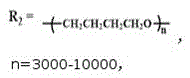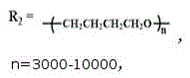CN115073701A - 一种阴离子型茜红素基水性聚氨酯荧光乳液及其制备方法 - Google Patents
一种阴离子型茜红素基水性聚氨酯荧光乳液及其制备方法 Download PDFInfo
- Publication number
- CN115073701A CN115073701A CN202210818399.9A CN202210818399A CN115073701A CN 115073701 A CN115073701 A CN 115073701A CN 202210818399 A CN202210818399 A CN 202210818399A CN 115073701 A CN115073701 A CN 115073701A
- Authority
- CN
- China
- Prior art keywords
- alizarin
- anionic
- emulsion
- waterborne polyurethane
- butanediol
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Pending
Links
- RGCKGOZRHPZPFP-UHFFFAOYSA-N alizarin Chemical compound C1=CC=C2C(=O)C3=C(O)C(O)=CC=C3C(=O)C2=C1 RGCKGOZRHPZPFP-UHFFFAOYSA-N 0.000 title claims abstract description 93
- 239000004814 polyurethane Substances 0.000 title claims abstract description 69
- 239000000839 emulsion Substances 0.000 title claims abstract description 68
- 229920002635 polyurethane Polymers 0.000 title claims abstract description 68
- 125000000129 anionic group Chemical group 0.000 title claims abstract description 36
- 238000002360 preparation method Methods 0.000 title abstract description 13
- 238000004945 emulsification Methods 0.000 title description 2
- LYCAIKOWRPUZTN-UHFFFAOYSA-N Ethylene glycol Chemical compound OCCO LYCAIKOWRPUZTN-UHFFFAOYSA-N 0.000 claims abstract description 42
- WERYXYBDKMZEQL-UHFFFAOYSA-N butane-1,4-diol Chemical compound OCCCCO WERYXYBDKMZEQL-UHFFFAOYSA-N 0.000 claims abstract description 42
- -1 polytetramethylene Polymers 0.000 claims abstract description 23
- 239000005058 Isophorone diisocyanate Substances 0.000 claims abstract description 22
- NIMLQBUJDJZYEJ-UHFFFAOYSA-N isophorone diisocyanate Chemical compound CC1(C)CC(N=C=O)CC(C)(CN=C=O)C1 NIMLQBUJDJZYEJ-UHFFFAOYSA-N 0.000 claims abstract description 22
- WGCNASOHLSPBMP-UHFFFAOYSA-N hydroxyacetaldehyde Natural products OCC=O WGCNASOHLSPBMP-UHFFFAOYSA-N 0.000 claims abstract description 21
- ZMXDDKWLCZADIW-UHFFFAOYSA-N N,N-Dimethylformamide Chemical compound CN(C)C=O ZMXDDKWLCZADIW-UHFFFAOYSA-N 0.000 claims abstract description 15
- XLYOFNOQVPJJNP-UHFFFAOYSA-N water Chemical compound O XLYOFNOQVPJJNP-UHFFFAOYSA-N 0.000 claims abstract description 13
- 239000000126 substance Substances 0.000 claims abstract description 11
- 239000003054 catalyst Substances 0.000 claims abstract description 8
- 239000008367 deionised water Substances 0.000 claims abstract description 7
- 229910021641 deionized water Inorganic materials 0.000 claims abstract description 7
- 239000002994 raw material Substances 0.000 claims abstract description 5
- 238000007334 copolymerization reaction Methods 0.000 claims abstract description 3
- 238000006243 chemical reaction Methods 0.000 claims description 14
- 238000003756 stirring Methods 0.000 claims description 12
- 238000001816 cooling Methods 0.000 claims description 10
- 238000010008 shearing Methods 0.000 claims description 8
- 238000000034 method Methods 0.000 claims description 6
- 239000000376 reactant Substances 0.000 claims description 6
- PTBDIHRZYDMNKB-UHFFFAOYSA-N 2,2-Bis(hydroxymethyl)propionic acid Chemical compound OCC(C)(CO)C(O)=O PTBDIHRZYDMNKB-UHFFFAOYSA-N 0.000 claims description 4
- HPQUMJNDQVOTAZ-UHFFFAOYSA-N 2,2-dihydroxypropanoic acid Chemical compound CC(O)(O)C(O)=O HPQUMJNDQVOTAZ-UHFFFAOYSA-N 0.000 claims description 4
- 238000001035 drying Methods 0.000 claims description 4
- 238000010438 heat treatment Methods 0.000 claims description 4
- 239000002808 molecular sieve Substances 0.000 claims description 4
- 239000012299 nitrogen atmosphere Substances 0.000 claims description 4
- 238000002203 pretreatment Methods 0.000 claims description 4
- URGAHOPLAPQHLN-UHFFFAOYSA-N sodium aluminosilicate Chemical compound [Na+].[Al+3].[O-][Si]([O-])=O.[O-][Si]([O-])=O URGAHOPLAPQHLN-UHFFFAOYSA-N 0.000 claims description 4
