JP5770183B2 - 生体液からの酸素除去 - Google Patents
生体液からの酸素除去 Download PDFInfo
- Publication number
- JP5770183B2 JP5770183B2 JP2012523121A JP2012523121A JP5770183B2 JP 5770183 B2 JP5770183 B2 JP 5770183B2 JP 2012523121 A JP2012523121 A JP 2012523121A JP 2012523121 A JP2012523121 A JP 2012523121A JP 5770183 B2 JP5770183 B2 JP 5770183B2
- Authority
- JP
- Japan
- Prior art keywords
- oxygen
- fluid
- microporous
- hollow tubes
- hydrophobic hollow
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Expired - Fee Related
Links
- QVGXLLKOCUKJST-UHFFFAOYSA-N atomic oxygen Chemical compound [O] QVGXLLKOCUKJST-UHFFFAOYSA-N 0.000 title claims description 153
- 239000001301 oxygen Substances 0.000 title claims description 153
- 229910052760 oxygen Inorganic materials 0.000 title claims description 153
- 239000013060 biological fluid Substances 0.000 title description 13
- 210000003743 erythrocyte Anatomy 0.000 claims description 94
- 239000012530 fluid Substances 0.000 claims description 78
- 239000006285 cell suspension Substances 0.000 claims description 53
- 239000007789 gas Substances 0.000 claims description 39
- 230000002209 hydrophobic effect Effects 0.000 claims description 24
- 238000009792 diffusion process Methods 0.000 claims description 15
- 238000000034 method Methods 0.000 claims description 14
- 239000006096 absorbing agent Substances 0.000 claims description 10
- 239000011148 porous material Substances 0.000 claims description 6
- 239000004743 Polypropylene Substances 0.000 claims description 3
- -1 polypropylene Polymers 0.000 claims description 3
- 229920001155 polypropylene Polymers 0.000 claims description 3
- 239000000835 fiber Substances 0.000 description 111
- 239000012510 hollow fiber Substances 0.000 description 61
- 239000003463 adsorbent Substances 0.000 description 58
- 210000004369 blood Anatomy 0.000 description 33
- 239000008280 blood Substances 0.000 description 33
- 238000003860 storage Methods 0.000 description 16
- 239000012528 membrane Substances 0.000 description 14
- 239000000725 suspension Substances 0.000 description 14
- 238000012545 processing Methods 0.000 description 11
- XKRFYHLGVUSROY-UHFFFAOYSA-N Argon Chemical compound [Ar] XKRFYHLGVUSROY-UHFFFAOYSA-N 0.000 description 10
- 239000012159 carrier gas Substances 0.000 description 10
- 230000036961 partial effect Effects 0.000 description 9
- IJGRMHOSHXDMSA-UHFFFAOYSA-N Atomic nitrogen Chemical compound N#N IJGRMHOSHXDMSA-UHFFFAOYSA-N 0.000 description 8
- 239000002245 particle Substances 0.000 description 7
- 239000000463 material Substances 0.000 description 6
- 108010054147 Hemoglobins Proteins 0.000 description 5
- 102000001554 Hemoglobins Human genes 0.000 description 5
- 229910052786 argon Inorganic materials 0.000 description 5
- 239000000306 component Substances 0.000 description 5
- 238000013461 design Methods 0.000 description 5
- 230000000694 effects Effects 0.000 description 5
- 230000004907 flux Effects 0.000 description 5
- 239000011261 inert gas Substances 0.000 description 5
- 239000003146 anticoagulant agent Substances 0.000 description 4
- 229940127219 anticoagulant drug Drugs 0.000 description 4
- 238000006243 chemical reaction Methods 0.000 description 4
- 238000004891 communication Methods 0.000 description 4
- 238000005516 engineering process Methods 0.000 description 4
- 210000000265 leukocyte Anatomy 0.000 description 4
- 229910052757 nitrogen Inorganic materials 0.000 description 4
- 230000008569 process Effects 0.000 description 4
- 238000012360 testing method Methods 0.000 description 4
- 230000002745 absorbent Effects 0.000 description 3
- 239000002250 absorbent Substances 0.000 description 3
