EP0149938A2 - Elektrochemisches Verfahren zur Entfernung von Gangstein, das metallische Elemente umgibt, in losem Material - Google Patents
Elektrochemisches Verfahren zur Entfernung von Gangstein, das metallische Elemente umgibt, in losem Material Download PDFInfo
- Publication number
- EP0149938A2 EP0149938A2 EP84402618A EP84402618A EP0149938A2 EP 0149938 A2 EP0149938 A2 EP 0149938A2 EP 84402618 A EP84402618 A EP 84402618A EP 84402618 A EP84402618 A EP 84402618A EP 0149938 A2 EP0149938 A2 EP 0149938A2
- Authority
- EP
- European Patent Office
- Prior art keywords
- basket
- gangue
- metallic elements
- cell
- anode
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Granted
Links
Images
Classifications
-
- C—CHEMISTRY; METALLURGY
- C25—ELECTROLYTIC OR ELECTROPHORETIC PROCESSES; APPARATUS THEREFOR
- C25F—PROCESSES FOR THE ELECTROLYTIC REMOVAL OF MATERIALS FROM OBJECTS; APPARATUS THEREFOR
- C25F1/00—Electrolytic cleaning, degreasing, pickling or descaling
-
- C—CHEMISTRY; METALLURGY
- C25—ELECTROLYTIC OR ELECTROPHORETIC PROCESSES; APPARATUS THEREFOR
- C25F—PROCESSES FOR THE ELECTROLYTIC REMOVAL OF MATERIALS FROM OBJECTS; APPARATUS THEREFOR
- C25F7/00—Constructional parts, or assemblies thereof, of cells for electrolytic removal of material from objects; Servicing or operating
Definitions
- the present invention relates to an electrochemical process for removing the gangue surrounding metallic elements in bulk materials, notably originating from archaeological excavations and, more particularly, from marine archaeological excavations.
- the present invention aims to provide a method allowing the treatment of large quantities of bulk materials to separate the metallic elements, thus making possible the treatment of materials from marine archaeological excavations.
- the cathodic treatment can be improved by placing cathodic conductive elements within the basket, for example vertical grids in addition to the central metallic part. It is also possible to add, during filling with the bulk materials, metallic chains or flexible metallic mesh which are thus embedded in the bulk mass.
- the anode is advantageously of cylindrical symmetry and arranged around the basket.
- the conductors forming the cathode and the anode can be made of any low-destructible material having sufficient conductivity. Iron or steel can be used, for example.
- the basket is preferably made of a material which does not conduct electricity to allow the anions (chlorides, sulphides) to migrate from the interior of the basket to the external anode. It can be made in particular of plastic.
- the basic aqueous bath which is contained in the cell is advantageously a bath containing from 8 to 40 grams of OH / 1 ions. It is possible for this purpose to use solutions of hydroxides or water-soluble carbonates, for example KOH, NaOH, Na 2 C0 3 , LiOH.
- the treatment can generally be carried out at a temperature close to room temperature (from 15 to 50 ° C).
- the treatment time depends on the nature of the gangue and its size on the surface and in volume. Generally, the processing time can vary from a few tens of hours to a few hundred hours and is typically 100 to 600 hours.
- An additional dechlorination treatment can be carried out in a conventional manner if necessary.
- the method according to the present invention also finds application in the processing of ores containing native metals and, in particular, precious metals such as gold or silver.
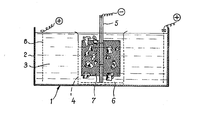
- the electrolysis cell 1 comprises a tank 2 of cylindrical shape. This tank contains an aqueous bath 3 consisting of an aqueous solution of potassium hydroxide at 5% by weight.
- a basket 4 of cylindrical plastic shape In the center of this tank is placed a basket 4 of cylindrical plastic shape, the side walls of which are pierced with holes.
- the assembly of the rod 5 and the grids 6 and 7 forms the cathode.
- a cylindrical grid 8 of stainless steel is arranged around the basket 4. This grid forms the anode.
- the basket can have a diameter of 40 cm and a height of 50 cm.
- a polarization is imposed between the anode and the cathode leading to a significant release of hydrogen by a current of 10A at approximately 3V of voltage.
- the polarization is maintained for 500 hours.
