WO2012157737A1 - 免疫誘導剤 - Google Patents
免疫誘導剤 Download PDFInfo
- Publication number
- WO2012157737A1 WO2012157737A1 PCT/JP2012/062750 JP2012062750W WO2012157737A1 WO 2012157737 A1 WO2012157737 A1 WO 2012157737A1 JP 2012062750 W JP2012062750 W JP 2012062750W WO 2012157737 A1 WO2012157737 A1 WO 2012157737A1
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- polypeptide
- cancer
- immunity
- cells
- inducing agent
- Prior art date
Links
- 230000036039 immunity Effects 0.000 title claims abstract description 78
- 230000001939 inductive effect Effects 0.000 title claims abstract description 71
- 108090000765 processed proteins & peptides Proteins 0.000 claims abstract description 168
- 102000004196 processed proteins & peptides Human genes 0.000 claims abstract description 161
- 229920001184 polypeptide Polymers 0.000 claims abstract description 159
- 206010028980 Neoplasm Diseases 0.000 claims abstract description 103
- 201000011510 cancer Diseases 0.000 claims abstract description 77
- 239000003795 chemical substances by application Substances 0.000 claims abstract description 59
- 125000003275 alpha amino acid group Chemical group 0.000 claims abstract description 43
- 150000001413 amino acids Chemical class 0.000 claims abstract description 24
- 238000011282 treatment Methods 0.000 claims abstract description 23
- 238000001727 in vivo Methods 0.000 claims abstract description 20
- 108091033319 polynucleotide Proteins 0.000 claims abstract description 20
- 102000040430 polynucleotide Human genes 0.000 claims abstract description 20
- 239000002157 polynucleotide Substances 0.000 claims abstract description 20
- 239000013598 vector Substances 0.000 claims abstract description 18
- 239000004480 active ingredient Substances 0.000 claims abstract description 8
- 230000036961 partial effect Effects 0.000 claims abstract description 5
- 102100022968 Katanin p60 ATPase-containing subunit A-like 1 Human genes 0.000 claims description 48
- 210000000612 antigen-presenting cell Anatomy 0.000 claims description 43
- 239000002671 adjuvant Substances 0.000 claims description 16
- 208000003174 Brain Neoplasms Diseases 0.000 claims description 8
- 208000026310 Breast neoplasm Diseases 0.000 claims description 8
- 101000975105 Homo sapiens Katanin p60 ATPase-containing subunit A-like 1 Proteins 0.000 claims description 8
- 206010006187 Breast cancer Diseases 0.000 claims description 7
- 102000013462 Interleukin-12 Human genes 0.000 claims description 7
- 108010065805 Interleukin-12 Proteins 0.000 claims description 7
- 206010058467 Lung neoplasm malignant Diseases 0.000 claims description 7
- 206010029260 Neuroblastoma Diseases 0.000 claims description 7
- 206010060862 Prostate cancer Diseases 0.000 claims description 7
- 208000000236 Prostatic Neoplasms Diseases 0.000 claims description 7
- 208000024770 Thyroid neoplasm Diseases 0.000 claims description 7
- 229940117681 interleukin-12 Drugs 0.000 claims description 7
- 201000007270 liver cancer Diseases 0.000 claims description 7
- 208000014018 liver neoplasm Diseases 0.000 claims description 7
- 201000005202 lung cancer Diseases 0.000 claims description 7
- 208000020816 lung neoplasm Diseases 0.000 claims description 7
- 201000002510 thyroid cancer Diseases 0.000 claims description 7
- 208000009956 adenocarcinoma Diseases 0.000 claims description 6
- 230000002708 enhancing effect Effects 0.000 claims description 6
- 230000001225 therapeutic effect Effects 0.000 claims description 6
- 230000000091 immunopotentiator Effects 0.000 claims description 5
- 208000032839 leukemia Diseases 0.000 claims description 5
- 201000006512 mast cell neoplasm Diseases 0.000 claims description 5
- 208000006971 mastocytoma Diseases 0.000 claims description 5
- 230000000069 prophylactic effect Effects 0.000 claims description 5
- 108091034117 Oligonucleotide Proteins 0.000 claims description 4
- 102100020715 Fms-related tyrosine kinase 3 ligand protein Human genes 0.000 claims description 3
- 101710162577 Fms-related tyrosine kinase 3 ligand protein Proteins 0.000 claims description 3
- 102000006992 Interferon-alpha Human genes 0.000 claims description 3
- 108010047761 Interferon-alpha Proteins 0.000 claims description 3
- 102000003996 Interferon-beta Human genes 0.000 claims description 3
- 108090000467 Interferon-beta Proteins 0.000 claims description 3
- 102000008070 Interferon-gamma Human genes 0.000 claims description 3
- 108010074328 Interferon-gamma Proteins 0.000 claims description 3
- 102000003810 Interleukin-18 Human genes 0.000 claims description 3
- 108090000171 Interleukin-18 Proteins 0.000 claims description 3
- 229960003130 interferon gamma Drugs 0.000 claims description 3
- 108010045648 interferon omega 1 Proteins 0.000 claims description 3
- 229960001388 interferon-beta Drugs 0.000 claims description 3
- CZMRCDWAGMRECN-UGDNZRGBSA-N Sucrose Chemical compound O[C@H]1[C@H](O)[C@@H](CO)O[C@@]1(CO)O[C@@H]1[C@H](O)[C@@H](O)[C@H](O)[C@@H](CO)O1 CZMRCDWAGMRECN-UGDNZRGBSA-N 0.000 claims 1
- 229930006000 Sucrose Natural products 0.000 claims 1
- 239000005720 sucrose Substances 0.000 claims 1
- 230000002265 prevention Effects 0.000 abstract description 16
- 210000004027 cell Anatomy 0.000 description 110
- 238000000034 method Methods 0.000 description 56
- 108090000623 proteins and genes Proteins 0.000 description 48
- 101710082999 Katanin p60 ATPase-containing subunit A-like 1 Proteins 0.000 description 47
- 210000001519 tissue Anatomy 0.000 description 39
- 241000282414 Homo sapiens Species 0.000 description 32
- 210000001744 T-lymphocyte Anatomy 0.000 description 27
- 239000002299 complementary DNA Substances 0.000 description 26
- 108020004414 DNA Proteins 0.000 description 24
- 230000014509 gene expression Effects 0.000 description 24
- 241000282472 Canis lupus familiaris Species 0.000 description 22
- 239000002609 medium Substances 0.000 description 22
- 102000004169 proteins and genes Human genes 0.000 description 21
- 239000013612 plasmid Substances 0.000 description 19
- 210000002966 serum Anatomy 0.000 description 19
- 125000000539 amino acid group Chemical group 0.000 description 18
- FAPWRFPIFSIZLT-UHFFFAOYSA-M Sodium chloride Chemical compound [Na+].[Cl-] FAPWRFPIFSIZLT-UHFFFAOYSA-M 0.000 description 17
- 241000588724 Escherichia coli Species 0.000 description 16
- 108091007433 antigens Proteins 0.000 description 14
- 102000036639 antigens Human genes 0.000 description 13
- 210000001151 cytotoxic T lymphocyte Anatomy 0.000 description 13
- 239000013604 expression vector Substances 0.000 description 13
- 241000282465 Canis Species 0.000 description 12
- 102100034922 T-cell surface glycoprotein CD8 alpha chain Human genes 0.000 description 12
- 239000000872 buffer Substances 0.000 description 12
- 210000004443 dendritic cell Anatomy 0.000 description 12
- 238000002360 preparation method Methods 0.000 description 12
- 241000699666 Mus <mouse, genus> Species 0.000 description 11
- 230000000259 anti-tumor effect Effects 0.000 description 11
- 239000000427 antigen Substances 0.000 description 11
- 101150021835 katnal1 gene Proteins 0.000 description 11
- 238000006243 chemical reaction Methods 0.000 description 10
- 238000012258 culturing Methods 0.000 description 10
- 239000012634 fragment Substances 0.000 description 10
- LFQSCWFLJHTTHZ-UHFFFAOYSA-N Ethanol Chemical compound CCO LFQSCWFLJHTTHZ-UHFFFAOYSA-N 0.000 description 9
- 239000011780 sodium chloride Substances 0.000 description 9
- 241001465754 Metazoa Species 0.000 description 8
- 229910002091 carbon monoxide Inorganic materials 0.000 description 8
- 230000001472 cytotoxic effect Effects 0.000 description 8
- 230000006698 induction Effects 0.000 description 8
- 239000002773 nucleotide Substances 0.000 description 8
- 125000003729 nucleotide group Chemical group 0.000 description 8
- 239000000243 solution Substances 0.000 description 8
- 210000004881 tumor cell Anatomy 0.000 description 8
- QKNYBSVHEMOAJP-UHFFFAOYSA-N 2-amino-2-(hydroxymethyl)propane-1,3-diol;hydron;chloride Chemical compound Cl.OCC(N)(CO)CO QKNYBSVHEMOAJP-UHFFFAOYSA-N 0.000 description 7
- 102000004127 Cytokines Human genes 0.000 description 7
- 108090000695 Cytokines Proteins 0.000 description 7
- 108010008281 Recombinant Fusion Proteins Proteins 0.000 description 7
- 102000007056 Recombinant Fusion Proteins Human genes 0.000 description 7
- 238000007796 conventional method Methods 0.000 description 7
- 239000007788 liquid Substances 0.000 description 7
- 210000005259 peripheral blood Anatomy 0.000 description 7
- 239000011886 peripheral blood Substances 0.000 description 7
- 238000003757 reverse transcription PCR Methods 0.000 description 7
- 239000000523 sample Substances 0.000 description 7
- FWMNVWWHGCHHJJ-SKKKGAJSSA-N 4-amino-1-[(2r)-6-amino-2-[[(2r)-2-[[(2r)-2-[[(2r)-2-amino-3-phenylpropanoyl]amino]-3-phenylpropanoyl]amino]-4-methylpentanoyl]amino]hexanoyl]piperidine-4-carboxylic acid Chemical compound C([C@H](C(=O)N[C@H](CC(C)C)C(=O)N[C@H](CCCCN)C(=O)N1CCC(N)(CC1)C(O)=O)NC(=O)[C@H](N)CC=1C=CC=CC=1)C1=CC=CC=C1 FWMNVWWHGCHHJJ-SKKKGAJSSA-N 0.000 description 6
- 241000283690 Bos taurus Species 0.000 description 6
- 241000283073 Equus caballus Species 0.000 description 6
- 241000287828 Gallus gallus Species 0.000 description 6
- 241000699670 Mus sp. Species 0.000 description 6
- -1 Val Chemical compound 0.000 description 6
- 210000001185 bone marrow Anatomy 0.000 description 6
- 230000000694 effects Effects 0.000 description 6
- 108020001507 fusion proteins Proteins 0.000 description 6
- 102000037865 fusion proteins Human genes 0.000 description 6
- 238000000338 in vitro Methods 0.000 description 6
- 210000000265 leukocyte Anatomy 0.000 description 6
- 239000002502 liposome Substances 0.000 description 6
- 108091008146 restriction endonucleases Proteins 0.000 description 6
- 229960005486 vaccine Drugs 0.000 description 6
- 108091032973 (ribonucleotides)n+m Proteins 0.000 description 5
- VYZAMTAEIAYCRO-BJUDXGSMSA-N Chromium-51 Chemical compound [51Cr] VYZAMTAEIAYCRO-BJUDXGSMSA-N 0.000 description 5
- 241000282326 Felis catus Species 0.000 description 5
- 108010017213 Granulocyte-Macrophage Colony-Stimulating Factor Proteins 0.000 description 5
- 102100039620 Granulocyte-macrophage colony-stimulating factor Human genes 0.000 description 5
- 241000282412 Homo Species 0.000 description 5
- 101100126865 Mus musculus Katnal1 gene Proteins 0.000 description 5
- 239000000654 additive Substances 0.000 description 5
- 238000003501 co-culture Methods 0.000 description 5
- 102000048547 human KATNAL1 Human genes 0.000 description 5
- 230000028993 immune response Effects 0.000 description 5
- 239000000203 mixture Substances 0.000 description 5
- 239000002245 particle Substances 0.000 description 5
- 210000003819 peripheral blood mononuclear cell Anatomy 0.000 description 5
- 102200082402 rs751610198 Human genes 0.000 description 5
- 239000006228 supernatant Substances 0.000 description 5
- 108090001005 Interleukin-6 Proteins 0.000 description 4
- 108091005804 Peptidases Proteins 0.000 description 4
- 238000012181 QIAquick gel extraction kit Methods 0.000 description 4
- 230000001464 adherent effect Effects 0.000 description 4
- 239000003153 chemical reaction reagent Substances 0.000 description 4
- 239000003814 drug Substances 0.000 description 4
- 210000002865 immune cell Anatomy 0.000 description 4
- BPHPUYQFMNQIOC-NXRLNHOXSA-N isopropyl beta-D-thiogalactopyranoside Chemical compound CC(C)S[C@@H]1O[C@H](CO)[C@H](O)[C@H](O)[C@H]1O BPHPUYQFMNQIOC-NXRLNHOXSA-N 0.000 description 4
- 238000004519 manufacturing process Methods 0.000 description 4
- 238000007911 parenteral administration Methods 0.000 description 4
- 238000012216 screening Methods 0.000 description 4
- 210000001550 testis Anatomy 0.000 description 4
- 206010009944 Colon cancer Diseases 0.000 description 3
- 108010041986 DNA Vaccines Proteins 0.000 description 3
- 229940021995 DNA vaccine Drugs 0.000 description 3
- 108010002350 Interleukin-2 Proteins 0.000 description 3
- 108090000978 Interleukin-4 Proteins 0.000 description 3
- 108010002586 Interleukin-7 Proteins 0.000 description 3
- 102000043129 MHC class I family Human genes 0.000 description 3
- 108091054437 MHC class I family Proteins 0.000 description 3
- 238000002835 absorbance Methods 0.000 description 3
- AVKUERGKIZMTKX-NJBDSQKTSA-N ampicillin Chemical compound C1([C@@H](N)C(=O)N[C@H]2[C@H]3SC([C@@H](N3C2=O)C(O)=O)(C)C)=CC=CC=C1 AVKUERGKIZMTKX-NJBDSQKTSA-N 0.000 description 3
- 229960000723 ampicillin Drugs 0.000 description 3
- 230000001093 anti-cancer Effects 0.000 description 3
- 239000002246 antineoplastic agent Substances 0.000 description 3
- 239000001506 calcium phosphate Substances 0.000 description 3
- 229910000389 calcium phosphate Inorganic materials 0.000 description 3
- 235000011010 calcium phosphates Nutrition 0.000 description 3
- 238000005119 centrifugation Methods 0.000 description 3
- 238000003776 cleavage reaction Methods 0.000 description 3
- 238000010367 cloning Methods 0.000 description 3
- 208000029742 colonic neoplasm Diseases 0.000 description 3
- 239000012228 culture supernatant Substances 0.000 description 3
- 238000011161 development Methods 0.000 description 3
- 201000010099 disease Diseases 0.000 description 3
- 208000037265 diseases, disorders, signs and symptoms Diseases 0.000 description 3
- 229940079593 drug Drugs 0.000 description 3
- 210000003527 eukaryotic cell Anatomy 0.000 description 3
- 238000001400 expression cloning Methods 0.000 description 3
- 235000013861 fat-free Nutrition 0.000 description 3
- 210000004700 fetal blood Anatomy 0.000 description 3
- 238000001361 intraarterial administration Methods 0.000 description 3
- 238000001990 intravenous administration Methods 0.000 description 3
- 210000004698 lymphocyte Anatomy 0.000 description 3
- 210000004962 mammalian cell Anatomy 0.000 description 3
- 239000012528 membrane Substances 0.000 description 3
- 235000013336 milk Nutrition 0.000 description 3
- 239000008267 milk Substances 0.000 description 3
- 210000004080 milk Anatomy 0.000 description 3
- 238000002156 mixing Methods 0.000 description 3
- 210000005087 mononuclear cell Anatomy 0.000 description 3
- 210000001236 prokaryotic cell Anatomy 0.000 description 3
- 230000007017 scission Effects 0.000 description 3
- 238000000926 separation method Methods 0.000 description 3
- 208000024891 symptom Diseases 0.000 description 3
- 230000001131 transforming effect Effects 0.000 description 3
- QORWJWZARLRLPR-UHFFFAOYSA-H tricalcium bis(phosphate) Chemical compound [Ca+2].[Ca+2].[Ca+2].[O-]P([O-])([O-])=O.[O-]P([O-])([O-])=O QORWJWZARLRLPR-UHFFFAOYSA-H 0.000 description 3
- 229920000936 Agarose Polymers 0.000 description 2
- 101000918303 Bos taurus Exostosin-2 Proteins 0.000 description 2
- 241000283707 Capra Species 0.000 description 2
- 108020004635 Complementary DNA Proteins 0.000 description 2
