EP0903405A2 - Composition pour lave-vaisselle - Google Patents
Composition pour lave-vaisselle Download PDFInfo
- Publication number
- EP0903405A2 EP0903405A2 EP98307432A EP98307432A EP0903405A2 EP 0903405 A2 EP0903405 A2 EP 0903405A2 EP 98307432 A EP98307432 A EP 98307432A EP 98307432 A EP98307432 A EP 98307432A EP 0903405 A2 EP0903405 A2 EP 0903405A2
- Authority
- EP
- European Patent Office
- Prior art keywords
- tablet
- dish washing
- silicone
- machine dish
- packaging system
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Withdrawn
Links
Images
Classifications
-
- C—CHEMISTRY; METALLURGY
- C11—ANIMAL OR VEGETABLE OILS, FATS, FATTY SUBSTANCES OR WAXES; FATTY ACIDS THEREFROM; DETERGENTS; CANDLES
- C11D—DETERGENT COMPOSITIONS; USE OF SINGLE SUBSTANCES AS DETERGENTS; SOAP OR SOAP-MAKING; RESIN SOAPS; RECOVERY OF GLYCEROL
- C11D3/00—Other compounding ingredients of detergent compositions covered in group C11D1/00
- C11D3/16—Organic compounds
- C11D3/37—Polymers
- C11D3/3703—Macromolecular compounds obtained otherwise than by reactions only involving carbon-to-carbon unsaturated bonds
- C11D3/373—Macromolecular compounds obtained otherwise than by reactions only involving carbon-to-carbon unsaturated bonds containing silicones
- C11D3/3742—Nitrogen containing silicones
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B65—CONVEYING; PACKING; STORING; HANDLING THIN OR FILAMENTARY MATERIAL
- B65D—CONTAINERS FOR STORAGE OR TRANSPORT OF ARTICLES OR MATERIALS, e.g. BAGS, BARRELS, BOTTLES, BOXES, CANS, CARTONS, CRATES, DRUMS, JARS, TANKS, HOPPERS, FORWARDING CONTAINERS; ACCESSORIES, CLOSURES, OR FITTINGS THEREFOR; PACKAGING ELEMENTS; PACKAGES
- B65D75/00—Packages comprising articles or materials partially or wholly enclosed in strips, sheets, blanks, tubes, or webs of flexible sheet material, e.g. in folded wrappers
- B65D75/28—Articles or materials wholly enclosed in composite wrappers, i.e. wrappers formed by associating or interconnecting two or more sheets or blanks
- B65D75/30—Articles or materials enclosed between two opposed sheets or blanks having their margins united, e.g. by pressure-sensitive adhesive, crimping, heat-sealing, or welding
- B65D75/32—Articles or materials enclosed between two opposed sheets or blanks having their margins united, e.g. by pressure-sensitive adhesive, crimping, heat-sealing, or welding one or both sheets or blanks being recessed to accommodate contents
- B65D75/325—Articles or materials enclosed between two opposed sheets or blanks having their margins united, e.g. by pressure-sensitive adhesive, crimping, heat-sealing, or welding one or both sheets or blanks being recessed to accommodate contents one sheet being recessed, and the other being a flat not- rigid sheet, e.g. puncturable or peelable foil
- B65D75/327—Articles or materials enclosed between two opposed sheets or blanks having their margins united, e.g. by pressure-sensitive adhesive, crimping, heat-sealing, or welding one or both sheets or blanks being recessed to accommodate contents one sheet being recessed, and the other being a flat not- rigid sheet, e.g. puncturable or peelable foil and forming several compartments
-
- C—CHEMISTRY; METALLURGY
- C11—ANIMAL OR VEGETABLE OILS, FATS, FATTY SUBSTANCES OR WAXES; FATTY ACIDS THEREFROM; DETERGENTS; CANDLES
- C11D—DETERGENT COMPOSITIONS; USE OF SINGLE SUBSTANCES AS DETERGENTS; SOAP OR SOAP-MAKING; RESIN SOAPS; RECOVERY OF GLYCEROL
- C11D17/00—Detergent materials or soaps characterised by their shape or physical properties
- C11D17/0047—Detergents in the form of bars or tablets
- C11D17/0065—Solid detergents containing builders
- C11D17/0073—Tablets
- C11D17/0091—Dishwashing tablets
-
- C—CHEMISTRY; METALLURGY
- C11—ANIMAL OR VEGETABLE OILS, FATS, FATTY SUBSTANCES OR WAXES; FATTY ACIDS THEREFROM; DETERGENTS; CANDLES
- C11D—DETERGENT COMPOSITIONS; USE OF SINGLE SUBSTANCES AS DETERGENTS; SOAP OR SOAP-MAKING; RESIN SOAPS; RECOVERY OF GLYCEROL
- C11D17/00—Detergent materials or soaps characterised by their shape or physical properties
- C11D17/04—Detergent materials or soaps characterised by their shape or physical properties combined with or containing other objects
- C11D17/041—Compositions releasably affixed on a substrate or incorporated into a dispensing means
-
- C—CHEMISTRY; METALLURGY
- C11—ANIMAL OR VEGETABLE OILS, FATS, FATTY SUBSTANCES OR WAXES; FATTY ACIDS THEREFROM; DETERGENTS; CANDLES
- C11D—DETERGENT COMPOSITIONS; USE OF SINGLE SUBSTANCES AS DETERGENTS; SOAP OR SOAP-MAKING; RESIN SOAPS; RECOVERY OF GLYCEROL
- C11D3/00—Other compounding ingredients of detergent compositions covered in group C11D1/00
- C11D3/16—Organic compounds
- C11D3/37—Polymers
- C11D3/3703—Macromolecular compounds obtained otherwise than by reactions only involving carbon-to-carbon unsaturated bonds
- C11D3/373—Macromolecular compounds obtained otherwise than by reactions only involving carbon-to-carbon unsaturated bonds containing silicones
-
- C—CHEMISTRY; METALLURGY
- C11—ANIMAL OR VEGETABLE OILS, FATS, FATTY SUBSTANCES OR WAXES; FATTY ACIDS THEREFROM; DETERGENTS; CANDLES
- C11D—DETERGENT COMPOSITIONS; USE OF SINGLE SUBSTANCES AS DETERGENTS; SOAP OR SOAP-MAKING; RESIN SOAPS; RECOVERY OF GLYCEROL
- C11D3/00—Other compounding ingredients of detergent compositions covered in group C11D1/00
- C11D3/39—Organic or inorganic per-compounds
- C11D3/3942—Inorganic per-compounds
Definitions
- the present invention relates to a tablet for use in a dish washing machine, a method of preparing the tablet and packaging for the tablet.
- Detergent tablets for dish-washing machines are liked by consumers as they find them are more convenient to use than alternative products such as powders and the like.
- Powder flowability is an important factor to consider in the manufacture of a tablet. Powders with a low flowability (less than 50 ml/s) can have two negative aspects namely: they can compact at the press feed line reducing throughput and they do not flow at a rate which is compatible with the pressing cycle and thus do not completely fill the tablet forming mold.
- Manufacture of tablets can also be difficult due to difficulty in ejecting the tablet from a mold. This difficulty means that the mechanical wear on the tabletting machine is increased and may flaws can occur on the tablet's surface
- binding and disintegrating agents may contain binding and disintegrating agents.
- An important property of these such agents is that they are compatible with the active ingredients in the tablet. Frequently an agent can have a dual role e.g., a binding agent also acts as a disintegrating agent.
- Effervescent systems have been included in a tablets formulation to increase their dissolution rate.
- Such effervescent systems includes weak acids or acid salts such as citric acid, maleic acid, sodium and hydrogen phosphates, in combination with a basic ingredient that liberates carbon dioxide when interacting with this source of acid. Examples include sodium and potassium carbonate and bicarbonate and sodium sesquicarbonate.
