EP0953867B1 - Verfahren zur spektralen Sensibilisierung von tafelförmigen Silberhalogenidkörnern - Google Patents
Verfahren zur spektralen Sensibilisierung von tafelförmigen Silberhalogenidkörnern Download PDFInfo
- Publication number
- EP0953867B1 EP0953867B1 EP98201401A EP98201401A EP0953867B1 EP 0953867 B1 EP0953867 B1 EP 0953867B1 EP 98201401 A EP98201401 A EP 98201401A EP 98201401 A EP98201401 A EP 98201401A EP 0953867 B1 EP0953867 B1 EP 0953867B1
- Authority
- EP
- European Patent Office
- Prior art keywords
- dye
- aggregating
- band
- silver halide
- silver
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Expired - Lifetime
Links
Classifications
-
- G—PHYSICS
- G03—PHOTOGRAPHY; CINEMATOGRAPHY; ANALOGOUS TECHNIQUES USING WAVES OTHER THAN OPTICAL WAVES; ELECTROGRAPHY; HOLOGRAPHY
- G03C—PHOTOSENSITIVE MATERIALS FOR PHOTOGRAPHIC PURPOSES; PHOTOGRAPHIC PROCESSES, e.g. CINE, X-RAY, COLOUR, STEREO-PHOTOGRAPHIC PROCESSES; AUXILIARY PROCESSES IN PHOTOGRAPHY
- G03C1/00—Photosensitive materials
- G03C1/005—Silver halide emulsions; Preparation thereof; Physical treatment thereof; Incorporation of additives therein
- G03C1/0051—Tabular grain emulsions
-
- G—PHYSICS
- G03—PHOTOGRAPHY; CINEMATOGRAPHY; ANALOGOUS TECHNIQUES USING WAVES OTHER THAN OPTICAL WAVES; ELECTROGRAPHY; HOLOGRAPHY
- G03C—PHOTOSENSITIVE MATERIALS FOR PHOTOGRAPHIC PURPOSES; PHOTOGRAPHIC PROCESSES, e.g. CINE, X-RAY, COLOUR, STEREO-PHOTOGRAPHIC PROCESSES; AUXILIARY PROCESSES IN PHOTOGRAPHY
- G03C1/00—Photosensitive materials
- G03C1/005—Silver halide emulsions; Preparation thereof; Physical treatment thereof; Incorporation of additives therein
- G03C1/06—Silver halide emulsions; Preparation thereof; Physical treatment thereof; Incorporation of additives therein with non-macromolecular additives
- G03C1/08—Sensitivity-increasing substances
- G03C1/10—Organic substances
- G03C1/12—Methine and polymethine dyes
-
- G—PHYSICS
- G03—PHOTOGRAPHY; CINEMATOGRAPHY; ANALOGOUS TECHNIQUES USING WAVES OTHER THAN OPTICAL WAVES; ELECTROGRAPHY; HOLOGRAPHY
- G03C—PHOTOSENSITIVE MATERIALS FOR PHOTOGRAPHIC PURPOSES; PHOTOGRAPHIC PROCESSES, e.g. CINE, X-RAY, COLOUR, STEREO-PHOTOGRAPHIC PROCESSES; AUXILIARY PROCESSES IN PHOTOGRAPHY
- G03C1/00—Photosensitive materials
- G03C1/005—Silver halide emulsions; Preparation thereof; Physical treatment thereof; Incorporation of additives therein
- G03C1/06—Silver halide emulsions; Preparation thereof; Physical treatment thereof; Incorporation of additives therein with non-macromolecular additives
- G03C1/08—Sensitivity-increasing substances
- G03C1/28—Sensitivity-increasing substances together with supersensitising substances
- G03C1/29—Sensitivity-increasing substances together with supersensitising substances the supersensitising mixture being solely composed of dyes ; Combination of dyes, even if the supersensitising effect is not explicitly disclosed
-
- G—PHYSICS
- G03—PHOTOGRAPHY; CINEMATOGRAPHY; ANALOGOUS TECHNIQUES USING WAVES OTHER THAN OPTICAL WAVES; ELECTROGRAPHY; HOLOGRAPHY
- G03C—PHOTOSENSITIVE MATERIALS FOR PHOTOGRAPHIC PURPOSES; PHOTOGRAPHIC PROCESSES, e.g. CINE, X-RAY, COLOUR, STEREO-PHOTOGRAPHIC PROCESSES; AUXILIARY PROCESSES IN PHOTOGRAPHY
- G03C5/00—Photographic processes or agents therefor; Regeneration of such processing agents
- G03C5/16—X-ray, infrared, or ultraviolet ray processes
- G03C5/17—X-ray, infrared, or ultraviolet ray processes using screens to intensify X-ray images
-
- G—PHYSICS
- G03—PHOTOGRAPHY; CINEMATOGRAPHY; ANALOGOUS TECHNIQUES USING WAVES OTHER THAN OPTICAL WAVES; ELECTROGRAPHY; HOLOGRAPHY
- G03C—PHOTOSENSITIVE MATERIALS FOR PHOTOGRAPHIC PURPOSES; PHOTOGRAPHIC PROCESSES, e.g. CINE, X-RAY, COLOUR, STEREO-PHOTOGRAPHIC PROCESSES; AUXILIARY PROCESSES IN PHOTOGRAPHY
- G03C1/00—Photosensitive materials
- G03C1/005—Silver halide emulsions; Preparation thereof; Physical treatment thereof; Incorporation of additives therein
- G03C1/06—Silver halide emulsions; Preparation thereof; Physical treatment thereof; Incorporation of additives therein with non-macromolecular additives
- G03C1/08—Sensitivity-increasing substances
- G03C1/09—Noble metals or mercury; Salts or compounds thereof; Sulfur, selenium or tellurium, or compounds thereof, e.g. for chemical sensitising
-
- G—PHYSICS
- G03—PHOTOGRAPHY; CINEMATOGRAPHY; ANALOGOUS TECHNIQUES USING WAVES OTHER THAN OPTICAL WAVES; ELECTROGRAPHY; HOLOGRAPHY
- G03C—PHOTOSENSITIVE MATERIALS FOR PHOTOGRAPHIC PURPOSES; PHOTOGRAPHIC PROCESSES, e.g. CINE, X-RAY, COLOUR, STEREO-PHOTOGRAPHIC PROCESSES; AUXILIARY PROCESSES IN PHOTOGRAPHY
- G03C1/00—Photosensitive materials
- G03C1/005—Silver halide emulsions; Preparation thereof; Physical treatment thereof; Incorporation of additives therein
- G03C1/06—Silver halide emulsions; Preparation thereof; Physical treatment thereof; Incorporation of additives therein with non-macromolecular additives
- G03C1/08—Sensitivity-increasing substances
- G03C1/10—Organic substances
- G03C1/12—Methine and polymethine dyes
- G03C1/14—Methine and polymethine dyes with an odd number of CH groups
- G03C1/18—Methine and polymethine dyes with an odd number of CH groups with three CH groups
-
- G—PHYSICS
- G03—PHOTOGRAPHY; CINEMATOGRAPHY; ANALOGOUS TECHNIQUES USING WAVES OTHER THAN OPTICAL WAVES; ELECTROGRAPHY; HOLOGRAPHY
- G03C—PHOTOSENSITIVE MATERIALS FOR PHOTOGRAPHIC PURPOSES; PHOTOGRAPHIC PROCESSES, e.g. CINE, X-RAY, COLOUR, STEREO-PHOTOGRAPHIC PROCESSES; AUXILIARY PROCESSES IN PHOTOGRAPHY
- G03C1/00—Photosensitive materials
- G03C1/005—Silver halide emulsions; Preparation thereof; Physical treatment thereof; Incorporation of additives therein
- G03C1/06—Silver halide emulsions; Preparation thereof; Physical treatment thereof; Incorporation of additives therein with non-macromolecular additives
- G03C1/08—Sensitivity-increasing substances
- G03C1/10—Organic substances
- G03C1/12—Methine and polymethine dyes
- G03C1/22—Methine and polymethine dyes with an even number of CH groups
-
- G—PHYSICS
- G03—PHOTOGRAPHY; CINEMATOGRAPHY; ANALOGOUS TECHNIQUES USING WAVES OTHER THAN OPTICAL WAVES; ELECTROGRAPHY; HOLOGRAPHY
- G03C—PHOTOSENSITIVE MATERIALS FOR PHOTOGRAPHIC PURPOSES; PHOTOGRAPHIC PROCESSES, e.g. CINE, X-RAY, COLOUR, STEREO-PHOTOGRAPHIC PROCESSES; AUXILIARY PROCESSES IN PHOTOGRAPHY
- G03C1/00—Photosensitive materials
- G03C1/005—Silver halide emulsions; Preparation thereof; Physical treatment thereof; Incorporation of additives therein
