US7253174B2 - N-heterocyclic inhibitors of TNF-α expression - Google Patents
N-heterocyclic inhibitors of TNF-α expression Download PDFInfo
- Publication number
- US7253174B2 US7253174B2 US11/485,075 US48507506A US7253174B2 US 7253174 B2 US7253174 B2 US 7253174B2 US 48507506 A US48507506 A US 48507506A US 7253174 B2 US7253174 B2 US 7253174B2
- Authority
- US
- United States
- Prior art keywords
- alkyl
- methyl
- dimethyl
- substituted
- mmol
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Expired - Lifetime
Links
- 108060008682 Tumor Necrosis Factor Proteins 0.000 title claims abstract description 47
- MZOFCQQQCNRIBI-VMXHOPILSA-N (3s)-4-[[(2s)-1-[[(2s)-1-[[(1s)-1-carboxy-2-hydroxyethyl]amino]-4-methyl-1-oxopentan-2-yl]amino]-5-(diaminomethylideneamino)-1-oxopentan-2-yl]amino]-3-[[2-[[(2s)-2,6-diaminohexanoyl]amino]acetyl]amino]-4-oxobutanoic acid Chemical compound OC[C@@H](C(O)=O)NC(=O)[C@H](CC(C)C)NC(=O)[C@H](CCCN=C(N)N)NC(=O)[C@H](CC(O)=O)NC(=O)CNC(=O)[C@@H](N)CCCCN MZOFCQQQCNRIBI-VMXHOPILSA-N 0.000 title claims abstract description 43
- 102000000852 Tumor Necrosis Factor-alpha Human genes 0.000 title claims abstract description 6
- 239000003112 inhibitor Substances 0.000 title description 7
- 150000001875 compounds Chemical class 0.000 claims abstract description 74
- 238000000034 method Methods 0.000 claims abstract description 38
- 239000008194 pharmaceutical composition Substances 0.000 claims abstract description 16
- 239000001257 hydrogen Substances 0.000 claims description 71
- 229910052739 hydrogen Inorganic materials 0.000 claims description 71
- 125000004435 hydrogen atom Chemical class [H]* 0.000 claims description 67
- 125000000217 alkyl group Chemical group 0.000 claims description 64
- 125000000623 heterocyclic group Chemical group 0.000 claims description 47
- 239000000203 mixture Substances 0.000 claims description 43
- -1 substituted Chemical class 0.000 claims description 40
- 208000037265 diseases, disorders, signs and symptoms Diseases 0.000 claims description 37
- 150000003839 salts Chemical class 0.000 claims description 33
- 125000000547 substituted alkyl group Chemical group 0.000 claims description 31
- 125000003545 alkoxy group Chemical group 0.000 claims description 27
- 229910052736 halogen Inorganic materials 0.000 claims description 27
- 150000002367 halogens Chemical class 0.000 claims description 27
- HBAQYPYDRFILMT-UHFFFAOYSA-N 8-[3-(1-cyclopropylpyrazol-4-yl)-1H-pyrazolo[4,3-d]pyrimidin-5-yl]-3-methyl-3,8-diazabicyclo[3.2.1]octan-2-one Chemical class C1(CC1)N1N=CC(=C1)C1=NNC2=C1N=C(N=C2)N1C2C(N(CC1CC2)C)=O HBAQYPYDRFILMT-UHFFFAOYSA-N 0.000 claims description 24
- 125000002496 methyl group Chemical group [H]C([H])([H])* 0.000 claims description 19
- 125000003118 aryl group Chemical group 0.000 claims description 18
- 208000035475 disorder Diseases 0.000 claims description 18
- 230000001404 mediated effect Effects 0.000 claims description 18
- 125000000753 cycloalkyl group Chemical group 0.000 claims description 16
- 238000011282 treatment Methods 0.000 claims description 16
- 241000124008 Mammalia Species 0.000 claims description 15
- 229910052794 bromium Inorganic materials 0.000 claims description 14
- 125000003107 substituted aryl group Chemical group 0.000 claims description 14
- 125000004093 cyano group Chemical group *C#N 0.000 claims description 13
- RAXXELZNTBOGNW-UHFFFAOYSA-N imidazole Chemical group C1=CNC=N1 RAXXELZNTBOGNW-UHFFFAOYSA-N 0.000 claims description 12
- 201000004681 Psoriasis Diseases 0.000 claims description 11
- 206010039073 rheumatoid arthritis Diseases 0.000 claims description 11
- UFHFLCQGNIYNRP-UHFFFAOYSA-N Hydrogen Chemical compound [H][H] UFHFLCQGNIYNRP-UHFFFAOYSA-N 0.000 claims description 10
- 125000005346 substituted cycloalkyl group Chemical group 0.000 claims description 10
- 229910052801 chlorine Inorganic materials 0.000 claims description 9
- 125000006416 CBr Chemical group BrC* 0.000 claims description 8
- 125000006414 CCl Chemical group ClC* 0.000 claims description 8
- 201000008482 osteoarthritis Diseases 0.000 claims description 8
- 208000001132 Osteoporosis Diseases 0.000 claims description 7
- 125000000956 methoxy group Chemical group [H]C([H])([H])O* 0.000 claims description 7
- 239000004480 active ingredient Substances 0.000 claims description 5
- 208000011231 Crohn disease Diseases 0.000 claims description 4
- FZWLAAWBMGSTSO-UHFFFAOYSA-N Thiazole Chemical group C1=CSC=N1 FZWLAAWBMGSTSO-UHFFFAOYSA-N 0.000 claims description 4
- 125000001495 ethyl group Chemical group [H]C([H])([H])C([H])([H])* 0.000 claims description 4
- 239000003937 drug carrier Substances 0.000 claims description 3
- WCPAKWJPBJAGKN-UHFFFAOYSA-N oxadiazole Chemical group C1=CON=N1 WCPAKWJPBJAGKN-UHFFFAOYSA-N 0.000 claims description 3
- 125000001425 triazolyl group Chemical group 0.000 claims description 2
- 102000002574 p38 Mitogen-Activated Protein Kinases Human genes 0.000 abstract description 34
- 108010068338 p38 Mitogen-Activated Protein Kinases Proteins 0.000 abstract description 34
- 230000000694 effects Effects 0.000 abstract description 15
- 230000005764 inhibitory process Effects 0.000 abstract description 7
- 238000004519 manufacturing process Methods 0.000 abstract description 7
- 230000016396 cytokine production Effects 0.000 abstract description 4
- XEKOWRVHYACXOJ-UHFFFAOYSA-N Ethyl acetate Chemical compound CCOC(C)=O XEKOWRVHYACXOJ-UHFFFAOYSA-N 0.000 description 94
- YMWUJEATGCHHMB-UHFFFAOYSA-N Dichloromethane Chemical compound ClCCl YMWUJEATGCHHMB-UHFFFAOYSA-N 0.000 description 70
- 239000000243 solution Substances 0.000 description 70
- 239000000047 product Substances 0.000 description 63
- 230000015572 biosynthetic process Effects 0.000 description 56
- 238000003786 synthesis reaction Methods 0.000 description 56
- IAZDPXIOMUYVGZ-UHFFFAOYSA-N Dimethylsulphoxide Chemical compound CS(C)=O IAZDPXIOMUYVGZ-UHFFFAOYSA-N 0.000 description 50
- 102100040247 Tumor necrosis factor Human genes 0.000 description 46
- XLYOFNOQVPJJNP-UHFFFAOYSA-N water Substances O XLYOFNOQVPJJNP-UHFFFAOYSA-N 0.000 description 39
- JGFZNNIVVJXRND-UHFFFAOYSA-N N,N-Diisopropylethylamine (DIPEA) Chemical compound CCN(C(C)C)C(C)C JGFZNNIVVJXRND-UHFFFAOYSA-N 0.000 description 37
- 235000019439 ethyl acetate Nutrition 0.000 description 32
- WYURNTSHIVDZCO-UHFFFAOYSA-N Tetrahydrofuran Chemical compound C1CCOC1 WYURNTSHIVDZCO-UHFFFAOYSA-N 0.000 description 30
- 239000012044 organic layer Substances 0.000 description 27
- UIIMBOGNXHQVGW-UHFFFAOYSA-M Sodium bicarbonate Chemical compound [Na+].OC([O-])=O UIIMBOGNXHQVGW-UHFFFAOYSA-M 0.000 description 22
- 239000000651 prodrug Substances 0.000 description 22
- 229940002612 prodrug Drugs 0.000 description 22
- 238000010898 silica gel chromatography Methods 0.000 description 22
- 239000002904 solvent Substances 0.000 description 21
- 201000010099 disease Diseases 0.000 description 19
- 208000027866 inflammatory disease Diseases 0.000 description 19
- VLKZOEOYAKHREP-UHFFFAOYSA-N n-Hexane Chemical compound CCCCCC VLKZOEOYAKHREP-UHFFFAOYSA-N 0.000 description 19
- WEVYAHXRMPXWCK-UHFFFAOYSA-N Acetonitrile Chemical compound CC#N WEVYAHXRMPXWCK-UHFFFAOYSA-N 0.000 description 18
- CSNNHWWHGAXBCP-UHFFFAOYSA-L Magnesium sulfate Chemical compound [Mg+2].[O-][S+2]([O-])([O-])[O-] CSNNHWWHGAXBCP-UHFFFAOYSA-L 0.000 description 18
- 0 [6*][V]C1=CC([11*])=[Y]C(C)=[W]1 Chemical compound [6*][V]C1=CC([11*])=[Y]C(C)=[W]1 0.000 description 18
- 239000003039 volatile agent Substances 0.000 description 18
- 238000006243 chemical reaction Methods 0.000 description 16
- 230000002829 reductive effect Effects 0.000 description 16
- OKKJLVBELUTLKV-UHFFFAOYSA-N Methanol Chemical compound OC OKKJLVBELUTLKV-UHFFFAOYSA-N 0.000 description 15
- ZMANZCXQSJIPKH-UHFFFAOYSA-N Triethylamine Chemical compound CCN(CC)CC ZMANZCXQSJIPKH-UHFFFAOYSA-N 0.000 description 15
- 238000000746 purification Methods 0.000 description 15
- 239000012453 solvate Substances 0.000 description 15
- 125000004432 carbon atom Chemical group C* 0.000 description 14
- 238000004128 high performance liquid chromatography Methods 0.000 description 14
- RYHBNJHYFVUHQT-UHFFFAOYSA-N 1,4-Dioxane Chemical compound C1COCCO1 RYHBNJHYFVUHQT-UHFFFAOYSA-N 0.000 description 13
- LFQSCWFLJHTTHZ-UHFFFAOYSA-N Ethanol Chemical compound CCO LFQSCWFLJHTTHZ-UHFFFAOYSA-N 0.000 description 13
- 238000003818 flash chromatography Methods 0.000 description 13
- QTBSBXVTEAMEQO-UHFFFAOYSA-N Acetic acid Chemical compound CC(O)=O QTBSBXVTEAMEQO-UHFFFAOYSA-N 0.000 description 12
- PMZURENOXWZQFD-UHFFFAOYSA-L Sodium Sulfate Chemical compound [Na+].[Na+].[O-]S([O-])(=O)=O PMZURENOXWZQFD-UHFFFAOYSA-L 0.000 description 12
- 229910052938 sodium sulfate Inorganic materials 0.000 description 12
- 235000011152 sodium sulphate Nutrition 0.000 description 12
- 210000004027 cell Anatomy 0.000 description 11
- 239000011541 reaction mixture Substances 0.000 description 11
- 229910000030 sodium bicarbonate Inorganic materials 0.000 description 11
