EP1672017B1 - Rubber compositions - Google Patents
Rubber compositions Download PDFInfo
- Publication number
- EP1672017B1 EP1672017B1 EP05112090A EP05112090A EP1672017B1 EP 1672017 B1 EP1672017 B1 EP 1672017B1 EP 05112090 A EP05112090 A EP 05112090A EP 05112090 A EP05112090 A EP 05112090A EP 1672017 B1 EP1672017 B1 EP 1672017B1
- Authority
- EP
- European Patent Office
- Prior art keywords
- rubber
- eto
- meo
- general formula
- mixture
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Active
Links
- 239000000203 mixture Substances 0.000 title claims abstract description 76
- 229920001971 elastomer Polymers 0.000 title claims abstract description 72
- 239000005060 rubber Substances 0.000 title claims abstract description 72
- -1 organosilane compound Chemical class 0.000 claims abstract description 23
- 239000000945 filler Substances 0.000 claims abstract description 12
- 125000002496 methyl group Chemical group [H]C([H])([H])* 0.000 claims abstract description 12
- 125000001495 ethyl group Chemical group [H]C([H])([H])C([H])([H])* 0.000 claims abstract description 8
- 229920006395 saturated elastomer Polymers 0.000 claims abstract description 7
- 125000001931 aliphatic group Chemical group 0.000 claims abstract description 5
- 125000003118 aryl group Chemical group 0.000 claims abstract description 5
- 125000001183 hydrocarbyl group Chemical group 0.000 claims abstract description 3
- 150000001282 organosilanes Chemical class 0.000 claims description 18
- 238000000034 method Methods 0.000 claims description 12
- 230000008569 process Effects 0.000 claims description 9
- 238000002360 preparation method Methods 0.000 claims description 6
- 238000004519 manufacturing process Methods 0.000 claims description 5
- 229920001515 polyalkylene glycol Polymers 0.000 claims description 5
- 238000013016 damping Methods 0.000 claims description 2
- 244000048879 Funtumia elastica Species 0.000 claims 1
- 125000001400 nonyl group Chemical group [H]C([*])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])[H] 0.000 claims 1
- 238000007789 sealing Methods 0.000 claims 1
- 125000001301 ethoxy group Chemical group [H]C([H])([H])C([H])([H])O* 0.000 abstract description 4
- 239000012752 auxiliary agent Substances 0.000 abstract 2
- 125000000217 alkyl group Chemical group 0.000 abstract 1
- 229910000077 silane Inorganic materials 0.000 description 21
- BLRPTPMANUNPDV-UHFFFAOYSA-N Silane Chemical compound [SiH4] BLRPTPMANUNPDV-UHFFFAOYSA-N 0.000 description 19
- LFQSCWFLJHTTHZ-UHFFFAOYSA-N Ethanol Chemical compound CCO LFQSCWFLJHTTHZ-UHFFFAOYSA-N 0.000 description 18
- 239000000047 product Substances 0.000 description 16
- 238000002156 mixing Methods 0.000 description 15
- 150000001875 compounds Chemical class 0.000 description 12
- 229920003048 styrene butadiene rubber Polymers 0.000 description 12
- VYPSYNLAJGMNEJ-UHFFFAOYSA-N Silicium dioxide Chemical compound O=[Si]=O VYPSYNLAJGMNEJ-UHFFFAOYSA-N 0.000 description 10
- YXFVVABEGXRONW-UHFFFAOYSA-N Toluene Chemical compound CC1=CC=CC=C1 YXFVVABEGXRONW-UHFFFAOYSA-N 0.000 description 9
- 230000000052 comparative effect Effects 0.000 description 9
- LYCAIKOWRPUZTN-UHFFFAOYSA-N Ethylene glycol Chemical compound OCCO LYCAIKOWRPUZTN-UHFFFAOYSA-N 0.000 description 7
- NINIDFKCEFEMDL-UHFFFAOYSA-N Sulfur Chemical compound [S] NINIDFKCEFEMDL-UHFFFAOYSA-N 0.000 description 7
- 239000003795 chemical substances by application Substances 0.000 description 7
- 239000003921 oil Substances 0.000 description 7
- 229920001451 polypropylene glycol Polymers 0.000 description 7
- 150000004756 silanes Chemical class 0.000 description 7
- 150000004760 silicates Chemical class 0.000 description 7
- 239000011593 sulfur Substances 0.000 description 7
- 229910052717 sulfur Inorganic materials 0.000 description 7
- 239000000725 suspension Substances 0.000 description 7
- 229920001223 polyethylene glycol Polymers 0.000 description 6
- 239000000243 solution Substances 0.000 description 6
- TXDNPSYEJHXKMK-UHFFFAOYSA-N sulfanylsilane Chemical class S[SiH3] TXDNPSYEJHXKMK-UHFFFAOYSA-N 0.000 description 6
- 238000012360 testing method Methods 0.000 description 6
- JOXIMZWYDAKGHI-UHFFFAOYSA-N toluene-4-sulfonic acid Chemical compound CC1=CC=C(S(O)(=O)=O)C=C1 JOXIMZWYDAKGHI-UHFFFAOYSA-N 0.000 description 6
- 238000004073 vulcanization Methods 0.000 description 6
- KAKZBPTYRLMSJV-UHFFFAOYSA-N Butadiene Chemical compound C=CC=C KAKZBPTYRLMSJV-UHFFFAOYSA-N 0.000 description 5
- PPBRXRYQALVLMV-UHFFFAOYSA-N Styrene Chemical compound C=CC1=CC=CC=C1 PPBRXRYQALVLMV-UHFFFAOYSA-N 0.000 description 5
- 238000005299 abrasion Methods 0.000 description 5
- 229920001577 copolymer Polymers 0.000 description 5
- DEQZTKGFXNUBJL-UHFFFAOYSA-N n-(1,3-benzothiazol-2-ylsulfanyl)cyclohexanamine Chemical compound C1CCCCC1NSC1=NC2=CC=CC=C2S1 DEQZTKGFXNUBJL-UHFFFAOYSA-N 0.000 description 5
- 239000000126 substance Substances 0.000 description 5
- ZQFHHQHREABVNX-UHFFFAOYSA-N triethoxy-(propyldisulfanyl)silane Chemical compound CCCSS[Si](OCC)(OCC)OCC ZQFHHQHREABVNX-UHFFFAOYSA-N 0.000 description 5
- OWRCNXZUPFZXOS-UHFFFAOYSA-N 1,3-diphenylguanidine Chemical compound C=1C=CC=CC=1NC(=N)NC1=CC=CC=C1 OWRCNXZUPFZXOS-UHFFFAOYSA-N 0.000 description 4
- IIFBEYQLKOBDQH-UHFFFAOYSA-N 3-chloropropyl-ethoxy-dimethylsilane Chemical compound CCO[Si](C)(C)CCCCl IIFBEYQLKOBDQH-UHFFFAOYSA-N 0.000 description 4
- OKTJSMMVPCPJKN-UHFFFAOYSA-N Carbon Chemical compound [C] OKTJSMMVPCPJKN-UHFFFAOYSA-N 0.000 description 4
- 239000002202 Polyethylene glycol Substances 0.000 description 4
- 235000021355 Stearic acid Nutrition 0.000 description 4
- XLOMVQKBTHCTTD-UHFFFAOYSA-N Zinc monoxide Chemical compound [Zn]=O XLOMVQKBTHCTTD-UHFFFAOYSA-N 0.000 description 4
- PNEYBMLMFCGWSK-UHFFFAOYSA-N aluminium oxide Inorganic materials [O-2].[O-2].[O-2].[Al+3].[Al+3] PNEYBMLMFCGWSK-UHFFFAOYSA-N 0.000 description 4
- 239000006229 carbon black Substances 0.000 description 4
- 235000019241 carbon black Nutrition 0.000 description 4
- YACLQRRMGMJLJV-UHFFFAOYSA-N chloroprene Chemical compound ClC(=C)C=C YACLQRRMGMJLJV-UHFFFAOYSA-N 0.000 description 4
- WITDFSFZHZYQHB-UHFFFAOYSA-N dibenzylcarbamothioylsulfanyl n,n-dibenzylcarbamodithioate Chemical compound C=1C=CC=CC=1CN(CC=1C=CC=CC=1)C(=S)SSC(=S)N(CC=1C=CC=CC=1)CC1=CC=CC=C1 WITDFSFZHZYQHB-UHFFFAOYSA-N 0.000 description 4
- 150000002430 hydrocarbons Chemical class 0.000 description 4
- QIQXTHQIDYTFRH-UHFFFAOYSA-N octadecanoic acid Chemical compound CCCCCCCCCCCCCCCCCC(O)=O QIQXTHQIDYTFRH-UHFFFAOYSA-N 0.000 description 4
- OQCDKBAXFALNLD-UHFFFAOYSA-N octadecanoic acid Natural products CCCCCCCC(C)CCCCCCCCC(O)=O OQCDKBAXFALNLD-UHFFFAOYSA-N 0.000 description 4
- 229920001748 polybutylene Polymers 0.000 description 4
- 229920000642 polymer Polymers 0.000 description 4
- 239000005077 polysulfide Substances 0.000 description 4
- 229920001021 polysulfide Polymers 0.000 description 4
- 150000008117 polysulfides Polymers 0.000 description 4
- 230000001012 protector Effects 0.000 description 4
- 239000008117 stearic acid Substances 0.000 description 4
- POAOYUHQDCAZBD-UHFFFAOYSA-N 2-butoxyethanol Chemical compound CCCCOCCO POAOYUHQDCAZBD-UHFFFAOYSA-N 0.000 description 3
- OKKJLVBELUTLKV-UHFFFAOYSA-N Methanol Chemical compound OC OKKJLVBELUTLKV-UHFFFAOYSA-N 0.000 description 3
- 239000005062 Polybutadiene Substances 0.000 description 3
- DNIAPMSPPWPWGF-UHFFFAOYSA-N Propylene glycol Chemical group CC(O)CO DNIAPMSPPWPWGF-UHFFFAOYSA-N 0.000 description 3
- 150000001343 alkyl silanes Chemical class 0.000 description 3
- 230000008878 coupling Effects 0.000 description 3
- 238000010168 coupling process Methods 0.000 description 3
- 238000005859 coupling reaction Methods 0.000 description 3
- 238000011049 filling Methods 0.000 description 3
- 239000000706 filtrate Substances 0.000 description 3
- 239000011521 glass Substances 0.000 description 3
- 229930195733 hydrocarbon Natural products 0.000 description 3
- 229910044991 metal oxide Inorganic materials 0.000 description 3
- 150000004706 metal oxides Chemical class 0.000 description 3
- 238000006116 polymerization reaction Methods 0.000 description 3
- 239000000377 silicon dioxide Substances 0.000 description 3
- 239000002904 solvent Substances 0.000 description 3
- 230000035882 stress Effects 0.000 description 3
- 239000010936 titanium Substances 0.000 description 3
- OHJYHAOODFPJOD-UHFFFAOYSA-N 2-(2-ethylhexoxy)ethanol Chemical compound CCCCC(CC)COCCO OHJYHAOODFPJOD-UHFFFAOYSA-N 0.000 description 2
- GZMAAYIALGURDQ-UHFFFAOYSA-N 2-(2-hexoxyethoxy)ethanol Chemical compound CCCCCCOCCOCCO GZMAAYIALGURDQ-UHFFFAOYSA-N 0.000 description 2
- 239000005995 Aluminium silicate Substances 0.000 description 2
- VQTUBCCKSQIDNK-UHFFFAOYSA-N Isobutene Chemical compound CC(C)=C VQTUBCCKSQIDNK-UHFFFAOYSA-N 0.000 description 2
- 239000006057 Non-nutritive feed additive Substances 0.000 description 2
- 229910020175 SiOH Inorganic materials 0.000 description 2
- 235000012211 aluminium silicate Nutrition 0.000 description 2
- 238000004458 analytical method Methods 0.000 description 2
- 238000006243 chemical reaction Methods 0.000 description 2
- 239000003153 chemical reaction reagent Substances 0.000 description 2
- 239000007822 coupling agent Substances 0.000 description 2
- 125000000118 dimethyl group Chemical group [H]C([H])([H])* 0.000 description 2
- 238000005516 engineering process Methods 0.000 description 2
- 239000012065 filter cake Substances 0.000 description 2
- 239000003365 glass fiber Substances 0.000 description 2
- 150000002334 glycols Chemical class 0.000 description 2
- WGCNASOHLSPBMP-UHFFFAOYSA-N hydroxyacetaldehyde Natural products OCC=O WGCNASOHLSPBMP-UHFFFAOYSA-N 0.000 description 2
- NLYAJNPCOHFWQQ-UHFFFAOYSA-N kaolin Chemical compound O.O.O=[Al]O[Si](=O)O[Si](=O)O[Al]=O NLYAJNPCOHFWQQ-UHFFFAOYSA-N 0.000 description 2
- 239000007788 liquid Substances 0.000 description 2
- 125000005358 mercaptoalkyl group Chemical group 0.000 description 2
- 238000006384 oligomerization reaction Methods 0.000 description 2
- 238000013031 physical testing Methods 0.000 description 2
- 239000004033 plastic Substances 0.000 description 2
- 229920003023 plastic Polymers 0.000 description 2
- 229920002857 polybutadiene Polymers 0.000 description 2
- 229920001195 polyisoprene Polymers 0.000 description 2
- 229920001296 polysiloxane Polymers 0.000 description 2
- 238000001556 precipitation Methods 0.000 description 2
- 239000011164 primary particle Substances 0.000 description 2
- 229910052710 silicon Inorganic materials 0.000 description 2
- 238000003756 stirring Methods 0.000 description 2
- 238000003860 storage Methods 0.000 description 2
- 229920003051 synthetic elastomer Polymers 0.000 description 2
- 239000005061 synthetic rubber Substances 0.000 description 2
- VTHOKNTVYKTUPI-UHFFFAOYSA-N triethoxy-[3-(3-triethoxysilylpropyltetrasulfanyl)propyl]silane Chemical compound CCO[Si](OCC)(OCC)CCCSSSSCCC[Si](OCC)(OCC)OCC VTHOKNTVYKTUPI-UHFFFAOYSA-N 0.000 description 2
- XLYOFNOQVPJJNP-UHFFFAOYSA-N water Substances O XLYOFNOQVPJJNP-UHFFFAOYSA-N 0.000 description 2
- 239000001993 wax Substances 0.000 description 2
- PUPZLCDOIYMWBV-UHFFFAOYSA-N (+/-)-1,3-Butanediol Chemical compound CC(O)CCO PUPZLCDOIYMWBV-UHFFFAOYSA-N 0.000 description 1
- KMYAABORDFJSLR-UHFFFAOYSA-N (carbamothioyltrisulfanyl) carbamodithioate Chemical compound NC(=S)SSSSC(N)=S KMYAABORDFJSLR-UHFFFAOYSA-N 0.000 description 1
- YXIWHUQXZSMYRE-UHFFFAOYSA-N 1,3-benzothiazole-2-thiol Chemical class C1=CC=C2SC(S)=NC2=C1 YXIWHUQXZSMYRE-UHFFFAOYSA-N 0.000 description 1
- DHKVCYCWBUNNQH-UHFFFAOYSA-N 2-[5-[2-(2,3-dihydro-1H-inden-2-ylamino)pyrimidin-5-yl]-1,3,4-oxadiazol-2-yl]-1-(1,4,5,7-tetrahydropyrazolo[3,4-c]pyridin-6-yl)ethanone Chemical compound C1C(CC2=CC=CC=C12)NC1=NC=C(C=N1)C1=NN=C(O1)CC(=O)N1CC2=C(CC1)C=NN2 DHKVCYCWBUNNQH-UHFFFAOYSA-N 0.000 description 1
- FMRSVUHIKQTOFR-UHFFFAOYSA-N 3-[ethoxy(dimethyl)silyl]propane-1-thiol Chemical compound CCO[Si](C)(C)CCCS FMRSVUHIKQTOFR-UHFFFAOYSA-N 0.000 description 1
- DCQBZYNUSLHVJC-UHFFFAOYSA-N 3-triethoxysilylpropane-1-thiol Chemical compound CCO[Si](OCC)(OCC)CCCS DCQBZYNUSLHVJC-UHFFFAOYSA-N 0.000 description 1
- NLHHRLWOUZZQLW-UHFFFAOYSA-N Acrylonitrile Chemical compound C=CC#N NLHHRLWOUZZQLW-UHFFFAOYSA-N 0.000 description 1
- 239000004604 Blowing Agent Substances 0.000 description 1
- 239000004971 Cross linker Substances 0.000 description 1
- VGGSQFUCUMXWEO-UHFFFAOYSA-N Ethene Chemical compound C=C VGGSQFUCUMXWEO-UHFFFAOYSA-N 0.000 description 1
- 239000004606 Fillers/Extenders Substances 0.000 description 1
- BRDWIEOJOWJCLU-LTGWCKQJSA-N GS-441524 Chemical compound C=1C=C2C(N)=NC=NN2C=1[C@]1(C#N)O[C@H](CO)[C@@H](O)[C@H]1O BRDWIEOJOWJCLU-LTGWCKQJSA-N 0.000 description 1
- 244000043261 Hevea brasiliensis Species 0.000 description 1
- 241001441571 Hiodontidae Species 0.000 description 1
- RRHGJUQNOFWUDK-UHFFFAOYSA-N Isoprene Natural products CC(=C)C=C RRHGJUQNOFWUDK-UHFFFAOYSA-N 0.000 description 1
- 229910052779 Neodymium Inorganic materials 0.000 description 1
- XUIMIQQOPSSXEZ-UHFFFAOYSA-N Silicon Chemical group [Si] XUIMIQQOPSSXEZ-UHFFFAOYSA-N 0.000 description 1
- GSEJCLTVZPLZKY-UHFFFAOYSA-N Triethanolamine Chemical compound OCCN(CCO)CCO GSEJCLTVZPLZKY-UHFFFAOYSA-N 0.000 description 1
- ZJCCRDAZUWHFQH-UHFFFAOYSA-N Trimethylolpropane Chemical compound CCC(CO)(CO)CO ZJCCRDAZUWHFQH-UHFFFAOYSA-N 0.000 description 1
- YKTSYUJCYHOUJP-UHFFFAOYSA-N [O--].[Al+3].[Al+3].[O-][Si]([O-])([O-])[O-] Chemical compound [O--].[Al+3].[Al+3].[O-][Si]([O-])([O-])[O-] YKTSYUJCYHOUJP-UHFFFAOYSA-N 0.000 description 1
- 239000012190 activator Substances 0.000 description 1
- 239000000654 additive Substances 0.000 description 1
- 230000000996 additive effect Effects 0.000 description 1
- 239000002318 adhesion promoter Substances 0.000 description 1
- 230000032683 aging Effects 0.000 description 1
- 229910052782 aluminium Inorganic materials 0.000 description 1
- 229960005363 aluminium oxide Drugs 0.000 description 1
- 239000010692 aromatic oil Substances 0.000 description 1
- 229910052788 barium Inorganic materials 0.000 description 1
- 238000009835 boiling Methods 0.000 description 1
- 229910052791 calcium Inorganic materials 0.000 description 1
- 239000011575 calcium Substances 0.000 description 1
- 239000000378 calcium silicate Substances 0.000 description 1
- 229910052918 calcium silicate Inorganic materials 0.000 description 1
- OYACROKNLOSFPA-UHFFFAOYSA-N calcium;dioxido(oxo)silane Chemical compound [Ca+2].[O-][Si]([O-])=O OYACROKNLOSFPA-UHFFFAOYSA-N 0.000 description 1
- 125000004432 carbon atom Chemical group C* 0.000 description 1
- 150000004649 carbonic acid derivatives Chemical class 0.000 description 1
- BJLJNLUARMMMLW-UHFFFAOYSA-N chloro-(3-chloropropyl)-dimethylsilane Chemical compound C[Si](C)(Cl)CCCCl BJLJNLUARMMMLW-UHFFFAOYSA-N 0.000 description 1
- 229920003193 cis-1,4-polybutadiene polymer Polymers 0.000 description 1
- 239000007859 condensation product Substances 0.000 description 1
- 229920003244 diene elastomer Polymers 0.000 description 1
- 238000004821 distillation Methods 0.000 description 1
- 239000012990 dithiocarbamate Substances 0.000 description 1
- 150000004659 dithiocarbamates Chemical class 0.000 description 1
- 239000000975 dye Substances 0.000 description 1
- 239000000839 emulsion Substances 0.000 description 1
- 125000003700 epoxy group Chemical group 0.000 description 1
- HQQADJVZYDDRJT-UHFFFAOYSA-N ethene;prop-1-ene Chemical group C=C.CC=C HQQADJVZYDDRJT-UHFFFAOYSA-N 0.000 description 1
- 238000001125 extrusion Methods 0.000 description 1
- 125000000524 functional group Chemical group 0.000 description 1
- 230000009477 glass transition Effects 0.000 description 1
- 150000002357 guanidines Chemical class 0.000 description 1
- 229910000856 hastalloy Inorganic materials 0.000 description 1
- 239000012760 heat stabilizer Substances 0.000 description 1
- 125000005842 heteroatom Chemical group 0.000 description 1
- TZMQHOJDDMFGQX-UHFFFAOYSA-N hexane-1,1,1-triol Chemical compound CCCCCC(O)(O)O TZMQHOJDDMFGQX-UHFFFAOYSA-N 0.000 description 1
- 230000007062 hydrolysis Effects 0.000 description 1
- 238000006460 hydrolysis reaction Methods 0.000 description 1
- 150000004679 hydroxides Chemical class 0.000 description 1
- 125000002887 hydroxy group Chemical group [H]O* 0.000 description 1
- TUMMOPTUHGUTLX-UHFFFAOYSA-N hydroxysilylformic acid Chemical compound O[SiH2]C(O)=O TUMMOPTUHGUTLX-UHFFFAOYSA-N 0.000 description 1
- 239000003112 inhibitor Substances 0.000 description 1
- 238000001746 injection moulding Methods 0.000 description 1
- 239000004611 light stabiliser Substances 0.000 description 1
- 229910052749 magnesium Inorganic materials 0.000 description 1
- 239000011777 magnesium Substances 0.000 description 1
- HCWCAKKEBCNQJP-UHFFFAOYSA-N magnesium orthosilicate Chemical compound [Mg+2].[Mg+2].[O-][Si]([O-])([O-])[O-] HCWCAKKEBCNQJP-UHFFFAOYSA-N 0.000 description 1
- 239000000391 magnesium silicate Substances 0.000 description 1
- 229910052919 magnesium silicate Inorganic materials 0.000 description 1
- 235000019792 magnesium silicate Nutrition 0.000 description 1
- 239000000463 material Substances 0.000 description 1
- 238000005259 measurement Methods 0.000 description 1
- 239000004005 microsphere Substances 0.000 description 1
