EP0309432A2 - Reinigungsverfahren und Gerät dafür - Google Patents
Reinigungsverfahren und Gerät dafürInfo
- Publication number
- EP0309432A2 EP0309432A2 EP88850310A EP88850310A EP0309432A2 EP 0309432 A2 EP0309432 A2 EP 0309432A2 EP 88850310 A EP88850310 A EP 88850310A EP 88850310 A EP88850310 A EP 88850310A EP 0309432 A2 EP0309432 A2 EP 0309432A2
- Authority
- EP
- European Patent Office
- Prior art keywords
- tank
- washing liquid
- cleaning
- liquid tank
- tenside
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Granted
Links
- 238000000034 method Methods 0.000 title claims abstract description 66
- 238000004140 cleaning Methods 0.000 title claims abstract description 41
- 239000000126 substance Substances 0.000 claims abstract description 30
- 238000006065 biodegradation reaction Methods 0.000 claims abstract description 23
- 235000015097 nutrients Nutrition 0.000 claims abstract description 18
- 244000005700 microbiome Species 0.000 claims abstract description 14
- 230000004936 stimulating effect Effects 0.000 claims abstract 2
- 239000007788 liquid Substances 0.000 claims description 58
- 238000005406 washing Methods 0.000 claims description 42
- 241000894006 Bacteria Species 0.000 claims description 34
- 239000000356 contaminant Substances 0.000 claims description 19
- 239000010802 sludge Substances 0.000 claims description 11
- 239000003513 alkali Substances 0.000 claims description 8
- QVGXLLKOCUKJST-UHFFFAOYSA-N atomic oxygen Chemical compound [O] QVGXLLKOCUKJST-UHFFFAOYSA-N 0.000 claims description 5
- 239000013505 freshwater Substances 0.000 claims description 5
- 238000010438 heat treatment Methods 0.000 claims description 5
- 229910052760 oxygen Inorganic materials 0.000 claims description 5
- 239000001301 oxygen Substances 0.000 claims description 5
- 239000000654 additive Substances 0.000 claims description 2
- 238000005238 degreasing Methods 0.000 abstract description 22
- 229930195733 hydrocarbon Natural products 0.000 abstract description 5
- 150000002430 hydrocarbons Chemical class 0.000 abstract description 5
- 239000004094 surface-active agent Substances 0.000 description 44
- 239000000243 solution Substances 0.000 description 31
- 239000003921 oil Substances 0.000 description 29
- HEMHJVSKTPXQMS-UHFFFAOYSA-M Sodium hydroxide Chemical compound [OH-].[Na+] HEMHJVSKTPXQMS-UHFFFAOYSA-M 0.000 description 28
- -1 greases Substances 0.000 description 15
- 235000011121 sodium hydroxide Nutrition 0.000 description 10
- 239000000203 mixture Substances 0.000 description 9
- 239000002253 acid Substances 0.000 description 7
- 150000001412 amines Chemical class 0.000 description 7
- XLYOFNOQVPJJNP-UHFFFAOYSA-N water Substances O XLYOFNOQVPJJNP-UHFFFAOYSA-N 0.000 description 7
- KWYUFKZDYYNOTN-UHFFFAOYSA-M Potassium hydroxide Chemical compound [OH-].[K+] KWYUFKZDYYNOTN-UHFFFAOYSA-M 0.000 description 6
- 235000014113 dietary fatty acids Nutrition 0.000 description 6
- 239000000194 fatty acid Substances 0.000 description 6
- 229930195729 fatty acid Natural products 0.000 description 6
- 150000003839 salts Chemical class 0.000 description 6
- 239000004115 Sodium Silicate Substances 0.000 description 5
- 125000000217 alkyl group Chemical group 0.000 description 5
- 125000004432 carbon atom Chemical group C* 0.000 description 5
- 125000002091 cationic group Chemical group 0.000 description 5
- 230000000694 effects Effects 0.000 description 5
- NTHWMYGWWRZVTN-UHFFFAOYSA-N sodium silicate Chemical compound [Na+].[Na+].[O-][Si]([O-])=O NTHWMYGWWRZVTN-UHFFFAOYSA-N 0.000 description 5
- 229910052911 sodium silicate Inorganic materials 0.000 description 5
- PXHVJJICTQNCMI-UHFFFAOYSA-N Nickel Chemical compound [Ni] PXHVJJICTQNCMI-UHFFFAOYSA-N 0.000 description 4
- 229910052799 carbon Inorganic materials 0.000 description 4
- 239000004519 grease Substances 0.000 description 4
- 239000008237 rinsing water Substances 0.000 description 4
- 235000019795 sodium metasilicate Nutrition 0.000 description 4
- OKTJSMMVPCPJKN-UHFFFAOYSA-N Carbon Chemical compound [C] OKTJSMMVPCPJKN-UHFFFAOYSA-N 0.000 description 3
- IAYPIBMASNFSPL-UHFFFAOYSA-N Ethylene oxide Chemical class C1CO1 IAYPIBMASNFSPL-UHFFFAOYSA-N 0.000 description 3
- 229910019142 PO4 Inorganic materials 0.000 description 3
- QAOWNCQODCNURD-UHFFFAOYSA-N Sulfuric acid Chemical class OS(O)(=O)=O QAOWNCQODCNURD-UHFFFAOYSA-N 0.000 description 3
- 229940045714 alkyl sulfonate alkylating agent Drugs 0.000 description 3
- 150000008052 alkyl sulfonates Chemical class 0.000 description 3
- 239000003637 basic solution Substances 0.000 description 3
- 230000001804 emulsifying effect Effects 0.000 description 3
- 229910052751 metal Inorganic materials 0.000 description 3
- 239000002184 metal Substances 0.000 description 3
- 150000002739 metals Chemical class 0.000 description 3
- 235000021317 phosphate Nutrition 0.000 description 3
- 229920001223 polyethylene glycol Polymers 0.000 description 3
- JCBPETKZIGVZRE-UHFFFAOYSA-N 2-aminobutan-1-ol Chemical compound CCC(N)CO JCBPETKZIGVZRE-UHFFFAOYSA-N 0.000 description 2
- 241000589291 Acinetobacter Species 0.000 description 2
- NLXLAEXVIDQMFP-UHFFFAOYSA-N Ammonia chloride Chemical compound [NH4+].[Cl-] NLXLAEXVIDQMFP-UHFFFAOYSA-N 0.000 description 2
- 241000186063 Arthrobacter Species 0.000 description 2
- 241000186146 Brevibacterium Species 0.000 description 2
- CURLTUGMZLYLDI-UHFFFAOYSA-N Carbon dioxide Chemical compound O=C=O CURLTUGMZLYLDI-UHFFFAOYSA-N 0.000 description 2
- VYZAMTAEIAYCRO-UHFFFAOYSA-N Chromium Chemical compound [Cr] VYZAMTAEIAYCRO-UHFFFAOYSA-N 0.000 description 2
- 241000588921 Enterobacteriaceae Species 0.000 description 2
- WQZGKKKJIJFFOK-GASJEMHNSA-N Glucose Natural products OC[C@H]1OC(O)[C@H](O)[C@@H](O)[C@@H]1O WQZGKKKJIJFFOK-GASJEMHNSA-N 0.000 description 2
- KFZMGEQAYNKOFK-UHFFFAOYSA-N Isopropanol Chemical compound CC(C)O KFZMGEQAYNKOFK-UHFFFAOYSA-N 0.000 description 2
- TWRXJAOTZQYOKJ-UHFFFAOYSA-L Magnesium chloride Chemical compound [Mg+2].[Cl-].[Cl-] TWRXJAOTZQYOKJ-UHFFFAOYSA-L 0.000 description 2
- 241000187654 Nocardia Species 0.000 description 2
- 229920000388 Polyphosphate Polymers 0.000 description 2
- 241000589516 Pseudomonas Species 0.000 description 2
- 241000168225 Pseudomonas alcaligenes Species 0.000 description 2
- 241000589630 Pseudomonas pseudoalcaligenes Species 0.000 description 2
- 241000605118 Thiobacillus Species 0.000 description 2
- 241000589634 Xanthomonas Species 0.000 description 2
- 150000001335 aliphatic alkanes Chemical class 0.000 description 2
- 150000004996 alkyl benzenes Chemical class 0.000 description 2
- 125000002947 alkylene group Chemical group 0.000 description 2
- 125000000129 anionic group Chemical group 0.000 description 2
- 125000003118 aryl group Chemical group 0.000 description 2
- 150000001735 carboxylic acids Chemical class 0.000 description 2
- 230000015556 catabolic process Effects 0.000 description 2
