JP2005293750A - Method and device for recording and reproduction, and magnetic recording medium - Google Patents
Method and device for recording and reproduction, and magnetic recording medium Download PDFInfo
- Publication number
- JP2005293750A JP2005293750A JP2004109035A JP2004109035A JP2005293750A JP 2005293750 A JP2005293750 A JP 2005293750A JP 2004109035 A JP2004109035 A JP 2004109035A JP 2004109035 A JP2004109035 A JP 2004109035A JP 2005293750 A JP2005293750 A JP 2005293750A
- Authority
- JP
- Japan
- Prior art keywords
- recording medium
- magnetic
- recording
- magnetic recording
- information signal
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Pending
Links
- 230000005291 magnetic effect Effects 0.000 title claims abstract description 122
- 238000000034 method Methods 0.000 title claims abstract description 35
- 230000004044 response Effects 0.000 claims abstract description 33
- 229910000859 α-Fe Inorganic materials 0.000 claims description 24
- AJCDFVKYMIUXCR-UHFFFAOYSA-N oxobarium;oxo(oxoferriooxy)iron Chemical group [Ba]=O.O=[Fe]O[Fe]=O.O=[Fe]O[Fe]=O.O=[Fe]O[Fe]=O.O=[Fe]O[Fe]=O.O=[Fe]O[Fe]=O.O=[Fe]O[Fe]=O AJCDFVKYMIUXCR-UHFFFAOYSA-N 0.000 claims description 20
- 238000003672 processing method Methods 0.000 claims description 10
- 239000010410 layer Substances 0.000 description 62
- 230000033458 reproduction Effects 0.000 description 33
- 239000000843 powder Substances 0.000 description 31
- 239000000696 magnetic material Substances 0.000 description 26
- 230000005294 ferromagnetic effect Effects 0.000 description 15
- 229910052751 metal Inorganic materials 0.000 description 13
- 239000002184 metal Substances 0.000 description 13
- ZWEHNKRNPOVVGH-UHFFFAOYSA-N 2-Butanone Chemical compound CCC(C)=O ZWEHNKRNPOVVGH-UHFFFAOYSA-N 0.000 description 12
- 239000011248 coating agent Substances 0.000 description 10
- 238000000576 coating method Methods 0.000 description 10
- 239000002245 particle Substances 0.000 description 10
- 239000006185 dispersion Substances 0.000 description 9
- 230000005415 magnetization Effects 0.000 description 7
- 238000007476 Maximum Likelihood Methods 0.000 description 5
- 238000009826 distribution Methods 0.000 description 5
- -1 polyethylene terephthalate Polymers 0.000 description 5
- 239000011701 zinc Substances 0.000 description 5
- UQSXHKLRYXJYBZ-UHFFFAOYSA-N Iron oxide Chemical compound [Fe]=O UQSXHKLRYXJYBZ-UHFFFAOYSA-N 0.000 description 4
- 229910045601 alloy Inorganic materials 0.000 description 4
- 239000000956 alloy Substances 0.000 description 4
- JHIVVAPYMSGYDF-UHFFFAOYSA-N cyclohexanone Chemical compound O=C1CCCCC1 JHIVVAPYMSGYDF-UHFFFAOYSA-N 0.000 description 4
- XEEYBQQBJWHFJM-UHFFFAOYSA-N iron Substances [Fe] XEEYBQQBJWHFJM-UHFFFAOYSA-N 0.000 description 4
- 239000006247 magnetic powder Substances 0.000 description 4
- 238000004519 manufacturing process Methods 0.000 description 4
- 229910052725 zinc Inorganic materials 0.000 description 4
- KDLHZDBZIXYQEI-UHFFFAOYSA-N Palladium Chemical compound [Pd] KDLHZDBZIXYQEI-UHFFFAOYSA-N 0.000 description 3
- 229910052782 aluminium Inorganic materials 0.000 description 3
- 239000006229 carbon black Substances 0.000 description 3
- 229910017052 cobalt Inorganic materials 0.000 description 3
- 239000010941 cobalt Substances 0.000 description 3
- GUTLYIVDDKVIGB-UHFFFAOYSA-N cobalt atom Chemical compound [Co] GUTLYIVDDKVIGB-UHFFFAOYSA-N 0.000 description 3
- 239000007788 liquid Substances 0.000 description 3
- 239000000203 mixture Substances 0.000 description 3
- 229920005749 polyurethane resin Polymers 0.000 description 3
- 230000008569 process Effects 0.000 description 3
- 229910052712 strontium Inorganic materials 0.000 description 3
- CIOAGBVUUVVLOB-UHFFFAOYSA-N strontium atom Chemical compound [Sr] CIOAGBVUUVVLOB-UHFFFAOYSA-N 0.000 description 3
- 229910052718 tin Inorganic materials 0.000 description 3
- 239000011135 tin Substances 0.000 description 3
- 238000007740 vapor deposition Methods 0.000 description 3
- QMMJWQMCMRUYTG-UHFFFAOYSA-N 1,2,4,5-tetrachloro-3-(trifluoromethyl)benzene Chemical compound FC(F)(F)C1=C(Cl)C(Cl)=CC(Cl)=C1Cl QMMJWQMCMRUYTG-UHFFFAOYSA-N 0.000 description 2
- 238000004438 BET method Methods 0.000 description 2
- VTYYLEPIZMXCLO-UHFFFAOYSA-L Calcium carbonate Chemical compound [Ca+2].[O-]C([O-])=O VTYYLEPIZMXCLO-UHFFFAOYSA-L 0.000 description 2
- OKTJSMMVPCPJKN-UHFFFAOYSA-N Carbon Chemical compound [C] OKTJSMMVPCPJKN-UHFFFAOYSA-N 0.000 description 2
- 229910052684 Cerium Inorganic materials 0.000 description 2
- 229910052779 Neodymium Inorganic materials 0.000 description 2
- 229910052777 Praseodymium Inorganic materials 0.000 description 2
- 235000021355 Stearic acid Nutrition 0.000 description 2
- MCMNRKCIXSYSNV-UHFFFAOYSA-N Zirconium dioxide Chemical compound O=[Zr]=O MCMNRKCIXSYSNV-UHFFFAOYSA-N 0.000 description 2
- XAGFODPZIPBFFR-UHFFFAOYSA-N aluminium Chemical compound [Al] XAGFODPZIPBFFR-UHFFFAOYSA-N 0.000 description 2
- 229910052787 antimony Inorganic materials 0.000 description 2
- 229910052797 bismuth Inorganic materials 0.000 description 2
- 229910052796 boron Inorganic materials 0.000 description 2
- 229910052799 carbon Inorganic materials 0.000 description 2
- 238000006243 chemical reaction Methods 0.000 description 2
- 229910052804 chromium Inorganic materials 0.000 description 2
- 239000011651 chromium Substances 0.000 description 2
- 229910052802 copper Inorganic materials 0.000 description 2
- 239000010949 copper Substances 0.000 description 2
- 239000013078 crystal Substances 0.000 description 2
- 238000001035 drying Methods 0.000 description 2
- 238000005516 engineering process Methods 0.000 description 2
- 238000009472 formulation Methods 0.000 description 2
- 229910052737 gold Inorganic materials 0.000 description 2
- 239000010931 gold Substances 0.000 description 2
- 229910052746 lanthanum Inorganic materials 0.000 description 2
- 150000002739 metals Chemical class 0.000 description 2
- 229910052750 molybdenum Inorganic materials 0.000 description 2
- 229910052759 nickel Inorganic materials 0.000 description 2
- PXHVJJICTQNCMI-UHFFFAOYSA-N nickel Substances [Ni] PXHVJJICTQNCMI-UHFFFAOYSA-N 0.000 description 2
- 229910052758 niobium Inorganic materials 0.000 description 2
- QIQXTHQIDYTFRH-UHFFFAOYSA-N octadecanoic acid Chemical compound CCCCCCCCCCCCCCCCCC(O)=O QIQXTHQIDYTFRH-UHFFFAOYSA-N 0.000 description 2
- OQCDKBAXFALNLD-UHFFFAOYSA-N octadecanoic acid Natural products CCCCCCCC(C)CCCCCCCCC(O)=O OQCDKBAXFALNLD-UHFFFAOYSA-N 0.000 description 2
- 229910052763 palladium Inorganic materials 0.000 description 2
- 229910052698 phosphorus Inorganic materials 0.000 description 2
- 229920003207 poly(ethylene-2,6-naphthalate) Polymers 0.000 description 2
- 239000011112 polyethylene naphthalate Substances 0.000 description 2
- 229920001228 polyisocyanate Polymers 0.000 description 2
- 239000005056 polyisocyanate Substances 0.000 description 2
- 229910052702 rhenium Inorganic materials 0.000 description 2
- 229910052703 rhodium Inorganic materials 0.000 description 2
- 239000010948 rhodium Substances 0.000 description 2
- 229910052710 silicon Inorganic materials 0.000 description 2
- 229910052709 silver Inorganic materials 0.000 description 2
- 239000011029 spinel Substances 0.000 description 2
- 229910052596 spinel Inorganic materials 0.000 description 2
- 239000008117 stearic acid Substances 0.000 description 2
- 229910052717 sulfur Inorganic materials 0.000 description 2
- 229910052719 titanium Inorganic materials 0.000 description 2
- 239000010936 titanium Substances 0.000 description 2
- 229910052720 vanadium Inorganic materials 0.000 description 2
- 229910052727 yttrium Inorganic materials 0.000 description 2
- 229910018072 Al 2 O 3 Inorganic materials 0.000 description 1
- PNEYBMLMFCGWSK-UHFFFAOYSA-N Alumina Chemical compound [O-2].[O-2].[O-2].[Al+3].[Al+3] PNEYBMLMFCGWSK-UHFFFAOYSA-N 0.000 description 1
- ZOXJGFHDIHLPTG-UHFFFAOYSA-N Boron Chemical compound [B] ZOXJGFHDIHLPTG-UHFFFAOYSA-N 0.000 description 1
- VYZAMTAEIAYCRO-UHFFFAOYSA-N Chromium Chemical compound [Cr] VYZAMTAEIAYCRO-UHFFFAOYSA-N 0.000 description 1
- 229910020630 Co Ni Inorganic materials 0.000 description 1
- RYGMFSIKBFXOCR-UHFFFAOYSA-N Copper Chemical compound [Cu] RYGMFSIKBFXOCR-UHFFFAOYSA-N 0.000 description 1
- 229910002440 Co–Ni Inorganic materials 0.000 description 1