- 230000004048 modification Effects 0.000 description 1
- 238000012986 modification Methods 0.000 description 1
- 229920003052 natural elastomer Polymers 0.000 description 1
- 229920001194 natural rubber Polymers 0.000 description 1
- QEFYFXOXNSNQGX-UHFFFAOYSA-N neodymium atom Chemical compound [Nd] QEFYFXOXNSNQGX-UHFFFAOYSA-N 0.000 description 1
- SLCVBVWXLSEKPL-UHFFFAOYSA-N neopentyl glycol Chemical compound OCC(C)(C)CO SLCVBVWXLSEKPL-UHFFFAOYSA-N 0.000 description 1
- 238000011022 operating instruction Methods 0.000 description 1
- 150000007524 organic acids Chemical class 0.000 description 1
- 235000005985 organic acids Nutrition 0.000 description 1
- 150000003961 organosilicon compounds Chemical class 0.000 description 1
- SOQBVABWOPYFQZ-UHFFFAOYSA-N oxygen(2-);titanium(4+) Chemical class [O-2].[O-2].[Ti+4] SOQBVABWOPYFQZ-UHFFFAOYSA-N 0.000 description 1
- WXZMFSXDPGVJKK-UHFFFAOYSA-N pentaerythritol Chemical compound OCC(CO)(CO)CO WXZMFSXDPGVJKK-UHFFFAOYSA-N 0.000 description 1
- 239000000049 pigment Substances 0.000 description 1
- 239000004014 plasticizer Substances 0.000 description 1
- 238000007639 printing Methods 0.000 description 1
- 238000012545 processing Methods 0.000 description 1
- 238000010992 reflux Methods 0.000 description 1
- 230000002787 reinforcement Effects 0.000 description 1
- 239000012763 reinforcing filler Substances 0.000 description 1
- 238000010057 rubber processing Methods 0.000 description 1
- 229930195734 saturated hydrocarbon Natural products 0.000 description 1
- SCPYDCQAZCOKTP-UHFFFAOYSA-N silanol Chemical compound [SiH3]O SCPYDCQAZCOKTP-UHFFFAOYSA-N 0.000 description 1
- 239000010703 silicon Substances 0.000 description 1
- 239000007858 starting material Substances 0.000 description 1
- 230000003068 static effect Effects 0.000 description 1
- 125000001424 substituent group Chemical group 0.000 description 1
- 238000006467 substitution reaction Methods 0.000 description 1
- 150000005846 sugar alcohols Polymers 0.000 description 1
- 238000009864 tensile test Methods 0.000 description 1
- CZDYPVPMEAXLPK-UHFFFAOYSA-N tetramethylsilane Chemical group C[Si](C)(C)C CZDYPVPMEAXLPK-UHFFFAOYSA-N 0.000 description 1
- 229920001169 thermoplastic Polymers 0.000 description 1
- 239000004416 thermosoftening plastic Substances 0.000 description 1
- 150000003585 thioureas Chemical class 0.000 description 1
- OGIDPMRJRNCKJF-UHFFFAOYSA-N titanium oxide Inorganic materials [Ti]=O OGIDPMRJRNCKJF-UHFFFAOYSA-N 0.000 description 1
- FBBATURSCRIBHN-UHFFFAOYSA-N triethoxy-[3-(3-triethoxysilylpropyldisulfanyl)propyl]silane Chemical compound CCO[Si](OCC)(OCC)CCCSSCCC[Si](OCC)(OCC)OCC FBBATURSCRIBHN-UHFFFAOYSA-N 0.000 description 1
- 229930195735 unsaturated hydrocarbon Natural products 0.000 description 1
- 239000011800 void material Substances 0.000 description 1
- 229910052725 zinc Inorganic materials 0.000 description 1
- 239000011787 zinc oxide Substances 0.000 description 1
Classifications
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07F—ACYCLIC, CARBOCYCLIC OR HETEROCYCLIC COMPOUNDS CONTAINING ELEMENTS OTHER THAN CARBON, HYDROGEN, HALOGEN, OXYGEN, NITROGEN, SULFUR, SELENIUM OR TELLURIUM
- C07F7/00—Compounds containing elements of Groups 4 or 14 of the Periodic System
- C07F7/02—Silicon compounds
- C07F7/08—Compounds having one or more C—Si linkages
- C07F7/18—Compounds having one or more C—Si linkages as well as one or more C—O—Si linkages
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08K—Use of inorganic or non-macromolecular organic substances as compounding ingredients
- C08K5/00—Use of organic ingredients
- C08K5/54—Silicon-containing compounds
- C08K5/541—Silicon-containing compounds containing oxygen
- C08K5/5415—Silicon-containing compounds containing oxygen containing at least one Si—O bond
- C08K5/5419—Silicon-containing compounds containing oxygen containing at least one Si—O bond containing at least one Si—C bond
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08K—Use of inorganic or non-macromolecular organic substances as compounding ingredients
- C08K3/00—Use of inorganic substances as compounding ingredients
- C08K3/34—Silicon-containing compounds
- C08K3/36—Silica
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08K—Use of inorganic or non-macromolecular organic substances as compounding ingredients
- C08K5/00—Use of organic ingredients
- C08K5/54—Silicon-containing compounds
- C08K5/548—Silicon-containing compounds containing sulfur
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08L—COMPOSITIONS OF MACROMOLECULAR COMPOUNDS
- C08L21/00—Compositions of unspecified rubbers
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08L—COMPOSITIONS OF MACROMOLECULAR COMPOUNDS
- C08L9/00—Compositions of homopolymers or copolymers of conjugated diene hydrocarbons
- C08L9/06—Copolymers with styrene
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08L—COMPOSITIONS OF MACROMOLECULAR COMPOUNDS
- C08L9/00—Compositions of homopolymers or copolymers of conjugated diene hydrocarbons
Definitions
- the invention relates to rubber mixtures, a process for their preparation and their use.
- hydrolyzable sulfur-containing organosilicon compounds are capable of reacting with hydroxyl group-containing fillers, such as natural and synthetic silicates, carbonates, glasses and metal oxides. They are used for surface modification and adhesion promotion. In the rubber processing industry, they are used as adhesion promoters between the reinforcing filler and the polymer used ( Angew. Chem. 98, (1986) 237-253 . DE2141159 . DE2212239 . DE19544469A1 . US3978103 . US4048206 . EP784072A1 ).
- polysulfan alkyltrialkoxysilanes
- polysulfan alkyltrialkoxysilanes
- a disadvantage of using the trialkoxyfunctional silanes is the emission of volatile hydrocarbons, which in practice is mainly methanol and ethanol.
- Dialkyl monoalkoxysilyl polysulfides are known. On reason The dialkylmonoalkoxy group has a lower emission of volatile hydrocarbons than trialkoxy compounds.
- Dialkylmonoalkoxysilylpolysulfide Disadvantage of Dialkylmonoalkoxysilylpolysulfide is the poor abrasion and tear propagation resistance.
- the object of the present invention is to provide rubber mixtures in whose production a low emission of volatile hydrocarbons occurs and the rubber mixtures have an improved tear propagation resistance compared to rubber mixtures with known silanes.
- At least one of R 2 or R 3 may be an -O- (YO) m -X group.
- R 4 may preferably be CH 2 , CH 2 CH 2 , CH 2 CH 2 CH 2 , CH 2 CH 2 CH 2 CH 2 , CH (CH 3 ), CH 2 CH (CH 3 ), CH (CH 3 ) CH 2 , C (CH 3 ) 2 , CH (C 2 H 5 ), CH 2 CH 2 CH (CH 3 ) or CH 2 CH (CH 3 ) CH 2 .
- the organosilane of the general formula I can consist of a mixture of compounds of organosilanes of the general formula I.
- the mixtures may contain compounds of organosilanes having the same or different m.
- the mixtures of organosilanes may contain compounds having the same or different Y groups.
- the mixtures may contain compounds of organosilanes having the same or different R 1 , R 2 , R 3 or R 4 groups.
- oligo- and polysiloxanes can be formed by addition of water.
- the oligo- and polysiloxanes may be obtained by oligomerization or co-oligomerization of the corresponding alkoxysilane compounds of general formula I by addition of water and additive addition and procedure known to those skilled in the art.
- the resulting oligo- or polymerization products can in the Organosilane compounds of general formula I may be included.
- the organosilane of the general formula I may also be an oligo- or polymerization product of the organosilane compound of the general formula I.
- the organosilane of general formula I may be a mixture of oligo- or polymerization products of the organosilane compound of general formula I and uncondensed organosilane compound of general formula I.
- the organosilane of the general formula I can be added to the mixing process both in pure form and supported on an inert organic or inorganic carrier, as well as prereacted with an organic or inorganic carrier.
- Preferred support materials may be precipitated or fumed silicas, waxes, thermoplastics, natural or synthetic silicates, natural or synthetic oxides, especially alumina, or carbon blacks.
- the organosilanes of the general formula I can also be added to the mixing process in a pre-reacted form with the filler to be used.
- Amorphous silicas prepared by precipitation of solutions of silicates having BET surface areas of from 20 to 400 m 2 / g in amounts of from 5 to 150 parts by weight, based in each case on 100 parts of rubber, may preferably be used.
- the rubber raw mixtures and vulcanizates according to the invention may contain further rubber auxiliaries, such as reaction accelerators, aging inhibitors, heat stabilizers, light stabilizers, antiozonants, processing aids, plasticizers, tackifiers, blowing agents, dyes, pigments, waxes, extenders, organic acids, Retarder, metal oxides or activators, such as triethanolamine or hexanetriol.
- rubber auxiliaries such as reaction accelerators, aging inhibitors, heat stabilizers, light stabilizers, antiozonants, processing aids, plasticizers, tackifiers, blowing agents, dyes, pigments, waxes, extenders, organic acids, Retarder, metal oxides or activators, such as triethanolamine or hexanetriol.
- Polyalkylene glycols may be polyethylene glycols, polypropylene glycols and / or polybutylene glycols.
- the polyalkylene glycols may have a molecular weight between 50 and 50,000 g / mol, preferably between 50 and 20,000 g / mol, more preferably between 200 and 10,000 g / mol, most preferably between 400 and 6,000 g / mol, most preferably between 500 and 3,000 g / mol, have.
- the polypropylene glycols may be hydrocarbon-terminated polypropylene glycol Alk-O- (CH 2 -CH (CH 3 ) -O) yl -H or Alk-O- (CH 2 -CH (CH 3 ) -O) yl- alk, where y is I and Alk have the abovementioned meaning.
- the polybutylene glycols may include hydrocarbon- terminated polybutylene glycol , Alk-O- (CH 2 -CH 2 -CH 2 -CH 2 -O) yl -H, Alk-O- (CH 2 -CH (CH 3 ) -CH 2 -O) yl -H , Alk-O- (CH 2 -CH 2 -CH 2 -CH 2 -O) yl -alk or Alk-O- (CH 2 -CH (CH 3 ) -CH 2 -O) yl- alk, where y I and Alk have the abovementioned meaning.
- Polyalkylene glycols may be polyethylene glycol, polypropylene glycol, polybutylene glycol, or etherified with mixtures thereof neopentyl glycol HO-CH 2 -C (Me) 2 -CH 2 -OH, pentaerythritol C (CH 2 -OH) 4 or trimethylolpropane CH 3 -CH 2 - C (CH 2 -OH) 3 , where the repeat units of ethylene glycol, propylene glycol or / and butylene glycol in the etherified polyalcohols are between 2 and 100, preferably between 2 and 50, more preferably between 3 and 30, very particularly preferably between 3 and 15, are, be.
- the rubber auxiliaries can be used in known amounts, which depend inter alia on the intended use. Usual amounts are, for example, amounts of 0.1 to 50 wt .-%, preferably 0.1 to 30 wt .-%, based on rubber. As crosslinkers sulfur or sulfur-donating substances can be used.
- the rubber mixtures according to the invention may additionally contain vulcanization accelerators. For example, mercaptobenzothiazoles, sulfenamides, guanidines, thiurams, dithiocarbamates, thioureas and thiocarbonates can be used as suitable vulcanization accelerators.
- the vulcanization accelerators and sulfur can be used in amounts of 0.1 to 10 wt .-%, preferably 0.1 to 5 wt .-%, based on rubber.
- Another object of the invention is a process for the preparation of the rubber mixture according to the invention, which is characterized by mixing the rubber, filler, optionally further rubber auxiliaries and at least one organosilane of the general formula I.
- the vulcanization of the rubber mixtures according to the invention can be carried out at temperatures of 100 to 200 ° C, preferably 130 to 180 ° C, optionally under pressure of 10 to 200 bar.
- the blending of the rubbers with the filler, optionally rubber auxiliaries and the organosilane of the general formula I can be carried out in known mixing units, such as rollers, internal mixers and mixing extruders.
- the rubber blends of the present invention may be used to make molded articles, for example, for the manufacture of pneumatic tires, tire treads, cable jackets, hoses, power belts, conveyor belts, roller coverings, tires, shoe soles, gaskets and damping elements.
- the rubber mixtures according to the invention show an improved tear propagation resistance.
- the resulting suspension is filtered and the filter cake washed with toluene.
- the filtrate is freed from the solvent on a rotary evaporator.
- the resulting suspension is filtered, the filter cake washed with toluene and the filtrate freed again from the toluene on a rotary evaporator.
- the suspension is cooled, filtered and the residue washed with ethanol.
- the filtrate is freed from the solvent on a rotary evaporator at 20-400 mbar and 60-90 ° C and filtered again. There are 769.2 g of an orange liquid isolated.
- a Bruker DRX 500 NMR instrument is used according to the rules and operating instructions known to the person skilled in the art.
- the measurement frequencies are 99.35 MHz for 29 Si cores and 500.13 MHz for 1 H cores.
- the reference is tetramethylsilane (TMS).
- the recipe used for the rubber compounds is given in Table 1 below.
- the unit phr means parts by weight, based on 100 parts of the raw rubber used.
- the silanes of the invention are metered in the same weight.
- the general procedure for the preparation of rubber compounds and their vulcanizates is described in the book: Rubber Technology Handbook ", W. Hofmann, Hanser Verlag 1994 described.
- the coupling reagents Si 69, a bis- (triethoxy-silylpropyl) tetrasulfide (TESPT), and VP Si 263, a 3-mercaptopropyl (triethoxysilane) (MPTES), are sales products of Degussa AG.
- the coupling reagent VP Si 208, an octylsilyltriethoxysilane, is as Alkylsilane is a processing aid and is a sales product of Degussa AG.
- the polymer VSL 5025-1 is a polymerized in solution SBR copolymer of Bayer AG, with a styrene content of 25 wt .-% and a butadiene content of 75 wt .-%.
- the copolymer contains 37.5 phr of oil and has a Mooney viscosity (ML 1 + 4/100 ° C) of 50.
- the polymer Buna CB 24 is a cis-1,4-polybutadiene (neodymium type) from Bayer AG, with a cis-1,4 content of at least 96% and a Mooney viscosity of 44 ⁇ 5.
- Ultrasil 7000 GR is an easily dispersible silica from Degussa AG and has a BET surface area of 170 m 2 / g.
- the aromatic oil used is Naftolen ZD from Chemetall, Vulkanox 4020 is PPD from Bayer AG and Protektor G3108 is an Ozontikwachs the Paramelt B.V ..
- Vulkacit D (DPG) and Vulkacit CZ (CBS) are commercial products of Bayer AG.
- Perkacit TBzTD tetrabenzyl thiuram tetrasulfide
- the rubber mixtures are prepared in an internal mixer according to the mixing instructions in Table 2.
- Table 2 step 1 Einstellunqen mixing unit Werner & Pfleiderer E type number of revolutions 60 min -1 stamp printing 5.5 bar void 1,58 l filling level 0.56 Flow temp. 70 ° C mixing process 0 to 1 min Buna VSL 5025-1 + Buna CB 24 1 to 2 min 1/2 silica, ZnO, stearic acid, naftoles ZD, silane 2 to 4 min 1/2 silica, Vulkanox, protector 4 min Clean 4 to 5 min Mix 5 min ventilate 5 to 6 min Mix and extend Batch temp.
- Table 3 summarizes the rubber testing methods. Table 3: Physical testing Standard / Conditions Tensile test on the ring, 23 ° C DIN 53504, ISO 37 Tensile strength (MPa) Voltage values (MPa) Elongation at break (%) Tearing test after Graves DIN 53 515 DIN abrasion, 10 N force (mm 3 ) DIN 53 516 Ball Rebound, 60 ° C (%) ASTM D 5308
- Table 4 shows the results of the rubber technical examination.
- Table 4 vulcanizate unit Mixture 1 Mixture 2 Mixture 3 Mixture 4 (Ref.) (Ref.) (Ref.) (Ref.) (Ref.) tensile strenght [MPa] 12.8 15.2 12.2 15.2 Voltage value 100% [MPa] 1.2 1.2 1.3 1.2 Voltage value 300% [MPa] 5.8 6.2 6.1 6.7 Voltage value 300% / 100% [-] 4.8 5.2 4.7 5.6 elongation [%] 480 480 460 460 Tear cons-standing [N / mm] 53 63 45 74 Ball rebound (60 ° C) [%] 65.5 69.5 65.1 69.1 DIN abrasion [mm 3 ] 88 66 82 59
- the dimethyl variant of mercaptosilane shows significant improvements in important properties.
- the stress value at 300% elongation, the reinforcement factor (stress value 300% / 100%), the tear propagation resistance and the DIN abrasion are significantly better than with the corresponding triethoxy variant (MPTES) (mixture 2). They are also significantly better than TESPT (mixture 1) and the corresponding dimethyl variant DMESPT (mixture 3).
- Example 7 HS-CH 2 -CH 2 -CH 2 -Si (Me) (OEt) [(O-CH (CH 3 ) -CH 2 ) 16 -OC 4 H 9 ]
- Example 8 HS-CH 2 -CH 2 -CH 2 -Si (Me) (OEt) [(O-CH 2 -CH 2 ) 4 -O-CH 2 -CH (Et) -C 4 H 9 ]
- HS-CH 2 -CH 2 -CH 2 -SiMe (OEt) 2 is based on EP 1 538 152 A1 Example 4 prepared.
- the starting materials used are Cl-CH 2 -CH 2 -CH 2 -SiMe (OEt) 2 , Cl-CH 2 -CH 2 -CH 2 -SiMeCl 2 , NaSH (dried) and ethanol.
- the suspension obtained is filtered, freed from the solvent and the silane purified by distillation.
- the recipe used for the rubber compounds is given in Table 5 below.
- the mixtures differ in the added coupling agent as indicated in Table 6.
- the mixing instructions are listed in Table 2.