- FBPFZTCFMRRESA-FSIIMWSLSA-N D-Glucitol Natural products OC[C@H](O)[C@H](O)[C@@H](O)[C@H](O)CO FBPFZTCFMRRESA-FSIIMWSLSA-N 0.000 description 2
- 241000702421 Dependoparvovirus Species 0.000 description 2
- 108010010803 Gelatin Proteins 0.000 description 2
- 102000005720 Glutathione transferase Human genes 0.000 description 2
- 108010070675 Glutathione transferase Proteins 0.000 description 2
- DHMQDGOQFOQNFH-UHFFFAOYSA-N Glycine Chemical compound NCC(O)=O DHMQDGOQFOQNFH-UHFFFAOYSA-N 0.000 description 2
- VEXZGXHMUGYJMC-UHFFFAOYSA-N Hydrochloric acid Chemical compound Cl VEXZGXHMUGYJMC-UHFFFAOYSA-N 0.000 description 2
- 241000124008 Mammalia Species 0.000 description 2
- PXHVJJICTQNCMI-UHFFFAOYSA-N Nickel Chemical compound [Ni] PXHVJJICTQNCMI-UHFFFAOYSA-N 0.000 description 2
- 102000035195 Peptidases Human genes 0.000 description 2
- 239000004365 Protease Substances 0.000 description 2
- 102100037486 Reverse transcriptase/ribonuclease H Human genes 0.000 description 2
- 238000012300 Sequence Analysis Methods 0.000 description 2
- VYPSYNLAJGMNEJ-UHFFFAOYSA-N Silicium dioxide Chemical compound O=[Si]=O VYPSYNLAJGMNEJ-UHFFFAOYSA-N 0.000 description 2
- 229920006355 Tefzel Polymers 0.000 description 2
- XSQUKJJJFZCRTK-UHFFFAOYSA-N Urea Chemical compound NC(N)=O XSQUKJJJFZCRTK-UHFFFAOYSA-N 0.000 description 2
- 241000700618 Vaccinia virus Species 0.000 description 2
- 241000700605 Viruses Species 0.000 description 2
- JLCPHMBAVCMARE-UHFFFAOYSA-N [3-[[3-[[3-[[3-[[3-[[3-[[3-[[3-[[3-[[3-[[3-[[5-(2-amino-6-oxo-1H-purin-9-yl)-3-[[3-[[3-[[3-[[3-[[3-[[5-(2-amino-6-oxo-1H-purin-9-yl)-3-[[5-(2-amino-6-oxo-1H-purin-9-yl)-3-hydroxyoxolan-2-yl]methoxy-hydroxyphosphoryl]oxyoxolan-2-yl]methoxy-hydroxyphosphoryl]oxy-5-(5-methyl-2,4-dioxopyrimidin-1-yl)oxolan-2-yl]methoxy-hydroxyphosphoryl]oxy-5-(6-aminopurin-9-yl)oxolan-2-yl]methoxy-hydroxyphosphoryl]oxy-5-(6-aminopurin-9-yl)oxolan-2-yl]methoxy-hydroxyphosphoryl]oxy-5-(6-aminopurin-9-yl)oxolan-2-yl]methoxy-hydroxyphosphoryl]oxy-5-(6-aminopurin-9-yl)oxolan-2-yl]methoxy-hydroxyphosphoryl]oxyoxolan-2-yl]methoxy-hydroxyphosphoryl]oxy-5-(5-methyl-2,4-dioxopyrimidin-1-yl)oxolan-2-yl]methoxy-hydroxyphosphoryl]oxy-5-(4-amino-2-oxopyrimidin-1-yl)oxolan-2-yl]methoxy-hydroxyphosphoryl]oxy-5-(5-methyl-2,4-dioxopyrimidin-1-yl)oxolan-2-yl]methoxy-hydroxyphosphoryl]oxy-5-(5-methyl-2,4-dioxopyrimidin-1-yl)oxolan-2-yl]methoxy-hydroxyphosphoryl]oxy-5-(6-aminopurin-9-yl)oxolan-2-yl]methoxy-hydroxyphosphoryl]oxy-5-(6-aminopurin-9-yl)oxolan-2-yl]methoxy-hydroxyphosphoryl]oxy-5-(4-amino-2-oxopyrimidin-1-yl)oxolan-2-yl]methoxy-hydroxyphosphoryl]oxy-5-(4-amino-2-oxopyrimidin-1-yl)oxolan-2-yl]methoxy-hydroxyphosphoryl]oxy-5-(4-amino-2-oxopyrimidin-1-yl)oxolan-2-yl]methoxy-hydroxyphosphoryl]oxy-5-(6-aminopurin-9-yl)oxolan-2-yl]methoxy-hydroxyphosphoryl]oxy-5-(4-amino-2-oxopyrimidin-1-yl)oxolan-2-yl]methyl [5-(6-aminopurin-9-yl)-2-(hydroxymethyl)oxolan-3-yl] hydrogen phosphate Polymers Cc1cn(C2CC(OP(O)(=O)OCC3OC(CC3OP(O)(=O)OCC3OC(CC3O)n3cnc4c3nc(N)[nH]c4=O)n3cnc4c3nc(N)[nH]c4=O)C(COP(O)(=O)OC3CC(OC3COP(O)(=O)OC3CC(OC3COP(O)(=O)OC3CC(OC3COP(O)(=O)OC3CC(OC3COP(O)(=O)OC3CC(OC3COP(O)(=O)OC3CC(OC3COP(O)(=O)OC3CC(OC3COP(O)(=O)OC3CC(OC3COP(O)(=O)OC3CC(OC3COP(O)(=O)OC3CC(OC3COP(O)(=O)OC3CC(OC3COP(O)(=O)OC3CC(OC3COP(O)(=O)OC3CC(OC3COP(O)(=O)OC3CC(OC3COP(O)(=O)OC3CC(OC3COP(O)(=O)OC3CC(OC3COP(O)(=O)OC3CC(OC3CO)n3cnc4c(N)ncnc34)n3ccc(N)nc3=O)n3cnc4c(N)ncnc34)n3ccc(N)nc3=O)n3ccc(N)nc3=O)n3ccc(N)nc3=O)n3cnc4c(N)ncnc34)n3cnc4c(N)ncnc34)n3cc(C)c(=O)[nH]c3=O)n3cc(C)c(=O)[nH]c3=O)n3ccc(N)nc3=O)n3cc(C)c(=O)[nH]c3=O)n3cnc4c3nc(N)[nH]c4=O)n3cnc4c(N)ncnc34)n3cnc4c(N)ncnc34)n3cnc4c(N)ncnc34)n3cnc4c(N)ncnc34)O2)c(=O)[nH]c1=O JLCPHMBAVCMARE-UHFFFAOYSA-N 0.000 description 2
- 239000008351 acetate buffer Substances 0.000 description 2
- 239000011543 agarose gel Substances 0.000 description 2
- 229940041181 antineoplastic drug Drugs 0.000 description 2
- 239000012298 atmosphere Substances 0.000 description 2
- 210000003719 b-lymphocyte Anatomy 0.000 description 2
- 238000010805 cDNA synthesis kit Methods 0.000 description 2
- 239000000969 carrier Substances 0.000 description 2
- 230000015556 catabolic process Effects 0.000 description 2
- 239000013611 chromosomal DNA Substances 0.000 description 2
- 239000013599 cloning vector Substances 0.000 description 2
- 230000000295 complement effect Effects 0.000 description 2
- 238000006731 degradation reaction Methods 0.000 description 2
- 238000004925 denaturation Methods 0.000 description 2
- 230000036425 denaturation Effects 0.000 description 2
- 239000003085 diluting agent Substances 0.000 description 2
- LOKCTEFSRHRXRJ-UHFFFAOYSA-I dipotassium trisodium dihydrogen phosphate hydrogen phosphate dichloride Chemical compound P(=O)(O)(O)[O-].[K+].P(=O)(O)([O-])[O-].[Na+].[Na+].[Cl-].[K+].[Cl-].[Na+] LOKCTEFSRHRXRJ-UHFFFAOYSA-I 0.000 description 2
- 238000004520 electroporation Methods 0.000 description 2
- 238000010828 elution Methods 0.000 description 2
- 239000000839 emulsion Substances 0.000 description 2
- 239000003623 enhancer Substances 0.000 description 2
- QHSJIZLJUFMIFP-UHFFFAOYSA-N ethene;1,1,2,2-tetrafluoroethene Chemical compound C=C.FC(F)=C(F)F QHSJIZLJUFMIFP-UHFFFAOYSA-N 0.000 description 2
- 238000010195 expression analysis Methods 0.000 description 2
- 238000007710 freezing Methods 0.000 description 2
- 230000008014 freezing Effects 0.000 description 2
- 239000007789 gas Substances 0.000 description 2
- 239000008273 gelatin Substances 0.000 description 2
- 229920000159 gelatin Polymers 0.000 description 2
- 235000019322 gelatine Nutrition 0.000 description 2
- 235000011852 gelatine desserts Nutrition 0.000 description 2
- 238000010353 genetic engineering Methods 0.000 description 2
- PCHJSUWPFVWCPO-UHFFFAOYSA-N gold Chemical compound [Au] PCHJSUWPFVWCPO-UHFFFAOYSA-N 0.000 description 2
- 229910052737 gold Inorganic materials 0.000 description 2
- 239000010931 gold Substances 0.000 description 2
- 229960000789 guanidine hydrochloride Drugs 0.000 description 2
- PJJJBBJSCAKJQF-UHFFFAOYSA-N guanidinium chloride Chemical compound [Cl-].NC(N)=[NH2+] PJJJBBJSCAKJQF-UHFFFAOYSA-N 0.000 description 2
- 238000009169 immunotherapy Methods 0.000 description 2
- 238000002347 injection Methods 0.000 description 2
- 239000007924 injection Substances 0.000 description 2
- 238000007918 intramuscular administration Methods 0.000 description 2
- 210000002540 macrophage Anatomy 0.000 description 2
- HQKMJHAJHXVSDF-UHFFFAOYSA-L magnesium stearate Chemical compound [Mg+2].CCCCCCCCCCCCCCCCCC([O-])=O.CCCCCCCCCCCCCCCCCC([O-])=O HQKMJHAJHXVSDF-UHFFFAOYSA-L 0.000 description 2
- 239000000463 material Substances 0.000 description 2
- 108020004999 messenger RNA Proteins 0.000 description 2
- 125000001360 methionine group Chemical group N[C@@H](CCSC)C(=O)* 0.000 description 2
- 230000004048 modification Effects 0.000 description 2
- 238000012986 modification Methods 0.000 description 2
- 239000013642 negative control Substances 0.000 description 2
- 230000007935 neutral effect Effects 0.000 description 2
- 102000039446 nucleic acids Human genes 0.000 description 2
- 108020004707 nucleic acids Proteins 0.000 description 2
- 150000007523 nucleic acids Chemical class 0.000 description 2
- 239000003921 oil Substances 0.000 description 2
- 239000000546 pharmaceutical excipient Substances 0.000 description 2
- 238000002205 phenol-chloroform extraction Methods 0.000 description 2
- 239000008363 phosphate buffer Substances 0.000 description 2
- 239000002953 phosphate buffered saline Substances 0.000 description 2
- 239000013600 plasmid vector Substances 0.000 description 2
- 235000019833 protease Nutrition 0.000 description 2
- 235000019419 proteases Nutrition 0.000 description 2
- 238000000746 purification Methods 0.000 description 2
- 239000012521 purified sample Substances 0.000 description 2
- 230000002829 reductive effect Effects 0.000 description 2
- 230000004044 response Effects 0.000 description 2
- 238000010839 reverse transcription Methods 0.000 description 2
- 238000012163 sequencing technique Methods 0.000 description 2
- 238000000527 sonication Methods 0.000 description 2
- 239000000600 sorbitol Substances 0.000 description 2
- 235000010356 sorbitol Nutrition 0.000 description 2
- ATHGHQPFGPMSJY-UHFFFAOYSA-N spermidine Chemical compound NCCCCNCCCN ATHGHQPFGPMSJY-UHFFFAOYSA-N 0.000 description 2
- 230000004936 stimulating effect Effects 0.000 description 2
- 238000007920 subcutaneous administration Methods 0.000 description 2
- 238000006467 substitution reaction Methods 0.000 description 2
- 238000001356 surgical procedure Methods 0.000 description 2
- 230000004083 survival effect Effects 0.000 description 2
- 238000003786 synthesis reaction Methods 0.000 description 2
- 239000006188 syrup Substances 0.000 description 2
- 235000020357 syrup Nutrition 0.000 description 2
- 230000008685 targeting Effects 0.000 description 2
- 238000000108 ultra-filtration Methods 0.000 description 2
- 241000701161 unidentified adenovirus Species 0.000 description 2
- 241001430294 unidentified retrovirus Species 0.000 description 2
- 238000003260 vortexing Methods 0.000 description 2
- MZOFCQQQCNRIBI-VMXHOPILSA-N (3s)-4-[[(2s)-1-[[(2s)-1-[[(1s)-1-carboxy-2-hydroxyethyl]amino]-4-methyl-1-oxopentan-2-yl]amino]-5-(diaminomethylideneamino)-1-oxopentan-2-yl]amino]-3-[[2-[[(2s)-2,6-diaminohexanoyl]amino]acetyl]amino]-4-oxobutanoic acid Chemical compound OC[C@@H](C(O)=O)NC(=O)[C@H](CC(C)C)NC(=O)[C@H](CCCN=C(N)N)NC(=O)[C@H](CC(O)=O)NC(=O)CNC(=O)[C@@H](N)CCCCN MZOFCQQQCNRIBI-VMXHOPILSA-N 0.000 description 1
- YYGNTYWPHWGJRM-UHFFFAOYSA-N (6E,10E,14E,18E)-2,6,10,15,19,23-hexamethyltetracosa-2,6,10,14,18,22-hexaene Chemical compound CC(C)=CCCC(C)=CCCC(C)=CCCC=C(C)CCC=C(C)CCC=C(C)C YYGNTYWPHWGJRM-UHFFFAOYSA-N 0.000 description 1
- 125000003088 (fluoren-9-ylmethoxy)carbonyl group Chemical group 0.000 description 1
- QAPSNMNOIOSXSQ-YNEHKIRRSA-N 1-[(2r,4s,5r)-4-[tert-butyl(dimethyl)silyl]oxy-5-(hydroxymethyl)oxolan-2-yl]-5-methylpyrimidine-2,4-dione Chemical compound O=C1NC(=O)C(C)=CN1[C@@H]1O[C@H](CO)[C@@H](O[Si](C)(C)C(C)(C)C)C1 QAPSNMNOIOSXSQ-YNEHKIRRSA-N 0.000 description 1
- BFSVOASYOCHEOV-UHFFFAOYSA-N 2-diethylaminoethanol Chemical compound CCN(CC)CCO BFSVOASYOCHEOV-UHFFFAOYSA-N 0.000 description 1
- QRXMUCSWCMTJGU-UHFFFAOYSA-N 5-bromo-4-chloro-3-indolyl phosphate Chemical compound C1=C(Br)C(Cl)=C2C(OP(O)(=O)O)=CNC2=C1 QRXMUCSWCMTJGU-UHFFFAOYSA-N 0.000 description 1
- 244000215068 Acacia senegal Species 0.000 description 1
- 208000007860 Anus Neoplasms Diseases 0.000 description 1
- 241000416162 Astragalus gummifer Species 0.000 description 1
- 241000255789 Bombyx mori Species 0.000 description 1
- 101100348617 Candida albicans (strain SC5314 / ATCC MYA-2876) NIK1 gene Proteins 0.000 description 1
- 241000282693 Cercopithecidae Species 0.000 description 1
- 108020004705 Codon Proteins 0.000 description 1
- 229920002261 Corn starch Polymers 0.000 description 1
- FBPFZTCFMRRESA-JGWLITMVSA-N D-glucitol Chemical compound OC[C@H](O)[C@@H](O)[C@H](O)[C@H](O)CO FBPFZTCFMRRESA-JGWLITMVSA-N 0.000 description 1
- 229920002307 Dextran Polymers 0.000 description 1
- 238000011510 Elispot assay Methods 0.000 description 1
- 241000991587 Enterovirus C Species 0.000 description 1
- 102000004190 Enzymes Human genes 0.000 description 1
- 108090000790 Enzymes Proteins 0.000 description 1
- 241001198387 Escherichia coli BL21(DE3) Species 0.000 description 1
- 108010074860 Factor Xa Proteins 0.000 description 1
- 241000282324 Felis Species 0.000 description 1
- 101150112014 Gapdh gene Proteins 0.000 description 1
- YRMZCZIRHYCNHX-RYUDHWBXSA-N Glu-Phe-Gly Chemical compound [H]N[C@@H](CCC(O)=O)C(=O)N[C@@H](CC1=CC=CC=C1)C(=O)NCC(O)=O YRMZCZIRHYCNHX-RYUDHWBXSA-N 0.000 description 1
- 102100031181 Glyceraldehyde-3-phosphate dehydrogenase Human genes 0.000 description 1
- 229920000084 Gum arabic Polymers 0.000 description 1
- HVLSXIKZNLPZJJ-TXZCQADKSA-N HA peptide Chemical compound C([C@@H](C(=O)N[C@@H](CC(O)=O)C(=O)N[C@@H](C(C)C)C(=O)N1[C@@H](CCC1)C(=O)N[C@@H](CC(O)=O)C(=O)N[C@@H](CC=1C=CC(O)=CC=1)C(=O)N[C@@H](C)C(O)=O)NC(=O)[C@H]1N(CCC1)C(=O)[C@@H](N)CC=1C=CC(O)=CC=1)C1=CC=C(O)C=C1 HVLSXIKZNLPZJJ-TXZCQADKSA-N 0.000 description 1
- 102100028972 HLA class I histocompatibility antigen, A alpha chain Human genes 0.000 description 1
- 102100028976 HLA class I histocompatibility antigen, B alpha chain Human genes 0.000 description 1
- 102100028971 HLA class I histocompatibility antigen, C alpha chain Human genes 0.000 description 1
- 108010075704 HLA-A Antigens Proteins 0.000 description 1
- 108010088729 HLA-A*02:01 antigen Proteins 0.000 description 1
- 108010035452 HLA-A1 Antigen Proteins 0.000 description 1
- 108010036972 HLA-A11 Antigen Proteins 0.000 description 1
- 108010013476 HLA-A24 Antigen Proteins 0.000 description 1
- 108010018475 HLA-A31 antigen Proteins 0.000 description 1
- 108010058607 HLA-B Antigens Proteins 0.000 description 1
- 108010070087 HLA-B37 Antigen Proteins 0.000 description 1
- 108010091938 HLA-B7 Antigen Proteins 0.000 description 1
- 108010039075 HLA-B8 Antigen Proteins 0.000 description 1
- 108010052199 HLA-C Antigens Proteins 0.000 description 1
- 101100281953 Homo sapiens GAPDH gene Proteins 0.000 description 1
- 101001066129 Homo sapiens Glyceraldehyde-3-phosphate dehydrogenase Proteins 0.000 description 1
- 101001046677 Homo sapiens Integrin alpha-V Proteins 0.000 description 1
- 101100126864 Homo sapiens KATNAL1 gene Proteins 0.000 description 1
- 101001005728 Homo sapiens Melanoma-associated antigen 1 Proteins 0.000 description 1
- 102100022337 Integrin alpha-V Human genes 0.000 description 1
- 102000014150 Interferons Human genes 0.000 description 1
- 108010050904 Interferons Proteins 0.000 description 1
- 108010002386 Interleukin-3 Proteins 0.000 description 1
- 102000015696 Interleukins Human genes 0.000 description 1
- 108010063738 Interleukins Proteins 0.000 description 1
- GUBGYTABKSRVRQ-QKKXKWKRSA-N Lactose Natural products OC[C@H]1O[C@@H](O[C@H]2[C@H](O)[C@@H](O)C(O)O[C@@H]2CO)[C@H](O)[C@@H](O)[C@H]1O GUBGYTABKSRVRQ-QKKXKWKRSA-N 0.000 description 1
- 239000006142 Luria-Bertani Agar Substances 0.000 description 1
- 102100025050 Melanoma-associated antigen 1 Human genes 0.000 description 1
- YNOVBMBQSQTLFM-DCAQKATOSA-N Met-Asn-Leu Chemical compound [H]N[C@@H](CCSC)C(=O)N[C@@H](CC(N)=O)C(=O)N[C@@H](CC(C)C)C(O)=O YNOVBMBQSQTLFM-DCAQKATOSA-N 0.000 description 1
- 108091022875 Microtubule Proteins 0.000 description 1
- 102000029749 Microtubule Human genes 0.000 description 1
- 241000711408 Murine respirovirus Species 0.000 description 1
- 101100013786 Mus musculus Gapdh gene Proteins 0.000 description 1
- 101000819572 Mus musculus Glyceraldehyde-3-phosphate dehydrogenase Proteins 0.000 description 1
- 239000000020 Nitrocellulose Substances 0.000 description 1
- 206010029719 Nonspecific reaction Diseases 0.000 description 1
- 208000008589 Obesity Diseases 0.000 description 1
