WO2022220232A1 - すい臓がん検出用蛍光プローブ - Google Patents
すい臓がん検出用蛍光プローブ Download PDFInfo
- Publication number
- WO2022220232A1 WO2022220232A1 PCT/JP2022/017566 JP2022017566W WO2022220232A1 WO 2022220232 A1 WO2022220232 A1 WO 2022220232A1 JP 2022017566 W JP2022017566 W JP 2022017566W WO 2022220232 A1 WO2022220232 A1 WO 2022220232A1
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- pancreatic cancer
- fluorescent probe
- tissue
- cancer
- residue
- Prior art date
Links
- 206010061902 Pancreatic neoplasm Diseases 0.000 title claims abstract description 83
- 208000015486 malignant pancreatic neoplasm Diseases 0.000 title claims abstract description 83
- 201000002528 pancreatic cancer Diseases 0.000 title claims abstract description 83
- 208000008443 pancreatic carcinoma Diseases 0.000 title claims abstract description 83
- 239000007850 fluorescent dye Substances 0.000 title claims abstract description 75
- 238000001514 detection method Methods 0.000 title claims abstract description 10
- 150000003839 salts Chemical class 0.000 claims abstract description 20
- 150000001875 compounds Chemical class 0.000 claims abstract description 19
- 206010028980 Neoplasm Diseases 0.000 claims description 80
- 201000011510 cancer Diseases 0.000 claims description 72
- 238000000034 method Methods 0.000 claims description 42
- 239000000523 sample Substances 0.000 claims description 38
- 238000001356 surgical procedure Methods 0.000 claims description 31
- 238000000799 fluorescence microscopy Methods 0.000 claims description 30
- 125000004435 hydrogen atom Chemical group [H]* 0.000 claims description 30
- 125000000217 alkyl group Chemical group 0.000 claims description 28
- 125000001424 substituent group Chemical group 0.000 claims description 24
- 239000000203 mixture Substances 0.000 claims description 21
- 238000002073 fluorescence micrograph Methods 0.000 claims description 16
- 210000000496 pancreas Anatomy 0.000 claims description 10
- 125000003630 glycyl group Chemical group [H]N([H])C([H])([H])C(*)=O 0.000 claims description 9
- 125000005843 halogen group Chemical group 0.000 claims description 9
- 125000001500 prolyl group Chemical group [H]N1C([H])(C(=O)[*])C([H])([H])C([H])([H])C1([H])[H] 0.000 claims description 9
- 125000001570 methylene group Chemical group [H]C([H])([*:1])[*:2] 0.000 claims description 8
- 230000005284 excitation Effects 0.000 claims description 7
- 125000001997 phenyl group Chemical group [H]C1=C([H])C([H])=C(*)C([H])=C1[H] 0.000 claims description 7
- DHMQDGOQFOQNFH-UHFFFAOYSA-N Glycine Chemical compound NCC(O)=O DHMQDGOQFOQNFH-UHFFFAOYSA-N 0.000 claims description 6
- 125000000539 amino acid group Chemical group 0.000 claims description 6
- 125000002887 hydroxy group Chemical group [H]O* 0.000 claims description 6
- VEYYWZRYIYDQJM-UHFFFAOYSA-N n-α-acetyllysine Chemical group CC(=O)NC(C(O)=O)CCCCN VEYYWZRYIYDQJM-UHFFFAOYSA-N 0.000 claims description 6
- YZCKVEUIGOORGS-IGMARMGPSA-N Protium Chemical compound [1H] YZCKVEUIGOORGS-IGMARMGPSA-N 0.000 claims description 5
- 238000002674 endoscopic surgery Methods 0.000 claims description 5
- ONIBWKKTOPOVIA-UHFFFAOYSA-N Proline Natural products OC(=O)C1CCCN1 ONIBWKKTOPOVIA-UHFFFAOYSA-N 0.000 claims description 4
- 239000004471 Glycine Substances 0.000 claims description 3
- ONIBWKKTOPOVIA-BYPYZUCNSA-N L-Proline Chemical compound OC(=O)[C@@H]1CCCN1 ONIBWKKTOPOVIA-BYPYZUCNSA-N 0.000 claims description 3
- ROHFNLRQFUQHCH-YFKPBYRVSA-N L-leucine Chemical compound CC(C)C[C@H](N)C(O)=O ROHFNLRQFUQHCH-YFKPBYRVSA-N 0.000 claims description 3
- OUYCCCASQSFEME-QMMMGPOBSA-N L-tyrosine Chemical compound OC(=O)[C@@H](N)CC1=CC=C(O)C=C1 OUYCCCASQSFEME-QMMMGPOBSA-N 0.000 claims description 3
- ROHFNLRQFUQHCH-UHFFFAOYSA-N Leucine Natural products CC(C)CC(N)C(O)=O ROHFNLRQFUQHCH-UHFFFAOYSA-N 0.000 claims description 3
- 230000001678 irradiating effect Effects 0.000 claims description 3
- 125000001909 leucine group Chemical group [H]N(*)C(C(*)=O)C([H])([H])C(C([H])([H])[H])C([H])([H])[H] 0.000 claims description 3
- OUYCCCASQSFEME-UHFFFAOYSA-N tyrosine Natural products OC(=O)C(N)CC1=CC=C(O)C=C1 OUYCCCASQSFEME-UHFFFAOYSA-N 0.000 claims description 3
- 125000001493 tyrosinyl group Chemical group [H]OC1=C([H])C([H])=C(C([H])=C1[H])C([H])([H])C([H])(N([H])[H])C(*)=O 0.000 claims description 3
- 125000000291 glutamic acid group Chemical group N[C@@H](CCC(O)=O)C(=O)* 0.000 claims description 2
- 210000001519 tissue Anatomy 0.000 description 84
- -1 n-octyl Chemical group 0.000 description 21
- 108010067722 Dipeptidyl Peptidase 4 Proteins 0.000 description 12
- 239000006166 lysate Substances 0.000 description 12
- 102100025012 Dipeptidyl peptidase 4 Human genes 0.000 description 11
- 239000000654 additive Substances 0.000 description 10
- 238000002512 chemotherapy Methods 0.000 description 9
- 210000004923 pancreatic tissue Anatomy 0.000 description 9
- 210000002563 splenic artery Anatomy 0.000 description 9
- 102000004190 Enzymes Human genes 0.000 description 8
- 108090000790 Enzymes Proteins 0.000 description 8
- 238000002271 resection Methods 0.000 description 7
- 238000012216 screening Methods 0.000 description 7
- 125000003545 alkoxy group Chemical group 0.000 description 6
- 125000003277 amino group Chemical group 0.000 description 6
- 239000012634 fragment Substances 0.000 description 6
- 230000008595 infiltration Effects 0.000 description 6
- 238000001764 infiltration Methods 0.000 description 6
- CSCPPACGZOOCGX-UHFFFAOYSA-N Acetone Chemical compound CC(C)=O CSCPPACGZOOCGX-UHFFFAOYSA-N 0.000 description 5
- LFQSCWFLJHTTHZ-UHFFFAOYSA-N Ethanol Chemical compound CCO LFQSCWFLJHTTHZ-UHFFFAOYSA-N 0.000 description 5
- 239000000872 buffer Substances 0.000 description 5
- 238000007490 hematoxylin and eosin (H&E) staining Methods 0.000 description 5
- 238000011532 immunohistochemical staining Methods 0.000 description 5
- 239000000243 solution Substances 0.000 description 5
- 238000005507 spraying Methods 0.000 description 5
- 241000282412 Homo Species 0.000 description 4
- 125000002947 alkylene group Chemical group 0.000 description 4
- 125000003118 aryl group Chemical group 0.000 description 4
- 210000004204 blood vessel Anatomy 0.000 description 4
- 125000004432 carbon atom Chemical group C* 0.000 description 4
- 239000003795 chemical substances by application Substances 0.000 description 4
- 230000004044 response Effects 0.000 description 4
- 239000007787 solid Substances 0.000 description 4
- 102100036969 Dipeptidyl peptidase 9 Human genes 0.000 description 3
- 101710087005 Dipeptidyl peptidase 9 Proteins 0.000 description 3
- LYCAIKOWRPUZTN-UHFFFAOYSA-N Ethylene glycol Chemical compound OCCO LYCAIKOWRPUZTN-UHFFFAOYSA-N 0.000 description 3
- ZMXDDKWLCZADIW-UHFFFAOYSA-N N,N-Dimethylformamide Chemical compound CN(C)C=O ZMXDDKWLCZADIW-UHFFFAOYSA-N 0.000 description 3
- 208000009956 adenocarcinoma Diseases 0.000 description 3
- 235000001014 amino acid Nutrition 0.000 description 3
- 230000009400 cancer invasion Effects 0.000 description 3
- 230000008859 change Effects 0.000 description 3
- 238000006243 chemical reaction Methods 0.000 description 3
- 239000003153 chemical reaction reagent Substances 0.000 description 3
- 238000003745 diagnosis Methods 0.000 description 3
- 238000004090 dissolution Methods 0.000 description 3
- 230000002255 enzymatic effect Effects 0.000 description 3
- 238000011156 evaluation Methods 0.000 description 3
- 238000002156 mixing Methods 0.000 description 3
- 238000011227 neoadjuvant chemotherapy Methods 0.000 description 3
- 230000001575 pathological effect Effects 0.000 description 3
- 239000002904 solvent Substances 0.000 description 3
- 230000035899 viability Effects 0.000 description 3
- INZOTETZQBPBCE-NYLDSJSYSA-N 3-sialyl lewis Chemical compound O[C@H]1[C@H](O)[C@H](O)[C@H](C)O[C@H]1O[C@H]([C@H](O)CO)[C@@H]([C@@H](NC(C)=O)C=O)O[C@H]1[C@H](O)[C@@H](O[C@]2(O[C@H]([C@H](NC(C)=O)[C@@H](O)C2)[C@H](O)[C@H](O)CO)C(O)=O)[C@@H](O)[C@@H](CO)O1 INZOTETZQBPBCE-NYLDSJSYSA-N 0.000 description 2
- 241000282472 Canis lupus familiaris Species 0.000 description 2
- 108010022366 Carcinoembryonic Antigen Proteins 0.000 description 2
- 102100025475 Carcinoembryonic antigen-related cell adhesion molecule 5 Human genes 0.000 description 2
- IAZDPXIOMUYVGZ-UHFFFAOYSA-N Dimethylsulphoxide Chemical compound CS(C)=O IAZDPXIOMUYVGZ-UHFFFAOYSA-N 0.000 description 2
- 102100036968 Dipeptidyl peptidase 8 Human genes 0.000 description 2
- 101710087011 Dipeptidyl peptidase 8 Proteins 0.000 description 2
- 102000001301 EGF receptor Human genes 0.000 description 2
- 108060006698 EGF receptor Proteins 0.000 description 2
- 241000282326 Felis catus Species 0.000 description 2
- 206010016654 Fibrosis Diseases 0.000 description 2
- KFZMGEQAYNKOFK-UHFFFAOYSA-N Isopropanol Chemical compound CC(C)O KFZMGEQAYNKOFK-UHFFFAOYSA-N 0.000 description 2
- 241000124008 Mammalia Species 0.000 description 2
- 208000035346 Margins of Excision Diseases 0.000 description 2
- 239000002253 acid Substances 0.000 description 2
- 125000002252 acyl group Chemical group 0.000 description 2
- 239000012736 aqueous medium Substances 0.000 description 2
- 230000008901 benefit Effects 0.000 description 2
- 239000007853 buffer solution Substances 0.000 description 2
- 125000004122 cyclic group Chemical group 0.000 description 2
- 125000000753 cycloalkyl group Chemical group 0.000 description 2
- 206010012601 diabetes mellitus Diseases 0.000 description 2
- 238000005516 engineering process Methods 0.000 description 2
- 125000000816 ethylene group Chemical group [H]C([H])([*:1])C([H])([H])[*:2] 0.000 description 2
