JP6505235B2 - リポソームを用いるアミロイド斑のmriイメージング - Google Patents
リポソームを用いるアミロイド斑のmriイメージング Download PDFInfo
- Publication number
- JP6505235B2 JP6505235B2 JP2017538918A JP2017538918A JP6505235B2 JP 6505235 B2 JP6505235 B2 JP 6505235B2 JP 2017538918 A JP2017538918 A JP 2017538918A JP 2017538918 A JP2017538918 A JP 2017538918A JP 6505235 B2 JP6505235 B2 JP 6505235B2
- Authority
- JP
- Japan
- Prior art keywords
- phospholipid
- polymer
- aromatic
- liposome composition
- substituted
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Active
Links
- 239000002502 liposome Substances 0.000 title claims description 118
- 208000037259 Amyloid Plaque Diseases 0.000 title claims description 66
- 238000003384 imaging method Methods 0.000 title claims description 23
- 239000000203 mixture Substances 0.000 claims description 86
- 125000003118 aryl group Chemical group 0.000 claims description 69
- 239000002245 particle Substances 0.000 claims description 54
- 229910052739 hydrogen Inorganic materials 0.000 claims description 40
- 239000001257 hydrogen Substances 0.000 claims description 40
- 239000012528 membrane Substances 0.000 claims description 36
- 238000002595 magnetic resonance imaging Methods 0.000 claims description 35
- 229920000642 polymer Polymers 0.000 claims description 26
- 239000003795 chemical substances by application Substances 0.000 claims description 25
- 150000003904 phospholipids Chemical class 0.000 claims description 25
- 238000001514 detection method Methods 0.000 claims description 22
- 125000004432 carbon atom Chemical group C* 0.000 claims description 20
- HVYWMOMLDIMFJA-DPAQBDIFSA-N cholesterol Chemical compound C1C=C2C[C@@H](O)CC[C@]2(C)[C@@H]2[C@@H]1[C@@H]1CC[C@H]([C@H](C)CCCC(C)C)[C@@]1(C)CC2 HVYWMOMLDIMFJA-DPAQBDIFSA-N 0.000 claims description 18
- 208000024827 Alzheimer disease Diseases 0.000 claims description 13
- 230000002708 enhancing effect Effects 0.000 claims description 13
- 230000002285 radioactive effect Effects 0.000 claims description 13
- 125000002947 alkylene group Chemical group 0.000 claims description 12
- KILNVBDSWZSGLL-KXQOOQHDSA-N 1,2-dihexadecanoyl-sn-glycero-3-phosphocholine Chemical group CCCCCCCCCCCCCCCC(=O)OC[C@H](COP([O-])(=O)OCC[N+](C)(C)C)OC(=O)CCCCCCCCCCCCCCC KILNVBDSWZSGLL-KXQOOQHDSA-N 0.000 claims description 11
- 125000005647 linker group Chemical group 0.000 claims description 11
- 235000012000 cholesterol Nutrition 0.000 claims description 9
- 125000004435 hydrogen atom Chemical class [H]* 0.000 claims description 9
- 210000002682 neurofibrillary tangle Anatomy 0.000 claims description 9
- CZPWVGJYEJSRLH-UHFFFAOYSA-N Pyrimidine Chemical compound C1=CN=CN=C1 CZPWVGJYEJSRLH-UHFFFAOYSA-N 0.000 claims description 8
- 125000000956 methoxy group Chemical group [H]C([H])([H])O* 0.000 claims description 7
- 229920001223 polyethylene glycol Polymers 0.000 claims description 7
- 238000004458 analytical method Methods 0.000 claims description 6
- QPCDCPDFJACHGM-UHFFFAOYSA-N N,N-bis{2-[bis(carboxymethyl)amino]ethyl}glycine Chemical compound OC(=O)CN(CC(O)=O)CCN(CC(=O)O)CCN(CC(O)=O)CC(O)=O QPCDCPDFJACHGM-UHFFFAOYSA-N 0.000 claims description 4
- 239000002202 Polyethylene glycol Substances 0.000 claims description 4
- 229920001477 hydrophilic polymer Polymers 0.000 claims description 4
- 229960003330 pentetic acid Drugs 0.000 claims description 4
- LYRFLYHAGKPMFH-UHFFFAOYSA-N octadecanamide Chemical compound CCCCCCCCCCCCCCCCCC(N)=O LYRFLYHAGKPMFH-UHFFFAOYSA-N 0.000 claims description 3
- 230000004044 response Effects 0.000 claims description 3
- 150000000921 Gadolinium Chemical class 0.000 claims 2
- 150000001875 compounds Chemical class 0.000 description 190
- 239000000562 conjugate Substances 0.000 description 74
- -1 C 1 -C 6 alkyl Chemical group 0.000 description 60
- 230000027455 binding Effects 0.000 description 56
- 241000699670 Mus sp. Species 0.000 description 55
- 241000699666 Mus <mouse, genus> Species 0.000 description 39
- 125000000217 alkyl group Chemical group 0.000 description 39
- 125000001072 heteroaryl group Chemical group 0.000 description 36
- 239000003446 ligand Substances 0.000 description 36
- 210000004556 brain Anatomy 0.000 description 28
- 238000000034 method Methods 0.000 description 25
- XEKOWRVHYACXOJ-UHFFFAOYSA-N Ethyl acetate Chemical compound CCOC(C)=O XEKOWRVHYACXOJ-UHFFFAOYSA-N 0.000 description 24
- 150000002431 hydrogen Chemical class 0.000 description 24
- 125000002887 hydroxy group Chemical group [H]O* 0.000 description 24
- UFHFLCQGNIYNRP-UHFFFAOYSA-N Hydrogen Chemical compound [H][H] UFHFLCQGNIYNRP-UHFFFAOYSA-N 0.000 description 23
- 125000002950 monocyclic group Chemical group 0.000 description 23
- OKKJLVBELUTLKV-UHFFFAOYSA-N Methanol Chemical compound OC OKKJLVBELUTLKV-UHFFFAOYSA-N 0.000 description 21
- 229910052736 halogen Inorganic materials 0.000 description 20
- 150000002367 halogens Chemical class 0.000 description 20
- ZMXDDKWLCZADIW-UHFFFAOYSA-N N,N-Dimethylformamide Chemical compound CN(C)C=O ZMXDDKWLCZADIW-UHFFFAOYSA-N 0.000 description 18
- MOFVSTNWEDAEEK-UHFFFAOYSA-M indocyanine green Chemical group [Na+].[O-]S(=O)(=O)CCCCN1C2=CC=C3C=CC=CC3=C2C(C)(C)C1=CC=CC=CC=CC1=[N+](CCCCS([O-])(=O)=O)C2=CC=C(C=CC=C3)C3=C2C1(C)C MOFVSTNWEDAEEK-UHFFFAOYSA-M 0.000 description 18
- 229960004657 indocyanine green Drugs 0.000 description 18
- VLKZOEOYAKHREP-UHFFFAOYSA-N n-Hexane Chemical compound CCCCCC VLKZOEOYAKHREP-UHFFFAOYSA-N 0.000 description 18
- LFQSCWFLJHTTHZ-UHFFFAOYSA-N Ethanol Chemical compound CCO LFQSCWFLJHTTHZ-UHFFFAOYSA-N 0.000 description 17
- 239000000243 solution Substances 0.000 description 17
- 125000003367 polycyclic group Chemical group 0.000 description 16
- 238000005481 NMR spectroscopy Methods 0.000 description 15
- 230000008499 blood brain barrier function Effects 0.000 description 15
- 210000001218 blood-brain barrier Anatomy 0.000 description 15
- 125000001424 substituent group Chemical group 0.000 description 15
- XFFSCOOTVXBLCK-QAVVBOBSSA-N OC(=O)c1cc(ccc1O)\N=N\c1ccc(cc1)-c1ccc(cc1)\N=N\c1ccc(O)c(c1)C(O)=O Chemical compound OC(=O)c1cc(ccc1O)\N=N\c1ccc(cc1)-c1ccc(cc1)\N=N\c1ccc(O)c(c1)C(O)=O XFFSCOOTVXBLCK-QAVVBOBSSA-N 0.000 description 14
- 239000013522 chelant Substances 0.000 description 13
- 230000000875 corresponding effect Effects 0.000 description 13
- JUJWROOIHBZHMG-UHFFFAOYSA-N Pyridine Chemical group C1=CC=NC=C1 JUJWROOIHBZHMG-UHFFFAOYSA-N 0.000 description 12
- 125000003342 alkenyl group Chemical group 0.000 description 12
- 125000000623 heterocyclic group Chemical group 0.000 description 12
- MVJKUWANBPLVFJ-GORDUTHDSA-N 2-[N-methyl-4-[(E)-2-pyrimidin-4-ylethenyl]anilino]ethanol Chemical compound CN(CCO)C1=CC=C(C=C1)\C=C\C1=NC=NC=C1 MVJKUWANBPLVFJ-GORDUTHDSA-N 0.000 description 11
- 241001465754 Metazoa Species 0.000 description 11
- 230000001965 increasing effect Effects 0.000 description 11
- 238000005259 measurement Methods 0.000 description 11
- 239000002953 phosphate buffered saline Substances 0.000 description 11
- 238000002360 preparation method Methods 0.000 description 11
- 230000008685 targeting Effects 0.000 description 11
- 125000003178 carboxy group Chemical group [H]OC(*)=O 0.000 description 10
- 150000003839 salts Chemical class 0.000 description 10
- 125000000472 sulfonyl group Chemical group *S(*)(=O)=O 0.000 description 10
- CBRMVHHBOZCVFN-DUXPYHPUSA-N 2-methoxy-4-[(E)-2-pyrimidin-4-ylethenyl]phenol Chemical compound COC1=C(C=CC(=C1)\C=C\C1=NC=NC=C1)O CBRMVHHBOZCVFN-DUXPYHPUSA-N 0.000 description 9
- 125000003917 carbamoyl group Chemical group [H]N([H])C(*)=O 0.000 description 9
- 125000006577 C1-C6 hydroxyalkyl group Chemical group 0.000 description 8
- HEDRZPFGACZZDS-UHFFFAOYSA-N Chloroform Chemical compound ClC(Cl)Cl HEDRZPFGACZZDS-UHFFFAOYSA-N 0.000 description 8
- IOJUPLGTWVMSFF-UHFFFAOYSA-N benzothiazole Chemical compound C1=CC=C2SC=NC2=C1 IOJUPLGTWVMSFF-UHFFFAOYSA-N 0.000 description 8
- 238000006243 chemical reaction Methods 0.000 description 8
- 230000013595 glycosylation Effects 0.000 description 8
- 238000006206 glycosylation reaction Methods 0.000 description 8
- 210000001320 hippocampus Anatomy 0.000 description 8
- 239000012216 imaging agent Substances 0.000 description 8
- 230000007170 pathology Effects 0.000 description 8
- 238000012360 testing method Methods 0.000 description 8
- 238000012879 PET imaging Methods 0.000 description 7
- 230000015572 biosynthetic process Effects 0.000 description 7
- 238000002347 injection Methods 0.000 description 7
- 239000007924 injection Substances 0.000 description 7
- 229920000233 poly(alkylene oxides) Polymers 0.000 description 7
- 230000002829 reductive effect Effects 0.000 description 7
- 230000009261 transgenic effect Effects 0.000 description 7
- XLYOFNOQVPJJNP-UHFFFAOYSA-N water Substances O XLYOFNOQVPJJNP-UHFFFAOYSA-N 0.000 description 7
- CSCPPACGZOOCGX-UHFFFAOYSA-N Acetone Chemical compound CC(C)=O CSCPPACGZOOCGX-UHFFFAOYSA-N 0.000 description 6
- 241000282472 Canis lupus familiaris Species 0.000 description 6
- PEDCQBHIVMGVHV-UHFFFAOYSA-N Glycerine Chemical compound OCC(O)CO PEDCQBHIVMGVHV-UHFFFAOYSA-N 0.000 description 6
- PXIPVTKHYLBLMZ-UHFFFAOYSA-N Sodium azide Chemical compound [Na+].[N-]=[N+]=[N-] PXIPVTKHYLBLMZ-UHFFFAOYSA-N 0.000 description 6
- 125000003710 aryl alkyl group Chemical group 0.000 description 6
- 229910052799 carbon Inorganic materials 0.000 description 6
- 238000005314 correlation function Methods 0.000 description 6
- 238000009472 formulation Methods 0.000 description 6
- 125000005842 heteroatom Chemical group 0.000 description 6
- 150000002632 lipids Chemical class 0.000 description 6
- 230000004807 localization Effects 0.000 description 6
- 230000005291 magnetic effect Effects 0.000 description 6
- 238000010172 mouse model Methods 0.000 description 6
- 238000002600 positron emission tomography Methods 0.000 description 6
- UMJSCPRVCHMLSP-UHFFFAOYSA-N pyridine Natural products COC1=CC=CN=C1 UMJSCPRVCHMLSP-UHFFFAOYSA-N 0.000 description 6
- 238000010898 silica gel chromatography Methods 0.000 description 6