- G03C1/06—Silver halide emulsions; Preparation thereof; Physical treatment thereof; Incorporation of additives therein with non-macromolecular additives
- G03C1/40—Dyestuffs not covered by the groups G03C1/08 - G03C1/38 or G03C1/42
-
- G—PHYSICS
- G03—PHOTOGRAPHY; CINEMATOGRAPHY; ANALOGOUS TECHNIQUES USING WAVES OTHER THAN OPTICAL WAVES; ELECTROGRAPHY; HOLOGRAPHY
- G03C—PHOTOSENSITIVE MATERIALS FOR PHOTOGRAPHIC PURPOSES; PHOTOGRAPHIC PROCESSES, e.g. CINE, X-RAY, COLOUR, STEREO-PHOTOGRAPHIC PROCESSES; AUXILIARY PROCESSES IN PHOTOGRAPHY
- G03C1/00—Photosensitive materials
- G03C1/005—Silver halide emulsions; Preparation thereof; Physical treatment thereof; Incorporation of additives therein
- G03C1/06—Silver halide emulsions; Preparation thereof; Physical treatment thereof; Incorporation of additives therein with non-macromolecular additives
- G03C1/08—Sensitivity-increasing substances
- G03C1/09—Noble metals or mercury; Salts or compounds thereof; Sulfur, selenium or tellurium, or compounds thereof, e.g. for chemical sensitising
- G03C2001/091—Gold
-
- G—PHYSICS
- G03—PHOTOGRAPHY; CINEMATOGRAPHY; ANALOGOUS TECHNIQUES USING WAVES OTHER THAN OPTICAL WAVES; ELECTROGRAPHY; HOLOGRAPHY
- G03C—PHOTOSENSITIVE MATERIALS FOR PHOTOGRAPHIC PURPOSES; PHOTOGRAPHIC PROCESSES, e.g. CINE, X-RAY, COLOUR, STEREO-PHOTOGRAPHIC PROCESSES; AUXILIARY PROCESSES IN PHOTOGRAPHY
- G03C1/00—Photosensitive materials
- G03C1/005—Silver halide emulsions; Preparation thereof; Physical treatment thereof; Incorporation of additives therein
- G03C1/06—Silver halide emulsions; Preparation thereof; Physical treatment thereof; Incorporation of additives therein with non-macromolecular additives
- G03C1/08—Sensitivity-increasing substances
- G03C1/09—Noble metals or mercury; Salts or compounds thereof; Sulfur, selenium or tellurium, or compounds thereof, e.g. for chemical sensitising
- G03C2001/096—Sulphur sensitiser
-
- G—PHYSICS
- G03—PHOTOGRAPHY; CINEMATOGRAPHY; ANALOGOUS TECHNIQUES USING WAVES OTHER THAN OPTICAL WAVES; ELECTROGRAPHY; HOLOGRAPHY
- G03C—PHOTOSENSITIVE MATERIALS FOR PHOTOGRAPHIC PURPOSES; PHOTOGRAPHIC PROCESSES, e.g. CINE, X-RAY, COLOUR, STEREO-PHOTOGRAPHIC PROCESSES; AUXILIARY PROCESSES IN PHOTOGRAPHY
- G03C1/00—Photosensitive materials
- G03C1/005—Silver halide emulsions; Preparation thereof; Physical treatment thereof; Incorporation of additives therein
- G03C1/06—Silver halide emulsions; Preparation thereof; Physical treatment thereof; Incorporation of additives therein with non-macromolecular additives
- G03C1/08—Sensitivity-increasing substances
- G03C1/09—Noble metals or mercury; Salts or compounds thereof; Sulfur, selenium or tellurium, or compounds thereof, e.g. for chemical sensitising
- G03C2001/097—Selenium
Definitions
- the present invention relates to a method of preparing a photosensitive emulsion having spectrally sensitized ⁇ 111 ⁇ tabular silver halide crystals showing an improvement in sensitivity (speed) to (preferably) visible green light, emitted by intensifying screens having green-light emitting luminescent phosphors, wherein said emulsion is suitable for use in a light-sensitive silver halide photographic film material applicable in a radiographic screen-film combination comprising at least one X-ray intensifying screen in operative association with the said film material.
- tabular grains are mainly concerned with high sensitive silver bromide or silver bromoiodide emulsions.
- Tabular grain emulsions having a high aspect ratio are known to provide several advantages over more conventional spherical grains as e.g. a high covering power, a high (spectral) sensitivity and a lower coating weight, which saves costs in manufacturing. Said lower coating weight is especially preferred if rapid processing applications are required, which is nowadays an ever more returning demand.
- Spectrally sensitizing dyes are well known in the art of photography, especially for green and red sensitization of flat tabular grains, whereas for blue and/or ultraviolet sensitization the number of examples is rather limited. Further it is known to use in radiography combinations of green-emitting phosphor screens with film materials containing green sensitized tabular grain emulsions. After processing of exposed emulsion grains residual amounts of dyes may be present, especially due to the presence of huge amounts of spectral sensitizing dyes as tabular grains have a large specific surface capable of adsorbing said huge amounts. Those huge amounts are further in favour of high speed and image quality (especially sharpness), required in diagnostic imaging where it is further of utmost importance to reduce irradiation of the patient to minimum levels.
- a method for preparing a tabular silver halide emulsion wherein ⁇ 111 ⁇ tabular silver halide grains rich in silver bromide having an aspect ratio of at least 2:1, an average crystal diameter between 0.3 ⁇ m and 3.0 ⁇ m and an average thickness of less than 0.50 ⁇ m account for at least 50 % of the total projective surface area of said grains, and wherein said method comprises the steps of precipitating, spectrally sensitizing and chemically ripening said grains, characterized by spectrally sensitizing the emulsion grains by adding before chemically ripening at least one J-band aggregating and at least one non-J-band aggregating dye, further defined as in claim 1.
- Spectrally sensitizing dyes are adsorbed on the surface of tabular silver halide grains in an amount sufficient to optimally sensitize said tabular silver halide grains.
- Spectrally sensitizing dyes are adsorbed on the surface of tabular silver halide grains in an amount sufficient to optimally sensitize said tabular silver halide grains.
- sensitization maximum of a sensitizing dye in a silver halide emulsion also called M-band sensitization due to molecular absorption, is situated near the absorption maximum of the free dye in an aqueous solution.
- Certain sensitizing dyes have a sharp sensitization band, or even peak, at a slightly longer wavelength (about 20-50 nm) than the wavelength corresponding to the M-band sensitization maximum. This is known in the art as “J-band sensitization” and the dyes are known as “J-band spectrally sensitizing dyes” or "J-band aggregating dyes”. Opposite thereto are the so-called “non-J-band aggregating dyes" which do not show the phenomenon of showing the sharp band or peak as described.
