JP2010511475A - 人体の植物性機能を最適化する方法、およびそのための装置 - Google Patents
人体の植物性機能を最適化する方法、およびそのための装置 Download PDFInfo
- Publication number
- JP2010511475A JP2010511475A JP2009540194A JP2009540194A JP2010511475A JP 2010511475 A JP2010511475 A JP 2010511475A JP 2009540194 A JP2009540194 A JP 2009540194A JP 2009540194 A JP2009540194 A JP 2009540194A JP 2010511475 A JP2010511475 A JP 2010511475A
- Authority
- JP
- Japan
- Prior art keywords
- antenna
- exposure
- electromagnetic
- human body
- radiation
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Pending
Links
- 238000000034 method Methods 0.000 title claims abstract description 30
- 230000010287 polarization Effects 0.000 claims abstract description 16
- 230000005672 electromagnetic field Effects 0.000 claims abstract description 13
- 230000005670 electromagnetic radiation Effects 0.000 claims abstract description 5
- 230000008859 change Effects 0.000 claims description 4
- 239000000463 material Substances 0.000 claims description 4
- 239000005445 natural material Substances 0.000 claims 1
- 230000005855 radiation Effects 0.000 abstract description 33
- 230000001225 therapeutic effect Effects 0.000 abstract description 19
- 239000003814 drug Substances 0.000 abstract description 8
- 229940079593 drug Drugs 0.000 abstract description 7
- 238000005516 engineering process Methods 0.000 abstract description 3
- 230000037037 animal physiology Effects 0.000 abstract description 2
- 238000011282 treatment Methods 0.000 description 31
- 210000001519 tissue Anatomy 0.000 description 26
- 230000000694 effects Effects 0.000 description 13
- 230000004907 flux Effects 0.000 description 12
- 206010020843 Hyperthermia Diseases 0.000 description 10
- 208000006673 asthma Diseases 0.000 description 10
- 230000036031 hyperthermia Effects 0.000 description 10
- 230000004044 response Effects 0.000 description 10
- 230000003993 interaction Effects 0.000 description 8
- 230000008569 process Effects 0.000 description 7
- 238000002560 therapeutic procedure Methods 0.000 description 7
- 208000037265 diseases, disorders, signs and symptoms Diseases 0.000 description 6
- 206010061218 Inflammation Diseases 0.000 description 5
- 238000010521 absorption reaction Methods 0.000 description 5
- 201000010099 disease Diseases 0.000 description 5
- 230000004054 inflammatory process Effects 0.000 description 5
- 208000030603 inherited susceptibility to asthma Diseases 0.000 description 5
- XLYOFNOQVPJJNP-UHFFFAOYSA-N water Substances O XLYOFNOQVPJJNP-UHFFFAOYSA-N 0.000 description 5
- 229920000742 Cotton Polymers 0.000 description 4
- 241001465754 Metazoa Species 0.000 description 4
- 238000001467 acupuncture Methods 0.000 description 4
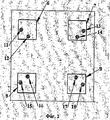
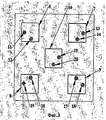
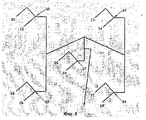
- 238000010586 diagram Methods 0.000 description 4
- 238000000940 electromagnetic therapy Methods 0.000 description 4
- 230000005713 exacerbation Effects 0.000 description 4
- 230000006870 function Effects 0.000 description 4
- 238000010438 heat treatment Methods 0.000 description 4
- 230000003902 lesion Effects 0.000 description 4
- 230000033001 locomotion Effects 0.000 description 4
- 210000000056 organ Anatomy 0.000 description 4
- 238000013459 approach Methods 0.000 description 3
- 238000013461 design Methods 0.000 description 3
- 230000007246 mechanism Effects 0.000 description 3
- 238000011160 research Methods 0.000 description 3
- 230000029058 respiratory gaseous exchange Effects 0.000 description 3
- 239000000758 substrate Substances 0.000 description 3
- 238000012546 transfer Methods 0.000 description 3
