EP3347081B1 - Apparatus for delivery of substances into bone - Google Patents
Apparatus for delivery of substances into bone Download PDFInfo
- Publication number
- EP3347081B1 EP3347081B1 EP15771668.9A EP15771668A EP3347081B1 EP 3347081 B1 EP3347081 B1 EP 3347081B1 EP 15771668 A EP15771668 A EP 15771668A EP 3347081 B1 EP3347081 B1 EP 3347081B1
- Authority
- EP
- European Patent Office
- Prior art keywords
- bone
- objects
- delivery arrangement
- object delivery
- arrangement comprises
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Active
Links
- 210000000988 bone and bone Anatomy 0.000 title claims description 92
- 238000012384 transportation and delivery Methods 0.000 title claims description 22
- 239000000126 substance Substances 0.000 title claims description 10
- 210000001519 tissue Anatomy 0.000 claims description 33
- 238000002604 ultrasonography Methods 0.000 claims description 32
- 238000000151 deposition Methods 0.000 claims description 30
- 230000008021 deposition Effects 0.000 claims description 26
- 238000009792 diffusion process Methods 0.000 claims description 15
- 230000014759 maintenance of location Effects 0.000 claims description 10
- 230000004807 localization Effects 0.000 claims description 6
- 230000017531 blood circulation Effects 0.000 claims description 5
- 230000010412 perfusion Effects 0.000 claims description 4
- 230000003044 adaptive effect Effects 0.000 claims description 3
- 210000001185 bone marrow Anatomy 0.000 claims description 3
- 230000009466 transformation Effects 0.000 claims description 3
- 230000003213 activating effect Effects 0.000 claims description 2
- 230000003321 amplification Effects 0.000 claims description 2
- 239000002086 nanomaterial Substances 0.000 claims description 2
- 238000003199 nucleic acid amplification method Methods 0.000 claims description 2
- 239000003814 drug Substances 0.000 description 11
- 206010017076 Fracture Diseases 0.000 description 10
- 239000004480 active ingredient Substances 0.000 description 10
- 229940079593 drug Drugs 0.000 description 10
- 238000000034 method Methods 0.000 description 9
- 230000003287 optical effect Effects 0.000 description 6
- 230000032258 transport Effects 0.000 description 6
- 208000010392 Bone Fractures Diseases 0.000 description 5
- 238000010521 absorption reaction Methods 0.000 description 5
- -1 anticholesterols Substances 0.000 description 5
- 239000003102 growth factor Substances 0.000 description 5
- 210000004872 soft tissue Anatomy 0.000 description 5
- 230000001225 therapeutic effect Effects 0.000 description 5
- 208000020084 Bone disease Diseases 0.000 description 4
- 238000013459 approach Methods 0.000 description 4
- 230000008468 bone growth Effects 0.000 description 4
- 239000003795 chemical substances by application Substances 0.000 description 4
- 238000005516 engineering process Methods 0.000 description 4
- 239000002872 contrast media Substances 0.000 description 3
- 238000001514 detection method Methods 0.000 description 3
- 230000000694 effects Effects 0.000 description 3
- 238000001727 in vivo Methods 0.000 description 3
- 238000007726 management method Methods 0.000 description 3
- CIWBSHSKHKDKBQ-JLAZNSOCSA-N Ascorbic acid Chemical compound OC[C@H](O)[C@H]1OC(=O)C(O)=C1O CIWBSHSKHKDKBQ-JLAZNSOCSA-N 0.000 description 2
- 208000001132 Osteoporosis Diseases 0.000 description 2
- 230000004913 activation Effects 0.000 description 2
- 230000032683 aging Effects 0.000 description 2
- 239000001961 anticonvulsive agent Substances 0.000 description 2
- 239000002246 antineoplastic agent Substances 0.000 description 2
- 210000001188 articular cartilage Anatomy 0.000 description 2
- 150000001875 compounds Chemical class 0.000 description 2
- 239000004020 conductor Substances 0.000 description 2
- 230000001054 cortical effect Effects 0.000 description 2
- 239000003172 expectorant agent Substances 0.000 description 2
- 230000035876 healing Effects 0.000 description 2
- 239000004615 ingredient Substances 0.000 description 2
- 229910052500 inorganic mineral Inorganic materials 0.000 description 2
- 239000011707 mineral Substances 0.000 description 2
- 239000003149 muscarinic antagonist Substances 0.000 description 2
- 230000035699 permeability Effects 0.000 description 2
- 230000000472 traumatic effect Effects 0.000 description 2
- 238000012285 ultrasound imaging Methods 0.000 description 2
- RBTBFTRPCNLSDE-UHFFFAOYSA-N 3,7-bis(dimethylamino)phenothiazin-5-ium Chemical compound C1=CC(N(C)C)=CC2=[S+]C3=CC(N(C)C)=CC=C3N=C21 RBTBFTRPCNLSDE-UHFFFAOYSA-N 0.000 description 1
- LRFVTYWOQMYALW-UHFFFAOYSA-N 9H-xanthine Chemical class O=C1NC(=O)NC2=C1NC=N2 LRFVTYWOQMYALW-UHFFFAOYSA-N 0.000 description 1
- 102000055006 Calcitonin Human genes 0.000 description 1
- 108060001064 Calcitonin Proteins 0.000 description 1
- OYPRJOBELJOOCE-UHFFFAOYSA-N Calcium Chemical compound [Ca] OYPRJOBELJOOCE-UHFFFAOYSA-N 0.000 description 1
- ZZZCUOFIHGPKAK-UHFFFAOYSA-N D-erythro-ascorbic acid Natural products OCC1OC(=O)C(O)=C1O ZZZCUOFIHGPKAK-UHFFFAOYSA-N 0.000 description 1
- MPBVHIBUJCELCL-UHFFFAOYSA-N Ibandronate Chemical compound CCCCCN(C)CCC(O)(P(O)(O)=O)P(O)(O)=O MPBVHIBUJCELCL-UHFFFAOYSA-N 0.000 description 1
- 206010027452 Metastases to bone Diseases 0.000 description 1
- 229930182555 Penicillin Natural products 0.000 description 1
- 208000006735 Periostitis Diseases 0.000 description 1
- 239000000150 Sympathomimetic Substances 0.000 description 1
- 108010049264 Teriparatide Proteins 0.000 description 1
- 102000004887 Transforming Growth Factor beta Human genes 0.000 description 1
- 108090001012 Transforming Growth Factor beta Proteins 0.000 description 1
- 229930003268 Vitamin C Natural products 0.000 description 1
- 229930003316 Vitamin D Natural products 0.000 description 1
- QYSXJUFSXHHAJI-XFEUOLMDSA-N Vitamin D3 Natural products C1(/[C@@H]2CC[C@@H]([C@]2(CCC1)C)[C@H](C)CCCC(C)C)=C/C=C1\C[C@@H](O)CCC1=C QYSXJUFSXHHAJI-XFEUOLMDSA-N 0.000 description 1
- 230000005856 abnormality Effects 0.000 description 1
- 239000002253 acid Substances 0.000 description 1
- 239000011149 active material Substances 0.000 description 1
- 239000003741 agents affecting lipid metabolism Substances 0.000 description 1
- 229940035676 analgesics Drugs 0.000 description 1
- 230000000578 anorexic effect Effects 0.000 description 1
- 239000005557 antagonist Substances 0.000 description 1
- 239000000730 antalgic agent Substances 0.000 description 1
- 230000000507 anthelmentic effect Effects 0.000 description 1
- 239000000921 anthelmintic agent Substances 0.000 description 1
- 229940124339 anthelmintic agent Drugs 0.000 description 1
- 239000003242 anti bacterial agent Substances 0.000 description 1
- 239000004004 anti-anginal agent Substances 0.000 description 1
- 230000003627 anti-cholesterol Effects 0.000 description 1
- 230000001078 anti-cholinergic effect Effects 0.000 description 1
- 230000003556 anti-epileptic effect Effects 0.000 description 1
- 229940121363 anti-inflammatory agent Drugs 0.000 description 1
- 239000002260 anti-inflammatory agent Substances 0.000 description 1
- 230000001022 anti-muscarinic effect Effects 0.000 description 1
- 239000000043 antiallergic agent Substances 0.000 description 1
- 229940124345 antianginal agent Drugs 0.000 description 1
- 239000003416 antiarrhythmic agent Substances 0.000 description 1
- 229940088710 antibiotic agent Drugs 0.000 description 1