- 239000000654 additive Substances 0.000 description 3
- 239000012503 blood component Substances 0.000 description 3
- 230000003247 decreasing effect Effects 0.000 description 3
- 238000010586 diagram Methods 0.000 description 3
- 238000009826 distribution Methods 0.000 description 3
- 239000004744 fabric Substances 0.000 description 3
- 238000004519 manufacturing process Methods 0.000 description 3
- 239000000203 mixture Substances 0.000 description 3
- 206010018910 Haemolysis Diseases 0.000 description 2
- XEEYBQQBJWHFJM-UHFFFAOYSA-N Iron Chemical compound [Fe] XEEYBQQBJWHFJM-UHFFFAOYSA-N 0.000 description 2
- 108010064719 Oxyhemoglobins Proteins 0.000 description 2
- 230000000996 additive effect Effects 0.000 description 2
- 239000000853 adhesive Substances 0.000 description 2
- 230000001070 adhesive effect Effects 0.000 description 2
- 230000008859 change Effects 0.000 description 2
- 230000007423 decrease Effects 0.000 description 2
- 238000010494 dissociation reaction Methods 0.000 description 2
- 230000005593 dissociations Effects 0.000 description 2
- 230000005484 gravity Effects 0.000 description 2
- 230000036541 health Effects 0.000 description 2
- 230000008588 hemolysis Effects 0.000 description 2
- 238000011835 investigation Methods 0.000 description 2
- 238000012986 modification Methods 0.000 description 2
- 230000004048 modification Effects 0.000 description 2
- 239000004417 polycarbonate Substances 0.000 description 2
- 229920000515 polycarbonate Polymers 0.000 description 2
- 208000007056 sickle cell anemia Diseases 0.000 description 2
- DHKHKXVYLBGOIT-UHFFFAOYSA-N 1,1-Diethoxyethane Chemical compound CCOC(C)OCC DHKHKXVYLBGOIT-UHFFFAOYSA-N 0.000 description 1
- 229920004943 Delrin® Polymers 0.000 description 1
- 241000282412 Homo Species 0.000 description 1
- 241001465754 Metazoa Species 0.000 description 1
- 208000018020 Sickle cell-beta-thalassemia disease syndrome Diseases 0.000 description 1
- FAPWRFPIFSIZLT-UHFFFAOYSA-M Sodium chloride Chemical compound [Na+].[Cl-] FAPWRFPIFSIZLT-UHFFFAOYSA-M 0.000 description 1
- 206010043391 Thalassaemia beta Diseases 0.000 description 1
- 239000011358 absorbing material Substances 0.000 description 1
- 239000011354 acetal resin Substances 0.000 description 1
- 238000007792 addition Methods 0.000 description 1
- 230000002411 adverse Effects 0.000 description 1
- WYTGDNHDOZPMIW-RCBQFDQVSA-N alstonine Natural products C1=CC2=C3C=CC=CC3=NC2=C2N1C[C@H]1[C@H](C)OC=C(C(=O)OC)[C@H]1C2 WYTGDNHDOZPMIW-RCBQFDQVSA-N 0.000 description 1
- 238000013459 approach Methods 0.000 description 1
- 210000002565 arteriole Anatomy 0.000 description 1
- 230000009286 beneficial effect Effects 0.000 description 1
- 230000005540 biological transmission Effects 0.000 description 1
- 230000036760 body temperature Effects 0.000 description 1
- 230000015556 catabolic process Effects 0.000 description 1
- 210000004027 cell Anatomy 0.000 description 1
- 230000001684 chronic effect Effects 0.000 description 1
- 230000006378 damage Effects 0.000 description 1
- 238000006731 degradation reaction Methods 0.000 description 1
- 230000012953 feeding on blood of other organism Effects 0.000 description 1
- 238000007667 floating Methods 0.000 description 1
- 210000002216 heart Anatomy 0.000 description 1
- 238000010348 incorporation Methods 0.000 description 1
- 238000009434 installation Methods 0.000 description 1
- 229910052742 iron Inorganic materials 0.000 description 1
- 239000007788 liquid Substances 0.000 description 1
- 210000004072 lung Anatomy 0.000 description 1
- 238000013178 mathematical model Methods 0.000 description 1
- 238000012856 packing Methods 0.000 description 1
