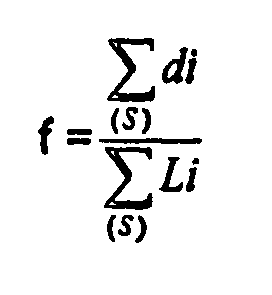EP1818422B2 - Ferritic stainless steel with 19% of chromium stabilised with niobium - Google Patents
Ferritic stainless steel with 19% of chromium stabilised with niobium Download PDFInfo
- Publication number
- EP1818422B2 EP1818422B2 EP07290039A EP07290039A EP1818422B2 EP 1818422 B2 EP1818422 B2 EP 1818422B2 EP 07290039 A EP07290039 A EP 07290039A EP 07290039 A EP07290039 A EP 07290039A EP 1818422 B2 EP1818422 B2 EP 1818422B2
- Authority
- EP
- European Patent Office
- Prior art keywords
- sheet
- ferritic
- steel sheet
- stainless steel
- niobium
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Active
Links
- 229910001220 stainless steel Inorganic materials 0.000 title claims abstract description 18
- 239000010955 niobium Substances 0.000 title abstract description 67
- 229910052758 niobium Inorganic materials 0.000 title abstract description 22
- GUCVJGMIXFAOAE-UHFFFAOYSA-N niobium atom Chemical compound [Nb] GUCVJGMIXFAOAE-UHFFFAOYSA-N 0.000 title abstract description 22
- 239000011651 chromium Substances 0.000 title abstract description 19
- 229910052804 chromium Inorganic materials 0.000 title abstract description 14
- VYZAMTAEIAYCRO-UHFFFAOYSA-N Chromium Chemical compound [Cr] VYZAMTAEIAYCRO-UHFFFAOYSA-N 0.000 title abstract description 11
- XEEYBQQBJWHFJM-UHFFFAOYSA-N Iron Chemical compound [Fe] XEEYBQQBJWHFJM-UHFFFAOYSA-N 0.000 claims abstract description 55
- 229910000831 Steel Inorganic materials 0.000 claims abstract description 54
- 239000010959 steel Substances 0.000 claims abstract description 54
- 239000000203 mixture Substances 0.000 claims abstract description 17
- 239000011265 semifinished product Substances 0.000 claims abstract description 8
- 229910052742 iron Inorganic materials 0.000 claims abstract description 7
- 239000012535 impurity Substances 0.000 claims abstract description 4
- 239000002244 precipitate Substances 0.000 claims description 29
- 238000001556 precipitation Methods 0.000 claims description 21
- 150000001875 compounds Chemical class 0.000 claims description 13
- 238000004519 manufacturing process Methods 0.000 claims description 8
- 238000000034 method Methods 0.000 claims description 7
- 230000000737 periodic effect Effects 0.000 claims description 4
- 238000003723 Smelting Methods 0.000 claims 1
- 239000000567 combustion gas Substances 0.000 claims 1
- IJGRMHOSHXDMSA-UHFFFAOYSA-N Atomic nitrogen Chemical compound N#N IJGRMHOSHXDMSA-UHFFFAOYSA-N 0.000 abstract description 24
- 239000010936 titanium Substances 0.000 abstract description 21
- 229910052799 carbon Inorganic materials 0.000 abstract description 15
- 229910052719 titanium Inorganic materials 0.000 abstract description 15
- OKTJSMMVPCPJKN-UHFFFAOYSA-N Carbon Chemical compound [C] OKTJSMMVPCPJKN-UHFFFAOYSA-N 0.000 abstract description 13
- RTAQQCXQSZGOHL-UHFFFAOYSA-N Titanium Chemical compound [Ti] RTAQQCXQSZGOHL-UHFFFAOYSA-N 0.000 abstract description 12
- 229910052757 nitrogen Inorganic materials 0.000 abstract description 12
- 229910052726 zirconium Inorganic materials 0.000 abstract description 9
- 229910052750 molybdenum Inorganic materials 0.000 abstract description 8
- 229910052710 silicon Inorganic materials 0.000 abstract description 8
- 229910052720 vanadium Inorganic materials 0.000 abstract description 8
- LEONUFNNVUYDNQ-UHFFFAOYSA-N vanadium atom Chemical compound [V] LEONUFNNVUYDNQ-UHFFFAOYSA-N 0.000 abstract description 8
- ZOKXTWBITQBERF-UHFFFAOYSA-N Molybdenum Chemical compound [Mo] ZOKXTWBITQBERF-UHFFFAOYSA-N 0.000 abstract description 6
- PXHVJJICTQNCMI-UHFFFAOYSA-N Nickel Chemical compound [Ni] PXHVJJICTQNCMI-UHFFFAOYSA-N 0.000 abstract description 6
- XUIMIQQOPSSXEZ-UHFFFAOYSA-N Silicon Chemical compound [Si] XUIMIQQOPSSXEZ-UHFFFAOYSA-N 0.000 abstract description 6
- QCWXUUIWCKQGHC-UHFFFAOYSA-N Zirconium Chemical compound [Zr] QCWXUUIWCKQGHC-UHFFFAOYSA-N 0.000 abstract description 6
- 229910052782 aluminium Inorganic materials 0.000 abstract description 6
- 239000011733 molybdenum Substances 0.000 abstract description 6
- 239000010703 silicon Substances 0.000 abstract description 6
- XAGFODPZIPBFFR-UHFFFAOYSA-N aluminium Chemical compound [Al] XAGFODPZIPBFFR-UHFFFAOYSA-N 0.000 abstract description 5
- RYGMFSIKBFXOCR-UHFFFAOYSA-N Copper Chemical compound [Cu] RYGMFSIKBFXOCR-UHFFFAOYSA-N 0.000 abstract description 4
- NINIDFKCEFEMDL-UHFFFAOYSA-N Sulfur Chemical compound [S] NINIDFKCEFEMDL-UHFFFAOYSA-N 0.000 abstract description 4
- 239000010949 copper Substances 0.000 abstract description 4
- 229910052802 copper Inorganic materials 0.000 abstract description 4
- 229910052717 sulfur Inorganic materials 0.000 abstract description 4
- 239000011593 sulfur Substances 0.000 abstract description 4
- PWHULOQIROXLJO-UHFFFAOYSA-N Manganese Chemical compound [Mn] PWHULOQIROXLJO-UHFFFAOYSA-N 0.000 abstract description 3
- ATJFFYVFTNAWJD-UHFFFAOYSA-N Tin Chemical compound [Sn] ATJFFYVFTNAWJD-UHFFFAOYSA-N 0.000 abstract description 3
- 238000005266 casting Methods 0.000 abstract description 3
- 239000010941 cobalt Substances 0.000 abstract description 3
- 229910017052 cobalt Inorganic materials 0.000 abstract description 3
- GUTLYIVDDKVIGB-UHFFFAOYSA-N cobalt atom Chemical compound [Co] GUTLYIVDDKVIGB-UHFFFAOYSA-N 0.000 abstract description 3
- 229910052748 manganese Inorganic materials 0.000 abstract description 3
- 239000011572 manganese Substances 0.000 abstract description 3
- 229910052759 nickel Inorganic materials 0.000 abstract description 3
- 238000007493 shaping process Methods 0.000 abstract description 2
- BHEPBYXIRTUNPN-UHFFFAOYSA-N hydridophosphorus(.) (triplet) Chemical compound [PH] BHEPBYXIRTUNPN-UHFFFAOYSA-N 0.000 abstract 2
- 238000002360 preparation method Methods 0.000 abstract 2
- 239000002245 particle Substances 0.000 abstract 1
- 238000003303 reheating Methods 0.000 abstract 1
- 238000007254 oxidation reaction Methods 0.000 description 20
- 230000003647 oxidation Effects 0.000 description 18
- 125000004122 cyclic group Chemical group 0.000 description 9
- 238000002524 electron diffraction data Methods 0.000 description 5
- 238000001953 recrystallisation Methods 0.000 description 5
- 230000006399 behavior Effects 0.000 description 4
- 230000007423 decrease Effects 0.000 description 4
- 230000002349 favourable effect Effects 0.000 description 4
- OAICVXFJPJFONN-UHFFFAOYSA-N Phosphorus Chemical compound [P] OAICVXFJPJFONN-UHFFFAOYSA-N 0.000 description 3
- 206010070834 Sensitisation Diseases 0.000 description 3
- 230000005540 biological transmission Effects 0.000 description 3
- 238000005260 corrosion Methods 0.000 description 3
- 230000007797 corrosion Effects 0.000 description 3
- 230000014509 gene expression Effects 0.000 description 3
- 238000005098 hot rolling Methods 0.000 description 3
- 229910000765 intermetallic Inorganic materials 0.000 description 3
- 239000010410 layer Substances 0.000 description 3
- 150000001247 metal acetylides Chemical class 0.000 description 3
- 229910052698 phosphorus Inorganic materials 0.000 description 3
- 239000011574 phosphorus Substances 0.000 description 3
- 230000008313 sensitization Effects 0.000 description 3
- 238000004627 transmission electron microscopy Methods 0.000 description 3
- 238000007792 addition Methods 0.000 description 2
- 230000001186 cumulative effect Effects 0.000 description 2
- 239000007789 gas Substances 0.000 description 2
- 229910001068 laves phase Inorganic materials 0.000 description 2
- 150000004767 nitrides Chemical class 0.000 description 2
- 238000011084 recovery Methods 0.000 description 2
- 238000004611 spectroscopical analysis Methods 0.000 description 2
- 230000000087 stabilizing effect Effects 0.000 description 2
- 238000005382 thermal cycling Methods 0.000 description 2
- 238000004458 analytical method Methods 0.000 description 1
- 238000004873 anchoring Methods 0.000 description 1
- 238000005452 bending Methods 0.000 description 1
- 230000015572 biosynthetic process Effects 0.000 description 1
- 238000005097 cold rolling Methods 0.000 description 1
- 238000009749 continuous casting Methods 0.000 description 1
- 238000001723 curing Methods 0.000 description 1
- 230000001627 detrimental effect Effects 0.000 description 1
- 239000006185 dispersion Substances 0.000 description 1
- 238000009826 distribution Methods 0.000 description 1
- 230000000694 effects Effects 0.000 description 1
- 238000001493 electron microscopy Methods 0.000 description 1
- 229940082150 encore Drugs 0.000 description 1
- 238000002149 energy-dispersive X-ray emission spectroscopy Methods 0.000 description 1
- 230000001747 exhibiting effect Effects 0.000 description 1
- 238000013007 heat curing Methods 0.000 description 1
- 238000010438 heat treatment Methods 0.000 description 1
- 238000012423 maintenance Methods 0.000 description 1
- 230000001376 precipitating effect Effects 0.000 description 1
- 239000006104 solid solution Substances 0.000 description 1
- 239000000243 solution Substances 0.000 description 1
- 229910052596 spinel Inorganic materials 0.000 description 1
- 239000011029 spinel Substances 0.000 description 1
- 239000000126 substance Substances 0.000 description 1
- 239000000758 substrate Substances 0.000 description 1
- 239000002344 surface layer Substances 0.000 description 1