- 102000001554 Hemoglobins Human genes 0.000 description 2
- 108010054147 Hemoglobins Proteins 0.000 description 2
- 206010028980 Neoplasm Diseases 0.000 description 2
- 102000057297 Pepsin A Human genes 0.000 description 2
- 108090000284 Pepsin A Proteins 0.000 description 2
- 206010036790 Productive cough Diseases 0.000 description 2
- 230000009286 beneficial effect Effects 0.000 description 2
- 229940124748 beta 2 agonist Drugs 0.000 description 2
- 201000011510 cancer Diseases 0.000 description 2
- 239000000969 carrier Substances 0.000 description 2
- 230000001684 chronic effect Effects 0.000 description 2
- 238000004891 communication Methods 0.000 description 2
- 230000004665 defense response Effects 0.000 description 2
- 238000011161 development Methods 0.000 description 2
- 230000018109 developmental process Effects 0.000 description 2
- 238000002651 drug therapy Methods 0.000 description 2
- 230000005520 electrodynamics Effects 0.000 description 2
- 230000002708 enhancing effect Effects 0.000 description 2
- 230000002496 gastric effect Effects 0.000 description 2
- 238000012544 monitoring process Methods 0.000 description 2
- 238000013546 non-drug therapy Methods 0.000 description 2
- 238000013021 overheating Methods 0.000 description 2
- 230000001717 pathogenic effect Effects 0.000 description 2
- 238000011084 recovery Methods 0.000 description 2
- 210000002345 respiratory system Anatomy 0.000 description 2
- 230000000630 rising effect Effects 0.000 description 2
- 230000035807 sensation Effects 0.000 description 2
- 238000001228 spectrum Methods 0.000 description 2
- 210000002784 stomach Anatomy 0.000 description 2
- 239000000126 substance Substances 0.000 description 2
- 208000024891 symptom Diseases 0.000 description 2
- 238000012360 testing method Methods 0.000 description 2
- 208000024172 Cardiovascular disease Diseases 0.000 description 1
- 206010011224 Cough Diseases 0.000 description 1
- 102000004190 Enzymes Human genes 0.000 description 1
- 108090000790 Enzymes Proteins 0.000 description 1
- 206010020751 Hypersensitivity Diseases 0.000 description 1
- 208000019693 Lung disease Diseases 0.000 description 1
- 206010057190 Respiratory tract infections Diseases 0.000 description 1
- 206010039897 Sedation Diseases 0.000 description 1
- 208000019802 Sexually transmitted disease Diseases 0.000 description 1
- 208000003443 Unconsciousness Diseases 0.000 description 1
- 206010046306 Upper respiratory tract infection Diseases 0.000 description 1
- 206010065813 Vaginal fistula Diseases 0.000 description 1
- 210000001015 abdomen Anatomy 0.000 description 1
- 230000009471 action Effects 0.000 description 1
- 230000002411 adverse Effects 0.000 description 1
- 150000001413 amino acids Chemical class 0.000 description 1
- 238000004458 analytical method Methods 0.000 description 1
- 230000006399 behavior Effects 0.000 description 1
- 230000008901 benefit Effects 0.000 description 1
- 230000004071 biological effect Effects 0.000 description 1
- 230000008827 biological function Effects 0.000 description 1
- 230000005540 biological transmission Effects 0.000 description 1
- 230000015572 biosynthetic process Effects 0.000 description 1
- 230000017531 blood circulation Effects 0.000 description 1
- 210000000621 bronchi Anatomy 0.000 description 1
- 206010006451 bronchitis Diseases 0.000 description 1
- 230000007883 bronchodilation Effects 0.000 description 1
- 244000309464 bull Species 0.000 description 1
- 238000004364 calculation method Methods 0.000 description 1
- 239000002775 capsule Substances 0.000 description 1
- JJWKPURADFRFRB-UHFFFAOYSA-N carbonyl sulfide Chemical compound O=C=S JJWKPURADFRFRB-UHFFFAOYSA-N 0.000 description 1
