JP2007242496A - Nonaqueous electrolyte secondary battery - Google Patents
Nonaqueous electrolyte secondary battery Download PDFInfo
- Publication number
- JP2007242496A JP2007242496A JP2006065202A JP2006065202A JP2007242496A JP 2007242496 A JP2007242496 A JP 2007242496A JP 2006065202 A JP2006065202 A JP 2006065202A JP 2006065202 A JP2006065202 A JP 2006065202A JP 2007242496 A JP2007242496 A JP 2007242496A
- Authority
- JP
- Japan
- Prior art keywords
- mass
- battery
- electrolyte
- added
- except
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Granted
Links
Images
Classifications
-
- Y—GENERAL TAGGING OF NEW TECHNOLOGICAL DEVELOPMENTS; GENERAL TAGGING OF CROSS-SECTIONAL TECHNOLOGIES SPANNING OVER SEVERAL SECTIONS OF THE IPC; TECHNICAL SUBJECTS COVERED BY FORMER USPC CROSS-REFERENCE ART COLLECTIONS [XRACs] AND DIGESTS
- Y02—TECHNOLOGIES OR APPLICATIONS FOR MITIGATION OR ADAPTATION AGAINST CLIMATE CHANGE
- Y02E—REDUCTION OF GREENHOUSE GAS [GHG] EMISSIONS, RELATED TO ENERGY GENERATION, TRANSMISSION OR DISTRIBUTION
- Y02E60/00—Enabling technologies; Technologies with a potential or indirect contribution to GHG emissions mitigation
- Y02E60/10—Energy storage using batteries
Abstract
Description
本発明は、リチウムを吸蔵放出する電極と電解質とを備える非水電解質二次電池に関する。 The present invention relates to a non-aqueous electrolyte secondary battery including an electrode that occludes and releases lithium and an electrolyte.
リチウムイオン電池の充放電サイクル寿命特性を向上させるために、電解液の添加剤が数多く提案されている。例えば、負極に金属リチウムを用いた非水電解質二次電池において、リチウムと反応し難い不飽和の炭素−炭素結合を含む化合物としてビニルエチレンカーボネートを溶媒に用いることが提案されている(特許文献1参照)。また、負極に炭素材料を用いた非水電解質において、ビニルエチレンカーボネート誘導体を使用することが提案されている(特許文献2参照)。さらに、黒鉛系負極を用いたリチウム電池において、電解液の分解を抑制するために、ビニレンカーボネート及びその誘電体を含有する電解液が提案されている(特許文献3参照)。また、軽金属を用いたリチウム電池において、負極上での電解液の還元分解反応を抑制するために、電解液にフルオロエチレンカーボネートを添加することが提案されている(特許文献4参照)。
しかし、上述した環状炭酸エステル誘導体を電解液に添加した場合、充放電サイクル寿命特性は向上するが、充電状態の電池を高温で放置した際に正極上で環状炭酸エステル誘導体が酸化分解される際に炭酸ガスを発生するため、電池の内圧が上昇し、電池厚みが増大するという問題があった。さらに、環状炭酸エステル誘導体を過剰に添加した場合、正極及び負極上で環状炭酸エステル誘導体が分解される際に発生した分解物がセパレータの目詰まりを誘発したり、また、正極及び負極上にリチウムイオンの移動抵抗の高い被膜として存在するために、放電性能が低下し、充放電サイクル寿命特性(容量保持率)が低下するという問題があった。 However, when the above-mentioned cyclic carbonate derivative is added to the electrolyte, the charge / discharge cycle life characteristics are improved. However, when the charged battery is left at a high temperature, the cyclic carbonate derivative is oxidatively decomposed on the positive electrode. Since carbon dioxide gas is generated, the internal pressure of the battery rises and the battery thickness increases. Furthermore, when the cyclic carbonate derivative is added excessively, a decomposition product generated when the cyclic carbonate derivative is decomposed on the positive electrode and the negative electrode induces clogging of the separator, or lithium on the positive electrode and the negative electrode. Since it exists as a film having a high ion migration resistance, there is a problem that the discharge performance is lowered and the charge / discharge cycle life characteristic (capacity retention) is lowered.
上記問題を改善する方法として、上記環状炭酸エステル誘導体の添加量を減量することが考えられるが、環状炭酸エステル誘導体を減量した場合は負極被膜が十分に形成されず、充放電サイクルにともない負極上で電解液の分解が進行し、電解液が枯渇するため、充放電サイクル寿命特性が低下するという問題がある。逆に過剰に添加した場合は、負極の被膜抵抗が大きくなり、負極上に金属リチウムが析出したり、環状炭酸エステル誘導体の分解物によってセパレータの目詰まりが生じ易くなるので、充放電サイクル寿命特性が低下するという問題がある。 As a method for improving the above problem, it is conceivable to reduce the amount of the cyclic carbonate derivative added. However, when the amount of the cyclic carbonate derivative is reduced, the negative electrode film is not sufficiently formed, and the negative electrode film is formed on the negative electrode due to the charge / discharge cycle. However, since the decomposition of the electrolytic solution proceeds and the electrolytic solution is depleted, the charge / discharge cycle life characteristic is deteriorated. On the other hand, when added excessively, the film resistance of the negative electrode is increased, and metallic lithium is deposited on the negative electrode, or the separator is easily clogged by the decomposition product of the cyclic carbonate derivative. There is a problem that decreases.
本発明は斯かる事情に鑑みてなされたものであり、電解質に環状炭酸エステル誘導体と、ビニルエステル誘導体とを含有させることにより、充放電サイクル寿命特性を向上出来ると共に、電池を高温で保存した際の電池厚みの増大を抑制することが出来る非水電解質二次電池を提供することを目的とする。 The present invention has been made in view of such circumstances, and by adding a cyclic carbonate derivative and a vinyl ester derivative to the electrolyte, the charge / discharge cycle life characteristics can be improved and the battery can be stored at a high temperature. An object of the present invention is to provide a non-aqueous electrolyte secondary battery that can suppress an increase in battery thickness.
第1発明に係る非水電解質二次電池は、リチウムを吸蔵放出する電極と電解質とを備える非水電解質二次電池において、前記電解質は、下記化1で表される環状炭酸エステル誘導体、及び下記化2で表される環状炭酸エステル誘導体のうちの少なくとも1種の化合物と、ビニルエステル誘導体とを含んでなることを特徴とする。
A nonaqueous electrolyte secondary battery according to a first aspect of the present invention is a nonaqueous electrolyte secondary battery comprising an electrode that occludes and releases lithium and an electrolyte, wherein the electrolyte is a cyclic carbonate derivative represented by the following
(式中、R1 及びR2 は、それぞれ独立して水素原子、アルキル基、不飽和結合を含む炭化水素基、アルコキシ基、ハロゲン、ハロゲンを有するアルキル基、又はアリール基である。但し、R1及びR2 はエーテル結合を含んでもよい。) (In the formula, R 1 and R 2 are each independently a hydrogen atom, an alkyl group, a hydrocarbon group containing an unsaturated bond, an alkoxy group, a halogen, an alkyl group having a halogen, or an aryl group. 1 and R 2 may contain an ether bond.)
(式中、R3 はアルキル基、不飽和結合を含む炭化水素基、アルコキシ基、ハロゲン、ハロゲンを有するアルキル基、又はアリール基である。R4 は、水素原子、アルキル基、不飽和結合を含む炭化水素基、アルコキシ基、ハロゲン、ハロゲンを有するアルキル基、又はアリール基である。但し、R3 及び4 はエーテル結合を含んでもよい。化2はプロピレンカーボネートを除く。) (In the formula, R 3 is an alkyl group, a hydrocarbon group containing an unsaturated bond, an alkoxy group, a halogen, a halogen-containing alkyl group, or an aryl group. R 4 is a hydrogen atom, an alkyl group, or an unsaturated bond. A hydrocarbon group, an alkoxy group, a halogen, a halogen-containing alkyl group, or an aryl group, wherein R 3 and 4 may contain an ether bond (Chemical Formula 2 excludes propylene carbonate.)
第2発明に係る非水電解質二次電池は、第1発明において、前記環状炭酸エステル誘導体は、ビニレンカーボネート、ビニルエチレンカーボネート、エチニルエチレンカーボネート、ジビニルエチレンカーボネート、ビニロキシエチレンカーボネート、モノフルオロエチレンカーボネート及びモノクロロエチレンカーボネートのうちの少なくとも1つであることを特徴とする。 The nonaqueous electrolyte secondary battery according to a second invention is the non-aqueous electrolyte secondary battery according to the first invention, wherein the cyclic carbonate derivative is vinylene carbonate, vinyl ethylene carbonate, ethynyl ethylene carbonate, divinyl ethylene carbonate, vinyloxyethylene carbonate, monofluoroethylene carbonate, and It is at least one of monochloroethylene carbonate.
第3発明に係る非水電解質二次電池は、第1又は第2発明において、前記ビニルエステル誘導体は、酢酸ビニル、ジケテン、プロピオン酸ビニル、酪酸ビニル、カプロン酸ビニル、モノクロロ酢酸ビニル、モノフルオロ酸ビニル、メタクリル酸ビニル、クロトン酸ビニル、シクロヘキサンカルボン酸ビニル、安息香酸ビニル及び桂皮酸ビニルのうちの少なくとも1つであることを特徴とする。 The nonaqueous electrolyte secondary battery according to a third aspect of the present invention is the first or second aspect, wherein the vinyl ester derivative is vinyl acetate, diketene, vinyl propionate, vinyl butyrate, vinyl caproate, vinyl monochloroacetate, monofluoro acid. It is characterized by being at least one of vinyl, vinyl methacrylate, vinyl crotonate, vinyl cyclohexanecarboxylate, vinyl benzoate and vinyl cinnamate.
第4発明に係る非水電解質二次電池は、第1乃至第3発明の何れかにおいて、前記電解質の前記環状炭酸エステル誘導体の含有量は0.1質量%以上3質量%以下であることを特徴とする。 In the nonaqueous electrolyte secondary battery according to a fourth aspect of the present invention, in any one of the first to third aspects, the content of the cyclic carbonate derivative of the electrolyte is 0.1% by mass or more and 3% by mass or less. Features.
第5発明に係る非水電解質二次電池は、第1乃至第4発明の何れかにおいて、前記電解質の前記ビニルエステル誘導体の含有量は0.01質量%以上2質量%以下であることを特徴とする。 The nonaqueous electrolyte secondary battery according to a fifth aspect of the present invention is any one of the first to fourth aspects, wherein the content of the vinyl ester derivative in the electrolyte is 0.01% by mass or more and 2% by mass or less. And
第1発明においては、電解質に環状炭酸エステル誘導体と、環状又は鎖状のビニルエステル誘導体とを含有させた場合、環状炭酸エステル誘導体及びビニルエステル誘導体を混合した添加剤が還元分解することによって形成される負極被膜が電解液の分解を抑制し、初期充放電効率が高くなる。そのため、初期容量の低減を抑制することができ、初期容量の大きい電池が得られる。また、負極の被膜抵抗が大きく上がらないため、負極上の金属リチウムデンドライトが発生し難くなる。 In the first invention, when the cyclic carbonate derivative and the cyclic or chain vinyl ester derivative are contained in the electrolyte, the additive formed by mixing the cyclic carbonate derivative and the vinyl ester derivative is formed by reductive decomposition. The negative electrode coating suppresses the decomposition of the electrolytic solution and increases the initial charge / discharge efficiency. Therefore, a reduction in initial capacity can be suppressed, and a battery having a large initial capacity can be obtained. Moreover, since the film resistance of the negative electrode does not increase greatly, it is difficult to generate metallic lithium dendrite on the negative electrode.
また、ビニルエステル誘導体は環状の炭酸エステルよりも酸化分解を受け難く、また酸化分解物が気体でないため分解の際にガスの発生が少なく、高温放置時のガス発生を抑制することが出来る。さらに、ビニルエステル誘導体が負極表面に被膜を形成し、電解液成分が負極上で還元され、ガスを発生する反応を抑制することが出来る。また、ビニルエステル誘導体は、負極上の環状炭酸エステル誘導体の分解反応や、正負極上での電解液の分解反応を抑制するため、高温放置時のガス発生が少なく、電池厚みの増加を抑制でき、また充放電サイクル寿命特性も高くなる。 In addition, vinyl ester derivatives are less susceptible to oxidative degradation than cyclic carbonates, and since the oxidative decomposition product is not a gas, less gas is generated during decomposition, and gas generation when left at high temperatures can be suppressed. Furthermore, the vinyl ester derivative forms a film on the surface of the negative electrode, and the electrolyte component is reduced on the negative electrode, thereby suppressing a reaction that generates gas. In addition, the vinyl ester derivative suppresses the decomposition reaction of the cyclic carbonate derivative on the negative electrode and the decomposition reaction of the electrolyte solution on the positive and negative electrodes. Further, the charge / discharge cycle life characteristics are also improved.
また、ビニルエステル誘導体は、環状炭酸エステル誘導体と同様に負極上に被膜を形成し、負極上での電解液の分解を抑制するが、ビニルエステル誘導体単独又は環状炭酸エステル誘導体単独がそれぞれ形成する負極被膜よりも、ビニルエステル誘導体及び環状炭酸エステル誘導体を混合した場合に形成される負極被膜の方が安定であり、相乗効果によって充放電サイクル寿命特性がさらに向上する。 Further, the vinyl ester derivative forms a film on the negative electrode similarly to the cyclic carbonate derivative and suppresses the decomposition of the electrolytic solution on the negative electrode, but the negative electrode formed by the vinyl ester derivative alone or the cyclic carbonate derivative alone, respectively. The negative electrode film formed when a vinyl ester derivative and a cyclic carbonate derivative are mixed is more stable than the film, and the charge / discharge cycle life characteristics are further improved by a synergistic effect.
第2発明においては、前記環状炭酸エステル誘導体として、ビニレンカーボネート、ビニルエチレンカーボネート、エチニルエチレンカーボネート、ジビニルエチレンカーボネート、ビニロキシエチレンカーボネート、モノフルオロエチレンカーボネート又はモノクロロエチレンカーボネートを用いる。 In the second invention, vinylene carbonate, vinyl ethylene carbonate, ethynyl ethylene carbonate, divinyl ethylene carbonate, vinyloxy ethylene carbonate, monofluoroethylene carbonate or monochloroethylene carbonate is used as the cyclic carbonate derivative.
第3発明においては、前記ビニルエステル誘導体として酢酸ビニル、ジケテン、プロピオン酸ビニル、酪酸ビニル、カプロン酸ビニル、モノクロロ酢酸ビニル、モノフルオロ酸ビニル、メタクリル酸ビニル、クロトン酸ビニル、シクロヘキサンカルボン酸ビニル、安息香酸ビニル、桂皮酸ビニルを用いる。 In the third invention, the vinyl ester derivative is vinyl acetate, diketene, vinyl propionate, vinyl butyrate, vinyl caproate, vinyl monochloroacetate, vinyl monofluoroate, vinyl methacrylate, vinyl crotonic acid, vinyl cyclohexanecarboxylate, benzoic acid. Vinyl acid and vinyl cinnamate are used.
