Summary of the invention
The purpose of this invention is to provide a kind of small thorowax droplet with the preparation of new type natural substrate adjuvant.
Another object of the present invention provides a kind of preparation method of small thorowax droplet pharmaceutical preparation.
The selected substrate adjuvant of the present invention is resulting by a large amount of tests, it is little to have molecular weight, soluble in water, and molten diffusing speed is faster, pure natural degree height, toxic and side effects is lower, and can reduce the medicine irritation abnormal smells from the patient, has the oral cavity of improvement acid-base value during the buccal of oral cavity, improve the characteristics of oral cavity smell, the used substrate adjuvant of the present invention is the agent of food sedan-chair flavor, takes that mouthfeel is good, the acceptant characteristics of patient, is the direction of following substrate adjuvant development.
Drug component of the present invention, the selection of consumption and adjuvant thereof also gropes to sum up to draw through the inventor in a large number, each amounts of components all has curative effect preferably in following ranges: Radix Bupleuri 1~5%, Rhizoma Pinelliae Preparatum 15~20%, Radix Scutellariae 15~20%, Radix Codonopsis 15~20%, Fructus Jujubae 15~20%, Radix Glycyrrhizae 10~15%, Rhizoma Zingiberis Recens 8~13%, above-mentioned medical material is 0.1: 1~1: 1 through the weight ratio of extracting resulting extract and adjuvant, wherein adjuvant comprises filler and plasticity substrate, said filler is selected from the natural adjuvant of following one or more plant origins: erythritol, sorbitol, fructose, D-ribonic acid-gamma lactone, arabitol, trehalose, D-ribose, low melting-point agarose, Lac, xylitol, Raffinose, glucose, malic acid, citric acid, isomalt, lactose, maltose etc., and they contain the water of crystallization chemical compound; Said plasticity substrate is selected from the natural adjuvant of following one or more plant origins: starch and derivant thereof, cellulose and derivant thereof, arabic gum, dextran, chitin, sesbania gum, carrageenan, Ficus elastica, Furcellaran, tragakanta, carrageenin, tamarind gum, pectin, xanthan gum, alginic acid and salt thereof, dextrin, cyclodextrin, agar, lactose; Described starch and derivant thereof such as pregelatinized Starch, modified starch, hydroxypropyl starch, carboxymethyl starch, described cellulose and derivant thereof such as methylcellulose, microcrystalline Cellulose, sodium carboxymethyl cellulose, hydroxypropyl emthylcellulose, cross-linking sodium carboxymethyl cellulose, hydroxyethylmethyl-cellulose, hydroxyethyl-cellulose, hydroxypropyl cellulose; Preferred drug component consumption of the present invention and adjuvant thereof be chosen as Radix Bupleuri 3.7%, Rhizoma Pinelliae Preparatum 18.5%, Radix Scutellariae 18.5%, Radix Codonopsis 18.5%, Fructus Jujubae 18.5%g, Radix Glycyrrhizae 12.3%, Rhizoma Zingiberis Recens 10%, above-mentioned medical material is 0.1: 1~0.6: 1 through the weight ratio of extracting resulting extract and adjuvant, and filler adjuvant wherein is selected from following one or more the natural adjuvant of plant origin: sorbitol, xylitol, lactose, maltose, and they contain the water of crystallization chemical compound; Plasticity substrate wherein is selected from following one or more the natural adjuvant of plant origin: pregelatinized Starch, carboxymethyl starch, methylcellulose, sodium carboxymethyl cellulose, hydroxypropyl emthylcellulose, arabic gum, alginic acid, dextrin, cyclodextrin, agar, lactose; Best drug component consumption of the present invention and adjuvant thereof to be chosen as the weight ratio of extracting resulting drug extract and adjuvant be 0.2~1: 0.4: 1, filler adjuvant wherein was selected from following one or more the natural adjuvant of plant origin: xylitol, lactose; Plasticity substrate wherein is selected from following one or more the natural adjuvant of plant origin: starch, arabic gum.
Can also contain chemosynthesis adjuvant and animal origin adjuvant in the above-mentioned dressing, wherein filler comprises phenylglycol, hexadecanol, octadecanol, sodium stearate, tristerin, tripalmitin, carbamide, polyoxyethylene monostearate, polyoxyethylene alkyl ether; Wherein plasticity substrate comprises polyvinylpyrrolidone, crospolyvinylpyrrolidone, carbomer, polyvinyl alcohol, acrylic resin, poloxamer, gelatin.
In screening to above adjuvant, we find: plant colloid such as carrageenan, the tragakanta, pectin, agar, arabic gum, Ficus elastica, tamarind gum, locust bean gum, Pseudobulbus Bletillae (Rhizoma Bletillae) glue, guar gum, Konjac glucomannan, it is big that plant colloids such as POLY-karaya have viscosity, mobile poor, characteristics such as do not solidify after the condensation, and arabic gum has high dense low sticking character, can be mixed with the aqueous solution of 50% concentration and still have flowability, this is one of not available characteristics of other hydrophilic colloid, arabic gum has at high temperature, under the low concentration, can ooze, but not condensation, at low temperature, under the high concentration, be difficult for oozing, but characteristics such as energy condensation.Polysaccharide such as polysaccharide such as starch and derivant thereof (as gelling starch, carboxymethyl starch etc.), cellulose derivative (as methylcellulose, sodium carboxymethyl cellulose, hydroxypropyl emthylcellulose etc.), alginic acid, dextrin, cyclodextrin, lactose, in screening, find alginic acid have viscosity big, be the fruit jelly sample, dextrin has the colloid sample, characteristics such as lactose coagulability difference; And starch and derivant thereof are materials commonly used in the medical science adjuvant, thus in polysaccharide preferred starch and derivant thereof.Polyhydric alcohol such as sorbitol (88~102 ℃), xylitol (88~94.5 ℃), lactose (70~80 ℃), mannitol (166~169 ℃), maltose alcohol (135~140 ℃), isomalt polyhydric alcohol such as (98~103 ℃) screen, and find that it has following characteristics as drop pill substrate: sorbitol, lactose, isomalt are mobile poor; Mannitol, maltose alcohol fusing point are too high; The xylitol coagulability is poor slightly.After Preliminary screening, preferred xylitol, lactose, sorbitol in the selection of polyhydric alcohol, the best is an xylitol.Xylitol has following characteristics as drop pill substrate: in the time of 91 ℃, molten condition has appearred in xylitol, but not fusion fully, cooling rapidly, it separates out crystallization very soon, xylitol mixes the back good fluidity with extractum at certain proportion, can drip and can condensation, but be condensed into Powdered thing, loosely organized, the toughness extreme difference, that pinches is promptly broken.Organic acid and salt, alkali such as citric acid (100 ℃), sorbic acid (133 ℃), succinic acid (181~189 ℃), sodium acetate organic acid such as (58 ℃) and salt, alkali, its as drop pill substrate have fusing point too high, with Chinese medical concrete can't mixing etc. shortcoming.
Because of above single adjuvant existing shortcoming in as the drop pill preparation process, particularly we by above-mentioned Preliminary screening after, determine two kinds of adjuvants are used and screen: mainly be that above various adjuvants are carried out combined sorting, final determine following several: the plant colloid cooperates with the plant colloid, the cooperating of polyhydric alcohol and polyhydric alcohol, polyhydric alcohol and plant colloidally cooperate, the cooperating of the cooperating of xylitol and arabic gum, lactose and arabic gum, based on the composite auxiliary material of xylitol.Find preferred the cooperation to be xylitol, lactose and the compound use of other adjuvant, this kind combination has following characteristics: make up with mannitol: can drip not condensation; Make up with sorbic acid: both do not dissolve each other; Make up with lactose: can drip the energy condensation, but frangible; Make up with pomelo-pectin, tragakanta, sodium alginate: viscosity is big, can't drip; Make up with arabic gum: can drip, coagulability is poor slightly; Make up with dextrin: can drip, coagulability is poor slightly; Make up with starch: can drip, coagulability is also better.Determine that at last best of breed is that xylitol cooperates with arabic gum with starch, xylitol with starch, lactose.
At xylitol and starch, lactose and starch, in the research of xylitol and arabic gum combination, xylitol and starch Application of composite being prepared some required in the process of drop pill factors investigated, mainly is to the xylitol type, condensed fluid, condensate temperature influences the drop pill mouldability, xylitol and starch proportion influence mouldability, temperature is to the influence of drop pill mouldability, the extractum amount influences the drop pill mouldability, mixing time influences the drop pill mouldability, the dropper bore is to the influence of drop pill particle diameter, the formulation optimization of drop pill, the Preliminary Determination of drop pill, dissolve scattered time limit is investigated.Find that the solid xylitol has three types of powder, granular and crystallinity, and the easiest fusion of powder xylitol, again can fine dissolving be dispersed in the mixed liquor that starch, extractum forms, good fluidity, drippage is easy, and granular and crystalline xyhose alcohol is difficult for fusion, solubility property is also slightly poor, the mix flow that they and starch, extractum form is relatively poor, viscosity is very big, almost cannot drip, and therefore drips first-selected powder xylitol in the system process at drop pill.
