WO1993000085A1 - Stabilized retinoid-containing skin care compositions - Google Patents
Stabilized retinoid-containing skin care compositions Download PDFInfo
- Publication number
- WO1993000085A1 WO1993000085A1 PCT/US1991/004471 US9104471W WO9300085A1 WO 1993000085 A1 WO1993000085 A1 WO 1993000085A1 US 9104471 W US9104471 W US 9104471W WO 9300085 A1 WO9300085 A1 WO 9300085A1
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- skin care
- care composition
- oil
- water
- acid
- Prior art date
Links
Classifications
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K47/00—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient
- A61K47/06—Organic compounds, e.g. natural or synthetic hydrocarbons, polyolefins, mineral oil, petrolatum or ozokerite
- A61K47/16—Organic compounds, e.g. natural or synthetic hydrocarbons, polyolefins, mineral oil, petrolatum or ozokerite containing nitrogen, e.g. nitro-, nitroso-, azo-compounds, nitriles, cyanates
- A61K47/18—Amines; Amides; Ureas; Quaternary ammonium compounds; Amino acids; Oligopeptides having up to five amino acids
- A61K47/183—Amino acids, e.g. glycine, EDTA or aspartame
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K31/00—Medicinal preparations containing organic active ingredients
- A61K31/045—Hydroxy compounds, e.g. alcohols; Salts thereof, e.g. alcoholates
- A61K31/07—Retinol compounds, e.g. vitamin A
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K31/00—Medicinal preparations containing organic active ingredients
- A61K31/11—Aldehydes
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K31/00—Medicinal preparations containing organic active ingredients
- A61K31/21—Esters, e.g. nitroglycerine, selenocyanates
- A61K31/215—Esters, e.g. nitroglycerine, selenocyanates of carboxylic acids
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K31/00—Medicinal preparations containing organic active ingredients
- A61K31/21—Esters, e.g. nitroglycerine, selenocyanates
- A61K31/215—Esters, e.g. nitroglycerine, selenocyanates of carboxylic acids
- A61K31/22—Esters, e.g. nitroglycerine, selenocyanates of carboxylic acids of acyclic acids, e.g. pravastatin
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K31/00—Medicinal preparations containing organic active ingredients
- A61K31/21—Esters, e.g. nitroglycerine, selenocyanates
- A61K31/215—Esters, e.g. nitroglycerine, selenocyanates of carboxylic acids
- A61K31/22—Esters, e.g. nitroglycerine, selenocyanates of carboxylic acids of acyclic acids, e.g. pravastatin
- A61K31/23—Esters, e.g. nitroglycerine, selenocyanates of carboxylic acids of acyclic acids, e.g. pravastatin of acids having a carboxyl group bound to a chain of seven or more carbon atoms
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K47/00—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient
- A61K47/06—Organic compounds, e.g. natural or synthetic hydrocarbons, polyolefins, mineral oil, petrolatum or ozokerite
- A61K47/08—Organic compounds, e.g. natural or synthetic hydrocarbons, polyolefins, mineral oil, petrolatum or ozokerite containing oxygen, e.g. ethers, acetals, ketones, quinones, aldehydes, peroxides
- A61K47/10—Alcohols; Phenols; Salts thereof, e.g. glycerol; Polyethylene glycols [PEG]; Poloxamers; PEG/POE alkyl ethers
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K47/00—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient
- A61K47/06—Organic compounds, e.g. natural or synthetic hydrocarbons, polyolefins, mineral oil, petrolatum or ozokerite
- A61K47/08—Organic compounds, e.g. natural or synthetic hydrocarbons, polyolefins, mineral oil, petrolatum or ozokerite containing oxygen, e.g. ethers, acetals, ketones, quinones, aldehydes, peroxides
- A61K47/12—Carboxylic acids; Salts or anhydrides thereof
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K47/00—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient
- A61K47/06—Organic compounds, e.g. natural or synthetic hydrocarbons, polyolefins, mineral oil, petrolatum or ozokerite
- A61K47/20—Organic compounds, e.g. natural or synthetic hydrocarbons, polyolefins, mineral oil, petrolatum or ozokerite containing sulfur, e.g. dimethyl sulfoxide [DMSO], docusate, sodium lauryl sulfate or aminosulfonic acids
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K8/00—Cosmetics or similar toiletry preparations
- A61K8/02—Cosmetics or similar toiletry preparations characterised by special physical form
- A61K8/04—Dispersions; Emulsions
- A61K8/06—Emulsions
- A61K8/064—Water-in-oil emulsions, e.g. Water-in-silicone emulsions
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K8/00—Cosmetics or similar toiletry preparations
- A61K8/18—Cosmetics or similar toiletry preparations characterised by the composition
- A61K8/30—Cosmetics or similar toiletry preparations characterised by the composition containing organic compounds
- A61K8/40—Cosmetics or similar toiletry preparations characterised by the composition containing organic compounds containing nitrogen
- A61K8/44—Aminocarboxylic acids or derivatives thereof, e.g. aminocarboxylic acids containing sulfur; Salts; Esters or N-acylated derivatives thereof
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K8/00—Cosmetics or similar toiletry preparations
- A61K8/18—Cosmetics or similar toiletry preparations characterised by the composition
- A61K8/30—Cosmetics or similar toiletry preparations characterised by the composition containing organic compounds
- A61K8/67—Vitamins
- A61K8/671—Vitamin A; Derivatives thereof, e.g. ester of vitamin A acid, ester of retinol, retinol, retinal
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K9/00—Medicinal preparations characterised by special physical form
- A61K9/0012—Galenical forms characterised by the site of application
- A61K9/0014—Skin, i.e. galenical aspects of topical compositions
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P17/00—Drugs for dermatological disorders
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61Q—SPECIFIC USE OF COSMETICS OR SIMILAR TOILETRY PREPARATIONS
- A61Q17/00—Barrier preparations; Preparations brought into direct contact with the skin for affording protection against external influences, e.g. sunlight, X-rays or other harmful rays, corrosive materials, bacteria or insect stings
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61Q—SPECIFIC USE OF COSMETICS OR SIMILAR TOILETRY PREPARATIONS
- A61Q19/00—Preparations for care of the skin
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K2800/00—Properties of cosmetic compositions or active ingredients thereof or formulation aids used therein and process related aspects
- A61K2800/40—Chemical, physico-chemical or functional or structural properties of particular ingredients
- A61K2800/51—Chelating agents
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K2800/00—Properties of cosmetic compositions or active ingredients thereof or formulation aids used therein and process related aspects
- A61K2800/40—Chemical, physico-chemical or functional or structural properties of particular ingredients
- A61K2800/52—Stabilizers
- A61K2800/522—Antioxidants; Radical scavengers
Definitions
- This invention relates to skin care compositions containing retinoids which generally improve the quality of the skin, particularly human facial skin. More particularly, the present invention relates to chemically stable skin care compositions comprising a water-in-oil emulsion and certain retinoids and to methods for making such compositions .
- Retinoic acid also known as Vitamin A acid or tretinoin
- Vitamin A acid or tretinoin is well-known for the treatment of such skin conditions as acne and products containing retinoic acid are commercially available in various forms from the Dermatological Division of Ortho Pharmaceutical Corporation.
- Such products include Retin A* creams, an oil-in-water emulsion of retinoic acid containing as an oil-soluble antioxidant, butylated hydroxytoluene (BHT); Retin A* liquid, a solution of retinoic acid in a polyethylene glycol/ethanol solvent employing BHT as an antioxidant; and Retin A* gel, which comprises retinoic acid in a gel vehicle comprising ethyl alcohol as the solvent, hydroxypropyl cellulose as the thickener or gelling agent and BHT as an antioxidant.
- BHT butylated hydroxytoluene
- Retin A* liquid a solution of retinoic acid in a polyethylene glycol/ethanol solvent employing BHT as an antioxidant
- Retin A* gel which comprises retinoic acid in a gel vehicle comprising ethyl alcohol as the solvent, hydroxypropyl cellulose as the thickener or gelling agent and BHT as an antioxidant.
- retinoids such as, for example, retinol (Vitamin A alcohol), retinal (Vitamin A aldehyde) and retinyl esters such as retinyl acetate and retinyl palmitate would be preferred over retinoic acid.
- a preferred form is retinol. This is because retinol is an endogenous compound naturally occurring in the human body and essential for good growth, differentiation of epithelial tissues and reproduction. Retinol is also preferred because it has a much larger safety margin than other retinoids such as retinoic acid.
