THERAPEUTICALLY USEFUL 2-AMINOTETRALIN DERIVATIVES
FIELD OF THE INVENTION
The present invention is related to new 1,2,3,4-tetrahydro-2-naphthylamines, to processes for preparing such compounds, pharmaceutical preparation of such compounds and the use of such compounds in manufacture of a pharmaceutical preparation.
BACKGROUND OF THE INVENTION
Psychiatric diseases are thought to be due to dysfunctions in monoaminergic neuronal systems, particularly those involving serotonin (5-HT) and dopamine (DA).
Anxiety is associated with increased activity in 5-HT systems. In animals where
5-HT has been depleted, benzodiazepine anxiolytics are not active in anti-anxiety assays that they otherwise are effective in. Seronotin neurons have autoreceptors that, when activated by agonists, depress firing rates of 5-HT cells. These receptors are of the 5- HT1A subtype. 5-HT1A agonists are anxiolytic. Buspirone is a marketed 5-HT1A agonist that is an anxiolytic. Gepirone is another 5-HT1A agonist with clinically demonstrated anti-anxiety activities.
Depression is a psychiatric condition thought to be associated with decreased 5- HT release. Many anti-depressants potentiate the effects of 5-HT by blocking the termination of activity through reuptake into nerve terminals. 5-HT1A agonists can can activate postsynaptically; they thus may also be anti-depressants. Gepirone has already been demonstrated to have ameliorative effects on some depressive endpoints in some patients.
Serotonin is also involved in the regulation of feeding and sexual behavior and in cardiovascular regulation. Thus, 5-HT1A agonists may be useful in treating overeating and sexual dysfunction. These compounds have been shown to alter feeding and sexual behavior in animals. They may also be useful in the treatment of obsessive/compulsive disorders, alcohol abuse and violent behavior. 5-HT1A agonists are also known to depress sympathetic nerve discharge and thus lower blood pressure. Thus, they may be useful in treating hypertension, congestive heart failure (by reducing cardiovascular afterload) and heart attack (be removing sympathetic drive to the heart).
Schizophrenia is thought to be due to hyperactivity in DA systems. Thus, currently available anti-psychotics are DA antagonists. Dopamine autoreceptors depress DA neuron firing rates, DA synthesis and release. Thus DA autoreceptor agonists can
also be expected to be anti-psychotics. DA agonists are also useful for treating Parkinsonism, a disease caused by degeneration of DA neurons, and hyperprolactinemia, since DA agonists depress prolactin release.
Dopamine autoreceptor antagonists are a new class of drugs that increase release of DA by releasing the DA neuron from autoreceptor control. Thus, these drugs can be expected to be useful in conditions treatable with amphetamine and other similar stimulants which directly release DA. However, DA autoreceptor agonists will be much milder stimulants because, rather than directly releasing DA, they simply increase the release associated with the normal DA activity by releasing the cell from autoreceptor control. Thus, DA autoreceptor antagonists can be expected to be useful in treating overeating, attention deficit disorders, psychiatric, cognitive and motor retardation in demented and elderly patients, and in treating nausea and dizziness with space travel.
The compounds of the present invention have a variety of effects at 5-HT1A and DA receptors, and offer a variety of utilities associated with those activities.
Clinically, 5-HT1A agonists have also demonstrated anxiolytic properties. The drug, Buspirone, is the only currently available marketed 5-HT1A agonist having anxiolytic activity. This compound antagonizes dopamine receptors at the same dose it stimulates 5-HT1A receptors. A similar drug, Gepirone, also has dopamine antagonist properties. These dopamine antagonist properties reduce the clinical utility of these compounds however because long term treatment with dopamine antagonists can produce tardive dyskinesia.
The search for new CNS active compounds is focused on finding compounds with selective 5-HT1A receptor agonist effects without detrimentally influencing central dopamine receptors.
Drugs acting on central dopamine transmission are clinically effective in treating a variety of central nervous system disorders such as parkinsonism, schizophrenia, and manic-depressive illness. In parkinsonism, for example, the nigro-neostriatal hypofunction can be restored by an increase in postsynaptic dopamine receptor stimulation. In schizophrenia, the condition can be normalized by achieving a decrease in postsynaptic dopamine receptor stimulation. Classical anti-psychotic agents directly block the postsynaptic dopamine receptor. The same effect can be achieved by inhibition of intraneuronal presynaptic events essential for the maintenance of adequate neuro- transmission, transport mechanism and transmitter synthesis.
In recent years a large body of pharmacological, biochemical and electrophysical evidence has provided considerable support in favor of the existence of a specific population of central autoregulatory dopamine receptors located in the dopaminergic neuron itself. These receptors are part of a homeostatic mechanism that modulates nerve impulse flow and transmitter synthesis and regulates the amount of dopamine released from the nerve endings.
Direct dopamine receptor agonists, like apomorphine, are able to activate the dopamine autoreceptors as well as the post synaptic dopamine receptors. The effects of autoreceptor stimulation appear to predominate when apomorphine is administered at low doses, whereas at higher doses the attenuation of dopamine transmission is outweighed by the enhancement of postsynaptic receptor stimulation. The anti-psychotic and anti- dyskinetic effects in man of low doses of apomoiphine are likely due to the autoreceptor- stimulator properties of this dopamine receptor agonist. This body of knowledge indicates dopamine receptor stimulants with a high selectivity for central nervous dop- amine autoreceptors would be valuable in treating psychiatric disorders.
INFORMATION DISCLOSURE STATEMENT
The following documents could be important in the examination of this application.
Arvidsson, L.-E., et al., J. Med. Chem., 24, 921 (1981), describes hydroxy-2- aminotetralins where the amine is substituted with one n-propyl, one benzyl or two n- propyl substitutents. The 5-, 6-, and 7-hydroxy compounds are described as active central dopamine-receptor agonists and the 8-hydroxy compound is described as a central
5-HT receptor agonist devoid of dopamine receptor stimulating activity.
Arvidsson, L.-E., et al., J. Med. Chem., 27, 45 (1984), describes 2-aminotetra- lins where the amine is substituted with one or two methyl, ethyl, n-propyl, i-propyl, n- butyl, or benzyl substituents. The 2-piperidinyltetralin is also described. Several of these compounds were found to be potent 5-HT agonists devoid of dopamine-mimetic effects.
Arvidsson, L.-E., et al., J. Med. Chem., 30, 2105 (1987), describes 8-hydroxy- 1-methyl-2-(di-n-propylamino)tetralins. These compounds were 5-HT receptor agonists.
The Arvidsson, L.-E. et al 8-hydroxy and 8-methoxy tetralin compounds are also disclosed in Derwent documents 00389J/47, 94981D/51 and 045535J.48.
McDermed, et al., J. Med. Chem., 18, 362 (1975) describes 5,6 dihydroxy-2-
aminotetralins. In addition, the 5,8 and 7,8 disubstituted compounds are also disclosed. The amine can be a mono or di substituted with simple alkyl groups, benzyl groups alkylalkoxy groups or the amine can be a 5 or 6 membered hydrocarbon or heterocyclic amine. These compounds are indicated to have dopaminergic properties although certain compounds are reported to be inactive.
McDermed, et al., J. Med. Chem., 19, 547 (1976) describes 5-,
6-, or 7-hydroxy-2-dipropylaminotetralins. These compounds are described as dopaminergic compounds.
Rusterholz, et al., J. Med. Chem., 19, 99 (1976) describes 5,8 disubstituted-2- aminotetralins with the amine being substituted with hydrogen, methyl, or cyanopropyl groups. Some of these compounds are potent prolactin inhibitors and believed to be dopamine agonists.
Ames, et al., J. Chem. Soc. 2636 (1965) describes the preparation of a large number of compounds, where the aromatic ring is substituted by methoxy, ethoxy, n- or iso-propoxy, or n-, sec- or tert-butoxy group in the 5 or 8 position and the amine is substituted by hydrogen or alkyl groups having 1-4 carbon atoms. The compounds are indicated to be prepared for pharmacological testing. However, no utility or pharmacological activity is yet known for the compounds just mentioned.
