JP2008538722A - 難揮発性流体を精留精製する方法 - Google Patents
難揮発性流体を精留精製する方法 Download PDFInfo
- Publication number
- JP2008538722A JP2008538722A JP2008502306A JP2008502306A JP2008538722A JP 2008538722 A JP2008538722 A JP 2008538722A JP 2008502306 A JP2008502306 A JP 2008502306A JP 2008502306 A JP2008502306 A JP 2008502306A JP 2008538722 A JP2008538722 A JP 2008538722A
- Authority
- JP
- Japan
- Prior art keywords
- group
- fluid
- volatile
- auxiliary
- mixture
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Pending
Links
- 238000000034 method Methods 0.000 title claims abstract description 79
- 239000012530 fluid Substances 0.000 title claims abstract description 61
- 238000000926 separation method Methods 0.000 claims abstract description 34
- 238000006243 chemical reaction Methods 0.000 claims abstract description 17
- 239000002904 solvent Substances 0.000 claims description 50
- 239000000203 mixture Substances 0.000 claims description 49
- 239000002608 ionic liquid Substances 0.000 claims description 41
- 239000000126 substance Substances 0.000 claims description 27
- 239000000654 additive Substances 0.000 claims description 24
- 230000000996 additive effect Effects 0.000 claims description 22
- 239000007788 liquid Substances 0.000 claims description 19
- 229920000642 polymer Polymers 0.000 claims description 19
- 238000000605 extraction Methods 0.000 claims description 15
- 239000012528 membrane Substances 0.000 claims description 15
- 238000000638 solvent extraction Methods 0.000 claims description 9
- 238000000622 liquid--liquid extraction Methods 0.000 claims description 8
- 239000000463 material Substances 0.000 claims description 4
- 238000011043 electrofiltration Methods 0.000 claims description 2
- 238000001962 electrophoresis Methods 0.000 claims description 2
- 150000001875 compounds Chemical class 0.000 abstract description 5
- 230000001568 sexual effect Effects 0.000 abstract 1
- -1 phosphonium ions Chemical class 0.000 description 185
- 125000002496 methyl group Chemical group [H]C([H])([H])* 0.000 description 64
- 125000001495 ethyl group Chemical group [H]C([H])([H])C([H])([H])* 0.000 description 50
- 229910052739 hydrogen Inorganic materials 0.000 description 40
- 239000001257 hydrogen Substances 0.000 description 40
- 125000004108 n-butyl group Chemical group [H]C([H])([H])C([H])([H])C([H])([H])C([H])([H])* 0.000 description 30
- 125000002777 acetyl group Chemical group [H]C([H])([H])C(*)=O 0.000 description 29
- 150000002431 hydrogen Chemical class 0.000 description 19
- UHOVQNZJYSORNB-UHFFFAOYSA-N Benzene Chemical compound C1=CC=CC=C1 UHOVQNZJYSORNB-UHFFFAOYSA-N 0.000 description 18
- UFHFLCQGNIYNRP-UHFFFAOYSA-N Hydrogen Chemical compound [H][H] UFHFLCQGNIYNRP-UHFFFAOYSA-N 0.000 description 17
- 239000000047 product Substances 0.000 description 17
- 210000004379 membrane Anatomy 0.000 description 15
- 238000000746 purification Methods 0.000 description 14
- OCJBOOLMMGQPQU-UHFFFAOYSA-N 1,4-dichlorobenzene Chemical compound ClC1=CC=C(Cl)C=C1 OCJBOOLMMGQPQU-UHFFFAOYSA-N 0.000 description 13
- 238000004821 distillation Methods 0.000 description 13
- 239000012535 impurity Substances 0.000 description 11
- 230000000694 effects Effects 0.000 description 10
- 125000000623 heterocyclic group Chemical group 0.000 description 9
- 125000001997 phenyl group Chemical group [H]C1=C([H])C([H])=C(*)C([H])=C1[H] 0.000 description 9
- RTZKZFJDLAIYFH-UHFFFAOYSA-N Diethyl ether Chemical compound CCOCC RTZKZFJDLAIYFH-UHFFFAOYSA-N 0.000 description 8
- 125000004432 carbon atom Chemical group C* 0.000 description 8
- IQQRAVYLUAZUGX-UHFFFAOYSA-N 1-butyl-3-methylimidazolium Chemical compound CCCCN1C=C[N+](C)=C1 IQQRAVYLUAZUGX-UHFFFAOYSA-N 0.000 description 7
- 150000001768 cations Chemical class 0.000 description 7
- 238000000895 extractive distillation Methods 0.000 description 7
- 125000000524 functional group Chemical group 0.000 description 7
- 125000005842 heteroatom Chemical group 0.000 description 7
- 150000002576 ketones Chemical class 0.000 description 7
- 238000010992 reflux Methods 0.000 description 7
- 125000004434 sulfur atom Chemical group 0.000 description 7
- XDTMQSROBMDMFD-UHFFFAOYSA-N Cyclohexane Chemical compound C1CCCCC1 XDTMQSROBMDMFD-UHFFFAOYSA-N 0.000 description 6
- 150000001450 anions Chemical class 0.000 description 6
- 125000003118 aryl group Chemical group 0.000 description 6
- 125000000954 2-hydroxyethyl group Chemical group [H]C([*])([H])C([H])([H])O[H] 0.000 description 5
- LFQSCWFLJHTTHZ-UHFFFAOYSA-N Ethanol Chemical compound CCO LFQSCWFLJHTTHZ-UHFFFAOYSA-N 0.000 description 5
- 241000183024 Populus tremula Species 0.000 description 5
- 125000003545 alkoxy group Chemical group 0.000 description 5
- 125000000217 alkyl group Chemical group 0.000 description 5
- 125000004104 aryloxy group Chemical group 0.000 description 5
- QVGXLLKOCUKJST-UHFFFAOYSA-N atomic oxygen Chemical compound [O] QVGXLLKOCUKJST-UHFFFAOYSA-N 0.000 description 5
- 239000000460 chlorine Substances 0.000 description 5
- 239000007789 gas Substances 0.000 description 5
- 229910052736 halogen Inorganic materials 0.000 description 5