- 230000010412 perfusion Effects 0.000 description 1
- 239000004033 plastic Substances 0.000 description 1
- 229920003023 plastic Polymers 0.000 description 1
- 229920000642 polymer Polymers 0.000 description 1
- 239000002861 polymer material Substances 0.000 description 1
- 229920006324 polyoxymethylene Polymers 0.000 description 1
- 229920002635 polyurethane Polymers 0.000 description 1
- 239000004814 polyurethane Substances 0.000 description 1
- 238000004382 potting Methods 0.000 description 1
- 238000011084 recovery Methods 0.000 description 1
- 230000002829 reductive effect Effects 0.000 description 1
- 230000000717 retained effect Effects 0.000 description 1
- 230000002441 reversible effect Effects 0.000 description 1
- 238000004904 shortening Methods 0.000 description 1
- 239000011780 sodium chloride Substances 0.000 description 1
- 238000010408 sweeping Methods 0.000 description 1
- 238000002560 therapeutic procedure Methods 0.000 description 1
- 239000011800 void material Substances 0.000 description 1
Images
Classifications
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B01—PHYSICAL OR CHEMICAL PROCESSES OR APPARATUS IN GENERAL
- B01D—SEPARATION
- B01D63/00—Apparatus in general for separation processes using semi-permeable membranes
- B01D63/02—Hollow fibre modules
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B01—PHYSICAL OR CHEMICAL PROCESSES OR APPARATUS IN GENERAL
- B01D—SEPARATION
- B01D63/00—Apparatus in general for separation processes using semi-permeable membranes
- B01D63/02—Hollow fibre modules
- B01D63/021—Manufacturing thereof
- B01D63/022—Encapsulating hollow fibres
- B01D63/0221—Encapsulating hollow fibres using a mould
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61M—DEVICES FOR INTRODUCING MEDIA INTO, OR ONTO, THE BODY; DEVICES FOR TRANSDUCING BODY MEDIA OR FOR TAKING MEDIA FROM THE BODY; DEVICES FOR PRODUCING OR ENDING SLEEP OR STUPOR
- A61M1/00—Suction or pumping devices for medical purposes; Devices for carrying-off, for treatment of, or for carrying-over, body-liquids; Drainage systems
- A61M1/02—Blood transfusion apparatus
- A61M1/0209—Multiple bag systems for separating or storing blood components
- A61M1/0218—Multiple bag systems for separating or storing blood components with filters
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61M—DEVICES FOR INTRODUCING MEDIA INTO, OR ONTO, THE BODY; DEVICES FOR TRANSDUCING BODY MEDIA OR FOR TAKING MEDIA FROM THE BODY; DEVICES FOR PRODUCING OR ENDING SLEEP OR STUPOR
- A61M1/00—Suction or pumping devices for medical purposes; Devices for carrying-off, for treatment of, or for carrying-over, body-liquids; Drainage systems
- A61M1/34—Filtering material out of the blood by passing it through a membrane, i.e. hemofiltration or diafiltration
- A61M1/3472—Filtering material out of the blood by passing it through a membrane, i.e. hemofiltration or diafiltration with treatment of the filtrate
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61M—DEVICES FOR INTRODUCING MEDIA INTO, OR ONTO, THE BODY; DEVICES FOR TRANSDUCING BODY MEDIA OR FOR TAKING MEDIA FROM THE BODY; DEVICES FOR PRODUCING OR ENDING SLEEP OR STUPOR
- A61M1/00—Suction or pumping devices for medical purposes; Devices for carrying-off, for treatment of, or for carrying-over, body-liquids; Drainage systems
- A61M1/34—Filtering material out of the blood by passing it through a membrane, i.e. hemofiltration or diafiltration
- A61M1/3472—Filtering material out of the blood by passing it through a membrane, i.e. hemofiltration or diafiltration with treatment of the filtrate