第4発明においては、電解質の環状炭酸エステル誘導体の含有量を0.1質量%以上3質量%以下にすると、充放電サイクル寿命特性を向上させ、高温放置時の電池厚みの増加を抑制することが出来ると共に、初期容量の低下を抑制することが出来る。環状炭酸エステル誘導体の含有量が0.1質量%未満の場合は、充放電サイクル寿命特性が向上せず、3質量%を超える場合は、初期容量が低下し、電池厚みが増加し、放電性能が低下する。 In the fourth invention, when the content of the cyclic carbonate derivative of the electrolyte is 0.1% by mass or more and 3% by mass or less, the charge / discharge cycle life characteristics are improved, and the increase in the battery thickness when left at high temperature is suppressed. And a decrease in the initial capacity can be suppressed. When the content of the cyclic carbonate derivative is less than 0.1% by mass, the charge / discharge cycle life characteristics are not improved. When the content exceeds 3% by mass, the initial capacity decreases, the battery thickness increases, and the discharge performance. Decreases.
第5発明においては、電解質のビニルエステル誘導体の含有量が0.01質量%以上2質量%以下であるため、充放電サイクル寿命特性を向上させ、高温放置時の電池厚みの増加を抑制することが出来ると共に、初期容量の低下を抑制することが出来る。ビニルエステル誘導体の含有量が0.01質量%未満の場合は、電池厚みが増加、放電性能が低下し、2質量%を超える場合は、初期容量が低下する。 In the fifth invention, since the content of the vinyl ester derivative of the electrolyte is 0.01% by mass or more and 2% by mass or less, the charge / discharge cycle life characteristics are improved, and an increase in battery thickness when left at high temperature is suppressed. And a decrease in the initial capacity can be suppressed. When the content of the vinyl ester derivative is less than 0.01% by mass, the battery thickness increases and the discharge performance decreases, and when it exceeds 2% by mass, the initial capacity decreases.
本発明によれば、充放電サイクル寿命特性(容量保持率)を向上出来ると共に、電池を高温で保存した際の電池厚みの増加を抑制することが出来る。 ADVANTAGE OF THE INVENTION According to this invention, while being able to improve a charge / discharge cycle life characteristic (capacity retention), the increase in battery thickness at the time of storing a battery at high temperature can be suppressed.
以下、好適な実施例を用いて本発明を説明するが、本発明は本実施例により何ら制限されるものではなく、その主旨を変更しない範囲において、適宜変更して実施することが出来る。
(実施例1)
図1は本発明に係る非水電解質二次電池の例を示す概略断面図である。図1において、1は非水電解質二次電池(以下、電池という)、2は電極群、3は負極、4は正極、5はセパレータ、6は電池ケース、7は電池蓋、8は安全弁、9は負極端子、10は負極リードである。電極群2は、負極3と正極4とをセパレータ5を介して扁平状に巻回したものである。電極群2は電池ケース6に収納してあり、電池ケース6の開口部は、安全弁8及び負極端子9が設けられた電池蓋7をレーザ溶接して密閉している。負極端子9は負極リード10と接続され、正極4は電池ケース6内面と接続されている。電池1は角型であり、厚みは4.2mmである。
Hereinafter, the present invention will be described with reference to preferred examples. However, the present invention is not limited to the examples, and can be appropriately modified and implemented without departing from the spirit of the present invention.
Example 1
FIG. 1 is a schematic cross-sectional view showing an example of a nonaqueous electrolyte secondary battery according to the present invention. In FIG. 1, 1 is a non-aqueous electrolyte secondary battery (hereinafter referred to as a battery), 2 is an electrode group, 3 is a negative electrode, 4 is a positive electrode, 5 is a separator, 6 is a battery case, 7 is a battery lid, 8 is a safety valve, 9 is a negative electrode terminal, and 10 is a negative electrode lead. The
正極4については、活物質としてLiCoO2 94質量%と、導電助剤としてアセチレンブラック3質量%と、結着剤としてポリフッ化ビニリデン(PVDF)3質量%とを混合した正極合剤を、N−メチル−2−ピロリドン(NMP)に分散させることによりペーストを調製した。このペーストを厚さ20μmのアルミニウム集電体に均一に塗布し、乾燥させた後、ロールプレスで圧縮形成することにより正極4を作製した。
For the
負極3については、活物質として黒鉛95質量%と、結着剤としてカルボキシメチルセルロース3質量%、スチレンブタジエンゴム2質量%とを混合し、蒸留水を適宜加えて分散させ、スラリーを調製した。このスラリーを厚さ15μmの銅集電体に均一に塗布、乾燥させた後、100℃で5時間乾燥させ、ロールプレスで圧縮形成することにより負極3を作製した。
Regarding the
セパレータ5は、厚さ20μmの微多孔性ポリエチレンフィルムを用いた。電解質は、エチレンカーボネート(EC)とエチルメチルカーボネート(EMC)とジエチルカーボネート(DEC)との体積比3:2:5混合溶媒にLiPF6 を1.1mol/L溶解させた電解質に、さらに電解質の総量に対してビニレンカーボネート(VC(1,3-dioxolen-2-one)、化3参照)を0.1質量%、ジケテン(DK(4-methyleneoxetan-2-one)、化4参照)を0.01質量%添加したものを用いた。
As the
(実施例2)
電解質の総量に対してVCを0.5質量%、DKを0.01質量%添加し、それ以外は実施例1と同様の電池を作製した。
(実施例3)
電解質の総量に対してVCを1質量%、DKを0.01質量%添加し、それ以外は実施例1と同様の電池を作製した。
(実施例4)
電解質の総量に対してVCを2質量%、DKを0.01質量%添加し、それ以外は実施例1と同様の電池を作製した。
(実施例5)
電解質の総量に対してVCを3質量%、DKを0.01質量%添加し、それ以外は実施例1と同様の電池を作製した。
(実施例6)
電解質の総量に対してVCを5質量%、DKを0.01質量%添加し、それ以外は実施例1と同様の電池を作製した。
(Example 2)
A battery was manufactured in the same manner as in Example 1 except that 0.5% by mass of VC and 0.01% by mass of DK were added to the total amount of the electrolyte.
(Example 3)
A battery was manufactured in the same manner as in Example 1 except that 1% by mass of VC and 0.01% by mass of DK were added to the total amount of the electrolyte.
Example 4
A battery was manufactured in the same manner as in Example 1 except that 2% by mass of VC and 0.01% by mass of DK were added to the total amount of the electrolyte.
(Example 5)
A battery was manufactured in the same manner as in Example 1 except that 3% by mass of VC and 0.01% by mass of DK were added to the total amount of the electrolyte.
(Example 6)
A battery was manufactured in the same manner as in Example 1 except that 5% by mass of VC and 0.01% by mass of DK were added to the total amount of the electrolyte.
(実施例7)
電解質の総量に対してVCを0.1質量%、DKを0.1質量%添加し、それ以外は実施例1と同様の電池を作製した。
(実施例8)
電解質の総量に対してVCを0.5質量%、DKを0.1質量%添加し、それ以外は実施例1と同様の電池を作製した。
(実施例9)
電解質の総量に対してVCを1質量%、DKを0.1質量%添加し、それ以外は実施例1と同様の電池を作製した。
(実施例10)
電解質の総量に対してVCを2質量%、DKを0.1質量%添加し、それ以外は実施例1と同様の電池を作製した。
(実施例11)
電解質の総量に対してVCを3質量%、DKを0.1質量%添加し、それ以外は実施例1と同様の電池を作製した。
(実施例12)
電解質の総量に対してVCを5質量%、DKを0.1質量%添加し、それ以外は実施例1と同様の電池を作製した。
(Example 7)
A battery was manufactured in the same manner as in Example 1 except that 0.1% by mass of VC and 0.1% by mass of DK were added to the total amount of the electrolyte.
(Example 8)
A battery was manufactured in the same manner as in Example 1 except that 0.5% by mass of VC and 0.1% by mass of DK were added to the total amount of the electrolyte.
Example 9
A battery was manufactured in the same manner as in Example 1 except that 1% by mass of VC and 0.1% by mass of DK were added to the total amount of the electrolyte.
(Example 10)
A battery was manufactured in the same manner as in Example 1 except that 2% by mass of VC and 0.1% by mass of DK were added to the total amount of the electrolyte.
(Example 11)
A battery was manufactured in the same manner as in Example 1 except that 3% by mass of VC and 0.1% by mass of DK were added to the total amount of the electrolyte.
(Example 12)
A battery was manufactured in the same manner as in Example 1 except that 5% by mass of VC and 0.1% by mass of DK were added to the total amount of the electrolyte.
(実施例13)
電解質の総量に対してVCを0.1質量%、DKを0.5質量%添加し、それ以外は実施例1と同様の電池を作製した。
(実施例14)
電解質の総量に対してVCを0.5質量%、DKを0.5質量%添加し、それ以外は実施例1と同様の電池を作製した。
(実施例15)
電解質の総量に対してVCを1質量%、DKを0.5質量%添加し、それ以外は実施例1と同様の電池を作製した。
(実施例16)
電解質の総量に対してVCを2質量%、DKを0.5質量%添加し、それ以外は実施例1と同様の電池を作製した。
(実施例17)
電解質の総量に対してVCを3質量%、DKを0.5質量%添加し、それ以外は実施例1と同様の電池を作製した。
(実施例18)
電解質の総量に対してVCを5質量%、DKを0.5質量%添加し、それ以外は実施例1と同様の電池を作製した。
(Example 13)
A battery was manufactured in the same manner as in Example 1 except that 0.1% by mass of VC and 0.5% by mass of DK were added to the total amount of the electrolyte.
(Example 14)
A battery was manufactured in the same manner as in Example 1 except that 0.5% by mass of VC and 0.5% by mass of DK were added to the total amount of the electrolyte.
(Example 15)
A battery was manufactured in the same manner as in Example 1 except that 1% by mass of VC and 0.5% by mass of DK were added to the total amount of the electrolyte.
(Example 16)
A battery was manufactured in the same manner as in Example 1 except that 2% by mass of VC and 0.5% by mass of DK were added to the total amount of the electrolyte.
(Example 17)
A battery was manufactured in the same manner as in Example 1 except that 3% by mass of VC and 0.5% by mass of DK were added to the total amount of the electrolyte.
(Example 18)
A battery was manufactured in the same manner as in Example 1 except that 5% by mass of VC and 0.5% by mass of DK were added to the total amount of the electrolyte.
(実施例19)
電解質の総量に対してVCを0.1質量%、DKを1質量%添加し、それ以外は実施例1と同様の電池を作製した。
(実施例20)
電解質の総量に対してVCを0.5質量%、DKを1質量%添加し、それ以外は実施例1と同様の電池を作製した。
(実施例21)
電解質の総量に対してVCを1質量%、DKを1質量%添加し、それ以外は実施例1と同様の電池を作製した。
(実施例22)
電解質の総量に対してVCを2質量%、DKを1質量%添加し、それ以外は実施例1と同様の電池を作製した。
(実施例23)
電解質の総量に対してVCを3質量%、DKを1質量%添加し、それ以外は実施例1と同様の電池を作製した。
(実施例24)
電解質の総量に対してVCを5質量%、DKを1質量%添加し、それ以外は実施例1と同様の電池を作製した。
Example 19
A battery was manufactured in the same manner as in Example 1 except that 0.1% by mass of VC and 1% by mass of DK were added to the total amount of the electrolyte.
(Example 20)
A battery was manufactured in the same manner as in Example 1 except that 0.5% by mass of VC and 1% by mass of DK were added to the total amount of the electrolyte.
(Example 21)
A battery was manufactured in the same manner as in Example 1 except that 1% by mass of VC and 1% by mass of DK were added to the total amount of the electrolyte.
(Example 22)
A battery was manufactured in the same manner as in Example 1 except that 2% by mass of VC and 1% by mass of DK were added to the total amount of the electrolyte.
(Example 23)
A battery was manufactured in the same manner as in Example 1 except that 3% by mass of VC and 1% by mass of DK were added to the total amount of the electrolyte.
(Example 24)
A battery was manufactured in the same manner as in Example 1 except that 5% by mass of VC and 1% by mass of DK were added to the total amount of the electrolyte.
(実施例25)
電解質の総量に対してVCを0.1質量%、DKを2質量%添加し、それ以外は実施例1と同様の電池を作製した。
(実施例26)
電解質の総量に対してVCを0.5質量%、DKを2質量%添加し、それ以外は実施例1と同様の電池を作製した。
(実施例27)
電解質の総量に対してVCを1質量%、DKを2質量%添加し、それ以外は実施例1と同様の電池を作製した。
(実施例28)
電解質の総量に対してVCを2質量%、DKを2質量%添加し、それ以外は実施例1と同様の電池を作製した。
(実施例29)
電解質の総量に対してVCを3質量%、DKを2質量%添加し、それ以外は実施例1と同様の電池を作製した。
(実施例30)
電解質の総量に対してVCを5質量%、DKを2質量%添加し、それ以外は実施例1と同様の電池を作製した。
(Example 25)
A battery was manufactured in the same manner as in Example 1 except that 0.1% by weight of VC and 2% by weight of DK were added to the total amount of the electrolyte.
(Example 26)
A battery was manufactured in the same manner as in Example 1 except that 0.5% by mass of VC and 2% by mass of DK were added to the total amount of the electrolyte.
(Example 27)
A battery was manufactured in the same manner as in Example 1 except that 1% by mass of VC and 2% by mass of DK were added to the total amount of the electrolyte.
(Example 28)
A battery was manufactured in the same manner as in Example 1 except that 2% by mass of VC and 2% by mass of DK were added to the total amount of the electrolyte.
(Example 29)
A battery was manufactured in the same manner as in Example 1 except that 3% by mass of VC and 2% by mass of DK were added to the total amount of the electrolyte.
(Example 30)
A battery was manufactured in the same manner as in Example 1 except that 5% by mass of VC and 2% by mass of DK were added to the total amount of the electrolyte.
(実施例31)
電解質の総量に対してVCを0.1質量%、DKを3質量%添加し、それ以外は実施例1と同様の電池を作製した。
(実施例32)
電解質の総量に対してVCを0.5質量%、DKを3質量%添加し、それ以外は実施例1と同様の電池を作製した。
(実施例33)
電解質の総量に対してVCを1質量%、DKを3質量%添加し、それ以外は実施例1と同様の電池を作製した。
(実施例34)
電解質の総量に対してVCを2質量%、DKを3質量%添加し、それ以外は実施例1と同様の電池を作製した。
(実施例35)
電解質の総量に対してVCを3質量%、DKを3質量%添加し、それ以外は実施例1と同様の電池を作製した。
(実施例36)
電解質の総量に対してVCを5質量%、DKを3質量%添加し、それ以外は実施例1と同様の電池を作製した。
(Example 31)
A battery was manufactured in the same manner as in Example 1 except that 0.1% by mass of VC and 3% by mass of DK were added to the total amount of the electrolyte.
(Example 32)
A battery was manufactured in the same manner as in Example 1 except that 0.5% by mass of VC and 3% by mass of DK were added to the total amount of the electrolyte.
(Example 33)
A battery was manufactured in the same manner as in Example 1 except that 1% by mass of VC and 3% by mass of DK were added to the total amount of the electrolyte.
(Example 34)
A battery was manufactured in the same manner as in Example 1 except that 2% by mass of VC and 3% by mass of DK were added to the total amount of the electrolyte.
(Example 35)
A battery was manufactured in the same manner as in Example 1 except that 3% by mass of VC and 3% by mass of DK were added to the total amount of the electrolyte.
(Example 36)
A battery was manufactured in the same manner as in Example 1 except that 5% by mass of VC and 3% by mass of DK were added to the total amount of the electrolyte.
