KR20080098597A - 통증의 치료를 위한 벤조이소인돌 유도체 - Google Patents
통증의 치료를 위한 벤조이소인돌 유도체 Download PDFInfo
- Publication number
- KR20080098597A KR20080098597A KR1020087018902A KR20087018902A KR20080098597A KR 20080098597 A KR20080098597 A KR 20080098597A KR 1020087018902 A KR1020087018902 A KR 1020087018902A KR 20087018902 A KR20087018902 A KR 20087018902A KR 20080098597 A KR20080098597 A KR 20080098597A
- Authority
- KR
- South Korea
- Prior art keywords
- oxy
- difluoromethyl
- benzo
- dihydro
- acetic acid
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Ceased
Links
- CPFIEQURHGECRC-UHFFFAOYSA-N CCOC(c(c(C(OCC)=O)c(c1c2cccc1)OCc1ccccc1)c2OCc1ccccc1)=O Chemical compound CCOC(c(c(C(OCC)=O)c(c1c2cccc1)OCc1ccccc1)c2OCc1ccccc1)=O CPFIEQURHGECRC-UHFFFAOYSA-N 0.000 description 1
Classifications
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D209/00—Heterocyclic compounds containing five-membered rings, condensed with other rings, with one nitrogen atom as the only ring hetero atom
- C07D209/56—Ring systems containing three or more rings
- C07D209/58—[b]- or [c]-condensed
- C07D209/62—Naphtho [c] pyrroles; Hydrogenated naphtho [c] pyrroles
- C07D209/64—Naphtho [c] pyrroles; Hydrogenated naphtho [c] pyrroles with an oxygen atom in position 1
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K31/00—Medicinal preparations containing organic active ingredients
- A61K31/33—Heterocyclic compounds
- A61K31/395—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins
- A61K31/40—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins having five-membered rings with one nitrogen as the only ring hetero atom, e.g. sulpiride, succinimide, tolmetin, buflomedil
- A61K31/403—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins having five-membered rings with one nitrogen as the only ring hetero atom, e.g. sulpiride, succinimide, tolmetin, buflomedil condensed with carbocyclic rings, e.g. carbazole
- A61K31/4035—Isoindoles, e.g. phthalimide
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K45/00—Medicinal preparations containing active ingredients not provided for in groups A61K31/00 - A61K41/00
- A61K45/06—Mixtures of active ingredients without chemical characterisation, e.g. antiphlogistics and cardiaca
