EP3346052A1 - Method for manufacturing processed fiber, processed fiber, method for suppressing damage to animal fiber, and method for processing animal fiber - Google Patents
Method for manufacturing processed fiber, processed fiber, method for suppressing damage to animal fiber, and method for processing animal fiber Download PDFInfo
- Publication number
- EP3346052A1 EP3346052A1 EP16841846.5A EP16841846A EP3346052A1 EP 3346052 A1 EP3346052 A1 EP 3346052A1 EP 16841846 A EP16841846 A EP 16841846A EP 3346052 A1 EP3346052 A1 EP 3346052A1
- Authority
- EP
- European Patent Office
- Prior art keywords
- fibers
- bleaching
- keratin
- hydrolyzed keratin
- hairs
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Pending
Links
Images
Classifications
-
- D—TEXTILES; PAPER
- D06—TREATMENT OF TEXTILES OR THE LIKE; LAUNDERING; FLEXIBLE MATERIALS NOT OTHERWISE PROVIDED FOR
- D06M—TREATMENT, NOT PROVIDED FOR ELSEWHERE IN CLASS D06, OF FIBRES, THREADS, YARNS, FABRICS, FEATHERS OR FIBROUS GOODS MADE FROM SUCH MATERIALS
- D06M15/00—Treating fibres, threads, yarns, fabrics, or fibrous goods made from such materials, with macromolecular compounds; Such treatment combined with mechanical treatment
- D06M15/01—Treating fibres, threads, yarns, fabrics, or fibrous goods made from such materials, with macromolecular compounds; Such treatment combined with mechanical treatment with natural macromolecular compounds or derivatives thereof
- D06M15/15—Proteins or derivatives thereof
-
- D—TEXTILES; PAPER
- D06—TREATMENT OF TEXTILES OR THE LIKE; LAUNDERING; FLEXIBLE MATERIALS NOT OTHERWISE PROVIDED FOR
- D06L—DRY-CLEANING, WASHING OR BLEACHING FIBRES, FILAMENTS, THREADS, YARNS, FABRICS, FEATHERS OR MADE-UP FIBROUS GOODS; BLEACHING LEATHER OR FURS
- D06L4/00—Bleaching fibres, filaments, threads, yarns, fabrics, feathers or made-up fibrous goods; Bleaching leather or furs
-
- D—TEXTILES; PAPER
- D06—TREATMENT OF TEXTILES OR THE LIKE; LAUNDERING; FLEXIBLE MATERIALS NOT OTHERWISE PROVIDED FOR
- D06L—DRY-CLEANING, WASHING OR BLEACHING FIBRES, FILAMENTS, THREADS, YARNS, FABRICS, FEATHERS OR MADE-UP FIBROUS GOODS; BLEACHING LEATHER OR FURS
- D06L4/00—Bleaching fibres, filaments, threads, yarns, fabrics, feathers or made-up fibrous goods; Bleaching leather or furs
- D06L4/10—Bleaching fibres, filaments, threads, yarns, fabrics, feathers or made-up fibrous goods; Bleaching leather or furs using agents which develop oxygen
- D06L4/12—Bleaching fibres, filaments, threads, yarns, fabrics, feathers or made-up fibrous goods; Bleaching leather or furs using agents which develop oxygen combined with specific additives
-
- D—TEXTILES; PAPER
- D06—TREATMENT OF TEXTILES OR THE LIKE; LAUNDERING; FLEXIBLE MATERIALS NOT OTHERWISE PROVIDED FOR
- D06P—DYEING OR PRINTING TEXTILES; DYEING LEATHER, FURS OR SOLID MACROMOLECULAR SUBSTANCES IN ANY FORM
- D06P1/00—General processes of dyeing or printing textiles, or general processes of dyeing leather, furs, or solid macromolecular substances in any form, classified according to the dyes, pigments, or auxiliary substances employed
- D06P1/44—General processes of dyeing or printing textiles, or general processes of dyeing leather, furs, or solid macromolecular substances in any form, classified according to the dyes, pigments, or auxiliary substances employed using insoluble pigments or auxiliary substances, e.g. binders
- D06P1/46—General processes of dyeing or printing textiles, or general processes of dyeing leather, furs, or solid macromolecular substances in any form, classified according to the dyes, pigments, or auxiliary substances employed using insoluble pigments or auxiliary substances, e.g. binders using compositions containing natural macromolecular substances or derivatives thereof
-
- D—TEXTILES; PAPER
- D06—TREATMENT OF TEXTILES OR THE LIKE; LAUNDERING; FLEXIBLE MATERIALS NOT OTHERWISE PROVIDED FOR
- D06M—TREATMENT, NOT PROVIDED FOR ELSEWHERE IN CLASS D06, OF FIBRES, THREADS, YARNS, FABRICS, FEATHERS OR FIBROUS GOODS MADE FROM SUCH MATERIALS
- D06M2101/00—Chemical constitution of the fibres, threads, yarns, fabrics or fibrous goods made from such materials, to be treated
- D06M2101/02—Natural fibres, other than mineral fibres
- D06M2101/10—Animal fibres
Definitions
- the present invention relates to a method for manufacturing processed fibers bleached and/or dyed, the processed fiber, a method for suppressing damage to animal fibers, and a method for processing animal fibers.
- the texture and hue of a fiber product made of animal fibers are important factors in determining the product value.
- the texture of animal fibers is expressed as "tender”, “smooth”, “frictional feel”, or the like, and it is thought that animal fibers have the most excellent texture when these fibers are collected and no chemical treatment other than scouring is performed.
- dyeing is performed in accordance with purposes, and when a vivid hue such as white or extremely light color to light color is required, it is necessary to bleach fibers prior to dyeing.
- oxidative bleaching by hydrogen peroxide or reductive bleaching by hydrosulfite or sodium bisulfite has been performed.
- these types of bleaching require long-term heating and an alkali agent, and thus fibers are damaged, causing deterioration of unique textures such as tenderness and frictional feel of animal fibers.
- Patent Literature 1 a bleaching sub-agent containing a polycarboxylic acid-based polymer and an amine-based compound, and a treating method by means of an alkaline protease, hydroxyalkyl phosphine, and derivatives thereof (Patent Literature 2) have been proposed.
- Patent Literature 2 a bleaching sub-agent containing a polycarboxylic acid-based polymer and an amine-based compound
- Patent Literature 2 a treating method by means of an alkaline protease, hydroxyalkyl phosphine, and derivatives thereof
- Patent Literature 3 a method applying an ultraviolet absorbing agent
- Patent Literature 4 a method applying a lower phosphorous acid and a lower phosphate to animal fibers
- Patent Literature 5 a method applying a fluorescent whitening agent to animal fibers
- their effects are not necessarily sufficient, and the textures are harmed, and their safety with regard to the human body may be also a concern.
- Patent Literatures 6 to 8 Conventionly, methods for providing fibers with the properties of an animal-derived protein by combining the animal-derived protein and animal fibers have been proposed (Patent Literatures 6 to 8). For example, it has been reported that use of sericin together with a dichlorotriazine-based compound provides fibers with durability and shape stability (Patent Literature 6); that in shape fixing, cloth made of animal protein fibers is soaked for absorption into a solution of a collagen protein derivative and a fibroin protein or a keratin solution, and thereby, the texture and touch feel are maintained (Patent Literature 7); that the physical properties of a protein fiber product is improved and the color of the protein fiber product is deepened by soaking the protein fiber product in an animal hair protein aqueous solution and a bridging agent (Patent Literature 8); and the like.
- a keratin hydrolytic product has the effect of suppressing deterioration of the texture and the light resistance of animal fibers caused by bleaching and/or dyeing.
- the present invention relates to providing a method for manufacturing processed fibers, the processed fibers, a method for suppressing damage to animal fibers, and a method for processing animal fibers, where a unique texture and light resistance of animal fibers are kept and a desired hue is realized even if the animal fibers are bleached and/or dyed.
- the inventors of the present invention found that when soaking animal fibers in a hydrolyzed keratin solution in any step for bleaching and/or dyeing the animal fibers, processed fibers with a desired hue are manufactured, while damage to the processed fibers is suppressed and while the original texture and light resistance of the animal fibers are kept.
- the present invention has the following aspects 1) to 10).
- the method for manufacturing processed fibers, the method for suppressing damage to animal fibers, and the method for processing animal fibers, in accordance with the present invention fiber damage occurring when bleaching and/or dyeing animal fibers is suppressed or repaired, and processed fibers where strength and light resistance are kept without impairing a unique texture of the animal fibers are manufactured. Also, because a hydrolytic product obtained by hydrolyzing naturally derived keratin protein is used, the method according to the present invention is implemented substantially safely to the human body, the environment, and the like.
- a method for manufacturing processed fibers of the present invention is one of the methods for manufacturing processed fibers with bleaching and dyeing animal fibers and has the step for soaking the fibers in a hydrolyzed keratin solution.
- animal fibers are those animal fibers used in spinning, and the form thereof may be short fibers, long fibers, spun yarn, knitted fabric, woven fabric, felt, or the like, and may be apparel products such as clothes, bedding, cushions, and stuffed toys.
- examples of the animal fibers include sheep wool, cashmere, mohair, camel hair, llama, alpaca, vicuna, angora, mink, and silk. These animal fibers may be used alone or in combination.
- the processed fibers of the present invention are obtained with bleaching and/or dyeing the above-described animal fibers, and thus, the processed fibers encompass bleached fibers, dyed fibers, and bleached and dyed fibers.
- bleaching is performed in order to increase the whiteness of animal fibers and is performed with an aqueous solution of hydrogen peroxide and a surface active agent, an aqueous solution of hydrogen peroxide and sodium silicate, or the like.
- dyeing is performed without particular limitation with a liquid flow method, a jigger method, a beam method, a cold pad-batch method, a pad steam method, a pad roll method, a continuous method, or the like, and a suitable dye and a suitable dyeing method may be selected in accordance with the type of animal fibers.
- the hydrolyzed keratins may be a hydrolytic product obtained by decomposing a keratin-containing raw material or a keratin extracted from the raw material under a reduction condition or the like by acid, alkali, peroxide, an enzyme, or the like, and the methods are all known (for example, see JP 2005-247692A , JP 2006-124341A , JP 2008-247925A , and JP H6-116300A ). Among them, hydrolytic products with an alkali or a peroxide are preferable.