- ISKQADXMHQSTHK-UHFFFAOYSA-N [4-(aminomethyl)phenyl]methanamine Chemical compound NCC1=CC=C(CN)C=C1 ISKQADXMHQSTHK-UHFFFAOYSA-N 0.000 claims description 3
- KSBAEPSJVUENNK-UHFFFAOYSA-L tin(ii) 2-ethylhexanoate Chemical compound [Sn+2].CCCCC(CC)C([O-])=O.CCCCC(CC)C([O-])=O KSBAEPSJVUENNK-UHFFFAOYSA-L 0.000 claims description 3
- UKLDJPRMSDWDSL-UHFFFAOYSA-L [dibutyl(dodecanoyloxy)stannyl] dodecanoate Chemical compound CCCCCCCCCCCC(=O)O[Sn](CCCC)(CCCC)OC(=O)CCCCCCCCCCC UKLDJPRMSDWDSL-UHFFFAOYSA-L 0.000 claims description 2
- 239000012975 dibutyltin dilaurate Substances 0.000 claims description 2
- 239000012974 tin catalyst Substances 0.000 claims 1
- 239000003973 paint Substances 0.000 abstract description 5
- 238000001514 detection method Methods 0.000 abstract description 4
- 239000013013 elastic material Substances 0.000 abstract description 4
- 238000011161 development Methods 0.000 abstract description 3
- 239000003814 drug Substances 0.000 abstract description 3
- 229940079593 drug Drugs 0.000 abstract description 3
- HFVAFDPGUJEFBQ-UHFFFAOYSA-M alizarin red S Chemical compound [Na+].O=C1C2=CC=CC=C2C(=O)C2=C1C=C(S([O-])(=O)=O)C(O)=C2O HFVAFDPGUJEFBQ-UHFFFAOYSA-M 0.000 description 47
- 230000000052 comparative effect Effects 0.000 description 15
- UFHFLCQGNIYNRP-UHFFFAOYSA-N Hydrogen Chemical compound [H][H] UFHFLCQGNIYNRP-UHFFFAOYSA-N 0.000 description 10
- 239000001257 hydrogen Substances 0.000 description 10
- 229910052739 hydrogen Inorganic materials 0.000 description 10
- 238000010521 absorption reaction Methods 0.000 description 8
- 239000000975 dye Substances 0.000 description 6
- 229920000642 polymer Polymers 0.000 description 6
- 238000005033 Fourier transform infrared spectroscopy Methods 0.000 description 4
- 238000004458 analytical method Methods 0.000 description 4
- JQVDAXLFBXTEQA-UHFFFAOYSA-N dibutylamine Chemical compound CCCCNCCCC JQVDAXLFBXTEQA-UHFFFAOYSA-N 0.000 description 4
- 238000002329 infrared spectrum Methods 0.000 description 4
- 238000002371 ultraviolet--visible spectrum Methods 0.000 description 4
- RZVHIXYEVGDQDX-UHFFFAOYSA-N 9,10-anthraquinone Chemical group C1=CC=C2C(=O)C3=CC=CC=C3C(=O)C2=C1 RZVHIXYEVGDQDX-UHFFFAOYSA-N 0.000 description 3
- 239000004970 Chain extender Substances 0.000 description 3
- 230000000694 effects Effects 0.000 description 3
- 239000000178 monomer Substances 0.000 description 3
- 239000002904 solvent Substances 0.000 description 3
- 238000012360 testing method Methods 0.000 description 3
- 238000001291 vacuum drying Methods 0.000 description 3
- AFCARXCZXQIEQB-UHFFFAOYSA-N N-[3-oxo-3-(2,4,6,7-tetrahydrotriazolo[4,5-c]pyridin-5-yl)propyl]-2-[[3-(trifluoromethoxy)phenyl]methylamino]pyrimidine-5-carboxamide Chemical compound O=C(CCNC(=O)C=1C=NC(=NC=1)NCC1=CC(=CC=C1)OC(F)(F)F)N1CC2=C(CC1)NN=N2 AFCARXCZXQIEQB-UHFFFAOYSA-N 0.000 description 2
- PYKYMHQGRFAEBM-UHFFFAOYSA-N anthraquinone Natural products CCC(=O)c1c(O)c2C(=O)C3C(C=CC=C3O)C(=O)c2cc1CC(=O)OC PYKYMHQGRFAEBM-UHFFFAOYSA-N 0.000 description 2
- 150000004056 anthraquinones Chemical class 0.000 description 2
- CSCPPACGZOOCGX-WFGJKAKNSA-N deuterated acetone Substances [2H]C([2H])([2H])C(=O)C([2H])([2H])[2H] CSCPPACGZOOCGX-WFGJKAKNSA-N 0.000 description 2
- 238000010586 diagram Methods 0.000 description 2
- 238000001506 fluorescence spectroscopy Methods 0.000 description 2
- 125000001570 methylene group Chemical group [H]C([H])([*:1])[*:2] 0.000 description 2
- 230000003287 optical effect Effects 0.000 description 2
- 125000001997 phenyl group Chemical group [H]C1=C([H])C([H])=C(*)C([H])=C1[H] 0.000 description 2
- 238000002464 physical blending Methods 0.000 description 2
- 238000006862 quantum yield reaction Methods 0.000 description 2
- 238000001228 spectrum Methods 0.000 description 2
- 238000002076 thermal analysis method Methods 0.000 description 2
