CN111902137A - 增强5-羟色氨酸的生物利用度的组合物和方法 - Google Patents
增强5-羟色氨酸的生物利用度的组合物和方法 Download PDFInfo
- Publication number
- CN111902137A CN111902137A CN201980021717.5A CN201980021717A CN111902137A CN 111902137 A CN111902137 A CN 111902137A CN 201980021717 A CN201980021717 A CN 201980021717A CN 111902137 A CN111902137 A CN 111902137A
- Authority
- CN
- China
- Prior art keywords
- htp
- carbidopa
- low dose
- formulation
- hydroxytryptamine
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Pending
Links
- LDCYZAJDBXYCGN-VIFPVBQESA-N 5-hydroxy-L-tryptophan Chemical compound C1=C(O)C=C2C(C[C@H](N)C(O)=O)=CNC2=C1 LDCYZAJDBXYCGN-VIFPVBQESA-N 0.000 title claims abstract description 193
- 238000000034 method Methods 0.000 title claims abstract description 44
- 239000000203 mixture Substances 0.000 title claims abstract description 42
- 230000002708 enhancing effect Effects 0.000 title claims description 10
- LDCYZAJDBXYCGN-UHFFFAOYSA-N oxitriptan Natural products C1=C(O)C=C2C(CC(N)C(O)=O)=CNC2=C1 LDCYZAJDBXYCGN-UHFFFAOYSA-N 0.000 title abstract description 174
- 229940000681 5-hydroxytryptophan Drugs 0.000 title abstract description 172
- 229960004205 carbidopa Drugs 0.000 claims abstract description 133
- TZFNLOMSOLWIDK-JTQLQIEISA-N carbidopa (anhydrous) Chemical compound NN[C@@](C(O)=O)(C)CC1=CC=C(O)C(O)=C1 TZFNLOMSOLWIDK-JTQLQIEISA-N 0.000 claims abstract description 132
- 238000009472 formulation Methods 0.000 claims abstract description 40
- 238000013268 sustained release Methods 0.000 claims abstract description 25
- 239000012730 sustained-release form Substances 0.000 claims abstract description 25
- 239000008194 pharmaceutical composition Substances 0.000 claims abstract description 12
- QZAYGJVTTNCVMB-UHFFFAOYSA-N serotonin Chemical compound C1=C(O)C=C2C(CCN)=CNC2=C1 QZAYGJVTTNCVMB-UHFFFAOYSA-N 0.000 claims description 199
- 238000011282 treatment Methods 0.000 claims description 39
- 239000002552 dosage form Substances 0.000 claims description 23
- 241000282414 Homo sapiens Species 0.000 claims description 20
- 239000003623 enhancer Substances 0.000 claims description 13
- 239000003826 tablet Substances 0.000 claims description 13
- 230000015572 biosynthetic process Effects 0.000 claims description 12
- 239000003814 drug Substances 0.000 claims description 11
- 239000003112 inhibitor Substances 0.000 claims description 11
- 239000002775 capsule Substances 0.000 claims description 10
- 230000036470 plasma concentration Effects 0.000 claims description 10
- 239000002243 precursor Substances 0.000 claims description 9
- 239000012729 immediate-release (IR) formulation Substances 0.000 claims description 8
- 229940124834 selective serotonin reuptake inhibitor Drugs 0.000 claims description 7
- 208000021384 Obsessive-Compulsive disease Diseases 0.000 claims description 6
- 206010042458 Suicidal ideation Diseases 0.000 claims description 6
- CGIGDMFJXJATDK-UHFFFAOYSA-N indomethacin Chemical compound CC1=C(CC(O)=O)C2=CC(OC)=CC=C2N1C(=O)C1=CC=C(Cl)C=C1 CGIGDMFJXJATDK-UHFFFAOYSA-N 0.000 claims description 6
- 238000009097 single-agent therapy Methods 0.000 claims description 6
- 208000019901 Anxiety disease Diseases 0.000 claims description 5
- 208000018737 Parkinson disease Diseases 0.000 claims description 5
- 230000005856 abnormality Effects 0.000 claims description 5
- 230000036651 mood Effects 0.000 claims description 5
- 239000012896 selective serotonin reuptake inhibitor Substances 0.000 claims description 5
- 208000024827 Alzheimer disease Diseases 0.000 claims description 4
- 229940123685 Monoamine oxidase inhibitor Drugs 0.000 claims description 4
- 230000036506 anxiety Effects 0.000 claims description 4
- 239000002899 monoamine oxidase inhibitor Substances 0.000 claims description 4
- 150000003839 salts Chemical class 0.000 claims description 4
- 239000000952 serotonin receptor agonist Substances 0.000 claims description 4
- 239000007909 solid dosage form Substances 0.000 claims description 4
- AHOUBRCZNHFOSL-YOEHRIQHSA-N (+)-Casbol Chemical compound C1=CC(F)=CC=C1[C@H]1[C@H](COC=2C=C3OCOC3=CC=2)CNCC1 AHOUBRCZNHFOSL-YOEHRIQHSA-N 0.000 claims description 3
- WSEQXVZVJXJVFP-HXUWFJFHSA-N (R)-citalopram Chemical compound C1([C@@]2(C3=CC=C(C=C3CO2)C#N)CCCN(C)C)=CC=C(F)C=C1 WSEQXVZVJXJVFP-HXUWFJFHSA-N 0.000 claims description 3
- RTHCYVBBDHJXIQ-MRXNPFEDSA-N (R)-fluoxetine Chemical compound O([C@H](CCNC)C=1C=CC=CC=1)C1=CC=C(C(F)(F)F)C=C1 RTHCYVBBDHJXIQ-MRXNPFEDSA-N 0.000 claims description 3
- KWTSXDURSIMDCE-QMMMGPOBSA-N (S)-amphetamine Chemical compound C[C@H](N)CC1=CC=CC=C1 KWTSXDURSIMDCE-QMMMGPOBSA-N 0.000 claims description 3
- 206010003591 Ataxia Diseases 0.000 claims description 3
- 206010003805 Autism Diseases 0.000 claims description 3
- 208000020706 Autistic disease Diseases 0.000 claims description 3
- 208000001640 Fibromyalgia Diseases 0.000 claims description 3
- 208000030990 Impulse-control disease Diseases 0.000 claims description 3
- 208000019695 Migraine disease Diseases 0.000 claims description 3
- 208000012902 Nervous system disease Diseases 0.000 claims description 3
- AHOUBRCZNHFOSL-UHFFFAOYSA-N Paroxetine hydrochloride Natural products C1=CC(F)=CC=C1C1C(COC=2C=C3OCOC3=CC=2)CNCC1 AHOUBRCZNHFOSL-UHFFFAOYSA-N 0.000 claims description 3
- 201000009916 Postpartum depression Diseases 0.000 claims description 3
- 208000027030 Premenstrual dysphoric disease Diseases 0.000 claims description 3
- 206010041250 Social phobia Diseases 0.000 claims description 3
- 229940123445 Tricyclic antidepressant Drugs 0.000 claims description 3
- 229940025084 amphetamine Drugs 0.000 claims description 3
- 208000030963 borderline personality disease Diseases 0.000 claims description 3
- 229960001653 citalopram Drugs 0.000 claims description 3
- USRHYDPUVLEVMC-FQEVSTJZSA-N dapoxetine Chemical compound C1([C@H](CCOC=2C3=CC=CC=C3C=CC=2)N(C)C)=CC=CC=C1 USRHYDPUVLEVMC-FQEVSTJZSA-N 0.000 claims description 3
- 229960005217 dapoxetine Drugs 0.000 claims description 3
- WSEQXVZVJXJVFP-FQEVSTJZSA-N escitalopram Chemical compound C1([C@]2(C3=CC=C(C=C3CO2)C#N)CCCN(C)C)=CC=C(F)C=C1 WSEQXVZVJXJVFP-FQEVSTJZSA-N 0.000 claims description 3
- 229960004341 escitalopram Drugs 0.000 claims description 3
- 229960002464 fluoxetine Drugs 0.000 claims description 3
- 229960004038 fluvoxamine Drugs 0.000 claims description 3
- CJOFXWAVKWHTFT-XSFVSMFZSA-N fluvoxamine Chemical compound COCCCC\C(=N/OCCN)C1=CC=C(C(F)(F)F)C=C1 CJOFXWAVKWHTFT-XSFVSMFZSA-N 0.000 claims description 3
- 229960000905 indomethacin Drugs 0.000 claims description 3
- 235000012054 meals Nutrition 0.000 claims description 3
- 206010027599 migraine Diseases 0.000 claims description 3
- 239000002767 noradrenalin uptake inhibitor Substances 0.000 claims description 3
- 229940127221 norepinephrine reuptake inhibitor Drugs 0.000 claims description 3
- 229960002296 paroxetine Drugs 0.000 claims description 3
- 208000028173 post-traumatic stress disease Diseases 0.000 claims description 3
- 239000000651 prodrug Substances 0.000 claims description 3
- 229940002612 prodrug Drugs 0.000 claims description 3
- 208000020016 psychiatric disease Diseases 0.000 claims description 3
- 229960002073 sertraline Drugs 0.000 claims description 3
- VGKDLMBJGBXTGI-SJCJKPOMSA-N sertraline Chemical compound C1([C@@H]2CC[C@@H](C3=CC=CC=C32)NC)=CC=C(Cl)C(Cl)=C1 VGKDLMBJGBXTGI-SJCJKPOMSA-N 0.000 claims description 3
- 239000003029 tricyclic antidepressant agent Substances 0.000 claims description 3
- YQNWZWMKLDQSAC-UHFFFAOYSA-N vortioxetine Chemical compound CC1=CC(C)=CC=C1SC1=CC=CC=C1N1CCNCC1 YQNWZWMKLDQSAC-UHFFFAOYSA-N 0.000 claims description 3
- 229960002263 vortioxetine Drugs 0.000 claims description 3
- 208000006096 Attention Deficit Disorder with Hyperactivity Diseases 0.000 claims description 2
- 208000036864 Attention deficit/hyperactivity disease Diseases 0.000 claims description 2
- 208000026331 Disruptive, Impulse Control, and Conduct disease Diseases 0.000 claims description 2
- 208000011688 Generalised anxiety disease Diseases 0.000 claims description 2
- 208000009668 Neurobehavioral Manifestations Diseases 0.000 claims description 2
- 208000028552 Treatment-Resistant Depressive disease Diseases 0.000 claims description 2
- 238000011360 adjunctive therapy Methods 0.000 claims description 2
- 208000013404 behavioral symptom Diseases 0.000 claims description 2
- 230000001149 cognitive effect Effects 0.000 claims description 2
- 208000029364 generalized anxiety disease Diseases 0.000 claims description 2
- 229940079322 interferon Drugs 0.000 claims description 2
- 208000024714 major depressive disease Diseases 0.000 claims description 2
- 238000004519 manufacturing process Methods 0.000 claims description 2
- 208000019906 panic disease Diseases 0.000 claims description 2
