WO2004003187A2 - Mpg added to fermentation - Google Patents
Mpg added to fermentation Download PDFInfo
- Publication number
- WO2004003187A2 WO2004003187A2 PCT/DK2003/000455 DK0300455W WO2004003187A2 WO 2004003187 A2 WO2004003187 A2 WO 2004003187A2 DK 0300455 W DK0300455 W DK 0300455W WO 2004003187 A2 WO2004003187 A2 WO 2004003187A2
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- fermentation
- polypeptide
- interest
- added
- mpg
- Prior art date
Links
Classifications
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12N—MICROORGANISMS OR ENZYMES; COMPOSITIONS THEREOF; PROPAGATING, PRESERVING, OR MAINTAINING MICROORGANISMS; MUTATION OR GENETIC ENGINEERING; CULTURE MEDIA
- C12N9/00—Enzymes; Proenzymes; Compositions thereof; Processes for preparing, activating, inhibiting, separating or purifying enzymes
- C12N9/14—Hydrolases (3)
- C12N9/24—Hydrolases (3) acting on glycosyl compounds (3.2)
- C12N9/2402—Hydrolases (3) acting on glycosyl compounds (3.2) hydrolysing O- and S- glycosyl compounds (3.2.1)
- C12N9/2405—Glucanases
- C12N9/2408—Glucanases acting on alpha -1,4-glucosidic bonds
- C12N9/2411—Amylases
- C12N9/2414—Alpha-amylase (3.2.1.1.)
- C12N9/2417—Alpha-amylase (3.2.1.1.) from microbiological source
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12N—MICROORGANISMS OR ENZYMES; COMPOSITIONS THEREOF; PROPAGATING, PRESERVING, OR MAINTAINING MICROORGANISMS; MUTATION OR GENETIC ENGINEERING; CULTURE MEDIA
- C12N1/00—Microorganisms, e.g. protozoa; Compositions thereof; Processes of propagating, maintaining or preserving microorganisms or compositions thereof; Processes of preparing or isolating a composition containing a microorganism; Culture media therefor
- C12N1/38—Chemical stimulation of growth or activity by addition of chemical compounds which are not essential growth factors; Stimulation of growth by removal of a chemical compound
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12N—MICROORGANISMS OR ENZYMES; COMPOSITIONS THEREOF; PROPAGATING, PRESERVING, OR MAINTAINING MICROORGANISMS; MUTATION OR GENETIC ENGINEERING; CULTURE MEDIA
- C12N9/00—Enzymes; Proenzymes; Compositions thereof; Processes for preparing, activating, inhibiting, separating or purifying enzymes
Definitions
- the present invention relates to a method of increasing solubility of a polypeptide of interest during fermentation.
- the polypeptides are fermented in yields that are above their solubility limit, meaning that they may be present in the culture broth in a partly precipitated form.
- the precipitate may be in the form of crystals or as amorphous precipitates. This causes problems in recovery where special measures have to be taken to solubilize the crystals/amorphous precipitate before removing the cells and other solids from the culture broth. These measures often result in yield losses.
- the purpose of this invention is therefore to provide a simple and efficient solution to the above described problem.
- the polypeptide of interest can be prevented from crystallizing or precipitating by adding a carbohydrate and/or a polyol and/or a derivative thereof and/or a polymer to the culture medium before and/or during fermentation, wherein the microorganism is not, or only to a low extent, able to metabolize said carbohydrate and/or said polyol and/or said derivative thereof; in particular the present invention deals with:
- a method for fermenting a microorganism, producing a polypeptide of interest, in a culture medium of at least 50 litres comprising: adding one or more compounds selected from the group consisting of 1,2-propandiol, 1,3- propandiol, ethylene glycol, trehalose, xylitol, arabitol, dulcitol, mannitol, erythritol, cellobiose, sorbitol and a polyether having an average molecular weight less than 1000, to the culture medium before and/or during fermentation, wherein the compound is low metabolizable measured by (OD ⁇ -OD ⁇ )/(OD r OD ⁇ ) ⁇ 25% as defined herein.
- the present invention deals with a new and surprisingly effective way of preventing the polypeptide of interest to crystallize or precipitate during the fermentation.
- MPG monopropylene glycol
- microorganisms The microorganism (the microbial strain) according to the invention may be obtained from microorganisms of any genus.
- the polypeptide of interest may be obtained from a bacterial or a fungal source.
- the polypeptide of interest may be obtained from a gram positive bacterium such as a Bacillus strain, e.g., Bacillus alkalophilus, Bacillus atnyloliquefaciens, Bacillus brevis, Bacillus circulans, Bacillus coagulans, Bacillus lautus, Bacillus lentus, Bacillus licheniformis, Bacillus megaterium, Bacillus stearothermophilus, Bacillus subtilis, or Bacillus thuringiensis; or a Streptomyces strain, e.g., Streptomyces IMdans or Streptomyces murinus; or from a gram negative bacterium, e.g., E.
- a Bacillus strain e.g., Bacillus alkalophilus, Bacillus atnyloliquefaciens, Bacillus brevis, Bacillus circulans, Bacillus coagulans, Bacillus lautus, Bacillus lentus,
- the polypeptide of interest may be obtained from a fungal source, e.g. from a yeast strain such as a Candida, Kluyveromyces, Pichia, Saccharomyces, Schizosaccharomyces, or Yarrowia strain, e.g., Saccharomyces carlsbergensis, Saccharomyces cerevisiae, Saccharomyces diastaticus, Saccharomyces douglasii, Saccharomyces kluyveri, Saccharomyces norbensis or Saccharomyces oviformis strain.
- yeast strain such as a Candida, Kluyveromyces, Pichia, Saccharomyces, Schizosaccharomyces, or Yarrowia strain, e.g., Saccharomyces carlsbergensis, Saccharomyces cerevisiae, Saccharomyces diastaticus, Saccharomyces douglasii, Saccharomyces kluyveri
- the polypeptide of interest may be obtained from a filamentous fungal strain such as an Acremonium, Aspergillus, Aureobasidium, Cryptococcus, Filibasidium, Fusarium, Humicola, Magnaporthe, Mucor, Myceliophthora, Neocallimastix, Neurospora, Paecilomyces, Penicillium, Piromyces, Schizophyllum, Talaromyces, Thermoascus, Thielavia, Tolypocladium, or Trichoderma strain, in particular the polypeptide of interest may be obtained from an Aspergillus aculeatus, Aspergillus awamori, Aspergillus foetidus, Aspergillus japonicus, Aspergillus nidulans, Aspergillus niger, Aspergillus oryzae, Fusarium bact dioides, Fusarium cerealis, Fusarium crookwellense, Fu
- the term "obtained from” as used herein in connection with a given source shall mean that the polypeptide of interest is produced by the source or by a cell in which a gene from the source has been inserted.