- retinol is stored in the human body largely in an inactive ester form, e.g. retinyl palmitate and, to some extent, retinyl acetate.
- the aldehyde, retinal also a preferred form, is an active metabolite of retinol and is needed for visual function. Accordingly, attention has turned toward formulating skin care compositions which contain these preferred, naturally occurring retinoids.
- oil-in-water emulsions have been preferred in that, as compared to water-in-oil emulsions for example, they are non-occlusive, non-greasy, compatible with other such emulsion products, easy to remove from the skin and are regarded as more aesthetically pleasing as well as being more economical to manufacture.
- chemical stability of the active ingredient it has been experienced that the retinoic acid in the oil phase is, in the main, well protected by including in such oil phase an oil soluble antioxidant.
- the aforementioned Retin A* cream is an oil-in-water emulsion containing retinoic acid and BHT, an oil-soluble antioxidant.
- an oil-in-water emulsion of retinoic acid which may include an oil-soluble antioxidant such as BHT or dl- ⁇ -tocopherol and a chelating agent e.g. ethylenediaminetetraacetic acid (EDTA).
- EDTA ethylenediaminetetraacetic acid
- a tanning composition is described which may include, among other ingredients Vitamin A in an oil-in-water emulsion containing Vitamin E and citric acid.
- Patent 4,247,547 still another form of a retinoic acid containing composition, namely a gel, is disclosed and is protected by an antioxidant selected from the group consisting of butylated hydroxytoluene, butylated hydroxyanisole (BHA), ascorbic acid (Vitamin C), propyl gallate, and ⁇ -tocopherol (Vitamin E).
- BHA butylated hydroxytoluene
- BHA butylated hydroxyanisole
- Ascorbic acid Vitamin C
- propyl gallate propyl gallate
- ⁇ -tocopherol Vitamin E
- retinoids including for example retinol, retinal and retinyl esters such as retinyl acetate and retinyl palmitate, and these unsurprisingly emulate the formulas of the commercial retinoic acid compositions i.e. are oil-in-water emulsions protected by oil-soluble antioxidants.
- retinoids such as retinol, retinyl acetate and retinyl palmitate are formulated in water-in-oil emulsions.
- a stable composition comprising retinol, retinyl acetate and retinyl palmitate may consist of retinol in a water-in-oil emulsion wherein the emulsion further include two oil-soluble antioxidants, BHT and BHA.
- Avon Products, Inc. the assignee of U.S. 4,826,828, sells two skin care products called Bioadvance and Bioadvance 2000. Each of these products is supplied in two bottles, portions of which are mixed together just prior to use. The first bottle contains what is called a "skin lotion", while the second bottle contains what is called a "fortifier".
- the “skin lotion” is a water-in-oil emulsion having a number of ingredients which include water, emulsifiers, silicone and vegetable oils, preservatives, emollients and butylated hydroxytoluene (BHT).
- the "fortifier” is a solution which contains a number of ingredients including cyclomethicone (a silicone oil), denatured ethanol, an emulsifier (Polysorbate 20), retinol, retinyl acetate, retinyl palmitate, BHT and BHA.
- U.S. 4,720,353 to Bell discloses water-in-oil emulsion carriers for various medicaments and drugs intended for topical application to the skin.
- Water soluble, miscible or dispersible drugs may be incorporated into the aqueous phase of the emulsion.
- Oil-soluble, miscible or dispersible drugs may be incorporated into the oil phase.
- Drugs which may be incorporated into the emulsion include derivatives of retinoic acid.
- Ingredients which may optionally be added to the emulsion include a preservative such as methyl paraben, propyl paraben or imidazolidinyl urea or an antioxidant such as butylated hydroxyanisole and a water or oil soluble vitamin such as vitamin C, tocopherol linoleate and the like.
- a preservative such as methyl paraben, propyl paraben or imidazolidinyl urea or an antioxidant such as butylated hydroxyanisole and a water or oil soluble vitamin such as vitamin C, tocopherol linoleate and the like.
- EP 0 343 444 A2 to Siemer et al discloses cosmetic preparations based on retinyl palmitate.
- Example 3 discloses a night cream in the form of an water-in-oil type emulsion comprising retinyl palmitate and butylated hydroxyanisole (BHA).
- BHA butylated hydroxyanisole
- EP 0 330496 A2 to Batt is directed to skin treatment compositions comprising a topically acceptable base and an effective amount of at least one ester of retinol, said compositions being useful in the treatment of photoaged skin.
- Example 6 discloses a water-in-oil emulsion comprising Vitamin A propionate and BHT, an oil-soluble antioxidant.
- retinoids may be successfully stabilized against chemical degradation by incorporating them into water-in-oil emulsions comprising a specifically defined stabilizing system.
- stability satisfactory for a commercial product has been achievable for certain specific retinoids only by utilizing a specific form of emulsion, i.e. water-in-oil, and then, only by employing a specific stabilizing system.
- retinoids which can be stabilized against chemical degradation in accordance with the principles of the present invention are retinol (Vitamin A alcohol), retinal (Vitamin A aldehyde), retinyl acetate, retinyl palmitate and mixtures thereof.
- the "chemical stability” or “stability" of a retinoid is defined in terms of the percentage of the specified retinoid which is retained in its original chemical form after the composition has been stored for a specified period of time at a specified temperture.
- the original concentration of all-trans retinol in an absolute ethanol solution were 0.20% by weight and, after two (2) weeks storage at room temperature (21°C ⁇ 1°C), the concentration of all-trans retinol were 0.18% by weight, then the original solution of all-trans retinol in absolute ethanol would be characterized as having a chemical stability of retinol of 90% after two weeks storage at room temperature.
- a commercially usable composition should exhibit a stability of at least about 60% of the active retinoid (s) after 13 weeks storage at 40°C .
- such compositions further exhibit a stability of at least about 80% after 13 weeks storage at room temperature .
- a skin care composition comprising a water-in-oil emulsion and a retinoid selected from the group consisting of retinol , retinal , retinyl acetate , retinyl palmitate and mixtures thereof , said composition further comprising a stabilizing system selected from the group consisting of : a) a chelating agent and at least one oil-soluble antioxidant; b) a chelating agent and at least one water-soluble antioxidant; and c) antioxidant present in each of the oil and water phases of said emulsion; said composition retaining at least about 60% of said retinoids after 13 weeks storage at 40°C.
- the skin care composition includes one of the aforementioned retinoids. a chelating agent, a water-soluble antioxidant and an oil- soluble antioxidant. Further, in a preferred embodiment of the invention, the composition retains at least about 90% of such retinoids after 13 weeks storage at 21°C.
- the composition of the invention is in the form of a particular type of emulsion, namely water- in-oil.
- emulsion namely water- in-oil.
- the generally accepted concept of an emulsion applies i.e., an intimate mixture of two immiscible liquids which remains unseparated for an acceptable shelf life (is physically stable) at or about room temperature.
- shelf life is physically stable
- both phases initially tend to form droplets. Thereafter, when the agitation ceases, the droplets quickly coalesce, and the two liquids tend to separate.
- an emulsion may be formed and physically stabilized and the lifetime of the droplets in intimate mixture materially increased if a compound, referred to as an emulsifier, is added to the immiscible liquids.
- a compound referred to as an emulsifier
- an emulsifier usually only one phase persists in droplet form for a prolonged period of time, and this is referred to as the internal phase which is surrounded by an external phase.
- An oil-in-water emulsion is one in which the external phase (also called the continuous phase) comprises water and the internal phase (also called the discontinuous or disperse phase) comprises an oil.
- a water-in-oil emulsion is one in which the external phase comprises an oil and the internal phase comprises water.
- compositions such as the ones containing retinoic acid are oil-in-water emulsion systems.
- certain retinoid compounds in particular, retinol, retinal, and the retinyl esters tend to be chemically unstable, i.e. they degrade, either by way of oxidation or isomerization, and are, therefore, not available to perform in their desired manner. While this is not clearly understood, it is believed that this degradation occurs as a result of the rapid diffusion of oxygen through the external water phase to the internal oil phase containing the retinoid, and degradation of the retinoid then occurs. Since the diffusion of oxygen is greater in a water phase than an oil phase, an oil-in-water system is more prone to such degradation. Precisely why this occurs for certain selected retinoids and not for others, is not yet understood.