- 108010038807 Oligopeptides Proteins 0.000 description 1
- 102000015636 Oligopeptides Human genes 0.000 description 1
- 239000012648 POLY-ICLC Substances 0.000 description 1
- 102000007079 Peptide Fragments Human genes 0.000 description 1
- 108010033276 Peptide Fragments Proteins 0.000 description 1
- 239000002202 Polyethylene glycol Substances 0.000 description 1
- 229920001213 Polysorbate 20 Polymers 0.000 description 1
- 241000288906 Primates Species 0.000 description 1
- 108091034057 RNA (poly(A)) Proteins 0.000 description 1
- 101100007329 Saccharomyces cerevisiae (strain ATCC 204508 / S288c) COS1 gene Proteins 0.000 description 1
- 241000235343 Saccharomycetales Species 0.000 description 1
- 241000607142 Salmonella Species 0.000 description 1
- 241000235347 Schizosaccharomyces pombe Species 0.000 description 1
- 229920002684 Sepharose Polymers 0.000 description 1
- 241000710960 Sindbis virus Species 0.000 description 1
- BHEOSNUKNHRBNM-UHFFFAOYSA-N Tetramethylsqualene Natural products CC(=C)C(C)CCC(=C)C(C)CCC(C)=CCCC=C(C)CCC(C)C(=C)CCC(C)C(C)=C BHEOSNUKNHRBNM-UHFFFAOYSA-N 0.000 description 1
- 229920001615 Tragacanth Polymers 0.000 description 1
- 108060008682 Tumor Necrosis Factor Proteins 0.000 description 1
- 102000000852 Tumor Necrosis Factor-alpha Human genes 0.000 description 1
- 241000269370 Xenopus <genus> Species 0.000 description 1
- HMNZFMSWFCAGGW-XPWSMXQVSA-N [3-[hydroxy(2-hydroxyethoxy)phosphoryl]oxy-2-[(e)-octadec-9-enoyl]oxypropyl] (e)-octadec-9-enoate Chemical compound CCCCCCCC\C=C\CCCCCCCC(=O)OCC(COP(O)(=O)OCCO)OC(=O)CCCCCCC\C=C\CCCCCCCC HMNZFMSWFCAGGW-XPWSMXQVSA-N 0.000 description 1
- 210000000683 abdominal cavity Anatomy 0.000 description 1
- 239000000205 acacia gum Substances 0.000 description 1
- 235000010489 acacia gum Nutrition 0.000 description 1
- 230000021736 acetylation Effects 0.000 description 1
- 238000006640 acetylation reaction Methods 0.000 description 1
- 239000002253 acid Substances 0.000 description 1
- 238000005903 acid hydrolysis reaction Methods 0.000 description 1
- 230000002378 acidificating effect Effects 0.000 description 1
- 230000003213 activating effect Effects 0.000 description 1
- 230000000996 additive effect Effects 0.000 description 1
- 238000001042 affinity chromatography Methods 0.000 description 1
- 229940037003 alum Drugs 0.000 description 1
- 238000004458 analytical method Methods 0.000 description 1
- 210000004102 animal cell Anatomy 0.000 description 1
- 238000000137 annealing Methods 0.000 description 1
- 230000030741 antigen processing and presentation Effects 0.000 description 1
- 210000001367 artery Anatomy 0.000 description 1
- 238000003556 assay Methods 0.000 description 1
- 239000011230 binding agent Substances 0.000 description 1
- 230000015572 biosynthetic process Effects 0.000 description 1
- 210000004369 blood Anatomy 0.000 description 1
- 239000008280 blood Substances 0.000 description 1
- 230000036760 body temperature Effects 0.000 description 1
- 230000037396 body weight Effects 0.000 description 1
- 210000002798 bone marrow cell Anatomy 0.000 description 1
- 210000004556 brain Anatomy 0.000 description 1
- 201000000488 breast squamous cell carcinoma Diseases 0.000 description 1
- 230000005907 cancer growth Effects 0.000 description 1
- 238000002619 cancer immunotherapy Methods 0.000 description 1
- 239000012830 cancer therapeutic Substances 0.000 description 1
- 239000002775 capsule Substances 0.000 description 1
- 239000004202 carbamide Substances 0.000 description 1
- 238000004113 cell culture Methods 0.000 description 1
- 239000006285 cell suspension Substances 0.000 description 1
- 230000008859 change Effects 0.000 description 1
- 239000013522 chelant Substances 0.000 description 1
- 238000002512 chemotherapy Methods 0.000 description 1
- 210000004978 chinese hamster ovary cell Anatomy 0.000 description 1
- XFIOKOXROGCUQX-UHFFFAOYSA-N chloroform;guanidine;phenol Chemical compound NC(N)=N.ClC(Cl)Cl.OC1=CC=CC=C1 XFIOKOXROGCUQX-UHFFFAOYSA-N 0.000 description 1
- 238000004587 chromatography analysis Methods 0.000 description 1
- 239000011651 chromium Substances 0.000 description 1
- 210000001072 colon Anatomy 0.000 description 1
- 238000010276 construction Methods 0.000 description 1
- 239000008120 corn starch Substances 0.000 description 1
- 210000004748 cultured cell Anatomy 0.000 description 1
- 230000016396 cytokine production Effects 0.000 description 1
- GVJHHUAWPYXKBD-UHFFFAOYSA-N d-alpha-tocopherol Natural products OC1=C(C)C(C)=C2OC(CCCC(C)CCCC(C)CCCC(C)C)(C)CCC2=C1C GVJHHUAWPYXKBD-UHFFFAOYSA-N 0.000 description 1
- 230000034994 death Effects 0.000 description 1
- 231100000517 death Toxicity 0.000 description 1
- 229940039227 diagnostic agent Drugs 0.000 description 1
- 239000000032 diagnostic agent Substances 0.000 description 1
- 238000000502 dialysis Methods 0.000 description 1
- PRAKJMSDJKAYCZ-UHFFFAOYSA-N dodecahydrosqualene Natural products CC(C)CCCC(C)CCCC(C)CCCCC(C)CCCC(C)CCCC(C)C PRAKJMSDJKAYCZ-UHFFFAOYSA-N 0.000 description 1
- 239000002552 dosage form Substances 0.000 description 1
- 241001493065 dsRNA viruses Species 0.000 description 1
- 230000008030 elimination Effects 0.000 description 1
- 238000003379 elimination reaction Methods 0.000 description 1
- 239000002158 endotoxin Substances 0.000 description 1
- 238000005516 engineering process Methods 0.000 description 1
- 238000001976 enzyme digestion Methods 0.000 description 1
- 238000003114 enzyme-linked immunosorbent spot assay Methods 0.000 description 1
- 239000006167 equilibration buffer Substances 0.000 description 1
- 238000011156 evaluation Methods 0.000 description 1
- 238000002474 experimental method Methods 0.000 description 1
- 239000013613 expression plasmid Substances 0.000 description 1
- 238000001914 filtration Methods 0.000 description 1
- 238000000684 flow cytometry Methods 0.000 description 1
- 125000005519 fluorenylmethyloxycarbonyl group Chemical group 0.000 description 1
- 238000009472 formulation Methods 0.000 description 1
- 238000002523 gelfiltration Methods 0.000 description 1
- 108020004445 glyceraldehyde-3-phosphate dehydrogenase Proteins 0.000 description 1
- 108010082286 glycyl-seryl-alanine Proteins 0.000 description 1
- 239000008187 granular material Substances 0.000 description 1
- 230000036541 health Effects 0.000 description 1
- 239000001307 helium Substances 0.000 description 1
- 229910052734 helium Inorganic materials 0.000 description 1
- SWQJXJOGLNCZEY-UHFFFAOYSA-N helium atom Chemical compound [He] SWQJXJOGLNCZEY-UHFFFAOYSA-N 0.000 description 1
- 210000003958 hematopoietic stem cell Anatomy 0.000 description 1
- 210000001320 hippocampus Anatomy 0.000 description 1
- 239000012510 hollow fiber Substances 0.000 description 1
- 102000047486 human GAPDH Human genes 0.000 description 1
- 230000002209 hydrophobic effect Effects 0.000 description 1
- 230000005847 immunogenicity Effects 0.000 description 1
- 230000016784 immunoglobulin production Effects 0.000 description 1
- 230000001976 improved effect Effects 0.000 description 1
- 239000000411 inducer Substances 0.000 description 1
- 238000011081 inoculation Methods 0.000 description 1
- 229940079322 interferon Drugs 0.000 description 1
- 229940047122 interleukins Drugs 0.000 description 1
- 230000003834 intracellular effect Effects 0.000 description 1
- 238000004255 ion exchange chromatography Methods 0.000 description 1
- 238000001155 isoelectric focusing Methods 0.000 description 1
- 230000006122 isoprenylation Effects 0.000 description 1
- 210000003292 kidney cell Anatomy 0.000 description 1
- 239000008101 lactose Substances 0.000 description 1
- 229920006008 lipopolysaccharide Polymers 0.000 description 1
- 210000001165 lymph node Anatomy 0.000 description 1
- 235000019359 magnesium stearate Nutrition 0.000 description 1
- 229930182817 methionine Natural products 0.000 description 1
- 238000000520 microinjection Methods 0.000 description 1
- 210000004688 microtubule Anatomy 0.000 description 1
- 238000010369 molecular cloning Methods 0.000 description 1
- 210000003205 muscle Anatomy 0.000 description 1
- 230000007498 myristoylation Effects 0.000 description 1
- 210000003757 neuroblast Anatomy 0.000 description 1
- 229910052759 nickel Inorganic materials 0.000 description 1
- 229920001220 nitrocellulos Polymers 0.000 description 1
- 235000020824 obesity Nutrition 0.000 description 1
- 210000000056 organ Anatomy 0.000 description 1
- 229940127084 other anti-cancer agent Drugs 0.000 description 1
- 239000008188 pellet Substances 0.000 description 1
- 239000008194 pharmaceutical composition Substances 0.000 description 1
- 230000026731 phosphorylation Effects 0.000 description 1
- 238000006366 phosphorylation reaction Methods 0.000 description 1
- 210000002826 placenta Anatomy 0.000 description 1
- 108700002563 poly ICLC Proteins 0.000 description 1
- 229940115270 poly iclc Drugs 0.000 description 1
- 229920001223 polyethylene glycol Polymers 0.000 description 1
- 239000000256 polyoxyethylene sorbitan monolaurate Substances 0.000 description 1
- 235000010486 polyoxyethylene sorbitan monolaurate Nutrition 0.000 description 1
- 239000004800 polyvinyl chloride Substances 0.000 description 1
- 229920000915 polyvinyl chloride Polymers 0.000 description 1
- 239000000843 powder Substances 0.000 description 1
- 239000002244 precipitate Substances 0.000 description 1
- 238000001556 precipitation Methods 0.000 description 1
- 238000002203 pretreatment Methods 0.000 description 1
- 230000003449 preventive effect Effects 0.000 description 1
- 239000000047 product Substances 0.000 description 1
- 238000011321 prophylaxis Methods 0.000 description 1
- 230000001681 protective effect Effects 0.000 description 1
- 239000001397 quillaja saponaria molina bark Substances 0.000 description 1
- 238000001959 radiotherapy Methods 0.000 description 1
- 239000011535 reaction buffer Substances 0.000 description 1
- 238000011160 research Methods 0.000 description 1
- 238000004366 reverse phase liquid chromatography Methods 0.000 description 1
- 238000005185 salting out Methods 0.000 description 1
- 229930182490 saponin Natural products 0.000 description 1
- 239000000377 silicon dioxide Substances 0.000 description 1
- 238000002415 sodium dodecyl sulfate polyacrylamide gel electrophoresis Methods 0.000 description 1
- 239000002904 solvent Substances 0.000 description 1
- 229940063673 spermidine Drugs 0.000 description 1
- 229940031439 squalene Drugs 0.000 description 1
- TUHBEKDERLKLEC-UHFFFAOYSA-N squalene Natural products CC(=CCCC(=CCCC(=CCCC=C(/C)CCC=C(/C)CC=C(C)C)C)C)C TUHBEKDERLKLEC-UHFFFAOYSA-N 0.000 description 1
- 208000013274 squamous cell breast carcinoma Diseases 0.000 description 1
- 238000010186 staining Methods 0.000 description 1
- 230000000638 stimulation Effects 0.000 description 1
- 239000000829 suppository Substances 0.000 description 1
- 239000004094 surface-active agent Substances 0.000 description 1
- 239000000725 suspension Substances 0.000 description 1
- 239000003826 tablet Substances 0.000 description 1
- 239000000454 talc Substances 0.000 description 1
- 229910052623 talc Inorganic materials 0.000 description 1
- 238000012360 testing method Methods 0.000 description 1
- 229960001295 tocopherol Drugs 0.000 description 1
- 229930003799 tocopherol Natural products 0.000 description 1
- 235000010384 tocopherol Nutrition 0.000 description 1
- 239000011732 tocopherol Substances 0.000 description 1
- 239000000196 tragacanth Substances 0.000 description 1
- 235000010487 tragacanth Nutrition 0.000 description 1
- 229940116362 tragacanth Drugs 0.000 description 1
- 241001529453 unidentified herpesvirus Species 0.000 description 1
- 238000002255 vaccination Methods 0.000 description 1
- 108700026220 vif Genes Proteins 0.000 description 1
- 239000013603 viral vector Substances 0.000 description 1
- 239000011782 vitamin Substances 0.000 description 1
- 229940088594 vitamin Drugs 0.000 description 1
- 229930003231 vitamin Natural products 0.000 description 1
- 235000013343 vitamin Nutrition 0.000 description 1
- XLYOFNOQVPJJNP-UHFFFAOYSA-N water Substances O XLYOFNOQVPJJNP-UHFFFAOYSA-N 0.000 description 1
- GVJHHUAWPYXKBD-IEOSBIPESA-N α-tocopherol Chemical compound OC1=C(C)C(C)=C2O[C@@](CCC[C@H](C)CCC[C@H](C)CCCC(C)C)(C)CCC2=C1C GVJHHUAWPYXKBD-IEOSBIPESA-N 0.000 description 1
Images
Classifications
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K39/00—Medicinal preparations containing antigens or antibodies
- A61K39/0005—Vertebrate antigens
- A61K39/0011—Cancer antigens
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K31/00—Medicinal preparations containing organic active ingredients
- A61K31/70—Carbohydrates; Sugars; Derivatives thereof
- A61K31/7088—Compounds having three or more nucleosides or nucleotides
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K38/00—Medicinal preparations containing peptides
- A61K38/16—Peptides having more than 20 amino acids; Gastrins; Somatostatins; Melanotropins; Derivatives thereof
- A61K38/17—Peptides having more than 20 amino acids; Gastrins; Somatostatins; Melanotropins; Derivatives thereof from animals; from humans
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K38/00—Medicinal preparations containing peptides
- A61K38/16—Peptides having more than 20 amino acids; Gastrins; Somatostatins; Melanotropins; Derivatives thereof
- A61K38/17—Peptides having more than 20 amino acids; Gastrins; Somatostatins; Melanotropins; Derivatives thereof from animals; from humans
- A61K38/1703—Peptides having more than 20 amino acids; Gastrins; Somatostatins; Melanotropins; Derivatives thereof from animals; from humans from vertebrates
- A61K38/1709—Peptides having more than 20 amino acids; Gastrins; Somatostatins; Melanotropins; Derivatives thereof from animals; from humans from vertebrates from mammals
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K38/00—Medicinal preparations containing peptides
- A61K38/16—Peptides having more than 20 amino acids; Gastrins; Somatostatins; Melanotropins; Derivatives thereof
- A61K38/17—Peptides having more than 20 amino acids; Gastrins; Somatostatins; Melanotropins; Derivatives thereof from animals; from humans
- A61K38/19—Cytokines; Lymphokines; Interferons
- A61K38/20—Interleukins [IL]
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K38/00—Medicinal preparations containing peptides
- A61K38/16—Peptides having more than 20 amino acids; Gastrins; Somatostatins; Melanotropins; Derivatives thereof
- A61K38/17—Peptides having more than 20 amino acids; Gastrins; Somatostatins; Melanotropins; Derivatives thereof from animals; from humans
- A61K38/19—Cytokines; Lymphokines; Interferons
- A61K38/20—Interleukins [IL]
- A61K38/208—IL-12
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K38/00—Medicinal preparations containing peptides
- A61K38/16—Peptides having more than 20 amino acids; Gastrins; Somatostatins; Melanotropins; Derivatives thereof