- 238000006243 chemical reaction Methods 0.000 description 2
- 229910052804 chromium Inorganic materials 0.000 description 2
- 239000011651 chromium Substances 0.000 description 2
- 230000006835 compression Effects 0.000 description 2
- 238000007906 compression Methods 0.000 description 2
- 229910052802 copper Inorganic materials 0.000 description 2
- 239000010949 copper Substances 0.000 description 2
- 238000000354 decomposition reaction Methods 0.000 description 2
- 238000006731 degradation reaction Methods 0.000 description 2
- 239000003599 detergent Substances 0.000 description 2
- 150000002148 esters Chemical class 0.000 description 2
- 150000004665 fatty acids Chemical class 0.000 description 2
- 239000008103 glucose Substances 0.000 description 2
- 238000002347 injection Methods 0.000 description 2
- 239000007924 injection Substances 0.000 description 2
- 239000000463 material Substances 0.000 description 2
- 230000002906 microbiologic effect Effects 0.000 description 2
- 229910052750 molybdenum Inorganic materials 0.000 description 2
- 229910052759 nickel Inorganic materials 0.000 description 2
- 230000001590 oxidative effect Effects 0.000 description 2
- 239000001205 polyphosphate Substances 0.000 description 2
- 235000011176 polyphosphates Nutrition 0.000 description 2
- 229910052700 potassium Inorganic materials 0.000 description 2
- 238000000926 separation method Methods 0.000 description 2
- RYCLIXPGLDDLTM-UHFFFAOYSA-J tetrapotassium;phosphonato phosphate Chemical compound [K+].[K+].[K+].[K+].[O-]P([O-])(=O)OP([O-])([O-])=O RYCLIXPGLDDLTM-UHFFFAOYSA-J 0.000 description 2
- 239000001993 wax Substances 0.000 description 2
- HXKKHQJGJAFBHI-UHFFFAOYSA-N 1-aminopropan-2-ol Chemical compound CC(O)CN HXKKHQJGJAFBHI-UHFFFAOYSA-N 0.000 description 1
- IOAOAKDONABGPZ-UHFFFAOYSA-N 2-amino-2-ethylpropane-1,3-diol Chemical compound CCC(N)(CO)CO IOAOAKDONABGPZ-UHFFFAOYSA-N 0.000 description 1
- UXFQFBNBSPQBJW-UHFFFAOYSA-N 2-amino-2-methylpropane-1,3-diol Chemical compound OCC(N)(C)CO UXFQFBNBSPQBJW-UHFFFAOYSA-N 0.000 description 1
- 241000605222 Acidithiobacillus ferrooxidans Species 0.000 description 1
- 241000605272 Acidithiobacillus thiooxidans Species 0.000 description 1
- 241000588624 Acinetobacter calcoaceticus Species 0.000 description 1
- 241000193792 Aerococcus viridans Species 0.000 description 1
- 241000607534 Aeromonas Species 0.000 description 1
- 241000193830 Bacillus <bacterium> Species 0.000 description 1
- 239000004215 Carbon black (E152) Substances 0.000 description 1
- ZAMOUSCENKQFHK-UHFFFAOYSA-N Chlorine atom Chemical compound [Cl] ZAMOUSCENKQFHK-UHFFFAOYSA-N 0.000 description 1
- 241000588919 Citrobacter freundii Species 0.000 description 1
- 241000588917 Citrobacter koseri Species 0.000 description 1
- RYGMFSIKBFXOCR-UHFFFAOYSA-N Copper Chemical compound [Cu] RYGMFSIKBFXOCR-UHFFFAOYSA-N 0.000 description 1
- 241000186216 Corynebacterium Species 0.000 description 1
- RWSOTUBLDIXVET-UHFFFAOYSA-N Dihydrogen sulfide Chemical class S RWSOTUBLDIXVET-UHFFFAOYSA-N 0.000 description 1
- 241000588724 Escherichia coli Species 0.000 description 1
- VGGSQFUCUMXWEO-UHFFFAOYSA-N Ethene Chemical compound C=C VGGSQFUCUMXWEO-UHFFFAOYSA-N 0.000 description 1
- 239000005977 Ethylene Substances 0.000 description 1
- 241000588749 Klebsiella oxytoca Species 0.000 description 1
- 241000588747 Klebsiella pneumoniae Species 0.000 description 1
- BAVYZALUXZFZLV-UHFFFAOYSA-N Methylamine Chemical compound NC BAVYZALUXZFZLV-UHFFFAOYSA-N 0.000 description 1
- ZOKXTWBITQBERF-UHFFFAOYSA-N Molybdenum Chemical compound [Mo] ZOKXTWBITQBERF-UHFFFAOYSA-N 0.000 description 1
- 241000588772 Morganella morganii Species 0.000 description 1
- 241000589588 Myroides odoratus Species 0.000 description 1
- 241000588912 Pantoea agglomerans Species 0.000 description 1
- ZLMJMSJWJFRBEC-UHFFFAOYSA-N Potassium Chemical compound [K] ZLMJMSJWJFRBEC-UHFFFAOYSA-N 0.000 description 1
- 241000588767 Proteus vulgaris Species 0.000 description 1
- 241000589540 Pseudomonas fluorescens Species 0.000 description 1
- 241000589776 Pseudomonas putida Species 0.000 description 1
- 241000589614 Pseudomonas stutzeri Species 0.000 description 1
- 241000863432 Shewanella putrefaciens Species 0.000 description 1
- QAOWNCQODCNURD-UHFFFAOYSA-L Sulfate Chemical compound [O-]S([O-])(=O)=O QAOWNCQODCNURD-UHFFFAOYSA-L 0.000 description 1
- ULUAUXLGCMPNKK-UHFFFAOYSA-N Sulfobutanedioic acid Chemical class OC(=O)CC(C(O)=O)S(O)(=O)=O ULUAUXLGCMPNKK-UHFFFAOYSA-N 0.000 description 1
- GSEJCLTVZPLZKY-UHFFFAOYSA-N Triethanolamine Chemical compound OCCN(CCO)CCO GSEJCLTVZPLZKY-UHFFFAOYSA-N 0.000 description 1
- 239000007983 Tris buffer Substances 0.000 description 1
- HCHKCACWOHOZIP-UHFFFAOYSA-N Zinc Chemical compound [Zn] HCHKCACWOHOZIP-UHFFFAOYSA-N 0.000 description 1
- 230000004913 activation Effects 0.000 description 1
- 125000002252 acyl group Chemical group 0.000 description 1
- 238000005273 aeration Methods 0.000 description 1
- 238000013019 agitation Methods 0.000 description 1
- 229910052783 alkali metal Inorganic materials 0.000 description 1
- 150000001340 alkali metals Chemical class 0.000 description 1
- 150000001447 alkali salts Chemical class 0.000 description 1
- 150000008055 alkyl aryl sulfonates Chemical class 0.000 description 1
- 150000008051 alkyl sulfates Chemical class 0.000 description 1
- 229910000147 aluminium phosphate Inorganic materials 0.000 description 1
- 125000003368 amide group Chemical group 0.000 description 1
- 150000001408 amides Chemical class 0.000 description 1
- 235000019270 ammonium chloride Nutrition 0.000 description 1
- 150000003863 ammonium salts Chemical class 0.000 description 1
- 239000007864 aqueous solution Substances 0.000 description 1
- IPTLKMXBROVJJF-UHFFFAOYSA-N azanium;methyl sulfate Chemical compound N.COS(O)(=O)=O IPTLKMXBROVJJF-UHFFFAOYSA-N 0.000 description 1
- 230000008953 bacterial degradation Effects 0.000 description 1
- 230000000721 bacterilogical effect Effects 0.000 description 1
- 239000001569 carbon dioxide Substances 0.000 description 1
- 229910002092 carbon dioxide Inorganic materials 0.000 description 1
- 239000003518 caustics Substances 0.000 description 1
- 239000003795 chemical substances by application Substances 0.000 description 1
- 239000000460 chlorine Substances 0.000 description 1
- 229910052801 chlorine Inorganic materials 0.000 description 1
- 238000000576 coating method Methods 0.000 description 1
- 229910017052 cobalt Inorganic materials 0.000 description 1
- 239000010941 cobalt Substances 0.000 description 1
- GUTLYIVDDKVIGB-UHFFFAOYSA-N cobalt atom Chemical compound [Co] GUTLYIVDDKVIGB-UHFFFAOYSA-N 0.000 description 1
- 150000001875 compounds Chemical class 0.000 description 1
- 238000007796 conventional method Methods 0.000 description 1
- 239000010730 cutting oil Substances 0.000 description 1
