JP2014201572A - Muscle protein synthesis signal enhancer - Google Patents
Muscle protein synthesis signal enhancer Download PDFInfo
- Publication number
- JP2014201572A JP2014201572A JP2013081115A JP2013081115A JP2014201572A JP 2014201572 A JP2014201572 A JP 2014201572A JP 2013081115 A JP2013081115 A JP 2013081115A JP 2013081115 A JP2013081115 A JP 2013081115A JP 2014201572 A JP2014201572 A JP 2014201572A
- Authority
- JP
- Japan
- Prior art keywords
- muscle
- mass
- fat globule
- milk fat
- amino acid
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Granted
Links
- 230000014616 translation Effects 0.000 title claims abstract description 30
- 102000008934 Muscle Proteins Human genes 0.000 title claims abstract description 29
- 108010074084 Muscle Proteins Proteins 0.000 title claims abstract description 29
- 238000001243 protein synthesis Methods 0.000 title claims abstract description 28
- 239000003623 enhancer Substances 0.000 title claims abstract description 15
- 239000012528 membrane Substances 0.000 claims abstract description 62
- 108010071421 milk fat globule Proteins 0.000 claims abstract description 55
- 150000001413 amino acids Chemical class 0.000 claims abstract description 52
- 210000003205 muscle Anatomy 0.000 claims abstract description 50
- 229940024606 amino acid Drugs 0.000 claims description 51
- 206010028289 Muscle atrophy Diseases 0.000 claims description 23
- 230000020763 muscle atrophy Effects 0.000 claims description 22
- 201000000585 muscular atrophy Diseases 0.000 claims description 22
- 230000001965 increasing effect Effects 0.000 claims description 21
- 239000003795 chemical substances by application Substances 0.000 claims description 14
- 239000003112 inhibitor Substances 0.000 claims description 10
- OCUSNPIJIZCRSZ-ZTZWCFDHSA-N (2s)-2-amino-3-methylbutanoic acid;(2s)-2-amino-4-methylpentanoic acid;(2s,3s)-2-amino-3-methylpentanoic acid Chemical compound CC(C)[C@H](N)C(O)=O.CC[C@H](C)[C@H](N)C(O)=O.CC(C)C[C@H](N)C(O)=O OCUSNPIJIZCRSZ-ZTZWCFDHSA-N 0.000 claims description 3
- 235000013305 food Nutrition 0.000 abstract description 28
- 239000003814 drug Substances 0.000 abstract description 17
- 229940079593 drug Drugs 0.000 abstract description 8
- 230000002708 enhancing effect Effects 0.000 abstract description 5
- 239000000463 material Substances 0.000 abstract description 5
- 239000008267 milk Substances 0.000 description 19
- 210000004080 milk Anatomy 0.000 description 19
- 235000013336 milk Nutrition 0.000 description 18
- ROHFNLRQFUQHCH-YFKPBYRVSA-N L-leucine Chemical compound CC(C)C[C@H](N)C(O)=O ROHFNLRQFUQHCH-YFKPBYRVSA-N 0.000 description 17
- 230000007423 decrease Effects 0.000 description 15
- 229960003136 leucine Drugs 0.000 description 15
- 238000000034 method Methods 0.000 description 15
- 210000002027 skeletal muscle Anatomy 0.000 description 15
- ROHFNLRQFUQHCH-UHFFFAOYSA-N Leucine Natural products CC(C)CC(N)C(O)=O ROHFNLRQFUQHCH-UHFFFAOYSA-N 0.000 description 13
- 230000000694 effects Effects 0.000 description 13
- 239000000203 mixture Substances 0.000 description 10
- 235000018102 proteins Nutrition 0.000 description 9
- 102000004169 proteins and genes Human genes 0.000 description 9
- 108090000623 proteins and genes Proteins 0.000 description 9
- 238000002360 preparation method Methods 0.000 description 8
- 239000000047 product Substances 0.000 description 7
- 108010013238 70-kDa Ribosomal Protein S6 Kinases Proteins 0.000 description 6
- 230000037396 body weight Effects 0.000 description 6
- 235000014121 butter Nutrition 0.000 description 6
- 235000015155 buttermilk Nutrition 0.000 description 6
- 241001465754 Metazoa Species 0.000 description 5
- 150000002632 lipids Chemical class 0.000 description 5
- 150000003904 phospholipids Chemical class 0.000 description 5
- 230000001629 suppression Effects 0.000 description 5
- 208000010428 Muscle Weakness Diseases 0.000 description 4
- 206010028372 Muscular weakness Diseases 0.000 description 4
- 108010065917 TOR Serine-Threonine Kinases Proteins 0.000 description 4
- 102000013530 TOR Serine-Threonine Kinases Human genes 0.000 description 4
- 230000032683 aging Effects 0.000 description 4
- 239000002552 dosage form Substances 0.000 description 4
- 235000019197 fats Nutrition 0.000 description 4
- 238000009472 formulation Methods 0.000 description 4
- 230000036541 health Effects 0.000 description 4
- 230000026731 phosphorylation Effects 0.000 description 4
- 238000006366 phosphorylation reaction Methods 0.000 description 4
- 230000001225 therapeutic effect Effects 0.000 description 4
- CSCPPACGZOOCGX-UHFFFAOYSA-N Acetone Chemical compound CC(C)=O CSCPPACGZOOCGX-UHFFFAOYSA-N 0.000 description 3
- 241000282412 Homo Species 0.000 description 3
- 241000700159 Rattus Species 0.000 description 3
- 239000012141 concentrate Substances 0.000 description 3
- 235000008504 concentrate Nutrition 0.000 description 3
- 235000013365 dairy product Nutrition 0.000 description 3
- -1 feed Substances 0.000 description 3
- 239000003921 oil Substances 0.000 description 3
- 230000001737 promoting effect Effects 0.000 description 3
- 210000002966 serum Anatomy 0.000 description 3
- 238000001262 western blot Methods 0.000 description 3
- RTZKZFJDLAIYFH-UHFFFAOYSA-N Diethyl ether Chemical compound CCOCC RTZKZFJDLAIYFH-UHFFFAOYSA-N 0.000 description 2
- AGPKZVBTJJNPAG-WHFBIAKZSA-N L-isoleucine Chemical compound CC[C@H](C)[C@H](N)C(O)=O AGPKZVBTJJNPAG-WHFBIAKZSA-N 0.000 description 2
- 235000019454 L-leucine Nutrition 0.000 description 2
- 239000004395 L-leucine Substances 0.000 description 2
- KDXKERNSBIXSRK-YFKPBYRVSA-N L-lysine Chemical compound NCCCC[C@H](N)C(O)=O KDXKERNSBIXSRK-YFKPBYRVSA-N 0.000 description 2
- COLNVLDHVKWLRT-QMMMGPOBSA-N L-phenylalanine Chemical compound OC(=O)[C@@H](N)CC1=CC=CC=C1 COLNVLDHVKWLRT-QMMMGPOBSA-N 0.000 description 2
- KZSNJWFQEVHDMF-BYPYZUCNSA-N L-valine Chemical compound CC(C)[C@H](N)C(O)=O KZSNJWFQEVHDMF-BYPYZUCNSA-N 0.000 description 2
- KDXKERNSBIXSRK-UHFFFAOYSA-N Lysine Natural products NCCCCC(N)C(O)=O KDXKERNSBIXSRK-UHFFFAOYSA-N 0.000 description 2
- 239000004472 Lysine Substances 0.000 description 2
- 206010028311 Muscle hypertrophy Diseases 0.000 description 2
- 108091007960 PI3Ks Proteins 0.000 description 2
- 102000003993 Phosphatidylinositol 3-kinases Human genes 0.000 description 2
- 108090000430 Phosphatidylinositol 3-kinases Proteins 0.000 description 2
- 102000002278 Ribosomal Proteins Human genes 0.000 description 2
- 108010000605 Ribosomal Proteins Proteins 0.000 description 2
- KZSNJWFQEVHDMF-UHFFFAOYSA-N Valine Natural products CC(C)C(N)C(O)=O KZSNJWFQEVHDMF-UHFFFAOYSA-N 0.000 description 2
- 108010046377 Whey Proteins Proteins 0.000 description 2
- 102000007544 Whey Proteins Human genes 0.000 description 2
- 230000004913 activation Effects 0.000 description 2
- 239000004480 active ingredient Substances 0.000 description 2
- 235000013361 beverage Nutrition 0.000 description 2
- 210000004369 blood Anatomy 0.000 description 2
- 239000008280 blood Substances 0.000 description 2
- 239000002775 capsule Substances 0.000 description 2
- 210000004027 cell Anatomy 0.000 description 2
- 239000003153 chemical reaction reagent Substances 0.000 description 2
- 238000006731 degradation reaction Methods 0.000 description 2
- 201000010099 disease Diseases 0.000 description 2
- 208000037265 diseases, disorders, signs and symptoms Diseases 0.000 description 2
- 238000001035 drying Methods 0.000 description 2
- 239000003797 essential amino acid Substances 0.000 description 2
- 235000020776 essential amino acid Nutrition 0.000 description 2
- 239000000796 flavoring agent Substances 0.000 description 2
- 235000013355 food flavoring agent Nutrition 0.000 description 2
- 230000037406 food intake Effects 0.000 description 2
- 238000005194 fractionation Methods 0.000 description 2
- 239000003205 fragrance Substances 0.000 description 2
- 230000006870 function Effects 0.000 description 2
- 150000002327 glycerophospholipids Chemical class 0.000 description 2
- 230000002209 hydrophobic effect Effects 0.000 description 2
- NOESYZHRGYRDHS-UHFFFAOYSA-N insulin Chemical compound N1C(=O)C(NC(=O)C(CCC(N)=O)NC(=O)C(CCC(O)=O)NC(=O)C(C(C)C)NC(=O)C(NC(=O)CN)C(C)CC)CSSCC(C(NC(CO)C(=O)NC(CC(C)C)C(=O)NC(CC=2C=CC(O)=CC=2)C(=O)NC(CCC(N)=O)C(=O)NC(CC(C)C)C(=O)NC(CCC(O)=O)C(=O)NC(CC(N)=O)C(=O)NC(CC=2C=CC(O)=CC=2)C(=O)NC(CSSCC(NC(=O)C(C(C)C)NC(=O)C(CC(C)C)NC(=O)C(CC=2C=CC(O)=CC=2)NC(=O)C(CC(C)C)NC(=O)C(C)NC(=O)C(CCC(O)=O)NC(=O)C(C(C)C)NC(=O)C(CC(C)C)NC(=O)C(CC=2NC=NC=2)NC(=O)C(CO)NC(=O)CNC2=O)C(=O)NCC(=O)NC(CCC(O)=O)C(=O)NC(CCCNC(N)=N)C(=O)NCC(=O)NC(CC=3C=CC=CC=3)C(=O)NC(CC=3C=CC=CC=3)C(=O)NC(CC=3C=CC(O)=CC=3)C(=O)NC(C(C)O)C(=O)N3C(CCC3)C(=O)NC(CCCCN)C(=O)NC(C)C(O)=O)C(=O)NC(CC(N)=O)C(O)=O)=O)NC(=O)C(C(C)CC)NC(=O)C(CO)NC(=O)C(C(C)O)NC(=O)C1CSSCC2NC(=O)C(CC(C)C)NC(=O)C(NC(=O)C(CCC(N)=O)NC(=O)C(CC(N)=O)NC(=O)C(NC(=O)C(N)CC=1C=CC=CC=1)C(C)C)CC1=CN=CN1 NOESYZHRGYRDHS-UHFFFAOYSA-N 0.000 description 2
