WO2008015945A1 - ORGANIC LUMINESCENT DEVICE AND BENZO[k]FLUORANTHENE COMPOUND - Google Patents
ORGANIC LUMINESCENT DEVICE AND BENZO[k]FLUORANTHENE COMPOUND Download PDFInfo
- Publication number
- WO2008015945A1 WO2008015945A1 PCT/JP2007/064615 JP2007064615W WO2008015945A1 WO 2008015945 A1 WO2008015945 A1 WO 2008015945A1 JP 2007064615 W JP2007064615 W JP 2007064615W WO 2008015945 A1 WO2008015945 A1 WO 2008015945A1
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- group
- substituted
- unsubstituted
- compound
- general formula
- Prior art date
Links
- -1 BENZO[k]FLUORANTHENE COMPOUND Chemical class 0.000 title claims abstract description 112
- HAXBIWFMXWRORI-UHFFFAOYSA-N BkF Natural products C1=CC(C2=CC3=CC=CC=C3C=C22)=C3C2=CC=CC3=C1 HAXBIWFMXWRORI-UHFFFAOYSA-N 0.000 title claims abstract description 42
- 125000000217 alkyl group Chemical group 0.000 claims abstract description 27
- 125000000623 heterocyclic group Chemical group 0.000 claims abstract description 27
- 125000003710 aryl alkyl group Chemical group 0.000 claims abstract description 26
- 125000001997 phenyl group Chemical group [H]C1=C([H])C([H])=C(*)C([H])=C1[H] 0.000 claims abstract description 14
- 150000002894 organic compounds Chemical class 0.000 claims abstract description 11
- 150000001875 compounds Chemical class 0.000 claims description 170
- 125000003118 aryl group Chemical group 0.000 claims description 18
- 125000005843 halogen group Chemical group 0.000 claims description 18
- 125000003545 alkoxy group Chemical group 0.000 claims description 17
- 125000004435 hydrogen atom Chemical group [H]* 0.000 claims description 9
- 125000003983 fluorenyl group Chemical group C1(=CC=CC=2C3=CC=CC=C3CC12)* 0.000 claims description 3
- 125000000843 phenylene group Chemical group C1(=C(C=CC=C1)*)* 0.000 claims description 2
- NAWXUBYGYWOOIX-SFHVURJKSA-N (2s)-2-[[4-[2-(2,4-diaminoquinazolin-6-yl)ethyl]benzoyl]amino]-4-methylidenepentanedioic acid Chemical compound C1=CC2=NC(N)=NC(N)=C2C=C1CCC1=CC=C(C(=O)N[C@@H](CC(=C)C(O)=O)C(O)=O)C=C1 NAWXUBYGYWOOIX-SFHVURJKSA-N 0.000 claims 1
- 230000003287 optical effect Effects 0.000 abstract description 2
- 229910052736 halogen Inorganic materials 0.000 abstract 1
- 150000002367 halogens Chemical class 0.000 abstract 1
- 150000002431 hydrogen Chemical class 0.000 abstract 1
- 229910052739 hydrogen Inorganic materials 0.000 abstract 1
- 239000001257 hydrogen Substances 0.000 abstract 1
- YXFVVABEGXRONW-UHFFFAOYSA-N Toluene Chemical compound CC1=CC=CC=C1 YXFVVABEGXRONW-UHFFFAOYSA-N 0.000 description 93
- 239000010410 layer Substances 0.000 description 66
- 238000004519 manufacturing process Methods 0.000 description 66
- 239000000463 material Substances 0.000 description 45
- XEKOWRVHYACXOJ-UHFFFAOYSA-N Ethyl acetate Chemical compound CCOC(C)=O XEKOWRVHYACXOJ-UHFFFAOYSA-N 0.000 description 42
- IJGRMHOSHXDMSA-UHFFFAOYSA-N Atomic nitrogen Chemical compound N#N IJGRMHOSHXDMSA-UHFFFAOYSA-N 0.000 description 30
- 239000012044 organic layer Substances 0.000 description 24
- 239000010408 film Substances 0.000 description 22
- 238000000034 method Methods 0.000 description 19
- 239000000203 mixture Substances 0.000 description 19
- 239000000243 solution Substances 0.000 description 19
- 239000000758 substrate Substances 0.000 description 19
- 230000015572 biosynthetic process Effects 0.000 description 17
- IMNFDUFMRHMDMM-UHFFFAOYSA-N N-Heptane Chemical compound CCCCCCC IMNFDUFMRHMDMM-UHFFFAOYSA-N 0.000 description 16
- 239000012298 atmosphere Substances 0.000 description 16
- 229910052757 nitrogen Inorganic materials 0.000 description 15
- 238000001228 spectrum Methods 0.000 description 15
- XLYOFNOQVPJJNP-UHFFFAOYSA-N water Chemical compound O XLYOFNOQVPJJNP-UHFFFAOYSA-N 0.000 description 15
- LFQSCWFLJHTTHZ-UHFFFAOYSA-N Ethanol Chemical compound CCO LFQSCWFLJHTTHZ-UHFFFAOYSA-N 0.000 description 14
- 239000012046 mixed solvent Substances 0.000 description 14
- NFHFRUOZVGFOOS-UHFFFAOYSA-N palladium;triphenylphosphane Chemical compound [Pd].C1=CC=CC=C1P(C=1C=CC=CC=1)C1=CC=CC=C1.C1=CC=CC=C1P(C=1C=CC=CC=1)C1=CC=CC=C1.C1=CC=CC=C1P(C=1C=CC=CC=1)C1=CC=CC=C1.C1=CC=CC=C1P(C=1C=CC=CC=1)C1=CC=CC=C1 NFHFRUOZVGFOOS-UHFFFAOYSA-N 0.000 description 14
- 229920005989 resin Polymers 0.000 description 13
- 239000011347 resin Substances 0.000 description 13
- 230000005525 hole transport Effects 0.000 description 12
- 229940125782 compound 2 Drugs 0.000 description 11
- 239000002904 solvent Substances 0.000 description 11
- 238000005160 1H NMR spectroscopy Methods 0.000 description 10
- HEDRZPFGACZZDS-MICDWDOJSA-N Trichloro(2H)methane Chemical compound [2H]C(Cl)(Cl)Cl HEDRZPFGACZZDS-MICDWDOJSA-N 0.000 description 10
- 238000003786 synthesis reaction Methods 0.000 description 10
- 238000010521 absorption reaction Methods 0.000 description 9
- 238000000862 absorption spectrum Methods 0.000 description 9
- GVEPBJHOBDJJJI-UHFFFAOYSA-N fluoranthene Chemical compound C1=CC(C2=CC=CC=C22)=C3C2=CC=CC3=C1 GVEPBJHOBDJJJI-UHFFFAOYSA-N 0.000 description 9
- 230000006870 function Effects 0.000 description 9
- 238000001840 matrix-assisted laser desorption--ionisation time-of-flight mass spectrometry Methods 0.000 description 9
- 238000000870 ultraviolet spectroscopy Methods 0.000 description 9
- 238000001771 vacuum deposition Methods 0.000 description 9
- 238000001035 drying Methods 0.000 description 8
- 239000011521 glass Substances 0.000 description 8
- 238000004020 luminiscence type Methods 0.000 description 8
- 230000008569 process Effects 0.000 description 8
- 238000010898 silica gel chromatography Methods 0.000 description 8
- PMZURENOXWZQFD-UHFFFAOYSA-L Sodium Sulfate Chemical compound [Na+].[Na+].[O-]S([O-])(=O)=O PMZURENOXWZQFD-UHFFFAOYSA-L 0.000 description 7
- 239000007864 aqueous solution Substances 0.000 description 7
- 239000012153 distilled water Substances 0.000 description 7
- 239000000284 extract Substances 0.000 description 7
- 238000002347 injection Methods 0.000 description 7
- 239000007924 injection Substances 0.000 description 7
- 229920002545 silicone oil Polymers 0.000 description 7
- 229910052938 sodium sulfate Inorganic materials 0.000 description 7
- 235000011152 sodium sulphate Nutrition 0.000 description 7
- HPALAKNZSZLMCH-UHFFFAOYSA-M sodium;chloride;hydrate Chemical class O.[Na+].[Cl-] HPALAKNZSZLMCH-UHFFFAOYSA-M 0.000 description 7
- KHNYNFUTFKJLDD-UHFFFAOYSA-N Benzo[j]fluoranthene Chemical group C1=CC(C=2C3=CC=CC=C3C=CC=22)=C3C2=CC=CC3=C1 KHNYNFUTFKJLDD-UHFFFAOYSA-N 0.000 description 6
- KFZMGEQAYNKOFK-UHFFFAOYSA-N Isopropanol Chemical compound CC(C)O KFZMGEQAYNKOFK-UHFFFAOYSA-N 0.000 description 6
- CDBYLPFSWZWCQE-UHFFFAOYSA-L Sodium Carbonate Chemical compound [Na+].[Na+].[O-]C([O-])=O CDBYLPFSWZWCQE-UHFFFAOYSA-L 0.000 description 6
- 230000006866 deterioration Effects 0.000 description 6
- 125000003914 fluoranthenyl group Chemical group C1(=CC=C2C=CC=C3C4=CC=CC=C4C1=C23)* 0.000 description 6
- 125000005581 pyrene group Chemical group 0.000 description 6
- 239000010409 thin film Substances 0.000 description 6
- GDTBXPJZTBHREO-UHFFFAOYSA-N bromine Substances BrBr GDTBXPJZTBHREO-UHFFFAOYSA-N 0.000 description 5
- 229910052794 bromium Inorganic materials 0.000 description 5
- 239000002019 doping agent Substances 0.000 description 5
- 238000005259 measurement Methods 0.000 description 5
- 229910052751 metal Inorganic materials 0.000 description 5
- 239000002184 metal Substances 0.000 description 5
- 150000003220 pyrenes Chemical group 0.000 description 5
- 125000001424 substituent group Chemical group 0.000 description 5
- MBWSEPXTVAGLJS-UHFFFAOYSA-N 3-bromo-7,12-diphenylbenzo[k]fluoranthene Chemical compound C=12C3=CC=CC=1C(Br)=CC=C2C(C(=C1C=CC=CC1=1)C=2C=CC=CC=2)=C3C=1C1=CC=CC=C1 MBWSEPXTVAGLJS-UHFFFAOYSA-N 0.000 description 4
- HEDRZPFGACZZDS-UHFFFAOYSA-N Chloroform Chemical compound ClC(Cl)Cl HEDRZPFGACZZDS-UHFFFAOYSA-N 0.000 description 4
- 125000004054 acenaphthylenyl group Chemical group C1(=CC2=CC=CC3=CC=CC1=C23)* 0.000 description 4
- FJDQFPXHSGXQBY-UHFFFAOYSA-L caesium carbonate Chemical compound [Cs+].[Cs+].[O-]C([O-])=O FJDQFPXHSGXQBY-UHFFFAOYSA-L 0.000 description 4
- 229910000024 caesium carbonate Inorganic materials 0.000 description 4
- 230000005284 excitation Effects 0.000 description 4
- 230000009477 glass transition Effects 0.000 description 4
- 238000010438 heat treatment Methods 0.000 description 4
- WFOVEDJTASPCIR-UHFFFAOYSA-N 3-[(4-methyl-5-pyridin-4-yl-1,2,4-triazol-3-yl)methylamino]-n-[[2-(trifluoromethyl)phenyl]methyl]benzamide Chemical compound N=1N=C(C=2C=CN=CC=2)N(C)C=1CNC(C=1)=CC=CC=1C(=O)NCC1=CC=CC=C1C(F)(F)F WFOVEDJTASPCIR-UHFFFAOYSA-N 0.000 description 3
- 229910001148 Al-Li alloy Inorganic materials 0.000 description 3
- CTQNGGLPUBDAKN-UHFFFAOYSA-N O-Xylene Chemical compound CC1=CC=CC=C1C CTQNGGLPUBDAKN-UHFFFAOYSA-N 0.000 description 3
- ZMANZCXQSJIPKH-UHFFFAOYSA-N Triethylamine Chemical compound CCN(CC)CC ZMANZCXQSJIPKH-UHFFFAOYSA-N 0.000 description 3
- HXGDTGSAIMULJN-UHFFFAOYSA-N acetnaphthylene Natural products C1=CC(C=C2)=C3C2=CC=CC3=C1 HXGDTGSAIMULJN-UHFFFAOYSA-N 0.000 description 3
- 229910052782 aluminium Inorganic materials 0.000 description 3
- XAGFODPZIPBFFR-UHFFFAOYSA-N aluminium Chemical compound [Al] XAGFODPZIPBFFR-UHFFFAOYSA-N 0.000 description 3
- 239000011248 coating agent Substances 0.000 description 3
- 238000000576 coating method Methods 0.000 description 3
- 229940125904 compound 1 Drugs 0.000 description 3
- 229940125773 compound 10 Drugs 0.000 description 3
- 229940126214 compound 3 Drugs 0.000 description 3
- 229940125898 compound 5 Drugs 0.000 description 3
- 229910052731 fluorine Inorganic materials 0.000 description 3
- AMGQUBHHOARCQH-UHFFFAOYSA-N indium;oxotin Chemical compound [In].[Sn]=O AMGQUBHHOARCQH-UHFFFAOYSA-N 0.000 description 3
- ZLVXBBHTMQJRSX-VMGNSXQWSA-N jdtic Chemical compound C1([C@]2(C)CCN(C[C@@H]2C)C[C@H](C(C)C)NC(=O)[C@@H]2NCC3=CC(O)=CC=C3C2)=CC=CC(O)=C1 ZLVXBBHTMQJRSX-VMGNSXQWSA-N 0.000 description 3
- PQXKHYXIUOZZFA-UHFFFAOYSA-M lithium fluoride Chemical compound [Li+].[F-] PQXKHYXIUOZZFA-UHFFFAOYSA-M 0.000 description 3
- 229910044991 metal oxide Inorganic materials 0.000 description 3
- 150000004706 metal oxides Chemical class 0.000 description 3
- PXHVJJICTQNCMI-UHFFFAOYSA-N nickel Substances [Ni] PXHVJJICTQNCMI-UHFFFAOYSA-N 0.000 description 3
- 229910000029 sodium carbonate Inorganic materials 0.000 description 3
- 125000000999 tert-butyl group Chemical group [H]C([H])([H])C(*)(C([H])([H])[H])C([H])([H])[H] 0.000 description 3
- ZKSVYBRJSMBDMV-UHFFFAOYSA-N 1,3-diphenyl-2-benzofuran Chemical compound C1=CC=CC=C1C1=C2C=CC=CC2=C(C=2C=CC=CC=2)O1 ZKSVYBRJSMBDMV-UHFFFAOYSA-N 0.000 description 2
- CTPUUDQIXKUAMO-UHFFFAOYSA-N 1-bromo-3-iodobenzene Chemical compound BrC1=CC=CC(I)=C1 CTPUUDQIXKUAMO-UHFFFAOYSA-N 0.000 description 2
- UCCUXODGPMAHRL-UHFFFAOYSA-N 1-bromo-4-iodobenzene Chemical compound BrC1=CC=C(I)C=C1 UCCUXODGPMAHRL-UHFFFAOYSA-N 0.000 description 2
- FZJPZRROCUUMOI-UHFFFAOYSA-N 1-bromofluoranthene Chemical compound C1=CC=C2C3=CC=CC=C3C3=C2C1=CC=C3Br FZJPZRROCUUMOI-UHFFFAOYSA-N 0.000 description 2
- 125000001255 4-fluorophenyl group Chemical group [H]C1=C([H])C(*)=C([H])C([H])=C1F 0.000 description 2
- ZCYVEMRRCGMTRW-UHFFFAOYSA-N 7553-56-2 Chemical group [I] ZCYVEMRRCGMTRW-UHFFFAOYSA-N 0.000 description 2
- CSCPPACGZOOCGX-UHFFFAOYSA-N Acetone Chemical compound CC(C)=O CSCPPACGZOOCGX-UHFFFAOYSA-N 0.000 description 2
- 239000004925 Acrylic resin Substances 0.000 description 2
- 229920000178 Acrylic resin Polymers 0.000 description 2
- WKBOTKDWSSQWDR-UHFFFAOYSA-N Bromine atom Chemical group [Br] WKBOTKDWSSQWDR-UHFFFAOYSA-N 0.000 description 2
- UFHFLCQGNIYNRP-VVKOMZTBSA-N Dideuterium Chemical group [2H][2H] UFHFLCQGNIYNRP-VVKOMZTBSA-N 0.000 description 2
- YCKRFDGAMUMZLT-UHFFFAOYSA-N Fluorine atom Chemical compound [F] YCKRFDGAMUMZLT-UHFFFAOYSA-N 0.000 description 2
- KDLHZDBZIXYQEI-UHFFFAOYSA-N Palladium Chemical compound [Pd] KDLHZDBZIXYQEI-UHFFFAOYSA-N 0.000 description 2
- XLOMVQKBTHCTTD-UHFFFAOYSA-N Zinc monoxide Chemical compound [Zn]=O XLOMVQKBTHCTTD-UHFFFAOYSA-N 0.000 description 2
- JFBZPFYRPYOZCQ-UHFFFAOYSA-N [Li].[Al] Chemical compound [Li].[Al] JFBZPFYRPYOZCQ-UHFFFAOYSA-N 0.000 description 2
- 229910045601 alloy Inorganic materials 0.000 description 2
- 239000000956 alloy Substances 0.000 description 2
- 125000002102 aryl alkyloxo group Chemical group 0.000 description 2
- QVGXLLKOCUKJST-UHFFFAOYSA-N atomic oxygen Chemical compound [O] QVGXLLKOCUKJST-UHFFFAOYSA-N 0.000 description 2
- 239000011230 binding agent Substances 0.000 description 2
- 125000006267 biphenyl group Chemical group 0.000 description 2
- 230000000903 blocking effect Effects 0.000 description 2
- 238000006243 chemical reaction Methods 0.000 description 2
- 239000000460 chlorine Substances 0.000 description 2
- 229910052801 chlorine Inorganic materials 0.000 description 2
- 238000004140 cleaning Methods 0.000 description 2
- 238000000151 deposition Methods 0.000 description 2
- 230000008021 deposition Effects 0.000 description 2
- 239000007772 electrode material Substances 0.000 description 2
- 230000007613 environmental effect Effects 0.000 description 2
- 125000001495 ethyl group Chemical group [H]C([H])([H])C([H])([H])* 0.000 description 2
- 230000002349 favourable effect Effects 0.000 description 2
- 150000002220 fluorenes Chemical class 0.000 description 2
- 150000008376 fluorenones Chemical class 0.000 description 2
- 239000011737 fluorine Substances 0.000 description 2
- 150000002430 hydrocarbons Chemical group 0.000 description 2
- 229910052740 iodine Inorganic materials 0.000 description 2
- 238000004768 lowest unoccupied molecular orbital Methods 0.000 description 2
- 125000002496 methyl group Chemical group [H]C([H])([H])* 0.000 description 2
- 125000003261 o-tolyl group Chemical group [H]C1=C([H])C(*)=C(C([H])=C1[H])C([H])([H])[H] 0.000 description 2
- 125000002524 organometallic group Chemical group 0.000 description 2
- 150000004866 oxadiazoles Chemical class 0.000 description 2
- 150000007978 oxazole derivatives Chemical class 0.000 description 2