- 230000000593 degrading effect Effects 0.000 description 1
- ZBCBWPMODOFKDW-UHFFFAOYSA-N diethanolamine Chemical compound OCCNCCO ZBCBWPMODOFKDW-UHFFFAOYSA-N 0.000 description 1
- 229940043237 diethanolamine Drugs 0.000 description 1
- 125000004185 ester group Chemical group 0.000 description 1
- 125000001033 ether group Chemical group 0.000 description 1
- 150000002170 ethers Chemical class 0.000 description 1
- 238000000605 extraction Methods 0.000 description 1
- 125000001153 fluoro group Chemical group F* 0.000 description 1
- 238000010353 genetic engineering Methods 0.000 description 1
- 150000002334 glycols Chemical class 0.000 description 1
- 238000000227 grinding Methods 0.000 description 1
- 229910052736 halogen Inorganic materials 0.000 description 1
- 150000002367 halogens Chemical class 0.000 description 1
- 231100001261 hazardous Toxicity 0.000 description 1
- 125000000623 heterocyclic group Chemical group 0.000 description 1
- 125000001165 hydrophobic group Chemical group 0.000 description 1
- 150000002500 ions Chemical class 0.000 description 1
- 229940102253 isopropanolamine Drugs 0.000 description 1
- 239000000314 lubricant Substances 0.000 description 1
- 239000011777 magnesium Substances 0.000 description 1
- 229910052749 magnesium Inorganic materials 0.000 description 1
- 229910001629 magnesium chloride Inorganic materials 0.000 description 1
- 229910052748 manganese Inorganic materials 0.000 description 1
- 239000011572 manganese Substances 0.000 description 1
- WPBNNNQJVZRUHP-UHFFFAOYSA-L manganese(2+);methyl n-[[2-(methoxycarbonylcarbamothioylamino)phenyl]carbamothioyl]carbamate;n-[2-(sulfidocarbothioylamino)ethyl]carbamodithioate Chemical compound [Mn+2].[S-]C(=S)NCCNC([S-])=S.COC(=O)NC(=S)NC1=CC=CC=C1NC(=S)NC(=O)OC WPBNNNQJVZRUHP-UHFFFAOYSA-L 0.000 description 1
- 238000004519 manufacturing process Methods 0.000 description 1
- VUZPPFZMUPKLLV-UHFFFAOYSA-N methane;hydrate Chemical compound C.O VUZPPFZMUPKLLV-UHFFFAOYSA-N 0.000 description 1
- 230000000813 microbial effect Effects 0.000 description 1
- 239000002480 mineral oil Substances 0.000 description 1
- 238000002156 mixing Methods 0.000 description 1
- 239000011733 molybdenum Substances 0.000 description 1
- 229940076266 morganella morganii Drugs 0.000 description 1
- 150000002780 morpholines Chemical class 0.000 description 1
- 229910052757 nitrogen Inorganic materials 0.000 description 1
- SNQQPOLDUKLAAF-UHFFFAOYSA-N nonylphenol Chemical compound CCCCCCCCCC1=CC=CC=C1O SNQQPOLDUKLAAF-UHFFFAOYSA-N 0.000 description 1
- 238000005457 optimization Methods 0.000 description 1
- 238000006213 oxygenation reaction Methods 0.000 description 1
- 239000002304 perfume Substances 0.000 description 1
- 150000004714 phosphonium salts Chemical class 0.000 description 1
- NBIIXXVUZAFLBC-UHFFFAOYSA-N phosphoric acid Substances OP(O)(O)=O NBIIXXVUZAFLBC-UHFFFAOYSA-N 0.000 description 1
- 150000003013 phosphoric acid derivatives Chemical class 0.000 description 1
- 229910052698 phosphorus Inorganic materials 0.000 description 1
- 239000011295 pitch Substances 0.000 description 1
- 231100000614 poison Toxicity 0.000 description 1
- 229920001451 polypropylene glycol Polymers 0.000 description 1
- 239000011591 potassium Substances 0.000 description 1
- 235000007686 potassium Nutrition 0.000 description 1
- 159000000001 potassium salts Chemical class 0.000 description 1
- 239000003755 preservative agent Substances 0.000 description 1
- 150000003141 primary amines Chemical class 0.000 description 1
- 239000010734 process oil Substances 0.000 description 1
- 102000004196 processed proteins & peptides Human genes 0.000 description 1
- 108090000765 processed proteins & peptides Proteins 0.000 description 1
- 102000004169 proteins and genes Human genes 0.000 description 1
- 108090000623 proteins and genes Proteins 0.000 description 1
- 229940007042 proteus vulgaris Drugs 0.000 description 1
- 230000008929 regeneration Effects 0.000 description 1
- 238000011069 regeneration method Methods 0.000 description 1
- 230000001105 regulatory effect Effects 0.000 description 1
- 150000003335 secondary amines Chemical class 0.000 description 1
- 238000004062 sedimentation Methods 0.000 description 1
- 150000004760 silicates Chemical class 0.000 description 1
- 239000000344 soap Substances 0.000 description 1
- 239000001509 sodium citrate Substances 0.000 description 1
- NLJMYIDDQXHKNR-UHFFFAOYSA-K sodium citrate Chemical compound O.O.[Na+].[Na+].[Na+].[O-]C(=O)CC(O)(CC([O-])=O)C([O-])=O NLJMYIDDQXHKNR-UHFFFAOYSA-K 0.000 description 1
- DZCAZXAJPZCSCU-UHFFFAOYSA-K sodium nitrilotriacetate Chemical compound [Na+].[Na+].[Na+].[O-]C(=O)CN(CC([O-])=O)CC([O-])=O DZCAZXAJPZCSCU-UHFFFAOYSA-K 0.000 description 1
- 235000019832 sodium triphosphate Nutrition 0.000 description 1
- 238000010561 standard procedure Methods 0.000 description 1
- 238000003756 stirring Methods 0.000 description 1
- 125000000565 sulfonamide group Chemical group 0.000 description 1
- 229910052717 sulfur Inorganic materials 0.000 description 1
- 238000004381 surface treatment Methods 0.000 description 1
- 239000011269 tar Substances 0.000 description 1
- 150000003512 tertiary amines Chemical class 0.000 description 1
- 239000003440 toxic substance Substances 0.000 description 1
- UNXRWKVEANCORM-UHFFFAOYSA-I triphosphate(5-) Chemical compound [O-]P([O-])(=O)OP([O-])(=O)OP([O-])([O-])=O UNXRWKVEANCORM-UHFFFAOYSA-I 0.000 description 1
- 239000002699 waste material Substances 0.000 description 1
- 229910052725 zinc Inorganic materials 0.000 description 1
- 239000011701 zinc Substances 0.000 description 1
Images
Classifications
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B08—CLEANING
- B08B—CLEANING IN GENERAL; PREVENTION OF FOULING IN GENERAL
- B08B3/00—Cleaning by methods involving the use or presence of liquid or steam
- B08B3/04—Cleaning involving contact with liquid
- B08B3/08—Cleaning involving contact with liquid the liquid having chemical or dissolving effect
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B08—CLEANING
- B08B—CLEANING IN GENERAL; PREVENTION OF FOULING IN GENERAL
- B08B3/00—Cleaning by methods involving the use or presence of liquid or steam
- B08B3/04—Cleaning involving contact with liquid
- B08B3/10—Cleaning involving contact with liquid with additional treatment of the liquid or of the object being cleaned, e.g. by heat, by electricity or by vibration
- B08B3/102—Cleaning involving contact with liquid with additional treatment of the liquid or of the object being cleaned, e.g. by heat, by electricity or by vibration with means for agitating the liquid
-
- C—CHEMISTRY; METALLURGY
- C11—ANIMAL OR VEGETABLE OILS, FATS, FATTY SUBSTANCES OR WAXES; FATTY ACIDS THEREFROM; DETERGENTS; CANDLES
- C11D—DETERGENT COMPOSITIONS; USE OF SINGLE SUBSTANCES AS DETERGENTS; SOAP OR SOAP-MAKING; RESIN SOAPS; RECOVERY OF GLYCEROL