- J-band aggregating spectrally sensitizing dyes consist of the nearly planar dye molecules adsorbed to silver halide surfaces along their long edge with their molecular planes stacked parallel to each other forming a two-dimensio-nal crystal. According to theoretical calculations bathochromic absorption shifts, characteristic for the presence of J-aggregates depend on the angle of slippage between successive molecular planes.
- ACFEM analytical color fluorescence electron microscopy
- ULESEM ultra low energy scanning electron microscopy
- AFM atomic force microscopy
- a method for preparing a tabular silver halide emulsion wherein ⁇ 111 ⁇ tabular silver halide grains having an average aspect ratio of at least 2:1, an average crystal diameter from 0.3 ⁇ m to 3.0 ⁇ m and an average thickness of less than 0.50 ⁇ m account for at least 50 % of the total projective surface area of said grains, said method comprising the steps of precipitating, spectrally sensitizing and chemically ripening, characterized by spectrally sensitizing the said grains by adding before chemically ripening at least one J-band aggregating and at least one non-J-band aggregating dye.
- said J-band aggregating dye has a maximum J-band absorption in the wavelength range from 540 nm to 555 nm and wherein said non-J-band aggregating dye absorbs irradiation having a wavelength of less than 500 nm, more preferably between 380 and 500 nm and still more preferably between 420 and 460 nm.
- said J-band aggregating dye is a cyanine dye and wherein said non-J-band aggregating dye is a merocyanine dye.
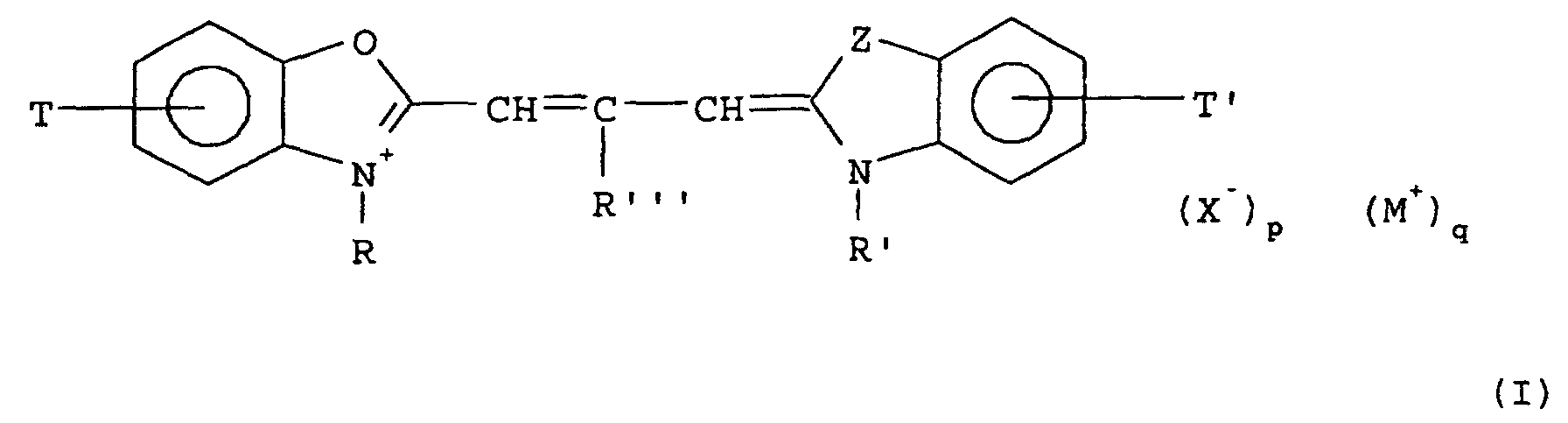
- said cyanine dye corresponds to the formula wherein
- R"' is -C 2 H 5
- Z is an oxygen atom
- at least one of R and R' represents
- R" ' is hydrogen, Z is an nitrogen atom, T represents 5-phenyl, 5-Cl, 5-OCH 3 or 5-CH 3 and wherein T' represents 5,6-(Cl) 2 ; 5-CN-6-Cl; 5-CF 3 -6-Cl; 5-Cl; 5-CN, 5-CF 3 , 5-CHF 2 , 5-SO 2 CH 3 or 5-SO 2 R''''' (R''''' representing a fluoro-subsituted or non-fluoro-substituted alkyl group), 5-COOR'''''' and S-SO 2 -N(R x ) (R y ) or 5-CO-N(R x ) (R y ), wherein R x and R y each independently represent substituted or unsubstited alkyl groups, which may form a ring with the N-atom to which they are attached.
- Z represents oxygen and T and T' each represent Cl or T represents Cl and T' represents Phenyl or vice versa and wherein further R and R' represent all combinations of the formulae - (CH 2 ) n SO 3 - wherein n equals 2, 3 or 4, - (CH 2 ) 2 -CH(CH 3 )-SO 3 - and - CH 2 -CHY-CH 2 -SO 3 - wherein Y represents -OH or -Cl; and wherein R, corresponding to the formulae given above, is combined with R' corresponding to the formulae - (CH 2 ) j H wherein j equals 1, 2, 3 or 4; -CH 2 -Phen-SO 3 - - CH 2 -Phen-COOH - (CH 2 ) k -Phen-COOH wherein k equals 1, 2 or 3.
- said spectral sensitiser is anhydro-5,5'-dichloro-3,3'-bis(n-sulphobutyl)-9-ethyloxacarbocyanine hydroxide or anhydro-5,5'-dichloro-3,3'-bis(n-sulphopropyl)-9-ethyloxa-carbocyanine hydroxide.
- non-J-aggregating dye used in combination with the J-aggreagating dye as described hereinbefore corresponds to the general formula (II), wherein
- said dye corresponds to the general formula (II)
- the alkyl chain containing at least one solubilizing group corresponds to one of the following formulae: (CH 2 ) m SO 3 - or (CH 2 ) m N(R)SO 3 - wherein m equals 1, 2, 3 or 4, and wherein R has the same meaning as R 1 to R 4 ; (CH 2 ) 2 CH(Y)SO 3 - wherein Y represents CH 3 -, -Cl or -OH; (CH 2 ) c OSO 3 - wherein c equals 2 or 3; (CH 2 ) d CO 2 - (H + ) wherein d equals 1, 2 or 3; (CH 2 ) s SO 2 - (CH 2 ) t H wherein s equals 2 or 3 and t equals 1 or 2; - (CH 2 ) f -Phen-W, wherein W represents -COO - or SO 3 - ; Ph
- said dye corresponds to formula (III) or formula (IV) wherein Q, M + , n', R 1 and R 2 have the same meaning as hereinbefore.
- said dye corresponds to the formula (V) wherein R 1 , R 2 , n' and M + have the same meaning as herinbefore.
- said dye corresponds to the formula (VI) or (VII)
- a molar ratio of non-J-band aggregating to J-band aggregating dyes is from 1:20 up to 10:1 more preferably from 1:5 up to 5:1 and still more preferably from 1:5 up to 5:4.
- the concrete dye anhydro-5,5'-dichloro-3,3'-bis(n-sulphobutyl)-9-ethyl-oxacarbocyanine hydroxide or anhydro-5,5'-dichloro-3,3'-bis(n-sulphopropyl)-9-ethyloxacarbocyanine hydroxide or its sulphopropyl derivative is thus added in an amount of from 0.20 up to a maximum amount of about 1 mmole per mole of silver, whereas the dye according to the formula (VII) hereinbefore is added in an amount of more than about 0.15 up to at most about 1.20 mmole per mole of silver. Opposite thereto, if used alone up to 2.8 mmole of J-band aggregating dye per mole of silver are added in order to get well-spectrally sensitized tabular emulsion grains.
