WO2008032980A1 - Novel stigmasterol derivative or pharmaceutically acceptable salt thereof, method of producing the same, and composition containing the same to inhibit obesity or to prevent and treat hyperlipidemia - Google Patents
Novel stigmasterol derivative or pharmaceutically acceptable salt thereof, method of producing the same, and composition containing the same to inhibit obesity or to prevent and treat hyperlipidemia Download PDFInfo
- Publication number
- WO2008032980A1 WO2008032980A1 PCT/KR2007/004395 KR2007004395W WO2008032980A1 WO 2008032980 A1 WO2008032980 A1 WO 2008032980A1 KR 2007004395 W KR2007004395 W KR 2007004395W WO 2008032980 A1 WO2008032980 A1 WO 2008032980A1
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- compound
- formula
- obesity
- cholesterol
- stigmasterol
- Prior art date
Links
- 208000008589 Obesity Diseases 0.000 title claims abstract description 46
- 235000020824 obesity Nutrition 0.000 title claims abstract description 46
- HCXVJBMSMIARIN-PHZDYDNGSA-N stigmasterol Chemical class C1C=C2C[C@@H](O)CC[C@]2(C)[C@@H]2[C@@H]1[C@@H]1CC[C@H]([C@H](C)/C=C/[C@@H](CC)C(C)C)[C@@]1(C)CC2 HCXVJBMSMIARIN-PHZDYDNGSA-N 0.000 title claims abstract description 38
- 239000000203 mixture Substances 0.000 title claims abstract description 30
- 150000003839 salts Chemical class 0.000 title claims abstract description 28
- 238000000034 method Methods 0.000 title claims abstract description 20
- 208000031226 Hyperlipidaemia Diseases 0.000 title claims abstract description 16
- 150000001875 compounds Chemical class 0.000 claims description 60
- OILXMJHPFNGGTO-UHFFFAOYSA-N (22E)-(24xi)-24-methylcholesta-5,22-dien-3beta-ol Natural products C1C=C2CC(O)CCC2(C)C2C1C1CCC(C(C)C=CC(C)C(C)C)C1(C)CC2 OILXMJHPFNGGTO-UHFFFAOYSA-N 0.000 claims description 13
- OQMZNAMGEHIHNN-UHFFFAOYSA-N 7-Dehydrostigmasterol Natural products C1C(O)CCC2(C)C(CCC3(C(C(C)C=CC(CC)C(C)C)CCC33)C)C3=CC=C21 OQMZNAMGEHIHNN-UHFFFAOYSA-N 0.000 claims description 13
- HZYXFRGVBOPPNZ-UHFFFAOYSA-N UNPD88870 Natural products C1C=C2CC(O)CCC2(C)C2C1C1CCC(C(C)=CCC(CC)C(C)C)C1(C)CC2 HZYXFRGVBOPPNZ-UHFFFAOYSA-N 0.000 claims description 13
- LGJMUZUPVCAVPU-UHFFFAOYSA-N beta-Sitostanol Natural products C1CC2CC(O)CCC2(C)C2C1C1CCC(C(C)CCC(CC)C(C)C)C1(C)CC2 LGJMUZUPVCAVPU-UHFFFAOYSA-N 0.000 claims description 13
- 229940032091 stigmasterol Drugs 0.000 claims description 13
- BFDNMXAIBMJLBB-UHFFFAOYSA-N stigmasterol Natural products CCC(C=CC(C)C1CCCC2C3CC=C4CC(O)CCC4(C)C3CCC12C)C(C)C BFDNMXAIBMJLBB-UHFFFAOYSA-N 0.000 claims description 13
- 235000016831 stigmasterol Nutrition 0.000 claims description 13
- 235000013305 food Nutrition 0.000 claims description 11
- 238000006243 chemical reaction Methods 0.000 claims description 10
- 239000007864 aqueous solution Substances 0.000 claims description 6
- KDLHZDBZIXYQEI-UHFFFAOYSA-N palladium Substances [Pd] KDLHZDBZIXYQEI-UHFFFAOYSA-N 0.000 claims description 6
- VHUUQVKOLVNVRT-UHFFFAOYSA-N Ammonium hydroxide Chemical compound [NH4+].[OH-] VHUUQVKOLVNVRT-UHFFFAOYSA-N 0.000 claims description 5
- NXJCBFBQEVOTOW-UHFFFAOYSA-L palladium(2+);dihydroxide Chemical compound O[Pd]O NXJCBFBQEVOTOW-UHFFFAOYSA-L 0.000 claims description 4
- 239000008194 pharmaceutical composition Substances 0.000 claims 1
- HVYWMOMLDIMFJA-DPAQBDIFSA-N cholesterol Chemical compound C1C=C2C[C@@H](O)CC[C@]2(C)[C@@H]2[C@@H]1[C@@H]1CC[C@H]([C@H](C)CCCC(C)C)[C@@]1(C)CC2 HVYWMOMLDIMFJA-DPAQBDIFSA-N 0.000 abstract description 44
- 210000004369 blood Anatomy 0.000 abstract description 26
- 239000008280 blood Substances 0.000 abstract description 26
- 235000012000 cholesterol Nutrition 0.000 abstract description 22
- 239000003814 drug Substances 0.000 abstract description 16
- 230000037396 body weight Effects 0.000 abstract description 13
- 108010023302 HDL Cholesterol Proteins 0.000 abstract description 12
- 238000008214 LDL Cholesterol Methods 0.000 abstract description 11
- 108010028554 LDL Cholesterol Proteins 0.000 abstract description 10
- UFTFJSFQGQCHQW-UHFFFAOYSA-N triformin Chemical compound O=COCC(OC=O)COC=O UFTFJSFQGQCHQW-UHFFFAOYSA-N 0.000 abstract description 10
- 229940079593 drug Drugs 0.000 abstract description 8
- 235000013402 health food Nutrition 0.000 abstract description 5
- 230000002401 inhibitory effect Effects 0.000 abstract description 4
- 235000002639 sodium chloride Nutrition 0.000 description 28
- 238000004519 manufacturing process Methods 0.000 description 26
- MSWZFWKMSRAUBD-UHFFFAOYSA-N beta-D-galactosamine Natural products NC1C(O)OC(CO)C(O)C1O MSWZFWKMSRAUBD-UHFFFAOYSA-N 0.000 description 18
- MSWZFWKMSRAUBD-IVMDWMLBSA-N 2-amino-2-deoxy-D-glucopyranose Chemical compound N[C@H]1C(O)O[C@H](CO)[C@@H](O)[C@@H]1O MSWZFWKMSRAUBD-IVMDWMLBSA-N 0.000 description 16
- 229960002442 glucosamine Drugs 0.000 description 16
- XLYOFNOQVPJJNP-UHFFFAOYSA-N water Substances O XLYOFNOQVPJJNP-UHFFFAOYSA-N 0.000 description 16
- OKKJLVBELUTLKV-UHFFFAOYSA-N Methanol Chemical compound OC OKKJLVBELUTLKV-UHFFFAOYSA-N 0.000 description 15
- 230000002829 reductive effect Effects 0.000 description 13
- QTBSBXVTEAMEQO-UHFFFAOYSA-N Acetic acid Chemical compound CC(O)=O QTBSBXVTEAMEQO-UHFFFAOYSA-N 0.000 description 12
- YMWUJEATGCHHMB-UHFFFAOYSA-N Dichloromethane Chemical compound ClCCl YMWUJEATGCHHMB-UHFFFAOYSA-N 0.000 description 12
- 241001465754 Metazoa Species 0.000 description 10
- 208000037265 diseases, disorders, signs and symptoms Diseases 0.000 description 10
- 238000011282 treatment Methods 0.000 description 10
- 235000013361 beverage Nutrition 0.000 description 9
- 239000003960 organic solvent Substances 0.000 description 9
- LFQSCWFLJHTTHZ-UHFFFAOYSA-N Ethanol Chemical compound CCO LFQSCWFLJHTTHZ-UHFFFAOYSA-N 0.000 description 8
- 241000699670 Mus sp. Species 0.000 description 8
- 201000010099 disease Diseases 0.000 description 8
- 239000000243 solution Substances 0.000 description 8
- 241000700159 Rattus Species 0.000 description 7
- 239000002253 acid Substances 0.000 description 7
- 238000002360 preparation method Methods 0.000 description 7
- LCGLNKUTAGEVQW-UHFFFAOYSA-N Dimethyl ether Chemical compound COC LCGLNKUTAGEVQW-UHFFFAOYSA-N 0.000 description 6
- 108090001030 Lipoproteins Proteins 0.000 description 6
- 102000004895 Lipoproteins Human genes 0.000 description 6
- -1 alkali metal salt Chemical class 0.000 description 6
- KRKNYBCHXYNGOX-UHFFFAOYSA-N citric acid Chemical compound OC(=O)CC(O)(C(O)=O)CC(O)=O KRKNYBCHXYNGOX-UHFFFAOYSA-N 0.000 description 6
- 239000003925 fat Substances 0.000 description 6
- 235000019197 fats Nutrition 0.000 description 6
- PEDCQBHIVMGVHV-UHFFFAOYSA-N Glycerine Chemical compound OCC(O)CO PEDCQBHIVMGVHV-UHFFFAOYSA-N 0.000 description 5
- VEXZGXHMUGYJMC-UHFFFAOYSA-N Hydrochloric acid Chemical compound Cl VEXZGXHMUGYJMC-UHFFFAOYSA-N 0.000 description 5
- 210000001789 adipocyte Anatomy 0.000 description 5
- 230000000694 effects Effects 0.000 description 5
- 230000036541 health Effects 0.000 description 5
- 235000009200 high fat diet Nutrition 0.000 description 5
- 239000000463 material Substances 0.000 description 5
- 239000013642 negative control Substances 0.000 description 5
- 239000000126 substance Substances 0.000 description 5
- 238000002560 therapeutic procedure Methods 0.000 description 5
- CIWBSHSKHKDKBQ-JLAZNSOCSA-N Ascorbic acid Chemical compound OC[C@H](O)[C@H]1OC(=O)C(O)=C1O CIWBSHSKHKDKBQ-JLAZNSOCSA-N 0.000 description 4
- AFVFQIVMOAPDHO-UHFFFAOYSA-N Methanesulfonic acid Chemical compound CS(O)(=O)=O AFVFQIVMOAPDHO-UHFFFAOYSA-N 0.000 description 4
- 240000003768 Solanum lycopersicum Species 0.000 description 4
- 239000004480 active ingredient Substances 0.000 description 4
- 239000002775 capsule Substances 0.000 description 4
- HGCIXCUEYOPUTN-UHFFFAOYSA-N cyclohexene Chemical compound C1CCC=CC1 HGCIXCUEYOPUTN-UHFFFAOYSA-N 0.000 description 4
- 235000005911 diet Nutrition 0.000 description 4
- 230000037213 diet Effects 0.000 description 4
- 238000009207 exercise therapy Methods 0.000 description 4
- 235000015203 fruit juice Nutrition 0.000 description 4
- HQKMJHAJHXVSDF-UHFFFAOYSA-L magnesium stearate Chemical compound [Mg+2].CCCCCCCCCCCCCCCCCC([O-])=O.CCCCCCCCCCCCCCCCCC([O-])=O HQKMJHAJHXVSDF-UHFFFAOYSA-L 0.000 description 4
- 239000013641 positive control Substances 0.000 description 4
- 235000014347 soups Nutrition 0.000 description 4
- JOXIMZWYDAKGHI-UHFFFAOYSA-N toluene-4-sulfonic acid Chemical compound CC1=CC=C(S(O)(=O)=O)C=C1 JOXIMZWYDAKGHI-UHFFFAOYSA-N 0.000 description 4
- 238000005160 1H NMR spectroscopy Methods 0.000 description 3
- CSCPPACGZOOCGX-UHFFFAOYSA-N Acetone Chemical compound CC(C)=O CSCPPACGZOOCGX-UHFFFAOYSA-N 0.000 description 3
- WEVYAHXRMPXWCK-UHFFFAOYSA-N Acetonitrile Chemical compound CC#N WEVYAHXRMPXWCK-UHFFFAOYSA-N 0.000 description 3
- 206010003210 Arteriosclerosis Diseases 0.000 description 3
- FEWJPZIEWOKRBE-JCYAYHJZSA-N Dextrotartaric acid Chemical compound OC(=O)[C@H](O)[C@@H](O)C(O)=O FEWJPZIEWOKRBE-JCYAYHJZSA-N 0.000 description 3
- XEKOWRVHYACXOJ-UHFFFAOYSA-N Ethyl acetate Chemical compound CCOC(C)=O XEKOWRVHYACXOJ-UHFFFAOYSA-N 0.000 description 3
- 102000016267 Leptin Human genes 0.000 description 3
- 108010092277 Leptin Proteins 0.000 description 3
- 235000007688 Lycopersicon esculentum Nutrition 0.000 description 3
- ZMXDDKWLCZADIW-UHFFFAOYSA-N N,N-Dimethylformamide Chemical compound CN(C)C=O ZMXDDKWLCZADIW-UHFFFAOYSA-N 0.000 description 3
- MUBZPKHOEPUJKR-UHFFFAOYSA-N Oxalic acid Chemical compound OC(=O)C(O)=O MUBZPKHOEPUJKR-UHFFFAOYSA-N 0.000 description 3
- KWYUFKZDYYNOTN-UHFFFAOYSA-M Potassium hydroxide Chemical compound [OH-].[K+] KWYUFKZDYYNOTN-UHFFFAOYSA-M 0.000 description 3
- FAPWRFPIFSIZLT-UHFFFAOYSA-M Sodium chloride Chemical compound [Na+].[Cl-] FAPWRFPIFSIZLT-UHFFFAOYSA-M 0.000 description 3
- HEMHJVSKTPXQMS-UHFFFAOYSA-M Sodium hydroxide Chemical compound [OH-].[Na+] HEMHJVSKTPXQMS-UHFFFAOYSA-M 0.000 description 3
- 239000000654 additive Substances 0.000 description 3
- 208000011775 arteriosclerosis disease Diseases 0.000 description 3
- 238000007681 bariatric surgery Methods 0.000 description 3
- 235000015278 beef Nutrition 0.000 description 3
- 230000015572 biosynthetic process Effects 0.000 description 3
- 150000001720 carbohydrates Chemical class 0.000 description 3
- 235000014633 carbohydrates Nutrition 0.000 description 3
- 235000014171 carbonated beverage Nutrition 0.000 description 3
- 210000000845 cartilage Anatomy 0.000 description 3
- 230000008859 change Effects 0.000 description 3
- 239000003795 chemical substances by application Substances 0.000 description 3
- 229940125904 compound 1 Drugs 0.000 description 3
- 238000007796 conventional method Methods 0.000 description 3
- 235000013365 dairy product Nutrition 0.000 description 3
- 239000012153 distilled water Substances 0.000 description 3
- 235000006694 eating habits Nutrition 0.000 description 3
- 235000013882 gravy Nutrition 0.000 description 3
- 208000019622 heart disease Diseases 0.000 description 3
- 229940088597 hormone Drugs 0.000 description 3
- 239000005556 hormone Substances 0.000 description 3
- 235000008960 ketchup Nutrition 0.000 description 3
- NRYBAZVQPHGZNS-ZSOCWYAHSA-N leptin Chemical compound O=C([C@H](CO)NC(=O)[C@H](CC(C)C)NC(=O)[C@H](CC(O)=O)NC(=O)[C@H](CC(C)C)NC(=O)[C@H](CCC(N)=O)NC(=O)[C@H](CC=1C2=CC=CC=C2NC=1)NC(=O)[C@H](CC(C)C)NC(=O)[C@@H](NC(=O)[C@H](CC(O)=O)NC(=O)[C@H](CCC(N)=O)NC(=O)[C@H](CC(C)C)NC(=O)[C@H](CO)NC(=O)CNC(=O)[C@H](CCC(N)=O)NC(=O)[C@@H](N)CC(C)C)CCSC)N1CCC[C@H]1C(=O)NCC(=O)N[C@@H](CS)C(O)=O NRYBAZVQPHGZNS-ZSOCWYAHSA-N 0.000 description 3
- 229940039781 leptin Drugs 0.000 description 3
- 230000008018 melting Effects 0.000 description 3
- 238000002844 melting Methods 0.000 description 3
- 230000004060 metabolic process Effects 0.000 description 3
- 230000037323 metabolic rate Effects 0.000 description 3
- 235000013336 milk Nutrition 0.000 description 3
- 239000008267 milk Substances 0.000 description 3
- 210000004080 milk Anatomy 0.000 description 3
- VLKZOEOYAKHREP-UHFFFAOYSA-N n-Hexane Chemical compound CCCCCC VLKZOEOYAKHREP-UHFFFAOYSA-N 0.000 description 3
- 235000012149 noodles Nutrition 0.000 description 3
- 150000007524 organic acids Chemical class 0.000 description 3