- C11D3/00—Other compounding ingredients of detergent compositions covered in group C11D1/00
- C11D3/16—Organic compounds
- C11D3/38—Products with no well-defined composition, e.g. natural products
- C11D3/381—Microorganisms
-
- C—CHEMISTRY; METALLURGY
- C23—COATING METALLIC MATERIAL; COATING MATERIAL WITH METALLIC MATERIAL; CHEMICAL SURFACE TREATMENT; DIFFUSION TREATMENT OF METALLIC MATERIAL; COATING BY VACUUM EVAPORATION, BY SPUTTERING, BY ION IMPLANTATION OR BY CHEMICAL VAPOUR DEPOSITION, IN GENERAL; INHIBITING CORROSION OF METALLIC MATERIAL OR INCRUSTATION IN GENERAL
- C23G—CLEANING OR DE-GREASING OF METALLIC MATERIAL BY CHEMICAL METHODS OTHER THAN ELECTROLYSIS
- C23G5/00—Cleaning or de-greasing metallic material by other methods; Apparatus for cleaning or de-greasing metallic material with organic solvents
-
- C11D2111/40—
Definitions
- the present invention relates to a method for cleaning objects and materials, and particularly, although not exclusively, for removing organic substances such as hydrocarbons, greases, waxes, oils, tars, pitches, proteins and peptides, particularly oils and greases and their decomposition products for biodegradation thereof.
- the invention also relates to apparatus for carrying out the method.
- the invention relates more specifically, although not exclusively, to degreasing biologically objects which are contaminated with cutting oils, lubricants, grinding oils or process oils, prior to the further treatment of said objects.
- Degreasing is an absolute necessity when the objects concerned are to be treated by processes such as phosphating processes, electrolytic processes, varnishing etc., if good results are to be achieved.
- Conventional degreasing of such objects is more often than not effected with a caustic soda solution, or lye, at high bath temperatures, which affords a good result initially.
- the bath gradually becomes enriched with oils removed from the objects and the degreasing capacity of the bath will fall-off radically when the oil contents of the bath exceeds 0.5 % by weight. This also applies to degreasing baths, which are maintained at temperatures of from 90 to 95°C. Objects which have been treated with a caustic soda solution are also difficult to wash clean.
- the objects are instead washed with tensides which emulsify the organic substances, whereafter the emulsified hydrocarbons are degraded to carbon dioxide and water, by adding nutrient salts to the bath, so as to activate the microorganisms present in the contaminating ssubstances and therewith result in the degradation of said substances.
- the novel method thus enables tensides to be used for cleaning purposes instead of caustic soda solutions. Furthermore, the tensides are not consumed to the same extent as the caustic soda solutions used in conventional methods, because the cleaning bath composed in accordance with the invention can be used effectively for two years owing to the degradation of entrant oils and greases. Conventional degreasing processes using caustic soda solutions can only be used effectively for a short period of time. Furthermore, the inventive cleaning process can be carried out at a low temperature.
- Objects or materials cleaned with the aid of tensides can also be washed easily.
- the use of tensides has been prohibited by the fact that their degreasing ability falls off rapidly with increasing oil levels, thereby necessitating quick replenishment of the tensides. Because of the tenside costs incurred hereby, such replenishment has rendered the process highly expensive.
- the proposed bacteria degradation or decomposition, of the contaminants present enables, for the first time, tensides to be used in an efficient and rational fashion.
- the present invention thus relates to a method for cleaning objects which is characterized by cleaning said objects by treating them with an aqueous solution of tensides such as to emulsify the organic contaminants present, and by adding nutrient salts to the tenside solution so as to initiate the growth of microorganisms for the purpose of biologically degrading the organic substances so that the tenside solution substantially is unaffected, the biodegradation being performed separately from or simultaneously with the cleaning.
- the oil is preferably accumulated in the cleaning bath, by adding thereto a tenside solution of basic pH 7-14, particularly by adding a basic tenside solution of pH 9-11.
- Cleaning can be effected with any kind of water-soluble tenside whatsoever, such as anionic, cationic, nonionic and amphoteric tensides.
- These can be tensides which are not-readily degradable by microorganisms, such as the halogen-containing (chloro-bromo- and fluoro-containing) tensides and heterocyclic tensides.
- Biodegradable tensides are preferably used, however, so that sludge, slime, separated from the process will not contaminate the surroundings. It is correct procedure, however, to control the process so that the microorganisms will not degrade the tensides.
- This control process is preferably effected by ensuring that the hydrocarbon content of the bath will not fall beneath 50 mg/liter and that the tenside content will not rise above 15 % by weight.
- the tensides will probably work at a pH of 7 up to an alkaline pH-value which will not block microbial growth. This pH-value is, at present, about 9.5, but may conceivably increase through genetic manipulation of the microorganisms. It has been found that good cleaning and degreasing results are achieved at alkaline pH-values above 8.5.
- the cleaning and biodegradation processes can be carried out at pH-values of from 7 to 14.
- the pH-value is preferably adjusted to between 9.0 and 9.5.
- the tenside content is preferably maintained at between 1-15 o/o by weight, preferably between 2-5 0/ 0 by weight in the case of objects soiled to normal levels, and between 5 and 10% by weight in the case of heavily soiled or contaminated objects.
- the tenside content of the cleaning bath may be maintained at a high level, while the bath in which biogradation takes place is maintained at the aforesaid tenside level.
- the content of organic substances should not fall beneath 50 mg/ml in the biodegradation process, since bacteria can begin to consume the tensides at lower contents of organic contaminants.
- the pH-value may fall rapidly as a result of the high consumption of emulsifying chemicals and also as a result of acid generation by dead bacteria.
- the contaminant content such as oil and grease should be maintained between 50-1000 mg/ml, preferably between 50 and 250 mg/ml, by adding to the system nutrient substances and pH-increasing substances.
- the temperature has also a decisive significance on optimum cleaning capacity.
- the temperature of the cleaning bath may be between 20 and 100°C.