- 229910020517 Co—Ti Inorganic materials 0.000 description 1
- 229910020521 Co—Zn Inorganic materials 0.000 description 1
- 229910017061 Fe Co Inorganic materials 0.000 description 1
- 229910001030 Iron–nickel alloy Inorganic materials 0.000 description 1
- ZOKXTWBITQBERF-UHFFFAOYSA-N Molybdenum Chemical compound [Mo] ZOKXTWBITQBERF-UHFFFAOYSA-N 0.000 description 1
- 229910003271 Ni-Fe Inorganic materials 0.000 description 1
- 239000000020 Nitrocellulose Substances 0.000 description 1
- 240000007594 Oryza sativa Species 0.000 description 1
- 235000007164 Oryza sativa Nutrition 0.000 description 1
- QLZHNIAADXEJJP-UHFFFAOYSA-N Phenylphosphonic acid Chemical compound OP(O)(=O)C1=CC=CC=C1 QLZHNIAADXEJJP-UHFFFAOYSA-N 0.000 description 1
- OAICVXFJPJFONN-UHFFFAOYSA-N Phosphorus Chemical compound [P] OAICVXFJPJFONN-UHFFFAOYSA-N 0.000 description 1
- 239000004952 Polyamide Substances 0.000 description 1
- 239000004962 Polyamide-imide Substances 0.000 description 1
- 239000004695 Polyether sulfone Substances 0.000 description 1
- 239000004698 Polyethylene Substances 0.000 description 1
- 239000004642 Polyimide Substances 0.000 description 1
- 239000004743 Polypropylene Substances 0.000 description 1
- 108010076504 Protein Sorting Signals Proteins 0.000 description 1
- XUIMIQQOPSSXEZ-UHFFFAOYSA-N Silicon Chemical compound [Si] XUIMIQQOPSSXEZ-UHFFFAOYSA-N 0.000 description 1
- BQCADISMDOOEFD-UHFFFAOYSA-N Silver Chemical compound [Ag] BQCADISMDOOEFD-UHFFFAOYSA-N 0.000 description 1
- 229910019146 Sn—Co—Ti Inorganic materials 0.000 description 1
- 229910009075 Sn—Zn—Co Inorganic materials 0.000 description 1
- NINIDFKCEFEMDL-UHFFFAOYSA-N Sulfur Chemical compound [S] NINIDFKCEFEMDL-UHFFFAOYSA-N 0.000 description 1
- ATJFFYVFTNAWJD-UHFFFAOYSA-N Tin Chemical compound [Sn] ATJFFYVFTNAWJD-UHFFFAOYSA-N 0.000 description 1
- RTAQQCXQSZGOHL-UHFFFAOYSA-N Titanium Chemical compound [Ti] RTAQQCXQSZGOHL-UHFFFAOYSA-N 0.000 description 1
- HCHKCACWOHOZIP-UHFFFAOYSA-N Zinc Chemical compound [Zn] HCHKCACWOHOZIP-UHFFFAOYSA-N 0.000 description 1
- 229910007564 Zn—Co Inorganic materials 0.000 description 1
- 238000010521 absorption reaction Methods 0.000 description 1
- 239000002253 acid Substances 0.000 description 1
- 239000012790 adhesive layer Substances 0.000 description 1
- JZQOJFLIJNRDHK-CMDGGOBGSA-N alpha-irone Chemical compound CC1CC=C(C)C(\C=C\C(C)=O)C1(C)C JZQOJFLIJNRDHK-CMDGGOBGSA-N 0.000 description 1
- WATWJIUSRGPENY-UHFFFAOYSA-N antimony atom Chemical compound [Sb] WATWJIUSRGPENY-UHFFFAOYSA-N 0.000 description 1
- 229910052788 barium Inorganic materials 0.000 description 1
- DSAJWYNOEDNPEQ-UHFFFAOYSA-N barium atom Chemical compound [Ba] DSAJWYNOEDNPEQ-UHFFFAOYSA-N 0.000 description 1
- 239000011324 bead Substances 0.000 description 1
- JCXGWMGPZLAOME-UHFFFAOYSA-N bismuth atom Chemical compound [Bi] JCXGWMGPZLAOME-UHFFFAOYSA-N 0.000 description 1
- 229910000019 calcium carbonate Inorganic materials 0.000 description 1
- WETINTNJFLGREW-UHFFFAOYSA-N calcium;iron;tetrahydrate Chemical compound O.O.O.O.[Ca].[Fe].[Fe] WETINTNJFLGREW-UHFFFAOYSA-N 0.000 description 1
- 238000004364 calculation method Methods 0.000 description 1
- 230000015556 catabolic process Effects 0.000 description 1
- GWXLDORMOJMVQZ-UHFFFAOYSA-N cerium Chemical compound [Ce] GWXLDORMOJMVQZ-UHFFFAOYSA-N 0.000 description 1
- 230000008859 change Effects 0.000 description 1
- 238000004140 cleaning Methods 0.000 description 1
- 239000002131 composite material Substances 0.000 description 1
- 229920001577 copolymer Polymers 0.000 description 1
- 238000006731 degradation reaction Methods 0.000 description 1
- 238000010586 diagram Methods 0.000 description 1
- 239000004744 fabric Substances 0.000 description 1
- 230000002349 favourable effect Effects 0.000 description 1
- 229910052732 germanium Inorganic materials 0.000 description 1
- PCHJSUWPFVWCPO-UHFFFAOYSA-N gold Chemical compound [Au] PCHJSUWPFVWCPO-UHFFFAOYSA-N 0.000 description 1
- 238000010438 heat treatment Methods 0.000 description 1
- XLYOFNOQVPJJNP-UHFFFAOYSA-M hydroxide Chemical compound [OH-] XLYOFNOQVPJJNP-UHFFFAOYSA-M 0.000 description 1
- 230000006872 improvement Effects 0.000 description 1
- 239000012535 impurity Substances 0.000 description 1
- 229910052742 iron Inorganic materials 0.000 description 1
- XMNVMZIXNKZAJB-UHFFFAOYSA-N iron(3+);lead(2+);oxygen(2-) Chemical compound [O-2].[O-2].[O-2].[O-2].[O-2].[Fe+3].[Fe+3].[Pb+2].[Pb+2] XMNVMZIXNKZAJB-UHFFFAOYSA-N 0.000 description 1
- FZLIPJUXYLNCLC-UHFFFAOYSA-N lanthanum atom Chemical compound [La] FZLIPJUXYLNCLC-UHFFFAOYSA-N 0.000 description 1
- 239000011133 lead Substances 0.000 description 1
- 229910052748 manganese Inorganic materials 0.000 description 1
- 239000011572 manganese Substances 0.000 description 1
- WPBNNNQJVZRUHP-UHFFFAOYSA-L manganese(2+);methyl n-[[2-(methoxycarbonylcarbamothioylamino)phenyl]carbamothioyl]carbamate;n-[2-(sulfidocarbothioylamino)ethyl]carbamodithioate Chemical compound [Mn+2].[S-]C(=S)NCCNC([S-])=S.COC(=O)NC(=S)NC1=CC=CC=C1NC(=S)NC(=O)OC WPBNNNQJVZRUHP-UHFFFAOYSA-L 0.000 description 1
- 239000011733 molybdenum Substances 0.000 description 1
- QEFYFXOXNSNQGX-UHFFFAOYSA-N neodymium atom Chemical compound [Nd] QEFYFXOXNSNQGX-UHFFFAOYSA-N 0.000 description 1
- 229920001220 nitrocellulos Polymers 0.000 description 1
- 239000004745 nonwoven fabric Substances 0.000 description 1
- 230000006911 nucleation Effects 0.000 description 1
- 238000010899 nucleation Methods 0.000 description 1
- 239000003921 oil Substances 0.000 description 1
- 230000003287 optical effect Effects 0.000 description 1
- 239000011574 phosphorus Substances 0.000 description 1
- 229920002492 poly(sulfone) Polymers 0.000 description 1
- 229920002647 polyamide Polymers 0.000 description 1
- 229920002312 polyamide-imide Polymers 0.000 description 1
- 229920000515 polycarbonate Polymers 0.000 description 1
- 239000004417 polycarbonate Substances 0.000 description 1
- 229920006393 polyether sulfone Polymers 0.000 description 1
- 229920000573 polyethylene Polymers 0.000 description 1
- 229920000139 polyethylene terephthalate Polymers 0.000 description 1
- 239000005020 polyethylene terephthalate Substances 0.000 description 1
- 229920001721 polyimide Polymers 0.000 description 1
- 229920001155 polypropylene Polymers 0.000 description 1
- PUDIUYLPXJFUGB-UHFFFAOYSA-N praseodymium atom Chemical compound [Pr] PUDIUYLPXJFUGB-UHFFFAOYSA-N 0.000 description 1
- 239000011164 primary particle Substances 0.000 description 1
- 239000011241 protective layer Substances 0.000 description 1
- 239000002994 raw material Substances 0.000 description 1
- 230000008929 regeneration Effects 0.000 description 1
- 238000011069 regeneration method Methods 0.000 description 1
- 229920005989 resin Polymers 0.000 description 1
- 239000011347 resin Substances 0.000 description 1
- WUAPFZMCVAUBPE-UHFFFAOYSA-N rhenium atom Chemical compound [Re] WUAPFZMCVAUBPE-UHFFFAOYSA-N 0.000 description 1
- MHOVAHRLVXNVSD-UHFFFAOYSA-N rhodium atom Chemical compound [Rh] MHOVAHRLVXNVSD-UHFFFAOYSA-N 0.000 description 1
- 235000009566 rice Nutrition 0.000 description 1
- 230000000630 rising effect Effects 0.000 description 1
- 239000004576 sand Substances 0.000 description 1
- 229910052706 scandium Inorganic materials 0.000 description 1
- SIXSYDAISGFNSX-UHFFFAOYSA-N scandium atom Chemical compound [Sc] SIXSYDAISGFNSX-UHFFFAOYSA-N 0.000 description 1
- 239000010703 silicon Substances 0.000 description 1
- 239000004332 silver Substances 0.000 description 1
- 239000010944 silver (metal) Substances 0.000 description 1
- 229910001220 stainless steel Inorganic materials 0.000 description 1
- 239000010935 stainless steel Substances 0.000 description 1
- 238000006467 substitution reaction Methods 0.000 description 1
- 125000000542 sulfonic acid group Chemical group 0.000 description 1
- 239000011593 sulfur Substances 0.000 description 1
- 239000002335 surface treatment layer Substances 0.000 description 1
- 229920003002 synthetic resin Polymers 0.000 description 1
- 239000000057 synthetic resin Substances 0.000 description 1
- 229910052715 tantalum Inorganic materials 0.000 description 1
- GUVRBAGPIYLISA-UHFFFAOYSA-N tantalum atom Chemical compound [Ta] GUVRBAGPIYLISA-UHFFFAOYSA-N 0.000 description 1
- 229910052714 tellurium Inorganic materials 0.000 description 1