- 238000009864 tensile test Methods 0.000 description 1
- 238000003466 welding Methods 0.000 description 1
- 229910000859 α-Fe Inorganic materials 0.000 description 1
Images
Classifications
-
- C—CHEMISTRY; METALLURGY
- C22—METALLURGY; FERROUS OR NON-FERROUS ALLOYS; TREATMENT OF ALLOYS OR NON-FERROUS METALS
- C22C—ALLOYS
- C22C38/00—Ferrous alloys, e.g. steel alloys
- C22C38/02—Ferrous alloys, e.g. steel alloys containing silicon
-
- C—CHEMISTRY; METALLURGY
- C21—METALLURGY OF IRON
- C21D—MODIFYING THE PHYSICAL STRUCTURE OF FERROUS METALS; GENERAL DEVICES FOR HEAT TREATMENT OF FERROUS OR NON-FERROUS METALS OR ALLOYS; MAKING METAL MALLEABLE, e.g. BY DECARBURISATION OR TEMPERING
- C21D6/00—Heat treatment of ferrous alloys
- C21D6/002—Heat treatment of ferrous alloys containing Cr
-
- C—CHEMISTRY; METALLURGY
- C21—METALLURGY OF IRON
- C21D—MODIFYING THE PHYSICAL STRUCTURE OF FERROUS METALS; GENERAL DEVICES FOR HEAT TREATMENT OF FERROUS OR NON-FERROUS METALS OR ALLOYS; MAKING METAL MALLEABLE, e.g. BY DECARBURISATION OR TEMPERING
- C21D8/00—Modifying the physical properties by deformation combined with, or followed by, heat treatment
- C21D8/02—Modifying the physical properties by deformation combined with, or followed by, heat treatment during manufacturing of plates or strips
- C21D8/0247—Modifying the physical properties by deformation combined with, or followed by, heat treatment during manufacturing of plates or strips characterised by the heat treatment
- C21D8/0273—Final recrystallisation annealing
-
- C—CHEMISTRY; METALLURGY
- C21—METALLURGY OF IRON
- C21D—MODIFYING THE PHYSICAL STRUCTURE OF FERROUS METALS; GENERAL DEVICES FOR HEAT TREATMENT OF FERROUS OR NON-FERROUS METALS OR ALLOYS; MAKING METAL MALLEABLE, e.g. BY DECARBURISATION OR TEMPERING
- C21D9/00—Heat treatment, e.g. annealing, hardening, quenching or tempering, adapted for particular articles; Furnaces therefor
- C21D9/0068—Heat treatment, e.g. annealing, hardening, quenching or tempering, adapted for particular articles; Furnaces therefor for particular articles not mentioned below
-
- C—CHEMISTRY; METALLURGY
- C22—METALLURGY; FERROUS OR NON-FERROUS ALLOYS; TREATMENT OF ALLOYS OR NON-FERROUS METALS
- C22C—ALLOYS
- C22C38/00—Ferrous alloys, e.g. steel alloys
- C22C38/001—Ferrous alloys, e.g. steel alloys containing N
-
- C—CHEMISTRY; METALLURGY
- C22—METALLURGY; FERROUS OR NON-FERROUS ALLOYS; TREATMENT OF ALLOYS OR NON-FERROUS METALS
- C22C—ALLOYS
- C22C38/00—Ferrous alloys, e.g. steel alloys
- C22C38/008—Ferrous alloys, e.g. steel alloys containing tin
-
- C—CHEMISTRY; METALLURGY
- C22—METALLURGY; FERROUS OR NON-FERROUS ALLOYS; TREATMENT OF ALLOYS OR NON-FERROUS METALS
- C22C—ALLOYS
- C22C38/00—Ferrous alloys, e.g. steel alloys
- C22C38/04—Ferrous alloys, e.g. steel alloys containing manganese
-
- C—CHEMISTRY; METALLURGY
- C22—METALLURGY; FERROUS OR NON-FERROUS ALLOYS; TREATMENT OF ALLOYS OR NON-FERROUS METALS
- C22C—ALLOYS
- C22C38/00—Ferrous alloys, e.g. steel alloys
- C22C38/18—Ferrous alloys, e.g. steel alloys containing chromium
- C22C38/20—Ferrous alloys, e.g. steel alloys containing chromium with copper
-
- C—CHEMISTRY; METALLURGY
- C22—METALLURGY; FERROUS OR NON-FERROUS ALLOYS; TREATMENT OF ALLOYS OR NON-FERROUS METALS
- C22C—ALLOYS
- C22C38/00—Ferrous alloys, e.g. steel alloys
- C22C38/18—Ferrous alloys, e.g. steel alloys containing chromium
- C22C38/22—Ferrous alloys, e.g. steel alloys containing chromium with molybdenum or tungsten
-
- C—CHEMISTRY; METALLURGY
- C22—METALLURGY; FERROUS OR NON-FERROUS ALLOYS; TREATMENT OF ALLOYS OR NON-FERROUS METALS
- C22C—ALLOYS
- C22C38/00—Ferrous alloys, e.g. steel alloys
- C22C38/18—Ferrous alloys, e.g. steel alloys containing chromium
- C22C38/24—Ferrous alloys, e.g. steel alloys containing chromium with vanadium
-
- C—CHEMISTRY; METALLURGY
- C22—METALLURGY; FERROUS OR NON-FERROUS ALLOYS; TREATMENT OF ALLOYS OR NON-FERROUS METALS
- C22C—ALLOYS
- C22C38/00—Ferrous alloys, e.g. steel alloys
- C22C38/18—Ferrous alloys, e.g. steel alloys containing chromium
- C22C38/26—Ferrous alloys, e.g. steel alloys containing chromium with niobium or tantalum
-
- C—CHEMISTRY; METALLURGY
- C22—METALLURGY; FERROUS OR NON-FERROUS ALLOYS; TREATMENT OF ALLOYS OR NON-FERROUS METALS
- C22C—ALLOYS
- C22C38/00—Ferrous alloys, e.g. steel alloys
- C22C38/18—Ferrous alloys, e.g. steel alloys containing chromium
- C22C38/30—Ferrous alloys, e.g. steel alloys containing chromium with cobalt
-
- C—CHEMISTRY; METALLURGY
- C22—METALLURGY; FERROUS OR NON-FERROUS ALLOYS; TREATMENT OF ALLOYS OR NON-FERROUS METALS
- C22C—ALLOYS
- C22C38/00—Ferrous alloys, e.g. steel alloys
- C22C38/18—Ferrous alloys, e.g. steel alloys containing chromium
- C22C38/40—Ferrous alloys, e.g. steel alloys containing chromium with nickel
- C22C38/44—Ferrous alloys, e.g. steel alloys containing chromium with nickel with molybdenum or tungsten
-
- C—CHEMISTRY; METALLURGY
- C22—METALLURGY; FERROUS OR NON-FERROUS ALLOYS; TREATMENT OF ALLOYS OR NON-FERROUS METALS
- C22C—ALLOYS
- C22C38/00—Ferrous alloys, e.g. steel alloys
- C22C38/18—Ferrous alloys, e.g. steel alloys containing chromium
- C22C38/40—Ferrous alloys, e.g. steel alloys containing chromium with nickel
- C22C38/46—Ferrous alloys, e.g. steel alloys containing chromium with nickel with vanadium
-
- C—CHEMISTRY; METALLURGY
- C22—METALLURGY; FERROUS OR NON-FERROUS ALLOYS; TREATMENT OF ALLOYS OR NON-FERROUS METALS
- C22C—ALLOYS
- C22C38/00—Ferrous alloys, e.g. steel alloys
- C22C38/18—Ferrous alloys, e.g. steel alloys containing chromium
- C22C38/40—Ferrous alloys, e.g. steel alloys containing chromium with nickel
- C22C38/48—Ferrous alloys, e.g. steel alloys containing chromium with nickel with niobium or tantalum
-
- C—CHEMISTRY; METALLURGY
- C22—METALLURGY; FERROUS OR NON-FERROUS ALLOYS; TREATMENT OF ALLOYS OR NON-FERROUS METALS
- C22C—ALLOYS
- C22C38/00—Ferrous alloys, e.g. steel alloys
- C22C38/18—Ferrous alloys, e.g. steel alloys containing chromium
- C22C38/40—Ferrous alloys, e.g. steel alloys containing chromium with nickel
- C22C38/52—Ferrous alloys, e.g. steel alloys containing chromium with nickel with cobalt
-
- C—CHEMISTRY; METALLURGY
- C21—METALLURGY OF IRON
- C21D—MODIFYING THE PHYSICAL STRUCTURE OF FERROUS METALS; GENERAL DEVICES FOR HEAT TREATMENT OF FERROUS OR NON-FERROUS METALS OR ALLOYS; MAKING METAL MALLEABLE, e.g. BY DECARBURISATION OR TEMPERING
- C21D2211/00—Microstructure comprising significant phases
- C21D2211/004—Dispersions; Precipitations
-
- C—CHEMISTRY; METALLURGY
- C21—METALLURGY OF IRON
- C21D—MODIFYING THE PHYSICAL STRUCTURE OF FERROUS METALS; GENERAL DEVICES FOR HEAT TREATMENT OF FERROUS OR NON-FERROUS METALS OR ALLOYS; MAKING METAL MALLEABLE, e.g. BY DECARBURISATION OR TEMPERING
- C21D2211/00—Microstructure comprising significant phases
- C21D2211/005—Ferrite
-
- F—MECHANICAL ENGINEERING; LIGHTING; HEATING; WEAPONS; BLASTING
- F01—MACHINES OR ENGINES IN GENERAL; ENGINE PLANTS IN GENERAL; STEAM ENGINES
- F01N—GAS-FLOW SILENCERS OR EXHAUST APPARATUS FOR MACHINES OR ENGINES IN GENERAL; GAS-FLOW SILENCERS OR EXHAUST APPARATUS FOR INTERNAL COMBUSTION ENGINES
- F01N13/00—Exhaust or silencing apparatus characterised by constructional features ; Exhaust or silencing apparatus, or parts thereof, having pertinent characteristics not provided for in, or of interest apart from, groups F01N1/00 - F01N5/00, F01N9/00, F01N11/00
- F01N13/08—Other arrangements or adaptations of exhaust conduits
- F01N13/10—Other arrangements or adaptations of exhaust conduits of exhaust manifolds
-
- F—MECHANICAL ENGINEERING; LIGHTING; HEATING; WEAPONS; BLASTING
- F01—MACHINES OR ENGINES IN GENERAL; ENGINE PLANTS IN GENERAL; STEAM ENGINES
- F01N—GAS-FLOW SILENCERS OR EXHAUST APPARATUS FOR MACHINES OR ENGINES IN GENERAL; GAS-FLOW SILENCERS OR EXHAUST APPARATUS FOR INTERNAL COMBUSTION ENGINES
- F01N13/00—Exhaust or silencing apparatus characterised by constructional features ; Exhaust or silencing apparatus, or parts thereof, having pertinent characteristics not provided for in, or of interest apart from, groups F01N1/00 - F01N5/00, F01N9/00, F01N11/00
- F01N13/16—Selection of particular materials
-
- F—MECHANICAL ENGINEERING; LIGHTING; HEATING; WEAPONS; BLASTING
- F01—MACHINES OR ENGINES IN GENERAL; ENGINE PLANTS IN GENERAL; STEAM ENGINES
- F01N—GAS-FLOW SILENCERS OR EXHAUST APPARATUS FOR MACHINES OR ENGINES IN GENERAL; GAS-FLOW SILENCERS OR EXHAUST APPARATUS FOR INTERNAL COMBUSTION ENGINES
- F01N2530/00—Selection of materials for tubes, chambers or housings
- F01N2530/02—Corrosion resistive metals
- F01N2530/04—Steel alloys, e.g. stainless steel
Definitions
- the invention relates to a ferritic stainless steel, said to be 19% Niobium stabilized Cr, and its use for parts subjected to high temperatures, in particular above 950-1000.degree.
- a good resistance to oxidation and a good mechanical resistance at high temperature are simultaneously sought: high mechanical characteristics, good resistance to creep and to thermal fatigue.
- the high-temperature mechanical behavior must also be adapted to the thermal cycles associated with the accelerator-deceleration phases of the engines.
- some parts such as exhaust manifolds require good cold formability to be shaped by bending or hydroforming.
