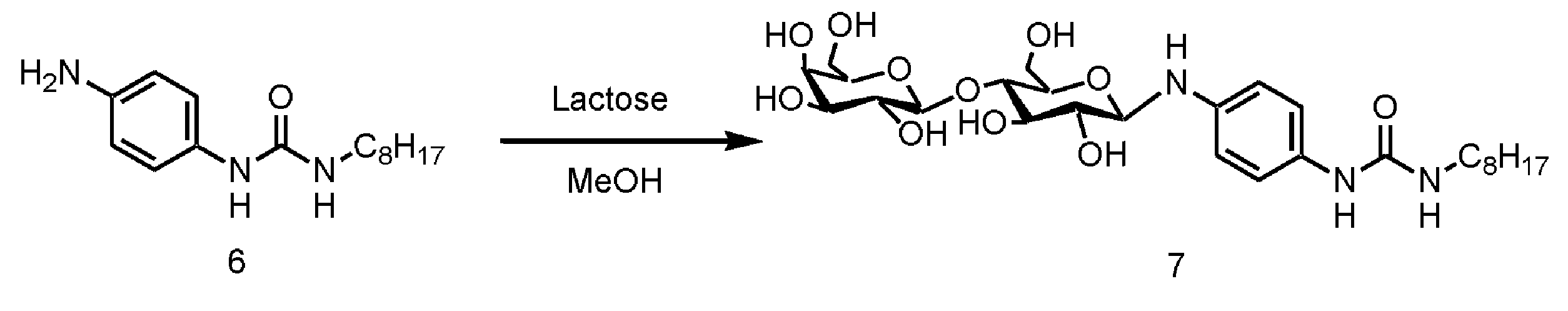
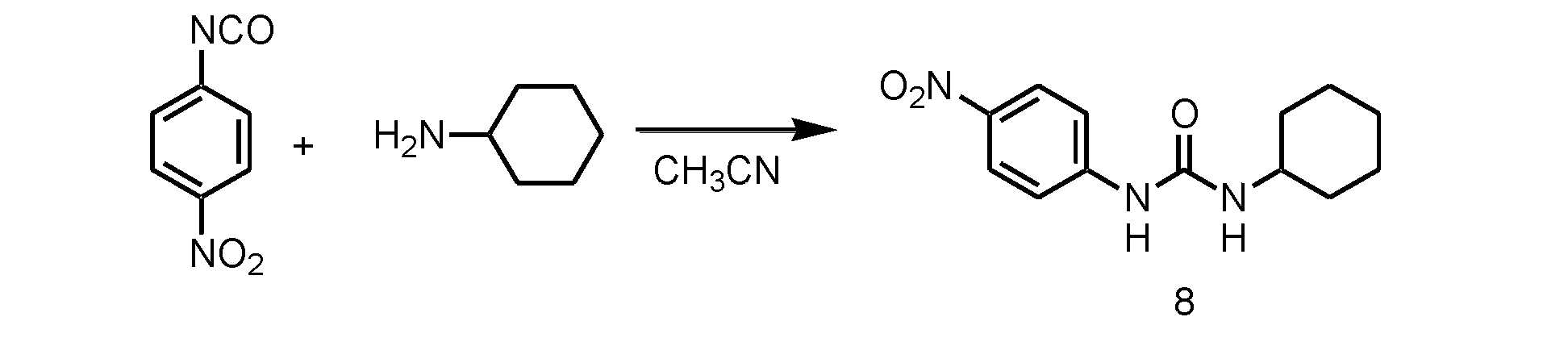
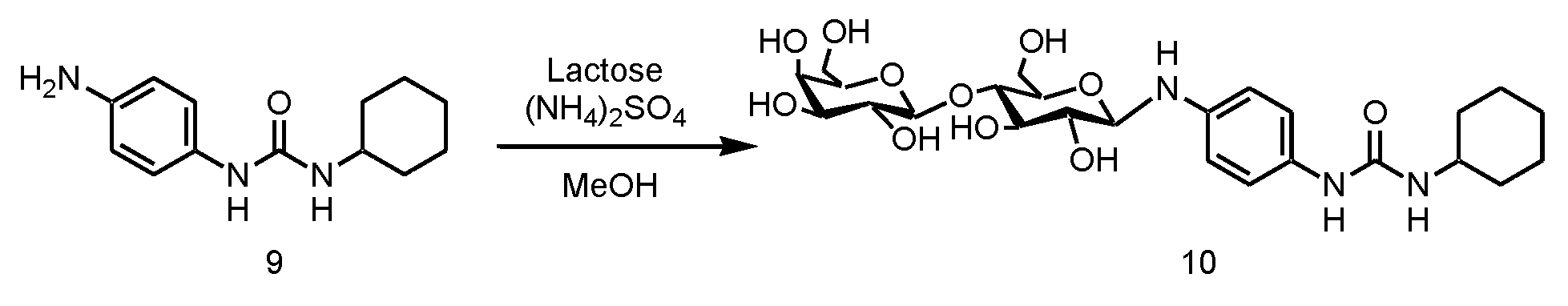
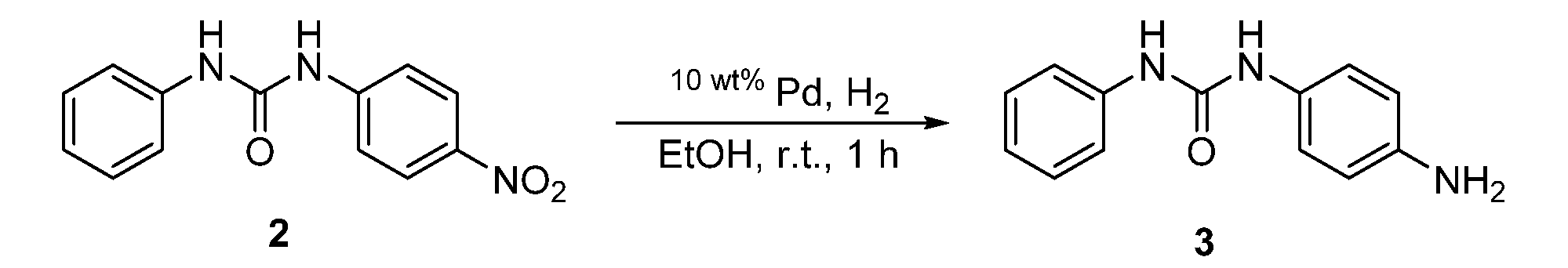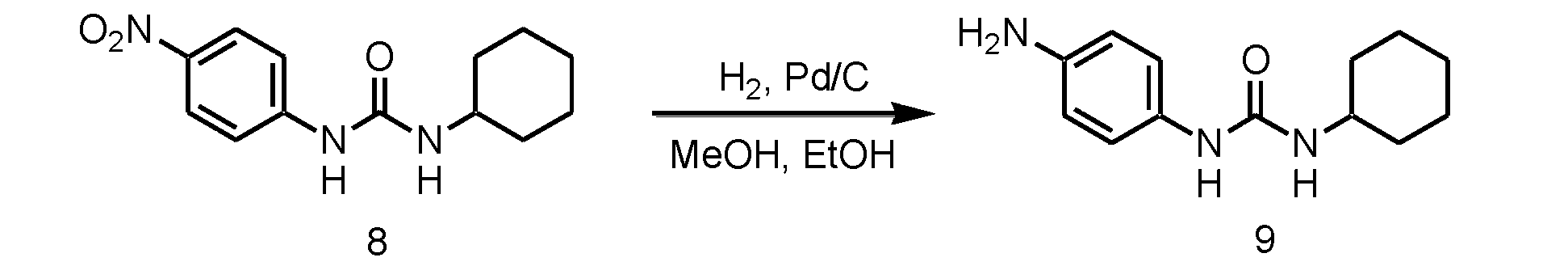WO2018070546A1 - 新規ヒドロゲル化剤 - Google Patents
新規ヒドロゲル化剤 Download PDFInfo
- Publication number
- WO2018070546A1 WO2018070546A1 PCT/JP2017/037423 JP2017037423W WO2018070546A1 WO 2018070546 A1 WO2018070546 A1 WO 2018070546A1 JP 2017037423 W JP2017037423 W JP 2017037423W WO 2018070546 A1 WO2018070546 A1 WO 2018070546A1
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- group
- carbon atoms
- hydrogelator
- compound
- formula
- Prior art date
Links
- 0 *NC(Nc(cc1)ccc1N)=O Chemical compound *NC(Nc(cc1)ccc1N)=O 0.000 description 3
- OISZIRHDSSOUKW-UHFFFAOYSA-N Nc(cc1)ccc1NC(Nc1ccccc1)=O Chemical compound Nc(cc1)ccc1NC(Nc1ccccc1)=O OISZIRHDSSOUKW-UHFFFAOYSA-N 0.000 description 2
- XVRHKKFLRSZHFG-MOIXXUDNSA-N CC(C1)[C@H]1OC([C@H](C(CO)OC1O[C@H](C(CO)O[C@H](C2O)Nc(cc3)ccc3NC(NC)=O)[C@@H]2O)O)[C@@H]1O Chemical compound CC(C1)[C@H]1OC([C@H](C(CO)OC1O[C@H](C(CO)O[C@H](C2O)Nc(cc3)ccc3NC(NC)=O)[C@@H]2O)O)[C@@H]1O XVRHKKFLRSZHFG-MOIXXUDNSA-N 0.000 description 1
- QHHPMKKJBURYIQ-UHFFFAOYSA-N [O-][N+](c(cc1)ccc1NC(Nc1ccccc1)=O)=O Chemical compound [O-][N+](c(cc1)ccc1NC(Nc1ccccc1)=O)=O QHHPMKKJBURYIQ-UHFFFAOYSA-N 0.000 description 1
Images
Classifications
-
- C—CHEMISTRY; METALLURGY
- C09—DYES; PAINTS; POLISHES; NATURAL RESINS; ADHESIVES; COMPOSITIONS NOT OTHERWISE PROVIDED FOR; APPLICATIONS OF MATERIALS NOT OTHERWISE PROVIDED FOR
- C09K—MATERIALS FOR MISCELLANEOUS APPLICATIONS, NOT PROVIDED FOR ELSEWHERE
- C09K3/00—Materials not provided for elsewhere
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07H—SUGARS; DERIVATIVES THEREOF; NUCLEOSIDES; NUCLEOTIDES; NUCLEIC ACIDS
- C07H15/00—Compounds containing hydrocarbon or substituted hydrocarbon radicals directly attached to hetero atoms of saccharide radicals
- C07H15/20—Carbocyclic rings
- C07H15/203—Monocyclic carbocyclic rings other than cyclohexane rings; Bicyclic carbocyclic ring systems
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K47/00—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient
- A61K47/06—Organic compounds, e.g. natural or synthetic hydrocarbons, polyolefins, mineral oil, petrolatum or ozokerite
- A61K47/26—Carbohydrates, e.g. sugar alcohols, amino sugars, nucleic acids, mono-, di- or oligo-saccharides; Derivatives thereof, e.g. polysorbates, sorbitan fatty acid esters or glycyrrhizin
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K8/00—Cosmetics or similar toiletry preparations
- A61K8/18—Cosmetics or similar toiletry preparations characterised by the composition
- A61K8/30—Cosmetics or similar toiletry preparations characterised by the composition containing organic compounds
- A61K8/60—Sugars; Derivatives thereof
Definitions
- the present invention relates to a novel gelling agent, and in particular, to a novel hydrogelling agent containing a monourea compound having a sugar structure that can form a gel with respect to an aqueous solvent.
- gels are added to paints and resins in the field of paints and resins to adjust fluidity, or gelate waste oil, waste liquid, wastewater, etc. to form solids, prevent water pollution, etc. It is widely used in various fields such as industrial field, daily necessities field, environmental field, medical field, cosmetics field, food field, agricultural field, living body related field, and analytical field.
- a gel is a structure in which a fluid such as water or an organic solvent is contained in a three-dimensional network structure formed of a chemical substance. An organogel is used when the fluid is an organic solvent, and a hydrogel when the fluid is water. That's it.
- An object of the present invention is to provide a novel gelling agent that can be prepared by a simple technique and has a structure that has not been proposed so far, particularly a novel gelling agent that can form a hydrogel.
- the present inventors have applied a monourea compound having a sugar structure as a gelling agent. Surprisingly, gels can be formed in various aqueous solvents. As a result, the present invention was completed.