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P1/00—Drugs for disorders of the alimentary tract or the digestive system
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P1/00—Drugs for disorders of the alimentary tract or the digestive system
- A61P1/04—Drugs for disorders of the alimentary tract or the digestive system for ulcers, gastritis or reflux esophagitis, e.g. antacids, inhibitors of acid secretion, mucosal protectants
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P1/00—Drugs for disorders of the alimentary tract or the digestive system
- A61P1/10—Laxatives
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P1/00—Drugs for disorders of the alimentary tract or the digestive system
- A61P1/12—Antidiarrhoeals
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P1/00—Drugs for disorders of the alimentary tract or the digestive system
- A61P1/16—Drugs for disorders of the alimentary tract or the digestive system for liver or gallbladder disorders, e.g. hepatoprotective agents, cholagogues, litholytics
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P11/00—Drugs for disorders of the respiratory system
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P11/00—Drugs for disorders of the respiratory system
- A61P11/02—Nasal agents, e.g. decongestants
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P11/00—Drugs for disorders of the respiratory system
- A61P11/06—Antiasthmatics
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P13/00—Drugs for disorders of the urinary system
- A61P13/12—Drugs for disorders of the urinary system of the kidneys
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P17/00—Drugs for dermatological disorders
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P17/00—Drugs for dermatological disorders
- A61P17/02—Drugs for dermatological disorders for treating wounds, ulcers, burns, scars, keloids, or the like
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P17/00—Drugs for dermatological disorders
- A61P17/06—Antipsoriatics
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P19/00—Drugs for skeletal disorders
- A61P19/02—Drugs for skeletal disorders for joint disorders, e.g. arthritis, arthrosis
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P19/00—Drugs for skeletal disorders