- one or more non-J-band aggregating dye(s) is (are) present in a total amount of more than 0.15 mmole per mole of silver and one or more J-band aggregating dye(s) is (are) present in a total amount of less than 1 mmole per mole of silver.
- the tabular silver halide emulsion used in the method of the present invention is an emulsion comprising ⁇ 111 ⁇ tabular silver halide grains having an average aspect ratio of at least 2:1, an average crystal diameter from 0.3 ⁇ m to 3.0 ⁇ m and an average thickness of less than 0.50 ⁇ m account for at least 50 % of the total projective surface area of said grains.
- said average thickness of the said tabular silver halide grains is less than 0.30 ⁇ m, and preferably even less than 0.20 ⁇ m, i.e. not less than about 0.06 ⁇ m.
- said grains have an average aspect ratio of at least 5:1, more preferably between 5:1 and 20:1 and still more preferably between 8:1 and 15:1.
- said average crystal diameter calculated as an average equivalent circular diameter from electron microscopic data obtained from all tabular grains present (which account for at least 50 % of the total projective surface area of said grains as told hereinbefore) is more preferably from 0.3 ⁇ m up to 2.0 ⁇ m and even more preferably from 0.4 ⁇ m up to 1.0 ⁇ m.
- said halide is selected from the group consisting of bromide, chloride and iodide.
- said bromide content is preferably at least 90 mole % and more preferably at least 95 mole % and even at least 98 mole %, whereas the silver iodide content is preferably less than 2 mole %, more preferably less than 1 mole %, still more preferably less than 0.2 mole % and even not more than 0.05 mole % in order to prevent inhibition in the developing step, thereby making rapid processing possible.
- said chloride content is preferably at least 90 mole %, more preferably at least 95 mole % and even at least 98 mole %, whereas the silver iodide content is preferably less than 2 mole %, more preferably less than 1 mole %, still more preferably less than 0.2 mole % and even not more than 0.05 mole %, again in order to prevent inhibition of the developing step, thereby promoting rapid processing.
- silver iodide can be added by double jet addition of (preferably diluted) silver nitrate and potassium iodide solutions, by conversion making use therefore from a (preferably diluted) potassium iodide solution, by addition of iodide ions generated rapidly from an organic iodide releasing agent as has been described e.g.
- ultrafine (homogeneous) silver iodide Lippmann emulsions also called “micrate emulsions”
- ⁇ 111 ⁇ tabular crystals as described emulsions used in the method according to the present invention comprise a binder.
- a survey of binders useful in the method of the present invention has been described in Research Disclosure No. 38957, published September 1, 1996, Chapter II. More detailed descriptions thereof can be found in EP-Application No. 96203203, filed November 15, 1996 wherein the use of different types of gelatin as commonly used hydrophilic binder has been described in the emulsion preparation of tabular grains rich in silver bromide and in EP-A 0 677 773, wherein colloidal silica has been described as an alternative useful binder, just as cationic starch described e.g. in EP-A 0 758 758.
- Tabular silver halide emulsions prepared by the method of the present invention can be chemically sensitized as described e.g. in "Chimie et Physique Photographique” by P. Glafkides, in “Photographic Emulsion Chemistry” by G.F. Duffin, in “Making and Coating Photographic Emulsion” by V.L. Zelikman et al, and in “Die Grundlagen der Photographischen mit Silberhalogeniden” edited by H. Frieser and published by Akademische Verlagsgesellschaft (1968). Chemical sensitisation has e.g. also been described in Research Disclosure N° 38957 (1996), Chapter IV.
- chemical sensitization can be carried out by effecting the ripening in the presence of small amounts of compounds containing sulphur e.g. thiosulphate, thiocyanate, thioureas, sulphites, mercapto compounds, and rhodamines.
- Said compounds containing sulphur can also be, at least partially, replaced by compounds containing selenium and/or tellurium.
- the emulsions may be sensitized also by means of gold-sulphur, gold-sulphur-selenium, gold-selenium ripeners or by means of reductors e.g. tin compounds as described in GB Patent 789,823, amines, hydrazine derivatives, formamidine-sulphinic acids, and silane compounds.
- the J-band and non-J-band forming spectral sensitizers are added before addition of the chemical sensitizing compounds mentioned hereinbefore as has been disclosed already in the earliest patents with respect to tabular grain emulsions filed by E. Kodak in 1981, more particularly in US-A's 4,425,425 and 4,439,520. It is not important whether the non-J-band aggregating dye or the J-band aggregating dye(s) is(are) added as the first dye(s) to the tabular grain emulsions in the method of the present invention. Even a mixture of both of them can be made before addition to the emulsion.
- a light-sensitive silver halide emulsion comprising tabular silver halide grains having an aspect ratio of at least 2:1, an average crystal diameter between 0.3 ⁇ m and 3.0 ⁇ m and an average thickness of less than 0.50 ⁇ m accounting for at least 50 % of the total projective surface area of said grains, wherefore preferred embodiments have already been described hereinbefore, and wherein said grains have at least one J-band aggregating spectrally sensitizing dye and at least one non-J-band aggregating dye the particular embodiments of which have also already been described hereinbefore.
- Light-sensitive silver halide emulsions are thus provided wherein one or more non-J-band aggregating dye(s) is (are) present in an amount of more than 0.15 mmole per mole of silver and wherein one or more J-band aggregating dye(s) is (are) present in an amount of less than 1 mmole per mole of silver. More in detail it should be clear that the amount of J-band aggregating spectrally sensitizing dye is always less than the normally (optimally) added amount when only one or more dyes of the same J-band aggregating type are present.
- heterocyclic nitrogen-containing compounds such as benzothiazolium salts, nitroimidazoles, nitrobenzimidazoles, chlorobenzimidazoles, bromobenzimidazoles, mercaptothiazoles, mercaptobenzothiazoles, mercaptobenzimidazoles, mercaptothiadiazoles, aminotriazoles, benzotriazoles (preferably 5-methyl-benzotriazole), nitrobenzo-triazoles, mercaptotetrazoles, in particular 1-phenyl-5-mercapto-tetrazole, mercaptopyrimidines, mercaptotriazines, benzothiazoline-2-thione, oxazoline-thione, triazaindenes, tetrazaindenes and pentazaindenes, especially those described by Birr in Z.
- benzothiazolium salts such as benzothiazolium salts, nitroimidazoles, nitrobenzimidazoles,
- gelatin as most preferred binder
- silver halide ratio ranging from 0.3 to 1.0 is then obtained, wherein extra gelatin added is not required to have a compostion as specific as in the preparation step of the grains according to the method of the present invention.
- Another binder may also be added instead of or in addition to gelatin.
- Useful vehicles, vehicle extenders, vehicle-like addenda and vehicle related addenda have been described e.g. in Research Disclosure N° 38957 (1996), Chapter II and in EP-A's 0 528 476 and 0 831 362.
- gelatinous emulsions comprising tabular grains and at least one J-band aggregating dye and at least one non-J-band aggregating dye can be used in various types of photographic elements, e.g. black and white silver halide photographic materials, like materials used for X-ray diagnostic purposes.
- a light-sensitive silver halide photographic material more preferably an X-ray photographic material, is further provided, said material comprising a support, at least one hydrophilic colloid layer on at least one side of said support and one or more photosensitive layer(s) comprising a silver halide photographic emulsion according to the present invention as set forth hereinbefore.
- the binder of the photographic material having at least one gelatinous emulsion prepared according to the method of the present invention can be forehardened with appropriate hardening agents such as those of the epoxide type, those of the ethylenimine type, those of the vinylsulfone type e.g. 1,3-vinylsulphonyl-2-propanol, chromium salts e.g. chromium acetate and chromium alum, aldehydes e.g. formaldehyde, glyoxal, and glutaraldehyde, N-methylol compounds e.g.
