WO2023025312A1 - 使用th-302治疗parp抑制剂耐药的患者 - Google Patents
使用th-302治疗parp抑制剂耐药的患者 Download PDFInfo
- Publication number
- WO2023025312A1 WO2023025312A1 PCT/CN2022/115284 CN2022115284W WO2023025312A1 WO 2023025312 A1 WO2023025312 A1 WO 2023025312A1 CN 2022115284 W CN2022115284 W CN 2022115284W WO 2023025312 A1 WO2023025312 A1 WO 2023025312A1
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- cancer
- tumor
- mutations
- brca2
- brca1
- Prior art date
Links
- 239000012661 PARP inhibitor Substances 0.000 title claims abstract description 68
- 229940121906 Poly ADP ribose polymerase inhibitor Drugs 0.000 title claims abstract description 68
- 239000003814 drug Substances 0.000 claims abstract description 132
- 229940079593 drug Drugs 0.000 claims abstract description 122
- 238000011282 treatment Methods 0.000 claims abstract description 83
- 238000000034 method Methods 0.000 claims abstract description 19
- 206010028980 Neoplasm Diseases 0.000 claims description 159
- FAQDUNYVKQKNLD-UHFFFAOYSA-N olaparib Chemical group FC1=CC=C(CC2=C3[CH]C=CC=C3C(=O)N=N2)C=C1C(=O)N(CC1)CCN1C(=O)C1CC1 FAQDUNYVKQKNLD-UHFFFAOYSA-N 0.000 claims description 64
- 229960000572 olaparib Drugs 0.000 claims description 63
- 150000001875 compounds Chemical class 0.000 claims description 60
- 206010021143 Hypoxia Diseases 0.000 claims description 59
- 230000035772 mutation Effects 0.000 claims description 57
- 108700020462 BRCA2 Proteins 0.000 claims description 42
- 102000052609 BRCA2 Human genes 0.000 claims description 42
- 201000011510 cancer Diseases 0.000 claims description 41
- 102000036365 BRCA1 Human genes 0.000 claims description 39
- 101150008921 Brca2 gene Proteins 0.000 claims description 39
- 108700020463 BRCA1 Proteins 0.000 claims description 36
- 206010061902 Pancreatic neoplasm Diseases 0.000 claims description 36
- 208000015486 malignant pancreatic neoplasm Diseases 0.000 claims description 36
- 201000002528 pancreatic cancer Diseases 0.000 claims description 36
- 208000008443 pancreatic carcinoma Diseases 0.000 claims description 36
- 101150072950 BRCA1 gene Proteins 0.000 claims description 35
- 206010064571 Gene mutation Diseases 0.000 claims description 32
- 230000007954 hypoxia Effects 0.000 claims description 32
- 108090000623 proteins and genes Proteins 0.000 claims description 27
- 206010005003 Bladder cancer Diseases 0.000 claims description 22
- 208000007097 Urinary Bladder Neoplasms Diseases 0.000 claims description 22
- 201000005112 urinary bladder cancer Diseases 0.000 claims description 22
- 206010033128 Ovarian cancer Diseases 0.000 claims description 21
- 206010058467 Lung neoplasm malignant Diseases 0.000 claims description 20
- 201000005202 lung cancer Diseases 0.000 claims description 20
- 208000020816 lung neoplasm Diseases 0.000 claims description 20
- 206010061535 Ovarian neoplasm Diseases 0.000 claims description 19
- 238000002360 preparation method Methods 0.000 claims description 14
- 201000001342 Fallopian tube cancer Diseases 0.000 claims description 13
- 208000013452 Fallopian tube neoplasm Diseases 0.000 claims description 13
- 208000026149 Primary peritoneal carcinoma Diseases 0.000 claims description 13
- 238000009097 single-agent therapy Methods 0.000 claims description 13
- 206010006187 Breast cancer Diseases 0.000 claims description 12
- 102000011724 DNA Repair Enzymes Human genes 0.000 claims description 12
- 108010076525 DNA Repair Enzymes Proteins 0.000 claims description 12
- 206010041067 Small cell lung cancer Diseases 0.000 claims description 12
- 208000000587 small cell lung carcinoma Diseases 0.000 claims description 12
- 210000001519 tissue Anatomy 0.000 claims description 12
- 208000026310 Breast neoplasm Diseases 0.000 claims description 11
- 206010009944 Colon cancer Diseases 0.000 claims description 11
- 208000029742 colonic neoplasm Diseases 0.000 claims description 11
- 210000004602 germ cell Anatomy 0.000 claims description 11
- 229950011068 niraparib Drugs 0.000 claims description 11
- 208000005718 Stomach Neoplasms Diseases 0.000 claims description 10
- PCHKPVIQAHNQLW-CQSZACIVSA-N niraparib Chemical compound N1=C2C(C(=O)N)=CC=CC2=CN1C(C=C1)=CC=C1[C@@H]1CCCNC1 PCHKPVIQAHNQLW-CQSZACIVSA-N 0.000 claims description 10
- HMABYWSNWIZPAG-UHFFFAOYSA-N rucaparib Chemical compound C1=CC(CNC)=CC=C1C(N1)=C2CCNC(=O)C3=C2C1=CC(F)=C3 HMABYWSNWIZPAG-UHFFFAOYSA-N 0.000 claims description 10
- 229950004707 rucaparib Drugs 0.000 claims description 10
- 206010060862 Prostate cancer Diseases 0.000 claims description 9
- 208000000236 Prostatic Neoplasms Diseases 0.000 claims description 9
- 208000015634 Rectal Neoplasms Diseases 0.000 claims description 9
- HWGQMRYQVZSGDQ-HZPDHXFCSA-N chembl3137320 Chemical compound CN1N=CN=C1[C@H]([C@H](N1)C=2C=CC(F)=CC=2)C2=NNC(=O)C3=C2C1=CC(F)=C3 HWGQMRYQVZSGDQ-HZPDHXFCSA-N 0.000 claims description 9
- 125000000524 functional group Chemical group 0.000 claims description 9
- 206010017758 gastric cancer Diseases 0.000 claims description 9
- 201000007270 liver cancer Diseases 0.000 claims description 9
- 208000014018 liver neoplasm Diseases 0.000 claims description 9
- 206010038038 rectal cancer Diseases 0.000 claims description 9
- 201000001275 rectum cancer Diseases 0.000 claims description 9
- 201000011549 stomach cancer Diseases 0.000 claims description 9
- 229950004550 talazoparib Drugs 0.000 claims description 9
- 238000002560 therapeutic procedure Methods 0.000 claims description 9
- XJGXCBHXFWBOTN-UHFFFAOYSA-N 4-[[4-fluoro-3-[2-(trifluoromethyl)-6,8-dihydro-5h-[1,2,4]triazolo[1,5-a]pyrazine-7-carbonyl]phenyl]methyl]-2h-phthalazin-1-one Chemical compound C1CN2N=C(C(F)(F)F)N=C2CN1C(=O)C1=CC(CC=2C3=CC=CC=C3C(=O)NN=2)=CC=C1F XJGXCBHXFWBOTN-UHFFFAOYSA-N 0.000 claims description 8
- DENYZIUJOTUUNY-MRXNPFEDSA-N (2R)-14-fluoro-2-methyl-6,9,10,19-tetrazapentacyclo[14.2.1.02,6.08,18.012,17]nonadeca-1(18),8,12(17),13,15-pentaen-11-one Chemical compound FC=1C=C2C=3C=4C(CN5[C@@](C4NC3C1)(CCC5)C)=NNC2=O DENYZIUJOTUUNY-MRXNPFEDSA-N 0.000 claims description 7
- 230000001771 impaired effect Effects 0.000 claims description 7
- 208000002154 non-small cell lung carcinoma Diseases 0.000 claims description 7
- 229950007072 pamiparib Drugs 0.000 claims description 7
- 208000016691 refractory malignant neoplasm Diseases 0.000 claims description 7
- 208000029729 tumor suppressor gene on chromosome 11 Diseases 0.000 claims description 7
- 230000004913 activation Effects 0.000 claims description 6
- 230000009885 systemic effect Effects 0.000 claims description 6
- 206010071981 BRCA2 gene mutation Diseases 0.000 claims description 5
- 238000001514 detection method Methods 0.000 claims description 5
- -1 preferably Proteins 0.000 claims description 4
- 206010071980 BRCA1 gene mutation Diseases 0.000 claims description 3
- 239000003795 chemical substances by application Substances 0.000 claims description 3
- UGJWRPJDTDGERK-UHFFFAOYSA-N evofosfamide Chemical compound CN1C(COP(=O)(NCCBr)NCCBr)=CN=C1[N+]([O-])=O UGJWRPJDTDGERK-UHFFFAOYSA-N 0.000 abstract description 143
- 229950009988 evofosfamide Drugs 0.000 abstract description 143
- 210000004027 cell Anatomy 0.000 description 86
- 238000012360 testing method Methods 0.000 description 35
- 241000282414 Homo sapiens Species 0.000 description 34
- 241000699670 Mus sp. Species 0.000 description 34
- 230000005764 inhibitory process Effects 0.000 description 31
- 238000002474 experimental method Methods 0.000 description 26
- 238000007920 subcutaneous administration Methods 0.000 description 24
- 229950002376 tirapazamine Drugs 0.000 description 24
- 210000004881 tumor cell Anatomy 0.000 description 24
- QVMPZNRFXAKISM-UHFFFAOYSA-N tirapazamine Chemical compound C1=CC=C2[N+]([O-])=NC(=N)N(O)C2=C1 QVMPZNRFXAKISM-UHFFFAOYSA-N 0.000 description 22
- 230000000694 effects Effects 0.000 description 21
- 229940002612 prodrug Drugs 0.000 description 17
- 239000000651 prodrug Substances 0.000 description 17
- 230000001146 hypoxic effect Effects 0.000 description 16
- 229920000776 Poly(Adenosine diphosphate-ribose) polymerase Polymers 0.000 description 15
- 101710179684 Poly [ADP-ribose] polymerase Proteins 0.000 description 14
- 102100023712 Poly [ADP-ribose] polymerase 1 Human genes 0.000 description 14
- 230000037396 body weight Effects 0.000 description 14
- 102000004190 Enzymes Human genes 0.000 description 13
- 108090000790 Enzymes Proteins 0.000 description 13
- 238000000338 in vitro Methods 0.000 description 13
- 238000011081 inoculation Methods 0.000 description 13
- 229910052760 oxygen Inorganic materials 0.000 description 13
- 239000003981 vehicle Substances 0.000 description 13
- 239000002775 capsule Substances 0.000 description 12
- BASFCYQUMIYNBI-UHFFFAOYSA-N platinum Chemical compound [Pt] BASFCYQUMIYNBI-UHFFFAOYSA-N 0.000 description 12
- 241001465754 Metazoa Species 0.000 description 11
- 239000002552 dosage form Substances 0.000 description 11
- 230000009467 reduction Effects 0.000 description 11
- 238000010171 animal model Methods 0.000 description 10
- 230000012010 growth Effects 0.000 description 10
- 229940100357 olaparib 50 mg Drugs 0.000 description 10
- 239000003826 tablet Substances 0.000 description 10
- 208000031648 Body Weight Changes Diseases 0.000 description 9
- 238000004458 analytical method Methods 0.000 description 9
- 230000004579 body weight change Effects 0.000 description 9
- 239000002609 medium Substances 0.000 description 9
- 230000035755 proliferation Effects 0.000 description 9
- 238000011160 research Methods 0.000 description 9
- 206010067484 Adverse reaction Diseases 0.000 description 8
- 108091007743 BRCA1/2 Proteins 0.000 description 8
- 230000005778 DNA damage Effects 0.000 description 8
- 231100000277 DNA damage Toxicity 0.000 description 8
- 230000006838 adverse reaction Effects 0.000 description 8
- 230000000259 anti-tumor effect Effects 0.000 description 8
- 230000002401 inhibitory effect Effects 0.000 description 8
- 230000035945 sensitivity Effects 0.000 description 8
- WQZGKKKJIJFFOK-GASJEMHNSA-N Glucose Natural products OC[C@H]1OC(O)[C@H](O)[C@@H](O)[C@@H]1O WQZGKKKJIJFFOK-GASJEMHNSA-N 0.000 description 7
- 231100000135 cytotoxicity Toxicity 0.000 description 7
- 230000003013 cytotoxicity Effects 0.000 description 7
- 230000006378 damage Effects 0.000 description 7
- 101150042537 dld1 gene Proteins 0.000 description 7
- 238000011156 evaluation Methods 0.000 description 7
- 239000007924 injection Substances 0.000 description 7
- 238000002347 injection Methods 0.000 description 7
- 230000008439 repair process Effects 0.000 description 7
- 239000000243 solution Substances 0.000 description 7
- 230000001225 therapeutic effect Effects 0.000 description 7
- 230000004614 tumor growth Effects 0.000 description 7
- 230000000996 additive effect Effects 0.000 description 6
- 230000001093 anti-cancer Effects 0.000 description 6
- 230000004663 cell proliferation Effects 0.000 description 6
- 230000008859 change Effects 0.000 description 6
- 229940090044 injection Drugs 0.000 description 6
- 229910052697 platinum Inorganic materials 0.000 description 6
- 230000001875 tumorinhibitory effect Effects 0.000 description 6
- 229950011257 veliparib Drugs 0.000 description 6
- JNAHVYVRKWKWKQ-CYBMUJFWSA-N veliparib Chemical compound N=1C2=CC=CC(C(N)=O)=C2NC=1[C@@]1(C)CCCN1 JNAHVYVRKWKWKQ-CYBMUJFWSA-N 0.000 description 6
- 108020004414 DNA Proteins 0.000 description 5
- 241000699666 Mus <mouse, genus> Species 0.000 description 5
- 239000002246 antineoplastic agent Substances 0.000 description 5
- 229940041181 antineoplastic drug Drugs 0.000 description 5
- 238000002512 chemotherapy Methods 0.000 description 5
- 238000002648 combination therapy Methods 0.000 description 5
- 230000002950 deficient Effects 0.000 description 5
- 239000011521 glass Substances 0.000 description 5
- 229940093181 glucose injection Drugs 0.000 description 5
- 230000020520 nucleotide-excision repair Effects 0.000 description 5
- 230000002829 reductive effect Effects 0.000 description 5
- 210000003462 vein Anatomy 0.000 description 5
- 230000033616 DNA repair Effects 0.000 description 4
- 230000033590 base-excision repair Effects 0.000 description 4
- 201000010099 disease Diseases 0.000 description 4
- 208000037265 diseases, disorders, signs and symptoms Diseases 0.000 description 4
- 230000006801 homologous recombination Effects 0.000 description 4
- 238000002744 homologous recombination Methods 0.000 description 4
- 230000007246 mechanism Effects 0.000 description 4
- 239000000203 mixture Substances 0.000 description 4
- 230000007959 normoxia Effects 0.000 description 4
- 230000036961 partial effect Effects 0.000 description 4
- 230000001717 pathogenic effect Effects 0.000 description 4
- 239000000546 pharmaceutical excipient Substances 0.000 description 4