- ZMXDDKWLCZADIW-UHFFFAOYSA-N N,N-Dimethylformamide Chemical compound CN(C)C=O ZMXDDKWLCZADIW-UHFFFAOYSA-N 0.000 description 10
- 206010063837 Reperfusion injury Diseases 0.000 description 10
- 150000001412 amines Chemical class 0.000 description 10
- 206010003246 arthritis Diseases 0.000 description 10
- 125000000449 nitro group Chemical group [O-][N+](*)=O 0.000 description 10
- 235000017557 sodium bicarbonate Nutrition 0.000 description 10
- 238000003756 stirring Methods 0.000 description 10
- WKBOTKDWSSQWDR-UHFFFAOYSA-N Bromine atom Chemical compound [Br] WKBOTKDWSSQWDR-UHFFFAOYSA-N 0.000 description 9
- 208000006673 asthma Diseases 0.000 description 9
- GDTBXPJZTBHREO-UHFFFAOYSA-N bromine Substances BrBr GDTBXPJZTBHREO-UHFFFAOYSA-N 0.000 description 9
- 239000012043 crude product Substances 0.000 description 9
- 239000002158 endotoxin Substances 0.000 description 9
- 229910052943 magnesium sulfate Inorganic materials 0.000 description 9
- 201000006417 multiple sclerosis Diseases 0.000 description 9
- 229960001407 sodium bicarbonate Drugs 0.000 description 9
- DTQVDTLACAAQTR-UHFFFAOYSA-N trifluoroacetic acid Substances OC(=O)C(F)(F)F DTQVDTLACAAQTR-UHFFFAOYSA-N 0.000 description 9
- 208000024827 Alzheimer disease Diseases 0.000 description 8
- 201000001320 Atherosclerosis Diseases 0.000 description 8
- 206010007559 Cardiac failure congestive Diseases 0.000 description 8
- RTZKZFJDLAIYFH-UHFFFAOYSA-N Diethyl ether Chemical compound CCOCC RTZKZFJDLAIYFH-UHFFFAOYSA-N 0.000 description 8
- 206010019280 Heart failures Diseases 0.000 description 8
- 208000022559 Inflammatory bowel disease Diseases 0.000 description 8
- 206010040070 Septic Shock Diseases 0.000 description 8
- 125000004429 atom Chemical group 0.000 description 8
- 239000000460 chlorine Chemical group 0.000 description 8
- 239000003480 eluent Substances 0.000 description 8
- 125000004433 nitrogen atom Chemical group N* 0.000 description 8
- NROKBHXJSPEDAR-UHFFFAOYSA-M potassium fluoride Chemical compound [F-].[K+] NROKBHXJSPEDAR-UHFFFAOYSA-M 0.000 description 8
- NXFCCUBWWTWZGE-UHFFFAOYSA-N 3-amino-n,4-dimethylbenzamide Chemical compound CNC(=O)C1=CC=C(C)C(N)=C1 NXFCCUBWWTWZGE-UHFFFAOYSA-N 0.000 description 7
- 206010001052 Acute respiratory distress syndrome Diseases 0.000 description 7
- VEXZGXHMUGYJMC-UHFFFAOYSA-N Hydrochloric acid Chemical compound Cl VEXZGXHMUGYJMC-UHFFFAOYSA-N 0.000 description 7
- 208000013616 Respiratory Distress Syndrome Diseases 0.000 description 7
- 208000011341 adult acute respiratory distress syndrome Diseases 0.000 description 7
- 201000000028 adult respiratory distress syndrome Diseases 0.000 description 7
- 229920006008 lipopolysaccharide Polymers 0.000 description 7
- 125000001971 neopentyl group Chemical group [H]C([*])([H])C(C([H])([H])[H])(C([H])([H])[H])C([H])([H])[H] 0.000 description 7
- 108090000623 proteins and genes Proteins 0.000 description 7
- 125000001424 substituent group Chemical group 0.000 description 7
- CSCPPACGZOOCGX-UHFFFAOYSA-N Acetone Chemical compound CC(C)=O CSCPPACGZOOCGX-UHFFFAOYSA-N 0.000 description 6
- 206010006895 Cachexia Diseases 0.000 description 6
- OKTJSMMVPCPJKN-UHFFFAOYSA-N Carbon Chemical compound [C] OKTJSMMVPCPJKN-UHFFFAOYSA-N 0.000 description 6
- 206010009900 Colitis ulcerative Diseases 0.000 description 6
- 206010018364 Glomerulonephritis Diseases 0.000 description 6
- 201000005569 Gout Diseases 0.000 description 6
- 201000006704 Ulcerative Colitis Diseases 0.000 description 6
- 239000012267 brine Substances 0.000 description 6
- 229910052799 carbon Inorganic materials 0.000 description 6
- 239000002552 dosage form Substances 0.000 description 6
- IXCSERBJSXMMFS-UHFFFAOYSA-N hydrogen chloride Substances Cl.Cl IXCSERBJSXMMFS-UHFFFAOYSA-N 0.000 description 6
- 229910000041 hydrogen chloride Inorganic materials 0.000 description 6
- XHXFXVLFKHQFAL-UHFFFAOYSA-N phosphoryl trichloride Chemical compound ClP(Cl)(Cl)=O XHXFXVLFKHQFAL-UHFFFAOYSA-N 0.000 description 6
- 230000002685 pulmonary effect Effects 0.000 description 6
- HPALAKNZSZLMCH-UHFFFAOYSA-M sodium;chloride;hydrate Chemical compound O.[Na+].[Cl-] HPALAKNZSZLMCH-UHFFFAOYSA-M 0.000 description 6
- DEDKKOOGYIMMBC-UHFFFAOYSA-N 2,6-dichloro-5-fluoropyridine-3-carbonitrile Chemical compound FC1=CC(C#N)=C(Cl)N=C1Cl DEDKKOOGYIMMBC-UHFFFAOYSA-N 0.000 description 5
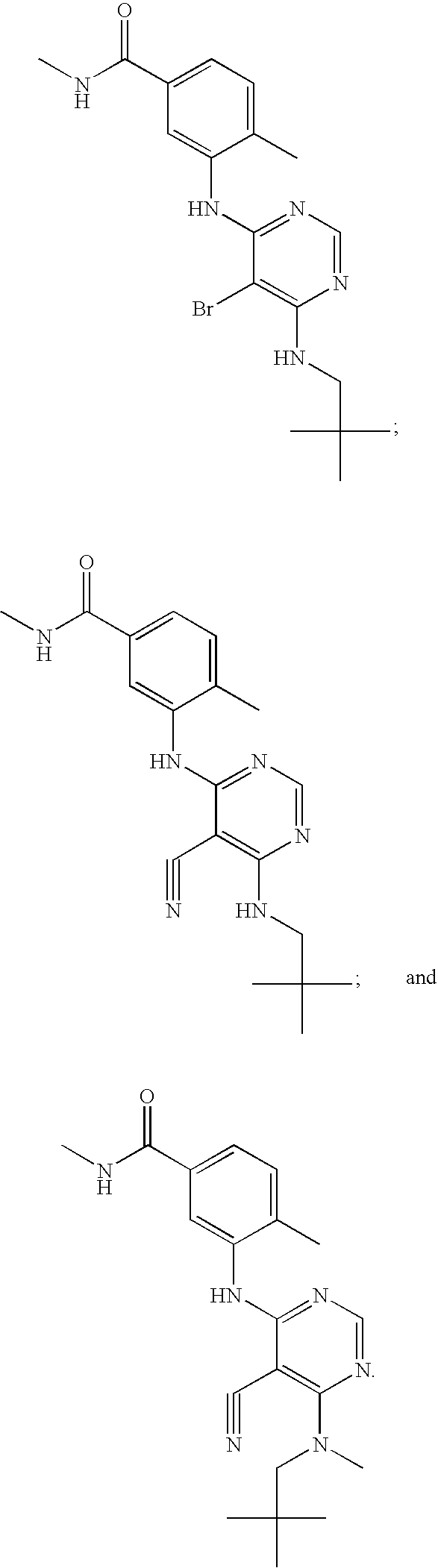
- CBSQWYUTOWGHBP-UHFFFAOYSA-N 3-[[5-bromo-6-(2,2-dimethylpropylamino)pyrimidin-4-yl]amino]-n,4-dimethylbenzamide Chemical compound CNC(=O)C1=CC=C(C)C(NC=2C(=C(NCC(C)(C)C)N=CN=2)Br)=C1 CBSQWYUTOWGHBP-UHFFFAOYSA-N 0.000 description 5
- UONISFJXHMSTQN-UHFFFAOYSA-N 3-[[5-cyano-6-[2,2-dimethylpropyl(methyl)amino]-2-(4-methyl-1,4-diazepan-1-yl)pyrimidin-4-yl]amino]-n-methoxy-4-methylbenzamide Chemical compound CONC(=O)C1=CC=C(C)C(NC=2C(=C(N(C)CC(C)(C)C)N=C(N=2)N2CCN(C)CCC2)C#N)=C1 UONISFJXHMSTQN-UHFFFAOYSA-N 0.000 description 5
- FLLNOJOVPMTRHA-UHFFFAOYSA-N 3-[[6-[2,2-dimethylpropyl(methyl)amino]-5-nitropyrimidin-4-yl]amino]-n,4-dimethylbenzamide Chemical compound CNC(=O)C1=CC=C(C)C(NC=2C(=C(N(C)CC(C)(C)C)N=CN=2)[N+]([O-])=O)=C1 FLLNOJOVPMTRHA-UHFFFAOYSA-N 0.000 description 5
- SQIYHQDCDNQPPX-UHFFFAOYSA-N 3-amino-n-methoxy-4-methylbenzamide Chemical compound CONC(=O)C1=CC=C(C)C(N)=C1 SQIYHQDCDNQPPX-UHFFFAOYSA-N 0.000 description 5
- GLOOTGZVAHKTJS-UHFFFAOYSA-N 4,6-dichloro-2-methylsulfanylpyrimidine-5-carbonitrile Chemical compound CSC1=NC(Cl)=C(C#N)C(Cl)=N1 GLOOTGZVAHKTJS-UHFFFAOYSA-N 0.000 description 5
- ZKHQWZAMYRWXGA-UHFFFAOYSA-N Adenosine triphosphate Natural products C1=NC=2C(N)=NC=NC=2N1C1OC(COP(O)(=O)OP(O)(=O)OP(O)(O)=O)C(O)C1O ZKHQWZAMYRWXGA-UHFFFAOYSA-N 0.000 description 5
- 208000023275 Autoimmune disease Diseases 0.000 description 5
- 102000004127 Cytokines Human genes 0.000 description 5
- 108090000695 Cytokines Proteins 0.000 description 5
- 208000002193 Pain Diseases 0.000 description 5
- 208000007536 Thrombosis Diseases 0.000 description 5
- 125000004414 alkyl thio group Chemical group 0.000 description 5
- 230000003110 anti-inflammatory effect Effects 0.000 description 5
- 230000000747 cardiac effect Effects 0.000 description 5
- 230000001684 chronic effect Effects 0.000 description 5
- 229910052731 fluorine Inorganic materials 0.000 description 5
- 229940125721 immunosuppressive agent Drugs 0.000 description 5
- 239000003018 immunosuppressive agent Substances 0.000 description 5
- 230000004054 inflammatory process Effects 0.000 description 5
- 230000002401 inhibitory effect Effects 0.000 description 5
- 239000010410 layer Substances 0.000 description 5
- QRUYXKCCDNOAAX-UHFFFAOYSA-N n,2,2-trimethylpropan-1-amine;hydrochloride Chemical compound Cl.CNCC(C)(C)C QRUYXKCCDNOAAX-UHFFFAOYSA-N 0.000 description 5
- 230000036407 pain Effects 0.000 description 5
- 239000002244 precipitate Substances 0.000 description 5
- 125000006239 protecting group Chemical group 0.000 description 5
- OLCSDVRURHCBDO-UHFFFAOYSA-N 3-[(5-bromo-2-chloropyrimidin-4-yl)amino]-n,4-dimethylbenzamide Chemical compound CNC(=O)C1=CC=C(C)C(NC=2C(=CN=C(Cl)N=2)Br)=C1 OLCSDVRURHCBDO-UHFFFAOYSA-N 0.000 description 4
- LWPGQUGXUYWLHN-UHFFFAOYSA-N 3-[(6-chloropyrimidin-4-yl)amino]-n,4-dimethylbenzamide Chemical compound CNC(=O)C1=CC=C(C)C(NC=2N=CN=C(Cl)C=2)=C1 LWPGQUGXUYWLHN-UHFFFAOYSA-N 0.000 description 4
- XQWSBVPVQYFDNI-UHFFFAOYSA-N 3-[[5-bromo-6-[2,2-dimethylpropyl(methyl)amino]pyrimidin-4-yl]amino]-n,4-dimethylbenzamide Chemical compound CNC(=O)C1=CC=C(C)C(NC=2C(=C(N(C)CC(C)(C)C)N=CN=2)Br)=C1 XQWSBVPVQYFDNI-UHFFFAOYSA-N 0.000 description 4
- ZXQDCBDWEGQQGQ-UHFFFAOYSA-N 3-[[5-cyano-6-(2,2-dimethylpropylamino)-3-fluoropyridin-2-yl]amino]-n-methoxy-4-methylbenzamide Chemical compound CONC(=O)C1=CC=C(C)C(NC=2C(=CC(=C(NCC(C)(C)C)N=2)C#N)F)=C1 ZXQDCBDWEGQQGQ-UHFFFAOYSA-N 0.000 description 4
- FZMANIZMBKDOKM-UHFFFAOYSA-N 3-[[5-cyano-6-[2,2-dimethylpropyl(methyl)amino]-2-methylsulfinylpyrimidin-4-yl]amino]-n-methoxy-4-methylbenzamide Chemical compound CONC(=O)C1=CC=C(C)C(NC=2C(=C(N(C)CC(C)(C)C)N=C(N=2)S(C)=O)C#N)=C1 FZMANIZMBKDOKM-UHFFFAOYSA-N 0.000 description 4
- VKYBTXNSTBNBBV-UHFFFAOYSA-N 3-[[5-cyano-6-[2,2-dimethylpropyl(methyl)amino]-2-methylsulfonylpyrimidin-4-yl]amino]-n,4-dimethylbenzamide Chemical compound CNC(=O)C1=CC=C(C)C(NC=2C(=C(N(C)CC(C)(C)C)N=C(N=2)S(C)(=O)=O)C#N)=C1 VKYBTXNSTBNBBV-UHFFFAOYSA-N 0.000 description 4
- SCEVBRBKKQZTKM-UHFFFAOYSA-N 5-[[6-chloro-5-(1-methylindol-5-yl)-1H-benzimidazol-2-yl]oxy]-N-hydroxy-2-methylbenzamide Chemical compound ClC=1C(=CC2=C(NC(=N2)OC=2C=CC(=C(C(=O)NO)C=2)C)C=1)C=1C=C2C=CN(C2=CC=1)C SCEVBRBKKQZTKM-UHFFFAOYSA-N 0.000 description 4
- 208000006386 Bone Resorption Diseases 0.000 description 4
- ZAMOUSCENKQFHK-UHFFFAOYSA-N Chlorine atom Chemical compound [Cl] ZAMOUSCENKQFHK-UHFFFAOYSA-N 0.000 description 4
- 102100023401 Dual specificity mitogen-activated protein kinase kinase 6 Human genes 0.000 description 4
- PXGOKWXKJXAPGV-UHFFFAOYSA-N Fluorine Chemical compound FF PXGOKWXKJXAPGV-UHFFFAOYSA-N 0.000 description 4
- 101000624426 Homo sapiens Dual specificity mitogen-activated protein kinase kinase 6 Proteins 0.000 description 4