- the number average molecular weight of this hydrolyzed keratin is preferably 10,000 or less, more preferably 8,000 or less, more preferably 5,000 or less, more preferably 3,000 or less, and more preferably 2,000 or less, and is preferably 130 or more, more preferably 150 or more, more preferably 200 or more, and more preferably 300 or more. Also, the number average molecular weight thereof is preferably 10,000 to 130, more preferably 8,000 to 130, more preferably 5,000 to 130, more preferably 3,000 to 150, more preferably 2,000 to 200, and more preferably 2,000 to 300.
- the keratin-containing raw material may be directly decomposed.
- an alkali metal hydroxide such as sodium hydroxide or potassium hydroxide, an alkaline earth metal hydroxides such as calcium hydroxide, or an alkaline compound such as ammonia is used, and in general, it is preferable to process the keratin-containing raw material at a concentration of 0.1 to 0.8 mol/L, preferably at a concentration of 0.2 to 0.5 mol/L, in general, at 20 to 120°C, and preferably for 0.1 to 72 hours.
- hydrolysis by a peroxide is performed by a peroxide such as hydrogen peroxide, performic acid, and peracetic acid, and in general, it is preferable to process the keratin-containing raw material at a concentration of 1 to 10%, preferably at a concentration of 3 to 8%, in general, at room temperature to 100°C, and preferably for 30 minutes to 48 hours.
- a peroxide such as hydrogen peroxide, performic acid, and peracetic acid
- this hydrolytic product may be cationized, silylated, acylated, or alkyl cationized so as to form derivatives of the hydrolyzed keratin, and these derivatives may also be used as long as the effects of the present invention are obtained.
- Examples of the keratin-containing raw materials include hairs of animals such as birds, sheep, horses, pigs, alpacas, mohair, angora, and cashmere, and among these, feathers obtained from birds or sheep wool is preferable, and it is particularly preferable to use feathers.
- any feathers such as down, feathers, and small feathers obtained from land birds such as chickens, quails, and turkeys, and water birds such as geese, call ducks, domestic ducks, European ducks, Peking ducks, and eider ducks, in particular, feathers of water birds are preferable.
- examples of preferred hydrolyzed keratins include hydrolyzed keratins derived from sheep wool or feathers, hydrolyzed keratins derived from feathers are more preferable, and alkaline hydrolyzed keratins or oxidatively hydrolyzed keratins derived from feathers are even more preferable.
- hydrolyzed keratin solutions examples include aqueous and ethanol solutions, and aqueous solutions are preferable.
- lipid components such as a chelating agent, a metal salt, ceramide, and fatty acid esters, an organic acid such as citric acid or ascorbic acid, a nonionic surface active agent, a cationic surface active agent, an amphoteric surface active agent, higher alcohols, lower alcohols, animal and vegetable oils, silicone oils, natural polysaccharides, an animal or plant extract, a hydrolytic product derived from animals or plants and derivatives thereof, a pH regulator, an antiseptic agent, and the like, in a range of not impairing the effects of the keratin.
- lipid components such as a chelating agent, a metal salt, ceramide, and fatty acid esters, an organic acid such as citric acid or ascorbic acid, a nonionic surface active agent, a cationic surface active agent, an amphoteric surface active agent, higher alcohols, lower alcohols, animal and vegetable oils, silicone oils, natural polysaccharides, an animal or plant extract, a hydrolytic product derived
- the concentration of the hydrolyzed keratin in the hydrolyzed keratin solution is preferably 0.01 mass% or more, more preferably 0.03 mass% or more, more preferably 0.05 mass% or more, and more preferably 0.1 mass% or more, and preferably 1 mass% or less, more preferably 0.7 mass% or less, and more preferably 0.5 mass% or less. Also, the concentration thereof is preferably 0.01 to 1 mass%, more preferably 0.03 to 0.7 mass%, more preferably 0.05 to 0.7 mass% or more, and more preferably 0.1 to 0.5 mass%.
- the animal fibers are preferably soaked in the hydrolyzed keratin solution, in general, at 30 to 80°C for 20 to 60 minutes, and are more preferably soaked at 40 to 60°C for 30 to 60 minutes.
- the animal fibers are soaked in the hydrolyzed keratin solution in a pretreatment process before a bleaching process and/or a dyeing process, or in a post-treatment process after a bleaching process and/or a dyeing process, and it is particularly preferable to implement both the pretreatment process and the post-treatment process.
- a pretreatment process before a bleaching process and/or a dyeing process
- a post-treatment process after a bleaching process and/or a dyeing process
- it is particularly preferable to implement both the pretreatment process and the post-treatment process For example, when dyeing is performed subsequent to bleaching, it is preferable to perform the soaking process twice, before the bleaching and after the dyeing, or three times before the bleaching after the bleachings (before the dyeing), and after the dyeing.
- the processed fibers of the present invention obtained thus, fiber damage due to bleaching and/or dyeing is suppressed or repaired. Namely, in the processed fibers of the present invention, the unique texture (for example, the touch feel) of animal fibers is kept in spite of the bleaching and/or dyeing, and the strength and the light resistance are kept without being reduced.
- the method including the step for soaking these fibers in the hydrolyzed keratin solution may serve as a method for suppressing damage to the fibers in bleaching and/or dyeing.
- the method, including the step for soaking the animal fibers in the hydrolyzed keratin solution, before or after, or before and after the fibers are bleached and/or dyed may serve as a processing method for suppressing the damage to the fibers.
- the present method for suppressing damage to animal fibers and the present method for processing animal fibers are useful for protecting the animal fibers from bleaching and/or dyeing and for keeping the texture of the fibers, or for keeping the light resistance.
- Texture refers to the material feel felt when a human touches the material, such as the touch feel, skin feel, or comfortability of feel
- unique texture of the animal fibers refers to the "tenderness”, “smoothness”, “frictional feel”, “suppleness”, “softness”, “sturdiness”, “bulkiness”, or the like.
- light resistance refers to resistance against deterioration caused by light (yellowing or decoloring of fibers).
- the dyeing (light resistant) fastness of fibers was measured in conformity with JIS L 0842, the third exposure method. The higher the grade obtained through this test is, the higher the fastness is.
- Fiber damage testing method Alkaline solubility method
- the alkaline solubility of fibers was measured in conformity with JIS L 1081, alkaline solubility method. The smaller the value obtained in this test is, the less damaged the fibers are.
- Fiber damage testing method Coloring method (methylene blue method)
- the fiber damage was determined in conformity with JIS L 1081, coloring (methylene blue method).
- the damage level is determined by means of a color concentration because undamaged fibers turn light blue and damaged fibers turn deep blue.
- ATR-FTIR spectra of fibers were recorded with a PerkinElmer Spectrum One FTIR spectrophotometer, manufactured by PerkinElmer, Inc., provided in Universal ATR Sampling Accessory. The measurements were performed under the condition that the accumulation number was 16 and the resolution was 4 cm -1 , and 4000 to 400 cm -1 .
- a peak for sulfonic acid was obtained at 1040 cm -1 .
- This sulfonic acid was derived from cystine in the fibers, and the cystine underwent cleavage due to excessive oxidation of cystine and changed into sulfonic acid.
- the lower the peak at 1040 cm -1 is, the lower the content of sulfonic acid is; indicating that excessive oxidation of fibers in the bleaching process has been suppressed.
- Panelist evaluation was performed on specimens. 25 panelists who had many opportunities to evaluate cashmere fibers in business touched specimens freely and evaluated the touch feel with the following criteria, and average values were obtained.
- SDS-PAGE electrophoresis was observed using a ready-made gel (e-PAGEL manufactured by ATTO CORPORATION). Dyeing was performed with Coomassie brilliant blue, and decoloring was performed with 10% acetic acid.
- Production Example 1 Production of sheep wool-derived solubilized (non hydrolytic) keratin
- Production Example 2 Production of feather-derived solubilized (non-hydrolytic) keratin
- the sheep wool was changed to feathers, and feather-derived solubilized keratin, not hydrolyzed, was produced similarly to Production Example 1.
- Production Example 3 Production of sheep wool-derived oxidatively hydrolyzed keratin
- Production Example 4 Production of feather-derived oxidatively hydrolyzed keratin
- the sheep wool was changed to feathers, and feather-derived oxidatively hydrolyzed keratin was produced similarly to Production Example 3.
- Production Example 5 Production of sheep wool-derived alkaline hydrolyzed keratin
- the sheep wool was changed to feathers, and feather-derived alkaline hydrolyzed keratin was produced similarly to Production Example 5,
- FIG. 1 shows the results of molecular weight analysis with SDS-PAGE for keratins produced in Production Examples 1 to 6.
- the feather and sheep wool-derived solubilized keratins With the feather and sheep wool-derived solubilized keratins, bands with a molecular weight of 10,000 or more were confirmed, whereas with the feather and sheep wool-derived hydrolyzed keratins, only bands with a molecular weight of 10,000 or less were confirmed.
- the feather and sheep wool-derived solubilized keratins are found to contain keratin molecules with molecular weight of 10,000 or more, and the feather and sheep wool-derived hydrolyzed keratins contain only keratin molecules with a molecular weight of 10,000 or less.
- Pretreatment was performed by soaking cashmere hairs in an aqueous solution containing 0.1% feather-derived oxidatively hydrolyzed keratin produced in Production Example 4 at 40°C for 60 minutes. Thereafter, the hairs were soaked in a diluted solution of 35% hydrogen peroxide water with the concentration of 20 cc/L at a liquid ratio of 1: 20, and were bleached at 60°C for 1 hour. After bleaching, the hairs were soaked in a 0.1% hydrolyzed keratin solution, at a liquid ratio of 1: 20 at 40°C for 60 minutes. The hairs were sufficiently washed with water and then dried, and thus feather-derived oxidatively hydrolyzed keratin treated hairs were produced.
- Feather-derived alkaline hydrolyzed keratin treated hairs were produced similarly to Embodiment 1, where the feather-derived oxidatively hydrolyzed keratin was changed to the feather-derived alkaline hydrolyzed keratin produced in Production Example 6.
- Sheep wool-derived oxidatively hydrolyzed keratin treated hairs were produced similarly to Embodiment 1, while the feather-derived oxidatively hydrolyzed keratin was changed to the sheep wool-derived oxidatively hydrolyzed keratin produced in Production Example 3.