- 235000015067 sauces Nutrition 0.000 description 3
- GQHTUMJGOHRCHB-UHFFFAOYSA-N 2,3,4,6,7,8,9,10-octahydropyrimido[1,2-a]azepine Chemical compound C1CCCCN2CCCN=C21 GQHTUMJGOHRCHB-UHFFFAOYSA-N 0.000 description 2
- QTBSBXVTEAMEQO-UHFFFAOYSA-M Acetate Chemical compound CC([O-])=O QTBSBXVTEAMEQO-UHFFFAOYSA-M 0.000 description 2
- CURLTUGMZLYLDI-UHFFFAOYSA-N Carbon dioxide Chemical compound O=C=O CURLTUGMZLYLDI-UHFFFAOYSA-N 0.000 description 2
- 208000020446 Cardiac disease Diseases 0.000 description 2
- 229920002101 Chitin Polymers 0.000 description 2
- 229920002261 Corn starch Polymers 0.000 description 2
- RGHNJXZEOKUKBD-SQOUGZDYSA-N D-gluconic acid Chemical compound OC[C@@H](O)[C@@H](O)[C@H](O)[C@@H](O)C(O)=O RGHNJXZEOKUKBD-SQOUGZDYSA-N 0.000 description 2
- IAZDPXIOMUYVGZ-UHFFFAOYSA-N Dimethylsulphoxide Chemical compound CS(C)=O IAZDPXIOMUYVGZ-UHFFFAOYSA-N 0.000 description 2
- 239000004278 EU approved seasoning Substances 0.000 description 2
- 229930091371 Fructose Natural products 0.000 description 2
- 239000005715 Fructose Substances 0.000 description 2
- RFSUNEUAIZKAJO-ARQDHWQXSA-N Fructose Chemical compound OC[C@H]1O[C@](O)(CO)[C@@H](O)[C@@H]1O RFSUNEUAIZKAJO-ARQDHWQXSA-N 0.000 description 2
- VZCYOOQTPOCHFL-OWOJBTEDSA-N Fumaric acid Chemical compound OC(=O)\C=C\C(O)=O VZCYOOQTPOCHFL-OWOJBTEDSA-N 0.000 description 2
- IAJILQKETJEXLJ-UHFFFAOYSA-N Galacturonsaeure Natural products O=CC(O)C(O)C(O)C(O)C(O)=O IAJILQKETJEXLJ-UHFFFAOYSA-N 0.000 description 2
- WQZGKKKJIJFFOK-GASJEMHNSA-N Glucose Chemical compound OC[C@H]1OC(O)[C@H](O)[C@@H](O)[C@@H]1O WQZGKKKJIJFFOK-GASJEMHNSA-N 0.000 description 2
- 108010010234 HDL Lipoproteins Proteins 0.000 description 2
- 102000015779 HDL Lipoproteins Human genes 0.000 description 2
- 206010020710 Hyperphagia Diseases 0.000 description 2
- 206010020772 Hypertension Diseases 0.000 description 2
- 108010007622 LDL Lipoproteins Proteins 0.000 description 2
- 102000007330 LDL Lipoproteins Human genes 0.000 description 2
- 206010028980 Neoplasm Diseases 0.000 description 2
- NBIIXXVUZAFLBC-UHFFFAOYSA-N Phosphoric acid Chemical compound OP(O)(O)=O NBIIXXVUZAFLBC-UHFFFAOYSA-N 0.000 description 2
- JUJWROOIHBZHMG-UHFFFAOYSA-N Pyridine Chemical compound C1=CC=NC=C1 JUJWROOIHBZHMG-UHFFFAOYSA-N 0.000 description 2
- CDBYLPFSWZWCQE-UHFFFAOYSA-L Sodium Carbonate Chemical compound [Na+].[Na+].[O-]C([O-])=O CDBYLPFSWZWCQE-UHFFFAOYSA-L 0.000 description 2
- QAOWNCQODCNURD-UHFFFAOYSA-N Sulfuric acid Chemical compound OS(O)(=O)=O QAOWNCQODCNURD-UHFFFAOYSA-N 0.000 description 2
- FEWJPZIEWOKRBE-UHFFFAOYSA-N Tartaric acid Natural products [H+].[H+].[O-]C(=O)C(O)C(O)C([O-])=O FEWJPZIEWOKRBE-UHFFFAOYSA-N 0.000 description 2
- WYURNTSHIVDZCO-UHFFFAOYSA-N Tetrahydrofuran Chemical compound C1CCOC1 WYURNTSHIVDZCO-UHFFFAOYSA-N 0.000 description 2
- DTQVDTLACAAQTR-UHFFFAOYSA-N Trifluoroacetic acid Chemical compound OC(=O)C(F)(F)F DTQVDTLACAAQTR-UHFFFAOYSA-N 0.000 description 2
- 241000209140 Triticum Species 0.000 description 2
- 235000021307 Triticum Nutrition 0.000 description 2
- 150000007513 acids Chemical class 0.000 description 2
- 231100000215 acute (single dose) toxicity testing Toxicity 0.000 description 2
- 238000011047 acute toxicity test Methods 0.000 description 2
- 229910052783 alkali metal Inorganic materials 0.000 description 2
- 229910052784 alkaline earth metal Inorganic materials 0.000 description 2
- 230000003627 anti-cholesterol Effects 0.000 description 2
- 230000003579 anti-obesity Effects 0.000 description 2
- 239000002585 base Substances 0.000 description 2
- 230000003542 behavioural effect Effects 0.000 description 2
- WPYMKLBDIGXBTP-UHFFFAOYSA-N benzoic acid Chemical compound OC(=O)C1=CC=CC=C1 WPYMKLBDIGXBTP-UHFFFAOYSA-N 0.000 description 2
- 230000036765 blood level Effects 0.000 description 2
- 235000008429 bread Nutrition 0.000 description 2
- 238000009395 breeding Methods 0.000 description 2
- 230000001488 breeding effect Effects 0.000 description 2
- 159000000007 calcium salts Chemical class 0.000 description 2
- 201000011510 cancer Diseases 0.000 description 2
- 239000012141 concentrate Substances 0.000 description 2
- 235000008504 concentrate Nutrition 0.000 description 2
- 235000014510 cooky Nutrition 0.000 description 2
- 239000008120 corn starch Substances 0.000 description 2
- 230000007812 deficiency Effects 0.000 description 2
- XBDQKXXYIPTUBI-UHFFFAOYSA-N dimethylselenoniopropionate Natural products CCC(O)=O XBDQKXXYIPTUBI-UHFFFAOYSA-N 0.000 description 2
- 208000035475 disorder Diseases 0.000 description 2
- 239000003937 drug carrier Substances 0.000 description 2
- 239000000796 flavoring agent Substances 0.000 description 2
- 235000013312 flour Nutrition 0.000 description 2
- 230000037406 food intake Effects 0.000 description 2
- 235000011194 food seasoning agent Nutrition 0.000 description 2
- 235000011187 glycerol Nutrition 0.000 description 2
- 229940093915 gynecological organic acid Drugs 0.000 description 2
- 235000015243 ice cream Nutrition 0.000 description 2
- 239000004615 ingredient Substances 0.000 description 2
- 239000003112 inhibitor Substances 0.000 description 2
- NOESYZHRGYRDHS-UHFFFAOYSA-N insulin Chemical compound N1C(=O)C(NC(=O)C(CCC(N)=O)NC(=O)C(CCC(O)=O)NC(=O)C(C(C)C)NC(=O)C(NC(=O)CN)C(C)CC)CSSCC(C(NC(CO)C(=O)NC(CC(C)C)C(=O)NC(CC=2C=CC(O)=CC=2)C(=O)NC(CCC(N)=O)C(=O)NC(CC(C)C)C(=O)NC(CCC(O)=O)C(=O)NC(CC(N)=O)C(=O)NC(CC=2C=CC(O)=CC=2)C(=O)NC(CSSCC(NC(=O)C(C(C)C)NC(=O)C(CC(C)C)NC(=O)C(CC=2C=CC(O)=CC=2)NC(=O)C(CC(C)C)NC(=O)C(C)NC(=O)C(CCC(O)=O)NC(=O)C(C(C)C)NC(=O)C(CC(C)C)NC(=O)C(CC=2NC=NC=2)NC(=O)C(CO)NC(=O)CNC2=O)C(=O)NCC(=O)NC(CCC(O)=O)C(=O)NC(CCCNC(N)=N)C(=O)NCC(=O)NC(CC=3C=CC=CC=3)C(=O)NC(CC=3C=CC=CC=3)C(=O)NC(CC=3C=CC(O)=CC=3)C(=O)NC(C(C)O)C(=O)N3C(CCC3)C(=O)NC(CCCCN)C(=O)NC(C)C(O)=O)C(=O)NC(CC(N)=O)C(O)=O)=O)NC(=O)C(C(C)CC)NC(=O)C(CO)NC(=O)C(C(C)O)NC(=O)C1CSSCC2NC(=O)C(CC(C)C)NC(=O)C(NC(=O)C(CCC(N)=O)NC(=O)C(CC(N)=O)NC(=O)C(NC(=O)C(N)CC=1C=CC=CC=1)C(C)C)CC1=CN=CN1 NOESYZHRGYRDHS-UHFFFAOYSA-N 0.000 description 2
- JVTAAEKCZFNVCJ-UHFFFAOYSA-N lactic acid Chemical compound CC(O)C(O)=O JVTAAEKCZFNVCJ-UHFFFAOYSA-N 0.000 description 2
- 229940041476 lactose 100 mg Drugs 0.000 description 2
- 210000004185 liver Anatomy 0.000 description 2
- 235000019359 magnesium stearate Nutrition 0.000 description 2
- 229910052751 metal Inorganic materials 0.000 description 2
- 239000002184 metal Substances 0.000 description 2
- 150000007522 mineralic acids Chemical class 0.000 description 2
- 210000003205 muscle Anatomy 0.000 description 2
- 235000005985 organic acids Nutrition 0.000 description 2
- 201000008482 osteoarthritis Diseases 0.000 description 2
- 235000020830 overeating Nutrition 0.000 description 2
- 150000004804 polysaccharides Polymers 0.000 description 2
- BWHMMNNQKKPAPP-UHFFFAOYSA-L potassium carbonate Chemical compound [K+].[K+].[O-]C([O-])=O BWHMMNNQKKPAPP-UHFFFAOYSA-L 0.000 description 2
- 239000000843 powder Substances 0.000 description 2
- 238000001671 psychotherapy Methods 0.000 description 2
- 238000000926 separation method Methods 0.000 description 2
- 210000002966 serum Anatomy 0.000 description 2
- 238000010898 silica gel chromatography Methods 0.000 description 2
- SQGYOTSLMSWVJD-UHFFFAOYSA-N silver(1+) nitrate Chemical compound [Ag+].[O-]N(=O)=O SQGYOTSLMSWVJD-UHFFFAOYSA-N 0.000 description 2
- GGCZERPQGJTIQP-UHFFFAOYSA-N sodium;9,10-dioxoanthracene-2-sulfonic acid Chemical compound [Na+].C1=CC=C2C(=O)C3=CC(S(=O)(=O)O)=CC=C3C(=O)C2=C1 GGCZERPQGJTIQP-UHFFFAOYSA-N 0.000 description 2
- 239000007787 solid Substances 0.000 description 2
- 230000035882 stress Effects 0.000 description 2
- 235000000346 sugar Nutrition 0.000 description 2
- 230000009182 swimming Effects 0.000 description 2
- 239000006188 syrup Substances 0.000 description 2
- 235000020357 syrup Nutrition 0.000 description 2
- 239000011975 tartaric acid Substances 0.000 description 2
- 235000002906 tartaric acid Nutrition 0.000 description 2
- 210000001519 tissue Anatomy 0.000 description 2
- VZCYOOQTPOCHFL-UHFFFAOYSA-N trans-butenedioic acid Natural products OC(=O)C=CC(O)=O VZCYOOQTPOCHFL-UHFFFAOYSA-N 0.000 description 2
- 235000015192 vegetable juice Nutrition 0.000 description 2
- 235000013311 vegetables Nutrition 0.000 description 2
- 238000009423 ventilation Methods 0.000 description 2
- 235000013343 vitamin Nutrition 0.000 description 2
- 229930003231 vitamin Natural products 0.000 description 2
- 239000011782 vitamin Substances 0.000 description 2
- 229940088594 vitamin Drugs 0.000 description 2
- 239000013585 weight reducing agent Substances 0.000 description 2
- QBYIENPQHBMVBV-HFEGYEGKSA-N (2R)-2-hydroxy-2-phenylacetic acid Chemical compound O[C@@H](C(O)=O)c1ccccc1.O[C@@H](C(O)=O)c1ccccc1 QBYIENPQHBMVBV-HFEGYEGKSA-N 0.000 description 1
- RTBFRGCFXZNCOE-UHFFFAOYSA-N 1-methylsulfonylpiperidin-4-one Chemical compound CS(=O)(=O)N1CCC(=O)CC1 RTBFRGCFXZNCOE-UHFFFAOYSA-N 0.000 description 1
- OWEGMIWEEQEYGQ-UHFFFAOYSA-N 100676-05-9 Natural products OC1C(O)C(O)C(CO)OC1OCC1C(O)C(O)C(O)C(OC2C(OC(O)C(O)C2O)CO)O1 OWEGMIWEEQEYGQ-UHFFFAOYSA-N 0.000 description 1
- XNWFRZJHXBZDAG-UHFFFAOYSA-N 2-METHOXYETHANOL Chemical compound COCCO XNWFRZJHXBZDAG-UHFFFAOYSA-N 0.000 description 1
- MSWZFWKMSRAUBD-GASJEMHNSA-N 2-amino-2-deoxy-D-galactopyranose Chemical compound N[C@H]1C(O)O[C@H](CO)[C@H](O)[C@@H]1O MSWZFWKMSRAUBD-GASJEMHNSA-N 0.000 description 1
- MIDXCONKKJTLDX-UHFFFAOYSA-N 3,5-dimethylcyclopentane-1,2-dione Chemical compound CC1CC(C)C(=O)C1=O MIDXCONKKJTLDX-UHFFFAOYSA-N 0.000 description 1
- HVCOBJNICQPDBP-UHFFFAOYSA-N 3-[3-[3,5-dihydroxy-6-methyl-4-(3,4,5-trihydroxy-6-methyloxan-2-yl)oxyoxan-2-yl]oxydecanoyloxy]decanoic acid;hydrate Chemical compound O.OC1C(OC(CC(=O)OC(CCCCCCC)CC(O)=O)CCCCCCC)OC(C)C(O)C1OC1C(O)C(O)C(O)C(C)O1 HVCOBJNICQPDBP-UHFFFAOYSA-N 0.000 description 1
- 208000021769 Acute sensory ataxic neuropathy Diseases 0.000 description 1
- 108010011485 Aspartame Proteins 0.000 description 1
- 241000894006 Bacteria Species 0.000 description 1
- 208000034577 Benign intracranial hypertension Diseases 0.000 description 1
- 239000005711 Benzoic acid Substances 0.000 description 1
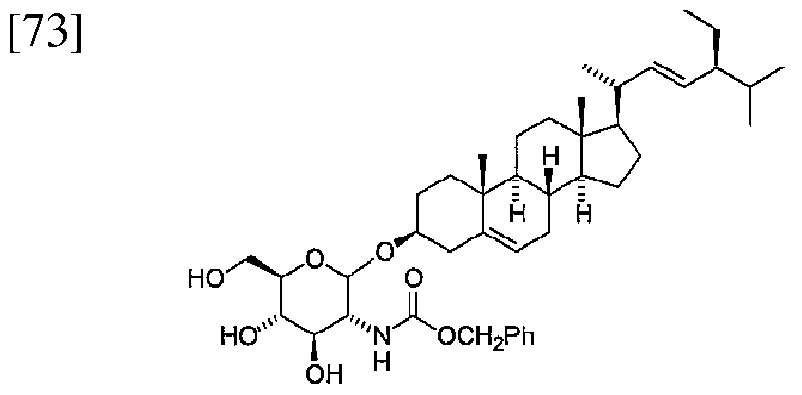
- 0 CC(C)[C@](C=C)*=C[C@@](C)[C@@](CC1)[C@@](C)(CC2)[C@@]1[C@]1C2[C@@](C)(CC[C@@](C2)OC([C@](C3OC(C)=O)NC(OCc4ccccc4)=O)O[C@](COC(C)=O)[C@]3OC(C)=O)C2=CC1 Chemical compound CC(C)[C@](C=C)*=C[C@@](C)[C@@](CC1)[C@@](C)(CC2)[C@@]1[C@]1C2[C@@](C)(CC[C@@](C2)OC([C@](C3OC(C)=O)NC(OCc4ccccc4)=O)O[C@](COC(C)=O)[C@]3OC(C)=O)C2=CC1 0.000 description 1
- 208000024172 Cardiovascular disease Diseases 0.000 description 1
- KRKNYBCHXYNGOX-UHFFFAOYSA-K Citrate Chemical compound [O-]C(=O)CC(O)(CC([O-])=O)C([O-])=O KRKNYBCHXYNGOX-UHFFFAOYSA-K 0.000 description 1
- 229920000858 Cyclodextrin Polymers 0.000 description 1
- FBPFZTCFMRRESA-FSIIMWSLSA-N D-Glucitol Natural products OC[C@H](O)[C@H](O)[C@@H](O)[C@H](O)CO FBPFZTCFMRRESA-FSIIMWSLSA-N 0.000 description 1
- ZZZCUOFIHGPKAK-UHFFFAOYSA-N D-erythro-ascorbic acid Natural products OCC1OC(=O)C(O)=C1O ZZZCUOFIHGPKAK-UHFFFAOYSA-N 0.000 description 1
- FBPFZTCFMRRESA-JGWLITMVSA-N D-glucitol Chemical compound OC[C@H](O)[C@@H](O)[C@H](O)[C@H](O)CO FBPFZTCFMRRESA-JGWLITMVSA-N 0.000 description 1
- RGHNJXZEOKUKBD-UHFFFAOYSA-N D-gluconic acid Natural products OCC(O)C(O)C(O)C(O)C(O)=O RGHNJXZEOKUKBD-UHFFFAOYSA-N 0.000 description 1
- 229920001353 Dextrin Polymers 0.000 description 1
- 239000004375 Dextrin Substances 0.000 description 1
- 239000004386 Erythritol Substances 0.000 description 1
- UNXHWFMMPAWVPI-UHFFFAOYSA-N Erythritol Natural products OCC(O)C(O)CO UNXHWFMMPAWVPI-UHFFFAOYSA-N 0.000 description 1
- WHUUTDBJXJRKMK-UHFFFAOYSA-N Glutamic acid Natural products OC(=O)C(N)CCC(O)=O WHUUTDBJXJRKMK-UHFFFAOYSA-N 0.000 description 1
- 244000068988 Glycine max Species 0.000 description 1
- 235000010469 Glycine max Nutrition 0.000 description 1
- AEMRFAOFKBGASW-UHFFFAOYSA-N Glycolic acid Chemical compound OCC(O)=O AEMRFAOFKBGASW-UHFFFAOYSA-N 0.000 description 1
- 229930186217 Glycolipid Natural products 0.000 description 1
- 229920002683 Glycosaminoglycan Polymers 0.000 description 1