- 206010061218 Inflammation Diseases 0.000 description 4
- 102000000589 Interleukin-1 Human genes 0.000 description 4
- 108010002352 Interleukin-1 Proteins 0.000 description 4
- BAVYZALUXZFZLV-UHFFFAOYSA-N Methylamine Chemical compound NC BAVYZALUXZFZLV-UHFFFAOYSA-N 0.000 description 4
- 102100038280 Prostaglandin G/H synthase 2 Human genes 0.000 description 4
- JUJWROOIHBZHMG-UHFFFAOYSA-N Pyridine Chemical compound C1=CC=NC=C1 JUJWROOIHBZHMG-UHFFFAOYSA-N 0.000 description 4
- HEDRZPFGACZZDS-MICDWDOJSA-N Trichloro(2H)methane Chemical compound [2H]C(Cl)(Cl)Cl HEDRZPFGACZZDS-MICDWDOJSA-N 0.000 description 4
- 238000003556 assay Methods 0.000 description 4
- 230000024279 bone resorption Effects 0.000 description 4
- KXZJHVJKXJLBKO-UHFFFAOYSA-N chembl1408157 Chemical compound N=1C2=CC=CC=C2C(C(=O)O)=CC=1C1=CC=C(O)C=C1 KXZJHVJKXJLBKO-UHFFFAOYSA-N 0.000 description 4
- 238000001914 filtration Methods 0.000 description 4
- 239000011737 fluorine Substances 0.000 description 4
- 125000001072 heteroaryl group Chemical group 0.000 description 4
- 125000002887 hydroxy group Chemical group [H]O* 0.000 description 4
- 208000015181 infectious disease Diseases 0.000 description 4
- 239000000543 intermediate Substances 0.000 description 4
- UQGXHNDRCRTZAC-UHFFFAOYSA-N n,2,2-trimethylpropan-1-amine Chemical compound CNCC(C)(C)C UQGXHNDRCRTZAC-UHFFFAOYSA-N 0.000 description 4
- 235000003270 potassium fluoride Nutrition 0.000 description 4
- 239000011698 potassium fluoride Substances 0.000 description 4
- 238000002360 preparation method Methods 0.000 description 4
- 102000004169 proteins and genes Human genes 0.000 description 4
- 239000007787 solid Substances 0.000 description 4
- 238000012360 testing method Methods 0.000 description 4
- 230000000699 topical effect Effects 0.000 description 4
- 230000006433 tumor necrosis factor production Effects 0.000 description 4
- BAUWRHPMUVYFOD-UHFFFAOYSA-N 1-methylpiperidin-4-ol Chemical compound CN1CCC(O)CC1 BAUWRHPMUVYFOD-UHFFFAOYSA-N 0.000 description 3
- XDIAMRVROCPPBK-UHFFFAOYSA-N 2,2-dimethylpropan-1-amine Chemical compound CC(C)(C)CN XDIAMRVROCPPBK-UHFFFAOYSA-N 0.000 description 3
- FQFWAUUFFGBSPE-UHFFFAOYSA-N 2-chloro-6-[2,2-dimethylpropyl(methyl)amino]-5-fluoropyridine-3-carbonitrile Chemical compound CC(C)(C)CN(C)C1=NC(Cl)=C(C#N)C=C1F FQFWAUUFFGBSPE-UHFFFAOYSA-N 0.000 description 3
- YIYSSVJBDXRCCE-UHFFFAOYSA-N 3-[(6-chloro-5-cyano-2-methylsulfanylpyrimidin-4-yl)amino]-n,4-dimethylbenzamide Chemical compound CNC(=O)C1=CC=C(C)C(NC=2C(=C(Cl)N=C(SC)N=2)C#N)=C1 YIYSSVJBDXRCCE-UHFFFAOYSA-N 0.000 description 3
- RAVHIBMPQXXIFO-UHFFFAOYSA-N 3-[[3-cyano-6-(2,2-dimethylpropylamino)-5-fluoropyridin-2-yl]amino]-n-methoxy-4-methylbenzamide Chemical compound CONC(=O)C1=CC=C(C)C(NC=2C(=CC(F)=C(NCC(C)(C)C)N=2)C#N)=C1 RAVHIBMPQXXIFO-UHFFFAOYSA-N 0.000 description 3
- YQHZBICMXSGGPM-UHFFFAOYSA-N 3-[[5-amino-6-[2,2-dimethylpropyl(methyl)amino]pyrimidin-4-yl]amino]-n,4-dimethylbenzamide Chemical compound CNC(=O)C1=CC=C(C)C(NC=2C(=C(N(C)CC(C)(C)C)N=CN=2)N)=C1 YQHZBICMXSGGPM-UHFFFAOYSA-N 0.000 description 3
- MOWBVAHTPQPQFH-UHFFFAOYSA-N 3-[[5-cyano-2-[2,2-dimethylpropyl(methyl)amino]-6-(4-methyl-1,4-diazepan-1-yl)pyrimidin-4-yl]amino]-n-ethoxy-4-methylbenzamide Chemical compound CCONC(=O)C1=CC=C(C)C(NC=2C(=C(N3CCN(C)CCC3)N=C(N=2)N(C)CC(C)(C)C)C#N)=C1 MOWBVAHTPQPQFH-UHFFFAOYSA-N 0.000 description 3
- PSPJBZZYNOEUAW-UHFFFAOYSA-N 3-[[5-cyano-4-[2,2-dimethylpropyl(methyl)amino]-6-(4-methyl-1,4-diazepan-1-yl)pyrimidin-2-yl]amino]-n-methoxy-4-methylbenzamide Chemical compound CONC(=O)C1=CC=C(C)C(NC=2N=C(C(C#N)=C(N(C)CC(C)(C)C)N=2)N2CCN(C)CCC2)=C1 PSPJBZZYNOEUAW-UHFFFAOYSA-N 0.000 description 3
- OZXQHVWVERIBQL-UHFFFAOYSA-N 3-[[5-cyano-6-[2,2-dimethylpropyl(methyl)amino]-2-(4-methyl-1,4-diazepan-1-yl)pyrimidin-4-yl]amino]-n,4-dimethylbenzamide Chemical compound CNC(=O)C1=CC=C(C)C(NC=2C(=C(N(C)CC(C)(C)C)N=C(N=2)N2CCN(C)CCC2)C#N)=C1 OZXQHVWVERIBQL-UHFFFAOYSA-N 0.000 description 3
- QFNDHSLMOVLXDJ-UHFFFAOYSA-N 3-[[5-cyano-6-[2,2-dimethylpropyl(methyl)amino]-2-(methylamino)pyrimidin-4-yl]amino]-n,4-dimethylbenzamide Chemical compound CNC(=O)C1=CC=C(C)C(NC=2C(=C(N(C)CC(C)(C)C)N=C(NC)N=2)C#N)=C1 QFNDHSLMOVLXDJ-UHFFFAOYSA-N 0.000 description 3
- XJGYVASDBIPRAF-UHFFFAOYSA-N 3-[[5-cyano-6-[2,2-dimethylpropyl(methyl)amino]-2-(methylamino)pyrimidin-4-yl]amino]-n-methoxy-4-methylbenzamide Chemical compound CC(C)(C)CN(C)C1=NC(NC)=NC(NC=2C(=CC=C(C=2)C(=O)NOC)C)=C1C#N XJGYVASDBIPRAF-UHFFFAOYSA-N 0.000 description 3
- IKMWKACSKQPBOI-UHFFFAOYSA-N 3-[[5-cyano-6-[2,2-dimethylpropyl(methyl)amino]-2-methylsulfanylpyrimidin-4-yl]amino]-n,4-dimethylbenzamide Chemical compound CNC(=O)C1=CC=C(C)C(NC=2C(=C(N(C)CC(C)(C)C)N=C(SC)N=2)C#N)=C1 IKMWKACSKQPBOI-UHFFFAOYSA-N 0.000 description 3
- WTVNJECUUZTSSA-UHFFFAOYSA-N 3-[[5-cyano-6-[2,2-dimethylpropyl(methyl)amino]-3-fluoropyridin-2-yl]amino]-n-methoxy-4-methylbenzamide Chemical compound CONC(=O)C1=CC=C(C)C(NC=2C(=CC(=C(N(C)CC(C)(C)C)N=2)C#N)F)=C1 WTVNJECUUZTSSA-UHFFFAOYSA-N 0.000 description 3
- MRHGOAKYYORQGQ-UHFFFAOYSA-N 4,6-dichloro-2-methylsulfanylpyrimidine-5-carbaldehyde Chemical compound CSC1=NC(Cl)=C(C=O)C(Cl)=N1 MRHGOAKYYORQGQ-UHFFFAOYSA-N 0.000 description 3
- ZPCPRYSSTJVFOS-UHFFFAOYSA-N 4-[2,2-dimethylpropyl(methyl)amino]-2-methylsulfanyl-6-[2-methyl-5-(1h-1,2,4-triazol-5-yl)anilino]pyrimidine-5-carbonitrile Chemical compound CC(C)(C)CN(C)C1=NC(SC)=NC(NC=2C(=CC=C(C=2)C=2NC=NN=2)C)=C1C#N ZPCPRYSSTJVFOS-UHFFFAOYSA-N 0.000 description 3
- YNRLJYPVRJBZGD-UHFFFAOYSA-N 4-n-(2,2-dimethylpropyl)-4-n-methyl-6-n-[2-methyl-5-(1h-1,2,4-triazol-5-yl)phenyl]-5-nitropyrimidine-4,6-diamine Chemical compound CC(C)(C)CN(C)C1=NC=NC(NC=2C(=CC=C(C=2)C=2NC=NN=2)C)=C1[N+]([O-])=O YNRLJYPVRJBZGD-UHFFFAOYSA-N 0.000 description 3
- AOHDWLIIXDURIG-UHFFFAOYSA-N 6-chloro-n-(2,2-dimethylpropyl)-n-methyl-5-nitropyrimidin-4-amine Chemical compound CC(C)(C)CN(C)C1=NC=NC(Cl)=C1[N+]([O-])=O AOHDWLIIXDURIG-UHFFFAOYSA-N 0.000 description 3
- 208000020084 Bone disease Diseases 0.000 description 3
- VPINENFZKNQTMZ-QGZVFWFLSA-N CNC(=O)C1=CC=C(C)C(NC2=NC(N(C)CC(C)(C)C)=NC(N[C@@H]3CCNC3)=C2C#N)=C1 Chemical compound CNC(=O)C1=CC=C(C)C(NC2=NC(N(C)CC(C)(C)C)=NC(N[C@@H]3CCNC3)=C2C#N)=C1 VPINENFZKNQTMZ-QGZVFWFLSA-N 0.000 description 3
- PCERTJMLECOVDM-QGZVFWFLSA-N CNC(=O)C1=CC=C(C)C(NC2=NC(N[C@@H]3CCNC3)=C(C#N)C(N(C)CC(C)(C)C)=N2)=C1 Chemical compound CNC(=O)C1=CC=C(C)C(NC2=NC(N[C@@H]3CCNC3)=C(C#N)C(N(C)CC(C)(C)C)=N2)=C1 PCERTJMLECOVDM-QGZVFWFLSA-N 0.000 description 3
- RLNZVPTUOMGUGY-UHFFFAOYSA-N CONC(=O)C1=CC=C(C)C(NC2=NC(OCCC3CCCN3C)=NC(N(C)CC(C)(C)C)=C2C#N)=C1 Chemical compound CONC(=O)C1=CC=C(C)C(NC2=NC(OCCC3CCCN3C)=NC(N(C)CC(C)(C)C)=C2C#N)=C1 RLNZVPTUOMGUGY-UHFFFAOYSA-N 0.000 description 3
- 108020004635 Complementary DNA Proteins 0.000 description 3
- KCXVZYZYPLLWCC-UHFFFAOYSA-N EDTA Chemical compound OC(=O)CN(CC(O)=O)CCN(CC(O)=O)CC(O)=O KCXVZYZYPLLWCC-UHFFFAOYSA-N 0.000 description 3
- 102000004190 Enzymes Human genes 0.000 description 3
- 108090000790 Enzymes Proteins 0.000 description 3
- 108010044467 Isoenzymes Proteins 0.000 description 3
- 206010028980 Neoplasm Diseases 0.000 description 3
- 108091000080 Phosphotransferase Proteins 0.000 description 3
- 108010029485 Protein Isoforms Proteins 0.000 description 3
- 102000001708 Protein Isoforms Human genes 0.000 description 3
- 239000002260 anti-inflammatory agent Substances 0.000 description 3
- 229940121363 anti-inflammatory agent Drugs 0.000 description 3
- 239000003153 chemical reaction reagent Substances 0.000 description 3
- 239000003814 drug Substances 0.000 description 3
- 229940088598 enzyme Drugs 0.000 description 3
- 239000000706 filtrate Substances 0.000 description 3
- 239000012467 final product Substances 0.000 description 3
- 108020001507 fusion proteins Proteins 0.000 description 3
- 102000037865 fusion proteins Human genes 0.000 description 3
- 125000001449 isopropyl group Chemical group [H]C([H])([H])C([H])(*)C([H])([H])[H] 0.000 description 3
- 108020004999 messenger RNA Proteins 0.000 description 3
- 230000004770 neurodegeneration Effects 0.000 description 3
- 208000015122 neurodegenerative disease Diseases 0.000 description 3
- 239000000041 non-steroidal anti-inflammatory agent Substances 0.000 description 3
- 229940021182 non-steroidal anti-inflammatory drug Drugs 0.000 description 3
- 102000020233 phosphotransferase Human genes 0.000 description 3
- LPNYRYFBWFDTMA-UHFFFAOYSA-N potassium tert-butoxide Chemical compound [K+].CC(C)(C)[O-] LPNYRYFBWFDTMA-UHFFFAOYSA-N 0.000 description 3
- 238000012746 preparative thin layer chromatography Methods 0.000 description 3
- 230000002265 prevention Effects 0.000 description 3
- 239000012264 purified product Substances 0.000 description 3
- 150000003254 radicals Chemical class 0.000 description 3
- 238000010992 reflux Methods 0.000 description 3
- 239000011734 sodium Substances 0.000 description 3
- 239000000758 substrate Substances 0.000 description 3
- YLQBMQCUIZJEEH-UHFFFAOYSA-N tetrahydrofuran Natural products C=1C=COC=1 YLQBMQCUIZJEEH-UHFFFAOYSA-N 0.000 description 3