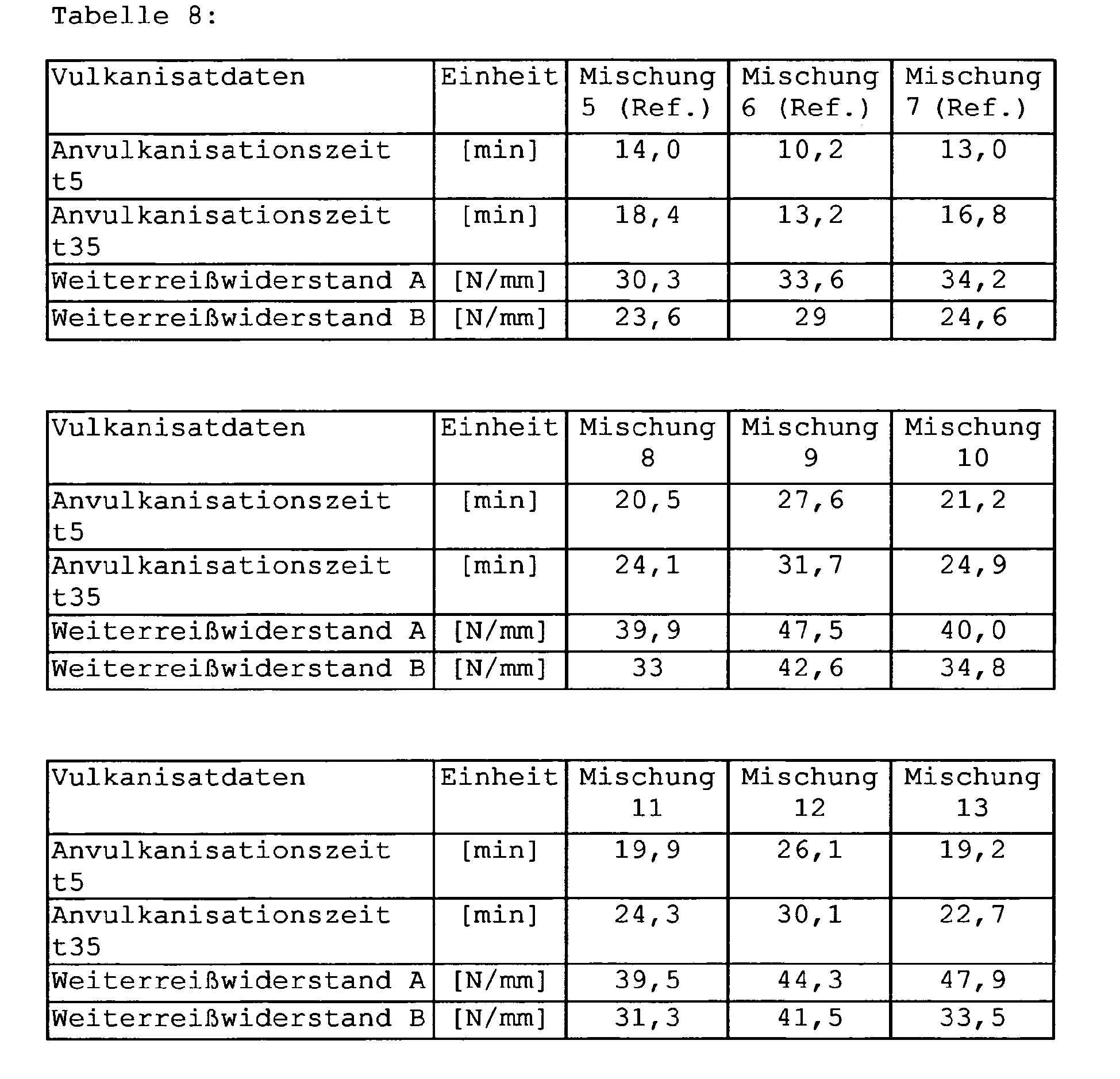
- Table 5 substance Mixtures 5 to 13 [Phr] 1st stage Buna VSL 5025-1 96 Buna CB 24 30 Ultrasil 7000 GR 80 Silanes from Table 6 2 ZnO 3 stearic acid 2 Naftolen ZD 10 Vulcanox 4020 1.5 Protector G 3108 1 2nd stage Batch level 1 3rd stage Batch level 2 Vulkacit D 2 Vulkacit CZ 1.5 Perkacit TBzTD 0.2 sulfur 1.5 Mixture no.
- silane 5 (Ref.) VP Si 263 6 (Ref.) Silane from Comparative Example 1 7 (Ref.) Silane from Comparative Example 3 8th Silane from Example 3 9 Silane from Example 4 10 Silane from Example 5 11 Silane from example 6 12 Silane from example 7 13 Silane from Example 8
- Table 7 summarizes the methods for rubber testing. Table 7: Physical testing Standard / Conditions Vulcanization behavior, 130 ° C DIN 53523/4, ISO 667 Scorch time t 5 Scorch time t 35 Tear propagation THE A ASTM D 624 Tear propagation THE B ASTM D 624
- Table 8 shows the results of the rubber testing.
- the tear propagation resistances of the mixtures 8 to 13 are markedly better than those of the blends 5, 6, 7.
- the mixtures with the long-chain alcohol silanes as substituents show an improved tear propagation behavior compared to the other mixtures. This applies both to unsubstituted mercaptosilane from mixture 5 (VP Si 263) and to the mercaptosilanes with a methyl group (mixture 7) and with two methyl groups (mixture 6).
- Mixtures 8 through 13 also show improved Mooney Scorch data. This is associated with improved processing safety, for example in the extrusion of tire treads or injection molding.
- the recipe used for the rubber compounds is given in Table 9 below.
- the mixtures differ in the added coupling agent based on equimolar dosage as indicated in Table 10.
- the mixing instructions are listed in Table 2.
- Table 9 substance Mixtures 14 to 21 [Phr] 1st stage Buna VSL 5025-1 96 Buna CB 24 30 Ultrasil 7000 GR 80 Silanes from Table 10 equimolar ZnO 3 stearic acid 2 Naftolen ZD 10 Vulcanox 4020 1.5 Protector G 3108 1 2nd stage Batch level 1 3rd stage Batch level 2 Vulkacit D 2 Vulkacit CZ 1.5 Perkacit TBzTD 0.2 sulfur 1.5 Mixture no.
- silane phr 14 (Ref.) VP Si 263 2.00 15 (Ref.) Silane from Comparative Example 3 1.75 16 Silane from Example 3 4.31 17 Silane from Example 4 9.68 18 Silane from Example 5 3.82 19 Silane from example 6 2.97 20 Silane from example 7 9.80 21 Silane from Example 8 3.94
- Table 11 shows the results of the rubber testing.
- the mixtures with the silanes from Examples 3 to 8 show advantages both in the tear propagation behavior and in the scorch behavior towards unsubstituted mercaptosilane and mercaptosilane having one methyl and two ethoxy groups.
Abstract
Description
Die Erfindung betrifft Kautschukmischungen, ein Verfahren zu ihrer Herstellung und ihrer Verwendung.The invention relates to rubber mixtures, a process for their preparation and their use.
Es ist bekannt, daß hydrolysierbare schwefelhaltige Organosiliciumverbindungen in der Lage sind, mit Hydroxylgruppen enthaltenden Füllstoffen, wie natürlichen und synthetischen Silicaten, Carbonaten, Gläsern und Metalloxiden, zu reagieren. Sie werden dabei zur Oberflächenmodifizierung und Haftvermittlung verwendet. In der kautschukverarbeitenden Industrie werden sie als Haftvermittler zwischen dem verstärkenden Füllstoff und dem eingesetzten Polymer eingesetzt (
Desweiteren ist die Verwendung von mercaptofunktionalisierten Organosilanen in Kautschukmischungen bekannt (
Ein Nachteil der Verwendung der trialkoxyfunktionellen Silane ist die Emission flüchtiger Kohlenwasserstoffe, wobei es sich in der Praxis hauptsächlich um Methanol und Ethanol handelt.A disadvantage of using the trialkoxyfunctional silanes is the emission of volatile hydrocarbons, which in practice is mainly methanol and ethanol.
Aus
Aus
Nachteil der Dialkylmonoalkoxysilylpolysulfide ist der schlechte Abrieb und Weiterreißwiderstand.Disadvantage of Dialkylmonoalkoxysilylpolysulfide is the poor abrasion and tear propagation resistance.
Aufgabe der vorliegenden Erfindung ist es, Kautschukmischungen zur Verfügung zu stellen, bei deren Herstellung eine geringe Emission flüchtiger Kohlenwasserstoffe auftritt und die Kautschukmischungen einen verbesserten Weiterreißwiderstand gegenüber Kautschukmischungen mit bekannten Silanen aufweisen.The object of the present invention is to provide rubber mixtures in whose production a low emission of volatile hydrocarbons occurs and the rubber mixtures have an improved tear propagation resistance compared to rubber mixtures with known silanes.
Gegenstand der Erfindung sind Kautschukmischungen, enthaltend Kautschuk, Füllstoffe, gegebenenfalls weitere Kautschukhilfsmittel und mindestens ein Organosilan der allgemeinen Formel I,
R1R2R3SiR4-SH (I)
wobei R1 Methyl oder Ethyl ist,
R2 -O-(Y-O)m-X ist, mit Y= verzweigte oder unverzweigte, gesättigte oder ungesättigte zweibindige Kohlenwasserstoffgruppe, vorzugsweise CH2, CH2CH2, CH2CH(CH3) oder CH(CH3)CH2, X eine C1- bis C9-Alkylgruppe, vorzugsweise Methyl oder Ethyl, und m=1-40, bevorzugt 2-30, besonders bevorzugt 3 bis 25, ganz besonders bevorzugt 4 bis 20, außerordentlich bevorzugt 10 bis 20, ist,
R3 Methyl, Ethyl, Methoxy, Ethoxy oder R2 ist,
und R4 eine verzweigte oder unverzweigte, gesättigte oder ungesättigte, aliphatische, aromatische oder gemischt aliphatische /aromatische zweibindige C1-C12 Kohlenwasserstoffgruppe ist.The invention relates to rubber mixtures containing rubber, fillers, optionally further rubber auxiliaries and at least one organosilane of the general formula I,
R 1 R 2 R 3 SiR 4 -SH (I)
where R 1 is methyl or ethyl,
R 2 is -O- (YO) m -X, with Y = branched or unbranched, saturated or unsaturated divalent hydrocarbon group, preferably CH 2 , CH 2 CH 2 , CH 2 CH (CH 3 ) or CH (CH 3 ) CH 2 , X is a C 1 - to C 9 -alkyl group, preferably methyl or ethyl, and m = 1-40, preferably 2-30, more preferably 3 to 25, very particularly preferably 4 to 20, most preferably 10 to 20,
R 3 is methyl, ethyl, methoxy, ethoxy or R 2 ,
and R 4 is a branched or unbranched, saturated or unsaturated, aliphatic, aromatic or mixed aliphatic / aromatic C 1 -C 12 hydrocarbyl group.
Vorzugsweise kann mindestens eine der Gruppen R2 oder R3 eine -O-(Y-O)m-X Gruppe sein.Preferably, at least one of R 2 or R 3 may be an -O- (YO) m -X group.
R4 kann bevorzugt CH2, CH2CH2, CH2CH2CH2, CH2CH2CH2CH2, CH(CH3), CH2CH(CH3), CH(CH3)CH2, C(CH3)2, CH(C2H5), CH2CH2CH(CH3) oder CH2CH(CH3)CH2 sein.R 4 may preferably be CH 2 , CH 2 CH 2 , CH 2 CH 2 CH 2 , CH 2 CH 2 CH 2 CH 2 , CH (CH 3 ), CH 2 CH (CH 3 ), CH (CH 3 ) CH 2 , C (CH 3 ) 2 , CH (C 2 H 5 ), CH 2 CH 2 CH (CH 3 ) or CH 2 CH (CH 3 ) CH 2 .
Verbindungen der Formel I können sein:
-
[(C4H9O-(CH2-CH2O)2] (Me)2Si(CH2)3SH,
-
[(C4H9O-(CH2-CH2O)3] (Me)2Si(CH2)3SH,
-
[(C4H9O-(CH2-CH2O)4] (Me)2Si(CH2)3SH,
-
[(C4H9O-(CH2-CH2O)5] (Me)2Si(CH2)3SH,
-
[(C4H9O-(CH2-CH2O)6] (Me)2Si(CH2)3SH,
-
[(C5H11O-(CH2-CH2O)2] (Me)2Si(CH2)3SH,
-
[(C5H11O-(CH2-CH2O)3] (Me)2Si(CH2)3SH,
-
[(C5H11O-(CH2-CH2O)4] (Me)2Si(CH2)3SH,
-
[(C5H11O-(CH2-CH2O)5] (Me)2Si(CH2)3SH,
-
[(C5H11O-(CH2-CH2O)6] (Me)2Si(CH2)3SH,
-
[(C6H13O-(CH2-CH2O)2] (Me)2Si(CH2)3SH,
-
[(C6H13O-(CH2-CH2O)3] (Me)2Si(CH2)3SH,
-
[(C6H13O-(CH2-CH2O)4] (Me)2Si(CH2)3SH,
-
[(C6H13O-(CH2-CH2O)5] (Me)2Si(CH2)3SH,
-
[(C6H13O-(CH2-CH2O)6] (Me)2Si(CH2)3SH,
-
[(C7H15O-(CH2-CH2O)2] (Me)2Si(CH2)3SH,
-
[(C7H15O-(CH2-CH2O)3] (Me)2Si(CH2)3SH,
-
[(C7H15O-(CH2-CH2O)4] (Me)2Si(CH2)3SH,
-
[(C7H15O-(CH2-CH2O)5] (Me)2Si(CH2)3SH,
-
[(C7H15O-(CH2-CH2O)6] (Me)2Si(CH2)3SH,
-
[(C8H17O-(CH2-CH2O)2] (Me)2Si(CH2)3SH,
-
[(C8H17O-(CH2-CH2O)3] (Me)2Si(CH2)3SH,
-
[(C8H17O-(CH2-CH2O)4] (Me)2Si(CH2)3SH,
-
[(C8H17O-(CH2-CH2O)5] (Me)2Si(CH2)3SH,
-
[(C8H17O-(CH2-CH2O)6] (Me)2Si(CH2)3SH,
-
[(C9H19O-(CH2-CH2O)2](Me)2Si(CH)3SH,
-
[(C9H19O-(CH2-CH2O)3](Me)2Si(CH2)3SH,
-
[(C9H19O-(CH2-CH2O)4](Me)2Si(CH)3SH,
-
[(C9H19O-(CH2-CH2O)5](Me)2Si(CH2)3SH,
-
[(C9H19O-(CH2-CH2O)6](Me)2Si(CH)3SH,
-
[(C4H9O-(CH2-CH2O)2]2(Me)Si(CH2)3SH,
-
[(C4H9O-(CH2-CH2O)3]2(Me)Si(CH2)3SH,
-
[(C4H9O-(CH2-CH2)4]2(Me)Si(CH2)3SH,
-
[(C9H9O-(CH2-CH2O)5]2(Me)Si(CH2)3SH,
-
[(C4H9O-(CH2-CH2O)6]2(Me)Si(CH2)3SH,
-
[(C5H11O-(CH2-CH2O)2]2(Me)Si(CH2)3SH,
-
[(C5H11O-(CH2-CH2O)3]2(Me)Si(CH2)3SH,
-
[(C5H11O-(CH2-CH2O)4]2(Me)Si(CH2)3SH,
-
[(C5H11O-(CH2-CH2O)5]2(Me)Si(CH2)3SH,
-
[(C5H11O-(CH2-CH2O)6]2(Me)Si(CH2)3SH,
-
[(C6H13O-(CH2-CH2O)2]2(Me)Si(CH2)3SH,
-
[(C6H13O-(CH2-CH2O)3]2(Me)Si(CH2)3SH,
-
[(C6H13O-(CH2-CH2O)4]2(Me)Si(CH2)3SH,
-
[(C6H13O-(CH2-CH2O)5]2(Me)Si(CH2)3SH,
-
[(C6H13O-(CH2-CH2O)6]2(Me)Si(CH2)3SH,
-
[(C7H15O-(CH2-CH2O)2]2(Me)Si(CH2)3SH,
-
[(C7H15O-(CH2-CH2O)3]2(Me)Si(CH2)3SH,
-
[(C7H15O-(CH2-CH2O)4]2(Me)Si(CH2)3SH,
-
[(C7H15O-(CH2-CH2O)5]2(Me)Si(CH2)3SH,
-
[(C7H15O-(CH2-CH2O)6]2(Me)Si(CH2)3SH,
-
[(C8H17O-(CH2-CH2O)2]2(Me)Si(CH2)3SH,
-
[(C8H17O-(CH2-CH2O)3]2(Me)Si(CH2)3SH,
-
[(C8H17O-(CH2-CH2O)4]2(Me)Si(CH2)3SH,
-
[(C8H17O-(CH2-CH2O)5]2(Me)Si(CH2)3SH,
-
[(C8H17O-(CH2-CH2O)6]2(Me)Si(CH2)3SH,
-
[(C9H19O-(CH2-CH2O)2]2(Me)Si(CH2)3SH,
- [(C9H19O-(CH2-CH2O)3]2(Me)Si(CH2)3SH,
-
[(C9H19O-(CH2-CH2O)4]2(Me)Si(CH2)3SH,
-
[(C9H19O-(CH2-CH2O)5]2(Me)Si(CH2)3SH,
-
[(C9H19O-(CH2-CH2O)6]2(Me)Si(CH2)3SH,
-
[(C4H9O-(CH2-CH2O)2](Me)(EtO)Si(CH2)3SH,
-
[(C4H9O-(CH2-CH2O)3](Me)(EtO)Si(CH2)3SH,
-
[(C4H9O-(CH2-CH2O)4](Me)(EtO)Si(CH2)3SH,
-
[(C4H9O-(CH2-CH2O)5](Me)(EtO)Si(CH2)3SH,
-
[(C4H9O-(CH2-CH2O)6](Me)(EtO)Si(CH2)3SH,
-
[(C5H11O-(CH2-CH2O)2](Me)(EtO)Si(CH2)3SH,
-
[(C5H11O-(CH2-CH2O)3](Me)(EtO)Si(CH2)3SH,
-
[(C5H11O-(CH2-CH2O)4](Me)(EtO)Si(CH2)3SH,
-
[(C5H11O-(CH2-CH2O)5](Me)(EtO)Si(CH2)3SH,
[(C5H11O-(CH2-CH2O)6](Me)(EtO)Si(CH2)3SH,
-
[(C9H13O-(CH2-CH2O)2](Me)(EtO)Si(CH2)3SH,
-
[(C6H13O-(CH2-CH2O)3](Me)(EtO)Si(CH2)3SH,
-
[(C6H13O-(CH2-CH2O)4](Me)(EtO)Si(CH2)3SH,
-
[(C6H13O-(CH2-CH2O)5](Me)(EtO)Si(CH2)3SH,
-
[(C5H13O-(CH2-CH2O)6](Me)(EtO)Si(CH2)3SH,
-
[(C7H15O-(CH2-CH2O)2](Me)(EtO)Si(CH2)3SH,
-
[(C7H15O-(CH2-CH2O)3](Me)(EtO)Si(CH2)3SH,
-
[(C7H15O-(CH2-CH2O)4](Me)(EtO)Si(CH2)3SH,
-
[(C7H15O-(CH2-CH2O)5](Me)(EtO)Si(CH2)3SH,
-
[(C7H15O-(CH2-CH2O)6](Me)(EtO)Si(CH2)3SH,
-
[(C8H17O-(CH2-CH2O)2](Me)(EtO)Si(CH2)3SH,
-
[(C8H17O-(CH2-CH2O)3](Me)(EtO)Si(CH2)3SH,
-
[(C8H17O-(CH2-CH2O)4](Me)(EtO)Si(CH2)3SH,
-
[(C8H17O-(CH2-CH2O)5](Me)(EtO)Si(CH2)3SH,
-
[(C8H17O-(CH2-CH2O)6](Me)(EtO)Si(CH2)3SH,
-
[(C9H19O-(CH2-CH2O)2](Me)(EtO)Si(CH2)3SH,
-
[(C9H19O-(CH2-CH2O)3](Me)(EtO)Si(CH2)3SH,
- [(C9H19O-(CH2-CH2O)4](Me)(EtO)Si(CH2)3SH,
-
[(C9H19O-(CH2CH2O)5](Me)(EtO)Si(CH2)3SH,
-
[(C9H19O-(CH2-CH2O)6](Me)(EtO)Si(CH2)3SH,
-
[(C4H9O-(CH2-CH2O)2](Me)(MeO)Si(CH2)3SH,
-
[(C4H9O-(CH2-CH2O)3](Me)(MeO)Si(CH2)3SH,
-
[(C4H9O-(CH2-CH2O)4](Me)(MeO)Si(CH2)3SH,
-
[(C4H9O-(CH2-CH2O)5](Me)(MeO)Si(CH2)3SH,
-
[(C4H9O-(CH2-CH2)6](Me)(MeO)Si(CH2)3SH,
-
[(C5H11O-(CH2-CH2O)2](Me)(MeO)Si(CH2)3SH,
-
[(C5H11O-(CH2-CH2O)3](Me)(MeO)Si(CH2)3SH,
-
[(C5H11O-(CH2-CH2O)4](Me)(MeO)Si(CH2)3SH,
-
[(C5H11O-(CH2-CH2O)5](Me)(MeO)Si(CH2)3SH,
-
[(C5H11O-(CH2-CH2O)6](Me)(MeO)Si(CH2)3SH,
-
[(C5H13O-(CH2-CH2O)2](Me)(MeO)Si(CH2)3SH,
-
[(C6H13O-(CH2-CH2O)3](Me)(MeO)Si(CH2)3SH,
-
[(C6H13O-(CH2-CH2O)4](Me)(MeO)Si(CH2)3SH,
-
[(C6H13O-(CH2-CH2O)5](Me)(MeO)Si(CH2)3SH,
-
[(C6H13O-(CH2-CH2O)6](Me)(MeO)Si(CH2)3SH,
-
[(C7H15O-(CH2-CH2O)2](Me)(MeO)Si(CH2)3SH,
-
[(C7H15O-(CH2-CH2O)3](Me)(MeO)Si(CH2)3SH,
-
[(C7H15O-(CH2-CH2O)4](Me)(MeO)Si(CH2)3SH,
-
[(C7H15O-(CH2-CH2O)5](Me)(MeO)Si(CH2)3SH,
-
[(C7H15O-(CH2-CH2O)6](Me)(MeO)Si(CH2)3SH,
-
[(C8H17O-(CH2-CH2O)2](Me)(MeO)Si(CH2)3SH,
-
[(C8H17O-(CH2-CH2O)3](Me)(MeO)Si(CH2)3SH,
-
[(C8H17O-(CH2-CH2O)4](Me)(MeO)Si(CH2)3SH,
-
[(C8H17O-(CH2-CH2O)5](Me)(MeO)Si(CH2)3SH,
-
[(C8H17O-(CH2-CH2O)6](Me)(MeO)Si(CH2)3SH,
-
[(C9H19O-(CH2-CH2O)2](Me)(MeO)Si(CH2)3SH,
-
[(C9H19O-(CH2-CH2O)3](Me)(MeO)Si(CH2)3SH,
-
[(C9H19O-(CH2-CH2O)4](Me)(MeO)Si(CH2)3SH,
- [(C9H19O-(CH2-CH2O)5](Me)(MeO)Si(CH2)3SH oder
-
[(C9H19O-(CH2-CH2O)6](Me)(MeO)Si(CH2)3SH, mit Me = CH3 und Et = CH2CH3.