- 208000018127 Idiopathic intracranial hypertension Diseases 0.000 description 1
- DGAQECJNVWCQMB-PUAWFVPOSA-M Ilexoside XXIX Chemical compound C[C@@H]1CC[C@@]2(CC[C@@]3(C(=CC[C@H]4[C@]3(CC[C@@H]5[C@@]4(CC[C@@H](C5(C)C)OS(=O)(=O)[O-])C)C)[C@@H]2[C@]1(C)O)C)C(=O)O[C@H]6[C@@H]([C@H]([C@@H]([C@H](O6)CO)O)O)O.[Na+] DGAQECJNVWCQMB-PUAWFVPOSA-M 0.000 description 1
- 102000004877 Insulin Human genes 0.000 description 1
- 108090001061 Insulin Proteins 0.000 description 1
- CKLJMWTZIZZHCS-REOHCLBHSA-N L-aspartic acid Chemical compound OC(=O)[C@@H](N)CC(O)=O CKLJMWTZIZZHCS-REOHCLBHSA-N 0.000 description 1
- WHUUTDBJXJRKMK-VKHMYHEASA-N L-glutamic acid Chemical compound OC(=O)[C@@H](N)CCC(O)=O WHUUTDBJXJRKMK-VKHMYHEASA-N 0.000 description 1
- 231100000111 LD50 Toxicity 0.000 description 1
- JVTAAEKCZFNVCJ-UHFFFAOYSA-M Lactate Chemical compound CC(O)C([O-])=O JVTAAEKCZFNVCJ-UHFFFAOYSA-M 0.000 description 1
- GUBGYTABKSRVRQ-QKKXKWKRSA-N Lactose Natural products OC[C@H]1O[C@@H](O[C@H]2[C@H](O)[C@@H](O)C(O)O[C@@H]2CO)[C@H](O)[C@@H](O)[C@H]1O GUBGYTABKSRVRQ-QKKXKWKRSA-N 0.000 description 1
- 208000002720 Malnutrition Diseases 0.000 description 1
- 229920002774 Maltodextrin Polymers 0.000 description 1
- 239000005913 Maltodextrin Substances 0.000 description 1
- GUBGYTABKSRVRQ-PICCSMPSSA-N Maltose Natural products O[C@@H]1[C@@H](O)[C@H](O)[C@@H](CO)O[C@@H]1O[C@@H]1[C@@H](CO)OC(O)[C@H](O)[C@H]1O GUBGYTABKSRVRQ-PICCSMPSSA-N 0.000 description 1
- 241000699666 Mus <mouse, genus> Species 0.000 description 1
- GRYLNZFGIOXLOG-UHFFFAOYSA-N Nitric acid Chemical compound O[N+]([O-])=O GRYLNZFGIOXLOG-UHFFFAOYSA-N 0.000 description 1
- 229910019142 PO4 Inorganic materials 0.000 description 1
- 208000002193 Pain Diseases 0.000 description 1
- 229920002230 Pectic acid Polymers 0.000 description 1
- NBIIXXVUZAFLBC-UHFFFAOYSA-L Phosphate ion(2-) Chemical compound OP([O-])([O-])=O NBIIXXVUZAFLBC-UHFFFAOYSA-L 0.000 description 1
- ZLMJMSJWJFRBEC-UHFFFAOYSA-N Potassium Chemical compound [K] ZLMJMSJWJFRBEC-UHFFFAOYSA-N 0.000 description 1
- OFOBLEOULBTSOW-UHFFFAOYSA-N Propanedioic acid Natural products OC(=O)CC(O)=O OFOBLEOULBTSOW-UHFFFAOYSA-N 0.000 description 1
- IWYDHOAUDWTVEP-UHFFFAOYSA-N R-2-phenyl-2-hydroxyacetic acid Natural products OC(=O)C(O)C1=CC=CC=C1 IWYDHOAUDWTVEP-UHFFFAOYSA-N 0.000 description 1
- KEAYESYHFKHZAL-UHFFFAOYSA-N Sodium Chemical compound [Na] KEAYESYHFKHZAL-UHFFFAOYSA-N 0.000 description 1
- UIIMBOGNXHQVGW-DEQYMQKBSA-M Sodium bicarbonate-14C Chemical compound [Na+].O[14C]([O-])=O UIIMBOGNXHQVGW-DEQYMQKBSA-M 0.000 description 1
- UEDUENGHJMELGK-HYDKPPNVSA-N Stevioside Chemical compound O([C@@H]1[C@@H](O)[C@H](O)[C@@H](CO)O[C@H]1O[C@]12C(=C)C[C@@]3(C1)CC[C@@H]1[C@@](C)(CCC[C@]1([C@@H]3CC2)C)C(=O)O[C@H]1[C@@H]([C@@H](O)[C@H](O)[C@@H](CO)O1)O)[C@@H]1O[C@H](CO)[C@@H](O)[C@H](O)[C@H]1O UEDUENGHJMELGK-HYDKPPNVSA-N 0.000 description 1
- KDYFGRWQOYBRFD-UHFFFAOYSA-N Succinic acid Natural products OC(=O)CCC(O)=O KDYFGRWQOYBRFD-UHFFFAOYSA-N 0.000 description 1
- 229930006000 Sucrose Natural products 0.000 description 1
- CZMRCDWAGMRECN-UGDNZRGBSA-N Sucrose Chemical compound O[C@H]1[C@H](O)[C@@H](CO)O[C@@]1(CO)O[C@@H]1[C@H](O)[C@@H](O)[C@H](O)[C@@H](CO)O1 CZMRCDWAGMRECN-UGDNZRGBSA-N 0.000 description 1
- 244000269722 Thea sinensis Species 0.000 description 1
- ZMANZCXQSJIPKH-UHFFFAOYSA-N Triethylamine Chemical compound CCN(CC)CC ZMANZCXQSJIPKH-UHFFFAOYSA-N 0.000 description 1
- 229930003268 Vitamin C Natural products 0.000 description 1
- TVXBFESIOXBWNM-UHFFFAOYSA-N Xylitol Natural products OCCC(O)C(O)C(O)CCO TVXBFESIOXBWNM-UHFFFAOYSA-N 0.000 description 1
- 230000003187 abdominal effect Effects 0.000 description 1
- 238000010521 absorption reaction Methods 0.000 description 1
- 125000002777 acetyl group Chemical group [H]C([H])([H])C(*)=O 0.000 description 1
- 230000002378 acidificating effect Effects 0.000 description 1
- 230000004913 activation Effects 0.000 description 1
- 231100000403 acute toxicity Toxicity 0.000 description 1
- 230000007059 acute toxicity Effects 0.000 description 1
- 230000032683 aging Effects 0.000 description 1
- 235000013334 alcoholic beverage Nutrition 0.000 description 1
- IAJILQKETJEXLJ-RSJOWCBRSA-N aldehydo-D-galacturonic acid Chemical compound O=C[C@H](O)[C@@H](O)[C@@H](O)[C@H](O)C(O)=O IAJILQKETJEXLJ-RSJOWCBRSA-N 0.000 description 1
- IAJILQKETJEXLJ-QTBDOELSSA-N aldehydo-D-glucuronic acid Chemical compound O=C[C@H](O)[C@@H](O)[C@H](O)[C@H](O)C(O)=O IAJILQKETJEXLJ-QTBDOELSSA-N 0.000 description 1
- 239000000783 alginic acid Substances 0.000 description 1
- 235000010443 alginic acid Nutrition 0.000 description 1
- 229920000615 alginic acid Polymers 0.000 description 1
- 229960001126 alginic acid Drugs 0.000 description 1
- 150000004781 alginic acids Chemical class 0.000 description 1
- 150000008044 alkali metal hydroxides Chemical class 0.000 description 1
- 229910001860 alkaline earth metal hydroxide Inorganic materials 0.000 description 1
- 229910000147 aluminium phosphate Inorganic materials 0.000 description 1
- 125000003277 amino group Chemical group 0.000 description 1
- 239000000908 ammonium hydroxide Substances 0.000 description 1
- JFCQEDHGNNZCLN-UHFFFAOYSA-N anhydrous glutaric acid Natural products OC(=O)CCCC(O)=O JFCQEDHGNNZCLN-UHFFFAOYSA-N 0.000 description 1
- 239000003963 antioxidant agent Substances 0.000 description 1
- 235000006708 antioxidants Nutrition 0.000 description 1
- 239000002830 appetite depressant Substances 0.000 description 1
- 235000015197 apple juice Nutrition 0.000 description 1
- 239000007900 aqueous suspension Substances 0.000 description 1
- 206010003246 arthritis Diseases 0.000 description 1
- 239000008122 artificial sweetener Substances 0.000 description 1
- 235000021311 artificial sweeteners Nutrition 0.000 description 1
- 235000010323 ascorbic acid Nutrition 0.000 description 1
- 229960005070 ascorbic acid Drugs 0.000 description 1
- 239000011668 ascorbic acid Substances 0.000 description 1
- 239000000605 aspartame Substances 0.000 description 1
- 235000010357 aspartame Nutrition 0.000 description 1
- IAOZJIPTCAWIRG-QWRGUYRKSA-N aspartame Chemical compound OC(=O)C[C@H](N)C(=O)N[C@H](C(=O)OC)CC1=CC=CC=C1 IAOZJIPTCAWIRG-QWRGUYRKSA-N 0.000 description 1
- 229960003438 aspartame Drugs 0.000 description 1
- 235000003704 aspartic acid Nutrition 0.000 description 1
- 229960005261 aspartic acid Drugs 0.000 description 1
- 238000011888 autopsy Methods 0.000 description 1
- 239000000022 bacteriostatic agent Substances 0.000 description 1
- 235000010233 benzoic acid Nutrition 0.000 description 1
- WQZGKKKJIJFFOK-VFUOTHLCSA-N beta-D-glucose Chemical compound OC[C@H]1O[C@@H](O)[C@H](O)[C@@H](O)[C@@H]1O WQZGKKKJIJFFOK-VFUOTHLCSA-N 0.000 description 1
- OQFSQFPPLPISGP-UHFFFAOYSA-N beta-carboxyaspartic acid Natural products OC(=O)C(N)C(C(O)=O)C(O)=O OQFSQFPPLPISGP-UHFFFAOYSA-N 0.000 description 1
- GUBGYTABKSRVRQ-QUYVBRFLSA-N beta-maltose Chemical compound OC[C@H]1O[C@H](O[C@H]2[C@H](O)[C@@H](O)[C@H](O)O[C@@H]2CO)[C@H](O)[C@@H](O)[C@@H]1O GUBGYTABKSRVRQ-QUYVBRFLSA-N 0.000 description 1
- 239000011230 binding agent Substances 0.000 description 1
- 238000010876 biochemical test Methods 0.000 description 1
- 230000036772 blood pressure Effects 0.000 description 1
- 210000004204 blood vessel Anatomy 0.000 description 1
- 210000004556 brain Anatomy 0.000 description 1
- 239000000872 buffer Substances 0.000 description 1
- 239000007975 buffered saline Substances 0.000 description 1
- KDYFGRWQOYBRFD-NUQCWPJISA-N butanedioic acid Chemical compound O[14C](=O)CC[14C](O)=O KDYFGRWQOYBRFD-NUQCWPJISA-N 0.000 description 1
- 235000014121 butter Nutrition 0.000 description 1
- FJDQFPXHSGXQBY-UHFFFAOYSA-L caesium carbonate Chemical compound [Cs+].[Cs+].[O-]C([O-])=O FJDQFPXHSGXQBY-UHFFFAOYSA-L 0.000 description 1
- 229910000024 caesium carbonate Inorganic materials 0.000 description 1
- 235000012970 cakes Nutrition 0.000 description 1
- 239000001506 calcium phosphate Substances 0.000 description 1
- 229910000389 calcium phosphate Inorganic materials 0.000 description 1
- 235000011010 calcium phosphates Nutrition 0.000 description 1
- 235000019577 caloric intake Nutrition 0.000 description 1
- 235000013736 caramel Nutrition 0.000 description 1
- 125000004432 carbon atom Chemical group C* 0.000 description 1
- 239000001569 carbon dioxide Substances 0.000 description 1
- 229910002092 carbon dioxide Inorganic materials 0.000 description 1
- 235000015190 carrot juice Nutrition 0.000 description 1
- 210000004027 cell Anatomy 0.000 description 1
- 210000000170 cell membrane Anatomy 0.000 description 1
- 210000002421 cell wall Anatomy 0.000 description 1
- 239000003153 chemical reaction reagent Substances 0.000 description 1
- 239000007810 chemical reaction solvent Substances 0.000 description 1
- 235000015218 chewing gum Nutrition 0.000 description 1
- 229940112822 chewing gum Drugs 0.000 description 1
- 235000019219 chocolate Nutrition 0.000 description 1
- 201000001352 cholecystitis Diseases 0.000 description 1
- 239000000084 colloidal system Substances 0.000 description 1
- 239000003086 colorant Substances 0.000 description 1
- 238000004737 colorimetric analysis Methods 0.000 description 1
- 235000009508 confectionery Nutrition 0.000 description 1
- 239000002537 cosmetic Substances 0.000 description 1
- 235000012495 crackers Nutrition 0.000 description 1
- 239000013078 crystal Substances 0.000 description 1
- 230000006378 damage Effects 0.000 description 1
- 230000003247 decreasing effect Effects 0.000 description 1
- 235000019425 dextrin Nutrition 0.000 description 1
- 206010012601 diabetes mellitus Diseases 0.000 description 1
- 235000018823 dietary intake Nutrition 0.000 description 1
- 230000029087 digestion Effects 0.000 description 1
- NBIIXXVUZAFLBC-UHFFFAOYSA-M dihydrogenphosphate Chemical compound OP(O)([O-])=O NBIIXXVUZAFLBC-UHFFFAOYSA-M 0.000 description 1
- 239000003085 diluting agent Substances 0.000 description 1
- 150000002016 disaccharides Chemical class 0.000 description 1
- 239000002270 dispersing agent Substances 0.000 description 1
- 206010013663 drug dependence Diseases 0.000 description 1
- 235000005686 eating Nutrition 0.000 description 1
- 235000013399 edible fruits Nutrition 0.000 description 1
- 239000000839 emulsion Substances 0.000 description 1
- 230000002124 endocrine Effects 0.000 description 1
- 230000037149 energy metabolism Effects 0.000 description 1
- 230000002255 enzymatic effect Effects 0.000 description 1
- 235000019414 erythritol Nutrition 0.000 description 1
- UNXHWFMMPAWVPI-ZXZARUISSA-N erythritol Chemical compound OC[C@H](O)[C@H](O)CO UNXHWFMMPAWVPI-ZXZARUISSA-N 0.000 description 1
- 229940009714 erythritol Drugs 0.000 description 1
- 210000003743 erythrocyte Anatomy 0.000 description 1
- 238000001704 evaporation Methods 0.000 description 1
- 230000008020 evaporation Effects 0.000 description 1
- 235000013410 fast food Nutrition 0.000 description 1
- 239000000706 filtrate Substances 0.000 description 1
- 235000019634 flavors Nutrition 0.000 description 1
- 235000013373 food additive Nutrition 0.000 description 1
- 239000002778 food additive Substances 0.000 description 1
- 235000012041 food component Nutrition 0.000 description 1
- 235000013355 food flavoring agent Nutrition 0.000 description 1
- 239000005417 food ingredient Substances 0.000 description 1
- 235000003599 food sweetener Nutrition 0.000 description 1
- 239000001530 fumaric acid Substances 0.000 description 1
- 235000011087 fumaric acid Nutrition 0.000 description 1
- 230000002496 gastric effect Effects 0.000 description 1
- 239000007903 gelatin capsule Substances 0.000 description 1
- 239000011521 glass Substances 0.000 description 1
- 239000000174 gluconic acid Substances 0.000 description 1
- 235000012208 gluconic acid Nutrition 0.000 description 1
- 239000008103 glucose Substances 0.000 description 1
- 229940097043 glucuronic acid Drugs 0.000 description 1
- 235000013922 glutamic acid Nutrition 0.000 description 1
- 239000004220 glutamic acid Substances 0.000 description 1
- 150000004676 glycans Polymers 0.000 description 1
- 150000002337 glycosamines Chemical class 0.000 description 1
- 239000008187 granular material Substances 0.000 description 1
