WO2015181507A1 - Nouveaux composes de type 5-acylsulfanyl-histidine en tant que precurseurs des 5-sulfanylhistidines correspondantes et de leurs disulfures - Google Patents
Nouveaux composes de type 5-acylsulfanyl-histidine en tant que precurseurs des 5-sulfanylhistidines correspondantes et de leurs disulfures Download PDFInfo
- Publication number
- WO2015181507A1 WO2015181507A1 PCT/FR2015/051416 FR2015051416W WO2015181507A1 WO 2015181507 A1 WO2015181507 A1 WO 2015181507A1 FR 2015051416 W FR2015051416 W FR 2015051416W WO 2015181507 A1 WO2015181507 A1 WO 2015181507A1
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- compound
- histidine
- acid
- derivatives
- acetylsulfanyl
- Prior art date
Links
- 0 C*(*)(*)C(CC(*C*1(*)*)=C1NC(C)=O)C(O)=O Chemical compound C*(*)(*)C(CC(*C*1(*)*)=C1NC(C)=O)C(O)=O 0.000 description 2
- ARAQDIDMFIPYQR-YFKPBYRVSA-N CC(Sc1c(C[C@@H](C(O)=O)N)[nH]cn1)=O Chemical compound CC(Sc1c(C[C@@H](C(O)=O)N)[nH]cn1)=O ARAQDIDMFIPYQR-YFKPBYRVSA-N 0.000 description 1
- YIWFXZNIBQBFHR-LURJTMIESA-N NCC(N[C@@H](Cc1cnc[nH]1)C(O)=O)=O Chemical compound NCC(N[C@@H](Cc1cnc[nH]1)C(O)=O)=O YIWFXZNIBQBFHR-LURJTMIESA-N 0.000 description 1
Classifications
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D403/00—Heterocyclic compounds containing two or more hetero rings, having nitrogen atoms as the only ring hetero atoms, not provided for by group C07D401/00
- C07D403/02—Heterocyclic compounds containing two or more hetero rings, having nitrogen atoms as the only ring hetero atoms, not provided for by group C07D401/00 containing two hetero rings
- C07D403/12—Heterocyclic compounds containing two or more hetero rings, having nitrogen atoms as the only ring hetero atoms, not provided for by group C07D401/00 containing two hetero rings linked by a chain containing hetero atoms as chain links
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D233/00—Heterocyclic compounds containing 1,3-diazole or hydrogenated 1,3-diazole rings, not condensed with other rings
- C07D233/54—Heterocyclic compounds containing 1,3-diazole or hydrogenated 1,3-diazole rings, not condensed with other rings having two double bonds between ring members or between ring members and non-ring members
- C07D233/66—Heterocyclic compounds containing 1,3-diazole or hydrogenated 1,3-diazole rings, not condensed with other rings having two double bonds between ring members or between ring members and non-ring members with hetero atoms or with carbon atoms having three bonds to hetero atoms with at the most one bond to halogen, e.g. ester or nitrile radicals, directly attached to ring carbon atoms
- C07D233/84—Sulfur atoms
Definitions
- the present invention relates to:
- the present invention relates to novel 5-acylsulfanylhistidine compounds and their derivatives, as well as their methods of preparation and their use as precursors of the corresponding 5-sulfanylthistides and their disulfides. More particularly, this invention relates to the synthesis of novel 5-acylsulfanyl-histidine compounds and their derivatives, their salts as direct precursors of the corresponding 5-sulfanylthistides and their disulfides.
- the recent IUPAC "suifanyl” nomenclature for the "-SH” group is applied for the compounds described in the invention instead of the different terms previously used in the literature such as "thiohistidine",
- a first example is constituted by the group of adenochromines
- the second approach consisted of a nucleophilic substitution of a 5-bromoimidazole nucleus activated by an electron-withdrawing group carboxaldehyde CHO (Ohba M., Nishimura Y., Kato M. and Fujii T. Tetrahedron 1999, 55, 4999-5016) in the context of the synthesis of imbricatine.
- a 5-bromoimidazole nucleus activated by an electron-withdrawing group carboxaldehyde CHO (Ohba M., Nishimura Y., Kato M. and Fujii T. Tetrahedron 1999, 55, 4999-5016) in the context of the synthesis of imbricatine.
- Ovothiols A, B and C see De Luna et al., J. Phys. Chemistry, 2013, DOI: 10.1021 / jp402514w;
- the 5-acylsulfanyl-histidine compounds and their derivatives would be very good precursors of 5-sulfanylhistidine and its derivatives. Since these 5-acylsulfanyl-histidine compounds and their derivatives are not known, it would therefore be necessary to have a synthesis method which makes it possible to directly introduce an acylsulfanyl group at the 5-position of a histidine. Such a method, to our knowledge, has never been described so far. This new method of direct introduction of an acylsulfanyl group at the 5-position of histidine or of one of its derivatives would be all the more interesting since it could be carried out without a protective group and in water as a solvent. reaction.
- One of the aims of the present invention is therefore to provide new compounds of the 5-acylsulfanyl-histidine type and their derivatives which may be precursors of the corresponding 5-sulfanylhistidines and their disulfides.
- Another object of the present invention is a process for the preparation of these novel 5-acylsulfanyl-histidine compounds and their derivatives using a new method for the direct introduction of an acylsulfanyl group at the 5-position of a histidine or the one of its derivatives without a protective group and in water as a solvent.
- Another object of the present invention is the use of these novel 5-acylsulfanyl-histidine compounds and their derivatives as precursors of the corresponding 5-sulfanylhistidines and their disulfides.
- the present invention therefore aims:
- the subject of the present invention is new 5-acylsulfanyl-histidine compounds and their derivatives corresponding to the following general formula (I):
- R 1 H, alkyl, in particular CH 3 ;
- R 5 alkyl, in particular methyl, phenyl
- the invention encompasses all stereoisomers, diastereoisomers and enantiomers, especially at the carbon atom carrying the COOH group, taken alone or as a mixture.
