WO2010004231A2 - Nouveaux composes, leur utilisation comme medicaments, et leur procede de preparation - Google Patents
Nouveaux composes, leur utilisation comme medicaments, et leur procede de preparation Download PDFInfo
- Publication number
- WO2010004231A2 WO2010004231A2 PCT/FR2009/051388 FR2009051388W WO2010004231A2 WO 2010004231 A2 WO2010004231 A2 WO 2010004231A2 FR 2009051388 W FR2009051388 W FR 2009051388W WO 2010004231 A2 WO2010004231 A2 WO 2010004231A2
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- independently
- formula
- compound
- group
- another
- Prior art date
Links
- 0 C*(C*(C)Oc1ccc(CCNC(*)=*)cc1)Oc1ccc(CCNC(*)=*)cc1 Chemical compound C*(C*(C)Oc1ccc(CCNC(*)=*)cc1)Oc1ccc(CCNC(*)=*)cc1 0.000 description 8
- UQFQONCQIQEYPJ-UHFFFAOYSA-N C[n]1nccc1 Chemical compound C[n]1nccc1 UQFQONCQIQEYPJ-UHFFFAOYSA-N 0.000 description 1
Classifications
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07C—ACYCLIC OR CARBOCYCLIC COMPOUNDS
- C07C271/00—Derivatives of carbamic acids, i.e. compounds containing any of the groups, the nitrogen atom not being part of nitro or nitroso groups
- C07C271/06—Esters of carbamic acids
- C07C271/08—Esters of carbamic acids having oxygen atoms of carbamate groups bound to acyclic carbon atoms
- C07C271/10—Esters of carbamic acids having oxygen atoms of carbamate groups bound to acyclic carbon atoms with the nitrogen atoms of the carbamate groups bound to hydrogen atoms or to acyclic carbon atoms
- C07C271/16—Esters of carbamic acids having oxygen atoms of carbamate groups bound to acyclic carbon atoms with the nitrogen atoms of the carbamate groups bound to hydrogen atoms or to acyclic carbon atoms to carbon atoms of hydrocarbon radicals substituted by singly-bound oxygen atoms
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P31/00—Antiinfectives, i.e. antibiotics, antiseptics, chemotherapeutics
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P31/00—Antiinfectives, i.e. antibiotics, antiseptics, chemotherapeutics
- A61P31/04—Antibacterial agents
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P31/00—Antiinfectives, i.e. antibiotics, antiseptics, chemotherapeutics
- A61P31/10—Antimycotics
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P31/00—Antiinfectives, i.e. antibiotics, antiseptics, chemotherapeutics
- A61P31/12—Antivirals
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P33/00—Antiparasitic agents
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07C—ACYCLIC OR CARBOCYCLIC COMPOUNDS
- C07C257/00—Compounds containing carboxyl groups, the doubly-bound oxygen atom of a carboxyl group being replaced by a doubly-bound nitrogen atom, this nitrogen atom not being further bound to an oxygen atom, e.g. imino-ethers, amidines
- C07C257/10—Compounds containing carboxyl groups, the doubly-bound oxygen atom of a carboxyl group being replaced by a doubly-bound nitrogen atom, this nitrogen atom not being further bound to an oxygen atom, e.g. imino-ethers, amidines with replacement of the other oxygen atom of the carboxyl group by nitrogen atoms, e.g. amidines
- C07C257/14—Compounds containing carboxyl groups, the doubly-bound oxygen atom of a carboxyl group being replaced by a doubly-bound nitrogen atom, this nitrogen atom not being further bound to an oxygen atom, e.g. imino-ethers, amidines with replacement of the other oxygen atom of the carboxyl group by nitrogen atoms, e.g. amidines having carbon atoms of amidino groups bound to acyclic carbon atoms
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07C—ACYCLIC OR CARBOCYCLIC COMPOUNDS
- C07C279/00—Derivatives of guanidine, i.e. compounds containing the group, the singly-bound nitrogen atoms not being part of nitro or nitroso groups
- C07C279/04—Derivatives of guanidine, i.e. compounds containing the group, the singly-bound nitrogen atoms not being part of nitro or nitroso groups having nitrogen atoms of guanidine groups bound to acyclic carbon atoms of a carbon skeleton
- C07C279/08—Derivatives of guanidine, i.e. compounds containing the group, the singly-bound nitrogen atoms not being part of nitro or nitroso groups having nitrogen atoms of guanidine groups bound to acyclic carbon atoms of a carbon skeleton being further substituted by singly-bound oxygen atoms
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07C—ACYCLIC OR CARBOCYCLIC COMPOUNDS
- C07C279/00—Derivatives of guanidine, i.e. compounds containing the group, the singly-bound nitrogen atoms not being part of nitro or nitroso groups
- C07C279/04—Derivatives of guanidine, i.e. compounds containing the group, the singly-bound nitrogen atoms not being part of nitro or nitroso groups having nitrogen atoms of guanidine groups bound to acyclic carbon atoms of a carbon skeleton
- C07C279/12—Derivatives of guanidine, i.e. compounds containing the group, the singly-bound nitrogen atoms not being part of nitro or nitroso groups having nitrogen atoms of guanidine groups bound to acyclic carbon atoms of a carbon skeleton being further substituted by nitrogen atoms not being part of nitro or nitroso groups
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07C—ACYCLIC OR CARBOCYCLIC COMPOUNDS
- C07C279/00—Derivatives of guanidine, i.e. compounds containing the group, the singly-bound nitrogen atoms not being part of nitro or nitroso groups
- C07C279/20—Derivatives of guanidine, i.e. compounds containing the group, the singly-bound nitrogen atoms not being part of nitro or nitroso groups containing any of the groups, X being a hetero atom, Y being any atom, e.g. acylguanidines
- C07C279/24—Y being a hetero atom
Definitions
- Novel compounds their use as medicaments, and process for their preparation
- the invention relates to novel compounds, their use as medicaments and their method of preparation.
- antiseptics and / or antibiotics are known and marketed, for example hexamidine and chlorhexidine.
- NSS nosocomial infections
- the agents responsible for IN can be bacteria, viruses or fungi.
- IN means any infection occurring within at least 48 hours (for bacterial infections) or within a period corresponding to the incubation period of the germ (for viral and fungal infections) after admission or at the following a hospitalization, and if she was absent on admission to the hospital.
- the nosocomial character of an infection of the surgical site is asserted if it occurs within 30 days after the intervention, or in the year if it involves the implantation of foreign material (prosthesis, implant), and this although the patient is no longer hospitalized.
- NNs are worrying because of their significant morbidity, associated mortality, significant additional hospital costs and the emergence of bacteria resistant to several families of antibiotics (BMR: Multi-Resistant Bacteria).
- NAs urinary tract infections (30%), respiratory infections (15%), surgical site infections (14%), and skin and soft tissue infections (10%).
- bacteria appear on the front line. The most commonly involved are Escherichia coli (25%) (intestinal commensal flora bacterium, responsible for urinary tract infections), Staphylococcus aureus (19%) (commensal cutaneous and nasal flora bacterium, responsible for skin infections and surgical site), Pseudomonas aeruginosa (10%) (opportunistic bacterium, responsible for severe pneumonias and skin infections), Enterococcus spp (6%) (intestinal commensal flora bacterium mainly responsible for urinary tract infections).
- MRSA Meticillin-Resistant Staphylococcus aureus
- glycopeptide-resistant enterococci (10%) (Vancomycin-resistant enterococci: vancomycin is a member of the glycopeptide family with teicoplanin), (iii) Pseudomonas aeruginosa resistant to cephalosporins. 3rd generation
- One aspect of the invention is to provide novel compounds for combating bacterial, viral, fungal and parasitic infections.
- One aspect of the invention relates to the use of compounds for the preparation of a medicament for the treatment of bacterial, viral, fungal and parasitic infections.
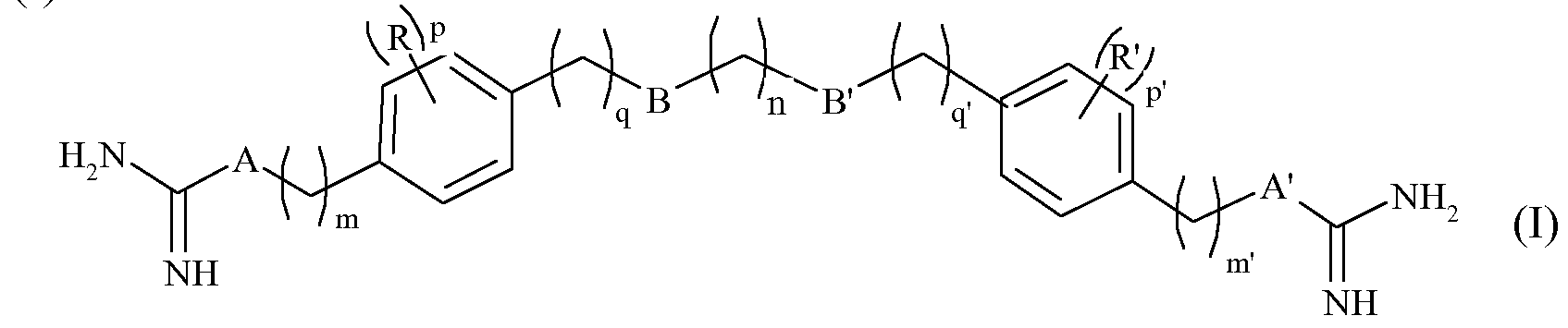
- the invention relates to the use of at least one compound of formula
- n represents an integer from 1 to 12, in particular from 1 to 8, m and m 'independently of one another represent integers from 0 to 8, especially 1 to 8, - q and q 'represent, independently of each other, integers from 0 to 2, p and p' independently of one another represent integers from 0 to 4, - A and A 'independently represent one of the other a CH 2 group, an NH group or a NR "group, in which R" is an alkyl group of 1 to 3 carbon atoms, linear or branched, - B and B 'represent independently one of the another an oxygen atom or a CH 2 group,
- R and R ' are, independently of one another, a halogen, such as chlorine, bromine, iodine or fluorine atoms, or an alkyl group of 1 to 3 carbon atoms, linear or branched, or a physiologically acceptable acid salt derived from a compound of formula (I) such as a hydrochloride, a formate, a trifluoroacetate or an oxalate (HOOCCOOH),
- linear or branched alkyl group of 1 to 3 carbon atoms denotes methyl, ethyl, n-propyl and isopropyl groups.
- physiologically acceptable acid salt means that the compounds of the formula (I), defined above, may exist in the form of amidinium (when A and / or A 'are -CH 2 -) or guanidinium (when A and / or A 'represents (nt) NH or NR ”) by reaction of an inorganic acid or an organic acid with a compound of formula (I).