At ratio of adjuvant molding is found in the sex research, in the combination of xylitol and starch, lactose and starch, xylitol and arabic gum, low melting point substrate adjuvant is 1: 0~1: 1.5 with the ratio of the weight of plasticity substrate adjuvant, be preferably 1: 0.1~1: 0.9, the best is 1: 0.1~1: 0.5.Low melting point substrate adjuvant of being formed within this scope and plasticity substrate adjuvant, the drug matrices fused solution all can ooze, and can condensation.Specific to each combination, xylitol is preferably 1: 0.2 with the ratio of the weight of starch~1: 0.3, and lactose is preferably 1: 0.2 with the ratio of the weight of starch~1: 0.3, and the ratio of the weight of xylitol and arabic gum is preferably 1: 0.2~and 1: 0.4.Find that in the research of temperature temperature is big especially to the influence of drop pill mouldability to the influence of drop pill mouldability, when temperature is too low, owing to the too big effect that oozes that influences drop pill of viscosity of substrate, when temperature is too high, not condensation of drop pill.Find that mixing time can have influence on the mouldability of drop pill in mixing time in to the sex research of drop pill molding, mixing time is too short, and mobile poor, influence oozes, and mixing time is oversize, influences the condensation of drop pill.Dripping under the system temperature, mixing time in 1~120 minute all can, suitable mixing time was at 10~30 minutes.Consider that mixing time can not be too short in the suitability for industrialized production, adopt the method that low temperature stirs for a long time, high temperature drips system.Find that in the research of dropper bore to the influence of drop pill particle diameter the dropper bore influences the size of drop pill and the flowability of fusion substrate, the system effect is dripped in influence.Drop pill diminishes and diminishes along with bore, but after 1.4 millimeters, along with the bore change of size that diminishes is not obvious, but the matrix flow reduction, system is dripped in influence.
So in the preparation method of preparation, medicine mixes mixing time with the substrate adjuvant be 10~30 minutes; The mixed heating and melting temperature of medicine and substrate adjuvant is 45~115 ℃, dripping the system temperature is 45~95 ℃, and liquid coolant is liquid paraffin, methyl-silicone oil or vegetable oil (Oleum Glycines, Semen Ricini wet goods), and the temperature of liquid coolant is-20~25 ℃, dropper mouth internal diameter is 1.0~4.0 millimeters; Preferred heating and melting temperature is 60~85 ℃, and dripping a system temperature is 60~85 ℃, and condensing agent is liquid paraffin, methyl-silicone oil, and the condensing agent temperature is 0~18 ℃, and the dropper bore is 1.1~3.5 millimeters, and the difference of dropper mouth external diameter and internal diameter is less for well; Best heating and melting temperature is that to make temperature be that 64 ℃, dropper bore are that 1.2~2.5 millimeters, condensing agent are 0 ℃ methyl-silicone oil to 64 ℃, droplet.
The substrate adjuvant of the best of the present invention is xylitol and starch, and xylitol is 1: 0.2~1: 0.3 with the ratio of the weight of starch; Or be lactose and starch, lactose is 1: 0.2~1: 0.3 with the ratio of the weight of starch; Or be xylitol and arabic gum, the ratio of the weight of xylitol and arabic gum is 1: 0.2~1: 0.4.
Xylitol is a kind of natural plant sweetening agent, approve through World Health Organization (WHO), xylitol is a kind of safest sweeting agent, countries in the world are extensive use of in fields such as food and oral-cavity articles, xylitol enters the help that need not insulin in the cell, when sugar utilizes obstacle, can not cause blood sugar increasing yet, can improve diabetics symptom, have the ketoplastic effect of powerful inhibition, can promote the generation of liver glycogen, directly infiltrate the Developmental and Metabolic Disorder that tissue is participated in metabolism, can be corrected protein, fat and steroid; Xylose is the internal metabolism intermediate product, and body has higher toleration to it.Clinical practice proves: the highest oral dosis tolerata can reach 220g every day, and intravenous drip every day can reach 100g.The oral 25700mg/Kg of median lethal dose(LD 50) (LD50) mice, quiet notes 6400mg/Kg, the quiet notes of rat 6200mg/Kg.
Medicine mesostroma adjuvant of the present invention and amount of drug are than can being the scope that allows on the galenic pharmacy, medicine described here can be that crude drug also can be the effective ingredient extract, in order to adapt to industrialized great production, the ratio range of mesostroma adjuvant of the present invention and medicine refers to the weight proportion of adjuvant and extract drugs extractum, and the substrate adjuvant is 1: 0.1~1: 1 with the ratio of the weight of drug extract; Preferred substrate adjuvant is 1: 0.1~1: 0.6 with the ratio of the weight of extract drugs extractum; Best substrate adjuvant is 1: 0.2~1: 0.4 with the ratio of the extraction extractum weight of medicine.
Medicine of the present invention can adopt the preparation of Chinese medicine preparation conventional method.The preparation of effective ingredient of the present invention can be adopted following method: water extraction, decoction and alcohol sedimentation technique, extraction, infusion process, percolation, reflux extraction, continuous backflow extraction method, macroreticular resin absorbing method preparation.For example, these crude drug pulverize mix homogeneously can be made powder takes after mixing it with water; Also can be with these medicines decocting together, the condensed water decocting liquid is made oral liquid then; But, preferably adopt following technology to extract, but this can not limit protection scope of the present invention to raw material in order to make each crude drug of this medicine better bring into play drug effect.
The preparation method of medicine of the present invention is as follows:
(a) get Radix Bupleuri 1~5%, Rhizoma Pinelliae Preparatum 15~20%, Radix Scutellariae 15~20%, Radix Codonopsis 15~20%, Fructus Jujubae 15~20%, Radix Glycyrrhizae 10~15%, Rhizoma Zingiberis Recens 8~13% is standby;
(b) get Radix Bupleuri, add water distillation and extraction, volatile oil and decocting liquid, with last medicinal residues extracting in water, merge with the decocting liquid that begins to distill gained, suitably concentrate, put coldly, add ethanol precipitation, leave standstill, get supernatant concentration and reclaim ethanol and get concentrated solution (1), standby; Follow the example of the Rhizoma Pinelliae and Rhizoma Zingiberis Recens, add ethanol percolation, concentrated percolate gets concentrated solution (2), and is standby; Get Radix Codonopsis, Fructus Jujubae and Radix Glycyrrhizae, extracting in water adds ethanol precipitation, leaves standstill, and gets supernatant concentration recovery ethanol and gets concentrated solution (3), and is standby; Get Radix Scutellariae, extracting in water, extracting solution add dilute hydrochloric acid and regulate pH value, are incubated, and leave standstill, and filter, and precipitation is regulated pH value with the second alcohol and water, and vacuum drying gets Radix Scutellariae extract; (1), (2), (3) concentrated solution of gained are merged, continue to be condensed into thick paste (4);
(c) in appropriate amount of auxiliary materials, add above-mentioned thick paste (4), Radix Scutellariae extract, Radix Bupleuri volatile oil fully mixes, mixture is at 45~115 ℃ of heating and meltings, stir, mixing time is 1~120 minute, insulation, at 45~95 ℃ of temperature following system, dropper bore is 1.0~4.0 millimeters, splash in-20~25 ℃ liquid paraffin, methyl-silicone oil or the vegetable oil, make drop pill, promptly.
Preferred process for preparing medicine of the present invention comprises the following steps:
(a) take off by recipe quantity and state medical material: Radix Bupleuri 3.7%, Rhizoma Pinelliae Preparatum 18.5%, Radix Scutellariae 18.5%, Radix Codonopsis 18.5%, Fructus Jujubae 18.5%g, Radix Glycyrrhizae 12.3%, Rhizoma Zingiberis Recens 10% is standby;
(b) get Radix Bupleuri, add water distillation and extraction 3 times, each 3 hours extraction times, get volatile oil and decocting liquid, with last medicinal residues extracting in water 2 times, each 1 hour, merge with the decocting liquid that begins to distill gained, suitably being concentrated into 50 ℃ of following relative densities is 1.20, put coldly, add ethanol precipitation and reach 70%, leave standstill to containing alcohol amount, get supernatant concentration recovery ethanol and get concentrated solution (1), standby; Follow the example of the Rhizoma Pinelliae and Rhizoma Zingiberis Recens, add 70% ethanol percolation, concentrated percolate gets concentrated solution (2), and is standby, get Radix Codonopsis, Fructus Jujubae and Radix Glycyrrhizae, extracting in water 3 times, each 1 hour, it was 1.18 that merge extractive liquid, is concentrated into 50 ℃ of following relative densities, add ethanol precipitation and reach 60% to containing the alcohol amount, leave standstill, get supernatant concentration recovery ethanol and get concentrated solution (3), standby; Get Radix Scutellariae, add 10 times of water gagings and extract 2 times, each 1 hour, it was 1.8,80 ℃ of insulations that extracting solution adds dilute hydrochloric acid adjusting pH value, leaves standstill, and filters, and precipitation is regulated pH value to 5.5 with the second alcohol and water, and vacuum drying gets Radix Scutellariae extract; (1), (2), (3) concentrated solution of gained are merged, continue to be condensed into thick paste (4);
(c) in appropriate amount of auxiliary materials, add above-mentioned thick paste (4), Radix Scutellariae extract, Radix Bupleuri volatile oil, fully mix, mixture stirs at 60~85 ℃ of heating and meltings, mixing time is 10~30 minutes, insulation is 1.1~3.5 millimeters at 60~85 ℃ of temperature following system, dropper bore, splashes in 0~18 ℃ the liquid paraffin, methyl-silicone oil, liquid coolant is use up and wiped to the drop pill drop that forms, back packing to be dried is made drop pill, promptly.