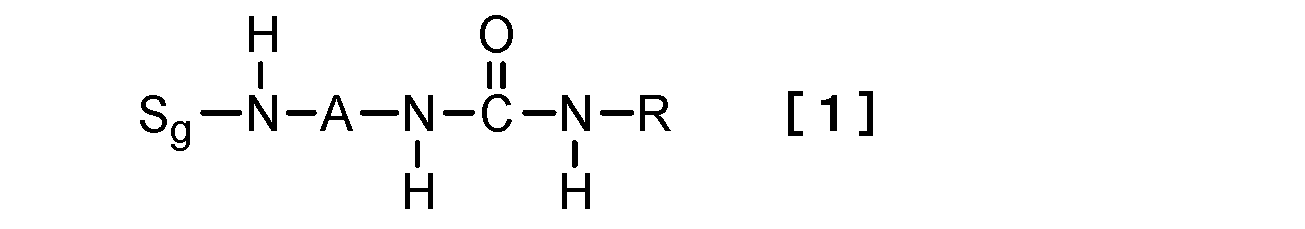
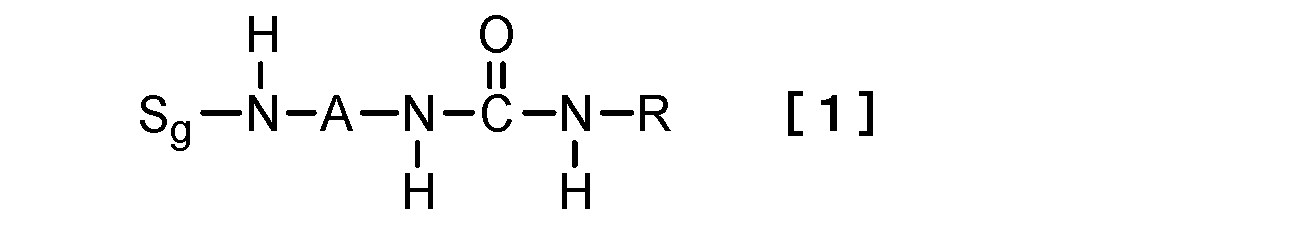
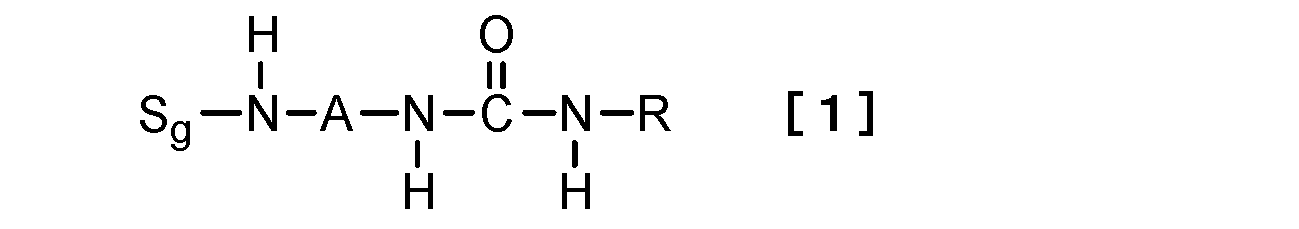
- this invention relates to the hydrogelator which consists of a compound represented by following formula [1] as a 1st viewpoint.
- S g represents a sugar group
- A represents a divalent linking group
- R is a linear or branched alkyl group having 1 to 15 carbon atoms, a cyclic alkyl group having 3 to 15 carbon atoms, or a linear or branched alkenyl group having 2 to 15 carbon atoms.
- the present invention relates to the hydrogelator according to the first aspect, in which the sugar group Sg is a monovalent group having a monosaccharide or disaccharide-derived structure.
- the linking group A is a linear alkylene group having 1 to 15 carbon atoms or an arylene group having 6 to 15 carbon atoms
- R is a linear alkyl group having 1 to 15 carbon atoms, a cyclic alkyl group having 3 to 10 carbon atoms, an unsubstituted or alkyl group having 1 to 10 carbon atoms, a halogen atom, and the number of carbon atoms
- the present invention relates to a hydrogel comprising the hydrogelator according to any one of the first aspect to the third aspect and an aqueous solvent.
- the present invention relates to a compound represented by the following formula [1]. (Where S g represents a sugar group, A represents a divalent linking group, R is a linear or branched alkyl group having 1 to 15 carbon atoms, a cyclic alkyl group having 3 to 15 carbon atoms, or a linear or branched alkenyl group having 2 to 15 carbon atoms.
- the hydrogelator of the present invention has an effect that an aqueous solvent can be gelled to form a gel. Moreover, the hydrogelator of this invention has the effect that it has high gelatinization ability. Furthermore, the hydrogelator of the present invention has an effect that it can be produced by a simple technique.
- FIG. 1 is a photograph of each sample tube after cooling in a gelation test of a hydrogelator comprising a compound of formula 4 with respect to various aqueous solvents in Example 2 (Aqueous solvent used: pure water, phosphate buffer) , Physiological saline, 30% 1,3-propanediol, 70% 1,3-propanediol, 30% ethanol, 70% ethanol) (Sus: suspended state, PG: partially gelled).
- Aqueous solvent used pure water, phosphate buffer
- Physiological saline 30% 1,3-propanediol
- 70% 1,3-propanediol 30% ethanol
- 70% ethanol 70% ethanol
- Example 2 is a photograph of each sample tube after cooling in a gelation test of a hydrogelator comprising a compound of formula 7 with respect to various aqueous solvents in Example 2 (aqueous solvent: pure water, phosphate buffer) Saline, 30% 1,3-propanediol, 70% 1,3-propanediol, 30% ethanol, 70% ethanol) (Sus: suspended state).
- aqueous solvent pure water, phosphate buffer
- FIG. 3 is a photograph of each sample tube after cooling in a gelation test of a hydrogelator comprising a compound of formula 10 with respect to various aqueous solvents in Example 2 (Aqueous solvent used: pure water, phosphate buffer) , Physiological saline, 30% 1,3-propanediol, 70% 1,3-propanediol, 30% ethanol, 70% ethanol) (Sol: solution is not viscous).
- FIG. 4 is a photograph of each sample tube after cooling in a gelation test of a hydrogelator comprising a compound of Formula 4 with respect to aqueous solvents having various pH values in Example 3 (aqueous solvent used; pH 1 to 5: Hydrochloric acid aqueous solution, pH 7: pure water, pH 8 to 10: sodium hydroxide aqueous solution) (Sus: suspended state, PG: partially gelled).
- aqueous solvent used pH 1 to 5: Hydrochloric acid aqueous solution, pH 7: pure water, pH 8 to 10: sodium hydroxide aqueous solution
- Sus suspended state
- PG partially gelled
- Example 5 is a photograph of each sample tube after cooling in a gelation test of a hydrogelator comprising a compound of formula 7 with respect to aqueous solvents of various pHs in Example 3 (aqueous solvent used; pH 1 to 5: hydrochloric acid) Aqueous solution, pH 7: pure water, pH 8-10: aqueous sodium hydroxide solution (Sus: suspended state).
- FIG. 6 is a photograph of each sample tube after cooling in a gelation test of a hydrogelator comprising a compound of formula 10 with respect to aqueous solvents having various pH values in Example 3 (aqueous solvent used; pH 1 to 5: hydrochloric acid) Aqueous solution, pH 7: pure water, pH 8-10: sodium hydroxide aqueous solution) (Sol: solution is not viscous).
- aqueous solvent used pH 1 to 5: hydrochloric acid
- pH 8-10 sodium hydroxide aqueous solution
- Sol solution is not viscous.
- FIG. 7 is a photograph of each sample tube after cooling in a gelation test of a hydrogelator comprising a compound of Formula 4 with respect to various aqueous solvents in Example 4
- Aqueous solvent used pure water (minimum gelation concentration) : 1.0 wt%), hydrochloric acid aqueous solution (pH 3) (minimum gelation concentration: 0.5 wt%), sodium hydroxide aqueous solution (pH 10) (minimum gelation concentration: 1.0 wt%), 30% ethanol (minimum gelation) Concentration: 1.0 wt%)].
- FIG. 1 Aqueous solvent used: pure water (minimum gelation concentration) : 1.0 wt%), hydrochloric acid aqueous solution (pH 3) (minimum gelation concentration: 0.5 wt%), sodium hydroxide aqueous solution (pH 10) (minimum gelation concentration: 1.0 wt%), 30% ethanol (minimum gelation) Concentration:
- FIG. 8 is a photograph of each sample tube after cooling in a gelation test of a hydrogelator comprising a compound of Formula 7 with respect to various aqueous solvents in Example 4
- Aqueous solvent used pure water (minimum gelation concentration) : 0.1 wt%), sodium hydroxide aqueous solution (pH 10) (minimum gelation concentration: 0.1 wt%), 30% ethanol (minimum gelation concentration: 0.1 wt%), 30% 1,3-propanediol ( Minimum gelation concentration: 0.1 wt%), 70% 1,3-propanediol (minimum gelation concentration: 0.5 wt%)].
- FIG. 1 Aqueous solvent used: pure water (minimum gelation concentration) : 0.1 wt%), sodium hydroxide aqueous solution (pH 10) (minimum gelation concentration: 0.1 wt%), 30% ethanol (minimum gelation concentration: 0.1 wt%), 30% 1,3-propaned
- FIG. 9 is a photograph of each sample tube after cooling in a gelation test of a hydrogelator comprising a compound of formula 10 with respect to various aqueous solvents in Example 4
- Aqueous solvent used pure water (minimum gelation concentration) : 0.5 wt%), hydrochloric acid aqueous solution (pH 3) (minimum gelation concentration: 1.0 wt%), sodium hydroxide aqueous solution (pH 10) (minimum gelation concentration: 0.5 wt%), physiological saline (minimum gelation) Concentration: 0.5 wt%), 30% 1,3-propanediol (minimum gelation concentration: 1.0 wt%)].
- FIG. 1 Aqueous solvent used: pure water (minimum gelation concentration) : 0.5 wt%), hydrochloric acid aqueous solution (pH 3) (minimum gelation concentration: 1.0 wt%), sodium hydroxide aqueous solution (pH 10) (
- FIG. 10 is a photograph of each sample tube after cooling in a gelation test of a hydrogelator composed of the compound of formula 10 with respect to heat treatment (40 ° C.) in Example 5 (gelation concentration: 0.5 wt%). 1.0 wt%).
- FIG. 11 is a photograph of each sample tube after cooling in a gelation test of a hydrogelator composed of the compound of formula 10 with respect to heat treatment (60 ° C.) in Example 5 (gelation concentration: 0.5 wt%). 1.0 wt%).
- FIG. 12 is a photograph of each sample tube after standing in a gelation test of a hydrogelator composed of the compound of Formula 10 for ultrasonic treatment in Example 5 (gelation concentration: 1.0 wt%).
- FIG. 13 is a photograph of each sample tube after standing in the gelation test of the hydrogelator composed of the compound of Formula 10 for vibration treatment in Example 5 (gelation concentration: 1.0 wt%).
- FIG. 14 is a photograph of a sample tube after cooling in a gelation test of a hydrogelator composed of the compound of Formula 4 in Example 7 (gelation concentration: 1.0 wt%).
- FIG. 15 is a photograph of a sample tube after cooling in a gelation test of a hydrogelator comprising a compound of formula 7 in Example 8 [(left) gelation concentration: 0.1 wt%, (right) gel Concentration: 0.5 wt%] (G: gelation).
- FIG. 16 is a photograph of a sample tube after cooling in a gelation test of a hydrogelator composed of the compound of formula 10 in Example 9.
- FIG. 17 is a graph showing the results of hydrogel transparency evaluation in Example 10.
- FIG. 18 is a photograph of a sample tube after standing in a gelation test of a hydrogelator comprising a compound of Formula 10 for vibration treatment in Example 11 [Left: Photograph of a sample tube before vibration treatment, middle: Photograph of vibration treatment, right: Photograph of sample tube after standing].
- FIG. 19 is a photograph of a scanning electron microscope (SEM) image in Example 12.
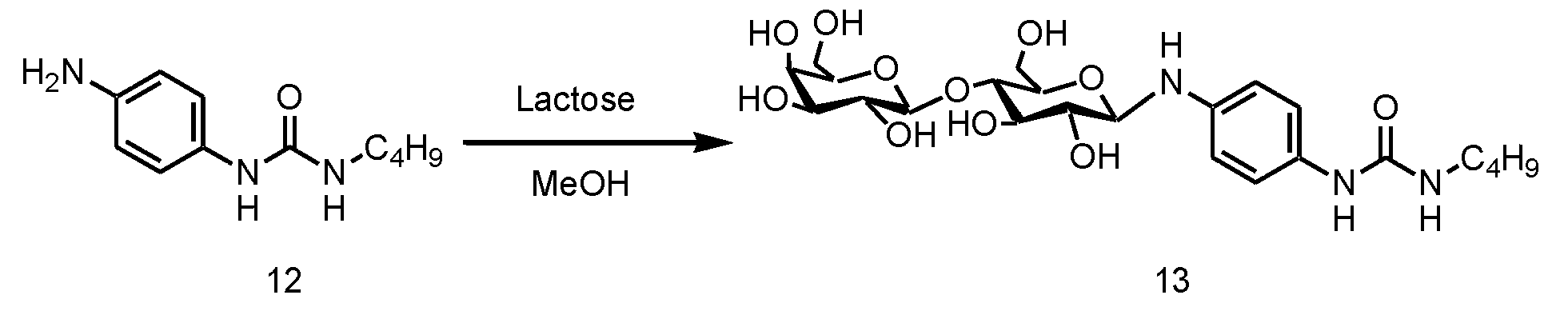
- FIG. 20 is a photograph of a sample tube after cooling in a gelation test of a hydrogelator composed of the compound of Formula 13 in Example 14.