- 238000010186 staining Methods 0.000 description 6
- 239000011550 stock solution Substances 0.000 description 6
- 102000001049 Amyloid Human genes 0.000 description 5
- 108010094108 Amyloid Proteins 0.000 description 5
- 241000283690 Bos taurus Species 0.000 description 5
- 0 CCCN(CC*)c1ccc(C=C[C@](CC(OC)=N2)N=C2OC)cc1 Chemical compound CCCN(CC*)c1ccc(C=C[C@](CC(OC)=N2)N=C2OC)cc1 0.000 description 5
- YLQBMQCUIZJEEH-UHFFFAOYSA-N Furan Chemical group C=1C=COC=1 YLQBMQCUIZJEEH-UHFFFAOYSA-N 0.000 description 5
- SIKJAQJRHWYJAI-UHFFFAOYSA-N Indole Chemical compound C1=CC=C2NC=CC2=C1 SIKJAQJRHWYJAI-UHFFFAOYSA-N 0.000 description 5
- VYPSYNLAJGMNEJ-UHFFFAOYSA-N Silicium dioxide Chemical compound O=[Si]=O VYPSYNLAJGMNEJ-UHFFFAOYSA-N 0.000 description 5
- 210000005013 brain tissue Anatomy 0.000 description 5
- 210000003710 cerebral cortex Anatomy 0.000 description 5
- 230000009137 competitive binding Effects 0.000 description 5
- 230000021615 conjugation Effects 0.000 description 5
- 125000004122 cyclic group Chemical group 0.000 description 5
- 125000000753 cycloalkyl group Chemical group 0.000 description 5
- 238000001125 extrusion Methods 0.000 description 5
- 229910052731 fluorine Inorganic materials 0.000 description 5
- 238000003364 immunohistochemistry Methods 0.000 description 5
- 239000002105 nanoparticle Substances 0.000 description 5
- 229910052760 oxygen Inorganic materials 0.000 description 5
- 125000001997 phenyl group Chemical group [H]C1=C([H])C([H])=C(*)C([H])=C1[H] 0.000 description 5
- 238000000159 protein binding assay Methods 0.000 description 5
- 125000006413 ring segment Chemical group 0.000 description 5
- SRLOHQKOADWDBV-NRONOFSHSA-M sodium;[(2r)-2,3-di(octadecanoyloxy)propyl] 2-(2-methoxyethoxycarbonylamino)ethyl phosphate Chemical compound [Na+].CCCCCCCCCCCCCCCCCC(=O)OC[C@H](COP([O-])(=O)OCCNC(=O)OCCOC)OC(=O)CCCCCCCCCCCCCCCCC SRLOHQKOADWDBV-NRONOFSHSA-M 0.000 description 5
- 239000000126 substance Substances 0.000 description 5
- 125000005346 substituted cycloalkyl group Chemical group 0.000 description 5
- KBPLFHHGFOOTCA-UHFFFAOYSA-N 1-Octanol Chemical compound CCCCCCCCO KBPLFHHGFOOTCA-UHFFFAOYSA-N 0.000 description 4
- 229940126062 Compound A Drugs 0.000 description 4
- IAZDPXIOMUYVGZ-UHFFFAOYSA-N Dimethylsulphoxide Chemical compound CS(C)=O IAZDPXIOMUYVGZ-UHFFFAOYSA-N 0.000 description 4
- WSFSSNUMVMOOMR-UHFFFAOYSA-N Formaldehyde Chemical compound O=C WSFSSNUMVMOOMR-UHFFFAOYSA-N 0.000 description 4
- NLDMNSXOCDLTTB-UHFFFAOYSA-N Heterophylliin A Natural products O1C2COC(=O)C3=CC(O)=C(O)C(O)=C3C3=C(O)C(O)=C(O)C=C3C(=O)OC2C(OC(=O)C=2C=C(O)C(O)=C(O)C=2)C(O)C1OC(=O)C1=CC(O)=C(O)C(O)=C1 NLDMNSXOCDLTTB-UHFFFAOYSA-N 0.000 description 4
- 241000282412 Homo Species 0.000 description 4
- KYQCOXFCLRTKLS-UHFFFAOYSA-N Pyrazine Chemical group C1=CN=CC=N1 KYQCOXFCLRTKLS-UHFFFAOYSA-N 0.000 description 4
- KAESVJOAVNADME-UHFFFAOYSA-N Pyrrole Chemical group C=1C=CNC=1 KAESVJOAVNADME-UHFFFAOYSA-N 0.000 description 4
- WYURNTSHIVDZCO-UHFFFAOYSA-N Tetrahydrofuran Chemical compound C1CCOC1 WYURNTSHIVDZCO-UHFFFAOYSA-N 0.000 description 4
- YTPLMLYBLZKORZ-UHFFFAOYSA-N Thiophene Chemical group C=1C=CSC=1 YTPLMLYBLZKORZ-UHFFFAOYSA-N 0.000 description 4
- 125000003545 alkoxy group Chemical group 0.000 description 4
- 150000001412 amines Chemical class 0.000 description 4
- 150000001540 azides Chemical class 0.000 description 4
- 230000008901 benefit Effects 0.000 description 4
- 125000002619 bicyclic group Chemical group 0.000 description 4
- 229910052794 bromium Inorganic materials 0.000 description 4
- 239000000460 chlorine Substances 0.000 description 4
- 229910052801 chlorine Inorganic materials 0.000 description 4
- 230000001419 dependent effect Effects 0.000 description 4
- 238000002073 fluorescence micrograph Methods 0.000 description 4
- 210000005153 frontal cortex Anatomy 0.000 description 4
- 125000001041 indolyl group Chemical group 0.000 description 4
- 238000002372 labelling Methods 0.000 description 4
- 238000001554 negative-stained transmission electron micrograph Methods 0.000 description 4
- 239000002904 solvent Substances 0.000 description 4
- 238000003786 synthesis reaction Methods 0.000 description 4
- 210000001519 tissue Anatomy 0.000 description 4
- 150000003852 triazoles Chemical group 0.000 description 4
- LVNGJLRDBYCPGB-LDLOPFEMSA-N (R)-1,2-distearoylphosphatidylethanolamine Chemical compound CCCCCCCCCCCCCCCCCC(=O)OC[C@H](COP([O-])(=O)OCC[NH3+])OC(=O)CCCCCCCCCCCCCCCCC LVNGJLRDBYCPGB-LDLOPFEMSA-N 0.000 description 3
- 102000013455 Amyloid beta-Peptides Human genes 0.000 description 3
- 108010090849 Amyloid beta-Peptides Proteins 0.000 description 3
- OKTJSMMVPCPJKN-UHFFFAOYSA-N Carbon Chemical compound [C] OKTJSMMVPCPJKN-UHFFFAOYSA-N 0.000 description 3
- PXGOKWXKJXAPGV-UHFFFAOYSA-N Fluorine Chemical compound FF PXGOKWXKJXAPGV-UHFFFAOYSA-N 0.000 description 3
- ZRALSGWEFCBTJO-UHFFFAOYSA-N Guanidine Chemical compound NC(N)=N ZRALSGWEFCBTJO-UHFFFAOYSA-N 0.000 description 3
- 101000617536 Homo sapiens Presenilin-1 Proteins 0.000 description 3
- PIWKPBJCKXDKJR-UHFFFAOYSA-N Isoflurane Chemical compound FC(F)OC(Cl)C(F)(F)F PIWKPBJCKXDKJR-UHFFFAOYSA-N 0.000 description 3
- 239000000232 Lipid Bilayer Substances 0.000 description 3
- 102100022033 Presenilin-1 Human genes 0.000 description 3
- YXFVVABEGXRONW-UHFFFAOYSA-N Toluene Chemical compound CC1=CC=CC=C1 YXFVVABEGXRONW-UHFFFAOYSA-N 0.000 description 3
- 230000003941 amyloidogenesis Effects 0.000 description 3
- 125000004429 atom Chemical group 0.000 description 3
- 238000011888 autopsy Methods 0.000 description 3
- 125000001164 benzothiazolyl group Chemical group S1C(=NC2=C1C=CC=C2)* 0.000 description 3
- 125000003354 benzotriazolyl group Chemical group N1N=NC2=C1C=CC=C2* 0.000 description 3
- 239000000872 buffer Substances 0.000 description 3
- 238000010226 confocal imaging Methods 0.000 description 3
- 230000008021 deposition Effects 0.000 description 3
- 238000003745 diagnosis Methods 0.000 description 3
- 238000000502 dialysis Methods 0.000 description 3
- 238000009826 distribution Methods 0.000 description 3
- 229940079593 drug Drugs 0.000 description 3
- 239000003814 drug Substances 0.000 description 3
- 239000011737 fluorine Substances 0.000 description 3
- 125000005843 halogen group Chemical group 0.000 description 3
- 125000004475 heteroaralkyl group Chemical group 0.000 description 3
- 230000002209 hydrophobic effect Effects 0.000 description 3
- 125000002768 hydroxyalkyl group Chemical group 0.000 description 3
- 125000002883 imidazolyl group Chemical group 0.000 description 3
- PZOUSPYUWWUPPK-UHFFFAOYSA-N indole Natural products CC1=CC=CC2=C1C=CN2 PZOUSPYUWWUPPK-UHFFFAOYSA-N 0.000 description 3
- RKJUIXBNRJVNHR-UHFFFAOYSA-N indolenine Natural products C1=CC=C2CC=NC2=C1 RKJUIXBNRJVNHR-UHFFFAOYSA-N 0.000 description 3
- 229910052740 iodine Inorganic materials 0.000 description 3
- 229960002725 isoflurane Drugs 0.000 description 3
- 230000007774 longterm Effects 0.000 description 3
- 239000012046 mixed solvent Substances 0.000 description 3
- 125000001624 naphthyl group Chemical group 0.000 description 3
- 238000005192 partition Methods 0.000 description 3
- LPNYRYFBWFDTMA-UHFFFAOYSA-N potassium tert-butoxide Chemical compound [K+].CC(C)(C)[O-] LPNYRYFBWFDTMA-UHFFFAOYSA-N 0.000 description 3
- 125000004076 pyridyl group Chemical group 0.000 description 3
- 125000000714 pyrimidinyl group Chemical group 0.000 description 3
- 230000005855 radiation Effects 0.000 description 3
- 239000011541 reaction mixture Substances 0.000 description 3
- 238000001228 spectrum Methods 0.000 description 3
- 238000000264 spin echo pulse sequence Methods 0.000 description 3
- 229910052717 sulfur Inorganic materials 0.000 description 3
- 239000000725 suspension Substances 0.000 description 3
- 238000012800 visualization Methods 0.000 description 3
- NRJAVPSFFCBXDT-HUESYALOSA-N 1,2-distearoyl-sn-glycero-3-phosphocholine Chemical compound CCCCCCCCCCCCCCCCCC(=O)OC[C@H](COP([O-])(=O)OCC[N+](C)(C)C)OC(=O)CCCCCCCCCCCCCCCCC NRJAVPSFFCBXDT-HUESYALOSA-N 0.000 description 2
- FCEHBMOGCRZNNI-UHFFFAOYSA-N 1-benzothiophene Chemical compound C1=CC=C2SC=CC2=C1 FCEHBMOGCRZNNI-UHFFFAOYSA-N 0.000 description 2
- TXLHNFOLHRXMAU-UHFFFAOYSA-N 2-(4-benzylphenoxy)-n,n-diethylethanamine;hydron;chloride Chemical compound Cl.C1=CC(OCCN(CC)CC)=CC=C1CC1=CC=CC=C1 TXLHNFOLHRXMAU-UHFFFAOYSA-N 0.000 description 2
- QKNYBSVHEMOAJP-UHFFFAOYSA-N 2-amino-2-(hydroxymethyl)propane-1,3-diol;hydron;chloride Chemical compound Cl.OCC(N)(CO)CO QKNYBSVHEMOAJP-UHFFFAOYSA-N 0.000 description 2
- YSDDPNWGLSGZRC-UHFFFAOYSA-N 4-[bis(2-hydroxyethyl)amino]benzaldehyde Chemical compound OCCN(CCO)C1=CC=C(C=O)C=C1 YSDDPNWGLSGZRC-UHFFFAOYSA-N 0.000 description 2
- LVILGAOSPDLNRM-UHFFFAOYSA-N 4-methylpyrimidine Chemical compound CC1=CC=NC=N1 LVILGAOSPDLNRM-UHFFFAOYSA-N 0.000 description 2
- YYROPELSRYBVMQ-UHFFFAOYSA-N 4-toluenesulfonyl chloride Chemical compound CC1=CC=C(S(Cl)(=O)=O)C=C1 YYROPELSRYBVMQ-UHFFFAOYSA-N 0.000 description 2
- 206010002091 Anaesthesia Diseases 0.000 description 2
- IJGRMHOSHXDMSA-UHFFFAOYSA-N Atomic nitrogen Chemical compound N#N IJGRMHOSHXDMSA-UHFFFAOYSA-N 0.000 description 2
- WKBOTKDWSSQWDR-UHFFFAOYSA-N Bromine atom Chemical compound [Br] WKBOTKDWSSQWDR-UHFFFAOYSA-N 0.000 description 2
- 125000000882 C2-C6 alkenyl group Chemical group 0.000 description 2
- 208000005145 Cerebral amyloid angiopathy Diseases 0.000 description 2
- ZAMOUSCENKQFHK-UHFFFAOYSA-N Chlorine atom Chemical compound [Cl] ZAMOUSCENKQFHK-UHFFFAOYSA-N 0.000 description 2
- IAZDPXIOMUYVGZ-WFGJKAKNSA-N Dimethyl sulfoxide Chemical compound [2H]C([2H])([2H])S(=O)C([2H])([2H])[2H] IAZDPXIOMUYVGZ-WFGJKAKNSA-N 0.000 description 2
- 229910052688 Gadolinium Inorganic materials 0.000 description 2
- OAKJQQAXSVQMHS-UHFFFAOYSA-N Hydrazine Chemical compound NN OAKJQQAXSVQMHS-UHFFFAOYSA-N 0.000 description 2
- 239000007987 MES buffer Substances 0.000 description 2
- 241000699660 Mus musculus Species 0.000 description 2