- microorganism used according to the present invention may be modified in such a way that it is not, or only to a low extent, able to metabolize the chosen carbohydrate and/or polyol and/or derivative thereof; e.g., the original microorganism is able to metabolize glycerol or cyclodextrin but the modified microorganism is not, or only to a low extent.
- the polypeptide of interest may be a peptide or a protein.
- a preferred peptide according to this invention contains from 2 to 100 amino acids; preferably from 10 to 80 amino acids; more preferably from 15 to 60 amino acids; even more preferably from 15 to 40 amino acids.
- the protein is an enzyme, in particular a hydrolase (class 1)
- proteases include those of animal, vegetable or microbial origin. Microbial origin is preferred. Chemically modified or protein engineered mutants are included.
- the protease may be an acid protease, a serine protease or a metallo protease, preferably an alkaline microbial protease or a trypsin-like protease.
- alkaline proteases are subtilisins, especially those derived from Bacillus, e.g., subtilisin Novo, subtilisin Carlsberg, subtilisin 309, subtilisin 147 and subtilisin 168 (described in WO 5 89/06279).
- trypsin-like proteases are trypsin (e.g. of porcine or bovine origin) and the Fusarium protease described in WO 89/06270 and WO 94/25583.
- proteases examples include the variants described in WO 92/19729, WO 98/20115, WO 98/20116, and WO 98/34946, especially the variants with substitutions in one or more of the following positions: 27, 36, 57, 76, 87, 97, 101, 104, 120, 123, 167, 170, 194,
- Preferred commercially available protease enzymes include ALCALASETM, SAVINASETM, PRIMASETM, DURALASETM, ESPERASETM, RELASETM and KANNASETM (Novozymes A/S), MAXATASETM, MAXACALTM, MAXAPEMTM, PROPERASETM, PURAFECTTM, PURAFECT OXPTM, FN2TM, and FN3TM (Genencor International Inc.).
- Suitable lipases include those of bacterial or fungal origin. Chemically modified or protein engineered mutants are included. Examples of useful lipases include lipases from Humicola (synonym Thermomyces), e.g. from H. lanuginosa (T. lanuginosus) as described in EP 258 068 and EP 305 216 or from H. insolens as described in WO 96/13580, a Pseudomonas lipase, e.g. from P. alcaligenes or P. pseudoalcaligenes (EP 218 272), P.
- lipase variants such as those described in WO 92/05249, WO
- Preferred commercially available lipase enzymes include LIPOLASETM, LIPOLASE ULTRATM and LIPEXTM (Novozymes A/S).
- Amylases Suitable amylases (alpha and/or beta) include those of bacterial or fungal origin.
- Amylases include, for example, alpha-amylases obtained from Bacillus, e.g. a special strain of B. licheniformis, described in more detail in GB 1,296,839.
- amylases examples are the variants described in WO 94/02597, WO 94/18314, WO 96/23873, WO 97/43424, and WO 01/66712, especially the variants with
- amylases are DURAMYLTM, TERMAMYLTM, FUNGAMYLTM, NATALASETM, TERMAMYL LCTM, TERMAMYL SCTM, LIQUIZYME-XTM and BANTM (Novozymes A/S), RAPIDASETM and PURASTARTM (from Genencor International Inc.).
- Cellulases include those of bacterial or fungal origin. Chemically modified or protein engineered mutants are included. Suitable cellulases include cellulases from the genera Bacillus, Pseudomonas, Humicola, Fusarium, Thielavia, Acremonium, e.g.
- cellulases are the alkaline or neutral cellulases having colour care benefits.
- Examples of such cellulases are cellulases described in EP 0 495 257, EP 0
- Oxidoreductases Oxidoreductases that may be treated according to the invention include peroxidases, and oxidases such as laccases, and catalases.
- hydrolases are carbohydrolases including MANNAWAYTM.
- Other preferred enzymes are transferases, lyases, isomerases, and ligases.
- the present invention may be useful for any fermentation in industrial scale, e.g. for any fermentation having culture media of at least 50 litres, preferably at least 100 litres, more preferably at least 500 litres, even more preferably at least 1000 litres, in particular at least 5000 litres.
- the microbial strain may be fermented by any method known in the art.
- the fermentation medium may be a complex medium comprising complex nitrogen and/or carbon sources, such as soybean meal, soy protein, soy protein hydrolysate, cotton seed meal, corn steep liquor, yeast extract, casein, casein hydrolysate, potato protein, potato protein hydrolysate, molasses, and the like.
- the fermentation medium may be a chemically defined media, e.g. as defined in WO 98/37179.
- the fermentation may be performed as a batch, a fed-batch, a repeated fed- batch or a continuous fermentation process.
- a fed-batch process either none or part of the compounds comprising one or more of the structural and/or catalytic elements is added to the medium before the start of the fermentation and either all or the remaining part, respectively, of the compounds comprising one or more of the structural and/or catalytic elements is fed during the fermentation process.
- the compounds which are selected for feeding can be fed together or separate from each other to the fermentation process.
- the complete start medium is additionally fed during fermentation.
- the start medium can be fed together with or separate from the structural element feed(s).
- part of the fermentation broth comprising the biomass is removed at time intervals, whereas in a continuous process, the removal of part of the fermentation broth occurs continuously.
- the fermentation process is thereby replenished with a portion of fresh medium corresponding to the amount of withdrawn fermentation broth.
- a fed-batch, a repeated fed-batch process or a continuous fermentation process is preferred.
- Slowly metabolizable carbohydrates such as pullulan, limit dextrin, and trehalose may be used according to the present invention.
- the slowly metabolizable carbohydrate is added to the culture medium either prior to inoculation or after inoculation at an amount of at least 0.1 % (w/w); in particular at an amount of at least 0.5% (w/w).
- the slowly metabolizable carbohydrate is added to the culture medium either prior to inoculation or after inoculation at an amount of up to 10% w/w; preferably at an amount of up to 8% w/w; more preferably at an amount of up to 6% w/w; more preferably at an amount of up to 5% w/w; more preferably at an amount of up to 4% w/w; more preferably at an amount of up to 3% w/w; more preferably at an amount of up to 2% w/w; even more preferably at an amount of up to 1% w/w.
- a very useful subgroup of carbohydrates, polyols may be used according to the invention. Any polyol may be used. However, a polyol selected from the group consisting of 1 ,2-propandiol (monopropylene glycol), 1 ,3-propandiol, glycerol, ethylene glycol, xylitol, arabitol, dulcitol, mannitol, erythritol, cellobiose and sorbitol, is preferred.
- glycerol some polyols, e.g. glycerol, are rather easily metabolized by most cells, but the uptake of e.g. glycerol can be blocked, meaning that glycerol may be used according to the present invention.
- the polyol is added to the culture medium either prior to inoculation or after inoculation at an amount of at least 0.1 % (w/w); in particular at an amount of at least 0.5% (w/w).
