상기 목적을 달성하기 위하여 본 발명은, 완전한 이뮤노글로불린(immunoglobulin) 형태의 항체를 살아있는 세포의 세포막을 세포내재화(endocytosis)를 통해 능동적으로 침투하고 세포질에 위치시키는 방법으로서, 상기 항체는 세포질 침투능을 가지는 경쇄가변영역 (VL)을 포함하는 것인 방법을 제공한다. In order to achieve the above object, the present invention provides a method for actively infiltrating a cell membrane of a living cell through endocytosis and placing it in the cytoplasm of an antibody of a complete immunoglobulin form, wherein the antibody has a cytoplasmic infiltration ability. The branch provides a light chain variable region (VL).
이하 본 발명을 상세히 설명한다.Hereinafter, the present invention will be described in detail.
본 발명의 상기 방법은 완전한 이뮤노글로불린(immunoglobulin) 형태의 항체를 세포내재화(endocytosis)를 통해서 능동적으로 살아있는 세포의 세포막을 침투 및 엔도좀 탈출(endosome escape) 기작을 유도하여 세포질에 위치하도록 유도할 수 있는 경쇄가변영역 (VL)에 의하여, 완전한 이뮤노글로불린(immunoglobulin) 형태의 항체가 살아있는 세포의 세포막을 침투하여 세포질에 위치할 수 있다. The method of the present invention is to induce the complete immunoglobulin (antimunoglobulin) type of antibody to be located in the cytoplasm by endocytosis to actively penetrate the cell membrane of living cells and induce endosomal escape mechanism By means of the variable light chain variable region (VL), antibodies in the form of complete immunoglobulin (immunoglobulin) can penetrate the cell membranes of living cells and be located in the cytoplasm.
즉, 본 발명의 항체는 살아있는 세포의 세포 내부로 침투할 수 있는 능력 및 세포질에 위치할 수 있는 능력을 모두 발휘하는 완전한 이뮤노글로불린 형태의 항체이며, 이의 일부 절편에 해당하는 항체 경쇄 가변 영역은 그 자체자체로서 세포 내부로 침투하고 세포질에 위치하는 능력을 발휘한다 That is, the antibody of the present invention is an antibody of the complete immunoglobulin type that exhibits both the ability to penetrate into the cells of living cells and to be located in the cytoplasm, and the antibody light chain variable region corresponding to some fragments thereof. As itself, it exerts its ability to penetrate inside the cell and locate in the cytoplasm.
도 1은 본 발명의 항체 또는 항체 경쇄 가변 영역의 세포에서의 활성을 도식화한 것이다. Figure 1 shows the activity in the cells of the antibody or antibody light chain variable region of the present invention.
상기 항체는 키메릭, 인간, 또는 인간화된 항체일 수 있다.The antibody may be a chimeric, human, or humanized antibody.
또한, 상기 항체는 IgG, IgM, IgA, IgD 또는 IgE일 수 있으며, 예를 들면, IgG1, IgG2, IgG3, IgG4, IgM, IgE, IgA1, IgA5, 또는 IgD 타입일 수 있으며, 가장 바람직하게는 IgG 타입의 단일클론항체일 수 있다.In addition, the antibody may be IgG, IgM, IgA, IgD or IgE, for example IgG1, IgG2, IgG3, IgG4, IgM, IgE, IgA1, IgA5, or IgD type, most preferably IgG Type of monoclonal antibody.
완전한 이뮤노글로불린 형태의 항체는 2개의 전장(full length) 경쇄 및 2개의 전장 중쇄를 가지는 구조이며, 각각의 경쇄는 중쇄와 이황화 결합(disulfide bond, SS-bond) 항체의 불변 영역은 중쇄 불변 영역과 경쇄 불변 영역으로 나뉘어지며, 중쇄 불변 영역은 감마(γ), 뮤(μ), 알파(α), 델타(δ) 및 엡실론(ε) 타입을 가지고, 서브클래스로 감마1(γ1), 감마2(γ2), 감마3(γ3), 감마4(γ4), 알파1(α1) 및 알파2(α2)를 가진다. 경쇄의 불변 영역은 카파(κ) 및 람다(λ) 타입을 가진다. A full immunoglobulin type antibody has a structure having two full length light chains and two full length heavy chains, each light chain having a heavy chain constant region of a heavy chain and disulfide bond (SS-bond) antibody. It is divided into light chain constant region and heavy chain constant region has gamma (γ), mu (μ), alpha (α), delta (δ) and epsilon (ε) type, and subclasses gamma 1 (γ1), gamma 2 (γ 2), gamma 3 (γ 3), gamma 4 (γ 4), alpha 1 (α 1), and alpha 2 (α 2). The constant regions of the light chains have kappa (κ) and lambda (λ) types.
"중쇄(heavy chain)"는 항원에 특이성을 부여하기 위해 충분한 가변 영역 서열을 갖는 아미노산 서열을 포함하는 가변 영역 도메인 VH 및 3 개의 불변 영역 도메인 CH1, CH2 및 CH3를 포함하는 전장 중쇄 및 이의 단편을 모두 포함하는 의미로 해석된다. 또한, 용어 "경쇄(light chain)"는 항원에 특이성을 부여하기 위한 충분한 가변영역 서열을 갖는 아미노산 서열을 포함하는 가변 영역 도메인 VL 및 불변 영역 도메인 CL을 포함하는 전장 경쇄 및 이의 단편을 모두 포함하는 의미로 해석된다. "Heavy chain" refers to a full-length heavy chain and fragment thereof comprising a variable region domain VH comprising an amino acid sequence having sufficient variable region sequence to confer specificity to an antigen and three constant region domains CH1, CH2 and CH3 Are interpreted to include all. In addition, the term “light chain” includes both the full-length light chain and fragments thereof comprising the variable region domain VL and the constant region domain CL comprising an amino acid sequence having sufficient variable region sequence to confer specificity to the antigen. It is interpreted as meaning.
상기 세포질 침투능은 세포 내재화(endocytosis)를 통해서 능동적으로 살아있는 세포에 침투한 후 엔도좀 탈출(endosome escape)에 의하여 세포질에 위치하는 것일 수 있다. The cytoplasmic penetrating ability may be located in the cytoplasm by endosome escape after actively infiltrating living cells through endocytosis.
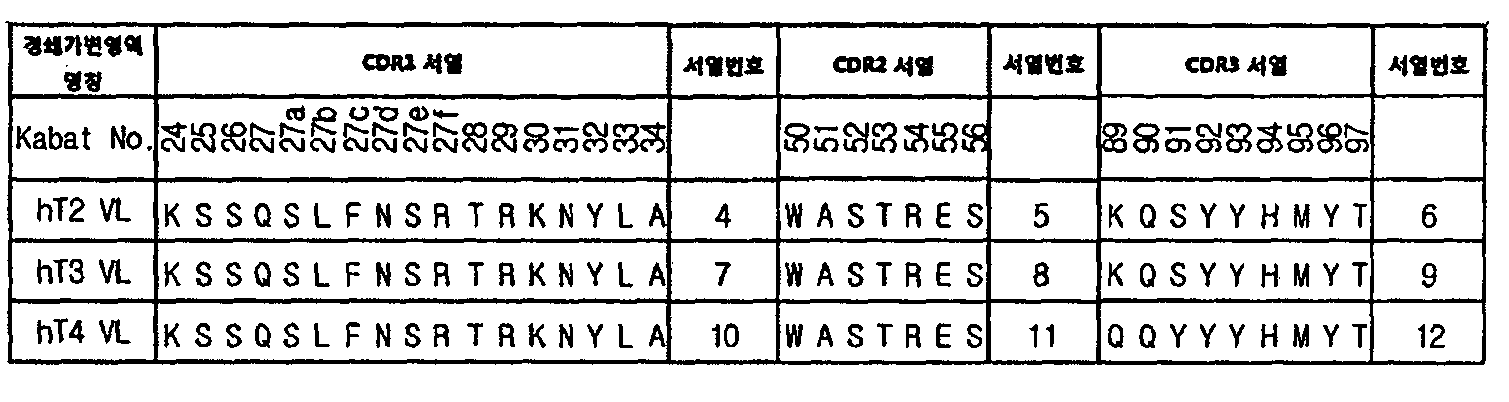
본 발명의 일 양상에서, 세포질 침투능을 가지는 경쇄 가변영역은 서열번호 4, 7 및 10으로 이루어진 군으로부터 선택된 아미노산 서열로 이루어진 CDR1 또는 이와 상동성이 90% 이상인 서열; 및 In one aspect of the present invention, the light chain variable region having cytoplasmic penetration ability is CDR1 consisting of an amino acid sequence selected from the group consisting of SEQ ID NO: 4, 7 and 10 or a sequence having at least 90% homology thereto; And
서열번호 6, 9, 및 12로 이루어진 군으로부터 선택된 아미노산 서열로 이루어진 CDR3 또는 이와 상동성이 90% 이상인 서열을 포함할 수 있다. CDR3 consisting of an amino acid sequence selected from the group consisting of SEQ ID NOs: 6, 9, and 12 or a sequence having at least 90% homology thereto.
상기 CDR1, CDR2, 및 CDR3의 서열정보는 하기와 같다. Sequence information of the CDR1, CDR2, and CDR3 is as follows.
[규칙 제91조에 의한 정정 11.08.2015]
[Correction under Rule 91 11.08.2015]
상기 경쇄가변영역은 더욱 바람직하게는 서열번호 5, 8, 및 11로 이루어진 군으로부터 선택된 아미노산 서열로 이루어진 CDR2 또는 이와 상동성이 90% 이상인 서열을 더 포함할 수 있다.More preferably, the light chain variable region may further include a CDR2 consisting of an amino acid sequence selected from the group consisting of SEQ ID NOs: 5, 8, and 11 or a sequence having at least 90% homology thereto.