- PORWMNRCUJJQNO-UHFFFAOYSA-N tellurium atom Chemical compound [Te] PORWMNRCUJJQNO-UHFFFAOYSA-N 0.000 description 1
- 229910052721 tungsten Inorganic materials 0.000 description 1
- WFKWXMTUELFFGS-UHFFFAOYSA-N tungsten Chemical compound [W] WFKWXMTUELFFGS-UHFFFAOYSA-N 0.000 description 1
- 239000010937 tungsten Substances 0.000 description 1
- LEONUFNNVUYDNQ-UHFFFAOYSA-N vanadium atom Chemical compound [V] LEONUFNNVUYDNQ-UHFFFAOYSA-N 0.000 description 1
- 238000004017 vitrification Methods 0.000 description 1
- XLYOFNOQVPJJNP-UHFFFAOYSA-N water Substances O XLYOFNOQVPJJNP-UHFFFAOYSA-N 0.000 description 1
- 238000004804 winding Methods 0.000 description 1
- VWQVUPCCIRVNHF-UHFFFAOYSA-N yttrium atom Chemical compound [Y] VWQVUPCCIRVNHF-UHFFFAOYSA-N 0.000 description 1
Images
Classifications
-
- G—PHYSICS
- G11—INFORMATION STORAGE
- G11B—INFORMATION STORAGE BASED ON RELATIVE MOVEMENT BETWEEN RECORD CARRIER AND TRANSDUCER
- G11B20/00—Signal processing not specific to the method of recording or reproducing; Circuits therefor
- G11B20/10—Digital recording or reproducing
- G11B20/10009—Improvement or modification of read or write signals
- G11B20/10305—Improvement or modification of read or write signals signal quality assessment
- G11B20/10324—Improvement or modification of read or write signals signal quality assessment asymmetry of the recorded or reproduced waveform
- G11B20/10333—Improvement or modification of read or write signals signal quality assessment asymmetry of the recorded or reproduced waveform wherein the asymmetry is linked to domain bloom
-
- G—PHYSICS
- G11—INFORMATION STORAGE
- G11B—INFORMATION STORAGE BASED ON RELATIVE MOVEMENT BETWEEN RECORD CARRIER AND TRANSDUCER
- G11B20/00—Signal processing not specific to the method of recording or reproducing; Circuits therefor
- G11B20/10—Digital recording or reproducing
- G11B20/10009—Improvement or modification of read or write signals
-
- G—PHYSICS
- G11—INFORMATION STORAGE
- G11B—INFORMATION STORAGE BASED ON RELATIVE MOVEMENT BETWEEN RECORD CARRIER AND TRANSDUCER
- G11B20/00—Signal processing not specific to the method of recording or reproducing; Circuits therefor
- G11B20/10—Digital recording or reproducing
- G11B20/10009—Improvement or modification of read or write signals
- G11B20/10046—Improvement or modification of read or write signals filtering or equalising, e.g. setting the tap weights of an FIR filter
-
- G—PHYSICS
- G11—INFORMATION STORAGE
- G11B—INFORMATION STORAGE BASED ON RELATIVE MOVEMENT BETWEEN RECORD CARRIER AND TRANSDUCER
- G11B20/00—Signal processing not specific to the method of recording or reproducing; Circuits therefor
- G11B20/10—Digital recording or reproducing
- G11B20/10009—Improvement or modification of read or write signals
- G11B20/10046—Improvement or modification of read or write signals filtering or equalising, e.g. setting the tap weights of an FIR filter
- G11B20/10055—Improvement or modification of read or write signals filtering or equalising, e.g. setting the tap weights of an FIR filter using partial response filtering when writing the signal to the medium or reading it therefrom
-
- G—PHYSICS
- G11—INFORMATION STORAGE
- G11B—INFORMATION STORAGE BASED ON RELATIVE MOVEMENT BETWEEN RECORD CARRIER AND TRANSDUCER
- G11B5/00—Recording by magnetisation or demagnetisation of a record carrier; Reproducing by magnetic means; Record carriers therefor
- G11B5/02—Recording, reproducing, or erasing methods; Read, write or erase circuits therefor
- G11B5/027—Analogue recording
- G11B5/035—Equalising
-
- G—PHYSICS
- G11—INFORMATION STORAGE
- G11B—INFORMATION STORAGE BASED ON RELATIVE MOVEMENT BETWEEN RECORD CARRIER AND TRANSDUCER
- G11B5/00—Recording by magnetisation or demagnetisation of a record carrier; Reproducing by magnetic means; Record carriers therefor
- G11B5/62—Record carriers characterised by the selection of the material
- G11B5/68—Record carriers characterised by the selection of the material comprising one or more layers of magnetisable material homogeneously mixed with a bonding agent
- G11B5/70—Record carriers characterised by the selection of the material comprising one or more layers of magnetisable material homogeneously mixed with a bonding agent on a base layer
- G11B5/706—Record carriers characterised by the selection of the material comprising one or more layers of magnetisable material homogeneously mixed with a bonding agent on a base layer characterised by the composition of the magnetic material
- G11B5/70626—Record carriers characterised by the selection of the material comprising one or more layers of magnetisable material homogeneously mixed with a bonding agent on a base layer characterised by the composition of the magnetic material containing non-metallic substances
- G11B5/70642—Record carriers characterised by the selection of the material comprising one or more layers of magnetisable material homogeneously mixed with a bonding agent on a base layer characterised by the composition of the magnetic material containing non-metallic substances iron oxides
- G11B5/70678—Ferrites
Landscapes
- Engineering & Computer Science (AREA)
- Signal Processing (AREA)
- Magnetic Record Carriers (AREA)
- Signal Processing For Digital Recording And Reproducing (AREA)
Abstract
Description
本発明は、記録再生方法および記録再生装置ならびに磁気記録媒体に関し、特に、高記録密度の磁気記録媒体に適した記録再生方法および記録再生装置、ならびに磁気記録媒体に関する。 The present invention relates to a recording / reproducing method, a recording / reproducing apparatus, and a magnetic recording medium, and more particularly to a recording / reproducing method, a recording / reproducing apparatus, and a magnetic recording medium suitable for a high recording density magnetic recording medium.
近年、磁気記録テープや磁気ディスク等の磁気記録媒体における記録密度の向上は目覚しいものがある。そのため、記録媒体の高記録密度化に対応して、記録再生装置に関する種々の技術が提案され、また、実用化されている。例えば、記録再生ヘッドとしてMRヘッドの採用、記録媒体とヘッドとのインターフェイスの改善などに種々の技術が提案され、また、実用化されている。さらに、信号処理技術の面においても、高密度記録化に伴うS/N比の劣化を挽回するため、パーシャルレスポンス(PR:Partial Response)方式と最尤復号(ML:Maximum Likelihood)方式を組み合わせたPRML方式が、磁気ディスク、デジタルVTR、コンピュータバックアップ用磁気テープ、光ディスク等の記録媒体を用いる記録再生装置で実用化されている(特許文献1参照)。このPRML信号処理方式は、等化処理する信号系列の項数に応じて、PR4ML(PR(1,0,−1)ML)、EPR4ML(PR(1,1,−1,−1)ML)、EEPR4ML(PR(1,2,0,−2,−1)ML)、EEEPR4ML(PR(1,3,2,−2,−3,−1)ML)等の各種の方式が知られている。 In recent years, there has been a remarkable improvement in recording density in magnetic recording media such as magnetic recording tapes and magnetic disks. For this reason, various techniques relating to recording / reproducing apparatuses have been proposed and put into practical use in response to the increase in recording density of recording media. For example, various technologies have been proposed and put into practical use for adopting an MR head as a recording / reproducing head and improving an interface between a recording medium and the head. Furthermore, in terms of signal processing technology, the partial response (PR) method and the maximum likelihood decoding (ML) method are combined in order to compensate for the degradation of the S / N ratio accompanying high-density recording. The PRML system has been put into practical use in a recording / reproducing apparatus using a recording medium such as a magnetic disk, a digital VTR, a computer backup magnetic tape, an optical disk (see Patent Document 1). In this PRML signal processing method, PR4ML (PR (1, 0, -1) ML), EPR4ML (PR (1, 1, -1, -1) ML) are selected according to the number of terms of the signal sequence to be equalized. Various methods such as EEPR4ML (PR (1, 2, 0, -2, -1) ML), EEEPR4ML (PR (1, 3, 2, -2, -3, -1) ML) are known. Yes.
そして、用いる磁性体の特性に応じて、最適なパーシャルレスポンス信号処理を適用すれば、磁気記録媒体に高密度に記録されている情報信号を低エラーレートで復元できるため、有効である。例えば、磁性体として六方晶系フェライトを含む磁性層を有する磁気記録媒体は、高密度記録での再生出力が高く、また、低ノイズであるという特性を有する。しかし、六方晶系フェライトは、その結晶構造に由来して、面内配向または無配向においても垂直方向の磁化成分を有するため、孤立反転再生波形は、面内配向の孤立反転再生波形と垂直方向の孤立反転再生波形とを足し合わせた特有の波形となり、例えば、図2に示すように、非対称性の波形を示す。こうした六方晶系フェライトを磁性体として用いる場合に得られる特有の孤立反転再生波形に対して、面内方向の磁化を記録した磁気記録媒体に対して最適化された従来のPRML信号処理を適用しても、最適な信号処理ができず、高密度記録での再生出力が高く、低ノイズである、という六方晶系フェライト磁性体に特有な特性を活かすことができなかった。
そこで本発明の課題は、高記録密度の磁気記録媒体からの情報信号の再生に際して、パーシャルレスポンス信号処理方式に最適化した記録再生方法および記録再生装置を提供することにある。 Accordingly, an object of the present invention is to provide a recording / reproducing method and a recording / reproducing apparatus that are optimized for a partial response signal processing system when reproducing an information signal from a magnetic recording medium having a high recording density.