- mineral acids such as hydrochloric, hydrobromic, hydroiodic, sulfuric, tartaric, phosphoric or with organic acids such as formic acid, acetic acid, trifluoroacetic acid, propionic, benzoic, maleic, fumaric, succinic, citric, oxalic, glyoxylic, aspartic, alkanesulfonic such as methanesulfonic, trifluoromethanesulfonic, ethanesulfonic, arylsulphonic acids such as benzene- and para-toluenesulfonic acids .
- mineral acids such as hydrochloric, hydrobromic, hydroiodic, sulfuric, tartaric, phosphoric
- organic acids such as formic acid, acetic acid, trifluoroacetic acid, propionic, benzoic, maleic, fumaric, succinic, citric, oxalic, glyoxylic, aspartic, alkanesulfonic such
- an alkyl radical is understood to mean a group comprising 1 to 6 linear or cyclic carbon atoms, optionally branched,
- substituted alkyl radical means an alkyl group substituted with one or more fluorine atoms, or substituted with an alkenyl group containing one or more carbon-carbon double bonds, or substituted with one or more OH, SH or NH 2 or COOH, as well as their enantiomers, and their diastereoisomers.
- aryl radical it is understood a phenyl group optionally fluorinated or polyfluorinated, and optionally comprising one or more OH or SH or NH 2 or COOH functions
- ⁇ -amino-acyl radical it is understood the acyl radical of any amino-proteogenic acid, that is to say any amino-acid used in the composition of proteins found in the plant, animal world, including man .
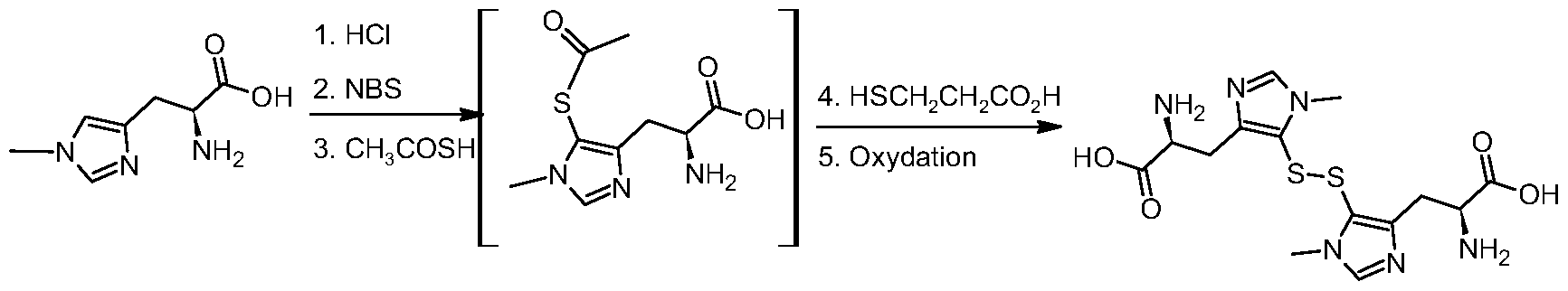
- the subject of the invention is also a process A for the preparation of the new compounds of the 5-acylsulfanyl-histidine type and their derivatives of general formula (I), explained in the attached FIG. 1, and characterized in that it includes the following steps:
- the halogenium ion generating agent X + may be:
- the polar protic solvent may be water or an aqueous solution.
- the carbothioic acid sulfur-containing reagent may be, for example, thioacetic acid; or thiobenzoic acid, or mixtures thereof.
- the sulfur-containing reagent of the carbothioic acid salt type may be, for example, potassium thioacetate, optionally in admixture with a carbothioic acid mentioned above.
- the temperature will be between 0-5 ° C.
- the subject of the present invention is the use of the abovementioned 5-acylsulfanyl compounds of formula (I) or of its derivatives, for the preparation of corresponding 5-sulfanyl-histidine compounds and their disulfides described below.
- the invention covers novel 5-sulfanylhistidine compounds and their derivatives corresponding to the following general formula (II):
- R 1 H then R 2 , R 3 and R 4 can not be simultaneously H.
- the invention encompasses all stereoisomers, diastereoisomers and enantiomers, in particular at the carbon atom carrying the COOH group, as well as all the corresponding disulfides, taken alone or as a mixture.
- mineral acids such as hydrochloric, hydrobromic, hydroiodic, sulfuric, tartaric, phosphoric or with organic acids such as formic acid, acetic acid, trifluoroacetic acid, propionic acid, benzoic, maleic, fumaric, succinic, citric, oxalic, glyoxylic, aspartic, alkanesulfonic, such as methanesulfonic, trifluoromethanesulfonic, ethanesulfonic, arylsulphonic acids such as benzene and paratoluene sulfonic acids.
- mineral acids such as hydrochloric, hydrobromic, hydroiodic, sulfuric, tartaric, phosphoric
- organic acids such as formic acid, acetic acid, trifluoroacetic acid, propionic acid
- benzoic maleic, fumaric, succinic, citric, oxalic, glyoxylic, aspartic, alkanesulfonic
- an alkyl radical is understood to mean a group comprising 1 to 6 linear or cyclic carbon atoms, optionally branched,
- substituted alkyl radical is understood to mean an alkyl group substituted with one or more fluorine atoms, or substituted by an alkenyl group containing one or more carbon-carbon double bonds, or substituted by one or more OH or SH or NH 2 or COOH functions, as well as their enantiomers, and their diastereoisomers.
- aryl radical it is understood a phenyl group optionally fluorinated or polyfluorinated, and optionally comprising one or more OH or SH or NH 2 or COOH functions
- acyl radical of any amino acid proteogenic, ie any amino acid involved in the composition of proteins found in the plant world, animal, including man.
- the new compounds of the 5-sulfanylhistidine type and their derivatives corresponding to the general formula (II), as well as their disulfides, could prove to be nutritional, cosmetic or medicinal active ingredients.