EPO Application No.89304935.3 discloses 2-amino 1,2,3,4-tetrahydronapthalenes as selective inhibitors of serotonin reuptake. It has a publication date subsequent to filing date of the parent application of this case.
German Patent DE 2 803 582 describes 2-aminotetralins where the aromatic ring is substituted on the 5,6,7 or 8 position with the group R1, where R1 is hydrogen, alkanoyl having 1 to 20 carbon atoms or a group -CO-(CH2)n-R7, n is a number 0 to 5, R7 is a phenyl group with substituents as defined further, R2 is hydrogen, hydroxy, halogen or alkylsulfonylamino, R3 is hydrogen, R4 is hydrogen, CH2OH, CH2O-CO-R8 or CH2-O-CO-(CH2)n-R7 with further definition and R5 and R6 are hydrogen, alkyl or aryl or aralkyl groups further defined or R5 and R6 are together an alkylene with 4 to 6 carbon atoms. The compounds are disclosed as having pharmacodynamic activity in particular a stimulating effect on alpha- and beta-adrenoceptors and dopamine receptors. Among the compounds described are compounds having the group R10 in the 8 position and having R2 or R4 other than hydrogen.
Great Britain Patent 1,377,356 describes 2-aminotetralins where the aromatic ring
is substituted on the 5, 6,7 or 8 position by R1, where R1 is hydrogen or methyl, the aliphatic ring is substituted by R2, where R2 is alkyl having 1-6 carbon atoms, and the amine is substituted by R3, where R3 is hydrogen or alkyl having 1-6 carbon atoms are described. Such compounds are stated to possess analgesic activity. 1,1-Dimethyl-2- (N,N-dimethylamino)-7-hydroxytetralin is mentioned as one example of a compound covered by the patent. This compound is also described in Chem. Ab., 79: 146294b as having analgesic and intestinal movement accelerating actions.
J. Pharm. Sci., 67, 880-82 (1978) describes the compound 1-methyl-2-(cyclopro- pylamino)-5-methoxytetralinand indicates the compound possess local anesthetic activity.
Derwent documents 58,247B/32, 40 378A/23, 83-729388/32, 83-729387/32,
29348D/17 and 06733V/05 refer to 8-carboxyamino tetralins. Additional 07633V/05 refers to 8-amido and 8-alkylamido tetralin.
EPO patent application EPO 270947 (1988) discloses 8-hydroxy and 8-methoxy- tetralins.
EPO patent application EPO 272 534 (1988) discloses aminotetralins including
8-amido compounds.
The references cited herein are disclosures describing work related to the invention:
Hjorth, S.; Carlsson, A; Lindberg, P.; Sanchez, D.; Wikstron, H.; Arvidsson, L.-E.; Hacksell, U.; Nilsson, J.L.G., J. Neural Transm., 1982, 55, page 169.
Mellin, C; Bjork, L.; Karlen, A.; Johansson, A.M.; Sundell, S.; Kenne, L.; Nelson, D.L.; Anden, N.-E.; Hacksell, U., J. Med. Chem., 1988, 31, page 1130.
Cossery, J.M.; Gozlan, H.; Spampinato, U.; Perdicakis, C; Guillaumet, G.: Pichat, L.; Hamon, M., European J. Pharmacol., 1987, pages 140-143.
SUMMARY OF THE INVENTION
A compound having the formula I
wherein R is hydrogen or halogen,
wherein R1 is
(a) -hydrogen, (k) -CSNR5R6
(b) -OR4, (l) -OSO2CF3
(c) -SR4,
(d) -CON R5R6,
(e) -CN,
(f) -het,
(g) -C(O)het,
(h) -CF3
(i) -SO2NR5 R6
(j) -5-oxazolyl
wherein R2 and R3 are independently
(a) -hydrogen,
(b) -(C1-C8) alkyl,
(c) -(C2-C8) alkenyl,
(d) -(C2-C8) alkynyl,
(e) -(CH2)m-(C3-C8)cycloalkyl,
(f) -(CH2)m-(C3-C8)cycloalkenyl,
(g) -(CH2)m-aryl,
(h) -(CH2)m-CO2R4,
(i) -(CH2)m-OR4,
wherein R4, R5 and R5 are independently
(a) -hydrogen,
(b) -(C1-C4)alkyl,
(c) -(C1-C4)alkenyl,
(d) -(C3-C8)cycloalkyl
and wherein X is (CH2)n
m = 0-4
n = 4-8
p = 0,1
and pharmaceutically acceptable acid addition salts thereof;
with the provisos that when R
1 is hydroxy or methoxy and R
2 is hydrogen cannot
be piperidino, piperazino or homopiperazino.
Selected compounds of this invention possess selective pharmacological properties and are useful in treating central nervous system disorders including anti-depression symptoms, anti-psychotic symptoms, anxiolytic symptoms, panic attacks, obsessive- compulsive disturbances, senile dementia, emotional disturbances related to dementia disorders, and stimulation of sexual activity. Selected compounds of this invention are also useful to alleviate aggressive behavior, confusional delirious states and impotence. Selected compounds of this invention are further useful as anti-diabetic, anti-obesity, anti-hypertensive agents and for treating sexual impotency. Compounds of this invention are also useful as antitussive agents.
Processes for preparation of these compounds, their pharmaceutical use and pharmaceutical preparations employing such compounds constitute further aspects of the invention.
An object of the invention is to provide compounds for therapeutic use, especially compounds having a therapeutic activity in the central nervous system. Another object is to provide compounds having an effect on the 5-HT1A receptor in mammals including man. A further object of this invention is to provide compounds having an effect on the subclass of dopamine receptors known as the D2 receptor.
DETAILED DESCRIPTION OF THE INVENTION
The compounds of this invention are identified in two ways: by the descriptive name and reference to labelled structures contained in appropriate charts. In appropriate situations, the proper stereochemistry is also represented in the charts.
In this document the parenthetical term (Cn-Cm) is inclusive such that a compound of (C1-C8) would include compounds of one to 8 carbons and their isomeric forms. The various carbon moieties are defined as follows: Alkyl refers to an aliphatic hydrocarbon radical and includes branched or unbranched forms such as methyl, ethyl, n-propyl, isopropyl, n-butyl, isobutyl, sec-butyl, t-butyl, n-pentyl, isopentyl, neo-pentyl, n-hexyl, isohexyl, n-heptyl, isoheptyl, and n-octyl.
Alkoxy as represented by -OR1 when R1 is (C1-C8) alkyl refers to an alkyl radical which is attached to the remainder of the molecule by oxygen and includes branched or unbranched forms such as methoxy, ethoxy, n-propoxy, isopropoxy, n-
butoxy, isobutoxy, sec-butoxy, t-butoxy, n-pentoxy, isopentoxy, neo-pentoxy, n-hexoxy, isohexoxy, n-heptoxy, isoheptoxy, and n-octoxy.
Alkenyl refers to a radical of an aliphatic unsaturated hydrocarbon having a double bond and includes both branched and unbranched forms such as ethenyl, 1- methyl-1-ethenyl, 1-propenyl, 2-propenyl, 1-butenyl, 2-butenyl, 3-butenyl, 2-methyl-1- butenyl, 1-pentenyl, allyl, 3-pentenyl, 4-pentenyl, 1-methyl-4-pentenyl, 3-methyl-1- pentenyl, 3-methyl-allyl, 1-hexenyl, 2-hexenyl, 3-hexenyl, 4-hexenyl, 1-methyl-4- hexenyl, 3-methyl-1-hexenyl, 3-methyl-2-hexenyl, 1-heptenyl, 2-heptenyl, 3-heptenyl, 4-heptenyl, 1-methyl-4-heptenyl, 3-methyl- 1-heptenyl, 3-methyl-2-heptenyl, 1-octenyl, 2-octenyl, or 3-octenyl.