- 229960000310 isoleucine Drugs 0.000 description 2
- AGPKZVBTJJNPAG-UHFFFAOYSA-N isoleucine Natural products CCC(C)C(N)C(O)=O AGPKZVBTJJNPAG-UHFFFAOYSA-N 0.000 description 2
- 239000007788 liquid Substances 0.000 description 2
- 238000002156 mixing Methods 0.000 description 2
- 230000004220 muscle function Effects 0.000 description 2
- 230000012042 muscle hypertrophy Effects 0.000 description 2
- 235000016709 nutrition Nutrition 0.000 description 2
- COLNVLDHVKWLRT-UHFFFAOYSA-N phenylalanine Natural products OC(=O)C(N)CC1=CC=CC=C1 COLNVLDHVKWLRT-UHFFFAOYSA-N 0.000 description 2
- 229960005190 phenylalanine Drugs 0.000 description 2
- 230000037081 physical activity Effects 0.000 description 2
- 239000000843 powder Substances 0.000 description 2
- 238000001556 precipitation Methods 0.000 description 2
- 239000003755 preservative agent Substances 0.000 description 2
- 239000002994 raw material Substances 0.000 description 2
- 230000019491 signal transduction Effects 0.000 description 2
- 239000007787 solid Substances 0.000 description 2
- 239000002904 solvent Substances 0.000 description 2
- 239000003381 stabilizer Substances 0.000 description 2
- 239000006228 supernatant Substances 0.000 description 2
- 239000013589 supplement Substances 0.000 description 2
- 239000006188 syrup Substances 0.000 description 2
- 235000020357 syrup Nutrition 0.000 description 2
- 239000003826 tablet Substances 0.000 description 2
- 238000012360 testing method Methods 0.000 description 2
- 238000012549 training Methods 0.000 description 2
- 238000013519 translation Methods 0.000 description 2
- 238000011282 treatment Methods 0.000 description 2
- 239000004474 valine Substances 0.000 description 2
- 229960004295 valine Drugs 0.000 description 2
- 238000005406 washing Methods 0.000 description 2
- XLYOFNOQVPJJNP-UHFFFAOYSA-N water Substances O XLYOFNOQVPJJNP-UHFFFAOYSA-N 0.000 description 2
- 235000021119 whey protein Nutrition 0.000 description 2
- 102000011690 Adiponectin Human genes 0.000 description 1
- 108010076365 Adiponectin Proteins 0.000 description 1
- 239000004475 Arginine Substances 0.000 description 1
- 206010003694 Atrophy Diseases 0.000 description 1
- 238000009020 BCA Protein Assay Kit Methods 0.000 description 1
- YDNKGFDKKRUKPY-JHOUSYSJSA-N C16 ceramide Natural products CCCCCCCCCCCCCCCC(=O)N[C@@H](CO)[C@H](O)C=CCCCCCCCCCCCCC YDNKGFDKKRUKPY-JHOUSYSJSA-N 0.000 description 1
- 101100361281 Caenorhabditis elegans rpm-1 gene Proteins 0.000 description 1
- 241000282472 Canis lupus familiaris Species 0.000 description 1
- 101000678286 Danio rerio Eukaryotic translation initiation factor 4E-binding protein 3-like Proteins 0.000 description 1
- 101000800913 Dictyostelium discoideum Eukaryotic translation initiation factor 4E-1A-binding protein homolog Proteins 0.000 description 1
- 101000800906 Drosophila melanogaster Eukaryotic translation initiation factor 4E-binding protein Proteins 0.000 description 1
- 241000282326 Felis catus Species 0.000 description 1
- 239000004606 Fillers/Extenders Substances 0.000 description 1
- JZNWSCPGTDBMEW-UHFFFAOYSA-N Glycerophosphorylethanolamin Natural products NCCOP(O)(=O)OCC(O)CO JZNWSCPGTDBMEW-UHFFFAOYSA-N 0.000 description 1
- 108090000288 Glycoproteins Proteins 0.000 description 1
- 102000003886 Glycoproteins Human genes 0.000 description 1
- 101001059454 Homo sapiens Serine/threonine-protein kinase MARK2 Proteins 0.000 description 1
- 102000004877 Insulin Human genes 0.000 description 1
- 108090001061 Insulin Proteins 0.000 description 1
- 206010022489 Insulin Resistance Diseases 0.000 description 1
- 108090000723 Insulin-Like Growth Factor I Proteins 0.000 description 1
- 102000004218 Insulin-Like Growth Factor I Human genes 0.000 description 1
- AHLPHDHHMVZTML-BYPYZUCNSA-N L-Ornithine Chemical compound NCCC[C@H](N)C(O)=O AHLPHDHHMVZTML-BYPYZUCNSA-N 0.000 description 1
- ODKSFYDXXFIFQN-BYPYZUCNSA-P L-argininium(2+) Chemical compound NC(=[NH2+])NCCC[C@H]([NH3+])C(O)=O ODKSFYDXXFIFQN-BYPYZUCNSA-P 0.000 description 1
- HNDVDQJCIGZPNO-YFKPBYRVSA-N L-histidine Chemical compound OC(=O)[C@@H](N)CC1=CN=CN1 HNDVDQJCIGZPNO-YFKPBYRVSA-N 0.000 description 1
- FFEARJCKVFRZRR-BYPYZUCNSA-N L-methionine Chemical compound CSCC[C@H](N)C(O)=O FFEARJCKVFRZRR-BYPYZUCNSA-N 0.000 description 1
- AYFVYJQAPQTCCC-GBXIJSLDSA-N L-threonine Chemical compound C[C@@H](O)[C@H](N)C(O)=O AYFVYJQAPQTCCC-GBXIJSLDSA-N 0.000 description 1
- QIVBCDIJIAJPQS-VIFPVBQESA-N L-tryptophane Chemical compound C1=CC=C2C(C[C@H](N)C(O)=O)=CNC2=C1 QIVBCDIJIAJPQS-VIFPVBQESA-N 0.000 description 1
- 102000004895 Lipoproteins Human genes 0.000 description 1
- 108090001030 Lipoproteins Proteins 0.000 description 1
- 102000014171 Milk Proteins Human genes 0.000 description 1
- 108010011756 Milk Proteins Proteins 0.000 description 1
- 102000015728 Mucins Human genes 0.000 description 1
- 108010063954 Mucins Proteins 0.000 description 1
- 241000699670 Mus sp. Species 0.000 description 1
- 208000029549 Muscle injury Diseases 0.000 description 1
- 208000000112 Myalgia Diseases 0.000 description 1
- SQVRNKJHWKZAKO-PFQGKNLYSA-N N-acetyl-beta-neuraminic acid Chemical compound CC(=O)N[C@@H]1[C@@H](O)C[C@@](O)(C(O)=O)O[C@H]1[C@H](O)[C@H](O)CO SQVRNKJHWKZAKO-PFQGKNLYSA-N 0.000 description 1
- CRJGESKKUOMBCT-VQTJNVASSA-N N-acetylsphinganine Chemical compound CCCCCCCCCCCCCCC[C@@H](O)[C@H](CO)NC(C)=O CRJGESKKUOMBCT-VQTJNVASSA-N 0.000 description 1
- 208000008589 Obesity Diseases 0.000 description 1
- AHLPHDHHMVZTML-UHFFFAOYSA-N Orn-delta-NH2 Natural products NCCCC(N)C(O)=O AHLPHDHHMVZTML-UHFFFAOYSA-N 0.000 description 1
- UTJLXEIPEHZYQJ-UHFFFAOYSA-N Ornithine Natural products OC(=O)C(C)CCCN UTJLXEIPEHZYQJ-UHFFFAOYSA-N 0.000 description 1
- 241000283973 Oryctolagus cuniculus Species 0.000 description 1
- 208000001132 Osteoporosis Diseases 0.000 description 1
- 239000002033 PVDF binder Substances 0.000 description 1
- 229940124158 Protease/peptidase inhibitor Drugs 0.000 description 1
- 108091008611 Protein Kinase B Proteins 0.000 description 1
- 102100033810 RAC-alpha serine/threonine-protein kinase Human genes 0.000 description 1
- 208000034189 Sclerosis Diseases 0.000 description 1
- 102100028904 Serine/threonine-protein kinase MARK2 Human genes 0.000 description 1
- 102000012010 Sialomucins Human genes 0.000 description 1
- 108010061228 Sialomucins Proteins 0.000 description 1
- 238000000692 Student's t-test Methods 0.000 description 1
- AYFVYJQAPQTCCC-UHFFFAOYSA-N Threonine Natural products CC(O)C(N)C(O)=O AYFVYJQAPQTCCC-UHFFFAOYSA-N 0.000 description 1
- 239000004473 Threonine Substances 0.000 description 1
- QIVBCDIJIAJPQS-UHFFFAOYSA-N Tryptophan Natural products C1=CC=C2C(CC(N)C(O)=O)=CNC2=C1 QIVBCDIJIAJPQS-UHFFFAOYSA-N 0.000 description 1
- 208000027418 Wounds and injury Diseases 0.000 description 1
- 238000010521 absorption reaction Methods 0.000 description 1
- 230000002378 acidificating effect Effects 0.000 description 1
- 230000009471 action Effects 0.000 description 1
- 239000013543 active substance Substances 0.000 description 1
- 230000001919 adrenal effect Effects 0.000 description 1
- VREFGVBLTWBCJP-UHFFFAOYSA-N alprazolam Chemical compound C12=CC(Cl)=CC=C2N2C(C)=NN=C2CN=C1C1=CC=CC=C1 VREFGVBLTWBCJP-UHFFFAOYSA-N 0.000 description 1
- BFNBIHQBYMNNAN-UHFFFAOYSA-N ammonium sulfate Chemical compound N.N.OS(O)(=O)=O BFNBIHQBYMNNAN-UHFFFAOYSA-N 0.000 description 1
- 229910052921 ammonium sulfate Inorganic materials 0.000 description 1
- 235000011130 ammonium sulphate Nutrition 0.000 description 1
- 238000004458 analytical method Methods 0.000 description 1
- 239000003963 antioxidant agent Substances 0.000 description 1
- ODKSFYDXXFIFQN-UHFFFAOYSA-N arginine Natural products OC(=O)C(N)CCCNC(N)=N ODKSFYDXXFIFQN-UHFFFAOYSA-N 0.000 description 1
- 239000011230 binding agent Substances 0.000 description 1
- 230000015572 biosynthetic process Effects 0.000 description 1
- 235000015895 biscuits Nutrition 0.000 description 1
- 210000004204 blood vessel Anatomy 0.000 description 1
- 150000005693 branched-chain amino acids Chemical class 0.000 description 1
- 235000008429 bread Nutrition 0.000 description 1
- 238000009395 breeding Methods 0.000 description 1
- 230000001488 breeding effect Effects 0.000 description 1
- 239000000872 buffer Substances 0.000 description 1
- 239000006172 buffering agent Substances 0.000 description 1
- 235000014171 carbonated beverage Nutrition 0.000 description 1
- 230000015556 catabolic process Effects 0.000 description 1
- 230000006037 cell lysis Effects 0.000 description 1
- 230000005754 cellular signaling Effects 0.000 description 1
- 229940106189 ceramide Drugs 0.000 description 1
- ZVEQCJWYRWKARO-UHFFFAOYSA-N ceramide Natural products CCCCCCCCCCCCCCC(O)C(=O)NC(CO)C(O)C=CCCC=C(C)CCCCCCCCC ZVEQCJWYRWKARO-UHFFFAOYSA-N 0.000 description 1