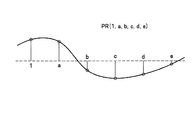
前記課題を解決するため、本発明は、磁気記録媒体に記録された情報信号をパーシャルレスポンス信号処理方式により等化復調して再生する記録再生方法であって、磁気記録媒体から再生される孤立反転再生波形の非対称性を示すγがγ>0である波形を有する場合には、パーシャルレスポンスの係数項(1,a1,a2,・・・,am(m=2nまたは2n+1:nは1以上の整数)が下記の式(1)または式(2)を満たす条件で、γがγ<0である波形を有する場合には、前記パーシャルレスポンスの係数項が下記の式(3)または式(4)を満たす条件で等化復調することを特徴とする記録再生方法を提供する。
この記録再生方法では、磁気記録媒体から再生される孤立反転再生波形の非対称性に応じて、パーシャルレスポンスの係数項(1,a1,a2,・・・,am(m=2nまたは2n+1:nは1以上の整数)の前半の項の和と、後半の項の和を前記の式(1)および式(2)または前記の式(3)および式(4)を満たすように等化復調することによって、六方晶系フェライト磁性体の特性に最適なPRML信号処理を適用することができる。 In this recording / reproducing method, partial response coefficient terms (1, a 1 , a 2 ,..., A m (m = 2n or 2n + 1) are selected according to the asymmetry of the isolated inverted reproduction waveform reproduced from the magnetic recording medium. : N is an integer of 1 or more) so that the sum of the first half term and the sum of the second half term satisfy the above formula (1) and formula (2) or the above formula (3) and formula (4), etc. By optimizing and demodulating, it is possible to apply PRML signal processing optimum for the characteristics of the hexagonal ferrite magnetic material.
また、本発明は、磁気記録媒体に記録された情報信号をパーシャルレスポンス信号処理方式により等化処理する等化手段と、等化処理された情報信号を復調する復調手段とを有する記録再生装置であって、前記磁気記録媒体から再生される孤立反転再生波形の非対称性を示すγがγ>0である波形を有する場合、前記復調手段は、前記等化手段によって等化処理された前記情報信号についてのパーシャルレスポンスの係数項(1,a1,a2,・・・,am(m=2nまたは2n+1:nは1以上の整数))が前記の式(1)または式(2)を満たす条件で復調し、前記γがγ<0である波形を有する場合には、前記復調手段は、前記等化手段によって等化処理された前記情報信号についてのパーシャルレスポンスの係数項(1,a1,a2,・・・,am(m=2nまたは2n+1:nは1以上の整数))が下記の式(3)または式(4)を満たす条件で復調することを特徴とする記録再生装置を提供する。 The present invention also relates to a recording / reproducing apparatus having an equalizing means for equalizing an information signal recorded on a magnetic recording medium by a partial response signal processing method and a demodulating means for demodulating the equalized information signal. When the γ indicating the asymmetry of the isolated inverted reproduction waveform reproduced from the magnetic recording medium has a waveform where γ> 0, the demodulating unit is configured to equalize the information signal by the equalizing unit. The coefficient terms (1, a 1 , a 2 ,..., A m (m = 2n or 2n + 1: n is an integer equal to or greater than 1)) for the partial response for the above formula (1) or (2) When demodulating under a satisfying condition and the γ has a waveform with γ <0, the demodulating means includes a partial response coefficient term (1, a) for the information signal equalized by the equalizing means. 1, a 2, ··, a m (m = 2n or 2n + 1: n is an integer of 1 or more) to provide a recording and reproducing apparatus characterized by) demodulates the condition satisfying the formula (3) or formula (4) below.
この記録再生装置では、磁気記録媒体から再生される孤立反転再生波形の非対称性に応じて、等化手段によって等化処理された前記情報信号についてのパーシャルレスポンスの係数項の前半の項の和と、後半の項の和を前記の式(1)および式(2)または前記の式(3)および式(4)を満たすように、復調手段が情報信号を復調することによって、六方晶系フェライト磁性体の特性に最適なパーシャルレスポンス信号処理を行なうことができる。 In this recording / reproducing apparatus, according to the asymmetry of the isolated inverted reproduction waveform reproduced from the magnetic recording medium, the sum of the first half of the coefficient terms of the partial response for the information signal equalized by the equalizing means The demodulating means demodulates the information signal so that the sum of the latter term satisfies the above formulas (1) and (2) or the above formulas (3) and (4). It is possible to perform the partial response signal processing optimal for the characteristics of the magnetic material.
本発明において、磁気記録媒体から再生される孤立反転再生波形の非対称性を示すγとは、孤立反転再生波形のピーク位置が前半の波形部分または後半の波形部分のいずれに偏っているかを示す指標であり、例えば、図2に示す左右非対称の孤立反転再生波形の半値幅をPW50、半値幅PW50のうちの右側の幅をPW1、左側の幅をPW2としたとき、下記式(5)で表わされる比率を言う。
γ(%)=[[(PW1)−(PW2)]÷(PW50)]×100 ・・・(5)
In the present invention, γ indicating the asymmetry of the isolated inverted reproduction waveform reproduced from the magnetic recording medium is an index indicating whether the peak position of the isolated inverted reproduction waveform is biased to the first half waveform portion or the second half waveform portion. For example, when the half-value width of the left-right asymmetric isolated inverted reproduction waveform shown in FIG. 2 is PW50, the right-side width of the half-value width PW50 is PW1, and the left-side width is PW2, the following expression (5) is expressed. Say the ratio.
γ (%) = [[(PW1) − (PW2)] ÷ (PW50)] × 100 (5)
さらに、本発明は、前記記録再生装置に用いられる磁気記録媒体を提供する。 Furthermore, the present invention provides a magnetic recording medium used for the recording / reproducing apparatus.
本発明の記録再生方法および記録再生装置は、磁気記録媒体に記録された情報信号の再生に際して、磁気記録媒体の磁性層を形成する磁性体の特性に最適なパーシャルレスポンス信号処理を行なうことによって、高密度に情報が記録された磁気記録媒体から高S/N比の再生信号を得ることができる。そのため、再生信号を低エラーレートで得ることができる。特に、六方晶系フェライト磁性体を用いる磁気記録媒の場合には、高密度記録での再生出力が高く、低ノイズである、という六方晶系フェライト磁性体の特性を活かすことができる。 The recording / reproducing method and recording / reproducing apparatus of the present invention perform partial response signal processing optimal for the characteristics of the magnetic material forming the magnetic layer of the magnetic recording medium when reproducing the information signal recorded on the magnetic recording medium, A reproduction signal having a high S / N ratio can be obtained from a magnetic recording medium on which information is recorded at high density. Therefore, a reproduction signal can be obtained at a low error rate. In particular, in the case of a magnetic recording medium using a hexagonal ferrite magnetic material, the characteristics of the hexagonal ferrite magnetic material, such as high reproduction output at high density recording and low noise, can be utilized.
また、本発明の磁気記録媒体は、前記記録再生装置において、磁性層を形成する磁性体の特性に最適なパーシャルレスポンス信号処理を行なうことによって、磁性体の特性を発揮して、高密度に情報を記録でき、かつ高S/N比の再生信号を得ることができる。そのため、再生信号を低エラーレートで得ることができる。 Further, the magnetic recording medium of the present invention exhibits the characteristics of the magnetic material by performing the partial response signal processing optimum for the characteristics of the magnetic material forming the magnetic layer in the recording / reproducing apparatus, thereby providing high-density information. Can be recorded, and a reproduction signal having a high S / N ratio can be obtained. Therefore, a reproduction signal can be obtained at a low error rate.
以下、本発明の記録再生方法および記録再生装置ならびに磁気記録媒体について説明する。
本発明の磁気記録媒体は、支持体の一方または両方の面に、記録再生装置の記録ヘッドによって情報信号が書き込まれる磁性体を含む磁性層を有し、その磁性層の下に非磁性層を有するものである。この磁気記録媒体の具体例として、磁気テープ、磁気ディスク(ハードディスク、フレキシブルディスク)、Feを主成分とする強磁性合金粉末等の磁性体を含む磁性層に磁気記録ヘッドによって情報信号を残留磁化の形態で記録することができる記録媒体が挙げられる。
The recording / reproducing method, recording / reproducing apparatus, and magnetic recording medium of the present invention will be described below.
The magnetic recording medium of the present invention has a magnetic layer containing a magnetic material to which an information signal is written by a recording head of a recording / reproducing apparatus on one or both sides of a support, and a nonmagnetic layer is formed under the magnetic layer. It is what you have. As a specific example of this magnetic recording medium, an information signal is remanently magnetized by a magnetic recording head on a magnetic layer containing a magnetic material such as a magnetic tape, a magnetic disk (hard disk, flexible disk), a ferromagnetic alloy powder containing Fe as a main component. A recording medium that can be recorded in a form is mentioned.
支持体としては、ポリエチレンテレフタレート、ポリエチレン、ポリプロピレン、ポリカーボネート、ポリエチレンナフタレート、ポリアミド、ポリアミドイミド、ポリイミド、ポリサルホン、ポリエーテルサルホンなどの各種の合成樹脂、およびアルミ、ステンレスなどの金属からなるフイルム、板等を磁気記録媒体の用途、形態等に応じて、適宜成形されたものを用いることができる。 Supports include polyethylene terephthalate, polyethylene, polypropylene, polycarbonate, polyethylene naphthalate, polyamide, polyamideimide, polyimide, polysulfone, polyethersulfone, and other synthetic resins, and films and plates made of metals such as aluminum and stainless steel. Or the like can be used according to the application, form, etc. of the magnetic recording medium.
この磁気記録媒体は、記録ヘッドまたは再生ヘッドと摺接する場合には、磁性層とは反対側の支持体の表面に摺接を円滑にするバック層を有するものが好ましい。
また、磁気記録媒体は、非磁性層、磁性層、バック層以外の層を有していてもよい。例えば、軟磁性粉末を含む軟磁性層、第2の磁性層、クッション層、オーバーコート層、接着層、保護層を有していてもよい。これらの層は、その機能を有効に発揮することができるように適切な位置に設けることができる。磁性層の厚さは、10〜300nmが好ましく、さらに好ましくは10〜200nmであり、特に好ましくは10〜100nmである。また、非磁性層は0.5〜3μmにすることができる。非磁性層の厚さは、磁性層よりも厚いことが望ましい。
When the magnetic recording medium is in sliding contact with the recording head or the reproducing head, the magnetic recording medium preferably has a back layer that makes the sliding contact smooth on the surface of the support opposite to the magnetic layer.
