CN100528156C - 在疼痛治疗中成瘾的预防 - Google Patents
在疼痛治疗中成瘾的预防 Download PDFInfo
- Publication number
- CN100528156C CN100528156C CNB028098277A CN02809827A CN100528156C CN 100528156 C CN100528156 C CN 100528156C CN B028098277 A CNB028098277 A CN B028098277A CN 02809827 A CN02809827 A CN 02809827A CN 100528156 C CN100528156 C CN 100528156C
- Authority
- CN
- China
- Prior art keywords
- gaba
- analgesic
- days
- medicament
- gvg
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Expired - Fee Related
Links
- 206010012335 Dependence Diseases 0.000 title claims description 42
- 208000002193 Pain Diseases 0.000 title abstract description 36
- 230000036407 pain Effects 0.000 title abstract description 36
- 230000002265 prevention Effects 0.000 title description 5
- 230000000202 analgesic effect Effects 0.000 claims abstract description 83
- PJDFLNIOAUIZSL-UHFFFAOYSA-N vigabatrin Chemical compound C=CC(N)CCC(O)=O PJDFLNIOAUIZSL-UHFFFAOYSA-N 0.000 claims abstract description 53
- 239000000203 mixture Substances 0.000 claims abstract description 42
- 239000003814 drug Substances 0.000 claims description 84
- BQJCRHHNABKAKU-KBQPJGBKSA-N morphine Chemical compound O([C@H]1[C@H](C=C[C@H]23)O)C4=C5[C@@]12CCN(C)[C@@H]3CC5=CC=C4O BQJCRHHNABKAKU-KBQPJGBKSA-N 0.000 claims description 50
- 229960005181 morphine Drugs 0.000 claims description 25
- 208000010513 Stupor Diseases 0.000 claims description 14
- 238000002360 preparation method Methods 0.000 claims description 4
- 238000000034 method Methods 0.000 abstract description 16
- 239000003445 gaba agent Substances 0.000 abstract 2
- 229960003692 gamma aminobutyric acid Drugs 0.000 description 71
- BTCSSZJGUNDROE-UHFFFAOYSA-N gamma-aminobutyric acid Chemical compound NCCCC(O)=O BTCSSZJGUNDROE-UHFFFAOYSA-N 0.000 description 71
- VYFYYTLLBUKUHU-UHFFFAOYSA-N dopamine Chemical compound NCCC1=CC=C(O)C(O)=C1 VYFYYTLLBUKUHU-UHFFFAOYSA-N 0.000 description 58
- 241001465754 Metazoa Species 0.000 description 30
- 229960003638 dopamine Drugs 0.000 description 29
- 230000000694 effects Effects 0.000 description 17
- -1 for adding guest Chemical compound 0.000 description 17
- 241000124008 Mammalia Species 0.000 description 15
- UGJMXCAKCUNAIE-UHFFFAOYSA-N Gabapentin Chemical compound OC(=O)CC1(CN)CCCCC1 UGJMXCAKCUNAIE-UHFFFAOYSA-N 0.000 description 14
- GVGLGOZIDCSQPN-PVHGPHFFSA-N Heroin Chemical compound O([C@H]1[C@H](C=C[C@H]23)OC(C)=O)C4=C5[C@@]12CCN(C)[C@@H]3CC5=CC=C4OC(C)=O GVGLGOZIDCSQPN-PVHGPHFFSA-N 0.000 description 14
- 210000003169 central nervous system Anatomy 0.000 description 14
- 229960002069 diamorphine Drugs 0.000 description 14
- 229940079593 drug Drugs 0.000 description 14
- 150000001875 compounds Chemical class 0.000 description 13
- 238000002347 injection Methods 0.000 description 12
- 239000007924 injection Substances 0.000 description 12
- FAPWRFPIFSIZLT-UHFFFAOYSA-M Sodium chloride Chemical compound [Na+].[Cl-] FAPWRFPIFSIZLT-UHFFFAOYSA-M 0.000 description 11
- IBALRBWGSVJPAP-HEHNFIMWSA-N progabide Chemical compound C=1C(F)=CC=C(O)C=1C(=N/CCCC(=O)N)/C1=CC=C(Cl)C=C1 IBALRBWGSVJPAP-HEHNFIMWSA-N 0.000 description 10
- 229960002752 progabide Drugs 0.000 description 10
- 239000011780 sodium chloride Substances 0.000 description 10
- ZGLIFVFRIOKQLE-LVZFUZTISA-N chembl2104573 Chemical compound C=1C(Cl)=CC=C(O)C=1C(=N/CCCC)/C1=CC=CC=C1Cl ZGLIFVFRIOKQLE-LVZFUZTISA-N 0.000 description 9
- 229950004395 fengabine Drugs 0.000 description 9
- SJZRECIVHVDYJC-UHFFFAOYSA-N 4-hydroxybutyric acid Chemical compound OCCCC(O)=O SJZRECIVHVDYJC-UHFFFAOYSA-N 0.000 description 8
- 230000036592 analgesia Effects 0.000 description 8
- OROGSEYTTFOCAN-DNJOTXNNSA-N codeine Chemical compound C([C@H]1[C@H](N(CC[C@@]112)C)C3)=C[C@H](O)[C@@H]1OC1=C2C3=CC=C1OC OROGSEYTTFOCAN-DNJOTXNNSA-N 0.000 description 8
- 238000011534 incubation Methods 0.000 description 8
- 230000002787 reinforcement Effects 0.000 description 8
- 239000000126 substance Substances 0.000 description 8
- 241000700159 Rattus Species 0.000 description 7
- KJADKKWYZYXHBB-XBWDGYHZSA-N Topiramic acid Chemical compound C1O[C@@]2(COS(N)(=O)=O)OC(C)(C)O[C@H]2[C@@H]2OC(C)(C)O[C@@H]21 KJADKKWYZYXHBB-XBWDGYHZSA-N 0.000 description 7
- 230000008859 change Effects 0.000 description 7
- 238000006243 chemical reaction Methods 0.000 description 7
- 230000001143 conditioned effect Effects 0.000 description 7
- NIJJYAXOARWZEE-UHFFFAOYSA-N di-n-propyl-acetic acid Natural products CCCC(C(O)=O)CCC NIJJYAXOARWZEE-UHFFFAOYSA-N 0.000 description 7
- 210000004744 fore-foot Anatomy 0.000 description 7
- 229960002870 gabapentin Drugs 0.000 description 7
- 239000000523 sample Substances 0.000 description 7
- 230000000638 stimulation Effects 0.000 description 7
- 238000002560 therapeutic procedure Methods 0.000 description 7
- 229960004394 topiramate Drugs 0.000 description 7
- MSRILKIQRXUYCT-UHFFFAOYSA-M valproate semisodium Chemical compound [Na+].CCCC(C(O)=O)CCC.CCCC(C([O-])=O)CCC MSRILKIQRXUYCT-UHFFFAOYSA-M 0.000 description 7
- 229960000604 valproic acid Drugs 0.000 description 7
- 210000001015 abdomen Anatomy 0.000 description 6
- 238000004458 analytical method Methods 0.000 description 6
- OROGSEYTTFOCAN-UHFFFAOYSA-N hydrocodone Natural products C1C(N(CCC234)C)C2C=CC(O)C3OC2=C4C1=CC=C2OC OROGSEYTTFOCAN-UHFFFAOYSA-N 0.000 description 6
- 239000007788 liquid Substances 0.000 description 6
- 238000011160 research Methods 0.000 description 6
- 150000003839 salts Chemical class 0.000 description 6
- 238000003639 Student–Newman–Keuls (SNK) method Methods 0.000 description 5
- 239000000730 antalgic agent Substances 0.000 description 5
- 230000037396 body weight Effects 0.000 description 5
- 206010013663 drug dependence Diseases 0.000 description 5
- 230000001939 inductive effect Effects 0.000 description 5
- 208000011117 substance-related disease Diseases 0.000 description 5
- 108010060511 4-Aminobutyrate Transaminase Proteins 0.000 description 4
- 102100035923 4-aminobutyrate aminotransferase, mitochondrial Human genes 0.000 description 4
- DEXMFYZAHXMZNM-UHFFFAOYSA-N Narceine Chemical compound OC(=O)C1=C(OC)C(OC)=CC=C1C(=O)CC1=C(CCN(C)C)C=C(OCO2)C2=C1OC DEXMFYZAHXMZNM-UHFFFAOYSA-N 0.000 description 4
- 239000008896 Opium Substances 0.000 description 4
- 239000000443 aerosol Substances 0.000 description 4
- 229940035676 analgesics Drugs 0.000 description 4
- 238000010171 animal model Methods 0.000 description 4
- 229960004126 codeine Drugs 0.000 description 4
- 239000007859 condensation product Substances 0.000 description 4
- 235000019788 craving Nutrition 0.000 description 4
- XYYVYLMBEZUESM-UHFFFAOYSA-N dihydrocodeine Natural products C1C(N(CCC234)C)C2C=CC(=O)C3OC2=C4C1=CC=C2OC XYYVYLMBEZUESM-UHFFFAOYSA-N 0.000 description 4
- 201000010099 disease Diseases 0.000 description 4
- 208000037265 diseases, disorders, signs and symptoms Diseases 0.000 description 4
- 150000002148 esters Chemical class 0.000 description 4
- 230000005764 inhibitory process Effects 0.000 description 4
- 238000007912 intraperitoneal administration Methods 0.000 description 4
- 238000001690 micro-dialysis Methods 0.000 description 4
- 210000002569 neuron Anatomy 0.000 description 4
- 210000001009 nucleus accumben Anatomy 0.000 description 4
- 229960001027 opium Drugs 0.000 description 4
- 239000000843 powder Substances 0.000 description 4
- 102000005962 receptors Human genes 0.000 description 4
- 108020003175 receptors Proteins 0.000 description 4
- 230000001105 regulatory effect Effects 0.000 description 4
- 208000024891 symptom Diseases 0.000 description 4
- 238000012360 testing method Methods 0.000 description 4
- 230000001225 therapeutic effect Effects 0.000 description 4
- 239000003981 vehicle Substances 0.000 description 4
- QTBSBXVTEAMEQO-UHFFFAOYSA-N Acetic acid Chemical compound CC(O)=O QTBSBXVTEAMEQO-UHFFFAOYSA-N 0.000 description 3
- FBPFZTCFMRRESA-FSIIMWSLSA-N D-Glucitol Natural products OC[C@H](O)[C@H](O)[C@@H](O)[C@H](O)CO FBPFZTCFMRRESA-FSIIMWSLSA-N 0.000 description 3
- IJVCSMSMFSCRME-KBQPJGBKSA-N Dihydromorphine Chemical compound O([C@H]1[C@H](CC[C@H]23)O)C4=C5[C@@]12CCN(C)[C@@H]3CC5=CC=C4O IJVCSMSMFSCRME-KBQPJGBKSA-N 0.000 description 3
- IAYPIBMASNFSPL-UHFFFAOYSA-N Ethylene oxide Chemical compound C1CO1 IAYPIBMASNFSPL-UHFFFAOYSA-N 0.000 description 3
- OKKJLVBELUTLKV-UHFFFAOYSA-N Methanol Chemical compound OC OKKJLVBELUTLKV-UHFFFAOYSA-N 0.000 description 3
- 229920003171 Poly (ethylene oxide) Polymers 0.000 description 3
- AFCGFAGUEYAMAO-UHFFFAOYSA-N acamprosate Chemical compound CC(=O)NCCCS(O)(=O)=O AFCGFAGUEYAMAO-UHFFFAOYSA-N 0.000 description 3
- 229960004047 acamprosate Drugs 0.000 description 3
- 230000008901 benefit Effects 0.000 description 3
- 210000004556 brain Anatomy 0.000 description 3
- 239000003795 chemical substances by application Substances 0.000 description 3
- KRKNYBCHXYNGOX-UHFFFAOYSA-N citric acid Chemical compound OC(=O)CC(O)(C(O)=O)CC(O)=O KRKNYBCHXYNGOX-UHFFFAOYSA-N 0.000 description 3
- 230000003750 conditioning effect Effects 0.000 description 3
- 235000014113 dietary fatty acids Nutrition 0.000 description 3
- 230000005284 excitation Effects 0.000 description 3
- 239000000194 fatty acid Substances 0.000 description 3
- 229930195729 fatty acid Natural products 0.000 description 3
- 150000004665 fatty acids Chemical class 0.000 description 3
- 210000001035 gastrointestinal tract Anatomy 0.000 description 3
- FQZPXSRKCOWUEI-UHFFFAOYSA-N hexadecyl 4-aminobutanoate Chemical compound CCCCCCCCCCCCCCCCOC(=O)CCCN FQZPXSRKCOWUEI-UHFFFAOYSA-N 0.000 description 3
- 230000002427 irreversible effect Effects 0.000 description 3
- 230000003533 narcotic effect Effects 0.000 description 3
- 239000000651 prodrug Substances 0.000 description 3
- 229940002612 prodrug Drugs 0.000 description 3
- 230000004044 response Effects 0.000 description 3
- 239000007787 solid Substances 0.000 description 3
- 239000000600 sorbitol Substances 0.000 description 3
- 239000007921 spray Substances 0.000 description 3
- 239000006188 syrup Substances 0.000 description 3
- 235000020357 syrup Nutrition 0.000 description 3
- 239000003826 tablet Substances 0.000 description 3
- YQYVFVRQLZMJKJ-JBBXEZCESA-N (+)-cyclazocine Chemical compound C([C@@]1(C)C2=CC(O)=CC=C2C[C@@H]2[C@@H]1C)CN2CC1CC1 YQYVFVRQLZMJKJ-JBBXEZCESA-N 0.000 description 2
- UVITTYOJFDLOGI-UHFFFAOYSA-N (1,2,5-trimethyl-4-phenylpiperidin-4-yl) propanoate Chemical compound C=1C=CC=CC=1C1(OC(=O)CC)CC(C)N(C)CC1C UVITTYOJFDLOGI-UHFFFAOYSA-N 0.000 description 2
- PJDFLNIOAUIZSL-YFKPBYRVSA-N (4r)-4-azaniumylhex-5-enoate Chemical compound C=C[C@H](N)CCC(O)=O PJDFLNIOAUIZSL-YFKPBYRVSA-N 0.000 description 2
- PJDFLNIOAUIZSL-RXMQYKEDSA-N (4s)-4-azaniumylhex-5-enoate Chemical compound C=C[C@@H](N)CCC(O)=O PJDFLNIOAUIZSL-RXMQYKEDSA-N 0.000 description 2
- TVYLLZQTGLZFBW-ZBFHGGJFSA-N (R,R)-tramadol Chemical compound COC1=CC=CC([C@]2(O)[C@H](CCCC2)CN(C)C)=C1 TVYLLZQTGLZFBW-ZBFHGGJFSA-N 0.000 description 2