- 238000011518 platinum-based chemotherapy Methods 0.000 description 4
- 239000000843 powder Substances 0.000 description 4
- 239000012624 DNA alkylating agent Substances 0.000 description 3
- 229940126161 DNA alkylating agent Drugs 0.000 description 3
- 206010061818 Disease progression Diseases 0.000 description 3
- 208000007571 Ovarian Epithelial Carcinoma Diseases 0.000 description 3
- 206010039491 Sarcoma Diseases 0.000 description 3
- QVGXLLKOCUKJST-UHFFFAOYSA-N atomic oxygen Chemical compound [O] QVGXLLKOCUKJST-UHFFFAOYSA-N 0.000 description 3
- 230000009286 beneficial effect Effects 0.000 description 3
- 238000004113 cell culture Methods 0.000 description 3
- 239000006285 cell suspension Substances 0.000 description 3
- 230000001419 dependent effect Effects 0.000 description 3
- 230000005750 disease progression Effects 0.000 description 3
- 231100000673 dose–response relationship Toxicity 0.000 description 3
- 230000034431 double-strand break repair via homologous recombination Effects 0.000 description 3
- 239000000796 flavoring agent Substances 0.000 description 3
- 230000006870 function Effects 0.000 description 3
- 238000001802 infusion Methods 0.000 description 3
- 231100000225 lethality Toxicity 0.000 description 3
- 239000000463 material Substances 0.000 description 3
- 230000006780 non-homologous end joining Effects 0.000 description 3
- 239000001301 oxygen Substances 0.000 description 3
- 230000037361 pathway Effects 0.000 description 3
- 230000008569 process Effects 0.000 description 3
- 102000004169 proteins and genes Human genes 0.000 description 3
- 230000000306 recurrent effect Effects 0.000 description 3
- 230000004044 response Effects 0.000 description 3
- 206010055113 Breast cancer metastatic Diseases 0.000 description 2
- 201000009030 Carcinoma Diseases 0.000 description 2
- 230000000970 DNA cross-linking effect Effects 0.000 description 2
- LFQSCWFLJHTTHZ-UHFFFAOYSA-N Ethanol Chemical compound CCO LFQSCWFLJHTTHZ-UHFFFAOYSA-N 0.000 description 2
- 206010025323 Lymphomas Diseases 0.000 description 2
- 241000124008 Mammalia Species 0.000 description 2
- 102100023897 NADPH-cytochrome P450 reductase Human genes 0.000 description 2
- 208000005890 Neuroma Diseases 0.000 description 2
- 108090000854 Oxidoreductases Proteins 0.000 description 2
- 102000004316 Oxidoreductases Human genes 0.000 description 2
- 229920003171 Poly (ethylene oxide) Polymers 0.000 description 2
- 229920000297 Rayon Polymers 0.000 description 2
- 208000000453 Skin Neoplasms Diseases 0.000 description 2
- 241000711955 Turkey rhinotracheitis virus Species 0.000 description 2
- 239000004480 active ingredient Substances 0.000 description 2
- 239000002671 adjuvant Substances 0.000 description 2
- 239000003963 antioxidant agent Substances 0.000 description 2
- 235000006708 antioxidants Nutrition 0.000 description 2
- FUNYDODCWWGJJA-UHFFFAOYSA-N bis(2-bromoethylamino)phosphinic acid Chemical compound BrCCNP(=O)(O)NCCBr FUNYDODCWWGJJA-UHFFFAOYSA-N 0.000 description 2
- BJQHLKABXJIVAM-UHFFFAOYSA-N bis(2-ethylhexyl) phthalate Chemical compound CCCCC(CC)COC(=O)C1=CC=CC=C1C(=O)OCC(CC)CCCC BJQHLKABXJIVAM-UHFFFAOYSA-N 0.000 description 2
- 239000012830 cancer therapeutic Substances 0.000 description 2
- 239000004359 castor oil Substances 0.000 description 2
- 235000019438 castor oil Nutrition 0.000 description 2
- 230000030833 cell death Effects 0.000 description 2
- 230000001413 cellular effect Effects 0.000 description 2
- 238000006243 chemical reaction Methods 0.000 description 2
- 229940044683 chemotherapy drug Drugs 0.000 description 2
- 239000007910 chewable tablet Substances 0.000 description 2
- 229940068682 chewable tablet Drugs 0.000 description 2
- 239000011248 coating agent Substances 0.000 description 2
- 208000035250 cutaneous malignant susceptibility to 1 melanoma Diseases 0.000 description 2
- 231100000599 cytotoxic agent Toxicity 0.000 description 2
- 239000002619 cytotoxin Substances 0.000 description 2
- 230000034994 death Effects 0.000 description 2
- 230000007423 decrease Effects 0.000 description 2
- 230000002939 deleterious effect Effects 0.000 description 2
- 238000011161 development Methods 0.000 description 2
- 239000008121 dextrose Substances 0.000 description 2
- 239000003085 diluting agent Substances 0.000 description 2
- 239000007884 disintegrant Substances 0.000 description 2
- 239000007919 dispersible tablet Substances 0.000 description 2
- 229940126534 drug product Drugs 0.000 description 2
- 238000002651 drug therapy Methods 0.000 description 2
- 239000002662 enteric coated tablet Substances 0.000 description 2
- 239000000945 filler Substances 0.000 description 2
- 235000013355 food flavoring agent Nutrition 0.000 description 2
- 235000003599 food sweetener Nutrition 0.000 description 2
- 238000003304 gavage Methods 0.000 description 2
- ZEMPKEQAKRGZGQ-XOQCFJPHSA-N glycerol triricinoleate Natural products CCCCCC[C@@H](O)CC=CCCCCCCCC(=O)OC[C@@H](COC(=O)CCCCCCCC=CC[C@@H](O)CCCCCC)OC(=O)CCCCCCCC=CC[C@H](O)CCCCCC ZEMPKEQAKRGZGQ-XOQCFJPHSA-N 0.000 description 2
- 239000008187 granular material Substances 0.000 description 2
- 239000001963 growth medium Substances 0.000 description 2
- 238000005304 joining Methods 0.000 description 2
- 208000032839 leukemia Diseases 0.000 description 2
- 238000011068 loading method Methods 0.000 description 2
- 239000000314 lubricant Substances 0.000 description 2
- 238000009115 maintenance therapy Methods 0.000 description 2
- 230000010534 mechanism of action Effects 0.000 description 2
- 201000001441 melanoma Diseases 0.000 description 2
- 230000033607 mismatch repair Effects 0.000 description 2
- 239000002773 nucleotide Substances 0.000 description 2
- 125000003729 nucleotide group Chemical group 0.000 description 2
- 229940100688 oral solution Drugs 0.000 description 2
- 239000006191 orally-disintegrating tablet Substances 0.000 description 2
- 239000000825 pharmaceutical preparation Substances 0.000 description 2
- 239000000049 pigment Substances 0.000 description 2
- 238000007747 plating Methods 0.000 description 2
- 239000003755 preservative agent Substances 0.000 description 2
- 150000003254 radicals Chemical class 0.000 description 2
- 238000011946 reduction process Methods 0.000 description 2
- 230000002441 reversible effect Effects 0.000 description 2
- 238000012163 sequencing technique Methods 0.000 description 2
- 239000002904 solvent Substances 0.000 description 2
- 206010041823 squamous cell carcinoma Diseases 0.000 description 2
- 239000000829 suppository Substances 0.000 description 2
- 239000004094 surface-active agent Substances 0.000 description 2
- 239000000375 suspending agent Substances 0.000 description 2
- 239000003765 sweetening agent Substances 0.000 description 2
- 230000002195 synergetic effect Effects 0.000 description 2
- 238000002054 transplantation Methods 0.000 description 2
- XLYOFNOQVPJJNP-UHFFFAOYSA-N water Substances O XLYOFNOQVPJJNP-UHFFFAOYSA-N 0.000 description 2
- WVWOOAYQYLJEFD-UHFFFAOYSA-N 1-(2-nitroimidazol-1-yl)-3-piperidin-1-ylpropan-2-ol Chemical compound C1=CN=C([N+]([O-])=O)N1CC(O)CN1CCCCC1 WVWOOAYQYLJEFD-UHFFFAOYSA-N 0.000 description 1
- NHBKXEKEPDILRR-UHFFFAOYSA-N 2,3-bis(butanoylsulfanyl)propyl butanoate Chemical compound CCCC(=O)OCC(SC(=O)CCC)CSC(=O)CCC NHBKXEKEPDILRR-UHFFFAOYSA-N 0.000 description 1
- YZBAXVICWUUHGG-UHFFFAOYSA-N 2-[[4-[2-[dimethyl(oxido)azaniumyl]ethylamino]-5,8-dihydroxy-9,10-dioxoanthracen-1-yl]amino]-n,n-dimethylethanamine oxide Chemical compound O=C1C2=C(O)C=CC(O)=C2C(=O)C2=C1C(NCC[N+](C)(C)[O-])=CC=C2NCC[N+](C)([O-])C YZBAXVICWUUHGG-UHFFFAOYSA-N 0.000 description 1
- GZSOKPMDWVRVMG-UHFFFAOYSA-N 2-[n-(2-bromoethyl)-2,4-dinitro-6-(2-phosphonooxyethylcarbamoyl)anilino]ethyl methanesulfonate Chemical compound CS(=O)(=O)OCCN(CCBr)C1=C(C(=O)NCCOP(O)(O)=O)C=C([N+]([O-])=O)C=C1[N+]([O-])=O GZSOKPMDWVRVMG-UHFFFAOYSA-N 0.000 description 1
- YZEUHQHUFTYLPH-UHFFFAOYSA-N 2-nitroimidazole Chemical compound [O-][N+](=O)C1=NC=CN1 YZEUHQHUFTYLPH-UHFFFAOYSA-N 0.000 description 1
- 102100022584 3-keto-steroid reductase/17-beta-hydroxysteroid dehydrogenase 7 Human genes 0.000 description 1
- 101710135980 3-keto-steroid reductase/17-beta-hydroxysteroid dehydrogenase 7 Proteins 0.000 description 1
- LCPFHXWLJMNKNC-PFEQFJNWSA-N 4-methylbenzenesulfonate;2-[4-[(3s)-piperidin-1-ium-3-yl]phenyl]indazole-7-carboxamide Chemical compound CC1=CC=C(S([O-])(=O)=O)C=C1.N1=C2C(C(=O)N)=CC=CC2=CN1C(C=C1)=CC=C1[C@@H]1CCC[NH2+]C1 LCPFHXWLJMNKNC-PFEQFJNWSA-N 0.000 description 1
- 208000003200 Adenoma Diseases 0.000 description 1
- 206010001233 Adenoma benign Diseases 0.000 description 1
- MXPOCMVWFLDDLZ-NSCUHMNNSA-N Apaziquone Chemical compound CN1C(\C=C\CO)=C(CO)C(C2=O)=C1C(=O)C=C2N1CC1 MXPOCMVWFLDDLZ-NSCUHMNNSA-N 0.000 description 1
- 238000011729 BALB/c nude mouse Methods 0.000 description 1
- 238000008673 BRACAnalysis Methods 0.000 description 1
- 206010004146 Basal cell carcinoma Diseases 0.000 description 1
- 208000003174 Brain Neoplasms Diseases 0.000 description 1
- 241000282465 Canis Species 0.000 description 1
- 206010008263 Cervical dysplasia Diseases 0.000 description 1
- 206010061809 Cervix carcinoma stage 0 Diseases 0.000 description 1
- 206010053138 Congenital aplastic anaemia Diseases 0.000 description 1
- 101710112752 Cytotoxin Proteins 0.000 description 1
- 230000007035 DNA breakage Effects 0.000 description 1
- 239000012623 DNA damaging agent Substances 0.000 description 1
- 230000004543 DNA replication Effects 0.000 description 1
- 238000009007 Diagnostic Kit Methods 0.000 description 1
- 206010012735 Diarrhoea Diseases 0.000 description 1
- 206010059866 Drug resistance Diseases 0.000 description 1
- 241000283073 Equus caballus Species 0.000 description 1
- 208000006168 Ewing Sarcoma Diseases 0.000 description 1
- 102000009095 Fanconi Anemia Complementation Group A protein Human genes 0.000 description 1
- 108010087740 Fanconi Anemia Complementation Group A protein Proteins 0.000 description 1
- 201000004939 Fanconi anemia Diseases 0.000 description 1
- 241000282324 Felis Species 0.000 description 1
- 241000282326 Felis catus Species 0.000 description 1
- 208000007569 Giant Cell Tumors Diseases 0.000 description 1
- 102100034533 Histone H2AX Human genes 0.000 description 1
- 101100437864 Homo sapiens BRCA1 gene Proteins 0.000 description 1
- 101001067891 Homo sapiens Histone H2AX Proteins 0.000 description 1
- 101001112118 Homo sapiens NADPH-cytochrome P450 reductase Proteins 0.000 description 1
- 208000037147 Hypercalcaemia Diseases 0.000 description 1
- 208000007766 Kaposi sarcoma Diseases 0.000 description 1
- 206010023774 Large cell lung cancer Diseases 0.000 description 1
- 102000003960 Ligases Human genes 0.000 description 1
- 108090000364 Ligases Proteins 0.000 description 1
- 206010027476 Metastases Diseases 0.000 description 1
- HRHKSTOGXBBQCB-UHFFFAOYSA-N Mitomycin E Natural products O=C1C(N)=C(C)C(=O)C2=C1C(COC(N)=O)C1(OC)C3N(C)C3CN12 HRHKSTOGXBBQCB-UHFFFAOYSA-N 0.000 description 1
- 241001529936 Murinae Species 0.000 description 1
- FXHOOIRPVKKKFG-UHFFFAOYSA-N N,N-Dimethylacetamide Chemical compound CN(C)C(C)=O FXHOOIRPVKKKFG-UHFFFAOYSA-N 0.000 description 1
- 206010028813 Nausea Diseases 0.000 description 1
- 206010029260 Neuroblastoma Diseases 0.000 description 1
- 241000283973 Oryctolagus cuniculus Species 0.000 description 1
- 239000012270 PD-1 inhibitor Substances 0.000 description 1
- 239000012668 PD-1-inhibitor Substances 0.000 description 1
- 208000000821 Parathyroid Neoplasms Diseases 0.000 description 1
- 241000009328 Perro Species 0.000 description 1
- 206010035226 Plasma cell myeloma Diseases 0.000 description 1
- 102000012338 Poly(ADP-ribose) Polymerases Human genes 0.000 description 1
- 108010061844 Poly(ADP-ribose) Polymerases Proteins 0.000 description 1
- 239000012979 RPMI medium Substances 0.000 description 1
- 241000700159 Rattus Species 0.000 description 1
- PLXBWHJQWKZRKG-UHFFFAOYSA-N Resazurin Chemical compound C1=CC(=O)C=C2OC3=CC(O)=CC=C3[N+]([O-])=C21 PLXBWHJQWKZRKG-UHFFFAOYSA-N 0.000 description 1
- 201000000582 Retinoblastoma Diseases 0.000 description 1
- 201000010208 Seminoma Diseases 0.000 description 1
- 206010070834 Sensitisation Diseases 0.000 description 1
- 208000021712 Soft tissue sarcoma Diseases 0.000 description 1
- 229930006000 Sucrose Natural products 0.000 description 1
- CZMRCDWAGMRECN-UGDNZRGBSA-N Sucrose Chemical compound O[C@H]1[C@H](O)[C@@H](CO)O[C@@]1(CO)O[C@@H]1[C@H](O)[C@@H](O)[C@H](O)[C@@H](CO)O1 CZMRCDWAGMRECN-UGDNZRGBSA-N 0.000 description 1
- 241000282898 Sus scrofa Species 0.000 description 1
- 229940123237 Taxane Drugs 0.000 description 1
- 208000024770 Thyroid neoplasm Diseases 0.000 description 1
- 102000004142 Trypsin Human genes 0.000 description 1
- 108090000631 Trypsin Proteins 0.000 description 1