- 238000011084 recovery Methods 0.000 claims description 2
- 239000006215 rectal suppository Substances 0.000 claims description 2
- 229940100618 rectal suppository Drugs 0.000 claims description 2
- 208000019116 sleep disease Diseases 0.000 claims description 2
- 239000003174 triple reuptake inhibitor Substances 0.000 claims description 2
- 229960003740 vilazodone Drugs 0.000 claims description 2
- SGEGOXDYSFKCPT-UHFFFAOYSA-N vilazodone Chemical compound C1=C(C#N)C=C2C(CCCCN3CCN(CC3)C=3C=C4C=C(OC4=CC=3)C(=O)N)=CNC2=C1 SGEGOXDYSFKCPT-UHFFFAOYSA-N 0.000 claims description 2
- 206010001497 Agitation Diseases 0.000 claims 1
- 208000025966 Neurological disease Diseases 0.000 claims 1
- 238000013019 agitation Methods 0.000 claims 1
- 210000004556 brain Anatomy 0.000 description 34
- 241000699670 Mus sp. Species 0.000 description 19
- 230000000694 effects Effects 0.000 description 19
- 102000003823 Aromatic-L-amino-acid decarboxylases Human genes 0.000 description 17
- 108090000121 Aromatic-L-amino-acid decarboxylases Proteins 0.000 description 17
- DUUGKQCEGZLZNO-UHFFFAOYSA-N 5-hydroxyindoleacetic acid Chemical compound C1=C(O)C=C2C(CC(=O)O)=CNC2=C1 DUUGKQCEGZLZNO-UHFFFAOYSA-N 0.000 description 14
- 241000282412 Homo Species 0.000 description 12
- 208000037265 diseases, disorders, signs and symptoms Diseases 0.000 description 12
- WTDRDQBEARUVNC-LURJTMIESA-N L-DOPA Chemical compound OC(=O)[C@@H](N)CC1=CC=C(O)C(O)=C1 WTDRDQBEARUVNC-LURJTMIESA-N 0.000 description 11
- 230000002411 adverse Effects 0.000 description 11
- 230000001225 therapeutic effect Effects 0.000 description 11
- 238000002560 therapeutic procedure Methods 0.000 description 11
- WTDRDQBEARUVNC-UHFFFAOYSA-N L-Dopa Natural products OC(=O)C(N)CC1=CC=C(O)C(O)=C1 WTDRDQBEARUVNC-UHFFFAOYSA-N 0.000 description 10
- 238000006243 chemical reaction Methods 0.000 description 10
- 230000002496 gastric effect Effects 0.000 description 10
- 229960004502 levodopa Drugs 0.000 description 10
- 238000003786 synthesis reaction Methods 0.000 description 10
- 210000001519 tissue Anatomy 0.000 description 10
- 201000010099 disease Diseases 0.000 description 9
- 230000001965 increasing effect Effects 0.000 description 9
- BNQDCRGUHNALGH-UHFFFAOYSA-N benserazide Chemical compound OCC(N)C(=O)NNCC1=CC=C(O)C(O)=C1O BNQDCRGUHNALGH-UHFFFAOYSA-N 0.000 description 7
- 229960000911 benserazide Drugs 0.000 description 7
- 229940079593 drug Drugs 0.000 description 7
- VYFYYTLLBUKUHU-UHFFFAOYSA-N dopamine Chemical compound NCCC1=CC=C(O)C(O)=C1 VYFYYTLLBUKUHU-UHFFFAOYSA-N 0.000 description 6
- 210000001035 gastrointestinal tract Anatomy 0.000 description 6
- 230000001839 systemic circulation Effects 0.000 description 6
- 108091032151 5-hydroxytryptamine receptor family Proteins 0.000 description 5
- 238000010521 absorption reaction Methods 0.000 description 5
- 238000011260 co-administration Methods 0.000 description 5
- 210000000936 intestine Anatomy 0.000 description 5
- 230000002093 peripheral effect Effects 0.000 description 5
- 239000007787 solid Substances 0.000 description 5
- 230000009885 systemic effect Effects 0.000 description 5
- 102000040125 5-hydroxytryptamine receptor family Human genes 0.000 description 4
- 241000699666 Mus <mouse, genus> Species 0.000 description 4
- 238000009098 adjuvant therapy Methods 0.000 description 4
- 230000007812 deficiency Effects 0.000 description 4
- 235000013305 food Nutrition 0.000 description 4
- 230000007774 longterm Effects 0.000 description 4
- 230000004060 metabolic process Effects 0.000 description 4
- 206010012735 Diarrhoea Diseases 0.000 description 3
- 102000004190 Enzymes Human genes 0.000 description 3
- 108090000790 Enzymes Proteins 0.000 description 3
- QIVBCDIJIAJPQS-VIFPVBQESA-N L-tryptophane Chemical compound C1=CC=C2C(C[C@H](N)C(O)=O)=CNC2=C1 QIVBCDIJIAJPQS-VIFPVBQESA-N 0.000 description 3
- 241001465754 Metazoa Species 0.000 description 3
- 206010028813 Nausea Diseases 0.000 description 3
- 241000283984 Rodentia Species 0.000 description 3
- QIVBCDIJIAJPQS-UHFFFAOYSA-N Tryptophan Natural products C1=CC=C2C(CC(N)C(O)=O)=CNC2=C1 QIVBCDIJIAJPQS-UHFFFAOYSA-N 0.000 description 3
- 102000005506 Tryptophan Hydroxylase Human genes 0.000 description 3
- 108010031944 Tryptophan Hydroxylase Proteins 0.000 description 3
- 206010047700 Vomiting Diseases 0.000 description 3
- 150000001413 amino acids Chemical class 0.000 description 3
- 210000003169 central nervous system Anatomy 0.000 description 3
- 230000001684 chronic effect Effects 0.000 description 3
- 238000011278 co-treatment Methods 0.000 description 3
- 230000000295 complement effect Effects 0.000 description 3
- 150000001875 compounds Chemical class 0.000 description 3
- 238000013270 controlled release Methods 0.000 description 3
- 208000035475 disorder Diseases 0.000 description 3
- 229960003638 dopamine Drugs 0.000 description 3
- 238000013265 extended release Methods 0.000 description 3
- 239000000284 extract Substances 0.000 description 3
- 238000010579 first pass effect Methods 0.000 description 3
- 230000005764 inhibitory process Effects 0.000 description 3
- 230000000968 intestinal effect Effects 0.000 description 3
- 230000008693 nausea Effects 0.000 description 3
- 238000001543 one-way ANOVA Methods 0.000 description 3
- 230000003285 pharmacodynamic effect Effects 0.000 description 3
- 238000010149 post-hoc-test Methods 0.000 description 3
- 230000008673 vomiting Effects 0.000 description 3
- 150000004803 5-hydroxytryptophans Chemical class 0.000 description 2
- 206010006550 Bulimia nervosa Diseases 0.000 description 2
- 102000004031 Carboxy-Lyases Human genes 0.000 description 2
- 108090000489 Carboxy-Lyases Proteins 0.000 description 2
- 241000124008 Mammalia Species 0.000 description 2
- 208000008589 Obesity Diseases 0.000 description 2
- 206010000059 abdominal discomfort Diseases 0.000 description 2
- 239000004480 active ingredient Substances 0.000 description 2
- 230000008901 benefit Effects 0.000 description 2
- 208000015114 central nervous system disease Diseases 0.000 description 2
- 238000001514 detection method Methods 0.000 description 2
- 238000009826 distribution Methods 0.000 description 2
- 230000029142 excretion Effects 0.000 description 2
- 230000006870 function Effects 0.000 description 2
- 238000004128 high performance liquid chromatography Methods 0.000 description 2
- 230000002401 inhibitory effect Effects 0.000 description 2
- 239000000543 intermediate Substances 0.000 description 2
- YGPSJZOEDVAXAB-UHFFFAOYSA-N kynurenine Chemical compound OC(=O)C(N)CC(=O)C1=CC=CC=C1N YGPSJZOEDVAXAB-UHFFFAOYSA-N 0.000 description 2
- 238000012423 maintenance Methods 0.000 description 2
- 239000011159 matrix material Substances 0.000 description 2
- 230000007246 mechanism Effects 0.000 description 2
- 230000002503 metabolic effect Effects 0.000 description 2
- 210000002569 neuron Anatomy 0.000 description 2
- 239000002858 neurotransmitter agent Substances 0.000 description 2
- 235000020824 obesity Nutrition 0.000 description 2
- VLTRZXGMWDSKGL-UHFFFAOYSA-N perchloric acid Chemical compound OCl(=O)(=O)=O VLTRZXGMWDSKGL-UHFFFAOYSA-N 0.000 description 2
- 230000001242 postsynaptic effect Effects 0.000 description 2
- 230000003389 potentiating effect Effects 0.000 description 2
- 208000024891 symptom Diseases 0.000 description 2
- 230000000946 synaptic effect Effects 0.000 description 2
- 229940124597 therapeutic agent Drugs 0.000 description 2
- 231100000331 toxic Toxicity 0.000 description 2
- 230000002588 toxic effect Effects 0.000 description 2
- 230000032258 transport Effects 0.000 description 2
- 238000011269 treatment regimen Methods 0.000 description 2
- -1 vilazone Chemical compound 0.000 description 2
- 210000001835 viscera Anatomy 0.000 description 2
- SFLSHLFXELFNJZ-QMMMGPOBSA-N (-)-norepinephrine Chemical compound NC[C@H](O)C1=CC=C(O)C(O)=C1 SFLSHLFXELFNJZ-QMMMGPOBSA-N 0.000 description 1
- UCTWMZQNUQWSLP-VIFPVBQESA-N (R)-adrenaline Chemical compound CNC[C@H](O)C1=CC=C(O)C(O)=C1 UCTWMZQNUQWSLP-VIFPVBQESA-N 0.000 description 1
- 229930182837 (R)-adrenaline Natural products 0.000 description 1
- 208000004998 Abdominal Pain Diseases 0.000 description 1
- QTBSBXVTEAMEQO-UHFFFAOYSA-M Acetate Chemical compound CC([O-])=O QTBSBXVTEAMEQO-UHFFFAOYSA-M 0.000 description 1
- 241000283690 Bos taurus Species 0.000 description 1
- 208000032841 Bulimia Diseases 0.000 description 1
- 241000282472 Canis lupus familiaris Species 0.000 description 1
- 239000004215 Carbon black (E152) Substances 0.000 description 1
- 206010010356 Congenital anomaly Diseases 0.000 description 1
- 241000938605 Crocodylia Species 0.000 description 1
- 206010012289 Dementia Diseases 0.000 description 1
- 241000283086 Equidae Species 0.000 description 1
- 241000282326 Felis catus Species 0.000 description 1