- IYNWSQDZXMGGGI-NUEKZKHPSA-N 3-hydroxymorphinan Chemical compound C1CCC[C@H]2[C@H]3CC4=CC=C(O)C=C4[C@]21CCN3 IYNWSQDZXMGGGI-NUEKZKHPSA-N 0.000 description 2
- USSIQXCVUWKGNF-UHFFFAOYSA-N 6-(dimethylamino)-4,4-diphenylheptan-3-one Chemical compound C=1C=CC=CC=1C(CC(C)N(C)C)(C(=O)CC)C1=CC=CC=C1 USSIQXCVUWKGNF-UHFFFAOYSA-N 0.000 description 2
- VTYYLEPIZMXCLO-UHFFFAOYSA-L Calcium carbonate Chemical compound [Ca+2].[O-]C([O-])=O VTYYLEPIZMXCLO-UHFFFAOYSA-L 0.000 description 2
- CURLTUGMZLYLDI-UHFFFAOYSA-N Carbon dioxide Chemical compound O=C=O CURLTUGMZLYLDI-UHFFFAOYSA-N 0.000 description 2
- RGHNJXZEOKUKBD-SQOUGZDYSA-N D-gluconic acid Chemical compound OC[C@@H](O)[C@@H](O)[C@H](O)[C@@H](O)C(O)=O RGHNJXZEOKUKBD-SQOUGZDYSA-N 0.000 description 2
- 206010013754 Drug withdrawal syndrome Diseases 0.000 description 2
- 108010065372 Dynorphins Proteins 0.000 description 2
- 241000196324 Embryophyta Species 0.000 description 2
- OGDVEMNWJVYAJL-LEPYJNQMSA-N Ethyl morphine Chemical compound C([C@H]1[C@H](N(CC[C@@]112)C)C3)=C[C@H](O)[C@@H]1OC1=C2C3=CC=C1OCC OGDVEMNWJVYAJL-LEPYJNQMSA-N 0.000 description 2
- VEXZGXHMUGYJMC-UHFFFAOYSA-N Hydrochloric acid Chemical compound Cl VEXZGXHMUGYJMC-UHFFFAOYSA-N 0.000 description 2
- ALFGKMXHOUSVAD-UHFFFAOYSA-N Ketobemidone Chemical compound C=1C=CC(O)=CC=1C1(C(=O)CC)CCN(C)CC1 ALFGKMXHOUSVAD-UHFFFAOYSA-N 0.000 description 2
- OZYUPQUCAUTOBP-QXAKKESOSA-N Levallorphan Chemical compound C([C@H]12)CCC[C@@]11CCN(CC=C)[C@@H]2CC2=CC=C(O)C=C21 OZYUPQUCAUTOBP-QXAKKESOSA-N 0.000 description 2
- JAQUASYNZVUNQP-USXIJHARSA-N Levorphanol Chemical compound C1C2=CC=C(O)C=C2[C@]23CCN(C)[C@H]1[C@@H]2CCCC3 JAQUASYNZVUNQP-USXIJHARSA-N 0.000 description 2
- XADCESSVHJOZHK-UHFFFAOYSA-N Meperidine Chemical compound C=1C=CC=CC=1C1(C(=O)OCC)CCN(C)CC1 XADCESSVHJOZHK-UHFFFAOYSA-N 0.000 description 2
- IDBPHNDTYPBSNI-UHFFFAOYSA-N N-(1-(2-(4-Ethyl-5-oxo-2-tetrazolin-1-yl)ethyl)-4-(methoxymethyl)-4-piperidyl)propionanilide Chemical compound C1CN(CCN2C(N(CC)N=N2)=O)CCC1(COC)N(C(=O)CC)C1=CC=CC=C1 IDBPHNDTYPBSNI-UHFFFAOYSA-N 0.000 description 2
- UIQMVEYFGZJHCZ-SSTWWWIQSA-N Nalorphine Chemical compound C([C@@H](N(CC1)CC=C)[C@@H]2C=C[C@@H]3O)C4=CC=C(O)C5=C4[C@@]21[C@H]3O5 UIQMVEYFGZJHCZ-SSTWWWIQSA-N 0.000 description 2
- ONBWJWYUHXVEJS-ZTYRTETDSA-N Normorphine Chemical compound C([C@@H](NCC1)[C@@H]2C=C[C@@H]3O)C4=CC=C(O)C5=C4[C@@]21[C@H]3O5 ONBWJWYUHXVEJS-ZTYRTETDSA-N 0.000 description 2
- MITFXPHMIHQXPI-UHFFFAOYSA-N Oraflex Chemical compound N=1C2=CC(C(C(O)=O)C)=CC=C2OC=1C1=CC=C(Cl)C=C1 MITFXPHMIHQXPI-UHFFFAOYSA-N 0.000 description 2
- BRUQQQPBMZOVGD-XFKAJCMBSA-N Oxycodone Chemical compound O=C([C@@H]1O2)CC[C@@]3(O)[C@H]4CC5=CC=C(OC)C2=C5[C@@]13CCN4C BRUQQQPBMZOVGD-XFKAJCMBSA-N 0.000 description 2
- UQCNKQCJZOAFTQ-ISWURRPUSA-N Oxymorphone Chemical compound O([C@H]1C(CC[C@]23O)=O)C4=C5[C@@]12CCN(C)[C@@H]3CC5=CC=C4O UQCNKQCJZOAFTQ-ISWURRPUSA-N 0.000 description 2
- NBIIXXVUZAFLBC-UHFFFAOYSA-N Phosphoric acid Chemical compound OP(O)(O)=O NBIIXXVUZAFLBC-UHFFFAOYSA-N 0.000 description 2
- 208000004550 Postoperative Pain Diseases 0.000 description 2
- 102100038931 Proenkephalin-A Human genes 0.000 description 2
- 102100024622 Proenkephalin-B Human genes 0.000 description 2
- 208000007271 Substance Withdrawal Syndrome Diseases 0.000 description 2
- 239000000556 agonist Substances 0.000 description 2
- 229960001391 alfentanil Drugs 0.000 description 2
- KGYFOSCXVAXULR-UHFFFAOYSA-N allylprodine Chemical compound C=1C=CC=CC=1C1(OC(=O)CC)CCN(C)CC1CC=C KGYFOSCXVAXULR-UHFFFAOYSA-N 0.000 description 2
- 229950004361 allylprodine Drugs 0.000 description 2
- 229960001349 alphaprodine Drugs 0.000 description 2
- UVAZQQHAVMNMHE-XJKSGUPXSA-N alphaprodine Chemical compound C=1C=CC=CC=1[C@@]1(OC(=O)CC)CCN(C)C[C@@H]1C UVAZQQHAVMNMHE-XJKSGUPXSA-N 0.000 description 2
- LKYQLAWMNBFNJT-UHFFFAOYSA-N anileridine Chemical compound C1CC(C(=O)OCC)(C=2C=CC=CC=2)CCN1CCC1=CC=C(N)C=C1 LKYQLAWMNBFNJT-UHFFFAOYSA-N 0.000 description 2
- 229960002512 anileridine Drugs 0.000 description 2
- 238000003975 animal breeding Methods 0.000 description 2
- 230000003042 antagnostic effect Effects 0.000 description 2
- 238000013459 approach Methods 0.000 description 2
- 239000007900 aqueous suspension Substances 0.000 description 2
- WPYMKLBDIGXBTP-UHFFFAOYSA-N benzoic acid Chemical compound OC(=O)C1=CC=CC=C1 WPYMKLBDIGXBTP-UHFFFAOYSA-N 0.000 description 2
- RDJGWRFTDZZXSM-RNWLQCGYSA-N benzylmorphine Chemical compound O([C@@H]1[C@]23CCN([C@H](C4)[C@@H]3C=C[C@@H]1O)C)C1=C2C4=CC=C1OCC1=CC=CC=C1 RDJGWRFTDZZXSM-RNWLQCGYSA-N 0.000 description 2
- FLKWNFFCSSJANB-UHFFFAOYSA-N bezitramide Chemical compound O=C1N(C(=O)CC)C2=CC=CC=C2N1C(CC1)CCN1CCC(C#N)(C=1C=CC=CC=1)C1=CC=CC=C1 FLKWNFFCSSJANB-UHFFFAOYSA-N 0.000 description 2
- 229960004611 bezitramide Drugs 0.000 description 2
- RMRJXGBAOAMLHD-IHFGGWKQSA-N buprenorphine Chemical compound C([C@]12[C@H]3OC=4C(O)=CC=C(C2=4)C[C@@H]2[C@]11CC[C@]3([C@H](C1)[C@](C)(O)C(C)(C)C)OC)CN2CC1CC1 RMRJXGBAOAMLHD-IHFGGWKQSA-N 0.000 description 2
- 229960001736 buprenorphine Drugs 0.000 description 2
- IFKLAQQSCNILHL-QHAWAJNXSA-N butorphanol Chemical compound N1([C@@H]2CC3=CC=C(C=C3[C@@]3([C@]2(CCCC3)O)CC1)O)CC1CCC1 IFKLAQQSCNILHL-QHAWAJNXSA-N 0.000 description 2
- 229960001113 butorphanol Drugs 0.000 description 2
- 239000011575 calcium Substances 0.000 description 2
- 239000002775 capsule Substances 0.000 description 2
- 210000004027 cell Anatomy 0.000 description 2
- 210000001175 cerebrospinal fluid Anatomy 0.000 description 2
- 239000003153 chemical reaction reagent Substances 0.000 description 2
- 230000001684 chronic effect Effects 0.000 description 2
- GPZLDQAEBHTMPG-UHFFFAOYSA-N clonitazene Chemical compound N=1C2=CC([N+]([O-])=O)=CC=C2N(CCN(CC)CC)C=1CC1=CC=C(Cl)C=C1 GPZLDQAEBHTMPG-UHFFFAOYSA-N 0.000 description 2
- 229950001604 clonitazene Drugs 0.000 description 2
- 239000003086 colorant Substances 0.000 description 2
- 229950002213 cyclazocine Drugs 0.000 description 2
- 229950003851 desomorphine Drugs 0.000 description 2
- LNNWVNGFPYWNQE-GMIGKAJZSA-N desomorphine Chemical compound C1C2=CC=C(O)C3=C2[C@]24CCN(C)[C@H]1[C@@H]2CCC[C@@H]4O3 LNNWVNGFPYWNQE-GMIGKAJZSA-N 0.000 description 2
- WDEFBBTXULIOBB-WBVHZDCISA-N dextilidine Chemical compound C=1C=CC=CC=1[C@@]1(C(=O)OCC)CCC=C[C@H]1N(C)C WDEFBBTXULIOBB-WBVHZDCISA-N 0.000 description 2
- 229960003701 dextromoramide Drugs 0.000 description 2
- INUNXTSAACVKJS-OAQYLSRUSA-N dextromoramide Chemical compound C([C@@H](C)C(C(=O)N1CCCC1)(C=1C=CC=CC=1)C=1C=CC=CC=1)N1CCOCC1 INUNXTSAACVKJS-OAQYLSRUSA-N 0.000 description 2
- 229960004193 dextropropoxyphene Drugs 0.000 description 2
- XLMALTXPSGQGBX-GCJKJVERSA-N dextropropoxyphene Chemical compound C([C@](OC(=O)CC)([C@H](C)CN(C)C)C=1C=CC=CC=1)C1=CC=CC=C1 XLMALTXPSGQGBX-GCJKJVERSA-N 0.000 description 2
- 229960003461 dezocine Drugs 0.000 description 2
- VTMVHDZWSFQSQP-VBNZEHGJSA-N dezocine Chemical compound C1CCCC[C@H]2CC3=CC=C(O)C=C3[C@]1(C)[C@H]2N VTMVHDZWSFQSQP-VBNZEHGJSA-N 0.000 description 2
- RXTHKWVSXOIHJS-UHFFFAOYSA-N diampromide Chemical compound C=1C=CC=CC=1N(C(=O)CC)CC(C)N(C)CCC1=CC=CC=C1 RXTHKWVSXOIHJS-UHFFFAOYSA-N 0.000 description 2
- 229950001059 diampromide Drugs 0.000 description 2
- 229960000920 dihydrocodeine Drugs 0.000 description 2
- RBOXVHNMENFORY-DNJOTXNNSA-N dihydrocodeine Chemical compound C([C@H]1[C@H](N(CC[C@@]112)C)C3)C[C@H](O)[C@@H]1OC1=C2C3=CC=C1OC RBOXVHNMENFORY-DNJOTXNNSA-N 0.000 description 2
- 239000003085 diluting agent Substances 0.000 description 2
- RHUWRJWFHUKVED-UHFFFAOYSA-N dimenoxadol Chemical compound C=1C=CC=CC=1C(C(=O)OCCN(C)C)(OCC)C1=CC=CC=C1 RHUWRJWFHUKVED-UHFFFAOYSA-N 0.000 description 2
- 229950011187 dimenoxadol Drugs 0.000 description 2
- QIRAYNIFEOXSPW-UHFFFAOYSA-N dimepheptanol Chemical compound C=1C=CC=CC=1C(CC(C)N(C)C)(C(O)CC)C1=CC=CC=C1 QIRAYNIFEOXSPW-UHFFFAOYSA-N 0.000 description 2
- 229950004655 dimepheptanol Drugs 0.000 description 2
- CANBGVXYBPOLRR-UHFFFAOYSA-N dimethylthiambutene Chemical compound C=1C=CSC=1C(=CC(C)N(C)C)C1=CC=CS1 CANBGVXYBPOLRR-UHFFFAOYSA-N 0.000 description 2
- 229950005563 dimethylthiambutene Drugs 0.000 description 2
- LQGIXNQCOXNCRP-UHFFFAOYSA-N dioxaphetyl butyrate Chemical compound C=1C=CC=CC=1C(C=1C=CC=CC=1)(C(=O)OCC)CCN1CCOCC1 LQGIXNQCOXNCRP-UHFFFAOYSA-N 0.000 description 2
- 229950008972 dioxaphetyl butyrate Drugs 0.000 description 2
- SVDHSZFEQYXRDC-UHFFFAOYSA-N dipipanone Chemical compound C=1C=CC=CC=1C(C=1C=CC=CC=1)(C(=O)CC)CC(C)N1CCCCC1 SVDHSZFEQYXRDC-UHFFFAOYSA-N 0.000 description 2
- 229960002500 dipipanone Drugs 0.000 description 2
- ZOWQTJXNFTWSCS-IAQYHMDHSA-N eptazocine Chemical compound C1N(C)CC[C@@]2(C)C3=CC(O)=CC=C3C[C@@H]1C2 ZOWQTJXNFTWSCS-IAQYHMDHSA-N 0.000 description 2
- 229950010920 eptazocine Drugs 0.000 description 2
- WGJHHMKQBWSQIY-UHFFFAOYSA-N ethoheptazine Chemical compound C=1C=CC=CC=1C1(C(=O)OCC)CCCN(C)CC1 WGJHHMKQBWSQIY-UHFFFAOYSA-N 0.000 description 2
- 229960000569 ethoheptazine Drugs 0.000 description 2
- MORSAEFGQPDBKM-UHFFFAOYSA-N ethylmethylthiambutene Chemical compound C=1C=CSC=1C(=CC(C)N(C)CC)C1=CC=CS1 MORSAEFGQPDBKM-UHFFFAOYSA-N 0.000 description 2
- 229950006111 ethylmethylthiambutene Drugs 0.000 description 2
- PXDBZSCGSQSKST-UHFFFAOYSA-N etonitazene Chemical compound C1=CC(OCC)=CC=C1CC1=NC2=CC([N+]([O-])=O)=CC=C2N1CCN(CC)CC PXDBZSCGSQSKST-UHFFFAOYSA-N 0.000 description 2
- 229950004538 etonitazene Drugs 0.000 description 2
- 229960002428 fentanyl Drugs 0.000 description 2
- IVLVTNPOHDFFCJ-UHFFFAOYSA-N fentanyl citrate Chemical compound OC(=O)CC(O)(C(O)=O)CC(O)=O.C=1C=CC=CC=1N(C(=O)CC)C(CC1)CCN1CCC1=CC=CC=C1 IVLVTNPOHDFFCJ-UHFFFAOYSA-N 0.000 description 2
- 230000006870 function Effects 0.000 description 2
- LLPOLZWFYMWNKH-CMKMFDCUSA-N hydrocodone Chemical compound C([C@H]1[C@H](N(CC[C@@]112)C)C3)CC(=O)[C@@H]1OC1=C2C3=CC=C1OC LLPOLZWFYMWNKH-CMKMFDCUSA-N 0.000 description 2
- 229960000240 hydrocodone Drugs 0.000 description 2
- WVLOADHCBXTIJK-YNHQPCIGSA-N hydromorphone Chemical compound O([C@H]1C(CC[C@H]23)=O)C4=C5[C@@]12CCN(C)[C@@H]3CC5=CC=C4O WVLOADHCBXTIJK-YNHQPCIGSA-N 0.000 description 2
- 229960001410 hydromorphone Drugs 0.000 description 2
- WTJBNMUWRKPFRS-UHFFFAOYSA-N hydroxypethidine Chemical compound C=1C=CC(O)=CC=1C1(C(=O)OCC)CCN(C)CC1 WTJBNMUWRKPFRS-UHFFFAOYSA-N 0.000 description 2
- 229950008496 hydroxypethidine Drugs 0.000 description 2
- 238000001727 in vivo Methods 0.000 description 2
- 230000001965 increasing effect Effects 0.000 description 2
- CGIGDMFJXJATDK-UHFFFAOYSA-N indomethacin Chemical compound CC1=C(CC(O)=O)C2=CC(OC)=CC=C2N1C(=O)C1=CC=C(Cl)C=C1 CGIGDMFJXJATDK-UHFFFAOYSA-N 0.000 description 2
- 230000000968 intestinal effect Effects 0.000 description 2
- 230000007794 irritation Effects 0.000 description 2
