WO2001035943A2 - Dextrose and insulin fluid formulation for intravenous infusion - Google Patents
Dextrose and insulin fluid formulation for intravenous infusion Download PDFInfo
- Publication number
- WO2001035943A2 WO2001035943A2 PCT/IN2000/000110 IN0000110W WO0135943A2 WO 2001035943 A2 WO2001035943 A2 WO 2001035943A2 IN 0000110 W IN0000110 W IN 0000110W WO 0135943 A2 WO0135943 A2 WO 0135943A2
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- dextrose
- insulin
- vitamins
- fluids
- solutions
- Prior art date
Links
Classifications
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K9/00—Medicinal preparations characterised by special physical form
- A61K9/0012—Galenical forms characterised by the site of application
- A61K9/0019—Injectable compositions; Intramuscular, intravenous, arterial, subcutaneous administration; Compositions to be administered through the skin in an invasive manner
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K31/00—Medicinal preparations containing organic active ingredients
- A61K31/045—Hydroxy compounds, e.g. alcohols; Salts thereof, e.g. alcoholates
- A61K31/07—Retinol compounds, e.g. vitamin A
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K31/00—Medicinal preparations containing organic active ingredients
- A61K31/13—Amines
- A61K31/14—Quaternary ammonium compounds, e.g. edrophonium, choline
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K31/00—Medicinal preparations containing organic active ingredients
- A61K31/16—Amides, e.g. hydroxamic acids
- A61K31/164—Amides, e.g. hydroxamic acids of a carboxylic acid with an aminoalcohol, e.g. ceramides
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K31/00—Medicinal preparations containing organic active ingredients
- A61K31/185—Acids; Anhydrides, halides or salts thereof, e.g. sulfur acids, imidic, hydrazonic or hydroximic acids
- A61K31/19—Carboxylic acids, e.g. valproic acid
- A61K31/195—Carboxylic acids, e.g. valproic acid having an amino group
- A61K31/197—Carboxylic acids, e.g. valproic acid having an amino group the amino and the carboxyl groups being attached to the same acyclic carbon chain, e.g. gamma-aminobutyric acid [GABA], beta-alanine, epsilon-aminocaproic acid, pantothenic acid
- A61K31/198—Alpha-aminoacids, e.g. alanine, edetic acids [EDTA]
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K31/00—Medicinal preparations containing organic active ingredients
- A61K31/33—Heterocyclic compounds
- A61K31/335—Heterocyclic compounds having oxygen as the only ring hetero atom, e.g. fungichromin
- A61K31/35—Heterocyclic compounds having oxygen as the only ring hetero atom, e.g. fungichromin having six-membered rings with one oxygen as the only ring hetero atom
- A61K31/352—Heterocyclic compounds having oxygen as the only ring hetero atom, e.g. fungichromin having six-membered rings with one oxygen as the only ring hetero atom condensed with carbocyclic rings, e.g. methantheline
- A61K31/353—3,4-Dihydrobenzopyrans, e.g. chroman, catechin
- A61K31/355—Tocopherols, e.g. vitamin E
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K31/00—Medicinal preparations containing organic active ingredients
- A61K31/33—Heterocyclic compounds
- A61K31/335—Heterocyclic compounds having oxygen as the only ring hetero atom, e.g. fungichromin
- A61K31/365—Lactones
- A61K31/375—Ascorbic acid, i.e. vitamin C; Salts thereof
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K31/00—Medicinal preparations containing organic active ingredients
- A61K31/33—Heterocyclic compounds
- A61K31/395—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins
- A61K31/435—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins having six-membered rings with one nitrogen as the only ring hetero atom
- A61K31/44—Non condensed pyridines; Hydrogenated derivatives thereof
- A61K31/4415—Pyridoxine, i.e. Vitamin B6
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K31/00—Medicinal preparations containing organic active ingredients
- A61K31/33—Heterocyclic compounds
- A61K31/395—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins
- A61K31/435—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins having six-membered rings with one nitrogen as the only ring hetero atom
- A61K31/44—Non condensed pyridines; Hydrogenated derivatives thereof
- A61K31/455—Nicotinic acids, e.g. niacin; Derivatives thereof, e.g. esters, amides
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K31/00—Medicinal preparations containing organic active ingredients
- A61K31/33—Heterocyclic compounds