- 230000004761 fibrosis Effects 0.000 description 2
- 238000002189 fluorescence spectrum Methods 0.000 description 2
- 238000012632 fluorescent imaging Methods 0.000 description 2
- 239000008187 granular material Substances 0.000 description 2
- 208000014829 head and neck neoplasm Diseases 0.000 description 2
- 230000002055 immunohistochemical effect Effects 0.000 description 2
- 238000011503 in vivo imaging Methods 0.000 description 2
- 230000003902 lesion Effects 0.000 description 2
- QDLAGTHXVHQKRE-UHFFFAOYSA-N lichenxanthone Natural products COC1=CC(O)=C2C(=O)C3=C(C)C=C(OC)C=C3OC2=C1 QDLAGTHXVHQKRE-UHFFFAOYSA-N 0.000 description 2
- 239000007788 liquid Substances 0.000 description 2
- 238000005259 measurement Methods 0.000 description 2
- 230000002093 peripheral effect Effects 0.000 description 2
- 230000002980 postoperative effect Effects 0.000 description 2
- 239000000047 product Substances 0.000 description 2
- 235000018102 proteins Nutrition 0.000 description 2
- 102000004169 proteins and genes Human genes 0.000 description 2
- 108090000623 proteins and genes Proteins 0.000 description 2
- 125000003808 silyl group Chemical group [H][Si]([H])([H])[*] 0.000 description 2
- 239000012453 solvate Substances 0.000 description 2
- 238000010186 staining Methods 0.000 description 2
- 239000000126 substance Substances 0.000 description 2
- 238000006467 substitution reaction Methods 0.000 description 2
- 230000004083 survival effect Effects 0.000 description 2
- 239000003826 tablet Substances 0.000 description 2
- 238000012360 testing method Methods 0.000 description 2
- 238000012800 visualization Methods 0.000 description 2
- 108010058566 130-nm albumin-bound paclitaxel Proteins 0.000 description 1
- JNGRENQDBKMCCR-UHFFFAOYSA-N 2-(3-amino-6-iminoxanthen-9-yl)benzoic acid;hydrochloride Chemical compound [Cl-].C=12C=CC(=[NH2+])C=C2OC2=CC(N)=CC=C2C=1C1=CC=CC=C1C(O)=O JNGRENQDBKMCCR-UHFFFAOYSA-N 0.000 description 1
- ZCYVEMRRCGMTRW-UHFFFAOYSA-N 7553-56-2 Chemical group [I] ZCYVEMRRCGMTRW-UHFFFAOYSA-N 0.000 description 1
- ZGXJTSGNIOSYLO-UHFFFAOYSA-N 88755TAZ87 Chemical compound NCC(=O)CCC(O)=O ZGXJTSGNIOSYLO-UHFFFAOYSA-N 0.000 description 1
- 206010052747 Adenocarcinoma pancreas Diseases 0.000 description 1
- 206010006187 Breast cancer Diseases 0.000 description 1
- 208000026310 Breast neoplasm Diseases 0.000 description 1
- WKBOTKDWSSQWDR-UHFFFAOYSA-N Bromine atom Chemical group [Br] WKBOTKDWSSQWDR-UHFFFAOYSA-N 0.000 description 1
- 208000001333 Colorectal Neoplasms Diseases 0.000 description 1
- 108010016626 Dipeptides Proteins 0.000 description 1
- 208000000461 Esophageal Neoplasms Diseases 0.000 description 1
- 101710107035 Gamma-glutamyltranspeptidase Proteins 0.000 description 1
- WQZGKKKJIJFFOK-GASJEMHNSA-N Glucose Natural products OC[C@H]1OC(O)[C@H](O)[C@@H](O)[C@@H]1O WQZGKKKJIJFFOK-GASJEMHNSA-N 0.000 description 1
- 101710173228 Glutathione hydrolase proenzyme Proteins 0.000 description 1
- 238000000585 Mann–Whitney U test Methods 0.000 description 1
- 206010030155 Oesophageal carcinoma Diseases 0.000 description 1
- 101150114468 TUB1 gene Proteins 0.000 description 1
- 208000024770 Thyroid neoplasm Diseases 0.000 description 1
- ZMANZCXQSJIPKH-UHFFFAOYSA-N Triethylamine Chemical class CCN(CC)CC ZMANZCXQSJIPKH-UHFFFAOYSA-N 0.000 description 1
- 206010052428 Wound Diseases 0.000 description 1
- 238000002835 absorbance Methods 0.000 description 1
- 238000010521 absorption reaction Methods 0.000 description 1
- 238000000862 absorption spectrum Methods 0.000 description 1
- 125000001931 aliphatic group Chemical group 0.000 description 1
- 125000004453 alkoxycarbonyl group Chemical group 0.000 description 1
- 125000003282 alkyl amino group Chemical group 0.000 description 1
- 125000004414 alkyl thio group Chemical group 0.000 description 1
- 150000001408 amides Chemical class 0.000 description 1
- 150000001413 amino acids Chemical class 0.000 description 1
- 229960002749 aminolevulinic acid Drugs 0.000 description 1
- 150000003863 ammonium salts Chemical class 0.000 description 1
- 239000000427 antigen Substances 0.000 description 1
- 102000036639 antigens Human genes 0.000 description 1
- 108091007433 antigens Proteins 0.000 description 1
- 125000003710 aryl alkyl group Chemical group 0.000 description 1
- 125000001769 aryl amino group Chemical group 0.000 description 1
- 125000005110 aryl thio group Chemical group 0.000 description 1
- WQZGKKKJIJFFOK-VFUOTHLCSA-N beta-D-glucose Chemical compound OC[C@H]1O[C@@H](O)[C@H](O)[C@@H](O)[C@@H]1O WQZGKKKJIJFFOK-VFUOTHLCSA-N 0.000 description 1
- 230000015572 biosynthetic process Effects 0.000 description 1
- 210000000481 breast Anatomy 0.000 description 1
- KVUAALJSMIVURS-ZEDZUCNESA-L calcium folinate Chemical compound [Ca+2].C1NC=2NC(N)=NC(=O)C=2N(C=O)C1CNC1=CC=C(C(=O)N[C@@H](CCC([O-])=O)C([O-])=O)C=C1 KVUAALJSMIVURS-ZEDZUCNESA-L 0.000 description 1
- 159000000007 calcium salts Chemical class 0.000 description 1
- 125000002837 carbocyclic group Chemical group 0.000 description 1
- 125000003178 carboxy group Chemical group [H]OC(*)=O 0.000 description 1
- 150000007942 carboxylates Chemical class 0.000 description 1
- 230000000973 chemotherapeutic effect Effects 0.000 description 1
- 229910052801 chlorine Inorganic materials 0.000 description 1
- 125000001309 chloro group Chemical group Cl* 0.000 description 1
- 150000001860 citric acid derivatives Chemical class 0.000 description 1
- 210000002808 connective tissue Anatomy 0.000 description 1
- 238000009109 curative therapy Methods 0.000 description 1
- 125000000392 cycloalkenyl group Chemical group 0.000 description 1
- 125000002933 cyclohexyloxy group Chemical group C1(CCCCC1)O* 0.000 description 1
- 125000001887 cyclopentyloxy group Chemical group C1(CCCC1)O* 0.000 description 1
- 230000003247 decreasing effect Effects 0.000 description 1
- 125000004663 dialkyl amino group Chemical group 0.000 description 1
- 201000010099 disease Diseases 0.000 description 1
- 208000037265 diseases, disorders, signs and symptoms Diseases 0.000 description 1
- 239000012153 distilled water Substances 0.000 description 1
- 238000001839 endoscopy Methods 0.000 description 1
- 201000004101 esophageal cancer Diseases 0.000 description 1
- 125000001495 ethyl group Chemical group [H]C([H])([H])C([H])([H])* 0.000 description 1
- 229910052731 fluorine Inorganic materials 0.000 description 1
- 125000001153 fluoro group Chemical group F* 0.000 description 1
- 125000000524 functional group Chemical group 0.000 description 1
- 102000006640 gamma-Glutamyltransferase Human genes 0.000 description 1
- SDUQYLNIPVEERB-QPPQHZFASA-N gemcitabine Chemical compound O=C1N=C(N)C=CN1[C@H]1C(F)(F)[C@H](O)[C@@H](CO)O1 SDUQYLNIPVEERB-QPPQHZFASA-N 0.000 description 1
- 229960005277 gemcitabine Drugs 0.000 description 1
- 239000008103 glucose Substances 0.000 description 1
- 150000002333 glycines Chemical class 0.000 description 1
- 125000001072 heteroaryl group Chemical group 0.000 description 1
- 125000000623 heterocyclic group Chemical group 0.000 description 1
- 150000003840 hydrochlorides Chemical class 0.000 description 1
- 230000007062 hydrolysis Effects 0.000 description 1
- 238000006460 hydrolysis reaction Methods 0.000 description 1
- 125000002768 hydroxyalkyl group Chemical group 0.000 description 1
- 238000003384 imaging method Methods 0.000 description 1
- 125000002883 imidazolyl group Chemical group 0.000 description 1
- 238000000338 in vitro Methods 0.000 description 1
- 238000011534 incubation Methods 0.000 description 1
- MOFVSTNWEDAEEK-UHFFFAOYSA-M indocyanine green Chemical compound [Na+].[O-]S(=O)(=O)CCCCN1C2=CC=C3C=CC=CC3=C2C(C)(C)C1=CC=CC=CC=CC1=[N+](CCCCS([O-])(=O)=O)C2=CC=C(C=CC=C3)C3=C2C1(C)C MOFVSTNWEDAEEK-UHFFFAOYSA-M 0.000 description 1
- 229960004657 indocyanine green Drugs 0.000 description 1
- 239000003112 inhibitor Substances 0.000 description 1
- 239000007924 injection Substances 0.000 description 1
- 238000002347 injection Methods 0.000 description 1
- 229910052500 inorganic mineral Inorganic materials 0.000 description 1
- 230000002608 insulinlike Effects 0.000 description 1
- 229910052740 iodine Inorganic materials 0.000 description 1
- 125000000959 isobutyl group Chemical group [H]C([H])([H])C([H])(C([H])([H])[H])C([H])([H])* 0.000 description 1
- 125000004491 isohexyl group Chemical group C(CCC(C)C)* 0.000 description 1
- 125000001972 isopentyl group Chemical group [H]C([H])([H])C([H])(C([H])([H])[H])C([H])([H])C([H])([H])* 0.000 description 1
- 125000001449 isopropyl group Chemical group [H]C([H])([H])C([H])(*)C([H])([H])[H] 0.000 description 1
- 238000002372 labelling Methods 0.000 description 1
- 238000002357 laparoscopic surgery Methods 0.000 description 1
- 201000007270 liver cancer Diseases 0.000 description 1
- 208000014018 liver neoplasm Diseases 0.000 description 1
- 210000004072 lung Anatomy 0.000 description 1
- 210000001365 lymphatic vessel Anatomy 0.000 description 1
- 238000007433 macroscopic evaluation Methods 0.000 description 1
- 159000000003 magnesium salts Chemical class 0.000 description 1
- 239000000463 material Substances 0.000 description 1
- 229910052751 metal Inorganic materials 0.000 description 1
- 239000002184 metal Substances 0.000 description 1
- AFVFQIVMOAPDHO-UHFFFAOYSA-M methanesulfonate group Chemical class CS(=O)(=O)[O-] AFVFQIVMOAPDHO-UHFFFAOYSA-M 0.000 description 1
- 125000002496 methyl group Chemical group [H]C([H])([H])* 0.000 description 1
- 239000011707 mineral Substances 0.000 description 1
- 150000002780 morpholines Chemical class 0.000 description 1