- inorganic acids for obtaining physiologically acceptable salts include, but are not limited to, hydrochloric acid, hydrobromic acid, nitric acid, carbonic acid, formic acid monohydrogenocarbonic acid, phosphoric acid, monohydrogenphosphoric acid, dihydrogenphosphoric acid, perchloric acid, sulfuric acid, monohydrogenosulfuric acid, hydroiodic acid.
- organic acids for obtaining physiologically acceptable salts include, but are not limited to, acetic acid, lactic acid, propionic acid, butyric acid, isobutyric acid, lactic acid, palmic acid, maleic acid, glutamic acid, hydroxymaleic acid, malonic acid, benzoic acid, succinic acid, glycolic acid, suberic acid, fumaric acid, mandelic acid, phthalic acid, salicylic acid, benzenesulfonic acid, toluenesulfonic acid, citric acid, tartaric acid, methanesulfonic acid, hydroxynaphthoic acid.
- Amino acid salts such as arginates and their equivalents are also included as well as salts of organic acids such as glucuronic acid or galacturonic acid and their equivalents (see, for example, Berge et al, " Pharmaceutical Salts, "Journal of Pharmaceutical Science, 1977, 66, 1-19).
- n represents an integer from 1 to 12
- n means all integers 1, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11 and 12.
- m and m independently of one another represent integers from 0 to 8 "denote all integers 0, 1, 2, 3, 4, 5, 6, 7 and 8. This definition is applies to formula (I) but also to the other formulas below.
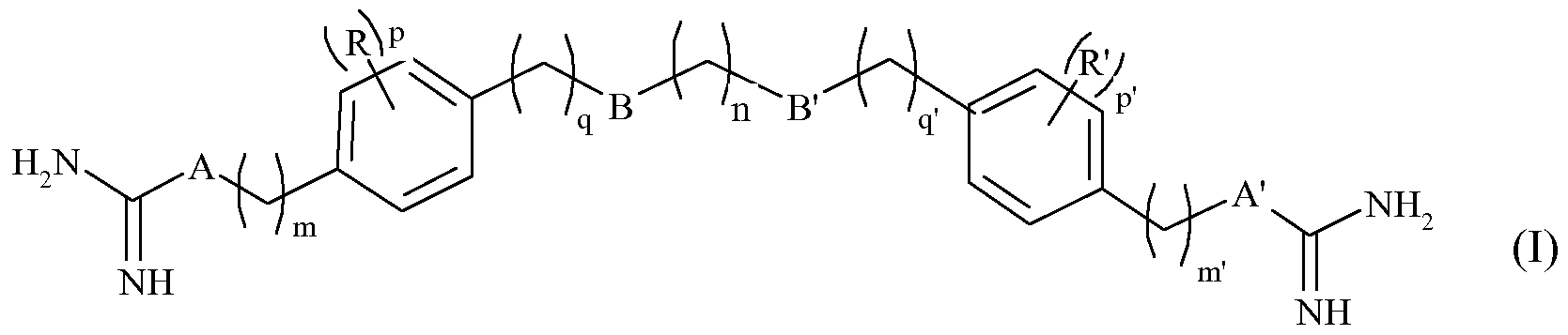
- the invention also relates to the use of at least one compound of formula (I) below:
- n represents an integer from 1 to 12, especially from 1 to 8, m and m 'independently of one another represent integers from 2 to 8, q and q' independently represent one on the other hand integers from 0 to 2, p and p 'independently of one another represent integers from 0 to 4, - A and A' represent, independently of one another, a CH group 2 , an NH group or a NR "group, wherein R" is an alkyl group of 1 to 3 carbon atoms, linear or branched,
- B and B ' represent, independently of one another, an oxygen atom or a CH 2 group; - R and R' are, independently of each other, a halogen, such as chlorine, bromine, iodine or fluorine, or an alkyl group of 1 to 3 carbon atoms, linear or branched, or a physio-logically acceptable acid salt derived from a compound of formula (I) such as a hydrochloride, a formate, a trifluoroacetate or an oxalate (HOOCCOOH), for the preparation of a medicament for the treatment of pathologies associated with bacterial, viral, fungal and parasitic infections.
- a halogen such as chlorine, bromine, iodine or fluorine
- an alkyl group of 1 to 3 carbon atoms linear or branched
- a physio-logically acceptable acid salt derived from a compound of formula (I) such as a hydrochloride, a formate, a trifluoroacetate
- the invention also relates to the use of at least one compound of formula (I) below:
- n represents an integer from 1 to 12, in particular from 1 to 8, m and m 'are equal to 2, q and q' independently of one another represent integers from 0 to 2, p and p 'represent, independently of one another, integers from 0 to 4,
- a and A 'independently of one another are CH 2 , NH or NR ", wherein R" is straight or branched alkyl of 1 to 3 carbon atoms,
- B and B ' represent, independently of one another, an oxygen atom or a CH 2 group
- R and R ' are independently of each other a halogen, such as chlorine, bromine, iodine or fluorine, or an alkyl group of 1 to 3 carbon atoms, linear or branched, or a salt of physio-logically acceptable acid derived from a compound of formula (I) such as a hydrochloride, a formate, a trifluoroacetate or an oxalate (HOOCCOOH),
- the invention also relates to the use of at least one compound of formula (I) below:
- n represents an integer from 1 to 12, in particular from 1 to 8, m and m 'independently of one another represent integers from 0 to 8, especially 1 to 8, q and q'; are equal to 0, p and p 'represent, independently of one another, integers from 0 to 4,
- a and A 'independently of one another are CH 2 , NH or NR ", wherein R" is straight or branched alkyl of 1 to 3 carbon atoms,
- B and B ' represent, independently of one another, an oxygen atom or a CH 2 group
- R and R ' are independently of each other a halogen, such as chlorine, bromine, iodine or fluorine, or an alkyl group of 1 to 3 carbon atoms, linear or branched, or a salt of physio-logically acceptable acid derived from a compound of formula (I) such as a hydrochloride, a formate, a trifluoroacetate or an oxalate (HOOCCOOH),
- the invention also relates to the use of at least one compound of formula (I)
- n represents an integer from 1 to 12, in particular from 1 to 8, m and m 'represent, independently of one another, integers from 0 to 8, especially 1 to 8, q and q 'represent, independently of each other, integers from 0 to 2, p and p' are equal to 0,
- a and A 'independently of one another are CH 2 , NH or NR ", wherein R" is straight or branched alkyl of 1 to 3 carbon atoms,
- B and B ' represent, independently of one another, an oxygen atom or a CH 2 group, or a physiologically acceptable acid salt derived from a compound of formula (I) such as a hydrochloride, a formate, trifluoroacetate or oxalate (HOOCCOOH),
- the invention also relates to the use of at least one compound of formula (I) below:
- n represents an integer from 1 to 12, especially from 1 to 8, m and m 'are equal to 2, - q and q' are equal to 0, p and p 'independently represent one of other integers from 0 to 4,
- a and A 'independently of one another are CH 2 , NH or NR ", wherein R" is straight or branched alkyl of 1 to 3 carbon atoms,
- B and B ' represent, independently of one another, an oxygen atom or a CH 2 group
- R and R ' are independently of each other a halogen, such as chlorine, bromine, iodine or fluorine, or an alkyl group of 1 to 3 carbon atoms, linear or branched, or a salt of physio-logically acceptable acid derived from a compound of formula (I) such as a hydrochloride, a formate, a trifluoroacetate or an oxalate (HOOCCOOH), for the preparation of a medicament for the treatment of pathologies associated with bacterial, viral, fungal and parasitic infections.
- a halogen such as chlorine, bromine, iodine or fluorine
- an alkyl group of 1 to 3 carbon atoms linear or branched
- a salt of physio-logically acceptable acid derived from a compound of formula (I) such as a hydrochloride, a formate, a trifluoroacetate or an oxalate (HOOCCOOH)
- the invention also relates to the use of at least one compound of formula (I) below:
- n represents an integer from 1 to 12, in particular from 1 to 8, m and m 'are equal to 2, q and q' independently of one another represent integers from 0 to 2, p and p 'are equal to 0,
- a and A 'independently of one another are CH 2 , NH or NR ", wherein R" is straight or branched alkyl of 1 to 3 carbon atoms,
- B and B ' represent, independently of one another, an oxygen atom or a CH 2 group, or a physiologically acceptable acid salt derived from a compound of formula (I) such as a hydrochloride, a formate, trifluoroacetate or oxalate (HOOCCOOH),
- the invention also relates to the use of at least one compound of formula (I) below:
- n represents an integer from 1 to 12, in particular from 1 to 8, m and m 'independently of one another represent integers from 0 to 8, especially 1 to 8,
- a and A 'independently of one another are CH 2 , NH or NR ", wherein R" is straight or branched alkyl of 1 to 3 carbon atoms,
- B and B ' represent, independently of one another, an oxygen atom or a CH 2 group, or a physiologically acceptable acid salt derived from a compound of formula (I) such as a hydrochloride, a formate, trifluoroacetate or oxalate (HOOCCOOH),
- the invention also relates to the use of at least one compound of formula (I) below:
- n represents an integer from 1 to 12, in particular from 1 to 8, m and m 'are equal to 2, q and q' are equal to 0, - p and p 'are equal to 0,
- a and A 'independently of one another are CH 2 , NH or NR ", wherein R" is straight or branched alkyl of 1 to 3 carbon atoms,
- B and B ' represent, independently of one another, an oxygen atom or a CH 2 group, or a physiologically acceptable acid salt derived from a compound of formula (I) such as a hydrochloride, a formate, a trifluoroacetate or an oxalate (HOOCCOOH),
- the invention also relates to the use of at least one compound of formula (Ia) below:
- n represents an integer from 1 to 12, in particular from 1 to 8, m and m 'independently of one another represent integers from 0 to 8, especially 1 to 8, q and q'; independently of one another represent integers from 0 to 2, p and p 'independently of one another represent integers from 0 to 4,
- a and A 'independently of one another represent an NH group or a group
- R is an alkyl group of 1 to 3 carbon atoms, linear or branched
- B and B ' represent, independently of one another, an oxygen atom or a CH 2 group
- R and R ' are independently of each other a halogen, such as chlorine, bromine, iodine or fluorine, or an alkyl group of 1 to 3 carbon atoms, linear or branched, or a salt of physio-logically acceptable acid derived from a compound of formula (I) such as a hydrochloride, a formate, a trifluoroacetate or an oxalate (HOOCCOOH), for the preparation of a medicament for the treatment of pathologies associated with bacterial infections, viral, fungal and parasitic.