(実施例37)
電解質の総量に対してVCを0.1質量%、酢酸ビニル(VA(vinyl acetate)、化5参照)を0.01質量%添加し、それ以外は実施例1と同様の電池を作製した。
(Example 37)
A battery was manufactured in the same manner as in Example 1 except that 0.1% by mass of VC and 0.01% by mass of vinyl acetate (VA (vinyl acetate), see Chemical formula 5) were added to the total amount of the electrolyte.
(実施例38)
電解質の総量に対してVCを0.5質量%、VAを0.01質量%添加し、それ以外は実施例1と同様の電池を作製した。
(実施例39)
電解質の総量に対してVCを1質量%、VAを0.01質量%添加し、それ以外は実施例1と同様の電池を作製した。
(実施例40)
電解質の総量に対してVCを2質量%、VAを0.01質量%添加し、それ以外は実施例1と同様の電池を作製した。
(実施例41)
電解質の総量に対してVCを3質量%、VAを0.01質量%添加し、それ以外は実施例1と同様の電池を作製した。
(実施例42)
電解質の総量に対してVCを5質量%、VAを0.01質量%添加し、それ以外は実施例1と同様の電池を作製した。
(Example 38)
A battery was manufactured in the same manner as in Example 1 except that 0.5% by mass of VC and 0.01% by mass of VA were added to the total amount of the electrolyte.
(Example 39)
A battery was manufactured in the same manner as in Example 1 except that 1% by mass of VC and 0.01% by mass of VA were added to the total amount of the electrolyte.
(Example 40)
A battery was manufactured in the same manner as in Example 1 except that 2% by mass of VC and 0.01% by mass of VA were added to the total amount of the electrolyte.
(Example 41)
A battery was manufactured in the same manner as in Example 1 except that 3% by mass of VC and 0.01% by mass of VA were added to the total amount of the electrolyte.
(Example 42)
A battery was manufactured in the same manner as in Example 1 except that 5% by mass of VC and 0.01% by mass of VA were added to the total amount of the electrolyte.
(実施例43)
電解質の総量に対してVCを0.1質量%、VAを0.1質量%添加し、それ以外は実施例1と同様の電池を作製した。
(実施例44)
電解質の総量に対してVCを0.5質量%、VAを0.1質量%添加し、それ以外は実施例1と同様の電池を作製した。
(実施例45)
電解質の総量に対してVCを1質量%、VAを0.1質量%添加し、それ以外は実施例1と同様の電池を作製した。
(実施例46)
電解質の総量に対してVCを2質量%、VAを0.1質量%添加し、それ以外は実施例1と同様の電池を作製した。
(実施例47)
電解質の総量に対してVCを3質量%、VAを0.1質量%添加し、それ以外は実施例1と同様の電池を作製した。
(実施例48)
電解質の総量に対してVCを5質量%、VAを0.1質量%添加し、それ以外は実施例1と同様の電池を作製した。
(Example 43)
A battery was manufactured in the same manner as in Example 1 except that 0.1% by mass of VC and 0.1% by mass of VA were added to the total amount of the electrolyte.
(Example 44)
A battery was manufactured in the same manner as in Example 1 except that 0.5% by mass of VC and 0.1% by mass of VA were added to the total amount of the electrolyte.
(Example 45)
A battery was manufactured in the same manner as in Example 1 except that 1% by mass of VC and 0.1% by mass of VA were added to the total amount of the electrolyte.
(Example 46)
A battery was manufactured in the same manner as in Example 1 except that 2% by mass of VC and 0.1% by mass of VA were added to the total amount of the electrolyte.
(Example 47)
A battery was manufactured in the same manner as in Example 1 except that 3% by mass of VC and 0.1% by mass of VA were added to the total amount of the electrolyte.
(Example 48)
A battery was manufactured in the same manner as in Example 1 except that 5% by mass of VC and 0.1% by mass of VA were added to the total amount of the electrolyte.
(実施例49)
電解質の総量に対してVCを0.1質量%、VAを0.5質量%添加し、それ以外は実施例1と同様の電池を作製した。
(実施例50)
電解質の総量に対してVCを0.5質量%、VAを0.5質量%添加し、それ以外は実施例1と同様の電池を作製した。
(実施例51)
電解質の総量に対してVCを1質量%、VAを0.5質量%添加し、それ以外は実施例1と同様の電池を作製した。
(実施例52)
電解質の総量に対してVCを2質量%、VAを0.5質量%添加し、それ以外は実施例1と同様の電池を作製した。
(実施例53)
電解質の総量に対してVCを3質量%、VAを0.5質量%添加し、それ以外は実施例1と同様の電池を作製した。
(実施例54)
電解質の総量に対してVCを5質量%、VAを0.5質量%添加し、それ以外は実施例1と同様の電池を作製した。
(Example 49)
A battery was manufactured in the same manner as in Example 1 except that 0.1 mass% of VC and 0.5 mass% of VA were added to the total amount of the electrolyte.
(Example 50)
A battery was manufactured in the same manner as in Example 1 except that 0.5% by mass of VC and 0.5% by mass of VA were added to the total amount of the electrolyte.
(Example 51)
A battery was manufactured in the same manner as in Example 1 except that 1% by mass of VC and 0.5% by mass of VA were added to the total amount of the electrolyte.
(Example 52)
A battery was manufactured in the same manner as in Example 1 except that 2% by mass of VC and 0.5% by mass of VA were added to the total amount of the electrolyte.
(Example 53)
A battery was manufactured in the same manner as in Example 1 except that 3% by mass of VC and 0.5% by mass of VA were added to the total amount of the electrolyte.
(Example 54)
A battery was manufactured in the same manner as in Example 1 except that 5% by mass of VC and 0.5% by mass of VA were added to the total amount of the electrolyte.
(実施例55)
電解質の総量に対してVCを0.1質量%、VAを1質量%添加し、それ以外は実施例1と同様の電池を作製した。
(実施例56)
電解質の総量に対してVCを0.5質量%、VAを1質量%添加し、それ以外は実施例1と同様の電池を作製した。
(実施例57)
電解質の総量に対してVCを1質量%、VAを1質量%添加し、それ以外は実施例1と同様の電池を作製した。
(実施例58)
電解質の総量に対してVCを2質量%、VAを1質量%添加し、それ以外は実施例1と同様の電池を作製した。
(実施例59)
電解質の総量に対してVCを3質量%、VAを1質量%添加し、それ以外は実施例1と同様の電池を作製した。
(実施例60)
電解質の総量に対してVCを5質量%、VAを1質量%添加し、それ以外は実施例1と同様の電池を作製した。
(Example 55)
A battery was manufactured in the same manner as in Example 1 except that 0.1% by weight of VC and 1% by weight of VA were added to the total amount of the electrolyte.
(Example 56)
A battery was manufactured in the same manner as in Example 1 except that 0.5% by mass of VC and 1% by mass of VA were added to the total amount of the electrolyte.
(Example 57)
A battery was manufactured in the same manner as in Example 1 except that 1% by mass of VC and 1% by mass of VA were added to the total amount of the electrolyte.
(Example 58)
A battery was manufactured in the same manner as in Example 1 except that 2% by mass of VC and 1% by mass of VA were added to the total amount of the electrolyte.
(Example 59)
A battery was manufactured in the same manner as in Example 1 except that 3% by mass of VC and 1% by mass of VA were added to the total amount of the electrolyte.
(Example 60)
A battery was manufactured in the same manner as in Example 1 except that 5% by mass of VC and 1% by mass of VA were added to the total amount of the electrolyte.
(実施例61)
電解質の総量に対してVCを0.1質量%、VAを2質量%添加し、それ以外は実施例1と同様の電池を作製した。
(実施例62)
電解質の総量に対してVCを0.5質量%、VAを2質量%添加し、それ以外は実施例1と同様の電池を作製した。
(実施例63)
電解質の総量に対してVCを1質量%、VAを2質量%添加し、それ以外は実施例1と同様の電池を作製した。
(実施例64)
電解質の総量に対してVCを2質量%、VAを2質量%添加し、それ以外は実施例1と同様の電池を作製した。
(実施例65)
電解質の総量に対してVCを3質量%、VAを2質量%添加し、それ以外は実施例1と同様の電池を作製した。
(実施例66)
電解質の総量に対してVCを5質量%、VAを2質量%添加し、それ以外は実施例1と同様の電池を作製した。
(Example 61)
A battery was manufactured in the same manner as in Example 1 except that 0.1% by weight of VC and 2% by weight of VA were added to the total amount of the electrolyte.
(Example 62)
A battery was manufactured in the same manner as in Example 1 except that 0.5% by mass of VC and 2% by mass of VA were added to the total amount of the electrolyte.
(Example 63)
A battery was manufactured in the same manner as in Example 1 except that 1% by mass of VC and 2% by mass of VA were added to the total amount of the electrolyte.
(Example 64)
A battery was manufactured in the same manner as in Example 1 except that 2% by mass of VC and 2% by mass of VA were added to the total amount of the electrolyte.
(Example 65)
A battery was manufactured in the same manner as in Example 1 except that 3% by mass of VC and 2% by mass of VA were added to the total amount of the electrolyte.
Example 66
A battery was manufactured in the same manner as in Example 1 except that 5% by mass of VC and 2% by mass of VA were added to the total amount of the electrolyte.
(実施例67)
電解質の総量に対してVCを0.1質量%、VAを3質量%添加し、それ以外は実施例1と同様の電池を作製した。
(実施例68)
電解質の総量に対してVCを0.5質量%、VAを3質量%添加し、それ以外は実施例1と同様の電池を作製した。
(実施例69)
電解質の総量に対してVCを1質量%、VAを3質量%添加し、それ以外は実施例1と同様の電池を作製した。
(実施例70)
電解質の総量に対してVCを2質量%、VAを3質量%添加し、それ以外は実施例1と同様の電池を作製した。
(実施例71)
電解質の総量に対してVCを3質量%、VAを3質量%添加し、それ以外は実施例1と同様の電池を作製した。
(実施例72)
電解質の総量に対してVCを5質量%、VAを3質量%添加し、それ以外は実施例1と同様の電池を作製した。
(Example 67)
A battery was manufactured in the same manner as in Example 1 except that 0.1% by mass of VC and 3% by mass of VA were added to the total amount of the electrolyte.
(Example 68)
A battery was manufactured in the same manner as in Example 1 except that 0.5% by mass of VC and 3% by mass of VA were added to the total amount of the electrolyte.
(Example 69)
A battery was manufactured in the same manner as in Example 1 except that 1% by mass of VC and 3% by mass of VA were added to the total amount of the electrolyte.
(Example 70)
A battery was manufactured in the same manner as in Example 1 except that 2% by mass of VC and 3% by mass of VA were added to the total amount of the electrolyte.
(Example 71)
A battery was manufactured in the same manner as in Example 1 except that 3% by mass of VC and 3% by mass of VA were added to the total amount of the electrolyte.
(Example 72)
A battery was manufactured in the same manner as in Example 1 except that 5% by mass of VC and 3% by mass of VA were added to the total amount of the electrolyte.
(実施例73)
電解質の総量に対してモノフルオロエチレンカーボネート(FEC(4-fluoro-1,3-dioxolan-2-one)、化6参照)を0.1質量%、DKを0.01質量%添加し、それ以外は実施例1と同様の電池を作製した。
(Example 73)
0.1% by mass of monofluoroethylene carbonate (FEC (4-fluoro-1,3-dioxolan-2-one), see Chemical formula 6) and 0.01% by mass of DK are added to the total amount of the electrolyte. A battery was prepared in the same manner as in Example 1 except for the above.
(実施例74)
電解質の総量に対してFECを0.5質量%、DKを0.01質量%添加し、それ以外は実施例1と同様の電池を作製した。
(実施例75)
電解質の総量に対してFECを1質量%、DKを0.01質量%添加し、それ以外は実施例1と同様の電池を作製した。
(実施例76)
電解質の総量に対してFECを2質量%、DKを0.01質量%添加し、それ以外は実施例1と同様の電池を作製した。
(実施例77)
電解質の総量に対してFECを3質量%、DKを0.01質量%添加し、それ以外は実施例1と同様の電池を作製した。
(実施例78)
電解質の総量に対してFECを5質量%、DKを0.01質量%添加し、それ以外は実施例1と同様の電池を作製した。
(Example 74)
A battery was manufactured in the same manner as in Example 1 except that 0.5% by mass of FEC and 0.01% by mass of DK were added to the total amount of the electrolyte.
(Example 75)
A battery was manufactured in the same manner as in Example 1 except that 1% by mass of FEC and 0.01% by mass of DK were added to the total amount of the electrolyte.
(Example 76)
A battery was manufactured in the same manner as in Example 1 except that 2% by mass of FEC and 0.01% by mass of DK were added to the total amount of the electrolyte.
(Example 77)
A battery was manufactured in the same manner as in Example 1 except that 3% by mass of FEC and 0.01% by mass of DK were added to the total amount of the electrolyte.
(Example 78)
A battery was manufactured in the same manner as in Example 1 except that 5% by mass of FEC and 0.01% by mass of DK were added to the total amount of the electrolyte.
(実施例79)
電解質の総量に対してFECを0.1質量%、DKを0.1質量%添加し、それ以外は実施例1と同様の電池を作製した。
(実施例80)
電解質の総量に対してFECを0.5質量%、DKを0.1質量%添加し、それ以外は実施例1と同様の電池を作製した。
(実施例81)
電解質の総量に対してFECを1質量%、DKを0.1質量%添加し、それ以外は実施例1と同様の電池を作製した。
(実施例82)
電解質の総量に対してFECを2質量%、DKを0.1質量%添加し、それ以外は実施例1と同様の電池を作製した。
(実施例83)
電解質の総量に対してFECを3質量%、DKを0.1質量%添加し、それ以外は実施例1と同様の電池を作製した。
(実施例84)
電解質の総量に対してFECを5質量%、DKを0.1質量%添加し、それ以外は実施例1と同様の電池を作製した。
(Example 79)
A battery was manufactured in the same manner as in Example 1 except that 0.1% by mass of FEC and 0.1% by mass of DK were added to the total amount of the electrolyte.
(Example 80)
A battery was manufactured in the same manner as in Example 1 except that 0.5% by mass of FEC and 0.1% by mass of DK were added to the total amount of the electrolyte.
(Example 81)
A battery was manufactured in the same manner as in Example 1 except that 1% by mass of FEC and 0.1% by mass of DK were added to the total amount of the electrolyte.
(Example 82)
A battery was manufactured in the same manner as in Example 1 except that 2% by mass of FEC and 0.1% by mass of DK were added to the total amount of the electrolyte.
(Example 83)
A battery was manufactured in the same manner as in Example 1 except that 3% by mass of FEC and 0.1% by mass of DK were added to the total amount of the electrolyte.
(Example 84)
A battery was manufactured in the same manner as in Example 1 except that 5% by mass of FEC and 0.1% by mass of DK were added to the total amount of the electrolyte.