- 239000001301 oxygen Substances 0.000 description 2
- 229910052760 oxygen Inorganic materials 0.000 description 2
- 125000002080 perylenyl group Chemical group C1(=CC=C2C=CC=C3C4=CC=CC5=CC=CC(C1=C23)=C45)* 0.000 description 2
- 125000001791 phenazinyl group Chemical group C1(=CC=CC2=NC3=CC=CC=C3N=C12)* 0.000 description 2
- BASFCYQUMIYNBI-UHFFFAOYSA-N platinum Chemical compound [Pt] BASFCYQUMIYNBI-UHFFFAOYSA-N 0.000 description 2
- 229920003227 poly(N-vinyl carbazole) Polymers 0.000 description 2
- 229920005990 polystyrene resin Polymers 0.000 description 2
- 229920000123 polythiophene Polymers 0.000 description 2
- 239000011241 protective layer Substances 0.000 description 2
- 150000003216 pyrazines Chemical class 0.000 description 2
- 125000001742 pyren-1-yl group Chemical group [H]C1=C([H])C2=C([H])C([H])=C3C(*)=C([H])C([H])=C4C([H])=C([H])C(=C1[H])C2=C34 0.000 description 2
- 125000001486 pyren-4-yl group Chemical group [H]C1=C(C2=C34)C([H])=C([H])C([H])=C2C([*])=C([H])C3=C([H])C([H])=C([H])C4=C1[H] 0.000 description 2
- 125000004076 pyridyl group Chemical group 0.000 description 2
- 125000000168 pyrrolyl group Chemical group 0.000 description 2
- 238000006862 quantum yield reaction Methods 0.000 description 2
- 238000010791 quenching Methods 0.000 description 2
- 230000000171 quenching effect Effects 0.000 description 2
- 125000001567 quinoxalinyl group Chemical group N1=C(C=NC2=CC=CC=C12)* 0.000 description 2
- 238000010992 reflux Methods 0.000 description 2
- 238000007789 sealing Methods 0.000 description 2
- 229920002050 silicone resin Polymers 0.000 description 2
- 229910052709 silver Inorganic materials 0.000 description 2
- 239000004332 silver Substances 0.000 description 2
- 239000002356 single layer Substances 0.000 description 2
- 239000007787 solid Substances 0.000 description 2
- 238000004528 spin coating Methods 0.000 description 2
- 238000004544 sputter deposition Methods 0.000 description 2
- PJANXHGTPQOBST-UHFFFAOYSA-N stilbene Chemical class C=1C=CC=CC=1C=CC1=CC=CC=C1 PJANXHGTPQOBST-UHFFFAOYSA-N 0.000 description 2
- 239000000126 substance Substances 0.000 description 2
- 238000006467 substitution reaction Methods 0.000 description 2
- 229940042055 systemic antimycotics triazole derivative Drugs 0.000 description 2
- 125000001544 thienyl group Chemical group 0.000 description 2
- QAEDZJGFFMLHHQ-UHFFFAOYSA-N trifluoroacetic anhydride Chemical compound FC(F)(F)C(=O)OC(=O)C(F)(F)F QAEDZJGFFMLHHQ-UHFFFAOYSA-N 0.000 description 2
- 239000008096 xylene Substances 0.000 description 2
- LVEYOSJUKRVCCF-UHFFFAOYSA-N 1,3-Bis(diphenylphosphino)propane Substances C=1C=CC=CC=1P(C=1C=CC=CC=1)CCCP(C=1C=CC=CC=1)C1=CC=CC=C1 LVEYOSJUKRVCCF-UHFFFAOYSA-N 0.000 description 1
- YJTKZCDBKVTVBY-UHFFFAOYSA-N 1,3-Diphenylbenzene Chemical group C1=CC=CC=C1C1=CC=CC(C=2C=CC=CC=2)=C1 YJTKZCDBKVTVBY-UHFFFAOYSA-N 0.000 description 1
- BCMCBBGGLRIHSE-UHFFFAOYSA-N 1,3-benzoxazole Chemical class C1=CC=C2OC=NC2=C1 BCMCBBGGLRIHSE-UHFFFAOYSA-N 0.000 description 1
- CBRJPFGIXUFMTM-WDEREUQCSA-N 1-[(2S,5R)-2-methyl-5-(7H-pyrrolo[2,3-d]pyrimidin-4-ylamino)piperidin-1-yl]prop-2-en-1-one Chemical compound N1=CN=C(C2=C1NC=C2)N[C@@H]2CC[C@@H](N(C2)C(C=C)=O)C CBRJPFGIXUFMTM-WDEREUQCSA-N 0.000 description 1
- HYZJCKYKOHLVJF-UHFFFAOYSA-N 1H-benzimidazole Chemical class C1=CC=C2NC=NC2=C1 HYZJCKYKOHLVJF-UHFFFAOYSA-N 0.000 description 1
- 125000000453 2,2,2-trichloroethyl group Chemical group [H]C([H])(*)C(Cl)(Cl)Cl 0.000 description 1
- 125000004206 2,2,2-trifluoroethyl group Chemical group [H]C([H])(*)C(F)(F)F 0.000 description 1
- 125000003163 2-(2-naphthyl)ethyl group Chemical group [H]C1=C([H])C([H])=C2C([H])=C(C([H])=C([H])C2=C1[H])C([H])([H])C([H])([H])* 0.000 description 1
- 125000006280 2-bromobenzyl group Chemical group [H]C1=C([H])C(Br)=C(C([H])=C1[H])C([H])([H])* 0.000 description 1
- 125000005999 2-bromoethyl group Chemical group 0.000 description 1
- 125000006282 2-chlorobenzyl group Chemical group [H]C1=C([H])C(Cl)=C(C([H])=C1[H])C([H])([H])* 0.000 description 1
- 125000001340 2-chloroethyl group Chemical group [H]C([H])(Cl)C([H])([H])* 0.000 description 1
- 125000004847 2-fluorobenzyl group Chemical group [H]C1=C([H])C(F)=C(C([H])=C1[H])C([H])([H])* 0.000 description 1
- 125000004777 2-fluoroethyl group Chemical group [H]C([H])(F)C([H])([H])* 0.000 description 1
- 125000004198 2-fluorophenyl group Chemical group [H]C1=C([H])C(F)=C(*)C([H])=C1[H] 0.000 description 1
- 125000000094 2-phenylethyl group Chemical group [H]C1=C([H])C([H])=C(C([H])=C1[H])C([H])([H])C([H])([H])* 0.000 description 1
- AYMGBIYWAZRPJY-UHFFFAOYSA-N 2-tert-butylpyrene Chemical compound C1=CC=C2C=CC3=CC(C(C)(C)C)=CC4=CC=C1C2=C43 AYMGBIYWAZRPJY-UHFFFAOYSA-N 0.000 description 1
- MGADZUXDNSDTHW-UHFFFAOYSA-N 2H-pyran Chemical class C1OC=CC=C1 MGADZUXDNSDTHW-UHFFFAOYSA-N 0.000 description 1
- GNIBJWUSOXCGLD-UHFFFAOYSA-N 3-bromo-7,12-ditert-butylbenzo[k]fluoranthene Chemical compound C1=CC=C2C(C(C)(C)C)=C(C=3C4=C5C=CC(Br)=C4C=CC=3)C5=C(C(C)(C)C)C2=C1 GNIBJWUSOXCGLD-UHFFFAOYSA-N 0.000 description 1
- APNZIMKIIKRHKS-UHFFFAOYSA-N 3-bromobenzo[k]fluoranthene Chemical compound C12=CC3=CC=CC=C3C=C2C2=CC=CC3=C2C1=CC=C3Br APNZIMKIIKRHKS-UHFFFAOYSA-N 0.000 description 1
- 125000006279 3-bromobenzyl group Chemical group [H]C1=C([H])C(=C([H])C(Br)=C1[H])C([H])([H])* 0.000 description 1
- 125000003852 3-chlorobenzyl group Chemical group [H]C1=C([H])C(=C([H])C(Cl)=C1[H])C([H])([H])* 0.000 description 1
- GYKIOFRJZAGNQX-UHFFFAOYSA-N 3-fluoranthen-8-ylbenzo[k]fluoranthene Chemical compound C1=CC(C2=CC=C(C=C22)C3=C4C=CC=C5C=6C(C(C=C3)=C54)=CC3=CC=CC=C3C=6)=C3C2=CC=CC3=C1 GYKIOFRJZAGNQX-UHFFFAOYSA-N 0.000 description 1
- 125000006284 3-fluorobenzyl group Chemical group [H]C1=C([H])C(=C([H])C(F)=C1[H])C([H])([H])* 0.000 description 1
- 125000004180 3-fluorophenyl group Chemical group [H]C1=C([H])C(*)=C([H])C(F)=C1[H] 0.000 description 1
- LZPWAYBEOJRFAX-UHFFFAOYSA-N 4,4,5,5-tetramethyl-1,3,2$l^{2}-dioxaborolane Chemical compound CC1(C)O[B]OC1(C)C LZPWAYBEOJRFAX-UHFFFAOYSA-N 0.000 description 1
- DEYUTQGOXIQQBT-UHFFFAOYSA-N 4-bromo-3,7,12-triphenylbenzo[k]fluoranthene Chemical compound C12=C3C(Br)=CC=C2C(C(=C2C=CC=CC2=C2C=4C=CC=CC=4)C=4C=CC=CC=4)=C2C1=CC=C3C1=CC=CC=C1 DEYUTQGOXIQQBT-UHFFFAOYSA-N 0.000 description 1
- NTPFGGJISCMKQN-UHFFFAOYSA-N 4-bromo-3-methyl-7,12-diphenylbenzo[k]fluoranthene Chemical compound C=12C3=CC=C(Br)C=1C(C)=CC=C2C(C(=C1C=CC=CC1=1)C=2C=CC=CC=2)=C3C=1C1=CC=CC=C1 NTPFGGJISCMKQN-UHFFFAOYSA-N 0.000 description 1
- 125000006281 4-bromobenzyl group Chemical group [H]C1=C([H])C(=C([H])C([H])=C1Br)C([H])([H])* 0.000 description 1
- 125000006283 4-chlorobenzyl group Chemical group [H]C1=C([H])C(=C([H])C([H])=C1Cl)C([H])([H])* 0.000 description 1
- 125000004801 4-cyanophenyl group Chemical group [H]C1=C([H])C(C#N)=C([H])C([H])=C1* 0.000 description 1
- 125000004860 4-ethylphenyl group Chemical group [H]C1=C([H])C(=C([H])C([H])=C1*)C([H])([H])C([H])([H])[H] 0.000 description 1
- 125000004176 4-fluorobenzyl group Chemical group [H]C1=C([H])C(=C([H])C([H])=C1F)C([H])([H])* 0.000 description 1
- 125000004861 4-isopropyl phenyl group Chemical group [H]C1=C([H])C(=C([H])C([H])=C1*)C([H])(C([H])([H])[H])C([H])([H])[H] 0.000 description 1
- 125000004172 4-methoxyphenyl group Chemical group [H]C1=C([H])C(OC([H])([H])[H])=C([H])C([H])=C1* 0.000 description 1
- 125000000590 4-methylphenyl group Chemical group [H]C1=C([H])C(=C([H])C([H])=C1*)C([H])([H])[H] 0.000 description 1
- 125000004199 4-trifluoromethylphenyl group Chemical group [H]C1=C([H])C(=C([H])C([H])=C1*)C(F)(F)F 0.000 description 1
- UXSHNPXCELYWAW-UHFFFAOYSA-N 5,6-dibromo-1,2-dihydroacenaphthylene Chemical compound C1CC2=CC=C(Br)C3=C2C1=CC=C3Br UXSHNPXCELYWAW-UHFFFAOYSA-N 0.000 description 1
- BPPBSKDZDJKJLJ-UHFFFAOYSA-N 5,6-dibromoacenaphthylene Chemical group C1=CC2=CC=C(Br)C3=C2C1=CC=C3Br BPPBSKDZDJKJLJ-UHFFFAOYSA-N 0.000 description 1
- QALKJGMGKYKMKE-UHFFFAOYSA-N 5-bromo-1,2-dihydroacenaphthylene Chemical compound C1CC2=CC=CC3=C2C1=CC=C3Br QALKJGMGKYKMKE-UHFFFAOYSA-N 0.000 description 1
- PSRUTZHGMSPRPZ-UHFFFAOYSA-N 5-bromoacenaphthylene Chemical group C1=CC2=CC=CC3=C2C1=CC=C3Br PSRUTZHGMSPRPZ-UHFFFAOYSA-N 0.000 description 1
- YONDEBNCTLTCRE-UHFFFAOYSA-N 7,12-diphenylbenzo[k]fluoranthene Chemical compound C1=CC=CC=C1C(C1=CC=CC=C11)=C(C=2C3=C4C=CC=C3C=CC=2)C4=C1C1=CC=CC=C1 YONDEBNCTLTCRE-UHFFFAOYSA-N 0.000 description 1
- FCNCGHJSNVOIKE-UHFFFAOYSA-N 9,10-diphenylanthracene Chemical class C1=CC=CC=C1C(C1=CC=CC=C11)=C(C=CC=C2)C2=C1C1=CC=CC=C1 FCNCGHJSNVOIKE-UHFFFAOYSA-N 0.000 description 1
- NIXOWILDQLNWCW-UHFFFAOYSA-N Acrylic acid Chemical compound OC(=O)C=C NIXOWILDQLNWCW-UHFFFAOYSA-N 0.000 description 1
- ROFVEXUMMXZLPA-UHFFFAOYSA-N Bipyridyl Chemical group N1=CC=CC=C1C1=CC=CC=N1 ROFVEXUMMXZLPA-UHFFFAOYSA-N 0.000 description 1
- QXQJVMFNIXQEQF-UHFFFAOYSA-N CC(C)(C)c1cc(cc2)c3c4c2ccc(S2OC(C)(C)C(C)(C)O2)c4ccc3c1 Chemical compound CC(C)(C)c1cc(cc2)c3c4c2ccc(S2OC(C)(C)C(C)(C)O2)c4ccc3c1 QXQJVMFNIXQEQF-UHFFFAOYSA-N 0.000 description 1
- OYPRJOBELJOOCE-UHFFFAOYSA-N Calcium Chemical compound [Ca] OYPRJOBELJOOCE-UHFFFAOYSA-N 0.000 description 1
- ZAMOUSCENKQFHK-UHFFFAOYSA-N Chlorine atom Chemical compound [Cl] ZAMOUSCENKQFHK-UHFFFAOYSA-N 0.000 description 1
- VYZAMTAEIAYCRO-UHFFFAOYSA-N Chromium Chemical compound [Cr] VYZAMTAEIAYCRO-UHFFFAOYSA-N 0.000 description 1
- RYGMFSIKBFXOCR-UHFFFAOYSA-N Copper Chemical compound [Cu] RYGMFSIKBFXOCR-UHFFFAOYSA-N 0.000 description 1
- 239000004641 Diallyl-phthalate Substances 0.000 description 1
- 238000005698 Diels-Alder reaction Methods 0.000 description 1
- 238000003547 Friedel-Crafts alkylation reaction Methods 0.000 description 1
- DGAQECJNVWCQMB-PUAWFVPOSA-M Ilexoside XXIX Chemical compound C[C@@H]1CC[C@@]2(CC[C@@]3(C(=CC[C@H]4[C@]3(CC[C@@H]5[C@@]4(CC[C@@H](C5(C)C)OS(=O)(=O)[O-])C)C)[C@@H]2[C@]1(C)O)C)C(=O)O[C@H]6[C@@H]([C@H]([C@@H]([C@H](O6)CO)O)O)O.[Na+] DGAQECJNVWCQMB-PUAWFVPOSA-M 0.000 description 1
- WHXSMMKQMYFTQS-UHFFFAOYSA-N Lithium Chemical compound [Li] WHXSMMKQMYFTQS-UHFFFAOYSA-N 0.000 description 1
- FYYHWMGAXLPEAU-UHFFFAOYSA-N Magnesium Chemical compound [Mg] FYYHWMGAXLPEAU-UHFFFAOYSA-N 0.000 description 1
- CBENFWSGALASAD-UHFFFAOYSA-N Ozone Chemical compound [O-][O+]=O CBENFWSGALASAD-UHFFFAOYSA-N 0.000 description 1
- 239000005062 Polybutadiene Substances 0.000 description 1
- 239000004695 Polyether sulfone Substances 0.000 description 1
- 239000004698 Polyethylene Substances 0.000 description 1
- 229920000265 Polyparaphenylene Chemical class 0.000 description 1
- 239000004734 Polyphenylene sulfide Substances 0.000 description 1
- ZLMJMSJWJFRBEC-UHFFFAOYSA-N Potassium Chemical compound [K] ZLMJMSJWJFRBEC-UHFFFAOYSA-N 0.000 description 1
- NRCMAYZCPIVABH-UHFFFAOYSA-N Quinacridone Chemical class N1C2=CC=CC=C2C(=O)C2=C1C=C1C(=O)C3=CC=CC=C3NC1=C2 NRCMAYZCPIVABH-UHFFFAOYSA-N 0.000 description 1
- KJTLSVCANCCWHF-UHFFFAOYSA-N Ruthenium Chemical compound [Ru] KJTLSVCANCCWHF-UHFFFAOYSA-N 0.000 description 1
- BUGBHKTXTAQXES-UHFFFAOYSA-N Selenium Chemical compound [Se] BUGBHKTXTAQXES-UHFFFAOYSA-N 0.000 description 1
- BQCADISMDOOEFD-UHFFFAOYSA-N Silver Chemical compound [Ag] BQCADISMDOOEFD-UHFFFAOYSA-N 0.000 description 1
- 238000006161 Suzuki-Miyaura coupling reaction Methods 0.000 description 1
- ATJFFYVFTNAWJD-UHFFFAOYSA-N Tin Chemical compound [Sn] ATJFFYVFTNAWJD-UHFFFAOYSA-N 0.000 description 1
- RTAQQCXQSZGOHL-UHFFFAOYSA-N Titanium Chemical compound [Ti] RTAQQCXQSZGOHL-UHFFFAOYSA-N 0.000 description 1
- 229920001807 Urea-formaldehyde Polymers 0.000 description 1
- JHYLKGDXMUDNEO-UHFFFAOYSA-N [Mg].[In] Chemical compound [Mg].[In] JHYLKGDXMUDNEO-UHFFFAOYSA-N 0.000 description 1
- 239000006096 absorbing agent Substances 0.000 description 1
- 239000011354 acetal resin Substances 0.000 description 1
- 125000000641 acridinyl group Chemical group C1(=CC=CC2=NC3=CC=CC=C3C=C12)* 0.000 description 1
- FZEYVTFCMJSGMP-UHFFFAOYSA-N acridone Chemical class C1=CC=C2C(=O)C3=CC=CC=C3NC2=C1 FZEYVTFCMJSGMP-UHFFFAOYSA-N 0.000 description 1
- 229920000122 acrylonitrile butadiene styrene Polymers 0.000 description 1
- 230000009471 action Effects 0.000 description 1
- 125000005073 adamantyl group Chemical group C12(CC3CC(CC(C1)C3)C2)* 0.000 description 1
- 239000000654 additive Substances 0.000 description 1
- 239000000853 adhesive Substances 0.000 description 1
- 230000001070 adhesive effect Effects 0.000 description 1
- 239000012790 adhesive layer Substances 0.000 description 1
- SNAAJJQQZSMGQD-UHFFFAOYSA-N aluminum magnesium Chemical compound [Mg].[Al] SNAAJJQQZSMGQD-UHFFFAOYSA-N 0.000 description 1
- 125000003277 amino group Chemical group 0.000 description 1
- 150000008425 anthrones Chemical class 0.000 description 1
- 125000005428 anthryl group Chemical group [H]C1=C([H])C([H])=C2C([H])=C3C(*)=C([H])C([H])=C([H])C3=C([H])C2=C1[H] 0.000 description 1
- 239000003963 antioxidant agent Substances 0.000 description 1
- 229940027991 antiseptic and disinfectant quinoline derivative Drugs 0.000 description 1
- 150000001491 aromatic compounds Chemical class 0.000 description 1
- 125000004104 aryloxy group Chemical group 0.000 description 1
- 125000003828 azulenyl group Chemical group 0.000 description 1
- 125000004618 benzofuryl group Chemical group O1C(=CC2=C1C=CC=C2)* 0.000 description 1
- IOJUPLGTWVMSFF-UHFFFAOYSA-N benzothiazole Chemical class C1=CC=C2SC=NC2=C1 IOJUPLGTWVMSFF-UHFFFAOYSA-N 0.000 description 1
- 125000004196 benzothienyl group Chemical group S1C(=CC2=C1C=CC=C2)* 0.000 description 1
- 125000001797 benzyl group Chemical group [H]C1=C([H])C([H])=C(C([H])=C1[H])C([H])([H])* 0.000 description 1
- 230000005540 biological transmission Effects 0.000 description 1
- QUDWYFHPNIMBFC-UHFFFAOYSA-N bis(prop-2-enyl) benzene-1,2-dicarboxylate Chemical compound C=CCOC(=O)C1=CC=CC=C1C(=O)OCC=C QUDWYFHPNIMBFC-UHFFFAOYSA-N 0.000 description 1
- KGBXLFKZBHKPEV-UHFFFAOYSA-N boric acid Chemical compound OB(O)O KGBXLFKZBHKPEV-UHFFFAOYSA-N 0.000 description 1
- 239000004327 boric acid Substances 0.000 description 1
- 125000005997 bromomethyl group Chemical group 0.000 description 1