본 발명의 일 구체예에서 상기 경쇄가변영역은 서열번호 4의 CDR1, 서열번호 5의 CDR2, 및 서열번호 6의 CDR3를 포함한 것일 수 있다.In one embodiment of the present invention, the light chain variable region may include CDR1 of SEQ ID NO: 4, CDR2 of SEQ ID NO: 5, and CDR3 of SEQ ID NO: 6.
또한, 본 발명의 다른 구체예에서 상기 경쇄 가변영역은 서열번호 7의 CDR1, 서열번호 8의 CDR2, 및 서열번호 9의 CDR3를 포함한 것일 수 있다.In another embodiment of the present invention, the light chain variable region may include CDR1 of SEQ ID NO. 7, CDR2 of SEQ ID NO. 8, and CDR3 of SEQ ID NO.
또한, 본 발명의 다른 양상에서 상기 경쇄 가변영역은 서열번호 10의 CDR1, 서열번호 11의 CDR2, 및 서열번호 12의 CDR3를 포함한 것일 수 있다.In another aspect of the present invention, the light chain variable region may include CDR1 of SEQ ID NO: 10, CDR2 of SEQ ID NO: 11, and CDR3 of SEQ ID NO: 12.
본 발명의 일 양상에서, 상기 경쇄가변영역은 경쇄 가변 영역의 N 말단으로부터 2번째 및 4번째 아미노산이 각각 류신 (leucine, L) 및 메티오닌 (Methionine, M)으로 치환된 것일 수 있다.In one aspect of the present invention, the light chain variable region may be one in which the second and fourth amino acids from the N terminus of the light chain variable region are substituted with leucine (L) and methionine (M), respectively.
이는 FR(framework)에 위치하고 있는 CDR 구조결정부위(Vernier zone)에 포함되는 잔기 중 세포질 침투능이 보존된 CDR 구조를 얻기 위해 중요한 2번째, 4번째 잔기를 치환시킨 것이다.This is to replace the important second and fourth residues in order to obtain the CDR structure of the cytoplasmic permeability among the residues included in the CDR (Vernier zone) located in the FR (framework).
또한, 본 발명의 일 양상에서, 상기 경쇄가변영역은 경쇄 가변 영역의 N 말단으로부터 9, 10, 13, 17, 19, 21, 22, 42, 45, 58, 60, 79, 및 85번째 아미노산이 각각 In addition, in one aspect of the present invention, the light chain variable region includes amino acids 9, 10, 13, 17, 19, 21, 22, 42, 45, 58, 60, 79, and 85 from the N terminus of the light chain variable region. each
세린 (serine, S), 세린 (serine, S), 알라닌 (Alanine, A), 발린 (Valine, V), 아스파트 산 (aspartic acid, D), 발린 (Valine, V), 이소류신 (Isoleucine, I), 트리오닌 (Threonine, T), 라이신 (Lysine, K), 라이신 (Lysine, K), 발린 (Valine, V), 세린 (serine, S), 글루타민 (Glutamine, Q) 및 트리오닌 (Threonine, T)으로 치환된 것일 수 있다.Serine (S), Serine (S), Alanine (A), Valine (V), Aspartic acid (D), Valine (V), Isoleucine (Isoleucine, I ), Trionine (T), lysine (Lysine, K), lysine (Lysine, K), valine (V), serine (S), glutamine (Q) and trionine (Threonine, It may be substituted with T).
이는 FDA 승인 후 시장에 판매되고 있는 인간화 치료용 항체 중 안정성이 매우 높으며 VH3소그룹의 중쇄가변영역과 Vκ1소그룹의 경쇄가변영역으로 이루어진 Trastuzumab(Herceptin)의 경쇄가변영역과의 서열 분석을 통해 FR(Framework) 에 위치하고 있는 총 14개의 잔기에 차이가 있음을 확인하여 이를 치환한 것이다. It is very stable among humanized therapeutic antibodies on the market after FDA approval, and FR (Framework) is analyzed through sequence analysis of the light chain variable region of Trastuzumab (Herceptin) consisting of the heavy chain variable region of VH3 subgroup and the light chain variable region of Vκ1 subgroup. A total of 14 residues located at) were found to have been replaced.
또한, 본 발명의 다른 양상에서, 상기 경쇄 가변 영역은 경쇄 가변 영역의 N 말단으로부터 89 번째 및 91번째 아미노산이 각각 글루타민 (Glutamine, Q) 및 타이로신 (Tyrosine, Y)으로 치환된 것일 수 있다.In another aspect of the present invention, the light chain variable region may be one in which the 89th and 91th amino acids are substituted with glutamine (Q) and tyrosine (Yyrosine, Y) from the N terminus of the light chain variable region, respectively.
이는 인간 항체 가변영역 간의 인터페이스(VH-VL interface) 분석을 통해 기존 세포질 침투 경쇄 가변 영역의 마우스 유래 CDR3에 위치한 2개의 잔기가 차이가 있음을 확인하여 이를 치환한 것이다. This was confirmed by substituting the two residues located in the mouse-derived CDR3 of the existing cytoplasmic penetration light chain variable region through analysis of the interface between the human antibody variable region (VH-VL interface).
본 발명의 바람직한 양상에서, 상기 경쇄 가변영역은 서열번호 1, 2, 및 3으로 이루어진 군으로부터 선택된 아미노산으로 이루어진 것일 수 있다.In a preferred aspect of the present invention, the light chain variable region may be composed of amino acids selected from the group consisting of SEQ ID NO: 1, 2, and 3.
상기 서열번호의 서열정보는 하기와 같다. Sequence information of the sequence number is as follows.
세포질 침투 인간화 경쇄가변영역(VL) 단일도메인 명칭 및 서열Cytoplasmic Penetration Humanized Light Chain Variable Region (VL) Monodomain Name and Sequence
단, 본 명세서에서 제공하는 서열번호에 명시된 모든 잔기 번호는 Kabat 번호를 사용하였다 (Kabat EA et al., 1991).However, all residue numbers specified in SEQ ID NOs provided herein were used Kabat number (Kabat EA et al., 1991).
본 발명의 일 양상에서, 상기 항체가 막을 투과하여, 세포질에 위치하는 세포는 살아있는 동물 세포일 수 있다. 즉 상기 항체는 상기 항체는 살아있는 동물 세포에 능동적으로 침투하는 것일 수 있다. In one aspect of the invention, the antibody penetrates the membrane, so that the cell located in the cytoplasm may be a live animal cell. That is, the antibody may be one in which the antibody actively penetrates live animal cells.
또한, 본 발명의 일 양상에서, 상기 항체는 세포질뿐 만 아니라 세포질 내에 존재하는 다양한 세포소기관 및 세포내 존재하는 분자들을 표적할 수 있으며, 그 종류를 제한하지 않으나, 예를 들면 세포질, 핵, 미토콘드리아, 소포체, 세포내 고분자 (organelle macromolecule)를 표적할 수 있다.In addition, in one aspect of the present invention, the antibody may target not only the cytoplasm but also various organelles and molecules present in the cell, but not limited to, for example, cytoplasm, nucleus, mitochondria , Endoplasmic reticulum, and intracellular macromolecules.
또한, 본 발명의 일 양상에서, 상기 세포내 고분자는 단백질, 지질, DNA 또는 RNA일 수 있다. 더욱 구체적으로, 상기 단백질은 세포 성장, 세포증식, 세포주기, DNA 수리, DNA 보전, 전사, 복제, 번역 또는 세포내 이동의 제어와 관련된 것 일 수 있으며, 상기 단백질은 인산기, 카르복실산기, 메틸기, 설페이트기, 지질, 수산기, 또는 아미드기로 변형 또는 활성화되거나, 돌연변이된 것 일 수 있다.In addition, in one aspect of the present invention, the intracellular polymer may be a protein, lipid, DNA or RNA. More specifically, the protein may be related to control of cell growth, cell proliferation, cell cycle, DNA repair, DNA preservation, transcription, replication, translation or intracellular migration, wherein the protein is a phosphate group, a carboxylic acid group, a methyl group And may be modified, activated, or mutated by sulfate, lipid, hydroxyl, or amide groups.
본 발명의 가장 바람직한 구체예에서, 상기 항체는 세포질 내 활성화된 RAS를 표적하여 특이적으로 결합하는 것일 수 있다. 상기 활성화된 RAS는 GTP가 결합된 종양관련인자로서, 상기 RAS는 돌연변이 형태일 수 있다. 상기 RAS의 돌연변이는 질환과 관계되는 다양한 형태로 그 종류에 제한이 없으나, 예를 들면, KRas, HRas, NRas 의 12번 Glycine, 13번 Glycine, 61번 glutamine 잔기들의 치환된 돌연변이들 일 수 있다. In the most preferred embodiment of the invention, the antibody may be to specifically bind to target activated RAS in the cytoplasm. The activated RAS is a tumor-associated factor to which GTP is bound, and the RAS may be in a mutant form. The RAS mutations are various forms related to the disease, but there is no limitation in the type thereof. For example, the RAS mutations may be substituted mutations of Glycine No. 12, Glycine No. 13, glutamine No. 61 of KRas, HRas, and NRas.
본 발명의 일 양상에서, 상기 세포질 내 활성화된 RAS와의 결합력은 항체의 중쇄가변영역 (VH)에 의한 것일 수 있다. In one aspect of the present invention, the binding force to the activated RAS in the cytoplasm may be due to the heavy chain variable region (VH) of the antibody.