Best process for preparing medicine of the present invention comprises the following steps:
(a) take off by recipe quantity and state medical material: Radix Bupleuri 3.7%, Rhizoma Pinelliae Preparatum 18.5%, Radix Scutellariae 18.5%, Radix Codonopsis 18.5%, Fructus Jujubae 18.5%g, Radix Glycyrrhizae 12.3%, Rhizoma Zingiberis Recens 10% is standby;
(b) get Radix Bupleuri, add water distillation and extraction 3 times, each 3 hours extraction times, get volatile oil and decocting liquid, with last medicinal residues extracting in water 2 times, each 1 hour, merge with the decocting liquid that begins to distill gained, suitably being concentrated into 50 ℃ of following relative densities is 1.20, put coldly, add ethanol precipitation and reach 70%, leave standstill to containing alcohol amount, get supernatant concentration recovery ethanol and get concentrated solution (1), standby; Follow the example of the Rhizoma Pinelliae and Rhizoma Zingiberis Recens, add 70% ethanol percolation, concentrated percolate gets concentrated solution (2), and is standby; Get Radix Codonopsis, Fructus Jujubae and Radix Glycyrrhizae, extracting in water 3 times, each 1 hour, it was 1.18 that merge extractive liquid, is concentrated into 50 ℃ of following relative densities, added ethanol precipitation and reached 60% to containing the alcohol amount, left standstill, and got supernatant concentration recovery ethanol and got concentrated solution (3), standby; Get Radix Scutellariae, add 10 times of water gagings and extract 2 times, each 1 hour, it was 1.8,80 ℃ of insulations that extracting solution adds dilute hydrochloric acid adjusting pH value, leaves standstill, and filters, and precipitation is regulated pH value to 5.5 with the second alcohol and water, and vacuum drying gets Radix Scutellariae extract; (1), (2), (3) concentrated solution of gained are merged, continue to be condensed into thick paste (4);
Wherein the Radix Bupleuri distillation time in the step (b) is 1~5 hour, and preferable range is 2~4 hours, and Best Times is be extracted as 1~3 time of 3 hours medicinal residues, and optimum condition is 2 times, is 0.5~1.5 hour each time, and Best Times is 1 hour; Concentrated extracting solution to relative density is 50 ℃ following 1.10~1.15; Ethanol precipitation is 50~90% to containing the alcohol amount, and preferable range is 60~80%, and optimum condition is 70%; In the step (b), the concentration of the percolate of Rhizoma Pinelliae Preparatum and Rhizoma Zingiberis Recens is 50~90%.Preferable range is 60~80%, and optimum condition is 70%; In the step (b), Radix Codonopsis, Fructus Jujubae and liquorice beverage extract 2~4 times, and optimum condition is 3 times; Be 0.5~1.5 hour, optimum condition is 1 hour at every turn; Concentrated extracting solution to relative density is 50 ℃ following 1.15~1.20; Ethanol precipitation is 40~80% to containing the alcohol amount, and preferable range is 50~70%, and optimum condition is 60%; In the step (d), the Radix Scutellariae amount of water is 5~15 times, and preferable range is 8~12 times, and optimum condition is 10 times; It is 1.0~2.5 that hydrochloric acid is regulated pH value, and optimum condition is 1.5~2.0; Insulation is at 60~100 ℃, and optimum condition is 70~90 ℃, and optimum condition is 80 ℃; Precipitation is regulated pH value to 4~7 with the second alcohol and water, and optimum condition is 5~6; In the step (b), it is 70 ℃ following 1.25~1.30 that the concentrated solution after the merging continues to be concentrated into relative density;
More than form when producing and to increase or to reduce according to corresponding ratio, as large-scale production can be unit with kilogram or with the ton, small-scale production can be unit with the gram also, and weight can increase or reduce, but the crude drug material weight proportion constant rate between each composition.
More than each single medicinal material, especially adjuvant drug, messenger drug or adjuvant drug and messenger drug in forming, can be replaced by suitable Chinese medicine individually or simultaneously with the identical property of medicine, effect, it is constant to replace back Chinese medicine preparation and drug effect thereof.
Medicine of the present invention can be determined usage and dosage according to patient's situation in use, but every day 1-3 time, and every day, each crude drug consumption was as the criterion with the state-promulgated pharmacopoeia dosage, was no more than the pharmacopeia ormal weight.
The drop pill that the present invention is prepared, conventional drop pill advantage is simple as preparing except having, steady quality, can make liquid medicine solidification, convenient drug administration, efficient, quick-acting, its biggest advantage is:
1, the selected adjuvant pure natural of the present invention degree height: the substrate adjuvant that employed substrate adjuvant derives from natural plants or originates based on natural plants among the present invention, selected substrate adjuvant is xylitol and starch or lactose and starch or xylitol and arabic gum, this substrate adjuvant has pure natural degree height, toxic and side effects is low, mouthfeel is good, dissolve scattered time limit is short, rapid-action, it is a kind of new medium adjuvant, can be used for substituting present chemosynthesis adjuvant, the drop pill made from this kind adjuvant, it is low to solve the pure natural degree that present drop pill substrate faced, and more and more can not satisfy people and require back to nature, take low toxicity, the problem of the pure natural medical that has no side effect.
2, some problems in the outlet of solution Chinese medicine: medicine of the present invention also can solve Chinese medicine preparation, some problems of in exit procedure, being run into of dropping pill formulation particularly, solve because different countries, especially the European countries of industry prosperity are to the difference identification of the selected adjuvant of Chinese medicine dropping pill formulation, overcome as the selected adjuvant Polyethylene Glycol of the dropping pill formulation of the health food outlet defective in some national food additive catalogue not, improve the Chinese medicine dripping pills preparation and move towards the international market, strengthen the competitiveness of international market.
3, solve the relatively poor problem of dropping pill formulation taste and further improve drug effect speed (dissolve scattered time limit): the medicinal dropping ball made from this kind substrate adjuvant of the present invention, can improve Chinese medicine preparation, the particularly present not good shortcoming of dropping pill formulation taste, improve mouthfeel, more easy for patients to accept, and the drop pill that adopts the selected adjuvant of medicine of the present invention to make has shorter dissolve scattered time limit, make drug effect faster, be that alternate attack of chill and fever is treated in a kind of onset faster, feeling of fullness and disecomfort in the chest and hypochondrium, vexed happiness is told, the medicine of bitter taste in the mouth and dry throat.
4, higher safety and solve some problems in the drop pill storage process: the selected substrate of the present invention is not only additive, nutrient commonly used in the food industry, and can do medicinal, but do not see that it uses as the drug matrices adjuvant, therefore, with regard to substrate, be perfectly safe, have no side effect, a large amount of evidences, the drop pill made from this adjuvant can reduce effective ingredient separating out in storage process, the sticking ball of drop pill, easy shortcomings such as moisture absorption deliquescing, but the big production of suitability for industrialized.
In order to understand the present invention better, drip test explanation advantages of the present invention such as, drop pill soft durometer different, the sticking ball of drop pill below with the little Radix Bupleuri of new substrate with dissolve scattered time limit, the ball method of double differences of the small thorowax droplet made for the substrate adjuvant with the Polyethylene Glycol.
Test example 1: dissolve scattered time limit, the different contrast experiment's example of the ball method of double differences
In vitro tests
The present invention be that the small thorowax droplet that adjuvant is made compares with the Polyethylene Glycol, by measuring dissolve scattered time limit, investigate its good releasing effect; By measuring indexs such as the ball method of double differences is different, investigate whether it ripe in preparation technology, whether be fit to suitability for industrialized production.
1. test medication: the new substrate small thorowax droplet of the present invention (newly) is the small thorowax droplet (old) that adjuvant is made with the Polyethylene Glycol.
2. method and result:
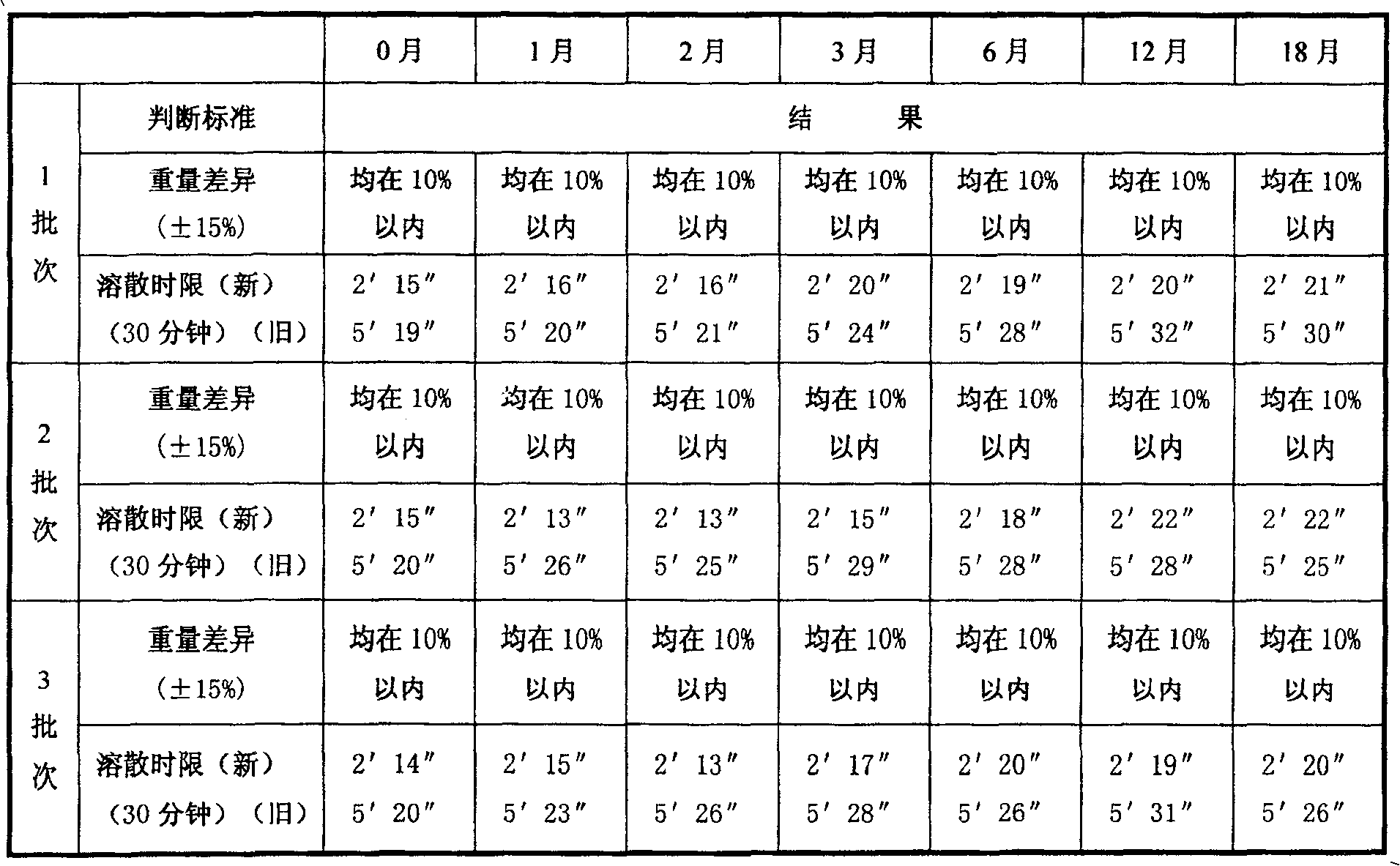
Dissolve scattered time limit: by " method is measured under this item of Chinese pharmacopoeia; The ball method of double differences is different: by " method is measured under this item of Chinese pharmacopoeia.Result of the test sees Table 1.