- 239000003146 anticoagulant agent Substances 0.000 description 1
- 229940127219 anticoagulant drug Drugs 0.000 description 1
- 229940125681 anticonvulsant agent Drugs 0.000 description 1
- 239000000935 antidepressant agent Substances 0.000 description 1
- 229940005513 antidepressants Drugs 0.000 description 1
- 239000003472 antidiabetic agent Substances 0.000 description 1
- 229940125708 antidiabetic agent Drugs 0.000 description 1
- 229960003965 antiepileptics Drugs 0.000 description 1
- 229940121375 antifungal agent Drugs 0.000 description 1
- 239000003429 antifungal agent Substances 0.000 description 1
- 229940030225 antihemorrhagics Drugs 0.000 description 1
- 229940125715 antihistaminic agent Drugs 0.000 description 1
- 239000000739 antihistaminic agent Substances 0.000 description 1
- 239000002220 antihypertensive agent Substances 0.000 description 1
- 229940030600 antihypertensive agent Drugs 0.000 description 1
- 239000003926 antimycobacterial agent Substances 0.000 description 1
- 229940034982 antineoplastic agent Drugs 0.000 description 1
- 239000000939 antiparkinson agent Substances 0.000 description 1
- 239000000164 antipsychotic agent Substances 0.000 description 1
- 239000003200 antithyroid agent Substances 0.000 description 1
- 229940043671 antithyroid preparations Drugs 0.000 description 1
- 239000003434 antitussive agent Substances 0.000 description 1
- 229940124584 antitussives Drugs 0.000 description 1
- 239000003443 antiviral agent Substances 0.000 description 1
- 239000002249 anxiolytic agent Substances 0.000 description 1
- 230000000949 anxiolytic effect Effects 0.000 description 1
- 210000001367 artery Anatomy 0.000 description 1
- 239000003212 astringent agent Substances 0.000 description 1
- 230000008901 benefit Effects 0.000 description 1
- 239000002876 beta blocker Substances 0.000 description 1
- 239000010836 blood and blood product Substances 0.000 description 1
- 229940125691 blood product Drugs 0.000 description 1
- 239000003633 blood substitute Substances 0.000 description 1
- 230000037180 bone health Effects 0.000 description 1
- 230000037118 bone strength Effects 0.000 description 1
- BBBFJLBPOGFECG-VJVYQDLKSA-N calcitonin Chemical compound N([C@H](C(=O)N[C@@H](CC(C)C)C(=O)NCC(=O)N[C@@H](CCCCN)C(=O)N[C@@H](CC(C)C)C(=O)N[C@@H](CO)C(=O)N[C@@H](CCC(N)=O)C(=O)N[C@@H](CCC(O)=O)C(=O)N[C@@H](CC(C)C)C(=O)N[C@@H](CC=1NC=NC=1)C(=O)N[C@@H](CCCCN)C(=O)N[C@@H](CC(C)C)C(=O)N[C@@H](CCC(N)=O)C(=O)N[C@@H]([C@@H](C)O)C(=O)N[C@@H](CC=1C=CC(O)=CC=1)C(=O)N1[C@@H](CCC1)C(=O)N[C@@H](CCCNC(N)=N)C(=O)N[C@@H]([C@@H](C)O)C(=O)N[C@@H](CC(N)=O)C(=O)N[C@@H]([C@@H](C)O)C(=O)NCC(=O)N[C@@H](CO)C(=O)NCC(=O)N[C@@H]([C@@H](C)O)C(=O)N1[C@@H](CCC1)C(N)=O)C(C)C)C(=O)[C@@H]1CSSC[C@H](N)C(=O)N[C@@H](CO)C(=O)N[C@@H](CC(N)=O)C(=O)N[C@@H](CC(C)C)C(=O)N[C@@H](CO)C(=O)N[C@@H]([C@@H](C)O)C(=O)N1 BBBFJLBPOGFECG-VJVYQDLKSA-N 0.000 description 1
- 229960004015 calcitonin Drugs 0.000 description 1
- 239000011575 calcium Substances 0.000 description 1
- 229910052791 calcium Inorganic materials 0.000 description 1
- 235000001465 calcium Nutrition 0.000 description 1
- 210000004027 cell Anatomy 0.000 description 1
- 230000000718 cholinopositive effect Effects 0.000 description 1
- 238000010276 construction Methods 0.000 description 1
- 229940039231 contrast media Drugs 0.000 description 1
- 239000013068 control sample Substances 0.000 description 1
- 206010061428 decreased appetite Diseases 0.000 description 1
- 230000003247 decreasing effect Effects 0.000 description 1
- 230000007812 deficiency Effects 0.000 description 1
- 229960001251 denosumab Drugs 0.000 description 1
- 238000011161 development Methods 0.000 description 1
- 230000018109 developmental process Effects 0.000 description 1
- 208000037265 diseases, disorders, signs and symptoms Diseases 0.000 description 1
- 208000035475 disorder Diseases 0.000 description 1
- 239000002934 diuretic Substances 0.000 description 1
- 229940030606 diuretics Drugs 0.000 description 1
- 230000003291 dopaminomimetic effect Effects 0.000 description 1
- 230000005672 electromagnetic field Effects 0.000 description 1
- 230000003419 expectorant effect Effects 0.000 description 1
- 229940066493 expectorants Drugs 0.000 description 1
- 238000002474 experimental method Methods 0.000 description 1
- 230000006870 function Effects 0.000 description 1
- 239000003163 gonadal steroid hormone Substances 0.000 description 1
- 230000012010 growth Effects 0.000 description 1
- 230000000025 haemostatic effect Effects 0.000 description 1
- 230000036541 health Effects 0.000 description 1
- 238000010438 heat treatment Methods 0.000 description 1
- 230000003054 hormonal effect Effects 0.000 description 1
- 239000003326 hypnotic agent Substances 0.000 description 1
- 230000000147 hypnotic effect Effects 0.000 description 1
- 229960005236 ibandronic acid Drugs 0.000 description 1
- 238000005286 illumination Methods 0.000 description 1
- 238000003384 imaging method Methods 0.000 description 1
- 230000001506 immunosuppresive effect Effects 0.000 description 1
- 229960003444 immunosuppressant agent Drugs 0.000 description 1
- 239000003018 immunosuppressive agent Substances 0.000 description 1
- 238000011065 in-situ storage Methods 0.000 description 1
- 230000003993 interaction Effects 0.000 description 1
- 150000002632 lipids Chemical class 0.000 description 1
- 239000000463 material Substances 0.000 description 1
- 230000007246 mechanism Effects 0.000 description 1
- 239000012528 membrane Substances 0.000 description 1
- 229960000907 methylthioninium chloride Drugs 0.000 description 1
- 230000000510 mucolytic effect Effects 0.000 description 1
- 229940066491 mucolytics Drugs 0.000 description 1
- 239000000472 muscarinic agonist Substances 0.000 description 1
- 229940035363 muscle relaxants Drugs 0.000 description 1
- 239000003158 myorelaxant agent Substances 0.000 description 1
- 239000002121 nanofiber Substances 0.000 description 1
- 239000002073 nanorod Substances 0.000 description 1
- 239000002698 neuron blocking agent Substances 0.000 description 1
- 235000006286 nutrient intake Nutrition 0.000 description 1
- 239000013307 optical fiber Substances 0.000 description 1
- 238000000879 optical micrograph Methods 0.000 description 1
- 229940094443 oxytocics prostaglandins Drugs 0.000 description 1
- 239000000734 parasympathomimetic agent Substances 0.000 description 1
- 230000001499 parasympathomimetic effect Effects 0.000 description 1
- 229940005542 parasympathomimetics Drugs 0.000 description 1
- 230000000849 parathyroid Effects 0.000 description 1
- 230000037361 pathway Effects 0.000 description 1
- 230000035515 penetration Effects 0.000 description 1
- 150000002960 penicillins Chemical class 0.000 description 1
- 210000003460 periosteum Anatomy 0.000 description 1
- 239000011148 porous material Substances 0.000 description 1
- 230000008569 process Effects 0.000 description 1
- 150000003180 prostaglandins Chemical class 0.000 description 1
- 229940121896 radiopharmaceutical Drugs 0.000 description 1
- 239000012217 radiopharmaceutical Substances 0.000 description 1
- 230000002799 radiopharmaceutical effect Effects 0.000 description 1
- 230000009467 reduction Effects 0.000 description 1
- 238000007634 remodeling Methods 0.000 description 1
- 238000011160 research Methods 0.000 description 1
- 230000004044 response Effects 0.000 description 1