Landscapes
- Chemical & Material Sciences (AREA)
- Engineering & Computer Science (AREA)
- Chemical Kinetics & Catalysis (AREA)
- Electrochemistry (AREA)
- Materials Engineering (AREA)
- Metallurgy (AREA)
- Organic Chemistry (AREA)
- Electrolytic Production Of Metals (AREA)
- Electrolytic Production Of Non-Metals, Compounds, Apparatuses Therefor (AREA)
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| FR8400922A FR2558486B1 (fr) | 1984-01-20 | 1984-01-20 | Procede electrochimique pour enlever la gangue entourant des elements metalliques dans des materiaux en vrac |
| FR8400922 | 1984-01-20 |
Publications (3)
| Publication Number | Publication Date |
|---|---|
| EP0149938A2 true EP0149938A2 (de) | 1985-07-31 |
| EP0149938A3 EP0149938A3 (en) | 1985-08-21 |
| EP0149938B1 EP0149938B1 (de) | 1987-07-08 |
Family
ID=9300345
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| EP19840402618 Expired EP0149938B1 (de) | 1984-01-20 | 1984-12-17 | Elektrochemisches Verfahren zur Entfernung von Gangstein, das metallische Elemente umgibt, in losem Material |
Country Status (7)
| Country | Link |
|---|---|
| EP (1) | EP0149938B1 (de) |
| EG (1) | EG16769A (de) |
| ES (1) | ES8605304A1 (de) |
| FR (1) | FR2558486B1 (de) |
| GR (1) | GR850103B (de) |
| TR (1) | TR22020A (de) |
| YU (1) | YU7585A (de) |
Cited By (3)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| FR2626586A1 (fr) * | 1988-02-01 | 1989-08-04 | Electricite De France | Procede electrochimique pour le nettoyage de materiaux en feuille et dispositifs pour sa mise en oeuvre |
| EP0474365A1 (de) * | 1990-08-31 | 1992-03-11 | United Kingdom Atomic Energy Authority | Filterreinigung |
| WO2001040551A1 (en) * | 1999-12-01 | 2001-06-07 | Qinetiq Limited | Method of electrochemical in situ disposal of metal structures |
Family Cites Families (2)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| GB505273A (en) * | 1936-08-01 | 1939-05-01 | John Galbraith Quiros Worledge | Electro-chemical process for renovating table plate and other metal surfaces |
| US3497445A (en) * | 1967-07-28 | 1970-02-24 | Economics Lab | Apparatus for cleaning and detarnishing silverware |
-
1984
- 1984-01-20 FR FR8400922A patent/FR2558486B1/fr not_active Expired
- 1984-12-17 EP EP19840402618 patent/EP0149938B1/de not_active Expired
- 1984-12-21 TR TR2202084A patent/TR22020A/xx unknown
-
1985
- 1985-01-15 GR GR850103A patent/GR850103B/el unknown
- 1985-01-16 ES ES539612A patent/ES8605304A1/es not_active Expired
- 1985-01-18 YU YU7585A patent/YU7585A/xx unknown
- 1985-01-19 EG EG2985A patent/EG16769A/xx active
Cited By (3)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| FR2626586A1 (fr) * | 1988-02-01 | 1989-08-04 | Electricite De France | Procede electrochimique pour le nettoyage de materiaux en feuille et dispositifs pour sa mise en oeuvre |
| EP0474365A1 (de) * | 1990-08-31 | 1992-03-11 | United Kingdom Atomic Energy Authority | Filterreinigung |
| WO2001040551A1 (en) * | 1999-12-01 | 2001-06-07 | Qinetiq Limited | Method of electrochemical in situ disposal of metal structures |
Also Published As
| Publication number | Publication date |
|---|---|
| EP0149938B1 (de) | 1987-07-08 |
| ES539612A0 (es) | 1986-04-01 |
| ES8605304A1 (es) | 1986-04-01 |
| EG16769A (en) | 1993-04-30 |
| EP0149938A3 (en) | 1985-08-21 |
| FR2558486A1 (fr) | 1985-07-26 |
| YU7585A (en) | 1988-04-30 |
| TR22020A (tr) | 1986-01-13 |
| GR850103B (de) | 1985-05-16 |
| FR2558486B1 (fr) | 1986-06-13 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| Chen et al. | Mineralogical characterization of a copper anode and the anode slimes from the La Caridad copper refinery of Mexicana de Cobre | |
| US6319391B1 (en) | Removal of metal from graphite | |
| US3772003A (en) | Process for the electrolytic recovery of lead, silver and zinc from their ore | |
| EP0149938B1 (de) | Elektrochemisches Verfahren zur Entfernung von Gangstein, das metallische Elemente umgibt, in losem Material | |