- the subject of the invention is also a process B for the preparation of the compounds of the 5- sulfanylhistidine type and of their derivatives of general formula (II) obtained from the compounds of the 5-acylsulfanyl-histidine type and of their derivatives of general formula (I) described in process A above, and characterized in that it comprises the following steps:
- the polar protic solvent may be water or an aqueous solution.
- the thiol may be, for example, mercaptopropionic acid or dithiotreitol, or mixtures thereof.
- the temperature may be between 20 and 130 ° C.
- the Applicant demonstrates the ability of the compounds of general formula (I) to be precursors of 5-sulfanylhistidine compounds and their derivatives of general formula (II) after hydrolysis.
- the subject of the invention is also a process C for the preparation of disulfides of 5-sulfanylhistidines and their derivatives:
- the Applicant demonstrates the ability of 5-acylsulfanyl-histidine compounds of general formula (I) to be disulfide precursors of 5-sulfanylhistidines and its derivatives after hydrolysis and oxidation.
- the invention also relates to a "one-pot" process D for the preparation of the 5-sulfanylhistidine derivatives and their corresponding disulfides from the corresponding histidine derivatives, by combining the processes A with B or with C, and characterized in that it comprises the following steps:
- the invention comprises 4 figures.
- FIG. 2 Representative spectrum ( 1 H NMR, 400 MHz) of the reaction mixture obtained in Example 1, preparation of L-5-acetylsulfanyl-histidine (Compound 1)
- FIG. 3 Representative spectrum ( 1 H NMR, 400 MHz) of the reaction mixture obtained in Example 3, preparation of L-5-acetylsulfanyl-O, N (dimethyl) -histidine (Compound 2)
- FIG. 4 Representative spectrum ( 1 H NMR, 400MHz) of the reaction mixture obtained in Example 5, preparation of L-5-acetylsulfanyl-aN, N, N (trimethyl) -histidine (Compound 3)
- the temperature is either the ambient temperature or a given temperature in degrees Celsius
- the pressure is the atmospheric pressure, unless otherwise indicated.
- the reagents used are commercially available from international suppliers such as SAF (France), Alfa Aesar, Fisher Scientific, TCI Europe, Bachem (Switzerland, AKOS (Germany) except the following compounds: N-methyl-histidine hydrochloride, N, N- Dimethyl Histidine Hydrochloride Hydrate and L-Hercynin which have been prepared according to the protocols cited.
- the 1 H-NMR analyzes were recorded at 400 MHz or 300 MHz in D 2 0 or a mixture D 2 0 / DC1, using the signal of HOD (4.79 ppm) as internal reference. The chemical shifts are noted in ppm, and the multiplicity of the signals indicated by the following symbols: s (singlet), d (doublet), t (triplet), q (quartet), and m (multiplet). The coupling constants are noted in hertz (Hz). 13 C-NMR analyzes are recorded at 75 MHz in D 2 0 or D 2 0 / DC 1. Analyses of mass are obtained by chemical ionization at atmospheric pressure (APCI-MS). Melting points were measured with an apparatus from Stuart Scientific.
- APCI-MS atmospheric pressure
- HPLC analyzes were performed on an Acquity device (Waters), using two types of columns: A. Kromasil Diol 250x4.6 column (5pm).
- the mobile phase used is a mixture of solvent A (10/90 H 2 O / acetonitrile + 0.05% TFA) and solvent B (50/50 H 2 O / acetonitrile + 0.05% TFA), with a gradient varying in 10 minutes from 90% A to 100% B and a flow rate of 1.2 ml / min.
- the mobile phase used is a mixture of solvent A (100% H 2 O + 0.2% HCOOH) and solvent B (100% acetonitrile + 0.2% HCOOH), with a gradient varying in 8 minutes from 100% A to 40%. % A and a flow rate of 1 ml / min.
- the detection is carried out with a universal ELSD detector (Sedere).
- the hydrochloride of L-histidine monohydrate (52.93 g, 250 mmol, 1 eq) is dissolved in 1.5 l of deionized water, and the solution is cooled to 0 ° C in 30 minutes. With vigorous stirring the bromine (16.7 ml, 51.93 g, 325 mmol, 1.3 eq) was added dropwise very rapidly. The solution turns red. The thioacetic acid (73.3 ml, 78.46 g, 1 mole, 4 eq.) Is added very rapidly: the solution becomes discolored immediately and changes from red to light yellow. Stirring is vigorously maintained at 0 ° C for 1 h.
- the L-histidine hydrochloride monohydrate (10.48 g, 50 mmol, 1 eq) is dissolved in 300 ml of deionized water containing a 37% concentrated hydrochloric acid solution (4.17 ml, 4.92 g). 50 mmol, 1 eq) and the solution is cooled to 0 ° C. The agitation is kept very strong. N-bromosuccinimide (11.56 g, 65 mmol, 1.3 eq) is added in one portion: the mixture becomes clear orange after 30 seconds. The temperature rises to 1 ° C.
- Compound 1 is obtained with a reaction yield of 75 mol% as calculated from the 1 H NMR spectrogram (in the reaction mixture).
- Compound 2 is obtained with a reaction yield of 70 mol% as calculated from the NMR-H 1 spectrogram.
- Compound 2 is purified on a silica column using an ethyl acetate / ethanol gradient followed by elution with water.
- a low intensity singlet corresponding to the hydrolysed product (compound 18b) is detected at 8.33 ppm.
- Example 5 Preparation of L-5-acetylsulfanyl-a, N, N, N (trimethyl) -histidine (Compound 3) by activation with N-bromosuccinimide and reaction with thioacetic acid
- Example 7 Preparation of L-5-acetylsulfanyl- (X, N (glycinyl) -histidine (Compound 4) by activation with N-bromosuccioacetic acid
- N (glycinyl) -histidine 212 mg, 1 mmol, 1 eq. Is dissolved in 7 ml of demineralized water and 1 ml of acetonitrile. Then a solution of concentrated hydrochloric acid at 37% (170 ⁇ l, 2 mmol, 2 eq.) Is added and the solution is cooled to 0 ° C. With vigorous stirring, N-bromosuccinimide (230 mg, 1.3 mmol, 1.3 eq) was added. After 3 minutes, the thioacetic acid (370 ⁇ l, 5.0 mmol, 5 eq) is added very rapidly. Stirring is maintained at 0 ° C for 30 minutes.