본 발명의 일 양상에서, 상기 중쇄가변영역은 In one aspect of the invention, the heavy chain variable region is
서열번호 14의 CDR1 또는 이와 상동성이 90% 이상인 아미노산 서열;The CDR1 of SEQ ID NO: 14 or an amino acid sequence having at least 90% homology with it;
서열번호 15의 CDR2 또는 이와 상동성이 90% 이상인 아미노산서열; 및CDR2 of SEQ ID NO: 15 or an amino acid sequence having a homology of 90% or more; And
서열번호 16의 CDR3 또는 이와 상동성이 90% 이상인 아미노산 서열;을 포함할 수 있다. CDR3 of SEQ ID NO: 16 or an amino acid sequence having 90% or more homology thereto;
상기 서열번호의 서열정보는 하기와 같다. Sequence information of the sequence number is as follows.
본 발명의 더욱 바람직한 구체예에서, 중쇄가변영역은 서열번호 13의 아미노산으로 이루어질 수 있다.In a more preferred embodiment of the invention, the heavy chain variable region may consist of the amino acid of SEQ ID NO: 13.
상기 서열번호의 서열정보는 하기와 같다.Sequence information of the sequence number is as follows.
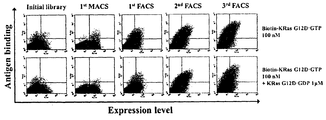
상기 RAS에 특이적으로 결합 및 저해하는 중쇄가변영역은 하기와 같은 방법에 의하여 스크리닝 하였다. The heavy chain variable region that specifically binds to and inhibits the RAS was screened by the following method.
본 발명의 일 구체예에서는 구축되어진 인간 중쇄가변영역(VH)과 중쇄불변영역(CH1)이 융합되어 있는 상태로 CDR1, CDR2, CDR3 지역에 총 18개 잔기에 대해서 인위적인 돌연변이가 유도된 라이브러리를 이용하여 선별하였다.In one embodiment of the present invention, a library in which artificial mutations are induced for a total of 18 residues in CDR1, CDR2, and CDR3 regions while the constructed human heavy chain variable region (VH) and heavy chain constant region (CH1) are fused. By selection.
또한 본 발명의 일 구체예에서는 상기 인간 중쇄가변영역(VH)과 중쇄불변영역(CH1)이 융합되어 있는 라이브러리를 이용하여 세포질 침투 인간화 경쇄가변영역(VL)과 결합된 상태에서도 활성화된(GTP가 결합된) RAS에 대해서 특이적으로 결합이 가능한 중쇄가변영역을 선별하였다. In addition, in one embodiment of the present invention using a library in which the human heavy chain variable region (VH) and the heavy chain constant region (CH1) is fused to the cytoplasmic penetration humanized light chain variable region (VL) is activated even in the state coupled to (GTP is The heavy chain variable region capable of binding specifically to RAS) was selected.
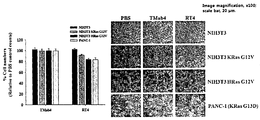
본 발명의 일 구체예에서는 표적분자로서 활성화된(GTP가 결합된) RAS 돌연변이체인 KRas G12D을 사용하였다. 본 발명의 일 구체예에 있어서 발암관련 RAS 돌연변이는 주로 12번, 13번, 61번 잔기에서 발생하며, 12번, 13번 잔기는 RAS 단백질의 P-loop에 위치하고 있으며, RAS 단백질과 결합해 있는 GTP를 가수분해하여 비활성화된 형태로 단백질 구조 변화를 유도하는 GAP(GTPase-activating protein)의 결합에 영향을 준다. 또한 61번 잔기는 GAP의 가수분해 활성 부위와 결합하여 GTP가수분해를 막는 역할을 하므로 다양한 발암관련 RAS 돌연변이는 RAS G12D 돌연변이와 신호전달에 관련된 지역(Switch I, Switch II)이 같으므로 KRas G12D 돌연변이에 한정되지 아니한다.In one embodiment of the present invention, KRas G12D, which is an activated (GTP coupled) RAS mutant, was used as a target molecule. In one embodiment of the present invention, carcinogenesis-related RAS mutations occur mainly at residues 12, 13 and 61, and residues 12 and 13 are located in the P-loop of the RAS protein and are bound to the RAS protein. Hydrolysis of GTP affects the binding of GAP (GTPase-activating protein), which induces protein structural changes in an inactive form. In addition, residue 61 binds to the hydrolytic activity of GAP and prevents GTP hydrolysis. Therefore, various carcinogenic RAS mutations have the same region as the RAS G12D mutation and signal transduction (Switch I, Switch II). It is not limited to.
또한, 상기 일 구체예에 있어서 NRas, HRas는 KRas와 G도메인이라고 불리는 촉매도메인(catalytic domain) 1번부터 165번 잔기까지의 유사도가 85% 이상이며, 이중 하위 신호물질과 결합하는 부위 Switch I(32번 내지 38번), Switch II(59번 내지 67번) 도메인은 100% 일치한다. 다만, 165번부터 189번까지의 C-말단 초가변형 부위는 15 %의 유사도를 지니고 있으나 구조적으로 하위 신호전달에 영향을 주지 않으므로 표적분자로 사용한 활성화된 KRas G12D에 한정되지 아니한다.In addition, in one embodiment, NRas and HRas have 85% or more similarity between residues of the catalytic domain 1 to 165 called KRas and G domain, and the site Switch I (which binds to the lower signaling material) 32-38), Switch II (59-67) domains are 100% identical. However, the C-terminal hypervariable regions 165 to 189 have a similarity of 15% but are not limited to activated KRas G12D used as target molecules because they do not structurally affect downstream signaling.
본 발명의 일 구체예에 있어서 효모세포표면발현 시스템을 이용하여 중쇄가변영역(VH)과 중쇄불변영역(CH1)이 발현된 상태에서 활성화된(GTP가 결합된) RAS에 대해서 초기 선별 이후 세포질 침투 경쇄가변영역(VL)과 경쇄불변영역(CL)을 포함하는 경쇄를 발현 및 분비하는 효모와 효모접합을 통해 이 결합된 Fab형태로 선별하였다. In one embodiment of the present invention using the yeast cell surface expression system cytoplasmic infiltration after the initial screening for RAS activated (GTP coupled) in the state in which the heavy chain variable region (VH) and heavy chain constant region (CH1) is expressed A light chain comprising a light chain variable region (VL) and a light chain constant region (CL) was selected in the form of the bound Fab through yeast and yeast conjugation expressing and secreting a light chain.
또한, 본 발명의 일 양상은 완전한 이뮤노글로불린(immunoglobulin) 형태의 항체가 세포막을 투과하여 세포질에 위치하는 것을 유도하는 경쇄가변영역 (VL)을 제공한다.In addition, one aspect of the present invention provides a light chain variable region (VL) that induces a complete immunoglobulin form of antibody to penetrate the cell membrane and located in the cytoplasm.
본 발명의 일 구체예에서 상기 경쇄가변영역은 서열번호 4, 7 및 10으로 이루어진 군으로부터 선택된 아미노산 서열로 이루어진 CDR1 또는 이와 상동성이 90% 이상인 서열; 및 In one embodiment of the present invention, the light chain variable region is CDR1 consisting of an amino acid sequence selected from the group consisting of SEQ ID NOs: 4, 7, and 10 or a sequence having a homology of 90% or more; And
서열번호 6, 9, 및 12로 이루어진 군으로부터 선택된 아미노산 서열로 이루어진 CDR3 또는 이와 상동성이 90% 이상인 서열을 포함하는 것일 수 있다.CDR3 consisting of an amino acid sequence selected from the group consisting of SEQ ID NOs: 6, 9, and 12, or may include a sequence having at least 90% homology thereto.
또한, 일 구체예에서, 상기 경쇄가변영역 (VL)은 경쇄 가변 영역의 N 말단으로부터 2번째 및 4번째 아미노산이 각각 류신 (leucine, L) 및 메티오닌 (Methionine, M)으로 치환된 것 일 수 있다. In addition, in one embodiment, the light chain variable region (VL) may be a second and fourth amino acid is substituted with leucine (L) and methionine (Methionine, M) from the N terminal of the light chain variable region, respectively. .
또한, 본 발명의 다른 구체예에서 상기 경쇄가변영역은 경쇄 가변 영역의 N 말단으로부터 9, 10, 13, 17, 19, 21, 22, 42, 45, 58, 60, 79, 및 85번째 아미노산이 각각 In another embodiment of the present invention, the light chain variable region includes amino acids 9, 10, 13, 17, 19, 21, 22, 42, 45, 58, 60, 79, and 85 from the N terminus of the light chain variable region. each
세린 (serine, S), 세린 (serine, S), 알라닌 (Alanine, A), 발린 (Valine, V), 아스파트 산 (aspartic acid, D), 발린 (Valine, V), 이소류신 (Isoleucine, I), 트리오닌 (Threonine, T), 라이신 (Lysine, K), 라이신 (Lysine, K), 발린 (Valine, V), 세린 (serine, S), 글루타민 (Glutamine, Q) 및 트리오닌 (Threonine, T)으로 치환된 것일 수 있다.Serine (S), Serine (S), Alanine (A), Valine (V), Aspartic acid (D), Valine (V), Isoleucine (Isoleucine, I ), Trionine (T), lysine (Lysine, K), lysine (Lysine, K), valine (V), serine (S), glutamine (Q) and trionine (Threonine, It may be substituted with T).