- the present invention aims to solve the problems mentioned above. It aims in particular at providing a ferritic stainless steel that has good heat resistance, that is to say a high resistance to creep, thermal fatigue and oxidation at higher periodic temperatures of use at 950 ° C and cold forming ability close to existing grades.
- the subject of the invention is a ferritic stainless steel sheet according to claim 1.
- the steel piece resulting from this sheet contains an intergranular precipitation comprising at least 80% of cubic Fe 2 Nb 3 compounds.
- the linear fraction f of ferritic grain boundaries exhibiting a precipitation of cubic Fe 2 Nb 3 compounds is greater than or equal to 5%.
- the invention also relates to a method of manufacturing a ferritic stainless steel sheet according to claim 4.
- Another subject of the invention is a method for manufacturing a part according to which a sheet of ferritic stainless steel manufactured according to the above process is supplied, forming the sheet to obtain a part, and then subjecting the part to a or several thermal cycles in a temperature range between 650 and 1050 ° C for a cumulative duration of more than 30 minutes.
- the invention also relates to the use of a steel sheet according to the characteristics described above, or manufactured by the method described above, for the manufacture of parts subjected to a periodic temperature of use higher than 950 ° C, including automobile exhaust gas exhaust manifolds, burners, heat exchangers, turbocharger housings, or boilers.
- carbon increases mechanical characteristics at high temperatures, in particular creep resistance.
- the carbon tends to precipitate in the form of carbides M 23 C 6 or M 7 C 3 at a temperature below about 900 ° C.
- This precipitation generally located at grain boundaries, can lead to a depletion of chromium in the vicinity of these joints and thus to an awareness of intergranular corrosion.
- This sensitization can occur especially in the Heat Affected Zones in welding that have been heated to very high temperatures.
- the carbon content must therefore be limited to 0.03% to obtain a satisfactory resistance to intergranular corrosion and not to reduce the formability.
- the carbon content must satisfy a relationship with molybdenum, niobium and nitrogen, as will be explained later.
- chromium is a very effective element for increasing the resistance to oxidation during thermal cycling. To fulfill this role, a minimum content of 0.3% by weight is necessary. The inventors have also demonstrated that the weight contents of chromium and silicon must obey the relationship: Cr + 5 Si ⁇ 20%, so as to obtain good resistance to cyclic oxidation at 1000 ° C.
- the silicon content must be limited to 1% by weight.
- Sulfur and phosphorus are impurities that decrease hot ductility and formability. Phosphorus easily segregates at grain boundaries and decreases cohesion. As such, the sulfur and phosphorus contents must be respectively less than or equal to 0.01 and 0.04% by weight.
- Chromium is an essential element for stabilizing the ferritic phase and increasing the resistance to oxidation.
- its minimum content must be greater than or equal to 18% in order to obtain a ferritic structure at any temperature and to obtain good resistance to cyclic oxidation. Its maximum content must not, however, exceed 22%, otherwise the mechanical resistance to the ambient temperature will be excessively increased and the fitness ability will be reduced consecutively.
- Nickel is a gamma element that increases the ductility of steel. In order to maintain a ferritic single-phase structure, its content must be less than or equal to 0.5% by weight.
- Molybdenum not only increases the high temperature resistance but also the resistance to oxidation. However, above 2.5% by weight of Mo, the yield strength and room temperature resistance are excessively increased, ductility and workability decrease. As will be discussed below, molybdenum must also satisfy a relationship with niobium, carbon and nitrogen, to obtain satisfactory mechanical strength and creep resistance at 1000 ° C and fatigue resistance. between 100 ° C and 1000 ° C.
- Copper has a heat-curing effect. In excessive quantities, however, it reduces the ductility during hot rolling. As such, the copper content must be less than or equal to 0.5% by weight.
- the inventors have demonstrated that the contents of titanium, aluminum and zirconium must be jointly limited in order to obtain a more intense precipitation of cubic Fe 2 Nb 3 : this precipitation of compounds intermetallic acting at high temperature, provides good resistance to cyclic oxidation and creep at 1000 ° C.
- the weight contents of Ti, Zr, Al must be limited to 0.02% each, and the sum of their contents must be such that: Ti + Al + Zr 0,0 0.030%.
- niobium precipitates, not in the form of Fe 2 Nb 3 , but from 650 ° C in the form of Fe 2 Nb compounds, less effective to resist creep.
- Niobium is an important element of the invention. Usually, this element can be used as a stabilizing element in ferritic stainless steels: in fact, the sensitization phenomenon mentioned above can be avoided by the addition of elements forming carbides or carbonitrides which are very thermally stable. In this way, carbon and nitrogen are minimized in solution, and subsequent precipitation of carbides and nitrides of chromium is avoided. Niobium (as well as titanium and, to a lesser extent, zirconium and vanadium) thus stably fixes carbon and nitrogen.
- niobium content is greater than 1% by weight, the hardening obtained is too important, the steel is less easily deformable and recrystallization after cold rolling is more difficult .
- Vanadium is an element that increases resistance to high temperature. In order to ensure satisfactory adhesion of the oxide layer formed during use at high temperature and to ensure good resistance to oxidation, the inventors have shown that the titanium and vanadium contents must satisfy the relationship: V +10 Ti ⁇ 0.06%. However, the vanadium content should be limited to 0.2% in order not to reduce the formability.
- nitrogen increases the mechanical characteristics. However, nitrogen tends to precipitate at grain boundaries as nitrides, thus reducing corrosion resistance. In order to limit sensitization problems, the nitrogen content must be less than or equal to 0.03%.
- the average grain size of the steel in the delivery state is between 10 and 60 micrometers, the subsequent precipitation of intermetallic compounds also making it possible to stabilize the grain size during use.
- a grain size of less than 10 microns has a detrimental effect on intergranular creep.
- a grain size greater than 60 microns will lead to the appearance of unsightly surface irregularities, or "orange peel", when shaping at room temperature.
- the steels according to the invention comprise an intergranular precipitation of Fe 2 Nb 3 compounds of cubic structure, after a heat treatment of between 650 ° C. and 1050 ° C. for a time greater than 30 minutes.
- the Fe 2 Nb 3 precipitates are very much in the majority of the intergranular precipitates, that is to say they represent more than 80% of the intergranular population.
- the nature and distribution of these precipitates are very favorable to resist creep, in comparison with Fe 2 Nb precipitates, or Laves phases.
- the Fe 2 Nb compounds which precipitate in intra- or intergranular form are stable only up to 950 ° C., unlike stable Fe 2 Nb 3 precipitates up to 1050 ° C.
- the structure of the steel in the delivery state is completely recrystallized: in this way, the Subsequent precipitation of Fe 2 Nb 3 compounds occurs in a very homogeneous manner.
- the expression f thus translates the degree of recovery of ferritic grain boundaries by a precipitation of cubic Fe 2 Nb 3 .
- the inventors have highlighted, as the present figure 2 , that the creep resistance in a so-called sag-test, was very much improved when the linear fraction of cubic Fe 2 Nb 3 precipitates was greater than or equal to 5%: under these conditions, these precipitates play a role. very effective anchoring of joints and slow creep.
- the sheet is then scoured and the sheet is then rolled under the usual conditions, for example by applying a reduction ratio of 30 to 90%.
- the cold-rolled sheet is then annealed at a temperature T R and for a time t R.
- T R and t R are chosen such that a complete recrystallization with an average ferritic grain size of between 10 and 60 microns is obtained.
- An increase in T R and t R increases the recrystallization rate as well as the average grain size.
- a temperature T R of between 1030 and 1130 ° C. and a time t R of between 10 seconds and 3 minutes make it possible simultaneously to obtain a complete recrystallization and a mean ferritic grain size of between 10 and 60 microns.
- the steel sheet is in the delivery condition.
- a part can then be manufactured from this sheet steel by implementing common modes of deformation, such as stamping, hydroforming or folding.
- common modes of deformation such as stamping, hydroforming or folding.
- one or more thermal cycles in a temperature range between 650 and 1050 ° C for a cumulative time greater than 30 minutes lead to a precipitation of Fe 2 Nb 3 and an increase of creep resistance.
- This resistance is particularly high when the linear fraction f of ferritic grain boundaries comprising a precipitation of Fe 2 Nb 3 compounds is greater than or equal to 5%.
- the recrystallization is complete and the average ferritic grain size is between 10 and 60 micrometers.
- the figure 3 annexed illustrates the precipitates observed after creep tests at 1000 ° C in the flows I1, I2, I3 and I4 according to the invention.
- the presence of intra- and especially intergranular precipitates covering a large part of the ferritic grain boundaries is noted.
- Analyzes by energy dispersive spectrometry (EDS) and wavelength (WDS) reveal that more than 80% of these precipitates consist of niobium and iron, of Fe 2 Nb 3 stoichiometry, and that they do not contain neither carbon nor nitrogen.
- figure 6 shows precipitates observed in the reference steels R3, R4 and R5.
- the degree of recovery of the ferritic grain boundaries by these precipitates is very low after creep tests at 1000 ° C.
- Experimental electron diffraction patterns and theoretical views along the zone axis of these precipitates are plotted respectively at Figures 7 and 8 .
- the EDS analysis and the diffraction examinations reveal that they are precipitated Fe 2 Nb, or Laves phase, of hexagonal network.
- the steels I1 to I4 according to the invention combine good mechanical properties when hot: mechanical resistance, resistance to creep, thermal fatigue and cyclic oxidation.
- the R1 and R2 steels have a combination of insufficient titanium and vanadium: the resistances to cyclic oxidation and thermal fatigue are unsatisfactory due to the lack of adhesion of the oxide layers to the substrate steel.
- R5 steel also has an excessive titanium content and a combination (Ti + Al + Zr) unsatisfactory. Moreover, its combination: Mo + 3 ⁇ Nb, is insufficient. As a result, the steel does not exhibit satisfactory mechanical properties at high temperature, in particular creep.
- R6 steel has an insufficient chromium content as well as combinations: Mo + 3 ⁇ Nb, Cr + 5% Si insufficient. Despite the presence of Fe 2 Nb 3 compounds, the properties of oxidation resistance and high temperature mechanical properties are insufficient.
- the steels according to the invention will be used with advantage for the manufacture of parts subjected to a temperature periodic use greater than 950 ° C, and in particular exhaust gas collectors in the automotive field, burners, heat exchangers or envelopes turbochargers, boilers.
Landscapes
- Chemical & Material Sciences (AREA)
- Engineering & Computer Science (AREA)
- Materials Engineering (AREA)
- Mechanical Engineering (AREA)
- Metallurgy (AREA)
- Organic Chemistry (AREA)
- Physics & Mathematics (AREA)
- Thermal Sciences (AREA)
- Crystallography & Structural Chemistry (AREA)
- Heat Treatment Of Sheet Steel (AREA)
- Treatment Of Steel In Its Molten State (AREA)
- Catalysts (AREA)
Abstract
Description
L'invention concerne un acier inoxydable ferritique, dit à 19%Cr stabilisé au niobium, et son utilisation pour des pièces soumises à des températures élevées, en particulier à plus de 950-1000°C.The invention relates to a ferritic stainless steel, said to be 19% Niobium stabilized Cr, and its use for parts subjected to high temperatures, in particular above 950-1000.degree.
Pour certaines applications telles que les pièces situées dans les parties chaudes des lignes d'échappement pour l'automobile, on recherche simultanément une bonne résistance à l'oxydation et une bonne tenue mécanique à haute température : caractéristiques mécaniques élevées, bonnes tenues au fluage et à la fatigue thermique. La tenue mécanique à haute température doit être également adaptée aux cycles thermiques associés aux phases d'accélérations-décélérations des moteurs. En outre, certaines parties telles que les collecteurs des gaz d'échappement requièrent une bonne formabilité à froid pour être mises en forme par pliage ou par hydroformage.For certain applications such as the parts located in the hot parts of the exhaust lines for the automobile, a good resistance to oxidation and a good mechanical resistance at high temperature are simultaneously sought: high mechanical characteristics, good resistance to creep and to thermal fatigue. The high-temperature mechanical behavior must also be adapted to the thermal cycles associated with the accelerator-deceleration phases of the engines. In addition, some parts such as exhaust manifolds require good cold formability to be shaped by bending or hydroforming.