- A61K31/395—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins
- A61K31/495—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins having six-membered rings with two or more nitrogen atoms as the only ring heteroatoms, e.g. piperazine or tetrazines
- A61K31/505—Pyrimidines; Hydrogenated pyrimidines, e.g. trimethoprim
- A61K31/506—Pyrimidines; Hydrogenated pyrimidines, e.g. trimethoprim not condensed and containing further heterocyclic rings
- A61K31/51—Thiamines, e.g. vitamin B1
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K31/00—Medicinal preparations containing organic active ingredients
- A61K31/33—Heterocyclic compounds
- A61K31/395—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins
- A61K31/495—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins having six-membered rings with two or more nitrogen atoms as the only ring heteroatoms, e.g. piperazine or tetrazines
- A61K31/505—Pyrimidines; Hydrogenated pyrimidines, e.g. trimethoprim
- A61K31/519—Pyrimidines; Hydrogenated pyrimidines, e.g. trimethoprim ortho- or peri-condensed with heterocyclic rings
- A61K31/525—Isoalloxazines, e.g. riboflavins, vitamin B2
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K31/00—Medicinal preparations containing organic active ingredients
- A61K31/59—Compounds containing 9, 10- seco- cyclopenta[a]hydrophenanthrene ring systems
- A61K31/593—9,10-Secocholestane derivatives, e.g. cholecalciferol, i.e. vitamin D3
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K31/00—Medicinal preparations containing organic active ingredients
- A61K31/70—Carbohydrates; Sugars; Derivatives thereof
- A61K31/7004—Monosaccharides having only carbon, hydrogen and oxygen atoms
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K31/00—Medicinal preparations containing organic active ingredients
- A61K31/70—Carbohydrates; Sugars; Derivatives thereof
- A61K31/7135—Compounds containing heavy metals
- A61K31/714—Cobalamins, e.g. cyanocobalamin, i.e. vitamin B12
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K31/00—Medicinal preparations containing organic active ingredients
- A61K31/70—Carbohydrates; Sugars; Derivatives thereof
- A61K31/715—Polysaccharides, i.e. having more than five saccharide radicals attached to each other by glycosidic linkages; Derivatives thereof, e.g. ethers, esters
- A61K31/726—Glycosaminoglycans, i.e. mucopolysaccharides
- A61K31/727—Heparin; Heparan
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K33/00—Medicinal preparations containing inorganic active ingredients
- A61K33/04—Sulfur, selenium or tellurium; Compounds thereof
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K33/00—Medicinal preparations containing inorganic active ingredients
- A61K33/14—Alkali metal chlorides; Alkaline earth metal chlorides
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K33/00—Medicinal preparations containing inorganic active ingredients
- A61K33/42—Phosphorus; Compounds thereof
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K38/00—Medicinal preparations containing peptides
- A61K38/04—Peptides having up to 20 amino acids in a fully defined sequence; Derivatives thereof
- A61K38/05—Dipeptides
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K38/00—Medicinal preparations containing peptides
- A61K38/16—Peptides having more than 20 amino acids; Gastrins; Somatostatins; Melanotropins; Derivatives thereof
- A61K38/17—Peptides having more than 20 amino acids; Gastrins; Somatostatins; Melanotropins; Derivatives thereof from animals; from humans
- A61K38/22—Hormones
- A61K38/28—Insulins
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K45/00—Medicinal preparations containing active ingredients not provided for in groups A61K31/00 - A61K41/00
- A61K45/06—Mixtures of active ingredients without chemical characterisation, e.g. antiphlogistics and cardiaca
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K47/00—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient
- A61K47/02—Inorganic compounds
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K47/00—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient
- A61K47/06—Organic compounds, e.g. natural or synthetic hydrocarbons, polyolefins, mineral oil, petrolatum or ozokerite
- A61K47/26—Carbohydrates, e.g. sugar alcohols, amino sugars, nucleic acids, mono-, di- or oligo-saccharides; Derivatives thereof, e.g. polysorbates, sorbitan fatty acid esters or glycyrrhizin
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P1/00—Drugs for disorders of the alimentary tract or the digestive system