- QHIWVLPBUQWDMQ-UHFFFAOYSA-N butyl prop-2-enoate;methyl 2-methylprop-2-enoate;prop-2-enoic acid Chemical compound OC(=O)C=C.COC(=O)C(C)=C.CCCCOC(=O)C=C QHIWVLPBUQWDMQ-UHFFFAOYSA-N 0.000 description 1
- DKEFXFSHBYIQBW-UHFFFAOYSA-N c1cc(-c2cc(-c3ccccn3)c(cc3)c4c2ccc(c(-c2ncccc2)c2)c4c3c2-c2ccccn2)ncc1 Chemical compound c1cc(-c2cc(-c3ccccn3)c(cc3)c4c2ccc(c(-c2ncccc2)c2)c4c3c2-c2ccccn2)ncc1 DKEFXFSHBYIQBW-UHFFFAOYSA-N 0.000 description 1
- 229910052791 calcium Inorganic materials 0.000 description 1
- 239000011575 calcium Substances 0.000 description 1
- 238000004364 calculation method Methods 0.000 description 1
- 125000000609 carbazolyl group Chemical group C1(=CC=CC=2C3=CC=CC=C3NC12)* 0.000 description 1
- 239000000969 carrier Substances 0.000 description 1
- 238000005266 casting Methods 0.000 description 1
- 239000003054 catalyst Substances 0.000 description 1
- 239000000919 ceramic Substances 0.000 description 1
- 125000001309 chloro group Chemical group Cl* 0.000 description 1
- 125000004218 chloromethyl group Chemical group [H]C([H])(Cl)* 0.000 description 1
- 229910052804 chromium Inorganic materials 0.000 description 1
- 239000011651 chromium Substances 0.000 description 1
- 150000001846 chrysenes Chemical class 0.000 description 1
- 125000002676 chrysenyl group Chemical group C1(=CC=CC=2C3=CC=C4C=CC=CC4=C3C=CC12)* 0.000 description 1
- 229910017052 cobalt Inorganic materials 0.000 description 1
- 239000010941 cobalt Substances 0.000 description 1
- GUTLYIVDDKVIGB-UHFFFAOYSA-N cobalt atom Chemical compound [Co] GUTLYIVDDKVIGB-UHFFFAOYSA-N 0.000 description 1
- 239000003086 colorant Substances 0.000 description 1
- 238000009833 condensation Methods 0.000 description 1
- 230000005494 condensation Effects 0.000 description 1
- 229920001940 conductive polymer Polymers 0.000 description 1
- 239000000470 constituent Substances 0.000 description 1
- 229920001577 copolymer Polymers 0.000 description 1
- 229910052802 copper Inorganic materials 0.000 description 1
- 239000010949 copper Substances 0.000 description 1
- 150000001882 coronenes Chemical class 0.000 description 1
- ZYGHJZDHTFUPRJ-UHFFFAOYSA-N coumarin Chemical class C1=CC=C2OC(=O)C=CC2=C1 ZYGHJZDHTFUPRJ-UHFFFAOYSA-N 0.000 description 1
- 125000004093 cyano group Chemical group *C#N 0.000 description 1
- 125000001995 cyclobutyl group Chemical group [H]C1([H])C([H])([H])C([H])(*)C1([H])[H] 0.000 description 1
- 125000000113 cyclohexyl group Chemical group [H]C1([H])C([H])([H])C([H])([H])C([H])(*)C([H])([H])C1([H])[H] 0.000 description 1
- 125000004210 cyclohexylmethyl group Chemical group [H]C([H])(*)C1([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C1([H])[H] 0.000 description 1
- 125000001511 cyclopentyl group Chemical group [H]C1([H])C([H])([H])C([H])([H])C([H])(*)C1([H])[H] 0.000 description 1
- 125000004851 cyclopentylmethyl group Chemical group C1(CCCC1)C* 0.000 description 1
- 125000001559 cyclopropyl group Chemical group [H]C1([H])C([H])([H])C1([H])* 0.000 description 1
- 230000009849 deactivation Effects 0.000 description 1
- SWXVUIWOUIDPGS-UHFFFAOYSA-N diacetone alcohol Natural products CC(=O)CC(C)(C)O SWXVUIWOUIDPGS-UHFFFAOYSA-N 0.000 description 1
- 239000010432 diamond Substances 0.000 description 1
- 229910003460 diamond Inorganic materials 0.000 description 1
- 125000004987 dibenzofuryl group Chemical group C1(=CC=CC=2OC3=C(C21)C=CC=C3)* 0.000 description 1
- 125000004988 dibenzothienyl group Chemical group C1(=CC=CC=2SC3=C(C21)C=CC=C3)* 0.000 description 1
- 125000001664 diethylamino group Chemical group [H]C([H])([H])C([H])([H])N(*)C([H])([H])C([H])([H])[H] 0.000 description 1
- 125000001028 difluoromethyl group Chemical group [H]C(F)(F)* 0.000 description 1
- 125000002147 dimethylamino group Chemical group [H]C([H])([H])N(*)C([H])([H])[H] 0.000 description 1
- 238000007598 dipping method Methods 0.000 description 1
- 230000000694 effects Effects 0.000 description 1
- 238000010893 electron trap Methods 0.000 description 1
- 238000000295 emission spectrum Methods 0.000 description 1
- 239000003822 epoxy resin Substances 0.000 description 1
- 125000002534 ethynyl group Chemical group [H]C#C* 0.000 description 1
- 125000003993 fluoranthen-1-yl group Chemical group [H]C1=C([H])C([H])=C2C(=C1[H])C1=C([H])C([H])=C([H])C3=C([H])C([H])=C(*)C2=C13 0.000 description 1
- 150000002219 fluoranthenes Chemical class 0.000 description 1
- 125000001153 fluoro group Chemical group F* 0.000 description 1
- 125000004216 fluoromethyl group Chemical group [H]C([H])(F)* 0.000 description 1
- 125000002541 furyl group Chemical group 0.000 description 1
- 239000007789 gas Substances 0.000 description 1
- PCHJSUWPFVWCPO-UHFFFAOYSA-N gold Chemical compound [Au] PCHJSUWPFVWCPO-UHFFFAOYSA-N 0.000 description 1
- 229910052737 gold Inorganic materials 0.000 description 1
- 239000010931 gold Substances 0.000 description 1
- 230000005283 ground state Effects 0.000 description 1
- 150000002366 halogen compounds Chemical class 0.000 description 1
- LNEPOXFFQSENCJ-UHFFFAOYSA-N haloperidol Chemical compound C1CC(O)(C=2C=CC(Cl)=CC=2)CCN1CCCC(=O)C1=CC=C(F)C=C1 LNEPOXFFQSENCJ-UHFFFAOYSA-N 0.000 description 1
- 125000002192 heptalenyl group Chemical group 0.000 description 1
- 229940083761 high-ceiling diuretics pyrazolone derivative Drugs 0.000 description 1
- 238000004770 highest occupied molecular orbital Methods 0.000 description 1
- 150000007857 hydrazones Chemical class 0.000 description 1
- 125000002887 hydroxy group Chemical group [H]O* 0.000 description 1
- 125000004029 hydroxymethyl group Chemical group [H]OC([H])([H])* 0.000 description 1
- 150000002460 imidazoles Chemical class 0.000 description 1
- 230000006872 improvement Effects 0.000 description 1
- 229910052738 indium Inorganic materials 0.000 description 1
- APFVFJFRJDLVQX-UHFFFAOYSA-N indium atom Chemical compound [In] APFVFJFRJDLVQX-UHFFFAOYSA-N 0.000 description 1
- LHJOPRPDWDXEIY-UHFFFAOYSA-N indium lithium Chemical compound [Li].[In] LHJOPRPDWDXEIY-UHFFFAOYSA-N 0.000 description 1
- 229910003437 indium oxide Inorganic materials 0.000 description 1
- PJXISJQVUVHSOJ-UHFFFAOYSA-N indium(iii) oxide Chemical compound [O-2].[O-2].[O-2].[In+3].[In+3] PJXISJQVUVHSOJ-UHFFFAOYSA-N 0.000 description 1
- 125000003406 indolizinyl group Chemical group C=1(C=CN2C=CC=CC12)* 0.000 description 1
- 238000007641 inkjet printing Methods 0.000 description 1
- 229910010272 inorganic material Inorganic materials 0.000 description 1
- 239000011147 inorganic material Substances 0.000 description 1
- 229940079865 intestinal antiinfectives imidazole derivative Drugs 0.000 description 1
- 239000011630 iodine Substances 0.000 description 1
- 125000000959 isobutyl group Chemical group [H]C([H])([H])C([H])(C([H])([H])[H])C([H])([H])* 0.000 description 1
- 125000001972 isopentyl group Chemical group [H]C([H])([H])C([H])(C([H])([H])[H])C([H])([H])C([H])([H])* 0.000 description 1
- 125000001449 isopropyl group Chemical group [H]C([H])([H])C([H])(*)C([H])([H])[H] 0.000 description 1
- 125000005956 isoquinolyl group Chemical group 0.000 description 1
- 239000011133 lead Substances 0.000 description 1
- 229910052744 lithium Inorganic materials 0.000 description 1
- 125000000040 m-tolyl group Chemical group [H]C1=C([H])C(*)=C([H])C(=C1[H])C([H])([H])[H] 0.000 description 1
- 229910052749 magnesium Inorganic materials 0.000 description 1
- 239000011777 magnesium Substances 0.000 description 1
- SJCKRGFTWFGHGZ-UHFFFAOYSA-N magnesium silver Chemical compound [Mg].[Ag] SJCKRGFTWFGHGZ-UHFFFAOYSA-N 0.000 description 1
- 235000021184 main course Nutrition 0.000 description 1
- WPBNNNQJVZRUHP-UHFFFAOYSA-L manganese(2+);methyl n-[[2-(methoxycarbonylcarbamothioylamino)phenyl]carbamothioyl]carbamate;n-[2-(sulfidocarbothioylamino)ethyl]carbamodithioate Chemical compound [Mn+2].[S-]C(=S)NCCNC([S-])=S.COC(=O)NC(=S)NC1=CC=CC=C1NC(=S)NC(=O)OC WPBNNNQJVZRUHP-UHFFFAOYSA-L 0.000 description 1
- 125000005395 methacrylic acid group Chemical group 0.000 description 1
- 125000006384 methylpyridyl group Chemical group 0.000 description 1
- 230000004048 modification Effects 0.000 description 1
- 238000012986 modification Methods 0.000 description 1
- 238000004776 molecular orbital Methods 0.000 description 1
- 125000004108 n-butyl group Chemical group [H]C([H])([H])C([H])([H])C([H])([H])C([H])([H])* 0.000 description 1
- 125000003136 n-heptyl group Chemical group [H]C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])* 0.000 description 1
- 125000001280 n-hexyl group Chemical group C(CCCCC)* 0.000 description 1
- 125000000740 n-pentyl group Chemical group [H]C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])* 0.000 description 1
- 125000004123 n-propyl group Chemical group [H]C([H])([H])C([H])([H])C([H])([H])* 0.000 description 1
- 150000002790 naphthalenes Chemical class 0.000 description 1
- 125000001624 naphthyl group Chemical group 0.000 description 1
- 125000004593 naphthyridinyl group Chemical group N1=C(C=CC2=CC=CN=C12)* 0.000 description 1
- 125000001971 neopentyl group Chemical group [H]C([*])([H])C(C([H])([H])[H])(C([H])([H])[H])C([H])([H])[H] 0.000 description 1
- 229910052759 nickel Inorganic materials 0.000 description 1
- VOFUROIFQGPCGE-UHFFFAOYSA-N nile red Chemical compound C1=CC=C2C3=NC4=CC=C(N(CC)CC)C=C4OC3=CC(=O)C2=C1 VOFUROIFQGPCGE-UHFFFAOYSA-N 0.000 description 1
- 150000004767 nitrides Chemical class 0.000 description 1
- 125000000449 nitro group Chemical group [O-][N+](*)=O 0.000 description 1
- 125000002868 norbornyl group Chemical group C12(CCC(CC1)C2)* 0.000 description 1
- 125000001715 oxadiazolyl group Chemical group 0.000 description 1
- AICOOMRHRUFYCM-ZRRPKQBOSA-N oxazine, 1 Chemical compound C([C@@H]1[C@H](C(C[C@]2(C)[C@@H]([C@H](C)N(C)C)[C@H](O)C[C@]21C)=O)CC1=CC2)C[C@H]1[C@@]1(C)[C@H]2N=C(C(C)C)OC1 AICOOMRHRUFYCM-ZRRPKQBOSA-N 0.000 description 1
- 125000002971 oxazolyl group Chemical group 0.000 description 1
- 238000007254 oxidation reaction Methods 0.000 description 1
- 229910052763 palladium Inorganic materials 0.000 description 1
- 125000005003 perfluorobutyl group Chemical group FC(F)(F)C(F)(F)C(F)(F)C(F)(F)* 0.000 description 1
- 125000005004 perfluoroethyl group Chemical group FC(F)(F)C(F)(F)* 0.000 description 1
- 125000005009 perfluoropropyl group Chemical group FC(C(C(F)(F)F)(F)F)(F)* 0.000 description 1
- 230000002093 peripheral effect Effects 0.000 description 1
- 150000002987 phenanthrenes Chemical class 0.000 description 1
- 125000004934 phenanthridinyl group Chemical group C1(=CC=CC2=NC=C3C=CC=CC3=C12)* 0.000 description 1
- 150000005041 phenanthrolines Chemical class 0.000 description 1
- 125000005561 phenanthryl group Chemical group 0.000 description 1
- 239000005011 phenolic resin Substances 0.000 description 1
- 150000004986 phenylenediamines Chemical class 0.000 description 1
- 238000005424 photoluminescence Methods 0.000 description 1
- 238000000103 photoluminescence spectrum Methods 0.000 description 1
- IEQIEDJGQAUEQZ-UHFFFAOYSA-N phthalocyanine Chemical class N1C(N=C2C3=CC=CC=C3C(N=C3C4=CC=CC=C4C(=N4)N3)=N2)=C(C=CC=C2)C2=C1N=C1C2=CC=CC=C2C4=N1 IEQIEDJGQAUEQZ-UHFFFAOYSA-N 0.000 description 1
- 125000001388 picenyl group Chemical group C1(=CC=CC2=CC=C3C4=CC=C5C=CC=CC5=C4C=CC3=C21)* 0.000 description 1
- 239000002985 plastic film Substances 0.000 description 1
- 239000004014 plasticizer Substances 0.000 description 1
- 229910052697 platinum Inorganic materials 0.000 description 1
- 229920000553 poly(phenylenevinylene) Polymers 0.000 description 1
- 229920002492 poly(sulfone) Polymers 0.000 description 1
- 229920002037 poly(vinyl butyral) polymer Polymers 0.000 description 1
- 229920006122 polyamide resin Polymers 0.000 description 1
- 229920000767 polyaniline Polymers 0.000 description 1
- 229920001230 polyarylate Polymers 0.000 description 1
- 229920002857 polybutadiene Polymers 0.000 description 1
- 229920005668 polycarbonate resin Polymers 0.000 description 1
- 239000004431 polycarbonate resin Substances 0.000 description 1
- 125000003367 polycyclic group Chemical group 0.000 description 1
- 229920000647 polyepoxide Polymers 0.000 description 1
- 229920001225 polyester resin Polymers 0.000 description 1
- 239000004645 polyester resin Substances 0.000 description 1
- 229920006393 polyether sulfone Polymers 0.000 description 1
- 229920000573 polyethylene Polymers 0.000 description 1
- 229920013716 polyethylene resin Polymers 0.000 description 1
- 229920001721 polyimide Polymers 0.000 description 1
- 239000009719 polyimide resin Substances 0.000 description 1
- 229920000642 polymer Polymers 0.000 description 1
- 229920006324 polyoxymethylene Polymers 0.000 description 1
- 229920000069 polyphenylene sulfide Polymers 0.000 description 1
- 229920000128 polypyrrole Polymers 0.000 description 1
- 229920005749 polyurethane resin Polymers 0.000 description 1
- 150000004033 porphyrin derivatives Chemical class 0.000 description 1
- BITYAPCSNKJESK-UHFFFAOYSA-N potassiosodium Chemical compound [Na].[K] BITYAPCSNKJESK-UHFFFAOYSA-N 0.000 description 1
- 229910052700 potassium Inorganic materials 0.000 description 1
- 239000011591 potassium Substances 0.000 description 1
- 239000002243 precursor Substances 0.000 description 1
- 125000001436 propyl group Chemical group [H]C([*])([H])C([H])([H])C([H])([H])[H] 0.000 description 1
- 239000012264 purified product Substances 0.000 description 1
- 125000003373 pyrazinyl group Chemical group 0.000 description 1
- JEXVQSWXXUJEMA-UHFFFAOYSA-N pyrazol-3-one Chemical class O=C1C=CN=N1 JEXVQSWXXUJEMA-UHFFFAOYSA-N 0.000 description 1
- 150000003219 pyrazolines Chemical class 0.000 description 1
- 125000001725 pyrenyl group Chemical group 0.000 description 1
- 125000002098 pyridazinyl group Chemical group 0.000 description 1
- 125000000714 pyrimidinyl group Chemical group 0.000 description 1
- 239000010453 quartz Substances 0.000 description 1
- 125000002294 quinazolinyl group Chemical group N1=C(N=CC2=CC=CC=C12)* 0.000 description 1
- 125000002943 quinolinyl group Chemical class N1=C(C=CC2=CC=CC=C12)* 0.000 description 1
- 125000005493 quinolyl group Chemical group 0.000 description 1