- CHJJGSNFBQVOTG-UHFFFAOYSA-N N-methyl-guanidine Natural products CNC(N)=N CHJJGSNFBQVOTG-UHFFFAOYSA-N 0.000 description 2
- 238000011887 Necropsy Methods 0.000 description 2
- ZCQWOFVYLHDMMC-UHFFFAOYSA-N Oxazole Chemical group C1=COC=N1 ZCQWOFVYLHDMMC-UHFFFAOYSA-N 0.000 description 2
- PCNDJXKNXGMECE-UHFFFAOYSA-N Phenazine Chemical group C1=CC=CC2=NC3=CC=CC=C3N=C21 PCNDJXKNXGMECE-UHFFFAOYSA-N 0.000 description 2
- 229920003171 Poly (ethylene oxide) Polymers 0.000 description 2
- WTKZEGDFNFYCGP-UHFFFAOYSA-N Pyrazole Chemical group C=1C=NNC=1 WTKZEGDFNFYCGP-UHFFFAOYSA-N 0.000 description 2
- FZWLAAWBMGSTSO-UHFFFAOYSA-N Thiazole Chemical group C1=CSC=N1 FZWLAAWBMGSTSO-UHFFFAOYSA-N 0.000 description 2
- XSQUKJJJFZCRTK-UHFFFAOYSA-N Urea Chemical compound NC(N)=O XSQUKJJJFZCRTK-UHFFFAOYSA-N 0.000 description 2
- 238000010521 absorption reaction Methods 0.000 description 2
- 125000003282 alkyl amino group Chemical group 0.000 description 2
- 125000000304 alkynyl group Chemical group 0.000 description 2
- 150000001408 amides Chemical class 0.000 description 2
- 230000037005 anaesthesia Effects 0.000 description 2
- 238000013459 approach Methods 0.000 description 2
- 125000001769 aryl amino group Chemical group 0.000 description 2
- 125000005334 azaindolyl group Chemical group N1N=C(C2=CC=CC=C12)* 0.000 description 2
- 125000003785 benzimidazolyl group Chemical group N1=C(NC2=C1C=CC=C2)* 0.000 description 2
- 125000000499 benzofuranyl group Chemical group O1C(=CC2=C1C=CC=C2)* 0.000 description 2
- 125000005874 benzothiadiazolyl group Chemical group 0.000 description 2
- 125000004196 benzothienyl group Chemical group S1C(=CC2=C1C=CC=C2)* 0.000 description 2
- 125000004541 benzoxazolyl group Chemical group O1C(=NC2=C1C=CC=C2)* 0.000 description 2
- 210000004204 blood vessel Anatomy 0.000 description 2
- GDTBXPJZTBHREO-UHFFFAOYSA-N bromine Substances BrBr GDTBXPJZTBHREO-UHFFFAOYSA-N 0.000 description 2
- 150000001721 carbon Chemical group 0.000 description 2
- 239000003054 catalyst Substances 0.000 description 2
- 210000001175 cerebrospinal fluid Anatomy 0.000 description 2
- 208000010877 cognitive disease Diseases 0.000 description 2
- 239000012141 concentrate Substances 0.000 description 2
- 239000002872 contrast media Substances 0.000 description 2
- OPQARKPSCNTWTJ-UHFFFAOYSA-L copper(ii) acetate Chemical compound [Cu+2].CC([O-])=O.CC([O-])=O OPQARKPSCNTWTJ-UHFFFAOYSA-L 0.000 description 2
- 230000001054 cortical effect Effects 0.000 description 2
- 125000000113 cyclohexyl group Chemical group [H]C1([H])C([H])([H])C([H])([H])C([H])(*)C([H])([H])C1([H])[H] 0.000 description 2
- 238000011026 diafiltration Methods 0.000 description 2
- 238000002405 diagnostic procedure Methods 0.000 description 2
- 125000004925 dihydropyridyl group Chemical group N1(CC=CC=C1)* 0.000 description 2
- SWSQBOPZIKWTGO-UHFFFAOYSA-N dimethylaminoamidine Natural products CN(C)C(N)=N SWSQBOPZIKWTGO-UHFFFAOYSA-N 0.000 description 2
- ZUOUZKKEUPVFJK-UHFFFAOYSA-N diphenyl Chemical compound C1=CC=CC=C1C1=CC=CC=C1 ZUOUZKKEUPVFJK-UHFFFAOYSA-N 0.000 description 2
- LOKCTEFSRHRXRJ-UHFFFAOYSA-I dipotassium trisodium dihydrogen phosphate hydrogen phosphate dichloride Chemical compound P(=O)(O)(O)[O-].[K+].P(=O)(O)([O-])[O-].[Na+].[Na+].[Cl-].[K+].[Cl-].[Na+] LOKCTEFSRHRXRJ-UHFFFAOYSA-I 0.000 description 2
- 230000009977 dual effect Effects 0.000 description 2
- 239000000428 dust Substances 0.000 description 2
- 238000001493 electron microscopy Methods 0.000 description 2
- 238000005516 engineering process Methods 0.000 description 2
- 230000005284 excitation Effects 0.000 description 2
- 239000000835 fiber Substances 0.000 description 2
- MHMNJMPURVTYEJ-UHFFFAOYSA-N fluorescein-5-isothiocyanate Chemical compound O1C(=O)C2=CC(N=C=S)=CC=C2C21C1=CC=C(O)C=C1OC1=CC(O)=CC=C21 MHMNJMPURVTYEJ-UHFFFAOYSA-N 0.000 description 2
- 238000000799 fluorescence microscopy Methods 0.000 description 2
- 230000006870 function Effects 0.000 description 2
- 125000002541 furyl group Chemical group 0.000 description 2
- MXZROTBGJUUXID-UHFFFAOYSA-K gadobenic acid Chemical compound [H+].[H+].[Gd+3].[O-]C(=O)CN(CC([O-])=O)CCN(CC(=O)[O-])CCN(CC([O-])=O)C(C([O-])=O)COCC1=CC=CC=C1 MXZROTBGJUUXID-UHFFFAOYSA-K 0.000 description 2
- UIWYJDYFSGRHKR-UHFFFAOYSA-N gadolinium atom Chemical compound [Gd] UIWYJDYFSGRHKR-UHFFFAOYSA-N 0.000 description 2
- 150000002314 glycerols Chemical class 0.000 description 2
- 239000013029 homogenous suspension Substances 0.000 description 2
- 125000002632 imidazolidinyl group Chemical group 0.000 description 2
- 125000004857 imidazopyridinyl group Chemical group N1C(=NC2=C1C=CC=N2)* 0.000 description 2
- 238000000338 in vitro Methods 0.000 description 2
- 238000011534 incubation Methods 0.000 description 2
- 125000003392 indanyl group Chemical group C1(CCC2=CC=CC=C12)* 0.000 description 2
- 125000003453 indazolyl group Chemical group N1N=C(C2=C1C=CC=C2)* 0.000 description 2
- 230000003993 interaction Effects 0.000 description 2
- OKJPEAGHQZHRQV-UHFFFAOYSA-N iodoform Chemical compound IC(I)I OKJPEAGHQZHRQV-UHFFFAOYSA-N 0.000 description 2
- 125000002183 isoquinolinyl group Chemical group C1(=NC=CC2=CC=CC=C12)* 0.000 description 2
- 239000010410 layer Substances 0.000 description 2
- 238000004949 mass spectrometry Methods 0.000 description 2
- 239000011159 matrix material Substances 0.000 description 2
- 125000002496 methyl group Chemical group [H]C([H])([H])* 0.000 description 2
- 230000004048 modification Effects 0.000 description 2
- 238000012986 modification Methods 0.000 description 2
- 125000002757 morpholinyl group Chemical group 0.000 description 2
- 239000012120 mounting media Substances 0.000 description 2
- 238000003333 near-infrared imaging Methods 0.000 description 2
- 125000001971 neopentyl group Chemical group [H]C([*])([H])C(C([H])([H])[H])(C([H])([H])[H])C([H])([H])[H] 0.000 description 2
- WCPAKWJPBJAGKN-UHFFFAOYSA-N oxadiazole Chemical group C1=CON=N1 WCPAKWJPBJAGKN-UHFFFAOYSA-N 0.000 description 2
- 125000002971 oxazolyl group Chemical group 0.000 description 2
- 125000004043 oxo group Chemical group O=* 0.000 description 2
- VATYWCRQDJIRAI-UHFFFAOYSA-N p-aminobenzaldehyde Chemical compound NC1=CC=C(C=O)C=C1 VATYWCRQDJIRAI-UHFFFAOYSA-N 0.000 description 2
- 230000035699 permeability Effects 0.000 description 2
- 239000000546 pharmaceutical excipient Substances 0.000 description 2
- 125000004193 piperazinyl group Chemical group 0.000 description 2
- 239000004417 polycarbonate Substances 0.000 description 2
- 229920000515 polycarbonate Polymers 0.000 description 2
- 229920001451 polypropylene glycol Polymers 0.000 description 2
- 239000000843 powder Substances 0.000 description 2
- 150000003138 primary alcohols Chemical class 0.000 description 2
- LVTJOONKWUXEFR-FZRMHRINSA-N protoneodioscin Natural products O(C[C@@H](CC[C@]1(O)[C@H](C)[C@@H]2[C@]3(C)[C@H]([C@H]4[C@@H]([C@]5(C)C(=CC4)C[C@@H](O[C@@H]4[C@H](O[C@H]6[C@@H](O)[C@@H](O)[C@@H](O)[C@H](C)O6)[C@@H](O)[C@H](O[C@H]6[C@@H](O)[C@@H](O)[C@@H](O)[C@H](C)O6)[C@H](CO)O4)CC5)CC3)C[C@@H]2O1)C)[C@H]1[C@H](O)[C@H](O)[C@H](O)[C@@H](CO)O1 LVTJOONKWUXEFR-FZRMHRINSA-N 0.000 description 2
- 238000000746 purification Methods 0.000 description 2
- 125000000561 purinyl group Chemical group N1=C(N=C2N=CNC2=C1)* 0.000 description 2
- 125000003373 pyrazinyl group Chemical group 0.000 description 2
- 125000003226 pyrazolyl group Chemical group 0.000 description 2
- PBMFSQRYOILNGV-UHFFFAOYSA-N pyridazine Chemical group C1=CC=NN=C1 PBMFSQRYOILNGV-UHFFFAOYSA-N 0.000 description 2
- 125000002098 pyridazinyl group Chemical group 0.000 description 2
- 125000000168 pyrrolyl group Chemical group 0.000 description 2
- 125000002943 quinolinyl group Chemical group N1=C(C=CC2=CC=CC=C12)* 0.000 description 2
- 238000002603 single-photon emission computed tomography Methods 0.000 description 2
- 241000894007 species Species 0.000 description 2
- 230000000087 stabilizing effect Effects 0.000 description 2
- PJANXHGTPQOBST-UHFFFAOYSA-N stilbene Chemical group C=1C=CC=CC=1C=CC1=CC=CC=C1 PJANXHGTPQOBST-UHFFFAOYSA-N 0.000 description 2
- 125000003107 substituted aryl group Chemical group 0.000 description 2
- 125000000147 tetrahydroquinolinyl group Chemical group N1(CCCC2=CC=CC=C12)* 0.000 description 2
- 125000003507 tetrahydrothiofenyl group Chemical group 0.000 description 2
- 125000003831 tetrazolyl group Chemical group 0.000 description 2
- 210000001103 thalamus Anatomy 0.000 description 2
- VLLMWSRANPNYQX-UHFFFAOYSA-N thiadiazole Chemical group C1=CSN=N1.C1=CSN=N1 VLLMWSRANPNYQX-UHFFFAOYSA-N 0.000 description 2
- 125000000335 thiazolyl group Chemical group 0.000 description 2
- RBNBDIMXFJYDLQ-UHFFFAOYSA-N thieno[3,2-d]pyrimidine Chemical compound C1=NC=C2SC=CC2=N1 RBNBDIMXFJYDLQ-UHFFFAOYSA-N 0.000 description 2
- 125000001544 thienyl group Chemical group 0.000 description 2
- 238000004809 thin layer chromatography Methods 0.000 description 2
- 229930192474 thiophene Chemical group 0.000 description 2
- JOXIMZWYDAKGHI-UHFFFAOYSA-N toluene-4-sulfonic acid Chemical compound CC1=CC=C(S(O)(=O)=O)C=C1 JOXIMZWYDAKGHI-UHFFFAOYSA-N 0.000 description 2
- 238000011830 transgenic mouse model Methods 0.000 description 2
- 125000001425 triazolyl group Chemical group 0.000 description 2
- YUOLAYZNDYRBEZ-UHFFFAOYSA-N (6-oxo-1h-pyridin-3-yl)boronic acid Chemical compound OB(O)C=1C=CC(=O)NC=1 YUOLAYZNDYRBEZ-UHFFFAOYSA-N 0.000 description 1
- FNQJDLTXOVEEFB-UHFFFAOYSA-N 1,2,3-benzothiadiazole Chemical compound C1=CC=C2SN=NC2=C1 FNQJDLTXOVEEFB-UHFFFAOYSA-N 0.000 description 1
- SLLFVLKNXABYGI-UHFFFAOYSA-N 1,2,3-benzoxadiazole Chemical compound C1=CC=C2ON=NC2=C1 SLLFVLKNXABYGI-UHFFFAOYSA-N 0.000 description 1
- SLKDGVPOSSLUAI-PGUFJCEWSA-N 1,2-dihexadecanoyl-sn-glycero-3-phosphoethanolamine zwitterion Chemical compound CCCCCCCCCCCCCCCC(=O)OC[C@H](COP(O)(=O)OCCN)OC(=O)CCCCCCCCCCCCCCC SLKDGVPOSSLUAI-PGUFJCEWSA-N 0.000 description 1
- BCMCBBGGLRIHSE-UHFFFAOYSA-N 1,3-benzoxazole Chemical compound C1=CC=C2OC=NC2=C1 BCMCBBGGLRIHSE-UHFFFAOYSA-N 0.000 description 1