- the polyol is added to the culture medium either prior to inoculation or after inoculation at an amount of up to 10% w/w; preferably at an amount of up to 8% w/w; more preferably at an amount of up to 6% w/w; more preferably at an amount of up to 5% w/w; more preferably at an amount of up to 4% w/w; more preferably at an amount of up to 3% w/w; more preferably at an amount of up to 2% w/w; even more preferably at an amount of up to 1 % w/w.
- Derivatives Another very useful subgroup of carbohydrates, derivatives, may be used according to the invention.
- Derivatives that may be used include maillard products, methyl glycosides, glucoronic acids, amino sugars, or N-acetyl glucosamines.
- the derivative is added to the culture medium either prior to inoculation or after inoculation at an amount of at least 0.1 % (w/w); in particular at an amount of at least 0.5% (w/w).
- the derivative is added to the culture medium either prior to inoculation or after inoculation at an amount of up to 10% w/w; preferably at an amount of up to 8% w/w; more preferably at an amount of up to 6% w/w; more preferably at an amount of up to 5% w/w; more preferably at an amount of up to 4% w/w; more preferably at an amount of up to 3% w/w; more preferably at an amount of up to 2% w/w; even more preferably at an amount of up to 1 % w/w.
- Polymers such as polyethers having an average molecular weight less than 1000; preferably an average molecular weight less than 900; more preferably an average molecular weight less than 800; even more preferably an average molecular weight less than 700, (e.g. polyethylene glycol 200 (PEG 200), polyethylene glycol 400 (PEG 400)), or their derivatives including block polymers or block copolymers of polyethylene oxide and polypropylene oxide, wherein the ends of the polymers may further be protected by an acyl group or an alkyl group, may also be used according to the present invention.
- PEG 200 polyethylene glycol 200
- PEG 400 polyethylene glycol 400
- block copolymers of polyethylene oxide and polypropylene oxide wherein the ends of the polymers may further be protected by an acyl group or an alkyl group, may also be used according to the present invention.
- the polymer is added to the culture medium either prior to inoculation or after inoculation at an amount of at least 0.1 % (w/w); in particular at an amount of at least 0.5% (w/w).
- the polymer is added to the culture medium either prior to inoculation or after inoculation at an amount of up to 10% w/w; preferably at an amount of up to 8% w/w; more preferably at an amount of up to 6% w/w; more preferably at an amount of up to 5% w/w; more preferably at an amount of up to 4% w/w; more preferably at an amount of up to 3% w/w; more preferably at an amount of up to 2% w/w; even more preferably at an amount of up to 1 % w/w.
- Salts In addition to adding a slowly metabolizable carbohydrate it may also be an advantage to add a salt to the fermentation medium (see e.g. Example 4).
- a preferred salt is selected from the group consisting of a chloride, a sulphate, a phosphate, a nitrate, and an ammonium salt; e.g. NaCI, KCI, Na2SO4, K2SO4.
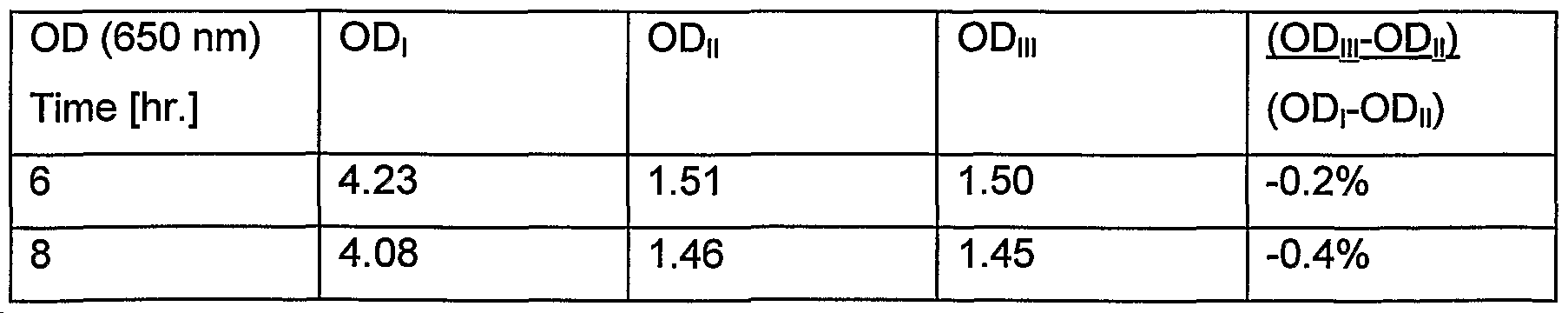
- the following test may be used to check whether a microorganism, producing a polypeptide of interest, is not, or only to a low extent, able to metabolize a given compound:
- a suitable media for the growth of the microorganism of interest is chosen.
- the media is characterized by the following parameters: a: The media contains glucose as the only carbohydrate source. b. When glucose is removed the media should only be able to support growth of a significantly lower biomass (less than 50%).
- the growth of the microorganism of interest is then compared in the following 3 media: I: Normal media (with glucose as the only carbohydrate source) II: Media I without glucose III: Media I without glucose, but with the same C-mol of the compound to be tested.
- the growth is then followed for a period of 8 hr in the 3 above mentioned media. Inoculation is done with a concentration of biomass that will secure that the normal media is outgrown in 75% of the time frame. The amount of biomass is measured as optical density (OD) at 650 nm. OD obtained in the different media is measured.
- OD optical density
- the compound to be tested is defined as low metabolizable, if (OD,i r OD
- -OD ⁇ ) ⁇ 5%; more preferably (OD discardrOD, ⁇ )/(ODrOD, ⁇ ) 0%
- Example 1 various compounds are tested according to this test.
- a further aspect of the invention concerns the downstream processing of the fermentation broth.
- the polypeptide of interest may be recovered from the fermentation broth, using standard technology developed for the polypeptide of interest.
- the relevant downstream processing technology to be applied depends on the nature of the polypeptide of interest.
- a process for the recovery of a polypeptide of interest from a fermentation broth will typically (but is not limited to) involve some or all of the following steps:
- the yield of the polypeptide of interest is much higher in the recovery when the crystal formation is reduced or eliminated by adding of , e.g. MPG, during fermentation.
- Part B 5 g/l Potassiumdihydrogenphosphate pH adjusted to 6.7 with NaOH.
- Part C carbon source equivalent to 0.08 mol carbon per liter (e.g. 2.5 g/l glucose)
- Demineralized water is used for the preparation of all media.
- the media was prepared with 2.5 g/l MPG; 2.5 g/l PEG 200; and 2.5 g/l sucrose, respectively. This should therefore result in a lightly higher OD in these media compared to the intended media, if the compounds were easily metabolized.
- the shake flasks were then incubated at 37°C at 300 rpm in 8 hr.