- Sheep wool-derived alkaline hydrolyzed keratin treated hairs were produced similarly to Embodiment 1, while the feather-derived oxidatively hydrolyzed keratin was changed to the sheep wool-derived alkaline hydrolyzed keratin produced in Production Example 5.
- Sheep wool-derived solubilized keratin treated hairs were produced similarly to Embodiment 1, while the feather-derived oxidatively hydrolyzed keratin was changed to the sheep wool-derived solubilized keratin produced in Production Example 1.
- Feather-derived solubilized keratin treated hairs were produced similarly to Embodiment 1, while the feather-derived oxidatively hydrolyzed keratin was changed to the feather-derived solubilized keratin produced in Production Example 2.
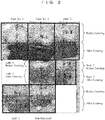
- FIG. 2 shows photographs of cotton-like fibers of Embodiments 1 to 4, Comparative Examples 1, 2, and unprocessed cashmere fibers, before and after the methylene blue dyeing.
- the fiber damage is evaluated on color concentrations because undamaged fibers turn light blue and damaged fibers turn deep blue.
- Comparative Examples 1, 2 were more deeply colored than unprocessed fibers.
- Embodiments 1 to 4 were more light in the color than Comparative Examples 1, 2, and in particular, Embodiments 1, 2 were equivalent to the unprocessed fibers in the color.
- Embodiments 1 to 4 suppressed, also according to the methylene blue dyeing method, fiber damage caused by bleaching, as is similar to the alkaline solubility method.
- the hydrolyzed keratins with an average molecular weight of 10,000 or less are found to effectively suppress fiber damage caused by bleaching.
- Embodiments 5 to 7 and Comparative Example 3 Keratin treatment before bleaching
- Pretreatment was performed by soaking cashmere hairs whose place of origin was different from those used in Embodiment 1 in a 0.5% feather-derived oxidatively hydrolyzed keratin solution, at a liquid ratio of 1: 20, at 40°C for 30 minutes. Thereafter, the hairs were soaked in a diluted solution obtained of 35% hydrogen peroxide water with the concentration 20 cc/L, at a liquid ratio of 1: 20, and were bleached at 60°C for 1 hour. The hairs were sufficiently washed with water and then dried to produce hydrolyzed keratin treated hairs.
- Hydrolyzed keratin treated hairs were produced similarly to Embodiment 5, while the hairs were soaked in a feather-derived oxidatively hydrolyzed keratin solution, at 60 °C.
- Feather-derived alkaline hydrolyzed keratin treated hairs were produced similarly to Embodiment 5, while the hairs were soaked in a feather-derived oxidatively hydrolyzed keratin solution, at 80 °C.
- Cashmere hairs similar to those used in Embodiment 5 were soaked in a diluted solution of 35% hydrogen peroxide water in the concentration of 20 cc/L of, at a liquid ratio of 1: 20, and were bleached at 60°C for 1 hour. The hairs were sufficiently washed with water and then dried; thus keratin untreated hairs were produced.
- Embodiments 5 to 7 shows ATR-FTIR spectra of unprocessed hairs, Embodiments 5 to 7, and Comparative Example 3.
- the unprocessed hairs showed no peaks for sulfonic acid at 1040 cm -1
- Embodiments 5 to 7 and Comparative Example 3 showed peaks for sulfonic acid at 1040 cm -1 , and thus, it was suggested that excessive oxidation caused by bleaching occurred.
- Embodiments 5 to 7 had weaker peak intensities at 1040 cm -1 , and thus, it was suggested that excessive oxidation caused by bleaching was suppressed.
- Bleached cashmere hairs were soaked in a 0.1% feather-derived oxidatively hydrolyzed keratin solution, at a liquid ratio of 1: 20, at 40°C for 60 minutes. After the hairs were sufficiently washed with water, they were dried, and hydrolyzed keratin treated hairs were produced.
- Embodiment 8 The fiber damages and light resistant fastnesses of Embodiment 8 and the bleached cashmere hairs are listed in Table 4.
- the fiber damage was smaller than the bleached cashmere hairs, suggesting the recovery from the fiber damage.
- the light resistant fastness increased, suggesting the increased light resistance.
- Table 4 Keratin Treatment Fiber Damage Light Resistant Fastness Processing Agent Temperatur e When Emb. 8 Feather-Derived Oxidatively Hydrolyzed Keratin 40°C After Bleaching 62.6 4 or mor e Un-processed - - - 71.2 3 to 4 *unprocessed: bleached cashmere hairs without keratin treatment
- Bleached and dyed cashmere hairs were soaked in a 0.5% feather-derived oxidatively hydrolyzed keratin solution, at a liquid ratio of 1: 20, at 60°C, for 20 minutes.
- the hairs were sufficiently washed with water and then dried, and thus, hydrolyzed keratin treated hairs were produced.
- Hydrolyzed keratin treated hairs were produced similarly to Embodiment 7, while the hairs were soaked in a feather-derived oxidatively hydrolyzed keratin solution, at 80 °C.
- Embodiments 9 and 10 The light resistant fastness of Embodiments 9 and 10 and the bleached and dyed cashmere hairs are listed in Table 5. Embodiments 9 and 10 showed increased light resistant fastness.
- Table 5 Keratin Treatment Light Resistant Fastness Processed Temperature When Emb. 9 Feather-Derived Oxidatively Hydrolyzed Keratin 60°C After Bleaching and Dyeing 3 to 4 Emb. 10 80°C After Bleaching and Dyeing 3 to 4 Unprocessed - - - 3 *unprocessed: bleached and dyed cashmere hairs that did not undergo keratin treatment
- Pretreatment was performed by soaking cashmere hairs used in Embodiment 5, in a 0.1% feather-derived oxidatively hydrolyzed keratin solution, at a liquid ratio of 1: 20, at 40°C, for 60 minutes. Thereafter, the hairs were soaked in a diluted solution of 35% hydrogen peroxide water having a concentration of 20 cc/L, at a liquid ratio of 1: 20, and were bleached at 60°C for 1 hour. After bleaching, the post-treatment was performed by soaking the hairs in a 0.1% feather-derived oxidatively hydrolyzed keratin solution, at a liquid ratio of 1: 20, at 40°C, for 60 minutes. The hairs were sufficiently washed with water and then dried, and thus hydrolyzed keratin pre/post-treated hairs. were produced.
- Hydrolyzed keratin treated hairs were produced similarly to Embodiment 9, while the hairs were soaked in a feather-derived oxidatively hydrolyzed keratin solution, at 30 °C.
- FIG. 4 shows ATR-FTIR spectra of unprocessed hairs, Embodiments 11 and 12, and Comparative Example 3.
- the unprocessed hairs showed no peaks for sulfonic acid at 1040 cm -1
- Embodiments 11 and 12 and Comparative Example 3 showed peaks for sulfonic acid at 1040 cm -1 , and thus, excessive oxidation seems caused by bleaching.
- Embodiments 11 and 12 had weaker peak intensities at 1040 cm -1 , and thus, excessive oxidation caused by bleaching seems suppressed.
- Pretreatment was performed by soaking cashmere hairs used in Embodiment 5, in a 0.1% feather-derived oxidatively hydrolyzed keratin solution, at a liquid ratio of 1: 20, at 80°C, for 30 minutes. Thereafter, the hairs were soaked in a diluted solution of 35% hydrogen peroxide water with the concentration of 20 cc/L, at a liquid ratio of 1: 20, and were bleached at 60°C for 1 hour. After bleaching, dyeing was performed by a 0.03% acidic dye.
- Hydrolyzed keratin treated hairs were produced similarly to Embodiment 13, while the hairs were soaked in a 0.5% feather-derived oxidatively hydrolyzed keratin solution.
- Cashmere hairs similar to Embodiment 5 were soaked in a diluted solution of 35% hydrogen peroxide water with the concentration of 20 cc/L, at a liquid ratio of 1: 20, and were bleached at 60°C for 1 hour. After bleaching, dyeing was performed by a 0.03% acidic dye. The hairs were sufficiently washed with water and then dried, and thus, keratin untreated hairs were produced.
- Embodiments 13 and 14 and Comparative Example 4 are listed in Table 7. Compared with Comparative Example 4, in Embodiments 13 and 14, the fiber damages were small, revealing the suppression of the fiber damage. Also, the light resistant fastness increased, revealing the increase in the light resistance.
- Table 7 Keratin Treatment Fiber Damage Light Resistance Fastness Processing Agent Temperature When Emb. 13 Feather Oxidatively Hydrolyzed Keratin 80°C Before and After Bleaching and Dyeing 22.6 3 to 4 Emb. 14 80°C Before and After Bleaching and Dyeing 26.6 4 or more Com. Ex. 4 - - Before and After Bleaching and Dyeing 29.3 3
- Pretreatment was performed by soaking spun sheep wool yarn in a 0.1% feather-derived oxidatively hydrolyzed keratin solution, at a liquid ratio of 1: 20, at 40°C. for 60 minutes. Thereafter, the hairs were soaked in a diluted solution of 35% hydrogen peroxide water (20 cc/L solution), at a liquid ratio of 1: 20, and were bleached at 60°C for 1 hour. After bleaching, post-treatment was performed by soaking the hairs in a 0.1% feather-derived oxidatively hydrolyzed keratin solution, at a liquid ratio of 1: 20, at 40°C, for 60 minutes. The hairs were sufficiently washed with water and then dried, and thus, hydrolyzed keratin treated spun yarn was produced.
- the spun sheep wool yarn was soaked in a diluted solution of 35% hydrogen peroxide water (20 cc/L solution), at a liquid ratio of 1: 20, and was bleached at 60°C for 1 hour.
- the yarn was sufficiently washed with water and then dried, and thus bleached sheep wool spun yarn was produced.
- Hydrolyzed keratin treated cashmere spun yarn was produced similarly to Embodiment 15, while the sheep wool yarn was changed to cashmere yarn.
- Hydrolyzed keratin treated silk spun yarn was produced similarly to Embodiment 15, while the sheep wool yarn was changed to silk yarn.
- Hydrolyzed keratin treated alpaca spun yarn was produced similarly to Embodiment 15, while the sheep wool yarn was changed to alpaca yarn.
- Hydrolyzed keratin treated mixed spun yarn was produced similarly to Embodiment 15, while the sheep wool yarn was changed to mixed yarn of 80% mohair and 20% sheep wool.