- dimethylol-urea and methyloldimethylhydantoin dioxan derivatives e.g. 2,3-dihydroxy-dioxan, active vinyl compounds e.g. 1,3,5-triacryloyl-hexahydro-s-triazine, active halogen compounds e.g. 2,4-dichloro-6-hydroxy-s-triazine, and mucohalogenic acids e.g. mucochloric acid and mucophenoxychloric acid.
- These hardeners can be used alone or in combination.
- the binder can also be hardened with fast-reacting hardeners such as carbamoylpyridinium salts as disclosed in US-A 4,063,952 and with the onium compounds as disclosed in EP-A 0 408 143.
- said photographic element or material comprises a support and on one or on each side thereof one or more silver halide emulsion layer(s) coated from a gelatinous emulsion prepared according to the method of this invention. More specifically said photographic material is a single-side or double-side coated X-ray material.
- the single-side coated X-ray material may contain one single emulsion layer, as it is the case for many applications, or it can be built up by two or even more emulsion layers.
- a material with a single or a duplitized emulsion layer coated on one or both sides of the support thus contains at least one gelatinous silver halide emulsion according to the invention.
- the material contains blue, green and red sensitive layers each of which can be single coated, but merely consist of double or even triple layers.
- the photographic material may contain several light-insensitive layers, e.g. a protective layer, one or more backing layers, one or more subbing layers, one or more intermediate layers e.g. filter layers and even an afterlayer containing e.g. the hardening agent(s), the antistatic agent(s), filter dyes for safety-light purposes etc.
- the photographic element may further comprise various kinds of coating physical property modifying addenda as described in Research Disclosure N° 38957 (1996), Chapter IX, wherein coating aids, plasticizers and lubricants, antistats and matting agents have been described.
- Development acceleration can be accomplished by incorporating in the emulsion layer or adjacent layers various compounds, preferably polyalkylene derivatives having a molecular weight of at least 400 such as those described in e.g. US-P's 3,038,805; 4,038,075 and 4,292,400 as well as in EP-A's 0 634 688 and 0 674 215.
- the photographic element may further comprise various other additives such as e.g. compounds improving the dimensional stability of the photographic element, UV-absorbers, spacing agents and plasticizers.
- Suitable additives for improving the dimensional stability of the photographic element are e.g. dispersions of a water-soluble or hardly soluble synthetic polymer e.g.
- Suitable UV-absorbers are e.g. aryl-substituted benzotriazole compounds as described in US-P 3,533,794, 4-thiazolidone compounds as described in US-P's 3,314,794 and 3,352,681, benzophenone compounds as described in JP-A 2784/71, cinnamic ester compounds as described in US-P's 3,705,805 and 3,707,375, butadiene compounds as described in US-P 4,045,229, and benzoxazole compounds as described in US-A 3,700,455 and those described in RD N° 38957 (1996), Chapter VI, wherein also suitable optical brighteners are mentioned.
- Spacing agents can be present of which, in general, the average particle size is comprised between 0.2 and 10 ⁇ m. Spacing agents can be soluble or insoluble in alkali. Alkali-insoluble spacing agents usually remain permanently in the photographic element, whereas alkali-soluble spacing agents usually are removed therefrom in an alkaline processing bath. Suitable spacing agents can be made e.g. of polymethyl methacrylate, of copolymers of acrylic acid and methyl methacrylate, and of hydroxypropylmethyl cellulose hexahydrophthalate. Other suitable spacing agents have been described in US-A 4,614,708.
- the photographic material can contain several non-light sensitive layers, e.g. an antistress topcoat layer, one or more backing layers, and one or more intermediate layers eventually containing filter- or antihalation dyes that absorb scattering light and thus promote the image sharpness.
- Suitable light-absorbing dyes used in these intermediate layers are described in e.g. US-A's 4,092,168 and 4,311,787, in DE-A 2,453,217 and in GB-A 7,907,440. Situated in such an intermediate layer between the emulsion layers and the support there will be only a small negligable loss in sensitivity but in rapid processing conditions decoloration of the filter dye layers may form a problem. Therefor it should be recommended to decrease the thickness of the whole coated layer packet resulting in shorter drying times after washing in the processing cycle.
- a radiographic screen-film combination may be provided comprising at least one X-ray intensifying screen in operative association with a material as described hereinbefore.
- the addition of appropriate filter dyes to the screens may be recommended.
- specific dyes as MAKROLEX ORANGE G or GG, trademarked products of BAYER AG.
- One or more backing layers can be provided at the non-light sensitive side of the support of materials coated with at least one emulsion layer at only one side of the support. These layers which can serve as anti-curl layer can contain e.g. matting agents like silica particles, lubricants, antistatic agents, light absorbing dyes, opacifying agents, e.g. titanium oxide and the usual ingredients like hardeners and wetting agents.
- the support of the photographic material may be opaque or transparent, e.g. a paper support or resin support.
- a paper support preference is given to one coated at one or both sides with an ⁇ -olefin polymer, e.g. a polyethylene layer which optionally contains an antihalation dye or pigment.
- an organic resin support e.g. cellulose nitrate film, cellulose acetate film, poly(vinyl acetal) film, polystyrene film, poly(ethylene terephthalate) or poly(ethylene naphthalate) film, polycarbonate film, polyvinylchloride film or poly- ⁇ -olefin films such as polyethylene or polypropylene film.
- the thickness of such organic resin film is preferably comprised between 0.07 and 0.35 mm.
- These organic resin supports are preferably coated with a subbing layer which can contain water insoluble particles such as silica or titanium dioxide.
- the photographic material containing tabular grains prepared according to the present invention can be image-wise exposed by any convenient radiation source in accordance with its specific application.
- processing conditions and composition of processing solutions are dependent from the specific type of photographic material in which the tabular grains prepared according to the present invention are applied.
- materials for X-ray diagnostic purposes said materials may be adapted to rapid processing conditions.
- an automatically operating processing apparatus is used provided with a system for automatic regeneration of the processing solutions.
- the forehardened material may be processed using one-part package chemistry or three-part package chemistry, depending on the processing application determining the degree of hardening required in said processing cycle. Applications within total processing times of 30 seconds and lower up to 90 seconds, known as common praxis, are possible. From an ecological point of view it is e.g.
- spectral sensitizing dyes are called “J-band spectrally sensitizing dyes” or “J-band aggregating dyes” when the said dyes produce J-aggregates if adsorbed on the surface of the silver halide grains.
- J-aggregates are characterized by a sharp absorption band, called J-band, with a bathochromic shift with respect to the absorption maximum of the free dye in a diluted aqueous solution.
- a spectral sensitizing dye was added in following amounts in mole/mole of silver halide: 0 (blank); 1 x 10 -6 ; 1 x 10 -5 ; 1 x 10 -4 ; 1 x 10 -3 ; said emulsion having a pH of 5.5, a UAg of 70 mV (potential versus silver/silver chloride reference electrode) at 38°C in a concentration of 60 g of silver nitrate per liter of the samples.
- the absorption spectra were taken for all samples, inclusive for the "blank" sample.
- the said dye is called a "J-aggregating” or "J-band aggregating” dye.
- Emulsion preparation :
- An emulsion having silver iodobromide emulsion crystals containing 1.0 mole % of silver iodide was prepared by following preparation steps, making use of the 3 solutions as set forth hereinbefore: a nucleation and a first neutralization step, a second neutralization step and a growth step, followed by a third and a fourth neutralization step, and a second growth step.
- Precipitation proceeded, making use of the double jet technique with continuous control of the pAg or pBr value, said value being defined as the negative logarithm of the silver ion or of the bromide ion concentration respectively, and with application, where required, of continuously varying addition rates of solutions to the reaction vessel.
- the emulsion 150 g of gelatin were added in order to get a weight ratio of gelatin to silver nitrate of 0.4, the emulsion containing an amount of silver bromoiodide equivalent with 190 g of silver nitrate per kg.