- 125000002757 morpholinyl group Chemical group 0.000 description 1
- 229940126619 mouse monoclonal antibody Drugs 0.000 description 1
- 125000004108 n-butyl group Chemical group [H]C([H])([H])C([H])([H])C([H])([H])C([H])([H])* 0.000 description 1
- 125000003136 n-heptyl group Chemical group [H]C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])* 0.000 description 1
- 125000001298 n-hexoxy group Chemical group [H]C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])O* 0.000 description 1
- 125000001280 n-hexyl group Chemical group C(CCCCC)* 0.000 description 1
- 125000000740 n-pentyl group Chemical group [H]C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])* 0.000 description 1
- 125000004123 n-propyl group Chemical group [H]C([H])([H])C([H])([H])C([H])([H])* 0.000 description 1
- 125000001624 naphthyl group Chemical group 0.000 description 1
- 125000001971 neopentyl group Chemical group [H]C([*])([H])C(C([H])([H])[H])(C([H])([H])[H])C([H])([H])[H] 0.000 description 1
- 150000002823 nitrates Chemical class 0.000 description 1
- 230000003287 optical effect Effects 0.000 description 1
- 210000000056 organ Anatomy 0.000 description 1
- 239000003960 organic solvent Substances 0.000 description 1
- 150000003891 oxalate salts Chemical class 0.000 description 1
- 125000004043 oxo group Chemical group O=* 0.000 description 1
- 125000004430 oxygen atom Chemical group O* 0.000 description 1
- 229960001592 paclitaxel Drugs 0.000 description 1
- 201000002094 pancreatic adenocarcinoma Diseases 0.000 description 1
- 229910052698 phosphorus Inorganic materials 0.000 description 1
- 239000002504 physiological saline solution Substances 0.000 description 1
- 150000003053 piperidines Chemical class 0.000 description 1
- 159000000001 potassium salts Chemical class 0.000 description 1
- 239000000843 powder Substances 0.000 description 1
- 230000008569 process Effects 0.000 description 1
- 125000004805 propylene group Chemical group [H]C([H])([H])C([H])([*:1])C([H])([H])[*:2] 0.000 description 1
- 125000004076 pyridyl group Chemical group 0.000 description 1
- 125000000719 pyrrolidinyl group Chemical group 0.000 description 1
- 150000003254 radicals Chemical class 0.000 description 1
- 238000001959 radiotherapy Methods 0.000 description 1
- 239000002994 raw material Substances 0.000 description 1
- 102000005962 receptors Human genes 0.000 description 1
- 108020003175 receptors Proteins 0.000 description 1
- 238000012552 review Methods 0.000 description 1
- MYFATKRONKHHQL-UHFFFAOYSA-N rhodamine 123 Chemical compound [Cl-].COC(=O)C1=CC=CC=C1C1=C2C=CC(=[NH2+])C=C2OC2=CC(N)=CC=C21 MYFATKRONKHHQL-UHFFFAOYSA-N 0.000 description 1
- 238000005070 sampling Methods 0.000 description 1
- 229920006395 saturated elastomer Polymers 0.000 description 1
- 125000002914 sec-butyl group Chemical group [H]C([H])([H])C([H])([H])C([H])(*)C([H])([H])[H] 0.000 description 1
- 230000035945 sensitivity Effects 0.000 description 1
- 159000000000 sodium salts Chemical class 0.000 description 1
- 239000007858 starting material Substances 0.000 description 1
- 125000000020 sulfo group Chemical group O=S(=O)([*])O[H] 0.000 description 1
- 150000003467 sulfuric acid derivatives Chemical class 0.000 description 1
- 238000007910 systemic administration Methods 0.000 description 1
- RCINICONZNJXQF-MZXODVADSA-N taxol Chemical compound O([C@@H]1[C@@]2(C[C@@H](C(C)=C(C2(C)C)[C@H](C([C@]2(C)[C@@H](O)C[C@H]3OC[C@]3([C@H]21)OC(C)=O)=O)OC(=O)C)OC(=O)[C@H](O)[C@@H](NC(=O)C=1C=CC=CC=1)C=1C=CC=CC=1)O)C(=O)C1=CC=CC=C1 RCINICONZNJXQF-MZXODVADSA-N 0.000 description 1
- 125000000999 tert-butyl group Chemical group [H]C([H])([H])C(*)(C([H])([H])[H])C([H])([H])[H] 0.000 description 1
- JOXIMZWYDAKGHI-UHFFFAOYSA-N toluene-4-sulfonic acid Chemical class CC1=CC=C(S(O)(=O)=O)C=C1 JOXIMZWYDAKGHI-UHFFFAOYSA-N 0.000 description 1
- 238000011200 topical administration Methods 0.000 description 1
- 201000007423 tubular adenocarcinoma Diseases 0.000 description 1
- XLYOFNOQVPJJNP-UHFFFAOYSA-N water Chemical compound O XLYOFNOQVPJJNP-UHFFFAOYSA-N 0.000 description 1
Images
Classifications
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K49/00—Preparations for testing in vivo
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07K—PEPTIDES
- C07K5/00—Peptides containing up to four amino acids in a fully defined sequence; Derivatives thereof
- C07K5/04—Peptides containing up to four amino acids in a fully defined sequence; Derivatives thereof containing only normal peptide links
- C07K5/06—Dipeptides
- C07K5/06008—Dipeptides with the first amino acid being neutral
- C07K5/06017—Dipeptides with the first amino acid being neutral and aliphatic
-
- G—PHYSICS
- G01—MEASURING; TESTING
- G01N—INVESTIGATING OR ANALYSING MATERIALS BY DETERMINING THEIR CHEMICAL OR PHYSICAL PROPERTIES
- G01N21/00—Investigating or analysing materials by the use of optical means, i.e. using sub-millimetre waves, infrared, visible or ultraviolet light
- G01N21/62—Systems in which the material investigated is excited whereby it emits light or causes a change in wavelength of the incident light
- G01N21/63—Systems in which the material investigated is excited whereby it emits light or causes a change in wavelength of the incident light optically excited
- G01N21/64—Fluorescence; Phosphorescence
-
- G—PHYSICS
- G01—MEASURING; TESTING
- G01N—INVESTIGATING OR ANALYSING MATERIALS BY DETERMINING THEIR CHEMICAL OR PHYSICAL PROPERTIES
- G01N33/00—Investigating or analysing materials by specific methods not covered by groups G01N1/00 - G01N31/00
- G01N33/48—Biological material, e.g. blood, urine; Haemocytometers
Definitions
- the present invention relates to a fluorescent probe for pancreatic cancer detection and a method for detecting pancreatic cancer cells or cancer tissues using the same.
- Pancreatic cancer is one of the major life-threatening diseases (eg, Non-Patent Document 1).
- Non-Patent Document 1 Despite recent advances in chemotherapy and radiotherapy, complete resection of cancer tissue remains the mainstay of curative therapy (2-3).
- Non-Patent Document 4 it may be more difficult to identify viable cancer tissue even by pathological examination of resected specimens (Non-Patent Document 5).
- Urano, and collaborators reported a new fluorescence imaging technique using activatable probes.
- This probe is initially non-fluorescent, but emits a visible fluorescent signal upon hydrolysis by ⁇ -glutamyl transpeptidase overexpressed in cancer cells (Non-Patent Document 6). Since then, more than 400 activatable fluorescent probes consisting of fluorescent substances such as amino acids or glucose and hydroxymethylrhodamine green (HMRG) have been developed (7-8) and are used for breast, esophageal and liver cancer. , lung, head and neck, colorectal and thyroid cancers.
- HMRG hydroxymethylrhodamine green
- An object of the present invention is to provide a fluorescent probe that can specifically detect pancreatic cancer. Another object of the present invention is to provide a method for detecting pancreatic cancer cells or cancer tissue using the fluorescent probe.
- the present inventors collected cancer tissue fragments and non-cancerous pancreatic tissue fragments from resected pancreatic cancer specimens, prepared lysates, and used an enzyme probe library consisting of approximately 400 types of HMRG derivative probes to identify pancreatic cancer. As a result of searching for a fluorescent probe that can be specifically detected, the present inventors have found that an HMRG derivative probe having a specific structure can specifically detect pancreatic cancer, and completed the present invention.
- a fluorescent probe for detecting pancreatic cancer comprising a compound represented by the following general formula (I) or a salt thereof.
- R 1 represents a hydrogen atom or 1 to 4 identical or different substituents bonded to the benzene ring
- R2 , R3 , R4 , R5 , R6 , and R7 each independently represent a hydrogen atom, a hydroxyl group, an alkyl group, or a halogen atom
- R 8 , R 9 and R 10 each independently represent a hydrogen atom or an alkyl group
- X represents a C 1 -C 3 alkylene group
- A is a proline residue
- B is an amino acid residue selected from glycine, leucine, proline, tyrosine or N ⁇ -acetyl-lysine residues
- A is linked to NH in the adjacent formula by forming an amide bond
- B is linked to A by forming an amide bond.
- R1 , R2 , R3, R4 , R5, R6 , R7 , R8, R9 and R10 are hydrogen atoms, and X is a methylene group, [ 1 ] or [ 2].
- a fluorescent probe for detecting pancreatic cancer comprising a compound or a salt thereof selected from the following group.
- [6] A pancreatic cancer detection kit comprising the fluorescent probe of any one of [1] to [4].
- a composition for diagnosing pancreatic cancer comprising the fluorescent probe of any one of [1] to [4].
- the composition for diagnosing pancreatic cancer according to [7] which is used for cancer surgical treatment or cancer examination.
- the composition for diagnosing pancreatic cancer according to [8], wherein the cancer surgical treatment is open surgery or endoscopic surgery.
- a method for detecting cancerous cells or tissue in the pancreas comprising: [11] A method for detecting pancreatic cancer, comprising the steps of (a) applying the fluorescent probe according to any one of [1] to [4] to a clinical specimen of pancreatic cancer, and (b) The method, comprising measuring a fluorescence image of a clinical specimen of pancreatic cancer to which the fluorescent probe is applied.