- the bath should be maintained at a temperature of between 20 and 80°C, preferably between 30 and 40° C, and more preferably between 35 and 40°C, which has been found to be a spectacular working range in the case of mesophilic bacteria. Good cleaning results are obtained in this temperature range while, at the same time, the low temperature used requires only a low energy input.
- the degreasing temperature In the case of certain degreasing processes used, for instance, to remove waxes and paraffins, the degreasing temperature must lie above 50-S0°C, in which case it may be suitable to use separate facilities for degreasing and biodegradation purposes.
- the method is preferably made aerobic by introducing air, which can be atomized with the aid of nozzles.
- R is an alkyl residue having a long chain with 8-20 carbon atoms
- R' is a short alkyl residue having 1-8 carbon atoms or H
- X is an alkylene residue, particularly -(CH 2 ) n when n is 1, 2 or 3.
- the tensides used in accordance with the invention may be anionic tensides, such as detergents and soaps, e.g. salts of carboxylic acids, suitably alkali, particularly potassium salts and amine salts (mono-, di- and triethanolamine salts), morpholine salts of fatty acids R-COO-,
- Sulfuric acid esters can also be used, such as sulfated oils and fatty acids, such as sulfuric acid esters
- Alkyl sulfonates can also be used, such as simple alkyl sulfonates
- Alkylaryl sulfonates can also be used, such as alkyl naphthalene sulfonates
- cationic tensides particularly with chlorine or methylsulfate ions as catanions, e.g. amine salts, primary, secondary and teriary amine salts
- Amphoteric tensides can also be used, such as betaines
- Nonionic tensides can also be used, such as ethylene oxide adducts, such as alkyl polyethylene glycols R-(O-CH 2 -CH 2 ) n -OH,
- n about 0.3-0.7x 2 , where n is the number of moles ethylene oxide per mole of starting substance and c i's the number of carbon atoms in the hydrophobic residue).
- Fatty acid monoglycerides are also used anhydrosorbite-monofatty acid esters
- tensides can either be used individually or in mixtures. Cationic and nonionic tensides and mixtures thereof are used in particular, especially nondionic ethylene oxice adducts. Examples of tensides which can be used in this regard include 616 Allrent (contains nonionic tensides 2-nonyl-phenol, cationic tensides alkyl polyglycolether ammonium methyl sulfate, tetra potassium pyrophosphate, sodium citrate, preservatives, isopropanol, perfume, water and trisodium nitrilo triacetate).
- Via® Surf, Radion®, Meggem 8510 0 ethylene oxide adducts, glycols, phosphates, silicates. These detergents often contain an alkaline substance which will not block microbiological growth and auxiliary washing agents such as polyphosphates.
- the substances used to adjust the pH of the system shall be soluble in water, although they should not have a negative effect on the tensides and the microbiological conversion.
- Examples of such basic substances include alkali salts of basic pyrosulfates M 4 P 2 0 7 , where M signifies an alkali or alkali metal, preferably potassium, polyphosphates, tripolyphosphate, metasilicates such as sodium metasilicate, and primary, secondary, tertiary amines, particularly water soluble and/or grease-emulsifying primary, secondary and tertiary alkanol amines preferably having 1-10 carbon atoms and optionally substituted on the alkyl part, e.g.
- the pH-value may fall rapidly due to high consumption of emulsifying chemicals and to the generation of acid by dead bacteria. Consequently, in order to prevent the tenside-consumption from becoming excessive, the nutrient solution introduced to the bath may also contain a pH-increasing substance suitable for tensides, e.g. one of the aforementioned.
- sodium metasilicate or alternatively amines are used.
- Tensides based on sodium metasilicate are not suitable for use in a number of industrial surface -treatment processes, such as electrolytic processes for applying chromium, nickel and other metals. In the case of these processes, the alkaline substance used must be based on amines.
- the nutrient substances charged in accordance with the invention are those conventionally used for the cultivation of microorganisms. These substances shall contain N, S, Mg, K, P, and a carbon source, and may also contain trace metals such as Zn, Mn, Cu, Co, Mo.
- a suitable mixture will contain one part by weight Mg 2+ , one part by weight S0 2- , 8 parts by weight K 4+ , 32 parts by weight P0 3- , 80 parts by weight NH + , a carbon source in the form of glucose 1600 parts by weight, minor quantities of zink, manganese, copper, cobalt and molybdenum, pH-increasing substances, e.g. alkali and pH-lowering acid, e.g. H 3 P0 4 , 0 2 3000 parts by weight in the form oxygen or air.
- the composition of the nutrient substances forms part of the present state of the art and can be readily established by the skilled person.
- microorganisms activated in accordance with the present invention are the following:
- the inventive method enables tensides to be used effectively and in a rational manner to clean industrial goods prior to subjecting said goods to further treatment processes, degreasing of said goods having been previously effected mainly with caustic soda solutions.
- the method enables tensides to be used, because the content organic contaminants in the cleaning bath can be held constantly at a low level.
- the bath can be used for a period of up to about 2 years as opposed to a period of about 1 month in the case of the earlier known cleaning method, depending upon the quantity of contaminated objects, or goods, degreased in the bath.
- the objects cleaned in accordance with the inventive method can also be washed easily free from the cleaning solution.
- the present inventive method can also be carried out at low temperatures, in the region of 35-40° C, instead of the relatively high temperatures of 90-95° C used in the earlier degreasing process. This means that energy can be saved. Since energy can be saved and the chemicals can be used more effectively, there is obtained a cost reduction of 60-80%.
- the invention also relates to apparatus for carrying out the inventive cleaning method.
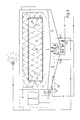
- This apparatus is characterized by a cleansing tank which is located above a washing liquid tank and above a rinsing liquid tank, the bottom of the cleansing tank being totally or partially common to the ceiling of the washing liquid tank and the rinsing liquid tank respectively.
- the bottoms of the three tanks may be conical in shape.
- the cleansing tank may have any desired shape, such as round, square, rectangular, although it is preferably open and round.
- the washing liquid and rinsing liquid tanks may also have any desired shape.
- the tanks are preferably closed.
- the rinsing liquid and washing liquid tanks may have a ceiling or roof whose total surface area is greater than or smaller than the bottom surface of the cleansing tank. These ceilings may also coincide fully or partially with the bottom of the cleansing tank. Thus, the rinsing liquid tank and washing liquid tank may project beyond the bottom of the cleansing tank or coincide with the outer boundaries thereof, as illustrated in Fig. 3, or the ceilings may leave part of the outer bottom surface of the cleansing tank free.
- the three tanks for cleansing and holding washing liquid and rinsing liquid respectively are preferably configured in a first cylindrical tank having a conical bottom.
- the tank is first divided transversely of its longitudinal axis, so as to obtain an upper, cleansing tank, and then diametrically downwards beneath the cleansing tank, so as to obtain a washing liquid tank and a rinsing liquid tank.
- Preferably, only the conical part of the first cylindrical tank is divided into a tank for washing liquid and a tank for rinsing liquid as illustrated in Fig. 3.
- the first cylindrical tank may also be partitioned transversely to the longitudinal axis, above the conical part, so that the washing-liquid and rinsing-liquid tanks obtain an upper cylindrical part and a lower conical part.
- outlet openings from the cleansing tank to the washing liquid tank and the rinsing liquid tank respectively openings for further transportation of washing and rinsing liquid from respective washing and rinsing liquid tanks to the cleansing tank, closeable outlets from respective washing liquid and rinsing liquid tanks, closeable inlets for fresh water, washing liquid and additives to the washing liquid tank and a closeable inlet for supplying fresh water to the rinsing liquid tank.
- the rinsing liquid and washing liquid tanks may include means for heating the contents of said tanks and also means for aerating said tanks.
- the washing liquid tank preferably includes aerating means.
- the washing liquid tank may also include agitating or stirring means. Agitation of the tank contents, however, is preferably effected through aerating of the tank.