- 235000013339 cereals Nutrition 0.000 description 1
- 235000013353 coffee beverage Nutrition 0.000 description 1
- 239000003086 colorant Substances 0.000 description 1
- 238000007796 conventional method Methods 0.000 description 1
- 239000006071 cream Substances 0.000 description 1
- 230000006378 damage Effects 0.000 description 1
- 230000003247 decreasing effect Effects 0.000 description 1
- 238000001514 detection method Methods 0.000 description 1
- 238000003745 diagnosis Methods 0.000 description 1
- 238000000502 dialysis Methods 0.000 description 1
- 235000005911 diet Nutrition 0.000 description 1
- 230000000378 dietary effect Effects 0.000 description 1
- 235000015872 dietary supplement Nutrition 0.000 description 1
- 230000004069 differentiation Effects 0.000 description 1
- 239000003085 diluting agent Substances 0.000 description 1
- 239000007884 disintegrant Substances 0.000 description 1
- 239000002270 dispersing agent Substances 0.000 description 1
- 230000035622 drinking Effects 0.000 description 1
- 239000003995 emulsifying agent Substances 0.000 description 1
- 238000005516 engineering process Methods 0.000 description 1
- 239000000835 fiber Substances 0.000 description 1
- 238000001914 filtration Methods 0.000 description 1
- 235000015203 fruit juice Nutrition 0.000 description 1
- 235000011389 fruit/vegetable juice Nutrition 0.000 description 1
- 235000013376 functional food Nutrition 0.000 description 1
- 150000002270 gangliosides Chemical class 0.000 description 1
- 239000000499 gel Substances 0.000 description 1
- 238000002523 gelfiltration Methods 0.000 description 1
- 150000002305 glucosylceramides Chemical class 0.000 description 1
- 235000020251 goat milk Nutrition 0.000 description 1
- 239000008187 granular material Substances 0.000 description 1
- 239000003102 growth factor Substances 0.000 description 1
- 239000000122 growth hormone Substances 0.000 description 1
- HNDVDQJCIGZPNO-UHFFFAOYSA-N histidine Natural products OC(=O)C(N)CC1=CN=CN1 HNDVDQJCIGZPNO-UHFFFAOYSA-N 0.000 description 1
- 239000003906 humectant Substances 0.000 description 1
- 238000003119 immunoblot Methods 0.000 description 1
- 238000001727 in vivo Methods 0.000 description 1
- 238000002347 injection Methods 0.000 description 1
- 239000007924 injection Substances 0.000 description 1
- 208000014674 injury Diseases 0.000 description 1
- 229940125396 insulin Drugs 0.000 description 1
- 238000004255 ion exchange chromatography Methods 0.000 description 1
- 235000015110 jellies Nutrition 0.000 description 1
- 239000008274 jelly Substances 0.000 description 1
- 201000010260 leiomyoma Diseases 0.000 description 1
- 239000000314 lubricant Substances 0.000 description 1
- 210000005075 mammary gland Anatomy 0.000 description 1
- 238000004519 manufacturing process Methods 0.000 description 1
- 208000030159 metabolic disease Diseases 0.000 description 1
- 229930182817 methionine Natural products 0.000 description 1
- 238000001471 micro-filtration Methods 0.000 description 1
- 230000005486 microgravity Effects 0.000 description 1
- 235000021239 milk protein Nutrition 0.000 description 1
- 210000000663 muscle cell Anatomy 0.000 description 1
- 208000013465 muscle pain Diseases 0.000 description 1
- 210000001087 myotubule Anatomy 0.000 description 1
- 210000000653 nervous system Anatomy 0.000 description 1
- VVGIYYKRAMHVLU-UHFFFAOYSA-N newbouldiamide Natural products CCCCCCCCCCCCCCCCCCCC(O)C(O)C(O)C(CO)NC(=O)CCCCCCCCCCCCCCCCC VVGIYYKRAMHVLU-UHFFFAOYSA-N 0.000 description 1
- 235000012149 noodles Nutrition 0.000 description 1
- 235000015097 nutrients Nutrition 0.000 description 1
- 230000035764 nutrition Effects 0.000 description 1
- 230000000050 nutritive effect Effects 0.000 description 1
- 235000020824 obesity Nutrition 0.000 description 1
- 210000000056 organ Anatomy 0.000 description 1
- 239000003960 organic solvent Substances 0.000 description 1
- 230000008520 organization Effects 0.000 description 1
- 229960003104 ornithine Drugs 0.000 description 1
- 238000007911 parenteral administration Methods 0.000 description 1
- 239000000137 peptide hydrolase inhibitor Substances 0.000 description 1
- 239000000546 pharmaceutical excipient Substances 0.000 description 1
- WTJKGGKOPKCXLL-RRHRGVEJSA-N phosphatidylcholine Chemical compound CCCCCCCCCCCCCCCC(=O)OC[C@H](COP([O-])(=O)OCC[N+](C)(C)C)OC(=O)CCCCCCCC=CCCCCCCCC WTJKGGKOPKCXLL-RRHRGVEJSA-N 0.000 description 1
- 150000008104 phosphatidylethanolamines Chemical class 0.000 description 1
- 230000035790 physiological processes and functions Effects 0.000 description 1
- 229920002981 polyvinylidene fluoride Polymers 0.000 description 1
- 230000002335 preservative effect Effects 0.000 description 1
- 230000000750 progressive effect Effects 0.000 description 1
- 230000017854 proteolysis Effects 0.000 description 1
- 235000020185 raw untreated milk Nutrition 0.000 description 1
- 230000009467 reduction Effects 0.000 description 1
- 230000001105 regulatory effect Effects 0.000 description 1
- 230000002040 relaxant effect Effects 0.000 description 1
- 230000004044 response Effects 0.000 description 1
- 230000000284 resting effect Effects 0.000 description 1
- 208000001076 sarcopenia Diseases 0.000 description 1
- 235000013580 sausages Nutrition 0.000 description 1
- 230000028327 secretion Effects 0.000 description 1
- 238000000926 separation method Methods 0.000 description 1
- 235000020183 skimmed milk Nutrition 0.000 description 1
- 210000003875 slow muscle fiber Anatomy 0.000 description 1
- 238000002415 sodium dodecyl sulfate polyacrylamide gel electrophoresis Methods 0.000 description 1
- 235000014214 soft drink Nutrition 0.000 description 1
- 239000000243 solution Substances 0.000 description 1
- 238000000638 solvent extraction Methods 0.000 description 1
- 230000000087 stabilizing effect Effects 0.000 description 1
- 150000003431 steroids Chemical class 0.000 description 1
- 210000002784 stomach Anatomy 0.000 description 1
- 239000000829 suppository Substances 0.000 description 1
- 238000001356 surgical procedure Methods 0.000 description 1
- 208000024891 symptom Diseases 0.000 description 1
- 230000002195 synergetic effect Effects 0.000 description 1
- 238000003786 synthesis reaction Methods 0.000 description 1
- 235000013616 tea Nutrition 0.000 description 1
- 238000002560 therapeutic procedure Methods 0.000 description 1
- 239000002562 thickening agent Substances 0.000 description 1
- 210000001519 tissue Anatomy 0.000 description 1
- 150000003626 triacylglycerols Chemical class 0.000 description 1
- 208000001072 type 2 diabetes mellitus Diseases 0.000 description 1
- 235000012431 wafers Nutrition 0.000 description 1
Images
Landscapes
- Coloring Foods And Improving Nutritive Qualities (AREA)
- Medicines Containing Material From Animals Or Micro-Organisms (AREA)
- Acyclic And Carbocyclic Compounds In Medicinal Compositions (AREA)
Abstract
Description
本発明は、筋量の増加或いは低下抑制効果を発揮する医薬品、食品等に関する。 The present invention relates to pharmaceuticals, foods, and the like that exert an effect of suppressing increase or decrease in muscle mass.
運動やスポーツ競技において、骨格筋は最も重要な運動器である。我々は骨格筋を収縮又は弛緩させることにより身体活動を行う。また骨格筋は活動するためのエネルギー源として、血液中の糖や脂肪を取り込んで消費することから、最大のエネルギー消費組織でもある。その他にも骨格筋は関節の安定化や姿勢の保持、血管、臓器の保護等の役割を有している。 Skeletal muscle is the most important exercise device in exercise and sports competition. We do physical activity by contracting or relaxing skeletal muscle. Skeletal muscle is also the largest energy consuming organization because it takes in and consumes sugar and fat in the blood as an energy source for activity. In addition, skeletal muscle plays a role of stabilizing joints, maintaining posture, protecting blood vessels and organs, and the like.
骨格筋量は、筋蛋白質の合成と分解のバランスにより一定量に保たれており、通常の生活の中で骨格筋量が急激に変動することは考えられない。しかし、加齢に伴い骨格筋量や筋力等の筋機能は低下してくる。我々の骨格筋は、30歳を過ぎると10年毎に約5%前後の割合で減少し、60歳を過ぎるとその減少率は更に加速すること、また高齢期の骨格筋の減少には、速筋(タイプII)繊維の選択的な減少が特徴であると報告されている(非特許文献1)。加齢に伴う骨格筋量や筋機能の低下は、転倒による怪我を引き起こし、身体活動量の低下を引き起こし、それに伴い骨粗鬆症、肥満やインスリン抵抗性等の代謝障害の危険性を高めると言われている(非特許文献2)。 Skeletal muscle mass is maintained at a constant level due to the balance between muscle protein synthesis and degradation, and skeletal muscle mass cannot be expected to fluctuate rapidly during normal life. However, muscle functions such as skeletal muscle mass and muscle strength decrease with age. Our skeletal muscles decrease at a rate of about 5% every 10 years after the age of 30, and the rate of reduction further accelerates after the age of 60. It has been reported that a selective decrease in fast muscle (type II) fibers is characteristic (Non-patent Document 1). It is said that the decrease in skeletal muscle mass and muscle function associated with aging causes injury due to falls, resulting in a decrease in physical activity, and accordingly increases the risk of metabolic disorders such as osteoporosis, obesity and insulin resistance. (Non-Patent Document 2).
