EP0678772A1 - Lichtempfindliches Silberchlorobromojodid- oder Silbuchlorojodid-Tafelkörner enthaltendes Material - Google Patents
Lichtempfindliches Silberchlorobromojodid- oder Silbuchlorojodid-Tafelkörner enthaltendes Material Download PDFInfo
- Publication number
- EP0678772A1 EP0678772A1 EP95200651A EP95200651A EP0678772A1 EP 0678772 A1 EP0678772 A1 EP 0678772A1 EP 95200651 A EP95200651 A EP 95200651A EP 95200651 A EP95200651 A EP 95200651A EP 0678772 A1 EP0678772 A1 EP 0678772A1
- Authority
- EP
- European Patent Office
- Prior art keywords
- emulsion
- silver
- tabular grains
- emulsions
- tabular
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Withdrawn
Links
- 239000000463 material Substances 0.000 title claims abstract description 64
- 229910052709 silver Inorganic materials 0.000 title claims abstract description 51
- 239000004332 silver Substances 0.000 title claims abstract description 51
- BQCADISMDOOEFD-UHFFFAOYSA-N Silver Chemical compound [Ag] BQCADISMDOOEFD-UHFFFAOYSA-N 0.000 title claims abstract description 27
- QZRGKCOWNLSUDK-UHFFFAOYSA-N Iodochlorine Chemical compound ICl QZRGKCOWNLSUDK-UHFFFAOYSA-N 0.000 title description 9
- 239000000839 emulsion Substances 0.000 claims abstract description 122
- -1 silver halide Chemical class 0.000 claims abstract description 62
- 239000013078 crystal Substances 0.000 claims abstract description 59
- VEXZGXHMUGYJMC-UHFFFAOYSA-M Chloride anion Chemical compound [Cl-] VEXZGXHMUGYJMC-UHFFFAOYSA-M 0.000 claims abstract description 20
- XMBWDFGMSWQBCA-UHFFFAOYSA-N hydrogen iodide Chemical compound I XMBWDFGMSWQBCA-UHFFFAOYSA-N 0.000 claims abstract description 19
- HOLVRJRSWZOAJU-UHFFFAOYSA-N [Ag].ICl Chemical compound [Ag].ICl HOLVRJRSWZOAJU-UHFFFAOYSA-N 0.000 claims abstract description 13
- 230000003595 spectral effect Effects 0.000 claims description 18
- GFFGJBXGBJISGV-UHFFFAOYSA-N Adenine Chemical compound NC1=NC=NC2=C1N=CN2 GFFGJBXGBJISGV-UHFFFAOYSA-N 0.000 claims description 17
- 150000001875 compounds Chemical class 0.000 claims description 17
- SQGYOTSLMSWVJD-UHFFFAOYSA-N silver(1+) nitrate Chemical compound [Ag+].[O-]N(=O)=O SQGYOTSLMSWVJD-UHFFFAOYSA-N 0.000 claims description 17
- XLYOFNOQVPJJNP-UHFFFAOYSA-N water Substances O XLYOFNOQVPJJNP-UHFFFAOYSA-N 0.000 claims description 17
- 229930024421 Adenine Natural products 0.000 claims description 15
- 231100000489 sensitizer Toxicity 0.000 claims description 11
- 125000000217 alkyl group Chemical group 0.000 claims description 9
- 238000010521 absorption reaction Methods 0.000 claims description 8
- 125000004429 atom Chemical group 0.000 claims description 8
- XLYOFNOQVPJJNP-UHFFFAOYSA-M hydroxide Chemical compound [OH-] XLYOFNOQVPJJNP-UHFFFAOYSA-M 0.000 claims description 7
- 229910001961 silver nitrate Inorganic materials 0.000 claims description 7
- 229910052757 nitrogen Inorganic materials 0.000 claims description 5
- IJGRMHOSHXDMSA-UHFFFAOYSA-N Atomic nitrogen Chemical compound N#N IJGRMHOSHXDMSA-UHFFFAOYSA-N 0.000 claims description 4
- 125000004433 nitrogen atom Chemical group N* 0.000 claims description 4
- 125000003118 aryl group Chemical group 0.000 claims description 3
- 229910052739 hydrogen Inorganic materials 0.000 claims description 3
- 239000001257 hydrogen Substances 0.000 claims description 3
- 125000004435 hydrogen atom Chemical class [H]* 0.000 claims description 3
- 125000004430 oxygen atom Chemical group O* 0.000 claims description 3
- 230000007935 neutral effect Effects 0.000 claims description 2
- 125000001424 substituent group Chemical group 0.000 claims description 2
- 238000012545 processing Methods 0.000 abstract description 44
- 230000008929 regeneration Effects 0.000 abstract description 4
- 238000011069 regeneration method Methods 0.000 abstract description 4
- 239000010410 layer Substances 0.000 description 35
- 239000003795 chemical substances by application Substances 0.000 description 26
- 229910021607 Silver chloride Inorganic materials 0.000 description 24
- HKZLPVFGJNLROG-UHFFFAOYSA-M silver monochloride Chemical compound [Cl-].[Ag+] HKZLPVFGJNLROG-UHFFFAOYSA-M 0.000 description 24
- 230000035945 sensitivity Effects 0.000 description 20
- NLKNQRATVPKPDG-UHFFFAOYSA-M potassium iodide Chemical compound [K+].[I-] NLKNQRATVPKPDG-UHFFFAOYSA-M 0.000 description 18
- 229920000159 gelatin Polymers 0.000 description 17
- 235000019322 gelatine Nutrition 0.000 description 17
- 108010010803 Gelatin Proteins 0.000 description 16
- 239000000975 dye Substances 0.000 description 16
- 239000008273 gelatin Substances 0.000 description 16
- 235000011852 gelatine desserts Nutrition 0.000 description 16
- 238000002360 preparation method Methods 0.000 description 16
- 239000000203 mixture Substances 0.000 description 12
- 230000005070 ripening Effects 0.000 description 12
- CPELXLSAUQHCOX-UHFFFAOYSA-M Bromide Chemical compound [Br-] CPELXLSAUQHCOX-UHFFFAOYSA-M 0.000 description 11
- 238000000576 coating method Methods 0.000 description 11
- 239000000126 substance Substances 0.000 description 11
- FAPWRFPIFSIZLT-UHFFFAOYSA-M Sodium chloride Chemical compound [Na+].[Cl-] FAPWRFPIFSIZLT-UHFFFAOYSA-M 0.000 description 10
- GZPBVLUEICLBOA-UHFFFAOYSA-N 4-(dimethylamino)-3,5-dimethylphenol Chemical compound CN(C)C1=C(C)C=C(O)C=C1C GZPBVLUEICLBOA-UHFFFAOYSA-N 0.000 description 9
- 238000007792 addition Methods 0.000 description 9
- 238000006243 chemical reaction Methods 0.000 description 9
- 230000006911 nucleation Effects 0.000 description 9
- 238000010899 nucleation Methods 0.000 description 9
- 238000001556 precipitation Methods 0.000 description 9
- QIGBRXMKCJKVMJ-UHFFFAOYSA-N Hydroquinone Chemical compound OC1=CC=C(O)C=C1 QIGBRXMKCJKVMJ-UHFFFAOYSA-N 0.000 description 8
- 206010070834 Sensitisation Diseases 0.000 description 8
- 239000002253 acid Substances 0.000 description 7
- 239000011248 coating agent Substances 0.000 description 7
- 239000002612 dispersion medium Substances 0.000 description 7
- QTBSBXVTEAMEQO-UHFFFAOYSA-N Acetic acid Chemical compound CC(O)=O QTBSBXVTEAMEQO-UHFFFAOYSA-N 0.000 description 6
- 239000002202 Polyethylene glycol Substances 0.000 description 6
- KWYUFKZDYYNOTN-UHFFFAOYSA-M Potassium hydroxide Chemical compound [OH-].[K+] KWYUFKZDYYNOTN-UHFFFAOYSA-M 0.000 description 6
- 229960000643 adenine Drugs 0.000 description 6
- 238000011161 development Methods 0.000 description 6
- 230000018109 developmental process Effects 0.000 description 6
- 238000000034 method Methods 0.000 description 6
- 229920001223 polyethylene glycol Polymers 0.000 description 6
- 239000004848 polyfunctional curative Substances 0.000 description 6
- IOLCXVTUBQKXJR-UHFFFAOYSA-M potassium bromide Chemical compound [K+].[Br-] IOLCXVTUBQKXJR-UHFFFAOYSA-M 0.000 description 6
- 150000001252 acrylic acid derivatives Chemical class 0.000 description 5
- 238000001035 drying Methods 0.000 description 5
- 239000011780 sodium chloride Substances 0.000 description 5
- 239000004793 Polystyrene Substances 0.000 description 4
- 230000000052 comparative effect Effects 0.000 description 4
- 238000001816 cooling Methods 0.000 description 4
- 238000009826 distribution Methods 0.000 description 4
- 239000002609 medium Substances 0.000 description 4
- 229920002223 polystyrene Polymers 0.000 description 4
- BWHMMNNQKKPAPP-UHFFFAOYSA-L potassium carbonate Chemical compound [K+].[K+].[O-]C([O-])=O BWHMMNNQKKPAPP-UHFFFAOYSA-L 0.000 description 4
- 238000011160 research Methods 0.000 description 4
- 239000011347 resin Substances 0.000 description 4
- 229920005989 resin Polymers 0.000 description 4
- 238000005406 washing Methods 0.000 description 4
- SXRSQZLOMIGNAQ-UHFFFAOYSA-N Glutaraldehyde Chemical compound O=CCCCC=O SXRSQZLOMIGNAQ-UHFFFAOYSA-N 0.000 description 3
- VYPSYNLAJGMNEJ-UHFFFAOYSA-N Silicium dioxide Chemical compound O=[Si]=O VYPSYNLAJGMNEJ-UHFFFAOYSA-N 0.000 description 3
- HEMHJVSKTPXQMS-UHFFFAOYSA-M Sodium hydroxide Chemical compound [OH-].[Na+] HEMHJVSKTPXQMS-UHFFFAOYSA-M 0.000 description 3
- GWEVSGVZZGPLCZ-UHFFFAOYSA-N Titan oxide Chemical compound O=[Ti]=O GWEVSGVZZGPLCZ-UHFFFAOYSA-N 0.000 description 3
- 239000006096 absorbing agent Substances 0.000 description 3
- 150000007513 acids Chemical class 0.000 description 3
- 239000000654 additive Substances 0.000 description 3
- MTHSVFCYNBDYFN-UHFFFAOYSA-N diethylene glycol Chemical compound OCCOCCO MTHSVFCYNBDYFN-UHFFFAOYSA-N 0.000 description 3
- 230000000694 effects Effects 0.000 description 3
- 230000006870 function Effects 0.000 description 3
- 230000002401 inhibitory effect Effects 0.000 description 3
- 239000011241 protective layer Substances 0.000 description 3
- 230000002829 reductive effect Effects 0.000 description 3