- the hydrogelator of the present invention comprises a monourea compound having a sugar structure represented by the following formula [1].
- S g represents a sugar group
- A represents a divalent linking group
- R is a linear or branched alkyl group having 1 to 15 carbon atoms, a cyclic alkyl group having 3 to 15 carbon atoms, or a linear or branched alkenyl group having 2 to 15 carbon atoms.
- the sugar group represented by S g a monovalent group comprising a structure derived from sugars, specifically, a removing one residue of the hydroxy group from a sugar.
- the sugar group may be substituted with a substituent such as an alkyl group (such as a methyl group or an ethyl group) and a sulfo group.
- a substituent such as an alkyl group (such as a methyl group or an ethyl group) and a sulfo group.
- the kind of the sugar is not particularly limited, it is preferably a monosaccharide or a disaccharide, and more preferably a disaccharide, from the viewpoint of gelation activity.
- pentose or hexose is preferable.
- pentose include ribose, deoxyribose, and fructose.
- hexose include glucose, mannose, galactose, methyl- ⁇ -glucose, and methyl- ⁇ -mannose.
- hexose is preferable, and glucose, mannose, galactose, methyl- ⁇ -glucose, and methyl- ⁇ -mannose are particularly preferable.
- disaccharide examples include lactose, maltose, isomaltose, trehalose, sucrose, and cellobiose, among which lactose is particularly preferable.
- the linking group represented by A is not particularly limited, and for example, a linear or branched alkylene group having 1 to 15 carbon atoms, a cyclic alkylene group having 3 to 15 carbon atoms, and 6 carbon atoms. Thru
- or 15 arylene groups etc. are mentioned.
- Examples of the linear or branched alkylene group having 1 to 15 carbon atoms include a methylene group, an ethylene group, a propylene group, a butylene group, and an octylene group, and a group obtained by branching these groups. Is mentioned.
- the cyclic alkylene group having 3 to 15 carbon atoms includes not only a group consisting of only a cyclic alkylene group (for example, a cyclopropylene group, a cyclobutylene group, a cyclohexene group, etc.), but also a ring such as a cyclopentyl ring or a cyclohexyl ring.
- Examples thereof include linear and / or branched alkylene groups having a structure and having 3 to 15 carbon atoms.
- Examples of the arylene group having 6 to 15 carbon atoms include a phenylene group, a naphthylene group, and an anthrylene group.
- Examples of the linear or branched alkyl group having 1 to 15 carbon atoms represented by R include methyl group, ethyl group, propyl group, butyl group, pentyl group, hexyl group, heptyl group, octyl group, nonyl group, decyl group. Groups, undecyl groups, dodecyl groups, tridecyl groups, tetradecyl groups and pentadecyl groups, and groups formed by branching these groups.
- Examples of the cyclic alkyl group having 3 to 15 carbon atoms represented by R include not only a cyclic alkyl group (for example, a cyclopropyl group, a cyclobutyl group, a cyclopentyl group, a cyclohexyl group, a cycloheptyl group, etc.) Examples thereof include linear and / or branched alkyl groups having a ring structure such as a cyclopentyl ring and a cyclohexyl ring and having 3 to 15 carbon atoms.
- Examples of the linear or branched alkenyl group having 2 to 15 carbon atoms represented by R include a vinyl group, an allyl group, a butenyl group, a pentenyl group, a hexenyl group, a heptenyl group and an octenyl group, and these groups. Examples include branched groups.
- Examples of the aryl group having 6 to 18 carbon atoms represented by R include a phenyl group, 1-naphthyl group, 2-naphthyl group, 1-anthryl group, 2-anthryl group, 9-anthryl group, 1-phenanthryl group, 2- Examples thereof include a phenanthryl group, a 3-phenanthryl group, a 4-phenanthryl group, and a 9-phenanthryl group.
- alkyl group having 1 to 10 carbon atoms examples include linear, branched or cyclic alkyl groups, such as methyl group, ethyl group, n -Propyl group, i-propyl group, cyclopropyl group, n-butyl group, i-butyl group, s-butyl group, t-butyl group, cyclobutyl group, 1-methyl-cyclopropyl group, 2-methyl-cyclopropyl Group, n-pentyl group, 1-methyl-n-butyl group, 2-methyl-n-butyl group, 3-methyl-n-butyl group, 1,1-dimethyl-n-propyl group, 1,2-dimethyl group -N-propyl group, 2,2-dimethyl-n-propyl group, 1-ethyl-n-propyl group, cyclopentyl group, 1-methyl-cyclobutyl groups, such as methyl group, ethyl group, n
- alkoxy group having 1 to 10 carbon atoms as the substituent examples include groups in which the above alkyl group is bonded to oxygen, such as a methoxy group, an ethoxy group, an n-propoxy group, an i-propoxy group, and an n-butoxy group.
- Examples of the aryloxy group having 6 to 18 carbon atoms as the substituent include groups in which the aryl group is bonded to oxygen.
- a phenyloxy group phenoxy group
- ⁇ -naphthyloxy group ⁇ -naphthyloxy group
- ⁇ -naphthyloxy group and the like.
- Examples thereof include a naphthyloxy group and a 9-phenanthryloxy group.
- halogen atom examples include a chlorine atom, a bromine atom, and an iodine atom.
- alkylcarbonyl group having 2 to 10 carbon atoms as the substituent examples include groups in which the alkyl group is bonded to a carbonyl group, such as a methylcarbonyl group, an ethylcarbonyl group, an n-propylcarbonyl group, i- Propylcarbonyl group, cyclopropylcarbonyl group, n-butylcarbonyl group, i-butylcarbonyl group, s-butylcarbonyl group, t-butylcarbonyl group, cyclobutylcarbonyl group, 1-methyl-cyclopropylcarbonyl group, 2-methyl -Cyclopropylcarbonyl group, n-pentylcarbonyl group, 1-methyl-n-butylcarbonyl group, 2-methyl-n-butylcarbonyl group, 3-methyl-n-butylcarbonyl group, 1,1-dimethyl-n- Propylcarbonyl group, 1,2-dimethyl-but
- Examples of the aralkyl group having 7 to 18 carbon atoms as the substituent include groups in which a hydrogen atom of the alkyl group is substituted with an aryl group, such as a benzyl group, a phenethyl group, a 3-phenylpropyl group, a 4-phenylbutyl group, 5-phenylpentyl group, 6-phenylhexyl group, ⁇ -naphthylmethyl group, ⁇ -naphthylmethyl group, 1-anthrylmethyl group, 2-anthrylmethyl group, 9-anthrylmethyl group, 1-phenanthryl Methyl group, 2-phenanthrylmethyl group, 3-phenanthrylmethyl group, 4-phenanthrylmethyl group, 9-phenanthrylmethyl group, ⁇ -naphthylethyl group, ⁇ -naphthylethyl group, 1-anthone Tolylethyl group, 2-anthrylethyl group, 9-
- the S g is preferably a monovalent group having a structure derived from a disaccharide, Most preferably, it is a monovalent group having a structure derived from lactose.
- A is preferably a linear alkylene group having 1 to 15 carbon atoms or an arylene group having 6 to 15 carbon atoms, and a linear alkylene group having 1 to 10 carbon atoms or carbon.
- An arylene group having 6 to 10 atoms is more preferable, and a phenylene group is most preferable.
- R is preferably a linear alkyl group having 1 to 15 carbon atoms, more preferably a linear alkyl group having 4 to 15 carbon atoms, and a straight chain having 4 to 12 carbon atoms.
- a chain alkyl group is particularly preferable, and a butyl group, an n-octyl group and a dodecyl group are most preferable.
- R is preferably a cyclic alkyl group having 3 to 10 carbon atoms, and most preferably a cyclohexyl group.
- R is a phenyl group which is unsubstituted or substituted with at least one substituent selected from the group consisting of an alkyl group having 1 to 10 carbon atoms, a halogen atom and an aralkyl group having 7 to 18 carbon atoms. Preferably there is. More preferably, R is substituted with at least one substituent selected from the group consisting of an unsubstituted or alkyl group having 1 to 5 carbon atoms, a halogen atom, and an aralkyl group having 7 to 10 carbon atoms.
- R is preferably a phenyl group, and particularly from the viewpoint of expressing better gelation ability, the above R is an unsubstituted phenyl group, or a phenyl group substituted with a tert-butyl group, a benzyl group, or a bromo group. Is preferred.
- the compound represented by the formula [1] which is the hydrogelator of the present invention, is self-assembled when it is added to various aqueous solvents described later to form secondary aggregates such as fiber or lamellar, These are considered to contribute to the gelation of the solvent. Therefore, as said S g , A and R in the formula [1], the compound represented by the formula [1] with respect to the high affinity of the secondary assembly to the solvent to be gelled or the solvent to be gelled In consideration of the high solubility as, a suitable or optimal group can be selected.
- the compound represented by the said Formula [1] is also the object of this invention.
- the definitions of S g , A and R in the compound represented by the formula [1] are as described in [Hydrogelling agent].
- the compound represented by the formula [1] can be easily synthesized, for example, by the following method (Step 1 to Step 3).
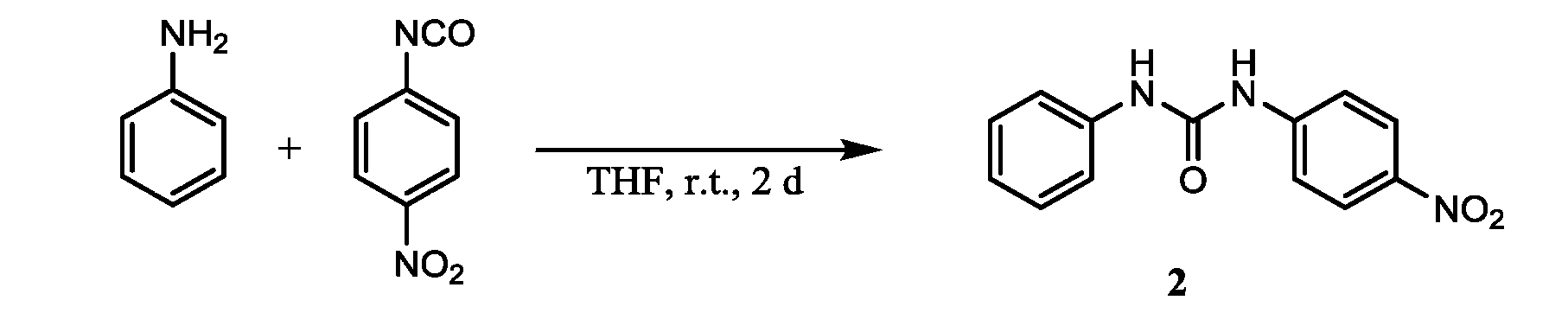
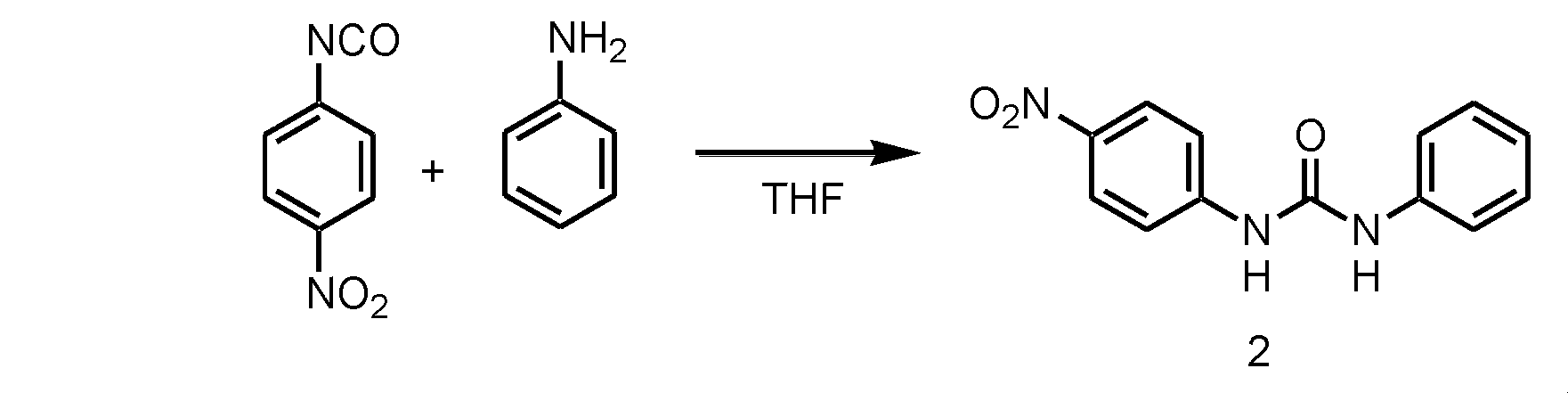
- step 1 the NO 2 substituted ester compound (a) of isocyanic acid is reacted with the amine compound (b).