- MPG is only very slowly or not at all metabolized by the strain used in this example. It is also clear that the culture is fully outgrown after 6 hr as the OD is not increased in medium I going from 6 to 8 hr.
- sucrose is easy metabolized by the strain used in this example.
- Polypeptide of interest an alpha-amylase variant described in WO 01/66712
- LB agar 10 g/l peptone from casein; 5 g/l yeast extract; 10 g/l Sodium Chloride; 12 g/l
- Transfer buffer M-9 buffer (deionized water is used): Di-Sodiumhydrogenphosphate, 2H2O 8.8 g/l;
- PRK-50 110 g/l soy grits; Di-Sodiumhydrogenphosphate, 2H2O 5 g/l; pH adjusted to 8.0 with NaOH/H3PO4 before sterilization.
- the agar was then washed with M-9 buffer, and the optical density (OD) at 650 nm of the resulting cell suspension was measured.
- the shake flask was incubated at 37°C at 300 rpm for 20 hr.
- the fermentation in the main fermentor (fermentation tank) was started by inoculating the main fermentor with the growing culture from the shake flask.
- the inoculated volume was
- Standard lab fermentors were used equipped with a temperature control system, pH control with ammonia water and phosphoric acid, dissolved oxygen electrode to measure >20% oxygen saturation through the entire fermentation.
- the pH was kept between 6.8 and 7.2 using ammonia water and phosphoric acid
- Fermentation B was run as described above but 50 g/L MPG (monopropyleneglycol) was added to the make-up medium before inoculation.
- Fermentation C was run as described above but 50 g/L MPG was added to the fermentation 24 hours after inoculation.
- sample II was centrifuged at 15000 g av in 20 minutes at
- sample I and sample Il sup The alpha-amylase activities in sample I and sample Il sup was then measured by methods known within the art (for example method for alpha-amylase activity measurements described in WO 95/26397 can be used). However, when measuring samples where the enzyme can be in a partly solid form, samples have to be treated with urea prior to analysis.
- sample I and sample ll SUb are diluted in the urea-buffer as described above before being analysed.
- the enzyme activity in sample ll sup is a measure of the soluble activity
- the activity in sample I is a measure of the total activity (both soluble and crystallized and precipitated activity).
- Two fermentations made in a scale larger than 50 liter were made with a Bacillus strain that produces an alpha-amylase with a low solubility.
- One of the fermentations had an addition of 2 % (w/w) MPG 24 hours after inoculation; here enzyme crystals were not visible in a microscope (40X).
- the other fermentation was a reference fermentation without any addition of MPG; here crystals were visible in the broth in a microscope (40X).
- the two batches were treated equal in a series of flocculations where pH was adjusted to 4 different set points and all additions are in % w/w.
- the flocculation consisted of an addition of 200 % water; 2 % CaCI2; 1.2 % AI2(OH)5CI; pH adjustment to a set point (see table below); 0.5 % Superfloc C591 and 0.2 to 0.3 % of Superfloc A130 depending of the pH. All flocculations were based on 100 g culture broth and all additions were done under stirring conditions. The flocculations were done at room temperature (approximately 20°C). The samples were centrifuged and the yield of the alpha-amylase activity were determined in the supernatant fractions.
- Table 6 Flocculation of sludge from first flocculation at different pH to illustrate the efl ect of MPG added to the fermentation.
- the yield is much higher in the recovery of the enzyme if the crystal formation can be reduced or eliminated by addition of MPG during fermentation.
- MPG added is 50 g/l MPG based on the volume of the make-up media before inoculation.
- Salt concentration amounts are based on volume of the make-up media before inoculation.
- Fermentation D is the reference process for this study, while the fermentations E, F, G, H, I had extra mineral salt added to the make-up media in the amounts found in table 7.
- Samples were taken after 3 days of fermentation. The samples were split into two identical parts (sample I and sample II). Sample II was centrifuged at 15000 g av in 20 minutes at 38°C. The resulting supernatant was then filtered through a 0.2 ⁇ m filter (Sartorius Minisart, order no.: 16534) (sample ll sup ).
- the enzyme activity in sample ll sup is a measure of the soluble activity
- the activity in sample I is a measure of the total activity (both soluble and crystallized and precipitated activity).
- Table 8 The following activities were found after 3 days of fermentation.
- the total activity found in fermentation D is set to 100%, and the soluble activity (i.e. the activity in sample ll sup ) is given relative to the activity in sample I for the specific fermentation.
- sample II The reason for the activity being higher in sample II, compared to sample I in some cases is that in the cases where all enzyme is in solution, removal of the cells increases the enzyme concentration in the sample, as the cells have a significant volume, but only a low enzyme concentration.
- a solution of alpha-amylase (e.g. recovered from the broth described in Example 2 by conventional means) is concentrated at pH 10.5-11 at 40°C.
- the concentrate is filtered through a 0.2 ⁇ m filter at 40°C. Aliquots of 25 mL is transferred to 7 vials, to which is then added one of the following polyols:
- a magnetic bar is placed in each vial, and the pH is adjusted to pH 7.5.
- the vials are then left at room temperature with slow stirring for two days.
- the results presented in the table below are given as the activity in the mother liquor relative to the total activity in the sample before filtration (in %).