-
[(C 4 H 9 O- (CH 2 -CH 2 O) 2 ] (Me) 2 Si (CH 2 ) 3 SH,
-
[(C 4 H 9 O- (CH 2 -CH 2 O) 3 ] (Me) 2 Si (CH 2 ) 3 SH,
-
[(C 4 H 9 O- (CH 2 -CH 2 O) 4 ] (Me) 2 Si (CH 2 ) 3 SH,
-
[(C 4 H 9 O- (CH 2 -CH 2 O) 5 ] (Me) 2 Si (CH 2 ) 3 SH,
-
[(C 4 H 9 O- (CH 2 -CH 2 O) 6 ] (Me) 2 Si (CH 2 ) 3 SH,
-
[(C 5 H 11 O- (CH 2 -CH 2 O) 2 ] (Me) 2 Si (CH 2 ) 3 SH,
-
[(C 5 H 11 O- (CH 2 -CH 2 O) 3 ] (Me) 2 Si (CH 2 ) 3 SH,
-
[(C 5 H 11 O- (CH 2 -CH 2 O) 4 ] (Me) 2 Si (CH 2 ) 3 SH,
-
[(C 5 H 11 O- (CH 2 -CH 2 O) 5 ] (Me) 2 Si (CH 2 ) 3 SH,
-
[(C 5 H 11 O- (CH 2 -CH 2 O) 6 ] (Me) 2 Si (CH 2 ) 3 SH,
-
[(C 6 H 13 O- (CH 2 -CH 2 O) 2 ] (Me) 2 Si (CH 2 ) 3 SH,
-
[(C 6 H 13 O- (CH 2 -CH 2 O) 3 ] (Me) 2 Si (CH 2 ) 3 SH,
-
[(C 6 H 13 O- (CH 2 -CH 2 O) 4 ] (Me) 2 Si (CH 2 ) 3 SH,
-
[(C 6 H 13 O- (CH 2 -CH 2 O) 5 ] (Me) 2 Si (CH 2 ) 3 SH,
-
[(C 6 H 13 O- (CH 2 -CH 2 O) 6 ] (Me) 2 Si (CH 2 ) 3 SH,
-
[(C 7 H 15 O- (CH 2 -CH 2 O) 2 ] (Me) 2 Si (CH 2 ) 3 SH,
-
[(C 7 H 15 O- (CH 2 -CH 2 O) 3 ] (Me) 2 Si (CH 2 ) 3 SH,
-
[(C 7 H 15 O- (CH 2 -CH 2 O) 4 ] (Me) 2 Si (CH 2 ) 3 SH,
-
[(C 7 H 15 O- (CH 2 -CH 2 O) 5 ] (Me) 2 Si (CH 2 ) 3 SH,
-
[(C 7 H 15 O- (CH 2 -CH 2 O) 6 ] (Me) 2 Si (CH 2 ) 3 SH,
-
[(C 8 H 17 O- (CH 2 -CH 2 O) 2 ] (Me) 2 Si (CH 2 ) 3 SH,
-
[(C 8 H 17 O- (CH 2 -CH 2 O) 3 ] (Me) 2 Si (CH 2 ) 3 SH,
-
[(C 8 H 17 O- (CH 2 -CH 2 O) 4 ] (Me) 2 Si (CH 2 ) 3 SH,
-
[(C 8 H 17 O- (CH 2 -CH 2 O) 5 ] (Me) 2 Si (CH 2 ) 3 SH,
-
[(C 8 H 17 O- (CH 2 -CH 2 O) 6 ] (Me) 2 Si (CH 2 ) 3 SH,
-
[(C 9 H 19 O- (CH 2 -CH 2 O) 2 ] (Me) 2 Si (CH) 3 SH,
-
[(C 9 H 19 O- (CH 2 -CH 2 O) 3 ] (Me) 2 Si (CH 2 ) 3 SH,
-
[(C 9 H 19 O- (CH 2 -CH 2 O) 4 ] (Me) 2 Si (CH) 3 SH,
-
[(C 9 H 19 O- (CH 2 -CH 2 O) 5 ] (Me) 2 Si (CH 2 ) 3 SH,
-
[(C 9 H 19 O- (CH 2 -CH 2 O) 6 ] (Me) 2 Si (CH) 3 SH,
-
[(C 4 H 9 O- (CH 2 -CH 2 O) 2 ] 2 (Me) Si (CH 2 ) 3 SH,
-
[(C 4 H 9 O- (CH 2 -CH 2 O) 3 ] 2 (Me) Si (CH 2 ) 3 SH,
-
[(C 4 H 9 O- (CH 2 -CH 2 ) 4 ] 2 (Me) Si (CH 2 ) 3 SH,
-
[(C 9 H 9 O- (CH 2 -CH 2 O) 5 ] 2 (Me) Si (CH 2 ) 3 SH,
-
[(C 4 H 9 O- (CH 2 -CH 2 O) 6 ] 2 (Me) Si (CH 2 ) 3 SH,
-
[(C 5 H 11 O- (CH 2 -CH 2 O) 2 ] 2 (Me) Si (CH 2 ) 3 SH,
-
[(C 5 H 11 O- (CH 2 -CH 2O) 3] 2 (Me) Si (CH 2) 3 SH,
-
[(C 5 H 11 O- (CH 2 -CH 2 O) 4 ] 2 (Me) Si (CH 2 ) 3 SH,
-
[(C 5 H 11 O- (CH 2 -CH 2 O) 5 ] 2 (Me) Si (CH 2 ) 3 SH,
-
[(C 5 H 11 O- (CH 2 -CH 2 O) 6 ] 2 (Me) Si (CH 2 ) 3 SH,
-
[(C 6 H 13 O- (CH 2 -CH 2 O) 2 ] 2 (Me) Si (CH 2 ) 3 SH,
-
[(C 6 H 13 O- (CH 2 -CH 2 O) 3 ] 2 (Me) Si (CH 2 ) 3 SH,
-
[(C 6 H 13 O- (CH 2 -CH 2 O) 4 ] 2 (Me) Si (CH 2 ) 3 SH,
-
[(C 6 H 13 O- (CH 2 -CH 2 O) 5 ] 2 (Me) Si (CH 2 ) 3 SH,
-
[(C 6 H 13 O- (CH 2 -CH 2 O) 6 ] 2 (Me) Si (CH 2 ) 3 SH,
-
[(C 7 H 15 O- (CH 2 -CH 2 O) 2 ] 2 (Me) Si (CH 2 ) 3 SH,
-
[(C 7 H 15 O- (CH 2 -CH 2 O) 3 ] 2 (Me) Si (CH 2 ) 3 SH,
-
[(C 7 H 15 O- (CH 2 -CH 2 O) 4 ] 2 (Me) Si (CH 2 ) 3 SH,
-
[(C 7 H 15 O- (CH 2 -CH 2 O) 5 ] 2 (Me) Si (CH 2 ) 3 SH,
-
[(C 7 H 15 O- (CH 2 -CH 2 O) 6 ] 2 (Me) Si (CH 2 ) 3 SH,
-
[(C 8 H 17 O- (CH 2 -CH 2 O) 2 ] 2 (Me) Si (CH 2 ) 3 SH,
-
[(C 8 H 17 O- (CH 2 -CH 2 O) 3 ] 2 (Me) Si (CH 2 ) 3 SH,
-
[(C 8 H 17 O- (CH 2 -CH 2 O) 4 ] 2 (Me) Si (CH 2 ) 3 SH,
-
[(C 8 H 17 O- (CH 2 -CH 2 O) 5 ] 2 (Me) Si (CH 2 ) 3 SH,
-
[(C 8 H 17 O- (CH 2 -CH 2 O) 6 ] 2 (Me) Si (CH 2 ) 3 SH,
-
[(C 9 H 19 O- (CH 2 -CH 2 O) 2 ] 2 (Me) Si (CH 2 ) 3 SH,
- [(C 9 H 19 O- (CH 2 -CH 2 O) 3 ] 2 (Me) Si (CH 2 ) 3 SH,
-
[(C 9 H 19 O- (CH 2 -CH 2 O) 4 ] 2 (Me) Si (CH 2 ) 3 SH,
-
[(C 9 H 19 O- (CH 2 -CH 2 O) 5 ] 2 (Me) Si (CH 2 ) 3 SH,
-
[(C 9 H 19 O- (CH 2 -CH 2 O) 6 ] 2 (Me) Si (CH 2 ) 3 SH,
-
[(C 4 H 9 O- (CH 2 -CH 2 O) 2 ] (Me) (EtO) Si (CH 2 ) 3 SH,
-
[(C 4 H 9 O- (CH 2 -CH 2 O) 3 ] (Me) (EtO) Si (CH 2 ) 3 SH,
-
[(C 4 H 9 O- (CH 2 -CH 2 O) 4 ] (Me) (EtO) Si (CH 2 ) 3 SH,
-
[(C 4 H 9 O- (CH 2 -CH 2 O) 5 ] (Me) (EtO) Si (CH 2 ) 3 SH,
-
[(C 4 H 9 O- (CH 2 -CH 2 O) 6 ] (Me) (EtO) Si (CH 2 ) 3 SH,
-
[(C 5 H 11 O- (CH 2 -CH 2 O) 2 ] (Me) (EtO) Si (CH 2 ) 3 SH,
-
[(C 5 H 11 O- (CH 2 -CH 2 O) 3 ] (Me) (EtO) Si (CH 2 ) 3 SH,
-
[(C 5 H 11 O- (CH 2 -CH 2 O) 4 ] (Me) (EtO) Si (CH 2 ) 3 SH,
-
[(C 5 H 11 O- (CH 2 -CH 2 O) 5 ] (Me) (EtO) Si (CH 2 ) 3 SH,
[(C 5 H 11 O- (CH 2 -CH 2 O) 6 ] (Me) (EtO) Si (CH 2 ) 3 SH,
-
[(C 9 H 13 O- (CH 2 -CH 2 O) 2 ] (Me) (EtO) Si (CH 2 ) 3 SH,
-
[(C 6 H 13 O- (CH 2 -CH 2O) 3] (Me) (EtO) Si (CH 2) 3 SH,
-
[(C 6 H 13 O- (CH 2 -CH 2 O) 4 ] (Me) (EtO) Si (CH 2 ) 3 SH,
-
[(C 6 H 13 O- (CH 2 -CH 2 O) 5 ] (Me) (EtO) Si (CH 2 ) 3 SH,
-
[(C 5 H 13 O- (CH 2 -CH 2 O) 6 ] (Me) (EtO) Si (CH 2 ) 3 SH,
-
[(C 7 H 15 O- (CH 2 -CH 2 O) 2 ] (Me) (EtO) Si (CH 2 ) 3 SH,
-
[(C 7 H 15 O- (CH 2 -CH 2 O) 3 ] (Me) (EtO) Si (CH 2 ) 3 SH,
-
[(C 7 H 15 O- (CH 2 -CH 2 O) 4 ] (Me) (EtO) Si (CH 2 ) 3 SH,
-
[(C 7 H 15 O- (CH 2 -CH 2 O) 5 ] (Me) (EtO) Si (CH 2 ) 3 SH,
-
[(C 7 H 15 O- (CH 2 -CH 2 O) 6 ] (Me) (EtO) Si (CH 2 ) 3 SH,
-
[(C 8 H 17 O- (CH 2 -CH 2O) 2] (Me) (EtO) Si (CH 2) 3 SH,
-
[(C 8 H 17 O- (CH 2 -CH 2 O) 3 ] (Me) (EtO) Si (CH 2 ) 3 SH,
-
[(C 8 H 17 O- (CH 2 -CH 2 O) 4 ] (Me) (EtO) Si (CH 2 ) 3 SH,
-
[(C 8 H 17 O- (CH 2 -CH 2 O) 5 ] (Me) (EtO) Si (CH 2 ) 3 SH,
-
[(C 8 H 17 O- (CH 2 -CH 2 O) 6 ] (Me) (EtO) Si (CH 2 ) 3 SH,
-
[(C 9 H 19 O- (CH 2 -CH 2 O) 2 ] (Me) (EtO) Si (CH 2 ) 3 SH,
-
[(C 9 H 19 O- (CH 2 -CH 2 O) 3 ] (Me) (EtO) Si (CH 2 ) 3 SH,
- [(C 9 H 19 O- (CH 2 -CH 2 O) 4 ] (Me) (EtO) Si (CH 2 ) 3 SH,
-
[(C 9 H 19 O- (CH 2 CH 2 O) 5 ] (Me) (EtO) Si (CH 2 ) 3 SH,
-
[(C 9 H 19 O- (CH 2 -CH 2 O) 6 ] (Me) (EtO) Si (CH 2 ) 3 SH,
-
[(C 4 H 9 O- (CH 2 -CH 2 O) 2 ] (Me) (MeO) Si (CH 2 ) 3 SH,
-
[(C 4 H 9 O- (CH 2 -CH 2 O) 3 ] (Me) (MeO) Si (CH 2 ) 3 SH,
-
[(C 4 H 9 O- (CH 2 -CH 2 O) 4 ] (Me) (MeO) Si (CH 2 ) 3 SH,
-
[(C 4 H 9 O- (CH 2 -CH 2 O) 5 ] (Me) (MeO) Si (CH 2 ) 3 SH,
-
[(C 4 H 9 O- (CH 2 -CH 2 ) 6 ] (Me) (MeO) Si (CH 2 ) 3 SH,
-
[(C 5 H 11 O- (CH 2 -CH 2 O) 2 ] (Me) (MeO) Si (CH 2 ) 3 SH,
-
[(C 5 H 11 O- (CH 2 -CH 2 O) 3 ] (Me) (MeO) Si (CH 2 ) 3 SH,
-
[(C 5 H 11 O- (CH 2 -CH 2 O) 4 ] (Me) (MeO) Si (CH 2 ) 3 SH,
-
[(C 5 H 11 O- (CH 2 -CH 2 O) 5 ] (Me) (MeO) Si (CH 2 ) 3 SH,
-
[(C 5 H 11 O- (CH 2 -CH 2 O) 6 ] (Me) (MeO) Si (CH 2 ) 3 SH,
-
[(C 5 H 13 O- (CH 2 -CH 2 O) 2 ] (Me) (MeO) Si (CH 2 ) 3 SH,
-
[(C 6 H 13 O- (CH 2 -CH 2 O) 3 ] (Me) (MeO) Si (CH 2 ) 3 SH,
-
[(C 6 H 13 O- (CH 2 -CH 2 O) 4 ] (Me) (MeO) Si (CH 2 ) 3 SH,
-
[(C 6 H 13 O- (CH 2 -CH 2 O) 5 ] (Me) (MeO) Si (CH 2 ) 3 SH,
-
[(C 6 H 13 O- (CH 2 -CH 2 O) 6 ] (Me) (MeO) Si (CH 2 ) 3 SH,
-
[(C 7 H 15 O- (CH 2 -CH 2 O) 2 ] (Me) (MeO) Si (CH 2 ) 3 SH,
-
[(C 7 H 15 O- (CH 2 -CH 2 O) 3 ] (Me) (MeO) Si (CH 2 ) 3 SH,
-
[(C 7 H 15 O- (CH 2 -CH 2 O) 4 ] (Me) (MeO) Si (CH 2 ) 3 SH,
-
[(C 7 H 15 O- (CH 2 -CH 2 O) 5 ] (Me) (MeO) Si (CH 2 ) 3 SH,
-
[(C 7 H 15 O- (CH 2 -CH 2 O) 6 ] (Me) (MeO) Si (CH 2 ) 3 SH,
-
[(C 8 H 17 O- (CH 2 -CH 2 O) 2 ] (Me) (MeO) Si (CH 2 ) 3 SH,
-
[(C 8 H 17 O- (CH 2 -CH 2 O) 3 ] (Me) (MeO) Si (CH 2 ) 3 SH,
-
[(C 8 H 17 O- (CH 2 -CH 2 O) 4 ] (Me) (MeO) Si (CH 2 ) 3 SH,
-
[(C 8 H 17 O- (CH 2 -CH 2O) 5] (Me) (MeO) Si (CH 2) 3 SH,
-
[(C 8 H 17 O- (CH 2 -CH 2 O) 6 ] (Me) (MeO) Si (CH 2 ) 3 SH,
-
[(C 9 H 19 O- (CH 2 -CH 2 O) 2 ] (Me) (MeO) Si (CH 2 ) 3 SH,
-
[(C 9 H 19 O- (CH 2 -CH 2 O) 3 ] (Me) (MeO) Si (CH 2 ) 3 SH,
-
[(C 9 H 19 O- (CH 2 -CH 2 O) 4 ] (Me) (MeO) Si (CH 2 ) 3 SH,
- [(C 9 H 19 O- (CH 2 -CH 2 O) 5 ] (Me) (MeO) Si (CH 2 ) 3 SH or
-
[(C 9 H 19 O- (CH 2 -CH 2 O) 6 ] (Me) (MeO) Si (CH 2 ) 3 SH, with Me = CH 3 and Et = CH 2 CH 3 .