- the present invention relates to a tablet that dissolves rapidly when added to water and is easy to manufacture accordingly the present invention relates to a machine dish washing tablet comprising a bleaching system, a silicone and less than 10 wt% of surfactant.
- the invention also relates to a process for preparing machine dish washing tablets comprising the step of adding a silicone emulsion to a formulation that is to be tableted.
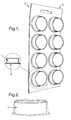
- the invention also relates to a dish washing tablet(1) and a packaging system (2) the packaging system comprising a body (3) shaped to receive a tablet and a cover(4) which keeps the tablet in the body.
- a dish washing tablet (1) and a packaging system (2) the packaging system comprising a body (3) shaped to receive a tablet and a cover(4) which keeps the tablet in the body.
- the use of this packaging system to store dish washing tablet is also described.
- the tablets according to the present invention not only dissolve quickly in the dish washing machine but they are easy to process in that the powder used to manufacture the tablets has good flowability, does not adhere to the punch's surface and the tablets produced are easily ejected from the mold.
- tablets of the invention can be prepared by conventional presses instead of powerful presses which are commonly used to for making detergent tablets.
- Conventional presses are those with engines with up to 5hp, a sole compression stage with a working capacity of up to 12t/cm 2 and production rate of 18000 tablets per hour.
- the cost of these presses is up to 8 times smaller than the double-stage large presses with a high pressing rate normally used to prepare detergent tablets.
- the tablets of the invention comprise a silicone, preferably in the form of an emulsion.
- the addition of water to the tablet via the emulsion is beneficial because it can prevent the powder from granulating.
- the granulation of the powder to from granules greater than 10mm is undesirable because it causes difficulties when press feeding press feeding.
- the silicones used are preferably polymers or copolymers of dimethyl silicone or aminomethoxy functionalised dimethyl silicone. Mixtures of the polymers may also be used.
- the level of silicone emulsion added to the product is from 0.5 wt% to 10 wt% of the total product, preferably from 1 to 3 wt%.
- the level of silicone within the silicone emulsion is from 2 wt% to 50 wt% of the total weight of the silicone emulsion, preferably from 5 to 15 wt%.
- any emulsifier may be used, preferably the emulsifier is a surfactant, most preferred as the emulsifier a nonionic surfactant.
- silicone emulsions preferentially used in the present invention are those available from Dow Corning under the trademarks Dow Corning 36, 346, and 347; Dow Corning HV-490, Dow Corning 190 and 93; Dow Corning 531 and 536; Dow Corning 8075, 7242, Dow Corning 2-8707 and OSI-SAG 470,
- the bleaching systems of the invention are preferably oxygen based.
- the oxygen bleaching agents of the compositions include organic peroxy acids and diacylperoxides.
- Typical monoperoxy acids useful herein include alkyl peroxy acids and aryl peroxy acids such as:
- Typical diperoxy acids useful herein include alkyl diperoxy acids and aryl diperoxy acids, such as:
- a typical diacylperoxide useful herein includes dibenzoylperoxide.
- Inorganic peroxygen compounds are especially suitable for the present invention.
- these materials useful in the invention are salts of monopersulfate, perborate monohydrate, perborate tetrahydrate, and percarbonate. sodium perborate and sodium percarbonate are particularly preferred.
- the organic peroxy acid is present in the composition in an amount such that the level of organic peroxy acid in the wash solution is 1 ppm to 300 ppm AvOx, preferably 2 ppm to 200 ppm AvOx.
- Typical levels of organic peroxy acid are from 2 wt% to 15 wt% of the total formulation.
- the oxygen bleaching agent may be incorporated directly into the formulation or may be encapsulated by any number of encapsulation techniques.
- Suitable peracid precursors for peroxy bleach compounds have been amply described in the literature, including GB Nos. 836,988; 855,735; 907,356; 907;358; 907,950; 1,003,310 and 1,246,339; US-A-3,332,882 and US-A-4,128,494.
- Typical examples of precursors are polyacylated alkylene diamines, such as N,N,N',N'-tetraacetylethylene diamine (TAED) and N,N,N',N'-tetraacetylmethylene diamine (TAMD); acylated glycolurils, such as tetraacetylglycoluril (TAGU); triacetylcyanurate, sodium sulfophenyl ethyl carbonic acid ester, sodium acetyloxybenene sulfonate (SABS), sodium nonanoyloxy benzene sulfonate (SNOBS) and choline sulfophenyl carbonate.
- SABS sodium acetyloxybenene sulfonate
- SNOBS sodium nonanoyloxy benzene sulfonate
- choline sulfophenyl carbonate choline sulfophenyl carbonate
- Peroxybenzoic acid precursors are known in the art, e.g., as described in GB-A-836,988. Examples of suitable precursors are phenylbenzoate; phenyl p-nitrobenzoate; o-nitrophenyl benzoate; o-carboxyphenyl benzoate; p-bromophenylbenzoate; sodium or potassium benzoyloxy benzene-sulfonate; and benzoic anhydride.
- Preferred peroxygen bleach precursors are sodium p-benzoyloxybenzene sulfonate, N,N,N',N'-tetraacetylethylene diamine, sodium nonanoyloxybenzene sulfonate and choline sulfophenyl carbonate.
- the peroxygen bleach precursors may be present in the composition in an amount from 1 to 20 wt. %, preferably from 1 to 15 wt. %, most preferably from 2 to 10 weight %.
- a source of hydrogen peroxide is required.
- the hydrogen peroxide source is preferably a compound that delivers hydrogen peroxide on dissolution.
- Preferred sources of hydrogen peroxide are sodium perborate, either as mono- or tetrahydrate and sodium percarbonate.
- the source of hydrogen peroxide, when included in the composition, is present at a level from 1% to 30% by weight, preferably from 2% to 25% by weight, most preferably from 4% to 20% by weight.
- An effective amount of a bleach catalyst can also be present in the first layer.
- organic catalysts are available such as the sulfonimines as described in US-A-5,041,232; US-A-5,047,163 and US-A-5,463,115.
- Transition metal bleach catalysts are also useful especially those based on manganese, iron, cobalt, titanium, molybdenum, nickel, chromium, copper, ruthenium, tungsten and mixtures thereof. These include simple water-soluble salts such as those of iron, manganese and cobalt as well as catalysts containing complex ligands.
- Suitable examples of manganese catalysts containing organic ligands are described in US-A-4,728,455, US-A-5,114,606, US-A-5,153,161, US-A-5,194,416, US-A-5,227,084, US-A-5,244,594, US-A-5,246,612, US-A-5,246,621, US-A-5,256,779, US-A-5,274,147, US-A-5,280,117 and EP-A-544,440, EP-A-544,490, EP-A-549,271 and EP-A-549,272.
- Preferred examples of these catalysts include Mn IV 2 (u-O) 2 (1,4,7-trimethyl-1,4,7-triazacyclononane) 2 (PF 6 ) 2 , Mn III 2 (u-O) 1 (u-OAC) 2 (1,4,7-trimethyl-1,4,7-triazacyclononane) 2 (CIO 4 ) 2 , Mn IV 4 (u-O) 6 (1,4,7-triazacyclononane) 4 (CIO 4 ) 4 , Mn III Mn IV 4 (u-O) 1 (u-OAC) 2 (1,4,7-trimethyl-1,4,7triazacyclononane) 2 (ClO 4 ) 3 , Mn IV (1,4,7-trimethyl-1,4,7-triazacyclononane)-(OCH 3 ) 3 (PF 6 ), and mixtures thereof.
- metal-based bleach catalysts include those disclosed in US-A-4,430,243 and US-A-5,l14,611.