- a halogen such as chlorine, bromine, iodine or fluorine
- an alkyl group of 1 to 3 carbon atoms linear or branched
- a salt of physio-logically acceptable acid derived from a compound of formula (I) such as a hydrochloride, a formate, a trifluoroacetate or an oxalate (HOOCCOOH)
- the compounds of formula (Ia) correspond to the compounds of formula (I) in which A and A 'represent, independently of one another, an NH group or an NR "group.
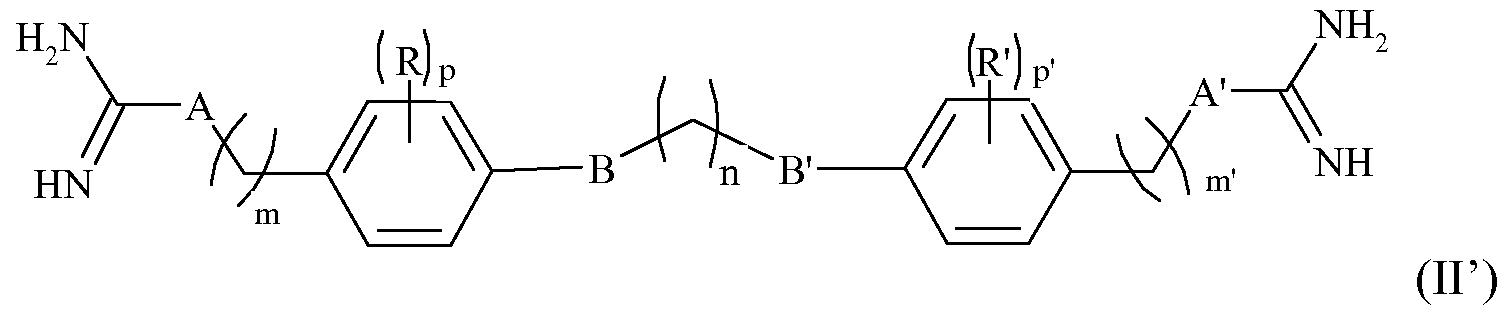
- the formula of the compounds for the use as defined above is the following formula (II):
- B, B ', R, R', m, m ', n, p, p' are as defined above, and, - A and A 'represent, independently of one another, a CH 2 group, an NH group or a NR "group, in which R" is a linear or branched alkyl group of 1 to 3 carbon atoms, or a physiologically acceptable acid salt derived from a compound of formula (II) such as hydrochloride, formate, trifluoroacetate or oxalate (HOOCCOOH).
- a compound of formula (II) such as hydrochloride, formate, trifluoroacetate or oxalate (HOOCCOOH).
- B, B ', R, R', m, m ', n, p, p' are as defined above, and,
- a and A 'independently of one another are NH or NR ", wherein R" is linear or branched alkyl of 1 to 3 carbon atoms, or a physiologically acid salt; acceptable derivative of a compound of formula (IF) such as hydrochloride, formate, trifluoroacetate or oxalate (HOOCCOOH).
- IF acceptable derivative of a compound of formula (IF) such as hydrochloride, formate, trifluoroacetate or oxalate (HOOCCOOH).
- the formula of the compounds of formula (IF) corresponds to formula (Ia), in which q and q 'are equal to 0.
- R represents a CH 2 group, an NH group or a NR "group, in which R" is an alkyl group of 1 to 3 carbon atoms. carbon, linear or branched, or a physio-logically acceptable acid salt derived from a compound of formula (III) such as a hydrochloride, a formate, a trifluoroacetate or an oxalate (HOOCCOOH).
- a compound of formula (III) such as a hydrochloride, a formate, a trifluoroacetate or an oxalate (HOOCCOOH).
- the formula of the compounds of formula (III) corresponds to formula (II), in which: m and m 'on the one hand, and p and p' on the other hand, are equal, and, - A and A 'd on the one hand, and B and B 'on the other, represent the same atom or group.
- the compounds of formula (III) are symmetrical.
- the formula of the compounds for the use as defined above is the following formula (HF):
- B, R, m, n, p are as defined above, and, - A represents an NH group or a NR "group, in which R" is an alkyl group of 1 to 3 carbon atoms, linear or branched or a physiologically acceptable acid salt derived from a compound of formula (III) such as hydrochloride, formate, trifluoroacetate or oxalate (HOOCCOOH).
- a compound of formula (III) such as hydrochloride, formate, trifluoroacetate or oxalate (HOOCCOOH).
- the formula of the compounds of formula (HF) corresponds to the formula (IF), in which: m and m 'on the one hand, and p and p' on the other hand, are equal, and
- a and A 'on the one hand, and B and B' on the other hand, represent the same atom or group.
- the compounds of formula (HF) are symmetrical.
- the formula of the compound for use as defined above is the following formula (IV):
- n 1, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11 or 12, or a physiologically acceptable acid salt derived from a compound of formula (IV)
- the formula of the compounds of formula (IV) corresponds to formula (III), in which A represents an NH group, B an oxygen atom, p is equal to 0 and m is equal to 2.
- the compounds of formula (IV) are symmetrical.
- the formula of the compound for use as defined above is the following formula 6:
- a physiologically acceptable acid salt derived from a compound of formula 6 such as hydrochloride, formate, trifluoroacetate or oxalate (HOOCCOOH).
- the formula of the compounds of formula 6 corresponds to formula (IV), wherein n is 6.
- the compound of formula 6 is symmetrical.
- the formula of the compound for the use as defined above is the formula (II) as defined above in which: m is different from m ', and / or p is different from p 'and / or R is different from R' and / or A is different from A 'and / or
- the compounds of the invention are involved in the treatment of pathologies caused by the bacteria of Table 1.
- Table 1 Bacteria causing pathologies treated with the compounds according to the invention.
- the compounds of the invention are involved in the treatment of pathologies caused by multidrug-resistant bacteria (BMR) responsible for nosocomial and / or community infections.
- BMR multidrug-resistant bacteria
- the compounds of the invention are involved in the treatment of pathologies caused by viruses belonging to different families, such as: viral hepatitis A, B & D, C (families of Picornaviridae, Hepadnavi ⁇ dae, Flaviviridae) ; AIDS, leukemia, cancer ... (Family Retroviridae); smallpox ... (Family Poxviridae); oral and / or genital herpes, chickenpox, shingles, mononucleosis, roselose, lymphoma ... (Family Herpesviridae); SARS, colds ...
- viruses belonging to different families such as: viral hepatitis A, B & D, C (families of Picornaviridae, Hepadnavi ⁇ dae, Flaviviridae) ; AIDS, leukemia, cancer ... (Family Retroviridae); smallpox ... (Family Poxviridae); oral
- the compounds of the invention are involved in the treatment of pathologies caused by fungi, in particular the following fungi: Candida spp., Aspergillus spp., Epidermophyton spp., Trichophyton spp., Microsporum spp. and Pneumocystis jiroveci.
- the compounds of the invention are involved in the treatment of pathologies caused by the parasites of Table 2.
- Table 2 Parasites causing pathologies treated with the compounds according to the invention.
- pathologies caused by parasites may for example be parasitic diseases due to protozoa or worms, scabies, myases and pediculosis.
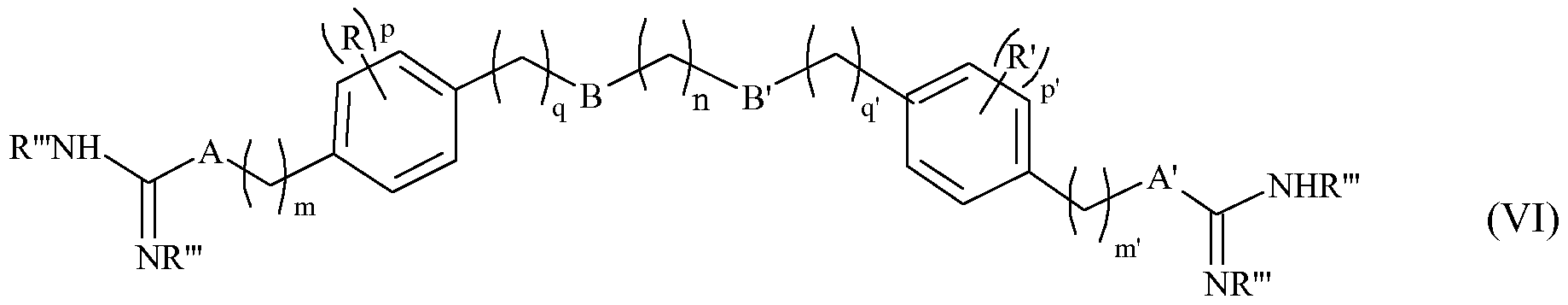
- the invention relates to the compound of formula (VI) below:
- n represents an integer from 1 to 12, in particular from 1 to 8, q and q 'independently of one another represent integers from 0 to 2, p and p' independently of one another represent integers from 0 to 4,
- B and B ' represent, independently of one another, an oxygen atom or a CH 2 group
- R and R ' represent, independently of one another, a halogen, such as chlorine, bromine, iodine or fluorine atoms, or an alkyl group of 1 to 3 carbon atoms, linear or branched,
- R '" represents a hydrogen or a group protecting the amine function, chosen especially from the groups Boc, Fmoc, Bn, Z, if A and A' represent, independently of one another, a CH 2 group, then m and m 'independently of one another represent integers from 1 to 8, if A and A' independently of one another represent an NH group or a group
- a physiologically acceptable acid salt derived from a compound of formula (VI) such as a hydrochloride, a formate, a trifluoroacetate or an oxalate (HOOCCOOH),
- the new compounds defined above can be used according to the present invention.
- the compounds of formula (VI), with the exception of the two compounds above, are new.
- linear or branched alkyl group of 1 to 3 carbon atoms denotes methyl, ethyl, n-propyl and isopropyl groups.
- physiologically acceptable acid salt means that the compounds of the formula (I), defined above, may exist in the form of amidinium (when A and / or A 'are -CH 2 -) or guanidinium (when A and / or A 'represents (nt) NH or NR ”) by reaction of an inorganic acid or an organic acid with a compound of formula (I).
- inorganic acids for obtaining physiologically acceptable salts include, but are not limited to, hydrochloric acid, hydrobromic acid, nitric acid, carbonic acid, formic acid, monohydrogenocarbonic acid, phosphoric acid, monohydrogenphosphoric acid, dihydrogenphosphoric acid, perchloric acid, sulfuric acid, monohydrogenosulfuric acid, hydroiodic acid.