- 229940125723 sedative agent Drugs 0.000 description 1
- 239000000932 sedative agent Substances 0.000 description 1
- 238000001338 self-assembly Methods 0.000 description 1
- 238000000527 sonication Methods 0.000 description 1
- 150000003431 steroids Chemical class 0.000 description 1
- 239000000021 stimulant Substances 0.000 description 1
- 230000035882 stress Effects 0.000 description 1
- 210000005065 subchondral bone plate Anatomy 0.000 description 1
- 230000001975 sympathomimetic effect Effects 0.000 description 1
- 229940064707 sympathomimetics Drugs 0.000 description 1
- 238000012385 systemic delivery Methods 0.000 description 1
- 229940065721 systemic for obstructive airway disease xanthines Drugs 0.000 description 1
- 230000002123 temporal effect Effects 0.000 description 1
- OGBMKVWORPGQRR-UMXFMPSGSA-N teriparatide Chemical compound C([C@H](NC(=O)[C@H](CCSC)NC(=O)[C@H](CC(C)C)NC(=O)[C@H](CCC(N)=O)NC(=O)[C@@H](NC(=O)[C@H](CCC(O)=O)NC(=O)[C@H](CO)NC(=O)[C@@H](NC(=O)[C@@H](N)CO)C(C)C)[C@@H](C)CC)C(=O)N[C@@H](CC(N)=O)C(=O)N[C@@H](CC(C)C)C(=O)NCC(=O)N[C@@H](CCCCN)C(=O)N[C@@H](CC=1N=CNC=1)C(=O)N[C@@H](CC(C)C)C(=O)N[C@@H](CC(N)=O)C(=O)N[C@@H](CO)C(=O)N[C@@H](CCSC)C(=O)N[C@@H](CCC(O)=O)C(=O)N[C@@H](CCCNC(N)=N)C(=O)N[C@@H](C(C)C)C(=O)N[C@@H](CCC(O)=O)C(=O)N[C@@H](CC=1C2=CC=CC=C2NC=1)C(=O)N[C@@H](CC(C)C)C(=O)N[C@@H](CCCNC(N)=N)C(=O)N[C@@H](CCCCN)C(=O)N[C@@H](CCCCN)C(=O)N[C@@H](CC(C)C)C(=O)N[C@@H](CCC(N)=O)C(=O)N[C@@H](CC(O)=O)C(=O)N[C@@H](C(C)C)C(=O)N[C@@H](CC=1N=CNC=1)C(=O)N[C@@H](CC(N)=O)C(=O)N[C@@H](CC=1C=CC=CC=1)C(O)=O)C1=CNC=N1 OGBMKVWORPGQRR-UMXFMPSGSA-N 0.000 description 1
- 229960005460 teriparatide Drugs 0.000 description 1
- ZRKFYGHZFMAOKI-QMGMOQQFSA-N tgfbeta Chemical compound C([C@H](NC(=O)[C@H](C(C)C)NC(=O)CNC(=O)[C@H](CCC(O)=O)NC(=O)[C@H](CCCNC(N)=N)NC(=O)[C@H](CC(N)=O)NC(=O)[C@H](CC(C)C)NC(=O)[C@H]([C@@H](C)O)NC(=O)[C@H](CCC(O)=O)NC(=O)[C@H]([C@@H](C)O)NC(=O)[C@H](CC(C)C)NC(=O)CNC(=O)[C@H](C)NC(=O)[C@H](CO)NC(=O)[C@H](CCC(N)=O)NC(=O)[C@@H](NC(=O)[C@H](C)NC(=O)[C@H](C)NC(=O)[C@@H](NC(=O)[C@H](CC(C)C)NC(=O)[C@@H](N)CCSC)C(C)C)[C@@H](C)CC)C(=O)N[C@@H]([C@@H](C)O)C(=O)N[C@@H](C(C)C)C(=O)N[C@@H](CC=1C=CC=CC=1)C(=O)N[C@@H](C)C(=O)N1[C@@H](CCC1)C(=O)N[C@@H]([C@@H](C)O)C(=O)N[C@@H](CC(N)=O)C(=O)N[C@@H](CCC(O)=O)C(=O)N[C@@H](C)C(=O)N[C@@H](CC=1C=CC=CC=1)C(=O)N[C@@H](CCCNC(N)=N)C(=O)N[C@@H](C)C(=O)N[C@@H](CC(C)C)C(=O)N1[C@@H](CCC1)C(=O)N1[C@@H](CCC1)C(=O)N[C@@H](CCCNC(N)=N)C(=O)N[C@@H](CCC(O)=O)C(=O)N[C@@H](CCCNC(N)=N)C(=O)N[C@@H](CO)C(=O)N[C@@H](CCCNC(N)=N)C(=O)N[C@@H](CC(C)C)C(=O)N[C@@H](CC(C)C)C(O)=O)C1=CC=C(O)C=C1 ZRKFYGHZFMAOKI-QMGMOQQFSA-N 0.000 description 1
- 238000002560 therapeutic procedure Methods 0.000 description 1
- 210000001685 thyroid gland Anatomy 0.000 description 1
- 229930003231 vitamin Natural products 0.000 description 1
- 235000013343 vitamin Nutrition 0.000 description 1
- 239000011782 vitamin Substances 0.000 description 1
- 229940088594 vitamin Drugs 0.000 description 1
- 235000019154 vitamin C Nutrition 0.000 description 1
- 239000011718 vitamin C Substances 0.000 description 1
- 235000019166 vitamin D Nutrition 0.000 description 1
- 239000011710 vitamin D Substances 0.000 description 1
- 150000003710 vitamin D derivatives Chemical class 0.000 description 1
- 229940046008 vitamin d Drugs 0.000 description 1
Images
Classifications
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61M—DEVICES FOR INTRODUCING MEDIA INTO, OR ONTO, THE BODY; DEVICES FOR TRANSDUCING BODY MEDIA OR FOR TAKING MEDIA FROM THE BODY; DEVICES FOR PRODUCING OR ENDING SLEEP OR STUPOR
- A61M37/00—Other apparatus for introducing media into the body; Percutany, i.e. introducing medicines into the body by diffusion through the skin
- A61M37/0092—Other apparatus for introducing media into the body; Percutany, i.e. introducing medicines into the body by diffusion through the skin using ultrasonic, sonic or infrasonic vibrations, e.g. phonophoresis
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61B—DIAGNOSIS; SURGERY; IDENTIFICATION
- A61B17/00—Surgical instruments, devices or methods, e.g. tourniquets
- A61B17/56—Surgical instruments or methods for treatment of bones or joints; Devices specially adapted therefor
- A61B17/58—Surgical instruments or methods for treatment of bones or joints; Devices specially adapted therefor for osteosynthesis, e.g. bone plates, screws, setting implements or the like
- A61B17/88—Osteosynthesis instruments; Methods or means for implanting or extracting internal or external fixation devices
- A61B17/8802—Equipment for handling bone cement or other fluid fillers
- A61B17/8805—Equipment for handling bone cement or other fluid fillers for introducing fluid filler into bone or extracting it
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61B—DIAGNOSIS; SURGERY; IDENTIFICATION
- A61B17/00—Surgical instruments, devices or methods, e.g. tourniquets
- A61B17/56—Surgical instruments or methods for treatment of bones or joints; Devices specially adapted therefor
- A61B17/58—Surgical instruments or methods for treatment of bones or joints; Devices specially adapted therefor for osteosynthesis, e.g. bone plates, screws, setting implements or the like
- A61B17/88—Osteosynthesis instruments; Methods or means for implanting or extracting internal or external fixation devices
- A61B17/885—Tools for expanding or compacting bones or discs or cavities therein
- A61B17/8852—Tools for expanding or compacting bones or discs or cavities therein capable of being assembled or enlarged, or changing shape, inside the bone or disc
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61B—DIAGNOSIS; SURGERY; IDENTIFICATION
- A61B17/00—Surgical instruments, devices or methods, e.g. tourniquets
- A61B17/56—Surgical instruments or methods for treatment of bones or joints; Devices specially adapted therefor
- A61B2017/564—Methods for bone or joint treatment
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61F—FILTERS IMPLANTABLE INTO BLOOD VESSELS; PROSTHESES; DEVICES PROVIDING PATENCY TO, OR PREVENTING COLLAPSING OF, TUBULAR STRUCTURES OF THE BODY, e.g. STENTS; ORTHOPAEDIC, NURSING OR CONTRACEPTIVE DEVICES; FOMENTATION; TREATMENT OR PROTECTION OF EYES OR EARS; BANDAGES, DRESSINGS OR ABSORBENT PADS; FIRST-AID KITS
- A61F2/00—Filters implantable into blood vessels; Prostheses, i.e. artificial substitutes or replacements for parts of the body; Appliances for connecting them with the body; Devices providing patency to, or preventing collapsing of, tubular structures of the body, e.g. stents
- A61F2/02—Prostheses implantable into the body
- A61F2/28—Bones
- A61F2002/286—Bone stimulation by mechanical vibrations for enhancing ossification
- A61F2002/2864—Bone stimulation by mechanical vibrations for enhancing ossification by ultrasonic or acoustic vibrations
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61M—DEVICES FOR INTRODUCING MEDIA INTO, OR ONTO, THE BODY; DEVICES FOR TRANSDUCING BODY MEDIA OR FOR TAKING MEDIA FROM THE BODY; DEVICES FOR PRODUCING OR ENDING SLEEP OR STUPOR
- A61M2210/00—Anatomical parts of the body
- A61M2210/02—Bones
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61N—ELECTROTHERAPY; MAGNETOTHERAPY; RADIATION THERAPY; ULTRASOUND THERAPY
- A61N7/00—Ultrasound therapy
- A61N2007/0004—Applications of ultrasound therapy
- A61N2007/0013—Fracture healing
Definitions
- the invention relates to bone healthcare and health management.