- A61P19/06—Antigout agents, e.g. antihyperuricemic or uricosuric agents
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P19/00—Drugs for skeletal disorders
- A61P19/08—Drugs for skeletal disorders for bone diseases, e.g. rachitism, Paget's disease
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P25/00—Drugs for disorders of the nervous system
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P25/00—Drugs for disorders of the nervous system
- A61P25/02—Drugs for disorders of the nervous system for peripheral neuropathies
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P25/00—Drugs for disorders of the nervous system
- A61P25/04—Centrally acting analgesics, e.g. opioids
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P25/00—Drugs for disorders of the nervous system
- A61P25/06—Antimigraine agents
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P25/00—Drugs for disorders of the nervous system
- A61P25/14—Drugs for disorders of the nervous system for treating abnormal movements, e.g. chorea, dyskinesia
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P25/00—Drugs for disorders of the nervous system
- A61P25/14—Drugs for disorders of the nervous system for treating abnormal movements, e.g. chorea, dyskinesia
- A61P25/16—Anti-Parkinson drugs
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P25/00—Drugs for disorders of the nervous system
- A61P25/28—Drugs for disorders of the nervous system for treating neurodegenerative disorders of the central nervous system, e.g. nootropic agents, cognition enhancers, drugs for treating Alzheimer's disease or other forms of dementia
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P27/00—Drugs for disorders of the senses
- A61P27/02—Ophthalmic agents
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P27/00—Drugs for disorders of the senses
- A61P27/02—Ophthalmic agents
- A61P27/06—Antiglaucoma agents or miotics
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P29/00—Non-central analgesic, antipyretic or antiinflammatory agents, e.g. antirheumatic agents; Non-steroidal antiinflammatory drugs [NSAID]
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P37/00—Drugs for immunological or allergic disorders
- A61P37/08—Antiallergic agents