- 125000001140 1,4-phenylene group Chemical group [H]C1=C([H])C([*:2])=C([H])C([H])=C1[*:1] 0.000 description 1
- YBYIRNPNPLQARY-UHFFFAOYSA-N 1H-indene Natural products C1=CC=C2CC=CC2=C1 YBYIRNPNPLQARY-UHFFFAOYSA-N 0.000 description 1
- UCOZSLOKWQWKJD-DAFODLJHSA-N 2-[N-(2-hydroxyethyl)-4-[(E)-2-pyrimidin-4-ylethenyl]anilino]ethanol Chemical compound N1=CN=C(C=C1)/C=C/C1=CC=C(C=C1)N(CCO)CCO UCOZSLOKWQWKJD-DAFODLJHSA-N 0.000 description 1
- LDLCZOVUSADOIV-UHFFFAOYSA-N 2-bromoethanol Chemical compound OCCBr LDLCZOVUSADOIV-UHFFFAOYSA-N 0.000 description 1
- GTZBELRIIUXTNQ-UHFFFAOYSA-N 2-hydroxyacetaldehyde Chemical compound OCC=O.OCC=O GTZBELRIIUXTNQ-UHFFFAOYSA-N 0.000 description 1
- SDTMFDGELKWGFT-UHFFFAOYSA-N 2-methylpropan-2-olate Chemical compound CC(C)(C)[O-] SDTMFDGELKWGFT-UHFFFAOYSA-N 0.000 description 1
- 125000000094 2-phenylethyl group Chemical group [H]C1=C([H])C([H])=C(C([H])=C1[H])C([H])([H])C([H])([H])* 0.000 description 1
- AKXKFZDCRYJKTF-UHFFFAOYSA-N 3-Hydroxypropionaldehyde Chemical compound OCCC=O AKXKFZDCRYJKTF-UHFFFAOYSA-N 0.000 description 1
- FWBHETKCLVMNFS-UHFFFAOYSA-N 4',6-Diamino-2-phenylindol Chemical compound C1=CC(C(=N)N)=CC=C1C1=CC2=CC=C(C(N)=N)C=C2N1 FWBHETKCLVMNFS-UHFFFAOYSA-N 0.000 description 1
- JOCUIVLSLBBESN-UHFFFAOYSA-N 4-[2-hydroxyethyl(methyl)amino]benzaldehyde Chemical compound OCCN(C)C1=CC=C(C=O)C=C1 JOCUIVLSLBBESN-UHFFFAOYSA-N 0.000 description 1
- CNRGMQRNYAIBTN-UHFFFAOYSA-N 5-hydroxypentanal Chemical compound OCCCCC=O CNRGMQRNYAIBTN-UHFFFAOYSA-N 0.000 description 1
- FPFTWHJPEMPAGE-UHFFFAOYSA-N 6-hydroxy caproaldehyde Chemical compound OCCCCCC=O FPFTWHJPEMPAGE-UHFFFAOYSA-N 0.000 description 1
- ZCYVEMRRCGMTRW-UHFFFAOYSA-N 7553-56-2 Chemical compound [I] ZCYVEMRRCGMTRW-UHFFFAOYSA-N 0.000 description 1
- 239000005964 Acibenzolar-S-methyl Substances 0.000 description 1
- 208000000044 Amnesia Diseases 0.000 description 1
- KNFSHMMZCUZSAM-QPJJXVBHSA-N CCCN(C)c1ccc(/C=C/c2ccncn2)cc1 Chemical compound CCCN(C)c1ccc(/C=C/c2ccncn2)cc1 KNFSHMMZCUZSAM-QPJJXVBHSA-N 0.000 description 1
- DWGLXOPOUYOHSN-ZZXKWVIFSA-N CCCN(CCO)c1ccc(/C=C/c2ncncc2)cc1 Chemical compound CCCN(CCO)c1ccc(/C=C/c2ncncc2)cc1 DWGLXOPOUYOHSN-ZZXKWVIFSA-N 0.000 description 1
- UWJOMBQWSOLRIB-UHFFFAOYSA-N CCCOCCOCCNC(CC(C)(C)OCCC(C)(C)OC(NCCC(C)C)=O)=O Chemical compound CCCOCCOCCNC(CC(C)(C)OCCC(C)(C)OC(NCCC(C)C)=O)=O UWJOMBQWSOLRIB-UHFFFAOYSA-N 0.000 description 1
- 241000282465 Canis Species 0.000 description 1
- KXDHJXZQYSOELW-UHFFFAOYSA-M Carbamate Chemical compound NC([O-])=O KXDHJXZQYSOELW-UHFFFAOYSA-M 0.000 description 1
- KXDHJXZQYSOELW-UHFFFAOYSA-N Carbamic acid Chemical group NC(O)=O KXDHJXZQYSOELW-UHFFFAOYSA-N 0.000 description 1
- 208000028698 Cognitive impairment Diseases 0.000 description 1
- 206010012289 Dementia Diseases 0.000 description 1
- 241000283074 Equus asinus Species 0.000 description 1
- JOYRKODLDBILNP-UHFFFAOYSA-N Ethyl urethane Chemical compound CCOC(N)=O JOYRKODLDBILNP-UHFFFAOYSA-N 0.000 description 1
- KMTRUDSVKNLOMY-UHFFFAOYSA-N Ethylene carbonate Chemical compound O=C1OCCO1 KMTRUDSVKNLOMY-UHFFFAOYSA-N 0.000 description 1
- IAYPIBMASNFSPL-UHFFFAOYSA-N Ethylene oxide Chemical compound C1CO1 IAYPIBMASNFSPL-UHFFFAOYSA-N 0.000 description 1
- 206010015866 Extravasation Diseases 0.000 description 1
- CTKINSOISVBQLD-UHFFFAOYSA-N Glycidol Chemical compound OCC1CO1 CTKINSOISVBQLD-UHFFFAOYSA-N 0.000 description 1
- 208000016988 Hemorrhagic Stroke Diseases 0.000 description 1
- AVXURJPOCDRRFD-UHFFFAOYSA-N Hydroxylamine Chemical compound ON AVXURJPOCDRRFD-UHFFFAOYSA-N 0.000 description 1
- 239000002841 Lewis acid Substances 0.000 description 1
- 239000002616 MRI contrast agent Substances 0.000 description 1
- 208000026139 Memory disease Diseases 0.000 description 1
- AFVFQIVMOAPDHO-UHFFFAOYSA-N Methanesulfonic acid Chemical compound CS(O)(=O)=O AFVFQIVMOAPDHO-UHFFFAOYSA-N 0.000 description 1
- 150000001204 N-oxides Chemical class 0.000 description 1
- SNYRPCZQHPPTJQ-HNQUOIGGSA-N OCC[n]1c2ccc(/C=C/c3ncnc(O)c3)cc2cc1 Chemical compound OCC[n]1c2ccc(/C=C/c3ncnc(O)c3)cc2cc1 SNYRPCZQHPPTJQ-HNQUOIGGSA-N 0.000 description 1
- OAICVXFJPJFONN-UHFFFAOYSA-N Phosphorus Chemical compound [P] OAICVXFJPJFONN-UHFFFAOYSA-N 0.000 description 1
- ZLMJMSJWJFRBEC-UHFFFAOYSA-N Potassium Chemical compound [K] ZLMJMSJWJFRBEC-UHFFFAOYSA-N 0.000 description 1
- 206010036790 Productive cough Diseases 0.000 description 1
- FAPWRFPIFSIZLT-UHFFFAOYSA-M Sodium chloride Chemical compound [Na+].[Cl-] FAPWRFPIFSIZLT-UHFFFAOYSA-M 0.000 description 1
- 229930182558 Sterol Natural products 0.000 description 1
- 229930006000 Sucrose Natural products 0.000 description 1
- CZMRCDWAGMRECN-UGDNZRGBSA-N Sucrose Chemical compound O[C@H]1[C@H](O)[C@@H](CO)O[C@@]1(CO)O[C@@H]1[C@H](O)[C@@H](O)[C@H](O)[C@@H](CO)O1 CZMRCDWAGMRECN-UGDNZRGBSA-N 0.000 description 1
- UCKMPCXJQFINFW-UHFFFAOYSA-N Sulphide Chemical compound [S-2] UCKMPCXJQFINFW-UHFFFAOYSA-N 0.000 description 1
- COQLPRJCUIATTQ-UHFFFAOYSA-N Uranyl acetate Chemical compound O.O.O=[U]=O.CC(O)=O.CC(O)=O COQLPRJCUIATTQ-UHFFFAOYSA-N 0.000 description 1
- BZHJMEDXRYGGRV-UHFFFAOYSA-N Vinyl chloride Chemical group ClC=C BZHJMEDXRYGGRV-UHFFFAOYSA-N 0.000 description 1
- DCKQGTRHDBMYRO-UHFFFAOYSA-N [Gd+3].C(CCCCCCCCCCCCCCCCC)[NH-].C(CCCCCCCCCCCCCCCCC)[NH-].C(CCCCCCCCCCCCCCCCC)[NH-] Chemical compound [Gd+3].C(CCCCCCCCCCCCCCCCC)[NH-].C(CCCCCCCCCCCCCCCCC)[NH-].C(CCCCCCCCCCCCCCCCC)[NH-] DCKQGTRHDBMYRO-UHFFFAOYSA-N 0.000 description 1
- MXZROTBGJUUXID-UHFFFAOYSA-I [Gd+3].[O-]C(=O)CN(CC([O-])=O)CCN(CC(=O)[O-])CCN(CC([O-])=O)C(C([O-])=O)COCC1=CC=CC=C1 Chemical compound [Gd+3].[O-]C(=O)CN(CC([O-])=O)CCN(CC(=O)[O-])CCN(CC([O-])=O)C(C([O-])=O)COCC1=CC=CC=C1 MXZROTBGJUUXID-UHFFFAOYSA-I 0.000 description 1
- 239000002253 acid Substances 0.000 description 1
- 230000009056 active transport Effects 0.000 description 1
- 125000005073 adamantyl group Chemical group C12(CC3CC(CC(C1)C3)C2)* 0.000 description 1
- 125000005426 adeninyl group Chemical group N1=C(N=C2N=CNC2=C1N)* 0.000 description 1
- 125000004183 alkoxy alkyl group Chemical group 0.000 description 1
- 125000005262 alkoxyamine group Chemical group 0.000 description 1
- 125000000278 alkyl amino alkyl group Chemical group 0.000 description 1
- 125000004947 alkyl aryl amino group Chemical group 0.000 description 1
- 230000029936 alkylation Effects 0.000 description 1
- 238000005804 alkylation reaction Methods 0.000 description 1
- VREFGVBLTWBCJP-UHFFFAOYSA-N alprazolam Chemical compound C12=CC(Cl)=CC=C2N2C(C)=NN=C2CN=C1C1=CC=CC=C1 VREFGVBLTWBCJP-UHFFFAOYSA-N 0.000 description 1
- 125000003368 amide group Chemical group 0.000 description 1
- 150000001409 amidines Chemical class 0.000 description 1
- 125000004103 aminoalkyl group Chemical group 0.000 description 1
- 150000001448 anilines Chemical class 0.000 description 1
- 125000002178 anthracenyl group Chemical group C1(=CC=CC2=CC3=CC=CC=C3C=C12)* 0.000 description 1
- 239000012736 aqueous medium Substances 0.000 description 1
- 125000002102 aryl alkyloxo group Chemical group 0.000 description 1
- 125000000732 arylene group Chemical group 0.000 description 1
- 125000004104 aryloxy group Chemical group 0.000 description 1
- 238000003556 assay Methods 0.000 description 1
- 125000005602 azabenzimidazolyl group Chemical group 0.000 description 1
- 125000002393 azetidinyl group Chemical group 0.000 description 1
- 125000004069 aziridinyl group Chemical group 0.000 description 1
- 125000003828 azulenyl group Chemical group 0.000 description 1
- RFRXIWQYSOIBDI-UHFFFAOYSA-N benzarone Chemical compound CCC=1OC2=CC=CC=C2C=1C(=O)C1=CC=C(O)C=C1 RFRXIWQYSOIBDI-UHFFFAOYSA-N 0.000 description 1
- 125000005605 benzo group Chemical group 0.000 description 1
- 125000004622 benzoxazinyl group Chemical group O1NC(=CC2=C1C=CC=C2)* 0.000 description 1
- 125000001797 benzyl group Chemical group [H]C1=C([H])C([H])=C(C([H])=C1[H])C([H])([H])* 0.000 description 1
- 125000000440 benzylamino group Chemical group [H]N(*)C([H])([H])C1=C([H])C([H])=C([H])C([H])=C1[H] 0.000 description 1
- JSMRMEYFZHIPJV-UHFFFAOYSA-N bicyclo[2.1.1]hexane Chemical group C1C2CC1CC2 JSMRMEYFZHIPJV-UHFFFAOYSA-N 0.000 description 1
- 235000010290 biphenyl Nutrition 0.000 description 1
- 239000004305 biphenyl Substances 0.000 description 1
- 239000008280 blood Substances 0.000 description 1
- 210000004369 blood Anatomy 0.000 description 1
- 230000037396 body weight Effects 0.000 description 1
- 239000012267 brine Substances 0.000 description 1
- FJDQFPXHSGXQBY-UHFFFAOYSA-L caesium carbonate Chemical compound [Cs+].[Cs+].[O-]C([O-])=O FJDQFPXHSGXQBY-UHFFFAOYSA-L 0.000 description 1
- 229910000024 caesium carbonate Inorganic materials 0.000 description 1
- 150000004657 carbamic acid derivatives Chemical class 0.000 description 1
- 239000004202 carbamide Substances 0.000 description 1
- 125000002837 carbocyclic group Chemical group 0.000 description 1
- 125000002915 carbonyl group Chemical group [*:2]C([*:1])=O 0.000 description 1
- 125000004181 carboxyalkyl group Chemical group 0.000 description 1
- 239000003153 chemical reaction reagent Substances 0.000 description 1
- 125000002603 chloroethyl group Chemical group [H]C([*])([H])C([H])([H])Cl 0.000 description 1
- XBWRJSSJWDOUSJ-UHFFFAOYSA-L chromium(ii) chloride Chemical compound Cl[Cr]Cl XBWRJSSJWDOUSJ-UHFFFAOYSA-L 0.000 description 1
- 125000000259 cinnolinyl group Chemical group N1=NC(=CC2=CC=CC=C12)* 0.000 description 1
- 230000008045 co-localization Effects 0.000 description 1
- 238000004440 column chromatography Methods 0.000 description 1
- 238000002591 computed tomography Methods 0.000 description 1
- 238000012790 confirmation Methods 0.000 description 1
- IQFVPQOLBLOTPF-HKXUKFGYSA-L congo red Chemical compound [Na+].[Na+].C1=CC=CC2=C(N)C(/N=N/C3=CC=C(C=C3)C3=CC=C(C=C3)/N=N/C3=C(C4=CC=CC=C4C(=C3)S([O-])(=O)=O)N)=CC(S([O-])(=O)=O)=C21 IQFVPQOLBLOTPF-HKXUKFGYSA-L 0.000 description 1
- 238000011109 contamination Methods 0.000 description 1