- Iron and manganese salts of aminocarboxylic acids in general are useful herein including iron and manganese aminocarboxylate salts disclosed for bleaching in the photographic color processing arts.
- a particularly useful transition metal salt is derived from ethylenediaminedisuccinate and any complex of this ligand with iron or manganese.
- Another type of bleach catalyst is a water soluble complex of manganese (II), (III), and/or (IV) with a ligand which is a non-carboxylate polyhydroxy compound having at least three consecutive C-OH groups.
- Preferred ligands include sorbitol, iditol, dulsitol, mannitol, xylithol, arabitol, adonitol, mesoerythritol, meso-inositol, lactose and mixtures thereof. Especially preferred is sorbitol.
- US-A-5,114,611 teaches a bleach catalyst comprising a complex of transition metals, including manganese, cobalt, iron or copper with an non-(macro)-cyclic ligand.
- transition metals including manganese, cobalt, iron or copper
- non-(macro)-cyclic ligand include Mn gluconate, Mn(CF 3 SO 3 ) 2 , and binuclear Mn complexed with tetra-N-dentate and bi-N-dentate ligands, including [bipy 2 Mn III (u-O) 2 Mn IV bipy 2 ]-(CIO 4 ) 3 .
- bleach catalysts are described, for example, in EP-A-408,131 (cobalt complexes), EP-A-384,503 and EP-A-306,089 (metallo-porphyrins), US-A-4,728,455 (manganese/multidenate ligand), US-A-4,711,748 (absorbed manganese on aluminosilicate), US-A-4,601,845 (aluminosilicate support with manganese, zinc or magnesium salt), US-A-4,626,373 (manganese/ligand), US-A-4,119,557 (ferric complex), US-A-4,430,243 (Chelants with manganese cations and non-catalytic metal cations), and US-A-4,728,455 (manganese gluconates).
- WO 96/23860 describe cobalt catalysts of the type [Co n L m X p ] Z Y z , where L is an organic ligand molecule containing more than one heteroatom selected from N, P, O and S; X is a co-ordinating species; n is preferably 1 or 2; m is preferably 1 to 5; p is preferably 0 to 4 and Y is a counterion.
- L is an organic ligand molecule containing more than one heteroatom selected from N, P, O and S
- X is a co-ordinating species
- n is preferably 1 or 2
- m is preferably 1 to 5
- p is preferably 0 to 4
- Y is a counterion.
- One example of such a catalyst is N,N'-Bis(salicylidene)ethylenediaminecobalt (II).
- transition-metal containing bleach catalysts can be prepared in the situ by the reaction of a transition-metal salt with suitable chelating agent, for example, a mixture of manganese sulfate and ethylenediaminedisuccinate.
- suitable chelating agent for example, a mixture of manganese sulfate and ethylenediaminedisuccinate.
- Highly colored transition metal-containing bleach catalysts may be co-processed with zeolites to reduce the color impact.
- the bleach catalyst is typically incorporated at a level of 0.0001 to 10% by wt., preferably 0.001 to 5% by weight.
- compositions of this invention can contain all manner of detergent builders commonly taught for use in machine dishwashing or other cleaning compositions.
- the builders can include any of the conventional inorganic and organic water-soluble builder salts, or mixtures thereof and comprise 5 to 90% by weight, preferably from 10 to 80% by weight of the cleaning composition.
- phosphorus-containing inorganic builders when present, include the water-soluble salts, especially alkali metal pyrophosphates, orthophosphates and polyphosphates.
- specific examples of inorganic phosphate builders include sodium and potassium tripolyphosphates, pyrophosphates and hexametaphosphates.
- non-phosphorus-containing inorganic builders when present, include water-soluble alkali metal carbonates, bicarbonates, sesquicarbonates, borates, silicates, including layered silicates such as SKS-6 ex. Hoechst, metasilicates, and crystalline and amorphous aluminosilicates. Specific examples include sodium carbonate (with or without calcite seeds), potassium carbonate, sodium and potassium bicarbonates, silicates including layered silicates and zeolites.
- Organic detergent builders can also be used in the present invention.
- organic builders include alkali metal citrates, succinates, malonates, fatty acid sulfonates, fatty acid carboxylates, nitrilotriacetates, phytates, phosphonates, alkanehydroxyphosphonates, oxydisuccinates, alkyl and alkenyl disuccinates, oxydiacetates, carboxymethyloxy succinates, ethylenediamine tetraacetates, tartrate monosuccinates, tartrate disuccinates, tartrate monoacetates, tartrate diacetates, oxidized starches, oxidized heteropolymeric polysaccharides, polyhydroxysulfonates, polycarboxylates such as polyacrylates, polymaleates, polyacetates, polyhydroxyacrylates, polyacrylate/polymaleate and polyacrylate/ polymethacrylate copolymers, acrylate/maleate/vinyl alcohol terpolymers, aminopol
- Alkali metal citrates, nitrilotriacetates, oxydisuccinates, polyphosphonates, acrylate/maleate copolymers and acrylate/maleate/vinyl alcohol terpolymers are especially preferred organic builders.
- the buffering system may be present in order to deliver a pH of about 6 to about 11 in the wash water.
- Materials which may be selected for the buffering system include water-soluble alkali metal carbonates, bicarbonates, sesquicarbonates, borates, silicates, layered silicates such as SKS-6 ex Hoechst, metasilicates, phytic acid, citric acid, borate and crystalline and amorphous aluminosilicates and mixtures thereof.
- Preferred examples include sodium and potassium carbonate, sodium and potassium bicarbonates, borates and silicates, including layered silicates
- a surfactant may be included in the tablet including anionic, nonionic, cationic, amphoteric, zwitteronic surfactants and mixtures of these surface active agents.
- anionic, nonionic, cationic, amphoteric, zwitteronic surfactants and mixtures of these surface active agents are well known in the detergent arts and are described at length at "Surface Active Agents and Detergents", Vol. 2 by Schwartz, Perry and Birch, Interscience Publishers, Inc., 1959, herein incorporated by reference.
- Preferred surfactants are one or a mixture of:
- Anionic synthetic detergents can be broadly described as surface active compounds with one or more negatively charged functional groups.
- An important class of anionic compounds are the water-soluble salts, particularly the alkali metal salts, of organic sulfur reaction products having in their molecular structure an alkyl radical containing from about 6 to 24 carbon atoms and a radical selected from the group consisting of sulfonic and sulfuric acid ester radicals.
- R 1 OSO 3 M where R 1 is a primary alkyl group of 8 to 18 carbon atoms and M is a solubilizing cation.
- the alkyl group R 1 may have a mixture of chain lengths. It is preferred that at least two thirds of the R 1 alkyl groups have a chain length of 8 to 14 carbon atoms. This will be the case if R 1 is coconut alkyl, for example.
- the solubilizing cation may be a range of cations which are in general monovalent and confer water solubility. Alkali metal, notably sodium, is especially envisaged. Other possibilities are ammonium and substituted ammonium ions, such as trialkanolammonium or trialkylammonium.
- R 2 CH (SO 3 M) CO 2 R 3 where R 2 is an alkyl group of 6 to 16 atoms, R 3 is an alkyl group of 1 to 4 carbon atoms and M is a solubilizing cation.
- the group R 2 may have a mixture of chain lengths. Preferably at least two thirds of these groups have 6 to 12 carbon atoms. This will be the case when the moiety R 2 CH(-)CO 2 (-) is derived from a coconut source, for instance. It is preferred that R 3 is a straight chain alkyl, notably methyl or ethyl.
- Alkyl Benzene Sulfonates R 4 ArSO 3 M where R 4 is an alkyl group of 8 to 18 carbon atoms, Ar is a benzene ring (C 6 H 4 ) and M is a solubilizing cation.