- 150000002367 halogens Chemical class 0.000 description 5
- 229910052760 oxygen Inorganic materials 0.000 description 5
- 239000001301 oxygen Substances 0.000 description 5
- 239000007858 starting material Substances 0.000 description 5
- 125000001731 2-cyanoethyl group Chemical group [H]C([H])(*)C([H])([H])C#N 0.000 description 4
- 239000012752 auxiliary agent Substances 0.000 description 4
- 230000015572 biosynthetic process Effects 0.000 description 4
- 238000009835 boiling Methods 0.000 description 4
- 238000001704 evaporation Methods 0.000 description 4
- 230000008020 evaporation Effects 0.000 description 4
- 125000004435 hydrogen atom Chemical group [H]* 0.000 description 4
- 125000004433 nitrogen atom Chemical group N* 0.000 description 4
- 239000012071 phase Substances 0.000 description 4
- 238000004064 recycling Methods 0.000 description 4
- 230000000630 rising effect Effects 0.000 description 4
- 150000003839 salts Chemical class 0.000 description 4
- 238000003786 synthesis reaction Methods 0.000 description 4
- XLYOFNOQVPJJNP-UHFFFAOYSA-N water Substances O XLYOFNOQVPJJNP-UHFFFAOYSA-N 0.000 description 4
- QTBSBXVTEAMEQO-UHFFFAOYSA-M Acetate Chemical compound CC([O-])=O QTBSBXVTEAMEQO-UHFFFAOYSA-M 0.000 description 3
- QGZKDVFQNNGYKY-UHFFFAOYSA-O Ammonium Chemical compound [NH4+] QGZKDVFQNNGYKY-UHFFFAOYSA-O 0.000 description 3
- OKKJLVBELUTLKV-UHFFFAOYSA-N Methanol Chemical compound OC OKKJLVBELUTLKV-UHFFFAOYSA-N 0.000 description 3
- 238000010521 absorption reaction Methods 0.000 description 3
- 239000002253 acid Substances 0.000 description 3
- 150000001335 aliphatic alkanes Chemical class 0.000 description 3
- 150000001336 alkenes Chemical class 0.000 description 3
- 125000003178 carboxy group Chemical group [H]OC(*)=O 0.000 description 3
- 125000001841 imino group Chemical group [H]N=* 0.000 description 3
- 150000002500 ions Chemical class 0.000 description 3
- 229910052757 nitrogen Inorganic materials 0.000 description 3
- 125000004430 oxygen atom Chemical group O* 0.000 description 3
- 125000001436 propyl group Chemical group [H]C([*])([H])C([H])([H])C([H])([H])[H] 0.000 description 3
- 238000001256 steam distillation Methods 0.000 description 3
- 229910052717 sulfur Inorganic materials 0.000 description 3
- 125000006702 (C1-C18) alkyl group Chemical group 0.000 description 2
- XUAXVBUVQVRIIQ-UHFFFAOYSA-N 1-butyl-2,3-dimethylimidazol-3-ium Chemical compound CCCCN1C=C[N+](C)=C1C XUAXVBUVQVRIIQ-UHFFFAOYSA-N 0.000 description 2
- JYARJXBHOOZQQD-UHFFFAOYSA-N 1-butyl-3-ethylimidazol-1-ium Chemical compound CCCC[N+]=1C=CN(CC)C=1 JYARJXBHOOZQQD-UHFFFAOYSA-N 0.000 description 2
- NNLHWTTWXYBJBQ-UHFFFAOYSA-N 1-butyl-4-methylpyridin-1-ium Chemical compound CCCC[N+]1=CC=C(C)C=C1 NNLHWTTWXYBJBQ-UHFFFAOYSA-N 0.000 description 2
- 125000004200 2-methoxyethyl group Chemical group [H]C([H])([H])OC([H])([H])C([H])([H])* 0.000 description 2
- IJGRMHOSHXDMSA-UHFFFAOYSA-N Atomic nitrogen Chemical compound N#N IJGRMHOSHXDMSA-UHFFFAOYSA-N 0.000 description 2
- KXDHJXZQYSOELW-UHFFFAOYSA-N Carbamic acid Chemical group NC(O)=O KXDHJXZQYSOELW-UHFFFAOYSA-N 0.000 description 2
- ZAMOUSCENKQFHK-UHFFFAOYSA-N Chlorine atom Chemical compound [Cl] ZAMOUSCENKQFHK-UHFFFAOYSA-N 0.000 description 2
- 229920000028 Gradient copolymer Polymers 0.000 description 2
- NBIIXXVUZAFLBC-UHFFFAOYSA-N Phosphoric acid Chemical compound OP(O)(O)=O NBIIXXVUZAFLBC-UHFFFAOYSA-N 0.000 description 2
- QAOWNCQODCNURD-UHFFFAOYSA-L Sulfate Chemical compound [O-]S([O-])(=O)=O QAOWNCQODCNURD-UHFFFAOYSA-L 0.000 description 2
- LSNNMFCWUKXFEE-UHFFFAOYSA-N Sulfurous acid Chemical compound OS(O)=O LSNNMFCWUKXFEE-UHFFFAOYSA-N 0.000 description 2
- 150000007513 acids Chemical class 0.000 description 2
- 125000004448 alkyl carbonyl group Chemical group 0.000 description 2
- VSCWAEJMTAWNJL-UHFFFAOYSA-K aluminium trichloride Chemical compound Cl[Al](Cl)Cl VSCWAEJMTAWNJL-UHFFFAOYSA-K 0.000 description 2
- 150000001408 amides Chemical class 0.000 description 2
- 125000003277 amino group Chemical group 0.000 description 2
- 150000001491 aromatic compounds Chemical class 0.000 description 2
- 125000005129 aryl carbonyl group Chemical group 0.000 description 2
- 125000001797 benzyl group Chemical group [H]C1=C([H])C([H])=C(C([H])=C1[H])C([H])([H])* 0.000 description 2
- 125000000484 butyl group Chemical group [H]C([*])([H])C([H])([H])C([H])([H])C([H])([H])[H] 0.000 description 2
- 150000001735 carboxylic acids Chemical class 0.000 description 2
- 229910052801 chlorine Inorganic materials 0.000 description 2
- 238000011109 contamination Methods 0.000 description 2
- 238000000354 decomposition reaction Methods 0.000 description 2
- 238000010586 diagram Methods 0.000 description 2
- 238000010790 dilution Methods 0.000 description 2
- 239000012895 dilution Substances 0.000 description 2
- 125000002147 dimethylamino group Chemical group [H]C([H])([H])N(*)C([H])([H])[H] 0.000 description 2
- 125000003438 dodecyl group Chemical group [H]C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])* 0.000 description 2
- 230000005684 electric field Effects 0.000 description 2
- 238000005868 electrolysis reaction Methods 0.000 description 2