- 235000019674 grape juice Nutrition 0.000 description 1
- 238000001794 hormone therapy Methods 0.000 description 1
- 235000003642 hunger Nutrition 0.000 description 1
- XMBWDFGMSWQBCA-UHFFFAOYSA-N hydrogen iodide Chemical compound I XMBWDFGMSWQBCA-UHFFFAOYSA-N 0.000 description 1
- QAOWNCQODCNURD-UHFFFAOYSA-M hydrogensulfate Chemical compound OS([O-])(=O)=O QAOWNCQODCNURD-UHFFFAOYSA-M 0.000 description 1
- 229940071870 hydroiodic acid Drugs 0.000 description 1
- 125000002887 hydroxy group Chemical group [H]O* 0.000 description 1
- 210000003016 hypothalamus Anatomy 0.000 description 1
- 238000005286 illumination Methods 0.000 description 1
- 239000007972 injectable composition Substances 0.000 description 1
- 229910052500 inorganic mineral Inorganic materials 0.000 description 1
- 235000008446 instant noodles Nutrition 0.000 description 1
- 229940125396 insulin Drugs 0.000 description 1
- 238000007912 intraperitoneal administration Methods 0.000 description 1
- 239000004310 lactic acid Substances 0.000 description 1
- 235000014655 lactic acid Nutrition 0.000 description 1
- 239000008101 lactose Substances 0.000 description 1
- 231100000518 lethal Toxicity 0.000 description 1
- 230000001665 lethal effect Effects 0.000 description 1
- 108010022197 lipoprotein cholesterol Proteins 0.000 description 1
- 239000007788 liquid Substances 0.000 description 1
- 239000000314 lubricant Substances 0.000 description 1
- VZCYOOQTPOCHFL-UPHRSURJSA-N maleic acid Chemical compound OC(=O)\C=C/C(O)=O VZCYOOQTPOCHFL-UPHRSURJSA-N 0.000 description 1
- 239000011976 maleic acid Substances 0.000 description 1
- 230000001071 malnutrition Effects 0.000 description 1
- 235000000824 malnutrition Nutrition 0.000 description 1
- 229940035034 maltodextrin Drugs 0.000 description 1
- IWYDHOAUDWTVEP-UHFFFAOYSA-M mandelate Chemical compound [O-]C(=O)C(O)C1=CC=CC=C1 IWYDHOAUDWTVEP-UHFFFAOYSA-M 0.000 description 1
- 229960002510 mandelic acid Drugs 0.000 description 1
- 235000013372 meat Nutrition 0.000 description 1
- 230000003340 mental effect Effects 0.000 description 1
- HEBKCHPVOIAQTA-UHFFFAOYSA-N meso ribitol Natural products OCC(O)C(O)C(O)CO HEBKCHPVOIAQTA-UHFFFAOYSA-N 0.000 description 1
- 229940098779 methanesulfonic acid Drugs 0.000 description 1
- 229920000609 methyl cellulose Polymers 0.000 description 1
- 239000001923 methylcellulose Substances 0.000 description 1
- 235000010981 methylcellulose Nutrition 0.000 description 1
- 235000010755 mineral Nutrition 0.000 description 1
- 239000011707 mineral Substances 0.000 description 1
- 150000002772 monosaccharides Chemical class 0.000 description 1
- 210000003097 mucus Anatomy 0.000 description 1
- 201000000585 muscular atrophy Diseases 0.000 description 1
- 235000021096 natural sweeteners Nutrition 0.000 description 1
- 229910017604 nitric acid Inorganic materials 0.000 description 1
- 235000015097 nutrients Nutrition 0.000 description 1
- 208000015380 nutritional deficiency disease Diseases 0.000 description 1
- 229920001542 oligosaccharide Polymers 0.000 description 1
- 150000002482 oligosaccharides Chemical class 0.000 description 1
- 235000006408 oxalic acid Nutrition 0.000 description 1
- 230000036407 pain Effects 0.000 description 1
- 229910052763 palladium Inorganic materials 0.000 description 1
- 239000002245 particle Substances 0.000 description 1
- LCLHHZYHLXDRQG-ZNKJPWOQSA-N pectic acid Chemical compound O[C@@H]1[C@@H](O)[C@@H](O)O[C@H](C(O)=O)[C@@H]1OC1[C@H](O)[C@@H](O)[C@@H](OC2[C@@H]([C@@H](O)[C@@H](O)[C@H](O2)C(O)=O)O)[C@@H](C(O)=O)O1 LCLHHZYHLXDRQG-ZNKJPWOQSA-N 0.000 description 1
- 239000000825 pharmaceutical preparation Substances 0.000 description 1
- NBIIXXVUZAFLBC-UHFFFAOYSA-K phosphate Chemical compound [O-]P([O-])([O-])=O NBIIXXVUZAFLBC-UHFFFAOYSA-K 0.000 description 1
- 239000010452 phosphate Substances 0.000 description 1
- 239000006187 pill Substances 0.000 description 1
- 235000013550 pizza Nutrition 0.000 description 1
- 229920000515 polycarbonate Polymers 0.000 description 1
- 239000004417 polycarbonate Substances 0.000 description 1
- 206010036067 polydipsia Diseases 0.000 description 1
- 239000010318 polygalacturonic acid Substances 0.000 description 1
- 229920001282 polysaccharide Polymers 0.000 description 1
- 239000005017 polysaccharide Substances 0.000 description 1
- 229910052700 potassium Inorganic materials 0.000 description 1
- 239000011591 potassium Substances 0.000 description 1
- XAEFZNCEHLXOMS-UHFFFAOYSA-M potassium benzoate Chemical compound [K+].[O-]C(=O)C1=CC=CC=C1 XAEFZNCEHLXOMS-UHFFFAOYSA-M 0.000 description 1
- 229910000028 potassium bicarbonate Inorganic materials 0.000 description 1
- 235000015497 potassium bicarbonate Nutrition 0.000 description 1
- 239000011736 potassium bicarbonate Substances 0.000 description 1
- 229910000027 potassium carbonate Inorganic materials 0.000 description 1
- 235000011181 potassium carbonates Nutrition 0.000 description 1
- NTTOTNSKUYCDAV-UHFFFAOYSA-N potassium hydride Chemical compound [KH] NTTOTNSKUYCDAV-UHFFFAOYSA-N 0.000 description 1
- 229910000105 potassium hydride Inorganic materials 0.000 description 1
- TYJJADVDDVDEDZ-UHFFFAOYSA-M potassium hydrogencarbonate Chemical compound [K+].OC([O-])=O TYJJADVDDVDEDZ-UHFFFAOYSA-M 0.000 description 1
- 230000001376 precipitating effect Effects 0.000 description 1
- 239000003755 preservative agent Substances 0.000 description 1
- 230000002335 preservative effect Effects 0.000 description 1
- 230000002265 prevention Effects 0.000 description 1
- 235000020991 processed meat Nutrition 0.000 description 1
- 235000019260 propionic acid Nutrition 0.000 description 1
- 230000001681 protective effect Effects 0.000 description 1
- 235000018102 proteins Nutrition 0.000 description 1
- 102000004169 proteins and genes Human genes 0.000 description 1
- 108090000623 proteins and genes Proteins 0.000 description 1
- YQUVCSBJEUQKSH-UHFFFAOYSA-N protochatechuic acid Natural products OC(=O)C1=CC=C(O)C(O)=C1 YQUVCSBJEUQKSH-UHFFFAOYSA-N 0.000 description 1
- 208000001381 pseudotumor cerebri Diseases 0.000 description 1
- 208000002815 pulmonary hypertension Diseases 0.000 description 1
- 239000008213 purified water Substances 0.000 description 1
- UMJSCPRVCHMLSP-UHFFFAOYSA-N pyridine Natural products COC1=CC=CN=C1 UMJSCPRVCHMLSP-UHFFFAOYSA-N 0.000 description 1
- IUVKMZGDUIUOCP-BTNSXGMBSA-N quinbolone Chemical compound O([C@H]1CC[C@H]2[C@H]3[C@@H]([C@]4(C=CC(=O)C=C4CC3)C)CC[C@@]21C)C1=CCCC1 IUVKMZGDUIUOCP-BTNSXGMBSA-N 0.000 description 1
- 239000002994 raw material Substances 0.000 description 1
- 238000010992 reflux Methods 0.000 description 1
- 238000012552 review Methods 0.000 description 1
- 235000019204 saccharin Nutrition 0.000 description 1
- CVHZOJJKTDOEJC-UHFFFAOYSA-N saccharin Chemical compound C1=CC=C2C(=O)NS(=O)(=O)C2=C1 CVHZOJJKTDOEJC-UHFFFAOYSA-N 0.000 description 1
- 229940081974 saccharin Drugs 0.000 description 1
- 239000000901 saccharin and its Na,K and Ca salt Substances 0.000 description 1
- 235000013580 sausages Nutrition 0.000 description 1
- HFHDHCJBZVLPGP-UHFFFAOYSA-N schardinger α-dextrin Chemical compound O1C(C(C2O)O)C(CO)OC2OC(C(C2O)O)C(CO)OC2OC(C(C2O)O)C(CO)OC2OC(C(O)C2O)C(CO)OC2OC(C(C2O)O)C(CO)OC2OC2C(O)C(O)C1OC2CO HFHDHCJBZVLPGP-UHFFFAOYSA-N 0.000 description 1
- 230000028327 secretion Effects 0.000 description 1
- 229910001961 silver nitrate Inorganic materials 0.000 description 1
- 201000002859 sleep apnea Diseases 0.000 description 1
- 235000011888 snacks Nutrition 0.000 description 1
- 229910052708 sodium Inorganic materials 0.000 description 1
- 239000011734 sodium Substances 0.000 description 1
- 229910000029 sodium carbonate Inorganic materials 0.000 description 1
- 239000011780 sodium chloride Substances 0.000 description 1
- 239000012312 sodium hydride Substances 0.000 description 1
- 229910000104 sodium hydride Inorganic materials 0.000 description 1
- 159000000000 sodium salts Chemical class 0.000 description 1
- 239000012453 solvate Substances 0.000 description 1
- 239000002904 solvent Substances 0.000 description 1
- 239000011877 solvent mixture Substances 0.000 description 1
- 239000000600 sorbitol Substances 0.000 description 1
- 229960002920 sorbitol Drugs 0.000 description 1
- 235000010356 sorbitol Nutrition 0.000 description 1
- 239000003381 stabilizer Substances 0.000 description 1
- 239000007858 starting material Substances 0.000 description 1
- 239000008223 sterile water Substances 0.000 description 1
- 229940013618 stevioside Drugs 0.000 description 1
- OHHNJQXIOPOJSC-UHFFFAOYSA-N stevioside Natural products CC1(CCCC2(C)C3(C)CCC4(CC3(CCC12C)CC4=C)OC5OC(CO)C(O)C(O)C5OC6OC(CO)C(O)C(O)C6O)C(=O)OC7OC(CO)C(O)C(O)C7O OHHNJQXIOPOJSC-UHFFFAOYSA-N 0.000 description 1
- 235000019202 steviosides Nutrition 0.000 description 1
- 238000003756 stirring Methods 0.000 description 1
- 238000007920 subcutaneous administration Methods 0.000 description 1
- 208000011117 substance-related disease Diseases 0.000 description 1
- KDYFGRWQOYBRFD-UHFFFAOYSA-L succinate(2-) Chemical compound [O-]C(=O)CCC([O-])=O KDYFGRWQOYBRFD-UHFFFAOYSA-L 0.000 description 1
- 239000005720 sucrose Substances 0.000 description 1
- 150000005846 sugar alcohols Chemical class 0.000 description 1
- 239000004094 surface-active agent Substances 0.000 description 1
- 239000003765 sweetening agent Substances 0.000 description 1
- 208000024891 symptom Diseases 0.000 description 1
- 238000003786 synthesis reaction Methods 0.000 description 1
- 239000003826 tablet Substances 0.000 description 1
- 229940095064 tartrate Drugs 0.000 description 1
- 235000013616 tea Nutrition 0.000 description 1
- YLQBMQCUIZJEEH-UHFFFAOYSA-N tetrahydrofuran Natural products C=1C=COC=1 YLQBMQCUIZJEEH-UHFFFAOYSA-N 0.000 description 1
- 239000000892 thaumatin Substances 0.000 description 1
- 235000010436 thaumatin Nutrition 0.000 description 1
- 239000002562 thickening agent Substances 0.000 description 1
- 235000015193 tomato juice Nutrition 0.000 description 1
- 230000000699 topical effect Effects 0.000 description 1
- 231100000419 toxicity Toxicity 0.000 description 1
- 230000001988 toxicity Effects 0.000 description 1
- 231100000820 toxicity test Toxicity 0.000 description 1
- QORWJWZARLRLPR-UHFFFAOYSA-H tricalcium bis(phosphate) Chemical compound [Ca+2].[Ca+2].[Ca+2].[O-]P([O-])([O-])=O.[O-]P([O-])([O-])=O QORWJWZARLRLPR-UHFFFAOYSA-H 0.000 description 1
- WKOLLVMJNQIZCI-UHFFFAOYSA-N vanillic acid Chemical compound COC1=CC(C(O)=O)=CC=C1O WKOLLVMJNQIZCI-UHFFFAOYSA-N 0.000 description 1
- TUUBOHWZSQXCSW-UHFFFAOYSA-N vanillic acid Natural products COC1=CC(O)=CC(C(O)=O)=C1 TUUBOHWZSQXCSW-UHFFFAOYSA-N 0.000 description 1
- 210000001075 venae cavae Anatomy 0.000 description 1
- 235000019154 vitamin C Nutrition 0.000 description 1
- 239000011718 vitamin C Substances 0.000 description 1
- 150000003722 vitamin derivatives Chemical class 0.000 description 1
- 235000019786 weight gain Nutrition 0.000 description 1
- 239000000811 xylitol Substances 0.000 description 1
- 235000010447 xylitol Nutrition 0.000 description 1
- HEBKCHPVOIAQTA-SCDXWVJYSA-N xylitol Chemical compound OC[C@H](O)[C@@H](O)[C@H](O)CO HEBKCHPVOIAQTA-SCDXWVJYSA-N 0.000 description 1
- 229960002675 xylitol Drugs 0.000 description 1
Classifications
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07J—STEROIDS
- C07J9/00—Normal steroids containing carbon, hydrogen, halogen or oxygen substituted in position 17 beta by a chain of more than two carbon atoms, e.g. cholane, cholestane, coprostane
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07J—STEROIDS
- C07J53/00—Steroids in which the cyclopenta(a)hydrophenanthrene skeleton has been modified by condensation with a carbocyclic rings or by formation of an additional ring by means of a direct link between two ring carbon atoms, including carboxyclic rings fused to the cyclopenta(a)hydrophenanthrene skeleton are included in this class
Definitions
- the present invention relates to a novel stigmasterol derivative or a pharmaceutically acceptable salt thereof, a method of producing the same, and a composition containing the same to inhibit obesity or to prevent and treat hyperlipidemia.