Three batches of small thorowax droplets made from the new medium adjuvant of table 1 (newly) with the polyethylene glycol 6000 be small thorowax droplet (old) dissolve scattered time limit made of adjuvant, weight differential relatively
Test data shows, the dissolve scattered time limit of new substrate small thorowax droplet is lacking of the small thorowax droplet made of adjuvant with the Polyethylene Glycol; The ball method of double differences of the small thorowax droplet that new and old substrate adjuvant is made is different all in the pharmacopeia prescribed limit.The result of the test explanation, the molten diffusing speed of the small thorowax droplet made from novel adjuvant is faster, is more conducive to medicine and plays a role in the shortest time, and the ball method of double differences is different all to be controlled in the pharmacopeia prescribed limit, the alternative present chemosynthesis adjuvant of this natural substrates adjuvant is described, but suitability for industrialized production.
Test example 2: the present invention with the polyethylene glycol 6000 be the sticking ball comparative observation of small thorowax droplet soft durometer, drop pill that adjuvant is made
The present invention be that the small thorowax droplet that adjuvant is made compares with the Polyethylene Glycol, by measuring indexs such as above-mentioned, investigate its effect.
1. test medication: the new substrate small thorowax droplet of the present invention (newly) is provided by the Jinshili Medicine Research ﹠. Development Co., Ltd., Tianjin; With the Polyethylene Glycol is the small thorowax droplet (old) that adjuvant is made, and is provided by the Jinshili Medicine Research ﹠. Development Co., Ltd., Tianjin.
2. method and result:
Get three batches of new, old substrate small thorowax droplets, be loaded in the porcelain vase respectively, and use the bottle stopper good seal.Putting it into the bottom has in the exsiccator of saturated Nacl (humidity 75%) solution, exsiccator is put into 40 ℃ of drying baker of constant temperature again, and timing sampling is observed situations such as drop pill soft durometer, the sticking ball of drop pill, the results are shown in Table 2.1, table 2.2.
Three batches in table 2.1 is that the small thorowax droplet reserved sample observing that adjuvant is made compares with the polyethylene glycol 6000
Table 2.2: three batches of small thorowax droplets made from the new medium adjuvant (newly) with the polyethylene glycol 6000 be small thorowax droplet (old) character observation made of adjuvant relatively
Test data shows, new substrate small thorowax droplet soft durometer changes and be that the small thorowax droplet made of adjuvant is similar, strong slightly with the Polyethylene Glycol; The sticking ball variation of the drop pill of new substrate small thorowax droplet, firmness change and be that the small thorowax droplet made of adjuvant is similar with the Polyethylene Glycol.The result of the test explanation, the sticking ball of the small thorowax droplet that new and old substrate adjuvant is made changes, firmness change is similar, and the alternative present chemosynthesis adjuvant of this natural substrates adjuvant is described, but suitability for industrialized production.
In vivo test:
This test illustrates beneficial effect of the present invention by the effect of interior resisting virus.
Healthy Kunming kind white mice, male and female half and half, female unpregnancy, body weight 14-16g are selected in experiment for use.By after the body weight layering, be divided into normal control group (not gastric infusion), model control group (irritate stomach and give distilled water), virazole matched group (virazole 0.07g/kg is given in intramuscular injection) and Herba Sidae Rhombifoliae soup drop pill matched group (irritate stomach and give Herba Sidae Rhombifoliae soup ball 1.2g/kg) at random earlier.Every group of 16 animals.Each group is by different groups and dosed administration (2 times/day) two days later, and except that the normal control group, all the other white mice are after the ether light anaesthesia, with 10LD
50Influenza virus (FM
1) infection of drop nose.After allowing it revive naturally, continue administration, continuous 4 days (totally 6 days).Observe the situations such as activity, feed, hair color, defecation of animal every day, and record dead animal number (dissect the dead animal back of should weighing immediately, takes out lung and claim weight in wet base, calculates the lung index one by one, and obtain lung index suppression ratio).To fasting after the administration in the 4th day of virus back 8 hours, each animal was weighed, and dissection is got the Mus lung and claimed weight in wet base, calculates the lung index one by one, and obtains lung index suppression ratio.
Lung index=lung weight in wet base (g) ÷ body weight (g) * 100
Lung index suppression ratio=(virus control group lung index average-test group lung index average) ÷ virus control group lung index average * 100%
The lung exponential quantity is big more, and the expression pulmonary lesion is serious more.The result carries out the statistical procedures rank rear in following table.
Table Herba Sidae Rhombifoliae soup drop pill infects the influence of white mice pulmonary lesion to influenza virus (FM1 strain)
Compare with model control group: * * P<0.01.
As can be seen from the above table, to the little viral pneumonia that causes from Mus of influenza infection, Herba Sidae Rhombifoliae soup drop pill group can lower its lung index, increases the exponential suppression ratio of lung, compares with the virus model group, and P<0.01 has the difference of highly significant.
The specific embodiment
The present invention is further illustrated below in conjunction with specific embodiment, and following this embodiment only is used to the present invention is described and to the present invention without limits.
Embodiment 1
(a) get Radix Bupleuri 33.3g, Rhizoma Pinelliae Preparatum 166.7g, Radix Scutellariae 166.7g, Radix Codonopsis 166.7g, Fructus Jujubae 166.7g, Radix Glycyrrhizae 111.1g, Rhizoma Zingiberis Recens 88.9g, xylitol 65.5g, arabic gum 18.5g are standby;
(b) get Radix Bupleuri, add water distillation and extraction 3 times, each 3 hours extraction times, get volatile oil and decocting liquid, with last medicinal residues extracting in water 2 times, each 1 hour, merge with the decocting liquid that begins to distill gained, suitably being concentrated into 50 ℃ of following relative densities is 1.20, put coldly, add ethanol precipitation and reach 70%, leave standstill to containing alcohol amount, get supernatant concentration recovery ethanol and get concentrated solution (1), standby; Follow the example of the Rhizoma Pinelliae and Rhizoma Zingiberis Recens, add 70% ethanol percolation, concentrated percolate gets concentrated solution (2), and is standby, get Radix Codonopsis, Fructus Jujubae and Radix Glycyrrhizae, extracting in water 3 times, each 1 hour, it was 1.18 that merge extractive liquid, is concentrated into 50 ℃ of following relative densities, add ethanol precipitation and reach 60% to containing the alcohol amount, leave standstill, get supernatant concentration recovery ethanol and get concentrated solution (3), standby; Get Radix Scutellariae, add 10 times of water gagings and extract 2 times, each 1 hour, it was 1.8,80 ℃ of insulations that extracting solution adds dilute hydrochloric acid adjusting pH value, leaves standstill, and filters, and precipitation is regulated pH value to 5.5 with the second alcohol and water, and vacuum drying gets Radix Scutellariae extract; (1), (2), (3) concentrated solution of gained are merged, continue to be condensed into thick paste (4);
(c) in xylitol and starch mixture, add above-mentioned thick paste (4), Radix Scutellariae extract, Radix Bupleuri volatile oil fully mixes, mixture is at 105~115 ℃ of heating and meltings, stir, mixing time is 10 minutes, insulation, at 62~66 ℃ of temperature following system, dropper bore is 1.20~2.0 millimeters, splash in 0~8 ℃ the methyl-silicone oil, make drop pill, promptly.