The magnetic recording medium may have a layer other than the nonmagnetic layer, the magnetic layer, and the back layer. For example, it may have a soft magnetic layer containing soft magnetic powder, a second magnetic layer, a cushion layer, an overcoat layer, an adhesive layer, and a protective layer. These layers can be provided at appropriate positions so that their functions can be effectively exhibited. The thickness of the magnetic layer is preferably 10 to 300 nm, more preferably 10 to 200 nm, and particularly preferably 10 to 100 nm. The nonmagnetic layer can be 0.5 to 3 μm. The nonmagnetic layer is preferably thicker than the magnetic layer.
磁性体としては、強磁性金属粉末または六方晶系フェライト粉末が用いられる。
強磁性金属粉末の具体例としては、Fe、Ni、Fe−Co、Fe−Ni、Co−Ni、Co−Ni−Fe等の金属単体または合金が挙げられ、金属成分の20質量%以下の範囲内で、アルミニウム、ケイ素、硫黄、スカンジウム、チタン、バナジウム、クロム、マンガン、銅、亜鉛、イットリウム、モリブデン、ロジウム、パラジウム、金、錫、アンチモン、ホウ素、バリウム、タンタル、タングステン、レニウム、銀、鉛、リン、ランタン、セリウム、プラセオジム、ネオジム、テルル、ビスマス等を含むことができる。また、強磁性金属粉末は、少量の水、水酸化物または酸化物を含むものであってもよい。
As the magnetic material, ferromagnetic metal powder or hexagonal ferrite powder is used.
Specific examples of the ferromagnetic metal powder include simple metals or alloys such as Fe, Ni, Fe—Co, Fe—Ni, Co—Ni, Co—Ni—Fe, and the range of 20% by mass or less of the metal component. Within, aluminum, silicon, sulfur, scandium, titanium, vanadium, chromium, manganese, copper, zinc, yttrium, molybdenum, rhodium, palladium, gold, tin, antimony, boron, barium, tantalum, tungsten, rhenium, silver, lead , Phosphorus, lanthanum, cerium, praseodymium, neodymium, tellurium, bismuth, and the like. The ferromagnetic metal powder may contain a small amount of water, hydroxide or oxide.
強磁性粉末の平均粒径サイズは、20〜60nmが好ましい。用いる強磁性粉末が、針状等である場合には、平均長軸長が30〜100nm、好ましくは35〜90nm、さらに好ましくは40〜80nmである。平均長軸長を100nm以下とすることで、ノイズが低減でき、良好な信号のSNが得られる。また、平均長軸長を30nm以上とすることで良好な保持力Hcが確保できる。粉末粒子の平均針状比は3〜10、好ましくは3〜8、さらに好ましくは4〜8である。板状である場合には、平均粉体サイズは、平均板径で表され、好ましくは25〜35nmであり、平均板状比は、2〜5が好ましい。 The average particle size of the ferromagnetic powder is preferably 20 to 60 nm. When the ferromagnetic powder to be used is needle-shaped or the like, the average major axis length is 30 to 100 nm, preferably 35 to 90 nm, and more preferably 40 to 80 nm. By setting the average major axis length to 100 nm or less, noise can be reduced and a good signal SN can be obtained. Moreover, favorable holding power Hc can be ensured by setting the average major axis length to 30 nm or more. The average acicular ratio of the powder particles is 3 to 10, preferably 3 to 8, and more preferably 4 to 8. In the case of a plate shape, the average powder size is expressed by an average plate diameter, preferably 25 to 35 nm, and the average plate ratio is preferably 2 to 5.
また、強磁性金属粉末は、SBET(BET比表面積)が通常40〜80m2/g、好ましくは50〜70m2/gであるものである。結晶子サイズは、通常、10〜25nm、好ましくは11〜22nmである。強磁性金属粉末のpHは7以上が好ましい。 The ferromagnetic metal powder has an S BET (BET specific surface area) of usually 40 to 80 m 2 / g, preferably 50 to 70 m 2 / g. The crystallite size is usually 10 to 25 nm, preferably 11 to 22 nm. The pH of the ferromagnetic metal powder is preferably 7 or more.
これらの強磁性金属粉末は、公知の方法に従って製造することができる。強磁性金属粉末の形状に特に制限はないが、通常、針状、粒状、サイコロ状、米粒状および板状のものなどが使用される。特に、針状の強磁性粉末を使用することが好ましい。 These ferromagnetic metal powders can be produced according to known methods. Although there is no restriction | limiting in particular in the shape of a ferromagnetic metal powder, Usually, a needle shape, a granular form, a dice shape, a rice granular shape, a plate shape etc. are used. In particular, it is preferable to use acicular ferromagnetic powder.
強磁性金属粉末の抗磁力Hcは、144〜300kA/mが好ましく、160〜224kA/mがさらに好ましい。また、その飽和磁化は、85〜150A・m2/kgが好ましく、100〜130A・m2/kgがさらに好ましい。 The coercive force Hc of the ferromagnetic metal powder is preferably 144 to 300 kA / m, and more preferably 160 to 224 kA / m. Further, the saturation magnetization is preferably 85~150A · m 2 / kg, more preferably 100~130A · m 2 / kg.
六方晶系フェライトとしては、バリウムフェライト、ストロンチウムフェライト、鉛フェライト、カルシウムフェライトおよびこれらの各種の各置換体、例えば、Co置換体等が挙げられる。具体的にはマグネトプランバイト型のバリウムフェライトおよびストロンチウムフェライト、スピネルで粒子表面を被覆したマグネトプランバイト型フェライト、さらに一部スピネル相を含有した複合マグネトプランバイト型のバリウムフェライトおよびストロンチウムフェライト等が挙げられ、その他所定の原子以外にAl、Si、S、Nb、Sn、Ti、V、Cr、Cu、Y、Mo、Rh、Pd、Ag、Sn、Sb、Te、W、Re、Au、Bi、La、Ce、Pr、Nd、P、Co、Mn、Zn、Ni、B、Ge、Nbなどの原子を含んでいてもよい。一般にはCo−Zn、Co−Ti、Co−Ti−Zr、Co−Ti−Zn、Ni−Ti−Zn、Nb−Zn−Co、Sn−Zn−Co、Sn−Co−Ti、Nb−Zn等の元素を添加した物を使用することができる。またW型六方晶系フェライトを用いることも可能である。さらに、原料・製法に由来する特有の不純物を含有しているものでもよい。これらの六方晶系フェライトは、六角板状の粉末の形態で用いられる。 Examples of the hexagonal ferrite include barium ferrite, strontium ferrite, lead ferrite, calcium ferrite, and various substitutes thereof, for example, Co substitutes. Specific examples include magnetoplumbite-type barium ferrite and strontium ferrite, magnetoplumbite-type ferrite whose particle surface is coated with spinel, and composite magnetoplumbite-type barium ferrite and strontium ferrite partially containing a spinel phase. In addition to predetermined atoms, Al, Si, S, Nb, Sn, Ti, V, Cr, Cu, Y, Mo, Rh, Pd, Ag, Sn, Sb, Te, W, Re, Au, Bi, It may contain atoms such as La, Ce, Pr, Nd, P, Co, Mn, Zn, Ni, B, Ge, and Nb. In general, Co-Zn, Co-Ti, Co-Ti-Zr, Co-Ti-Zn, Ni-Ti-Zn, Nb-Zn-Co, Sn-Zn-Co, Sn-Co-Ti, Nb-Zn, etc. The thing which added these elements can be used. It is also possible to use W-type hexagonal ferrite. Furthermore, the thing containing the characteristic impurity originating in a raw material and a manufacturing method may be used. These hexagonal ferrites are used in the form of hexagonal plate-like powder.
六方晶系フェライト磁性粉の平均板径を50nm以下、平均厚さを15nm以下とすることで、高密度記録、特にMRヘッドで再生する場合にノイズを低減させ、高S/Nが得られる。BET法による比表面積は通常、30〜200m2/gであり、50〜100m2/gが好ましい。比表面積は概ね粉体板径と板厚からの算術計算値と符合する。板径・板厚の分布は狭いほど好ましい。分布は正規分布ではない場合が多いが、計算して粉体サイズに対する標準偏差で表すとσ/(平均板径または平均板厚)=0.1〜0.5である。粉体サイズ分布をシャープにするには粉体生成反応系をできるだけ均一にすると共に、生成した粉体に分布改良処理を施すことも行われている。たとえば酸溶液中で超微細粉体を選別的に溶解する方法等も知られている。ガラス化結晶法では、熱処理を複数回行い、核生成と成長を分離することでより均一な粉体を得ている。磁性粉で測定された抗磁力Hcは40〜400kA/m程度まで作成できるが、144〜300kA/mが好ましい。高Hcの方が高密度記録に有利であるが、記録ヘッドの能力で制限される。Hcは粉体サイズ(板径・板厚)、含有元素の種類と量、元素の置換サイト、粉体生成反応条件等により制御できる。 By setting the average plate diameter of the hexagonal ferrite magnetic powder to 50 nm or less and the average thickness to 15 nm or less, noise can be reduced and high S / N can be obtained when reproducing with high-density recording, particularly with an MR head. The specific surface area according to the BET method is usually 30 to 200 m 2 / g, and preferably 50 to 100 m 2 / g. The specific surface area generally agrees with the arithmetic calculation value from the powder plate diameter and plate thickness. The narrower the distribution of plate diameter and plate thickness, the better. In many cases, the distribution is not a normal distribution. When expressed as a standard deviation with respect to the powder size, σ / (average plate diameter or average plate thickness) = 0.1 to 0.5. In order to sharpen the powder size distribution, the powder production reaction system is made as uniform as possible, and the produced powder is subjected to a distribution improving process. For example, a method of selectively dissolving ultrafine powder in an acid solution is also known. In the vitrification crystal method, a more uniform powder is obtained by performing heat treatment a plurality of times to separate nucleation and growth. The coercive force Hc measured with the magnetic powder can be made up to about 40 to 400 kA / m, but preferably 144 to 300 kA / m. Higher Hc is more advantageous for high density recording, but is limited by the capacity of the recording head. Hc can be controlled by the powder size (plate diameter / plate thickness), the type and amount of the contained element, the substitution site of the element, the powder production reaction conditions, and the like.