- 210000000845 cartilage Anatomy 0.000 description 1
- 230000001413 cellular effect Effects 0.000 description 1
- 210000003679 cervix uteri Anatomy 0.000 description 1
- 210000003109 clavicle Anatomy 0.000 description 1
- 238000010835 comparative analysis Methods 0.000 description 1
- 239000012141 concentrate Substances 0.000 description 1
- 230000003750 conditioning effect Effects 0.000 description 1
- 238000007796 conventional method Methods 0.000 description 1
- 230000007123 defense Effects 0.000 description 1
- 230000018044 dehydration Effects 0.000 description 1
- 238000006297 dehydration reaction Methods 0.000 description 1
- 230000002939 deleterious effect Effects 0.000 description 1
- 230000001419 dependent effect Effects 0.000 description 1
- 238000003745 diagnosis Methods 0.000 description 1
- 208000035475 disorder Diseases 0.000 description 1
- 229940088598 enzyme Drugs 0.000 description 1
- 230000005284 excitation Effects 0.000 description 1
- 230000005281 excited state Effects 0.000 description 1
- 239000003172 expectorant agent Substances 0.000 description 1
- 230000003419 expectorant effect Effects 0.000 description 1
- 230000002349 favourable effect Effects 0.000 description 1
- 239000003862 glucocorticoid Substances 0.000 description 1
- 150000002337 glycosamines Chemical class 0.000 description 1
- 230000036541 health Effects 0.000 description 1
- 230000013632 homeostatic process Effects 0.000 description 1
- 230000001976 improved effect Effects 0.000 description 1
- 230000006872 improvement Effects 0.000 description 1
- 238000001727 in vivo Methods 0.000 description 1
- 238000011065 in-situ storage Methods 0.000 description 1
- 230000001939 inductive effect Effects 0.000 description 1
- 208000015181 infectious disease Diseases 0.000 description 1
- 230000002757 inflammatory effect Effects 0.000 description 1
- 230000007774 longterm Effects 0.000 description 1
- 230000004199 lung function Effects 0.000 description 1
- 230000002503 metabolic effect Effects 0.000 description 1
- 230000004048 modification Effects 0.000 description 1
- 238000012986 modification Methods 0.000 description 1
- 230000003287 optical effect Effects 0.000 description 1
- 230000010355 oscillation Effects 0.000 description 1
- 230000001575 pathological effect Effects 0.000 description 1
- 230000000149 penetrating effect Effects 0.000 description 1
- 229940111202 pepsin Drugs 0.000 description 1
- 238000001050 pharmacotherapy Methods 0.000 description 1
- 230000010363 phase shift Effects 0.000 description 1
- 108091008695 photoreceptors Proteins 0.000 description 1
- 230000037081 physical activity Effects 0.000 description 1
- 230000006461 physiological response Effects 0.000 description 1
- 238000004393 prognosis Methods 0.000 description 1
- 230000001681 protective effect Effects 0.000 description 1
- 206010037833 rales Diseases 0.000 description 1
- 230000009467 reduction Effects 0.000 description 1
- 230000008929 regeneration Effects 0.000 description 1
- 238000011069 regeneration method Methods 0.000 description 1
- 230000004202 respiratory function Effects 0.000 description 1
- 208000020029 respiratory tract infectious disease Diseases 0.000 description 1
- 230000036280 sedation Effects 0.000 description 1
- 208000024794 sputum Diseases 0.000 description 1
- 210000003802 sputum Anatomy 0.000 description 1
- 230000003637 steroidlike Effects 0.000 description 1
- 230000009897 systematic effect Effects 0.000 description 1
- 230000009885 systemic effect Effects 0.000 description 1
- 210000000115 thoracic cavity Anatomy 0.000 description 1