- Fig. 3 When using the illustrated apparatus, goods or objects A to be cleaned are placed in the cleansing tank 1, e.g. with the aid of a basket.
- the valve 6 is opened and a pump activated, whereupon washing liquid comprising tenside and alkali is pumped up from the tank 2 and applied to the objects or goods under powerful pressure. This can either be carried out manually or through nozzles installed in the cleansing tank 1.
- the washing liquid emulsifies oil, grease and like contaminants.
- the valve 4 in the cleansing tank 1 is opened, and the used washing liquid is allowed to run back to the washing liquid tank 2.
- valve 7 is then opened and a pump activated, whereupon rinsing water is sprayed, in the aforedescribed manner, over the objects, to rinse washing liquid therefrom.
- the valve 5 in the cleansing tank 1 is then opened and the rinsing water is allowed to run back to the tank 3.
- oil and grease will have accumulated in the washing liquid in the tank 2.
- continuous metering of tensides, alkali and nutrient salts can be commenced and the pH-value adjusted to 8.5-9.5, so as to enable maximum growth of the microorganisms.
- the washing liquid tank which in the case of the illustrated apparatus is the tank in which the contaminants are broken down biologically, is aerated and the contents thereof agitated with the aid of air entering through an air conduit 16.
- the pH-value falls with increasing oil contents.
- the pH-value is therefore adjusted to the aforesaid pH-range, so as to maintain the oil content within the range of 100-500 mg/liter.
- the method is facilitated by the fact that the lower part of the apparatus is conical in shape. Because the washing liquid tank 2 is conical it has a confined lowest point to which contaminants and dead bacteria are able to sink and from which the contaminants can be removed continuously to a waste outlet, through the valve 8.
- the rinsing water tank 3 is also conical in shape and incorporates an outlet valve 9.
- the bacteria population can be run at low temperatures, preferably at 35-40° C in the case of a thermophilic process or at 40-60° C in the case of mesophilic bacteria. These temperature ranges can be maintained in the washing liquid tank 2 with the aid of the heating means 13.
- the tank contents may also be heated with hot air.
- the temperature of the contents of the rinsing water tank 3 can also be regulated, with the aid of the means 14. Optimization of the washing process for high bacteria populations, by adjusting the pH-value and the nutrient substances and fresh tensides, enables the tenside consumption to be kept as low as 10°/o of the calculated amount of oil introduced to the system. Oxygen is supplied continuously, by aeration, and the oxygen blow is positioned in a manner such that dead bacteria and entrant contaminants can be removed continuously through the valve 6. This affords optimum bacteria growth conditions and enables tensides to be used effectively and rationally for degreasing industrial objects and goods.
- a stream of objects, or goods, to be cleansed are conveyed by a transverse conveyer or drum to the process bath 17 see Fig. 1) and are left in the bath for about 2-20 minutes, so that oil and contaminants are washed off.
- Air is blown with a motor 18 and a fan 19 through line 20 into the bath continuously from the bottom of said bath, so as to thorougly agitate the bath contents and maximize the oxygen supply.
- Bath liquid is fed through a valve 21 with a pump 22 continuously from the bath to a separator unit 23, for extraction of contaminants and dead bacteria through a valve 24 to a sludge tank 25. Sludge, or slime, is withdrawn from the separator compression zone.
- Tensides are metered to the bath continuously from a tank 26 at the same rate as the tensides are consumed.
- Nutrient solution, Camex Bio 104-1 is metered to the bath intermittently from a tank 27, so as to maintain the set-point values set on the pH-meter constant. The best mesophilic bacteria culture is obtained when the bath temperature is maintained at 38 ⁇ 1°C.
- the nutrient solution and the tenside solution are fed through valves 28, 29, 30 and 31 respectively with pumps 32 and 33 and are mixed with separated solution from the separator 23 through valve 34 in a mixing unit 35 and fed to the process tank 17.
- a thermostate 36 and a heater 37 are placed in the process tank 16. Fresh water is fed into the tank 17 through a line 37.
- the tenside solution used is Camex Bio 104, which comprises a mixture of 1-5% by weight sodium metasilicate, 5-10% tetrapotassium pyrophosphate, 5-10 % nonionic and cationic tensides, having a pH of 13. This solution is diluted to a 5%-tenside solution of pH 10.
- the nutrient solution used has the following basic composition
- This solution is diluted with water to 300 liters. 2 kg of this basic solution is mixed with 210 kg NH 4 Si0 3 and the mixture diluted to 600 liters.
- Example 1 above is repeated but with the difference that the nutrient solution is mixed with amine instead of sodium silicate, in accordance with the following.
- 2 kg of basic solution are mixed with 400 kg alkaline amine such as diethanol amine or trietanol amine.
- the mix is diluted with water to 600 liters.
- This example is particularly suited for application with goods or objects which are to be coated with chromium, nickel or other metals by electrolytic coating processes.
- the objects or goods are passed through a washing machine 39 on a conveyor belt 40 and then rinced in a rincing unit 42.
- a 50/o-tenside solution is mixed from the basic solution according to Example 1 at a temperature of 50-90° C, whereafter the solution is applied under high pressure to the surfaces of the objects or goods, so as to dissolve and emulsify oil and contaminants present thereon. Washing and rincing is done through nozzzles 42. Part of the washing liquid is transferred to a biodegradation unit 43 for the reduction of oil and contaminants.
- Nutrient solution as in example 1 is added to the biodegradation unit 43 from a tank 44.
- Fresh tenside solution is fed to the regeneration unit 43 from a tank 45.
- the temperature in the biodegradation unit is lowered to 20-30° C, upon oxygenation of the unit contents, by injecting air thereinto. Consequently, a mesophilic bacteria culture obtains optimized growth conditions when the flow passes through the biological unit.
- the flow is then passed to the central separation part, in which dead bacteria and sludge, slime, are passed to a compression zone for removal to a sludge tank 46. Air is also injected in the cleaning tank 39. Heaters are arranged in all tanks 39, 41 and 43.
- the biodegradation process is controlled in the same manner as that described in Example 1 and the injection of air and metering of tensides is effected continuously, whereas the alkaline nutrient solution is metered intermittently, to a pH-value of 9.4-9.5.