본 발명의 다른 구체예에서 상기 경쇄가변영역은 경쇄 가변 영역의 N 말단으로부터 89 번째 및 91번째 아미노산이 각각 글루타민 (Glutamine, Q) 및 타이로신 (Tyrosine, Y)으로 치환된 것 일 수 있다.In another embodiment of the present invention, the light chain variable region may be one in which the 89th and 91th amino acids are substituted with glutamine (Glutamine, Q) and tyrosine (Y) from the N terminus of the light chain variable region, respectively.
또한, 본 발명의 바람직한 구체예에서, 상기 경쇄 가변영역은 서열번호 1, 2, 및 3으로 이루어진 군으로부터 선택된 아미노산 서열로 이루어진 것일 수 있다. In addition, in a preferred embodiment of the present invention, the light chain variable region may be composed of an amino acid sequence selected from the group consisting of SEQ ID NO: 1, 2, and 3.
본 발명에 따른 경쇄 가변영역의 세포 침투능은 세포 내재화(endocytosis)를 통해 침투한 후 엔도좀 탈출에 의한 것 일 수 있다.The cell penetration capacity of the light chain variable region according to the present invention may be due to endosomal escape after infiltrating through endocytosis.
또한, 본 발명의 일 양상은 상기 경쇄가변영역을 포함하는 항체를 제공한다. In addition, an aspect of the present invention provides an antibody comprising the light chain variable region.
본 발명의 구체예에서, 상기 항체는 세포막을 투과하여 세포질에 위치하는 것일 수 있으며, 상기 항체는 키메릭, 인간, 또는 인간화된 항체일 수 있다. 상기 항체는 IgG, IgM, IgA, IgD 및 IgE로 이루어진 군으로부터 선택된 것일 수 있다. 또한, 상기 항체는 세포질, 핵, 미토콘드리아, 소포체, 세포내 고분자 (organelle macromolecule)를 표적하는 것일 수 있으며, 상기 세포내 고분자는 단백질, 지질, DNA 또는 RNA일 수 있다. 상기 단백질은 세포 성장, 세포증식, 세포주기, DNA 수리, DNA 보전, 전사, 복제, 번역 또는 세포내 이동의 제어와 관련된 것 일 수 있다. 본 발명의 바람직한 구체예에서, 상기 항체는 세포질 내 활성화된 RAS에 특이적으로 결합하는 것일 수 있으며, 상기 세포질 내의 활성화된 RAS에 특이적으로 결합하는 중쇄 가변영역 (VH)에 의한 것일 수 있다. 상기 활성화된 RAS는 돌연변이된 형태일 수 있다.In an embodiment of the present invention, the antibody may be located in the cytoplasm through the cell membrane, and the antibody may be a chimeric, human, or humanized antibody. The antibody may be selected from the group consisting of IgG, IgM, IgA, IgD and IgE. In addition, the antibody may be to target the cytoplasm, nucleus, mitochondria, endoplasmic reticulum, intracellular macromolecule (organelle macromolecule), the intracellular polymer may be protein, lipid, DNA or RNA. The protein may be related to control of cell growth, cell proliferation, cell cycle, DNA repair, DNA preservation, transcription, replication, translation or intracellular migration. In a preferred embodiment of the present invention, the antibody may be specifically binding to activated RAS in the cytoplasm, it may be by a heavy chain variable region (VH) that specifically binds to the activated RAS in the cytoplasm. The activated RAS may be in mutated form.
또한, 본 발명의 구체예에 따른 중쇄가변영역은 In addition, the heavy chain variable region according to an embodiment of the present invention
서열번호 14의 CDR1 또는 이와 상동성이 90% 이상인 아미노산 서열;The CDR1 of SEQ ID NO: 14 or an amino acid sequence having at least 90% homology with it;
서열번호 15의 CDR2 또는 이와 상동성이 90% 이상인 아미노산서열; 및CDR2 of SEQ ID NO: 15 or an amino acid sequence having a homology of 90% or more; And
서열번호 16의 CDR3 또는 이와 상동성이 90% 이상인 아미노산 서열;을 포함할 수 있다. CDR3 of SEQ ID NO: 16 or an amino acid sequence having 90% or more homology thereto;
본 발명의 더욱 바람직한 구체예에서, 중쇄가변영역은 서열번호 13의 아미노산으로 이루어질 수 있다.In a more preferred embodiment of the invention, the heavy chain variable region may consist of the amino acid of SEQ ID NO: 13.
또한, 본 발명의 일 양상은 상기 항체에 융합된, 펩타이드, 단백질, 소분자 약물, 나노입자 및 리포좀으로 이루어진 군으로부터 선택된 생체활성분자를 제공한다. In addition, an aspect of the present invention provides a bioactive molecule selected from the group consisting of peptides, proteins, small molecule drugs, nanoparticles and liposomes, fused to the antibody.
상기 단백질은 단백질은 항체, 항체의 단편, 면역 글로불린, 펩타이드, 효소, 성장인자 (growth factor), 사이토카인 (cytokine), 전사인자, 독소, 항원성 펩티드, 호르몬, 운반 단백질, 운동 기능 단백질, 수용체, 신호(signaling) 단백질, 저장 단백질, 막 단백질, 막횡단(transmembrane) 단백질, 내부(internal) 단백질, 외부(external) 단백질, 분비 단백질, 바이러스 단백질, 당 단백질, 절단된 단백질, 단백질 복합체, 또는 화학적으로 개질된 단백질 등일 수 있다. The protein may be an antibody, a fragment of an antibody, an immunoglobulin, a peptide, an enzyme, a growth factor, a cytokine, a transcription factor, a toxin, an antigenic peptide, a hormone, a carrier protein, a motor function protein, a receptor , Signaling proteins, storage proteins, membrane proteins, transmembrane proteins, internal proteins, external proteins, secreted proteins, viral proteins, glycoproteins, truncated proteins, protein complexes, or chemical Modified proteins, and the like.
본 발명의 구체적인 실시예에서는 세포질 침투를 통하여 활성화된(GTP와 결합하는) RAS를 특이적 결합 및 저해하는 완전 이뮤노글로불린 형태 항체의 경쇄가변영역의 N-말단에 RGD4C 펩타이드를 융합한 형태를 제공한다. 상기 본 발명의 일 구체예에 있어서 경쇄가변영역 N-말단과 RGD4C 펩타이드는 (G4S)1 연결자(linker)로 융합되는 것이 바람직하지만 이에 제한 되지 않는다.In a specific embodiment of the present invention provides a form in which the RGD4C peptide is fused to the N-terminus of the light chain variable region of a complete immunoglobulin-type antibody that specifically binds to and inhibits RAS activated (which binds to GTP) through cellular infiltration. do. In one embodiment of the present invention, the light chain variable region N-terminus and the RGD4C peptide are preferably fused to (G 4 S) 1 linker, but are not limited thereto.
본 발명에 있어서 소분자 약물은 약 1000 달톤 미만의 분자량을 지니며 질병의 치료제로서 활성도를 지니는 유기 화합물, 무기 화합물 또는 유기금속 화합물을 나타내는 것으로 본원에서 광범위하게 사용된다. 본원에서 소분자 약물은 올리고펩티드 및 약 1000 달톤 미만의 분자량을 지니는 그 밖의 바이오분자(biomolecule)를 포함한다.Small molecule drugs in the present invention are used broadly herein to denote organic compounds, inorganic compounds or organometallic compounds having a molecular weight of less than about 1000 Daltons and having activity as therapeutic agents for the disease. Small molecule drugs herein include oligopeptides and other biomolecules having a molecular weight of less than about 1000 Daltons.
본 발명에 있어서, 나노입자(nanoparticle)는 직경 1 내지 1000 nm 크기를 갖는 물질들로 이루어진 입자를 의미하며, 상기 나노 입자는 금속 나노 입자, 금속 나노 입자 코어 및 상기 코어를 둘러싸는 금속 쉘로 구성되는 금속/금속 코어쉘 복합체, 금속 나노 입자 코어 및 상기 코어를 둘러싸는 비금속 쉘로 구성되는 금속/비금속 코어쉘 또는 비금속 나노 입자 코어 및 상기 코어를 둘러싸는 금속 쉘로 구성되는 비금속/금속 코어쉘 복합체일 수 있다. 일 구체예에 따르면, 상기 금속은 금, 은, 구리, 알루미늄, 니켈, 팔라듐, 백금, 자성철 및 그의 산화물로부터 선택되는 것일 수 있으나, 이에 한정하지는 않으며, 상기 비금속은 실리카, 폴리스티렌, 라텍스 및 아크릴레이트 계열의 물질로부터 선택되는 것일 수 있으나, 이에 한정하지는 않는다.In the present invention, the nanoparticle (nanoparticle) means a particle made of a material having a diameter of 1 to 1000 nm, the nanoparticle is composed of a metal nanoparticle, a metal nanoparticle core and a metal shell surrounding the core It may be a metal / nonmetal coreshell composed of a metal / metal coreshell composite, a metal nanoparticle core and a nonmetal shell surrounding the core, or a nonmetal / metal coreshell composite composed of a nonmetal nanoparticle core and a metal shell surrounding the core. . According to one embodiment, the metal may be selected from gold, silver, copper, aluminum, nickel, palladium, platinum, magnetic iron and oxides thereof, but is not limited thereto, and the nonmetal may be silica, polystyrene, latex, and acrylic. It may be selected from the rate-based material, but is not limited thereto.