- the invention deals with detection of a weak bone and healing of the weak or fractured bone in vivo.
- Bone diseases are disorders in remodeling of bone tissue. As a result, bones can become mechanically weak. Reduction of bone mineral density (BMD) is a natural process related to aging after the age of 20. However, some bone diseases, such as osteoporosis, can cause excessive loss of BMD. Deficiencies in nutrient intake (e.g., calcium and vitamin D and C), hormonal imbalance and cell abnormalities can also cause bone disorders.
- BMD bone mineral density
- Bone fractures are labelled low-impact fractures and high-impact fractures.
- the low-impact (or fragility) fractures are predominantly caused by deteriorated bone strength, which results from aging or bone disease, and can occur due to a mechanical impact following e.g. slipping or falling.
- High-impact (or traumatic) fractures require excessive stress caused by traumatic accidents and can occur in healthy bone. Bone is considered weak when the risk for fragility fractures is increased.
- Weak bone is typically treated by systemic delivery of drug and growth factors. Such drugs and drug-like factors are absorbed throughout the body. Therefore, high doses may be required to gain sufficient therapeutic effects in the bone. However, the drug, especially at high drug doses, may cause side effects outside fracture sites, some of which may be severe.
- the object of this invention is to accomplish an improved technology for transport and deposition of objects into bone for effective and controllable localized management of bone health.
- a kit comprising an object delivery arrangement for delivering objects into bone, and retention means configured to counteract the passive diffusion out from the target and formed by one of a covering layer having a lower perfusion coefficient than the embracing tissues, an active object having a size sufficient to prevent passive diffusion out of the target in the bone, a substance that expands and covers the target site in the bone, subsequent application of mechanical waves for maintaining the substance in the target, after application of mechanical waves for depositing the substance, said arrangement comprising an ultrasound, photo-acoustics or plasma source configured to generate localized mechanical waves, and said arrangement being configured to perform localized deposition of the objects near bone, expose the objects and the bone to said localized mechanical waves to force the objects into the bone, and perform retention of the deposited objects in the bone, by using said retention means, so as to prevent the deposed objects from escaping the target site in the bone.
- the invention is defined in the appended set of claims.
- the invention is based on generation of localized mechanical waves into a tissue, and localized deposition of the objects near bone, and deposition of the objects to the bone by the effect of said mechanical waves.
- the direction of transportation and deposition of objects into bone tissue is not limited to transport and deposition from bone periosteal (i.e. outer) surface into bone tissue.
- the transportation and deposition of objects can also be achieved from any surface of a cavity (e.g. endosteal surface) or pore into bone tissue.
- the benefit of the invention is that the proposed conjunction of means permits an enhanced therapeutic power and advanced management of the therapeutic effect compared to the closest prior art.
- an object delivery arrangement for delivering objects into bone.
- the delivery object is, for example, a drug molecule or molecules for osteoporosis treatment.
- the arrangement comprises means for generating localized mechanical waves into a tissue.
- Said means are for example at least one of mechanical wave emitter 108, 113, energy conductor 109, sound source 111, and waveguide 112.
- Said means 108, 111 are for example an ultrasound transducer or an ultrasound source.
- the arrangement further comprises means for performing localized deposition of the objects 103 contained within a material boundary 104 near bone interface 107, and means for exposing the objects and the bone to said mechanical waves, obtaining deposition 110 of the objects to the bone.
- Said means for performing localized deposition are for example at least one of hollow structure 101, 201, cutting edge 102, reservoir 200 and syringe 203.
- the means for exposing are for example at least one of means according to reference signs 108, 109, 111, 112 and 113.
- the means 108, 109, 111, 112, 113 can penetrate skin 105 or tissues 106.
- the arrangement according to the present invention preferably comprises means for transporting the objects to the bone 107 in order to obtain deposition of the objects 110 to the bone.
- the means for transporting are for example at least one of means according to reference signs 101, 102, 103, 108 and 109.
- the location of weak bone is detected by quantitative ultrasound (QUS) or ultrasound imaging. After defining the weak bone, the deposition of objects into the weak bone can be achieved with mechanical waves generated with the same or a different ultrasound system than used for QUS or ultrasound imaging.
- the arrangement according to the present invention can comprise means, i.e. at least one of means according to reference signs 108, 113, 109, 111, 112 and 204a-e, for generating localized mechanical waves into the tissue to perform the localization on the basis of at least one of high-intensity focused ultrasound (HIFU) 204c, topological guides for ultrasound 112 and electromagnetic steering of the objects to improve focusing of the diffusion. It is essential for localized deposition to localize the driving mechanical (or sound) wave field inside the tissue, at the preferred point at or near the bone (e.g. a weak part of the bone).
- HIFU high-intensity focused ultrasound
- Localization of the driving mechanical wave field is realized either by means of high-intensity focused ultrasound (HIFU) or topological ultrasound (waveguides or high-order topologies i.e. fractal structures). Localization can also be realized by means of a counter electrode, or electromagnetic fields that steer the field or objects, to improve focusing of the diffusion.
- HIFU high-intensity focused ultrasound
- topological ultrasound waveguides or high-order topologies i.e. fractal structures.
- the arrangement can comprise means, i.e. at least one of means according to reference signs 108, 113, 109, 111, 112 and 204a-e, for generating localized mechanical waves into a tissue on the basis of time reversal ultrasound performing adaptive focusing.
- Time reversal ultrasound permits adaptive focusing through an inhomogeneous medium, such as soft tissue or trabecular bone.
- the arrangement can also comprise means, i.e. at least one of means according to reference signs 108, 113, 109, 111, 112, 200, 201, 203 and 204a-e, for performing localized deposition of the objects near bone based on photo-acoustic transformation in order to generate mechanical waves near objects.
- Photo-acoustic transformation also permits introduction of the sound source inside the tissue.
- the tissue is irradiated by electromagnetic waves (e.g. laser pulse), which penetrate and absorb into the tissue.
- the absorption causes localized thermal expansion, which results in emission of mechanical waves (i.e. sound field) at the point of thermal expansion.
- the resulting sound field is tunable by parameters of the optical beam (e.g.
- the energy of the electromagnetic beam can be absorbed in any parts of the tissue or in the objects that are being deposited or a combination of thereof.
- the optical absorption coefficients characteristic to different layers of the soft tissue and bone are functions of the optical wavelength.
- tuning of the optical wavelength permits e.g. maximization of the absorption ratio between the bone and soft tissue and results localization of the sound source at or near the bone.
- the localization can be also obtained by using a point source (e.g. 108, 113, 204a-b), independent of the technique of implementation.
- the arrangement comprises means for selecting the objects from a reservoir of objects and forcing the objects into the bone.
- Objects e.g. molecules
- the reservoir of objects e.g. solution
- the arrangement can comprise means, i.e. at least one of means according to reference signs 108, 113, 109, 111, 112, 200, 201, 203 and 204a-e, for performing retention of objects 103, 110 by depositing in addition to the objects a covering layer having a lower perfusion coefficient than the embracing tissues.
- the purpose of retention is to prevent the deposed objects from escaping the target (bone).
- Retention is realized by deposing another covering layer that has much lower perfusion coefficient compared to those of the embracing tissues. This can also be realized by depositing a substance that expands and covers the target.
- An alternative embodiment to this approach is using an active object, which is too large for passive diffusion, but can be actively deposited using the method described in the invention.
- the large size then prevents passive diffusion out of the target. Retention can also be controlled by subsequent sonication: the first application of mechanical waves deposits the substance into the target, followed by several applications of mechanical waves that maintain the substance in the target (counteract the passive diffusion out from the target).
- the arrangement can comprise means, i.e. at least one of means according to reference signs 101, 102, 103, 108, 113, 109, 111, 112, 200, 201, 203 and 204a-e, for activating objects selectively at different time points.
- Objects that are inactive in the tissue are first driven in, to form a reservoir 103, 104, 110 of the objects in the tissue. After this, the objects are collectively or selectively activated e.g. by means of mechanical waves, electromagnetic waves or temperature. Selective activation permits activation at different time points, e.g. one ingredient of the objects can be activated directly after drive in and another ingredient can be activated later. This can be considered to be e.g.
- the different drugs are encapsulated in or on surface of e.g. gas voids (with or without lipid shells or equivalent) of various sizes corresponding to various resonant frequencies. After driving in, the encapsulated objects are released at desired moments by sonicating at the resonant frequency corresponding to the release of objects desired.
- gas voids with or without lipid shells or equivalent
- the arrangement can also comprise means, i.e. at least one of means according to reference signs 108, 113, 109, 111, 112, 204a-e, for affecting tissue 107, 105, 106, 110 and 205 by mechanical vibrations.