- Hydrolyzed keratin treated angora spun yarn was produced similarly to Embodiment 15, while the sheep wool yarn was changed to angora yarn.
- Embodiments 16 to 20 Animal hairs similar to Embodiments 16 to 20 were soaked in a diluted solution of 35% hydrogen peroxide water(20 cc/L solution), at a liquid ratio of 1: 20, and were bleached at 60°C for 1 hour. The hairs were sufficiently washed with water and then dried, and thus keratin untreated spun yarn was produced.
- Pretreatment was performed by soaking cashmere hairs in a 0.1% aqueous solution containing commercially available hydrolyzed keratin (average molecular weight of 1,000), at a temperature of 40°C, for 60 minutes. Thereafter, the hairs were soaked in a diluted solution of 35% hydrogen peroxide water (20 cc/L solution) at a liquid ratio of 1: 20, and were bleached at 60°C for 1 hour. After bleaching, the hairs were soaked in a 0.1% hydrolyzed keratin solution, at a liquid ratio of 1: 20, at 40°C for 60 minutes. The hairs were sufficiently washed with water and then dried, and thus hydrolyzed keratin treated hairs were produced.
- Hydrolyzed collagen treated hairs were produced similarly to Embodiment 21, while the hydrolyzed keratin was changed to a commercially available hydrolyzed collagen (average molecular weight of 1,000).
- Hydrolyzed silk treated hairs were produced similarly to Embodiment 21, while the hydrolyzed keratin was changed to commercially available hydrolyzed silk (average molecular weight of 1,000).
- FIG. 5 shows photographs of cotton-like fibers, before and after methylene blue dyeing on Embodiment 21, Comparative Examples 11 and 12.
- the fiber damage is evaluated with a color concentration because undamaged fibers turn light blue and damaged fibers turn deep blue.
- Comparative Examples 11 and 12 were more deeply colored than unprocessed fibers.
- Embodiment 21 was more lightly colored than Comparative Examples 11 and 12. This suggests that with Embodiment 21, in the evaluation with the methylene blue dyeing method as well, fiber damage caused by bleaching was suppressed similarly to the alkaline solubility method.
- the hydrolyzed keratin, among hydrolyzed proteins effectively suppressed fiber damage.
Abstract
Description
- The present invention relates to a method for manufacturing processed fibers bleached and/or dyed, the processed fiber, a method for suppressing damage to animal fibers, and a method for processing animal fibers.
- The texture and hue of a fiber product made of animal fibers are important factors in determining the product value. The texture of animal fibers is expressed as "tender", "smooth", "frictional feel", or the like, and it is thought that animal fibers have the most excellent texture when these fibers are collected and no chemical treatment other than scouring is performed. Also, with regard to the hue, dyeing is performed in accordance with purposes, and when a vivid hue such as white or extremely light color to light color is required, it is necessary to bleach fibers prior to dyeing. Thus, conventionally, oxidative bleaching by hydrogen peroxide or reductive bleaching by hydrosulfite or sodium bisulfite has been performed. However, these types of bleaching require long-term heating and an alkali agent, and thus fibers are damaged, causing deterioration of unique textures such as tenderness and frictional feel of animal fibers.
- In view of this, attempts have been made to develop a bleaching method by which the texture does not deteriorate and sufficient whiteness is obtained. For example, a bleaching sub-agent (Patent Literature 1) containing a polycarboxylic acid-based polymer and an amine-based compound, and a treating method by means of an alkaline protease, hydroxyalkyl phosphine, and derivatives thereof (Patent Literature 2) have been proposed. However, it cannot be said that they sufficiently suppress deterioration of the texture and maintain sufficient light resistance.
- Also, a method applying an ultraviolet absorbing agent (Patent Literature 3), a method applying a lower phosphorous acid and a lower phosphate to animal fibers (Patent Literature 4), a method applying a fluorescent whitening agent to animal fibers (Patent Literature 5), and the like have been proposed for improving the light resistance of animal fibers. However, their effects are not necessarily sufficient, and the textures are harmed, and their safety with regard to the human body may be also a concern.
- On the other hand, conventionally, methods for providing fibers with the properties of an animal-derived protein by combining the animal-derived protein and animal fibers have been proposed (
Patent Literatures 6 to 8). For example, it has been reported that use of sericin together with a dichlorotriazine-based compound provides fibers with durability and shape stability (Patent Literature 6); that in shape fixing, cloth made of animal protein fibers is soaked for absorption into a solution of a collagen protein derivative and a fibroin protein or a keratin solution, and thereby, the texture and touch feel are maintained (Patent Literature 7); that the physical properties of a protein fiber product is improved and the color of the protein fiber product is deepened by soaking the protein fiber product in an animal hair protein aqueous solution and a bridging agent (Patent Literature 8); and the like. - However, it has not previously been known that a keratin hydrolytic product has the effect of suppressing deterioration of the texture and the light resistance of animal fibers caused by bleaching and/or dyeing.
-
- Patent Literature 1:
JP 2005-146442A - Patent Literature 2:
JP H11-172580A - Patent Literature 3:
JP H2-242970A - Patent Literature 4:
JP H5-156573A - Patent Literature 5:
JP 2001-518919A - Patent Literature 6:
JP2004-44055A - Patent Literature 7:
JP H1-280074A - Patent Literature 8:
JP H6-341058A - The present invention relates to providing a method for manufacturing processed fibers, the processed fibers, a method for suppressing damage to animal fibers, and a method for processing animal fibers, where a unique texture and light resistance of animal fibers are kept and a desired hue is realized even if the animal fibers are bleached and/or dyed.
- As a result of intensive studies in light of the above-described issues, the inventors of the present invention found that when soaking animal fibers in a hydrolyzed keratin solution in any step for bleaching and/or dyeing the animal fibers, processed fibers with a desired hue are manufactured, while damage to the processed fibers is suppressed and while the original texture and light resistance of the animal fibers are kept.
- The present invention has the following aspects 1) to 10).
- 1) A method for manufacturing processed fibers where animal fibers are bleached and/or dyed having the step for soaking the fibers in a hydrolyzed keratin solution.
- 2) The method according to 1), where the hydrolyzed keratin solution has a concentration of 0.01 to 1 mass%.
- 3) The method according to 1) or 2), where the step for soaking the fibers in the hydrolyzed keratin solution is repeatedly performed before and after bleaching and/or dyeing.
- 4) The method according to any of 1) to 3), where a hydrolyzed keratin is one derived from sheep wool or feather.
- 5) The method according to any of 1) to 4), where a hydrolyzed keratin has a number average molecular weight of 10,000 to 130.
- 6) The method according to any of 1) to 5), where the animal fibers are one or more selected from sheep wool, cashmere, mohair, camel hair, llama, alpaca, vicuna, angora, mink, and silk.
- 7) The method according to any of 1) to 6), where the animal fibers are spun yarn.
- 8) A method for suppressing damage to animal fibers, performing the step for soaking said fibers in a hydrolyzed keratin solution, when bleaching and/or dyeing said fibers.
- 9) A method for processing animal fibers, performing the step for soaking the fibers in a hydrolyzed keratin solution, before, after, or before and after bleaching and/or dyeing.
- 10) A processed fiber, manufactured according to one of the methods of 1) to 7).
- According to the method for manufacturing processed fibers, the method for suppressing damage to animal fibers, and the method for processing animal fibers, in accordance with the present invention, fiber damage occurring when bleaching and/or dyeing animal fibers is suppressed or repaired, and processed fibers where strength and light resistance are kept without impairing a unique texture of the animal fibers are manufactured. Also, because a hydrolytic product obtained by hydrolyzing naturally derived keratin protein is used, the method according to the present invention is implemented substantially safely to the human body, the environment, and the like.
-
-
FIG. 1 shows SDS-PAGE of a hydrolyzed keratin. -
FIG. 2 shows photographs of cotton-like fibers before and after methylene blue dyeing. -
FIG. 3 shows ATR-FTIR spectra ofEmbodiments 5 to 7 and Comparative Example 3. -
FIG. 4 shows ATR-FTIR spectra ofEmbodiments -
FIG. 5 shows photographs of cotton-like fibers before and after methylene blue dyeing. - A method for manufacturing processed fibers of the present invention is one of the methods for manufacturing processed fibers with bleaching and dyeing animal fibers and has the step for soaking the fibers in a hydrolyzed keratin solution.
- In the present invention, animal fibers are those animal fibers used in spinning, and the form thereof may be short fibers, long fibers, spun yarn, knitted fabric, woven fabric, felt, or the like, and may be apparel products such as clothes, bedding, cushions, and stuffed toys.
- Here, examples of the animal fibers include sheep wool, cashmere, mohair, camel hair, llama, alpaca, vicuna, angora, mink, and silk. These animal fibers may be used alone or in combination.
- The processed fibers of the present invention are obtained with bleaching and/or dyeing the above-described animal fibers, and thus, the processed fibers encompass bleached fibers, dyed fibers, and bleached and dyed fibers.
- In the present invention, bleaching is performed in order to increase the whiteness of animal fibers and is performed with an aqueous solution of hydrogen peroxide and a surface active agent, an aqueous solution of hydrogen peroxide and sodium silicate, or the like. Also, dyeing is performed without particular limitation with a liquid flow method, a jigger method, a beam method, a cold pad-batch method, a pad steam method, a pad roll method, a continuous method, or the like, and a suitable dye and a suitable dyeing method may be selected in accordance with the type of animal fibers.
- In the present invention, the hydrolyzed keratins may be a hydrolytic product obtained by decomposing a keratin-containing raw material or a keratin extracted from the raw material under a reduction condition or the like by acid, alkali, peroxide, an enzyme, or the like, and the methods are all known (for example, see
JP 2005-247692A JP 2006-124341A JP 2008-247925A JP H6-116300A - Considering the sorption and permeability of the hydrolyzed keratin to fibers, the number average molecular weight of this hydrolyzed keratin, measured through gel filtration analysis, is preferably 10,000 or less, more preferably 8,000 or less, more preferably 5,000 or less, more preferably 3,000 or less, and more preferably 2,000 or less, and is preferably 130 or more, more preferably 150 or more, more preferably 200 or more, and more preferably 300 or more. Also, the number average molecular weight thereof is preferably 10,000 to 130, more preferably 8,000 to 130, more preferably 5,000 to 130, more preferably 3,000 to 150, more preferably 2,000 to 200, and more preferably 2,000 to 300.