- A61P1/16—Drugs for disorders of the alimentary tract or the digestive system for liver or gallbladder disorders, e.g. hepatoprotective agents, cholagogues, litholytics
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P17/00—Drugs for dermatological disorders
- A61P17/02—Drugs for dermatological disorders for treating wounds, ulcers, burns, scars, keloids, or the like
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P25/00—Drugs for disorders of the nervous system
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P25/00—Drugs for disorders of the nervous system
- A61P25/28—Drugs for disorders of the nervous system for treating neurodegenerative disorders of the central nervous system, e.g. nootropic agents, cognition enhancers, drugs for treating Alzheimer's disease or other forms of dementia
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P3/00—Drugs for disorders of the metabolism
- A61P3/02—Nutrients, e.g. vitamins, minerals
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P3/00—Drugs for disorders of the metabolism
- A61P3/08—Drugs for disorders of the metabolism for glucose homeostasis
- A61P3/10—Drugs for disorders of the metabolism for glucose homeostasis for hyperglycaemia, e.g. antidiabetics
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P31/00—Antiinfectives, i.e. antibiotics, antiseptics, chemotherapeutics
- A61P31/04—Antibacterial agents
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P35/00—Antineoplastic agents
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P39/00—General protective or antinoxious agents
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P39/00—General protective or antinoxious agents
- A61P39/02—Antidotes
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P43/00—Drugs for specific purposes, not provided for in groups A61P1/00-A61P41/00
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P7/00—Drugs for disorders of the blood or the extracellular fluid
- A61P7/02—Antithrombotic agents; Anticoagulants; Platelet aggregation inhibitors
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P9/00—Drugs for disorders of the cardiovascular system
- A61P9/10—Drugs for disorders of the cardiovascular system for treating ischaemic or atherosclerotic diseases, e.g. antianginal drugs, coronary vasodilators, drugs for myocardial infarction, retinopathy, cerebrovascula insufficiency, renal arteriosclerosis
Definitions
- Ringers lactate solutions etc. All these have been used, as basic fluids when patient needed administration of therapeutic fluids and other ingredients. In certain special circumstances, fluids like
- the body is unable to utilise, the energy substrate and electrolytes available, in view of high levels of catacholamines, along with qualitative and quantitative deficiency of insulin, and disturbed hormonal levels.
- the fluids needed are broadly categorised for use to suit the various situations. They are to be in packing of 500 ml for calculations .
- the following abbreviations are used to denote the fluid and their possible place of use.
- G - Gangal N - Non diabetic, D - Diabetic, E - Electrolytes, H - Heparin, V - Vitamins, I - Insulin, where supplemental Potassium is added they are marked as K.
- Dext - Dextrose Dextrose.
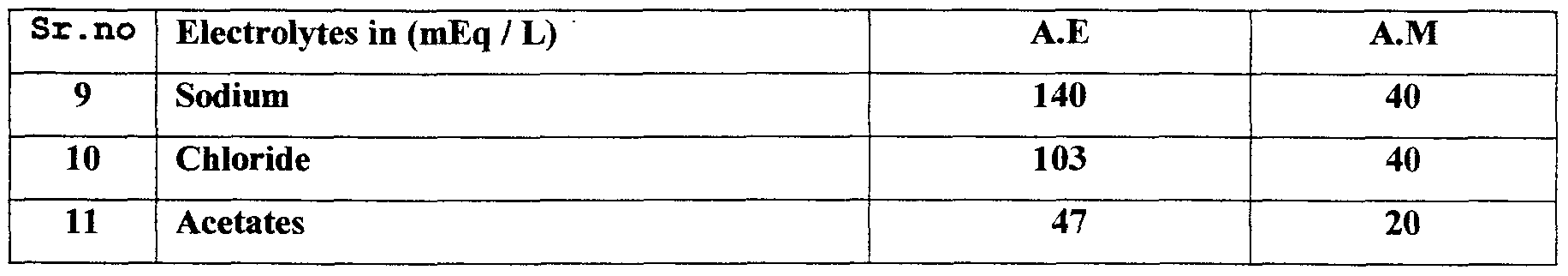
- A.E Actual electrolytes to replace the Extra cellular electrolytes
- A.M Actual electrolytes needed in Maintenance solutions as are presently available, to be taken as 500 ml packing for values of their contents.
- the fluids needed to treat patients who are said to be non-diabetic, as are known to day, are called GN 1 containing the ingredients as stated in the abbreviated text form in the columns
- the GD 9 solutions are supplemented with Electrolytes and Vitamins to gain GD 14 - EV solutions.
- the GD 9 group of fluids is supplemented with Heparin and Vitamins. They provide GD 15 - H V group of solutions.
- the GD 9 combination fluids are supplemented with Electrolytes, Heparin and Vitamins. These fluids form the GD 16 - EHV. Group of solutions
- the tables below provide information about A.E, the Actual electrolytes in Extra cellular replacement and A.M, Actual electrolytes in Maintenance solutions as are available.
- the values expressed in the table against each item is the actual content of the ingredients in 100 ml of each fluids as are in use today.
- Oil Soluble Vitamins A, D, E, are made water soluble to be used here.
- the formulations consist of the following and other ingredients mentioned in the above clarifications and in the text.