- means i.e. at least one of means according to reference signs 108, 113, 109, 111, 112, 204a-e, for affecting tissue 107, 105, 106, 110 and 205 by mechanical vibrations.
- one, two or several components are driven in the tissue and then treated (shaken) by mechanical vibration. This shaking causes merging of the components to larger aggregates that cannot escape from the target tissue (e.g. bone) or whose escape rate is decreased.
- the arrangement according to the present invention can comprise nanostructure means to control diffusion and to amplify the diffusion.
- Nano-swimmers or functionalized nano-rods permit improved control of the diffusion and amplification of the diffusion.
- the same can also be accomplished e.g. by nano motors, which are controlled by at least one of external field, internal field, external power source and internal power source.
- the arrangement can comprise means (204a-e) for exposing the objects and the bone to said mechanical waves to deposit the objects to the bone utilizing at least one of blood circulation and the bone marrow cavity for the transportation of the objects.
- the objects are driven in bone from the inside (e.g. endosteal side) (means 204b), utilizing blood circulation and/or the bone marrow cavity for the initial transport of the objects to the treatment site, e.g. the fracture site, and then exploiting mechanical waves to deposit the objects into the bone.
- the arrangement according to the present invention can comprise multi-center-frequency means, i.e. at least one of means according to reference signs 108, 113, 109, 111, 112 and 204a-e, to generate mechanical waves of at least two different frequencies in order to improve transportation of the objects and deposition of the objects.
- multi-center-frequency means i.e. at least one of means according to reference signs 108, 113, 109, 111, 112 and 204a-e, to generate mechanical waves of at least two different frequencies in order to improve transportation of the objects and deposition of the objects.
- a kilohertz frequency transducer e.g. 204d
- a megahertz frequency transducer e.g. 204e
- the means i.e. at least one of means according to reference signs 108, 113, 109, 111, 112 and 204a-e, for generating localized mechanical waves into a tissue can comprise a plasma source.
- the driving pressure field is generated by a plasma source.
- the plasma source can be realized e.g. by a focused laser or a spark gap.
- FIG. 1 depicts the preferred embodiment of the invention.
- a catheter (101) featuring a cutting edge (102) perforates the skin and tissue.
- the catheter delivers the objects (103) and it forms an object reservoir (103) with boundary 104 near the bone surface (107).
- the sound source comprises energy conductor (109) and a sound emitter (108).
- the sound emitter 108 may be e.g. a flat piezo, a focused piezo, spark, laser induced spark, EMUT (Energy Mode Ultrasound Transducer), CMUT (Capacitive micromachined ultrasonic transducers), PMUT (Piezoelectric Micromachined Ultrasonic Transducers) and equivalent.
- the mechanical wave generated by the sound emitter translates the objects into the bone (110).
- the sound source (111) is located outside the tissue and the mechanical wave is transmitted to the tissue and active ingredient via a waveguide (112).
- a waveguide (112) e.g. 10 - 500kHz mechanical wave is transmitted through at least one of waveguide (112), active ingredient (103), tissue (106) and sound emitter (108). This mechanical wave alters the permeability of the bone membrane.
- Another, e.g. 0,5 - 50 MHz mechanical wave is subsequently transmitted to the boundary. This mechanical wave deposits the active ingredient from the reservoir to the bone.
- 111 is a light source and the light wave is guided to reservoir 103, 104 and bone 107 through an optical fiber 112 or reflecting inner wall of a catheter 101.
- the light wave is absorbed by active ingredient 103 or bone 107 to generate light-induced sound waves for translating the active ingredient 103 into the bone 107.
- the object can be e.g. molecules, drugs, vehicles carrying the object, imaging contrast agent, minerals or nanofibers.
- Biologically active materials that may be of interest include analgesics, antagonists, anti-inflammatory agents, anthelmintics, antianginal agents, antiarrhythmic agents, antibiotics (including penicillins), anticholesterols, anticoagulants, anticonvulsants, antidepressants, antidiabetic agents, antiepileptics, antigonadotropins, antihistamines, antihypertensive agents, antimuscarinic agents, antimycobacterial agents, antineoplastic agents, antipsychotic agents, immunosuppressants, antithyroid agents, antiviral agents, antifungal agents, anxiolytic sedatives (hypnotics and neuroleptics), astringents, beta-adrenoceptor blocking agents, blood products and substitutes, anti-cancer agents, cardiacinotropic agents, contrast media, corticosterioids, cough suppressants (expectorants
- Exemplary medicaments can be e.g. ibandronic acid, zolendronic acid, teriparatide, denosumab, TGF-beta, FGF-beta and BB1/biopharm,
- the sound emitter (108) is a confocal transducer featuring two transducers of different center frequencies.
- the two frequencies generate a third frequency which acts as the wave translating the active ingredient.
- catheter wall (101) or waveguide (112) acts as a "cold finger", i.e. heat energy is absorbed from the tissues exposed to ultrasound induced heating.
- Figure 2 depicts another means of translating objects, e.g. active ingredients, into bone.
- a syringe (203) loaded with the objects containing active ingredient (200), is connected with a needle (201) to a major artery which transports the objects with the blood flow to the treatment site.
- An ultrasound generator (204a) comprising at least one of 101, 103, 108, 111, 112, 113, 109 generates the ultrasound which locally translates the drug into the bone.
- the ultrasound system operates intravenously (204b).
- the ultrasound waves are focused through the skin and tissue to the bone - reservoir 103 boundary 104, 107 with an ultrasound generator (204c).
- a combination ultrasound system comprising of two different transducers, one of which (204d) translates the active ingredient 205 through the tissue and skin (such as sonophoresis), whereas the other one (204e) translates the active ingredient into the bone.
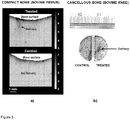
- Figure 3 shows preliminary results on compact cortical and spongy cancellous bone.
- (a) Optical microscopy image of cortical bone into which we have delivered contrast agent (methylene blue; image on top) by using high-intensity focused ultrasound (HIU) (Parameters: sine burst frequency: 2.17 MHz; cycles per burst: 200, pulse-repetition frequency: 1000 Hz).
- the gray scale represents optical absorption.
- the ultrasound beam enhanced the delivery, as is indicated by an arrow. There is no similar effect seen in a control sample (image on bottom), extracted from the same piece of bone and treated consistently but without ultrasound.
- (b) Photograph of the result of a related experiment in cancellous bone (sine burst frequency: 2.17 MHz; cycles per burst: 100, pulse-repetition frequency: 600 Hz).
- Localized delivery of objects into the bone includes transport and deposition according to the preferred or alternative embodiments of the invention as described in Figures 1 and 2 .
- one object or a group of objects is transported into the bone.
- a second object is transported into bone.
- the second object is delivered close to the pathways through which the first object has travelled to prevent washout of first object.
- the second object can alternatively self-assemble with itself or with the first object to create large-sized constructs (e.g. via mechanisms such as self-assembly) to slow down or prevent washout of the objects with therapeutic effect.
- the role of the second object can also be to catalyze the therapeutic effect of first object.
- the catalyzation can be achieved also by exposing at least one of the objects to mechanical or electromagnetic waves.
- Localized deposition is realized by employing one of the presented or different combinations of the presented techniques and methods.
Description
- The invention relates to bone healthcare and health management. The invention deals with detection of a weak bone and healing of the weak or fractured bone in vivo.
- Bone diseases are disorders in remodeling of bone tissue. As a result, bones can become mechanically weak. Reduction of bone mineral density (BMD) is a natural process related to aging after the age of 20. However, some bone diseases, such as osteoporosis, can cause excessive loss of BMD. Deficiencies in nutrient intake (e.g., calcium and vitamin D and C), hormonal imbalance and cell abnormalities can also cause bone disorders.
- Bone fractures are labelled low-impact fractures and high-impact fractures. The low-impact (or fragility) fractures are predominantly caused by deteriorated bone strength, which results from aging or bone disease, and can occur due to a mechanical impact following e.g. slipping or falling. High-impact (or traumatic) fractures require excessive stress caused by traumatic accidents and can occur in healthy bone. Bone is considered weak when the risk for fragility fractures is increased.
- There is a need for methods to detect and heal weak bones, preferably before fractures occur.
- Localized inference of bone quality techniques are being developed by several research groups. To this end, quantitative ultrasound (QUS) is one of the most promising approaches. Yet, ultrasonic detection of clinically relevant fracture sites such as the hip and vertebrae is challenging and requires further development.
- Weak bone is typically treated by systemic delivery of drug and growth factors. Such drugs and drug-like factors are absorbed throughout the body. Therefore, high doses may be required to gain sufficient therapeutic effects in the bone. However, the drug, especially at high drug doses, may cause side effects outside fracture sites, some of which may be severe.