- IFKPLJWIEQBPGG-UHFFFAOYSA-N isomethadone Chemical compound C=1C=CC=CC=1C(C(C)CN(C)C)(C(=O)CC)C1=CC=CC=C1 IFKPLJWIEQBPGG-UHFFFAOYSA-N 0.000 description 2
- 229950009272 isomethadone Drugs 0.000 description 2
- 229960003029 ketobemidone Drugs 0.000 description 2
- 229960000263 levallorphan Drugs 0.000 description 2
- RCYBMSQOSGJZLO-BGWNEDDSSA-N levophenacylmorphan Chemical compound C([C@]12CCCC[C@H]1[C@H]1CC3=CC=C(C=C32)O)CN1CC(=O)C1=CC=CC=C1 RCYBMSQOSGJZLO-BGWNEDDSSA-N 0.000 description 2
- 229950007939 levophenacylmorphan Drugs 0.000 description 2
- 229960003406 levorphanol Drugs 0.000 description 2
- 229950010274 lofentanil Drugs 0.000 description 2
- IMYHGORQCPYVBZ-NLFFAJNJSA-N lofentanil Chemical compound CCC(=O)N([C@@]1([C@@H](CN(CCC=2C=CC=CC=2)CC1)C)C(=O)OC)C1=CC=CC=C1 IMYHGORQCPYVBZ-NLFFAJNJSA-N 0.000 description 2
- 239000000463 material Substances 0.000 description 2
- 238000005259 measurement Methods 0.000 description 2
- 229960000365 meptazinol Drugs 0.000 description 2
- JLICHNCFTLFZJN-HNNXBMFYSA-N meptazinol Chemical compound C=1C=CC(O)=CC=1[C@@]1(CC)CCCCN(C)C1 JLICHNCFTLFZJN-HNNXBMFYSA-N 0.000 description 2
- 229950009131 metazocine Drugs 0.000 description 2
- YGSVZRIZCHZUHB-COLVAYQJSA-N metazocine Chemical compound C1C2=CC=C(O)C=C2[C@]2(C)CCN(C)[C@@]1([H])[C@@H]2C YGSVZRIZCHZUHB-COLVAYQJSA-N 0.000 description 2
- 229960001797 methadone Drugs 0.000 description 2
- NPZXCTIHHUUEEJ-CMKMFDCUSA-N metopon Chemical compound O([C@@]1(C)C(=O)CC[C@@H]23)C4=C5[C@@]13CCN(C)[C@@H]2CC5=CC=C4O NPZXCTIHHUUEEJ-CMKMFDCUSA-N 0.000 description 2
- 229950006080 metopon Drugs 0.000 description 2
- 238000012986 modification Methods 0.000 description 2
- 230000004048 modification Effects 0.000 description 2
- 230000008450 motivation Effects 0.000 description 2
- GODGZZGKTZQSAL-VXFFQEMOSA-N myrophine Chemical compound C([C@@H]1[C@@H]2C=C[C@@H]([C@@H]3OC4=C5[C@]23CCN1C)OC(=O)CCCCCCCCCCCCC)C5=CC=C4OCC1=CC=CC=C1 GODGZZGKTZQSAL-VXFFQEMOSA-N 0.000 description 2
- 229950007471 myrophine Drugs 0.000 description 2
- NETZHAKZCGBWSS-CEDHKZHLSA-N nalbuphine Chemical compound C([C@]12[C@H]3OC=4C(O)=CC=C(C2=4)C[C@@H]2[C@]1(O)CC[C@@H]3O)CN2CC1CCC1 NETZHAKZCGBWSS-CEDHKZHLSA-N 0.000 description 2
- 229960000805 nalbuphine Drugs 0.000 description 2
- 229960000938 nalorphine Drugs 0.000 description 2
- 230000007935 neutral effect Effects 0.000 description 2
- 229960004300 nicomorphine Drugs 0.000 description 2
- HNDXBGYRMHRUFN-CIVUWBIHSA-N nicomorphine Chemical compound O([C@H]1C=C[C@H]2[C@H]3CC=4C5=C(C(=CC=4)OC(=O)C=4C=NC=CC=4)O[C@@H]1[C@]52CCN3C)C(=O)C1=CC=CN=C1 HNDXBGYRMHRUFN-CIVUWBIHSA-N 0.000 description 2
- 229950011519 norlevorphanol Drugs 0.000 description 2
- WCJFBSYALHQBSK-UHFFFAOYSA-N normethadone Chemical compound C=1C=CC=CC=1C(CCN(C)C)(C(=O)CC)C1=CC=CC=C1 WCJFBSYALHQBSK-UHFFFAOYSA-N 0.000 description 2
- 229960004013 normethadone Drugs 0.000 description 2
- 229950006134 normorphine Drugs 0.000 description 2
- WCDSHELZWCOTMI-UHFFFAOYSA-N norpipanone Chemical compound C=1C=CC=CC=1C(C=1C=CC=CC=1)(C(=O)CC)CCN1CCCCC1 WCDSHELZWCOTMI-UHFFFAOYSA-N 0.000 description 2
- 229950007418 norpipanone Drugs 0.000 description 2
- 239000000014 opioid analgesic Substances 0.000 description 2
- 229940005483 opioid analgesics Drugs 0.000 description 2
- 229960002085 oxycodone Drugs 0.000 description 2
- 229960005118 oxymorphone Drugs 0.000 description 2
- VOKSWYLNZZRQPF-GDIGMMSISA-N pentazocine Chemical compound C1C2=CC=C(O)C=C2[C@@]2(C)[C@@H](C)[C@@H]1N(CC=C(C)C)CC2 VOKSWYLNZZRQPF-GDIGMMSISA-N 0.000 description 2
- 229960005301 pentazocine Drugs 0.000 description 2
- 230000010412 perfusion Effects 0.000 description 2
- 229960000482 pethidine Drugs 0.000 description 2
- 239000000546 pharmaceutical excipient Substances 0.000 description 2
- LOXCOAXRHYDLOW-UHFFFAOYSA-N phenadoxone Chemical compound C=1C=CC=CC=1C(C=1C=CC=CC=1)(C(=O)CC)CC(C)N1CCOCC1 LOXCOAXRHYDLOW-UHFFFAOYSA-N 0.000 description 2
- 229950004540 phenadoxone Drugs 0.000 description 2
- ZQHYKVKNPWDQSL-KNXBSLHKSA-N phenazocine Chemical compound C([C@@]1(C)C2=CC(O)=CC=C2C[C@@H]2[C@@H]1C)CN2CCC1=CC=CC=C1 ZQHYKVKNPWDQSL-KNXBSLHKSA-N 0.000 description 2
- 229960000897 phenazocine Drugs 0.000 description 2
- CFBQYWXPZVQQTN-QPTUXGOLSA-N phenomorphan Chemical compound C([C@]12CCCC[C@H]1[C@H]1CC3=CC=C(C=C32)O)CN1CCC1=CC=CC=C1 CFBQYWXPZVQQTN-QPTUXGOLSA-N 0.000 description 2
- 229950011496 phenomorphan Drugs 0.000 description 2
- IPOPQVVNCFQFRK-UHFFFAOYSA-N phenoperidine Chemical compound C1CC(C(=O)OCC)(C=2C=CC=CC=2)CCN1CCC(O)C1=CC=CC=C1 IPOPQVVNCFQFRK-UHFFFAOYSA-N 0.000 description 2
- 229960004315 phenoperidine Drugs 0.000 description 2
- PXXKIYPSXYFATG-UHFFFAOYSA-N piminodine Chemical compound C1CC(C(=O)OCC)(C=2C=CC=CC=2)CCN1CCCNC1=CC=CC=C1 PXXKIYPSXYFATG-UHFFFAOYSA-N 0.000 description 2
- 229950006445 piminodine Drugs 0.000 description 2
- 229940068170 pirinitramide Drugs 0.000 description 2
- IHEHEFLXQFOQJO-UHFFFAOYSA-N piritramide Chemical compound C1CC(C(=O)N)(N2CCCCC2)CCN1CCC(C#N)(C=1C=CC=CC=1)C1=CC=CC=C1 IHEHEFLXQFOQJO-UHFFFAOYSA-N 0.000 description 2
- 210000002442 prefrontal cortex Anatomy 0.000 description 2
- 230000003518 presynaptic effect Effects 0.000 description 2
- 230000008569 process Effects 0.000 description 2
- 108010041071 proenkephalin Proteins 0.000 description 2
- ZXWAUWBYASJEOE-UHFFFAOYSA-N proheptazine Chemical compound C=1C=CC=CC=1C1(OC(=O)CC)CCCN(C)CC1C ZXWAUWBYASJEOE-UHFFFAOYSA-N 0.000 description 2
- 239000003380 propellant Substances 0.000 description 2
- XJKQCILVUHXVIQ-UHFFFAOYSA-N properidine Chemical compound C=1C=CC=CC=1C1(C(=O)OC(C)C)CCN(C)CC1 XJKQCILVUHXVIQ-UHFFFAOYSA-N 0.000 description 2
- 229950004345 properidine Drugs 0.000 description 2
- ZBAFFZBKCMWUHM-UHFFFAOYSA-N propiram Chemical compound C=1C=CC=NC=1N(C(=O)CC)C(C)CN1CCCCC1 ZBAFFZBKCMWUHM-UHFFFAOYSA-N 0.000 description 2
- 229950003779 propiram Drugs 0.000 description 2
- QELSKZZBTMNZEB-UHFFFAOYSA-N propylparaben Chemical compound CCCOC(=O)C1=CC=C(O)C=C1 QELSKZZBTMNZEB-UHFFFAOYSA-N 0.000 description 2
- AJPJDKMHJJGVTQ-UHFFFAOYSA-M sodium dihydrogen phosphate Chemical compound [Na+].OP(O)([O-])=O AJPJDKMHJJGVTQ-UHFFFAOYSA-M 0.000 description 2
- 238000013222 sprague-dawley male rat Methods 0.000 description 2
- 201000009032 substance abuse Diseases 0.000 description 2
- 239000000758 substrate Substances 0.000 description 2
- GGCSSNBKKAUURC-UHFFFAOYSA-N sufentanil Chemical compound C1CN(CCC=2SC=CC=2)CCC1(COC)N(C(=O)CC)C1=CC=CC=C1 GGCSSNBKKAUURC-UHFFFAOYSA-N 0.000 description 2
- 229960004739 sufentanil Drugs 0.000 description 2
- 229960001402 tilidine Drugs 0.000 description 2
- 229960004380 tramadol Drugs 0.000 description 2
- TVYLLZQTGLZFBW-GOEBONIOSA-N tramadol Natural products COC1=CC=CC([C@@]2(O)[C@@H](CCCC2)CN(C)C)=C1 TVYLLZQTGLZFBW-GOEBONIOSA-N 0.000 description 2
- LLPOLZWFYMWNKH-UHFFFAOYSA-N trans-dihydrocodeinone Natural products C1C(N(CCC234)C)C2CCC(=O)C3OC2=C4C1=CC=C2OC LLPOLZWFYMWNKH-UHFFFAOYSA-N 0.000 description 2
- RJMIEHBSYVWVIN-LLVKDONJSA-N (2r)-2-[4-(3-oxo-1h-isoindol-2-yl)phenyl]propanoic acid Chemical compound C1=CC([C@H](C(O)=O)C)=CC=C1N1C(=O)C2=CC=CC=C2C1 RJMIEHBSYVWVIN-LLVKDONJSA-N 0.000 description 1
- LNAZSHAWQACDHT-XIYTZBAFSA-N (2r,3r,4s,5r,6s)-4,5-dimethoxy-2-(methoxymethyl)-3-[(2s,3r,4s,5r,6r)-3,4,5-trimethoxy-6-(methoxymethyl)oxan-2-yl]oxy-6-[(2r,3r,4s,5r,6r)-4,5,6-trimethoxy-2-(methoxymethyl)oxan-3-yl]oxyoxane Chemical compound CO[C@@H]1[C@@H](OC)[C@H](OC)[C@@H](COC)O[C@H]1O[C@H]1[C@H](OC)[C@@H](OC)[C@H](O[C@H]2[C@@H]([C@@H](OC)[C@H](OC)O[C@@H]2COC)OC)O[C@@H]1COC LNAZSHAWQACDHT-XIYTZBAFSA-N 0.000 description 1
- RDJGLLICXDHJDY-NSHDSACASA-N (2s)-2-(3-phenoxyphenyl)propanoic acid Chemical compound OC(=O)[C@@H](C)C1=CC=CC(OC=2C=CC=CC=2)=C1 RDJGLLICXDHJDY-NSHDSACASA-N 0.000 description 1
- GUHPRPJDBZHYCJ-SECBINFHSA-N (2s)-2-(5-benzoylthiophen-2-yl)propanoic acid Chemical compound S1C([C@H](C(O)=O)C)=CC=C1C(=O)C1=CC=CC=C1 GUHPRPJDBZHYCJ-SECBINFHSA-N 0.000 description 1
- MDKGKXOCJGEUJW-VIFPVBQESA-N (2s)-2-[4-(thiophene-2-carbonyl)phenyl]propanoic acid Chemical compound C1=CC([C@@H](C(O)=O)C)=CC=C1C(=O)C1=CC=CS1 MDKGKXOCJGEUJW-VIFPVBQESA-N 0.000 description 1
- AGTSSZRZBSNTGQ-ITZCFHCWSA-N (2s,3r)-2-[[(2s)-2-[[(2s)-2-[[(2s)-6-amino-2-[[(2s)-2-[[(2s)-5-amino-2-[[(2s)-2-[[(2s)-2-[[(2s)-2-[[(2s)-2-[[2-[[2-[[(2s)-2-amino-3-(4-hydroxyphenyl)propanoyl]amino]acetyl]amino]acetyl]amino]-3-phenylpropanoyl]amino]-4-methylpentanoyl]amino]-5-(diaminomet Chemical compound C([C@@H](C(=O)N[C@@H](CC(C)C)C(=O)N[C@@H](CCCN=C(N)N)C(=O)N[C@@H](CCCN=C(N)N)C(=O)N[C@@H](CCC(N)=O)C(=O)N[C@@H](CC=1C=CC=CC=1)C(=O)N[C@@H](CCCCN)C(=O)N[C@@H](C(C)C)C(=O)N[C@@H](C(C)C)C(=O)N[C@@H]([C@@H](C)O)C(O)=O)NC(=O)CNC(=O)CNC(=O)[C@@H](N)CC=1C=CC(O)=CC=1)C1=CC=CC=C1 AGTSSZRZBSNTGQ-ITZCFHCWSA-N 0.000 description 1
- VHBDDUMTLJSZDR-VYKNHSEDSA-N (4r,4ar,7s,7ar,12bs)-3-methyl-2,4,4a,7,7a,13-hexahydro-1h-4,12-methanobenzofuro[3,2-e]isoquinoline-7,9-diol;nitric acid Chemical compound O[N+]([O-])=O.O([C@H]1[C@H](C=C[C@H]23)O)C4=C5[C@@]12CCN(C)[C@@H]3CC5=CC=C4O VHBDDUMTLJSZDR-VYKNHSEDSA-N 0.000 description 1
- LGFMXOTUSSVQJV-NEYUFSEYSA-N (4r,4ar,7s,7ar,12bs)-9-methoxy-3-methyl-2,4,4a,7,7a,13-hexahydro-1h-4,12-methanobenzofuro[3,2-e]isoquinoline-7-ol;(4r,4ar,7s,7ar,12bs)-3-methyl-2,4,4a,7,7a,13-hexahydro-1h-4,12-methanobenzofuro[3,2-e]isoquinoline-7,9-diol;1-[(3,4-dimethoxyphenyl)methyl]-6 Chemical compound Cl.Cl.Cl.O([C@H]1[C@H](C=C[C@H]23)O)C4=C5[C@@]12CCN(C)[C@@H]3CC5=CC=C4O.C([C@H]1[C@H](N(CC[C@@]112)C)C3)=C[C@H](O)[C@@H]1OC1=C2C3=CC=C1OC.C1=C(OC)C(OC)=CC=C1CC1=NC=CC2=CC(OC)=C(OC)C=C12 LGFMXOTUSSVQJV-NEYUFSEYSA-N 0.000 description 1
- BJEPYKJPYRNKOW-REOHCLBHSA-N (S)-malic acid Chemical compound OC(=O)[C@@H](O)CC(O)=O BJEPYKJPYRNKOW-REOHCLBHSA-N 0.000 description 1
- ODIGIKRIUKFKHP-UHFFFAOYSA-N (n-propan-2-yloxycarbonylanilino) acetate Chemical compound CC(C)OC(=O)N(OC(C)=O)C1=CC=CC=C1 ODIGIKRIUKFKHP-UHFFFAOYSA-N 0.000 description 1
- LVGUZGTVOIAKKC-UHFFFAOYSA-N 1,1,1,2-tetrafluoroethane Chemical compound FCC(F)(F)F LVGUZGTVOIAKKC-UHFFFAOYSA-N 0.000 description 1
- DDMOUSALMHHKOS-UHFFFAOYSA-N 1,2-dichloro-1,1,2,2-tetrafluoroethane Chemical compound FC(F)(Cl)C(F)(F)Cl DDMOUSALMHHKOS-UHFFFAOYSA-N 0.000 description 1
- IXPNQXFRVYWDDI-UHFFFAOYSA-N 1-methyl-2,4-dioxo-1,3-diazinane-5-carboximidamide Chemical compound CN1CC(C(N)=N)C(=O)NC1=O IXPNQXFRVYWDDI-UHFFFAOYSA-N 0.000 description 1
- IIZPXYDJLKNOIY-JXPKJXOSSA-N 1-palmitoyl-2-arachidonoyl-sn-glycero-3-phosphocholine Chemical compound CCCCCCCCCCCCCCCC(=O)OC[C@H](COP([O-])(=O)OCC[N+](C)(C)C)OC(=O)CCC\C=C/C\C=C/C\C=C/C\C=C/CCCCC IIZPXYDJLKNOIY-JXPKJXOSSA-N 0.000 description 1
- KLIVRBFRQSOGQI-UHFFFAOYSA-N 2-(11-oxo-6h-benzo[c][1]benzothiepin-3-yl)acetic acid Chemical compound S1CC2=CC=CC=C2C(=O)C2=CC=C(CC(=O)O)C=C12 KLIVRBFRQSOGQI-UHFFFAOYSA-N 0.000 description 1