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P43/00—Drugs for specific purposes, not provided for in groups A61P1/00-A61P41/00
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P7/00—Drugs for disorders of the blood or the extracellular fluid
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P7/00—Drugs for disorders of the blood or the extracellular fluid
- A61P7/06—Antianaemics
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P9/00—Drugs for disorders of the cardiovascular system
- A61P9/10—Drugs for disorders of the cardiovascular system for treating ischaemic or atherosclerotic diseases, e.g. antianginal drugs, coronary vasodilators, drugs for myocardial infarction, retinopathy, cerebrovascula insufficiency, renal arteriosclerosis
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D209/00—Heterocyclic compounds containing five-membered rings, condensed with other rings, with one nitrogen atom as the only ring hetero atom
- C07D209/56—Ring systems containing three or more rings
- C07D209/58—[b]- or [c]-condensed
- C07D209/62—Naphtho [c] pyrroles; Hydrogenated naphtho [c] pyrroles
- C07D209/66—Naphtho [c] pyrroles; Hydrogenated naphtho [c] pyrroles with oxygen atoms in positions 1 and 3
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K2300/00—Mixtures or combinations of active ingredients, wherein at least one active ingredient is fully defined in groups A61K31/00 - A61K41/00
Landscapes
- Health & Medical Sciences (AREA)
- Chemical & Material Sciences (AREA)
- Organic Chemistry (AREA)
- Life Sciences & Earth Sciences (AREA)
- Engineering & Computer Science (AREA)
- Bioinformatics & Cheminformatics (AREA)
- Veterinary Medicine (AREA)
- Public Health (AREA)
- General Health & Medical Sciences (AREA)
- Medicinal Chemistry (AREA)
- Animal Behavior & Ethology (AREA)
- Pharmacology & Pharmacy (AREA)
- General Chemical & Material Sciences (AREA)
- Chemical Kinetics & Catalysis (AREA)
- Nuclear Medicine, Radiotherapy & Molecular Imaging (AREA)
- Biomedical Technology (AREA)
- Neurosurgery (AREA)
- Neurology (AREA)
- Pulmonology (AREA)
- Pain & Pain Management (AREA)
- Rheumatology (AREA)
- Physical Education & Sports Medicine (AREA)
- Dermatology (AREA)
- Ophthalmology & Optometry (AREA)
- Urology & Nephrology (AREA)