- Tissue treatment based on localized delivery and release of drugs has been reported for soft tissue sites. In particular, a recent report details ultrasound-aided delivery and release in articular cartilage (Nieminen et al., Ultrasound Med Biol 41(8):2259-2268, 2015) and subchondral bone through articular cartilage (Nieminen et al., Ultrasonics Symposium (IUS), 2012 IEEE International, pages 1869 - 1872). For bone metastases, there are reports on localized ultrasound-aided release of drugs, first transported into the vicinity of the therapy site by blood circulation (Staruch et al., Radiology 263(1):117-127, 2012). However, there is no known method to do simultaneous release and deposition. Moreover, there is no known methodology that would permit construction of a hand-held device for detection of weak bone (site with fracture risk) followed by instant localized treatment.
- In prior art document
US6231528 B1 is presented an in vivo technology for using ultrasound in conjunction with a biomedical compound or bone growth factor to induce healing, growth and ingrowth responses in bone. To this end, non-invasively applied ultrasonic stimulus is operative to transport the bone growth factor from the external surface of the soft tissue to the bone and to synergistically enhance the interaction between the bone growth factor and the bone. This technology does not involve deposition of the ultrasonically transported objects and describes the use of ultrasound for delivery only in the context of extracorporeal ultrasound transducer, ultrasound pulser, biomedical compounds and bone growth factors. In addition, the technology does not incorporate focused ultrasonic waves which are vital for highly localized treatment. - Furthermore, reference is made to document
US2015/125808 A1 . - The object of this invention is to accomplish an improved technology for transport and deposition of objects into bone for effective and controllable localized management of bone health. This is achieved by a kit, comprising an object delivery arrangement for delivering objects into bone, and retention means configured to counteract the passive diffusion out from the target and formed by one of a covering layer having a lower perfusion coefficient than the embracing tissues, an active object having a size sufficient to prevent passive diffusion out of the target in the bone, a substance that expands and covers the target site in the bone, subsequent application of mechanical waves for maintaining the substance in the target, after application of mechanical waves for depositing the substance, said arrangement comprising an ultrasound, photo-acoustics or plasma source configured to generate localized mechanical waves, and said arrangement being configured to perform localized deposition of the objects near bone, expose the objects and the bone to said localized mechanical waves to force the objects into the bone, and perform retention of the deposited objects in the bone, by using said retention means, so as to prevent the deposed objects from escaping the target site in the bone.
- The invention is defined in the appended set of claims. The invention is based on generation of localized mechanical waves into a tissue, and localized deposition of the objects near bone, and deposition of the objects to the bone by the effect of said mechanical waves.
- The direction of transportation and deposition of objects into bone tissue is not limited to transport and deposition from bone periosteal (i.e. outer) surface into bone tissue. The transportation and deposition of objects can also be achieved from any surface of a cavity (e.g. endosteal surface) or pore into bone tissue.
- The benefit of the invention is that the proposed conjunction of means permits an enhanced therapeutic power and advanced management of the therapeutic effect compared to the closest prior art.
-
- Figure 1
- shows preferred embodiments of the invention.
- Figure 2
- shows alternative embodiments of the invention.
- Figure 3
- shows preliminary results.
- In the present invention is presented an object delivery arrangement for delivering objects into bone. The delivery object is, for example, a drug molecule or molecules for osteoporosis treatment. The arrangement comprises means for generating localized mechanical waves into a tissue. Said means are for example at least one of
mechanical wave emitter energy conductor 109,sound source 111, andwaveguide 112. Said means 108, 111 are for example an ultrasound transducer or an ultrasound source. The arrangement further comprises means for performing localized deposition of theobjects 103 contained within amaterial boundary 104 nearbone interface 107, and means for exposing the objects and the bone to said mechanical waves, obtainingdeposition 110 of the objects to the bone. Said means for performing localized deposition are for example at least one ofhollow structure cutting edge 102,reservoir 200 andsyringe 203. The means for exposing are for example at least one of means according toreference signs means skin 105 ortissues 106. - The arrangement according to the present invention preferably comprises means for transporting the objects to the
bone 107 in order to obtain deposition of theobjects 110 to the bone. The means for transporting are for example at least one of means according toreference signs - The arrangement according to the present invention can comprise means, i.e. at least one of means according to
reference signs ultrasound 112 and electromagnetic steering of the objects to improve focusing of the diffusion. It is essential for localized deposition to localize the driving mechanical (or sound) wave field inside the tissue, at the preferred point at or near the bone (e.g. a weak part of the bone). Localization of the driving mechanical wave field is realized either by means of high-intensity focused ultrasound (HIFU) or topological ultrasound (waveguides or high-order topologies i.e. fractal structures). Localization can also be realized by means of a counter electrode, or electromagnetic fields that steer the field or objects, to improve focusing of the diffusion. - In one embodiment according to the present invention the arrangement can comprise means, i.e. at least one of means according to
reference signs - The arrangement can also comprise means, i.e. at least one of means according to
reference signs - In another embodiment according to the present invention, the arrangement comprises means for selecting the objects from a reservoir of objects and forcing the objects into the bone. Objects (e.g. molecules) are selected from the reservoir of objects (e.g. solution) and are forced into the bone.
- The arrangement can comprise means, i.e. at least one of means according to
reference signs objects - In one further embodiment according to the present invention the arrangement can comprise means, i.e. at least one of means according to
reference signs reservoir - The arrangement can also comprise means, i.e. at least one of means according to
reference signs tissue - In nanotechnology embodiments according to the present invention the arrangement according to the present invention can comprise nanostructure means to control diffusion and to amplify the diffusion. Nano-swimmers or functionalized nano-rods permit improved control of the diffusion and amplification of the diffusion. The same can also be accomplished e.g. by nano motors, which are controlled by at least one of external field, internal field, external power source and internal power source.
- In one further embodiment according to the present invention the arrangement can comprise means (204a-e) for exposing the objects and the bone to said mechanical waves to deposit the objects to the bone utilizing at least one of blood circulation and the bone marrow cavity for the transportation of the objects. Alternatively, instead of driving from the periosteal side of the bone, the objects are driven in bone from the inside (e.g. endosteal side) (means 204b), utilizing blood circulation and/or the bone marrow cavity for the initial transport of the objects to the treatment site, e.g. the fracture site, and then exploiting mechanical waves to deposit the objects into the bone.
- In further embodiments the arrangement according to the present invention can comprise multi-center-frequency means, i.e. at least one of means according to
reference signs - Generation of sound waves by at least at two distinct center-frequencies can enhance the drive in. For instance, a kilohertz frequency transducer (e.g. 204d) can be used to increase the permeability at the bone surface (e.g. periosteum or endosteum) and a megahertz frequency transducer (e.g. 204e) can be used to push the objects in.
- In embodiments according to the present invention the means, i.e. at least one of means according to
reference signs -
Figure 1 depicts the preferred embodiment of the invention. A catheter (101) featuring a cutting edge (102) perforates the skin and tissue. The catheter delivers the objects (103) and it forms an object reservoir (103) withboundary 104 near the bone surface (107). The sound source comprises energy conductor (109) and a sound emitter (108). Thesound emitter 108 may be e.g. a flat piezo, a focused piezo, spark, laser induced spark, EMUT (Energy Mode Ultrasound Transducer), CMUT (Capacitive micromachined ultrasonic transducers), PMUT (Piezoelectric Micromachined Ultrasonic Transducers) and equivalent. The mechanical wave generated by the sound emitter translates the objects into the bone (110). In another embodiment the sound source (111) is located outside the tissue and the mechanical wave is transmitted to the tissue and active ingredient via a waveguide (112). In one embodiment of the invention e.g. 10 - 500kHz mechanical wave is transmitted through at least one of waveguide (112), active ingredient (103), tissue (106) and sound emitter (108). This mechanical wave alters the permeability of the bone membrane. Another, e.g. 0,5 - 50 MHz mechanical wave is subsequently transmitted to the boundary. This mechanical wave deposits the active ingredient from the reservoir to the bone. - According to one
embodiment 111 is a light source and the light wave is guided toreservoir bone 107 through anoptical fiber 112 or reflecting inner wall of acatheter 101. The light wave is absorbed byactive ingredient 103 orbone 107 to generate light-induced sound waves for translating theactive ingredient 103 into thebone 107. - The object can be e.g. molecules, drugs, vehicles carrying the object, imaging contrast agent, minerals or nanofibers. Biologically active materials that may be of interest include analgesics, antagonists, anti-inflammatory agents, anthelmintics, antianginal agents, antiarrhythmic agents, antibiotics (including penicillins), anticholesterols, anticoagulants, anticonvulsants, antidepressants, antidiabetic agents, antiepileptics, antigonadotropins, antihistamines, antihypertensive agents, antimuscarinic agents, antimycobacterial agents, antineoplastic agents, antipsychotic agents, immunosuppressants, antithyroid agents, antiviral agents, antifungal agents, anxiolytic sedatives (hypnotics and neuroleptics), astringents, beta-adrenoceptor blocking agents, blood products and substitutes, anti-cancer agents, cardiacinotropic agents, contrast media, corticosterioids, cough suppressants (expectorants and muco-lytics), diuretics, dopaminergics (antiparkinsonian agents), haemostatics, immunosuppressive and immunoactive agents, lipid regulating agents, muscle relaxants, parasympathomimetics, parathyroid calcitonin and biphospho-nates, prostaglandins, radiopharmaceuticals, sex hormones (including steroids), anti-allergic agents, stimulants and anorexics, sympathomimetics, thyroid agents, vasidilators, neuron blocking agents, anticholinergic and choli-nomimetic agents, antimuscarinic and muscarinic agents, vitamins, and xanthines. Exemplary medicaments can be e.g. ibandronic acid, zolendronic acid, teriparatide, denosumab, TGF-beta, FGF-beta and BB1/biopharm, According to one embodiment, the sound emitter (108) is a confocal transducer featuring two transducers of different center frequencies. According to one embodiment, the two frequencies generate a third frequency which acts as the wave translating the active ingredient.