- A61K38/17—Peptides having more than 20 amino acids; Gastrins; Somatostatins; Melanotropins; Derivatives thereof from animals; from humans
- A61K38/19—Cytokines; Lymphokines; Interferons
- A61K38/21—Interferons [IFN]
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K38/00—Medicinal preparations containing peptides
- A61K38/16—Peptides having more than 20 amino acids; Gastrins; Somatostatins; Melanotropins; Derivatives thereof
- A61K38/17—Peptides having more than 20 amino acids; Gastrins; Somatostatins; Melanotropins; Derivatives thereof from animals; from humans
- A61K38/19—Cytokines; Lymphokines; Interferons
- A61K38/21—Interferons [IFN]
- A61K38/212—IFN-alpha
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K38/00—Medicinal preparations containing peptides
- A61K38/16—Peptides having more than 20 amino acids; Gastrins; Somatostatins; Melanotropins; Derivatives thereof
- A61K38/17—Peptides having more than 20 amino acids; Gastrins; Somatostatins; Melanotropins; Derivatives thereof from animals; from humans
- A61K38/19—Cytokines; Lymphokines; Interferons
- A61K38/21—Interferons [IFN]
- A61K38/215—IFN-beta
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K38/00—Medicinal preparations containing peptides
- A61K38/16—Peptides having more than 20 amino acids; Gastrins; Somatostatins; Melanotropins; Derivatives thereof
- A61K38/17—Peptides having more than 20 amino acids; Gastrins; Somatostatins; Melanotropins; Derivatives thereof from animals; from humans
- A61K38/19—Cytokines; Lymphokines; Interferons
- A61K38/21—Interferons [IFN]
- A61K38/217—IFN-gamma
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K39/00—Medicinal preparations containing antigens or antibodies
- A61K39/46—Cellular immunotherapy
- A61K39/461—Cellular immunotherapy characterised by the cell type used
- A61K39/4611—T-cells, e.g. tumor infiltrating lymphocytes [TIL], lymphokine-activated killer cells [LAK] or regulatory T cells [Treg]
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K39/00—Medicinal preparations containing antigens or antibodies
- A61K39/46—Cellular immunotherapy
- A61K39/464—Cellular immunotherapy characterised by the antigen targeted or presented
- A61K39/4643—Vertebrate antigens
- A61K39/4644—Cancer antigens
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K45/00—Medicinal preparations containing active ingredients not provided for in groups A61K31/00 - A61K41/00
- A61K45/06—Mixtures of active ingredients without chemical characterisation, e.g. antiphlogistics and cardiaca
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P35/00—Antineoplastic agents
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P35/00—Antineoplastic agents
- A61P35/02—Antineoplastic agents specific for leukemia
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P37/00—Drugs for immunological or allergic disorders
- A61P37/02—Immunomodulators
- A61P37/04—Immunostimulants
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P43/00—Drugs for specific purposes, not provided for in groups A61P1/00-A61P41/00
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07K—PEPTIDES
- C07K14/00—Peptides having more than 20 amino acids; Gastrins; Somatostatins; Melanotropins; Derivatives thereof
- C07K14/435—Peptides having more than 20 amino acids; Gastrins; Somatostatins; Melanotropins; Derivatives thereof from animals; from humans
- C07K14/46—Peptides having more than 20 amino acids; Gastrins; Somatostatins; Melanotropins; Derivatives thereof from animals; from humans from vertebrates
- C07K14/47—Peptides having more than 20 amino acids; Gastrins; Somatostatins; Melanotropins; Derivatives thereof from animals; from humans from vertebrates from mammals
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K39/00—Medicinal preparations containing antigens or antibodies
- A61K2039/51—Medicinal preparations containing antigens or antibodies comprising whole cells, viruses or DNA/RNA
- A61K2039/53—DNA (RNA) vaccination
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K38/00—Medicinal preparations containing peptides
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K39/00—Medicinal preparations containing antigens or antibodies
Definitions
- the present invention relates to a novel immunity-inducing agent useful as an agent for treating and / or preventing cancer.
- Cancer is a disease that occupies the top cause of all deaths, and the current treatment is a combination of radiation therapy and chemotherapy, mainly surgery. Despite the recent development of new surgical methods and the discovery of new anti-cancer drugs, the therapeutic results of cancer have not improved much except for some cancers. In recent years, advances in molecular biology and cancer immunology have identified cancer antigens recognized by cytotoxic T cells that respond to cancer and genes encoding cancer antigens, and expectations for antigen-specific immunotherapy have increased. Yes.
- Non-patent Document 1 In immunotherapy, in order to reduce side effects, peptides or proteins recognized as antigens are hardly present in normal cells but are required to be specifically present in cancer cells.
- Boon et al. Of Ludwig Laboratories in Belgium isolated human melanoma antigen MAGE1 recognized by CD8 positive T cells by a cDNA expression cloning method using autologous cancer cell lines and cancer reactive T cells (Non-patent Document 1). .
- SEREX seological identification of recombinant by expression cloning
- identifies a tumor antigen recognized by an antibody produced in response to own cancer in the body of a cancer patient by incorporating a gene expression cloning technique It has been reported (Patent Literature 1, Non-Patent Literature 2), and several cancer antigens have been isolated by this method. Furthermore, clinical trials for cancer immunotherapy have been started targeting some of them.
- dogs and cats are known to have many tumors such as breast tumors and squamous cell carcinomas, and are ranked high in the disease statistics of dogs and cats.
- tumors such as breast tumors and squamous cell carcinomas
- Most dogs and cat tumors are noticed by the owner after the tumor has progressed and the tumor has grown, and even if they come to the hospital and are removed by surgery or administered human drugs (such as anticancer drugs) Already too late to die soon after treatment. Under such circumstances, if cancer therapeutics and preventives effective for dogs and cats become available, it is expected that the use for canine cancer will be opened.
- Katanin p60 subunit A-like1 was identified as a protein having a microtubule binding domain (Patent Literature 2, Non-Patent Literature 3). However, there is no report that the KATNAL1 protein has immunity-inducing activity against cancer cells, whereby the protein is useful for treatment or prevention of cancer.
- An object of the present invention is to find a novel polypeptide useful as an agent for treating and / or preventing cancer, and to provide use of the polypeptide as an immunity-inducing agent.
- a cDNA encoding a protein that binds to an antibody present in serum derived from a cancer-bearing living body by a SEREX method using a dog testis-derived cDNA library and the serum of a cancer-bearing dog.
- a canine katanin p60 subunit A-like1 hereinafter referred to as KATNAL1
- SEQ ID NO: 2 was prepared.
- human and mouse KATNAL1 having the amino acid sequences shown in SEQ ID NOs: 4 and 6 were prepared.
- KATNAL1 polypeptides are specifically expressed in breast cancer, brain tumor, perianal adenocarcinoma, neuroblastoma, mastocytoma, liver cancer, prostate cancer, lung cancer, thyroid cancer or leukemia tissue or cell It was. Furthermore, it has been found that by administering these KATNAL1 to a living body, immune cells against KATNAL1 can be induced in the living body, and in vivo tumors expressing KATNAL1 can be regressed.
- a recombinant vector capable of expressing a polynucleotide encoding a KATNAL1 polypeptide or a fragment thereof induces an antitumor effect in vivo against a cancer expressing KATNAL1.
- the polypeptide of KATNAL1 is presented by antigen-presenting cells and has the ability to activate and proliferate cytotoxic T cells specific for the peptide (immunity-inducing activity). Is useful for the treatment and / or prevention of cancer, and antigen-presenting cells in contact with the polypeptide and T-cells in contact with the antigen-presenting cells are useful for the treatment and / or prevention of cancer.
- the present invention has been completed.
- the present invention has the following features. (1) comprising at least one polypeptide selected from the following polypeptides (a) to (c) and having immunity-inducing activity, or a polynucleotide encoding the polypeptide, An immunity-inducing agent comprising a recombinant vector capable of expressing the polypeptide as an active ingredient.
- An immunity-inducing agent comprising a recombinant vector capable of expressing the polypeptide as an active ingredient.
- a polypeptide comprising 7 or more consecutive amino acids in the amino acid sequence set forth in SEQ ID NOs: 4, 2, 8, 10, 12 in the sequence listing.
- the immunity-inducing agent according to (1) wherein the polypeptide having immunity-inducing activity is a polypeptide having an amino acid sequence described in SEQ ID NOs: 4, 2, 8, 10, 12 in the sequence listing.
- the immunity-inducing agent according to (1) or (2) which is a treatment agent for antigen-presenting cells.
- the immunity-inducing agent according to (1) or (2) which is a therapeutic and / or prophylactic agent for cancer.
- the immunity-inducing agent according to (4), wherein the cancer is a cancer that expresses KATNAL1.
- the immunopotentiator comprises Freund's incomplete adjuvant, Montanide, poly IC and derivatives thereof, CpG oligonucleotide, interleukin 12, interleukin 18, interferon ⁇ , interferon ⁇ , interferon ⁇ , interferon ⁇ , interferon ⁇ , and Flt3 ligand.
- the immunity-inducing agent according to (7) which is at least one selected from the group consisting of:
- the present invention provides a novel immunity-inducing agent useful for cancer treatment and / or prevention.
- a novel immunity-inducing agent useful for cancer treatment and / or prevention.
- the polypeptide used in the present invention when administered to a living body, immune cells can be induced in the living body, and further, cancer that has already occurred is reduced or retracted. be able to. Therefore, the polypeptide is useful for treating or preventing cancer.
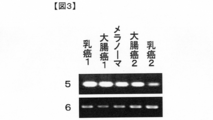
- Reference number 1 Expression pattern of canine KATNAL1 gene in each tissue and cell line of dog
- Reference number 2 Expression pattern of canine GAPDH gene in each tissue and cell line of dog. It is a figure which shows the expression pattern in the human normal tissue and a tumor tissue, or a cancer cell line of the identified KATNAL1 gene.
- polypeptide contained as an active ingredient in the immunity-inducing agent of the present invention examples include the following.
- polypeptide refers to a molecule formed by peptide bonding of a plurality of amino acids, and not only a polypeptide molecule having a large number of amino acids but also a low molecular weight molecule having a small number of amino acids.
- Oligopeptides and full-length proteins are also included.
- the full-length protein of KATNAL1 having the amino acid sequences shown in SEQ ID NOs: 2, 4, 8, 10, and 12 is also included.
- a polypeptide comprising an immunity-inducing activity consisting of seven or more consecutive amino acids in a polypeptide having the amino acid sequence shown in SEQ ID NO: 4, 2, 8, 10, 12 in the sequence listing (b) (a ) A polypeptide having 85% or more sequence identity with a polypeptide of (7) and having an immunity-inducing activity consisting of 7 or more amino acids (c) containing a polypeptide of (a) or (b) as a partial sequence, A polypeptide having immunity-inducing activity.
- “having an amino acid sequence” means that amino acid residues are arranged in such an order. Therefore, for example, “a polypeptide having the amino acid sequence represented by SEQ ID NO: 2” has the amino acid sequence of Met Asn Leu Ala covered and Glu Phe Gly Ser Ala represented by SEQ ID NO: 490. A polypeptide of the size of an amino acid residue is meant.
- “polypeptide having the amino acid sequence represented by SEQ ID NO: 2” may be abbreviated as “polypeptide of SEQ ID NO: 2”. The same applies to the expression “having a base sequence”. In this case, the term “comprising” may be replaced by the expression “consisting of”.
- immuno-inducing activity means the ability to induce immune cells that secrete cytokines such as interferon in vivo.
- Whether or not the above polypeptide has immunity-inducing activity can be confirmed using, for example, a known Elispot assay.
- cells such as peripheral blood mononuclear cells are obtained from a living body administered with a polypeptide whose immunity-inducing activity is to be evaluated, and the cells are referred to as the polypeptide.
- the number of immune cells in the cells can be measured by co-culturing and measuring the amount of cytokine production from the cells using a specific antibody, whereby the immunity-inducing activity can be evaluated.
- the tumor when the recombinant polypeptides (a) to (c) above are administered to a cancer-bearing organism, the tumor can be regressed due to its immunity-inducing activity. Therefore, the immunity-inducing activity can also be evaluated as the ability to suppress the growth of cancer cells or to reduce or eliminate cancer tissue (tumor) (hereinafter referred to as “anti-tumor activity”).
- anti-tumor activity The antitumor activity of a polypeptide is confirmed by, for example, examining whether the tumor is actually reduced by administering the polypeptide to a cancer-bearing living body, as specifically described in the examples below. can do.
- T cells stimulated with the polypeptide that is, T cells brought into contact with antigen-presenting cells presenting the polypeptide
- the antitumor activity of the polypeptide can also be evaluated.
- the T cell and the antigen-presenting cell can be contacted by co-culturing both in a liquid medium.
- the cytotoxic activity can be measured by a known method called a 51 Cr release assay described in, for example, Int. J. Cancer, 58: p317,1994.
- the polypeptide is used for cancer treatment and / or prevention, it is not particularly limited, but it is preferable to evaluate immunity induction activity using antitumor activity as an index.
- amino acid sequences shown in SEQ ID NOs: 2, 4, 8, 10, and 12 of the sequence listing disclosed by the present invention are derived from cancer-bearing dogs by the SEREX method using a dog testis-derived cDNA library and the serum of cancer-bearing dogs.