- A61M1/3475—Filtering material out of the blood by passing it through a membrane, i.e. hemofiltration or diafiltration with treatment of the filtrate with filtrate treatment agent in the same enclosure as the membrane
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61M—DEVICES FOR INTRODUCING MEDIA INTO, OR ONTO, THE BODY; DEVICES FOR TRANSDUCING BODY MEDIA OR FOR TAKING MEDIA FROM THE BODY; DEVICES FOR PRODUCING OR ENDING SLEEP OR STUPOR
- A61M1/00—Suction or pumping devices for medical purposes; Devices for carrying-off, for treatment of, or for carrying-over, body-liquids; Drainage systems
- A61M1/34—Filtering material out of the blood by passing it through a membrane, i.e. hemofiltration or diafiltration
- A61M1/3472—Filtering material out of the blood by passing it through a membrane, i.e. hemofiltration or diafiltration with treatment of the filtrate
- A61M1/3486—Biological, chemical treatment, e.g. chemical precipitation; treatment by absorbents
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61M—DEVICES FOR INTRODUCING MEDIA INTO, OR ONTO, THE BODY; DEVICES FOR TRANSDUCING BODY MEDIA OR FOR TAKING MEDIA FROM THE BODY; DEVICES FOR PRODUCING OR ENDING SLEEP OR STUPOR
- A61M1/00—Suction or pumping devices for medical purposes; Devices for carrying-off, for treatment of, or for carrying-over, body-liquids; Drainage systems
- A61M1/36—Other treatment of blood in a by-pass of the natural circulatory system, e.g. temperature adaptation, irradiation ; Extra-corporeal blood circuits
- A61M1/3679—Other treatment of blood in a by-pass of the natural circulatory system, e.g. temperature adaptation, irradiation ; Extra-corporeal blood circuits by absorption
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B01—PHYSICAL OR CHEMICAL PROCESSES OR APPARATUS IN GENERAL
- B01D—SEPARATION
- B01D63/00—Apparatus in general for separation processes using semi-permeable membranes
- B01D63/02—Hollow fibre modules
- B01D63/04—Hollow fibre modules comprising multiple hollow fibre assemblies
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61M—DEVICES FOR INTRODUCING MEDIA INTO, OR ONTO, THE BODY; DEVICES FOR TRANSDUCING BODY MEDIA OR FOR TAKING MEDIA FROM THE BODY; DEVICES FOR PRODUCING OR ENDING SLEEP OR STUPOR
- A61M1/00—Suction or pumping devices for medical purposes; Devices for carrying-off, for treatment of, or for carrying-over, body-liquids; Drainage systems
- A61M1/02—Blood transfusion apparatus
- A61M1/0209—Multiple bag systems for separating or storing blood components
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61M—DEVICES FOR INTRODUCING MEDIA INTO, OR ONTO, THE BODY; DEVICES FOR TRANSDUCING BODY MEDIA OR FOR TAKING MEDIA FROM THE BODY; DEVICES FOR PRODUCING OR ENDING SLEEP OR STUPOR
- A61M1/00—Suction or pumping devices for medical purposes; Devices for carrying-off, for treatment of, or for carrying-over, body-liquids; Drainage systems
- A61M1/02—Blood transfusion apparatus
- A61M1/0272—Apparatus for treatment of blood or blood constituents prior to or for conservation, e.g. freezing, drying or centrifuging
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61M—DEVICES FOR INTRODUCING MEDIA INTO, OR ONTO, THE BODY; DEVICES FOR TRANSDUCING BODY MEDIA OR FOR TAKING MEDIA FROM THE BODY; DEVICES FOR PRODUCING OR ENDING SLEEP OR STUPOR
- A61M1/00—Suction or pumping devices for medical purposes; Devices for carrying-off, for treatment of, or for carrying-over, body-liquids; Drainage systems
- A61M1/02—Blood transfusion apparatus
- A61M1/0281—Apparatus for treatment of blood or blood constituents prior to transfusion, e.g. washing, filtering or thawing
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61M—DEVICES FOR INTRODUCING MEDIA INTO, OR ONTO, THE BODY; DEVICES FOR TRANSDUCING BODY MEDIA OR FOR TAKING MEDIA FROM THE BODY; DEVICES FOR PRODUCING OR ENDING SLEEP OR STUPOR