- 229940124594 traditional oriental medicine Drugs 0.000 description 1
- 208000016261 weight loss Diseases 0.000 description 1
- 230000004580 weight loss Effects 0.000 description 1
- 210000002268 wool Anatomy 0.000 description 1
Images
Classifications
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61N—ELECTROTHERAPY; MAGNETOTHERAPY; RADIATION THERAPY; ULTRASOUND THERAPY
- A61N5/00—Radiation therapy
- A61N5/02—Radiation therapy using microwaves
-
- H—ELECTRICITY
- H01—ELECTRIC ELEMENTS
- H01Q—ANTENNAS, i.e. RADIO AERIALS
- H01Q21/00—Antenna arrays or systems
- H01Q21/06—Arrays of individually energised antenna units similarly polarised and spaced apart
- H01Q21/061—Two dimensional planar arrays
- H01Q21/065—Patch antenna array
-
- H—ELECTRICITY
- H01—ELECTRIC ELEMENTS
- H01Q—ANTENNAS, i.e. RADIO AERIALS
- H01Q21/00—Antenna arrays or systems
- H01Q21/24—Combinations of antenna units polarised in different directions for transmitting or receiving circularly and elliptically polarised waves or waves linearly polarised in any direction
Landscapes
- Health & Medical Sciences (AREA)
- Engineering & Computer Science (AREA)
- Biomedical Technology (AREA)
- General Health & Medical Sciences (AREA)
- Veterinary Medicine (AREA)
- Radiology & Medical Imaging (AREA)
- Life Sciences & Earth Sciences (AREA)
- Animal Behavior & Ethology (AREA)
- Pathology (AREA)
- Public Health (AREA)
- Nuclear Medicine, Radiotherapy & Molecular Imaging (AREA)
- Radiation-Therapy Devices (AREA)
- Electrotherapy Devices (AREA)
- Farming Of Fish And Shellfish (AREA)
- Magnetic Treatment Devices (AREA)
- Waveguide Aerials (AREA)
- Surgical Instruments (AREA)
- Hydroponics (AREA)
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| RU2006144755/14A RU2339413C2 (ru) | 2006-12-07 | 2006-12-07 | Способ оптимизации вегетативных функций организма человека и устройство для его осуществления |
| PCT/RU2007/000569 WO2008069692A1 (fr) | 2006-12-07 | 2007-10-16 | Procédé d'optimisation de l'état fonctionnel de systèmes végétatifs de l'organisme et dispositif de sa mise en oeuvre |
Publications (2)
| Publication Number | Publication Date |
|---|---|
| JP2010511475A true JP2010511475A (ja) | 2010-04-15 |
| JP2010511475A5 JP2010511475A5 (OSRAM) | 2013-02-14 |
Family
ID=40193384
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| JP2009540194A Pending JP2010511475A (ja) | 2006-12-07 | 2007-10-16 | 人体の植物性機能を最適化する方法、およびそのための装置 |
Country Status (14)
| Country | Link |
|---|---|
| US (1) | US20090254157A1 (OSRAM) |
| EP (1) | EP2095842B1 (OSRAM) |
| JP (1) | JP2010511475A (OSRAM) |
| CN (1) | CN101626807B (OSRAM) |
| AT (1) | ATE545448T1 (OSRAM) |
| AU (1) | AU2007328552B2 (OSRAM) |
| BR (1) | BRPI0720130A2 (OSRAM) |
| CA (1) | CA2671312A1 (OSRAM) |
| DK (1) | DK2095842T3 (OSRAM) |
| ES (1) | ES2382630T3 (OSRAM) |
| NZ (1) | NZ577862A (OSRAM) |
| PL (1) | PL2095842T3 (OSRAM) |
| PT (1) | PT2095842E (OSRAM) |
| RU (1) | RU2339413C2 (OSRAM) |
Cited By (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JP2022553317A (ja) * | 2019-10-21 | 2022-12-22 | セラバイオニック インコーポレイティド | 対象の治療のための電気システム |
Families Citing this family (4)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US20080172105A1 (en) * | 2007-01-17 | 2008-07-17 | Ws Far Ir Medical Technology Co., Ltd. | Method for preventing and/or ameliorating inflammation |
| CN108273194A (zh) * | 2017-01-05 | 2018-07-13 | 山东量子生物科技有限公司 | 量子芯灸片 |
| CN114824830A (zh) * | 2022-06-29 | 2022-07-29 | 盛纬伦(深圳)通信技术有限公司 | 一种圆极化旁瓣抑制的贴片阵列天线 |
| CN118557899B (zh) * | 2024-08-01 | 2024-10-22 | 南昌大学附属康复医院(南昌大学第四附属医院) | 分布式微波神经调控装置及相关产品 |
