JP6407952B2 - 水溶液からの有機化合物及びクロラミンの除去 - Google Patents
水溶液からの有機化合物及びクロラミンの除去 Download PDFInfo
- Publication number
- JP6407952B2 JP6407952B2 JP2016500803A JP2016500803A JP6407952B2 JP 6407952 B2 JP6407952 B2 JP 6407952B2 JP 2016500803 A JP2016500803 A JP 2016500803A JP 2016500803 A JP2016500803 A JP 2016500803A JP 6407952 B2 JP6407952 B2 JP 6407952B2
- Authority
- JP
- Japan
- Prior art keywords
- carbon
- chloramine
- sulfur
- medium
- carbon substrate
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Active
Links
- QDHHCQZDFGDHMP-UHFFFAOYSA-N Chloramine Chemical compound ClN QDHHCQZDFGDHMP-UHFFFAOYSA-N 0.000 title claims description 99
- 150000002894 organic compounds Chemical class 0.000 title claims description 49
- 239000007864 aqueous solution Substances 0.000 title claims description 46
- OKTJSMMVPCPJKN-UHFFFAOYSA-N Carbon Chemical compound [C] OKTJSMMVPCPJKN-UHFFFAOYSA-N 0.000 claims description 225
- NINIDFKCEFEMDL-UHFFFAOYSA-N Sulfur Chemical compound [S] NINIDFKCEFEMDL-UHFFFAOYSA-N 0.000 claims description 101
- 229910052717 sulfur Inorganic materials 0.000 claims description 99
- 239000011593 sulfur Substances 0.000 claims description 94
- 150000001875 compounds Chemical class 0.000 claims description 79
- 238000000034 method Methods 0.000 claims description 52
- 239000000243 solution Substances 0.000 claims description 14
- 230000003647 oxidation Effects 0.000 claims description 11
- 238000007254 oxidation reaction Methods 0.000 claims description 11
- 238000005211 surface analysis Methods 0.000 claims description 5
- 239000013626 chemical specie Substances 0.000 claims description 2
- 229910052799 carbon Inorganic materials 0.000 description 181
- 239000000758 substrate Substances 0.000 description 103
- IJGRMHOSHXDMSA-UHFFFAOYSA-N Atomic nitrogen Chemical compound N#N IJGRMHOSHXDMSA-UHFFFAOYSA-N 0.000 description 62
- HEDRZPFGACZZDS-UHFFFAOYSA-N Chloroform Chemical compound ClC(Cl)Cl HEDRZPFGACZZDS-UHFFFAOYSA-N 0.000 description 52
- 239000007795 chemical reaction product Substances 0.000 description 46
- 229910052757 nitrogen Inorganic materials 0.000 description 36
- 229910052751 metal Inorganic materials 0.000 description 33
- 239000002184 metal Substances 0.000 description 33
- XLYOFNOQVPJJNP-UHFFFAOYSA-N water Substances O XLYOFNOQVPJJNP-UHFFFAOYSA-N 0.000 description 33
- QVGXLLKOCUKJST-UHFFFAOYSA-N atomic oxygen Chemical compound [O] QVGXLLKOCUKJST-UHFFFAOYSA-N 0.000 description 30
- 238000001914 filtration Methods 0.000 description 30
- 229910052760 oxygen Inorganic materials 0.000 description 30
- 239000001301 oxygen Substances 0.000 description 30
- 238000010438 heat treatment Methods 0.000 description 27
- 239000000463 material Substances 0.000 description 26
- 150000003839 salts Chemical class 0.000 description 22
- 239000000460 chlorine Substances 0.000 description 20
- 239000002245 particle Substances 0.000 description 20
- 238000011282 treatment Methods 0.000 description 20
- ZAMOUSCENKQFHK-UHFFFAOYSA-N Chlorine atom Chemical compound [Cl] ZAMOUSCENKQFHK-UHFFFAOYSA-N 0.000 description 19
- 229910052801 chlorine Inorganic materials 0.000 description 19
- 239000011148 porous material Substances 0.000 description 17
- QJGQUHMNIGDVPM-UHFFFAOYSA-N nitrogen group Chemical group [N] QJGQUHMNIGDVPM-UHFFFAOYSA-N 0.000 description 15
- 238000012545 processing Methods 0.000 description 14
- 238000006243 chemical reaction Methods 0.000 description 13
- 239000001257 hydrogen Substances 0.000 description 12
- 229910052739 hydrogen Inorganic materials 0.000 description 12
- 239000000356 contaminant Substances 0.000 description 11
- 239000000203 mixture Substances 0.000 description 11
- 239000000126 substance Substances 0.000 description 11
- 239000002023 wood Substances 0.000 description 11
- 150000001450 anions Chemical class 0.000 description 10
- 239000003651 drinking water Substances 0.000 description 10
- 235000020188 drinking water Nutrition 0.000 description 10
- 239000012530 fluid Substances 0.000 description 10
- -1 trihalomethanes Substances 0.000 description 10
- 239000007789 gas Substances 0.000 description 9
- 239000011159 matrix material Substances 0.000 description 9
- 238000004833 X-ray photoelectron spectroscopy Methods 0.000 description 8
- 239000007788 liquid Substances 0.000 description 8
- 235000013162 Cocos nucifera Nutrition 0.000 description 7
- 244000060011 Cocos nucifera Species 0.000 description 7
- 238000004458 analytical method Methods 0.000 description 7
- 150000001768 cations Chemical class 0.000 description 7
- 150000002739 metals Chemical class 0.000 description 7
- 239000000047 product Substances 0.000 description 7
- 239000012085 test solution Substances 0.000 description 7
- 238000012360 testing method Methods 0.000 description 7
- XEEYBQQBJWHFJM-UHFFFAOYSA-N Iron Chemical compound [Fe] XEEYBQQBJWHFJM-UHFFFAOYSA-N 0.000 description 6
- 230000003197 catalytic effect Effects 0.000 description 6
- 238000009792 diffusion process Methods 0.000 description 6
- 150000002431 hydrogen Chemical class 0.000 description 6
- 229910052976 metal sulfide Inorganic materials 0.000 description 6
- 238000001228 spectrum Methods 0.000 description 6
- 239000003570 air Substances 0.000 description 5
- QSHDDOUJBYECFT-UHFFFAOYSA-N mercury Chemical compound [Hg] QSHDDOUJBYECFT-UHFFFAOYSA-N 0.000 description 5
- 229910052753 mercury Inorganic materials 0.000 description 5
- 125000004433 nitrogen atom Chemical group N* 0.000 description 5
- 230000008569 process Effects 0.000 description 5
- 239000002904 solvent Substances 0.000 description 5
- 125000004434 sulfur atom Chemical group 0.000 description 5
- QGZKDVFQNNGYKY-UHFFFAOYSA-N Ammonia Chemical compound N QGZKDVFQNNGYKY-UHFFFAOYSA-N 0.000 description 4
- UFHFLCQGNIYNRP-UHFFFAOYSA-N Hydrogen Chemical compound [H][H] UFHFLCQGNIYNRP-UHFFFAOYSA-N 0.000 description 4
- 239000012298 atmosphere Substances 0.000 description 4
- 125000004429 atom Chemical group 0.000 description 4
- 230000008859 change Effects 0.000 description 4
- 239000003245 coal Substances 0.000 description 4
- 238000000354 decomposition reaction Methods 0.000 description 4
- 239000012535 impurity Substances 0.000 description 4
- 235000019988 mead Nutrition 0.000 description 4
- 239000002994 raw material Substances 0.000 description 4
- 239000011257 shell material Substances 0.000 description 4
- 239000007787 solid Substances 0.000 description 4
- 229960002135 sulfadimidine Drugs 0.000 description 4