- A61M1/00—Suction or pumping devices for medical purposes; Devices for carrying-off, for treatment of, or for carrying-over, body-liquids; Drainage systems
- A61M1/14—Dialysis systems; Artificial kidneys; Blood oxygenators ; Reciprocating systems for treatment of body fluids, e.g. single needle systems for hemofiltration or pheresis
- A61M1/16—Dialysis systems; Artificial kidneys; Blood oxygenators ; Reciprocating systems for treatment of body fluids, e.g. single needle systems for hemofiltration or pheresis with membranes
- A61M1/1621—Constructional aspects thereof
- A61M1/1623—Disposition or location of membranes relative to fluids
- A61M1/1625—Dialyser of the outside perfusion type, i.e. blood flow outside hollow membrane fibres or tubes
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61M—DEVICES FOR INTRODUCING MEDIA INTO, OR ONTO, THE BODY; DEVICES FOR TRANSDUCING BODY MEDIA OR FOR TAKING MEDIA FROM THE BODY; DEVICES FOR PRODUCING OR ENDING SLEEP OR STUPOR
- A61M1/00—Suction or pumping devices for medical purposes; Devices for carrying-off, for treatment of, or for carrying-over, body-liquids; Drainage systems
- A61M1/14—Dialysis systems; Artificial kidneys; Blood oxygenators ; Reciprocating systems for treatment of body fluids, e.g. single needle systems for hemofiltration or pheresis
- A61M1/16—Dialysis systems; Artificial kidneys; Blood oxygenators ; Reciprocating systems for treatment of body fluids, e.g. single needle systems for hemofiltration or pheresis with membranes
- A61M1/1621—Constructional aspects thereof
- A61M1/1623—Disposition or location of membranes relative to fluids
- A61M1/1627—Dialyser of the inside perfusion type, i.e. blood flow inside hollow membrane fibres or tubes
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61M—DEVICES FOR INTRODUCING MEDIA INTO, OR ONTO, THE BODY; DEVICES FOR TRANSDUCING BODY MEDIA OR FOR TAKING MEDIA FROM THE BODY; DEVICES FOR PRODUCING OR ENDING SLEEP OR STUPOR
- A61M2202/00—Special media to be introduced, removed or treated
- A61M2202/02—Gases
- A61M2202/0208—Oxygen
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B01—PHYSICAL OR CHEMICAL PROCESSES OR APPARATUS IN GENERAL
- B01D—SEPARATION
- B01D2311/00—Details relating to membrane separation process operations and control
- B01D2311/13—Use of sweep gas
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B01—PHYSICAL OR CHEMICAL PROCESSES OR APPARATUS IN GENERAL
- B01D—SEPARATION
- B01D2311/00—Details relating to membrane separation process operations and control
- B01D2311/26—Further operations combined with membrane separation processes
- B01D2311/2626—Absorption or adsorption
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B01—PHYSICAL OR CHEMICAL PROCESSES OR APPARATUS IN GENERAL
- B01D—SEPARATION
- B01D2313/00—Details relating to membrane modules or apparatus
- B01D2313/40—Adsorbents within the flow path
Description
本願は、2009年7月31日付で出願された米国仮特許出願第61/230,575号の権利を主張し、この特許出願の開示が参照によって本明細書に組み込まれる。
本発明は、米国国立衛生研究所の米国心肺血液研究所によってNew Health Sciences社に授与された助成金番号R44HL088848−01に基づいて政府支援で行われた。政府は、本発明にある種の権利を有している。
・R(ファイバ内径)と、
・pO2in(入口酸素分圧)と、
・▲D▼(血液中の酸素の拡散率)と、
・Vmax(ファイバ中の血液の速度)と、
・f(p*)=(BCHb/σ)dSO2/dpO2(CHbは、ヘモグロビンの濃度であり、Bは、ヘモグロビンの酸素容量であり、σは、血液中の酸素の溶解度であり、dSO2/dpO2は、オキシヘモグロビン解離曲線の勾配である)と、
を含む。
Claims (20)
- 赤血球を含む流体中の酸素の濃度を低減する装置であって、
ハウジングと、
流体入口と、
流体出口と、
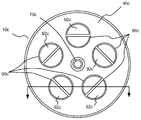
前記ハウジングの内部に延在し、前記流体の流れに適合する、前記流体入口及び前記流体出口と流体接続している、複数の微小孔性で疎水性の中空管と、
前記複数の微小孔性で疎水性の中空管の中を流れる前記流体から酸素の輸送を容易化するために前記複数の微小孔性で疎水性の中空管の外面で酸素の濃度を低減する運搬システムと、を備え、