- 102000014630 G protein-coupled serotonin receptor activity proteins Human genes 0.000 description 1
- 241000287828 Gallus gallus Species 0.000 description 1
- 241000219726 Griffonia simplicifolia Species 0.000 description 1
- 108010025020 Nerve Growth Factor Proteins 0.000 description 1
- 102000007072 Nerve Growth Factors Human genes 0.000 description 1
- 208000000224 Night Terrors Diseases 0.000 description 1
- 206010067482 No adverse event Diseases 0.000 description 1
- 241000282320 Panthera leo Species 0.000 description 1
- 241001494479 Pecora Species 0.000 description 1
- 208000001647 Renal Insufficiency Diseases 0.000 description 1
- 206010039966 Senile dementia Diseases 0.000 description 1
- 208000013738 Sleep Initiation and Maintenance disease Diseases 0.000 description 1
- 206010041010 Sleep terror Diseases 0.000 description 1
- 241000251539 Vertebrata <Metazoa> Species 0.000 description 1
- 230000003281 allosteric effect Effects 0.000 description 1
- 230000004075 alteration Effects 0.000 description 1
- 238000010171 animal model Methods 0.000 description 1
- 239000000935 antidepressant agent Substances 0.000 description 1
- 229940005513 antidepressants Drugs 0.000 description 1
- 239000003420 antiserotonin agent Substances 0.000 description 1
- 230000006907 apoptotic process Effects 0.000 description 1
- 238000013459 approach Methods 0.000 description 1
- 210000000467 autonomic pathway Anatomy 0.000 description 1
- 230000009286 beneficial effect Effects 0.000 description 1
- 239000000090 biomarker Substances 0.000 description 1
- 230000001851 biosynthetic effect Effects 0.000 description 1
- 239000008280 blood Substances 0.000 description 1
- 210000004369 blood Anatomy 0.000 description 1
- 210000005013 brain tissue Anatomy 0.000 description 1
- 229940097987 carbidopa 10 mg Drugs 0.000 description 1
- 230000003197 catalytic effect Effects 0.000 description 1
- 210000004027 cell Anatomy 0.000 description 1
- 230000002490 cerebral effect Effects 0.000 description 1
- 235000013330 chicken meat Nutrition 0.000 description 1
- 210000001072 colon Anatomy 0.000 description 1
- 238000002648 combination therapy Methods 0.000 description 1
- 239000002131 composite material Substances 0.000 description 1
- 238000011262 co‐therapy Methods 0.000 description 1
- 230000007423 decrease Effects 0.000 description 1
- 235000005911 diet Nutrition 0.000 description 1
- 230000000378 dietary effect Effects 0.000 description 1
- 231100000676 disease causative agent Toxicity 0.000 description 1
- 231100000673 dose–response relationship Toxicity 0.000 description 1
- 238000012377 drug delivery Methods 0.000 description 1
- 238000002651 drug therapy Methods 0.000 description 1
- 230000004064 dysfunction Effects 0.000 description 1
- 235000005686 eating Nutrition 0.000 description 1
- 229960005139 epinephrine Drugs 0.000 description 1
- 238000002474 experimental method Methods 0.000 description 1
- 210000001723 extracellular space Anatomy 0.000 description 1
- 238000000605 extraction Methods 0.000 description 1
- 239000012530 fluid Substances 0.000 description 1
- 239000012458 free base Substances 0.000 description 1
- 210000005153 frontal cortex Anatomy 0.000 description 1
- 230000005176 gastrointestinal motility Effects 0.000 description 1
- 239000008187 granular material Substances 0.000 description 1
- 230000009546 growth abnormality Effects 0.000 description 1
- 230000036541 health Effects 0.000 description 1
- 229930195733 hydrocarbon Natural products 0.000 description 1
- 150000002430 hydrocarbons Chemical class 0.000 description 1
- 210000003405 ileum Anatomy 0.000 description 1
- 208000026278 immune system disease Diseases 0.000 description 1
- 230000001771 impaired effect Effects 0.000 description 1
- 238000001727 in vivo Methods 0.000 description 1
- 239000004615 ingredient Substances 0.000 description 1
- 206010022437 insomnia Diseases 0.000 description 1
- 210000001630 jejunum Anatomy 0.000 description 1
- 201000006370 kidney failure Diseases 0.000 description 1
- 230000000670 limiting effect Effects 0.000 description 1
- 150000002632 lipids Chemical class 0.000 description 1
- 239000007788 liquid Substances 0.000 description 1
- RLAWWYSOJDYHDC-BZSNNMDCSA-N lisinopril Chemical compound C([C@H](N[C@@H](CCCCN)C(=O)N1[C@@H](CCC1)C(O)=O)C(O)=O)CC1=CC=CC=C1 RLAWWYSOJDYHDC-BZSNNMDCSA-N 0.000 description 1
- 235000019988 mead Nutrition 0.000 description 1
- 230000010534 mechanism of action Effects 0.000 description 1
- 239000012528 membrane Substances 0.000 description 1
- 208000030159 metabolic disease Diseases 0.000 description 1
- 230000037353 metabolic pathway Effects 0.000 description 1
- 238000006241 metabolic reaction Methods 0.000 description 1
- 239000002207 metabolite Substances 0.000 description 1
- 239000011325 microbead Substances 0.000 description 1
- 230000003278 mimic effect Effects 0.000 description 1
- 239000008185 minitablet Substances 0.000 description 1
- 238000012986 modification Methods 0.000 description 1
- 230000004048 modification Effects 0.000 description 1
- 230000035772 mutation Effects 0.000 description 1
- 230000001537 neural effect Effects 0.000 description 1
- 210000002241 neurite Anatomy 0.000 description 1
- 230000000955 neuroendocrine Effects 0.000 description 1
- 230000004766 neurogenesis Effects 0.000 description 1
- 230000000926 neurological effect Effects 0.000 description 1
- 239000003900 neurotrophic factor Substances 0.000 description 1
- 229960002748 norepinephrine Drugs 0.000 description 1
- SFLSHLFXELFNJZ-UHFFFAOYSA-N norepinephrine Natural products NCC(O)C1=CC=C(O)C(O)=C1 SFLSHLFXELFNJZ-UHFFFAOYSA-N 0.000 description 1
- 229940126701 oral medication Drugs 0.000 description 1
- 210000000056 organ Anatomy 0.000 description 1
- 230000003204 osmotic effect Effects 0.000 description 1
- 230000008058 pain sensation Effects 0.000 description 1
- 230000036961 partial effect Effects 0.000 description 1
- 230000007170 pathology Effects 0.000 description 1
- 230000037361 pathway Effects 0.000 description 1
- 235000020030 perry Nutrition 0.000 description 1
- 230000000144 pharmacologic effect Effects 0.000 description 1
- 230000004962 physiological condition Effects 0.000 description 1
- 230000001766 physiological effect Effects 0.000 description 1
- 230000003518 presynaptic effect Effects 0.000 description 1
- 230000002265 prevention Effects 0.000 description 1
- 230000001737 promoting effect Effects 0.000 description 1
- 238000011002 quantification Methods 0.000 description 1
- 238000004445 quantitative analysis Methods 0.000 description 1
- 238000009790 rate-determining step (RDS) Methods 0.000 description 1
- 210000000664 rectum Anatomy 0.000 description 1
- 230000002829 reductive effect Effects 0.000 description 1
- 230000004044 response Effects 0.000 description 1
- 238000012552 review Methods 0.000 description 1
- 230000028327 secretion Effects 0.000 description 1
- 241000894007 species Species 0.000 description 1
- 230000004936 stimulating effect Effects 0.000 description 1
- 210000002784 stomach Anatomy 0.000 description 1
- 230000005062 synaptic transmission Effects 0.000 description 1
- 230000002195 synergetic effect Effects 0.000 description 1
- 230000008337 systemic blood flow Effects 0.000 description 1
- 231100000419 toxicity Toxicity 0.000 description 1
- 230000001988 toxicity Effects 0.000 description 1
- 238000005303 weighing Methods 0.000 description 1
Images
Classifications
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K31/00—Medicinal preparations containing organic active ingredients
- A61K31/33—Heterocyclic compounds
- A61K31/395—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins
- A61K31/40—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins having five-membered rings with one nitrogen as the only ring hetero atom, e.g. sulpiride, succinimide, tolmetin, buflomedil
- A61K31/403—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins having five-membered rings with one nitrogen as the only ring hetero atom, e.g. sulpiride, succinimide, tolmetin, buflomedil condensed with carbocyclic rings, e.g. carbazole
- A61K31/404—Indoles, e.g. pindolol
- A61K31/405—Indole-alkanecarboxylic acids; Derivatives thereof, e.g. tryptophan, indomethacin
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K31/00—Medicinal preparations containing organic active ingredients
- A61K31/13—Amines
- A61K31/135—Amines having aromatic rings, e.g. ketamine, nortriptyline
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K31/00—Medicinal preparations containing organic active ingredients
- A61K31/13—Amines
- A61K31/135—Amines having aromatic rings, e.g. ketamine, nortriptyline
- A61K31/138—Aryloxyalkylamines, e.g. propranolol, tamoxifen, phenoxybenzamine