- 125000003754 ethoxycarbonyl group Chemical group C(=O)(OCC)* 0.000 description 2
- KWIUHFFTVRNATP-UHFFFAOYSA-N glycine betaine Chemical compound C[N+](C)(C)CC([O-])=O KWIUHFFTVRNATP-UHFFFAOYSA-N 0.000 description 2
- 229920001519 homopolymer Polymers 0.000 description 2
- 150000002430 hydrocarbons Chemical class 0.000 description 2
- 125000002887 hydroxy group Chemical group [H]O* 0.000 description 2
- 229920000587 hyperbranched polymer Polymers 0.000 description 2
- 239000000543 intermediate Substances 0.000 description 2
- 125000001449 isopropyl group Chemical group [H]C([H])([H])C([H])(*)C([H])([H])[H] 0.000 description 2
- 230000008018 melting Effects 0.000 description 2
- 238000002844 melting Methods 0.000 description 2
- 238000010327 methods by industry Methods 0.000 description 2
- 125000001160 methoxycarbonyl group Chemical group [H]C([H])([H])OC(*)=O 0.000 description 2
- 125000000636 p-nitrophenyl group Chemical group [H]C1=C([H])C(=C([H])C([H])=C1*)[N+]([O-])=O 0.000 description 2
- 238000005192 partition Methods 0.000 description 2
- 125000001501 propionyl group Chemical group O=C([*])C([H])([H])C([H])([H])[H] 0.000 description 2
- CZPWVGJYEJSRLH-UHFFFAOYSA-P pyrimidine-1,3-diium Chemical compound C1=C[NH+]=C[NH+]=C1 CZPWVGJYEJSRLH-UHFFFAOYSA-P 0.000 description 2
- 230000008929 regeneration Effects 0.000 description 2
- 238000011069 regeneration method Methods 0.000 description 2
- 229920006395 saturated elastomer Polymers 0.000 description 2
- 125000002914 sec-butyl group Chemical group [H]C([H])([H])C([H])([H])C([H])(*)C([H])([H])[H] 0.000 description 2
- 238000004088 simulation Methods 0.000 description 2
- 125000000999 tert-butyl group Chemical group [H]C([H])([H])C(*)(C([H])([H])[H])C([H])([H])[H] 0.000 description 2
- 125000003944 tolyl group Chemical group 0.000 description 2
- 239000012808 vapor phase Substances 0.000 description 2
- 239000012855 volatile organic compound Substances 0.000 description 2
- 125000004178 (C1-C4) alkyl group Chemical group 0.000 description 1
- 125000000229 (C1-C4)alkoxy group Chemical group 0.000 description 1
- ZXMGHDIOOHOAAE-UHFFFAOYSA-N 1,1,1-trifluoro-n-(trifluoromethylsulfonyl)methanesulfonamide Chemical compound FC(F)(F)S(=O)(=O)NS(=O)(=O)C(F)(F)F ZXMGHDIOOHOAAE-UHFFFAOYSA-N 0.000 description 1
- GIWQSPITLQVMSG-UHFFFAOYSA-N 1,2-dimethylimidazole Chemical compound CC1=NC=CN1C GIWQSPITLQVMSG-UHFFFAOYSA-N 0.000 description 1
- UMZDENILBZKMFY-UHFFFAOYSA-N 1,2-dimethylpyridin-1-ium Chemical compound CC1=CC=CC=[N+]1C UMZDENILBZKMFY-UHFFFAOYSA-N 0.000 description 1
- NOBVKAMFUBMCCA-UHFFFAOYSA-N 1,3,4,5-tetramethylimidazol-1-ium Chemical compound CC1=C(C)[N+](C)=CN1C NOBVKAMFUBMCCA-UHFFFAOYSA-N 0.000 description 1
- CDIWYWUGTVLWJM-UHFFFAOYSA-N 1,3,4-trimethylimidazol-1-ium Chemical compound CC1=C[N+](C)=CN1C CDIWYWUGTVLWJM-UHFFFAOYSA-N 0.000 description 1
- CLHRGZGPVVFEAO-UHFFFAOYSA-N 1,3-dibutyl-2-methylimidazol-1-ium Chemical compound CCCCN1C=C[N+](CCCC)=C1C CLHRGZGPVVFEAO-UHFFFAOYSA-N 0.000 description 1
- NWXVIUBYBJUOAY-UHFFFAOYSA-N 1,3-dibutylimidazol-1-ium Chemical compound CCCCN1C=C[N+](CCCC)=C1 NWXVIUBYBJUOAY-UHFFFAOYSA-N 0.000 description 1
- HVVRUQBMAZRKPJ-UHFFFAOYSA-N 1,3-dimethylimidazolium Chemical compound CN1C=C[N+](C)=C1 HVVRUQBMAZRKPJ-UHFFFAOYSA-N 0.000 description 1
- HQNBJNDMPLEUDS-UHFFFAOYSA-N 1,5-dimethylimidazole Chemical compound CC1=CN=CN1C HQNBJNDMPLEUDS-UHFFFAOYSA-N 0.000 description 1
- BXKLAIZZZVWDLP-UHFFFAOYSA-N 1-butyl-2-ethyl-5-methylimidazole Chemical compound CCCCN1C(C)=CN=C1CC BXKLAIZZZVWDLP-UHFFFAOYSA-N 0.000 description 1
- DRRUKZGOEVGCED-UHFFFAOYSA-N 1-butyl-2-ethyl-6-methylpyridin-1-ium Chemical compound CCCC[N+]1=C(C)C=CC=C1CC DRRUKZGOEVGCED-UHFFFAOYSA-N 0.000 description 1
- FWEIDDZCICNFFR-UHFFFAOYSA-N 1-butyl-2-ethylimidazole Chemical compound CCCCN1C=CN=C1CC FWEIDDZCICNFFR-UHFFFAOYSA-N 0.000 description 1
- GYZXRPOUUZKBAT-UHFFFAOYSA-N 1-butyl-2-ethylpyridin-1-ium Chemical compound CCCC[N+]1=CC=CC=C1CC GYZXRPOUUZKBAT-UHFFFAOYSA-N 0.000 description 1
- WHLZPGRDRYCVRQ-UHFFFAOYSA-N 1-butyl-2-methylimidazole Chemical compound CCCCN1C=CN=C1C WHLZPGRDRYCVRQ-UHFFFAOYSA-N 0.000 description 1
- BHIGPVGNEXDQBL-UHFFFAOYSA-N 1-butyl-2-methylpyridin-1-ium Chemical compound CCCC[N+]1=CC=CC=C1C BHIGPVGNEXDQBL-UHFFFAOYSA-N 0.000 description 1
- RIDWYWYHKGNNOF-UHFFFAOYSA-N 1-butyl-3,4,5-trimethylimidazol-3-ium Chemical compound CCCCN1C=[N+](C)C(C)=C1C RIDWYWYHKGNNOF-UHFFFAOYSA-N 0.000 description 1
- QVHIOPQEIQXVPH-UHFFFAOYSA-N 1-butyl-5-methylimidazole Chemical compound CCCCN1C=NC=C1C QVHIOPQEIQXVPH-UHFFFAOYSA-N 0.000 description 1
- LDVVBLGHGCHZBJ-UHFFFAOYSA-N 1-decyl-3-methylimidazolium Chemical compound CCCCCCCCCCN1C=C[N+](C)=C1 LDVVBLGHGCHZBJ-UHFFFAOYSA-N 0.000 description 1
- 125000004343 1-phenylethyl group Chemical group [H]C1=C([H])C([H])=C(C([H])=C1[H])C([H])(*)C([H])([H])[H] 0.000 description 1
- 125000004206 2,2,2-trifluoroethyl group Chemical group [H]C([H])(*)C(F)(F)F 0.000 description 1
- 125000003456 2,6-dinitrophenyl group Chemical group [H]C1=C([H])C(=C(*)C(=C1[H])[N+]([O-])=O)[N+]([O-])=O 0.000 description 1