- organic acids for obtaining physiologically acceptable salts include, but are not limited to, acetic acid, lactic acid, propionic acid, butyric acid, isobutyric acid, palmic acid, maleic acid, glutamic acid, hydroxymaleic acid, malonic acid, benzoic acid, succinic acid, glycolic acid, suberic acid, fumaric acid, mandelic acid, phthalic acid, salicylic acid, benzenesulfonic acid, ⁇ -toluenesulfonic acid, citric acid, tartaric acid, methanesulfonic acid, hydroxynaphthoic acid.
- Amino acid salts such as arginates and their equivalents are also included as well as salts of organic acids such as glucuronic acid or galacturonic acid and their equivalents (see, for example, Berge et al, " Pharmaceutical Salts, "Journal of Pharmaceutical Science, 1977, 66, 1-19).
- the invention relates to the compound of formula (VI) defined above, in which: m and m 'independently of one another represent integers from 2 to 8.
- the invention relates to the compound of formula
- the invention relates to the compound of formula (VI) defined above, in which: q and q 'are equal to 0.
- the invention relates to the compound of formula (VI) defined above, in which: p and p 'are equal to 0.
- the invention relates to the compound of formula (VI) defined above, in which: m and m 'are equal to 2, and q and q' are equal to 0 to 2.
- the invention relates to the compound of formula (VI) defined above, in which: m and m 'are equal to 2, and p and p' are equal to 0.
- the invention relates to the compound of formula (VI) defined above, in which: q and q 'are equal to 0, and p and p' are equal to 0.
- the invention relates to the compound of formula (VI) defined above, in which: m and m 'are equal to 2, q and q 'are 0, and p and p' are 0.
- the invention relates to the compound of formula (VI) defined above, in which: n represents an integer from 6 to 12, in particular from 8 to 10.
- the invention relates to the compound of formula (VI) defined above, in which: R, R 'represent, independently of one another, an alkyl group of 1 to 3 carbon atoms, linear or branched.
- the invention relates to the compound of formula (VI) defined above, in which: p and p 'are equal to 0.
- the invention relates to the compound of formula (VI) defined above, in which: n represents an integer ranging from 6 to 12, in particular from 8 to 10, and R, R 'represent independently from each other an alkyl group of 1 to 3 carbon atoms, linear or branched.
- the invention relates to the compound of formula (VI) defined above, in which: n represents an integer from 6 to 12, in particular from 8 to 10, and p and p 'are equal to to 0.
- the invention also relates to the compound of formula (VF) below:
- n represents an integer from 1 to 12, especially from 1 to 8, q and q 'independently of one another represent integers from 0 to 2, p and p' independently represent one of other integers from 0 to 4, B and B 'represent, independently of one another, an oxygen atom or a CH 2 group,
- R and R ' represent, independently of one another, a halogen, such as chlorine, bromine, iodine or fluorine atoms, or an alkyl group of 1 to 3 carbon atoms, linear or branched,
- R '" represents a hydrogen or a group protecting the amine function, chosen in particular from the groups Boc, Fmoc, Bn, Z,
- a and A ' represent, independently of one another, an NH group or a NR "group, in which R" is an alkyl group of 1 to 3 carbon atoms, linear or branched, then m and m' represent independently from one another integers from 2 to 8,
- a physiologically acceptable acid salt derived from a compound of formula (VI) such as a hydrochloride, a formate, a trifluoroacetate or an oxalate (HOOCCOOH),
- the new compounds defined above can be used according to the present invention.
- the formula of the compounds of formula (VF) corresponds to formula (VI) with A and A 'representing, independently of one another, an NH group or an NR "group.
- the invention relates to compounds of formula
- n represents an integer from 1 to 12, especially from 1 to 8, q and q 'independently of one another represent integers from 0 to 2, p and p' independently represent one on the other, whole numbers from 0 to 4,
- B and B ' represent, independently of one another, an oxygen atom or a CH 2 group
- R and R ' represent, independently of one another, a halogen, such as chlorine, bromine, iodine or fluorine, or an alkyl group of 1 to 3 carbon atoms, linear or branched, if A and A' independently of one another represent a group CH 2 , then m and m 'independently of one another represent integers from 1 to 8, if A and A' are independently of one another an NH group or a group
- a physiologically acceptable acid salt derived from a compound of formula (I) such as a hydrochloride, a formate, a trifluoroacetate or an oxalate (HOOCCOOH),
- the invention also relates to compounds of formula (Ia) below
- n represents an integer from 1 to 12, especially from 1 to 8, m and m 'independently of one another represent integers from 2 to 8, q and q' independently represent one of other integers from 0 to 2, p and p 'independently of one another represent integers from 0 to 4,
- a and A 'independently of one another represent an NH group or a group
- R is an alkyl group of 1 to 3 carbon atoms, linear or branched
- B and B ' represent, independently of one another, an oxygen atom or a CH 2 group; - R and R' are, independently of each other, a halogen, such as chlorine, bromine, iodine or fluorine, or an alkyl group of 1 to 3 carbon atoms, linear or branched, or a physio-logically acceptable acid salt derived from a compound of formula (Ia) such as a hydrochloride, a formate, a trifluoroacetate or an oxalate (HOOCCOOH), the compounds of the following formulas being excluded:
- the formula of the compounds of formula (Ia) corresponds to the formula (VF), in which R "represents a hydrogen
- the formula of the compounds of formula (Ia) corresponds to formula (I), in which A and A 'represent independently from each other an NH group or a NR "group.
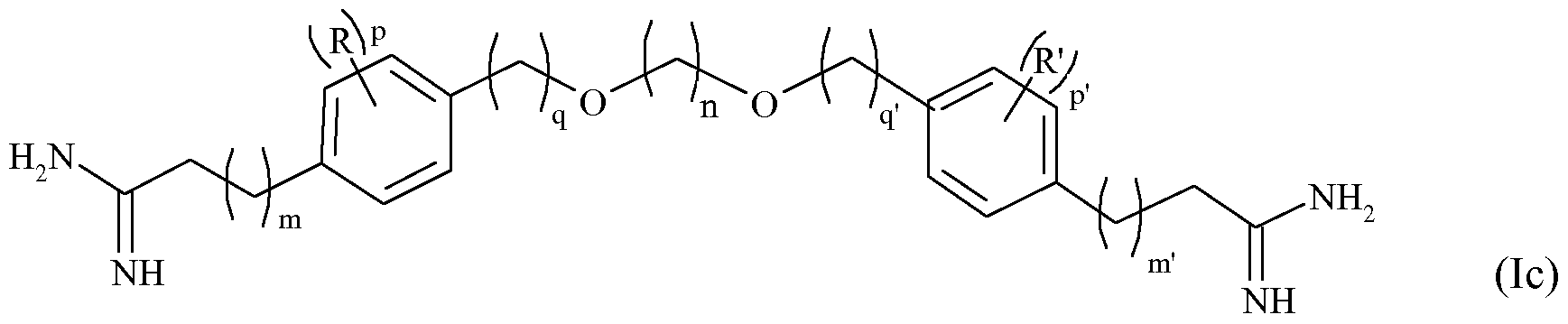
- the invention also relates to compounds of formula (Ib) below:
- n represents an integer from 1 to 12, especially from 1 to 8, m and m 'independently of one another represent integers from 1 to 8, q and q' independently represent one on the other, integers from 0 to 2, p and p 'independently of one another represent integers from 0 to 4, B and B' represent independently of each other an atom of oxygen or a CH 2 group, - R and R 'are independently of one another a halogen, such as chlorine, bromine, iodine or fluorine atoms, or an alkyl group of 1 to 3 carbon atoms, linear or branched, or a physiologically acceptable acid salt derived from a compound of formula (Ib) such as a hydrochloride, a formate, a trifluoroacetate or an oxalate (HOOCCOOH).
- a compound of formula (Ib) such as a hydrochloride, a formate, a trifluoroacetate or an oxalate (HOOCCO
- the formula of the compounds of formula (Ib) corresponds to formula (I), in which A and A 'represent a CH 2 group.
- the new compounds defined above can be used according to the present invention.
- the invention relates to compounds of formula (II) below:
- a physiologically acceptable acid salt derived from a compound of formula (II) such as hydrochloride, formate, trifluoroacetate or oxalate (HOOCCOOH).
- the formula of the compounds of formula (II) corresponds to formula (I), in which q and q 'are equal to 0.
- the compounds of formula (II) above, excluding the two compounds above, are new.
- the invention relates to compounds of formula (IF) below:
- a and A ' represent, independently of one another, an NH group or a NR "group, in which R" is an alkyl group of 1 to 3 carbon atoms, linear or branched, m and m' independently represent one of the other integers from 2 to 8,
- a physiologically acceptable acid salt derived from a compound of formula (IF) such as a hydrochloride, a formate, a trifluoroacetate or an oxalate (HOOCCOOH).
- a compound of formula (IF) such as a hydrochloride, a formate, a trifluoroacetate or an oxalate (HOOCCOOH).
- the formula of compounds of formula (IF) corresponds to formula (Ia), in which q and q 'are equal to 0.
- the formula of compounds (IF) corresponds to formula (II), in which A and A' represent independently of one another an NH group or a NR "group.
- the invention relates to compounds of formula (III) below:
- B, R, n, p are as defined above, and, if A represents a CH 2 group, then m represents an integer from 1 to 8, if A is an NH group or a NR "group, wherein R "is an alkyl group of 1 to 3 carbon atoms, linear or branched, then m represents an integer of 2 to 8,
- a physiologically acceptable acid salt derived from a compound of formula (III) such as hydrochloride, formate, trifluoroacetate or oxalate (HOOCCOOH).
- the formula of the compounds of formula (III) corresponds to formula (II), in which: m and m 'on the one hand, and p and p' on the other hand, are equal, and A and A 'd' on the one hand, and B and B 'on the other, represent the same atom or group.
- the compounds of formula (III) are symmetrical.
- the compounds of formula (III) above (excluding the two compounds above) are new.
- the invention relates to compounds of formula (IH ') below:
- A represents an NH group or a NR "group, in which R" is an alkyl group of 1 to 3 carbon atoms, linear or branched, and
- n represents an integer from 2 to 8, or a physiologically acceptable acid salt derived from a compound of formula (HF) such as a hydrochloride, a formate, a trifluoroacetate or an oxalate (HOOCCOOH).
- HF a physiologically acceptable acid salt derived from a compound of formula (HF) such as a hydrochloride, a formate, a trifluoroacetate or an oxalate (HOOCCOOH).
- a and A 'on the one hand, and B and B' on the other hand, represent the same atom or group.
- the formula of the compounds (HF) corresponds to the formula (III), in which A and A 'represent, independently of one another, an NH group or an NR "group.
- the invention relates to compounds of formula (IV) below:
- n 1, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11 or 12, or a physiologically acceptable acid salt derived from a compound of formula (IV) such as hydrochloride, formate, trifluoroacetate or oxalate (HOOCCOOH).