- In hydrolysis by an alkali or peroxide, the keratin-containing raw material may be directly decomposed.
- In alkaline hydrolysis, an alkali metal hydroxide such as sodium hydroxide or potassium hydroxide, an alkaline earth metal hydroxides such as calcium hydroxide, or an alkaline compound such as ammonia is used, and in general, it is preferable to process the keratin-containing raw material at a concentration of 0.1 to 0.8 mol/L, preferably at a concentration of 0.2 to 0.5 mol/L, in general, at 20 to 120°C, and preferably for 0.1 to 72 hours.
- Also, hydrolysis by a peroxide (oxidative hydrolysis) is performed by a peroxide such as hydrogen peroxide, performic acid, and peracetic acid, and in general, it is preferable to process the keratin-containing raw material at a concentration of 1 to 10%, preferably at a concentration of 3 to 8%, in general, at room temperature to 100°C, and preferably for 30 minutes to 48 hours.
- Also, this hydrolytic product may be cationized, silylated, acylated, or alkyl cationized so as to form derivatives of the hydrolyzed keratin, and these derivatives may also be used as long as the effects of the present invention are obtained.
- Examples of the keratin-containing raw materials include hairs of animals such as birds, sheep, horses, pigs, alpacas, mohair, angora, and cashmere, and among these, feathers obtained from birds or sheep wool is preferable, and it is particularly preferable to use feathers. Although it is possible to use any feathers such as down, feathers, and small feathers obtained from land birds such as chickens, quails, and turkeys, and water birds such as geese, call ducks, domestic ducks, European ducks, Peking ducks, and eider ducks, in particular, feathers of water birds are preferable.
- In the present invention, examples of preferred hydrolyzed keratins include hydrolyzed keratins derived from sheep wool or feathers, hydrolyzed keratins derived from feathers are more preferable, and alkaline hydrolyzed keratins or oxidatively hydrolyzed keratins derived from feathers are even more preferable.
- Examples of hydrolyzed keratin solutions include aqueous and ethanol solutions, and aqueous solutions are preferable.
- It is possible to blend, as appropriate, into this hydrolyzed keratin solution, lipid components such as a chelating agent, a metal salt, ceramide, and fatty acid esters, an organic acid such as citric acid or ascorbic acid, a nonionic surface active agent, a cationic surface active agent, an amphoteric surface active agent, higher alcohols, lower alcohols, animal and vegetable oils, silicone oils, natural polysaccharides, an animal or plant extract, a hydrolytic product derived from animals or plants and derivatives thereof, a pH regulator, an antiseptic agent, and the like, in a range of not impairing the effects of the keratin.
- In view of increasing the animal fiber's protective function and recovering function, and suppressing deterioration of the texture caused by an increase in the sorption amount of the hydrolyzed keratin into fibers, the concentration of the hydrolyzed keratin in the hydrolyzed keratin solution is preferably 0.01 mass% or more, more preferably 0.03 mass% or more, more preferably 0.05 mass% or more, and more preferably 0.1 mass% or more, and preferably 1 mass% or less, more preferably 0.7 mass% or less, and more preferably 0.5 mass% or less. Also, the concentration thereof is preferably 0.01 to 1 mass%, more preferably 0.03 to 0.7 mass%, more preferably 0.05 to 0.7 mass% or more, and more preferably 0.1 to 0.5 mass%.
- From the viewpoint of suppressing deterioration of the protective function and the recovering function for animal fibers caused by denaturation by heat or a decrease in the osmotic force, the animal fibers are preferably soaked in the hydrolyzed keratin solution, in general, at 30 to 80°C for 20 to 60 minutes, and are more preferably soaked at 40 to 60°C for 30 to 60 minutes.
- The animal fibers are soaked in the hydrolyzed keratin solution in a pretreatment process before a bleaching process and/or a dyeing process, or in a post-treatment process after a bleaching process and/or a dyeing process, and it is particularly preferable to implement both the pretreatment process and the post-treatment process. For example, when dyeing is performed subsequent to bleaching, it is preferable to perform the soaking process twice, before the bleaching and after the dyeing, or three times before the bleaching after the bleachings (before the dyeing), and after the dyeing.
- In the processed fibers of the present invention obtained thus, fiber damage due to bleaching and/or dyeing is suppressed or repaired. Namely, in the processed fibers of the present invention, the unique texture (for example, the touch feel) of animal fibers is kept in spite of the bleaching and/or dyeing, and the strength and the light resistance are kept without being reduced.
- Therefore, when bleaching and/or dyeing animal fibers, the method including the step for soaking these fibers in the hydrolyzed keratin solution may serve as a method for suppressing damage to the fibers in bleaching and/or dyeing. Also, the method, including the step for soaking the animal fibers in the hydrolyzed keratin solution, before or after, or before and after the fibers are bleached and/or dyed, may serve as a processing method for suppressing the damage to the fibers.
- The present method for suppressing damage to animal fibers and the present method for processing animal fibers are useful for protecting the animal fibers from bleaching and/or dyeing and for keeping the texture of the fibers, or for keeping the light resistance.
- Note that in the present invention, "texture" refers to the material feel felt when a human touches the material, such as the touch feel, skin feel, or comfortability of feel, and the unique texture of the animal fibers refers to the "tenderness", "smoothness", "frictional feel", "suppleness", "softness", "sturdiness", "bulkiness", or the like.
- Also, "light resistance" refers to resistance against deterioration caused by light (yellowing or decoloring of fibers).
- Hereinafter, the present invention will be more specifically described according to embodiments. However, the present invention is not limited to these embodiments.
- The dyeing (light resistant) fastness of fibers was measured in conformity with JIS L 0842, the third exposure method. The higher the grade obtained through this test is, the higher the fastness is.
- The alkaline solubility of fibers was measured in conformity with JIS L 1081, alkaline solubility method. The smaller the value obtained in this test is, the less damaged the fibers are.
- The fiber damage was determined in conformity with JIS L 1081, coloring (methylene blue method). The damage level is determined by means of a color concentration because undamaged fibers turn light blue and damaged fibers turn deep blue.
- ATR-FTIR spectra of fibers were recorded with a PerkinElmer Spectrum One FTIR spectrophotometer, manufactured by PerkinElmer, Inc., provided in Universal ATR Sampling Accessory. The measurements were performed under the condition that the accumulation number was 16 and the resolution was 4 cm-1, and 4000 to 400 cm-1. In the obtained spectra, a peak for sulfonic acid was obtained at 1040 cm-1. This sulfonic acid was derived from cystine in the fibers, and the cystine underwent cleavage due to excessive oxidation of cystine and changed into sulfonic acid. Thus, the lower the peak at 1040 cm-1 is, the lower the content of sulfonic acid is; indicating that excessive oxidation of fibers in the bleaching process has been suppressed.
- Single yarn tensile strength and elongation ratio were measured in conformity with JIS L 1095, spun yarn testing method.
- Panelist evaluation was performed on specimens. 25 panelists who had many opportunities to evaluate cashmere fibers in business touched specimens freely and evaluated the touch feel with the following criteria, and average values were obtained.
-
- 5: Excellent
- 4: Good
- 3: Fair
- 2: Poor and
- 1: Bad.
- SDS-PAGE electrophoresis was observed using a ready-made gel (e-PAGEL manufactured by ATTO CORPORATION). Dyeing was performed with Coomassie brilliant blue, and decoloring was performed with 10% acetic acid.
- Column for gel filtration: AsahipakGF-510HQ and AsahipakGF-310HQ manufactured by SHOWA DENKO K.K.
- Moving phase: CH3CN/H2O(45/55) + 0.1% trifluoroacetic acid (TFA) Flow rate: 0.5 ml/min
- Column temperature: 40°C
- UV detection condition: 215 nm
- Standard sample: catalase (Mw 230,000), aldolase (Mw 158,000), cow serum albumin (Mw 68,000), ovalbumin (Mw 45,000), chymotrypsinogen A (Mw 25,000), cytochrome C (Mw 12,500), insulin (Mw 5,808), bacitracin (Mw 1,400), Gly-Gly-Gly-Gly-Gly-Gly (Mw 360.3), Gly-Pro-Ala (Mw 243), and Phe (Mw 165).
- 300 mL of an 8 M urea solution (pH: 9.5) containing 0.05 M tris and 0.1 M dithiothreitol was added to 10 g of sheep wool and stirred at 4°C for 24 hours. Thereafter, 7.5 g of sodium sulfite and 18 g of sodium tetrathionate dihydrate were added and stirred at 4°C for 24 hours. After undissolved substances were removed by centrifugal separation, the pH of the mixture was adjusted to 5.2 with hydrochloric acid, and sheep wool-derived solubilized, but not hydrolyzed, keratin was produced.
- The sheep wool was changed to feathers, and feather-derived solubilized keratin, not hydrolyzed, was produced similarly to Production Example 1.
- Soaking 10 g of sheep wool in hydrogen peroxide water for three hours in order to oxidize it. Thereafter, the sheep wool was hydrolyzed by adding ammonia. Unreacted hydrogen peroxide and undecomposed substances were removed, and sheep wool-derived oxidatively hydrolyzed keratin was produced.
- The sheep wool was changed to feathers, and feather-derived oxidatively hydrolyzed keratin was produced similarly to Production Example 3.
- 100 g of 1.3% sodium hydroxide solution was added to 10 g of sheep wool and reacted at 120°C for 20 minutes. After cooling to room temperature, the pH was lowered to by hydrochloric acid, and the product was left for one night. After undecomposed substances were removed by centrifugal separation, the pH of the mixture was adjusted to 5.6 with sodium hydroxide, and sheep wool-derived alkaline hydrolyzed keratin was produced.
- The sheep wool was changed to feathers, and feather-derived alkaline hydrolyzed keratin was produced similarly to Production Example 5,
-
FIG. 1 shows the results of molecular weight analysis with SDS-PAGE for keratins produced in Production Examples 1 to 6. With the feather and sheep wool-derived solubilized keratins, bands with a molecular weight of 10,000 or more were confirmed, whereas with the feather and sheep wool-derived hydrolyzed keratins, only bands with a molecular weight of 10,000 or less were confirmed. Based on this, the feather and sheep wool-derived solubilized keratins are found to contain keratin molecules with molecular weight of 10,000 or more, and the feather and sheep wool-derived hydrolyzed keratins contain only keratin molecules with a molecular weight of 10,000 or less. - Also, the results of molecular weight measurement through gel filtration molecular weight analysis for the keratins produced in Production Examples 3 to 6 are listed in Table 1. Those keratins are found to contain keratin molecules with number average molecular weight of about 1000, regardless of whether the keratin-containing raw material is derived from and the hydrolysis method.