Abstract
Description
Claims
Priority Applications (5)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| AU2003243925A AU2003243925A1 (en) | 2002-07-01 | 2003-07-01 | Mpg added to fermentation |
| DE60325731T DE60325731D1 (en) | 2002-07-01 | 2003-07-01 | Monopropylenglykol als fermentationszusatz |
| EP03761447A EP1520012B1 (en) | 2002-07-01 | 2003-07-01 | Monopropylene glycol added to fermentation |
| DK03761447T DK1520012T3 (en) | 2002-07-01 | 2003-07-01 | Monopropylene glycol added to fermentation medium |
| US10/519,432 US20060127974A1 (en) | 2002-07-01 | 2003-07-01 | Mpg added to fermentation |
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| DKPA200201021 | 2002-07-01 | ||
| DKPA200201021 | 2002-07-01 |
Publications (2)
| Publication Number | Publication Date |
|---|---|
| WO2004003187A2 true WO2004003187A2 (en) | 2004-01-08 |
| WO2004003187A3 WO2004003187A3 (en) | 2004-03-18 |
Family
ID=29797022
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/DK2003/000455 WO2004003187A2 (en) | 2002-07-01 | 2003-07-01 | Mpg added to fermentation |
Country Status (8)
| Country | Link |
|---|---|
| US (1) | US20060127974A1 (en) |
| EP (1) | EP1520012B1 (en) |
| CN (1) | CN100529066C (en) |
| AT (1) | ATE420161T1 (en) |
| AU (1) | AU2003243925A1 (en) |
| DE (1) | DE60325731D1 (en) |
| DK (1) | DK1520012T3 (en) |
| WO (1) | WO2004003187A2 (en) |
Cited By (4)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2006136164A1 (en) * | 2005-06-24 | 2006-12-28 | Novozymes A/S | Mutant cells suitable for recombinant polypeptide production |
| CN100999742B (en) * | 2007-01-09 | 2011-05-11 | 清华大学 | High yield technology of producing 1,3 propyl diol by fermenting |
| EP2285823B1 (en) | 2008-06-09 | 2016-04-27 | Danisco US Inc. | Recovery of insoluble enzyme from fermentation broth and formulation of insoluble enzyme |
| WO2020249546A1 (en) | 2019-06-13 | 2020-12-17 | Basf Se | Method of recovering a protein from fermentation broth using a divalent cation |
Families Citing this family (2)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN104480152B (en) * | 2014-11-20 | 2017-09-01 | 武汉华士特工业生物技术开发有限公司 | A kind of method for improving docosahexaenoic acid content in schizochytrium limacinum grease |
| CA3083390A1 (en) * | 2017-11-29 | 2019-06-06 | Basf Se | Storage-stable enzyme preparations, their production and use |
Citations (52)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| GB1296839A (en) | 1969-05-29 | 1972-11-22 | ||
| GB1372034A (en) | 1970-12-31 | 1974-10-30 | Unilever Ltd | Detergent compositions |
| US4435307A (en) | 1980-04-30 | 1984-03-06 | Novo Industri A/S | Detergent cellulase |
| EP0218272A1 (en) | 1985-08-09 | 1987-04-15 | Gist-Brocades N.V. | Novel lipolytic enzymes and their use in detergent compositions |
| EP0258068A2 (en) | 1986-08-29 | 1988-03-02 | Novo Nordisk A/S | Enzymatic detergent additive |
| EP0260105A2 (en) | 1986-09-09 | 1988-03-16 | Genencor, Inc. | Preparation of enzymes having altered activity |
| EP0305216A1 (en) | 1987-08-28 | 1989-03-01 | Novo Nordisk A/S | Recombinant Humicola lipase and process for the production of recombinant humicola lipases |
| JPS6474992A (en) | 1987-09-16 | 1989-03-20 | Fuji Oil Co Ltd | Dna sequence, plasmid and production of lipase |
| WO1989006270A1 (en) | 1988-01-07 | 1989-07-13 | Novo-Nordisk A/S | Enzymatic detergent |
| WO1989006279A1 (en) | 1988-01-07 | 1989-07-13 | Novo-Nordisk A/S | Mutated subtilisin genes |
| EP0331376A2 (en) | 1988-02-28 | 1989-09-06 | Amano Pharmaceutical Co., Ltd. | Recombinant DNA, bacterium of the genus pseudomonas containing it, and process for preparing lipase by using it |
| WO1989009259A1 (en) | 1988-03-24 | 1989-10-05 | Novo-Nordisk A/S | A cellulase preparation |
| EP0407225A1 (en) | 1989-07-07 | 1991-01-09 | Unilever Plc | Enzymes and enzymatic detergent compositions |
| WO1991016422A1 (en) | 1990-04-14 | 1991-10-31 | Kali-Chemie Aktiengesellschaft | Alkaline bacillus lipases, coding dna sequences therefor and bacilli which produce these lipases |
| WO1992005249A1 (en) | 1990-09-13 | 1992-04-02 | Novo Nordisk A/S | Lipase variants |
| EP0495257A1 (en) | 1991-01-16 | 1992-07-22 | The Procter & Gamble Company | Compact detergent compositions with high activity cellulase |
| WO1992019729A1 (en) | 1991-05-01 | 1992-11-12 | Novo Nordisk A/S | Stabilized enzymes and detergent compositions |
| EP0531372A1 (en) | 1990-05-09 | 1993-03-17 | Novo Nordisk As | A cellulase preparation comprising an endoglucanase enzyme. |
| EP0531315A1 (en) | 1990-05-09 | 1993-03-17 | Novo Nordisk As | An enzyme capable of degrading cellulose or hemicellulose. |
| US5260202A (en) | 1988-09-07 | 1993-11-09 | Delta Biotechnology Limited | Fermentation method |
| WO1994001541A1 (en) | 1992-07-06 | 1994-01-20 | Novo Nordisk A/S | C. antarctica lipase and lipase variants |
| WO1994002597A1 (en) | 1992-07-23 | 1994-02-03 | Novo Nordisk A/S | MUTANT α-AMYLASE, DETERGENT, DISH WASHING AGENT, AND LIQUEFACTION AGENT |
| WO1994007998A1 (en) | 1992-10-06 | 1994-04-14 | Novo Nordisk A/S | Cellulase variants |
| WO1994018314A1 (en) | 1993-02-11 | 1994-08-18 | Genencor International, Inc. | Oxidatively stable alpha-amylase |
| WO1994025578A1 (en) | 1993-04-27 | 1994-11-10 | Gist-Brocades N.V. | New lipase variants for use in detergent applications |
| WO1994025583A1 (en) | 1993-05-05 | 1994-11-10 | Novo Nordisk A/S | A recombinant trypsin-like protease |
| WO1995006720A1 (en) | 1993-08-30 | 1995-03-09 | Showa Denko K.K. | Novel lipase, microorganism producing the lipase, process for producing the lipase, and use of the lipase |
| WO1995014783A1 (en) | 1993-11-24 | 1995-06-01 | Showa Denko K.K. | Lipase gene and variant lipase |
| WO1995022615A1 (en) | 1994-02-22 | 1995-08-24 | Novo Nordisk A/S | A method of preparing a variant of a lipolytic enzyme |
