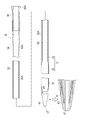
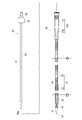
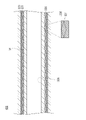
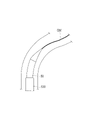
WO2022168719A1 - Cathéter percutané - Google Patents
Cathéter percutané Download PDFInfo
- Publication number
- WO2022168719A1 WO2022168719A1 PCT/JP2022/003023 JP2022003023W WO2022168719A1 WO 2022168719 A1 WO2022168719 A1 WO 2022168719A1 JP 2022003023 W JP2022003023 W JP 2022003023W WO 2022168719 A1 WO2022168719 A1 WO 2022168719A1
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- catheter
- blood
- tube
- stylet
- reinforcing body
- Prior art date
Links
- 229920005989 resin Polymers 0.000 claims abstract description 25
- 239000011347 resin Substances 0.000 claims abstract description 25
- 230000003014 reinforcing effect Effects 0.000 claims description 79
- 239000010410 layer Substances 0.000 claims description 25
- 239000012790 adhesive layer Substances 0.000 claims description 12
- 239000003351 stiffener Substances 0.000 claims 1
- 210000004204 blood vessel Anatomy 0.000 abstract description 29
- 230000002787 reinforcement Effects 0.000 abstract description 7
- 238000005253 cladding Methods 0.000 abstract 1
- 239000008280 blood Substances 0.000 description 81
- 210000004369 blood Anatomy 0.000 description 81
- 230000004087 circulation Effects 0.000 description 30
- 230000004048 modification Effects 0.000 description 27
- 238000012986 modification Methods 0.000 description 27
- 238000000034 method Methods 0.000 description 17
- 239000000463 material Substances 0.000 description 16
- 238000009941 weaving Methods 0.000 description 15
- PPBRXRYQALVLMV-UHFFFAOYSA-N Styrene Chemical compound C=CC1=CC=CC=C1 PPBRXRYQALVLMV-UHFFFAOYSA-N 0.000 description 14
- 230000036770 blood supply Effects 0.000 description 13
- 239000000853 adhesive Substances 0.000 description 12
- 230000001070 adhesive effect Effects 0.000 description 12
- 210000001631 vena cava inferior Anatomy 0.000 description 12
- 238000010586 diagram Methods 0.000 description 8
- 230000006870 function Effects 0.000 description 8
- 230000017531 blood circulation Effects 0.000 description 7
- -1 for example Substances 0.000 description 7
- 210000005245 right atrium Anatomy 0.000 description 7
- 229920001577 copolymer Polymers 0.000 description 6
- 238000001514 detection method Methods 0.000 description 6
- 210000003462 vein Anatomy 0.000 description 6
- JOYRKODLDBILNP-UHFFFAOYSA-N Ethyl urethane Chemical compound CCOC(N)=O JOYRKODLDBILNP-UHFFFAOYSA-N 0.000 description 5
- 238000003618 dip coating Methods 0.000 description 5
- 238000007654 immersion Methods 0.000 description 5
- 238000000465 moulding Methods 0.000 description 5
- 239000004698 Polyethylene Substances 0.000 description 4
- XUIMIQQOPSSXEZ-UHFFFAOYSA-N Silicon Chemical compound [Si] XUIMIQQOPSSXEZ-UHFFFAOYSA-N 0.000 description 4
- 230000008901 benefit Effects 0.000 description 4
- 238000009954 braiding Methods 0.000 description 4
- 230000002612 cardiopulmonary effect Effects 0.000 description 4
- 230000008602 contraction Effects 0.000 description 4
- 210000003191 femoral vein Anatomy 0.000 description 4
- 239000007789 gas Substances 0.000 description 4
- 210000004072 lung Anatomy 0.000 description 4
- 229920000573 polyethylene Polymers 0.000 description 4
- 229920002635 polyurethane Polymers 0.000 description 4
- 239000004814 polyurethane Substances 0.000 description 4
- 239000010703 silicon Substances 0.000 description 4
- 229910052710 silicon Inorganic materials 0.000 description 4
- BZHJMEDXRYGGRV-UHFFFAOYSA-N Vinyl chloride Chemical compound ClC=C BZHJMEDXRYGGRV-UHFFFAOYSA-N 0.000 description 3
- 229920001971 elastomer Polymers 0.000 description 3
