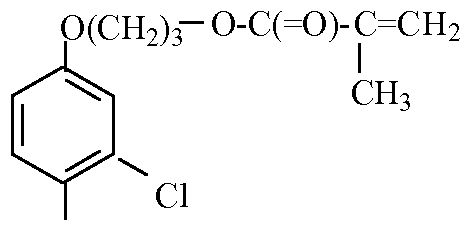WO2009153473A1 - Copolymère à blocs contenant un monomère photoactif porteur d'un groupement photoisomérisable, son utilisation dans une mémoire optique 3d - Google Patents
Copolymère à blocs contenant un monomère photoactif porteur d'un groupement photoisomérisable, son utilisation dans une mémoire optique 3d Download PDFInfo
- Publication number
- WO2009153473A1 WO2009153473A1 PCT/FR2009/050913 FR2009050913W WO2009153473A1 WO 2009153473 A1 WO2009153473 A1 WO 2009153473A1 FR 2009050913 W FR2009050913 W FR 2009050913W WO 2009153473 A1 WO2009153473 A1 WO 2009153473A1
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- block copolymer
- block
- copolymer according
- group
- monomer
- Prior art date
Links
Classifications
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08F—MACROMOLECULAR COMPOUNDS OBTAINED BY REACTIONS ONLY INVOLVING CARBON-TO-CARBON UNSATURATED BONDS
- C08F293/00—Macromolecular compounds obtained by polymerisation on to a macromolecule having groups capable of inducing the formation of new polymer chains bound exclusively at one or both ends of the starting macromolecule
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B82—NANOTECHNOLOGY
- B82Y—SPECIFIC USES OR APPLICATIONS OF NANOSTRUCTURES; MEASUREMENT OR ANALYSIS OF NANOSTRUCTURES; MANUFACTURE OR TREATMENT OF NANOSTRUCTURES
- B82Y10/00—Nanotechnology for information processing, storage or transmission, e.g. quantum computing or single electron logic
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08F—MACROMOLECULAR COMPOUNDS OBTAINED BY REACTIONS ONLY INVOLVING CARBON-TO-CARBON UNSATURATED BONDS
- C08F293/00—Macromolecular compounds obtained by polymerisation on to a macromolecule having groups capable of inducing the formation of new polymer chains bound exclusively at one or both ends of the starting macromolecule
- C08F293/005—Macromolecular compounds obtained by polymerisation on to a macromolecule having groups capable of inducing the formation of new polymer chains bound exclusively at one or both ends of the starting macromolecule using free radical "living" or "controlled" polymerisation, e.g. using a complexing agent
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08L—COMPOSITIONS OF MACROMOLECULAR COMPOUNDS
- C08L53/00—Compositions of block copolymers containing at least one sequence of a polymer obtained by reactions only involving carbon-to-carbon unsaturated bonds; Compositions of derivatives of such polymers
-
- G—PHYSICS
- G11—INFORMATION STORAGE
- G11B—INFORMATION STORAGE BASED ON RELATIVE MOVEMENT BETWEEN RECORD CARRIER AND TRANSDUCER
- G11B7/00—Recording or reproducing by optical means, e.g. recording using a thermal beam of optical radiation by modifying optical properties or the physical structure, reproducing using an optical beam at lower power by sensing optical properties; Record carriers therefor
- G11B7/24—Record carriers characterised by shape, structure or physical properties, or by the selection of the material
-
- G—PHYSICS
- G11—INFORMATION STORAGE
- G11B—INFORMATION STORAGE BASED ON RELATIVE MOVEMENT BETWEEN RECORD CARRIER AND TRANSDUCER
- G11B7/00—Recording or reproducing by optical means, e.g. recording using a thermal beam of optical radiation by modifying optical properties or the physical structure, reproducing using an optical beam at lower power by sensing optical properties; Record carriers therefor
- G11B7/24—Record carriers characterised by shape, structure or physical properties, or by the selection of the material
- G11B7/241—Record carriers characterised by shape, structure or physical properties, or by the selection of the material characterised by the selection of the material
- G11B7/242—Record carriers characterised by shape, structure or physical properties, or by the selection of the material characterised by the selection of the material of recording layers
- G11B7/244—Record carriers characterised by shape, structure or physical properties, or by the selection of the material characterised by the selection of the material of recording layers comprising organic materials only
- G11B7/245—Record carriers characterised by shape, structure or physical properties, or by the selection of the material characterised by the selection of the material of recording layers comprising organic materials only containing a polymeric component
Definitions
- the technology that is contemplated in the present invention is more particularly that of 3-D optical storage as described in international applications WO 01/73779 and WO 03/070689 and in Japanese Journal of Applied Physics. , Flight. 45, No. 28, 2006, pp. 1229-1234. It is based on the use of a photoisomerizable chromophore which is in two interconvertible thermodynamically stable isomeric forms under the effect of a light irradiation of appropriate wavelength. When no data has yet been recorded, one of the two forms has a majority. For data writing, this isomeric form is converted to the other by light irradiation having an appropriate wavelength. The conversion may result from a direct or indirect optical interaction (eg multiphoton).