生体内では、骨格筋量の増加は、タンパク質合成の増加につながるシグナル伝達系を通じて行われ、その中心的な因子としてmTOR(mammalian target of rapamycin)がある。インスリンやIGF−1などの増殖因子に応答して、ホスファチジルイノシトール3キナーゼ(PI3K)を介し、シグナル伝達経路下流のAKTがリン酸化され、mTORがリン酸化される。リン酸化されたmTORはさらに下流の、4E−BPやp70S6キナーゼ(S6K)をリン酸化する。p70S6キナーゼ(S6K)は、セリン/スレオニンキナーゼであり、リン酸化され活性化されると、翻訳を促進するS6リボソームタンパク質がリン酸化され活性化されることで、筋組織においては筋タンパク質の合成が促進される。従って、筋組織において上記シグナル系が活性化すれば骨格筋量が増加すると考えられる。 In vivo, skeletal muscle mass is increased through a signal transduction system that leads to an increase in protein synthesis, and mTOR (mammalian target of rapamycin) is a central factor. In response to growth factors such as insulin and IGF-1, AKT downstream of the signal transduction pathway is phosphorylated and mTOR is phosphorylated through phosphatidylinositol 3 kinase (PI3K). Phosphorylated mTOR further phosphorylates downstream 4E-BP and p70S6 kinase (S6K). p70S6 kinase (S6K) is a serine / threonine kinase. When phosphorylated and activated, the S6 ribosomal protein that promotes translation is phosphorylated and activated, so that muscle protein synthesis can be performed in muscle tissues. Promoted. Therefore, it is considered that the amount of skeletal muscle increases if the signal system is activated in muscle tissue.
これまで、骨格筋量を増加するための手段として、ステロイド剤や成長ホルモンの使用が知られているが、これらは筋力の低下や筋肉痛、副腎機能の低下等の副作用の問題を有している。骨格筋量を増加或いは維持することは、パフォーマンス向上を目指す運動愛好者やアスリートだけでなく、ヘルスケアの点で広く一般人においても有用であると考えられることから、より安全性の高い成分が求められ、例えば、アルギニン、リジン及びオルニチンを含むサプリメント(特許文献1)、L−ロイシンを一定量含有する高齢者の骨格筋量減少を防止又は改善するためのアミノ酸含有組成物(特許文献2)等が報告されている。 Until now, the use of steroids and growth hormones has been known as a means to increase skeletal muscle mass, but these have problems of side effects such as muscle weakness, muscle pain, and decreased adrenal function. Yes. Increasing or maintaining skeletal muscle mass is considered to be useful not only for exercise enthusiasts and athletes aiming at improving performance, but also for general people in terms of health care. For example, a supplement containing arginine, lysine and ornithine (Patent Document 1), an amino acid-containing composition for preventing or improving a decrease in skeletal muscle mass in elderly people containing a certain amount of L-leucine (Patent Document 2), etc. Has been reported.
一方、乳脂肪球皮膜(MFGM: milk fat globule membrane)は、乳中の脂肪球を皮膜している乳腺細胞由来の膜様成分であり、主としてリン脂質とリポタンパク質よりなる。乳脂肪球皮膜は、脂肪を乳汁中に分散させる機能があるが、それのみならず血中アディポネクチン増加及び/又は減少抑制効果(特許文献3)、学習能向上効果(特許文献4)、シアロムチンの分泌促進効果等(特許文献5)の生理機能を有することが知られている。また、最近では、乳脂肪球皮膜が、筋力を増大させることや持久力を向上させることが報告されている(特許文献6)。 On the other hand, milk fat globule membrane (MFGM) is a membrane-like component derived from mammary gland cells that coat fat globules in milk, and mainly consists of phospholipids and lipoproteins. The milk fat globule membrane has a function of dispersing fat in milk, but it also has an effect of suppressing increase and / or decrease of blood adiponectin (Patent Document 3), an effect of improving learning ability (Patent Document 4), and sialomucin. It is known to have physiological functions such as a secretion promoting effect (Patent Document 5). Recently, it has been reported that the milk fat globule membrane increases muscle strength and improves endurance (Patent Document 6).
しかしながら、乳脂肪球皮膜に筋量増加作用があること、更にこれとアミノ酸を併用して用いることにより、それぞれを単独で用いるよりも格段に筋タンパク質合成シグナルが増強され、筋量増加作用或いは筋萎縮抑制作用が大きくなることはこれまで全く知られていない。 However, the milk fat globule membrane has an effect of increasing muscle mass, and when used in combination with an amino acid, the muscle protein synthesis signal is remarkably enhanced as compared with the case where each of them is used alone. Until now, it has not been known at all that an atrophy-suppressing effect is increased.
本発明は、優れた筋タンパク質合成シグナルの増強作用を有し、筋肉量増加効果、筋萎縮抑制効果を発揮し、且つ安全性が高い医薬品、医薬部外品、食品、飼料及び医薬品や食品等に配合した場合に当該効果を発揮する素材を提供することに関する。 The present invention has an excellent muscle protein synthesis signal enhancing action, exerts an effect of increasing muscle mass, suppresses muscle atrophy, and has high safety, such as pharmaceuticals, quasi drugs, foods, feeds, pharmaceuticals and foods, etc. It is related with providing the raw material which exhibits the said effect, when mix | blending with.
本発明者らは、筋量増加において有効な成分の探索を行った結果、乳脂肪球皮膜とアミノ酸を併用して用いれば、各々を単独で用いる場合に比較して格段に優れた筋タンパク質合成シグナル増強作用があり、これが筋量増加、筋萎縮抑制等の効果を発揮し得る医薬品、医薬部外品、食品及び飼料等として有用であることを見出した。 As a result of searching for an effective component in increasing muscle mass, the present inventors have found that muscle protein synthesis is much better when milk fat globule membrane and amino acids are used in combination than when each is used alone. It has been found that it has a signal enhancing action, which is useful as a pharmaceutical, quasi-drug, food and feed that can exert effects such as an increase in muscle mass and suppression of muscle atrophy.
すなわち、本発明は、下記(1)〜(3)に係るものである。
(1)乳脂肪球皮膜とアミノ酸を組み合わせてなる筋タンパク質合成シグナル増強剤。
(2)乳脂肪球皮膜とアミノ酸を組み合わせてなる筋量増加剤。
(3)乳脂肪球皮膜とアミノ酸を組み合わせてなる筋萎縮抑制剤。
That is, the present invention relates to the following (1) to (3).
(1) A muscle protein synthesis signal enhancer comprising a milk fat globule membrane and an amino acid in combination.
(2) A muscle mass increasing agent comprising a combination of milk fat globule membrane and amino acids.
(3) A muscle atrophy inhibitor comprising a combination of milk fat globule membrane and amino acids.
本発明の筋タンパク質合成シグナル増強剤、筋量増加剤及び筋萎縮抑制剤は、高齢者を含む幅広い年齢層において、日常の活動時における筋量増加、筋萎縮抑制のための食品、医薬品、医薬部外品、飼料として又は医薬品や食品等に配合した場合に当該効果を発揮する素材として有用である。 The muscle protein synthesis signal enhancer, muscle mass increasing agent, and muscle atrophy inhibitor of the present invention are foods, pharmaceuticals, and pharmaceuticals for increasing muscle mass and suppressing muscle atrophy during daily activities in a wide range of age groups including the elderly. It is useful as a quasi-drug, feed, or a material that exhibits this effect when blended with pharmaceuticals or foods.
本発明における乳脂肪球皮膜は、乳中の脂肪球を皮膜する膜及びそれを構成する膜成分混合物と定義され、バターミルクやバターセーラム等の乳複合脂質高含有画分に多く含まれることが知られている。一般的に、乳脂肪球皮膜はタンパク質(約40〜45%)と脂質(約50〜55%)から構成されており、当該タンパク質としては、ミルクムチンと呼ばれる糖蛋白質(Mather IH、Biochim Biophys Acta.(1978) 514:25-36.)等を含むことが知られ、脂質としては、トリグリセライドやリン脂質(例えば、スフィンゴリン脂質、グリセロリン脂質等)が多く含まれ、これ以外にスフィンゴ糖脂質(例えば、グルコシルセラミド、ガングリオシド等)が含まれることが知られている(Keenan TW、Applied Science Publishers.(1983) pp89-pp130.)。また、リン脂質は、スフィンゴミエリン等のスフィンゴリン脂質が主であり、その他にホスファチジルコリンやホスファチジルエタノールアミン等のグリセロリン脂質が含まれる。 The milk fat globule membrane in the present invention is defined as a membrane that coats fat globules in milk and a membrane component mixture that constitutes the membrane, and it is often contained in fractions containing high amounts of milk complex lipids such as butter milk and butter serum. Are known. In general, milk fat globule membrane is composed of protein (about 40 to 45%) and lipid (about 50 to 55%). As the protein, glycoprotein called milk mucin (Mather IH, Biochim Biophys Acta. (1978) 514: 25-36.) And the like, and as the lipid, there are many triglycerides and phospholipids (for example, sphingophospholipid, glycerophospholipid, etc.), and in addition to this, sphingoglycolipids (for example, , Glucosylceramide, ganglioside, etc.) (Keenan TW, Applied Science Publishers. (1983) pp89-pp130.). The phospholipids are mainly sphingophospholipids such as sphingomyelin, and glycerophospholipids such as phosphatidylcholine and phosphatidylethanolamine are also included.
本発明の乳脂肪球皮膜としては、脂質の含有量が、乾燥物換算で、10質量%以上、好ましくは20質量%以上、より好ましくは30質量%以上であり、且つ100質量%以下、好ましくは90質量%以下、より好ましくは60質量%以下である。また、10〜100質量%、好ましくは20〜90質量%、より好ましくは30〜60質量%である。また、リン脂質の含有量が、乾燥物換算で、5質量%以上、好ましくは8質量%以上、より好ましくは15質量%以上、より好ましくは20質量%以上であり、且つ100質量%以下、好ましくは90質量%以下、より好ましくは80質量%以下、より好ましくは60質量%以下である。また、5〜100質量%、より好ましくは8〜90質量%、より好ましくは15〜80質量%、より好ましくは20〜60質量%である。 The milk fat globule membrane of the present invention has a lipid content of 10% by mass or more, preferably 20% by mass or more, more preferably 30% by mass or more, and 100% by mass or less, preferably in terms of dry matter. Is 90% by mass or less, more preferably 60% by mass or less. Moreover, it is 10-100 mass%, Preferably it is 20-90 mass%, More preferably, it is 30-60 mass%. The phospholipid content is 5% by mass or more, preferably 8% by mass or more, more preferably 15% by mass or more, more preferably 20% by mass or more, and 100% by mass or less, in terms of dry matter. Preferably it is 90 mass% or less, More preferably, it is 80 mass% or less, More preferably, it is 60 mass% or less. Moreover, it is 5-100 mass%, More preferably, it is 8-90 mass%, More preferably, it is 15-80 mass%, More preferably, it is 20-60 mass%.