- 150000003839 salts Chemical class 0.000 description 3
- ADZWSOLPGZMUMY-UHFFFAOYSA-M silver bromide Chemical compound [Ag]Br ADZWSOLPGZMUMY-UHFFFAOYSA-M 0.000 description 3
- ZUNKMNLKJXRCDM-UHFFFAOYSA-N silver bromoiodide Chemical compound [Ag].IBr ZUNKMNLKJXRCDM-UHFFFAOYSA-N 0.000 description 3
- BDHFUVZGWQCTTF-UHFFFAOYSA-N sulfonic acid Chemical compound OS(=O)=O BDHFUVZGWQCTTF-UHFFFAOYSA-N 0.000 description 3
- 239000004094 surface-active agent Substances 0.000 description 3
- YXIWHUQXZSMYRE-UHFFFAOYSA-N 1,3-benzothiazole-2-thiol Chemical class C1=CC=C2SC(S)=NC2=C1 YXIWHUQXZSMYRE-UHFFFAOYSA-N 0.000 description 2
- GGZHVNZHFYCSEV-UHFFFAOYSA-N 1-Phenyl-5-mercaptotetrazole Chemical compound SC1=NN=NN1C1=CC=CC=C1 GGZHVNZHFYCSEV-UHFFFAOYSA-N 0.000 description 2
- LRUDIIUSNGCQKF-UHFFFAOYSA-N 5-methyl-1H-benzotriazole Chemical compound C1=C(C)C=CC2=NNN=C21 LRUDIIUSNGCQKF-UHFFFAOYSA-N 0.000 description 2
- CIWBSHSKHKDKBQ-JLAZNSOCSA-N Ascorbic acid Chemical compound OC[C@H](O)[C@H]1OC(=O)C(O)=C1O CIWBSHSKHKDKBQ-JLAZNSOCSA-N 0.000 description 2
- 229920003171 Poly (ethylene oxide) Polymers 0.000 description 2
- 239000004698 Polyethylene Substances 0.000 description 2
- 239000004285 Potassium sulphite Substances 0.000 description 2
- 239000004133 Sodium thiosulphate Substances 0.000 description 2
- NINIDFKCEFEMDL-UHFFFAOYSA-N Sulfur Chemical compound [S] NINIDFKCEFEMDL-UHFFFAOYSA-N 0.000 description 2
- QAOWNCQODCNURD-UHFFFAOYSA-N Sulfuric acid Chemical compound OS(O)(=O)=O QAOWNCQODCNURD-UHFFFAOYSA-N 0.000 description 2
- 239000005864 Sulphur Substances 0.000 description 2
- SJOOOZPMQAWAOP-UHFFFAOYSA-N [Ag].BrCl Chemical compound [Ag].BrCl SJOOOZPMQAWAOP-UHFFFAOYSA-N 0.000 description 2
- 230000001133 acceleration Effects 0.000 description 2
- 125000001931 aliphatic group Chemical group 0.000 description 2
- LEQAOMBKQFMDFZ-UHFFFAOYSA-N alpha-ketodiacetal Natural products O=CC=O LEQAOMBKQFMDFZ-UHFFFAOYSA-N 0.000 description 2
- XYXNTHIYBIDHGM-UHFFFAOYSA-N ammonium thiosulfate Chemical compound [NH4+].[NH4+].[O-]S([O-])(=O)=S XYXNTHIYBIDHGM-UHFFFAOYSA-N 0.000 description 2
- 239000002216 antistatic agent Substances 0.000 description 2
- 150000001565 benzotriazoles Chemical class 0.000 description 2
- 239000011230 binding agent Substances 0.000 description 2
- 125000003178 carboxy group Chemical group [H]OC(*)=O 0.000 description 2
- 229920001577 copolymer Polymers 0.000 description 2
- 238000009792 diffusion process Methods 0.000 description 2
- 239000006185 dispersion Substances 0.000 description 2
- 230000002349 favourable effect Effects 0.000 description 2
- 239000004615 ingredient Substances 0.000 description 2
- 238000011068 loading method Methods 0.000 description 2
- 125000000956 methoxy group Chemical group [H]C([H])([H])O* 0.000 description 2
- 239000002245 particle Substances 0.000 description 2
- CMCWWLVWPDLCRM-UHFFFAOYSA-N phenidone Chemical compound N1C(=O)CCN1C1=CC=CC=C1 CMCWWLVWPDLCRM-UHFFFAOYSA-N 0.000 description 2
- 229920000573 polyethylene Polymers 0.000 description 2
- 229920000139 polyethylene terephthalate Polymers 0.000 description 2
- 239000005020 polyethylene terephthalate Substances 0.000 description 2
- 229910000027 potassium carbonate Inorganic materials 0.000 description 2
- BHZRJJOHZFYXTO-UHFFFAOYSA-L potassium sulfite Chemical compound [K+].[K+].[O-]S([O-])=O BHZRJJOHZFYXTO-UHFFFAOYSA-L 0.000 description 2
- 235000019252 potassium sulphite Nutrition 0.000 description 2
- 230000001235 sensitizing effect Effects 0.000 description 2
- GEHJYWRUCIMESM-UHFFFAOYSA-L sodium sulfite Chemical compound [Na+].[Na+].[O-]S([O-])=O GEHJYWRUCIMESM-UHFFFAOYSA-L 0.000 description 2
- AKHNMLFCWUSKQB-UHFFFAOYSA-L sodium thiosulfate Chemical compound [Na+].[Na+].[O-]S([O-])(=O)=S AKHNMLFCWUSKQB-UHFFFAOYSA-L 0.000 description 2
- 235000019345 sodium thiosulphate Nutrition 0.000 description 2
- 239000001117 sulphuric acid Substances 0.000 description 2
- 235000011149 sulphuric acid Nutrition 0.000 description 2
- UEUXEKPTXMALOB-UHFFFAOYSA-J tetrasodium;2-[2-[bis(carboxylatomethyl)amino]ethyl-(carboxylatomethyl)amino]acetate Chemical compound [Na+].[Na+].[Na+].[Na+].[O-]C(=O)CN(CC([O-])=O)CCN(CC([O-])=O)CC([O-])=O UEUXEKPTXMALOB-UHFFFAOYSA-J 0.000 description 2
- 238000012546 transfer Methods 0.000 description 2
- 238000000108 ultra-filtration Methods 0.000 description 2
- 125000000391 vinyl group Chemical group [H]C([*])=C([H])[H] 0.000 description 2
- JNYAEWCLZODPBN-JGWLITMVSA-N (2r,3r,4s)-2-[(1r)-1,2-dihydroxyethyl]oxolane-3,4-diol Chemical class OC[C@@H](O)[C@H]1OC[C@H](O)[C@H]1O JNYAEWCLZODPBN-JGWLITMVSA-N 0.000 description 1
- LUMLZKVIXLWTCI-NSCUHMNNSA-N (e)-2,3-dichloro-4-oxobut-2-enoic acid Chemical compound OC(=O)C(\Cl)=C(/Cl)C=O LUMLZKVIXLWTCI-NSCUHMNNSA-N 0.000 description 1
- BCMCBBGGLRIHSE-UHFFFAOYSA-N 1,3-benzoxazole Chemical class C1=CC=C2OC=NC2=C1 BCMCBBGGLRIHSE-UHFFFAOYSA-N 0.000 description 1
- YHMYGUUIMTVXNW-UHFFFAOYSA-N 1,3-dihydrobenzimidazole-2-thione Chemical class C1=CC=C2NC(S)=NC2=C1 YHMYGUUIMTVXNW-UHFFFAOYSA-N 0.000 description 1
- RYHBNJHYFVUHQT-UHFFFAOYSA-N 1,4-Dioxane Chemical class C1COCCO1 RYHBNJHYFVUHQT-UHFFFAOYSA-N 0.000 description 1
- YLVACWCCJCZITJ-UHFFFAOYSA-N 1,4-dioxane-2,3-diol Chemical compound OC1OCCOC1O YLVACWCCJCZITJ-UHFFFAOYSA-N 0.000 description 1
- IJHIIHORMWQZRQ-UHFFFAOYSA-N 1-(ethenylsulfonylmethylsulfonyl)ethene Chemical compound C=CS(=O)(=O)CS(=O)(=O)C=C IJHIIHORMWQZRQ-UHFFFAOYSA-N 0.000 description 1
- SIQZJFKTROUNPI-UHFFFAOYSA-N 1-(hydroxymethyl)-5,5-dimethylhydantoin Chemical compound CC1(C)N(CO)C(=O)NC1=O SIQZJFKTROUNPI-UHFFFAOYSA-N 0.000 description 1
- FYBFGAFWCBMEDG-UHFFFAOYSA-N 1-[3,5-di(prop-2-enoyl)-1,3,5-triazinan-1-yl]prop-2-en-1-one Chemical compound C=CC(=O)N1CN(C(=O)C=C)CN(C(=O)C=C)C1 FYBFGAFWCBMEDG-UHFFFAOYSA-N 0.000 description 1
- YGDWUQFZMXWDKE-UHFFFAOYSA-N 1-oxido-1,3-thiazole Chemical class [O-]S1=CN=C=C1 YGDWUQFZMXWDKE-UHFFFAOYSA-N 0.000 description 1
- JAAIPIWKKXCNOC-UHFFFAOYSA-N 1h-tetrazol-1-ium-5-thiolate Chemical class SC1=NN=NN1 JAAIPIWKKXCNOC-UHFFFAOYSA-N 0.000 description 1
- HAZJTCQWIDBCCE-UHFFFAOYSA-N 1h-triazine-6-thione Chemical class SC1=CC=NN=N1 HAZJTCQWIDBCCE-UHFFFAOYSA-N 0.000 description 1
- YKUDHBLDJYZZQS-UHFFFAOYSA-N 2,6-dichloro-1h-1,3,5-triazin-4-one Chemical compound OC1=NC(Cl)=NC(Cl)=N1 YKUDHBLDJYZZQS-UHFFFAOYSA-N 0.000 description 1
- SMZOUWXMTYCWNB-UHFFFAOYSA-N 2-(2-methoxy-5-methylphenyl)ethanamine Chemical compound COC1=CC=C(C)C=C1CCN SMZOUWXMTYCWNB-UHFFFAOYSA-N 0.000 description 1
- BDKLKNJTMLIAFE-UHFFFAOYSA-N 2-(3-fluorophenyl)-1,3-oxazole-4-carbaldehyde Chemical compound FC1=CC=CC(C=2OC=C(C=O)N=2)=C1 BDKLKNJTMLIAFE-UHFFFAOYSA-N 0.000 description 1
- NIXOWILDQLNWCW-UHFFFAOYSA-N 2-Propenoic acid Natural products OC(=O)C=C NIXOWILDQLNWCW-UHFFFAOYSA-N 0.000 description 1
- JKFYKCYQEWQPTM-UHFFFAOYSA-N 2-azaniumyl-2-(4-fluorophenyl)acetate Chemical compound OC(=O)C(N)C1=CC=C(F)C=C1 JKFYKCYQEWQPTM-UHFFFAOYSA-N 0.000 description 1
- PHPYXVIHDRDPDI-UHFFFAOYSA-N 2-bromo-1h-benzimidazole Chemical class C1=CC=C2NC(Br)=NC2=C1 PHPYXVIHDRDPDI-UHFFFAOYSA-N 0.000 description 1
- AYPSHJCKSDNETA-UHFFFAOYSA-N 2-chloro-1h-benzimidazole Chemical class C1=CC=C2NC(Cl)=NC2=C1 AYPSHJCKSDNETA-UHFFFAOYSA-N 0.000 description 1
- KRTDQDCPEZRVGC-UHFFFAOYSA-N 2-nitro-1h-benzimidazole Chemical class C1=CC=C2NC([N+](=O)[O-])=NC2=C1 KRTDQDCPEZRVGC-UHFFFAOYSA-N 0.000 description 1
- AGBXYHCHUYARJY-UHFFFAOYSA-N 2-phenylethenesulfonic acid Chemical class OS(=O)(=O)C=CC1=CC=CC=C1 AGBXYHCHUYARJY-UHFFFAOYSA-N 0.000 description 1
- KFZMGEQAYNKOFK-UHFFFAOYSA-N 2-propanol Substances CC(C)O KFZMGEQAYNKOFK-UHFFFAOYSA-N 0.000 description 1
- JSIAIROWMJGMQZ-UHFFFAOYSA-N 2h-triazol-4-amine Chemical class NC1=CNN=N1 JSIAIROWMJGMQZ-UHFFFAOYSA-N 0.000 description 1
- CBHTTYDJRXOHHL-UHFFFAOYSA-N 2h-triazolo[4,5-c]pyridazine Chemical class N1=NC=CC2=C1N=NN2 CBHTTYDJRXOHHL-UHFFFAOYSA-N 0.000 description 1
- OCVLSHAVSIYKLI-UHFFFAOYSA-N 3h-1,3-thiazole-2-thione Chemical class SC1=NC=CS1 OCVLSHAVSIYKLI-UHFFFAOYSA-N 0.000 description 1
- GDVFHEXRJFFDDB-UHFFFAOYSA-N 4-(5-sulfanylidene-2h-tetrazol-1-yl)benzoic acid Chemical compound C1=CC(C(=O)O)=CC=C1N1C(=S)N=NN1 GDVFHEXRJFFDDB-UHFFFAOYSA-N 0.000 description 1
- DSVIHYOAKPVFEH-UHFFFAOYSA-N 4-(hydroxymethyl)-4-methyl-1-phenylpyrazolidin-3-one Chemical compound N1C(=O)C(C)(CO)CN1C1=CC=CC=C1 DSVIHYOAKPVFEH-UHFFFAOYSA-N 0.000 description 1
- ZZEYCGJAYIHIAZ-UHFFFAOYSA-N 4-methyl-1-phenylpyrazolidin-3-one Chemical compound N1C(=O)C(C)CN1C1=CC=CC=C1 ZZEYCGJAYIHIAZ-UHFFFAOYSA-N 0.000 description 1
- UTMDJGPRCLQPBT-UHFFFAOYSA-N 4-nitro-1h-1,2,3-benzotriazole Chemical class [O-][N+](=O)C1=CC=CC2=NNN=C12 UTMDJGPRCLQPBT-UHFFFAOYSA-N 0.000 description 1