Embodiment 2
(a) get Radix Bupleuri 33.3g, Rhizoma Pinelliae Preparatum 166.7g, Radix Scutellariae 166.7g, Radix Codonopsis 166.7g, Fructus Jujubae 166.7g, Radix Glycyrrhizae 111.1g, Rhizoma Zingiberis Recens 88.9g, xylitol, arabic gum are standby;
(b) get Radix Bupleuri, add water distillation and extraction 1 time, 3 hours extraction times, get volatile oil and decocting liquid, with last medicinal residues extracting in water a time, the time is 1 hour, merges with the decocting liquid that begins to distill gained, suitably being concentrated into 50 ℃ of following relative densities is 1.10, put coldly, add ethanol precipitation and reach 50%, leave standstill to containing alcohol amount, get supernatant concentration recovery ethanol and get concentrated solution, standby; Follow the example of the Rhizoma Pinelliae and Rhizoma Zingiberis Recens, add 50% ethanol percolation, concentrated percolate gets concentrated solution, and is standby; Get Radix Codonopsis, Fructus Jujubae and Radix Glycyrrhizae, extracting in water 3 times, each 1 hour, it was 1.18 that merge extractive liquid, is concentrated into 50 ℃ of following relative densities, added ethanol precipitation and reached 60% to containing the alcohol amount, left standstill, and got supernatant concentration recovery ethanol and got concentrated solution, standby; Get Radix Scutellariae, add 10 times of water gagings and extract 2 times, each 1 hour, it was 1.0,60 ℃ of insulations that extracting solution adds dilute hydrochloric acid adjusting pH value, leaves standstill, and filters, and precipitation is regulated pH value to 4.0 with the second alcohol and water, and vacuum drying gets Radix Scutellariae extract; Concentrated solution merges, and continues to be condensed into thick paste, and 70 ℃ of following relative densities are 1.25 standby;
(c) with above-mentioned Radix Scutellariae extract, Radix Bupleuri volatile oil and thick paste and be 5 times of amounts of extract, xylitol: arabic gum is 5:1, the mixture mix homogeneously, mixture stirs at 60~85 ℃ of heating and meltings, mixing time is 10~30 minutes, insulation is 1.1~3.5 millimeters at 60~85 ℃ of temperature following system, dropper bore, splashes in 0~18 ℃ the methyl-silicone oil, liquid coolant is use up and wiped to the drop pill drop that forms, back packing to be dried is made drop pill, promptly.
Embodiment 3
(a) get Radix Bupleuri 33.3g, Rhizoma Pinelliae Preparatum 166.7g, Radix Scutellariae 166.7g, Radix Codonopsis 166.7g, Fructus Jujubae 166.7g, Radix Glycyrrhizae 111.1g, Rhizoma Zingiberis Recens 88.9g, lactose, starch are standby;
(b) get Radix Bupleuri, add water distillation and extraction 2 times, each 3 hours extraction times, get volatile oil and decocting liquid, with last medicinal residues extracting in water 2 times, each 1.5 hours, merge with the decocting liquid that begins to distill gained, suitably being concentrated into 50 ℃ of following relative densities is 1.30, put coldly, add ethanol precipitation and reach 50%, leave standstill to containing alcohol amount, get supernatant concentration recovery ethanol and get concentrated solution (1), standby; Follow the example of the Rhizoma Pinelliae and Rhizoma Zingiberis Recens, add 60% ethanol percolation, concentrated percolate gets concentrated solution (2), and is standby, get Radix Codonopsis, Fructus Jujubae and Radix Glycyrrhizae, extracting in water 2 times, each 0.5 hour, it was 1.18 that merge extractive liquid, is concentrated into 50 ℃ of following relative densities, add ethanol precipitation and reach 50% to containing the alcohol amount, leave standstill, get supernatant concentration recovery ethanol and get concentrated solution (3), standby; Get Radix Scutellariae, add 8 times of water gagings and extract 2 times, each 1 hour, it was 1.5,70 ℃ of insulations that extracting solution adds dilute hydrochloric acid adjusting pH value, leaves standstill, and filters, and precipitation is regulated pH value to 5.0 with the second alcohol and water, and vacuum drying gets Radix Scutellariae extract; (1), (2), (3) concentrated solution of gained are merged, continue to be condensed into thick paste (4);
(c) in appropriate amount of auxiliary materials, add above-mentioned thick paste (4), Radix Scutellariae extract, Radix Bupleuri volatile oil, fully mix, mixture stirs at 90~95 ℃ of heating and meltings, mixing time is 10 minutes, insulation is 1.21~2.5 millimeters at 62~65 ℃ of temperature following system, dropper bore, splashes in 0~8 ℃ the liquid paraffin, liquid coolant is use up and wiped to the drop pill drop that forms, back packing to be dried is made drop pill, promptly.
Embodiment 4
(a) get Radix Bupleuri 33.3g, Rhizoma Pinelliae Preparatum 166.7g, Radix Scutellariae 166.7g, Radix Codonopsis 166.7g, Fructus Jujubae 166.7g, Radix Glycyrrhizae 111.1g, Rhizoma Zingiberis Recens 88.9g, sorbitol 56.8g, xanthan gum 26.2g are standby;
(b) get Radix Bupleuri, add water distillation and extraction 4 times, each 3 hours extraction times, get volatile oil and decocting liquid, with last medicinal residues extracting in water 3 times, each 1 hour, merge with the decocting liquid that begins to distill gained, suitably being concentrated into 50 ℃ of following relative densities is 1.40, put coldly, add ethanol precipitation and reach 80%, leave standstill to containing alcohol amount, get supernatant concentration recovery ethanol and get concentrated solution (1), standby; Follow the example of the Rhizoma Pinelliae and Rhizoma Zingiberis Recens, add 80% ethanol percolation, concentrated percolate gets concentrated solution (2), and is standby; Get Radix Codonopsis, Fructus Jujubae and Radix Glycyrrhizae, extracting in water 4 times, each 1.5 hours, it was 1.19 that merge extractive liquid, is concentrated into 50 ℃ of following relative densities, added ethanol precipitation and reached 70% to containing the alcohol amount, left standstill, and got supernatant concentration recovery ethanol and got concentrated solution (3), standby; Get Radix Scutellariae, add 12 times of water gagings and extract 2 times, each 1 hour, it was 2.0,90 ℃ of insulations that extracting solution adds dilute hydrochloric acid adjusting pH value, leaves standstill, and filters, and precipitation is regulated pH value to 5.0 with the second alcohol and water, and vacuum drying gets Radix Scutellariae extract; (1), (2), (3) concentrated solution of gained are merged, continue to be condensed into thick paste (4);
(c) in sorbitol and xanthan gum mixtures, add above-mentioned thick paste (4), Radix Scutellariae extract, Radix Bupleuri volatile oil, fully mix, mixture stirs at 64 ℃ of heating and meltings, mixing time is 10~30 minutes, insulation is 1.2~2.5 millimeters at 64 ℃ of temperature following system, dropper bore, splashes in 0 ℃ the methyl-silicone oil, liquid coolant is use up and wiped to the drop pill drop that forms, back packing to be dried is made drop pill, promptly.
Embodiment 5
(a) get Radix Bupleuri 33.3g, Rhizoma Pinelliae Preparatum 166.7g, Radix Scutellariae 166.7g, Radix Codonopsis 166.7g, Fructus Jujubae 166.7g, Radix Glycyrrhizae 111.1g, Rhizoma Zingiberis Recens 88.9g, trehalose 85.6g, cyclodextrin 21.4g are standby;
(b) get Radix Bupleuri, add water distillation and extraction 5 times, each 3 hours extraction times, get volatile oil and decocting liquid, with last medicinal residues extracting in water 2 times, each 1 hour, merge with the decocting liquid that begins to distill gained, suitably being concentrated into 50 ℃ of following relative densities is 1.20, put coldly, add ethanol precipitation and reach 70%, leave standstill to containing alcohol amount, get supernatant concentration recovery ethanol and get concentrated solution (1), standby; Follow the example of the Rhizoma Pinelliae and Rhizoma Zingiberis Recens, add 70% ethanol percolation, concentrated percolate gets concentrated solution (2), and is standby; Get Radix Codonopsis, Fructus Jujubae and Radix Glycyrrhizae, extracting in water 3 times, each 1 hour, it was 1.18 that merge extractive liquid, is concentrated into 50 ℃ of following relative densities, added ethanol precipitation and reached 60% to containing the alcohol amount, left standstill, and got supernatant concentration recovery ethanol and got concentrated solution (3), standby; Get Radix Scutellariae, add 10 times of water gagings and extract 2 times, each 1 hour, it was 1.8,80 ℃ of insulations that extracting solution adds dilute hydrochloric acid adjusting pH value, leaves standstill, and filters, and precipitation is regulated pH value to 5.5 with the second alcohol and water, and vacuum drying gets Radix Scutellariae extract; (1), (2), (3) concentrated solution of gained are merged, continue to be condensed into thick paste (4);
(c) in trehalose and cyclodextrin mixt, add above-mentioned thick paste (4), Radix Scutellariae extract, Radix Bupleuri volatile oil, fully mix, mixture stirs at 85~93 ℃ of heating and meltings, mixing time is 15 minutes, insulation is 1.6~3.5 millimeters at 60~65 ℃ of temperature following system, dropper bore, splashes in 0~18 ℃ the methyl-silicone oil, liquid coolant is use up and wiped to the drop pill drop that forms, back packing to be dried is made drop pill, promptly.