Priority Applications (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| AT88850310T ATE88224T1 (de) | 1987-09-21 | 1988-09-19 | Reinigungsverfahren und geraet dafuer. |
Applications Claiming Priority (4)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| SE8703644 | 1987-09-21 | ||
| SE8703644A SE8703644D0 (sv) | 1987-09-21 | 1987-09-21 | Avfettningsforfarande |
| SE8801511A SE8801511L (sv) | 1987-09-21 | 1988-04-22 | Avfettningsfoerfarande och anordning daerfoer |
| SE8801511 | 1988-04-22 |
Publications (3)
| Publication Number | Publication Date |
|---|---|
| EP0309432A2 true EP0309432A2 (de) | 1989-03-29 |
| EP0309432A3 EP0309432A3 (de) | 1991-03-13 |
| EP0309432B1 EP0309432B1 (de) | 1993-04-14 |
Family
ID=26659951
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| EP88850310A Revoked EP0309432B1 (de) | 1987-09-21 | 1988-09-19 | Reinigungsverfahren und Gerät dafür |
Country Status (6)
| Country | Link |
|---|---|
| EP (1) | EP0309432B1 (de) |
| JP (1) | JPH01159387A (de) |
| AU (1) | AU613402B2 (de) |
| DE (1) | DE3880246T2 (de) |
| ES (1) | ES2041338T3 (de) |
| SE (1) | SE8801511L (de) |
Cited By (12)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| EP0588282A1 (de) * | 1992-09-14 | 1994-03-23 | Haldor Dr. Aamot | Reinigung- und Spülverfahren für industrielle Produkte |
| FR2706790A1 (en) * | 1993-06-21 | 1994-12-30 | Vitobio Sa | Process and biological composition for cleaning paint brushes and rollers |
| DE4340058A1 (de) * | 1993-11-24 | 1995-06-01 | Aamot Haldor | Verfahren zum Aufarbeiten gebrauchter Entfettungs- und Reinigungslösungen |
| WO1996004083A1 (en) * | 1994-08-01 | 1996-02-15 | B & S Research | Method and apparatus for remediating contaminated material |
| EP0715549A1 (de) * | 1993-07-16 | 1996-06-12 | Technological Resources Pty. Ltd. | Abbeizen von farbe, lack und anderen beschichtungen von metall- und legierungswerkstoffen |
| EP0784518A2 (de) * | 1994-09-30 | 1997-07-23 | Advanced Bioremediation Systems, Inc. | Vorrichtung zum mikrobiologischen waschen von teilen |
| DE19644714A1 (de) * | 1996-10-28 | 1998-04-30 | Klaus F Dr Ing Roth | Verfahren zum Abbau von Kühlschmierstoffen |
| US6019110A (en) * | 1994-09-30 | 2000-02-01 | Chemfree Corporation | Parts washing system |
| US6328045B1 (en) * | 1994-09-30 | 2001-12-11 | Chemfree Corporation | Parts washing system |
| US6391836B1 (en) * | 2001-01-16 | 2002-05-21 | Bioclean, Usa | Biological cleaning system which forms a conversion coating on substrates |
| WO2004113589A1 (de) * | 2003-06-25 | 2004-12-29 | Cb Chemie Und Biotechnologie Gmbh | Vorrichtung zur reinigung von mit öl und fett beschmutzten maschinenteilen nach art einer waschmaschine mit bioreaktor |
| DE102019005395A1 (de) * | 2019-07-24 | 2021-01-28 | Jens Fridolin Olschewski | Verfahren zur Aufbereitung von durch Mineralöl, natives Öl oder/und Teeröl verunreinigten festen Partikeln |
Citations (4)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US4124504A (en) * | 1977-05-25 | 1978-11-07 | George Munden | Process for treating caustic wash solutions |
| GB2065631A (en) * | 1979-12-20 | 1981-07-01 | Inst Francais Du Petrole | Despersant and biodegradant formulations |
| FR2528087A1 (fr) * | 1982-06-04 | 1983-12-09 | Nal Expl Oceans Centre | Procede de degradation et de dispersion des nappes d'hydrocarbures en mer par fermentation |
| EP0116151A1 (de) * | 1982-12-22 | 1984-08-22 | Henkel Kommanditgesellschaft auf Aktien | Verfahren zur Regenerierung bzw. zum Recycling von wässrigen Entfettungs- und Reinigungslösungen |
Family Cites Families (2)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| GB1574922A (en) * | 1976-09-24 | 1980-09-10 | Niigata Engineering Co Ltd | Method and unit for wastewater treatment by microorganisms |
| CH667449A5 (de) * | 1986-04-23 | 1988-10-14 | Sulzer Ag | Verfahren zur biologischen reinigung von wasser oder abwasser von organischen stickstoffhaltigen verunreinigungen. |
-
1988
- 1988-04-22 SE SE8801511A patent/SE8801511L/ not_active Application Discontinuation
- 1988-09-13 AU AU22163/88A patent/AU613402B2/en not_active Ceased
- 1988-09-19 EP EP88850310A patent/EP0309432B1/de not_active Revoked
- 1988-09-19 ES ES198888850310T patent/ES2041338T3/es not_active Expired - Lifetime
- 1988-09-19 DE DE88850310T patent/DE3880246T2/de not_active Expired - Fee Related
- 1988-09-20 JP JP63236204A patent/JPH01159387A/ja active Pending
Patent Citations (4)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US4124504A (en) * | 1977-05-25 | 1978-11-07 | George Munden | Process for treating caustic wash solutions |
| GB2065631A (en) * | 1979-12-20 | 1981-07-01 | Inst Francais Du Petrole | Despersant and biodegradant formulations |
| FR2528087A1 (fr) * | 1982-06-04 | 1983-12-09 | Nal Expl Oceans Centre | Procede de degradation et de dispersion des nappes d'hydrocarbures en mer par fermentation |
| EP0116151A1 (de) * | 1982-12-22 | 1984-08-22 | Henkel Kommanditgesellschaft auf Aktien | Verfahren zur Regenerierung bzw. zum Recycling von wässrigen Entfettungs- und Reinigungslösungen |
Cited By (28)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US5532162A (en) * | 1992-09-14 | 1996-07-02 | Aamot; Haldor | Elimination of used degreasing solution through biological degradation |
| EP0588282A1 (de) * | 1992-09-14 | 1994-03-23 | Haldor Dr. Aamot | Reinigung- und Spülverfahren für industrielle Produkte |
| FR2706790A1 (en) * | 1993-06-21 | 1994-12-30 | Vitobio Sa | Process and biological composition for cleaning paint brushes and rollers |
| EP0715549A1 (de) * | 1993-07-16 | 1996-06-12 | Technological Resources Pty. Ltd. | Abbeizen von farbe, lack und anderen beschichtungen von metall- und legierungswerkstoffen |
| EP0715549A4 (de) * | 1993-07-16 | 1996-07-17 | ||
| DE4340058A1 (de) * | 1993-11-24 | 1995-06-01 | Aamot Haldor | Verfahren zum Aufarbeiten gebrauchter Entfettungs- und Reinigungslösungen |
| US5772782A (en) * | 1994-08-01 | 1998-06-30 | B&S Research, Inc. | Method for remediating contaminated material |
| WO1996004083A1 (en) * | 1994-08-01 | 1996-02-15 | B & S Research | Method and apparatus for remediating contaminated material |
| US5589004A (en) * | 1994-08-01 | 1996-12-31 | B&S Research, Inc. | Method for remediating contaminated material |
| US5709234A (en) * | 1994-08-01 | 1998-01-20 | B&S Research, Inc. | Method and apparatus for remediating contaminated material |
| US6095163A (en) * | 1994-09-30 | 2000-08-01 | Chemfree Corporation | Parts washing system |
| US6019110A (en) * | 1994-09-30 | 2000-02-01 | Chemfree Corporation | Parts washing system |
| US6074491A (en) * | 1994-09-30 | 2000-06-13 | Chemfree Corporation | Parts washing system |
| EP0784518A2 (de) * | 1994-09-30 | 1997-07-23 | Advanced Bioremediation Systems, Inc. | Vorrichtung zum mikrobiologischen waschen von teilen |
| US6328045B1 (en) * | 1994-09-30 | 2001-12-11 | Chemfree Corporation | Parts washing system |
| US7980257B2 (en) | 1994-09-30 | 2011-07-19 | Zymo International, Inc. | Parts washing system |
| EP0784518B1 (de) * | 1994-09-30 | 2002-06-26 | ZYMO International Inc. | Vorrichtung zum mikrobiologischen waschen von teilen |
| DE19644714A1 (de) * | 1996-10-28 | 1998-04-30 | Klaus F Dr Ing Roth | Verfahren zum Abbau von Kühlschmierstoffen |
| WO2002057031A3 (en) * | 2001-01-16 | 2002-09-19 | Bio Clean Llc | Biological cleaning system |
| WO2002057031A2 (en) * | 2001-01-16 | 2002-07-25 | Bio Clean Llc | Biological cleaning system |
| EP1351782A2 (de) * | 2001-01-16 | 2003-10-15 | Bio Clean LLC | Biologisches reinigungssystem |