六方晶系フェライト粉末の飽和磁化σSは30〜70A・m2/kgが好ましい。σSは、微粉体になるほど小さくなる傾向がある。 The saturation magnetization σS of the hexagonal ferrite powder is preferably 30 to 70 A · m 2 / kg. σS tends to decrease as the powder becomes finer.
本発明において、六方晶系フェライトのバリウムフェライト粉末を含む分散液を支持体上に塗布して磁性層を形成した塗布型バリウムフェライト(BaFe)磁性層、特に、板径が40nm以下のバリウムフェライト磁性体を用いた塗布型バリウムフェライト磁性層を有する磁気記録媒体は、高密度記録(特に、100kfciを超える線記録密度)での再生出力に優れ、また低ノイズである特性を有するため、有効である。 In the present invention, a coated barium ferrite (BaFe) magnetic layer in which a magnetic layer is formed by coating a dispersion containing a barium ferrite powder of hexagonal ferrite on a support, particularly a barium ferrite magnetic layer having a plate diameter of 40 nm or less. A magnetic recording medium having a coated barium ferrite magnetic layer using a body is effective because it has excellent reproduction output in high-density recording (particularly linear recording density exceeding 100 kfci) and has low noise characteristics. .
一般に、磁性体として、酸化鉄またはコバルト含有強磁性酸化鉄を用いた磁気記録媒体では、再生される孤立反転再生波形は、γ≒0の非対称性を示し、磁性体として六方晶系フェライトを用いた磁気記録媒体では、再生される孤立反転再生波形は、γ>0の非対称性を示す。また、蒸着法を用いて形成された磁性層を有する磁気記録媒体では、再生される孤立反転再生波形はγ<0の比対称性を示す。したがって、この非対称性に応じて、パーシャルレスポンス信号処理を前記(1)および(2)、または(3)および(4)で表わされる条件を満たすように等化処理を行なうことにより、磁気記録媒体の磁性層を形成する磁性体に応じて最適なパーシャルレスポンス信号処理を行なうことができる。 In general, in a magnetic recording medium using iron oxide or cobalt-containing ferromagnetic iron oxide as a magnetic material, the isolated inversion reproduction waveform to be reproduced shows asymmetry of γ≈0, and hexagonal ferrite is used as the magnetic material. In the conventional magnetic recording medium, the isolated inverted reproduction waveform to be reproduced exhibits asymmetry of γ> 0. Further, in a magnetic recording medium having a magnetic layer formed by vapor deposition, the isolated inverted reproduction waveform to be reproduced exhibits a relative symmetry of γ <0. Therefore, according to this asymmetry, the partial response signal processing is equalized so as to satisfy the conditions represented by the above (1) and (2) or (3) and (4). Optimal partial response signal processing can be performed according to the magnetic material forming the magnetic layer.
すなわち、本発明の記録再生方法および記録再生装置においては、この磁気記録媒体から情報信号を再生するに際して、まず、記録再生ヘッドによって磁気記録媒体から読み出されるアナログ信号において、磁気記録媒体に用いられている磁性体に応じて、孤立反転再生波形の非対称性を示すγがγ>0であるか、γ≒0であるか、あるいはγ<0であるかを予め推測することができる。したがって、本発明の記録再生方法および記録再生装置においては、用いる磁気記録媒体の磁性体に応じて、再生される孤立反転再生波形の非対称性を示すγがγ>0である波形を有する場合には、パーシャルレスポンスの係数項(1,a1,a2,・・・,am(m=2nまたは2n+1:nは1以上の整数)が前記の式(1)または式(2)を満たす条件で、パーシャルレスポンス信号処理方式によって、磁気記録媒体から読み取られた情報信号が等化処理され、等化処理された信号を復調して、元の信号が復元される。また、孤立反転再生波形の非対称性を示すγがγ<0である波形を有する場合には、前記パーシャルレスポンスの係数項(1,a1,a2,・・・,am(m=2nまたは2n+1:nは1以上の整数)が前記の式(3)または式(4)を満たす条件で、パーシャルレスポンス信号処理方式によって、磁気記録媒体から読み取られた情報信号が等化処理され、等化処理された信号を復調して、元の信号が復元される。このように、孤立反転再生波形の非対称性に応じて、最適なパーシャルレスポンス信号処理、すなわち、PR4ML(PR(1,0,−1)ML)、EPR4ML(PR(1,1,−1,−1)ML)、EEPR4ML(PR(1,2,0,−2,−1)ML)、EEEPR4ML(PR(1,3,2,−2,−3,−1)ML)等のいずれのパーシャルレスポンス信号処理方式による等化処理を行なうかを選択することによって、再生信号の復元を低いエラーレートで行なうことができる。 That is, in the recording / reproducing method and recording / reproducing apparatus of the present invention, when reproducing an information signal from the magnetic recording medium, first, an analog signal read from the magnetic recording medium by the recording / reproducing head is used for the magnetic recording medium. Depending on the magnetic material, it can be preliminarily estimated whether γ indicating the asymmetry of the isolated inverted reproduction waveform is γ> 0, γ≈0, or γ <0. Therefore, in the recording / reproducing method and recording / reproducing apparatus of the present invention, when γ indicating asymmetry of the isolated inverted reproduction waveform to be reproduced has a waveform in which γ> 0, depending on the magnetic material of the magnetic recording medium to be used. Is a partial response coefficient term (1, a 1 , a 2 ,..., A m (m = 2n or 2n + 1: n is an integer of 1 or more) satisfies the above formula (1) or (2). Under certain conditions, the information signal read from the magnetic recording medium is equalized by the partial response signal processing method, and the equalized signal is demodulated to restore the original signal. , Which has an asymmetry of γ <0, the partial response coefficient terms (1, a 1 , a 2 ,..., A m (m = 2n or 2n + 1: n is 1). The above integer) is the above formula (3 Alternatively, the information signal read from the magnetic recording medium is equalized by the partial response signal processing method under the condition satisfying Expression (4), and the equalized signal is demodulated to restore the original signal. Thus, the optimum partial response signal processing, that is, PR4ML (PR (1,0, -1) ML), EPR4ML (PR (1,1, -1, -1) ML), EEPR4ML (PR (1, 2, 0, -2, -1) ML), EEEPR4ML (PR (1, 3, 2, -2, -3, -1) ML), etc. By selecting whether to perform equalization processing by the partial response signal processing method, the reproduction signal can be restored at a low error rate.
次に、磁性体としてバリウムフェライトを含む磁性層を有する磁気記録媒体を例に取って、本発明の記録再生方法および記録再生装置について、図1〜3を参照して説明する。
図1は、本発明に係る記録再生装置の構成例を示し、この記録再生装置10は、プリコーダ11と、記録再生アンプ12と、記録用ヘッド13と、再生用ヘッド14と、イコライザ(等化手段)15と、最尤復号器16と、デコーダ(復調手段)17とを備える。
Next, taking a magnetic recording medium having a magnetic layer containing barium ferrite as a magnetic material as an example, the recording / reproducing method and recording / reproducing apparatus of the present invention will be described with reference to FIGS.
FIG. 1 shows a configuration example of a recording / reproducing apparatus according to the present invention. This recording / reproducing
プリコーダ11は、データ記録の前に置くことにより、復調時に生じる誤りの伝搬を防げるようになる。
By placing the
記録再生アンプ12は、プリコーダ11によって符号化された信号を増幅し、また、後記の再生用ヘッド14によって生じた信号を増幅するものである。
記録用ヘッド13は、磁気記録媒体20の磁性層に含まれるバリウムフェライト磁性体を磁化させて、所定のクロック周期のデータを磁気記録媒体20に記録するものである。
The recording / reproducing amplifier 12 amplifies the signal encoded by the
The
再生用ヘッド14は、磁気記録媒体20の磁性層に摺接して、その磁性層の磁化の変化を読み取り、アナログの再生信号を得る。このアナログ信号は、磁気記録媒体20の磁性層に記録された信号を微分した信号であり、(1−D)の伝達特性で表される。
The reproducing
再生用ヘッド14による読み取りによって生じたアナログ信号の波形の一例を図2に示す。ここでは、磁気記録媒体20に記録されたパルス信号の立ち上がりのタイミングで生じる孤立反転再生波形を例にして説明する。
An example of a waveform of an analog signal generated by reading by the reproducing
図2に示す孤立反転再生波形は、正方向にピークを有し、そのピークの左右が非対称となっている。そして、この孤立反転再生波形は、ピーク値の半値幅PW50のうちの右側の幅PW1が左側の幅PW2よりも大きくなっている。これは、面内配向の磁化成分と、バリウムフェライトに特有の垂直磁化成分を足し合わせたものである。 The isolated inverted reproduction waveform shown in FIG. 2 has a peak in the positive direction, and the left and right sides of the peak are asymmetric. In this isolated inverted reproduction waveform, the right-side width PW1 of the half-value width PW50 of the peak value is larger than the left-side width PW2. This is the sum of the in-plane orientation magnetization component and the perpendicular magnetization component peculiar to barium ferrite.
なお、図2では、正方向にピークを持つ孤立反転再生波形をアナログ信号の波形として示したが、実際には、アナログ信号の波形は、正方向および負方向にピークを持つ2つの孤立反転再生波形が重なり合って構成される。これは、負方向にピークを持つ孤立反転再生波形も、磁気記録媒体20に記録されたパルス信号の立ち下がりのタイミングで生じるからである。
In FIG. 2, the isolated inverted reproduction waveform having a peak in the positive direction is shown as an analog signal waveform. However, in reality, the waveform of the analog signal has two isolated inverted reproductions having peaks in the positive direction and the negative direction. It is composed of overlapping waveforms. This is because an isolated inverted reproduction waveform having a peak in the negative direction is also generated at the falling timing of the pulse signal recorded on the
イコライザ15(等化手段)は、再生用ヘッド14から記録再生アンプ12を介して転送された信号をイコライズする。PR(1,a,b,c,d,e,・・・)の伝達特性が1+a・D+b・D2+c・D3+d・D4+e・D5+・・・=(1−D)(1+f1・D+f2・D2+f3・D3+f4・D4+・・・)のときに、具体的には、イコライザ15は、伝達特性が1+f1・D+f2・D2+f3・D3+f4・D4+・・・で表わされるようにイコライズを行う。
The equalizer 15 (equalizing means) equalizes the signal transferred from the reproducing
最尤復号器16は、イコライザ15によってイコライズされたデータの識別を行なう。最尤復号は、データ間に相関を持たせて記録再生したときに、最も確からしいデータ系列を検出する方法で周知の技術である。そして、デコーダ17は、等化処理された信号を元のデータ(例えば(0,1,0))に復号する。これにより、磁気記録媒体20に記録された記録データを元のデータに正しく復元することが可能となる。
The
以下、本発明の記録再生方法を具体的に行なった例を示す。 Hereinafter, an example in which the recording / reproducing method of the present invention is specifically performed will be described.