- 238000011160 research Methods 0.000 description 1
- 230000004044 response Effects 0.000 description 1
- YYMBJDOZVAITBP-UHFFFAOYSA-N rubrene Chemical compound C1=CC=CC=C1C(C1=C(C=2C=CC=CC=2)C2=CC=CC=C2C(C=2C=CC=CC=2)=C11)=C(C=CC=C2)C2=C1C1=CC=CC=C1 YYMBJDOZVAITBP-UHFFFAOYSA-N 0.000 description 1
- 229910052707 ruthenium Inorganic materials 0.000 description 1
- 125000002914 sec-butyl group Chemical group [H]C([H])([H])C([H])([H])C([H])(*)C([H])([H])[H] 0.000 description 1
- 229910052711 selenium Inorganic materials 0.000 description 1
- 239000011669 selenium Substances 0.000 description 1
- VYPSYNLAJGMNEJ-UHFFFAOYSA-N silicon dioxide Inorganic materials O=[Si]=O VYPSYNLAJGMNEJ-UHFFFAOYSA-N 0.000 description 1
- 229910052708 sodium Inorganic materials 0.000 description 1
- 239000011734 sodium Substances 0.000 description 1
- 238000001179 sorption measurement Methods 0.000 description 1
- 230000007480 spreading Effects 0.000 description 1
- 238000003892 spreading Methods 0.000 description 1
- 238000001308 synthesis method Methods 0.000 description 1
- 230000002194 synthesizing effect Effects 0.000 description 1
- 150000003518 tetracenes Chemical class 0.000 description 1
- 125000001935 tetracenyl group Chemical group C1(=CC=CC2=CC3=CC4=CC=CC=C4C=C3C=C12)* 0.000 description 1
- 150000004867 thiadiazoles Chemical class 0.000 description 1
- 125000001113 thiadiazolyl group Chemical group 0.000 description 1
- 150000007979 thiazole derivatives Chemical class 0.000 description 1
- 125000000335 thiazolyl group Chemical group 0.000 description 1
- 239000011135 tin Substances 0.000 description 1
- XOLBLPGZBRYERU-UHFFFAOYSA-N tin dioxide Chemical compound O=[Sn]=O XOLBLPGZBRYERU-UHFFFAOYSA-N 0.000 description 1
- 229910001887 tin oxide Inorganic materials 0.000 description 1
- 239000010936 titanium Substances 0.000 description 1
- 229910052719 titanium Inorganic materials 0.000 description 1
- 238000012546 transfer Methods 0.000 description 1
- TVIVIEFSHFOWTE-UHFFFAOYSA-K tri(quinolin-8-yloxy)alumane Chemical compound [Al+3].C1=CN=C2C([O-])=CC=CC2=C1.C1=CN=C2C([O-])=CC=CC2=C1.C1=CN=C2C([O-])=CC=CC2=C1 TVIVIEFSHFOWTE-UHFFFAOYSA-K 0.000 description 1
- 125000005259 triarylamine group Chemical group 0.000 description 1
- 150000003918 triazines Chemical class 0.000 description 1
- 125000003866 trichloromethyl group Chemical group ClC(Cl)(Cl)* 0.000 description 1
- 125000002023 trifluoromethyl group Chemical group FC(F)(F)* 0.000 description 1
- 125000003960 triphenylenyl group Chemical group C1(=CC=CC=2C3=CC=CC=C3C3=CC=CC=C3C12)* 0.000 description 1
- WFKWXMTUELFFGS-UHFFFAOYSA-N tungsten Chemical compound [W] WFKWXMTUELFFGS-UHFFFAOYSA-N 0.000 description 1
- 229910052721 tungsten Inorganic materials 0.000 description 1
- 239000010937 tungsten Substances 0.000 description 1
- 238000004506 ultrasonic cleaning Methods 0.000 description 1
- 238000007738 vacuum evaporation Methods 0.000 description 1
- 229910052720 vanadium Inorganic materials 0.000 description 1
- GPPXJZIENCGNKB-UHFFFAOYSA-N vanadium Chemical compound [V]#[V] GPPXJZIENCGNKB-UHFFFAOYSA-N 0.000 description 1
- 229920002554 vinyl polymer Polymers 0.000 description 1
- 229910052727 yttrium Inorganic materials 0.000 description 1
- VWQVUPCCIRVNHF-UHFFFAOYSA-N yttrium atom Chemical compound [Y] VWQVUPCCIRVNHF-UHFFFAOYSA-N 0.000 description 1
- YVTHLONGBIQYBO-UHFFFAOYSA-N zinc indium(3+) oxygen(2-) Chemical compound [O--].[Zn++].[In+3] YVTHLONGBIQYBO-UHFFFAOYSA-N 0.000 description 1
- 239000011787 zinc oxide Substances 0.000 description 1
Classifications
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07C—ACYCLIC OR CARBOCYCLIC COMPOUNDS
- C07C22/00—Cyclic compounds containing halogen atoms bound to an acyclic carbon atom
- C07C22/02—Cyclic compounds containing halogen atoms bound to an acyclic carbon atom having unsaturation in the rings
- C07C22/04—Cyclic compounds containing halogen atoms bound to an acyclic carbon atom having unsaturation in the rings containing six-membered aromatic rings
- C07C22/08—Cyclic compounds containing halogen atoms bound to an acyclic carbon atom having unsaturation in the rings containing six-membered aromatic rings containing fluorine
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07C—ACYCLIC OR CARBOCYCLIC COMPOUNDS
- C07C13/00—Cyclic hydrocarbons containing rings other than, or in addition to, six-membered aromatic rings
- C07C13/28—Polycyclic hydrocarbons or acyclic hydrocarbon derivatives thereof
- C07C13/32—Polycyclic hydrocarbons or acyclic hydrocarbon derivatives thereof with condensed rings
- C07C13/62—Polycyclic hydrocarbons or acyclic hydrocarbon derivatives thereof with condensed rings with more than three condensed rings
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07C—ACYCLIC OR CARBOCYCLIC COMPOUNDS
- C07C25/00—Compounds containing at least one halogen atom bound to a six-membered aromatic ring
- C07C25/18—Polycyclic aromatic halogenated hydrocarbons
- C07C25/22—Polycyclic aromatic halogenated hydrocarbons with condensed rings
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07C—ACYCLIC OR CARBOCYCLIC COMPOUNDS
- C07C255/00—Carboxylic acid nitriles
- C07C255/49—Carboxylic acid nitriles having cyano groups bound to carbon atoms of six-membered aromatic rings of a carbon skeleton
- C07C255/50—Carboxylic acid nitriles having cyano groups bound to carbon atoms of six-membered aromatic rings of a carbon skeleton to carbon atoms of non-condensed six-membered aromatic rings
- C07C255/51—Carboxylic acid nitriles having cyano groups bound to carbon atoms of six-membered aromatic rings of a carbon skeleton to carbon atoms of non-condensed six-membered aromatic rings containing at least two cyano groups bound to the carbon skeleton
-
- C—CHEMISTRY; METALLURGY
- C09—DYES; PAINTS; POLISHES; NATURAL RESINS; ADHESIVES; COMPOSITIONS NOT OTHERWISE PROVIDED FOR; APPLICATIONS OF MATERIALS NOT OTHERWISE PROVIDED FOR
- C09K—MATERIALS FOR MISCELLANEOUS APPLICATIONS, NOT PROVIDED FOR ELSEWHERE
- C09K11/00—Luminescent, e.g. electroluminescent, chemiluminescent materials
- C09K11/06—Luminescent, e.g. electroluminescent, chemiluminescent materials containing organic luminescent materials
-
- H—ELECTRICITY
- H05—ELECTRIC TECHNIQUES NOT OTHERWISE PROVIDED FOR
- H05B—ELECTRIC HEATING; ELECTRIC LIGHT SOURCES NOT OTHERWISE PROVIDED FOR; CIRCUIT ARRANGEMENTS FOR ELECTRIC LIGHT SOURCES, IN GENERAL
- H05B33/00—Electroluminescent light sources
- H05B33/12—Light sources with substantially two-dimensional radiating surfaces
- H05B33/14—Light sources with substantially two-dimensional radiating surfaces characterised by the chemical or physical composition or the arrangement of the electroluminescent material, or by the simultaneous addition of the electroluminescent material in or onto the light source
-
- H—ELECTRICITY
- H10—SEMICONDUCTOR DEVICES; ELECTRIC SOLID-STATE DEVICES NOT OTHERWISE PROVIDED FOR
- H10K—ORGANIC ELECTRIC SOLID-STATE DEVICES
- H10K50/00—Organic light-emitting devices
-
- H—ELECTRICITY
- H10—SEMICONDUCTOR DEVICES; ELECTRIC SOLID-STATE DEVICES NOT OTHERWISE PROVIDED FOR
- H10K—ORGANIC ELECTRIC SOLID-STATE DEVICES
- H10K50/00—Organic light-emitting devices
- H10K50/10—OLEDs or polymer light-emitting diodes [PLED]
- H10K50/11—OLEDs or polymer light-emitting diodes [PLED] characterised by the electroluminescent [EL] layers
-
- H—ELECTRICITY
- H10—SEMICONDUCTOR DEVICES; ELECTRIC SOLID-STATE DEVICES NOT OTHERWISE PROVIDED FOR
- H10K—ORGANIC ELECTRIC SOLID-STATE DEVICES
- H10K85/00—Organic materials used in the body or electrodes of devices covered by this subclass
- H10K85/60—Organic compounds having low molecular weight
- H10K85/615—Polycyclic condensed aromatic hydrocarbons, e.g. anthracene
- H10K85/622—Polycyclic condensed aromatic hydrocarbons, e.g. anthracene containing four rings, e.g. pyrene
-
- H—ELECTRICITY
- H10—SEMICONDUCTOR DEVICES; ELECTRIC SOLID-STATE DEVICES NOT OTHERWISE PROVIDED FOR
- H10K—ORGANIC ELECTRIC SOLID-STATE DEVICES
- H10K85/00—Organic materials used in the body or electrodes of devices covered by this subclass
- H10K85/60—Organic compounds having low molecular weight
- H10K85/615—Polycyclic condensed aromatic hydrocarbons, e.g. anthracene
- H10K85/623—Polycyclic condensed aromatic hydrocarbons, e.g. anthracene containing five rings, e.g. pentacene
-
- H—ELECTRICITY
- H10—SEMICONDUCTOR DEVICES; ELECTRIC SOLID-STATE DEVICES NOT OTHERWISE PROVIDED FOR
- H10K—ORGANIC ELECTRIC SOLID-STATE DEVICES
- H10K85/00—Organic materials used in the body or electrodes of devices covered by this subclass
- H10K85/60—Organic compounds having low molecular weight
- H10K85/615—Polycyclic condensed aromatic hydrocarbons, e.g. anthracene
- H10K85/626—Polycyclic condensed aromatic hydrocarbons, e.g. anthracene containing more than one polycyclic condensed aromatic rings, e.g. bis-anthracene
-
- H—ELECTRICITY
- H10—SEMICONDUCTOR DEVICES; ELECTRIC SOLID-STATE DEVICES NOT OTHERWISE PROVIDED FOR
- H10K—ORGANIC ELECTRIC SOLID-STATE DEVICES
- H10K85/00—Organic materials used in the body or electrodes of devices covered by this subclass
- H10K85/60—Organic compounds having low molecular weight
- H10K85/631—Amine compounds having at least two aryl rest on at least one amine-nitrogen atom, e.g. triphenylamine
- H10K85/633—Amine compounds having at least two aryl rest on at least one amine-nitrogen atom, e.g. triphenylamine comprising polycyclic condensed aromatic hydrocarbons as substituents on the nitrogen atom
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07C—ACYCLIC OR CARBOCYCLIC COMPOUNDS
- C07C2603/00—Systems containing at least three condensed rings
- C07C2603/02—Ortho- or ortho- and peri-condensed systems
- C07C2603/40—Ortho- or ortho- and peri-condensed systems containing four condensed rings
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07C—ACYCLIC OR CARBOCYCLIC COMPOUNDS
- C07C2603/00—Systems containing at least three condensed rings
- C07C2603/02—Ortho- or ortho- and peri-condensed systems
- C07C2603/52—Ortho- or ortho- and peri-condensed systems containing five condensed rings
-
- C—CHEMISTRY; METALLURGY
- C09—DYES; PAINTS; POLISHES; NATURAL RESINS; ADHESIVES; COMPOSITIONS NOT OTHERWISE PROVIDED FOR; APPLICATIONS OF MATERIALS NOT OTHERWISE PROVIDED FOR
- C09K—MATERIALS FOR MISCELLANEOUS APPLICATIONS, NOT PROVIDED FOR ELSEWHERE
- C09K2211/00—Chemical nature of organic luminescent or tenebrescent compounds
- C09K2211/10—Non-macromolecular compounds
- C09K2211/1003—Carbocyclic compounds
- C09K2211/1007—Non-condensed systems
-
- C—CHEMISTRY; METALLURGY
- C09—DYES; PAINTS; POLISHES; NATURAL RESINS; ADHESIVES; COMPOSITIONS NOT OTHERWISE PROVIDED FOR; APPLICATIONS OF MATERIALS NOT OTHERWISE PROVIDED FOR
- C09K—MATERIALS FOR MISCELLANEOUS APPLICATIONS, NOT PROVIDED FOR ELSEWHERE
- C09K2211/00—Chemical nature of organic luminescent or tenebrescent compounds
- C09K2211/10—Non-macromolecular compounds
- C09K2211/1003—Carbocyclic compounds
- C09K2211/1011—Condensed systems
-
- C—CHEMISTRY; METALLURGY
- C09—DYES; PAINTS; POLISHES; NATURAL RESINS; ADHESIVES; COMPOSITIONS NOT OTHERWISE PROVIDED FOR; APPLICATIONS OF MATERIALS NOT OTHERWISE PROVIDED FOR
- C09K—MATERIALS FOR MISCELLANEOUS APPLICATIONS, NOT PROVIDED FOR ELSEWHERE
- C09K2211/00—Chemical nature of organic luminescent or tenebrescent compounds
- C09K2211/10—Non-macromolecular compounds
- C09K2211/1003—Carbocyclic compounds
- C09K2211/1014—Carbocyclic compounds bridged by heteroatoms, e.g. N, P, Si or B
-
- C—CHEMISTRY; METALLURGY
- C09—DYES; PAINTS; POLISHES; NATURAL RESINS; ADHESIVES; COMPOSITIONS NOT OTHERWISE PROVIDED FOR; APPLICATIONS OF MATERIALS NOT OTHERWISE PROVIDED FOR
- C09K—MATERIALS FOR MISCELLANEOUS APPLICATIONS, NOT PROVIDED FOR ELSEWHERE
- C09K2211/00—Chemical nature of organic luminescent or tenebrescent compounds
- C09K2211/10—Non-macromolecular compounds
- C09K2211/1018—Heterocyclic compounds
- C09K2211/1025—Heterocyclic compounds characterised by ligands
- C09K2211/1029—Heterocyclic compounds characterised by ligands containing one nitrogen atom as the heteroatom
Definitions
- This invention relates to an organic luminescent device (organic EL (electroluminescent) device) using a benzo [k] fluoranthene compound, and relates to the benzo [k] fluoranthene compound.
- Organic luminescent devices are devices in which a thin film containing a fluorescent organic compound is held between an anode and a cathode, electrons and holes are injected from each electrode to produce excitons of the fluorescent organic compound, and the light is utilized that is emitted when the excitons return to the ground state.
- Recent progress seen in such organic luminescent devices is noticeable, and they have characteristics capable of realizing luminescent devices which can achieving high luminance at a low applied voltage, a variety of luminescent wavelengths and high-speed response, and are thin and light-weight. Thus, there is a possibility that they are applied to a wide variety of uses.
- An object of the present invention is to provide a compound for an organic luminescent device which takes on luminous hues with a very high degree of purity and has optical power with high efficiency, high luminance and a long life, and to provide an organic luminescent device using such a compound.
- a further object of the present invention is to . provide an organic luminescent device which can be fabricated with ease and can be manufactured at relatively low costs.
- the present inventors have conducted extensive research in order to solve the problems stated above. As a result, they have accomplished the present invention .
- the present invention provides an organic luminescent device constituted of an anode and a cathode at least one of which is transparent or semitransparent, and a layer containing an organic compound, held between a pair of electrodes consisting of the anode and the cathode, wherein; the layer containing an organic compound contains a benzo [k] fluoranthene compound represented by the following general formula (1) .