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K31/00—Medicinal preparations containing organic active ingredients
- A61K31/13—Amines
- A61K31/15—Oximes (>C=N—O—); Hydrazines (>N—N<); Hydrazones (>N—N=) ; Imines (C—N=C)
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K31/00—Medicinal preparations containing organic active ingredients
- A61K31/185—Acids; Anhydrides, halides or salts thereof, e.g. sulfur acids, imidic, hydrazonic or hydroximic acids
- A61K31/19—Carboxylic acids, e.g. valproic acid
- A61K31/195—Carboxylic acids, e.g. valproic acid having an amino group
- A61K31/197—Carboxylic acids, e.g. valproic acid having an amino group the amino and the carboxyl groups being attached to the same acyclic carbon chain, e.g. gamma-aminobutyric acid [GABA], beta-alanine, epsilon-aminocaproic acid, pantothenic acid
- A61K31/198—Alpha-aminoacids, e.g. alanine, edetic acids [EDTA]
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K31/00—Medicinal preparations containing organic active ingredients
- A61K31/33—Heterocyclic compounds
- A61K31/335—Heterocyclic compounds having oxygen as the only ring hetero atom, e.g. fungichromin
- A61K31/34—Heterocyclic compounds having oxygen as the only ring hetero atom, e.g. fungichromin having five-membered rings with one oxygen as the only ring hetero atom, e.g. isosorbide
- A61K31/343—Heterocyclic compounds having oxygen as the only ring hetero atom, e.g. fungichromin having five-membered rings with one oxygen as the only ring hetero atom, e.g. isosorbide condensed with a carbocyclic ring, e.g. coumaran, bufuralol, befunolol, clobenfurol, amiodarone
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K31/00—Medicinal preparations containing organic active ingredients
- A61K31/33—Heterocyclic compounds
- A61K31/395—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins
- A61K31/435—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins having six-membered rings with one nitrogen as the only ring hetero atom
- A61K31/44—Non condensed pyridines; Hydrogenated derivatives thereof
- A61K31/4406—Non condensed pyridines; Hydrogenated derivatives thereof only substituted in position 3, e.g. zimeldine
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K31/00—Medicinal preparations containing organic active ingredients
- A61K31/33—Heterocyclic compounds
- A61K31/395—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins
- A61K31/435—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins having six-membered rings with one nitrogen as the only ring hetero atom
- A61K31/44—Non condensed pyridines; Hydrogenated derivatives thereof
- A61K31/445—Non condensed piperidines, e.g. piperocaine
- A61K31/4523—Non condensed piperidines, e.g. piperocaine containing further heterocyclic ring systems
- A61K31/4525—Non condensed piperidines, e.g. piperocaine containing further heterocyclic ring systems containing a five-membered ring with oxygen as a ring hetero atom
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K31/00—Medicinal preparations containing organic active ingredients
- A61K31/33—Heterocyclic compounds
- A61K31/395—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins
- A61K31/435—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins having six-membered rings with one nitrogen as the only ring hetero atom
- A61K31/44—Non condensed pyridines; Hydrogenated derivatives thereof
- A61K31/445—Non condensed piperidines, e.g. piperocaine
- A61K31/4523—Non condensed piperidines, e.g. piperocaine containing further heterocyclic ring systems
- A61K31/454—Non condensed piperidines, e.g. piperocaine containing further heterocyclic ring systems containing a five-membered ring with nitrogen as a ring hetero atom, e.g. pimozide, domperidone
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K31/00—Medicinal preparations containing organic active ingredients
- A61K31/33—Heterocyclic compounds
- A61K31/395—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins
- A61K31/495—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins having six-membered rings with two or more nitrogen atoms as the only ring heteroatoms, e.g. piperazine or tetrazines
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K31/00—Medicinal preparations containing organic active ingredients
- A61K31/33—Heterocyclic compounds
- A61K31/395—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins
- A61K31/495—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins having six-membered rings with two or more nitrogen atoms as the only ring heteroatoms, e.g. piperazine or tetrazines
- A61K31/496—Non-condensed piperazines containing further heterocyclic rings, e.g. rifampin, thiothixene
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K45/00—Medicinal preparations containing active ingredients not provided for in groups A61K31/00 - A61K41/00
- A61K45/06—Mixtures of active ingredients without chemical characterisation, e.g. antiphlogistics and cardiaca
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K9/00—Medicinal preparations characterised by special physical form
- A61K9/0012—Galenical forms characterised by the site of application
- A61K9/0053—Mouth and digestive tract, i.e. intraoral and peroral administration
- A61K9/0065—Forms with gastric retention, e.g. floating on gastric juice, adhering to gastric mucosa, expanding to prevent passage through the pylorus
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P25/00—Drugs for disorders of the nervous system
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P25/00—Drugs for disorders of the nervous system
- A61P25/24—Antidepressants
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K2300/00—Mixtures or combinations of active ingredients, wherein at least one active ingredient is fully defined in groups A61K31/00 - A61K41/00
Abstract
本发明提供了一种在需要其的受试者中提高肠内施用5‑羟色氨酸(5‑HTP)的生物利用度的方法,所述方法包括肠内共同施用低剂量卡比多巴和所述5‑HTP,以及用于该方法的药物制剂。在一些实施方案中,5‑HTP和/或低剂量卡比多巴作为缓释制剂提供。
Description
政府支持声明
本发明是在政府支持下由美国国立卫生研究院(NIH)授予的联邦资助2R01MH079201-06A1完成的。政府对本发明享有某些权利。
背景技术
5-羟色氨酸(5-HTP)是5-羟色胺(aka,5-羟色胺,5-HT)的天然直接前体。据报道,5-HTP在与中枢神经系统功能有关的一系列疾病中具有治疗潜力(Turner等,2006a),但在人体中表现出快速吸收(TMax~1h)和快速消除(T1/2~2h)的能力(Gijsman等,2002;Westenberg等,1982)。5-HTP单独作为一个分子可能不适合药物治疗,因为施用后快速发生与CMax相关的不良事件,并且每天需要剂量4-6次以维持合理稳定的5-HTP暴露,这种剂量要求在一般治疗环境中是不现实且不切实际的(Jacobsen等,2016a)。
卡比多巴和苄丝肼是芳香族氨基酸脱羧酶(AAAD)的外围抑制剂(PDI)。在人类中,PDI协同治疗已被用于增强多巴胺前体左旋多巴用于帕金森氏病治疗的生物利用度(Freitas等,2016),以及5-HT前体5-HTP的生物利用度用于实验性治疗(Turner等,2006b)。对于5-HTP,PDI协同治疗还可减少人体胃肠道(GI)不良事件,例如恶心,腹泻,胃部不适,呕吐-与胃肠道内5-HTP转化为5-HT有关(Byerley等,1987)。
苄丝肼和卡比多巴通常与左旋多巴一起使用,左旋多巴:PDI的比例为4:1(Merck,2017;Roche,2015)。然而,苄丝肼比卡比多巴更有效。例如,口服施用后,苄丝肼是外周器官中AAAD的有效抑制剂比卡比多巴高10倍(Da Prada等,1987)。因此,苄丝肼的起始剂量约为卡比多巴的一半(Merck,2017;Roche,2015),并且苄丝肼和卡比多巴不能简单地进行剂量替换。
在人类慢性研究中,使用PDI(主要是卡比多巴)和5-HTP的报道使用的PDI剂量范围为150mg/天至1000mg/天。此类PDI剂量会在所有周围组织(即大脑外部的所有组织)中引起对AAAD的强烈系统性抑制。报道的5-HTP:PDI比值通常在4:1和10:1之间,类似于PDI与左旋多巴一起用于治疗帕金森氏病的情况。先前通过口服5-HTP和PDI治疗进行的慢性临床研究使用的PDI剂量>2mg/kg/天(假定患者体重70公斤),这将在所有系统性外周组织中强烈抑制AAAD。已报道的人类对卡比多巴和5-HTP的慢性研究(例如,Kahn和Westerberg,1985;van Hiele,1980;van Praag,1982)将产生>25ng/ml的平均卡比多巴血浆水平(VerhagenMetman等,2015;Yeh)等人,1989),通过对这里引用的参考文献进行推断。
当PDI以全身活性剂量例如卡比多巴1-2mg/kg/天,与左旋多巴一起给予对致命性帕金森氏病患者时,没有可能将归因于PDI治疗的不良反应,因为当将PDI和左旋多巴一起施用时,很难分开其影响。在没有左旋多巴的情况下,PDI治疗的安全性更加难以评估,因为PDI治疗对外周多巴胺、肾上腺素和去甲肾上腺素的作用(抑制合成)将因同时施用左旋多巴(增强合成)而抵消(Rose等,1988)。
PDI不是AAAD的特异性抑制剂;它们还可以抑制犬尿氨酸途径的酶,其异常与中枢神经系统、代谢和免疫疾病有关(Badawy and Bano,2016)。此外,在动物中长期PDI治疗即卡比多巴、苄丝肼而没有左旋多巴可以引起明显的毒性作用,包括肾脏供血不足和生长异常(Rauws等,1982;Yoshimura等,1987)。此外,在人类中,先天氨基酸脱羧酶缺乏会引起严重的自主神经、运动和其他症状,表明长期的PDI治疗可能具有广谱毒性作用(Manegold等,2009)。
总体而言,尚未建立在其他方面健康的人中不进行左旋多巴治疗的PDI治疗的长期安全性,并且PDI的药理学可能会对外周的各种生理系统产生负面影响。因此,如果可能的话,采用5-HTP和较低剂量的PDI(例如卡比多巴)的人体疗法将是更好的选择。
发明内容
如附图所示,出乎意料地发现,低剂量的卡比多巴,特别是高5-HTP:PDI比例,可以显著(>100%)增强5-HTP口服生物利用度,而在治疗有效范围中不会引起全身性卡比多巴血浆水平的升高。。
因此,本公开内容的一个方面提供了一种在受试者(例如人类受试者)中提高5-HTP的生物利用度的方法,该方法包括下述或由下述组成或基本上由下述组成:向所述受试者共同施用治疗有效量的5-HTP以及低剂量的卡比多巴,从而提高受试者的5-HTP生物利用度。
在一些实施方案中,低剂量卡比多巴产生亚临床的卡比多巴血浆水平,这导致卡比多巴仅在肠道中局部起作用以增强5-HTP生物利用度,这反过来使毒性和其他与全身卡比多巴接触有关的不良生理影响的风险最小化。
在一些实施方案中,一种增强肠内施用的5-HTP在需要其的人类受试者中的生物利用度的方法,包括:将低剂量卡比多巴与所述5-HTP肠内共同施用,所述低剂量卡比多巴的日剂量为约0.1或0.2至约0.5、0.6或0.8mg/kg/天(或每天约5或10mg至约35、50或60mg/天),从而提高肠内施用的5-HTP的生物利用度。
在一些实施方案中,5-HTP和卡比多巴以5-HTTP:卡比多巴的每日剂量比为100:1、80:1、60:1或50:1至40:1、30:1或20:1施用。
在一些实施方案中,在肠内共同施用所述低剂量卡比多巴后,受试者的卡比多巴血浆水平小于25、20、15、10、5或2ng/ml。
在一些实施方案中,每天一次,两次或三次施用5-HTP和低剂量卡比多巴。
在一些实施方案中,以选自口服速释制剂、口服缓释制剂、口服肠内凝胶、直肠栓剂及其组合的方式施用制剂。
在一些实施方案中,以相同或分开的剂型将5-HTP和卡比多巴作为5-HTP和低剂量卡比多巴的缓释(SR)制剂提供。
在一些实施方案中,所述受试者需要治疗精神病和/或神经病,例如情绪异常或冲动或攻击控制中的异常。在一些实施方案中,所述受试者需要治疗抑郁症、社交焦虑症、恐慌症、广泛性焦虑症、强迫症、冲动控制障碍、自杀倾向、边缘性人格障碍、纤维肌痛、共济失调,与神经系统疾病有关的情绪、认知或行为症状和焦虑(例如阿尔茨海默氏症,帕金森氏症),中风康复、自闭症、偏头痛、睡眠障碍、经前焦虑症、创伤后应激障碍、产后抑郁或干扰素治疗后抑郁。在一些实施方案中,所述受试者需要治疗抑郁症、焦虑症、自杀倾向、强迫症或ADHD。在一些实施方案中,所述受试者需要治疗抑郁症,重度抑郁症或抗治疗性抑郁症(用5-羟色胺再摄取抑制剂治疗后不能完全缓解)。