- GIQKIFWTIQDQMM-UHFFFAOYSA-N 5h-1,3-oxazole-2-thione Chemical compound S=C1OCC=N1 GIQKIFWTIQDQMM-UHFFFAOYSA-N 0.000 description 1
- 241001479434 Agfa Species 0.000 description 1
- NOWKCMXCCJGMRR-UHFFFAOYSA-N Aziridine Chemical compound C1CN1 NOWKCMXCCJGMRR-UHFFFAOYSA-N 0.000 description 1
- KAKZBPTYRLMSJV-UHFFFAOYSA-N Butadiene Chemical class C=CC=C KAKZBPTYRLMSJV-UHFFFAOYSA-N 0.000 description 1
- VYZAMTAEIAYCRO-UHFFFAOYSA-N Chromium Chemical compound [Cr] VYZAMTAEIAYCRO-UHFFFAOYSA-N 0.000 description 1
- 235000008733 Citrus aurantifolia Nutrition 0.000 description 1
- RWSOTUBLDIXVET-UHFFFAOYSA-N Dihydrogen sulfide Chemical class S RWSOTUBLDIXVET-UHFFFAOYSA-N 0.000 description 1
- 102000004190 Enzymes Human genes 0.000 description 1
- 108090000790 Enzymes Proteins 0.000 description 1
- VGGSQFUCUMXWEO-UHFFFAOYSA-N Ethene Chemical compound C=C VGGSQFUCUMXWEO-UHFFFAOYSA-N 0.000 description 1
- 239000005977 Ethylene Substances 0.000 description 1
- 239000001828 Gelatine Substances 0.000 description 1
- CTKINSOISVBQLD-UHFFFAOYSA-N Glycidol Chemical class OCC1CO1 CTKINSOISVBQLD-UHFFFAOYSA-N 0.000 description 1
- VVQNEPGJFQJSBK-UHFFFAOYSA-N Methyl methacrylate Chemical compound COC(=O)C(C)=C VVQNEPGJFQJSBK-UHFFFAOYSA-N 0.000 description 1
- 239000000020 Nitrocellulose Substances 0.000 description 1
- ISLYUUGUJKSGDZ-UHFFFAOYSA-N OC1=CC=NC2=NC=NN12 Chemical class OC1=CC=NC2=NC=NN12 ISLYUUGUJKSGDZ-UHFFFAOYSA-N 0.000 description 1
- IWPZUNZVUVXGPW-UHFFFAOYSA-N OS(=O)=S.Cc1ccccc1 Chemical compound OS(=O)=S.Cc1ccccc1 IWPZUNZVUVXGPW-UHFFFAOYSA-N 0.000 description 1
- 229910019142 PO4 Inorganic materials 0.000 description 1
- 239000004743 Polypropylene Substances 0.000 description 1
- ZLMJMSJWJFRBEC-UHFFFAOYSA-N Potassium Chemical compound [K] ZLMJMSJWJFRBEC-UHFFFAOYSA-N 0.000 description 1
- 229910021612 Silver iodide Inorganic materials 0.000 description 1
- LSNNMFCWUKXFEE-UHFFFAOYSA-N Sulfurous acid Chemical class OS(O)=O LSNNMFCWUKXFEE-UHFFFAOYSA-N 0.000 description 1
- ZMZDMBWJUHKJPS-UHFFFAOYSA-M Thiocyanate anion Chemical compound [S-]C#N ZMZDMBWJUHKJPS-UHFFFAOYSA-M 0.000 description 1
- 235000011941 Tilia x europaea Nutrition 0.000 description 1
- FJWGYAHXMCUOOM-QHOUIDNNSA-N [(2s,3r,4s,5r,6r)-2-[(2r,3r,4s,5r,6s)-4,5-dinitrooxy-2-(nitrooxymethyl)-6-[(2r,3r,4s,5r,6s)-4,5,6-trinitrooxy-2-(nitrooxymethyl)oxan-3-yl]oxyoxan-3-yl]oxy-3,5-dinitrooxy-6-(nitrooxymethyl)oxan-4-yl] nitrate Chemical compound O([C@@H]1O[C@@H]([C@H]([C@H](O[N+]([O-])=O)[C@H]1O[N+]([O-])=O)O[C@H]1[C@@H]([C@@H](O[N+]([O-])=O)[C@H](O[N+]([O-])=O)[C@@H](CO[N+]([O-])=O)O1)O[N+]([O-])=O)CO[N+](=O)[O-])[C@@H]1[C@@H](CO[N+]([O-])=O)O[C@@H](O[N+]([O-])=O)[C@H](O[N+]([O-])=O)[C@H]1O[N+]([O-])=O FJWGYAHXMCUOOM-QHOUIDNNSA-N 0.000 description 1
- XEIPQVVAVOUIOP-UHFFFAOYSA-N [Au]=S Chemical compound [Au]=S XEIPQVVAVOUIOP-UHFFFAOYSA-N 0.000 description 1
- DPSDYUYTQNAJJQ-UHFFFAOYSA-L [Na+].[Na+].OC(C)P(=O)([O-])OP(=O)[O-] Chemical compound [Na+].[Na+].OC(C)P(=O)([O-])OP(=O)[O-] DPSDYUYTQNAJJQ-UHFFFAOYSA-L 0.000 description 1
- 238000000862 absorption spectrum Methods 0.000 description 1
- DHKHKXVYLBGOIT-UHFFFAOYSA-N acetaldehyde Diethyl Acetal Natural products CCOC(C)OCC DHKHKXVYLBGOIT-UHFFFAOYSA-N 0.000 description 1
- 150000003926 acrylamides Chemical class 0.000 description 1
- 150000001253 acrylic acids Chemical class 0.000 description 1
- 150000008360 acrylonitriles Chemical class 0.000 description 1
- 238000004220 aggregation Methods 0.000 description 1
- 150000001299 aldehydes Chemical class 0.000 description 1
- 239000003513 alkali Substances 0.000 description 1
- 150000001336 alkenes Chemical class 0.000 description 1
- 125000003545 alkoxy group Chemical group 0.000 description 1
- 150000003973 alkyl amines Chemical class 0.000 description 1
- 150000001346 alkyl aryl ethers Chemical class 0.000 description 1
- 125000005907 alkyl ester group Chemical group 0.000 description 1
- 150000005215 alkyl ethers Chemical class 0.000 description 1
- 125000002947 alkylene group Chemical group 0.000 description 1
- 229940037003 alum Drugs 0.000 description 1
- 150000001412 amines Chemical class 0.000 description 1
- 235000001014 amino acid Nutrition 0.000 description 1
- 150000001413 amino acids Chemical class 0.000 description 1
- 125000004103 aminoalkyl group Chemical group 0.000 description 1
- 125000000129 anionic group Chemical group 0.000 description 1
- 230000002180 anti-stress Effects 0.000 description 1
- 238000000149 argon plasma sintering Methods 0.000 description 1
- 235000010323 ascorbic acid Nutrition 0.000 description 1
- 229960005070 ascorbic acid Drugs 0.000 description 1
- 239000011668 ascorbic acid Substances 0.000 description 1
- QVGXLLKOCUKJST-UHFFFAOYSA-N atomic oxygen Chemical compound [O] QVGXLLKOCUKJST-UHFFFAOYSA-N 0.000 description 1
- XHLMRAUSOZPJEM-UHFFFAOYSA-N benzenesulfonothioamide Chemical compound NS(=O)(=S)C1=CC=CC=C1 XHLMRAUSOZPJEM-UHFFFAOYSA-N 0.000 description 1
- 150000008366 benzophenones Chemical class 0.000 description 1
- IOJUPLGTWVMSFF-UHFFFAOYSA-N benzothiazole Chemical class C1=CC=C2SC=NC2=C1 IOJUPLGTWVMSFF-UHFFFAOYSA-N 0.000 description 1
- 235000021028 berry Nutrition 0.000 description 1
- 230000015572 biosynthetic process Effects 0.000 description 1
- 229910021538 borax Inorganic materials 0.000 description 1
- KGBXLFKZBHKPEV-UHFFFAOYSA-N boric acid Chemical compound OB(O)O KGBXLFKZBHKPEV-UHFFFAOYSA-N 0.000 description 1
- 239000004327 boric acid Substances 0.000 description 1
- 229940006460 bromide ion Drugs 0.000 description 1
- 244000309464 bull Species 0.000 description 1
- 150000001720 carbohydrates Chemical class 0.000 description 1
- 125000002091 cationic group Chemical group 0.000 description 1
- 229920002301 cellulose acetate Polymers 0.000 description 1
- 239000003153 chemical reaction reagent Substances 0.000 description 1
- 150000001844 chromium Chemical class 0.000 description 1
- 229910052804 chromium Inorganic materials 0.000 description 1
- 239000011651 chromium Substances 0.000 description 1
- WYYQVWLEPYFFLP-UHFFFAOYSA-K chromium(3+);triacetate Chemical compound [Cr+3].CC([O-])=O.CC([O-])=O.CC([O-])=O WYYQVWLEPYFFLP-UHFFFAOYSA-K 0.000 description 1
- 239000011247 coating layer Substances 0.000 description 1
- 239000000084 colloidal system Substances 0.000 description 1
- 238000012505 colouration Methods 0.000 description 1
- 239000007859 condensation product Substances 0.000 description 1
- QSAWQNUELGIYBC-UHFFFAOYSA-N cyclohexane-1,2-dicarboxylic acid Chemical compound OC(=O)C1CCCCC1C(O)=O QSAWQNUELGIYBC-UHFFFAOYSA-N 0.000 description 1
- 150000004691 decahydrates Chemical class 0.000 description 1
- 230000001419 dependent effect Effects 0.000 description 1
- 238000003745 diagnosis Methods 0.000 description 1
- 150000001991 dicarboxylic acids Chemical class 0.000 description 1
- 235000014113 dietary fatty acids Nutrition 0.000 description 1
- ZOMNIUBKTOKEHS-UHFFFAOYSA-L dimercury dichloride Chemical class Cl[Hg][Hg]Cl ZOMNIUBKTOKEHS-UHFFFAOYSA-L 0.000 description 1
- IYBUGHSUXJVLBQ-UHFFFAOYSA-L dipotassium;bromide;iodide Chemical compound [K+].[K+].[Br-].[I-] IYBUGHSUXJVLBQ-UHFFFAOYSA-L 0.000 description 1
- UQGFMSUEHSUPRD-UHFFFAOYSA-N disodium;3,7-dioxido-2,4,6,8,9-pentaoxa-1,3,5,7-tetraborabicyclo[3.3.1]nonane Chemical compound [Na+].[Na+].O1B([O-])OB2OB([O-])OB1O2 UQGFMSUEHSUPRD-UHFFFAOYSA-N 0.000 description 1
- AFOSIXZFDONLBT-UHFFFAOYSA-N divinyl sulfone Chemical compound C=CS(=O)(=O)C=C AFOSIXZFDONLBT-UHFFFAOYSA-N 0.000 description 1
- 238000004945 emulsification Methods 0.000 description 1
- 238000005516 engineering process Methods 0.000 description 1
- 125000004185 ester group Chemical group 0.000 description 1
- 125000001495 ethyl group Chemical group [H]C([H])([H])C([H])([H])* 0.000 description 1
- GWBBVOVXJZATQQ-UHFFFAOYSA-L etidronate disodium Chemical compound [Na+].[Na+].OP(=O)([O-])C(O)(C)P(O)([O-])=O GWBBVOVXJZATQQ-UHFFFAOYSA-L 0.000 description 1
- 238000005562 fading Methods 0.000 description 1
- 239000000194 fatty acid Substances 0.000 description 1
- 229930195729 fatty acid Natural products 0.000 description 1
- 125000003055 glycidyl group Chemical group C(C1CO1)* 0.000 description 1
- 150000002334 glycols Chemical class 0.000 description 1
- 229940015043 glyoxal Drugs 0.000 description 1
- PDMYFWLNGXIKEP-UHFFFAOYSA-K gold(3+);trithiocyanate Chemical compound [Au+3].[S-]C#N.[S-]C#N.[S-]C#N PDMYFWLNGXIKEP-UHFFFAOYSA-K 0.000 description 1
- 150000004820 halides Chemical class 0.000 description 1
- 150000002366 halogen compounds Chemical class 0.000 description 1
- 125000000623 heterocyclic group Chemical group 0.000 description 1