前記複数の微小孔性で疎水性の中空管は、0.01μmから0.5μmまでの範囲の細孔径を有する、装置。 - 前記ハウジングは、さらに、運搬入口と、運搬出口と、を備える、請求項1記載の装置。
- 前記運搬システムは、酸素以外のガスを含む流体である、請求項1記載の装置。
- 前記複数の微小孔性で疎水性の中空管は、150μmから200μmまでの範囲の管外径を有する、請求項1記載の装置。
- 前記複数の微小孔性で疎水性の中空管は、0.1μmから0.4μmの範囲の細孔径を有する、請求項1記載の装置。
- 前記複数の微小孔性で疎水性の中空管は、5000本から8000本までの数の範囲に及ぶ、請求項1記載の装置。
- 前記複数の微小孔性で疎水性の中空管は、10cmから50cmまでの長さの範囲に及ぶ、請求項1記載の装置。
- 前記運搬システムは、ポンプによって供給される酸素以外のガス、及び、加圧容器によって供給される酸素以外のガスからなる群から選択される流体又は真空源を含む、請求項3記載の装置。
- 前記運搬システムは、酸素吸収材を含む、請求項1記載の装置。
- 前記酸素吸収材は、前記複数の微小孔性で疎水性の中空管の少なくとも一部分の外面に固定化されている、請求項9記載の装置。
- 前記酸素吸収材は、前記複数の微小孔性で疎水性の中空管の外部にある前記ハウジングの容積内に位置付けられている、請求項9記載の装置。
- 複数の前記複数の微小孔性で疎水性の中空管が一つ以上の群として配置され、前記酸素吸収材は、前記一つ以上の群の長さの少なくとも一部分を囲む、請求項9記載の装置。
- 複数の管が前記複数の微小孔性で疎水性の中空管の長さの少なくとも一部分に亘って前記酸素吸収材の周囲の周りに配置されている、請求項9記載の装置。
- 前記運搬システムは、流体処理システムの流体経路の内部に接続可能である、請求項1記載の装置。
- 前記流体は、赤血球懸濁液を含む、請求項1記載の装置。
- 赤血球を含む流体中の酸素の濃度を低減する方法であって、
ハウジングの内部に延在する複数の微小孔性で疎水性の中空管内に前記流体を流し、
前記複数の微小孔性で疎水性の中空管は、0.01μmから0.5μmまでの範囲の細孔径を有し、
前記複数の微小孔性で疎水性の中空管の外面が、酸素の拡散を促進するように濃度勾配を保つことによって、前記流体より酸素の濃度を低く保つ運搬システムに流体接続されている、方法。 - 前記流体は、赤血球懸濁液を含む、請求項16記載の方法。
- 赤血球を含む流体中の酸素の濃度を低減する装置であって、
ハウジングと、
前記ハウジングの内部に延在し、複数の微小孔性で疎水性の中空管のルーメン内部にある酸素吸収材を有する、前記複数の微小孔性で疎水性の中空管と、
前記複数の微小孔性で疎水性の中空管の外部にある前記ハウジングの容積と流体接続している流体入口と、
前記容積と流体接続している流体出口と、
を備え、
前記複数の微小孔性で疎水性の中空管は、0.01μmから0.5μmまでの範囲の細孔径を有する、装置。 - 前記複数の微小孔性で疎水性の中空管は、ポンプ、加圧容器、及び真空源からなる群から選択されるデバイスと流体接続する、請求項1記載の装置。
- 前記複数の微小孔性で疎水性の中空管は、ポリプロピレンからなる、請求項1又は18記載の装置。
Applications Claiming Priority (3)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| US23057509P | 2009-07-31 | 2009-07-31 | |
| US61/230,575 | 2009-07-31 | ||
| PCT/US2010/044045 WO2011014855A2 (en) | 2009-07-31 | 2010-07-31 | Removal of oxygen from biological fluids |
Publications (2)
| Publication Number | Publication Date |
|---|---|
| JP2013500794A JP2013500794A (ja) | 2013-01-10 |
| JP5770183B2 true JP5770183B2 (ja) | 2015-08-26 |
Family
ID=43529974
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| JP2012523121A Expired - Fee Related JP5770183B2 (ja) | 2009-07-31 | 2010-07-31 | 生体液からの酸素除去 |
Country Status (7)
| Country | Link |
|---|---|
| US (1) | US20120129149A1 (ja) |
| EP (1) | EP2459247A4 (ja) |
| JP (1) | JP5770183B2 (ja) |
| AU (1) | AU2010278768A1 (ja) |
| CA (1) | CA2772320A1 (ja) |
| NZ (1) | NZ598410A (ja) |
| WO (1) | WO2011014855A2 (ja) |
Families Citing this family (18)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US8828226B2 (en) | 2003-03-01 | 2014-09-09 | The Trustees Of Boston University | System for assessing the efficacy of stored red blood cells using microvascular networks |
| US9199016B2 (en) | 2009-10-12 | 2015-12-01 | New Health Sciences, Inc. | System for extended storage of red blood cells and methods of use |
| US11284616B2 (en) | 2010-05-05 | 2022-03-29 | Hemanext Inc. | Irradiation of red blood cells and anaerobic storage |
| BR112012008683B8 (pt) | 2009-10-12 | 2022-11-08 | Hemanext Inc | dispositivo de armazenagem de sangue para armazenar sangue depletado de oxigênio e dióxido de carbono, dispositivo de depleção de oxigênio e dióxido de carbono, método para remover oxigênio e dióxido de carbono de hemácias, sistema de armazenagem de sangue, dispositivo de armazenagem de sangue, método para remover oxigênio de hemácias e método para aumentar os níveis de adenosina trifosfato (atp) nas hemácias |
| WO2011046963A1 (en) | 2009-10-12 | 2011-04-21 | New Health Sciences, Inc. | Oxygen depletion devices and methods for removing oxygen from red blood cells |
| WO2012027582A1 (en) | 2010-08-25 | 2012-03-01 | New Health Sciences | Method for enhancing red blood cell quality and survival during storage |
| PT2635114T (pt) | 2010-11-05 | 2020-06-16 | New Health Sciences Inc | Irradiação de glóbulos vermelhos e armazenamento anaeróbico |