Différentes nuances d'aciers inoxydables austénitiques ou ferritiques ont été proposées pour répondre aux exigences spécifiques des différentes zones de la ligne d'échappement. On a noté en particulier un développement de certaines nuances d'aciers inoxydables ferritiques : Ceci est dû à leur coût moins élevé que celui des aciers austénitiques ou réfractaires, ainsi qu'à leur meilleure tenue à l'oxydation cyclique, ce dernier point résultant d'une différence de coefficient de dilatation entre l'acier et la couche superficielle d'oxydes moindre pour les aciers ferritiques que pour les aciers austénitiques. On connaît ainsi des aciers inoxydables ferritiques à 17%Cr stabilisés avec 0,14% de titane et 0,5% de niobium (type EN 1.4509, AISI 441) Ce type de nuances n'est cependant pas adapté aux parties les plus chaudes des lignes d'échappement lorsque les températures sont supérieures à 950°C, car leur résistance au fluage est insuffisante et l'oxydation à haute température se produit de façon excessive. On connaît également des aciers inoxydables ferritiques à 14%Cr stabilisés avec 0,5% de niobium sans titane (type EN 1.4595). Ceux-ci présentent une tenue à haute température équivalente à celle des nuances précédentes, mais une meilleure aptitude à la mise en forme. Cependant la température maximale d'utilisation reste 950°C. Selon
La présente invention a pour but de résoudre les problèmes évoqués ci-dessus. Elle vise en particulier à mettre à disposition un acier inoxydable ferritique qui présente une bonne tenue à chaud, c'est-à-dire une résistance élevée au fluage, à la fatigue thermique et à l'oxydation à des températures d'utilisation périodique supérieures à 950°C ainsi qu'une aptitude à la mise en forme à froid proche des nuances existantes.The present invention aims to solve the problems mentioned above. It aims in particular at providing a ferritic stainless steel that has good heat resistance, that is to say a high resistance to creep, thermal fatigue and oxidation at higher periodic temperatures of use at 950 ° C and cold forming ability close to existing grades.
Dans ce but, l'invention a pour objet une tôle d'acier inoxydable ferritique selon la revendication 1.For this purpose, the subject of the invention is a ferritic stainless steel sheet according to
Préférentiellement, la pièce d'acier issue de cette tôle contient une précipitation intergranulaire comprenant au moins 80% de composés Fe2Nb3 cubiques.Preferably, the steel piece resulting from this sheet contains an intergranular precipitation comprising at least 80% of cubic Fe 2 Nb 3 compounds.
Préférentiellement encore, la fraction linéaire f de joints de grains ferritiques présentant une précipitation de composés Fe2Nb3 cubiques, est supérieure ou égale à 5%.Also preferably, the linear fraction f of ferritic grain boundaries exhibiting a precipitation of cubic Fe 2 Nb 3 compounds is greater than or equal to 5%.
L'invention a également pour objet un procédé de fabrication d'une tôle d'acier inoxydable ferritique, selon la revendication 4.The invention also relates to a method of manufacturing a ferritic stainless steel sheet according to claim 4.
L'invention a également pour objet un procédé de fabrication d'une pièce selon lequel on approvisionne une tôle d'acier inoxydable ferritique fabriquée selon le procédé ci-dessus, on forme la tôle pour obtenir une pièce, puis on soumet la pièce à un ou plusieurs cycles thermiques dans un domaine de températures comprises entre 650 et 1050°C pendant une durée cumulée supérieure à 30 minutes.Another subject of the invention is a method for manufacturing a part according to which a sheet of ferritic stainless steel manufactured according to the above process is supplied, forming the sheet to obtain a part, and then subjecting the part to a or several thermal cycles in a temperature range between 650 and 1050 ° C for a cumulative duration of more than 30 minutes.
L'invention a également pour objet l'utilisation d'une tôle d'acier selon les caractéristiques décrites ci-dessus, ou fabriquée par le procédé décrit ci-dessus, pour la fabrication de pièces soumises à une température d'utilisation périodique supérieure à 950°C, et notamment de collecteurs d'échappement de gaz de combustion dans le domaine automobile, de brûleurs, d'échangeurs à chaleur, d'enveloppes de turbocompresseurs, ou de chaudières.The invention also relates to the use of a steel sheet according to the characteristics described above, or manufactured by the method described above, for the manufacture of parts subjected to a periodic temperature of use higher than 950 ° C, including automobile exhaust gas exhaust manifolds, burners, heat exchangers, turbocharger housings, or boilers.
D'autres caractéristiques et avantages de l'invention apparaîtront au cours de la description ci-dessous donnée à titre d'exemple et faite en référence aux figures jointes suivantes :
- La
figure 1 définit schématiquement la fraction linéaire f de joints de grains ferritiques comportant une précipitation de Fe2Nb3 cubiques. - - La
figure 2 présente l'influence de la fraction f sur le comportement en fluage. - - La
figure 3 présente une observation en Microscopie Electronique en Transmission de précipités cubiques Fe2Nb3 dans une tôle laminée à froid et recuite d'un acier selon l'invention, après traitement de 100h à 1000°C. - - Les
figures 4 et 5 présentent respectivement des clichés de diffraction électronique et des clichés théoriques selon l'axe de zone de ces précipités Fe2Nb3. - - La
figure 6 présente une observation en Microscopie Electronique en Transmission de précipités hexagonaux Fe2Nb dans une tôle laminée à froid et recuite d'un acier de référence, après traitement de 100h à 1000°C. - - Les
figures 7 et 8 présentent respectivement des clichés de diffraction électronique et des clichés théoriques selon l'axe de zone de ces précipités Fe2Nb.
- The
figure 1 schematically defines the linear fraction f of ferritic grain boundaries with cubic Fe 2 Nb 3 precipitation. - - The
figure 2 shows the influence of fraction f on creep behavior. - - The
figure 3 presents an observation in Transmission Electron Microscopy of cubic Fe 2 Nb 3 precipitates in a cold-rolled and annealed sheet of a steel according to the invention, after treatment of 100 h at 1000 ° C. - - The
Figures 4 and 5 present respectively electron diffraction patterns and theoretical views along the zone axis of these Fe 2 Nb 3 precipitates. - - The
figure 6 presents an observation in Transmission Electron Microscopy of Fe 2 Nb hexagonal precipitates in a cold-rolled and annealed sheet of a reference steel, after treatment of 100h at 1000 ° C. - - The
Figures 7 and 8 show respectively electron diffraction patterns and theoretical views along the zone axis of these Fe 2 Nb precipitates.
En ce qui concerne la composition chimique de l'acier, le carbone augmente les caractéristiques mécaniques à haute température, en particulier la résistance au fluage. Cependant, en raison de sa solubilité très faible dans la ferrite, le carbone tend à précipiter sous forme de carbures M23C6 ou M7C3 à une température inférieure à 900°C environ. Cette précipitation généralement située aux joints de grains peut conduire à un appauvrissement en chrome au voisinage de ces joints et donc à une sensibilisation à la corrosion intergranulaire. Cette sensibilisation peut se rencontrer en particulier dans les Zones Affectées par la Chaleur en soudage qui ont été réchauffées à très haute température. La teneur en carbone doit donc être limitée à 0,03% pour obtenir une résistance satisfaisante à la corrosion intergranulaire ainsi que pour ne pas diminuer la formabilité. De plus, la teneur en carbone doit satisfaire à une relation avec le molybdène, le niobium et l'azote, comme il sera expliqué plus loin.With regard to the chemical composition of steel, carbon increases mechanical characteristics at high temperatures, in particular creep resistance. However, because of its very low solubility in ferrite, the carbon tends to precipitate in the form of carbides M 23 C 6 or M 7 C 3 at a temperature below about 900 ° C. This precipitation, generally located at grain boundaries, can lead to a depletion of chromium in the vicinity of these joints and thus to an awareness of intergranular corrosion. This sensitization can occur especially in the Heat Affected Zones in welding that have been heated to very high temperatures. The carbon content must therefore be limited to 0.03% to obtain a satisfactory resistance to intergranular corrosion and not to reduce the formability. In addition, the carbon content must satisfy a relationship with molybdenum, niobium and nitrogen, as will be explained later.
Le manganèse accroît les caractéristiques mécaniques. Au delà de 1 % en poids, la cinétique d'oxydation à chaud devient cependant trop rapide et une couche d'oxyde moins compacte se développe, formée de spinelle avec de la chromine.Manganese increases the mechanical characteristics. Above 1% by weight, the kinetics of hot oxidation however becomes too fast and a less compact oxide layer develops, formed of spinel with chromine.
Comme le chrome, le silicium est un élément très efficace pour accroître la résistance à l'oxydation lors de cycles thermiques. Pour assurer ce rôle, une teneur minimale de 0,3% en poids est nécessaire. Les inventeurs ont également mis en évidence que les teneurs pondérales en chrome et en silicium devaient obéir à la relation : Cr + 5 Si ≥ 20%, de façon à obtenir une bonne résistance à l'oxydation cyclique à 1000°C.Like chromium, silicon is a very effective element for increasing the resistance to oxidation during thermal cycling. To fulfill this role, a minimum content of 0.3% by weight is necessary. The inventors have also demonstrated that the weight contents of chromium and silicon must obey the relationship: Cr + 5 Si ≥ 20%, so as to obtain good resistance to cyclic oxidation at 1000 ° C.
Cependant, pour ne pas diminuer l'aptitude au laminage à chaud et la mise en forme à froid, la teneur en silicium doit être limitée à 1% en poids.However, in order not to decrease the hot rolling ability and the cold forming, the silicon content must be limited to 1% by weight.
Le soufre et le phosphore sont des impuretés qui diminuent la ductilité à chaud et la formabilité. Le phosphore ségrége facilement aux joints de grains et diminue leur cohésion. A ce titre, les teneurs en soufre et phosphore doivent être respectivement inférieures ou égales à 0,01 et 0,04% en poids.Sulfur and phosphorus are impurities that decrease hot ductility and formability. Phosphorus easily segregates at grain boundaries and decreases cohesion. As such, the sulfur and phosphorus contents must be respectively less than or equal to 0.01 and 0.04% by weight.
Le chrome est un élément essentiel pour la stabilisation de la phase ferritique et pour accroître la résistance à l'oxydation. En liaison avec les autres éléments de la composition, sa teneur minimale doit être supérieure ou égale à 18% afin d'obtenir une structure ferritique à toute température et d'obtenir une bonne résistance à l'oxydation cyclique. Sa teneur maximale ne doit pas cependant excéder 22% sous peine d'augmenter excessivement la résistance mécanique à l'ambiante et de diminuer consécutivement l'aptitude à la mise en forme.Chromium is an essential element for stabilizing the ferritic phase and increasing the resistance to oxidation. In connection with the other elements of the composition, its minimum content must be greater than or equal to 18% in order to obtain a ferritic structure at any temperature and to obtain good resistance to cyclic oxidation. Its maximum content must not, however, exceed 22%, otherwise the mechanical resistance to the ambient temperature will be excessively increased and the fitness ability will be reduced consecutively.
Le nickel est un élément gammagène qui augmente la ductilité de l'acier. Afin de conserver une structure monophasée ferritique, sa teneur doit être inférieure ou égale à 0,5% en poids.Nickel is a gamma element that increases the ductility of steel. In order to maintain a ferritic single-phase structure, its content must be less than or equal to 0.5% by weight.