在一些实施方案中,所述方法包括用5-HTP和低剂量卡比多巴进行单一疗法。在一些实施方案中,该方法包括用5-HTP和低剂量卡比多巴以及5-羟色胺增强剂的辅助疗法。在一些实施方案中,5-羟色胺增强剂选自5-羟色胺再摄取抑制剂、5-羟色胺去甲肾上腺素再摄取抑制剂、三重再摄取抑制剂、单胺氧化酶抑制剂、三环抗抑郁剂、5-羟色胺激动剂、苯丙胺、5-羟色胺前体、5-羟色胺原药、5-羟色胺生物合成中间体及其药学上可接受的盐。在一些实施方案中,5-羟色胺增强剂是选择性5-羟色胺再摄取抑制剂(SSRI)。在一些实施方案中,5-羟色胺增强剂选自以下组成的组:西酞普兰、达泊西汀、艾司西酞普兰、氟西汀、氟伏沙明、吲哚平、帕罗西汀、舍曲林、维拉唑酮、伏替西汀、齐美替丁及其组合。
在一些实施方案中,任选地以相同或分开的剂型口服施用5-HTP和低剂量卡比多巴。
在一些实施方案中,所述低剂量卡比多巴以约0.1或0.2至约0.5mg/kg/天(或每天约5或10mg至约35mg)的每日剂量提供。
在一些实施方案中,5-HTP以每天约0.1、0.2、0.5或0.75克至约1、4或6克的日剂量提供。
还提供了包含5-HTP和低剂量卡比多巴的适合肠内施用的药物制剂或部件试剂盒,所述制剂适于以每天一次,两次或三次施用。
在一些实施方案中,所述药物制剂或部件试剂盒中的低剂量卡比多巴以约0.1或0.2至约0.5、0.6或0.8mg/kg/天(或每天约5或10mg至35、50或60mg)的日剂量提供。
在一些实施方案中,5-HTP和低剂量卡比多巴在所述药物制剂或部件试剂盒中以从5-HTTP:卡比多巴为100:1、80:1、60:1或50:1至40:1、30:1或20:1的剂量比提供。
在一些实施方案中,所述制剂是适合于口服或直肠施用的固体剂型。
在一些实施方案中,5-HTP和/或卡比多巴的制剂为每日施用一次的一个药片、胶囊或其他制剂;或每日施用二次的两个药片、胶囊或其他制剂。
在一些实施方案中,所述制剂包含5-HTP和/或卡比多巴的缓释制剂。在一些实施方案中,所述缓释制剂包括胃滞留制剂。在一些实施方案中,将5-HTP和低剂量卡比多巴与膳食共同施用(例如,当以胃滞留制剂形式提供时)。
还提供了本文所述的低剂量卡比多巴在增强5-HTP在受试者(例如人类受试者)中的生物利用度的方法中的用途,所述方法包括以下步骤或由其组成或基本上由其组成:向所述受试者共同施用低剂量的卡比多巴与5-HTP,从而提高了受试者的5-HTP生物利用度。
还提供了本文所教导的低剂量卡比多巴在制备用于增强受试者(例如人类受试者)中5-HTP的生物利用度的药物的方法中的用途。
结合附图,在下面的描述中解释了本公开的前述方面和其他特征。
附图简述
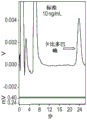
图1.低剂量卡比多巴对5HTP施用引起的5-HTP血浆升高的影响。卡比多巴剂量依赖性地增加5-HTP血浆升高。WT和5-HTHypo小鼠的作用相似(由于色氨酸羟化酶突变而导致选择性和部分脑5-HT缺乏的小鼠(Beaulieu等,2008)),可能反映了这两种基因型的小鼠在基线具有相似的5-HTP血浆水平。*表示与仅用5-HTP 200mg/kg/天治疗的组有统计学显著差异。单向方差分析,Dunnett事后检测。
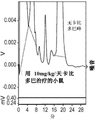
图2.低剂量卡比多巴对5-HTP诱导的脑5-HT组织升高的影响。卡比多巴剂量依赖性地增加了由5-HTP诱导的5-HT组织升高。这种作用在5-HTHypo小鼠中成比例地更加明显,这是由于其较低的内源性大脑5-HT合成在基线时具有较低的大脑5-HT水平。*表示与仅用5-HTP 200mg/kg/天治疗的组有统计学显著差异。单向方差分析,Dunnett事后检测。
图3.低剂量卡比多巴对5-HTP诱导的脑5-HIAA组织升高的影响。卡比多巴剂量依赖性地增加了5-HTP诱导的5-HIAA组织升高。这种作用在5-HTHypo小鼠中成比例地更加明显,这是由于其较低的内源性大脑5-HT合成在基线具有较低的大脑5-HIAA水平。*表示与仅用5-HTP 200mg/kg/天治疗的组有统计学显著差异。单向方差分析,Dunnett事后检测。
图4A-4D.血浆中卡比多巴的HPLC电化学定量。图4A:高氯酸中的10ng/ml标准。卡比多巴峰在24分钟时可见。图4B:来自未经卡比多巴处理的小鼠的血浆提取物。没有可见的卡比多巴峰。24分钟时的基线波动是检测器噪声。图4C:来自未用卡比多巴治疗的小鼠的血浆提取物,但在提取前用血浆卡比多巴峰达到2ng/ml的浓度。卡比多巴峰在24分钟时可见。图4D:来自用10mg/kg/天的卡比多巴治疗的小鼠的血浆提取物。没有可见的卡比多巴峰。如图所示,在24分钟时基线波动是检测器噪声。
图5A-5C.本发明作用机理的定性说明。在基线条件下(图5A),体循环中基本上不存在5-HTP。(在基线,脑5-HT是由色氨酸在脑中局部合成的。)单独进行5-HTP治疗(图5B)将适度升高全身循环中的5-HTP,并适度升高脑5-HT。与低剂量卡比多巴的共处理(图5C)将显著抑制肠道中的局部氨基酸脱羧酶,减少5-HTP的首过代谢,从而与单独使用5-HTP治疗后的脑5-HT相比使更多的5-HTP进入全身循环并引起更强的升高5-HTP治疗后。同时,在全身循环中,卡比多巴的水平将太稀而无法功能性抑制AAAD。符号:·,卡比多巴,,5-HTP。
发明详述
为了促进对本公开原理的理解,现在将参考优选实施方案,并且将使用特定语言来描述它们。然而,应当理解的是,不意图因此限制本发明的范围,如本文所示的对本发明的这种修改和进一步修改是本发明所涉及领域的技术人员通常会想到的。
冠词“一种”和“一个”在本文中用于指代该冠词的语法对象的一个或多个(即,至少一个)。举例来说,“一种元素”是指至少一个元素并且可以包括超过一个元素。
“大约”用于通过提供给定值可以“略高于”或“略低于”端点而不影响期望的结果,从而为数值范围端点提供灵活性。
本文中使用的术语“包括”、“包括”或“具有”及其变体意欲包括其后列出的元素及其等效物以及附加元素。作为“包括”、“包含”或“具有”某些元素的实施方案也被设想为“基本上由”和“由”这些特定元素组成。
除非本文另有说明,否则本文中对值范围的叙述仅仅是作为单独提及范围内的每个单独值的速记方法,并且每个单独的值被并入说明书中,如同在本文中单独叙述一样。例如,如果浓度范围规定为1%至50%,则表示本说明书中明确列举了2%至40%、10%至30%或1%至3%等数值。这些只是具体意图的示例,并且所述最低值和最高值之间所有可能的数值组合(包括列举的最低值和最高值)应被视为已在本说明书中明确说明。
如本文所用,“治疗”,“疗法”和/或“治疗方案”是指响应于由患者表现出的或患者可能易感的疾病,病症或生理状况而进行的临床干预。治疗的目的包括减轻或预防症状,减慢或停止疾病,病症或病状的发展或恶化和/或疾病,病症或病状的缓解。
术语“有效量”或“治疗有效量”是指足以实现有益或期望的生物学和/或临床结果的量。
如本文所用,术语“受试者”和“患者”在本文可互换使用,并且是指人类和非人类动物。本公开的术语“非人类动物”包括所有脊椎动物,例如哺乳动物和非哺乳动物,例如非人灵长类,绵羊,狗,猫,马,牛,鸡,两栖动物,爬行动物等。
除非另有定义,否则本文中使用的所有技术术语具有与本公开所属领域的普通技术人员通常所理解的相同含义。
I.5-羟色氨酸
“5-羟色氨酸”或“5-HTP”是体内5-羟色胺(aka 5-羟色胺,5-HT)的前体。在人体中,5-HTP是通过色氨酸羟化酶(外周中为亚型1,大多数神经元中为型2)从饮食色氨酸合成的(Jacobsen等,2016a)。5-HTP通过芳香族氨基酸脱羧酶(AAAD)迅速转化为5-HT,AAAD是一种在基线条件下不饱和的高容量普遍表达的酶(Bowsher,1986)。因此,5-HTP是5-HT的天然、直接和限速前体。
不论是口服还是胃肠外,外源给予5-HTP均可提高动物模型中脑5-HT的合成和功能水平(即细胞外5-HT,5-HTExt)(Jacobsen等,2016b;Perry和Fuller,1993),以及在人中的情况(Agren等,1991;Sargent等,1998)。
与5-HT系统的一般情况一样,非人哺乳动物物种例如啮齿类与人之间的5-HTP生物学和药理学非常相似。例如,在啮齿类和人中,5-HTP唯一已知的代谢命运是由AAAD转化为5-HT(Jacobsen等,2016a);在基线时,5-HTP血浆和组织水平非常低(Gijsman等,2002;Jacobsen等,2016b);并且单独使用外源性5-HTP只能适度地提高大脑5-HT功能的神经内分泌生物标志物,而外源性5-HTP可以与SSRI的施用同时协同作用,从而提高5-HT的功能,超出SSRI的作用(Fuller和Snoddy,1980;Sargent等人)等,1998)。因此,在啮齿动物中对5-HTP药理学的发现可以很好地转化用于人类。
5-HTP可以很容易地从植物Griffonia Simplicifolia的种子中获得,并且可以商购。还可以通过化学合成获得5-HTP(参见例如CN103554005A)。据报道,在人试验性实验中,5-HTP的外源施用具有治疗多种疾病的潜力,例如,作为单一疗法治疗抑郁症(Takahashi等,1976),作为辅助疗法治疗抑郁症(van Praag,1982),焦虑症(Kahn)(1987),肥胖(Cangiano等,1992),共济失调(Trouillas等,1988),偏头痛(Nicolodi和Sicuteri,1999),纤维肌痛(Caruso等,1990),失眠(Soulairac和Lambinet,1977)和睡惊症(Bruni等,2004)。
此外,作为单一疗法或辅助疗法的5-HTP将增强大脑中的5-HT功能,而5-HTP可以治疗其他疾病,如已知可由5-HT刺激性药物治疗的单一疗法或辅助疗法,包括但不限于:创伤后应激障碍(Connor等,1999),社交恐惧症(Lader等,2004),焦虑症(Coric等,2010),老年痴呆症(Viscogliosi等,2017),阿尔茨海默氏症相关痴呆(Bartels等,2018),强迫症(Blier和Bergeron,1996),经前烦躁不安(Steiner等,1995),产后抑郁症(Appleby等,1997),贪食症(Jackson等,2010),暴食症(Guerdjikova等,2008),中风恢复(Mead等,2013)和/或假性延髓情绪(Sloan等,1992)。此外,5-HTP药物可以治疗被认其病理为涉及5-HT缺乏症的疾病,包括但不限于冲动控制障碍(Carver等,2008),边缘性人格障碍(Brown等,1982),自杀倾向(Asberg,1997)和/或自闭症(Veenstra-VanderWeele等,2012)。
口服后,5-HTP迅速从上肠吸收(TMax约1h),然后迅速消除(转化为5-HT),半衰期为约2h(Gijsman等,2002;Westenberg等,1982)。快速吸收与CMax相关的胃肠道(GI)不良事件有关,例如恶心,腹泻,腹痛和呕吐(Byerley等,1987;Lowe等,2006)。这些不合意的不良事件是由于胃肠道内5-HTP意外转化为5-HT引起的(Turner等,2006b)。实际上,动物研究表明,在肠壁运输过程中,超过50%的外源性5-HTP转化为5-HT(Shindo等人,1977)。5-HT促进胃肠动力,液体分泌和疼痛感(Gershon,2013),与观察到的与5-HTP施用相关的胃肠不良事件相对应(Turner等,2006b)。为了减少5-HTP引起的GI不良事件,大多数试验中的5-HTP剂量必须在数天或数周内分多个步骤逐步提高(Alino等,1976;van Hiele,1980;van Praag,1982)。不幸的是,这使治疗变得复杂,损害了依从性,并损害了整体临床有效性(Claxton等,2001)。总而言之,5-HTP的快速药代动力学使得其天然速释形式的5-HTP难以用作治疗剂。实际上,当前尚无食品和药物管理局批准使用5-HTP作为活性成分的药品。
5-HTP的半衰期短,需要每天多次(即4-6次)剂量来维持合理稳定的5-HTP血浆暴露(Jacobsen等,2016a;van Praag,1982)。这种方案在现实生活中的治疗环境中是不切实际的。依从性低,并且治疗效果受损。幸运的是,缓释(SR)递送可以显著地且以出乎意料的程度增强5-HTP的药物特性(请参见Jacobsen等人的美国专利第9,468,627号和第8,969,400号),这得到了动物数据的支持(Jacobsen等人,2016b)。此外,5-HTP的口服生物利用度似乎不高(Gijsman等,2002)。当单独使用天然5-HTP时,治疗性研究通常使用高剂量的5-HTP,例如900mg/天用于治疗肥胖症(Cangiano等,1992)。由于固体剂型和/或每剂多个片剂或胶囊剂的尺寸大,高5-HTP剂量具有缺点。
如本文所用,“5-HTP的缓释制剂”是指具有以缓慢的速率释放5-HTP的能力的制剂,从而与速释制剂相比,血浆T1/2增加和/或TMax延迟。术语“慢速5-HTP”和“缓慢释放5-HTP”可互换使用,是指使5-HTP以比直接施用慢的速率在受试者体内释放的能力。参见Jacobsen等人的美国专利第9,468,627号,其全部内容通过引用合并于此。可用于此类制剂的其他术语包括但不限于“持续释放”,“控制释放”,“延长释放”和“随时间释放”。
例如,立即释放的口服5-HTP通常具有约2小时的T1/2,因此,缓释的5-HTP具有大于3、4、5、6或7小时的T1/2。在一些实施方案中,T1/2为至少8小时。在一些实施方案中,T1/2为8、10或12小时至24、48或72小时。
作为另一个例子,速释口服5-HTP的TMax为1-2小时。因此,在一些实施方案中,施用和/或配制5-HTP的缓释制剂以使5-HTP的Tmax(施用后最大血浆浓度的时间)为至少2小时,或2小时至12小时之间。
在一些实施方案中,以适合肠内施用的制剂的治疗有效量提供5-HTP。如本文所用,术语“治疗有效量”是指足以在受试者中显示益处的5-HTP的量。在一些实施方案中,以单位剂量提供制剂,用于每天一次或每天两次施用。参见Jacobsen等人的美国专利第8,969,400号,其全部内容通过引用合并于此。
在一些实施方案中,可以提供0.05至10克的5-HTP的日剂量(例如,作为用于每日一次施用的一个药片、胶囊或其他剂型,或作为用于每日两次施用的两个药片、胶囊或其他剂型,每个含有日剂量的一半)。在一些实施方案中,所述日剂量可以是每天0.01、0.05、0.1、0.2、0.5或0.75至5、8或10克。在一些实施方案中,所述日剂量可以是每天1至5克。在一些实施方案中,所述日剂量可以是每天1至3克。
在一些实施方案中,对于人类受试者,以每天约0.1、0.2、0.5或0.75克至每天约1、4或6克的日剂量提供5-HTP。
在一些实施方案中,施用5-HTP以便达到平均10-100ng/ml的血浆5-HTP水平。在一些实施方案中,施用5-HTP以便达到平均100-1000ng/ml的血浆5-HTP水平。在一些实施方案中,施用5-HTP以达到平均1000-10000ng/ml的血浆5-HTP水平。
II.低剂量卡比多巴
卡比多巴抑制芳香族氨基酸脱羧酶(AAAD),该酶催化5-HTP转化为5-HT,左旋多巴转化为多巴胺以及其他类似的代谢反应(Bowsher R.R.,1986)。卡比多巴的治疗维持剂量>1mg/kg/天(假设患者体重为70kg)(Merck,2017年;Pahwa等人,2014年;van Praag,1982年)。在稳定状态下,在治疗方案中,维持剂量下卡比多巴的平均血浆水平≥25ng/ml(范围:25-150ng/ml)(Verhagen Metman等人,2015;Yeh等人,1989)。
如本文所用,“低剂量”卡比多巴是指低于临床上通常使用的剂量。AAAD是催化5-HTP转化为5-HT的酶,以过量存在并且在基线条件下是不饱和的(Bowsher R.R.,1986)。结果,在基线条件下5-HTP水平非常低,反映出5-HT合成中的限速步骤是由色氨酸羟化酶催化从色氨酸形成5-HTP(Jacobsen等,2012a)。因此,最低水平的PDI(例如平均卡比多巴<25ng/ml(Verhagen Metman等人,2015;Yeh等人,1989))将不会对全身循环和内部器官中的AAAD活性产生实质性影响。
在一些实施方案中,低剂量卡比多巴以约0.1或0.2至约0.5、0.6或0.8mg/kg/天(或每天约5或10mg至约35、50或60mg)的日剂量提供给人类受试者。
在一些实施方案中,5-HTP和低剂量卡比多巴共同施用/剂量形式起作用,使得5-HTP向5-HT的转化基本上仅在肠中在5-HTP吸收的位点被抑制,而AAAD活性在全身循环、内部器官和大脑中将保持基本和功能不受抑制(参见图5A-5C)。
不希望受到理论的束缚,如本文教导的用低剂量卡比多巴在肠中对AAAD的选择性抑制可完全或部分地保护5-HTP免于在肠中由AAAD催化转化为5-HT。反过来,这允许更多的5-HTP进入全身血液循环并被转运到大脑,在那里5-HTP可以转化为5-HT并增强大脑的5-HT合成和5-HT功能。此外,由于大多数由5-HTP引起的不良事件是由肠道内转化为5-HT引起的(例如腹泻,恶心,胃肠不适,呕吐)(Byerley等,1987),本发明可减少此类5-HT相关的GI不良事件。
脑中5-HT功能增强可能表现为净5-HT突触和突触外释放增加以及细胞外5-HT(5-HTExt)水平升高。这导致通过脑中5-羟色胺受体的5-HT神经传递增加,这种作用可以发挥治疗作用。血清素受体的下游机制包括但不限于神经可塑性,电生理变化,大脑连通性调节,脑结构回路改变,脑功能回路改变,总体脑结构改变,神经突结构和补体改变,神经营养因子改变,神经发生改变,神经元数量和补体改变,非神经元细胞数量和补体改变以及凋亡改变。在一些实施方案中,所述共同施用可以进一步包括另一种5-羟色胺能治疗剂的施用。
如本文所用,“卡比多巴缓释制剂”是指具有以缓慢速率释放低剂量卡比多巴的能力的制剂,使得与速释制剂相比,血浆T1/2延迟和/或TMax降低。卡比多巴的缓释制剂可以与5-HTP一起在同一剂型或分开的剂型中提供。术语“缓速卡比多巴”和“缓释卡比多巴”可互换使用,是指与以速释剂型直接施用相比,使卡比多巴以比较慢的速率在受试者体内释放的能力。参见,例如,Han等人的US 2006/0013875(Pahwa等人,2014)。可用于此类制剂的其他术语包括但不限于“持续释放”,“控制释放”,“延长释放”和“随时间释放”。
III.配方和施用
本发明中使用的5-HTP和/或卡比多巴可以是游离碱;盐;偶联物(例如氨基酸偶联物,烃偶联物,脂质偶联物);改变吸收、分布、代谢和/或排泄特性的偶联物;或进行同位素修饰以改变吸收,分布,代谢和/或排泄特性。参见Tamir等人的美国专利第4,658,038号;Xiang等人的美国专利第7,101,912号;Jacobsen等人的美国专利第8,969,400号,其通过引用并入本文。
适用于5-HTP和/或卡比多巴肠内施用的固体药物剂型可以包括片剂,胶囊剂或颗粒剂,其可以使用本领域技术人员已知的用于制备药物制剂的标准药物方法来制备。实例包括但不限于胃滞留制剂(Lopes等,2016),可溶胀基质片剂(Verhagen Metman等,2015),易腐蚀基质片剂(Nokhodchi等,2012),渗透性片剂(Thombre等,2004)),迷你片剂(Mitra等,2017)和微珠(Freitas等,2016)。
在一些实施方案中,所述制剂是5-HTP和低剂量卡比多巴的缓释胃肠滞留制剂,其以相同或分开的剂型共同施用(参见综述,Lopes等,2016;也参见美国专利No.6,340,475、6,635,280、6,723340、9,161,911和9,980903)。
根据具体实施方案,可以将5-HTP和/或卡比多巴剂型与食物一起或不与食物一起施用。在一些实施方案中,所述剂型将与食物(例如通过进餐;参见美国专利第7,438,927号)一起服用/施用。在其他实施方案中,将在禁食时服用/施用所述剂型。
适用于肠施用的固体剂型可以将5-HTP和/或卡比多巴主要递送至胃,空肠,回肠,结肠或直肠或其组合。5-HTP和/或卡比多巴可以通过固体剂量在一段时间内释放,范围从在施用部位即时到达24小时,如适用于上述适应症。5-HTP和/或卡比多巴剂型也可以是液体,凝胶或半固体,或掺入固体成分的非固体。