| JP2014510731A (ja) | 2011-03-16 | 2014-05-01 | メイヨー ファンデーション フォー メディカル エデュケーション アンド リサーチ | 赤血球製剤および血小板製剤の有効貯蔵を延ばすための方法および材料 |
| US9067004B2 (en) * | 2011-03-28 | 2015-06-30 | New Health Sciences, Inc. | Method and system for removing oxygen and carbon dioxide during red cell blood processing using an inert carrier gas and manifold assembly |
| CN103492041B (zh) * | 2011-03-28 | 2017-02-08 | 新健康科学股份有限公司 | 用于在红细胞血液处理过程中使用惰性载气和歧管组件去除氧和二氧化碳的方法和系统 |
| DE102011052187A1 (de) * | 2011-07-27 | 2013-01-31 | Maquet Vertrieb Und Service Deutschland Gmbh | Anordnung zum Entfernen von Kohlenstoffdioxid aus einem exkorporalen Blutstrom mittels Inertgasen |
| ES2923571T3 (es) | 2011-08-10 | 2022-09-28 | Hemanext Inc | Dispositivo integrado de filtrado de leucocitos, oxígeno y/o CO2 y separación de plasma |
| GB201207543D0 (en) * | 2012-05-01 | 2012-06-13 | Haemair Ltd | Treatment of transfusion blood |
| AU2014223165B2 (en) * | 2013-02-28 | 2017-04-13 | Hemanext Inc. | Gas depletion and gas addition devices for blood treatment |
| KR20180012242A (ko) | 2015-03-10 | 2018-02-05 | 뉴 헬스 사이언시즈 인코포레이티드 | 산소 감소 1회용 키트, 장치 및 이의 사용 방법 |
| IL285359B2 (en) | 2015-04-23 | 2024-01-01 | Hemanext Inc | Anaerobic blood storage containers |
| CA2985799A1 (en) | 2015-05-18 | 2016-11-24 | New Health Sciences, Inc. | Methods for the storage of whole blood, and compositions thereof |
| EP3463466B1 (en) | 2016-05-27 | 2022-02-16 | Hemanext Inc. | Anaerobic blood storage and pathogen inactivation method |
Family Cites Families (17)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US4629544A (en) * | 1984-09-24 | 1986-12-16 | Aquanautics Corporation | Apparatus and method for reversibly removing ligands from carriers |
| IT1218450B (it) * | 1985-09-24 | 1990-04-19 | Teresa Borgione | Perfezionamenti agli ossigenatori per il sangue, a fibre cave |
| US5192320A (en) * | 1987-07-11 | 1993-03-09 | Dainippon Ink And Chemicals Inc. | Artificial lung and method of using it |
| JP2700170B2 (ja) * | 1987-07-11 | 1998-01-19 | 大日本インキ化学工業株式会社 | 膜型人工肺 |
| US5139668A (en) * | 1989-12-27 | 1992-08-18 | Alberta Research Corporation | Hollow fiber bundle element |
| US5194158A (en) * | 1990-06-15 | 1993-03-16 | Matson Stephen L | Radon removal system and process |
| GB2283015B (en) * | 1993-10-22 | 1998-05-13 | Chemitreat Pte Ltd | Membrane reactor for the removal of dissolved oxygen from water |
| US6610832B1 (en) * | 1995-03-23 | 2003-08-26 | Biopure Corporation | Preserving a hemoglobin blood substitute with a transparent overwrap |
| EP1093720B1 (en) * | 1995-03-23 | 2009-11-04 | Biopure Corporation | Stable polymerized hemoglobin blood-substitute |
| US5693230A (en) * | 1996-01-25 | 1997-12-02 | Gas Research Institute | Hollow fiber contactor and process |
| US6162396A (en) * | 1997-04-26 | 2000-12-19 | The Regents Of The University Of California | Blood storage device and method for oxygen removal |
| US6022477A (en) * | 1997-11-14 | 2000-02-08 | New Jersey Institute Of Technology | Method and apparatus for isolation purification of biomolecules |
| US6582496B1 (en) * | 2000-01-28 | 2003-06-24 | Mykrolis Corporation | Hollow fiber membrane contactor |
| TW531428B (en) * | 1999-01-29 | 2003-05-11 | Mykrolis Corp | Fluid-fluid phase contactors and methods of using and making the same |
| DE10327988B4 (de) * | 2002-12-18 | 2009-05-14 | Alpha Plan Gmbh | Filtermodul zur Aufbereitung von Flüssigkeiten |
| KR100721054B1 (ko) * | 2004-11-23 | 2007-05-25 | 주식회사 뉴하트바이오 | 혈액정화 및/또는 혈액산화용 필터모듈, 그를 이용한혈액정화 및 혈액산화 방법 그리고 그를 포함하는혈액정화 장치 |
| JP3772909B1 (ja) * | 2005-04-04 | 2006-05-10 | 東洋紡績株式会社 | 血液浄化器 |
-
2010