- the emulsion thus prepared was divided in 4 parts.
- the emulsion crystals were chemically ripened with sulphur and gold in the presence of green light absorbing J-aggregating dyes as spectral sensitizing dyes and non-J-aggregating dyes at 47°C for a time in order to get an optimized relationship between fog and sensitivity (speed).
- Addition sequence of the dyes was such that the non-J-aggregating dye (NJAD) was added after the green light absorbing J-aggregating dye (JAD).
- a total amount of green light absorbing JAD was 1.26 and 0.63 mmole/mole of silver in the two comparative coatings respectively and 0.63 mmole/mole in the two inventive coatings to which increasing amounts of NJAD were added (0.32 and 0.64 mmole/mole respectively).
- a protective coating composition was prepared containing per liter following ingredients in demineralized water:
- Bis-(vinylsulphonyl)-methane was added as a hardening agent in an amount in order to have a swelling degree of the material of not more than 200 % when immersing the hardened material for 3 minutes in demineralized water of 35°C.
- Exposure,sensitometric and densitometric data samples of these coatings were preserved for 3 days at a temperature of 57°C and 34% RH and were exposed at both sides with visible light by projection lamps (130V; 250W) having an exposure voltage of 90V during 0.1 s at a distance of 1.8 m from the film, through a continuously varying carbon-coated wedge (wedge constant 0.15) with a densitometric filter with a density of 0.30 and a "Corning filter 4010" as a filter with a density 2.64 (measured with a Macbeth TR 924 densitometer), transmitting green light in the range between 500 and 580 nm and with a transmission maximum at 525 nm.
- the exposed samples were processed during the 90 seconds cycle described below.
- the density as a function of the light dose was measured and therefrom were determined the following parameters:
- Stain was visually controlled : "Yes” if stain was still visible; “No” if there was no remarkable or disturbing stain density.
- CURIX 530 tradename of Agfa-Gevaert N.V. was used as an automatic processing machine. Processing sequence and conditions in the said CURIX 530 processing machine were following (expressed in seconds (sec.), temperature (in °C) added thereto: loading 3.4 sec. developing 23.4 sec./ 35°C in developer G138 (trade name) cross-over 3.8 sec. fixing 15.7 sec./ 35°C in fixer G334 (trade name) cross-over 3.8 sec. rinsing 15.7 sec./ 20°C. drying 32.2 sec. (cross-over time included) total time 98.0 sec.
- inventive samples Nos. 3-4 are illustrative for the present invention in that a remarkable gain in speed is attained if, before starting chemical ripening, to the tabular grain emulsion a non-J-band aggregating dye is added in an amount in order to compensate, in part (see 2 and 3 versus 1) or totally (see 4 versus 1), for the addition of a lower amount of absorbing J-band aggregating sensitizing dye (providing the absence of dye stain, but compare the loss in speed if only one half of the normally added amount of JAD is added as in sample No. 2 versus in sample No. 1.
- Example 2 The same emulsion as in Example 1 was prepared and was divided in 9 parts after redispersing.
- the emulsion crystals were chemically ripened with sulphur, selenium and gold in the presence of green light absorbing J-aggregating dyes as spectral sensitizing dyes and non-J-aggregating dyes at 47°C for a time in order to get an optimized relationship between fog and sensitivity (speed).
- Addition sequence of the dyes was such that the non-J-aggregating dye (NJAD) was always added after the green light absorbing J-aggregating dye (JAD).
- a total amount of added green light absorbing J-aggregating dye was always 1.26 mmole/mole of silver: if said total amount was not reached in the chemical ripening, an additional amount was added after chemical ripening in order to compensate therefore.
- inventive samples Nos. 10-13 are illustrative for the present invention in that a remarkable gain in speed is attained if, before starting chemical ripening, to the tabular grain emulsion a non-J-aggregating dye is added after addition of green light absorbing J-aggregating sensitizing dye.
- Example No. 1 The same emulsion as in Example No. 1 was prepared. After redispersion the said emulsion was divided into 10 parts. The emulsion crystals were chemically ripened with sulphur and gold in the presence of a green light absorbing J-aggregating dye as spectral sensitizing dye and a non-J-aggregating dye at 47°C for a time in order to get an optimized relationship between fog and sensitivity (speed). Addition sequence of the dyes was such that the non-J-aggregating dye (NJAD) was always added after the green light absorbing J-aggregating dye (JAD).
- NJAD non-J-aggregating dye
- Amounts of dyes (in mg/mole of silver) added before chemical ripening are given in the Table 5 hereinafter, together with sensitometric data obtained as explained hereinbefore, except for gradations (contrasts) and residual colour or dye stain. Exposures in these experiments were performed on the emulsion side of the single-side coated material only.
- the coating of the photographic materials was so that only one emulsion layer and one protective layer was present on only one side of the support.
- the coating solutions of the emulsion layers were prepared by adding solutions of the compounds indicated in Table 3 to the melted emulsion while stirring.
- the coating solution of the protective layer is given in Table 4.
- the viscosity and surface tension of the coating solutions were optimized according to the requirements of the coating method.
- the emulsion layer(s) and the protective layer were coated simultaneously on one side of a substrated polyester support having a thickness of 175 ⁇ m by means of conventional coating techniques.
- the silver coverage of the emulsions was about 7 g/m 2 , expressed as an equivalent amount of silver nitrate.
- Table 5 shows the sensitometric results in terms of sensitivity (as log E(xposure)): the lower the figure, the more sensitive is the emulsion) and overall contrast GG for the photographic strips coated, exposed and processed as set forth hereinbefore. Amounts of dyes are given in mole/mole of silver as in the Examples hereinbefore. The values given in Table 5 for the overall contrast GG the gradation was measured from the characteristic curve over a density range of 1.75 starting from a density value of 0.25 up to 2.00 above fog. Both gradations have been multiplied by a factor of 100.