- the group R 4 may be a mixture of chain lengths. Straight chains of 11 to 14 carbon atoms are preferred.
- Paraffin sulfonates having 8 to 22 carbon atoms, preferably 12 to 16 carbon atoms, in the alkyl moiety. These surfactants are commercially available as Hostapur SAS from Hoechst Celanese.
- Olefin sulfonates having 8 to 22 carbon atoms, preferably 12 to 16 carbon atoms.
- U.S. Patent No. 3,332,880 contains a description of suitable olefin sulfonates.
- Organic phosphate based anionic surfactants include organic phosphate esters such as complex mono- or diester phosphates of hydroxyl- terminated alkoxide condensates, or salts thereof. Included in the organic phosphate esters are phosphate ester derivatives of polyoxyalkylated alkylaryl phosphate esters, of ethoxylated linear alcohols and ethoxylates of phenol. Also included are nonionic alkoxylates having a sodium alkylenecarboxylate moiety linked to a terminal hydroxyl group of the nonionic through an ether bond. Counterions to the salts of all the foregoing may be those of alkali metal, alkaline earth metal, ammonium, alkanolammonium and alkylammonium types.
- Particularly preferred anionic surfactants are the fatty acid ester sulfonates with formula: R 2 CH(SO 3 M)CO 2 R 3 where the moiety R 2 CH(-)CO 2 (-) is derived from a coconut source and R 3 is either methyl or ethyl; primary alkyl sulfates with the formula: R 1 OSO 3 M wherein R 1 is a primary alkyl group of 10 to 18 carbon atoms and M is a sodium cation; and paraffin sulfonates, preferably with 12 to 16 carbon atoms to the alkyl moiety.
- Nonionic surfactants can be broadly defined as surface active compounds with one or more uncharged hydrophilic substituents.
- a major class of nonionic surfactants are those compounds produced by the condensation of alkylene oxide groups with an organic hydrophobic material which may be aliphatic or alkyl aromatic in nature.
- the length of the hydrophilic or polyoxyalkylene radical which is condensed with any particular hydrophobic group can be readily adjusted to yield a water-soluble compound having the desired degree of balance between hydrophilic and hydrophobic elements.
- Illustrative, but not limiting examples, of various suitable nonionic surfactant types are:
- polyoxyalkene condensates of aliphatic carboxylic acids whether linear- or branched-chain and unsaturated or saturated, especially ethoxylated and/or propoxylated aliphatic acids containing from about 8 to about 18 carbon atoms in the aliphatic chain and incorporating from about 2 to about 50 ethylene oxide and/or propylene oxide units.
- Suitable carboxylic acids include "coconut” fatty acids (derived from coconut oil) which contain an average of about 12 carbon atoms, "tallow” fatty acids (derived from tallow-class fats) which contain an average of about 18 carbon atoms, palmitic acid, myristic acid, stearic acid and lauric acid,
- polyoxyalkene condensates of aliphatic alcohols whether linear- or branched-chain and unsaturated or saturated, especially ethoxylated and/or propoxylated aliphatic alcohols containing from about 6 to about 24 carbon atoms and incorporating from about 2 to about 50 ethylene oxide and/or propylene oxide units.
- Suitable alcohols include "coconut” fatty alcohol, "tallow” fatty alcohol, lauryl alcohol, myristyl alcohol and oleyl alcohol.
- Ethoxylated fatty alcohols may be used alone or in admixture with anionic surfactants, especially the preferred surfactants above.
- the average chain lengths of the alkyl group R 5 in the general formula: R 5 O(CH 2 CH 2 O) n H is from 6 to 20 carbon atoms.
- the group R 5 may have chain lengths in a range from 9 to 18 carbon atoms.
- the average value of n should be at least 2.
- the numbers of ethylene oxide residues may be a statistical distribution around the average value. However, as is known, the distribution can be affected by the manufacturing process or altered by fractionation after ethoxylation.
- Particularly preferred ethoxylated fatty alcohols have a group R 5 which has 9 to 18 carbon atoms while n is from 2 to 8.
- nonionic surfactants having a formula: wherein R 6 is a linear alkyl hydrocarbon radical having an average of 6 to 18 carbon atoms, R 7 and R 8 are each linear alkyl hydrocarbons of about 1 to about 4 carbon atoms, x is an integer of from 1 to 6, y is an integer of from 4 to 20 and z is an integer from 4 to 25.
- One preferred nonionic surfactant of the above formula is Poly-Tergent SLF-18 7 a registered trademark of the Olin Corporation, New Haven, Conn. having a composition of the above formula where R 6 is a C 6 -C 10 linear alkyl mixture, R 7 and R 8 are methyl, x averages 3, y averages 12 and z averages 16.
- R 9 is a linear, aliphatic hydrocarbon radical having from about 4 to about 18 carbon atoms including mixtures thereof; and R 10 is a linear, aliphatic hydrocarbon radical having from about 2 to about 26 carbon atoms including mixtures thereof; j is an integer having a value of from 1 to about 3; k is an integer having a value from 5 to about 30; and z is an integer having a value of from 1 to about 3. Most preferred are compositons in which j is 1, k is from about 10 to about 20 and l is 1. These surfactants are described in WO 94/22800. Other preferred nonionic surfactants are linear fatty alcohol alkoxylates with a capped terminal group, as described in U.S. 4,340,766 to BASF. Particularly preferred is Plurafac LF403 ex. BASF.
- R 11 is a C 6 -C 24 linear or branched alkyl hydrocarbon radical and q is a number from 2 to 50; more preferably R 11 is a C 8 -C 18 linear alkyl mixture and q is a number from 2 to 15.
- polyoxyethylene or polyoxypropylene condensates of alkyl phenols whether linear- or branched-chain and unsaturated or saturated, containing from about 6 to 12 carbon atoms and incorporating from about 2 to about 25 moles of ethylene oxide and/or propylene oxide.
- the preferred polyoxyethylene derivatives are of sorbitan monolaurate, sorbitan trilaurate, sorbitan monopalmitate, sorbitan tripalmitate, sorbitan monostearate, sorbitan monoisostearate, sorbitan tripalmitate, sorbital tristearate, sorbitan monooleate, and sorbitan trioleate.
- the polyoxyethylene chains may contain between about 4 and 30 ethylene oxide units, preferably about 10 to 20.
- the sorbitan ester derivatives contain 1, 2 or 3 polyoxyethylene chains dependent upon whether they are mono-, di- or tri-acid esters.
- polyoxyethylene-polyoxypropylene block copolymers having formula: HO(CH 2 CH 2 O) a (CH(CH 3 ) CH 2 O) b (CH 2 CH 2 O) c H or HO(CH(CH 3 )CH 2 O) d (CH 2 CH 2 O) e (CH(CH 3 )CH 2 O) f H wherein a, b, c, d, e and f are integers from 1 to 350 reflecting the respective polyethylene oxide and polypropylene oxide blocks of said polymer.
- the polyoxyethylene component of the block polymer constitutes at least about 10% of the block polymer.
- the material preferably has a molecular weight of between about 1,000 and 15,000, more preferably from about 1,500 to about 6,000. These materials are well-known in the art. They are available under the trademark "Pluronic” and "Pluronic R", a product of BASF Corporation.
- Amine oxides having formula: R 12 R 13 R 14 N O wherein R 12 , R 13 and R 14 are saturated aliphatic radicals or substituted saturated aliphatic radicals.
- Preferable amine oxides are those wherein R 12 is an alkyl chain of about 10 to about 20 carbon atoms and R 13 and R 14 are methyl or ethyl groups or both R 12 and R 13 are alkyl chains of about 6 to about 14 carbon atoms and R 14 is a methyl or ethyl group.