- DCXHLPGLBYHNMU-UHFFFAOYSA-N 2-[1-(4-azidobenzoyl)-5-methoxy-2-methylindol-3-yl]acetic acid Chemical compound CC1=C(CC(O)=O)C2=CC(OC)=CC=C2N1C(=O)C1=CC=C(N=[N+]=[N-])C=C1 DCXHLPGLBYHNMU-UHFFFAOYSA-N 0.000 description 1
- TYCOFFBAZNSQOJ-UHFFFAOYSA-N 2-[4-(3-fluorophenyl)phenyl]propanoic acid Chemical compound C1=CC(C(C(O)=O)C)=CC=C1C1=CC=CC(F)=C1 TYCOFFBAZNSQOJ-UHFFFAOYSA-N 0.000 description 1
- JIEKMACRVQTPRC-UHFFFAOYSA-N 2-[4-(4-chlorophenyl)-2-phenyl-5-thiazolyl]acetic acid Chemical compound OC(=O)CC=1SC(C=2C=CC=CC=2)=NC=1C1=CC=C(Cl)C=C1 JIEKMACRVQTPRC-UHFFFAOYSA-N 0.000 description 1
- XKSAJZSJKURQRX-UHFFFAOYSA-N 2-acetyloxy-5-(4-fluorophenyl)benzoic acid Chemical compound C1=C(C(O)=O)C(OC(=O)C)=CC=C1C1=CC=C(F)C=C1 XKSAJZSJKURQRX-UHFFFAOYSA-N 0.000 description 1
- BMYNFMYTOJXKLE-UHFFFAOYSA-N 3-azaniumyl-2-hydroxypropanoate Chemical compound NCC(O)C(O)=O BMYNFMYTOJXKLE-UHFFFAOYSA-N 0.000 description 1
- 239000002999 4 aminobutyrate aminotransferase inhibitor Substances 0.000 description 1
- SYCHUQUJURZQMO-UHFFFAOYSA-N 4-hydroxy-2-methyl-1,1-dioxo-n-(1,3-thiazol-2-yl)-1$l^{6},2-benzothiazine-3-carboxamide Chemical compound OC=1C2=CC=CC=C2S(=O)(=O)N(C)C=1C(=O)NC1=NC=CS1 SYCHUQUJURZQMO-UHFFFAOYSA-N 0.000 description 1
- PJJGZPJJTHBVMX-UHFFFAOYSA-N 5,7-Dihydroxyisoflavone Chemical compound C=1C(O)=CC(O)=C(C2=O)C=1OC=C2C1=CC=CC=C1 PJJGZPJJTHBVMX-UHFFFAOYSA-N 0.000 description 1
- RZVAJINKPMORJF-UHFFFAOYSA-N Acetaminophen Chemical compound CC(=O)NC1=CC=C(O)C=C1 RZVAJINKPMORJF-UHFFFAOYSA-N 0.000 description 1
- 239000005995 Aluminium silicate Substances 0.000 description 1
- 206010002091 Anaesthesia Diseases 0.000 description 1
- 208000007415 Anhedonia Diseases 0.000 description 1
- BSYNRYMUTXBXSQ-UHFFFAOYSA-N Aspirin Chemical compound CC(=O)OC1=CC=CC=C1C(O)=O BSYNRYMUTXBXSQ-UHFFFAOYSA-N 0.000 description 1
- 208000008035 Back Pain Diseases 0.000 description 1
- KPYSYYIEGFHWSV-UHFFFAOYSA-N Baclofen Chemical compound OC(=O)CC(CN)C1=CC=C(Cl)C=C1 KPYSYYIEGFHWSV-UHFFFAOYSA-N 0.000 description 1
- 239000005711 Benzoic acid Substances 0.000 description 1
- 101800005049 Beta-endorphin Proteins 0.000 description 1
- 102400000748 Beta-endorphin Human genes 0.000 description 1
- LSNNMFCWUKXFEE-UHFFFAOYSA-M Bisulfite Chemical compound OS([O-])=O LSNNMFCWUKXFEE-UHFFFAOYSA-M 0.000 description 1
- 241000283690 Bos taurus Species 0.000 description 1
- OYPRJOBELJOOCE-UHFFFAOYSA-N Calcium Chemical compound [Ca] OYPRJOBELJOOCE-UHFFFAOYSA-N 0.000 description 1
- 241000282472 Canis lupus familiaris Species 0.000 description 1
- OIRAEJWYWSAQNG-UHFFFAOYSA-N Clidanac Chemical compound ClC=1C=C2C(C(=O)O)CCC2=CC=1C1CCCCC1 OIRAEJWYWSAQNG-UHFFFAOYSA-N 0.000 description 1
- UDIPTWFVPPPURJ-UHFFFAOYSA-M Cyclamate Chemical compound [Na+].[O-]S(=O)(=O)NC1CCCCC1 UDIPTWFVPPPURJ-UHFFFAOYSA-M 0.000 description 1
- RGHNJXZEOKUKBD-UHFFFAOYSA-N D-gluconic acid Natural products OCC(O)C(O)C(O)C(O)C(O)=O RGHNJXZEOKUKBD-UHFFFAOYSA-N 0.000 description 1
- FEWJPZIEWOKRBE-JCYAYHJZSA-N Dextrotartaric acid Chemical compound OC(=O)[C@H](O)[C@@H](O)C(O)=O FEWJPZIEWOKRBE-JCYAYHJZSA-N 0.000 description 1
- 239000004338 Dichlorodifluoromethane Substances 0.000 description 1
- 206010013654 Drug abuse Diseases 0.000 description 1
- 102400000242 Dynorphin A(1-17) Human genes 0.000 description 1
- 108010049140 Endorphins Proteins 0.000 description 1
- 102000009025 Endorphins Human genes 0.000 description 1
- 108090000790 Enzymes Proteins 0.000 description 1
- 102000004190 Enzymes Human genes 0.000 description 1
- 241000283073 Equus caballus Species 0.000 description 1
- OGDVEMNWJVYAJL-UHFFFAOYSA-N Ethylmorphine Natural products C1C(N(CCC234)C)C2C=CC(O)C3OC2=C4C1=CC=C2OCC OGDVEMNWJVYAJL-UHFFFAOYSA-N 0.000 description 1
- 241000282326 Felis catus Species 0.000 description 1
- 102000005915 GABA Receptors Human genes 0.000 description 1
- 108010005551 GABA Receptors Proteins 0.000 description 1
- 240000004859 Gamochaeta purpurea Species 0.000 description 1
- 108010010803 Gelatin Proteins 0.000 description 1
- HEFNNWSXXWATRW-UHFFFAOYSA-N Ibuprofen Chemical compound CC(C)CC1=CC=C(C(C)C(O)=O)C=C1 HEFNNWSXXWATRW-UHFFFAOYSA-N 0.000 description 1
- RGHNJXZEOKUKBD-QTBDOELSSA-N L-gulonic acid Chemical compound OC[C@H](O)[C@@H](O)[C@H](O)[C@H](O)C(O)=O RGHNJXZEOKUKBD-QTBDOELSSA-N 0.000 description 1
- GUBGYTABKSRVRQ-QKKXKWKRSA-N Lactose Natural products OC[C@H]1O[C@@H](O[C@H]2[C@H](O)[C@@H](O)C(O)O[C@@H]2CO)[C@H](O)[C@@H](O)[C@H]1O GUBGYTABKSRVRQ-QKKXKWKRSA-N 0.000 description 1
- URLZCHNOLZSCCA-VABKMULXSA-N Leu-enkephalin Chemical class C([C@@H](C(=O)N[C@@H](CC(C)C)C(O)=O)NC(=O)CNC(=O)CNC(=O)[C@@H](N)CC=1C=CC(O)=CC=1)C1=CC=CC=C1 URLZCHNOLZSCCA-VABKMULXSA-N 0.000 description 1
- SBDNJUWAMKYJOX-UHFFFAOYSA-N Meclofenamic Acid Chemical compound CC1=CC=C(Cl)C(NC=2C(=CC=CC=2)C(O)=O)=C1Cl SBDNJUWAMKYJOX-UHFFFAOYSA-N 0.000 description 1
- UEZVMMHDMIWARA-UHFFFAOYSA-N Metaphosphoric acid Chemical compound OP(=O)=O UEZVMMHDMIWARA-UHFFFAOYSA-N 0.000 description 1
- 241000699670 Mus sp. Species 0.000 description 1
- CMWTZPSULFXXJA-UHFFFAOYSA-N Naproxen Natural products C1=C(C(C)C(O)=O)C=CC2=CC(OC)=CC=C21 CMWTZPSULFXXJA-UHFFFAOYSA-N 0.000 description 1
- JZFPYUNJRRFVQU-UHFFFAOYSA-N Niflumic acid Chemical compound OC(=O)C1=CC=CN=C1NC1=CC=CC(C(F)(F)F)=C1 JZFPYUNJRRFVQU-UHFFFAOYSA-N 0.000 description 1
- GRYLNZFGIOXLOG-UHFFFAOYSA-N Nitric acid Chemical compound O[N+]([O-])=O GRYLNZFGIOXLOG-UHFFFAOYSA-N 0.000 description 1
- 241001504519 Papio ursinus Species 0.000 description 1
- 241001494479 Pecora Species 0.000 description 1
- 229920005372 Plexiglas® Polymers 0.000 description 1
- TVQZAMVBTVNYLA-UHFFFAOYSA-N Pranoprofen Chemical compound C1=CC=C2CC3=CC(C(C(O)=O)C)=CC=C3OC2=N1 TVQZAMVBTVNYLA-UHFFFAOYSA-N 0.000 description 1
- 241000288906 Primates Species 0.000 description 1
- 108010069820 Pro-Opiomelanocortin Proteins 0.000 description 1
- 239000000683 Pro-Opiomelanocortin Substances 0.000 description 1
- 241000700157 Rattus norvegicus Species 0.000 description 1
- 101800001440 Rimorphin Proteins 0.000 description 1
- 102400000235 Rimorphin Human genes 0.000 description 1
- 241000283984 Rodentia Species 0.000 description 1
- 208000008765 Sciatica Diseases 0.000 description 1
- 229920002472 Starch Polymers 0.000 description 1
- 229910000831 Steel Inorganic materials 0.000 description 1
- KDYFGRWQOYBRFD-UHFFFAOYSA-N Succinic acid Natural products OC(=O)CCC(O)=O KDYFGRWQOYBRFD-UHFFFAOYSA-N 0.000 description 1
- 229930006000 Sucrose Natural products 0.000 description 1
- CZMRCDWAGMRECN-UGDNZRGBSA-N Sucrose Chemical compound O[C@H]1[C@H](O)[C@@H](CO)O[C@@]1(CO)O[C@@H]1[C@H](O)[C@@H](O)[C@H](O)[C@@H](CO)O1 CZMRCDWAGMRECN-UGDNZRGBSA-N 0.000 description 1
- QAOWNCQODCNURD-UHFFFAOYSA-N Sulfuric acid Chemical compound OS(O)(=O)=O QAOWNCQODCNURD-UHFFFAOYSA-N 0.000 description 1
- FEWJPZIEWOKRBE-UHFFFAOYSA-N Tartaric acid Natural products [H+].[H+].[O-]C(=O)C(O)C(O)C([O-])=O FEWJPZIEWOKRBE-UHFFFAOYSA-N 0.000 description 1
- UWAOJIWUVCMBAZ-UHFFFAOYSA-N [1-[1-(4-chlorophenyl)cyclobutyl]-3-methylbutyl]-dimethylazanium;chloride Chemical compound Cl.C=1C=C(Cl)C=CC=1C1(C(N(C)C)CC(C)C)CCC1 UWAOJIWUVCMBAZ-UHFFFAOYSA-N 0.000 description 1
- 210000000683 abdominal cavity Anatomy 0.000 description 1
- 229960004892 acemetacin Drugs 0.000 description 1
- FSQKKOOTNAMONP-UHFFFAOYSA-N acemetacin Chemical compound CC1=C(CC(=O)OCC(O)=O)C2=CC(OC)=CC=C2N1C(=O)C1=CC=C(Cl)C=C1 FSQKKOOTNAMONP-UHFFFAOYSA-N 0.000 description 1
- 235000011054 acetic acid Nutrition 0.000 description 1
- DPXJVFZANSGRMM-UHFFFAOYSA-N acetic acid;2,3,4,5,6-pentahydroxyhexanal;sodium Chemical compound [Na].CC(O)=O.OCC(O)C(O)C(O)C(O)C=O DPXJVFZANSGRMM-UHFFFAOYSA-N 0.000 description 1
- 229960001138 acetylsalicylic acid Drugs 0.000 description 1
- 239000002253 acid Substances 0.000 description 1
- 239000012190 activator Substances 0.000 description 1
- 230000003044 adaptive effect Effects 0.000 description 1
- 239000000654 additive Substances 0.000 description 1
- 230000000996 additive effect Effects 0.000 description 1
- 239000000853 adhesive Substances 0.000 description 1
- 230000001070 adhesive effect Effects 0.000 description 1
- 108010041395 alpha-Endorphin Proteins 0.000 description 1
- BJEPYKJPYRNKOW-UHFFFAOYSA-N alpha-hydroxysuccinic acid Natural products OC(=O)C(O)CC(O)=O BJEPYKJPYRNKOW-UHFFFAOYSA-N 0.000 description 1
- 229910000147 aluminium phosphate Inorganic materials 0.000 description 1
- 235000012211 aluminium silicate Nutrition 0.000 description 1
- 230000037005 anaesthesia Effects 0.000 description 1
- 230000002421 anti-septic effect Effects 0.000 description 1
- 125000003118 aryl group Chemical group 0.000 description 1
- 239000012752 auxiliary agent Substances 0.000 description 1
- 229960000794 baclofen Drugs 0.000 description 1
- 229960005430 benoxaprofen Drugs 0.000 description 1
- 235000010233 benzoic acid Nutrition 0.000 description 1
- WOPZMFQRCBYPJU-NTXHZHDSSA-N beta-endorphin Chemical compound C([C@@H](C(=O)N[C@@H](CCCCN)C(=O)N[C@@H](CC(N)=O)C(=O)N[C@@H](C)C(=O)N[C@@H]([C@@H](C)CC)C(=O)N[C@@H]([C@@H](C)CC)C(=O)N[C@@H](CCCCN)C(=O)N[C@@H](CC(N)=O)C(=O)N[C@@H](C)C(=O)N[C@@H](CC=1C=CC(O)=CC=1)C(=O)N[C@@H](CCCCN)C(=O)N[C@@H](CCCCN)C(=O)NCC(=O)N[C@@H](CCC(N)=O)C(O)=O)NC(=O)[C@H](CC(C)C)NC(=O)[C@@H](NC(=O)[C@@H](NC(=O)[C@H](CC(C)C)NC(=O)[C@H]1N(CCC1)C(=O)[C@@H](NC(=O)[C@H](CCC(N)=O)NC(=O)[C@H](CO)NC(=O)[C@H](CCCCN)NC(=O)[C@H](CCC(N)=O)NC(=O)[C@H](CO)NC(=O)[C@@H](NC(=O)[C@H](CCSC)NC(=O)[C@H](CC=1C=CC=CC=1)NC(=O)CNC(=O)CNC(=O)[C@@H](N)CC=1C=CC(O)=CC=1)[C@@H](C)O)[C@@H](C)O)C(C)C)[C@@H](C)O)C1=CC=CC=C1 WOPZMFQRCBYPJU-NTXHZHDSSA-N 0.000 description 1
- 239000011230 binding agent Substances 0.000 description 1
- 238000011953 bioanalysis Methods 0.000 description 1
- 230000036983 biotransformation Effects 0.000 description 1
- 230000008499 blood brain barrier function Effects 0.000 description 1
- 210000001218 blood-brain barrier Anatomy 0.000 description 1
- 239000012267 brine Substances 0.000 description 1
- 150000007516 brønsted-lowry acids Chemical class 0.000 description 1
- 150000007528 brønsted-lowry bases Chemical class 0.000 description 1
- 229950005608 bucloxic acid Drugs 0.000 description 1
- IJTPQQVCKPZIMV-UHFFFAOYSA-N bucloxic acid Chemical compound ClC1=CC(C(=O)CCC(=O)O)=CC=C1C1CCCCC1 IJTPQQVCKPZIMV-UHFFFAOYSA-N 0.000 description 1
- 244000309464 bull Species 0.000 description 1
- KDYFGRWQOYBRFD-NUQCWPJISA-N butanedioic acid Chemical compound O[14C](=O)CC[14C](O)=O KDYFGRWQOYBRFD-NUQCWPJISA-N 0.000 description 1
- 229910052791 calcium Inorganic materials 0.000 description 1
- 229910000019 calcium carbonate Inorganic materials 0.000 description 1
- 235000010216 calcium carbonate Nutrition 0.000 description 1
- 239000001506 calcium phosphate Substances 0.000 description 1
- 229910000389 calcium phosphate Inorganic materials 0.000 description 1