Citations (5)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JPS6068868A (ja) * | 1983-06-16 | 1985-04-19 | リチヤ−ド ヒユ− カメロン ベンタル | 生体の代謝特性感化用電気装置 |
| JPH03143453A (ja) * | 1989-10-31 | 1991-06-19 | Shimadzu Corp | ハイパーサーミア装置 |
| JPH0857065A (ja) * | 1994-08-25 | 1996-03-05 | Akinobu Tomono | 変調式マイクロ波治療器 |
| US5507791A (en) * | 1993-08-31 | 1996-04-16 | Sit'ko; Sergei P. | Microwave resonance therapy |
| JP2006180444A (ja) * | 2004-12-22 | 2006-07-06 | Tatung Co | 円形分極アレイアンテナ |
Family Cites Families (10)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CA1266325A (en) * | 1985-07-23 | 1990-02-27 | Fumihiro Ito | Microwave antenna |
| GB2189080B (en) * | 1986-04-02 | 1989-11-29 | Thorn Emi Electronics Ltd | Microstrip antenna |
| US4932420A (en) * | 1988-10-07 | 1990-06-12 | Clini-Therm Corporation | Non-invasive quarter wavelength microwave applicator for hyperthermia treatment |
| SU1831343A3 (en) * | 1991-10-17 | 1993-07-30 | Stanislav N Darovskikh | Device for stimulation of functional state of biological object |
| CN1076605A (zh) * | 1993-01-07 | 1993-09-29 | 许陆文 | 高效微波治疗仪 |
| US5661494A (en) * | 1995-03-24 | 1997-08-26 | The United States Of America As Represented By The Administrator Of The National Aeronautics And Space Administration | High performance circularly polarized microstrip antenna |
| US5769879A (en) * | 1995-06-07 | 1998-06-23 | Medical Contouring Corporation | Microwave applicator and method of operation |
| US6097985A (en) * | 1999-02-09 | 2000-08-01 | Kai Technologies, Inc. | Microwave systems for medical hyperthermia, thermotherapy and diagnosis |
| RU34087U1 (ru) * | 2003-07-30 | 2003-11-27 | Черняков Геннадий Михайлович | Устройство для активизации регуляторных процессов биологического объекта |
| US7769468B2 (en) * | 2006-03-03 | 2010-08-03 | Bsd Medical Corporation | Transparent electromagnetic applicator and hyperthermia treatment method |
-
2006
- 2006-12-07 RU RU2006144755/14A patent/RU2339413C2/ru not_active IP Right Cessation
-
2007
- 2007-10-16 CA CA002671312A patent/CA2671312A1/en not_active Abandoned
- 2007-10-16 AT AT07861032T patent/ATE545448T1/de active
- 2007-10-16 CN CN2007800452613A patent/CN101626807B/zh not_active Expired - Fee Related
- 2007-10-16 BR BRPI0720130-3A2A patent/BRPI0720130A2/pt not_active IP Right Cessation
- 2007-10-16 NZ NZ577862A patent/NZ577862A/en not_active IP Right Cessation
- 2007-10-16 JP JP2009540194A patent/JP2010511475A/ja active Pending
- 2007-10-16 AU AU2007328552A patent/AU2007328552B2/en not_active Ceased
- 2007-10-16 DK DK07861032.6T patent/DK2095842T3/da active
- 2007-10-16 PL PL07861032T patent/PL2095842T3/pl unknown
- 2007-10-16 PT PT07861032T patent/PT2095842E/pt unknown
- 2007-10-16 ES ES07861032T patent/ES2382630T3/es active Active
- 2007-10-16 EP EP07861032A patent/EP2095842B1/en not_active Not-in-force
-
2009
- 2009-06-08 US US12/480,019 patent/US20090254157A1/en not_active Abandoned
Patent Citations (5)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JPS6068868A (ja) * | 1983-06-16 | 1985-04-19 | リチヤ−ド ヒユ− カメロン ベンタル | 生体の代謝特性感化用電気装置 |
| JPH03143453A (ja) * | 1989-10-31 | 1991-06-19 | Shimadzu Corp | ハイパーサーミア装置 |
| US5507791A (en) * | 1993-08-31 | 1996-04-16 | Sit'ko; Sergei P. | Microwave resonance therapy |
| JPH0857065A (ja) * | 1994-08-25 | 1996-03-05 | Akinobu Tomono | 変調式マイクロ波治療器 |
| JP2006180444A (ja) * | 2004-12-22 | 2006-07-06 | Tatung Co | 円形分極アレイアンテナ |
Cited By (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JP2022553317A (ja) * | 2019-10-21 | 2022-12-22 | セラバイオニック インコーポレイティド | 対象の治療のための電気システム |
Also Published As
| Publication number | Publication date |
|---|---|
| ATE545448T1 (de) | 2012-03-15 |
| PL2095842T3 (pl) | 2012-08-31 |