- 2 is an amino acid sequence of a KATNAL1 protein isolated as a polypeptide that binds to an antibody that specifically exists in the serum of the sera and as a human, bovine, horse, or chicken homologous factor (see Example 1).
- human KATNAL1 which is a human homologous factor of canine KATNAL1
- the sequence identity is 95% base sequence and amino acid sequence 98%.
- bovine KATNAL1 a bovine homologous factor, sequence identity is 91% base sequence, 97% amino acid sequence
- sequence identity is 87% and the amino acid sequence is 88%
- the sequence identity is 81% in the base sequence and 90% in the amino acid sequence.
- the polypeptide of the above (a) is 7 or more, preferably 8, 9 or 10 or more consecutive amino acids in the polypeptide having the amino acid sequence represented by SEQ ID NO: 2, 4, 8, 10, or 12.
- a polypeptide comprising immunity-inducing activity More preferably, the polypeptide is a polypeptide consisting of an amino acid sequence having a sequence identity of 85% or more with the amino acid sequence represented by SEQ ID NO: 4, and particularly preferably, the polypeptide is SEQ ID NO: 2, 4 , 8, 10, and 12.
- a polypeptide having about 7 amino acid residues or more can exhibit antigenicity and immunogenicity. Therefore, any polypeptide having 7 or more consecutive amino acid residues in the amino acid sequences of SEQ ID NOs: 2 and 4 can have immunity-inducing activity and can be used for preparing the immunity-inducing agent of the present invention.
- the polypeptide is taken up by antigen-presenting cells and then decomposed by peptidase in the cells to become smaller fragments and presented on the surface of the cells It is known that cytotoxic T cells and the like recognize this and selectively kill cells presenting the antigen.
- the size of the polypeptide presented on the surface of the antigen-presenting cell is relatively small, and is about 7 to 30 amino acids. Therefore, from the viewpoint of presentation on antigen-presenting cells, the polypeptide (a) is about 7 to 30 consecutive amino acids in the amino acid sequence represented by SEQ ID NOs: 2, 4, 8, 10, 12. This is one of the preferred embodiments, and more preferably it is composed of about 8 to 30 or 9 to 30 amino acids.
- These relatively small polypeptides may be presented directly on the cell surface on antigen-presenting cells without being taken up into antigen-presenting cells.
- Polypeptides taken up by antigen-presenting cells are cleaved at random positions by peptidases in the cells to generate various polypeptide fragments, and these polypeptide fragments are presented on the surface of antigen-presenting cells. Therefore, if a polypeptide having a large size such as the full-length region of SEQ ID NOs: 2, 4, 8, 10, 12 is administered, a polypeptide effective for inducing immunity via antigen-presenting cells due to degradation in the antigen-presenting cells. Peptide fragments are inevitably generated.
- a polypeptide having a large size can be preferably used for immunity induction via antigen-presenting cells, and the number of amino acids is 30 or more, more preferably 100 or more, more preferably 200 or more, more preferably 250 or more, and still more preferably.
- the polypeptide (b) is a polypeptide in which a small number (preferably one or several) of amino acid residues in the polypeptide (a) are substituted, deleted and / or inserted. It has 90% or more, preferably 95% or more, more preferably 98% or more, more preferably 99% or more or 99.5% or more sequence identity with the original sequence, and immunity-inducing activity
- a polypeptide having In general, a protein antigen may have almost the same antigenicity as the original protein even if a small number of amino acid residues in the amino acid sequence of the protein are substituted, deleted or inserted. It is well known to those skilled in the art.
- the polypeptide (b) can also exert immunity-inducing activity, it can be used for the preparation of the immunity-inducing agent of the present invention.
- the polypeptide (b) above one to several amino acid residues in the amino acid sequence represented by SEQ ID NOs: 2, 4, 8, 10, 12 are substituted, deleted and / or inserted. It is also preferred that the polypeptide is a modified polypeptide. “Several” in the present specification represents an integer of 2 to 10, preferably an integer of 2 to 6, and more preferably an integer of 2 to 4.
- sequence identity of amino acid sequences or base sequences means that both amino acid sequences (or bases) so that the amino acid residues (or bases) of the two amino acid sequences (or base sequences) to be compared match as much as possible. Sequence) and the number of matched amino acid residues (or the number of matched bases) divided by the total number of amino acid residues (or the total number of bases) is expressed as a percentage. In the above alignment, a gap is appropriately inserted in one or both of the two sequences to be compared as necessary. Such alignment of sequences can be performed using a known program such as BLAST, FASTA, CLUSTALW, and the like.
- the total number of amino acid residues is the number of residues obtained by counting one gap as one amino acid residue.
- the sequence identity (%) is the total number of amino acid residues in the longer sequence, and the amino acid residues that match. Calculated by dividing the number.
- the 20 kinds of amino acids constituting the natural protein include neutral amino acids having low polarity side chains (Gly, Ile, Val, Leu, Ala, Met, Pro), neutral amino acids having hydrophilic side chains (Asn). , Gln, Thr, Ser, Tyr, Cys), acidic amino acids (Asp, Glu), basic amino acids (Arg, Lys, His), and aromatic amino acids (Phe, Tyr, Trp) It is known that the properties of a polypeptide often do not change if substitution is made between these groups. Therefore, when substituting amino acid residues in the polypeptide of the above (a) of the present invention, substitution between these groups increases the possibility that immunity-inducing activity can be maintained, which is preferable. .
- the polypeptide (c) is a polypeptide that includes the polypeptide (a) or (b) as a partial sequence and has immunity-inducing activity. That is, it is a polypeptide obtained by adding another amino acid or polypeptide to one or both ends of the polypeptide (a) or (b) and having immunity-inducing activity.
- a polypeptide can also be used for the preparation of the immunity-inducing agent of the present invention.
- the above-mentioned polypeptides include, for example, the Fmoc method (fluorenylmethyloxycarbonyl method) and the tBoc method (t-butyloxycarbonyl method). It can synthesize
- polypeptide synthesized can also synthesize
- a polynucleotide encoding the above polypeptide is prepared, the polynucleotide is incorporated into an expression vector and introduced into a host cell, and the polypeptide is produced in the host cell.
- the desired polypeptide can be obtained.
- the polynucleotide encoding the above polypeptide can be easily prepared by a known genetic engineering technique or a conventional method using a commercially available nucleic acid synthesizer.
- DNA having the nucleotide sequence of SEQ ID NO: 1 is subjected to PCR using a canine chromosomal DNA or cDNA library as a template and a pair of primers designed to amplify the nucleotide sequence described in SEQ ID NO: 1.
- Any DNA having the nucleotide sequence of SEQ ID NO: 3 can be prepared in the same manner by using human chromosomal DNA or cDNA library as the template. PCR reaction conditions can be set as appropriate.
- one cycle of a reaction process consisting of 94 ° C. for 30 seconds (denaturation), 55 ° C. for 30 seconds to 1 minute (annealing), and 72 ° C. for 2 minutes (extension)
- the conditions include, but are not limited to, conditions of reacting at 72 ° C. for 7 minutes after 30 cycles.
- appropriate probes and primers are prepared based on the nucleotide sequence and amino acid sequence information shown in SEQ ID Nos. 1 and 3 in the sequence listing in the present specification, and a cDNA library for dogs, humans, etc. is prepared using them.
- the desired DNA can be isolated by screening for.
- the cDNA library is preferably prepared from cells, organs or tissues expressing the proteins of SEQ ID NOs: 2, 4.
- nucleotide sequence of the polynucleotide encoding the polypeptide (b) or (c) described above can be easily specified, such a polynucleotide can also be obtained by a conventional method using a commercially available nucleic acid synthesizer. It can be easily synthesized.
- the host cell may be any cell that can express the polypeptide.
- prokaryotic cells include Escherichia coli, and examples of eukaryotic cells include monkey kidney cells COS1, Chinese hamster ovary cells. Examples include, but are not limited to, cultured mammalian cells such as CHO, budding yeast, fission yeast, silkworm cells, and Xenopus egg cells.
- an expression vector having an origin, a promoter, a ribosome binding site, a DNA cloning site, a terminator and the like that can replicate in the prokaryotic cell is used as the expression vector.
- Examples of the expression vector for E. coli include pUC system, pBluescript II, pET expression system, pGEX expression system and the like.
- an expression vector for a eukaryotic cell having a promoter, a splicing region, a poly (A) addition site, etc. is used as the expression vector.
- expression vectors include pKA1, pCDM8, pSVK3, pMSG, pSVL, pBK-CMV, pBK-RSV, EBV vector, pRS, pcDNA3, pMSG, pYES2, and the like.
- a DNA encoding the above polypeptide is incorporated into such an expression vector, and after transforming a eukaryotic host cell with the vector, the resulting transformant is cultured, and the DNA encodes. Can be expressed in eukaryotic host cells.
- pIND / V5-His, pFLAG-CMV-2, pEGFP-N1, pEGFP-C1, etc. are used as an expression vector, a fusion protein to which various tags such as His tag, FLAG tag, myc tag HA tag, and GFP are added.
- the polypeptide can be expressed.
- a known method such as an electroporation method, a calcium phosphate method, a liposome method, or a DEAE dextran method can be used.
- Polypeptides obtained by the above methods include those in the form of fusion proteins with other arbitrary proteins as described above. Examples thereof include glutathione-S-transferase (GST) and a fusion protein with a His tag. A polypeptide in the form of such a fusion protein is also included in the scope of the present invention as the above-mentioned polypeptide (c). Furthermore, the polypeptide expressed in the transformed cell may be subjected to various modifications in the cell after being translated. Such post-translationally modified polypeptides are also included in the scope of the present invention as long as they have immunity-inducing activity. Examples of such translational modifications include elimination of N-terminal methionine, N-terminal acetylation, sugar chain addition, limited degradation by intracellular protease, myristoylation, isoprenylation, phosphorylation and the like.
- the immunity-inducing agent of the present invention can be used as a therapeutic and / or prophylactic agent for cancer.
- the above-mentioned polypeptide having immunity-inducing activity can be used in a method for treating and / or preventing cancer by immunity induction.
- the target cancer is not particularly limited as long as it expresses a gene encoding the polypeptide of SEQ ID NO: KATNAL1, but preferably breast cancer, brain tumor, perianal adenocarcinoma, neuroblastoma, obesity A cell tumor, liver cancer, prostate cancer, lung cancer, thyroid cancer or leukemia.
- the target animals are preferably mammals, more preferably mammals including primates, pet animals, domestic animals, sport animals, etc., particularly preferably humans, dogs or cats.
- the administration route to the living body of the immunity-inducing agent of the present invention may be oral administration or parenteral administration, but parenteral administration such as intramuscular administration, subcutaneous administration, intravenous administration and intraarterial administration is preferred.
- parenteral administration such as intramuscular administration, subcutaneous administration, intravenous administration and intraarterial administration is preferred.
- the immunity-inducing agent When used for the purpose of treating cancer, it can be administered to a regional lymph node in the vicinity of the tumor to be treated, as described in Examples below, in order to enhance the anticancer effect.
- the dose may be an amount effective for inducing immunity.
- it may be an amount effective for the treatment and / or prevention of cancer.
- the amount effective for the treatment and / or prevention of cancer is appropriately selected according to the size, symptoms, etc. of the tumor.
- the effective amount per day for the subject animal is 0.0001 to 1000 ⁇ g, preferably 0.
- the dose is 001 to 1000 ⁇ g and can be administered once or divided into several times. Preferably, it is divided into several times and administered every few days to several months.
- the immunity-inducing agent of the present invention can regress an already formed tumor. Therefore, since anticancer activity can be exerted on a small number of cancer cells at the early stage of development, onset and recurrence of cancer can be prevented if used before onset of cancer or after treatment of cancer. That is, the immunity-inducing agent of the present invention is useful for both treatment and prevention of cancer.
- the immunity-inducing agent of the present invention may consist only of a polypeptide, or may be appropriately mixed with additives such as pharmacologically acceptable carriers, diluents and excipients suitable for each administration form. It can also be formulated. Formulation methods and usable additives are well known in the field of pharmaceutical formulations, and any method and additive can be used. Specific examples of additives include diluents such as physiological buffers; excipients such as sugar, lactose, corn starch, calcium phosphate, sorbitol, glycine; syrup, gelatin, gum arabic, sorbitol, polyvinyl chloride, tragacanth, etc.
- additives include diluents such as physiological buffers; excipients such as sugar, lactose, corn starch, calcium phosphate, sorbitol, glycine; syrup, gelatin, gum arabic, sorbitol, polyvinyl chloride, tragacanth, etc.
- Binders such as magnesium stearate, polyethylene glycol, talc, silica and the like, but are not limited thereto.
- the dosage form include oral preparations such as tablets, capsules, granules, powders, and syrups, and parenteral preparations such as inhalants, injections, suppositories, and liquids. These preparations can be made by generally known production methods.
- the immunity-inducing agent of the present invention can be used in combination with an immunity enhancing agent capable of enhancing the immunological response in vivo.
- the immunopotentiator may be contained in the immunity-inducing agent of the present invention, or may be administered to a patient in combination with the immunity-inducing agent of the present invention as a separate composition.
- the above immunopotentiator examples include an adjuvant.
- Adjuvants provide a reservoir of antigen (extracellular or within macrophages) and can enhance immunological responses by activating macrophages and stimulating specific sets of lymphocytes, thus enhancing anticancer effects be able to. Therefore, in particular, when the immunity-inducing agent of the present invention is used for the treatment and / or prevention of cancer, it is preferable that the immunity-inducing agent further contains an adjuvant in addition to the above polypeptide as an active ingredient. Many types of adjuvants are well known in the art, and any adjuvant can be used.
- adjuvants include MPL (SmithKline Beecham), Salmonella minnesotaoRe 595 lipopolysaccharide and similar products obtained after acid hydrolysis; QA-21 saponin; DQS21 described in PCT application WO 96/33739 (SmithKline Beecham); QS-7, QS-17, QS-18 and QS-L1 (So, outside, 10 people, “Molecules and Cells (Molecules and cells), 1997, Vol. 7, p. 178-186); Freund's incomplete adjuvant; Freund's complete adjuvant; vitamins Montanide; Alum; CpG oligonucleotides (see, for example, Craig, et al., “Nature”, Vol.
- the mixing ratio of the adjuvant to the polypeptide is typically about 1:10 to 10: 1, preferably about 1: 5 to 5: 1, more preferably about 1: 1.
- adjuvants are not limited to the above examples, and other adjuvants known in the art may be used when administering the immunity-inducing agent of the present invention (for example, “Goding”, “Monoclonal antibodies: Principles and Practices (see Monoclonal Antibodies: Principles and Practices, 2nd edition, 1986). Methods for preparing polypeptide and adjuvant mixtures or emulsions are well known to those skilled in vaccination.
- factors that stimulate the immune response of the subject can also be used as the immune enhancer.
- various cytokines having the property of stimulating lymphocytes and antigen-presenting cells can be used in combination with the immune inducer of the present invention as an immune enhancer.
- Numerous cytokines capable of enhancing such immunological responses are known to those skilled in the art, such as interleukin-12 (IL-12), which has been shown to enhance the protective effects of vaccines, Examples include, but are not limited to, GM-CSF, IL-18, interferon ⁇ , interferon ⁇ , interferon ⁇ , interferon ⁇ , and Flt3 ligand.
- IL-12 interleukin-12
- Such factors can also be used as the above-mentioned immunity enhancing agent, and can be administered to patients in the immunity-inducing agent of the present invention or in combination with the immunity-inducing agent of the present invention as a separate composition.
- the polypeptide can be presented to the antigen-presenting cell by contacting the polypeptide with the antigen-presenting cell in vitro. That is, the above-described polypeptides (a) to (c) can be used as a treatment agent for antigen-presenting cells.
- dendritic cells or B cells carrying MHC class I molecules can be preferably used as antigen-presenting cells.
- MHC class I molecules have been identified and are well known. MHC molecules in humans are called HLA. Examples of HLA class I molecules include HLA-A, HLA-B, and HLA-C.
- HLA-A1 HLA-A0201, HLA-A0204, HLA-A0205, HLA-A0206, HLA-A0207, HLA-A11, HLA-A24, HLA-A31, HLA-A6801, HLA-B7, HLA-B8, HLA-B2705, HLA-B37, HLA-Cw0401, HLA-Cw0602 and the like.
- Dendritic cells or B cells carrying MHC class I molecules can be prepared from peripheral blood by well-known methods. For example, dendritic cells are induced from bone marrow, umbilical cord blood or patient peripheral blood using granulocyte-macrophage colony-stimulating factor (GM-CSF) and IL-3 (or IL-4), and tumor-related peptides are introduced into the culture system. Can be added to induce tumor-specific dendritic cells.