- 208000025865 Ulcer Diseases 0.000 description 1
- 206010047700 Vomiting Diseases 0.000 description 1
- 208000008383 Wilms tumor Diseases 0.000 description 1
- MUJMYVFVAWFUJL-SNAWJCMRSA-O [(e)-4-[[4-(3-bromo-4-chloroanilino)pyrido[3,4-d]pyrimidin-6-yl]amino]-4-oxobut-2-enyl]-dimethyl-[(3-methyl-5-nitroimidazol-4-yl)methyl]azanium Chemical compound CN1C=NC([N+]([O-])=O)=C1C[N+](C)(C)C\C=C\C(=O)NC(N=CC1=NC=N2)=CC1=C2NC1=CC=C(Cl)C(Br)=C1 MUJMYVFVAWFUJL-SNAWJCMRSA-O 0.000 description 1
- 230000002159 abnormal effect Effects 0.000 description 1
- 208000017733 acquired polycythemia vera Diseases 0.000 description 1
- 230000009471 action Effects 0.000 description 1
- 230000001154 acute effect Effects 0.000 description 1
- 208000009956 adenocarcinoma Diseases 0.000 description 1
- 230000001919 adrenal effect Effects 0.000 description 1
- 230000002411 adverse Effects 0.000 description 1
- 239000002168 alkylating agent Substances 0.000 description 1
- 229940100198 alkylating agent Drugs 0.000 description 1
- 208000007502 anemia Diseases 0.000 description 1
- 229950002465 apaziquone Drugs 0.000 description 1
- 208000019493 atypical carcinoid tumor Diseases 0.000 description 1
- 230000006399 behavior Effects 0.000 description 1
- 230000008901 benefit Effects 0.000 description 1
- 230000004071 biological effect Effects 0.000 description 1
- 230000031018 biological processes and functions Effects 0.000 description 1
- 230000033228 biological regulation Effects 0.000 description 1
- 210000004369 blood Anatomy 0.000 description 1
- 239000008280 blood Substances 0.000 description 1
- 210000000988 bone and bone Anatomy 0.000 description 1
- 210000004556 brain Anatomy 0.000 description 1
- 210000000481 breast Anatomy 0.000 description 1
- 210000000621 bronchi Anatomy 0.000 description 1
- 208000035269 cancer or benign tumor Diseases 0.000 description 1
- 239000000969 carrier Substances 0.000 description 1
- 230000025084 cell cycle arrest Effects 0.000 description 1
- 230000003833 cell viability Effects 0.000 description 1
- 238000012512 characterization method Methods 0.000 description 1
- QUQKKHBYEFLEHK-QNBGGDODSA-N chembl3137318 Chemical compound CC1=CC=C(S(O)(=O)=O)C=C1.CN1N=CN=C1[C@H]([C@H](N1)C=2C=CC(F)=CC=2)C2=NNC(=O)C3=C2C1=CC(F)=C3 QUQKKHBYEFLEHK-QNBGGDODSA-N 0.000 description 1
- 238000007385 chemical modification Methods 0.000 description 1
- 239000003153 chemical reaction reagent Substances 0.000 description 1
- 210000004978 chinese hamster ovary cell Anatomy 0.000 description 1
- 230000001684 chronic effect Effects 0.000 description 1
- 238000009643 clonogenic assay Methods 0.000 description 1
- 231100000096 clonogenic assay Toxicity 0.000 description 1
- 210000001072 colon Anatomy 0.000 description 1
- 229940000425 combination drug Drugs 0.000 description 1
- 239000012141 concentrate Substances 0.000 description 1
- 238000007796 conventional method Methods 0.000 description 1
- 239000013078 crystal Substances 0.000 description 1
- 231100000433 cytotoxic Toxicity 0.000 description 1
- 230000001472 cytotoxic effect Effects 0.000 description 1
- 230000007547 defect Effects 0.000 description 1
- 230000006735 deficit Effects 0.000 description 1
- 239000008355 dextrose injection Substances 0.000 description 1
- 229940000406 drug candidate Drugs 0.000 description 1
- 230000008030 elimination Effects 0.000 description 1
- 238000003379 elimination reaction Methods 0.000 description 1
- 230000009088 enzymatic function Effects 0.000 description 1
- 208000037828 epithelial carcinoma Diseases 0.000 description 1
- WCDWBPCFGJXFJZ-UHFFFAOYSA-N etanidazole Chemical compound OCCNC(=O)CN1C=CN=C1[N+]([O-])=O WCDWBPCFGJXFJZ-UHFFFAOYSA-N 0.000 description 1
- 229950006566 etanidazole Drugs 0.000 description 1
- IDGUHHHQCWSQLU-UHFFFAOYSA-N ethanol;hydrate Chemical compound O.CCO IDGUHHHQCWSQLU-UHFFFAOYSA-N 0.000 description 1
- 235000019634 flavors Nutrition 0.000 description 1
- 235000013305 food Nutrition 0.000 description 1
- 210000000232 gallbladder Anatomy 0.000 description 1
- 201000008361 ganglioneuroma Diseases 0.000 description 1
- 239000007789 gas Substances 0.000 description 1
- 208000005017 glioblastoma Diseases 0.000 description 1
- 210000002768 hair cell Anatomy 0.000 description 1
- 210000003128 head Anatomy 0.000 description 1
- 239000003481 heat shock protein 90 inhibitor Substances 0.000 description 1
- 210000005260 human cell Anatomy 0.000 description 1
- 230000000148 hypercalcaemia Effects 0.000 description 1
- 208000030915 hypercalcemia disease Diseases 0.000 description 1
- 206010020718 hyperplasia Diseases 0.000 description 1
- 238000009169 immunotherapy Methods 0.000 description 1
- 230000001976 improved effect Effects 0.000 description 1
- 230000006872 improvement Effects 0.000 description 1
- 238000001727 in vivo Methods 0.000 description 1
- 238000011534 incubation Methods 0.000 description 1
- 230000006698 induction Effects 0.000 description 1
- 238000001990 intravenous administration Methods 0.000 description 1
- 230000009545 invasion Effects 0.000 description 1
- 210000003734 kidney Anatomy 0.000 description 1
- 230000002147 killing effect Effects 0.000 description 1
- 210000000867 larynx Anatomy 0.000 description 1
- 201000010260 leiomyoma Diseases 0.000 description 1
- 231100000518 lethal Toxicity 0.000 description 1
- 230000001665 lethal effect Effects 0.000 description 1
- 239000012669 liquid formulation Substances 0.000 description 1
- 210000004185 liver Anatomy 0.000 description 1
- 210000004072 lung Anatomy 0.000 description 1
- 201000009546 lung large cell carcinoma Diseases 0.000 description 1
- 230000000527 lymphocytic effect Effects 0.000 description 1
- 230000002101 lytic effect Effects 0.000 description 1
- 238000012423 maintenance Methods 0.000 description 1
- 238000011418 maintenance treatment Methods 0.000 description 1
- 230000036210 malignancy Effects 0.000 description 1
- 230000003211 malignant effect Effects 0.000 description 1
- 108010082117 matrigel Proteins 0.000 description 1
- 230000001404 mediated effect Effects 0.000 description 1
- 230000002503 metabolic effect Effects 0.000 description 1
- 230000004060 metabolic process Effects 0.000 description 1
- 230000009401 metastasis Effects 0.000 description 1
- 230000001394 metastastic effect Effects 0.000 description 1
- 206010061289 metastatic neoplasm Diseases 0.000 description 1
- HRHKSTOGXBBQCB-VFWICMBZSA-N methylmitomycin Chemical compound O=C1C(N)=C(C)C(=O)C2=C1[C@@H](COC(N)=O)[C@@]1(OC)[C@H]3N(C)[C@H]3CN12 HRHKSTOGXBBQCB-VFWICMBZSA-N 0.000 description 1
- 230000004048 modification Effects 0.000 description 1
- 238000012986 modification Methods 0.000 description 1
- 201000005962 mycosis fungoides Diseases 0.000 description 1
- 201000000050 myeloid neoplasm Diseases 0.000 description 1
- 230000008693 nausea Effects 0.000 description 1
- 210000003739 neck Anatomy 0.000 description 1
- 230000035407 negative regulation of cell proliferation Effects 0.000 description 1
- 201000008026 nephroblastoma Diseases 0.000 description 1
- MDJFHRLTPRPZLY-UHFFFAOYSA-N nimorazole Chemical compound [O-][N+](=O)C1=CN=CN1CCN1CCOCC1 MDJFHRLTPRPZLY-UHFFFAOYSA-N 0.000 description 1
- 229960004918 nimorazole Drugs 0.000 description 1
- 238000011580 nude mouse model Methods 0.000 description 1
- 230000008520 organization Effects 0.000 description 1
- 230000002188 osteogenic effect Effects 0.000 description 1
- 201000008968 osteosarcoma Diseases 0.000 description 1
- 230000002611 ovarian Effects 0.000 description 1
- 208000012988 ovarian serous adenocarcinoma Diseases 0.000 description 1
- 201000003709 ovarian serous carcinoma Diseases 0.000 description 1
- 210000003101 oviduct Anatomy 0.000 description 1
- 210000000496 pancreas Anatomy 0.000 description 1
- 201000002530 pancreatic endocrine carcinoma Diseases 0.000 description 1
- 229940121655 pd-1 inhibitor Drugs 0.000 description 1
- 238000009520 phase I clinical trial Methods 0.000 description 1
- 238000009521 phase II clinical trial Methods 0.000 description 1
- 208000028591 pheochromocytoma Diseases 0.000 description 1
- NLNIWCUPDREYCA-VEGRVEBRSA-N phosphono dihydrogen phosphate;(2r,3r,4r)-2,3,4,5-tetrahydroxypentanal Chemical compound OP(O)(=O)OP(O)(O)=O.OC[C@@H](O)[C@@H](O)[C@@H](O)C=O NLNIWCUPDREYCA-VEGRVEBRSA-N 0.000 description 1
- 230000026731 phosphorylation Effects 0.000 description 1
- 238000006366 phosphorylation reaction Methods 0.000 description 1
- 229950010456 pimonidazole Drugs 0.000 description 1
- 239000004033 plastic Substances 0.000 description 1
- 230000005731 poly ADP ribosylation Effects 0.000 description 1
- 208000037244 polycythemia vera Diseases 0.000 description 1
- 238000006116 polymerization reaction Methods 0.000 description 1
- 239000000244 polyoxyethylene sorbitan monooleate Substances 0.000 description 1
- 235000010482 polyoxyethylene sorbitan monooleate Nutrition 0.000 description 1
- 229920000053 polysorbate 80 Polymers 0.000 description 1
- 229940068968 polysorbate 80 Drugs 0.000 description 1
- 229950004406 porfiromycin Drugs 0.000 description 1
- 208000030266 primary brain neoplasm Diseases 0.000 description 1
- 238000012545 processing Methods 0.000 description 1
- 230000002062 proliferating effect Effects 0.000 description 1
- 210000002307 prostate Anatomy 0.000 description 1
- 230000005855 radiation Effects 0.000 description 1
- 238000001959 radiotherapy Methods 0.000 description 1
- 239000002994 raw material Substances 0.000 description 1
- 239000000376 reactant Substances 0.000 description 1
- 102000005962 receptors Human genes 0.000 description 1
- 108020003175 receptors Proteins 0.000 description 1
- 210000000664 rectum Anatomy 0.000 description 1
- 230000010076 replication Effects 0.000 description 1
- 210000001995 reticulocyte Anatomy 0.000 description 1
- 238000012552 review Methods 0.000 description 1
- 201000009410 rhabdomyosarcoma Diseases 0.000 description 1
- 150000003839 salts Chemical class 0.000 description 1
- 238000012216 screening Methods 0.000 description 1
- 230000008313 sensitization Effects 0.000 description 1
- 230000001568 sexual effect Effects 0.000 description 1
- 210000003491 skin Anatomy 0.000 description 1
- 201000000849 skin cancer Diseases 0.000 description 1
- 206010040882 skin lesion Diseases 0.000 description 1
- 231100000444 skin lesion Toxicity 0.000 description 1
- 150000003384 small molecules Chemical class 0.000 description 1
- 239000012453 solvate Substances 0.000 description 1
- 230000000392 somatic effect Effects 0.000 description 1
- 241000894007 species Species 0.000 description 1
- 230000006641 stabilisation Effects 0.000 description 1
- 238000011105 stabilization Methods 0.000 description 1
- 210000002784 stomach Anatomy 0.000 description 1
- 201000000498 stomach carcinoma Diseases 0.000 description 1
- 238000003860 storage Methods 0.000 description 1
- 239000000126 substance Substances 0.000 description 1
- 239000005720 sucrose Substances 0.000 description 1
- 230000004083 survival effect Effects 0.000 description 1
- 208000024891 symptom Diseases 0.000 description 1
- 229940124652 talazoparib tosylate Drugs 0.000 description 1
- 230000008685 targeting Effects 0.000 description 1
- DKPFODGZWDEEBT-QFIAKTPHSA-N taxane Chemical class C([C@]1(C)CCC[C@@H](C)[C@H]1C1)C[C@H]2[C@H](C)CC[C@@H]1C2(C)C DKPFODGZWDEEBT-QFIAKTPHSA-N 0.000 description 1
- 229940126585 therapeutic drug Drugs 0.000 description 1
- 210000001685 thyroid gland Anatomy 0.000 description 1
- 231100000331 toxic Toxicity 0.000 description 1
- 230000002588 toxic effect Effects 0.000 description 1
- 230000001988 toxicity Effects 0.000 description 1
- 231100000419 toxicity Toxicity 0.000 description 1
- 238000012546 transfer Methods 0.000 description 1
- 238000011269 treatment regimen Methods 0.000 description 1
- 230000001960 triggered effect Effects 0.000 description 1
- 239000012588 trypsin Substances 0.000 description 1
- 231100000402 unacceptable toxicity Toxicity 0.000 description 1
- 241001529453 unidentified herpesvirus Species 0.000 description 1
- 238000012795 verification Methods 0.000 description 1
- 239000012905 visible particle Substances 0.000 description 1
- 230000008673 vomiting Effects 0.000 description 1
- 230000003442 weekly effect Effects 0.000 description 1
Images
Classifications
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K31/00—Medicinal preparations containing organic active ingredients
- A61K31/66—Phosphorus compounds
- A61K31/675—Phosphorus compounds having nitrogen as a ring hetero atom, e.g. pyridoxal phosphate
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P35/00—Antineoplastic agents
Definitions
- the invention relates to a method for treating cancer, especially a method for treating cancer patients resistant to PARP inhibitors (PARPi).