- 238000000265 homogenisation Methods 0.000 description 1
- 150000002429 hydrazines Chemical class 0.000 description 1
- ZMZDMBWJUHKJPS-UHFFFAOYSA-N hydrogen thiocyanate Natural products SC#N ZMZDMBWJUHKJPS-UHFFFAOYSA-N 0.000 description 1
- AKCUHGBLDXXTOM-UHFFFAOYSA-N hydroxy-oxo-phenyl-sulfanylidene-$l^{6}-sulfane Chemical compound SS(=O)(=O)C1=CC=CC=C1 AKCUHGBLDXXTOM-UHFFFAOYSA-N 0.000 description 1
- WYASEAQTEQVOJE-UHFFFAOYSA-N hydroxy-phenyl-sulfanylidene-$l^{4}-sulfane Chemical compound OS(=S)C1=CC=CC=C1 WYASEAQTEQVOJE-UHFFFAOYSA-N 0.000 description 1
- 125000002768 hydroxyalkyl group Chemical group 0.000 description 1
- 239000001866 hydroxypropyl methyl cellulose Substances 0.000 description 1
- UFVKGYZPFZQRLF-UHFFFAOYSA-N hydroxypropyl methyl cellulose Chemical compound OC1C(O)C(OC)OC(CO)C1OC1C(O)C(O)C(OC2C(C(O)C(OC3C(C(O)C(O)C(CO)O3)O)C(CO)O2)O)C(CO)O1 UFVKGYZPFZQRLF-UHFFFAOYSA-N 0.000 description 1
- 229920003088 hydroxypropyl methyl cellulose Polymers 0.000 description 1
- 235000010979 hydroxypropyl methyl cellulose Nutrition 0.000 description 1
- 238000010348 incorporation Methods 0.000 description 1
- 230000005764 inhibitory process Effects 0.000 description 1
- 230000000977 initiatory effect Effects 0.000 description 1
- 229910001505 inorganic iodide Inorganic materials 0.000 description 1
- 150000004694 iodide salts Chemical class 0.000 description 1
- 230000031700 light absorption Effects 0.000 description 1
- 239000004571 lime Substances 0.000 description 1
- 230000000670 limiting effect Effects 0.000 description 1
- 239000000314 lubricant Substances 0.000 description 1
- 238000004519 manufacturing process Methods 0.000 description 1
- 239000011159 matrix material Substances 0.000 description 1
- 239000006224 matting agent Substances 0.000 description 1
- 238000005259 measurement Methods 0.000 description 1
- 125000005395 methacrylic acid group Chemical class 0.000 description 1
- WSFSSNUMVMOOMR-NJFSPNSNSA-N methanone Chemical compound O=[14CH2] WSFSSNUMVMOOMR-NJFSPNSNSA-N 0.000 description 1
- ZAKLKBFCSHJIRI-UHFFFAOYSA-N mucochloric acid Natural products OC1OC(=O)C(Cl)=C1Cl ZAKLKBFCSHJIRI-UHFFFAOYSA-N 0.000 description 1
- 229920001220 nitrocellulos Polymers 0.000 description 1
- 150000004957 nitroimidazoles Chemical class 0.000 description 1
- 150000004010 onium ions Chemical class 0.000 description 1
- 150000002924 oxiranes Chemical class 0.000 description 1
- 229910052760 oxygen Inorganic materials 0.000 description 1
- 239000001301 oxygen Substances 0.000 description 1
- QUBQYFYWUJJAAK-UHFFFAOYSA-N oxymethurea Chemical compound OCNC(=O)NCO QUBQYFYWUJJAAK-UHFFFAOYSA-N 0.000 description 1
- 229950005308 oxymethurea Drugs 0.000 description 1
- 125000001997 phenyl group Chemical group [H]C1=C([H])C([H])=C(*)C([H])=C1[H] 0.000 description 1
- 235000021317 phosphate Nutrition 0.000 description 1
- XYFCBTPGUUZFHI-UHFFFAOYSA-O phosphonium Chemical compound [PH4+] XYFCBTPGUUZFHI-UHFFFAOYSA-O 0.000 description 1
- 150000003013 phosphoric acid derivatives Chemical class 0.000 description 1
- 239000000049 pigment Substances 0.000 description 1
- 239000004014 plasticizer Substances 0.000 description 1
- 229920003229 poly(methyl methacrylate) Polymers 0.000 description 1
- 229920001281 polyalkylene Polymers 0.000 description 1
- 229920001515 polyalkylene glycol Polymers 0.000 description 1
- 229920013639 polyalphaolefin Polymers 0.000 description 1
- 229920006289 polycarbonate film Polymers 0.000 description 1
- 229920000642 polymer Polymers 0.000 description 1
- 239000004926 polymethyl methacrylate Substances 0.000 description 1
- 229920000098 polyolefin Polymers 0.000 description 1
- 229920001155 polypropylene Polymers 0.000 description 1
- 229920001451 polypropylene glycol Polymers 0.000 description 1
- 239000004800 polyvinyl chloride Substances 0.000 description 1
- 229920000915 polyvinyl chloride Polymers 0.000 description 1
- 229910052700 potassium Inorganic materials 0.000 description 1
- 239000011591 potassium Substances 0.000 description 1
- ZNNZYHKDIALBAK-UHFFFAOYSA-M potassium thiocyanate Chemical compound [K+].[S-]C#N ZNNZYHKDIALBAK-UHFFFAOYSA-M 0.000 description 1
- 229940116357 potassium thiocyanate Drugs 0.000 description 1
- 238000004321 preservation Methods 0.000 description 1
- 239000000047 product Substances 0.000 description 1
- UGZVCHWAXABBHR-UHFFFAOYSA-O pyridin-1-ium-1-carboxamide Chemical class NC(=O)[N+]1=CC=CC=C1 UGZVCHWAXABBHR-UHFFFAOYSA-O 0.000 description 1
- HBCQSNAFLVXVAY-UHFFFAOYSA-N pyrimidine-2-thiol Chemical class SC1=NC=CC=N1 HBCQSNAFLVXVAY-UHFFFAOYSA-N 0.000 description 1
- 230000005855 radiation Effects 0.000 description 1
- 229930182490 saponin Natural products 0.000 description 1
- 150000007949 saponins Chemical class 0.000 description 1
- 235000017709 saponins Nutrition 0.000 description 1
- 150000003342 selenium Chemical class 0.000 description 1
- 230000008313 sensitization Effects 0.000 description 1
- 238000000926 separation method Methods 0.000 description 1
- 150000004756 silanes Chemical class 0.000 description 1
- 239000000377 silicon dioxide Substances 0.000 description 1
- 229940045105 silver iodide Drugs 0.000 description 1
- 235000017281 sodium acetate Nutrition 0.000 description 1
- 229940087562 sodium acetate trihydrate Drugs 0.000 description 1
- 235000010265 sodium sulphite Nutrition 0.000 description 1
- 239000004328 sodium tetraborate Substances 0.000 description 1
- 235000010339 sodium tetraborate Nutrition 0.000 description 1
- HLWRUJAIJJEZDL-UHFFFAOYSA-M sodium;2-[2-[bis(carboxymethyl)amino]ethyl-(carboxymethyl)amino]acetate Chemical compound [Na+].OC(=O)CN(CC(O)=O)CCN(CC(O)=O)CC([O-])=O HLWRUJAIJJEZDL-UHFFFAOYSA-M 0.000 description 1
- 238000001179 sorption measurement Methods 0.000 description 1
- 239000003381 stabilizer Substances 0.000 description 1
- 230000000087 stabilizing effect Effects 0.000 description 1
- 239000007858 starting material Substances 0.000 description 1
- 238000003756 stirring Methods 0.000 description 1
- 238000003860 storage Methods 0.000 description 1
- 150000003440 styrenes Chemical class 0.000 description 1
- 150000005846 sugar alcohols Polymers 0.000 description 1
- 229920001059 synthetic polymer Polymers 0.000 description 1
- JJJPTTANZGDADF-UHFFFAOYSA-N thiadiazole-4-thiol Chemical class SC1=CSN=N1 JJJPTTANZGDADF-UHFFFAOYSA-N 0.000 description 1
- DHCDFWKWKRSZHF-UHFFFAOYSA-L thiosulfate(2-) Chemical compound [O-]S([S-])(=O)=O DHCDFWKWKRSZHF-UHFFFAOYSA-L 0.000 description 1
- 150000003585 thioureas Chemical class 0.000 description 1
- 150000003606 tin compounds Chemical class 0.000 description 1
- 239000004408 titanium dioxide Substances 0.000 description 1
- OGIDPMRJRNCKJF-UHFFFAOYSA-N titanium oxide Inorganic materials [Ti]=O OGIDPMRJRNCKJF-UHFFFAOYSA-N 0.000 description 1
- 150000004684 trihydrates Chemical class 0.000 description 1
- 229920001567 vinyl ester resin Polymers 0.000 description 1
- 229920002554 vinyl polymer Polymers 0.000 description 1
- 239000000080 wetting agent Substances 0.000 description 1
- 239000004711 α-olefin Substances 0.000 description 1
Images
Classifications
-
- G—PHYSICS
- G03—PHOTOGRAPHY; CINEMATOGRAPHY; ANALOGOUS TECHNIQUES USING WAVES OTHER THAN OPTICAL WAVES; ELECTROGRAPHY; HOLOGRAPHY
- G03C—PHOTOSENSITIVE MATERIALS FOR PHOTOGRAPHIC PURPOSES; PHOTOGRAPHIC PROCESSES, e.g. CINE, X-RAY, COLOUR, STEREO-PHOTOGRAPHIC PROCESSES; AUXILIARY PROCESSES IN PHOTOGRAPHY
- G03C1/00—Photosensitive materials
- G03C1/005—Silver halide emulsions; Preparation thereof; Physical treatment thereof; Incorporation of additives therein
- G03C1/0051—Tabular grain emulsions
- G03C1/0053—Tabular grain emulsions with high content of silver chloride
-
- G—PHYSICS
- G03—PHOTOGRAPHY; CINEMATOGRAPHY; ANALOGOUS TECHNIQUES USING WAVES OTHER THAN OPTICAL WAVES; ELECTROGRAPHY; HOLOGRAPHY
- G03C—PHOTOSENSITIVE MATERIALS FOR PHOTOGRAPHIC PURPOSES; PHOTOGRAPHIC PROCESSES, e.g. CINE, X-RAY, COLOUR, STEREO-PHOTOGRAPHIC PROCESSES; AUXILIARY PROCESSES IN PHOTOGRAPHY
- G03C1/00—Photosensitive materials
- G03C1/005—Silver halide emulsions; Preparation thereof; Physical treatment thereof; Incorporation of additives therein
- G03C1/06—Silver halide emulsions; Preparation thereof; Physical treatment thereof; Incorporation of additives therein with non-macromolecular additives
- G03C1/08—Sensitivity-increasing substances
- G03C1/10—Organic substances
- G03C1/12—Methine and polymethine dyes
- G03C1/14—Methine and polymethine dyes with an odd number of CH groups
- G03C1/18—Methine and polymethine dyes with an odd number of CH groups with three CH groups
-
- G—PHYSICS