Verbindungen der Formel I mit X= C3H7, C4H9, C5H11, C6H13, C7H15, C8H17 oder C9H19 können sein:
[(X-O-(CH2-CH(CH3)O-)2](Me)(MeO)Si(CH2)3SH,
[(X-O-(CH2-CH(CH3)O-)3](Me)(MeO)Si(CH2)3SH,
[(X-O-(CH2-CH(CH3)O-)4](Me)(MeO)Si(CH2)3SH,
[(X-O-(CH2-CH(CH3)O-)5](Me)(MeO)Si(CH2)3SH,
[(X-O-(CH2-CH(CH3)O-)6](Me)(MeO)Si(CH2)3SH,
[(X-O-(CH2-CH(CH3)O-)7](Me)(MeO)Si(CH2)3SH,
[(X-O-(CH2-CH(CH3)O-)8](Me)(MeO)Si(CH2)3SH,
[(X-O-(CH2-CH(CH3)O-)9](Me)(MeO)Si(CH2)3SH,
[(X-O-(CH2-CH(CH3)O-)10](Me)(MeO)Si(CH2)3SH,
[(X-O-(CH2-CH(CH3)O-)11](Me)(MeO)Si(CH2)3SH,
[(X-O-(CH2-CH(CH3)O-)12](Me)(MeO)Si(CH2)3SH,
[(X-O-(CH2-CH(CH3)O-)13](Me)(MeO)Si(CH2)3SH,
[(X-O-(CH2-CH(CH3)O-)14](Me)(MeO)Si(CH2)3SH,
[(X-O-(CH2-CH(CH3)O-)15](Me)(MeO)Si(CH2)3SH,
[(X-O-(CH2-CH(CH3)O-)16](Me)(MeO)Si(CH2)3SH,
[(X-O-(CH2-CH(CH3)O-)17](Me)(MeO)Si(CH2)3SH,
[(X-O-(CH2-CH(CH3)O-)18](Me)(MeO)Si(CH2)3SH,
[(X-O-(CH2-CH(CH3)O-)19](Me)(MeO)Si(CH2)3SH,
[(X-O-(CH2-CH(CH3)O-)20](Me)(MeO)Si(CH2)3SH,
[(X-O-(CH2-CH(CH3)O-)2](Me)(EtO)Si(CH2)3SH,
[(X-O-(CH2-CH(CH3)O-)3](Me)(EtO)Si(CH2)3SH,
[(X-O-(CH2-CH(CH3)O-)4](Me)(EtO)Si(CH2)3SH,
[(X-O-(CH2-CH(CH3)O-)5](Me)(EtO)Si(CH2)3SH,
[(X-O-(CH2-CH(CH3)O-)6](Me)(EtO)Si(CH2)3SH,
[(X-O-(CH2-CH(CH3)O-)7](Me)(EtO)Si(CH2)3SH,
[(X-O-(CH2-CH(CH3)O-)8](Me)(EtO)Si(CH2)3SH,
[(X-O-(CH2-CH(CH3)O-)9](Me)(EtO)Si(CH2)3SH,
[(X-O-(CH2-CH(CH3)O-)10](Me)(EtO)Si(CH2)3SH,
[(X-O-(CH2-CH(CH3)O-)11](Me)(EtO)Si(CH2)3SH,
[(X-O-(CH2-CH(CH3)O-)12](Me)(EtO)Si(CH2)3SH,
[ (X-O- (CH2-CH (CH3) O-) 13] (Me) (EtO) Si (CH2) 3SH,
[(X-O-(CH2-CH(CH3)O-)14] (Me)(EtO) Si(CH2)3SH,
[(X-O-(CH2-CH(CH3)O-) 15] (Me)(EtO) Si(CH2)3SH,
[(X-O-(CH2-CH(CH3)O-)16](Me)(EtO)Si(CH2)3SH,
[(X-O-(CH2-CH(CH3)O-)17](Me)(EtO)Si(CH2)3SH,
[(X-O-(CH2-CH(CH3)O-)18](Me)(EtO)Si(CH2)3SH,
[(X-O-(CH2-CH(CH3)O-)19](Me)(EtO)Si(CH2)3SH,
[(X-O-(CH2-CH(CH3)O-)20](Me)(EtO)Si(CH2)3SH,
[(X-O-(CH2-CH(CH3)O-)19](Me)(EtO) Si(CH2)3SH,
[(X-O-(CH2-CH(CH3)O-)20](Me)(EtO)Si(CH2)3SH,
[(X-O-(CH2-CH(CH3)O-)2]2(Me)Si(CH2)3SH,
[(X-O-(CH2-CH(CH3)O-)3]2(Me)Si(CH2)3SH,
[(X-O-(CH2-CH(CH3)O-)4]2(Me)Si(CH2)3SH,
[(X-O-(CH2-CH(CH3)O-)5]2(Me)Si(CH2)3SH,
[(X-O-(CH2-CH(CH3)O-)6]2(Me)Si(CH2)3SH,
[(X-O-(CH2-CH(CH3)O-)7]2(Me)Si(CH2)3SH,
[(X-O-(CH2-CH(CH3)O-)8]2(Me)Si(CH2)3SH,
[(X-O-(CH2-CH(CH3)O-)9]2(Me)Si(CH2)3SH,
[(X-O-(CH2-CH(CH3)O-)10] 2(Me)Si(CH2)3SH,
[(X-O-(CH2-CH(CH3)O-)11]2(Me)Si(CH2)3SH,
[(X-O-(CH2-CH(CH3)O-)12]2(Me)Si(CH)3SH,
[(X-O-(CH2-CH(CH3)O-)13]2(Me)Si(CH2)3SH,
[(X-O-(CH2-CH(CH3)O-)14]2(Me)Si(CH2)3SH,
[(X-O-(CH2-CH(CH3)O-)15]2(Me)Si(CH2)3SH,
[(X-O-(CH2-CH(CH3)O-)16]2(Me)Si(CH2)3SH,
[(X-O-(CH2-CH(CH3)O-)17]2(Me)Si(CH2)3SH,
[(X-O-(CH2-CH(CH3)O-)18]2(Me)Si(CH2)3SH,
[(X-O-(CH2-CH(CH3)O-)19]2(Me)Si(CH2)3SH,
[(X-O-(CH2-CH(CH3)O-)20]2(Me)Si(CH2)3SH,
[(X-O-(CH2-CH(CH3)O-)2](Me)2Si(CH2)3SH,
[(X-O-(CH2-CH(CH3)O-)3](Me)2Si(CH)3SH,
[(X-O-(CH2-CH(CH3)O-)4](Me)2Si(CH2)3SH,
[(X-O-(CH2-CH(CH3)O-)5](Me)2Si(CH)3SH,
[(X-O-(CH2-CH(CH3)O-)6](Me)2Si(CH2)3SH,
[(X-O-(CH2-CH(CH3)O-)7](Me)2Si(CH)3SH,
[(X-O-(CH2-CH(CH3)O-)8](Me)2Si(CH2)3SH,
[(X-O-(CH2-CH(CH3)O-)9](Me)2Si(CH2)3SH,
[(X-O-(CH2-CH(CH3)O-)10](Me)2Si(CH2)3SH,
[(X-O-(CH2-CH(CH3)O-)11](Me)2Si(CH2)3SH,
[(X-O-(CH2-CH(CH3)0-)12](Me)2Si(CH2)3SH,
[(X-O-(CH2-CH(CH3)O-)13](Me)2Si(CH2)3SH,
[(X-O-(CH2-CH(CH3)O-)14](Me)2Si(CH2)3SH,
[(X-O-(CH2-CH(CH3)O-)15](Me)2Si(CH2)3SH,
[(X-O-(CH2-CH(CH3)O-)16](Me) 2Si(CH2)3SH,
[(X-O-(CH2-CH(CH3)O-)17](Me)2Si(CH2)3SH,
[(X-O-(CH2-CH(CH3)O-)18](Me)2Si(CH2)3SH,
[(X-O-(CH2-CH(CH3)O-)19](Me)2Si(CH2)3SHoder
[(X-O-(CH2-CH(CH3)O-)20](Me)2Si(CH2)3SH.
Compounds of the formula I where X =C 3 H 7 , C 4 H 9 , C 5 H 11 , C 6 H 13 , C 7 H 15 , C 8 H 17 or C 9 H 19 may be:
[(XO- (CH 2 -CH (CH 3 ) O-) 2 ] (Me) (MeO) Si (CH 2 ) 3 SH,
[(XO- (CH 2 -CH (CH 3 ) O-) 3 ] (Me) (MeO) Si (CH 2 ) 3 SH,
[(XO- (CH 2 -CH (CH 3 ) O-) 4 ] (Me) (MeO) Si (CH 2 ) 3 SH,
[(XO- (CH 2 -CH (CH 3 ) O-) 5 ] (Me) (MeO) Si (CH 2 ) 3 SH,
[(XO- (CH 2 -CH (CH 3 ) O-) 6 ] (Me) (MeO) Si (CH 2 ) 3 SH,
[(XO- (CH 2 -CH (CH 3 ) O-) 7 ] (Me) (MeO) Si (CH 2 ) 3 SH,
[(XO- (CH 2 -CH (CH 3 ) O-) 8 ] (Me) (MeO) Si (CH 2 ) 3 SH,
[(XO- (CH 2 -CH (CH 3 ) O-) 9 ] (Me) (MeO) Si (CH 2 ) 3 SH,
[(XO- (CH 2 -CH (CH 3 ) O-) 10 ] (Me) (MeO) Si (CH 2 ) 3 SH,
[(XO- (CH 2 -CH (CH 3 ) O-) 11 ] (Me) (MeO) Si (CH 2 ) 3 SH,
[(XO- (CH 2 -CH (CH 3 ) O-) 12 ] (Me) (MeO) Si (CH 2 ) 3 SH,
[(XO- (CH 2 -CH (CH 3 ) O-) 13 ] (Me) (MeO) Si (CH 2 ) 3 SH,
[(XO- (CH 2 -CH (CH 3 ) O-) 14 ] (Me) (MeO) Si (CH 2 ) 3 SH,
[(XO- (CH 2 -CH (CH 3 ) O-) 15 ] (Me) (MeO) Si (CH 2 ) 3 SH,
[(XO- (CH 2 -CH (CH 3 ) O-) 16 ] (Me) (MeO) Si (CH 2 ) 3 SH,
[(XO- (CH 2 -CH (CH 3 ) O-) 17 ] (Me) (MeO) Si (CH 2 ) 3 SH,
[(XO- (CH 2 -CH (CH 3 ) O-) 18 ] (Me) (MeO) Si (CH 2 ) 3 SH,
[(XO- (CH 2 -CH (CH 3 ) O-) 19 ] (Me) (MeO) Si (CH 2 ) 3 SH,
[(XO- (CH 2 -CH (CH 3 ) O-) 20 ] (Me) (MeO) Si (CH 2 ) 3 SH,
[(XO- (CH 2 -CH (CH 3 ) O-) 2 ] (Me) (EtO) Si (CH 2 ) 3 SH,
[(XO- (CH 2 -CH (CH 3 ) O-) 3 ] (Me) (EtO) Si (CH 2 ) 3 SH,
[(XO- (CH 2 -CH (CH 3 ) O-) 4 ] (Me) (EtO) Si (CH 2 ) 3 SH,
[(XO- (CH 2 -CH (CH 3 ) O-) 5 ] (Me) (EtO) Si (CH 2 ) 3 SH,
[(XO- (CH 2 -CH (CH 3 ) O-) 6 ] (Me) (EtO) Si (CH 2 ) 3 SH,
[(XO- (CH 2 -CH (CH 3 ) O-) 7 ] (Me) (EtO) Si (CH 2 ) 3 SH,
[(XO- (CH 2 -CH (CH 3 ) O-) 8 ] (Me) (EtO) Si (CH 2 ) 3 SH,
[(XO- (CH 2 -CH (CH 3 ) O-) 9 ] (Me) (EtO) Si (CH 2 ) 3 SH,
[(XO- (CH 2 -CH (CH 3 ) O-) 10 ] (Me) (EtO) Si (CH 2 ) 3 SH,
[(XO- (CH 2 -CH (CH 3 ) O-) 11 ] (Me) (EtO) Si (CH 2 ) 3 SH,
[(XO- (CH 2 -CH (CH 3 ) O-) 12 ] (Me) (EtO) Si (CH 2 ) 3 SH,
[(XO- (CH 2 -CH (CH 3 ) O-) 13 ] (Me) (EtO) Si (CH 2 ) 3 SH,
[(XO- (CH 2 -CH (CH 3 ) O-) 14 ] (Me) (EtO) Si (CH 2 ) 3 SH,
[(XO- (CH 2 -CH (CH 3 ) O-) 15 ] (Me) (EtO) Si (CH 2 ) 3 SH,
[(XO- (CH 2 -CH (CH 3 ) O-) 16 ] (Me) (EtO) Si (CH 2 ) 3 SH,
[(XO- (CH 2 -CH (CH 3 ) O-) 17 ] (Me) (EtO) Si (CH 2 ) 3 SH,
[(XO- (CH 2 -CH (CH 3 ) O-) 18 ] (Me) (EtO) Si (CH 2 ) 3 SH,
[(XO- (CH 2 -CH (CH 3 ) O-) 19 ] (Me) (EtO) Si (CH 2 ) 3 SH,
[(XO- (CH 2 -CH (CH 3 ) O-) 20 ] (Me) (EtO) Si (CH 2 ) 3 SH,
[(XO- (CH 2 -CH (CH 3 ) O-) 19 ] (Me) (EtO) Si (CH 2 ) 3 SH,
[(XO- (CH 2 -CH (CH 3 ) O-) 20 ] (Me) (EtO) Si (CH 2 ) 3 SH,
[(XO- (CH 2 -CH (CH 3 ) O-) 2 ] 2 (Me) Si (CH 2 ) 3 SH,
[(XO- (CH 2 -CH (CH 3 ) O-) 3 ] 2 (Me) Si (CH 2 ) 3 SH,
[(XO- (CH 2 -CH (CH 3 ) O-) 4 ] 2 (Me) Si (CH 2 ) 3 SH,
[(XO- (CH 2 -CH (CH 3 ) O-) 5 ] 2 (Me) Si (CH 2 ) 3 SH,
[(XO- (CH 2 -CH (CH 3 ) O-) 6 ] 2 (Me) Si (CH 2 ) 3 SH,
[(XO- (CH 2 -CH (CH 3 ) O-) 7 ] 2 (Me) Si (CH 2 ) 3 SH,
[(XO- (CH 2 -CH (CH 3 ) O-) 8 ] 2 (Me) Si (CH 2 ) 3 SH,
[(XO- (CH 2 -CH (CH 3 ) O-) 9 ] 2 (Me) Si (CH 2 ) 3 SH,
[(XO- (CH 2 -CH (CH 3 ) O-) 10 ] 2 (Me) Si (CH 2 ) 3 SH,
[(XO- (CH 2 -CH (CH 3 ) O-) 11 ] 2 (Me) Si (CH 2 ) 3 SH,
[(XO- (CH 2 -CH (CH 3 ) O-) 12 ] 2 (Me) Si (CH) 3 SH,
[(XO- (CH 2 -CH (CH 3 ) O-) 13 ] 2 (Me) Si (CH 2 ) 3 SH,
[(XO- (CH 2 -CH (CH 3 ) O-) 14 ] 2 (Me) Si (CH 2 ) 3 SH,
[(XO- (CH 2 -CH (CH 3 ) O-) 15 ] 2 (Me) Si (CH 2 ) 3 SH,
[(XO- (CH 2 -CH (CH 3 ) O-) 16 ] 2 (Me) Si (CH 2 ) 3 SH,
[(XO- (CH 2 -CH (CH 3 ) O-) 17 ] 2 (Me) Si (CH 2 ) 3 SH,
[(XO- (CH 2 -CH (CH 3 ) O-) 18 ] 2 (Me) Si (CH 2 ) 3 SH,
[(XO- (CH 2 -CH (CH 3 ) O-) 19 ] 2 (Me) Si (CH 2 ) 3 SH,
[(XO- (CH 2 -CH (CH 3 ) O-) 20 ] 2 (Me) Si (CH 2 ) 3 SH,
[(XO- (CH 2 -CH (CH 3 ) O-) 2 ] (Me) 2 Si (CH 2 ) 3 SH,
[(XO- (CH 2 -CH (CH 3 ) O-) 3 ] (Me) 2 Si (CH) 3 SH,
[(XO- (CH 2 -CH (CH 3 ) O-) 4 ] (Me) 2 Si (CH 2 ) 3 SH,
[(XO- (CH 2 -CH (CH 3 ) O-) 5 ] (Me) 2 Si (CH) 3 SH,
[(XO- (CH 2 -CH (CH 3 ) O-) 6 ] (Me) 2 Si (CH 2 ) 3 SH,
[(XO- (CH 2 -CH (CH 3 ) O-) 7 ] (Me) 2 Si (CH) 3 SH,
[(XO- (CH 2 -CH (CH 3 ) O-) 8 ] (Me) 2 Si (CH 2 ) 3 SH,
[(XO- (CH 2 -CH (CH 3 ) O-) 9 ] (Me) 2 Si (CH 2 ) 3 SH,
[(XO- (CH 2 -CH (CH 3 ) O-) 10 ] (Me) 2 Si (CH 2 ) 3 SH,
[(XO- (CH 2 -CH (CH 3 ) O-) 11 ] (Me) 2 Si (CH 2 ) 3 SH,
[(XO- (CH 2 -CH (CH 3 ) O-) 12 ] (Me) 2 Si (CH 2 ) 3 SH,
[(XO- (CH 2 -CH (CH 3 ) O-) 13 ] (Me) 2 Si (CH 2 ) 3 SH,
[(XO- (CH 2 -CH (CH 3 ) O-) 14 ] (Me) 2 Si (CH 2 ) 3 SH,
[(XO- (CH 2 -CH (CH 3 ) O-) 15 ] (Me) 2 Si (CH 2 ) 3 SH,
[(XO- (CH 2 -CH (CH 3 ) O-) 16 ] (Me) 2 Si (CH 2 ) 3 SH,
[(XO- (CH 2 -CH (CH 3 ) O-) 17 ] (Me) 2 Si (CH 2 ) 3 SH,
[(XO- (CH 2 -CH (CH 3 ) O-) 18 ] (Me) 2 Si (CH 2 ) 3 SH,
[(XO- (CH 2 -CH (CH 3 ) O-) 19 ] (Me) 2 Si (CH 2 ) 3 SH or
[(XO- (CH 2 -CH (CH 3 ) O-) 20 ] (Me) 2 Si (CH 2 ) 3 SH.
Das Organosilan der allgemeinen Formel I kann aus einem Gemisch von Verbindungen von Organosilanen der allgemeinen Formel I bestehen. Die Gemische können Verbindungen von Organosilanen mit denselben oder unterschiedlichen m enthalten. Die Gemische von Organosilanen können Verbindungen mit denselben oder unterschiedlichen Y-Gruppen enthalten. Die Gemische können Verbindungen von Organosilanen mit denselben oder unterschiedlichen R1-, R2- , R3- oder R4-Gruppen enthalten.The organosilane of the general formula I can consist of a mixture of compounds of organosilanes of the general formula I. The mixtures may contain compounds of organosilanes having the same or different m. The mixtures of organosilanes may contain compounds having the same or different Y groups. The mixtures may contain compounds of organosilanes having the same or different R 1 , R 2 , R 3 or R 4 groups.
Aus den Organosilanen der allgemeinen Formel I können durch Wasserzugabe Kondensationsprodukte, das heisst Oligo- und Polysiloxane gebildet werden. Die Oligo- und Polysiloxane können durch Oligomerisierung oder Cooligomerisierung der entsprechenden Alkoxysilanverbindungen der allgemeinen Formel I durch Wasserzugabe und dem Fachmann auf diesem Gebiet bekannte Additivzugabe und Verfahrensweise erhalten werden. Die so entstandenen Oligo- bzw. Polymerisierungsprodukte können in den Organsilanverbindungen der allgemeinen Formel I enthalten sein.From the organosilanes of the general formula I condensation products, ie oligo- and polysiloxanes can be formed by addition of water. The oligo- and polysiloxanes may be obtained by oligomerization or co-oligomerization of the corresponding alkoxysilane compounds of general formula I by addition of water and additive addition and procedure known to those skilled in the art. The resulting oligo- or polymerization products can in the Organosilane compounds of general formula I may be included.
Das Organosilan der allgemeinen Formel I kann auch ein Oligo- oder Polymerisierungsprodukt der Organosilanverbindung der allgemeinen Formel I sein. Das Organosilan der allgemeinen Formel I kann ein Gemisch aus Oligo- oder Polymerisierungsprodukten der Organosilanverbindung der allgemeinen Formel I und unkondensierter Organosilanverbindung der allgemeinen Formel I sein.The organosilane of the general formula I may also be an oligo- or polymerization product of the organosilane compound of the general formula I. The organosilane of general formula I may be a mixture of oligo- or polymerization products of the organosilane compound of general formula I and uncondensed organosilane compound of general formula I.
Das Organosilan der allgemeinen Formel I kann sowohl in reiner Form als auch aufgezogen auf einen inerten organischen oder anorganischen Träger, sowie vorreagiert mit einem organischen oder anorganischen Träger dem Mischprozeß zugegeben werden. Bevorzugte Trägermaterialien können gefällte oder pyrogene Kieselsäuren, Wachse, Thermoplaste, natürliche oder synthetische Silikate, natürliche oder synthetische Oxide, speziell Aluminiumoxid, oder Ruße sein. Desweiteren können die Organosilane der allgemeinen Formel I auch vorreagiert mit dem einzusetzenden Füllstoff dem Mischprozeß zugegeben werden.The organosilane of the general formula I can be added to the mixing process both in pure form and supported on an inert organic or inorganic carrier, as well as prereacted with an organic or inorganic carrier. Preferred support materials may be precipitated or fumed silicas, waxes, thermoplastics, natural or synthetic silicates, natural or synthetic oxides, especially alumina, or carbon blacks. Furthermore, the organosilanes of the general formula I can also be added to the mixing process in a pre-reacted form with the filler to be used.