Le molybdène accroît non seulement la résistance à haute température mais aussi la résistance à l'oxydation. Cependant, au delà de 2,5% en poids de Mo, la limite d'élasticité et la résistance à température ambiante sont accrues de façon excessive, la ductilité et l'aptitude à la mise en forme diminuent. Comme on le verra plus loin, le molybdène doit également satisfaire à une relation conjointement avec le niobium, le carbone et l'azote, pour obtenir une résistance mécanique et une résistance au fluage satisfaisantes à 1000°C ainsi qu'une résistance à la fatigue thermique entre 100°C et 1000°C.Molybdenum not only increases the high temperature resistance but also the resistance to oxidation. However, above 2.5% by weight of Mo, the yield strength and room temperature resistance are excessively increased, ductility and workability decrease. As will be discussed below, molybdenum must also satisfy a relationship with niobium, carbon and nitrogen, to obtain satisfactory mechanical strength and creep resistance at 1000 ° C and fatigue resistance. between 100 ° C and 1000 ° C.
Le cuivre a un effet durcissant à chaud. En quantité excessive, il diminue cependant la ductilité lors du laminage à chaud. A ce titre, la teneur en cuivre doit donc être inférieure ou égale à 0,5% en poids.Copper has a heat-curing effect. In excessive quantities, however, it reduces the ductility during hot rolling. As such, the copper content must be less than or equal to 0.5% by weight.
Les inventeurs ont mis en évidence que les teneurs en titane, en aluminium et en zirconium doivent être conjointement limitées afin d'obtenir une précipitation plus intense de Fe2Nb3 cubiques : cette précipitation de composés intermétalliques intervenant à haute température, permet d'obtenir une bonne tenue à l'oxydation cyclique et au fluage à 1000°C. Dans ce but, les teneurs pondérales en Ti, Zr, Al, doivent être limitées à 0,02% chacune, et la somme de leurs teneurs doit être telle que : Ti+Al+Zr≤ 0,030%. Dans le cas contraire, le niobium précipite, non pas sous forme de Fe2Nb3, mais à partir de 650°C sous forme de composés Fe2Nb, moins efficaces pour résister au fluage.The inventors have demonstrated that the contents of titanium, aluminum and zirconium must be jointly limited in order to obtain a more intense precipitation of cubic Fe 2 Nb 3 : this precipitation of compounds intermetallic acting at high temperature, provides good resistance to cyclic oxidation and creep at 1000 ° C. For this purpose, the weight contents of Ti, Zr, Al must be limited to 0.02% each, and the sum of their contents must be such that: Ti + Al +
Le niobium est un élément important de l'invention. Usuellement, cet élément peut être utilisé comme élément stabilisant dans les aciers inoxydables ferritiques : en effet, le phénomène de sensibilisation mentionné ci-dessus peut être évité par l'addition d'éléments formant des carbures ou des carbonitrures très stables thermiquement. De cette façon, on réduit le plus possible le carbone et l'azote en solution et on évite ainsi une précipitation ultérieure de carbures et de nitrures de chrome. Le niobium (ainsi que le titane et, dans une moindre mesure, le zirconium et le vanadium) fixe donc de façon stable le carbone et l'azote.Niobium is an important element of the invention. Usually, this element can be used as a stabilizing element in ferritic stainless steels: in fact, the sensitization phenomenon mentioned above can be avoided by the addition of elements forming carbides or carbonitrides which are very thermally stable. In this way, carbon and nitrogen are minimized in solution, and subsequent precipitation of carbides and nitrides of chromium is avoided. Niobium (as well as titanium and, to a lesser extent, zirconium and vanadium) thus stably fixes carbon and nitrogen.
Mais le niobium se combine également avec le fer pour former certains composés intermétalliques dans l'intervalle 650°C-1050°C: les inventeurs ont mis en évidence qu'une précipitation intergranulaire de Fe2Nb3 cubique intervenant à haute température pouvait être mise à profit pour augmenter les propriétés mécaniques à chaud. Ceci nécessite cependant les conditions suivantes :
- Si la teneur en Nb total de l'acier est inférieure à 0,2%, l'acier est insuffisamment stabilisé et la quantité de Fe2Nb3 précipité est insuffisante pour obtenir les propriétés visées à haute température.
- If the total Nb content of the steel is less than 0.2%, the steel is insufficiently stabilized and the amount of Fe 2 Nb 3 precipitated is insufficient to obtain the properties targeted at high temperature.
Pour obtenir cette précipitation favorable du niobium, les inventeurs ont également mis en évidence l'importance de la teneur en niobium effectif, désignée par ΔNb : le niobium effectif désigne la quantité de niobium en solution solide disponible pour précipiter avec le fer, en faisant l'hypothèse que le carbone et l'azote ont totalement précipité avec le niobium sous forme de carbonitrures NbCN. Dans ces conditions : ΔNb= Nb -7C - 7NTo obtain this favorable precipitation of niobium, the inventors have also demonstrated the importance of the effective niobium content, designated by ΔNb: the effective niobium denotes the amount of niobium in solid solution available for precipitating with iron, by making hypothesis that carbon and nitrogen precipitated completely with niobium as NbCN carbonitrides. Under these conditions: ΔNb = Nb -7C - 7N
Pour garantir une résistance mécanique et une résistance au fluage à 1000°C satisfaisantes, ainsi qu'une résistance à la fatigue thermique entre 100°C et 1000°C, les inventeurs ont mis en évidence que les teneurs en Mo et en ΔNb devaient excéder une valeur particulière de façon que :
Cependant, il convient par ailleurs de limiter les additions de niobium : Lorsque la teneur en niobium est supérieure à 1% en poids, le durcissement obtenu est trop important, l'acier est moins facilement déformable et la recristallisation après laminage à froid est plus difficile.However, it is also necessary to limit the additions of niobium: When the niobium content is greater than 1% by weight, the hardening obtained is too important, the steel is less easily deformable and recrystallization after cold rolling is more difficult .
Le vanadium est un élément qui augmente la résistance à haute température. Afin d'assurer une adhérence satisfaisante de la couche d'oxyde formée lors de l'utilisation à haute température et de garantir une bonne tenue à l'oxydation, les inventeurs ont mis en évidence que les teneurs en titane et vanadium doivent satisfaire à la relation : V +10 Ti ≥ 0,06%. Il convient cependant de limiter la teneur en vanadium à 0,2% pour ne pas diminuer la formabilité.Vanadium is an element that increases resistance to high temperature. In order to ensure satisfactory adhesion of the oxide layer formed during use at high temperature and to ensure good resistance to oxidation, the inventors have shown that the titanium and vanadium contents must satisfy the relationship: V +10 Ti ≥ 0.06%. However, the vanadium content should be limited to 0.2% in order not to reduce the formability.
Comme le carbone, l'azote augmente les caractéristiques mécaniques. Cependant, l'azote tend à précipiter aux joints de grains sous forme de nitrures, réduisant ainsi la résistance à la corrosion. Afin de limiter les problèmes de sensibilisation, la teneur en azote doit être inférieure ou égale à 0,03%.Like carbon, nitrogen increases the mechanical characteristics. However, nitrogen tends to precipitate at grain boundaries as nitrides, thus reducing corrosion resistance. In order to limit sensitization problems, the nitrogen content must be less than or equal to 0.03%.
Le cobalt est un élément durcissant à chaud mais qui dégrade la formabilité :
- A cet effet sa teneur doit être comprise entre 0,005
% et 0,05% en poids. - Afin d'éviter les problèmes de forgeabilité à chaud, la teneur en étain doit être inférieure ou égale à 0,05%.
- For this purpose its content must be between 0.005% and 0.05% by weight.
- In order to avoid hot forgeability problems, the tin content must be less than or equal to 0.05%.
Selon l'invention, la taille moyenne de grain de l'acier dans l'état de livraison est comprise entre 10 et 60 micromètres, la précipitation ultérieure de composés intermétalliques permettant aussi de stabiliser la taille de grain lors de l'utilisation. Une taille de grain inférieure à 10 micromètres a un effet néfaste sur le fluage intergranulaire. Une taille de grain supérieure à 60 micromètres va conduire à l'apparition d'irrégularités de surface inesthétiques, ou « peau d'orange », lors de la mise en forme à la température ambiante.According to the invention, the average grain size of the steel in the delivery state is between 10 and 60 micrometers, the subsequent precipitation of intermetallic compounds also making it possible to stabilize the grain size during use. A grain size of less than 10 microns has a detrimental effect on intergranular creep. A grain size greater than 60 microns will lead to the appearance of unsightly surface irregularities, or "orange peel", when shaping at room temperature.
Les aciers selon l'invention comportent une précipitation intergranulaire de composés Fe2Nb3 de structure cubique, après un traitement thermique compris entre 650°C et 1050°C pendant un temps supérieur à 30minutes. Selon l'invention, les précipités Fe2Nb3 sont très majoritaires parmi les précipités intergranulaires, c'est-à-dire représentent plus de 80% de la population intergranulaire. La nature et la répartition de ces précipités sont très favorables pour résister au fluage, en comparaison de précipités Fe2Nb, ou phases de Laves. Les composés Fe2Nb qui précipitent sous forme intra- ou intergranulaire ne sont stables que jusqu'à 950°C, contrairement aux précipités Fe2Nb3 stables jusqu'à 1050°C.The steels according to the invention comprise an intergranular precipitation of Fe 2 Nb 3 compounds of cubic structure, after a heat treatment of between 650 ° C. and 1050 ° C. for a time greater than 30 minutes. According to the invention, the Fe 2 Nb 3 precipitates are very much in the majority of the intergranular precipitates, that is to say they represent more than 80% of the intergranular population. The nature and distribution of these precipitates are very favorable to resist creep, in comparison with Fe 2 Nb precipitates, or Laves phases. The Fe 2 Nb compounds which precipitate in intra- or intergranular form are stable only up to 950 ° C., unlike stable Fe 2 Nb 3 precipitates up to 1050 ° C.
Selon l'invention, la structure de l'acier dans l'état de livraison est totalement recristallisée : de la sorte, la précipitation ultérieure des composés Fe2Nb3 intervient de façon très homogène.According to the invention, the structure of the steel in the delivery state is completely recrystallized: in this way, the Subsequent precipitation of Fe 2 Nb 3 compounds occurs in a very homogeneous manner.
Les inventeurs ont mis en évidence que l'efficacité des composés Fe2Nb3 cubiques était particulièrement accrue lorsque la fraction linéaire de joints de grains ferritiques qui présentaient une précipitation de ces composés, était supérieure ou égale à 5%. La définition de cette fraction linéaire f est donnée à la
L'expression f traduit donc le taux de recouvrement des joints de grains ferritiques par une précipitation de Fe2Nb3 cubiques. Les inventeurs ont mis en évidence, comme le présente la
La mise en oeuvre du procédé de fabrication d'une tôle d'acier inoxydable ferritique selon l'invention pourra notamment être la suivante :
- On approvisionne un acier de composition selon l'invention
- On procède à la coulée d'un demi-produit à partir de cet acier. Cette coulée peut être réalisée en lingot ou encore sous forme de brame produite par coulée continue (épaisseur allant généralement de quelques dizaines de millimètres pour les brames minces à quelques centaines de millimètres pour les brames classiques) ou de bandes minces entre cylindres d'acier contrarotatifs. Les demi-produits coulés sont tout d'abord portés à une température supérieure à 1000°C pour atteindre en tout point une température favorable aux déformations élevées que va subir l'acier lors du laminage à chaud.