본 발명에 있어서, 리포좀은 자기 스스로 회합할 수 있는, 수성 내부 구획을 둘러싸는 하나 이상의 지질 이중층 막으로 구성된다. 리포좀은 막 타입 및 그 크기에 의하여 특정 수 있다. 작은 유니라멜라 소포(SUV)는 단일막을 갖고 20nm 내지 50nm의 직경을 가질 수 있다. 큰 유니라멜라 소포(LUV)는 50nm이상의 직경을 가질 수 있다. 올리고라멜라 큰 소포 및 멀티라멜라 큰 소포는 다중, 일반적으로 동심원, 막 층을 가지고 직경이 100nm 이상일 수 있다. 여러 비동심원 막을 가진 리포좀, 즉 더 큰 소포 내에서 포함된 여러 작은 소포는 멀티소포성 소포(multivesicular vesicle)라고 한다.In the present invention, liposomes are composed of one or more lipid bilayer membranes surrounding an aqueous internal compartment that can associate themselves. Liposomes can be specified by membrane type and size. Small unilamellar vesicles (SUVs) have a single membrane and may have a diameter of 20 nm to 50 nm. Large uni-lamellar vesicles (LUV) may have a diameter of 50 nm or more. Oligolamella large vesicles and multilamellar large vesicles have multiple, generally concentric, membrane layers and may be 100 nm or more in diameter. Liposomes with multiple asymmetrical membranes, ie several small vesicles contained within larger vesicles, are called multivesicular vesicles.
본 발명에 있어서 “융합”은 기능 또는 구조가 다르거나 같은 두 분자를 일체화하는 것으로, 상기 단백질, 소분자 약물, 나노입자 또는 리포좀에 상기 종양 침투성 펩타이드가 결합할 수 있는 모든 물리, 화학적 또는 생물학적 방법에 의한 융합일 수 있다. 상기 융합은 바람직하게는 연결자 펩타이드에 의할 수 있으며, 이 연결자 펩타이드는 본 발명의 항체 경쇄 가변 영역, 항체, 또는 이의 절편의 다양한 위치에서 상기 생체 활성 분자와의 융합을 중계할 수 있다. In the present invention, "fusion" refers to the integration of two molecules having different or the same function or structure, and any physical, chemical or biological method in which the tumor-penetrating peptide can bind to the protein, small molecule drug, nanoparticle or liposome. By fusion. The fusion may preferably be by a linker peptide, which may relay fusion with the bioactive molecule at various positions in the antibody light chain variable region, antibody, or fragment thereof.
또한, 본 발명은 상기 항체, 또는 이에 융합된 펩타이드, 단백질, 소분자 약물, 나노입자 및 리포좀으로 이루어진 군으로부터 선택된 생체활성분자를 포함하는 암의 예방 또는 치료용 약학적 조성물을 제공한다.The present invention also provides a pharmaceutical composition for preventing or treating cancer, including a bioactive molecule selected from the group consisting of the antibody, or a peptide, a protein, a small molecule drug, a nanoparticle, and a liposome fused thereto.
본 발명에 따른 항체, 또는 이에 융합된 펩타이드, 단백질, 소분자 약물, 나노입자 및 리포좀으로 이루어진 군으로부터 선택된 생체활성분자를 이용하여 인간 항체 중쇄가변영역(VH)이 가지는 항원 고특이성 및 고친화도에 영향주지 않으면서 세포내부로 침투 및 세포질에 잔류하는 특성을 부여할 수 있으며, 이를 통해 현재 소분자 약물을 이용한 질병 치료에 표적물질로 분류되고 있는 세포질 내에 존재하면서 단백질과 단백질 사이에 넓고 평평한 표면을 통해 구조복합성 상호작용을 이루는 종양 및 질환 관련 인자에 대한 치료 및 진단에 높은 효과를 기대할 수 있다. 또한 기존 다양한 종양치료제의 주요 약물 저항성 관련 인자인 KRas 돌연변이를 선택적 저해가 가능하면서 기존 치료제와의 병행치료를 통해 효과적인 항암 활성을 기대할 수 있다.The high specificity and high affinity of the antigen of the human antibody heavy chain variable region (VH) is influenced by using a bioactive molecule selected from the group consisting of an antibody according to the present invention or peptides, proteins, small molecule drugs, nanoparticles, and liposomes fused thereto. It is able to penetrate into the cell and remain in the cytoplasm without being known, and it is present in the cytoplasm that is currently classified as a target for the treatment of diseases using small molecule drugs. A high effect can be expected in the treatment and diagnosis of tumor and disease related factors that make up complex interactions. In addition, while it is possible to selectively inhibit the KRas mutation, which is a major drug resistance-related factor of various existing tumor therapeutic agents, effective anticancer activity can be expected through concurrent treatment with existing therapeutic agents.
상기 암은 편평상피세포암, 소세포폐암, 비소세포폐암, 폐의 선암, 폐의 편평상피암, 복막암, 피부암, 피부 또는 안구내 흑색종, 직장암, 항문부근암, 식도암, 소장암, 내분비선암, 부갑상선암, 부신암, 연조직 육종, 요도암, 만성 또는 급성 백혈병, 림프구 림프종, 간세포암, 위장암, 췌장암, 교아종, 경부암, 난소암, 간암, 방광암, 간종양, 유방암, 결장암, 대장암, 자궁내막 또는 자궁암, 침샘암, 신장암, 간암, 전립선암, 음문암, 갑상선암, 간암 및 두경부암으로 이루어진 군으로부터 선택되는 것일 수 있다.The cancer includes squamous cell carcinoma, small cell lung cancer, non-small cell lung cancer, lung adenocarcinoma, lung squamous cell carcinoma, peritoneal cancer, skin cancer, skin or intraocular melanoma, rectal cancer, anal muscle cancer, esophageal cancer, small intestine cancer, endocrine adenocarcinoma, Parathyroid cancer, adrenal cancer, soft tissue sarcoma, urethral cancer, chronic or acute leukemia, lymphocyte lymphoma, hepatocellular carcinoma, gastrointestinal cancer, pancreatic cancer, glioblastoma, cervical cancer, ovarian cancer, liver cancer, bladder cancer, liver tumor, breast cancer, colon cancer, colon cancer, Endometrial or uterine cancer, salivary gland cancer, kidney cancer, liver cancer, prostate cancer, vulva cancer, thyroid cancer, liver cancer and head and neck cancer.
상기 조성물이 암의 예방 또는 치료용 약학적 조성물로 제조되는 경우, 상기 조성물은 약학적으로 허용되는 담체를 포함할 수 있다. 상기 조성물에 포함되는 약학적으로 허용되는 담체는 제제시에 통상적으로 이용되는 것으로서, 락토오스, 덱스트로오스, 수크로오스, 솔비톨, 만니톨, 전분, 아카시아 고무, 인산 칼슘, 알기네이트, 젤라틴, 규산 칼슘, 미세결정성 셀룰로오스, 폴리비닐피롤리돈, 셀룰로오스, 물, 시럽, 메틸 셀룰로오스, 메틸히드록시벤조에이트, 프로필히드록시벤조에이트, 활석, 스테아르산 마그네슘 및 미네랄 오일 등을 포함하나, 이에 한정되는 것은 아니다. 상기 약학적 조성물은 상기 성분들 이외에 윤활제, 습윤제, 감미제, 향미제, 유화제, 현탁제, 보존제 등을 추가로 포함할 수 있다.When the composition is prepared as a pharmaceutical composition for preventing or treating cancer, the composition may include a pharmaceutically acceptable carrier. Pharmaceutically acceptable carriers included in the composition are conventionally used in the preparation, lactose, dextrose, sucrose, sorbitol, mannitol, starch, acacia rubber, calcium phosphate, alginate, gelatin, calcium silicate, fine Crystalline cellulose, polyvinylpyrrolidone, cellulose, water, syrup, methyl cellulose, methylhydroxybenzoate, propylhydroxybenzoate, talc, magnesium stearate and mineral oil, and the like. The pharmaceutical composition may further include lubricants, wetting agents, sweeteners, flavoring agents, emulsifiers, suspending agents, preservatives, and the like, in addition to the above components.
상기 암의 예방 또는 치료용 약학적 조성물은 경구 또는 비경구로 투여할 수 있다. 비경구 투여인 경우에는 정맥내 주입, 피하 주입, 근육 주입, 복강 주입, 내피 투여, 국소 투여, 비내 투여, 폐내 투여 및 직장내 투여 등으로 투여할 수 있다. 경구 투여시, 단백질 또는 펩타이드는 소화가 되기 때문에 경구용 조성물은 활성 약제를 코팅하거나 위에서의 분해로부터 보호되도록 제형화 되어야 한다. 또한, 상기 조성물은 활성 물질이 표적 세포로 이동할 수 있는 임의의 장치에 의해 투여될 수 있다.The pharmaceutical composition for preventing or treating cancer may be administered orally or parenterally. In the case of parenteral administration, it can be administered by intravenous injection, subcutaneous injection, intramuscular injection, intraperitoneal injection, endothelial administration, topical administration, intranasal administration, pulmonary administration and rectal administration. In oral administration, because proteins or peptides are digested, oral compositions should be formulated to coat the active agent or to protect it from degradation in the stomach. In addition, the composition may be administered by any device in which the active substance may migrate to the target cell.
상기 암의 예방 또는 치료용 약학적 조성물의 적합한 투여량은 제제화 방법, 투여 방식, 환자의 연령, 체중, 성, 병적 상태, 음식, 투여 시간, 투여 경로, 배설 속도 및 반응 감응성과 같은 요인들에 의해 다양하게 처방될 수 있다. 상기 조성물의 바람직한 투여량은 성인 기준으로 0.001-100 ㎎/kg 범위 내이다. 용어 "약학적 유효량"은 암을 예방 또는 치료하는 데, 또는 혈관신생으로 인한 질환의 예방 또는 치료하는 데 충분한 양을 의미한다.Suitable dosages of the pharmaceutical compositions for the prophylaxis or treatment of cancer are dependent on factors such as the formulation method, mode of administration, age, weight, sex, morbidity, food, time of administration, route of administration, rate of excretion and response to response of the patient. It can be prescribed in various ways. Preferred dosages of the compositions are in the range of 0.001-100 mg / kg on an adult basis. The term "pharmaceutically effective amount" means an amount sufficient to prevent or treat cancer or to prevent or treat a disease due to angiogenesis.