- the present invention relates to a polymer for optical data storage in 3D. It also relates to the material obtained from this polymer as well as to the optical memory in 3D, in particular in disk form.
- the chromophores are attached to a polymer through the (co) polymerization of monomers carrying said chromophores.
- the application WO 2006/075327 also teaches the interest in increasing the concentration of chromophores so as to improve the recording sensitivity of the optical memory.
- concentration of monomers carrying chromophores increases, the mechanical properties of the polymer are affected and the material obtained is either too fragile or too soft to be manipulated easily. The need therefore exists to develop a rigid material that can be used in the field of 3D optical storage having good readability and data writing.
- US Patent 5023859 discloses an optical memory based on the use of a polymer carrying a photosensitive group of stilbene, spiropyran, azobenzene, bisazobenzene, trisazobenzene or azoxybenzene type.
- the polymer may be a block polymer but the exact nature of this block polymer is not specified.
- the polymer may be a polyalkylacrylate or a copolymer of the polyalkylacrylate, especially a copolymer with styrene. It may also be polymethyl methacrylate. It is not specified that it may be a block copolymer or that the chromophore is present in one of the blocks in particular.
- the invention relates to a block copolymer comprising:
- At least one soft block A having a T 9 between -55 ° C. and 0 ° C., preferably between -40 ° C. and -1 ° C.,
- At least one block B comprising at least one photoactive monomer carrying a photo-isomerizable chromophore.
- At least one soft block A or at least one block B means that the block copolymer can comprise one or more blocks A and one or more blocks B.
- the block B can comprise one or more photoactive monomers associated with another monomer.
- the block B may advantageously comprise a cooperative effect monomer.
- the photoactive monomer has the formula (I):
- X is H or CH 3 -;
- CR denotes a photo-isomerizable chromophore
- the block copolymer makes it possible to obtain a 3D optical memory.
- the invention also relates to the mixture comprising the block copolymer and a polymer which is a thermoplastic, a thermoplastic elastomer or a thermosetting and a 3D optical memory comprising the block copolymer or the polymer mixture.
- the invention also relates to the use of a block copolymer or a mixture of block copolymers as described above for performing optical data storage. - AT -
- T 9 denotes the glass transition temperature of a polymer, measured by DSC according to ASTM E1356.
- the T 9 of a monomer is also referred to as the T 9 of the homopolymer having a number-average molecular weight M n of at least 10,000 g / mol, obtained by radical polymerization of said monomer.
- M n number-average molecular weight
- ethyl acrylate has a T 9 of -24 ° C because the ethyl homopolyacrylate has a T 9 of -24 ° C. All percentages are by weight unless otherwise indicated.
- photoactive monomer is understood to mean a monomer carrying a photo-isomerizable CR chromophore group.
- the chromophore exists in two isomeric forms, for example cis / trans. The conversion from one form to another takes place under the action of a light irradiation of appropriate wavelength.
- the photoactive monomer has the formula (I):
- X is H or CH 3 -;
- R being H or a C 1 -C 10 alkyl group
- CR denotes a photoisomerizable chromophore
- the spacer group L has the function of improving the freedom of movement of the chromophore with respect to the copolymer chain so as to promote the conversion of the chromophore from one form to another. This improves the capacity and speed of reading.
- L is chosen such that G and CR are connected to each other by a sequence of 2 or more atoms which are linked together by covalent bonds.
- L can be chosen for example from groups (CRiR 2 ) m! O (CRIR 2) m, (OCRiR 2) m or (SCRiR 2) m wherein m is an integer greater than 2, preferably between 2 and 10 and R 1 and R 2 independently denote H, halogen or alkyl or aryl.
- R 1 and R 2 denote H.
- the chromophore CR is preferably of the diarylalkylene type existing in cis and trans isomeric forms. It may be one of the chromophores disclosed in WO 01/73779, WO 03/070689, WO 2006/075329 or WO 2006/075327.
- chromophore CR is chosen so that the energy barrier for isomerization is greater than 80 kJ / mol. Indeed, it is desirable that the isomerization is a very slow process at room temperature to avoid a loss of recorded data.
- the photoactive monomer has the formula (II):
- Ar 1 and Ar 2 denote aryl groups, optionally substituted with one or more substituents
- W 1 and W 2 are chosen from the groups H, -CN, -COOH, -COOR ', -OH, -SO 2 R', -NO 2 , R 'being a C 1 -C 10 alkyl or aryl group; .
- L is linked by covalent bonds to Ar 2 as well as to G.
- Ar 1 and Ar 2 denote aryl groups, substituted or unsubstituted. They are chosen for example independently of one another from phenyl, biphenyl, anthracene or phenanthrene groups. Substituent (s) are selected potential (s) from: H, C 1 -C 10 alkyl, NO 2, halogen or alkoxy, C 1 - C 10, NR "R"'with R “and R'” being H or C 1 -C 10 alkyl.