在一些实施方案中,肠溶剂型可以口服或直肠施用。可以根据治疗需要一次,两次或更频繁地施用。每剂可以施用一个或多个单位。5-HTP和/或卡比多巴剂型可根据待治疗对象的治疗需要,立即、在一天、数天、数周、数月或不确定的时间中施用。
在一些实施方案中,每个亚剂量(例如,在每天两次或更多次的施用中的第一次)包括一个药片、胶囊或其他剂型。在其他实施方案中,每个亚剂量包括两个或多个药片、胶囊或其他剂型。
可以在数天、数周或数月内使5-HTP和/或卡比多巴的剂量逐渐达到至最终剂量,或者可以从治疗开始就施用最终剂量强度。
5-HTP和卡比多巴的共同施用可作为单一疗法用于治疗,即无需其他伴随的5-羟色胺增强剂疗法。可选地,5-HTP和卡比多巴共同施用可以在治疗上用作辅助疗法,即与另一种5-羟色胺能疗法或5-羟色胺增强剂一起,包括但不限于5-羟色胺再摄取抑制剂,5-羟色胺去甲肾上腺素再摄取抑制剂,单胺氧化酶抑制剂,5-羟色胺激动剂,5-羟色胺拮抗剂,5-羟色胺受体变构调节剂,5-羟色胺前体,5-羟色胺合成辅助因子和/或5-羟色胺代谢途径中生物元素的调节剂。参见Jacobsen等人的美国专利第9,468,627号。
本文所用的“5-羟色胺增强剂”是指直接或间接增加5-羟色胺可用于在中枢神经系统中与突触后膜上的5-羟色胺受体结合或直接刺激5-羟色胺受体的任何化合物,包括但不限于不限于5-羟色胺再摄取抑制剂,单胺氧化酶抑制剂,三环抗抑郁药,5-羟色胺激动剂,苯丙胺,5-羟色胺前体,5-羟色胺前药,5-羟色胺生物合成中的中间体,辅因子及其药学上可接受的盐。这样的化合物可以单独施用或与其他5-羟色胺增强剂组合施用。
术语“SSRI”或“选择性5-羟色胺再摄取抑制剂”是指通常用作抗抑郁药的那些化合物,并与神经递质5-羟色胺的细胞外水平增加相关,其通过抑制神经递质5-羟色胺被突触前细胞摄入,增加可与突触后5-羟色胺受体结合的突触裂中的5-羟色胺水平。合适的SSRI的实例包括但不限于西酞普兰,达泊西汀,依西酞普兰,氟西汀,氟伏沙明,吲哚平,帕罗西汀,舍曲林,维拉唑酮,伏替西汀,齐美替丁及其组合。
5-HTP和低剂量卡比多巴可以以相同的剂型一起提供,也可以以包含独立剂型的部件试剂盒的分别提供,然后将它们组合在一起用于如本文所述的联合治疗。它们也可以包装并一起作为使用5-羟色胺增强剂(例如SSRI)的辅助疗法中的部件试剂盒的单独组分存在。
提供以下非限制性实施例以进一步描述和证明本发明。
实施例
方法
小鼠:使用成年小鼠,具有正常5-HT水平的野生型(WT)小鼠和具有降低的脑5-HT合成和水平的“5-HTHypo”小鼠(Beaulieu等,2008)。5-HTHypo小鼠是大脑5-HT缺乏症的自然模型,已知它是数种中枢神经系统疾病的致病因子,诸如抑郁和自杀倾向。
药物治疗:卡比多巴用作PDI。为了模拟在治疗过程中人中发生的口服药物传递,通过小鼠食物(标准食物)递送5-HTP和卡比多巴。这种方法随着时间分配了药物,从而提供了“缓释”(也就是持续释放,延长释放,随时间释放,控制释放)的测量方式。5-HTP的剂量为200mg/kg/天。除5-HTP以外,卡比多巴的剂量为2、5或10mg/kg/天,以评估卡比多巴对5-HTP治疗结果的影响。为了评估单独的卡比多巴的作用,还对小鼠的组施用了卡比多巴10mg/kg/天(最高的卡比多巴剂量),但未伴随5-HTP。与未治疗的小鼠进行比较。所有治疗的持续时间为14天,在小鼠实验中被认为是长期的。
样品收集:在治疗期间收集血液样品以评估5-HTP和卡比多巴的血浆水平。治疗结束时对小鼠实施安乐死,并收集脑组织(额叶皮层)。
定量分析:如所述(Jacobsen等,2012b),使用HPLC对血浆5-HTP,血浆卡比多巴,脑5-HT和脑5-羟基吲哚乙酸(5-HIAA,主要的5-HT代谢产物)的水平进行定量。血浆中5-HT和5-HTP的检出限为1ng/ml,血浆中卡比多巴的检出限为2ng/ml(图4C)。
结果与讨论
5-HTP处理可升高血浆5-HTP(图1),脑5-HT(图2)和脑5-HIAA(图3)的水平。单独使用卡比多巴,而未伴随5-HTP治疗,则没有效果。除了5-HTP以外,卡比多巴加倍且以剂量依赖方式地增强了5-HTP治疗对所有结果指标的作用。WT和5-HTHypo小鼠在血浆5-HTP的治疗作用方面没有差异。这是预料之中的,因为5-HTHypo小鼠携带的突变不会影响5-HTP的吸收和代谢(Beaulieu等,2008)。相反,在5-HTHypo小鼠中,对脑5-HT和5-HIAA的5HTP+/-卡比多巴治疗效果相对更明显。这可能部分反映了WT小鼠在基线时已经具有较高的5-HT组织水平。请注意,先前的数据表明,野生型小鼠中5-HT和5-HIAA的组织水平即使很小的升高,都转化为细胞外空间(5-HTExt)中5HT的功能活性池水平的显著升高(Jacobsen等等,2016b)。
总体而言,这些发现表明,这种5-HTP/低剂量卡比多巴治疗方案可能在与脑5-HT功能障碍相关的疾病(例如精神疾病)中特别相关,在5-HT水平正常以及5-HT普遍升高的患者和疾病中也如此,并且增强可能具有治疗作用。血浆卡比多巴水平低于检测极限,即<2ng/ml,甚至在最高剂量10mg/kg/天也是如此。
这表明即使在仅产生血浆暴露水平、至少比通常的全身活性水平低至少12倍的剂量(即25ng/ml,Verhagen等,2015;Yeh等,1989),卡比多巴仍显著提高5-HTP的生物利用度。据认为,潜在的机理是卡比多巴仅在上肠中局部起作用,以保护5-HTP在肠壁上运输期间免受首过代谢。在正常情况下,外源性5-HTP的首过代谢是众所周知的(Shindo等,1977)。此外,由于公认的是5-HTP的不良反应主要是由肠道中的5-HTP转化为5-HT引起的,因此这些数据表明,这种5-HTP/低剂量卡比多巴方案将提高人中5-HTP疗法的安全性和耐受性。
因此,比以前报告的剂量更低的卡比多巴剂量可以增强5-HTP的生物利用度和药效学(脑5-HT)作用。此外,卡比多巴的剂量基本上仅能有效地局部抑制肠道中的PDI活性,可以显著增强5-HTP的生物利用度和药效(脑5-HT)作用。5-HTP:卡比多巴的比例为20:1至100:1,远高于以前的报告,可有效提高5-HTP的生物利用度和药效(脑5-HT)作用。
这些数据证明了用低剂量卡比多巴(例如,约0.17至0.83mg/kg/天的剂量以增强5-HTP生物利用度)与5-HTP的共同治疗的人类治疗方法的可行性。注意,由于小鼠的新陈代谢较高,大约需要人日剂量的12倍才能产生相同的暴露量。因此,小鼠与人之间的“物种间比例因子”为1/12(Sharma和McNeill,2009)。
这些数据还证明了以20:1至100:1的5-HTTP:卡比多巴比例进行共治疗的方法以增强5-HTP生物利用度并减少5-HTP GI不良事件的可行性。
参考文献
Agren H,Reibring L,Hartvig P,Tedroff J,Bjurling P,Hornfeldt K,et al(1991).Low brain uptake of L-[11C]5-hydroxytryptophan in major depression:apositron emission tomography study on patients and healthy volunteers.ActaPsychiatr Scand 83(6):449-455.
Alino JJ,Gutierrez JL,Iglesias ML(1976).5-Hydroxytryptophan(5-HTP)anda MAOI(nialamide)in the treatment of depressions.A double-blind controlledstudy.Int Pharmacopsychiatry 11(1):8-15.
Appleby L,Warner R,Whitton A,Faragher B(1997).A controlled study offluoxetine and cognitive-behavioural counselling in the treatment ofpostnatal depression.Bmj 314(7085):932-936.
Asberg M(1997).Neurotransmitters and suicidal behavior.The evidencefrom cerebrospinal fluid studies.Ann N Y Acad Sci 836:158-181.
Badawy AA,Bano S(2016).Tryptophan Metabolism in Rat Liver AfterAdministration of Tryptophan,Kynurenine Metabolites,and KynureninaseInhibitors.Int J Tryptophan Res 9:51-65.
Bartels C,Wagner M,Wolfsgruber S,Ehrenreich H,Schneider A,Alzheimer'sDisease Neuroimaging I(2018).Impact of SSRI Therapy on Risk of ConversionFrom Mild Cognitive Impairment to Alzheimer's Dementia in Individuals WithPrevious Depression.Am J Psychiatry 175(3):232-241
Beaulieu JM,Zhang X,Rodriguiz RM,Sotnikova TD,Cools MJ,Wetsel WC,etal(2008).Role of GSK3 beta in behavioral abnormalities induced by serotonindeficiency.Proceedings of the National Academy of Sciences of the UnitedStates of America 105(4):1333-1338.
Blier P,Bergeron R(1996).Sequential administration of augmentationstrategies in treatment-resistant obsessive-compulsive disorder:preliminaryfindings.Int Clin Psychopharmacol 11(1):37-44.
Bowsher R.R.HDP(1986).Aromatic L-Amino Acid Decarboxylase.In:BoultonA.A.BGB,Yu P.H.(ed).Neurotransmitter Enzymes.Neuromethods(Series 1:Neurochemistry),.Humana Press.Vol 5.
Brown GL,Ebert MH,Goyer PF,Jimerson DC,Klein WJ,Bunney WE,et al(1982).Aggression,suicide,and serotonin:relationships to CSF aminemetabolites.Am J Psychiatry 139(6):741-746.
Bruni O,Ferri R,Miano S,Verrillo E(2004).L-5-Hydroxytryptophantreatment of sleep terrors in children.Eur J Pediatr 163(7):402-407.
Byerley WF,Judd LL,Reimherr FW,Grosser BI(1987).5-Hydroxytryptophan:areview of its antidepressant efficacy and adverse effects.J ClinPsychopharmacol 7(3):127-137.
Cangiano C,Ceci F,Cascino A,Del Ben M,Laviano A,Muscaritoli M,et al(1992).Eating behavior and adherence to dietary prescriptions in obese adultsubjects treated with5-hydroxytryptophan.Am J Clin Nutr 56(5):863-867.
Caruso I,Sarzi Puttini P,Cazzola M,Azzolini V(1990).Double-blindstudy of5-hydroxytryptophan versus placebo in the treatment of primaryfibromyalgia syndrome.The Journal of international medical research 18(3):201-209.
Carver CS,Johnson SL,Joormann J(2008).Serotonergic function,two-modemodels of self-regulation,and vulnerability to depression:what depression hasin common with impulsive aggression.Psychol Bull 134(6):912-943.
Claxton AJ,Cramer J,Pierce C(2001).A systematic review of theassociations between dose regimens and medication compliance.Clinicaltherapeutics 23(8):1296-1310.
Connor KM,Sutherland SM,Tupler LA,Malik ML,Davidson JR(1999).Fluoxetine in post-traumatic stress disorder.Randomised,double-blindstudy.Br J Psychiatry 175:17-22.
Coric V,Feldman HH,Oren DA,Shekhar A,Pultz J,Dockens RC,et al(2010).Multicenter,randomized,double-blind,active comparator and placebo-controlledtrial of a corticotropin-releasing factor receptor-1 antagonist ingeneralized anxiety disorder.Depress Anxiety 27(5):417-425.
Da Prada M,Kettler R,Zurcher G,Schaffner R,Haefely WE(1987).Inhibition of Decarboxylase and Levels of Dopa and 3-O-Methyldopa:AComparative Study of Benserazide versus Carbidopa in Rodents and of MadoparStandard versus Madopar HBS in Volunteers.Eur Neurol 27(suppl 1):9-20.
Freitas ME,Ruiz-Lopez M,Fox SH(2016).Novel Levodopa Formulations forParkinson's Disease.CNS drugs 30(11):1079-1095.
Fuller RW,Snoddy HD(1980).Effect of serotonin-releasing drugs onserum corticosterone concentration in rats.Neuroendocrinology 31(2):96-100.
Gasser UE,Fischer A,Timmermans JP,Arnet I(2013).Pharmaceuticalquality of seven generic Levodopa/Benserazide products compared with originalMadopar(R)/Prolopa(R).BMC Pharmacol Toxicol 14:24.
Gershon MD(2013).5-Hydroxytryptamine(serotonin)in thegastrointestinal tract.Curr Opin Endocrinol Diabetes Obes 20(1):14-21.
Gijsman HJ,van Gerven JM,de Kam ML,Schoemaker RC,Pieters MS,WeemaesM,et al(2002).Placebo-controlled comparison of three dose-regimens of 5-hydroxytryptophan challenge test in healthy volunteers.J Clin Psychopharmacol22(2):183-189.
Guerdjikova AI,McElroy SL,Kotwal R,Welge JA,Nelson E,Lake K,et al(2008).High-dose escitalopram in the treatment of binge-eating disorder withobesity:a placebo-controlled monotherapy trial.Hum Psychopharmacol 23(1):1-11.
Hsu A,Yao HM,Gupta S,Modi NB(2015).Comparison of the pharmacokineticsof an oral extended-release capsule formulation of carbidopa-levodopa(IPX066)with immediate-release carbidopa-levodopa(Sinemet),sustained-releasecarbidopa-levodopa(Sinemet CR),and carbidopa-levodopa-entacapone(Stalevo).Journal of clinical pharmacology.
Jackson CW,Cates M,Lorenz R(2010).Pharmacotherapy of eatingdisorders.Nutr Clin Pract 25(2):143-159.
Jacobsen JP,Krystal AD,Krishnan KR,Caron MG(2016a).Adjunctive 5-Hydroxytryptophan Slow-Release for Treatment-Resistant Depression:Clinicaland Preclinical Rationale.Trends Pharmacol Sci.
Jacobsen JP,Medvedev IO,Caron MG(2012a).The 5-HT deficiency theory ofdepression:perspectives from a naturalistic 5-HT deficiency model,thetryptophan hydroxylase 2Arg439His knockin mouse.Philos Trans R Soc Lond BBiol Sci 367(1601):2444-2459.
Jacobsen JP,Rudder ML,Roberts W,Royer EL,Robinson TJ,Oh A,et al(2016b).SSRI Augmentation by 5-Hydroxytryptophan Slow Release:MousePharmacodynamic Proof of Concept.Neuropsychopharmacology 41(9):2324-2334.