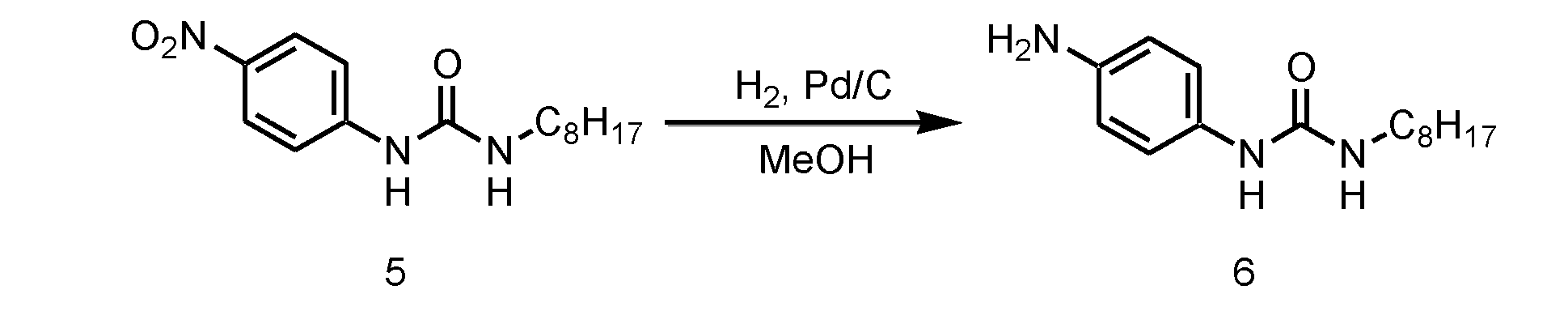
- step 2 the nitro group of the monourea compound (c) obtained in step 1 is reduced.
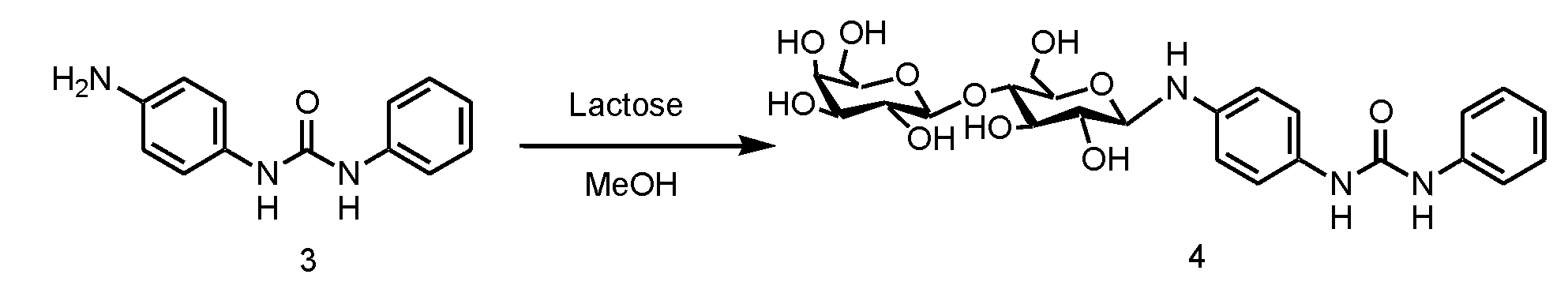
- step 3 the monourea compound (d) having an amino group obtained in step 2 is reacted with sugar (e).
- the usable organic solvent is not particularly limited as long as it dissolves the NO 2 substituted ester compound (a) of isocyanate and the amine compound (b).
- alcohols for example, methanol, ethanol, etc.
- cellosolves eg, methoxyethanol, ethoxyethanol, etc.
- aprotic polar organic solvents eg, N, N-dimethylformamide (DMF), dimethyl sulfoxide (DMSO), N, N— Dimethylacetamide, tetramethylurea, sulfolane, N-methylpyrrolidone, N, N-dimethylimidazolidinone, etc.
- ethers eg diethyl ether, diisopropyl ether, t-butyl methyl ether (TBME), tetrahydrofuran, geo Xanthine
- aliphatic hydrocarbons alipha
- the reaction can be appropriately selected from room temperature (around 25 ° C.) to the reflux temperature of the organic solvent used in the above reaction, and the reaction time can be appropriately selected from about 1 hour to 5 days.
- the solvent is distilled off, and if necessary, it can be purified by various purification methods such as various chromatographic methods, recrystallization methods, reprecipitation methods, distillation methods, and washing.
- the reaction in Step 2 is catalytic reduction.
- a known method such as a method using a hydrogenation catalyst such as nickel (Ni) or palladium (Pd) can be used.
- the solvent is distilled off, and if necessary, it can be purified by various purification methods such as various chromatographic methods, recrystallization methods, reprecipitation methods, distillation methods, and washing.
- the reaction in Step 3 is a dehydration reaction, and examples of the organic solvent that can be used for the dehydration reaction include the organic solvents mentioned in Step 1 above.
- the reaction can be appropriately selected from room temperature (around 25 ° C.) to the reflux temperature of the organic solvent used in the above reaction, and the reaction time can be appropriately selected from about 1 hour to 14 days.
- the solvent is distilled off, and if necessary, it can be purified by various purification methods such as various chromatographic methods, recrystallization methods, reprecipitation methods, distillation methods, and washing.
- the hydrogel of the present invention can be obtained by gelling a solvent with the above hydrogelator. Specifically, a manufacturing method in which a predetermined amount of a hydrogelator is heated and dissolved in a solvent and cooled, and a manufacturing method in which a predetermined amount of the hydrogelator is added to the solvent and subjected to ultrasonic treatment, vibration treatment, or the like. Is exemplified. Usually, it is preferable to dissolve completely by heating. In addition, when performing ultrasonic treatment or vibration treatment, the hydrogelator may or may not be heated. In this specification, gelation means that a fluid liquid substantially loses fluidity.
- the amount of the hydrogelator of the present invention is not particularly limited as long as the effect of the present invention is exhibited, but is usually 0.001 to 20% by mass with respect to the mass of the solvent to be gelled, For example, 0.05 to 5% by mass. In this specification and the like, mass% is also expressed as wt%.
- the solvent is not particularly limited as long as it does not prevent gelation, and preferred specific examples include an aqueous solvent.
- the hydrogel of the present invention can be formed containing the hydrogelator and an aqueous solvent.
- the “aqueous solvent” in the present invention is not particularly limited as long as it contains at least water as a solvent.
- water a buffer solution, an inorganic aqueous solution (an acidic aqueous solution, a basic aqueous solution, a neutral aqueous solution, etc.), And a mixed solvent of a hydrophilic organic solvent (referred to herein as a hydrophilic organic solution) and the like.
- buffer examples include sodium phosphate buffer, tris-HCl (Tris-HCl) buffer, 4- (2-hydroxyethyl) -1-piperazineethanesulfonic acid-sodium hydroxide ( HEPES-NaOH buffer, sodium acetate (NaOAc) buffer, borate buffer (Borate), borate-sodium hydroxide (Borate NaOH) buffer, tris-glycine-sodium dodecyl sulfate (Tris-glycine-SDS) Buffer, Tris-Borate-Ethylenediaminetetraacetic acid (Tris-Borate-EDTA) buffer, Phosphate buffered saline (PBS), Glycine-sodium hydroxide (Glycine-NaOH) buffer, Glycine-hydrochloric acid (Glycine- HCl) buffer , Phosphate (Phosphate) buffer, and sodium dihydrogen phosphate - sodium hydroxide
- the inorganic aqueous solution examples include an aqueous solution of an acid such as hydrochloric acid, an aqueous solution of an alkali such as sodium hydroxide, an aqueous solution of an inorganic salt such as a sodium salt or an ammonium salt (including seawater, saline, and physiological saline). It is done.
- an aqueous solution of an acid such as hydrochloric acid
- an aqueous solution of an alkali such as sodium hydroxide
- an aqueous solution of an inorganic salt such as a sodium salt or an ammonium salt (including seawater, saline, and physiological saline). It is done.
- the hydrophilic organic solvent in the hydrophilic organic solution is not particularly limited as long as it is an organic solvent that dissolves in water at an arbitrary ratio, and examples thereof include alcohol, acetone, cyclohexanone, acetonitrile, dioxane, glycerol, and dimethyl sulfoxide.
- the alcohol is preferably a water-soluble alcohol that is freely soluble in water, and more preferable examples include alcohols having 1 to 9 carbon atoms, polyhydric alcohols, higher alcohols, and glycerides.
- examples of the alcohol having 1 to 9 carbon atoms include methanol, ethanol, 2-propanol, i-butanol, pentanol, hexanol, 1-octanol, isooctanol, and the like.
- examples of the polyhydric alcohol include butanediol, Examples of the higher alcohol include octyldodecanol, stearyl alcohol, and oleyl alcohol; examples of the glycerides include trioctanoin, tri (caprylcaprylic acid) glyceryl, and glyceryl stearate.
- hydrophilic organic solvent used in the hydrophilic organic solution methanol, ethanol, 2-propanol, i-butanol, pentanol, hexanol, 1-octanol, isooctanol, acetone, cyclohexanone, acetonitrile, dioxane Glycerol, butanediol, propylene glycol, ethylene glycol and dimethyl sulfoxide are preferred, and ethanol and butanediol are more preferred.
- the ratio of the hydrophilic organic solvent in the hydrophilic organic solution used in the present invention is not particularly limited, and can be, for example, 10 wt% to 90 wt%.
- hydrogelator of the present invention can also be applied to a medium, which includes Dulbecco's modified Eagle medium (D-MEM), Roswell Park Memorial Institute 1640 medium (RPMI 1640 medium), and Ham F12 medium ( F12 medium) and the like.
- D-MEM Dulbecco's modified Eagle medium
- RPMI 1640 medium Roswell Park Memorial Institute 1640 medium
- F12 medium Ham F12 medium
- the hydrogelator of the present invention is heated and stirred or sonicated as necessary, in addition to the aforementioned water, buffer, medium, inorganic aqueous solution, hydrophilic organic solution, etc., which are media, and then left at room temperature. By doing so, a hydrogel can be obtained.
- the gel strength can be adjusted by the concentration of the hydrogelator.
- the hydrogel formed by the hydrogelator of the present invention has various additives (surfactant, ultraviolet absorber, moisturizer, etc.) as required, such as its application, within a range that does not inhibit the gelation ability of the hydrogelator.
- additives surfactant, ultraviolet absorber, moisturizer, etc.
- Organic compounds such as agents, preservatives, antioxidants, fragrances, and physiologically active substances (medicinal ingredients), and inorganic compounds such as titanium oxide, talc, mica, and water) can be mixed.
- the hydrogelator of the present invention can gel various aqueous solvents as described above, the hydrogelator of the present invention and the hydrogel obtained therefrom are used as a cosmetic substrate or a medical substrate, a gel electrolyte, a cell culture medium.
- Materials in various fields such as materials, base materials for preserving biomolecules such as cells and proteins, base materials for external use, base materials for biochemistry, base materials for foods, contact lenses, disposable diapers, artificial actuators, base materials for dry land agriculture Can be used for In addition, it can be widely used as a bioreactor carrier for enzymes and the like in research, medicine, analysis, and various industries.
- the apparatus and conditions used for sample preparation and physical property analysis are as follows.
- 1 H-NMR spectrum apparatus JNM ECA-600 (manufactured by JEOL Ltd.)
- Vortex mixer Device VORTEX3 (manufactured by IKA)
- Transmittance Device JASCO V-630 spectrometer (manufactured by JASCO Corporation)
- SEM Scanning electron microscope Apparatus: JEOL JSM-6300 spectrometer (manufactured by JEOL Ltd.)
- Example 2 Gel forming ability of hydrogelator (1)
- the three compounds synthesized in Example 1 were used as various aqueous solvents (pure water, phosphate buffer, physiological saline, 30% 1,3-propanediol, 70% 1,3-propanediol, The gel-forming ability was evaluated with respect to 30% ethanol and 70% ethanol.
- the gelation test was performed as follows. The hydrogelator was weighed into a 2.0 mL screw mouth sample tube, and various aqueous solvents were added thereto to prepare samples each having a hydrogelator concentration of 1.0 mass%. And it heated and melt
- the hydrogelator of the present invention can form a gel with respect to an aqueous solvent.