- the present invention has overcome these difficulties and instead, provides a water-in-oil emulsion composition containing at least one retinoid compound wherein the physical stability of the emulsion and the chemical stability of the active ingredients is excellent.
- the composition of this invention employs a chemical stabilizing system selected from the group consisting of a) a chelating agent and at least one oil-soluble antioxidant; b) a chelating agent and at least one water-soluble antioxidant; and c) antioxidant present in each of the oil and water phases of said emulsion;
- the water-soluble antioxidants which are useful in the compositions of the present invention include ascorbic acid, sodium sulfite, sodium metabisulfite, sodium bisulfite, sodium thiosulfite, sodium formaldehyde sulfoxylate, isoascorbic acid, thioglycerol, thiosorbitol, thiourea, thioglycolic acid, cysteine hydrochloride, 1,4-diazobicyclo-(2,2,2)-octane and mixtures thereof as well as any other known water-soluble antioxidant compatible with the other components of the compositions.
- the oil-soluble antioxidants which are useful in the compositions of the present invention include butylated hydroxytoluene (BHT), ascorbyl palmitate, butylated hydroxyanisole (BHA) , ⁇ -tocopherol, phenyl- ⁇ -naphthylamine, hydroquinone, propyl gallate, nordihydroguiaretic acid, and mixtures thereof as well as any other known oil-soluble antioxidant compatible with the other components of the compositions.
- BHT butylated hydroxytoluene
- BHA butylated hydroxyanisole
- ⁇ -tocopherol phenyl- ⁇ -naphthylamine
- hydroquinone propyl gallate
- nordihydroguiaretic acid and mixtures thereof as well as any other known oil-soluble antioxidant compatible with the other components of the compositions.
- the antioxidants should be utilized in a stabilizing effective amount and may range in total from about 0.001 to 5.0% based on the weight of the total
- the compositions include a chelating agent.
- the retinoid compounds of this invention are sensitive to metal ions and in particular to bi- and tri-valent cations and in certain instances, appear degrade rapidly in their presence.
- the chelating agent forms a complex with the metal ions thereby inactivating them and preventing them from affecting the retinoid compounds.
- Chelating agents which are useful in the compositions of the present invention include ethylenediamine tetraacetic acid (EDTA) and derivatives and salts thereof, dihydroxyethyl glycine, citric acid, tartaric acid, and mixtures thereof.
- the chelating agents should be utilized in a stabilizing effective amount and may range from about 0.01 to 2.0% based on the weight of the total composition, preferably from about 0.05 to 1.0%.
- the retinoid compounds which are useful in the compositions of the present invention consist of Vitamin A alcohol (retinol), Vitamin A aldehyde (retinal) and Vitamin A esters (retinyl acetate and retinyl palmitate). These retinoids are utilized in the compositions of the present invention in a therapeutically effective amount that may range from about 0.001 to 5.0% by weight of the total compositions, preferably from about 0.001 to 1.0%.
- the skin care compositions of the present invention comprising a water-in-oil emulsion can be in the format of cream or lotion formulations, as desired, by varying the relative quantities of the oil and water phases of the emulsion.
- the pH of the compositions should be in the range of from about 4 to about 9, and preferably from about 4 to about 7.
- Minerals oils, animal oils, vegetable oils and silicones have all been used in cosmetic creams and lotions of the emulsion type.
- other emollients and surface active agents have been incorporated in the emulsions, including glyceryl trioleate, acetylated sucrose distearate, sorbitan triolate, polyoxyethylene (1) monostearate, glycerol monooleate, sucrose distearate.
- Thickeners such as natural gums and synthetic polymers, as well as preservatives such as methylparaben, butyl paraben, propylparaben and phenoxyethanol, coloring agents and fragrances also are commonly included in such compositions.
- Other active ingredients such as sunscreen materials and antimicrobial materials may be utilized in the compositions of the present invention provided that they are physically and chemically compatible with the other components of the compositions.
- the essence of the present invention is not within the specific composition per se of the cream or lotion formulation, and any of the many formulations or compositions of the cream or lotion type currently utilized in skin care preparations can be employed provided that it is in a water-in-oil emulsion and is chemically compatible with the retinoid compounds.
- the ratio of the oil phase of the emulsion to the water phase can be from about 5:95 to 99:1.
- the actual ratio of the two phases will depend on the desired final product.
- the compositions of the present invention can be prepared by well-known mixing or blending procedures. Each phase of the emulsion is preferably separately prepared with all of the components contained in the appropriate phase except it is usually preferred to omit the retinoid compound.
- the emulsion is then formed normally by adding the water phase to the oil phase with agitation, and often the emulsion is cooled down when the retinoid compound is added.
- the portions be prepared under an oxygen depleted atmosphere such as a nitrogen or argon gas blanket. Commercially, it is envisioned that such oxygen depleted atmosphere may be obtained by operating under vacuum conditions and that the product be stored, prior to use, in blind-end containers, preferably aluminum tubes.
- retinol (Vitamin A alcohol) were prepared having the % w/w compositions set forth in Table 1. These emulsions were prepared according to the following procedure. The ingredients shown under the heading "Aqueous Phase Ingredients” were added to a first glass container equipped with a stainless steel stirrer and heated with stirring to 75-85°c under an argon gas blanket. The ingredients shown under the heading “Oil Phase Ingredients” were added to a second glass container equipped with a stainless steel stirrer and heated with stirring to about from 85° to 90°C under an argon gas blanket.
- the Retinoid Mixture was added with stirring.
- the emulsion was blanketed under argon gas and the temperature was maintained at about 50-53°C during the addition of the Retinoid Mixture.
- the emulsion was gradually cooled, with stirring and under an argon blanket, to room temperature (approximately 21°C).
- the finished emulsion was then transferred under argon gas blanketing to blind end aluminum tubes (2 ounce size) which were promptly crimped and tightly capped. The closed tubes were then set aside for determination of retinol stability after storage for various time periods at various temperatures.
- Retinol degrades under the influence of UV light. Accordingly, care must be taken at all stages of the emulsion preparation process to protect the retinol from exposure to UV light. This can be accomplished by turning out the lights in the processing area or by conducting the various handling and processing steps under yellow light.
- Mixture A the ingredient in the Oil Phase Ingredients designated as Mixture A consisted of 1.50 g myristyl myristate; 1.25 g oleic acid (Emersol 228); 1.25 g glyceryl stearate (Emerest 2400); 1.25 g stearic acid (Emersol 132); 1.00 g isopropyl palmitate; 1.00 stearoxytrimethylsilane (Dow Corning 580 Wax); 0.50 synthetic beeswax; 0.50 g stearyl alcohol; and 0.50 g cetyl alcohol.
- Mixture A consisted of 1.50 g myristyl myristate; 1.25 g oleic acid (Emersol 228); 1.25 g glyceryl stearate (Emerest 2400); 1.25 g stearic acid (Emersol 132); 1.00 g isopropyl palmitate; 1.00 stearoxytrimethylsilane (Dow Corning 580 Wax
- Mixture A was prepared by mixing the indicated ingredients in a glass container, stirring with heat until all ingredients were liquified and uniformly mixed; pouring the liquified mixture into shallow containers; and allowing the mixture to cool to ambient temperature.
- Concentrations of all-trans retinol in oil-in-water samples A, B and C in Table 1 were determined after storage for various time periods at various temperatures. Concentrations of retinol and other retinoids such as retinal (vitamin A aldehyde), retinyl acetate and retinyl palmitate can be determined by any suitable analytical procedure.
- retinoid concentrations were determined by a high performance liquid chromatography (HPLC) procedure in which the chromatograph was equipped with a reversed phase 5 micron C-8 column (25 cm in length ⁇ 4.6 mm in diameter) and a UV detector at 340nm.
- the sample to be analyzed was diluted with a solution of 50% by weight methanol and 50% by weight ethyl acetate to a concentration of 18 micrograms/ml and the retinoid was detected at 340nm.
- the gradient mobile phase consisted of an organic portion composed of 5 percent tetrahydrofuran in acetonitrile and an aqueous portion consisting of 0.05N ammonium acetate.
- the solvent program has an initial composition of 70% organic/30% aqueous which increases linearly to 80% organic/20% aqueous at 13 minutes, then again increases linearly to 100% organic at 15 minutes, where it stays until 19 minutes.
- the analytical conditions were run at a flow rate of 2 ml/min and thermostatically regulated at 40°C.