- a singlet corresponding to the excess of the thioacetic acid is detected at 2.48 ppm, a singlet at 2.78 ppm corresponding to the succinimide as well as low intensity signals corresponding to the secondary products, such as acetic acid detected at 2,0ppm.
- Compound 5 is obtained with a reaction yield of 65 mol% as calculated from the NMR spectrogram.
- the ethanol signals are detected at 18ppm and 3.65ppm.
- 1-methyl-hercynin (430 mg, 2 mmol, 1 eq) is dissolved in 15 ml of deionized water. Concentrated 37% hydrochloric acid (170 ⁇ l, 2 mmol, ⁇ 1 eq.) Is added and the solution is cooled to 0 ° C. With vigorous stirring, N-bromosuccinimide (465 mg, 2.6 mmol, 1.3 eq) was added. After 3 minutes, the thioacetic acid (740 ⁇ l, 10 mmol, 5 eq) is added very rapidly. Stirring is maintained at 0 ° C for 30 minutes.
- a singlet corresponding to the excess of the thioacetic acid is detected at 2.48 ppm, a singlet at 2.78 ppm corresponding to the succinimide as well as low intensity signals corresponding to the secondary products, such as acetic acid detected at 2,0ppm.
- Example 10 Preparation of L-5-acetylsulfanyl- ⁇ -N (L-alanyl) -histidine (Compound 7) by activation with N-bromosuccinimide and reaction with thioacetic acid
- the ⁇ , N (L-alanyl) -histidine (500 mg, 2.2 mmol, 1 eq) is dissolved in 15 ml of deionized water containing 37% concentrated hydrochloric acid solution (370 ⁇ , 4%). 4 mmol, 2 eq.) And the solution is cooled to 0 ° C. N-bromosuccinimide (510 mg, 2.9 mmol, 1.3 eq) is added in one portion: the mixture becomes clear orange after 30 seconds. After 3 minutes, the thioacetic acid (820 ⁇ l, 11.0 mmol, 5 eq) is added very rapidly. Stirring is vigorously maintained at 0 ° C for 30 minutes.
- Compound 7 is obtained with a reaction yield of 65 mol% as calculated from the NMR-H 1 spectrogram of a sample.
- reaction mixture is washed with 2x25 ml of ethyl acetate and the compound is then purified on a silica column (2/2/1 ethyl acetate / ethanol / water) to give 7 (410 mg, 54.degree. %, purity 88%) as a clear oil.
- a singlet corresponding to succinimide is detected at 2.68 ppm.
- N (pentanoyl) -histidine 450 mg, 1.43 mmol, 1 eq.
- 10 ml of demineralized water containing a 37% concentrated hydrochloric acid solution 120 ⁇ l, 1.43 ml). mmol, 1 eq.
- N-bromosuccinimide 330 mg, 1.86 mmol, 1.3 eq
- the thioacetic acid 530 ⁇ l, 7.15 mmol, 5 eq
- Stirring is vigorously maintained at 0 ° C for 30 minutes.
- Compound 8 is obtained with a reaction yield of 67 mol% as calculated from the NMR-H 1 spectrogram of a sample.
- the product is purified on a silica column (mixture 90% ethyl acetate / ethanol 3/1 and 10% water).
- the 5-acetylsulfanyl- ⁇ , N (pentanoyl) -histidine (compound 8) is obtained in the form of a transparent oil (320 mg, 64%, purity 90%).
- the 5-acylsulfanyl compounds are prepared in situ and then hydrolyzed directly, while stirring the reaction medium, preferably by heating.
- reaction medium The presence of a thiol, such as mercaptopropionic acid or dithiothreitol, is beneficial for the easy isolation of 5-sulfanylthistidine derivatives, but it is in no way necessary for the hydrolysis itself, as demonstrated in the literature.
- Example 22 "One pot" preparation of L-5-sulfanylhistidine via in situ preparation of 5-acetylsulfanylhistidine followed by hydrolysis (Compound 18)
- Example 23 "One pot" preparation of D-5-sulfanylhistidine via in-situ preparation of 5-acetylsulfanyl-histidine followed by hydrolysis (Compound 19)
- D-Histidine (3.92 g, 25 mmol, 1 eq) is dissolved in 150 ml deionized water and 37% concentrated hydrochloric acid solution (4.17 ml, 4.92 g, 50 mmol). 2 eq.), Then the solution is cooled to 0 ° C. The agitation is kept very strong. N-bromosuccinimide (5.78 g, 32.5 mmol, 1.3 eq) is added all at once: the solution becomes clear orange Thioacetic acid (7.33 ml, 7.85 g, 200 ml) mmol, 4 eq) is added all at once, stirring is vigorously maintained at 0 ° C.
- Example 24 "one pot” preparation of D, L-5-sulfanylhistidine via in situ preparation of 5-acylsulfanylhistidine followed by hydrolysis (Compound 20)
- the 3-mercaptopropionic acid (8.0 ml, 9.65 g, 90 mmol, 6 eq) is added and the solution is then heated at 100 ° C for 18 h. A precipitate corresponding to the disulfide of thioacetic acid and mercaptopropionic acid is removed by filtration. The filtrate is washed twice with 100 ml of ethyl acetate. After neutralization and crystallization in the presence of dithiothreitol (233 mg, 1.5 mmol, 0.1 eq.), 650 mg of D, L-5-sulfanylhistidine (Compound 20) (23%, 29% relative to the amount of intermediate SAc) are obtained in the form of a white solid.