Cycloalkyl refers to a radical of a saturated cyclic hydrocarbon such as cyclopropyl, cyclobutyl, cyclopentyl, cyclohexyl, cycloheptyl, or cyclooctyl.
Het refers to a five atom heterocyclic ring containing nitrogen, carbon and in some cases oxygen. It includes 2-pyrrolyl, 2-oxazolyl, 2-imidazolyl, 2-oxazolinyl, 2- imidazolinyl.
Halogen refers to bromine, chlorine or fluorine.
It will be apparent to those skilled in the art that compounds of this invention do contain chiral centers. The scope of this invention includes all enantiomeric or diastereomeric forms of Formula I compounds either in pure form or as mixtures of enantiomers or diastereomers. The compounds of Formula I contain 1-3 asymmetric carbon atoms in the aliphatic ring moiety, including the ring carbon atoms adjacent to the nitrogen atom. The therapeutic properties of the compounds may to a greater or lesser degree depend on the stereochemistry of a particular compound. Pure enantiomers as well as enantiomeric or diastereomeric mixtures are within the scope of the invention.
Both organic and inorganic acids can be employed to form non-toxic pharmaceutically acceptable acid addition salts of the compounds of this invention. Illustrative acids are sulfuric, nitric, phosphoric, hydrochloric, citric, acetic, lactic, tartaric, palmoic, ethanedisulfonic, sulfamic, succinic, cyclohexylsulfamic, fumaric, maleic, and benzoic acid. These salts are readily prepared by methods known in the art.
The compounds of this invention may be obtained by one of the following methods described below and outlined in the appropriate charts.
Chart A
Substituted 2-tetralone A-1, is subjected to reductive amination whose procedures
are known in the art in step 1. In step 2, when R1 is methoxy A-2 may be demethylated via procedure known in the art to yield A-3.
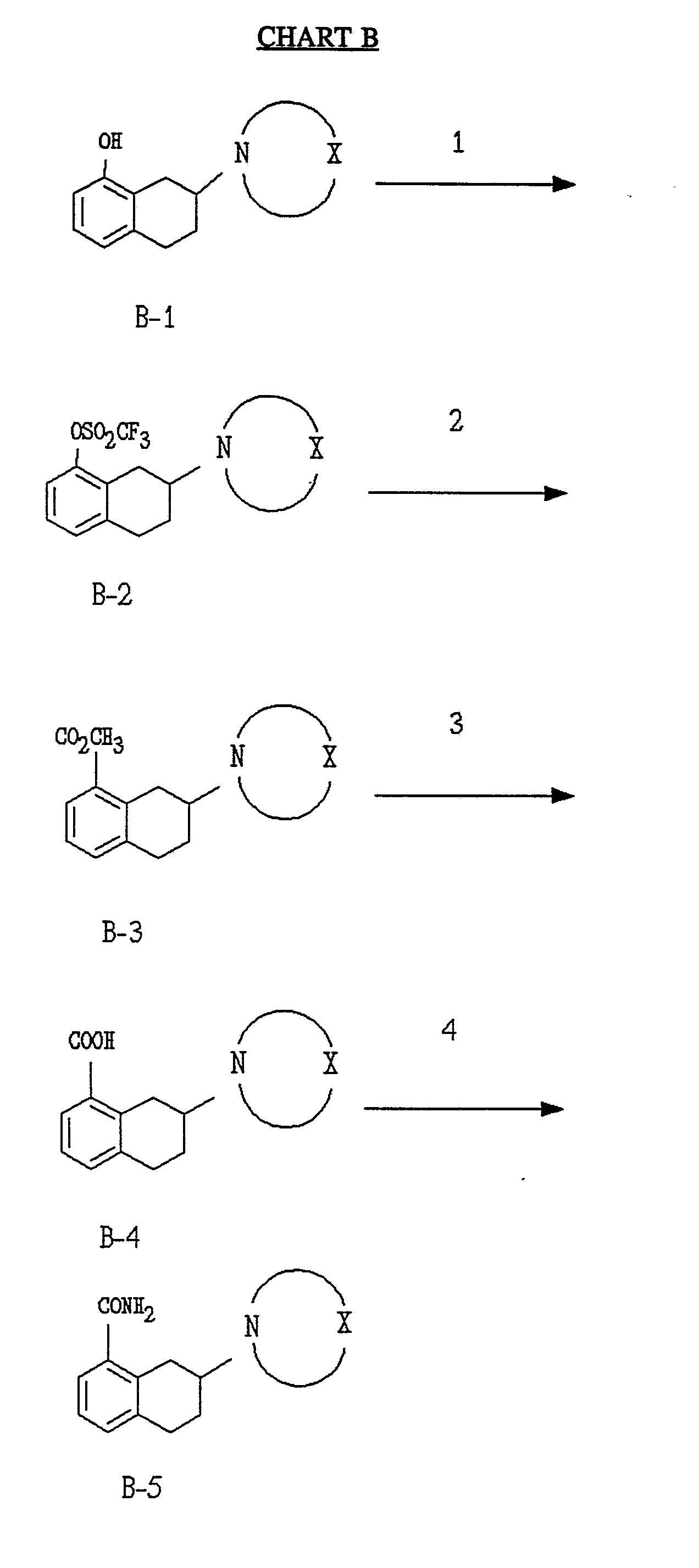
Chart B
In Step 1 of Chart B, the phenol B-1 is reacted with trifluoromethanesulfonic anhydride in the presence of a solvent according to methods well known in the art to yield the triflate B-2. The phenol B-1 can be prepared from the appropriately substituted tetralones by the process depicted in Chart A.
In Step 2, a solution of B-2 in a solvent mixture such as methanol/ DMF is reacted with carbon monoxide gas, palladium acetate, triethylamine and 1,3-bis(diphenylphos- phino) propane to form the carboxylic acid methyl ester B-3.
In Step 3, the methyl ester B-3 is hydrolyzed with sodium hydroxide in methanol. The resulting acid B-4 is then coupled in step 4 with ammonia in the presence of diethylcyanophosphonate and triethylamine in a solvent such as DMF to yield the carboxamide B-5.
Chart C
In step 1, acid chloride C-1 is treated with ethylene in the presence of aluminum trichloride to obtain tetralone C-2. C-2 is reductively aminated with the appropriate amine to yield C-3. C-3 is then dissolved in a solvent such as THF and reacted in the presence of t-butyllithium and trimethylsilyl isocyanate to yield amide C-4.
Chart D
In step 1, ketal D-2 is generated by stirring tetralone D-1 with ethylene glycol in the presence of an acid catalyst. In step 2, the 8-trifluoromethyl compound D-3 is easily obtained by heating a mixture of D-2, copper(I) iodide, sodium trifluoroacetate and N- methylpyrrolodone to 160°C. In step 3, hydrolysis using aqueous acid gives tetralone D-4 which is reductively aminated in step 4 using the known procedure of mixing the appropriate amine acetic acid, and sodium cyanoborohydride to yield D-5.
Chart E
In step 1, substituted 2-tetralone E-1 is alkylated at the 2-position to produce E-2 by reaction with an alkyl halide utilizing base in accordance with alkylation methods well known in the art. In step 2, E-2 is subjected to reductive amination to produce E-3. When R1 is methoxy. In step 3, E-3 is demethylated via procedures well known in the art to yield E-4.
Chart F
In step 1, substituted tetralone F-1 is reacted with dimethylcarbonate in the presence of base such as LDA to produce F-2. In step 2 F-2 is reacted with alkyl halide in the presence of base to produce F-3. In step 3, F-3 is decarboxylated to produce F-4. In step 4, F-4 is subjected to reductive amination to produce F-5. F-5 is demethylated when R1 is methoxy to yield F-6.
Chart G
In step 1, bromotetralone G-1 (C-2) is reductively aminated using typical conditions with the appropriate amine to obtain G-2. In step 2, G-2 is treated with t- butyllithium followed by dimethylformamide to obtain aldehyde G-3. This aldehyde is condensed in step 3 with "TOSMIC" under typical conditions to obtain the oxazole G-4.