- the retention time of retinol (Vitamin A alcohol) is about 6.4 minutes.
- the retention times of retinal (Vitamin A aldehyde), retinyl acetate, and retinyl palmitate are about 7.5 mins., 10.1 mins. and 18.7 mins., respectively.
- the HPLC results were found to be reproducible to better than a 3% range of standard deviation.
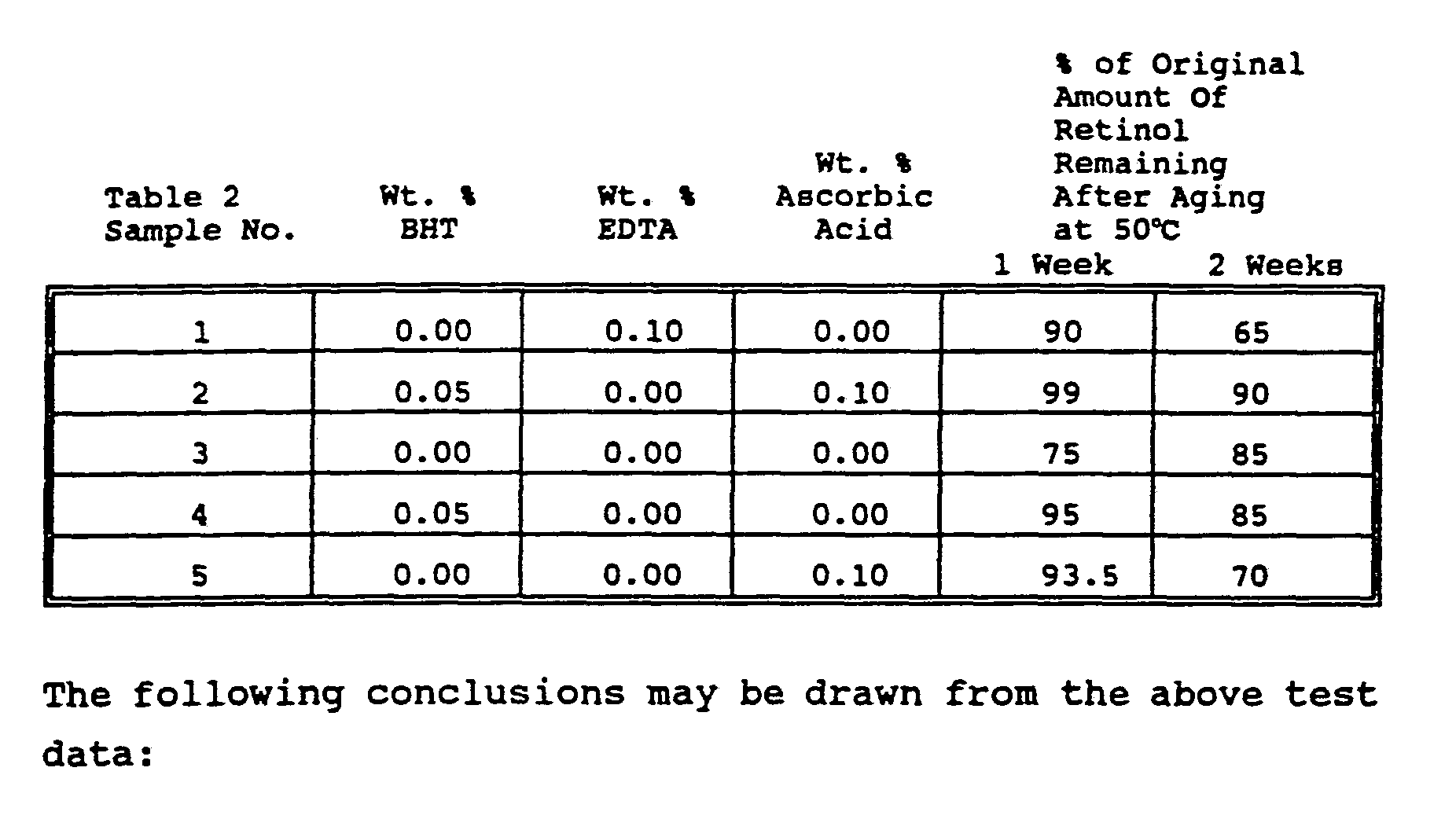
- EXAMPLE II Five water-in-oil (w/o) emulsions of retinol (Vitamin A alcohol) were prepared having the % w/w compositions set forth in Table 2. These emulsions were prepared according to the following procedure. The ingredients shown under the heading "Oil Phase Ingredients” were added under argon gas blanketing to a suitable sized glass beaker equipped with a stainless steel stirrer and heated with stirring to 80°C until uniformly mixed. The ingredients shown under the heading "Aqueous Phase Ingredients” were added under argon gas blanketing to a separate glass container equipped with a stainless steel stirrer and heated with stirring to 80°C until uniformly mixed. The pH of the Aqueous Phase Ingredients was adjusted to 4.7 using 50% NaOH.
- the Aqueous Phase Ingredients at 80°C were added to the Oil Phase Ingredients, also at 80°C, with agitation to form a water-in-oil emulsion, this addition step also being done under an argon gas blanket.
- the use of argon gas blanketing was continued through the preparation procedure being described.
- the water-in-oil emulsion was homogenized and cooled to about 50°C with stirring.
- the phenoxyethanol and the fragrance were then added to the emulsion.
- the retinol was then added to the emulsion which was then gradually cooled until the temperature was below 40°C.
- the water-in-oil emulsion was transferred under argon gas blanketing to small blind-end aluminum tubes which were promptly crimped and tightly capped.
- a water-in-oil emulsion of retinol containing neither an oil-soluble antioxidant, a water-soluble antioxidant nor a chelating agent (Table 2, comparative Sample 3) retained only 75% of its initial amount of retinol after one week aging at 50°C. After two weeks aging at 50°C, the analytical results indicated that 85% of the original amount of retinol remained in comparative Sample 3. This figure of 85% was thought to be inaccurate in view of the fact that only 75% of the original amount of retinol remained after one week aging. In any event, the chemical stability of retinol is poor and this emulsion cannot be considered the basis for a commercial product having acceptable long term chemical stability of retinol.
- a water-in-oil emulsion of retinol containing a water-soluble antioxidant (ascorbic acid) but containing neither an oil-soluble antioxidant or a chelating agent (Table 2, comparative Sample 5) was found to retain 93.5% of its original concentration of retinol after one week aging at 50°C. This represented somewhat of an improvement in chemical stability of retinol compared to comparative Sample 3 of Table 2 under the same storage conditions. However, after two weeks storage at 50°C, only 70% of the original amount of retinol remained in comparative Sample 5, Table 2. In view of the rapid fall off and low values comparative Sample 5 of Table 2 cannot be the basis for a commercial product having acceptable long term chemical stability of retinol.
- a water-in-oil emulsion of retinol containing an oil-soluble antioxidant (BHT) but containing neither a chelating agent nor a water-soluble antioxidant (Table 2, comparative Sample 4) was found to retain 95% of its original concentration of retinol after one week aging at 50°C and falls off rapidly to a low value of 85% after two weeks aging at the same temperature. This represents somewhat of an improvement over the chemical stability of retinol in comparative Sample 3 and comparative Sample 5 of Table 2. In view of the rapid decrease in retinol concentrations after two weeks and the relatively low quantity retained as retinol at that time, comparative Sample 4 of Table 2 is not suitable as the basis for a commercial product having acceptable chemical stability of retinol.
- Sample 2 was found to retain 99% of its original concentration of retinol after one week aging at 50°C and
- Table 2 could form the basis for a commercial product having acceptable long term chemical stability of retinol.
- Water-in-oil emulsions comprising retinol were prepared having the compositions given in Table 3. These emulsions were prepared by the same general procedure used to prepare the water-in-oil emulsions given in Table 2. After the water phase ingredients were added to the oil phase ingredients as described hereinabove, the remaining ingredients were added to the resulting water-in-oil emulsion in the order set forth in Table 3, with retinol being the last component to be added. As indicated earlier, the emulsions were prepared under argon gas blanketing (other inert gases, e.g. nitrogen or carbon dioxide could be used, if desired) to minimize the possibility of entraining oxygen during the various mixing steps. Measures were taken (as described above) to minimize the exposure of retinol to UV light.