- 235000011010 calcium phosphates Nutrition 0.000 description 1
- 125000004432 carbon atom Chemical group C* 0.000 description 1
- 239000001569 carbon dioxide Substances 0.000 description 1
- 229910002092 carbon dioxide Inorganic materials 0.000 description 1
- 239000001768 carboxy methyl cellulose Substances 0.000 description 1
- 229960003184 carprofen Drugs 0.000 description 1
- IVUMCTKHWDRRMH-UHFFFAOYSA-N carprofen Chemical compound C1=CC(Cl)=C[C]2C3=CC=C(C(C(O)=O)C)C=C3N=C21 IVUMCTKHWDRRMH-UHFFFAOYSA-N 0.000 description 1
- 239000000969 carrier Substances 0.000 description 1
- 230000001925 catabolic effect Effects 0.000 description 1
- 229940112822 chewing gum Drugs 0.000 description 1
- 235000015218 chewing gum Nutrition 0.000 description 1
- 238000004587 chromatography analysis Methods 0.000 description 1
- 208000022371 chronic pain syndrome Diseases 0.000 description 1
- 235000015165 citric acid Nutrition 0.000 description 1
- 229950010886 clidanac Drugs 0.000 description 1
- 238000007596 consolidation process Methods 0.000 description 1
- 238000013270 controlled release Methods 0.000 description 1
- 238000007796 conventional method Methods 0.000 description 1
- 238000002425 crystallisation Methods 0.000 description 1
- 230000008025 crystallization Effects 0.000 description 1
- 239000000625 cyclamic acid and its Na and Ca salt Substances 0.000 description 1
- 230000001934 delay Effects 0.000 description 1
- 230000002939 deleterious effect Effects 0.000 description 1
- 238000013461 design Methods 0.000 description 1
- PXBRQCKWGAHEHS-UHFFFAOYSA-N dichlorodifluoromethane Chemical compound FC(F)(Cl)Cl PXBRQCKWGAHEHS-UHFFFAOYSA-N 0.000 description 1
- 235000019404 dichlorodifluoromethane Nutrition 0.000 description 1
- 229940087091 dichlorotetrafluoroethane Drugs 0.000 description 1
- 229960001259 diclofenac Drugs 0.000 description 1
- DCOPUUMXTXDBNB-UHFFFAOYSA-N diclofenac Chemical compound OC(=O)CC1=CC=CC=C1NC1=C(Cl)C=CC=C1Cl DCOPUUMXTXDBNB-UHFFFAOYSA-N 0.000 description 1
- 239000002270 dispersing agent Substances 0.000 description 1
- 210000005064 dopaminergic neuron Anatomy 0.000 description 1
- 230000003291 dopaminomimetic effect Effects 0.000 description 1
- 239000002552 dosage form Substances 0.000 description 1
- 238000012377 drug delivery Methods 0.000 description 1
- 230000000857 drug effect Effects 0.000 description 1
- JMNJYGMAUMANNW-FIXZTSJVSA-N dynorphin a Chemical compound C([C@@H](C(=O)N[C@@H](CC(C)C)C(=O)N[C@@H](CCCNC(N)=N)C(=O)N[C@@H](CCCNC(N)=N)C(=O)N[C@@H]([C@@H](C)CC)C(=O)N[C@@H](CCCNC(N)=N)C(=O)N1[C@@H](CCC1)C(=O)N[C@@H](CCCCN)C(=O)N[C@@H](CC(C)C)C(=O)N[C@@H](CCCCN)C(=O)N[C@@H](CC=1C2=CC=CC=C2NC=1)C(=O)N[C@@H](CC(O)=O)C(=O)N[C@@H](CC(N)=O)C(=O)N[C@@H](CCC(N)=O)C(O)=O)NC(=O)CNC(=O)CNC(=O)[C@@H](N)CC=1C=CC(O)=CC=1)C1=CC=CC=C1 JMNJYGMAUMANNW-FIXZTSJVSA-N 0.000 description 1
- 230000005611 electricity Effects 0.000 description 1
- 230000008030 elimination Effects 0.000 description 1
- 238000003379 elimination reaction Methods 0.000 description 1
- 238000005516 engineering process Methods 0.000 description 1
- 230000002708 enhancing effect Effects 0.000 description 1
- 230000007613 environmental effect Effects 0.000 description 1
- 239000002532 enzyme inhibitor Substances 0.000 description 1
- 229940125532 enzyme inhibitor Drugs 0.000 description 1
- 125000001495 ethyl group Chemical group [H]C([H])([H])C([H])([H])* 0.000 description 1
- 229960004578 ethylmorphine Drugs 0.000 description 1
- 238000011156 evaluation Methods 0.000 description 1
- 238000002474 experimental method Methods 0.000 description 1
- 229960001419 fenoprofen Drugs 0.000 description 1
- 229960002679 fentiazac Drugs 0.000 description 1
- 229960004369 flufenamic acid Drugs 0.000 description 1
- LPEPZBJOKDYZAD-UHFFFAOYSA-N flufenamic acid Chemical compound OC(=O)C1=CC=CC=C1NC1=CC=CC(C(F)(F)F)=C1 LPEPZBJOKDYZAD-UHFFFAOYSA-N 0.000 description 1
- 229950007979 flufenisal Drugs 0.000 description 1
- 239000012530 fluid Substances 0.000 description 1
- 229950001284 fluprofen Drugs 0.000 description 1
- 229960002390 flurbiprofen Drugs 0.000 description 1
- SYTBZMRGLBWNTM-UHFFFAOYSA-N flurbiprofen Chemical compound FC1=CC(C(C(O)=O)C)=CC=C1C1=CC=CC=C1 SYTBZMRGLBWNTM-UHFFFAOYSA-N 0.000 description 1
- 238000009472 formulation Methods 0.000 description 1
- 238000004508 fractional distillation Methods 0.000 description 1
- 108010047064 gamma-Endorphin Proteins 0.000 description 1
- 229920000159 gelatin Polymers 0.000 description 1
- 239000008273 gelatin Substances 0.000 description 1
- 235000019322 gelatine Nutrition 0.000 description 1
- 235000011852 gelatine desserts Nutrition 0.000 description 1
- 229910021397 glassy carbon Inorganic materials 0.000 description 1
- 239000000174 gluconic acid Substances 0.000 description 1
- 235000012208 gluconic acid Nutrition 0.000 description 1
- 239000007902 hard capsule Substances 0.000 description 1
- UKACHOXRXFQJFN-UHFFFAOYSA-N heptafluoropropane Chemical compound FC(F)C(F)(F)C(F)(F)F UKACHOXRXFQJFN-UHFFFAOYSA-N 0.000 description 1
- BXWNKGSJHAJOGX-UHFFFAOYSA-N hexadecan-1-ol Chemical compound CCCCCCCCCCCCCCCCO BXWNKGSJHAJOGX-UHFFFAOYSA-N 0.000 description 1
- FBPFZTCFMRRESA-UHFFFAOYSA-N hexane-1,2,3,4,5,6-hexol Chemical compound OCC(O)C(O)C(O)C(O)CO FBPFZTCFMRRESA-UHFFFAOYSA-N 0.000 description 1
- 238000004128 high performance liquid chromatography Methods 0.000 description 1
- XMBWDFGMSWQBCA-UHFFFAOYSA-N hydrogen iodide Chemical compound I XMBWDFGMSWQBCA-UHFFFAOYSA-N 0.000 description 1
- 229940071870 hydroiodic acid Drugs 0.000 description 1
- 229960000905 indomethacin Drugs 0.000 description 1
- 229960004187 indoprofen Drugs 0.000 description 1
- 230000006698 induction Effects 0.000 description 1
- 238000001802 infusion Methods 0.000 description 1
- 239000003112 inhibitor Substances 0.000 description 1
- 229940102223 injectable solution Drugs 0.000 description 1
- 229940102213 injectable suspension Drugs 0.000 description 1
- 238000010255 intramuscular injection Methods 0.000 description 1
- 239000007927 intramuscular injection Substances 0.000 description 1
- 238000001990 intravenous administration Methods 0.000 description 1
- 239000007951 isotonicity adjuster Substances 0.000 description 1
- 229950002252 isoxicam Drugs 0.000 description 1
- YYUAYBYLJSNDCX-UHFFFAOYSA-N isoxicam Chemical compound OC=1C2=CC=CC=C2S(=O)(=O)N(C)C=1C(=O)NC=1C=C(C)ON=1 YYUAYBYLJSNDCX-UHFFFAOYSA-N 0.000 description 1
- NLYAJNPCOHFWQQ-UHFFFAOYSA-N kaolin Chemical compound O.O.O=[Al]O[Si](=O)O[Si](=O)O[Al]=O NLYAJNPCOHFWQQ-UHFFFAOYSA-N 0.000 description 1
- DKYWVDODHFEZIM-UHFFFAOYSA-N ketoprofen Chemical compound OC(=O)C(C)C1=CC=CC(C(=O)C=2C=CC=CC=2)=C1 DKYWVDODHFEZIM-UHFFFAOYSA-N 0.000 description 1
- 229960000991 ketoprofen Drugs 0.000 description 1
- 239000008101 lactose Substances 0.000 description 1
- 239000000787 lecithin Substances 0.000 description 1
- 235000010445 lecithin Nutrition 0.000 description 1
- 229940067606 lecithin Drugs 0.000 description 1
- 229940057995 liquid paraffin Drugs 0.000 description 1
- 239000007937 lozenge Substances 0.000 description 1
- 239000000314 lubricant Substances 0.000 description 1
- 229950001846 mabuprofen Drugs 0.000 description 1
- JVGUNCHERKJFCM-UHFFFAOYSA-N mabuprofen Chemical compound CC(C)CC1=CC=C(C(C)C(=O)NCCO)C=C1 JVGUNCHERKJFCM-UHFFFAOYSA-N 0.000 description 1
- 206010025482 malaise Diseases 0.000 description 1
- 239000001630 malic acid Substances 0.000 description 1
- 235000011090 malic acid Nutrition 0.000 description 1
- 238000007726 management method Methods 0.000 description 1
- 238000013507 mapping Methods 0.000 description 1
- 229960003803 meclofenamic acid Drugs 0.000 description 1
- 238000002483 medication Methods 0.000 description 1
- 229960003464 mefenamic acid Drugs 0.000 description 1
- 230000004060 metabolic process Effects 0.000 description 1
- 229920000609 methyl cellulose Polymers 0.000 description 1
- 239000001923 methylcellulose Substances 0.000 description 1
- 238000000520 microinjection Methods 0.000 description 1
- 238000002156 mixing Methods 0.000 description 1
- 150000002772 monosaccharides Chemical class 0.000 description 1
- 229910000403 monosodium phosphate Inorganic materials 0.000 description 1
- 235000019799 monosodium phosphate Nutrition 0.000 description 1
- XCKKIKBIPZJUET-VYKNHSEDSA-N morphine hydrochloride Chemical compound Cl.O([C@H]1[C@H](C=C[C@H]23)O)C4=C5[C@@]12CCN(C)[C@@H]3CC5=CC=C4O XCKKIKBIPZJUET-VYKNHSEDSA-N 0.000 description 1
- 239000002756 mu opiate receptor agonist Substances 0.000 description 1
- 210000004877 mucosa Anatomy 0.000 description 1
- QCOXCILKVHKOGO-UHFFFAOYSA-N n-(2-nitramidoethyl)nitramide Chemical compound [O-][N+](=O)NCCN[N+]([O-])=O QCOXCILKVHKOGO-UHFFFAOYSA-N 0.000 description 1
- 229960002009 naproxen Drugs 0.000 description 1
- CMWTZPSULFXXJA-VIFPVBQESA-N naproxen Chemical compound C1=C([C@H](C)C(O)=O)C=CC2=CC(OC)=CC=C21 CMWTZPSULFXXJA-VIFPVBQESA-N 0.000 description 1
- 230000001537 neural effect Effects 0.000 description 1
- 208000004296 neuralgia Diseases 0.000 description 1
- 239000002858 neurotransmitter agent Substances 0.000 description 1
- 229960000916 niflumic acid Drugs 0.000 description 1
- 229910017604 nitric acid Inorganic materials 0.000 description 1
- 229940121367 non-opioid analgesics Drugs 0.000 description 1
- 239000000041 non-steroidal anti-inflammatory agent Substances 0.000 description 1
- 229940021182 non-steroidal anti-inflammatory drug Drugs 0.000 description 1
- GYCKQBWUSACYIF-UHFFFAOYSA-N o-hydroxybenzoic acid ethyl ester Natural products CCOC(=O)C1=CC=CC=C1O GYCKQBWUSACYIF-UHFFFAOYSA-N 0.000 description 1
- 239000003921 oil Substances 0.000 description 1
- 235000019198 oils Nutrition 0.000 description 1
- 239000004006 olive oil Substances 0.000 description 1
- 235000008390 olive oil Nutrition 0.000 description 1
- 239000006186 oral dosage form Substances 0.000 description 1
- 229960002739 oxaprozin Drugs 0.000 description 1
- OFPXSFXSNFPTHF-UHFFFAOYSA-N oxaprozin Chemical compound O1C(CCC(=O)O)=NC(C=2C=CC=CC=2)=C1C1=CC=CC=C1 OFPXSFXSNFPTHF-UHFFFAOYSA-N 0.000 description 1
- 238000007254 oxidation reaction Methods 0.000 description 1
- DIVDFFZHCJEHGG-UHFFFAOYSA-N oxidopamine Chemical compound NCCC1=CC(O)=C(O)C=C1O DIVDFFZHCJEHGG-UHFFFAOYSA-N 0.000 description 1
- 229960003294 papaveretum Drugs 0.000 description 1
- 229960005489 paracetamol Drugs 0.000 description 1
- 239000008194 pharmaceutical composition Substances 0.000 description 1
- 229940124531 pharmaceutical excipient Drugs 0.000 description 1
- 150000003904 phospholipids Chemical class 0.000 description 1
- 230000035479 physiological effects, processes and functions Effects 0.000 description 1
- 239000006187 pill Substances 0.000 description 1
- 229960002702 piroxicam Drugs 0.000 description 1