- ASWVTGNCAZCNNR-UHFFFAOYSA-N sulfamethazine Chemical compound CC1=CC(C)=NC(NS(=O)(=O)C=2C=CC(N)=CC=2)=N1 ASWVTGNCAZCNNR-UHFFFAOYSA-N 0.000 description 4
- 238000010998 test method Methods 0.000 description 4
- UHOVQNZJYSORNB-UHFFFAOYSA-N Benzene Chemical compound C1=CC=CC=C1 UHOVQNZJYSORNB-UHFFFAOYSA-N 0.000 description 3
- LSNNMFCWUKXFEE-UHFFFAOYSA-M Bisulfite Chemical compound OS([O-])=O LSNNMFCWUKXFEE-UHFFFAOYSA-M 0.000 description 3
- OKKJLVBELUTLKV-UHFFFAOYSA-N Methanol Chemical compound OC OKKJLVBELUTLKV-UHFFFAOYSA-N 0.000 description 3
- HEMHJVSKTPXQMS-UHFFFAOYSA-M Sodium hydroxide Chemical compound [OH-].[Na+] HEMHJVSKTPXQMS-UHFFFAOYSA-M 0.000 description 3
- 230000008901 benefit Effects 0.000 description 3
- 239000006227 byproduct Substances 0.000 description 3
- 150000001721 carbon Chemical class 0.000 description 3
- 238000009838 combustion analysis Methods 0.000 description 3
- 238000002485 combustion reaction Methods 0.000 description 3
- 238000001514 detection method Methods 0.000 description 3
- OEBRKCOSUFCWJD-UHFFFAOYSA-N dichlorvos Chemical compound COP(=O)(OC)OC=C(Cl)Cl OEBRKCOSUFCWJD-UHFFFAOYSA-N 0.000 description 3
- 230000006870 function Effects 0.000 description 3
- RAXXELZNTBOGNW-UHFFFAOYSA-N imidazole Natural products C1=CNC=N1 RAXXELZNTBOGNW-UHFFFAOYSA-N 0.000 description 3
- 229910052742 iron Inorganic materials 0.000 description 3
- 229910052750 molybdenum Inorganic materials 0.000 description 3
- 229920000642 polymer Polymers 0.000 description 3
- 239000000843 powder Substances 0.000 description 3
- 238000000197 pyrolysis Methods 0.000 description 3
- 230000009467 reduction Effects 0.000 description 3
- 229910052709 silver Inorganic materials 0.000 description 3
- 239000004332 silver Substances 0.000 description 3
- 238000004659 sterilization and disinfection Methods 0.000 description 3
- 229910052721 tungsten Inorganic materials 0.000 description 3
- NLXLAEXVIDQMFP-UHFFFAOYSA-N Ammonia chloride Chemical compound [NH4+].[Cl-] NLXLAEXVIDQMFP-UHFFFAOYSA-N 0.000 description 2
- XKRFYHLGVUSROY-UHFFFAOYSA-N Argon Chemical compound [Ar] XKRFYHLGVUSROY-UHFFFAOYSA-N 0.000 description 2
- CURLTUGMZLYLDI-UHFFFAOYSA-N Carbon dioxide Chemical compound O=C=O CURLTUGMZLYLDI-UHFFFAOYSA-N 0.000 description 2
- RYGMFSIKBFXOCR-UHFFFAOYSA-N Copper Chemical compound [Cu] RYGMFSIKBFXOCR-UHFFFAOYSA-N 0.000 description 2
- ZRALSGWEFCBTJO-UHFFFAOYSA-N Guanidine Chemical compound NC(N)=N ZRALSGWEFCBTJO-UHFFFAOYSA-N 0.000 description 2
- MHAJPDPJQMAIIY-UHFFFAOYSA-N Hydrogen peroxide Chemical compound OO MHAJPDPJQMAIIY-UHFFFAOYSA-N 0.000 description 2
- ZOKXTWBITQBERF-UHFFFAOYSA-N Molybdenum Chemical compound [Mo] ZOKXTWBITQBERF-UHFFFAOYSA-N 0.000 description 2
- JUJWROOIHBZHMG-UHFFFAOYSA-N Pyridine Chemical compound C1=CC=NC=C1 JUJWROOIHBZHMG-UHFFFAOYSA-N 0.000 description 2
- BQCADISMDOOEFD-UHFFFAOYSA-N Silver Chemical compound [Ag] BQCADISMDOOEFD-UHFFFAOYSA-N 0.000 description 2
- QAOWNCQODCNURD-UHFFFAOYSA-L Sulfate Chemical compound [O-]S([O-])(=O)=O QAOWNCQODCNURD-UHFFFAOYSA-L 0.000 description 2
- RAHZWNYVWXNFOC-UHFFFAOYSA-N Sulphur dioxide Chemical compound O=S=O RAHZWNYVWXNFOC-UHFFFAOYSA-N 0.000 description 2
- YTPLMLYBLZKORZ-UHFFFAOYSA-N Thiophene Chemical compound C=1C=CSC=1 YTPLMLYBLZKORZ-UHFFFAOYSA-N 0.000 description 2
- RTAQQCXQSZGOHL-UHFFFAOYSA-N Titanium Chemical compound [Ti] RTAQQCXQSZGOHL-UHFFFAOYSA-N 0.000 description 2
- 239000004699 Ultra-high molecular weight polyethylene Substances 0.000 description 2
- 239000011149 active material Substances 0.000 description 2
- 229910021529 ammonia Inorganic materials 0.000 description 2
- ROOXNKNUYICQNP-UHFFFAOYSA-N ammonium persulfate Chemical compound [NH4+].[NH4+].[O-]S(=O)(=O)OOS([O-])(=O)=O ROOXNKNUYICQNP-UHFFFAOYSA-N 0.000 description 2
- 238000009835 boiling Methods 0.000 description 2
- 238000011088 calibration curve Methods 0.000 description 2
- 238000005660 chlorination reaction Methods 0.000 description 2
- 239000002131 composite material Substances 0.000 description 2
- 229910052802 copper Inorganic materials 0.000 description 2
- 239000010949 copper Substances 0.000 description 2
- 230000007423 decrease Effects 0.000 description 2
- 239000000645 desinfectant Substances 0.000 description 2
- 238000000502 dialysis Methods 0.000 description 2
- 239000003344 environmental pollutant Substances 0.000 description 2
- 239000012467 final product Substances 0.000 description 2
- 229920001903 high density polyethylene Polymers 0.000 description 2
- 239000004700 high-density polyethylene Substances 0.000 description 2
- 239000010903 husk Substances 0.000 description 2
- WQYVRQLZKVEZGA-UHFFFAOYSA-N hypochlorite Chemical compound Cl[O-] WQYVRQLZKVEZGA-UHFFFAOYSA-N 0.000 description 2
- 230000006872 improvement Effects 0.000 description 2
- 150000002500 ions Chemical class 0.000 description 2
- 238000005259 measurement Methods 0.000 description 2
- 239000011733 molybdenum Substances 0.000 description 2
- 239000012299 nitrogen atmosphere Substances 0.000 description 2
- 231100000719 pollutant Toxicity 0.000 description 2
- 239000012429 reaction media Substances 0.000 description 2
- 238000001179 sorption measurement Methods 0.000 description 2
- 241000894007 species Species 0.000 description 2
- 239000007858 starting material Substances 0.000 description 2
- XTQHKBHJIVJGKJ-UHFFFAOYSA-N sulfur monoxide Chemical class S=O XTQHKBHJIVJGKJ-UHFFFAOYSA-N 0.000 description 2
- UMGDCJDMYOKAJW-UHFFFAOYSA-N thiourea Chemical compound NC(N)=S UMGDCJDMYOKAJW-UHFFFAOYSA-N 0.000 description 2
- 239000010936 titanium Substances 0.000 description 2
- 229910052719 titanium Inorganic materials 0.000 description 2
- WFKWXMTUELFFGS-UHFFFAOYSA-N tungsten Chemical compound [W] WFKWXMTUELFFGS-UHFFFAOYSA-N 0.000 description 2
- 239000010937 tungsten Substances 0.000 description 2
- 229920000785 ultra high molecular weight polyethylene Polymers 0.000 description 2
- 239000012855 volatile organic compound Substances 0.000 description 2
- AOSFMYBATFLTAQ-UHFFFAOYSA-N 1-amino-3-(benzimidazol-1-yl)propan-2-ol Chemical compound C1=CC=C2N(CC(O)CN)C=NC2=C1 AOSFMYBATFLTAQ-UHFFFAOYSA-N 0.000 description 1
- WJFKNYWRSNBZNX-UHFFFAOYSA-N 10H-phenothiazine Chemical compound C1=CC=C2NC3=CC=CC=C3SC2=C1 WJFKNYWRSNBZNX-UHFFFAOYSA-N 0.000 description 1
- QGZKDVFQNNGYKY-UHFFFAOYSA-O Ammonium Chemical compound [NH4+] QGZKDVFQNNGYKY-UHFFFAOYSA-O 0.000 description 1
- OYPRJOBELJOOCE-UHFFFAOYSA-N Calcium Chemical compound [Ca] OYPRJOBELJOOCE-UHFFFAOYSA-N 0.000 description 1