- (a) applying a fluorescent probe containing a compound represented by the following general formula (I) or a salt thereof to a specimen surgically excised from the pancreas of a subject; and (b) the fluorescence A method of determining the presence of pancreatic cancer cells and/or identifying the extent of pancreatic cancer tissue in a subject comprising measuring a fluorescent image of a resected specimen to which a probe has been applied.
- R 1 represents a hydrogen atom or 1 to 4 identical or different substituents bonded to the benzene ring
- R2 , R3 , R4 , R5 , R6 , and R7 each independently represent a hydrogen atom, a hydroxyl group, an alkyl group, or a halogen atom
- R 8 , R 9 and R 10 each independently represent a hydrogen atom or an alkyl group
- X represents a C 1 -C 3 alkylene group
- A is a proline residue
- B is an amino acid residue selected from glycine, leucine, proline, tyrosine or N ⁇ -acetyl-lysine residues
- A is linked to NH in the adjacent formula by forming an amide bond
- B is linked to A by forming an amide bond.
- the present invention can provide a fluorescent probe that can specifically detect pancreatic cancer. Further, by using the fluorescent probe of the present invention, a method for detecting pancreatic cancer cells or cancer tissue can be provided.
- the fluorescent probe for detecting pancreatic cancer of the present invention enables real-time identification of viable pancreatic cancer tissue in human resected specimens.
- a method for determining the presence of pancreatic cancer cells in a subject and/or identifying the extent of pancreatic cancer tissue during surgical treatment of pancreatic cancer by using the fluorescent probes of the present invention can provide. This makes it possible to clearly distinguish cancerous tissue from surrounding non-cancerous tissue as a fluorescent region in a specimen surgically excised from the subject's pancreas.
- cancer infiltration that cannot be confirmed with the naked eye may occur around blood vessels such as the splenic artery. It is possible to visualize cancer infiltration around blood vessels that cannot be confirmed with the naked eye, which can reduce the amount of cancer left behind during surgery.
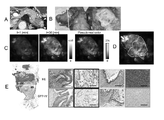
- FI enhancement of candidate probes in primary selection using lysates obtained from pancreatic cancer patients TBR based on FI increase 30 minutes after administration of five probes to tissue fragments is shown.
- Figure 2 shows the trend of FI increase in cancer and non-cancerous tissue fragments after GP-HMRG administration.
- Fluorescent imaging patient #2 on a whole surgical specimen using GP-HMRG showing a uniform increase in fluorescent signal in pancreatic cancer tissue.
- Fluorescent imaging (patient #6) on a whole surgical specimen using GP-HMRG showing a heterogeneous increase in fluorescent signal in pancreatic cancer tissue.
- Fluorescence imaging (patient #8) on a whole surgical specimen using GP-HMRG showing cancer invasion into the splenic artery. The time course of the fluorescence intensity of GP-HMRG upon addition of each enzyme is shown.
- halogen atom means a fluorine atom, a chlorine atom, a bromine atom, or an iodine atom.
- alkyl may be straight chain, branched chain, cyclic, or an aliphatic hydrocarbon group consisting of a combination thereof.
- the number of carbon atoms in the alkyl group is not particularly limited . ). When the number of carbon atoms is specified, it means “alkyl” having the number of carbon atoms within the specified range.
- C 1-8 alkyl includes methyl, ethyl, n-propyl, isopropyl, n-butyl, isobutyl, sec-butyl, tert-butyl, n-pentyl, isopentyl, neo-pentyl, n-hexyl, isohexyl, n-heptyl, n-octyl and the like are included.
- an alkyl group may have one or more optional substituents.
- substituents include, but are not limited to, alkoxy groups, halogen atoms, amino groups, mono- or di-substituted amino groups, substituted silyl groups, acyl groups, and the like.
- alkyl group When an alkyl group has more than one substituent, they may be the same or different.
- alkyl moieties of other substituents containing alkyl moieties eg, alkoxy groups, arylalkyl groups, etc.
- substituents include, but are not limited to, alkyl groups, alkoxy groups, hydroxyl groups, carboxyl groups, halogen atoms, sulfo groups, amino groups, alkoxycarbonyl groups, oxo groups, and the like. . These substituents may further have a substituent. Examples of such groups include, but are not limited to, halogenated alkyl groups, dialkylamino groups, and the like.
- alkoxy group refers to a structure in which the aforementioned alkyl group is bonded to an oxygen atom, and examples thereof include saturated alkoxy groups that are linear, branched, cyclic, or a combination thereof.
- methoxy, ethoxy, n-propoxy, isopropoxy, cyclopropoxy, n-butoxy, isobutoxy, s-butoxy, t-butoxy, cyclobutoxy, cyclopropylmethoxy, n- Pentyloxy group, cyclopentyloxy group, cyclopropylethyloxy group, cyclobutylmethyloxy group, n-hexyloxy group, cyclohexyloxy group, cyclopropylpropyloxy group, cyclobutylethyloxy group, cyclopentylmethyloxy group and the like are preferred. Examples include:
- alkylamino and arylamino mean an amino group in which the hydrogen atom of the -NH2 group is replaced with one or two of the above alkyl or aryl. Examples include methylamino, dimethylamino, ethylamino, diethylamino, ethylmethylamino, benzylamino and the like.
- alkylthio and arylthio mean groups in which a hydrogen atom of a --SH group is replaced with the above alkyl or aryl. Examples include methylthio, ethylthio, benzylthio and the like.
- ring structure means a heterocyclic or carbocyclic group when formed by the combination of two substituents, and such groups may be saturated, unsaturated, or aromatic. can be.
- substituents can form a ring structure with another substituent, and when such substituents are attached, the skilled artisan will appreciate the particular substitution, e.g. is formed.
- One embodiment of the fluorescent probe for detecting pancreatic cancer of the present invention is a fluorescent probe for detecting pancreatic cancer containing a compound represented by the following general formula (I) or a salt thereof (hereinafter referred to as "the fluorescent probe of the present invention (also called a probe).
- R 1 represents a hydrogen atom or 1 to 4 substituents bonded to a benzene ring.
- substituents include, but are not limited to, alkyl groups, alkoxy groups, halogen atoms, amino groups, mono- or di-substituted amino groups, substituted silyl groups, acyl groups, and the like.
- the benzene ring has two or more substituents, they may be the same or different.
- a hydrogen atom is preferred as R 1 .
- R2 , R3 , R4 , R5 , R6 , and R7 each independently represent a hydrogen atom, a hydroxyl group, an alkyl group, or a halogen atom. It is preferred that R 2 and R 7 are hydrogen atoms. It is also preferred that R 3 , R 4 , R 5 and R 6 are hydrogen atoms. More preferably, all of R 2 , R 3 , R 4 , R 5 , R 6 and R 7 are hydrogen atoms.
- R 8 , R 9 and R 10 each independently represent a hydrogen atom or an alkyl group.
- R 8 and R 9 represent an alkyl group, they may be the same or different.
- R 8 and R 9 are hydrogen atoms
- R 8 is an alkyl group and R 9 is preferably a hydrogen atom
- R 8 and R 9 are hydrogen atoms is more preferred.
- R 10 is preferably a hydrogen atom.
- X represents a C 1 -C 3 alkylene group.
- the alkylene group may be either a linear alkylene group or a branched alkylene group. Examples include a methylene group (--CH 2 --), an ethylene group (--CH 2 --CH 2 --), a propylene group (--CH 2 --CH 2 --CH 2 --), and a branched alkylene group --CH ( CH 3 )—, —CH 2 —CH(CH 3 )—, —CH(CH 2 CH 3 )—, and the like can also be used. Among these, a methylene group or an ethylene group is preferred, and a methylene group is more preferred.
- a in general formula (I) is a proline residue.
- a screening test was performed using an enzyme probe library consisting of about 400 kinds of HMRG derivative probes. A trend was found to show significant differences and ratios between FI increases in cancer and non-cancer lysates.
- the above Xaa (B in general formula (I)) is a glycine residue, a glutamic acid residue, a leucine residue, a proline residue, a tyrosine residue, or N ⁇ -acetyl-lysine. It was found to be an amino acid residue selected from residues. Furthermore, among these amino acid residues, B is preferably a glycine residue, a leucine residue, a proline residue, a tyrosine residue or an N ⁇ -acetyl-lysine residue, with a glycine residue being particularly preferred.
- A is linked to NH in the adjacent formula by forming an amide bond
- B is linked to A by forming an amide bond.
- the compound represented by general formula (I) may exist as a salt.
- Such salts include base addition salts, acid addition salts, amino acid salts and the like.
- base addition salts include metal salts such as sodium salts, potassium salts, calcium salts and magnesium salts, ammonium salts, or organic amine salts such as triethylamine salts, piperidine salts and morpholine salts.
- examples thereof include mineral acid salts such as hydrochlorides, sulfates and nitrates, and organic acid salts such as carboxylates, methanesulfonates, p-toluenesulfonates, citrates and oxalates.
- Glycine salt etc. can be illustrated as an amino acid salt. However, it is not limited to these salts.
- the compound represented by the general formula (I) may have one or more asymmetric carbon atoms depending on the type of substituent, and stereoisomers such as optical isomers or diastereoisomers are present. sometimes. All stereoisomers in pure form, any mixtures of stereoisomers, racemates, etc. are included within the scope of the present invention.
- the compound represented by general formula (I) or a salt thereof may exist as a hydrate or solvate, and any of these substances are included within the scope of the present invention.
- the type of solvent that forms the solvate is not particularly limited, but examples include solvents such as ethanol, acetone, and isopropanol.
- the compound represented by the general formula (I), for example, has amino groups at the 3- and 6-positions, using a xanthene compound having a 2-carboxyphenyl group or a 2-alkoxycarbonylphenyl group at the 9-position as a starting material.
- a xanthene compound having a 2-carboxyphenyl group or a 2-alkoxycarbonylphenyl group at the 9-position can be easily produced by converting a 2-carboxyphenyl group or a 2-alkoxycarbonylphenyl group at the 9-position to a hydroxyalkyl group and then acylating the amino group at the 3-position.
- Examples of the 3,6-diaminoxanthene compound that can be used as a raw material include, for example, Rhodamine 110 and Rhodamine 123, which are both commercially available. An appropriate xanthene compound can be selected accordingly.
- the fluorescent probe of the present invention may be used as a composition by blending additives that are commonly used for preparing reagents, if necessary.
- additives such as dissolution aids, pH adjusters, buffers, and tonicity agents can be used, and the blending amounts of these can be appropriately selected by those skilled in the art. be.
- These compositions can be provided as a composition in an appropriate form such as a mixture of powder forms, a freeze-dried product, granules, tablets, liquids, and the like.
- the compound represented by the general formula (I) or a salt thereof may be used as it is. may be used.
- additives for using the reagent in a physiological environment additives such as dissolution aids, pH adjusters, buffers, and tonicity agents can be used, and the amount of these additives can be appropriately selected by those skilled in the art. It is possible.
- These compositions are generally provided as compositions in suitable forms such as powdered mixtures, lyophilized products, granules, tablets, and liquids. can be applied by dissolving in
- the fluorescent probe of the present invention can be used, for example, during surgery, during examination, and after surgery.