- Diabetes (AREA)
- Epidemiology (AREA)
- Psychology (AREA)
- Immunology (AREA)
- Hematology (AREA)
- Orthopedic Medicine & Surgery (AREA)
- Vascular Medicine (AREA)
- Otolaryngology (AREA)
- Psychiatry (AREA)
- Hospice & Palliative Care (AREA)
- Heart & Thoracic Surgery (AREA)
- Gastroenterology & Hepatology (AREA)
- Cardiology (AREA)
- Pharmaceuticals Containing Other Organic And Inorganic Compounds (AREA)
- Indole Compounds (AREA)
Abstract
Description
| 시간 / 분 | %B |
| 0 | 3 |
| 0.1 | 3 |
| 4 | 97 |
| 4.8 | 97 |
| 4.9 | 3 |
| 5.0 | 3 |
Claims (14)
- 제1항에 있어서, R1 및 R2 중 하나가 C1 -4 알킬을 나타내고, 나머지 하나가 디플루오로메틸을 나타내는 화학식 I의 화합물.
- 제1항에 있어서, R1 및 R2가 둘 다 디플루오로메틸인 화학식 I의 화합물.
- 제1항 내지 제3항 중 어느 한 항에 있어서, X 및 Y가 둘 다 C=O를 나타내는 화학식 I의 화합물.
- 제1항 내지 제4항 중 어느 한 항에 있어서, R3이 H, F 또는 Cl인 화학식 I의 화합물.
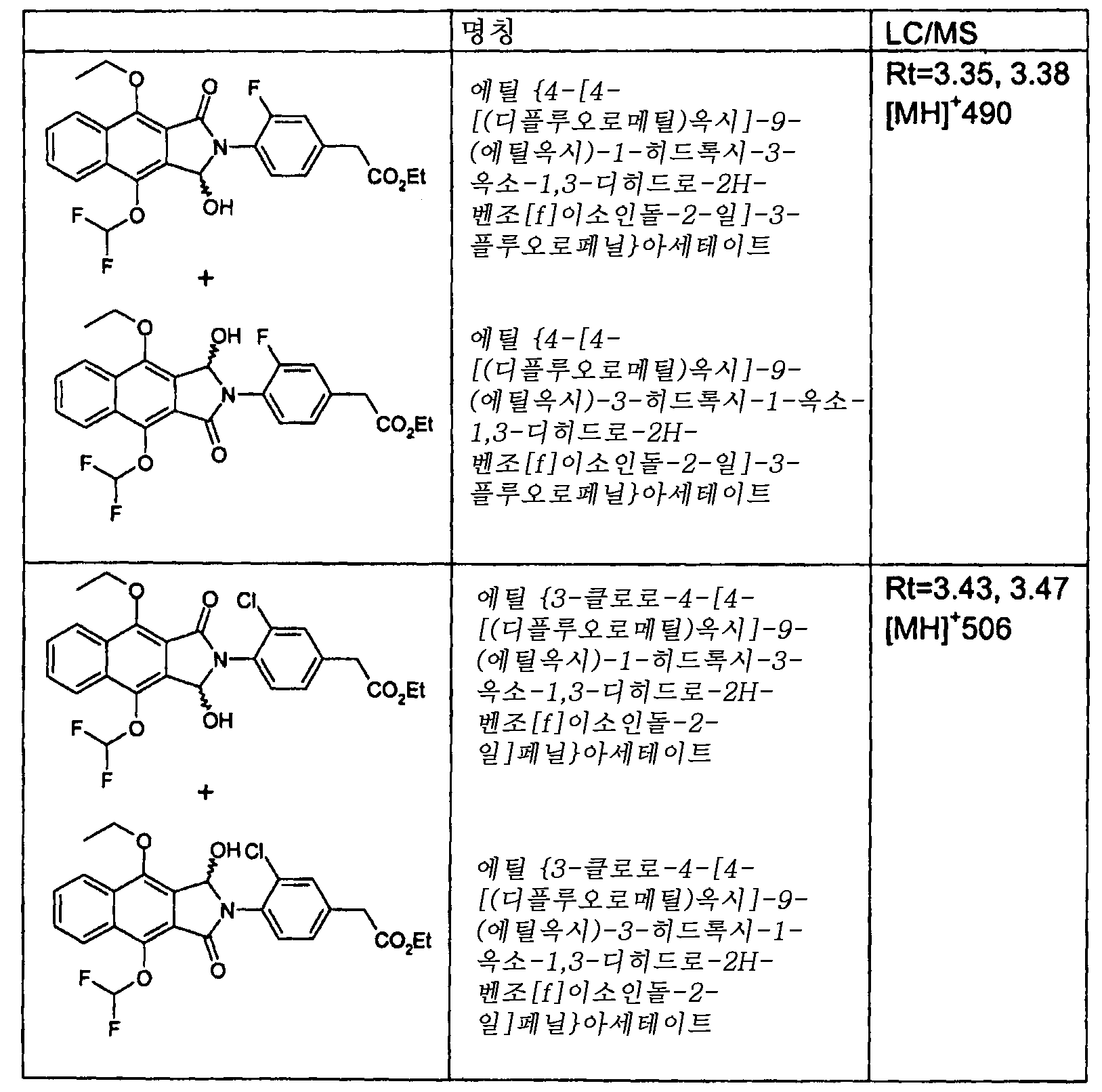
- 제1항에 있어서,(4-{4,9-비스[(디플루오로메틸)옥시]-1,3-디옥소-1,3-디히드로-2H-벤조[f]이소인돌-2-일}페닐)아세트산;(4-{4,9-비스[(디플루오로메틸)옥시]-1,3-디옥소-1,3-디히드로-2H-벤조[f]이소인돌-2-일}-3-클로로페닐)아세트산;(4-{4,9-비스[(디플루오로메틸)옥시]-1,3-디옥소-1,3-디히드로-2H-벤조[f]이소인돌-2-일}-3-플루오로페닐)아세트산;(4-{4,9-비스[(디플루오로메틸)옥시]-1,3-디옥소-1,3-디히드로-2H-벤조[f]이소인돌-2-일}-2-플루오로페닐)아세트산;(4-{4,9-비스[(디플루오로메틸)옥시]-1-옥소-1,3-디히드로-2H-벤조[f]이소인돌-2-일}-3-플루오로페닐)아세트산;(4-{4,9-비스[(디플루오로메틸)옥시]-1-옥소-1,3-디히드로-2H-벤조[f]이소인돌-2-일}-3-클로로페닐)아세트산;(4-{4,9-비스[(디플루오로메틸)옥시]-1-옥소-1,3-디히드로-2H-벤조[f]이소인돌-2-일}-2-플루오로페닐)아세트산;(4-{4-[(디플루오로메틸)옥시]-9-[(1-메틸에틸)옥시]-1,3-디옥소-1,3-디히드로-2H-벤조[f]이소인돌-2-일}페닐)아세트산;{4-[4-[(디플루오로메틸)옥시]-9-(에틸옥시)-1,3-디옥소-1,3-디히드로-2H-벤조[f]이소인돌-2-일]페닐}아세트산;{4-[4-[(디플루오로메틸)옥시]-9-(메틸옥시)-1,3-디옥소-1,3-디히드로-2H-벤조[f]이소인돌-2-일]페닐}아세트산;{4-[4-[(디플루오로메틸)옥시]-9-(에틸옥시)-1,3-디옥소-1,3-디히드로-2H-벤조[f]이소인돌-2-일]-3-플루오로페닐}아세트산;{3-클로로-4-[4-[(디플루오로메틸)옥시]-9-(에틸옥시)-1,3-디옥소-1,3-디히드로-2H-벤조[f]이소인돌-2-일]페닐}아세트산;{4-[4-[(디플루오로메틸)옥시]-9-(에틸옥시)-1,3-디옥소-1,3-디히드로-2H-벤조[f]이소인돌-2-일]-2-플루오로페닐}아세트산;{4-[4-[(디플루오로메틸)옥시]-1,3-디옥소-9-(프로필옥시)-1,3-디히드로-2H-벤조[f]이소인돌-2-일]-3-플루오로페닐}아세트산;{3-클로로-4-[4-[(디플루오로메틸)옥시]-1,3-디옥소-9-(프로필옥시)-1,3-디히드로-2H-벤조[f]이소인돌-2-일]페닐}아세트산;{4-[4-[(디플루오로메틸)옥시]-1,3-디옥소-9-(프로필옥시)-1,3-디히드로-2H-벤조[f]이소인돌-2-일]-2-플루오로페닐}아세트산;(4-{4-[(디플루오로메틸)옥시]-9-[(1-메틸에틸)옥시]-1,3-디옥소-1,3-디히드로-2H-벤조[f]이소인돌-2-일}-3-플루오로페닐)아세트산;(3-클로로-4-{4-[(디플루오로메틸)옥시]-9-[(1-메틸에틸)옥시]-1,3-디옥소-1,3-디히드로-2H-벤조[f]이소인돌-2-일}페닐)아세트산;(4-{4-[(디플루오로메틸)옥시]-9-[(1-메틸에틸)옥시]-1,3-디옥소-1,3-디히드로-2H-벤조[f]이소인돌