- the L-histidine hydrochloride monohydrate (14.82 g, 70 mmol, 1 eq) is dissolved in 126 ml of deionized water, and the solution is cooled to 0 ° C. With vigorous stirring, the bromine (4.32 ml, 13.42 g, 84 mmol, 1.2 eq.) Is added dropwise very rapidly. The solution becomes red. The thioacetic acid (18.0 ml, 19.2 g, 245 mmol, 3.5 eq) is added very rapidly. Stirring is vigorously maintained at 0 ° C for 20 minutes.
- the slightly yellow solution obtained is extracted twice with 120 ml of ethyl acetate. After hydrolysis under heat, oxidation and purification on the resin DOWEX 50WX2-400 the hydrochloride hydrate of the disulfide of the L-5-sulfanyl-N, N (dimethyl) -histidine (compound 24x4HClxH2O, 1.2 g, 41%) is obtained in the form of a beige powder.
- Compound 25 Compound 2 is prepared and purified by column as described in Example 3, using an ethyl acetate / ethanol gradient followed by elution with water. The aqueous fraction containing the pure compound 2 is placed in a water bath at 40 ° C., and heated with stirring for 8 hours. Samples are taken every 60 minutes, and the mixture analyzed by HPLC.
- the 3-mercaptopropionic acid (11.07 ml, 13.4 g, 125 mmol, 5 eq) is added to the aqueous phase and then the solution is heated at 130 ° C for 3h. After extraction, neutralization and crystallization in the presence of dithiothreitol (1.95 g (12.5 mmol, 0.5 eq)) L-5- sulfanyl - ⁇ , N, N, N (trimethyl) -histidine (Compound 26) (2.22 g, 38%, 58% relative to the amount of the intermediate SAc) is obtained in the form of a white powder (to be stored under an inert atmosphere).
- Compound 3 is prepared and purified by column as described in Example 5. 100 mg (0.33 mmol, 1 eq) of compound 3 are solubilized in 2.4 mL of D20. 172 mg of 3-mercaptopropionic acid (142 ⁇ l, 5 equivalents) are added, and the solution is heated to 40 ° C. Conversion is followed by 1 H-NMR and HPLC-ELSD. The hydrolysis yield of compound 3 is 90% after 3h (followed by
- Compound 3 is prepared and purified by column as described in Example 5. 170 mg (0.6 mmol) of compound 3 are solubilized in 10 mL of water, and the solution is heated to 90 ° C in air during 7h. The conversion is followed by HPLC. The hydrolysis of compound 3 is complete after 7h. The solution is evaporated to dryness. The residue is taken up in a mixture of 5 mL of methanol and 93 mg (0.6 mmol) of dithiothreitol. After stirring for 4 hours under an inert atmosphere, 2 ml of ethanol are added. A precipitate forms immediately, which is filtered and washed with ethanol (2x2mL) and then with ethyl ether (2x2mL). After drying, 104 mg (72%) of L-5-sulfanyl-N, N, N (trimethyl) histidine are obtained in the form of a beige powder.
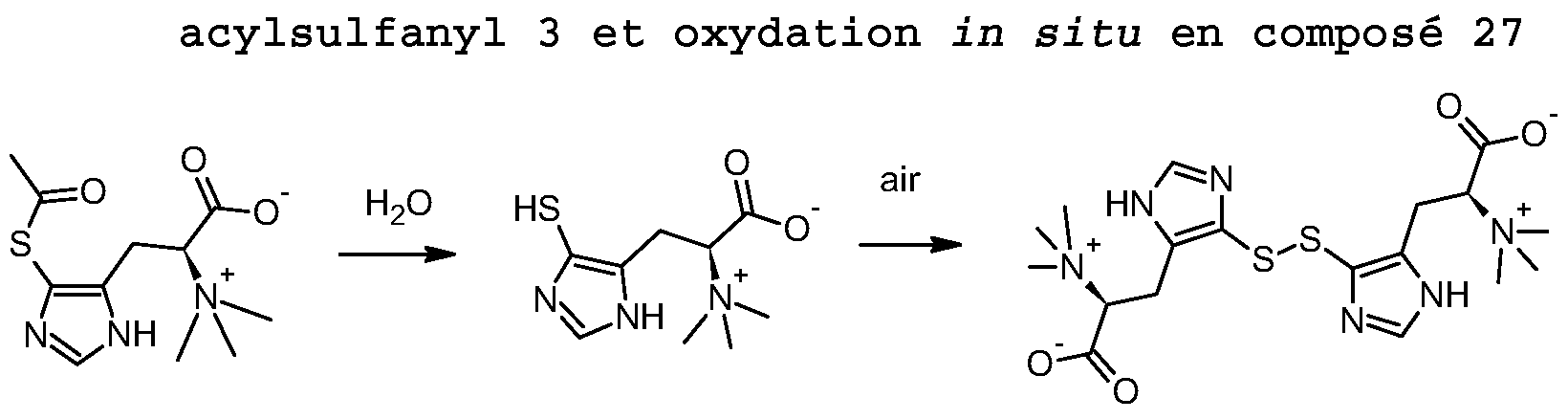
- L-5-sulfanyl-a, N, N, N (trimethyl) -histidine (Compound 26, 300 mg, 1.29 mmol, 1 eq) is dissolved in 50 ml of demineralized water. The colorless solution is stirred at room temperature for 4 days. After filtration and lyophilization of the filtrate, the disulfide of L-5-sulfanyl- ⁇ , ⁇ , ⁇ , ⁇ (trimethyl) -histidine (Compound 27) (263 mg, 89%) is obtained in the form of a yellow powder.
- Compound 3 is prepared and purified by column as described in Example 5, using an ethyl acetate / ethanol gradient followed by elution with water.
- the aqueous fraction containing pure compound 3 is placed in a water bath at 40 ° C, and heated with stirring for two days. Samples are taken every hour, and the mixture analyzed by HPLC.
- Compound 3 is prepared and purified by column as described in Example 5. 30 mg of compound 3 are solubilized in 600 ⁇ L of D 2 0, the solution transferred to an NMR tube, which is kept at room temperature. The conversion is monitored by 1 H-NMR. The hydrolysis of compound 3 is almost complete after 2 days, and a mixture containing disulfide 27 and thiol 26 ( ⁇ 3: 1) is obtained.