- 125000000022 2-aminoethyl group Chemical group [H]C([*])([H])C([H])([H])N([H])[H] 0.000 description 1
- 125000001340 2-chloroethyl group Chemical group [H]C([H])(Cl)C([H])([H])* 0.000 description 1
- 125000003006 2-dimethylaminoethyl group Chemical group [H]C([H])([H])N(C([H])([H])[H])C([H])([H])C([H])([H])* 0.000 description 1
- YQCACEPSJCNFNB-UHFFFAOYSA-N 2-ethyl-1,6-dimethylpyridin-1-ium Chemical compound CCC1=CC=CC(C)=[N+]1C YQCACEPSJCNFNB-UHFFFAOYSA-N 0.000 description 1
- UINDRJHZBAGQFD-UHFFFAOYSA-N 2-ethyl-1-methylimidazole Chemical compound CCC1=NC=CN1C UINDRJHZBAGQFD-UHFFFAOYSA-N 0.000 description 1
- BJMBCVIFMXOIIH-UHFFFAOYSA-N 2-ethyl-1-methylpyridin-1-ium Chemical compound CCC1=CC=CC=[N+]1C BJMBCVIFMXOIIH-UHFFFAOYSA-N 0.000 description 1
- 125000000094 2-phenylethyl group Chemical group [H]C1=C([H])C([H])=C(C([H])=C1[H])C([H])([H])C([H])([H])* 0.000 description 1
- SVTMPVRFMNPZTP-UHFFFAOYSA-N 3-butyl-1,4-dimethylimidazol-1-ium Chemical compound CCCCN1C=[N+](C)C=C1C SVTMPVRFMNPZTP-UHFFFAOYSA-N 0.000 description 1
- WXMVWUBWIHZLMQ-UHFFFAOYSA-N 3-methyl-1-octylimidazolium Chemical compound CCCCCCCCN1C=C[N+](C)=C1 WXMVWUBWIHZLMQ-UHFFFAOYSA-N 0.000 description 1
- 125000004042 4-aminobutyl group Chemical group [H]C([*])([H])C([H])([H])C([H])([H])C([H])([H])N([H])[H] 0.000 description 1
- 125000004800 4-bromophenyl group Chemical group [H]C1=C([H])C(*)=C([H])C([H])=C1Br 0.000 description 1
- 125000006283 4-chlorobenzyl group Chemical group [H]C1=C([H])C(=C([H])C([H])=C1Cl)C([H])([H])* 0.000 description 1
- 229910016467 AlCl 4 Inorganic materials 0.000 description 1
- 229910021591 Copper(I) chloride Inorganic materials 0.000 description 1
- SNRUBQQJIBEYMU-UHFFFAOYSA-N Dodecane Natural products CCCCCCCCCCCC SNRUBQQJIBEYMU-UHFFFAOYSA-N 0.000 description 1
- KIWBPDUYBMNFTB-UHFFFAOYSA-N Ethyl hydrogen sulfate Chemical compound CCOS(O)(=O)=O KIWBPDUYBMNFTB-UHFFFAOYSA-N 0.000 description 1
- PQBAWAQIRZIWIV-UHFFFAOYSA-N N-methylpyridinium Chemical compound C[N+]1=CC=CC=C1 PQBAWAQIRZIWIV-UHFFFAOYSA-N 0.000 description 1
- IOVCWXUNBOPUCH-UHFFFAOYSA-M Nitrite anion Chemical compound [O-]N=O IOVCWXUNBOPUCH-UHFFFAOYSA-M 0.000 description 1
- NBIIXXVUZAFLBC-UHFFFAOYSA-L Phosphate ion(2-) Chemical compound OP([O-])([O-])=O NBIIXXVUZAFLBC-UHFFFAOYSA-L 0.000 description 1
- RWRDLPDLKQPQOW-UHFFFAOYSA-O Pyrrolidinium ion Chemical compound C1CC[NH2+]C1 RWRDLPDLKQPQOW-UHFFFAOYSA-O 0.000 description 1
- NINIDFKCEFEMDL-UHFFFAOYSA-N Sulfur Chemical compound [S] NINIDFKCEFEMDL-UHFFFAOYSA-N 0.000 description 1
- DTQVDTLACAAQTR-UHFFFAOYSA-M Trifluoroacetate Chemical compound [O-]C(=O)C(F)(F)F DTQVDTLACAAQTR-UHFFFAOYSA-M 0.000 description 1
- 239000011358 absorbing material Substances 0.000 description 1
- 150000001252 acrylic acid derivatives Chemical class 0.000 description 1
- 150000001298 alcohols Chemical class 0.000 description 1
- 150000001334 alicyclic compounds Chemical class 0.000 description 1
- 150000007824 aliphatic compounds Chemical class 0.000 description 1
- 229920005603 alternating copolymer Polymers 0.000 description 1
- XAGFODPZIPBFFR-UHFFFAOYSA-N aluminium Chemical compound [Al] XAGFODPZIPBFFR-UHFFFAOYSA-N 0.000 description 1
- 229910052782 aluminium Inorganic materials 0.000 description 1
- 229910000147 aluminium phosphate Inorganic materials 0.000 description 1
- 125000003785 benzimidazolyl group Chemical group N1=C(NC2=C1C=CC=C2)* 0.000 description 1
- 125000001164 benzothiazolyl group Chemical group S1C(=NC2=C1C=CC=C2)* 0.000 description 1
- 125000004541 benzoxazolyl group Chemical group O1C(=NC2=C1C=CC=C2)* 0.000 description 1
- 125000003236 benzoyl group Chemical group [H]C1=C([H])C([H])=C(C([H])=C1[H])C(*)=O 0.000 description 1
- 125000001584 benzyloxycarbonyl group Chemical group C(=O)(OCC1=CC=CC=C1)* 0.000 description 1
- 229960003237 betaine Drugs 0.000 description 1
- 125000002619 bicyclic group Chemical group 0.000 description 1
- 125000000319 biphenyl-4-yl group Chemical group [H]C1=C([H])C([H])=C([H])C([H])=C1C1=C([H])C([H])=C([*])C([H])=C1[H] 0.000 description 1
- XHIHMDHAPXMAQK-UHFFFAOYSA-N bis(trifluoromethylsulfonyl)azanide;1-butylpyridin-1-ium Chemical compound CCCC[N+]1=CC=CC=C1.FC(F)(F)S(=O)(=O)[N-]S(=O)(=O)C(F)(F)F XHIHMDHAPXMAQK-UHFFFAOYSA-N 0.000 description 1
- 229920001400 block copolymer Polymers 0.000 description 1
- 238000004364 calculation method Methods 0.000 description 1
- 239000003990 capacitor Substances 0.000 description 1
- 229910052799 carbon Inorganic materials 0.000 description 1
- 125000002915 carbonyl group Chemical group [*:2]C([*:1])=O 0.000 description 1
- 239000003054 catalyst Substances 0.000 description 1
- 239000007795 chemical reaction product Substances 0.000 description 1
- 125000002668 chloroacetyl group Chemical group ClCC(=O)* 0.000 description 1
- 125000004218 chloromethyl group Chemical group [H]C([H])(Cl)* 0.000 description 1
- 125000000068 chlorophenyl group Chemical group 0.000 description 1
- JIVPVXMEBJLZRO-UHFFFAOYSA-N chlorthalidone Chemical compound C1=C(Cl)C(S(=O)(=O)N)=CC(C2(O)C3=CC=CC=C3C(=O)N2)=C1 JIVPVXMEBJLZRO-UHFFFAOYSA-N 0.000 description 1
- 238000004587 chromatography analysis Methods 0.000 description 1