- the excess body weight has a body mass index (BMI, an index of obesity that is obtained by dividing weight by the square of height) in the range of 25 or more and less than 30.
- BMI body mass index
- the obesity means that the body mass index is in the range of 30 or more.
- Obesity means that fat is excessively accumulated in the body and the ratio of fat to body weight is relatively high. Obesity is classified into simple obesity and secondary obesity (symptomatic obesity).
- sample obesity may be caused by overeating, insufficient exercise, and a reduced basal metabolic rate. Most of the cases that are clinically diagnosed are simple obesity. If simple obesity continues over a long period of time, various types of disorders may occur.
- Secondary obesity is caused by certain basis diseases and examples thereof may include endocrine obesity, hypothalamus obesity, hereditary obesity, or obesity resulting from medicine.
- the main cause of obesity is due to the excessive growth of adipocytes, and the excessive growth of adipocytes is caused by the lack of hormone, which is called leptin that is secreted by the order of the brain.
- the lack of leptin hormone is caused by overeating, reduced metabolism in the living body due to aging, and animal cholesterol that is excessively accumulated in the body, with the exception of the case of the hereditary leptin hormone deficiency.
- Cholesterol includes small spherical lipoprotein particles in the blood. Lipoprotein is classified into various types. Lipoprotein that carries cholesterol from the liver to the other tissues is called low density lipoprotein (LDL), and lipoprotein that carries cholesterol from the other tissues to the liver is called high density lipoprotein (HDL). Particularly, HDL-cholesterol functions to clean the blood vessel to reduce arteriosclerosis. Thus, in the case when the content of HDL-cholesterol is low, the occurrence of arteriosclerosis is increased similar to the case when the content of LDL- cholesterol is high, which is considered to be unhealthy.
- LDL low density lipoprotein
- HDL high density lipoprotein
- Obesity incurs diseases such as hyperlipidemia, hypertension, cardiovascular disease, pseudotumor cerebri, sleep apnea, cancer, pulmonary hypertension, cholecystitis, and osteoarthritis, or reduces movement. Furthermore, obesity may be a complication resulting from various types of diseases.
- obesity occurs when ingestion energy continues while being more than consumption energy. Some obese patients make an effort to reduce body weight and cholesterol by diet therapy and exercise therapy for the purpose of treating obesity. Additionally, patients are subjected to behavioral treatment, psychotherapy, drug treatment, or bariatric surgery (a surgical operation such as gastric banding).
- Det therapy is a method of controlling the total energy intake.
- diet therapy is problematic in that since a metabolic rate is reduced during rest, it is difficult to ensure a sufficient weight reduction.
- weight other than fat weight is reduced, such that physical and mental pains such as malnutrition disorder, hunger, or stress occur.
- Behavioral treatment or psychotherapy is used as an auxiliary means of diet therapy or exercise therapy, and not considered as a main treatment to obtain the desired effect.
- the obese person do a low-impact exercise such as swimming.
- medicine that has a low side effect and activation of energy metabolism may be used while the patient does a low-impact exercise such as swimming to increase muscle mass and promote consumption of energy due to an increase in basal metabolism, thereby reducing the possibility of obesity.
- stigmasterol is phytosterol that is contained in soybean milk and has a structure similar to that of animal cholesterol.
- stigmasterol is absorbed in the guts while competing with animal cholesterol to reduce the concentration of cholesterol in the body and suppress the excessive growth of adipocytes.
- calcium phosphate is accumulated in the joints or muscles and muscular atrophy occurs.
- Glucosamine is one of natural amino sugars that constitute the joints and the cartilages and its molecular formula is C H NO .
- Glucosamine has 6 carbon atoms,
- glucosamine and galactosamine are generically known as hexosamine.
- Glucosamine is a strong basic material that has colorless needle crystals, and is decomposed at 11O 0 C and dissolved in water.
- Natural glucosamine is present in a chitin form or a polysaccharide form such as mucopolysaccharides constituting the cell walls of bacteria or the cartilages or skins of animals.
- a large amount of proteins that are bonded to glucosamine are contained in blood or mucus of humans, and glycolipid that is bonded to glucosamine is present in the cell membrane of a red blood cell.
- chitin may be decomposed using a hydrochloric acid to produce glucosamine.
- glucosamine prevents acetate from being provided into the cell to suppress formation of acetyl GoA. As a result, the synthesis of fats and cholesterol is obstructed.
- Snce glucosamine is known as a material that helps make the joint and the cartilage strong, it is extensively used in medicines for degenerative arthritis, cosmetic compositions, foods, and the like.
- the present inventors have conducted studies on a compound capable of reducing the amount of cholesterol and preventing adipocytes from growing in the body, which has resulted in the finding of a novel stigmasterol derivative produced by the present inventors that reduces the contents of triglyceride, total cholesterol, and LDL-cholesterol in the blood, increases the content of HDL- cholesterol, and prevents body weight from being increased, thereby accomplishing the present invention.
- the present invention provides a novel stigmasterol derivative or a pharmaceutically acceptable salt thereof, a method of producing the same, and a composition containing the same to inhibit obesity or to prevent and treat hyperlipidemia. Best Mode for Carrying Out the Invention
- a novel stigmasterol derivative represented by the following Formula 1 or a pharmaceutically acceptable salt thereof is provided.
- the compound of the present invention may be used m the form of a pharmaceutically acceptable salt and a solvate according to the conventional method m the related art.
- acid addition salts produced with free acids are preferred.
- the acid addition salts are produced by the conventional method, for example, a method comprising the steps of dissolving a compound in an excessive amount of acid aqueous solution, and precipitating the salt using water-miscible organic solvents such as methanol, ethanol, acetone or acetomtrile.
- Acid or alcohol for example, glycol monomethyl ether
- the free acids organic acids and inorganic acids may be used.
- Examples of the inorganic acids include hydrochloric acid, phosphoric acid, sulfuric acid, nitric acid, and tartaric acid
- examples of the organic acids include methanesulfonic acid, p-toluenesulfonic acid, acetic acid, trifluoroacetic acid, citric acid, maleic acid, succinic acid, oxalic acid, benzoic acid, tartaric acid, fumaric acid, mandelic acid, propionic acid, lactic acid, glycollic acid, gluconic acid, galacturonic acid, glutamic acid, glutaric acid, glucuronic acid, aspartic acid, ascorbic acid, vanillic acid, and hydroiodic acid, but are not limited thereto.
- a pharmaceutically acceptable metal salt can be produced using a base.
- An alkali metal salt and alkaline earth metal salt can be obtained by a method, in which a compound is dissolved in an excessive amount of alkali metal hydroxide or alkaline earth metal hydroxide solution, filtered the undissolved salt, and then the filtrate is evaporated and dried.
- metal salts it is preferable that sodium, potassium, or calcium salt is pharmaceutically preferable, and the corresponding silver salt is obtained by reacting alkali metal salt or alkaline earth metal salt with a suitable silver salt (e.g. silver nitrate).
- a pharmaceutically acceptable salt of the compound represented by Formula 1 includes salts of acidic or basic groups, which can be present in the compound of Formula 1 unless otherwise specified.
- the pharmaceutically acceptable salt includes sodium salt, calcium salt, potassium salt of a hydroxy group
- other pharmaceutically acceptable salt of an amino group includes hydrochloride, hy- drobromide, sulfate, hydrogen sulfate, phosphate, hydrogen phosphate, dihydrogen phosphate, acetate, succinate, citrate, tartrate, lactate, mandelate, methanesulfonate (mesylate), and p-toluenesulfonate (tosylate).
- the salts can be produced by a known method for producing a salt or a production process in the related art.
- an intermediate compound of the stigmasterol derivative represented by the following Formula 3 or 4 or a pharmaceutically acceptable salt thereof is provided.
- the method of producing the stigmasterol derivative of the present invention comprises the steps of: [40] 1) producing the compound of Formula 3 by reacting stigmasterol and the compound of Formula 2, [41] 2) producing the compound of Formula 4 by reacting the compound of Formula 3 produced in step 1) and an ammonia aqueous solution, and [42] 3) producing the compound of Formula 1 by reacting the compound of Formula 4 produced in step 2) and palladium hydroxide [Pd(OH) ]. [43] [Reaction Scheme 1]
- the compound of Formula 2 as a starting material can be produced by the method described in Chemical. Reviews 1993, Vol. 93, No. 4, p. 1511 or the like or the similar method thereto, and used.
- step 1) stigmasterol and the compound of Formula 2 are dissolved in an organic solvent, and then BF OEt was added thereto. The mixture is subjected to reaction at normal temperature for 10-20 hours to produce the compound of Formula 3.
- the organic solvent is preferably selected from the group consisting of dimethyl ether, tetrahydrofuran, methylene chloride, methanol, cyclohexene, acetonitrile, dimethylformamide, and dimethyl sulfoxide, but is not limited thereto.
- step 2) the compound of Formula 3 produced in step 1) is dissolved in an organic solvent, and reacted with the ammonia aqueous solution in a water bath at 30 ⁇ 50°C for 10-20 hours to produce the compound of Formula 4.
- the organic solvent can be selected from the organic solvents used in step 1).
- the base is preferably selected from the group consisting of sodium hydride, potassium hydride, sodium hydroxide, potassium hydroxide, sodium carbonate, potassium carbonate, cesium carbonate, sodium bicarbonate, potassium bicarbonate, tri- ethylamme, pyridine, DBU, and ammonium hydroxide, but is not limited thereto.
- step 3 the compound of Formula 4 produced in step 2) is dissolved in an organic solvent and distilled water, and reacted with palladium hydroxide [Pd(OH) ] to produce the compound of Formula 1.
- the organic solvent used herein can be selected from the organic solvents used in step 1).
- composition containing the stigmasterol derivative represented by Formula 1 or the pharmaceutically acceptable salt thereof to inhibit obesity or to prevent and treat hyper- lipidemia is provided.
- the stigmasterol derivative according to the present invention has the excellent effects of decreasing blood levels of triglyceride, total cholesterol, and LDL cholesterol, increasing blood level of HDL-cholesterol, and inhibiting body weight gain.
- toxicity test was performed by orally administering the stigmasterol derivative according to the invention to a mouse, resulting in a Lethal Ebse 50 (LD )
- the stigmasterol derivative according to the present invention can be used as a medicine and as health food useful for inhibiting obesity or for preventing and treating hyperlipidemia.
- composition of the invention may contain at least one kind of active ingredient known in the art, which has the effect of inhibiting obesity or preventing or treating hyperlipidemia, in addition to the compound of Formula 1.
- the composition of the invention can include at least one pharmaceutically acceptable carrier, in addition to the active ingredients as described above.