- 238000004448 titration Methods 0.000 description 2
- 238000002211 ultraviolet spectrum Methods 0.000 description 2
- AVWRKZWQTYIKIY-UHFFFAOYSA-N urea-1-carboxylic acid Chemical compound NC(=O)NC(O)=O AVWRKZWQTYIKIY-UHFFFAOYSA-N 0.000 description 2
- BJZYYSAMLOBSDY-QMMMGPOBSA-N (2s)-2-butoxybutan-1-ol Chemical compound CCCCO[C@@H](CC)CO BJZYYSAMLOBSDY-QMMMGPOBSA-N 0.000 description 1
- CSCPPACGZOOCGX-UHFFFAOYSA-N Acetone Chemical class CC(C)=O CSCPPACGZOOCGX-UHFFFAOYSA-N 0.000 description 1
- KXDHJXZQYSOELW-UHFFFAOYSA-M Carbamate Chemical compound NC([O-])=O KXDHJXZQYSOELW-UHFFFAOYSA-M 0.000 description 1
- 241000282414 Homo sapiens Species 0.000 description 1
- 101001007415 Homo sapiens LEM domain-containing protein 1 Proteins 0.000 description 1
- 101001126084 Homo sapiens Piwi-like protein 2 Proteins 0.000 description 1
- 101000666379 Homo sapiens Transcription factor Dp family member 3 Proteins 0.000 description 1
- 102100028300 LEM domain-containing protein 1 Human genes 0.000 description 1
- 238000005481 NMR spectroscopy Methods 0.000 description 1
- XBXFGOSIPGWNLZ-UHFFFAOYSA-N O=C1C=C(CC(C)(C)C1)C.N=C=O Chemical compound O=C1C=C(CC(C)(C)C1)C.N=C=O XBXFGOSIPGWNLZ-UHFFFAOYSA-N 0.000 description 1
- 102100029365 Piwi-like protein 2 Human genes 0.000 description 1
- 102100038129 Transcription factor Dp family member 3 Human genes 0.000 description 1
- 238000005299 abrasion Methods 0.000 description 1
- 239000000853 adhesive Substances 0.000 description 1
- 230000001070 adhesive effect Effects 0.000 description 1
- 150000001408 amides Chemical class 0.000 description 1
- 238000004873 anchoring Methods 0.000 description 1
- 230000002238 attenuated effect Effects 0.000 description 1
- 230000009286 beneficial effect Effects 0.000 description 1
- 230000015572 biosynthetic process Effects 0.000 description 1
- 238000006555 catalytic reaction Methods 0.000 description 1
- 238000012512 characterization method Methods 0.000 description 1
- 239000003795 chemical substances by application Substances 0.000 description 1
- 238000004587 chromatography analysis Methods 0.000 description 1
- 238000000576 coating method Methods 0.000 description 1
- 150000001875 compounds Chemical class 0.000 description 1
- 238000007906 compression Methods 0.000 description 1
- 230000021615 conjugation Effects 0.000 description 1
- 238000000354 decomposition reaction Methods 0.000 description 1
- 230000007547 defect Effects 0.000 description 1
- 238000007865 diluting Methods 0.000 description 1
- 238000002474 experimental method Methods 0.000 description 1
- 239000000835 fiber Substances 0.000 description 1
- GNBHRKFJIUUOQI-UHFFFAOYSA-N fluorescein Chemical compound O1C(=O)C2=CC=CC=C2C21C1=CC=C(O)C=C1OC1=CC(O)=CC=C21 GNBHRKFJIUUOQI-UHFFFAOYSA-N 0.000 description 1
- 238000002189 fluorescence spectrum Methods 0.000 description 1
- 239000007850 fluorescent dye Substances 0.000 description 1
- 239000006260 foam Substances 0.000 description 1
- 231100000086 high toxicity Toxicity 0.000 description 1
- 125000002887 hydroxy group Chemical group [H]O* 0.000 description 1
- 230000003993 interaction Effects 0.000 description 1
- 150000002500 ions Chemical class 0.000 description 1
- 239000004973 liquid crystal related substance Substances 0.000 description 1
- 239000000463 material Substances 0.000 description 1
- 239000011159 matrix material Substances 0.000 description 1
- 238000002156 mixing Methods 0.000 description 1
- 239000000203 mixture Substances 0.000 description 1
- 238000012986 modification Methods 0.000 description 1
- 230000004048 modification Effects 0.000 description 1
- 238000000655 nuclear magnetic resonance spectrum Methods 0.000 description 1
- 239000000049 pigment Substances 0.000 description 1
- 239000002861 polymer material Substances 0.000 description 1
- 229920005862 polyol Polymers 0.000 description 1