Embodiment 6
(a) get Radix Bupleuri 33.3g, Rhizoma Pinelliae Preparatum 166.7g, Radix Scutellariae 166.7g, Radix Codonopsis 166.7g, Fructus Jujubae 166.7g, Radix Glycyrrhizae 111.1g, Rhizoma Zingiberis Recens 88.9g, lactose 101.5g, hydroxypropyl starch 15.6g are standby; Get Radix Bupleuri, add water distillation and extraction 2 times, each 3 hours extraction times, get volatile oil and decocting liquid, last medicinal residues are added water
(b) extract 2 times, each 1 hour, merge with the decocting liquid that begins to distill gained, suitably being concentrated into 50 ℃ of following relative densities is 1.20, puts coldly, adds ethanol precipitation and reaches 90% to containing the alcohol amount, leaves standstill, and gets supernatant concentration and reclaims ethanol and get concentrated solution (1), standby; Follow the example of the Rhizoma Pinelliae and Rhizoma Zingiberis Recens, add 90% ethanol percolation, concentrated percolate gets concentrated solution (2), and is standby; Get Radix Codonopsis, Fructus Jujubae and Radix Glycyrrhizae, extracting in water 3 times, each 1 hour, it was 1.18 that merge extractive liquid, is concentrated into 50 ℃ of following relative densities, added ethanol precipitation and reached 60% to containing the alcohol amount, left standstill, and got supernatant concentration recovery ethanol and got concentrated solution (3), standby; Get Radix Scutellariae, add 10 times of water gagings and extract 2 times, each 1 hour, it was 1.8,80 ℃ of insulations that extracting solution adds dilute hydrochloric acid adjusting pH value, leaves standstill, and filters, and precipitation is regulated pH value to 5.5 with the second alcohol and water, and vacuum drying gets Radix Scutellariae extract; (1), (2), (3) concentrated solution of gained are merged, continue to be condensed into thick paste (4);
(c) in lactose and hydroxypropyl starch mixture, add above-mentioned thick paste (4), Radix Scutellariae extract, Radix Bupleuri volatile oil, fully mix, mixture stirs at 70~75 ℃ of heating and meltings, mixing time is 60 minutes, insulation is 1.8~2.5 millimeters at 50~55 ℃ of temperature following system, dropper bore, splashes in 0~10 ℃ the liquid paraffin, liquid coolant is use up and wiped to the drop pill drop that forms, back packing to be dried is made drop pill, promptly.
Embodiment 7
(a) get Radix Bupleuri 33.3g, Rhizoma Pinelliae Preparatum 166.7g, Radix Scutellariae 166.7g, Radix Codonopsis 166.7g, Fructus Jujubae 166.7g, Radix Glycyrrhizae 111.1g, Rhizoma Zingiberis Recens 88.9g, xylitol, methylcellulose are standby;
(b) get Radix Bupleuri, add water distillation and extraction 2 times, each 3 hours extraction times, get volatile oil and decocting liquid, with last medicinal residues extracting in water 2 times, each 1 hour, merge with the decocting liquid that begins to distill gained, suitably being concentrated into 50 ℃ of following relative densities is 1.40, put coldly, add ethanol precipitation and reach 80%, leave standstill to containing alcohol amount, get supernatant concentration recovery ethanol and get concentrated solution (1), standby; Follow the example of the Rhizoma Pinelliae and Rhizoma Zingiberis Recens, add 80% ethanol percolation, concentrated percolate gets concentrated solution (2), and is standby, get Radix Codonopsis, Fructus Jujubae and Radix Glycyrrhizae, extracting in water 3 times, each 1 hour, it was 1.18 that merge extractive liquid, is concentrated into 50 ℃ of following relative densities, add ethanol precipitation and reach 60% to containing the alcohol amount, leave standstill, get supernatant concentration recovery ethanol and get concentrated solution (3), standby; Get Radix Scutellariae, add 10 times of water gagings and extract 2 times, each 1 hour, it was 1.8,80 ℃ of insulations that extracting solution adds dilute hydrochloric acid adjusting pH value, leaves standstill, and filters, and precipitation is regulated pH value to 5.5 with the second alcohol and water, and vacuum drying gets Radix Scutellariae extract; (1), (2), (3) concentrated solution of gained are merged, continue to be condensed into thick paste (4), 70 ℃ of following relative densities are 1.27;
(c) to above-mentioned thick paste (4), Radix Scutellariae extract, add in the Radix Bupleuri volatile oil 6 times of amounts, xylitol: methylcellulose is 1: 0.3 a adjuvant, fully mix, mixture stirs at 72~75 ℃ of heating and meltings, mixing time is 40 minutes, insulation is 1.2~2.0 millimeters at 60~86 ℃ of temperature following system, dropper bore, in droplets 3 ℃ the methyl-silicone oil, liquid coolant is use up and wiped to the drop pill drop that forms, back packing to be dried is made drop pill, promptly.
Embodiment 8
(a) get Radix Bupleuri 33.3g, Rhizoma Pinelliae Preparatum 166.7g, Radix Scutellariae 166.7g, Radix Codonopsis 166.7g, Fructus Jujubae 166.7g, Radix Glycyrrhizae 111.1g, Rhizoma Zingiberis Recens 88.9g, xylitol, starch are standby;
(b) get Radix Bupleuri, add water distillation and extraction 3 times, each 3 hours extraction times, get volatile oil and decocting liquid, with last medicinal residues extracting in water 2 times, each 1 hour, merge with the decocting liquid that begins to distill gained, suitably being concentrated into 50 ℃ of following relative densities is 1.40, put coldly, add ethanol precipitation and reach 60%, leave standstill to containing alcohol amount, get supernatant concentration recovery ethanol and get concentrated solution (1), standby; Follow the example of the Rhizoma Pinelliae and Rhizoma Zingiberis Recens, add 80% ethanol percolation, concentrated percolate gets concentrated solution (2), and is standby, get Radix Codonopsis, Fructus Jujubae and Radix Glycyrrhizae, extracting in water 3 times, each 1 hour, it was 1.18 that merge extractive liquid, is concentrated into 50 ℃ of following relative densities, add ethanol precipitation and reach 700% to containing the alcohol amount, leave standstill, get supernatant concentration recovery ethanol and get concentrated solution (3), standby; Get Radix Scutellariae, add 15 times of water gagings and extract 2 times, each 1 hour, it was 1.8,80 ℃ of insulations that extracting solution adds dilute hydrochloric acid adjusting pH value, leaves standstill, and filters, and precipitation is regulated pH value to 7 with the second alcohol and water, and vacuum drying gets Radix Scutellariae extract; (1), (2), (3) concentrated solution of gained are merged, continue to be condensed into thick paste (4), 70 ℃ of following relative densities are 1.30;
(c) to above-mentioned thick paste (4), Radix Scutellariae extract, add in the Radix Bupleuri volatile oil 5 times of amounts, xylitol: starch is 1: 0.25 mixture, fully mix, mixture stirs at 60~85 ℃ of heating and meltings, mixing time is 10~30 minutes, insulation is 1.1~3.5 millimeters at 60~85 ℃ of temperature following system, dropper bore, splashes in 0 ℃ the methyl-silicone oil, liquid coolant is use up and wiped to the drop pill drop that forms, back packing to be dried is made drop pill, promptly.
Embodiment 9
(a) get Radix Bupleuri 33.3g, Rhizoma Pinelliae Preparatum 166.7g, Radix Scutellariae 166.7g, Radix Codonopsis 166.7g, Fructus Jujubae 166.7g, Radix Glycyrrhizae 111.1g, Rhizoma Zingiberis Recens 88.9g, lactose, starch are standby;
(b) get Radix Bupleuri, add water distillation and extraction 2 times, each 3 hours extraction times, get volatile oil and decocting liquid, with last medicinal residues extracting in water 3 times, each 1.5 hours, merge with the decocting liquid that begins to distill gained, suitably being concentrated into 50 ℃ of following relative densities is 1.50, put coldly, add ethanol precipitation and reach 50%, leave standstill to containing alcohol amount, get supernatant concentration recovery ethanol and get concentrated solution (1), standby; Follow the example of the Rhizoma Pinelliae and Rhizoma Zingiberis Recens, add 90% ethanol percolation, concentrated percolate gets concentrated solution (2), and is standby, get Radix Codonopsis, Fructus Jujubae and Radix Glycyrrhizae, extracting in water 4 times, each 1.5 hours, it was 1.18 that merge extractive liquid, is concentrated into 50 ℃ of following relative densities, add ethanol precipitation and reach 60% to containing the alcohol amount, leave standstill, get supernatant concentration recovery ethanol and get concentrated solution (3), standby; Get Radix Scutellariae, add 10 times of water gagings and extract 2 times, each 1 hour, it was 2.5,80 ℃ of insulations that extracting solution adds dilute hydrochloric acid adjusting pH value, leaves standstill, and filters, and precipitation is regulated pH value to 7 with the second alcohol and water, and vacuum drying gets Radix Scutellariae extract; (1), (2), (3) concentrated solution of gained are merged, continue to be condensed into thick paste (4), 70 ℃ of following relative densities are 1.27;
(c) to above-mentioned thick paste (4), Radix Scutellariae extract, add in the Radix Bupleuri volatile oil 5 times of amounts, lactose: starch is 1: 0.3 adjuvant, fully mix, mixture stirs at 85 ℃ of heating and meltings, mixing time is 10~30 minutes, insulation is 1.7 millimeters at 65 ℃ of temperature following system, dropper bore, splashes in-10~5 ℃ the methyl-silicone oil, liquid coolant is use up and wiped to the drop pill drop that forms, back packing to be dried is made drop pill, promptly.