(実施例85)
電解質の総量に対してFECを0.1質量%、DKを0.5質量%添加し、それ以外は実施例1と同様の電池を作製した。
(実施例86)
電解質の総量に対してFECを0.5質量%、DKを0.5質量%添加し、それ以外は実施例1と同様の電池を作製した。
(実施例87)
電解質の総量に対してFECを1質量%、DKを0.5質量%添加し、それ以外は実施例1と同様の電池を作製した。
(実施例88)
電解質の総量に対してFECを2質量%、DKを0.5質量%添加し、それ以外は実施例1と同様の電池を作製した。
(実施例89)
電解質の総量に対してFECを3質量%、DKを0.5質量%添加し、それ以外は実施例1と同様の電池を作製した。
(実施例90)
電解質の総量に対してFECを5質量%、DKを0.5質量%添加し、それ以外は実施例1と同様の電池を作製した。
(Example 85)
A battery was manufactured in the same manner as in Example 1 except that 0.1% by mass of FEC and 0.5% by mass of DK were added to the total amount of the electrolyte.
(Example 86)
A battery was manufactured in the same manner as in Example 1 except that 0.5% by mass of FEC and 0.5% by mass of DK were added to the total amount of the electrolyte.
(Example 87)
A battery was manufactured in the same manner as in Example 1 except that 1% by mass of FEC and 0.5% by mass of DK were added to the total amount of the electrolyte.
(Example 88)
A battery was manufactured in the same manner as in Example 1 except that 2% by mass of FEC and 0.5% by mass of DK were added to the total amount of the electrolyte.
Example 89
A battery was manufactured in the same manner as in Example 1 except that 3% by mass of FEC and 0.5% by mass of DK were added to the total amount of the electrolyte.
(Example 90)
A battery was manufactured in the same manner as in Example 1 except that 5% by mass of FEC and 0.5% by mass of DK were added to the total amount of the electrolyte.
(実施例91)
電解質の総量に対してFECを0.1質量%、DKを1質量%添加し、それ以外は実施例1と同様の電池を作製した。
(実施例92)
電解質の総量に対してFECを0.5質量%、DKを1質量%添加し、それ以外は実施例1と同様の電池を作製した。
(実施例93)
電解質の総量に対してFECを1質量%、DKを1質量%添加し、それ以外は実施例1と同様の電池を作製した。
(実施例94)
電解質の総量に対してFECを2質量%、DKを1質量%添加し、それ以外は実施例1と同様の電池を作製した。
(実施例95)
電解質の総量に対してFECを3質量%、DKを1質量%添加し、それ以外は実施例1と同様の電池を作製した。
(実施例96)
電解質の総量に対してFECを5質量%、DKを1質量%添加し、それ以外は実施例1と同様の電池を作製した。
(Example 91)
A battery was manufactured in the same manner as in Example 1 except that 0.1% by mass of FEC and 1% by mass of DK were added to the total amount of the electrolyte.
(Example 92)
A battery was manufactured in the same manner as in Example 1 except that 0.5% by mass of FEC and 1% by mass of DK were added to the total amount of the electrolyte.
(Example 93)
A battery was manufactured in the same manner as in Example 1 except that 1% by mass of FEC and 1% by mass of DK were added to the total amount of the electrolyte.
(Example 94)
A battery was manufactured in the same manner as in Example 1 except that 2% by mass of FEC and 1% by mass of DK were added to the total amount of the electrolyte.
(Example 95)
A battery was manufactured in the same manner as in Example 1 except that 3% by mass of FEC and 1% by mass of DK were added to the total amount of the electrolyte.
Example 96
A battery was manufactured in the same manner as in Example 1 except that 5% by mass of FEC and 1% by mass of DK were added to the total amount of the electrolyte.
(実施例97)
電解質の総量に対してFECを0.1質量%、DKを2質量%添加し、それ以外は実施例1と同様の電池を作製した。
(実施例98)
電解質の総量に対してFECを0.5質量%、DKを2質量%添加し、それ以外は実施例1と同様の電池を作製した。
(実施例99)
電解質の総量に対してFECを1質量%、DKを2質量%添加し、それ以外は実施例1と同様の電池を作製した。
(実施例100)
電解質の総量に対してFECを2質量%、DKを2質量%添加し、それ以外は実施例1と同様の電池を作製した。
(実施例101)
電解質の総量に対してFECを3質量%、DKを2質量%添加し、それ以外は実施例1と同様の電池を作製した。
(実施例102)
電解質の総量に対してFECを5質量%、DKを2質量%添加し、それ以外は実施例1と同様の電池を作製した。
(Example 97)
A battery was manufactured in the same manner as in Example 1 except that 0.1% by mass of FEC and 2% by mass of DK were added to the total amount of the electrolyte.
(Example 98)
A battery was manufactured in the same manner as in Example 1 except that 0.5% by mass of FEC and 2% by mass of DK were added to the total amount of the electrolyte.
Example 99
A battery was manufactured in the same manner as in Example 1 except that 1% by mass of FEC and 2% by mass of DK were added to the total amount of the electrolyte.
(Example 100)
A battery was manufactured in the same manner as in Example 1 except that 2% by mass of FEC and 2% by mass of DK were added to the total amount of the electrolyte.
(Example 101)
A battery was manufactured in the same manner as in Example 1 except that 3% by mass of FEC and 2% by mass of DK were added to the total amount of the electrolyte.
(Example 102)
A battery was manufactured in the same manner as in Example 1 except that 5% by mass of FEC and 2% by mass of DK were added to the total amount of the electrolyte.
(実施例103)
電解質の総量に対してFECを0.1質量%、DKを3質量%添加し、それ以外は実施例1と同様の電池を作製した。
(実施例104)
電解質の総量に対してFECを0.5質量%、DKを3質量%添加し、それ以外は実施例1と同様の電池を作製した。
(実施例105)
電解質の総量に対してFECを1質量%、DKを3質量%添加し、それ以外は実施例1と同様の電池を作製した。
(実施例106)
電解質の総量に対してFECを2質量%、DKを3質量%添加し、それ以外は実施例1と同様の電池を作製した。
(実施例107)
電解質の総量に対してFECを3質量%、DKを3質量%添加し、それ以外は実施例1と同様の電池を作製した。
(実施例108)
電解質の総量に対してFECを5質量%、DKを3質量%添加し、それ以外は実施例1と同様の電池を作製した。
(Example 103)
A battery was manufactured in the same manner as in Example 1 except that 0.1% by mass of FEC and 3% by mass of DK were added to the total amount of the electrolyte.
(Example 104)
A battery was manufactured in the same manner as in Example 1 except that 0.5% by mass of FEC and 3% by mass of DK were added to the total amount of the electrolyte.
(Example 105)
A battery was manufactured in the same manner as in Example 1 except that 1% by mass of FEC and 3% by mass of DK were added to the total amount of the electrolyte.
(Example 106)
A battery was manufactured in the same manner as in Example 1 except that 2% by mass of FEC and 3% by mass of DK were added to the total amount of the electrolyte.
(Example 107)
A battery was manufactured in the same manner as in Example 1 except that 3% by mass of FEC and 3% by mass of DK were added to the total amount of the electrolyte.
(Example 108)
A battery was manufactured in the same manner as in Example 1 except that 5% by mass of FEC and 3% by mass of DK were added to the total amount of the electrolyte.
(実施例109)
電解質の総量に対してFECを0.1質量%、VAを0.01質量%添加し、それ以外は実施例1と同様の電池を作製した。
(実施例110)
電解質の総量に対してFECを0.5質量%、VAを0.01質量%添加し、それ以外は実施例1と同様の電池を作製した。
(実施例111)
電解質の総量に対してFECを1質量%、VAを0.01質量%添加し、それ以外は実施例1と同様の電池を作製した。
(実施例112)
電解質の総量に対してFECを2質量%、VAを0.01質量%添加し、それ以外は実施例1と同様の電池を作製した。
(実施例113)
電解質の総量に対してFECを3質量%、VAを0.01質量%添加し、それ以外は実施例1と同様の電池を作製した。
(実施例114)
電解質の総量に対してFECを5質量%、VAを0.01質量%添加し、それ以外は実施例1と同様の電池を作製した。
(Example 109)
A battery was manufactured in the same manner as in Example 1 except that 0.1% by mass of FEC and 0.01% by mass of VA were added to the total amount of the electrolyte.
(Example 110)
A battery was manufactured in the same manner as in Example 1 except that 0.5% by mass of FEC and 0.01% by mass of VA were added to the total amount of the electrolyte.
(Example 111)
A battery was manufactured in the same manner as in Example 1 except that 1% by mass of FEC and 0.01% by mass of VA were added to the total amount of the electrolyte.
(Example 112)
A battery was manufactured in the same manner as in Example 1 except that 2% by mass of FEC and 0.01% by mass of VA were added to the total amount of the electrolyte.
(Example 113)
A battery was manufactured in the same manner as in Example 1 except that 3% by mass of FEC and 0.01% by mass of VA were added to the total amount of the electrolyte.
(Example 114)
A battery was manufactured in the same manner as in Example 1 except that 5% by mass of FEC and 0.01% by mass of VA were added to the total amount of the electrolyte.
(実施例115)
電解質の総量に対してFECを0.1質量%、VAを0.1質量%添加し、それ以外は実施例1と同様の電池を作製した。
(実施例116)
電解質の総量に対してFECを0.5質量%、VAを0.1質量%添加し、それ以外は実施例1と同様の電池を作製した。
(実施例117)
電解質の総量に対してFECを1質量%、VAを0.1質量%添加し、それ以外は実施例1と同様の電池を作製した。
(実施例118)
電解質の総量に対してFECを2質量%、VAを0.1質量%添加し、それ以外は実施例1と同様の電池を作製した。
(実施例119)
電解質の総量に対してFECを3質量%、VAを0.1質量%添加し、それ以外は実施例1と同様の電池を作製した。
(実施例120)
電解質の総量に対してFECを5質量%、VAを0.1質量%添加し、それ以外は実施例1と同様の電池を作製した。
(Example 115)
A battery was manufactured in the same manner as in Example 1 except that 0.1% by mass of FEC and 0.1% by mass of VA were added to the total amount of the electrolyte.
(Example 116)
A battery was manufactured in the same manner as in Example 1 except that 0.5% by mass of FEC and 0.1% by mass of VA were added to the total amount of the electrolyte.
(Example 117)
A battery was manufactured in the same manner as in Example 1 except that 1% by mass of FEC and 0.1% by mass of VA were added to the total amount of the electrolyte.
(Example 118)
A battery was manufactured in the same manner as in Example 1 except that 2% by mass of FEC and 0.1% by mass of VA were added to the total amount of the electrolyte.
(Example 119)
A battery was manufactured in the same manner as in Example 1 except that 3% by mass of FEC and 0.1% by mass of VA were added to the total amount of the electrolyte.
(Example 120)
A battery was manufactured in the same manner as in Example 1 except that 5% by mass of FEC and 0.1% by mass of VA were added to the total amount of the electrolyte.
(実施例121)
電解質の総量に対してFECを0.1質量%、VAを0.5質量%添加し、それ以外は実施例1と同様の電池を作製した。
(実施例122)
電解質の総量に対してFECを0.5質量%、VAを0.5質量%添加し、それ以外は実施例1と同様の電池を作製した。
(実施例123)
電解質の総量に対してFECを1質量%、VAを0.5質量%添加し、それ以外は実施例1と同様の電池を作製した。
(実施例124)
電解質の総量に対してFECを2質量%、VAを0.5質量%添加し、それ以外は実施例1と同様の電池を作製した。
(実施例125)
電解質の総量に対してFECを3質量%、VAを0.5質量%添加し、それ以外は実施例1と同様の電池を作製した。
(実施例126)
電解質の総量に対してFECを5質量%、VAを0.5質量%添加し、それ以外は実施例1と同様の電池を作製した。
(Example 121)
A battery was manufactured in the same manner as in Example 1 except that 0.1% by mass of FEC and 0.5% by mass of VA were added to the total amount of the electrolyte.
(Example 122)
A battery was manufactured in the same manner as in Example 1 except that 0.5% by mass of FEC and 0.5% by mass of VA were added to the total amount of the electrolyte.
(Example 123)
A battery was manufactured in the same manner as in Example 1 except that 1% by mass of FEC and 0.5% by mass of VA were added to the total amount of the electrolyte.
(Example 124)
A battery was manufactured in the same manner as in Example 1 except that 2% by mass of FEC and 0.5% by mass of VA were added to the total amount of the electrolyte.
(Example 125)
A battery was manufactured in the same manner as in Example 1 except that 3% by mass of FEC and 0.5% by mass of VA were added to the total amount of the electrolyte.
(Example 126)
A battery was manufactured in the same manner as in Example 1 except that 5% by mass of FEC and 0.5% by mass of VA were added to the total amount of the electrolyte.
(実施例127)
電解質の総量に対してFECを0.1質量%、VAを1質量%添加し、それ以外は実施例1と同様の電池を作製した。
(実施例128)
電解質の総量に対してFECを0.5質量%、VAを1質量%添加し、それ以外は実施例1と同様の電池を作製した。
(実施例129)
電解質の総量に対してFECを1質量%、VAを1質量%添加し、それ以外は実施例1と同様の電池を作製した。
(実施例130)
電解質の総量に対してFECを2質量%、VAを1質量%添加し、それ以外は実施例1と同様の電池を作製した。
(実施例131)
電解質の総量に対してFECを3質量%、VAを1質量%添加し、それ以外は実施例1と同様の電池を作製した。
(実施例132)
電解質の総量に対してFECを5質量%、VAを1質量%添加し、それ以外は実施例1と同様の電池を作製した。
(Example 127)
A battery was manufactured in the same manner as in Example 1 except that 0.1% by mass of FEC and 1% by mass of VA were added to the total amount of the electrolyte.
(Example 128)
A battery was manufactured in the same manner as in Example 1 except that 0.5% by mass of FEC and 1% by mass of VA were added to the total amount of the electrolyte.
(Example 129)
A battery was manufactured in the same manner as in Example 1 except that 1% by mass of FEC and 1% by mass of VA were added to the total amount of the electrolyte.
(Example 130)
A battery was manufactured in the same manner as in Example 1 except that 2% by mass of FEC and 1% by mass of VA were added to the total amount of the electrolyte.
(Example 131)
A battery was manufactured in the same manner as in Example 1 except that 3% by mass of FEC and 1% by mass of VA were added to the total amount of the electrolyte.
(Example 132)
A battery was manufactured in the same manner as in Example 1 except that 5% by mass of FEC and 1% by mass of VA were added to the total amount of the electrolyte.
(実施例133)
電解質の総量に対してFECを0.1質量%、VAを2質量%添加し、それ以外は実施例1と同様の電池を作製した。
(実施例134)
電解質の総量に対してFECを0.5質量%、VAを2質量%添加し、それ以外は実施例1と同様の電池を作製した。
(実施例135)
電解質の総量に対してFECを1質量%、VAを2質量%添加し、それ以外は実施例1と同様の電池を作製した。
(実施例136)
電解質の総量に対してFECを2質量%、VAを2質量%添加し、それ以外は実施例1と同様の電池を作製した。
(実施例137)
電解質の総量に対してFECを3質量%、VAを2質量%添加し、それ以外は実施例1と同様の電池を作製した。
(実施例138)
電解質の総量に対してFECを5質量%、VAを2質量%添加し、それ以外は実施例1と同様の電池を作製した。
(Example 133)
A battery was manufactured in the same manner as in Example 1 except that 0.1% by mass of FEC and 2% by mass of VA were added to the total amount of the electrolyte.
(Example 134)
A battery was manufactured in the same manner as in Example 1 except that 0.5% by mass of FEC and 2% by mass of VA were added to the total amount of the electrolyte.
(Example 135)
A battery was manufactured in the same manner as in Example 1 except that 1% by mass of FEC and 2% by mass of VA were added to the total amount of the electrolyte.