[Table 1] Keratin-containing Raw Material Hydrolysis Method Average Molecular Weight Production Ex. 3 wool oxidation 725 Production Ex. 4 feather oxidation 927 Production Ex. 5 wool alkaline 1248 Production Ex. 6 feather alkaline 783 - Pretreatment was performed by soaking cashmere hairs in an aqueous solution containing 0.1% feather-derived oxidatively hydrolyzed keratin produced in Production Example 4 at 40°C for 60 minutes. Thereafter, the hairs were soaked in a diluted solution of 35% hydrogen peroxide water with the concentration of 20 cc/L at a liquid ratio of 1: 20, and were bleached at 60°C for 1 hour. After bleaching, the hairs were soaked in a 0.1% hydrolyzed keratin solution, at a liquid ratio of 1: 20 at 40°C for 60 minutes. The hairs were sufficiently washed with water and then dried, and thus feather-derived oxidatively hydrolyzed keratin treated hairs were produced.
- Feather-derived alkaline hydrolyzed keratin treated hairs were produced similarly to
Embodiment 1, where the feather-derived oxidatively hydrolyzed keratin was changed to the feather-derived alkaline hydrolyzed keratin produced in Production Example 6. - Sheep wool-derived oxidatively hydrolyzed keratin treated hairs were produced similarly to
Embodiment 1, while the feather-derived oxidatively hydrolyzed keratin was changed to the sheep wool-derived oxidatively hydrolyzed keratin produced in Production Example 3. - Sheep wool-derived alkaline hydrolyzed keratin treated hairs were produced similarly to
Embodiment 1, while the feather-derived oxidatively hydrolyzed keratin was changed to the sheep wool-derived alkaline hydrolyzed keratin produced in Production Example 5. - Sheep wool-derived solubilized keratin treated hairs were produced similarly to
Embodiment 1, while the feather-derived oxidatively hydrolyzed keratin was changed to the sheep wool-derived solubilized keratin produced in Production Example 1. - Feather-derived solubilized keratin treated hairs were produced similarly to
Embodiment 1, while the feather-derived oxidatively hydrolyzed keratin was changed to the feather-derived solubilized keratin produced in Production Example 2. - The results of the fiber damage test and average values of panelist evaluations (touch feels) on
Embodiments 1 to 4 and Comparative Examples 1, 2 are listed in Table 2. Compared with Comparative Examples 1, 2, where hydrolyzed keratin treatment was not performed, inEmbodiments 1 to 4, where the hydrolyzed keratin treatment was performed, the fiber damage caused by bleaching was suppressed and deterioration of the unique texture of cashmere fibers was suppressed.[Table 2] Keratin Treatment Fiber Damage Touch Feel Processing Agent When Emb. 1 Feather-Derived Oxidatively Hydrolyzed Keratin Before and After Bleaching 16.3 3.3±0.9 Emb. 2 Feather-Derived Alkaline Hydrolyzed Keratin Before and After Bleaching 15.7 3.2±1.0 Emb. 3 Wool-Derived Oxidatively Hydrolyzed keratin Before and Aft er Bleaching 16.0 2.9±0.7 Emb. 4 Wool-Derived Alkaline Hydrolyzed Keratin Before and After Bleaching 16.0 3.3±0.9 Com. Ex. 1 Wool-Derived Solubilized Keratin Before and Aft er Bleaching 17.3 2.5±0.8 Com. Ex. 2 Feather-Derived Solubilized Keratin Before and After Bleaching 16.4 2.7±0.9 Unprocessed - - 11.9 4.5±0.7 *unprocessed: cashmere hairs not bleached and without keratin treatment -
FIG. 2 shows photographs of cotton-like fibers ofEmbodiments 1 to 4, Comparative Examples 1, 2, and unprocessed cashmere fibers, before and after the methylene blue dyeing. The fiber damage is evaluated on color concentrations because undamaged fibers turn light blue and damaged fibers turn deep blue. As shown inFIG. 2 , Comparative Examples 1, 2 were more deeply colored than unprocessed fibers.Embodiments 1 to 4 were more light in the color than Comparative Examples 1, 2, and in particular,Embodiments Embodiments 1 to 4 suppressed, also according to the methylene blue dyeing method, fiber damage caused by bleaching, as is similar to the alkaline solubility method. - According to the results of fiber damage level and touch feel, the hydrolyzed keratins with an average molecular weight of 10,000 or less are found to effectively suppress fiber damage caused by bleaching.
-
Embodiments 5 to 7 and Comparative Example 3 (keratin treatment before bleaching) - Pretreatment was performed by soaking cashmere hairs whose place of origin was different from those used in
Embodiment 1 in a 0.5% feather-derived oxidatively hydrolyzed keratin solution, at a liquid ratio of 1: 20, at 40°C for 30 minutes. Thereafter, the hairs were soaked in a diluted solution obtained of 35% hydrogen peroxide water with the concentration 20 cc/L, at a liquid ratio of 1: 20, and were bleached at 60°C for 1 hour. The hairs were sufficiently washed with water and then dried to produce hydrolyzed keratin treated hairs. - Hydrolyzed keratin treated hairs were produced similarly to
Embodiment 5, while the hairs were soaked in a feather-derived oxidatively hydrolyzed keratin solution, at 60 °C. - Feather-derived alkaline hydrolyzed keratin treated hairs were produced similarly to
Embodiment 5, while the hairs were soaked in a feather-derived oxidatively hydrolyzed keratin solution, at 80 °C. - Cashmere hairs similar to those used in
Embodiment 5 were soaked in a diluted solution of 35% hydrogen peroxide water in the concentration of 20 cc/L of, at a liquid ratio of 1: 20, and were bleached at 60°C for 1 hour. The hairs were sufficiently washed with water and then dried; thus keratin untreated hairs were produced. -
1) The results of fiber damage test on unprocessed hairs,Embodiments 5 to 7, and Comparative Example 3 are listed in Table 3. Compared with Comparative Example 3, inEmbodiments 5 to 7, the fiber damage was smaller, revealing the suppression of the fiber damage caused by bleaching.[Table 3] Keratin Treatment Fiber Damage Processing Agent Temperature When Emb. 5 Feather-Derived Oxidatively Hydrolyzed Keratin 40°C Before Bleaching 25.7 Emb. 6 60°C Before Bleaching 28.1 Emb. 7 80°C Before Blea ching 30.3 Com. Ex. 3 - - Before Bleaching 31.7 Unprocessed - - - 19.0 *unprocessed: cashmere hairs not bleached and without keratin treatment
2)FIG. 3 shows ATR-FTIR spectra of unprocessed hairs,Embodiments 5 to 7, and Comparative Example 3. The unprocessed hairs showed no peaks for sulfonic acid at 1040 cm-1, whereasEmbodiments 5 to 7 and Comparative Example 3 showed peaks for sulfonic acid at 1040 cm-1, and thus, it was suggested that excessive oxidation caused by bleaching occurred. However, compared with Comparative Example 3,Embodiments 5 to 7 had weaker peak intensities at 1040 cm-1, and thus, it was suggested that excessive oxidation caused by bleaching was suppressed. - Bleached cashmere hairs were soaked in a 0.1% feather-derived oxidatively hydrolyzed keratin solution, at a liquid ratio of 1: 20, at 40°C for 60 minutes. After the hairs were sufficiently washed with water, they were dried, and hydrolyzed keratin treated hairs were produced.
- The fiber damages and light resistant fastnesses of Embodiment 8 and the bleached cashmere hairs are listed in Table 4. In Embodiment 8, the fiber damage was smaller than the bleached cashmere hairs, suggesting the recovery from the fiber damage. Also, the light resistant fastness increased, suggesting the increased light resistance.
[Table 4] Keratin Treatment Fiber Damage Light Resistant Fastness Processing Agent Temperatur e When Emb. 8 Feather-Derived Oxidatively Hydrolyzed Keratin 40°C After Bleaching 62.6 4 or mor e Un-processed - - - 71.2 3 to 4 *unprocessed: bleached cashmere hairs without keratin treatment - Bleached and dyed cashmere hairs were soaked in a 0.5% feather-derived oxidatively hydrolyzed keratin solution, at a liquid ratio of 1: 20, at 60°C, for 20 minutes. The hairs were sufficiently washed with water and then dried, and thus, hydrolyzed keratin treated hairs were produced.
- Hydrolyzed keratin treated hairs were produced similarly to
Embodiment 7, while the hairs were soaked in a feather-derived oxidatively hydrolyzed keratin solution, at 80 °C. - The light resistant fastness of Embodiments 9 and 10 and the bleached and dyed cashmere hairs are listed in Table 5. Embodiments 9 and 10 showed increased light resistant fastness.
[Table 5] Keratin Treatment Light Resistant Fastness Processed Temperature When Emb. 9 Feather-Derived Oxidatively Hydrolyzed Keratin 60°C After Bleaching and Dyeing 3 to 4 Emb. 10 80°C After Bleaching and Dyeing 3 to 4 Unprocessed - - - 3 *unprocessed: bleached and dyed cashmere hairs that did not undergo keratin treatment - Pretreatment was performed by soaking cashmere hairs used in
Embodiment 5, in a 0.1% feather-derived oxidatively hydrolyzed keratin solution, at a liquid ratio of 1: 20, at 40°C, for 60 minutes. Thereafter, the hairs were soaked in a diluted solution of 35% hydrogen peroxide water having a concentration of 20 cc/L, at a liquid ratio of 1: 20, and were bleached at 60°C for 1 hour. After bleaching, the post-treatment was performed by soaking the hairs in a 0.1% feather-derived oxidatively hydrolyzed keratin solution, at a liquid ratio of 1: 20, at 40°C, for 60 minutes. The hairs were sufficiently washed with water and then dried, and thus hydrolyzed keratin pre/post-treated hairs. were produced. - Hydrolyzed keratin treated hairs were produced similarly to Embodiment 9, while the hairs were soaked in a feather-derived oxidatively hydrolyzed keratin solution, at 30 °C.