<BaFe磁性層用塗布液の処方>
バリウムフェライト磁性粉 100部
ポリウレタン樹脂 14部
質量平均分子量:10000
スルホン酸官能基:0.05meq/g
研磨剤 8部
カーボンブラック(粒子サイズ:0.015μm) 0.5部
#55(旭カーボン社製)
ステアリン酸 0.5部
ブチルステアレート 2部
メチルエチルケトン 180部
ッシクロヘキサノン 100部
<Prescription of coating solution for BaFe magnetic layer>
Barium ferrite magnetic powder 100
Sulfonic acid functional group: 0.05 meq / g
Abrasive 8 parts carbon black (particle size: 0.015 μm) 0.5 parts # 55 (Asahi Carbon Co., Ltd.)
Stearic acid 0.5 part Butyl stearate 2 parts Methyl ethyl ketone 180 parts Cyclohexanone 100 parts
<非磁性層用塗布液の処方>
非磁性粉体:α酸化鉄 100部
平均一次粒子径:0.09μm、BET法による比表面積:50m2/g
pH:7 DBP吸油量:27〜38ml/100g、
表面処理層:Al2O3が粒子全体に対して8質量%存在
カーボンブラック 25部
コンダクテックスSC−U(コロンビアンカーボン社製)
塩化ビニル共重合体:MR104(日本ゼオン社製) 13部
ポリウレタン樹脂:UR8200(東洋紡社製9 5部
フェニルホスホン酸 3.5部
ブチルステアレート 1部
ステアリン酸 2部
メチルエチルケトン 205部
シクロヘキサノン 135部
<Prescription of coating solution for nonmagnetic layer>
Nonmagnetic powder: α iron oxide 100 parts Average primary particle size: 0.09 μm, specific surface area by BET method: 50 m 2 / g
pH: 7 DBP oil absorption: 27-38 ml / 100 g,
Surface treatment layer: Al 2 O 3 is present in an amount of 8% by mass based on the whole particle 25 parts of carbon black Conductex SC-U (manufactured by Columbian Carbon)
Vinyl chloride copolymer: MR104 (manufactured by Nippon Zeon Co., Ltd.) 13 parts Polyurethane resin: UR8200 (manufactured by Toyobo Co., Ltd. 9 parts 5 parts phenylphosphonic acid 3.5
<テープの製造>
上記の塗布液の処方にしたがって、各成分をニーダで混練した。得られた混練液を、1.0mmφのジルコニアビーズを分散部の容積に対し80%充填する量を入れた横型サンドミルにポンプで通液し、2000rpmで120分間(実質的に分散部に滞留した時間)分散処理を行なって磁性層用の分散液と非磁性層の分散液をそれぞれ調製した。得られた磁性層用の分散液には、さらにメチルエチルケトン3部を加え、1μmの平均粒径を有するフィルターを用いて濾過して磁性層形成用の塗布液を得た。また、非磁性層用の分散液には、ポリイソシアネートを2.5部、さらにメチルエチルケトン3部を加え、1μmの平均粒径を有するフィルターを用いて濾過して、非磁性層形成用の塗布液をそれぞれ調製した。
<Manufacture of tape>
Each component was kneaded with a kneader according to the formulation of the coating solution. The obtained kneaded liquid was pumped through a horizontal sand mill filled with 1.0 mmφ zirconia beads in an amount of 80% with respect to the volume of the dispersion part, and remained at 2000 rpm for 120 minutes (substantially stayed in the dispersion part. Time) Dispersion treatment was performed to prepare a dispersion for the magnetic layer and a dispersion for the nonmagnetic layer, respectively. To the obtained dispersion for magnetic layer, 3 parts of methyl ethyl ketone was further added and filtered using a filter having an average particle diameter of 1 μm to obtain a coating solution for forming a magnetic layer. Further, 2.5 parts of polyisocyanate and 3 parts of methyl ethyl ketone are added to the dispersion for the nonmagnetic layer, and the mixture is filtered using a filter having an average particle diameter of 1 μm to form a coating liquid for forming the nonmagnetic layer. Were prepared respectively.
得られた非磁性層形成用塗布液を、厚さ4μmのポリエチレンナフタレートベース上に乾燥後の厚さが1.5μmになるように塗布して乾燥させて非磁性層を形成した。その後、非磁性層の上に磁性層形成用の塗布液を磁性層の厚さが30〜210nmになるように逐次重層塗布を行ない、磁性層がまだ湿潤状態にあるうちに600mTの磁力を持つコバルト磁石と600mTの磁力を持つソレノイドにより磁性層内のBaFe磁性体を面内配向させ、さらに600mTのコバルト磁石で、垂直方向に磁界を印加して、斜めに磁性体を配向させ、磁性層の乾燥が終了するまでその垂直方向の磁界を保持した。次いで、7段のカレンダで温度90℃、線圧300kg/cm(294kN/m)にて処理を行なった。その後、非磁性層および磁性層を形成した面と反対側の面に、下記処方のバック層形成層塗布液を塗布して、厚み0.5μmのバック層を形成してウエブ原反を得た。 The obtained coating solution for forming a nonmagnetic layer was applied onto a polyethylene naphthalate base having a thickness of 4 μm so that the thickness after drying was 1.5 μm and dried to form a nonmagnetic layer. After that, the magnetic layer forming coating solution is successively applied onto the nonmagnetic layer so that the thickness of the magnetic layer is 30 to 210 nm, and the magnetic layer has a magnetic force of 600 mT while it is still wet. A BaFe magnetic body in the magnetic layer is oriented in the plane by a cobalt magnet and a solenoid having a magnetic force of 600 mT, and a magnetic field is applied obliquely by applying a magnetic field in the vertical direction with a 600 mT cobalt magnet. The vertical magnetic field was maintained until drying was completed. Next, the treatment was carried out with a seven-stage calendar at a temperature of 90 ° C. and a linear pressure of 300 kg / cm (294 kN / m). Thereafter, a back layer forming layer coating solution having the following formulation was applied to the surface opposite to the surface on which the nonmagnetic layer and the magnetic layer were formed to form a back layer having a thickness of 0.5 μm to obtain a web original fabric. .
バック層形成用塗布液
カーボンブラック(平均粒子サイズ:17nm) 100部
炭酸カルシウム(平均粒子サイズ:40nm) 80部
αアルミナ(平均粒子サイズ:200nm) 5部
分散液(ニトロセルロース樹脂、ポリウレタン樹脂、ポリイソシアネート)
Back layer forming coating solution Carbon black (average particle size: 17 nm) 100 parts Calcium carbonate (average particle size: 40 nm) 80 parts α-alumina (average particle size: 200 nm) 5 parts Dispersion liquid (nitrocellulose resin, polyurethane resin, poly Isocyanate)
前記のとおり、一面に非磁性層および磁性層を有し、他面にバック層を有するウエブ原反を、3.8mmの幅にスリットし、スリット品の送り出し部および巻き取り部を備える装置に不織布とカミソリブレードが磁性面に押し当たるように取り付け、テープクリーニング装置で磁性層の表面のクリーニングを行ない、BaFe磁性体を含む磁性層を有する磁気テープを得た。 As described above, an apparatus having a non-magnetic layer and a magnetic layer on one side and a web web having a back layer on the other side is slit to a width of 3.8 mm, and includes a delivery unit and a winding unit for slit products. The nonwoven fabric and the razor blade were attached so as to press against the magnetic surface, and the surface of the magnetic layer was cleaned with a tape cleaning device to obtain a magnetic tape having a magnetic layer containing a BaFe magnetic material.
BaFe磁性体を含む磁性層を有する磁気テープに、前記図1に示す構成を有する記録再生装置を用いて、規格化線密度K=(PW50)/(bit長)が異なるデータ波形を記録して、3種類の再生用サンプルを用意した。これらの3種の磁気テープのそれぞれから再生される孤立反転再生波形(γ=22%)について、PR(1,a,b,c)ML方式、PR(1,a,b,c,d)ML方式、PR(1,a,b,c,d,e)ML方式によって、特性項の前半の項の和の絶対値が後半の項の和の絶対値よりも小さい係数でのエラーレートを測定した。また、通常のPRML方式(=PR(1,1,−1,−1)ML方式、PR(1,2,0,−2,−1)ML方式、PR(1,3,2,−2,−3,−1)ML方式)でのエラーレートも測定した。 Data waveforms having different normalized linear densities K = (PW50) / (bit length) are recorded on a magnetic tape having a magnetic layer containing a BaFe magnetic material using the recording / reproducing apparatus having the configuration shown in FIG. Three types of playback samples were prepared. For the isolated inverted reproduction waveform (γ = 22%) reproduced from each of these three types of magnetic tapes, PR (1, a, b, c) ML system, PR (1, a, b, c, d) With the ML method and the PR (1, a, b, c, d, e) ML method, an error rate with a coefficient in which the absolute value of the sum of the first half of the characteristic term is smaller than the absolute value of the sum of the second half of the term is obtained. It was measured. Further, the normal PRML system (= PR (1,1, -1, -1) ML system, PR (1,2,0, -2, -1) ML system, PR (1,3,2, -2) , -3, -1) ML method) was also measured.
また、磁性体として、メタル(Feを主成分とする強磁性合金粉末、γ=3%)を用いた磁気テープ、ME(蒸着法による磁性層、γ=−13%)磁気テープについても、同様にして、エラーレートを測定した。
得られた結果を表1に示す。
The same applies to magnetic tapes using metal (ferromagnetic alloy powder containing Fe as a main component, γ = 3%) and ME (magnetic layer by vapor deposition, γ = -13%) magnetic tape as magnetic materials. The error rate was measured.
The obtained results are shown in Table 1.