Chart H
In step 1, substrate H-1 is treated with t-butyllithium followed by sulfur dioxide to obtain sulfonic acid H-2. In step 2, H-2 is treated with sodium hydride to obtain H-3 followed by treatment with N-chlorosuccinimide in step 3 to obtain sulfonylchloride H-4. Treatment of H-4 with ammonia in step 4 gives H-5 which is hydrolysized with aqueous acid to H-6 in step 5. In step 6, reductive amination using typical conditions using the appropriate amine gives H-7.
Chart I
Bromo compound I-1 (C-3) is treated with t-butyllithium followed by trimethyl- silylisothiocyanate to give 1-2.
Chart J
In addition compounds of J-1 can be converted to compounds of Formula I wherein R1 is arylcarbonyl by the process illustrated in Chart J. In Step 1 a solution of J-1 is reacted with the pyrrole-adduct in a solvent such as toluene in the presence of ethylmagnesium bromide to yield J-2.
In Step 2, J-2 is reacted with an appropriate amine in the presence of acetic acid, platinum oxide and absolute ethanol under a hydrogen atmosphere to yield compound J- 3.
Methods for preparing compounds of Formula I wherein R1 is hydrogen, -OR6, or -SR6 are illustrated by the processes illustrated in Charts A, E and F. In each of these processes, 2-tetralone derivatives are utilized as the starting material.
Methods for conducting reductive amination are well known in the art and any such methods may be used in the procedures described above. One such method
involves reacting the tetralone with an amine in the presence of sodium cyanoboro- hydride and glacial acetic acid in tetrahydrofuran/methanol.
The 8-amido compounds B-5 or C-4 can be converted to the corresponding 8- cyano compounds by reacting with a "Burgess salt" utilizing conditions well known in the art. The Burgess salt can be prepared by the procedure described in Organic Synthesis, 56, page 40.
In clinical practice the compounds of the present invention will normally be administered orally, rectally, or by injection, in the form of pharmaceutical preparations comprising the active ingredient either as a free base or as a pharmaceutically acceptable non-toxic, acid addition salt, such as the hydrochloride, lactate, acetate, sulfamate salt, in association with a pharmaceutically acceptable carrier. The use and administration to a patient to be treated in the clinic would be readily apparent to a person of ordinary skill in the art.
In therapeutical treatment the suitable daily doses of the compounds of the invention are 1-2000 mg for oral application, preferentially 50-500 mg, and 0.1-100 mg for parenteral application, preferentially 0.5-50 mg.
The compounds of this invention where R1 is in the 8 position in the aromatic ring are very selective 5-HT1A receptor agonists having little or no dopaminergic activity. The IC50 ratio of dopamine D2 to 5-HT1A in vitro binding data shown in Table 1 for one compound of this invention, demonstrates the selectivity for the 5-HT1A receptor. The compounds of this invention also have been shown to have high oral potency and a long duration of action. Both these features are beneficial to effective clinical treatment.
The utility of the compounds of this invention to treat central nervous system disorders is shown in behavioral, physiological and biochemical tests. The methods are given as follows:
Binding: Inhibition of 8-OH-DPAT binding in a bovine brain homogenate. Potency is given as nM dose required to inhibit 50% of DPAT binding (IC50). This test measures ability to bind to 5-hydroxytryptamine (5-HT1A) receptor.
Hypothermia: Starting with a dose of 30 mg/kg, four mice are injected subcutaneously with test compound. Twenty minutes later, the number of animals whose body temperature has decreased by 2°C. or more are counted. If all four animals reach criteria, the drug is considered "active", and subsequent readings are taken at 60 and 120
minutes after drug. The time for last statistically significant drug affect on mean body temperature is indicated in minutes. For all "active" compounds, doses are lowered by 0.5 log intervals until a dose which does not lower body temperature by 2°C. in any animal is found. Potency is given as mg/kg ED50 (dose required to depress temperature in two of four mice) as measured by Spearman-Karber statistics.
Sympathetic Nerve Discharge (SND): The i.v. mg/kg dose causing a 50% depression in SND in chloralose anestetized cats and the maximum inhibition of sympathetic activity observed in the dose range tested (0.001-1.0 mg/kg i.v.).
BP SND/MAX: The blood pressure of the chloralose anestetized cats in percent control at the dose causing 50 % depression in SND and the maximum reduction in blood pressure as percent of the control blood pressure in the same animals observed in the dose range tested (0.001-1.0 mg/kg i.v.).
CNS and anti-hypertensive biological data are shown in Tables 1 and 2 respectively.
The compounds of this invention are useful as anti-diabetic, anti-obesity, anti- hypertensive and anti-tussive agents. While all of the compounds do not have all of these pharmacological activities the utility of a particular compound can be determined by one skilled in the art utilizing the following tests.
Anti-diabetic
A. Testing For Blood Glucose Lowering In the KKAy Mouse
All KKAy mice used for screening are produced and selected by methods outlined by T. Fujita et al., Diabetes, 32, pp. 804-10 (1983). The screening is done in groups of six animals per group.
Pre-treatment non-fasting blood glucose (NFBG) samples are measured five days prior to the start of a screening run by previously described methodologies. These blood sugar values are used to place animals into groups with equal mean blood glucose concentrations and to eliminate any mice with a NFBG value <250 mg/dl. On day 0, compounds chosen to be run are incorporated into ground mouse chow (Purina 5015). Compounds are included at a rate of 1 mg/ gram of chow. Generally, 300 g of drugs containing diet is prepared for each group. Mice receiving ground chow only are the negative control.
Each screening run also uses ciglitazone (T. Fujita et al., supra) as a positive control (0.5 to 1.0 mg/ gram chow).
Initial body and food weights are taken on day one. Food is placed in a crock which contains an adequate amount to last for the length of the study. In order to acclimate the mice from pelleted mouse chow to ground mouse chow, they are fed the ground chow for nine days prior to use in the screen. On day four of treatment, a NFBG sample is again measured, as well as food and body weights. Food consumption measurements are used to determine an average mg/kg dose the mice received over the testing period, and to evaluate the compound's effect on food consumption.
Acceptance and activity are determined by the following criteria:
A. Negative Control
This group must not show a significant change (p < .05) from pre- to post- treatment. If there is a significant decrease in blood sugar, the run is not valid.
B. Positive Control
This group must show a significant depression in blood sugar mean levels from pre- to post-treatment. A lack of activity in this group would also invalidate the run.
C. Negative Control vs. Positive Control
This contrast must be significant. It is a further assurance that both control groups performed as expected.
D. Compound
A compound's activity is based on several criteria:
1. A significant decrease in blood sugar mean levels from pre- to post-treatment.
2. Negative control vs. compound: This contrast allows one to determine if these groups are dissimilar, which is required for the compound to be considered active.
II. Anti-obesity Activity
Upjohn Sprague-Dawley rats are housed individually and given food and water ad libitum. Food consumption is measured daily. The animals are orally dosed with 100 mg/kg or 200 mg/kg of the compound in Tween 80. Controls receive an equivalent volume of (0.25) of Tween 80. If the daily food consumption of the treated animals is in the range of 4 grams less than that of the control animals the compound is considered to have anorexic activity.
Experimental Procedures
Without further elaboration, it is believed that one skilled in the art can, using the preceding description, practice the present invention to its fullest extent. The following detailed examples describe how to prepare the various compounds and/or perform the various processes of the invention and are to be construed as merely illustrative, and not limitations of the preceding disclosure in any way whatsoever. Those skilled in the art will promptly recognize appropriate variations from the procedures both as to reactants and as to reaction conditions and techniques.