- Ar 1 is a phenyl or biphenyl group and Ar 2 is a phenyl or biphenyl group, each of the phenyl and / or biphenyl groups may be optionally substituted by one or more substituents, that is to say that the chromophore has for formula (V) or (VI):
- the optional substituents may be for example H, aryl, alkyl CRCI 0, NO 2, halogen or alkoxy CRCI 0.
- W 1 and W 2 are H or CN
- Ar 2 is phenyl or biphenyl
- Ar 1 is phenyl or biphenyl substituted para to R 5 O- or R 5 S-.
- R 5 denotes an alkyl or aryl group, substituted or unsubstituted.
- R 5 is a C 1 -C 4 alkyl group.
- R 5 may be, for example, a methyl, ethyl, propyl or butyl group.
- it may be the chromophore of formula (VII):
- W 1 and W 2 denote H or CN
- Ar 2 is a phenyl or biphenyl group
- Ar 1 is a biphenyl group substituted by R 5 O- or R 5 S-.
- it may be the chromophore of formula (VIII):
- MeAA or MeMMA are very particularly preferred:
- the trans isomer presents a greater fluorescence than cis; the trans isomer has a large biphotonic absorption cross section; the Stokes shift (“Stokes shift”) is greater than 100 nm (little overlap between the absorption and emission spectra with peaks at 375 and 485 nm respectively).
- Chromophores which have a low overlap, i.e. ⁇ 35% or even better ⁇ 20%, between the absorption and emission spectra are preferred (see, on this, page 22 of WO 2006/075327). . This makes it possible to increase the concentration of the chromophore and thus to promote the cooperative effect without impairing the quality of the signal during the reading.
- the overlap depends on both the Stokes displacement and the peak width.
- the overlap is defined as the absorbed emission percentage for a solution of the 0.01 M chromophore in a 1 cm passage vessel.
- the Stokes shift is> 100 nm.
- the measurement of Stokes displacement is well known to those skilled in the art: reference may be made in particular to the document DEKKER encyclopedia of nanoscience and nanotechnology by James A. Schwartz et al., Published: illustrated published by CRC Press, 2004, pages 4014 and following or encyclopedia of Optical Engineering: Las-Pho, pages 1025 and following, Ronald G. Driggers, illustrated edition published by CRC Press, 2003.
- This displacement is measured by comparing the emission spectra and absorption chromophore in a commercial spectrofluorometer. This displacement represents a physical property of a chromophore and is independent of the type of spectrofluorimeter used.
- the invention is not limited to the particular chromophores of the diarylalkylene type but can also be applied to other photoisomerizable chromophores, not including stilbene, spiropyran, azobenzene, bisazobenzene, trisazobenzene or azoxybenzene groups.
- a list of chromophores useful in the invention is found in the following documents US 5023859, US 6875833 and US 6641889.
- cooperative effect monomer means a compound of formula (VIII):
- Ar 3 denotes an aromatic group which may or may not be substituted by one or more substituents.
- This cooperative-effect monomer interacts with the chromophore and / or enhances the cooperative effect between the chromophores themselves, thereby improving the writing speed.
- An interpretation of the cooperative effect is that the monomer modifies the microenvironment of the chromophore and promotes photo-isomerization.
- the substituent for formula (VIII) is selected from: (i) halogens;
- Ar 3 is a phenyl group.
- the halogen group is chlorine.
- Ar 3 is chosen from the following groups:
- PEMA phenoxy ethyl methacrylate
- PPMA phenoxypropyl methacrylate
- PEA phenoxy ethyl acrylate
- Block A it can be "rigid” or “soft”.
- Block A is considered “rigid” when its glass transition temperature is above room temperature, 25 °.
- Block A is considered “soft” when its glass transition temperature is below 25 ° C.
- It has according to the invention a T 9 between -55 ° C and 0 ° C, preferably between -40 ° C and -1 ° C and is therefore soft.
- it also has a number average mass Mn> 1000 g / mol, advantageously> 5000 g / mol, preferably> 10000 g / mol.
- One of the functions of the soft block A is to obtain sufficient mechanical strength of the memory storage material.
- the soft block A is obtained from the polymerization of at least one vinyl, vinylidene, diene, olefinic, allylic or (meth) acrylic monomer such that the combination of monomers leads to a Tg of the block A ⁇ 20 ° C. and in particular at most equal to 0 ° C, for example between -30 ° C and -3 ° C.
- These monomers are chosen more particularly from vinylaromatic monomers such as styrene or substituted styrenes, especially alpha-methylstyrene, acrylic monomers such as acrylic acid or its salts, alkyl acrylates, cycloalkyl acrylates or aryl acrylates.