| WO1995024471A1 (en) | 1994-03-08 | 1995-09-14 | Novo Nordisk A/S | Novel alkaline cellulases |
| WO1995026397A1 (en) | 1994-03-29 | 1995-10-05 | Novo Nordisk A/S | Alkaline bacillus amylase |
| WO1995030744A2 (en) | 1994-05-04 | 1995-11-16 | Genencor International Inc. | Lipases with improved surfactant resistance |
| WO1995035381A1 (en) | 1994-06-20 | 1995-12-28 | Unilever N.V. | Modified pseudomonas lipases and their use |
| WO1996000292A1 (en) | 1994-06-23 | 1996-01-04 | Unilever N.V. | Modified pseudomonas lipases and their use |
| WO1996011262A1 (en) | 1994-10-06 | 1996-04-18 | Novo Nordisk A/S | An enzyme and enzyme preparation with endoglucanase activity |
| WO1996012012A1 (en) | 1994-10-14 | 1996-04-25 | Solvay S.A. | Lipase, microorganism producing same, method for preparing said lipase and uses thereof |
| WO1996013580A1 (en) | 1994-10-26 | 1996-05-09 | Novo Nordisk A/S | An enzyme with lipolytic activity |
| WO1996023873A1 (en) | 1995-02-03 | 1996-08-08 | Novo Nordisk A/S | Amylase variants |
| WO1996027002A1 (en) | 1995-02-27 | 1996-09-06 | Novo Nordisk A/S | Novel lipase gene and process for the production of lipase with the use of the same |
| WO1996029397A1 (en) | 1995-03-17 | 1996-09-26 | Novo Nordisk A/S | Novel endoglucanases |
| WO1997004079A1 (en) | 1995-07-14 | 1997-02-06 | Novo Nordisk A/S | A modified enzyme with lipolytic activity |
| WO1997007202A1 (en) | 1995-08-11 | 1997-02-27 | Novo Nordisk A/S | Novel lipolytic enzymes |
| US5648263A (en) | 1988-03-24 | 1997-07-15 | Novo Nordisk A/S | Methods for reducing the harshness of a cotton-containing fabric |
| WO1997043424A1 (en) | 1996-05-14 | 1997-11-20 | Genencor International, Inc. | MODIFIED α-AMYLASES HAVING ALTERED CALCIUM BINDING PROPERTIES |
| WO1998008940A1 (en) | 1996-08-26 | 1998-03-05 | Novo Nordisk A/S | A novel endoglucanase |
| WO1998012307A1 (en) | 1996-09-17 | 1998-03-26 | Novo Nordisk A/S | Cellulase variants |
| WO1998020115A1 (en) | 1996-11-04 | 1998-05-14 | Novo Nordisk A/S | Subtilase variants and compositions |
| WO1998020116A1 (en) | 1996-11-04 | 1998-05-14 | Novo Nordisk A/S | Subtilase variants and compositions |
| WO1998034946A1 (en) | 1997-02-12 | 1998-08-13 | Massachusetts Institute Of Technology | Daxx, a novel fas-binding protein that activates jnk and apoptosis |
| WO1998037179A2 (en) | 1997-02-20 | 1998-08-27 | Dsm N.V. | Fermentative production of valuable compounds on an industrial scale using chemically defined media |
| WO1999001544A1 (en) | 1997-07-04 | 1999-01-14 | Novo Nordisk A/S | FAMILY 6 ENDO-1,4-β-GLUCANASE VARIANTS AND CLEANING COMPOSIT IONS CONTAINING THEM |
| WO2001066712A2 (en) | 2000-03-08 | 2001-09-13 | Novozymes A/S | Variants with altered properties |
Family Cites Families (19)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US3111459A (en) * | 1959-10-19 | 1963-11-19 | Ajinomoto Kk | Method for preparation of inosine |
| US3222257A (en) * | 1961-04-19 | 1965-12-07 | Ajinomoto Kk | Process for producing nucleosides by microorganisms |
| GB1001173A (en) * | 1962-10-22 | 1965-08-11 | Miles Lab | Process for the production of galactose oxidase |
| US3951937A (en) * | 1973-12-20 | 1976-04-20 | The Community Blood Council Of Greater New York, Inc. | Large scale purification of hepatitis type B antigen using polyethylene glycol |
| DE2512735A1 (en) * | 1975-03-22 | 1976-09-30 | Henkel & Cie Gmbh | PROCESS FOR THE EXTRACTION OF PROTEINS FROM Aqueous PROTEIN SOLUTIONS |
| US4069216A (en) * | 1975-06-16 | 1978-01-17 | Edward Shanbrom, Inc. | Simplified methods for preparation of very high purity Factor VIII concentrate |
| JPS5953038B2 (en) * | 1979-04-07 | 1984-12-22 | メルシャン株式会社 | Manufacturing method of cyclodextrin |
| DD153495A3 (en) * | 1979-07-03 | 1982-01-13 | Petrolchemisches Kombinat | AUXILIARY COMBINATION TO IMPROVE MICROBIOLOGICAL PROCEDURES |
| JPS6030682A (en) * | 1983-07-30 | 1985-02-16 | Amano Pharmaceut Co Ltd | Production of beta-amylase |
| US4588691A (en) * | 1984-02-29 | 1986-05-13 | Johal Sarjit S | Method for preparation of Fraction I protein and by-products thereof |
| US4673647A (en) * | 1985-05-06 | 1987-06-16 | Miles Laboratories, Inc. | Process to solubilize enzymes and an enzyme liquid product produced thereby |
| US4728613A (en) * | 1985-09-04 | 1988-03-01 | Miles Laboratories, Inc. | Method for the recovery of extracellular enzymes from whole fermentation beer |
| CH667673A5 (en) * | 1988-01-22 | 1988-10-31 | Eidgenoess Tech Hochschule | Prodn. of fermentation broth with lignolytic activity - by growing fungi under nutrient limited conditions in stirred reactor and in presence of cell wall stabiliser |
| US5371198A (en) * | 1991-12-16 | 1994-12-06 | Novo Nordisk A/S | Method for protection of proteolysis-susceptible protein during protein production in a fluid medium |
| DE69333463D1 (en) * | 1992-05-18 | 2004-05-06 | Genencor Int | Bacteria producing alkaline proteases, and process for producing these alkaline proteases |
| JP3254457B2 (en) * | 1992-09-18 | 2002-02-04 | 株式会社日立製作所 | Method for forming rotor of oilless screw compressor and oilless screw compressor using the rotor |
| US5443981A (en) * | 1993-06-09 | 1995-08-22 | Rutgers, The State University | Acremonium typhinum ATCC 74228 which exhibits endoproteolytic activity |
| US5616693A (en) * | 1996-07-01 | 1997-04-01 | Alpha Therapeutic Corporation | Process for seperating alpha-1-proteinase inhibitor from COHN IV1 +1V4 paste |
| AU2002331306A1 (en) * | 2001-08-07 | 2003-02-24 | Novozymes North America, Inc. | Carbohydrates and polyols for dissolving protein crystals |
-
2003
- 2003-07-01 WO PCT/DK2003/000455 patent/WO2004003187A2/en not_active Application Discontinuation
- 2003-07-01 CN CNB038155788A patent/CN100529066C/en not_active Expired - Fee Related
- 2003-07-01 AU AU2003243925A patent/AU2003243925A1/en not_active Abandoned
- 2003-07-01 DE DE60325731T patent/DE60325731D1/en not_active Expired - Lifetime