- Ri is a group selected from the group consisting of a substituted or unsubstituted alkyl group, a substituted or unsubstituted aralkyl group and a substituted or unsubstituted heterocyclic ring group, and Ri' s may be the same or different;
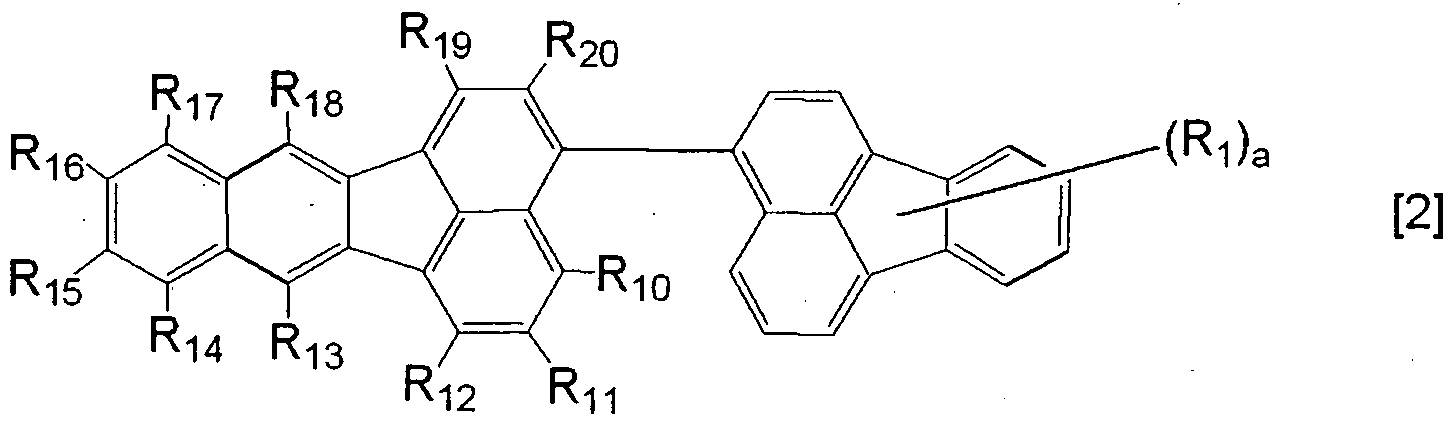
- Rio to R 2 O are each independently a group selected from the group consisting of a hydrogen atom, a halogen atom, a substituted or unsubstituted alkyl group, a substituted or unsubstituted aralkyl group, a substituted or unsubstituted phenyl group, a substituted or unsubstituted condensed bicyclic aromatic group and a substituted or unsubstituted heterocyclic group; and a is an integer of 0 or more and 9 or less.
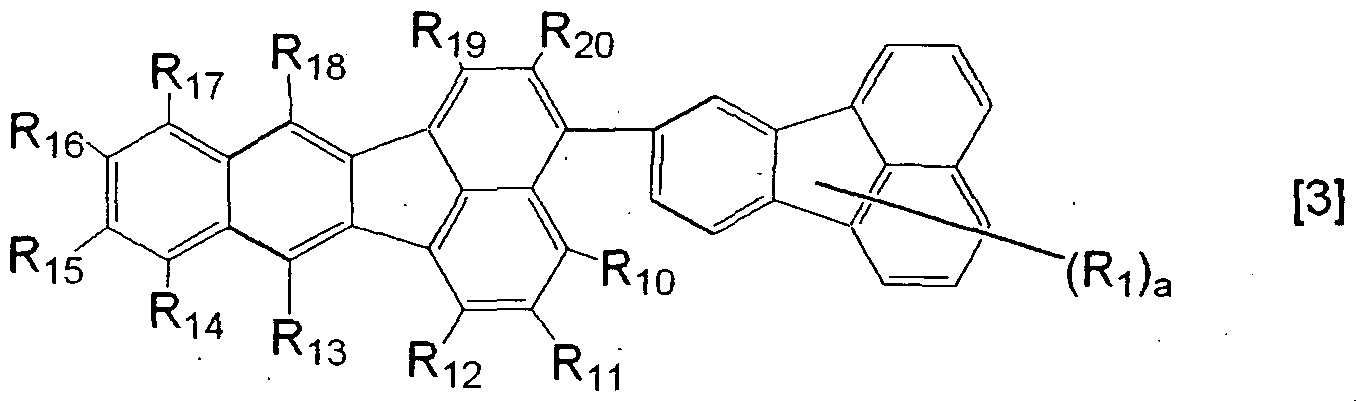
- the present invention also provides a benzo [k] fluoranthene compound represented by the following general formula (3).
- R 1 is a group selected from the group consisting of a substituted or unsubstituted alkyl group, a substituted or unsubstituted aralkyl group and a substituted or unsubstituted heterocyclic group, and Ri' s may be the same or different;
- Rio to R 2 O are each independently a group selected from the group consisting of a hydrogen atom, a halogen atom, a substituted or unsubstituted alkyl group, a substituted or unsubstituted aralkyl group, a substituted or unsubstituted alkoxyl group, a substituted or unsubstituted phenyl group, a substituted or unsubstituted condensed bicyclic aromatic group and a substituted or unsubstituted heterocyclic group; and a is an integer of 0 or more and 9 or less.
- the organic luminescent device and compound of the present invention affords highly efficient light emission at a low applied voltage, and also has high thermal stability and ensure superior durability.
- FIG. 1 is a sectional view showing an example of the organic luminescent device in the present invention.
- FIG. 2 is a graph showing a 1 H-NMR (CDCl 3 ) spectrum of Exemplary Compound 101.
- FIG. 3 is a graph showing a PL spectrum (excitation wavelength: 420 ran) of a toluene solution containing Exemplary Compound 101 in a concentration of 1 x 10 "5 mol/1.
- FIG. 4 is a graph showing a 1 H-NMR (CDCl 3 ) spectrum of Exemplary Compound H2-3.
- FIG. 5 is a graph showing a 1 H-NMR (CDCl 3 ) spectrum of Exemplary Compound H4-2.
- FIG. 6 is a graph showing a 1 H-NMR (CDCl 3 ) spectrum of Exemplary Compound H9-1.
- FIG . 7 is a graph showing a 1 H-NMR (CDCl 3 ) spectrum of Exemplary Compound H14 -3 .
- the benzo [k] fluoranthene compound of the present invention is represented by the general formula (1) .
- This compound can be used without regard to the substitution position of the fluoranthene ring at which the fluoranthene ring is bonded to the 3-position of the benzo [k] fluoranthene ring, and is specifically represented by any one of the following general formulas (2) and (3) and the general formulas (11) to (13) .
- a compound having the substituent at the ortho-position is particularly preferred in order to control concentration quenching due to cohesion. This is because the compound has a structure in which the phenyl rises from the plane of the benzofluoranthene ring in virtue of steric repulsion attributable to the substituent at the ortho- position.
- the benzo [ k] fluoranthene compound used in the present invention can be used as a material for organic luminescent devices.
- it when used for a light-emitting layer, it can be used alone in the light-emitting layer, and can be used as a dopant
- the light-emitting layer is formed of carrier-transporting host and guest materials, the main course that comes up with light emission consists of the following several steps.
- the desired energy movement and light emission in the respective steps take place through various deactivation processes and competitions.
- the present inventors have made various studies, and have discovered that a device using a compound in which fluoranthene and benzofluoranthene are combined with each other through one single bond especially as the host or guest of the light-emitting layer, emits blue light at high efficiency, keeps high luminance over a long period of time and is reduced in deterioration due to electrification.
- the molecular orbital has been calculated for 3- ( 8-fluoranthenyl ) -benzo [k] fluoranthene which is a compound having the basic skeleton of the benzo [k] fluoranthene compound used in the present invention.
- the LUMO orbital spreads from the fluoranthene to the benzofluoranthene and the HOMO orbital is localized at the benzofluoranthene .
- the present inventors conceived that it is possible to improve luminescent colors in virtue of the electron trapping performance and CT (charge transfer) performance attributable to the LUMO spreading from the fluoranthene to the benzofluoranthene .
- steric hindrance groups such as tert-butyl groups are introduced into the condensed-ring aromatic groups.
- the compound of the present invention can be used without regard to the substitution position of the fluoranthene ring at which the fluoranthene is bonded to the 3-position of the benzo [k] fluoranthene ring. Taking into account steric hindrance due to the peri- position, an effect on rotation restraint and further the process of synthesizing the compound, it is preferable that the fluoranthene ring is substituted at its 3-position.
- the substituted or unsubstituted alkyl group includes, but is not limited to, e.g., the following: A methyl group, a methyl-dl group, a methyl-d3 group, an ethyl group, an ethyl-d5 group, a n-propyl group, a n-butyl group, a n-pentyl group, a n-hexyl group, a n-heptyl group, a n-octyl group, a n-decyl group, an iso-propyl group, an iso-propyl-d7 group, an iso-butyl group, a sec-butyl group, a tert-butyl group, a tert-butyl-d9 group, an iso-pentyl group, a neopentyl group, a tert-octyl group, a fluoromethyl group
- the substituted or unsubstituted aralkyl group includes, but is not limited to, e.g., the following:
- the substituted or unsubstituted phenyl group includes, but is not limited to, e.g., the following: A phenyl group, a phenyl-d5 group, a 2- methylphenyl group, a 3-methylphenyl group, a 4- methylphenyl group, a 4-methoxyphenyl group, a 4- ethylphenyl group, a 2-fluorophenyl group, a 3- fluorophenyl group, a 4-fluorophenyl group, a 4- trifluoromethylphenyl group, a 3, 5-dimethylphenyl group, a 2, 6-dimethylphenyl group, a 2, 6-diethylphenyl group, a mesityl group, a 3-iso-propylphenyl group, a 3-tert- butylphenyl group, a 4-iso-propylphenyl group, a 4- tert-butylphenyl group, a 4-
- the substituted or unsubstituted condensed bicyclic aromatic group includes, but is not limited to, a naphthyl group, an azulene group and a heptalene group.
- the substituted or unsubstituted heterocyclic group include, but is not limited to, e.g., the following:
- the halogen atom includes a fluorine atom, a chlorine atom, a bromine atom and an iodine atom.
- a substituent (s ) the above substituents may each further have include (s), but is (are) not limited to, e.g., the following: Alkyl groups such as a methyl group, an ethyl group and a propyl group, aryl groups such as a "phenyl group and a biphenyl group, heterocyclic groups such as a thienyl group, a pyrrolyl group and a pyridyl group, amino groups such as a dimethylamino group, a diethylamino group, a dibenzylamino group, a diphenylamino group, a ditolylamino group and a dianisolylamino group, and the substituted or unsubstituted alkyl groups listed above; substituted or unsubstituted alk
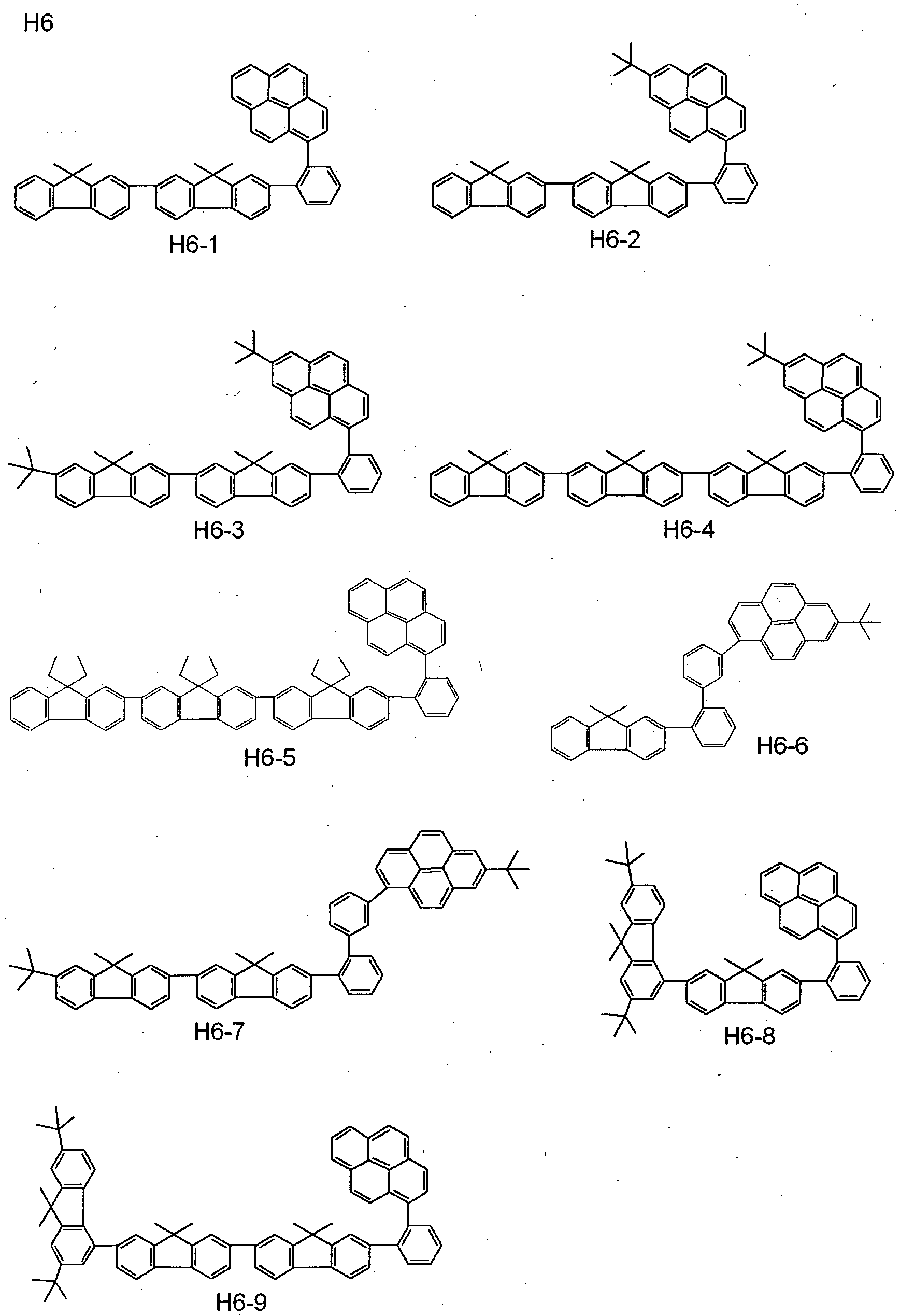
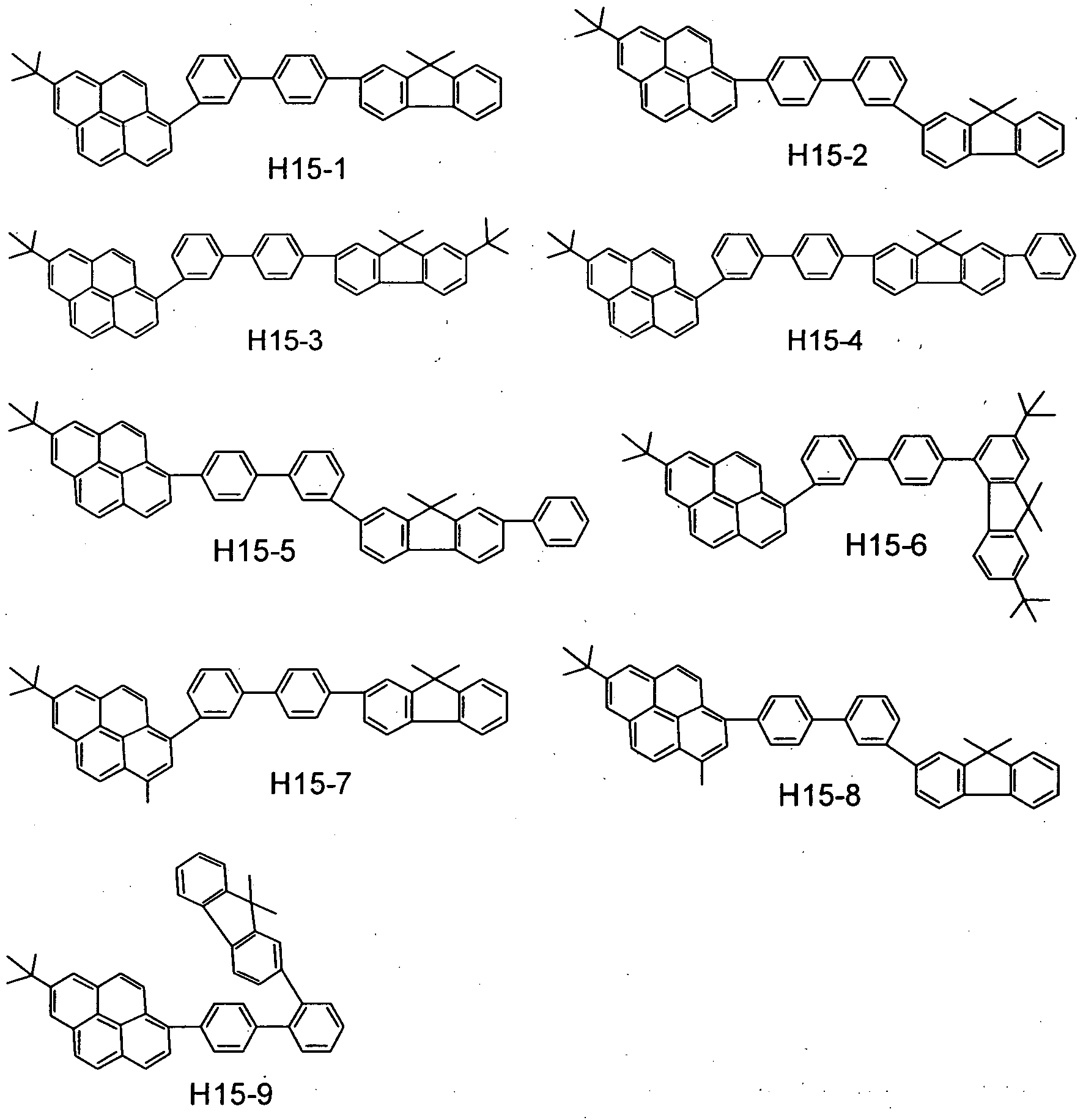
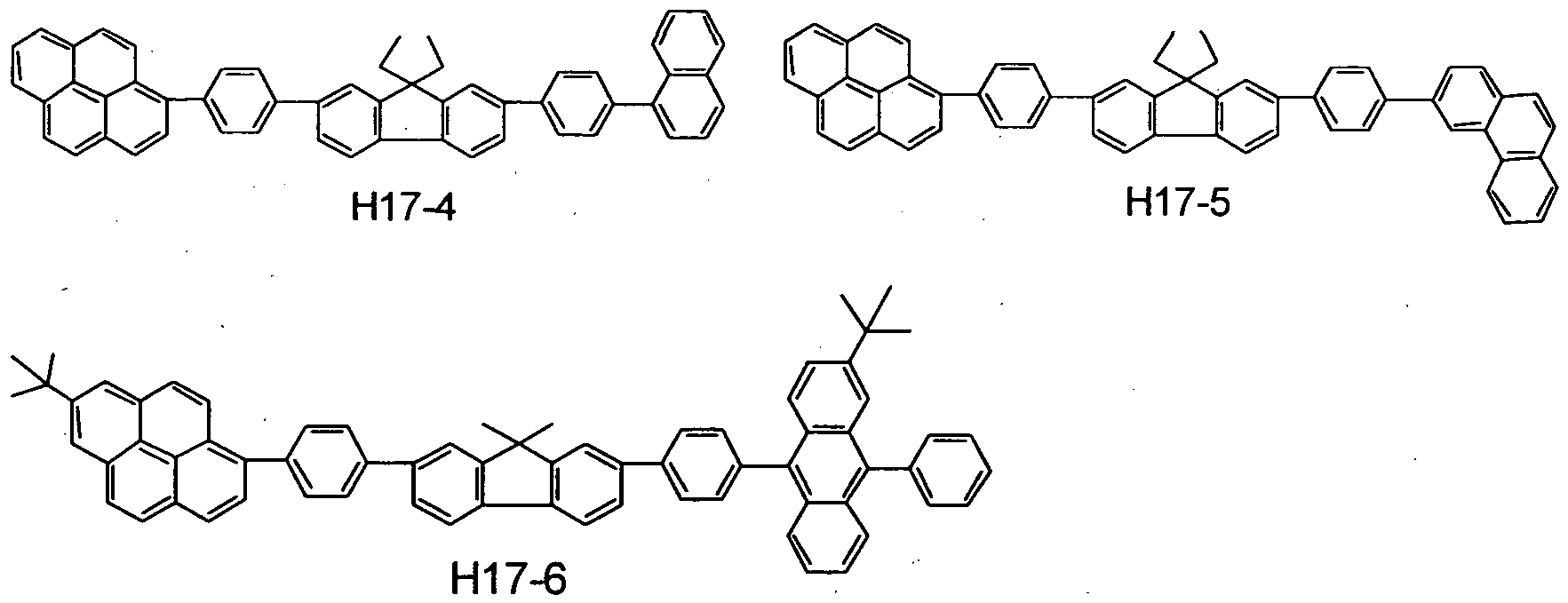
- the benzo [k] fluoranthene compound used in the present invention specifically include, but is not limited to, those shown in the following tables.