- GM-CSF granulocyte-macrophage colony-stimulating factor
- IL-3 or IL-4
- the cells used can be bone marrow and umbilical cord blood provided by healthy people, the patient's own bone marrow and peripheral blood, etc., but when using the patient's own autologous cells, it is highly safe and has serious side effects. It can also be expected to avoid it.
- Peripheral blood or bone marrow may be a fresh sample, a cryopreserved sample, or a cryopreserved sample.
- whole blood may be cultured, or only the leukocyte component may be separated and cultured, but the latter is more efficient and preferable.
- mononuclear cells may be separated among the leukocyte components.
- the whole cells constituting the bone marrow may be cultured, or mononuclear cells may be separated and cultured from this.
- Peripheral blood, its white blood cell components, and bone marrow cells include mononuclear cells, hematopoietic stem cells, immature dendritic cells, CD4 positive cells, and the like that are the origin of dendritic cells.
- the cytokine used has the characteristics that have been confirmed to be safe and physiologically active, it does not matter whether it is a natural type or a genetically modified type, and its production method is preferably ensured. Are used in the minimum amount required.
- the concentration of the cytokine to be added is not particularly limited as long as it is a concentration at which dendritic cells are induced.
- the total concentration of cytokines is preferably about 10 to 1000 ng / mL, more preferably about 20 to 500 ng / mL.
- the culture can be performed using a well-known medium usually used for culturing leukocytes.
- the culture temperature is not particularly limited as long as leukocyte growth is possible, but is most preferably about 37 ° C. which is the human body temperature.
- the gas environment during the culture is not particularly limited as long as leukocytes can be grown, but it is preferable to aerate 5% CO 2 .
- the culture period is not particularly limited as long as a necessary number of cells are induced, but it is usually performed for 3 days to 2 weeks.
- an appropriate device can be used as appropriate. However, it is preferable that safety is confirmed for medical use and the operation is stable and simple.
- laminated containers, multistage containers, roller bottles, spinner bottles, bag type incubators, hollow fiber columns, etc. can be used regardless of general containers such as petri dishes, flasks, and bottles. .
- the method of bringing the polypeptide into contact with the antigen-presenting cell in vitro can be performed by a well-known method. For example, it can be performed by culturing antigen-presenting cells in a culture solution containing the polypeptide.
- the peptide concentration in the medium is not particularly limited, but is usually about 1 to 100 ⁇ g / ml, preferably about 5 to 20 ⁇ g / ml.
- the cell density during the culture is not particularly limited, but is usually about 10 3 to 10 7 cells / ml, preferably about 5 ⁇ 10 4 to 5 ⁇ 10 6 cells / ml. Cultivation is preferably performed according to a conventional method in a 37 ° C., 5% CO 2 atmosphere.
- the length of the peptide that can be presented on the surface by antigen-presenting cells is usually about 30 amino acid residues at the maximum. Therefore, although not particularly limited, when the antigen-presenting cell and the polypeptide are contacted in vitro, the polypeptide may be prepared to a length of about 30 amino acid residues or less.
- an isolated antigen-presenting cell containing a complex of the polypeptide and MHC molecule can be prepared using the polypeptide.
- Such antigen-presenting cells can present the polypeptide to T cells in vivo or in vitro, and induce and proliferate cytotoxic T cells specific for the polypeptide.
- Antigen-presenting cells containing the above-described polypeptide and MHC molecule complex prepared as described above are contacted with T cells in vitro to induce cytotoxic T cells specific for the polypeptide. And can be grown. This can be done by co-culturing the antigen-presenting cells and T cells in a liquid medium. For example, it can be carried out by suspending antigen-presenting cells in a liquid medium, placing them in a container such as a well of a microplate, adding T cells thereto, and culturing.
- the mixing ratio of antigen-presenting cells and T cells during co-culture is not particularly limited, but is usually about 1: 1 to 1: 100, preferably about 1: 5 to 1:20 in terms of the number of cells.
- the density of antigen-presenting cells suspended in the liquid medium is not particularly limited, but is usually about 1 to 10 million cells / ml, preferably about 10,000 to 1 million cells / ml.
- the coculture is preferably performed in a 37 ° C., 5% CO 2 atmosphere according to a conventional method.
- the culture time is not particularly limited, but is usually 2 days to 3 weeks, preferably about 4 days to 2 weeks.
- the coculture is preferably performed in the presence of one or more interleukins such as IL-2, IL-6, IL-7 and IL-12.
- the concentration of IL-2 and IL-7 is usually about 5 to 20 U / ml
- the concentration of IL-6 is usually about 500 to 2000 U / ml
- the concentration of IL-12 is usually about 5 to 20 ng / ml.
- the above co-culture may be repeated once to several times by adding fresh antigen-presenting cells. For example, the operation of discarding the culture supernatant after co-culture, adding a fresh antigen-presenting cell suspension, and further co-culturing may be repeated once to several times.
- the conditions for each co-culture may be the same as described above.
- cytotoxic T cells specific for the polypeptide are induced and proliferated. Therefore, an isolated T cell that selectively binds the complex of the polypeptide and the MHC molecule can be prepared using the polypeptide.
- the KATNAL1 gene is used for breast cancer cells, breast cancer tissues, brain tumor cells, brain tumor tissues, perianal adenocarcinoma tissues, perianal adenocarcinoma cells, mastocytoma tissues, mastocytoma cells, neuroblasts It is specifically expressed in tumor cells, liver cancer cells, liver cancer tissues, prostate cancer cells, prostate cancer tissues, lung cancer cells, lung cancer tissues, thyroid cancer cells, thyroid cancer tissues and leukemia cells. Therefore, it is considered that KATNAL1 gene is present in significantly more than normal cells in these cancer types.
- cytotoxic T cells can damage cancer cells.
- the antigen-presenting cells presenting the polypeptide can induce and proliferate cytotoxic T cells specific for the polypeptide in vivo, the antigen-presenting cells are administered in vivo. In some cases, cancer cells can be damaged. That is, the cytotoxic T cell and the antigen-presenting cell prepared using the polypeptide are also useful as a cancer treatment and / or prevention agent, like the immunity-inducing agent of the present invention.
- the administration route of the therapeutic and / or prophylactic agent for cancer containing antigen-presenting cells or isolated T cells as an active ingredient is preferably parenteral administration such as intravenous administration or intraarterial administration.
- the dose is appropriately selected according to symptoms, administration purposes, etc., but is usually 1 to 10 trillion, preferably 1 million to 1 billion, and this is once every few days to several months. Administration is preferred.
- the preparation may be, for example, one in which cells are suspended in physiological buffer saline, and can be used in combination with other anticancer agents, cytokines, and the like.
- one or two or more additives well known in the pharmaceutical field can be added.
- the immunity-inducing agent of the present invention comprises a polynucleotide encoding the above-described polypeptides (a) to (c), and contains a recombinant vector capable of expressing the polypeptide in vivo as an active ingredient. There may be. As shown in the Examples described below, a recombinant vector capable of expressing such an antigen polypeptide is also called a gene vaccine.
- the vector used for producing the gene vaccine is not particularly limited as long as it can be expressed in the target animal cell (preferably in the mammalian cell), and may be a plasmid vector or a virus vector, and is known in the field of gene vaccines. Any of these vectors may be used.
- a polynucleotide such as DNA or RNA encoding the polypeptide can be easily prepared by a conventional method.
- the polynucleotide can be incorporated into a vector by a method well known to those skilled in the art.
- the administration route of the gene vaccine is preferably a parenteral administration route such as intramuscular administration, subcutaneous administration, intravenous administration or intraarterial administration, and the dosage can be appropriately selected according to the type of antigen and the like.
- the weight of the gene vaccine per kg of body weight is about 0.1 ⁇ g to 100 mg, preferably about 1 ⁇ g to 10 mg.
- a polynucleotide encoding the above polypeptide in RNA virus or DNA virus such as retrovirus, adenovirus, adeno-associated virus, herpes virus, vaccinia virus, poxvirus, poliovirus, Sindbis virus, etc. And a method for infecting a target animal with this.
- retroviruses, adenoviruses, adeno-associated viruses, vaccinia viruses and the like are particularly preferred.
- Examples of other methods include a method in which an expression plasmid is directly administered into muscle (DNA vaccine method), a liposome method, a lipofectin method, a microinjection method, a calcium phosphate method, an electroporation method, and the like. The method is preferred.
- an in vivo method for directly introducing the gene into the body, and collecting a certain cell from the target animal and transferring the gene to the cell outside the body There is an ex vivo method that introduces the cells into the body and returns the cells to the body (Nikkei Science, April 1994, p20-45, Monthly Pharmaceutical Affairs, 1994, Vol. 36, No. 1, p. 23-48, Experimental Medicine) Special edition, 1994, Vol. 12, No. 15, and references cited therein), in vivo method is more preferable.
- an in vivo method When administered by an in vivo method, it can be administered by an appropriate administration route according to the disease or symptom or the like for the purpose of treatment. For example, it can be administered intravenously, artery, subcutaneously, intramuscularly. In the case of administration by an in vivo method, for example, it can be in the form of a preparation such as a liquid, but is generally an injection containing the DNA encoding the peptide of the present invention, which is an active ingredient. Depending on the case, customary carriers may be added.
- the liposome or membrane-fused liposome containing the DNA can be in the form of a liposome preparation such as a suspension, a freezing agent, or a centrifugal concentrated freezing agent.
- base sequence shown in SEQ ID NO: 1 includes the base sequence actually shown in SEQ ID NO: 1 as well as a complementary sequence thereto. Therefore, when saying “polynucleotide having the base sequence shown in SEQ ID NO: 1”, the single-stranded polynucleotide having the base sequence actually shown in SEQ ID NO: 1, its complementary base sequence Single-stranded polynucleotides having and double-stranded polynucleotides comprising these are included.
- any base sequence is appropriately selected, but those skilled in the art can easily select it.
- Example 1 Acquisition of novel cancer antigen protein by SEREX method (1) Preparation of cDNA library Total RNA was extracted from dog testis by acid-guanidinium-phenol-chloroform method (Acid guanidinium-phenol-chloroform method), and Oligotex -PolyA RNA was purified using dT30 mRNA purification Kit (Takara Shuzo Co., Ltd.) according to the protocol attached to the kit.
- a cDNA phage library was synthesized using the obtained mRNA (5 ⁇ g).
- the cDNA phage library was prepared by using cDNA Synthesis kit, Zap-cDNA Synthesis Kit, ZAP-cDNA GigapackIII Gold Cloning Kit (manufactured by STRATAGENE) according to the protocol attached to the kit.
- the size of the prepared cDNA phage library was 1 ⁇ 10 6 pfu / ml.
- the membrane was recovered, immersed in TBS (10 mM Tris-HCl, 150 mM NaCl pH 7.5) containing 0.5% nonfat dry milk, and shaken at 4 ° C. overnight to suppress nonspecific reaction.
- TBS 10 mM Tris-HCl, 150 mM NaCl pH 7.5
- This filter was reacted with serum of a patient dog diluted 500 times at room temperature for 2 to 3 hours.
- the serum pretreatment method is as follows. That is, ⁇ ZAP Express phage into which no foreign gene was inserted was infected with host E. coli (XL1-BLue MRF ′), and then cultured overnight at 37 ° C. on NZY plate medium. Next, a buffer of 0.2 M NaHCO 3 pH 8.3 containing 0.5 M NaCl was added to the plate and allowed to stand at 4 ° C. for 15 hours, and then the supernatant was recovered as an E. coli / phage extract. Next, the recovered E.
- coli / phage extract was passed through an NHS column (GE Healthcare Bio-Science) to immobilize the E. coli / phage derived protein.
- Serum dog serum was passed through and reacted with this protein-immobilized column, and antibodies adsorbed to E. coli and phage were removed from the serum.
- the serum fraction passed through the column was diluted 500 times with TBS containing 0.5% nonfat dry milk, and this was used as an immunoscreening material.
- the purified plasmid was analyzed for the full-length insert sequence by the primer walking method using the T3 primer shown in SEQ ID NO: 13 and the T7 primer shown in SEQ ID NO: 14.
- the gene sequence described in SEQ ID NO: 1 was obtained.
- a sequence identity search program BLAST search http://www.ncbi.nlm.nih.gov/BLAST/
- BLAST search http://www.ncbi.nlm.nih.gov/BLAST/
- sequence identity is 95% base sequence and amino acid sequence 98%.
- sequence identity is 85% base sequence, 94% amino acid sequence
- sequence identity is 91% nucleotide sequence, 97% amino acid sequence
- sequence identity is 87%, amino acid sequence 88%, chicken homologous chicken In KATNAL1, the sequence identity was 81% base sequence and 90% amino acid sequence.
- the human KATNAL1 base sequence is SEQ ID NO: 3, the amino acid sequence is SEQ ID NO: 4, the mouse KATNAL1 base sequence is SEQ ID NO: 5, the amino acid sequence is SEQ ID NO: 6, the bovine KATNAL1 base sequence is SEQ ID NO: 7, and the amino acid sequence is sequenced.
- the base sequence of horse KATNAL1 is shown in SEQ ID NO: 9
- the amino acid sequence is shown in SEQ ID NO: 10
- the base sequence of chicken KATNAL1 is shown in SEQ ID NO: 11
- the amino acid sequence is shown in SEQ ID NO: 12.
- Gene pool cDNA (manufactured by Invitrogen), QUICK-Clone cDNA (manufactured by Clontech) and Large-Insert cDNA Library (manufactured by Clontech) were used as cDNAs for human normal tissues (brain, hippocampus, testis, colon, placenta). .
- the PCR reaction was carried out using the obtained gene-specific primers (SEQ ID NOs: 15 and 16 for dog primers, SEQ ID NOs: 17 and 18 for human primers, and SEQ ID NOs: 19 and 20 for mouse primers) as follows.
- the canine KATNAL1 gene was not expressed in most tissues in healthy dog tissues, whereas strong expression was observed in dog tumor tissues.
- the expression of the human and mouse KATNAL1 genes was hardly confirmed in human and mouse normal tissues as in the canine KATNAL1 gene, and expression was detected in most cell lines in cancer cells (FIGS. 2 and 3).
- KATNAL1 gene was 5 times or more higher in breast cancer, colon cancer, thyroid cancer, liver cancer, prostate cancer, and lung cancer than in each normal tissue. This result shows that the antitumor agent targeting human KATNAL1 is expected to have a great difference between the drug efficacy and side effects without worrying about side effects in normal tissues.
- Example 2 Analysis of cancer antigenicity of KATNAL1 in vivo (1) Preparation of recombinant vector expressing KATNAL1 in vivo KATNAL1 in vivo by the following method based on the nucleotide sequence of SEQ ID NO: 5 A recombinant vector that expresses was produced. PCR was prepared from the mouse cancer cell line N2a (purchased from ATCC) in which expression was observed in Example 1.
- Primer HS polymerase (manufactured by Takara Shuzo Co., Ltd.) 1 ⁇ l of cDNA, two kinds of primers (described in SEQ ID NOs: 25 and 26) containing HindIII and XbaI restriction enzyme cleavage sequences, 0.4 ⁇ M, 0.2 mM dNTP, 1.25 U Add each reagent and the attached buffer so that the total volume becomes 50 ⁇ l, and use Thermal Cycler (manufactured by BIO RAD), 30 cycles of 98 ° C-10 seconds, 55 ° C-15 seconds, 72 ° C-4 minutes This was done by repeating.
- the above two types of primers amplify the region encoding the entire amino acid sequence of SEQ ID NO: 5. After PCR, the amplified DNA was electrophoresed on a 1% agarose gel, and a DNA fragment of about 1500 bp was purified using QIAquick Gel Extraction Kit (manufactured by QIAGEN).
- the purified DNA fragment was ligated to the cloning vector pCR-Blunt (manufactured by Invitrogen). After transforming this into E. coli, the plasmid was recovered, and it was confirmed by sequencing that the amplified gene fragment matched the target sequence.
- a plasmid corresponding to the target sequence was treated with HindIII and XbaI restriction enzymes, purified with QIAquick Gel Extraction Kit, and then inserted into a mammalian expression vector pcDNA3.1 (manufactured by Invitrogen) treated with HindIII and XbaI restriction enzymes. did.
- the use of this vector produces KATNAL1 protein in mammalian cells.
- gold particles 50 ⁇ g of gold particles (manufactured by Bio Rad), 100 ⁇ l of spermidine (manufactured by SIGMA), and 100 ⁇ l of 1M CaCl 2 (manufactured by SIGMA) were added to 100 ⁇ g of the plasmid DNA prepared above, and the mixture was stirred by vortexing and allowed to stand at room temperature for 10 minutes. (Hereinafter referred to as gold-DNA particles). After centrifugation at 3000 rpm for 1 minute, the supernatant was discarded and washed 3 times with 100% ethanol (manufactured by WAKO).
- Tefzel Tubing manufactured by Bio Rad
- Example 3 Production of human recombinant KATNAL1 protein and evaluation of immunity induction ability
- the purified DNA fragment was ligated to the cloning vector pCR-Blunt (manufactured by Invitrogen). After transforming this into E. coli, the plasmid was recovered, and it was confirmed by sequencing that the amplified gene fragment matched the target sequence.