- Amphoteric synthetic detergents can be broadly described as derivatives of aliphatic tertiary amines, in which the aliphatic radical may be straight chain or branched and wherein one of the aliphatic substituents contain from about 8 to about 18 carbons and one contains an anionic water-solubilizing group, i.e., carboxy, sulpho, sulphato, phosphato or phosphono.
- an anionic water-solubilizing group i.e., carboxy, sulpho, sulphato, phosphato or phosphono.
- Examples of compounds falling within this definition are sodium 3-dodecylamino propionate and sodium 2-dodecylamino propane sulfonate.
- Zwitterionic synthetic detergents can be broadly described as derivatives of aliphatic quaternary ammonium, phosphonium and sulphonium compounds in which the aliphatic radical may be straight chained or branched, and wherein one of the aliphatic substituents contains from about 8 to about 18 carbon atoms and one contains an anionic water-solubilizing group, e.g., carboxy, sulpho, sulphato, phosphato or phosphono. These compounds are frequently referred to as betaines. Besides alkyl betaines, alkyl amino and alkyl amido betaines are encompassed within this invention.
- R 15 is a monovalent organic radical (e.g., a monovalent saturated aliphatic, unsaturated aliphatic or aromatic radical such as alkyl, hydroxyalkyl, alkenyl, hydroxyalkenyl, aryl, alkylaryl, hydroxyalkylaryl, arylalkyl, alkenylaryl, arylalkenyl, etc.) containing from about 6 to about 30 (preferably from about 8 to 18 and more preferably from about 9 to about 13) carbon atoms; R 16 is a divalent hydrocarbon radical containing from 2 to about 4 carbon atoms such as ethylene, propylene or butylene (most preferably the unit (R 16 O) n represents repeating units of ethylene oxide, propylene oxide and/or random or block combinations thereof); n is a number having an average value of from 0 to about 12; Z 1 represents a moiety derived from a monovalent organic radical (e.g., a monovalent saturated aliphatic, unsaturated alipha
- Examples of commercially available materials from Henkel Techandit GmbH Aktien of Dusseldorf, Germany include APG 7 300, 325 and 350 with R 15 being C 9 -C 11 , n is 0 and p is 1.3, 1.6 and 1.8-2.2 respectively; APG 7 500 and 550 with R 15 is C 12 -C 13 , n is 0 and p is 1.3 and 1.8-2.2, respectively; and APG 7 600 with R 15 being C 12 -C 14 , n is 0 and p is 1.3.
- esters of glucose are contemplated especially, it is envisaged that corresponding materials based on other reducing sugars, such as galactose and mannose are also suitable.
- nonionic surfactants are polyoxyethylene and polyoxypropylene condensates of linear aliphatic alcohols.
- the preferred range of surfactant is from 0.5 to 30 % by wt., more preferably from 0.5 to 15% by wt of the composition.
- compositions herein may also optionally contain one or more transition metal chelating agents.
- Such chelating agents can be selected from the group consisting of amino carboxylates, amino phosphonates, polyfunctionally-substituted aromatic chelating agents and mixtures therein. Without intending to be bound by theory, it is believed that the benefit of these materials is due in part to their exceptional ability to remove iron and manganese ions from washing solutions by formation of soluble chelates.
- Amino carboxylates useful as optional chelating agents include ethylenediaminetetraacetates, N-hydroxyethylethylenediaminetriacetates, nitrilotriacetates, ethylenediamine tetraproprionates, triethylenetetraamine-hexaacetates, diethylenetriaminepentaacetates, ethylenediamine disuccinate, and ethanoldiglycines, alkali metal, ammonium, and substituted ammonium salts therein and mixtures therein.
- Amino phosphonates are also suitable for use as chelating agents in the compositions of the invention when at least low levels of total phosphorus are permitted in detergent compositions, and include ethylenediaminetetrakis (methylenephosphonates), nitrilotris (methylenephosphonates) and diethylenetriaminepentakis (methylenephosphonates). Preferably, these amino phosphonates do not contain alkyl or alkenyl groups with more than about 6 carbon atoms.
- Polyfunctionally-substituted aromatic chelating agents are also useful in the compositions herein. See US-A-3,812,044.
- Preferred compounds of this type in acid form are dihydroxydisulfobenzenes such as 1,2-dihydroxy-3,5-disulfobenzene.
- these chelating agents will generally comprise from 0.1% to 10% by weight of the detergent compositions herein. More preferably, if utilized, the chelating agents will comprise from 0.1% to 5.0% by weight of such composition.
- Enzymes capable of facilitating the removal of soils from a substrate may also be present in an amount of up to 10% by wt., preferably 1 to 5 wt. %.
- Such enzymes include proteases (e.g., Alcalase7, Savinase7 and Esperase7 from Novo Industries A/S and Purafect OxP7, ex. Genencor), amylases (e.g., Termamyl7 and Duramyl7 from Novo Industries and Purafect OxAm7, ex. Genencor).
- Suitable binding and disintegrating agents for use in tablets are described in the literature (see, for example, "Pharmaceutical Dosage Forms : Volume 1", 1989, Marcel Dekker Inc., ISBN 0-8246-804402). Both natural and synthetic polymeric materials are suitable.
- starch such as corn, rice and potato starch and starch derivatives such as U-Sperse MTM and U-SperseTM provided by National Starch PrimojelTM, carboxymethyl starch and sodium glycolate such as ExplotabTM, pre-gelatinized corn starches such as NationalTM 1551 and Starch TM 1500; cellulose and cellulose derivatives including carboxymethyl cellulose such as CourloseTM and NymcelTM, reticulated sodium carboxymethyl cellulose, such as Ac-Di-SolTM provided by FMC Corporation, microcystalline cellulose fibres such as HanflocTM, microcrystalline cellulose such as LatticeTM NT provided by FMC Corporation and AvicelTM PH provided by FMC Corporation, methyl cellulose, ethyl cellulose, hydroxypropyl cellulose and hydroxyprorylmethyl cellulose.
- starch such as corn, rice and potato starch and starch derivatives such as U-Sperse MTM and U-SperseTM provided by National Starch PrimojelTM, carboxymethyl star
- binding/disintegrating agents are polyvinylpolypyrrolidone products such as PlasdoneTM, PVPTM K-30 and PVPTM K-60 all provided by International Specialty Products; polyvinylpolypyrrolidine products, a crosslinked product of N-vinyl-2-pyrrolidone such as PolyplasdoneTM XL provided by International Specialty Products; polymethacrylates, polyvinyl alcohol and polyethylene glycols. Gums such as acacia, tragacanth, guar, locust bean gum and pectin, gelatin, sucrose and alginates are also suitable as binding/disintegrating agents.
- Suitable inorganic materials include magnesium-aluminium silicate such as VeegumTM HV provided by R.T. Vanderbilt Co.Inc., bentonite and montmorillonite such as GelwhiteTM provided by Southern Clay Products.
- Other suitable binding agents include monoglycerids such as ImwitorTM 191 provided by Huls America Inc., glyceril sterates such as ImwitorTM 900 provided by Huls America Inc., and palmoil glycerides such as ImwitorTM 940 provided by Huls America Inc..
- the most preferred binding/disintegrating agents are microcrystalline cellulose and polyethylene glycols. The most preferred polyethylene glycols have a molecular weight of about 2000 to about 15000.
- lubricant agents for aiding the tablet's processing, such as stearates, waxes, hydrogenated vegetable oils and polyethylene glycols and charges such as sugars, sodium sulphate and sodium chloride.
- the tablet may be in the form of a layered product.
- the level of components may vary in each layer. In some instances it may be desirable to have no or low levels of bleaching ingredient with bleach sensitive ingredients such as perfume in one layer and the bleaching ingredient in a separate layer.
- the tablets of the invention completely dissolve in 6 litres of water(4oFH) at 65°C within 14 minutes.