- the benzo [k] fluoranthene compound used in the present invention is represented by A-B.
- a and B the position at which these are combined with each other is shown. More specifically, as to Exemplary Compound 101, the combination is expressed as follows:
- 3-Bromobenzo [k] fluoranthene which is a precursor material of the benzo [ k] fluoranthene compound used in the present invention, can be synthesized by using a bromine compound of acenaphthylene.
- the bromine compound of acenaphthylene can be synthesized by oxidation reaction of 5-bromoacenaphthene as disclosed in, e.g., a document "Can. J. Chem., 70, 1015 (1992)".
- 5, 6-dibromoacenaphthene may be synthesized from 5, 6-dibromoacenaphthylene .
- the benzo [k] fluoranthene compound used in the present invention may be synthesized by, e.g., Suzuki- Miyaura coupling reaction of a bromine compound of corresponding benzo [k] fluoranthene with a pinacol borane compound derived from bromofluoranthene . It may similarly be synthesized by reacting a pinacol borane compound of benzofluoranthene with a bromine compound of fluoranthene . In place of the pinacol borane compound, boric acid may be used.
- Pinacol borane may be formed by reacting a halogen compound with 4,4,5,5- tetramethyl- [1, 3, 2] dioxabororane in, e.g., a toluene solvent and in the presence of triethylamine and a catalyst Ni (dppp) Cl .
- a tert-butyl group-substituted fluoranthene unit can be synthesized by Friedel-Crafts alkylation of bromofluoranthene .
- the organic luminescent device of the present invention has at least a pair of electrodes consisting of an anode and a cathode at least one of which is transparent or semitransparent, and one or two or more layers containing an organic compound, held between the pair of electrodes.
- at least one of the layer (s) containing an organic compound preferably at least one layer having a luminescent region, and more preferably a luminescent layer, contains at least one benzo [k] fluoranthene compound of the present invention, described above.
- the layer containing the benzo [k] fluoranthene compound may contain a second compound (a host material) .
- the host material is preferably a compound having a larger energy gap than the benzo [k] fluoranthene compound.
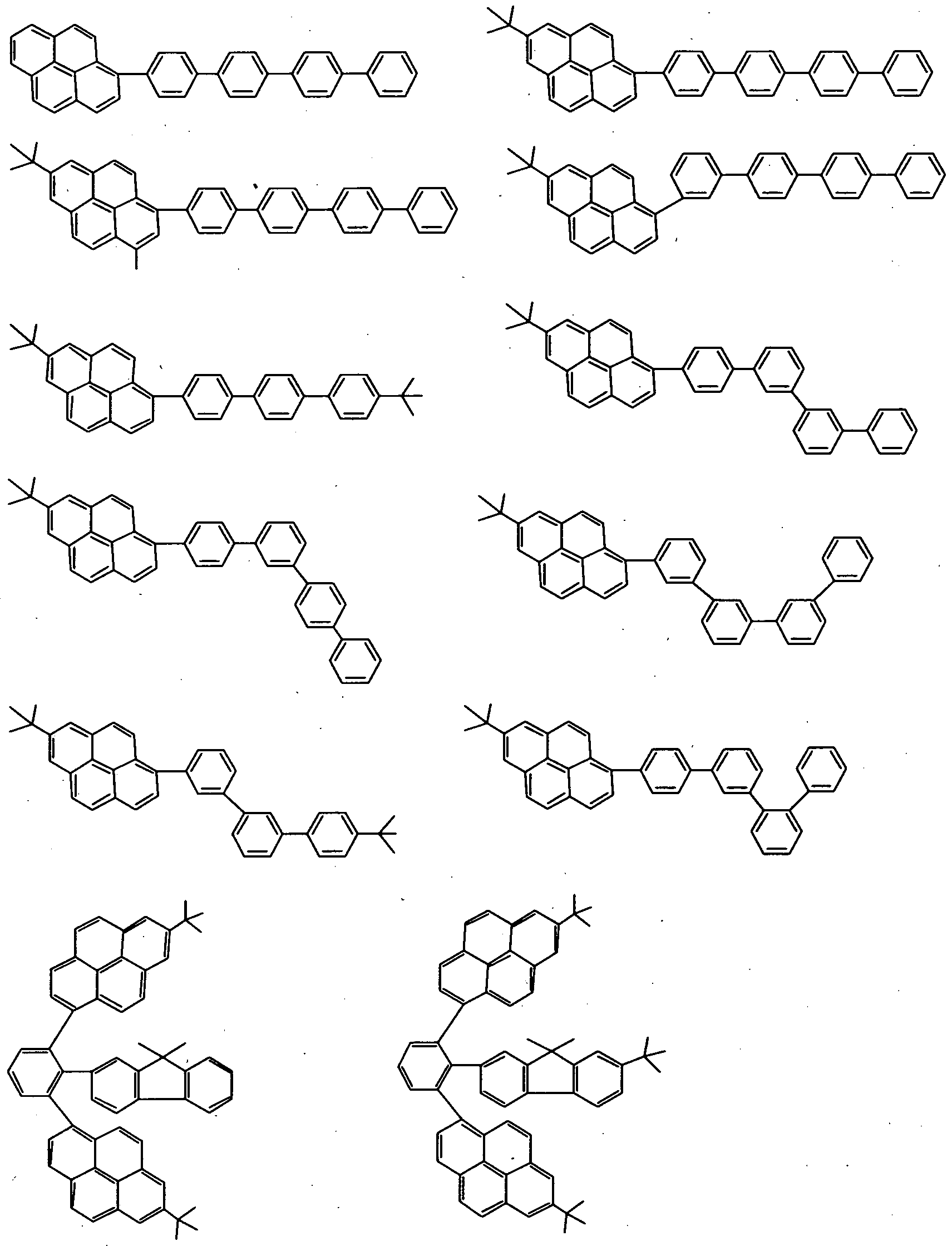
- the second compound includes, but is not limited to, e.g., such materials having a pyrene skeleton as shown below.
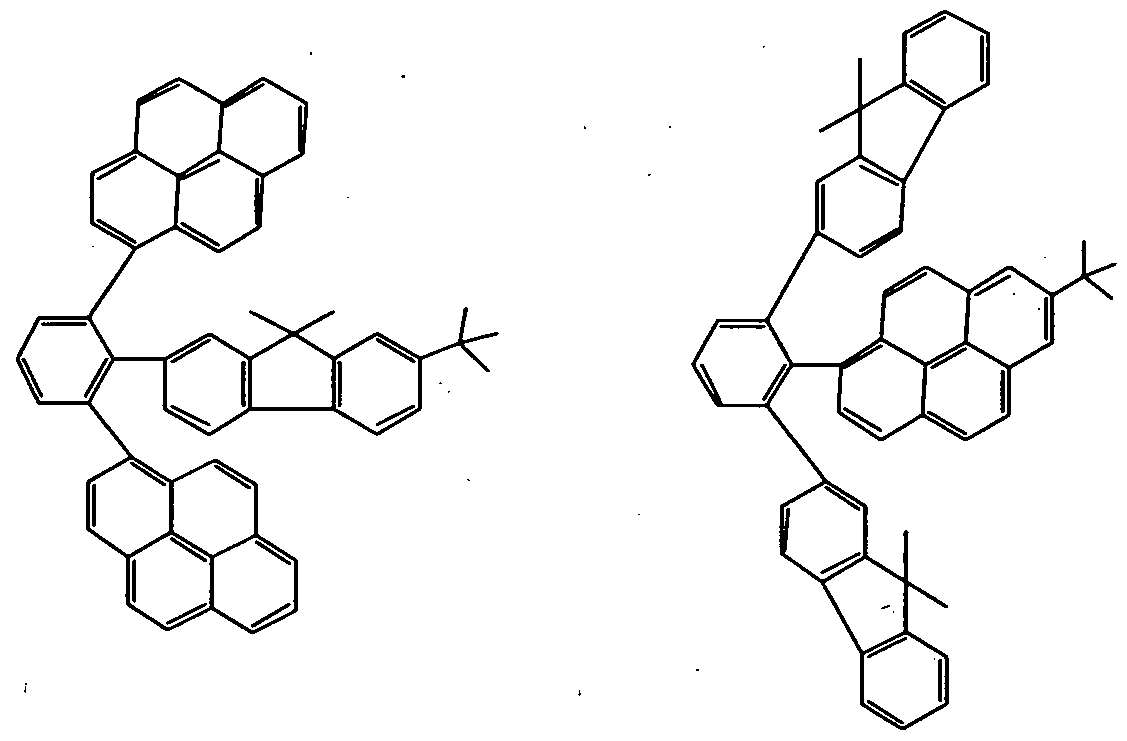
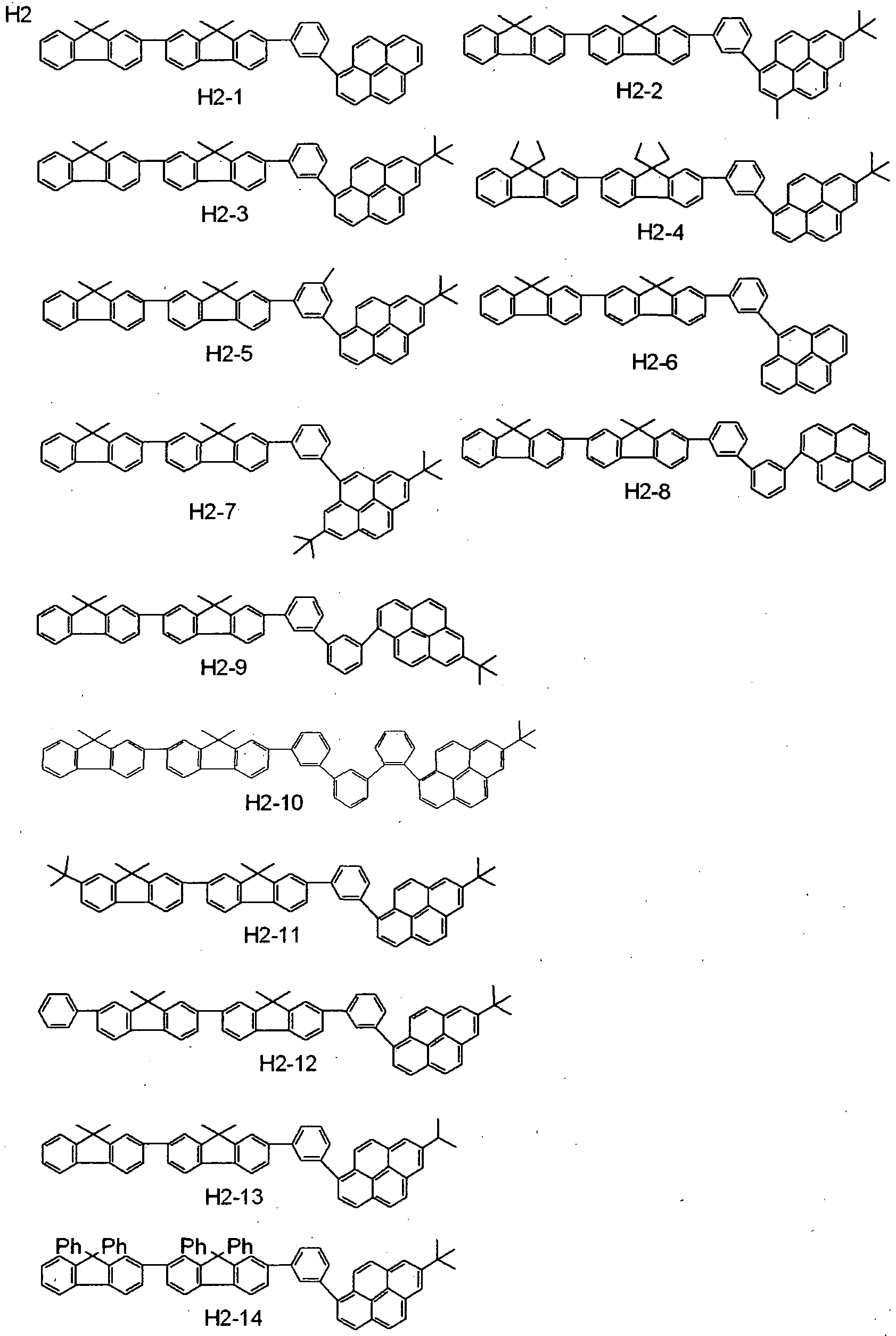
- the second compound further includes, but is not limited to, e.g., such materials having a condensed- ring aromatic group as shown below.
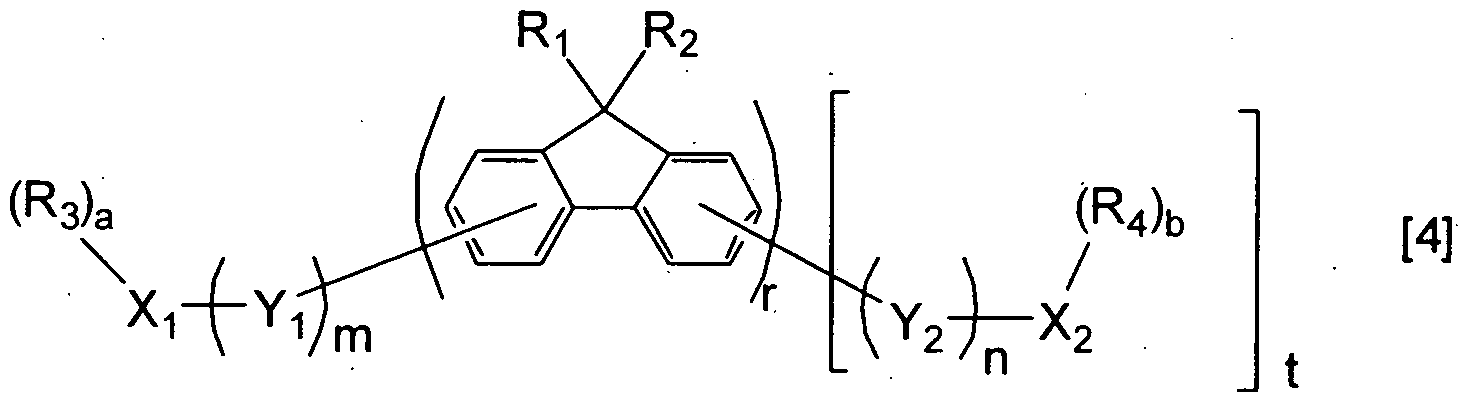
- a compound represented by the following general formula (4) preferably a compound wherein Yi and Y 2 of the general formula (4) are each independently represented by the following general formula (5), (6) or (7), and more preferably a compound represented by the following general formula (8) or further by the following general formula (9) or (10), may be used as the second compound, whereby the continuation of good light emission can be achieved with high luminous efficiency and reduced deterioration in luminance.
- R 1 and R 2 are each independently a group selected from the group consisting of a hydrogen atom, a halogen atom, a substituted or unsubstituted alkyl group, a substituted or unsubstituted aralkyl group, a substituted or unsubstituted alkoxyl group, a substituted or unsubstituted aryl group and a substituted or unsubstituted heterocyclic group, R 1 ' s may be the same or different, and R 2 ' s may be the same or different; R 3 and R 4 are each independently a group selected from the group consisting of a halogen atom, a substituted or unsubstituted alkyl group, a substituted or unsubstituted aralkyl group, a substituted or unsubstituted alkoxyl group, a substituted or unsubstituted aryl group and a substituted or unsubstituted heterocycl
- Xi and X 2 are each independently a substituted or unsubstituted aryl group or a substituted or unsubstituted heterocyclic ring group; a and b are each independently an integer of 0 to 3;
- Yi and Y 2 are each independently a substituted or unsubstituted phenylene group, Yi' s may be the same or different, and Y 2 ' s may be the same or different; m and n are each independently an integer of 1 to 3; t is 0 or 1, and, when t is 0, the terminal fluorenyl group may be substituted, at its position where it is substituted with Y 2 , with a group selected from the group consisting of a halogen atom, a substituted or unsubstituted alkyl group, a substituted or unsubstituted aralkyl group, a substituted or unsubstituted alkoxyl group and a substituted or unsubstituted phenyl group; and r is an integer of 1 to 5.
- R 5 and R 6 are each independently a group selected from the group consisting of a halogen atom, a substituted or unsubstituted alkyl group, a substituted or unsubstituted aralkyl group, a substituted or unsubstituted alkoxyl group, a substituted or unsubstituted aryl group and a substituted or unsubstituted heterocyclic group, R 5 ' s may be the same or different, and R 6 ' s may be the same or different; and c and d are each independently an integer of 0 to 4.
- R 7 is a group selected from the group consisting of a halogen atom, a substituted or unsubstituted alkyl group, a substituted or unsubstituted aralkyl group, a substituted or unsubstituted alkoxyl group, a substituted or unsubstituted aryl group and a substituted or unsubstituted heterocyclic ring group, R 7 ' s may be the same or different; and e is an integer of 0 to 4.
- the hydrogen substituents may be heavy hydrogen.
- Specific examples of the substituted or unsubstituted alkyl group, aralkyl group and heterocyclic group and the halogen atom may include those listed in regard to the above general formulas (1) to (3) and (11) to (13) .
- the substituted or unsubstituted alkoxyl group may include alkyloxyl groups having an alkyl group or an aralkyl group, aralkyloxyl groups, the substituted or unsubstituted aryl group described in regard to the above general formulas (1) to (3) and (11) to (13), and alkoxyl groups having a heterocyclic group.
