JP2010059155A - Agent for improving exercise function - Google Patents
Agent for improving exercise function Download PDFInfo
- Publication number
- JP2010059155A JP2010059155A JP2009183261A JP2009183261A JP2010059155A JP 2010059155 A JP2010059155 A JP 2010059155A JP 2009183261 A JP2009183261 A JP 2009183261A JP 2009183261 A JP2009183261 A JP 2009183261A JP 2010059155 A JP2010059155 A JP 2010059155A
- Authority
- JP
- Japan
- Prior art keywords
- fat globule
- globule membrane
- membrane component
- agent
- improving
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Granted
Links
- 239000012528 membrane Substances 0.000 claims abstract description 82
- 229920002527 Glycogen Polymers 0.000 claims abstract description 20
- 229940096919 glycogen Drugs 0.000 claims abstract description 20
- 239000004480 active ingredient Substances 0.000 claims abstract description 18
- 210000003205 muscle Anatomy 0.000 claims description 47
- 230000007659 motor function Effects 0.000 claims description 33
- 230000006870 function Effects 0.000 claims description 23
- 230000002929 anti-fatigue Effects 0.000 claims description 13
- 238000009825 accumulation Methods 0.000 claims description 10
- 230000001737 promoting effect Effects 0.000 abstract description 8
- 230000003387 muscular Effects 0.000 abstract description 5
- 238000005086 pumping Methods 0.000 abstract 2
- 235000019197 fats Nutrition 0.000 description 64
- 235000013305 food Nutrition 0.000 description 30
- 235000013336 milk Nutrition 0.000 description 30
- 239000008267 milk Substances 0.000 description 30
- 210000004080 milk Anatomy 0.000 description 30
- 108010071421 milk fat globule Proteins 0.000 description 17
- 230000009182 swimming Effects 0.000 description 16
- 150000002632 lipids Chemical class 0.000 description 15
- 230000009471 action Effects 0.000 description 14
- 230000006872 improvement Effects 0.000 description 13
- 238000000034 method Methods 0.000 description 13
- 239000000203 mixture Substances 0.000 description 13
- 108090000623 proteins and genes Proteins 0.000 description 13
- 230000000694 effects Effects 0.000 description 12
- 150000003904 phospholipids Chemical class 0.000 description 12
- 239000003814 drug Substances 0.000 description 11
- 230000014509 gene expression Effects 0.000 description 11
- 235000005911 diet Nutrition 0.000 description 10
- 235000018102 proteins Nutrition 0.000 description 10
- 102000004169 proteins and genes Human genes 0.000 description 10
- 239000011734 sodium Substances 0.000 description 10
- 230000037213 diet Effects 0.000 description 9
- 230000037406 food intake Effects 0.000 description 9
- 235000012631 food intake Nutrition 0.000 description 9
- VLKZOEOYAKHREP-UHFFFAOYSA-N n-Hexane Chemical compound CCCCCC VLKZOEOYAKHREP-UHFFFAOYSA-N 0.000 description 9
- 210000002027 skeletal muscle Anatomy 0.000 description 9
- 230000002269 spontaneous effect Effects 0.000 description 9
- 239000003795 chemical substances by application Substances 0.000 description 8
- 235000008504 concentrate Nutrition 0.000 description 8
- 239000012141 concentrate Substances 0.000 description 8
- 238000012360 testing method Methods 0.000 description 8
- 238000009395 breeding Methods 0.000 description 7
- 230000001488 breeding effect Effects 0.000 description 7
- 229940079593 drug Drugs 0.000 description 7
- CIWBSHSKHKDKBQ-JLAZNSOCSA-N Ascorbic acid Chemical compound OC[C@H](O)[C@H]1OC(=O)C(O)=C1O CIWBSHSKHKDKBQ-JLAZNSOCSA-N 0.000 description 6
- KFZMGEQAYNKOFK-UHFFFAOYSA-N Isopropanol Chemical compound CC(C)O KFZMGEQAYNKOFK-UHFFFAOYSA-N 0.000 description 6
- 230000037396 body weight Effects 0.000 description 6
- 235000015155 buttermilk Nutrition 0.000 description 6
- 239000011575 calcium Substances 0.000 description 6
- KRKNYBCHXYNGOX-UHFFFAOYSA-N citric acid Chemical compound OC(=O)CC(O)(C(O)=O)CC(O)=O KRKNYBCHXYNGOX-UHFFFAOYSA-N 0.000 description 6
- 238000009472 formulation Methods 0.000 description 6
- 239000007788 liquid Substances 0.000 description 6
- 238000002360 preparation method Methods 0.000 description 6
- 239000000047 product Substances 0.000 description 6
- 235000000891 standard diet Nutrition 0.000 description 6
- XLYOFNOQVPJJNP-UHFFFAOYSA-N water Substances O XLYOFNOQVPJJNP-UHFFFAOYSA-N 0.000 description 6
- 241000699670 Mus sp. Species 0.000 description 5
- 235000014121 butter Nutrition 0.000 description 5
- 235000016709 nutrition Nutrition 0.000 description 5
- YDNKGFDKKRUKPY-JHOUSYSJSA-N C16 ceramide Natural products CCCCCCCCCCCCCCCC(=O)N[C@@H](CO)[C@H](O)C=CCCCCCCCCCCCCC YDNKGFDKKRUKPY-JHOUSYSJSA-N 0.000 description 4
- CRJGESKKUOMBCT-VQTJNVASSA-N N-acetylsphinganine Chemical compound CCCCCCCCCCCCCCC[C@@H](O)[C@H](CO)NC(C)=O CRJGESKKUOMBCT-VQTJNVASSA-N 0.000 description 4
- 229940106189 ceramide Drugs 0.000 description 4
- ZVEQCJWYRWKARO-UHFFFAOYSA-N ceramide Natural products CCCCCCCCCCCCCCC(O)C(=O)NC(CO)C(O)C=CCCC=C(C)CCCCCCCCC ZVEQCJWYRWKARO-UHFFFAOYSA-N 0.000 description 4
- 235000013365 dairy product Nutrition 0.000 description 4
- 238000002224 dissection Methods 0.000 description 4
- 239000002552 dosage form Substances 0.000 description 4
- VVGIYYKRAMHVLU-UHFFFAOYSA-N newbouldiamide Natural products CCCCCCCCCCCCCCCCCCCC(O)C(O)C(O)C(CO)NC(=O)CCCCCCCCCCCCCCCCC VVGIYYKRAMHVLU-UHFFFAOYSA-N 0.000 description 4
- 239000003921 oil Substances 0.000 description 4
- 235000019198 oils Nutrition 0.000 description 4
- CSCPPACGZOOCGX-UHFFFAOYSA-N Acetone Chemical compound CC(C)=O CSCPPACGZOOCGX-UHFFFAOYSA-N 0.000 description 3
- 241001465754 Metazoa Species 0.000 description 3
- OKKJLVBELUTLKV-UHFFFAOYSA-N Methanol Chemical compound OC OKKJLVBELUTLKV-UHFFFAOYSA-N 0.000 description 3
- 238000004458 analytical method Methods 0.000 description 3
- 235000013361 beverage Nutrition 0.000 description 3
- 239000002775 capsule Substances 0.000 description 3
- 238000007796 conventional method Methods 0.000 description 3
- 230000007423 decrease Effects 0.000 description 3
- 239000000796 flavoring agent Substances 0.000 description 3
- 150000002270 gangliosides Chemical class 0.000 description 3
- 150000002305 glucosylceramides Chemical class 0.000 description 3
- 239000008187 granular material Substances 0.000 description 3
- 235000015110 jellies Nutrition 0.000 description 3
- 239000008274 jelly Substances 0.000 description 3
- 235000020124 milk-based beverage Nutrition 0.000 description 3
- 230000035764 nutrition Effects 0.000 description 3
- 239000000843 powder Substances 0.000 description 3
- -1 softeners Substances 0.000 description 3
- 239000007787 solid Substances 0.000 description 3
- 239000000243 solution Substances 0.000 description 3
- 239000003381 stabilizer Substances 0.000 description 3
- 230000000638 stimulation Effects 0.000 description 3
- 239000013589 supplement Substances 0.000 description 3
- 235000020357 syrup Nutrition 0.000 description 3
- 239000006188 syrup Substances 0.000 description 3
- 239000003826 tablet Substances 0.000 description 3
- GVJHHUAWPYXKBD-UHFFFAOYSA-N (±)-α-Tocopherol Chemical compound OC1=C(C)C(C)=C2OC(CCCC(C)CCCC(C)CCCC(C)C)(C)CCC2=C1C GVJHHUAWPYXKBD-UHFFFAOYSA-N 0.000 description 2
- NHBKXEKEPDILRR-UHFFFAOYSA-N 2,3-bis(butanoylsulfanyl)propyl butanoate Chemical compound CCCC(=O)OCC(SC(=O)CCC)CSC(=O)CCC NHBKXEKEPDILRR-UHFFFAOYSA-N 0.000 description 2
- IJGRMHOSHXDMSA-UHFFFAOYSA-N Atomic nitrogen Chemical compound N#N IJGRMHOSHXDMSA-UHFFFAOYSA-N 0.000 description 2
- HEDRZPFGACZZDS-UHFFFAOYSA-N Chloroform Chemical compound ClC(Cl)Cl HEDRZPFGACZZDS-UHFFFAOYSA-N 0.000 description 2
- 240000000560 Citrus x paradisi Species 0.000 description 2
- ZZZCUOFIHGPKAK-UHFFFAOYSA-N D-erythro-ascorbic acid Natural products OCC1OC(=O)C(O)=C1O ZZZCUOFIHGPKAK-UHFFFAOYSA-N 0.000 description 2
- RTZKZFJDLAIYFH-UHFFFAOYSA-N Diethyl ether Chemical compound CCOCC RTZKZFJDLAIYFH-UHFFFAOYSA-N 0.000 description 2
- 241000699666 Mus <mouse, genus> Species 0.000 description 2
- DFPAKSUCGFBDDF-UHFFFAOYSA-N Nicotinamide Chemical compound NC(=O)C1=CC=CN=C1 DFPAKSUCGFBDDF-UHFFFAOYSA-N 0.000 description 2
- NBIIXXVUZAFLBC-UHFFFAOYSA-N Phosphoric acid Chemical compound OP(O)(O)=O NBIIXXVUZAFLBC-UHFFFAOYSA-N 0.000 description 2
- 229930006000 Sucrose Natural products 0.000 description 2
- CZMRCDWAGMRECN-UGDNZRGBSA-N Sucrose Chemical compound O[C@H]1[C@H](O)[C@@H](CO)O[C@@]1(CO)O[C@@H]1[C@H](O)[C@@H](O)[C@H](O)[C@@H](CO)O1 CZMRCDWAGMRECN-UGDNZRGBSA-N 0.000 description 2
- 229930003268 Vitamin C Natural products 0.000 description 2
- 108010046377 Whey Proteins Proteins 0.000 description 2
- 102000007544 Whey Proteins Human genes 0.000 description 2
- 229940024606 amino acid Drugs 0.000 description 2
- 150000001413 amino acids Chemical class 0.000 description 2
- 238000001035 drying Methods 0.000 description 2
- 235000013399 edible fruits Nutrition 0.000 description 2
- 230000037080 exercise endurance Effects 0.000 description 2
- 238000004186 food analysis Methods 0.000 description 2
- 235000013355 food flavoring agent Nutrition 0.000 description 2
- 239000003205 fragrance Substances 0.000 description 2
- 150000002327 glycerophospholipids Chemical class 0.000 description 2
- 150000002339 glycosphingolipids Chemical class 0.000 description 2
- 238000004895 liquid chromatography mass spectrometry Methods 0.000 description 2
- HQKMJHAJHXVSDF-UHFFFAOYSA-L magnesium stearate Chemical compound [Mg+2].CCCCCCCCCCCCCCCCCC([O-])=O.CCCCCCCCCCCCCCCCCC([O-])=O HQKMJHAJHXVSDF-UHFFFAOYSA-L 0.000 description 2