- 125000003088 (fluoren-9-ylmethoxy)carbonyl group Chemical group 0.000 description 2
- WSLDOOZREJYCGB-UHFFFAOYSA-N 1,2-Dichloroethane Chemical compound ClCCCl WSLDOOZREJYCGB-UHFFFAOYSA-N 0.000 description 2
- 238000005160 1H NMR spectroscopy Methods 0.000 description 2
- IBKKZHDEHVVGBN-UHFFFAOYSA-N 2-chloro-6-(2,2-dimethylpropylamino)-5-fluoropyridine-3-carbonitrile Chemical compound CC(C)(C)CNC1=NC(Cl)=C(C#N)C=C1F IBKKZHDEHVVGBN-UHFFFAOYSA-N 0.000 description 2
- ZAQFXOXZMRYLLE-UHFFFAOYSA-N 2-methyl-5-(1h-1,2,4-triazol-5-yl)aniline Chemical compound C1=C(N)C(C)=CC=C1C1=NN=CN1 ZAQFXOXZMRYLLE-UHFFFAOYSA-N 0.000 description 2
- FBVFHNYPUGBROK-UHFFFAOYSA-N 3-[(6-chloro-3-cyano-5-fluoropyridin-2-yl)amino]-n-methoxy-4-methylbenzamide Chemical compound CONC(=O)C1=CC=C(C)C(NC=2C(=CC(F)=C(Cl)N=2)C#N)=C1 FBVFHNYPUGBROK-UHFFFAOYSA-N 0.000 description 2
- NGEKCRASOVPFHI-UHFFFAOYSA-N 3-[(6-chloro-5-cyano-2-methylsulfanylpyrimidin-4-yl)amino]-n-methoxy-4-methylbenzamide Chemical compound CONC(=O)C1=CC=C(C)C(NC=2C(=C(Cl)N=C(SC)N=2)C#N)=C1 NGEKCRASOVPFHI-UHFFFAOYSA-N 0.000 description 2
- PYVFNJNDMMVFQN-UHFFFAOYSA-N 3-[(6-chloro-5-cyano-3-fluoropyridin-2-yl)amino]-n-methoxy-4-methylbenzamide Chemical compound CONC(=O)C1=CC=C(C)C(NC=2C(=CC(=C(Cl)N=2)C#N)F)=C1 PYVFNJNDMMVFQN-UHFFFAOYSA-N 0.000 description 2
- GWOZCMSNQYRWMZ-UHFFFAOYSA-N 3-[[2-[3-(dimethylamino)propylamino]-6-[2,2-dimethylpropyl(methyl)amino]-5-nitropyrimidin-4-yl]amino]-n,4-dimethylbenzamide Chemical compound CNC(=O)C1=CC=C(C)C(NC=2C(=C(N(C)CC(C)(C)C)N=C(NCCCN(C)C)N=2)[N+]([O-])=O)=C1 GWOZCMSNQYRWMZ-UHFFFAOYSA-N 0.000 description 2
- YUGFSINFHHWZDZ-UHFFFAOYSA-N 3-[[3-cyano-6-[2,2-dimethylpropyl(methyl)amino]-5-fluoropyridin-2-yl]amino]-n-methoxy-4-methylbenzamide Chemical compound CONC(=O)C1=CC=C(C)C(NC=2C(=CC(F)=C(N(C)CC(C)(C)C)N=2)C#N)=C1 YUGFSINFHHWZDZ-UHFFFAOYSA-N 0.000 description 2
- HNVPGJXZTYTKKI-UHFFFAOYSA-N 3-[[5-acetamido-6-[2,2-dimethylpropyl(methyl)amino]pyrimidin-4-yl]amino]-n,4-dimethylbenzamide Chemical compound CNC(=O)C1=CC=C(C)C(NC=2C(=C(N(C)CC(C)(C)C)N=CN=2)NC(C)=O)=C1 HNVPGJXZTYTKKI-UHFFFAOYSA-N 0.000 description 2
- PADKCSXGWUKVND-UHFFFAOYSA-N 3-[[5-bromo-6-(2,2-dimethylpropylamino)-2-(methylamino)pyrimidin-4-yl]amino]-n,4-dimethylbenzamide Chemical compound CNC(=O)C1=CC=C(C)C(NC=2C(=C(NCC(C)(C)C)N=C(NC)N=2)Br)=C1 PADKCSXGWUKVND-UHFFFAOYSA-N 0.000 description 2
- GAEINKNHTGGMKX-UHFFFAOYSA-N 3-[[5-bromo-6-[2,2-dimethylpropyl(methyl)amino]-2-(methylamino)pyrimidin-4-yl]amino]-n,4-dimethylbenzamide Chemical compound CNC(=O)C1=CC=C(C)C(NC=2C(=C(N(C)CC(C)(C)C)N=C(NC)N=2)Br)=C1 GAEINKNHTGGMKX-UHFFFAOYSA-N 0.000 description 2
- RMYMFFZYKNHCFJ-UHFFFAOYSA-N 3-[[5-bromo-6-[2,2-dimethylpropyl(methyl)amino]-2-methylsulfanylpyrimidin-4-yl]amino]-n,4-dimethylbenzamide Chemical compound CNC(=O)C1=CC=C(C)C(NC=2C(=C(N(C)CC(C)(C)C)N=C(SC)N=2)Br)=C1 RMYMFFZYKNHCFJ-UHFFFAOYSA-N 0.000 description 2
- DOYRTFWVJGIOFF-UHFFFAOYSA-N 3-[[5-cyano-4-[2,2-dimethylpropyl(methyl)amino]pyrimidin-2-yl]amino]-n,4-dimethylbenzamide Chemical compound CNC(=O)C1=CC=C(C)C(NC=2N=C(C(C#N)=CN=2)N(C)CC(C)(C)C)=C1 DOYRTFWVJGIOFF-UHFFFAOYSA-N 0.000 description 2
- MCMHMGZBNVWWJB-UHFFFAOYSA-N 3-[[5-cyano-6-[2,2-dimethylpropyl(methyl)amino]-2-methylsulfanylpyrimidin-4-yl]amino]-n-methoxy-4-methylbenzamide Chemical compound CONC(=O)C1=CC=C(C)C(NC=2C(=C(N(C)CC(C)(C)C)N=C(SC)N=2)C#N)=C1 MCMHMGZBNVWWJB-UHFFFAOYSA-N 0.000 description 2
- ZOVOCXLTKOTVBY-UHFFFAOYSA-N 3-[[6-(2,2-dimethylpropylamino)pyrimidin-4-yl]amino]-n,4-dimethylbenzamide Chemical compound CNC(=O)C1=CC=C(C)C(NC=2N=CN=C(NCC(C)(C)C)C=2)=C1 ZOVOCXLTKOTVBY-UHFFFAOYSA-N 0.000 description 2
- GVELABHRQHAXDQ-UHFFFAOYSA-N 3-[[6-[2,2-dimethylpropyl(methyl)amino]-2-(1-methylpiperidin-4-yl)oxy-5-nitropyrimidin-4-yl]amino]-n,4-dimethylbenzamide Chemical compound CNC(=O)C1=CC=C(C)C(NC=2C(=C(N(C)CC(C)(C)C)N=C(OC3CCN(C)CC3)N=2)[N+]([O-])=O)=C1 GVELABHRQHAXDQ-UHFFFAOYSA-N 0.000 description 2
- TZWQMSZHBCUZQM-UHFFFAOYSA-N 3-[[6-[2,2-dimethylpropyl(methyl)amino]-2-methylsulfanyl-5-nitropyrimidin-4-yl]amino]-n,4-dimethylbenzamide Chemical compound CNC(=O)C1=CC=C(C)C(NC=2C(=C(N(C)CC(C)(C)C)N=C(SC)N=2)[N+]([O-])=O)=C1 TZWQMSZHBCUZQM-UHFFFAOYSA-N 0.000 description 2
- MRDOGNAGJIUIPO-UHFFFAOYSA-N 3-[[6-[2,2-dimethylpropyl(methyl)amino]-2-methylsulfonyl-5-nitropyrimidin-4-yl]amino]-n,4-dimethylbenzamide Chemical compound CNC(=O)C1=CC=C(C)C(NC=2C(=C(N(C)CC(C)(C)C)N=C(N=2)S(C)(=O)=O)[N+]([O-])=O)=C1 MRDOGNAGJIUIPO-UHFFFAOYSA-N 0.000 description 2
- KFEGQHGOJKTFMG-UHFFFAOYSA-N 3-[[6-[2,2-dimethylpropyl(methyl)amino]pyrimidin-4-yl]amino]-n,4-dimethylbenzamide Chemical compound CNC(=O)C1=CC=C(C)C(NC=2N=CN=C(C=2)N(C)CC(C)(C)C)=C1 KFEGQHGOJKTFMG-UHFFFAOYSA-N 0.000 description 2
- HCTISZQLTGAYOX-UHFFFAOYSA-N 4,6-dichloro-5-nitropyrimidine Chemical compound [O-][N+](=O)C1=C(Cl)N=CN=C1Cl HCTISZQLTGAYOX-UHFFFAOYSA-N 0.000 description 2
- XJPZKYIHCLDXST-UHFFFAOYSA-N 4,6-dichloropyrimidine Chemical compound ClC1=CC(Cl)=NC=N1 XJPZKYIHCLDXST-UHFFFAOYSA-N 0.000 description 2
- FXVUECNXZPZMCF-QGZVFWFLSA-N 4-[2,2-dimethylpropyl(methyl)amino]-6-[2-methyl-5-(1h-1,2,4-triazol-5-yl)anilino]-2-[[(3r)-pyrrolidin-3-yl]amino]pyrimidine-5-carbonitrile Chemical compound N=1C(NC=2C(=CC=C(C=2)C=2NC=NN=2)C)=C(C#N)C(N(CC(C)(C)C)C)=NC=1N[C@@H]1CCNC1 FXVUECNXZPZMCF-QGZVFWFLSA-N 0.000 description 2
- OPTJUOMHYBCQHW-UHFFFAOYSA-N 4-[2,2-dimethylpropyl(methyl)amino]-6-[2-methyl-5-(1h-1,2,4-triazol-5-yl)anilino]pyrimidine-5-carbonitrile Chemical compound CC(C)(C)CN(C)C1=NC=NC(NC=2C(=CC=C(C=2)C=2NC=NN=2)C)=C1C#N OPTJUOMHYBCQHW-UHFFFAOYSA-N 0.000 description 2
- AEXCUJUYEZIWJV-UHFFFAOYSA-N 4-hydroxy-2-methylsulfanyl-1h-pyrimidin-6-one Chemical compound CSC1=NC(O)=CC(=O)N1 AEXCUJUYEZIWJV-UHFFFAOYSA-N 0.000 description 2
- BEJLDIRNTMYPOY-UHFFFAOYSA-N 4-n-(2,2-dimethylpropyl)-4-n-methyl-6-n-[2-methyl-5-(1h-1,2,4-triazol-5-yl)phenyl]pyrimidine-4,5,6-triamine Chemical compound CC(C)(C)CN(C)C1=NC=NC(NC=2C(=CC=C(C=2)C=2NC=NN=2)C)=C1N BEJLDIRNTMYPOY-UHFFFAOYSA-N 0.000 description 2
- KQWOCDRMDSBBDH-UHFFFAOYSA-N 5-(5-chloro-2-fluorophenyl)-1,3,5,7,8,9-hexahydro-[1,2,5]oxadiazolo[3,4-b][1,4]benzodiazepin-6-one Chemical compound FC1=CC=C(Cl)C=C1C1C(C(=O)CCC2)=C2N=C2NONC2=N1 KQWOCDRMDSBBDH-UHFFFAOYSA-N 0.000 description 2
- PAYRUJLWNCNPSJ-UHFFFAOYSA-N Aniline Chemical compound NC1=CC=CC=C1 PAYRUJLWNCNPSJ-UHFFFAOYSA-N 0.000 description 2
- XKRFYHLGVUSROY-UHFFFAOYSA-N Argon Chemical compound [Ar] XKRFYHLGVUSROY-UHFFFAOYSA-N 0.000 description 2
- 108091003079 Bovine Serum Albumin Proteins 0.000 description 2
- DIGBZOXKXJRDIB-UHFFFAOYSA-N CC#N.CCN(CC)CC.CONC(=O)C1=CC=C(C)C(N)=C1.[C-]#[N+]C1=C(Cl)N=C(Cl)C(F)=C1.[C-]#[N+]C1=CC(F)=C(NC2=CC(C(=O)NOC)=CC=C2C)N=C1Cl Chemical compound CC#N.CCN(CC)CC.CONC(=O)C1=CC=C(C)C(N)=C1.[C-]#[N+]C1=C(Cl)N=C(Cl)C(F)=C1.[C-]#[N+]C1=CC(F)=C(NC2=CC(C(=O)NOC)=CC=C2C)N=C1Cl DIGBZOXKXJRDIB-UHFFFAOYSA-N 0.000 description 2
- BJELTIRSGBXBSF-UHFFFAOYSA-N CNC(=O)C1=CC=C(C)C(N)=C1.CNC(=O)C1=CC=C(C)C(NC2=NC=NC(Cl)=C2)=C1.ClC1=CC(Cl)=NC=N1 Chemical compound CNC(=O)C1=CC=C(C)C(N)=C1.CNC(=O)C1=CC=C(C)C(NC2=NC=NC(Cl)=C2)=C1.ClC1=CC(Cl)=NC=N1 BJELTIRSGBXBSF-UHFFFAOYSA-N 0.000 description 2
- KPNFPQGZGYOXSA-QGZVFWFLSA-N CNC(=O)C1=CC=C(C)C(NC2=NC(N[C@@H]3CCNC3)=NC(N(C)CC(C)(C)C)=C2C#N)=C1 Chemical compound CNC(=O)C1=CC=C(C)C(NC2=NC(N[C@@H]3CCNC3)=NC(N(C)CC(C)(C)C)=C2C#N)=C1 KPNFPQGZGYOXSA-QGZVFWFLSA-N 0.000 description 2
- VBXGFLKOKADANI-UHFFFAOYSA-N CNC(=O)C1=CC=C(C)C(NC2=NC(OCCC3CCCN3C)=NC(N(C)CC(C)(C)C)=C2C#N)=C1 Chemical compound CNC(=O)C1=CC=C(C)C(NC2=NC(OCCC3CCCN3C)=NC(N(C)CC(C)(C)C)=C2C#N)=C1 VBXGFLKOKADANI-UHFFFAOYSA-N 0.000 description 2
- UHDVMBPCTMXDQZ-QGZVFWFLSA-N CONC(=O)C1=CC=C(C)C(NC2=NC(N[C@@H]3CCNC3)=NC(N(C)CC(C)(C)C)=C2C#N)=C1 Chemical compound CONC(=O)C1=CC=C(C)C(NC2=NC(N[C@@H]3CCNC3)=NC(N(C)CC(C)(C)C)=C2C#N)=C1 UHDVMBPCTMXDQZ-QGZVFWFLSA-N 0.000 description 2
- 239000004215 Carbon black (E152) Substances 0.000 description 2
- 208000035473 Communicable disease Diseases 0.000 description 2
- 108010037462 Cyclooxygenase 2 Proteins 0.000 description 2
- 238000002965 ELISA Methods 0.000 description 2
- 108010008165 Etanercept Proteins 0.000 description 2
- WTDHULULXKLSOZ-UHFFFAOYSA-N Hydroxylamine hydrochloride Chemical compound Cl.ON WTDHULULXKLSOZ-UHFFFAOYSA-N 0.000 description 2
- 206010020751 Hypersensitivity Diseases 0.000 description 2
- SIKJAQJRHWYJAI-UHFFFAOYSA-N Indole Chemical compound C1=CC=C2NC=CC2=C1 SIKJAQJRHWYJAI-UHFFFAOYSA-N 0.000 description 2
- 102000004890 Interleukin-8 Human genes 0.000 description 2
- 108090001007 Interleukin-8 Proteins 0.000 description 2
- 102100034069 MAP kinase-activated protein kinase 2 Human genes 0.000 description 2
- 101710141394 MAP kinase-activated protein kinase 2 Proteins 0.000 description 2
- TWRXJAOTZQYOKJ-UHFFFAOYSA-L Magnesium chloride Chemical compound [Mg+2].[Cl-].[Cl-] TWRXJAOTZQYOKJ-UHFFFAOYSA-L 0.000 description 2
- 208000034578 Multiple myelomas Diseases 0.000 description 2
- LRHPLDYGYMQRHN-UHFFFAOYSA-N N-Butanol Chemical compound CCCCO LRHPLDYGYMQRHN-UHFFFAOYSA-N 0.000 description 2