- A composition steel is supplied according to the invention
- A semi-finished product is cast from this steel. This casting can be carried out in ingot or in the form of slab produced by continuous casting (thickness generally ranging from a few tens of millimeters for thin slabs to a few hundred millimeters for conventional slabs) or thin strips between contrarotating steel rolls . The cast semifinished products are first brought to a temperature above 1000 ° C to reach at any point a temperature favorable to the high deformations that will undergo the steel during hot rolling.
On décape puis on lamine ensuite la tôle dans les conditions usuelles, en appliquant par exemple un taux de réduction de 30 à 90%. On recuit ensuite la tôle laminée à froid à une température TR et pendant une durée tR.It is then scoured and the sheet is then rolled under the usual conditions, for example by applying a reduction ratio of 30 to 90%. The cold-rolled sheet is then annealed at a temperature T R and for a time t R.
Ces paramètres TR et tR sont choisis de telle sorte que l'on obtienne une recristallisation complète avec une taille moyenne de grain ferritique comprise entre 10 et 60 micromètres. Un accroissement de TR et de tR augmente le taux de recristallisation ainsi que la taille moyenne de grain. Une température TR comprise entre 1030 et 1130°C et un temps tR compris entre 10 secondes et 3 minutes permettent d'obtenir simultanément une recristallisation complète et une taille moyenne de grain ferritique comprise entre 10 et 60 micromètres.These parameters T R and t R are chosen such that a complete recrystallization with an average ferritic grain size of between 10 and 60 microns is obtained. An increase in T R and t R increases the recrystallization rate as well as the average grain size. A temperature T R of between 1030 and 1130 ° C. and a time t R of between 10 seconds and 3 minutes make it possible simultaneously to obtain a complete recrystallization and a mean ferritic grain size of between 10 and 60 microns.
A ce stade, la tôle d'acier est à l'état de livraison. Une pièce peut être alors fabriquée à partir de cette tôle d'acier en mettant en oeuvre des modes usuels de déformation, tels qu'emboutissage, hydroformage ou pliage. Lorsque l'on met en oeuvre la tôle d'acier à l'état de livraison avec une taille de grain ferritique comprise entre 10 et 60 micromètres, celle-ci présente simultanément une bonne résistance à la formation d'irrégularités de surface lors d'une mise en forme à froid et une bonne résistance au fluage lors de son utilisation à haute température.At this point, the steel sheet is in the delivery condition. A part can then be manufactured from this sheet steel by implementing common modes of deformation, such as stamping, hydroforming or folding. When the sheet steel is used in the delivery state with a ferritic grain size of between 10 and 60 micrometers, it simultaneously has good resistance to the formation of surface irregularities when cold forming and good creep resistance when used at high temperatures.
Lors de la mise en service ultérieure de la pièce, un ou plusieurs cycles thermiques dans un domaine de températures comprises entre 650 et 1050°C pendant une durée cumulée supérieure à 30 minutes, conduisent à une précipitation de Fe2Nb3 et un accroissement de la résistance au fluage.During the subsequent commissioning of the part, one or more thermal cycles in a temperature range between 650 and 1050 ° C for a cumulative time greater than 30 minutes lead to a precipitation of Fe 2 Nb 3 and an increase of creep resistance.
Cette résistance est particulièrement élevée lorsque la fraction linéaire f de joints de grains ferritiques comportant une précipitation des composés Fe2Nb3 est supérieure ou égale à 5%.This resistance is particularly high when the linear fraction f of ferritic grain boundaries comprising a precipitation of Fe 2 Nb 3 compounds is greater than or equal to 5%.
A titre d'exemple non limitatif, les résultats suivants vont montrer les caractéristiques avantageuses conférées par l'invention.By way of non-limiting example, the following results will show the advantageous characteristics conferred by the invention.
On a élaboré des aciers dont la composition exprimée en pourcentage pondéral, figure au tableau 1 ci-dessous. Outre les aciers I1 à I4 selon l'invention, on a indiqué à titre de comparaison la composition d'aciers de référence R1 à R6. On a également porté au tableau 2, la valeur des expressions : Ti + AI + Zr, Cr + 5 Si, Mo+3 (Nb- 7C- 7N), V +10 Ti, pour chacune des compositions ci-dessus.Steels were developed whose composition, expressed as a percentage by weight, is shown in Table 1 below. In addition to the steels I1 to I4 according to the invention, the composition of reference steels R1 to R6 has been indicated for comparison. The value of the expressions: Ti + Al + Zr, Cr + 5 Si, Mo + 3 (Nb-7C-7N), V +10 Ti, for each of the above compositions is also shown in Table 2.
Après coulée, ces aciers ont été réchauffés à une température supérieure à 1000°C, laminés à chaud jusqu'à une épaisseur de 3mm, décapés puis laminés à froid jusqu'à une épaisseur de 1,5mm. Les tôles d'acier ont été ensuite recuites à une température de 1100°C pendant une durée de 30 secondes.
Valeurs soulignées : non conformes à l'invention
Valeurs soulignées : non conformes à l'invention
Underlined values: not in accordance with the invention
Underlined values: not in accordance with the invention
Le tableau 3 présente le résultat d'un certain nombre d'essais effectués à hautes températures sur ces aciers ou d'observations réalisées après ces cycles à hautes températures. Ces essais sont destinés à apprécier le comportement mécanique, particulièrement dans des conditions d'utilisation à température supérieure ou égale à 950°C :
- après avoir soumis les tôles d'aciers à une température de 1000°C pendant une durée de 100h, on a examiné l'état de précipitation par Microscopie Electronique en Transmission. Des analyses par spectrométrie à dispersion d'énergie (EDS) ont été effectuées pour déterminer les éléments composant ces précipités. Des clichés de diffraction électronique (
figure 4 ) ont été comparés à des clichés théoriques selon l'axe de zone (figure 5 ) - On a effectué des essais de traction mécanique à 950°C et 1000°C selon la norme ASTM E21-92, et mesuré la résistance mécanique Rm. La tenue mécanique à chaud est considérée comme insuffisante lorsque Rm est inférieure à 18 MPa à 950°C ou lorsque Rm est inférieure à 10MPa à 1000°C.
- La résistance à la fatigue thermique a été évaluée grâce un test consistant à soumettre une éprouvette bridée de 1,5mm d'épaisseur à un cyclage thermique dont les températures minimale et maximale sont égales à 100°C et 1000°C. On mesure le nombre de cycles jusqu'à rupture. Le résultat est jugé satisfaisant lorsque le nombre de cycles à rupture dépasse 3500.
- La tenue au fluage a été mesurée au moyen d'un essai, dit «sag test » : un échantillon de 1,5mm d'épaisseur posé sur deux appuis ponctuels écartés de 200 mm est porté à 1000°C. On mesure la flèche après 100 h de maintien en température. Le résultat est considéré comme non satisfaisant lorsque la flèche excède 10 mm.
- La résistance à l'oxydation cyclique a été mesurée par la perte de masse, après enlèvement de l'oxyde, d'une éprouvette après 600 cycles entre la température ambiante et 1000°C, le cycle comprenant un temps de maintien de 20 minutes à 1000°C pour une durée totale de cycle de 30min. Le résultat est insuffisant lorsque la perte de masse est supérieure à 40 g/cm2.
- after subjecting the steel sheets to a temperature of 1000 ° C for a period of 100h, the precipitation state was examined by Transmission Electron Microscopy. Energy dispersive spectrometry (EDS) analyzes were performed to determine the elements composing these precipitates. Electron diffraction patterns (
figure 4 ) were compared with theoretical views along the zone axis (figure 5 ) - Mechanical tensile tests were conducted at 950 ° C and 1000 ° C according to ASTM E21-92, and the mechanical strength R m was measured. The mechanical resistance to heat is considered insufficient when R m is less than 18 MPa at 950 ° C or when R m is less than 10 MPa at 1000 ° C.
- The thermal fatigue resistance was evaluated by means of a test consisting in subjecting a 1.5 mm thick flanged specimen to thermal cycling whose minimum and maximum temperatures are equal to 100 ° C. and 1000 ° C. The number of cycles is measured until failure. The result is considered satisfactory when the number of cycles to rupture exceeds 3500.
- The creep resistance was measured by means of a so-called "sag test" test: a 1.5 mm thick sample placed on two point supports spaced apart by 200 mm is brought to 1000 ° C. The arrow is measured after 100 hours of temperature maintenance. The result is considered unsatisfactory when the arrow exceeds 10 mm.
- The resistance to cyclic oxidation was measured by the mass loss, after removal of the oxide, of a test piece after 600 cycles between room temperature and 1000 ° C., the cycle comprising a holding time of 20 minutes at 1000 ° C for a total cycle time of 30min. The result is insufficient when the loss of mass is greater than 40 g / cm 2 .
Dans les aciers selon l'invention, le recristallisation est complète et la taille moyenne de grain ferritique est comprise entre 10 et 60 micromètres.In the steels according to the invention, the recrystallization is complete and the average ferritic grain size is between 10 and 60 micrometers.
La
Par comparaison, la
Les aciers I1 à I4 selon l'invention combinent de bonnes propriétés mécaniques à chaud : résistance mécanique, résistance au fluage, à la fatigue thermique et à l'oxydation cyclique.The steels I1 to I4 according to the invention combine good mechanical properties when hot: mechanical resistance, resistance to creep, thermal fatigue and cyclic oxidation.
Les aciers R1 et R2 présentent une combinaison de titane et de vanadium insuffisante : les résistances à l'oxydation cyclique et à la fatigue thermique ne sont pas satisfaisantes en raison du manque d'adhésion des couches d'oxydes avec l'acier substrat.The R1 and R2 steels have a combination of insufficient titanium and vanadium: the resistances to cyclic oxidation and thermal fatigue are unsatisfactory due to the lack of adhesion of the oxide layers to the substrate steel.
Dans les aciers R3, R4 et R5 de référence, les teneurs en Ti ou en Zr, en Ti+AI+Zr sont excessives, ce qui ne permet pas la précipitation du niobium sous forme de Fe2Nb3 : c'est au contraire Fe2Nb qui précipite vers 650-800°C lors de l'utilisation à haute température. Ces précipités relativement grossiers, présents en faible quantité aux joints de grains et totalement remis en solution à des températures supérieures à 950°C, sont peu efficaces pour améliorer la tenue au fluage. Par comparaison, les précipités Fe2Nb3 des aciers selon l'invention sont plus stables, même au delà de 950°C. Corrélativement la tenue au fluage et la résistance à l'oxydation cyclique des aciers R3 à R5 sont insuffisantes.In reference steels R3, R4 and R5, the contents of Ti or Zr, Ti + Al + Zr are excessive, which does not allow the precipitation of niobium as Fe 2 Nb3: it is on the contrary Fe 2 Nb that precipitates around 650-800 ° C during use at high temperature. These relatively coarse precipitates, present in small amounts at the grain boundaries and totally redissolved at temperatures above 950 ° C, are not very effective in improving the creep resistance. In comparison, the Fe 2 Nb 3 precipitates of the steels according to the invention are more stable, even beyond 950 ° C. Correlatively, the creep resistance and the cyclic oxidation resistance of the R3 to R5 steels are insufficient.
L'acier R5 a également une teneur excessive en titane et une combinaison (Ti+Al+Zr) non satisfaisante. De plus, sa combinaison : Mo+3ΔNb, est insuffisante. En conséquence, l'acier ne présente pas de propriétés mécaniques satisfaisantes à haute température, en particulier de fluage.R5 steel also has an excessive titanium content and a combination (Ti + Al + Zr) unsatisfactory. Moreover, its combination: Mo + 3ΔNb, is insufficient. As a result, the steel does not exhibit satisfactory mechanical properties at high temperature, in particular creep.