- the term "surgery” includes any surgery, including endoscopic or laparoscopic surgery.
- the term “examination” includes examination using an endoscope, treatment such as excision and sampling of tissue accompanying the examination, and examination performed on tissue separated and collected from a living body. These terms should be interpreted in the broadest sense and not exclusive in any way.
- cancer tissue means any tissue containing cancer cells.
- tissue should be interpreted in the broadest sense, including a part or the whole of an organ, and should not be interpreted restrictively in any way.
- diagnosis should be interpreted in the broadest sense, including macroscopically or microscopically confirming the presence of cancerous tissue in any living body site.
- One aspect of the present invention is a composition for detecting pancreatic cancer, containing the fluorescent probe of the present invention.
- pancreatic cancer diagnostic composition comprising the fluorescent probe of the present invention.
- Another aspect of the present invention is a composition for diagnosing pancreatic cancer used for cancer surgical treatment or cancer examination, containing the fluorescent probe of the present invention.
- cancer surgical treatment includes open surgery and endoscopic surgery.
- Detection method using fluorescent probe comprises a step of applying the fluorescent probe of the present invention to a tissue taken from the pancreas of a subject; A step of irradiating the tissue after application with excitation light, and a step of detecting fluorescence from the tissue, A method for detecting pancreatic cancer cells or tissue comprising:
- the subject includes humans and mammals other than humans (eg, dogs, cats, etc.).
- step (a) In order to apply the fluorescent probe to the tissue collected from the pancreas of the subject in the above step (a), for example, using a lysate prepared from a cancerous or non-cancerous tissue sample, for example, wells of a 384 plate, etc. This includes, but is not limited to, applying to
- Another embodiment of the present invention is a method for detecting pancreatic cancer, comprising the steps of (a) applying the fluorescent probe of the present invention to a clinical sample of pancreatic cancer, and (b) the fluorescent probe measuring the fluorescence image of a clinical specimen of pancreatic cancer to which is applied.
- step (a) above can be performed, for example, by spraying a solution of the fluorescent probe locally or entirely onto the clinical sample.
- the detection method and detection method of the present invention can further include observing the fluorescence response using fluorescence imaging means.
- fluorescence imaging means As means for observing the fluorescence response, a fluorometer having a wide measurement wavelength can be used, but the fluorescence response can also be visualized using fluorescence imaging means capable of displaying a two-dimensional image. By using the means of fluorescence imaging, the fluorescence response can be visualized in two dimensions, so that cancer cells or tissues can be visualized instantaneously.
- a device known in the art can be used as the fluorescence imaging device.
- it is also possible to detect the reaction between the sample to be measured and the fluorescent probe by means of a change in the ultraviolet-visible absorption spectrum (for example, a change in absorbance at a specific absorption wavelength).
- the method of using the fluorescent probe of the present invention is not particularly limited, and it can be used in the same manner as conventionally known fluorescent probes.
- the compound of the present invention or a salt thereof is added to an aqueous medium such as physiological saline or a buffer solution, or a mixture of a water-miscible organic solvent such as ethanol, acetone, ethylene glycol, dimethylsulfoxide and dimethylformamide and an aqueous medium. is dissolved, the solution is added to an appropriate buffer containing cells or tissues, and the fluorescence spectrum is measured.
- the fluorescent probe of the present invention may be used in the form of a composition in combination with suitable additives.
- the concentration of the compound of the present invention in the fluorescent probe of the present invention can be determined appropriately according to the type of cells to be measured, the measurement conditions, and the like.
- Another embodiment of the present invention is a kit for detecting pancreatic cancer cells or tissues, containing the fluorescent probe of the present invention.
- the fluorescent probe of the present invention is usually prepared as a solution. It can also be applied by dissolving in distilled water for injection or an appropriate buffer solution.
- kit may contain other reagents and the like as necessary.
- additives such as dissolution aids, pH adjusters, buffers, tonicity agents, and the like can be used, and the amount of these additives can be appropriately selected by those skilled in the art.
- the applied concentration of the fluorescent probe of the present invention is not particularly limited, but for example, a solution with a concentration of about 0.1 to 10 ⁇ M can be applied.
- Another embodiment of the present invention comprises: Determining the presence of pancreatic cancer cells in a subject, comprising applying a fluorescent probe comprising the represented compound or a salt thereof, and (b) measuring a fluorescent image of a resection specimen to which said fluorescent probe is applied. and/or identifying the range of pancreatic cancer tissue (hereinafter also referred to as "the identification method of the present invention, etc.”).
- the subject includes humans and mammals other than humans (eg, dogs, cats, etc.).
- R 1 , R 2 , R 3 , R 4 , R 5 , R 6 , R 7 , X, A, and B are as described in detail above.
- B in general formula (I) is a glycine residue.
- R 1 , R 2 , R 3 , R 4 , R 5 , R 6 , R 7 , R 8 , R 9 and R 10 are hydrogen atoms and X is preferably a methylene group.
- the identification method and the like of the present invention can further include visualizing the fluorescence image using fluorescence imaging means.
- the details of the fluorescence imaging means are as described in the detection method and detection method of the present invention.
- the identification method, etc. of the present invention can be performed during surgical treatment of pancreatic cancer.
- surgical treatment of pancreatic cancer includes open wound surgery and endoscopic surgery.
- the fluorescent region is the surrounding cancer tissue. It becomes possible to distinguish clearly from non-cancerous tissue.
- the identification method and the like of the present invention it is possible to identify cancer tissue rapidly and in real time based on the viability of cancer cells. As a result, it is possible to reduce the number of pathological examinations of tissues suspected of having cancer during surgery.
- cancer infiltration that cannot be confirmed with the naked eye may occur around blood vessels such as the splenic artery. It is possible to visualize cancer infiltration that cannot be confirmed with the naked eye around blood vessels, and this can reduce the amount of cancer left behind during surgery.
- fluorescence imaging such as by the identification method of the present invention, can visualize the spread of viable pancreatic cancer cells in real time, which is useful for intraoperative diagnosis of surgical margins and preoperative endoscopic evaluation of intraductal lesions. It is also useful for
- tissue pieces were collected from resected specimens of patients who underwent radical resection for pancreatic cancer. (Informed consent was obtained in the previous case before collection)
- Non-Patent Documents 6 to 8 lysates prepared from tissue fragments of cancerous and non-cancerous regions were mixed with fluorescent probes, respectively, and the increase in fluorescence intensity was measured. , candidate fluorescent probes were selected. Briefly, 5 ⁇ L of lysate (protein concentration: 0.20 mg/dL) was dropped into wells of a black 384 plate containing 15 ⁇ L of a library of dipeptide-HMRG compounds (Non-Patent Document 8). Final concentrations of candidate probes and lysate proteins were 1.0 ⁇ M and 0.050 mg/dL, respectively.
- FI fluorescence intensity
- FI was calculated by extracting fluorescence images at 540 nm and subtracting the mean fluorescence value of the region of interest (ROI) at 30 min from the fluorescence intensity of the same region at 1 min.
- ROI region of interest
- TBR tumor/background ratio
- DPP-IV Dipeptidyl peptidase 4
- its analogous enzyme was considered to be the target enzyme overexpressed in pancreatic cancer tissue.
- F-7000 Hitachi (Tokyo)
- human recombinant DPP-IV 50 ⁇ L; D4943, Sigma-Aldrich
- DPP-VIII 1.0 ⁇ g; ab162872, abcam
- DPP-IX 1.0 ⁇ g ; ab79621, abcam
- Excitation and emission wavelengths were set at 495 and 525 nm, respectively.
- DPP-IV expression in cut surfaces of surgical resection specimens was assessed by immunohistochemical (IHC) staining.
- the antibody used was an anti-DPP-IV mouse monoclonal antibody (TA500733; Origene Technologies Inc. (Rockville, Md.)).
- Antigen retrieval was performed at 110° C. for 15 minutes.
- the concentration of anti-DPP-IV antibody was 1:100, and incubation was carried out at 4°C overnight.
- IHC staining results were evaluated by a pathologist (MT) blinded to the fluorescence imaging results.
- MT pathologist
- Example 1 Primary and secondary probe screening 5
- a fluorescent probe (XaaP-HMRG) consisting of a dipeptide with a proline at the C-terminus that binds to HMRG with amide bonds was used to detect cancer.
- Fig. 1A There was a tendency for the difference and the increase ratio to be large between the increase in FI and the non-cancer lysate (Fig. 1A).
- five probes (AcKP-, GP-, LP-, PP-, and YP-HMRG) were selected as candidate probes and advanced to secondary screening.
- secondary screening cancerous and non-cancerous tissue pieces were collected from 11 patients.
- FIG. 1 shows the FI enhancement of candidate probes in primary and secondary screens using lysates and tissue sections obtained from pancreatic cancer patients.
- fluorescence imaging using lysates from five patients detected six types of probes (AcKP-, EP -, GP-, LP-, PP-, and YP-HMRG) showed a significant increase in FI in cancer tissue (C, solid line) in contrast to non-cancerous tissue (N, dotted line).
- FIG. 1B shows the FI increase 30 minutes after each probe administration for the tissue strips.
- the plot on the left is the result for cancerous tissue and the plot on the right is the result for non-cancerous tissue.
- the FI increase ratio of GP-HMRG cancer/non-cancerous area was AcKP-HMRG (0.95-1.36), LP-HMRG (1.10-2.38), PP-HMRG (2.37- 3.20) and among the remaining candidates including YP-HMRG (1.62-3.01) (range, 2.70-6.10). Bars indicate median values.
- FIG. 1C shows changes in FI increase over time in cancer and non-cancerous tissue fragments after administration of GP-HMRG.
- GP-HMRG was selected as a pancreatic cancer-labeling fluorescent probe, and the ability of fluorescence imaging to detect cancer was evaluated using surgically resected specimens.
- Example 2 Fluorescence Imaging of Whole Surgical Specimens Using GP-HMRG The cancer detectability of fluorescence imaging was evaluated by spraying GP-HMRG on cut surfaces of whole surgical specimens of 8 pancreatic adenocarcinoma patients immediately after resection. Patient backgrounds are summarized in Table 1. Neoadjuvant chemotherapy (NAC) with gemcitabine and nab-paclitaxel was indicated in two patients. Three patients were treated for diabetes but did not receive a DPP-IV inhibitor preoperatively.
- DM diabetes
- NAC preoperative chemotherapy
- PD pancreaticoduodenectomy
- DP tail pancreatectomy
- DP-CAR tail pancreatectomy pancreatectomy with celiac axis resection
- TBR tumor/background ratio
- tub 1/tub 2 well/moderately differentiated tubular adenocarcinoma adenocarcinoma
- por poorly differentiated adenocarcinoma
- the average TBR of fluorescence images 30 minutes after GP-HMRG spraying was 1.96 (range, 1.31-2.04).
- patient numbers: 1, 2, 4 and 5 with TBR ranging from 1.93 to 3.10, the fluorescence signal of the cancer tissue was nearly uniform and macroscopically distinct from the surrounding pancreatic tissue. were identifiable (Fig. 2).
- FIG. 2 shows fluorescence imaging (patient #2) on a whole surgical specimen using GP-HMRG showing a uniform increase in fluorescence signal in pancreatic cancer tissue.