- Water is the main and the essential natural component of the body, and is also the main vehicle / media through which the body's circulation is maintained. This makes it possible the essentials of life and function are made available from the body to the cell level and receives back the wastes from the cell level to the organ to be eliminated out of the body. It helps to mentain physiological state in and around the body, congenial to sustain normal life.
- the volume of the water needed by the body varies with the functional level, the problems faced and the environment. Accordingly the volume of fluid needed to be infused, to balance the need and the output and also to maintain an effective circulation, are required into consideration to asses.
- Dextrose A major source of energy out of all other essential energy substrates. A must for living and sustaining life.
- Dextrose in the form of 5% as an isotonic solution has been the main stay of intravenous infusion in all forms of conditions where infusion forms the treatment as well as a resuscitative measure. In states of stress, shock, and critical conditions, this isotonic solution is now understood to have hypotonic solution effect.
- This type of fluids are likely to cause enormous harmful effects.
- the fluids we use in hypertonic form enable us to provide needed higher calories along with insulin, and also avoid effects of hypotonic solutions.
- it forms the main source of energy to be supplemented by amino acids, and emulsified fat solutions. This helps to meet the needs of essential amino acids, and also fats; along with it enables to prevent the bad and rebound effects of prolonged use of carbohydrates alone.
- Na Sodium chloride forms a major and a main electrolyte. It is also called the second pump to maintain the circulation.
- Nacl is an isotonic solution, commonly used as such or in combination with Dextrose solutions. It is one of the main electrolyte, the body needs. It is mainly an extra cellular electrolyte with a small intracellular component, having a reciprocal gradient relation with K ion responding to any effective agents. The intake and output needs to be balanced. The body contains a large amount, nearing around 5000 mEq of Na, with an average need of, 80 - 100, mEq / Day. This is mostly lost in the urine. In the normal individual, on one hand there is a large reserve, on the other hand there is an excellent mechanism to conserve it by normal functioning kidney. This can result in bringing urinary loss of Na to near zero, in the absence of intake ofNa. Still large fluctuations do occur with resultant serious problems. Hence it needs to be managed properly to retain homeostasis
- Ca Calcium similarly forms a major cation component of the body, having role to play in homeostasis, membrane potentials, inter cellular junctions and cell functions.
- Calcium forms around 2% of the bodyweight. Ninety nine % of it is in the form of bone. Part of it is constantly changing to contribute to the extra-cellular component. This is under the influence of simple ion exchange. Paratharmone, Vit D, Corticosteroids cause disturbances in Calcium content.
- any variations in the extra-cellular component affect the homeostasis. This in turn results in deranged cellular functions.
- Average Indian diet may contain as low as 0.2 Gms as against the daily need of 0.5 to 0.6 gm on an average. The needs are higher in later terms of pregnancy and lactation, thus needing up to around 1 gram of Calcium a day.
- Intracellular Ca regulates cell metabolism, it's function and inter junction permeability.
- Ca is an important component of the cell membrane and competes with Na. Raised Ca, in blood increases the threshold for contraction and low Ca leads to tetany.
- Ca also plays role in coagulation of blood, coupling mechanism between excitation and contraction of muscles, and secretary mechanism of Insulin.
- K Potassium mainly an intracellular electrolyte having a significant role to play in cell membrane potentials and cell functions.
- Potassium chloride KC1 solution is available for intravenous use, needs to be diluted considerably to use. Normally there is about 3500 mEq of Potassium in the body, of which 98%is intracellular and hardly 2% is in extra-cellular compartment. This small part of the Potassium plays a major role in the functioning of the smooth, skeletal, and cardiac muscles. As a result any variations that occur in its level can certainly cause considerable problems.
- electrolytes As are included in the A.E, extra cellular electrolyte replacement component and A.M, electrolyte maintenance replacement component solutions are also essential to the body's function and are required to be maintained in balance.
- Insulin Insulin is an essential hormone required in the process of metabolism of carbohydrates, proteins, and fats. Involving almost all organs in the body. It is now fairly well established, in clinical states needing intravenous infusions, there is qualitative and quantitative deficiency of insulin.
- Heparin It is a naturally occurring substance, not occurring in blood to a measurable extent, prevents blood coagulation. It plays significant role in cases of trauma induced on any account. Blood coagulation is essentially a series of enzymatic reactions involving a number of plasma proteins. Many of these are present only in traces. Within an intact healthy circulatory system these enzymes, for the most part, are in an inactive form, when the blood is shed on wettable surfaces, a series of auto catalytic enzymatic reaction is initiated, this is a process of more complex and confused in results, interpretations, and opinions.