Jacobsen JP,Siesser WB,Sachs BD,Peterson S,Cools MJ,Setola V,et al(2012b).Deficient serotonin neurotransmission and depression-like serotoninbiomarker alterations in tryptophan hydroxylase 2(Tph2)loss-of-functionmice.Molecular psychiatry 17(7):694-704.
Jacobsen JPRD,NC,US),Caron,Marc G.(Hillsborough,NC,US)(2016).Slow-release formulations of 5-hydroxytryptophan as an adjunct to pro-serotonergictherapies.Duke University(Durham,NC,US):United States.
Kahn RS,Westenberg HG,Verhoeven WM,Gispen-de Wied CC,Kamerbeek WD(1987).Effect of a serotonin precursor and uptake inhibitor in anxietydisorders;a double-blind comparison of 5-hydroxytryptophan,clomipramine andplacebo.Int Clin Psychopharmacol 2(1):33-45.
Lader M,Stender K,Burger V,Nil R(2004).Efficacy and tolerability ofescitalopram in 12-and 24-week treatment of social anxiety disorder:randomised,double-blind,placebo-controlled,fixed-dose study.Depress Anxiety19(4):241-248.
Lopes CM,Bettencourt C,Rossi A,Buttini F,Barata P(2016).Overview ongastroretentive drug delivery systems for improving drug bioavailability.IntJ Pharm 510(1):144-158.
Lowe SL,Yeo KP,Teng L,Soon DK,Pan A,Wise SD,et al(2006).L-5-Hydroxytryptophanaugments the neuroendocrine response to aSSRI.Psychoneuroendocrinology 31(4):473-484.
Manegold C,Hoffmann GF,Degen I,Ikonomidou H,Knust A,Laass MW,et al(2009).Aromatic L-amino acid decarboxylase deficiency:clinical features,drugtherapy and follow-up.J Inherit Metab Dis 32(3):371-380.
Mead GE,Hsieh CF,Lee R,Kutlubaev M,Claxton A,Hankey GJ,et al(2013).Selective serotonin reuptake inhibitors for stroke recovery:a systematicreview and meta-analysis.Stroke;a journal of cerebral circulation 44(3):844-850.
Mitra B,Chang J,Wu SJ,Wolfe CN,Ternik RL,Gunter TZ,et al(2017).Feasibility of mini-tablets as a flexible drug delivery tool.Int J Pharm 525(1):149-159.
Nicolodi M,Sicuteri F(1999).L-5-hydroxytryptophan can preventnociceptive disorders in man.Adv Exp Med Biol 467:177-182.
Nokhodchi A,Raja S,Patel P,Asare-Addo K(2012).The role of oralcontrolled release matrix tablets in drug delivery systems.Bioimpacts 2(4):175-187.
Pahwa R,Lyons KE,Hauser RA,Fahn S,Jankovic J,Pourcher E,et al(2014).Randomized trial of IPX066,carbidopa/levodopa extended release,in earlyParkinson's disease.Parkinsonism Relat Disord 20(2):142-148.
Perry KW,Fuller RW(1993).Extracellular 5-hydroxytryptamineconcentration in rat hypothalamus after administration of fluoxetine plus L-5-hydroxytryptophan.J Pharm Pharmacol 45(8):759-761.
Rauws AG,Vos JG,Garbis-Berkvens JM,Peters PW,de Vries T,van Logten MJ(1982).Comparative 90-day toxicity of two decarboxylase inhibitors,benserazide and carbidopa,in the rat.Toxicol Appl Pharmacol 66(2):201-220.
Rose S,Jenner P,Marsden CD(1988).The effect of carbidopa on plasmaand muscle levels of L-dopa,dopamine,and their metabolites following L-dopaadministration to rats.Mov Disord 3(2):117-125.
Sargent PA,Williamson DJ,Cowen PJ(1998).Brain 5-HT neurotransmissionduring paroxetine treatment.The British journal of psychiatry:the journal ofmental science 172:49-52.
Sharma V,McNeill JH(2009).To scale or not to scale:the principles ofdose extrapolation.Br J Pharmacol 157(6):907-921.
Shindo H,Komai T,Kawai K(1977).Mechanism of intestinal absorption andbrain uptake of L-5-hydroxytryptophan in rats,as compared to those of L-3,4-dihydroxyphenylalanine.Chem Pharm Bull(Tokyo)25(6):1417-1425.
Sloan RL,Brown KW,Pentland B(1992).Fluoxetine as a treatment foremotional lability after brain injury.Brain Inj 6(4):315-319.
Soulairac A,Lambinet H(1977).[Effect of 5-hydroxytryptophan,aserotonin precursor,on sleep disorders].Ann Med Psychol(Paris)1(5):792-798.
Steiner M,Steinberg S,Stewart D,Carter D,Berger C,Reid R,et al(1995).Fluoxetine in the treatment of premenstrual dysphoria.Canadian Fluoxetine/Premenstrual Dysphoria Collaborative Study Group.N Engl J Med 332(23):1529-1534.
Takahashi S,Takahashi R,Masumura I,Miike A(1976).Measurement of5-hydroxyindole compounds during L-5-HTP treatment in depressed patients.FoliaPsychiatr Neurol Jpn 30(4):463-473.
Thombre AG,Appel LE,Chidlaw MB,Daugherity PD,Dumont F,Evans LA,et al(2004).Osmotic drug delivery using swellable-core technology.J ControlRelease 94(1):75-89.
Trouillas P,Brudon F,Adeleine P(1988).Improvement of cerebellarataxia with levorotatory form of 5-hydroxytryptophan.A double-blind studywith quantified data processing.Arch Neurol 45(11):1217-1222.
Turner EH,Loftis JM,Blackwell AD(2006a).Serotonin a la carte:supplementation with the serotonin precursor 5-hydroxytryptophan.PharmacolTher 109(3):325-338.
Turner EH,Loftis JM,Blackwell AD(2006b).Serotonin a la carte:supplementation with the serotonin precursor 5-hydroxytryptophan.PharmacolTher 109(3):325-338.
van Hiele LJ(1980).l-5-Hydroxytryptophan in depression:the firstsubstitution therapy in psychiatry?The treatment of 99 out-patients with'therapy-resistant'depressions.Neuropsychobiology 6(4):230-240.
van Praag HM(1982).Serotonin precursors in the treatment ofdepression.Advances in biochemical psychopharmacology 34:259-286.
Van Woert MH,Rosenbaum D,Howieson J,Bowers MB,Jr.(1977).Long-termtherapy of myoclonus and other neurologic disorders with L-5-hydroxytryptophan and carbidopa.The New England journal of medicine 296(2):70-75.
Veenstra-VanderWeele J,Muller CL,Iwamoto H,Sauer JE,Owens WA,Shah CR,et al(2012).Autism gene variant causes hyperserotonemia,serotonin receptorhypersensitivity,social impairment and repetitive behavior.Proceedings of theNational Academy of Sciences of the United States of America 109(14):5469-5474.
Verhagen Metman L,Stover N,Chen C,Cowles VE,Sweeney M(2015).Gastroretentive carbidopa/levodopa,DM-1992,for the treatment of advancedParkinson's disease.Mov Disord 30(9):1222-1228.
Viscogliosi G,Chiriac IM,Ettorre E(2017).Efficacy and Safety ofCitalopram Compared to Atypical Antipsychotics on Agitation in Nursing HomeResidents With Alzheimer Dementia.J Am Med Dir Assoc 18(9):799-802.
Westenberg HG,Gerritsen TW,Meijer BA,van Praag HM(1982).Kinetics ofl-5-hydroxytryptophan in healthy subjects.Psychiatry Res 7(3):373-385.
Yeh KC,August TF,Bush DF,Lasseter KC,Musson DG,Schwartz S,et al(1989).Pharmacokinetics and bioavailability of Sinemet CR:a summary of humanstudies.Neurology 39(11Suppl 2):25-38.
Yoshimura M,Kambara S,Takahashi H,Okabayashi H,Ijichi H(1987).Involvement of dopamine in development of hypertension in spontaneouslyhypertensive rat:effect of carbidopa,inhibitor of peripheral dopadecarboxylase.Clin Exp Hypertens A 9(10):1585-1599.
本说明书中提及的任何专利或出版物都说明本发明所属领域的技术人员的水平。这些专利和出版物在本文中以引用方式并入,其程度如同每一个单独的出版物被明确地和单独地说明为通过引用并入一样。如有冲突,以本说明书包括定义为准。
本领域技术人员将容易理解,本发明很好地适于实现所述目的并获得所述的目标和优势,以及其中固有的那些。本文所描述的本发明目前通过优选实施方案代表,其是示例性的,并不意图作为对本发明范围的限制。本领域技术人员将理解如权利要求书的范围所界定的本发明的精神中所包含的变化和其他应用。
Claims (30)
1.一种在需要其的人类受试者中增强肠内施用的5-HTP的生物利用度的方法,所述方法包括:
将低剂量卡比多巴与所述5-HTP肠内共同施用,所述低剂量卡比多巴的日剂量为约0.1或0.2至约0.5、0.6或0.8mg/kg/天(或每天约5或10mg至约35、50或60mg)
从而提高肠内施用的5-HTP的生物利用度。
2.根据权利要求1所述的方法,其中所述5-HTP和低剂量卡比多巴以100:1至20:1的5-HTTP:卡比多巴的日剂量比例施用。
3.根据权利要求1或权利要求2所述的方法,其中在肠内共同施用所述低剂量卡比多巴时,所述受试者的卡比多巴血浆水平小于25、20、15、10、5或2ng/ml。
4.根据前述权利要求中任一项所述的方法,其中所述5-HTP和低剂量卡比多巴每天施用一次,两次或三次。
5.根据前述权利要求中任一项的方法,其中5-HTP和/或低剂量卡比多巴的制剂以选自口服速释制剂、口服缓释制剂、口服肠内凝胶、直肠栓剂及其组合的方式施用。
6.根据前述权利要求中任一项所述的方法,其中所述5-HTP和低剂量卡比多巴以相同或分开的剂型在5-HTP和低剂量卡比多巴的缓释(SR)制剂中提供。
7.根据前述权利要求中任一项所述的方法,其中所述SR制剂是胃滞留制剂。
8.根据权利要求7所述的方法,其中所述5-HTP和低剂量卡比多巴与膳食共同施用。
9.根据前述权利要求中任一项的方法,其中所述受试者需要治疗抑郁、社交焦虑症、恐慌症、广泛性焦虑症、强迫症(OCD)、冲动控制障碍、自杀倾向、边缘性人格障碍、纤维肌痛、共济失调、与神经疾病(例如阿尔茨海默氏症、帕金森氏症)有关的情绪、认知、或行为症状和躁动、中风恢复、自闭症、偏头痛、睡眠障碍、经前烦躁不安、创伤后应激障碍、产后抑郁或干扰素治疗后抑郁。
10.根据前述权利要求中任一项所述的方法,其中所述受试者需要治疗精神疾病和/或神经疾病,例如情绪异常或冲动或攻击控制中的异常。
11.根据前述权利要求中任一项所述的方法,其中所述受试者需要治疗抑郁、焦虑、自杀倾向、强迫症或ADHD。
12.根据前述权利要求中任一项所述的方法,其中所述受试者需要治疗抑郁症、重度抑郁症或抗治疗性抑郁症。
13.根据前述权利要求中任一项的方法,其中所述方法包括用5-HTP和卡比多巴进行单药治疗。
14.根据前述权利要求中任一项所述的方法,其中所述方法包括使用5-HTP和卡比多巴以及5-羟色胺增强剂的辅助疗法。
15.根据权利要求14所述的方法,其中所述5-羟色胺增强剂选自5-羟色胺再摄取抑制剂、5-羟色胺去甲肾上腺素再摄取抑制剂、三重再摄取抑制剂、单胺氧化酶抑制剂、三环抗抑郁药、5-羟色胺激动剂、苯丙胺、5-羟色胺前体、5-羟色胺原药、5-羟色胺生物合成中的中间体,及其药学上可接受的盐。
16.根据权利要求14所述的方法,其中所述5-羟色胺增强剂是选择性5-羟色胺再摄取抑制剂(SSRI)。
17.根据权利要求14所述的方法,其中所述5-羟色胺增强剂选自以下组成的组:西酞普兰、达泊西汀、依西酞普兰、氟西汀、氟伏沙明、吲哚平、帕罗西汀、舍曲林、维拉唑酮、伏地西汀、齐美替丁及其组合。
18.根据前述权利要求中任一项所述的方法,其中所述5-HTP和低剂量卡比多巴以相同或分开的剂型口服施用。
19.根据前述权利要求中任一项所述的方法,其中所述低剂量卡比多巴以每天约0.1或0.2至约0.5mg/kg/天(或每天约5或10mg至约35mg)的日剂量提供。
20.根据前述权利要求中任一项所述的方法,其中以每天约0.1、0.2、0.5或0.75克至每天约1、4或6克的日剂量提供所述5-HTP。
21.适合肠内施用的药物制剂或部件试剂盒,其包含5-HTP和低剂量卡比多巴,所述制剂适合每日一次、两次或三次施用。
22.根据权利要求21所述的药物制剂或部件试剂盒,其中所述低剂量卡比多巴以下述日剂量提供:约0.1或0.2mg/kg/天至约0.5、0.6或0.8mg/kg/天(或每天约5或10mg至每天约35、50或60mg)。
23.根据权利要求21或22所述的药物制剂或部件试剂盒,其中所述5-HTP和低剂量卡比多巴在所述剂型中以100∶1至20∶1的5-HTP∶卡比多巴的剂量比提供。
24.根据权利要求21-23中任一项所述的药物制剂或部件试剂盒,其中所述制剂是适合于口服或直肠施用的固体剂型。
25.根据权利要求21至24中任一项所述的药物制剂或部件试剂盒,其中5-HTP和/或低剂量卡比多巴的制剂是一个药片、一个胶囊或其他每日一次剂量的制剂;和或两个药片、两个胶囊或其它每天两次剂量(合计为每日剂量)的制剂。
26.根据权利要求21-25中任一项所述的药物制剂或部件试剂盒,其中所述药物制剂或部件试剂盒包括5-HTP和低剂量卡比多巴的缓释(SR)制剂。
27.根据权利要求26所述的方法,其中所述SR制剂是胃滞留制剂。
28.根据权利要求27所述的方法,其中所述5-HTP和低剂量卡比多巴与膳食共同施用。
29.低剂量卡比多巴在增强5-HTP在人类受试者中的生物利用度的方法中的用途,所述方法包括向该受试者肠内共同施用低剂量的卡比多巴与5-HTP、或由上述步骤组成、或基本上由上述步骤组成,所述低剂量卡比多巴以约0.1或0.2至约0.5、0.6或0.8mg/kg/天(或每天约5或10mg至约35、50或60mg)的日剂量提供,从而增强所述受试者中5-HTP的生物利用度。
30.低剂量卡比多巴在制备用于增强人类受试者中5-HTP的生物利用度的药物的方法中的用途,所述低剂量卡比多巴以约0.1或0.2至约0.5、0.6或0.8mg/kg/天(或每天约5或10mg至约35、50或60mg)的每日剂量提供用于肠内施用。
Applications Claiming Priority (5)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| US201862623073P | 2018-01-29 | 2018-01-29 | |
| US62/623,073 | 2018-01-29 | ||
| US201862743816P | 2018-10-10 | 2018-10-10 | |
| US62/743,816 | 2018-10-10 | ||