- 235000012054 meals Nutrition 0.000 description 2
- 108020004999 messenger RNA Proteins 0.000 description 2
- BDAGIHXWWSANSR-UHFFFAOYSA-N methanoic acid Natural products OC=O BDAGIHXWWSANSR-UHFFFAOYSA-N 0.000 description 2
- 230000004118 muscle contraction Effects 0.000 description 2
- 230000035790 physiological processes and functions Effects 0.000 description 2
- BASFCYQUMIYNBI-UHFFFAOYSA-N platinum Chemical compound [Pt] BASFCYQUMIYNBI-UHFFFAOYSA-N 0.000 description 2
- 238000001556 precipitation Methods 0.000 description 2
- 239000003755 preservative agent Substances 0.000 description 2
- 239000008213 purified water Substances 0.000 description 2
- 239000002994 raw material Substances 0.000 description 2
- 210000001908 sarcoplasmic reticulum Anatomy 0.000 description 2
- 238000000926 separation method Methods 0.000 description 2
- 210000002966 serum Anatomy 0.000 description 2
- 235000020183 skimmed milk Nutrition 0.000 description 2
- 235000014214 soft drink Nutrition 0.000 description 2
- 239000002904 solvent Substances 0.000 description 2
- 239000005720 sucrose Substances 0.000 description 2
- XOAAWQZATWQOTB-UHFFFAOYSA-N taurine Chemical compound NCCS(O)(=O)=O XOAAWQZATWQOTB-UHFFFAOYSA-N 0.000 description 2
- 235000013616 tea Nutrition 0.000 description 2
- 229930003231 vitamin Natural products 0.000 description 2
- 235000013343 vitamin Nutrition 0.000 description 2
- 239000011782 vitamin Substances 0.000 description 2
- 229940088594 vitamin Drugs 0.000 description 2
- 235000019154 vitamin C Nutrition 0.000 description 2
- 239000011718 vitamin C Substances 0.000 description 2
- 235000021119 whey protein Nutrition 0.000 description 2
- WMBWREPUVVBILR-WIYYLYMNSA-N (-)-Epigallocatechin-3-o-gallate Chemical compound O([C@@H]1CC2=C(O)C=C(C=C2O[C@@H]1C=1C=C(O)C(O)=C(O)C=1)O)C(=O)C1=CC(O)=C(O)C(O)=C1 WMBWREPUVVBILR-WIYYLYMNSA-N 0.000 description 1
- JLPULHDHAOZNQI-ZTIMHPMXSA-N 1-hexadecanoyl-2-(9Z,12Z-octadecadienoyl)-sn-glycero-3-phosphocholine Chemical compound CCCCCCCCCCCCCCCC(=O)OC[C@H](COP([O-])(=O)OCC[N+](C)(C)C)OC(=O)CCCCCCC\C=C/C\C=C/CCCCC JLPULHDHAOZNQI-ZTIMHPMXSA-N 0.000 description 1
- MIDXCONKKJTLDX-UHFFFAOYSA-N 3,5-dimethylcyclopentane-1,2-dione Chemical compound CC1CC(C)C(=O)C1=O MIDXCONKKJTLDX-UHFFFAOYSA-N 0.000 description 1
- OSWFIVFLDKOXQC-UHFFFAOYSA-N 4-(3-methoxyphenyl)aniline Chemical compound COC1=CC=CC(C=2C=CC(N)=CC=2)=C1 OSWFIVFLDKOXQC-UHFFFAOYSA-N 0.000 description 1
- 108091006112 ATPases Proteins 0.000 description 1
- 102000057290 Adenosine Triphosphatases Human genes 0.000 description 1
- 102000011690 Adiponectin Human genes 0.000 description 1
- 108010076365 Adiponectin Proteins 0.000 description 1
- 206010001497 Agitation Diseases 0.000 description 1
- 108010011485 Aspartame Proteins 0.000 description 1
- AUNGANRZJHBGPY-UHFFFAOYSA-N D-Lyxoflavin Natural products OCC(O)C(O)C(O)CN1C=2C=C(C)C(C)=CC=2N=C2C1=NC(=O)NC2=O AUNGANRZJHBGPY-UHFFFAOYSA-N 0.000 description 1
- 229920001353 Dextrin Polymers 0.000 description 1
- 239000004375 Dextrin Substances 0.000 description 1
- 108700039887 Essential Genes Proteins 0.000 description 1
- 239000004606 Fillers/Extenders Substances 0.000 description 1
- PEDCQBHIVMGVHV-UHFFFAOYSA-N Glycerol Natural products OCC(O)CO PEDCQBHIVMGVHV-UHFFFAOYSA-N 0.000 description 1
- JZNWSCPGTDBMEW-UHFFFAOYSA-N Glycerophosphorylethanolamin Natural products NCCOP(O)(=O)OCC(O)CO JZNWSCPGTDBMEW-UHFFFAOYSA-N 0.000 description 1
- 108090000288 Glycoproteins Proteins 0.000 description 1
- 102000003886 Glycoproteins Human genes 0.000 description 1
- 238000007696 Kjeldahl method Methods 0.000 description 1
- AGPKZVBTJJNPAG-WHFBIAKZSA-N L-isoleucine Chemical compound CC[C@H](C)[C@H](N)C(O)=O AGPKZVBTJJNPAG-WHFBIAKZSA-N 0.000 description 1
- ROHFNLRQFUQHCH-YFKPBYRVSA-N L-leucine Chemical compound CC(C)C[C@H](N)C(O)=O ROHFNLRQFUQHCH-YFKPBYRVSA-N 0.000 description 1
- KZSNJWFQEVHDMF-BYPYZUCNSA-N L-valine Chemical compound CC(C)[C@H](N)C(O)=O KZSNJWFQEVHDMF-BYPYZUCNSA-N 0.000 description 1
- GUBGYTABKSRVRQ-QKKXKWKRSA-N Lactose Natural products OC[C@H]1O[C@@H](O[C@H]2[C@H](O)[C@@H](O)C(O)O[C@@H]2CO)[C@H](O)[C@@H](O)[C@H]1O GUBGYTABKSRVRQ-QKKXKWKRSA-N 0.000 description 1
- ROHFNLRQFUQHCH-UHFFFAOYSA-N Leucine Natural products CC(C)CC(N)C(O)=O ROHFNLRQFUQHCH-UHFFFAOYSA-N 0.000 description 1
- 229920000161 Locust bean gum Polymers 0.000 description 1
- 239000004909 Moisturizer Substances 0.000 description 1
- 102000015728 Mucins Human genes 0.000 description 1
- 108010063954 Mucins Proteins 0.000 description 1
- SQVRNKJHWKZAKO-PFQGKNLYSA-N N-acetyl-beta-neuraminic acid Chemical compound CC(=O)N[C@@H]1[C@@H](O)C[C@@](O)(C(O)=O)O[C@H]1[C@H](O)[C@H](O)CO SQVRNKJHWKZAKO-PFQGKNLYSA-N 0.000 description 1
- 208000008589 Obesity Diseases 0.000 description 1
- 235000004347 Perilla Nutrition 0.000 description 1
- 244000124853 Perilla frutescens Species 0.000 description 1
- AUNGANRZJHBGPY-SCRDCRAPSA-N Riboflavin Chemical compound OC[C@@H](O)[C@@H](O)[C@@H](O)CN1C=2C=C(C)C(C)=CC=2N=C2C1=NC(=O)NC2=O AUNGANRZJHBGPY-SCRDCRAPSA-N 0.000 description 1
- 102000012010 Sialomucins Human genes 0.000 description 1
- 108010061228 Sialomucins Proteins 0.000 description 1
- 108010073771 Soybean Proteins Proteins 0.000 description 1
- 238000000692 Student's t-test Methods 0.000 description 1
- 244000269722 Thea sinensis Species 0.000 description 1
- 244000000188 Vaccinium ovalifolium Species 0.000 description 1
- KZSNJWFQEVHDMF-UHFFFAOYSA-N Valine Natural products CC(C)C(N)C(O)=O KZSNJWFQEVHDMF-UHFFFAOYSA-N 0.000 description 1
- 229930003471 Vitamin B2 Natural products 0.000 description 1
- QYSXJUFSXHHAJI-XFEUOLMDSA-N Vitamin D3 Natural products C1(/[C@@H]2CC[C@@H]([C@]2(CCC1)C)[C@H](C)CCCC(C)C)=C/C=C1\C[C@@H](O)CCC1=C QYSXJUFSXHHAJI-XFEUOLMDSA-N 0.000 description 1
- 229930003427 Vitamin E Natural products 0.000 description 1
- 238000010521 absorption reaction Methods 0.000 description 1
- 230000002378 acidificating effect Effects 0.000 description 1
- 230000036982 action potential Effects 0.000 description 1
- 230000002411 adverse Effects 0.000 description 1
- 238000005273 aeration Methods 0.000 description 1
- 229910000147 aluminium phosphate Inorganic materials 0.000 description 1
- VZTDIZULWFCMLS-UHFFFAOYSA-N ammonium formate Chemical compound [NH4+].[O-]C=O VZTDIZULWFCMLS-UHFFFAOYSA-N 0.000 description 1
- BFNBIHQBYMNNAN-UHFFFAOYSA-N ammonium sulfate Chemical compound N.N.OS(O)(=O)=O BFNBIHQBYMNNAN-UHFFFAOYSA-N 0.000 description 1
- 229910052921 ammonium sulfate Inorganic materials 0.000 description 1
- 235000011130 ammonium sulphate Nutrition 0.000 description 1
- 238000000540 analysis of variance Methods 0.000 description 1
- 239000003963 antioxidant agent Substances 0.000 description 1
- 235000006708 antioxidants Nutrition 0.000 description 1
- 235000010323 ascorbic acid Nutrition 0.000 description 1
- 239000011668 ascorbic acid Substances 0.000 description 1
- 229960005070 ascorbic acid Drugs 0.000 description 1
- IAOZJIPTCAWIRG-QWRGUYRKSA-N aspartame Chemical compound OC(=O)C[C@H](N)C(=O)N[C@H](C(=O)OC)CC1=CC=CC=C1 IAOZJIPTCAWIRG-QWRGUYRKSA-N 0.000 description 1
- 235000010357 aspartame Nutrition 0.000 description 1
- 239000000605 aspartame Substances 0.000 description 1
- 229960003438 aspartame Drugs 0.000 description 1
- 230000000386 athletic effect Effects 0.000 description 1
- 239000011230 binding agent Substances 0.000 description 1
- 235000015895 biscuits Nutrition 0.000 description 1
- 210000004369 blood Anatomy 0.000 description 1
- 239000008280 blood Substances 0.000 description 1
- 235000008429 bread Nutrition 0.000 description 1
- 239000000872 buffer Substances 0.000 description 1
- 239000006172 buffering agent Substances 0.000 description 1
- 230000003185 calcium uptake Effects 0.000 description 1
- 235000013736 caramel Nutrition 0.000 description 1
- 235000014171 carbonated beverage Nutrition 0.000 description 1
- 239000000679 carrageenan Substances 0.000 description 1
- 235000010418 carrageenan Nutrition 0.000 description 1
- 229920001525 carrageenan Polymers 0.000 description 1
- 229940113118 carrageenan Drugs 0.000 description 1
- 239000000969 carrier Substances 0.000 description 1
- 239000005018 casein Substances 0.000 description 1
- BECPQYXYKAMYBN-UHFFFAOYSA-N casein, tech. Chemical compound NCCCCC(C(O)=O)N=C(O)C(CC(O)=O)N=C(O)C(CCC(O)=N)N=C(O)C(CC(C)C)N=C(O)C(CCC(O)=O)N=C(O)C(CC(O)=O)N=C(O)C(CCC(O)=O)N=C(O)C(C(C)O)N=C(O)C(CCC(O)=N)N=C(O)C(CCC(O)=N)N=C(O)C(CCC(O)=N)N=C(O)C(CCC(O)=O)N=C(O)C(CCC(O)=O)N=C(O)C(COP(O)(O)=O)N=C(O)C(CCC(O)=N)N=C(O)C(N)CC1=CC=CC=C1 BECPQYXYKAMYBN-UHFFFAOYSA-N 0.000 description 1
- 235000021240 caseins Nutrition 0.000 description 1
- 210000004027 cell Anatomy 0.000 description 1
- 239000001913 cellulose Substances 0.000 description 1
- 229920002678 cellulose Polymers 0.000 description 1
- 239000011248 coating agent Substances 0.000 description 1
- 235000013353 coffee beverage Nutrition 0.000 description 1
- 239000003086 colorant Substances 0.000 description 1
- 230000036461 convulsion Effects 0.000 description 1
- 238000001816 cooling Methods 0.000 description 1
- 239000006071 cream Substances 0.000 description 1
- 230000007812 deficiency Effects 0.000 description 1
- 235000019425 dextrin Nutrition 0.000 description 1
- 238000000502 dialysis Methods 0.000 description 1