상기 조성물은 당해 당업자가 용이하게 실시할 수 있는 방법에 따라, 약학적으로 허용되는 담체 및/또는 부형제를 이용하여 제제화함으로써 단위 용량 형태로 제조되거나 또는 다용량 용기 내에 내입시켜 제조될 수 있다. 이때 제형은 오일 또는 수성 매질중의 용액, 현탁액, 시럽제 또는 유화액 형태이거나 엑스제, 산제, 분말제, 과립제, 정제 또는 캅셀제 형태일 수도 있으며, 분산제 또는 안정화제를 추가적으로 포함할 수 있다. 또한, 상기 조성물은 개별 치료제로 투여하거나 다른 치료제와 병용하여 투여될 수 있고, 종래의 치료제와는 순차적 또는 동시에 투여될 수 있다. 한편, 상기 조성물은 항체 또는 항원 결합 단편을 포함하므로, 면역 리포좀으로 제형화될 수 있다. 항체를 포함하는 리포좀은 당업계에 널리 알려진 방법에 따라 제조될 수 있다. 상기 면역 리포좀은 포스파티딜콜린, 콜레스테롤 및 폴리에틸렌글리콜-유도체화된 포스파티딜에탄올아민을 포함하는 지질 조성물로서 역상 증발법에 의해 제조될 수 있다. 예를 들어, 항체의 Fab' 단편은 디설파이드-교체 반응을 통해 리포좀에 접합될 수 있다. 독소루비신과 같은 화학치료제가 추가로 리포좀 내에 포함될 수 있다.The composition may be prepared in unit dose form or formulated into a multi-dose container by formulating with a pharmaceutically acceptable carrier and / or excipient, according to methods readily available to those skilled in the art. The formulation may be in the form of solutions, suspensions, syrups or emulsions in oils or aqueous media, or in the form of extracts, powders, powders, granules, tablets or capsules, and may further comprise dispersants or stabilizers. In addition, the composition may be administered as a separate therapeutic agent or in combination with other therapeutic agents, and may be administered sequentially or simultaneously with conventional therapeutic agents. On the other hand, since the composition comprises an antibody or antigen-binding fragment, it can be formulated as an immune liposome. Liposomes comprising the antibody can be prepared according to methods well known in the art. The immune liposomes can be prepared by reverse phase evaporation as a lipid composition comprising phosphatidylcholine, cholesterol and polyethyleneglycol-derivatized phosphatidylethanolamine. For example, Fab 'fragments of antibodies can be conjugated to liposomes via disulfide-replacement reactions. Chemotherapeutic agents such as doxorubicin may further be included in the liposomes.
또한, 본 발명은 상기 항체, 또는 이에 융합된 펩타이드, 단백질, 소분자 약물, 나노입자 및 리포좀으로 이루어진 군으로부터 선택된 생체활성분자를 포함하는 암의 진단용 조성물을 제공한다.The present invention also provides a composition for diagnosing cancer comprising a bioactive molecule selected from the group consisting of the antibody, or a peptide, a protein, a small molecule drug, a nanoparticle, and a liposome fused thereto.
본 명세서에서 사용된 용어 "진단"은 병태 생리의 존재 또는 특징을 확인하는 것을 의미한다. 본 발명에서의 진단은 암의 발병 여부 및 경과를 확인하는 것이다.As used herein, the term “diagnosis” refers to identifying the presence or characteristic of pathophysiology. Diagnosis in the present invention is to determine the onset and progress of cancer.
상기 완전한 이뮤노글로불린 형태의 항체 및 이의 절편은 영상을 통하여 암을 진단하기 위하여 분자 영상용 형광체와 결합할 수 있다. The complete immunoglobulin form of the antibody and fragments thereof may be combined with a phosphor for molecular imaging to diagnose cancer through imaging.
상기 분자 영상용 형광체는 형광을 발생시키는 모든 물질을 말하며, 적색이나 근적외선(near-infrared)의 형광을 발광하는 것이 바람직하며, 양자 수득량(quantaum yield)이 높은 형광체가 더욱 바람직하나 이에 한정되지 않는다.The fluorescent material for molecular imaging refers to any material generating fluorescence, and preferably emits red or near-infrared fluorescence, and more preferably, a phosphor having a high quantum yield is more preferred, but is not limited thereto. .
상기 분자 영상용 형광체는 상기 완전한 이뮤노글로불린 형태의 항체 및 이의 절편에 특이적으로 결합하는 종양 침투성 펩타이드와 결합할 수 있는 형광체, 형광 단백질 또는 기타 영상용 물질이 바람직하나 이에 한정되지 않는다.The molecular imaging phosphor is preferably, but not limited to, a phosphor, a fluorescent protein or other imaging material capable of binding to a tumor-penetrating peptide specifically binding to the antibody and fragments thereof of the complete immunoglobulin form.
형광체는 플루오레신(fluorescein), 보디피(BODYPY), 테트라메틸로드아민(Trtramethylrhodamine), 알렉사(Alexa), 시아닌(Cyanine), 알로피코시아닌(allopicocyanine) 또는 이들의 유도체가 바람직하나 이에 한정되지 않는다.The phosphor is preferably fluorescein, BODYPY, tetramethylrhodamine, Alexa, cyanine, allopicocyanine or derivatives thereof, but not limited thereto. Do not.
형광 단백질은 Dronpa 단백질, 형광 발색 유전자(EGFP), 적색 형광 프로테인(red fluorescent protein, DsRFP), 근적외선 형광을 나타내는 시아닌 형광체인 Cy5.5 또는 기타 형광 단백질이 바람직하나 이에 한정되지 않는다.The fluorescent protein is preferably, but not limited to, Dronpa protein, fluorescent color gene (EGFP), red fluorescent protein (DsRFP), Cy5.5 or other fluorescent protein which is a near infrared fluorescence.
기타 영상용 물질은 산화철, 방사성 동위원소 등이 바람직하나 이에 한정되지 않으며, MR, PET과 같은 영상 장비에 응용될 수 있다.Other imaging materials are preferably iron oxide, radioisotopes, etc., but are not limited thereto, and may be applied to imaging equipment such as MR and PET.
또한 본 발명은 상기 경쇄가변영역 또는 이를 포함하는 항체 또는 이의 절편을 코딩하는 폴리뉴클레오티드를 제공한다.In another aspect, the present invention provides a polynucleotide encoding the light chain variable region or an antibody or fragment thereof.
상기 “폴리뉴클레오티드(polynucleotide)”는 단일가닥 또는 이중가닥 형태로 존재하는 디옥시리보뉴클레오티드 또는 리보뉴클레오티드의 중합체이다. RNA 게놈 서열, DNA(gDNA 및 cDNA) 및 이로부터 전사되는 RNA 서열을 포괄하며, 특별하게 다른 언급이 없는 한 천연의 폴리뉴클레오티드의 유사체를 포함한다.The "polynucleotide" is a polymer of deoxyribonucleotides or ribonucleotides present in single- or double-stranded form. It encompasses RNA genomic sequences, DNA (gDNA and cDNA) and RNA sequences transcribed therefrom and includes analogs of natural polynucleotides unless specifically stated otherwise.
상기 폴리뉴클레오티드는 상기 기술한 경쇄 가변영역을 코딩하는 뉴클레오티드 서열뿐만 아니라, 그 서열에 상보적인(complementary) 서열도 포함한다. 상기 상보적인 서열은 완벽하게 상보적인 서열뿐만 아니라, 실질적으로 상보적인 서열도 포함한다. 이는 당업계에 공지된 가혹 조건(stringent conditions) 하에서, 예를 들어, 서열번호 1 내지 3 중 어느 하나의 아미노산 서열을 코딩하는 뉴클레오티드 서열과 혼성화될 수 있는 서열을 의미한다.The polynucleotide includes not only the nucleotide sequence encoding the light chain variable region described above, but also a complementary sequence to the sequence. Such complementary sequences include sequences that are substantially complementary, as well as sequences that are substantially complementary. This means a sequence that can hybridize under stringent conditions known in the art, for example, with a nucleotide sequence encoding the amino acid sequence of any one of SEQ ID NOS: 1-3.
또한 상기 폴리뉴클레오티드는 변형될 수 있다. 상기 변형은 뉴클레오티드의 추가, 결실 또는 비보존적 치환 또는 보존적 치환을 포함한다. 상기 아미노산 서열을 코딩하는 폴리뉴클레오티드는 상기 뉴클레오티드 서열에 대하여 실질적인 동일성을 나타내는 뉴클레오티드 서열도 포함하는 것으로 해석된다. 상기의 실질적인 동일성은, 상기 뉴클레오티드 서열과 임의의 다른 서열을 최대한 대응되도록 얼라인하고, 당업계에서 통상적으로 이용되는 알고리즘을 이용하여 얼라인된 서열을 분석한 경우에, 최소 80%의 상동성, 최소 90%의 상동성 또는 최소 95%의 상동성을 나타내는 서열일 수 있다.The polynucleotide may also be modified. Such modifications include addition, deletion or non-conservative substitutions or conservative substitutions of nucleotides. The polynucleotide encoding the amino acid sequence is to be interpreted to also include a nucleotide sequence showing substantial identity to the nucleotide sequence. The substantial identity is at least 80% homology when the nucleotide sequence is aligned with any other sequence as closely as possible and the aligned sequence is analyzed using algorithms commonly used in the art. A sequence exhibiting at least 90% homology or at least 95% homology.