(Example 136)
A battery was manufactured in the same manner as in Example 1 except that 2% by mass of FEC and 2% by mass of VA were added to the total amount of the electrolyte.
(Example 137)
A battery was manufactured in the same manner as in Example 1 except that 3% by mass of FEC and 2% by mass of VA were added to the total amount of the electrolyte.
(Example 138)
A battery was manufactured in the same manner as in Example 1 except that 5% by mass of FEC and 2% by mass of VA were added to the total amount of the electrolyte.
(実施例139)
電解質の総量に対してFECを0.1質量%、VAを3質量%添加し、それ以外は実施例1と同様の電池を作製した。
(実施例140)
電解質の総量に対してFECを0.5質量%、VAを3質量%添加し、それ以外は実施例1と同様の電池を作製した。
(実施例141)
電解質の総量に対してFECを1質量%、VAを3質量%添加し、それ以外は実施例1と同様の電池を作製した。
(実施例142)
電解質の総量に対してFECを2質量%、VAを3質量%添加し、それ以外は実施例1と同様の電池を作製した。
(実施例143)
電解質の総量に対してFECを3質量%、VAを3質量%添加し、それ以外は実施例1と同様の電池を作製した。
(実施例144)
電解質の総量に対してFECを5質量%、VAを3質量%添加し、それ以外は実施例1と同様の電池を作製した。
(Example 139)
A battery was manufactured in the same manner as in Example 1 except that 0.1% by mass of FEC and 3% by mass of VA were added to the total amount of the electrolyte.
(Example 140)
A battery was manufactured in the same manner as in Example 1 except that 0.5% by mass of FEC and 3% by mass of VA were added to the total amount of the electrolyte.
(Example 141)
A battery was manufactured in the same manner as in Example 1 except that 1% by mass of FEC and 3% by mass of VA were added to the total amount of the electrolyte.
(Example 142)
A battery was manufactured in the same manner as in Example 1 except that 2% by mass of FEC and 3% by mass of VA were added to the total amount of the electrolyte.
(Example 143)
A battery was manufactured in the same manner as in Example 1 except that 3% by mass of FEC and 3% by mass of VA were added to the total amount of the electrolyte.
(Example 144)
A battery was manufactured in the same manner as in Example 1 except that 5% by mass of FEC and 3% by mass of VA were added to the total amount of the electrolyte.
(実施例145)
電解質の総量に対してビニルエチレンカーボネート(VEC(4-vinyl-1,3-dioxolan-2-one)、化7参照)を0.5質量%、DKを0.5質量%添加し、それ以外は実施例1と同様の電池を作製した。
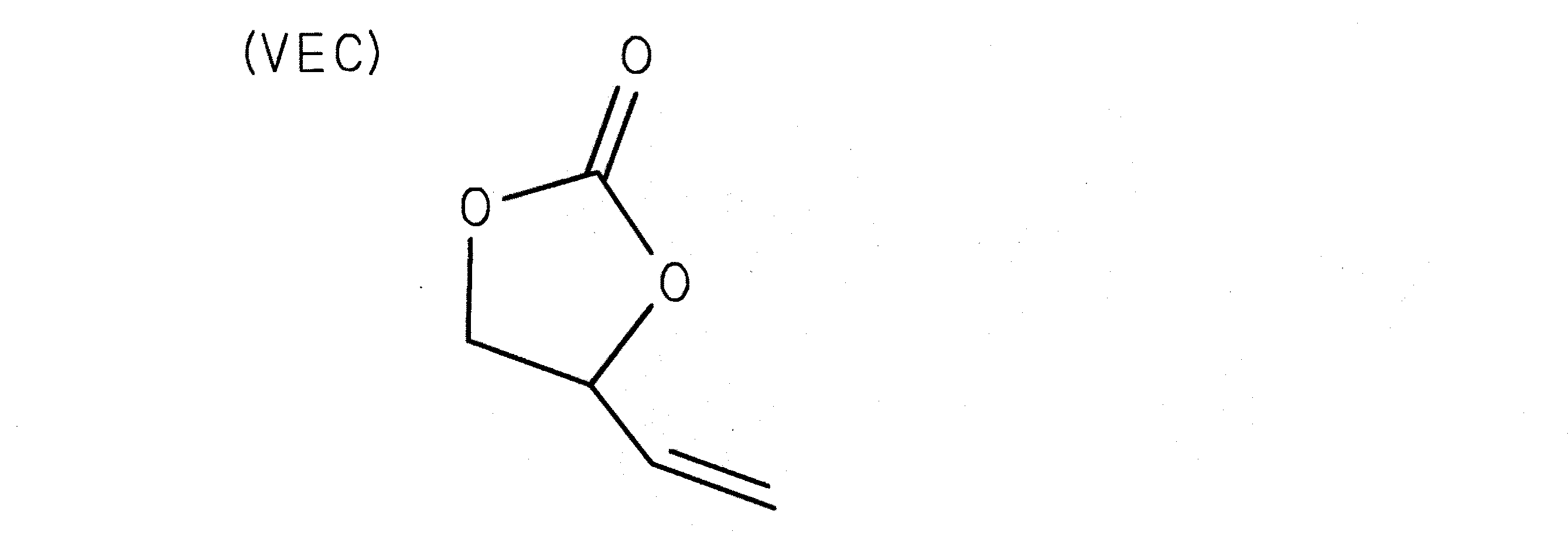
(Example 145)
Add 0.5% by mass of vinyl ethylene carbonate (VEC (4-vinyl-1,3-dioxolan-2-one), see chemical formula 7), 0.5% by mass of DK to the total amount of electrolyte, and other than that Produced the same battery as in Example 1.
(実施例146)
電解質の総量に対してVECを0.5質量%、VAを0.5質量%添加し、それ以外は実施例1と同様の電池を作製した。
(実施例147)
電解質の総量に対してジビニルエチレンカーボネート(DVEC(4,5-divinyl-1,3-dioxolan-2-one)、化8参照)を0.5質量%、DKを0.5質量%添加し、それ以外は実施例1と同様の電池を作製した。
(Example 146)
A battery was manufactured in the same manner as in Example 1 except that 0.5% by mass of VEC and 0.5% by mass of VA were added to the total amount of the electrolyte.
(Example 147)
0.5% by mass of divinylethylene carbonate (DVEC (4,5-divinyl-1,3-dioxolan-2-one), see Chemical Formula 8) and 0.5% by mass of DK are added to the total amount of the electrolyte, Otherwise, a battery similar to that of Example 1 was produced.
(実施例148)
電解質の総量に対してDVECを0.5質量%、VAを0.5質量%添加し、それ以外は実施例1と同様の電池を作製した。
(実施例149)
電解質の総量に対してALOM−EC(4-(prop-2-enyloxymethyl)-1,3-dioxolan-2-one、化9参照)を0.5質量%、DKを0.5質量%添加し、それ以外は実施例1と同様の電池を作製した。
(Example 148)
A battery was manufactured in the same manner as in Example 1 except that 0.5% by mass of DVEC and 0.5% by mass of VA were added to the total amount of the electrolyte.
(Example 149)
Add 0.5% by mass of ALOM-EC (4- (prop-2-enyloxymethyl) -1,3-dioxolan-2-one, see Chemical formula 9) and 0.5% by mass of DK to the total amount of electrolyte. Otherwise, a battery similar to that of Example 1 was produced.
(実施例150)
電解質の総量に対してALOM−ECを0.5質量%、VAを0.5質量%添加し、それ以外は実施例1と同様の電池を作製した。
(実施例151)
電解質の総量に対してPGOM−EC(4-(prop-2-ynyloxymethyl)-1,3-dioxolan-2-one、化10参照)を0.5質量%、DKを0.5質量%添加し、それ以外は実施例1と同様の電池を作製した。
(Example 150)
A battery was manufactured in the same manner as in Example 1 except that 0.5% by mass of ALOM-EC and 0.5% by mass of VA were added to the total amount of the electrolyte.
(Example 151)
Add 0.5% by mass of PGOM-EC (4- (prop-2-ynyloxymethyl) -1,3-dioxolan-2-one, see Chemical Formula 10) and 0.5% by mass of DK to the total amount of electrolyte. Otherwise, a battery similar to that of Example 1 was produced.
(実施例152)
電解質の総量に対してPGOM−ECを0.5質量%、VAを0.5質量%添加し、それ以外は実施例1と同様の電池を作製した。
(実施例153)
電解質の総量に対してモノクロロエチレンカーボネート(ClEC(4-chloro-1,3-dioxolan-2-one)、化11参照)を0.5質量%、DKを0.5質量%添加し、それ以外は実施例1と同様の電池を作製した。
(Example 152)
A battery was manufactured in the same manner as in Example 1 except that 0.5% by mass of PGOM-EC and 0.5% by mass of VA were added to the total amount of the electrolyte.
(Example 153)
Add 0.5% by mass of monochloroethylene carbonate (ClEC (4-chloro-1,3-dioxolan-2-one), see Chemical formula 11) and 0.5% by mass of DK with respect to the total amount of electrolyte. Produced the same battery as in Example 1.
(実施例154)
電解質の総量に対してClECを0.5質量%、VAを0.5質量%添加し、それ以外は実施例1と同様の電池を作製した。
(実施例155)
電解質の総量に対してVCを0.5質量%、FECを0.5質量%、DKを0.5質量%添加し、それ以外は実施例1と同様の電池を作製した。
(実施例156)
電解質の総量に対してVCを1質量%、FECを1質量%、DKを0.5質量%添加し、それ以外は実施例1と同様の電池を作製した。
(実施例157)
電解質の総量に対してVCを1質量%、プロピオン酸ビニル0.5質量%(化12参照)添加し、それ以外は実施例1と同様の電池を作製した。
(Example 154)
A battery was manufactured in the same manner as in Example 1 except that 0.5% by mass of ClEC and 0.5% by mass of VA were added to the total amount of the electrolyte.
(Example 155)
A battery was manufactured in the same manner as in Example 1 except that 0.5% by mass of VC, 0.5% by mass of FEC, and 0.5% by mass of DK were added to the total amount of the electrolyte.
(Example 156)
A battery was manufactured in the same manner as in Example 1 except that 1% by mass of VC, 1% by mass of FEC, and 0.5% by mass of DK were added to the total amount of the electrolyte.
(Example 157)
A battery was manufactured in the same manner as in Example 1 except that 1% by mass of VC and 0.5% by mass of vinyl propionate (see Chemical formula 12) were added to the total amount of the electrolyte.
(実施例158)
電解質の総量に対してVCを1質量%、酪酸ビニル0.5質量%(化13参照)添加し、それ以外は実施例1と同様の電池を作製した。
(Example 158)
A battery was manufactured in the same manner as in Example 1 except that 1% by mass of VC and 0.5% by mass of vinyl butyrate (see Chemical formula 13) were added to the total amount of the electrolyte.
(実施例159)
電解質の総量に対してVCを1質量%、カプロン酸ビニル0.5質量%(化14参照)添加し、それ以外は実施例1と同様の電池を作製した。
(Example 159)
A battery was manufactured in the same manner as in Example 1 except that 1% by mass of VC and 0.5% by mass of vinyl caproate (see Chemical formula 14) were added to the total amount of the electrolyte.
(実施例160)
電解質の総量に対してVCを1質量%、モノクロロ酢酸ビニル0.5質量%(化15参照)添加し、それ以外は実施例1と同様の電池を作製した。
(Example 160)
A battery was manufactured in the same manner as in Example 1 except that 1% by mass of VC and 0.5% by mass of monochlorovinyl acetate (see Chemical formula 15) were added to the total amount of the electrolyte.
(実施例161)
電解質の総量に対してVCを1質量%、モノフルオロ酢酸ビニル0.5質量%(化16参照)添加し、それ以外は実施例1と同様の電池を作製した。
(Example 161)
A battery was manufactured in the same manner as in Example 1 except that 1% by mass of VC and 0.5% by mass of vinyl monofluoroacetate (see Chemical formula 16) were added to the total amount of the electrolyte.
(実施例162)
電解質の総量に対してVCを1質量%、メタクリル酸ビニル0.5質量%(化17参照)添加し、それ以外は実施例1と同様の電池を作製した。
(Example 162)
A battery was manufactured in the same manner as in Example 1 except that 1% by mass of VC and 0.5% by mass of vinyl methacrylate (see Chemical formula 17) were added to the total amount of the electrolyte.
(実施例163)
電解質の総量に対してVCを1質量%、クロトン酸ビニル0.5質量%(化18参照)添加し、それ以外は実施例1と同様の電池を作製した。
(Example 163)
A battery was manufactured in the same manner as in Example 1 except that 1% by mass of VC and 0.5% by mass of vinyl crotonate (see Chemical formula 18) were added to the total amount of the electrolyte.
(実施例164)
電解質の総量に対してVCを1質量%、シクロヘキサンカルボン酸ビニル0.5質量%(化19参照)添加し、それ以外は実施例1と同様の電池を作製した。
(Example 164)
A battery was manufactured in the same manner as in Example 1 except that 1% by mass of VC and 0.5% by mass of vinyl cyclohexanecarboxylate (see Chemical formula 19) were added to the total amount of the electrolyte.
(実施例165)
電解質の総量に対してVCを1質量%、安息香酸ビニル0.5質量%(化20参照)添加し、それ以外は実施例1と同様の電池を作製した。
(Example 165)
A battery was manufactured in the same manner as in Example 1 except that 1% by mass of VC and 0.5% by mass of vinyl benzoate (see Chemical formula 20) were added to the total amount of the electrolyte.
(実施例166)
電解質の総量に対してVCを1質量%、桂皮酸ビニル0.5質量%(化21参照)添加し、それ以外は実施例1と同様の電池を作製した。
(Example 166)
A battery was manufactured in the same manner as in Example 1 except that 1% by mass of VC and 0.5% by mass of vinyl cinnamate (see Chemical formula 21) were added to the total amount of the electrolyte.
(比較例1)
電解質に添加剤を添加しておらず、それ以外は実施例1と同様の電池を作製した。
(比較例2)
電解質の総量に対してDKを0.01質量%添加し、それ以外は実施例1と同様の電池を作製した。
(比較例3)
電解質の総量に対してDKを0.1質量%添加し、それ以外は実施例1と同様の電池を作製した。
(比較例4)
電解質の総量に対してDKを0.5質量%添加し、それ以外は実施例1と同様の電池を作製した。
(比較例5)
電解質の総量に対してDKを1質量%添加し、それ以外は実施例1と同様の電池を作製した。
(比較例6)
電解質の総量に対してDKを2質量%添加し、それ以外は実施例1と同様の電池を作製した。
(比較例7)
電解質の総量に対してDKを3質量%添加し、それ以外は実施例1と同様の電池を作製した。
(Comparative Example 1)
A battery was prepared in the same manner as in Example 1 except that no additive was added to the electrolyte.
(Comparative Example 2)
A battery was manufactured in the same manner as in Example 1 except that 0.01% by mass of DK was added to the total amount of the electrolyte.
(Comparative Example 3)
A battery was prepared in the same manner as in Example 1 except that 0.1% by mass of DK was added to the total amount of the electrolyte.
(Comparative Example 4)
A battery was prepared in the same manner as in Example 1 except that 0.5% by mass of DK was added to the total amount of the electrolyte.
(Comparative Example 5)
A battery was prepared in the same manner as in Example 1 except that 1% by mass of DK was added to the total amount of the electrolyte.