- hydroxyalkyl acrylates such as 2-hydroxyethyl acrylate
- ether alkyl acrylates such as acrylate methoxyethyl, alkoxy- or aryloxy-polyalkylene glycol acrylates such as methoxypolyethylene glycol acrylates, ethoxypolyethylene glycol acrylates, methoxypolypropylene glycol acrylates, methoxy-polyethylene glycol-polypropylene glycol acrylates or mixtures thereof
- aminoalkyl acrylates such as 2- (dimethylamino) ethyl acrylate (ADAME), fluorinated acrylates, silylated acrylates, phosphorus acrylates such as phosphorus acrylates alkylene glycol, methacrylic monomers such as methacrylic acid or its salts, alkyl, cycloalkyl,
- the soft block A is preferably obtained from styrene and / or (meth) acrylic monomer (s) and / or alkyl acrylate.
- the block A comprises, as the main monomer (s), styrene and / or MMA and / or butyl acrylate or ethyl hexyl acrylate.
- it comprises at least 50% of butyl acrylate or 2-ethylhexyl acrylate.
- Block A is intended to confer the mechanical properties of strength and / or rigidity of the finished material.
- the soft block A may contain, in addition to the above monomers, monomers (s) composing the block (s) B, especially photoactive monomer or cooperative-effect monomer.
- the block A may contain residues of the monomer (s) constituting the block B.
- the block (s) A may comprise or monomers (s) initially introduced (s) to prepare the block (s) B.
- the soft block A may comprise by weight from 40 to 100% styrene and / or acrylate butyl or 2 ethyl hexyl, from 0 to 30% of at least one comonomer chosen from the list defined above and from 1 to 30% of at least one photoactive monomer, the total being 100%.
- block B it comprises at least one photoactive monomer and optionally at least one other monomer copolymerizable with the photoactive monomer.
- the said other monomer may be chosen from the list of monomers defined above for block A. It may also be a monomer with a cooperative effect.
- the content by weight of photoactive monomer in block B may range from 5 to 100%.
- the monomer that is copolymerized with the photoactive monomer is a cooperative effect monomer. It is preferably TCLP, PEMA, TCLPa or PEA.
- Block B comprises, for example by weight, from 10 to 80% of at least one photoactive monomer, from 10 to 80% of at least one cooperative-effect monomer and optionally one or more other comonomers chosen from the previous list (the total doing 100%).
- the block B may contain monomer (s) making up the block (s) A.
- the block B may contain monomer residues constituting the block A.
- the block (es) B may comprise monomer (s) initially introduced (s) to prepare the block (s) A.
- the block B may comprise in weight of 40 to 100% of active monomer and / or monomer with a cooperative effect, from 0 to 60% of at least one monomer chosen from the list defined above for the synthesis of block A, the total being 100%.
- the block copolymer of the invention it comprises at least one soft block A and at least one block B comprising at least one photoactive monomer.
- a block copolymer consists of adjacent blocks that are constitutionally different, ie, blocks comprising units derived from different monomers or the same monomer, but according to a composition or a sequential distribution of different patterns.
- a block copolymer may for example be a diblock, triblock or star copolymer.
- the block copolymer is such that block (s) A and block (es) B are incompatible, that is to say that they have a Flory interaction parameter. - Huggins ⁇ AB > 0 at room temperature (parameter well known by those skilled in the art and described in particular in the chemistry and physico-chemistry of polymers, by M. Fontanille and Y. Gnanou, Dunod 2002). This results in a microseparation of phases with the formation of a two-phase structure on a macroscopic scale.
- the block copolymer is then nanostructured, that is to say that domains are formed whose size is less than 100 nm, preferably between 5 and 50 nm.
- Nanostructuring has the advantage of leading to a transparent material. In addition, this makes it possible to obtain domains concentrated in chromophores because there is no "dilution" by the block (s) A, which makes it possible to promote the cooperative effect between chromophores (with an increase in the writing speed).
- the block copolymer is preferably a triblock copolymer B-A-B 'comprising a central block A connected by covalent bonds to two lateral blocks B and B' (that is to say arranged on each side of the central block A).
- B and B ' may be the same or different (this type of copolymer is sometimes also noted B-b-A-b-B').
- It can also be a triblock copolymer ABA 'comprising a central block B connected by covalent bonds to two lateral blocks A and A (that is to say arranged on each side of the central block B) and which comprise chromophore patterns.
- a and A ' may be the same or different.
- more complex structures can be obtained, for example with a number of blocks greater than or equal to 2, for example 5 blocks, B "-A'-B'- AB, 6 blocks, ...
- the synthesized block copolymer can therefore consist of a single structure or a mixture of different structures, more or less complex.
- the mechanical and optical properties obtained can then vary widely depending on the block copolymer used in the 3D storage material.
- the structuring at the nanoscale obtained by the incompatibility of blocks A and B remains a property common to the various block copolymers which are the subject of the present invention.