- G03—PHOTOGRAPHY; CINEMATOGRAPHY; ANALOGOUS TECHNIQUES USING WAVES OTHER THAN OPTICAL WAVES; ELECTROGRAPHY; HOLOGRAPHY
- G03C—PHOTOSENSITIVE MATERIALS FOR PHOTOGRAPHIC PURPOSES; PHOTOGRAPHIC PROCESSES, e.g. CINE, X-RAY, COLOUR, STEREO-PHOTOGRAPHIC PROCESSES; AUXILIARY PROCESSES IN PHOTOGRAPHY
- G03C1/00—Photosensitive materials
- G03C1/005—Silver halide emulsions; Preparation thereof; Physical treatment thereof; Incorporation of additives therein
- G03C1/46—Silver halide emulsions; Preparation thereof; Physical treatment thereof; Incorporation of additives therein having more than one photosensitive layer
-
- G—PHYSICS
- G03—PHOTOGRAPHY; CINEMATOGRAPHY; ANALOGOUS TECHNIQUES USING WAVES OTHER THAN OPTICAL WAVES; ELECTROGRAPHY; HOLOGRAPHY
- G03C—PHOTOSENSITIVE MATERIALS FOR PHOTOGRAPHIC PURPOSES; PHOTOGRAPHIC PROCESSES, e.g. CINE, X-RAY, COLOUR, STEREO-PHOTOGRAPHIC PROCESSES; AUXILIARY PROCESSES IN PHOTOGRAPHY
- G03C1/00—Photosensitive materials
- G03C1/005—Silver halide emulsions; Preparation thereof; Physical treatment thereof; Incorporation of additives therein
- G03C1/0051—Tabular grain emulsions
- G03C2001/0055—Aspect ratio of tabular grains in general; High aspect ratio; Intermediate aspect ratio; Low aspect ratio
-
- G—PHYSICS
- G03—PHOTOGRAPHY; CINEMATOGRAPHY; ANALOGOUS TECHNIQUES USING WAVES OTHER THAN OPTICAL WAVES; ELECTROGRAPHY; HOLOGRAPHY
- G03C—PHOTOSENSITIVE MATERIALS FOR PHOTOGRAPHIC PURPOSES; PHOTOGRAPHIC PROCESSES, e.g. CINE, X-RAY, COLOUR, STEREO-PHOTOGRAPHIC PROCESSES; AUXILIARY PROCESSES IN PHOTOGRAPHY
- G03C5/00—Photographic processes or agents therefor; Regeneration of such processing agents
- G03C5/16—X-ray, infrared, or ultraviolet ray processes
- G03C2005/168—X-ray material or process
-
- G—PHYSICS
- G03—PHOTOGRAPHY; CINEMATOGRAPHY; ANALOGOUS TECHNIQUES USING WAVES OTHER THAN OPTICAL WAVES; ELECTROGRAPHY; HOLOGRAPHY
- G03C—PHOTOSENSITIVE MATERIALS FOR PHOTOGRAPHIC PURPOSES; PHOTOGRAPHIC PROCESSES, e.g. CINE, X-RAY, COLOUR, STEREO-PHOTOGRAPHIC PROCESSES; AUXILIARY PROCESSES IN PHOTOGRAPHY
- G03C2200/00—Details
- G03C2200/03—111 crystal face
-
- G—PHYSICS
- G03—PHOTOGRAPHY; CINEMATOGRAPHY; ANALOGOUS TECHNIQUES USING WAVES OTHER THAN OPTICAL WAVES; ELECTROGRAPHY; HOLOGRAPHY
- G03C—PHOTOSENSITIVE MATERIALS FOR PHOTOGRAPHIC PURPOSES; PHOTOGRAPHIC PROCESSES, e.g. CINE, X-RAY, COLOUR, STEREO-PHOTOGRAPHIC PROCESSES; AUXILIARY PROCESSES IN PHOTOGRAPHY
- G03C5/00—Photographic processes or agents therefor; Regeneration of such processing agents
- G03C5/16—X-ray, infrared, or ultraviolet ray processes
Definitions
- the present invention relates to silver halide emulsions comprising tabular grains rich in chloride and to photographic materials containing such emulsions.
- Tabular grains are known in the photographic art for quite some time. As early as 1961 Berry et al described the preparation and growth of tabular silver bromoiodide grains in "Photographic Science and Engineering, Vol 5, No 6". A discussion of tabular grains appeared in Duffin, Photographic Emulsion Chemistry, Focal Press, 1966, p. 66-72.
- High aspect ratio tabular grains exhibit several pronounced photographic advantages. Thanks to their particular morphology greater amounts of spectral sensitizers can be adsorbed per mole of silver halide if compared with classical globular grains. As a consequence such spectrally sensitized tabular grains show an improved speed-granularity relationship and a wide separation between their blue and minus blue speed. Sharpness of photographic images can be improved using tabular grains thanks to their lower light scattering properties again compared with conventional globular emulsion grains. In color negative materials the conventional sequence of the light sensitive-layers can be altered and the yellow filter layer can be omitted. In developed black-and-white images high covering power is obtained even at high hardening levels. Alternatively reduced silver halide coverages can be achieved if required, resulting again in improved sharpness. In double-side coated radiographic materials the presence of tabular grains reduces the so-called cross-over which is the dominant factor for sharpness in such materials.
- Iodide and bromide ions can be incorporated into the grains, rich in chloride, during the nucleation step and/or during the growth step.
- iodide concentrations up to 3 mole % and even more preferably to limit them to 1.0 mole % or less, based on the total silver amount as higher concentrations retard development and lead to unsatisfactory sensitivities.
- the velocity of fixation can be disturbed in that case and as a consequence residual colouration may be unavoidable.
- the tabular silver chlorobromoiodide emulsion crystals used according to this invention preferably contain an amount of silver bromide of at most 10 mole %. To reduce the amount of replenisher in the processing, it is even more preferable to reduce the amount of bromide ions to less then 5 mole %.
- Bromide ions can be provided from at least one inorganic and/or organic agent providing bromide ions.
- Preferred emulsions comprising silver chlorobromoiodide or chloroiodide tabular crystals used according to this invention are those wherein an amount of 50 to 100 mole % of the total amount of iodide ions is located at the surface of the said emulsion crystals.
- the presence of iodide ions at the surface of the silver chloroiodide or silver chlorobromoiodide tabular crystals is particularly preferred as upon spectral sensitisation, an improved adsorption of the spectral sensitizer and an improved light absorption is obtained as the quantum efficiency detected in the photochemical processes is increased.
- Iodide ions are incorporated at the surface of said crystals by means of the so-called conversion technique.
- Iodide ions can be provided by the addition to the reaction vessel of inorganic iodide salts as e.g. potassium iodide, but for kinetic reasons, especially to obtain a more homogeneous iodide distribution, not only in the crystal lattice but over the whole crystal population, it can be more preferable to add iodide ions provided by organic agents releasing iodide ions or even by a combination of inorganic and organic agents providing iodide ions.
- inorganic iodide salts as e.g. potassium iodide
- organic agents releasing iodide ions or even by a combination of inorganic and organic agents providing iodide ions.
- Such agents have been described in EP-A 563 701, 563 708 and 561 415 and EP-Applications 93202899.6, filed October 15, 1993, and 93203040.6, filed October 29, 1993.
- Said agents releasing iodide ions promote a homogenous distribution of iodide ions in the crystal lattice and over the crystal population of the silver chlorobromoiodide or chloroiodide tabular crystals as the precipitation proceeds after homogenisation of the reagents added to the reaction vessel that are slowly releasing iodide ions.
- emulsions comprising tabular grains rich in chloride, having a flat hexagonal shape, are very useful in photographic elements if said grains contain at least 75 mole % of chloride, if at least 50 % of the total projected area of all the grains is provided by said tabular grains, if said tabular grains exhibit an average aspect ratio, defined as the ratio of the average equivalent diameter of the projective tabular crystal surface and its average thickness, of at least 8:1, and if the average thickness is from 0.08 ⁇ m to 0.2 ⁇ m as is illustrated in the Examples given hereinafter.
- Ultrathin grains with a thickness of less than 0.06 ⁇ m have been given in EP-A 0 534 325 In accordance with this invention it is preferred that at least 70 % of the total projected area is provided by said tabular grains, and even more preferably at least 90 %. In accordance with this invention an aspect ratio of at most 100:1 and more preferably of up to 50:1 is further preferred.
- tabular silver chlorobromoiodide or chloroiodide emulsion crystals are obtained having an amount of residual adenin of at most 0.3 mg per g of the equivalent amount of silver nitrate which is realized by applying the procedure set forth above.
- a low amount of residual adenin is in favour of spectral and chemical sensitisation and of the developability of the tabular crystals in the processing.
- Adenine is well-known as a habit modifying agent in the preparation of tabular crystals rich in chloride having (111) major faces as has been shown in EP-A 0 481 133.
- Additional gelatin can be added at a later stage of the emulsion preparation, e.g. after washing, to establish optimal coating conditions and/or to establish the required thickness of the coated emulsion layer.