- SECXISVLQFMRJM-UHFFFAOYSA-N N-Methylpyrrolidone Chemical compound CN1CCCC1=O SECXISVLQFMRJM-UHFFFAOYSA-N 0.000 description 2
- 239000012826 P38 inhibitor Substances 0.000 description 2
- KDLHZDBZIXYQEI-UHFFFAOYSA-N Palladium on carbon Substances [Pd] KDLHZDBZIXYQEI-UHFFFAOYSA-N 0.000 description 2
- 206010035226 Plasma cell myeloma Diseases 0.000 description 2
- 108050003267 Prostaglandin G/H synthase 2 Proteins 0.000 description 2
- 102000001253 Protein Kinase Human genes 0.000 description 2
- KYQCOXFCLRTKLS-UHFFFAOYSA-N Pyrazine Chemical compound C1=CN=CC=N1 KYQCOXFCLRTKLS-UHFFFAOYSA-N 0.000 description 2
- 206010037660 Pyrexia Diseases 0.000 description 2
- CZPWVGJYEJSRLH-UHFFFAOYSA-N Pyrimidine Chemical compound C1=CN=CN=C1 CZPWVGJYEJSRLH-UHFFFAOYSA-N 0.000 description 2
- SMWDFEZZVXVKRB-UHFFFAOYSA-N Quinoline Chemical compound N1=CC=CC2=CC=CC=C21 SMWDFEZZVXVKRB-UHFFFAOYSA-N 0.000 description 2
- 229940124639 Selective inhibitor Drugs 0.000 description 2
- 206010040047 Sepsis Diseases 0.000 description 2
- VYPSYNLAJGMNEJ-UHFFFAOYSA-N Silicium dioxide Chemical compound O=[Si]=O VYPSYNLAJGMNEJ-UHFFFAOYSA-N 0.000 description 2
- FAPWRFPIFSIZLT-UHFFFAOYSA-M Sodium chloride Chemical compound [Na+].[Cl-] FAPWRFPIFSIZLT-UHFFFAOYSA-M 0.000 description 2
- 208000006011 Stroke Diseases 0.000 description 2
- YTPLMLYBLZKORZ-UHFFFAOYSA-N Thiophene Chemical compound C=1C=CSC=1 YTPLMLYBLZKORZ-UHFFFAOYSA-N 0.000 description 2
- 208000036142 Viral infection Diseases 0.000 description 2
- 230000004913 activation Effects 0.000 description 2
- 238000007792 addition Methods 0.000 description 2
- 238000001042 affinity chromatography Methods 0.000 description 2
- 125000003342 alkenyl group Chemical group 0.000 description 2
- 230000007815 allergy Effects 0.000 description 2
- 150000001413 amino acids Chemical class 0.000 description 2
- 230000002491 angiogenic effect Effects 0.000 description 2
- 239000012911 assay medium Substances 0.000 description 2
- 210000004369 blood Anatomy 0.000 description 2
- 239000008280 blood Substances 0.000 description 2
- 201000011510 cancer Diseases 0.000 description 2
- 239000002775 capsule Substances 0.000 description 2
- 239000011203 carbon fibre reinforced carbon Substances 0.000 description 2
- 239000006285 cell suspension Substances 0.000 description 2
- 239000002299 complementary DNA Substances 0.000 description 2
- 238000010511 deprotection reaction Methods 0.000 description 2
- 230000001066 destructive effect Effects 0.000 description 2
- 206010012601 diabetes mellitus Diseases 0.000 description 2
- 150000004985 diamines Chemical class 0.000 description 2
- 239000012091 fetal bovine serum Substances 0.000 description 2
- RWSXRVCMGQZWBV-WDSKDSINSA-N glutathione Chemical compound OC(=O)[C@@H](N)CCC(=O)N[C@@H](CS)C(=O)NCC(O)=O RWSXRVCMGQZWBV-WDSKDSINSA-N 0.000 description 2
- 229930195733 hydrocarbon Natural products 0.000 description 2
- 150000002430 hydrocarbons Chemical group 0.000 description 2
- 239000005457 ice water Substances 0.000 description 2
- 238000001727 in vivo Methods 0.000 description 2
- 208000014674 injury Diseases 0.000 description 2
- AWJUIBRHMBBTKR-UHFFFAOYSA-N isoquinoline Chemical compound C1=NC=CC2=CC=CC=C21 AWJUIBRHMBBTKR-UHFFFAOYSA-N 0.000 description 2
- IVSZLXZYQVIEFR-UHFFFAOYSA-N m-xylene Chemical group CC1=CC=CC(C)=C1 IVSZLXZYQVIEFR-UHFFFAOYSA-N 0.000 description 2
- 235000019341 magnesium sulphate Nutrition 0.000 description 2
- 230000004060 metabolic process Effects 0.000 description 2
- 239000003226 mitogen Substances 0.000 description 2
- 230000004048 modification Effects 0.000 description 2
- 238000012986 modification Methods 0.000 description 2
- QNDOJFRYZPCHJA-UHFFFAOYSA-N n-[(4,6-dichloro-2-methylsulfanylpyrimidin-5-yl)methylidene]hydroxylamine Chemical compound CSC1=NC(Cl)=C(C=NO)C(Cl)=N1 QNDOJFRYZPCHJA-UHFFFAOYSA-N 0.000 description 2
- XWCCTMBMQUCLSI-UHFFFAOYSA-N n-ethyl-n-propylpropan-1-amine Chemical compound CCCN(CC)CCC XWCCTMBMQUCLSI-UHFFFAOYSA-N 0.000 description 2
- 229910052757 nitrogen Inorganic materials 0.000 description 2
- 150000007530 organic bases Chemical class 0.000 description 2
- 238000012261 overproduction Methods 0.000 description 2
- 210000003819 peripheral blood mononuclear cell Anatomy 0.000 description 2
- 102000013415 peroxidase activity proteins Human genes 0.000 description 2
- 108040007629 peroxidase activity proteins Proteins 0.000 description 2
- 239000000546 pharmaceutical excipient Substances 0.000 description 2
- 239000012071 phase Substances 0.000 description 2
- 125000001997 phenyl group Chemical group [H]C1=C([H])C([H])=C(*)C([H])=C1[H] 0.000 description 2
- 230000026731 phosphorylation Effects 0.000 description 2
- 238000006366 phosphorylation reaction Methods 0.000 description 2
- 239000012286 potassium permanganate Substances 0.000 description 2
- 230000003389 potentiating effect Effects 0.000 description 2
- 230000008569 process Effects 0.000 description 2
- 230000000770 proinflammatory effect Effects 0.000 description 2
- 230000002062 proliferating effect Effects 0.000 description 2
- 108060006633 protein kinase Proteins 0.000 description 2
- UMJSCPRVCHMLSP-UHFFFAOYSA-N pyridine Natural products COC1=CC=CN=C1 UMJSCPRVCHMLSP-UHFFFAOYSA-N 0.000 description 2
- 125000000714 pyrimidinyl group Chemical group 0.000 description 2
- XSCHRSMBECNVNS-UHFFFAOYSA-N quinoxaline Chemical compound N1=CC=NC2=CC=CC=C21 XSCHRSMBECNVNS-UHFFFAOYSA-N 0.000 description 2
- 230000009467 reduction Effects 0.000 description 2
- 238000006722 reduction reaction Methods 0.000 description 2
- 230000004044 response Effects 0.000 description 2
- 229920006395 saturated elastomer Polymers 0.000 description 2
- 230000036303 septic shock Effects 0.000 description 2
- 230000035939 shock Effects 0.000 description 2
- 150000003384 small molecules Chemical class 0.000 description 2
- JQWHASGSAFIOCM-UHFFFAOYSA-M sodium periodate Chemical compound [Na+].[O-]I(=O)(=O)=O JQWHASGSAFIOCM-UHFFFAOYSA-M 0.000 description 2
- 239000012265 solid product Substances 0.000 description 2
- 239000007858 starting material Substances 0.000 description 2
- 150000003431 steroids Chemical class 0.000 description 2
- 239000012258 stirred mixture Substances 0.000 description 2
- 229910052717 sulfur Inorganic materials 0.000 description 2
- UOGYFORZZWCMJZ-HXUWFJFHSA-N tert-butyl (3r)-3-[[4-[2,2-dimethylpropyl(methyl)amino]-6-[2-methyl-5-(methylcarbamoyl)anilino]pyrimidin-2-yl]amino]pyrrolidine-1-carboxylate Chemical compound CNC(=O)C1=CC=C(C)C(NC=2N=C(N[C@H]3CN(CC3)C(=O)OC(C)(C)C)N=C(C=2)N(C)CC(C)(C)C)=C1 UOGYFORZZWCMJZ-HXUWFJFHSA-N 0.000 description 2
- CMIBWIAICVBURI-SSDOTTSWSA-N tert-butyl (3r)-3-aminopyrrolidine-1-carboxylate Chemical compound CC(C)(C)OC(=O)N1CC[C@@H](N)C1 CMIBWIAICVBURI-SSDOTTSWSA-N 0.000 description 2
- 230000001225 therapeutic effect Effects 0.000 description 2
- FYSNRJHAOHDILO-UHFFFAOYSA-N thionyl chloride Chemical compound ClS(Cl)=O FYSNRJHAOHDILO-UHFFFAOYSA-N 0.000 description 2
- 150000003852 triazoles Chemical group 0.000 description 2
- 230000009385 viral infection Effects 0.000 description 2
- 230000003612 virological effect Effects 0.000 description 2
- 125000004502 1,2,3-oxadiazolyl group Chemical group 0.000 description 1
- 125000001399 1,2,3-triazolyl group Chemical group N1N=NC(=C1)* 0.000 description 1
- 125000001376 1,2,4-triazolyl group Chemical group N1N=C(N=C1)* 0.000 description 1
- 125000004520 1,3,4-thiadiazolyl group Chemical group 0.000 description 1
- 125000003363 1,3,5-triazinyl group Chemical group N1=C(N=CN=C1)* 0.000 description 1
- BDNKZNFMNDZQMI-UHFFFAOYSA-N 1,3-diisopropylcarbodiimide Chemical compound CC(C)N=C=NC(C)C BDNKZNFMNDZQMI-UHFFFAOYSA-N 0.000 description 1
- JPRPJUMQRZTTED-UHFFFAOYSA-N 1,3-dioxolanyl Chemical group [CH]1OCCO1 JPRPJUMQRZTTED-UHFFFAOYSA-N 0.000 description 1
- HKDFRDIIELOLTJ-UHFFFAOYSA-N 1,4-dithianyl Chemical group [CH]1CSCCS1 HKDFRDIIELOLTJ-UHFFFAOYSA-N 0.000 description 1
- ASOKPJOREAFHNY-UHFFFAOYSA-N 1-Hydroxybenzotriazole Chemical compound C1=CC=C2N(O)N=NC2=C1 ASOKPJOREAFHNY-UHFFFAOYSA-N 0.000 description 1
- FXHRAKUEZPSMLJ-UHFFFAOYSA-N 1-methyl-1,4-diazepane Chemical compound CN1CCCNCC1 FXHRAKUEZPSMLJ-UHFFFAOYSA-N 0.000 description 1
- 125000001637 1-naphthyl group Chemical group [H]C1=C([H])C([H])=C2C(*)=C([H])C([H])=C([H])C2=C1[H] 0.000 description 1
- HYZJCKYKOHLVJF-UHFFFAOYSA-N 1H-benzimidazole Chemical compound C1=CC=C2NC=NC2=C1 HYZJCKYKOHLVJF-UHFFFAOYSA-N 0.000 description 1
- DZTIFMWYYHCREC-UHFFFAOYSA-N 2,4-dichloropyrimidine-5-carbonyl chloride Chemical compound ClC(=O)C1=CN=C(Cl)N=C1Cl DZTIFMWYYHCREC-UHFFFAOYSA-N 0.000 description 1
- SXGZJKUKBWWHRA-UHFFFAOYSA-N 2-(N-morpholiniumyl)ethanesulfonate Chemical compound [O-]S(=O)(=O)CC[NH+]1CCOCC1 SXGZJKUKBWWHRA-UHFFFAOYSA-N 0.000 description 1
- NGNBDVOYPDDBFK-UHFFFAOYSA-N 2-[2,4-di(pentan-2-yl)phenoxy]acetyl chloride Chemical compound CCCC(C)C1=CC=C(OCC(Cl)=O)C(C(C)CCC)=C1 NGNBDVOYPDDBFK-UHFFFAOYSA-N 0.000 description 1