- 230000000378 dietary effect Effects 0.000 description 1
- 235000014113 dietary fatty acids Nutrition 0.000 description 1
- 235000015872 dietary supplement Nutrition 0.000 description 1
- 239000003085 diluting agent Substances 0.000 description 1
- 239000007884 disintegrant Substances 0.000 description 1
- 239000002270 dispersing agent Substances 0.000 description 1
- 239000003995 emulsifying agent Substances 0.000 description 1
- 210000002472 endoplasmic reticulum Anatomy 0.000 description 1
- 239000000194 fatty acid Substances 0.000 description 1
- 229930195729 fatty acid Natural products 0.000 description 1
- 238000001914 filtration Methods 0.000 description 1
- 235000019634 flavors Nutrition 0.000 description 1
- 235000012041 food component Nutrition 0.000 description 1
- 235000019253 formic acid Nutrition 0.000 description 1
- 239000008369 fruit flavor Substances 0.000 description 1
- 235000015203 fruit juice Nutrition 0.000 description 1
- 235000011389 fruit/vegetable juice Nutrition 0.000 description 1
- 235000013376 functional food Nutrition 0.000 description 1
- WIGCFUFOHFEKBI-UHFFFAOYSA-N gamma-tocopherol Natural products CC(C)CCCC(C)CCCC(C)CCCC1CCC2C(C)C(O)C(C)C(C)C2O1 WIGCFUFOHFEKBI-UHFFFAOYSA-N 0.000 description 1
- 238000002523 gelfiltration Methods 0.000 description 1
- 239000003349 gelling agent Substances 0.000 description 1
- 235000011187 glycerol Nutrition 0.000 description 1
- 235000020251 goat milk Nutrition 0.000 description 1
- 230000036541 health Effects 0.000 description 1
- 230000002401 inhibitory effect Effects 0.000 description 1
- 239000007924 injection Substances 0.000 description 1
- 238000002347 injection Methods 0.000 description 1
- 229910052500 inorganic mineral Inorganic materials 0.000 description 1
- 238000004255 ion exchange chromatography Methods 0.000 description 1
- 229960000310 isoleucine Drugs 0.000 description 1
- AGPKZVBTJJNPAG-UHFFFAOYSA-N isoleucine Natural products CCC(C)C(N)C(O)=O AGPKZVBTJJNPAG-UHFFFAOYSA-N 0.000 description 1
- 230000003907 kidney function Effects 0.000 description 1
- 239000008101 lactose Substances 0.000 description 1
- 239000000711 locust bean gum Substances 0.000 description 1
- 235000010420 locust bean gum Nutrition 0.000 description 1
- 239000000314 lubricant Substances 0.000 description 1
- 235000019359 magnesium stearate Nutrition 0.000 description 1
- 230000014759 maintenance of location Effects 0.000 description 1
- 210000005075 mammary gland Anatomy 0.000 description 1
- 238000004519 manufacturing process Methods 0.000 description 1
- 239000000463 material Substances 0.000 description 1
- 229940126601 medicinal product Drugs 0.000 description 1
- 238000001471 micro-filtration Methods 0.000 description 1
- 239000011707 mineral Substances 0.000 description 1
- 230000001333 moisturizer Effects 0.000 description 1
- 210000000663 muscle cell Anatomy 0.000 description 1
- 229960003966 nicotinamide Drugs 0.000 description 1
- 235000005152 nicotinamide Nutrition 0.000 description 1
- 239000011570 nicotinamide Substances 0.000 description 1
- 229910052757 nitrogen Inorganic materials 0.000 description 1
- 235000012149 noodles Nutrition 0.000 description 1
- 235000015097 nutrients Nutrition 0.000 description 1
- 235000020824 obesity Nutrition 0.000 description 1
- 229940100688 oral solution Drugs 0.000 description 1
- 239000003960 organic solvent Substances 0.000 description 1
- 238000007911 parenteral administration Methods 0.000 description 1
- 239000000546 pharmaceutical excipient Substances 0.000 description 1
- WTJKGGKOPKCXLL-RRHRGVEJSA-N phosphatidylcholine Chemical compound CCCCCCCCCCCCCCCC(=O)OC[C@H](COP([O-])(=O)OCC[N+](C)(C)C)OC(=O)CCCCCCCC=CCCCCCCCC WTJKGGKOPKCXLL-RRHRGVEJSA-N 0.000 description 1
- 150000008104 phosphatidylethanolamines Chemical class 0.000 description 1
- 229910052697 platinum Inorganic materials 0.000 description 1
- 229920000642 polymer Polymers 0.000 description 1
- 150000008442 polyphenolic compounds Chemical class 0.000 description 1
- 235000013824 polyphenols Nutrition 0.000 description 1
- 235000021251 pulses Nutrition 0.000 description 1
- 235000020185 raw untreated milk Nutrition 0.000 description 1
- 238000003753 real-time PCR Methods 0.000 description 1
- 229960002477 riboflavin Drugs 0.000 description 1
- 235000013580 sausages Nutrition 0.000 description 1
- 230000028327 secretion Effects 0.000 description 1
- WXMKPNITSTVMEF-UHFFFAOYSA-M sodium benzoate Chemical compound [Na+].[O-]C(=O)C1=CC=CC=C1 WXMKPNITSTVMEF-UHFFFAOYSA-M 0.000 description 1
- 235000010234 sodium benzoate Nutrition 0.000 description 1
- 239000004299 sodium benzoate Substances 0.000 description 1
- 238000000638 solvent extraction Methods 0.000 description 1
- 229940001941 soy protein Drugs 0.000 description 1
- 235000012424 soybean oil Nutrition 0.000 description 1
- 239000003549 soybean oil Substances 0.000 description 1
- 239000008347 soybean phospholipid Substances 0.000 description 1
- 239000006228 supernatant Substances 0.000 description 1
- 239000004094 surface-active agent Substances 0.000 description 1
- 229960003080 taurine Drugs 0.000 description 1
- UIERGBJEBXXIGO-UHFFFAOYSA-N thiamine mononitrate Chemical compound [O-][N+]([O-])=O.CC1=C(CCO)SC=[N+]1CC1=CN=C(C)N=C1N UIERGBJEBXXIGO-UHFFFAOYSA-N 0.000 description 1
- 239000002562 thickening agent Substances 0.000 description 1
- 230000007704 transition Effects 0.000 description 1
- 150000003626 triacylglycerols Chemical class 0.000 description 1
- 239000004474 valine Substances 0.000 description 1
- 235000019164 vitamin B2 Nutrition 0.000 description 1
- 239000011716 vitamin B2 Substances 0.000 description 1
- QYSXJUFSXHHAJI-YRZJJWOYSA-N vitamin D3 Chemical compound C1(/[C@@H]2CC[C@@H]([C@]2(CCC1)C)[C@H](C)CCCC(C)C)=C\C=C1\C[C@@H](O)CCC1=C QYSXJUFSXHHAJI-YRZJJWOYSA-N 0.000 description 1
- 235000005282 vitamin D3 Nutrition 0.000 description 1
- 239000011647 vitamin D3 Substances 0.000 description 1
- 235000019165 vitamin E Nutrition 0.000 description 1
- 239000011709 vitamin E Substances 0.000 description 1
- 229940046009 vitamin E Drugs 0.000 description 1
- 229940023308 vitamin b6 20 mg Drugs 0.000 description 1
- 229940021056 vitamin d3 Drugs 0.000 description 1
- 150000003722 vitamin derivatives Chemical class 0.000 description 1
- 230000002747 voluntary effect Effects 0.000 description 1
- 235000012431 wafers Nutrition 0.000 description 1
- 235000013618 yogurt Nutrition 0.000 description 1
- UHVMMEOXYDMDKI-JKYCWFKZSA-L zinc;1-(5-cyanopyridin-2-yl)-3-[(1s,2s)-2-(6-fluoro-2-hydroxy-3-propanoylphenyl)cyclopropyl]urea;diacetate Chemical compound [Zn+2].CC([O-])=O.CC([O-])=O.CCC(=O)C1=CC=C(F)C([C@H]2[C@H](C2)NC(=O)NC=2N=CC(=CC=2)C#N)=C1O UHVMMEOXYDMDKI-JKYCWFKZSA-L 0.000 description 1
Images
Abstract
Description
本発明は、運動機能向上効果を発揮する医薬品、食品等に関する。 The present invention relates to pharmaceuticals, foods, and the like that exhibit an effect of improving motor function.
一般的に、筋力等の運動能力の向上には、運動トレーニングとバランスの良い栄養補給が重要と考えられている。最近では、運動愛好者やアスリートにおいて、より効率的に筋力向上を図るため、単にトレーニングを行うだけでなく、サプリメント等の栄養補給を併用する試みがなされている(特許文献1)。 In general, exercise training and well-balanced nutrition are considered important for improving athletic ability such as muscle strength. In recent years, exercise enthusiasts and athletes have attempted to use nutrition supplements such as supplements in addition to training to improve muscle strength more efficiently (Patent Document 1).
しかしながら、一部のタンパク質やアミノ酸を過剰摂取した状態でトレーニングを行うことは、腎機能等に悪影響を及ぼす原因になりうることが懸念されている(非特許文献1)。
一方、運動愛好者やアスリート以外においても、無理なダイエットよって栄養成分の体内補給が不足し、骨格筋の減少、及び筋力や持久力をはじめとする運動機能が衰退すること、更には、運動機能衰退に伴う疲労が問題視されている。
従って、パフォーマンス向上を目指す運動愛好者やアスリートだけでなく、肥満軽減を目指す一般人においても、効率的な運動機能向上方法が望まれている。
However, there is a concern that training with excessive intake of some proteins and amino acids may cause adverse effects on renal function and the like (Non-patent Document 1).
On the other hand, even for non-exercise enthusiasts and athletes, due to an unreasonable diet, there is a shortage of nutritional components in the body, skeletal muscle loss, and motor functions such as muscle strength and endurance decline, and motor functions Fatigue associated with decline is regarded as a problem.
Therefore, not only exercise enthusiasts and athletes aiming at performance improvement, but also general people aiming at alleviating obesity, an efficient method for improving motor function is desired.
斯かる観点から、運動機能向上作用を有する成分の探索が行われ、例えば、茶カテキンによる持久力向上作用(特許文献2)や重合体果実ポリフェノール(特許文献3)に筋力向上作用等が報告されている。 From such a point of view, a component having a motor function improving action is searched, and for example, endurance improving action by tea catechin (Patent Document 2) and polymer fruit polyphenol (Patent Document 3) are reported. ing.
脂肪球皮膜成分は、乳腺より分泌される乳脂肪球を被覆している膜であって、脂肪を乳汁中に分散させる機能を有するのみならず、新生動物の食物として多くの生理的機能を有している。例えば、血中アディポネクチン増加及び/又は減少抑制効果(特許文献4)、学習能向上効果(特許文献5)、シアロムチンの分泌促進効果等(特許文献6)の生理機能を有することが知られている。 The fat globule membrane component is a film that coats milk fat globule secreted from the mammary gland and has not only the function of dispersing fat in milk but also has many physiological functions as food for newborn animals. is doing. For example, it is known to have physiological functions such as blood adiponectin increase and / or decrease inhibitory effect (Patent Document 4), learning ability improving effect (Patent Document 5), sialomucin secretion promoting effect and the like (Patent Document 6). .