| BRPI0720130A2 (pt) | 2014-11-18 |
| NZ577862A (en) | 2011-07-29 |
| AU2007328552A1 (en) | 2008-06-12 |
| DK2095842T3 (da) | 2012-06-11 |
| CN101626807B (zh) | 2012-09-05 |
| AU2007328552B2 (en) | 2011-03-31 |
| EP2095842A4 (en) | 2010-05-26 |
| US20090254157A1 (en) | 2009-10-08 |
| EP2095842A8 (en) | 2009-11-11 |
| PT2095842E (pt) | 2012-07-16 |
| CA2671312A1 (en) | 2008-06-12 |
| EP2095842A1 (en) | 2009-09-02 |
| CN101626807A (zh) | 2010-01-13 |
| RU2339413C2 (ru) | 2008-11-27 |
| EP2095842B1 (en) | 2012-02-15 |
| ES2382630T3 (es) | 2012-06-12 |
| RU2006144755A (ru) | 2008-06-20 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| Chicheł et al. | Hyperthermia–description of a method and a review of clinical applications | |
| Szasz et al. | Oncothermia: principles and practices | |
| JP4309348B2 (ja) | 深部腫瘍向けの単極フェイズドアレイ温熱療法アプリケータ | |
| CN102176948B (zh) | 具有目标反馈信号调制的射频热疗装置 | |
| RU2509579C2 (ru) | Устройство для гипертермии | |
| CN104640601B (zh) | 用于个性化治疗和诊断的rf热疗装置 | |
| DE60116495T2 (de) | System zur wärmebehandlung der prostata | |
| JP2003530976A (ja) | マイクロ波を用いた乳房病巣を治療する方法および装置 | |
| US9669231B1 (en) | Apparatus and method for hyperthermic treatments | |
| JP2010511475A (ja) | 人体の植物性機能を最適化する方法、およびそのための装置 | |
| US20080140063A1 (en) | Non-invasive method and system for using radio frequency induced hyperthermia to treat medical diseases | |
| Habash et al. | Principles, applications, risks and benefits of therapeutic hyperthermia | |
| Sardari et al. | Cancer treatment with hyperthermia | |
| US20090132015A1 (en) | Method and System for Using Directional Antennas in Medical Treatments | |
| Cabuy | Hyperthermia in cancer treatment | |
| Dunlop et al. | Has hyperthermia a place in cancer treatment? | |
| Singh | Microwave applicators for hyperthermia treatment of cancer: An overview | |
| Panda | Application of hyperthermia for cancer treatment: various techniques and recent advancement | |
| Cepeda et al. | Coaxial double slot antenna design for interstitial hyperthermia in muscle using a finite element computer modeling | |
| WO2008069692A1 (fr) | Procédé d'optimisation de l'état fonctionnel de systèmes végétatifs de l'organisme et dispositif de sa mise en oeuvre | |
| Szasz et al. | Hyperthermia results and challenges | |
| Androulakis et al. | Preclinical prototype validation and characterization of a thermobrachytherapy system for interstitial hyperthermia and high-dose-rate brachytherapy | |
| Losito et al. | E-field distribution improvement by new hyperthermia applicators | |
| Guerrero et al. | Principles of Hyperthermia Physics and Clinical Application | |
| Hand | Hyperthermia: Challenging applications of physics in cancer therapy |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| A977 | Report on retrieval |
Free format text: JAPANESE INTERMEDIATE CODE: A971007 Effective date: 20111228 |
|
| A131 | Notification of reasons for refusal |
Free format text: JAPANESE INTERMEDIATE CODE: A131 Effective date: 20120105 |
|
| A521 | Request for written amendment filed |
Free format text: JAPANESE INTERMEDIATE CODE: A523 Effective date: 20120323 |
|
| A131 | Notification of reasons for refusal |
Free format text: JAPANESE INTERMEDIATE CODE: A131 Effective date: 20120921 |
|
| A521 | Request for written amendment filed |
Free format text: JAPANESE INTERMEDIATE CODE: A523 Effective date: 20121214 |
|
| A524 | Written submission of copy of amendment under article 19 pct |
Free format text: JAPANESE INTERMEDIATE CODE: A524 Effective date: 20121214 |
|
| A02 | Decision of refusal |
Free format text: JAPANESE INTERMEDIATE CODE: A02 Effective date: 20130625 |