Als Füllstoffe können für die erfindungsgemäßen Kautschukmischungen die folgenden Füllstoffe eingesetzt werden:
- Ruße: Die hierbei zu verwendenden Ruße sind nach dem Flammruß-, Furnace-, Gasruß oder Thermal-Verfahren hergestellt und besitzen BET-Oberflächen von 20 bis 200 m2/g. Die Ruße können gegebenenfalls auch Heteroatome, wie zum Beispiel Si, enthalten.
- Amorphe Kieselsäuren, hergestellt zum Beispiel durch Fällung von Lösungen von Silikaten oder Flammenhydrolyse von Siliciumhalogeniden mit spezifischen Oberflächen von 5 bis 1000 m2/g, vorzugsweise 20 bis 400 m2/g (BET-Oberfläche) und mit Primärteilchengrößen von 10 bis 400 nm. Die Kieselsäuren können gegebenenfalls auch als Mischoxide mit anderen Metalloxiden, wie Al-, Mg-, Ca-, Ba-, Zn- und Titanoxiden, vorliegen.
- Synthetische Silikate, wie Aluminiumsilikat, Erdalkalisilikate, wie Magnesiumsilikat oder Calciumsilikat, mit BET-Oberflächen von 20 bis 400 m2/g und Primärteilchendurchmessern von 10 bis 400 nm.
- Synthetische oder natürliche Aluminiumoxide und - hydroxide.
- Natürliche Silikate, wie Kaolin und andere natürlich vorkommende Kieselsäuren.
- Glasfaser und Glasfaserprodukte (Matten, Stränge) oder Mikroglaskugeln.
- Carbon blacks: The carbon blacks to be used in this case are produced by the flame black, furnace, gas black or thermal process and have BET surface areas of from 20 to 200 m 2 / g. Optionally, the carbon blacks may also contain heteroatoms, such as Si.
- Amorphous silicas prepared, for example, by precipitation of solutions of silicates or flame hydrolysis of silicon halides specific surface areas of from 5 to 1000 m 2 / g, preferably from 20 to 400 m 2 / g (BET surface area) and with primary particle sizes of from 10 to 400 nm. The silicas may also be used as mixed oxides with other metal oxides such as Al, Mg and , Ca, Ba, Zn and titanium oxides.
- Synthetic silicates, such as aluminum silicate, alkaline earth silicates, such as magnesium silicate or calcium silicate, with BET surface areas of 20 to 400 m 2 / g and primary particle diameters of 10 to 400 nm.
- Synthetic or natural aluminas and hydroxides.
- Natural silicates, such as kaolin and other naturally occurring silicas.
- Glass fiber and glass fiber products (mats, strands) or glass microspheres.
Bevorzugt können amorphe Kieselsäuren, hergestellt durch Fällung von Lösungen von Silikaten, mit BET-Oberflächen von 20 bis 400 m2/g in Mengen von 5 bis 150 Gew.-Teilen, jeweils bezogen auf 100 Teile Kautschuk, eingesetzt werden.Amorphous silicas prepared by precipitation of solutions of silicates having BET surface areas of from 20 to 400 m 2 / g in amounts of from 5 to 150 parts by weight, based in each case on 100 parts of rubber, may preferably be used.
Für die Herstellung der erfindungsgemäßen Kautschukmischungen können neben Naturkautschuk auch Synthesekautschuke eingesetzt werden. Bevorzugte Synthesekautschuke sind beispielsweise bei
- Polybutadien (BR),
- Polyisopren (IR),
- Styrol/Butadien-Copolymerisate, beispielsweise Emulsions-SBR (E-SBR) oder Lösungs-SBR (L-SBR), bevorzugt mit Styrolgehalten von 1 bis 60, besonders bevorzugt 5 bis 50 Gew.-% (SBR),
- Chloropren (CR),
- Isobutylen/Isopren-Copolymerisate (IIR),
- Butadien/Acrylnitril-Copolymere mit Acrylnitrilgehalten von 5 bis 60, vorzugsweise 10 bis 50 Gew.-% (NBR),
- teilhydrierter oder vollständig hydrierter NBR-Kautschuk (HNBR),
- Ethylen/Propylen/Dien-Copolymerisate (EPDM),
- oben genannte Kautschuke, die zusätzlich funktionelle Gruppen besitzen, wie z.B. Carboxy- , Silanol- oder Epoxygruppen, beispielsweise Epoxidierter NR, Carboxy-funktionalisierter NBR oder Silanol- (-SiOH) bzw. Siloxyfunktionalisierter (-Si-OR) SBR,
- Polybutadiene (BR),
- Polyisoprene (IR),
- Styrene / butadiene copolymers, for example emulsion SBR (E-SBR) or solution SBR (L-SBR), preferably with styrene contents of 1 to 60, more preferably 5 to 50 wt.% (SBR),
- Chloroprene (CR),
- Isobutylene / isoprene copolymers (IIR),
- Butadiene / acrylonitrile copolymers having acrylonitrile contents of 5 to 60, preferably 10 to 50 wt .-% (NBR),
- partially hydrogenated or fully hydrogenated NBR rubber (HNBR),
- Ethylene / propylene / diene copolymers (EPDM),
- abovementioned rubbers which additionally have functional groups, for example carboxy, silanol or epoxy groups, for example epoxidized NR, carboxy-functionalized NBR or silanol (-SiOH) or siloxy-functionalized (-Si-OR) SBR,
Die erfindungsgemäßen Kautschukrohmischungen und - vulkanisate können weitere Kautschukhilfsmittel, wie Reaktionsbeschleuniger, Alterungsschutzmittel, Wärmestabilisatoren, Lichtschutzmittel, Ozonschutzmittel, Verarbeitungshilfsmittel, Weichmacher, Tackifier, Treibmittel, Farbstoffe, Pigmente, Wachse, Streckmittel, organische Säuren, Verzögerer, Metalloxide oder Aktivatoren, wie Triethanolamin oder Hexantriol, enthalten.The rubber raw mixtures and vulcanizates according to the invention may contain further rubber auxiliaries, such as reaction accelerators, aging inhibitors, heat stabilizers, light stabilizers, antiozonants, processing aids, plasticizers, tackifiers, blowing agents, dyes, pigments, waxes, extenders, organic acids, Retarder, metal oxides or activators, such as triethanolamine or hexanetriol.
Weitere Kautschukhilfsmittel können Polyalkylenglykole sein. Polyalkylenglykole können Polyethylenglykole, Polypropylenglykole oder/und Polybutylenglykole sein.Other rubber auxiliaries may be polyalkylene glycols. Polyalkylene glycols may be polyethylene glycols, polypropylene glycols and / or polybutylene glycols.
Die Polyalkylenglykole können ein Molekulargewicht zwischen 50 und 50.000 g/mol, bevorzugt zwischen 50 und 20.000 g/mol, besonders bevorzugt zwischen 200 und 10.000 g/mol, ganz besonders bevorzugt zwischen 400 und 6.000 g/mol, außerordentlich bevorzugt zwischen 500 und 3.000 g/mol, haben.The polyalkylene glycols may have a molecular weight between 50 and 50,000 g / mol, preferably between 50 and 20,000 g / mol, more preferably between 200 and 10,000 g / mol, most preferably between 400 and 6,000 g / mol, most preferably between 500 and 3,000 g / mol, have.
Die Polyethylenglykole können kohlenwasserstoffterminiertes Polyethylenglykol Alk-O-(CH2-CH2-O)yI-H bzw. Alk-(CH2-CH2-O)yI-Alk sein, mit yI = 2-25, bevorzugt yI = 2-15, besonders bevorzugt yI = 3-8 und 10-14, ganz besonders bevorzugt yI = 3-6 und 10-13, und Alk gleich ein verzweigter oder unverzweigter, unsubstituierter oder substituierter, gesättigter oder ungesättigter Kohlenwasserstoff mit 1 bis 35, bevorzugt 4 bis 25, besonders bevorzugt 6 bis 20, ganz besonders bevorzugt 10 bis 20, außerordentlich bevorzugt 11 bis 14, Kohlenstoffatomen.The polyethylene glycols may be hydrocarbon-terminated polyethylene glycol Alk-O- (CH 2 -CH 2 -O) yl -H or alk- (CH 2 -CH 2 -O) yl- alk, with y I = 2-25, preferably y I = 2-15, more preferably y I = 3-8 and 10-14, most preferably y I = 3-6 and 10-13, and Alk is a branched or unbranched, unsubstituted or substituted, saturated or unsaturated hydrocarbon with 1 to 35, preferably 4 to 25, more preferably 6 to 20, most preferably 10 to 20, most preferably 11 to 14, carbon atoms.
Die Polypropylenglykole können kohlenwasserstoffterminiertes Polypropylenglykol Alk-O-(CH2-CH(CH3)-O)yI-H bzw. Alk-O-(CH2-CH(CH3)-O)yI-Alk sein, wobei yI und Alk die oben genannte Bedeutung haben.The polypropylene glycols may be hydrocarbon-terminated polypropylene glycol Alk-O- (CH 2 -CH (CH 3 ) -O) yl -H or Alk-O- (CH 2 -CH (CH 3 ) -O) yl- alk, where y is I and Alk have the abovementioned meaning.
Die Polybutylenglykole können kohlenwasserstoffterminiertes Polybutylenglykol Alk-O-(CH2-CH2-CH2-CH2-O)yI-H, Alk-O- (CH2-CH (CH3) -CH2-O)yI-H, Alk-O-(CH2-CH2-CH2-CH2-O)yI-Alk oder Alk-O-(CH2-CH (CH3)-CH2-O)yI-Alk sein, wobei yI und Alk die oben genannte Bedeutung haben.The polybutylene glycols may include hydrocarbon- terminated polybutylene glycol , Alk-O- (CH 2 -CH 2 -CH 2 -CH 2 -O) yl -H, Alk-O- (CH 2 -CH (CH 3 ) -CH 2 -O) yl -H , Alk-O- (CH 2 -CH 2 -CH 2 -CH 2 -O) yl -alk or Alk-O- (CH 2 -CH (CH 3 ) -CH 2 -O) yl- alk, where y I and Alk have the abovementioned meaning.
Polyalkylenglykole können polyethylenglykol-, polypropylenglykol-, polybutylenglykol-, oder mit Mischungen davon verethertes Neopentylglykol HO-CH2-C(Me)2-CH2-OH, Pentaerythrit C(CH2-OH)4 oder Trimethylolpropan CH3-CH2-C(CH2-OH)3, wobei die Wiederholeinheiten von Ethylenglykol, Propylenglykol oder/und Butylenglykol in den veretherten Polyalkoholen zwischen 2 und 100, bevorzugt zwischen 2 und 50, besonders bevorzugt zwischen 3 und 30, ganz besonders bevorzugt zwischen 3 und 15, sind, sein.Polyalkylene glycols may be polyethylene glycol, polypropylene glycol, polybutylene glycol, or etherified with mixtures thereof neopentyl glycol HO-CH 2 -C (Me) 2 -CH 2 -OH, pentaerythritol C (CH 2 -OH) 4 or trimethylolpropane CH 3 -CH 2 - C (CH 2 -OH) 3 , where the repeat units of ethylene glycol, propylene glycol or / and butylene glycol in the etherified polyalcohols are between 2 and 100, preferably between 2 and 50, more preferably between 3 and 30, very particularly preferably between 3 and 15, are, be.
Die Kautschukhilfsmittel können in bekannten Mengen, die sich unter anderem nach dem Verwendungszweck richten, eingesetzt werden. Übliche Mengen sind zum Beispiel Mengen von 0,1 bis 50 Gew.-%, vorzugsweise 0,1 bis 30 Gew.-%, bezogen auf Kautschuk. Als Vernetzer können Schwefel oder schwefelspendende Substanzen eingesetzt werden. Die erfindungsgemäßen Kautschukmischungen können darüber hinaus Vulkanisationsbeschleuniger enthalten. Beispielsweise können als geeignete Vulkanisationsbeschleuniger Mercaptobenzthiazole, Sulfenamide, Guanidine, Thiurame, Dithiocarbamate, Thioharnstoffe und Thiocarbonate eingesetzt werden. Die Vulkanisationsbeschleuniger und Schwefel können in Mengen von 0,1 bis 10 Gew.-%, bevorzugt 0.1 bis 5 Gew.-%, bezogen auf Kautschuk, eingesetzt werden.The rubber auxiliaries can be used in known amounts, which depend inter alia on the intended use. Usual amounts are, for example, amounts of 0.1 to 50 wt .-%, preferably 0.1 to 30 wt .-%, based on rubber. As crosslinkers sulfur or sulfur-donating substances can be used. The rubber mixtures according to the invention may additionally contain vulcanization accelerators. For example, mercaptobenzothiazoles, sulfenamides, guanidines, thiurams, dithiocarbamates, thioureas and thiocarbonates can be used as suitable vulcanization accelerators. The vulcanization accelerators and sulfur can be used in amounts of 0.1 to 10 wt .-%, preferably 0.1 to 5 wt .-%, based on rubber.
Ein weiterer Gegenstand der Erfindung ist ein Verfahren zur Herstellung der erfindungsgemäßen Kautschukmischung, welches dadurch gekennzeichnet ist, daß man den Kautschuk, Füllstoff, gegebenenfalls weitere Kautschukhilfsmittel und mindestens ein Organosilan der allgemeinen Formel I mischt.Another object of the invention is a process for the preparation of the rubber mixture according to the invention, which is characterized by mixing the rubber, filler, optionally further rubber auxiliaries and at least one organosilane of the general formula I.
Die Vulkanisation der erfindungsgemäßen Kautschukmischungen kann bei Temperaturen von 100 bis 200°C, bevorzugt 130 bis 180 °C, gegebenenfalls unter Druck von 10 bis 200 bar erfolgen. Die Abmischung der Kautschuke mit dem Füllstoff, gegebenenfalls Kautschukhilfsmitteln und dem Organosilan der allgemeinen Formel I kann in bekannten Mischaggregaten, wie Walzen, Innenmischern und Mischextrudern, durchgeführt werden.The vulcanization of the rubber mixtures according to the invention can be carried out at temperatures of 100 to 200 ° C, preferably 130 to 180 ° C, optionally under pressure of 10 to 200 bar. The blending of the rubbers with the filler, optionally rubber auxiliaries and the organosilane of the general formula I can be carried out in known mixing units, such as rollers, internal mixers and mixing extruders.
Die erfindungsgemäßen Kautschukmischungen können zur Herstellung von Formkörpern, zum Beispiel für die Herstellung von Luftreifen, Reifenlaufflächen, Kabelmänteln, Schläuchen, Treibriemen, Förderbändern, Walzenbelägen, Reifen, Schuhsohlen, Dichtungsringen und Dämpfungselementen, verwendet werden.The rubber blends of the present invention may be used to make molded articles, for example, for the manufacture of pneumatic tires, tire treads, cable jackets, hoses, power belts, conveyor belts, roller coverings, tires, shoe soles, gaskets and damping elements.
Die erfindungsgemäßen Kautschukmischungen zeigen einen verbesserten Weiterreißwiderstand.The rubber mixtures according to the invention show an improved tear propagation resistance.
In einem Autoklaven mit Glasdoppelmantel und Hastelloy C22 Deckel + Armaturen (Buechi AG) werden bei Raumtemperatur 37,5 g getrocknetes NaSH und 600 ml trockenes Ethanol vorgelegt. Die Suspension wird erwärmt und 20 min bei 50°C gerührt. Es wird eine Mischung aus 100 g 3-Chlorpropyl(dimethylethoxysilan) und 5 g 3-Chlorpropyl(dimethylchlorsilan) mit einer Druckbürette zu der Suspension gegeben. Zu der Mischung werden weitere 200 ml Ethanol gegeben und unter Rühren auf 93-96°C erwärmt. Die Temperatur wird für 180 min gehalten. Die Mischung wird anschließend auf Raumtemperatur abgekühlt.37.5 g of dried NaSH and 600 ml of dry ethanol are initially charged at room temperature in an autoclave with glass double jacket and Hastelloy C22 lid + fittings (Buechi AG). The suspension is heated and stirred at 50 ° C for 20 min. A mixture of 100 g of 3-chloropropyl (dimethylethoxysilane) and 5 g of 3-chloropropyl (dimethylchlorosilane) is added to the suspension using a pressure burette. Another 200 ml of ethanol are added to the mixture and heated to 93-96 ° C. with stirring. The temperature is held for 180 min. The mixture is then cooled to room temperature.
Die entstandene Suspension wird filtriert und der Filterkuchen mit Toluol gewaschen. Das Filtrat wird am Rotationsverdampfer vom Lösungsmittel befreit. Die erhaltene Suspension wird filtriert, der Filterkuchen mit Toluol gewaschen und das Filtrat nochmals am Rotationsverdampfer vom Toluol befreit.The resulting suspension is filtered and the filter cake washed with toluene. The filtrate is freed from the solvent on a rotary evaporator. The resulting suspension is filtered, the filter cake washed with toluene and the filtrate freed again from the toluene on a rotary evaporator.
Es werden 88,3 g flüssiges, farbloses Produkt erhalten.There are obtained 88.3 g of liquid, colorless product.
In einen 2000 ml Vierhalskolben werden 700 ml Ethanol mit 337 g getrocknetem Na2S4 (1,94 mol) und 700 g 3-Chlorpropyl(dimethylethoxysilan) (3,88 mol) eingewogen und unter Rühren zum Sieden erhitzt. Die Reaktionslösung wird für 270 min unter Rückfluß gekocht. Der Suspension werden 3 g 3-Chlorpropyl(dimethylethoxysilan) zugesetzt und für weitere 30 min auf Rückfluß erhitzt.In a 2000 ml four-necked flask, 700 ml of ethanol with 337 g of dried Na 2 S 4 (1.94 mol) and 700 g of 3-chloropropyl (dimethylethoxysilane) (3.88 mol) weighed and heated to boiling with stirring. The reaction solution is refluxed for 270 minutes. 3 g of 3-chloropropyl (dimethylethoxysilane) are added to the suspension and heated to reflux for a further 30 min.
Die Suspension wird abgekühlt, filtriert und der Rückstand mit Ethanol gewaschen. Das Filtrat wird am Rotationsverdampfer bei 20-400 mbar und 60-90°C vom Lösungsmittel befreit und nochmals filtriert. Es werden 769,2 g einer orangen Flüssigkeit isoliert.The suspension is cooled, filtered and the residue washed with ethanol. The filtrate is freed from the solvent on a rotary evaporator at 20-400 mbar and 60-90 ° C and filtered again. There are 769.2 g of an orange liquid isolated.
1. 1H-NMR
3-Chlorpropyl(dimethyl-ethoxysilan)
2. 29Si-NMR
Das Vergleichsbeispiel 2 enthält 1,6 mol-% dimerisiertes [(EtO)Me2Si-CH2-CH2-CH2-]2Sx .
1. 1 H NMR
3-chloropropyl (dimethyl-ethoxysilane)
2. 29 Si NMR
Comparative Example 2 contains 1.6 mol% of dimerized [(EtO) Me 2 Si-CH 2 -CH 2 -CH 2 -] 2 S x .
Für die Analyse des Vergleichsproduktes wird ein DRX 500 NMR-Gerät der Firma Bruker gemäß den dem Fachmann bekannten Regeln und Bedienvorschriften verwendet. Die Messfrequenzen sind 99,35 MHz für 29Si-Kerne und 500,13 MHz für 1H-Kerne. Als Referenz dient Tetramethylsilan (TMS).For the analysis of the comparison product, a Bruker DRX 500 NMR instrument is used according to the rules and operating instructions known to the person skilled in the art. The measurement frequencies are 99.35 MHz for 29 Si cores and 500.13 MHz for 1 H cores. The reference is tetramethylsilane (TMS).
Die Analyse von Bis(alkoxysilylorganyl)polysulfiden und Mercaptoorganyl(alkoxysilanen) und deren Gemischen wird beispielsweise beschrieben in
Die für die Kautschukmischungen verwendete Rezeptur ist in der folgenden Tabelle 1 angegeben. Dabei bedeutet die Einheit phr Gewichtsanteile, bezogen auf 100 Teile des eingesetzten Rohkautschuks. Die erfindungsgemäßen Silane werden gewichtsgleich dosiert. Das allgemeine Verfahren zur Herstellung von Kautschukmischungen und deren Vulkanisate ist in dem Buch: "
Die Kopplungsreagenzien Si 69, ein Bis-(triethoxy-silylpropyl)tetrasulfid (TESPT), und VP Si 263, ein 3-Mercaptopropyl(triethoxysilan) (MPTES), sind Verkaufsprodukte der Degussa AG. Das Kopplungsreagenz VP Si 208, ein Octylsilyltriethoxysilan, ist als Alkylsilan ein Verarbeitungshilfsmittel und ist ein Verkaufsprodukt der Degussa AG.