- KISWVXRQTGLFGD-UHFFFAOYSA-N 2-[[2-[[6-amino-2-[[2-[[2-[[5-amino-2-[[2-[[1-[2-[[6-amino-2-[(2,5-diamino-5-oxopentanoyl)amino]hexanoyl]amino]-5-(diaminomethylideneamino)pentanoyl]pyrrolidine-2-carbonyl]amino]-3-hydroxypropanoyl]amino]-5-oxopentanoyl]amino]-5-(diaminomethylideneamino)p Chemical compound C1CCN(C(=O)C(CCCN=C(N)N)NC(=O)C(CCCCN)NC(=O)C(N)CCC(N)=O)C1C(=O)NC(CO)C(=O)NC(CCC(N)=O)C(=O)NC(CCCN=C(N)N)C(=O)NC(CO)C(=O)NC(CCCCN)C(=O)NC(C(=O)NC(CC(C)C)C(O)=O)CC1=CC=C(O)C=C1 KISWVXRQTGLFGD-UHFFFAOYSA-N 0.000 description 1
- 102100027324 2-hydroxyacyl-CoA lyase 1 Human genes 0.000 description 1
- CAQFTFGWNKAGCI-UHFFFAOYSA-N 2-n-(2-methylphenyl)-6-[(4-pyridin-2-ylpiperazin-1-yl)methyl]-1,3,5-triazine-2,4-diamine Chemical compound CC1=CC=CC=C1NC1=NC(N)=NC(CN2CCN(CC2)C=2N=CC=CC=2)=N1 CAQFTFGWNKAGCI-UHFFFAOYSA-N 0.000 description 1
- 125000001622 2-naphthyl group Chemical group [H]C1=C([H])C([H])=C2C([H])=C(*)C([H])=C([H])C2=C1[H] 0.000 description 1
- 125000001698 2H-pyranyl group Chemical group O1C(C=CC=C1)* 0.000 description 1
- JAOJYKNVTHDLIM-UHFFFAOYSA-N 3-[[2-[2,2-dimethylpropyl(methyl)amino]-6-(4-methyl-1,4-diazepan-1-yl)pyrimidin-4-yl]amino]-n-ethoxy-4-methylbenzamide Chemical compound CCONC(=O)C1=CC=C(C)C(NC=2N=C(N=C(C=2)N2CCN(C)CCC2)N(C)CC(C)(C)C)=C1 JAOJYKNVTHDLIM-UHFFFAOYSA-N 0.000 description 1
- TWLPPYQBNUXIBA-UHFFFAOYSA-N 3-[[3-cyano-2-[2,2-dimethylpropyl(methyl)amino]-6-(1-methylpiperidin-4-yl)oxypyridin-4-yl]amino]-n,4-dimethylbenzamide Chemical compound CNC(=O)C1=CC=C(C)C(NC=2C(=C(N(C)CC(C)(C)C)N=C(OC3CCN(C)CC3)C=2)C#N)=C1 TWLPPYQBNUXIBA-UHFFFAOYSA-N 0.000 description 1
- NASBSWVDAYXHBV-UHFFFAOYSA-N 3-[[4-[2,2-dimethylpropyl(methyl)amino]-6-(4-methyl-1,4-diazepan-1-yl)pyrimidin-2-yl]amino]-n-methoxy-4-methylbenzamide Chemical compound CONC(=O)C1=CC=C(C)C(NC=2N=C(C=C(N=2)N(C)CC(C)(C)C)N2CCN(C)CCC2)=C1 NASBSWVDAYXHBV-UHFFFAOYSA-N 0.000 description 1
- UHDVMBPCTMXDQZ-UHFFFAOYSA-N 3-[[5-cyano-6-[2,2-dimethylpropyl(methyl)amino]-2-(pyrrolidin-3-ylamino)pyrimidin-4-yl]amino]-n-methoxy-4-methylbenzamide Chemical compound CONC(=O)C1=CC=C(C)C(NC=2C(=C(N(C)CC(C)(C)C)N=C(NC3CNCC3)N=2)C#N)=C1 UHDVMBPCTMXDQZ-UHFFFAOYSA-N 0.000 description 1
- RTADBVMDCJJLDI-UHFFFAOYSA-N 3-[[6-(2,2-dimethylpropylamino)-2-(methylamino)pyrimidin-4-yl]amino]-n,4-dimethylbenzamide Chemical compound CNC(=O)C1=CC=C(C)C(NC=2N=C(NC)N=C(NCC(C)(C)C)C=2)=C1 RTADBVMDCJJLDI-UHFFFAOYSA-N 0.000 description 1
- WMECFLJXJWTRGH-UHFFFAOYSA-N 3-[[6-[2,2-dimethylpropyl(methyl)amino]-2-(methylamino)pyrimidin-4-yl]amino]-n,4-dimethylbenzamide Chemical compound CNC(=O)C1=CC=C(C)C(NC=2N=C(NC)N=C(C=2)N(C)CC(C)(C)C)=C1 WMECFLJXJWTRGH-UHFFFAOYSA-N 0.000 description 1
- JMQFXVLEWYNNLS-UHFFFAOYSA-N 3-[[6-[2,2-dimethylpropyl(methyl)amino]-2-methylsulfanylpyrimidin-4-yl]amino]-n,4-dimethylbenzamide Chemical compound CNC(=O)C1=CC=C(C)C(NC=2N=C(SC)N=C(C=2)N(C)CC(C)(C)C)=C1 JMQFXVLEWYNNLS-UHFFFAOYSA-N 0.000 description 1
- VEDROZWXRQZVEJ-UHFFFAOYSA-N 3-[bromo-[2-(1-methylpiperidin-4-yl)oxypyrimidin-4-yl]amino]-n,4-dimethylbenzamide Chemical compound CNC(=O)C1=CC=C(C)C(N(Br)C=2N=C(OC3CCN(C)CC3)N=CC=2)=C1 VEDROZWXRQZVEJ-UHFFFAOYSA-N 0.000 description 1
- MKSTXMDSWOHKOR-UHFFFAOYSA-N 3-aminopyrrolidine-1-carboxylic acid Chemical compound NC1CCN(C(O)=O)C1 MKSTXMDSWOHKOR-UHFFFAOYSA-N 0.000 description 1
- 125000004364 3-pyrrolinyl group Chemical group [H]C1=C([H])C([H])([H])N(*)C1([H])[H] 0.000 description 1
- GHAWBARMICSLQS-UHFFFAOYSA-N 4,6-dichloro-2-methylsulfanyl-5-nitropyrimidine Chemical compound CSC1=NC(Cl)=C([N+]([O-])=O)C(Cl)=N1 GHAWBARMICSLQS-UHFFFAOYSA-N 0.000 description 1
- IJMVIGVHTYZQDE-UHFFFAOYSA-N 4-[2,2-dimethylpropyl(methyl)amino]-2-methylsulfonyl-6-[2-methyl-5-(1h-1,2,4-triazol-5-yl)anilino]pyrimidine-5-carbonitrile Chemical compound CC(C)(C)CN(C)C1=NC(S(C)(=O)=O)=NC(NC=2C(=CC=C(C=2)C=2NC=NN=2)C)=C1C#N IJMVIGVHTYZQDE-UHFFFAOYSA-N 0.000 description 1
- QCQCHGYLTSGIGX-GHXANHINSA-N 4-[[(3ar,5ar,5br,7ar,9s,11ar,11br,13as)-5a,5b,8,8,11a-pentamethyl-3a-[(5-methylpyridine-3-carbonyl)amino]-2-oxo-1-propan-2-yl-4,5,6,7,7a,9,10,11,11b,12,13,13a-dodecahydro-3h-cyclopenta[a]chrysen-9-yl]oxy]-2,2-dimethyl-4-oxobutanoic acid Chemical compound N([C@@]12CC[C@@]3(C)[C@]4(C)CC[C@H]5C(C)(C)[C@@H](OC(=O)CC(C)(C)C(O)=O)CC[C@]5(C)[C@H]4CC[C@@H]3C1=C(C(C2)=O)C(C)C)C(=O)C1=CN=CC(C)=C1 QCQCHGYLTSGIGX-GHXANHINSA-N 0.000 description 1
- WAISNFWNFICZOV-UHFFFAOYSA-N 4-chloro-2-methylsulfanyl-6-[2-methyl-5-(1h-1,2,4-triazol-5-yl)anilino]pyrimidine-5-carbonitrile Chemical compound CSC1=NC(Cl)=C(C#N)C(NC=2C(=CC=C(C=2)C=2NC=NN=2)C)=N1 WAISNFWNFICZOV-UHFFFAOYSA-N 0.000 description 1
- LMCNSMCHUYZUFF-UHFFFAOYSA-N 4-hydroxy-2-methylsulfanyl-5-nitro-1h-pyrimidin-6-one Chemical compound CSC1=NC(O)=C([N+]([O-])=O)C(O)=N1 LMCNSMCHUYZUFF-UHFFFAOYSA-N 0.000 description 1
- KOFBNBCOGKLUOM-UHFFFAOYSA-N 4-methyl-3-nitrobenzonitrile Chemical compound CC1=CC=C(C#N)C=C1[N+]([O-])=O KOFBNBCOGKLUOM-UHFFFAOYSA-N 0.000 description 1
- 125000001826 4H-pyranyl group Chemical group O1C(=CCC=C1)* 0.000 description 1
- YGKOOYJKBBADLL-UHFFFAOYSA-N 5-(4-methyl-3-nitrophenyl)-1h-1,2,4-triazole Chemical compound C1=C([N+]([O-])=O)C(C)=CC=C1C1=NN=CN1 YGKOOYJKBBADLL-UHFFFAOYSA-N 0.000 description 1
- 208000030507 AIDS Diseases 0.000 description 1
- ZKHQWZAMYRWXGA-KQYNXXCUSA-J ATP(4-) Chemical compound C1=NC=2C(N)=NC=NC=2N1[C@@H]1O[C@H](COP([O-])(=O)OP([O-])(=O)OP([O-])([O-])=O)[C@@H](O)[C@H]1O ZKHQWZAMYRWXGA-KQYNXXCUSA-J 0.000 description 1
- 208000031261 Acute myeloid leukaemia Diseases 0.000 description 1
- 229920000936 Agarose Polymers 0.000 description 1
- 206010002556 Ankylosing Spondylitis Diseases 0.000 description 1
- 239000004475 Arginine Substances 0.000 description 1
- 206010055128 Autoimmune neutropenia Diseases 0.000 description 1
- 210000002237 B-cell of pancreatic islet Anatomy 0.000 description 1
- 208000032791 BCR-ABL1 positive chronic myelogenous leukemia Diseases 0.000 description 1
- 208000004429 Bacillary Dysentery Diseases 0.000 description 1
- 241000713704 Bovine immunodeficiency virus Species 0.000 description 1
- 201000006474 Brain Ischemia Diseases 0.000 description 1
- SWFVRUANOZHHFE-UHFFFAOYSA-N C#CC1=CN=C(NC2=CC(C(=O)NC)=CC=C2C)N=C1N(C)CC(C)(C)C.CNC(=O)C1=CC=C(C)C(NC2=NC(NC3CCCC3)=NC=C2Br)=C1.CNC(=O)C1=CC=C(C)C(NC2=NC(OC3CCN(C)CC3)=NC=C2Br)=C1.CNC(=O)C1=CC=C(C)C(NC2=NC=CC(N(C)CC(C)(C)C)=C2Br)=C1 Chemical compound C#CC1=CN=C(NC2=CC(C(=O)NC)=CC=C2C)N=C1N(C)CC(C)(C)C.CNC(=O)C1=CC=C(C)C(NC2=NC(NC3CCCC3)=NC=C2Br)=C1.CNC(=O)C1=CC=C(C)C(NC2=NC(OC3CCN(C)CC3)=NC=C2Br)=C1.CNC(=O)C1=CC=C(C)C(NC2=NC=CC(N(C)CC(C)(C)C)=C2Br)=C1 SWFVRUANOZHHFE-UHFFFAOYSA-N 0.000 description 1
- VZXFTFAGWBIVRN-UHFFFAOYSA-N C#CC1=CN=C(NC2=CC(C(=O)NC)=CC=C2C)N=C1N(C)CC(C)(C)C.CNC(=O)C1=CC=C(C)C(NC2=NC=CC(N(C)CC(C)(C)C)=C2Br)=C1.CNC(=O)C1=CC=C(C)C(NC2=NC=CC(N(C)CC(C)(C)C)=C2N)=C1.CNC(=O)C1=CC=C(C)C(NC2=NC=CC(N(C)CC(C)(C)C)=C2NC(C)=O)=C1 Chemical compound C#CC1=CN=C(NC2=CC(C(=O)NC)=CC=C2C)N=C1N(C)CC(C)(C)C.CNC(=O)C1=CC=C(C)C(NC2=NC=CC(N(C)CC(C)(C)C)=C2Br)=C1.CNC(=O)C1=CC=C(C)C(NC2=NC=CC(N(C)CC(C)(C)C)=C2N)=C1.CNC(=O)C1=CC=C(C)C(NC2=NC=CC(N(C)CC(C)(C)C)=C2NC(C)=O)=C1 VZXFTFAGWBIVRN-UHFFFAOYSA-N 0.000 description 1
- VKFKWPXYLFZXCH-ZAHSCVQWSA-N C.C.CC(C)(C)OC(=O)N1CC[C@@H](N)C1.CONC(=O)C1=CC=C(C)C(NC2=C(C#N)C(N(C)CC(C)(C)C)=NC(N[C@@H]3CCNC3)=N2)=C1.CONC(=O)C1=CC=C(C)C(NC2=C(C#N)C(N(C)CC(C)(C)C)=NC(S(C)=O)=N2)=C1.Cl Chemical compound C.C.CC(C)(C)OC(=O)N1CC[C@@H](N)C1.CONC(=O)C1=CC=C(C)C(NC2=C(C#N)C(N(C)CC(C)(C)C)=NC(N[C@@H]3CCNC3)=N2)=C1.CONC(=O)C1=CC=C(C)C(NC2=C(C#N)C(N(C)CC(C)(C)C)=NC(S(C)=O)=N2)=C1.Cl VKFKWPXYLFZXCH-ZAHSCVQWSA-N 0.000 description 1
- MEAUVVQCQIDBLU-OIEOOTLDSA-N C.CNC(=O)C1=CC(NC2=CC(F)=NC(N[C@@H]3CCN(C(=O)OC(C)(C)C)C3)=N2)=C(C)C=C1.CNC(=O)C1=CC(NC2=CC(N(C)CC(C)(C)C)=NC(N[C@@H]3CCN(C(=O)OC(C)(C)C)C3)=N2)=C(C)C=C1.CNCC(C)(C)C.Cl Chemical compound C.CNC(=O)C1=CC(NC2=CC(F)=NC(N[C@@H]3CCN(C(=O)OC(C)(C)C)C3)=N2)=C(C)C=C1.CNC(=O)C1=CC(NC2=CC(N(C)CC(C)(C)C)=NC(N[C@@H]3CCN(C(=O)OC(C)(C)C)C3)=N2)=C(C)C=C1.CNCC(C)(C)C.Cl MEAUVVQCQIDBLU-OIEOOTLDSA-N 0.000 description 1
- DSWWSLXEJFUXJM-UHFFFAOYSA-N C=C(NC)C1=CC=C(C)C(NC2=NC(S(C)(=O)=O)=NC(N(C)CC(C)(C)C)=C2C#N)=C1.CNC(=O)C1=CC=C(C)C(NC2=NC(C)=NC(N(C)CC(C)(C)C)=C2C#N)=C1.CNC(=O)C1=CC=C(C)C(NC2=NC=CC(N(C)CC(C)(C)C)=C2N)=C1.CONC(=O)C1=CC=C(C)C(NC2=NC(OC3CCN(C)CC3)=NC(N(C)CC(C)(C)C)=C2C#N)=C1 Chemical compound C=C(NC)C1=CC=C(C)C(NC2=NC(S(C)(=O)=O)=NC(N(C)CC(C)(C)C)=C2C#N)=C1.CNC(=O)C1=CC=C(C)C(NC2=NC(C)=NC(N(C)CC(C)(C)C)=C2C#N)=C1.CNC(=O)C1=CC=C(C)C(NC2=NC=CC(N(C)CC(C)(C)C)=C2N)=C1.CONC(=O)C1=CC=C(C)C(NC2=NC(OC3CCN(C)CC3)=NC(N(C)CC(C)(C)C)=C2C#N)=C1 DSWWSLXEJFUXJM-UHFFFAOYSA-N 0.000 description 1
- WOSOVIWDIHGYEF-UHFFFAOYSA-N CC#N.CC(C)(C)CN.CCN(CC)CC.[C-]#[N+]C1=C(Cl)N=C(Cl)C(F)=C1.[C-]#[N+]C1=C(Cl)N=C(NCC(C)(C)C)C(F)=C1 Chemical compound CC#N.CC(C)(C)CN.CCN(CC)CC.[C-]#[N+]C1=C(Cl)N=C(Cl)C(F)=C1.[C-]#[N+]C1=C(Cl)N=C(NCC(C)(C)C)C(F)=C1 WOSOVIWDIHGYEF-UHFFFAOYSA-N 0.000 description 1
- OYKOMINEPKKGON-UHFFFAOYSA-N CC#N.CCN(CC)CC.CNCC(C)(C)C.Cl.[C-]#[N+]C1=C(Cl)N=C(Cl)C(F)=C1.[C-]#[N+]C1=C(Cl)N=C(N(C)CC(C)(C)C)C(F)=C1 Chemical compound CC#N.CCN(CC)CC.CNCC(C)(C)C.Cl.[C-]#[N+]C1=C(Cl)N=C(Cl)C(F)=C1.[C-]#[N+]C1=C(Cl)N=C(N(C)CC(C)(C)C)C(F)=C1 OYKOMINEPKKGON-UHFFFAOYSA-N 0.000 description 1