- OXBLHERUFWYNTN-UHFFFAOYSA-M copper(I) chloride Chemical compound [Cu]Cl OXBLHERUFWYNTN-UHFFFAOYSA-M 0.000 description 1
- ORTQZVOHEJQUHG-UHFFFAOYSA-L copper(II) chloride Chemical compound Cl[Cu]Cl ORTQZVOHEJQUHG-UHFFFAOYSA-L 0.000 description 1
- 238000002425 crystallisation Methods 0.000 description 1
- 230000008025 crystallization Effects 0.000 description 1
- 125000004093 cyano group Chemical group *C#N 0.000 description 1
- 125000004122 cyclic group Chemical group 0.000 description 1
- 150000003997 cyclic ketones Chemical class 0.000 description 1
- 125000006639 cyclohexyl carbonyl group Chemical group 0.000 description 1
- 125000000113 cyclohexyl group Chemical group [H]C1([H])C([H])([H])C([H])([H])C([H])(*)C([H])([H])C1([H])[H] 0.000 description 1
- 125000000640 cyclooctyl group Chemical group [H]C1([H])C([H])([H])C([H])([H])C([H])([H])C([H])(*)C([H])([H])C([H])([H])C1([H])[H] 0.000 description 1
- 125000006638 cyclopentyl carbonyl group Chemical group 0.000 description 1
- 125000001511 cyclopentyl group Chemical group [H]C1([H])C([H])([H])C([H])([H])C([H])(*)C1([H])[H] 0.000 description 1
- 125000003074 decanoyl group Chemical group [H]C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C(*)=O 0.000 description 1
- 125000002704 decyl group Chemical group [H]C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])* 0.000 description 1
- 239000007857 degradation product Substances 0.000 description 1
- 125000004188 dichlorophenyl group Chemical group 0.000 description 1
- 125000001664 diethylamino group Chemical group [H]C([H])([H])C([H])([H])N(*)C([H])([H])C([H])([H])[H] 0.000 description 1
- 125000004212 difluorophenyl group Chemical group 0.000 description 1
- NBIIXXVUZAFLBC-UHFFFAOYSA-M dihydrogenphosphate Chemical compound OP(O)([O-])=O NBIIXXVUZAFLBC-UHFFFAOYSA-M 0.000 description 1
- 125000005805 dimethoxy phenyl group Chemical group 0.000 description 1
- 230000005518 electrochemistry Effects 0.000 description 1
- 238000000909 electrodialysis Methods 0.000 description 1
- 239000000839 emulsion Substances 0.000 description 1
- 125000001301 ethoxy group Chemical group [H]C([H])([H])C([H])([H])O* 0.000 description 1
- 230000002349 favourable effect Effects 0.000 description 1
- 238000001640 fractional crystallisation Methods 0.000 description 1
- 150000002240 furans Chemical class 0.000 description 1
- 125000002541 furyl group Chemical group 0.000 description 1
- 238000004817 gas chromatography Methods 0.000 description 1
- 238000007429 general method Methods 0.000 description 1
- 150000004820 halides Chemical class 0.000 description 1
- 125000003187 heptyl group Chemical group [H]C([*])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])[H] 0.000 description 1
- 125000004051 hexyl group Chemical group [H]C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])* 0.000 description 1
- 125000005935 hexyloxycarbonyl group Chemical group 0.000 description 1
- 229930195733 hydrocarbon Natural products 0.000 description 1
- 125000005462 imide group Chemical group 0.000 description 1
- 125000001041 indolyl group Chemical group 0.000 description 1
- 125000005928 isopropyloxycarbonyl group Chemical group [H]C([H])([H])C([H])(OC(*)=O)C([H])([H])[H] 0.000 description 1
- 125000000400 lauroyl group Chemical group O=C([*])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])[H] 0.000 description 1
- 239000007791 liquid phase Substances 0.000 description 1
- 239000000314 lubricant Substances 0.000 description 1
- 238000005259 measurement Methods 0.000 description 1
- JZMJDSHXVKJFKW-UHFFFAOYSA-M methyl sulfate(1-) Chemical compound COS([O-])(=O)=O JZMJDSHXVKJFKW-UHFFFAOYSA-M 0.000 description 1
- 125000001421 myristyl group Chemical group [H]C([*])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])[H] 0.000 description 1
- 125000006606 n-butoxy group Chemical group 0.000 description 1
- 125000000740 n-pentyl group Chemical group [H]C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])* 0.000 description 1
- 125000000449 nitro group Chemical group [O-][N+](*)=O 0.000 description 1
- YPJKMVATUPSWOH-UHFFFAOYSA-N nitrooxidanyl Chemical compound [O][N+]([O-])=O YPJKMVATUPSWOH-UHFFFAOYSA-N 0.000 description 1
- 125000002868 norbornyl group Chemical group C12(CCC(CC1)C2)* 0.000 description 1
- 125000002347 octyl group Chemical group [H]C([*])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])[H] 0.000 description 1
- 150000007524 organic acids Chemical class 0.000 description 1
- 235000005985 organic acids Nutrition 0.000 description 1
- 239000003960 organic solvent Substances 0.000 description 1
- 125000000913 palmityl group Chemical group [H]C([*])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])[H] 0.000 description 1
- 125000001147 pentyl group Chemical group C(CCCC)* 0.000 description 1
- 239000012466 permeate Substances 0.000 description 1
- 125000000951 phenoxy group Chemical group [H]C1=C([H])C([H])=C(O*)C([H])=C1[H] 0.000 description 1
- 125000004437 phosphorous atom Chemical group 0.000 description 1
- 229910052698 phosphorus Inorganic materials 0.000 description 1
- 229920000962 poly(amidoamine) Polymers 0.000 description 1