本発明の乳脂肪球皮膜は、乳原料から遠心分離法や有機溶剤抽出法等の公知の方法により調製することができる。 The milk fat globule membrane of the present invention can be prepared from a milk raw material by a known method such as a centrifugal separation method or an organic solvent extraction method.
乳原料としては、牛乳やヤギ乳等が挙げられるが、牛乳を用いるのが好ましい。また、乳原料には、生乳、脱脂乳や加工乳等の乳の他、乳製品も含まれ、乳製品としては、バターミルク、バターオイル、バターセーラム、ホエータンパク質濃縮物(WPC)等が挙げられる。バターミルクは、牛乳等を遠心分離して得られるクリームからバター粒を製造する際に得られるが、乳脂肪球皮膜はバターミルク中に多く含まれているので、バターミルクをそのまま使用することができる。同様に、バターオイルを製造する際に生じるバターセーラム中にも乳脂肪球皮膜が多く含まれるのでバターセーラムをそのまま使用してもよい。 Examples of the milk material include milk and goat milk, but it is preferable to use milk. In addition to milk such as raw milk, skim milk and processed milk, dairy products include dairy products. Examples of dairy products include buttermilk, butter oil, buttersarum, whey protein concentrate (WPC) and the like. It is done. Buttermilk is obtained when producing butter grains from cream obtained by centrifuging milk etc., but since milk fat globule membrane is contained in a lot of buttermilk, buttermilk can be used as it is. it can. Similarly, since a lot of milk fat globule membrane is contained in the butter serum produced when producing the butter oil, the butter serum may be used as it is.
乳脂肪球皮膜の調製法としては、例えば、乳やホエータンパク質濃縮物(WPC)等の乳製品をエーテルやアセトンで抽出する方法(特開平3−47192号公報)、バターミルクを酸性域に調整、等電点沈殿を行うことにより生じたタンパク質を除去し、上清を精密濾過膜処理して得られる濃縮液を乾燥する方法(特許第3103218号公報)等が挙げられる。また、バターセーラム中よりタンパク質を凝集除去後に濾過濃縮し乾燥する方法(特開2007−89535号公報)等も使用することができる。本製法によると、例えば、乳由来の複合脂質を乾燥物中20質量%以上含有する乳脂肪球皮膜を調製することができる。 As a method for preparing a milk fat globule membrane, for example, a method of extracting milk products such as milk and whey protein concentrate (WPC) with ether or acetone (JP-A-3-47192), butter milk is adjusted to an acidic range And a method of removing a protein produced by performing isoelectric precipitation and drying a concentrated solution obtained by subjecting the supernatant to a microfiltration membrane treatment (Japanese Patent No. 3103218). Further, a method of filtering and concentrating the protein after removing the protein from the buttersarum and drying it (Japanese Patent Application Laid-Open No. 2007-89535) can also be used. According to this production method, for example, a milk fat globule membrane containing 20% by mass or more of milk-derived complex lipid in the dried product can be prepared.
斯くして調製された乳脂肪球皮膜は、必要に応じて、さらに透析、硫安分画、ゲルろ過、等電点沈殿、イオン交換クロマトグラフィー、溶媒分画等の手法により精製することにより純度を高めたものを使用してもよい。 The milk fat globule membrane thus prepared is further purified by a method such as dialysis, ammonium sulfate fractionation, gel filtration, isoelectric precipitation, ion exchange chromatography, solvent fractionation, etc. as necessary. You may use what was raised.
なお、乳脂肪球皮膜の形態は、特に限定されず、液状、半固体状や個体状、粉状等の何れでもよく、これらを単独で又は2種以上組み合わせて用いてもよい。
牛乳由来の乳脂肪球皮膜は、食経験が豊富であり、高純度かつ安価なものも市販されており、それらを用いるのが特に好ましい。例えば、メグレジャパン(株)「BSCP」、雪印乳業(株)「ミルクセラミドMC−5」、(株)ニュージーランドミルクプロダクツ「Phospholipid Concentrate」等が挙げられる。
In addition, the form of the milk fat globule membrane is not particularly limited, and any of liquid, semi-solid, solid, and powder may be used, and these may be used alone or in combination of two or more.
Milk fat globule membranes derived from milk have abundant dietary experience, and high purity and inexpensive products are also commercially available, and it is particularly preferable to use them. Examples include Megre Japan Co., Ltd. “BSCP”, Snow Brand Milk Products Co., Ltd. “Milk Ceramide MC-5”, and New Zealand Milk Products “Phospholipid Concentrate”.
一方、本発明のアミノ酸としては、必須アミノ酸、すなわち、ヒスチジン、イソロイシン、ロイシン、リジン、メチオニン、フェニルアラニン、スレオニン、バリン、トリプトファンから選ばれる1種以上が好ましい。このうち、筋肉量増加の点から、ロイシン、イソロイシン、バリン、フェニルアラニンのような疎水性アミノ酸が好ましく、更にはロイシン、イソロイシン及びバリンのような分岐鎖アミノ酸が好ましく、ロイシンがより好ましい。尚、当該アミノ酸はL型であるのが好ましいが、D型であってもよい。当該アミノ酸は、市販品をそのまま用いてもよく、また、化学合成したものを用いても良い。 On the other hand, the amino acid of the present invention is preferably an essential amino acid, that is, one or more selected from histidine, isoleucine, leucine, lysine, methionine, phenylalanine, threonine, valine, and tryptophan. Of these, from the viewpoint of increasing muscle mass, hydrophobic amino acids such as leucine, isoleucine, valine and phenylalanine are preferred, branched chain amino acids such as leucine, isoleucine and valine are preferred, and leucine is more preferred. The amino acid is preferably L type, but may be D type. As the amino acid, a commercially available product may be used as it is, or a chemically synthesized product may be used.
本発明の乳脂肪球皮膜とアミノ酸を組み合わせてなる筋タンパク質合成シグナル増強剤、筋量増加剤及び筋萎縮抑制剤(以下、「筋タンパク質合成シグナル増強剤等」とも称する)は、配合剤として、それぞれの有効量を、すなわちそれぞれを有効成分として、適当な配合比において一つの剤型に製剤化したものでも、またそれぞれの有効量を含有する単独の製剤を同時に又は間隔を空けて別々に使用できるようにしたキットであってもよい。 A muscle protein synthesis signal enhancer, a muscle mass increase agent, and a muscle atrophy inhibitor (hereinafter also referred to as “muscle protein synthesis signal enhancer etc.”), which are a combination of the milk fat globule membrane of the present invention and an amino acid, Each effective amount, that is, each formulated as an active ingredient and formulated into one dosage form at an appropriate blending ratio, or a single formulation containing each effective amount is used simultaneously or separately at intervals It may be a kit that can be used.
本発明の筋タンパク質合成シグナル増強剤等における、乳脂肪球皮膜とアミノ酸の組み合わせの割合は、適宜選択することができるが、例えば乳脂肪球皮膜1質量部に対して、アミノ酸は、好ましくは0.05質量部以上、より好ましくは0.2質量部以上であり、且つ好ましくは30質量部以下、より好ましくは10質量部以下である、また、好ましくは0.05〜30質量部、より好ましくは0.2〜10質量部である。 The ratio of the combination of milk fat globule membrane and amino acid in the muscle protein synthesis signal enhancer or the like of the present invention can be appropriately selected. For example, the amino acid is preferably 0 with respect to 1 part by mass of the milk fat globule membrane. 0.05 parts by mass or more, more preferably 0.2 parts by mass or more, and preferably 30 parts by mass or less, more preferably 10 parts by mass or less, and preferably 0.05 to 30 parts by mass, more preferably Is 0.2 to 10 parts by mass.
後記実施例に示すように、乳脂肪球皮膜及びアミノ酸を組み合わせてラットに投与した場合、筋タンパク質合成に関わるp70S6キナーゼ(S6K)がリン酸化される。p70S6キナーゼ(S6K)がリン酸化され活性化されると、翻訳を促進するS6リボソームタンパク質がリン酸化され活性化され、筋組織において筋タンパク質の合成が促進され、骨格筋量が増加することから、乳脂肪球皮膜及びアミノ酸の組み合わせは、筋タンパク質合成シグナル増強剤、筋量増加剤又は筋萎縮抑制剤として使用することができる。ここで、当該使用は、ヒト若しくは非ヒト動物、又はそれらに由来する検体における使用であり得、また治療的使用であっても非治療的使用であってもよい。ここで、「非治療的」とは、医療行為を含まない概念、すなわち人間を手術、治療又は診断する方法を含まない概念、より具体的には医師又は医師の指示を受けた者が人間に対して手術、治療又は診断を実施する方法を含まない概念である。
また、乳脂肪球皮膜及びアミノ酸の組み合わせは、筋タンパク質合成シグナル増強剤、筋量増加剤又は筋萎縮抑制剤を製造するために使用することができる。
As shown in Examples below, when a combination of milk fat globule membrane and amino acid is administered to rats, p70S6 kinase (S6K) involved in muscle protein synthesis is phosphorylated. When p70S6 kinase (S6K) is phosphorylated and activated, the S6 ribosomal protein that promotes translation is phosphorylated and activated, promoting muscle protein synthesis in muscle tissue and increasing skeletal muscle mass, The combination of milk fat globule membrane and amino acid can be used as a muscle protein synthesis signal enhancer, muscle mass enhancer or muscle atrophy inhibitor. Here, the use can be in humans or non-human animals, or specimens derived therefrom, and can be either therapeutic or non-therapeutic. Here, “non-therapeutic” means a concept that does not include medical practice, that is, a concept that does not include a method for operating, treating, or diagnosing a person, more specifically, a doctor or a person who has received instructions from a doctor It is a concept that does not include a method for performing surgery, treatment, or diagnosis on the subject.
Moreover, the combination of milk fat globule membrane and amino acids can be used to produce a muscle protein synthesis signal enhancer, muscle mass increase agent, or muscle atrophy inhibitor.
したがって、乳脂肪球皮膜及びアミノ酸の組み合わせは、例えば加齢に伴って生ずる筋量の減少や筋萎縮に対して有効である。
本発明において、「筋量増加」とは、筋肉量を増加させること意味し、筋量低下の抑制も含まれる。また、「筋萎縮」とは、筋蛋白質の分解速度が合成速度を上回ることにより、筋蛋白質が減少する、もしくは筋細胞が減少し、結果的に筋量が低下することをいい、長期間の安静臥床や骨折等によるギプス固定、或いは微小重力暴露によるもの(廃用性筋萎縮という)と筋萎縮性側策硬化症(ALS)等の疾病による進行性筋萎縮に大別される。さらに、加齢に伴っても筋萎縮と同様の症状が起きることがあり、これは加齢性筋減弱症(サルコペニア)と呼ばれている。したがって「筋萎縮の抑制」とは、不活動や加齢、疾病等による筋量の低下を抑制することをいう。
Therefore, the combination of milk fat globule membrane and amino acid is effective for, for example, the decrease in muscle mass and muscle atrophy caused by aging.