L'acier R6 présente une teneur insuffisante en chrome ainsi que des combinaisons : Mo+3ΔNb, Cr+5%Si insuffisantes. En dépit de la présence de composés Fe2Nb3, les propriétés de résistance à l'oxydation et les propriétés mécaniques à haute température sont insuffisantes.R6 steel has an insufficient chromium content as well as combinations: Mo + 3ΔNb, Cr + 5% Si insufficient. Despite the presence of Fe 2 Nb 3 compounds, the properties of oxidation resistance and high temperature mechanical properties are insufficient.
Les aciers selon l'invention seront utilisés avec profit pour la fabrication de pièces soumises à une température d'utilisation périodique supérieure à 950°C, et notamment de collecteurs d'échappement de gaz de combustion dans le domaine automobile, de brûleurs, d'échangeurs à chaleur ou d'enveloppes de turbocompresseurs, de chaudières.The steels according to the invention will be used with advantage for the manufacture of parts subjected to a temperature periodic use greater than 950 ° C, and in particular exhaust gas collectors in the automotive field, burners, heat exchangers or envelopes turbochargers, boilers.
Claims (6)
- Ferritic stainless steel sheet, the composition of which comprises, the contents being expressed by weight:C ≤ 0.03%;Mn ≤ 1 %;0.3 ≤ Si ≤ 1%;S ≤ 0.01% ;P ≤ 0.04% ;18% ≤ Cr ≤ 22%;Ni ≤ 0.5% ;Mo ≤ 2.5% ;Cu ≤ 0.5% ;Ti ≤ 0.02%;Zr ≤ 0.02% ;Al ≤ 0.02% ;0.2% ≤ Nb ≤ 1% ;V ≤ 0.2% ;N ≤ 0.03% ;0.005% ≤ Co ≤ 0.05% ;Sn ≤ 0.05%;it being understood that:Ti + Al + Zr ≤ 0.030%;Cr + 5 Si ≥ 20%;Mo + 3 (Nb-7C-7N) ≥ 1.5%;V + 10 Ti ≥ 0.06%,the remainder of the composition being composed of iron and inevitable impurities resulting from the smelting, of which the structure is completely recrystallized and the average ferritic grain size is between 10 and 60 micrometres.
- Part manufactured from a steel sheet according to Claim 1, characterized in that it contains an intergranular precipitation comprising at least 80% of cubic Fe2Nb3 compounds.
- Part according to Claim 2, characterized in that the linear fraction f of ferritic grain boundaries having a precipitation of cubic Fe2Nb3 compounds is greater than or equal to 5%, the fraction "f" is defined by
denotes the total length of the grain boundaries comprising Fe2Nb3 precipitates relative to the surface (S) in question and - Process for manufacturing a ferritic stainless steel sheet, according to which:- a steel of composition according to Claim 1 is provided;- a semi-finished product is then cast from this steel;- said semi-finished product is brought to a temperature above 1000°C;- said semi-finished product is hot-rolled to obtain a hot-rolled sheet;- said sheet is cold-rolled; and- said cold-rolled sheet is annealed at a temperature TR between 1030 and 1130°C for a time tR between 10 seconds and 3 minutes, in order to obtain a completely recrystallized structure with a ferritic grain size between 10 and 60 micrometres.
- Process for manufacturing a part according to which a ferritic stainless steel sheet manufactured according to Claim 4 is provided, then:- said sheet is formed in order to obtain a part; then- said part is subjected to one or more heat cycles in a temperature range between 650 and 1050°C for a combined time greater than 30 minutes.
- Use of the steel sheet according to either one of Claims 1 or 3, or manufactured by a process according to Claim 4, for manufacturing parts subjected to a periodic operating temperature above 950°C, and especially combustion gas exhaust manifolds in the automotive field, burners, heat exchangers, turbocharger housings and boilers.
Priority Applications (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| SI200730015T SI1818422T2 (en) | 2006-02-08 | 2007-01-11 | Ferritic stainless steel with 19% of chromium stabilised with niobium |
| EP07290039A EP1818422B2 (en) | 2006-02-08 | 2007-01-11 | Ferritic stainless steel with 19% of chromium stabilised with niobium |
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| EP06290231A EP1818421A1 (en) | 2006-02-08 | 2006-02-08 | Ferritic, niobium-stabilised 19% chromium stainless steel |
| EP07290039A EP1818422B2 (en) | 2006-02-08 | 2007-01-11 | Ferritic stainless steel with 19% of chromium stabilised with niobium |
Publications (3)
| Publication Number | Publication Date |
|---|---|
| EP1818422A1 EP1818422A1 (en) | 2007-08-15 |
| EP1818422B1 EP1818422B1 (en) | 2008-12-10 |
| EP1818422B2 true EP1818422B2 (en) | 2012-07-18 |
Family
ID=36691479
Family Applications (2)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| EP06290231A Withdrawn EP1818421A1 (en) | 2006-02-08 | 2006-02-08 | Ferritic, niobium-stabilised 19% chromium stainless steel |
| EP07290039A Active EP1818422B2 (en) | 2006-02-08 | 2007-01-11 | Ferritic stainless steel with 19% of chromium stabilised with niobium |
Family Applications Before (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| EP06290231A Withdrawn EP1818421A1 (en) | 2006-02-08 | 2006-02-08 | Ferritic, niobium-stabilised 19% chromium stainless steel |
Country Status (7)
| Country | Link |
|---|---|
| EP (2) | EP1818421A1 (en) |
| AT (1) | ATE417134T1 (en) |
| DE (1) | DE602007000326D1 (en) |
| DK (1) | DK1818422T4 (en) |
| ES (1) | ES2317629T5 (en) |
| PT (1) | PT1818422E (en) |
| SI (1) | SI1818422T2 (en) |
Families Citing this family (12)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2013179616A1 (en) * | 2012-05-28 | 2013-12-05 | Jfeスチール株式会社 | Ferritic stainless steel |
| IN2015DN01710A (en) | 2012-09-03 | 2015-05-22 | Aperam Stainless France | |
| FI124995B (en) | 2012-11-20 | 2015-04-15 | Outokumpu Oy | Ferritic stainless steel |
| JP6159775B2 (en) * | 2014-10-31 | 2017-07-05 | 新日鐵住金ステンレス株式会社 | Ferritic stainless steel with excellent resistance to exhaust gas condensate corrosion and brazing, and method for producing the same |
| EP3214198B1 (en) * | 2014-10-31 | 2022-06-01 | NIPPON STEEL Stainless Steel Corporation | Ferrite-based stainless steel with high resistance to corrosiveness caused by exhaust gas and condensation and high brazing properties and method for manufacturing same |
| KR102206415B1 (en) * | 2016-09-02 | 2021-01-22 | 제이에프이 스틸 가부시키가이샤 | Ferritic stainless steel |
| KR102400403B1 (en) * | 2016-12-21 | 2022-05-23 | 제이에프이 스틸 가부시키가이샤 | Ferritic stainless steel |
| US20210032731A1 (en) * | 2018-01-31 | 2021-02-04 | Jfe Steel Corporation | Ferritic stainless steel |
| SI3670692T1 (en) | 2018-12-21 | 2022-11-30 | Outokumpu Oyj, | Ferritic stainless steel |
| KR102259806B1 (en) * | 2019-08-05 | 2021-06-03 | 주식회사 포스코 | Ferritic stainless steel with improved creep resistance at high temperature and method for manufacturing the ferritic stainless steel |
| CN112375995B (en) * | 2021-01-15 | 2021-05-07 | 江苏省沙钢钢铁研究院有限公司 | 400 MPa-grade corrosion-resistant steel bar and production method thereof |
| CN116479330A (en) * | 2023-04-27 | 2023-07-25 | 山西常达精密金属科技有限公司 | Ultra-pure ferrite stainless steel material for welding automobile exhaust system and preparation thereof |
Citations (2)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JPH04224657A (en) † | 1990-12-26 | 1992-08-13 | Kawasaki Steel Corp | Ferritic stainless steel excellent in strength at high temperature and toughness in weld heat-affected zone |
| JPH04280948A (en) † | 1991-03-08 | 1992-10-06 | Nippon Yakin Kogyo Co Ltd | Ferritic stainless steel excellent in tougness and corrosion resistance |
Family Cites Families (5)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US4010049A (en) * | 1975-10-06 | 1977-03-01 | Jones & Laughlin Steel Corporation | Columbium-stabilized high chromium ferritic stainless steels containing zirconium |
| FR2589482B1 (en) * | 1985-11-05 | 1987-11-27 | Ugine Gueugnon Sa | STAINLESS STEEL FERRITIC STEEL SHEET OR STRIP, ESPECIALLY FOR EXHAUST SYSTEMS |
| JP2696584B2 (en) * | 1990-03-24 | 1998-01-14 | 日新製鋼株式会社 | Ferrite heat-resistant stainless steel with excellent low-temperature toughness, weldability and heat resistance |
| FR2798394B1 (en) * | 1999-09-09 | 2001-10-26 | Ugine Sa | FERRITIC STEEL WITH 14% CHROMIUM STABILIZED IN NIOBIUM AND ITS USE IN THE AUTOMOTIVE FIELD |
| US6426039B2 (en) * | 2000-07-04 | 2002-07-30 | Kawasaki Steel Corporation | Ferritic stainless steel |
-
2006
- 2006-02-08 EP EP06290231A patent/EP1818421A1/en not_active Withdrawn
-
2007
- 2007-01-11 DK DK07290039.2T patent/DK1818422T4/en active
- 2007-01-11 EP EP07290039A patent/EP1818422B2/en active Active
- 2007-01-11 SI SI200730015T patent/SI1818422T2/en unknown
- 2007-01-11 AT AT07290039T patent/ATE417134T1/en active
- 2007-01-11 PT PT07290039T patent/PT1818422E/en unknown
- 2007-01-11 ES ES07290039T patent/ES2317629T5/en active Active
- 2007-01-11 DE DE602007000326T patent/DE602007000326D1/en active Active
Patent Citations (2)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JPH04224657A (en) † | 1990-12-26 | 1992-08-13 | Kawasaki Steel Corp | Ferritic stainless steel excellent in strength at high temperature and toughness in weld heat-affected zone |
| JPH04280948A (en) † | 1991-03-08 | 1992-10-06 | Nippon Yakin Kogyo Co Ltd | Ferritic stainless steel excellent in tougness and corrosion resistance |
Non-Patent Citations (1)
| Title |
|---|
| J.H. Schmitt et al: "Some Recent Trends in Niobium Ferritic Stainless Steels", Proceedings of Symposium at the occasion of the 30 years anniversary of Niobium Products Co. GmbH, Düsseldorf, 20-05-2005. † |
Also Published As
| Publication number | Publication date |
|---|---|
| EP1818422B1 (en) | 2008-12-10 |
| DK1818422T3 (en) | 2009-02-23 |
| DE602007000326D1 (en) | 2009-01-22 |
| SI1818422T1 (en) | 2009-04-30 |
| EP1818422A1 (en) | 2007-08-15 |
| EP1818421A1 (en) | 2007-08-15 |
| DK1818422T4 (en) | 2012-10-29 |
| PT1818422E (en) | 2009-01-30 |
| ES2317629T3 (en) | 2009-04-16 |
| SI1818422T2 (en) | 2012-11-30 |
| ATE417134T1 (en) | 2008-12-15 |
| ES2317629T5 (en) | 2012-12-26 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| EP1818422B2 (en) | Ferritic stainless steel with 19% of chromium stabilised with niobium | |
| US6478897B1 (en) | Heat-resistant alloy wire | |
| EP1819461B1 (en) | Method of producing austentic iron/carbon/manganese steel sheets having very high strength and elongation characteristics and excellent homogeneity | |
| EP1844173B1 (en) | Method for producing austenitic iron-carbon-manganese metal sheets, and sheets produced thereby | |
| EP1734143B1 (en) | Ferritic stainless steel sheet excellent in formability and method for production thereof | |
| KR101256268B1 (en) | Austenitic stainless steel | |
| EP1867740B1 (en) | Low thermal expansion Ni-base superalloy | |
| JP5353501B2 (en) | High temperature hydrogen gas storage steel container having excellent hydrogen resistance and method for producing the same | |
| WO2008145872A1 (en) | Low density steel with good stamping capability | |
| FR2876708A1 (en) | PROCESS FOR MANUFACTURING COLD-ROLLED CARBON-MANGANESE AUSTENITIC STEEL TILES WITH HIGH CORROSION RESISTANT MECHANICAL CHARACTERISTICS AND SHEETS THUS PRODUCED | |
| US20130004360A1 (en) | Ferritic stainless steel sheet excellent in oxidation resistance and ferritic stainless steel sheet excellent in heat resistance and method of production of same | |
| JPH11302801A (en) | High chromium-high nickel alloy excellent in stress corrosion cracking resistance | |
| WO2019045001A1 (en) | Alloy plate and gasket | |
| CN114502760A (en) | Ferritic stainless steel sheet, method for producing same, and ferritic stainless steel member | |
| JP2004277860A (en) | Heat-resistant alloy for high-strength exhaust valve excellent in overaging resistance | |
| EP1083241B1 (en) | Ferritic niobium-stabilised 14% chromium steel and its use in the car industry | |