- 229920000333 poly(propyleneimine) Polymers 0.000 description 1
- 229920000728 polyester Polymers 0.000 description 1
- 229920000223 polyglycerol Polymers 0.000 description 1
- 238000001556 precipitation Methods 0.000 description 1
- 238000002360 preparation method Methods 0.000 description 1
- 238000004237 preparative chromatography Methods 0.000 description 1
- 125000006410 propenylene group Chemical group 0.000 description 1
- 125000004742 propyloxycarbonyl group Chemical group 0.000 description 1
- 125000001725 pyrenyl group Chemical group 0.000 description 1
- JUJWROOIHBZHMG-UHFFFAOYSA-O pyridinium Chemical compound C1=CC=[NH+]C=C1 JUJWROOIHBZHMG-UHFFFAOYSA-O 0.000 description 1
- 125000004076 pyridyl group Chemical group 0.000 description 1
- 238000000197 pyrolysis Methods 0.000 description 1
- 229920005604 random copolymer Polymers 0.000 description 1
- 239000012925 reference material Substances 0.000 description 1
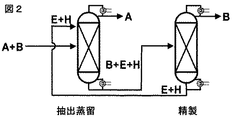
- 238000007670 refining Methods 0.000 description 1
- 239000012465 retentate Substances 0.000 description 1
- IKGXIBQEEMLURG-NVPNHPEKSA-N rutin Chemical compound O[C@@H]1[C@H](O)[C@@H](O)[C@H](C)O[C@H]1OC[C@@H]1[C@@H](O)[C@H](O)[C@@H](O)[C@H](OC=2C(C3=C(O)C=C(O)C=C3OC=2C=2C=C(O)C(O)=CC=2)=O)O1 IKGXIBQEEMLURG-NVPNHPEKSA-N 0.000 description 1
- 125000005930 sec-butyloxycarbonyl group Chemical group [H]C([H])([H])C([H])([H])C([H])(OC(*)=O)C([H])([H])[H] 0.000 description 1
- 239000007787 solid Substances 0.000 description 1
- 239000011343 solid material Substances 0.000 description 1
- 239000003381 stabilizer Substances 0.000 description 1
- 125000004079 stearyl group Chemical group [H]C([*])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])[H] 0.000 description 1
- 238000006467 substitution reaction Methods 0.000 description 1
- 239000011593 sulfur Substances 0.000 description 1
- 239000000725 suspension Substances 0.000 description 1
- 125000005931 tert-butyloxycarbonyl group Chemical group [H]C([H])([H])C(OC(*)=O)(C([H])([H])[H])C([H])([H])[H] 0.000 description 1
- 125000001973 tert-pentyl group Chemical group [H]C([H])([H])C([H])([H])C(*)(C([H])([H])[H])C([H])([H])[H] 0.000 description 1
- 125000001544 thienyl group Chemical group 0.000 description 1
- 150000003568 thioethers Chemical class 0.000 description 1
- 125000003396 thiol group Chemical group [H]S* 0.000 description 1
- JOXIMZWYDAKGHI-UHFFFAOYSA-N toluene-4-sulfonic acid Chemical compound CC1=CC=C(S(O)(=O)=O)C=C1 JOXIMZWYDAKGHI-UHFFFAOYSA-N 0.000 description 1
- 125000005425 toluyl group Chemical group 0.000 description 1
- 238000012546 transfer Methods 0.000 description 1
- 125000003866 trichloromethyl group Chemical group ClC(Cl)(Cl)* 0.000 description 1
- ITMCEJHCFYSIIV-UHFFFAOYSA-M triflate Chemical compound [O-]S(=O)(=O)C(F)(F)F ITMCEJHCFYSIIV-UHFFFAOYSA-M 0.000 description 1
- 125000004044 trifluoroacetyl group Chemical group FC(C(=O)*)(F)F 0.000 description 1
- 125000002023 trifluoromethyl group Chemical group FC(F)(F)* 0.000 description 1
- 125000005023 xylyl group Chemical group 0.000 description 1
Images
Classifications
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D233/00—Heterocyclic compounds containing 1,3-diazole or hydrogenated 1,3-diazole rings, not condensed with other rings
- C07D233/54—Heterocyclic compounds containing 1,3-diazole or hydrogenated 1,3-diazole rings, not condensed with other rings having two double bonds between ring members or between ring members and non-ring members
- C07D233/56—Heterocyclic compounds containing 1,3-diazole or hydrogenated 1,3-diazole rings, not condensed with other rings having two double bonds between ring members or between ring members and non-ring members with only hydrogen atoms or radicals containing only hydrogen and carbon atoms, attached to ring carbon atoms
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B01—PHYSICAL OR CHEMICAL PROCESSES OR APPARATUS IN GENERAL
- B01D—SEPARATION
- B01D3/00—Distillation or related exchange processes in which liquids are contacted with gaseous media, e.g. stripping
- B01D3/14—Fractional distillation or use of a fractionation or rectification column
- B01D3/143—Fractional distillation or use of a fractionation or rectification column by two or more of a fractionation, separation or rectification step
- B01D3/146—Multiple effect distillation
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B01—PHYSICAL OR CHEMICAL PROCESSES OR APPARATUS IN GENERAL
- B01D—SEPARATION
- B01D3/00—Distillation or related exchange processes in which liquids are contacted with gaseous media, e.g. stripping
- B01D3/34—Distillation or related exchange processes in which liquids are contacted with gaseous media, e.g. stripping with one or more auxiliary substances
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07C—ACYCLIC OR CARBOCYCLIC COMPOUNDS
- C07C7/00—Purification; Separation; Use of additives
- C07C7/04—Purification; Separation; Use of additives by distillation
- C07C7/05—Purification; Separation; Use of additives by distillation with the aid of auxiliary compounds