- a gelatin to silver halide ratio the silver halide being expressed as the equivalent amount of silver nitrate, ranging from 0.3 to 1.0 is then obtained.
- spectral sensitisation traditionally follows the completion of chemical sensitisation. However in connection with tabular grains according to this invention, it is highly contemplated that spectral sensitisation can occur simultaneously with or even precede completely the chemical sensitisation step.
- a spectral sensitising dye may be added to the emulsion crystals just before cooling of the dispersion at the end of the growth stage, but in principle the addition of said dye may be performed at any stage of the precipitation, during or after redispersing or before, during or after chemical ripening. The addition can further be performed in one or more portions.
- the tabular silver chlorobromoiodide or chloroiodide emulsion crystals are spectrally sensitised with at least one spectral sensitiser having a J-aggregating structure when adsorbed at the crystal surface of the tabular grains according to this invention.
- tabular silver chlorobromoiodide or chloroiodide emulsion crystals according to this invention are spectrally sensitised with at least one dye having a structure corresponding to the general formula (I) given below.
- Z represents a nitrogen or an oxygen atom and is substituted with R'' if Z is a nitrogen atom
- R, R' and R'' each independently represent substituted or unsubstituted alkyl
- R''' represents hydrogen, substituted or unsubstituted alkyl or substituted or unsubstituted aryl
- T and T' may each independently represent the usual substituents known in the art
- (X ⁇ ) p represents an negatively charged atom or group of atoms to compensate for the positive charge(s) present on the dye
- (M+) q represents a positively charged atom or group of atoms to compensate for the negative charge(s) present on the dye
- p and q each represent an integer in order to get an electronically neutral compound.
- Another class of preferred spectral sensitisers suitable for use together with the emulsion crystals according to this invention are those of the type where R''' is hydrogen, Z is an nitrogen atom, wherein T represents 5-phenyl, 5-Cl, 5-OCH3 and 5-CH3 and wherein T' represents 5,6-(Cl)2; 5-CN-6-Cl; 5-CF3-6-Cl; 5-Cl; 5-CN, 5-CF3, 5-CHF2, 5-SO2CH3, 5-SO2R''''' (R''''' representing a fluoro-subsituted or non-fluoro-substituted alkylgroup), 5-COOR'''''' and 5-SO2-N(R x ) (R y ) or 5-CO-N(R x ) (R y ), wherein R x and R y each independently represent substituted or unsubstited alkyl groups, which may form a ring with the N-atom to which they are attached.
- Especially preferred structures of the type wherein Z represents oxygen are those wherein T and T' each represent Cl or T represents Cl and T' represents Phenyl or vice versa and wherein R and R' represent all combinations of the formulae -(CH2) n SO3 ⁇ wherein n equals 2, 3 or 4, -(CH2)2-CH(CH3)-SO3 ⁇ and -CH2-CHY-CH2-SO3 ⁇ wherein Y represents -OH or -Cl; and wherein R, corresponding to the formulae given above, is combined with R' corresponding to the formulae -(CH2) j H wherein j equals 1, 2, 3 or 4; -CH2-Phen-SO3 ⁇ -CH2-Phen-COOH -(CH2) k -Phen-COOH wherein k equals 1, 2 or 3.
- An example of a useful spectral sensitiser according to the general formula given above is anhydro-5,5'-dichloro-3,3'-bis(n-sulphobutyl)-9-ethyloxacarbo-cyanine hydroxide or anhydro-5,5'-dichloro-3,3'-bis(n-sulphopropyl)-9-ethyloxacarbocyanine hydroxide.
- a suitable mixture of spectral sensitizers that is applied is anhydro-5,5'-dichloro-3,3'-bis(n-sulphobutyl)-9-ethyl oxacarbocyanine hydroxide or anhydro-5,5'-dichloro-3,3'-bis(n-sulphopropyl)-9-ethyloxacarbo-cyanine hydroxide together with anhydro-5,5'-dicyano-1,1'-diethyl-3,3'-di(2-acetoxyethyl)ethylimidacarbocyanine bromide.
- Two or more types of tabular silver halide emulsions that have been prepared differently can be mixed for forming a photographic emulsion for use in accordance with the present invention.
- the size distribution of the grains in the emulsions comprising the silver chlorobromoiodide or chloroiodide tabular grains according to the present invention can be monodisperse or heterodisperse, the monodisperse distribution being preferred.
- the emulsion comprising the silver halide tabular grains in connection with the present invention can be chemically sensitised as described e.g. in "Chimie et Physique Photographique” by P. Glafkides, in “Photographic Emulsion Chemistry” by G.F. Duffin, in “Making and Coating Photographic Emulsion” by V.L. Zelikman et al, and in "Die Grundlagen der Photographischen mit Silberhalogeniden” edited by H. Frieser and published by Akademische Verlagsgesellschaft (1968).
- chemical sensitisation can be carried out by effecting the ripening in the presence of small amounts of compounds containing sulphur e.g.
- emulsions can be sensitized also by means of gold-sulphur ripeners or by means of reductors e.g. tin compounds as described in GB 789,823, amines, hydrazine derivatives, formamidine-sulphinic acids, and silane compounds.
- reductors e.g. tin compounds as described in GB 789,823, amines, hydrazine derivatives, formamidine-sulphinic acids, and silane compounds.
- the silver halide emulsion layer(s) in accordance with the present invention or the non-light-sensitive layers may comprise compounds preventing the formation of fog or stabilizing the photographic characteristics during the production or storage of the photographic elements or during the photographic treatment thereof.
- Many known compounds can be added as fog-inhibiting agent or stabilizer to the silver halide emulsion layer or to other coating layers in water-permeable relationship therewith such as an undercoat or a topcoat protective layer. Suitable examples are e.g.
- heterocyclic nitrogen-containing compounds such as benzothiazolium salts, nitroimidazoles, nitrobenzimidazoles, chlorobenzimidazoles, bromobenzimidazoles, mercaptothiazoles, mercaptobenzothiazoles, mercaptobenzimidazoles, mercaptothiadiazoles, aminotriazoles, benzotriazoles (preferably 5-methyl-benzotriazole), nitrobenzotriazoles, mercaptotetrazoles, in particular 1-phenyl-5-mercapto-tetrazole, mercaptopyrimidines, mercaptotriazines, benzothiazoline-2-thione, oxazoline-thione, triazaindenes, tetrazaindenes and pentazaindenes, especially those described by Birr in Z.
- benzothiazolium salts such as benzothiazolium salts, nitroimidazoles, nitrobenzimidazoles, chlor
- the photographic tabular grains in connection with the present invention can be used in various types of photographic elements. Because of their content rich in chloride such crystals are normally used for those applications which do not require extreme high sensitivity as in graphic arts e.g. recording materials for the output of scanners, phototypesetters and imagesetters, duplicating materials, radiographic hard-copy materials, diffusion transfer materials and black-and-white or colour print materials which produce prints starting from negatives in amateur or professional still photography or prints for cinematographic exhibition.
- the photographic material according to this invention comprises a support and on at least one side thereof at least one silver halide light-sensitive emulsion layer comprising silver chlorobromoiodide and/or chloroiodide tabular crystals as described hereinbefore.
- Development acceleration can be accomplished by incorporating in the emulsion layer or adjacent layers various compounds, preferably polyalkylene derivatives having a molecular weight of at least 400 such as those described in e.g. US Patents 3,038,805; 4,038,075 and 4,292,400 as well as in EP-A 634 688 and 94200639, filed March 11, 1994.
- the photographic element of the present invention may further comprise various kinds of surface-active agents in the photographic emulsion layer or in at least one other hydrophilic colloid layer.
- Suitable surface-active agents include non-ionic agents such as saponins, alkylene oxides e.g.
- polyethylene glycol polyethylene glycol/polypropylene glycol condensation products, polyethylene glycol alkyl ethers or polyethylene glycol alkylaryl ethers, polyethylene glycol esters, polyethylene glycol sorbitan esters, polyalkylene glycol alkylamines or alkylamides, silicone-polyethylene oxide adducts, glycidol derivatives, fatty acid esters of polyhydric alcohols and alkyl esters of saccharides; anionic agents comprising an acid group such as a carboxy, sulpho, phospho, sulphuric or phosphoric ester group; ampholytic agents such as aminoacids, aminoalkyl sulphonic acids, aminoalkyl sulphates or phosphates, alkyl betaines, and amine-N-oxides; and cationic agents such as alkylamine salts, aliphatic, aromatic, or heterocyclic quaternary ammonium salts, aliphatic or heterocyclic ring
- Such surface-active agents can be used for various purposes e.g. as coating aids, as compounds preventing electric charges, as compounds improving slidability, as compounds facilitating dispersive emulsification, as compounds preventing or reducing adhesion, and as compounds improving the photographic characteristics e.g higher contrast, sensitization, and development acceleration.
- Preferred surface-active coating agents are compounds containing perfluorinated alkyl groups.
- the photographic element of the present invention may further comprise various other additives such as e.g. compounds improving the dimensional stability of the photographic element, UV-absorbers, spacing agents and plasticizers.
- Suitable additives for improving the dimensional stability of the photographic element are e.g. dispersions of a water-soluble or hardly soluble synthetic polymer e.g. polymers of alkyl(meth)acrylates, alkoxy(meth)acrylates, glycidyl (meth)acrylates, (meth)acrylamides, vinyl esters, acrylonitriles, olefins, and styrenes, or copolymers of the above with acrylic acids, methacrylic acids, Alpha-Beta-unsaturated dicarboxylic acids, hydroxyalkyl (meth)acrylates, sulphoalkyl (meth)acrylates, and styrene sulphonic acids.
- a water-soluble or hardly soluble synthetic polymer e.g. polymers of alkyl(meth)acrylates, alkoxy(meth)acrylates, glycidyl (meth)acrylates, (meth)acrylamides, vinyl esters,
- UV-absorbers are e.g. aryl-substituted benzotriazole compounds as described in US Patent 3,533,794, 4-thiazolidone compounds as described in US Patent 3,314,794 and 3,352,681, benzophenone compounds as described in JP-A 2784/71, cinnamic ester compounds as described in US Patents 3,705,805 and 3,707,375, butadiene compounds as described in US Patent 4,045,229, and benzoxazole compounds as described in US Patent 3,700,455.
- UV-absorbers are especially useful in colour materials where they prevent the fading by light of the colour images formed after processing.
- Spacing agents can be present of which, in general, the average particle size is comprised between 0.2 and 10 ⁇ m. Spacing agents can be soluble or insoluble in alkali. Alkali-insoluble spacing agents usually remain permanently in the photographic element, whereas alkali-soluble spacing agents usually are removed therefrom in an alkaline processing bath. Suitable spacing agents can be made e.g. of polymethyl methacrylate, of copolymers of acrylic acid and methyl methacrylate, and of hydroxypropylmethyl cellulose hexahydrophthalate. Other suitable spacing agents have been described in US Patent 4,614,708.
- the photographic material can contain several non-light sensitive layers, e.g. an antistress topcoat layer, one or more backing layers, and one or more intermediate layers eventually containing filter- or antihalation dyes that absorb scattering light and thus promote the image sharpness.
- Suitable light-absorbing dyes used in these intermediate layers are described in e.g. US Patents 4,092,168, US 4,311,787, DE 2,453,217, GB Patent 7,907,440, EP-A's 0 586 748 and 0 587 230, US-P 5,344,749 and EP-A No. 93203382.2, filed December 2, 1993.