-
1) The results of fiber damage test and average values of panelist evaluations are listed in Table 6 regarding unprocessed hairs,Embodiments Embodiments Embodiments [Table 6] Keratin Treatment Fiber Damage Light Resistant Fastness Touch Feel Processing Agent Temperature When Emb. 11 Feather Derived Oxidatively Hydrolyzed Keratin 40°C Before and After Bleaching 19.1 4 or more 3.1 ±0.8 Emb. 12 30°C Before and After Bleaching 28.3 4 or more 3.2 ±0.9 Com. Ex. 3 - - Before and After Bleaching 31.7 4 2.4 ±0.6 Unprocessed - - - 19.0 4 or more 4.2 ±0.8 *unprocessed: cashmere hairs not bleached and without keratin treatment
2)FIG. 4 shows ATR-FTIR spectra of unprocessed hairs,Embodiments Embodiments - Pretreatment was performed by soaking cashmere hairs used in
Embodiment 5, in a 0.1% feather-derived oxidatively hydrolyzed keratin solution, at a liquid ratio of 1: 20, at 80°C, for 30 minutes. Thereafter, the hairs were soaked in a diluted solution of 35% hydrogen peroxide water with the concentration of 20 cc/L, at a liquid ratio of 1: 20, and were bleached at 60°C for 1 hour. After bleaching, dyeing was performed by a 0.03% acidic dye. post-treatment was performed on the bleached and dyed cashmere hairs by soaking the hairs in a 0.1% feather-derived oxidatively hydrolyzed keratin solution, at a liquid ratio of 1: 20, at 80°C, for 30 minutes. The hairs were sufficiently washed with water and then dried, and thus hydrolyzed keratin pre/post-treated hairs were produced. - Hydrolyzed keratin treated hairs were produced similarly to Embodiment 13, while the hairs were soaked in a 0.5% feather-derived oxidatively hydrolyzed keratin solution.
- Cashmere hairs similar to
Embodiment 5 were soaked in a diluted solution of 35% hydrogen peroxide water with the concentration of 20 cc/L, at a liquid ratio of 1: 20, and were bleached at 60°C for 1 hour. After bleaching, dyeing was performed by a 0.03% acidic dye. The hairs were sufficiently washed with water and then dried, and thus, keratin untreated hairs were produced. - The fiber damage and light resistant fastness of Embodiments 13 and 14 and Comparative Example 4 are listed in Table 7. Compared with Comparative Example 4, in Embodiments 13 and 14, the fiber damages were small, revealing the suppression of the fiber damage. Also, the light resistant fastness increased, revealing the increase in the light resistance.
[Table 7] Keratin Treatment Fiber Damage Light Resistance Fastness Processing Agent Temperature When Emb. 13 Feather Oxidatively Hydrolyzed Keratin 80°C Before and After Bleaching and Dyeing 22.6 3 to 4 Emb. 14 80°C Before and After Bleaching and Dyeing 26.6 4 or more Com. Ex. 4 - - Before and After Bleaching and Dyeing 29.3 3 - Pretreatment was performed by soaking spun sheep wool yarn in a 0.1% feather-derived oxidatively hydrolyzed keratin solution, at a liquid ratio of 1: 20, at 40°C. for 60 minutes. Thereafter, the hairs were soaked in a diluted solution of 35% hydrogen peroxide water (20 cc/L solution), at a liquid ratio of 1: 20, and were bleached at 60°C for 1 hour. After bleaching, post-treatment was performed by soaking the hairs in a 0.1% feather-derived oxidatively hydrolyzed keratin solution, at a liquid ratio of 1: 20, at 40°C, for 60 minutes. The hairs were sufficiently washed with water and then dried, and thus, hydrolyzed keratin treated spun yarn was produced.
- The spun sheep wool yarn was soaked in a diluted solution of 35% hydrogen peroxide water (20 cc/L solution), at a liquid ratio of 1: 20, and was bleached at 60°C for 1 hour. The yarn was sufficiently washed with water and then dried, and thus bleached sheep wool spun yarn was produced.
- The yarn strength and yarn elongation are listed in Table 8 regarding the raw yarn, Embodiment 15, and Comparative Example 5. Compared with Comparative Example 5, Embodiment 15 showed nearly equivalent values to the raw yarn before bleaching, and thus, yarn properties in the embodiment less changed in spite of the bleaching.
[Table 8] Keratin Treatment Yarn Strength (g) Yarn Elongation (%) Processing Agent Temperature When Emb. 15 Feather-Derived Oxidatively Hydrolyzed Keratin 40°C Before and After Bleaching 258.7 10.7 Com. Ex. 5 - - Before and After Bleaching 267.5 9.7 Raw Yarn - - - 252.0 12.9 *raw yarn: spun yarn not bleached and without keratin treatment - Hydrolyzed keratin treated cashmere spun yarn was produced similarly to Embodiment 15, while the sheep wool yarn was changed to cashmere yarn.
- Hydrolyzed keratin treated silk spun yarn was produced similarly to Embodiment 15, while the sheep wool yarn was changed to silk yarn.
- Hydrolyzed keratin treated alpaca spun yarn was produced similarly to Embodiment 15, while the sheep wool yarn was changed to alpaca yarn.
- Hydrolyzed keratin treated mixed spun yarn was produced similarly to Embodiment 15, while the sheep wool yarn was changed to mixed yarn of 80% mohair and 20% sheep wool.
- Hydrolyzed keratin treated angora spun yarn was produced similarly to Embodiment 15, while the sheep wool yarn was changed to angora yarn.
- Animal hairs similar to Embodiments 16 to 20 were soaked in a diluted solution of 35% hydrogen peroxide water(20 cc/L solution), at a liquid ratio of 1: 20, and were bleached at 60°C for 1 hour. The hairs were sufficiently washed with water and then dried, and thus keratin untreated spun yarn was produced.
- The yarn strength and yarn elongation of the raw yarn of animal hairs, Embodiments 16 to 20, and Comparative Examples 6 to 10 are listed in Table 9. By bleaching, the yarn strength and the yarn elongation tend to decrease. However, the keratin treatment is found to suppress a decrease in the yarn strength and also the yarn elongation.
[Table 9] Count Yarn Strength (g) Yarn Elongation (%) Cashmere Raw Yarn 2/20 390.5 11.8 Emb. 16 2/20 392.9 11.2 Com. Ex. 6 2/20 323.7 10.1 Silk Raw Yarn 2/60 911.0 9.0 Emb. 17 2/60 904.5 9.0 Com. Ex. 7 2/60 893.0 9.0 Alpaca Raw Yarn 2/24 554.0 16.1 Emb. 18 2/24 599.4 15.6 Com. Ex. 8 2/24 524.0 14.3 Mohair Wool Raw Yarn 1/24 265.9 18.3 Emb. 19 1/24 277.9 15.9 Com. Ex. 9 1/24 239.9 15.4 Angola Raw Yarn 2/24 419.4 14.7 Emb. 20 2/24 406.3 14.1 Com. Ex. 10 2/24 346.5 13.2 * raw yarn: spun yarn not bleached and without keratin treatment - Pretreatment was performed by soaking cashmere hairs in a 0.1% aqueous solution containing commercially available hydrolyzed keratin (average molecular weight of 1,000), at a temperature of 40°C, for 60 minutes. Thereafter, the hairs were soaked in a diluted solution of 35% hydrogen peroxide water (20 cc/L solution) at a liquid ratio of 1: 20, and were bleached at 60°C for 1 hour. After bleaching, the hairs were soaked in a 0.1% hydrolyzed keratin solution, at a liquid ratio of 1: 20, at 40°C for 60 minutes. The hairs were sufficiently washed with water and then dried, and thus hydrolyzed keratin treated hairs were produced.
- Hydrolyzed collagen treated hairs were produced similarly to
Embodiment 21, while the hydrolyzed keratin was changed to a commercially available hydrolyzed collagen (average molecular weight of 1,000). - Hydrolyzed silk treated hairs were produced similarly to
Embodiment 21, while the hydrolyzed keratin was changed to commercially available hydrolyzed silk (average molecular weight of 1,000). - The results of fiber damage test (alkaline solubility method) and the light resistant fastness of
Embodiments 21 and Comparative Examples 11 and 12 are listed in Table 10. Compared with Comparative Examples 11 and 12 treated with hydrolyzed collagen and hydrolyzed silk,Embodiment 21 treated with hydrolyzed keratin had the least fiber damage and the highest light resistant fastness.[Table 10] Hydrolyzed Protein Treatment Fiber Damage Light Resistant Fastness Processing Agent When Emb. 21 Hydrolyzed Keratin Before and After Bleaching 22.8 4 or more Com. Ex. 11 Hydrolyzed Collagen Before and After Bleaching 27.5 4 Com. Ex. 12 Hydrolyzed Silk Before and After Bleaching 24.5 4 -
FIG. 5 shows photographs of cotton-like fibers, before and after methylene blue dyeing onEmbodiment 21, Comparative Examples 11 and 12. The fiber damage is evaluated with a color concentration because undamaged fibers turn light blue and damaged fibers turn deep blue. According toFIG. 5 , Comparative Examples 11 and 12 were more deeply colored than unprocessed fibers.Embodiment 21 was more lightly colored than Comparative Examples 11 and 12. This suggests that withEmbodiment 21, in the evaluation with the methylene blue dyeing method as well, fiber damage caused by bleaching was suppressed similarly to the alkaline solubility method. Thus, the hydrolyzed keratin, among hydrolyzed proteins, effectively suppressed fiber damage.
Claims (10)
- A method for manufacturing processed fibers by bleaching and/or dyeing animal fibers, performing a step for soaking said fibers in a solution of hydrolyzed keratin.
- The method according to claim 1, wherein the hydrolyzed keratin solution has a concentration of 0.01 to 1 mass%.
- The method according to claim 1 or 2, wherein the step for soaking the fibers in the hydrolyzed keratin solution is performed before and after bleaching and/or dyeing.
- The method according to any one of claims 1 to 3, wherein the hydrolyzed keratin is one derived from sheep wool or feather.
- The method according to any one of claims 1 to 4, wherein the hydrolyzed keratin has a number average molecular weight of 10,000 to 130.
- The method according to any one of claims 1 to 5, wherein the animal fibers are one or more selected from sheep wool, cashmere, mohair, camel hair, llama, alpaca, vicuna, angora, mink, and silk.