- 238000003780 insertion Methods 0.000 description 3
- 230000037431 insertion Effects 0.000 description 3
- 239000012528 membrane Substances 0.000 description 3
- 230000002093 peripheral effect Effects 0.000 description 3
- 239000005060 rubber Substances 0.000 description 3
- 239000012781 shape memory material Substances 0.000 description 3
- 210000002620 vena cava superior Anatomy 0.000 description 3
- CURLTUGMZLYLDI-UHFFFAOYSA-N Carbon dioxide Chemical compound O=C=O CURLTUGMZLYLDI-UHFFFAOYSA-N 0.000 description 2
- MYMOFIZGZYHOMD-UHFFFAOYSA-N Dioxygen Chemical compound O=O MYMOFIZGZYHOMD-UHFFFAOYSA-N 0.000 description 2
- RRHGJUQNOFWUDK-UHFFFAOYSA-N Isoprene Chemical compound CC(=C)C=C RRHGJUQNOFWUDK-UHFFFAOYSA-N 0.000 description 2
- 239000004677 Nylon Substances 0.000 description 2
- 230000004323 axial length Effects 0.000 description 2
- 230000000740 bleeding effect Effects 0.000 description 2
- MTAZNLWOLGHBHU-UHFFFAOYSA-N butadiene-styrene rubber Chemical compound C=CC=C.C=CC1=CC=CC=C1 MTAZNLWOLGHBHU-UHFFFAOYSA-N 0.000 description 2
- 239000011248 coating agent Substances 0.000 description 2
- 238000000576 coating method Methods 0.000 description 2
- 230000007423 decrease Effects 0.000 description 2
- 229910001882 dioxygen Inorganic materials 0.000 description 2
- 239000005038 ethylene vinyl acetate Substances 0.000 description 2
- 229920006244 ethylene-ethyl acrylate Polymers 0.000 description 2
- 210000004731 jugular vein Anatomy 0.000 description 2
- 238000009940 knitting Methods 0.000 description 2
- 239000007788 liquid Substances 0.000 description 2
- 229910052751 metal Inorganic materials 0.000 description 2
- 239000002184 metal Substances 0.000 description 2
- 239000000203 mixture Substances 0.000 description 2
- 229920001778 nylon Polymers 0.000 description 2
- 229920001200 poly(ethylene-vinyl acetate) Polymers 0.000 description 2
- 229920000346 polystyrene-polyisoprene block-polystyrene Polymers 0.000 description 2
- 230000001105 regulatory effect Effects 0.000 description 2
- 229920003048 styrene butadiene rubber Polymers 0.000 description 2
- 229920000468 styrene butadiene styrene block copolymer Polymers 0.000 description 2
- 229920002725 thermoplastic elastomer Polymers 0.000 description 2
- 229920002803 thermoplastic polyurethane Polymers 0.000 description 2
- 239000010936 titanium Substances 0.000 description 2
- 229910052719 titanium Inorganic materials 0.000 description 2
- 229920000178 Acrylic resin Polymers 0.000 description 1
- 239000004925 Acrylic resin Substances 0.000 description 1
- RYGMFSIKBFXOCR-UHFFFAOYSA-N Copper Chemical compound [Cu] RYGMFSIKBFXOCR-UHFFFAOYSA-N 0.000 description 1
- 244000043261 Hevea brasiliensis Species 0.000 description 1
- 229920002614 Polyether block amide Polymers 0.000 description 1
- 239000004743 Polypropylene Substances 0.000 description 1
- 208000007536 Thrombosis Diseases 0.000 description 1
- RTAQQCXQSZGOHL-UHFFFAOYSA-N Titanium Chemical compound [Ti] RTAQQCXQSZGOHL-UHFFFAOYSA-N 0.000 description 1
- 229920000800 acrylic rubber Polymers 0.000 description 1
- 150000001336 alkenes Chemical class 0.000 description 1
- 239000000956 alloy Substances 0.000 description 1
- 229910045601 alloy Inorganic materials 0.000 description 1
- 210000000709 aorta Anatomy 0.000 description 1
- 210000001367 artery Anatomy 0.000 description 1
- 230000005540 biological transmission Effects 0.000 description 1
- 230000015572 biosynthetic process Effects 0.000 description 1
- 229920005549 butyl rubber Polymers 0.000 description 1
- 229910002092 carbon dioxide Inorganic materials 0.000 description 1
- 239000001569 carbon dioxide Substances 0.000 description 1
- 238000002680 cardiopulmonary resuscitation Methods 0.000 description 1