(Comparative Example 6)
A battery was prepared in the same manner as in Example 1 except that 2% by mass of DK was added to the total amount of the electrolyte.
(Comparative Example 7)
A battery was prepared in the same manner as in Example 1 except that 3% by mass of DK was added to the total amount of the electrolyte.
(比較例8)
電解質の総量に対してVCを0.1質量%添加し、それ以外は実施例1と同様の電池を作製した。
(比較例9)
電解質の総量に対してVCを0.5質量%添加し、それ以外は実施例1と同様の電池を作製した。
(比較例10)
電解質の総量に対してVCを1質量%添加し、それ以外は実施例1と同様の電池を作製した。
(比較例11)
電解質の総量に対してVCを2質量%添加し、それ以外は実施例1と同様の電池を作製した。
(比較例12)
電解質の総量に対してVCを3質量%添加し、それ以外は実施例1と同様の電池を作製した。
(比較例13)
電解質の総量に対してVCを5質量%添加し、それ以外は実施例1と同様の電池を作製した。
(Comparative Example 8)
A battery was manufactured in the same manner as in Example 1 except that 0.1% by mass of VC was added to the total amount of the electrolyte.
(Comparative Example 9)
A battery was manufactured in the same manner as in Example 1 except that 0.5% by mass of VC was added to the total amount of the electrolyte.
(Comparative Example 10)
A battery was manufactured in the same manner as in Example 1 except that 1% by mass of VC was added to the total amount of the electrolyte.
(Comparative Example 11)
A battery was manufactured in the same manner as in Example 1 except that 2% by mass of VC was added to the total amount of the electrolyte.
(Comparative Example 12)
A battery was manufactured in the same manner as in Example 1 except that 3% by mass of VC was added to the total amount of the electrolyte.
(Comparative Example 13)
A battery was manufactured in the same manner as in Example 1 except that 5% by mass of VC was added to the total amount of the electrolyte.
(比較例14)
電解質の総量に対してVAを0.01質量%添加し、それ以外は実施例1と同様の電池を作製した。
(比較例15)
電解質の総量に対してVAを0.1質量%添加し、それ以外は実施例1と同様の電池を作製した。
(比較例16)
電解質の総量に対してVAを0.5質量%添加し、それ以外は実施例1と同様の電池を作製した。
(比較例17)
電解質の総量に対してVAを1質量%添加し、それ以外は実施例1と同様の電池を作製した。
(比較例18)
電解質の総量に対してVAを2質量%添加し、それ以外は実施例1と同様の電池を作製した。
(比較例19)
電解質の総量に対してVAを3質量%添加し、それ以外は実施例1と同様の電池を作製した。
(Comparative Example 14)
A battery was manufactured in the same manner as in Example 1 except that 0.01% by mass of VA was added to the total amount of the electrolyte.
(Comparative Example 15)
A battery was manufactured in the same manner as in Example 1 except that 0.1% by mass of VA was added to the total amount of the electrolyte.
(Comparative Example 16)
A battery was manufactured in the same manner as in Example 1 except that 0.5% by mass of VA was added to the total amount of the electrolyte.
(Comparative Example 17)
A battery was manufactured in the same manner as in Example 1 except that 1% by mass of VA was added to the total amount of the electrolyte.
(Comparative Example 18)
A battery was manufactured in the same manner as in Example 1 except that 2% by mass of VA was added to the total amount of the electrolyte.
(Comparative Example 19)
A battery was manufactured in the same manner as in Example 1 except that 3% by mass of VA was added to the total amount of the electrolyte.
(比較例20)
電解質の総量に対してFECを0.1質量%添加し、それ以外は実施例1と同様の電池を作製した。
(比較例21)
電解質の総量に対してFECを0.5質量%添加し、それ以外は実施例1と同様の電池を作製した。
(比較例22)
電解質の総量に対してFECを1質量%添加し、それ以外は実施例1と同様の電池を作製した。
(比較例23)
電解質の総量に対してFECを2質量%添加し、それ以外は実施例1と同様の電池を作製した。
(比較例24)
電解質の総量に対してFECを3質量%添加し、それ以外は実施例1と同様の電池を作製した。
(比較例25)
電解質の総量に対してFECを5質量%添加し、それ以外は実施例1と同様の電池を作製した。
(Comparative Example 20)
A battery was manufactured in the same manner as in Example 1 except that 0.1% by mass of FEC was added to the total amount of the electrolyte.
(Comparative Example 21)
A battery was manufactured in the same manner as in Example 1 except that 0.5% by mass of FEC was added to the total amount of the electrolyte.
(Comparative Example 22)
A battery was manufactured in the same manner as in Example 1 except that 1% by mass of FEC was added to the total amount of the electrolyte.
(Comparative Example 23)
A battery was manufactured in the same manner as in Example 1 except that 2% by mass of FEC was added to the total amount of the electrolyte.
(Comparative Example 24)
A battery was manufactured in the same manner as in Example 1 except that 3% by mass of FEC was added to the total amount of the electrolyte.
(Comparative Example 25)
A battery was manufactured in the same manner as in Example 1 except that 5% by mass of FEC was added to the total amount of the electrolyte.
(比較例26)
電解質の総量に対してVECを0.5質量%添加し、それ以外は実施例1と同様の電池を作製した。
(比較例27)
電解質の総量に対してVECを1質量%添加し、それ以外は実施例1と同様の電池を作製した。
(比較例28)
電解質の総量に対してDVECを0.5質量%添加し、それ以外は実施例1と同様の電池を作製した。
(比較例29)
電解質の総量に対してDVECを1質量%添加し、それ以外は実施例1と同様の電池を作製した。
(Comparative Example 26)
A battery was manufactured in the same manner as in Example 1 except that 0.5% by mass of VEC was added to the total amount of the electrolyte.
(Comparative Example 27)
A battery was manufactured in the same manner as in Example 1 except that 1% by mass of VEC was added to the total amount of the electrolyte.
(Comparative Example 28)
A battery was manufactured in the same manner as in Example 1 except that 0.5% by mass of DVEC was added to the total amount of the electrolyte.
(Comparative Example 29)
A battery was manufactured in the same manner as in Example 1 except that 1% by mass of DVEC was added to the total amount of the electrolyte.
(比較例30)
電解質の総量に対してALOM−ECを0.5質量%添加し、それ以外は実施例1と同様の電池を作製した。
(比較例31)
電解質の総量に対してALOM−ECを1質量%添加し、それ以外は実施例1と同様の電池を作製した。
(比較例32)
電解質の総量に対してPGOM−ECを0.5質量%添加し、それ以外は実施例1と同様の電池を作製した。
(比較例33)
電解質の総量に対してPGOM−ECを1質量%添加し、それ以外は実施例1と同様の電池を作製した。
(Comparative Example 30)
A battery was manufactured in the same manner as in Example 1 except that 0.5% by mass of ALOM-EC was added to the total amount of the electrolyte.
(Comparative Example 31)
A battery was manufactured in the same manner as in Example 1 except that 1% by mass of ALOM-EC was added to the total amount of the electrolyte.
(Comparative Example 32)
A battery was manufactured in the same manner as in Example 1 except that 0.5% by mass of PGOM-EC was added to the total amount of the electrolyte.
(Comparative Example 33)
A battery was manufactured in the same manner as in Example 1 except that 1% by mass of PGOM-EC was added to the total amount of the electrolyte.
(比較例34)
電解質の総量に対してCIECを0.5質量%添加し、それ以外は実施例1と同様の電池を作製した。
(比較例35)
電解質の総量に対してCIECを1質量%添加し、それ以外は実施例1と同様の電池を作製した。
(比較例36)
電解質の総量に対してVCを0.5質量%、FECを0.5質量%添加し、それ以外は実施例1と同様の電池を作製した。
(比較例37)
電解質の総量に対してVCを1質量%、FECを1質量%添加し、それ以外は実施例1と同様の電池を作製した。
(Comparative Example 34)
A battery was manufactured in the same manner as in Example 1 except that 0.5% by mass of CIEC was added to the total amount of the electrolyte.
(Comparative Example 35)
A battery was manufactured in the same manner as in Example 1 except that 1% by mass of CIEC was added to the total amount of the electrolyte.
(Comparative Example 36)
A battery was manufactured in the same manner as in Example 1 except that 0.5% by mass of VC and 0.5% by mass of FEC were added to the total amount of the electrolyte.
(Comparative Example 37)
A battery was manufactured in the same manner as in Example 1 except that 1% by mass of VC and 1% by mass of FEC were added to the total amount of the electrolyte.
(比較例38)
電解質の総量に対してプロピオン酸ビニルを0.5質量%添加し、それ以外は実施例1と同様の電池を作製した。
(比較例39)
電解質の総量に対して酪酸ビニルを0.5質量%添加し、それ以外は実施例1と同様の電池を作製した。
(比較例40)
電解質の総量に対してモノクロロ酢酸ビニルを0.5質量%添加し、それ以外は実施例1と同様の電池を作製した。
(比較例41)
電解質の総量に対してモノフルオロ酢酸ビニルを0.5質量%添加し、それ以外は実施例1と同様の電池を作製した。
(比較例42)
電解質の総量に対してメタクリル酸ビニルを0.5質量%添加し、それ以外は実施例1と同様の電池を作製した。
(Comparative Example 38)
A battery was manufactured in the same manner as in Example 1 except that 0.5% by mass of vinyl propionate was added to the total amount of the electrolyte.
(Comparative Example 39)
A battery was prepared in the same manner as in Example 1 except that 0.5% by mass of vinyl butyrate was added to the total amount of the electrolyte.
(Comparative Example 40)
A battery was manufactured in the same manner as in Example 1 except that 0.5% by mass of vinyl monochloroacetate was added to the total amount of the electrolyte.
(Comparative Example 41)
A battery was manufactured in the same manner as in Example 1 except that 0.5% by mass of vinyl monofluoroacetate was added to the total amount of the electrolyte.
(Comparative Example 42)
A battery was prepared in the same manner as in Example 1 except that 0.5% by mass of vinyl methacrylate was added to the total amount of the electrolyte.
(比較例43)
電解質の総量に対してクロトン酸ビニルを0.5質量%添加し、それ以外は実施例1と同様の電池を作製した。
(比較例44)
電解質の総量に対してシクロヘキサンカルボン酸ビニルを0.5質量%添加し、それ以外は実施例1と同様の電池を作製した。
(比較例45)
電解質の総量に対して安息香酸ビニルを0.5質量%添加し、それ以外は実施例1と同様の電池を作製した。
(比較例46)
電解質の総量に対して桂皮酸ビニルを0.5質量%添加し、それ以外は実施例1と同様の電池を作製した。
(Comparative Example 43)
A battery was manufactured in the same manner as in Example 1 except that 0.5% by mass of vinyl crotonate was added to the total amount of the electrolyte.
(Comparative Example 44)
A battery was manufactured in the same manner as in Example 1 except that 0.5% by mass of vinylcyclohexanecarboxylate was added to the total amount of the electrolyte.
(Comparative Example 45)
A battery was manufactured in the same manner as in Example 1 except that 0.5% by mass of vinyl benzoate was added to the total amount of the electrolyte.
(Comparative Example 46)
A battery was manufactured in the same manner as in Example 1 except that 0.5% by mass of vinyl cinnamate was added to the total amount of the electrolyte.
上述した各実施例及び各比較例の電池に対し、後述する初期容量、電池厚み、容量保持率(充放電サイクル寿命特性)を確認した。初期容量については、作製した電池を、電流800mAで4.2Vまで3時間定電流定電圧充電し、その後電流800mAで3Vまで放電を行い、放電容量[mAh](初期容量)を確認した。 The initial capacity, battery thickness, and capacity retention rate (charge / discharge cycle life characteristics) described later were confirmed for the batteries of the above-described Examples and Comparative Examples. Regarding the initial capacity, the produced battery was charged at a constant current and a constant voltage to 4.2 V at a current of 800 mA for 3 hours, and then discharged to 3 V at a current of 800 mA to confirm the discharge capacity [mAh] (initial capacity).
電池厚みについては、作製した電池を、電流800mAで4.2Vまで3時間定電流定電圧充電した後、85℃の恒温槽中で50時間放置した後、電池の厚さ[mm](電池厚み)を確認した。 Regarding the battery thickness, the prepared battery was charged at a current of 800 mA to 4.2 V at a constant current and a constant voltage for 3 hours, then left in a constant temperature bath at 85 ° C. for 50 hours, and then the battery thickness [mm] (battery thickness) )It was confirmed.
容量保持率については、25℃の環境下で、電流800mAで4.2Vまで3時間定電流定電圧充電し、その後電流800mAで3Vまで放電を行い、その後も上述した充電及び放電を繰返し500サイクル行い、初期放電容量に対する500サイクル後の放電容量の比[%](容量保持率)を求めた。 Regarding the capacity retention rate, the battery was charged at a constant current and a constant voltage for 3 hours at a current of 800 mA to 4.2 V in an environment of 25 ° C., and then discharged to 3 V at a current of 800 mA. The ratio [%] (capacity retention) of the discharge capacity after 500 cycles to the initial discharge capacity was determined.
各実施例及び各比較例の電池の初期容量、電池厚み、容量保持率を表1〜9に示す。 Tables 1 to 9 show the initial capacity, battery thickness, and capacity retention rate of the batteries of Examples and Comparative Examples.
また、実施例1〜144、比較例1〜25に関しては、初期容量、電池厚み、容量保持率を添加剤の添加量に応じて並べ替えたものを表10〜表25に示す。 Moreover, about Examples 1-144 and Comparative Examples 1-25, what rearranged initial capacity, battery thickness, and capacity | capacitance retention rate according to the addition amount of the additive is shown in Table 10-Table 25.
表14,17,20,23等に示すように、0.01質量%以上2質量%以下のDK又はVAを添加することにより初期容量が向上する。また、0.1質量%以上3質量%以下のVC又はFECを添加することにより初期容量が向上する。 As shown in Tables 14, 17, 20, 23, etc., the initial capacity is improved by adding 0.01% by mass or more and 2% by mass or less of DK or VA. Moreover, initial capacity improves by adding 0.1 mass% or more and 3 mass% or less of VC or FEC.
表15,18,21,24等に示すように、0.01質量%以上のDK又はVAを添加することにより電池厚みの増加が抑制される。 As shown in Tables 15, 18, 21, 24, etc., an increase in battery thickness is suppressed by adding 0.01% by mass or more of DK or VA.
比較例のように、環状炭酸エステル誘導体としてのVC、FEC、VEC、DVEC、ALOM−EC、PGOM−EC、及びCIECの何れかを単独で添加したり、ビニルエステル誘導体としてのDK、VA、プロピオン酸ビニル、酪酸ビニル、モノクロロ酢酸ビニル、モノフルオロ酢酸ビニル、メタクリル酸ビニル、クロトン酸ビニル、シクロヘキサンカルボン酸ビニル、安息香酸ビニル及び桂皮酸ビニルの何れかを単独で添加するよりも、実施例のように、環状炭酸エステル誘導体とビニルエステル誘導体とを混合して添加する方が、初期容量、容量保持率(サイクル特性)が向上すると共に高温放置時の電池厚みの増加を抑制することが分かる。 As in the comparative example, any one of VC, FEC, VEC, DVEC, ALOM-EC, PGOM-EC, and CIEC as a cyclic carbonate derivative is added alone, or DK, VA, and propion as vinyl ester derivatives Example 1 rather than adding any one of vinyl acid vinyl, vinyl butyrate, vinyl monochloroacetate, vinyl monofluoroacetate, vinyl methacrylate, vinyl crotonate, vinyl cyclohexanecarboxylate, vinyl benzoate and vinyl cinnamate alone In addition, it can be seen that the addition of a mixture of a cyclic carbonate derivative and a vinyl ester derivative improves initial capacity and capacity retention (cycle characteristics) and suppresses an increase in battery thickness when left at high temperature.