- a plasmid corresponding to the target sequence was treated with EcoRI and XhoI restriction enzymes, purified with QIAquick Gel Extraction Kit, and then inserted into an expression vector pET30a (manufactured by Novagen) for Escherichia coli treated with EcoRI and XhoI restriction enzymes. .
- This vector a His-tagged recombinant protein can be produced.
- This plasmid was transformed into E. coli BL21 (DE3) for expression, and the target protein was expressed in E. coli by inducing expression with 1 mM IPTG.
- the cells were suspended in 50 mM Tris-HCl buffer (pH 8.0) and sonicated on ice.
- the E. coli sonication solution was centrifuged at 6000 rpm for 20 minutes, and the resulting supernatant was used as a soluble fraction and the precipitate was used as an insoluble fraction.
- the insoluble fraction was suspended in 50 mM Tris-HCl buffer (pH 8.0) and centrifuged at 6000 rpm for 15 minutes. This operation was repeated twice to perform a protease removal operation.
- the column unadsorbed fraction was washed with 0.5 M sodium chloride-containing 0.1 M acetate buffer (pH 4.0), 10 times the volume of the column, and immediately, 0.5 M sodium chloride-containing 0.1 M Elution was performed with an acetate buffer (pH 3.0) to obtain a purified fraction, and this purified fraction was subsequently used as a material for administration tests.
- the target protein in each elution fraction was confirmed by Coomassie staining performed according to a conventional method.
- the purified sample obtained by the above method was replaced with a reaction buffer solution (50 mM Tris-HCl, 100 mM NaCl, 5 mM CaCl 2 (pH 8.0)), and attached using FactorXa Cleavage Capture Kit (manufactured by Novagen).
- the His tag was cleaved with Factor Xa protease and the target protein was purified.
- 12 ml of the purified sample obtained by the above method was replaced with a physiological phosphate buffer (manufactured by Nissui Pharmaceutical) using ultrafiltration NANOSEP 10K OMEGA (manufactured by PALL), and then HT tafrin. Aseptic filtration was performed with Acrodisc 0.22 ⁇ m (manufactured by PALL), and this was used for the experiment.
- KATNAL1 protein-reactive CD8 positive cytotoxic T cells Peripheral blood was isolated from a healthy person and overlaid on Lymphocyte separation medium (OrganonpTeknika, Durham, NC) at 1,500 rpm at room temperature. Centrifuged for minutes. Fractions containing peripheral blood mononuclear cells (PBMC) were collected and washed three times (or more) in cold phosphate buffer to obtain PBMC. The obtained PBMC was suspended in 20 ml of AIM-V medium (Life Technologies, Inc., Grand Island, NY, USA) and allowed to attach in a culture flask (Falcon) for 2 hours under conditions of 37 ° C. and 5% CO 2 . . Non-adherent cells were used for T cell preparation, and adherent cells were used to prepare dendritic cells.
- Lymphocyte separation medium OrganonpTeknika, Durham, NC
- AIM-V medium Life Technologies, Inc., Grand Island, NY, USA
- adherent cells were cultured in AIM-V medium in the presence of IL-4 (1000 U / ml) and GM-CSF (1000 U / ml).
- the non-adherent cell population obtained after 6 days was collected, human recombinant KATNAL1 protein was added at a concentration of 10 ⁇ g / ml, and cultured at 37 ° C. under 5% CO 2 for 4 hours.
- IL-4 1000 U / ml
- GM-CSF 1000 U / ml
- IL-6 1000 U / ml, Genzyme, Cambridge, MA
- IL-1 ⁇ 10 ng / ml, Genzyme, Cambridge, MA
- the prepared dendritic cells were suspended in AIM-V medium at a cell density of 1 ⁇ 10 6 cells / ml, human recombinant KATNAL1 protein was added again at a concentration of 10 ⁇ g / ml, and 37 ° C. using a 96-well plate. The cells were cultured for 4 hours under the condition of 5% CO 2 .
- each culture supernatant was discarded and dendritic cells treated with human recombinant protein and X-irradiated in the same manner as above were treated with 10% human AB serum (Nabi, Miami, FL), IL-7 (10 U / ml). , Genzyme, Cambridge, MA) and suspended in AIM-V medium containing IL-2 (10 U / ml, Genzyme, Cambridge, MA) (cell density: 1 ⁇ 10 5 cells / ml), 24-well plate 1 well 1 ⁇ 10 5 cells were added per cell and further cultured. The same operation was repeated 4 to 6 times every 7 days, and then the stimulated T cells were collected, and the induction of CD8 positive T cells was confirmed by flow cytometry.
- T98G (Stein GH et al, J. Cell Physiol, 99:.. 43-54 (1979), purchased from ATCC) were collected 10 5 to centrifuge tube 50ml capacity 100 ⁇ Ci of chromium 51 was added and incubated at 37 ° C. for 2 hours. Thereafter, the plate was washed 3 times with AIM-V medium containing 10% human AB serum, added 10 3 times per well of 96-well V-bottom plate, and further suspended in AIM-V medium containing 10% human AB serum.
- CD8 positive T cells stimulated with human recombinant protein have cytotoxic activity against T98G.
- CD8 positive T cells induced with a negative control protein did not show cytotoxic activity. Therefore, it was revealed that the human recombinant protein used in the present invention has the ability to induce CD8 positive cytotoxic T cells capable of damaging tumor cells.
- the cytotoxic activity was determined by mixing 10 5 stimulation-induced CD8 positive T cells and 10 3 malignant brain tumor cell lines T98G incorporated with chromium 51, and culturing for 4 hours. The amount of chromium 51 released into the medium is measured, and the cytotoxic activity of CD8 positive T cells calculated by Formula 1 against T98G is shown.
- Cytotoxic activity (%) Chromium 51 release from T98G (cpm) / CD51 release from target cells added with 1N hydrochloric acid (cpm) ⁇ 100 when CD8 positive T cells are added.
- the present invention is useful for the treatment and / or prevention of cancer because it provides an immunity-inducing agent containing a polypeptide that exhibits antitumor activity against various cancers.
Abstract
Description
(1)以下の(a)ないし(c)のいずれかのポリペプチド類から選択され、かつ免疫誘導活性を有する少なくとも1つのポリペプチド、又は該ポリペプチドをコードするポリヌクレオチドを含み、生体内で該ポリペプチドを発現可能な組換えベクター、を有効成分として含有する免疫誘導剤。
(a)配列表の配列番号4、2、8、10、12に記載のアミノ酸配列中の連続する7個以上のアミノ酸から成るポリペプチド。
(b)前記(a)のポリペプチドと85%以上の配列同一性を有し、かつ7個以上のアミノ酸から成るポリペプチド。
(c)前記(a)又は(b)のポリペプチドを部分配列として含むポリペプチド。
(2)前記免疫誘導活性を有するポリペプチドが配列表の配列番号4、2、8、10、12記載のアミノ酸配列を有するポリペプチドである、(1)に記載の免疫誘導剤。
(3)抗原提示細胞の処理剤である、(1)又は(2)記載の免疫誘導剤。
(4)癌の治療及び/又は予防剤である、(1)又は(2)に記載の免疫誘導剤。
(5)前記癌がKATNAL1を発現する癌である、(4)に記載の免疫誘導剤。
(6)前記癌が乳癌、脳腫瘍、肛門周囲腺癌、神経芽腫、肥満細胞腫、肝臓癌、前立腺癌、肺癌、甲状腺癌又は白血病である、(4)又は(5)に記載の免疫誘導剤。
(7)免疫増強剤をさらに含む(1)~(6)のいずれかに記載の免疫誘導剤。
(8)前記免疫増強剤が、フロイントの不完全アジュバント、モンタニド、ポリICおよびその誘導体、CpGオリゴヌクレオチド、インターロイキン12、インターロイキン18、インターフェロンα、インターフェロンβ、インターフェロンω、インターフェロンγ並びにFlt3リガンドから成る群より選ばれる少なくとも一つである、(7)記載の免疫誘導剤。
(b)(a)のポリペプチドと85%以上の配列同一性を有し、7個以上のアミノ酸から成る、免疫誘導活性を有するポリペプチド
(c)(a)又は(b)のポリペプチドを部分配列として含み、免疫誘導活性を有するポリペプチド。
上記したポリペプチドは、例えば、Fmoc法(フルオレニルメチルオキシカルボニル法)、tBoc法(t-ブチルオキシカルボニル法)等の化学合成法に従って合成することができる。また、各種の市販のペプチド合成機を利用して常法により合成することもできる。また、公知の遺伝子工学的手法を用いて、上記ポリペプチドをコードするポリヌクレオチドを調製し、該ポリヌクレオチドを発現ベクターに組み込んで宿主細胞に導入し、該宿主細胞中でポリペプチドを生産させることにより、目的とするポリペプチドを得ることができる。
(1)cDNAライブラリーの作製
犬の精巣から酸-グアニジウム-フェノール-クロロフォルム法(Acid guanidium-Phenol-Chloroform法)により全RNAを抽出し、Oligotex-dT30 mRNA purification Kit(宝酒造株式会社製)を用いてキット添付のプロトコールに従ってポリA RNAを精製した。
上記作製したcDNAファージライブラリーを用いて、イムノスクリーニングを行った。具体的にはΦ90×15mmのNZYアガロースプレートに2340クローンとなるように宿主大腸菌(XL1-Blue MRF')に感染させ、42℃、3~4時間培養し、溶菌斑(プラーク)を作らせ、IPTG(イソプロピル-β-D-チオガラクトシド)を浸透させたニトロセルロースメンブレン(Hybond C Extra:GE Healthecare Bio-Sciece社製)でプレートを37℃で4時間覆うことによりタンパク質を誘導・発現させ、メンブレンにタンパク質を転写した。その後メンブレンを回収し0.5%脱脂粉乳を含むTBS(10mM Tris-HCl、150mM NaCl pH7.5)に浸し4℃で一晩振盪することによって非特異反応を抑制した。このフィルターを500倍希釈した患犬血清と室温で2~3時間反応させた。
上記方法により単離した1個の陽性クローンを塩基配列解析に供するため、ファージベクターからプラスミドベクターに転換する操作を行った。具体的には宿主大腸菌(XL1-Blue MRF')を吸光度OD600が1.0となるよう調製した溶液200μlと、精製したファージ溶液100μlさらにExAssist helper phage(STRATAGENE社製)1μlを混合した後37℃で15分間反応後、LB培地を3ml添加し37℃で2.5~3時間培養を行い、直ちに70℃の水浴にて20分間保温した後、4℃、1000×g、15分間遠心を行い、上清をファージミド溶液として回収した。次いでファージミド宿主大腸菌(SOLR)を吸光度OD600が1.0となるよう調製した溶液200μlと、精製したファージ溶液10μlを混合した後37℃で15分間反応させ、50μlをアンピシリン(終濃度50μg/ml)含有LB寒天培地に播き37℃一晩培養した。トランスフォームしたSOLRのシングルコロニーを採取し、アンピシリン(終濃度50μg/ml)含有LB培地37℃にて培養後、QIAGEN plasmid Miniprep Kit(キアゲン社製)を使って目的のインサートを持つプラスミドDNAを精製した。
上記方法により得られた遺伝子に対しイヌ、ヒト及びマウスの正常組織及び各種細胞株における発現をRT-PCR(Reverse Transcription-PCR)法により調べた。逆転写反応は以下の通り行なった。すなわち、各組織50~100mg及び各細胞株5~10×106個の細胞からTRIZOL試薬(invitrogen社製)を用いて添付のプロトコールに従い全RNAを抽出した。この全RNAを用いてSuperscript First-Strand Synthesis System for RT-PCR(invitrogen社製)により添付のプロトコールに従いcDNAを合成した。ヒト正常組織(脳、海馬、精巣、結腸、胎盤)のcDNAは、ジーンプールcDNA(invitrogen社製)、QUICK-Clone cDNA(クロンテック社製)及びLarge-Insert cDNA Library(クロンテク社製)を用いた。PCR反応は、取得した遺伝子特異的なプライマー(イヌプライマーは配列番号15及び16、ヒトプライマーは配列番号17及び18、マウスプライマーは配列番号19及び20に記載)を用いて以下の通り行った。すなわち、逆転写反応により調製したサンプル0.25μl、上記プライマーを各2μM、0.2mMの各dNTP、0.65UのExTaqポリメラーゼ(宝酒造株式会社製)となるように各試薬と添付バッファーを加え全量を25μlとし、Thermal Cycler(BIO RAD社製)を用いて、94℃-30秒、55℃-30秒、72℃-1分のサイクルを30回繰り返して行った。比較対照のため、GAPDH特異的なプライマー(イヌ及びヒトGAPDHプライマーは配列番号21及び22、マウスGAPDHプライマーは配列番号23及び24に記載)も同時に用いた。その結果、図1に示すように、イヌKATNAL1遺伝子は、健常なイヌ組織ではほとんどの組織で発現が見られず、一方イヌ腫瘍組織では強い発現が見られた。ヒト及びマウスKATNAL1遺伝子の発現も、イヌKATNAL1遺伝子と同様、ヒト及びマウス正常組織ではほとんど発現が確認できず、癌細胞では大部分の細胞株で発現が検出された(図2、3)。
上記方法により得られた遺伝子に対しヒトの正常組織における発現を定量的RT-PCR(Reverse Transcription-PCR)法により調べた。ヒト正常組織及び癌組織のcDNAは、Tissue scan Real Time cancer survey Panel I(ORIGENE社製)を用いた。また定量RT―PCRはBIO RAD社製社製のCFX96Real Time Cystem―C1000Thermal Cyclerを用いて実施した。PCR反応は、取得した遺伝子特異的なプライマー(配列番号17及び18に記載)を用いて以下の通り行った。すなわち、cDNAサンプル5μl、上記プライマーを各2μM、2XSYBR Premix Ex TaqIIポリメラーゼ(宝酒造株式会社製)の各試薬と添付バッファーを加え全量を20μlとし、94℃-30秒、55℃-30秒、72℃-1分のサイクルを30回繰り返して行った。その結果、KATNAL1遺伝子は、乳癌、大腸癌、甲状腺癌、肝臓癌、前立腺癌、肺癌においてそれぞれの正常組織と比較してすべて5倍以上発現が高かった。本結果は、ヒトKATNAL1を標的とした抗腫瘍剤が、正常組織での副作用を全く心配すること無く、薬剤の薬効と副作用の大きな乖離を期待させる。
(1)生体内でKATNAL1を発現する組換えベクターの作製
配列番号5の塩基配列を基に、以下の方法にて、生体内でKATNAL1を発現する組換えベクターを作製した。PCRは、実施例1において発現の見られたマウス癌細胞株N2a(ATCCより購入)より調製した。cDNAを1μl、HindIII及びXbaI制限酵素切断配列を含む2種類のプライマー(配列番号25及び26に記載)を各0.4μM、0.2mM dNTP、1.25UのPrimeSTAR HSポリメラーゼ(宝酒造株式会社製)となるように各試薬と添付バッファーを加え全量を50μlとし、Thermal Cycler(BIO RAD社製)を用いて、98℃-10秒、55℃-15秒、72℃-4分のサイクルを30回繰り返すことにより行った。なお、上記2種類のプライマーは、配列番号5のアミノ酸配列全長をコードする領域を増幅するものであった。PCR後、増幅されたDNAを1% アガロースゲルにて電気泳動し、QIAquick Gel Extraction Kit(QIAGEN社製)を用いて約1500bpのDNA断片を精製した。
10匹のA/Jマウス(7週齢、雄、日本SLC社から購入)及びBalb/cマウス(7週齢、雄、日本SLC社から購入)に対して、上記で作製したチューブを遺伝子銃に固定し、純ヘリウムガスを用いて400psiの圧力で、剃毛したマウスの腹腔にDNAワクチンを7日ごとに計3回、経皮投与した後に(プラスミドDNA接種量は2μg/匹になる)マウス神経芽腫細胞株N2a細胞、大腸癌細胞株CT26を各々1×106個を移植して抗腫瘍効果を評価した(予防モデル)。なお、コントロールとして、KATNAL1遺伝子が挿入されていないプラスミドDNAを各モデルで10匹ずつ投与した。
(1)ヒト組換えKATNAL1タンパク質の作製
配列番号3の遺伝子を基に、以下の方法にてヒトKATNAL1の組換えタンパク質を作製した。PCRは、実施例1で作製した各種組織・細胞cDNAよりRT-PCR法による発現が確認できたcDNAを1μl、EcoRI及びXhoI制限酵素切断配列を含む2種類のプライマー(配列番号27及び28に記載)を各0.4μM,0.2mM dNTP,1.25UのPrimeSTAR HSポリメラーゼ(宝酒造社製)となるように各試薬と添付バッファーを加え全量を50μlとし、Thermal Cycler(BIO RAD社製)を用いて、98℃-10秒、55℃-15秒、72℃-4分のサイクルを30回繰り返すことにより行った。なお、上記2種類のプライマーは、配列番号4のアミノ酸配列全長をコードする領域を増幅するものであった。PCR後、増幅されたDNAを1%アガロースゲルにて電気泳動し、QIAquick Gel Extraction Kit(QIAGEN社製)を用いて約1500bpのDNA断片を精製した。
上記で得られた、配列番号4を発現する組換え大腸菌を100μg/ml アンピシリン含有LB培地にて600nmでの吸光度が0.7付近になるまで37℃で培養後、イソプロピル-β-D-1-チオガラクトピラノシド終濃度が1mMとなるよう添加し、さらに37℃で4時間培養した。その後4800rpmで10分間遠心し集菌した。この菌体ペレットをリン酸緩衝化生理食塩水に懸濁し、さらに4800rpmで10分間遠心し菌体の洗浄を行った。
健常人から末梢血を分離し、Lymphocyte separation medium(OrganonpTeknika,Durham,NC)に重層して1,500rpmで室温で20分間遠心分離した。末梢血単核球(PBMC)を含有する画分を回収し、冷リン酸塩緩衝液中で3回(又はそれ以上)洗浄し、PBMCを得た。得られたPBMCをAIM-V培地(Life Technololgies,Inc.,米国ニューヨーク州Grand Island)20mlに懸濁し、培養フラスコ(Falcon)中に37℃、5%CO2の条件下で2時間付着させた。非付着細胞はT細胞調製に用い、付着細胞は樹状細胞を調製するために用いた。
Claims (8)