-2-일}-2-플루오로페닐)아세트산;{3-클로로-4-[4-[(디플루오로메틸)옥시]-9-(에틸옥시)-1-옥소-1,3-디히드로-2H-벤조[f]이소인돌-2-일]페닐}아세트산;{3-클로로-4-[9-[(디플루오로메틸)옥시]-4-(에틸옥시)-1-옥소-1,3-디히드로-2H-벤조[f]이소인돌-2-일]페닐}아세트산;{4-[9-[(디플루오로메틸)옥시]-4-(메틸옥시)-1-옥소-1,3-디히드로-2H-벤조[f]이소인돌-2-일]페닐}아세트산;{4-[4-[(디플루오로메틸)옥시]-9-(메틸옥시)-1-옥소-1,3-디히드로-2H-벤조[f]이소인돌-2-일]페닐}아세트산;{4-[9-[(디플루오로메틸)옥시]-4-(에틸옥시)-1-옥소-1,3-디히드로-2H-벤조[f]이소인돌-2-일]-3-플루오로페닐}아세트산;{4-[4-[(디플루오로메틸)옥시]-9-(에틸옥시)-1-옥소-1,3-디히드로-2H-벤조[f]이소인돌-2-일]-3-플루오로페닐}아세트산;{4-[9-[(디플루오로메틸)옥시]-4-(에틸옥시)-1-옥소-1,3-디히드로-2H-벤조[f]이소인돌-2-일]-2-플루오로페닐}아세트산;4-[4-[(디플루오로메틸)옥시]-9-(에틸옥시)-1-옥소-1,3-디히드로-2H-벤조[f]이소인돌-2-일]-2-플루오로페닐}아세트산;{3-클로로-4-[4-[(디플루오로메틸)옥시]-1-옥소-9-(프로필옥시)-1,3-디히드로-2H-벤조[f]이소인돌-2-일]페닐}아세트산;{4-[9-[(디플루오로메틸)옥시]-1-옥소-4-(프로필옥시)-1,3-디히드로-2H-벤조[f]이소인돌-2-일]-3-플루오로페닐}아세트산;{4-[4-[(디플루오로메틸)옥시]-1-옥소-9-(프로필옥시)-1,3-디히드로-2H-벤조[f]이소인돌-2-일]-3-플루오로페닐}아세트산;{4-[9-[(디플루오로메틸)옥시]-1-옥소-4-(프로필옥시)-1,3-디히드로-2H-벤조[f]이소인돌-2-일]-2-플루오로페닐}아세트산;{4-[4-[(디플루오로메틸)옥시]-1-옥소-9-(프로필옥시)-1,3-디히드로-2H-벤조[f]이소인돌-2-일]-2-플루오로페닐}아세트산;(3-클로로-4-{9-[(디플루오로메틸)옥시]-4-[(1-메틸에틸)옥시]-1-옥소-1,3-디히드로-2H-벤조[f]이소인돌-2-일}페닐)아세트산;(3-클로로-4-{4-[(디플루오로메틸)옥시]-9-[(1-메틸에틸)옥시]-1-옥소-1,3-디히드로-2H-벤조[f]이소인돌-2-일}페닐)아세트산;(4-{9-[(디플루오로메틸)옥시]-4-[(1-메틸에틸)옥시]-1-옥소-1,3-디히드로-2H-벤조[f]이소인돌-2-일}-3-플루오로페닐)아세트산;(4-{4-[(디플루오로메틸)옥시]-9-[(1-메틸에틸)옥시]-1-옥소-1,3-디히드로-2H-벤조[f]이소인돌-2-일}-3-플루오로페닐)아세트산;(4-{9-[(디플루오로메틸)옥시]-4-[(1-메틸에틸)옥시]-1-옥소-1,3-디히드로-2H-벤조[f]이소인돌-2-일}-2-플루오로페닐)아세트산;(4-{4-[(디플루오로메틸)옥시]-9-[(1-메틸에틸)옥시]-1-옥소-1,3-디히드로-2H-벤조[f]이소인돌-2-일}-2-플루오로페닐)아세트산;또는 이들의 제약상 허용되는 유도체로 이루어진 군으로부터 선택된 화학식 I의 화합물.
- 하기 화학식 II의 화합물을 적합한 염기와 반응시키고, 임의로 그 후 생성된 화합물의 제약상 허용되는 유도체를 형성하고/거나, 화학식 I의 화합물을 또다른 것으로 전환시키는 단계를 포함하는, X 및 Y 중 하나가 C=O를 나타내고 나머지 하나가 CH2를 나타내고, R1, R2 및 R3이 제1항에 정의된 바와 같은 제1항에 따른 화학식 I의 화합물을 제조하는 방법.<화학식 II>식 중, X 및 Y 중 하나는 C=O를 나타내고, 나머지 하나는 CH2를 나타내고; R1, R2 및 R3은 제1항에 정의된 바와 같고; R4는 C1 -6 알킬을 나타낸다.
- 제1항에 있어서, 인간 의약 또는 수의약에 사용하기 위한 화학식 I의 화합물.
- 제1항에 있어서, EP4 수용체에서 PGE2의 작용 또는 작용의 상실에 의해 매개되는 증상의 치료에 사용하기 위한 화학식 I의 화합물.
- EP4 수용체에서 PGE2의 작용 또는 작용의 상실에 의해 매개되는 증상을 앓고 있는 인간 또는 동물 대상체에게 유효량의 제1항에 따른 화학식 I의 화합물을 투여하는 것을 포함하는, EP4 수용체에서 PGE2의 작용 또는 작용의 상실에 의해 매개되는 증상을 앓고 있는 인간 또는 동물 대상체를 치료하는 방법.
- EP4 수용체에서 PGE2의 작용에 의해 매개되는 증상의 치료용 의약을 제조하기 위한 제1항에 따른 화학식 I의 화합물의 용도.
- 제1항에 따른 화학식 I의 화합물, 및 그에 대한 하나 이상의 허용되는 담체 또는 희석제를 포함하는 제약 조성물.