In the present invention, “increase in muscle mass” means to increase muscle mass, and includes suppression of muscle mass decline. In addition, “muscle atrophy” refers to a decrease in muscle protein or a decrease in muscle cells, resulting in a decrease in muscle mass due to the degradation rate of muscle protein exceeding the synthesis rate. It can be roughly divided into cast fixation due to resting bed and fractures, or microgravity exposure (referred to as disuse muscle atrophy) and progressive muscle atrophy due to diseases such as amyotrophic side sclerosis (ALS). Furthermore, symptoms similar to muscle atrophy may occur with aging, and this is called age-related muscle weakness (sarcopenia). Therefore, “suppression of muscle atrophy” refers to suppression of a decrease in muscle mass due to inactivity, aging, disease, or the like.
一般に筋断面積と筋力との間には相関関係が認められ(福永哲夫:ヒトの絶対筋力−超音波による体肢組成・筋力の分析−杏林書院 1978)、このことは筋肥大が筋力の増加を伴うことを示しているが、トレーニングによる筋力の増加は筋横断面積の増加よりも大きいという報告されている(Davis J et al. Eur J Appl Physiol 57 1988)。また、トレーニングが進むについて、筋線維横断面積あたりの筋力が低下することも示されている(Ploutsz LL et al. J Appl Physiol 76 1994)。このような理由として、筋横断面積以外にも、運動神経系や筋肉に含まれる速筋線維と遅筋線維の割合が筋力に影響するからと考えられる(Komi P et al. Acta Physiol Scand 100 1997、Sale DG et al. Med Sci Sports Exerc 20 1998)。このことから、筋力は様々な因子により調節され、筋肥大の筋力の増加は単なる一対一の関係で結ばれているわけではない。したがって、本発明の筋量増加剤及び筋萎縮抑制剤は、筋力向上剤、持久力向上剤とは異なるものである。 In general, there is a correlation between muscle cross-sectional area and muscle strength (Tetsuo Fukunaga: Human absolute muscle strength-Analysis of limb composition and strength by ultrasound-Kyorin Shoin 1978). This is because muscle hypertrophy increases muscle strength. However, it has been reported that the increase in muscle strength due to training is greater than the increase in cross-muscle area (Davis J et al. Eur J Appl Physiol 57 1988). It has also been shown that muscle strength decreases per muscle fiber cross-sectional area as training progresses (Ploutsz LL et al. J Appl Physiol 76 1994). The reason for this is thought to be that the ratio of fast and slow muscle fibers contained in the motor nervous system and muscles affects muscle strength in addition to the cross-muscle area (Komi P et al. Acta Physiol Scand 100 1997 Sale DG et al. Med Sci Sports Exerc 20 1998). For this reason, muscle strength is regulated by various factors, and the increase in muscle strength due to muscle hypertrophy is not simply linked in a one-to-one relationship. Therefore, the muscle mass increasing agent and the muscle atrophy inhibitor of the present invention are different from the muscle strength improving agent and the endurance improving agent.
本発明の筋タンパク質合成シグナル増強剤等は、それ自体、ヒトを含む動物に摂取又は投与した場合に筋量増加、及び筋萎縮抑制の各効果を発揮する、ヒト若しくは動物用の医薬品、医薬部外品、食品、又は飼料であってもよく、或いは当該医薬品、医薬部外品、食品又は飼料に配合して使用される素材又は製剤であってもよい。 The muscle protein synthesis signal enhancer and the like of the present invention itself is a drug for humans or animals, or a pharmaceutical part that exhibits the effects of increasing muscle mass and suppressing muscle atrophy when ingested or administered to animals including humans. The product may be a quasi-drug, food, or feed, or may be a material or formulation that is used in combination with the drug, quasi-drug, food, or feed.
また、当該食品には、運動不足者や中高年者、ベッドレスト者、或いはアスリートや運動愛好者における筋量増加、速筋増加、筋分化促進、及び筋損傷抑制をコンセプトとし、必要に応じてその旨を表示した食品、機能性食品、病者用食品、特定保健用食品が包含される。 The food also has the concept of increasing muscle mass, increasing fast muscles, promoting muscle differentiation, and suppressing muscle damage in those who are underexercise, middle-aged and elderly, bed resters, athletes and exercise enthusiasts. This includes foods with indications, functional foods, foods for the sick, and foods for specified health use.
本発明の乳脂肪球皮膜及びアミノ酸を含有する上記医薬品の投与形態としては、例えば錠剤、カプセル剤、顆粒剤、散剤、シロップ剤等による経口投与又は注射剤、坐剤、吸入薬、経皮吸収剤、外用剤等による非経口投与が挙げられる。また、このような種々の剤型の製剤は、本発明の乳脂肪球皮膜及びアミノ酸をそれぞれ単独で、又は他の薬学的に許容される賦形剤、結合剤、増量剤、崩壊剤、界面活性剤、滑沢剤、分散剤、緩衝剤、保存剤、嬌味剤、香料、被膜剤、担体、希釈剤、乳脂肪球皮膜以外の薬効成分等を適宜組み合わせて調製することができる。また、これらの投与形態のうち、好ましい形態は経口投与であり、経口用液体製剤は、嬌味剤、緩衝剤、安定化剤等を加えて常法により調製することができる。 Examples of the dosage form of the above-mentioned pharmaceutical containing the milk fat globule membrane and amino acid of the present invention include oral administration by tablet, capsule, granule, powder, syrup, etc., or injection, suppository, inhalant, and transdermal absorption. Parenteral administration by an agent, an external preparation and the like. In addition, the preparations of such various dosage forms include the milk fat globule membrane and amino acid of the present invention alone, or other pharmaceutically acceptable excipients, binders, extenders, disintegrants, interfaces. An active agent, a lubricant, a dispersing agent, a buffering agent, a preservative, a flavoring agent, a fragrance, a film agent, a carrier, a diluent, a medicinal component other than milk fat globule film, and the like can be appropriately combined. Of these dosage forms, the preferred form is oral administration, and oral liquid preparations can be prepared by conventional methods with the addition of flavoring agents, buffers, stabilizers and the like.
経口投与用製剤中の乳脂肪球皮膜の含有量は、一般的に製剤全質量の0.05質量%以上、好ましくは0.2質量%以上であり、且つ30質量%以下、好ましくは10質量%以下である。また0.05〜30質量%、好ましくは0.2〜10質量%である。
経口投与用製剤中のアミノ酸の含有量は、一般的に製剤全質量の0.05質量%以上、好ましくは0.2質量%以上であり、且つ30質量%以下、好ましくは10質量%以下である。また0.05〜30質量%、好ましくは0.2〜10質量%である。
The content of the milk fat globule membrane in the preparation for oral administration is generally 0.05% by mass or more, preferably 0.2% by mass or more, and 30% by mass or less, preferably 10% by mass based on the total mass of the formulation. % Or less. Moreover, it is 0.05-30 mass%, Preferably it is 0.2-10 mass%.
The content of amino acids in the preparation for oral administration is generally 0.05% by mass or more, preferably 0.2% by mass or more, and 30% by mass or less, preferably 10% by mass or less of the total mass of the formulation. is there. Moreover, it is 0.05-30 mass%, Preferably it is 0.2-10 mass%.
本発明の乳脂肪球皮膜及びアミノ酸を含有する上記食品の形態としては、清涼飲料水、茶系飲料、コーヒー飲料、果汁飲料、炭酸飲料、ジュース、ゼリー、ウエハース、ビスケット、パン、麺、ソーセージ等の飲食品や栄養食等の各種食品の他、さらには、上述した経口投与製剤と同様の形態(錠剤、カプセル剤、シロップ等)の栄養補給用組成物が挙げられる。 Examples of the form of the food containing the milk fat globule membrane and amino acid of the present invention include soft drinks, tea beverages, coffee beverages, fruit juice beverages, carbonated beverages, juices, jelly, wafers, biscuits, breads, noodles, sausages, etc. In addition to various foods such as foods and beverages and nutritive foods, further, a nutritional supplement composition in the same form (tablet, capsule, syrup, etc.) as the above-mentioned oral administration preparations can be mentioned.
種々の形態の食品は、本発明の乳脂肪球皮膜及びアミノ酸を単独で、又は他の食品材料や、溶剤、軟化剤、油、乳化剤、防腐剤、香科、安定剤、着色剤、酸化防止剤、保湿剤、増粘剤、本発明以外の有効成分等を適宜組み合わせて調製することができる。 Various types of foods can be obtained by using the milk fat globule membrane and amino acids of the present invention alone, or other food materials, solvents, softeners, oils, emulsifiers, preservatives, fragrances, stabilizers, colorants, antioxidants. It can be prepared by appropriately combining agents, humectants, thickeners, active ingredients other than the present invention, and the like.
また、病者用食品、例えば適当量の栄養補給が困難な高齢者やベッドレスト状態の病者に対する食品としては、経腸栄養剤等の栄養組成物の形態とすることが可能である。 Moreover, as food for sick people, for example, food for elderly people who are difficult to supplement with an appropriate amount of nutrition or sick people in bed rest, it is possible to use a nutritional composition such as enteral nutrient.
また、飼料としては、ウサギ、ラット、マウス等に用いる小動物用飼料、犬、猫等に用いるペットフード等の飼料等が挙げられ、上記食品と同様の形態に調製できる。 Examples of the feed include feed for small animals used for rabbits, rats, mice, etc., feed for pet foods used for dogs, cats, and the like, and can be prepared in the same form as the above foods.
当該食品又は飼料中の乳脂肪球皮膜の含有量は、一般的に製剤全質量の0.05質量%以上、好ましくは0.2質量%以上であり、且つ30質量%以下、好ましくは10質量%以下である。また0.05〜30質量%、好ましくは0.2〜10質量%である。
当該食品飼料中のアミノ酸の含有量は、一般的に製剤全質量の0.05質量%以上、好ましくは0.2質量%以上であり、且つ30質量%以下、好ましくは10質量%以下である。また0.05〜30質量%、好ましくは0.2〜10質量%である。
The content of the milk fat globule membrane in the food or feed is generally 0.05% by mass or more, preferably 0.2% by mass or more, and 30% by mass or less, preferably 10% by mass with respect to the total mass of the preparation. % Or less. Moreover, it is 0.05-30 mass%, Preferably it is 0.2-10 mass%.
The content of amino acids in the food feed is generally 0.05% by mass or more, preferably 0.2% by mass or more, and 30% by mass or less, preferably 10% by mass or less of the total mass of the preparation. . Moreover, it is 0.05-30 mass%, Preferably it is 0.2-10 mass%.