- PARP inhibitors PARPi
- PARPi drug Olaparib The first human clinical trial of PARPi drug Olaparib confirmed for the first time that PARPi can inhibit the growth of tumor cells carrying BRCA1/2 mutations, which is mainly based on the theory of synthetic lethality (Ashworth, A., & Lord, CJ (2018 ).Synthetic lethal therapies for cancer: what's next after PARP inhibitors?.Nature rev iews.Clinical oncology,15(9),564–576.https://doi.org/10.1038/s41571-018-0055-6): PARP inhibitors can inhibit the DNA single-strand damage repair function of PARP, resulting in a large number of single-strand DNA damage in cells that cannot be repaired in time.
- Double-strand DNA damage with strong cytotoxicity can be mediated by BRCA1 and BRCA2 proteins in normal cells.
- BRCA1/2-deficient tumor cells double-strand DNA damage cannot be repaired, resulting in the eventual death of tumor cells.
- PARPi was originally developed for radiotherapy and chemotherapy sensitization, and preclinical studies support that PARPi can be developed as a single drug for the treatment of BRCA1/2 gene deficient cancers. Therefore, the initial target population for PA RPi-BRCA hypothesis verification was selected as carriers of BRCA1/2 germline mutation (gBRCA1/2).
- Niraparib and Rucaparib Two other PARPi have been approved in ovarian cancer: Niraparib and Rucaparib: FDA and EMA approve Niraparib maintenance regimen (regardless of BRCA1/2 status); Rucaparib It is also registered by the FDA and EMA as an optional treatment option for patients with BRCA1/2 mutation-related ovarian cancer who have previously received two lines of chemotherapy; and talazoparib has also been approved by the FDA for the treatment of BRCA Mutant/HER-2-negative metastatic breast cancer (Mateo, J., Lord, CJ, Serr a, V., Tutt, A., J., Castroviejo-Bermejo, M., Cruz, C., Oaknin, A., Kaye, SB, & de Bono, JS (2019). A decade of clinical development of PARP inhibitors in perspective. Annals of oncology: official journal of the European Society for Medical Oncology, 30(9), 1437–1447. https://doi.org/10.1093/annonc/md
- TH-302 (Evofosfamide, cas number 918633-87-1) is a 2-nitroimidazole-triggered hypoxia-activated prodrug (HAP) bromoifosfamide developed by Threshold Corporation of the United States. Under hypoxia, the inactive TH-302 prodrug released highly toxic Br-IPM. TH-302 has broad-spectrum biological activity in vivo and in vitro, specific hypoxia selective activation activity, induction of H2AX phosphorylation and DNA cross-linking activity, resulting in cell cycle arrest, so this compound has been developed by many pharmaceutical companies and scientific research institutes Development of anticancer drugs.
- HAP hypoxia-activated prodrug
- TH-302 has broad-spectrum activity against various tumors, and has excellent hypoxia-selective activity enhancement effect.
- Cytochrome P450 oxidoreductase reduces the prodrug TH-302 to obtain intermediate free radical anion, and then the free radical anion is unstable and decomposed into cytotoxic cytotoxin Br-IPM to play a role.
- the key step in this step is the one-electron reduction process. Studies have confirmed that the presence of oxygen will reverse the one-electron reduction process. It can be restored to produce stronger cytotoxicity.
- DNA repair mutant cell lines based on Chinese hamster ovary cells including cell lines lacking base excision, nucleotide excision, non-homologous end-joining repair or homologous end-joining repair (the cell line is a cell line lacking homologous source-dependent repair cell line) to test the in vitro cytotoxicity of TH-302.
- the study found that cell lines lacking homologous end-joining repair alone or combined with nucleotide excision were significantly more sensitive to TH-302 hypoxia. However, cell lines with defective base excision, nucleotide excision, or non-homologous end-joining repair alone had no effect on TH-302 sensitivity.
- H460 cell line human large cell lung cancer cells
- HCT116 cell line human colon cancer cell
- A375 cell line human malignant melanoma cells
- hypoxia targeting compositions and combinations thereof with a parp inhibitor and methods of use thereof it is pointed out that the use of hypoxia-activated drugs or their prodrugs (such as apaziquone, AQ4N, etanidazole, evofosfamide (TH-302), nimorazole, pimonidazole, porfiromycin, PR-104, tarloxotinib, tirapazamine (tirapazamine)) and PARPi have an additive effect when combined with PARPi.
- hypoxia-activated drugs or their prodrugs such as apaziquone, AQ4N, etanidazole, evofosfamide (TH-302), nimorazole, pimonidazole, porfiromycin, PR-104, tarloxotinib, tirapazamine (tirapazamine)
- PARPi have an additive effect when combined with PARPi.
- TH-302 single drug can have excellent therapeutic effect on PARPi-resistant cancer models!
- the present application provides the following cancer treatment methods.
- Therapeutic method which uses the drug containing the hypoxia-activated compound of formula (1) as a single drug or in combination to treat PARPi-resistant cancer and tumor patients:
- each R is independently selected from H, -CH 3 , -CH 2 CH 3
- each X is independently selected from Cl, Br, MsO, TsO and other leaving functional groups.
- Therapeutic method which uses the drug containing the hypoxia-activated compound of formula (1) in combination with PARP inhibitors to treat cancer and tumor patients resistant to PARP inhibitors:
- each R is independently selected from H, -CH 3 , -CH 2 CH 3
- each X is independently selected from Cl, Br, MsO, TsO and other leaving functional groups.
- the medicines mentioned herein refer to medicines or preparations, the prepared medicines contain the hypoxia-activated compound of formula (1) or their salts or solvates as active ingredients in a specific dose range, and/or the prepared medicines are specific dosage ranges. Dosage form, specific mode of administration.
- the prepared medicines, medicaments and preparations may also contain pharmaceutically acceptable auxiliary materials or excipients.
- the medicine can be in any dosage form for clinical application, such as tablet, suppository, dispersible tablet, enteric-coated tablet, chewable tablet, orally disintegrating tablet, capsule, sugar-coated agent, granule, dry powder, oral solution, small needle for injection , Freeze-dried powder for injection or large infusion.
- the pharmaceutically acceptable adjuvant or excipient in the medicine may include one or more of the following: diluent, solubilizer, disintegrant, suspending agent, lubricant, viscose Mixtures, fillers, flavoring agents, sweeteners, antioxidants, surfactants, preservatives, coating agents, and pigments, etc.
- Threshold related patents: WO2010048330A1, WO2012142520A2, and WO2008083101A1.
- Threshold related patents: WO2010048330A1, WO2012142520A2, and WO2008083101A1.
- the present invention will The text of the above application is incorporated in its entirety.
- TH-302 or its similar compounds It is a DNA alkylating agent anticancer drug with a wide range of cancer treatment potentials.
- Cancer refers to leukemias, lymphomas, carcinomas and other malignancies (including solid tumors) of potentially unrestricted growth that can spread locally by invasion and systemically by metastasis.
- cancers that can be treated include, but are not limited to, adrenal, bone, brain, breast, bronchi, colon and/or rectum, gallbladder, head and neck, kidney, larynx, liver, lung, nervous tissue, pancreas, prostate, Cancer of the parathyroid, skin, stomach, and thyroid.
- cancers include acute and chronic lymphocytic and granulocytic neoplasms, adenocarcinoma, adenoma, basal cell carcinoma, cervical dysplasia and carcinoma in situ, Ewing's sarcoma, epidermoid carcinoma, giant cell tumor, multiple Type glioblastoma, hair cell tumor, enteric ganglioneuroma, proliferative corneal neuroma, islet cell carcinoma, Kaposi's sarcoma, leiomyoma, leukemia, lymphoma, malignant carcinoid tumor, malignant melanoma , malignant hypercalcemia, marfanoid tumor, medullary epithelial carcinoma, metastatic skin cancer, mucosal neuroma, myeloma, mycosis fungoides, neuroblastoma, osteosarcoma, osteogenic and other sarcomas, Ovarian tumor, pheochromocytoma, polycythemia
- PARP is an enzyme, the full name is polyadenylate diphosphate-ribose polymerase (Poly ADP-ribose Polymerase, PARP).
- PARP is a DNA repair enzyme that plays a key role in the DNA repair pathway. PARP is activated when DNA is damaged and broken. As a molecular sensor of DNA damage, it has the function of recognizing and binding to the position of DNA breakage, and then activates and catalyzes the poly ADP-ribosylation of receptor proteins to participate in the DNA repair process. .
- PARP inhibitors inhibit the work of PARP enzymes, so that these PARP enzymes, which are equivalent to "repairmen", cannot work normally. If DNA damage is not repaired, cells will die.
- PARP inhibitors are used to specifically enter cancer cells, the PARP enzyme activity is inhibited and cannot function normally, and the DNA of cancer cells cannot be repaired. In this way, PARP inhibitors can only kill cancer cells but not normal cells.
- Synthetic lethality means that when two different genes (BRCA) or protein (PRAP) change at the same time, it will lead to cell death. However, if only one of these two genes/proteins is abnormal, it will not cause cell death.
- PARP inhibitors are compounds that inhibit PARP enzymes, that is, all substances that can inhibit the activity of PARP enzymes are PARP inhibitors.
- the PARP inhibitor here essentially refers to the drug containing the active ingredient of PARP inhibitor.
- Talazoparib indicated for adults with deleterious or suspected deleterious germline BRCA-mutated (gBRCAm) HER2-negative locally advanced or metastatic breast cancer.
- the commercially available dosage form is 0.25 mg/1 mg talazoparib tosylate capsules, 1 mg orally once a day, considering treatment interruption or dose reduction in case of adverse reactions:
- the oral dose is reduced to 0.75mg (three 0.25mg capsules), once a day;
- the oral dose is reduced to 0.5mg (two 0.25mg capsules), once a day;
- the oral dose was reduced to 0.25 mg (one 0.25 mg capsule), once a day.
- Niraparib for the maintenance treatment of adult patients with platinum-sensitive recurrent epithelial ovarian cancer, fallopian tube cancer or primary peritoneal cancer after complete remission or partial remission of platinum-containing chemotherapy.
- the commercially available dosage form is 100mg Niraparib Tosylate Capsules, 300mg orally once a day, until disease progression or intolerable adverse reactions occur. In the case of adverse reactions, consider treatment interruption or dose reduction:
- a second dose reduction can be made, from 2 capsules (200 mg) per day to 1 capsule per day (100 mg);
- Rucaparib for women with advanced ovarian cancer whose tumors carry a specific gene mutation (harmful BRCA) and who have been treated with two or more chemotherapy drugs.
- the commercially available dosage forms are tablets: 200mg, 250mg and 300mg, three specifications.
- the recommended dose is 600 mg orally twice daily with or without food. Continue treatment until disease progression or unacceptable toxicity. For adverse reactions, consider interruption of treatment or dose reduction.
- Olaparib for treatment-na ⁇ ve adult patients with germline or somatic BRCA-mutated (gBRCAm or sBRCAm) advanced epithelial ovarian, fallopian tube, or primary peritoneal cancer in complete or partial response to platinum-based chemotherapy Maintenance therapy after chemotherapy; maintenance therapy for adult patients with platinum-sensitive recurrent epithelial ovarian cancer, fallopian tube cancer or primary peritoneal cancer after complete remission or partial remission of platinum-based chemotherapy.