- 2003-07-01 DK DK03761447T patent/DK1520012T3/en active
- 2003-07-01 EP EP03761447A patent/EP1520012B1/en not_active Expired - Lifetime
- 2003-07-01 US US10/519,432 patent/US20060127974A1/en not_active Abandoned
- 2003-07-01 AT AT03761447T patent/ATE420161T1/en not_active IP Right Cessation
Patent Citations (57)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| GB1296839A (en) | 1969-05-29 | 1972-11-22 | ||
| GB1372034A (en) | 1970-12-31 | 1974-10-30 | Unilever Ltd | Detergent compositions |
| US4435307A (en) | 1980-04-30 | 1984-03-06 | Novo Industri A/S | Detergent cellulase |
| EP0218272A1 (en) | 1985-08-09 | 1987-04-15 | Gist-Brocades N.V. | Novel lipolytic enzymes and their use in detergent compositions |
| EP0258068A2 (en) | 1986-08-29 | 1988-03-02 | Novo Nordisk A/S | Enzymatic detergent additive |
| EP0260105A2 (en) | 1986-09-09 | 1988-03-16 | Genencor, Inc. | Preparation of enzymes having altered activity |
| EP0305216A1 (en) | 1987-08-28 | 1989-03-01 | Novo Nordisk A/S | Recombinant Humicola lipase and process for the production of recombinant humicola lipases |
| JPS6474992A (en) | 1987-09-16 | 1989-03-20 | Fuji Oil Co Ltd | Dna sequence, plasmid and production of lipase |
| WO1989006270A1 (en) | 1988-01-07 | 1989-07-13 | Novo-Nordisk A/S | Enzymatic detergent |
| WO1989006279A1 (en) | 1988-01-07 | 1989-07-13 | Novo-Nordisk A/S | Mutated subtilisin genes |
| EP0331376A2 (en) | 1988-02-28 | 1989-09-06 | Amano Pharmaceutical Co., Ltd. | Recombinant DNA, bacterium of the genus pseudomonas containing it, and process for preparing lipase by using it |
| US5691178A (en) | 1988-03-22 | 1997-11-25 | Novo Nordisk A/S | Fungal cellulase composition containing alkaline CMC-endoglucanase and essentially no cellobiohydrolase |
| WO1989009259A1 (en) | 1988-03-24 | 1989-10-05 | Novo-Nordisk A/S | A cellulase preparation |
| US5648263A (en) | 1988-03-24 | 1997-07-15 | Novo Nordisk A/S | Methods for reducing the harshness of a cotton-containing fabric |
| US5776757A (en) | 1988-03-24 | 1998-07-07 | Novo Nordisk A/S | Fungal cellulase composition containing alkaline CMC-endoglucanase and essentially no cellobiohydrolase and method of making thereof |
| US5260202A (en) | 1988-09-07 | 1993-11-09 | Delta Biotechnology Limited | Fermentation method |
| EP0407225A1 (en) | 1989-07-07 | 1991-01-09 | Unilever Plc | Enzymes and enzymatic detergent compositions |
| WO1991016422A1 (en) | 1990-04-14 | 1991-10-31 | Kali-Chemie Aktiengesellschaft | Alkaline bacillus lipases, coding dna sequences therefor and bacilli which produce these lipases |
| EP0531372A1 (en) | 1990-05-09 | 1993-03-17 | Novo Nordisk As | A cellulase preparation comprising an endoglucanase enzyme. |
| EP0531315A1 (en) | 1990-05-09 | 1993-03-17 | Novo Nordisk As | An enzyme capable of degrading cellulose or hemicellulose. |
| US5686593A (en) | 1990-05-09 | 1997-11-11 | Novo Nordisk A/S | Enzyme capable of degrading cellulose or hemicellulose |
| US5457046A (en) | 1990-05-09 | 1995-10-10 | Novo Nordisk A/S | Enzyme capable of degrading cellullose or hemicellulose |
| US5763254A (en) | 1990-05-09 | 1998-06-09 | Novo Nordisk A/S | Enzyme capable of degrading cellulose or hemicellulose |
| WO1992005249A1 (en) | 1990-09-13 | 1992-04-02 | Novo Nordisk A/S | Lipase variants |
| EP0495257A1 (en) | 1991-01-16 | 1992-07-22 | The Procter & Gamble Company | Compact detergent compositions with high activity cellulase |
| WO1992019729A1 (en) | 1991-05-01 | 1992-11-12 | Novo Nordisk A/S | Stabilized enzymes and detergent compositions |
| WO1994001541A1 (en) | 1992-07-06 | 1994-01-20 | Novo Nordisk A/S | C. antarctica lipase and lipase variants |
| WO1994002597A1 (en) | 1992-07-23 | 1994-02-03 | Novo Nordisk A/S | MUTANT α-AMYLASE, DETERGENT, DISH WASHING AGENT, AND LIQUEFACTION AGENT |
| WO1994007998A1 (en) | 1992-10-06 | 1994-04-14 | Novo Nordisk A/S | Cellulase variants |
| WO1994018314A1 (en) | 1993-02-11 | 1994-08-18 | Genencor International, Inc. | Oxidatively stable alpha-amylase |
| WO1994025578A1 (en) | 1993-04-27 | 1994-11-10 | Gist-Brocades N.V. | New lipase variants for use in detergent applications |
| WO1994025583A1 (en) | 1993-05-05 | 1994-11-10 | Novo Nordisk A/S | A recombinant trypsin-like protease |
| WO1995006720A1 (en) | 1993-08-30 | 1995-03-09 | Showa Denko K.K. | Novel lipase, microorganism producing the lipase, process for producing the lipase, and use of the lipase |
| WO1995014783A1 (en) | 1993-11-24 | 1995-06-01 | Showa Denko K.K. | Lipase gene and variant lipase |
| WO1995022615A1 (en) | 1994-02-22 | 1995-08-24 | Novo Nordisk A/S | A method of preparing a variant of a lipolytic enzyme |
| WO1995024471A1 (en) | 1994-03-08 | 1995-09-14 | Novo Nordisk A/S | Novel alkaline cellulases |
| WO1995026397A1 (en) | 1994-03-29 | 1995-10-05 | Novo Nordisk A/S | Alkaline bacillus amylase |
| WO1995030744A2 (en) | 1994-05-04 | 1995-11-16 | Genencor International Inc. | Lipases with improved surfactant resistance |
| WO1995035381A1 (en) | 1994-06-20 | 1995-12-28 | Unilever N.V. | Modified pseudomonas lipases and their use |
| WO1996000292A1 (en) | 1994-06-23 | 1996-01-04 | Unilever N.V. | Modified pseudomonas lipases and their use |
| WO1996011262A1 (en) | 1994-10-06 | 1996-04-18 | Novo Nordisk A/S | An enzyme and enzyme preparation with endoglucanase activity |
| WO1996012012A1 (en) | 1994-10-14 | 1996-04-25 | Solvay S.A. | Lipase, microorganism producing same, method for preparing said lipase and uses thereof |
| WO1996013580A1 (en) | 1994-10-26 | 1996-05-09 | Novo Nordisk A/S | An enzyme with lipolytic activity |
| WO1996023873A1 (en) | 1995-02-03 | 1996-08-08 | Novo Nordisk A/S | Amylase variants |
| WO1996027002A1 (en) | 1995-02-27 | 1996-09-06 | Novo Nordisk A/S | Novel lipase gene and process for the production of lipase with the use of the same |