- ZPUNYVRYQROZLN-UHFFFAOYSA-N CC(=O)O.CNC(=O)C1=CC=C(C)C(NC2=C(C#N)C(N(C)CC(C)(C)C)=NC(S(C)(=O)=O)=N2)=C1.CNC(=O)C1=CC=C(C)C(NC2=C(C#N)C(N(C)CC(C)(C)C)=NC(SC)=N2)=C1.O=[Mn](=O)(=O)(=O)[K] Chemical compound CC(=O)O.CNC(=O)C1=CC=C(C)C(NC2=C(C#N)C(N(C)CC(C)(C)C)=NC(S(C)(=O)=O)=N2)=C1.CNC(=O)C1=CC=C(C)C(NC2=C(C#N)C(N(C)CC(C)(C)C)=NC(SC)=N2)=C1.O=[Mn](=O)(=O)(=O)[K] ZPUNYVRYQROZLN-UHFFFAOYSA-N 0.000 description 1
- YRUAVKSSYOBQII-UHFFFAOYSA-M CC(C)(C)CN(C)c(nc(NCCCN(C)C)nc1Nc2cc(C(NC)=O)ccc2C)c1[IH]([O-])=O Chemical compound CC(C)(C)CN(C)c(nc(NCCCN(C)C)nc1Nc2cc(C(NC)=O)ccc2C)c1[IH]([O-])=O YRUAVKSSYOBQII-UHFFFAOYSA-M 0.000 description 1
- LVHGRZOGMZFWDS-UHFFFAOYSA-M CC(C)(C)CN(C)c(nc(nc1Nc2cc(C(NC)=O)ccc2C)OC2CCN(C)CC2)c1[IH]([O-])=O Chemical compound CC(C)(C)CN(C)c(nc(nc1Nc2cc(C(NC)=O)ccc2C)OC2CCN(C)CC2)c1[IH]([O-])=O LVHGRZOGMZFWDS-UHFFFAOYSA-M 0.000 description 1
- PWYPBJYPZKHOLL-UHFFFAOYSA-N CC(C)(C)CN.CNC(=O)C1=CC=C(C)C(NC2=CC(Cl)=NC=N2)=C1.CNC(=O)C1=CC=C(C)C(NC2=CC(NCC(C)(C)C)=NC=N2)=C1 Chemical compound CC(C)(C)CN.CNC(=O)C1=CC=C(C)C(NC2=CC(Cl)=NC=N2)=C1.CNC(=O)C1=CC=C(C)C(NC2=CC(NCC(C)(C)C)=NC=N2)=C1 PWYPBJYPZKHOLL-UHFFFAOYSA-N 0.000 description 1
- RGIABZDBGQMABG-UHFFFAOYSA-N CC(C)(C)CN.[C-]#[N+]C1=C(Cl)N=C(NC2=C(C)C=CC(C(=O)NOC)=C2)C(F)=C1.[C-]#[N+]C1=C(NCC(C)(C)C)N=C(NC2=C(C)C=CC(C(=O)NOC)=C2)C(F)=C1 Chemical compound CC(C)(C)CN.[C-]#[N+]C1=C(Cl)N=C(NC2=C(C)C=CC(C(=O)NOC)=C2)C(F)=C1.[C-]#[N+]C1=C(NCC(C)(C)C)N=C(NC2=C(C)C=CC(C(=O)NOC)=C2)C(F)=C1 RGIABZDBGQMABG-UHFFFAOYSA-N 0.000 description 1
- OZWIOHZFEWSMPL-UHFFFAOYSA-M CC(C)(C)CNc(ncnc1Nc2cc(C(NC)=O)ccc2C)c1[IH]([O-])=O Chemical compound CC(C)(C)CNc(ncnc1Nc2cc(C(NC)=O)ccc2C)c1[IH]([O-])=O OZWIOHZFEWSMPL-UHFFFAOYSA-M 0.000 description 1
- KLTARYAJSJDRBZ-UHFFFAOYSA-N CC(C)(C)OC(=O)N1CCC(N)C1.CNC(=O)C1=CC(N)=C(C)C=C1.CNC(=O)C1=CC=C(C)C(NC2=CC(F)=NC(F)=N2)=C1.CNC(=O)C1=CC=C(C)C(NC2=CC(F)=NC(N(C)CC(C)(C)C)=N2)=C1.CNCC(C)(C)C.FC1=CC(F)=NC(F)=N1 Chemical compound CC(C)(C)OC(=O)N1CCC(N)C1.CNC(=O)C1=CC(N)=C(C)C=C1.CNC(=O)C1=CC=C(C)C(NC2=CC(F)=NC(F)=N2)=C1.CNC(=O)C1=CC=C(C)C(NC2=CC(F)=NC(N(C)CC(C)(C)C)=N2)=C1.CNCC(C)(C)C.FC1=CC(F)=NC(F)=N1 KLTARYAJSJDRBZ-UHFFFAOYSA-N 0.000 description 1
- STZKKIRMWDNOAB-UHFFFAOYSA-N CC(C)(C)OC(=O)N1CCC(N)C1.[C-]#[N+]C1=C(N(C)CC(C)(C)C)N=C(NC2CCN(C(=O)OC(C)(C)C)C2)N=C1NC1=CC(C(=O)NC)=CC=C1C.[C-]#[N+]C1=C(N(C)CC(C)(C)C)N=C(NC2CCNC2)N=C1NC1=CC(C(=O)NC)=CC=C1C.[C-]#[N+]C1=C(N(C)CC(C)(C)C)N=C(S(C)(=O)=O)N=C1NC1=CC(C(=O)NC)=CC=C1C Chemical compound CC(C)(C)OC(=O)N1CCC(N)C1.[C-]#[N+]C1=C(N(C)CC(C)(C)C)N=C(NC2CCN(C(=O)OC(C)(C)C)C2)N=C1NC1=CC(C(=O)NC)=CC=C1C.[C-]#[N+]C1=C(N(C)CC(C)(C)C)N=C(NC2CCNC2)N=C1NC1=CC(C(=O)NC)=CC=C1C.[C-]#[N+]C1=C(N(C)CC(C)(C)C)N=C(S(C)(=O)=O)N=C1NC1=CC(C(=O)NC)=CC=C1C STZKKIRMWDNOAB-UHFFFAOYSA-N 0.000 description 1
- YGPJWFYJZVTRKO-FDWYJAJHSA-N CC(C)(C)OC(=O)N1CC[C@@H](N)C1.CC1=C(NC2=C(C#N)C(N(C)CC(C)(C)C)=NC(N[C@@H]3CCNC3)=N2)C=C(C2=NN=CN2)C=C1.CC1=C(NC2=C(C#N)C(N(C)CC(C)(C)C)=NC(S(C)(=O)=O)=N2)C=C(C2=NN=CN2)C=C1.Cl Chemical compound CC(C)(C)OC(=O)N1CC[C@@H](N)C1.CC1=C(NC2=C(C#N)C(N(C)CC(C)(C)C)=NC(N[C@@H]3CCNC3)=N2)C=C(C2=NN=CN2)C=C1.CC1=C(NC2=C(C#N)C(N(C)CC(C)(C)C)=NC(S(C)(=O)=O)=N2)C=C(C2=NN=CN2)C=C1.Cl YGPJWFYJZVTRKO-FDWYJAJHSA-N 0.000 description 1
- YCPGRIWNIYFQIU-PVOIQKRISA-N CC(C)(C)OC(=O)N1CC[C@@H](N)C1.CNC(=O)C1=CC=C(C)C(NC2=C(Br)C=NC(Cl)=N2)=C1.CNC(=O)C1=CC=C(C)C(NC2=C(Br)C=NC(N[C@@H]3CCNC3)=N2)=C1 Chemical compound CC(C)(C)OC(=O)N1CC[C@@H](N)C1.CNC(=O)C1=CC=C(C)C(NC2=C(Br)C=NC(Cl)=N2)=C1.CNC(=O)C1=CC=C(C)C(NC2=C(Br)C=NC(N[C@@H]3CCNC3)=N2)=C1 YCPGRIWNIYFQIU-PVOIQKRISA-N 0.000 description 1
- AYRQUWYVTJWDOO-UHFFFAOYSA-N CC1=C(N)C=C(C2=NN=CN2)C=C1.CC1=C(NC2=C([N+](=O)[O-])C(N(C)CC(C)(C)C)=NC=N2)C=C(C2=NN=CN2)C=C1.CN(CC(C)(C)C)C1=C([N+](=O)[O-])C(Cl)=NC=N1 Chemical compound CC1=C(N)C=C(C2=NN=CN2)C=C1.CC1=C(NC2=C([N+](=O)[O-])C(N(C)CC(C)(C)C)=NC=N2)C=C(C2=NN=CN2)C=C1.CN(CC(C)(C)C)C1=C([N+](=O)[O-])C(Cl)=NC=N1 AYRQUWYVTJWDOO-UHFFFAOYSA-N 0.000 description 1
- MHBHGPKTNIFKLY-NMRXSTGRSA-N CC1=C(NC2=C(C#N)C(N(C)CC(C)(C)C)=NC(N[C@@H]3CCNC3)=N2)C=C(C2=NN=CN2)C=C1.CC1=CC=C(C2=NN=CN2)C=C1N.CSC1=NC(Cl)=C(C#N)C(Cl)=N1.CSC1=NC(NC2=C(C)C=CC(C3=NN=CN3)=C2)=C(C#N)C(Cl)=N1.Cl Chemical compound CC1=C(NC2=C(C#N)C(N(C)CC(C)(C)C)=NC(N[C@@H]3CCNC3)=N2)C=C(C2=NN=CN2)C=C1.CC1=CC=C(C2=NN=CN2)C=C1N.CSC1=NC(Cl)=C(C#N)C(Cl)=N1.CSC1=NC(NC2=C(C)C=CC(C3=NN=CN3)=C2)=C(C#N)C(Cl)=N1.Cl MHBHGPKTNIFKLY-NMRXSTGRSA-N 0.000 description 1
- LFIOCXPKLFKWEV-UHFFFAOYSA-N CC1=C(NC2=C(C#N)C(N(C)CC(C)(C)C)=NC(S(C)(=O)=O)=N2)C=C(C2=NN=CN2)C=C1.CSC1=NC(NC2=C(C)C=CC(C3=NN=CN3)=C2)=C(C#N)C(N(C)CC(C)(C)C)=N1 Chemical compound CC1=C(NC2=C(C#N)C(N(C)CC(C)(C)C)=NC(S(C)(=O)=O)=N2)C=C(C2=NN=CN2)C=C1.CSC1=NC(NC2=C(C)C=CC(C3=NN=CN3)=C2)=C(C#N)C(N(C)CC(C)(C)C)=N1 LFIOCXPKLFKWEV-UHFFFAOYSA-N 0.000 description 1
- IQLVNYKBFVKISY-UHFFFAOYSA-N CC1=C(NC2=C(C#N)C(N(C)CC(C)(C)C)=NC=N2)C=C(C2=NN=CN2)C=C1.CSC1=NC(NC2=C(C)C=CC(C3=NN=CN3)=C2)=C(C#N)C(N(C)CC(C)(C)C)=N1 Chemical compound CC1=C(NC2=C(C#N)C(N(C)CC(C)(C)C)=NC=N2)C=C(C2=NN=CN2)C=C1.CSC1=NC(NC2=C(C)C=CC(C3=NN=CN3)=C2)=C(C#N)C(N(C)CC(C)(C)C)=N1 IQLVNYKBFVKISY-UHFFFAOYSA-N 0.000 description 1
- ZYYNDAURHYNVJD-UHFFFAOYSA-N CC1=C(NC2=C(N)C(N(C)CC(C)(C)C)=NC=N2)C=C(C2=NN=CN2)C=C1.CC1=C(NC2=C([N+](=O)[O-])C(N(C)CC(C)(C)C)=NC=N2)C=C(C2=NN=CN2)C=C1.CO Chemical compound CC1=C(NC2=C(N)C(N(C)CC(C)(C)C)=NC=N2)C=C(C2=NN=CN2)C=C1.CC1=C(NC2=C([N+](=O)[O-])C(N(C)CC(C)(C)C)=NC=N2)C=C(C2=NN=CN2)C=C1.CO ZYYNDAURHYNVJD-UHFFFAOYSA-N 0.000 description 1
- IXRVLUWIEMIGTC-UHFFFAOYSA-N CC1=C(NC2=C([N+](=O)[O-])C(N(C)CC(C)(C)C)=NC=N2)C=C(C2=NN=CN2)C=C1.CN(CC(C)(C)C)C1=C([N+](=O)[O-])C(Cl)=NC=N1.CNCC(C)(C)C.Cl.O=[N+]([O-])C1=C(Cl)N=CN=C1Cl Chemical compound CC1=C(NC2=C([N+](=O)[O-])C(N(C)CC(C)(C)C)=NC=N2)C=C(C2=NN=CN2)C=C1.CN(CC(C)(C)C)C1=C([N+](=O)[O-])C(Cl)=NC=N1.CNCC(C)(C)C.Cl.O=[N+]([O-])C1=C(Cl)N=CN=C1Cl IXRVLUWIEMIGTC-UHFFFAOYSA-N 0.000 description 1
- UYFPNUZLSGFGLS-UHFFFAOYSA-N CC1=CC=C(C#N)C=C1[N+](=O)[O-].CCO.CCOC(=N)C1=CC=C(C)C([N+](=O)[O-])=C1.Cl Chemical compound CC1=CC=C(C#N)C=C1[N+](=O)[O-].CCO.CCOC(=N)C1=CC=C(C)C([N+](=O)[O-])=C1.Cl UYFPNUZLSGFGLS-UHFFFAOYSA-N 0.000 description 1
- KLIQLCUBDBEBRN-UHFFFAOYSA-N CC1=CC=C(C2=NN=CC2)C=C1NC1=C(N)C(N(C)CC(C)(C)C)=NC=N1.CC1=CC=C(C2=NN=CN2)C=C1NC1=C(N(=O)[O-])C(N(C)CC(C)(C)C)=NC=N1.CNC(=O)C1=CC(NC2=NC(NC3CCCC3)=NC(N(C)CC(C)(C)C)=C2C#N)=C(C)C=C1.CNC(=O)C1=CC=C(C)C(NC2=NC=CC(N(C)CC(C)(C)C)=C2NC(C)=O)=C1 Chemical compound CC1=CC=C(C2=NN=CC2)C=C1NC1=C(N)C(N(C)CC(C)(C)C)=NC=N1.CC1=CC=C(C2=NN=CN2)C=C1NC1=C(N(=O)[O-])C(N(C)CC(C)(C)C)=NC=N1.CNC(=O)C1=CC(NC2=NC(NC3CCCC3)=NC(N(C)CC(C)(C)C)=C2C#N)=C(C)C=C1.CNC(=O)C1=CC=C(C)C(NC2=NC=CC(N(C)CC(C)(C)C)=C2NC(C)=O)=C1 KLIQLCUBDBEBRN-UHFFFAOYSA-N 0.000 description 1
- PLHSIMFVKWMMGQ-UHFFFAOYSA-N CC1=CC=C(C2=NN=CN2)C=C1NC1=C(C#N)C(N(C)CC(C)(C)C)=NC(NC2CCNC2)=N1.CNC(=O)C1=CC=C(C)C(NC2=NC(N3CCCN(C)CC3)=NC(N(C)CC(C)(C)C)=C2C#N)=C1.CNC(=O)C1=CC=C(C)C(NC2=NC(OCCC3CCCN3C)=NC(N(C)CC(C)(C)C)=C2C#N)=C1.CONC(=O)C1=CC=C(C)C(NC2=NC(N3CCCN(C)CC3)=NC(N(C)CC(C)(C)C)=C2C#N)=C1 Chemical compound CC1=CC=C(C2=NN=CN2)C=C1NC1=C(C#N)C(N(C)CC(C)(C)C)=NC(NC2CCNC2)=N1.CNC(=O)C1=CC=C(C)C(NC2=NC(N3CCCN(C)CC3)=NC(N(C)CC(C)(C)C)=C2C#N)=C1.CNC(=O)C1=CC=C(C)C(NC2=NC(OCCC3CCCN3C)=NC(N(C)CC(C)(C)C)=C2C#N)=C1.CONC(=O)C1=CC=C(C)C(NC2=NC(N3CCCN(C)CC3)=NC(N(C)CC(C)(C)C)=C2C#N)=C1 PLHSIMFVKWMMGQ-UHFFFAOYSA-N 0.000 description 1
- FFKJVMIGIQCRIL-UHFFFAOYSA-N CC1=CC=C(C2=NN=CN2)C=C1NC1=C(C#N)C(N(C)CC(C)(C)C)=NC=N1.CCC1=NC(N(C)CC(C)(C)C)=C(Cl)C(NC2=CC(C(=O)NC)=CC=C2C)=N1.CCC1=NC(NC)=C(Cl)C(NC2=CC(C(=O)NC)=CC=C2C)=N1.CNC(=O)C1=CC=C(C)C(NC2=NC(N(C)CC(C)(C)C)=NC(N(C)CC(C)(C)C)=C2Cl)=C1 Chemical compound CC1=CC=C(C2=NN=CN2)C=C1NC1=C(C#N)C(N(C)CC(C)(C)C)=NC=N1.CCC1=NC(N(C)CC(C)(C)C)=C(Cl)C(NC2=CC(C(=O)NC)=CC=C2C)=N1.CCC1=NC(NC)=C(Cl)C(NC2=CC(C(=O)NC)=CC=C2C)=N1.CNC(=O)C1=CC=C(C)C(NC2=NC(N(C)CC(C)(C)C)=NC(N(C)CC(C)(C)C)=C2Cl)=C1 FFKJVMIGIQCRIL-UHFFFAOYSA-N 0.000 description 1
- DZSHGWKBCCHJOC-UHFFFAOYSA-N CC1=CC=C(C2=NN=CN2)C=C1[N+](=O)[O-].CCOC(=N)C1=CC=C(C)C([N+](=O)[O-])=C1.Cl.[H]C(=O)NN Chemical compound CC1=CC=C(C2=NN=CN2)C=C1[N+](=O)[O-].CCOC(=N)C1=CC=C(C)C([N+](=O)[O-])=C1.Cl.[H]C(=O)NN DZSHGWKBCCHJOC-UHFFFAOYSA-N 0.000 description 1
- PMDQNNFEFLCILB-UHFFFAOYSA-N CC1CCC2CCCCC2C1.CC1Cc2ccccc2C1 Chemical compound CC1CCC2CCCCC2C1.CC1Cc2ccccc2C1 PMDQNNFEFLCILB-UHFFFAOYSA-N 0.000 description 1
- YJKFOZMSXHHTHY-UHFFFAOYSA-N CCC1=NC(N(C)CC(C)(C)C)=C(Br)C(NC2=CC(C(=O)NC)=CC=C2C)=N1.CCC1=NC(N(C)CC(C)(C)C)=C(Br)C(NC2=CC(C(=O)NC)=CC=C2C)=N1.CCC1=NC(N(C)CC(C)(C)C)=C([N+](=O)[O-])C(NC2=CC(C(=O)NC)=CC=C2C)=N1.CNC(=O)C1=CC=C(C)C(NC2=NC(NCCCN(C)C)=NC(N(C)CC(C)(C)C)=C2C#N)=C1 Chemical compound CCC1=NC(N(C)CC(C)(C)C)=C(Br)C(NC2=CC(C(=O)NC)=CC=C2C)=N1.CCC1=NC(N(C)CC(C)(C)C)=C(Br)C(NC2=CC(C(=O)NC)=CC=C2C)=N1.CCC1=NC(N(C)CC(C)(C)C)=C([N+](=O)[O-])C(NC2=CC(C(=O)NC)=CC=C2C)=N1.CNC(=O)C1=CC=C(C)C(NC2=NC(NCCCN(C)C)=NC(N(C)CC(C)(C)C)=C2C#N)=C1 YJKFOZMSXHHTHY-UHFFFAOYSA-N 0.000 description 1