- Vitamins Fat and water-soluble vitamins are organic compounds, forming as components or cofactors for enzymes concerned with metabolism of carbohydrates, fats, and proteins.
- Vitamin A -Daily dose of around 10,000 to 20,000 I.U in the form of made as water soluble could be essential to maintain the integrity of the epithelial cells, synthesis of glucocortcoids, cholesterol, and for somatic growth, upper respiratory tract epithelium.
- Vitamin Bi It is mainly concerned with Carbohydrate metabolism. IV / IM administration of large doses may cause anaphylactic shock and interfere with action of other B vitamins.
- B 3 - Nicotinic acid is required in Protein metabolism, necessary in respiration Be - Indirectly affects carbohydrate metabolism, directly the catacholamines, gamma-amino butyric acid, etc., B 7 - is involved in fundamental biological reactions.
- B ⁇ is necessary in maintenance of function of the nervous system and conduction mechanism.
- C - Concerned with cellular respiration / oxidation-reduction reactions, and formation of collagen, intercellular matrix development of cartilage, wound healing, carbohydrate metabolism.
- Vitamin D - Facilitates interdepartmental metabolism between vitamins, and maintains Ca. homeostasis. It is quite important in the acute conditions, like mobilisation of insulin, coupling mechanism of excitation and contraction mechanism of muscles. These have some relevance to diabetes normal organ function and essential hypertension.
- E - Alpha Tocopheryl Acetate is said to be essential to maintain integrity and stability of biological membranes, normal structure and function of the nervous system and is mostly used on an empirical base. It is known to be antioxidant.
- Choline Chloride is akin to B group of Vitamins but synthesised in the human body. It plays an important role as a structural component of tissues and in biological methilation reactions. It is a precursor of acetylcholine, and some hormones. Its biogenesis is universal in nature. It helps to spare amino acids in protein synthesis.
- DL - Methionine is sulphur containing aminoacid and inseparable dietary component. Methionine has lipotropic action, and is considered as useful adjutant of treatment of liver diseases. It enhances synthesis of glutathione, necessary for detoxification of toxic metabolites of paracetamol. Average needs are around 200 to 500 mgm.
- Inosital - occurs in nearly all plant, and animal cell, suggesting it to be an essential cell constituent. It is found to be a constituent of an amylase enzyme system. To a certain extent it is synthesised by the intestinal flora. It possesses weak lipotropic activity prevents fatty liver. It could be beneficial in diabetic neuropathy.
Landscapes
- Health & Medical Sciences (AREA)
- Life Sciences & Earth Sciences (AREA)
- Chemical & Material Sciences (AREA)
- Pharmacology & Pharmacy (AREA)
- Veterinary Medicine (AREA)
- Public Health (AREA)
- General Health & Medical Sciences (AREA)
- Medicinal Chemistry (AREA)
- Animal Behavior & Ethology (AREA)
- Epidemiology (AREA)
- General Chemical & Material Sciences (AREA)
- Chemical Kinetics & Catalysis (AREA)
- Engineering & Computer Science (AREA)
- Nuclear Medicine, Radiotherapy & Molecular Imaging (AREA)
- Bioinformatics & Cheminformatics (AREA)
- Organic Chemistry (AREA)
- Diabetes (AREA)
- Inorganic Chemistry (AREA)
- Molecular Biology (AREA)
- Biomedical Technology (AREA)
- Gastroenterology & Hepatology (AREA)
- Hematology (AREA)
- Neurology (AREA)
- Neurosurgery (AREA)
- Dermatology (AREA)
- Toxicology (AREA)
- Immunology (AREA)
- Obesity (AREA)
- Endocrinology (AREA)
- Proteomics, Peptides & Aminoacids (AREA)
- Communicable Diseases (AREA)
- Psychiatry (AREA)
- Nutrition Science (AREA)
- Hospice & Palliative Care (AREA)
- Oncology (AREA)
- Biochemistry (AREA)
- Heart & Thoracic Surgery (AREA)
- Oil, Petroleum & Natural Gas (AREA)
- Cardiology (AREA)
- Zoology (AREA)
Abstract
Description
Claims