- the substituted or unsubstituted alkoxyl group more specifically includes, but is not limited to, a methoxyl group, an ethoxyl group, a propoxyl group, a 2-ethyl-octyloxyl group, a phenoxyl group, a 4-tert- butylphenoxyl group, a benzyloxyl group and a thienyloxyl group.
- the substituted or unsubstituted aryl group includes the phenyl group and condensed bicyclic aromatic group described previously, and besides a substituted or unsubstituted tricyclic or higher condensed-ring aromatic group.
- the substituted or unsubstituted, condensed tri or more cyclic aromatic group includes, but is not limited to, an acenaphthylenyl group, an anthryl group, a phenanthryl group, a pyrenyl group, a fluoranthenyl group, a benzofluoranthenyl group, an acephenanthrylenyl group, an aceanthrylenyl group, a chrysenyl group, a dibenzochrysenyl group, a benzoanthryl group, a naphthacenyl group, a picenyl group, a fluorenyl group and a triphenylenyl group.
- the compound wherein Y 1 and Y 2 are each independently a group selected from the groups represented by the general formulas (5) and (6) and at least one of Xi and X 2 is a substituted or unsubstituted pyrene ring includes, but is not limited to, e.g., the following materials.
- the compound wherein Yi and Y 2 are each independently a group selected from the . groups represented by the general formulas (5) and (6) and at least one of Xi and X 2 is a group other than the pyrene ring includes, but is not limited to, e.g., the following materials.
- the compound wherein one of X x and X 2 is a substituted or unsubstituted pyrene ring and the other is a condensed-ring hydrocarbon skeleton other than the pyrene ring includes, but is not limited to, e.g., the following materials.
- the compound wherein Yi and Y 2 are each independently a group selected from the groups represented by the general formula (7) and at least one of Xi and X 2 is a substituted or unsubstituted pyrene ring includes, but is not limited to, e.g., the following materials.
- the compound wherein Yi and Y 2 are each independently a group selected from the groups represented by the general formulas (5) to (7) and at least one of Xi and X 2 is a pyrene ring includes, but is not limited to, e.g., the following materials.
- the compound wherein Yi and Y 2 are each independently a group selected from the groups represented by the general formula (7) and at least one of Xi and X 2 is a group other than a pyrene ring includes, but is not limited to, e.g., the following materials .
- the compound wherein Yi and Y 2 are each independently a group selected from the groups represented by the general formula (7) and one of X 1 and X 2 is a substituted or unsubstituted pyrene ring and the other is a condensed-ring hydrocarbon skeleton other than a pyrene ring may include, but is not limited to, e.g., the following materials.
- the dopant concentration is 0.01% by mass or more and 80% by mass or less, and preferably 1% by mass or more and 40% by mass or less, based on the mass of the host material.
- the dopant material may be contained uniformly, or with a concentration gradient, in the whole layer formed of the host material, or may partially be contained in some regions to form regions of a host material layer containing no dopant material.
- FIG. 1 illustrates a preferred example of the organic luminescent device of the present invention.
- FIG. 1 is a sectional view showing an example of the organic luminescent device of the present invention, which is constituted of a substrate 1, and an anode 2, a hole transport layer 5, a luminescent layer 3, an electron transport layer 6 and a cathode 4 in this order provided on the substrate.
- This device is one in which the functions of carrier transport and luminescence are separated, and is used in appropriate combination with a compound having each of a hole transport property, an electron transport property and a luminescence property.
- a device which is constituted of a substrate 1, and an anode 2, a luminescent layer 3 and a cathode 4 in this order provided on the substrate.
- the luminescent device used in this case is useful where the device itself has hole transportability, electron transportability and luminescence performance, or where compounds having the respective properties are used in the form of a mixture.
- a material having either of hole transport function or electron transport function or both of them is used as a luminescent material in any one of the layers.
- This device is useful where such a material is used in combination with a mere hole transporting material or electron transporting material having no luminescent property.
- the luminescent layer consists of either of the hole transport layer 5 or the electron transport layer 6.
- FIG. 1 may be so constituted as to be provided with a hole injection layer inserted on the anode 2 side. This is effective in improving adherence between the anode 2 and the hole transport layer 5 and improving hole injection, and is effective in low-voltage luminescence.
- FIG. 1 may be so constituted as to be provided with a layer (hole/exciton blocking layer) which is inserted between the luminescent layer 3 and the electron transport layer 6 to prevent holes or excitons from passing through toward the cathode 4 side, .
- a compound having a very high ionization potential may be used in the hole/exciton blocking layer to provide a constitution very effective in improving luminous efficiency.
- the device constitution given in FIG. 1 and the foregoing show very basic constitutions, and the constitution of the organic luminescent device using the compound of the present invention is by no means limited thereto.
- the device may have various constitutions such that an insulating layer is provided at the interface between an electrode and an organic layer, an adhesive layer or an interference layer is provided, and the hole transport layer is constituted of two layers different in ionization potential .
- the organic luminescent device of the present invention may be used in any forms of the device constitution given in FIG. 1 and the, foregoing .
- an organic layer using the benzo [ k] fluoranthene compound of the present invention is useful as the luminescent layer, the electron transport layer or the hole transport layer.
- a layer formed by vacuum deposition (vacuum evaporation) or solution coating can not easily be crystallized and has a superior stability over time.
- the above benzo [k] fluoranthene compound is used as a constituent of, in particular, the luminescent layer, and may optionally be used together with a hole transporting compound, a luminescent compound and/or an electron transporting compound which are/is of a low molecular type or polymer type hitherto known in the art.
- a hole injecting and transporting material facilitates the injection of holes from the anode and has mobility good enough to transport the injected holes to the luminescent layer.
- Low-molecular and high-molecular materials having hole injection and transport functions include, but is not limited to, the following:
- Triarylamine derivatives Triarylamine derivatives, phenylenediamine derivatives, triazole derivatives, oxadiazole derivatives, imidazole derivatives, pyrazoline derivatives, pyrazolone derivatives, oxazole derivatives, fluorenone derivatives, hydrazone derivatives, stilbene derivatives, phthalocyanine derivatives, porphyrin derivatives, poly (vinylcarbazole) , poly (silylene) , poly (thiophene) , and other conductive high-molecular materials.
- Materials concerned chiefly in a luminescence function which are usable besides the compounds used in the organic luminescent device of the present invention includes, but are not limited to, the following: High-molecular derivatives such as polycyclic condensation aromatic compounds (e.g., naphthalene derivatives, phenanthrene derivatives, fluorene derivatives, pyrene derivatives, tetracene derivatives, coronene derivatives, chrysene derivatives, perylene derivatives, 9, 10-diphenylanthracene derivatives, and rubrene) , quinacridone derivatives, acridone derivatives, coumarine derivatives, pyrane derivatives, Nile Red, pyrazine derivatives, benzoimidazole derivatives, benzothiazole derivatives, benzoxazole derivatives, stilbene derivatives, organometallic complexes (e.g., organoaluminum complexes such as tris ( 8-quinolinolato) aluminum,
- An electron injecting and transporting material may be selected from materials which facilitate the injection of electrons from the cathode and have a function to transport injected electrons to the luminescent layer, taking into account, e.g., a balance with the carrier mobility of the hole transporting material.
- Materials having electron injection and transport functions include, but is not limited to, the following:
- the layer containing the benzo [k] fluoranthene compound of the present invention and the layers formed of other organic compounds are formed by any of methods shown below.
- the layers are commonly formed by forming thin films by means of vacuum deposition, ionization deposition, sputtering, plasma deposition or known application (e.g., spin coating, dipping, casting, the LB (Langmuir Blodgett) process, or ink-jet printing) of a coating material prepared by dissolving a material in a suitable solvent.
- the films may be formed using materials in combination with a suitable binder resin.
- the binder resin may be selected from extensive binding resins, and includes, but is not limited to, the following:
- Polyvinyl carbazole resins polycarbonate resins, polyester resins, polyarylate resins, polystyrene resins, ABS resins, polybutadiene resins, polyurethane resins, acrylic resins, methacrylic resins, butyral resins, polyvinyl acetal resins, polyamide resins, polyimide resins, polyethylene resins, polyether sulfone resins, diallyl phthalate resins, phenol resins, epoxy resins, silicone resins, polysulfone resins, and urea resins. These may be used alone or in the form of a mixture of two or more types as copolymers . Additives such as known plasticizers, antioxidants and ultraviolet absorbers may optionally be used in combination .
- Materials for the anode having a work function as large as possible are favorable.
- the following is usable metal simple substances such as gold, platinum, silver, copper, nickel, palladium, cobalt, selenium, vanadium and tungsten, or alloys of any of these; and metal oxides such as tin oxide, zinc oxide, indium oxide, indium-tin oxide (ITO), and indium-zinc oxide.
- metal simple substances such as gold, platinum, silver, copper, nickel, palladium, cobalt, selenium, vanadium and tungsten, or alloys of any of these
- metal oxides such as tin oxide, zinc oxide, indium oxide, indium-tin oxide (ITO), and indium-zinc oxide.
- conductive polymers such as polyaniline, polypyrrole, polythiophene, and polyphenylene sulfide. Any of these electrode materials may be used alone or in a combination of two or more types.
- the anode may be constituted of a single layer
- Materials for the cathode having a small work function are favorable.
- the following are usable: metal simple substances such as lithium, sodium, potassium, calcium, magnesium, aluminum, indium, ruthenium, titanium, manganese, yttrium, silver, lead, tin, and chromium; or alloys of two or more of these, such as lithium-indium, sodium-potassium, magnesium-silver, aluminum-lithium, aluminum-magnesium, and magnesium-indium.
- Metal oxides such as indium-tin oxide (ITO) may also be used. Any of these electrode materials may be used alone or in a combination of two or more types.
- the cathode may be constituted of a single layer or a multiple layer.
- the substrate used in the present invention there are no particular limitations thereon. It is possible to use opaque substrates made of, for example, metal or ceramics, and transparent substrates made of, for example, glass, quartz and plastic sheets. A color filter film, a fluorescent color conversion filter film, or a dielectric reflecting film may also be used as the substrate to control luminescence light.
- a protective layer or a sealing layer may be provided in order to prevent contact with oxygen or moisture.
- the protective layer may include diamond thin films; films of inorganic materials such as metal oxides and metal nitrides; films of high-molecular materials such as fluorine resins, polyparaxylene, polyethylene, silicone resins and polystyrene resins; and photocurable resins.
- the device may be covered with glass, a gas-impermeable film or metal, and packaged with a suitable sealing resin.
- the organic luminescent device of the present invention may be so produced as to be connected with thin-film transistors (TFTs) fabricated on the substrate .
- TFTs thin-film transistors
- the device may be of either bottom emission (a constitution in which light is taken out from the substrate side) or top emission (a constitution in which light is taken out from the opposite side of the substrate. )
- Exemplary Compound No. 101 of the present invention can be produced by, e.g., a process described below.
- Exemplary Compound No. 102 4-bromo-7, 12-di (4-tert-butylphenyl) benzo [k] fluo- ranthene .
- Exemplary Compound No. 106-1 4-bromo-7, 12-di (2-methylphenyl) benzo [k] fluo- ranthene .
- Exemplary Compound No. 110 4-bromo-7, 12-di (4-fluorophenyl) benzo [k] fluo- ranthene .
- Exemplary Compound No. 118 can be synthesized in the same manner as in Production Example 1 except that the following compound is used in place of the 2- (fluoranthen-3-yl) -A 1 4, 5, 5-tetramethyl- [1, 3, 2 ] dioxabororane used in Production Example 1.
- 2- (5, 8-di-tertbutylfluoranthen-3-yl) -4,4,5,5- tetramethyl- [1,3,2] dioxabororane Production Examples 9 to 12
- Exemplary Compound No. 201 2-(fluoranthen-8-yl) -4, 4, 5, 5-tetramethyl- [1, 3, 2 ] dioxabororane .
- Exemplary Compound No. 223-2 2- ( fluoranthen-1-yl) -4, 4 , 5, 5-tetramethyl- [1,3,2] dioxabororane .
- Exemplary Compound No. H2-3 of the present invention can be produced by, e.g., a process described below.
- the mixture obtained was cooled to room temperature, followed by adding water, toluene and ethyl acetate to separate an organic layer.
- the aqueous layer was further extracted (twice) with the mixed solvent of toluene and ethyl acetate, and the extract obtained was added to the organic layer solution having been separated first.
- This organic layer was washed with saturated brine, followed by drying with sodium sulfate.
- IXIALDI-TOF MS matrix-assisted laser desorption/ ionization time-of-flight mass spectrometry ascertained 718.4 which was M + of this compound. Further, 1 H-NMR measurement ascertained the structure of this compound (see FIG. 4) .
- Exemplary Compound No. H2-13 2- (7-iso-propylpyren-l-yl) -4, 4, 5, 5-tetramethyl- [1,3,2] dioxabororane .
- Exemplary Compound No. H10-2 2- (lO-phenylanthracen-9-yl) -4,4,5, 5-tetramethyl- [1,3,2] dioxabororane .
- Exemplary Compound No. Hll-2 2- (phenanthren-9-yl) -4, 4, 5, 5-tetramethyl- [1,3,2] dioxabororane.
- Exemplary Compound No. H2-4 can be synthesized in the same manner as in Production Example 13 except that 2- [2- (9' , 9' -diethylfluoren-2' -yl) -9, 9-diethylfluoren-7- yl-4 , 4 , 5, 5-tetramethyl- [ 1, 3, 2] dioxabororane is used in place of the intermediate (III) used in Production Example 13.
- Production Example 22 2- [2- (9' , 9' -diethylfluoren-2' -yl) -9, 9-diethylfluoren-7- yl-4 , 4 , 5, 5-tetramethyl- [ 1, 3, 2] dioxabororane is used in place of the intermediate (III) used in Production Example 13.
- Exemplary Compound No. H6-2 can be synthesized in the same manner as in Production Example 13 except that l-bromo-2-iodobenzene is used in place of the 1-bromo- 3-iodobenzene) used in Production Example 13.
- Production Example 23 (Production of Exemplary Compound No. H4-2)
- Exemplary Compound No. H4-2 of the present invention can be produced by, e.g., a process described below.
- Exemplary Compound No. H4-4 2- (7-tert-butyl-3-methylpyren-l-yl) -4, 4,5,5- tetramethyl- [1,3,2] dioxabororane .
- Exemplary Compound No. H4-5 2- (pyren-4-yl) -4,4,5, 5-tetramethyl- [1,3,2] dioxabororane .
- Exemplary Compound No. H4-8 2- (7-iso-propylpyren-l-yl) -4,4,5, 5-tetramethyl- [1,3,2] dioxabororane .
- Exemplary Compound No. H9-1 of the present invention can be produced by, e.g., a process described below.
- Exemplary Compound No. H9-6 can be synthesized in the same manner as in Production Example 31 except that l-bromo-2-iodobenzene is used in place of l-bromo-3- iodobenzene used in Production Example 31.
- Exemplary Compound No. H14-3 used in the present invention can be produced by, e.g., a process described below.
- An intermediate (X) was produced in the same manner as in the intermediate (VII) except that 1- bromo-4-iodobenzene was used in place of the l-bromo-3- iodobenzene) .
- the mixture obtained was cooled to room temperature, followed by adding water, toluene and ethyl acetate to separate an organic layer.
- the aqueous layer was further extracted (twice) with the mixed solvent of toluene and ethyl acetate, and the extract obtained was added to the organic layer solution having been separated first.
- This organic layer was washed with saturated brine, followed by drying with sodium sulfate.
- MALDI-TOF MS matrix-assisted laser desorption/ ionization time-of-flight mass spectrometry
- An organic luminescent device having the structure shown in FIG. 1 was fabricated in the following way.
- an indium-tin oxide (ITO) film as the anode 2 was formed by sputtering in a layer thickness of 120 run, and was used as a transparent conductive support substrate.
- ITO indium-tin oxide
- This substrate was subjected to ultrasonic cleaning successively with acetone and isopropyl alcohol (IPA), and subsequently to cleaning with pure water, followed by drying. It was further subjected to UV/ozone cleaning, and was used as the transparent conductive support substrate.
- IPA acetone and isopropyl alcohol
- This solution was dropped on the above ITO electrode, and spin-coating was performed first at the number of revolutions of 500 rpm for 10 seconds and then at the number of revolutions of 1,000 rpm for 40 seconds to form a thin film. Thereafter, the film was dried for 10 minutes in a 80 0 C vacuum oven to completely remove the solvent remaining in the thin film to form the hole transport layer 5.
- Exemplary Compound No. 101 and Compound 2 as a second compound, represented by the following structural formula were vacuum-deposited together (mass ratio: 5:9) to form the luminescent layer 3 of 30 nm in thickness.
- the film formation by vacuum deposition was performed under conditions of a degree of vacuum of 1.0 * 10 "4 Pa and a rate of film formation of 0.1 nm/sec or more and 0.2 nm/sec or less.
- a 2, 9-bis [2- (9, 9' -dimethylfluorenyl) -1, 10-phenanthroline film was formed by vacuum deposition in a layer thickness of 30 nm.
- the film formation by vacuum deposition was performed under conditions of a degree of vacuum of 1.0 * 10 ⁇ 4 Pa and a rate of film formation of 0.1 nm/sec or more and 0.2 nm/sec or less.
- a lithium fluoride (LiF) film was formed by vacuum deposition in a layer thickness of 0.5 nm, and an aluminum film of 100 nm in thickness was further formed by vacuum deposition.
- An organic luminescent device (organic EL device) was fabricated having the aluminum-lithium film as an electron injection electrode (cathode 4) .
- the film formation by vacuum deposition was performed under conditions of a degree of vacuum of 1.0 * 10 "4 Pa and a rate of film formation of 0.1 nm/sec for the lithium fluoride film and 0.5 nm/sec or more and 1.0 nm/sec or less for the aluminum film.
- the organic EL device thus obtained was covered with a protecting glass plate in an atmosphere of dry air and sealed with an acrylic-resin adhesive so as not to cause device deterioration due to adsorption of moisture .
- Exemplary Compound No. 101 had an energy gap of 2.81 eV.
- a method was used in which absorption spectra were measured with an ultraviolet- visible spectrophotometer Hitachi U-3010 and the values were calculated from absorption ends of the spectra.
- An organic luminescent device was fabricated in the same manner as in Example 1 except that Compound 4 represented by the following structural formula was used in place of Compound 1, a film of the above Compound 3 was formed by vacuum deposition in a layer thickness of 15 nm to form the hole transport layer 5 and Exemplary Compound No. H2-3 was used as the second compound in place of the above Compound 2.
- Exemplary Compound No. H2-3 had an energy gap of 3.18 eV and Exemplary Compound No. 101 had an energy gap of 2.81 eV.
- a method was used in which absorption spectra were measured with an ultraviolet-visible spectrophotometer Hitachi U-3010 and the values were calculated from absorption ends of the spectra.
- An organic luminescent device was fabricated in the same manner as in Example 3 except that Exemplary Compound No. H14-3 was used as the second compound in place of the above Compound 2.