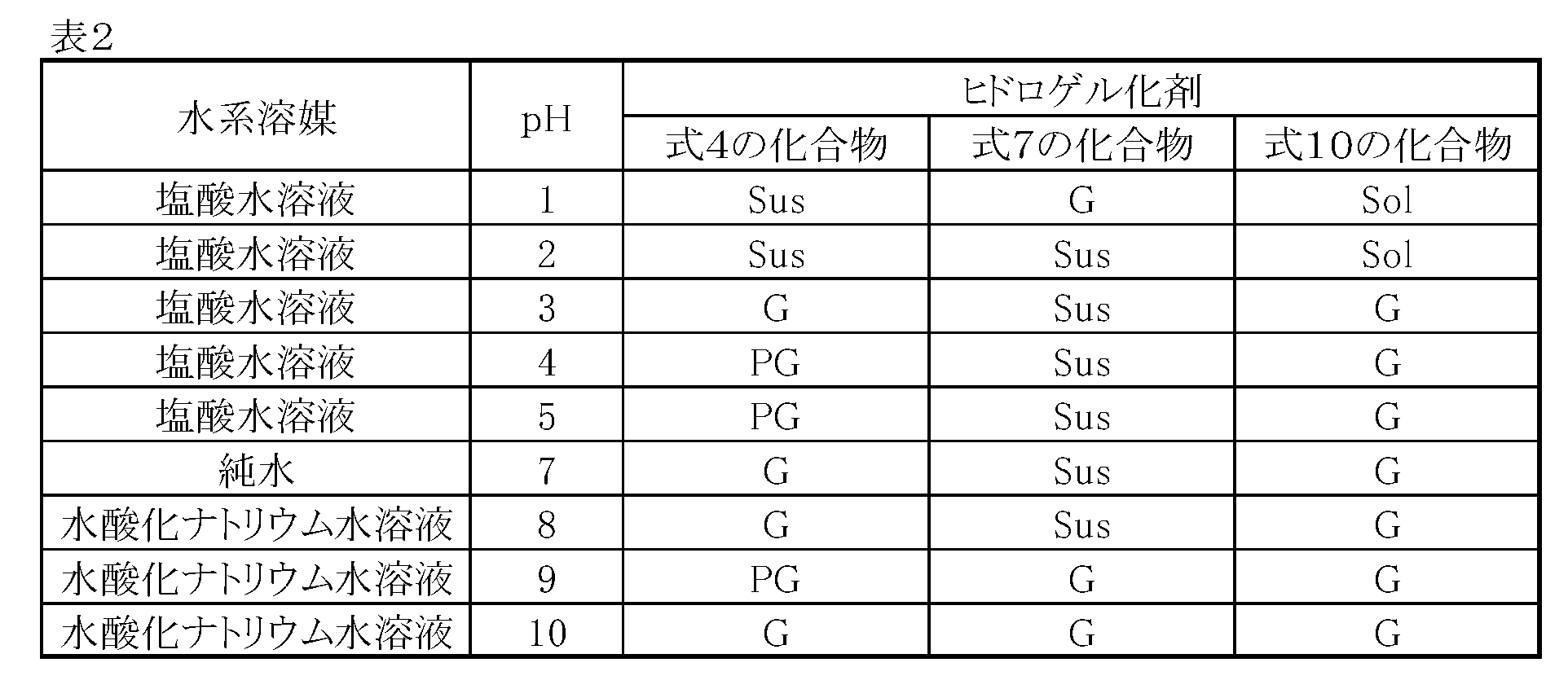
- Example 3 pH responsiveness of hydrogelator
- aqueous solvents hydroochloric acid aqueous solution, pure water, sodium hydroxide aqueous solution having various pHs were gelled, and the pH responsiveness of the gelation ability was examined.
- the gelation test was performed in the same procedure as in [Example 2].
- the evaluation results are shown in Table 2.
- the photograph of each sample tube after standing is shown in FIG. 4 (photograph of each sample tube after cooling in the gelation test of the hydrogelator comprising the compound of formula 4), and FIG. 5 (hydrogelation comprising the compound of formula 7).
- FIG. 6 photograph of each sample tube after cooling in the gelation test of the hydrogelator comprising the compound of formula 10) shows the photograph of each sample tube after cooling in the gelation test of the agent.
- the hydrogelator of the present invention can form a gel with respect to an aqueous solvent having a pH in the range of 1 to 10.
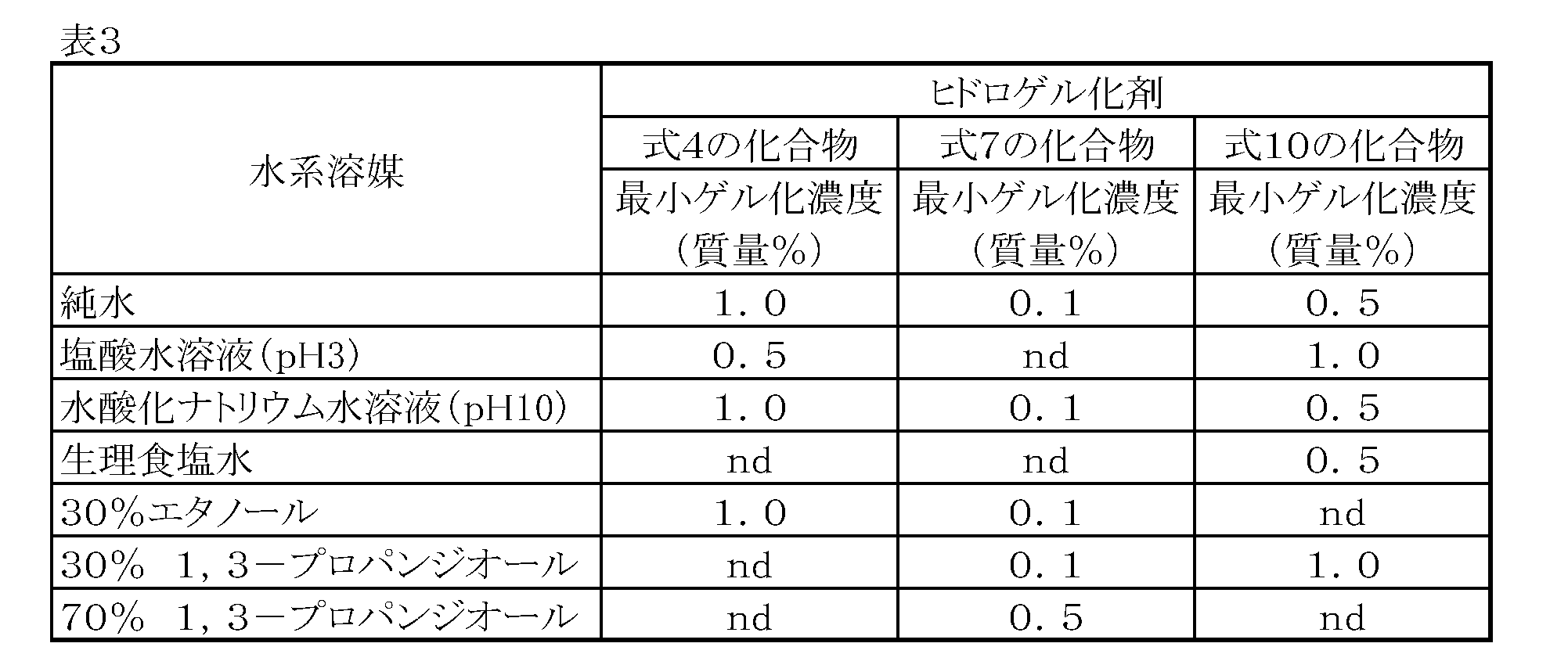
- Example 4 Evaluation of minimum gelation concentration of hydrogelator
- the three compounds synthesized in Example 1 were used as hydrogelators, and various aqueous solvents [pure water, hydrochloric acid aqueous solution (pH 3), sodium hydroxide aqueous solution (pH 10), physiological saline, 30% ethanol, 30% 1,3 -Minimal gelation concentration for propanediol, 70% 1,3-propanediol] [Concentration to form a gel that does not collapse even when the sample tube is inverted (not including partially gelled gel)] Asked.
- the gelation test was performed in the same procedure as in [Example 2]. The evaluation results are shown in Table 3. Fig.
- FIG. 9 shows a photograph of each sample tube after cooling in the gelation test of the agent.
- Example 5 Gel forming ability of hydrogelator (2)
- gel forming ability was evaluated for various gelation treatments [heat treatment (40 ° C. and 60 ° C.), ultrasonic treatment, vibration treatment]. .
- the gelation test by various gelation treatments was performed as follows.
- the hydrogelator was weighed into a 2.0 mL screw mouth sample tube, and pure water was added thereto to prepare a sample having a hydrogelator concentration of 1.0 mass%. Then, using a handy massager (manufactured by SLIVE), vibration was applied to the lower part of the sample tube for about 3 minutes. Then, it left still for 30 minutes at room temperature, and observed formation of the gel.
- Table 4 shows the evaluation results. Further, photographs of the sample tubes after standing are shown in FIG. 10 [photograph of a sample tube cooled after heating treatment (40 ° C.) of a hydrogelator comprising a compound of formula 10] and FIG. 11 [comprising a compound of formula 10 Photo of the sample tube cooled after the heat treatment (60 ° C.) of the hydrogelator], FIG. 12 [Photo of the sample tube allowed to stand after sonication of the hydrogelator comprising the compound of formula 10] and FIG. [Photo of a sample tube which was allowed to stand after vibration treatment of a hydrogelator comprising a compound of formula 10]
- the hydrogelator of the present invention can form a hydrogel by performing ultrasonic treatment or vibration treatment at room temperature in addition to heat treatment.
- Example 7 Gel forming ability of hydrogelator (3)
- the hydrogelator the compound represented by Formula 4 synthesized in Example 6
- 500 ⁇ L of pure water was added thereto, and the concentration of the hydrogelator was 1.0% by mass.
- Samples were prepared. And it was made to heat-dissolve on a 120 degreeC hotplate. Then, when the obtained solution was left still at room temperature, the cloudy hydrogel was obtained.
- a photograph of the sample tube after cooling in the gelation test is shown in FIG.
- Example 8 Gel forming ability of hydrogelator (4)
- the hydrogelator the compound represented by formula 7 synthesized in Example 6
- 500 ⁇ L of pure water was added thereto, and the concentration of the hydrogelator was Samples of 0.1 wt% and 0.5 wt% were prepared. And it was made to heat-dissolve on a 120 degreeC hotplate. Then, when each obtained solution was left still at room temperature, hydrogel was obtained, respectively. The minimum gelation concentration was 0.1% by mass, and a transparent hydrogel was produced.
- a photograph of the sample tube after cooling in the gelation test is shown in FIG.
- the compound represented by Formula 7 synthesized in Example 6 includes hydrochloric acid or aqueous sodium hydroxide solution having a pH of 4 to 9, physiological saline, a mixture of ethanol and water, and 1,3-propanediol. Gel formation was also possible with a mixed solution of water.
- Example 9 Gel forming ability of hydrogelator (5)
- a sample was prepared by adding a hydrogelator (compound represented by Formula 10 synthesized in Example 6) and 500 ⁇ L of pure water to a sample tube. And it was made to heat-dissolve on a 120 degreeC hotplate. Then, when the obtained solution was left still at room temperature, hydrogel was obtained. The minimum gelation concentration was 0.3% by mass, and a transparent hydrogel was produced.
- the photograph of the sample tube after standing to cool in the gelation test is shown in FIG.
- Example 10 Evaluation of transparency of hydrogel
- a hydrogelator (compound represented by Formula 10 synthesized in Example 6) was added to the sample tube, and pure water was added thereto, so that the concentration of the hydrogelator was 0.1% by mass and 0.3% by mass, respectively.
- %, 0.5 wt%, 0.75 wt% and 1.0 wt% samples were prepared. And it was made to heat-dissolve on a 120 degreeC hotplate. Thereafter, when each of the obtained solutions was allowed to stand at room temperature, the hydrogelling agent having a concentration of 0.1% by mass was in a non-viscous state (sol). was gotten. And the transmittance
- Example 11 Gel forming ability of hydrogelator (6)
- a hydrogelator Using the compound represented by Formula 10 synthesized in Example 6 as a hydrogelator, gel forming ability with respect to vibration treatment was evaluated.
- the gelation test by vibration treatment was performed as follows. A sample was prepared by adding a hydrogelator and pure water to the sample tube. Then, using a vortex mixer, vibration was applied to the lower part of the sample tube for about 5 minutes (2500 rpm). Then, when left still at room temperature, hydrogel was obtained (FIG. 18). The minimum gelation concentration was 0.5% by mass.
- the compound represented by Formula 10 synthesized in Example 6 includes hydrochloric acid or sodium hydroxide aqueous solution having a pH of 3 to 10, physiological saline, phosphate buffer, a mixture of ethanol and water, and 1 Gels could be formed even with a mixture of 1,3-propanediol and water by vibration treatment.
- Example 12 Scanning electron microscope observation of hydrogel
- a hydrogelator compound represented by Formula 10 synthesized in Example 6
- 500 ⁇ L of pure water is added thereto, and the concentration of the hydrogelator is 1.0% by mass.
- Samples were prepared. And it was made to heat-dissolve on a 120 degreeC hotplate. Then, when the obtained solution was left still at room temperature, hydrogel was obtained. And the obtained hydrogel was observed with the scanning electron microscope (SEM).