- the blocks A and A comprise, as major monomer (s), styrene and / or MMA and / or alkyl acrylate;
- the blocks B and B ' comprise, by weight, from 10 to 60% of at least one photoactive monomer, from 10 to 60% of at least one cooperative-effect monomer and optionally a monomer from the preceding list of monomer mentioned for the block A (the total making 100%), which is preferably an alkyl (meth) acrylate, more particularly methyl methacrylate.
- the block copolymer may be used alone or in admixture with another polymer which has sufficient transparency in the wavelength range used for writing or reading as well as low birefringence. It can be a thermoplastic, a thermoplastic elastomer or a thermosetting. This characteristic is important for the 3D optical memory technology for which it is necessary for the light ray to reach each layer of the memory without being disturbed.
- a thermoplastic such as a homo- or copolymer of methyl methacrylate or styrene or a polycarbonate is preferably used.
- the mixture comprises, by weight, from 50 to 100%, advantageously from 75 to 100%, preferably from 90 to 100%, of the block copolymer, respectively from 0 to 50%, advantageously from 0 to 25%, preferably 5 to 10%.
- the block copolymer and / or the mixture of block copolymers may also optionally comprise various additives (antistatic, lubricant, colorant, plasticizer, antioxidant, anti-UV, etc.). Process for obtaining the block copolymer
- the block copolymer is obtained using polymerization techniques known to those skilled in the art.
- One of these polymerization techniques may be anionic polymerization as it is for example taught in the following documents FR 2762604, FR 2761997 and FR 2761995. It may also be the controlled radical polymerization technique which comprises several variants depending on the nature of the control agent that is used.
- SFRP Stable Free Radical Polymerization
- ATRP Atom Transfer Radical Polymerization
- RAFT Reversible Addition Fragmentation Transfer
- sulfur-containing products such as dithioesters, trithiocarbonates, xanthates or dithiocarbamates.
- Controlled radical polymerization with T nitroxide control is the preferred technique for obtaining the block copolymer of the invention. Indeed, this technique does not require working under conditions as severe as the anionic polymerization (that is to say, no moisture, temperature ⁇ 100 0 C). It also makes it possible to polymerize a wide range of monomers. It can be conducted under various conditions, for example by mass, solvent or dispersed medium such as suspension or emulsion in water.
- stable free radical denotes a radical that is so persistent and non-reactive with respect to air and moisture in the ambient air that it can be handled and stored for a much longer period than the majority free radicals (see Accounts of Chemical
- the stable free radical is thus distinguished from free radicals whose lifetime is ephemeral (from a few milliseconds to a few seconds) as the free radicals derived from the usual initiators of polymerization such as peroxides, hydroperoxides or azo initiators. Free radicals initiating polymerization tend to accelerate polymerization whereas stable free radicals generally tend to slow it down. It can be said that a free radical is stable within the meaning of the present invention if it is not a polymerization initiator and if, under the usual conditions of the invention, the average lifetime of the radical is at least one minute.
- the nitroxide T is represented by the structure (IX):
- R 9 CR 11 Rio (IX) wherein R 6, R7, Rs, R9, R I O and R 11 denote linear or branched alkyl groups C 1 -C 2 O, preferably C 1 -C 10 such as methyl, ethyl, propyl, butyl, isopropyl, isobutyl, tert-butyl, neopentyl, substituted or unsubstituted, C 6 -C 30 aryls substituted or unsubstituted by one or more substituents, such as benzyl, aryl (phenyl), cyclic saturated C 1 -C 30 and wherein the groups R 6 and R 9 may be part of an optionally substituted cyclic structure R 6 -CNC-R 6 which may be chosen from:
- x denoting an integer between 1 and 12.
- R 3 and R b denote identical or different alkyl groups having from 1 to 40 carbon atoms, optionally linked to each other so as to form a ring and optionally substituted with hydroxyl, alkoxy or amino groups,
- R L designating a monovalent group of molar mass greater than 16 g / mol, preferably greater than 30 g / mol.
- the group R L may for example have a molar mass of between 40 and 450 g / mol.
- Z 1 and Z 2 which may be the same or different, may be selected from alkyl, cycloalkyl, alkoxyl, aryloxyl, aryl, aralkyloxy, perfluoroalkyl, aralkyl and may include from 1 to 20 carbon atoms; Z 1 and / or Z 2 may also be a halogen atom such as a chlorine, bromine or fluorine atom.
- R L is a phosphonate group of formula: in which R c and R d are two identical or different alkyl groups, optionally linked so as to form a ring, comprising from 1 to 40 carbon atoms, optionally substituted or not by one or more substituents as described above.
- the stable nitroxide radical originates from a molecule which splits into two radicals, one nitroxide, regulator of the polymerization, the other initiator of the polymerization.
- a molecule capable of forming in situ the stable nitroxide radical under the effect of a rise in temperature mention may be made of the BLOCBUILDER ® manufactured and marketed by the Applicant.