- 제13항에 있어서, 하나 이상의 추가 치료제를 포함하는 제약 조성물.
Applications Claiming Priority (4)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| GB0602238.8 | 2006-02-03 | ||
| GB0602238A GB0602238D0 (en) | 2006-02-03 | 2006-02-03 | Novel compounds |
| GB0615094.0 | 2006-07-28 | ||
| GB0615094A GB0615094D0 (en) | 2006-07-28 | 2006-07-28 | Novel compounds |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| KR20080098597A true KR20080098597A (ko) | 2008-11-11 |
Family
ID=38042653
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| KR1020087018902A Ceased KR20080098597A (ko) | 2006-02-03 | 2007-02-01 | 통증의 치료를 위한 벤조이소인돌 유도체 |
Country Status (15)
| Country | Link |
|---|---|
| US (1) | US8207213B2 (ko) |
| EP (1) | EP1979317B1 (ko) |
| JP (1) | JP5118063B2 (ko) |
| KR (1) | KR20080098597A (ko) |
| AU (1) | AU2007211483B2 (ko) |
| BR (1) | BRPI0708025A2 (ko) |
| CA (1) | CA2640274A1 (ko) |
| CR (1) | CR10210A (ko) |
| EA (1) | EA014428B1 (ko) |
| ES (1) | ES2393800T3 (ko) |
| IL (1) | IL192933A0 (ko) |
| MA (1) | MA30308B1 (ko) |
| NO (1) | NO20083441L (ko) |
| SG (1) | SG169378A1 (ko) |
| WO (1) | WO2007088189A1 (ko) |
Families Citing this family (6)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US8110681B2 (en) | 2006-03-17 | 2012-02-07 | The United States Of America As Represented By The Secretary, Department Of Health And Human Services | Compounds for the treatment of spinal muscular atrophy and other uses |
| GB0615111D0 (en) * | 2006-07-28 | 2006-09-06 | Glaxo Group Ltd | Novel compounds |
| GB0620619D0 (en) * | 2006-10-17 | 2006-11-29 | Glaxo Group Ltd | Novel compounds |
| GB0623203D0 (en) * | 2006-11-21 | 2007-01-03 | Glaxo Group Ltd | Novel compounds |
| GB0721611D0 (en) | 2007-11-02 | 2007-12-12 | Glaxo Group Ltd | Novel compounds |
| EP2338485A1 (de) * | 2009-12-14 | 2011-06-29 | Grünenthal GmbH | Substituierte 1,3-Dioxoisoindoline als Arzneimittel |
Family Cites Families (4)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| GB0030541D0 (en) | 2000-12-14 | 2001-01-31 | Glaxo Group Ltd | Medical uses |
| GB0031295D0 (en) * | 2000-12-21 | 2001-01-31 | Glaxo Group Ltd | Naphthalene derivatives |
| GB0103269D0 (en) * | 2001-02-09 | 2001-03-28 | Glaxo Group Ltd | Napthalene derivatives |
| GB0615111D0 (en) * | 2006-07-28 | 2006-09-06 | Glaxo Group Ltd | Novel compounds |
-
2007
- 2007-02-01 EA EA200870223A patent/EA014428B1/ru not_active IP Right Cessation
- 2007-02-01 SG SG201100786-1A patent/SG169378A1/en unknown
- 2007-02-01 EP EP07704312A patent/EP1979317B1/en active Active
- 2007-02-01 ES ES07704312T patent/ES2393800T3/es active Active
- 2007-02-01 AU AU2007211483A patent/AU2007211483B2/en not_active Ceased
- 2007-02-01 CA CA002640274A patent/CA2640274A1/en not_active Abandoned
- 2007-02-01 JP JP2008552808A patent/JP5118063B2/ja not_active Expired - Fee Related
- 2007-02-01 WO PCT/EP2007/050991 patent/WO2007088189A1/en not_active Ceased
- 2007-02-01 BR BRPI0708025-5A patent/BRPI0708025A2/pt not_active IP Right Cessation