- 230000002596 correlated effect Effects 0.000 description 1
- 230000008878 coupling Effects 0.000 description 1
- 238000010168 coupling process Methods 0.000 description 1
- 238000005859 coupling reaction Methods 0.000 description 1
- 150000001913 cyanates Chemical class 0.000 description 1
- 125000000000 cycloalkoxy group Chemical group 0.000 description 1
- 125000004367 cycloalkylaryl group Chemical group 0.000 description 1
- 125000001995 cyclobutyl group Chemical group [H]C1([H])C([H])([H])C([H])(*)C1([H])[H] 0.000 description 1
- 125000000582 cycloheptyl group Chemical group [H]C1([H])C([H])([H])C([H])([H])C([H])([H])C([H])(*)C([H])([H])C1([H])[H] 0.000 description 1
- 125000000640 cyclooctyl group Chemical group [H]C1([H])C([H])([H])C([H])([H])C([H])([H])C([H])(*)C([H])([H])C([H])([H])C1([H])[H] 0.000 description 1
- 125000001511 cyclopentyl group Chemical group [H]C1([H])C([H])([H])C([H])([H])C([H])(*)C1([H])[H] 0.000 description 1
- 125000001559 cyclopropyl group Chemical group [H]C1([H])C([H])([H])C1([H])* 0.000 description 1
- 125000004855 decalinyl group Chemical group C1(CCCC2CCCCC12)* 0.000 description 1
- 238000010511 deprotection reaction Methods 0.000 description 1
- 230000001687 destabilization Effects 0.000 description 1
- 238000011161 development Methods 0.000 description 1
- 125000004985 dialkyl amino alkyl group Chemical group 0.000 description 1
- 125000004663 dialkyl amino group Chemical group 0.000 description 1
- FHIVAFMUCKRCQO-UHFFFAOYSA-N diazinon Chemical compound CCOP(=S)(OCC)OC1=CC(C)=NC(C(C)C)=N1 FHIVAFMUCKRCQO-UHFFFAOYSA-N 0.000 description 1
- 235000005911 diet Nutrition 0.000 description 1
- 230000000378 dietary effect Effects 0.000 description 1
- 235000014113 dietary fatty acids Nutrition 0.000 description 1
- 125000000723 dihydrobenzofuranyl group Chemical group O1C(CC2=C1C=CC=C2)* 0.000 description 1
- 125000001070 dihydroindolyl group Chemical group N1(CCC2=CC=CC=C12)* 0.000 description 1
- IJKVHSBPTUYDLN-UHFFFAOYSA-N dihydroxy(oxo)silane Chemical compound O[Si](O)=O IJKVHSBPTUYDLN-UHFFFAOYSA-N 0.000 description 1
- 238000007865 diluting Methods 0.000 description 1
- KPUWHANPEXNPJT-UHFFFAOYSA-N disiloxane Chemical compound [SiH3]O[SiH3] KPUWHANPEXNPJT-UHFFFAOYSA-N 0.000 description 1
- 125000005883 dithianyl group Chemical group 0.000 description 1
- 229960003722 doxycycline Drugs 0.000 description 1
- XQTWDDCIUJNLTR-CVHRZJFOSA-N doxycycline monohydrate Chemical compound O.O=C1C2=C(O)C=CC=C2[C@H](C)[C@@H]2C1=C(O)[C@]1(O)C(=O)C(C(N)=O)=C(O)[C@@H](N(C)C)[C@@H]1[C@H]2O XQTWDDCIUJNLTR-CVHRZJFOSA-N 0.000 description 1
- 238000013535 dynamic contrast enhanced MRI Methods 0.000 description 1
- 238000002296 dynamic light scattering Methods 0.000 description 1
- 230000000694 effects Effects 0.000 description 1
- 238000004993 emission spectroscopy Methods 0.000 description 1
- 238000005538 encapsulation Methods 0.000 description 1
- 239000003623 enhancer Substances 0.000 description 1
- 150000002148 esters Chemical class 0.000 description 1
- 125000001495 ethyl group Chemical group [H]C([H])([H])C([H])([H])* 0.000 description 1
- 238000002474 experimental method Methods 0.000 description 1
- 230000036251 extravasation Effects 0.000 description 1
- 229930195729 fatty acid Natural products 0.000 description 1
- 239000000194 fatty acid Substances 0.000 description 1
- 150000004665 fatty acids Chemical class 0.000 description 1
- 238000007667 floating Methods 0.000 description 1
- YNDIAUKFXKEXSV-CRYLGTRXSA-N florbetapir F-18 Chemical compound C1=CC(NC)=CC=C1\C=C\C1=CC=C(OCCOCCOCC[18F])N=C1 YNDIAUKFXKEXSV-CRYLGTRXSA-N 0.000 description 1
- 125000003983 fluorenyl group Chemical group C1(=CC=CC=2C3=CC=CC=C3CC12)* 0.000 description 1
- 238000007306 functionalization reaction Methods 0.000 description 1
- 230000004927 fusion Effects 0.000 description 1
- 125000003147 glycosyl group Chemical group 0.000 description 1
- 150000004820 halides Chemical class 0.000 description 1
- 125000001188 haloalkyl group Chemical group 0.000 description 1
- 230000026030 halogenation Effects 0.000 description 1
- 238000005658 halogenation reaction Methods 0.000 description 1
- 230000036541 health Effects 0.000 description 1
- 125000002192 heptalenyl group Chemical group 0.000 description 1
- 125000005549 heteroarylene group Chemical group 0.000 description 1
- 125000004415 heterocyclylalkyl group Chemical group 0.000 description 1
- 125000005844 heterocyclyloxy group Chemical group 0.000 description 1
- 230000000971 hippocampal effect Effects 0.000 description 1
- 238000010562 histological examination Methods 0.000 description 1
- 238000007489 histopathology method Methods 0.000 description 1
- 150000007857 hydrazones Chemical class 0.000 description 1
- 150000002433 hydrophilic molecules Chemical class 0.000 description 1
- 125000002636 imidazolinyl group Chemical group 0.000 description 1
- 125000005945 imidazopyridyl group Chemical group 0.000 description 1
- 150000003949 imides Chemical class 0.000 description 1
- 150000002466 imines Chemical class 0.000 description 1
- 230000002055 immunohistochemical effect Effects 0.000 description 1
- 230000001771 impaired effect Effects 0.000 description 1
- 238000001727 in vivo Methods 0.000 description 1
- 238000011503 in vivo imaging Methods 0.000 description 1
- 125000003454 indenyl group Chemical group C1(C=CC2=CC=CC=C12)* 0.000 description 1
- 125000003387 indolinyl group Chemical group N1(CCC2=CC=CC=C12)* 0.000 description 1
- 125000003406 indolizinyl group Chemical group C=1(C=CN2C=CC=CC12)* 0.000 description 1
- 238000009616 inductively coupled plasma Methods 0.000 description 1
- 230000003834 intracellular effect Effects 0.000 description 1
- 208000020658 intracerebral hemorrhage Diseases 0.000 description 1
- 238000010253 intravenous injection Methods 0.000 description 1
- 230000026045 iodination Effects 0.000 description 1
- 238000006192 iodination reaction Methods 0.000 description 1
- 239000011630 iodine Substances 0.000 description 1
- GHXZPUGJZVBLGC-UHFFFAOYSA-N iodoethene Chemical compound IC=C GHXZPUGJZVBLGC-UHFFFAOYSA-N 0.000 description 1
- 125000000959 isobutyl group Chemical group [H]C([H])([H])C([H])(C([H])([H])[H])C([H])([H])* 0.000 description 1
- 239000012948 isocyanate Substances 0.000 description 1
- 150000002513 isocyanates Chemical class 0.000 description 1
- 125000000904 isoindolyl group Chemical group C=1(NC=C2C=CC=CC12)* 0.000 description 1
- 125000001972 isopentyl group Chemical group [H]C([H])([H])C([H])(C([H])([H])[H])C([H])([H])C([H])([H])* 0.000 description 1
- 125000001449 isopropyl group Chemical group [H]C([H])([H])C([H])(*)C([H])([H])[H] 0.000 description 1
- 125000001786 isothiazolyl group Chemical group 0.000 description 1
- 150000002540 isothiocyanates Chemical class 0.000 description 1
- 125000000842 isoxazolyl group Chemical group 0.000 description 1
- 150000007517 lewis acids Chemical class 0.000 description 1
- 206010025135 lupus erythematosus Diseases 0.000 description 1
- 230000014759 maintenance of location Effects 0.000 description 1
- 238000004519 manufacturing process Methods 0.000 description 1
- 239000003550 marker Substances 0.000 description 1
- 238000001819 mass spectrum Methods 0.000 description 1
- 239000000463 material Substances 0.000 description 1
- 238000000816 matrix-assisted laser desorption--ionisation Methods 0.000 description 1
- 230000007246 mechanism Effects 0.000 description 1
- 230000006984 memory degeneration Effects 0.000 description 1
- 208000023060 memory loss Diseases 0.000 description 1
- 238000003328 mesylation reaction Methods 0.000 description 1
- OKKJLVBELUTLKV-VMNATFBRSA-N methanol-d1 Chemical compound [2H]OC OKKJLVBELUTLKV-VMNATFBRSA-N 0.000 description 1
- 238000000386 microscopy Methods 0.000 description 1
- 125000004108 n-butyl group Chemical group [H]C([H])([H])C([H])([H])C([H])([H])C([H])([H])* 0.000 description 1
- 125000003136 n-heptyl group Chemical group [H]C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])* 0.000 description 1
- 125000001280 n-hexyl group Chemical group C(CCCCC)* 0.000 description 1
- HKUFIYBZNQSHQS-UHFFFAOYSA-N n-octadecyloctadecan-1-amine Chemical compound CCCCCCCCCCCCCCCCCCNCCCCCCCCCCCCCCCCCC HKUFIYBZNQSHQS-UHFFFAOYSA-N 0.000 description 1
- 125000000740 n-pentyl group Chemical group [H]C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])* 0.000 description 1
- 125000004123 n-propyl group Chemical group [H]C([H])([H])C([H])([H])C([H])([H])* 0.000 description 1
- 229940031182 nanoparticles iron oxide Drugs 0.000 description 1
- 125000004593 naphthyridinyl group Chemical group N1=C(C=CC2=CC=CN=C12)* 0.000 description 1
- 210000001577 neostriatum Anatomy 0.000 description 1
- 230000004770 neurodegeneration Effects 0.000 description 1
- 208000015122 neurodegenerative disease Diseases 0.000 description 1
- 230000002981 neuropathic effect Effects 0.000 description 1
- 150000002825 nitriles Chemical class 0.000 description 1
- 125000000449 nitro group Chemical group [O-][N+](*)=O 0.000 description 1
- 229910052757 nitrogen Inorganic materials 0.000 description 1
- 125000006574 non-aromatic ring group Chemical group 0.000 description 1
- 125000000962 organic group Chemical group 0.000 description 1
- 239000012044 organic layer Substances 0.000 description 1
- 230000003204 osmotic effect Effects 0.000 description 1
- 230000002018 overexpression Effects 0.000 description 1
- 125000001715 oxadiazolyl group Chemical group 0.000 description 1
- IVMHDOBGNQOUHO-UHFFFAOYSA-N oxathiane Chemical compound C1CCSOC1 IVMHDOBGNQOUHO-UHFFFAOYSA-N 0.000 description 1
- 150000002923 oximes Chemical class 0.000 description 1
- 150000002924 oxiranes Chemical class 0.000 description 1
- 230000005298 paramagnetic effect Effects 0.000 description 1
- 239000008188 pellet Substances 0.000 description 1
- 230000035515 penetration Effects 0.000 description 1
- 230000000737 periodic effect Effects 0.000 description 1
- 230000002688 persistence Effects 0.000 description 1