- argon gas blanketing other inert gases, e.g. nitrogen or carbon dioxide could be used, if desired
- water-in- oil emulsions comprising retinol, a chelating agent and either a water-soluble antioxidant or an oil-soluble antioxidant retain greater than 90% of their original concentration of retinol after being aged for 13 weeks at room temperature (21°C). These same emulsions retain at least about 89% of their original concentration of retinol after being aged for 13 weeks at 40°C.
- a water-in-oil emulsion comprising retinol, a chelating agent, a water-soluble antioxidant and an oil-soluble antioxidant (Table 3, Sample 3) was found to retain 100% of its original amount of retinol after 13 weeks aging at room temperature, 96% of the original amount of retinol after 13 weeks aging at 40°C and 94% of the original amount of retinol after 13 weeks aging at 50°C. The same emulsion was found to retain 97% of its original amount of retinol after 26 weeks storage at 21°C, 92% of its original amount of retinol after 26 weeks storage at 40°, and 95.3% of its original amount of retinol after 52 weeks storage at room temperature.
- Two water-in-oil emulsions one comprising retinyl acetate and the other comprising retinyl palmitate, were prepared having the % w/w compositions set forth in Table 4, Samples 3 and 4, respectively. These two water-in-oil emulsions were prepared by the same general procedure used to prepare the water-in-oil emulsions shown in Tables 2 and 3.
- two oil-in-water emulsions one comprising retinyl acetate and the other comprising retinyl palmitate, were prepared having the % w/w compositions shown in Table 4, comparative Samples 1 and 2, respectively. These two oil-in-water emulsions were prepared by the same general procedure used to prepare the oil-in-water emulsions shown in Table 1.
- Mixture A consisted of 1.50 g myristyl myristate; 1.25 g oleic acid (Emersol 228); 1.25 g glyceryl stearate (Emerest 2400); 1.25 g stearic acid (Emersol 132); 1.00 g isopropyl palmitate; 1.00 stearoxytrimethylsilane (Dow Corning 580 Wax); 0.50 synthetic beeswax; 0.50 g stearyl alcohol; and 0.50 g cetyl alcohol.
- Mixture A was prepared by mixing the indicated ingredients in a glass container; stirring with heat until all ingredients were liquified and uniformly mixed; pouring the liquified mixture into shallow containers; and allowing the mixture to cool to ambient temperature.
- test results show that when retinyl acetate is formulated in the mentioned water-in-oil emulsion, ninety-one percent (91%) of the original amount of retinyl acetate is retained after 13 weeks storage at room temperature (21°C) , fifty-eight percent ( 58%) of the original amount of retinyl acetate is retained after 13 weeks storage at 40°C and sixteen percent ( 16%) of the original amount of retinyl acetate is retained after 13 weeks storage at 50°C.
- a water-in-oil cream composition was prepared according to the following procedure.
- a suitable sized glass beaker held under argon gas blanketing and fitted with a stainless steel stirrer 250 g mineral oil, 2 g propylparaben, 60 g Elfacos C-26, 50 g Elfacos E200, 30 g Elfacos ST-9, 10 g stearoxytrimethyIsilane, 10 g dimethicone (50cstk), and 0.5 g butylated hydroxytoluene (BHT) were heated to 80°C and mixed.
- BHT butylated hydroxytoluene
- the resulting water-in-oil emulsion was found to retain 100% of its original amount of retinol after 4 weeks aging at room temperature (21°C). 97.8% of the original amount of retinol was retained after 4 weeks aging at 40°C and 96.6% of the original amount was retained after 4 weeks agin at 50°C. After 13 weeks aging, the emulsion retained the following percentages of its original amount of retinol: 99.1% at room temperature; 96.2% at 40°C; and 94% at 50°C. After 26 weeks aging, the emulsion retained the following percentages of its original amount of retinol: 97% at room temperature and 92% at 40°C. This is excellent chemical stability of the retinol.
- a water-in-oil cream composition was prepared in accordance with the procedure of Example V with the amount of retinol being increased to 1.25% w/w and consists of the following ingredients:
- the resulting water-in-oil emulsion retained 93% of its original amount of retinol after aging for 13 weeks at room temperature ( 21°C) .
- a water-in-oil cream composition was prepared in accordance with the procedure of Example V with 0.5g propyl gallate replacing the butylated hydroxytoluene and omitting ascorbic acid.
- the composition consisted of the following ingredients: Ingredient % w/w mineral oil 25.000
- Vitamin A alcohol retinol
- the resulting water-in-oil emulsion composition was an off-white, water-in-oil cream which was found to retain 91% of its original retinol concentration after 2 weeks aging at room temperature (21°C), 84.8% after 2 weeks aging at 40°C and 86.8% after 2 weeks aging at 50°C.
- the emulsion was found to retain 79% of its original amount of retinol after 8 weeks aging at room temperature (21°C), 58% after 8 weeks aging at 40°C, and 65% after 8 weeks aging at 50°C.
- the emulsion was found to retain 77% of its original amount of retinol after 13 weeks aging at room temperature, 78% after 13 weeks aging at 40°C and 59% after 13 weeks aging at 50°C.
- EXAMPLE VIII A water-in-oil cream composition was prepared in accordance with the procedure of Example V with 0.5 g nordihydroguiaretic acid replacing the butylated hydroxytoluene and omitting the ascorbic acid.
- the composition consisted of the following ingredients:
- the resulting water-in-oil emulsion composition was found to retain 96.8% of its original amount of retinol after 2 weeks aging at room temperature (21°C), 87.6% after 2 weeks aging at 40°C and 91.2% after 2 weeks aging at 50°C.
- the composition was found to retain 91.2% of its original amount of retinol after 8 weeks aging at room temperature, 72.6% after 8 weeks aging at 40°C, and 50.8% after 8 weeks aging at 50°C.
- the composition was found to retain 91.6% of its original amount of retinol after 13 weeks aging at room temperature, 69.0% after 13 weeks aging at 40°C, and 59.3% after 13 weeks aging at 50°C.
- a water-in-oil cream composition was prepared in accordance with the procedure of Example V with 0.5 g monothioglycerol replacing the ascorbic acid and omitting the butylated hydroxytoluene.
- the composition consisted of the following ingredients:
- the resulting water-in-oil emulsion composition was found to retain 92.8% of its original amount of retinol after 4 weeks aging at room temperature (21°C), 92.8% after 4 weeks aging at 40°C, and 90.4% after 4 weeks aging at 50°C.
- the composition was found to retain 90.8% of its original level of retinol after 8 weeks aging at room temperature. 82.4% after 8 weeks aging at 40°C, and 65.3% after 8 weeks aging at 50°C.
- the composition was found to retain 80% of its original level of retinol after 13 weeks aging at room temperature, 62.3% after 13 weeks aging at 40°C, and 65.7% after 13 weeks aging at 50°C.
- a water-in-oil cream composition was prepared in accordance with the procedure of Example V with 1.0 g sodium metabisulfite replacing the ascorbic acid and consisted of the following ingredients:
- composition After 13 weeks aging, the composition was found to retain 96% of its original level of retinol.
- a water-in-oil cream composition was prepared in accordance with the procedure of Example V with 0.5 g sodium formaldehyde sulfoxylate replacing the ascorbic acid and omitting the butylated hydroxytoluene.
- the composition consisted of the following ingredients:
- a water-in-oil cream composition was prepared in accordance with the procedure of Example V with 0.5 g 1,4 diazobicyclo-(2,2,2)-octane replacing the ascorbic acid and omitting the butylated hydroxytoluene.
- the composition consisted of the following ingredients: Ingredient % w/w mineral oil 25.000
- the resulting water-in-oil emulsion composition was an off-white cream and was found to retain 91.8% of its original amount of retinol after 13 weeks aging at room temperature (21°C).
- a water-in-oil cream composition was prepared in accordance with the procedure of Example V and consisted of the following ingredients: Ingredient % w/w mineral oil 25.000
- EXAMPLE XIV A water-in-oil cream composition was prepared in accordance with the procedure of Example V with the addition of 20 g silicon dioxide to the phase containing the mineral oil and consisted of the following ingredients: Ingredient % w/w mineral oil 25.000
- the resulting water-in-oil emulsion composition was an off-white cream.
- a retinol cream composition was prepared according to the following procedure.
- a suitable sized glass beaker blanketed with argon gas and fitted with a stainless steel stirrer 10g PVP/Eicosene Copolymer, 5g Phenoxyethanol, 2.45g Polysorbate 20, 1.54g Dimethicone, 10g Arlacel 481, 25g PEG-7 Hydrogenated Castor Oil, 15g C12-15 Alcohols Benzoate, 85g Cetearyl Octanoate, 20g Silicon Dioxide, and 0.25g BHT were heated to 80°C and mixed.