- 101710178035 Chorismate synthase 2 Proteins 0.000 description 1
- YGBZFOQXPOGACY-CUDHKJQZSA-N Communic acid Natural products CC(C=C)=CC[C@H]1C(=C)CC[C@@H]2[C@]1(C)CCC[C@]2(C)C(O)=O YGBZFOQXPOGACY-CUDHKJQZSA-N 0.000 description 1
- 101710152694 Cysteine synthase 2 Proteins 0.000 description 1
- BWGNESOTFCXPMA-UHFFFAOYSA-N Dihydrogen disulfide Chemical compound SS BWGNESOTFCXPMA-UHFFFAOYSA-N 0.000 description 1
- RWSOTUBLDIXVET-UHFFFAOYSA-N Dihydrogen sulfide Chemical compound S RWSOTUBLDIXVET-UHFFFAOYSA-N 0.000 description 1
- 241000196324 Embryophyta Species 0.000 description 1
- 239000012028 Fenton's reagent Substances 0.000 description 1
- MBMLMWLHJBBADN-UHFFFAOYSA-N Ferrous sulfide Chemical compound [Fe]=S MBMLMWLHJBBADN-UHFFFAOYSA-N 0.000 description 1
- 239000004831 Hot glue Substances 0.000 description 1
- RAXXELZNTBOGNW-UHFFFAOYSA-O Imidazolium Chemical compound C1=C[NH+]=CN1 RAXXELZNTBOGNW-UHFFFAOYSA-O 0.000 description 1
- JEGUVXRNDRXUDN-UHFFFAOYSA-N Labdatriene Natural products C1CCC(C)(C(O)=O)C2CCC(=C)C(CCC(=C)C=C)C21C JEGUVXRNDRXUDN-UHFFFAOYSA-N 0.000 description 1
- FYYHWMGAXLPEAU-UHFFFAOYSA-N Magnesium Chemical compound [Mg] FYYHWMGAXLPEAU-UHFFFAOYSA-N 0.000 description 1
- PWHULOQIROXLJO-UHFFFAOYSA-N Manganese Chemical compound [Mn] PWHULOQIROXLJO-UHFFFAOYSA-N 0.000 description 1
- BZLVMXJERCGZMT-UHFFFAOYSA-N Methyl tert-butyl ether Chemical compound COC(C)(C)C BZLVMXJERCGZMT-UHFFFAOYSA-N 0.000 description 1
- 229920001410 Microfiber Polymers 0.000 description 1
- PNUMGDFCIASAFI-UHFFFAOYSA-M N#C[S-].N#CS.[Na+] Chemical compound N#C[S-].N#CS.[Na+] PNUMGDFCIASAFI-UHFFFAOYSA-M 0.000 description 1
- CHJJGSNFBQVOTG-UHFFFAOYSA-N N-methyl-guanidine Natural products CNC(N)=N CHJJGSNFBQVOTG-UHFFFAOYSA-N 0.000 description 1
- GRYLNZFGIOXLOG-UHFFFAOYSA-N Nitric acid Chemical compound O[N+]([O-])=O GRYLNZFGIOXLOG-UHFFFAOYSA-N 0.000 description 1
- BPQQTUXANYXVAA-UHFFFAOYSA-N Orthosilicate Chemical compound [O-][Si]([O-])([O-])[O-] BPQQTUXANYXVAA-UHFFFAOYSA-N 0.000 description 1
- CBENFWSGALASAD-UHFFFAOYSA-N Ozone Chemical compound [O-][O+]=O CBENFWSGALASAD-UHFFFAOYSA-N 0.000 description 1
- 239000004698 Polyethylene Substances 0.000 description 1
- ZLMJMSJWJFRBEC-UHFFFAOYSA-N Potassium Chemical compound [K] ZLMJMSJWJFRBEC-UHFFFAOYSA-N 0.000 description 1
- OEYVFRVNVPKHQQ-UHFFFAOYSA-N Pyrimidin-4-yl-Methanol Chemical compound OCC1=CC=NC=N1 OEYVFRVNVPKHQQ-UHFFFAOYSA-N 0.000 description 1
- 239000005708 Sodium hypochlorite Substances 0.000 description 1
- LSNNMFCWUKXFEE-UHFFFAOYSA-N Sulfurous acid Chemical compound OS(O)=O LSNNMFCWUKXFEE-UHFFFAOYSA-N 0.000 description 1
- UCKMPCXJQFINFW-UHFFFAOYSA-N Sulphide Chemical compound [S-2] UCKMPCXJQFINFW-UHFFFAOYSA-N 0.000 description 1
- 229920010741 Ultra High Molecular Weight Polyethylene (UHMWPE) Polymers 0.000 description 1
- XSQUKJJJFZCRTK-UHFFFAOYSA-N Urea Natural products NC(N)=O XSQUKJJJFZCRTK-UHFFFAOYSA-N 0.000 description 1
- 239000005083 Zinc sulfide Substances 0.000 description 1
- QCWXUUIWCKQGHC-UHFFFAOYSA-N Zirconium Chemical compound [Zr] QCWXUUIWCKQGHC-UHFFFAOYSA-N 0.000 description 1
- ZJVPRPLANXCJHZ-UHFFFAOYSA-N [C].ClN Chemical compound [C].ClN ZJVPRPLANXCJHZ-UHFFFAOYSA-N 0.000 description 1
- SHMVRUMGFMGYGG-UHFFFAOYSA-N [Mo].S=O Chemical compound [Mo].S=O SHMVRUMGFMGYGG-UHFFFAOYSA-N 0.000 description 1
- 239000002253 acid Substances 0.000 description 1
- 230000004913 activation Effects 0.000 description 1
- 238000007792 addition Methods 0.000 description 1
- 239000000654 additive Substances 0.000 description 1
- 230000004075 alteration Effects 0.000 description 1
- 239000012080 ambient air Substances 0.000 description 1
- 235000019270 ammonium chloride Nutrition 0.000 description 1
- HIVLDXAAFGCOFU-UHFFFAOYSA-N ammonium hydrosulfide Chemical compound [NH4+].[SH-] HIVLDXAAFGCOFU-UHFFFAOYSA-N 0.000 description 1
- 229910001870 ammonium persulfate Inorganic materials 0.000 description 1
- BFNBIHQBYMNNAN-UHFFFAOYSA-N ammonium sulfate Chemical compound N.N.OS(O)(=O)=O BFNBIHQBYMNNAN-UHFFFAOYSA-N 0.000 description 1
- 229910052921 ammonium sulfate Inorganic materials 0.000 description 1
- 235000011130 ammonium sulphate Nutrition 0.000 description 1
- XYXNTHIYBIDHGM-UHFFFAOYSA-N ammonium thiosulfate Chemical compound [NH4+].[NH4+].[O-]S([O-])(=O)=S XYXNTHIYBIDHGM-UHFFFAOYSA-N 0.000 description 1
- 238000013459 approach Methods 0.000 description 1
- 229910052786 argon Inorganic materials 0.000 description 1
- 239000011230 binding agent Substances 0.000 description 1
- WVMYSOZCZHQCSG-UHFFFAOYSA-N bis(sulfanylidene)zirconium Chemical compound S=[Zr]=S WVMYSOZCZHQCSG-UHFFFAOYSA-N 0.000 description 1
- 239000011575 calcium Substances 0.000 description 1
- 229910052791 calcium Inorganic materials 0.000 description 1
- 125000004432 carbon atom Chemical group C* 0.000 description 1
- 239000001569 carbon dioxide Substances 0.000 description 1
- 229910002092 carbon dioxide Inorganic materials 0.000 description 1
- DXHPZXWIPWDXHJ-UHFFFAOYSA-N carbon monosulfide Chemical compound [S+]#[C-] DXHPZXWIPWDXHJ-UHFFFAOYSA-N 0.000 description 1
- 239000003575 carbonaceous material Substances 0.000 description 1
- 125000002915 carbonyl group Chemical group [*:2]C([*:1])=O 0.000 description 1
- 239000003610 charcoal Substances 0.000 description 1
- 229910052729 chemical element Inorganic materials 0.000 description 1
- 239000003153 chemical reaction reagent Substances 0.000 description 1
- GRWVQDDAKZFPFI-UHFFFAOYSA-H chromium(III) sulfate Chemical compound [Cr+3].[Cr+3].[O-]S([O-])(=O)=O.[O-]S([O-])(=O)=O.[O-]S([O-])(=O)=O GRWVQDDAKZFPFI-UHFFFAOYSA-H 0.000 description 1
- 239000000567 combustion gas Substances 0.000 description 1
- 238000004891 communication Methods 0.000 description 1
- 238000010924 continuous production Methods 0.000 description 1
- 238000001816 cooling Methods 0.000 description 1
- 229910000365 copper sulfate Inorganic materials 0.000 description 1
- ARUVKPQLZAKDPS-UHFFFAOYSA-L copper(II) sulfate Chemical compound [Cu+2].[O-][S+2]([O-])([O-])[O-] ARUVKPQLZAKDPS-UHFFFAOYSA-L 0.000 description 1
- OMZSGWSJDCOLKM-UHFFFAOYSA-N copper(II) sulfide Chemical compound [S-2].[Cu+2] OMZSGWSJDCOLKM-UHFFFAOYSA-N 0.000 description 1
- 239000011258 core-shell material Substances 0.000 description 1
- XLJMAIOERFSOGZ-UHFFFAOYSA-M cyanate Chemical compound [O-]C#N XLJMAIOERFSOGZ-UHFFFAOYSA-M 0.000 description 1
- 238000013461 design Methods 0.000 description 1
- 238000004455 differential thermal analysis Methods 0.000 description 1