- 125000001792 phenanthrenyl group Chemical group C1(=CC=CC=2C3=CC=CC=C3C=CC12)* 0.000 description 1
- DHRLEVQXOMLTIM-UHFFFAOYSA-N phosphoric acid;trioxomolybdenum Chemical compound O=[Mo](=O)=O.O=[Mo](=O)=O.O=[Mo](=O)=O.O=[Mo](=O)=O.O=[Mo](=O)=O.O=[Mo](=O)=O.O=[Mo](=O)=O.O=[Mo](=O)=O.O=[Mo](=O)=O.O=[Mo](=O)=O.O=[Mo](=O)=O.O=[Mo](=O)=O.OP(O)(O)=O DHRLEVQXOMLTIM-UHFFFAOYSA-N 0.000 description 1
- 229910052698 phosphorus Inorganic materials 0.000 description 1
- 239000011574 phosphorus Substances 0.000 description 1
- 125000004592 phthalazinyl group Chemical group C1(=NN=CC2=CC=CC=C12)* 0.000 description 1
- 229920000136 polysorbate Polymers 0.000 description 1
- 239000011591 potassium Substances 0.000 description 1
- 229910052700 potassium Inorganic materials 0.000 description 1
- 125000002924 primary amino group Chemical group [H]N([H])* 0.000 description 1
- 230000008569 process Effects 0.000 description 1
- 108090000765 processed proteins & peptides Proteins 0.000 description 1
- RAMTXCRMKBFPRG-UHFFFAOYSA-N prop-2-ynyl carbonochloridate Chemical compound ClC(=O)OCC#C RAMTXCRMKBFPRG-UHFFFAOYSA-N 0.000 description 1
- 125000006239 protecting group Chemical group 0.000 description 1
- 125000001042 pteridinyl group Chemical group N1=C(N=CC2=NC=CN=C12)* 0.000 description 1
- 125000004309 pyranyl group Chemical group O1C(C=CC=C1)* 0.000 description 1
- 125000003072 pyrazolidinyl group Chemical group 0.000 description 1
- 125000000719 pyrrolidinyl group Chemical group 0.000 description 1
- 125000001422 pyrrolinyl group Chemical group 0.000 description 1
- 125000006085 pyrrolopyridyl group Chemical group 0.000 description 1
- 238000011002 quantification Methods 0.000 description 1
- 125000002294 quinazolinyl group Chemical group N1=C(N=CC2=CC=CC=C12)* 0.000 description 1
- 125000001567 quinoxalinyl group Chemical group N1=C(C=NC2=CC=CC=C12)* 0.000 description 1
- 239000012217 radiopharmaceutical Substances 0.000 description 1
- 229940121896 radiopharmaceutical Drugs 0.000 description 1
- 230000002799 radiopharmaceutical effect Effects 0.000 description 1
- 238000011084 recovery Methods 0.000 description 1
- 238000006268 reductive amination reaction Methods 0.000 description 1
- 238000011160 research Methods 0.000 description 1
- 230000002441 reversible effect Effects 0.000 description 1
- 238000012552 review Methods 0.000 description 1
- 238000007363 ring formation reaction Methods 0.000 description 1
- 238000011808 rodent model Methods 0.000 description 1
- 229920006395 saturated elastomer Polymers 0.000 description 1
- 125000002914 sec-butyl group Chemical group [H]C([H])([H])C([H])([H])C([H])(*)C([H])([H])[H] 0.000 description 1
- 150000003333 secondary alcohols Chemical class 0.000 description 1
- 230000035945 sensitivity Effects 0.000 description 1
- 210000002966 serum Anatomy 0.000 description 1
- 239000000741 silica gel Substances 0.000 description 1
- 229910002027 silica gel Inorganic materials 0.000 description 1
- RYMZZMVNJRMUDD-HGQWONQESA-N simvastatin Chemical compound C([C@H]1[C@@H](C)C=CC2=C[C@H](C)C[C@@H]([C@H]12)OC(=O)C(C)(C)CC)C[C@@H]1C[C@@H](O)CC(=O)O1 RYMZZMVNJRMUDD-HGQWONQESA-N 0.000 description 1
- 239000002356 single layer Substances 0.000 description 1
- 239000002002 slurry Substances 0.000 description 1
- 235000010378 sodium ascorbate Nutrition 0.000 description 1
- 229960005055 sodium ascorbate Drugs 0.000 description 1
- PPASLZSBLFJQEF-RKJRWTFHSA-M sodium ascorbate Substances [Na+].OC[C@@H](O)[C@H]1OC(=O)C(O)=C1[O-] PPASLZSBLFJQEF-RKJRWTFHSA-M 0.000 description 1
- 239000011780 sodium chloride Substances 0.000 description 1
- PPASLZSBLFJQEF-RXSVEWSESA-M sodium-L-ascorbate Chemical compound [Na+].OC[C@H](O)[C@H]1OC(=O)C(O)=C1[O-] PPASLZSBLFJQEF-RXSVEWSESA-M 0.000 description 1
- HPALAKNZSZLMCH-UHFFFAOYSA-M sodium;chloride;hydrate Chemical compound O.[Na+].[Cl-] HPALAKNZSZLMCH-UHFFFAOYSA-M 0.000 description 1
- 239000011877 solvent mixture Substances 0.000 description 1
- 208000024794 sputum Diseases 0.000 description 1
- 210000003802 sputum Anatomy 0.000 description 1
- 150000003432 sterols Chemical class 0.000 description 1
- 235000003702 sterols Nutrition 0.000 description 1
- 238000003756 stirring Methods 0.000 description 1
- 125000005415 substituted alkoxy group Chemical group 0.000 description 1
- 125000000547 substituted alkyl group Chemical group 0.000 description 1
- 238000006467 substitution reaction Methods 0.000 description 1
- 239000000758 substrate Substances 0.000 description 1
- 239000005720 sucrose Substances 0.000 description 1
- 229940124530 sulfonamide Drugs 0.000 description 1
- 150000003456 sulfonamides Chemical class 0.000 description 1
- 150000003457 sulfones Chemical class 0.000 description 1
- 150000003462 sulfoxides Chemical class 0.000 description 1
- 230000001629 suppression Effects 0.000 description 1
- 208000024891 symptom Diseases 0.000 description 1
- 125000000999 tert-butyl group Chemical group [H]C([H])([H])C(*)(C([H])([H])[H])C([H])([H])[H] 0.000 description 1
- 125000003718 tetrahydrofuranyl group Chemical group 0.000 description 1
- 125000005944 tetrahydroimidazopyridyl group Chemical group 0.000 description 1
- 125000005888 tetrahydroindolyl group Chemical group 0.000 description 1
- 125000001712 tetrahydronaphthyl group Chemical group C1(CCCC2=CC=CC=C12)* 0.000 description 1
- 125000001412 tetrahydropyranyl group Chemical group 0.000 description 1
- 125000004632 tetrahydrothiopyranyl group Chemical group S1C(CCCC1)* 0.000 description 1
- 125000001113 thiadiazolyl group Chemical group 0.000 description 1
- 125000001984 thiazolidinyl group Chemical group 0.000 description 1
- 125000004001 thioalkyl group Chemical group 0.000 description 1
- 150000003567 thiocyanates Chemical class 0.000 description 1
- JADVWWSKYZXRGX-UHFFFAOYSA-M thioflavine T Chemical compound [Cl-].C1=CC(N(C)C)=CC=C1C1=[N+](C)C2=CC=C(C)C=C2S1 JADVWWSKYZXRGX-UHFFFAOYSA-M 0.000 description 1
- 150000003573 thiols Chemical class 0.000 description 1
- 125000004568 thiomorpholinyl group Chemical group 0.000 description 1
- 125000003944 tolyl group Chemical group 0.000 description 1
- 125000005490 tosylate group Chemical group 0.000 description 1
- 238000007070 tosylation reaction Methods 0.000 description 1
- DKZBBWMURDFHNE-UHFFFAOYSA-N trans-coniferylaldehyde Natural products COC1=CC(C=CC=O)=CC=C1O DKZBBWMURDFHNE-UHFFFAOYSA-N 0.000 description 1
- 230000007704 transition Effects 0.000 description 1
- 230000032258 transport Effects 0.000 description 1
- 125000004306 triazinyl group Chemical group 0.000 description 1
- ITMCEJHCFYSIIV-UHFFFAOYSA-M triflate Chemical compound [O-]S(=O)(=O)C(F)(F)F ITMCEJHCFYSIIV-UHFFFAOYSA-M 0.000 description 1
- 125000002023 trifluoromethyl group Chemical group FC(F)(F)* 0.000 description 1
- 239000003656 tris buffered saline Substances 0.000 description 1
- MWOOGOJBHIARFG-UHFFFAOYSA-N vanillin Chemical compound COC1=CC(C=O)=CC=C1O MWOOGOJBHIARFG-UHFFFAOYSA-N 0.000 description 1
- 230000002792 vascular Effects 0.000 description 1
- 239000003981 vehicle Substances 0.000 description 1
- 230000002861 ventricular Effects 0.000 description 1
- 125000000391 vinyl group Chemical group [H]C([*])=C([H])[H] 0.000 description 1
- 229920002554 vinyl polymer Polymers 0.000 description 1
- PIAOXUVIBAKVSP-UHFFFAOYSA-N γ-hydroxybutyraldehyde Chemical compound OCCCC=O PIAOXUVIBAKVSP-UHFFFAOYSA-N 0.000 description 1
Images
Classifications
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K49/00—Preparations for testing in vivo
- A61K49/06—Nuclear magnetic resonance [NMR] contrast preparations; Magnetic resonance imaging [MRI] contrast preparations
- A61K49/18—Nuclear magnetic resonance [NMR] contrast preparations; Magnetic resonance imaging [MRI] contrast preparations characterised by a special physical form, e.g. emulsions, microcapsules, liposomes
- A61K49/1806—Suspensions, emulsions, colloids, dispersions
- A61K49/1812—Suspensions, emulsions, colloids, dispersions liposomes, polymersomes, e.g. immunoliposomes
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K49/00—Preparations for testing in vivo
- A61K49/001—Preparation for luminescence or biological staining
- A61K49/0013—Luminescence
- A61K49/0017—Fluorescence in vivo
- A61K49/0019—Fluorescence in vivo characterised by the fluorescent group, e.g. oligomeric, polymeric or dendritic molecules
- A61K49/0021—Fluorescence in vivo characterised by the fluorescent group, e.g. oligomeric, polymeric or dendritic molecules the fluorescent group being a small organic molecule
- A61K49/0032—Methine dyes, e.g. cyanine dyes
- A61K49/0034—Indocyanine green, i.e. ICG, cardiogreen
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K49/00—Preparations for testing in vivo
- A61K49/001—Preparation for luminescence or biological staining
- A61K49/0063—Preparation for luminescence or biological staining characterised by a special physical or galenical form, e.g. emulsions, microspheres
- A61K49/0069—Preparation for luminescence or biological staining characterised by a special physical or galenical form, e.g. emulsions, microspheres the agent being in a particular physical galenical form
- A61K49/0076—Preparation for luminescence or biological staining characterised by a special physical or galenical form, e.g. emulsions, microspheres the agent being in a particular physical galenical form dispersion, suspension, e.g. particles in a liquid, colloid, emulsion
- A61K49/0084—Preparation for luminescence or biological staining characterised by a special physical or galenical form, e.g. emulsions, microspheres the agent being in a particular physical galenical form dispersion, suspension, e.g. particles in a liquid, colloid, emulsion liposome, i.e. bilayered vesicular structure
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K49/00—Preparations for testing in vivo
- A61K49/06—Nuclear magnetic resonance [NMR] contrast preparations; Magnetic resonance imaging [MRI] contrast preparations