- the commercially available dosage forms are tablets: 150mg and 100mg, two specifications. The recommended dose is 300 mg (two 150 mg tablets) twice daily, equivalent to a total daily dose of 600 mg. 100mg tablet for dose reduction use:
- treatment interruption or dose reduction may be considered;
- the recommended dose is reduced to 250 mg (one 150 mg tablet, one 100 mg tablet), taken twice a day (equivalent to a total daily dose of 500 mg);
- the recommended dose is reduced to 200 mg (two 100 mg tablets) taken twice daily (equivalent to a total daily dose of 400 mg).
- Fluzoparib for the treatment of patients with platinum-sensitive recurrent ovarian cancer, fallopian tube cancer or primary peritoneal cancer with germline BRCA mutation (gBRCAm) who have previously received second-line or more chemotherapy.
- Commercially available capsules 50mg specification.
- Weekly doses of 480 mg/m 2 to about 670 mg/m 2 or, for example, 575 mg/m 2 are administered intravenously.
- TH-302 (Concentrate for Administration Solution) for clinical trials is a sterile liquid formulation of TH-302.
- TH-302 was formulated with 70% absolute ethanol, 25% dimethylacetamide and 5% polysorbate 80. It is supplied by the sponsor in a 10 mL glass vial with a rubber stopper and flip-off closure.
- TH-302 drug product is a clear, colorless to pale yellow solution substantially free of visible particles.
- each single-use vial contains TH-302 drug product in a nominal fill volume of 6.5 mL (equivalent to 100 mg/mL) and is clearly labeled as The batch number, route of administration, required storage conditions, name of the sponsor and appropriate precautionary labels required by applicable regulations are disclosed. It needs to be diluted according to the pharmacy manual before administration.
- TH-302 Dilute to a total volume of 500 mL (1000 mL for a total dose > 1000 mg) with commercially available 5% dextrose in water before administration to obtain the desired final concentration.
- Each dose of TH-302 was prepared with 5% dextrose in water without bis(2-ethylhexyl) phthalate (DEHP-free) and administered intravenously using a DEHP-free IV administration set.
- lyophilized preparations developed by Threshold can also be used:
- a solution (20 mL) of TH-302 (100 mg) and sucrose (1 g) was added to a lyophilized vial and lyophilized to produce a lyophilized unit dosage form of TH-302 with a drug loading of less than 5 mg/cm 3 .
- the unit dosage form is dissolved in 5% dextrose injection, and an appropriate amount of the solution is administered to the patient;
- TH-302 lyophilized preparations for injection are prepared in 100mL glass vials with a drug loading of 100mg/100ml. Store under controlled conditions, inject 250mL of 5% glucose injection into a freeze-dried preparation bottle when used, and infuse intravenously within 30 minutes through an infusion pump.
- Monotherapy that is, monotherapy.
- Combined use that is, combined drug therapy.
- Monotherapy refers to the use of only one anticancer drug in a course of treatment.
- Combination therapy refers to the simultaneous or successive use of two or more anticancer drugs in one course of treatment.
- combination therapy needs to explore different dosages and administration cycles according to the characteristics of the disease and the types of drugs used in combination. Only based on the above conditions can the combination drug treatment plan obtained through exploration be able to achieve better therapeutic effects than single drug therapy.
- the drug dosage and dosage cycle of the single drug and combined treatment regimens need to be obtained through clinical trials with reference to the dosage and dosage regimen of TH-302 and its similar compounds and PARPi mentioned above.
- damaged DNA repair enzymes are selected from:
- Nucleotide excision repair enzyme (nucleotide excision repair) damage
- base excision repair enzyme base excision repair
- Mismatch repair enzyme mismatch repair
- Impairment of one or more of the repair enzymes in the Fanconi's anemia pathway is Impairment of one or more of the repair enzymes in the Fanconi's anemia pathway.
- any one or more of damaged homologous recombination DNA repair enzymes, damaged nucleotide excision repair enzymes, and damaged base excision repair enzymes more preferably a single damaged homologous recombination DNA repair enzyme Or both homologous recombination DNA repair enzyme damage and nucleotide excision repair enzyme damage.
- the patient's tumor or cancer tissue is detected to have any one or two gene mutations in the genes corresponding to BRCA1 and BRCA2; or the patient is detected to have any one of the genes corresponding to BRCA1 and BRCA2 A genetic mutation or both genetic mutations.
- BRACAnalysis CDx a companion detection kit for Olaparib, is used to detect BRCA gene mutations in blood samples of ovarian cancer patients;
- BRCA1/2 Gene Mutation Detection Kit (Combined Probe-Anchor Polymerization Sequencing Method), which is used to detect germline mutations in exon regions and adjacent intron regions of BRCA1/2 genes in clinically diagnosed ovarian cancer and breast cancer patients carry out qualitative testing;
- Human BRCA1 gene and BRCA2 gene mutation detection kit (reversible terminal termination sequencing method), which can be used for the relevant medication guidance of PARP inhibitor olaparib tablets.
- BRCA1 and BRCA2 mutations include germline mutations (gBRCAm) and systemic mutations (sBRCAm) of BRCA1 and BRCA2 mutations.
- hypoxia-activating compound of formula (1) is selected from compounds of the following structures:
- TH-302 is preferred.
- the cancer and tumor are selected from ovarian cancer, breast cancer, pancreatic cancer, fallopian tube cancer, primary peritoneal cancer, gastric cancer, prostate cancer, lung cancer, liver cancer, colon cancer, rectal cancer, bladder cancer, etc.
- the lung cancer is preferably Non-small cell lung cancer, small cell lung cancer.
- Therapeutic method uses the drug monotherapy that contains the hypoxia activation compound of following formula to treat Olaparib Olaparib resistant ovarian cancer, breast cancer, pancreatic cancer, fallopian tube cancer, primary peritoneal cancer, gastric cancer, prostate cancer, non Patients with small cell lung cancer, small cell lung cancer, liver cancer, colon cancer, rectal cancer, bladder cancer:
- the patient's tumor or cancer tissue is detected to have any one or two gene mutations in the genes corresponding to BRCA1 and BRCA2; or the patient is detected to have any one of the genes corresponding to BRCA1 and BRCA2 A genetic mutation or both genetic mutations.
- Therapeutic method which uses the drug containing the hypoxia activation compound of the following formula in combination with Olaparib to treat Olaparib-resistant ovarian cancer, breast cancer, pancreatic cancer, fallopian tube cancer, primary peritoneal cancer, gastric cancer , prostate cancer, non-small cell lung cancer, small cell lung cancer, liver cancer, colon cancer, rectal cancer, bladder cancer patients:
- the patient's tumor or cancer tissue is detected to have any one or two gene mutations in the genes corresponding to BRCA1 and BRCA2; or the patient is detected to have any one of the genes corresponding to BRCA1 and BRCA2 A genetic mutation or both genetic mutations.
- a method of treatment comprising the following steps:
- the drug containing the hypoxia-activating compound of formula (1) is used for treatment alone or in combination with PARP inhibitors:
- each R is independently selected from H, -CH 3 , -CH 2 CH 3
- each X is independently selected from Cl, Br, MsO, TsO and other leaving functional groups.
- the TMB (Tumor Mutation Burden) level of the gene mutation is medium.
- hypoxia-activated compound of formula (1) in the preparation of drugs for the treatment of cancer in patients as a single drug or in combination with PARP inhibitors:
- the patient is a PARP inhibitor-resistant patient
- Each R is independently selected from H, -CH 3 , -CH 2 CH 3 , and each X is independently selected from Cl, Br, MsO, TsO and other leaving functional groups.
- the patient has impaired DNA repair enzymes
- the patient's tumor or cancer tissue is detected to have any one or two gene mutations in the genes corresponding to BRCA1 and BRCA2; or
- the patient is detected to have any one gene mutation or two gene mutations in the genes corresponding to BRCA1 and BRCA2.
- BRCA1 and BRCA2 mutations include germline mutations (gBRCAm) and systemic mutations (sBRCAm) of BRCA1 and BRCA2 mutations.
- hypoxia-activating compound of the formula (I) is selected from compounds of the following structures:
- the PARP inhibitor is selected from the group consisting of Olaparib, Rucaparib, Niraparib, Talazoparib, Fluzoparib, Pamiparib; or
- the cancer and tumor are selected from ovarian cancer, breast cancer, pancreatic cancer, fallopian tube cancer, primary peritoneal cancer, gastric cancer, prostate cancer, non-small cell lung cancer, small cell lung cancer, liver cancer, colon cancer, rectal cancer, and bladder cancer; or
- the TMB (Tumor Mutation Burden) level of the gene mutation is moderate.
- TMB Tumor mutation load (burden) is tumor gene mutation load
- TMB Tumor mutation load
- Mb represents every million bases
- CheckMate-032 This is a Phase II clinical trial of 401 advanced lung cancer patients who failed first-line treatment, receiving PD-1 inhibitors alone or in combination with Yipimu.
- TMB Tumor mutation load
- the effective rates of the three groups are 62%, 20%, and 23%, respectively, and the effective rate of the high TMB group is 3 times; and the median overall survival of the three groups were: 22.0 months, 3.6 months, 3.4 months - 22.0 months and 3.4 months, a difference of 6 times!
- the present invention also provides a drug for treating PARP inhibitor-resistant cancer and tumor patients, the drug contains the hypoxia-activating compound of formula (I), and the drug can be used alone or in combination to treat PARP inhibitor-resistant cancer , Tumor patients:
- each R is independently selected from H, -CH 3 , -CH 2 CH 3
- each X is independently selected from Cl, Br, MsO, TsO and other leaving functional groups.
- said patient has impaired DNA repair enzymes; or
- the patient's tumor or cancer tissue is detected to have any one or two gene mutations in the genes corresponding to BRCA1 and BRCA2; or
- the patient is detected to have any one gene mutation or two gene mutations in the genes corresponding to BRCA1 and BRCA2.
- the BRCA1 and BRCA2 mutations include germline mutations (gBRCAm) and systemic mutations (sBRCAm) of BRCA1 and BRCA2 mutations.
- hypoxia-activating compound of formula (1) is selected from compounds of the following structures:
- the PARP inhibitor is selected from the group consisting of Olaparib, Rucaparib, Niraparib, Talazoparib, Fluzoparib, Pamiparib; or
- the cancer and tumor are selected from ovarian cancer, breast cancer, pancreatic cancer, fallopian tube cancer, primary peritoneal cancer, gastric cancer, prostate cancer, non-small cell lung cancer, small cell lung cancer, liver cancer, colon cancer, rectal cancer, and bladder cancer; or
- the TMB (Tumor Mutation Burden) level of the gene mutation is moderate.
- the above-mentioned medicine should also add pharmaceutically acceptable auxiliary materials or excipients according to the characteristics of medicines, medicines and preparations.
- the medicine can be in any dosage form for clinical application, such as tablet, suppository, dispersible tablet, enteric-coated tablet, chewable tablet, orally disintegrating tablet, capsule, sugar-coated agent, granule, dry powder, oral solution, small needle for injection , Freeze-dried powder for injection or large infusion.
- the pharmaceutically acceptable adjuvant or excipient in the medicine may include one or more of the following: diluent, solubilizer, disintegrant, suspending agent, lubricant, viscose Mixtures, fillers, flavoring agents, sweeteners, antioxidants, surfactants, preservatives, coating agents, and pigments, etc.
- the above drugs can be used alone or in combination with PARPi drugs for treatment.
- Fig. 1 is the inhibitory rate curve of compound TH-302 and tirapazamine (tirapazamine) to Capan-1 cell line under normoxic and hypoxic conditions, wherein, con.Log (nM) represents the concentration value under the unit of nmol/L The logarithm value with base 10, inhibition represents inhibition rate;
- Fig. 2 is the inhibitory rate curve of compound TH-302 and tirapazamine (tirapazamine) to Capan-1 cell line under normoxic and hypoxic conditions, wherein, con.Log (nM) represents the concentration value under the unit of nmol/L The logarithm value with base 10, inhibition represents inhibition rate;
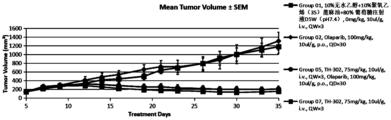
- Fig. 3 is the growth curve of the tumor volume of mice in each group in the subcutaneous model of human pancreatic cancer Capan-1;
- Figure 4 is a graph showing the relative tumor inhibition rates of mice in each group in the subcutaneous model of human pancreatic cancer Capan-1;
- Figure 5 is a graph of the body weight of mice in each group in the subcutaneous model of human pancreatic cancer Capan-1;
- Figure 6 is a graph showing the percent change in body weight of mice in each group in the subcutaneous model of human pancreatic cancer Capan-1;
- Fig. 7 is the growth curve of the tumor volume of mice in each group in the pancreatic cancer Capan-1CDX model
- Figure 8 is a graph showing the change in body weight over time in each treatment group in the pancreatic cancer Capan-1CDX model
- Figure 9 is a growth curve of the tumor volume of each group of mice in the lung cancer LU6429 PDX model
- Figure 10 is a graph showing the body weight of each treatment group over time in the lung cancer LU6429 PDX model
- Figure 11 is a growth curve of the tumor volume of each group of mice in the bladder cancer BL3325 PDX model
- Figure 12 is a graph showing the change in body weight over time in each treatment group in the bladder cancer BL3325 PDX model.
- a patient and “individual” are used interchangeably and refer to a mammal in need of cancer treatment.
- the patient is a human.
- the patient is a human being diagnosed with cancer.
- a "patient” or “individual” may refer to a non-human mammal, such as a non-human primate, dog, cat, rabbit, pig, mouse, for screening, characterization and evaluation of drugs and therapies or rats.
- Prodrug refers to a compound (or drug) that, upon administration or administration, is metabolized or otherwise converted to a biologically active or more active compound (or drug) with respect to at least one property.
- Prodrugs are chemically modified relative to the drug in such a way that they are less or inactive relative to the drug, but the chemical modification allows the corresponding drug to be produced by metabolism or other biological processes after administration of the prodrug.
- Prodrugs may have altered metabolic stability or delivery characteristics, fewer side effects or toxicity, or improved flavor relative to the active drug.
- Prodrugs can be synthesized using reactants other than the corresponding drug.
- Treating or “treating a patient” refers to administering, using or administering to a patient a therapeutically effective amount of a drug relevant to the present invention.