- FIG. 2A shows preoperative contrast-enhanced CT (arrow) of pancreatic body cancer.
- FIG. 2B shows the macroscopic image OR of the DP specimen after making a section of the tumor, and C is an enlarged view of the section containing the tumor.
- FIG. 2D shows the increase in fluorescence signal after spraying GP-HMRG on the cut surface.
- FIG. 2E shows a fluorescence image (left) and a pseudo-real color image (right) of the cut surface 30 minutes after probe administration.
- FIG. 2F shows the relationship between fluorescence signal and distribution of cancer tissue (solid white line) and surrounding pancreatic tissue (dotted white line) based on the macroscopic image (B) of the sample.
- FIG. 2G shows a low-magnification histopathological image of hematoxylin-eosin (H&E) staining corresponding to the fluorescent image (left, dashed lines indicate cancer borders). Magnifications of H&E and IHC staining of DPP-IV in cancer tissue (red) and pancreatic tissue (blue) are also shown (right). Scale bar is 100 ⁇ m.
- FIG. 3 shows fluorescence imaging (patient #6) on a whole surgical specimen using GP-HMRG showing a heterogeneous increase in fluorescence signal in pancreatic cancer tissue.
- FIG. 3A shows preoperative contrast-enhanced CT (arrow) of pancreatic head cancer.
- FIG. 3B shows a gross image of a PD specimen (left) and a section along the dotted line containing the tumor (right).
- FIG. 3C shows the increase in fluorescence signal after sprinkling GP-HMRG on the cut surface.
- FIG. 3D shows the relationship between fluorescence signals and distribution of cancer tissue (solid white line) and peripheral pancreatic tissue (dotted white line) based on the macroscopic image (B) of the sample.
- FIG. 3E is a low-magnification histopathological image of H&E staining corresponding to the fluorescence image (left, dotted line indicates cancer border). Magnifications of DPP-IV H&E and IHC staining in cancer tissue (solid white line) and pancreatic tissue (dotted white line) are also shown (right). Scale bar is 100 ⁇ m.
- FIG. 4 shows fluorescence imaging (patient #8) on a whole surgical specimen using GP-HMRG showing cancer invasion into the splenic artery.
- FIG. 4A shows preoperative contrast-enhanced CT (arrow) of pancreatic body cancer.
- Figure 4B is a macroscopic image of a DP specimen (left) and a section along the dotted line containing the tumor (right). indicates
- FIG. 4C shows the increase in fluorescence signal after spraying GP-HMRG on the cut surface and the pseudo-real color image at 30 minutes.
- FIG. 4D shows the relationship between fluorescence signals and distribution of cancer tissue (solid white line) and peripheral pancreatic tissue (dotted white line) based on the macroscopic image (B) of the sample. Arrows indicate the splenic artery.
- FIG. 4A shows preoperative contrast-enhanced CT (arrow) of pancreatic body cancer.
- Figure 4B is a macroscopic image of a DP specimen (left) and a section along the dotted line
- 4E shows a low-magnification histopathological image of H&E staining corresponding to the fluorescence image (left, dashed lines indicate cancer borders). Fluorescent (red) and minor fluorescent (black) portions of the main tumor by DPP-IV H&E and IHC staining, viable cancer infiltrates around the splenic arteries (green), and enlarged views of non-cancerous pancreatic tissue (blue) are also shown. shown (right). Scale bar is 100 ⁇ m.
- FIG. 5 shows the time course of fluorescence intensity of GP-HMRG when each enzyme was added. Excitation/emission wavelengths were 495 nm/525 nm.
- FIG. 5A shows the result of adding DPP-IV
- FIG. 5B shows the result of adding DPP-VIII
- FIG. 5C shows the result of adding DPP-IX.
- IHC staining on resected samples from 8 patients showed no obvious difference in the expression level of DPP-IV between cancer tissue and surrounding pancreatic tissue (Table 1 and Table 1). 2-4).
- fluorescence imaging with GP-HMRG has the potential to visualize the spread of viable pancreatic cancer cells in real time, which is useful for intraoperative diagnosis of surgical margins and preoperative endoscopy of intraluminal lesions. It suggests that it is also useful for mirror evaluation.
- a major advantage of using activatable probes is that it allows rapid and real-time identification of cancerous tissue based on enzymatic activity, viability of cancer cells. Indeed, the present study confirmed an increase in fluorescence signal in cancer tissue 1 minute after topical administration of GP-HMRG, and furthermore, FI in cancer tissue decreased, possibly as a result of fibrosis and mucinous changes due to preoperative chemotherapy.
- a fluorescence imaging technique has also been developed for intraoperative identification of pancreatic cancer using a novel fluorophore that targets the factor 1 receptor (IGF-1R).
- these techniques which are based on systemic administration of 'deactivatable' probes, usually require longer intervals for washout of the fluorescent agent from background tissue, which can be activated by intraoperative local administration. can result in a lower TBR compared to the use of simple probes.
- the technology also has potential advantages in elucidating cancer tissue viability and enzymatic activity, which may allow prediction of chemotherapeutic sensitivity and postoperative outcome.
- fluorescence imaging using GP-HMRG enables rapid and real-time visualization of pancreatic cancer based on the enzymatic activity of cancer tissue.
Abstract
Description
しかしながら、手術中の癌の広がりの境界を正確に同定することはしばしば困難であり、このことから癌組織の不完全な除去および好ましくない術後生存につながる可能性がある(非特許文献2~3)。特に術前化学療法(非特許文献4)を受けている患者では、切除標本の病理学的検査によっても生存可能な癌組織の同定がより困難になる可能性がある(非特許文献5)。
[1] 以下の一般式(I)で表される化合物又はその塩を含む、すい臓がん検出用蛍光プローブ。
(式中、
R1は水素原子又はベンゼン環に結合する1~4個の同一又は異なる置換基を表し;
R2、R3、R4、R5、R6、及びR7は、それぞれ独立して、水素原子、ヒドロキシル基、アルキル基、又はハロゲン原子を表し;
R8、R9及びR10は、それぞれ独立して、水素原子又はアルキル基を示し;
XはC1-C3アルキレン基を表し;
Aは、プロリン残基であり;
Bは、グリシン残基、ロイシン残基、プロリン残基、チロシン残基又はNα-アセチル-リシン残基から選択されるアミノ酸残基であり;
ここで、Aは隣接する式中のNHとアミド結合を形成して連結し、BはAとアミド結合を形成して連結している。)
[2]Bがグリシン残基である、[1]に記載の蛍光プローブ。
[3]R1、R2、R3、R4、R5、R6、R7、R8、R9及びR10が水素原子であり、Xがメチレン基である、[1]又は[2]に記載の蛍光プローブ。
[4]以下の群から選択される化合物又はその塩を含む、すい臓がん検出用蛍光プローブ。
[5][1]~[4]のいずれか1項に記載の蛍光プローブを含む、すい臓がん検出用組成物。
[6][1]~[4]のいずれか1項に記載の蛍光プローブを含む、すい臓がん検出用キット。
[7][1]~[4]のいずれか1項に記載の蛍光プローブを含む、すい臓がん診断用組成物。
[8]がん外科治療又はがん検査に用いられる[7]に記載のすい臓がん診断用組成物。
[9]前記がん外科治療が、開創手術又は鏡視下手術である[8]に記載のすい臓がん診断用組成物。
[10][1]~[4]のいずれか1項に記載の蛍光プローブを、被検体のすい臓から採取した組織に適用する工程、
適用後の前記組織に対して励起光を照射する工程、及び
前記組織からの蛍光を検出する工程、
を含む、すい臓のがん細胞またはがん組織の検出方法。
[11]すい臓がんを検知する方法であって、(a)[1]~[4]のいずれか1項に記載の蛍光プローブをすい臓がんの臨床検体に適用する工程、及び(b)前記蛍光プローブを適用したすい臓がんの臨床検体の蛍光像を測定することを含む、前記方法。