しかし、脂肪球皮膜成分が、持久力をはじめとする運動機能や疲労に対して与える影響については、これまで全く知られていない。 However, the effect of fat globule membrane components on endurance and other motor functions and fatigue has never been known.
本発明は、食経験が豊富で安全性が高く、優れた運動機能向上作用、持久力向上作用、抗疲労作用、筋力向上作用、グリコーゲン蓄積促進作用、又は筋ポンプ機能向上作用を示す医薬品、医薬部外品、食品及び飼料を提供することに関する。 The present invention is a medicinal product or pharmaceutical having rich dietary experience and high safety and having excellent motor function improving action, endurance improving action, anti-fatigue action, muscle strength improving action, glycogen accumulation promoting action, or muscle pump function improving action It relates to providing quasi-drugs, food and feed.
本発明者らは、身体機能向上において有効な成分の探索を行った結果、乳由来成分の中から、脂肪球皮膜成分に運動機能向上作用、持久力向上作用、抗疲労作用、筋力向上作用、グリコーゲン蓄積促進作用、又は筋ポンプ機能向上作用の効果があり、これが当該作用効果を発揮し得る医薬品、医薬部外品、食品及び飼料として有用であることを見出した。 As a result of searching for an effective component in improving the body function, the present inventors, as a result of the milk-derived component, the fat globule film component, the motor function improving action, endurance improving action, anti-fatigue action, muscle strength improving action, It has been found that there is an effect of promoting the accumulation of glycogen or the function of improving the muscle pump function, which is useful as a pharmaceutical, a quasi-drug, a food and a feed that can exhibit the effect.
すなわち、本発明は、下記(1)〜(6)に係るものである。
(1)脂肪球皮膜成分を有効成分とする運動機能向上剤。
(2)脂肪球皮膜成分を有効成分とする持久力向上剤。
(3)脂肪球皮膜成分を有効成分とする抗疲労剤。
(4)脂肪球皮膜成分を有効成分とする筋力向上剤。
(5)脂肪球皮膜成分を有効成分とするグリコーゲン蓄積促進剤。
(6)脂肪球皮膜成分を有効成分とする筋ポンプ機能向上剤。
That is, the present invention relates to the following (1) to (6).
(1) A motor function improver comprising a fat globule membrane component as an active ingredient.
(2) Endurance improver comprising a fat globule membrane component as an active ingredient.
(3) An anti-fatigue agent comprising a fat globule film component as an active ingredient.
(4) A muscle strength improver comprising a fat globule membrane component as an active ingredient.
(5) A glycogen accumulation promoter comprising a fat globule membrane component as an active ingredient.
(6) A muscle pump function improver comprising a fat globule membrane component as an active ingredient.
本発明の運動機能向上剤、持久力向上剤、抗疲労剤、筋力向上剤、グリコーゲン蓄積促進剤及び筋ポンプ機能向上剤は、運動及び日常の動作及び労働を含む広義の運動に対して、運動機能の向上、持久力向上、抗疲労、筋力向上、グリコーゲン蓄積促進、又は筋ポンプ機能向上のための食品、医薬品、医薬部外品又は飼料として有用である。 The motor function improver, endurance improver, anti-fatigue agent, muscle strength improver, glycogen accumulation promoter and muscle pump function improver of the present invention are exercises in a broad sense including exercise and daily movements and labor. It is useful as a food, pharmaceutical, quasi-drug or feed for improving function, endurance, anti-fatigue, muscle strength, promoting glycogen accumulation, or improving muscle pump function.
本発明における脂肪球皮膜成分としては、乳中の脂肪球を皮膜している成分、及びそれに由来する膜成分混合物が挙げられる。
脂肪球皮膜成分は、バターミルクやバターセーラム等の乳複合脂質高含有画分に多く含まれることが知られ、脂肪球皮膜成分の乾燥量の約半分が脂質で構成されている(三浦晋、FOOD STYLE21、2009)。当該脂質としては、トリグリセライドやリン脂質(例えば、スフィンゴリン脂質、グリセロリン脂質等)が多く含まれ、これ以外にスフィンゴ糖脂質(例えば、グルコシルセラミド、ガングリオシド等)が含まれることが知られている(Keenan、Applied Sci、1983)。また、脂質以外の成分としては、ミルクムチンと呼ばれる糖蛋白質(Mather、Biochim Biophys Acta、1978)等の蛋白質が含まれることが知られている。
Examples of the fat globule membrane component in the present invention include a component that coats a fat globule in milk and a membrane component mixture derived therefrom.
The fat globule membrane component is known to be contained in a large amount in milk complex lipid-rich fractions such as buttermilk and buttersarum, and about half of the dry amount of the fat globule membrane component is composed of lipids (Miura Aoi, FOOD STYLE21, 2009). As the lipid, it is known that many triglycerides and phospholipids (for example, sphingophospholipids, glycerophospholipids, etc.) are included, and in addition, glycosphingolipids (for example, glucosylceramide, ganglioside, etc.) are included ( Keenan, Applied Sci, 1983). In addition, it is known that components other than lipids include proteins such as glycoprotein called milk mucin (Mather, Biochim Biophys Acta, 1978).
本発明における脂肪球皮膜成分に含まれるリン脂質としては、スフィンゴミエリン等のスフィンゴリン脂質の他、ホスファチジルコリンやホスファチジルエタノールアミン等のグリセロリン脂質が挙げられる。この中で、乳由来の特徴的なリン脂質であるスフィンゴミエリンが脂肪球皮膜成分に含まれることが好ましい。 Examples of the phospholipid contained in the fat globule membrane component in the present invention include glycerophospholipids such as phosphatidylcholine and phosphatidylethanolamine in addition to sphingophospholipids such as sphingomyelin. Of these, sphingomyelin, which is a characteristic phospholipid derived from milk, is preferably contained in the fat globule membrane component.
本発明における脂肪球皮膜成分中の脂質の含有量は、特に限定されないが、乾燥物換算で、20〜100質量%、より50〜90質量%であるのが好ましい。
本発明における脂肪球皮膜成分中のリン脂質の含有量は、特に限定されないが、乾燥物換算で、10〜100質量%、より15〜85質量%、更に20〜70質量%であるのが好ましい。
脂肪球皮膜成分中の各リン脂質の含有量は、特に限定されないが、例えば、スフィンゴミエリンの含有量は、脂肪球皮膜成分中、乾燥物換算で、1〜50質量%、より2〜30質量%、更に3〜25質量%、より更に4〜20質量%であるのが好ましい。
Although content of the lipid in the fat globule membrane component in the present invention is not particularly limited, it is preferably 20 to 100% by mass, more preferably 50 to 90% by mass in terms of dry matter.
The content of the phospholipid in the fat globule membrane component in the present invention is not particularly limited, but is preferably 10 to 100% by mass, more preferably 15 to 85% by mass, and further preferably 20 to 70% by mass in terms of dry matter. .
The content of each phospholipid in the fat globule membrane component is not particularly limited. For example, the content of sphingomyelin is 1 to 50 mass% in terms of dry matter in the fat globule membrane component, and more 2 to 30 mass%. %, 3 to 25% by mass, and more preferably 4 to 20% by mass.
本発明の脂肪球皮膜成分としては、乳原料等から遠心分離法や有機溶剤抽出法等の各種脂肪球皮膜成分の調製法により得たものを用いてもよい。さらに、透析、硫安分画、ゲルろ過、等電点沈殿、イオン交換クロマトグラフィー、溶媒分画等の手法により精製することにより純度を高めたものを用いてもよい。 As the fat globule membrane component of the present invention, those obtained by preparing various fat globule membrane components such as a centrifugal separation method or an organic solvent extraction method from a milk raw material or the like may be used. Furthermore, you may use what improved purity by refine | purifying with methods, such as a dialysis, an ammonium sulfate fraction, gel filtration, isoelectric precipitation, ion-exchange chromatography, and a solvent fraction.
本発明の脂肪球皮膜成分の乳原料としては、牛乳やヤギ乳等が挙げられるが、乳の中でも牛乳由来の脂肪球皮膜成分は、食経験が豊富であり、高純度かつ安価なものも上市されており、特に好ましい。
また、乳原料には、生乳、脱脂乳や加工乳等の乳の他、乳製品も含まれるが、乳製品としては、バターミルク、バターオイル、バターセーラム、ホエータンパク質濃縮物(WPC)等が挙げられる。
Examples of the milk raw material for the fat globule membrane component of the present invention include milk and goat milk, but among milk, the fat globule membrane component derived from milk has abundant food experience, and high purity and inexpensive products are also marketed. It is particularly preferable.
In addition to milk such as raw milk, skim milk and processed milk, dairy products include dairy products. Examples of dairy products include buttermilk, butter oil, buttersarum, whey protein concentrate (WPC) and the like. Can be mentioned.
本発明の脂肪球皮膜成分の調製は、例えば、乳やホエータンパク質濃縮物(WPC)、バターミルクや脱脂粉乳等の乳製品をエーテルやアセトンで抽出する方法(特開平3−47192号公報)、バターミルクを酸性域に調整、等電点沈殿を行うことにより生じたタンパク質を除去し、上清を精密濾過膜処理して得られる濃縮液を乾燥する方法(特許第3103218号公報)等により行うことができる。
また、バターセーラム中よりタンパク質を凝集除去後に濾過濃縮し乾燥する方法(特開2007−89535号公報)等も使用することができる。本製法によると、例えば、乳由来の複合脂質を乾燥物中20質量%以上含有する脂肪球皮膜成分を調製することができる。なお、脂肪球皮膜成分の形態は、特に限定されず、液状、半固体状や個体状、粉状等の何れでもよく、これらを単独で又は2種以上組み合わせて用いてもよい。
Preparation of the fat globule membrane component of the present invention is, for example, a method of extracting dairy products such as milk, whey protein concentrate (WPC), buttermilk and skim milk powder with ether or acetone (JP-A-3-47192), It is performed by a method (patent No. 3103218) or the like that removes proteins produced by adjusting buttermilk to an acidic region and performing isoelectric precipitation and drying the concentrate obtained by microfiltration membrane treatment of the supernatant. be able to.
Further, a method of filtering and concentrating the protein after removing the protein from the buttersarum and drying it (Japanese Patent Application Laid-Open No. 2007-89535) can also be used. According to this production method, for example, a fat globule membrane component containing 20% by mass or more of milk-derived complex lipid in the dried product can be prepared. The form of the fat globule membrane component is not particularly limited, and may be any of liquid, semi-solid, solid, powder, etc., and these may be used alone or in combination of two or more.
また、脂肪球皮膜成分として、市販品を用いることもできる。斯かる市販品としては、メグレジャパン(株)「BSCP」、雪印乳業(株)「ミルクセラミドMC-5」、(株)ニュージーランドミルクプロダクツ「Phospholipid Concentrate シリーズ(500, 700)」等が挙げられる。
また、脂肪球皮膜成分は牛乳等を遠心分離して得られるクリームからバター粒を製造する際に得られるバターミルク中に多く含まれているので、バターミルクをそのまま使用してもよい。同様に、脂肪球皮膜成分はバターオイルを製造する際に生じるバターセーラム中に多く含まれているので、バターセーラムをそのまま使用してもよい。
Moreover, a commercial item can also be used as a fat globule membrane component. Examples of such commercial products include Megre Japan Co., Ltd. “BSCP”, Snow Brand Milk Products Co., Ltd. “Milk Ceramide MC-5”, New Zealand Milk Products Co., Ltd. “Phospholipid Concentrate Series (500, 700)”, and the like.
Further, since the fat globule membrane component is contained in a large amount in butter milk obtained when producing butter granules from a cream obtained by centrifuging milk or the like, butter milk may be used as it is. Similarly, since the fat globule membrane component is contained in a large amount in the butter serum produced when producing the butter oil, the butter serum may be used as it is.