Landscapes
- Health & Medical Sciences (AREA)
- Life Sciences & Earth Sciences (AREA)
- Chemical & Material Sciences (AREA)
- Veterinary Medicine (AREA)
- Public Health (AREA)
- General Health & Medical Sciences (AREA)
- Animal Behavior & Ethology (AREA)
- Epidemiology (AREA)
- Pharmacology & Pharmacy (AREA)
- Medicinal Chemistry (AREA)
- General Chemical & Material Sciences (AREA)
- Oil, Petroleum & Natural Gas (AREA)
- Engineering & Computer Science (AREA)
- Chemical Kinetics & Catalysis (AREA)
- Dermatology (AREA)
- Birds (AREA)
- Emergency Medicine (AREA)
- Proteomics, Peptides & Aminoacids (AREA)
- Bioinformatics & Cheminformatics (AREA)
- Organic Chemistry (AREA)
- Nuclear Medicine, Radiotherapy & Molecular Imaging (AREA)
- Dispersion Chemistry (AREA)
- Cosmetics (AREA)
- Acyclic And Carbocyclic Compounds In Medicinal Compositions (AREA)
Abstract
Description
Claims
Priority Applications (9)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| KR1019930700583A KR100250700B1 (en) | 1991-06-27 | 1991-06-28 | Stabilized retinoid-containing skin care compositions |
| DE69130020T DE69130020T2 (en) | 1991-06-27 | 1991-06-28 | STABILIZED, RETINOID CONTAINING SKIN CARE PREPARATIONS |
| EP91913705A EP0549592B2 (en) | 1991-06-27 | 1991-06-28 | Stabilized retinoid-containing skin care compositions |
| CA002090104A CA2090104C (en) | 1991-06-27 | 1991-06-28 | Stabilized retinoid-containing skin care compositions |
| BR9106891A BR9106891A (en) | 1991-06-27 | 1991-06-28 | Compositions for skin treatment |
| AU82997/91A AU664973B2 (en) | 1991-06-27 | 1991-06-28 | Stabilized retinoid-containing water-in-oil skin care compositions |
| NO930660A NO304405B1 (en) | 1991-06-27 | 1993-02-24 | skin Care Compositions |
| FI930887A FI930887A (en) | 1991-06-27 | 1993-02-26 | HUDVAORDKOMPOSITIONER |
| FI981664A FI104407B (en) | 1991-06-27 | 1998-07-27 | A process for preparing skin care compositions |
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| US71926491A | 1991-06-27 | 1991-06-27 | |
| US719,264 | 1991-06-27 |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| WO1993000085A1 true WO1993000085A1 (en) | 1993-01-07 |
Family
ID=24889399
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/US1991/004471 WO1993000085A1 (en) | 1991-06-27 | 1991-06-28 | Stabilized retinoid-containing skin care compositions |
Country Status (22)
| Country | Link |
|---|---|
| EP (1) | EP0549592B2 (en) |
| JP (2) | JP3435510B2 (en) |
| KR (1) | KR100250700B1 (en) |
| AU (1) | AU664973B2 (en) |
| BR (1) | BR9106891A (en) |
| CA (1) | CA2090104C (en) |
| CO (1) | CO4130180A1 (en) |
| DE (1) | DE69130020T2 (en) |
| EG (1) | EG19852A (en) |
| ES (1) | ES2120417T5 (en) |
| FI (2) | FI930887A (en) |
| MA (1) | MA22240A1 (en) |
| MX (1) | MX187577B (en) |
| MY (2) | MY131215A (en) |
| NO (1) | NO304405B1 (en) |
| NZ (2) | NZ272557A (en) |
| PH (1) | PH31832A (en) |
| PT (1) | PT98512B (en) |
| SG (1) | SG66224A1 (en) |
| TW (1) | TW203009B (en) |
| WO (1) | WO1993000085A1 (en) |
| ZA (1) | ZA915285B (en) |
Cited By (29)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO1994009756A1 (en) * | 1992-11-05 | 1994-05-11 | Unilever Plc | Retinol containing cosmetic composition |
| FR2714595A1 (en) * | 1993-12-30 | 1995-07-07 | Oreal | Water-in-oil emulsion containing retinol, its use and its conditioning. |
| WO1995025507A1 (en) * | 1994-03-22 | 1995-09-28 | Pierre Fabre Dermo-Cosmetique | Retinal-containing composition |
| WO1997021423A1 (en) * | 1995-12-11 | 1997-06-19 | The Procter & Gamble Company | Topical retinoid composition |
| WO1997031620A2 (en) * | 1996-03-01 | 1997-09-04 | Johnson & Johnson Consumer Products, Inc. | Topical compositions comprising an oil-in-water emulsion and a retinoid |
| US5744148A (en) * | 1996-09-20 | 1998-04-28 | Chesebrough-Pond's Usa Co., Division Of Conopco, Inc. | Stabilization of an unstable retinoid in oil-in-water emulsions for skin care compositions |
| US5851538A (en) * | 1995-12-29 | 1998-12-22 | Advanced Polymer Systems, Inc. | Retinoid formulations in porous microspheres for reduced irritation and enhanced stability |
| US5891470A (en) * | 1998-04-17 | 1999-04-06 | Advanced Polymer Systems, Inc. | Softgel formulation containing retinol |
| WO1999053904A2 (en) * | 1998-04-17 | 1999-10-28 | Advanced Polymer Systems, Inc. | Softgel-compatible composition containing retinol |
| US5976555A (en) * | 1994-09-07 | 1999-11-02 | Johnson & Johnson Consumer Products, Inc. | Topical oil-in-water emulsions containing retinoids |
| US6124274A (en) * | 1997-10-15 | 2000-09-26 | Basf Aktiengesellschaft | Use of ascorbyl 2'-phosphates for stabilizing vitamin A and/or vitamin A derivatives in cosmetic and pharmaceutical preparations |
| EP1055720A2 (en) * | 1999-05-28 | 2000-11-29 | JOHNSON & JOHNSON CONSUMER COMPANIES, INC. | Compositions for stabilizing oxygen-labile species |
| US6193956B1 (en) | 1995-09-06 | 2001-02-27 | Johnson & Johnson Consumer Companies, Inc. | Topical compositions |
| US6207717B1 (en) | 1999-01-12 | 2001-03-27 | Dow Corning Corporation | Entrapment of vitamins with an elastomeric silicone polyether |
| USH2043H1 (en) | 1997-05-23 | 2002-08-06 | The Procter & Gamble Company | Skin care compositions |
| WO2004012696A1 (en) * | 2002-07-24 | 2004-02-12 | Basf Aktiengesellschaft | Retinoid-containing preparations |
| WO2004032893A1 (en) * | 2002-10-11 | 2004-04-22 | Kose Corporation | Oily solid cosmetic and method for production thereof |
| WO2005070373A1 (en) * | 2004-01-22 | 2005-08-04 | Basf Aktiengesellschaft | Retinoid-containing preparations |
| USRE38813E1 (en) | 1990-01-29 | 2005-10-04 | Johnson & Johnson Consumer Companies, Inc. | Retinoid compositions containing a water soluble antioxidant and a chelator |
| EP1714640A1 (en) * | 1992-07-13 | 2006-10-25 | Shiseido Company, Ltd. | Stabilised external skin treatment composition comprising retinol. |
| US7604812B2 (en) | 2000-12-15 | 2009-10-20 | Patrick Franke | Hypoallergenic and non-irritant skin care formulations |
| US8481011B2 (en) | 2005-12-14 | 2013-07-09 | Pierre Fabre Dermo-Cosmetique | Use of polyunsaturated compounds as whitening agents |
| WO2018111952A1 (en) | 2016-12-13 | 2018-06-21 | The Procter & Gamble Company | Stable personal care compositions containing a retinoid |
| WO2021123337A1 (en) | 2019-12-20 | 2021-06-24 | L'oreal | Retinol-based composition |
| WO2021121677A1 (en) | 2019-12-20 | 2021-06-24 | L'oreal | Retinol-based serum |
| WO2021123233A1 (en) | 2019-12-20 | 2021-06-24 | L'oreal | Retinol-based composition |
| FR3111074A1 (en) | 2020-06-08 | 2021-12-10 | L'oreal | Retinol-based composition |
| FR3111075A1 (en) | 2020-06-08 | 2021-12-10 | L'oreal | Retinol-based composition |
| US11197826B2 (en) | 2015-12-23 | 2021-12-14 | Novartis Ag | Oil-in-water emulsions including retinoic acid |
Families Citing this family (11)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JPH0632722A (en) * | 1992-07-13 | 1994-02-08 | Shiseido Co Ltd | External preparation for skin |