- 238000012545 processing Methods 0.000 description 1
- 238000003786 synthesis reaction Methods 0.000 description 1
- BFKJFAAPBSQJPD-UHFFFAOYSA-N tetrafluoroethene Chemical group FC(F)=C(F)F BFKJFAAPBSQJPD-UHFFFAOYSA-N 0.000 description 1
- CZDYPVPMEAXLPK-UHFFFAOYSA-N tetramethylsilane Chemical compound C[Si](C)(C)C CZDYPVPMEAXLPK-UHFFFAOYSA-N 0.000 description 1
- 238000002411 thermogravimetry Methods 0.000 description 1
- 229920002725 thermoplastic elastomer Polymers 0.000 description 1
- 238000000870 ultraviolet spectroscopy Methods 0.000 description 1
- 230000004580 weight loss Effects 0.000 description 1
Images
Classifications
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08G—MACROMOLECULAR COMPOUNDS OBTAINED OTHERWISE THAN BY REACTIONS ONLY INVOLVING UNSATURATED CARBON-TO-CARBON BONDS
- C08G18/00—Polymeric products of isocyanates or isothiocyanates
- C08G18/06—Polymeric products of isocyanates or isothiocyanates with compounds having active hydrogen
- C08G18/28—Polymeric products of isocyanates or isothiocyanates with compounds having active hydrogen characterised by the compounds used containing active hydrogen
- C08G18/65—Low-molecular-weight compounds having active hydrogen with high-molecular-weight compounds having active hydrogen
- C08G18/66—Compounds of groups C08G18/42, C08G18/48, or C08G18/52
- C08G18/6666—Compounds of group C08G18/48 or C08G18/52
- C08G18/6692—Compounds of group C08G18/48 or C08G18/52 with compounds of group C08G18/34
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08G—MACROMOLECULAR COMPOUNDS OBTAINED OTHERWISE THAN BY REACTIONS ONLY INVOLVING UNSATURATED CARBON-TO-CARBON BONDS
- C08G18/00—Polymeric products of isocyanates or isothiocyanates
- C08G18/06—Polymeric products of isocyanates or isothiocyanates with compounds having active hydrogen
- C08G18/28—Polymeric products of isocyanates or isothiocyanates with compounds having active hydrogen characterised by the compounds used containing active hydrogen
- C08G18/30—Low-molecular-weight compounds
- C08G18/32—Polyhydroxy compounds; Polyamines; Hydroxyamines
- C08G18/3203—Polyhydroxy compounds
- C08G18/3206—Polyhydroxy compounds aliphatic
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08G—MACROMOLECULAR COMPOUNDS OBTAINED OTHERWISE THAN BY REACTIONS ONLY INVOLVING UNSATURATED CARBON-TO-CARBON BONDS
- C08G18/00—Polymeric products of isocyanates or isothiocyanates
- C08G18/06—Polymeric products of isocyanates or isothiocyanates with compounds having active hydrogen
- C08G18/28—Polymeric products of isocyanates or isothiocyanates with compounds having active hydrogen characterised by the compounds used containing active hydrogen
- C08G18/30—Low-molecular-weight compounds
- C08G18/34—Carboxylic acids; Esters thereof with monohydroxyl compounds
- C08G18/348—Hydroxycarboxylic acids
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08G—MACROMOLECULAR COMPOUNDS OBTAINED OTHERWISE THAN BY REACTIONS ONLY INVOLVING UNSATURATED CARBON-TO-CARBON BONDS
- C08G18/00—Polymeric products of isocyanates or isothiocyanates
- C08G18/06—Polymeric products of isocyanates or isothiocyanates with compounds having active hydrogen
- C08G18/28—Polymeric products of isocyanates or isothiocyanates with compounds having active hydrogen characterised by the compounds used containing active hydrogen
- C08G18/30—Low-molecular-weight compounds
- C08G18/38—Low-molecular-weight compounds having heteroatoms other than oxygen
- C08G18/3855—Low-molecular-weight compounds having heteroatoms other than oxygen having sulfur
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08G—MACROMOLECULAR COMPOUNDS OBTAINED OTHERWISE THAN BY REACTIONS ONLY INVOLVING UNSATURATED CARBON-TO-CARBON BONDS
- C08G18/00—Polymeric products of isocyanates or isothiocyanates
- C08G18/06—Polymeric products of isocyanates or isothiocyanates with compounds having active hydrogen