- the R L group may also comprise at least one aromatic ring such as the phenyl radical or the naphthyl radical, substituted for example by one or more alkyl radicals comprising from 1 to 10 carbon atoms.
- nitroxides of formula (X) are preferred because they make it possible to obtain good control of the radical polymerization of (meth) acrylic monomers.
- Alkoxyamines of formula (XIII) are preferred:
- Z is a multivalent group and o is an integer of 1 to 10 inclusive.
- Z is a group capable of releasing several radical sites after thermal activation and rupture of the covalent ZT bond. Examples of groups Z are found on pages 15 to 18 of international application WO 2006/061523.
- Z is a divalent group, i.e. the integer o is 2.
- the central block is first prepared by polymerizing with the alkoxyamine the monomer mixture leading to the central block.
- the polymerization takes place with or without a solvent or in a dispersed medium.
- the mixture is heated to a temperature above the activation temperature of the alkoxyamine.
- the monomer (s) leading to the side blocks is added. It is possible that at the end of the preparation of the central block, there remain monomers not completely consumed that we can choose to eliminate or not before the preparation of the side blocks.
- the removal may for example consist of precipitating in a non-solvent, recovering and drying the central block. If one chooses not to remove the monomers not entirely consumed, they can polymerize with the monomers introduced to prepare the side blocks.
- the optical principles underlying the present invention are the same as those described in international applications WO 01/73779 and WO 03/070689.
- the writing is based on the conversion of one isomeric form to another under the effect of a light irradiation.
- the conversion requires having a chromophore in an excited state, which requires absorption at an energy level E.
- the absorption of two photons is facilitated by combining the energy of at least two photons of one or more photons.
- a plurality of light beams (x) having energy levels E 1 and E 2 which may be different from E.
- the two light beams are in the UV, visible or near-infrared range.
- only one light beam is used and the conversion is the result of a two-photon absorption process.
- the reading may be based on a linear or non-linear electronic excitation process.
- the emission spectra of the two isomers are different and the emission is collected using a suitable reading device.
- a non-linear process such as Raman scattering or a Four Wave Mixing Process (“Four Wave Mixing Process") can be employed.
- a small volume element of the 3D memory contains the chromophores in a major isomeric form or else under the other.
- the volume element therefore contains information stored in a well-defined and localized area of the memory and is characterized by an optical signal different from that of its immediate environment.
- the invention also relates to the 3D optical memory (or 3D optical storage unit) comprising the block copolymer or the mixture of block copolymers of the invention and which is used to record (store) the data.
- a 3D memory is a memory that stores data at any point (defined by three coordinates x, y and z) of the volume of the memory.
- a 3D memory allows storage of data in several virtual layers (or virtual levels). The volume of the 3D memory is therefore related to the physical volume occupied by it.
- the 3D memory can be obtained by injecting the block copolymer or the block polymer blend.
- This transformation technique is known to the plasturgists and consists in injecting under pressure the material in the molten state in a mold (in this connection, reference can be made to the Summary of plastics, Nathan, 4th edition, ISBN 2-12-355352-2, pp. 141 - 156). The material is melted and compressed using an extruder. It is also possible to superpose several layers comprising the block copolymer or the block copolymer mixture of the invention as taught in the international application WO 2006/075329.
- the 3D optical memory is in the form of a disk which makes it possible to set it in rotation, the writing or reading head being fixed.
- the disc may be obtained by injection or molding of the block copolymer or the block copolymer mixture if it has the appropriate mechanical characteristics. It can also be obtained by depositing the block copolymer or the copolymer mixture on a rigid and transparent support in the range of wavelengths used for writing and / or reading.
- the BLOCBUILDER ® corresponds to the product of formula:
- the butyl polyacrylate co styrene obtained is then withdrawn from the reactor.
- the Tg of the measured block A is -5 ° C.
- the reaction is carried out at 116 ° C. with stirring for 3 hours.
- the reaction product is then withdrawn. It corresponds to the expected triblock polymer.
- Step 1 synthesis of the soft block A
- the reaction is conducted at 1 18 0 C for 190 minutes.
- the resulting reaction product is treated to remove unreacted monomers.
- the butyl polyacrylate co styrene obtained is then withdrawn from the reactor.
- the Tg of the measured block A is -5 ° C.
- Step 2 synthesis of block B
- Block A 60 g of Block A are charged with 240 g of MeMMA, 240 g of PEMA and 880 g of toluene. The reaction is carried out at 116 ° C. with stirring for 3 hours.
- reaction product is then withdrawn. It corresponds to the expected triblock copolymer.
- Step 1 synthesis of the soft block A
- the butyl polyacrylate co styrene obtained is then withdrawn from the reactor.
- the Tg of the measured block A is -5 ° C.
- the reaction is carried out at 116 ° C. with stirring for 3 hours.
- the reaction product is then withdrawn. It corresponds to the expected diblock copolymer.