또한, 본 발명의 일 양상은 항체의 경쇄가변영역을, 살아있는 세포 내부로 침투하여 세포질에 위치하는 특성을 가진 경쇄가변영역으로 치환하는 단계를 포함하는, In addition, an aspect of the present invention includes the step of replacing the light chain variable region of the antibody with a light chain variable region having a characteristic of infiltrating into the living cell and located in the cytoplasm,
살아있는 세포내부로 침투하여 세포질에 위치하는 항체의 제조방법을 제공할 수 있다. It is possible to provide a method for producing an antibody that penetrates into living cells and is located in the cytoplasm.
또한, 본 발명의 일 구체예는 기존의 완전 이뮤노글로불린 형태의 항체의 경쇄가변영역(VL)을 세포질 침투 경쇄가변영역(VL)과 치환하여, 치환된 완전한 이뮤노글로불린 형태의 단일클론항체가 상기 세포질 침투능을 가지는 완전 이뮤노글로불린 형태의 단일클론항체의 세포질 침투와 같은 특성을 갖게 하는 방법을 제공할 수 있다.In addition, one embodiment of the present invention is to replace the light chain variable region (VL) of the existing full immunoglobulin form of the antibody with the cytoplasmic penetration light chain variable region (VL), the monoclonal antibody of the substituted full immunoglobulin form It is possible to provide a method of having the same characteristics as the cellular penetration of a monoclonal antibody in the form of a complete immunoglobulin having the cytoplasmic penetration ability.
본 발명의 구체예에서, 세포질 침투 경쇄가변영역(VL)을 이용한 세포내부로 침투하여 세포질에 분포하는 완전 이뮤노글로불린 형태의 항체 제조방법의 예는 아래와 같다.In an embodiment of the present invention, an example of a method for producing a fully immunoglobulin-type antibody which penetrates into the cell using the cytoplasmic infiltration light chain variable region (VL) and distributes the cytoplasm is as follows.
(1) 상기 인간 경쇄가변영역(VL) 및 인간 경쇄불변영역(CL)을 포함하는 경쇄에서 경쇄가변영역(VL)을 세포질에 침투하는 인간화 경쇄가변영역(VL)으로 치환된 핵산(nucleic acids)을 클로닝한 세포질 침투 경쇄 발현 벡터를 제조하는 단계;(1) a nucleic acid substituted with a humanized light chain variable region (VL) that penetrates the light chain variable region (VL) into the cytoplasm in a light chain including the human light chain variable region (VL) and the human light chain constant region (CL) Preparing a cytoplasmic infiltrating light chain expression vector which has been cloned;
(2) 상기 제조된 경쇄와 상호작용하여 완전한 이뮤노글로불린 형태의 항체 발현을 위한 중쇄가변영역(VH) 및 중쇄불변영역(CH1-hinge-CH2-CH3)이 포함된 중쇄를 코딩하는 핵산(nucleic acides)를 클로닝한 중쇄 발현 벡터를 제조하는 단계;(2) a nucleic acid encoding a heavy chain including a heavy chain variable region (VH) and a heavy chain constant region (CH1-hinge-CH2-CH3) for interacting with the prepared light chain to express the antibody in the form of a complete immunoglobulin preparing a heavy chain expression vector cloned from the acides);
(3) 상기 제조된 경쇄, 중쇄 발현벡터를 단백질 발현용 동물세포에 동시 형질전환하여 세포내부 침투 및 세포질 잔류 인간화 경쇄가변영역(VL)을 포함하는 완전한 이뮤노글로불린 형태의 항체를 발현하는 단계; 및(3) co-transforming the prepared light and heavy chain expression vectors into animal cells for protein expression to express the complete immunoglobulin type antibody including intracellular infiltration and cytoplasmic residual humanized light chain variable region (VL); And
(4) 상기 발현된 세포질 침투능을 가지는 완전한 이뮤노글로불린 형태의 항체를 정제 및 회수하는 단계.(4) purifying and recovering the antibody in the form of a complete immunoglobulin having the expressed cytoplasmic permeability.
상기 방법은 경쇄를 발현하는 벡터와 중쇄를 발현하는 벡터를 발현하여 세포질 침투능을 가지는 완전한 이뮤노글로불린 형태의 항체를 제조할 수 있다. 또한, 세포내부의 특정 단백질을 인지할 수 있는 중쇄가변영역을 포함하는 중쇄를 발현하는 벡터와 함께 형질전환을 통하여, 세포내부로 침투하고 세포질에 분포하여 특정 단백질과 결합할 수 잇는 항체를 발현할 수 있다. 벡터는 경쇄와 중쇄를 하나의 벡터에서 동시에 발현되는 벡터 시스템이거나 또는 각각 별도의 벡터에서 발현시키는 시스템 모두 가능하다. 후자의 경우, 두 벡터는 동시 형질전환(co-transfomation) 및 표적 형질전환(targeted transformation)을 통하여 숙주 세포로 도입될 수 있다.The method can express a vector expressing a light chain and a vector expressing a heavy chain to prepare antibodies in the form of complete immunoglobulins having cytoplasmic penetrating ability. In addition, through transformation with a vector expressing a heavy chain comprising a heavy chain variable region capable of recognizing a specific protein within a cell, the antibody may penetrate into the cell and be distributed in the cytoplasm to express an antibody capable of binding to a specific protein. Can be. The vector can be either a vector system in which the light and heavy chains are simultaneously expressed in one vector or a system in which each is expressed in a separate vector. In the latter case, both vectors can be introduced into the host cell via co-transfomation and targeted transformation.
본 발명에서 사용하는 용어 "벡터(vector)"는 숙주 세포에서 목적 유전자를 발현시키기 위한 수단을 의미한다. 예를 들어, 플라스미드 벡터, 코즈미드 벡터 및 박테리오파아지 벡터, 아데노바이러스 벡터, 레트로바이러스 벡터 및 아데노-연관 바이러스 벡터와 같은 바이러스 벡터를 포함한다. 상기 재조합 벡터로 사용될 수 있는 벡터는 당업계에서 종종 사용되는 플라스미드(예를 들면, pSC101, pGV1106, pACYC177, ColE1, pKT230, pME290, pBR322, pUC8/9, pUC6, pBD9, pHC79, pIJ61, pLAFR1, pHV14, pGEX 시리즈, pET 시리즈 및 pUC19 등), 파지(예를 들면, λgt4λB, λ-Charon, λΔz1 및 M13 등) 또는 바이러스(예를 들면, CMV, SV40 등)를 조작하여 제작될 수 있다.As used herein, the term "vector" means a means for expressing a gene of interest in a host cell. For example, viral vectors such as plasmid vectors, cosmid vectors and bacteriophage vectors, adenovirus vectors, retrovirus vectors, and adeno-associated virus vectors are included. Vectors that can be used as the recombinant vector are plasmids often used in the art (eg, pSC101, pGV1106, pACYC177, ColE1, pKT230, pME290, pBR322, pUC8 / 9, pUC6, pBD9, pHC79, pIJ61, pLAFR1, pHV14). , pGEX series, pET series, pUC19 and the like), phage (e.g., λgt4λB, λ-Charon, λΔz1 and M13, etc.) or viruses (e.g., CMV, SV40, etc.).
상기 재조합 벡터에서 본 발명에서 제공하는 경쇄가변영역, 및 경쇄불변영역(CL), 중쇄가변영역(VH) 및 중쇄불변영역(CH1-hinge-CH2-CH3)은 프로모터에 작동적으로 연결될 수 있다. 용어 "작동적으로 연결된(operatively linked)"은 뉴클레오티드 발현 조절 서열(예를 들면, 프로모터 서열)과 다른 뉴클레오티드 서열 사이의 기능적인 결합을 의미한다. 따라서, 이에 의해 상기 조절 서열은 상기 다른 뉴클레오티드 서열의 전사 및/또는 해독을 조절할 수 있다.In the recombinant vector, the light chain variable region, the light chain constant region (CL), the heavy chain variable region (VH), and the heavy chain constant region (CH1-hinge-CH2-CH3) provided in the present invention may be operably linked to a promoter. The term "operatively linked" means a functional bond between a nucleotide expression control sequence (eg, a promoter sequence) and another nucleotide sequence. Thus, the regulatory sequence can thereby regulate transcription and / or translation of the other nucleotide sequence.
상기 재조합 벡터는, 전형적으로 클로닝을 위한 벡터 또는 발현을 위한 벡터로서 구축될 수 있다. 상기 발현용 벡터는 당업계에서 식물, 동물 또는 미생물에서 외래의 단백질을 발현하는데 사용되는 통상의 것을 사용할 수 있다. 상기 재조합 벡터는 당업계에 공지된 다양한 방법을 통해 구축될 수 있다. The recombinant vector can typically be constructed as a vector for cloning or a vector for expression. The expression vector may be a conventional one used in the art to express foreign proteins in plants, animals or microorganisms. The recombinant vector may be constructed through various methods known in the art.