- C08G18/28—Polymeric products of isocyanates or isothiocyanates with compounds having active hydrogen characterised by the compounds used containing active hydrogen
- C08G18/40—High-molecular-weight compounds
- C08G18/48—Polyethers
- C08G18/4854—Polyethers containing oxyalkylene groups having four carbon atoms in the alkylene group
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08G—MACROMOLECULAR COMPOUNDS OBTAINED OTHERWISE THAN BY REACTIONS ONLY INVOLVING UNSATURATED CARBON-TO-CARBON BONDS
- C08G18/00—Polymeric products of isocyanates or isothiocyanates
- C08G18/06—Polymeric products of isocyanates or isothiocyanates with compounds having active hydrogen
- C08G18/28—Polymeric products of isocyanates or isothiocyanates with compounds having active hydrogen characterised by the compounds used containing active hydrogen
- C08G18/65—Low-molecular-weight compounds having active hydrogen with high-molecular-weight compounds having active hydrogen
- C08G18/66—Compounds of groups C08G18/42, C08G18/48, or C08G18/52
- C08G18/6666—Compounds of group C08G18/48 or C08G18/52
- C08G18/667—Compounds of group C08G18/48 or C08G18/52 with compounds of group C08G18/32 or polyamines of C08G18/38
- C08G18/6674—Compounds of group C08G18/48 or C08G18/52 with compounds of group C08G18/32 or polyamines of C08G18/38 with compounds of group C08G18/3203
-
- C—CHEMISTRY; METALLURGY
- C09—DYES; PAINTS; POLISHES; NATURAL RESINS; ADHESIVES; COMPOSITIONS NOT OTHERWISE PROVIDED FOR; APPLICATIONS OF MATERIALS NOT OTHERWISE PROVIDED FOR
- C09K—MATERIALS FOR MISCELLANEOUS APPLICATIONS, NOT PROVIDED FOR ELSEWHERE
- C09K11/00—Luminescent, e.g. electroluminescent, chemiluminescent materials
- C09K11/02—Use of particular materials as binders, particle coatings or suspension media therefor
- C09K11/025—Use of particular materials as binders, particle coatings or suspension media therefor non-luminescent particle coatings or suspension media
-
- C—CHEMISTRY; METALLURGY
- C09—DYES; PAINTS; POLISHES; NATURAL RESINS; ADHESIVES; COMPOSITIONS NOT OTHERWISE PROVIDED FOR; APPLICATIONS OF MATERIALS NOT OTHERWISE PROVIDED FOR
- C09K—MATERIALS FOR MISCELLANEOUS APPLICATIONS, NOT PROVIDED FOR ELSEWHERE
- C09K11/00—Luminescent, e.g. electroluminescent, chemiluminescent materials
- C09K11/06—Luminescent, e.g. electroluminescent, chemiluminescent materials containing organic luminescent materials
-
- C—CHEMISTRY; METALLURGY
- C09—DYES; PAINTS; POLISHES; NATURAL RESINS; ADHESIVES; COMPOSITIONS NOT OTHERWISE PROVIDED FOR; APPLICATIONS OF MATERIALS NOT OTHERWISE PROVIDED FOR
- C09K—MATERIALS FOR MISCELLANEOUS APPLICATIONS, NOT PROVIDED FOR ELSEWHERE
- C09K2211/00—Chemical nature of organic luminescent or tenebrescent compounds
- C09K2211/14—Macromolecular compounds
- C09K2211/1408—Carbocyclic compounds
- C09K2211/1416—Condensed systems
Landscapes
- Chemical & Material Sciences (AREA)
- Organic Chemistry (AREA)
- Health & Medical Sciences (AREA)
- Chemical Kinetics & Catalysis (AREA)
- Medicinal Chemistry (AREA)
- Polymers & Plastics (AREA)
- Engineering & Computer Science (AREA)
- Materials Engineering (AREA)
- Polyurethanes Or Polyureas (AREA)
Abstract
本发明公开一种阴离子型茜红素基水性聚氨酯荧光乳液及其制备方法:利用异佛二酮二异氰酸酯、聚丁二醇与茜红素进行共聚反应,通过‑HNCOO‑化学键将茜红素引入聚氨酯分子链中;所述阴离子型茜红素基水性聚氨酯荧光乳液包括以下质量比的原料:异佛二酮二异氰酸酯、聚丁二醇、茜红素、N,N‑二甲基甲酰胺、1,4‑丁二醇、催化剂、去离子水的质量比=24:36:(1.36‑1.58):(20‑25):4:(0.030‑0.056):100。本发明产品与水有更好的相容性,且在稳定性以及机械性能上都得到显著提升,高效地解决了茜红素荧光产率低而导致发光效率低的问题,让荧光更好的释放,可在防伪标识、交通标志、弹性材料、生物显影、生化检测、药物示踪、荧光油墨、荧光涂料等方面广泛应用。
Description
技术领域
本发明属于高分子材料加工领域,具体涉及一种阴离子型茜红素基水性聚氨酯荧光乳液的制备方法。
背景技术
聚氨酯(PU)是满足现代社会高度多样化需求的最通用的高分子材料,如涂料、粘合剂、纤维、泡沫和热塑性弹性体。由于聚氨酯基染料聚合物在工业上有很大的应用前景,如光化学治疗、热压敏感记录、光记录、化学催化、液晶显示等高新领域,使人类对其需求与日俱增。目前,高分子聚合物染料的制备主要集中在将染料单体物理锚定在聚合物基质中,因此,导致染料分子易迁移,其色牢度、耐磨性、加工性能较差,毒性较高。例如,彩色聚氨酯主要由混合颜料和聚氨酯乳液通过物理共混的方式将二者混合,然而这种方法制出的产品颜色黯淡、亮度不足,染色能力和色牢度差,且耐磨性也较差。目前,利用小分子荧光剂作为扩链剂引入到水性聚氨酯主链及其产品的荧光性能的鲜有被相关文献及专利报道或记载,尚未被完整系统地研究。