- 238000007416 differential thermogravimetric analysis Methods 0.000 description 1
- SWSQBOPZIKWTGO-UHFFFAOYSA-N dimethylaminoamidine Natural products CN(C)C(N)=N SWSQBOPZIKWTGO-UHFFFAOYSA-N 0.000 description 1
- 229910001882 dioxygen Inorganic materials 0.000 description 1
- 238000010494 dissociation reaction Methods 0.000 description 1
- 230000005593 dissociations Effects 0.000 description 1
- 238000004821 distillation Methods 0.000 description 1
- 238000009826 distribution Methods 0.000 description 1
- TXKMVPPZCYKFAC-UHFFFAOYSA-N disulfur monoxide Inorganic materials O=S=S TXKMVPPZCYKFAC-UHFFFAOYSA-N 0.000 description 1
- 238000007580 dry-mixing Methods 0.000 description 1
- 238000010981 drying operation Methods 0.000 description 1
- 230000000694 effects Effects 0.000 description 1
- 230000007613 environmental effect Effects 0.000 description 1
- 150000002118 epoxides Chemical class 0.000 description 1
- 238000002474 experimental method Methods 0.000 description 1
- 239000000835 fiber Substances 0.000 description 1
- 238000011049 filling Methods 0.000 description 1
- 239000006260 foam Substances 0.000 description 1
- 125000000524 functional group Chemical group 0.000 description 1
- 230000005484 gravity Effects 0.000 description 1
- ZRALSGWEFCBTJO-UHFFFAOYSA-O guanidinium Chemical compound NC(N)=[NH2+] ZRALSGWEFCBTJO-UHFFFAOYSA-O 0.000 description 1
- 230000036541 health Effects 0.000 description 1
- 239000004009 herbicide Substances 0.000 description 1
- 229910000037 hydrogen sulfide Inorganic materials 0.000 description 1
- ZMZDMBWJUHKJPS-UHFFFAOYSA-N hydrogen thiocyanate Natural products SC#N ZMZDMBWJUHKJPS-UHFFFAOYSA-N 0.000 description 1
- QAOWNCQODCNURD-UHFFFAOYSA-M hydrogensulfate Chemical compound OS([O-])(=O)=O QAOWNCQODCNURD-UHFFFAOYSA-M 0.000 description 1
- 238000005470 impregnation Methods 0.000 description 1
- 239000011261 inert gas Substances 0.000 description 1
- 230000003993 interaction Effects 0.000 description 1
- 230000001788 irregular Effects 0.000 description 1
- 229910052747 lanthanoid Inorganic materials 0.000 description 1
- 150000002602 lanthanoids Chemical class 0.000 description 1
- 229920005610 lignin Polymers 0.000 description 1
- 238000011068 loading method Methods 0.000 description 1
- 239000011777 magnesium Substances 0.000 description 1
- 229910052749 magnesium Inorganic materials 0.000 description 1
- 229910052748 manganese Inorganic materials 0.000 description 1
- 239000011572 manganese Substances 0.000 description 1
- 229940099596 manganese sulfate Drugs 0.000 description 1
- 239000011702 manganese sulphate Substances 0.000 description 1
- 235000007079 manganese sulphate Nutrition 0.000 description 1
- WPBNNNQJVZRUHP-UHFFFAOYSA-L manganese(2+);methyl n-[[2-(methoxycarbonylcarbamothioylamino)phenyl]carbamothioyl]carbamate;n-[2-(sulfidocarbothioylamino)ethyl]carbamodithioate Chemical compound [Mn+2].[S-]C(=S)NCCNC([S-])=S.COC(=O)NC(=S)NC1=CC=CC=C1NC(=S)NC(=O)OC WPBNNNQJVZRUHP-UHFFFAOYSA-L 0.000 description 1
- SQQMAOCOWKFBNP-UHFFFAOYSA-L manganese(II) sulfate Chemical compound [Mn+2].[O-]S([O-])(=O)=O SQQMAOCOWKFBNP-UHFFFAOYSA-L 0.000 description 1
- 238000002844 melting Methods 0.000 description 1
- 230000008018 melting Effects 0.000 description 1
- 229910044991 metal oxide Inorganic materials 0.000 description 1
- 150000004706 metal oxides Chemical class 0.000 description 1
- VNWKTOKETHGBQD-UHFFFAOYSA-N methane Chemical compound C VNWKTOKETHGBQD-UHFFFAOYSA-N 0.000 description 1
- 239000003658 microfiber Substances 0.000 description 1
- 238000012986 modification Methods 0.000 description 1
- 230000004048 modification Effects 0.000 description 1
- CWQXQMHSOZUFJS-UHFFFAOYSA-N molybdenum disulfide Chemical compound S=[Mo]=S CWQXQMHSOZUFJS-UHFFFAOYSA-N 0.000 description 1
- YNAVUWVOSKDBBP-UHFFFAOYSA-O morpholinium Chemical compound [H+].C1COCCN1 YNAVUWVOSKDBBP-UHFFFAOYSA-O 0.000 description 1
- 229910052758 niobium Inorganic materials 0.000 description 1
- 239000010955 niobium Substances 0.000 description 1
- GUCVJGMIXFAOAE-UHFFFAOYSA-N niobium atom Chemical compound [Nb] GUCVJGMIXFAOAE-UHFFFAOYSA-N 0.000 description 1
- 229910017604 nitric acid Inorganic materials 0.000 description 1
- 239000011368 organic material Substances 0.000 description 1
- 239000003960 organic solvent Substances 0.000 description 1
- 239000007800 oxidant agent Substances 0.000 description 1
- YGBZFOQXPOGACY-UHFFFAOYSA-N ozic acid Natural products C1CCC(C(O)=O)(C)C2C1(C)C(CC=C(C=C)C)C(=C)CC2 YGBZFOQXPOGACY-UHFFFAOYSA-N 0.000 description 1
- 239000011236 particulate material Substances 0.000 description 1
- 239000003415 peat Substances 0.000 description 1
- 230000000737 periodic effect Effects 0.000 description 1
- 239000002957 persistent organic pollutant Substances 0.000 description 1
- 239000000575 pesticide Substances 0.000 description 1
- 229950000688 phenothiazine Drugs 0.000 description 1
- 239000006069 physical mixture Substances 0.000 description 1
- 229920000573 polyethylene Polymers 0.000 description 1
- 239000005077 polysulfide Substances 0.000 description 1
- 229920001021 polysulfide Polymers 0.000 description 1
- 150000008117 polysulfides Polymers 0.000 description 1
- 239000011591 potassium Substances 0.000 description 1
- 229910052700 potassium Inorganic materials 0.000 description 1
- 239000012286 potassium permanganate Substances 0.000 description 1
- 238000002360 preparation method Methods 0.000 description 1
- 238000000746 purification Methods 0.000 description 1
- UMJSCPRVCHMLSP-UHFFFAOYSA-N pyridine Natural products COC1=CC=CN=C1 UMJSCPRVCHMLSP-UHFFFAOYSA-N 0.000 description 1
- WHMDPDGBKYUEMW-UHFFFAOYSA-N pyridine-2-thiol Chemical compound SC1=CC=CC=N1 WHMDPDGBKYUEMW-UHFFFAOYSA-N 0.000 description 1
- JUJWROOIHBZHMG-UHFFFAOYSA-O pyridinium Chemical compound C1=CC=[NH+]C=C1 JUJWROOIHBZHMG-UHFFFAOYSA-O 0.000 description 1
- 238000011002 quantification Methods 0.000 description 1
- 239000000376 reactant Substances 0.000 description 1
- 239000007845 reactive nitrogen species Substances 0.000 description 1
- 239000012925 reference material Substances 0.000 description 1
- 238000011160 research Methods 0.000 description 1
- 150000003384 small molecules Chemical class 0.000 description 1
- GCLGEJMYGQKIIW-UHFFFAOYSA-H sodium hexametaphosphate Chemical compound [Na]OP1(=O)OP(=O)(O[Na])OP(=O)(O[Na])OP(=O)(O[Na])OP(=O)(O[Na])OP(=O)(O[Na])O1 GCLGEJMYGQKIIW-UHFFFAOYSA-H 0.000 description 1
- SUKJFIGYRHOWBL-UHFFFAOYSA-N sodium hypochlorite Chemical compound [Na+].Cl[O-] SUKJFIGYRHOWBL-UHFFFAOYSA-N 0.000 description 1