- a physiologically acceptable acid salt derived from a compound of formula (IV) such as hydrochloride, formate, trifluoroacetate or oxalate (HOOCCOOH).
- the formula of the compounds of formula (IV) corresponds to formula (III), in which A represents an NH group, B an oxygen atom, p is equal to 0 and m is equal to 2.
- the compounds of formula (IV) are symmetrical.
- the compounds of formula (IV) above (excluding the two compounds above) are new.
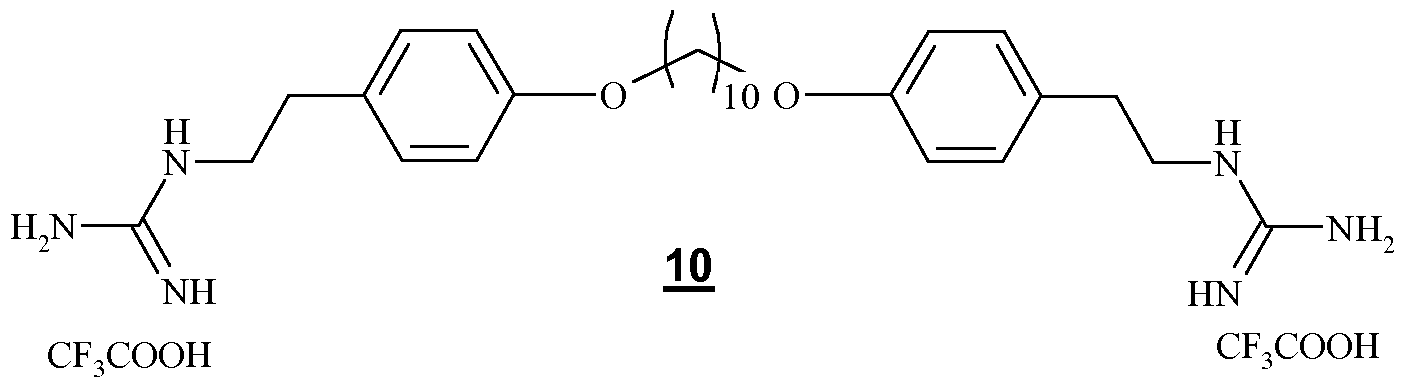
- the invention relates to compounds of formula 6 below:
- a physiologically acceptable acid salt derived from a compound of formula 6 such as hydrochloride, formate, trifluoroacetate or oxalate (HOOCCOOH).
- the formula of the compounds of formula 6 corresponds to formula (IV), wherein n is 6.
- the compound of formula 6 is symmetrical.
- the compound of formula 6 above is new.
- the invention relates to compounds of formula
- the invention relates to a pharmaceutical composition
- a pharmaceutical composition comprising as active substance at least one compound as defined above, in combination with a pharmaceutically acceptable vehicle.
- the pharmaceutical composition comprises as active substance:
- the first case corresponds to the compounds of formulas (VI), (I) and (II).
- the second case corresponds to the compounds of formulas (III), (IV) and 6, for which m is equal to m '. These compounds are soluble in aqueous solution.
- the pharmaceutical composition comprises as active substance: a compound as defined above, in particular for which the sum m + m '+ n is greater than 10, or a compound as defined above. above, for which the sum 2m + n is greater than 10, said pharmaceutical composition being formulated in hydroalcoholic solution.
- the first case corresponds to the compounds of formulas (VI), (I) and (II).
- the second case corresponds to the compounds of formulas (III), (IV) and 6, for which m is equal to m '.
- hydroalcoholic solution denotes a mixture of water and at least one alcohol, especially chosen from ethanol, isopropyl alcohol and benzyl alcohol.
- the pharmaceutical composition as defined above is orally administrable, and in said pharmaceutical composition, the active substance is at a concentration of about 0.1 to about 5 mg / kg body weight.
- the pharmaceutical composition as defined above is administrable topically, and in said pharmaceutical composition, the active substance is at a concentration of about 0.1% to about 1% relative to to the total weight in the pharmaceutical composition.
- topically means that the pharmaceutical composition is applied to localized locations, for example the skin or mucosa.
- compositions in capsule, tablet, powder, cream, lotion, aqueous or hydroalcoholic solution, mouthwash, eye drops, milk, foam, gel, spray or powder, for example.
- the invention also relates to the process for preparing a compound of formula (Ia) comprising the following steps of:
- n represents an integer from 1 to 12, in particular from 1 to 8, m and m 'independently of one another represent integers from 2 to 8, q and q' independently represent one of the other integers from 0 to 2, p and p 'independently of one another represent integers from 0 to 4, A and A 'independently of one another represent an NH group or a group
- R is an alkyl group of 1 to 3 carbon atoms, linear or branched
- B and B ' represent, independently of one another, an oxygen atom or a CH 2 group
- R and R ' represent, independently of one another, a halogen, such as chlorine, bromine, iodine or fluorine atoms, or an alkyl group of 1 to 3 carbon atoms, linear or branched,
- Y represents a protective group of amines, in particular Boc or Fmoc.
- n represents an integer from 1 to 12, especially from 1 to 8, m and m 'independently of one another represent integers from 2 to 8, q and q' independently represent one on the other integers from 0 to 2, p and p 'independently of one another represent integers from 0 to 4,
- a and A 'independently of one another represent an NH group or a group
- R is an alkyl group of 1 to 3 carbon atoms, linear or branched
- B and B ' represent, independently of one another, an oxygen atom or a CH 2 group
- R and R ' represent, independently of one another, a halogen, such as chlorine, bromine, iodine or fluorine atoms, or an alkyl group of 1 to 3 carbon atoms, linear or branched, or,
- GP represents a Leaving Group such as -SR, -NTf or N and Y represents a protective group of amines, in particular Boc or Fmoc,
- n represents an integer from 1 to 12, especially from 1 to 8, - m and m 'independently of one another represent integers from 2 to 8, q and q' independently represent number of integers from 0 to 2, p and p 'independently of one another represent integers from 0 to 4, A and A' represent, independently of one another, an NH group or a group NR ", in which R" is an alkyl group of 1 to 3 carbon atoms, linear or branched,
- B and B ' represent, independently of one another, an oxygen atom or a CH 2 group
- R and R ' represent, independently of one another, a halogen, such as chlorine, bromine, iodine or fluorine atoms, or an alkyl group of 1 to 3 carbon atoms, linear or branched,
- Y represents a protective group of amines, in particular Boc or Fmoc.
- n represents an integer from 1 to 12, especially from 1 to 8, m and m 'independently of one another represent integers from 2 to 8, q and q' independently represent number of integers from 0 to 2, p and p 'independently of one another represent integers from 0 to 4, - A and A' represent, independently of one another, a group NH or a group
- R is an alkyl group of 1 to 3 carbon atoms, linear or branched
- B and B ' represent, independently of one another, an oxygen atom or a CH 2 group; - R and R' are, independently of each other, a halogen, such as chlorine, bromine, iodine or fluorine, or an alkyl group of 1 to 3 carbon atoms, linear or branched, or,
- an acid salt derived from a compound of formula (Ia) such as a hydrochloride, a formate, a trifluoroacetate or an oxalate (HOOCCOOH).
- linear or branched alkyl group of 1 to 3 carbon atoms denotes methyl, ethyl, n-propyl and isopropyl groups.
- the neutral form of the compound can be obtained by neutralizing the medium after reaction.
- the isolation of the compounds is facilitated when the compounds of formula (Ia), and in particular (XF), are in the form of acid salts.
- Step b) makes it possible to form guanidine function.
- This step is generally carried out in the presence of a base, for example triethylamine or diisopropylethylamine, and as an organic solvent, such as dichloromethane, although the use of other organic solvents is also possible, for example THF, methanol, acetonitrile, dioxane, or a mixture of these solvents.
- a base for example triethylamine or diisopropylethylamine
- organic solvent such as dichloromethane
- the method of preparation, as defined above, of a compound of formula (IV) comprises:
- n 1, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11 or 12
- Y is a protecting group for amines, especially Boc or Fmoc.
- n 1, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11 or 12, or
- an acid salt derived from a compound of formula (XXI) such as a hydrochloride, a formate, a trifluoroacetate or an oxalate (HOOCCOOH),
- GP represents a Leaving Group such as -SR, -NTf or -N and Y represents a protective group of amines, in particular Boc or Fmoc,
- n 1, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11 or 12
- Y is a protecting group for amines, especially Boc or Fmoc.
- n 1, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11 or 12, or
- an acid salt derived from a compound of formula (IV) such as a hydrochloride, a formate, a trifluoroacetate or an oxalate (HOOCCOOH).
- the invention relates to the compound of formula (X ') below in which: n represents an integer from 1 to 12, especially from 1 to 8, m and m 'independently of one another represent integers from 2 to 8, q and q' independently represent one on the other integers from 0 to 2, - p and p 'independently of one another represent integers from 0 to 4,
- a and A 'independently of one another represent an NH group or a group
- R is an alkyl group of 1 to 3 carbon atoms, linear or branched
- B and B ' represent, independently of one another, an oxygen atom or a CH 2 group
- R and R 'independently of one another represent a halogen, such as chlorine, bromine, iodine or fluorine, or an alkyl group of 1 to 3 carbon atoms, linear or branched, Y represents a protective group amines, especially Boc or Fmoc.
- n 1, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11 or 12
- Y is a protecting group for amines, especially Boc or Fmoc.
- linear or branched alkyl group of 1 to 3 carbon atoms denotes methyl, ethyl, n-propyl and isopropyl groups.
- the compound of formula (X ') as defined above is an intermediate for the preparation of compounds of formula (I) such that defined above, and the compound of formula (XX) as defined above is an intermediate for the preparation of compounds of formula (IV) as defined above.
- XXX wherein X 1 and X 'are, independently of each other, a halogen, especially chlorine, bromine or iodine, n represents an integer from 1 to 12; , especially from 1 to 8,
- n represents an integer from 1 to 8
- q represents an integer from 0 to 2
- p represents an integer of 0 to 4
- R represents a halogen, such as chlorine, bromine, iodine or fluorine atoms, or an alkyl group of 1 to 3 carbon atoms, linear or branched,
- R ' represents a halogen, such as chlorine, bromine, iodine or fluorine atoms, or an alkyl group of 1 to 3 carbon atoms, linear or branched, steps a) and b) can be performed simultaneously ("one pot" reaction) or successively,
- n represents an integer from 1 to 12, especially from 1 to 8, m and m 'independently of one another represent integers from 1 to 8, q and q' independently represent one of the other of integers from 0 to 2, p and p 'independently of one another represent integers from 0 to 4, R and R' are independently of each other a halogen, such as chlorine, bromine, iodine or fluorine atoms, or an alkyl group of 1 to 3 carbon atoms, linear or branched,
- n represents an integer from 1 to 12, in particular from 1 to 8, m and m 'independently of one another represent integers from 1 to 8, and q' and q 'independently represent one of the other of integers from 0 to 2, p and p 'independently of one another represent integers from 0 to 4,
- R and R' are independently of each other a halogen, such as chlorine, bromine, iodine or fluorine atoms, or an alkyl group of 1 to 3 carbon atoms, linear or branched, or a physiologically acceptable acid salt derived from a compound of formula (Ic) such as a hydrochloride, a formate, a trifluoroacetate or an oxalate (HOOCCOOH).