发明内容
针对现有技术的不足之处,本发明的目的在于提供一种阴离子型茜红素基水性聚氨酯荧光乳液及其制备方法,通过将二佛异酮异氰酸酯和聚丁二醇和9,10-二氢-3,4-二羟基-9,10-二氧代-2-蒽磺酸单钠盐(茜红素)反应,制备红色荧光的茜红素基水性聚氨酯乳液,其具有稳定性高,在乳液或固化膜中都有较稳定的荧光强度,可以应用于防伪标识、交通标志、弹性材料、化学检测、荧光油墨、荧光涂料等领域。
本发明的技术方案概述如下:
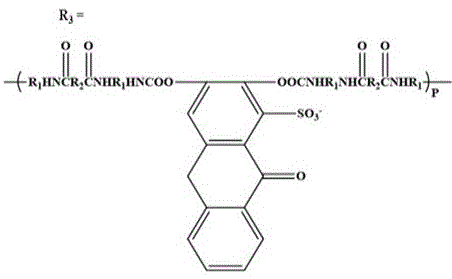
一种阴离子型茜红素基水性聚氨酯荧光乳液:利用异佛二酮二异氰酸酯、聚丁二醇与茜红素进行共聚反应,通过-HNCOO-化学键将茜红素引入聚氨酯分子链中;所述阴离子型茜红素基水性聚氨酯荧光乳液包括以下质量比的原料:异佛二酮二异氰酸酯、聚丁二醇、茜红素、N,N-二甲基甲酰胺、1,4-丁二醇、催化剂、去离子水的质量比=24: 36: (1.36-1.58): (20-25):4: (0.030-0.056):100;
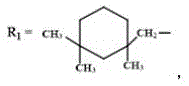
所述阴离子型茜红素基水性聚氨酯的化学式为:
优选的是,所述阴离子型茜红素基水性聚氨酯荧光乳液的原料还可包括2,2-二羟甲基丙酸。
优选的是,所述2,2-二羟甲基丙酸和1,4-丁二醇的质量比=4.74:4。
优选的是,所述催化剂为有机锡催化剂,包括辛酸亚锡、二月桂酸二丁基锡、二醋酸二丁基锡中的一种或多种。
一种阴离子型茜红素基水性聚氨酯荧光乳液的制备方法,包括以下步骤:S1:预处理异佛二酮二异氰酸酯、聚丁二醇及1,4-丁二醇;
S2:将茜红素加入N,N-二甲基甲酰胺中,搅拌溶解后,得茜红素溶液;
S3:向四口反应釜中加入异佛二酮二异氰酸酯、聚丁二醇和茜红素溶液,在氮气气氛下剧烈搅拌,再缓慢升温至70-100℃,滴加催化剂,搅拌反应至体系残留-NCO含量达到理论值为止;
S4:根据反应过程是否添加二羟基丙酸,该步骤分为以下两种情形:
将S3所得反应物冷却至60-80℃,在30 min内逐滴加入1,4-丁二醇后,50-70℃反应2h,得反应后乳液;
或将S3所得反应物冷却至60-80℃,在30 min内逐滴加入1,4-丁二醇后,再继续加入二羟基丙酸,50-70℃反应2h,得反应后乳液;
S5:将S4所得反应后乳液冷却至30-50℃,加入去离子水,再以3000 rpm的剪切速率持续剪切30 min,即得所述阴离子型茜红素基水性聚氨酯荧光乳液。
优选的是,所述异佛二酮二异氰酸酯、聚丁二醇的预处理方法为:将异佛二酮二异氰酸酯、聚丁二醇在60-100℃下真空干燥30min。
优选的是,所述1,4-丁二醇的预处理方法为:将1,4-丁二醇在4Å分子筛中干燥24h。
本发明的有益效果:
1、本发明以茜红素作为聚氨酯的扩链剂,通过共价键将茜红素引入到聚氨酯主链上,制备水性聚氨酯荧光乳液(AH-WPUS),其中AH分子中含有Na+,使聚氨酯聚合物与水有更好的相容性,且在稳定性以及机械性能上都得到显著提升。
2、本发明将茜红素小分子荧光单体通过-HNCOO-化学键形式键合到聚氨酯分子链中,高效地解决了茜红素荧光产率低而导致发光效率低的问题,让荧光更好的释放。一方面,聚氨酯分子链可以有效地限制茜红素分子的运动来确保茜红素荧光分子以发光的形式释放能量,大大的提高量子产率;另一方面,提高了茜红素的加工性和稳定性。
3、本发明制出的阴离子型茜红素基水性聚氨酯荧光乳液具有较强的荧光效应,可在防伪标识、交通标志、弹性材料、生物显影、生化检测、药物示踪、荧光油墨、荧光涂料等方面得到应用,具有非常广阔的市场前景。
4、本发明制备方法步骤少、反应条件温和、成本低,制备的产品稳定性好、荧光性能优异。
附图说明
图1为本发明阴离子型茜红素基水性聚氨酯荧光乳液的制备方法流程图;
图2为本发明阴离子型茜红素基水性聚氨酯荧光乳液制备方法的合成路线图;
图3为实施例1-2制出的阴离子型茜红素基水性聚氨酯荧光乳液及对比例1中的茜红素荧光基团和水性聚氨酯乳液的红外光谱图(n=3000-10000,p=5000-12000,r=10000-20000);
图4为实施例1-2制出的阴离子型茜红素基水性聚氨酯荧光乳液的核磁氢谱图;
图5为实施例1-2制出的阴离子型茜红素基水性聚氨酯荧光乳液、对比例1中的水性聚氨酯乳液及所制出的混合乳液的紫外光谱图;
图6为实施例2制出的阴离子型茜红素基水性聚氨酯荧光乳液及对比例1中的茜红素荧光基团的荧光发射图。
具体实施方式
下面结合实施例对本发明做进一步的详细说明,以令本领域技术人员参照说明书文字能够据以实施。
实施例1
一种阴离子型茜红素基水性聚氨酯荧光乳液的制备方法,包括以下步骤:
S1:预处理异佛二酮二异氰酸酯、聚丁二醇:将24g异佛二酮二异氰酸酯、36g聚丁二醇在60℃下真空干燥30min;
预处理1,4-丁二醇:将4g的1,4-丁二醇在4 Å分子筛中干燥24h;
S2:将1.36g茜红素加入20gN,N-二甲基甲酰胺中,搅拌溶解后,得茜红素溶液;
S3:向四口反应釜中加入S1所得异佛二酮二异氰酸酯、聚丁二醇和S2所得茜红素溶液,在氮气气氛下剧烈搅拌,再缓慢升温至70℃,滴加0.030g辛酸亚锡,搅拌反应至体系残留-NCO含量达到理论值(通过标准的二正丁胺滴定法测定)为止;
S4:将S3所得反应物冷却至60℃,在30 min内逐滴加入S1所得1,4-丁二醇,再继续加入4.74g二羟基丙酸后,50℃反应2h,得反应后乳液;
S5:将S4所得反应后乳液冷却至30℃,加入100g去离子水,再以3000 rpm的剪切速率持续剪切30 min,即得所述阴离子型茜红素基水性聚氨酯荧光乳液(记作AH-WPU1)。
实施例2
一种阴离子型茜红素基水性聚氨酯荧光乳液的制备方法,包括以下步骤:
S1:预处理异佛二酮二异氰酸酯、聚丁二醇:将24g异佛二酮二异氰酸酯、36g聚丁二醇在100℃下真空干燥30min;
预处理1,4-丁二醇:将4g的1,4-丁二醇在4 Å分子筛中干燥24h;
S2:将1.58g茜红素加入20gN,N-二甲基甲酰胺中,搅拌溶解后,得茜红素溶液;
S3:向四口反应釜中加入S1所得异佛二酮二异氰酸酯、聚丁二醇和S2所得茜红素溶液,在氮气气氛下剧烈搅拌,再缓慢升温至100℃,滴加0.056g二醋酸二丁基锡,搅拌反应至体系残留-NCO含量达到理论值(通过标准的二正丁胺滴定法测定)为止;
S4:将S3所得反应物冷却至80℃,在30 min内逐滴加入S1所得1,4-丁二醇,70℃反应2h,得反应后乳液;
S5:将S4所得反应后乳液冷却至50℃,加入100g去离子水,再以3000 rpm的剪切速率持续剪切30 min,即得所述阴离子型茜红素基水性聚氨酯荧光乳液(记作AH-WPU2)。
对比例1为水性聚氨酯乳液和茜红素的共混物,其制备方法与实施例2相同,区别在于:在S3步骤不加入茜红素溶液,直至S5步骤制出水性聚氨酯乳液(记作WPU)再与茜红素溶液(记作AH)进行物理混合,即得茜红素与水性聚氨酯混合乳液(记作AH+WPU)。
对实施例1-2及对比例1所制备出水性聚氨酯乳液进行性能表征及测试
1、实验仪器:FTIR傅立叶转换红外线光谱仪(BrukerAVANCE III 4OOM,美国布鲁克公司)、热分析仪(STA-409PC型,德国耐驰公司)、紫外光谱分析(5500PC型,上海元析仪器有限公司)、荧光光谱分析(FL-4700型,美国Agilent公司)。
2、实验方法
乳液成膜过程:将乳液倾倒在四氟乙烯板上分别在环境温度和真空干燥箱中除去水分,除尽即得薄膜。
红外光谱(FT-IR):使用衰减全发射(ATR)方式,直接用成膜样品进行测试,测定样品的红外谱图。
核磁共振氢谱分析(1H-NMR):以四甲基硅烷(TMS)为标准物,氘代丙酮(Acetone-D6)为溶剂。
紫外可见光谱(UV-Vis):在400-800nm的波长范围的UV-5500PC装置上在获得。以水作为溶剂,将乳液浓度稀释为1 ×10-3mol/L。
荧光光谱(FL4700),以水作为溶剂将乳液梯度稀释到1×10-3-1×10-5mol/L,在400-800 nm下获得,出入口狭缝都设置成5nm,电压为400V。
热重分析测试:样品在80℃干燥固化2h。在25℃-600 ℃的温度变化范围内,对薄膜进行热失重分析则用TGA仪器进行测试,控制以10℃/min的升温速率。
3、实验结果:
(1)FTIR分析
图3为实施例1-2制出的AH-WPU乳液及对比例1中的AH基团和WPU乳液的红外光谱图,由图3可知,典型的脲基甲酸酯中C=O的特征吸收峰在1644 cm-1出现。同时,典型的羟基吸收峰和AH分子结构中3400至3500 cm-1之间的峰值NCO吸收峰在所有AH-WPU中均消失,表明AH中的OH和IPDI中的NCO已完全反应,AH与聚氨酯共价连接。此外,聚氨酯的特征吸收峰位于3350cm-1(νN-H),2953 cm-1和2855 cm-1(νCH2和νCH3),1710 cm-1(νC= O),1239 cm-1(νC–O氨基甲酸酯)和1111cm-1(PTMG中的νC–O–C),这些特征基团的出现表明AH-WPU已经成功合成。