| WO1996029397A1 (en) | 1995-03-17 | 1996-09-26 | Novo Nordisk A/S | Novel endoglucanases |
| WO1997004079A1 (en) | 1995-07-14 | 1997-02-06 | Novo Nordisk A/S | A modified enzyme with lipolytic activity |
| WO1997007202A1 (en) | 1995-08-11 | 1997-02-27 | Novo Nordisk A/S | Novel lipolytic enzymes |
| WO1997043424A1 (en) | 1996-05-14 | 1997-11-20 | Genencor International, Inc. | MODIFIED α-AMYLASES HAVING ALTERED CALCIUM BINDING PROPERTIES |
| WO1998008940A1 (en) | 1996-08-26 | 1998-03-05 | Novo Nordisk A/S | A novel endoglucanase |
| WO1998012307A1 (en) | 1996-09-17 | 1998-03-26 | Novo Nordisk A/S | Cellulase variants |
| WO1998020115A1 (en) | 1996-11-04 | 1998-05-14 | Novo Nordisk A/S | Subtilase variants and compositions |
| WO1998020116A1 (en) | 1996-11-04 | 1998-05-14 | Novo Nordisk A/S | Subtilase variants and compositions |
| WO1998034946A1 (en) | 1997-02-12 | 1998-08-13 | Massachusetts Institute Of Technology | Daxx, a novel fas-binding protein that activates jnk and apoptosis |
| WO1998037179A2 (en) | 1997-02-20 | 1998-08-27 | Dsm N.V. | Fermentative production of valuable compounds on an industrial scale using chemically defined media |
| WO1999001544A1 (en) | 1997-07-04 | 1999-01-14 | Novo Nordisk A/S | FAMILY 6 ENDO-1,4-β-GLUCANASE VARIANTS AND CLEANING COMPOSIT IONS CONTAINING THEM |
| WO2001066712A2 (en) | 2000-03-08 | 2001-09-13 | Novozymes A/S | Variants with altered properties |
Non-Patent Citations (1)
| Title |
|---|
| DARTOIS ET AL., BIOCHEMICA ET BIOPHYSICA ACTA, vol. 1131, 1993, pages 253 - 360 |
Cited By (5)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2006136164A1 (en) * | 2005-06-24 | 2006-12-28 | Novozymes A/S | Mutant cells suitable for recombinant polypeptide production |
| CN100999742B (en) * | 2007-01-09 | 2011-05-11 | 清华大学 | High yield technology of producing 1,3 propyl diol by fermenting |
| EP2285823B1 (en) | 2008-06-09 | 2016-04-27 | Danisco US Inc. | Recovery of insoluble enzyme from fermentation broth and formulation of insoluble enzyme |
| EP2285823B2 (en) † | 2008-06-09 | 2024-03-06 | Danisco US Inc. | Recovery of insoluble enzyme from fermentation broth and formulation of insoluble enzyme |
| WO2020249546A1 (en) | 2019-06-13 | 2020-12-17 | Basf Se | Method of recovering a protein from fermentation broth using a divalent cation |
Also Published As
| Publication number | Publication date |
|---|---|
| AU2003243925A8 (en) | 2004-01-19 |
| CN1665923A (en) | 2005-09-07 |
| DK1520012T3 (en) | 2009-04-27 |
| DE60325731D1 (en) | 2009-02-26 |
| WO2004003187A3 (en) | 2004-03-18 |
| US20060127974A1 (en) | 2006-06-15 |
| ATE420161T1 (en) | 2009-01-15 |
| EP1520012B1 (en) | 2009-01-07 |
| AU2003243925A1 (en) | 2004-01-19 |
| EP1520012A2 (en) | 2005-04-06 |
| CN100529066C (en) | 2009-08-19 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| Negi et al. | Optimization of culture parameters to enhance production of amylase and protease from Aspergillus awamori in a single fermentation | |
| EP3959326B1 (en) | Industrial fermentation process for microbial cells using a fed-batch pre-culture | |
| WO2004003216A2 (en) | Sterilization of a fermentation medium comprising hydrolysed n-source | |
| JP6306519B2 (en) | Advanced fermentation control | |
| Senthilkumar et al. | Amylase production by Bacillus sp. using cassava as substrate | |
| CN100487114C (en) | Crystal harvest from fermentation broth | |
| WO2004003187A2 (en) | Mpg added to fermentation | |
| US20070015254A1 (en) | Crystal harvest from fermentation broth | |
| JP2014521356A (en) | Reduction of culture viscosity by adding manganese | |
| US20120122183A1 (en) | Flocculation with divalent salt and phosphate | |
| US20130309722A1 (en) | Use of browned glucose as a feed substrate | |
| US8889395B2 (en) | Crystal metabolite recovery | |
| US20100173286A1 (en) | Mutant Cells Suitable for Recombinant Polypeptide Production | |
| US7019120B2 (en) | Cloud-point extraction of enzymes and polypeptides from a fermentation broth using a non-ionic surfactant | |
| CN111032856A (en) | Use of FCA control based on pH | |
| EP2213723A1 (en) | Isomaltose for fungus fermentation | |
| US20220275355A1 (en) | Method of Recovering a Protein from Fermentation Broth Using a Divalent Cation | |
| US20040033567A1 (en) | Hydrolysed N-source |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| AK | Designated states |
Kind code of ref document: A2 Designated state(s): AE AG AL AM AT AU AZ BA BB BG BR BY BZ CA CH CN CO CR CU CZ DE DK DM DZ EC EE ES FI GB GD GE GH GM HR HU ID IL IN IS JP KE KG KP KR KZ LC LK LR LS LT LU LV MA MD MG MK MN MW MX MZ NI NO NZ OM PG PH PL PT RO RU SC SD SE SG SK SL SY TJ TM TN TR TT TZ UA UG US UZ VC VN YU ZA ZM ZW |
|
| AL | Designated countries for regional patents |
Kind code of ref document: A2 Designated state(s): GH GM KE LS MW MZ SD SL SZ TZ UG ZM ZW AM AZ BY KG KZ MD RU TJ TM AT BE BG CH CY CZ DE DK EE ES FI FR GB GR HU IE IT LU MC NL PT RO SE SI SK TR BF BJ CF CG CI CM GA GN GQ GW ML MR NE SN TD TG |
|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application | ||
| WWE | Wipo information: entry into national phase |
Ref document number: 2003761447 Country of ref document: EP |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 20038155788 Country of ref document: CN |
|
| WWP | Wipo information: published in national office |
Ref document number: 2003761447 Country of ref document: EP |
|
| ENP | Entry into the national phase |
Ref document number: 2006127974 Country of ref document: US Kind code of ref document: A1 |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 10519432 Country of ref document: US |
|
| WWP | Wipo information: published in national office |
Ref document number: 10519432 Country of ref document: US |
|
| NENP | Non-entry into the national phase |
Ref country code: JP |
|
| WWW | Wipo information: withdrawn in national office |
Country of ref document: JP |