- the Pinner reaction comprises two steps: a reaction of the nitrile function with ethanol in the presence of acid, in particular hydrochloric acid, followed by a reaction with ammonia, to result in the formation of the amidine function .
- n represents an integer from 1 to 12, especially from 1 to 8, q and q 'independently of one another represent integers from 0 to 2, p and p' independently represent one on the other, whole numbers from 0 to 4,
- R and R ' are, independently of one another, a halogen, such as chlorine, bromine, iodine or fluorine atoms, or an alkyl group of 1 to 3 carbon atoms, linear or branched,
- n represents an integer from 1 to 12, especially from 1 to 8, q and q 'independently of one another represent integers from 0 to 2, p and p' independently represent one of other integers from 0 to 4, R and R 'are, independently of one another, a halogen, such as chlorine, bromine, iodine or fluorine, or an alkyl group of 1 to 3 carbon atoms, linear or branched,
- X represents a leaving group, in particular a halogen atom such as the chlorine, bromine or iodine atoms
- n represents an integer from 1 to 12, especially from 1 to 8, m and m 'independently of one another represent integers from 1 to 8, q and q' independently represent one on the other integers from 0 to 2, p and p 'independently of one another represent integers from 0 to 4,
- R and R ' are, independently of one another, a halogen, such as chlorine, bromine, iodine or fluorine atoms, or an alkyl group of 1 to 3 carbon atoms, linear or branched,
- n represents an integer from 1 to 12, in particular from 1 to 8, m and m 'independently of one another represent integers from 1 to 8, and q' and q 'independently represent one of the other of integers from 0 to 2, p and p 'independently of one another represent integers from 0 to 4,
- R and R' are independently of each other a halogen, such as chlorine, bromine, iodine or fluorine atoms, or an alkyl group of 1 to 3 carbon atoms, linear or branched,
- a physiologically acceptable acid salt derived from a compound of formula (Id) such as hydrochloride, formate, trifluoroacetate or oxalate (HOOCCOOH).
- the residual aqueous phase is added with 40 ml of AcOEt, and the pH of the mixture is lowered to 7-8 using an aqueous solution of 1M HCl.
- the phases are then separated and the aqueous phase is washed with AcOEt (2 * 15 mL).
- the combined organic phases are then dried over Na 2 SO 4 , filtered and evaporated.
- the crude is then purified by chromatographic column (Al 2 O 3 , CH 2 Cl 2 / Hex, 85:15) to give N- (t-butoxycarbonyl) -2- (4-hydroxyphenyl) ethylamine (0.98 g 73%).
- N- (t-butoxycarbonyl) -2- (4-hydroxyphenyl) ethylamine (1.5 g, 6.32 mmol, leq) is dissolved in CH 3 CN (freshly distilled over CaH 2 ) and added with K 2 CO 3 (1.74 g, 12.64 mmol, 2 eq).
- the mixture is refluxed under argon for about two hours before the addition of dichlorohexane (0.55 mL, 3.79 mmol, 0.6 eq.). After 24 hours at reflux, 0.3 equivalents of dichloro-hexane are added. After 48 hours of reaction, the return to ambient temperature is followed by evaporation of the solvent.
- the crude obtained is solubilized in CH 2 Cl 2 and filtered on sintered glass to remove the mineral impurities.
- the filtrate is then concentrated to a minimum of solvent and precipitated by addition of MeOH accompanied by cold evaporation.
- the solid obtained is then filtered, rinsed with MeOH and dried under vacuum. It corresponds to the expected compound in the form of a white powder (0.52 g;
- a variant using DMF or another amide (dimethylacetamide, N-methylpyrrolidone for example) as solvent is applicable at room temperature.
- UV-Vis H 2 O: 195 (9326.7); 221 (2238.7).
- 1,6-bis (4- (2-guanidinoethyl) phenoxy) hexane using respectively diiodomethane, dibromopropane, dibromobutane, dibromopentane, dibromoheptane, dibromooctane, dibromononane, dibromodecane and dibromdodecane in place of dichloro-hexane), or the corresponding dichloro-, dibromo-, diiodo or di-para-toluenesulfonylalkanes.
- 1,7-bis (4- (2-guanidinoethyl) phenoxy) heptane compound 1H-NMR (400 MHz, D 2 O): 1.42 (m, 6H, OCH 2 CH 2 CH 2 ); 1.72 (m, 4 ⁇ ,
- Variants using DMF in the presence of NaH at 50 ° C. or CH 3 CN in the presence of K 2 CO 3 at 90 ° C. are also applicable.
- the bis-propionitrile previously obtained is hydrolyzed in a mixture of acetic acid / concentrated sulfuric acid / water at approximately 100 ° C.
- the addition of cold water precipitates the diacid which is collected by filtration, washed with water and dried under water. empty.
- This diacid is then converted into ethyl diester by treatment with a mixture of concentrated sulfuric acid / ethanol at reflux, followed by precipitation by addition of water, filtration, washing with water and drying, then chromatography if necessary.
- An alternative to this esterification may be the reaction of the diacid with two equivalents of KOH (or CsOH) in water, followed by lyophilization, yielding the double potassium salt (or cesium).
- This salt is reacted with about two equivalents of ethyl halide (or tosylate or mesylate) in anhydrous dimethylformamide.
- an excess of ice water is added to give a precipitate of the diester (or a gum) which is separated by filtration (or decantation).
- the crude bis (4- (2- (ethoxycarbonyl) ethyl) phenoxy) alkane diester is purified by chromatography.
- the ethyl diester previously obtained is treated in anhydrous THF (or diethyl ether) with one equivalent of lithium aluminum hydride.
- a chromatographic control incorporating a treatment of the sample according to the standard of this reaction is carried out before stopping the reaction.
- Excess hydride is destroyed by careful addition of water, or ethyl acetate.
- the solid residue is removed by filtration, and evaporation of the filtrate in vacuo gives the desired bis (4- (hydroxypropyl) phenoxy) alkane.
- the latter can be purified on a short column of silica or alumina.
- the bis (4- (hydroxypropyl) phenoxy) alkane above is solubilized in dichloromethane, with two equivalents of triphenylphosphine.
- the solution is cooled to 0 ° C., and an excess of carbon tetrachloride is added.
- the reaction medium is stirred at room temperature and the reaction is monitored by chromatography; when all the diol has disappeared, the solvents are evaporated and the residue is chromatographed on silica or alumina to give the desired bis (4- (chloropropyl) phenoxy) alkane.
- the dibromo derivative can be obtained in a similar manner by replacing carbon tetrachloride with carbon tetrabromide.
- a synthetic alternative may be the addition of an excess of thionyl chloride at 0 ° C. to a solution of diol in anhydrous dichloromethane.
- the reaction is monitored by chromatography. When the alcohol is consumed, the solvent and the excess of thionyl chloride are distilled or evaporated under vacuum, to give the crude dichlorinated derivative, which can be chromatographed.
- the above bis (4- (chloropropyl) phenoxy) alkane is dissolved in dry dimethylsulfoxide, and sixteen equivalents of potassium cyanide (or sodium) are added all at once.
- the mixture is heated under an inert atmosphere at 80 ° C.
- the reaction is monitored by chromatography. When all the dichlorinated derivative has been consumed, the mixture is cooled to room temperature and ice water is added to give a precipitate (or gum), isolated by filtration or decantation. After drying, the solid or gum obtained is chromatographed to give the desired bis (4- (cyanopropyl) phenoxy) alkane.
- the solid obtained is solubilized in a minimum of CH 2 Cl 2 and again precipitated by the addition of an excess of MeOH. After filtration, washing with MeOH and drying, the pure expected compound is obtained in the form of white flakes (0.45 g, 41%).
- Variants using DMF in the presence of NaH at 50 ° C. or CH 3 CN in the presence of K 2 CO 3 at 90 ° C. are also applicable.
- 1,10-Bis (4- (2-cyanoethyl) phenoxy) decane are prepared following the procedures described above to give 1,1-bis (4- (2-cyanoethyl) phenoxy) methane (respectively using dibromopropane, dibromobutane, dibromopentane, dibromohexane, dibromoheptane, dibromooctane, dibromononane and dichlorodecane in place of dichloromethane), or the corresponding dichloro- or diiodo or di-para-toluenesulfonylalkanes.
- a solution of a bis (4- (cyanoalkyl) phenoxy) alkane derivative in a mixture of ethanol and anhydrous benzene is cooled to 0 ° C. and is then treated by bubbling gaseous hydrochloric acid for approximately 1 hour.
- the acid solution is stored at 4 ° C for several days.
- the volatile portion of hydrochloric acid is removed by bubbling dry nitrogen, and the degassed solution is evaporated to dryness under reduced pressure.
- the evaporation residue containing bis (4- (ethyl) imidatoalkyl) phenoxy alkane dihydrochloride is taken up in an IM solution of ammonia in ethanol.
- the mixture is heated at 50-60 ° C. under anhydrous conditions during 2-3h, then left stirring at room temperature overnight.
- the resulting medium is filtered and then diethyl ether is added, precipitating the desired bis (4- (amidinoalkyl) phenoxy) alkane dihydrochloride.
- the bis (4- (cyanoalkyl) phenoxy) alkane compound is dissolved in an EtOH / benzene mixture and is bubbled under stirring at 4 ° C HCl gas for about two hours. The mixture is then left stirring at room temperature for 48 hours before the evaporation in vacuo of the solvents.
- the oil obtained is then solubilized in a minimum of EtOH and the expected bis-imidate is precipitated by addition of a large excess of Et2O.
- the white precipitate is then filtered on frit, washed with Et 2 O and dried under vacuum.
- the desired compound is obtained in the form of a white powder.
- EtOH and gaseous NH 3 was bubbled for about 1 h 30 min.
- the medium is then refluxed under argon for 5 to 6 hours.
- the return to ambient temperature is followed by the precipitation of the bis-amidine salt by adding an excess of Et 2 O in the medium.