[12]蛍光イメージング手段を用いて前記蛍光像を可視化することを更なる特徴とする、[11]に記載の方法。
[13](a)被検体のすい臓から外科的に切除された標本に、以下の一般式(I)で表される化合物又はその塩を含む蛍光プローブを適用する工程、及び(b)前記蛍光プローブを適用した切除標本の蛍光像を測定することを含む、被検体におけるすい臓がん細胞の存在を決定する、及び/又は、すい臓がん組織の範囲を同定する、方法。
(式中、
R1は水素原子又はベンゼン環に結合する1~4個の同一又は異なる置換基を表し;
R2、R3、R4、R5、R6、及びR7は、それぞれ独立して、水素原子、ヒドロキシル基、アルキル基、又はハロゲン原子を表し;
R8、R9及びR10は、それぞれ独立して、水素原子又はアルキル基を示し;
XはC1-C3アルキレン基を表し;
Aは、プロリン残基であり;
Bは、グリシン残基、ロイシン残基、プロリン残基、チロシン残基又はNα-アセチル-リシン残基から選択されるアミノ酸残基であり;
ここで、Aは隣接する式中のNHとアミド結合を形成して連結し、BはAとアミド結合を形成して連結している。)
[14]Bがグリシン残基である、[13]に記載の方法。
[15]R1、R2、R3、R4、R5、R6、R7、R8、R9及びR10が水素原子であり、Xがメチレン基である、[13]又は[14]に記載の方法。
[16]蛍光イメージング手段を用いて前記蛍光像を可視化することを更なる特徴とする、[13]~[15]のいずれか1項に記載の方法。
[17][13]~[16]のいずれか1項に記載の方法が、すい臓がんの外科治療中に行われる、被検体におけるすい臓がん細胞の存在を決定する、及び/又は、すい臓がん組織の範囲を同定する、方法。
[18]前記すい臓がんの外科治療が、開創手術又は鏡視下手術である、[17]に記載の方法。
を提供するものである。
本発明のすい臓がん検出用蛍光プローブにより、ヒト切除標本における生存すい臓癌組織のリアルタイム同定が可能である。
また、術前化学療法を受けた患者では、脾動脈等の血管の周囲で肉眼では確認できない癌浸潤が発生することがあるが、本発明の同定方法を用いると、蛍光イメージングにより脾動脈等の血管の周囲の肉眼では確認できない癌浸潤の可視化が可能であり、これにより手術中のがんの取り残しを低減することが可能である。
本明細書中において、「ハロゲン原子」とは、フッ素原子、塩素原子、臭素原子、又はヨウ素原子を意味する。
また、R10は水素原子であることが好ましい。
ここで、Aは隣接する式中のNHとアミド結合を形成して連結し、BはAとアミド結合を形成して連結している。
ここで、がん外科治療には、開創手術及び鏡視下手術が含まれる。
本発明のもう1つの実施態様は、本発明の蛍光プローブを、被検体のすい臓から採取した組織に適用する工程、
適用後の前記組織に対して励起光を照射する工程、及び
前記組織からの蛍光を検出する工程、
を含む、すい臓のがん細胞またはがん組織の検出方法である。
ここで、被検体には、ヒト、及びヒト以外の哺乳類(例えば、イヌ、ネコ等)が含まれる。
また、本発明の蛍光プローブ中の本発明の化合物の濃度は、測定する細胞等の種類や測定条件等に応じて適切に定めることができる。
本発明のもう1つの実施態様は、(a)被検体のすい臓から外科的に切除された標本に、以下の一般式(I)で表される化合物又はその塩を含む蛍光プローブを適用する工程、及び(b)前記蛍光プローブを適用した切除標本の蛍光像を測定することを含む、被検体におけるすい臓がん細胞の存在を決定する、及び/又は、すい臓がん組織の範囲を同定する、方法である(以下「本発明の同定方法等」とも言う)。
ここで、被検体には、ヒト、及びヒト以外の哺乳類(例えば、イヌ、ネコ等)が含まれる。
本研究は東京大学医学部附属病院治験審査委員会(IRB No.2957- [11])により承認された。
2017年4月から2020年12月までの期間に、膵癌に対し根治的切除術を施行した患者の切除標本から、組織片を採取した。(採取にあたり、前例でインフォームドコンセントを行い、同意を得た)
最初に、非特許文献6~8に記載されている手順に則って、癌部・非癌部の組織片から作成したライセートをそれぞれ蛍光プローブと混合して蛍光強度の上昇値を測定し、候補となる蛍光プローブを選定した。概要を述べると、ジペプチド-HMRG化合物のライブラリー(非特許文献8)15μLが配列された黒色384プレートのウェルへ、5μLのライセート(蛋白質濃度:0.20mg/dL)を滴下した。候補プローブ及びライセート蛋白質の最終濃度はそれぞれ1.0μM及び0.050mg/dLであった。
各試料の蛍光強度(FI)は、37℃でインキュベートしながら、Envision Multilabel Plate Reader(PerkinElmer(米国マサチューセッツ州))により、混合後0分後と60分後を測定した。励起及び発光波長はそれぞれ485nm及び535nmに設定した。FI増加は以下のように定義した。
(FI増加)=(60分後のFI)-(0分後のFI)
癌および非癌ライセートにおけるFI増加の差または比が上位10%にあったプローブを候補プローブとして選定し、次の評価を行った。
癌部および非癌部組織の組織片を8ウェルプレートに配置し、候補蛍光プローブを直接散布した。各蛍光プローブの濃度および量は、50μMおよび200μLとした。蛍光プローブの投与後0分(前)、1分、3分、5分、10分、15分、20分、25分及び30分に蛍光画像を撮像した。蛍光画像はMaestro in Vivo画像システム(PerkinElmer(米国マサチューセッツ州))により取得し、撮影条件は485~480nmおよび490nmの励起および発光波長とした。FIは、540nmでの蛍光画像を抽出し、30分での関心領域(ROI)の平均蛍光値から1分での同領域の蛍光地を差し引くことにより計算した。癌と非癌組織間でFIの差最大であったものを膵癌標識蛍光プローブとして選定した。
膵癌検体を摘出した直後に、すい臓癌組織の最大径を含むように切断した。次に、前述の蛍光プローブ(4mL、50μM)を切断面に直接散布し、前述のように、Maestro in Vivoイメージングシステムを用いて蛍光イメージングを行った。蛍光イメージングの精度は、外科医(RT)と病理医(MT)が、同じ面の病理組織学的所見を参照して評価した。加えて、腫瘍/バックグラウンド比(TBR)も、切断面のマクロ/顕微鏡所見と対応する蛍光画像に基づいて、癌部と非癌部におけるプローブ投与後1分から30分までの平均FIの増加を測定することにより算出した。
ジペプチジルペプチダーゼ4(DPP-IV)あるいはその類似酵素がすい臓癌組織で過剰発現している標的酵素であると考えられた。まず候補蛍光プローブをDPP-IVや類似酵素が活性化するかを調べた。F-7000(日立(東京都))を用いて、ヒト組換えDPP-IV(50μL;D4943、Sigma-Aldrich)、DPP-VIII (1.0μg;ab162872、abcam)、DPP-IX(1.0μg;ab79621、abcam)を3.0mLのプローブ溶液(1.0μM)中に注入し、2,000秒間のFIの変化を測定した。励起及び発光波長はそれぞれ495及び525nmに設定した。
さらに、外科的切除標本の切断面におけるDPP-IVの発現を、免疫組織化学(IHC)染色によって評価した。抗体は、抗DPP-IVマウスモノクローナル抗体(TA500733;Origene Technologies Inc.(メリーランド州ロックビル))を用いた。抗原賦活化は110℃で15分間行った。抗DPP-IV抗体の濃度は1:100とし、インキュベーションは4℃、Overnightで行った。IHC染色の結果は、蛍光イメージングの結果を盲検化して、病理学者(MT)が評価した。
一次及び二次プローブスクリーニング
5切除検体から作成したライセートと309種類の候補プローブを用いた一次選択では、HMRGとアミド結合するC末端にプロリンを持つジペプチドから成る蛍光プローブ(XaaP-HMRG)が、癌と非癌ライセートにおけるFI増加の間で差や増加比が大きい傾向を示した(図1A)。これらの中で、5つのプローブ(AcKP-、GP-、LP-、PP-、及びYP-HMRG)を、候補プローブとして選定し2次スクリーニングへ進んだ。2次スクリーニングでは、11人の患者から癌部・非癌部組織片を採取した。
具体的には、図1Aで示すように、5人の患者から得たライセートを用いた蛍光イメージングは、HMRGとアミド結合するC末端にプロリンを持つジペプチドから成る6種類のプローブ(AcKP-、EP-、GP-、LP-、PP-、及びYP-HMRG)において、非癌組織(N、点線)とは対照的に癌組織(C、実線)においてFIの有意な増加を示した。
GP-HMRGの癌部/非癌部のFI増加比は、AcKP-HMRG(0.95~1.36)、LP-HMRG(1.10~2.38)、PP-HMRG(2.37~3.20)及びYP-HMRG(1.62~3.01)を含む残りの候補の中でも、最も高かった(範囲、2.70-6.10)。バーは中央値を示す。
また、図1Cは、GP-HMRG投与後の癌及び非癌組織断片における経時的なFI増加の推移を示す。
GP-HMRGを用いた全外科標本の蛍光イメージング
すい臓腺癌患者8名の切除直後の全外科標本の切断面にGP-HMRGをスプレーすることにより、蛍光イメージングの癌検出能を評価した。患者背景を表1にまとめた。ゲムシタビンとナブパクリタキセルによる術前補助化学療法(NAC)が2人の患者で適応となった。3名の患者は糖尿病で治療されたが、術前にDPP-IV阻害剤は投与されなかった。
DM:糖尿病、NAC:術前化学療法、PD:膵頭十二指腸切除術、DP:膵体尾部切除術、DP-CAR:膵体尾部切除術
pancreatectomy with celiac axis resection:腹腔動脈幹合併膵体尾部切除術
TBR:腫瘍/バックグラウンド比、tub 1/tub 2:高/中分化型管状腺癌
adenocarcinoma, por:poorly differentiated adenocarcinoma(低分化腺癌)
*:Evans分類に基づく
**:脾動脈への神経周囲およびリンパ管への生存癌細胞の浸潤
図2のAは、膵体部癌の術前造影CT(矢印)を示す。
図2のBは、腫瘍の割面を作成した後のDP標本の肉眼の画像ORを示し、Cは腫瘍を含む切断面の拡大図である。
図2のDは、切断面にGP-HMRGをスプレーした後の蛍光シグナルの増加を示す。
図2のEは、プローブ投与30分後の切断面の蛍光画像(左)と擬似リアルカラー画像(右)を示す。
図2のFは、試料の肉眼画像(B)に基づく癌組織(白実線)および周辺すい臓組織(白点線)の蛍光シグナルと分布の間の関係を示す。
図2のGは、蛍光画像(左,点線はがん境界を示す)に対応するヘマトキシリン-エオジン(H&E)染色の低倍率組織病理学的画像を示す。癌組織(赤)およびすい臓組織(青)におけるDPP-IVのH&E染色およびIHC染色の拡大図も示す 右)。スケールバーは、100μmである。
図3は、すい臓癌組織における蛍光シグナルの不均一な増加を示す、GP-HMRGを用いた全外科標本上の蛍光イメージング(患者番号6)を示す。
図3のAは、膵頭部癌の術前造影CT(矢印)を示す。
図3のBは、PD標本の肉眼像(左)および腫瘍を含む点線に沿った切断面(右)を示す。
図3のCは、切断面にGP-HMRGを散布した後の蛍光シグナルの増加を示す。
図3のDは、試料の肉眼像(B)に基づく癌組織(白実線)および周辺すい臓組織(白点線)の蛍光シグナルと分布の関係を示す。
図3のEは、蛍光画像に対応するH&E染色の低倍率病理組織像(左,点線はがん境界を示す)。癌組織(白実線)およびすい臓組織(白点線)におけるDPP-IVのH&E染色およびIHC染色の拡大図も示す(右)。スケールバーは、100μmである。
図4のAは、膵体部癌の術前造影CT(矢印)を示す。
図4のBは、DP標本の肉眼の画像(左)および腫瘍を含む点線に沿った切断面(右)
を示す。
図4のCは、GP-HMRGを切断面にスプレーした後の蛍光シグナルの増加と30分での擬似リアルカラー画像を示す。
図4のDは、試料の肉眼画像(B)に基づく癌組織(白実線)および周辺すい臓組織(白点線)の蛍光シグナルと分布の関係を示す。矢印は脾動脈を示す。
図4のEは、蛍光画像に対応するH&E染色の低倍率病理組織像(左、点線はがん境界を示す)を示す。DPP-IVのH&E染色およびIHC染色による主腫瘍の蛍光(赤)および少量の蛍光(黒)部分、脾動脈周囲の生存癌浸潤(緑)、および非癌性すい臓組織(青)の拡大図も示している(右)。スケールバーは、100μmである。
GP-HMRGを活性化する標的酵素の同定
DPP添加後のGP-HMRGのin vitro蛍光スペクトルは、プローブがDPP-IVおよびDPP-IXとの反応で高蛍光HMRGに変換されることを示した(図5)。
ここで、図5は、各酵素添加時のGP-HMRGの蛍光強度の経時変化を示す。励起/発光波長は495nm/525nmであった。図5のAは DPP-IV、図5のBはDPP-VIII、図5のCは DPP-IXを添加した結果である。
一方、上記の結果とは異なり、8人の患者の切除した試料でのIHC染色は、癌組織と周辺すい臓組織の間のDPP-IVの発現量の明らかな違いを示さなかった(表1及び図2~4)。
本研究では、活性化可能な309種類の蛍光プローブの候補を用いてスクリーニングを行い、すい臓癌組織の同定のためにGP-HMRGを選定した。新鮮切除標本の切断面上のGP-HMRGを用いた蛍光イメージングは、8名の患者のうち4名において高い(>1.9)TBRを有する均一な蛍光領域として癌組織を可視化した。残りの4例では、癌組織の蛍光シグナルは不均一であり、癌組織での蛍光シグナルは非癌組織よりも高いものの、周囲の非癌組織との明確な識別には不十分であった。しかしながら、術前化学療法を受けた1人の患者では、蛍光イメージングにより脾動脈周囲の肉眼では確認できない癌浸潤の可視化が可能となった。これらの結果は、GP-HMRGを用いた蛍光イメージングが、viableなすい臓癌細胞の広がりをリアルタイムで可視化する可能性を有し、これは外科的切除縁の術中診断および管内病変の術前内視鏡評価にも有用であることを示唆する。
最近、いくつかの研究では、5-アミノレブリン酸、インドシアニングリーン、および炭水化物抗原19-9(CA19-9)、癌胎児性抗原(CEA)、上皮成長因子受容体(EGFR)、およびインスリン様成長因子1受容体(IGF-1R)を標的とする新規フルオロフォアを用いたすい臓癌の術中同定のための蛍光イメージング技術も開発された。
しかしながら、「非活性化可能な」プローブの全身投与に基づくこれらの技術は、通常、バックグラウンド組織からの蛍光剤のウォッシュアウトのためにより長い間隔を必要とし、それは、術中局所投与による活性化可能なプローブの使用と比較して、より低いTBRをもたらし得る。これに対して、本技術は癌組織の生存性と酵素活性の解明にも潜在的な利点があり、化学療法に対する感受性と術後転帰の予測を可能にし得る。
Claims (18)
- 以下の一般式(I)で表される化合物又はその塩を含む、すい臓がん検出用蛍光プローブ。
(式中、