- the total solid material within the tablet is preferably in the range from 0.005 to 1.0mm. in order to maximize the dissolution rate and increase the tablet's resistance.
- the addition order of the components of within the formulation can alter the appearance of the tablet.
- a tablet in the context of the present invention can be defined as any particulate material compacted to form a solid material, the tablet comprising at least one thousand particulates.
- the tablet has a diameter of 0.5 cm.
- the packaging system of the present invention comprises a body (3) shaped to receive a tablet and a cover(4) which keeps the tablet in the body.
- a cover which keeps the tablet in the body.
- the cover covering the tablet is metallic.
- the packaging system avoids the physical contact of a user with the detergent tablet when placing the tablet in the dish washing machine.
- Another important aspect of the present invention is the combined use of the detergent material of the present invention with a packaging of the type here illustrated in figure 1 and 2, as examples.
- a detergent tablet according to the present invention (Example 1) was prepared as follows.
- a dissolution test was performed in actual use conditions by placing a tablet in a plastic basket within a dish-washing machine Enxuta brand - Futura model, acquired in 1996. The selected cycle was set to a complete wash and the time taken for the tablet to completely dissolve was measured.
- the obtained values are average values of 3 washes for each product.
- the table given below demonstrates the tablet according to the invention dissolves completely within 14 minutes, while the comparative examples take longer than 14 minutes to completely dissolve.
- Examples A to E are commercially available tablets.
Landscapes
- Chemical & Material Sciences (AREA)
- Engineering & Computer Science (AREA)
- Life Sciences & Earth Sciences (AREA)
- Chemical Kinetics & Catalysis (AREA)
- Oil, Petroleum & Natural Gas (AREA)
- Wood Science & Technology (AREA)
- Organic Chemistry (AREA)
- Composite Materials (AREA)
- Mechanical Engineering (AREA)
- Inorganic Chemistry (AREA)
- Detergent Compositions (AREA)
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| BR9704788 | 1997-09-23 | ||
| BR9704788-0A BR9704788A (pt) | 1997-09-23 | 1997-09-23 | Processo para aumentar a dissolução de tabletes detergentes para máquinas de lavar louça,combinaçaõ de composição detergente com sistema de embalagem ,e,embalagem |
Publications (2)
| Publication Number | Publication Date |
|---|---|
| EP0903405A2 true EP0903405A2 (fr) | 1999-03-24 |
| EP0903405A3 EP0903405A3 (fr) | 1999-08-11 |
Family
ID=4068055
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| EP98307432A Withdrawn EP0903405A3 (fr) | 1997-09-23 | 1998-09-14 | Composition pour lave-vaisselle |
Country Status (7)
| Country | Link |
|---|---|
| EP (1) | EP0903405A3 (fr) |
| AR (1) | AR017131A1 (fr) |
| AU (1) | AU8522598A (fr) |
| BR (1) | BR9704788A (fr) |
| CA (1) | CA2247017A1 (fr) |
| NZ (1) | NZ331906A (fr) |
| ZA (1) | ZA988506B (fr) |
Cited By (28)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO1999063045A1 (fr) * | 1998-06-03 | 1999-12-09 | Henkel Kommanditgesellschaft Auf Aktien | Corps moules detergents stables a la rupture et se dissolvant rapidement |
| EP1041138A1 (fr) * | 1999-04-02 | 2000-10-04 | Unilever Plc | Composition de nettoyage à utilisation domestique |
| GB2355245A (en) * | 1999-10-12 | 2001-04-18 | Mcbride Robert Ltd | Detergent packaging system |
| WO2002051704A1 (fr) * | 2000-12-22 | 2002-07-04 | Henkel Kommanditgesellschaft Auf Aktien | Procede pour produire un conditionnement garni de pastilles et conditionnement correspondant |
| US6540081B2 (en) | 2001-09-06 | 2003-04-01 | Ecolab Inc. | Unit dose blister pack product dispenser |
| US6610643B1 (en) * | 1998-10-05 | 2003-08-26 | Henkel Kommanditgesellschaft Auf Aktien | Detergent tablet packaging for dishwashers |
| WO2003074647A1 (fr) * | 2002-03-06 | 2003-09-12 | Reckitt Benckiser N.V. | Ameliorations apportees a un contenant |
| US6673765B1 (en) | 1995-05-15 | 2004-01-06 | Ecolab Inc. | Method of making non-caustic solid cleaning compositions |
| US6962266B2 (en) | 2002-10-04 | 2005-11-08 | Ecolab Inc. | Method and apparatus for using a unit dose dispenser |
| WO2007007034A1 (fr) * | 2005-07-08 | 2007-01-18 | Reckitt Benckiser N.V. | Article de nettoyage et procédé de nettoyage |
| WO2012049053A1 (fr) * | 2010-10-14 | 2012-04-19 | Unilever Plc | Emballage comprenant une composition de lessive, distributeur d'emballage et procédé de lavage au moyen du distributeur et de l'emballage |
| US8329112B2 (en) | 2006-10-30 | 2012-12-11 | Reckitt Benckiser N.V. | Multi-dosing detergent delivery device |
| US8338357B2 (en) | 2006-01-21 | 2012-12-25 | Reckitt Benckiser N.V. | Multiple dosing ware washing article |
| US8375962B2 (en) | 2006-01-21 | 2013-02-19 | Reckitt Benckiser N. V. | Dosage element and chamber |
| US8815018B2 (en) | 2007-05-30 | 2014-08-26 | Reckitt Benckiser N.V. | Detergent dosing device |
| US9062281B2 (en) | 2010-10-14 | 2015-06-23 | Conopco, Inc. | Particulate detergent compositions comprising fluorescer |
| US9273271B2 (en) | 2010-10-14 | 2016-03-01 | Conopco Inc. | Laundry detergent particles |
| US9284517B2 (en) | 2010-10-14 | 2016-03-15 | Conopco Inc. | Laundry detergent particle |
| US9290724B2 (en) | 2010-10-14 | 2016-03-22 | Conopco, Inc. | Laundry detergent particles |
| US9290725B2 (en) | 2010-10-14 | 2016-03-22 | Conopco Inc. | Laundry detergent particles |
| US9290723B2 (en) | 2010-10-14 | 2016-03-22 | Conopco Inc. | Laundry detergent particles |
| US9365811B2 (en) | 2010-10-14 | 2016-06-14 | Conopco Inc. | Manufacture of coated particulate detergents |
| DE102015213199A1 (de) | 2015-07-14 | 2017-01-19 | Henkel Ag & Co. Kgaa | Blisterverpackung und Verfahren zur Entnahme eines Produkts aus der Blisterverpackung |
| DE102015218216A1 (de) | 2015-09-22 | 2017-03-23 | Henkel Ag & Co. Kgaa | Blisterverpackung und Verfahren zur Entnahme einer Tablette aus der Blisterverpackung |
| DE102015218214A1 (de) | 2015-09-22 | 2017-03-23 | Henkel Ag & Co. Kgaa | Blisterverpackung, Umverpackung für Blisterverpackung und Gebinde mit Umverpackung |
| DE102015218217A1 (de) | 2015-09-22 | 2017-03-23 | Henkel Ag & Co. Kgaa | Blisterverpackung mit asymmetrischer Tablette sowie asymmetrische Tablette |