本発明の乳脂肪球皮膜及びアミノ酸の組み合わせを医薬品又は食品として、或いは医薬又は食品に配合して使用する場合の成人1人当たりの1日の投与又は摂取量は、通常、乳脂肪球皮膜として10mg以上、好ましくは100mg以上であり、且つ10000mg以下、好ましくは5000mg以下である。また、10〜10000mg、好ましくは 100〜5000mgである。また、アミノ酸として、10mg以上、好ましくは100mg以上であり、且つ10000mg以下、好ましくは5000mg以下である。また、10〜10000mg、好ましくは100〜5000mgである。
投与又は摂取対象としては、それを必要としているヒト等であるが、例えば筋肉減弱症の患者や加齢性筋肉減弱症のヒト、それらの予備軍などが好適に挙げられる。
When the combination of milk fat globule membrane and amino acid of the present invention is used as a medicine or food, or blended with a medicine or food, the daily administration or intake per adult is usually 10 mg as milk fat globule membrane. As mentioned above, Preferably it is 100 mg or more, and is 10,000 mg or less, Preferably it is 5000 mg or less. Moreover, it is 10-10000 mg, Preferably it is 100-5000 mg. Further, the amino acid is 10 mg or more, preferably 100 mg or more, and 10000 mg or less, preferably 5000 mg or less. Moreover, it is 10-10000 mg, Preferably it is 100-5000 mg.
The administration or ingestion target is a human or the like in need thereof, for example, a patient with muscle weakness, a human with age-related muscle weakness, or a reserve army thereof.
上述した実施形態に関し、本発明においては以下の態様が開示される。
<1>乳脂肪球皮膜及びアミノ酸を組み合わせてなる筋タンパク質合成シグナル増強剤。
<2>乳脂肪球皮膜及びアミノ酸を組み合わせてなる筋量増加剤。
<3>乳脂肪球皮膜とアミノ酸を組み合わせてなる筋萎縮抑制剤。
<4>筋タンパク質合成シグナル増強剤を製造するための乳脂肪球皮膜及びアミノ酸の組み合わせの使用。
<5>筋量増加剤を製造するための乳脂肪球皮膜及びアミノ酸の組み合わせの使用。
<6>筋萎縮抑制剤を製造するための乳脂肪球皮膜及びアミノ酸の組み合わせの使用。
<7>筋タンパク質合成シグナル増強に使用するための乳脂肪球皮膜及びアミノ酸の組み合わせ。
<8>筋量増加に使用するための乳脂肪球皮膜及びアミノ酸の組み合わせ。
<9>筋萎縮抑制に使用するための乳脂肪球皮膜及びアミノ酸の組み合わせ。
<10>乳脂肪球皮膜及びアミノ酸の有効量を組み合わせて投与又は摂取することによる筋タンパク質合成シグナル増強方法。
<11>乳脂肪球皮膜及びアミノ酸の有効量を組み合わせて投与又は摂取することによる筋量増加方法。
<12>乳脂肪球皮膜及びアミノ酸の有効量を組み合わせて投与又は摂取することによる筋萎縮抑制方法。
<13>上記<1>〜<12>において、アミノ酸は必須アミノ酸である。
<14>上記<1>〜<12>において、アミノ酸は疎水性アミノ酸である。
<15>上記<1>〜<12>において、アミノ酸は分岐アミノ酸である。
<16>上記<1>〜<12>において、アミノ酸はロイシン、イソロイシン及びバリンから選ばれる分岐アミノ酸である。
<17>上記<1>〜<12>において、アミノ酸はロイシンである。
<18>上記<1>〜<12>において、乳脂肪球皮膜とアミノ酸の質量比が、1:0.05〜1:30である。
<19>上記<7>〜<9>において、使用は非治療的使用である。
<20>上記<10>〜<12>において、方法は非治療的方法である。
<21>上記<10>〜<12>において、投与又は摂取の対象は、それぞれ筋量増加を必要とする若しくは希望する動物又はヒト、筋萎縮抑制を必要とする若しくは希望する動物又はヒトである。
<22>上記<1>、<4>、<7>、<10>において、筋タンパク質合成シグナル増強は、好適にはp70S6キナーゼのリン酸化である。
With respect to the above-described embodiment, the following aspects are disclosed in the present invention.
<1> A muscle protein synthesis signal enhancer comprising a milk fat globule membrane and an amino acid in combination.
<2> A muscle mass increasing agent comprising a combination of a milk fat globule membrane and an amino acid.
<3> A muscle atrophy inhibitor comprising a combination of a milk fat globule membrane and an amino acid.
<4> Use of a combination of milk fat globule membrane and amino acid for producing a muscle protein synthesis signal enhancer.
<5> Use of a combination of milk fat globule membrane and amino acid for producing a muscle mass increasing agent.
<6> Use of a combination of milk fat globule membrane and amino acid for producing a muscle atrophy inhibitor.
<7> A combination of milk fat globule membrane and amino acid for use in enhancing muscle protein synthesis signal.
<8> A combination of milk fat globule membrane and amino acid for use in increasing muscle mass.
<9> A combination of milk fat globule membrane and amino acid for use in suppressing muscle atrophy.
<10> A method for enhancing muscle protein synthesis signal by administering or ingesting a combination of an effective amount of milk fat globule membrane and an amino acid.
<11> A method for increasing muscle mass by administering or ingesting a combination of an effective amount of milk fat globule membrane and an amino acid.
<12> A method for suppressing muscle atrophy by administering or ingesting a milk fat globule membrane and an effective amount of an amino acid in combination.
<13> In the above items <1> to <12>, an amino acid is an essential amino acid.
<14> In the above items <1> to <12>, the amino acid is a hydrophobic amino acid.
<15> In the above items <1> to <12>, the amino acid is a branched amino acid.
<16> In the above items <1> to <12>, the amino acid is a branched amino acid selected from leucine, isoleucine and valine.
<17> In the above items <1> to <12>, the amino acid is leucine.
<18> In the above items <1> to <12>, the mass ratio between the milk fat globule membrane and the amino acid is 1: 0.05 to 1:30.
<19> In the above <7> to <9>, the use is a non-therapeutic use.
<20> In the above <10> to <12>, the method is a non-therapeutic method.
<21> In the above items <10> to <12>, the administration or ingestion target is an animal or human that requires or desires muscle mass increase, or an animal or human that requires or desires muscle atrophy suppression, respectively. .
<22> In the above <1>, <4>, <7>, <10>, the muscle protein synthesis signal enhancement is preferably phosphorylation of p70S6 kinase.
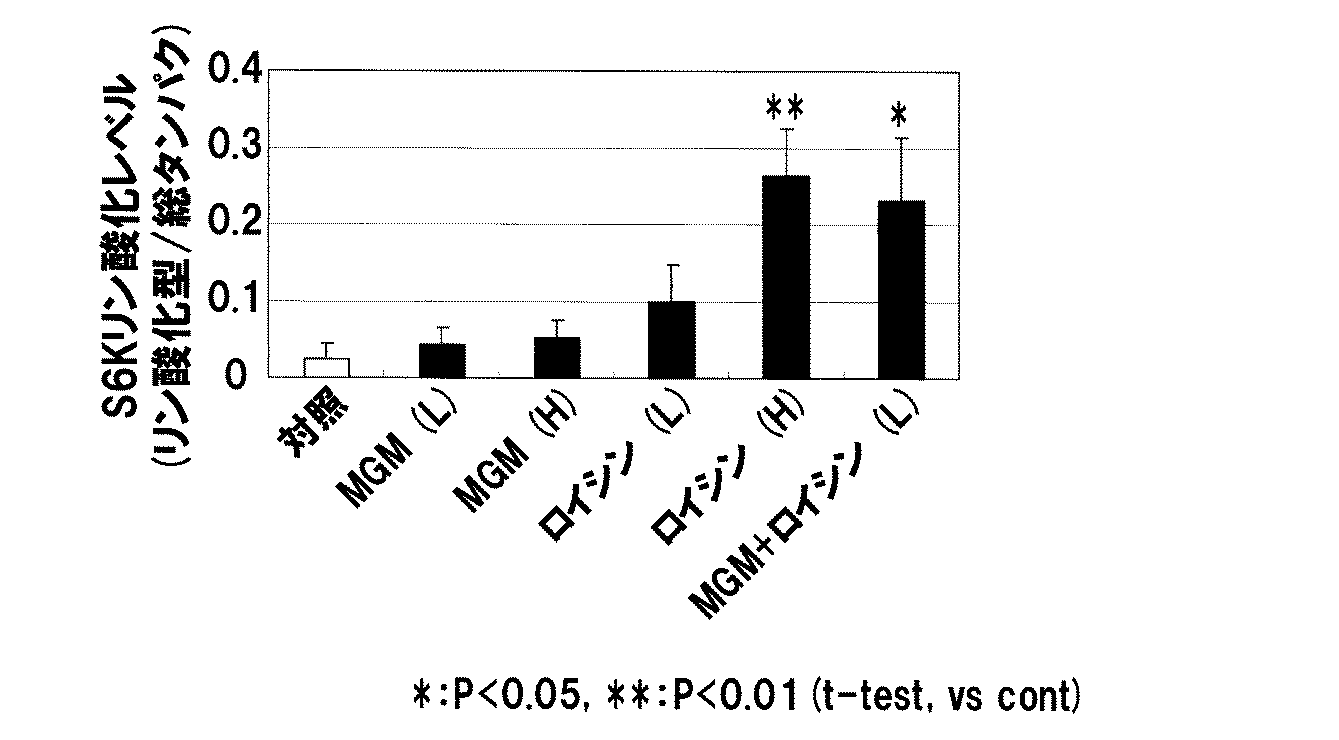
試験例1: 乳脂肪球皮膜及びロイシンの筋タンパク質合成シグナル増強作用 Test Example 1: Enhancement of muscle protein synthesis signal by milk fat globule membrane and leucine
乳脂肪球皮膜(MGM)及びロイシンの筋タンパク質合成シグナル作用に対する評価を下記の通り行った。乳脂肪球皮膜としては、アーラ・フーズ社製PL 20を使用した。ロイシンは関東化学株式会社製のL−ロイシンを使用した。 The milk fat globule membrane (MGM) and leucine were evaluated for the muscle protein synthesis signal action as follows. As the milk fat globule membrane, PL 20 manufactured by Ara Foods was used. As leucine, L-leucine manufactured by Kanto Chemical Co., Ltd. was used.