- the 3-mercaptopropionic acid (5.26 ml, 6.36 g, 60 mmol, 6 eq) is added and the solution is heated at 80 ° C overnight. The solution is cooled to ambient temperature and then extracted with 4 times 50 ml of ethyl acetate. The aqueous phase is purified on silica to yield L-5-sulfanyl- ⁇ (acetyl) histidine disulfide hydrochloride (compound 28) as an orange oil (520 mg, 17%, 36% relative to the amount of intermediate SAc).
- the disulfide hydrochloride of L-5-sulfanyl- ⁇ , N (acetyl) -histidine (Compound 28) (520 mg, 834 pmol, 1 eq.) Is dissolved in 50 ml of water and the pH of the solution brown is adjusted to 4.5 by adding NH 4 OH. 3-Mercaptopropionic acid (4.38 ml, 5.31 g, 50 mmol, 5 eq) is added. The solution is heated at 70 ° C for 2h. The solution is extracted with 4 times 50 ml of ethyl acetate. The aqueous phase is evaporated to dryness to give L-5-sulfanyl- ⁇ , N (acetyl) -histidine (Compound 29) (390 mg, 86%) as a beige solid.
- Example 30 "One pot” preparation of L-5-sulfanyl carnosine via in-situ preparation of 5-acylsulfanylhistidine followed by hydrolysis (Compound 30)
- 1-methyl-L-histidine (0.84 g, 5 mmol, 1 eq.) Is dissolved in 35 ml of demineralized water and a 37% solution of concentrated hydrochloric acid (835 ⁇ l (10 mmol, 2 eq. ) is added, then the solution is cooled to 1 ° C. The stirring is maintained very strong N-bromosuccinimide (1.17 g, 6.5 mmol, 1.3 eq.) is added rapidly. minutes, the thioacetic acid (2.57 ml, 2.74 g, 35 mmol, 7 eq) is added very rapidly, stirring is vigorous at 0 ° C. for 30 minutes, the solution is extracted with 40 ml.
- L-5-sulfanyl-1-methyl-histidine disulfide (Compound 25) (427 mg, 0.52 mmol, 1 eq) is suspended in 25 ml of methanol. The medium is heated to 50 ° C. and then dithiothreitol (299 mg, 1.92 mmol, 2 eq.) Is added. After stirring for 1 h at room temperature and precipitation with ethyl ether, L-5-sulfanyl-1-methyl-Histidine (Iso-ovothiol A, Compound 32) (295 mg, 69%) is obtained as a a slightly greyish powder.
- Example 32 Preparation of L-5-sulfanyl- (X, N, N (dimethyl) -1-methyl-histidine disulfide by hydrolysis of the 5-acetylsulfanyl- ⁇ , N, N (dimethyl) -1-methyl- Histidine followed by oxidation in air (Compound 33)
- Compound 5 is prepared and purified by column as described in Example 8. 180 mg (0.63 mmol, 1 eq) of compound 5 are solubilized in 20 ml of water. The clear solution is stirred in the presence of oxygen for 20 hours at room temperature. After lyophilization, the disulfide L-5-sulfanyl-a, N, N (dimethyl) -1-methyl-histidine (Compound 33, 98%) is obtained in the form of a greenish amorphous solid.
- Example 33 "One pot" preparation of L-5-sulfanyl- ⁇ , N, N, N (trimethyl) -1-methyl-histidine disulfide (Compound 34 dihydrochloride) via in-situ preparation of L-5- acetylsulfanyl- ⁇ , N, N (trimethyl) -1-methyl-histidine followed by hydrolysis and oxidation
- 1-methyl-hercynin (510 mg, 2 mmol, 1 eq.) Is dissolved in 15 ml of deionized water containing 37% concentrated hydrochloric acid solution (170 ⁇ l, 2 mmol, 1 eq.), Then the solution is cooled to 0 ° C. The agitation is kept very strong. N-bromosuccinimide (465 mg, 2.6 mmol, 1.3 eq) is added rapidly. After 3 minutes, the thioacetic acid (740 ⁇ l, 10 mmol, 5 eq) is added very rapidly. Stirring is vigorously maintained at 0 ° C for 30 minutes.
- the mixture is extracted with 2x20 ml of ethyl acetate and then diluted in 160 ml of ethyl acetate / ethanol (3/1) for purification on a silica column (ethyl acetate / ethanol / water 2 / 2/1).
- the slightly pink oil obtained is oxidized with dimethyl sulfoxide (140 ⁇ l, 2 mmol, 1 eq.) In a solution of glacial acetic acid. The solution is heated for one hour at 80 ° C.
- the disulfide dihydrochloride of L-5-sulfanyl- ⁇ , ⁇ , ⁇ , ⁇ , (trimethyl) -1-methyl-histidine (compound 34) is obtained after purification on a silica column (acetate of ethyl / ethanol / water 2/2/1 then elution with 0.5M hydrochloric acid) as a slightly yellow oil (110 mg, 10%).
- Compound 7 is prepared and purified by column as described in Example 10. 340 mg (1 mmol, 1 eq) of compound 7 are solubilized in 20 ml of water. The clear solution is shaken in the absence of oxygen for 6 days at room temperature. After evaporation to dryness, L-5-sulfanyl- ⁇ , N (L-alanyl) -histidine (Compound 35, 92%) is obtained in the form of a beige amorphous solid.