Bei dem Polymer VSL 5025-1 handelt es sich um ein in Lösung polymerisiertes SBR-Copolymer der Bayer AG, mit einem Styrolgehalt von 25 Gew.-% und einem Butadiengehalt von 75 Gew.-%. Das Copolymer enthält 37,5 phr Öl und weist eine Mooney-Viskosität (ML 1+4/100°C) von 50 auf.The polymer VSL 5025-1 is a polymerized in solution SBR copolymer of Bayer AG, with a styrene content of 25 wt .-% and a butadiene content of 75 wt .-%. The copolymer contains 37.5 phr of oil and has a Mooney viscosity (ML 1 + 4/100 ° C) of 50.
Bei dem Polymer Buna CB 24 handelt es sich um ein cis-1,4-Polybutadien (Neodymtyp) der Bayer AG, mit cis-1,4-Gehalt von mindestens 96 % und einer Mooney-Viskosität von 44±5.The polymer Buna CB 24 is a cis-1,4-polybutadiene (neodymium type) from Bayer AG, with a cis-1,4 content of at least 96% and a Mooney viscosity of 44 ± 5.
Ultrasil 7000 GR ist eine leicht dispergierbare Kieselsäure der Degussa AG und besitzt eine BET-Oberfläche von 170 m2/g.Ultrasil 7000 GR is an easily dispersible silica from Degussa AG and has a BET surface area of 170 m 2 / g.
Als aromatisches öl wird Naftolen ZD der Chemetall verwendet, bei Vulkanox 4020 handelt es sich um PPD der Bayer AG und Protektor G3108 ist ein Ozonschutzwachs der Paramelt B.V.. Vulkacit D (DPG) und Vulkacit CZ (CBS) sind Handelsprodukte der Bayer AG. Perkacit TBzTD (Tetrabenzylthiuramtetrasulfid) ist ein Produkt von Flexsys N.V..The aromatic oil used is Naftolen ZD from Chemetall, Vulkanox 4020 is PPD from Bayer AG and Protektor G3108 is an Ozonschutzwachs the Paramelt B.V .. Vulkacit D (DPG) and Vulkacit CZ (CBS) are commercial products of Bayer AG. Perkacit TBzTD (tetrabenzyl thiuram tetrasulfide) is a product of Flexsys N.V.
Die Kautschukmischungen werden in einem Innenmischer entsprechend der Mischvorschrift in Tabelle 2 hergestellt.
Drehzahlvariation halten
5* links, 5* rechts einschneiden und 6* bei weitem Walzenspalt (6 mm) und 3* bei engem Walzenspalt (3 mm)
Fell ausziehen.
Keep the speed variation
5 * left, 5 * right cut and 6 * wide nip (6 mm) and 3 * narrow nip (3 mm)
Take off the coat.
In Tabelle 3 sind die Methoden für die Gummitestung zusammengestellt.
Die Tabelle 4 zeigt die Ergebnisse der gummitechnischen Prüfung.
Daß Mercaptosilane eine höhere Kopplungsausbeute und damit Verstärkung als ein Polysulfid besitzen, ist aus
In
Im Gegensatz zu den vorgenannten Beobachtungen bei den Polysulfiden aus
In einem Rundkolben werden 86,64 g HS-CH2-CH2-CH2-Si(Me)(OMe)2, 163,29 g Polypropylenglykolmonobutylether (CAS 9003-13-8, Firma Aldrich, Mw = 340 g/mol) und 0,23 g p-Toluolsulfonsäure gemischt. Die Mischung wird am Rotationsverdampfer bei 150-155°C Ölbadtemperatur und 100-400 mbar für 6,5 h behandelt. Der freiwerdende, flüchtige Alkohol wird abdestilliert. Die isolierte Auswaage des Produktes beträgt 236 g.In a round bottomed flask, 86.64 g of HS-CH 2 -CH 2 -CH 2 -Si (Me) (OMe) 2 , 163.29 g of polypropylene glycol monobutyl ether (CAS 9003-13-8, Aldrich, M w = 340 g / mol) and 0.23 g of p-toluenesulfonic acid. The mixture is treated on a rotary evaporator at 150-155 ° C oil bath temperature and 100-400 mbar for 6.5 h. The liberated, volatile alcohol is distilled off. The isolated weight of the product is 236 g.
In einem Rundkolben werden 86,64 g HS-CH2-CH2-CH2-Si(Me)(OMe)2 , 480,03 g Polypropylenglykolmonobutylether (CAS 9003-13-8, Firma Aldrich, Mw= 1000 g/mol) und 0,23 g p-Toluolsulfonsäure gemischt. Die Mischung wird am Rotationsverdampfer bei 145-155°C Ölbadtemperatur und 100-400 mbar für 4,5 h behandelt. Der freiwerdende, flüchtige Alkohol wird abdestilliert. Die isolierte Auswaage des Produktes beträgt 552 g.86.64 g of HS-CH 2 -CH 2 -CH 2 -Si (Me) (OMe) 2 , 480.03 g of polypropylene glycol monobutyl ether (CAS 9003-13-8, Aldrich, M w = 1000 g / mol) and 0.23 g of p-toluenesulfonic acid. The mixture is treated on a rotary evaporator at 145-155 ° C oil bath temperature and 100-400 mbar for 4.5 h. The liberated, volatile alcohol is distilled off. The isolated weight of the product is 552 g.
In einem Rundkolben werden 86,62 g HS-CH2-CH2-CH2-Si(Me) (OMe)2, 147 g Polyethylenglykol-mono-2-ethylhexyl-ether (Aduxol HEX-04, CAS 26468-86-0, Firma Schärer & Schläpfer AG) und 0,5 g Ti(OBu)4 gemischt. Die Mischung wird am Rotationsverdampfer bei 125-135°C Ölbadtemperatur und 150-300 mbar für 4,5 h behandelt. Der freiwerdende, flüchtige Alkohol wird abdestilliert. Die isolierte Auswaage des Produktes beträgt 214 g.86.62 g of HS-CH 2 -CH 2 -CH 2 -Si (Me) (OMe) 2 , 147 g of polyethylene glycol mono-2-ethylhexyl ether (aduxol HEX-04, CAS 26468-86- 0, Schärer & Schläpfer AG) and 0.5 g of Ti (OBu) 4 mixed. The mixture is treated on a rotary evaporator at 125-135 ° C oil bath temperature and 150-300 mbar for 4.5 h. The liberated, volatile alcohol is distilled off. The isolated weight of the product is 214 g.
In einem Rundkolben werden 50 g HS-CH2-CH2-CH2-Si(Me) (OEt)2 , 45,7 g Diethylenglykol-monohexyl-ether (CAS 112-59-4, bezogen über Merck/VWR International) und 0,23 g Ti(OBu)4 gemischt. Die Mischung wird am Rotationsverdampfer bei 130-135°C Ölbadtemperatur und 100-300 mbar für 6 h behandelt. Der freiwerdende, flüchtige Alkohol wird abdestilliert. Die Menge an isoliertem Produkt beträgt 80 g.In a round-bottom flask, 50 g of HS-CH 2 -CH 2 -CH 2 -Si (Me) (OEt) 2 , 45.7 g of diethylene glycol monohexyl ether (CAS 112-59-4, obtained from Merck / VWR International) and 0.23 g of Ti (OBu) 4 mixed. The mixture is treated on a rotary evaporator at 130-135 ° C oil bath temperature and 100-300 mbar for 6 h. The liberated, volatile alcohol is distilled off. The amount of isolated product is 80 g.
In einem Rundkolben werden 80 g HS-CH2-CH2-CH2-Si(Me)(OEt)2, 384,07 g Polypropylenglykolmonobutylether (CAS 9003-13-8, Firma Aldrich, Mw = 1000 g/mol) und 0,2 g p-Toluolsulfonsäure gemischt. Die Mischung wird am Rotationsverdampfer bei 145-155°C Ölbadtemperatur und 100-300 mbar für 6 h behandelt. Der freiwerdende, flüchtige Alkohol wird abdestilliert. Die Auswaage des erhaltenen Produktes beträgt 448 g.In a round-bottom flask, 80 g of HS-CH 2 -CH 2 -CH 2 -Si (Me) (OEt) 2 , 384.07 g of polypropylene glycol monobutyl ether (CAS 9003-13-8, Aldrich, M w = 1000 g / mol) and 0.2 g of p-toluenesulfonic acid. The mixture is treated on a rotary evaporator at 145-155 ° C oil bath temperature and 100-300 mbar for 6 h. The liberated, volatile alcohol is distilled off. The weight of the product obtained is 448 g.
In einem Rundkolben werden 50 g HS-CH2-CH2-CH2-Si(Me) (OEt)2 , 73,5 g Polyethylenglykol-mono-2-ethylhexyl-ether (Aduxol HEX-04, CAS 26468-86-0, Firma Schärer & Schläpfer AG) und 0,3 g Ti(OBu)4 gemischt. Die Mischung wird am Rotationsverdampfer bei 125-135°C Ölbadtemperatur und 150-300 mbar für 4,5 h behandelt. Der freiwerdende, flüchtige Alkohol wird abdestilliert. Die Auswaage an erhaltenem Produkt beträgt 108 g.In a round-bottom flask, 50 g of HS-CH 2 -CH 2 -CH 2 -Si (Me) (OEt) 2 , 73.5 g of polyethylene glycol mono-2-ethylhexyl ether (Aduxol HEX-04, CAS 26468-86- 0, Schärer & Schläpfer AG) and 0.3 g of Ti (OBu) 4 mixed. The mixture is treated on a rotary evaporator at 125-135 ° C oil bath temperature and 150-300 mbar for 4.5 h. The liberated, volatile alcohol is distilled off. The weight of the product obtained is 108 g.
HS-CH2-CH2-CH2-SiMe(OEt)2 wird in Anlehnung an
Die für die Kautschukmischungen verwendete Rezeptur ist in der folgenden Tabelle 5 angegeben. Die Mischungen unterscheiden sich in dem zugegebenen Kopplungsagens wie in Tabelle 6 angegeben. Die Mischvorschrift ist in Tabelle 2 aufgeführt.
In Tabelle 7 sind die Methoden für die Gummitestung zusammengestellt.
Die Tabelle 8 zeigt die Ergebnisse der gummitechnischen Prüfung.
Wie man anhand der Vulkanisatergebnisse erkennen kann, sind die Weiterreißwiderstände der Mischungen 8 bis 13 deutlich besser als die der Mischungen 5, 6, 7. Die Mischungen mit den Silanen mit langkettigem Alkohol als Substituenten zeigen ein verbessertes Weiterreißverhalten gegenüber den anderen Mischungen. Dies gilt sowohl gegenüber unsubstituiertem Mercaptosilan aus Mischung 5 (VP Si 263) als auch gegenüber den Mercaptosilanen mit einer Methylgruppe (Mischung 7) und mit zwei Methylgruppen (Mischung 6). Die Mischungen 8 bis 13 zeigen zudem noch verbesserte Mooney-Scorch Daten. Damit verbunden ist eine verbesserte Verarbeitungssicherheit, z.B. bei der Extrusion von Reifenlaufflächen oder dem Spritzgießen.As can be seen from the results of the vulcanization, the tear propagation resistances of the mixtures 8 to 13 are markedly better than those of the blends 5, 6, 7. The mixtures with the long-chain alcohol silanes as substituents show an improved tear propagation behavior compared to the other mixtures. This applies both to unsubstituted mercaptosilane from mixture 5 (VP Si 263) and to the mercaptosilanes with a methyl group (mixture 7) and with two methyl groups (mixture 6). Mixtures 8 through 13 also show improved Mooney Scorch data. This is associated with improved processing safety, for example in the extrusion of tire treads or injection molding.
Die für die kautschukmischungen verwendete Rezeptur ist in der folgenden Tabelle 9 angegeben. Die Mischungen unterscheiden sich in dem zugegebenen Kopplungsagens auf Basis equimolarer Dosierung wie in Tabelle 10 angegeben. Die Mischvorschrift ist in Tabelle 2 aufgeführt.
Die durchgeführten Prüfungen sind in Tabelle 7 aufgelistet. Die Tabelle 11 zeigt die Ergebnisse der gummitechnischen Prüfung.
Auch bei equimolarer Dosierung zeigen die Mischungen mit den Silanen aus den Beispielen 3 bis 8 Vorteile sowohl im Weiterreißverhalten als auch im Scorch-Verhalten gegenüber unsubstituiertem Mercaptosilan und Mercaptosilan mit einer Methyl- und zwei Ethoxygruppen.Even with equimolar metering, the mixtures with the silanes from Examples 3 to 8 show advantages both in the tear propagation behavior and in the scorch behavior towards unsubstituted mercaptosilane and mercaptosilane having one methyl and two ethoxy groups.
Claims (6)
- Rubber mixtures comprising rubber, fillers, optionally further rubber auxiliaries and at least one organosilane of the general formula I,
R1R2R3SiR4-SH (I)
where R1 is methyl or ethyl,
R2 is -O-(Y-O)m-X, where Y is a branched or unbranched, saturated or unsaturated divalent hydrocarbon group, X is a C1- to C9-alkyl group and m is 1-40,
R3 is methyl, ethyl, methoxy, ethoxy or R2
and R4 is a branched or unbranched, saturated or unsaturated, aliphatic, aromatic or mixed aliphatic/aromatic divalent C1-C12 hydrocarbon group. - Rubber mixtures according to Claim 1, characterized in that the organosilane of the general formula I comprises oligomerized or polymerized organosilanes of the general formula I.
- Rubber mixtures according to Claim 1, characterized in that the rubber auxiliary is a polyalkylene glycol.
- Process for the preparation of rubber mixtures according to Claim 1, characterized in that the rubber, filler, optionally further rubber auxiliaries and at least one organosilane of the general formula I are mixed.
- Use of the rubber mixture according to Claim 1 for the production of shaped articles.
- Use of the rubber mixture according to Claim 5 for the production of pneumatic tyres, tyre treads, cable sheathing, hoses, drive belts, conveyor belts, roller coverings, tyres, shoe soles, sealing rings and damping elements.
Priority Applications (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| SI200531368T SI1672017T1 (en) | 2004-12-18 | 2005-12-14 | Rubber compositions |
| PL05112090T PL1672017T3 (en) | 2004-12-18 | 2005-12-14 | Rubber compositions |
Applications Claiming Priority (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| DE102004061014A DE102004061014A1 (en) | 2004-12-18 | 2004-12-18 | rubber compounds |
Publications (3)
| Publication Number | Publication Date |
|---|---|
| EP1672017A2 EP1672017A2 (en) | 2006-06-21 |
| EP1672017A3 EP1672017A3 (en) | 2007-05-09 |
| EP1672017B1 true EP1672017B1 (en) | 2011-07-13 |
Family
ID=36061580
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| EP05112090A Active EP1672017B1 (en) | 2004-12-18 | 2005-12-14 | Rubber compositions |
Country Status (17)
| Country | Link |
|---|---|
| US (1) | US7384997B2 (en) |
| EP (1) | EP1672017B1 (en) |
| JP (1) | JP4420894B2 (en) |
| KR (1) | KR101222258B1 (en) |
| CN (1) | CN1789315B (en) |
| AT (1) | ATE516326T1 (en) |
| BR (1) | BRPI0506287B1 (en) |
| CA (1) | CA2530346C (en) |
| DE (1) | DE102004061014A1 (en) |
| ES (1) | ES2368568T3 (en) |
| MX (1) | MXPA05013860A (en) |
| MY (1) | MY139418A (en) |
| PL (1) | PL1672017T3 (en) |
| PT (1) | PT1672017E (en) |
| RU (1) | RU2404207C2 (en) |
| SI (1) | SI1672017T1 (en) |
| TW (1) | TWI389958B (en) |
Families Citing this family (29)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| DE102004030737A1 (en) * | 2004-06-25 | 2006-01-12 | Degussa Ag | Method and device for extracting substances from silane-modified fillers |
| DE102005057801A1 (en) * | 2005-01-20 | 2006-08-03 | Degussa Ag | mercaptosilanes |
| DE102005020535B3 (en) * | 2005-05-03 | 2006-06-08 | Degussa Ag | Preparation of mercapto organyl(alkoxysilane) comprises reaction of bis(alkoxysilylorganyl)polysulfide with hydrogen in the presence of an alcohol and a doped metal catalyst (containing e.g. (iron) compound and a doping component) |
| DE102005038791A1 (en) * | 2005-08-17 | 2007-02-22 | Degussa Ag | New organosilicon compounds based on triethanolamine, useful as components of rubber mixtures for making e.g. tires, tubes and cable coatings |
| DE102005060122A1 (en) * | 2005-12-16 | 2007-06-21 | Degussa Gmbh | Process for the preparation of (mercaptoorganyl) alkyl polyether silanes |
| DE102006004062A1 (en) * | 2006-01-28 | 2007-08-09 | Degussa Gmbh | rubber compounds |
| DE102006027235A1 (en) * | 2006-06-09 | 2008-01-17 | Evonik Degussa Gmbh | rubber compounds |
| DE102006033310A1 (en) * | 2006-07-17 | 2008-01-31 | Evonik Degussa Gmbh | Mixtures of silicon-containing coupling reagents |
| DE102006041356A1 (en) * | 2006-09-01 | 2008-03-20 | Evonik Degussa Gmbh | Process for the preparation of organosilanes |
| US7550524B2 (en) * | 2006-10-06 | 2009-06-23 | Momentive Performance Materials Inc. | Elastomer composition containing mercaptofunctional silane and process for making same |
| JP2008115326A (en) * | 2006-11-07 | 2008-05-22 | Sumitomo Rubber Ind Ltd | Rubber composition and tire having tread using the same |
| JPWO2008123306A1 (en) | 2007-03-27 | 2010-07-15 | 株式会社ブリヂストン | Method for producing rubber composition for tire tread |
| ITTO20080053A1 (en) * | 2008-01-24 | 2009-07-25 | Bridgestone Corp | MIXES INCLUDING TRIALCOSSIMERCAPTOALCHIL-SILANI |
| IT1394126B1 (en) * | 2008-10-22 | 2012-05-25 | Bridgestone Corp | TREAD COMPOUND FOR WINTER TIRES |
| JP2012520830A (en) * | 2009-03-20 | 2012-09-10 | エボニック デグサ ゲーエムベーハー | Method for producing organosilane |
| RU2496809C2 (en) * | 2009-04-28 | 2013-10-27 | Бриджстоун Корпорейшн | Pneumatic tyre |
| CN102884111B (en) * | 2009-12-03 | 2014-09-24 | 米其林集团总公司 | Filler blending for rubber formulations |
| EP2517899A1 (en) * | 2011-04-29 | 2012-10-31 | Lanxess Deutschland GmbH | Method for manufacturing rubber mixtures |
| WO2013058390A1 (en) | 2011-10-21 | 2013-04-25 | 株式会社ブリヂストン | Rubber composition and manufacturing method for same |
| DE102012205642A1 (en) * | 2012-04-05 | 2013-10-10 | Evonik Industries Ag | Mercaptosilane Russ mix |
| EP2736049B1 (en) * | 2012-11-23 | 2018-01-03 | Nexans | Storage-stable crosslinkable polymer mixture based on chlorinated polymer |
| US10011687B2 (en) * | 2013-05-08 | 2018-07-03 | Dow Silicones Corporation | Hydrophilic organosilanes |
| CN104558010B (en) * | 2015-01-27 | 2017-10-27 | 荆州市江汉精细化工有限公司 | A kind of mercaptosilane coupling agents preparation method of low VOC emission |
| DE102015224450A1 (en) * | 2015-12-07 | 2017-06-08 | Evonik Degussa Gmbh | rubber compounds |
| DE102017221277A1 (en) | 2017-11-28 | 2019-05-29 | Evonik Degussa Gmbh | Silane mixtures and process for their preparation |
| DE102018213774A1 (en) * | 2018-08-16 | 2020-02-20 | Evonik Operations Gmbh | rubber compounds |
| EP4023460A1 (en) * | 2020-12-30 | 2022-07-06 | The Goodyear Tire & Rubber Company | Sulfur curable rubber formulation, tire component and silica |
| CN115850826A (en) * | 2021-09-23 | 2023-03-28 | 中国石油化工股份有限公司 | Rubber auxiliary agent, rubber composition, vulcanized rubber, and preparation method and application thereof |
| CN114395173A (en) * | 2021-12-13 | 2022-04-26 | 东莞市瑞拓五金橡塑有限公司 | Formula and preparation method of high-temperature-resistant rubber sealing ring |
Family Cites Families (31)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| NL301721A (en) | 1962-12-21 | 1900-01-01 | ||
| US4076550A (en) | 1971-08-17 | 1978-02-28 | Deutsche Gold- Und Silber-Scheideanstalt Vormals Roessler | Reinforcing additive |
| US3997356A (en) | 1971-08-17 | 1976-12-14 | Deutsche Gold- Und Silber-Scheideanstalt Vormals Roessler | Reinforcing additive |
| US3978103A (en) | 1971-08-17 | 1976-08-31 | Deutsche Gold- Und Silber-Scheideanstalt Vormals Roessler | Sulfur containing organosilicon compounds |
| BE787691A (en) | 1971-08-17 | 1973-02-19 | Degussa | ORGANOSILICIC COMPOUNDS CONTAINING SULFUR |
| US3873489A (en) | 1971-08-17 | 1975-03-25 | Degussa | Rubber compositions containing silica and an organosilane |
| US4048206A (en) | 1975-04-22 | 1977-09-13 | Mikhail Grigorievich Voronkov | Process for the production of 1-organylsilatranes and carbofunctional derivatives thereof |
| DE2933345C2 (en) * | 1979-08-17 | 1983-01-20 | Degussa Ag, 6000 Frankfurt | Vulcanizable rubber mixture based on halogen-free rubbers and process for vulcanizing these rubber mixtures |
| DE19512543C2 (en) * | 1995-04-06 | 1999-02-25 | Continental Ag | Tread compound for pneumatic tires, process for their manufacture and their use |
| KR100473410B1 (en) * | 1995-08-16 | 2005-09-02 | 오시 스페셜티스, 인코포레이티드 | Stable Silane Composition Supported by Silica Carriers |
| DE19544469A1 (en) | 1995-09-23 | 1997-03-27 | Degussa | Vulcanizable rubber compounds containing organosilane compounds and silica and process for producing them |
| FR2743564A1 (en) | 1996-01-11 | 1997-07-18 | Michelin & Cie | RUBBER COMPOSITIONS FOR SILICA-BASED TIRE CASINGS CONTAINING A REINFORCING ADDITIVE BASED ON A FUNCTIONALIZED POLYORGANOSILOXANE AND AN ORGANOSILANE COMPOUND. |
| US5914364A (en) | 1996-03-11 | 1999-06-22 | The Goodyear Tire & Rubber Company | Silica reinforced rubber composition and tire with tread |