- administering or “administering” "using" a drug to a patient refers to direct administration or administration (which may be administered or administered to the patient by a medical professional or may be self-administered or administered) and/or indirect administration or administration, which may prescribe the drug the behavior of.
- direct administration or administration which may be administered or administered to the patient by a medical professional or may be self-administered or administered
- indirect administration or administration which may prescribe the drug the behavior of.
- a physician who instructs a patient to self-administer or administer a drug and/or provides a prescription for the drug to the patient is administering or administering the drug to the patient.
- a "therapeutically effective amount" of a drug means that when administered or administered or used to a patient with cancer, it will have a desired therapeutic effect (such as alleviation, improvement, remission or elimination of the clinical manifestations of one or more cancers in the patient) amount of drug.
- a therapeutic effect does not necessarily occur through the administration or administration of one dose, and may only occur after the administration or administration of a series of doses. Thus, a therapeutically effective amount may be administered or administered in one or more doses.
- Treatment of a condition or patient refers to steps taken to obtain beneficial or desired results (including clinical results).
- beneficial or desired clinical outcomes include, but are not limited to, alleviation or amelioration of one or more symptoms of cancer; reduction in extent of disease; delay or slowing of disease progression; amelioration, remission or stabilization of disease state; or other beneficial outcomes.
- treatment of cancer can result in a partial response or stable disease.
- Tumor cell refers to a tumor cell of any appropriate species (eg, a mammal such as murine, canine, feline, equine or human).
- TH-302 is a small molecule prodrug that can be activated under hypoxic conditions and release cytotoxins to kill tumor cells and tumor tissues.
- DLD1 human colon cancer cell line
- BRCA2 knockout DLD1-BRCA2-/- tumor cell line to detect Whether there is any difference in the killing ability of TH-302 to the above two tumor cell lines with or without BRCA2 protein expression.
- This clonogenic assay uses IC 90 (90% inhibitory concentration) value as an evaluation of the ability of compound TH-302 to kill cells.
- the specific experimental method is as follows:
- DLD1 and DLD1-BRCA2-/- cells were cultured in RPMI medium, added with 10% FBS and 1% double antibody, and cultured at 37°C and 5% CO 2 .
- the cells are routinely cultured until the cell saturation is 80%-90%, and the cells are collected when the number reaches the requirement.
- the cells were cultured in a 37°C, 5% CO 2 incubator for 2 days.
- the cells were resuspended with 3 mL of culture medium and seeded in a 6-well culture plate, wherein the density of DLD1-BRCA2-/- cells was 2000/well, and the density of DLD1 cells was 300/well.
- the IC 90 values of the test compound TH-302 in the two cells measured by the above-mentioned experimental method are listed in Table 1 below.
- Table 1 IC 90 data of the inhibitory effect of compound TH-302 on two tumor cell lines under hypoxic conditions
- the applicant specifically studied the in vitro cell proliferation inhibition experiments of hypoxia-activated anticancer prodrugs TH-302 and tirapazamine in Capan-1 and BxPc-3 cell lines under normoxia and hypoxia conditions, respectively.
- the Capan-1 cell line is a BRCA mutant cell line
- the BxPc-3 cell line is a BRCA wild-type cell line, that is, a non-BRCA mutant cell line.
- This experiment was used to verify the difference in the sensitivity of TH-302 and tirapazamine, which are both hypoxia-activated anticancer prodrugs, to BRCA mutations.
- hypoxic workstation Adjusts the hypoxic workstation to an anaerobic environment (O 2 ⁇ 0.01%) and confirm hypoxia in the workstation using an oxygen indicator.
- the 24-well plate with glass inserts was sent to the hypoxic workstation.
- the IC 50 of TH-302 in the BRCA mutant Capan-1 cell line was 0.82 ⁇ M
- the IC 50 in the BRCA wild-type BxPc-3 cell line was 3.07 ⁇ M.
- the difference is 3.7 times, indicating that BRCA mutation makes TH-302 have stronger proliferation inhibitory activity on tumor cell lines, that is, BRCA mutation will enhance the sensitivity of tumor cells to TH-302 drug.
- Tirapazamine which is also a hypoxia-activated anticancer prodrug, had an IC 50 of 29.06 ⁇ M in the BRCA-mutated Capan-1 cell line under hypoxic conditions, and an IC 50 of 29.06 ⁇ M in the BRCA wild-type BxPc-3 cell line.
- the IC 50 was 33.23 ⁇ M, with a difference of 1.1 times, and there was no significant difference, which indicated that tirapazamine had no correlation with BRCA mutations, that is, BRCA mutations did not significantly affect the proliferation inhibitory activity of tirapazamine on tumor cell lines, that is, BRCA The mutation did not enhance the sensitivity of tumor cells to the drug tirapazamine.
- Capan-1 CDX model is an olaparib-resistant model with a BRCA2 pathogenic variant.
- Each BALB/c female nude mouse was inoculated subcutaneously with 5 ⁇ 10 5 Capan-1 cells on the lower back of the right side, and the cells were resuspended in 1:1 PBS and Matrigel (0.1ml/mouse), and a total of 64 female mice were inoculated .
- the date of inoculation is June 23, 2021.
- the average tumor volume is 140mm 3 , the patients will be randomly divided into groups according to tumor size.
- test drug Olaparib 100mg/kg single drug group (Group 2), TH-302 75mg/kg and Olaparib 100mg/kg combined administration group (Group 5), TH-302 75mg/kg single drug group (Group 7) And 10% absolute ethanol + 10% polyoxyethylene (35) castor oil + 80% glucose injection D5W (pH7.4) vehicle control group, a total of 7 groups, 6 mice in each group.
- vehicle control group, TH-302 single drug and combined drug groups were given tail vein administration once a week for a total of three weeks; the test drug Olaparib group was administered orally by gavage once a day for a total of three weeks. Medicine for 30 days.
- the curative effect is evaluated according to the relative tumor inhibition rate TGI (%), and the safety evaluation is carried out according to the animal body weight change and death.
- the test drug Olaparib 100mg/kg (Group 2) treatment group had no tumor inhibitory effect on the 35th day after tumor cell inoculation, and the relative tumor inhibition rate TGI (%) was -7.1%, which was not statistically significantly different from the control group (p>0.05).
- the combined treatment group (Group 5) of Olaparib 100mg/kg and TH-302 75mg/kg had a significant tumor inhibitory effect on the 35th day after tumor cell inoculation, and there was a statistically significant difference compared with the control group (p ⁇ 0.001 ), the relative tumor inhibition rate TGI (%) was 84.47%.
- TH-302 75mg/kg monotherapy group (Group 7) showed a significant tumor inhibitory effect on the 35th day after tumor cell inoculation (Day 35), which was statistically significantly different from the control group (p ⁇ 0.001 ), the relative tumor inhibition rate TGI (%) was 87.66%.
- TGI tumor inhibition rate
- mice The tumor volumes of mice in different groups were measured on different days, and the average values were obtained. The results are shown in Table 7 below.
- Table 7 Changes in tumor volume of mice in each group with treatment time in the Capan-1 model of human pancreatic cancer (unit: mm 3 )
- Table 8 Drug efficacy analysis table of each group in the subcutaneous model of human pancreatic cancer Capan-1
- T/C% Relative tumor proliferation rate
- T and C are the average relative tumor volume (RTV) of the treatment group and the control group at a specific time point, respectively.
- Table 9 Relative tumor inhibition rates of tumors in each group in the human pancreatic cancer Capan-1 subcutaneous model
- mice The body weights of mice in different groups were measured on different days, and the average values were obtained. The results are shown in Table 10 below.
- Table 10 Body weight of mice on different days of inoculation in the subcutaneous animal model experiment of human pancreatic cancer Capan-1 treated with drugs
- Figure 5 is, the body weight curves of mice in each group in the subcutaneous model of human pancreatic cancer Capan-1.
- Capan-1 CDX model is indeed an Olaparib-resistant model, and Olaparib has no inhibitory effect on the tumor growth of this model, that is, it is resistant to Olaparib;
- TH-302 monotherapy has a good therapeutic effect on Olaparib-resistant pancreatic cancer (TGI is 82.67%);
- TH-302 combined with Olaparib has a good therapeutic effect on Olaparib-resistant pancreatic cancer (TGI is 87.43%);
- the TH-302 monotherapy group had a slightly better antitumor effect, but there was no significant difference (p>0.05);
- the inventors also studied the efficacy and safety of TH-302 in the olaparib-resistant lung cancer LU6429 PDX model and bladder cancer BL3325 PDX model.
- mice were subcutaneously inoculated with human pancreatic cancer Capan-1 cells to establish a subcutaneous transplantation model of human pancreatic cancer.
- the test is divided into test drug Olaparib 100mg/kg single drug group (Group 2), TH-302 50mg/kg single drug group (Group 3, QD), TH-302 100mg/kg single drug group (Group 4), TH-302 50mg/kg single drug group (Group 5, QW), TH-302 25mg/kg single drug group (Group 6), TH-302 25mg/kg and Olaparib 100mg/kg combined administration group (Group 7) and 10% no Water ethanol + 10% polyoxyethylene (35) castor oil + 80% glucose injection D5W (pH7.4) vehicle control group (Group 1), a total of 7 groups, 6 mice in each group.
- Olaparib 100mg/kg single drug group Group 2
- TH-302 50mg/kg single drug group Group 3, QD
- TH-302 100mg/kg single drug group Group 4
- the vehicle control group and each test drug of TH-302 were administered by tail vein injection.
- the TH-302 50mg/kg single-drug group (Group 3, QD) was administered once a day for 3 consecutive days, rested for 4 days, and then rested for two weeks; then administered daily for 3 consecutive days.
- TH-302 100mg/kg (Group 4, QW), 50mg/kg (Group 5, QW), 25mg/kg (Group 6, QW) single drug group and combined administration of TH-302 25mg/kg and Olaparib 100mg/kg TH-302 in Group 7 was administered once a week for a total of three weeks.
- the test drug Olaparib in each group was administered orally by gavage, once a day, for a total of 30 days.
- the specific administration route, dose and scheme in the human pancreatic cancer Capan-1 animal model are shown in Table 12.
- Table 12 Administration routes, doses and regimens in human-derived pancreatic cancer Capan-1 animal model
- the dosing volume is 10 ⁇ L/g.
- the tumor growth of each treatment group and control group was recorded on different days of the experiment, as shown in Table 13, and the growth curves of the tumor volumes of mice in each group are shown in FIG. 7 .
- the curative effect was evaluated according to the relative tumor proliferation rate and the relative tumor inhibition rate, and the efficacy analysis of each group is shown in Table 14.
- the results of the mouse body weight change rate on the 43rd day are shown in Table 15.
- the body weight of each treatment group changed with time The change curve is shown in Figure 8.
- Relative tumor proliferation rate, T/C% that is, at a certain time point, the percentage value of the relative tumor volume or tumor weight between the treatment group and the control group.
- T/C% T TW /C TW ⁇ 100% (T TW : the average tumor weight at the end of the experiment in the treatment group; C TW : the average tumor weight at the end of the experiment in the vehicle control group).
- T and C are the relative tumor volume (RTV) or tumor weight (TW) of the treatment group and the control group at a specific time point, respectively).
- Table 13 Changes in tumor volume of mice in each group with treatment time in the Capan-1 model of human pancreatic cancer (mm 3 )
- Table 14 Drug efficacy analysis table of each group in the subcutaneous model of human pancreatic cancer Capan-1
- Table 15 Body weight changes in each group in the human pancreatic cancer Capan-1 subcutaneous model
- mice in the vehicle control group were 1301.38 mm 3 on day 43 after tumor cell inoculation (Day 43).
- the average tumor volume of the test drug Olaparib 100mg/kg (Group 2) treatment group was 846.86mm 3 on the 43rd day after tumor cell inoculation (Day 43), and the relative tumor inhibition rate TGI (%) was 37.43%. There was no statistically significant difference (p>0.05).
- the test drug TH-302 50mg/kg treatment group (Group 3, QD) had an average tumor volume of 146.99mm 3 on the 43rd day after tumor cell inoculation (Day 43), which was statistically significantly different from the control group ( p ⁇ 0.001), the relative tumor inhibition rate TGI (%) was 89.17%.
- the test drug TH-302 100mg/kg treatment group (Group 4) had an average tumor volume of 124.68mm 3 on the 43rd day after tumor cell inoculation (Day 43), which was statistically significantly different from the control group (p ⁇ 0.001), and the relative tumor inhibition rate TGI (%) was 90.89%.
- the test drug TH-302 50mg/kg treatment group (Group 5, QW) had an average tumor volume of 263.45mm 3 on the 43rd day after tumor cell inoculation (Day 43), which was statistically significantly different from the control group ( p ⁇ 0.001), the relative tumor inhibition rate TGI (%) was 80.18%, and the complete tumor inhibition rate was 33.3%.
- TH-302 100mg/kg (Group 4), 50mg/kg (Group 5, QW) and 25mg/kg (Group 6) monotherapy groups showed a dose-dependent antitumor effect.
- TH-302 100mg/kg (Group 4, QW), 50mg/kg (Group 5, QW) monotherapy group compared with TH-302 25mg/kg monotherapy group (Group 6, QW) had statistically significant Sex difference (all p ⁇ 0.001).
- the combination therapy group of Olaparib 100mg/kg and TH-302 25mg/kg (Group 7) has better anti-tumor effect than Olaparib 100mg/kg (Group 2) and TH-302 25mg/kg single drug group (Group 6), statistics There are significant differences (p ⁇ 0.05 and p ⁇ 0.001), and the specific dose combination of Olaparib and TH-302 can show a synergistic effect on tumor inhibition.
- the LU6429 PDX model is an olaparib-resistant model with a BRCA2 pathogenic mutation.
- mice Balb/c nude female mice were inoculated subcutaneously
- the tumor mass of lung cancer LU6429 was used to establish the subcutaneous transplantation tumor model of human lung cancer.
- the test is divided into test drug Olaparib 50mg/kg single drug group (Group 02), TH-302 80mg/kg single drug group (Group 03), TH-302 40mg/kg single drug group (Group 04), TH-302 20mg/kg single drug group (Group 04), TH-302 20mg/kg kg single drug group (Group 05), TH-302 40mg/kg and Olaparib 50mg/kg combined administration group (Group 06), and glucose injection vehicle control group (Group 01).