- the polymer solutions obtained in Examples 1 to 3 are precipitated in a large quantity of methanol at room temperature, filtered, washed and dried.
- the product obtained is then shaped by compression-molding at 150 0 C for 10 min to form a disc of 2 cm in diameter and 2 mm thick.
- the light transmission is greater than 80% over the entire visible range.
- This disk is then subjected to a static read-write test of data using a suitable laser device. There was a recording of the data on the disc.
Abstract
Description
Claims
Priority Applications (3)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| EP09766035A EP2291423A1 (fr) | 2008-05-27 | 2009-05-15 | Copolymère à blocs contenant un monomère photoactif porteur d'un groupement photoisomérisable, son utilisation dans une mémoire optique 3d |
| US12/995,025 US20110129636A1 (en) | 2008-05-27 | 2009-05-15 | Block Copolymer Containing a Photoactive Monomer Bearing a Photoisomerizable Group, Use Thereof in a 3D Optical Memory |
| JP2011511058A JP2011522917A (ja) | 2008-05-27 | 2009-05-15 | 光異性化可能な基を有する光活性モノマーを含むブロックコポリマーと、3d光メモリでのその使用 |
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| FR0853429A FR2931827A1 (fr) | 2008-05-27 | 2008-05-27 | Copolymere a blocs contenant un monomere photoactif porteur d'un groupement photoisomerisable, son utilisation dans une memoire optique 3d. |
| FR0853429 | 2008-05-27 |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| WO2009153473A1 true WO2009153473A1 (fr) | 2009-12-23 |
Family
ID=40104749
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/FR2009/050913 WO2009153473A1 (fr) | 2008-05-27 | 2009-05-15 | Copolymère à blocs contenant un monomère photoactif porteur d'un groupement photoisomérisable, son utilisation dans une mémoire optique 3d |
Country Status (6)
| Country | Link |
|---|---|
| US (1) | US20110129636A1 (fr) |
| EP (1) | EP2291423A1 (fr) |
| JP (1) | JP2011522917A (fr) |
| KR (1) | KR20110021975A (fr) |
| FR (1) | FR2931827A1 (fr) |
| WO (1) | WO2009153473A1 (fr) |
Cited By (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JP2014534272A (ja) * | 2011-08-25 | 2014-12-18 | ロリク アーゲーRolic Ag | 光反応性化合物 |
Citations (2)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2003070689A2 (fr) * | 2002-02-21 | 2003-08-28 | Mempile Inc. | Composes donateur-accepteur-donateur lies a un polymere et leur utilisation dans une memoire optique tridimensionnelle |
| FR2909093A1 (fr) * | 2006-11-28 | 2008-05-30 | Arkema France | Memoire optique 3d comprenant un copolymere a blocs contenant un monomere photoactif porteur d'un groupement photoisomerisable. |
Family Cites Families (9)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| DE3810722A1 (de) * | 1988-03-30 | 1989-10-12 | Roehm Gmbh | Vorrichtung zur reversiblen optischen datenspeicherung |
| US5763548A (en) * | 1995-03-31 | 1998-06-09 | Carnegie-Mellon University | (Co)polymers and a novel polymerization process based on atom (or group) transfer radical polymerization |
| US5789487A (en) * | 1996-07-10 | 1998-08-04 | Carnegie-Mellon University | Preparation of novel homo- and copolymers using atom transfer radical polymerization |
| DE19720288A1 (de) * | 1997-05-15 | 1998-11-19 | Bayer Ag | Homopolymere mit hoher photoinduzierbarer Doppelbrechung |
| IL135309A0 (en) * | 2000-03-28 | 2001-05-20 | Ortal Apert | Three-dimensional optical memory |
| CN1513176A (zh) * | 2001-03-28 | 2004-07-14 | 拜尔公司 | 在信息层中含有三氮菁染料作为吸光性化合物的光学数据存储介质 |
| US20090283727A1 (en) * | 2005-01-12 | 2009-11-19 | Mempile Inc. C/O Phs Corporate Services, Inc. | Disks for data storage |
| WO2006075328A1 (fr) * | 2005-01-12 | 2006-07-20 | Mempile Inc. | Composes presentant un pouvoir d'absorption biphotonique ameliore pour des applications non lineaires |
| EP1842184A2 (fr) * | 2005-01-12 | 2007-10-10 | Mempile Inc. | Fabrication de multiplaques pour memoire optique amelioree |
-
2008
- 2008-05-27 FR FR0853429A patent/FR2931827A1/fr active Pending
-
2009
- 2009-05-15 US US12/995,025 patent/US20110129636A1/en not_active Abandoned
- 2009-05-15 EP EP09766035A patent/EP2291423A1/fr not_active Withdrawn
- 2009-05-15 KR KR1020107029264A patent/KR20110021975A/ko not_active Application Discontinuation
- 2009-05-15 JP JP2011511058A patent/JP2011522917A/ja not_active Abandoned
- 2009-05-15 WO PCT/FR2009/050913 patent/WO2009153473A1/fr active Application Filing
Patent Citations (2)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2003070689A2 (fr) * | 2002-02-21 | 2003-08-28 | Mempile Inc. | Composes donateur-accepteur-donateur lies a un polymere et leur utilisation dans une memoire optique tridimensionnelle |