- QYSPLQLAKJAUJT-UHFFFAOYSA-N piroxicam Chemical compound OC=1C2=CC=CC=C2S(=O)(=O)N(C)C=1C(=O)NC1=CC=CC=N1 QYSPLQLAKJAUJT-UHFFFAOYSA-N 0.000 description 1
- 229960000851 pirprofen Drugs 0.000 description 1
- PIDSZXPFGCURGN-UHFFFAOYSA-N pirprofen Chemical compound ClC1=CC(C(C(O)=O)C)=CC=C1N1CC=CC1 PIDSZXPFGCURGN-UHFFFAOYSA-N 0.000 description 1
- 239000004926 polymethyl methacrylate Substances 0.000 description 1
- 239000001267 polyvinylpyrrolidone Substances 0.000 description 1
- 235000013855 polyvinylpyrrolidone Nutrition 0.000 description 1
- 229920000036 polyvinylpyrrolidone Polymers 0.000 description 1
- 229960003101 pranoprofen Drugs 0.000 description 1
- 108010034422 pre-prodynorphin Proteins 0.000 description 1
- 239000002243 precursor Substances 0.000 description 1
- 108090000765 processed proteins & peptides Proteins 0.000 description 1
- 238000012545 processing Methods 0.000 description 1
- 210000004129 prosencephalon Anatomy 0.000 description 1
- 230000009467 reduction Effects 0.000 description 1
- 238000009738 saturating Methods 0.000 description 1
- 238000012216 screening Methods 0.000 description 1
- 230000001953 sensory effect Effects 0.000 description 1
- 230000000862 serotonergic effect Effects 0.000 description 1
- 229960003466 sibutramine hydrochloride Drugs 0.000 description 1
- 239000011734 sodium Substances 0.000 description 1
- 239000000661 sodium alginate Substances 0.000 description 1
- 235000010413 sodium alginate Nutrition 0.000 description 1
- 229940005550 sodium alginate Drugs 0.000 description 1
- 235000019812 sodium carboxymethyl cellulose Nutrition 0.000 description 1
- 229920001027 sodium carboxymethylcellulose Polymers 0.000 description 1
- 229960001462 sodium cyclamate Drugs 0.000 description 1
- 229940067741 sodium octyl sulfate Drugs 0.000 description 1
- 159000000000 sodium salts Chemical class 0.000 description 1
- HPALAKNZSZLMCH-UHFFFAOYSA-M sodium;chloride;hydrate Chemical compound O.[Na+].[Cl-] HPALAKNZSZLMCH-UHFFFAOYSA-M 0.000 description 1
- WFRKJMRGXGWHBM-UHFFFAOYSA-M sodium;octyl sulfate Chemical compound [Na+].CCCCCCCCOS([O-])(=O)=O WFRKJMRGXGWHBM-UHFFFAOYSA-M 0.000 description 1
- 239000007901 soft capsule Substances 0.000 description 1
- 239000002904 solvent Substances 0.000 description 1
- 125000006850 spacer group Chemical group 0.000 description 1
- 239000003381 stabilizer Substances 0.000 description 1
- 239000008107 starch Substances 0.000 description 1
- 235000019698 starch Nutrition 0.000 description 1
- 239000010959 steel Substances 0.000 description 1
- 230000004936 stimulating effect Effects 0.000 description 1
- 238000007920 subcutaneous administration Methods 0.000 description 1
- 239000005720 sucrose Substances 0.000 description 1
- 229950005175 sudoxicam Drugs 0.000 description 1
- IIACRCGMVDHOTQ-UHFFFAOYSA-M sulfamate Chemical compound NS([O-])(=O)=O IIACRCGMVDHOTQ-UHFFFAOYSA-M 0.000 description 1
- 229960000894 sulindac Drugs 0.000 description 1
- MLKXDPUZXIRXEP-MFOYZWKCSA-N sulindac Chemical compound CC1=C(CC(O)=O)C2=CC(F)=CC=C2\C1=C/C1=CC=C(S(C)=O)C=C1 MLKXDPUZXIRXEP-MFOYZWKCSA-N 0.000 description 1
- 239000001117 sulphuric acid Substances 0.000 description 1
- 235000011149 sulphuric acid Nutrition 0.000 description 1
- 239000000829 suppository Substances 0.000 description 1
- 229960004492 suprofen Drugs 0.000 description 1
- 239000000375 suspending agent Substances 0.000 description 1
- 239000000725 suspension Substances 0.000 description 1
- 230000002195 synergetic effect Effects 0.000 description 1
- 239000011975 tartaric acid Substances 0.000 description 1
- 235000002906 tartaric acid Nutrition 0.000 description 1
- 231100001274 therapeutic index Toxicity 0.000 description 1
- PBJUNZJWGZTSKL-MRXNPFEDSA-N tiagabine Chemical compound C1=CSC(C(=CCCN2C[C@@H](CCC2)C(O)=O)C2=C(C=CS2)C)=C1C PBJUNZJWGZTSKL-MRXNPFEDSA-N 0.000 description 1
- 229960001918 tiagabine Drugs 0.000 description 1
- 229960001312 tiaprofenic acid Drugs 0.000 description 1
- 229950002345 tiopinac Drugs 0.000 description 1
- 229960002905 tolfenamic acid Drugs 0.000 description 1
- YEZNLOUZAIOMLT-UHFFFAOYSA-N tolfenamic acid Chemical compound CC1=C(Cl)C=CC=C1NC1=CC=CC=C1C(O)=O YEZNLOUZAIOMLT-UHFFFAOYSA-N 0.000 description 1
- 229960001017 tolmetin Drugs 0.000 description 1
- UPSPUYADGBWSHF-UHFFFAOYSA-N tolmetin Chemical compound C1=CC(C)=CC=C1C(=O)C1=CC=C(CC(O)=O)N1C UPSPUYADGBWSHF-UHFFFAOYSA-N 0.000 description 1
- JOXIMZWYDAKGHI-UHFFFAOYSA-N toluene-4-sulfonic acid Chemical compound CC1=CC=C(S(O)(=O)=O)C=C1 JOXIMZWYDAKGHI-UHFFFAOYSA-N 0.000 description 1
- 230000001988 toxicity Effects 0.000 description 1
- 231100000419 toxicity Toxicity 0.000 description 1
- QORWJWZARLRLPR-UHFFFAOYSA-H tricalcium bis(phosphate) Chemical compound [Ca+2].[Ca+2].[Ca+2].[O-]P([O-])([O-])=O.[O-]P([O-])([O-])=O QORWJWZARLRLPR-UHFFFAOYSA-H 0.000 description 1
- CYRMSUTZVYGINF-UHFFFAOYSA-N trichlorofluoromethane Chemical compound FC(Cl)(Cl)Cl CYRMSUTZVYGINF-UHFFFAOYSA-N 0.000 description 1
- 210000003462 vein Anatomy 0.000 description 1
- 239000000080 wetting agent Substances 0.000 description 1
- 229950007802 zidometacin Drugs 0.000 description 1
- NXSIJWJXMWBCBX-NWKQFZAZSA-N α-endorphin Chemical compound C([C@@H](C(=O)N[C@@H](CCSC)C(=O)N[C@@H]([C@@H](C)O)C(=O)N[C@@H](CO)C(=O)N[C@@H](CCC(O)=O)C(=O)N[C@@H](CCCCN)C(=O)N[C@@H](CO)C(=O)N[C@@H](CCC(N)=O)C(=O)N[C@@H]([C@@H](C)O)C(=O)N1[C@@H](CCC1)C(=O)N[C@@H](CC(C)C)C(=O)N[C@@H](C(C)C)C(=O)N[C@@H]([C@@H](C)O)C(O)=O)NC(=O)CNC(=O)CNC(=O)[C@@H](N)CC=1C=CC(O)=CC=1)C1=CC=CC=C1 NXSIJWJXMWBCBX-NWKQFZAZSA-N 0.000 description 1
- GASYAMBJHBRTOE-WHDBNHDESA-N γ-endorphin Chemical compound C([C@@H](C(=O)N[C@@H](CCSC)C(=O)N[C@@H]([C@@H](C)O)C(=O)N[C@@H](CO)C(=O)N[C@@H](CCC(O)=O)C(=O)N[C@@H](CCCCN)C(=O)N[C@@H](CO)C(=O)N[C@@H](CCC(N)=O)C(=O)N[C@@H]([C@@H](C)O)C(=O)N1[C@@H](CCC1)C(=O)N[C@@H](CC(C)C)C(=O)N[C@@H](C(C)C)C(=O)N[C@@H]([C@@H](C)O)C(=O)N[C@@H](CC(C)C)C(O)=O)NC(=O)CNC(=O)CNC(=O)[C@@H](N)CC=1C=CC(O)=CC=1)C1=CC=CC=C1 GASYAMBJHBRTOE-WHDBNHDESA-N 0.000 description 1
Images
Classifications
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K31/00—Medicinal preparations containing organic active ingredients
- A61K31/185—Acids; Anhydrides, halides or salts thereof, e.g. sulfur acids, imidic, hydrazonic or hydroximic acids
- A61K31/19—Carboxylic acids, e.g. valproic acid
- A61K31/195—Carboxylic acids, e.g. valproic acid having an amino group
- A61K31/197—Carboxylic acids, e.g. valproic acid having an amino group the amino and the carboxyl groups being attached to the same acyclic carbon chain, e.g. gamma-aminobutyric acid [GABA], beta-alanine, epsilon-aminocaproic acid or pantothenic acid
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K45/00—Medicinal preparations containing active ingredients not provided for in groups A61K31/00 - A61K41/00
- A61K45/06—Mixtures of active ingredients without chemical characterisation, e.g. antiphlogistics and cardiaca
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K31/00—Medicinal preparations containing organic active ingredients
- A61K31/33—Heterocyclic compounds
- A61K31/395—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins
- A61K31/435—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins having six-membered rings with one nitrogen as the only ring hetero atom
- A61K31/47—Quinolines; Isoquinolines
- A61K31/485—Morphinan derivatives, e.g. morphine, codeine
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P25/00—Drugs for disorders of the nervous system
- A61P25/04—Centrally acting analgesics, e.g. opioids
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P25/00—Drugs for disorders of the nervous system
- A61P25/30—Drugs for disorders of the nervous system for treating abuse or dependence
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P25/00—Drugs for disorders of the nervous system
- A61P25/30—Drugs for disorders of the nervous system for treating abuse or dependence
- A61P25/36—Opioid-abuse
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P29/00—Non-central analgesic, antipyretic or antiinflammatory agents, e.g. antirheumatic agents; Non-steroidal antiinflammatory drugs [NSAID]
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P39/00—General protective or antinoxious agents
- A61P39/02—Antidotes
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P43/00—Drugs for specific purposes, not provided for in groups A61P1/00-A61P41/00
Landscapes
- Health & Medical Sciences (AREA)
- Pharmacology & Pharmacy (AREA)
- Veterinary Medicine (AREA)
- Chemical & Material Sciences (AREA)
- Public Health (AREA)
- General Health & Medical Sciences (AREA)
- Medicinal Chemistry (AREA)
- Animal Behavior & Ethology (AREA)
- Life Sciences & Earth Sciences (AREA)
- Nuclear Medicine, Radiotherapy & Molecular Imaging (AREA)
- Organic Chemistry (AREA)
- General Chemical & Material Sciences (AREA)
- Chemical Kinetics & Catalysis (AREA)
- Epidemiology (AREA)
- Engineering & Computer Science (AREA)
- Bioinformatics & Cheminformatics (AREA)
- Neurology (AREA)
- Biomedical Technology (AREA)
- Neurosurgery (AREA)
- Addiction (AREA)
- Pain & Pain Management (AREA)
- Psychiatry (AREA)
- Emergency Medicine (AREA)
- Toxicology (AREA)
- Rheumatology (AREA)
- Pharmaceuticals Containing Other Organic And Inorganic Compounds (AREA)
- Acyclic And Carbocyclic Compounds In Medicinal Compositions (AREA)
- Medicines That Contain Protein Lipid Enzymes And Other Medicines (AREA)
Abstract
本发明提供了一种用于治疗疼痛的组合物。该组合物包括药用镇痛药和GABA能药剂,如γ-乙烯基GABA,其能有效地减轻或消除该镇痛药的致瘾性。本发明还包括一种通过给予GABA能药剂来减轻或消除镇痛药的致瘾性的方法。
Description
本发明由美国政府资助,合同号为DE-AC02-98CH10886,由美国能源部签定。美国政府拥有本发明的一定权利。
本发明的背景技术
本发明涉及成瘾的预防。更具体地说,本发明涉及给予化合物来预防对经常在疼痛治疗中给予的镇痛药的成瘾。
多年以来,治疗术后疼痛和与慢性疼痛有关的疾病一直是最麻烦和困难的医学领域之一。一般说来,治疗是如此之差以致已颁布了法规来确保患者获得适当疼痛治疗的权利。
疼痛治疗的主要问题来自不能量化疼痛控制方式的适当性。临床问题是复杂的,但显然不适当的疼痛控制可导致过高的发病率和过多不良的临床结果。
很久以来已经知道准确的疼痛控制将改善临床结果并伴随很小或没有致瘾性。然而,在典型的临床情况下,几乎不可能获得准确的疼痛控制,因为疼痛的强度一般会波动并且很少随时间保持恒定。在另一方面,用超过控制疼痛所需要的麻醉剂进行治疗经常导致慢性药物成瘾和其不幸的临床和社会后果。参见B.Meier and M.Petersen,“Medicine Merchants/Uses and Abuses:Use of PainkillerGrows Quickly,Along With Wide Spread Abuse,”New York Times,2001年3月5日,在A1。
一般来说,内科医生已选择从属地治疗疼痛,因为它们真正担心以下风险:在消除需要麻醉剂治疗的内科疾病很久以后,人将对麻醉剂成瘾。
因此,需要能够给予有效的、但致瘾的镇痛药,而没有出现对这类镇痛药成瘾的有害的副作用。
发明简述
本发明提供用于治疗哺乳动物疼痛的组合物。该组合物包括具有致瘾性的药用镇痛药和有效地减轻或消除该镇痛药的致瘾性的GABA能药剂。在一个优选的具体实施例中,镇痛药是麻醉性镇痛药。
GABA能药剂可以是任何能增强GABA能系统或提高中枢神经系统中细胞外和内源性GABA水平的药剂。优选的GABA能药剂包括γ-乙烯基-γ-氨基丁酸(GVG)、加巴喷丁、丙戊酸、氟柳双胺、γ-羟基丁酸、酚加宾、十六烷基GABA、托吡酯、替加宾、以及阿坎酸(同型-钙-乙酰牛磺酸)。GABA能药剂也可包括GABA能药剂的药用盐、GABA能药剂的对映体或外消旋混合物、或上述的任何组合。GVG是最优选的。
在一个优选具体实施例中,致瘾性包括出现对镇痛药的依赖性或耐受性。
还提供一种方法用于减轻或消除镇痛药在宿主中的致瘾性。该方法包括给予宿主具有致瘾性的镇痛药,并且还给予该宿主有效地减轻或消除该镇痛药的致瘾性的GABA能药剂。优选地,GABA能药剂与镇痛药同时给予。GABA能药剂可在镇痛药之前、之后、或与镇痛药同时给予、或其任何组合。在一个优选的具体实施例中,镇痛药和GABA能药剂是以单个组合物同时给予。
在一个单独的优选具体实施例中,减轻或消除镇痛药的致瘾性是在没有对GABA能药剂的厌恶或欲望反应的条件下发生。在一个单独的优选具体实施例中,致瘾性的减轻或消除是用条件性位置偏爱(CPP)来测量。