- Exemplary Compound No. H14-3 had an energy gap of 3.03 eV. To examine the energy gap, a method was used in which an absorption spectrum was measured with an ultraviolet-visible spectrophotometer Hitachi U-3010 and the value was calculated from an absorption end of the spectrum.
- Example 5 In the device 'of this Example, light emission of 8.3 cd/A in current efficiency was observed at an applied voltage of 4 V. The voltage was further applied to this device for 100 hours in an atmosphere of nitrogen to ascertain that good light emission continued.
- Example 5
- Compound 5 had an energy gap of 3.06 eV. To examine the energy gap, a method was used in which an absorption spectrum was measured with an ultraviolet- visible spectrophotometer Hitachi U-3010 and the value was calculated from an absorption end of the spectrum. Compound 5
- Example 6 An organic luminescent device was fabricated in the same manner as in Example 3 except that Compound 6 represented by the following structural formula was used as the second compound in place of the above Compound 2.
- Compound 6 had an energy gap of 2.99 eV. To examine the energy gap, a method was used in which an absorption spectrum was measured with an ultraviolet- visible spectrophotometer Hitachi U-3010 and the value was calculated from an absorption end of the spectrum.
- Compound 7 had an energy gap of 2.99 eV. To examine the energy gap, a method was used in which an absorption spectrum was measured with an ultraviolet- visible spectrophotometer Hitachi U-3010 and the value was calculated from an absorption end of the spectrum. Compound 7
- Compound 8 had an energy gap of 3.12 eV. To examine the energy gap, a method was used in which an absorption spectrum was measured with an ultraviolet- visible spectrophotometer Hitachi U-3010 and the value was calculated from an absorption end of the spectrum. Compound 8
- An organic luminescent device was fabricated in the same manner as in Example 3 except that Compound 9 represented by the following structural formula was used as the second compound in place of the above Compound 2.
- Compound 9 had an energy gap of 2.88 eV. To examine the energy gap, a method was used in which an absorption spectrum was measured with an ultraviolet- visible spectrophotometer Hitachi U-3010 and the value was calculated from an absorption end of the spectrum.
- Example 10 In the device of this Example, light emission of 6.4 cd/A in current efficiency was observed at an applied voltage of 4 V. The voltage was further applied to this device for 100 hours in an atmosphere of nitrogen to ascertain that good light emission continued.
- Example 10
- Compound 10 had an energy gap of 2.99 eV. To examine the energy gap, a method was used in which an absorption spectrum was measured with an ultraviolet- visible spectrophotometer Hitachi U-3010 and the value was calculated from an absorption end of the spectrum. Compound 10
Landscapes
- Chemical & Material Sciences (AREA)
- Organic Chemistry (AREA)
- Engineering & Computer Science (AREA)
- Materials Engineering (AREA)
- Physics & Mathematics (AREA)
- Spectroscopy & Molecular Physics (AREA)
- Optics & Photonics (AREA)
- Electroluminescent Light Sources (AREA)
- Organic Low-Molecular-Weight Compounds And Preparation Thereof (AREA)
Abstract
Description
Claims
Priority Applications (3)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| EP07768469A EP2049616A4 (en) | 2006-08-04 | 2007-07-18 | Organic luminescent device and benzo[k]fluoranthene compound |
| US12/296,574 US7977869B2 (en) | 2006-08-04 | 2007-07-18 | Organic luminescent device and benzo[k]fluoranthene compound |
| CN2007800137888A CN101426882B (en) | 2006-08-04 | 2007-07-18 | Organic luminescent device and benzo[k]fluoranthene compound |
Applications Claiming Priority (4)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| JP2006213606 | 2006-08-04 | ||
| JP2006-213606 | 2006-08-04 | ||
| JP2007120565 | 2007-05-01 | ||
| JP2007-120565 | 2007-05-01 |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| WO2008015945A1 true WO2008015945A1 (en) | 2008-02-07 |
Family
ID=38997124
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/JP2007/064615 WO2008015945A1 (en) | 2006-08-04 | 2007-07-18 | ORGANIC LUMINESCENT DEVICE AND BENZO[k]FLUORANTHENE COMPOUND |
Country Status (6)
| Country | Link |
|---|---|
| US (1) | US7977869B2 (en) |
| EP (1) | EP2049616A4 (en) |
| JP (1) | JP4455625B2 (en) |
| KR (1) | KR101019876B1 (en) |
| CN (1) | CN101426882B (en) |
| WO (1) | WO2008015945A1 (en) |
Cited By (7)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2009123344A1 (en) * | 2008-04-01 | 2009-10-08 | Canon Kabushiki Kaisha | Organic light-emitting device |
| GB2463040A (en) * | 2008-08-28 | 2010-03-03 | Cambridge Display Tech Ltd | Light emitting polymer |
| WO2010047403A1 (en) * | 2008-10-22 | 2010-04-29 | Canon Kabushiki Kaisha | Organic light-emitting device |
| EP2190044A2 (en) | 2008-11-19 | 2010-05-26 | Canon Kabushiki Kaisha | Light-emitting device, image display apparatus, and novel organic compound |
| WO2011018886A1 (en) | 2009-08-10 | 2011-02-17 | Canon Kabushiki Kaisha | Organic compound and organic light-emitting device using the same |
| EP2197060A3 (en) * | 2008-12-10 | 2012-07-18 | Canon Kabushiki Kaisha | Organic light-emitting device |
| US8569751B2 (en) | 2009-08-20 | 2013-10-29 | Canon Kabushiki Kaisha | Heterocyclic compound and organic light-emitting device using the same |
Families Citing this family (18)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JP5074754B2 (en) | 2006-03-31 | 2012-11-14 | キヤノン株式会社 | Novel compound and organic light emitting device using the same |
| EP2085371B1 (en) * | 2006-11-15 | 2015-10-07 | Idemitsu Kosan Co., Ltd. | Fluoranthene compound, organic electroluminescent device using the fluoranthene compound, and organic electroluminescent material-containing solution |
| JP5142589B2 (en) * | 2007-05-28 | 2013-02-13 | キヤノン株式会社 | Indenochrysene derivative and organic light emitting device using the same |
| WO2009066808A1 (en) * | 2007-11-22 | 2009-05-28 | Gracel Display Inc. | Aromatic electroluminescent compounds with high efficiency and electroluminescent device using the same |
| WO2009066815A1 (en) * | 2007-11-23 | 2009-05-28 | Gracel Display Inc. | Luminescent compounds and electroluminescent device using the same |
| JP5335284B2 (en) * | 2008-05-22 | 2013-11-06 | キヤノン株式会社 | Fused polycyclic compound and organic light emitting device having the same |
| JP5376857B2 (en) * | 2008-08-04 | 2013-12-25 | キヤノン株式会社 | Fused polycyclic compound and organic light emitting device using the same |
| JP5627209B2 (en) * | 2009-09-14 | 2014-11-19 | キヤノン株式会社 | Novel pyrene compound and organic EL device having the same |
| JP5578832B2 (en) * | 2009-11-05 | 2014-08-27 | キヤノン株式会社 | Novel fluorenylamine compound and organic light emitting device having the same |
| US8929054B2 (en) * | 2010-07-21 | 2015-01-06 | Cleanvolt Energy, Inc. | Use of organic and organometallic high dielectric constant material for improved energy storage devices and associated methods |
| TW201215659A (en) * | 2010-10-08 | 2012-04-16 | Idemitsu Kosan Co | Benzo[k]fluoranthene derivative and organic electroluminescence element containing same |
| JP5777408B2 (en) | 2011-05-30 | 2015-09-09 | キヤノン株式会社 | Fused polycyclic compound and organic light emitting device using the same |
| EP2745093B1 (en) * | 2011-08-16 | 2021-12-15 | Waters Technologies Corporation | Flow cells for differential refractive index detection |
| JP2015233023A (en) * | 2012-08-31 | 2015-12-24 | 出光興産株式会社 | Organic electroluminescent device |
| JP6187080B2 (en) * | 2012-10-11 | 2017-08-30 | Tdk株式会社 | Electroluminescent device |
| WO2014145559A2 (en) | 2013-03-15 | 2014-09-18 | Cleanvolt Energy, Inc. | Improved electrodes and currents through the use of organic and organometallic high dielectric constant materials in energy storage devices and associated methods |
| JP7033397B2 (en) * | 2016-03-29 | 2022-03-10 | 住友化学株式会社 | Light emitting element |
| KR102383315B1 (en) | 2018-03-30 | 2022-04-06 | 캐논 가부시끼가이샤 | Organic light emitting element, display apparatus, image pickup apparatus, and illumination apparatus |
Citations (10)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JPH1112205A (en) * | 1997-04-18 | 1999-01-19 | Mitsui Chem Inc | Fluoranthene derivative and organic electroluminescent element |
| JP2000178212A (en) | 1998-12-17 | 2000-06-27 | Mitsui Chemicals Inc | Hydrocarbon compound and organic electroluminescent element |
| JP2002025776A (en) | 2000-07-11 | 2002-01-25 | Mitsui Chemicals Inc | Hydrocarbon compound and organic electroluminescent element |
| JP2002313575A (en) | 2001-04-09 | 2002-10-25 | Mitsui Chemicals Inc | Hydrocarbon compound and organic electroluminescent element |
| JP2004002351A (en) * | 2002-03-27 | 2004-01-08 | Tdk Corp | Organic el device |
| JP2005068087A (en) * | 2003-08-26 | 2005-03-17 | Mitsui Chemicals Inc | Benzofluoranthene compound and organic electroluminescent element containing benzofluoranthene compound |
| WO2005090365A1 (en) * | 2004-03-18 | 2005-09-29 | Sharp Kabushiki Kaisha | Organosilanes, process for production of the same, and use thereof |
| US20060158102A1 (en) * | 2004-12-01 | 2006-07-20 | Idemitsu Kosan Co., Ltd. | Organic electroluminescent device |
| JP2007191061A (en) * | 2006-01-19 | 2007-08-02 | Nissan Motor Co Ltd | Vehicle body front part structure |
| WO2007100010A1 (en) | 2006-02-28 | 2007-09-07 | Idemitsu Kosan Co., Ltd. | Organic electroluminescent device |
Family Cites Families (8)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JP4473414B2 (en) * | 2000-06-06 | 2010-06-02 | 三井化学株式会社 | Hydrocarbon compounds and organic electroluminescent devices |
| JP4240841B2 (en) * | 2001-04-27 | 2009-03-18 | キヤノン株式会社 | Organic light emitting device |
| US7049011B2 (en) * | 2002-03-14 | 2006-05-23 | Tdk Corporation | Organic electroluminescent device |
| JP3840235B2 (en) * | 2003-06-27 | 2006-11-01 | キヤノン株式会社 | Organic light emitting device |
| JP4065547B2 (en) * | 2004-04-12 | 2008-03-26 | キヤノン株式会社 | Fluorene compound and organic light emitting device using the same |
| JP4429149B2 (en) * | 2004-11-26 | 2010-03-10 | キヤノン株式会社 | Fluorene compound and organic light emitting device |
| US7411799B2 (en) * | 2005-07-27 | 2008-08-12 | Texas Instruments Incorporated | Apparatus and method for regulating a switching device |
| JP5074754B2 (en) | 2006-03-31 | 2012-11-14 | キヤノン株式会社 | Novel compound and organic light emitting device using the same |
-
2007
- 2007-07-18 CN CN2007800137888A patent/CN101426882B/en not_active Expired - Fee Related
- 2007-07-18 US US12/296,574 patent/US7977869B2/en active Active
- 2007-07-18 KR KR1020097004453A patent/KR101019876B1/en not_active IP Right Cessation
- 2007-07-18 EP EP07768469A patent/EP2049616A4/en not_active Withdrawn
- 2007-07-18 WO PCT/JP2007/064615 patent/WO2008015945A1/en active Application Filing
- 2007-08-03 JP JP2007202583A patent/JP4455625B2/en not_active Expired - Fee Related
Patent Citations (10)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JPH1112205A (en) * | 1997-04-18 | 1999-01-19 | Mitsui Chem Inc | Fluoranthene derivative and organic electroluminescent element |
| JP2000178212A (en) | 1998-12-17 | 2000-06-27 | Mitsui Chemicals Inc | Hydrocarbon compound and organic electroluminescent element |
| JP2002025776A (en) | 2000-07-11 | 2002-01-25 | Mitsui Chemicals Inc | Hydrocarbon compound and organic electroluminescent element |
| JP2002313575A (en) | 2001-04-09 | 2002-10-25 | Mitsui Chemicals Inc | Hydrocarbon compound and organic electroluminescent element |
| JP2004002351A (en) * | 2002-03-27 | 2004-01-08 | Tdk Corp | Organic el device |
| JP2005068087A (en) * | 2003-08-26 | 2005-03-17 | Mitsui Chemicals Inc | Benzofluoranthene compound and organic electroluminescent element containing benzofluoranthene compound |
| WO2005090365A1 (en) * | 2004-03-18 | 2005-09-29 | Sharp Kabushiki Kaisha | Organosilanes, process for production of the same, and use thereof |
| US20060158102A1 (en) * | 2004-12-01 | 2006-07-20 | Idemitsu Kosan Co., Ltd. | Organic electroluminescent device |
| JP2007191061A (en) * | 2006-01-19 | 2007-08-02 | Nissan Motor Co Ltd | Vehicle body front part structure |
| WO2007100010A1 (en) | 2006-02-28 | 2007-09-07 | Idemitsu Kosan Co., Ltd. | Organic electroluminescent device |
Non-Patent Citations (3)
| Title |
|---|
| CAN. J. CHEM., vol. 70, 1992, pages 1015 |
| J. AM. CHEM. SOC., vol. 74, 1952, pages 1075 |
| See also references of EP2049616A4 |
Cited By (17)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US20100327274A1 (en) * | 2008-04-01 | 2010-12-30 | Canon Kabushiki Kaisha | Organic light-emitting device |
| JP2009267378A (en) * | 2008-04-01 | 2009-11-12 | Canon Inc | Organic light-emitting element |
| WO2009123344A1 (en) * | 2008-04-01 | 2009-10-08 | Canon Kabushiki Kaisha | Organic light-emitting device |
| GB2463040A (en) * | 2008-08-28 | 2010-03-03 | Cambridge Display Tech Ltd | Light emitting polymer |
| GB2463040B (en) * | 2008-08-28 | 2012-10-31 | Cambridge Display Tech Ltd | Light-emitting material |
| DE112009002093T5 (en) | 2008-08-28 | 2011-07-07 | Cambridge Display Technology Ltd. | Light-emitting material and device |
| EP2338185A4 (en) * | 2008-10-22 | 2012-04-04 | Canon Kk | Organic light-emitting device |
| EP2338185A1 (en) * | 2008-10-22 | 2011-06-29 | Canon Kabushiki Kaisha | Organic light-emitting device |
| CN102197506A (en) * | 2008-10-22 | 2011-09-21 | 佳能株式会社 | Organic light-emitting device |
| WO2010047403A1 (en) * | 2008-10-22 | 2010-04-29 | Canon Kabushiki Kaisha | Organic light-emitting device |
| CN101740725A (en) * | 2008-11-19 | 2010-06-16 | 佳能株式会社 | Light-emitting device, image display apparatus, and novel organic compound |
| EP2190044A3 (en) * | 2008-11-19 | 2012-02-29 | Canon Kabushiki Kaisha | Light-emitting device, image display apparatus, and novel organic compound |
| EP2190044A2 (en) | 2008-11-19 | 2010-05-26 | Canon Kabushiki Kaisha | Light-emitting device, image display apparatus, and novel organic compound |
| EP2197060A3 (en) * | 2008-12-10 | 2012-07-18 | Canon Kabushiki Kaisha | Organic light-emitting device |
| US8455113B2 (en) | 2008-12-10 | 2013-06-04 | Canon Kabushiki Kaisha | Organic light-emitting device |
| WO2011018886A1 (en) | 2009-08-10 | 2011-02-17 | Canon Kabushiki Kaisha | Organic compound and organic light-emitting device using the same |
| US8569751B2 (en) | 2009-08-20 | 2013-10-29 | Canon Kabushiki Kaisha | Heterocyclic compound and organic light-emitting device using the same |
Also Published As
| Publication number | Publication date |
|---|---|
| US20090278447A1 (en) | 2009-11-12 |
| KR101019876B1 (en) | 2011-03-04 |
| EP2049616A1 (en) | 2009-04-22 |
| EP2049616A4 (en) | 2012-10-17 |
| CN101426882B (en) | 2012-07-04 |
| JP2008297535A (en) | 2008-12-11 |
| US7977869B2 (en) | 2011-07-12 |
| CN101426882A (en) | 2009-05-06 |
| KR20090035638A (en) | 2009-04-09 |
| JP4455625B2 (en) | 2010-04-21 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| US7977869B2 (en) | Organic luminescent device and benzo[k]fluoranthene compound | |
| US7309533B2 (en) | Substituted anthryl derivative and electroluminescence device using the same | |
| US8026664B2 (en) | Compound and organic light emitting device | |
| JP4677221B2 (en) | Organic light emitting device | |
| JP5089235B2 (en) | Fused heterocyclic compounds and organic light emitting devices | |
| KR101124749B1 (en) | Indenochrysene derivative and organic light-emitting device using same | |
| JP4429149B2 (en) | Fluorene compound and organic light emitting device | |
| JP4035482B2 (en) | Substituted anthryl derivative and organic light emitting device using the same | |
| JP5366505B2 (en) | Indenopyrene compound and organic light-emitting device using the same | |
| WO2005000787A1 (en) | Aminoanthryl derivative substitution compound and organic electroluminescence device using the same | |
| JP2007186449A (en) | Compound substituted with aminobisanthryl-derived group and organic light-emitting element by using the same | |
| US20080286611A1 (en) | Benzo(ghi)fluoranthene derivative and organic light emitting device using the same | |
| EP1487779A1 (en) | Oligofluorenylene compounds | |
| JP2006219392A (en) | Bisanthryl derived group-substituted compound and organic light emitting device | |
| WO2008111540A1 (en) | Fused ring aromatic compound and organic light-emitting device using same | |
| JP2007169182A (en) | Fluorenylene compound and organic light-emitting element using it | |
| JP5031294B2 (en) | Amine compounds and organic light emitting devices | |
| JP2010111635A (en) | New azaindenochrysene derivative and organic light-emitting element | |
| US8318326B2 (en) | Fused polycyclic compounds and organic light-emitting device using the same |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application |
Ref document number: 07768469 Country of ref document: EP Kind code of ref document: A1 |
|
| REEP | Request for entry into the european phase |
Ref document number: 2007768469 Country of ref document: EP |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 2007768469 Country of ref document: EP |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 12296574 Country of ref document: US |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 200780013788.8 Country of ref document: CN |
|
| NENP | Non-entry into the national phase |
Ref country code: DE |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 1020097004453 Country of ref document: KR |
|
| NENP | Non-entry into the national phase |
Ref country code: RU |