- mice in each group There were 6 groups in this study, 6 mice in each group, in which the vehicle control group and TH-302 were given tail vein administration once a week for a total of 3 weeks; Olaparib was administered by intragastric administration and administered daily Medicine 1 time, a total of 28 days of administration.
- Table 16 shows the administration route, dosage and regimen in the lung cancer LU6429 animal model.
- Table 16 Administration route, dosage and regimen in animal model of lung cancer LU6429
- the administration volume is 10 ⁇ L/g; 2. QD ⁇ 28: once a day for 28 consecutive days; 3. QW ⁇ 3: once a week for 3 consecutive weeks.
- the tumor growth of each treatment group and control group was recorded on different days of the experiment, as shown in Table 17, and the growth curves of the tumor volumes of mice in each group are shown in FIG. 9 .
- the curative effect was evaluated according to the relative tumor proliferation rate and the relative tumor inhibition rate, and the efficacy analysis of each group is shown in Table 18. Record the changes in body weight after administration of the treatment group and the control group.
- the safety of each group in the subcutaneous model of lung cancer LU6429 is shown in Table 19, and the body weight change results of the mice on day 25 are shown in Table 10.
- Table 17 In Changes of tumor volume in each group of mice in lung cancer LU6429 model with treatment time (mm 3 )
- Table 18 In Drug efficacy analysis table of each group in the subcutaneous model of lung cancer LU6429
- Olaparib alone did not produce tumor inhibitory effect, showing the resistance of lung cancer LU6429 PDX model to Olaparib.
- the TH-302 80mg/kg (Group 03) dose treatment group the tumor of 1 mouse was completely cleared, and the clearance rate was 16.7%.
- the test drug TH-302 80mg/kg, 40mg/kg and 20mg/kg treatment groups had statistically significant differences (p ⁇ 0.05), showing dose dependence.
- mice in each test drug treatment group did not lose weight and were well tolerated.
- the BL3325 PDX model is an olaparib-resistant model with a BRCA2 pathogenic mutation.
- mice Balb/c nude female mice were inoculated subcutaneously
- the tumor mass of bladder cancer BL3325 was used to establish a subcutaneous transplanted tumor model of human bladder cancer.
- the test is divided into test drug Olaparib 50mg/kg single drug group (Group 02), TH-302 80mg/kg single drug group (Group 03), TH-302 40mg/kg single drug group (Group 04), TH-302 20mg/kg single drug group (Group 04), TH-302 20mg/kg kg single drug group (Group 05), TH-302 40mg/kg and Olaparib 50mg/kg combined administration group (Group 06), and glucose injection vehicle control group (Group 01).
- mice in each group There were 6 groups in this study, 6 mice in each group, in which the vehicle control group and TH-302 were given tail vein administration once a week for a total of 3 weeks; Olaparib was administered by intragastric administration and administered daily 1 dose, for a total of 30 days.
- Table 20 shows the administration route, dosage and regimen in the bladder cancer BL3325 animal model.
- Table 20 Administration route, dosage and regimen in bladder cancer BL3325 animal model
- the administration volume is 10 ⁇ L/g; 2. QD ⁇ 30: once a day for 30 consecutive days; 3. QW ⁇ 3: once a week for 3 weeks; 4. i.v. is tail vein administration, p.o. is intragastric administration.
- Table 21 In Changes of tumor volume in each group of mice in bladder cancer BL3325 model with treatment time (mm 3 )
- Table 22 In Drug efficacy analysis table of each group in bladder cancer BL3325 subcutaneous model
- Table 23 In Body weight changes of each group in bladder cancer BL3325 subcutaneous model
- Test drug TH-302 in 80mg/kg, 40mg/kg, 20mg/kg dose alone treatment group, and TH-302 40mg/kg combined with Olaparib 50mg/kg treatment group Bladder cancer BL3325 subcutaneous models all have significant anti-tumor proliferation effects, and Olaparib 50mg/kg alone has no significant anti-tumor effect.
- the TH-302 80mg/kg treatment group Group 03
- the combined treatment effect of TH-302 40mg/kg and Olaparib 50mg/kg was significantly better than that of Olaparib 50mg/kg alone, better than that of TH-302 40mg/kg alone, and the difference was significant.
- mice in each test drug treatment group did not lose weight and were well tolerated.
- the PARP inhibitor selected in the examples of the present application is Olaparib, Rucaparib, Niraparib, Talazoparib, Fluzoparib, Pamiparib, etc. It is also a PARP inhibitor, and its mechanism of action is similar to that of Olaparib, both of which block the function of enzymes involved in the repair of damaged DNA. Therefore, it can be inferred that Rucaparib, Niraparib, and Talazopa PARPi such as Talazoparib, Fluzoparib, and Pamiparib have similar tumor inhibitory efficacy to Olaparib in the above experiment.
- TH-302 is the DNA alkylating agent activated by hypoxia, the general formula compound of claim 1:
Landscapes
- Health & Medical Sciences (AREA)
- General Health & Medical Sciences (AREA)
- Chemical & Material Sciences (AREA)
- Medicinal Chemistry (AREA)
- Veterinary Medicine (AREA)
- Public Health (AREA)
- Pharmacology & Pharmacy (AREA)
- Life Sciences & Earth Sciences (AREA)
- Animal Behavior & Ethology (AREA)
- Epidemiology (AREA)
- General Chemical & Material Sciences (AREA)
- Organic Chemistry (AREA)
- Nuclear Medicine, Radiotherapy & Molecular Imaging (AREA)
- Chemical Kinetics & Catalysis (AREA)
- Pharmaceuticals Containing Other Organic And Inorganic Compounds (AREA)
Abstract
Description
| 化合物 | IC50(μM) | 比率(21%O 2/0.01%O 2) |
| TH-302,21%O 2 | 93.45 | - |
| TH-302,<0.01%O 2 | 0.82 | 113.96 |
| 化合物 | IC50(μM) | 比率(21%O 2/0.01%O 2) |
| 替拉扎明,21%O 2 | >2000 | - |
| 替拉扎明,<0.01%O 2 | 29.06 | >68.82 |
| 化合物 | IC50(μM) | 比率(21%O 2/0.01%O 2) |
| TH-302,21%O 2 | 423.50 | - |
| TH-302,<0.01%O 2 | 3.07 | 137.95 |
| 化合物 | IC50(μM) | 比率(21%O 2/0.01%O 2) |
| 替拉扎明,21%O 2 | >2000 | - |
| 替拉扎明,<0.01%O 2 | 33.23 | >60.19 |
Claims (16)
- 根据权利要求1所述的治疗方法,其使用含有式(I)的乏氧激活化合物的药物联用PARP抑制剂治疗PARP抑制剂耐药的癌症、肿瘤患者。
- 根据权利要求1或2所述的治疗方法,其中,所述患者的DNA修复酶受损;或所述患者的肿瘤或癌组织被检测出具有BRCA1、BRCA2对应的基因中的任意一个基因突变或两个基因突变;或所述患者被检测出具有BRCA1、BRCA2对应的基因中的任意一个基因突变或两个基因突变。
- 根据权利要求3所述的治疗方法,其中,BRCA1、BRCA2突变包括胚系突变(gBRCAm)和体系突变(sBRCAm)的BRCA1、BRCA2突变。
- 根据权利要求1或2所述的治疗方法,其中,所述PARP抑制剂选自奥拉帕利Olaparib、芦卡帕利Rucaparib、尼拉帕利Niraparib、他拉唑帕利Talazoparib、氟唑帕利Fluzoparib、帕米帕利Pamiparib;所述癌症、肿瘤选自卵巢癌、乳腺癌、胰腺癌、输卵管癌、原发性腹膜癌、胃癌、前列腺癌、肝癌、结肠癌、直肠癌、肺癌、膀胱癌,所述肺癌优选为非小细胞肺癌、小细胞肺癌。
- 根据权利要求3或4或7或8或9任一项所述的治疗方法,其中,所述的基因突变的TMB(肿瘤基因突变负荷)水平为中。
- 根据权利要求11所述的用途,其中,所述患者的DNA修复酶受损;或所述患者的肿瘤或癌组织被检测出具有BRCA1、BRCA2对应的基因中的任意一个基因突变或两个基因突变;或所述患者被检测出具有BRCA1、BRCA2对应的基因中的任意一个基因突变或两个基因突变。
- 根据权利要求12所述的用途,其中,所述BRCA1、BRCA2突变包括胚系突变(gBRCAm)和体系突变(sBRCAm)的BRCA1、BRCA2突变;所述基因突变的TMB(肿瘤基因突变负荷)水平为中。
- 根据权利要求15所述的药物,其中,所述患者的DNA修复酶受损;或所述患者的肿瘤或癌组织被检测出具有BRCA1、BRCA2对应的基因中的任意一个基因突变或两个基因突变;或所述患者被检测出具有BRCA1、BRCA2对应的基因中的任意一个基因突变或两个基因突变,优先的,BRCA1、BRCA2突变包括胚系突变(gBRCAm)和体系突变(sBRCAm)的BRCA1、BRCA2突变。
Priority Applications (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| KR1020247008354A KR20240051965A (ko) | 2021-08-27 | 2022-08-26 | Th-302를 이용한 parp 억제제에 내성이 있는 환자의 치료 |
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| CN202110996009 | 2021-08-27 | ||
| CN202110996009.2 | 2021-08-27 |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| WO2023025312A1 true WO2023025312A1 (zh) | 2023-03-02 |
Family
ID=85322487
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/CN2022/115284 WO2023025312A1 (zh) | 2021-08-27 | 2022-08-26 | 使用th-302治疗parp抑制剂耐药的患者 |
Country Status (2)
| Country | Link |
|---|---|
| KR (1) | KR20240051965A (zh) |
| WO (1) | WO2023025312A1 (zh) |
Cited By (2)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2023174319A1 (zh) * | 2022-03-15 | 2023-09-21 | 深圳艾欣达伟医药科技有限公司 | 治疗brca突变癌症患者的方法 |
| WO2024061346A1 (zh) * | 2022-09-22 | 2024-03-28 | 深圳艾欣达伟医药科技有限公司 | 乏氧激活的化合物在制备治疗癌症患者的药物中的用途 |
Citations (32)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2004087075A2 (en) | 2003-03-28 | 2004-10-14 | Threshold Pharmaceuticals, Inc. | Compositions and methods for treating cancer |
| WO2007002931A2 (en) | 2005-06-29 | 2007-01-04 | Threshold Pharmaceuticals, Inc. | Phosphoramidate alkylator prodrugs |
| WO2008083101A1 (en) | 2006-12-26 | 2008-07-10 | Threshold Pharmaceuticals, Inc. | Phosphoramidate alkylator prodrugs for the treatment of cancer |
| WO2008151253A1 (en) | 2007-06-04 | 2008-12-11 | Threshold Pharmaceuticals, Inc. | Hypoxia activated prodrugs of antineoplastic agents |
| WO2009018163A1 (en) | 2007-07-27 | 2009-02-05 | Threshold Pharmaceuticals, Inc. | Hypoxia activated prodrugs of anthracyclines |
| WO2009033165A1 (en) | 2007-09-06 | 2009-03-12 | Threshold Pharmaceuticals, Inc. | Hypoxia activated prodrugs of bis-alkylating agents |
| WO2010048330A1 (en) | 2008-10-21 | 2010-04-29 | Threshold Pharmaceuticals, Inc. | Treatment of cancer using hypoxia activated prodrugs |
| WO2012006032A2 (en) | 2010-06-28 | 2012-01-12 | Threshold Pharmaceuticals, Inc. | Treatment of blood cancer |
| WO2012009288A2 (en) | 2010-07-12 | 2012-01-19 | Threshold Pharmaceuticals, Inc. | Administration of hypoxia activated prodrugs and antiangiogenic agents for the treatment of cancer |
| WO2012135757A2 (en) | 2011-04-01 | 2012-10-04 | Threshold Pharmaceuticals, Inc. | Methods for treating cancer |
| WO2012142520A2 (en) | 2011-04-15 | 2012-10-18 | Threshold Pharmaceuticals, Inc. | Unit dose form for oral administration |
| WO2012145684A1 (en) | 2011-04-22 | 2012-10-26 | The United States Of America, As Represented By The Secretary, Department Of Health & Human Services | Transient hypoxia inducers and their use |
| WO2013096687A1 (en) | 2011-12-22 | 2013-06-27 | Threshold Pharmaceuticals, Inc. | Administration of hypoxia activated prodrugs in combination with chk1 inhibitors for treating cancer |
| WO2013096684A1 (en) | 2011-12-22 | 2013-06-27 | Threshold Pharmaceuticals, Inc. | Hypoxia activated prodrugs and mtor inhibitors for treating cancer |
| WO2013116385A1 (en) | 2012-01-31 | 2013-08-08 | Threshold Pharmaceuticals, Inc. | Predictive biomarker for hypoxia-activated prodrug therapy |
| WO2013126539A1 (en) | 2012-02-21 | 2013-08-29 | Threshold Pharmaceuticals Inc. | Treatment of cancer |
| WO2013177633A1 (en) | 2012-06-01 | 2013-12-05 | Bionomics Limited | Combination therapy involving a vascular disrupting agent and an agent which targets hypoxia |
| WO2014062856A1 (en) | 2012-10-16 | 2014-04-24 | Halozyme, Inc. | Hypoxia and hyaluronan and markers thereof for diagnosis and monitoring of diseases and conditions and related methods |
| WO2014169035A1 (en) | 2013-04-10 | 2014-10-16 | Threshold Pharmaceuticals, Inc. | Predictive and response biomarker for th-302 anti-cancer therapy |
| WO2015013448A1 (en) | 2013-07-26 | 2015-01-29 | Threshold Pharmaceuticals, Inc. | Treatment of pancreatic cancer with a combination of a hypoxia-acti vated prodrug and a taxane |
| WO2015051921A1 (en) | 2013-10-10 | 2015-04-16 | Merck Patent Gmbh | Synthesis of 1-alkyl-2-amino-imidazol-5-carboxylic acid ester via calpha-substituted n-alkyl-glycine ester derivatives |