- 239000011877 solvent mixture Substances 0.000 description 1
- 238000005507 spraying Methods 0.000 description 1
- IIACRCGMVDHOTQ-UHFFFAOYSA-M sulfamate Chemical compound NS([O-])(=O)=O IIACRCGMVDHOTQ-UHFFFAOYSA-M 0.000 description 1
- CADICXFYUNYKGD-UHFFFAOYSA-N sulfanylidenemanganese Chemical compound [Mn]=S CADICXFYUNYKGD-UHFFFAOYSA-N 0.000 description 1
- NYPFJVOIAWPAAV-UHFFFAOYSA-N sulfanylideneniobium Chemical compound [Nb]=S NYPFJVOIAWPAAV-UHFFFAOYSA-N 0.000 description 1
- 150000003457 sulfones Chemical class 0.000 description 1
- 150000003462 sulfoxides Chemical class 0.000 description 1
- 150000003463 sulfur Chemical class 0.000 description 1
- 239000008400 supply water Substances 0.000 description 1
- 238000006557 surface reaction Methods 0.000 description 1
- 238000005979 thermal decomposition reaction Methods 0.000 description 1
- VOVUARRWDCVURC-UHFFFAOYSA-N thiirane Chemical compound C1CS1 VOVUARRWDCVURC-UHFFFAOYSA-N 0.000 description 1
- BRNULMACUQOKMR-UHFFFAOYSA-N thiomorpholine Chemical compound C1CSCCN1 BRNULMACUQOKMR-UHFFFAOYSA-N 0.000 description 1
- 229930192474 thiophene Natural products 0.000 description 1
- 229940006280 thiosulfate ion Drugs 0.000 description 1
- DHCDFWKWKRSZHF-UHFFFAOYSA-L thiosulfate(2-) Chemical compound [O-]S([S-])(=O)=O DHCDFWKWKRSZHF-UHFFFAOYSA-L 0.000 description 1
- 150000003852 triazoles Chemical class 0.000 description 1
- NRZWQKGABZFFKE-UHFFFAOYSA-N trimethylsulfonium Chemical compound C[S+](C)C NRZWQKGABZFFKE-UHFFFAOYSA-N 0.000 description 1
- YRYSAWZMIRQUBO-UHFFFAOYSA-N trimethylsulfoxonium Chemical compound C[S+](C)(C)=O YRYSAWZMIRQUBO-UHFFFAOYSA-N 0.000 description 1
- ITRNXVSDJBHYNJ-UHFFFAOYSA-N tungsten disulfide Chemical compound S=[W]=S ITRNXVSDJBHYNJ-UHFFFAOYSA-N 0.000 description 1
- 238000011144 upstream manufacturing Methods 0.000 description 1
- 229910052984 zinc sulfide Inorganic materials 0.000 description 1
- DRDVZXDWVBGGMH-UHFFFAOYSA-N zinc;sulfide Chemical compound [S-2].[Zn+2] DRDVZXDWVBGGMH-UHFFFAOYSA-N 0.000 description 1
- 229910052726 zirconium Inorganic materials 0.000 description 1
Images
Classifications
-
- C—CHEMISTRY; METALLURGY
- C02—TREATMENT OF WATER, WASTE WATER, SEWAGE, OR SLUDGE
- C02F—TREATMENT OF WATER, WASTE WATER, SEWAGE, OR SLUDGE
- C02F1/00—Treatment of water, waste water, or sewage
- C02F1/28—Treatment of water, waste water, or sewage by sorption
- C02F1/283—Treatment of water, waste water, or sewage by sorption using coal, charred products, or inorganic mixtures containing them
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B01—PHYSICAL OR CHEMICAL PROCESSES OR APPARATUS IN GENERAL
- B01J—CHEMICAL OR PHYSICAL PROCESSES, e.g. CATALYSIS OR COLLOID CHEMISTRY; THEIR RELEVANT APPARATUS
- B01J20/00—Solid sorbent compositions or filter aid compositions; Sorbents for chromatography; Processes for preparing, regenerating or reactivating thereof
- B01J20/02—Solid sorbent compositions or filter aid compositions; Sorbents for chromatography; Processes for preparing, regenerating or reactivating thereof comprising inorganic material
- B01J20/20—Solid sorbent compositions or filter aid compositions; Sorbents for chromatography; Processes for preparing, regenerating or reactivating thereof comprising inorganic material comprising free carbon; comprising carbon obtained by carbonising processes
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B01—PHYSICAL OR CHEMICAL PROCESSES OR APPARATUS IN GENERAL
- B01J—CHEMICAL OR PHYSICAL PROCESSES, e.g. CATALYSIS OR COLLOID CHEMISTRY; THEIR RELEVANT APPARATUS
- B01J20/00—Solid sorbent compositions or filter aid compositions; Sorbents for chromatography; Processes for preparing, regenerating or reactivating thereof
- B01J20/28—Solid sorbent compositions or filter aid compositions; Sorbents for chromatography; Processes for preparing, regenerating or reactivating thereof characterised by their form or physical properties
- B01J20/28002—Solid sorbent compositions or filter aid compositions; Sorbents for chromatography; Processes for preparing, regenerating or reactivating thereof characterised by their form or physical properties characterised by their physical properties
- B01J20/28011—Other properties, e.g. density, crush strength
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B01—PHYSICAL OR CHEMICAL PROCESSES OR APPARATUS IN GENERAL
- B01J—CHEMICAL OR PHYSICAL PROCESSES, e.g. CATALYSIS OR COLLOID CHEMISTRY; THEIR RELEVANT APPARATUS
- B01J20/00—Solid sorbent compositions or filter aid compositions; Sorbents for chromatography; Processes for preparing, regenerating or reactivating thereof
- B01J20/30—Processes for preparing, regenerating, or reactivating
- B01J20/32—Impregnating or coating ; Solid sorbent compositions obtained from processes involving impregnating or coating
- B01J20/3202—Impregnating or coating ; Solid sorbent compositions obtained from processes involving impregnating or coating characterised by the carrier, support or substrate used for impregnation or coating
- B01J20/3204—Inorganic carriers, supports or substrates
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B01—PHYSICAL OR CHEMICAL PROCESSES OR APPARATUS IN GENERAL
- B01J—CHEMICAL OR PHYSICAL PROCESSES, e.g. CATALYSIS OR COLLOID CHEMISTRY; THEIR RELEVANT APPARATUS
- B01J20/00—Solid sorbent compositions or filter aid compositions; Sorbents for chromatography; Processes for preparing, regenerating or reactivating thereof
- B01J20/30—Processes for preparing, regenerating, or reactivating
- B01J20/32—Impregnating or coating ; Solid sorbent compositions obtained from processes involving impregnating or coating
- B01J20/3231—Impregnating or coating ; Solid sorbent compositions obtained from processes involving impregnating or coating characterised by the coating or impregnating layer
- B01J20/3234—Inorganic material layers
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B01—PHYSICAL OR CHEMICAL PROCESSES OR APPARATUS IN GENERAL
- B01J—CHEMICAL OR PHYSICAL PROCESSES, e.g. CATALYSIS OR COLLOID CHEMISTRY; THEIR RELEVANT APPARATUS
- B01J20/00—Solid sorbent compositions or filter aid compositions; Sorbents for chromatography; Processes for preparing, regenerating or reactivating thereof
- B01J20/30—Processes for preparing, regenerating, or reactivating
- B01J20/32—Impregnating or coating ; Solid sorbent compositions obtained from processes involving impregnating or coating