상기 재조합 벡터는 원핵 세포 또는 진핵 세포를 숙주로 하여 구축될 수 있다. 예를 들어, 사용되는 벡터가 발현 벡터이고, 원핵 세포를 숙주로 하는 경우에는, 전사를 진행시킬 수 있는 강력한 프로모터(예를 들어, pLλ프로모터, trp 프로모터, lac 프로모터, tac 프로모터, T7 프로모터 등), 해독의 개시를 위한 라이보좀 결합 자리 및 전사/해독 종결 서열을 포함하는 것이 일반적이다. 진핵 세포를 숙주로 하는 경우에는, 벡터에 포함되는 진핵 세포에서 작동하는 복제원점은 f1 복제원점, SV40 복제원점, pMB1 복제원점, 아데노 복제원점, AAV 복제원점, CMV 복제원점 및 BBV 복제원점 등을 포함하나, 이에 한정되는 것은 아니다. 또한, 포유동물 세포의 게놈으로부터 유래된 프로모터(예를 들어, 메탈로티오닌 프로모터) 또는 포유동물 바이러스로부터 유래된 프로모터(예를 들어, 아데노바이러스 후기 프로모터, 백시니아 바이러스 7.5K 프로모터, SV40 프로모터, 사이토메갈로바이러스(CMV) 프로모터 및 HSV의 tk 프로모터)가 이용될 수 있으며, 전사 종결 서열로서 폴리아데닐화 서열을 일반적으로 갖는다.The recombinant vector may be constructed using prokaryotic or eukaryotic cells as hosts. For example, when the vector used is an expression vector and the prokaryotic cell is a host, a strong promoter (for example, a pLλ promoter, trp promoter, lac promoter, tac promoter, T7 promoter, etc.) capable of promoting transcription It is common to include ribosome binding sites and transcription / detox termination sequences for initiation of translation. In the case of using a eukaryotic cell as a host, replication origins that operate in eukaryotic cells included in the vector include f1 origin, SV40 origin, pMB1 origin, adeno origin, AAV origin, CMV origin, and BBV origin. Including but not limited to. In addition, promoters derived from the genome of mammalian cells (eg, metallothionine promoters) or promoters derived from mammalian viruses (eg, adenovirus late promoters, vaccinia virus 7.5K promoters, SV40 promoters, Cytomegalovirus (CMV) promoter and tk promoter of HSV) can be used and generally have a polyadenylation sequence as a transcription termination sequence.
본 발명의 또 다른 양상은 상기 재조합 벡터로 형질전환된 숙주세포를 제공할 수 있다.Another aspect of the invention can provide a host cell transformed with the recombinant vector.
숙주 세포는 당업계에 공지된 어떠한 숙주 세포도 이용할 수 있으며, 원핵 세포로는, 예를 들어, E. coli JM109, E. coli BL21, E. coli RR1, E. coli LE392, E. coli B, E. coli X 1776, E. coli W3110, 바실러스 서브틸리스, 바실러스 츄린겐시스와 같은 바실러스 속 균주, 그리고 살모넬라 티피무리움, 세라티아 마르세슨스 및 다양한 슈도모나스 종과 같은 장내균과 균주 등이 있으며, 진핵 세포에 형질 전환시키는 경우에는 숙주 세포로서, 효모(Saccharomyce cerevisiae), 곤충 세포, 식물 세포 및 동물 세포, 예를 들어, SP2/0, CHO(Chinese hamster ovary) K1, CHO DG44, PER.C6, W138, BHK, COS-7, 293, HepG2, Huh7, 3T3, RIN 및 MDCK 세포주 등이 이용될 수 있다.The host cell may use any host cell known in the art, and prokaryotic cells include, for example, E. coli JM109, E. coli BL21, E. coli RR1, E. coli LE392, E. coli B, Strains of the genus Bacillus, such as E. coli X 1776, E. coli W3110, Bacillus subtilis, Bacillus thuringiensis, and enterococci and strains such as Salmonella typhimurium, Serratia marsons and various Pseudomonas species. In the case of transformation into cells, as a host cell, yeast (Saccharomyce cerevisiae), insect cells, plant cells and animal cells, for example, SP2 / 0, Chinese hamster ovary K1, CHO DG44, PER.C6, W138 , BHK, COS-7, 293, HepG2, Huh7, 3T3, RIN and MDCK cell lines and the like can be used.
본 발명의 또 다른 양상은 상기 숙주세포를 배양하는 단계를 포함하는, 세포내부로 침투하여 세포질에 위치하는 완전한 이뮤노글로불린 형태의 항체 제조방법을 제공할 수 있다.Another aspect of the present invention can provide a method for producing a complete immunoglobulin-type antibody in the cytoplasm to penetrate into the cell, comprising culturing the host cell.
상기 재조합 벡터의 숙주 세포 내로의 삽입은, 당업계에 널리 알려진 삽입 방법을 사용할 수 있다. 상기 운반 방법은 예를 들어, 숙주 세포가 원핵 세포인 경우, CaCl2 방법 또는 전기 천공 방법 등을 사용할 수 있고, 숙주 세포가 진핵 세포인 경우에는, 미세 주입법, 칼슘 포스페이트 침전법, 전기 천공법, 리포좀-매개 형질감염법 및 유전자 밤바드먼트 등을 사용할 수 있으나, 이에 한정하지는 않는다. E. coli 등의 미생물을 이용하는 경우 생산성은 동물세포 등에 비하여 높은 편이나 당화(glycosylation) 문제로 인해 인택트(intact)한 Ig 형태의 항체 생산에는 적당하지 않지만, Fab 및 Fv와 같은 항원 결합 단편의 생산에는 사용될 수 있다.Insertion of the recombinant vector into the host cell, can be used insertion methods well known in the art. For example, when the host cell is a prokaryotic cell, a CaCl 2 method or an electroporation method may be used. When the host cell is a eukaryotic cell, a micro-injection method, calcium phosphate precipitation method, electroporation method, Liposome-mediated transfection and gene bombardment may be used, but is not limited thereto. When using microorganisms such as E. coli, productivity is higher than that of animal cells, but is not suitable for the production of intact Ig-type antibodies due to glycosylation problems. However, antigen-binding fragments such as Fab and Fv Can be used for production.
상기 형질 전환된 숙주 세포를 선별하는 방법은 선택 표지에 의해 발현되는 표현형을 이용하여, 당업계에 널리 알려진 방법에 따라 용이하게 실시할 수 있다. 예를 들어, 상기 선택 표지가 특정 항생제 내성 유전자인 경우에는, 상기 항생제가 함유된 배지에서 형질전환체를 배양함으로써 형질전환체를 용이하게 선별할 수 있다.The method of selecting the transformed host cell can be easily carried out according to methods well known in the art using a phenotype expressed by a selection label. For example, when the selection marker is a specific antibiotic resistance gene, the transformant can be easily selected by culturing the transformant in a medium containing the antibiotic.
또한, 본 발명의 일 양상은 상기 살아있는 세포내부로 침투하여 세포질에 분포하는 완전한 이뮤노글로불린 형태의 항체를 이용하여 세포질 침투를 통해 세포질에 위치하고 있는 활성화된(GTP가 결합된) 종양관련인자 RAS에 특이적 결합 및 활성을 저해하는 완전한 이뮤노글로불린 형태의 항체의 제조방법을 제공할 수 있다. In addition, an aspect of the present invention is to the activated (GTP coupled) tumor-associated factor RAS located in the cytoplasm through cytoplasmic infiltration using the antibody of the complete immunoglobulin form that penetrates into the living cell and distributed in the cytoplasm It is possible to provide a method for preparing a complete immunoglobulin form of antibody that inhibits specific binding and activity.
상기 일 양상 구체예에 있어서, 활성화된(GTP가 결합된) RAS 특이적 결합능을 가지고 있는 중쇄가변영역(VH)을 이용한 동물 세포내부로 침투하고 세포질에 분포하여 세포질에 위치하고 있는 활성화된(GTP가 결합된) RAS와 특이적 결합하는 완전 이뮤노글로불린 형태의 항체는 하기와 같은 방법으로 제조될 수 있다.In one embodiment of the above aspect, the activated (GTP conjugated) GRA which is penetrated into the animal cell using the heavy chain variable region (VH) having RAS specific binding ability and distributed in the cytoplasm and located in the cytoplasm Antibodies in the form of fully immunoglobulin that specifically bind to bound RAS can be prepared by the following method.
(1) 활성화된(GTP가 결합된) RAS와 특이적 결합하는 인간 중쇄가변영역(VH)과 중쇄불변영역(CH1-hinge-CH2-CH3)이 포함된 핵산(nucleic acides)를 클로닝한 중쇄 발현 벡터를 제조하는 단계;(1) Heavy chain expression by cloning of nucleic acid containing human heavy chain variable region (VH) and heavy chain constant region (CH1-hinge-CH2-CH3) that specifically bind to activated (GTP-bound) RAS Preparing a vector;
(2) 상기 제조된 중쇄 발현 백터와 세포 침투 경쇄 발현벡터를 단백질 발현용 동물세포에 동시 형질전환하여 살아있는 세포내부로 침투 및 세포질 분포하여 활성화된(GTP가 결합된) RAS를 특이적 인지하는 완전 이뮤노글로불린 형태의 항체를 발현하는 단계; 및(2) The co-transformation of the prepared heavy chain expression vector and the cell infiltration light chain expression vector into animal cells for protein expression infiltrate into living cells and cytoplasmic distribution to completely recognize specific activated RAS (GTP-coupled). Expressing an antibody in an immunoglobulin form; And
(2) 상기 발현된 활성화된(GTP가 결합된) RAS를 특이적 인지하는 세포질 침투능을 가지는 완전한 이뮤노글로불린 형태의 항체를 정제, 회수하는 단계.(2) Purifying and recovering an antibody of a complete immunoglobulin form having cytoplasmic penetrating ability to specifically recognize the expressed activated (GTP bound) RAS.