| Moats et al. | The role of electrolyte additives on passivation behaviour during copper electrorefining | |
| Chen et al. | Mineralogical characterization of anode slimes—9. The reaction of Kidd creek anode slimes with various lixiviants | |
| EP0620607A1 (de) | Verfahren zur elektrolytischen Behandlung von Altbatterien | |
| Cheng et al. | Fundamental studies of copper anode passivation during electrorefining: Part II. Surface morphology | |
| Chen et al. | Mineralogical Characterization of Anode Slimes: Part V—Nickel-Rich Copper Anodes from the CCR Division of Noranda Minerals Inc. | |
| RU2064344C1 (ru) | Магнитогравитационный сепаратор | |
| CN110438555A (zh) | 一种电化学回收hw13类废弃线路板上贵金属的反应装置及方法 | |
| FR2513904A1 (fr) | Procede et installation pour recuperer des composants d'accumulateurs | |
| Li et al. | Effect of additives on anode passivation in direct electrolysis process of copper—nickel based alloy scraps | |
| FR2502187A1 (fr) | Procede et dispositif pour l'electrotraitement de materiaux composites pulverulents | |
| EP0143051B1 (de) | Verfahren zur Entfernung von Gangstein von Metallstücken | |
| Manilevich et al. | Improvement of the efficiency of electrochemical refining of cobalt | |
| SU1613591A1 (ru) | Система кучного выщелачивани полезных ископаемых | |
| EP4198153A1 (de) | Verfahren zur selektiven solubilisierung einer nickelbasierten schicht eines mehrschichtigen stapels | |
| EP0443929A1 (de) | Verfahren zur Vernichtung von elektrischen Altbatterien und Wiedergewinnung der verschiedenen Komponenten | |
| FR2617508A1 (fr) | Structure anodique pour raffinage electrolytique | |
| US491686A (en) | Ore-amalgamator | |
| EP2188092B1 (de) | Vorrichtung und verfahren zur reinigung von metallteilen | |
| Tomasek et al. | Gold extraction from the electronical scrap | |
| US640718A (en) | Process of extracting precious metals. | |
| US590524A (en) | Apparatus for extracting precious metals from black sand |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| PUAI | Public reference made under article 153(3) epc to a published international application that has entered the european phase |
Free format text: ORIGINAL CODE: 0009012 |
|
| PUAL | Search report despatched |
Free format text: ORIGINAL CODE: 0009013 |
|
| AK | Designated contracting states |
Designated state(s): BE GB IT NL |
|
| AK | Designated contracting states |
Designated state(s): BE GB IT NL |
|
| 17P | Request for examination filed |
Effective date: 19851207 |
|
| 17Q | First examination report despatched |
Effective date: 19860813 |
|
| GRAA | (expected) grant |
Free format text: ORIGINAL CODE: 0009210 |
|
| AK | Designated contracting states |
Kind code of ref document: B1 Designated state(s): BE GB IT NL |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: NL Effective date: 19870708 |
|
| ITF | It: translation for a ep patent filed | ||
| NLV1 | Nl: lapsed or annulled due to failure to fulfill the requirements of art. 29p and 29m of the patents act | ||
| PLBE | No opposition filed within time limit |
Free format text: ORIGINAL CODE: 0009261 |
|
| STAA | Information on the status of an ep patent application or granted ep patent |
Free format text: STATUS: NO OPPOSITION FILED WITHIN TIME LIMIT |
|
| 26N | No opposition filed | ||
| PGFP | Annual fee paid to national office [announced via postgrant information from national office to epo] |
Ref country code: GB Payment date: 19921211 Year of fee payment: 9 |
|
| ITTA | It: last paid annual fee | ||
| PGFP | Annual fee paid to national office [announced via postgrant information from national office to epo] |
Ref country code: BE Payment date: 19930115 Year of fee payment: 9 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: GB Effective date: 19931217 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: BE Effective date: 19931231 |
|
| BERE | Be: lapsed |
Owner name: ELECTRICITE DE FRANCE SERVICE NATIONAL Effective date: 19931231 |
|
| GBPC | Gb: european patent ceased through non-payment of renewal fee |
Effective date: 19931217 |