本発明の脂肪球皮膜成分は、後記実施例に示すように、マウスにおいて、自発運動量を有意に向上させ、ひらめ筋の筋力を有意に増大させたことから、運動機能向上作用、筋力向上作用を有し、また、遊泳持久力を向上させたことから、持久力向上作用、抗疲労作用を有する。また、本発明の脂肪球皮膜成分は、マウスにおいて、骨格筋中のグリコーゲン含量を有意に増加させ、筋ポンプ機能に関連する遺伝子の発現量を有意に増加させることから、グリコーゲン蓄積促進作用、筋ポンプ機能向上作用を有する。
従って、脂肪球皮膜成分は、運動機能向上剤、持久力向上剤、抗疲労剤、筋力向上剤、グリコーゲン蓄積促進剤、筋ポンプ機能向上剤(以下、「運動機能向上剤等」とする。)として、使用することができ、さらにこれらの剤を製造するために使用することができる。このとき、当該運動機能向上剤等には、脂肪球皮膜成分を単独で、又はこれ以外に、必要に応じて適宜選択した、担体、安定化剤等の、配合すべき後述の対象物において許容されるものを使用してもよい。なお、当該製剤は配合すべき対象物に応じて常法により製造することができる。
The fat globule membrane component of the present invention, as shown in the examples below, significantly improved the amount of spontaneous exercise and significantly increased the muscle strength of the soleus muscle in mice. Moreover, since it has improved swimming endurance, it has endurance improvement action and anti-fatigue action. In addition, the fat globule membrane component of the present invention significantly increases the glycogen content in skeletal muscle and significantly increases the expression level of genes related to muscle pump function in mice. Has an effect of improving the pump function.
Therefore, the fat globule membrane component is an exercise function improver, endurance improver, anti-fatigue agent, muscle strength improver, glycogen accumulation promoter, muscle pump function improver (hereinafter referred to as “motor function improver”). And can be used to produce these agents as well. In this case, the motor function improver and the like are acceptable in the later-described objects to be blended, such as a carrier, a stabilizer, and the like, which are appropriately selected as necessary, in addition to the fat globule membrane component. You may use what is done. In addition, the said formulation can be manufactured by a conventional method according to the target object which should be mix | blended.
そして、運動機能向上剤等は、運動機能向上、持久力向上、抗疲労、筋力向上、グリコーゲン蓄積促進、又は筋ポンプ機能向上の各効果を発揮する、ヒト若しくは動物用の医薬品、医薬部外品、食品、又は飼料の有効成分として配合して使用可能である。また、脂肪球皮膜成分は、運動不足者や中高年者、ベッドレスト者、或いはアスリートにおける運動機能向上、持久力向上、抗疲労、筋力向上、グリコーゲン蓄積促進、又は筋ポンプ機能向上をコンセプトとし、必要に応じてその旨を表示した食品、機能性食品、病者用食品、特定保健用食品に応用できる。 Motor function improvers and the like are drugs or quasi drugs for humans or animals that exhibit the effects of motor function improvement, endurance improvement, anti-fatigue, muscle strength improvement, glycogen accumulation promotion, or muscle pump function improvement. It can be used as an active ingredient in foods or feeds. In addition, fat globule membrane components are required for the concept of improving exercise function, endurance, anti-fatigue, muscle strength, promoting glycogen accumulation, or improving muscle pump function in people with exercise deficiency, middle-aged and elderly people, bed rest persons, or athletes. It can be applied to foods, functional foods, foods for the sick, and foods for specified health use.
本発明の運動機能向上剤等を医薬品、医薬部外品の有効成分として用いた場合の投与形態としては、例えば錠剤、カプセル剤、顆粒剤、散剤、シロップ剤等による経口投与又は注射剤、坐剤、吸入薬、経皮吸収剤、外用剤等による非経口投与が挙げられる。また、このような種々の剤型の製剤を調製するには、本発明の運動機能向上剤等を単独で、又は他の薬学的に許容される賦形剤、結合剤、増量剤、崩壊剤、界面活性剤、滑沢剤、分散剤、緩衝剤、保存剤、嬌味剤、香料、被膜剤、担体、希釈剤、脂肪球皮膜成分以外の薬効成分等を適宜組み合わせて用いることができる。また、これらの投与形態のうち、好ましい形態は経口投与であり、経口用液体製剤を調製する場合は、嬌味剤、緩衝剤、安定化剤等を加えて常法により製造することができる。 Examples of the dosage form when the motor function improver of the present invention is used as an active ingredient of pharmaceuticals and quasi-drugs include oral administration or injections such as tablets, capsules, granules, powders, and syrups. Parenteral administration such as an agent, an inhalation agent, a transdermal absorption agent, and an external preparation. In order to prepare such various dosage forms, the motor function improver of the present invention alone or other pharmaceutically acceptable excipients, binders, extenders, disintegrants. , Surfactants, lubricants, dispersants, buffers, preservatives, flavoring agents, fragrances, coating agents, carriers, diluents, medicinal components other than fat globule film components, and the like can be used in appropriate combinations. Of these dosage forms, the preferred form is oral administration, and when an oral liquid preparation is prepared, it can be produced by a conventional method by adding a flavoring agent, a buffering agent, a stabilizer and the like.
本発明の運動機能向上剤等を食品の有効成分として用いた場合の形態としては、牛乳、加工乳、乳飲料、ヨーグルト、清涼飲料水、茶系飲料、コーヒー飲料、果汁飲料、炭酸飲料、ジュース、ゼリー、ウエハース、ビスケット、パン、麺、ソーセージ等の飲食品や栄養食等の各種食品の他、さらには、上述した経口投与製剤と同様の形態(錠剤、カプセル剤、シロップ等)の栄養補給用組成物が挙げられる。 As the form when the motor function improver of the present invention is used as an active ingredient of food, milk, processed milk, milk drink, yogurt, soft drink, tea-based drink, coffee drink, fruit drink, carbonated drink, juice In addition to foods and drinks such as jelly, wafers, biscuits, bread, noodles, sausages and various foods such as nutritional foods, nutritional supplements in the same form (tablets, capsules, syrups, etc.) as the above-mentioned oral preparations Composition for use.
種々の形態の食品を調製するには、本発明の運動機能向上剤等を単独で、又は他の食品材料や、溶剤、軟化剤、油、乳化剤、防腐剤、香科、安定剤、着色剤、酸化防止剤、保湿剤、増粘剤、脂肪球皮膜成分以外の有効成分等を適宜組み合わせて運動機能向上用食品、持久力向上用食品、抗疲労用食品、筋力向上用食品、ペットフード等に配合することが可能である。 To prepare various forms of food, the motor function improver of the present invention alone, or other food materials, solvents, softeners, oils, emulsifiers, preservatives, fragrances, stabilizers, colorants , Antioxidants, moisturizers, thickeners, active ingredients other than fat globule membrane components, etc., in combination as appropriate, food for improving motor function, food for improving endurance, food for anti-fatigue, food for improving muscle strength, pet food, etc. It is possible to blend in.
また、本発明の運動機能向上剤等は、適当量の栄養補給が困難な高齢者やベッドレスト状態の病者においては、経腸栄養剤等の栄養組成物の形態として配合することが可能である。 In addition, the motor function improver of the present invention can be blended in the form of a nutritional composition such as an enteral nutrient in elderly people who are difficult to supplement with an appropriate amount of nutrition or in bed-rested patients. is there.
本発明の運動機能向上剤等を飼料の有効成分として用いた場合は、上記食品と同様の形態に使用することができる。 When the motor function improver or the like of the present invention is used as an active ingredient in feed, it can be used in the same form as the food.
上記飲料、例えば乳飲料、清涼飲料水、茶系飲料等に対する脂肪球皮膜成分(乾燥物換算)の含有量は、飲料中、通常0.001〜3.0質量%、さらに0.01〜2.0質量%、特に0.1〜1.0質量%とするのが好ましい。 The content of the fat globule membrane component (in terms of dry matter) for the beverages such as milk beverages, soft drinks, tea beverages, etc. is usually 0.001 to 3.0% by mass, and further 0.01 to 2 in the beverage. It is preferably 0.0% by mass, particularly 0.1 to 1.0% by mass.
上記以外の食品や飼料、また医薬品、例えば錠剤、顆粒剤、カプセル剤等の経口用固形製剤、内服液剤、シロップ剤等の経口用液体製剤の場合には、上記以外の食品、飼料又は医薬品等の各全量中における脂肪球皮膜成分(乾燥物換算)の含有量は、通常0.02〜80質量%、さらに0.2〜75質量%、特に2〜50質量%とするのが好ましい。尚、脂肪球皮膜成分は、溶解状態であっても、分散状態であっても良く、その存在状態は問わない。 In the case of foods and feeds other than the above, and pharmaceuticals such as oral solid preparations such as tablets, granules and capsules, and oral liquid preparations such as oral liquids and syrups, foods, feeds or pharmaceuticals other than those described above The content of the fat globule film component (in terms of dry matter) in the total amount is usually 0.02 to 80% by mass, more preferably 0.2 to 75% by mass, and particularly preferably 2 to 50% by mass. In addition, the fat globule membrane component may be in a dissolved state or a dispersed state, and the presence state thereof does not matter.
本発明の運動機能向上剤等の摂取量は、剤形や用途によって異なるが、脂肪球皮膜成分(乾燥物換算)として、成人に対して1日あたり、10〜10000mg/60kg体重とするのが好ましく、特に100〜5000mg/60kg体重、更に500〜5000mg/60kg体重とするのが好ましい。 The intake of the motor function improver and the like of the present invention varies depending on the dosage form and use, but it is 10 to 10,000 mg / 60 kg body weight per day for adults as a fat globule membrane component (in terms of dry matter). Particularly preferred is 100 to 5000 mg / 60 kg body weight, more preferably 500 to 5000 mg / 60 kg body weight.
以下に本発明の代表的な試験例と実施例を示す。 The typical test examples and examples of the present invention are shown below.
試験例1
脂肪球皮膜成分が自発運動量および筋力向上に及ぼす効果を検討した。脂肪球皮膜成分としては、メグレジャパン社製 BSCPを使用した(表1:飼料の組成)。
BSCPには、乾燥物換算で、蛋白質 49質量%(以下、「%」とする)及び脂質 39%が含まれ、スフィンゴリン脂質としてスフィンゴミエリン 3.7%が含まれ、スフィンゴ糖脂質としてグルコシルセラミド 2.4%,およびガングリオシド 0.4%が含まれていた。
Test example 1
We investigated the effects of fat globule membrane components on spontaneous exercise and muscle strength. As the fat globule film component, BSCP manufactured by Megre Japan was used (Table 1: Composition of feed).
BSCP contains 49% by mass of protein (hereinafter referred to as “%”) and 39% of lipid, 3.7% of sphingomyelin as sphingophospholipid, and 2.4% of glucosylceramide as glycosphingolipid in terms of dry matter. , And 0.4% ganglioside.
脂肪球皮膜成分中の蛋白質及び脂質の分析方法としては、ケルダール法(神立誠著、最新食品分析法、同文書院)及びレーゼゴットリーブ法(日本食品工業学会編、食品分析法、光琳)で行なった。
また、脂肪球皮膜成分中のリン脂質の分析は、LC-MS法にて行なった。すなわち、脂肪球皮膜成分よりクロロホルム/メタノール(=2:1)を用いて脂質画分を抽出し、窒素気流下で乾固した後、ヘキサン/イソプロパノール(=95:5)に溶解した。この試料を、下記LC-MS分析に供し、リン脂質の定量を行った。
具体的な分析手段としては、以下のものを用いた。
カラム:Inertsil SIL 100A-3 (GLサイエンス社、1.5mm×150mm)
カラム温度:40℃
流速:0.1 mL/min
検出器:アジレント、1100 LC/MSD
移動相:A液(ヘキサン:イソプロパノール:ギ酸=95:5:0.1)およびB液(ヘキサン:イソプロパノール:50mMギ酸アンモニウム=25:65:10)のグラジエント分離
Proteins and lipids in fat globule membrane components are analyzed by the Kjeldahl method (Makoto Kateri, latest food analysis method, the same documentary institute) and the Rosette Gottlieb method (edited by the Japan Food Industry Association, food analysis method, Korin). It was.