- JAD NJAD Speed GG Stain 1 (comp.) 1.230 0 0.126 275 YES 2 (comp.) 0.941 0 0.129 285 YES 3 (comp.) 0.701 0 0.150 270 SLIGHT 4 (comp.) 0.470 0 0.168 247 NO 5 (inv.) 0.941 0.314 0.133 275 SLIGHT 6 (inv.) 0.701 0.314 0.135 309 NO 7 (inv.) 0.470 0.314 0.155 285 NO 8 (inv.) 0.941 0.629 0.142 305 SLIGHT 9 (inv.) 0.701 0.629 0.146 312 NO 10 (inv.) 0.470 0.629 0.156 312 NO
Landscapes
- Physics & Mathematics (AREA)
- Chemical & Material Sciences (AREA)
- General Physics & Mathematics (AREA)
- Chemical Kinetics & Catalysis (AREA)
- Engineering & Computer Science (AREA)
- Materials Engineering (AREA)
- Spectroscopy & Molecular Physics (AREA)
- Silver Salt Photography Or Processing Solution Therefor (AREA)
- Conversion Of X-Rays Into Visible Images (AREA)
Claims (11)
- Ein Verfahren zur Herstellung einer tafelförmige Silberhalogenidkörner enthaltenden Emulsion, wobei tafelförmige {111}-Silberhalogenidkörner mit einem mittleren Aspektverhältnis von zumindest 2:1, einem mittleren Kristalldurchmesser zwischen 0,3 µm und 3,0 µm und einer mittleren Stärke von weniger als 0,50 µm zumindest 50% der gesamten projizierten Oberfläche der Körner ausmachen und wobei das Verfahren als Schritte die Fällung, die spektrale Sensibilisierung und die chemische Reifung der Körner umfasst, dadurch gekennzeichnet, dass die spektrale Sensibilisierung der Körner durch Zugabe vor der chemischen Reifung von zumindest einem zur Bildung von J-Aggregaten befähigten Farbstoff und zumindest einem nicht zur Bildung von J-Aggregaten befähigten Farbstoff erfolgt, wobei der zur Bildung von J-Aggregaten befähigte Farbstoff eine maximale Absorption zur Bildung von J-Aggregaten im Wellenlängenbereich über 500 nm aufweist und der nicht zur Bildung von J-Aggregaten befähigte Farbstoff Strahlung mit einer Wellenlänge von weniger als 500 nm absorbiert, wobei der zur Bildung von J-Aggregaten befähigte Farbstoff ein Cyaninfarbstoff und der nicht zur Bildung von J-Aggregaten befähigte Farbstoff ein Merocyaninfarbstoff ist, wobei der Cyaninfarbstoff folgender Formel entspricht in der :und wobei der nicht zur Bildung von J-Aggregaten befähigte Farbstoff folgender allgemeiner Formel (II) entspricht : in der :Z ein Stickstoffatom oder ein Sauerstoffatom bedeutet und durch R" substituiert ist, wenn Z ein Stickstoffatom ist,R, R' und R" unabhängig voneinander jeweils eine gegebenenfalls substituierte Alkylgruppe bedeuten,R" ' ein Wasserstoffatom, eine gegebenenfalls substituierte Alkylgruppe oder eine gegebenenfalls substituierte Arylgruppe bedeutet,T und T' unabhängig voneinander jeweils die üblichen, den Fachleuten bekannten Substituenten bedeuten können,(X-)p ein negativ geladenes Atom oder eine Gruppe von Atomen zum Ausgleichen der positiven Ladung(en) auf dem Farbstoff bedeutet, und(M+)q ein positiv geladenes Atom oder eine Gruppe von Atomen zum Ausgleichen der negativen Ladung(en) auf dem Farbstoff bedeutet, wobei p und q jeweils eine ganze Zahl bedeuten, durch die eine elektronisch neutrale Verbindung erhalten wird,Y die zur Bildung eines heterocyclischen 5- oder 6-gliedrigen Ringes benötigten Atome bedeutet,Q S, NR3, O oder -(C=O)-N(R4)- bedeutet,M+ ein Kation bedeutet,n' 0, 1 oder 2 bedeutet,b 0 oder 1 bedeutet, undR1, R2, R3 und R4 unabhängig voneinander jeweils eine C1-C5-Alkylkette bedeuten,mit der Maßgabe, dass R2, R3 und/oder R4 eine solubilisierende Gruppe oder eine latente alkalilöslich machende Gruppe enthält (enthalten).
- Verfahren nach Anspruch 1, dadurch gekennzeichnet, dass der Gesamtanteil des (der) nicht zur Bildung von J-Aggregaten befähigten Farbstoffe(s) mehr als 0,15 mMol pro Mol Silber und der Gesamtanteil des (der) zur Bildung von J-Aggregaten befähigten Farbstoffe(s) weniger als 1 mMol pro Mol Silber beträgt.
- Verfahren nach Anspruch 2, dadurch gekennzeichnet, dass das Molverhältnis des nicht zur Bildung von J-Aggregaten befähigten Farbstoffes zum zur Bildung von J-Aggregaten befähigten Farbstoff zwischen 1:20 und 10:1 liegt.
- Verfahren nach Anspruch 2, dadurch gekennzeichnet, dass das Molverhältnis des nicht zur Bildung von J-Aggregaten befähigten Farbstoffes zum zur Bildung von J-Aggregaten befähigten Farbstoff zwischen 1:5 und 5:1 liegt.
- Verfahren nach Anspruch 2, dadurch gekennzeichnet, dass das Molverhältnis des nicht zur Bildung von J-Aggregaten befähigten Farbstoffes zum zur Bildung von J-Aggregaten befähigten Farbstoff zwischen 1:5 und 5:4 liegt.
- Verfahren nach einem der Ansprüche 1 bis 5, dadurch gekennzeichnet, dass die chemische Reifung der tafelförmigen Körner in Gegenwart von Kombinationen von Schwefel-, Selen- und Goldionen liefernden Verbindungen erfolgt.
- Verfahren nach einem der Ansprüche 1 bis 6, dadurch gekennzeichnet, dass als Halogenid in den tafelförmigen Silberhalogenidkörnern Bromid, Chlorid oder Iodid verwendet wird.
- Lichtempfindliche Silberhalogenidemulsion, die nach einem Verfahren nach einem der Ansprüche 1 bis 7 erhältlich ist.
- Lichtempfindliche Silberhalogenidemulsion nach Anspruch 8, dadurch gekennzeichnet, dass der Gesamtanteil des (der) nicht zur Bildung von J-Aggregaten befähigten Farbstoffe(s) mehr als 0,15 mMol pro Mol Silber und der Gesamtanteil des (der) zur Bildung von J-Aggregaten befähigten Farbstoffe(s) weniger als 1 mMol pro Mol Silber beträgt.
- Lichtempfindliches fotografisches Silberhalogenid-Röntgenmaterial, enthaltend einen Träger, zumindest eine hydrophile Kolloidschicht auf zumindest einer Seite des Trägers und eine oder mehrere strahlungsempfindliche Schichten, die eine fotografische Silberhalogenidemulsion nach Anspruch 8 oder 9 enthalten.
- Eine radiografische Film-Folien-Kombination, die zumindest eine Röntgenverstärkerfolie in wirksamer Beziehung zu einem Material nach Anspruch 10 enthält.
Priority Applications (4)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| DE69825419T DE69825419T2 (de) | 1998-04-29 | 1998-04-29 | Verfahren zur spektralen Sensibilisierung von tafelförmigen Silberhalogenidkörnern |
| EP98201401A EP0953867B1 (de) | 1998-04-29 | 1998-04-29 | Verfahren zur spektralen Sensibilisierung von tafelförmigen Silberhalogenidkörnern |
| US09/271,372 US6300051B1 (en) | 1998-04-29 | 1999-03-18 | Method to spectrally sensitize tabular silver halide grains |
| JP11113176A JP2000066323A (ja) | 1998-04-29 | 1999-04-21 | 平板状ハロゲン化銀粒子をスペクトル増感するための方法 |
Applications Claiming Priority (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| EP98201401A EP0953867B1 (de) | 1998-04-29 | 1998-04-29 | Verfahren zur spektralen Sensibilisierung von tafelförmigen Silberhalogenidkörnern |
Publications (2)
| Publication Number | Publication Date |
|---|---|
| EP0953867A1 EP0953867A1 (de) | 1999-11-03 |
| EP0953867B1 true EP0953867B1 (de) | 2004-08-04 |
Family
ID=8233666
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| EP98201401A Expired - Lifetime EP0953867B1 (de) | 1998-04-29 | 1998-04-29 | Verfahren zur spektralen Sensibilisierung von tafelförmigen Silberhalogenidkörnern |
Country Status (3)
| Country | Link |
|---|---|
| EP (1) | EP0953867B1 (de) |
| JP (1) | JP2000066323A (de) |
| DE (1) | DE69825419T2 (de) |
Families Citing this family (4)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US6348293B1 (en) | 1999-11-26 | 2002-02-19 | Agfa-Gevaert | Radiographic film material exhibiting increased covering power and “colder” blue-black image tone |
| EP1103848B1 (de) * | 1999-11-26 | 2007-01-17 | Agfa-Gevaert | Lichtempfindliches photographisches Silberhalogenidfilmmaterial und eine Kombination eines radiographischen Verstärkungsschirms mit diesem Film |
| EP1103849A1 (de) * | 1999-11-26 | 2001-05-30 | Agfa-Gevaert | Radiographisches Element mit verbesserter Deckkraft und blauschwarzem Bildton |