| DE102016203354A1 (de) | 2016-03-01 | 2017-09-07 | Henkel Ag & Co. Kgaa | Blisterverpackung mit asymmetrischer Blisterhaube |
| EP4303290A1 (fr) * | 2022-07-05 | 2024-01-10 | Capsule Minimal Ltd. | Complexe de cartouche facilement soluble et systèmes industriels et procédés de fabrication associés |
Citations (14)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| GB1297087A (fr) * | 1969-10-15 | 1972-11-22 | ||
| FR2249949A1 (en) * | 1973-11-06 | 1975-05-30 | Saporta Francois De | Compacting detergent powder into pellets - packaged e.g. in continuous tapes, for accurate dosage in washing machine |
| EP0000076A1 (fr) * | 1977-06-01 | 1978-12-20 | THE PROCTER & GAMBLE COMPANY | Comprimé détergent |
| US4136045A (en) * | 1976-10-12 | 1979-01-23 | The Procter & Gamble Company | Detergent compositions containing ethoxylated nonionic surfactants and silicone containing suds suppressing agents |
| CH623537A5 (en) * | 1974-06-24 | 1981-06-15 | Sagapha Ag | Packaging for capsules divided into portions and having a pasty spot-removing means |
| DE3326459A1 (de) * | 1983-07-22 | 1985-01-31 | Etol-Werk GmbH & Co KG Chemische Fabrik, 7603 Oppenau | Verfahren zur herstellung eines geschirrspuelmittels und vorrichtung zur verarbeitung desselben |
| EP0225859A2 (fr) * | 1985-11-06 | 1987-06-16 | Ecolab Inc. | Distributeur de bloc solide chimique pour système de nettoyage |
| GB2200365A (en) * | 1987-01-26 | 1988-08-03 | Goodjet Ltd | Detergent composition |
| GB2210579A (en) * | 1987-10-02 | 1989-06-14 | Bristol Myers Co | Blister package and its manufacture |
| EP0391087A1 (fr) * | 1989-04-07 | 1990-10-10 | Freytag von Loringhoven, Andreas | Procédé et moyens pour l'addition de parfum à l'eau de lavage ou de rinçage |
| WO1991015568A1 (fr) * | 1990-04-02 | 1991-10-17 | Henkel Kommanditgesellschaft Auf Aktien | Comprimes detergents stables, bifonctionnels et sans phosphate pour lave-vaisselle |
| WO1996019561A1 (fr) * | 1994-12-22 | 1996-06-27 | The Procter & Gamble Company | Compositions a base de silicone |
| WO1997000311A1 (fr) * | 1995-06-16 | 1997-01-03 | The Procter & Gamble Company | Compositions d'agents de blanchiment comprenant des catalyseurs au cobalt |
| WO1997031096A1 (fr) * | 1996-02-23 | 1997-08-28 | Henkel Kommanditgesellschaft Auf Aktien | Procede de fabrication de pastilles d'agent detergent pour lave-vaisselle |
-
1997
- 1997-09-23 BR BR9704788-0A patent/BR9704788A/pt not_active Application Discontinuation
-
1998
- 1998-09-14 CA CA002247017A patent/CA2247017A1/fr not_active Abandoned
- 1998-09-14 EP EP98307432A patent/EP0903405A3/fr not_active Withdrawn
- 1998-09-15 NZ NZ331906A patent/NZ331906A/en unknown
- 1998-09-17 AU AU85225/98A patent/AU8522598A/en not_active Abandoned
- 1998-09-17 ZA ZA9808506A patent/ZA988506B/xx unknown
- 1998-09-22 AR ARP980104722A patent/AR017131A1/es not_active Application Discontinuation
Patent Citations (14)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| GB1297087A (fr) * | 1969-10-15 | 1972-11-22 | ||
| FR2249949A1 (en) * | 1973-11-06 | 1975-05-30 | Saporta Francois De | Compacting detergent powder into pellets - packaged e.g. in continuous tapes, for accurate dosage in washing machine |
| CH623537A5 (en) * | 1974-06-24 | 1981-06-15 | Sagapha Ag | Packaging for capsules divided into portions and having a pasty spot-removing means |
| US4136045A (en) * | 1976-10-12 | 1979-01-23 | The Procter & Gamble Company | Detergent compositions containing ethoxylated nonionic surfactants and silicone containing suds suppressing agents |
| EP0000076A1 (fr) * | 1977-06-01 | 1978-12-20 | THE PROCTER & GAMBLE COMPANY | Comprimé détergent |
| DE3326459A1 (de) * | 1983-07-22 | 1985-01-31 | Etol-Werk GmbH & Co KG Chemische Fabrik, 7603 Oppenau | Verfahren zur herstellung eines geschirrspuelmittels und vorrichtung zur verarbeitung desselben |
| EP0225859A2 (fr) * | 1985-11-06 | 1987-06-16 | Ecolab Inc. | Distributeur de bloc solide chimique pour système de nettoyage |
| GB2200365A (en) * | 1987-01-26 | 1988-08-03 | Goodjet Ltd | Detergent composition |
| GB2210579A (en) * | 1987-10-02 | 1989-06-14 | Bristol Myers Co | Blister package and its manufacture |
| EP0391087A1 (fr) * | 1989-04-07 | 1990-10-10 | Freytag von Loringhoven, Andreas | Procédé et moyens pour l'addition de parfum à l'eau de lavage ou de rinçage |
| WO1991015568A1 (fr) * | 1990-04-02 | 1991-10-17 | Henkel Kommanditgesellschaft Auf Aktien | Comprimes detergents stables, bifonctionnels et sans phosphate pour lave-vaisselle |
| WO1996019561A1 (fr) * | 1994-12-22 | 1996-06-27 | The Procter & Gamble Company | Compositions a base de silicone |
| WO1997000311A1 (fr) * | 1995-06-16 | 1997-01-03 | The Procter & Gamble Company | Compositions d'agents de blanchiment comprenant des catalyseurs au cobalt |
| WO1997031096A1 (fr) * | 1996-02-23 | 1997-08-28 | Henkel Kommanditgesellschaft Auf Aktien | Procede de fabrication de pastilles d'agent detergent pour lave-vaisselle |
Non-Patent Citations (1)
| Title |
|---|
| 0'LENICK A J ET AL: "SILANOL ESTERS: A NEW SERIES OF SILICONE COMPOUNDS" SOAP COSMETICS CHEMICAL SPECIALTIES, vol. 70, no. 8, 1 August 1994, pages 50-52, 54, XP000543178 * |
Cited By (35)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US6673765B1 (en) | 1995-05-15 | 2004-01-06 | Ecolab Inc. | Method of making non-caustic solid cleaning compositions |
| WO1999063045A1 (fr) * | 1998-06-03 | 1999-12-09 | Henkel Kommanditgesellschaft Auf Aktien | Corps moules detergents stables a la rupture et se dissolvant rapidement |
| US6610643B1 (en) * | 1998-10-05 | 2003-08-26 | Henkel Kommanditgesellschaft Auf Aktien | Detergent tablet packaging for dishwashers |
| EP1041138A1 (fr) * | 1999-04-02 | 2000-10-04 | Unilever Plc | Composition de nettoyage à utilisation domestique |
| GB2355245A (en) * | 1999-10-12 | 2001-04-18 | Mcbride Robert Ltd | Detergent packaging system |
| WO2002051704A1 (fr) * | 2000-12-22 | 2002-07-04 | Henkel Kommanditgesellschaft Auf Aktien | Procede pour produire un conditionnement garni de pastilles et conditionnement correspondant |
| US6540081B2 (en) | 2001-09-06 | 2003-04-01 | Ecolab Inc. | Unit dose blister pack product dispenser |
| US7407925B2 (en) | 2002-03-06 | 2008-08-05 | Reckitt Benckiser N.V. | Packaged detergent composition or additive and method for packaging the same |
| WO2003074647A1 (fr) * | 2002-03-06 | 2003-09-12 | Reckitt Benckiser N.V. | Ameliorations apportees a un contenant |