The analysis of phospholipids in the fat globule membrane component was performed by LC-MS method. That is, the lipid fraction was extracted from the fat globule membrane component using chloroform / methanol (= 2: 1), dried in a nitrogen stream, and then dissolved in hexane / isopropanol (= 95: 5). This sample was subjected to the following LC-MS analysis to quantify phospholipids.
As specific analysis means, the following were used.
Column: Inertsil SIL 100A-3 (GL Science, 1.5mm x 150mm)
Column temperature: 40 ° C
Flow rate: 0.1 mL / min
Detector: Agilent, 1100 LC / MSD
Mobile phase: Gradient separation of liquid A (hexane: isopropanol: formic acid = 95: 5: 0.1) and liquid B (hexane: isopropanol: 50 mM ammonium formate = 25: 65: 10)
1週間の予備飼育後、体重および自発運動量を基準に、これらが等しくなるよう、2群に分け、それぞれ、Cont(標準食摂取)群およびMFGM(乳脂肪球皮膜成分食1摂取)群とした(各群n=6)。群分け後4週間、回転カゴ式自発運動量測定装置 SW-15(メルクエスト)を使用し、マウスの自発運動量を計測した。4週間飼育後、解剖に供し、ひらめ筋の筋力を測定した。摘出筋の筋力測定は、Cannonらの方法(Biomed Sci Instrum, 2005)に準じて行った。すなわち、マウスよりひらめ筋を摘出、縫合糸(#5-0 silk)を用いてトランスデューサー(WPI, FORT100)に固定し、37℃のKrebs溶液中(95%-O2, 5%-CO2通気)に浸漬した。電気刺激は、2本のプラチナ電極より行った。刺激は、シングルパルス(twitch)を施した後、40Hz、330ms(1/s)の刺激を2分間(120秒)繰り返し(tetanic)、トランスデューサーより得られるシグナル(g/mg muscle)を筋力として測定した。Cont群とMFGM群間の有意差検定は、repeated measure ANOVA(自発運動量)、またはstudent's t-test(筋力)により行った。 After pre-breeding for 1 week, divided into 2 groups so that they are equal based on body weight and voluntary exercise amount, respectively, the Cont (standard diet intake) group and the MFGM (milk fat globule membrane component 1 intake) group, respectively. (N = 6 for each group). For 4 weeks after grouping, the mouse's spontaneous momentum was measured using a rotating basket type spontaneous momentum measuring device SW-15 (Melquest). After breeding for 4 weeks, it was subjected to dissection and the muscle strength of the soleus was measured. The muscle strength of the isolated muscle was measured according to the method of Cannon et al. (Biomed Sci Instrum, 2005). That is, the soleus muscle was removed from the mouse, fixed to the transducer (WPI, FORT100) using a suture (# 5-0 silk), and in Krebs solution at 37 ° C. (95% -O 2 , 5% -CO 2 Dipped in aeration). Electrical stimulation was performed from two platinum electrodes. For stimulation, after applying a single pulse (twitch), 40Hz, 330ms (1 / s) stimulation is repeated for 2 minutes (120 seconds) (tetanic), and the signal (g / mg muscle) obtained from the transducer is used as muscle strength. It was measured. The significant difference test between the Cont group and the MFGM group was performed by repeated measure ANOVA (spontaneous exercise amount) or student's t-test (muscle strength).
その結果、MFGM群では、Cont群に対して、期間中の自発運動量が有意な高値を示した(図1)。また、筋力は、Cont群に対してMFGM群が有意な高値を示した(図2)。
自発的な運動量は、持久力や筋力をはじめとする運動機能の総和であり、また、運動機能が向上することにより、身体疲労に対する耐性が向上すると考えられる。本試験において、脂肪球皮膜成分は、自発運動量および摘出筋の筋力を増加させたことから、運動機能向上、持久力向上、筋力向上、および抗疲労に有効であることが明らかとなった。
As a result, in the MFGM group, the amount of spontaneous exercise during the period was significantly higher than that in the Cont group (FIG. 1). In addition, the muscle strength of the MFGM group was significantly higher than the Cont group (FIG. 2).
Spontaneous exercise amount is the sum of motor functions including endurance and muscle strength, and it is considered that resistance to physical fatigue is improved by improving motor functions. In this test, the fat globule membrane component increased the amount of spontaneous exercise and the muscle strength of the extracted muscle, and thus was found to be effective in improving motor function, endurance, muscle strength, and anti-fatigue.
試験例2
脂肪球皮膜成分が持久力に及ぼす効果を検討した。脂肪球皮膜成分としては、ニュージーランドミルクプロダクツ社製Phospholipid Concentrate 700を使用した(表2:飼料の組成)。
Phospholipid Concentrate 700には、乾燥物換算で、脂質85%が含まれ、スフィンゴミエリン16.5%が含まれていた。
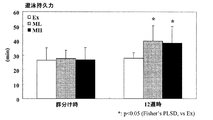
1週間の予備飼育後、7週齢の雄性BALB/cマウスを、体重と限界遊泳時間(=遊泳持久力)が等しくなるよう、3群に分け、各群を、Ex(標準食摂取+運動)群、ML(1%脂肪球皮膜成分食2摂取+運動)群、およびMH(3.5%脂肪球皮膜成分食3摂取+運動)群とした。遊泳持久力は、マウス用流水プール(京大松元式運動量測定流水槽)にて限界までの遊泳時間を測定した。群わけ後、試験食を給餌するとともに週2回の遊泳トレーニング(5 L/min, 30min)を施した。また、12週間飼育時に限界遊泳時間を計測し、脂肪球皮膜成分が持久力に及ぼす効果を検証した。
図3に、群分け時および12週時の遊泳持久力の結果を示す。ML群およびMH群の遊泳時間は、Ex群に対して有意な高値を示した。
Test example 2
The effect of fat globule membrane components on endurance was examined. As the fat globule membrane component, Phospholipid Concentrate 700 manufactured by New Zealand Milk Products was used (Table 2: Composition of feed).
Phospholipid Concentrate 700 contained 85% lipid and 16.5% sphingomyelin in terms of dry matter.
After 1 week of pre-breeding, 7-week-old male BALB / c mice were divided into 3 groups so that the body weight and limit swimming time (= swimming endurance) were equal, and each group was Ex (standard food intake + exercise) ) Group, ML (1% fat globule
FIG. 3 shows the results of swimming endurance at the time of grouping and at 12 weeks. The swimming time in the ML and MH groups was significantly higher than that in the Ex group.
この結果より、脂肪球皮膜成分が運動持久力の向上に有効であることが明らかとなった。さらに、運動持久力の向上は身体疲労耐性の向上を意味していることから、脂肪球皮膜成分は抗疲労に有効であることが明らかとなった。 From these results, it was revealed that the fat globule membrane component is effective in improving exercise endurance. Furthermore, since the improvement of exercise endurance means the improvement of physical fatigue resistance, it became clear that the fat globule membrane component is effective for anti-fatigue.
試験例3
脂肪球皮膜成分が骨格筋グリコーゲン含量、および遺伝子発現に及ぼす効果を検討した。脂肪球皮膜成分としては、雪印乳業(株)社製ミルクセラミドMC-5を使用した(表3:飼料の組成)。ミルクセラミドMC-5には、乾燥物換算で、蛋白質 21.2%および脂質 59.3%が含まれ、スフィンゴミエリン 6.9%が含まれていた。
Test example 3
The effects of fat globule membrane components on skeletal muscle glycogen content and gene expression were examined. As a fat globule membrane component, Milk Ceramide MC-5 manufactured by Snow Brand Milk Products Co., Ltd. was used (Table 3: Composition of feed). Milk ceramide MC-5 contained 21.2% protein and 59.3% lipid and 6.9% sphingomyelin in terms of dry matter.
1週間の予備飼育後、7週齢の雄性BALB/cマウスを、体重と限界遊泳時間(=遊泳持久力)が等しくなるよう、4群に分け、各群を、Cont(標準食摂取)群、MF(1%乳脂肪球皮膜成分食4摂取+運動)群、Ex(標準食摂取+運動)群、MF-Ex(1%乳脂肪球皮膜成分食4摂取+運動)群とした。群分け後、試験食を給餌した。Ex群、MF-Ex群については、週2回の遊泳トレーニング(5 L/min, 30min)を施し、Cont群、MF群については、遊泳トレーニングを施さなかった。
各群を12週間飼育時に限界遊泳時間を計測した。その後、13週間飼育時に解剖に供し、ひらめ筋の筋力測定を行った。解剖後、Xuら(J Cell Mol Med, 942-54, 2008)の方法に従って腓腹筋グリコーゲン含量を、また、ひらめ筋および腓腹筋における、Na+-K+ pumpβ1およびSERCA2の各遺伝子発現量を定量的PCR法(Murase et al, Biogerontology, 2009)により測定した。筋ポンプ機能関連の遺伝子発現量を測定する際に、用いたプライマー(配列番号1〜6)を表4に示す。なお、Na+-K+ pumpβ1は、Naポンプ機能に関連する遺伝子であり、SERCA2(sarcoplasmic /endoplasmic reticulum Ca2+ ATPase)は、Caポンプに関連する遺伝子として知られている。
After 1 week of preliminary breeding, 7-week-old male BALB / c mice were divided into 4 groups so that the body weight and limit swimming time (= swimming endurance) were equal, and each group was divided into Cont (standard food intake) groups. MF (1% milk fat globule
The limit swimming time was measured when each group was raised for 12 weeks. Thereafter, the animals were dissected for 13 weeks, and the muscle strength of the soleus was measured. After dissection, quantitative PCR of gastrocnemius glycogen content according to the method of Xu et al. (J Cell Mol Med, 942-54, 2008) and Na + -K + pumpβ1 and SERCA2 gene expression levels in soleus and gastrocnemius muscle Measured by the method (Murase et al, Biogerontology, 2009). Table 4 shows the primers (SEQ ID NOs: 1 to 6) used in measuring the gene expression level related to the muscle pump function. Na + -K + pump β1 is a gene related to Na pump function, and SERCA2 (sarcoplasmic / endoplasmic reticulum Ca 2+ ATPase) is known as a gene related to Ca pump.