<方法>
1週間の予備飼育後、6週齢の雄性SDラットを6群{1)対照群、2)MGM(0.68 g/kg体重:L)投与群、3)MGM(1.35 g/kg体重:H)投与群、4)ロイシン(0.68 g/kg体重:L)投与群、5)ロイシン(1.35 g/kg体重:H)投与群、6)MGM(0.68 g/kg体重:L)+ロイシン(0.68 g/kg体重:L)投与群}に分け(各群n=6)、各群18時間の絶食を行った。絶食後、水(対照群)、水に溶解したMGM、或いはロイシン(上記量)を胃ゾンデにて経口投与し、自由飲水・絶食下で1時間飼育を継続した。1時間後、ヒフク筋を摘出し、ウェスタンブロットサンプルに供した。
<Method>
6 weeks old male SD rats were grouped into 6 groups {1) control group, 2) MGM (0.68 g / kg body weight: L) administration group, 3) MGM (1.35 g / kg) Body weight: H) administration group, 4) Leucine (0.68 g / kg body weight: L) administration group, 5) Leucine (1.35 g / kg body weight: H) administration group, 6) MGM (0.68 g / kg) kg body weight: L) + leucine (0.68 g / kg body weight: L) administration group} (each group n = 6), and fasting was performed for 18 hours in each group. After fasting, water (control group), MGM dissolved in water, or leucine (the above amount) was orally administered with a stomach tube, and the breeding was continued for 1 hour under free drinking and fasting. One hour later, the fibroid muscle was removed and subjected to a Western blot sample.
ウェスタンブロッティング法は、以下の手順で行った。
Protease inhibitor cocktail set III(CALBIOCHEM)を添加したCelLytic(TM) MT Cell Lysis Reagent,For mammalian tissues(SIGMA)内で、凍結保存していたヒフク筋組織をホモジナイズし、遠心分離(12000rpm、4℃、10分)後、上清を回収した。各タンパク質濃度をBCA protein assay kit(PIERCE)にて測定し、全ての濃度を統一した。各サンプル20μgをSDS−PAGE(レディーゲル5−15% BioRad)にて分離後、Immun−Blot PVDF membrane For Protein Blotting(Bio−Rad)に転写した。作製したメンブレンを1%のBSA(Wako)にてブロッキングし、その後、表1に示す1次抗体と90分間反応させた。メンブレンを洗浄後、Anti−rabbit IgG,HRP−linked Antibody(Cell Signaling Technology)と60分間反応させ、再びメンブレンを洗浄後、ECL Prime Western Blotting Detection Reagent(GE Healthcare)にてS6Kタンパク質の検出を行った。活性化のシグナルは、p70S6キナーゼ(S6K)の活性化(リン酸化)を指標とし、S6Kの総タンパクおよびリン酸化型を認識する抗体を用い検出したバンドより、[リン酸化型量/総タンパク量]という値を求めて表現した。対照群との有意差検定は、student's t−testにより行った。
Western blotting was performed according to the following procedure.
Homogenized Hf muscle tissue that had been cryopreserved in a Celytic (TM) MT Cell Lysis Reagent, Formalmian tissues (SIGMA) supplemented with Protease inhibitor cocktail set III (CALBIOCHEM) at 4 ° C., rpm 1 Min)) and the supernatant was collected. Each protein concentration was measured by BCA protein assay kit (PIERCE), and all the concentrations were unified. 20 μg of each sample was separated by SDS-PAGE (Ready Gel 5-15% BioRad) and then transferred to Immuno-Blot PVDF membrane For Protein Blotting (Bio-Rad). The prepared membrane was blocked with 1% BSA (Wako), and then reacted with the primary antibodies shown in Table 1 for 90 minutes. After washing the membrane, it was reacted with Anti-rabbit IgG, HRP-linked Antibody (Cell Signaling Technology) for 60 minutes, and after washing the membrane again, ECL Prime Western Blotting Detection Reagent (GE Health was detected by GE Health 6). . The activation signal is based on activation (phosphorylation) of p70S6 kinase (S6K) as an index, and from the band detected using an antibody that recognizes the total protein and phosphorylated form of S6K [phosphorylated / total protein ] Was obtained and expressed. The significant difference test with the control group was performed by student's t-test.
<結果>
低濃度のMGMおよびロイシンの併用投与群で、相乗的なS6Kのリン酸化亢進が認められ、筋タンパク質合成促進が示唆された(図1)。
<Result>
In the combination administration group of low concentrations of MGM and leucine, synergistic enhancement of phosphorylation of S6K was observed, suggesting that muscle protein synthesis was promoted (FIG. 1).
Claims (4)
Priority Applications (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| JP2013081115A JP6158565B2 (en) | 2013-04-09 | 2013-04-09 | Muscle protein synthesis signal enhancer |
Applications Claiming Priority (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| JP2013081115A JP6158565B2 (en) | 2013-04-09 | 2013-04-09 | Muscle protein synthesis signal enhancer |
Publications (3)
| Publication Number | Publication Date |
|---|---|
| JP2014201572A true JP2014201572A (en) | 2014-10-27 |
| JP2014201572A5 JP2014201572A5 (en) | 2016-06-30 |
| JP6158565B2 JP6158565B2 (en) | 2017-07-05 |
Family
ID=52352333
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| JP2013081115A Active JP6158565B2 (en) | 2013-04-09 | 2013-04-09 | Muscle protein synthesis signal enhancer |
Country Status (1)
| Country | Link |
|---|---|
| JP (1) | JP6158565B2 (en) |
Cited By (3)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JP2016123322A (en) * | 2014-12-26 | 2016-07-11 | 花王株式会社 | Solid composition |
| JP2016123324A (en) * | 2014-12-26 | 2016-07-11 | 花王株式会社 | Solid composition |
| JP7462127B1 (en) | 2023-08-16 | 2024-04-05 | 株式会社Healthy Body | Muscle Hypertrophy Composition |
Citations (6)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JP2010059155A (en) * | 2008-08-07 | 2010-03-18 | Kao Corp | Agent for improving exercise function |
| WO2011096413A1 (en) * | 2010-02-03 | 2011-08-11 | 花王株式会社 | Agent for improving motility function |
| JP2011157330A (en) * | 2010-02-03 | 2011-08-18 | Kao Corp | Motor function improving agent |
| WO2012141316A1 (en) * | 2011-04-13 | 2012-10-18 | 味の素株式会社 | Nutritional composition |
| WO2012165345A1 (en) * | 2011-05-27 | 2012-12-06 | 株式会社明治 | Composition for preventing and/or ameliorating cancer-related irreversible metabolism disorders |
| WO2012170021A1 (en) * | 2011-06-08 | 2012-12-13 | Nestec S.A. | Nutritional compositions having exogenous milk fat globule membrane components |
-
2013
- 2013-04-09 JP JP2013081115A patent/JP6158565B2/en active Active
Patent Citations (6)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JP2010059155A (en) * | 2008-08-07 | 2010-03-18 | Kao Corp | Agent for improving exercise function |
| WO2011096413A1 (en) * | 2010-02-03 | 2011-08-11 | 花王株式会社 | Agent for improving motility function |
| JP2011157330A (en) * | 2010-02-03 | 2011-08-18 | Kao Corp | Motor function improving agent |
| WO2012141316A1 (en) * | 2011-04-13 | 2012-10-18 | 味の素株式会社 | Nutritional composition |
| WO2012165345A1 (en) * | 2011-05-27 | 2012-12-06 | 株式会社明治 | Composition for preventing and/or ameliorating cancer-related irreversible metabolism disorders |
| WO2012170021A1 (en) * | 2011-06-08 | 2012-12-13 | Nestec S.A. | Nutritional compositions having exogenous milk fat globule membrane components |
Cited By (3)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JP2016123322A (en) * | 2014-12-26 | 2016-07-11 | 花王株式会社 | Solid composition |
| JP2016123324A (en) * | 2014-12-26 | 2016-07-11 | 花王株式会社 | Solid composition |
| JP7462127B1 (en) | 2023-08-16 | 2024-04-05 | 株式会社Healthy Body | Muscle Hypertrophy Composition |
Also Published As
| Publication number | Publication date |
|---|---|
| JP6158565B2 (en) | 2017-07-05 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| JP6232412B2 (en) | Motor function improver | |
| JP5775657B2 (en) | Anti-fatigue agent containing amino acid composition | |
| JP5581071B2 (en) | Mitochondrial function improver | |
| TWI722992B (en) | Brain function improving agent, and preventive or therapeutic agent for cognitive dysfunction | |
| JP2009507044A (en) | Milk-derived composition and use for enhancing muscle mass or strength | |
| JP6027386B2 (en) | Muscle mass increasing agent | |
| CN105451578A (en) | Chromatographic separation of sugars using blend of cation exchange resins | |
| JP5972717B2 (en) | Cell membrane enhancer | |
| JP6158565B2 (en) | Muscle protein synthesis signal enhancer | |
| JP5841556B2 (en) | Composition of whey growth factor extract for reducing muscle inflammation | |
| US8268360B2 (en) | Motor function improver | |
| JP6254434B2 (en) | Agility improver | |
| JP7260951B2 (en) | knee pain reliever | |
| JP2016138059A (en) | Agent for improving stumble during walking | |
| JP6941445B2 (en) | Balance sensation improver | |
| JP7260952B2 (en) | shoulder pain reliever | |
| JP6823397B2 (en) | Stumbling improver while walking | |
| JP7397562B2 (en) | Lower back pain reliever | |
| JP6450823B2 (en) | Agility improver | |
| JP7260950B2 (en) | low back pain reliever | |
| JP6849390B2 (en) | Joint stiffness improving agent | |
| JP2016138058A (en) | Agent for improving neck stiffness | |
| JP2018016557A (en) | Joint flexibility improver |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| A621 | Written request for application examination |
Free format text: JAPANESE INTERMEDIATE CODE: A621 Effective date: 20160407 |
|
| A521 | Request for written amendment filed |
Free format text: JAPANESE INTERMEDIATE CODE: A523 Effective date: 20160513 |
|
| A131 | Notification of reasons for refusal |
Free format text: JAPANESE INTERMEDIATE CODE: A131 Effective date: 20170131 |
|
| A521 | Request for written amendment filed |
Free format text: JAPANESE INTERMEDIATE CODE: A523 Effective date: 20170330 |
|
| TRDD | Decision of grant or rejection written | ||
| A01 | Written decision to grant a patent or to grant a registration (utility model) |
Free format text: JAPANESE INTERMEDIATE CODE: A01 Effective date: 20170606 |
|
| A61 | First payment of annual fees (during grant procedure) |
Free format text: JAPANESE INTERMEDIATE CODE: A61 Effective date: 20170608 |
|
| R151 | Written notification of patent or utility model registration |
Ref document number: 6158565 Country of ref document: JP Free format text: JAPANESE INTERMEDIATE CODE: R151 |
|
| R250 | Receipt of annual fees |
Free format text: JAPANESE INTERMEDIATE CODE: R250 |
|
| R250 | Receipt of annual fees |
Free format text: JAPANESE INTERMEDIATE CODE: R250 |
|
| R250 | Receipt of annual fees |
Free format text: JAPANESE INTERMEDIATE CODE: R250 |
|
| R250 | Receipt of annual fees |
Free format text: JAPANESE INTERMEDIATE CODE: R250 |
|
| R250 | Receipt of annual fees |
Free format text: JAPANESE INTERMEDIATE CODE: R250 |