- C07C7/08—Purification; Separation; Use of additives by distillation with the aid of auxiliary compounds by extractive distillation
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07F—ACYCLIC, CARBOCYCLIC OR HETEROCYCLIC COMPOUNDS CONTAINING ELEMENTS OTHER THAN CARBON, HYDROGEN, HALOGEN, OXYGEN, NITROGEN, SULFUR, SELENIUM OR TELLURIUM
- C07F9/00—Compounds containing elements of Groups 5 or 15 of the Periodic Table
- C07F9/02—Phosphorus compounds
- C07F9/28—Phosphorus compounds with one or more P—C bonds
- C07F9/54—Quaternary phosphonium compounds
Landscapes
- Chemical & Material Sciences (AREA)
- Organic Chemistry (AREA)
- Chemical Kinetics & Catalysis (AREA)
- Engineering & Computer Science (AREA)
- Biochemistry (AREA)
- General Health & Medical Sciences (AREA)
- Molecular Biology (AREA)
- Life Sciences & Earth Sciences (AREA)
- Health & Medical Sciences (AREA)
- Analytical Chemistry (AREA)
- Oil, Petroleum & Natural Gas (AREA)
- Water Supply & Treatment (AREA)
- Organic Low-Molecular-Weight Compounds And Preparation Thereof (AREA)
- Vaporization, Distillation, Condensation, Sublimation, And Cold Traps (AREA)
- Other Resins Obtained By Reactions Not Involving Carbon-To-Carbon Unsaturated Bonds (AREA)
- Physical Or Chemical Processes And Apparatus (AREA)
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| DE102005013030A DE102005013030A1 (de) | 2005-03-22 | 2005-03-22 | Verfahren zur destillativen Reinigung schwerflüchtiger Fluide |
| PCT/EP2006/002553 WO2006100023A1 (de) | 2005-03-22 | 2006-03-21 | Verfahren zur destillativen reinigung schwerflüchtiger fluide |
Publications (2)
| Publication Number | Publication Date |
|---|---|
| JP2008538722A true JP2008538722A (ja) | 2008-11-06 |
| JP2008538722A5 JP2008538722A5 (enExample) | 2009-05-07 |
Family
ID=36579102
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| JP2008502306A Pending JP2008538722A (ja) | 2005-03-22 | 2006-03-21 | 難揮発性流体を精留精製する方法 |
Country Status (7)
| Country | Link |
|---|---|
| US (1) | US8277615B2 (enExample) |
| EP (1) | EP1866045B1 (enExample) |
| JP (1) | JP2008538722A (enExample) |
| CN (1) | CN101180112B (enExample) |
| AT (1) | ATE530237T1 (enExample) |
| DE (1) | DE102005013030A1 (enExample) |
| WO (1) | WO2006100023A1 (enExample) |
Cited By (3)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JP2010536571A (ja) * | 2007-08-31 | 2010-12-02 | ビーエーエスエフ ソシエタス・ヨーロピア | イオン性液体の蒸留 |
| JP2012518524A (ja) * | 2009-02-23 | 2012-08-16 | ビーエーエスエフ ソシエタス・ヨーロピア | 蒸留助剤の使用下でのイオン性液体の蒸留 |
| JP2013142081A (ja) * | 2012-01-12 | 2013-07-22 | Lanxess Deutschland Gmbh | メントール化合物異性体の分離 |
Families Citing this family (22)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| DE102005028451B4 (de) | 2005-06-17 | 2017-02-16 | Evonik Degussa Gmbh | Verfahren zum Transport von Wärme |
| DE102006024397B3 (de) * | 2006-05-24 | 2007-10-11 | Universität Rostock | Mehrphasen-Membran |
| EP2088389B1 (de) * | 2008-02-05 | 2017-05-10 | Evonik Degussa GmbH | Absorptionskältemaschine |
| EP2087930A1 (de) * | 2008-02-05 | 2009-08-12 | Evonik Degussa GmbH | Verfahren zur Absorption eines flüchtigen Stoffes in einem flüssigen Absorptionsmittel |
| RU2400469C2 (ru) * | 2008-12-09 | 2010-09-27 | Общество с ограниченной ответственностью "Новые технологии" | Способ очистки ацетона-сырца |
| DE102009000543A1 (de) | 2009-02-02 | 2010-08-12 | Evonik Degussa Gmbh | Verfahren, Absorptionsmedien und Vorrichtung zur Absorption von CO2 aus Gasmischungen |
| EP2243530A1 (de) * | 2009-04-21 | 2010-10-27 | LANXESS Deutschland GmbH | Verfahren zur Trennung aromatischer Verbindungen |
| DE102009047564A1 (de) | 2009-12-07 | 2011-06-09 | Evonik Degussa Gmbh | Arbeitsmedium für eine Absorptionskältemaschine |
| DE102011077377A1 (de) | 2010-11-12 | 2012-05-16 | Evonik Degussa Gmbh | Verfahren zur Absorption von sauren Gasen aus Gasmischungen |
| CA2855855A1 (en) | 2011-11-14 | 2013-05-23 | Evonik Degussa Gmbh | Method and device for the separation of acidic gases from a gas mixture |
| US9024079B2 (en) * | 2012-01-11 | 2015-05-05 | Lanxess Deutschland Gmbh | Separation of isomeric menthol compounds |
| DE102012200907A1 (de) | 2012-01-23 | 2013-07-25 | Evonik Industries Ag | Verfahren und Absorptionsmedium zur Absorption von CO2 aus einer Gasmischung |
| DE102012207509A1 (de) | 2012-05-07 | 2013-11-07 | Evonik Degussa Gmbh | Verfahren zur Absorption von CO2 aus einer Gasmischung |
| DE102015212749A1 (de) | 2015-07-08 | 2017-01-12 | Evonik Degussa Gmbh | Verfahren zur Entfeuchtung von feuchten Gasgemischen |
| NO20151452A1 (en) * | 2015-10-26 | 2017-04-27 | Norwegian Tech As | Method for separation of non-polar organic compounds from a material |
| DE102016210478A1 (de) | 2016-06-14 | 2017-12-14 | Evonik Degussa Gmbh | Verfahren zur Entfeuchtung von feuchten Gasgemischen |
| DE102016210481B3 (de) | 2016-06-14 | 2017-06-08 | Evonik Degussa Gmbh | Verfahren zum Reinigen einer ionischen Flüssigkeit |
| DE102016210484A1 (de) | 2016-06-14 | 2017-12-14 | Evonik Degussa Gmbh | Verfahren zur Entfeuchtung von feuchten Gasgemischen |
| DE102016210483A1 (de) | 2016-06-14 | 2017-12-14 | Evonik Degussa Gmbh | Verfahren und Absorptionsmittel zur Entfeuchtung von feuchten Gasgemischen |