- the gelatin binder of the photographic material according to this invention can be forehardened with appropriate hardening agents such as those of the epoxide type, those of the ethylenimine type, those of the vinylsulfone type e.g. 1,3-vinylsulphonyl-2-propanol, di-(vinyl-sulphonyl)-methane or ethylene di-(vinyl-sulphone), chromium salts e.g. chromium acetate and chromium alum, aldehydes e.g. formaldehyde, glyoxal, and glutaraldehyde, N-methylol compounds e.g.
- appropriate hardening agents such as those of the epoxide type, those of the ethylenimine type, those of the vinylsulfone type e.g. 1,3-vinylsulphonyl-2-propanol, di-(vinyl-sulphony
- dimethylolurea and methyloldimethylhydantoin dioxan derivatives e.g. 2,3-dihydroxy-dioxan, active vinyl compounds e.g. 1,3,5-triacryloyl-hexahydro-s-triazine, active halogen compounds e.g. 2,4-dichloro-6-hydroxy-s-triazine, and mucohalogenic acids e.g. mucochloric acid and mucophenoxychloric acid.
- These hardeners can be used alone or in combination.
- the binder can also be hardened with fast-reacting hardeners such as carbamoylpyridinium salts as disclosed in US-P 4,063,952 and with the onium compounds as disclosed in EP-A 408,143.
- the photographic material according to this invention is preferably hardened to such an extent that the total amount of water absorption is at most 10 g/m2 per side after immersing the material for 3 minutes in demineralized water of 25°C.
- the photographic element according to this invention can contain one single emulsion layer, as it is the case for many applications, or it can be built up by two or even more emulsion layers.
- a material with a single or a duplitized emulsion layer coated on one or both sides of the support may contain silver halide emulsions according to the invention.
- duplitized emulsions differing in photographic speed by at least 0.15 log E a gain in cross-over exposure in double side coated materials can be obtained.
- the photographic material may contain several light-insensitive layers, e.g. a protective layer, one or more backing layers, one or more subbing layers, one or more intermediate layers e.g. filter layers and even an afterlayer containing e.g. the hardening agent(s), the antistatic agent(s), filter dyes for safety-light purposes etc.
- One or more backing layers can be provided at the non-light sensitive side of the support of materials coated with at least one emulsion layer at only one side of the support.
- These layers which can serve as anti-curl layer can contain e.g. matting agents like silica particles, lubricants, antistatic agents, light absorbing dyes, opacifying agents, e.g. titanium oxide and the usual ingredients like hardeners and wetting agents.
- the photographic material according to this invention is a double side coated X-ray material with a cross-over percentage of less than 40% even in the absence of an antihalation undercoating.
- the cross-over percentage (% cross-over) is therein determined as follows.
- the double side coated materials are adjusted between a single green light emitting screen (CURIX ortho Regular : Agfa-Gevaert trade name) and a white paper, replacing the second screen.
- This film-screen element directed with its light emitting screen to the X-ray tube, is then exposed with varying X-ray doses, expressed as log E.
- the support of the photographic material may be opaque or transparent, e.g. a paper support or resin support.
- a paper support preference is given to one coated at one or both sides with an Alpha-olefin polymer, e.g. a polyethylene layer which optionally contains an anti-halation dye or pigment. It is also possible to use an organic resin support e.g.
- cellulose nitrate film cellulose acetate film, poly(vinyl acetal) film, polystyrene film, poly(ethylene terephthalate) film, polycarbonate film, polyvinylchloride film or poly-Alpha-olefin films such as polyethylene or polypropylene film.
- the thickness of such organic resin film is preferably comprised between 0.07 and 0.35 mm.
- These organic resin supports are preferably coated with a subbing layer which can contain water insoluble particles such as silica or titanium dioxide.
- the photographic material containing tabular grains prepared according to the present invention can be image-wise exposed by any convenient radiation source in accordance with its specific application.
- processing conditions and composition of processing solutions are dependent from the specific type of photographic material in which the tabular grains prepared according to the present invention are applied.
- materials for X-ray diagnostic purposes said materials may be adapted to rapid processing conditions.
- an automatically operating processing apparatus is used provided with a system for automatic regeneration of the processing solutions.
- the forehardened material may be processed using one-part package chemistry or three-part package chemistry, depending on the processing application determining the degree of hardening required in said processing cycle. Applications within total processing times of 30 seconds and lower up to 90 seconds, known as common praxis, are possible.
- processing of a photographic material comprising the steps of developing, fixing, rinsing and drying can proceed, with a total processing time of at most 45 seconds thanks to the high degree of hardening of the said material as set forth hereinbefore. More preferably a processing of at most 38 seconds is possible.
- processing proceeds in a hardener-free developer and/or fixer.
- the developing proceeds in a developer comprising ascorbic acid as a developing solution.
- Emulsion A tabular silver chloride emulsion
- the pH value of the said dispersing medium was adjusted to a value of 3.0 with sulphuric acid, and after the addition of 55.5 ml of polystyrene sulphonic acid the obtained flocculate was decanted and washed three times with an amount of 6 l of demineralized water in order to remove the soluble salts present.
- the thus obtained silver chloride tabular emulsion showed the grain characteristics as illustrated in Table 1.
- the average diameter d EM , average thickness d, average aspect ratio AR were obtained from electron microscopic photographs: the diameter of the grain was defined as the diameter of the circle having an area equal to the projected area of the grain as viewed in the said photographs.
- the average sphere equivalent diameter d M obtained from the measurement of electric reduction currents obtained by reduction of a silver halide grain with a microscopically fine electrode is given: the sphere equivalent diameter was defined as the diameter of a hypothetical spherical grain with the same volume as the corresponding tabular grain.
- Emulsions B1 to B6 comparative tabular silver chloroiodide emulsions
- Emulsions B1 to B6 For the preparation of the Emulsions B1 to B6, an amount of potassium iodide was added to B1, in order to obtain an iodide content in the tabular silver chloroiodide crystals of respectively 0.25 (B1); 0.50 (B2); 0.75 (B3); 1.00 (B4); 1.50 (B5) and 2.00 (B6) mole %.
- Grain characteristics are summarized in Table 1.
- Chemical ripening agents were gold thiocyanate, sodium thiosulphate as a source of sulphur and toluene thiosulphonic acid was used as predigestion agent.
- the amounts of each chemical ripening were optimized in order to obtain an optimal fog-sensitivity relationship after 2 hours at 57°C.
- each emulsion was stabilized with 1-p-carboxyphenyl-5-mercaptotetrazole and after addition of the normal coating additives the solutions were coated simultaneously together with a protective layer containing 1.3 g gelatine per m2 per side on both sides of a polyethylene terephthalate film support having a thickness of 175 ⁇ m.
- the resulting photographic material contained per side an amount of silver halide corresponding to 4.5 grams of AgNO3 per m2 and an amount of gelatin corresponding to 3.55 g/m2.
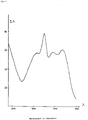
- the absorption spectra measured from the unexposed and unprocessed materials show an increase of the absorption density at a wavelength value of 510 nm and of 545 nm with respectively about 20 and 80 %, corresponding to the absorption maxima of the spectrally sensitizing dye adsorbed at the silver chloride crystal surface and in the gelatin matrix. From these data it can be concluded that the tabular silver chloride crystals containing iodide ions show an improved dye absorption if compared with tabular silver chloride crystals prepared iodide-free (see Fig. 1: absorption curve for iodide-free silver chloride crystals; Fig. 2: absorption curve for silver chloride crystals having a silver iodide containing phase at its crystal surface).
- Said processing composition is given hereinafter: demineralized water 500 ml hydroquinone 40 g phenidone B 1.6 g potassium bromide 5 g potassium thiocyanate 2 g aqueous potassium sulphite (655 g/l) 300 ml aqueous potassium carbonate (765 g/l) 80 ml aqueous potassium hydroxyde (755 g/l) 14 ml Trilon B (trade name for Na4EDTA from BASF) 8 ml Turpinal 2NZ (trade name for 1-hydroxy-ethyldiphosphonic acid disodium salt) 2 g Polyoxyethylene (M.W.: 200) 40 ml pH 10.70 Water to make 1 l
- CHLORDEV The concentrated, hardener-free developer, called “CHLORDEV” has to be diluted with the same amount of demineralised water (pH of developer ready-for-use: 10.46).
- pH of developer ready-for-use 10.46
- Table 2 the sensitometric results obtained are given.
- the density as a function of the light dose was measured and therefrom were determined the following parameters:
- Emulsions K1-K2 comparative tabular silver chlorobromide emulsion
- the said emulsions K1 and K2 were prepared without the presence of iodide ions, nor in B1, nor in B2. Instead of potassium iodide potassium bromide was added to B1 in an amount in order to obtain a silver chlorobromide emulsion having 5 mole % of bromide for K1 and 10 mole % of bromide for K2.
- a nucleation step was performed by introducing solution A and solution B simultaneously in dispersion medium C both at a flow rate of 60 ml/min during 30 seconds. After a physical ripening time of 20 min during which the temperature is raised to 70 °C a growth step was performed by introducing by a double jet during 28 minutes and 48 seconds solution A starting at a flow rate of 5 ml/min and linearly increasing the flow rate to an end value of 13.73 ml/min, and solution B at an increasing flow rate in order to maintain a constant mV value, measured by a silver electrode versus S.C.E., of +103 mV.
- the said growth step was continued at a the lower starting flow rate for solution A of 5 ml/min during 16 minutes while solution B was added at a flow rate so as to maintain the mV at a constant value of +135 mV.
- a further increase to a flow rate of 25 ml/min. in a time period of 41 minutes and 20 seconds for solution A was the next step, during which the pAg value was kept at +135 mV.
- Values of d M (in ⁇ m) were determined as described hereinbefore: values of 0.79 and 0.81 were measured for emulsions K1 and K2 resp..
- Emulsions L1-L2 comparative tabular silver chlorobromoiodide emulsions
- the emulsions K1, K2, L1 and L2 were coated in the same way as described hereinbefore for the emulsions A and B1-B6.
- Emulsions T1-T8 (inventive tabular silver chloroiodide emulsions)
- Emulsions T1-T8 For the preparation of the Emulsions T1-T8, an amount of potassium iodide was added to B1, just as for the comparative Emulsions B1-B6 in order to obtain an iodide content in the tabular silver chloroiodide crystals of 1.3 mole %.
- Variable parameters in the preparation method of the inventive Emulsions T1-T8 were the concentration of adenin in the reaction vessel (given in mg per 500 g of silver nitrate used in the precipitation, the pAg value (expressed in mV vs. S.C.E.) during the nucleation step and the temperature T (in °C) of the reaction vessel in the said nucleation step.
- Emulsions T1-T8 were chemically and spectrally sensitised and further coated in the same way as described hereinbefore for the Emulsions J, K and L.
- the coatings were exposed in the same way and the processing which was performed as in the G138 developer from Agfa-Gevaert containing hardening agent and in the experimental hardener-free developer CHLORDEV replacing G138, the composition of which is given above.
- Results obtained for fog F, sensitivity S, contrast G and maximum density DMAX with emulsions T1-T8 are summarized in Table 5, just as in Table 3.
- the specifications for a CURIX ORTHO film material, trade name product from Agfa-Gevaert, which have to be attained, are added to the Table 5.
- Emulsions TV1-TV3 (inventive tabular silver chloroiodide emulsions with a variable iodide profile)
- Emulsions TV1-TV3 For the preparation of the Emulsions TV1-TV3 an amount of potassium iodide was added to B1, and the preparation followed the steps just as for the inventive Emulsions T1-T8 with the difference that the amount of iodide was varied: 0.5 mole % for TV1; 1.0 mole % for TV2 and 2 mole % for TV3.