Preparation 1 7-(1-hexahydroazepinyl)-5,6,7,8-te rahydronaphthalene-1-trifluoromethyl- sulfonate (B-2, Chart B)
A solution of 2.1 g7-(1-hexahydroazepinyl)-5,6,7,8-tetrahydro-1-naphthalenoland
4.06 g pyridine in 100 mL CH2Cl2 was stirred and cooled to 0°C under a nitrogen atmosphere. The trifluoromethanesulfonic anhydride (4.32 g) was added dropwise over a 20 min period. The yellow solution was warmed to room temperature and stirred for 45 min. TLC aliquot showed no starting material present, the reaction was quenched with satd. NaHCO3 to pH > 8. The mixture was extracted with methylene chloride. The organic layers were washed with water, brine, dried (MgSO4), filtered and concentrated to yield an oil. Flash chromatography on 1 kg silica gel eluding with hexane/ethyl acetate (1:1) (collecting 45 mL fractions) yielded a pale yellow
oil (3.05 g, 95%)
Preparation 2 7-(1-Hexahydroazepinyl)-5,6,7,8-tetrahydronaphthalene-1-carboxylicacid methyl ester (B-3, Chart B)
A solution of 2.9 g 7-(1-hexaJιydroazepinyl)-5,6,7,8-tetrahydronaphthalene-1- trifluoromethylsulfonate and 2.13 mL triethyl amine in 9 mL methanol and 27 mL DMF was degassed with nitrogen through a syringe for 10 min. Carbon monoxide was then bubbled through the solution for 10 min. During this time, a solution if 172 mg palladium acetate and 379 mg in 7 mL DMF was dissolved and degassed with nitrogen for 10 min. This solution was added to the reaction , heated to 70°C and carbon monoxide gas bubbled through overnight. An aliquot was treated with satd. NaHCO3 and EtOAc and showed no starting material. Nitrogen was bubbled through the solution and then quenched with satd. NaHCO3. The mixture was extracted with ethyl acetate
(3 X 500 ml) and the combined organic layers were washed with brine, dried (MgSO4), filtered and concentrated to yield an oil. Flash chromatography using 400g silica gel and eluting with hexane/ethyl acetate (1:1) to yield an oil (1.56 g, 71%).
Preparation3 7-(1-Hexahydroazepinyl)-5,6,7,8-tetrahydronaphthalene-1-carboxylicacid
(B-4, Chart B)
A mixture of 1.56 g 7-(1-Hexahydroazepinyl)-5,6,7,8-tetrahydronaphthalene-1- carboxylic acid methyl ester , 1.57 mL 12 N NaOH, 1.57 mL water in 10 mL methanol was refluxed (70-80ºC) overnight. TLC showed no starting material remaining. The mixture was neutralized with 6 N HCl to pH 5-6 and concentrated to dryness using toluene and methanol. A white solid was recovered and used crude.
Preparation 4 8-bromo-2-tetralone (C-2, Chart C)
Substitute 2-bromophenylacetylchloride in the procedure detailed in A.H. Horn, C.J. Grol, D. Dijkstra, and A.H. Mulder, J. Med. Chem. 21, 825 (1978).
Preparation 5 8-bromo-2-(spiro-1,3-dioxolan-2-yl)tet-alin (D-2, Chart D)
8-Bromo-2-tetralone (29g), ethylene glycol (24g), p-toluenesulfonic acid (0.5g), and benzene (250 ml) were heated to reflux with azeotropic removal of water for 16 hr.
The solution was cooled and extracted with aq. sodium carbonate, water, and then brine. The solution was dried over anhydrous sodium sulfate and the solvent removed under vacuum.
Preparation 6 8-trifluoromethyl-2-(spiro-1,3-dioxolan-2-yl)tetralin (D-3, Chart D)
8-Bromo-2-(spiro-1,3-dioxolan-2-yl)tetralin(12.4g),sodiumtrifluoroacetate(255),
copper (I) iodide (17.5g) and N-methylpyrrolidone (368 ml) were heated under nitrogen to 160°C and maintained there for 4 hr. The solution was cooled and ether and hexane were added. The slurry was filtered through diatomaceous earth and the elutant was washed with water (3X) and brine. The solution was dried over anyhdrous sodium sulfate and the solvent removed under vacuum. Flash chromatography was performed, eluting with ether/ hexane (1:9) giving 9.9g of a pure liquid.
Preparation 7 8-trifluoromethyl-2-tetralone (D-4, Chart D)
8-Trifluoromethyl-2-(spiro-1,3-dioxolan-2-yl)tetralin (9.9g), water (15 ml), THF (120 ml), and 2 N aq. HCl (12 ml) were heated to 50°C for 15 hr. This solution was cooled and extracted with ether, washing the organic layer with aq. sodium bicarbonate and then brine. Drying over anhydrous sodium sulfate and solvent removal afforded a clear liquid.
Preparation 8 1,2,3,4-Tetrahydro-2-oxo-1-(2-propenyl)-naphthalene(E-2, Chart E) and 1,2,3,4-Tetrahydro-2-oxo-1,1-di-(2-propenyl)naphthalene
To a solution of 7.3 g (50 mmol) 2-tetralone in 75 mL THF in a 3-neck round- bottomed flask, equipped with a gas inlet and septum, was added 36.7 mL LDA (55 mmol, 1.5 M in cyclohexane) at -30°C under a nitrogen atmosphere. The solution was allowed to warm to 0°C over a 30-minute period and 5.6 mL (65 mmol) allyl bromide was added. TLC analysis was used to monitor the reaction. After stirring for 24 hours at room temperature, the reaction mixture was quenched with 10% sodium bisulfate to pH 2-3. After removal of THF under reduced pressure, the mixture was extracted with ethyl acetate (2 X 1 L) and the combined organic layers were washed with brine, dried (MgSO4), filtered and concentrated in vacuo. The crude product was purified by liquid chromatography on 800 g of silica gel 60 (230-400 m), eluting with 1 L of hexane, followed by 5 L of 5% ethyl acetate/hexane, and collecting 40 mL fractions. Fractions 65-82 gave 3.1 g (33%) of pure 1,2,3,4-Tetrahydro-2-oxo-1-(2-propenyl)-naphthalene as a light yellow oil.
1HNMR (CDCl3, TMS): 7.27-7.16 (m, 4H); 5.81-4.95 (m, 3H); (s, 3H); 3.54- 2.45 (m, 7H).
IR (film): v max 1717, 1640 and 1582 cm-1.
MS: M+ 186, other ions at m/z 168, 145, 128, 117.
TLC (Silica Gel GF): Rf = 0.51 in hexane/ethyl acetate (4:1).
on.
Fractions 41-64 gave 4.2 g (37%) of pure and 1,2,3,4-Tetrahydro-2-oxo-1,1-di-(2- propenyl)naphthalene as a colorless oil
Preparation 9 1,2,3,4-Tetrahydro-8-methoxy-2-oxo-1-(2-propenyl)-naphthalene and 1,2,3,4-Tetrahydro-8-methoxy-2-oxo-1-di-(2-propenyl)-naphthalene(E-2, Chart E) To a solution of 8.8 g (50 mmol) 8-methoxy-2-tetralone in 250 mL THF in a three-neck round-bottomed flask, equipped with a gas inlet and septum, was added 40 mL LDA (60 mmol, 1.5 M in cyclohexane, at -30°C under a nitrogen atmosphere. The solution was allowed to warm to 0°C over a 30-minute period and 6.5 mL (75 mmol) allylbromide was added. TLC analysis was used to monitor the reaction. After stirring the mixture at room temperature for three hours and at 40°C for one hour, the reaction mixture was quenched with 10% sodium bisulfate to pH 2-3. After removal of THF under reduced pressure, the mixture was extracted with ethyl acetate (2 X 1 L) and the combined organic layers were washed with brine, dried (MgSO4), filtered and concentrated in vacuo. The resulting oil (about 3b/22b=4 by LC purification in a small scale run) was used without purification in the next step. For the analytical purpose the small amount of the crude product (< 1 g) was purified by liquid chromatography on 185 g of silica gel 60(230-400 m), eluting with hexane/ acetone (19:1). Fractions homogeneous by TLC were combined and concentrated in vacuo. Pure title compounds were isolated as a light yellow oil.