- the pharmaceutically acceptable carrier include saline solution, sterile water, Ringer's solution, buffered saline solution, dextrose solution, maltodextrin solution, glycerol, ethanol and a mixture of one or more thereof.
- the composition may also contain other conventional additives such as antioxidants, buffers, and bacteriostatic agents.
- the composition may additionally contain diluents, dispersants, surfactants, binders, and lubricants in order to formulate it into injectable formulations such as aqueous solution, suspension, and emulsion, pills, capsules, granules, and tablets.
- injectable formulations such as aqueous solution, suspension, and emulsion, pills, capsules, granules, and tablets.
- the composition may preferably be formulated depending on particular diseases and its components, using the method described in Remington's Pharmaceutical Science (latest edition), Mack Publishing Cbmpany, Easton PA, which is a suitable method in the relevant field of art.
- composition of the invention may be administered orally or parenterally (for example, intravein, subcutaneous, intraperitoneal, or topical application).
- the dosage of the composition of the invention can vary depending on various factors, including the patient's weight, age, sex, health condition, and diet, and administration time, administration route, secretion rate, disease severity, etc.
- the compound of Formula 1 is administered at a daily dosage of about 5-30 mg/kg, preferably 10-25 mg/kg one time or several times.
- composition of the invention may be used alone or in combination with surgical operations, hormone therapies, chemical therapies, and other methods using biological reaction regulators, in order to inhibit obesity or to prevent or treat hyperlipidemia.
- the composition of the present invention can be added to a health food for the purpose of improving the diseases caused by obesity or hyperlipidemia.
- the compound of Formula 1 of the invention can be added as it is or with other foods or food ingredients, and suitably used according to the conventional method.
- the mixed amount of active ingredient can be suitably determined depending on its purpose (prevention, health, or treatment).
- the compound of Formula 1 of the invention is added 15% by weight or less, preferably 10% by weight or less, based on a raw material.
- the amount may be less than the above range. Snce there is no problem in safety, the active ingredient may be used in an amount more than the above range.
- the kind of food is not particularly limited.
- Examples of the food, to which the substance can be added include meat, sausage, bread, chocolate, candy, snack, cookie, pizza, instant noodle, other noodles, chewing gum, dairy products including icecream, various kinds of soup, beverage, tea, drink, alcoholic beverages and vitamin complex, and includes all kinds of typical health foods.
- the health beverage composition of the present invention may contain various flavors, natural carbohydrates or the like as an additional ingredient, similarly to a conventional beverage.
- natural carbohydrates include monosaccharides such as glucose, fructose, disaccharides such as maltose, sucrose, polysaccharides such as dextrin and cyclodextrin, sugaralcohols such as xylitol, sorbitol, erythritol.
- sweeteners include natural sweeteners such as thaumatin and stevioside, and artificial sweeteners such as saccharin and aspartame.
- the content of the natural carbohydrate is generally about 0.01-0.04 g, preferably about 0.02-O.03 g, based on 100 m£ of the composition of the present invention.
- the composition of the invention may contain various nutrients, vitamins, minerals, flavoring agent, coloring agent, pectic acid and a salt thereof, alginic acid and a salt thereof, organic acid, protective colloid thickener, pH control agent, stabilizer, preservative, glycerin, alcohol, carbonating agent used in carbonated beverages or the like, in addition to the above-mentioned. Further, the composition of the invention may contain fruit fleshes for production of natural fruit juice, fruit juice beverage, and vegetable beverage. The above ingredients can be used alone or in combination therewith. Generally, the content of the additives is, but not considered significant, 0.01-O.l parts by weight, based on 100 parts by weight of the composition of the present invention. Mode for the Invention
- LDL-cholesterol [Total cholesterol - HDL-cholesterol - (triglyceride / 5)]
- mice Sx-week-old ICR mice were used as test animals. The mice were allowed to freely ingest solid feed and water at the temperature of 23+1 0 C and the humidity of 60+5% before the test was performed. The test animals were divided into groups, and six mice were included in each group. The compound 1 that was produced in Example 1 was suspended in a 0.5% methyl cellulose solution and then orally administered in the amount of 1 g/kg/15 mH once. After the test material was administered, mice were observed for 14 days in views of appearance and death. The dead mice were subjected to an autopsy and observed in terms of grosslesions. The LD value was obtained by using the Richfield- Wilcoxon method.
- the compound of the present invention does not incur a change in toxicity until the amount is increased to 3000 mg/kg in respects to all the mice. Additionally, the compound is evaluated as a safe substance having the oral administration lethal dose 50 (LD ) of 3000 mg/kg or more.
- LD lethal dose 50
- PREPARATION EXAMPLE 2 Production of foods [133] Foods that contain the compound of Formula 1 according to the present invention were produced by using the following methods. [ 134] 1. Production of seasonings [135] 20-95 % by weight of compound of Formula 1 was used to produce seasonings that were healthy.
- Sde materials such as fructose liquid (0.5%), oligosaccharide (2%), sugar (2%), common salt (0.5%), and water (75%) and the compound of Formula 1 were homogeneously mixed with each other and instantaneously sterilized.
- the sterilized mixture was packed by using small-sized vessels such as glass bottles and pet bottles to produce beverages that were healthy.
- the stigmasterol derivative according to the present invention is advantageous in that the stigmasterol derivative reduces the contents of triglyceride, total cholesterol, and LDL-cholesterol in blood, increases the content of HDL-cholesterol, and suppresses an increase in body weight. Accordingly, it is possible to use the stigmasterol derivative according to the present invention as medicines and health foods that are useful to suppress obesity or to prevent and treat hyperlipidemia.
Abstract
The present invention relates to a novel stigmasterol derivative or a pharmaceutically acceptable salt thereof, a method of producing the same, and a composition comprising the same for inhibiting obesity or for preventing and treating hyperlipidemia. The stigmasterol derivative according to the present invention is advantageous in that the stigmasterol derivative reduces the contents of triglyceride, total cholesterol, and LDL-cholesterol in blood, increases the content of HDL-cholesterol, and suppresses an increase in body weight. Accordingly, it is possible to use the stigmasterol derivative according to the present invention as medicines and health foods that are useful to suppress obesity or to prevent and treat hyperlipidemia.
Description
Description
NOVEL STIGMASTEROL DERIVATIVE OR PHARMACEUTICALLY ACCEPTABLE SALT THEREOF, METHOD OF PRODUCING THE SAME, AND COMPOSITION CONTAINING THE SAME TO INHIBIT OBESITY OR TO PREVENT AND TREAT HYPERLIPIDEMIA Technical Field
[1] The present invention relates to a novel stigmasterol derivative or a pharmaceutically acceptable salt thereof, a method of producing the same, and a composition containing the same to inhibit obesity or to prevent and treat hyperlipidemia. Background Art
[2] In recent years, due to growth in the economy and changes in life style, dietary habits have significantly changed. In particular, excess body weight and obesity due to ingestion of high calorie food such as fastfood and insufficient exercise have considerably increased.
[3] The excess body weight has a body mass index (BMI, an index of obesity that is obtained by dividing weight by the square of height) in the range of 25 or more and less than 30. The obesity means that the body mass index is in the range of 30 or more.
[4] Two-thirds of Americans suffer from excess body weight or obesity. Excess body weight increases blood pressure and the amount of cholesterol, which causes various types of diseases in adults such as cardiac disorders, and obesity increases heart diseases, diabetes, arthritis, and some cancer.
[5] Excess body weight and obesity act as factors that increase the rate of occurrence of various types of diseases in adults such as arteriosclerosis, hypertension, hyperlipidemia or cardiac disorders in children or teenagers as well as adults.
[6] Obesity affects all age groups, and frequently occurs in the prime of life. It is known that obesity does not frequently occur in Asia due to their dietary habit of eating vegetables. However, currently, dietary habits and a residence environment are changing in Asia due to an influence of the Western civilization, thus increasing obesity.
[7] In addition, the modern people have reduced strength due to overworking excessive drinking, stress, or the like, and it is known that about 30-40% of them are obese and about 10% of them are seriously obese.
[8] Obesity means that fat is excessively accumulated in the body and the ratio of fat to body weight is relatively high. Obesity is classified into simple obesity and secondary obesity (symptomatic obesity).
[9] Smple obesity may be caused by overeating, insufficient exercise, and a reduced basal metabolic rate. Most of the cases that are clinically diagnosed are simple obesity. If simple obesity continues over a long period of time, various types of disorders may occur.
[10] Secondary obesity is caused by certain basis diseases and examples thereof may include endocrine obesity, hypothalamus obesity, hereditary obesity, or obesity resulting from medicine.
[11] The main cause of obesity is due to the excessive growth of adipocytes, and the excessive growth of adipocytes is caused by the lack of hormone, which is called leptin that is secreted by the order of the brain. The lack of leptin hormone is caused by overeating, reduced metabolism in the living body due to aging, and animal cholesterol that is excessively accumulated in the body, with the exception of the case of the hereditary leptin hormone deficiency.
[12] Cholesterol includes small spherical lipoprotein particles in the blood. Lipoprotein is classified into various types. Lipoprotein that carries cholesterol from the liver to the other tissues is called low density lipoprotein (LDL), and lipoprotein that carries cholesterol from the other tissues to the liver is called high density lipoprotein (HDL). Particularly, HDL-cholesterol functions to clean the blood vessel to reduce arteriosclerosis. Thus, in the case when the content of HDL-cholesterol is low, the occurrence of arteriosclerosis is increased similar to the case when the content of LDL- cholesterol is high, which is considered to be unhealthy.
[13] Obesity incurs diseases such as hyperlipidemia, hypertension, cardiovascular disease, pseudotumor cerebri, sleep apnea, cancer, pulmonary hypertension, cholecystitis, and osteoarthritis, or reduces movement. Furthermore, obesity may be a complication resulting from various types of diseases.
[14] Generally, obesity occurs when ingestion energy continues while being more than consumption energy. Some obese patients make an effort to reduce body weight and cholesterol by diet therapy and exercise therapy for the purpose of treating obesity. Additionally, patients are subjected to behavioral treatment, psychotherapy, drug treatment, or bariatric surgery (a surgical operation such as gastric banding).
[15] Det therapy is a method of controlling the total energy intake. However, diet therapy is problematic in that since a metabolic rate is reduced during rest, it is difficult to
ensure a sufficient weight reduction. In addition, even if the sufficient weight reduction is ensured, weight other than fat weight is reduced, such that physical and mental pains such as malnutrition disorder, hunger, or stress occur.
[16] In the case of exercise therapy, consumption energy is increased, the metabolic rate is improved during rest, resistance to insulin is avoided, and fat is reduced from the body. However, since it is necessary to do an aerobic exercise for 20 min per time and repeat the exercise three times or more a week, it is difficult to consistently maintain an exercise regimen.
[17] Behavioral treatment or psychotherapy is used as an auxiliary means of diet therapy or exercise therapy, and not considered as a main treatment to obtain the desired effect.
[18] In the case where the above-mentioned treatments are not useful or urgent treatment needs to be used because of serious obesity, drug treatment or bariatric surgery is used. However, bariatric surgery imposes a very heavy burden on a patient. Examples of medicine that is used to treat obesity by using the drug treatment include appetite depressants, digestion and absorption inhibitors, obesity inhibitors, or metabolism promotion agents. Furthermore, the medicine may incur a side effect such as drug dependence. Snce a patient may have a tolerance to medicine over a short period of time, it is difficult to perform the treatment over a long period of time.
[19] Additionally, in the case where exercise therapy is used, if an obese person does a high-impact exercise, the possibility of damage to underlying joints is increased. Therefore, it is preferable that the obese person do a low-impact exercise such as swimming. In this case, preferably, medicine that has a low side effect and activation of energy metabolism may be used while the patient does a low-impact exercise such as swimming to increase muscle mass and promote consumption of energy due to an increase in basal metabolism, thereby reducing the possibility of obesity.
[20] Meanwhile, stigmasterol is phytosterol that is contained in soybean milk and has a structure similar to that of animal cholesterol. However, stigmasterol is absorbed in the guts while competing with animal cholesterol to reduce the concentration of cholesterol in the body and suppress the excessive growth of adipocytes. In the case of stigmasterol deficiency, calcium phosphate is accumulated in the joints or muscles and muscular atrophy occurs.
[21] Glucosamine is one of natural amino sugars that constitute the joints and the cartilages and its molecular formula is C H NO . Glucosamine has 6 carbon atoms,
6 13 6 and glucosamine and galactosamine are generically known as hexosamine. Glucosamine is a strong basic material that has colorless needle crystals, and is
decomposed at 11O0C and dissolved in water. Natural glucosamine is present in a chitin form or a polysaccharide form such as mucopolysaccharides constituting the cell walls of bacteria or the cartilages or skins of animals. A large amount of proteins that are bonded to glucosamine are contained in blood or mucus of humans, and glycolipid that is bonded to glucosamine is present in the cell membrane of a red blood cell. Furthermore, chitin may be decomposed using a hydrochloric acid to produce glucosamine. During biosynthesis of glucosamine, glucosamine prevents acetate from being provided into the cell to suppress formation of acetyl GoA. As a result, the synthesis of fats and cholesterol is obstructed. Snce glucosamine is known as a material that helps make the joint and the cartilage strong, it is extensively used in medicines for degenerative arthritis, cosmetic compositions, foods, and the like. [22] Currently, the increase in obesity has become a social concern, and there is a need to develop goods that are capable of significantly reducing the amount of cholesterol and preventing adipocytes from growing in the body and efficiently reducing the weight unlike known stigmasterol or glucosamine. Disclosure of Invention Technical Problem
[23] With respect to this, the present inventors have conducted studies on a compound capable of reducing the amount of cholesterol and preventing adipocytes from growing in the body, which has resulted in the finding of a novel stigmasterol derivative produced by the present inventors that reduces the contents of triglyceride, total cholesterol, and LDL-cholesterol in the blood, increases the content of HDL- cholesterol, and prevents body weight from being increased, thereby accomplishing the present invention. Technical Solution
[24] The present invention provides a novel stigmasterol derivative or a pharmaceutically acceptable salt thereof, a method of producing the same, and a composition containing the same to inhibit obesity or to prevent and treat hyperlipidemia. Best Mode for Carrying Out the Invention
[25] According to an embodiment of the present invention, a novel stigmasterol derivative represented by the following Formula 1 or a pharmaceutically acceptable salt thereof is provided.
[28] The compound of the present invention may be used m the form of a pharmaceutically acceptable salt and a solvate according to the conventional method m the related art.
[29] As the pharmaceutically acceptable salt, acid addition salts produced with free acids are preferred. The acid addition salts are produced by the conventional method, for example, a method comprising the steps of dissolving a compound in an excessive amount of acid aqueous solution, and precipitating the salt using water-miscible organic solvents such as methanol, ethanol, acetone or acetomtrile. Acid or alcohol (for example, glycol monomethyl ether) in the same molar amount of compound and water is heated, and the mixture is dried by evaporation or the precipitated salt can be suction-filtered. At this time, as the free acids, organic acids and inorganic acids may be used. Examples of the inorganic acids include hydrochloric acid, phosphoric acid, sulfuric acid, nitric acid, and tartaric acid, and examples of the organic acids include methanesulfonic acid, p-toluenesulfonic acid, acetic acid, trifluoroacetic acid, citric acid, maleic acid, succinic acid, oxalic acid, benzoic acid, tartaric acid, fumaric acid, mandelic acid, propionic acid, lactic acid, glycollic acid, gluconic acid, galacturonic acid, glutamic acid, glutaric acid, glucuronic acid, aspartic acid, ascorbic acid, vanillic acid, and hydroiodic acid, but are not limited thereto.