(2)1H-NMR分析
图4为实施例1-2制出的AH-WPU乳液核磁氢谱图,由图4可知,1.05-0.95ppm来自IPDI分子结构上的氢,2.50-3.30 ppm来自PTMG多元醇上的氢,3.90 ppm大概为形成的酰胺上的氢,蒽醌结构上有苯环上的氢和一个亚甲基的氢的化学位移在谱图上的6.90-8.50ppm是典型的蒽醌结构上的氢,从核磁共振氢谱上可以得知AH已经与聚氨酯分子链发生了化学反应。
(3)UV-Vis分析
图5为实施例1-2制出的AH-WPU乳液及对比例1中的WPU乳液及所制出的混合乳液的紫外光谱图,由图5可知,WPU在UV-vis光谱中几乎没有吸收。AH-WPU显示两个与蒽醌单元有关的强吸收带,中心在683nm和542 nm。AH-WPU的吸收强度随AH含量的增加而增加。与AH+WPU相比,可以清楚地发现AH-WPU的最大吸收波长从543nm右移动到625nm的大概发生了82nm的偏移,这进一步证实了AH被共价固定在聚氨酯链。这可能是由于AH的蒽醌和WPU中的脲基甲酸酯的共轭作用,以及AH-WPU中的偶极分子与离子之间的相互作用,从而削弱了AH中的蒽醌的供电子能力。
(4)AH和AH-WPU的荧光光谱分析
图6为实施例2制出的AH-WPU乳液及对比例1中的AH基团的荧光发射图,由图6可知:实施例2及对比例1样品都有一个发射波长,且样品在380nm附近自然光下激发,且在实施例2加入AH后,荧光强度得到提高,是因为在AH-WPU中,整个化合物的结构含有较多的苯环结构而使得整个结构难以旋转,是一个典型的刚性结构,分子不易旋转振动,就很难与其他的分子发生碰撞使得能量大多以光的能量释放,发射荧光的强度就越高,但是同时刚性结构的物质也会存在极大的位阻,整个分子结构很难在一个平面上,导致共轭效应形成蓝移现象,因此,在光油墨、荧光涂料等领域有着广阔的市场前景。
(5)AH-WPU薄膜的热分析
表1为实施例1-2和对比例1的TG数据表
表1:
样品T10%/°CT30%/°CT50%/°CT80%/°C对比例1/AH+WPU260280300380实施例1/AH-WPU1262297322390实施例2/AH-WPU2268306340406
T10%、T30%、T50%、T80%分别表示样品失去10%质量、30%质量、50%质量、80%质量时对应的温度。
由上表可知,在AH量的增加下,样品的分解温度都有所增加,热稳定性也得到改善。
(6)AH-WPU薄膜的力学性能
表2为实施例1-2和对比例1的TG力学性能数据表
表2:
样品抗拉强度(MPa)断裂伸长率(%)对比例1/AH+WPU0.65178实施例1/AH-WPU11.25265实施例2/AH-WPU21.85259
由表2可知,相比于对比例1,实施例1-2的成膜样品的抗拉强度和断裂伸长率都得到了显著提升,因此,相比于对比例1物理共混的方式,通过共价键将茜红素荧光素键合到聚氨酯分子链中,能明显改善力学性能。
实施例1-2以茜红素作为聚氨酯的扩链剂,通过共价键将茜红素引入到聚氨酯主链上,制备水性聚氨酯荧光乳液(AH-WPUS),其中AH分子中含有Na+,使聚氨酯聚合物与水有更好的相容性,且在稳定性以及机械性能上都得到显著提升。
实施例1-2将茜红素小分子荧光单体通过-HNCOO-化学键形式键合到聚氨酯分子链中,高效地解决了茜红素荧光产率低而导致发光效率低的问题,让荧光更好的释放。一方面,聚氨酯分子链可以有效地限制茜红素分子的运动来确保茜红素荧光分子以发光的形式释放能量,大大的提高量子产率;另一方面,提高了茜红素的加工性和稳定性。
实施例1-2制出的阴离子型茜红素基水性聚氨酯荧光乳液具有较强的荧光效应,可在防伪标识、交通标志、弹性材料、生物显影、生化检测、药物示踪、荧光油墨、荧光涂料等方面得到应用,具有非常广阔的市场前景。
实施例1-2制备方法步骤少、反应条件温和、成本低,制备的产品稳定性好、荧光性能优异。
尽管本发明的实施方案已公开如上,但其并不仅仅限于说明书和实施方式中所列运用,它完全可以被适用于各种适合本发明的领域,对于熟悉本领域的人员而言,可容易地实现另外的修改,因此在不背离权利要求及等同范围所限定的一般概念下,本发明并不限于特定的细节。
Claims (7)
2.根据权利要求1所述的一种阴离子型茜红素基水性聚氨酯荧光乳液,其特征在于,所述阴离子型茜红素基水性聚氨酯荧光乳液的原料还可包括2,2-二羟甲基丙酸。
3.根据权利要求2所述的一种阴离子型茜红素基水性聚氨酯荧光乳液,其特征在于,所述2,2-二羟甲基丙酸和1,4-丁二醇的质量比=4.74:4。
4.根据权利要求1所述的一种阴离子型茜红素基水性聚氨酯荧光乳液,其特征在于,所述催化剂为有机锡催化剂,包括辛酸亚锡、二月桂酸二丁基锡、二醋酸二丁基锡中的一种或多种。
5.如权利要求1-4任一项所述的一种阴离子型茜红素基水性聚氨酯荧光乳液的制备方法,其特征在于,包括以下步骤:
S1:预处理异佛二酮二异氰酸酯、聚丁二醇及1,4-丁二醇;
S2:将茜红素加入N,N-二甲基甲酰胺中,搅拌溶解后,得茜红素溶液;
S3:向四口反应釜中加入异佛二酮二异氰酸酯、聚丁二醇和茜红素溶液,在氮气气氛下剧烈搅拌,再缓慢升温至70-100℃,滴加催化剂,搅拌反应至体系残留-NCO含量达到理论值为止;
S4:将S3所得反应物冷却至60-80℃,在30 min内逐滴加入1,4-丁二醇后,50-70℃反应2h,得反应后乳液;
或将S3所得反应物冷却至60-80℃,在30 min内逐滴加入1,4-丁二醇后,再继续加入二羟基丙酸,50-70℃反应2h,得反应后乳液;
S5:将S4所得反应后乳液冷却至30-50℃,加入去离子水,再以3000 rpm的剪切速率持续剪切30 min,即得所述阴离子型茜红素基水性聚氨酯荧光乳液。
6.根据权利要求5所述的的一种阴离子型茜红素基水性聚氨酯荧光乳液的制备方法,其特征在于,所述异佛二酮二异氰酸酯、聚丁二醇的预处理方法为:将异佛二酮二异氰酸酯、聚丁二醇在60-100℃下真空干燥30min。
7.根据权利要求5所述的的一种阴离子型茜红素基水性聚氨酯荧光乳液的制备方法,其特征在于,所述1,4-丁二醇的预处理方法为:将1,4-丁二醇在4 Å分子筛中干燥24h。
Priority Applications (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| CN202210818399.9A CN115073701A (zh) | 2022-07-13 | 2022-07-13 | 一种阴离子型茜红素基水性聚氨酯荧光乳液及其制备方法 |
Applications Claiming Priority (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| CN202210818399.9A CN115073701A (zh) | 2022-07-13 | 2022-07-13 | 一种阴离子型茜红素基水性聚氨酯荧光乳液及其制备方法 |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| CN115073701A true CN115073701A (zh) | 2022-09-20 |
Family
ID=83259454
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| CN202210818399.9A Pending CN115073701A (zh) | 2022-07-13 | 2022-07-13 | 一种阴离子型茜红素基水性聚氨酯荧光乳液及其制备方法 |
Country Status (1)
| Country | Link |
|---|---|
| CN (1) | CN115073701A (zh) |
Citations (5)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN102675587A (zh) * | 2012-05-22 | 2012-09-19 | 长春工业大学 | 环境pH响应性聚氨酯荧光发光医用材料的制备 |
| CN103172829A (zh) * | 2013-03-21 | 2013-06-26 | 中国科学技术大学 | 基于二异氰酸酯中发色团的荧光型水性聚氨酯乳液的制法 |
| US20190144594A1 (en) * | 2016-12-26 | 2019-05-16 | Public University Corporation Yokohama City University | Fluorescent Resin Composition, Molded Object and Medical Device, and Method for Producing Fluorescent Resin Composition |