Embodiment 10
(a) get Radix Bupleuri 33.3g, Rhizoma Pinelliae Preparatum 166.7g, Radix Scutellariae 166.7g, Radix Codonopsis 166.7g, Fructus Jujubae 166.7g, Radix Glycyrrhizae 111.1g, Rhizoma Zingiberis Recens 88.9g, xylitol, arabic gum are standby;
(b) get Radix Bupleuri, add water distillation and extraction 4 times, each 3 hours extraction times, get volatile oil and decocting liquid, with last medicinal residues extracting in water 2 times, each 1 hour, merge with the decocting liquid that begins to distill gained, suitably being concentrated into 50 ℃ of following relative densities is 1.20, put coldly, add ethanol precipitation and reach 70%, leave standstill to containing alcohol amount, get supernatant concentration recovery ethanol and get concentrated solution (1), standby; Follow the example of the Rhizoma Pinelliae and Rhizoma Zingiberis Recens, add 70% ethanol percolation, concentrated percolate gets concentrated solution (2), and is standby, get Radix Codonopsis, Fructus Jujubae and Radix Glycyrrhizae, extracting in water 3 times, each 1 hour, it was 1.18 that merge extractive liquid, is concentrated into 50 ℃ of following relative densities, add ethanol precipitation and reach 60% to containing the alcohol amount, leave standstill, get supernatant concentration recovery ethanol and get concentrated solution (3), standby; Get Radix Scutellariae, add 10 times of water gagings and extract 2 times, each 1 hour, it was 1.8,80 ℃ of insulations that extracting solution adds dilute hydrochloric acid adjusting pH value, leaves standstill, and filters, and precipitation is regulated pH value to 5.5 with the second alcohol and water, and vacuum drying gets Radix Scutellariae extract; (1), (2), (3) concentrated solution of gained are merged, continue to be condensed into thick paste (4), 70 ℃ of following relative densities are 1.28;
(c) to above-mentioned thick paste (4), Radix Scutellariae extract, add in the Radix Bupleuri volatile oil 3 times of amounts, xylitol: arabic gum is 1: 0.4 a adjuvant, fully mix, mixture stirs at 75 ℃ of heating and meltings, mixing time is 50 minutes, insulation is 1.21 millimeters at 70 ℃ of temperature following system, dropper bore, splashes in 5 ℃ the methyl-silicone oil, liquid coolant is use up and wiped to the drop pill drop that forms, back packing to be dried is made drop pill, promptly.
Embodiment 11
(a) get Radix Bupleuri 27g, Rhizoma Pinelliae Preparatum 144g, Radix Scutellariae 180g, Radix Codonopsis 180g, Fructus Jujubae 180g, Radix Glycyrrhizae 90g, Rhizoma Zingiberis Recens 99g, xylitol, chitin are standby;
(b) get Radix Bupleuri, add water distillation and extraction 3 times, each 3 hours extraction times, get volatile oil and decocting liquid, with last medicinal residues extracting in water 2 times, each 1 hour, merge with the decocting liquid that begins to distill gained, suitably being concentrated into 50 ℃ of following relative densities is 1.20, put coldly, add ethanol precipitation and reach 70%, leave standstill to containing alcohol amount, get supernatant concentration recovery ethanol and get concentrated solution (1), standby; Follow the example of the Rhizoma Pinelliae and Rhizoma Zingiberis Recens, add 70% ethanol percolation, concentrated percolate gets concentrated solution (2), and is standby, get Radix Codonopsis, Fructus Jujubae and Radix Glycyrrhizae, extracting in water 3 times, each 1 hour, it was 1.18 that merge extractive liquid, is concentrated into 50 ℃ of following relative densities, add ethanol precipitation and reach 60% to containing the alcohol amount, leave standstill, get supernatant concentration recovery ethanol and get concentrated solution (3), standby; Get Radix Scutellariae, add 10 times of water gagings and extract 2 times, each 1 hour, it was 1.8,80 ℃ of insulations that extracting solution adds dilute hydrochloric acid adjusting pH value, leaves standstill, and filters, and precipitation is regulated pH value to 5.5 with the second alcohol and water, and vacuum drying gets Radix Scutellariae extract; (1), (2), (3) concentrated solution of gained are merged, continue to be condensed into thick paste (4), 70 ℃ of following relative densities are 1.28;
(c) to above-mentioned thick paste (4), Radix Scutellariae extract, add in the Radix Bupleuri volatile oil 5.5 times of amounts, xylitol: chitin is 1: 0.6 a adjuvant, fully mix, mixture stirs at 100 ℃ of heating and meltings, mixing time is 5 minutes, insulation is 1.1 millimeters at 85 ℃ of temperature following system, dropper bore, splashes in 0 ℃ the methyl-silicone oil, liquid coolant is use up and wiped to the drop pill drop that forms, back packing to be dried is made drop pill, promptly.
Embodiment 12
(a) get Radix Bupleuri 45g, Rhizoma Pinelliae Preparatum 153g, Radix Scutellariae 171g, Radix Codonopsis 171g, Fructus Jujubae 171g, Radix Glycyrrhizae 99g, Rhizoma Zingiberis Recens 90g, lactose, Furcellaran are standby;
(b) get Radix Bupleuri, add water distillation and extraction 1 time, 3 hours extraction times, get volatile oil and decocting liquid, with last medicinal residues extracting in water a time, the time is 1 hour, merges with the decocting liquid that begins to distill gained, suitably being concentrated into 50 ℃ of following relative densities is 1.10, put coldly, add ethanol precipitation and reach 50%, leave standstill to containing alcohol amount, get supernatant concentration recovery ethanol and get concentrated solution (1), standby; Follow the example of the Rhizoma Pinelliae and Rhizoma Zingiberis Recens, add 50% ethanol percolation, concentrated percolate gets concentrated solution (2), and is standby, get Radix Codonopsis, Fructus Jujubae and Radix Glycyrrhizae, extracting in water 3 times, each 1 hour, it was 1.18 that merge extractive liquid, is concentrated into 50 ℃ of following relative densities, add ethanol precipitation and reach 60% to containing the alcohol amount, leave standstill, get supernatant concentration recovery ethanol and get concentrated solution (3), standby; Get Radix Scutellariae, add 10 times of water gagings and extract 2 times, each 1 hour, it was 1.0,60 ℃ of insulations that extracting solution adds dilute hydrochloric acid adjusting pH value, leaves standstill, and filters, and precipitation is regulated pH value to 4.0 with the second alcohol and water, and vacuum drying gets Radix Scutellariae extract; (1), (2), (3) concentrated solution of gained are merged, continue to be condensed into thick paste (4), 70 ℃ of following relative densities are 1.25;
(c) to above-mentioned thick paste (4), Radix Scutellariae extract, add in the Radix Bupleuri volatile oil 2.5 times of amounts, lactose: Furcellaran is in 1: 1 the adjuvant, fully mix, mixture stirs at 65 ℃ of heating and meltings, mixing time is 60 minutes, insulation is 1.21~3.5 millimeters at 60 ℃ of temperature following system, dropper bore, in droplets 8 ℃ the liquid paraffin, liquid coolant is use up and wiped to the drop pill drop that forms, back packing to be dried is made drop pill, promptly.
Embodiment 13
(a) get Radix Bupleuri 36g, Rhizoma Pinelliae Preparatum 162g, Radix Scutellariae 162g, Radix Codonopsis 180g, Fructus Jujubae 171g, Radix Glycyrrhizae 117g, Rhizoma Zingiberis Recens 72g, lactose, Ficus elastica are standby;
(b) get Radix Bupleuri, add water distillation and extraction 4 times, each 3 hours extraction times, get volatile oil and decocting liquid, with last medicinal residues extracting in water 2 times, each 1 hour, merge with the decocting liquid that begins to distill gained, suitably being concentrated into 50 ℃ of following relative densities is 1.20, put coldly, add ethanol precipitation and reach 70%, leave standstill to containing alcohol amount, get supernatant concentration recovery ethanol and get concentrated solution (1), standby; Follow the example of the Rhizoma Pinelliae and Rhizoma Zingiberis Recens, add 70% ethanol percolation, concentrated percolate gets concentrated solution (2), and is standby, get Radix Codonopsis, Fructus Jujubae and Radix Glycyrrhizae, extracting in water 3 times, each 1 hour, it was 1.18 that merge extractive liquid, is concentrated into 50 ℃ of following relative densities, add ethanol precipitation and reach 60% to containing the alcohol amount, leave standstill, get supernatant concentration recovery ethanol and get concentrated solution (3), standby; Get Radix Scutellariae, add 10 times of water gagings and extract 2 times, each 1 hour, it was 1.8,80 ℃ of insulations that extracting solution adds dilute hydrochloric acid adjusting pH value, leaves standstill, and filters, and precipitation is regulated pH value to 5.5 with the second alcohol and water, and vacuum drying gets Radix Scutellariae extract; (1), (2), (3) concentrated solution of gained are merged, continue to be condensed into thick paste (4), 70 ℃ of following relative densities are 1.28;
(c) to above-mentioned thick paste (4), Radix Scutellariae extract, add in the Radix Bupleuri volatile oil 4.5 times of amounts, lactose: the ratio of Ficus elastica weight is to add in 1: 0.5 the adjuvant, fully mix, mixture stirs at 75 ℃ of heating and meltings, mixing time is 125 minutes, insulation is 1.9 millimeters at 66 ℃ of temperature following system, dropper bore, splashes in 10 ℃ the vegetable oil, liquid coolant is use up and wiped to the drop pill drop that forms, back packing to be dried is made drop pill, promptly.