- 2007-02-01 US US12/278,013 patent/US8207213B2/en not_active Expired - Fee Related
- 2007-02-01 KR KR1020087018902A patent/KR20080098597A/ko not_active Ceased
-
2008
- 2008-07-21 IL IL192933A patent/IL192933A0/en unknown
- 2008-08-06 MA MA31160A patent/MA30308B1/fr unknown
- 2008-08-06 NO NO20083441A patent/NO20083441L/no not_active Application Discontinuation
- 2008-08-14 CR CR10210A patent/CR10210A/es not_active Application Discontinuation
Also Published As
| Publication number | Publication date |
|---|---|
| SG169378A1 (en) | 2011-03-30 |
| US8207213B2 (en) | 2012-06-26 |
| EA014428B1 (ru) | 2010-12-30 |
| JP5118063B2 (ja) | 2013-01-16 |
| EA200870223A1 (ru) | 2009-02-27 |
| NO20083441L (no) | 2008-09-30 |
| IL192933A0 (en) | 2009-02-11 |
| US20110092561A1 (en) | 2011-04-21 |
| MA30308B1 (fr) | 2009-04-01 |
| WO2007088189A1 (en) | 2007-08-09 |
| EP1979317A1 (en) | 2008-10-15 |
| ES2393800T3 (es) | 2012-12-28 |
| EP1979317B1 (en) | 2012-08-22 |
| AU2007211483B2 (en) | 2012-08-16 |
| CA2640274A1 (en) | 2007-08-09 |
| CR10210A (es) | 2008-10-29 |
| JP2010517925A (ja) | 2010-05-27 |
| BRPI0708025A2 (pt) | 2011-05-17 |
| AU2007211483A1 (en) | 2007-08-09 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| KR20090089868A (ko) | Ep4 수용체 작동제로서 벤즈아미드 유도체 | |
| JP5250427B2 (ja) | EP4受容体アゴニストとしてのベンゾ(f)イソインドール−2−イルフェニル酢酸誘導体 | |
| KR20080098597A (ko) | 통증의 치료를 위한 벤조이소인돌 유도체 | |
| US20100069457A1 (en) | Naphthalene Derivatives Used As EP4 Receptor Agonists | |
| US8026270B2 (en) | Benzo [F] isoindoles as EP4 receptor agonists | |
| JP2010517929A (ja) | ベンゾイソインドール誘導体およびep4受容体リガンドとしてのその使用 | |
| EP2081897B1 (en) | Isoindol derivatives as ep4 receptor agonists | |
| JP2010510282A (ja) | ベンゾインドール誘導体およびep4受容体アゴニストとしてのそれらの使用 | |
| CN101415680B (zh) | 用于治疗疼痛的苯并异吲哚衍生物 | |
| US20100273853A1 (en) | Novel isoindol derivatives as ep4 receptor agonists | |
| MX2008010050A (en) | Benzo (f) isoindol-2-ylphenyl acetic acid derivatives as ep4 receptor agonists |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| PA0105 | International application |
Patent event date: 20080731 Patent event code: PA01051R01D Comment text: International Patent Application |
|
| PG1501 | Laying open of application | ||
| A201 | Request for examination | ||
| PA0201 | Request for examination |
Patent event code: PA02012R01D Patent event date: 20120130 Comment text: Request for Examination of Application |
|
| E902 | Notification of reason for refusal | ||
| PE0902 | Notice of grounds for rejection |
Comment text: Notification of reason for refusal Patent event date: 20130822 Patent event code: PE09021S01D |
|
| E601 | Decision to refuse application | ||
| PE0601 | Decision on rejection of patent |
Patent event date: 20131129 Comment text: Decision to Refuse Application Patent event code: PE06012S01D Patent event date: 20130822 Comment text: Notification of reason for refusal Patent event code: PE06011S01I |