| US6962266B2 (en) | 2002-10-04 | 2005-11-08 | Ecolab Inc. | Method and apparatus for using a unit dose dispenser |
| GB2428247A (en) * | 2005-07-08 | 2007-01-24 | Reckitt Benckiser Nv | Reduced mass detergent tablet and packaging |
| WO2007007034A1 (fr) * | 2005-07-08 | 2007-01-18 | Reckitt Benckiser N.V. | Article de nettoyage et procédé de nettoyage |
| US8338357B2 (en) | 2006-01-21 | 2012-12-25 | Reckitt Benckiser N.V. | Multiple dosing ware washing article |
| US8375962B2 (en) | 2006-01-21 | 2013-02-19 | Reckitt Benckiser N. V. | Dosage element and chamber |
| US8329112B2 (en) | 2006-10-30 | 2012-12-11 | Reckitt Benckiser N.V. | Multi-dosing detergent delivery device |
| US8815018B2 (en) | 2007-05-30 | 2014-08-26 | Reckitt Benckiser N.V. | Detergent dosing device |
| CN103153813A (zh) * | 2010-10-14 | 2013-06-12 | 荷兰联合利华有限公司 | 包含洗衣组合物的包装、所述包装的分配器和利用所述分配器和所述包装进行洗涤的方法 |
| US9365811B2 (en) | 2010-10-14 | 2016-06-14 | Conopco Inc. | Manufacture of coated particulate detergents |
| US9062281B2 (en) | 2010-10-14 | 2015-06-23 | Conopco, Inc. | Particulate detergent compositions comprising fluorescer |
| CN103153813B (zh) * | 2010-10-14 | 2015-08-19 | 荷兰联合利华有限公司 | 包含洗衣组合物的包装、所述包装的分配器和利用所述分配器和所述包装进行洗涤的方法 |
| US9273271B2 (en) | 2010-10-14 | 2016-03-01 | Conopco Inc. | Laundry detergent particles |
| US9284517B2 (en) | 2010-10-14 | 2016-03-15 | Conopco Inc. | Laundry detergent particle |
| US9290724B2 (en) | 2010-10-14 | 2016-03-22 | Conopco, Inc. | Laundry detergent particles |
| US9290725B2 (en) | 2010-10-14 | 2016-03-22 | Conopco Inc. | Laundry detergent particles |
| US9290723B2 (en) | 2010-10-14 | 2016-03-22 | Conopco Inc. | Laundry detergent particles |
| WO2012049053A1 (fr) * | 2010-10-14 | 2012-04-19 | Unilever Plc | Emballage comprenant une composition de lessive, distributeur d'emballage et procédé de lavage au moyen du distributeur et de l'emballage |
| DE102015213199A1 (de) | 2015-07-14 | 2017-01-19 | Henkel Ag & Co. Kgaa | Blisterverpackung und Verfahren zur Entnahme eines Produkts aus der Blisterverpackung |
| WO2017009422A1 (fr) | 2015-07-14 | 2017-01-19 | Henkel Ag & Co. Kgaa | Emballage coque et procédé pour sortir un produit de l'emballage coque |
| DE102015218216A1 (de) | 2015-09-22 | 2017-03-23 | Henkel Ag & Co. Kgaa | Blisterverpackung und Verfahren zur Entnahme einer Tablette aus der Blisterverpackung |
| DE102015218214A1 (de) | 2015-09-22 | 2017-03-23 | Henkel Ag & Co. Kgaa | Blisterverpackung, Umverpackung für Blisterverpackung und Gebinde mit Umverpackung |
| DE102015218217A1 (de) | 2015-09-22 | 2017-03-23 | Henkel Ag & Co. Kgaa | Blisterverpackung mit asymmetrischer Tablette sowie asymmetrische Tablette |
| WO2017050538A1 (fr) * | 2015-09-22 | 2017-03-30 | Henkel Ag & Co. Kgaa | Emballage transparent renfermant une tablette asymétrique et tablette asymétrique |
| DE102016203354A1 (de) | 2016-03-01 | 2017-09-07 | Henkel Ag & Co. Kgaa | Blisterverpackung mit asymmetrischer Blisterhaube |
| WO2017148698A1 (fr) | 2016-03-01 | 2017-09-08 | Henkel Ag & Co. Kgaa | Emballage coque à coque asymétrique |
| EP4303290A1 (fr) * | 2022-07-05 | 2024-01-10 | Capsule Minimal Ltd. | Complexe de cartouche facilement soluble et systèmes industriels et procédés de fabrication associés |
Also Published As
| Publication number | Publication date |
|---|---|
| ZA988506B (en) | 2000-03-22 |
| NZ331906A (en) | 1999-10-28 |
| AR017131A1 (es) | 2001-08-22 |
| AU8522598A (en) | 1999-04-15 |
| BR9704788A (pt) | 1999-09-08 |
| CA2247017A1 (fr) | 1999-03-23 |
| EP0903405A3 (fr) | 1999-08-11 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| EP0903405A2 (fr) | Composition pour lave-vaisselle | |
| US5783540A (en) | Machine dishwashing tablets delivering a rinse aid benefit | |
| US5900395A (en) | Machine dishwashing tablets containing an oxygen bleach system | |
| EP0893491B1 (fr) | Compositions détergentes pour lave-vaisselle automatiques | |
| US5837663A (en) | Machine dishwashing tablets containing a peracid | |
| EP0998548B1 (fr) | Compositions pour lave-vaisselle renfermant des polymeres cationiques ou amphoteres solubles dans l'eau | |
| US20040058846A1 (en) | Copolymers that prevent glass from corroding | |
| US5705465A (en) | Anti-foam system for automatic dishwashing compositions | |
| US5480576A (en) | 1,3-N azole containing detergent compositions | |
| US20100144576A1 (en) | Dish detergent | |
| JP3171856B2 (ja) | 無水グリシンアクチベーターを含有するブリーチ添加剤組成物および漂白組成物 | |
| EP0846757B1 (fr) | Composition gélifiée pour lave-vaisselle | |
| US6345633B1 (en) | Automatic dishwashing compositions containing water soluble cationic surfactants | |
| AU729402B2 (en) | An anti-foam system based on hydrocarbon polymers and hydrophobic particulate solids | |
| AU9258498A (en) | Machine dishwashing compositions and rinse aid compositions | |
| US6334452B1 (en) | Automatic dishwashing compositions containing water soluble cationic surfactants | |
| WO1994019445A1 (fr) | Composition de lavage pour lave-vaisselle | |
| GB2367830A (en) | Process for preparing tablets |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| PUAI | Public reference made under article 153(3) epc to a published international application that has entered the european phase |
Free format text: ORIGINAL CODE: 0009012 |
|
| AK | Designated contracting states |
Kind code of ref document: A2 Designated state(s): DE ES FR GB IT |
|
| AX | Request for extension of the european patent |
Free format text: AL;LT;LV;MK;RO;SI |
|
| PUAL | Search report despatched |
Free format text: ORIGINAL CODE: 0009013 |
|
| AK | Designated contracting states |
Kind code of ref document: A3 Designated state(s): AT BE CH CY DE DK ES FI FR GB GR IE IT LI LU MC NL PT SE |
|
| AX | Request for extension of the european patent |
Free format text: AL;LT;LV;MK;RO;SI |
|
| 17P | Request for examination filed |
Effective date: 19990906 |
|
| RAP1 | Party data changed (applicant data changed or rights of an application transferred) |
Owner name: UNILEVER N.V. Owner name: UNILEVER PLC |
|
| AKX | Designation fees paid |
Free format text: DE ES FR GB IT |
|
| 17Q | First examination report despatched |
Effective date: 20020712 |
|
| STAA | Information on the status of an ep patent application or granted ep patent |
Free format text: STATUS: THE APPLICATION IS DEEMED TO BE WITHDRAWN |
|
| 18D | Application deemed to be withdrawn |
Effective date: 20030805 |