Embodiment 14
(a) get Radix Bupleuri 33.3g, Rhizoma Pinelliae Preparatum 166.7g, Radix Scutellariae 166.7g, Radix Codonopsis 166.7g, Fructus Jujubae 166.7g, Radix Glycyrrhizae 111.1g, Rhizoma Zingiberis Recens 88.9g, xylitol, tragakanta are standby;
(b) get Radix Bupleuri, add water distillation and extraction 3 times, each 3 hours extraction times, get volatile oil and decocting liquid, with last medicinal residues extracting in water 2 times, each 0.5 hour, merge with the decocting liquid that begins to distill gained, suitably being concentrated into 50 ℃ of following relative densities is 1.20, put coldly, add ethanol precipitation and reach 70%, leave standstill to containing alcohol amount, get supernatant concentration recovery ethanol and get concentrated solution (1), standby; Follow the example of the Rhizoma Pinelliae and Rhizoma Zingiberis Recens, add 70% ethanol percolation, concentrated percolate gets concentrated solution (2), and is standby, get Radix Codonopsis, Fructus Jujubae and Radix Glycyrrhizae, extracting in water 3 times, each 1 hour, it was 1.19 that merge extractive liquid, is concentrated into 50 ℃ of following relative densities, add ethanol precipitation and reach 70% to containing the alcohol amount, leave standstill, get supernatant concentration recovery ethanol and get concentrated solution (3), standby; Get Radix Scutellariae, add 10 times of water gagings and extract 2 times, each 1 hour, it was 2.0,90 ℃ of insulations that extracting solution adds dilute hydrochloric acid adjusting pH value, leaves standstill, and filters, and precipitation is regulated pH value to 5.5 with the second alcohol and water, and vacuum drying gets Radix Scutellariae extract;
(c) to above-mentioned thick paste (4), Radix Scutellariae extract, add in the Radix Bupleuri volatile oil 7 times of amounts, xylitol: the ratio of the weight of tragakanta is 1: 0.1 a adjuvant, fully mix, mixture stirs at 75 ℃ of heating and meltings, mixing time is 100 minutes, insulation is 3.5 millimeters at 45 ℃ of temperature following system, dropper bore, splashes in-10~5 ℃ the methyl-silicone oil, liquid coolant is use up and wiped to the drop pill drop that forms, back packing to be dried is made drop pill, promptly.
Embodiment 15
(a) get Radix Bupleuri 33.3g, Rhizoma Pinelliae Preparatum 166.7g, Radix Scutellariae 166.7g, Radix Codonopsis 166.7g, Fructus Jujubae 166.7g, Radix Glycyrrhizae 111.1g, Rhizoma Zingiberis Recens 88.9g, xylitol, starch are standby;
(b) get Radix Bupleuri, add water distillation and extraction 3 times, each 3 hours extraction times, get volatile oil and decocting liquid, with last medicinal residues extracting in water 2 times, each 0.5 hour, merge with the decocting liquid that begins to distill gained, suitably being concentrated into 50 ℃ of following relative densities is 1.20, put coldly, add ethanol precipitation and reach 70%, leave standstill to containing alcohol amount, get supernatant concentration recovery ethanol and get concentrated solution (1), standby; Follow the example of the Rhizoma Pinelliae and Rhizoma Zingiberis Recens, add 70% ethanol percolation, concentrated percolate gets concentrated solution (2), and is standby; Get Radix Codonopsis, Fructus Jujubae and Radix Glycyrrhizae, extracting in water 3 times, each 1 hour, it was 1.19 that merge extractive liquid, is concentrated into 50 ℃ of following relative densities, added ethanol precipitation and reached 70% to containing the alcohol amount, left standstill, and got supernatant concentration recovery ethanol and got concentrated solution (3), standby; Get Radix Scutellariae, add 10 times of water gagings and extract 2 times, each 1 hour, it was 2.0,90 ℃ of insulations that extracting solution adds dilute hydrochloric acid adjusting pH value, leaves standstill, and filters, and precipitation is regulated pH value to 5.5 with the second alcohol and water, and vacuum drying gets Radix Scutellariae extract; (1), (2), (3) concentrated solution of gained are merged, continue to be condensed into thick paste (4), 70 ℃ of following relative densities are 1.25;
(c) to above-mentioned thick paste (4), Radix Scutellariae extract, add in the Radix Bupleuri volatile oil 5.5 times of amounts, xylitol: 1: 0.25 the adjuvant of ratio of the weight of starch, fully mix, mixture stirs at 85 ℃ of heating and meltings, mixing time is 10~30 minutes, insulation is 1.1~3.5 millimeters at 85 ℃ of temperature following system, dropper bore, splashes into 0~5 ℃ liquid paraffin, liquid coolant is use up and wiped to the drop pill drop that forms, back packing to be dried is made drop pill, promptly.
Embodiment 16
(a) get Radix Bupleuri 33.3g, Rhizoma Pinelliae Preparatum 166.7g, Radix Scutellariae 166.7g, Radix Codonopsis 166.7g, Fructus Jujubae 166.7g, Radix Glycyrrhizae 111.1g, Rhizoma Zingiberis Recens 88.9g, lactose, starch are standby;
(b) get Radix Bupleuri, add water distillation and extraction 3 times, each 3 hours extraction times, get volatile oil and decocting liquid, with last medicinal residues extracting in water 2 times, each 0.5 hour, merge with the decocting liquid that begins to distill gained, suitably being concentrated into 50 ℃ of following relative densities is 1.20, put coldly, add ethanol precipitation and reach 80%, leave standstill to containing alcohol amount, get supernatant concentration recovery ethanol and get concentrated solution (1), standby; Follow the example of the Rhizoma Pinelliae and Rhizoma Zingiberis Recens, add 70% ethanol percolation, concentrated percolate gets concentrated solution (2), and is standby; Get Radix Codonopsis, Fructus Jujubae and Radix Glycyrrhizae, extracting in water 3 times, each 1 hour, it was 1.19 that merge extractive liquid, is concentrated into 50 ℃ of following relative densities, added ethanol precipitation and reached 70% to containing the alcohol amount, left standstill, and got supernatant concentration recovery ethanol and got concentrated solution (3), standby; Get Radix Scutellariae, add 10 times of water gagings and extract 2 times, each 1 hour, it was 1.5,80 ℃ of insulations that extracting solution adds dilute hydrochloric acid adjusting pH value, leaves standstill, and filters, and precipitation is regulated pH value to 5.5 with the second alcohol and water, and vacuum drying gets Radix Scutellariae extract; (1), (2), (3) concentrated solution of gained are merged, continue to be condensed into thick paste (4), 70 ℃ of following relative densities are 1.25;
(c) to above-mentioned thick paste (4), Radix Scutellariae extract, add in the Radix Bupleuri volatile oil 8 times of amounts, lactose: the ratio of starch weight is 1: 0.6 a adjuvant, fully mix, mixture stirs at 85~89 ℃ of heating and meltings, mixing time is 30 minutes, insulation is 1.8 millimeters at 63 ℃ of temperature following system, dropper bore, splashes in 0 ℃ the methyl-silicone oil, liquid coolant is use up and wiped to the drop pill drop that forms, back packing to be dried is made drop pill, promptly.
Embodiment 17
(a) get Radix Bupleuri 33.3g, Rhizoma Pinelliae Preparatum 166.7g, Radix Scutellariae 166.7g, Radix Codonopsis 166.7g, Fructus Jujubae 166.7g, Radix Glycyrrhizae 111.1g, Rhizoma Zingiberis Recens 88.9g, xylitol, arabic gum are standby;
(b) get Radix Bupleuri, add water distillation and extraction 3 times, each 3 hours extraction times, get volatile oil and decocting liquid, with last medicinal residues extracting in water 2 times, each 0.5 hour, merge with the decocting liquid that begins to distill gained, suitably being concentrated into 50 ℃ of following relative densities is 1.20, put coldly, add ethanol precipitation and reach 70%, leave standstill to containing alcohol amount, get supernatant concentration recovery ethanol and get concentrated solution (1), standby; Follow the example of the Rhizoma Pinelliae and Rhizoma Zingiberis Recens, add 70% ethanol percolation, concentrated percolate gets concentrated solution (2), and is standby; Get Radix Codonopsis, Fructus Jujubae and Radix Glycyrrhizae, extracting in water 3 times, each 1 hour, it was 1.15 that merge extractive liquid, is concentrated into 50 ℃ of following relative densities, added ethanol precipitation and reached 80% to containing the alcohol amount, left standstill, and got supernatant concentration recovery ethanol and got concentrated solution (3), standby; Get Radix Scutellariae, add 10 times of water gagings and extract 2 times, each 1 hour, it was 2.0,90 ℃ of insulations that extracting solution adds dilute hydrochloric acid adjusting pH value, leaves standstill, and filters, and precipitation is regulated pH value to 5.5 with the second alcohol and water, and vacuum drying gets Radix Scutellariae extract; (1), (2), (3) concentrated solution of gained are merged, continue to be condensed into thick paste (4);
(c) to above-mentioned thick paste (4), Radix Scutellariae extract, add in the Radix Bupleuri volatile oil 3 times of amounts, xylitol: the ratio of the weight of arabic gum is that 1: 0.4 amount is auxilliary, fully mix, mixture stirs at 95 ℃ of heating and meltings, mixing time is 15 minutes, insulation is 1.1~2.5 millimeters at 90 ℃ of temperature following system, dropper bore, splashes in 0~5 ℃ the methyl-silicone oil, liquid coolant is use up and wiped to the drop pill drop that forms, back packing to be dried is made drop pill, promptly.