- 以下の(a)ないし(c)のいずれかのポリペプチド類から選択され、かつ免疫誘導活性を有する少なくとも1つのポリペプチド、又は該ポリペプチドをコードするポリヌクレオチドを含み、生体内で該ポリペプチドを発現可能な組換えベクター、を有効成分として含有する免疫誘導剤。
(a)配列表の配列番号4、2、8、10、12に記載のアミノ酸配列中の連続する7個以上のアミノ酸から成るポリペプチド。
(b)前記(a)のポリペプチドと85%以上の配列同一性を有し、かつ7個以上のアミノ酸から成るポリペプチド。
(c)前記(a)又は(b)のポリペプチドを部分配列として含むポリペプチド。 - 前記免疫誘導活性を有するポリペプチドが配列表の配列番号4、2、8、10、12記載のアミノ酸配列を有するポリペプチドである、請求項1に記載の免疫誘導剤。
- 抗原提示細胞の処理剤である、請求項1又は2に記載の免疫誘導剤。
- 癌の治療及び/又は予防剤である、請求項1又は2に記載の免疫誘導剤。
- 前記癌がKATNAL1を発現する癌である、請求項4に記載の免疫誘導剤。
- 前記癌が乳癌、脳腫瘍、肛門周囲腺癌、神経芽腫、肥満細胞腫、肝臓癌、前立腺癌、肺癌、甲状腺癌又は白血病である、請求項4又は5に記載の免疫誘導剤。
- 免疫増強剤をさらに含む、請求項1~6のいずれかに記載の免疫誘導剤。
- 前記免疫増強剤が、フロイントの不完全アジュバント、モンタニド、ポリICおよびその誘導体、CpGオリゴヌクレオチド、インターロイキン12、インターロイキン18、インターフェロンα、インターフェロンβ、インターフェロンω、インターフェロンγ並びにFlt3リガンドから成る群より選ばれる少なくとも一つである、請求項7記載の免疫誘導剤。
Priority Applications (12)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| CN201280024226.4A CN103547284B (zh) | 2011-05-19 | 2012-05-18 | 免疫诱导剂 |
| JP2012525552A JP5954174B2 (ja) | 2011-05-19 | 2012-05-18 | 免疫誘導剤 |
| AU2012256757A AU2012256757B2 (en) | 2011-05-19 | 2012-05-18 | Immunity inducing agent |
| ES12784927.1T ES2595161T3 (es) | 2011-05-19 | 2012-05-18 | Agente inmunoinductor |
| CA2836568A CA2836568C (en) | 2011-05-19 | 2012-05-18 | Immunity induction agent |
| RU2013156368A RU2614386C2 (ru) | 2011-05-19 | 2012-05-18 | Средство, индуцирующее иммунитет |
| EP12784927.1A EP2711017B1 (en) | 2011-05-19 | 2012-05-18 | Immunity inducing agent |
| BR112013029704A BR112013029704A2 (pt) | 2011-05-19 | 2012-05-18 | Agente de indutor de imunidade e uso de um agente indutor de imunidade |
| DK12784927.1T DK2711017T3 (en) | 2011-05-19 | 2012-05-18 | Immunity-inducing agent |
| US14/118,271 US20140120059A1 (en) | 2011-05-19 | 2012-05-18 | Immunity inducing agent |
| KR1020137033212A KR101949186B1 (ko) | 2011-05-19 | 2012-05-18 | 면역 유도제 |
| US14/882,307 US10463725B2 (en) | 2011-05-19 | 2015-10-13 | Immunity inducing agent |
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| JP2011-112181 | 2011-05-19 | ||
| JP2011112181 | 2011-05-19 |
Related Child Applications (2)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| US14/118,271 A-371-Of-International US20140120059A1 (en) | 2011-05-19 | 2012-05-18 | Immunity inducing agent |
| US14/882,307 Division US10463725B2 (en) | 2011-05-19 | 2015-10-13 | Immunity inducing agent |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| WO2012157737A1 true WO2012157737A1 (ja) | 2012-11-22 |
Family
ID=47177051
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/JP2012/062750 WO2012157737A1 (ja) | 2011-05-19 | 2012-05-18 | 免疫誘導剤 |
Country Status (13)
| Country | Link |
|---|---|
| US (2) | US20140120059A1 (ja) |
| EP (1) | EP2711017B1 (ja) |
| JP (1) | JP5954174B2 (ja) |
| KR (1) | KR101949186B1 (ja) |
| CN (1) | CN103547284B (ja) |
| AU (1) | AU2012256757B2 (ja) |
| BR (1) | BR112013029704A2 (ja) |
| CA (1) | CA2836568C (ja) |
| DK (1) | DK2711017T3 (ja) |
| ES (1) | ES2595161T3 (ja) |
| PL (1) | PL2711017T3 (ja) |
| RU (1) | RU2614386C2 (ja) |
| WO (1) | WO2012157737A1 (ja) |
Cited By (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2016175309A1 (ja) * | 2015-04-30 | 2016-11-03 | 東レ株式会社 | 免疫誘導剤 |
Families Citing this family (2)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN105296603A (zh) * | 2014-07-28 | 2016-02-03 | 广州君赫生物科技有限公司 | 一种检测男性不育症Katnal1基因突变位点的试剂盒及其PCR扩增方法 |
| MX2018001298A (es) * | 2015-08-10 | 2018-05-17 | Toray Industries | Agente inductor de inmunidad. |
Citations (5)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO1996033739A1 (en) | 1995-04-25 | 1996-10-31 | Smithkline Beecham Biologicals S.A. | Vaccines containing a saponin and a sterol |
| US5698396A (en) | 1995-06-07 | 1997-12-16 | Ludwig Institute For Cancer Research | Method for identifying auto-immunoreactive substances from a subject |
| US6429304B1 (en) * | 1998-04-14 | 2002-08-06 | The Regents Of The University Of California | Nucleic acids encoding a katanin p60 subunit |
| JP2003532419A (ja) * | 2000-05-05 | 2003-11-05 | インサイト・ゲノミックス・インコーポレイテッド | 細胞骨格結合タンパク質 |
| JP2004008216A (ja) | 2002-03-22 | 2004-01-15 | Research Association For Biotechnology | 新規な全長cDNA |
Family Cites Families (10)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US153312A (en) * | 1874-07-21 | Improvement in handcuffs | ||
| US5595756A (en) * | 1993-12-22 | 1997-01-21 | Inex Pharmaceuticals Corporation | Liposomal compositions for enhanced retention of bioactive agents |
| CA2402563A1 (en) * | 1999-12-23 | 2001-07-26 | Hyseq, Inc. | Novel nucleic acids and polypeptides |
| US7108972B2 (en) * | 2001-02-21 | 2006-09-19 | Curagen Corporation | Proteins, polynucleotides encoding them and methods of using the same |
| US20040078804A1 (en) | 2001-05-03 | 2004-04-22 | Henry Yue | Cytoskeleton-associated proteins |
| US7193069B2 (en) | 2002-03-22 | 2007-03-20 | Research Association For Biotechnology | Full-length cDNA |
| CN1957092B (zh) * | 2004-02-23 | 2013-04-03 | 诺瓦提斯公司 | P53野生型作为使用与细胞毒剂组合的mTOR抑制剂治疗的生物标志 |
| BRPI0912462B1 (pt) * | 2008-08-05 | 2022-03-22 | Toray Industries, Inc | Uso de um agente indutor de imunidade, uso de um polipeptídeo e uso de um vetor recombinante |
| CN107261111B (zh) * | 2009-09-03 | 2022-02-01 | 东丽株式会社 | 免疫诱导剂 |
| JP2011092100A (ja) * | 2009-10-29 | 2011-05-12 | Dna Chip Research Inc | 生理的状態変化と生理的状態に変化を与える要因の効果を評価する遺伝子マーカー、評価方法、評価システム、及びコンピュータプログラム |
-
2012
- 2012-05-18 WO PCT/JP2012/062750 patent/WO2012157737A1/ja active Application Filing
- 2012-05-18 CN CN201280024226.4A patent/CN103547284B/zh not_active Expired - Fee Related
- 2012-05-18 JP JP2012525552A patent/JP5954174B2/ja not_active Expired - Fee Related
- 2012-05-18 EP EP12784927.1A patent/EP2711017B1/en not_active Not-in-force
- 2012-05-18 ES ES12784927.1T patent/ES2595161T3/es active Active
- 2012-05-18 BR BR112013029704A patent/BR112013029704A2/pt active Search and Examination
- 2012-05-18 CA CA2836568A patent/CA2836568C/en not_active Expired - Fee Related
- 2012-05-18 KR KR1020137033212A patent/KR101949186B1/ko active IP Right Grant
- 2012-05-18 PL PL12784927T patent/PL2711017T3/pl unknown
- 2012-05-18 RU RU2013156368A patent/RU2614386C2/ru active
- 2012-05-18 US US14/118,271 patent/US20140120059A1/en not_active Abandoned
- 2012-05-18 DK DK12784927.1T patent/DK2711017T3/en active
- 2012-05-18 AU AU2012256757A patent/AU2012256757B2/en not_active Ceased
-
2015
- 2015-10-13 US US14/882,307 patent/US10463725B2/en not_active Expired - Fee Related
Patent Citations (5)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO1996033739A1 (en) | 1995-04-25 | 1996-10-31 | Smithkline Beecham Biologicals S.A. | Vaccines containing a saponin and a sterol |
| US5698396A (en) | 1995-06-07 | 1997-12-16 | Ludwig Institute For Cancer Research | Method for identifying auto-immunoreactive substances from a subject |
| US6429304B1 (en) * | 1998-04-14 | 2002-08-06 | The Regents Of The University Of California | Nucleic acids encoding a katanin p60 subunit |
| JP2003532419A (ja) * | 2000-05-05 | 2003-11-05 | インサイト・ゲノミックス・インコーポレイテッド | 細胞骨格結合タンパク質 |
| JP2004008216A (ja) | 2002-03-22 | 2004-01-15 | Research Association For Biotechnology | 新規な全長cDNA |
Non-Patent Citations (14)
| Title |
|---|
| "Experimental Medicine", vol. 12, 1994 |
| "Molecular Cloning" |
| ERICKSON, H.S. ET AL.: "Searching for 13q key players in esophageal squamous-cell carcinogenesis by qRT-PCR of microdissected tissues", CANCER BIOMARKERS 1574-0153, vol. 4, no. 3, 2008, pages 139 - 140, XP008171487 * |
| GODING: "Monoclonal Antibodies: Principles and Practice", 1986 |
| INT. J. CANCER, vol. 58, 1994, pages 317 |
| KREIG, NATURE, vol. 374, pages 546 - 549 |
| NIKKEI, SCIENCE, April 1994 (1994-04-01), pages 20 - 45 |
| PROC. NATL. ACAD. SCI. USA, vol. 92, 1995, pages 11810 - 11813 |
| RIGDEN DJ. ET AL., FEBS LETT., vol. 583, no. 5, 2009, pages 872 - 8 |
| SAHIN, U. ET AL.: "Human neoplasms elicit multiple specific immune responses in the autologous host", PROCEEDINGS OF THE NATIONAL ACADEMY OF SCIENCES OF THE UNITED STATES OF AMERICA, vol. 92, no. 25, 1995, pages 11810 - 3, XP055135654 * |
| See also references of EP2711017A4 |
| SO, MOLECULES AND CELLS, vol. 7, 1997, pages 178 - 186 |
| STEIN GH ET AL., J. CELL PHYSIOL., vol. 99, 1979, pages 43 - 54 |
| THE PHARMACEUTICAL MONTHLY, vol. 36, no. 1, 1994, pages 23 - 48 |
Cited By (4)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2016175309A1 (ja) * | 2015-04-30 | 2016-11-03 | 東レ株式会社 | 免疫誘導剤 |
| JPWO2016175309A1 (ja) * | 2015-04-30 | 2018-02-22 | 東レ株式会社 | 免疫誘導剤 |
| JP7160462B2 (ja) | 2015-04-30 | 2022-10-25 | 東レ株式会社 | 免疫誘導剤 |
| US11478539B2 (en) | 2015-04-30 | 2022-10-25 | Toray Industries, Inc. | Immunity-inducing agent |
Also Published As
| Publication number | Publication date |
|---|---|
| US20140120059A1 (en) | 2014-05-01 |
| CN103547284A (zh) | 2014-01-29 |
| AU2012256757A1 (en) | 2014-01-09 |
| KR101949186B1 (ko) | 2019-02-18 |
| JP5954174B2 (ja) | 2016-07-20 |
| BR112013029704A2 (pt) | 2017-08-29 |
| AU2012256757B2 (en) | 2017-06-15 |
| RU2614386C2 (ru) | 2017-03-27 |
| JPWO2012157737A1 (ja) | 2014-07-31 |
| EP2711017A4 (en) | 2014-11-05 |
| CA2836568A1 (en) | 2012-11-22 |
| RU2013156368A (ru) | 2015-06-27 |
| EP2711017B1 (en) | 2016-08-03 |
| PL2711017T3 (pl) | 2017-01-31 |
| KR20140034224A (ko) | 2014-03-19 |
| ES2595161T3 (es) | 2016-12-28 |
| US10463725B2 (en) | 2019-11-05 |
| EP2711017A1 (en) | 2014-03-26 |
| CN103547284B (zh) | 2015-11-25 |
| DK2711017T3 (en) | 2016-11-07 |
| CA2836568C (en) | 2020-07-21 |
| US20160030540A1 (en) | 2016-02-04 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| JP5962739B2 (ja) | 免疫誘導剤 | |
| JP5874163B2 (ja) | 免疫誘導剤 | |
| JP5954174B2 (ja) | 免疫誘導剤 | |
| US10537623B2 (en) | Immunity induction agent | |
| US11478539B2 (en) | Immunity-inducing agent | |
| JP7087387B2 (ja) | 免疫誘導剤 | |
| JP7035534B2 (ja) | 免疫誘導剤 | |
| JP5504607B2 (ja) | 免疫誘導剤 | |
| JP5572938B2 (ja) | 免疫誘導剤 | |
| JP5589274B2 (ja) | 免疫誘導剤 | |
| JP2008247892A (ja) | 癌の治療及び/又は予防剤 |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| ENP | Entry into the national phase |
Ref document number: 2012525552 Country of ref document: JP Kind code of ref document: A |
|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application |
Ref document number: 12784927 Country of ref document: EP Kind code of ref document: A1 |
|
| ENP | Entry into the national phase |
Ref document number: 2836568 Country of ref document: CA |
|
| NENP | Non-entry into the national phase |
Ref country code: DE |
|
| REEP | Request for entry into the european phase |
Ref document number: 2012784927 Country of ref document: EP |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 2012784927 Country of ref document: EP |
|
| ENP | Entry into the national phase |
Ref document number: 20137033212 Country of ref document: KR Kind code of ref document: A |
|
| ENP | Entry into the national phase |
Ref document number: 2013156368 Country of ref document: RU Kind code of ref document: A |
|
| ENP | Entry into the national phase |
Ref document number: 2012256757 Country of ref document: AU Date of ref document: 20120518 Kind code of ref document: A |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 14118271 Country of ref document: US |
|
| REG | Reference to national code |
Ref country code: BR Ref legal event code: B01A Ref document number: 112013029704 Country of ref document: BR |
|
| ENP | Entry into the national phase |
Ref document number: 112013029704 Country of ref document: BR Kind code of ref document: A2 Effective date: 20131119 |