- 230000000052 comparative effect Effects 0.000 description 1
- 239000002131 composite material Substances 0.000 description 1
- 229910052802 copper Inorganic materials 0.000 description 1
- 239000010949 copper Substances 0.000 description 1
- 230000000916 dilatatory effect Effects 0.000 description 1
- 230000010339 dilation Effects 0.000 description 1
- 239000013013 elastic material Substances 0.000 description 1
- 239000005042 ethylene-ethyl acrylate Substances 0.000 description 1
- 210000001105 femoral artery Anatomy 0.000 description 1
- 230000004217 heart function Effects 0.000 description 1
- 239000012510 hollow fiber Substances 0.000 description 1
- 238000001727 in vivo Methods 0.000 description 1
- 229920003049 isoprene rubber Polymers 0.000 description 1
- 238000013008 moisture curing Methods 0.000 description 1
- 229920003052 natural elastomer Polymers 0.000 description 1
- 229920001194 natural rubber Polymers 0.000 description 1
- JRZJOMJEPLMPRA-UHFFFAOYSA-N olefin Natural products CCCCCCCC=C JRZJOMJEPLMPRA-UHFFFAOYSA-N 0.000 description 1
- 238000006213 oxygenation reaction Methods 0.000 description 1
- 229910052763 palladium Inorganic materials 0.000 description 1
- 230000000149 penetrating effect Effects 0.000 description 1
- 229920000636 poly(norbornene) polymer Polymers 0.000 description 1
- 229920000058 polyacrylate Polymers 0.000 description 1
- 229920001083 polybutene Polymers 0.000 description 1
- 229920000642 polymer Polymers 0.000 description 1
- 229920001155 polypropylene Polymers 0.000 description 1
- 229920001296 polysiloxane Polymers 0.000 description 1
- 102000004169 proteins and genes Human genes 0.000 description 1
- 108090000623 proteins and genes Proteins 0.000 description 1
- 230000009467 reduction Effects 0.000 description 1
- 230000000241 respiratory effect Effects 0.000 description 1
- 239000002210 silicon-based material Substances 0.000 description 1
- 229920002379 silicone rubber Polymers 0.000 description 1
- 239000004945 silicone rubber Substances 0.000 description 1
- 230000000087 stabilizing effect Effects 0.000 description 1
- 229920003051 synthetic elastomer Polymers 0.000 description 1
- 229920003002 synthetic resin Polymers 0.000 description 1
- 239000000057 synthetic resin Substances 0.000 description 1
- 239000005061 synthetic rubber Substances 0.000 description 1
- 230000007704 transition Effects 0.000 description 1
Images
Classifications
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61M—DEVICES FOR INTRODUCING MEDIA INTO, OR ONTO, THE BODY; DEVICES FOR TRANSDUCING BODY MEDIA OR FOR TAKING MEDIA FROM THE BODY; DEVICES FOR PRODUCING OR ENDING SLEEP OR STUPOR
- A61M1/00—Suction or pumping devices for medical purposes; Devices for carrying-off, for treatment of, or for carrying-over, body-liquids; Drainage systems
- A61M1/36—Other treatment of blood in a by-pass of the natural circulatory system, e.g. temperature adaptation, irradiation ; Extra-corporeal blood circuits
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61M—DEVICES FOR INTRODUCING MEDIA INTO, OR ONTO, THE BODY; DEVICES FOR TRANSDUCING BODY MEDIA OR FOR TAKING MEDIA FROM THE BODY; DEVICES FOR PRODUCING OR ENDING SLEEP OR STUPOR
- A61M25/00—Catheters; Hollow probes
Abstract
L'invention fournit un cathéter percutané dont l'étirement peut être inhibé, y compris lorsque le cathéter percutané se prend involontairement dans un vaisseau sanguin. Selon l'invention, un cathéter (30) possède un second corps de renforcement (331) tubulaire, une partie revêtement (332) revêtant au moins une partie du second corps de renforcement, et une seconde couche de résine (333) agencée de manière à recouvrir le second corps de renforcement et la partie revêtement.