R1は水素原子又はベンゼン環に結合する1~4個の同一又は異なる置換基を表し;
R2、R3、R4、R5、R6、及びR7は、それぞれ独立して、水素原子、ヒドロキシル基、アルキル基、又はハロゲン原子を表し;
R8、R9及びR10は、それぞれ独立して、水素原子又はアルキル基を示し;
XはC1-C3アルキレン基を表し;
Aは、プロリン残基であり;
Bは、グリシン残基、ロイシン残基、プロリン残基、チロシン残基又はNα-アセチル-リシン残基から選択されるアミノ酸残基であり;
ここで、Aは隣接する式中のNHとアミド結合を形成して連結し、BはAとアミド結合を形成して連結している。) - Bがグリシン残基である、請求項1に記載の蛍光プローブ。
- R1、R2、R3、R4、R5、R6、R7、R8、R9及びR10が水素原子であり、Xがメチレン基である、請求項1又は2に記載の蛍光プローブ。
- 請求項1~4のいずれか1項に記載の蛍光プローブを含む、すい臓がん検出用組成物。
- 請求項1~4のいずれか1項に記載の蛍光プローブを含む、すい臓がん検出用キット。
- 請求項1~4のいずれか1項に記載の蛍光プローブを含む、すい臓がん診断用組成物。
- がん外科治療又はがん検査に用いられる請求項7に記載のすい臓がん診断用組成物。
- 前記がん外科治療が、開創手術又は鏡視下手術である請求項8に記載のすい臓がん診断用組成物。
- 請求項1~4のいずれか1項に記載の蛍光プローブを、被検体のすい臓から採取した組織に適用する工程、
適用後の前記組織に対して励起光を照射する工程、及び
前記組織からの蛍光を検出する工程、
を含む、すい臓のがん細胞またはがん組織の検出方法。 - すい臓がんを検知する方法であって、(a)請求項1~4のいずれか1項に記載の蛍光プローブをすい臓がんの臨床検体に適用する工程、及び(b)前記蛍光プローブを適用したすい臓がんの臨床検体の蛍光像を測定することを含む、前記方法。
- 蛍光イメージング手段を用いて前記蛍光像を可視化することを更なる特徴とする、請求項11に記載の方法。
- (a)被検体のすい臓から外科的に切除された標本に、以下の一般式(I)で表される化合物又はその塩を含む蛍光プローブを適用する工程、及び(b)前記蛍光プローブを適用した切除標本の蛍光像を測定することを含む、被検体におけるすい臓がん細胞の存在を決定する、及び/又は、すい臓がん組織の範囲を同定する、方法。
(式中、
R1は水素原子又はベンゼン環に結合する1~4個の同一又は異なる置換基を表し;
R2、R3、R4、R5、R6、及びR7は、それぞれ独立して、水素原子、ヒドロキシル基、アルキル基、又はハロゲン原子を表し;
R8、R9及びR10は、それぞれ独立して、水素原子又はアルキル基を示し;
XはC1-C3アルキレン基を表し;
Aは、プロリン残基であり;
Bは、グリシン残基、グルタミン酸残基、ロイシン残基、プロリン残基、チロシン残基又はNα-アセチル-リシン残基から選択されるアミノ酸残基であり;
ここで、Aは隣接する式中のNHとアミド結合を形成して連結し、BはAとアミド結合を形成して連結している。) - Bがグリシン残基である、請求項13に記載の方法。
- R1、R2、R3、R4、R5、R6、R7、R8、R9及びR10が水素原子であり、Xがメチレン基である、請求項13又は14に記載の方法。
- 蛍光イメージング手段を用いて前記蛍光像を可視化することを更なる特徴とする、請求項13~15のいずれか1項に記載の方法。
- 請求項13~16のいずれか1項に記載の方法が、すい臓がんの外科治療中に行われる、被検体におけるすい臓がん細胞の存在を決定する、及び/又は、すい臓がん組織の範囲を同定する、方法。
- 前記すい臓がんの外科治療が、開創手術又は鏡視下手術である、請求項17に記載の方法。
Priority Applications (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| JP2023514649A JPWO2022220232A1 (ja) | 2021-04-12 | 2022-04-12 |
Applications Claiming Priority (4)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| US202163173574P | 2021-04-12 | 2021-04-12 | |
| US63/173,574 | 2021-04-12 | ||
| JP2021-081777 | 2021-05-13 | ||
| JP2021081777 | 2021-05-13 |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| WO2022220232A1 true WO2022220232A1 (ja) | 2022-10-20 |
Family
ID=83640069
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/JP2022/017566 WO2022220232A1 (ja) | 2021-04-12 | 2022-04-12 | すい臓がん検出用蛍光プローブ |
Country Status (2)
| Country | Link |
|---|---|
| JP (1) | JPWO2022220232A1 (ja) |
| WO (1) | WO2022220232A1 (ja) |
Citations (5)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JP2004533449A (ja) * | 2001-05-11 | 2004-11-04 | ボード オブ リージェンツ, ザ ユニバーシティ オブ テキサス システム | Cd26を発現している細胞に関連する疾患の治療としての抗cd26モノクローナル抗体 |
| WO2011087000A1 (ja) * | 2010-01-13 | 2011-07-21 | 国立大学法人 東京大学 | がん診断薬 |
| WO2013180181A1 (ja) * | 2012-05-30 | 2013-12-05 | 国立大学法人 東京大学 | 高感度膵液検出用蛍光プローブ、及び膵液検出方法 |
| WO2016006678A1 (ja) * | 2014-07-11 | 2016-01-14 | 国立大学法人 東京大学 | ジペプチジルペプチダーゼiv検出用蛍光プローブ |
| WO2020235567A1 (ja) * | 2019-05-21 | 2020-11-26 | 国立大学法人 東京大学 | 脳腫瘍の検出用蛍光プローブ |
-
2022
- 2022-04-12 JP JP2023514649A patent/JPWO2022220232A1/ja active Pending
- 2022-04-12 WO PCT/JP2022/017566 patent/WO2022220232A1/ja active Application Filing
Patent Citations (5)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JP2004533449A (ja) * | 2001-05-11 | 2004-11-04 | ボード オブ リージェンツ, ザ ユニバーシティ オブ テキサス システム | Cd26を発現している細胞に関連する疾患の治療としての抗cd26モノクローナル抗体 |
| WO2011087000A1 (ja) * | 2010-01-13 | 2011-07-21 | 国立大学法人 東京大学 | がん診断薬 |
| WO2013180181A1 (ja) * | 2012-05-30 | 2013-12-05 | 国立大学法人 東京大学 | 高感度膵液検出用蛍光プローブ、及び膵液検出方法 |
| WO2016006678A1 (ja) * | 2014-07-11 | 2016-01-14 | 国立大学法人 東京大学 | ジペプチジルペプチダーゼiv検出用蛍光プローブ |
| WO2020235567A1 (ja) * | 2019-05-21 | 2020-11-26 | 国立大学法人 東京大学 | 脳腫瘍の検出用蛍光プローブ |
Also Published As
| Publication number | Publication date |
|---|---|
| JPWO2022220232A1 (ja) | 2022-10-20 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| AU2017203340B2 (en) | Synthesis And Composition Of Amino Acid Linking Groups Conjugated To Compounds Used For The Targeted Imaging Of Tumors | |
| EP2524702B1 (en) | Diagnostic for cancer | |
| Fan et al. | Lighting-up breast cancer cells by a near-infrared fluorescent probe based on KIAA1363 enzyme-targeting | |
| US20170232119A1 (en) | Synthesis and composition of amino acid linking groups conjugated to compounds used for the targeted imaging of tumors | |
| KR20180123216A (ko) | Ca ix-표적 nir 염료 및 그의 용도 | |
| US9486545B2 (en) | Method of screening for colon cancer using biomarkers | |
| JP5523282B2 (ja) | 蛍光コバラミンおよびその使用 | |
| Liu et al. | In vivo molecular imaging of gastric cancer in human-murine xenograft models with confocal laser endomicroscopy using a tumor vascular homing peptide | |
| US20230417754A1 (en) | Near infrared-ii probes as high affinity targeting imaging agents and uses thereof | |
| US20100310459A1 (en) | Targeted Detection of Dysplasia In Barrett's Esophagus With A Novel Fluorescence-Labeled Polypeptide | |
| WO2022220232A1 (ja) | すい臓がん検出用蛍光プローブ | |
| WO2008005942A2 (en) | Activatable probes and methods of use | |
| EP3974438A1 (en) | Fluorescent probe for use in detection of brain tumor | |
| WO2021177060A1 (ja) | Lta1基質となる蛍光プローブ | |
| US20220144879A1 (en) | Fluorescent probe for detecting cancer | |
| JP5700392B2 (ja) | 生体試料の調製法 | |
| US11884638B2 (en) | Compound or salt thereof, composition for cysteine detection, fluorescent probe and composition for diagnosing cancer containing the same, method for detecting cysteine, method for providing information for diagnosing cancer, and method for producing compound | |
| Jiang et al. | Engineering a near-infrared LAP fluorescent probe with high sensitivity and selectivity for surgical resection of liver cancer | |
| CN116655516A (zh) | 用于前列腺癌特异性荧光探针的化合物、制备方法及其应用 | |
| Mathejczyk | Innovative NIR fluorescent probes for an improved tumor detection in vivo | |
| Muguruma et al. | Endoscopic Molecular Imaging in Gastrointestinal Oncology | |
| Paharia | Molecular Tissue Mapping and Real-time Image Guidance for Surgical Resection of Head and Neck Cancers–A Review | |
| Masatomo Beika et al. | Detection of Metastatic Lymph Nodes Using 5-Aminolevulinic Acid in Patients with Gastric Cancer |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application |
Ref document number: 22788157 Country of ref document: EP Kind code of ref document: A1 |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 2023514649 Country of ref document: JP |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 18286291 Country of ref document: US |
|
| NENP | Non-entry into the national phase |
Ref country code: DE |
|
| 122 | Ep: pct application non-entry in european phase |
Ref document number: 22788157 Country of ref document: EP Kind code of ref document: A1 |