| PCT/US2019/015391 WO2019148087A1 (en) | 2018-01-29 | 2019-01-28 | Compositions and methods of enhancing 5-hydroxytryptophan bioavailability |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| CN111902137A true CN111902137A (zh) | 2020-11-06 |
Family
ID=67395723
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| CN201980021717.5A Pending CN111902137A (zh) | 2018-01-29 | 2019-01-28 | 增强5-羟色氨酸的生物利用度的组合物和方法 |
Country Status (14)
| Country | Link |
|---|---|
| US (1) | US11337963B2 (zh) |
| EP (1) | EP3746058B1 (zh) |
| JP (2) | JP2021511365A (zh) |
| KR (1) | KR20200116110A (zh) |
| CN (1) | CN111902137A (zh) |
| AU (1) | AU2019211458A1 (zh) |
| BR (1) | BR112020015068A2 (zh) |
| CA (1) | CA3089068A1 (zh) |
| ES (1) | ES2948788T3 (zh) |
| IL (1) | IL276059B1 (zh) |
| MX (1) | MX2020007760A (zh) |
| RU (1) | RU2020125170A (zh) |
| SG (1) | SG11202007178VA (zh) |
| WO (1) | WO2019148087A1 (zh) |
Families Citing this family (3)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| EP4362919A1 (en) * | 2021-07-30 | 2024-05-08 | Evecxia Therapeutics, Inc. | 5-hydroxytryptophan gastroretentive dosage forms |
| WO2023064598A1 (en) | 2021-10-14 | 2023-04-20 | Evecxia Therapeutics, Inc. | A method for optimizing 5-hydroxytryptamine function in the brain for therapeutic purposes |
| DE102022103658A1 (de) * | 2022-02-16 | 2023-08-17 | Joachim Kamprad | Arzneimittelzusammensetzung enthaltend L-Tryptophan (L-Try) und L-5-Hydroxytryptophan (5-HTP) sowie einen peripheren Abbauhemmer |
Citations (4)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO1991007960A1 (fr) * | 1989-11-24 | 1991-06-13 | Paul Trouillas | Compositions pharmaceutiques destinees au traitement de l'ataxie et les troubles de l'equilibre |
| US6207699B1 (en) * | 1999-06-18 | 2001-03-27 | Richard Brian Rothman | Pharmaceutical combinations for treating obesity and food craving |
| US9468627B2 (en) * | 2010-10-22 | 2016-10-18 | Duke University | Slow-release formulations of 5-hydroxytryptophan as an adjunct to pro-serotonergic therapies |
| US20160375136A1 (en) * | 2010-02-05 | 2016-12-29 | Phosphagenics Limited | Carrier composition |
Family Cites Families (13)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US4658038A (en) | 1982-10-07 | 1987-04-14 | Research Foundation For Mental Hygiene, Inc. | N-acylated 5-hydroxytryptophan amide derivatives |
| US6635280B2 (en) | 1997-06-06 | 2003-10-21 | Depomed, Inc. | Extending the duration of drug release within the stomach during the fed mode |
| JP4083818B2 (ja) | 1997-06-06 | 2008-04-30 | ディポメド,インコーポレイティド | 高度可溶性薬物の制御された放出のための胃滞留性の経口薬物投与形 |
| TWI312285B (en) | 2001-10-25 | 2009-07-21 | Depomed Inc | Methods of treatment using a gastric retained gabapentin dosage |
| CA2409552A1 (en) | 2001-10-25 | 2003-04-25 | Depomed, Inc. | Gastric retentive oral dosage form with restricted drug release in the lower gastrointestinal tract |
| US6723340B2 (en) | 2001-10-25 | 2004-04-20 | Depomed, Inc. | Optimal polymer mixtures for gastric retentive tablets |
| US20060013875A1 (en) | 2002-05-29 | 2006-01-19 | Impax Laboratories, Inc. | Combination immediate release controlled release levodopa/carbidopa dosage forms |
| US7101912B2 (en) | 2002-12-06 | 2006-09-05 | Xenoport, Inc. | Carbidopa prodrugs and derivatives, and compositions and uses thereof |
| EP1751312A4 (en) * | 2004-05-21 | 2010-01-27 | Univ Duke | POLYMORPHISM IN TRYPTOPHAN HYDROXYLASE 2 REGULATING BRAIN SEROTONIN SYNTHESIS |
| US20070213370A1 (en) * | 2005-11-18 | 2007-09-13 | H. Lundbeck A/S | 5-HTP Combination Therapy |
| WO2009043834A1 (en) | 2007-10-01 | 2009-04-09 | Neurosearch A/S | Pharmaceutical compositions of 5-hydr0xytrypt0phan and serotonin-enhancing compound |
| MX340249B (es) | 2008-08-15 | 2016-07-01 | Depomed Inc | Composiciones farmacéuticas retentivas gástricas para tratamiento y prevención de transtornos del sistema nervioso central (cns). |
| CN103554005A (zh) | 2013-11-22 | 2014-02-05 | 长沙理工大学 | 一种左旋5-羟基色氨酸的简便合成新方法 |
-
2019
- 2019-01-28 EP EP19743636.3A patent/EP3746058B1/en active Active
- 2019-01-28 ES ES19743636T patent/ES2948788T3/es active Active
- 2019-01-28 KR KR1020207024221A patent/KR20200116110A/ko not_active Application Discontinuation
- 2019-01-28 JP JP2020541352A patent/JP2021511365A/ja active Pending
- 2019-01-28 US US15/733,423 patent/US11337963B2/en active Active
- 2019-01-28 WO PCT/US2019/015391 patent/WO2019148087A1/en unknown
- 2019-01-28 CN CN201980021717.5A patent/CN111902137A/zh active Pending
- 2019-01-28 MX MX2020007760A patent/MX2020007760A/es unknown
- 2019-01-28 BR BR112020015068-1A patent/BR112020015068A2/pt unknown
- 2019-01-28 SG SG11202007178VA patent/SG11202007178VA/en unknown
- 2019-01-28 RU RU2020125170A patent/RU2020125170A/ru unknown
- 2019-01-28 AU AU2019211458A patent/AU2019211458A1/en active Pending
- 2019-01-28 CA CA3089068A patent/CA3089068A1/en active Pending
- 2019-01-28 IL IL276059A patent/IL276059B1/en unknown
-
2023
- 2023-07-21 JP JP2023119314A patent/JP2023153871A/ja active Pending
Patent Citations (4)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO1991007960A1 (fr) * | 1989-11-24 | 1991-06-13 | Paul Trouillas | Compositions pharmaceutiques destinees au traitement de l'ataxie et les troubles de l'equilibre |
| US6207699B1 (en) * | 1999-06-18 | 2001-03-27 | Richard Brian Rothman | Pharmaceutical combinations for treating obesity and food craving |
| US20160375136A1 (en) * | 2010-02-05 | 2016-12-29 | Phosphagenics Limited | Carrier composition |
| US9468627B2 (en) * | 2010-10-22 | 2016-10-18 | Duke University | Slow-release formulations of 5-hydroxytryptophan as an adjunct to pro-serotonergic therapies |
Non-Patent Citations (1)
| Title |
|---|
| 张建平等: "《实用神经病学(第二版)》", vol. 1, 科学技术文献出版社, pages: 1058 - 171 * |
Also Published As
| Publication number | Publication date |
|---|---|
| EP3746058C0 (en) | 2023-06-07 |
| US20210346345A1 (en) | 2021-11-11 |
| IL276059A (en) | 2020-08-31 |
| MX2020007760A (es) | 2020-10-28 |
| AU2019211458A1 (en) | 2020-08-06 |
| SG11202007178VA (en) | 2020-08-28 |
| IL276059B1 (en) | 2024-04-01 |
| EP3746058A4 (en) | 2021-12-22 |
| JP2021511365A (ja) | 2021-05-06 |
| EP3746058B1 (en) | 2023-06-07 |
| WO2019148087A1 (en) | 2019-08-01 |
| JP2023153871A (ja) | 2023-10-18 |
| KR20200116110A (ko) | 2020-10-08 |
| US11337963B2 (en) | 2022-05-24 |
| ES2948788T3 (es) | 2023-09-19 |
| CA3089068A1 (en) | 2019-08-01 |
| RU2020125170A (ru) | 2022-02-28 |
| BR112020015068A2 (pt) | 2021-01-05 |
| EP3746058A1 (en) | 2020-12-09 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| Kadriu et al. | Glutamatergic neurotransmission: pathway to developing novel rapid-acting antidepressant treatments | |
| Palucha et al. | Metabotropic glutamate receptor ligands as possible anxiolytic and antidepressant drugs | |
| Cardinali et al. | Melatonin and its analogs in insomnia and depression | |
| Ferreira et al. | Summary of the recommendations of the EFNS/MDS‐ES review on therapeutic management of P arkinson's disease | |
| Tokita et al. | Roles of glutamate signaling in preclinical and/or mechanistic models of depression | |
| Turner et al. | Serotonin a la carte: supplementation with the serotonin precursor 5-hydroxytryptophan | |
| Serafini et al. | Pharmacological properties of glutamatergic drugs targeting NMDA receptors and their application in major depression | |
| Poleszak et al. | A complex interaction between glycine/NMDA receptors and serotonergic/noradrenergic antidepressants in the forced swim test in mice | |
| US20110230513A1 (en) | Pharmaceutical compositions for treatment of parkinsons disease | |
| EP3746058B1 (en) | Compositions and methods of enhancing 5-hydroxytryptophan bioavailability | |
| MX2008014840A (es) | Tratamiento de trastornos depresivos. | |
| Oertel et al. | Early (uncomplicated) Parkinson’s disease | |
| US20090264522A1 (en) | Methods of treating central nervous system disorders with a low dose combination of escitalopram and bupropion | |
| JP2017534613A (ja) | デクスメデトミジン製剤を使用する睡眠障害の予防または治療 | |
| EP2145620A2 (en) | Gaboxadol for treating depression and other affective disorders | |
| Capuano et al. | Flupirtine antinociception in the rat orofacial formalin test: an analysis of combination therapies with morphine and tramadol | |
| EP4319759A1 (en) | 1-methylxanthine-based bioactive composition and method of use thereof | |
| AU2005270573A1 (en) | Novel formulation for L-tryptophane comprising carbidopa/benserazide | |
| da Silva Leal et al. | Depletion of 5 hydroxy-triptamine (5-HT) affects the antidepressant-like effect of neuronal nitric oxide synthase inhibitor in mice | |
| Celikyurt et al. | Serotonin noradrenaline reuptake inhibitors (SNRIs) | |
| Faro et al. | Possible synergies between isatin, an endogenous MAO inhibitor, and antiparkinsonian agents on the dopamine release from striatum of freely moving rats. | |
| WO2015059638A2 (en) | Cns stimulant and opioid receptor antagonist combination as a non- addictive, non-aversive and synergistic anti-obesity treatment | |
| Müller | Safinamide for symptoms of Parkinson’s disease | |
| Lv et al. | Application of the concept of continuous dopaminergic stimulation for the management of Parkinson’s disease | |
| ZA200509356B (en) | Gaboxadol for treating depression and other affective disorders |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| PB01 | Publication | ||
| PB01 | Publication | ||
| SE01 | Entry into force of request for substantive examination | ||
| SE01 | Entry into force of request for substantive examination |