- Emulsions TV2 and TV3 no further additions of iodide were performed, so that the total amount of iodide ions for both Emulsions was 1 mole % and 2 mole % respectively.
- Emulsion TV1 an iodide content in the silver chloroiodide tabular crystals of 1.3 mole % was obtained by adding a further amount of 0.8 mole % at the end of the preparation stage just as for the Emulsions L1 and L2.
- Table 7 Em. d EM ( ⁇ m) d ( ⁇ m) AR d M ( ⁇ m) TV1 1.27 0.14 8.8 0.70 TV2 1.51 0.19 8.1 0.86 TV3 1.25 0.17 7.5 0.73
- Emulsions TV1-TV3 were coated in the same way as described hereinbefore for the Emulsions J, K, L and T.
- the coatings were exposed in the same way as well as the processing which was performed as in the developer G138, from Agfa-Gevaert, containing a hardener and in the hardener-free developer CHLORDEV replacing G138, the composition of which has been given hereinbefore.
- Results obtained for fog F, sensitivity S, contrast G and maximum density DMAX with the said emulsions are summarized in Table 7, just as in Tables 3 and 5.
- the specifications, which should be attained for a CURIX ORTHO film material, trade name from Agfa-Gevaert, processed in G138, are added to this Table.
- Emulsion TV1 lower amounts of iodide are leading to fog increase, loss in sensitivity and gradation; higher amounts although preventing fog increase are leading to a still more remarkable loss in sensitivity and gradation.
- iodide ions at the crystal surface of the silver chlorobromoiodide or silver chloroiodide crystals is thus particularly favourable.
- a suitable black image tone is observed for all samples of the materials coated from emulsions according to this invention.
Priority Applications (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| EP95200651A EP0678772A1 (de) | 1994-04-06 | 1995-03-17 | Lichtempfindliches Silberchlorobromojodid- oder Silbuchlorojodid-Tafelkörner enthaltendes Material |
Applications Claiming Priority (3)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| EP94200932 | 1994-04-06 | ||
| EP94200932 | 1994-04-06 | ||
| EP95200651A EP0678772A1 (de) | 1994-04-06 | 1995-03-17 | Lichtempfindliches Silberchlorobromojodid- oder Silbuchlorojodid-Tafelkörner enthaltendes Material |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| EP0678772A1 true EP0678772A1 (de) | 1995-10-25 |
Family
ID=26136154
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| EP95200651A Withdrawn EP0678772A1 (de) | 1994-04-06 | 1995-03-17 | Lichtempfindliches Silberchlorobromojodid- oder Silbuchlorojodid-Tafelkörner enthaltendes Material |
Country Status (1)
| Country | Link |
|---|---|
| EP (1) | EP0678772A1 (de) |
Cited By (6)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| EP0862088A1 (de) * | 1997-03-01 | 1998-09-02 | Agfa-Gevaert N.V. | System und Verfahren zur Röntgenbild Herstellung |
| US5912108A (en) * | 1996-12-30 | 1999-06-15 | Agfa-Gevaert, N.V. | Processing of a light-sensitive silver halide photographic material |
| EP0953867A1 (de) * | 1998-04-29 | 1999-11-03 | Agfa-Gevaert N.V. | Verfahren zur spektralen Sensibilisierung von tafelförmigen Silberhalogenidkörnern |
| EP0962820A1 (de) * | 1998-06-05 | 1999-12-08 | Agfa-Gevaert N.V. | Verarbeitung von radiographischen Materialien,die Emulsionskörner mit hohem Silberchlorid enthalten |
| US6300051B1 (en) | 1998-04-29 | 2001-10-09 | Agfa-Gevaert | Method to spectrally sensitize tabular silver halide grains |
| WO2005050315A1 (en) * | 2003-11-12 | 2005-06-02 | Eastman Kodak Company | High-speed radiographic film |
Citations (5)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JPH04161947A (ja) * | 1990-10-25 | 1992-06-05 | Fuji Photo Film Co Ltd | 写真用ハロゲン化銀乳剤の製造方法及びそれを用いた感光材料の現像処理方法 |
| EP0534325A1 (de) * | 1991-09-20 | 1993-03-31 | Eastman Kodak Company | Emulsionen mit ultradünnen tafelförmigen Körnern eines hohen Chloridgehaltes |
| EP0534395A1 (de) * | 1991-09-24 | 1993-03-31 | Eastman Kodak Company | Hochchloridhaltige Emulsionen hoher Tafelförmigkeit von aussergewöhnlicher Stabilität |
| EP0535467A1 (de) * | 1991-09-20 | 1993-04-07 | Eastman Kodak Company | Verfahren zur Herstellung von Emulsionen mit tafelförmigen Körnern von hohem Chloridgehalt (III) |
| EP0620479A1 (de) * | 1993-04-16 | 1994-10-19 | Eastman Kodak Company | Emulsion mit Iodid-Streifen aufweisenden tafelförmigen Körnern |
-
1995
- 1995-03-17 EP EP95200651A patent/EP0678772A1/de not_active Withdrawn
Patent Citations (5)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JPH04161947A (ja) * | 1990-10-25 | 1992-06-05 | Fuji Photo Film Co Ltd | 写真用ハロゲン化銀乳剤の製造方法及びそれを用いた感光材料の現像処理方法 |
| EP0534325A1 (de) * | 1991-09-20 | 1993-03-31 | Eastman Kodak Company | Emulsionen mit ultradünnen tafelförmigen Körnern eines hohen Chloridgehaltes |
| EP0535467A1 (de) * | 1991-09-20 | 1993-04-07 | Eastman Kodak Company | Verfahren zur Herstellung von Emulsionen mit tafelförmigen Körnern von hohem Chloridgehalt (III) |
| EP0534395A1 (de) * | 1991-09-24 | 1993-03-31 | Eastman Kodak Company | Hochchloridhaltige Emulsionen hoher Tafelförmigkeit von aussergewöhnlicher Stabilität |
| EP0620479A1 (de) * | 1993-04-16 | 1994-10-19 | Eastman Kodak Company | Emulsion mit Iodid-Streifen aufweisenden tafelförmigen Körnern |
Non-Patent Citations (1)
| Title |
|---|
| PATENT ABSTRACTS OF JAPAN vol. 16, no. 457 (P - 1426) 22 September 1992 (1992-09-22) * |
Cited By (8)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US5912108A (en) * | 1996-12-30 | 1999-06-15 | Agfa-Gevaert, N.V. | Processing of a light-sensitive silver halide photographic material |
| EP0862088A1 (de) * | 1997-03-01 | 1998-09-02 | Agfa-Gevaert N.V. | System und Verfahren zur Röntgenbild Herstellung |
| EP0953867A1 (de) * | 1998-04-29 | 1999-11-03 | Agfa-Gevaert N.V. | Verfahren zur spektralen Sensibilisierung von tafelförmigen Silberhalogenidkörnern |
| US6300051B1 (en) | 1998-04-29 | 2001-10-09 | Agfa-Gevaert | Method to spectrally sensitize tabular silver halide grains |
| EP0962820A1 (de) * | 1998-06-05 | 1999-12-08 | Agfa-Gevaert N.V. | Verarbeitung von radiographischen Materialien,die Emulsionskörner mit hohem Silberchlorid enthalten |
| US6066441A (en) * | 1998-06-05 | 2000-05-23 | Agfa-Gevaert, N.V. | Processing of radiographic materials having emulsion grains rich in silver chloride |
| WO2005050315A1 (en) * | 2003-11-12 | 2005-06-02 | Eastman Kodak Company | High-speed radiographic film |
| US6989223B2 (en) | 2003-11-12 | 2006-01-24 | Eastman Kodak Company | High-speed radiographic film |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| US5595864A (en) | Method for making tabular silver halide grains | |
| EP0712034B1 (de) | Verfahren zur spektralen Sensibilisierung von tafelförmigen Silberhalogenidkörnern | |
| EP0678772A1 (de) | Lichtempfindliches Silberchlorobromojodid- oder Silbuchlorojodid-Tafelkörner enthaltendes Material | |
| EP0503700B1 (de) | Verfahren zur Herstellung von Jodobromid-Emulsionen, die monodisperse tabulare Körner enthalten | |
| US5286621A (en) | Method for the preparation of tabular emulsion grains rich in chloride | |
| EP0569075B1 (de) | Verfahren zur Herstellung von tafelförmigen Silberhalogenid-Körnern | |
| EP0911687B1 (de) | Verfahren zur Herstellung von morphologisch homogenen silberbromidreichen tafelförmigen (111)-Kristallen | |
| US5498518A (en) | Chloride rich tabular grain emulsion with (100) major faces | |
| US6030762A (en) | Method of preparing {111} tabular silver chloro(bromo)iodide crystals | |
| US6277552B1 (en) | Shallow electron trap dopants in silver halide tabular grain emulsions for use in medical diagnostic imaging materials | |
| EP0651284A1 (de) | Fällung von Silberhalogenidkristallen, die lodid enthalten | |
| EP0843208A1 (de) | Verfahren zur Herstellung silberbromidreicher Tafelkörner in Gegenwart spezifischer Gelatinen | |
| US5733715A (en) | Silver halide photographic materials comprising silver bromide or bromoiodide emulsions having triangular tabular crystals | |
| EP0843207B1 (de) | Verfahren zur Herstellung einer verbesserten photographischen Emulsion mit chloridreichen Tafelkörnern | |
| EP0770909A1 (de) | Photographisches Silberhalogenidmehrschichtmaterial und Verfahren zu dessen Herstellung | |
| US5795704A (en) | Method of hardener-free processing of a forehardened silver halide photographic material | |
| EP0610608B1 (de) | Photographisches, lichtempfindliches Silberhalogenidmaterial zur Wiedergabe von medizinischen laseraufgezeichneten Bildern und Verfahren zur Verarbeitung | |
| US6436621B1 (en) | Multilayer silver halide photographic material and process for preparing the same | |
| EP0709730B1 (de) | Verfahren zur Verarbeitung eines photographischen Silberhalogenidmaterials | |
| US6136524A (en) | Light-sensitive emulsion having (100) tabular grains rich in silver chloride and method for preparing said grains | |
| US6083678A (en) | Method for preparing a light-sensitive emulsion having (100) tabular grains rich in silver chloride | |
| US6280915B1 (en) | Spectrally sensitized tabular grain photographic materials | |
| US6558892B2 (en) | Method of preparing ultrathin light-sensitive tabular grain emulsions rich in silver bromide | |
| EP0465728A1 (de) | Stabilisierung von tabularen Körnern durch Pyrimidinderivate | |
| EP0754964A1 (de) | Photographische Silberhalogenidmaterialien, die dreieckige Tafelkörner aufweisende Silberbromid- oder Bromidjodidemulsionen enthalten |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| PUAI | Public reference made under article 153(3) epc to a published international application that has entered the european phase |
Free format text: ORIGINAL CODE: 0009012 |
|
| AK | Designated contracting states |
Kind code of ref document: A1 Designated state(s): BE DE FR GB NL |
|
| 17P | Request for examination filed |
Effective date: 19960425 |
|
| R17P | Request for examination filed (corrected) |
Effective date: 19960425 |
|
| 17Q | First examination report despatched |
Effective date: 19971016 |
|
| STAA | Information on the status of an ep patent application or granted ep patent |
Free format text: STATUS: THE APPLICATION HAS BEEN WITHDRAWN |
|
| 18W | Application withdrawn |
Withdrawal date: 19980203 |