- B01J20/3231—Impregnating or coating ; Solid sorbent compositions obtained from processes involving impregnating or coating characterised by the coating or impregnating layer
- B01J20/3234—Inorganic material layers
- B01J20/3236—Inorganic material layers containing metal, other than zeolites, e.g. oxides, hydroxides, sulphides or salts
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B01—PHYSICAL OR CHEMICAL PROCESSES OR APPARATUS IN GENERAL
- B01J—CHEMICAL OR PHYSICAL PROCESSES, e.g. CATALYSIS OR COLLOID CHEMISTRY; THEIR RELEVANT APPARATUS
- B01J20/00—Solid sorbent compositions or filter aid compositions; Sorbents for chromatography; Processes for preparing, regenerating or reactivating thereof
- B01J20/30—Processes for preparing, regenerating, or reactivating
- B01J20/32—Impregnating or coating ; Solid sorbent compositions obtained from processes involving impregnating or coating
- B01J20/3231—Impregnating or coating ; Solid sorbent compositions obtained from processes involving impregnating or coating characterised by the coating or impregnating layer
- B01J20/3242—Layers with a functional group, e.g. an affinity material, a ligand, a reactant or a complexing group
- B01J20/3244—Non-macromolecular compounds
- B01J20/3246—Non-macromolecular compounds having a well defined chemical structure
- B01J20/3248—Non-macromolecular compounds having a well defined chemical structure the functional group or the linking, spacer or anchoring group as a whole comprising at least one type of heteroatom selected from a nitrogen, oxygen or sulfur, these atoms not being part of the carrier as such
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B01—PHYSICAL OR CHEMICAL PROCESSES OR APPARATUS IN GENERAL
- B01J—CHEMICAL OR PHYSICAL PROCESSES, e.g. CATALYSIS OR COLLOID CHEMISTRY; THEIR RELEVANT APPARATUS
- B01J20/00—Solid sorbent compositions or filter aid compositions; Sorbents for chromatography; Processes for preparing, regenerating or reactivating thereof
- B01J20/30—Processes for preparing, regenerating, or reactivating
- B01J20/32—Impregnating or coating ; Solid sorbent compositions obtained from processes involving impregnating or coating
- B01J20/3231—Impregnating or coating ; Solid sorbent compositions obtained from processes involving impregnating or coating characterised by the coating or impregnating layer
- B01J20/3242—Layers with a functional group, e.g. an affinity material, a ligand, a reactant or a complexing group
- B01J20/3244—Non-macromolecular compounds
- B01J20/3246—Non-macromolecular compounds having a well defined chemical structure
- B01J20/3248—Non-macromolecular compounds having a well defined chemical structure the functional group or the linking, spacer or anchoring group as a whole comprising at least one type of heteroatom selected from a nitrogen, oxygen or sulfur, these atoms not being part of the carrier as such
- B01J20/3251—Non-macromolecular compounds having a well defined chemical structure the functional group or the linking, spacer or anchoring group as a whole comprising at least one type of heteroatom selected from a nitrogen, oxygen or sulfur, these atoms not being part of the carrier as such comprising at least two different types of heteroatoms selected from nitrogen, oxygen or sulphur
-
- C—CHEMISTRY; METALLURGY
- C02—TREATMENT OF WATER, WASTE WATER, SEWAGE, OR SLUDGE
- C02F—TREATMENT OF WATER, WASTE WATER, SEWAGE, OR SLUDGE
- C02F1/00—Treatment of water, waste water, or sewage
- C02F1/28—Treatment of water, waste water, or sewage by sorption
- C02F1/288—Treatment of water, waste water, or sewage by sorption using composite sorbents, e.g. coated, impregnated, multi-layered
-
- C—CHEMISTRY; METALLURGY
- C02—TREATMENT OF WATER, WASTE WATER, SEWAGE, OR SLUDGE
- C02F—TREATMENT OF WATER, WASTE WATER, SEWAGE, OR SLUDGE
- C02F2101/00—Nature of the contaminant
- C02F2101/30—Organic compounds
-
- C—CHEMISTRY; METALLURGY
- C02—TREATMENT OF WATER, WASTE WATER, SEWAGE, OR SLUDGE
- C02F—TREATMENT OF WATER, WASTE WATER, SEWAGE, OR SLUDGE
- C02F2101/00—Nature of the contaminant
- C02F2101/30—Organic compounds
- C02F2101/36—Organic compounds containing halogen
-
- C—CHEMISTRY; METALLURGY
- C02—TREATMENT OF WATER, WASTE WATER, SEWAGE, OR SLUDGE
- C02F—TREATMENT OF WATER, WASTE WATER, SEWAGE, OR SLUDGE
- C02F2303/00—Specific treatment goals
- C02F2303/18—Removal of treatment agents after treatment
- C02F2303/185—The treatment agent being halogen or a halogenated compound
Landscapes
- Chemical & Material Sciences (AREA)
- Organic Chemistry (AREA)
- Analytical Chemistry (AREA)
- Chemical Kinetics & Catalysis (AREA)
- Inorganic Chemistry (AREA)
- Hydrology & Water Resources (AREA)
- Life Sciences & Earth Sciences (AREA)
- Engineering & Computer Science (AREA)
- Environmental & Geological Engineering (AREA)
- Water Supply & Treatment (AREA)
- Solid-Sorbent Or Filter-Aiding Compositions (AREA)
- Carbon And Carbon Compounds (AREA)
- Water Treatment By Sorption (AREA)
- Physical Water Treatments (AREA)
Description
「a」、「an」、及び「the」は互換可能に使用され、1又はそれよりも多くを意味する。
「及び/又は」は、記載される事例の一方又は両方が起こりうることを示すために使用され、例えば、A及び/又はBは、(A及びB)と(A又はB)とを含む。
本開示の濾過媒体を調製するのに使用される反応性化合物は、硫黄を含む。実施形態において、この反応性化合物は硫黄含有の反応性化合物、又は硫黄及び窒素含有の反応性化合物である。本明細書で使用されるとき、硫黄含有反応性化合物とは、硫黄を含有する任意の反応性物質を指し、これは単体硫黄を含み得る。一実施形態において、追加化合物を加えることができ、例えば窒素含有反応性化合物又は酸素を加えることができる。一実施形態において、この反応性化合物は金属塩であり得る。別の実施形態において、この反応性化合物は金属塩を含まない。
国際特許出願第US2012/052502号(参照により全体が本明細書に組み込まれる)は、単体硫黄、SO2、SOCl2、SO2Cl2、CS2、COS、H2S、及びエチレンスルフィド、及びエポキシドの硫黄類似体などの硫黄含有化合物の使用を開示しており、これらは炭素基材と共に熱処理される。
米国特許仮出願第61/699324号(2012年9月11日出願)(参照により全体が本明細書に組み込まれる)は、硫黄及び窒素含有塩の使用を開示している。一実施形態において、反応性化合物は、式[C]+y x[A]−x yで表わされる塩であり、式中、[C]はカチオン、[A]はアニオン、x及びyは独立に、少なくとも1である。これらの塩は、少なくとも1つの硫黄原子と、少なくとも1つの窒素原子を含む。
炭素基材と共に熱処理に使用される硫黄含有反応性化合物、並びに/又は硫黄及び窒素含有反応性化合物に加えて、更に、例えば窒素含有反応性化合物及び/又は酸素含有反応性化合物などの追加化合物を使用して、本開示の媒体を達成することもできる。
炭素基材は、粒状材料、粉末材料、繊維、チューブ、ウェブ、又はフォームであり得る。
単体炭素の反応は、一般的に、高い活性化エネルギーを示すため、高温で行われる。反応性化合物を炭素基材表面に導入するのに使用される反応は、硫黄化学種(及び、存在する場合は追加の反応性種)を熱により分解し、並びに、炭素基材との反応を可能にするのに十分な温度で、実施され得る。代表的な温度は、少なくとも200、250、300、400、又は更には500℃、かつ最高650、700、800、900、1000、1200又は更には1400℃を含む。本明細書で得られる生成物は、反応生成物又は媒体と呼ばれる。
炭素基材と、硫黄を含む反応性化合物との反応生成物は、本明細書において、互換可能に反応生成物又は媒体と呼ばれる。
一実施形態において、反応生成物は、マトリックス内に配置されて、フィルターを形成する。マトリックスは、チューブ又は水溶液が内部を通ることができるようにする他の構造体の表面上のウェブのポリマー含有複合体ブロックであり得る。一実施形態において、反応生成物は、ポリエチレン(例えば超高分子量ポリエチレン、又は高密度ポリエチレン(HDPE))などの結合材料と共に混合及び圧縮され得る。別の実施形態において、この反応生成物を、ウェブ(例えば、ブローンマイクロファイバ)内に充填してもよく、このウェブは、圧縮してもよいし又はしなくてもよい。これは、例えば、米国公開特許出願第2009/0039028(Eatonら)に記載されており、この全体が本明細書に組み込まれる。
本開示の媒体は、流体流れから、具体的には液体の流体流れから、より具体的には水性流体流れから、クロラミン及び/又は有機化合物を除去するのに使用され得る。