| CN110606928A (zh) * | 2019-08-28 | 2019-12-24 | 齐鲁工业大学 | 一种基于咔唑的荧光水性聚氨酯的合成方法 |
| CN114075320A (zh) * | 2021-06-18 | 2022-02-22 | 四川大学 | 一种发光颜色可调的荧光水性聚氨酯及其制备方法 |
-
2022
- 2022-07-13 CN CN202210818399.9A patent/CN115073701A/zh active Pending
Patent Citations (5)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN102675587A (zh) * | 2012-05-22 | 2012-09-19 | 长春工业大学 | 环境pH响应性聚氨酯荧光发光医用材料的制备 |
| CN103172829A (zh) * | 2013-03-21 | 2013-06-26 | 中国科学技术大学 | 基于二异氰酸酯中发色团的荧光型水性聚氨酯乳液的制法 |
| US20190144594A1 (en) * | 2016-12-26 | 2019-05-16 | Public University Corporation Yokohama City University | Fluorescent Resin Composition, Molded Object and Medical Device, and Method for Producing Fluorescent Resin Composition |
| CN110606928A (zh) * | 2019-08-28 | 2019-12-24 | 齐鲁工业大学 | 一种基于咔唑的荧光水性聚氨酯的合成方法 |
| CN114075320A (zh) * | 2021-06-18 | 2022-02-22 | 四川大学 | 一种发光颜色可调的荧光水性聚氨酯及其制备方法 |
Non-Patent Citations (1)
| Title |
|---|
| 李明君: ""绿色、红色和紫色水性聚氨酯染料的合成及性能研究"", 《中国优秀硕士学位论文全文数据库 工程科技I辑》, no. 02, pages 1 - 2 * |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| Fang et al. | Light-and heat-triggered polyurethane based on dihydroxyl anthracene derivatives for self-healing applications | |
| Xu et al. | Synthesis and characterization of hyperbranched polyurethane acrylates used as UV curable oligomers for coatings | |
| CN103172829B (zh) | 基于二异氰酸酯中发色团的荧光型水性聚氨酯乳液的制法 | |
| Mao et al. | Synthesis of polymeric dyes based on UV curable multifunctional waterborne polyurethane for textile coating | |
| Liu et al. | Stiff UV-Curable self-healing coating based on double reversible networks containing diels-alder cross-linking and hydrogen bonds | |
| CN101235195B (zh) | 阳离子水性聚氨酯/丙烯酸酯复合乳液及其制备方法 | |
| CN103254396B (zh) | 一种基于二元醇中发色团的荧光型水性聚氨酯及其制备方法 | |
| Hu et al. | Synthesis and characterization of novel renewable castor oil-based UV-curable polyfunctional polyurethane acrylate | |
| Xu et al. | Polyurethane modified epoxy acrylate resins containing ε-caprolactone unit | |
| Li et al. | High concentration acid-induced discoloration polymeric dyes fabricated with UV-curable azobenzene-lignin-based waterborne polyurethane | |
| Zhang et al. | A self-colored waterborne polyurethane film with natural curcumin as a chain extender and excellent UV-Absorbing properties | |
| Xu et al. | Synthesis of transparent covalently self-colored polyurethane based on anthraquinone chromophore chain extenders | |
| Mao et al. | Synthesis of blocked and branched waterborne polyurethanes for pigment printing applications | |
| Chen et al. | Fluorescent self-healing waterborne polyurethane based on naphthalimide derivatives and its application in anti-counterfeiting | |
| CN115960331B (zh) | 一种阻燃和紫外屏蔽水性聚氨酯及其制备方法 | |
| Ma et al. | Synthesis and properties of photocurable polyurethane acrylate for textile artificial leather | |
| Yan et al. | Thiourethane thermoset coatings from bio‐based thiols | |
| CN105601567A (zh) | 荧光染料单体、共聚型荧光聚氨酯乳液及荧光聚氨酯-丙烯酸酯共聚物乳液及其制备方法 | |
| Hu et al. | A photochromic waterborne polyurethane-based dye with chemically fixed azobenzene groups | |
| CN105949425B (zh) | 一种聚氨酯基黑色高分子染料、制备方法及应用 | |
| CN106832175A (zh) | 一种基于咔唑衍生物的双羟基荧光扩链剂及其制备与应用 | |
| Muroi et al. | Self-healing thiol-ene networks based on cyclodextrin-adamantane host-guest interactions | |
| CN107556454B (zh) | 一种水性聚氨酯的制备方法、内交联聚氨酯-丙烯酸酯及其制备方法 | |
| Liao et al. | Effect of adding curcumin on the characterization of castor oil-based waterborne polyurethane coating | |
| CN115926106B (zh) | 一种反应型聚氨酯阴离子乳化剂及其制备方法和应用 |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| PB01 | Publication | ||
| PB01 | Publication | ||
| SE01 | Entry into force of request for substantive examination | ||
| SE01 | Entry into force of request for substantive examination | ||
| RJ01 | Rejection of invention patent application after publication |
Application publication date: 20220920 |