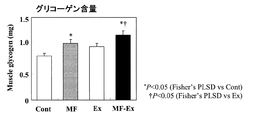
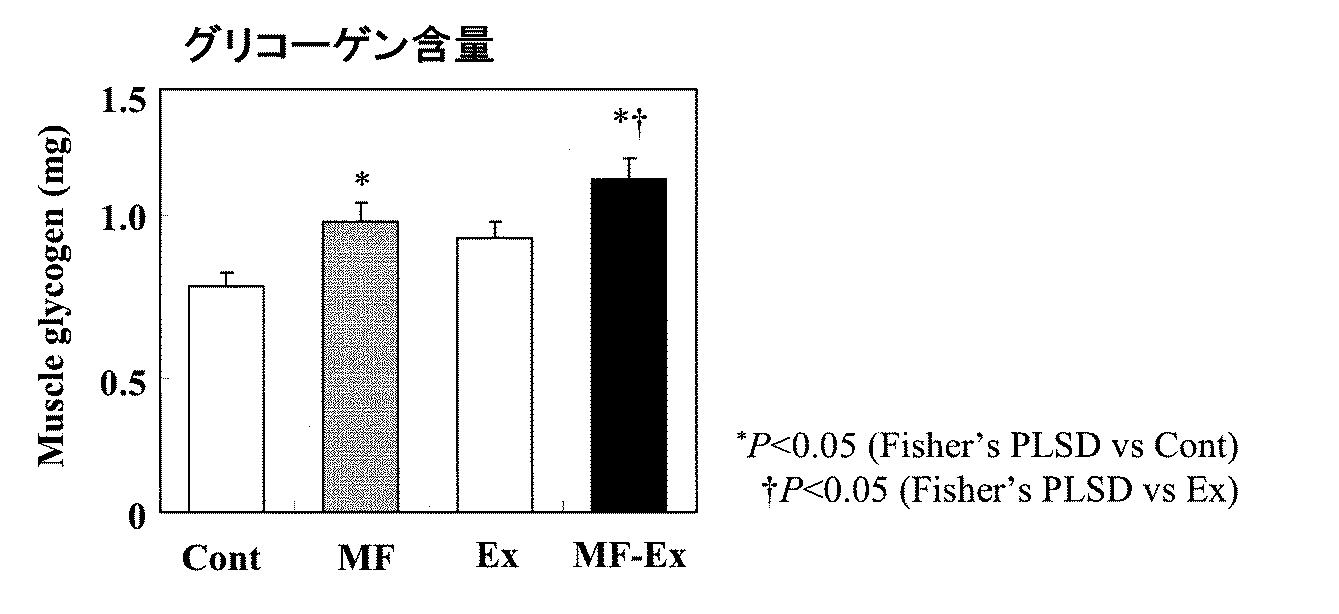
その結果、12週間飼育時における限界遊泳時間は、MF-Ex群(52.6 ( 4.5 min)でEx群(40.1 ( 3.8 min)に対して有意な高値を示し、乳脂肪球皮膜成分食摂取による持久力向上が認められた。また、13週間飼育時におけるひらめ筋筋力は、MF-Ex群が1.90 ( 0.08、Ex群が1.73 ( 0.07、Cont群が1.64 ( 0.07 (g/mg muscle)であり、乳脂肪球皮膜成分食摂取による筋力向上が認められた。
図4に、解剖時における腓腹筋グリコーゲン含量を示す。MF群およびMF-Ex群では、筋中グリコーゲンが有意な高値を示した。従って、乳脂肪球皮膜成分は、筋グリコーゲンの蓄積促進作用を有する事が明らかとなった。筋肉中のグリコーゲンは筋収縮の重要なエネルギー源であることから、乳脂肪球皮膜成分摂取による筋グリコーゲン量の増加は、運動機能向上に寄与すると考えられる。
As a result, the limit swimming time in the 12-week breeding period was significantly higher in the MF-Ex group (52.6 (4.5 min) than in the Ex group (40.1 (3.8 min)), and the endurance due to intake of milk fat globule membrane component foods In addition, the soleus muscle strength at 13 weeks of breeding was 1.90 (0.08 for the MF-Ex group, 1.73 (0.07 for the Ex group, 1.64 (0.07 (g / mg muscle) for the Cont group), The muscle strength was improved by the intake of milk fat globule membrane component food.
FIG. 4 shows the gastrocnemius glycogen content at the time of dissection. In the MF group and MF-Ex group, muscle glycogen showed a significantly high value. Therefore, it has been clarified that the milk fat globule membrane component has an action of promoting the accumulation of muscle glycogen. Since glycogen in muscle is an important energy source for muscle contraction, an increase in the amount of muscle glycogen due to intake of milk fat globule membrane component is considered to contribute to improvement of motor function.
図5に、解剖時のひらめ筋における筋ポンプ機能に関連する遺伝子発現量の結果を示す。MF群ではCont群に比し、MF-Ex群ではEx群に比し、それぞれ、Na+-K+ pumpβ1(Naポンプ)およびSERCA2(Caポンプ)の発現量が高値を示した。特に、MF-Ex群では、Cont群に比し、Na+-K+ pumpβ1(Naポンプ)およびSERCA2(Caポンプ)の発現量が有意な高値を示した。Naポンプは、活動電位保持を介して筋細胞膜の興奮性維持に、またCaポンプは、筋小胞体へのカルシウム取り込みに関与することが知られており、共に、筋収縮/筋力発揮において重要な役割を担っている。したがって、乳脂肪球皮膜成分は、筋ポンプ機能向上剤として有効であり、筋収縮関連ポンプ機能の向上を介して持久力や筋力等の運動機能向上に作用することが明らかとなった。 FIG. 5 shows the results of gene expression levels related to the muscle pump function in the soleus at the time of dissection. The expression levels of Na + -K + pumpβ1 (Na pump) and SERCA2 (Ca pump) were higher in the MF group than in the Cont group and in the MF-Ex group compared to the Ex group, respectively. In particular, the expression levels of Na + -K + pumpβ1 (Na pump) and SERCA2 (Ca pump) were significantly higher in the MF-Ex group than in the Cont group. Na pump is known to be involved in maintaining excitability of muscle cell membranes through action potential retention, and Ca pump is known to be involved in calcium uptake into the sarcoplasmic reticulum. Have a role. Therefore, it has been clarified that the milk fat globule membrane component is effective as a muscle pump function improving agent and acts on improvement of motor functions such as endurance and muscle strength through improvement of the muscle contraction related pump function.
製剤例
処方例1 運動機能向上用ゼリー食品
カラギーナンとローカストビーンガムの混合ゲル化剤0.65%、グレープフルーツの50%の濃縮果汁5.0%、クエン酸0.05%、ビタミンC0.05%、および脂肪球皮膜成分(ニュージーランドミルクプロダクツ社製 Phospholipid Concentrate 700: 脂質 85%,スフィンゴミエリン 16.5%)を2.0%混合し、これに水を加えて100%に調整し、65℃で溶解した。更に少量のグレープフルーツフレーバーを添加して85℃で5分間保持して殺菌処理後、100mLの容器に分注した。8時間静置して徐冷しながら5℃に冷却して、ゲル化させ、口に含んだ時に口溶け性が良好で、果実風味を有し食感良好な脂肪球皮膜成分を含有するゼリー食品を得た。
Formulation example Formulation example 1 Jelly food for improving motor function Carrageenan and locust bean gum mixed gelling agent 0.65%,
処方例2 運動機能向上用錠剤
アスコルビン酸180mg、クエン酸50mg、アスパルテーム12mg、ステアリン酸マグネシウム24mg、結晶セルロース120mg、乳糖274mg、および脂肪球皮膜成分(メグレ社製 BSCP: 蛋白質 49%, 脂質39%, スフィンゴミエリン 3.7%, グルコシルセラミド 2.4%, ガングリオシド 0.4%)440mgからなる処方(1日量2200mg)で、日本薬局方(製剤総則「錠剤」)に準じて錠剤を製造し、脂肪球皮膜成分を含有する錠剤を得た。
Formulation Example 2 Motor Function Improvement Tablet Ascorbic acid 180 mg,
処方例3 運動機能向上用ビタミン内服液
タウリン800mg、ショ糖2000mg、カラメル50mg、安息香酸ナトリウム30mg、ビタミンB1硝酸塩5mg、ビタミンB2 20mg、ビタミンB6 20mg、ビタミンC 2000mg、ビタミンE 100mg、ビタミンD3 2000IU、ニコチン酸アミド20mg、脂肪球皮膜成分(雪印乳業社製 ミルクセラミドMC-5: 蛋白質 21.2%, 脂質 59.3%, スフィンゴミエリン 6.9%)1000mg、ロイシン200mg、イソロイシン100mg、バリン100mgを適量の精製水に加えて溶解し、リン酸水溶液でpH3に調節した後、更に精製水を加えて全量を50mLとした。これを80℃で30分滅菌して、脂肪球皮膜成分及びアミノ酸類を含有する運動機能向上用飲料を得た。
Formulation Example 3 Vitamin oral solution for improving motor function Taurine 800 mg, sucrose 2000 mg,
処方例4 運動機能向上用乳系飲料
乳カゼイン3.4g、分離大豆タンパク質1.67g、デキストリン14.86g、ショ糖1.3g、大豆油1.75g、シソ油0.18g、大豆リン脂質0.14g、グリセリン脂肪酸エステル0.07g、ミネラル類0.60g、ビタミン類0.06g、脂肪球皮膜成分(ニュージーランドミルクプロダクツ社製 Phospholipid Concentrate 500: 脂質 89%, スフィンゴミエリン 7.8%)1.0gに精製水を加え、常法に従い、レトルト殺菌し、脂肪球皮膜成分を含有する運動機能向上用飲料(100mL)を得た。
Formulation Example 4 Milk Beverage for Improvement of Motor Function Milk Casein 3.4 g, Isolated Soy Protein 1.67 g, Dextrin 14.86 g, Sucrose 1.3 g, Soybean Oil 1.75 g, Perilla Oil 0.18 g,
Claims (6)
Priority Applications (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| JP2009183261A JP5882559B2 (en) | 2008-08-07 | 2009-08-06 | Motor function improver |
Applications Claiming Priority (3)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| JP2008203816 | 2008-08-07 | ||
| JP2008203816 | 2008-08-07 | ||
| JP2009183261A JP5882559B2 (en) | 2008-08-07 | 2009-08-06 | Motor function improver |
Related Child Applications (2)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| JP2014040738A Division JP2014141496A (en) | 2008-08-07 | 2014-03-03 | Exercise function-improving agent |
| JP2015243418A Division JP6232412B2 (en) | 2008-08-07 | 2015-12-14 | Motor function improver |
Publications (2)
| Publication Number | Publication Date |
|---|---|
| JP2010059155A true JP2010059155A (en) | 2010-03-18 |
| JP5882559B2 JP5882559B2 (en) | 2016-03-09 |
Family
ID=42186390
Family Applications (4)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| JP2009183261A Active JP5882559B2 (en) | 2008-08-07 | 2009-08-06 | Motor function improver |
| JP2014040738A Pending JP2014141496A (en) | 2008-08-07 | 2014-03-03 | Exercise function-improving agent |
| JP2015243418A Active JP6232412B2 (en) | 2008-08-07 | 2015-12-14 | Motor function improver |
| JP2016028047A Pending JP2016104805A (en) | 2008-08-07 | 2016-02-17 | Motor function improver |
Family Applications After (3)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| JP2014040738A Pending JP2014141496A (en) | 2008-08-07 | 2014-03-03 | Exercise function-improving agent |
| JP2015243418A Active JP6232412B2 (en) | 2008-08-07 | 2015-12-14 | Motor function improver |
| JP2016028047A Pending JP2016104805A (en) | 2008-08-07 | 2016-02-17 | Motor function improver |
Country Status (1)
| Country | Link |
|---|---|
| JP (4) | JP5882559B2 (en) |
Cited By (24)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2010134384A1 (en) * | 2009-05-20 | 2010-11-25 | よつ葉乳業株式会社 | Skin function-improving composition |
| WO2011096414A1 (en) * | 2010-02-03 | 2011-08-11 | 花王株式会社 | Mitochondrial function improver |
| WO2011096413A1 (en) * | 2010-02-03 | 2011-08-11 | 花王株式会社 | Agent for improving motility function |
| US8268360B2 (en) | 2010-08-26 | 2012-09-18 | Kao Corporation | Motor function improver |
| JP2012180311A (en) * | 2011-03-02 | 2012-09-20 | Kao Corp | Endurance improver |
| WO2013058229A1 (en) | 2011-10-17 | 2013-04-25 | 花王株式会社 | Agent for increasing amount of muscles |
| WO2014104316A1 (en) * | 2012-12-28 | 2014-07-03 | 花王株式会社 | Sphingomyelin-containing supplement |
| JP2014201572A (en) * | 2013-04-09 | 2014-10-27 | 花王株式会社 | Muscle protein synthesis signal enhancer |
| JP2015113322A (en) * | 2013-12-13 | 2015-06-22 | 花王株式会社 | Agility improvement agent |
| JP5816760B1 (en) * | 2014-06-27 | 2015-11-18 | 花王株式会社 | Solid composition |
| JP5816762B1 (en) * | 2014-06-27 | 2015-11-18 | 花王株式会社 | Solid composition |
| JP5816761B1 (en) * | 2014-06-27 | 2015-11-18 | 花王株式会社 | Solid composition |
| JP2016026491A (en) * | 2014-06-27 | 2016-02-18 | 花王株式会社 | Solid composition |
| JP2016123323A (en) * | 2014-12-26 | 2016-07-11 | 花王株式会社 | Solid composition |
| JP2016123322A (en) * | 2014-12-26 | 2016-07-11 | 花王株式会社 | Solid composition |
| JP2017077185A (en) * | 2015-10-19 | 2017-04-27 | 花王株式会社 | Beverage composition |
| JP2017088527A (en) * | 2015-11-06 | 2017-05-25 | 花王株式会社 | Production methods of milk fat globule membrane component-containing granulated materials |
| JP2017118821A (en) * | 2015-12-28 | 2017-07-06 | 花王株式会社 | Corn potage soup |
| JP2017197479A (en) * | 2016-04-28 | 2017-11-02 | 花王株式会社 | Solid composition |
| JP2017212961A (en) * | 2016-06-02 | 2017-12-07 | 花王株式会社 | Jelly-like solid food |
| JP2018083812A (en) * | 2017-11-30 | 2018-05-31 | 花王株式会社 | Agility improver |
| JP2018085965A (en) * | 2016-11-29 | 2018-06-07 | 花王株式会社 | milk beverage |
| JP2019182803A (en) * | 2018-04-13 | 2019-10-24 | 花王株式会社 | Muscle fatigue recovery agent |
| JP2021073268A (en) * | 2021-02-01 | 2021-05-13 | オリザ油化株式会社 | Muscle-enhancing agent |
Families Citing this family (2)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| EP3941451A4 (en) * | 2019-03-21 | 2022-12-14 | Fonterra Co-Operative Group Limited | Compositions comprising polar lipids for maintaining or increasing mobility and vitality |
| JP7365380B2 (en) * | 2021-09-22 | 2023-10-19 | 任天堂株式会社 | Game program, information processing system, information processing device, and game processing method |