Physical data for 1,2,3,4-tetrahydro-8-methoxy-2-oxo-2-(2-propenyl)naphthlene:
1HNMR (CDCl3, TMS): 7.21-6.76 (m, 3H); 5.73-4.87 (m. 3H); 3.82 (s, 3H); 3.88-3.82 (m, 1H); 3.32-2.43 (m, 6H).
IR (film): v max 1712,1640, 1586 cm-1.
MS: Calcd for C14H16O2: 216.1150.
Found: 216.1151.
Analysis: Calcd for C14H16O2: C, 77.75; H, 7.46.
Found: C, 77.56; H, 7.68.
TLC (Silica Gel GF): Rf = 0.32 in hexane/acetone (4:1)
Physical data for 1,2,3,4-tetrahydro-8-methoxy-2-oxo-1-di-(2-propenyl)- naphthlene:
1HNMR (CDCI3, TMS): 7.22-6.73 (m, 3H); 5.44-4.77 (m, 6H); 3.85 (s, 3H); 4.0-2.52 (m, 8H).
IR (film): v max 1712, 1639 and 1582 cm-1.
MS: Calcd for C17H20O2: 256.1463.
Found: 256.1470
Analysis: Calcd for C17H20O2: C, 79.65; H, 7.86.
Found: C, 79.56; H, 8.29.
TLC (Silica Gel GF): Rf=0.46 in hexane/acetone (19:1).
Preparation 10 1,2,3,4-Tetrahydro-5-methoxy-2-oxo-1-(2-propenyl)-naphthalene (E-2, Chart E) and 1,2,3,4-Tetrahydro-5-methoxy-2-oxo-1,1-di-(2-pro-penyl)-naρhthalene
To a solution of 5.3 g (30 mmol) 5-methoxy-2-tetralone in 45 mL THF in a three-neck round-bottomed flask, equipped with a gas inlet and septum, was added 22 mL LDA (33 mmol, 1.5 M in cyclohexane, at -30°C under a nitrogen atmosphere. The solution was allowed to warm to 0°C over a thirty-minute period and 3.4 mL (39 mmol) allylbromide was added. TLC analysis was used to monitor the reaction. After five hours of stirring, the reaction mixture was quenched with 10% sodium bisulfate to pH
2-3. After removal of THF under reduced pressure, the mixture was extracted with ethyl acetate (2 X 1 L) and the combined organic layers were washed with brine, dried
(MgSO4), filtered and concentrated in vacuo. The resulting oil was purified by liquid chromatography on 800 g of silica gel 60 (230-400 m), eluting with 1 L of hexane and
5 L of hexane-ethyl acetate (19: 1), and collecting 40 mL fractions. Fractions 45-87 gave
2.5 g (32.5%) of pure 1,2,3,4-tetrahydro-5-methoxy-2-oxo-1,1-di-(2-propenyl)- naphthalene as a near colorless oil and fractions 88-140 gave 1.07 g (16.5%) of pure
1,2,3,4-tetrahydro-5-methoxy-2-oxo-1-(2-propenyl)naphthaleneas a light yellow oil.
1HNMR (CDCl3, TMS): 7.23-6.77 (m, 3H); 5.75-4.97 (m, 3H); 3.85 (s, 3H); 3.52-2.49 (m, 7H).
IR (film): v max 1717, 1641 and 1586 cm-1.
MS: M+ 216, other ions at m/z 175, 159, 147.
TLC (Silica Gel GF): Rf = 0.42 in hexane-ethyl acetate (4:1).
Preparation 11 1,2,3,4-Tetrahydro-8-methoxy-1-(cyclopropylmethyl)-2-oxo-naphthalene (E-2, Chart E)
To a solution of 3.52 g (20 mmol) 8-methoxy-2-tetralone in 50 mL THF in a three-neck round-bottomed flask, equipped with a gas inlet and septum, was added 14.3 mL LDA (22 mmol, 1.5 M in cyclohexane) at -30°C under a nitrogen atmosphere. The solution was allowed to warm to 0°C over a thirty-minute period and 2.4 mL (24 mmol) allyl bromide was added. TLC analysis was used to monitor the reaction. After stirring
for two hours, the TLC analysis appeared to show little progress. The reaction mixture was therefore treated with 1.1 mL (12 mmol) allylbromide and the mixture was heated to reflux for 72 hours. The reaction mixture was quenched with 10% sodium bisulfate to pH 2-3. After removal of THF under reduced pressure, the mixture was extracted with ethyl acetate (2x 1 L) and the combined organic layers were washed with brine, dried (MgSO4), filtered and concentrated in vacuo. The resulting oil was purified by liquid chromatography on 400 g of silica gel 60 (230-400 m), eluting with hexane/acetone (9:1) and collecting 40 mL fractions. Fractions homogeneous by TLC were combined and concentrated in vacuo to give 4.5 g (97.8%) of pure title compound as a near colorless oil.
1HNMR (CDCl3, TMS): 7.20-6.75 (m, 3H); 3.91 (t, J=7 Hz, 1H); 3.81 (s, 3H); 3.33-1.62 (m, 6H); 0.64-0.09 (m, 5H).
IR (film): v max 1711 cm-1.
MS: Calcd for C15H18O2: 230.1307.
Found: 230.1290.
Analysis: Calcd for C15H18O2: C, 78.23; H, 7.88.
Found: C, 77.93; H, 8.06.
TLC (Silica Gel GF): Rf = 0.46 in hexane-acetone (4:1).
Preparation 12 1,2,3,4-Tetrahydro-8-methoxy-2-oxo-1-naphthalene-carboxylic Acid Methyl Ester (F-2, Chart F)
To a solution of 17.6 g (0.1 mol) 8-methoxy-2-tetralone in 200 mL THF in a three-neck round-bottomed flask, equipped with a gas inlet and septum, was added 86.7 mL LDA (0.13 mol, 1.5 M in cyclohexane) at -30° C under a nitrogen atmosphere. The solution was allowed to warm to 0°C over a thirty-minute period and 84.3 mL (1.0 mol) dimetiiylcarbonate was added. After refluxing for 24 hours (bath temperature 70°C), the TLC analysis indicated no starting material remaining. The reaction mixture was quenched with 1 N HCl to pH 2-3. After removal of THF under reduced pressure, the mixture was extracted with methylene chloride (2 X 1 L) and the combined organic layers were washed with brine, dried (MgSO4), filtered and concentrated in vacuo. The resulting oil was purified by flash chromatography on 1 Kg of silica gel 60 (230-400 m), eluting with 1 L hexane, 2 L 10%, 8 L 20% ethyl acetate/hexane and collecting 500 mL fractions. Fractions 7-9 gave 0.5 g (2%) of a yellow oil which was shown to be 1,1- dicarbomethoxy product by 1HNMR. Fractions 11-22 afforded 21.1 g (90%) of pure
title compound as a yellow oil.
1HNMR (CDCI3, TMS): 7.28-6.77 (m, 3H); 4.72 (s, 1H); 3.80 (s, 3H); 3.72- 2.17 (m, 7H).
IR (film): v max 1750, 1718 and 1588 cm-1.
MS: M+ 234, other ions at m/z 202, 191, 174, 147, 131, 115, 103, 91.
Analysis: Calcd for C13H14O4: C, 66.65; H, 6.02.
Found: C, 66.49; H, 5.93.
TLC (Silica Gel GF): Rf = 0.33 in hexane/ethyl acetate (3:1).