Priority Applications (3)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| AU30489/01A AU779798C (en) | 1999-11-15 | 2000-11-10 | Dextrose and insulin fluid formulation for intravenous infusion |
| JP2001537936A JP2003514014A (en) | 1999-11-15 | 2000-11-10 | Glucose and insulin containing liquid formulations for intravenous infusion |
| EP00990877A EP1231907A2 (en) | 1999-11-15 | 2000-11-10 | Dextrose and insulin fluid formulation for intravenous infusion |
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| IN1106MA1999 | 1999-11-15 | ||
| IN1106/MAS/99 | 1999-11-15 |
Publications (2)
| Publication Number | Publication Date |
|---|---|
| WO2001035943A2 true WO2001035943A2 (en) | 2001-05-25 |
| WO2001035943A3 WO2001035943A3 (en) | 2002-03-21 |
Family
ID=11096775
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/IN2000/000110 WO2001035943A2 (en) | 1999-11-15 | 2000-11-10 | Dextrose and insulin fluid formulation for intravenous infusion |
Country Status (7)
| Country | Link |
|---|---|
| EP (1) | EP1231907A2 (en) |
| JP (1) | JP2003514014A (en) |
| CN (1) | CN1284522C (en) |
| AU (1) | AU779798C (en) |
| RU (1) | RU2300367C2 (en) |
| WO (1) | WO2001035943A2 (en) |
| ZA (1) | ZA200204895B (en) |
Cited By (6)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JP2004002429A (en) * | 2002-05-29 | 2004-01-08 | Insignion Holdings Ltd | Composition and therapeutic application of the same |
| WO2007072147A2 (en) * | 2005-12-19 | 2007-06-28 | Ernst, Johanna, Catarina | Composition for diagnosing and treating circulatory system diseases |
| US9901623B2 (en) | 2015-08-27 | 2018-02-27 | Eli Lilly And Company | Rapid-acting insulin compositions |
| US10646551B2 (en) | 2012-11-13 | 2020-05-12 | Adocia | Rapid-acting insulin formulation comprising a substituted anionic compound |
| US11123406B2 (en) | 2014-12-16 | 2021-09-21 | Eli Lilly And Company | Rapid-acting insulin compositions |
| US11207384B2 (en) | 2017-06-01 | 2021-12-28 | Eli Lilly And Company | Rapid-acting insulin compositions |
Families Citing this family (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| RU2546286C2 (en) * | 2013-06-07 | 2015-04-10 | Федеральное государственное бюджетное научное учреждение "Всероссийский научно-исследовательский институт крахмалопродуктов" | Pharmaceutical substance and method of obtaining thereof |
Citations (3)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO1982003773A1 (en) * | 1981-04-27 | 1982-11-11 | Baxter Travenol Lab | Dialysis solution containing glucose,amino acids & insulin |
| WO1991018610A1 (en) * | 1990-05-28 | 1991-12-12 | Olle Ljungqvist Medical Ab | New use of glucose and a new solution of glucose |
| WO1995005833A1 (en) * | 1993-08-24 | 1995-03-02 | Albert Fay Hill | Pharmaceutical composition for immunoenhancement therapy |
-
2000
- 2000-11-10 EP EP00990877A patent/EP1231907A2/en not_active Withdrawn
- 2000-11-10 WO PCT/IN2000/000110 patent/WO2001035943A2/en active IP Right Grant
- 2000-11-10 AU AU30489/01A patent/AU779798C/en not_active Ceased
- 2000-11-10 RU RU2002116377/15A patent/RU2300367C2/en not_active IP Right Cessation
- 2000-11-10 JP JP2001537936A patent/JP2003514014A/en active Pending
- 2000-11-10 CN CNB008157014A patent/CN1284522C/en not_active Expired - Fee Related
-
2002
- 2002-06-14 ZA ZA200204895A patent/ZA200204895B/en unknown
Patent Citations (3)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO1982003773A1 (en) * | 1981-04-27 | 1982-11-11 | Baxter Travenol Lab | Dialysis solution containing glucose,amino acids & insulin |
| WO1991018610A1 (en) * | 1990-05-28 | 1991-12-12 | Olle Ljungqvist Medical Ab | New use of glucose and a new solution of glucose |
| WO1995005833A1 (en) * | 1993-08-24 | 1995-03-02 | Albert Fay Hill | Pharmaceutical composition for immunoenhancement therapy |
Non-Patent Citations (4)
| Title |
|---|