ビニルエステル誘導体を比較した場合、実施例51、実施例157〜159、比較例16、比較例38、39より、アルキル鎖長が長くなるに従い、高温放置時の電池厚みの増加が抑制されることが分かる。これは、耐酸化性が向上し、また、沸点が高くなるためと考えられる。
アルキル鎖長が長くなるに従い、容量保持率(サイクル特性)は若干低下している。これは、負極上に形成される負極保護被膜の性状の違いによるものであると考えられる。
When vinyl ester derivatives are compared, the increase in battery thickness at high temperature is suppressed as the alkyl chain length becomes longer than in Example 51, Examples 157 to 159, Comparative Example 16, and Comparative Examples 38 and 39. I understand. This is presumably because the oxidation resistance is improved and the boiling point is increased.
As the alkyl chain length increases, the capacity retention (cycle characteristics) slightly decreases. This is considered to be due to the difference in properties of the negative electrode protective film formed on the negative electrode.
実施例160、比較例40より、クロロ化されている場合、高温放置時の電池厚みの増加が抑制されることが分かる。これは、耐酸化性が向上したためと考えられる。
クロロ化されている場合、容量保持率は若干低下している。これは、塩素を含むことにより、負極保護被膜の性状が変わるためであると考えられる。
From Example 160 and Comparative Example 40, it can be seen that when chlorinated, the increase in battery thickness when left at high temperature is suppressed. This is thought to be due to improved oxidation resistance.
When chlorinated, the capacity retention is slightly reduced. This is considered to be because the properties of the negative electrode protective film change due to the inclusion of chlorine.
実施例161、比較例41より、フルオロ化されている場合、高温放置時の電池厚みの増加はさらに抑制されることが分かる。これは、耐酸化性が向上したためであると考えられる。容量保持率もクロロ化されている場合より良好である。これは、フッ素を含むので、LiF等の安定な負極保護被膜を形成するためであると考えられる。 From Example 161 and Comparative Example 41, it can be seen that when fluorinated, the increase in battery thickness when left at high temperature is further suppressed. This is considered to be due to the improved oxidation resistance. Capacity retention is also better than when chlorinated. This is considered to be for forming a stable negative electrode protective film such as LiF because it contains fluorine.
実施例162、163、比較例42、43の高温放置時の電池の膨れは、他より大きい。これは、ビニル基が複数あるので、耐酸化性が低下し、正極上で酸化分解したためであると考えられる。容量保持率は良好である。ビニル基が複数あるので、強固な負極保護被膜を形成したためと考えられる。 In Examples 162 and 163 and Comparative Examples 42 and 43, the swelling of the battery when left at high temperature is larger than the others. This is considered to be due to the fact that since there are a plurality of vinyl groups, the oxidation resistance is lowered and oxidative decomposition occurs on the positive electrode. The capacity retention is good. This is probably because a strong negative electrode protective film was formed because there are a plurality of vinyl groups.
実施例164、比較例44のようにシクロヘキシル基を有する場合、アルキル基を有する場合より高温放置時の電池の膨れが小さくなることが分かる。容量保持率は、アルキル基を有する場合と比較して良くなかった。負極保護被膜の性状の違いによるものと考えられる。 As shown in Example 164 and Comparative Example 44, when the cyclohexyl group is included, the battery swells when left at high temperature is smaller than when the alkyl group is included. The capacity retention was not good as compared with the case having an alkyl group. This is thought to be due to the difference in properties of the negative electrode protective film.
実施例165、166、比較例45、46より、シクロヘキシル基を有するよりフェニル基を有する方が、高温放置時の電池の膨れが大きくなることが分かる。これは、フェニル基を有する方が酸化分解を受けやすいためであると考えられる。容量保持率はシクロヘキシル基を有する場合より良好であった。負極保護被膜の性状によるものと考えられる。 From Examples 165 and 166 and Comparative Examples 45 and 46, it can be seen that a battery having a phenyl group has a larger swelling when left at a high temperature than a cyclohexyl group. This is considered to be because the one having a phenyl group is more susceptible to oxidative decomposition. The capacity retention was better than that having a cyclohexyl group. This is considered to be due to the properties of the negative electrode protective film.
以上より、0.1質量%以上3質量%以下の環状炭酸エステル誘導体と、0.01質量%以上2質量%以下のエステル誘導体とを添加することにより、電池の初期容量、容量保持率(サイクル特性)が向上すると共に高温放置時の電池厚さの増加を抑制することが出来ることが分かる。 From the above, by adding 0.1% by mass or more and 3% by mass or less of the cyclic carbonate derivative and 0.01% by mass or more and 2% by mass or less of the ester derivative, the initial capacity of the battery, capacity retention rate (cycle) It can be seen that the characteristics) can be improved and the increase in battery thickness when left at high temperature can be suppressed.
1 非水電解質二次電池
2 電極群
3 負極
4 正極
5 セパレータ
6 電池ケース
7 電池蓋
8 安全弁
9 負極端子
10 負極リード
DESCRIPTION OF
Claims (5)
前記電解質は、下記化1で表される環状炭酸エステル誘導体、及び下記化2で表される環状炭酸エステル誘導体のうちの少なくとも1種の化合物と、
ビニルエステル誘導体と
を含んでなることを特徴とする非水電解質二次電池。
(式中、R1 及びR2 は、それぞれ独立して水素原子、アルキル基、不飽和結合を含む炭化水素基、アルコキシ基、ハロゲン、ハロゲンを有するアルキル基、又はアリール基である。但し、R1及びR2 はエーテル結合を含んでもよい。)
(式中、R3 はアルキル基、不飽和結合を含む炭化水素基、アルコキシ基、ハロゲン、ハロゲンを有するアルキル基、又はアリール基である。R4 は、水素原子、アルキル基、不飽和結合を含む炭化水素基、アルコキシ基、ハロゲン、ハロゲンを有するアルキル基、又はアリール基である。但し、R3 及び4 はエーテル結合を含んでもよい。化2はプロピレンカーボネートを除く。) In a non-aqueous electrolyte secondary battery comprising an electrode that occludes and releases lithium and an electrolyte,
The electrolyte includes at least one compound selected from a cyclic carbonate derivative represented by the following chemical formula 1 and a cyclic carbonate derivative represented by the following chemical formula 2:
A non-aqueous electrolyte secondary battery comprising a vinyl ester derivative.
(In the formula, R 1 and R 2 are each independently a hydrogen atom, an alkyl group, a hydrocarbon group containing an unsaturated bond, an alkoxy group, a halogen, an alkyl group having a halogen, or an aryl group. 1 and R 2 may contain an ether bond.)
(In the formula, R 3 is an alkyl group, a hydrocarbon group containing an unsaturated bond, an alkoxy group, a halogen, a halogen-containing alkyl group, or an aryl group. R 4 is a hydrogen atom, an alkyl group, or an unsaturated bond. A hydrocarbon group, an alkoxy group, a halogen, a halogen-containing alkyl group, or an aryl group, wherein R 3 and 4 may contain an ether bond (Chemical Formula 2 excludes propylene carbonate.)
Priority Applications (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| JP2006065202A JP5160744B2 (en) | 2006-03-10 | 2006-03-10 | Nonaqueous electrolyte secondary battery |
Applications Claiming Priority (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| JP2006065202A JP5160744B2 (en) | 2006-03-10 | 2006-03-10 | Nonaqueous electrolyte secondary battery |
Publications (2)
| Publication Number | Publication Date |
|---|---|
| JP2007242496A true JP2007242496A (en) | 2007-09-20 |
| JP5160744B2 JP5160744B2 (en) | 2013-03-13 |
Family
ID=38587816
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| JP2006065202A Expired - Fee Related JP5160744B2 (en) | 2006-03-10 | 2006-03-10 | Nonaqueous electrolyte secondary battery |
Country Status (1)
| Country | Link |
|---|---|
| JP (1) | JP5160744B2 (en) |
Cited By (24)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JP2009152133A (en) * | 2007-12-21 | 2009-07-09 | Sony Corp | Nonaqueous electrolyte secondary battery |
| WO2011142412A1 (en) * | 2010-05-12 | 2011-11-17 | 三菱化学株式会社 | Nonaqueous-electrolyte secondary battery |
| WO2011142410A1 (en) * | 2010-05-12 | 2011-11-17 | 三菱化学株式会社 | Non-aqueous electrolytic solution, and non-aqueous electrolyte secondary battery |
| JP2012049106A (en) * | 2010-05-12 | 2012-03-08 | Mitsubishi Chemicals Corp | Nonaqueous electrolyte secondary battery |
| JP2012054231A (en) * | 2010-08-05 | 2012-03-15 | Mitsubishi Chemicals Corp | Nonaqueous electrolyte and nonaqueous electrolyte battery |
| WO2012035821A1 (en) * | 2010-09-16 | 2012-03-22 | 三菱化学株式会社 | Nonaqueous electrolyte and nonaqueous electrolyte secondary battery |
| JP2012064473A (en) * | 2010-09-16 | 2012-03-29 | Mitsubishi Chemicals Corp | Nonaqueous electrolyte and nonaqueous electrolyte battery |
| JP2012094501A (en) * | 2010-09-28 | 2012-05-17 | Mitsubishi Chemicals Corp | Nonaqueous electrolyte secondary battery |
| WO2012067248A1 (en) * | 2010-11-19 | 2012-05-24 | 三菱化学株式会社 | Process for producing 4-alkynyl-1,3-dioxolan-2-one derivative |
| JP2012109240A (en) * | 2010-10-29 | 2012-06-07 | Mitsubishi Chemicals Corp | Nonaqueous electrolyte secondary battery |
| WO2012141001A1 (en) * | 2011-04-11 | 2012-10-18 | 宇部興産株式会社 | Non-aqueous electrolyte solution and electricity-storage device using same |
| WO2012147502A1 (en) * | 2011-04-27 | 2012-11-01 | 昭和電工株式会社 | Non-aqueous electrolyte solution for secondary cell, and non-aqueous electrolyte secondary cell |
| JP2012227060A (en) * | 2011-04-21 | 2012-11-15 | Mitsubishi Chemicals Corp | Nonaqueous electrolyte and nonaqueous electrolyte battery using the same |
| JP2013239426A (en) * | 2011-11-11 | 2013-11-28 | Mitsubishi Chemicals Corp | Nonaqueous electrolyte secondary battery and nonaqueous electrolyte |
| CN103794817A (en) * | 2014-02-20 | 2014-05-14 | 福建创鑫科技开发有限公司 | Application of vinyl ethylene carbonate to lithium ion battery |
| KR101483126B1 (en) | 2008-03-07 | 2015-01-15 | 삼성에스디아이 주식회사 | Organic eletrolyte and lithium battery using the same |
| JPWO2013058235A1 (en) * | 2011-10-17 | 2015-04-02 | 宇部興産株式会社 | Non-aqueous electrolyte and power storage device using the same |
| WO2019102722A1 (en) * | 2017-11-21 | 2019-05-31 | ダイキン工業株式会社 | Electrolytic solution, electrochemical device, lithium ion secondary battery, and module |
| WO2019188210A1 (en) * | 2018-03-27 | 2019-10-03 | ダイキン工業株式会社 | Electrolyte solution, electrochemical device, lithium ion secondary battery, and module |
| US20190334207A1 (en) * | 2017-01-23 | 2019-10-31 | Lg Chem, Ltd. | Additive For Non-aqueous Electrolyte Solution, And Non-aqueous Electrolyte Solution For Lithium Secondary Battery And Lithium Secondary Battery Which Include The Same |
| WO2019207983A1 (en) * | 2018-04-25 | 2019-10-31 | ダイキン工業株式会社 | Electrolytic solution, electrochemical device, lithium-ion secondary battery, and module |
| CN111656595A (en) * | 2018-01-30 | 2020-09-11 | 大金工业株式会社 | Electrolyte solution, electrochemical device, lithium ion secondary battery, and assembly |
| CN114243111A (en) * | 2021-12-17 | 2022-03-25 | 松山湖材料实验室 | Lithium ion battery electrolyte additive, electrolyte and lithium ion battery |
| CN114361590A (en) * | 2021-12-17 | 2022-04-15 | 松山湖材料实验室 | Lithium ion battery electrolyte additive, electrolyte and lithium ion battery |
Citations (8)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JP2000299127A (en) * | 1999-02-12 | 2000-10-24 | Mitsui Chemicals Inc | Nonaqueous electrolyte and secondary battery using it |
| JP2001052743A (en) * | 1999-08-13 | 2001-02-23 | Mitsui Chemicals Inc | Nonaqueous electrolyte and secondary battery using the same |
| JP2002324580A (en) * | 2001-02-23 | 2002-11-08 | Mitsubishi Chemicals Corp | Nonaqueous electrolyte secondary battery |
| JP2004259681A (en) * | 2003-02-27 | 2004-09-16 | Sanyo Electric Co Ltd | Non-aqueous lithium secondary battery |
| WO2005020365A1 (en) * | 2003-08-26 | 2005-03-03 | Japan Aerospace Exploration Agency | Nonflammable nonaqueous electrolyte and lithium-ion battery containing the same |
| JP2006172950A (en) * | 2004-12-16 | 2006-06-29 | Nec Corp | Electrolyte for secondary battery, and the secondary battery |
| WO2006067957A1 (en) * | 2004-12-24 | 2006-06-29 | Matsushita Electric Industrial Co., Ltd. | Nonaqueous electrolyte for secondary battery and secondary battery comprising same |
| JP2006286606A (en) * | 2005-03-08 | 2006-10-19 | Sanyo Electric Co Ltd | Nonaqueous electrolyte secondary battery |
-
2006
- 2006-03-10 JP JP2006065202A patent/JP5160744B2/en not_active Expired - Fee Related
Patent Citations (8)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JP2000299127A (en) * | 1999-02-12 | 2000-10-24 | Mitsui Chemicals Inc | Nonaqueous electrolyte and secondary battery using it |
| JP2001052743A (en) * | 1999-08-13 | 2001-02-23 | Mitsui Chemicals Inc | Nonaqueous electrolyte and secondary battery using the same |
| JP2002324580A (en) * | 2001-02-23 | 2002-11-08 | Mitsubishi Chemicals Corp | Nonaqueous electrolyte secondary battery |
| JP2004259681A (en) * | 2003-02-27 | 2004-09-16 | Sanyo Electric Co Ltd | Non-aqueous lithium secondary battery |
| WO2005020365A1 (en) * | 2003-08-26 | 2005-03-03 | Japan Aerospace Exploration Agency | Nonflammable nonaqueous electrolyte and lithium-ion battery containing the same |
| JP2006172950A (en) * | 2004-12-16 | 2006-06-29 | Nec Corp | Electrolyte for secondary battery, and the secondary battery |
| WO2006067957A1 (en) * | 2004-12-24 | 2006-06-29 | Matsushita Electric Industrial Co., Ltd. | Nonaqueous electrolyte for secondary battery and secondary battery comprising same |
| JP2006286606A (en) * | 2005-03-08 | 2006-10-19 | Sanyo Electric Co Ltd | Nonaqueous electrolyte secondary battery |
Cited By (57)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JP2009152133A (en) * | 2007-12-21 | 2009-07-09 | Sony Corp | Nonaqueous electrolyte secondary battery |