[30] Further, a pharmaceutically acceptable metal salt can be produced using a base. An alkali metal salt and alkaline earth metal salt can be obtained by a method, in which a compound is dissolved in an excessive amount of alkali metal hydroxide or alkaline earth metal hydroxide solution, filtered the undissolved salt, and then the filtrate is evaporated and dried. In respects to metal salts, it is preferable that sodium, potassium, or calcium salt is pharmaceutically preferable, and the corresponding silver salt is obtained by reacting alkali metal salt or alkaline earth metal salt with a suitable silver salt (e.g. silver nitrate).
[31] A pharmaceutically acceptable salt of the compound represented by Formula 1 includes salts of acidic or basic groups, which can be present in the compound of Formula 1 unless otherwise specified. For example, the pharmaceutically acceptable
salt includes sodium salt, calcium salt, potassium salt of a hydroxy group, and other pharmaceutically acceptable salt of an amino group includes hydrochloride, hy- drobromide, sulfate, hydrogen sulfate, phosphate, hydrogen phosphate, dihydrogen phosphate, acetate, succinate, citrate, tartrate, lactate, mandelate, methanesulfonate (mesylate), and p-toluenesulfonate (tosylate). Further, the salts can be produced by a known method for producing a salt or a production process in the related art.
[32] In another embodiment of the present invention, an intermediate compound of the stigmasterol derivative represented by the following Formula 3 or 4 or a pharmaceutically acceptable salt thereof is provided.
[33] [Formula 3]
[35] [Formula 4]
[37]
[38] According to another embodiment of the present invention, a method of producing the stigmasterol derivative represented by the above Formula 1 is provided. [39] As shown in the following Reaction Scheme 1, the method of producing the stigmasterol derivative of the present invention comprises the steps of: [40] 1) producing the compound of Formula 3 by reacting stigmasterol and the compound of Formula 2, [41] 2) producing the compound of Formula 4 by reacting the compound of Formula 3 produced in step 1) and an ammonia aqueous solution, and [42] 3) producing the compound of Formula 1 by reacting the compound of Formula 4 produced in step 2) and palladium hydroxide [Pd(OH) ]. [43] [Reaction Scheme 1]
[45] In Reaction Scheme 1, the compound of Formula 2 as a starting material can be produced by the method described in Chemical. Reviews 1993, Vol. 93, No. 4, p. 1511 or the like or the similar method thereto, and used.
[46] In step 1), stigmasterol and the compound of Formula 2 are dissolved in an organic solvent, and then BF OEt was added thereto. The mixture is subjected to reaction at normal temperature for 10-20 hours to produce the compound of Formula 3.
[47] The organic solvent is preferably selected from the group consisting of dimethyl ether, tetrahydrofuran, methylene chloride, methanol, cyclohexene, acetonitrile, dimethylformamide, and dimethyl sulfoxide, but is not limited thereto.
[48] In step 2), the compound of Formula 3 produced in step 1) is dissolved in an organic solvent, and reacted with the ammonia aqueous solution in a water bath at 30~50°C for 10-20 hours to produce the compound of Formula 4.
[49] The organic solvent can be selected from the organic solvents used in step 1). The base is preferably selected from the group consisting of sodium hydride, potassium hydride, sodium hydroxide, potassium hydroxide, sodium carbonate, potassium carbonate, cesium carbonate, sodium bicarbonate, potassium bicarbonate, tri- ethylamme, pyridine, DBU, and ammonium hydroxide, but is not limited thereto.
[50] In step 3), the compound of Formula 4 produced in step 2) is dissolved in an organic solvent and distilled water, and reacted with palladium hydroxide [Pd(OH) ] to produce the compound of Formula 1. The organic solvent used herein can be selected from the organic solvents used in step 1).
[51]
[52] According to still another embodiment of the present invention, a composition containing the stigmasterol derivative represented by Formula 1 or the pharmaceutically acceptable salt thereof to inhibit obesity or to prevent and treat hyper- lipidemia is provided.
[53] The stigmasterol derivative according to the present invention has the excellent effects of decreasing blood levels of triglyceride, total cholesterol, and LDL cholesterol, increasing blood level of HDL-cholesterol, and inhibiting body weight gain.
[54] Further, toxicity test was performed by orally administering the stigmasterol derivative according to the invention to a mouse, resulting in a Lethal Ebse 50 (LD )
50 after oral administration of 3000 mg/kg or more, which indicates that the stigmasterol derivative is a safe substance.
[55] Accordingly, the stigmasterol derivative according to the present invention can be used as a medicine and as health food useful for inhibiting obesity or for preventing and treating hyperlipidemia.
[56] The composition of the invention may contain at least one kind of active ingredient known in the art, which has the effect of inhibiting obesity or preventing or treating hyperlipidemia, in addition to the compound of Formula 1.
[57] For administration, the composition of the invention can include at least one pharmaceutically acceptable carrier, in addition to the active ingredients as described above. Examples of the pharmaceutically acceptable carrier include saline solution, sterile water, Ringer's solution, buffered saline solution, dextrose solution, maltodextrin solution, glycerol, ethanol and a mixture of one or more thereof. If necessary, the composition may also contain other conventional additives such as antioxidants, buffers, and bacteriostatic agents. Moreover, the composition may additionally contain diluents, dispersants, surfactants, binders, and lubricants in order to formulate it into injectable formulations such as aqueous solution, suspension, and emulsion, pills, capsules, granules, and tablets. Furthermore, the composition may preferably be formulated depending on particular diseases and its components, using the method described in Remington's Pharmaceutical Science (latest edition), Mack Publishing
Cbmpany, Easton PA, which is a suitable method in the relevant field of art.
[58] The composition of the invention may be administered orally or parenterally (for example, intravein, subcutaneous, intraperitoneal, or topical application). The dosage of the composition of the invention can vary depending on various factors, including the patient's weight, age, sex, health condition, and diet, and administration time, administration route, secretion rate, disease severity, etc. The compound of Formula 1 is administered at a daily dosage of about 5-30 mg/kg, preferably 10-25 mg/kg one time or several times.
[59] The composition of the invention may be used alone or in combination with surgical operations, hormone therapies, chemical therapies, and other methods using biological reaction regulators, in order to inhibit obesity or to prevent or treat hyperlipidemia.
[60] The composition of the present invention can be added to a health food for the purpose of improving the diseases caused by obesity or hyperlipidemia. In the case of using the compound of Formula 1 of the invention as a food additive, the compound of Formula 1 can be added as it is or with other foods or food ingredients, and suitably used according to the conventional method. The mixed amount of active ingredient can be suitably determined depending on its purpose (prevention, health, or treatment). Generally, during the production of food or beverage, the compound of Formula 1 of the invention is added 15% by weight or less, preferably 10% by weight or less, based on a raw material. However, in the case of taking over a long period of time for the purpose of health and hygiene or for the purpose of controlling health, the amount may be less than the above range. Snce there is no problem in safety, the active ingredient may be used in an amount more than the above range.
[61] The kind of food is not particularly limited. Examples of the food, to which the substance can be added, include meat, sausage, bread, chocolate, candy, snack, cookie, pizza, instant noodle, other noodles, chewing gum, dairy products including icecream, various kinds of soup, beverage, tea, drink, alcoholic beverages and vitamin complex, and includes all kinds of typical health foods.
[62] The health beverage composition of the present invention may contain various flavors, natural carbohydrates or the like as an additional ingredient, similarly to a conventional beverage. Examples of the natural carbohydrates include monosaccharides such as glucose, fructose, disaccharides such as maltose, sucrose, polysaccharides such as dextrin and cyclodextrin, sugaralcohols such as xylitol, sorbitol, erythritol. Examples of the sweeteners include natural sweeteners such as thaumatin and stevioside, and artificial sweeteners such as saccharin and aspartame. The content of
the natural carbohydrate is generally about 0.01-0.04 g, preferably about 0.02-O.03 g, based on 100 m£ of the composition of the present invention.
[63] The composition of the invention may contain various nutrients, vitamins, minerals, flavoring agent, coloring agent, pectic acid and a salt thereof, alginic acid and a salt thereof, organic acid, protective colloid thickener, pH control agent, stabilizer, preservative, glycerin, alcohol, carbonating agent used in carbonated beverages or the like, in addition to the above-mentioned. Further, the composition of the invention may contain fruit fleshes for production of natural fruit juice, fruit juice beverage, and vegetable beverage. The above ingredients can be used alone or in combination therewith. Generally, the content of the additives is, but not considered significant, 0.01-O.l parts by weight, based on 100 parts by weight of the composition of the present invention. Mode for the Invention
[64] Hereinafter, the preferred Examples are provided for better understanding. However, these Examples are for the illustrative purpose only, and the invention is not intended to be limited by these Examples.
[65] EXAMPLE 1 : Production of stigmasterol derivative
[66] 1. Production of acetic acid
4-acetoxy-2-acetoxymethyl-5-benzyloxycarbonylamino-6-[17-(4-ethyl-l,5-dimethy l-2-hexenyl)-10,13-dimethyl-2,3,4,7,8,9,10,ll,12,13,14,15,16,17-tetradecahydro-lH -cydopenta[a]phenanthren-3-yloxy] -tetrahydro-pyran-3-yl ester (3)
[68] Acetic acid
4-acetoxy-2-acetoxymethyl-5-benzyloxycarbonylamino-6-(2,2,2-trichloroacetimidoylo xy)-tetrahydro-pyran-3-yl ester (10 g, 0.0171 mol) and stigmasterol (7.0 g, 0.0171 mol) were dissolved in 250 m# of dimethyl ether, 0.69 mH of BF OEt was added
3 2 thereto, and the mixture was stirred at normal temperature for 16 hours. After the reaction was completed, the reaction solvent was removed to obtain a concentrate. After the concentrate was dissolved in methylene chloride, separation was performed by means of silica gel column chromatography using a solvent mixture of ethyl acetate
: hexane (1:3) to obtain the compound (3). [69] Yield : 21%,
[70] Melting point : 165-1670C,
[71] 1H-NMR(SOOMHz, CDCl ) : 0.68-2.28(43H, m), 1.97(3H,s), 2.01(3H,s), 2.07(3H,s),
3
3.47(2H,m), 3.61(lH,m), 4.10(lH,d), 4.25(lH,m), 4.8O(1H, m), 5.02(2H,t), 5.11(3H,m), 5.30(lH,s), 5.35(lH,m), 7.33(5H,s)
[72] 2. Production of
{2-[17-(4-ethyl-l,5-dimethyl-2-hexenyl)-10,13-dimethyl-2,3,4,7,8,9,10,ll,12,13,14,l 5,16,17,-tetradecahydro-lH-cyclopenta[a]phenanthren-3-yloxyl]-4,5-dihydro-6-hy droxymethyl-tetrahydro-pyran-S-ylj-carbamic acid benzyl ester (4)
[74] Acetic acid
4-acetoxy-2-acetoxymethyl-5-benzyloxycarbonylamino-6-[17-(4-methyl-l,5-dimethyl- 2-hexenyl)-10,13-dimethyl-2,3,4,7,8,9,10,l l,12,13,14,15,16,17-tetradecahydro-lH-cyc lopenta[a]phenanthren-3-yloxy]-tetrahydro-pyran-3-yl ester (5.0 g, 0.006 mol) that was produced in the above 1 was dissolved in 30 mt of methylene chloride, 80 m£ of methanol was added, 25 m£ of ammonia aqueous solution was added, and the resulting solution was stirred in a water bath at 400C for 16 hours while the stopper was put on the reactor. After the reaction, produced solids were filtered, washed with water, and dried to obtain the compound (4).
[75] Yield : 85%,
[76] Melting point : 193-1950C,
[77] 1H-NMR(SOOMHz, CDCl ) : 0.68-2.26(43H, m), 3.29(2H,m), 3.42(1H, m),
3
3.5-3.8(3H,m), 4.58(2H,m), 5.10(3H,s), 5.28(2H,s), 6.10(lH,m), 7.30(5H,s) [78] 3. Production of
5-amino-6-[17-(4-ethyl-l,5-dimethyl-hexenyl)-10,13-dimethyl-2,3,4,7,8,9,10,ll,12, 13,14,15,16,17-tetradecahydro-lH-cyclopenta[a]phenanthren-3-yloxy]-2-hydrome thyl-tetrahydro-pyran-3,4-diol (1)
[80] 220 rd of methanol, 110 m# of cyclohexene, and 20 m# of distilled water were sequentially added to {2-[17-(4-ethyl-l,5-dimethyl - hexenyl)-10,13-dimethyl-2,3,4,7,8,9,10,l l,12,13,14,15,16,17-tetradecahydro-lH-cyclo penta[a]phenanthren-3-yloxyl] -4,5-dihydro-6-hydroxymethyl-tetrahydro-pyran-3-yl } -c arbamic acid benzyl ester (2.0 g, 0.0028 mol) that was produced in the above 2, 5.0 g of palladium hydroxide [Pd(OH) ] was added, and reflux was performed for 2 hours while stirring was carried out. After the reaction was completed, palladium was removed by using cellite and the filtered solution was concentrated. The concentrated solution was dissolved in a developing solvent (methylene chloride:methanol = 8:1) and separation was performed by using silica gel column chromatography to obtain the compound (1).
[81] Yield : 55%,
[82] Melting point : 2550C,
[83] 1H-NMR(SOOMHz, CDCl ) : 0.68-2.17(43H, m), 2.42(2H,m), 3.11 (4H,m),
3
3.35(2H,m), 3.69(lH,m), 4.26(2H,m), 4.48(lH,t), 4.96(lH,s), 5.08(1H, m), 5.17(lH,m), 5.37(lH,s)
[84]
[85] EXPERIMENTAL EXAMPLE 1 : Anti-obesity, anti-lipid and anti- cholesterol test
[86] In order to evaluate anti-obesity, anti-lipid and anti-cholesterol abilities of the compound of Formula 1 according to the present invention, the following test was performed.
[87] 30 three- week-old SD male rats that were obtained from Central Lab Animal, Inc. were used as subject animals. In respects to a breeding condition, the temperature was 23+30C, the relative humidity was 55+10%, the number of ventilation was 10-20 ventilation/time, the lighting time of the fluorescent lamp was 12 hours/day (the switch was turned on at 8 a.m. and the switch was turned off at 8 p.m.), and the intensity of illumination was 150~300 Lux. The subject animals were divided into five groups, and the six rats were bred in each group in the polycarbonate breeding box (size : 220 W x
360 L x 200 H mm).
[88] After the subject animals became adapted to new environment for 1 week, the rats were allowed to freely ingest AIN-76A high fat diet (Dytes Inc, USA) as feed for 4 weeks. Subterranean water that was acceptable to drink was sterilized by using a micro drum filter and a UV sterilizer and the rats were allowed to freely drink the sterilized water by using the sterilized water bottle.
[89] While the same feed was given to the rats that became fat for 6 weeks, distilled water was orally administered to the first group (negative control group), 400 rrg/kg of the compound 1 that was produced in Example 1 was orally administered to the second group (test group 1), 800 mg/kg of the compound 1 that was produced in Example 1 was orally administered to the third group (test group 2), 800 mg/kg of stigmas terol that was the control medicine was orally administered to the fourth group (positive control group 1), and 800 mg/kg of glucosamine that was the control medicine was orally administered to the fifth group (positive control group 2).
[90] Before the test was finished, the rats that fasted for 16 hours were anesthetized using dimethyl ether and had their blood taken from the abdominal venae cavae. After the blood was centrifuged, serum was obtained. The contents of triglyceride, total cholesterol, and HDL-cholesterol in the serum were measured by using a quantitative kit reagent (ASAN Pharmaceutical Cb.) according to an enzymatic colorimetric method, and the content of LDL-cholesterol was calculated by using the following Friedewald equation.