| DATABASE EMBASE [Online] ELSEVIER SCIENCE PUBLISHERS, AMSTERDAM, NL; HASLAUER F. ET AL: "[The effect of an infusion of glucose- insulin -potassium and of heparin on the concentration of plasma free fatty acids and blood glucose]. DIE WIRKUNG EINER INFUSION VON GLUKOSE- INSULIN -KALIUM UND VON HEPARIN AUF DIE KONZENTRATION DER FREIEN FETTSAUREN DES PLASMAS UND DER BLUTGLUKOSE." retrieved from STN Database accession no. 83253537 XP002171110 & WIENER KLINISCHE WOCHENSCHRIFT, (1983) 95/17 (618-621). CODEN: WKWOAO, * |
| DATABASE EMBASE [Online] ELSEVIER SCIENCE PUBLISHERS, AMSTERDAM, NL; MARCUARD S.P. ET AL: "Availability of insulin from total parenteral nutrition solutions." retrieved from STN Database accession no. 90165762 XP002171111 & JOURNAL OF PARENTERAL AND ENTERAL NUTRITION, (1990) 14/3 (262-264). , * |
| DATABASE MEDLINE [Online] US NATIONAL LIBRARY OF MEDICINE (NLM), BETHESDA, MD, US; WEBER S S ET AL: "Availability of insulin from parenteral nutrient solutions." retrieved from STN Database accession no. 77179246 XP002171112 & AMERICAN JOURNAL OF HOSPITAL PHARMACY, (1977 APR) 34 (4) 353-7. , * |
| SHIZGAL H M ET AL: "INSULIN AND EFFICACY OF TOTAL PARENTERAL NUTRITION" AMERICAN JOURNAL OF CLINICAL NUTRITION,BETHESDA,MD,US, vol. 50, 1989, pages 1355-1363, XP000569039 ISSN: 0002-9165 * |
Cited By (13)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JP2004002429A (en) * | 2002-05-29 | 2004-01-08 | Insignion Holdings Ltd | Composition and therapeutic application of the same |
| GB2427824A (en) * | 2002-05-29 | 2007-01-10 | Insignion Holdings Ltd | Treatment of proliferative or viral conditions |
| GB2392618B (en) * | 2002-05-29 | 2007-02-21 | Insignion Holdings Ltd | Compositions for therapeutic use |
| WO2007072147A2 (en) * | 2005-12-19 | 2007-06-28 | Ernst, Johanna, Catarina | Composition for diagnosing and treating circulatory system diseases |
| WO2007072147A3 (en) * | 2005-12-19 | 2007-11-01 | Ernst Johanna Catarina | Composition for diagnosing and treating circulatory system diseases |
| US10646551B2 (en) | 2012-11-13 | 2020-05-12 | Adocia | Rapid-acting insulin formulation comprising a substituted anionic compound |
| US10881716B2 (en) | 2012-11-13 | 2021-01-05 | Adocia | Rapid-acting insulin formulation comprising a substituted anionic compound |
| US11324808B2 (en) | 2012-11-13 | 2022-05-10 | Adocia | Rapid-acting insulin formulation comprising a substituted anionic compound |
| US11123406B2 (en) | 2014-12-16 | 2021-09-21 | Eli Lilly And Company | Rapid-acting insulin compositions |
| US11872266B2 (en) | 2014-12-16 | 2024-01-16 | Eli Lilly And Company | Rapid-acting insulin compositions |
| US9901623B2 (en) | 2015-08-27 | 2018-02-27 | Eli Lilly And Company | Rapid-acting insulin compositions |
| US10925931B2 (en) | 2015-08-27 | 2021-02-23 | Eli Lilly And Company | Rapid-acting insulin compositions |
| US11207384B2 (en) | 2017-06-01 | 2021-12-28 | Eli Lilly And Company | Rapid-acting insulin compositions |
Also Published As
| Publication number | Publication date |
|---|---|
| RU2002116377A (en) | 2004-01-27 |
| CN1390118A (en) | 2003-01-08 |
| RU2300367C2 (en) | 2007-06-10 |
| EP1231907A2 (en) | 2002-08-21 |
| AU779798B2 (en) | 2005-02-10 |
| JP2003514014A (en) | 2003-04-15 |
| AU3048901A (en) | 2001-05-30 |
| WO2001035943A3 (en) | 2002-03-21 |
| CN1284522C (en) | 2006-11-15 |
| ZA200204895B (en) | 2003-02-12 |
| AU779798C (en) | 2005-11-17 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| Musso | Magnesium metabolism in health and disease | |
| Marinella | The refeeding syndrome and hypophosphatemia | |
| Levine et al. | Hypocalcemic heart failure | |
| US5032608A (en) | Method and substrate composition for treating atherosclerosis | |
| Velez et al. | Severe acute metabolic acidosis (acute beriberi): an avoidable complication of total parenteral nutrition | |
| Kraft | Phosphorus and calcium: a review for the adult nutrition support clinician | |
| Clark et al. | Treatment of hypophosphatemia in patients receiving specialized nutrition support using a graduated dosing scheme: results from a prospective clinical trial | |
| AU2022204450A1 (en) | Uses of oxygenated cholesterol sulfates (OCS) | |