| GR1002207B (en) * | 1992-08-06 | 1996-03-27 | Johnson & Johnson Consumer | Skin care compositions containing imidazoles. |
| KR100355892B1 (en) * | 2000-01-06 | 2002-10-12 | 주식회사 코리아나화장품 | Skin care composition containing Retinol and Tetradibutyl Pentaerithrityl Hydroxyhydrocinnamate |
| DE10048125A1 (en) * | 2000-09-28 | 2002-04-18 | Beiersdorf Ag | High water emulsion type W / O preparations, with medium polar and / or nonpolar lipids and one or more A-O-B-O-A surfactant polyethers |
| DE10048366A1 (en) | 2000-09-29 | 2002-04-11 | Beiersdorf Ag | Emulsion compositions W / O with increased water content, with medium-polar and / or nonpolar lipids and one or more surfactant polyethers of the type A-O-B-O-A and with at least one substance selected from the group of cationic polymers |
| DE10048427A1 (en) * | 2000-09-29 | 2002-04-11 | Beiersdorf Ag | Water-in-oil emulsions with a high water content, useful for cosmetic and medicinal applications, include a alkylene oxide copolymer nonionic surfactant and an anionic and/or amphoteric polymer |
| DE10048429A1 (en) * | 2000-09-29 | 2002-04-11 | Beiersdorf Ag | Emulsion compositions W / O with increased water content, with medium polar and / or nonpolar lipids and one or more surfactant polyethers of type A-O-B-O-A and a substance selected from the group of nonionic polymers |
| JP4119296B2 (en) * | 2003-04-14 | 2008-07-16 | 株式会社コーセー | Cosmetics |
| JP2006045080A (en) * | 2004-08-02 | 2006-02-16 | Pola Chem Ind Inc | Skin care preparation suitable as quasi-drug |
| DE102006062568A1 (en) | 2006-12-29 | 2008-07-03 | Henkel Kgaa | Stabilizing the color and/or odor of a water-containing cosmetic or pharmaceutical composition comprises adding a mixture of a phenolic antioxidant and a complexing agent |
| JP6386713B2 (en) * | 2013-10-03 | 2018-09-05 | 国立大学法人千葉大学 | Preventive and / or therapeutic agent for cerebral circulation disorder |
Citations (4)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US4214000A (en) * | 1978-10-30 | 1980-07-22 | Johnson & Johnson | Zinc salt of all-trans-retinoic acid for the treatment of acne |
| US4603146A (en) * | 1984-05-16 | 1986-07-29 | Kligman Albert M | Methods for retarding the effects of aging of the skin |
| US4720353A (en) * | 1987-04-14 | 1988-01-19 | Richardson-Vicks Inc. | Stable pharmaceutical w/o emulsion composition |
| US4877805A (en) * | 1985-07-26 | 1989-10-31 | Kligman Albert M | Methods for treatment of sundamaged human skin with retinoids |
Family Cites Families (4)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| DE3372843D1 (en) * | 1982-05-15 | 1987-09-10 | Beecham Group Plc | Skin treatment compositions |
| DE3514724A1 (en) † | 1985-04-24 | 1986-10-30 | Albin F. Dr. 4200 Oberhausen Jereb | Ointment for the prevention and treatment of disorders of the eyes and skin |
| JPH06105042B2 (en) * | 1989-06-30 | 1994-12-21 | マツダ株式会社 | Engine intake system |
| JP3014780B2 (en) † | 1990-01-29 | 2000-02-28 | ジヨンソン・アンド・ジヨンソン・コンシユーマー・プロダクツ・インコーポレーテツド | Skin care composition |
-
1991
- 1991-06-28 SG SG1996001619A patent/SG66224A1/en unknown
- 1991-06-28 EP EP91913705A patent/EP0549592B2/en not_active Expired - Lifetime
- 1991-06-28 JP JP51304691A patent/JP3435510B2/en not_active Expired - Lifetime
- 1991-06-28 WO PCT/US1991/004471 patent/WO1993000085A1/en active IP Right Grant
- 1991-06-28 ES ES91913705T patent/ES2120417T5/en not_active Expired - Lifetime
- 1991-06-28 CA CA002090104A patent/CA2090104C/en not_active Expired - Lifetime
- 1991-06-28 BR BR9106891A patent/BR9106891A/en not_active IP Right Cessation
- 1991-06-28 DE DE69130020T patent/DE69130020T2/en not_active Expired - Lifetime
- 1991-06-28 KR KR1019930700583A patent/KR100250700B1/en not_active IP Right Cessation
- 1991-06-28 AU AU82997/91A patent/AU664973B2/en not_active Expired
- 1991-07-08 ZA ZA915285A patent/ZA915285B/en unknown
- 1991-07-10 PH PH42767A patent/PH31832A/en unknown
- 1991-07-17 MY MYPI91001291A patent/MY131215A/en unknown
- 1991-07-17 MY MYPI98003948A patent/MY132824A/en unknown
- 1991-07-18 MX MX9100278A patent/MX187577B/en unknown
- 1991-07-18 CO CO92344280A patent/CO4130180A1/en unknown
- 1991-07-19 NZ NZ272557A patent/NZ272557A/en unknown
- 1991-07-19 NZ NZ239051A patent/NZ239051A/en not_active IP Right Cessation
- 1991-07-29 MA MA22518A patent/MA22240A1/en unknown
- 1991-07-31 PT PT98512A patent/PT98512B/en not_active IP Right Cessation
- 1991-08-09 TW TW80106279A patent/TW203009B/zh not_active IP Right Cessation
- 1991-09-07 EG EG53091A patent/EG19852A/en active
-
1993
- 1993-02-24 NO NO930660A patent/NO304405B1/en not_active IP Right Cessation
- 1993-02-26 FI FI930887A patent/FI930887A/en unknown
-
1998
- 1998-07-27 FI FI981664A patent/FI104407B/en active
-
2003
- 2003-03-20 JP JP2003077559A patent/JP3927511B2/en not_active Expired - Lifetime
Patent Citations (4)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US4214000A (en) * | 1978-10-30 | 1980-07-22 | Johnson & Johnson | Zinc salt of all-trans-retinoic acid for the treatment of acne |
| US4603146A (en) * | 1984-05-16 | 1986-07-29 | Kligman Albert M | Methods for retarding the effects of aging of the skin |
| US4877805A (en) * | 1985-07-26 | 1989-10-31 | Kligman Albert M | Methods for treatment of sundamaged human skin with retinoids |
| US4720353A (en) * | 1987-04-14 | 1988-01-19 | Richardson-Vicks Inc. | Stable pharmaceutical w/o emulsion composition |
Non-Patent Citations (1)
| Title |
|---|
| See also references of EP0549592A4 * |
Cited By (49)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| USRE38813E1 (en) | 1990-01-29 | 2005-10-04 | Johnson & Johnson Consumer Companies, Inc. | Retinoid compositions containing a water soluble antioxidant and a chelator |
| EP1714640A1 (en) * | 1992-07-13 | 2006-10-25 | Shiseido Company, Ltd. | Stabilised external skin treatment composition comprising retinol. |
| WO1994009756A1 (en) * | 1992-11-05 | 1994-05-11 | Unilever Plc | Retinol containing cosmetic composition |
| EP0953338A1 (en) * | 1993-12-30 | 1999-11-03 | L'oreal | Packaging device for a product |
| EP0666076A2 (en) * | 1993-12-30 | 1995-08-09 | L'oreal | Water in oil emulsion containing retinol |
| EP0666076A3 (en) * | 1993-12-30 | 1997-01-02 | Oreal | Water in oil emulsion containing retinol. |
| US5800596A (en) * | 1993-12-30 | 1998-09-01 | L'oreal | Water-in-oil emulsion containing retinol, its use and its packaging |
| US5656672A (en) * | 1993-12-30 | 1997-08-12 | L'oreal | Water-in-oil emulsion containing retinol its use |
| FR2714595A1 (en) * | 1993-12-30 | 1995-07-07 | Oreal | Water-in-oil emulsion containing retinol, its use and its conditioning. |
| FR2717686A1 (en) * | 1994-03-22 | 1995-09-29 | Fabre Pierre Cosmetique | Dermo-cosmetic composition and preparation process. |
| US5776986A (en) * | 1994-03-22 | 1998-07-07 | Pierre Fabre Dermo-Cosmetique | Composition containing retinal |