- the precipitate is filtered, washed with ether and dried under vacuum.
- the expected product is obtained in the form of a white powder.
- the 1,2-diphenylalkane is treated with dichloromethylether in the presence of zinc chloride (Hager et al., J. AM Pharm Ass, 1952, XLI, 3 (6), 115-118, Reichstein and Oppenauer, HeIv. Chim. Acta, 1933, 16, 1380) or with chloromethyl-n-octyl ether in the presence of titanium tetrachloride, or any other chloromethylating agent, to give bis (para-chloromethyl) -diphenylalkane.
- zinc chloride Hager et al., J. AM Pharm Ass, 1952, XLI, 3 (6), 115-118, Reichstein and Oppenauer, HeIv. Chim. Acta, 1933, 16, 1380
- chloromethyl-n-octyl ether in the presence of titanium tetrachloride, or any other chloromethylating agent, to give bis (para-chloromethyl) -diphenylalkane.
- Bis (para-chloromethyl) -diphenylalkane is treated with NaCN in a hydroalcoholic mixture (Hager et al., J. AM Pharm Ass, 1952, XLI, 3 (6), 115-118, Reichstein and Oppenauer, HeIv. Chim. Acta, 1933, 16, 1380), or in any other solvent promoting this nucleophilic substitution to give the bis (para-cyanomethyl) -diphenylalkane
- the bis (4-cyanoalkyl) -diphenylalkane in question is dissolved in anhydrous THF under an inert atmosphere.
- borane complex 1M THF in tetrahydrofuran are added, and the mixture is stirred for 30 minutes at room temperature and then brought to reflux for 3 to 4 hours. After cooling to room temperature, the mixture is added to excess methanol to destroy the excess borane. Trimethoxyborane, methanol and tetrahydrofuran are evaporated. The evaporation residue is taken up in a methanol / water / hydrochloric acid mixture, and the mixture is refluxed for 2 hours.
- the solvents are evaporated to dryness to give the desired bis (4 - ((aminomethyl) alkyl) diphenylalkane dihydrochloride
- the free bisamine is obtained by treatment of an aqueous solution of hydrochloride in the presence of a mineral base, followed by The organic phase is dried over sodium sulphate, filtered and evaporated under reduced pressure.
- the bis (4-aminoalkyl) phenyl alkane dihydrochloride is suspended in anhydrous CH 2 Cl 2 and solubilized by the addition of a minimum of MeOH. Triethylamine is then added, as well as ⁇ /, N'-Bis (tert-butoxycarbonyl) -N "-triflylguanidine and the whole is placed at room temperature, under an inert atmosphere and with stirring. The solvent is removed by evaporation under The crude product obtained is solubilized with CH 2 Cl 2 , washed with an aqueous solution of 2M NaHSO 4 , then with a saturated aqueous solution of NaHCO 3, and the organic phase is then dried over Na 2 SO 4 , filtered, concentrated and dried. under vacuum to give the protected compound.
- a solution of a bis (4- (cyanoalkyl) phenyl) alkane derivative in a mixture of ethanol and anhydrous benzene is cooled to 0 ° C. and is then treated by bubbling gaseous hydrochloric acid for approximately 1 hour.
- the acid solution is stored at 4 ° C for several days.
- the volatile portion of hydrochloric acid is removed by bubbling dry nitrogen, and the degassed solution is evaporated to dryness under reduced pressure.
- the evaporation residue, containing bis (4- (ethyl) imidatoalkyl) phenyl) alkane dihydrochloride is taken up in an IM solution of ammonia in ethanol.
- Escherichia coli ATCC 25922 Pseudomonas aeruginosa ATCC 27853, Staphylococcus aureus ATCC 25923 & ATCC 29213 and Enterococcus faecalis ATCC 29212 .
- the study also included 5 clinical isolates obtained from rectal swabs and / or blood culture:
- EfR2 E. faecalis resistant to teicoplanin and vancomycin (van A gene)
- PaR1 P. aeruginosa overexpressing an efflux pump
- the isolates were chosen on the basis of their resistance profile. Resistance profiles were determined on VITEK2 (BioMerieux, France).
- the bacteria are cultured either on Mueller-Hinton agar (Difco, 225250) or Mueller-Hinton broth (Difco, 275730), at 35 ° C (Grare M, Mourer M, Fontanay S, Regnouf- de- Vains JB, Finance C, Duval RE.
- Gram M Mourer M
- Fontanay S Regnouf- de- Vains JB
- Finance C Duval RE.
- In vitro activity of para-guanidinoethylcalix [4] arene against susceptible and antibiotic-resistant Gram-negative and Gram-positive bacteria. J.
- the different molecules tested were prepared in the form of aqueous solution at 10 -2 mol / l.
- the solutions used for the biological tests are freshly prepared and stored for a maximum of one week at 4 ° C.
- the solutions are filtered through a 0.22 ⁇ m pore filter (Millex®GP, 0.22 ⁇ m, Millipore, France) before each test.
- guanidino compounds 1, 3, 4, 6, 7, 9 and 10 in the form of trifluoroacetic acid salts, amidino compounds 103, 104, 106, 107, 108 and 110 in the form of salts.
- MICs Minimum Inhibitory Concentrations
- bacterial suspensions are prepared from an isolated colony taken from Mueller-Hinton agar taken up in 5 mL of Mueller-Hinton broth. After 24 hours of growth, the bacterial suspensions are diluted in sterile distilled water to obtain an inoculum of 10 5 - 10 6 CFU / mL. The purity of the bacterial suspensions is controlled by agar isolation and Gram stain.
- the MICs are determined using an ELISA plate reader (at 540 nm, Multiskan EX, Thermo Electron Corporation, France) as the concentrations of active substance for the absorbance is comparable to that of the negative control (culture medium alone or with the drug, without the inoculum). The results are the average of 4 independent experiments.
- MIC Minimum inhibitory concentrations
- Table 3.1 MIC values (mg / L) obtained by the liquid microdilution method, according to the procedures of CLSI and CA-SFM.
- Table 3.2 MIC values (mg / L) obtained by the liquid microdilution method, according to the procedures of CLSI and CA-SFM.
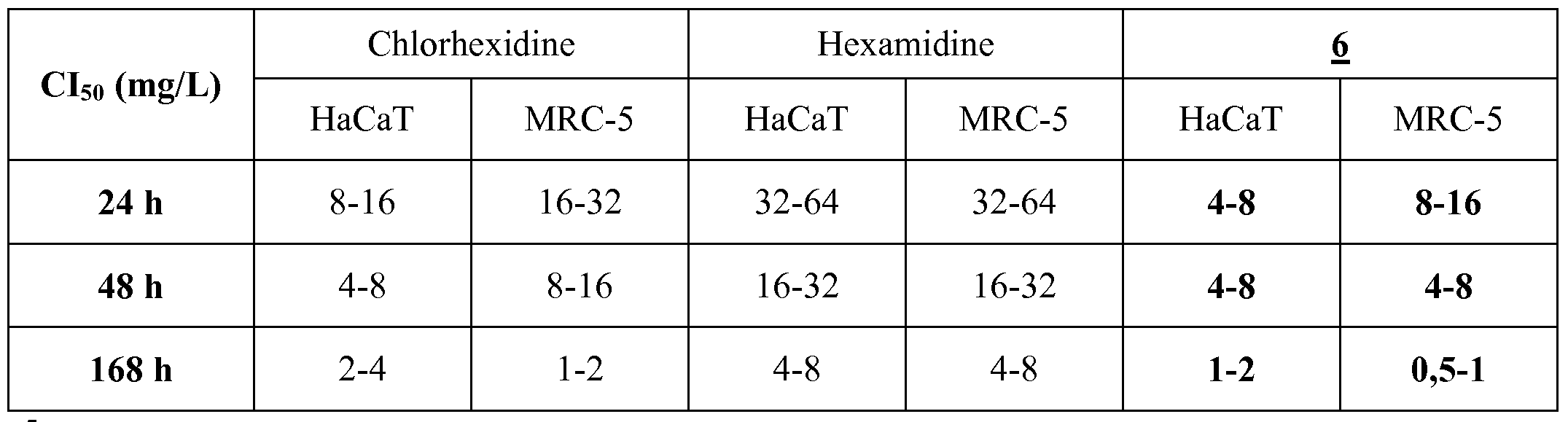
- both hexamidine and chlorhexidine have broad-spectrum antibacterial activity (ie, antibacterial activity against Gram-positive and Gram-negative bacteria), with these two molecules (hexamidine and chlorhexidine) having a lower activity on P. aeruginosa.
- hexamidine and chlorhexidine exhibit comparable antibacterial activity whether reference bacteria (ATCC strains) or clinical isolates are considered.
- hexamidine and chlorhexidine show better activity on gram-positive shells (S. aureus and Enterococcus spp., With the exception of chlorhexidine with respect to E. coli bacteria.
- These molecules show activity on Gram-positive and Gram-negative bacteria, with a higher activity on Gram-positive cocci (and more particularly Staphylococcus spp., And S. aureus), and 6 maintain comparable activity on isolates. clinics.
- Compound 6 exhibits an overall antibacterial activity better than hexamidine and comparable to that of chlorhexidine, in particular as regards its activity with respect to Staphylococci.
- Compounds 9 and 10 both show very good activity on the 5 reference strains tested, equivalent or superior to those of compound 6.
- the eukaryotic cells used in our study are the HaCaT cells (human keratinocytes) which were rationally provided by the Pierre Fabre Institute (Toulouse, France); and MRC-5 cells (human embryonic fibroblasts) which were obtained from BioMerieux (Lyon, France). These cells are grown in DMEM ("Dulbecco's Modified Eagle's Medium", Invitrogen 61965) for HaCaT cells, and MEM ("Modified Eagle's Medium", Invitrogen 41090); 10% fetal calf serum (SVF, Invitrogen, 10270, Lot 40Q5150K) in a humid chamber at 37 ° C. under 5% CO 2 .
- the cytotoxicity test is based on the incorporation of a vital dye, Neutral Red, into the lysosomes of viable cells, following exposure of the cells to the different concentrations of the test molecules.
- the amount of dye, after extraction from the lysosomes, is quantified using a spectrophotometer and compared to that obtained in the case of "control" cells, not exposed to the test molecules.
- the medium contained in each of the wells is removed and replaced with 200 ⁇ L medium without phenol red (Invitrogen, 51200) containing 50 ⁇ g. / mL Neutral Red for an additional 3 h incubation.
- the cells are then rinsed 3 times with a solution of PBS. After the last PBS wash, 200 ⁇ L of a 1% acetic acid / 50% ethanol solution is added to each of the wells to extract the dye from the viable cell lysosomes.