Landscapes
- Chemical & Material Sciences (AREA)
- Organic Chemistry (AREA)
- Pharmaceuticals Containing Other Organic And Inorganic Compounds (AREA)
- Plural Heterocyclic Compounds (AREA)
Abstract
Description
Claims
Priority Applications (6)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| CN201580040441.7A CN106573895B (zh) | 2014-05-30 | 2015-05-29 | 作为相应的5-硫烷基组氨酸和其二硫化物的前体的新型5-酰基硫烷基-组氨酸化合物 |
| US15/314,628 US9926300B2 (en) | 2014-05-30 | 2015-05-29 | 5-acylsulfanyl-histidine compounds as precursors of the corresponding 5-sulfanylhistidines and their disulfides |
| CA2949334A CA2949334A1 (fr) | 2014-05-30 | 2015-05-29 | Composes de type 5-acylsulfanyl-histidine en tant que precurseurs des 5-sulfanylhistidines correspondantes et de leurs disulfures |
| EP15735988.6A EP3148973A1 (fr) | 2014-05-30 | 2015-05-29 | Nouveaux composes de type 5-acylsulfanyl-histidine en tant que precurseurs des 5-sulfanylhistidines correspondantes et de leurs disulfures |
| US15/891,433 US20180258077A1 (en) | 2014-05-30 | 2018-02-08 | 5-sulfanylhistidines and their disulfides and method of preparation thereof |
| US16/580,375 US20200017479A1 (en) | 2014-05-30 | 2019-09-24 | Novel 5-acylsulfanyl-histidine compounds as precursors of the corresponding 5-sulfanylhistidines and their disulfides |
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| FR1454935A FR3021655B1 (fr) | 2014-05-30 | 2014-05-30 | Nouveaux composes de type 5-acylsulfanyl-histidine en tant que precurseurs des 5-sulfanylhistidines correspondantes et de leurs disulfures |
| FR1454935 | 2014-05-30 |
Related Child Applications (2)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| US15/314,628 A-371-Of-International US9926300B2 (en) | 2014-05-30 | 2015-05-29 | 5-acylsulfanyl-histidine compounds as precursors of the corresponding 5-sulfanylhistidines and their disulfides |
| US15/891,433 Division US20180258077A1 (en) | 2014-05-30 | 2018-02-08 | 5-sulfanylhistidines and their disulfides and method of preparation thereof |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| WO2015181507A1 true WO2015181507A1 (fr) | 2015-12-03 |
Family
ID=51352623
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/FR2015/051416 WO2015181507A1 (fr) | 2014-05-30 | 2015-05-29 | Nouveaux composes de type 5-acylsulfanyl-histidine en tant que precurseurs des 5-sulfanylhistidines correspondantes et de leurs disulfures |
Country Status (6)
| Country | Link |
|---|---|
| US (2) | US9926300B2 (fr) |
| EP (1) | EP3148973A1 (fr) |
| CN (2) | CN112194632A (fr) |
| CA (1) | CA2949334A1 (fr) |
| FR (1) | FR3021655B1 (fr) |
| WO (1) | WO2015181507A1 (fr) |
Families Citing this family (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| IT202100026639A1 (it) | 2021-10-18 | 2023-04-18 | Stazione Zoologica Anton Dohrn | Metodologia di sistema di ingegneria genetica a base di diatomee per la produzione ecosostenibile di ovotioli |
Family Cites Families (3)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US4898878A (en) * | 1987-10-02 | 1990-02-06 | The Board Of Regents Of The University Of Washington | Antioxidant thiohistidine compounds |
| FR2950889B1 (fr) | 2009-10-06 | 2011-11-18 | Tetrahedron | Procede de synthese de l'ergothioneine et analogues |
| FR2950890B1 (fr) | 2009-10-06 | 2011-12-30 | Tetrahedron | Procede de synthese de la 2-thiohistidine et analogues |
-
2014
- 2014-05-30 FR FR1454935A patent/FR3021655B1/fr not_active Expired - Fee Related
-
2015
- 2015-05-29 CN CN202010802288.XA patent/CN112194632A/zh active Pending
- 2015-05-29 CA CA2949334A patent/CA2949334A1/fr not_active Abandoned
- 2015-05-29 US US15/314,628 patent/US9926300B2/en active Active
- 2015-05-29 EP EP15735988.6A patent/EP3148973A1/fr not_active Withdrawn
- 2015-05-29 CN CN201580040441.7A patent/CN106573895B/zh not_active Expired - Fee Related
- 2015-05-29 WO PCT/FR2015/051416 patent/WO2015181507A1/fr active Application Filing
-
2018
- 2018-02-08 US US15/891,433 patent/US20180258077A1/en not_active Abandoned
Non-Patent Citations (28)
| Title |
|---|
| ANDREAS SPALTENSTEIN ET AL: "New synthesis of 4- and 5-imidazolethiols", THE JOURNAL OF ORGANIC CHEMISTRY, vol. 52, no. 14, 1 July 1987 (1987-07-01), pages 2977 - 2979, XP055137821, ISSN: 0022-3263, DOI: 10.1021/jo00390a004 * |
| BAILLY ET AL., BIOORG. MED. CHEM., vol. 11, 2003, pages 4623 - 4630 |
| BRAUNSHAUSEN A.; SEEBECK F., JACS, vol. 133, 2011, pages 1757 |
| CAROLL A.; AVREY V.M., J. NAT. PROD., vol. 72, 2009, pages 696 - 699 |
| DE LUNA ET AL., J. PHYS. CHEMISTRY, 2013 |
| HAND C.E.; HONEK J.F., J. NAT. PROD., vol. 68, 2005, pages 293 - 308 |
| HENG SONG ET AL: "Regioselectivity of the Oxidative C-S Bond Formation in Ergothioneine and Ovothiol Biosyntheses", ORGANIC LETTERS, vol. 15, no. 18, 20 September 2013 (2013-09-20), pages 4854 - 4857, XP055138255, ISSN: 1523-7060, DOI: 10.1021/ol402275t * |