Priority Applications (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| JP2022579488A JPWO2022168719A1 (fr) | 2021-02-03 | 2022-01-27 |
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| JP2021015855 | 2021-02-03 | ||
| JP2021-015855 | 2021-02-03 |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| WO2022168719A1 true WO2022168719A1 (fr) | 2022-08-11 |
Family
ID=82741517
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/JP2022/003023 WO2022168719A1 (fr) | 2021-02-03 | 2022-01-27 | Cathéter percutané |
Country Status (2)
| Country | Link |
|---|---|
| JP (1) | JPWO2022168719A1 (fr) |
| WO (1) | WO2022168719A1 (fr) |
Citations (2)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JPH02102665A (ja) * | 1988-10-11 | 1990-04-16 | Terumo Corp | カテーテル |
| WO2018193601A1 (fr) * | 2017-04-20 | 2018-10-25 | 朝日インテック株式会社 | Cathéter |
-
2022
- 2022-01-27 WO PCT/JP2022/003023 patent/WO2022168719A1/fr active Application Filing
- 2022-01-27 JP JP2022579488A patent/JPWO2022168719A1/ja active Pending
Patent Citations (2)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JPH02102665A (ja) * | 1988-10-11 | 1990-04-16 | Terumo Corp | カテーテル |
| WO2018193601A1 (fr) * | 2017-04-20 | 2018-10-25 | 朝日インテック株式会社 | Cathéter |
Also Published As
| Publication number | Publication date |
|---|---|
| JPWO2022168719A1 (fr) | 2022-08-11 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| US9022916B2 (en) | Transseptal cannula, tip, delivery system, and method | |
| JP6813571B2 (ja) | 経皮カテーテルおよび経皮カテーテル用チューブの製造方法 | |
| WO2014138146A2 (fr) | Canule trans-septale, pointe, système de pose et procédé | |
| US11260159B2 (en) | Percutaneous catheter and percutaneous catheter assembly | |
| JP6876710B2 (ja) | カテーテル | |
| WO2022168719A1 (fr) | Cathéter percutané | |
| JP6813570B2 (ja) | 経皮カテーテル | |
| WO2022168720A1 (fr) | Cathéter percutané | |
| JP7157283B1 (ja) | スタイレットおよびカテーテル組立体 | |
| WO2006118103A1 (fr) | Ensemble de pompage a ballonnet intra-aortique | |
| JP6730430B2 (ja) | 経皮カテーテル組立体、経皮カテーテル、およびダイレーター | |
| WO2021172155A1 (fr) | Cathéter percutané | |
| WO2021182070A1 (fr) | Stylet | |
| WO2021177053A1 (fr) | Cathéter percutané | |
| WO2021177165A1 (fr) | Cathéter percutané et procédé d'utilisation de cathéter percutané | |
| JP2023032536A (ja) | シースおよび医療用組立体 | |
| US20210290904A1 (en) | Catheter assembly | |
| JP2023000024A (ja) | 経皮カテーテル | |
| WO2020137708A1 (fr) | Ensemble cathéter |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application |
Ref document number: 22749593 Country of ref document: EP Kind code of ref document: A1 |
|
| ENP | Entry into the national phase |
Ref document number: 2022579488 Country of ref document: JP Kind code of ref document: A |
|
| NENP | Non-entry into the national phase |
Ref country code: DE |
|
| 122 | Ep: pct application non-entry in european phase |
Ref document number: 22749593 Country of ref document: EP Kind code of ref document: A1 |