镇痛药可以是任何镇痛药。具有致瘾性的镇痛药,例如,麻醉性镇痛药,是优选的。麻醉性镇痛药的实例包括阿芬他尼、烯丙罗定、阿法罗定、阿尼利定、苄吗啡、苯腈米特、丁丙诺啡、布托啡诺、氯尼他秦、可待因、环佐辛、地索吗啡、右吗拉胺、地佐辛、地恩丙胺、双氢可待因、双氢吗啡、地美沙朵、地美庚醇、二甲噻丁、吗苯丁酯、地匹哌酮、依他佐辛、依索庚嗪、乙甲噻丁、乙基吗啡、依托尼秦、芬太尼、海洛因、氢可酮、氢吗啡酮、羟哌替啶、异美沙酮、凯托米酮、左洛啡烷、左啡诺、左芬啡烷、洛芬太尼、哌替啶、美普他酚、美他佐辛、美沙酮、美托酮、吗啡、麦罗啡、纳布啡、那碎因、尼可吗啡、去甲左啡诺、去甲美沙酮、烯丙吗啡、去甲吗啡、诺匹哌酮、阿片、羟考酮、羟吗啡酮、阿片全碱、喷他佐辛、苯吗庚酮、非诺啡烷、非那佐辛、苯哌利定、匹米诺定、哌腈米特、propheptazine、二甲哌替啶、丙哌利定、丙吡兰、右丙氧芬、舒芬太尼、曲马朵、替利定、其盐类、任何前述的混合物、混合的μ-激动剂/拮抗药、μ-拮抗药组合。
优选的GABA能药剂包括γ-乙烯基-γ-氨基丁酸(GVG)、加巴喷丁、丙戊酸、氟柳双胺、γ-羟基丁酸、酚加宾、十六烷基GABA、托吡酯、替加宾、阿坎酸(同型-钙-乙酰牛磺酸)、其药用盐、其对映体或外消旋混合物、或其任何组合。GVG是最优选的。
在本发明的方法中,宿主通常是哺乳动物。人宿主是最优选的。
GABA能药剂的优选量将有所不同。例如,对人而言,优选地,GVG给予的量为约500mg/天至约6g/天。优选地,加巴喷丁给予人的量为约600mg/天至约3600mg/天。优选地,丙戊酸给予人的量为约500mg/天至约2500mg/天。优选地,托吡酯给予人的量为约100mg/天至约1000mg/天。优选地,氟柳双胺给予人的量为约1000mg/天至约3000mg/天。优选地,酚加宾给予人的量为约700mg/天至约4000mg/天。优选地,γ-羟基丁酸给予人的量为约1000mg/天至约5000mg/天。
本发明的组合物可减轻或消除镇痛药的致瘾性而没有干扰镇痛药的治疗效果。该组合物的这种活性具有这样的结果:通过减轻或消除作为治疗后发病主要来源的成瘾,增加了镇痛剂的治疗指数。因此,该组合物使得能够完全和持续疼痛控制,而较少担心产生治疗后药物成瘾和滥用。
附图简要描述
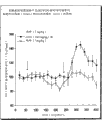
图1是曲线图,说明GVG对吗啡诱导的伏隔核(NAcc)的DA释放的影响。
发明详述
本发明的组合物包括适合用于减轻疼痛的镇痛药化合物和有效地减弱或消除镇痛药的致瘾性的GABA能药剂。通过在一个组合物中一起给予这些化合物,本发明的组合物可减轻或消除给予用来治疗疼痛的化合物的致瘾性。
在治疗术后疼痛时(外科手术和矫形术)频繁给予镇痛药。这种疼痛的特点通常在于波动但在几天至几周和几个月的时间范围内强度逐渐减弱并且需要行进性(ongoing)疼痛治疗。镇痛药也经常用于治疗慢性疼痛疾病,如慢性背痛和坐骨神经痛和其他神经痛。
镇痛药化合物可以是用于治疗疼痛的任何药用镇痛药,其在本技术领域是已知的。也可以使用镇痛药的组合。然而,当镇痛剂具有致瘾性时,该化合物的益处才获得最大实现。
具有致瘾性的镇痛药在本文定义为那些镇痛药,其被认识到在单次给予或重复给予短期或长时间后可出现(develop)躯体和/或心理依赖,和/或通过短期或长时间重复给予明显出现对其镇痛作用的耐受性。
这类镇痛药的实例包括麻醉性镇痛药。麻醉性镇痛药习惯地用于治疗疼痛。麻醉性镇痛药包括,例如,阿片类镇痛药如阿芬他尼、烯丙罗定、阿法罗定、阿尼利定、苄吗啡、苯腈米特、丁丙诺啡、布托啡诺、氯尼他秦、可待因、环佐辛、地索吗啡、右吗拉胺、地佐辛、地恩丙胺、双氢可待因、双氢吗啡、地美沙朵、地美庚醇、二甲噻丁、吗苯丁酯、地匹哌酮、依他佐辛、依索庚嗪、乙甲噻丁、乙基吗啡、依托尼秦、芬太尼、海洛因、氢可酮、氢吗啡酮、羟哌替啶、异美沙酮、凯托米酮、左洛啡烷、左啡诺、左芬啡烷、洛芬太尼、哌替啶、美普他酚、美他佐辛、美沙酮、美托酮、吗啡、麦罗啡、纳布啡、那碎因、尼可吗啡、去甲左啡诺、去甲美沙酮、烯丙吗啡、去甲吗啡、诺匹哌酮、阿片、羟考酮、羟吗啡酮、阿片全碱、喷他佐辛、苯吗庚酮、非诺啡烷、非那佐辛、苯哌利定、匹米诺定、哌腈米特、propheptazine、二甲哌替啶、丙哌利定、丙吡兰、右丙氧芬、舒芬太尼、曲马朵、替利定、其盐类、任何前述的混合物、混合的μ-激动剂/拮抗药、μ-拮抗药组合等等。
实例还包括作为内源性吗啡样物质的镇痛肽,例如,脑啡肽类如蛋氨酸脑啡肽和亮氨酸脑啡肽;内啡肽类如α-内啡肽、β-内啡肽、以及γ-内啡肽;以及强啡肽类如强啡肽A和强啡肽B,以及其前体,其实例包括前脑啡肽类如前脑啡肽、前阿黑皮素原(propiomelanocortins)、以及前强啡肽原(prodynorphins)。
镇痛药并不限于任何实物形态。镇痛药可以是,例如,固体或液体。两个液体形态镇痛药的实例是可待因糖浆和Brompton鸡尾酒(cocktail)。
本发明的组合物可进一步包括一种或多种附加药物,其可以或不能与本发明所使用的镇痛药起增效作用。这类附加药物的实例包括非甾类抗炎药,包括布洛芬、双氯芬酸、萘普生、苯噁洛芬、氟比洛芬、非诺洛芬、氟布芬、酮洛芬、吲哚洛芬、吡洛芬(piroprofen)、卡洛芬、噁丙嗪、普拉洛芬(pramoprofen)、莫罗洛芬(muroprofen)、曲美洛芬(trioxaprofen)、舒洛芬、氨布洛芬、噻洛芬酸、氟洛芬、布氯酸、吲哚美辛、舒林酸、托美丁、佐美酸、硫平酸、齐多美辛、阿西美辛、芬替酸、环氯茚酸、oxpinac、甲芬那酸、甲氧芬那酸、氟芬那酸、尼氟酸、托芬那酸、diflurisal、氟苯柳、吡罗昔康、舒多昔康或伊索昔康等等。其他适当的可包括在本发明的剂型中的附加药物包括对乙酰氨基酚、阿司匹林、以及其他非阿片类镇痛药。
GABA能药剂,如在本文所定义的,是增强GABA能系统或提高中枢神经系统(CNS)中细胞外内源性GABA水平的药剂。这类组合物或药剂包括增强在CNS中GABA产生或释放的药剂。如在本文所使用的,增强或增加内源性中枢神经系统GABA水平被定义为:在哺乳动物活体内,较之正常水平,能大幅提高或上调GABA水平。优选地,内源性中枢神经系统GABA水平可比正常水平升高至少约10%至约1000%。
GABA能药剂包括但不限于γ-乙烯基-γ-氨基丁酸(GVG)、加巴喷丁、丙戊酸、氟柳双胺、γ-羟基丁酸、酚加宾、十六烷基GABA、托吡酯、替加宾、以及阿坎酸(同型-钙-乙酰牛磺酸)。GABA能药剂还包括GABA能药剂的药用盐、以及GABA能药剂的对映体或外消旋混合物、或上述的任何组合。
不同的对映体可由手性起始材料合成,或外消旋酸盐可通过在化学上早已成熟的常规方法加以解析,如非对映盐类的手性层析、分馏结晶等等。
加巴喷丁的商品名为可自美国Parke-Davis获得。丙戊酸的商品名为和可自美国Abbott获得。氟柳双胺的商品名为可自法国Synthelabo获得。氟柳双胺的化学式为C17H16N2O2。酚加宾的商品名为SL79229,可自法国Synthelabo获得。酚加宾的化学式为C17H17C12NO。γ-羟基丁酸可自Sigma化学试剂公司获得。γ-羟基丁酸的化学式为C4H7O3Na。托吡酯是化学式为C12H21NO8S的氨基磺酸酯取代的单糖,商品名为可自美国McNeil获得。
GABA能药剂还包括组合物或药剂,其包括GABA的前体药物、或者在化学结构中以GABA作为组分的药物。在中枢神经系统中,这些前体药物经代谢、酶解、或非酶解的途径生物转化成或切割成GABA,从而具有药理活性。GABA前体药物的例子之一便是氟柳双胺,它在通过血脑屏障后,可增加内源性中枢神经系统GABA水平。
γ-乙烯基-γ-氨基丁酸(GVG)是一种选择性的和不可逆的GABA转氨酶(GABA-T)抑制剂,而后者已知可以增强GABA能的抑制。GVG为C6H11NO2或4-氨基-5-己酸,商品名为可自Hoechst Marion Roussel购得。GVG不与任何受体或再摄取复合物结合,但可通过选择性地和不可逆地抑制GABA转氨酶(GABA-T,通常对GABA进行分解代谢的酶类)的活性而提高内源性细胞内GABA水平。
如本文所使用的,GVG包括外消旋化合物或混合物,其中包括等量的S(+)-γ-乙烯基-γ-氨基丁酸、和R(-)-γ-乙烯基-γ-氨基丁酸。该GVG的外消旋化合物的商品名为由Aventis PharmaAG生产。
GVG含有不对称的碳原子,因此能够以对映体的形式存在。本发明包含GVG的任何对映体形式,包括GVG的外消旋体或外消旋混合物。在某些情况下,使用某一对映体,可能比使用其它对映体或外消旋体或外消旋混合物,在本发明的方法中具有更大的优势(即药效更好)。这种优势可由本领域熟练的技术人员很快确定。例如,对映体S(+)-γ-乙烯基-γ-氨基丁酸在提高内源性细胞内GABA水平方面比对映体R(-)-γ-乙烯基-γ-氨基丁酸更有效。
如本文所使用的,药用盐包括那些可形成盐的酸和碱,它们本身并不显著提高化合物的毒性。适宜的盐的一些例子包括无机酸盐,如盐酸、氢碘酸、氢溴酸、磷酸、偏磷酸、硝酸、和硫酸,以及有机酸盐,如酒石酸、醋酸、柠檬酸、苹果酸、苯甲酸、乙醇酸、葡萄糖酸、古洛糖酸、琥珀酸、芳基磺酸(如对甲苯磺酸)等等。
就本发明的组合物而言,设想了所有给药方式。优选系统给药方式,如口服和胃肠道外给药。本发明的组合物的给药还可包括控释递药系统,其在本技术领域是已知的。
依据已知的和建立起来的惯例,通常该组合物将用一种或多种药用组分进行配制。因而,该组合物可配制成,例如,液体、粉末、酏剂、可注射溶液、或混悬液等等。用于口服的剂型可提供成片剂、胶囊片剂、或硬胶囊剂,其中药理活性组分是与惰性固体稀释剂如碳酸钙、磷酸钙、或高岭土进行混合,或提供成软胶囊剂,其中将活性组分与含油介质(如液体石蜡或橄榄油)进行混合。
适用于胃肠道外给药的药物组合物的实例包括,例如,用于皮下、静脉、和肌内注射的注射剂、点滴注射、栓剂、吸入剂、透皮制剂、透粘膜制剂(transmucosal preparations)、以及贴剂。药理和药用添加剂的实例包括,例如,赋形剂、崩解剂或崩解助剂、粘合剂、润滑剂、涂布剂、着色剂、稀释剂、基质、增溶剂或增溶助剂、等渗剂(isotonicities)、pH调节剂、稳定剂、推进剂、以及胶粘剂。
含水混悬液可包括诸如悬浮剂这样的药用赋形剂,例如,羧甲基纤维素钠、甲基纤维素、羟丙基甲基纤维素、藻酸钠、聚乙烯吡咯烷酮、西黄蓍胶、和阿拉伯树胶;分散剂或湿润剂如天然存在的磷脂,例如卵磷脂,或环氧烷烃与脂肪酸的缩合产物如聚氧烯烃硬脂酸酯,或环氧乙烷与长链脂族醇类的缩合产物如十七乙烯-氧化十六醇(heptadecaethylene-oxycetanol),或环氧乙烷与偏酯(衍生自脂肪酸和己糖醇)的缩合产物如聚氧乙烯山梨醇含油酸基化合物(polyoxyethylene sorbitol monoleate),或环氧乙烷与偏酯(衍生自脂肪酸和己糖醇酐)的缩合产物如聚氧乙烯山梨糖醇酐含油酸基化合物。含水混悬液还可包括一种或多种防腐剂如乙基或正丙基对羟基苯甲酸酯、一种或多种着色剂、一种或多种增香剂、以及一种或多种甜味剂,如蔗糖、糖精、或环己氨基磺酸钠或环己氨基磺酸钙。
就经鼻给药而言,本发明的化合物可用作,例如,液体喷雾剂、散剂、或以滴剂的形式。对于通过吸入法给药来说,根据本发明的化合物是方便地以气溶胶喷雾剂(aerosol spray)的形式递送,其来自加压的容器(packs)或喷雾器,同时采用适当的推进剂,例如,二氯二氟甲烷、三氯氟甲烷、二氯四氟乙烷、四氟乙烷、七氟丙烷、二氧化碳、或其他适合的气体。在加压气雾剂的情况下,剂量单位的确定可通过提供阀门来递送经计量的量。可以配制用于吸入器或隔离物的,例如,明胶的胶囊剂和弹射剂,其含有本发明的化合物和适当的粉末基质(如乳糖或淀粉)的粉末混合物。
镇痛药的剂量是有效预防疼痛症状出现或治疗宿主遭受的某些疼痛症状的量,其在本领域是已知的。在组合物中镇痛药的量可适当地进行选择,其取决于,例如,给药途径、依赖性出现和/或出现耐受性的程度、给药的目的如预防疾病或治疗疾病的给药、以及患者的年龄或体重。此外,由于本发明的组合物可有效抑制或消除镇痛药的致瘾性,更高剂量的镇痛药可给予宿主。
镇痛药的“有效量”是指足以引出所希望的药理或治疗效应的量,因而导致有效预防或治疗疼痛。疼痛的预防表现为拖延或推迟疼痛的发作。疼痛的治疗表现为减少与疼痛相关的疼痛症状或改善疼痛症状的复发。在本发明的组合物中的镇痛药的特效量将会变化,其取决于(除了其它许多因素之外)要治疗的个体、疼痛的来源、和严重程度、以及给予的镇痛药的特定类型,其在本领域是已知的。对人而言,例如,有效量可从约5μg到约1000mg不等,优选从约50μg至约100mg。
在本发明的组合物中的GABA能药剂的量是有效减轻或消除镇痛药的致瘾性的量。优选地,GABA能药剂也以将在宿主中的任何潜在副作用减小到最少的量给予。有效量将有所不同,取决于所给予的镇痛药的剂量。GABA能药剂的量可由于额外的因素而变化,例如,给药途径、镇痛药的致瘾性、宿主对镇痛药的耐受性、以及年龄或体重。在优选的具体实施例中,GABA能药剂也将对镇痛药的治疗效果具有很小或没有影响。就人而言,例如,组合物中GABA能药剂的优选量是约0.5至约5克。
至于剂量的实例,其中本发明的组合物包括麻醉性镇痛剂如盐酸吗啡或硝酸吗啡且其量为从约10至30mg,本发明的组合物通常可包括约25至约1000mg、优选约100至约500mg的GABA能药剂,如GVG。
用本文所描述的镇痛药化合物进行镇痛治疗的宿主或患者一般是哺乳动物。哺乳动物包括,例如,人、狒狒及其它灵长类、以及宠物动物(如狗和猫)、实验动物(如大鼠和小鼠)、及家畜(如马、羊、母牛)等。
不为理论所限制,相信麻醉性镇痛剂的致瘾性与其对中枢神经系统(CNS)的中端脑多巴胺(DA)强化/报偿通路的药理学作用相联系。这些通路中的多巴胺能传导系统是由γ-氨基丁酸(GABA)所调节。
已经发现,致瘾性药物如在治疗疼痛中使用的吗啡和其他镇痛药物增强前脑的中端脑报偿/强化回路中的多巴胺(DA),产生增强的大脑的报偿作用,其构成药物使用者所谓的“高潮状态”。这些多巴胺系统功能的改变也已经对正在恢复的成瘾者药物渴求和用药习惯的复发中得到体现。例如,吗啡可以通过与DA载体(DAT)结合和阻碍DA向突触前末梢的再摄取而作用于这些DA系统。已有足够的证据证明,成瘾药物的致瘾性,是与中枢神经系统报偿/强化通路的再摄取过程被阻断相联系的。
人们相信,麻醉性镇痛药可抑制单胺类的突触前再摄取。中皮质边缘系DA系统中的多巴胺能神经元,其细胞体存在于腹盖区(VTA),且主要向伏隔核(NAcc)处投射,据认为该神经元参与麻醉性强化。对腹盖区(VTA)报偿中枢进行电刺激,可提高NAcc中细胞外DA水平,而NAcc中6-羟基-多巴胺的损害可消除成瘾药物的自身给药。体内微透析研究证实,吗啡可增加NAcc中细胞外的多巴胺水平的能力。
NAcc和腹侧苍白球处的γ-氨基丁酸(GABA)能神经元向腹盖区(VTA)的DA神经元投射。药理学和电生理学研究显示,这些投射是抑制性的。腹盖区DA神经元的抑制可能是GABAB受体刺激的结果。此外,向腹盖区微注射巴氯芬,通过这些受体亚型的作用,能够减少NAcc中DA的浓度。总而言之,很明显,GABA的药理学使用可以通过调节腹盖区DA神经元而影响NAcc处DA水平。
基于麻醉性镇痛药可增加细胞外NAcc中DA水平和GABA可抑制相同核中的DA,本发明人已表明,GABA能药剂可削弱麻醉性镇痛药诱导的细胞外DA的变化。例如,GVG显著削弱大鼠伏隔核(NAcc)中吗啡诱导的DA增加。参见实施例1。
因而,本发明人已开发了一种药理学策略,该策略针对GABA能神经递质系统,该系统不同于中端脑中的DA报偿/强化系统,但与其在功能上有关联,以避免在给予镇痛药来治疗疼痛时的致瘾性。然而,这一新方法没有用一种直接的GABA激动剂来针对GABA受体复合物,而是使用了GABA能药剂,利用了可提高内源性GABA水平的不可逆酶抑制剂的长效作用,而避免了GABA激动剂由于直接作用于受体本身而带来的致瘾性。因此,通过干扰产生渴求和报偿的过程而不干扰镇痛药减少宿主疼痛的能力,GABA能药剂可消除镇痛药的致瘾性。
相应地,提供了一种用于减轻或消除镇痛药的致瘾性,如上述在宿主中所定义的。该方法包括给予宿主镇痛药,并且给予有效量的GABA能药剂。
如前面所论述的,镇痛药可以是任何用于治疗疼痛的药用镇痛药,如在本领域已知的,其包括镇痛药的组合。在优选的具体实施例中,这些镇痛药具有致瘾性。这些镇痛药也不限于任何物理形式。镇痛药可以是,例如,固体或液体。两个液体形式镇痛药的实例是可待因糖浆和Brompton鸡尾酒。
如前面所论述的,具有致瘾性的镇痛药在本文定义为那些镇痛药,其被认识到在单次给予或重复给予短期或长时间后可出现依赖性,和/或通过短期或长时间重复给予显著出现对其镇痛作用的耐受性。
GABA能药剂的有效量,如在本文所定义的,是有效减轻或消除镇痛药的致瘾性的量。优选地,GABA能药剂还以将在宿主中的任何潜在副作用减小到最少的量给予。有效量将有所不同,取决于所给予的镇痛药的剂量。GABA能药剂的量可由于额外的因素而变化,例如,给药途径、镇痛药的致瘾性、宿主对镇痛药的耐受性、以及年龄或体重。在优选的具体实施例中,GABA能药剂也将对镇痛药的治疗效果具有很小或没有影响。
GABA能药剂的有效量应该足以提高内源性中枢神经系统GABA水平。如在本文所使用的,增加内源性中枢神经系统GABA水平是定义为:在哺乳动物活体内,较之正常水平,能大幅度提高或上调GABA水平。优选地,内源性中枢神经系统GABA水平可比正常水平升高至少约10%至约1000%。
在哺乳动物中有效量GVG的实例包括约10mg/kg/天至约100mg/kg/天的量,优选约25mg/kg/天至约80mg/kg/天。对人来说,较好的范围是从约500mg/天至约6g/天,更好为从1g/天至4g/天。
在哺乳动物中有效量加巴喷丁的实例包括约10mg/kg/天至约40mg/kg/天的量,优选约15mg/kg/天至约30mg/kg/天。对人来说,较好的范围是从约600mg/天至约3600mg/天,更好为从900mg/天至2400mg/天。加巴喷丁的商品名为可自美国Parke-Davis获得。
在哺乳动物中有效量的丙戊酸的实例包括约10mg/kg/天至约60mg/kg/天的量,优选约15mg/kg/天至约30mg/kg/天。对人来说,较好的范围是从约500mg/天至约2500mg/天,更好为从750mg/天至1750mg/天。丙戊酸的商品名为和可自美国Abbott获得。
在哺乳动物中有效量的托吡酯的实例包括约5mg/kg/天至约80mg/kg/天的量,优选约5mg/kg/天至约15mg/kg/天。对人来说,较好的范围是从约100mg/天至约1000mg/天,更好为从200mg/天至600mg/天。托吡酯的商品名为可自美国McNeil获得。
在哺乳动物中有效量氟柳双胺的实例包括约5mg/kg/天至约75mg/kg/天的量,优选15mg/kg/天至约45mg/kg/天。对人来说,较好的范围是从约1000mg/天至约3000mg/天,更好为从1500mg/天至2500mg/天。氟柳双胺的商品名为可自法国Synthelabo获得。氟柳双胺的化学式为C17H16N2O2。
在哺乳动物中有效量酚加宾的实例包括约5mg/kg/天至约80mg/kg/天的量,优选15mg/kg/天至约50mg/kg/天。对人来说,较好的范围是从约700mg/天至约4000mg/天,更好为从1000mg/天至3000mg/天。酚加宾的商品名为SL79229,可自法国Synthelabo获得。酚加宾的化学式为C17H17C12NO。
在哺乳动物中有效量的γ-羟基丁酸的实例包括约5mg/kg/天至约100mg/kg/天的量,优选10mg/kg/天至约80mg/kg/天。对人来说,较好的范围是从约700mg/天至约5000mg/天,更好为从1000mg/天至4000mg/天。作为钠盐的γ-羟基丁酸可自Sigma化学试剂公司获得。
强迫性用药包括三个独立成分:耐受、心理依赖、以及躯体依赖。耐受可产生一种需求:在几次使用药物后,需要增加药物剂量,以达到同样程度的作用。躯体依赖是反复使用药物后产生的适应状态,其表现为药物给予中断时强烈的躯体障碍。心理依赖的特征为对药物强烈的内驱力、渴望、或使用,而使用者感觉这种效应对保持良好状态是必需的。参见Feldman,R.S.和Quenzer,L.F.,“Fundamentals of Neuropsychopharmocology”(《神经心理药理学基础》)418-422(Sinaur Associates,inc.)(1984),全文结合于此作为参考。根据前述定义,在此所述的“依赖性特征”包括所有与强迫性用药有关、能够被宿主的生物化学组成、生理和心理特性所影响的特征。
报偿/激励效应指的是:任何能够产生快感缺失或者提高后天学会的反应(learned response)的可能性的镇痛刺激。这与“强化”是同义的。就实验动物而言,通过使用据信可测量报偿的方法,可判断一种刺激是否具有报偿性(rewarding)。所用方法为测量刺激能否产生接近反应,又称欲望反应,还是戒断反应,即动物对刺激进行躲避,又叫厌恶反应。条件性位置偏爱(CPP)是一测量接近(欲望)反应或戒断(厌恶)反应的方法。人们可以推断,报偿刺激可以导致接近行为。事实上,报偿的定义之一即为可引发接近行为的刺激。此外,报偿的结果是增加与报偿有关的刺激的激励特性。
报偿也可以通过确定一种报偿的递送是否视一种特殊反应而定来进行测量,从而增加该反应在相似条件下重复出现的可能,即强化方法。例如,一只大鼠以某一固定次数击压一小棒以获取药物注射,这便是一个强化的例子。另一种测量报偿效应的方法是,通过与中性环境刺激的多重配对,确定一种刺激(如一种药物)是否能使以前为中性的环境刺激引发原来只与药物有关的行为效应。这就是条件强化。CPP可被视为条件强化的一种形式。
一种药物的激励动机值可以用条件性位置偏爱(CPP)来估算。动物在一种没有药物的状态下接受测试,以决定与从前曾接受盐水的环境比较,它们是否更倾向于从前曾接受药物的环境。在CPP模式中,动物在一种截然不同的环境下接受药物,而在另一环境下接受合适的载体。CPP模式在评价实验动物对药物的激励动机效应时被广泛使用。在进行条件化或与药物配对后,如果该动物,在无药物的情况下,一贯地选择从前与药物相关的环境,那么可以推论:药物的欲望值已经被编码入大脑,而且在无药物存在时仍容易受影响。CPP反映在,相对于注射载体的对照动物,动物在有与药物有关的刺激存在的环境中花费更长时间。
有人假定,由于人类对药物的渴求通常是由从前与用药有关的感觉刺激所引起的,类似CPP这样的条件化模式可以用来建立实验动物渴求反应的模型。
如在本文所使用的,对镇痛药的渴求指的是希望自身给予曾经被哺乳动物使用过的镇痛药。该哺乳动物不需要用镇痛药来防止戒断症状。
如前面所论述的,本发明的方法设想任何给药形式。GABA能药剂可在镇痛药之前、期间、或与镇痛药同时给予、或其任何组合。优选同时给药。如果GABA能药剂和镇痛药同时给予,则可使用如上所述的本发明的组合物。
通过胃肠道外和肠道途径进行系统给药是优选的。例如,GABA能药剂可静脉内或腹腔内(i.p.)给予。
还曾尝试过口服或肠道给药。诸如片剂、胶囊剂、丸剂、锭剂、酏剂、混悬剂、糖浆剂、糯米纸囊剂、嚼用口胶剂(chewing gum)等剂型都能够用来提供GABA能药剂。
下述实施例是用来帮助进一步理解本发明。使用的特定材料和条件是为了进一步说明本发明而不是限制其合理的范围。
实施例1
在自由活动的大鼠中进行探查,内源性GABA活性增加对吗啡诱导的伏隔核(NAcc)的细胞外多巴胺浓度的影响。
所有动物均使用IACUC认可的实验程序,并严格遵循NIH准则。成年雄性Sprague-Dawley大鼠(体重200-300g,Taconic农场)在12∶12的明/暗条件下,饲养在动物饲养设施中。动物分成6组(n=3-6),研究开始前至少4天,将动物麻醉,以趋实体的方式将硅化的指引套管植入右侧NAcc(位于前囟前方2.0mm、侧方1.0mm、皮质表面腹侧7.0mm)和额叶前部皮质(PFC)。将微透析探针(2.0mm,Bioanalytical Systems(生物分析系统公司),BAS,WestLafayette,IN)置入引导套管内,使用CMA/100微灌流泵(BAS),以2.0μl/分钟的流速,通过探针给予人工脑脊液(ACSF,155.0mMNa-,1.1mM Ca2-,2.9mM K-,132.76mM Cl-,和0.83mM Mg2-)。
将动物放在碗中,插入探针,用ACSF灌流过夜。在研究当天,最少要注射3个样品,以确定基线稳定性。收集样品20分钟,在线注射(CMA/160,BAS)。3个稳定样品的平均多巴胺浓度定为对照值(100%),所有随后处理值都换算成对照值的百分比。一旦建立起稳定的基线,便经腹腔(i.p.)注射吗啡。高效液相层析(HPLC)系统由一个BAS逆相柱(3.0μC-18)、一个BAS LC-4C电化学传感器(带有一个双/玻璃碳电极,电压设为650mV)、一台使用商业软件包(Chromograph Bioanalytical Systems)进行在线分析数据的计算机、以及一个双笔图表记录仪组成。流动相(流速1.0ml/分钟)的组成如下:7.0%甲醇、50mM磷酸二氢钠(sodium phosphatemonobasic)、1.0mM辛基硫酸钠、以及0.1mM EDNA,pH值4.0。多巴胺洗脱7.5分钟。