- Absorbance is measured at 540 nm, with a reference wavelength at 690 nm, using an ELISA plate reader (Multiskan EX, Thermo Electron Corporation, France). The results are the average of the values obtained for 8 wells per concentration and per time unit. Each experiment was performed 3 times.
- Table 4 IC 50 determined from the values obtained by the viability test (at MTT). HaCaT and MRC-5 cell lines were cultured as an adherent culture on 96-well microplates.
- the compounds were added at concentrations of 1 to 256 mg / L for 24, 48 and 168 h.
- IC50s are representative of three independent determinations.
- Table 5 CC50 determined from the values obtained by the cytotoxicity test (at RN). HaCaT and MRC-5 cell lines were cultured as an adherent culture on 96-well microplates.
- the compounds were added at concentrations of 1 to 256 mg / L for 24, 48 and 168 h.
- CC50s are representative of three independent determinations.
- Table 6 Selectivity index obtained for 5 reference strains, after 24 hours of exposure to the compound, on HaCaT cell lines. (1,1,6-bis (4- (2-guanidinoethyl) phenoxy) hexane.
- the IS values are globally very close to each other, which shows the interest of compound 6 vis-à-vis "commercial” compounds widely advocated "in town and in hospital” for Gram bacteria infections. positive, and more particularly S. aureus infections.
Landscapes
- Chemical & Material Sciences (AREA)
- Organic Chemistry (AREA)
- Health & Medical Sciences (AREA)
- Life Sciences & Earth Sciences (AREA)
- Veterinary Medicine (AREA)
- Chemical Kinetics & Catalysis (AREA)
- General Health & Medical Sciences (AREA)
- Medicinal Chemistry (AREA)
- Nuclear Medicine, Radiotherapy & Molecular Imaging (AREA)
- Pharmacology & Pharmacy (AREA)
- Public Health (AREA)
- Animal Behavior & Ethology (AREA)
- General Chemical & Material Sciences (AREA)
- Communicable Diseases (AREA)
- Oncology (AREA)
- Tropical Medicine & Parasitology (AREA)
- Virology (AREA)
- Pharmaceuticals Containing Other Organic And Inorganic Compounds (AREA)
- Organic Low-Molecular-Weight Compounds And Preparation Thereof (AREA)
- Acyclic And Carbocyclic Compounds In Medicinal Compositions (AREA)
Abstract
Description
Claims
Priority Applications (4)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| NZ591021A NZ591021A (en) | 2008-07-10 | 2009-07-10 | Phenoxyalkanes or phenylalkanes with guanidine or imine groups, and their use in treating various infections |
| EP09794039A EP2303837A2 (fr) | 2008-07-10 | 2009-07-10 | Nouveaux composes, leur utilisation comme medicaments, et leur procede de preparation |
| US13/003,330 US8703825B2 (en) | 2008-07-10 | 2009-07-10 | Compounds, use thereof as medicaments, and method of preparation thereof |
| CA2730304A CA2730304A1 (fr) | 2008-07-10 | 2009-07-10 | Nouveaux composes, leur utilisation comme medicaments, et leur procede de preparation |
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| FR0803940A FR2933692B1 (fr) | 2008-07-10 | 2008-07-10 | Nouveaux composes, leur utilisation comme medicaments, et leur procede de preparation |
| FR08/03940 | 2008-07-10 |
Publications (2)
| Publication Number | Publication Date |
|---|---|
| WO2010004231A2 true WO2010004231A2 (fr) | 2010-01-14 |
| WO2010004231A3 WO2010004231A3 (fr) | 2010-03-04 |
Family
ID=40289121
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/FR2009/051388 WO2010004231A2 (fr) | 2008-07-10 | 2009-07-10 | Nouveaux composes, leur utilisation comme medicaments, et leur procede de preparation |
Country Status (6)
| Country | Link |
|---|---|
| US (1) | US8703825B2 (fr) |
| EP (1) | EP2303837A2 (fr) |
| CA (1) | CA2730304A1 (fr) |
| FR (1) | FR2933692B1 (fr) |
| NZ (1) | NZ591021A (fr) |
| WO (1) | WO2010004231A2 (fr) |
Cited By (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2012147092A3 (fr) * | 2011-03-03 | 2013-03-14 | Cadila Healthcare Limited | Nouveaux sels d'un inhibiteur de la dpp-4 |
Citations (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| GB938042A (en) | 1961-03-30 | 1963-09-25 | Wellcome Found | Phenoxyalkanes |
Family Cites Families (4)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US3896235A (en) * | 1970-09-01 | 1975-07-22 | Burroughs Wellcome Co | Anthelmintic ethers and composition thereof |
| DE2658329C2 (de) * | 1976-12-23 | 1983-01-13 | Hoechst Ag, 6000 Frankfurt | Nitroaminäther, Verfahren zu ihrer Herstellung und ihre Verwendung als Disazoverbindungen |
| US20040006092A1 (en) * | 2001-08-31 | 2004-01-08 | Neurochem, Inc. | Amidine derivatives for treating amyloidosis |
| JP2006501160A (ja) * | 2002-06-07 | 2006-01-12 | ユニバーシティ オブ ノース カロライナ アット チャペル ヒル | アミロイド症を処置するためのアミジン誘導体 |
-
2008
- 2008-07-10 FR FR0803940A patent/FR2933692B1/fr active Active
-
2009
- 2009-07-10 NZ NZ591021A patent/NZ591021A/xx not_active IP Right Cessation
- 2009-07-10 CA CA2730304A patent/CA2730304A1/fr not_active Abandoned
- 2009-07-10 EP EP09794039A patent/EP2303837A2/fr not_active Withdrawn
- 2009-07-10 US US13/003,330 patent/US8703825B2/en not_active Expired - Fee Related
- 2009-07-10 WO PCT/FR2009/051388 patent/WO2010004231A2/fr active Application Filing
Patent Citations (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| GB938042A (en) | 1961-03-30 | 1963-09-25 | Wellcome Found | Phenoxyalkanes |
Non-Patent Citations (1)
| Title |
|---|
| DARDONVILLE ET AL., J MED. CHEM., vol. 49, no. 12, 2006, pages 3748 |
Cited By (3)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2012147092A3 (fr) * | 2011-03-03 | 2013-03-14 | Cadila Healthcare Limited | Nouveaux sels d'un inhibiteur de la dpp-4 |
| CN104788456A (zh) * | 2011-03-03 | 2015-07-22 | 卡迪拉保健有限公司 | Dpp-iv抑制剂的新的盐 |
| US9108972B2 (en) | 2011-03-03 | 2015-08-18 | Cadila Healthcare Limited | Salts of DPP-IV inhibitor |
Also Published As
| Publication number | Publication date |
|---|---|
| US20110184070A1 (en) | 2011-07-28 |
| FR2933692A1 (fr) | 2010-01-15 |
| CA2730304A1 (fr) | 2010-01-14 |
| NZ591021A (en) | 2012-11-30 |
| EP2303837A2 (fr) | 2011-04-06 |
| WO2010004231A3 (fr) | 2010-03-04 |
| US8703825B2 (en) | 2014-04-22 |
| FR2933692B1 (fr) | 2022-12-02 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| EP3463294B1 (fr) | Compositions sous forme d'une solution aqueuse injectable comprenant du glucagon humain et un co-polyaminoacide statistique | |
| FI95800C (fi) | Menetelmä terapeuttisesti käyttökelpoisten amino- ja nitrosubstituoitujen trisyklisten yhdisteiden valmistamiseksi | |
| US20150018564A1 (en) | Amide compound or salt thereof, biofilm inhibitor, biofilm remover and disinfectant containing the same | |
| US10562842B2 (en) | Use of polyaminoisoprenyl derivatives in antibiotic or antiseptic treatment | |
| US20190269719A1 (en) | Polyguanidine polymers and methods of use thereof | |
| EP0402266A2 (fr) | Lauramides N-substitués, leur préparation et compositions les contenant | |
| EP0288352B1 (fr) | Dérivés aromatiques, leur préparation et leur utilisation comme antimicrobiens | |
| US20100298206A1 (en) | Hybrid oligomers, their preparation process and pharmaceutical compositions containing them | |
| EP0087378B1 (fr) | Ethers-oximes d'alcoylaminoalcools comme médicaments, produits nouveaux et procédés pour leur préparation | |
| WO2010004231A2 (fr) | Nouveaux composes, leur utilisation comme medicaments, et leur procede de preparation | |
| EP1640368A2 (fr) | Composés urées cycliques et leur procédé de préparation | |
| US8664384B2 (en) | N6-(ferrocenmethyl)quinazolin-2,4,6-triamina (H2) and the derivatives and prodrugs thereof as antileishmanial, antiprotozoal, antiparasitic and antimicrobial agents | |
| RU2004129734A (ru) | Производные изохинолона | |
| BRPI0718014A2 (pt) | Composto de amida ou sal do mesmo, e inibidor de formação de biopelícula, removedor de biopelícula e bactericida, cada um utilizando o composto de amida ou sal do mesmo. | |
| EP4294820A1 (fr) | Peptides cycliques pénétrant les cellules avec trois résidus hydrophobes ou plus | |
| JP2014501750A5 (fr) | ||
| CN113227088A (zh) | 单环β-内酰胺化合物在制药中的应用 | |
| EP3237015B1 (fr) | Derives hydroxybisphosphoniques hydrosolubles de la doxorubicine | |
| WO2008050858A1 (fr) | Composé amide, son sel et agent d'élimination de biofilm les utilisant | |
| US20040214780A1 (en) | Peptide derivative, and pharmaceutically acceptable salt thereof, process for producing the same, and use thereof | |
| US8722719B2 (en) | Amine compound and use for same | |
| JP2008184406A (ja) | 医薬組成物 | |
| JP2003221353A (ja) | ポリエチレングリコールおよびその製造方法 | |
| US20070043008A1 (en) | Condensed Bicyclo[3.1.0] Hex-4-EN Derivatives Useful as Antibacterial Agents | |
| DK142846B (da) | Bakteriostatisk salt af 1,3-bis-(beta-ethylhexyl)-5-amino-5-methylhexahydropyrimidin til anvendelse i tand og mundplejemidler samt fremgangsmaade til fremstilling heraf |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application |
Ref document number: 09794039 Country of ref document: EP Kind code of ref document: A2 |
|
| ENP | Entry into the national phase |
Ref document number: 2730304 Country of ref document: CA |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 2009794039 Country of ref document: EP |
|
| NENP | Non-entry into the national phase |
Ref country code: DE |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 218/DELNP/2011 Country of ref document: IN |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 591021 Country of ref document: NZ |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 13003330 Country of ref document: US |