| Rhoda et al. | Fluid and electrolyte management: putting a plan in motion | |
| Young | Role of potassium in preventive cardiovascular medicine | |
| AU779798C (en) | Dextrose and insulin fluid formulation for intravenous infusion | |
| McCarthy | A practical approach to the management of patients with chronic renal failure | |
| Blumenkrantz et al. | Managing the nutritional concerns of the patient undergoing peritoneal dialysis | |
| Sacks et al. | Observations of hypophosphatemia and its management in nutrition support | |
| AU8033487A (en) | Method and substrate composition for treating atherosclerosis | |
| Marsano et al. | Effects of alcohol on electrolytes and minerals | |
| US6372264B1 (en) | Method of reducing calcified arterial plaque buildup and cellular malfunction and for balancing ionic calcium | |
| Lim et al. | Managing hypocalcemia in massive blood transfusion | |
| VANDER WOUDE et al. | Addition of branched-chain amino acids to parenteral nutrition of stressed critically ill patients | |
| Schneir et al. | Lithium Toxicity from the Addition of an ACE Inhibitor with an Unexpected Type I Brugada Pattern ECG: Case Files of the Medical Toxicology Fellowship at the University of California, San Diego | |
| Gooch | Identifying acid-base and electrolyte imbalances | |
| Podd | Hyperphosphatemia: understanding the role of phosphate metabolism | |
| Parker | Calcium Alkali Syndrome Treated With Hemodialysis | |
| Whang | Medical and health aspects of potassium | |
| Galica | Parenteral nutrition |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| AK | Designated states |
Kind code of ref document: A2 Designated state(s): AE AG AL AM AT AU AZ BA BB BG BR BY BZ CA CH CN CR CU CZ DE DK DM DZ EE ES FI GB GD GE GH GM HR HU ID IL IN IS JP KE KG KP KR KZ LC LK LR LS LT LU LV MA MD MG MK MN MW MX MZ NO NZ PL PT RO RU SD SE SG SI SK SL TJ TM TR TT TZ UA UG US UZ VN YU ZA ZW |
|
| AL | Designated countries for regional patents |
Kind code of ref document: A2 Designated state(s): GH GM KE LS MW MZ SD SL SZ TZ UG ZW AM AZ BY KG KZ MD RU TJ TM AT BE CH CY DE DK ES FI FR GB GR IE IT LU MC NL PT SE TR BF BJ CF CG CI CM GA GN GW ML MR NE SN TD TG |
|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application | ||
| DFPE | Request for preliminary examination filed prior to expiration of 19th month from priority date (pct application filed before 20040101) | ||
| AK | Designated states |
Kind code of ref document: A3 Designated state(s): AE AG AL AM AT AU AZ BA BB BG BR BY BZ CA CH CN CR CU CZ DE DK DM DZ EE ES FI GB GD GE GH GM HR HU ID IL IN IS JP KE KG KP KR KZ LC LK LR LS LT LU LV MA MD MG MK MN MW MX MZ NO NZ PL PT RO RU SD SE SG SI SK SL TJ TM TR TT TZ UA UG US UZ VN YU ZA ZW |
|
| AL | Designated countries for regional patents |
Kind code of ref document: A3 Designated state(s): GH GM KE LS MW MZ SD SL SZ TZ UG ZW AM AZ BY KG KZ MD RU TJ TM AT BE CH CY DE DK ES FI FR GB GR IE IT LU MC NL PT SE TR BF BJ CF CG CI CM GA GN GW ML MR NE SN TD TG |
|
| ENP | Entry into the national phase in: |
Ref country code: JP Ref document number: 2001 537936 Kind code of ref document: A Format of ref document f/p: F |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 008157014 Country of ref document: CN |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 2002/04895 Country of ref document: ZA Ref document number: 200204895 Country of ref document: ZA Ref document number: 30489/01 Country of ref document: AU |
|
| ENP | Entry into the national phase in: |
Ref country code: RU Ref document number: 2002 2002116377 Kind code of ref document: A Format of ref document f/p: F |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 2000990877 Country of ref document: EP |
|
| WWP | Wipo information: published in national office |
Ref document number: 2000990877 Country of ref document: EP |
|
| REG | Reference to national code |
Ref country code: DE Ref legal event code: 8642 |
|
| WWG | Wipo information: grant in national office |
Ref document number: 30489/01 Country of ref document: AU |