- A61K49/08—Nuclear magnetic resonance [NMR] contrast preparations; Magnetic resonance imaging [MRI] contrast preparations characterised by the carrier
- A61K49/10—Organic compounds
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K49/00—Preparations for testing in vivo
- A61K49/06—Nuclear magnetic resonance [NMR] contrast preparations; Magnetic resonance imaging [MRI] contrast preparations
- A61K49/08—Nuclear magnetic resonance [NMR] contrast preparations; Magnetic resonance imaging [MRI] contrast preparations characterised by the carrier
- A61K49/10—Organic compounds
- A61K49/12—Macromolecular compounds
- A61K49/126—Linear polymers, e.g. dextran, inulin, PEG
Landscapes
- Health & Medical Sciences (AREA)
- Veterinary Medicine (AREA)
- Public Health (AREA)
- Epidemiology (AREA)
- Life Sciences & Earth Sciences (AREA)
- Animal Behavior & Ethology (AREA)
- General Health & Medical Sciences (AREA)
- Chemical & Material Sciences (AREA)
- Nuclear Medicine, Radiotherapy & Molecular Imaging (AREA)
- Radiology & Medical Imaging (AREA)
- Dispersion Chemistry (AREA)
- Biomedical Technology (AREA)
- Engineering & Computer Science (AREA)
- Medicinal Chemistry (AREA)
- Immunology (AREA)
- Medicines Containing Antibodies Or Antigens For Use As Internal Diagnostic Agents (AREA)
- Medicinal Preparation (AREA)
- Acyclic And Carbocyclic Compounds In Medicinal Compositions (AREA)
- Pharmaceuticals Containing Other Organic And Inorganic Compounds (AREA)
- Polyethers (AREA)
Description
本出願は、2014年10月8日出願の米国仮特許出願第62/061,514号、および2015年2月2日出願の同第62/111,057号の優先権を主張するものであり、その各々は参照により全体が本明細書に組み込まれる。
特定の実施形態を、実施例の形式で以下に記載する。本発明のすべての潜在的な用途を描写することは不可能である。したがって、実施形態は、かなり詳細に記載してあるが、添付の特許請求の範囲をそのような詳細に、もしくは任意の特定の実施形態に制限するか、またはいかなる方法で限定することを意図するものではない。
図1A、1B、および1Cは、化合物i〜xiii、および既知の化合物MeXO4、SB−13およびフロルベタピルを含む様々な化合物のCLogP値および構造を表形式で示す。CLogPは、Log[オクタノール中の濃度/水中の濃度]としての、オクタノール中の化合物の水に対する相対濃度についての分配係数の対数である。より低いCLogP値は、より高い親水性に相当する。MeXO4はアミロイド斑に対する高い親和性を示したが、発明者らはSB−13および臨床に使用されるフロルベタピル中のスチルベン構造が結合における感度および特異性を提供し得ると判断した。発明者らはさらに、ベンゾチアゾールまたはピリミジル部分が既知の化合物と比較してより低いCLogPおよび増加したH結合能を有する類似体を生成し得ると判断した。加えて、ヒドロキシエチル基(複数可)で窒素をキャッピングすると、さらに親水性が増加し、リポソームの脂質二重層中への挿入の代わりに、リポソームの外部の水性媒質中でリガンドが浮遊するのを維持する能力を高めることができた。したがって、i〜xiiiなどの化合物は、ゆえに、スチルベンコアの親水性を増加して二重層を不安定にすることなくリポソーム表面上に提示されるリガンドの能力を増強すると一般的に予想される側鎖修飾を用いて考案した。化合物i〜xiiiはそれぞれ、以前の化合物MeXO4よりも数桁親水性である。理論に束縛されるものではないが、より高い親水性は、例えば、リポソーム外の親水性環境中の、結合に利用可能な構成での対応する化合物の存在を増加させることによって、およびアミロイドβ線維との結合相互作用を増加させることなどによって、アミロイドβ線維との相互作用ならびに結合を促進できると考えられる。
Αβ線維を、全体の教示が参照により本明細書に組み込まれるKlunkら、Ann Neurol,2004;55:306−19の方法に従って合成した。簡潔に述べると、Αβ(1−40)ペプチド(rPeptide,Bogart,GA)を433μg/mL(100μΜ)の最終濃度にpH7.4のリン酸緩衝生理食塩水中に溶解した。この溶液を室温で4時間、700rpmで磁気撹拌棒を用いて撹拌して原線維の形成を促進した。原液を等分し、将来の使用のために−80℃で保存した。原線維の均質な懸濁液を維持するために、結合アッセイ用のアリコートを取り出す前に、ストック溶液を十分に撹拌した。原線維の均質な懸濁液を確実にするために、結合アッセイ用のアリコートを取り出す前にストック溶液を十分に撹拌した。
結合アッセイ:化合物または化合物−標識リポソームのアミロイド線維との結合親和性を、以下の方法によって測定した。競合アッセイ用のリガンド−標識リポソーム、リガンドストック溶液、およびクリサミンGを10mMのTris−HCl、pH7.4で500nMに希釈した。原線維ストック溶液を、200μLの反応混合物中の化合物のストック溶液または化合物標識リポソームと混合し、0.0625〜2.0μΜの試験化合物を有する原線維の最終濃度(20μΜ)を得た。結合混合物を室温で1時間インキュベートした。続いて、インキュベートした結合混合物を16,400rpmで20分間遠心分離して原線維を分離した。原線維沈殿物をTris−HClで2回洗浄した。次いで、蛍光を、それぞれ405nmおよび535nmの励起波長ならびに発光波長を用いて、FilterMax F−5マルチモードプレートリーダー(Molecular devices,Sunnyvale,CA)で測定した。競合結合アッセイは、20μΜの原線維、1.0μΜの試験化合物および様々な量の非蛍光競合物質のクリサミンGを使用し、1時間インキュベートした。
粒径を測定するために、押出プロセスからの定期的な試料および透析濾過後の最終試料をPBSで希釈し、自己相関システムに取り付けられたゴニオメータベースの動的光散乱システム(BI−90,Brookhaven Instruments Corporation,Holtsville,NY)で測定した。532nmの固体レーザーを光源として使用し、90℃で、光電子増倍管で判別検出されるまで調整された試料の濃度から、毎秒約100kカウント(kcps)を得た。相関関数を、相関ビンの指数関数的に離間したセットを使用して測定し、相関関数の初期の指数関数的な降下を捕捉する少なくとも10チャネル、および長期残光を捕捉する10チャネルを確実にした。ダストの特徴である大きな粒径の混入を示す、ベースラインよりも有意に高い長期相関関数を示した相関関数スライスを排除するダスト識別アルゴリズムを用いて、各試料について2分間の相関関数を平均した。得られた平均相関関数を、CONTINアルゴリズムを用いて分析し、体積平均分布を使用して、平均サイズおよび標準偏差を推定した。全ての分布は実質的に単峰性であった(主ピークにおける体積の99%以上)。
脂質二重層の表面上に表面Gdキレートで装飾したリポソームを調製した(ビス−ステアリル鎖がリポソームの二重層中に挿入するようにGdキレートが脂質アンカーBSA−DTPA−Gdにコンジュゲートされる)。図10Aに示すように、これらのリポソームは、低磁場強度で高いGdモル緩和を示した。図10Aにおいて、例示的なリポソームを、より低い磁場強度で遊離Gdキレート(GadobenateDimelglumine、MULTIHANCE(商標)、Bracco Imaging、Monroe Township、NJ)と比較しているが、その差は、遊離キレートが表面提示よりも緩和的である9.4Tでの点まで、より高い磁場強度で徐々に失われる。
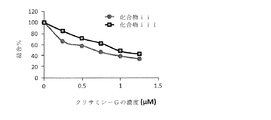
化合物iiiのリガンドを使用してアミロイド斑を標的とするデュアルGdリポソームを、したがって、マウス中のアミロイド斑を画像化するそれらの能力について試験した。デュアルGd/化合物iiiリポソームをマウス内に静脈内注射し、1T MRIスキャナーでT1加重スピンエコーシーケンス(TE=32ms、TR=770ms)を用いて4日後にスキャンした。デュアル−Gd/化合物iiiリポソームは、アミロイド保有マウスの2つの異なる系統(15ヶ月齢のTetO/APPswe−indおよび9ヶ月齢のTg2576)において、アミロイド沈着に特徴的なパターンでシグナル増強をもたらした。同じマウスの注射前スキャンおよび同様に注射したアミロイド陰性マウス、ならびに同じ粒子の非標的バージョン(すなわち、化合物iiiを含まない)を注射したアミロイド陽性マウスでは、比較してシグナルは得られなかった。未結合リポソームが循環から除去されるように、4日の遅延を計画した。PEG化リポソームは約24時間の循環半減期を有しており、過去の経験から、4日の遅延が血液プールからのリポソームのほぼ完全な除去に十分であることが示唆された。
リポソームはまた、近赤外イメージングのために、二重層に約0.6モル%のインドシアニングリーン(ICG)を含有していた。図14A〜H4は、死後のマウスの脳の近赤外イメージングの結果を示す。Tg2576マウスの脳切片の共焦点イメージングにより、トランスジェニックマウスの皮質および海馬におけるICGシグナルの増強が明らかになり、非トランスジェニック対照(図14B)と比較した場合APP+脳において局在性がより高い(図14A)ことが示された。高倍率では、図14Eが、図14Aに対応するアミロイド沈着物の特徴的な点状構造を明らかにした;図14Fは、図14Bの対照に相当する。
合成の取り組みは、図1A〜1B中の最低のCLogP値を有する3つの化合物(化合物ii、化合物iおよび化合物iii)に焦点を当てた。合成および精製は効率的であり、全収率は約90%であった。化合物iiおよび化合物iiiを、アミロイド線維の結合および特異性について試験し、非常に類似した結果が得られたことから、アミロイド結合能力は、これらの分子の比較的高い親水性によって顕著に影響されないことが示唆された。実際には、MeXO4で行った類似の研究と比較して本分子はMeXO4よりも非常に強くアミロイド線維に結合するようであり、クリサミン−Gで置換された結合種はMeXO4での80%超と比較して、わずか約60%である。化合物iiiを、そのより低いCLogP値およびわずかにより高いアミロイド結合能力に基づいて、リード候補として選択した。
Claims (15)
- リン脂質−ポリマー−芳香族コンジュゲートを含む膜を含む、リポソーム組成物であって、
前記リン脂質−ポリマー−芳香族コンジュゲート中の芳香族部分が、
によって表され、
前記リン脂質−ポリマー−芳香族コンジュゲート中のリン脂質−ポリマー部分が、
R2は、アルキレンまたはアルコキシアルキレンのうちの1つである1〜6個の炭素原子を含む連結基であり;
R3は、水素、C1−C6アルキル、またはC1−C6アルコキシアルキルであり、且つ、水素以外のR3は、0個、1個または複数の−OHで置換されており;
ピリミジンPは、−OH、−O−アルキルおよび−NH2のうちの0個、1個または複数で置換されており;
nは、約60〜約100の整数であり;
mは、12、13、14、15、16、17、または18のうちの1つである、
リポソーム組成物。 - 前記膜によってカプセル化されたものまたは前記膜に結合したもののうちの少なくとも1つである非放射性磁気共鳴イメージング(MRI)コントラスト増強剤をさらに含む、請求項1に記載のリポソーム組成物。
- 前記膜が、第1のリン脂質および第2のリン脂質をさらに含み、前記第2のリン脂質が、親水性ポリマーで誘導体化されている、請求項1に記載のリポソーム組成物。
- 患者中のアミロイド沈着物の画像化における使用のためのリポソーム組成物であって、
前記リポソーム組成物が、リン脂質−ポリマー−芳香族コンジュゲート(前記リン脂質−ポリマー−芳香族コンジュゲート中の芳香族部分は、
前記使用が、
検出可能な量の前記リポソーム組成物を前記患者中に導入するステップ;
前記リポソーム組成物が1個以上のアミロイド沈着物と結合するのに十分な時間を与えるステップ;および
1個以上のアミロイド沈着物と結合したリポソーム組成物を検出するステップ、
を含む、
リポソーム組成物。 - 前記検出するステップが、磁気共鳴イメージングを用いて検出することを含む、請求項7に記載のリポソーム組成物。
- 前記非放射性磁気共鳴イメージング(MRI)コントラスト増強剤が、前記膜によってカプセル化されており、且つ、前記膜に結合している、請求項7に記載のリポソーム組成物。
- 前記リポソーム組成物が、約100,000、125,000、150,000、165,000、180,000、190,000、および200,000のうちの少なくとも約1つ以上のmM−1s−1の粒子あたりの緩和により特徴付けられる、請求項9に記載のリポソーム組成物。
- 前記使用が、前記1個以上のアミロイド沈着物と結合したリポソーム組成物の検出に応じて前記患者をアルツハイマー病と診断することをさらに含む、請求項7に記載のリポソーム組成物。
- 前記使用が、
前記1個以上のアミロイド沈着物と結合したリポソーム組成物の検出に応じて、アルツハイマー病を潜在的に有するとして前記患者を特定すること;
タウ神経原線維濃縮体の分析に前記患者を供すること;および
前記1個以上のアミロイド沈着物と結合したリポソーム組成物の検出と共にタウ神経原線維濃縮体の存在を決定したら、前記患者をアルツハイマー病と診断すること、
をさらに含む、請求項7に記載のリポソーム組成物。 - 患者中のアミロイド沈着物を画像化するためのキットであって、
リン脂質−ポリマー−芳香族コンジュゲート(前記リン脂質−ポリマー−芳香族コンジュゲート中の芳香族部分は、
説明書であって、
検出可能な量の前記リポソーム組成物を前記患者中へ導入し;
前記リポソーム組成物が1個以上のアミロイド沈着物と結合するのに十分な時間を与え;
1個以上のアミロイド沈着物と結合したリポソーム組成物を検出する
ようにユーザーに指示する説明書、
を含むキット。 - 前記説明書が、1個以上のアミロイド沈着物と結合したリポソーム組成物の検出に応じて前記患者をアルツハイマー病と診断するように前記ユーザーにさらに指示する、請求項14に記載のキット。
Applications Claiming Priority (5)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| US201462061514P | 2014-10-08 | 2014-10-08 | |
| US62/061,514 | 2014-10-08 | ||
| US201562111057P | 2015-02-02 | 2015-02-02 | |
| US62/111,057 | 2015-02-02 | ||
| PCT/US2015/054732 WO2016057812A1 (en) | 2014-10-08 | 2015-10-08 | Mri imaging of amyloid plaque using liposomes |
Publications (3)
| Publication Number | Publication Date |
|---|---|
| JP2017532378A JP2017532378A (ja) | 2017-11-02 |
| JP2017532378A5 JP2017532378A5 (ja) | 2018-11-22 |
| JP6505235B2 true JP6505235B2 (ja) | 2019-04-24 |
Family
ID=55653789
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| JP2017538918A Active JP6505235B2 (ja) | 2014-10-08 | 2015-10-08 | リポソームを用いるアミロイド斑のmriイメージング |
Country Status (11)
| Country | Link |
|---|---|
| US (4) | US9744251B2 (ja) |
| EP (3) | EP4035688A1 (ja) |
| JP (1) | JP6505235B2 (ja) |
| KR (1) | KR102409017B1 (ja) |
| CN (1) | CN107106708B (ja) |
| AU (2) | AU2015330824A1 (ja) |
| CA (1) | CA2963941C (ja) |
| ES (1) | ES2743704T3 (ja) |
| HK (1) | HK1243335A1 (ja) |
| MX (1) | MX371295B (ja) |
| WO (1) | WO2016057812A1 (ja) |
Families Citing this family (17)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| PL2694116T3 (pl) | 2011-04-06 | 2018-12-31 | Board Of Regents Of The University Of Texas System | Nanocząstki na bazie lipidów |
| JP6130401B2 (ja) | 2012-01-20 | 2017-05-17 | アンナプラガダ,アナンス | 医学画像を客観的に特徴付けるための方法および組成物 |
| CN107106708B (zh) | 2014-10-08 | 2021-06-18 | 德克萨斯州儿童医院 | 使用脂质体进行的淀粉样蛋白斑mri成像 |
| AU2017299219B2 (en) | 2016-07-22 | 2022-12-22 | Ac Immune Sa | Compounds for imaging Tau protein aggregates |
| JP7059270B2 (ja) | 2016-07-22 | 2022-04-25 | エーシー・イミューン・エス・アー | タウタンパク質凝集体を画像化するための化合物 |
| US11052162B2 (en) | 2016-09-16 | 2021-07-06 | Texas Children's Hospital | Liposomal gadolinium (GD) contrast agent “NMRX” for T1-MRI |
| US11241510B2 (en) | 2016-11-30 | 2022-02-08 | Texas Children's Hospital | Hydrophilic fluorinated molecules for liposomal 19F MRI probes with unique MR signatures |
| WO2019145292A1 (en) | 2018-01-24 | 2019-08-01 | Ac Immune Sa | Azacarboline compounds for the detection of tau aggregates |
| EP3743423A1 (en) | 2018-01-24 | 2020-12-02 | AC Immune SA | Gamma-carboline compounds for the detection of tau aggregates |
| CN112384202B (zh) * | 2018-03-29 | 2023-03-21 | 微脉管治疗有限公司 | 用于检测和治疗阿尔茨海默病的组合物及方法 |
| CN112533928A (zh) | 2018-06-08 | 2021-03-19 | Ac免疫有限公司 | 用于诊断的新化合物 |
| EP3914230A4 (en) * | 2019-01-24 | 2023-06-07 | Alzeca Biosciences, Llc | <SMALLCAPS/>? ? ?FUNCTIONALIZED LIPOSOMES FOR IMAGE MISFOLDED PROTEINS |
| JP2022550237A (ja) * | 2019-07-08 | 2022-12-01 | アルゼカ バイオサイエンシズ、エルエルシー | タウ病理のためのターゲティングリガンド |
| WO2021155128A1 (en) * | 2020-01-29 | 2021-08-05 | Texas Children's Hospital | Targeted contrast agents for mri of amyloid deposition |
| CN115279424A (zh) * | 2020-02-12 | 2022-11-01 | 德克萨斯儿童医院 | 用于α-突触核蛋白沉积MRI的靶向对比剂 |
| US11779664B2 (en) | 2020-02-12 | 2023-10-10 | Texas Children's Hospital | Targeted contrast agents for MRI of alpha-synuclein deposition |