| US6472137B1 (en) | 1999-11-26 | 2002-10-29 | Agfa-Gevaert | Light-sensitive silver halide photographic film material and radiographic intensifying screen-film combination |
Family Cites Families (5)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JPH02167539A (ja) * | 1988-09-14 | 1990-06-27 | Fuji Photo Film Co Ltd | ハロゲン化銀写真感光材料 |
| US5141846A (en) * | 1990-10-18 | 1992-08-25 | Polaroid Corporation | Method for preparing photographic emulsion |
| JP2881504B2 (ja) * | 1991-01-21 | 1999-04-12 | コニカ株式会社 | ハロゲン化銀写真感光材料 |
| EP0678772A1 (de) * | 1994-04-06 | 1995-10-25 | Agfa-Gevaert N.V. | Lichtempfindliches Silberchlorobromojodid- oder Silbuchlorojodid-Tafelkörner enthaltendes Material |
| EP0712034B1 (de) * | 1994-11-11 | 2002-04-10 | Agfa-Gevaert | Verfahren zur spektralen Sensibilisierung von tafelförmigen Silberhalogenidkörnern |
-
1998
- 1998-04-29 DE DE69825419T patent/DE69825419T2/de not_active Expired - Fee Related
- 1998-04-29 EP EP98201401A patent/EP0953867B1/de not_active Expired - Lifetime
-
1999
- 1999-04-21 JP JP11113176A patent/JP2000066323A/ja active Pending
Also Published As
| Publication number | Publication date |
|---|---|
| JP2000066323A (ja) | 2000-03-03 |
| DE69825419T2 (de) | 2005-06-30 |
| DE69825419D1 (de) | 2004-09-09 |
| EP0953867A1 (de) | 1999-11-03 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| US5595864A (en) | Method for making tabular silver halide grains | |
| EP0933670B1 (de) | Lichtempfindliche Emulsion mit bromidreichen Tafelkörnern, die mit Thiocyanatkomplexen des Rhodiums dotiert sind | |
| EP0712034B1 (de) | Verfahren zur spektralen Sensibilisierung von tafelförmigen Silberhalogenidkörnern | |
| EP0953867B1 (de) | Verfahren zur spektralen Sensibilisierung von tafelförmigen Silberhalogenidkörnern | |
| US6200743B1 (en) | Radiation-sensitive emulsion, light-sensitive silver halide photographic film material and radiographic intensifying screen-film combination | |
| US6087085A (en) | Preparation method of morphologically homogeneous (111) tabular crystals rich in silver bromide | |
| EP0911687B1 (de) | Verfahren zur Herstellung von morphologisch homogenen silberbromidreichen tafelförmigen (111)-Kristallen | |
| US6300051B1 (en) | Method to spectrally sensitize tabular silver halide grains | |
| EP0678772A1 (de) | Lichtempfindliches Silberchlorobromojodid- oder Silbuchlorojodid-Tafelkörner enthaltendes Material | |
| EP0569075B1 (de) | Verfahren zur Herstellung von tafelförmigen Silberhalogenid-Körnern | |
| US5733715A (en) | Silver halide photographic materials comprising silver bromide or bromoiodide emulsions having triangular tabular crystals | |
| EP0843208A1 (de) | Verfahren zur Herstellung silberbromidreicher Tafelkörner in Gegenwart spezifischer Gelatinen | |
| EP0651284A1 (de) | Fällung von Silberhalogenidkristallen, die lodid enthalten | |
| US7129031B2 (en) | Radiographic silver halide photographic material having a good developing speed, an excellent image tone and low residual color after processing | |
| EP0752617B1 (de) | Erzeugungsverfahren für photographisches Silberhalogenidmaterial geeignet für schnelle Verarbeitungsanwendungen | |
| EP1045283B1 (de) | Strahlungsempfindliche Emulsion, lichtempfindlicher photographischer Silberhalogenidfilm und Kombination eines radiographischen Verstärkungsschirms mit einem Film | |
| US20020037485A1 (en) | Method of preparing ultrathin light-sensitive tabular grain emulsions rich in silver bromide | |
| EP0770909A1 (de) | Photographisches Silberhalogenidmehrschichtmaterial und Verfahren zu dessen Herstellung | |
| US6080536A (en) | Method of preparing (100) tabular grains rich in silver bromide | |
| US6436621B1 (en) | Multilayer silver halide photographic material and process for preparing the same | |
| US6136524A (en) | Light-sensitive emulsion having (100) tabular grains rich in silver chloride and method for preparing said grains | |
| US6280915B1 (en) | Spectrally sensitized tabular grain photographic materials | |
| EP0890875B1 (de) | Mehrschichtiges photographisches Silberhalogenidmaterial und bilderzeugendes Verfahren für Anwendungen in der zerstörungsfreien Prüfung mittels industrieller Radiographie | |
| US6312882B1 (en) | Silver halide emulsions comprising tabular crystals, emulsions and the processing thereof | |
| US6030757A (en) | Multilayer silver halide photographic material and image-forming method in industrial radiographic non-destructive testing applications |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| PUAI | Public reference made under article 153(3) epc to a published international application that has entered the european phase |
Free format text: ORIGINAL CODE: 0009012 |
|
| AK | Designated contracting states |
Kind code of ref document: A1 Designated state(s): BE DE FR GB |
|
| AX | Request for extension of the european patent |
Free format text: AL;LT;LV;MK;RO;SI |
|
| 17P | Request for examination filed |
Effective date: 20000503 |
|
| AKX | Designation fees paid |
Free format text: BE DE FR GB |
|
| RAP1 | Party data changed (applicant data changed or rights of an application transferred) |
Owner name: AGFA-GEVAERT |
|
| 17Q | First examination report despatched |
Effective date: 20030617 |
|
| GRAP | Despatch of communication of intention to grant a patent |
Free format text: ORIGINAL CODE: EPIDOSNIGR1 |
|
| GRAS | Grant fee paid |
Free format text: ORIGINAL CODE: EPIDOSNIGR3 |
|
| GRAA | (expected) grant |
Free format text: ORIGINAL CODE: 0009210 |
|
| AK | Designated contracting states |
Kind code of ref document: B1 Designated state(s): BE DE FR GB |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: BE Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20040804 |
|
| REG | Reference to a national code |
Ref country code: GB Ref legal event code: FG4D |
|
| REF | Corresponds to: |
Ref document number: 69825419 Country of ref document: DE Date of ref document: 20040909 Kind code of ref document: P |
|
| REG | Reference to a national code |
Ref country code: GB Ref legal event code: 746 Effective date: 20040921 |
|
| ET | Fr: translation filed | ||
| REG | Reference to a national code |
Ref country code: FR Ref legal event code: D6 |
|
| PLBE | No opposition filed within time limit |
Free format text: ORIGINAL CODE: 0009261 |
|
| STAA | Information on the status of an ep patent application or granted ep patent |
Free format text: STATUS: NO OPPOSITION FILED WITHIN TIME LIMIT |
|
| 26N | No opposition filed |
Effective date: 20050506 |
|
| REG | Reference to a national code |
Ref country code: GB Ref legal event code: 732E |
|
| REG | Reference to a national code |
Ref country code: FR Ref legal event code: TP |
|
| PGFP | Annual fee paid to national office [announced via postgrant information from national office to epo] |
Ref country code: DE Payment date: 20080421 Year of fee payment: 11 |
|
| PGFP | Annual fee paid to national office [announced via postgrant information from national office to epo] |
Ref country code: FR Payment date: 20080418 Year of fee payment: 11 |
|
| PGFP | Annual fee paid to national office [announced via postgrant information from national office to epo] |
Ref country code: GB Payment date: 20080421 Year of fee payment: 11 |
|
| GBPC | Gb: european patent ceased through non-payment of renewal fee |
Effective date: 20090429 |
|
| REG | Reference to a national code |
Ref country code: FR Ref legal event code: ST Effective date: 20091231 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: DE Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20091103 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: GB Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20090429 Ref country code: FR Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20091222 |