| EP2257652B1 (en) | Method of manufacturing sheets of austenitic stainless steel with high mechanical properties | |
| JP7341016B2 (en) | Ferritic stainless cold rolled steel sheet | |
| CN115198144B (en) | Heat-resistant alloy member, material used therefor, and method for producing same | |
| JP2020132919A (en) | Heat-resistant alloy and method for producing the same | |
| CN1093887C (en) | Austenitic stainless steel with good oxidation resistance | |
| JP4078881B2 (en) | Austenitic stainless steel sheet for heat exchanger | |
| FR2864108A1 (en) | Stainless steel with high mechanical strength and good elongation with an austenitic microstructure and limited martensite pockets for the fabrication of motor vehicle structural components | |
| JP2021080541A (en) | Heat-resistant alloy | |
| JPH11199987A (en) | Heat resistant alloy suitable for cold working |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| PUAI | Public reference made under article 153(3) epc to a published international application that has entered the european phase |
Free format text: ORIGINAL CODE: 0009012 |
|
| AK | Designated contracting states |
Kind code of ref document: A1 Designated state(s): AT BE BG CH CY CZ DE DK EE ES FI FR GB GR HU IE IS IT LI LT LU LV MC NL PL PT RO SE SI SK TR |
|
| AX | Request for extension of the european patent |
Extension state: AL BA HR MK YU |
|
| 17P | Request for examination filed |
Effective date: 20070914 |
|
| 17Q | First examination report despatched |
Effective date: 20071024 |
|
| AKX | Designation fees paid | ||
| RAX | Requested extension states of the european patent have changed |
Extension state: HR Payment date: 20080215 Extension state: MK Payment date: 20080215 Extension state: AL Payment date: 20080215 Extension state: BA Payment date: 20080215 |
|
| RBV | Designated contracting states (corrected) |
Designated state(s): AT BE BG CH CY CZ DE DK EE ES FI FR GB GR HU IE IS IT LI LT LU LV MC NL PL PT RO SE SI SK TR |
|
| RAX | Requested extension states of the european patent have changed |
Extension state: AL Payment date: 20080215 Extension state: HR Payment date: 20080215 Extension state: RS Payment date: 20080215 Extension state: BA Payment date: 20080215 Extension state: MK Payment date: 20080215 |
|
| REG | Reference to a national code |
Ref country code: DE Ref legal event code: 8566 |
|
| GRAP | Despatch of communication of intention to grant a patent |
Free format text: ORIGINAL CODE: EPIDOSNIGR1 |
|
| GRAS | Grant fee paid |
Free format text: ORIGINAL CODE: EPIDOSNIGR3 |
|
| GRAA | (expected) grant |
Free format text: ORIGINAL CODE: 0009210 |
|
| RAP1 | Party data changed (applicant data changed or rights of an application transferred) |
Owner name: ARCELORMITTAL-STAINLESS FRANCE |
|
| AK | Designated contracting states |
Kind code of ref document: B1 Designated state(s): AT BE BG CH CY CZ DE DK EE ES FI FR GB GR HU IE IS IT LI LT LU LV MC NL PL PT RO SE SI SK TR |
|
| AX | Request for extension of the european patent |
Extension state: AL BA HR MK RS |
|
| REG | Reference to a national code |
Ref country code: GB Ref legal event code: FG4D Free format text: NOT ENGLISH |
|
| REG | Reference to a national code |
Ref country code: CH Ref legal event code: EP |
|
| REG | Reference to a national code |
Ref country code: IE Ref legal event code: FG4D Free format text: LANGUAGE OF EP DOCUMENT: FRENCH |
|
| REF | Corresponds to: |
Ref document number: 602007000326 Country of ref document: DE Date of ref document: 20090122 Kind code of ref document: P |
|
| REG | Reference to a national code |
Ref country code: PT Ref legal event code: SC4A Free format text: AVAILABILITY OF NATIONAL TRANSLATION Effective date: 20090119 |
|
| REG | Reference to a national code |
Ref country code: DK Ref legal event code: T3 |
|
| REG | Reference to a national code |
Ref country code: SE Ref legal event code: TRGR |
|
| REG | Reference to a national code |
Ref country code: GR Ref legal event code: EP Ref document number: 20090400724 Country of ref document: GR |
|
| REG | Reference to a national code |
Ref country code: ES Ref legal event code: FG2A Ref document number: 2317629 Country of ref document: ES Kind code of ref document: T3 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: LT Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20081210 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: LV Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20081210 Ref country code: PL Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20081210 |
|
| REG | Reference to a national code |
Ref country code: IE Ref legal event code: FD4D |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: IE Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20081210 Ref country code: EE Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20081210 Ref country code: BG Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20090310 Ref country code: RO Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20081210 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: MC Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20090131 Ref country code: CZ Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20081210 Ref country code: IS Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20090410 |
|
| PLBI | Opposition filed |
Free format text: ORIGINAL CODE: 0009260 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: SK Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20081210 |
|
| PLAX | Notice of opposition and request to file observation + time limit sent |
Free format text: ORIGINAL CODE: EPIDOSNOBS2 |
|
| 26 | Opposition filed |
Opponent name: THYSSENKRUPP NIROSTA AG Effective date: 20090910 |
|
| NLR1 | Nl: opposition has been filed with the epo |
Opponent name: THYSSENKRUPP NIROSTA AG |
|
| PLAF | Information modified related to communication of a notice of opposition and request to file observations + time limit |
Free format text: ORIGINAL CODE: EPIDOSCOBS2 |
|
| PLBB | Reply of patent proprietor to notice(s) of opposition received |
Free format text: ORIGINAL CODE: EPIDOSNOBS3 |
|
| PGRI | Patent reinstated in contracting state [announced from national office to epo] |
Ref country code: IT Effective date: 20110501 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: HU Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20090611 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: CY Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20081210 |
|
| PUAH | Patent maintained in amended form |
Free format text: ORIGINAL CODE: 0009272 |
|
| STAA | Information on the status of an ep patent application or granted ep patent |
Free format text: STATUS: PATENT MAINTAINED AS AMENDED |
|
| 27A | Patent maintained in amended form |
Effective date: 20120718 |
|
| AK | Designated contracting states |
Kind code of ref document: B2 Designated state(s): AT BE BG CH CY CZ DE DK EE ES FI FR GB GR HU IE IS IT LI LT LU LV MC NL PL PT RO SE SI SK TR |
|
| AX | Request for extension of the european patent |
Extension state: AL BA HR MK RS |
|
| REG | Reference to a national code |
Ref country code: CH Ref legal event code: AEN Free format text: BREVET MAINTENU DANS UNE FORME MODIFIEE |
|
| REG | Reference to a national code |
Ref country code: DE Ref legal event code: R102 Ref document number: 602007000326 Country of ref document: DE Effective date: 20120718 |
|
| REG | Reference to a national code |
Ref country code: DK Ref legal event code: T4 |
|
| REG | Reference to a national code |
Ref country code: SE Ref legal event code: RPEO |
|
| REG | Reference to a national code |
Ref country code: NL Ref legal event code: T3 |
|
| REG | Reference to a national code |
Ref country code: ES Ref legal event code: DC2A Ref document number: 2317629 Country of ref document: ES Kind code of ref document: T5 Effective date: 20121226 |
|
| REG | Reference to a national code |
Ref country code: GR Ref legal event code: EP Ref document number: 20120402355 Country of ref document: GR Effective date: 20121122 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: LV Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20120718 |
|
| PGFP | Annual fee paid to national office [announced via postgrant information from national office to epo] |
Ref country code: LU Payment date: 20141218 Year of fee payment: 9 Ref country code: DK Payment date: 20141222 Year of fee payment: 9 Ref country code: GR Payment date: 20141217 Year of fee payment: 9 |
|
| PGFP | Annual fee paid to national office [announced via postgrant information from national office to epo] |
Ref country code: NL Payment date: 20141218 Year of fee payment: 9 Ref country code: PT Payment date: 20141217 Year of fee payment: 9 |
|
| PGFP | Annual fee paid to national office [announced via postgrant information from national office to epo] |
Ref country code: SI Payment date: 20141217 Year of fee payment: 9 |
|
| REG | Reference to a national code |
Ref country code: FR Ref legal event code: PLFP Year of fee payment: 10 |
|
| REG | Reference to a national code |
Ref country code: DK Ref legal event code: EBP Effective date: 20160131 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: LU Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20160111 |
|
| REG | Reference to a national code |
Ref country code: GR Ref legal event code: ML Ref document number: 20120402355 Country of ref document: GR Effective date: 20160803 |
|
| REG | Reference to a national code |
Ref country code: NL Ref legal event code: MM Effective date: 20160201 |
|
| REG | Reference to a national code |
Ref country code: SI Ref legal event code: KO00 Effective date: 20160906 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: NL Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20160201 Ref country code: GR Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20160803 Ref country code: PT Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20160711 Ref country code: SI Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20160112 |
|
| REG | Reference to a national code |
Ref country code: FR Ref legal event code: PLFP Year of fee payment: 11 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: DK Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20160131 |
|
| REG | Reference to a national code |
Ref country code: FR Ref legal event code: PLFP Year of fee payment: 12 |
|
| P01 | Opt-out of the competence of the unified patent court (upc) registered |
Effective date: 20230524 |
|
| PGFP | Annual fee paid to national office [announced via postgrant information from national office to epo] |
Ref country code: FR Payment date: 20231211 Year of fee payment: 18 Ref country code: FI Payment date: 20231222 Year of fee payment: 18 |
|
| PGFP | Annual fee paid to national office [announced via postgrant information from national office to epo] |
Ref country code: ES Payment date: 20240207 Year of fee payment: 18 |
|
| PGFP | Annual fee paid to national office [announced via postgrant information from national office to epo] |
Ref country code: AT Payment date: 20231222 Year of fee payment: 18 |
|
| PGFP | Annual fee paid to national office [announced via postgrant information from national office to epo] |
Ref country code: DE Payment date: 20240115 Year of fee payment: 18 Ref country code: GB Payment date: 20240119 Year of fee payment: 18 Ref country code: CH Payment date: 20240202 Year of fee payment: 18 |
|
| PGFP | Annual fee paid to national office [announced via postgrant information from national office to epo] |
Ref country code: TR Payment date: 20240108 Year of fee payment: 18 Ref country code: SE Payment date: 20240119 Year of fee payment: 18 Ref country code: IT Payment date: 20240110 Year of fee payment: 18 Ref country code: BE Payment date: 20240116 Year of fee payment: 18 |