- According to one embodiment, catheter wall (101) or waveguide (112) acts as a "cold finger", i.e. heat energy is absorbed from the tissues exposed to ultrasound induced heating.
-
Figure 2 depicts another means of translating objects, e.g. active ingredients, into bone. A syringe (203), loaded with the objects containing active ingredient (200), is connected with a needle (201) to a major artery which transports the objects with the blood flow to the treatment site. An ultrasound generator (204a), comprising at least one of 101, 103, 108, 111, 112, 113, 109 generates the ultrasound which locally translates the drug into the bone. In an alternative embodiment the ultrasound system operates intravenously (204b). In another alternative embodiment, the ultrasound waves are focused through the skin and tissue to the bone -reservoir 103boundary active ingredient 205 through the tissue and skin (such as sonophoresis), whereas the other one (204e) translates the active ingredient into the bone. -
Figure 3 shows preliminary results on compact cortical and spongy cancellous bone. (a) Optical microscopy image of cortical bone into which we have delivered contrast agent (methylene blue; image on top) by using high-intensity focused ultrasound (HIU) (Parameters: sine burst frequency: 2.17 MHz; cycles per burst: 200, pulse-repetition frequency: 1000 Hz). The gray scale represents optical absorption. The ultrasound beam enhanced the delivery, as is indicated by an arrow. There is no similar effect seen in a control sample (image on bottom), extracted from the same piece of bone and treated consistently but without ultrasound. (b) Photograph of the result of a related experiment in cancellous bone (sine burst frequency: 2.17 MHz; cycles per burst: 100, pulse-repetition frequency: 600 Hz). - Localized delivery of objects into the bone includes transport and deposition according to the preferred or alternative embodiments of the invention as described in
Figures 1 and2 . In one phase, one object or a group of objects is transported into the bone. In the second phase, a second object is transported into bone. The second object is delivered close to the pathways through which the first object has travelled to prevent washout of first object. The second object can alternatively self-assemble with itself or with the first object to create large-sized constructs (e.g. via mechanisms such as self-assembly) to slow down or prevent washout of the objects with therapeutic effect. The role of the second object can also be to catalyze the therapeutic effect of first object. The catalyzation can be achieved also by exposing at least one of the objects to mechanical or electromagnetic waves. - Localized deposition is realized by employing one of the presented or different combinations of the presented techniques and methods.
Claims (11)
- A kit, comprising
an object delivery arrangement (101, 111, 201, 203, 204a) for delivering objects (110) into bone, and retention means (108, 113, 109, 111, 112, 200, 201, 203, 204a-e) formed by one of- a covering layer having a lower perfusion coefficient than the embracing tissues,- an active object having a size sufficient to prevent passive diffusion out of the target in the bone,- a substance configured to expand and cover the target site in the bone, and- means for subsequent application of mechanical waves for maintaining the substance in the target, after application of mechanical waves for depositing the substance,wherein said object delivery arrangement comprises an ultrasound, photo-acoustics or plasma source configured to generate localized mechanical waves, and
wherein said object delivery arrangement is configured to- perform localized deposition of the objects near bone,- expose the objects and the bone to said localized mechanical waves to force the objects into the bone, and- wherein said retention means is configured to perform retention of the deposited objects in the bone, so as to prevent the deposed objects from escaping the target site in the bone. - A kit according to claim 1, characterised in that the object delivery arrangement comprises means (101, 102, 103, 108, 109) for transporting the objects to the bone in order to obtain deposition of the objects into the bone.
- A kit according to claim 1, characterised in that the object delivery arrangement comprises means (108, 113) for generating localized mechanical waves into the tissue performing the localization on the basis of at least one of high-intensity focused ultrasound (HIFU), waveguide, electromagnetic steering of the wave field and electromagnetic steering of the object in order to improve focusing of the diffusion.
- A kit according to claim 1, characterised in that the object delivery arrangement comprises means (108, 113) for generating localized mechanical waves into a tissue on the basis of time reversal ultrasound performing adaptive focusing.
- A kit according to claim 1, characterised in that the object delivery arrangement comprises means for performing localized deposition of the objects near bone on the basis of photo-acoustic transformation in order to localize the means for generating localized mechanical waves inside the tissue and in order to reduce ultrasonic energy deposition in tissue adjacent to bone relative to that in bone.
- A kit according to claim 1, characterised in that the object delivery arrangement comprises means for selecting objects from the reservoir of objects and forcing the objects into the bone tissue.
- A kit according to claim 1, characterised in that the object delivery arrangement comprises means for activating objects selectively at different time points.
- A kit according to claim 1, characterised in that the object delivery arrangement comprises means for affecting a target structure by mechanical vibrations for enhanced deposition.
- A kit according to claim 1, characterised in that the object delivery arrangement comprises nanostructure means to achieve at least one of control diffusion and amplification of the diffusion.
- A kit according to claim 1, characterised in that the object delivery arrangement comprises means (204a-e) for exposing the objects and the bone to said mechanical waves to obtain deposition of the objects to the bone utilizing at least one of blood circulation and the bone marrow cavity for the transportation of the objects.
- A kit according to claim 1, characterised in that the object delivery arrangement comprises multi-center-frequency means to generate mechanical waves of at least two different frequencies in order to improve transportation of the objects and deposition of the objects.
Applications Claiming Priority (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| PCT/FI2015/050589 WO2017042422A1 (en) | 2015-09-09 | 2015-09-09 | Apparatus for delivery of substances into bone |
Publications (2)
| Publication Number | Publication Date |
|---|---|
| EP3347081A1 EP3347081A1 (en) | 2018-07-18 |
| EP3347081B1 true EP3347081B1 (en) | 2020-03-04 |
Family
ID=54207543
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| EP15771668.9A Active EP3347081B1 (en) | 2015-09-09 | 2015-09-09 | Apparatus for delivery of substances into bone |
Country Status (7)
| Country | Link |
|---|---|
| US (1) | US20180193075A1 (en) |
| EP (1) | EP3347081B1 (en) |
| JP (1) | JP6474941B2 (en) |
| CN (1) | CN108025165B (en) |
| AU (1) | AU2015408449B2 (en) |
| ES (1) | ES2796236T3 (en) |
| WO (1) | WO2017042422A1 (en) |
Family Cites Families (7)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JPH11267229A (en) * | 1998-03-19 | 1999-10-05 | Takeshi Nishisaka | Bone forming device |
| US6231528B1 (en) | 1999-01-15 | 2001-05-15 | Jonathan J. Kaufman | Ultrasonic and growth factor bone-therapy: apparatus and method |
| US9302087B2 (en) * | 2002-02-14 | 2016-04-05 | Gholam A. Peyman | Method and composition for hyperthermally treating cells |
| US8551096B2 (en) * | 2009-05-13 | 2013-10-08 | Boston Scientific Scimed, Inc. | Directional delivery of energy and bioactives |
| EP2718433A4 (en) * | 2011-06-10 | 2015-07-29 | Univ New York State Res Found | Method and device for non-invasive acoustic stimulation of stem cells and progenitor cells in a patient |
| GB201212222D0 (en) * | 2012-07-10 | 2012-08-22 | Univ Dundee | Improved apparatus and method for mineralising biological materials |
| US10835767B2 (en) * | 2013-03-08 | 2020-11-17 | Board Of Regents, The University Of Texas System | Rapid pulse electrohydraulic (EH) shockwave generator apparatus and methods for medical and cosmetic treatments |
-
2015
- 2015-09-09 JP JP2018512417A patent/JP6474941B2/en active Active
- 2015-09-09 AU AU2015408449A patent/AU2015408449B2/en active Active
- 2015-09-09 EP EP15771668.9A patent/EP3347081B1/en active Active
- 2015-09-09 WO PCT/FI2015/050589 patent/WO2017042422A1/en active Application Filing
- 2015-09-09 ES ES15771668T patent/ES2796236T3/en active Active
- 2015-09-09 CN CN201580083006.2A patent/CN108025165B/en active Active
-
2018
- 2018-03-09 US US15/917,044 patent/US20180193075A1/en not_active Abandoned
Non-Patent Citations (1)
| Title |
|---|
| None * |
Also Published As
| Publication number | Publication date |
|---|---|
| WO2017042422A1 (en) | 2017-03-16 |
| JP6474941B2 (en) | 2019-02-27 |
| CN108025165A (en) | 2018-05-11 |
| US20180193075A1 (en) | 2018-07-12 |
| AU2015408449B2 (en) | 2018-04-12 |
| JP2018527093A (en) | 2018-09-20 |
| AU2015408449A1 (en) | 2018-03-22 |
| CN108025165B (en) | 2019-01-01 |
| ES2796236T3 (en) | 2020-11-26 |
| EP3347081A1 (en) | 2018-07-18 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| JP7016325B2 (en) | Methods and equipment for extraction and delivery of entities | |
| US11534187B2 (en) | Acoustic therapy device | |