- 2010-07-31 CA CA2772320A patent/CA2772320A1/en not_active Abandoned
- 2010-07-31 EP EP10805155.8A patent/EP2459247A4/en not_active Withdrawn
- 2010-07-31 NZ NZ598410A patent/NZ598410A/en not_active IP Right Cessation
- 2010-07-31 WO PCT/US2010/044045 patent/WO2011014855A2/en active Application Filing
- 2010-07-31 US US13/387,528 patent/US20120129149A1/en not_active Abandoned
- 2010-07-31 JP JP2012523121A patent/JP5770183B2/ja not_active Expired - Fee Related
- 2010-07-31 AU AU2010278768A patent/AU2010278768A1/en not_active Abandoned
Also Published As
| Publication number | Publication date |
|---|---|
| NZ598410A (en) | 2014-04-30 |
| WO2011014855A2 (en) | 2011-02-03 |
| AU2010278768A1 (en) | 2012-03-15 |
| JP2013500794A (ja) | 2013-01-10 |
| US20120129149A1 (en) | 2012-05-24 |
| WO2011014855A3 (en) | 2011-06-16 |
| EP2459247A4 (en) | 2013-08-28 |
| CA2772320A1 (en) | 2011-02-03 |
| EP2459247A2 (en) | 2012-06-06 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| JP5770183B2 (ja) | 生体液からの酸素除去 | |
| JP6796664B2 (ja) | 血液処理のためのガス枯渇化およびガス添加デバイス | |
| US8569052B2 (en) | Oxygen depletion devices and methods for removing oxygen from red blood cells | |
| CN107530377B (zh) | 氧减少一次性套件、装置及其使用方法 | |
| AU2016253545B2 (en) | Integrated leukocyte, oxygen and/or co2 depletion, and plasma separation filter device | |
| US10213543B2 (en) | Blood purification column | |
| JP3948736B2 (ja) | 血液透析器 |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| A621 | Written request for application examination |
Free format text: JAPANESE INTERMEDIATE CODE: A621 Effective date: 20130718 |
|
| A977 | Report on retrieval |
Free format text: JAPANESE INTERMEDIATE CODE: A971007 Effective date: 20140327 |
|
| A131 | Notification of reasons for refusal |
Free format text: JAPANESE INTERMEDIATE CODE: A131 Effective date: 20140401 |
|
| A601 | Written request for extension of time |
Free format text: JAPANESE INTERMEDIATE CODE: A601 Effective date: 20140613 |
|
| A602 | Written permission of extension of time |
Free format text: JAPANESE INTERMEDIATE CODE: A602 Effective date: 20140620 |
|
| A601 | Written request for extension of time |
Free format text: JAPANESE INTERMEDIATE CODE: A601 Effective date: 20140729 |
|
| A602 | Written permission of extension of time |
Free format text: JAPANESE INTERMEDIATE CODE: A602 Effective date: 20140805 |
|
| A521 | Request for written amendment filed |
Free format text: JAPANESE INTERMEDIATE CODE: A523 Effective date: 20140828 |
|
| A131 | Notification of reasons for refusal |
Free format text: JAPANESE INTERMEDIATE CODE: A131 Effective date: 20150224 |
|
| A521 | Request for written amendment filed |
Free format text: JAPANESE INTERMEDIATE CODE: A523 Effective date: 20150514 |
|
| TRDD | Decision of grant or rejection written | ||
| A01 | Written decision to grant a patent or to grant a registration (utility model) |
Free format text: JAPANESE INTERMEDIATE CODE: A01 Effective date: 20150602 |
|
| A61 | First payment of annual fees (during grant procedure) |
Free format text: JAPANESE INTERMEDIATE CODE: A61 Effective date: 20150624 |
|
| R150 | Certificate of patent or registration of utility model |
Ref document number: 5770183 Country of ref document: JP Free format text: JAPANESE INTERMEDIATE CODE: R150 |
|
| LAPS | Cancellation because of no payment of annual fees | ||
| S533 | Written request for registration of change of name |
Free format text: JAPANESE INTERMEDIATE CODE: R313533 |
|
| R360 | Written notification for declining of transfer of rights |
Free format text: JAPANESE INTERMEDIATE CODE: R360 |
|
| R370 | Written measure of declining of transfer procedure |
Free format text: JAPANESE INTERMEDIATE CODE: R370 |