- The method according to any one of claims 1 to 6, wherein the animal fibers are spun yarn.
- A method for suppressing damage in animal fibers, performing a step for soaking the animal fibers in a hydrolyzed keratin solution when bleaching and/or dyeing said fibers.
- A method for processing animal fibers, performing a step for soaking said fibers in a hydrolyzed keratin solution before, after, or before and after bleaching and/or dyeing.
- A processed fiber manufactured according to the method in one of claims 1 to 7.
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| JP2015171068 | 2015-08-31 | ||
| PCT/JP2016/075315 WO2017038814A1 (en) | 2015-08-31 | 2016-08-30 | Method for manufacturing processed fiber, processed fiber, method for suppressing damage to animal fiber, and method for processing animal fiber |
Publications (2)
| Publication Number | Publication Date |
|---|---|
| EP3346052A1 true EP3346052A1 (en) | 2018-07-11 |
| EP3346052A4 EP3346052A4 (en) | 2019-05-01 |
Family
ID=58187543
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| EP16841846.5A Pending EP3346052A4 (en) | 2015-08-31 | 2016-08-30 | Method for manufacturing processed fiber, processed fiber, method for suppressing damage to animal fiber, and method for processing animal fiber |
Country Status (8)
| Country | Link |
|---|---|
| EP (1) | EP3346052A4 (en) |
| JP (1) | JP6587690B2 (en) |
| KR (1) | KR102129971B1 (en) |
| CN (1) | CN108026692A (en) |
| AU (1) | AU2016317514B2 (en) |
| MY (1) | MY193256A (en) |
| NZ (1) | NZ740282A (en) |
| WO (1) | WO2017038814A1 (en) |
Cited By (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US11814782B2 (en) | 2017-05-15 | 2023-11-14 | Shima Seiki Mfg., Ltd. | Surface-processed fiber, method for manufacturing same, thread, and fiber product |
Families Citing this family (4)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US20210017672A1 (en) * | 2018-03-22 | 2021-01-21 | Shima Seiki Mfg., Ltd. | Protein fiber crimping method, protein fiber production method, protein fibers, spun yarn, and textile product |
| CN110565357A (en) * | 2019-06-11 | 2019-12-13 | 邵阳阳光发品有限公司 | Method for bleaching human hair fibers by complexing catalysis |
| CN110453522B (en) * | 2019-06-20 | 2022-02-18 | 天津科技大学 | Method for manufacturing non-woven wallpaper base paper by using 100% natural fibers |
| JP7473955B2 (en) | 2020-04-03 | 2024-04-24 | 西川株式会社 | Animal hair product manufacturing method |
Family Cites Families (14)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JPH01280074A (en) | 1988-05-02 | 1989-11-10 | Daito Boshoku Kk | Fabric and production of the same |
| JPH02242970A (en) | 1988-08-04 | 1990-09-27 | Honda Motor Co Ltd | Method for improving light and heat resistance of wool fiber |
| JPH05156573A (en) | 1991-03-11 | 1993-06-22 | Kanebo Ltd | Animal hair-based textile product with ultra-high light resistance and its production |
| JP3265329B2 (en) * | 1993-05-31 | 2002-03-11 | 中日本繊維工業協同組合 | How to process protein fiber products |
| JP3256748B2 (en) * | 1993-06-30 | 2002-02-12 | ライオン株式会社 | Liquid softener composition for clothing |
| JPH07300771A (en) * | 1994-04-21 | 1995-11-14 | Kiyoujiyuu:Kk | Shape memory wool and its production |
| JP2001518919A (en) | 1997-03-25 | 2001-10-16 | チバ スペシャルティ ケミカルズ ホールディング インコーポレーテッド | Optical brightener |
| JPH11172580A (en) | 1997-08-21 | 1999-06-29 | Nippon Chem Ind Co Ltd | Agent for treating animal hair, treating bath and treatment |
| JPH11139940A (en) * | 1997-10-31 | 1999-05-25 | Seiwa Kasei Kk | Preliminarily treating agent for dyeing hair |
| JP4157338B2 (en) | 2002-07-13 | 2008-10-01 | 敏夫 萩原 | Modification method for textile materials |
| JP2005146442A (en) | 2003-11-12 | 2005-06-09 | Nicca Chemical Co Ltd | Bleaching auxiliary for wool-based fiber and method for bleaching wool-based fiber |
| JP4931010B2 (en) * | 2007-04-13 | 2012-05-16 | 東洋羽毛工業株式会社 | Hair treatment method |
| CN103194919A (en) * | 2013-04-24 | 2013-07-10 | 太仓市其盛化纤厂 | Printing method beneficial to improving color saturation of wool fabric |
| CN103735428B (en) * | 2013-12-31 | 2015-04-08 | 广州市白云区大荣精细化工有限公司 | Keratin hair conditioner and preparation method thereof |
-
2016
- 2016-08-30 WO PCT/JP2016/075315 patent/WO2017038814A1/en active Application Filing
- 2016-08-30 NZ NZ740282A patent/NZ740282A/en unknown
- 2016-08-30 AU AU2016317514A patent/AU2016317514B2/en active Active
- 2016-08-30 EP EP16841846.5A patent/EP3346052A4/en active Pending
- 2016-08-30 KR KR1020187009161A patent/KR102129971B1/en active IP Right Grant
- 2016-08-30 CN CN201680050316.9A patent/CN108026692A/en active Pending
- 2016-08-30 MY MYPI2018700748A patent/MY193256A/en unknown
- 2016-08-30 JP JP2017538044A patent/JP6587690B2/en active Active
Cited By (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US11814782B2 (en) | 2017-05-15 | 2023-11-14 | Shima Seiki Mfg., Ltd. | Surface-processed fiber, method for manufacturing same, thread, and fiber product |
Also Published As
| Publication number | Publication date |
|---|---|
| WO2017038814A1 (en) | 2017-03-09 |
| JPWO2017038814A1 (en) | 2018-08-16 |
| KR20180042429A (en) | 2018-04-25 |
| CN108026692A (en) | 2018-05-11 |
| NZ740282A (en) | 2019-07-26 |
| KR102129971B1 (en) | 2020-07-03 |
| AU2016317514B2 (en) | 2019-07-25 |
| EP3346052A4 (en) | 2019-05-01 |
| MY193256A (en) | 2022-09-28 |
| AU2016317514A1 (en) | 2018-03-22 |
| JP6587690B2 (en) | 2019-10-09 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| EP3346052A1 (en) | Method for manufacturing processed fiber, processed fiber, method for suppressing damage to animal fiber, and method for processing animal fiber | |
| CN105648771A (en) | Antibacterial deodorant and anti-wrinkle finishing process | |
| Pekhtasheva et al. | Biodegradation and biodeterioration of some natural polymers | |
| CN102560696B (en) | Production method of natural colorful mulberry silk floss | |
| JP2010132595A (en) | Hair-treating agent having effect of protecting hair, and damage prevention and hair restoration | |
| JPH0610272A (en) | Finishing agent for woven fabric | |
| JP2009144282A (en) | Synthetic keratin fiber and method for producing synthetic keratin fiber | |
| US20150107616A1 (en) | Methods for processing keratinous fibers, and uses thereof | |
| JPH03213574A (en) | Modification of zootic hair fiber | |
| CN112618408B (en) | Composite protein hair restoration composition and application thereof | |
| Fleet et al. | Sunlight exposure caused yellowing and increased mineral content in wool | |
| CN103494354A (en) | Shell fabric formed by blending pearl fibers and copper ammonia fibers | |
| KR102185434B1 (en) | A method of producing eco-friendly hemp fibers including enzymatic treatment step | |
| JP2015078143A (en) | Hair treatment method | |
| Pekhtasheva et al. | Biodegradation and biodeterioration of some natural polymers | |
| CN110055771A (en) | A kind of bleaching of textile material and method of modifying and purposes | |
| JPH08134778A (en) | Method for processing fiber product | |
| Hassan | Possum fiber—A wonderful creation of nature | |
| JP3895217B2 (en) | Textile product and manufacturing method thereof | |
| WO2018109956A1 (en) | Biological-component bonded fiber and method for manufacturing same | |
| KR101465195B1 (en) | Uv protection finishing method of cellulose textile products | |
| JP2012087428A (en) | Antibacterial feather and method for producing the same | |
| JP2001234471A (en) | Method for processing cellulosic fiber | |
| CN106491490A (en) | A kind of fibroin albumen composite aquogel of anti-inflammation and preparation method thereof | |
| JP2009174111A (en) | Branched protein fiber, method for producing the same, floccular matter, branched protein fiber spun yarn, branched protein fiber-containing spun yarn, fabric, and nonwoven fabric |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| STAA | Information on the status of an ep patent application or granted ep patent |
Free format text: STATUS: THE INTERNATIONAL PUBLICATION HAS BEEN MADE |
|
| PUAI | Public reference made under article 153(3) epc to a published international application that has entered the european phase |
Free format text: ORIGINAL CODE: 0009012 |
|
| STAA | Information on the status of an ep patent application or granted ep patent |
Free format text: STATUS: REQUEST FOR EXAMINATION WAS MADE |
|
| 17P | Request for examination filed |
Effective date: 20180323 |
|
| AK | Designated contracting states |
Kind code of ref document: A1 Designated state(s): AL AT BE BG CH CY CZ DE DK EE ES FI FR GB GR HR HU IE IS IT LI LT LU LV MC MK MT NL NO PL PT RO RS SE SI SK SM TR |
|
| AX | Request for extension of the european patent |
Extension state: BA ME |
|
| DAV | Request for validation of the european patent (deleted) | ||
| DAX | Request for extension of the european patent (deleted) | ||
| A4 | Supplementary search report drawn up and despatched |
Effective date: 20190403 |
|
| RIC1 | Information provided on ipc code assigned before grant |
Ipc: D06M 101/12 20060101ALI20190328BHEP Ipc: D06L 4/12 20170101ALI20190328BHEP Ipc: D06M 15/15 20060101AFI20190328BHEP Ipc: D06P 1/46 20060101ALI20190328BHEP Ipc: D06L 4/00 20170101ALI20190328BHEP |
|
| STAA | Information on the status of an ep patent application or granted ep patent |
Free format text: STATUS: EXAMINATION IS IN PROGRESS |
|
| 17Q | First examination report despatched |
Effective date: 20220929 |