| CN115970009A (zh) * | 2023-02-08 | 2023-04-18 | 丽水市中心医院 | Vhpk多肽修饰的荧光纳米脂质体给药系统、其制备方法及应用 |
Family Cites Families (35)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US5204085A (en) | 1985-01-08 | 1993-04-20 | Mallinckrodt Medical, Inc. | Method for enhancing the safety of metal-ligand chelates as X-ray contrast agents |
| CA2185810A1 (en) | 1994-03-28 | 1995-10-05 | Jo Klaveness | Liposomes |
| US6071532A (en) | 1996-10-15 | 2000-06-06 | Emory University | Synthesis of glycophospholipid and peptide-phospholipid conjugates and uses thereof |
| ATE287700T1 (de) | 1999-11-30 | 2005-02-15 | Univ Arizona | Strahlung-sensitive liposomen |
| US7029655B2 (en) | 2000-10-04 | 2006-04-18 | California Institute Of Technology | Magnetic resonance imaging agents for in vivo labeling and detection of amyloid deposits |
| WO2002094191A2 (en) | 2001-05-23 | 2002-11-28 | New York University | Detection of alzheimer's amyloid by magnetic resonance imaging |
| US7138136B2 (en) | 2002-03-05 | 2006-11-21 | Cleveland State University | Agglomerated particles for aerosol drug delivery |
| EP1641742A4 (en) * | 2003-05-01 | 2006-11-29 | Nst Neurosurvival Technologies | COMPOUNDS BINDING SELECTIVELY TO MEMBRANES OF APOPTOTIC CELLS |
| US7208174B2 (en) | 2003-12-18 | 2007-04-24 | Hoffmann-La Roche Inc. | Liposome compositions |
| US20100031378A1 (en) | 2008-08-04 | 2010-02-04 | Edwards Joel A | Novel gene disruptions, compositions and methods relating thereto |
| US8357351B2 (en) | 2004-04-21 | 2013-01-22 | Ananth Annapragada | Nano-scale contrast agents and methods of use |
| US7713517B2 (en) | 2004-04-21 | 2010-05-11 | Marval Biosciences, Inc. | Compositions and methods for enhancing contrast in imaging |
| US20070292354A1 (en) | 2004-09-23 | 2007-12-20 | Guerbet | Contrast Agents Encapsulating Systems for Cest Imaging |
| JP2009511568A (ja) | 2005-10-13 | 2009-03-19 | ワラタ ファーマシューティカルズ, インコーポレイテッド | イノシトール誘導体、ならびに異常なタンパク質の折りたたみまたは凝集、あるいはアミロイドの形成、沈着、蓄積または残存を特徴とする疾患の処置におけるその使用 |
| US20070160658A1 (en) | 2005-10-20 | 2007-07-12 | The Penn State Research Foundation | Delivery system for diagnostic and therapeutic agents |
| US20090123047A1 (en) | 2007-03-21 | 2009-05-14 | Yfantis Spyros A | Method and system for characterizing prostate images |
| EP1982733A1 (en) | 2007-04-17 | 2008-10-22 | Bayer Schering Pharma Aktiengesellschaft | Use of a contrast agent for magnetic resonance imaging of endoleaks |
| US7858803B2 (en) | 2007-04-27 | 2010-12-28 | The General Hospital Corporation | Imaging tracers for early detection and treatment of amyloid plaques caused by Alzheimer's disease and related disorders |
| CA2695204A1 (en) | 2007-07-26 | 2009-01-29 | Nanoscan Imaging, Llc | Methods for imaging using improved nanoparticulate contrast agents |
| US8642013B2 (en) | 2007-12-05 | 2014-02-04 | Ananth Annapragada | Nano-scale contrast agents and methods of use |
| US20100286067A1 (en) * | 2008-01-08 | 2010-11-11 | Biogenerix Ag | Glycoconjugation of polypeptides using oligosaccharyltransferases |
| ITMI20081052A1 (it) | 2008-06-10 | 2009-12-11 | Univ Milano Bicocca | Liposomi in grado di legare efficacemente il peptide beta-amiloide |
| WO2009152445A1 (en) | 2008-06-13 | 2009-12-17 | Marval Biosciences, Inc. | Imaging of atherosclerotic plaques using liposomal imaging agents |
| US20110208064A1 (en) | 2008-07-31 | 2011-08-25 | Ran Chongzhao | Curcumin Derivatives for Amyloid-Beta Plaque Imaging |
| AR074760A1 (es) | 2008-12-18 | 2011-02-09 | Metabolex Inc | Agonistas del receptor gpr120 y usos de los mismos en medicamentos para el tratamiento de diabetes y el sindrome metabolico. |
| EP2410990A4 (en) | 2009-03-19 | 2015-05-20 | Marval Biosciences Inc | COMPOSITIONS AND METHODS FOR CONTRAST GAINING IN IMAGING |
| WO2011045415A2 (en) | 2009-10-15 | 2011-04-21 | Guerbet | New imaging agents and their use for the diagnostic in vivo of neurodegenerative diseases, notably alzheimer's disease and derivative diseases |
| CA2801182A1 (en) * | 2010-06-16 | 2011-12-22 | Metabolex, Inc. | Gpr120 receptor agonists and uses thereof |
| CA2822021C (en) | 2011-03-02 | 2018-01-02 | Sensulin, Llc | Vesicle compositions |
| PL2694116T3 (pl) | 2011-04-06 | 2018-12-31 | Board Of Regents Of The University Of Texas System | Nanocząstki na bazie lipidów |
| GB201112056D0 (en) | 2011-07-14 | 2011-08-31 | Univ Leuven Kath | Antibodies |
| JP6130401B2 (ja) | 2012-01-20 | 2017-05-17 | アンナプラガダ,アナンス | 医学画像を客観的に特徴付けるための方法および組成物 |
| US9377472B2 (en) | 2012-04-30 | 2016-06-28 | Amoneta Diagnostics | Biological complex specific for Alzheimer's disease detection in vitro and use thereof |
| KR20150127245A (ko) | 2013-03-14 | 2015-11-16 | 켈탁시스, 인코퍼레이티드 | 류코트라이엔 a4 가수분해효소의 저해제 |
| CN107106708B (zh) | 2014-10-08 | 2021-06-18 | 德克萨斯州儿童医院 | 使用脂质体进行的淀粉样蛋白斑mri成像 |
-
2015
- 2015-10-08 CN CN201580066712.6A patent/CN107106708B/zh active Active
- 2015-10-08 JP JP2017538918A patent/JP6505235B2/ja active Active
- 2015-10-08 ES ES15849750T patent/ES2743704T3/es active Active
- 2015-10-08 US US14/878,745 patent/US9744251B2/en active Active
- 2015-10-08 CA CA2963941A patent/CA2963941C/en active Active
- 2015-10-08 WO PCT/US2015/054732 patent/WO2016057812A1/en active Application Filing
- 2015-10-08 KR KR1020177012354A patent/KR102409017B1/ko active IP Right Grant
- 2015-10-08 AU AU2015330824A patent/AU2015330824A1/en not_active Abandoned
- 2015-10-08 EP EP22152987.8A patent/EP4035688A1/en active Pending
- 2015-10-08 EP EP15849750.3A patent/EP3204051B1/en active Active
- 2015-10-08 MX MX2017004695A patent/MX371295B/es active IP Right Grant
- 2015-10-08 EP EP19172751.0A patent/EP3542828A1/en not_active Withdrawn
-
2016
- 2016-12-01 US US15/366,667 patent/US10537649B2/en active Active
-
2018
- 2018-02-27 HK HK18102837.3A patent/HK1243335A1/zh unknown
-
2019
- 2019-11-08 US US16/677,751 patent/US20200069820A1/en not_active Abandoned
-
2020
- 2020-02-14 US US16/791,068 patent/US11141495B2/en active Active
-
2021
- 2021-05-25 AU AU2021203388A patent/AU2021203388B2/en active Active
Also Published As
| Publication number | Publication date |
|---|---|
| US20200069820A1 (en) | 2020-03-05 |
| EP3204051A4 (en) | 2018-06-27 |
| KR20170103748A (ko) | 2017-09-13 |
| CA2963941C (en) | 2023-08-01 |
| KR102409017B1 (ko) | 2022-06-16 |
| US9744251B2 (en) | 2017-08-29 |
| CN107106708A (zh) | 2017-08-29 |
| US10537649B2 (en) | 2020-01-21 |
| BR112017007238A2 (pt) | 2018-01-16 |
| CN107106708B (zh) | 2021-06-18 |
| AU2021203388A1 (en) | 2021-06-24 |
| EP3204051B1 (en) | 2019-05-29 |
| EP4035688A1 (en) | 2022-08-03 |
| AU2015330824A1 (en) | 2017-05-25 |
| US20200179540A1 (en) | 2020-06-11 |
| EP3204051A1 (en) | 2017-08-16 |
| ES2743704T3 (es) | 2020-02-20 |
| US20170080111A1 (en) | 2017-03-23 |
| MX371295B (es) | 2020-01-24 |
| CA2963941A1 (en) | 2016-04-14 |
| HK1243335A1 (zh) | 2018-07-13 |
| WO2016057812A1 (en) | 2016-04-14 |
| US20160101197A1 (en) | 2016-04-14 |
| JP2017532378A (ja) | 2017-11-02 |
| AU2021203388B2 (en) | 2024-05-30 |
| MX2017004695A (es) | 2018-01-24 |
| US11141495B2 (en) | 2021-10-12 |
| EP3542828A1 (en) | 2019-09-25 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| JP6505235B2 (ja) | リポソームを用いるアミロイド斑のmriイメージング | |
| Tanifum et al. | A novel liposomal nanoparticle for the imaging of amyloid plaque by magnetic resonance imaging | |
| CN104144708B (zh) | 基于脂质的纳米粒子 | |
| Cai et al. | Near‐infrared fluorescence imaging of gastrin releasing peptide receptor targeting in prostate cancer lymph node metastases | |
| Kim et al. | An aptamer‐based magnetic resonance imaging contrast agent for detecting oligomeric amyloid‐β in the brain of an Alzheimer's disease mouse model | |
| JP7545393B2 (ja) | 誤って折りたたまれたタンパク質を画像化するための機能化リポソーム | |
| US10124078B2 (en) | Lipid-based nanoparticles | |
| BR112017007238B1 (pt) | Composição lipossomal compreendendo um conjugado fosfolipídiopolímero-aromático e um agente potencializador de contraste de imagem de ressonância magnética não radioativo para geração de imagem de depósitos amiloides | |
| Andreozzi | The Development of Multimodal Imaging Probes for Visualizing Neuroinflammation in Alzheimer's Disease and Traumatic Brain Injury | |
| Wang | Development of novel molecular imaging agents for myelination |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| A521 | Request for written amendment filed |
Free format text: JAPANESE INTERMEDIATE CODE: A523 Effective date: 20170908 |
|
| A521 | Request for written amendment filed |
Free format text: JAPANESE INTERMEDIATE CODE: A523 Effective date: 20181005 |
|
| A621 | Written request for application examination |
Free format text: JAPANESE INTERMEDIATE CODE: A621 Effective date: 20181005 |
|
| A871 | Explanation of circumstances concerning accelerated examination |
Free format text: JAPANESE INTERMEDIATE CODE: A871 Effective date: 20181005 |
|
| A975 | Report on accelerated examination |
Free format text: JAPANESE INTERMEDIATE CODE: A971005 Effective date: 20181018 |
|
| A131 | Notification of reasons for refusal |
Free format text: JAPANESE INTERMEDIATE CODE: A131 Effective date: 20181120 |
|
| A521 | Request for written amendment filed |
Free format text: JAPANESE INTERMEDIATE CODE: A523 Effective date: 20190214 |
|
| TRDD | Decision of grant or rejection written | ||
| A01 | Written decision to grant a patent or to grant a registration (utility model) |
Free format text: JAPANESE INTERMEDIATE CODE: A01 Effective date: 20190226 |
|
| A61 | First payment of annual fees (during grant procedure) |
Free format text: JAPANESE INTERMEDIATE CODE: A61 Effective date: 20190326 |
|
| R150 | Certificate of patent or registration of utility model |
Ref document number: 6505235 Country of ref document: JP Free format text: JAPANESE INTERMEDIATE CODE: R150 |
|
| R250 | Receipt of annual fees |
Free format text: JAPANESE INTERMEDIATE CODE: R250 |
|
| R250 | Receipt of annual fees |
Free format text: JAPANESE INTERMEDIATE CODE: R250 |
|
| R250 | Receipt of annual fees |
Free format text: JAPANESE INTERMEDIATE CODE: R250 |