| EP3257843A1 (en) | 2016-06-14 | 2017-12-20 | Evonik Degussa GmbH | Method of preparing a high purity imidazolium salt |
| EP3257568B1 (de) | 2016-06-14 | 2019-09-18 | Evonik Degussa GmbH | Verfahren zur entfeuchtung von feuchten gasgemischen mit ionischen flüssigkeiten |
| CN111631986B (zh) * | 2020-06-23 | 2021-09-17 | 赫禧(广州)生物科技有限公司 | 一种植物活性成分提纯方法及其在化妆品的应用 |
Citations (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2005016484A1 (de) * | 2003-08-05 | 2005-02-24 | Basf Aktiengesellschaft | Recycling von ionischen flüssigkeiten bei der extraktiv-destillation |
Family Cites Families (11)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US2080064A (en) * | 1935-10-25 | 1937-05-11 | Shell Dev | Distillation process for the purification of alcohols |
| US2357028A (en) * | 1943-08-04 | 1944-08-29 | Shell Dev | Solvent extraction process |
| JPS5424982B1 (enExample) * | 1969-06-06 | 1979-08-24 | ||
| US4726894A (en) * | 1985-12-27 | 1988-02-23 | The Foxboro Company | Distillation cut point control |
| DE3613975A1 (de) | 1986-04-25 | 1987-10-29 | Bayer Ag | Trennung von diastereomeren |
| CN1022291C (zh) * | 1992-04-17 | 1993-10-06 | 天津大学 | 吸附蒸馏分离方法 |
| JPH09165348A (ja) | 1995-12-15 | 1997-06-24 | Mitsubishi Chem Corp | シクロヘキセンの分離方法 |
| US5731453A (en) * | 1996-03-12 | 1998-03-24 | Ube Industries, Ltd. | Process for producing a diaryl carbonate |
| US6616831B1 (en) * | 1997-09-03 | 2003-09-09 | Gtc Technology Inc. | Aromatics separation process and method of retrofitting existing equipment for same |
| KR100846957B1 (ko) * | 2001-03-20 | 2008-07-17 | 바스프 에스이 | 선택성 첨가제로서의 이온성 액체를 이용한 근비등 또는 공비 혼합물의 분리 방법 |
| US8002953B2 (en) * | 2007-07-13 | 2011-08-23 | Amt International Inc. | Low-energy extractive distillation process for dehydration of aqueous ethanol |
-
2005
- 2005-03-22 DE DE102005013030A patent/DE102005013030A1/de not_active Withdrawn
-
2006
- 2006-03-21 JP JP2008502306A patent/JP2008538722A/ja active Pending
- 2006-03-21 AT AT06723567T patent/ATE530237T1/de active
- 2006-03-21 EP EP06723567A patent/EP1866045B1/de not_active Not-in-force
- 2006-03-21 WO PCT/EP2006/002553 patent/WO2006100023A1/de not_active Ceased
- 2006-03-21 US US11/909,343 patent/US8277615B2/en not_active Expired - Fee Related
- 2006-03-21 CN CN2006800178366A patent/CN101180112B/zh not_active Expired - Fee Related
Patent Citations (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2005016484A1 (de) * | 2003-08-05 | 2005-02-24 | Basf Aktiengesellschaft | Recycling von ionischen flüssigkeiten bei der extraktiv-destillation |
Cited By (3)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JP2010536571A (ja) * | 2007-08-31 | 2010-12-02 | ビーエーエスエフ ソシエタス・ヨーロピア | イオン性液体の蒸留 |
| JP2012518524A (ja) * | 2009-02-23 | 2012-08-16 | ビーエーエスエフ ソシエタス・ヨーロピア | 蒸留助剤の使用下でのイオン性液体の蒸留 |
| JP2013142081A (ja) * | 2012-01-12 | 2013-07-22 | Lanxess Deutschland Gmbh | メントール化合物異性体の分離 |
Also Published As
| Publication number | Publication date |
|---|---|
| WO2006100023A9 (de) | 2007-12-13 |
| US20080283383A1 (en) | 2008-11-20 |
| DE102005013030A1 (de) | 2006-09-28 |
| EP1866045B1 (de) | 2011-10-26 |
| CN101180112B (zh) | 2011-02-02 |
| WO2006100023A1 (de) | 2006-09-28 |
| EP1866045A1 (de) | 2007-12-19 |
| CN101180112A (zh) | 2008-05-14 |
| US8277615B2 (en) | 2012-10-02 |
| ATE530237T1 (de) | 2011-11-15 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| JP2008538722A (ja) | 難揮発性流体を精留精製する方法 | |
| CA2440528C (en) | Ionic liquids as selective additives for the separation of close-boiling or azeotropic mixtures | |
| EP1654046B1 (de) | Destillatives verfahren zur trennung von engsiedenden oder azeotropen gemischen unter verwendung von ionischen flüssigkeiten | |
| US20040188350A1 (en) | Purification or work-up of ionic liquids by means of adsorptive separation processes | |
| KR101116453B1 (ko) | 추출 증류에서 생성된 이온성 액체의 리사이클링 | |
| ES2284050T3 (es) | Procedimiento para la obtencion de halogenoalcanos a partir de alcoholes. | |
| US20080249307A1 (en) | Method for Producing Ethylene Amines | |
| EP1817090B1 (de) | Verfahren zur chemischen umsetzung und auftrennung eines gemisches in einer kolonne | |
| US20120067715A1 (en) | Method for purifying carboxylic acids containing halogen compounds |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| A521 | Request for written amendment filed |
Free format text: JAPANESE INTERMEDIATE CODE: A523 Effective date: 20090317 |
|
| A621 | Written request for application examination |
Free format text: JAPANESE INTERMEDIATE CODE: A621 Effective date: 20090317 |
|
| A977 | Report on retrieval |
Free format text: JAPANESE INTERMEDIATE CODE: A971007 Effective date: 20101025 |
|
| A131 | Notification of reasons for refusal |
Free format text: JAPANESE INTERMEDIATE CODE: A131 Effective date: 20101109 |
|
| A601 | Written request for extension of time |
Free format text: JAPANESE INTERMEDIATE CODE: A601 Effective date: 20110207 |
|
| A602 | Written permission of extension of time |
Free format text: JAPANESE INTERMEDIATE CODE: A602 Effective date: 20110215 |
|
| A02 | Decision of refusal |
Free format text: JAPANESE INTERMEDIATE CODE: A02 Effective date: 20110705 |