| HENG SONG: "Regioselectivity of the oxidative C-S Bond Formation in Ergothioneine and Ovothiol Biosyntheses", ORGANIC LETTERS, vol. 15, no. 18, 20 September 2013 (2013-09-20), pages 4854 - 4857, XP055138255, DOI: doi:10.1021/ol402275t |
| HOLLER ET AL., JOC, 1987, pages 4421 - 4423 |
| HOLLER ET AL., JOC, vol. 20, 1987, pages 4421 - 4423 |
| HOPKINS P. ET AL., JOC, vol. 52, 1987, pages 2977,4420 |
| ITO S.; NARDI G.; PROTA G., JCS CHEM. COMM., 1976, pages 1042 |
| ITO S.; PROTA G., JCS CHEM. COMM., 1977, pages 251 - 252 |
| K. A. PIEZ ET AL., J. BIOL. CHEM., vol. 194, 1952, pages 669 - 672 |
| MASHABELA ET AL., CHEM. COMM., vol. 49, 2013, pages 7714 - 7716 |
| MASHABELA G.; SEEBECK F., JCS CHEM. COMM., 2013, pages 7714 - 7716 |
| OGT R.N.; SPIES H.S.C.; STEENKAMP D.J., EUR. J. BIOCHEM., vol. 268, 2001, pages 5229 - 5241 |
| OHBA M ET AL: "Synthetic Studies on the Starfish Alkaloid Imbricatine. A Chiral Synthesis of Tri-O-methylimbricatine", TETRAHEDRON, ELSEVIER SCIENCE PUBLISHERS, AMSTERDAM, NL, vol. 55, no. 16, 16 April 1999 (1999-04-16), pages 4999 - 5016, XP004161084, ISSN: 0040-4020, DOI: 10.1016/S0040-4020(99)00199-4 * |
| OHBA M.; NISHIMURA Y.; KATO M.; FUJII T., TETRAHEDRON, vol. 55, 1999, pages 4999 - 5016 |
| PALUMBA ET AL., THL, 1982, pages 3207 - 3208 |
| PATHIRANA C.; ANDERSEN R.J., J. AM. CHEM. SOC., vol. 108, 1986, pages 8288 - 8289 |
| PING LIU ET AL: "Discovery of MK-3168: A PET Tracer for Imaging Brain Fatty Acid Amide Hydrolase", ACS MEDICINAL CHEMISTRY LETTERS, vol. 4, no. 6, 13 June 2013 (2013-06-13), pages 509 - 513, XP055138171, ISSN: 1948-5875, DOI: 10.1021/ml4000996 * |
| ROSSI F.; NARDI G.; PALUMBO A.; PROTA G., COMP. BIOCHEM. PHYSIOL., vol. 80B, 1985, pages 843 - 845 |
| SHOSUKE ITO ET AL: "Structures of Adenochrornines A and B, the Iron(III) binding Amino-acids of a Unique Group of Peptides, Adenochromes from Octopus vulgaris", JOURNAL OF THE CHEMICAL SOCIETY, CHEMICAL COMMUNICATIONS, 1 January 1976 (1976-01-01), pages 1042 - 1043, XP055206868 * |
| SPALTENSTEIN, THE JOURNAL OF ORGANIC CHEMISTRY, vol. 52, no. 14, pages 2977 - 2979 |
| TOD P. HOLLER ET AL: "Synthesis and structure reassignment of mercaptohistidines of marine origin. Syntheses of L-ovothiols A and C", THE JOURNAL OF ORGANIC CHEMISTRY, vol. 52, no. 19, 1 September 1987 (1987-09-01), pages 4420 - 4421, XP055138088, ISSN: 0022-3263, DOI: 10.1021/jo00228a060 * |
| TURNER E.; KLEVIT R.E.; SHAPIRO B.M., J. BIOL. CHEM., vol. 261, 1986, pages 13056 |
| V. N. REINHOLD ET AL., J. MED. CHEM., vol. 11, 1968, pages 258 - 260 |
Also Published As
| Publication number | Publication date |
|---|---|
| CA2949334A1 (fr) | 2015-12-03 |
| US20180258077A1 (en) | 2018-09-13 |
| US9926300B2 (en) | 2018-03-27 |
| FR3021655A1 (fr) | 2015-12-04 |
| CN106573895A (zh) | 2017-04-19 |
| CN112194632A (zh) | 2021-01-08 |
| EP3148973A1 (fr) | 2017-04-05 |
| US20170190691A1 (en) | 2017-07-06 |
| FR3021655B1 (fr) | 2016-07-08 |
| CN106573895B (zh) | 2020-11-24 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| CA2776836C (fr) | Procede de synthese de l'ergothioneine et analogues | |
| EP3148973A1 (fr) | Nouveaux composes de type 5-acylsulfanyl-histidine en tant que precurseurs des 5-sulfanylhistidines correspondantes et de leurs disulfures | |
| CA2776833C (fr) | Procede de synthese de la 2-thiohistidine et analogues | |
| US20200017479A1 (en) | Novel 5-acylsulfanyl-histidine compounds as precursors of the corresponding 5-sulfanylhistidines and their disulfides | |
| EP0915858B1 (fr) | Intermediaires pour la preparation de 2-imidazoline-5-ones | |
| AU750368B2 (en) | Process for preparing 4-substituted-1H-indole-3-glyoxamides | |
| FR2965561A1 (fr) | Procede de preparation d’un acide amine a partir de 2-aminobutyrolactone | |
| CN116730969B (zh) | 一种β-拉帕醌-氨基酸缀合物及其制备方法和应用 | |
| EP1620440B1 (fr) | Synthese de composes heterocycliques substitues | |
| WO2000021923A1 (fr) | Procede de preparation de toluenesulfinates | |
| CA2347718A1 (fr) | Procede de preparation de 4-1h-indole-3-glyoxamides substituees | |
| JPH09255672A (ja) | 光学活性3−(p−メトキシフェニル)グリシッド酸アルカリ金属塩の製造法 | |
| PL218006B1 (pl) | Sposób otrzymywania sulfidów |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application |
Ref document number: 15735988 Country of ref document: EP Kind code of ref document: A1 |
|
| ENP | Entry into the national phase |
Ref document number: 2949334 Country of ref document: CA |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 15314628 Country of ref document: US |
|
| NENP | Non-entry into the national phase |
Ref country code: DE |
|
| REEP | Request for entry into the european phase |
Ref document number: 2015735988 Country of ref document: EP |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 2015735988 Country of ref document: EP |