| FR2909093A1 (fr) * | 2006-11-28 | 2008-05-30 | Arkema France | Memoire optique 3d comprenant un copolymere a blocs contenant un monomere photoactif porteur d'un groupement photoisomerisable. |
Cited By (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JP2014534272A (ja) * | 2011-08-25 | 2014-12-18 | ロリク アーゲーRolic Ag | 光反応性化合物 |
Also Published As
| Publication number | Publication date |
|---|---|
| US20110129636A1 (en) | 2011-06-02 |
| FR2931827A1 (fr) | 2009-12-04 |
| JP2011522917A (ja) | 2011-08-04 |
| EP2291423A1 (fr) | 2011-03-09 |
| KR20110021975A (ko) | 2011-03-04 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| Schumers et al. | Light‐responsive block copolymers | |
| WO2008065301A1 (fr) | Memoire optique 3d comprenant un copolymere a blocs contenant un monomere photoactif porteur d'un groupement photoisomerisable | |
| WO2006106277A2 (fr) | Copolymere sequence a fonctions acides modulable et composition adhesive et thermoplastique le contenant | |
| EP2655031A1 (fr) | Plaques coulées nanostructurées réticulées | |
| US7018684B2 (en) | Block copolymers for optical data storage | |
| FR3040171A1 (fr) | Composition comprenant un reseau de polymeres reticules, prepare par copolymerisation radicalaire, comprenant des liaisons pendantes et des points de reticulation echangeables par reaction d'echange aldehyde-imine et/ou par reaction d'echange imine-imine, procede de preparation et utilisation | |
| FR2805268A1 (fr) | Polymeres thermoreversibles a fonctions mitroxyde | |
| FR2608615A1 (fr) | Composition d'un melange de polymeres, appropriee comme materiau optique | |
| EP2097480B1 (fr) | Memoire optique 3d comprenant des particules multicouches comprenant un monomere photoactif porteur d'un groupement photoisomerisable | |
| WO2009153473A1 (fr) | Copolymère à blocs contenant un monomère photoactif porteur d'un groupement photoisomérisable, son utilisation dans une mémoire optique 3d | |
| CA2590548A1 (fr) | Plaques coulees acryliques renforcees a l'impact | |
| WO2009037412A1 (fr) | Composition renfermant un homopolymere d'acide lactique et un copolymere a blocs | |
| WO2003037983A2 (fr) | Materiau polymere thermoplastique et disques moules | |
| EP3233933B1 (fr) | Polymerisation radicalaire d'alcoxyamines a basse temperature | |
| FR2920431A1 (fr) | Procede de preparation d'un materiau transparent de type alliage de polymere thermodurcissable/polymere thermoplastique et son application dans l'optique pour la fabrication de verres organiques. | |
| EP3233932B1 (fr) | Polymérisation radicalaire d'alcoxyamines à basse température | |
| FR2809726A1 (fr) | Monomeres monofonctionnels, compositions polymerisables les renfermant, resines et articles ophtalmiques obtenus a partir de ceux-ci | |
| CA3032226A1 (fr) | Polyester thermoplastique amorphe pour la fabrication d'articles optiques | |
| EP2030998A1 (fr) | Procédé de préparation d'un article moulé transparent à base d'un alliage de polymer thermoplastique et de polymère thermodurcissable | |
| WO2021105637A1 (fr) | Microgels et leurs applications photoniques et interferentielles stimulables | |
| FR3041972A1 (fr) | Adhesif comprenant des copolymeres a blocs, pour l'encapsulation de dispositifs electroniques flexibles, ameliorant la protection contre la permeabilite aux gaz | |
| WO2007135342A1 (fr) | Procede de coextrusion multicouche utilisant une couche ductile intermediaire | |
| FR2833962A1 (fr) | Disques moules pour supports d'enregistrement d'informations audio et/ou optiques en copolymere methacrylique a resistance amelioree au stockage | |
| FR2866342A1 (fr) | Disques moules pour supports d'enregistrement d'informations a base de copolymeres blocs nanostructures | |
| WO2010097561A2 (fr) | Nouveaux copolymères amphiphiles comprenant un groupe colorant, leur procédé de préparation et leurs utilisations comme pigments |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application |
Ref document number: 09766035 Country of ref document: EP Kind code of ref document: A1 |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 2011511058 Country of ref document: JP |
|
| NENP | Non-entry into the national phase |
Ref country code: DE |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 2009766035 Country of ref document: EP |
|
| ENP | Entry into the national phase |
Ref document number: 20107029264 Country of ref document: KR Kind code of ref document: A |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 12995025 Country of ref document: US |