| US5780538A (en) | 1996-03-11 | 1998-07-14 | The Goodyear Tire & Rubber Company | Silica reinforced rubber composition and tire with tread |
| AR016766A1 (en) * | 1997-07-11 | 2001-08-01 | Rhodia Chimie Sa | A PROCEDURE FOR THE PREPARATION OF POLYGANOSILOXANS (POS) WITH TIOL FUNCTIONS, AND THE POSSIBLE TO BE OBTAINED BY MEANS OF THIS PROCEDURE |
| PT958298E (en) * | 1997-08-21 | 2003-09-30 | Crompton Corp | MERCAPTHOSILAN COUPLING AGENTS BLOCKED FOR REINFORCED RUBBER |
| JPH11286575A (en) * | 1998-04-01 | 1999-10-19 | Nippon Zeon Co Ltd | Diene-based polymer composition |
| DE19825796A1 (en) * | 1998-06-10 | 1999-12-16 | Degussa | New oligomeric organosilane polysulfanes, their use in rubber mixtures and for the production of moldings |
| US6342560B1 (en) * | 1999-08-19 | 2002-01-29 | Ppg Industries Ohio, Inc. | Chemically modified fillers and polymeric compositions containing same |
| JP3530088B2 (en) * | 1999-10-25 | 2004-05-24 | 住友ゴム工業株式会社 | Rubber composition and method for producing the same |
| US6518335B2 (en) | 2000-01-05 | 2003-02-11 | Crompton Corporation | Sulfur-containing silane coupling agents |
| DE10015309A1 (en) | 2000-03-28 | 2001-10-18 | Degussa | Rubber compounds |
| US6433065B1 (en) * | 2000-10-13 | 2002-08-13 | Bridgestone Corporation | Silica-reinforced rubber compounded with mercaptosilanes and alkyl alkoxysilanes |
| US6635700B2 (en) * | 2000-12-15 | 2003-10-21 | Crompton Corporation | Mineral-filled elastomer compositions |
| JP4759150B2 (en) * | 2001-02-16 | 2011-08-31 | 株式会社ブリヂストン | tire |
| DE10132941A1 (en) * | 2001-07-06 | 2003-01-23 | Degussa | Oligomeric organosilanes, process for their preparation and their use |
| ES2252606T3 (en) * | 2001-08-06 | 2006-05-16 | Degussa Ag | ORGANOSILICIO COMPOUNDS. |
| JP2003183446A (en) * | 2001-10-05 | 2003-07-03 | Kao Corp | Storage elastic modulus improver for rubber |
| DE10223658A1 (en) * | 2002-05-28 | 2003-12-18 | Degussa | Organosilicon compounds, process for their preparation and their use |
| JP2004010689A (en) * | 2002-06-05 | 2004-01-15 | Bridgestone Corp | Tire |
| US20040014840A1 (en) * | 2002-07-11 | 2004-01-22 | Uniroyal Chemical Company | Rubber compositions and methods for improving scorch safety and hysteretic properties of the compositions |
-
2004
- 2004-12-18 DE DE102004061014A patent/DE102004061014A1/en not_active Withdrawn
-
2005
- 2005-12-14 AT AT05112090T patent/ATE516326T1/en active
- 2005-12-14 PT PT05112090T patent/PT1672017E/en unknown
- 2005-12-14 PL PL05112090T patent/PL1672017T3/en unknown
- 2005-12-14 SI SI200531368T patent/SI1672017T1/en unknown
- 2005-12-14 EP EP05112090A patent/EP1672017B1/en active Active
- 2005-12-14 ES ES05112090T patent/ES2368568T3/en active Active
- 2005-12-15 TW TW094144542A patent/TWI389958B/en not_active IP Right Cessation
- 2005-12-15 US US11/300,374 patent/US7384997B2/en active Active
- 2005-12-16 CA CA2530346A patent/CA2530346C/en not_active Expired - Fee Related
- 2005-12-16 MX MXPA05013860A patent/MXPA05013860A/en active IP Right Grant
- 2005-12-16 RU RU2005139404/05A patent/RU2404207C2/en active
- 2005-12-16 MY MYPI20055961A patent/MY139418A/en unknown
- 2005-12-16 KR KR1020050124338A patent/KR101222258B1/en active IP Right Grant
- 2005-12-19 JP JP2005365395A patent/JP4420894B2/en not_active Expired - Fee Related
- 2005-12-19 BR BRPI0506287A patent/BRPI0506287B1/en not_active IP Right Cessation
- 2005-12-19 CN CN2005100229191A patent/CN1789315B/en not_active Expired - Fee Related
Also Published As
| Publication number | Publication date |
|---|---|
| DE102004061014A1 (en) | 2006-06-29 |
| BRPI0506287A (en) | 2006-10-03 |
| PT1672017E (en) | 2011-09-22 |
| JP4420894B2 (en) | 2010-02-24 |
| MXPA05013860A (en) | 2006-06-19 |
| ATE516326T1 (en) | 2011-07-15 |
| MY139418A (en) | 2009-09-30 |
| CN1789315A (en) | 2006-06-21 |
| ES2368568T3 (en) | 2011-11-18 |
| EP1672017A2 (en) | 2006-06-21 |
| TWI389958B (en) | 2013-03-21 |
| BRPI0506287B1 (en) | 2016-07-26 |
| JP2006169538A (en) | 2006-06-29 |
| KR101222258B1 (en) | 2013-01-15 |
| SI1672017T1 (en) | 2011-10-28 |
| EP1672017A3 (en) | 2007-05-09 |
| RU2404207C2 (en) | 2010-11-20 |
| KR20060069787A (en) | 2006-06-22 |
| RU2005139404A (en) | 2007-06-27 |
| CN1789315B (en) | 2011-07-20 |
| US20060160935A1 (en) | 2006-07-20 |
| CA2530346A1 (en) | 2006-06-18 |
| TW200634078A (en) | 2006-10-01 |
| US7384997B2 (en) | 2008-06-10 |
| PL1672017T3 (en) | 2011-11-30 |
| CA2530346C (en) | 2010-05-18 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| EP1672017B1 (en) | Rubber compositions | |
| EP2026982B1 (en) | Rubber mixtures | |
| EP1829922B1 (en) | Latex compositions | |
| EP1285926B1 (en) | Organosilicon compounds | |
| EP1533336B1 (en) | Rubber mixtures | |
| EP1976926B1 (en) | Rubber mixtures | |
| DE10327624B3 (en) | Organosilicon compounds, process for their preparation, and their use | |
| EP2041146B1 (en) | Mixtures of silicon-containing coupling reagents | |
| EP1683801B1 (en) | Mercaptosilanes | |
| EP1754749B1 (en) | Rubber mixtures | |
| KR101125514B1 (en) | Carbon black | |
| EP0992505A2 (en) | Sulfanyl silanes | |
| DE19915281A1 (en) | Rubber compounds | |
| EP1367059B1 (en) | Organosilicon compounds, their preparation and use | |
| EP1394166B1 (en) | Organosilicon Compounds | |
| EP3094686B1 (en) | Oligomeric organosilanes, the production thereof and the use thereof in rubber mixtures | |
| CN112566971B (en) | Rubber mixture | |
| EP3830095B1 (en) | Thioether silanes, method for the production thereof, and use thereof | |
| DE10163941C1 (en) | Organosilicon compounds with one or two long-chain alkoxy groups attached to silicon, used as coupling agents for fillers in rubber mixtures, e.g. for tires or tubing are new | |
| DE10208590A1 (en) | Rubber mixture used especially for production of tires and tire treads contains a silicate filler and a combination of mercaptoalkyl-silane and organoxy group-containing siloxane |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| PUAI | Public reference made under article 153(3) epc to a published international application that has entered the european phase |
Free format text: ORIGINAL CODE: 0009012 |
|
| 17P | Request for examination filed |
Effective date: 20051214 |
|
| AK | Designated contracting states |
Kind code of ref document: A2 Designated state(s): AT BE BG CH CY CZ DE DK EE ES FI FR GB GR HU IE IS IT LI LT LU LV MC NL PL PT RO SE SI SK TR |
|
| AX | Request for extension of the european patent |
Extension state: AL BA HR MK YU |
|
| RAP1 | Party data changed (applicant data changed or rights of an application transferred) |
Owner name: DEGUSSA GMBH |
|
| PUAL | Search report despatched |
Free format text: ORIGINAL CODE: 0009013 |
|
| AK | Designated contracting states |
Kind code of ref document: A3 Designated state(s): AT BE BG CH CY CZ DE DK EE ES FI FR GB GR HU IE IS IT LI LT LU LV MC NL PL PT RO SE SI SK TR |
|
| AX | Request for extension of the european patent |
Extension state: AL BA HR MK YU |
|
| 17Q | First examination report despatched |
Effective date: 20070712 |
|
| RAP1 | Party data changed (applicant data changed or rights of an application transferred) |
Owner name: EVONIK DEGUSSA GMBH |
|
| AKX | Designation fees paid |
Designated state(s): AT BE BG CH CY CZ DE DK EE ES FI FR GB GR HU IE IS IT LI LT LU LV MC NL PL PT RO SE SI SK TR |
|
| RAP1 | Party data changed (applicant data changed or rights of an application transferred) |
Owner name: EVONIK DEGUSSA GMBH |
|
| GRAP | Despatch of communication of intention to grant a patent |
Free format text: ORIGINAL CODE: EPIDOSNIGR1 |
|
| GRAS | Grant fee paid |
Free format text: ORIGINAL CODE: EPIDOSNIGR3 |
|
| GRAA | (expected) grant |
Free format text: ORIGINAL CODE: 0009210 |
|
| AK | Designated contracting states |
Kind code of ref document: B1 Designated state(s): AT BE BG CH CY CZ DE DK EE ES FI FR GB GR HU IE IS IT LI LT LU LV MC NL PL PT RO SE SI SK TR |
|
| REG | Reference to a national code |
Ref country code: GB Ref legal event code: FG4D Free format text: NOT ENGLISH |
|
| REG | Reference to a national code |
Ref country code: CH Ref legal event code: EP |
|
| REG | Reference to a national code |
Ref country code: IE Ref legal event code: FG4D Free format text: LANGUAGE OF EP DOCUMENT: GERMAN |
|
| REG | Reference to a national code |
Ref country code: DE Ref legal event code: R096 Ref document number: 502005011611 Country of ref document: DE Effective date: 20110908 |
|
| REG | Reference to a national code |
Ref country code: RO Ref legal event code: EPE |
|
| REG | Reference to a national code |
Ref country code: PT Ref legal event code: SC4A Free format text: AVAILABILITY OF NATIONAL TRANSLATION Effective date: 20110915 |
|
| REG | Reference to a national code |
Ref country code: NL Ref legal event code: T3 |
|
| REG | Reference to a national code |
Ref country code: SK Ref legal event code: T3 Ref document number: E 10043 Country of ref document: SK |
|
| REG | Reference to a national code |
Ref country code: ES Ref legal event code: FG2A Ref document number: 2368568 Country of ref document: ES Kind code of ref document: T3 Effective date: 20111118 |
|
| REG | Reference to a national code |
Ref country code: PL Ref legal event code: T3 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: LT Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20110713 Ref country code: IS Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20111113 Ref country code: SE Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20110713 |
|
| REG | Reference to a national code |
Ref country code: IE Ref legal event code: FD4D |
|
| REG | Reference to a national code |
Ref country code: HU Ref legal event code: AG4A Ref document number: E012268 Country of ref document: HU |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: LV Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20110713 Ref country code: GR Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20111014 Ref country code: CY Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20110713 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: IE Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20110713 |
|
| PLBE | No opposition filed within time limit |
Free format text: ORIGINAL CODE: 0009261 |
|
| STAA | Information on the status of an ep patent application or granted ep patent |
Free format text: STATUS: NO OPPOSITION FILED WITHIN TIME LIMIT |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: EE Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20110713 |
|
| 26N | No opposition filed |
Effective date: 20120416 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: DK Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20110713 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: MC Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20111231 |
|
| REG | Reference to a national code |
Ref country code: CH Ref legal event code: PL |
|
| REG | Reference to a national code |
Ref country code: DE Ref legal event code: R097 Ref document number: 502005011611 Country of ref document: DE Effective date: 20120416 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: CH Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20111231 Ref country code: LI Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20111231 |
|
| REG | Reference to a national code |
Ref country code: AT Ref legal event code: MM01 Ref document number: 516326 Country of ref document: AT Kind code of ref document: T Effective date: 20111214 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: AT Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20111214 Ref country code: BG Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20111013 |
|
| REG | Reference to a national code |
Ref country code: FR Ref legal event code: PLFP Year of fee payment: 11 |
|
| REG | Reference to a national code |
Ref country code: FR Ref legal event code: PLFP Year of fee payment: 12 |
|
| REG | Reference to a national code |
Ref country code: FR Ref legal event code: PLFP Year of fee payment: 13 |
|
| REG | Reference to a national code |
Ref country code: DE Ref legal event code: R081 Ref document number: 502005011611 Country of ref document: DE Owner name: EVONIK OPERATIONS GMBH, DE Free format text: FORMER OWNER: EVONIK DEGUSSA GMBH, 45128 ESSEN, DE |
|
| REG | Reference to a national code |
Ref country code: BE Ref legal event code: HC Owner name: EVONIK OPERATIONS GMBH; DE Free format text: DETAILS ASSIGNMENT: CHANGE OF OWNER(S), CHANGEMENT DE NOM DU PROPRIETAIRE; FORMER OWNER NAME: EVONIK DEGUSSA GMBH Effective date: 20191122 |
|
| REG | Reference to a national code |
Ref country code: SK Ref legal event code: TC4A Ref document number: E 10043 Country of ref document: SK Owner name: EVONIK OPERATIONS GMBH, ESSEN, DE Effective date: 20200525 |
|
| REG | Reference to a national code |
Ref country code: LU Ref legal event code: HC Owner name: EVONIK OPERATIONS GMBH; DE Free format text: FORMER OWNER: EVONIK DEGUSSA GMBH Effective date: 20200505 |
|
| REG | Reference to a national code |
Ref country code: ES Ref legal event code: PC2A Owner name: EVONIK OPERATIONS GMBH Effective date: 20200622 |
|
| REG | Reference to a national code |
Ref country code: HU Ref legal event code: HC9C Owner name: EVONIK OPERATIONS GMBH, DE Free format text: FORMER OWNER(S): EVONIK DEGUSSA GMBH, DE |
|
| REG | Reference to a national code |
Ref country code: FI Ref legal event code: PCE Owner name: EVONIK OPERATIONS GMBH |
|
| REG | Reference to a national code |
Ref country code: NL Ref legal event code: HC Owner name: EVONIK OPERATIONS GMBH; DE Free format text: DETAILS ASSIGNMENT: CHANGE OF OWNER(S), CHANGE OF OWNER(S) NAME Effective date: 20200928 |
|
| REG | Reference to a national code |
Ref country code: SI Ref legal event code: SP73 Owner name: EVONIK OPERATIONS GMBH; DE Effective date: 20201013 |
|
| PGFP | Annual fee paid to national office [announced via postgrant information from national office to epo] |
Ref country code: FI Payment date: 20201222 Year of fee payment: 16 Ref country code: DE Payment date: 20201211 Year of fee payment: 16 Ref country code: FR Payment date: 20201223 Year of fee payment: 16 Ref country code: GB Payment date: 20201223 Year of fee payment: 16 |
|
| PGFP | Annual fee paid to national office [announced via postgrant information from national office to epo] |
Ref country code: BE Payment date: 20201221 Year of fee payment: 16 |
|
| PGFP | Annual fee paid to national office [announced via postgrant information from national office to epo] |
Ref country code: NL Payment date: 20201221 Year of fee payment: 16 Ref country code: LU Payment date: 20201223 Year of fee payment: 16 |
|
| PGFP | Annual fee paid to national office [announced via postgrant information from national office to epo] |
Ref country code: IT Payment date: 20201224 Year of fee payment: 16 |
|
| PGFP | Annual fee paid to national office [announced via postgrant information from national office to epo] |
Ref country code: HU Payment date: 20210117 Year of fee payment: 16 Ref country code: TR Payment date: 20201211 Year of fee payment: 16 Ref country code: ES Payment date: 20210219 Year of fee payment: 16 |
|
| PGFP | Annual fee paid to national office [announced via postgrant information from national office to epo] |
Ref country code: PT Payment date: 20211202 Year of fee payment: 17 Ref country code: SK Payment date: 20211221 Year of fee payment: 17 Ref country code: RO Payment date: 20211202 Year of fee payment: 17 |
|
| PGFP | Annual fee paid to national office [announced via postgrant information from national office to epo] |
Ref country code: SI Payment date: 20211207 Year of fee payment: 17 |
|
| PGFP | Annual fee paid to national office [announced via postgrant information from national office to epo] |
Ref country code: PL Payment date: 20211203 Year of fee payment: 17 Ref country code: CZ Payment date: 20211214 Year of fee payment: 17 |
|
| REG | Reference to a national code |
Ref country code: DE Ref legal event code: R119 Ref document number: 502005011611 Country of ref document: DE |
|
| REG | Reference to a national code |
Ref country code: FI Ref legal event code: MAE |
|
| REG | Reference to a national code |
Ref country code: NL Ref legal event code: MM Effective date: 20220101 |
|
| GBPC | Gb: european patent ceased through non-payment of renewal fee |
Effective date: 20211214 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: FI Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20211214 |
|
| REG | Reference to a national code |
Ref country code: BE Ref legal event code: MM Effective date: 20211231 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: NL Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20220101 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: LU Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20211214 Ref country code: GB Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20211214 Ref country code: DE Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20220701 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: HU Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20211215 Ref country code: FR Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20211231 Ref country code: BE Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20211231 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: IT Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20211214 |
|
| REG | Reference to a national code |
Ref country code: ES Ref legal event code: FD2A Effective date: 20230217 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: ES Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20211215 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: RO Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20221214 Ref country code: PT Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20230614 Ref country code: CZ Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20221214 |
|
| REG | Reference to a national code |
Ref country code: SK Ref legal event code: MM4A Ref document number: E 10043 Country of ref document: SK Effective date: 20221214 |
|
| REG | Reference to a national code |
Ref country code: SI Ref legal event code: KO00 Effective date: 20201013 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: SK Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20221214 Ref country code: SI Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20221215 |