- SEM scanning electron microscope
- a photograph of a scanning electron microscope (SEM) image is shown in FIG. As shown in FIG. 19, fibrous aggregates were observed.
- Example 14 Gel forming ability of hydrogelator (7)
- a hydrogelator (compound represented by Formula 13) was added to the sample tube, and 500 ⁇ L of pure water was added thereto to prepare a sample. And it was made to heat-dissolve on a 120 degreeC hotplate. Then, when the obtained solution was left still at room temperature, hydrogel was obtained. The minimum gelation concentration was 1.0% by mass, and a translucent hydrogel was produced.
- a photograph of the sample tube after cooling in the gelation test is shown in FIG.
- the compound represented by Formula 13 was able to form a gel with respect to hydrochloric acid having a pH of 4 to 10 or a sodium hydroxide aqueous solution, physiological saline, and a mixed solution of ethanol and water.
- Example 15 Gel forming ability of hydrogelator (8)
- a sample was prepared by adding a hydrogelator (compound represented by Formula 16) and pure water to a sample tube. And it was made to heat-dissolve on a 120 degreeC hotplate. Thereafter, when the obtained solution was allowed to stand at room temperature, a suspension was obtained without gelation.
- the compound represented by Formula 16 was able to form a gel with respect to a mixed solution of ethanol and water and a mixed solution of 1,3-propanediol / water.
Landscapes
- Chemical & Material Sciences (AREA)
- Health & Medical Sciences (AREA)
- Organic Chemistry (AREA)
- Life Sciences & Earth Sciences (AREA)
- Engineering & Computer Science (AREA)
- General Health & Medical Sciences (AREA)
- Biochemistry (AREA)
- Molecular Biology (AREA)
- Biotechnology (AREA)
- Genetics & Genomics (AREA)
- Materials Engineering (AREA)
- Public Health (AREA)
- Epidemiology (AREA)
- Veterinary Medicine (AREA)
- Animal Behavior & Ethology (AREA)
- Chemical Kinetics & Catalysis (AREA)
- Pharmacology & Pharmacy (AREA)
- Medicinal Chemistry (AREA)
- General Chemical & Material Sciences (AREA)
- Oil, Petroleum & Natural Gas (AREA)
- Birds (AREA)
- Cosmetics (AREA)
- Organic Low-Molecular-Weight Compounds And Preparation Thereof (AREA)
- Medicinal Preparation (AREA)
- General Preparation And Processing Of Foods (AREA)
- Saccharide Compounds (AREA)
Abstract
Description
これまでにも、種々のヒドロゲルやヒドロゲル化剤が提案されており、その一例として、各種単糖類から誘導された糖誘導体を用いたゲル化剤が報告されている(特許文献1)。
本発明は、簡便な手法によって作製することができる、これまで提案されていない構造を有する新規なゲル化剤、特にヒドロゲルを形成できる新規なゲル化剤を提供することを課題とする。
Sgは糖基を表し、
Aは2価の連結基を表し、
Rは、炭素原子数1乃至15の直鎖状若しくは分岐鎖状のアルキル基、炭素原子数3乃至15の環状のアルキル基、炭素原子数2乃至15の直鎖状若しくは分岐鎖状のアルケニル基、又は未置換の若しくは炭素原子数1乃至10のアルキル基、炭素原子数1乃至10のアルコキシ基、炭素原子数6乃至18のアリールオキシ基、ハロゲン原子、ニトロ基、フェニル基、炭素原子数2乃至10のアルキルカルボニル基及び炭素原子数7乃至18のアラルキル基からなる群から選択される少なくとも1つの置換基で置換された炭素原子数6乃至18のアリール基を表す。)
第2観点として、前記糖基Sgが単糖又は二糖由来の構造からなる1価の基である、第1観点に記載のヒドロゲル化剤に関する。
第3観点として、前記連結基Aが炭素原子数1乃至15の直鎖状アルキレン基又は炭素原子数6乃至15のアリーレン基であり、
前記Rが炭素原子数1乃至15の直鎖状のアルキル基、炭素原子数3乃至10の環状のアルキル基、又は未置換の若しくは炭素原子数1乃至10のアルキル基、ハロゲン原子及び炭素原子数7乃至18のアラルキル基からなる群から選択される少なくとも1つの置換基で置換されたフェニル基である、
第2観点に記載のヒドロゲル化剤に関する。
第4観点として、第1観点乃至第3観点のいずれか1項に記載のヒドロゲル化剤と、水系溶媒よりなるヒドロゲルに関する。
第5観点として、下記式[1]で表される化合物に関する。
Sgは糖基を表し、
Aは2価の連結基を表し、
Rは、炭素原子数1乃至15の直鎖状若しくは分岐鎖状のアルキル基、炭素原子数3乃至15の環状のアルキル基、炭素原子数2乃至15の直鎖状若しくは分岐鎖状のアルケニル基、又は未置換の若しくは炭素原子数1乃至10のアルキル基、炭素原子数1乃至10のアルコキシ基、炭素原子数6乃至18のアリールオキシ基、ハロゲン原子、ニトロ基、フェニル基、炭素原子数2乃至10のアルキルカルボニル基及び炭素原子数7乃至18のアラルキル基からなる群から選択される少なくとも1つの置換基で置換された炭素原子数6乃至18のアリール基を表す。)
また、本発明のヒドロゲル化剤は、高いゲル化能を有するという効果を奏する。
さらに、本発明のヒドロゲル化剤は、簡便な手法によって作製することができるという効果を奏する。
本発明のヒドロゲル化剤は、下記式[1]で表される、糖構造を有するモノウレア化合物からなる。
Sgは糖基を表し、
Aは2価の連結基を表し、
Rは、炭素原子数1乃至15の直鎖状若しくは分岐鎖状のアルキル基、炭素原子数3乃至15の環状のアルキル基、炭素原子数2乃至15の直鎖状若しくは分岐鎖状のアルケニル基、又は未置換の若しくは炭素原子数1乃至10のアルキル基、炭素原子数1乃至10のアルコキシ基、炭素原子数6乃至18のアリールオキシ基、ハロゲン原子、ニトロ基、フェニル基、炭素原子数2乃至10のアルキルカルボニル基及び炭素原子数7乃至18のアラルキル基からなる群から選択される少なくとも1つの置換基で置換された炭素原子数6乃至18のアリール基を表す。)
前記糖基は、アルキル基(メチル基やエチル基等)及びスルホ基等の置換基によって置換されていてもよい。
前記糖の種類は特に限定されないが、ゲル化活性の観点から、単糖又は二糖であることが好ましく、二糖であることがより好ましい。
前記5炭糖としては、リボース、デオキシリボース、及びフルクトース等が挙げられる。
前記6炭糖としては、グルコース、マンノース、ガラクトース、メチル-α-グルコース、及びメチル-α-マンノース等が挙げられる。
単糖の中でも、6炭糖が好ましく、グルコース、マンノース、ガラクトース、メチル-α-グルコース、及びメチル-α-マンノースが特に好ましい。
前記炭素原子数3乃至15の環状のアルキレン基としては、環状のアルキレン基のみからなる基(例えば、シクロプロピレン基、シクロブチレン基、シクロヘキセン基等)だけでなく、シクロペンチル環、シクロヘキシル環等の環構造を有する直鎖状及び/又は分岐鎖状のアルキレン基であって、炭素原子数が3乃至15の基であるものも挙げられる。
前記炭素原子数6乃至15のアリーレン基としては、フェニレン基、ナフチレン基、アントリレン基等が挙げられる。
Rが表す炭素原子数3乃至15の環状のアルキル基としては、環状のアルキル基のみからなる基(例えば、シクロプロピル基、シクロブチル基、シクロペンチル基、シクロヘキシル基、シクロヘプチル基等)だけでなく、シクロペンチル環、シクロヘキシル環等の環構造を有する直鎖状及び/又は分岐鎖状のアルキル基であって、炭素原子数が3乃至15の基であるものも挙げられる。
Rが表す炭素原子数2乃至15の直鎖状又は分岐鎖状のアルケニル基としては、ビニル基、アリル基、ブテニル基、ペンテニル基、ヘキセニル基、ヘプテニル基及びオクテニル基等、並びにこれらの基が分岐されてなる基が挙げられる。
また前記式[1]において、上記Aは炭素原子数1乃至15の直鎖状アルキレン基又は炭素原子数6乃至15のアリーレン基が好ましく、炭素原子数1乃至10の直鎖状アルキレン基又は炭素原子数6乃至10のアリーレン基がより好ましく、最も好ましくはフェニレン基である。
さらに前記式[1]において、上記Rは炭素原子数1乃至15の直鎖状アルキル基が好ましく、炭素原子数4乃至15の直鎖状アルキル基がより好ましく、炭素原子数4乃至12の直鎖状アルキル基が特に好ましく、最も好ましくはブチル基、n-オクチル基及びドデシル基である。
また上記Rは、炭素原子数3乃至10の環状のアルキル基が好ましく、最も好ましくはシクロヘキシル基である。
さらに上記Rは、未置換の又は炭素原子数1乃至10のアルキル基、ハロゲン原子及び炭素原子数7乃至18のアラルキル基からなる群から選択される少なくとも1つの置換基で置換されたフェニル基であることが好ましい。より好ましくは、上記Rは、未置換の又は炭素原子数1乃至5のアルキル基、ハロゲン原子及び炭素原子数7乃至10のアラルキル基からなる群から選択される少なくとも1つの置換基で置換されたフェニル基であることが好ましく、特により良好なゲル化能を発現する観点から、上記Rは、未置換のフェニル基、又は、tert-ブチル基、ベンジル基若しくはブロモ基置換のフェニル基であることが好ましい。
なお、前記式[1]で表される化合物中のSg、A及びRの定義については、[ヒドロゲル化剤]で記載したとおりである。
反応は、室温(25℃前後)乃至上記反応に使用する有機溶媒の還流温度以下にて適宜選択され得、反応時間は1時間~5日間程度から適宜選択され得る。
反応終了後、溶媒を留去し、必要に応じて各種クロマトグラフィー法、再結晶法、再沈殿法、蒸留法、洗浄等の公知の精製方法にて精製することができる。
反応終了後、溶媒を留去し、必要に応じて各種クロマトグラフィー法、再結晶法、再沈殿法、蒸留法、洗浄等の公知の精製方法にて精製することができる。
反応は、室温(25℃前後)乃至上記反応に使用する有機溶媒の還流温度以下にて適宜選択され得、反応時間は1時間~14日間程度から適宜選択され得る。
反応終了後、溶媒を留去し、必要に応じて各種クロマトグラフィー法、再結晶法、再沈殿法、蒸留法、洗浄等の公知の精製方法にて精製することができる。
本発明のヒドロゲルは、上記ヒドロゲル化剤で溶媒をゲル化させることにより、得ることができる。具体的には、溶媒に所定量のヒドロゲル化剤を加熱溶解させ、冷却するという製造方法、並びに溶媒に所定量のヒドロゲル化剤を加えて、超音波処理や振動処理などを施すという製造方法などが例示される。通常、加熱溶解の際には、完全に溶解させることが好ましい。また、超音波処理や振動処理などを施す際には、ヒドロゲル化剤は加熱してもしなくてもよい。
なお、本明細書において、ゲル化とは、流動性のある液体が実質的に流動性を失った状態となることをいう。
溶媒をゲル化するに際し、本発明のヒドロゲル化剤の使用量は、本発明の効果を奏する限り特に限定されないが、ゲル化される溶媒の質量に対して、通常0.001乃至20質量%、例えば0.05乃至5質量%である。なお、本明細書等では、質量%をwt%とも表記する。
本発明のヒドロゲルは、前記ヒドロゲル化剤と、水系溶媒を含みて形成され得る。
前記アルコールは、好ましくは水に自由に溶解する水溶性アルコールであり、より好ましくは炭素原子数1乃至9のアルコール、多価アルコール、高級アルコール、グリセライド類が挙げられる。
具体的には、炭素原子数1乃至9のアルコールとしては、メタノール、エタノール、2-プロパノール、i-ブタノール、ペンタノール、ヘキサノール、1-オクタノール、イソオクタノール等;多価アルコールとしては、ブタンジオール、エチレングリコール、プロピレングリコール、ポリプロピレングリコール等;高級アルコールとしては、オクチルドデカノール、ステアリルアルコール、オレイルアルコール等;グリセライド類としてはトリオクタノイン、トリ(カプリルカプリル酸)グリセリル、ステアリン酸グリセリル等が挙げられる。
これらの中でも、前記親水性有機溶液に使用する親水性有機溶媒としては、メタノール、エタノール、2-プロパノール、i-ブタノール、ペンタノール、ヘキサノール、1-オクタノール、イソオクタノール、アセトン、シクロヘキサノン、アセトニトリル、ジオキサン、グリセロール、ブタンジオール、プロピレングリコール、エチレングリコール及びジメチルスルホキシドが好ましく、エタノール及びブタンジオールがより好ましい。
本発明で用いる親水性有機溶液における親水性有機溶媒の割合は特に限定されないが、例えば10wt%乃至90wt%とすることができる。
(1)1H-NMRスペクトル
装置:JNM ECA-600(日本電子株式会社製)
(2)ボルテックスミキサー
装置:VORTEX3(IKA社製)
(3)透過率
装置:JASCO V-630 spectrometer(日本分光株式会社製)
(4)走査型電子顕微鏡(SEM)
装置:JEOL JSM-6300 spectrometer(日本電子株式会社製)
<式4で表される化合物の合成>
1H-NMR (400MHz, Acetone-d6) δ: 8.79 (1H, s), 8.34 (1H, s), 8.16 (2H, dd, J = 7.1, 2.2 Hz), 7.76 (2H, td, J= 6.2, 3.7 Hz), 7.52 (2H, d, J = 7.8 Hz), 7.29-7.25 (2H, m), 7.00 (1H, t, J= 7.6 Hz).