| US8568339B2 (en) | Single element ultrasound transducer with multiple driving circuits | |
| Xu et al. | Evolution of bubble clouds induced by pulsed cavitational ultrasound therapy-histotripsy | |
| JP2003526403A (en) | Method and kit for cavitation induced tissue treatment with low intensity ultrasound | |
| Couture et al. | In vivo targeted delivery of large payloads with an ultrasound clinical scanner | |
| JP2023111991A (en) | Novel transfection and drug delivery device | |
| US20200406017A1 (en) | Technique and method to locally deliver objects into bone | |
| US20070232987A1 (en) | One-hand-operated ultrasound transducer and method for delivering a controlled and uniform distribution of a sterile or a non-sterile topical reagent to skin for use in diagnostic, therapeutic, and aesthetic therapies | |
| EP3347081B1 (en) | Apparatus for delivery of substances into bone | |
| CN109414243A (en) | The external ultrasonic treated for spinal cord and spinal nerve generates therapeutic device, the equipment including the device and the method for implementing the device | |
| JPH07213622A (en) | Chemical dosing device | |
| KR20160119077A (en) | Liquid vehicle for suspension of undelivered particles | |
| Singh et al. | Ultrasound-guided Drug Delivery and Tissue Engineering | |
| van Blokland | Instigating and monitoring transdermal drug delivery using ultrasound-mediated cavitation | |
| JPH0698917A (en) | Ultrasonic cell membrane matching vibration treatment of malignant tumor | |
| DE102004004622A1 (en) | Arrangement for transdermal delivery of a medicament by sonophoresis comprises a medicament carrier, an ultrasound generator and a sonic coupler | |
| Visuri et al. | Optically generated ultrasound for enhanced drug delivery | |
| WO2004100850A2 (en) | Method of applying gas contact ultrasound therapy, surgery, and drug delivery |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| STAA | Information on the status of an ep patent application or granted ep patent |
Free format text: STATUS: THE INTERNATIONAL PUBLICATION HAS BEEN MADE |
|
| PUAI | Public reference made under article 153(3) epc to a published international application that has entered the european phase |
Free format text: ORIGINAL CODE: 0009012 |
|
| STAA | Information on the status of an ep patent application or granted ep patent |
Free format text: STATUS: REQUEST FOR EXAMINATION WAS MADE |
|
| 17P | Request for examination filed |
Effective date: 20180220 |
|
| AK | Designated contracting states |
Kind code of ref document: A1 Designated state(s): AL AT BE BG CH CY CZ DE DK EE ES FI FR GB GR HR HU IE IS IT LI LT LU LV MC MK MT NL NO PL PT RO RS SE SI SK SM TR |
|
| AX | Request for extension of the european patent |
Extension state: BA ME |
|
| DAV | Request for validation of the european patent (deleted) | ||
| DAX | Request for extension of the european patent (deleted) | ||
| GRAP | Despatch of communication of intention to grant a patent |
Free format text: ORIGINAL CODE: EPIDOSNIGR1 |
|
| STAA | Information on the status of an ep patent application or granted ep patent |
Free format text: STATUS: GRANT OF PATENT IS INTENDED |
|
| INTG | Intention to grant announced |
Effective date: 20190930 |
|
| RIN1 | Information on inventor provided before grant (corrected) |
Inventor name: MOILANEN, PETRO Inventor name: NIEMINEN, HEIKKI Inventor name: SALMI, ARI Inventor name: GARCIA PEREZ, ALEJANDRO Inventor name: HAEGGSTROEM, EDWARD |
|
| GRAS | Grant fee paid |
Free format text: ORIGINAL CODE: EPIDOSNIGR3 |
|
| GRAA | (expected) grant |
Free format text: ORIGINAL CODE: 0009210 |
|
| STAA | Information on the status of an ep patent application or granted ep patent |
Free format text: STATUS: THE PATENT HAS BEEN GRANTED |
|
| AK | Designated contracting states |
Kind code of ref document: B1 Designated state(s): AL AT BE BG CH CY CZ DE DK EE ES FI FR GB GR HR HU IE IS IT LI LT LU LV MC MK MT NL NO PL PT RO RS SE SI SK SM TR |
|
| REG | Reference to a national code |
Ref country code: GB Ref legal event code: FG4D |
|
| REG | Reference to a national code |
Ref country code: CH Ref legal event code: EP |
|
| REG | Reference to a national code |
Ref country code: AT Ref legal event code: REF Ref document number: 1239683 Country of ref document: AT Kind code of ref document: T Effective date: 20200315 |
|
| REG | Reference to a national code |
Ref country code: DE Ref legal event code: R096 Ref document number: 602015048206 Country of ref document: DE |
|
| REG | Reference to a national code |
Ref country code: IE Ref legal event code: FG4D |
|
| REG | Reference to a national code |
Ref country code: FI Ref legal event code: FGE |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: NO Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20200604 Ref country code: RS Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20200304 |
|
| REG | Reference to a national code |
Ref country code: NL Ref legal event code: MP Effective date: 20200304 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: GR Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20200605 Ref country code: HR Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20200304 Ref country code: LV Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20200304 Ref country code: SE Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20200304 Ref country code: BG Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20200604 |
|
| REG | Reference to a national code |
Ref country code: LT Ref legal event code: MG4D |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: NL Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20200304 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: PT Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20200729 Ref country code: RO Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20200304 Ref country code: CZ Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20200304 Ref country code: SM Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20200304 Ref country code: EE Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20200304 Ref country code: LT Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20200304 Ref country code: IS Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20200704 Ref country code: SK Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20200304 |
|
| REG | Reference to a national code |
Ref country code: AT Ref legal event code: MK05 Ref document number: 1239683 Country of ref document: AT Kind code of ref document: T Effective date: 20200304 |
|
| REG | Reference to a national code |
Ref country code: ES Ref legal event code: FG2A Ref document number: 2796236 Country of ref document: ES Kind code of ref document: T3 Effective date: 20201126 |
|
| REG | Reference to a national code |
Ref country code: DE Ref legal event code: R097 Ref document number: 602015048206 Country of ref document: DE |
|
| PLBE | No opposition filed within time limit |
Free format text: ORIGINAL CODE: 0009261 |
|
| STAA | Information on the status of an ep patent application or granted ep patent |
Free format text: STATUS: NO OPPOSITION FILED WITHIN TIME LIMIT |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: AT Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20200304 Ref country code: DK Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20200304 |
|
| 26N | No opposition filed |
Effective date: 20201207 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: PL Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20200304 Ref country code: SI Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20200304 |
|
| REG | Reference to a national code |
Ref country code: CH Ref legal event code: PL |
|
| REG | Reference to a national code |
Ref country code: BE Ref legal event code: MM Effective date: 20200930 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: LU Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20200909 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: IE Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20200909 Ref country code: CH Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20200930 Ref country code: BE Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20200930 Ref country code: LI Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20200930 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: TR Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20200304 Ref country code: MT Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20200304 Ref country code: CY Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20200304 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: MK Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20200304 Ref country code: MC Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20200304 Ref country code: AL Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20200304 |
|
| P01 | Opt-out of the competence of the unified patent court (upc) registered |
Effective date: 20230529 |
|
| PGFP | Annual fee paid to national office [announced via postgrant information from national office to epo] |
Ref country code: GB Payment date: 20230920 Year of fee payment: 9 Ref country code: FI Payment date: 20230920 Year of fee payment: 9 |
|
| PGFP | Annual fee paid to national office [announced via postgrant information from national office to epo] |
Ref country code: FR Payment date: 20230928 Year of fee payment: 9 Ref country code: DE Payment date: 20230920 Year of fee payment: 9 |
|
| PGFP | Annual fee paid to national office [announced via postgrant information from national office to epo] |
Ref country code: ES Payment date: 20231124 Year of fee payment: 9 |
|
| PGFP | Annual fee paid to national office [announced via postgrant information from national office to epo] |
Ref country code: IT Payment date: 20230927 Year of fee payment: 9 |