| WO2015069489A1 (en) | 2013-11-06 | 2015-05-14 | Merck Patent Gmbh | Predictive biomarker for hypoxia-activated prodrug therapy |
| WO2015171647A1 (en) | 2014-05-05 | 2015-11-12 | Threshold Pharmaceuticals, Inc. | Treatment of cancer |
| WO2016011195A1 (en) | 2014-07-17 | 2016-01-21 | Threshold Pharmaceuticals, Inc. | Th-302 solid forms and methods related thereto |
| WO2016011328A1 (en) | 2014-07-17 | 2016-01-21 | Baker Cheryl | Treatment of cancer with a combination of radiation, cerium oxide nanoparticles, and a chemotherapeutic agent |
| WO2016014390A1 (en) | 2014-07-25 | 2016-01-28 | Merck Patent Gmbh | Compositions and methods for mek inhibitor combination therapy in the treatment of cancer |
| WO2016081547A1 (en) | 2014-11-19 | 2016-05-26 | Merck Patent Gmbh | Predictive biomarker for hypoxia-activated prodrug therapy |
| WO2018026606A1 (en) | 2016-08-01 | 2018-02-08 | Threshold Pharmaceuticals, Inc. | Administration of hypoxia activated prodrugs in combination with immune modulatory agents for treating cancer |
| WO2019055786A1 (en) | 2017-09-14 | 2019-03-21 | Lankenau Institute For Medical Research | METHODS AND COMPOSITIONS FOR THE TREATMENT OF CANCER |
| WO2019173799A1 (en) | 2018-03-08 | 2019-09-12 | Caris Science, Inc. | Oligonucleotide probes and uses thereof |
| WO2020007106A1 (zh) | 2018-07-05 | 2020-01-09 | 深圳艾欣达伟医药科技有限公司 | 埃夫索胺的抗癌医药用途 |
| WO2020118251A2 (en) | 2018-12-07 | 2020-06-11 | The Board Of Trustees Of The Leland Stanford Junior University | Hypoxia targeting compositions and combinations thereof with a parp inhibitor and methods of use thereof |
-
2022
- 2022-08-26 WO PCT/CN2022/115284 patent/WO2023025312A1/zh active Application Filing
- 2022-08-26 KR KR1020247008354A patent/KR20240051965A/ko unknown
Patent Citations (32)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2004087075A2 (en) | 2003-03-28 | 2004-10-14 | Threshold Pharmaceuticals, Inc. | Compositions and methods for treating cancer |
| WO2007002931A2 (en) | 2005-06-29 | 2007-01-04 | Threshold Pharmaceuticals, Inc. | Phosphoramidate alkylator prodrugs |
| WO2008083101A1 (en) | 2006-12-26 | 2008-07-10 | Threshold Pharmaceuticals, Inc. | Phosphoramidate alkylator prodrugs for the treatment of cancer |
| WO2008151253A1 (en) | 2007-06-04 | 2008-12-11 | Threshold Pharmaceuticals, Inc. | Hypoxia activated prodrugs of antineoplastic agents |
| WO2009018163A1 (en) | 2007-07-27 | 2009-02-05 | Threshold Pharmaceuticals, Inc. | Hypoxia activated prodrugs of anthracyclines |
| WO2009033165A1 (en) | 2007-09-06 | 2009-03-12 | Threshold Pharmaceuticals, Inc. | Hypoxia activated prodrugs of bis-alkylating agents |
| WO2010048330A1 (en) | 2008-10-21 | 2010-04-29 | Threshold Pharmaceuticals, Inc. | Treatment of cancer using hypoxia activated prodrugs |
| WO2012006032A2 (en) | 2010-06-28 | 2012-01-12 | Threshold Pharmaceuticals, Inc. | Treatment of blood cancer |
| WO2012009288A2 (en) | 2010-07-12 | 2012-01-19 | Threshold Pharmaceuticals, Inc. | Administration of hypoxia activated prodrugs and antiangiogenic agents for the treatment of cancer |
| WO2012135757A2 (en) | 2011-04-01 | 2012-10-04 | Threshold Pharmaceuticals, Inc. | Methods for treating cancer |
| WO2012142520A2 (en) | 2011-04-15 | 2012-10-18 | Threshold Pharmaceuticals, Inc. | Unit dose form for oral administration |
| WO2012145684A1 (en) | 2011-04-22 | 2012-10-26 | The United States Of America, As Represented By The Secretary, Department Of Health & Human Services | Transient hypoxia inducers and their use |
| WO2013096687A1 (en) | 2011-12-22 | 2013-06-27 | Threshold Pharmaceuticals, Inc. | Administration of hypoxia activated prodrugs in combination with chk1 inhibitors for treating cancer |
| WO2013096684A1 (en) | 2011-12-22 | 2013-06-27 | Threshold Pharmaceuticals, Inc. | Hypoxia activated prodrugs and mtor inhibitors for treating cancer |
| WO2013116385A1 (en) | 2012-01-31 | 2013-08-08 | Threshold Pharmaceuticals, Inc. | Predictive biomarker for hypoxia-activated prodrug therapy |
| WO2013126539A1 (en) | 2012-02-21 | 2013-08-29 | Threshold Pharmaceuticals Inc. | Treatment of cancer |
| WO2013177633A1 (en) | 2012-06-01 | 2013-12-05 | Bionomics Limited | Combination therapy involving a vascular disrupting agent and an agent which targets hypoxia |
| WO2014062856A1 (en) | 2012-10-16 | 2014-04-24 | Halozyme, Inc. | Hypoxia and hyaluronan and markers thereof for diagnosis and monitoring of diseases and conditions and related methods |
| WO2014169035A1 (en) | 2013-04-10 | 2014-10-16 | Threshold Pharmaceuticals, Inc. | Predictive and response biomarker for th-302 anti-cancer therapy |
| WO2015013448A1 (en) | 2013-07-26 | 2015-01-29 | Threshold Pharmaceuticals, Inc. | Treatment of pancreatic cancer with a combination of a hypoxia-acti vated prodrug and a taxane |
| WO2015051921A1 (en) | 2013-10-10 | 2015-04-16 | Merck Patent Gmbh | Synthesis of 1-alkyl-2-amino-imidazol-5-carboxylic acid ester via calpha-substituted n-alkyl-glycine ester derivatives |
| WO2015069489A1 (en) | 2013-11-06 | 2015-05-14 | Merck Patent Gmbh | Predictive biomarker for hypoxia-activated prodrug therapy |
| WO2015171647A1 (en) | 2014-05-05 | 2015-11-12 | Threshold Pharmaceuticals, Inc. | Treatment of cancer |
| WO2016011195A1 (en) | 2014-07-17 | 2016-01-21 | Threshold Pharmaceuticals, Inc. | Th-302 solid forms and methods related thereto |
| WO2016011328A1 (en) | 2014-07-17 | 2016-01-21 | Baker Cheryl | Treatment of cancer with a combination of radiation, cerium oxide nanoparticles, and a chemotherapeutic agent |
| WO2016014390A1 (en) | 2014-07-25 | 2016-01-28 | Merck Patent Gmbh | Compositions and methods for mek inhibitor combination therapy in the treatment of cancer |
| WO2016081547A1 (en) | 2014-11-19 | 2016-05-26 | Merck Patent Gmbh | Predictive biomarker for hypoxia-activated prodrug therapy |
| WO2018026606A1 (en) | 2016-08-01 | 2018-02-08 | Threshold Pharmaceuticals, Inc. | Administration of hypoxia activated prodrugs in combination with immune modulatory agents for treating cancer |
| WO2019055786A1 (en) | 2017-09-14 | 2019-03-21 | Lankenau Institute For Medical Research | METHODS AND COMPOSITIONS FOR THE TREATMENT OF CANCER |
| WO2019173799A1 (en) | 2018-03-08 | 2019-09-12 | Caris Science, Inc. | Oligonucleotide probes and uses thereof |
| WO2020007106A1 (zh) | 2018-07-05 | 2020-01-09 | 深圳艾欣达伟医药科技有限公司 | 埃夫索胺的抗癌医药用途 |
| WO2020118251A2 (en) | 2018-12-07 | 2020-06-11 | The Board Of Trustees Of The Leland Stanford Junior University | Hypoxia targeting compositions and combinations thereof with a parp inhibitor and methods of use thereof |
Non-Patent Citations (8)
| Title |
|---|
| ASHWORTH, A.LORD, C. J.: "Synthetic Lethal Therapies for Cancer: What's Next after PARP Inhibitors?", NATURE REVIEWS. CLINICAL ONCOLOGY, vol. 15, no. 9, 2018, pages 564 - 576, XP036573545, DOI: 10.1038/s41571-018-0055-6 |
| CONROY, M.BORAD, M. J.BRYCE, A. H.: "Hypoxia-Activated Alkylating Agents in BRCAl-Mutant Ovarian Serous Carcinoma", CUREUS, vol. 9, no. 7, 2017, pages e1517 |
| HE LIZHAO-YI LIUNAYIYUAN WUYONG-CHANG CHENQUAN CHENGJING WANG: "PARP Inhibitor Resistance: the Underlying Mechanisms and Clinical Implications", MOL CANCER, vol. 19, no. 1, 20 June 2020 (2020-06-20), pages 107, XP055753848, DOI: 10.1186/s12943-020-01227-0 |
| MATEO, J.LORD, C. J.SERRA, V.TUTT, A.BALMANA, J.CASTROVIEJO-BERMEJO, M.CRUZ, C.OAKNIN, A.KAYE, S. BDE BONO, J. S.: "A Decade of Clinical Development of PARP Inhibitors in Perspective", ANNALS OF ONCOLOGY: OFFICIAL JOURNAL OF THE EUROPEAN SOCIETY FOR MEDICAL ONCOLOGY, vol. 30, no. 9, 2019, pages 1437 - 1447, XP055860728, DOI: 10.1093/annonc/mdz192 |
| MENG FEVANS J WBHUPATHI D ET AL.: "Molecular and Cellular Pharmacology of the Hypoxia-Activated Prodrug TH-302. [J", MOLECULAR CANCER THERAPEUTICS, vol. 11, no. 918633-87-1, 2012, pages 740, XP055039520, DOI: 10.1158/1535-7163.MCT-11-0634 |
| no. 912444-00-9 |
| ROSE, M.BURGESS, J. T.O'BYRNE, K.RICHARD, D. J.BOLDERSON, E.: "PARP Inhibitors: Clinical Relevance, Mechanisms of Action and Tumor Resistance", FRONTIERS IN CELL AND DEVELOPMENTAL BIOLOGY, vol. 8, 2020, pages 564601 |
| VENA FRANCESCA, JIA RUOCHEN, ESFANDIARI ARMAN, GARCIA-GOMEZ JUAN J., RODRIGUEZ-JUSTO MANUEL, MA JIANGUO, SYED SAKEENA, CROWLEY LIN: "MEK inhibition leads to BRCA2 downregulation and sensitization to DNA damaging agents in pancreas and ovarian cancer models", ONCOTARGET, vol. 9, no. 14, 20 February 2018 (2018-02-20), pages 11592 - 11603, XP093039635, DOI: 10.18632/oncotarget.24294 * |
Cited By (2)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2023174319A1 (zh) * | 2022-03-15 | 2023-09-21 | 深圳艾欣达伟医药科技有限公司 | 治疗brca突变癌症患者的方法 |
| WO2024061346A1 (zh) * | 2022-09-22 | 2024-03-28 | 深圳艾欣达伟医药科技有限公司 | 乏氧激活的化合物在制备治疗癌症患者的药物中的用途 |
Also Published As
| Publication number | Publication date |
|---|---|
| KR20240051965A (ko) | 2024-04-22 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| WO2023025312A1 (zh) | 使用th-302治疗parp抑制剂耐药的患者 | |
| Chang et al. | Phase I study of nanoliposomal irinotecan (PEP02) in advanced solid tumor patients | |
| US6979675B2 (en) | Treatment of cancer with 2-deoxyglucose | |
| CN106659716B (zh) | 阿吡莫德组合物及其使用方法 | |
| CN106488776B (zh) | 包含糖皮质激素和edo-s101的组合 | |
| CA2459822C (en) | Treatment of chronic myelogenous leukemia, resistant or intolerant to sti571, involving homoharringtonine alone or combined with other agents | |
| US20230011378A1 (en) | Combination Therapy For Treatment Of Cancer | |
| KR20140079831A (ko) | 췌장암 및/또는 담도암 치료약 | |
| TW202123943A (zh) | 用於癌症治療之考比西他 (cobicistat) | |
| CN113710658A (zh) | 用于治疗尤因肉瘤的喹啉类化合物或其药学上可接受的盐 | |
| AU2023202746A1 (en) | Combination of a BCL-2 inhibitor and a MCL-1 inhibitor, uses and pharmaceutical compositions thereof | |
| US11311548B2 (en) | Cancer therapy | |
| KR20220011647A (ko) | 소세포폐암의 병용 치료용 퀴놀린 유도체 | |
| WO2023198188A1 (zh) | 使用th-302单药或联用parp抑制剂治疗癌症的方法 | |
| WO2023174319A1 (zh) | 治疗brca突变癌症患者的方法 | |
| KR102607967B1 (ko) | 단백질 키나제 억제제 및 추가 화학요법제의 조합물 | |
| CN116159062A (zh) | 药物组合物及其用途 | |
| CN113116895A (zh) | 用于治疗神经母细胞瘤的喹啉衍生物 | |
| US11890292B2 (en) | Compositions, methods, systems and/or kits for preventing and/or treating neoplasms | |
| EP2928456A1 (en) | Methods and compositions for treatment of cancer | |
| WO2024061346A1 (zh) | 乏氧激活的化合物在制备治疗癌症患者的药物中的用途 | |
| US20220323465A1 (en) | Pharmaceutical combination and use thereof | |
| Eatock et al. | A dose-finding study of raltitrexed (tomudex) with cisplatin and epirubicin in advanced gastro-oesophageal adenocarcinoma | |
| CN115038447A (zh) | 用于治疗癌症的组合疗法 | |
| JP2014513698A (ja) | がん治療保護のためのヒ素の使用 |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application |
Ref document number: 22860658 Country of ref document: EP Kind code of ref document: A1 |
|
| ENP | Entry into the national phase |
Ref document number: 20247008354 Country of ref document: KR Kind code of ref document: A |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 2022860658 Country of ref document: EP |
|
| NENP | Non-entry into the national phase |
Ref country code: DE |
|
| ENP | Entry into the national phase |
Ref document number: 2022860658 Country of ref document: EP Effective date: 20240327 |