また、γ(=−13%)<0であるME(蒸着法による磁性層)磁気テープでは、前記式(3)または(4)の場合にエラーレートが低減することが分かった。
Further, it was found that the error rate is reduced in the case of the formula (3) or (4) in the ME (magnetic layer by vapor deposition method) magnetic tape in which γ (= −13%) <0.
10 記録再生装置
11 プリコーダ
12 記録再生アンプ
13 記録用ヘッド
14 再生用ヘッド
15 イコライザ
16 最尤復号器
17 デコーダ
20 磁気記録媒体
DESCRIPTION OF
Claims (9)
前記磁気記録媒体から再生される孤立反転再生波形の非対称性を示すγがγ>0である波形を有する情報信号を、パーシャルレスポンスの係数項(1,a1,a2,・・・,am(m=2nまたは2n+1:nは1以上の整数))が下記の式(1)または式(2)を満たす条件で等化復調することを特徴とする記録再生方法。
An information signal having a waveform in which γ representing the asymmetry of the isolated inverted reproduction waveform reproduced from the magnetic recording medium is γ> 0 is expressed as a partial response coefficient term (1, a 1 , a 2 ,..., A A recording / reproducing method, wherein m (m = 2n or 2n + 1: n is an integer of 1 or more)) is equalized and demodulated under a condition satisfying the following expression (1) or expression (2).
前記磁気記録媒体から再生される孤立反転再生波形の非対称性を示すγがγ<0である波形を有する情報信号を、パーシャルレスポンスの係数項(1,a1,a2,・・・,am(m=2nまたは2n+1:nは1以上の整数))が下記の式(3)または式(4)を満たす条件で等化復調することを特徴とする記録再生方法。
An information signal having a waveform in which γ representing the asymmetry of the isolated inverted reproduction waveform reproduced from the magnetic recording medium is γ <0 is represented by a coefficient term (1, a 1 , a 2 ,..., A A recording / reproducing method, wherein m (m = 2n or 2n + 1: n is an integer of 1 or more)) is equalized and demodulated under a condition satisfying the following expression (3) or expression (4).
前記磁気記録媒体から再生される孤立反転再生波形の非対称性を示すγがγ>0である波形を有する場合、前記復調手段は、前記等化手段によって等化処理された前記情報信号についてのパーシャルレスポンスの係数項(1,a1,a2,・・・,am(m=2nまたは2n+1:nは1以上の整数))が下記の式(1)または式(2)を満たす条件で復調することを特徴とする記録再生装置。
When γ indicating the asymmetry of the isolated inverted reproduction waveform reproduced from the magnetic recording medium has a waveform with γ> 0, the demodulating means performs partial processing on the information signal equalized by the equalizing means. Response coefficient terms (1, a 1 , a 2 ,..., A m (m = 2n or 2n + 1: n is an integer greater than or equal to 1)) satisfy the following formula (1) or formula (2) A recording / reproducing apparatus for demodulating.
前記磁気記録媒体から再生される孤立反転再生波形の非対称性を示すγがγ<0である波形を有する場合、前記復調手段は、前記等化手段によって等化処理された前記情報信号についてのパーシャルレスポンスの係数項(1,a1,a2,・・・,am(m=2nまたは2n+1:nは1以上の整数))が下記の式(3)または式(4)を満たす条件で復調することを特徴とする記録再生装置。
When γ indicating the asymmetry of the isolated inverted reproduction waveform reproduced from the magnetic recording medium has a waveform where γ <0, the demodulating means performs partial processing on the information signal equalized by the equalizing means. Response coefficient terms (1, a 1 , a 2 ,..., A m (m = 2n or 2n + 1: n is an integer greater than or equal to 1)) satisfy the following formula (3) or formula (4) A recording / reproducing apparatus for demodulating.
Priority Applications (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| JP2004109035A JP2005293750A (en) | 2004-04-01 | 2004-04-01 | Method and device for recording and reproduction, and magnetic recording medium |
| US11/146,025 US7295394B2 (en) | 2004-04-01 | 2005-06-07 | Reproducing method, reproducing apparatus, recording and reproducing apparatus, and magnetic recording medium |
Applications Claiming Priority (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| JP2004109035A JP2005293750A (en) | 2004-04-01 | 2004-04-01 | Method and device for recording and reproduction, and magnetic recording medium |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| JP2005293750A true JP2005293750A (en) | 2005-10-20 |
Family
ID=35060267
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| JP2004109035A Pending JP2005293750A (en) | 2004-04-01 | 2004-04-01 | Method and device for recording and reproduction, and magnetic recording medium |
Country Status (2)
| Country | Link |
|---|---|
| US (1) | US7295394B2 (en) |
| JP (1) | JP2005293750A (en) |
Families Citing this family (3)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US7236319B2 (en) * | 2005-06-08 | 2007-06-26 | Fujifilm Corporation | Reproducing method, reproducing apparatus, recording and reproducing apparatus, and magnetic recording medium |
| US7532425B2 (en) * | 2005-06-21 | 2009-05-12 | Fujifilm Corporation | Reproducing method, reproducing apparatus, recording and reproducing apparatus, and magnetic recording medium |
| US10755735B2 (en) * | 2018-06-29 | 2020-08-25 | Fujifilm Corporation | Apparatus capable of determining deviation amount between magnetic head and magnetic tape based on reading servo pattern |
Family Cites Families (3)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JP2840467B2 (en) * | 1991-02-07 | 1998-12-24 | キヤノン株式会社 | Magnetic recording and reproducing method |
| US6249398B1 (en) * | 1998-03-04 | 2001-06-19 | Maxtor Corporation | Class of fixed partial response targets in a PRML sampled data detection channel |
| JP3646684B2 (en) | 2001-09-20 | 2005-05-11 | 株式会社日立製作所 | Data recording / reproducing apparatus using partial response demodulation method |
-
2004
- 2004-04-01 JP JP2004109035A patent/JP2005293750A/en active Pending
-
2005
- 2005-06-07 US US11/146,025 patent/US7295394B2/en not_active Expired - Fee Related
Also Published As
| Publication number | Publication date |
|---|---|
| US20050225888A1 (en) | 2005-10-13 |
| US7295394B2 (en) | 2007-11-13 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| CN1129894C (en) | Multi-layer magnetic recording media and magnetoresistive driven systems | |
| US9728216B2 (en) | Feromagnetically coupled magnetic recording media | |
| JP2001023144A (en) | Magnetic recording medium and method of manufacturing the same | |
| US20090110961A1 (en) | Magnetic recording medium, method of manufacturing the same, and magnetic storage device | |
| JP2009110606A (en) | Magnetic recording medium, method for manufacturing the same, and magnetic storage device | |
| JP2008198316A (en) | Perpendicular magnetic recording medium, manufacturing method thereof, and magnetic recording apparatus | |
| JP2005251264A (en) | Perpendicular magnetic recording medium and manufacturing method thereof | |
| JP2005293750A (en) | Method and device for recording and reproduction, and magnetic recording medium | |
| US7532425B2 (en) | Reproducing method, reproducing apparatus, recording and reproducing apparatus, and magnetic recording medium | |
| JP2010009683A (en) | Magnetic recording medium and magnetic recording device | |
| JP7009417B2 (en) | Magnetic tape device | |
| JP4383336B2 (en) | Magnetic tape | |
| JP2008276863A (en) | Perpendicular magnetic recording medium, manufacturing method thereof, and magnetic recording apparatus | |
| US7236319B2 (en) | Reproducing method, reproducing apparatus, recording and reproducing apparatus, and magnetic recording medium | |
| WO2004075178A1 (en) | Vertical magnetic recording medium | |
| JP2005293753A (en) | Recording/reproducing method, recording/reproducing apparatus, and magnetic recording medium | |
| JP2005293754A (en) | Recording and reproducing method, recording and reproducing apparatus, and magnetic recording medium | |
| KR100935147B1 (en) | Vertical magnetic recording medium, manufacturing method thereof and magnetic recording device | |
| JP2005293752A (en) | Recording and reproducing method, recording and reproducing apparatus, and magnetic recording medium | |
| JP2004272982A (en) | Method for manufacturing perpendicular magnetic recording medium and perpendicular magnetic recording medium | |
| JP2005025820A (en) | Magnetic recording medium | |
| Bate | A survey of recent advances in magnetic recording materials | |
| JP2004178708A (en) | Magnetic recording medium and magnetic recorder | |
| JP3030279B2 (en) | Magnetic recording medium and magnetic recording / reproducing device | |
| JP2001043523A (en) | Magnetic recording medium and magnetic recording device |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| A711 | Notification of change in applicant |
Free format text: JAPANESE INTERMEDIATE CODE: A712 Effective date: 20061212 |
|
| RD02 | Notification of acceptance of power of attorney |
Free format text: JAPANESE INTERMEDIATE CODE: A7422 Effective date: 20061212 |
|
| A621 | Written request for application examination |
Free format text: JAPANESE INTERMEDIATE CODE: A621 Effective date: 20070209 |
|
| A977 | Report on retrieval |
Free format text: JAPANESE INTERMEDIATE CODE: A971007 Effective date: 20090223 |
|
| A131 | Notification of reasons for refusal |
Free format text: JAPANESE INTERMEDIATE CODE: A131 Effective date: 20090303 |
|
| A521 | Written amendment |
Free format text: JAPANESE INTERMEDIATE CODE: A523 Effective date: 20090501 |
|
| A131 | Notification of reasons for refusal |
Free format text: JAPANESE INTERMEDIATE CODE: A131 Effective date: 20090609 |
|
| RD13 | Notification of appointment of power of sub attorney |
Free format text: JAPANESE INTERMEDIATE CODE: A7433 Effective date: 20090722 |
|
| A521 | Written amendment |
Free format text: JAPANESE INTERMEDIATE CODE: A523 Effective date: 20090731 |
|
| A521 | Written amendment |
Free format text: JAPANESE INTERMEDIATE CODE: A821 Effective date: 20090724 |
|
| A131 | Notification of reasons for refusal |
Free format text: JAPANESE INTERMEDIATE CODE: A131 Effective date: 20090908 |
|
| A131 | Notification of reasons for refusal |
Free format text: JAPANESE INTERMEDIATE CODE: A131 Effective date: 20091208 |
|
| A02 | Decision of refusal |
Free format text: JAPANESE INTERMEDIATE CODE: A02 Effective date: 20100309 |