γ-乙烯基-γ-氨基丁酸(GVG)是一种不可逆的GABA转氨酶抑制剂,其在注射吗啡(7mg/kg)之前2.5小时经腹腔注射。在所有研究中,进行实验的前一天晚上,都将动物放在微透析碗中,通过微透析探针,以2.0μl/分钟的流速,灌注人工脑脊液(ACSF)。每次研究完毕时,将动物杀死,取出大脑,并且进行切片,以检验探针位置。
通过以趋实体方式植入的探针对NAcc中细胞外DA水平进行连续取样。其结果显示于图1中。单独给以吗啡提高NAcc中DA浓度至基线以上50%(p<0.01,T=3.79)。GVG剂量依赖地减弱NAcc中DA对吗啡的反应,其中事先给以150mg/kg GVG处理后无明显抑制,事先给以300mg/kg GVG处理后减弱62%(p<0.01,T=4.97),以及事先给以500mg/kg GVG处理后减弱67%(p<0.001,T=6.02)。这些数据表明GABA能系统是作为通过降低细胞外DA浓度来减轻或消除镇痛药致瘾性的目标。
实施例2
利用电热板试验研究了在雄性Sprague-Dawley大鼠中GVG对吗啡的镇痛效能的影响。在该模式中,对动物进行处理并放置在53℃的电热板上,接着测量舐前爪之一的潜伏期。该试验是用来筛选药剂,其具有中等到显著的镇痛作用,因为这些药物将增加舐前爪的潜伏期。
在接收载体或吗啡前2.5小时,用载体或GVG对大鼠进行处理。在将动物放置在电热板(恒温53℃)上之前30分钟,将载体或吗啡给予动物。通过测量放置在电热板上以后的舐前爪的潜伏期对镇痛效能进行评估。对每个处理组研究了总共10只大鼠。每个值表示舐前爪的潜伏期(单位为秒)±S.E.M。将结果列于表1中。
表1
预处理 处理 舐前爪的潜伏期(秒)
| 载体,1ml/kg | 载体,1ml/kg | 11±0.8 |
| GVG,300mg/kg | 载体,1ml/kg | 17±1.4+ |
| 载体,1ml/kg | 吗啡,10mg/kg | 27±3.0* |
| GVG,300mg/kg | 吗啡,10mg/kg | 27±2.9# |
*显著大于载体预处理/载体处理,p<0.01,方差分析和Student-Newman-Keuls检验。
#显著大于GVG预处理/载体处理,p<0.05,方差分析和Student-Newman-Keuls检验。
+显著大于载体预处理/载体处理,p<0.05,方差分析和Student-Newman-Keuls检验。
这些结果清楚表明,与载体处理的动物相比,腹腔注射10mg/kg的吗啡导致舐前爪的潜伏期显著增加,即吗啡产生镇痛效能。此外,GVG+载体比载体+载体产生显著更长的潜伏期。然而,与载体+吗啡相比,接收吗啡前2.5小时,给予GVG并不显著改变舐前爪的潜伏期。这些结果表明,腹腔注射300mg/kg的GVG并不改变吗啡的镇痛效能。
实施例3
然后研究了海洛因诱导的条件性位置偏爱。海洛因是有效的镇痛药,与吗啡类似。
在所有啮齿动物研究中,使用了雄性Sprague-Dawley大鼠(体重200-225g,Taconic农场,Germantown,NY)。在开始实验前,动物被允许适应动物饲养设施至少5天。使用了条件性位置偏爱(CPP)箱,如先前所述(Lepore et al.,1995),不同之处在于:并不是一个箱是完全白色,另一个箱是黑色;而是一个箱是完全浅蓝色并具有不锈钢底板,而第二个箱是浅蓝色并具有水平的黑条纹(2.5cm宽)且与平滑的树脂玻璃底板相隔3.8cm。在所有用GVG进行的CPP研究中,盐水体积是(1ml/kg),而海洛因剂量则是1.5mg/kg。盐水、海洛因、和GVG都是腹腔注射(i.p.)。习得阶段(acquisition phase)的调节步骤包括12个时间段,其连续进行12天。
CPP配对是:1)盐水/盐水2)盐水/海洛因3)GVG/盐水4)盐水/海洛因和GVG。每组的动物随机给定2×2因子设计,其中一个因子是配对箱,而另一个因子是调节次序。对接收盐水或海洛因的动物进行注射并限制在合适的室(compartment)中30分钟。在盐水或海洛因注射前3小时注射GVG并随后将动物放置在适当的箱中。这样做的原因在于:研究表明,在GVG给药后3至4小时GABA水平达到最大值。
在试验当天(12天),既不给予药物也不给予盐水并且允许动物在两箱之间自由走动15分钟。在每个箱中度过的时间是利用电偶联于计时器的自动红外光束进行记录。就对海洛因的CPP表达阶段而言,动物习惯和适应了海洛因,如在习得研究中所述的,但没有任何在表达研究中的动物在调节(conditioning)的日子里给予GVG。在试验当天(12天),在表达阶段被试验的动物,其与在习得阶段的动物不同,在它们被放置在仪器中之前2.5小时接收盐水或GVG并允许自由进入两箱15分钟。
将结果列于下面的表2中。
表2
1每个值表示在每个箱中度过的平均分钟数±S.E.M。对于每个处理配对研究了8至10只大鼠。
2载体是腹腔注射1ml/kg的0.9%盐水。
*显著大于所有其他组,p<0.01,方差分析和Student-Newman-Keuls检验。
#显著小于所有其他组,p<0.01,方差分析和Student-Newman-Keuls检验。
该结果清楚表明,300mg/kg的GVG阻断了海洛因诱导的CPP的表达。
因而,表1和表2的结果一起说明GVG能够阻断对海洛因(一种有特效的成瘾镇痛药)的渴求,同时并不降低减轻疼痛的镇痛功效。因而,GABA能药剂,如与阿片类镇痛药一起使用的GVG,将降低对镇痛药成瘾的可能性,而不降低其在疼痛治疗中的治疗效果。
尽管本发明已经参照优选具体实施例进行了说明,但是,对于本领域的技术人员来说,本发明可以有各种更改和变化,而不偏离本发明的精神。本发明的各种更改、变化、和等同物由所附的权利要求书的内容涵盖。
Claims (6)
1.γ-乙烯基-γ-氨基丁酸在制备用于减轻麻醉性镇痛药的致瘾性的药剂中的应用,所述用于减轻麻醉性镇痛药的致瘾性的药剂包括:
a)有效量的麻醉性镇痛药,以及
b)有效量的γ-乙烯基-γ-氨基丁酸。
2.根据权利要求1所述的应用,其中所述麻醉性镇痛药是吗啡。
3.根据权利要求1所述的应用,其中所述麻醉性镇痛药与所述γ-乙烯基-γ-氨基丁酸在同一组合物中。
4.据权利要求1所述的应用,其中所述γ-乙烯基-γ-氨基丁酸的量为500mg至5g。
5.用于减轻麻醉性镇痛药的致瘾性的组合物,所述组合物包括:
a)有效量的麻醉性镇痛药,以及
b)有效量的γ-乙烯基-γ-氨基丁酸。
6.根据权利要求5所述的组合物,其中所述麻醉性镇痛药是吗啡。
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| US09/853,548 US20020187996A1 (en) | 2001-05-14 | 2001-05-14 | Prevention of addiction in pain management |
| US09/853,548 | 2001-05-14 |
Publications (2)
| Publication Number | Publication Date |
|---|---|
| CN1531431A CN1531431A (zh) | 2004-09-22 |
| CN100528156C true CN100528156C (zh) | 2009-08-19 |
Family
ID=25316324
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| CNB028098277A Expired - Fee Related CN100528156C (zh) | 2001-05-14 | 2002-04-08 | 在疼痛治疗中成瘾的预防 |
Country Status (21)
| Country | Link |
|---|---|
| US (3) | US20020187996A1 (zh) |
| EP (1) | EP1392301B1 (zh) |
| JP (1) | JP2004534029A (zh) |
| KR (1) | KR20040020054A (zh) |
| CN (1) | CN100528156C (zh) |
| AT (1) | ATE473001T1 (zh) |
| AU (1) | AU2002342533B2 (zh) |
| BR (1) | BR0209613A (zh) |
| CA (1) | CA2446639C (zh) |
| CZ (1) | CZ20033380A3 (zh) |
| DE (1) | DE60236927D1 (zh) |
| ES (1) | ES2348637T3 (zh) |
| HR (1) | HRP20030892A2 (zh) |
| HU (1) | HUP0402326A3 (zh) |
| IL (1) | IL158762A0 (zh) |
| MX (1) | MXPA03010407A (zh) |
| NO (1) | NO331509B1 (zh) |
| NZ (1) | NZ529614A (zh) |
| PL (1) | PL368505A1 (zh) |
| WO (1) | WO2002091990A2 (zh) |
| YU (1) | YU89003A (zh) |
Families Citing this family (8)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2002009707A1 (en) * | 2000-07-31 | 2002-02-07 | Nycomed Danmark A/S | Fentanyl composition for nasal administration |
| DE10141650C1 (de) | 2001-08-24 | 2002-11-28 | Lohmann Therapie Syst Lts | Transdermales Therapeutisches System mit Fentanyl bzw. verwandten Substanzen |
| US20050043345A1 (en) * | 2003-08-22 | 2005-02-24 | Pfizer Inc | Pharmaceutical composition for the prevention and treatment of addiction in a mammal |
| US8652527B1 (en) | 2013-03-13 | 2014-02-18 | Upsher-Smith Laboratories, Inc | Extended-release topiramate capsules |
| US9101545B2 (en) | 2013-03-15 | 2015-08-11 | Upsher-Smith Laboratories, Inc. | Extended-release topiramate capsules |
| CN105497896A (zh) * | 2015-03-31 | 2016-04-20 | 苏州大学 | 一种肿瘤耐药的靶点及其应用 |
| JP2024504902A (ja) | 2020-12-04 | 2024-02-02 | ラボラトリオス シラネス、エセ.ア.デ セ.べ. | 痛みに対するオピオイド鎮痛薬と抗てんかん薬のコーティングされた安定な固体医薬組成物 |
| US11977085B1 (en) | 2023-09-05 | 2024-05-07 | Elan Ehrlich | Date rape drug detection device and method of using same |
Family Cites Families (2)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US6057368A (en) * | 1998-08-05 | 2000-05-02 | Brookhaven Science Associates Llc | Treatment of addiction and addiction-related behavior |
| US6541520B1 (en) * | 1998-08-05 | 2003-04-01 | Brookhaven Science Associates | Treatment of addiction and addiction-related behavior |
-
2001
- 2001-05-14 US US09/853,548 patent/US20020187996A1/en not_active Abandoned
-
2002
- 2002-04-08 KR KR10-2003-7014736A patent/KR20040020054A/ko not_active Application Discontinuation
- 2002-04-08 BR BR0209613-7A patent/BR0209613A/pt not_active IP Right Cessation
- 2002-04-08 AU AU2002342533A patent/AU2002342533B2/en not_active Ceased
- 2002-04-08 DE DE60236927T patent/DE60236927D1/de not_active Expired - Lifetime
- 2002-04-08 YU YU89003A patent/YU89003A/sh unknown
- 2002-04-08 JP JP2002588909A patent/JP2004534029A/ja active Pending
- 2002-04-08 CZ CZ20033380A patent/CZ20033380A3/cs unknown
- 2002-04-08 CN CNB028098277A patent/CN100528156C/zh not_active Expired - Fee Related
- 2002-04-08 WO PCT/US2002/011072 patent/WO2002091990A2/en active IP Right Grant
- 2002-04-08 HU HU0402326A patent/HUP0402326A3/hu unknown
- 2002-04-08 ES ES02769652T patent/ES2348637T3/es not_active Expired - Lifetime
- 2002-04-08 AT AT02769652T patent/ATE473001T1/de not_active IP Right Cessation
- 2002-04-08 IL IL15876202A patent/IL158762A0/xx unknown
- 2002-04-08 NZ NZ529614A patent/NZ529614A/en unknown
- 2002-04-08 CA CA2446639A patent/CA2446639C/en not_active Expired - Fee Related
- 2002-04-08 PL PL02368505A patent/PL368505A1/xx not_active Application Discontinuation
- 2002-04-08 MX MXPA03010407A patent/MXPA03010407A/es not_active Application Discontinuation
- 2002-04-08 EP EP02769652A patent/EP1392301B1/en not_active Expired - Lifetime
- 2002-04-18 US US10/124,660 patent/US6939876B2/en not_active Expired - Fee Related
-
2003
- 2003-01-02 US US10/335,800 patent/US20030134868A1/en not_active Abandoned
- 2003-11-05 HR HR20030892A patent/HRP20030892A2/xx not_active Application Discontinuation
- 2003-11-13 NO NO20035038A patent/NO331509B1/no not_active IP Right Cessation
Also Published As
| Publication number | Publication date |
|---|---|
| NO331509B1 (no) | 2012-01-16 |
| NO20035038D0 (no) | 2003-11-13 |
| WO2002091990A2 (en) | 2002-11-21 |
| JP2004534029A (ja) | 2004-11-11 |
| BR0209613A (pt) | 2004-12-07 |
| US6939876B2 (en) | 2005-09-06 |
| CN1531431A (zh) | 2004-09-22 |
| US20020187996A1 (en) | 2002-12-12 |
| ES2348637T3 (es) | 2010-12-10 |
| US20030134868A1 (en) | 2003-07-17 |
| IL158762A0 (en) | 2004-05-12 |
| PL368505A1 (en) | 2005-04-04 |
| EP1392301B1 (en) | 2010-07-07 |
| ATE473001T1 (de) | 2010-07-15 |
| CZ20033380A3 (cs) | 2005-03-16 |
| HUP0402326A2 (hu) | 2005-02-28 |
| CA2446639C (en) | 2010-11-02 |
| EP1392301A4 (en) | 2004-07-14 |
| CA2446639A1 (en) | 2002-11-21 |
| US20030004176A1 (en) | 2003-01-02 |
| DE60236927D1 (de) | 2010-08-19 |
| KR20040020054A (ko) | 2004-03-06 |
| YU89003A (sh) | 2006-08-17 |
| EP1392301A2 (en) | 2004-03-03 |
| MXPA03010407A (es) | 2004-05-05 |
| WO2002091990A3 (en) | 2003-03-27 |
| AU2002342533B2 (en) | 2007-07-05 |
| HUP0402326A3 (en) | 2008-04-28 |
| HRP20030892A2 (en) | 2005-08-31 |
| NZ529614A (en) | 2006-08-31 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| US5578645A (en) | Methods and compositions for treating acute or chronic pain and drug addiction | |
| TWI243674B (en) | Use of gamma vinylGABA (GVG) or a pharmaceutically acceptable salt thereof, or an enantiomer or a racemic mixture thereof | |
| US20060177381A1 (en) | Opiopathies | |
| CN100528156C (zh) | 在疼痛治疗中成瘾的预防 | |
| WO1983003541A1 (en) | Antagonism of central nervous system drugs by the administration of 4-aminopyridine alone or in combination with other drugs | |
| Skoogh et al. | Barbiturates depress vagal motor pathway to ferret trachea at ganglia | |
| Koob et al. | Opioids | |
| JP2017506244A (ja) | ノルイボガイン及び関連化合物を用いた治療方法 | |
| AU2002342533A1 (en) | Prevention of addiction in pain management | |
| EP2007388B1 (en) | Opiopathies | |
| Eddy et al. | Codeine and its alternates for pain and cough relief | |
| Vandam | Some aspects of the history of local anesthesia | |
| WO2021113757A1 (en) | Use of a kv7 potassium channel opener for treating pain | |
| CN112569237A (zh) | 伊马替尼及其衍生物与尼古丁或其类似物联用或复方在防治尼古丁成瘾与复吸中的应用 | |
| CN111214469A (zh) | 预防和治疗慢性疼痛的化合物及其应用 | |
| Klifto et al. | Pain management in burn patients: pharmacologic management of acute and chronic pain | |
| Akuzawa et al. | Effect of ASP8062 on morphine self-administration and morphine-induced respiratory suppression in monkeys | |
| JP2020526500A (ja) | 治療方法及びその剤形 | |
| Glaubach | Sulfapyridine potentiation of narcotic and toxic effects of papaverine in rats and rabbits | |
| CN115969850A (zh) | 小檗胺在制备镇痛药物中的应用 | |
| Karandikar et al. | Comparative study of CNS effects of opium in homeopathic potencies with pentazocine in mice | |
| CA3222744A1 (en) | Methods of treating or preventing conditions associated with opiate withdrawal or opiate relapse | |
| CN1476326A (zh) | 苯环利啶成瘾及苯环利啶成瘾相关行为的治疗 | |
| Madison | The effect of a neuropeptide FF receptor subtype-selective compound on morphine tolerance | |
| Dumke | Newer agents for the anesthesiologist |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| C06 | Publication | ||
| PB01 | Publication | ||
| C10 | Entry into substantive examination | ||
| SE01 | Entry into force of request for substantive examination | ||
| C14 | Grant of patent or utility model | ||
| GR01 | Patent grant | ||
| C17 | Cessation of patent right | ||
| CF01 | Termination of patent right due to non-payment of annual fee |
Granted publication date: 20090819 Termination date: 20110408 |