| AU701657B2 (en) * | 1994-03-22 | 1999-02-04 | Pierre Fabre Dermo-Cosmetique | Composition containing retinal |
| WO1995025507A1 (en) * | 1994-03-22 | 1995-09-28 | Pierre Fabre Dermo-Cosmetique | Retinal-containing composition |
| US6461622B2 (en) | 1994-09-07 | 2002-10-08 | Johnson & Johnson Consumer Companies, Inc. | Topical compositions |
| US5976555A (en) * | 1994-09-07 | 1999-11-02 | Johnson & Johnson Consumer Products, Inc. | Topical oil-in-water emulsions containing retinoids |
| US6193956B1 (en) | 1995-09-06 | 2001-02-27 | Johnson & Johnson Consumer Companies, Inc. | Topical compositions |
| WO1997021423A1 (en) * | 1995-12-11 | 1997-06-19 | The Procter & Gamble Company | Topical retinoid composition |
| US5851538A (en) * | 1995-12-29 | 1998-12-22 | Advanced Polymer Systems, Inc. | Retinoid formulations in porous microspheres for reduced irritation and enhanced stability |
| WO1997031620A3 (en) * | 1996-03-01 | 1998-01-08 | Johnson & Johnson Consumer | Topical compositions comprising an oil-in-water emulsion and a retinoid |
| WO1997031620A2 (en) * | 1996-03-01 | 1997-09-04 | Johnson & Johnson Consumer Products, Inc. | Topical compositions comprising an oil-in-water emulsion and a retinoid |
| US5744148A (en) * | 1996-09-20 | 1998-04-28 | Chesebrough-Pond's Usa Co., Division Of Conopco, Inc. | Stabilization of an unstable retinoid in oil-in-water emulsions for skin care compositions |
| USH2043H1 (en) | 1997-05-23 | 2002-08-06 | The Procter & Gamble Company | Skin care compositions |
| US6124274A (en) * | 1997-10-15 | 2000-09-26 | Basf Aktiengesellschaft | Use of ascorbyl 2'-phosphates for stabilizing vitamin A and/or vitamin A derivatives in cosmetic and pharmaceutical preparations |
| WO1999053904A3 (en) * | 1998-04-17 | 1999-12-23 | Advanced Polymer Systems Inc | Softgel-compatible composition containing retinol |
| WO1999053904A2 (en) * | 1998-04-17 | 1999-10-28 | Advanced Polymer Systems, Inc. | Softgel-compatible composition containing retinol |
| US5891470A (en) * | 1998-04-17 | 1999-04-06 | Advanced Polymer Systems, Inc. | Softgel formulation containing retinol |
| US6228894B1 (en) | 1998-04-17 | 2001-05-08 | Enhanced Derm Technologies, Inc. | Softgel-compatible composition containing retinol |
| US6207717B1 (en) | 1999-01-12 | 2001-03-27 | Dow Corning Corporation | Entrapment of vitamins with an elastomeric silicone polyether |
| EP1055720A3 (en) * | 1999-05-28 | 2001-03-07 | JOHNSON & JOHNSON CONSUMER COMPANIES, INC. | Compositions for stabilizing oxygen-labile species |
| EP1055720A2 (en) * | 1999-05-28 | 2000-11-29 | JOHNSON & JOHNSON CONSUMER COMPANIES, INC. | Compositions for stabilizing oxygen-labile species |
| US7604812B2 (en) | 2000-12-15 | 2009-10-20 | Patrick Franke | Hypoallergenic and non-irritant skin care formulations |
| WO2004012696A1 (en) * | 2002-07-24 | 2004-02-12 | Basf Aktiengesellschaft | Retinoid-containing preparations |
| WO2004032893A1 (en) * | 2002-10-11 | 2004-04-22 | Kose Corporation | Oily solid cosmetic and method for production thereof |
| WO2005070373A1 (en) * | 2004-01-22 | 2005-08-04 | Basf Aktiengesellschaft | Retinoid-containing preparations |
| DE102004003478A1 (en) * | 2004-01-22 | 2005-08-18 | Basf Ag | Retinoid-containing preparations |
| US8481011B2 (en) | 2005-12-14 | 2013-07-09 | Pierre Fabre Dermo-Cosmetique | Use of polyunsaturated compounds as whitening agents |
| US11197826B2 (en) | 2015-12-23 | 2021-12-14 | Novartis Ag | Oil-in-water emulsions including retinoic acid |
| WO2018111952A1 (en) | 2016-12-13 | 2018-06-21 | The Procter & Gamble Company | Stable personal care compositions containing a retinoid |
| US10493009B2 (en) | 2016-12-13 | 2019-12-03 | The Procter & Gamble Company | Stable personal care compositions containing a retinoid |
| WO2021123337A1 (en) | 2019-12-20 | 2021-06-24 | L'oreal | Retinol-based composition |
| WO2021123233A1 (en) | 2019-12-20 | 2021-06-24 | L'oreal | Retinol-based composition |
| FR3104977A1 (en) | 2019-12-20 | 2021-06-25 | L'oreal | Retinol based serum |
| FR3104976A1 (en) | 2019-12-20 | 2021-06-25 | L'oreal | Retinol-based composition |
| FR3104975A1 (en) | 2019-12-20 | 2021-06-25 | L'oreal | Retinol-based composition |
| WO2021121677A1 (en) | 2019-12-20 | 2021-06-24 | L'oreal | Retinol-based serum |
| FR3111074A1 (en) | 2020-06-08 | 2021-12-10 | L'oreal | Retinol-based composition |
| FR3111075A1 (en) | 2020-06-08 | 2021-12-10 | L'oreal | Retinol-based composition |
| WO2021250008A1 (en) | 2020-06-08 | 2021-12-16 | L'oreal | Retinol-based composition |
| WO2021250001A1 (en) | 2020-06-08 | 2021-12-16 | L'oreal | Retinol-based composition |
Also Published As
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| AU664973B2 (en) | Stabilized retinoid-containing water-in-oil skin care compositions | |
| US5559149A (en) | Skin care compositions containing retinoids | |
| AU674324B2 (en) | Compositions containing imidazoles | |
| EP0440398B1 (en) | Skin care compositions | |
| US6080393A (en) | Skin care composition comprising a retinoid | |
| US6193956B1 (en) | Topical compositions | |
| US5976555A (en) | Topical oil-in-water emulsions containing retinoids | |
| CA2247645C (en) | Topical compositions comprising an oil-in-water emulsion and a retinoid | |
| CA2336799C (en) | Improved stable topical ascorbic acid compositions | |
| EP0818988A2 (en) | Skin care compositions containing retinoids and liposomes | |
| WO1996031194A9 (en) | Skin care compositions containing retinoids and liposomes | |
| WO1996007396A9 (en) | Retinoid compositions | |
| US5583136A (en) | Retinoid containing skin care compositions containing imidazoles |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| AK | Designated states |
Kind code of ref document: A1 Designated state(s): AT AU BB BG BR CA CH CS DE DK ES FI GB HU JP KP KR LK LU MC MG MN MW NL NO PL RO SD SE SU |
|
| AL | Designated countries for regional patents |
Kind code of ref document: A1 Designated state(s): AT BE CH DE DK ES FR GB GR IT LU NL SE BF BJ CF CG CI CM GA GN ML MR SN TD TG |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 2090104 Country of ref document: CA |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 930887 Country of ref document: FI Ref document number: 1019930700583 Country of ref document: KR |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 1991913705 Country of ref document: EP |
|
| WWP | Wipo information: published in national office |
Ref document number: 1991913705 Country of ref document: EP |
|
| REG | Reference to national code |
Ref country code: DE Ref legal event code: 8642 |
|
| REG | Reference to national code |
Ref country code: DE Ref legal event code: 8642 |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 981664 Country of ref document: FI |
|
| WWG | Wipo information: grant in national office |
Ref document number: 1991913705 Country of ref document: EP |
|
| WWG | Wipo information: grant in national office |
Ref document number: 981664 Country of ref document: FI |