| EP1351782A4 (de) * | 2001-01-16 | 2004-05-12 | Bio Clean Llc | Biologisches reinigungssystem |
| US6884301B2 (en) * | 2001-01-16 | 2005-04-26 | Bioclean, Usa | Biological cleaning system comprising microbes for digesting oils and/or greases |
| EP2316583A1 (de) * | 2001-01-16 | 2011-05-04 | Bio Clean LLC | Biologisches Reinigungssystem |
| US6391836B1 (en) * | 2001-01-16 | 2002-05-21 | Bioclean, Usa | Biological cleaning system which forms a conversion coating on substrates |
| WO2004113589A1 (de) * | 2003-06-25 | 2004-12-29 | Cb Chemie Und Biotechnologie Gmbh | Vorrichtung zur reinigung von mit öl und fett beschmutzten maschinenteilen nach art einer waschmaschine mit bioreaktor |
| EP1491661A1 (de) * | 2003-06-25 | 2004-12-29 | CB Chemie und Biotechnologie GmbH | Vorrichtung zur Reinigung von mit Öl und Fett beschmutzten Maschinenteilen nach Art einer Waschmaschine mit Bioreaktor |
| DE102019005395A1 (de) * | 2019-07-24 | 2021-01-28 | Jens Fridolin Olschewski | Verfahren zur Aufbereitung von durch Mineralöl, natives Öl oder/und Teeröl verunreinigten festen Partikeln |
Also Published As
| Publication number | Publication date |
|---|---|
| ES2041338T3 (es) | 1993-11-16 |
| DE3880246D1 (de) | 1993-05-19 |
| DE3880246T2 (de) | 1993-11-25 |
| AU613402B2 (en) | 1991-08-01 |
| SE8801511D0 (sv) | 1988-04-22 |
| EP0309432A3 (de) | 1991-03-13 |
| SE8801511L (sv) | 1989-03-22 |
| AU2216388A (en) | 1989-03-23 |
| EP0309432B1 (de) | 1993-04-14 |
| JPH01159387A (ja) | 1989-06-22 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| EP0309432B1 (de) | Reinigungsverfahren und Gerät dafür | |
| US5532162A (en) | Elimination of used degreasing solution through biological degradation | |
| JP7261174B2 (ja) | 産業、機械及びレストラン機器を維持する材料及び方法 | |
| US5776351A (en) | Method for regeneration and closed loop recycling of contaminated cleaning solution | |
| EP0424340A2 (de) | Verfahren zum Beizen von Eisen- und Stahloberflächen | |
| CN101132994B (zh) | 废气废水处理装置以及废气废水处理方法 | |
| US5885459A (en) | Process for purifying and reusing surfactant-containing waste waters | |
| CN1325404C (zh) | 一种油田油泥砂无害化处理方法和处理装置 | |
| US20080011659A1 (en) | Water purification and treatment apparatus and treatment process using the apparatus | |
| US6063206A (en) | De-oiling process using enzymes | |
| US6057147A (en) | Apparatus and method for bioremediation of hydrocarbon-contaminated objects | |
| KR100242777B1 (ko) | 유기질 폐액의 폭기조와 그 폭기조를 이용한 폭기처리장치 | |
| US6884301B2 (en) | Biological cleaning system comprising microbes for digesting oils and/or greases | |
| JPH11323391A (ja) | 金属製品用水系脱脂処理浴及びその運転方法 | |
| JP6508503B1 (ja) | 種微生物の保管方法及び種微生物の保管装置 | |
| JP4654405B2 (ja) | 油生分解方法及び油生分解装置 | |
| Savchuk et al. | Research into processes of wastewater treatment at plants of meat processing industry by flotation and coagulation | |
| RU2214972C1 (ru) | Способ очистки воды | |
| CS200526B2 (en) | Waste water treatment method and device for making the said method | |
| KR101732680B1 (ko) | 도축시설 또는 육가공시설의 슬러지 저감장치 | |
| US4209489A (en) | Apparatus for treating spent hydrochloric acid | |
| DK176509B1 (da) | Fremgangsmåde til rensning af forurenet vand ved aktivslammetoden | |
| JPH11151497A (ja) | 廃水処理装置 | |
| PL161554B1 (pl) | Sposób chemicznego czyszczenia instalacji wodnych, zwłaszcza sieci centralnego ogrzewania | |
| KR20170101162A (ko) | 고농도 유지 폐수 처리 방법 |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| PUAI | Public reference made under article 153(3) epc to a published international application that has entered the european phase |
Free format text: ORIGINAL CODE: 0009012 |
|
| AK | Designated contracting states |
Kind code of ref document: A2 Designated state(s): AT BE CH DE ES FR GB GR IT LI LU NL SE |
|
| RHK1 | Main classification (correction) |
Ipc: C23G 1/36 |
|
| PUAL | Search report despatched |
Free format text: ORIGINAL CODE: 0009013 |
|
| AK | Designated contracting states |
Kind code of ref document: A3 Designated state(s): AT BE CH DE ES FR GB GR IT LI LU NL SE |
|
| 17P | Request for examination filed |
Effective date: 19910511 |
|
| 17Q | First examination report despatched |
Effective date: 19920513 |
|
| GRAA | (expected) grant |
Free format text: ORIGINAL CODE: 0009210 |
|
| AK | Designated contracting states |
Kind code of ref document: B1 Designated state(s): AT BE CH DE ES FR GB GR IT LI LU NL SE |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: BE Effective date: 19930414 Ref country code: IT Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT;WARNING: LAPSES OF ITALIAN PATENTS WITH EFFECTIVE DATE BEFORE 2007 MAY HAVE OCCURRED AT ANY TIME BEFORE 2007. THE CORRECT EFFECTIVE DATE MAY BE DIFFERENT FROM THE ONE RECORDED. Effective date: 19930414 Ref country code: CH Effective date: 19930414 Ref country code: AT Effective date: 19930414 Ref country code: LI Effective date: 19930414 Ref country code: SE Effective date: 19930414 Ref country code: NL Effective date: 19930414 |
|
| REF | Corresponds to: |
Ref document number: 88224 Country of ref document: AT Date of ref document: 19930415 Kind code of ref document: T |
|
| REF | Corresponds to: |
Ref document number: 3880246 Country of ref document: DE Date of ref document: 19930519 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: GR Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 19930716 |
|
| REG | Reference to a national code |
Ref country code: CH Ref legal event code: PL |
|
| ET | Fr: translation filed | ||
| PGFP | Annual fee paid to national office [announced via postgrant information from national office to epo] |
Ref country code: GB Payment date: 19930909 Year of fee payment: 6 |
|
| PGFP | Annual fee paid to national office [announced via postgrant information from national office to epo] |
Ref country code: ES Payment date: 19930910 Year of fee payment: 6 |
|
| NLV1 | Nl: lapsed or annulled due to failure to fulfill the requirements of art. 29p and 29m of the patents act | ||
| PGFP | Annual fee paid to national office [announced via postgrant information from national office to epo] |
Ref country code: FR Payment date: 19930916 Year of fee payment: 6 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: LU Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 19930930 |
|
| REG | Reference to a national code |
Ref country code: ES Ref legal event code: FG2A Ref document number: 2041338 Country of ref document: ES Kind code of ref document: T3 |
|
| PGFP | Annual fee paid to national office [announced via postgrant information from national office to epo] |
Ref country code: DE Payment date: 19931125 Year of fee payment: 6 |
|
| PLBI | Opposition filed |
Free format text: ORIGINAL CODE: 0009260 |
|
| 26 | Opposition filed |
Opponent name: DR.AAMOT, HALDOR Effective date: 19940114 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: GB Effective date: 19940919 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: ES Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 19940920 |
|
| GBPC | Gb: european patent ceased through non-payment of renewal fee |
Effective date: 19940919 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: FR Effective date: 19950531 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: DE Effective date: 19950601 |
|
| REG | Reference to a national code |
Ref country code: FR Ref legal event code: ST |
|
| RDAG | Patent revoked |
Free format text: ORIGINAL CODE: 0009271 |
|
| STAA | Information on the status of an ep patent application or granted ep patent |
Free format text: STATUS: PATENT REVOKED |
|
| 27W | Patent revoked |
Effective date: 19950812 |