Citations (4)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JP2007112793A (en) * | 2005-09-22 | 2007-05-10 | Snow Brand Milk Prod Co Ltd | Sphingomyelin-containing medicine, food and drink or fodder |
| JP2007246404A (en) * | 2006-03-14 | 2007-09-27 | Snow Brand Milk Prod Co Ltd | Learning ability-improving agent |
| WO2007138749A1 (en) * | 2006-05-31 | 2007-12-06 | Snow Brand Milk Products Co., Ltd. | Visceral fat accumulation inhibitor, and agent for promoting the increase in and/or inhibiting the decrease in blood adiponectin level |
| WO2008016108A1 (en) * | 2006-08-04 | 2008-02-07 | Snow Brand Milk Products Co., Ltd. | Agent for preventing infection |
Family Cites Families (2)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JP4852684B2 (en) * | 2005-09-30 | 2012-01-11 | 雪印メグミルク株式会社 | Milk-derived complex lipid-rich powder |
| JP5209335B2 (en) * | 2008-02-08 | 2013-06-12 | 日本甜菜製糖株式会社 | Continuous potted seedling transplanting machine for dense planting in soft and soft fields |
-
2009
- 2009-08-06 JP JP2009183261A patent/JP5882559B2/en active Active
-
2014
- 2014-03-03 JP JP2014040738A patent/JP2014141496A/en active Pending
-
2015
- 2015-12-14 JP JP2015243418A patent/JP6232412B2/en active Active
-
2016
- 2016-02-17 JP JP2016028047A patent/JP2016104805A/en active Pending
Patent Citations (4)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JP2007112793A (en) * | 2005-09-22 | 2007-05-10 | Snow Brand Milk Prod Co Ltd | Sphingomyelin-containing medicine, food and drink or fodder |
| JP2007246404A (en) * | 2006-03-14 | 2007-09-27 | Snow Brand Milk Prod Co Ltd | Learning ability-improving agent |
| WO2007138749A1 (en) * | 2006-05-31 | 2007-12-06 | Snow Brand Milk Products Co., Ltd. | Visceral fat accumulation inhibitor, and agent for promoting the increase in and/or inhibiting the decrease in blood adiponectin level |
| WO2008016108A1 (en) * | 2006-08-04 | 2008-02-07 | Snow Brand Milk Products Co., Ltd. | Agent for preventing infection |
Cited By (35)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JP5679966B2 (en) * | 2009-05-20 | 2015-03-04 | よつ葉乳業株式会社 | Skin function improving composition |
| JPWO2010134384A1 (en) * | 2009-05-20 | 2012-11-08 | よつ葉乳業株式会社 | Skin function improving composition |
| WO2010134384A1 (en) * | 2009-05-20 | 2010-11-25 | よつ葉乳業株式会社 | Skin function-improving composition |
| WO2011096414A1 (en) * | 2010-02-03 | 2011-08-11 | 花王株式会社 | Mitochondrial function improver |
| WO2011096413A1 (en) * | 2010-02-03 | 2011-08-11 | 花王株式会社 | Agent for improving motility function |
| US8268360B2 (en) | 2010-08-26 | 2012-09-18 | Kao Corporation | Motor function improver |
| JP2012180311A (en) * | 2011-03-02 | 2012-09-20 | Kao Corp | Endurance improver |
| WO2013058229A1 (en) | 2011-10-17 | 2013-04-25 | 花王株式会社 | Agent for increasing amount of muscles |
| JP2013100275A (en) * | 2011-10-17 | 2013-05-23 | Kao Corp | Agent for increasing amount of muscle |
| US9649322B2 (en) | 2012-12-28 | 2017-05-16 | Kao Corporation | Sphingomyelin-containing supplement |
| JP2014141476A (en) * | 2012-12-28 | 2014-08-07 | Kao Corp | Sphingomyelin-containing supplement |
| WO2014104316A1 (en) * | 2012-12-28 | 2014-07-03 | 花王株式会社 | Sphingomyelin-containing supplement |
| JP2014201572A (en) * | 2013-04-09 | 2014-10-27 | 花王株式会社 | Muscle protein synthesis signal enhancer |
| JP2015113322A (en) * | 2013-12-13 | 2015-06-22 | 花王株式会社 | Agility improvement agent |
| TWI552754B (en) * | 2014-06-27 | 2016-10-11 | Kao Corp | Solid composition |
| US9956246B2 (en) | 2014-06-27 | 2018-05-01 | Kao Corporation | Solid ingestible composition comprising a fat globule membrane component and water-soluble dietary fiber |
| WO2015198482A1 (en) * | 2014-06-27 | 2015-12-30 | 花王株式会社 | Solid composition |
| WO2015198480A1 (en) * | 2014-06-27 | 2015-12-30 | 花王株式会社 | Solid composition |
| WO2015198481A1 (en) * | 2014-06-27 | 2015-12-30 | 花王株式会社 | Solid composition |
| JP2016026491A (en) * | 2014-06-27 | 2016-02-18 | 花王株式会社 | Solid composition |
| JP5816762B1 (en) * | 2014-06-27 | 2015-11-18 | 花王株式会社 | Solid composition |
| JP5816760B1 (en) * | 2014-06-27 | 2015-11-18 | 花王株式会社 | Solid composition |
| JP5816761B1 (en) * | 2014-06-27 | 2015-11-18 | 花王株式会社 | Solid composition |
| JP2016123323A (en) * | 2014-12-26 | 2016-07-11 | 花王株式会社 | Solid composition |
| JP2016123322A (en) * | 2014-12-26 | 2016-07-11 | 花王株式会社 | Solid composition |
| JP2017077185A (en) * | 2015-10-19 | 2017-04-27 | 花王株式会社 | Beverage composition |
| JP2017088527A (en) * | 2015-11-06 | 2017-05-25 | 花王株式会社 | Production methods of milk fat globule membrane component-containing granulated materials |
| JP2017118821A (en) * | 2015-12-28 | 2017-07-06 | 花王株式会社 | Corn potage soup |
| JP2017197479A (en) * | 2016-04-28 | 2017-11-02 | 花王株式会社 | Solid composition |
| JP2017212961A (en) * | 2016-06-02 | 2017-12-07 | 花王株式会社 | Jelly-like solid food |
| JP2018085965A (en) * | 2016-11-29 | 2018-06-07 | 花王株式会社 | milk beverage |
| JP2018083812A (en) * | 2017-11-30 | 2018-05-31 | 花王株式会社 | Agility improver |
| JP2019182803A (en) * | 2018-04-13 | 2019-10-24 | 花王株式会社 | Muscle fatigue recovery agent |
| JP2021073268A (en) * | 2021-02-01 | 2021-05-13 | オリザ油化株式会社 | Muscle-enhancing agent |
| JP7104193B2 (en) | 2021-02-01 | 2022-07-20 | オリザ油化株式会社 | Muscle builders |
Also Published As
| Publication number | Publication date |
|---|---|
| JP2016104805A (en) | 2016-06-09 |
| JP2016121132A (en) | 2016-07-07 |
| JP5882559B2 (en) | 2016-03-09 |
| JP2014141496A (en) | 2014-08-07 |
| JP6232412B2 (en) | 2017-11-15 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| JP6232412B2 (en) | Motor function improver | |
| JP5581071B2 (en) | Mitochondrial function improver | |
| EP2039365B1 (en) | Visceral fat accumulation inhibitor, and agent for promoting the increase in and/or inhibiting the decrease in blood adiponectin level | |
| JP5507802B2 (en) | Muscle aging inhibitor | |
| KR20080108523A (en) | Fat accumulation inhibitor | |
| WO2016163245A1 (en) | Activator of energy metabolism in muscle cells | |
| JP5972717B2 (en) | Cell membrane enhancer | |
| US8268360B2 (en) | Motor function improver | |
| JP5922863B2 (en) | Motor function improver | |
| US20150238453A1 (en) | Bio-available n-acetyl creatine species and compositions thereof | |
| KR20190091768A (en) | Health assistance food for promoting growth | |
| JP5922862B2 (en) | Mitochondrial function improver | |
| JP2011504464A (en) | Novel blood pressure lowering composition | |
| JP6158565B2 (en) | Muscle protein synthesis signal enhancer | |
| JP6787595B2 (en) | Blood flow improver, royal jelly composition and method for producing royal jelly composition | |
| JP6225199B2 (en) | Motor function improver | |
| JP2021107400A (en) | Obesity suppressant | |
| JP7300243B2 (en) | nutritional composition | |
| JP6254434B2 (en) | Agility improver | |
| JP6450823B2 (en) | Agility improver | |
| TW201808120A (en) | Agent for improving physical fitness | |
| JP2008031080A (en) | Lipid utilization-promoting composition | |
| JP2016135765A (en) | Prophylactic agent for muscular contracture | |
| JP2018016561A (en) | Remedy for stumbling while walking | |
| Singh | DAIRY NUTRACEUTICALS AND FUNCTIONAL DAIRY FOODS: CURRENT STATUS, ISSUES AND CHALLENGES |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| A621 | Written request for application examination |
Free format text: JAPANESE INTERMEDIATE CODE: A621 Effective date: 20110811 |
|
| A131 | Notification of reasons for refusal |
Free format text: JAPANESE INTERMEDIATE CODE: A131 Effective date: 20130709 |
|
| A02 | Decision of refusal |
Free format text: JAPANESE INTERMEDIATE CODE: A02 Effective date: 20131203 |
|
| A521 | Request for written amendment filed |
Free format text: JAPANESE INTERMEDIATE CODE: A821 Effective date: 20140304 |
|
| A521 | Request for written amendment filed |
Free format text: JAPANESE INTERMEDIATE CODE: A523 Effective date: 20150803 |
|
| A521 | Request for written amendment filed |
Free format text: JAPANESE INTERMEDIATE CODE: A523 Effective date: 20150916 |
|
| A521 | Request for written amendment filed |
Free format text: JAPANESE INTERMEDIATE CODE: A523 Effective date: 20151214 |
|
| A61 | First payment of annual fees (during grant procedure) |
Free format text: JAPANESE INTERMEDIATE CODE: A61 Effective date: 20160204 |
|
| R151 | Written notification of patent or utility model registration |
Ref document number: 5882559 Country of ref document: JP Free format text: JAPANESE INTERMEDIATE CODE: R151 |
|
| R250 | Receipt of annual fees |
Free format text: JAPANESE INTERMEDIATE CODE: R250 |
|
| R250 | Receipt of annual fees |
Free format text: JAPANESE INTERMEDIATE CODE: R250 |
|
| R250 | Receipt of annual fees |
Free format text: JAPANESE INTERMEDIATE CODE: R250 |
|
| R250 | Receipt of annual fees |
Free format text: JAPANESE INTERMEDIATE CODE: R250 |
|
| R250 | Receipt of annual fees |
Free format text: JAPANESE INTERMEDIATE CODE: R250 |
|
| R250 | Receipt of annual fees |
Free format text: JAPANESE INTERMEDIATE CODE: R250 |