[91]
[92] LDL-cholesterol = [Total cholesterol - HDL-cholesterol - (triglyceride / 5)]
[93]
[94] The rats were isolated from each other, and the dietary intake and the weight were measured twice per week during the test.
[95] The content of triglyceride in the blood is described in Table 1 , the content of total cholesterol in the blood is described in Table 2, the content of HDL-cholesterol in the blood is described in Table 3, and the content of LDL-cholesterol in the blood is described in Table 4. In addition, a change in body weight is described in Table 5.
[96] Table 1
[Table 1]
[Table ]
(intent of triglyceride in blood
[97] Table 2 [Table 2] [Table ] Content of total cholesterol in blood
[98] Table 3
[Table 3]
[Table ]
(intent of HDL-cholesterol in blood
[99] Table 4 [Table 4] [Table ] Content of LDL-cholesterol in blood
[100]
[101] As shown in Tables 1 to 4, it could be seen that in the case where the compound according to the present invention was used, the content of triglyceride in the blood was reduced by 30-34%, the content of total cholesterol in the blood was reduced by
10-13%, the content of LDL-cholesterol in the blood was reduced by 10-14%, and the content of HDL-cholesterol in the blood was increased by 1-3%, as the results of biochemical test of blood in comparison with the high fat diet group (negative control group). Thus, it can be seen that the compound according to the present invention significantly improves the blood lipoprotein.
[102] However, in the case of stigmasterol and glucosamine that were the positive control medicines, the contents of triglyceride in the blood were reduced by 7.30% and 10.70%, the contents of total cholesterol in the blood were reduced by 4.90% and 1.90%, the contents of LDL-cholesterol in the blood were reduced by 6.20% and 1.19%, and the contents of HDL-cholesterol in the blood were insignificantly increased in comparison with the high fat diet group (negative control group). Thus, it can be seen that stigmasterol and glucosamine insignificantly improve the blood lipoprotein.
[103] Table 5 [Table 5] [Table ] Change in body weight
[104]
[105] As shown in Table 5, the compound according to the present invention reduces the weight by 7.8 to 12.4% in comparison with the high fat diet group (negative control group). In the case of stigmasterol and glucosamine that were the positive control medicines, the weights were reduced by 1.2% and 3.4% in comparison with the high
fat diet group (negative control group). [106] [107] EXPERIMENTAL EXAMPLE 2 : Acute toxicity test
[108] In order to evaluate acute toxicity of the stigmasterol derivative according to the present invention, the acute toxicity test was performed by using the Richfield- Wilcoxon method.
[109] Sx-week-old ICR mice were used as test animals. The mice were allowed to freely ingest solid feed and water at the temperature of 23+10C and the humidity of 60+5% before the test was performed. The test animals were divided into groups, and six mice were included in each group. The compound 1 that was produced in Example 1 was suspended in a 0.5% methyl cellulose solution and then orally administered in the amount of 1 g/kg/15 mH once. After the test material was administered, mice were observed for 14 days in views of appearance and death. The dead mice were subjected to an autopsy and observed in terms of grosslesions. The LD value was obtained by using the Richfield- Wilcoxon method.
[110] From the test results, it could be seen that there was no clinical symptom in the mice to which the test material was administered, and no dead mice were found.
[I l l] Therefore, the compound of the present invention does not incur a change in toxicity until the amount is increased to 3000 mg/kg in respects to all the mice. Additionally, the compound is evaluated as a safe substance having the oral administration lethal dose 50 (LD ) of 3000 mg/kg or more.
50
[112]
[113] Preparation Examples of the compositions of the present invention are described below. [114] PREPARATION EXAMPLE 1 : Production of pharmaceutical preparation
[115] 1. Production of powder [116] Compound of Formula 1 2 g [117] Lactose 1 g [118] The above-mentioned components were mixed with each other and then packed by using an airtight sheet to produce powder. [119] 2. Production of tablet [120] Compound of Formula 1 100 mg [121] Corn starch 100 mg [122] Lactose 100 mg
[123] Magnesium stearate 2 mg
[124] The above-mentioned components were mixed with each other and then tableted by using a typical production process to produce tablet. [125] 3. Production of capsule preparation [126] Compound of Formula 1 100 mg [127] Corn starch 100 mg [128] Lactose 100 mg [129] Magnesium stearate 2 mg [130] The above-mentioned components were mixed with each other and then packed in a gelatin capsule by using a typical production process of capsule preparation to produce capsule preparation. [131]
[132] PREPARATION EXAMPLE 2 : Production of foods [133] Foods that contain the compound of Formula 1 according to the present invention were produced by using the following methods. [ 134] 1. Production of seasonings [135] 20-95 % by weight of compound of Formula 1 was used to produce seasonings that were healthy.
[136] 2. Production of tomato ketchup and sauce [137] 0.2-1.0 % by weight of compound of Formula 1 was added to tomato ketchup or sauce to produce tomato ketchup or sauce that was healthy. [138] 3. Production of wheat flour foods [139] 0.5~5.0 % by weight of compound of Formula 1 was added to wheat flour and the resulting mixture was used to produce bread, cake, cookies, crackers, and noodles.
Thereby, foods that were healthy were obtained. [140] 4. Production of soup and gravy [141] 0.1~5.0 % by weight of compound of Formula 1 was added to soup and gravy to produce processed meat, and soup and gravy for noodles that were healthy. [ 142] 5. Production of ground beef [143] 10 % by weight of compound of Formula 1 was added to ground beef to produce ground beef that was healthy. [144] 6. Production of dairy products [145] 5~10 % by weight of compound of Formula 1 was added to milk and dairy products such as butter and ice cream were produced by using the milk. [146]
[147] PREPARATION EXAMPLE 3 : Production of beverages
[148] 1. Production of carbonated beverages
[149] Additives such as 5-10% of sugar, 0.05-O.3% of citric acid, 0.005-0.02% of caramel, and 0.1-1% of vitamin C were mixed with each other and 79-94% of purified water was added thereto to produce a syrup. The syrup was sterilized at 85-980C for 20-180 sec and then mixed with cold water at a ratio of 1:4. 0.5-0.82% of carbon dioxide was added to the resulting mixture to produce a carbonated beverage that contained the compound of Formula 1 according to the present invention.
[ 150] 2. Production of beverages that were healthy
[151] Sde materials such as fructose liquid (0.5%), oligosaccharide (2%), sugar (2%), common salt (0.5%), and water (75%) and the compound of Formula 1 were homogeneously mixed with each other and instantaneously sterilized. The sterilized mixture was packed by using small-sized vessels such as glass bottles and pet bottles to produce beverages that were healthy.
[152] 3. Production of vegetable juice
[153] 5 g of compound of Formula 1 was added to 1,000 mi, of tomato or carrot juice to produce vegetable juice that was healthy.
[ 154] 4. Production of fruit juice
[155] 1 g of compound of Formula 1 was added to 1,000 \ύ of apple or grape juice to produce fruit juice that was healthy. Industrial Applicability
[156] The stigmasterol derivative according to the present invention is advantageous in that the stigmasterol derivative reduces the contents of triglyceride, total cholesterol, and LDL-cholesterol in blood, increases the content of HDL-cholesterol, and suppresses an increase in body weight. Accordingly, it is possible to use the stigmasterol derivative according to the present invention as medicines and health foods that are useful to suppress obesity or to prevent and treat hyperlipidemia.
Claims
[1] A stigmasterol derivative represented by Formula 1 or a pharmaceutically acceptable salt thereof. [Formula 1]
[2] An intermediate compound of a stigmasterol derivative represented by Formula 3 or a pharmaceutically acceptable salt thereof. [Formula 3]
[3] An intermediate compound of a stigmasterol derivative represented by Formula 4 or a pharmaceutically acceptable salt thereof. [Formula 4]
[4] A method of producing the compound of claim 1 , which is represented by Reaction Scheme 1, the method comprising the steps of:
1) producing the compound of Formula 3 by reacting stigmasterol and the compound of Formula 2;
2) producing the compound of Formula 4 by reacting the compound of Formula
3 produced in the step 1) and an ammonia aqueous solution; and
3) producing the compound of Formula 1 by reacting the compound of Formula
4 produced in the step 2) and palladium hydroxide [Pd(OH) ].
[Reaction Scheme 1]
[5] A pharmaceutical composition containing the compound of claim 1 to suppress obesity or to prevent and treat hyperlipidemia. [6] A food composition that contains the compound of claim 1 and is useful to suppress obesity or to prevent hyperlipidemia.
Applications Claiming Priority (4)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| KR20060087875 | 2006-09-12 | ||
| KR10-2006-0087875 | 2006-09-12 | ||
| KR10-2007-0091861 | 2007-09-11 | ||
| KR1020070091861A KR100886466B1 (en) | 2006-09-12 | 2007-09-11 | New stigmasterol derivatives or pharmaceutically acceptable salts thereof, process for the preparation thereof and composition comprising the same for inhibiting obesity or for preventing and treating hyperlipidemia |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| WO2008032980A1 true WO2008032980A1 (en) | 2008-03-20 |
Family
ID=39183984
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/KR2007/004395 WO2008032980A1 (en) | 2006-09-12 | 2007-09-11 | Novel stigmasterol derivative or pharmaceutically acceptable salt thereof, method of producing the same, and composition containing the same to inhibit obesity or to prevent and treat hyperlipidemia |
Country Status (2)
| Country | Link |
|---|---|
| KR (1) | KR100886466B1 (en) |
| WO (1) | WO2008032980A1 (en) |
Cited By (3)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2009021740A2 (en) | 2007-08-15 | 2009-02-19 | Sanofis-Aventis | Substituted tetrahydronaphthalenes, process for the preparation thereof and the use thereof as medicaments |
| WO2011157827A1 (en) | 2010-06-18 | 2011-12-22 | Sanofi | Azolopyridin-3-one derivatives as inhibitors of lipases and phospholipases |
| EP2567959A1 (en) | 2011-09-12 | 2013-03-13 | Sanofi | 6-(4-Hydroxy-phenyl)-3-styryl-1H-pyrazolo[3,4-b]pyridine-4-carboxylic acid amide derivatives as kinase inhibitors |
Citations (3)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2000052029A1 (en) * | 1999-03-04 | 2000-09-08 | Eugene Science Inc. | Water-soluble sterol derivative for inhibiting cholesterol absorption and process for preparing the same |
| KR20020081834A (en) * | 2001-04-20 | 2002-10-30 | 주식회사 유엘바이오텍 | Serum cholesterol lowering agent and methods for preparing them |
| WO2005005453A2 (en) * | 2003-07-09 | 2005-01-20 | Forbes Medi-Tech Inc. | Novel compounds and compositions comprising sterols and/or stanols and cholesterol biosynthesis inhibitors and use thereof in treating or preventing a variety of diseases and conditions. |
-
2007
- 2007-09-11 KR KR1020070091861A patent/KR100886466B1/en active IP Right Grant
- 2007-09-11 WO PCT/KR2007/004395 patent/WO2008032980A1/en active Application Filing
Patent Citations (3)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2000052029A1 (en) * | 1999-03-04 | 2000-09-08 | Eugene Science Inc. | Water-soluble sterol derivative for inhibiting cholesterol absorption and process for preparing the same |
| KR20020081834A (en) * | 2001-04-20 | 2002-10-30 | 주식회사 유엘바이오텍 | Serum cholesterol lowering agent and methods for preparing them |
| WO2005005453A2 (en) * | 2003-07-09 | 2005-01-20 | Forbes Medi-Tech Inc. | Novel compounds and compositions comprising sterols and/or stanols and cholesterol biosynthesis inhibitors and use thereof in treating or preventing a variety of diseases and conditions. |
Non-Patent Citations (1)
| Title |
|---|
| KAZUNOBU TOSHIMA ET AL.: "Recent Progress in O-Glycosylation Methods and Its Application to Natural Products Synthesis", CHEMICAL REVIEWS, vol. 93, no. 4, June 1993 (1993-06-01), pages 1503 - 1531 * |
Cited By (3)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2009021740A2 (en) | 2007-08-15 | 2009-02-19 | Sanofis-Aventis | Substituted tetrahydronaphthalenes, process for the preparation thereof and the use thereof as medicaments |
| WO2011157827A1 (en) | 2010-06-18 | 2011-12-22 | Sanofi | Azolopyridin-3-one derivatives as inhibitors of lipases and phospholipases |
| EP2567959A1 (en) | 2011-09-12 | 2013-03-13 | Sanofi | 6-(4-Hydroxy-phenyl)-3-styryl-1H-pyrazolo[3,4-b]pyridine-4-carboxylic acid amide derivatives as kinase inhibitors |
Also Published As
| Publication number | Publication date |
|---|---|
| KR20080024075A (en) | 2008-03-17 |
| KR100886466B1 (en) | 2009-03-04 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| EP1952817B1 (en) | Aloe vera extract, method of producing aloe vera extract, and hyperglycemia improving agent | |
| US8470807B2 (en) | Antioxidant | |
| JP5121308B2 (en) | Composition for preventing, improving or treating metabolic syndrome | |
| US20170035725A1 (en) | Muscle atrophy inhibitor | |
| EP2218342A2 (en) | Novel nutraceutical compositions and use thereof | |
| WO2011077800A1 (en) | Hyperlipemia-ameliorating agent, anemia-ameliorating composition, uric-acid-level-reducing composition, and foods and beverages | |
| WO2011148657A1 (en) | Composition for prevention, amelioration or treatment of metabolic syndrome | |
| JPH0920660A (en) | Stress-resistant composition | |
| WO2011002033A1 (en) | Glucose metabolism-improving agent and glucose metabolism-improving composition | |
| AU2009318455B2 (en) | Antioxidant | |
| WO2008032980A1 (en) | Novel stigmasterol derivative or pharmaceutically acceptable salt thereof, method of producing the same, and composition containing the same to inhibit obesity or to prevent and treat hyperlipidemia | |
| AU2013298354A1 (en) | Compounds and their effects on appetite control and insulin sensitivity | |
| KR100966719B1 (en) | Composition comprising Orostachys japonicus extract for inhibiting obesity and for preventing or treating hyperlipidemia | |
| WO2019107531A1 (en) | Composition for suppressing increase in or reducing blood lipids | |
| JP2012505208A (en) | Novel use of pandoratin derivatives or Boesenbergia pandurata extract | |
| JP3634721B2 (en) | Preventive or therapeutic agent for hyperlipidemia | |
| WO2004083179A1 (en) | Remedy for diabetes | |
| JP4022350B2 (en) | Composition having an inhibitory effect on cholesterol elevation and an inhibitory effect on HDL-cholesterol lowering | |
| JP6391959B2 (en) | Non-alcoholic steatohepatitis ameliorating agent and ameliorating nutrition composition | |
| JP6194192B2 (en) | Adiponectin production promoter | |
| JP2011012005A (en) | Hyperlipidemia-ameliorating agent | |
| JP2016104816A (en) | Food product for inhibiting blood sugar level elevation | |
| JP6742853B2 (en) | Persistent quercetin glycoside preparation | |
| JP5801572B2 (en) | PPARγ activator | |
| WO2021255979A1 (en) | Anti-obesity composition and composition for oral administration |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application |
Ref document number: 07808187 Country of ref document: EP Kind code of ref document: A1 |
|
| NENP | Non-entry into the national phase |
Ref country code: DE |
|
| 122 | Ep: pct application non-entry in european phase |
Ref document number: 07808187 Country of ref document: EP Kind code of ref document: A1 |