| KR101483126B1 (en) | 2008-03-07 | 2015-01-15 | 삼성에스디아이 주식회사 | Organic eletrolyte and lithium battery using the same |
| KR101837785B1 (en) * | 2010-05-12 | 2018-03-12 | 미쯔비시 케미컬 주식회사 | Nonaqueous-electrolyte secondary battery |
| WO2011142412A1 (en) * | 2010-05-12 | 2011-11-17 | 三菱化学株式会社 | Nonaqueous-electrolyte secondary battery |
| WO2011142410A1 (en) * | 2010-05-12 | 2011-11-17 | 三菱化学株式会社 | Non-aqueous electrolytic solution, and non-aqueous electrolyte secondary battery |
| JP2012049106A (en) * | 2010-05-12 | 2012-03-08 | Mitsubishi Chemicals Corp | Nonaqueous electrolyte secondary battery |
| EP2571090A4 (en) * | 2010-05-12 | 2014-01-22 | Mitsubishi Chem Corp | Nonaqueous-electrolyte secondary battery |
| US20130071730A1 (en) * | 2010-05-12 | 2013-03-21 | Mitsubishi Chemical Corporation | Nonaqueous electrolyte secondary battery |
| EP2571090A1 (en) * | 2010-05-12 | 2013-03-20 | Mitsubishi Chemical Corporation | Nonaqueous-electrolyte secondary battery |
| KR101930558B1 (en) * | 2010-05-12 | 2018-12-18 | 미쯔비시 케미컬 주식회사 | Non-aqueous electrolytic solution, and non-aqueous electrolyte secondary battery |
| JP2012054231A (en) * | 2010-08-05 | 2012-03-15 | Mitsubishi Chemicals Corp | Nonaqueous electrolyte and nonaqueous electrolyte battery |
| WO2012035821A1 (en) * | 2010-09-16 | 2012-03-22 | 三菱化学株式会社 | Nonaqueous electrolyte and nonaqueous electrolyte secondary battery |
| CN103109410A (en) * | 2010-09-16 | 2013-05-15 | 三菱化学株式会社 | Nonaqueous electrolyte and nonaqueous electrolyte secondary battery |
| EP2618418A1 (en) * | 2010-09-16 | 2013-07-24 | Mitsubishi Chemical Corporation | Nonaqueous electrolyte and nonaqueous electrolyte secondary battery |
| US9553333B2 (en) | 2010-09-16 | 2017-01-24 | Mitsubishi Chemical Corporation | Nonaqueous electrolytic solution and nonaqueous electrolyte secondary battery |
| JP2012064473A (en) * | 2010-09-16 | 2012-03-29 | Mitsubishi Chemicals Corp | Nonaqueous electrolyte and nonaqueous electrolyte battery |
| EP2618418A4 (en) * | 2010-09-16 | 2014-01-22 | Mitsubishi Chem Corp | Nonaqueous electrolyte and nonaqueous electrolyte secondary battery |
| JP2012094501A (en) * | 2010-09-28 | 2012-05-17 | Mitsubishi Chemicals Corp | Nonaqueous electrolyte secondary battery |
| JP2012109240A (en) * | 2010-10-29 | 2012-06-07 | Mitsubishi Chemicals Corp | Nonaqueous electrolyte secondary battery |
| WO2012067248A1 (en) * | 2010-11-19 | 2012-05-24 | 三菱化学株式会社 | Process for producing 4-alkynyl-1,3-dioxolan-2-one derivative |
| JP6098507B2 (en) * | 2011-04-11 | 2017-03-22 | 宇部興産株式会社 | Nonaqueous electrolytic solution for lithium battery or lithium ion capacitor and power storage device using the same |
| JPWO2012141001A1 (en) * | 2011-04-11 | 2014-07-28 | 宇部興産株式会社 | Non-aqueous electrolyte and power storage device using the same |
| CN103477492A (en) * | 2011-04-11 | 2013-12-25 | 宇部兴产株式会社 | Non-aqueous electrolyte solution and electricity-storage device using same |
| WO2012141001A1 (en) * | 2011-04-11 | 2012-10-18 | 宇部興産株式会社 | Non-aqueous electrolyte solution and electricity-storage device using same |
| US9362595B2 (en) | 2011-04-11 | 2016-06-07 | Ube Industries, Ltd. | Nonaqueous electrolytic solution and energy storage device using same |
| JP2012227060A (en) * | 2011-04-21 | 2012-11-15 | Mitsubishi Chemicals Corp | Nonaqueous electrolyte and nonaqueous electrolyte battery using the same |
| WO2012147502A1 (en) * | 2011-04-27 | 2012-11-01 | 昭和電工株式会社 | Non-aqueous electrolyte solution for secondary cell, and non-aqueous electrolyte secondary cell |
| JPWO2013058235A1 (en) * | 2011-10-17 | 2015-04-02 | 宇部興産株式会社 | Non-aqueous electrolyte and power storage device using the same |
| JP2017112123A (en) * | 2011-11-11 | 2017-06-22 | 三菱化学株式会社 | Nonaqueous electrolyte secondary battery and nonaqueous electrolyte |
| JP2013239426A (en) * | 2011-11-11 | 2013-11-28 | Mitsubishi Chemicals Corp | Nonaqueous electrolyte secondary battery and nonaqueous electrolyte |
| JP2018190739A (en) * | 2011-11-11 | 2018-11-29 | 三菱ケミカル株式会社 | Nonaqueous electrolytic secondary battery, and nonaqueous electrolyte solution |
| CN103794817A (en) * | 2014-02-20 | 2014-05-14 | 福建创鑫科技开发有限公司 | Application of vinyl ethylene carbonate to lithium ion battery |
| US20190334207A1 (en) * | 2017-01-23 | 2019-10-31 | Lg Chem, Ltd. | Additive For Non-aqueous Electrolyte Solution, And Non-aqueous Electrolyte Solution For Lithium Secondary Battery And Lithium Secondary Battery Which Include The Same |
| US10923768B2 (en) * | 2017-01-23 | 2021-02-16 | Lg Chem, Ltd. | Alkynyl-containing compound additive for non-aqueous electrolyte solution, and non-aqueous electrolyte solution for lithium secondary battery and lithium secondary battery which include the same |
| EP4053959A1 (en) * | 2017-11-21 | 2022-09-07 | Daikin Industries, Ltd. | Electrolytic solution, electrochemical device, lithium ion secondary battery, and module |
| JP7048904B2 (en) | 2017-11-21 | 2022-04-06 | ダイキン工業株式会社 | Electrolytes, electrochemical devices, lithium-ion secondary batteries and modules |
| CN111373591A (en) * | 2017-11-21 | 2020-07-03 | 大金工业株式会社 | Electrolyte solution, electrochemical device, lithium ion secondary battery, and assembly |
| JPWO2019102722A1 (en) * | 2017-11-21 | 2020-09-10 | ダイキン工業株式会社 | Electrolyte, electrochemical device, lithium ion secondary battery and module |
| CN111373591B (en) * | 2017-11-21 | 2023-11-03 | 大金工业株式会社 | Electrolyte, electrochemical device, lithium ion secondary battery and assembly |
| JP7307361B2 (en) | 2017-11-21 | 2023-07-12 | ダイキン工業株式会社 | Electrolyte, electrochemical device, lithium ion secondary battery and module |
| US11664534B2 (en) | 2017-11-21 | 2023-05-30 | Daikin Industries, Ltd. | Electrolytic solution, electrochemical device, lithium ion secondary battery, and module |
| EP3703173A4 (en) * | 2017-11-21 | 2021-08-18 | Daikin Industries, Ltd. | Electrolytic solution, electrochemical device, lithium ion secondary battery, and module |
| JP2022037007A (en) * | 2017-11-21 | 2022-03-08 | ダイキン工業株式会社 | Electrolyte, electrochemical device, lithium-ion secondary battery, and module |
| WO2019102722A1 (en) * | 2017-11-21 | 2019-05-31 | ダイキン工業株式会社 | Electrolytic solution, electrochemical device, lithium ion secondary battery, and module |
| CN111656595A (en) * | 2018-01-30 | 2020-09-11 | 大金工业株式会社 | Electrolyte solution, electrochemical device, lithium ion secondary battery, and assembly |
| CN111656595B (en) * | 2018-01-30 | 2024-01-05 | 大金工业株式会社 | Electrolyte, electrochemical device, lithium ion secondary battery and assembly |
| KR102470813B1 (en) * | 2018-03-27 | 2022-11-28 | 다이킨 고교 가부시키가이샤 | Electrolyte, electrochemical device, lithium ion secondary battery and module |
| JP7161122B2 (en) | 2018-03-27 | 2022-10-26 | ダイキン工業株式会社 | Electrolyte, electrochemical device, lithium ion secondary battery and module |
| WO2019188210A1 (en) * | 2018-03-27 | 2019-10-03 | ダイキン工業株式会社 | Electrolyte solution, electrochemical device, lithium ion secondary battery, and module |
| JPWO2019188210A1 (en) * | 2018-03-27 | 2021-01-14 | ダイキン工業株式会社 | Electrolyte, electrochemical device, lithium ion secondary battery and module |
| CN111937215A (en) * | 2018-03-27 | 2020-11-13 | 大金工业株式会社 | Electrolyte solution, electrochemical device, lithium ion secondary battery, and module |
| KR20200121358A (en) * | 2018-03-27 | 2020-10-23 | 다이킨 고교 가부시키가이샤 | Electrolyte, electrochemical device, lithium ion secondary battery and module |
| US11916195B2 (en) | 2018-03-27 | 2024-02-27 | Daikin Industries, Ltd. | Electrolyte solution, electrochemical device, lithium ion secondary battery, and module |
| CN111937215B (en) * | 2018-03-27 | 2024-04-19 | 大金工业株式会社 | Electrolyte, electrochemical device, lithium ion secondary battery, and assembly |
| WO2019207983A1 (en) * | 2018-04-25 | 2019-10-31 | ダイキン工業株式会社 | Electrolytic solution, electrochemical device, lithium-ion secondary battery, and module |
| CN114361590A (en) * | 2021-12-17 | 2022-04-15 | 松山湖材料实验室 | Lithium ion battery electrolyte additive, electrolyte and lithium ion battery |
| CN114243111A (en) * | 2021-12-17 | 2022-03-25 | 松山湖材料实验室 | Lithium ion battery electrolyte additive, electrolyte and lithium ion battery |
Also Published As
| Publication number | Publication date |
|---|---|
| JP5160744B2 (en) | 2013-03-13 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| JP5160744B2 (en) | Nonaqueous electrolyte secondary battery | |
| JP4703203B2 (en) | Nonaqueous electrolyte secondary battery | |
| JP5738342B2 (en) | Method for suppressing reduction in life of redox shuttle agent | |
| JP4779651B2 (en) | Non-aqueous electrolyte and lithium secondary battery | |
| JP6396153B2 (en) | Lithium secondary battery | |
| JP7005587B2 (en) | Non-aqueous electrolyte composition | |
| JP4012174B2 (en) | Lithium battery with efficient performance | |
| JP2006216378A5 (en) | ||
| JP2001332297A (en) | Nonaqueous electrolytic solution and lithium secondary battery using the same | |
| JP2007504619A (en) | Non-aqueous lithium secondary battery with improved cycle characteristics and / or high temperature stability | |
| JP5474785B2 (en) | Non-aqueous electrolyte and secondary battery equipped with the same | |
| JP2003059529A (en) | Nonaqueous electrolyte solution and lithium secondary battery using same | |
| JP2009105069A (en) | Electrolyte for lithium secondary battery, and lithium secondary battery containing same | |
| JP5062459B2 (en) | Nonaqueous electrolyte secondary battery | |
| JP5235405B2 (en) | Nonaqueous electrolyte secondary battery | |
| JP2016048624A (en) | Lithium secondary battery | |
| JP2007173014A (en) | Nonaqueous electrolyte secondary battery | |
| JP2005268017A (en) | Nonaqueous electrolyte secondary battery | |
| JP6361113B2 (en) | Lithium ion secondary battery | |
| JP6755182B2 (en) | Lithium ion secondary battery | |
| JP4431941B2 (en) | Nonaqueous electrolyte secondary battery | |
| JP4765629B2 (en) | Non-aqueous electrolyte and lithium secondary battery | |
| JP5376142B2 (en) | Nonaqueous electrolyte and lithium battery using the same | |
| JP6224382B2 (en) | Lithium secondary battery | |
| JP2003317803A (en) | Nonaqueous electrolytic solution, and lithium secondary cell using the same |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| A621 | Written request for application examination |
Free format text: JAPANESE INTERMEDIATE CODE: A621 Effective date: 20080514 |
|
| A977 | Report on retrieval |
Free format text: JAPANESE INTERMEDIATE CODE: A971007 Effective date: 20110128 |
|
| A711 | Notification of change in applicant |
Free format text: JAPANESE INTERMEDIATE CODE: A711 Effective date: 20110405 |
|
| A521 | Written amendment |
Free format text: JAPANESE INTERMEDIATE CODE: A523 Effective date: 20110520 |
|
| RD02 | Notification of acceptance of power of attorney |
Free format text: JAPANESE INTERMEDIATE CODE: A7422 Effective date: 20110720 |
|
| A131 | Notification of reasons for refusal |
Free format text: JAPANESE INTERMEDIATE CODE: A131 Effective date: 20110823 |
|
| A521 | Written amendment |
Free format text: JAPANESE INTERMEDIATE CODE: A523 Effective date: 20111007 |
|
| A131 | Notification of reasons for refusal |
Free format text: JAPANESE INTERMEDIATE CODE: A131 Effective date: 20120508 |
|
| A521 | Written amendment |
Free format text: JAPANESE INTERMEDIATE CODE: A523 Effective date: 20120625 |
|
| TRDD | Decision of grant or rejection written | ||
| A01 | Written decision to grant a patent or to grant a registration (utility model) |
Free format text: JAPANESE INTERMEDIATE CODE: A01 Effective date: 20121127 |
|
| A61 | First payment of annual fees (during grant procedure) |
Free format text: JAPANESE INTERMEDIATE CODE: A61 Effective date: 20121213 |
|
| R150 | Certificate of patent or registration of utility model |
Ref document number: 5160744 Country of ref document: JP Free format text: JAPANESE INTERMEDIATE CODE: R150 Free format text: JAPANESE INTERMEDIATE CODE: R150 |
|
| FPAY | Renewal fee payment (event date is renewal date of database) |
Free format text: PAYMENT UNTIL: 20151221 Year of fee payment: 3 |
|
| S531 | Written request for registration of change of domicile |
Free format text: JAPANESE INTERMEDIATE CODE: R313531 |
|
| S111 | Request for change of ownership or part of ownership |
Free format text: JAPANESE INTERMEDIATE CODE: R313117 |
|
| R350 | Written notification of registration of transfer |
Free format text: JAPANESE INTERMEDIATE CODE: R350 |
|
| R350 | Written notification of registration of transfer |
Free format text: JAPANESE INTERMEDIATE CODE: R350 |
|
| LAPS | Cancellation because of no payment of annual fees |