1H-NMR (400MHz, Acetone-d6) δ: 7.97 (1H, s), 7.70 (1H, s), 7.52 (2H, d, J = 7.3 Hz), 7.22 (4H, dtd, J= 15.3, 6.1, 2.8 Hz), 6.95-6.91 (1H, m), 6.61 (2H, dt, J = 9.4, 2.4 Hz), 4.40 (1H, s).
1H-NMR (DMSO-d6) δ = 8.49 (1H, s), 8.22 (1H, s), 7.41 (2H, d, J = 7.8 Hz), 7.25 (2H, t, J = 7.8 Hz), 7.16 (2H, d, J = 8.8 Hz), 6.92 (1H, t, J = 7.6 Hz), 6.64 (2H, d, J = 8.8 Hz), 6.03 (1H, d, J = 7.8 Hz), 5.10 (1H, d, J = 3.9 Hz), 4.98 (1H, d, J = 5.4 Hz), 4.78 (1H, d, J = 5.4 Hz), 4.73 (1H, s), 4.66 (1H, t, J = 4.9 Hz), 4.51 (2H, dd, J = 10.5, 5.1 Hz), 4.38 (1H, t, J = 8.3 Hz), 4.24 (1H, d, J = 7.3 Hz), 3.70 (1H, s)
1H-NMR (400 MHz, DMSO-d6) δ = 9.19 (1H, s), 8.12 (2H, d, J = 9.3 Hz), 7.60 (2H, d, J= 9.3 Hz), 6.41 (1H, s), 3.08 (2H, q, J= 6.5 Hz), 1.42 (2H, s), 1.25 (11H, s), 0.83 (3H, t, J = 6.8 H)
1H-NMR (DMSO-d6) δ = 7.80 (1H, s), 6.98 (2H, d, J = 8.8 Hz), 6.45 (2H, d, J = 8.3 Hz), 5.85 (1H, s), 4.64 (1H, s), 3.01 (2H, q, J = 6.5 Hz), 1.38 (2H, s), 1.25 (11H, s), 0.86 (3H, t, J = 6.8 Hz)
1H-NMR (DMSO-d6) δ = 7.90 (1H, s), 7.08 (2H, d, J = 8.8 Hz), 6.58 (2H, d, J= 9.3 Hz), 5.90-5.87 (2H, m), 5.10 (1H, d, J= 3.9 Hz), 4.96 (1H, d, J = 5.4 Hz), 4.78 (1H, d, J = 4.9 Hz), 4.71 (1H, s), 4.67 (1H, t, J = 5.1 Hz), 4.50 (2H, dd, J = 12.4, 5.1 Hz), 4.35 (1H, t, J = 8.0 Hz), 4.23 (1H, d, J = 6.8 Hz), 3.70 (1H, dd, J = 10.0, 5.1 Hz), 3.02 (2H, q, J = 6.5 Hz), 1.39 (2H, t, J = 6.3 Hz), 1.26 (11H, s), 0.86 (3H, t, J = 6.8 Hz)
1H-NMR (ACETONE-d6) δ = 8.42 (1H, s), 8.13 (2H, td, J = 9.3, 2.4 Hz), 7.71 (2H, dt, J = 9.3, 2.4 Hz), 5.92 (1H, s), 3.62 (1H, s), 1.93-1.90 (2H, m), 1.72-1.70 (2H, m), 1.61-1.58 (1H, m), 1.40-1.34 (2H, m), 1.28-1.18 (3H, m).
1H-NMR (DMSO-d6) δ = 7.74 (1H, s), 6.98 (2H, d, J = 8.8 Hz), 6.45 (2H, d, J= 8.3 Hz), 5.80 (1H, d, J = 8.3 Hz), 4.64 (1H, s), 1.76-1.73 (2H, m), 1.63-1.61 (2H, m), 1.52-1.49 (1H, m), 1.28-1.25 (2H, m), 1.13-1.10 (3H, m)
1H-NMR (DMSO-d6) δ = 7.84 (1H, s), 7.08 (2H, d, J = 8.8 Hz), 6.58 (2H, d, J = 8.8 Hz), 5.91 (1H, d, J = 7.3 Hz), 5.84 (1H, d, J = 7.8 Hz), 5.10 (1H, d, J = 4.4 Hz), 4.97 (1H, d, J = 5.4 Hz), 4.79 (1H, d, J = 5.4 Hz), 4.72 (1H, s), 4.66 (1H, d, J = 5.4 Hz), 4.52-4.49 (2H, m), 4.35 (1H, t, J = 8.3 Hz), 4.23 (1H, d, J = 7.3 Hz)
実施例1で合成した3種の化合物をヒドロゲル化剤として、各種水系溶媒(純水、リン酸緩衝液、生理食塩水、30% 1,3-プロパンジオール、70% 1,3-プロパンジオール、30%エタノール、70%エタノール)に対するゲル形成能の評価を行った。
ゲル化試験は次のように行った。2.0mLのねじ口サンプル管に、ヒドロゲル化剤を量り取り、ここに各種水系溶媒を加えて、ヒドロゲル化剤の濃度が1.0質量%の試料をそれぞれ調製した。そして、120℃のホットプレート上にて加熱し、溶解させた。その後、室温にて一晩静置し、ゲルの形成を観察した。
静置放冷後、溶液の流動性が失われて、サンプル管を倒置しても溶液が流れ落ちない状態を「ゲル化」と判断した。表中の符号は形成したゲルの状態を示し(以下、本明細書中の他の表も同様である)、ゲル化したものは「G」、部分的にゲル化したものは「PG」、懸濁液の状態のものは「Sus」、サンプル管を倒置すると溶液が流れ落ち、溶液に粘性がない状態のものを「Sol」と評価した。また、評価しなかったものは「nd」とした。
評価結果を表1に示す。また静置後の各サンプル管の写真を図1(式4の化合物からなるヒドロゲル化剤のゲル化試験の放冷後の各サンプル管の写真)、図2(式7の化合物からなるヒドロゲル化剤のゲル化試験の放冷後の各サンプル管の写真)、及び図3(式10の化合物からなるヒドロゲル化剤のゲル化試験の放冷後の各サンプル管の写真)に示す。
水系溶媒として、種々のpHの水系溶媒(塩酸水溶液、純水、水酸化ナトリウム水溶液)のゲル化を行い、ゲル化能のpH応答性を調べた。ゲル化試験は[実施例2]の手順と同様の手順で行った。
評価結果を表2に示す。また静置後の各サンプル管の写真を図4(式4の化合物からなるヒドロゲル化剤のゲル化試験の放冷後の各サンプル管の写真)、図5(式7の化合物からなるヒドロゲル化剤のゲル化試験の放冷後の各サンプル管の写真)、及び図6(式10の化合物からなるヒドロゲル化剤のゲル化試験の放冷後の各サンプル管の写真)に示す。
実施例1で合成した3種の化合物をヒドロゲル化剤として、各種水系溶媒[純水、塩酸水溶液(pH3)、水酸化ナトリウム水溶液(pH10)、生理食塩水、30%エタノール、30% 1,3-プロパンジオール、70% 1,3-プロパンジオール]に対する最小ゲル化濃度[サンプル管を逆さにしてもゲルが崩壊しない程度のゲル(部分的にゲル化したものを含まない)を形成する濃度]を求めた。ゲル化試験は[実施例2]の手順と同様の手順で行った。
評価結果を表3に示す。また静置後の各サンプル管の写真を図7(式4の化合物からなるヒドロゲル化剤のゲル化試験の放冷後の各サンプル管の写真)、図8(式7の化合物からなるヒドロゲル化剤のゲル化試験の放冷後の各サンプル管の写真)、及び図9(式10の化合物からなるヒドロゲル化剤のゲル化試験の放冷後の各サンプル管の写真)に示す。
実施例1で合成した式10で表される化合物をヒドロゲル化剤として、各種ゲル化処理[加熱処理(40℃及び60℃)、超音波処理、振動処理]に対するゲル形成能の評価を行った。
各種ゲル化処理によるゲル化試験は次のように行った。
2.0mLのねじ口サンプル管に、所定量のヒドロゲル化剤を量り取り、ここに純水を加えて、表4に示す濃度(0.5質量%及び1.0質量%)の試料をそれぞれ調製した。そして、40℃のホットプレート上にて15分間加熱し、溶解させた。その後、室温にて30分間静置し、ゲルの形成を観察した。
加熱温度を60℃に変えた以外は、[加熱処理(40℃)]の手順と同様の手順で行った。
2.0mLのねじ口サンプル管に、ヒドロゲル化剤を量り取り、ここに純水を加えて、ヒドロゲル化剤の濃度が1.0質量%の試料を調製した。そして、ブランソン卓上超音波洗浄器(ブランソン社製2510J-DTH)を用いて、10分間超音波処理を行った。その後、室温にて30分間静置し、ゲルの形成を観察した。
2.0mLのねじ口サンプル管に、ヒドロゲル化剤を量り取り、ここに純水を加えて、ヒドロゲル化剤の濃度が1.0質量%の試料を調製した。そして、ハンディマッサージャー(スライヴ社製)を用いて、サンプル管の下部に振動を約3分間与えた。その後、室温にて30分間静置し、ゲルの形成を観察した。
<式4で表される化合物の合成>
サンプル管に、ヒドロゲル化剤(実施例6で合成した式4で表される化合物)5.05mgを量り取り、ここに純水500μLを加えて、ヒドロゲル化剤の濃度が1.0質量%の試料を調製した。そして、120℃のホットプレート上で加熱溶解させた。その後、得られた溶液を室温で静置すると、白濁したヒドロゲルが得られた。ゲル化試験の放冷後のサンプル管の写真を図14に示す。
サンプル管に、ヒドロゲル化剤(実施例6で合成した式7で表される化合物)0.52mg及び2.51mgをそれぞれ量り取り、ここに純水500μLを加えて、ヒドロゲル化剤の濃度がそれぞれ0.1質量%及び0.5質量%の試料を調製した。そして、120℃のホットプレート上で加熱溶解させた。その後、得られた各溶液を室温で静置すると、ヒドロゲルがそれぞれ得られた。最小ゲル化濃度は0.1質量%であり、透明なヒドロゲルが生成した。ゲル化試験の放冷後のサンプル管の写真を図15に示す。
また、実施例6で合成した式7で表される化合物は、純水以外に、pH4~9の塩酸又は水酸化ナトリウム水溶液、生理食塩水、エタノール及び水の混合液並びに1,3-プロパンジオール及び水の混合液に対してもゲル形成することができた。
サンプル管に、ヒドロゲル化剤(実施例6で合成した式10で表される化合物)及び純水500μLを加えて、試料を調製した。そして、120℃のホットプレート上で加熱溶解させた。その後、得られた溶液を室温で静置すると、ヒドロゲルが得られた。最小ゲル化濃度は0.3質量%であり、透明なヒドロゲルが生成した。ゲル化試験の放冷後のサンプル管の写真を図16に示す。
サンプル管に、ヒドロゲル化剤(実施例6で合成した式10で表される化合物)を加え、ここに純水を加えて、ヒドロゲル化剤の濃度がそれぞれ0.1質量%、0.3質量%、0.5質量%、0.75質量%及び1.0質量%の試料を調製した。そして、120℃のホットプレート上で加熱溶解させた。その後、得られた各溶液を室温で静置すると、ヒドロゲル化剤の濃度が0.1質量%のものは溶液に粘性がない状態(ゾル)であったが、それ以外のものについては、ヒドロゲルが得られた。そして、得られたゾル及びヒドロゲルの透過率を測定した。その結果を図17に示す。
図17に示すように、ヒドロゲル化剤の濃度が0.5質量%までは、ヒドロゲルは高い透明性を有するが、それよりも高い濃度のヒドロゲルでは、透過率は低下した。
実施例6で合成した式10で表される化合物をヒドロゲル化剤として、振動処理に対するゲル形成能の評価を行った。
振動処理によるゲル化試験は次のように行った。
サンプル管に、ヒドロゲル化剤及び純水を加えて、試料を調製した。そして、ボルテックスミキサーを用いて、サンプル管の下部に振動を約5分間(2500rpm)与えた。その後、室温で静置すると、ヒドロゲルが得られた(図18)。最小ゲル化濃度は0.5質量%であった。
また、実施例6で合成した式10で表される化合物は、純水以外に、pH3~10の塩酸又は水酸化ナトリウム水溶液、生理食塩水、リン酸緩衝液、エタノール及び水の混合液並びに1,3-プロパンジオール及び水の混合液に対しても、振動処理により、ゲル形成することができた。
サンプル管に、ヒドロゲル化剤(実施例6で合成した式10で表される化合物)5.0mgを量り取り、ここに純水500μLを加えて、ヒドロゲル化剤の濃度が1.0質量%の試料を調製した。そして、120℃のホットプレート上で加熱溶解させた。その後、得られた溶液を室温で静置すると、ヒドロゲルが得られた。そして、得られたヒドロゲルを走査型電子顕微鏡(SEM)にて観察した。走査型電子顕微鏡(SEM)像の写真を図19に示す。
図19に示すように、繊維状集合体が観察された。
<式13で表される化合物の合成>
サンプル管に、ヒドロゲル化剤(式13で表される化合物)を加え、ここに純水500μLを加えて、試料を調製した。そして、120℃のホットプレート上で加熱溶解させた。その後、得られた溶液を室温で静置すると、ヒドロゲルが得られた。最小ゲル化濃度は1.0質量%であり、半透明なヒドロゲルが生成した。ゲル化試験の放冷後のサンプル管の写真を図20に示す。
式13で表される化合物は、純水以外に、pH4~10の塩酸又は水酸化ナトリウム水溶液、生理食塩水並びにエタノール及び水の混合液に対してもゲル形成することができた。
サンプル管に、ヒドロゲル化剤(式16で表される化合物)及び純水を加えて、試料を調製した。そして、120℃のホットプレート上で加熱溶解させた。その後、得られた溶液を室温で静置すると、ゲル化せずに懸濁液が得られた。
一方、式16で表される化合物は、エタノール及び水の混合液並びに1,3-プロパンジオール/水の混合液に対して、ゲル形成することができた。
Claims (5)
- 下記式[1]で表される化合物からなるヒドロゲル化剤。
Sgは糖基を表し、
Aは2価の連結基を表し、
Rは、炭素原子数1乃至15の直鎖状若しくは分岐鎖状のアルキル基、炭素原子数3乃至15の環状のアルキル基、炭素原子数2乃至15の直鎖状若しくは分岐鎖状のアルケニル基、又は未置換の若しくは炭素原子数1乃至10のアルキル基、炭素原子数1乃至10のアルコキシ基、炭素原子数6乃至18のアリールオキシ基、ハロゲン原子、ニトロ基、フェニル基、炭素原子数2乃至10のアルキルカルボニル基及び炭素原子数7乃至18のアラルキル基からなる群から選択される少なくとも1つの置換基で置換された炭素原子数6乃至18のアリール基を表す。) - 前記糖基Sgが単糖又は二糖由来の構造からなる1価の基である、請求項1に記載のヒドロゲル化剤。
- 前記連結基Aが炭素原子数1乃至15の直鎖状アルキレン基又は炭素原子数6乃至15のアリーレン基であり、
前記Rが炭素原子数1乃至15の直鎖状のアルキル基、炭素原子数3乃至10の環状のアルキル基、又は未置換の若しくは炭素原子数1乃至10のアルキル基、ハロゲン原子及び炭素原子数7乃至18のアラルキル基からなる群から選択される少なくとも1つの置換基で置換されたフェニル基である、
請求項2に記載のヒドロゲル化剤。 - 請求項1乃至請求項3のいずれか1項に記載のヒドロゲル化剤と、水系溶媒よりなるヒドロゲル。
Priority Applications (5)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| JP2018545094A JP7133174B2 (ja) | 2016-10-14 | 2017-10-16 | 新規ヒドロゲル化剤 |
| CN201780061764.3A CN109790439A (zh) | 2016-10-14 | 2017-10-16 | 新的水凝胶化剂 |
| US16/341,939 US20190241598A1 (en) | 2016-10-14 | 2017-10-16 | Novel hydrogelator |
| EP17859395.0A EP3524656A4 (en) | 2016-10-14 | 2017-10-16 | NEW HYDROGEL |