Preparation 13 1,2,3,4-Tetrahydro-8-methoxy-2-oxo-3-(2-propenyl)-1-naphthalene- carboxylic acid methyl ester (F-3, Chart F)
A solution of 10.2 g (43.5 mmol) 1,2,3,4-tetrahydro-8-methoxy-2-oxo-1- naphthalene carboxylic acid methyl ester in 108 mL of THF in a three-neck, round- bottomed flask, equipped with a dropping funnel, was added dropwise 63.8 mL (95.7 mmol) of LDA (1.5 M in cyclohexane) at -30°C to -40°C under a nitrogen atmosphere. The solution was allowed to warm to 0°C and 6.0 mL (69.6 mmol) of allylbromide was added. After stirring the mixture for one hour at room temperature, TLC analysis showed no starting material remaining. The reaction was quenched with 3N hydrochloric acid to pH 2-3 and extracted with ethyl acetate (2x 1 L). The combined organic layers were washed with brine, dried (MgSO4), filtered, and concentrated in vacuo. The resulting oil was purified by liquid chromatography on 800 g silica gel 60 (230-400 m), eluting with hexane-acetone (3:1), and collecting 40 mL fractions. Fractions 36-63 gave 10.3 g (87 %) of pure title compound as a yellow oil.
1HNMR (CDCI3, TMS): 7.27-6.76 (m, 3H); 5.89-5.02 (m, 3H); 4.75, 4.59 (two s, 1H); 3.80, 3.81 (two s, 6H); 3.32-1.64 (m, 5H).
IR (film): v max 1751, 1717 and 1589 cm-1.
MS: M+ 274, other ions at m/z 242, 233, 214, 201, 187, 173, 159, 145.
Analysis: Calcd for C16H18O4: C, 70.05; H, 6.61.
Found: C, 69.73; H, 6.65.
TLC (Silica Gel GF): Rf = 0.34 in hexane-ethyl acetate (3:1).
Preparation 14 1,2,3,4-Tetrahydro-8-methoxy-3-(2-propenyl)-2-oxo-naphthalene (F-4, Chart F)
To a solution of 10.3 g (37.6 mmol) of 1,2,3,4-tetrahydro-8-methoxy-2-oxo-3-(2- propenyl)-1-naphthalene carboxylic acid methyl ester in 26.3 mL of DMSO and 1.1 mL
of water was added 1.9 g (45.1 mmol) of lithium chloride. The reaction mixture was heated at 125 °C (bath temperature) for five hours. TLC analysis showed no starting material remaining. The mixture was cooled to room temperature and extracted with ethyl acetate (1 L). The organic layer was washed with 10% aqueous calcium sulfate (an efficient way of removing DMSO from organic layer), dried (MgSO4), filtered and concentrated in vacuo. The crude product was purified by liquid chromatography on 800 g silica gel 60 (230-400 m), eluting with hexane-ethyl acetate (3:1), and collecting 40 mL fractions. Fractions 26-53 gave 7.65 g (94%) of pure title compound as a yellow oil.
1HNMR (CDCl3, TMS): 7.18-6.74 (m, 3H); 5.95-4.95 (m, 3H); 3.82 (s, 3H);
3.70-2.08 (m, 7H).
IR (film): v max 1756, 1710 and 1589 cm-1.
MS: M+ 216,other ions at m/z 185, 174, 159, 146, 134, 115, 104.
Analysis: Calcd for C14H16O2: C, 77.75; H, 7.46.
Found: C, 77.21; H, 7.65.
TLC (Silica Gel Gf): Rf = 0.53 in hexane-ethyl acetate (3:1).
Preparation 15 8-A minosulfonyl-2-(spiro-1,3-dioxola n-2-yl)tetralin. (H-2, Chart H)
Magnesium (3.83 g, 0.158 mol) was covered with dry tetrahydrofuran (250 mls), and 8-bromo-2-(spiro-1,3-dioxolan-2-yl)tetralin (28.29 g, 0.105 mol) was added. A few crystals of iodine were added, and the mixture was heated to reflux on the steam bath until the reaction became exothermic. The reaction was stirred at ambient temperature until the reaction subsided. The reaction mixture was refluxed gently on the steam bath for an additonal 40 minutes. The Grignard solution was removed from the excess magnesium via needle stock and cooled to -15°C. Sulfur dioxide gas was bubbled through the solution for 30 minutes. The mixture was diluted with diethylether and washed with dilute hydrochloric acid and brine containing sodium bicarbonate. The solution was dried (MgSO4), and the solvent was removed under vacuum to leave the sulfinic acid as an off-white solid (27.26 g).
(H-3, Chart H) Sodium hydride (5.3 g, 50% in oil, 0.11 mol) was washed twice with hexane, and covered with dry tetrahydrofuran (400 mis). A solution of the sulfinic acid (26.38 g, 0.104 mol) in dry tetrahydrofuran (300 mls) was added via needle stock.
The mixture was stirred at room temperature overnight and then heated at reflux for 15 minutes. The mixture was diluted with diethylether, and the precipitate was filtered
while blowing argon over the surface of the compound. The compound was washed several times with diethylether, and dried under vacuum leaving the sodium sulfinate as a solid (26.77 g).
(H-4, Chart H) A suspension of the sodium sulfinate (26.77 g, 0.0969 mol) in methylene chloride (400 mls) was cooled in ice, and N-chlorosuccinimide (13.75 g, 0.103 mol) was added. The mixture was stirred at room temperature for 2 hours. Diethylether was added, and the mixture was washed with water and brine. The solution was dried (MgSO4), and the solvent was removed under vacuum to leave the sulfonyl chloride as an amber solid (23.3 g).
(H-5, Chart H) A solution of the sulfonyl chloride (23.3 g) in tetrahydrofuran (80 mis) was added to an ice-cooled solution of ammonium hydroxide (100 mls) in acetone (500 mls). The cold bath was removed, and the mixture was stirred for 2 hours. The solvent was evaporated, and the residue was partioned between 4:1 diethylether/ tetrahydrofuran and brine. The solution was washed twice with 2% hydrochloric acid, sat. sodium bicarbonate, and brine. The solution was dried (MgSO4), and the solvent was removed under vacuum to leave the sulfonamide as a tan solid (19.8 g). A sample (0.75 g) was crystallized from ethyl acetate/hexane to give off-white crystals of the sulfonamide (0.68 g, m.p. 127-128°C).
Preparation 16 8-Aminosulfonyl-2-tetralone (H-6, Chart H).
8-Aminosulfonyl-2-(spiro-1,3-dioxolan-2-yl)tetralin (18.36 g, 0.0682 mol) was dissolved in acetone (400 mis), and p-toluenesulfonic acid (1.85 g, 9.7 mmol, 14 mol percent) was added. The mixture was stirred at room temperature for 21 hours. Saturated sodium bicarbonate (50 mls) was added, and the solvent was removed under vacuum. The residue was diluted with water and cooled in ice. The precipitate was filtered, washed with water, and dried under vacuum. The compound was boiled in ethyl acetate (350-400 mls) until most of the solid dissolved and then filtered. Hexane was added, and crystallization occurred leaving the ketone as an orange solid (10.34 g, m.p. 173-175°C).
Preparation 17 8-Carbomethoxy-2-[spiro-2-(1,3-dioxolyl)]tetralin (J-1, Chart J)
8-Trifluoromethylsulfonyl-2-[spiro-2-(1,3-dioxolyl)]tetralin (62 g, 183.4 mmol) was placed in a round bottom flask with palladium acetate (2.88 g, 7 mol%), bis(diphenylphosphino)propane (6.81 g, 9 mol%), diisopropylamine (70.3 ml, 2.2 eq.), methanol (183 ml) and dimethylsulfoxide (550 ml). The flask was thoroughly flushed with carbon monoxide, which was subsequently bubbled through the solution. The solution was heated to 70° and stirred for four hours. The solution was cooled and 400 ml. of methylene chloride and 800 ml of ether added. This solution was washed with water (4 x 500 ml) and brine (400 ml), and dried over anhydrous sodium sulfate. Solvent removal in vacuo and filtration through a plug of flash silica gel (6 cm x 30 cm) with ethyl acetate/hexane (25:75), followed by solvent removal afforded 39 g of the title compound (85% yield) as an oil.