実施形態1.水溶液からクロラミン及び有機化合物を除去するための方法であって、
クロラミン及び有機化合物を含む水溶液を提供する工程と、
水溶液を、多孔質炭素基材を含む媒体に接触させる工程と、を含み、多孔質炭素基材は少なくとも1.5質量%の硫黄を含む、方法。
少なくとも0.5ppmのクロラミン及び有機化合物を含む水溶液を、少なくとも1.5質量%の硫黄を有する多孔質炭素基材を含む媒体に接触させる工程と、溶出物を収集する工程とを含み、溶出物は、0.1ppm未満のクロラミンを含む、方法。
(i)炭素支持体の表面と、(ii)硫黄を含む反応性化合物とを熱処理することにより調製される媒体を提供する工程と、
媒体を、クロラミン及び有機化合物を含む水溶液に接触させる工程とを含む方法であって、
水溶液は、媒体に接触した後、減少した量のクロラミンと、減少した量の有機化合物とを有する、方法。
クロラミン試験
サンプル中の全塩素含有量から、水サンプル中のクロラミン含有量を求めた。全塩素(OCl−及びクロラミン)濃度は、Hach Companyが米国環境保護庁公定法(USEPA Method)330.5に相当すると主張する全塩素分析DPD法(DPD Total Chlorine Method)、Hach Method 8167により測定した。遊離塩素(OCl−)濃度は、Hach Companyが米国環境保護庁公定法330.5に相当すると主張する遊離クロラミン分析DPD法(DPD Free Chloramine Analysis)、Hach Method 8021により定期的に測定した。遊離塩素は無視できる程の低濃度(<0.2ppm)に保たれたため、全塩素分析は、水中のクロラミン濃度の良い推量となると考えられた。すべての試薬及び器具は、標準Hach Methodに記載されたものであり、Hach Company(Loveland,CO)より入手可能であった。
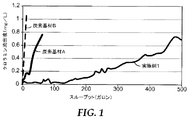
通過流システムにおけるクロラミン処理容量は、通過流試験方法により評価された。pHが7.6±0.25、総溶解固形物が200〜500mg/L、硬度がCaCO3換算で170mg/L未満、濁度が1比濁計濁度単位未満、温度が20±3℃である、クロラミン水溶液試験液を3mg/L、調製した。クロラミン濃度は、次亜塩素酸ナトリウム溶液を加え、次に塩化アンモニウム溶液を加えることにより、2.7〜3.3mg/Lに制御した。pHは、必要により、水酸化ナトリウムを加えることで調整した。
有機化合物を除去する処理容量は、クロロホルムを代理物として使用し、通過流試験方法により評価された。クロロホルムの試験溶液は、300μg/L±30μg/Lの平均クロロホルム濃度で調製した。
サンプル中の炭素、水素、窒素及び硫黄の重量パーセントは、LECO TruSpec Micro CHNS元素分析器(Laboratory Equipment Co.(St.Joseph,MI))を使用して燃焼により測定された。手短に言えば、サンプルを装置に設置し、雰囲気ガスでパージした。その後、サンプルを酸素存在下で1000℃超まで加熱して、サンプルを燃焼させた。その後、更なる酸化、還元、及び粒子除去のために、サンプルを第2炉に通した。その後、炭素、水素、窒素、及び硫黄の含有量を測定するために、燃焼ガスを各種検知器に通した。
サンプルの化学的状態及び元素組成は、X線光電子分光法により、Kratos Axis Ultra(商標)XPSシステム(Shimadzu Corp.(Columbia,MD))を使用し、ベース圧10−9Torr(1.3×10−9kPa)未満で分析された。単色AlKα(1486.6eV(238.18 aJ))X線源は、140ワット(14KV、10mA)で操作された。半球型電子エネルギー分析器は、概略測定では160eV(26aJ)、高解像度スペクトルでは20eV(3aJ)の、一定のパスエネルギーで動作した。結合エネルギー(BE)尺度は、C 1sピークのBEに対して較正された。このスペクトルは、サンプル表面に対して90°の射出角で検出された。データ処理は、PHI MultiPak V8.2B、2006及びCasa XPSバージョン2.3.16 Dev41ソフトウェアで実施した。表面組成は、適切なスコフィールドイオン化断面の補正を行った後、概略測定スペクトルで計測した光電子ピーク面積から計算された。報告されている全体の原子濃度は、無作為に選択された複数のサンプル領域で収集された概略測定スペクトルから誘導された平均値である。触媒官能基の表面成分は、C 1s、O 1s、N 1s及びS 2pコアレベルスペクトルのデコンボリューション/曲線あてはめ分析により決定された。曲線あてはめ分析は、ガウス/ローレンツGL関数の和、及びシャーリー型バックグラウンド除去法に基づいて行われた。
炭素基材Aは、更なる処理なしにそのまま使用した木材原料活性炭(公称80×325メッシュ、MeadWestvaco Specialty Chemicals(North Charleston,SC)より、商標名「AQUAGUARD 325」で入手)であった。炭素基材Aは現在、水中のクロラミン、塩素、味、及びにおいを制御するため特に設計された製品として市販されている。比類のないクロラミン高処理容量を有するとされ、クロラミン低減処理容量が重要であるユースポイント浄水フィルターに選択される触媒性炭素である。製品パンフレット「AQUAGUARD 200 and 325:Catalytic Activated Carbon」(2012年6月改訂)を参照。
炭素基材Bは、ヤシガラ活性炭(公称80×325メッシュ、Kuraray Chemical(Osaka,Japan)より、商標名「PGW100MP」で入手)であった。ヤシガラ活性炭は公称120マイクロメートルの平均粒径を有していた。
炭素基材Bをるつぼ内で180℃に加熱してから、撹拌しながら、単体硫黄(炭素1g当たり硫黄0.2g、Alfa Aesar(Ward Hill,MA)から入手、−325メッシュ、99.5%)を加えた。硫黄は溶融し、炭素基材B内に組み込まれた。
実施例2は、上記の実施例1の記述に従って調製され、上記の「表面分析」方法に従って試験された。実施例2の炭素基材は、炭素91.1原子%、窒素0.6原子%、酸素2.1原子%、及び硫黄5.3原子%を含むことが見出された。サンプル表面の5.3原子パーセントの硫黄のうち、7.4%が酸化状態−2、65.9%が酸化状態0、13.4%が酸化状態+2、9.5%が酸化状態+4、3.8%が酸化状態+6であった。
選択された炭素基材(80×325メッシュ公称粒径)を40cm3、ブレンダーに入れた。最大の非圧縮密度における炭素の体積を測定した。40cm3のTicona GUR 2126超高分子量ポリエチレン(UHMWPE)粉末(Ticona Engineering Polymers(Florence KY)から入手)を、最大の非圧縮密度で測定し、ブレンダーに入れた。この炭素とUHMWPEを3分間混合した。次に、この混合物を、外径1.35in(34.3mm)、内径0.375in(9.5mm)、長さ3.6in(91.4mm)の寸法を有する、コアが円筒状の中空である円筒状の型に定量的に移した。この型は、米国特許第8,206,627号(Stoufferら)に記述されているインパルス充填を使用して、最大の非圧縮密度まで充填した。この型に蓋をしてから、熱対流炉で50分間、180℃で加熱した。加熱後すぐに、この型をピストンで圧縮し、固定長さ3.1in(78.7mm)のブロックにした。この型を室温まで冷まし、得られたカーボンブロックを型から取り出した。ホットメルト接着剤を使用して、端キャップをこのブロックに接着した。
Claims (7)
- 水溶液からクロラミン及び有機化合物を除去するための方法であって、
クロラミン及び有機化合物を含む水溶液を提供する工程と、
前記水溶液を、活性炭を含む媒体に接触させる工程と、を含み、前記活性炭は少なくとも1.5質量%の硫黄を含む、方法。 - 前記活性炭の表面が、化学種COxSyを含み、式中、xは0.1以下、yは0.005〜0.3である、請求項1に記載の方法。
- 前記媒体の少なくとも0.2質量%が、XPS表面分析に基づいて0より大きい酸化状態の硫黄を含む、請求項1または2に記載の方法。
- 前記媒体が、0.6g/ccを超える嵩密度を有する、請求項1ないし3のいずれかに記載の方法。
- 前記媒体が、3%未満の灰分を有する、請求項1ないし4のいずれかに記載の方法。
- 水溶液からクロラミン及び有機化合物を除去するための方法であって、
少なくとも0.5ppmのクロラミン及び有機化合物を含む水溶液を、少なくとも1.5質量%の硫黄を有する活性炭を含む媒体に接触させる工程と、該溶液を収集する工程とを含み、該溶液は、0.1ppm未満のクロラミンを含む、方法。 - (i)活性炭の表面と、(ii)硫黄を含む反応性化合物とを熱処理することにより調製される少なくとも1.5質量%の硫黄を有する活性炭を含む媒体を提供する工程と、
前記媒体を、クロラミン及び有機化合物を含む水溶液に接触させる工程とを含む方法であって、
前記水溶液は、前記媒体に接触した後、減少した量のクロラミンと、減少した量の前記有機化合物とを有する、方法。
Applications Claiming Priority (3)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| US201361777017P | 2013-03-12 | 2013-03-12 | |
| US61/777,017 | 2013-03-12 | ||
| PCT/US2014/021670 WO2014164275A1 (en) | 2013-03-12 | 2014-03-07 | Removal of organic compounds and chloramine from aqueous solutions |
Related Child Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| JP2018140033A Division JP6595054B2 (ja) | 2013-03-12 | 2018-07-26 | 水溶液からの有機化合物及びクロラミンの除去 |
Publications (3)
| Publication Number | Publication Date |
|---|---|
| JP2016518242A JP2016518242A (ja) | 2016-06-23 |
| JP2016518242A5 JP2016518242A5 (ja) | 2017-04-06 |
| JP6407952B2 true JP6407952B2 (ja) | 2018-10-17 |
Family
ID=51658842
Family Applications (2)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| JP2016500803A Active JP6407952B2 (ja) | 2013-03-12 | 2014-03-07 | 水溶液からの有機化合物及びクロラミンの除去 |
| JP2018140033A Active JP6595054B2 (ja) | 2013-03-12 | 2018-07-26 | 水溶液からの有機化合物及びクロラミンの除去 |
Family Applications After (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| JP2018140033A Active JP6595054B2 (ja) | 2013-03-12 | 2018-07-26 | 水溶液からの有機化合物及びクロラミンの除去 |
Country Status (9)
| Country | Link |
|---|---|
| US (1) | US20160016820A1 (ja) |
| EP (1) | EP2969965B1 (ja) |
| JP (2) | JP6407952B2 (ja) |
| KR (1) | KR102239059B1 (ja) |
| CN (1) | CN105189364B (ja) |
| AU (1) | AU2014249524B2 (ja) |
| BR (1) | BR112015022508A2 (ja) |
| CA (1) | CA2904888A1 (ja) |
| WO (1) | WO2014164275A1 (ja) |
Families Citing this family (3)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2014164013A1 (en) | 2013-03-12 | 2014-10-09 | 3M Innovative Properties Company | Removal of chloramine and mercury from aqueous solutions |
| US10953386B2 (en) | 2017-12-29 | 2021-03-23 | Mannon Water (Singapore) PTE. LTD. | Filtration media for removing chloramine, chlorine, and ammonia, and method of making the same |
| WO2019160977A1 (en) * | 2018-02-13 | 2019-08-22 | Calgon Carbon Corporation | Chemical sorbent oxidation method and sorbents made therefrom |
Family Cites Families (20)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JPS5978915A (ja) * | 1982-10-25 | 1984-05-08 | Takeda Chem Ind Ltd | 硫黄担持活性炭の製造法 |
| JP2561368B2 (ja) * | 1990-05-30 | 1996-12-04 | 有限会社みかづき文化会館 | 循環システムおよびこれを用いた循環ろ過装置 |
| US5338458A (en) * | 1993-01-21 | 1994-08-16 | Calgon Carbon Corporation | Method for removing chloramine with catalytic carbon |