| KR1020197009688A KR20190067167A (ko) | 2016-10-14 | 2017-10-16 | 신규 하이드로겔화제 |
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| JP2016-202988 | 2016-10-14 | ||
| JP2016202988 | 2016-10-14 |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| WO2018070546A1 true WO2018070546A1 (ja) | 2018-04-19 |
Family
ID=61905653
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/JP2017/037423 WO2018070546A1 (ja) | 2016-10-14 | 2017-10-16 | 新規ヒドロゲル化剤 |
Country Status (7)
| Country | Link |
|---|---|
| US (1) | US20190241598A1 (ja) |
| EP (1) | EP3524656A4 (ja) |
| JP (1) | JP7133174B2 (ja) |
| KR (1) | KR20190067167A (ja) |
| CN (1) | CN109790439A (ja) |
| TW (1) | TW201829710A (ja) |
| WO (1) | WO2018070546A1 (ja) |
Cited By (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US20200270392A1 (en) * | 2019-02-25 | 2020-08-27 | Tokyo Electron Limited | Composite for film formation and film forming method |
Families Citing this family (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN112142805B (zh) * | 2020-09-09 | 2022-03-25 | 江南大学 | 一种n-烷基葡萄糖胺小分子醇凝胶及其制备方法和应用 |
Citations (4)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JP2002053840A (ja) * | 2000-08-07 | 2002-02-19 | Japan Science & Technology Corp | コレステロール部位と糖部位とを有する化合物から成る有機溶媒ゲル化剤 |
| JP2004262809A (ja) * | 2003-02-28 | 2004-09-24 | Noguchi Inst | ジアミド型ゲル化剤 |
| WO2012121394A1 (ja) | 2011-03-10 | 2012-09-13 | 国立大学法人静岡大学 | 置換芳香族化合物、ヒドロゲル化剤、ヒドロゲル、及び水系試料のゲル化方法 |
| JP2012180455A (ja) * | 2011-03-01 | 2012-09-20 | National Institute Of Advanced Industrial Science & Technology | 糖由来の低分子ヒドロゲル化剤、および前記ヒドロゲル化剤を有効成分とするヒドロゲル |
-
2017
- 2017-10-16 KR KR1020197009688A patent/KR20190067167A/ko unknown
- 2017-10-16 JP JP2018545094A patent/JP7133174B2/ja active Active
- 2017-10-16 CN CN201780061764.3A patent/CN109790439A/zh active Pending
- 2017-10-16 WO PCT/JP2017/037423 patent/WO2018070546A1/ja unknown
- 2017-10-16 US US16/341,939 patent/US20190241598A1/en not_active Abandoned
- 2017-10-16 TW TW106135366A patent/TW201829710A/zh unknown
- 2017-10-16 EP EP17859395.0A patent/EP3524656A4/en not_active Withdrawn
Patent Citations (4)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JP2002053840A (ja) * | 2000-08-07 | 2002-02-19 | Japan Science & Technology Corp | コレステロール部位と糖部位とを有する化合物から成る有機溶媒ゲル化剤 |
| JP2004262809A (ja) * | 2003-02-28 | 2004-09-24 | Noguchi Inst | ジアミド型ゲル化剤 |
| JP2012180455A (ja) * | 2011-03-01 | 2012-09-20 | National Institute Of Advanced Industrial Science & Technology | 糖由来の低分子ヒドロゲル化剤、および前記ヒドロゲル化剤を有効成分とするヒドロゲル |
| WO2012121394A1 (ja) | 2011-03-10 | 2012-09-13 | 国立大学法人静岡大学 | 置換芳香族化合物、ヒドロゲル化剤、ヒドロゲル、及び水系試料のゲル化方法 |
Non-Patent Citations (4)
| Title |
|---|
| AKAMA, SHUTO; YAMANAKA, MASAMICHI: "2B6-05 Tankotei de Gosei Kano na Teibi.nshl Hydrociel-ka-zai = Short-step synthesizable low molecular weight hydrogelator", 97TH ANNUAL MEETING OF THE CHEMICAL SOCIETY OF JAPAN IN SPRING KOEN YOKOSHU. , vol. 97, 3 March 2017 (2017-03-03), XP009513286 * |
| DATABASE Registry 10 November 2017 (2017-11-10), Database accession no. RN 109819-12-7 * |
| MATHISELVAM, MANOHARAN, ET AL.: "Synthesis and characterization of thiourea-and urea- linked glycolipids as low-molecular-weight hydrogelators", RSC ADVANCES , vol. 3, no. 34, 2013, pages 14528 - 14542, XP055588365 * |
| See also references of EP3524656A4 |
Cited By (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US20200270392A1 (en) * | 2019-02-25 | 2020-08-27 | Tokyo Electron Limited | Composite for film formation and film forming method |
Also Published As
| Publication number | Publication date |
|---|---|
| TW201829710A (zh) | 2018-08-16 |
| EP3524656A4 (en) | 2019-10-16 |
| KR20190067167A (ko) | 2019-06-14 |
| EP3524656A1 (en) | 2019-08-14 |
| JP7133174B2 (ja) | 2022-09-08 |
| CN109790439A (zh) | 2019-05-21 |
| JPWO2018070546A1 (ja) | 2019-09-05 |
| US20190241598A1 (en) | 2019-08-08 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| Wu et al. | Nitrogen and sulfur co-doped carbon quantum dots for highly selective and sensitive fluorescent detection of Fe (III) ions and L-cysteine | |
| Kamkaew et al. | Aza-BODIPY dyes with enhanced hydrophilicity | |
| Zhuang et al. | Ratiometric fluorescence probe for monitoring hydroxyl radical in live cells based on gold nanoclusters | |
| WO2018070546A1 (ja) | 新規ヒドロゲル化剤 | |
| Bhangu et al. | Sono-transformation of tannic acid into biofunctional ellagic acid micro/nanocrystals with distinct morphologies | |
| WO2002096457A3 (en) | Stable liquid formulations of antibodies | |
| Soldà et al. | C 60@ lysozyme: a new photosensitizing agent for photodynamic therapy | |
| CN104984349B (zh) | 一种负载表阿霉素的石墨烯量子点载药体系及其制备方法 | |
| Ge et al. | A thiol–thiosulfonate reaction providing a novel strategy for turn-on thiol sensing | |
| Hirohara et al. | Sugar and heavy atom effects of glycoconjugated chlorin palladium complex on photocytotoxicity | |
| Zheng et al. | Water-soluble two-photon absorbing nitrosyl complex for light-activated therapy through nitric oxide release | |
| US20180319957A1 (en) | Preparation of modified cellulose and its derivatives | |
| JP5885256B2 (ja) | トリアジン環含有重合体 | |
| Alnajjar et al. | Rational design of boron-dipyrromethene (BODIPY) reporter dyes for cucurbit [7] uril | |
| Ershov et al. | Condensation products of D-ribose with thiol-containing hydrazides and gold glyconanoparticles thereof | |
| Zhang et al. | A new xanthene-based platform for developing NIR fluorogenic probes for in vivo bioimaging | |
| Ho-Yin et al. | One-pot synthesis to prepare lignin/photoacid nanohybrids for multifunctional biosensors and photo-triggered singlet oxygen generation | |
| Fredy et al. | Diverted Natural Lossen-type Rearrangement for Bioconjugation through in Situ Myrosinase-Triggered Isothiocyanate Synthesis | |
| JP7066117B2 (ja) | オイルゲル化剤 | |
| Moiseeva et al. | Tetraiodophthalocyanines: Simple and convenient synthetic protocol and structural modification via Sonogashira cross-coupling reaction | |
| Seggio et al. | Visible light-activatable cyclodextrin-conjugates for the efficient delivery of nitric oxide with fluorescent reporter and their inclusion complexes with betaxolol | |
| Chen et al. | Synthesis and biological application of a water-soluble fluorescent probe based on coumarin derivatives for detection of ferric ion | |
| CN112142805B (zh) | 一种n-烷基葡萄糖胺小分子醇凝胶及其制备方法和应用 | |
| Jiao et al. | Meso pyridinium BODIPY-based long wavelength infrared mitochondria-targeting fluorescent probe with high photostability | |
| CN110372587A (zh) | 水杨酸偶氮8-羟基喹啉及其制备方法和应用 |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application |
Ref document number: 17859395 Country of ref document: EP Kind code of ref document: A1 |
|
| ENP | Entry into the national phase |
Ref document number: 2018545094 Country of ref document: JP Kind code of ref document: A |
|
| ENP | Entry into the national phase |
Ref document number: 20197009688 Country of ref document: KR Kind code of ref document: A |
|
| NENP | Non-entry into the national phase |
Ref country code: DE |
|
| ENP | Entry into the national phase |
Ref document number: 2017859395 Country of ref document: EP Effective date: 20190508 |