Preparation 18 (1,2,3,4-Tetrahydro-2-oxonaphthalene-8-yl)(2-pyrrolyl) ketone (J-2, Chart J)
Pyrrole (3.17 ml) was dissolved in toluene (40 ml) and cooled to 0° while ethylmagnesium bromide (15.2 ml of a 3 M solution in ether) was added. This solution was allowed to warm to 25° and stirred for 30 minutes. A solution of methyl 1,2,3,4- tetrahydrospiro-2-[2-(1,3-dioxolane)]naphthalene-8-yl-carboxylate (5.15 g, 20.8 mmol) dissolved in toluene (20 ml) was added and the solution refluxed for 24 hours. The solution was cooled and quenched by the addition of saturated aqueous ammonium chloride. Ether (100 ml) was added and the solution extracted. The organic layer was washed with water (2 x 100 ml), saturated aqueous sodium bicarbonate (50 ml) and brine (50 ml). Drying over anhydrous sodium sulfate and solvent removal in vacuo afforded a dark oil. This was placed in a solution of acetic acid/THF/water (3:1:1) and heated to 50° for five hours. After cooling, the solvent was removed in vacuo and the residue placed on a flash silica gel column (3 cm x 40 cm) and eluted with ethyl acetate/hexane (40:60) (added methylene chloride to dissolve crystallized product off the column. Solvent removal afforded 3.7 g of the title compound (74% yield) as light yellow needles (m.p. 174°C).
Example 1 -Octahydro-1-(1,2,3,4- tetrahydro-8-methoxy-2-naphthalenyl)-azocine hydrochloride (A-2, Chart A).
To a solution of 1.76 g (10 mmol) 8-methoxy tetralone and 5.66 g (50 mmol) hepta m ethylenamine in 30 mL MeOH/THF (1:1) was added HOAc dropwise to adjust
the pH to 4-5. The reaction mixture stirred for 15 minutes under N2, then 1.26 g (20 mmol) NaCNBH3 was added. When the reaction was complete by TLC (24 h), 1 N NaOH (25 mL) and H2O (200 mL) was added to quench the reaction. The solution was extracted with CH2Cl2 (2 X 500 mL) and the combined organic layers were washed with brine, dried (MgSO4), filtered and concentrated in vacuo. The resulting was purified by liquid chromatography on 400 g of silica gel 60 (230-400m), eluting with hexane/acetone (5:1). Fractions homogeneous by TLC were combined and concentrated in vacuo to give pure compound as an oil. The HCl salt was formed by using a MeOH/HCl solution. The title compound was recovered as a white solid by recrystallization using EtOAc/MeOH (2.65 g, 86%): mp. 211-213°C.
1HNMR (CDCl3, TMS): 7.13(t, 1H); 6.69 (t, 2H); 3.81 (s, 3H); 3.54 (m, 3H), 3.31-3.18 (m, 3H); 2.94-2.67 (m, 4H); 2.17 (m, 2H); 2.1-1.46 (m, 8H).
IR (mull): v max 2529, 2503, 1585, 1470, 1463, 1453, 1251 cm-1.
Analysis: Calcd for C18H27NOΗCl: C, 69.769; H, 9.108; N, 4.521.
Found: C, 69.6; H, 9.24; N, 4.65.
utilizing a procedure similar to that of Example 1 but substituting the appropriate starting materials there is obtained
Hexahydro-1-(1,2,3,4-tetrahydro-8-methoxy-2-naphthalenyl)-1H-azepine hydrochloride white solid: m.p. 236°-237°C.
1HNMR (CDCI3, TMS): 7.12-7.04 (t, 1H); 6.71-6.64 (q, 2H); 3.81 (s, 3H);
3.03-2.76 (m, 7H); 2.45 (q, 1H); 2.0 (m, 1H); 1.62 (m, 10H).
IR (mull): v m a x 3000, 2600, 2550, 1590, 1480 cm-1.
Analysis: Calcd for C17H25NO·HCl: C, 69.017; H, 8.859; N, 4.735.
Found: C, 68.91; H, 9.04; N, 4.67. Hexahydro-1-(1,2,3,4-tetrahydro-2-naphthalenyl)-1H-Azepine
Hydrochloride as a white solid: m.p. 243-245°C - Compound 1-3.
Example 2 (1-Hexahydroazepinyl)-5,6,7,8-tetrahydronaphthalene-1-carboxamide (B-5, Chart B) - Compound 2-1.
Gaseous ammonia was bubbled through a solution of 7-(1-Hexahydroazepinyl)- 5,6,7,8-tetrahydronaphthalene-1-carboxylic acid and 2.25 mL Triethylamine in 30 mL
DMF. After 10 min, 1.75 mL diethylcyanophosphonate was added and the solution was stirred overnight with ammonia bubbling through the solution. Direct spot TLC showed no starting material remaining. The reaction mixture was evaporated and dissolved in
10 mL methanol. This mixture was flash chromatographed on 200 g silica gel eluting with chloroform first, followed by chloroform/4M NH3 in methanol (95:5). Homogenous samples were combined and concentrated to yield 1 g of a solid. Crystallized from acetone. (Mpt.= 141°C)
Example 3 ( + )-1,2,3,4-tetrahydro-2-pyrrolidino-8-bromo-tetralin (C-3, Chart C) - Compound 3-1.
8-Bromo-2-tetralone (40 g., 177 mmol) and pyrrolidine (3 eq) were dissolved in methanol (350 mL) with bromocresol green as an indicator. Hydrochloric acid (3 eq) in methanol was added. Pyrrolidine (2 eq) was added and the solution stirred at 0°C for 30 minutes. Sodium cyanoborohydride (2 eq) was added and the solution stirred for another hour at 0°C. It was stirred at room temp for an hour then the solvents were removed under vacuum. Ether was added and sodium carbonate (sat'd, aq). The ether layer was washed with brine, dried over sodium sulfate, and stripped of solvent under vacuum. After chromatography, the yield of pure oil was 29 g, 58.5%. The racemate (21.6 g) and D-tartaric acid (1 eq) were dissolved in acetonitrile (1800 mL) and methanol (50 mL). The resulting white solid was recrystallized from acetonitrile until a constant optical rotation was achieved for the salt, [α]25= +34.07°, 165.0-166.5°C mp. Yield of the title compound as an oil was 2 g, = +61.27°.
Example 4 (+)-1,2,3,4-tetrahydro-2-py rrolidino-naphthalene-8-yl-carboxamide (C-4,
Chart C) - Compound 4-1.
(+)-8-Bromo-2-pyrrolidinotetralin (1.84 g, 6.57 mmol) was dissolved in tetrahydrofuran (13 mL) and cooled to -78°C. t-Butyllithium (1.7 M in hexane, 2.1 eq) was added. After 10 min, trimethylsilylisocyanate (2.5 eq) was added. After 10 min, the bath was removed and the solution allowed to warm to room temp. Sodium carbonate (sat'd, aq) was added and the product extracted with ether/chloroform. The organic layer was washed with water, brine, and then was dried over sodium sulfate.
It was stripped of solvent under vacuum to yield white solid. Recrystallization from acetonitrile gave 0.63 g (39.3%) of the hydrochloride of the title compound as white needles, 220.0- 224.0 °C, [α]
25 = +90.62° (228.5-230.5 mp for the hydrochloride
salt).
Utilizing a procedure similar to that of Example 4, but substituting the appropriate bromotetralin there is obtained
(-)-1,2,3,4-te rahydro-2-pyrrolidino-naphthalene-8-yl-carboxamide (mp 228.5-
230.5°C)
(+)-1,2,3,4-tetrahydro-2-(1-piperidinyl)-naphthalene-8-yl-carboxamide (mp 229°C)
(-)-1,2,3,4-tetrahydro-2-(1-piperidinyl)-naphthalene-8-yl-carboxamide
(mp 229°C).
The compound (+)1,2,3,4-tetrahydro-2-(1-pyrrolidinyl)naphthalene-8-yl- carboxamide and its salts, processes for preparing said compounds and methods of employing said compounds in therapy represent the best mode of carrying out the invention.