| DE9312280U1 (de) * | 1993-08-17 | 1993-10-28 | ETC Engineering & Technology Consulting GmbH, 89331 Burgau | Mittel zur Adsorption von Trihalogenmethanen (Haloformen) und AOX sowie zur Reduzierung von Chloraminen aus Wasser, insbesondere aus Schwimm- und Badebeckenwasser |
| JP3678251B2 (ja) * | 1994-09-13 | 2005-08-03 | 三菱瓦斯化学株式会社 | 高比表面積炭素材料の製造方法 |
| US6270822B1 (en) * | 1997-10-27 | 2001-08-07 | Tekquest Industries | Methods for removal of chlorine from water |
| US6706194B2 (en) * | 2002-05-08 | 2004-03-16 | Meadwestvaco Corporation | Method for removal of chloramines from drinking water |
| EP2021289B1 (en) | 2006-03-22 | 2011-12-14 | 3M Innovative Properties Company | Use of a filter medium |
| US20080047902A1 (en) * | 2006-08-28 | 2008-02-28 | Basf Catalysts Llc | Media for the removal of heavy metals and volatile byproducts from drinking water |
| WO2008027530A1 (en) * | 2006-09-01 | 2008-03-06 | Seldon Technologies, Llc | Nanostructured materials comprising support fibers coated with metal containing compounds and methods of using the same |
| WO2009140033A2 (en) * | 2008-05-14 | 2009-11-19 | 3M Innovative Properties Company | Filter medium and water filtration system including the same |
| TWI505987B (zh) * | 2010-03-31 | 2015-11-01 | Kuraray Chemical Kk | 活性碳及其用途 |
| AU2011264921B2 (en) * | 2010-06-08 | 2014-04-03 | Phillips 66 Company | Heavy metal removal methods and systems using porous supports impregnated with selenium |
| CN102000548A (zh) * | 2010-12-02 | 2011-04-06 | 上海纳米技术及应用国家工程研究中心有限公司 | 一种低浓度有机废气的改性活性炭吸附剂的制备方法 |
| EP2478957A1 (en) * | 2011-01-25 | 2012-07-25 | Norit Nederland B.V. | Production of catalytically active activated carbon |
| AU2012309078B2 (en) * | 2011-09-12 | 2015-11-26 | Solventum Intellectual Properties Company | Filtration medium comprising a carbon oxychalcogenide |
| US9878925B2 (en) * | 2011-12-22 | 2018-01-30 | 3M Innovative Properties Company | Filtration medium comprising a thermolysis product of a carbon oxychalcogenide and a metal salt, method of removing chloramine with this filtration medium and method of making this filtration medium |
| JP6148343B2 (ja) * | 2012-09-11 | 2017-06-14 | スリーエム イノベイティブ プロパティズ カンパニー | 窒素及び硫黄を含む濾材 |
| AU2014249631B2 (en) * | 2013-03-12 | 2016-11-24 | 3M Innovative Properties Company | Removal of chlorine and/or chloramine from aqueous solutions |
| WO2014164013A1 (en) * | 2013-03-12 | 2014-10-09 | 3M Innovative Properties Company | Removal of chloramine and mercury from aqueous solutions |
-
2014
- 2014-03-07 EP EP14779165.1A patent/EP2969965B1/en active Active
- 2014-03-07 AU AU2014249524A patent/AU2014249524B2/en not_active Ceased
- 2014-03-07 CN CN201480014561.5A patent/CN105189364B/zh active Active
- 2014-03-07 WO PCT/US2014/021670 patent/WO2014164275A1/en active Application Filing
- 2014-03-07 CA CA2904888A patent/CA2904888A1/en not_active Abandoned
- 2014-03-07 JP JP2016500803A patent/JP6407952B2/ja active Active
- 2014-03-07 US US14/773,469 patent/US20160016820A1/en not_active Abandoned
- 2014-03-07 BR BR112015022508A patent/BR112015022508A2/pt not_active IP Right Cessation
- 2014-03-07 KR KR1020157027475A patent/KR102239059B1/ko active IP Right Grant
-
2018
- 2018-07-26 JP JP2018140033A patent/JP6595054B2/ja active Active
Also Published As
| Publication number | Publication date |
|---|---|
| KR102239059B1 (ko) | 2021-04-12 |
| AU2014249524B2 (en) | 2017-01-19 |
| EP2969965A4 (en) | 2016-08-10 |
| AU2014249524A1 (en) | 2015-10-01 |
| US20160016820A1 (en) | 2016-01-21 |
| JP6595054B2 (ja) | 2019-10-23 |
| EP2969965B1 (en) | 2020-08-05 |
| JP2016518242A (ja) | 2016-06-23 |
| JP2018183781A (ja) | 2018-11-22 |
| CN105189364A (zh) | 2015-12-23 |
| CA2904888A1 (en) | 2014-10-09 |
| BR112015022508A2 (pt) | 2017-07-18 |
| EP2969965A1 (en) | 2016-01-20 |
| CN105189364B (zh) | 2019-01-08 |
| WO2014164275A1 (en) | 2014-10-09 |
| KR20150127157A (ko) | 2015-11-16 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| JP6611698B2 (ja) | 水溶液からの塩素及び/又はクロラミンの除去 | |
| US20200115255A1 (en) | Filtration medium comprising nitrogen and sulfur | |
| EP2755753B1 (en) | Filtration medium comprising a carbon oxychalcogenide | |
| JP6595054B2 (ja) | 水溶液からの有機化合物及びクロラミンの除去 | |
| JP6416185B2 (ja) | 水溶液からのクロラミン及び水銀の除去 |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| RD03 | Notification of appointment of power of attorney |
Free format text: JAPANESE INTERMEDIATE CODE: A7423 Effective date: 20170110 |
|
| RD04 | Notification of resignation of power of attorney |
Free format text: JAPANESE INTERMEDIATE CODE: A7424 Effective date: 20170117 |
|
| A521 | Request for written amendment filed |
Free format text: JAPANESE INTERMEDIATE CODE: A523 Effective date: 20170302 |
|
| A621 | Written request for application examination |
Free format text: JAPANESE INTERMEDIATE CODE: A621 Effective date: 20170302 |
|
| A977 | Report on retrieval |
Free format text: JAPANESE INTERMEDIATE CODE: A971007 Effective date: 20170627 |
|
| A131 | Notification of reasons for refusal |
Free format text: JAPANESE INTERMEDIATE CODE: A131 Effective date: 20170711 |
|
| A521 | Request for written amendment filed |
Free format text: JAPANESE INTERMEDIATE CODE: A523 Effective date: 20171006 |
|
| A02 | Decision of refusal |
Free format text: JAPANESE INTERMEDIATE CODE: A02 Effective date: 20180327 |
|
| A521 | Request for written amendment filed |
Free format text: JAPANESE INTERMEDIATE CODE: A523 Effective date: 20180726 |
|
| A911 | Transfer to examiner for re-examination before appeal (zenchi) |
Free format text: JAPANESE INTERMEDIATE CODE: A911 Effective date: 20180802 |
|
| TRDD | Decision of grant or rejection written | ||
| A01 | Written decision to grant a patent or to grant a registration (utility model) |
Free format text: JAPANESE INTERMEDIATE CODE: A01 Effective date: 20180821 |
|
| A61 | First payment of annual fees (during grant procedure) |
Free format text: JAPANESE INTERMEDIATE CODE: A61 Effective date: 20180919 |
|
| R150 | Certificate of patent or registration of utility model |
Ref document number: 6407952 Country of ref document: JP Free format text: JAPANESE INTERMEDIATE CODE: R150 |
|
| R250 | Receipt of annual fees |
Free format text: JAPANESE INTERMEDIATE CODE: R250 |
|
| R250 | Receipt of annual fees |
Free format text: JAPANESE INTERMEDIATE CODE: R250 |
|
| S111 | Request for change of ownership or part of ownership |
Free format text: JAPANESE INTERMEDIATE CODE: R313111 |
|
| R250 | Receipt of annual fees |
Free format text: JAPANESE INTERMEDIATE CODE: R250 |