- ZMHJWNVVARFOSE-UHFFFAOYSA-N CCC1=NC(N(C)CC(C)(C)C)=C(C#N)C(NC2=CC(C(=O)NC)=CC=C2C)=N1.CNC(=O)C1=CC=C(C)C(NC2=NC(OCC(C)(C)CN(C)C)=NC(N(C)CC(C)(C)C)=C2C#N)=C1.CNC(=O)C1=CC=C(C)C(NC2=NC(OCC3CCCN(C)C3)=NC(N(C)CC(C)(C)C)=C2C#N)=C1.CNC(=O)C1=CC=C(C)C(NC2=NC(OCCCN(C)C)=NC(N(C)CC(C)(C)C)=C2C#N)=C1 Chemical compound CCC1=NC(N(C)CC(C)(C)C)=C(C#N)C(NC2=CC(C(=O)NC)=CC=C2C)=N1.CNC(=O)C1=CC=C(C)C(NC2=NC(OCC(C)(C)CN(C)C)=NC(N(C)CC(C)(C)C)=C2C#N)=C1.CNC(=O)C1=CC=C(C)C(NC2=NC(OCC3CCCN(C)C3)=NC(N(C)CC(C)(C)C)=C2C#N)=C1.CNC(=O)C1=CC=C(C)C(NC2=NC(OCCCN(C)C)=NC(N(C)CC(C)(C)C)=C2C#N)=C1 ZMHJWNVVARFOSE-UHFFFAOYSA-N 0.000 description 1
- ZWSXCVZHCOODDZ-UHFFFAOYSA-N CCC1=NC(N(C)CC(C)(C)C)=C(C#N)C(NC2=CC(C(=O)NC)=CC=C2C)=N1.CNC(=O)C1=CC=C(C)C(NC2=NC=CC(N(C)CC(C)(C)C)=C2C#N)=C1.CNC(=O)C1=CC=C(C)C(NC2=NC=NC(NCC(C)(C)C)=C2Br)=C1.CNC(=O)C1=CC=C(C)C(NC2=NC=NC(NCC(C)(C)C)=C2C#N)=C1 Chemical compound CCC1=NC(N(C)CC(C)(C)C)=C(C#N)C(NC2=CC(C(=O)NC)=CC=C2C)=N1.CNC(=O)C1=CC=C(C)C(NC2=NC=CC(N(C)CC(C)(C)C)=C2C#N)=C1.CNC(=O)C1=CC=C(C)C(NC2=NC=NC(NCC(C)(C)C)=C2Br)=C1.CNC(=O)C1=CC=C(C)C(NC2=NC=NC(NCC(C)(C)C)=C2C#N)=C1 ZWSXCVZHCOODDZ-UHFFFAOYSA-N 0.000 description 1
- JSMJNEOTIMEMDH-UHFFFAOYSA-N CCC1=NC(NCC(C)(C)C)=C(Br)C(NC2=CC(C(=O)NC)=CC=C2C)=N1.CCN(C)C1=NC(OC2CCN(C)CC2)=NC(NC2=CC(C(=O)NC)=CC=C2C)=C1C#N.CNC(=O)C1=CC=C(C)C(NC2=NC(NCCCN(C)C)=NC(N(C)CC(C)(C)C)=C2[N+](=O)[O-])=C1.CNC(=O)C1=CC=C(C)C(NC2=NC(OC3CCN(C)CC3)=NC(N(C)C)=C2C#N)=C1 Chemical compound CCC1=NC(NCC(C)(C)C)=C(Br)C(NC2=CC(C(=O)NC)=CC=C2C)=N1.CCN(C)C1=NC(OC2CCN(C)CC2)=NC(NC2=CC(C(=O)NC)=CC=C2C)=C1C#N.CNC(=O)C1=CC=C(C)C(NC2=NC(NCCCN(C)C)=NC(N(C)CC(C)(C)C)=C2[N+](=O)[O-])=C1.CNC(=O)C1=CC=C(C)C(NC2=NC(OC3CCN(C)CC3)=NC(N(C)C)=C2C#N)=C1 JSMJNEOTIMEMDH-UHFFFAOYSA-N 0.000 description 1
- MSVDSWYRUACXRL-UHFFFAOYSA-N CCC1=NC(NCC(C)(C)C)=C(C#N)C(NC2=CC(C(=O)NC)=CC=C2C)=N1.CNC(=O)C1=CC=C(C)C(NC2=NC(NCC(C)(C)CN(C)C)=NC(N(C)CC(C)(C)C)=C2C#N)=C1.CNC(=O)C1=CC=C(C)C(NC2=NC(NCCC3CCCN3C)=NC(N(C)CC(C)(C)C)=C2C#N)=C1.CSC1=NC(NC2=CC(C3=NN=CN3)=CC=C2C)=C(C#N)C(N(C)CC(C)(C)C)=N1 Chemical compound CCC1=NC(NCC(C)(C)C)=C(C#N)C(NC2=CC(C(=O)NC)=CC=C2C)=N1.CNC(=O)C1=CC=C(C)C(NC2=NC(NCC(C)(C)CN(C)C)=NC(N(C)CC(C)(C)C)=C2C#N)=C1.CNC(=O)C1=CC=C(C)C(NC2=NC(NCCC3CCCN3C)=NC(N(C)CC(C)(C)C)=C2C#N)=C1.CSC1=NC(NC2=CC(C3=NN=CN3)=CC=C2C)=C(C#N)C(N(C)CC(C)(C)C)=N1 MSVDSWYRUACXRL-UHFFFAOYSA-N 0.000 description 1
- CUDWFDXOTSPPDG-UHFFFAOYSA-N CCCN(C)C1=NC(OC2CCN(C)CC2)=NC(NC2=CC(C(=O)NC)=CC=C2C)=C1C#N Chemical compound CCCN(C)C1=NC(OC2CCN(C)CC2)=NC(NC2=CC(C(=O)NC)=CC=C2C)=C1C#N CUDWFDXOTSPPDG-UHFFFAOYSA-N 0.000 description 1
- OJQCFEHRPFSOCY-UHFFFAOYSA-N CCCN(C)C1=NC(OC2CCN(C)CC2)=NC(NC2=CC(C(=O)NC)=CC=C2C)=C1C#N.CNC(=O)C1=CC=C(C)C(NC2=NC(OC3CCN(C)CC3)=NC(N(C)C(C)C)=C2C#N)=C1.CNC(=O)C1=CC=C(C)C(NC2=NC(OC3CCN(C)CC3)=NC(N(C)CC(C)(C)C)=C2[N+](=O)[O-])=C1.CNC(=O)C1=CC=C(C)C(NC2=NC(OC3CCN(C)CC3)=NC(N(C)CC(C)C)=C2C#N)=C1 Chemical compound CCCN(C)C1=NC(OC2CCN(C)CC2)=NC(NC2=CC(C(=O)NC)=CC=C2C)=C1C#N.CNC(=O)C1=CC=C(C)C(NC2=NC(OC3CCN(C)CC3)=NC(N(C)C(C)C)=C2C#N)=C1.CNC(=O)C1=CC=C(C)C(NC2=NC(OC3CCN(C)CC3)=NC(N(C)CC(C)(C)C)=C2[N+](=O)[O-])=C1.CNC(=O)C1=CC=C(C)C(NC2=NC(OC3CCN(C)CC3)=NC(N(C)CC(C)C)=C2C#N)=C1 OJQCFEHRPFSOCY-UHFFFAOYSA-N 0.000 description 1
- OUDZWGXQOUKBGV-UHFFFAOYSA-N CCN(C)C1=NC(OC2CCN(C)CC2)=NC(NC2=CC(C(=O)NC)=CC=C2C)=C1C#N Chemical compound CCN(C)C1=NC(OC2CCN(C)CC2)=NC(NC2=CC(C(=O)NC)=CC=C2C)=C1C#N OUDZWGXQOUKBGV-UHFFFAOYSA-N 0.000 description 1
- XEKZDGGRTQSDIU-UHFFFAOYSA-N CCONC(=O)C1=CC(NC2=NC(N(C)CC(C)(C)C)=NC(N3CCCN(C)CC3)=C2)=C(C)C=C1.CCONC(=O)C1=CC(NC2=NC(N(C)CC(C)(C)C)=NC(N3CCCN(C)CC3)=C2C#N)=C(C)C=C1 Chemical compound CCONC(=O)C1=CC(NC2=NC(N(C)CC(C)(C)C)=NC(N3CCCN(C)CC3)=C2)=C(C)C=C1.CCONC(=O)C1=CC(NC2=NC(N(C)CC(C)(C)C)=NC(N3CCCN(C)CC3)=C2C#N)=C(C)C=C1 XEKZDGGRTQSDIU-UHFFFAOYSA-N 0.000 description 1
- BIRZRXOPDQNIFC-UHFFFAOYSA-N CCONC(=O)C1=CC=C(C)C(NC2=NC(N(C)CC(C)(C)C)=NC(N3CCCN(C)CC3)=C2C#N)=C1.CONC(=O)C1=CC=C(C)C(NC2=C(F)C=C(C#N)C(N(C)CC(C)(C)C)=N2)=C1.CONC(=O)C1=CC=C(C)C(NC2=NC(N(C)CC(C)(C)C)=C(C#N)C(N3CCCN(C)CC3)=N2)=C1.[C-]#[N+]C1=C(NC2=CC(C(=O)NOC)=CC=C2C)N=C(NCC(C)(C)C)C(F)=C1 Chemical compound CCONC(=O)C1=CC=C(C)C(NC2=NC(N(C)CC(C)(C)C)=NC(N3CCCN(C)CC3)=C2C#N)=C1.CONC(=O)C1=CC=C(C)C(NC2=C(F)C=C(C#N)C(N(C)CC(C)(C)C)=N2)=C1.CONC(=O)C1=CC=C(C)C(NC2=NC(N(C)CC(C)(C)C)=C(C#N)C(N3CCCN(C)CC3)=N2)=C1.[C-]#[N+]C1=C(NC2=CC(C(=O)NOC)=CC=C2C)N=C(NCC(C)(C)C)C(F)=C1 BIRZRXOPDQNIFC-UHFFFAOYSA-N 0.000 description 1
- LWJPWIMDBLYXNR-XRSZJZRISA-N CClC(=O)O.CSC1=NC(Cl)=C(/C=N/O)C(Cl)=N1.CSC1=NC(Cl)=C(C=O)C(Cl)=N1.NO.O Chemical compound CClC(=O)O.CSC1=NC(Cl)=C(/C=N/O)C(Cl)=N1.CSC1=NC(Cl)=C(C=O)C(Cl)=N1.NO.O LWJPWIMDBLYXNR-XRSZJZRISA-N 0.000 description 1
- ZTVFHQDJEOPCKC-UHFFFAOYSA-N CN(C)CCCN.CNC(=O)C1=CC(NC2=C([N+](=O)[O-])C(N(C)CC(C)(C)C)=NC(NCCCN(C)C)=N2)=C(C)C=C1.CNC(=O)C1=CC(NC2=C([N+](=O)[O-])C(N(C)CC(C)(C)C)=NC(S(C)(=O)=O)=N2)=C(C)C=C1 Chemical compound CN(C)CCCN.CNC(=O)C1=CC(NC2=C([N+](=O)[O-])C(N(C)CC(C)(C)C)=NC(NCCCN(C)C)=N2)=C(C)C=C1.CNC(=O)C1=CC(NC2=C([N+](=O)[O-])C(N(C)CC(C)(C)C)=NC(S(C)(=O)=O)=N2)=C(C)C=C1 ZTVFHQDJEOPCKC-UHFFFAOYSA-N 0.000 description 1
- VRMYLVAUCCECFS-UHFFFAOYSA-N CN(CC(C)(C)C)C1=C([N+](=O)[O-])C(Cl)=NC=N1.CNC(=O)C1=CC=C(C)C(N)=C1.CNC(=O)C1=CC=C(C)C(NC2=NC=NC(N(C)CC(C)(C)C)=C2[N+](=O)[O-])=C1 Chemical compound CN(CC(C)(C)C)C1=C([N+](=O)[O-])C(Cl)=NC=N1.CNC(=O)C1=CC=C(C)C(N)=C1.CNC(=O)C1=CC=C(C)C(NC2=NC=NC(N(C)CC(C)(C)C)=C2[N+](=O)[O-])=C1 VRMYLVAUCCECFS-UHFFFAOYSA-N 0.000 description 1
- WYXCDQQFEDFPSK-UHFFFAOYSA-N CN.CNC(=O)C1=CC=C(C)C(NC2=C(C#N)C(N(C)CC(C)(C)C)=NC(NC)=N2)=C1.CNC(=O)C1=CC=C(C)C(NC2=C(C#N)C(N(C)CC(C)(C)C)=NC(S(C)(=O)=O)=N2)=C1 Chemical compound CN.CNC(=O)C1=CC=C(C)C(NC2=C(C#N)C(N(C)CC(C)(C)C)=NC(NC)=N2)=C1.CNC(=O)C1=CC=C(C)C(NC2=C(C#N)C(N(C)CC(C)(C)C)=NC(S(C)(=O)=O)=N2)=C1 WYXCDQQFEDFPSK-UHFFFAOYSA-N 0.000 description 1
- KGBMGZJFHMFRLH-UHFFFAOYSA-N CN.CNC1=NC(NC2=CC(C(=O)NOC)=CC=C2C)=C(C#N)C(N(C)CC(C)(C)C)=N1.CONC(=O)C1=CC=C(C)C(NC2=C(C#N)C(N(C)CC(C)(C)C)=NC(S(C)=O)=N2)=C1 Chemical compound CN.CNC1=NC(NC2=CC(C(=O)NOC)=CC=C2C)=C(C#N)C(N(C)CC(C)(C)C)=N1.CONC(=O)C1=CC=C(C)C(NC2=C(C#N)C(N(C)CC(C)(C)C)=NC(S(C)=O)=N2)=C1 KGBMGZJFHMFRLH-UHFFFAOYSA-N 0.000 description 1
- FSCIPNINVGZTBT-UHFFFAOYSA-N CN1CCC(O)CC1.CNC(=O)C1=CC(NC2=C([N+](=O)[O-])C(N(C)CC(C)(C)C)=NC(OC3CCN(C)CC3)=N2)=C(C)C=C1.CNC(=O)C1=CC(NC2=C([N+](=O)[O-])C(N(C)CC(C)(C)C)=NC(S(C)(=O)=O)=N2)=C(C)C=C1 Chemical compound CN1CCC(O)CC1.CNC(=O)C1=CC(NC2=C([N+](=O)[O-])C(N(C)CC(C)(C)C)=NC(OC3CCN(C)CC3)=N2)=C(C)C=C1.CNC(=O)C1=CC(NC2=C([N+](=O)[O-])C(N(C)CC(C)(C)C)=NC(S(C)(=O)=O)=N2)=C(C)C=C1 FSCIPNINVGZTBT-UHFFFAOYSA-N 0.000 description 1
- BGHRYUVRGKLNPT-UHFFFAOYSA-N CN1CCC(O)CC1.CNC(=O)C1=CC=C(C)C(NC2=C(Br)C=NC(Cl)=N2)=C1.CNC(=O)C1=CC=C(C)C(NC2=C(Br)C=NC(OC3CCN(C)CC3)=N2)=C1 Chemical compound CN1CCC(O)CC1.CNC(=O)C1=CC=C(C)C(NC2=C(Br)C=NC(Cl)=N2)=C1.CNC(=O)C1=CC=C(C)C(NC2=C(Br)C=NC(OC3CCN(C)CC3)=N2)=C1 BGHRYUVRGKLNPT-UHFFFAOYSA-N 0.000 description 1
- FBXFLYMVUPWCSC-UHFFFAOYSA-N CN1CCC(O)CC1.[C-]#[N+]C1=C(N(C)CC(C)(C)C)N=C(OC2CCN(C)CC2)N=C1NC1=CC(C(=O)NC)=CC=C1C.[C-]#[N+]C1=C(N(C)CC(C)(C)C)N=C(S(C)(=O)=O)N=C1NC1=CC(C(=O)NC)=CC=C1C Chemical compound CN1CCC(O)CC1.[C-]#[N+]C1=C(N(C)CC(C)(C)C)N=C(OC2CCN(C)CC2)N=C1NC1=CC(C(=O)NC)=CC=C1C.[C-]#[N+]C1=C(N(C)CC(C)(C)C)N=C(S(C)(=O)=O)N=C1NC1=CC(C(=O)NC)=CC=C1C FBXFLYMVUPWCSC-UHFFFAOYSA-N 0.000 description 1
- ZSNQCVYRWIXPBX-UHFFFAOYSA-N CN1CCCNCC1.CNC(=O)C1=CC=C(C)C(NC2=C(C#N)C(N(C)CC(C)(C)C)=NC(N3CCCN(C)CC3)=N2)=C1.CNC(=O)C1=CC=C(C)C(NC2=C(C#N)C(N(C)CC(C)(C)C)=NC(S(C)(=O)=O)=N2)=C1 Chemical compound CN1CCCNCC1.CNC(=O)C1=CC=C(C)C(NC2=C(C#N)C(N(C)CC(C)(C)C)=NC(N3CCCN(C)CC3)=N2)=C1.CNC(=O)C1=CC=C(C)C(NC2=C(C#N)C(N(C)CC(C)(C)C)=NC(S(C)(=O)=O)=N2)=C1 ZSNQCVYRWIXPBX-UHFFFAOYSA-N 0.000 description 1
- YQAOAFCCDBIECS-UHFFFAOYSA-N CN1CCCNCC1.CONC(=O)C1=CC=C(C)C(NC2=C(C#N)C(N(C)CC(C)(C)C)=NC(N3CCCN(C)CC3)=N2)=C1.CONC(=O)C1=CC=C(C)C(NC2=C(C#N)C(N(C)CC(C)(C)C)=NC(S(C)=O)=N2)=C1 Chemical compound CN1CCCNCC1.CONC(=O)C1=CC=C(C)C(NC2=C(C#N)C(N(C)CC(C)(C)C)=NC(N3CCCN(C)CC3)=N2)=C1.CONC(=O)C1=CC=C(C)C(NC2=C(C#N)C(N(C)CC(C)(C)C)=NC(S(C)=O)=N2)=C1 YQAOAFCCDBIECS-UHFFFAOYSA-N 0.000 description 1
- LTTODTZFNJSHMR-UHFFFAOYSA-N CNC(=O)C1=CC(N)=C(C)C=C1.CNCC(C)(C)C.O=[Mn](=O)(=O)(=O)[K].[C-]#[N+]C1=C(Cl)N=C(SC)N=C1Cl.[C-]#[N+]C1=C(Cl)N=C(SC)N=C1NC1=CC(C(=O)NC)=CC=C1C.[C-]#[N+]C1=C(N(C)CC(C)(C)C)N=C(SC)N=C1NC1=CC(C(=O)NC)=CC=C1C Chemical compound CNC(=O)C1=CC(N)=C(C)C=C1.CNCC(C)(C)C.O=[Mn](=O)(=O)(=O)[K].[C-]#[N+]C1=C(Cl)N=C(SC)N=C1Cl.[C-]#[N+]C1=C(Cl)N=C(SC)N=C1NC1=CC(C(=O)NC)=CC=C1C.[C-]#[N+]C1=C(N(C)CC(C)(C)C)N=C(SC)N=C1NC1=CC(C(=O)NC)=CC=C1C LTTODTZFNJSHMR-UHFFFAOYSA-N 0.000 description 1
- CGSUKJLRGMEMPA-GPIYMFQDSA-N CNC(=O)C1=CC(NC2=C(C#N)C(N(C)CC(C)(C)C)=NC(N[C@@H]3CCNC3)=N2)=C(C)C=C1.CNC(=O)C1=CC(NC2=CC(N(C)CC(C)(C)C)=NC(N[C@@H]3CCN(C(=O)OC(C)(C)C)C3)=N2)=C(C)C=C1 Chemical compound CNC(=O)C1=CC(NC2=C(C#N)C(N(C)CC(C)(C)C)=NC(N[C@@H]3CCNC3)=N2)=C(C)C=C1.CNC(=O)C1=CC(NC2=CC(N(C)CC(C)(C)C)=NC(N[C@@H]3CCN(C(=O)OC(C)(C)C)C3)=N2)=C(C)C=C1 CGSUKJLRGMEMPA-GPIYMFQDSA-N 0.000 description 1
- JSGFOGNTYVGPBT-UHFFFAOYSA-N CNC(=O)C1=CC(NC2=C([N+](=O)[O-])C(Cl)=NC(SC)=N2)=C(C)C=C1.CNC(=O)C1=CC=C(C)C(N)=C1.CSC1=NC(Cl)=C([N+](=O)[O-])C(Cl)=N1 Chemical compound CNC(=O)C1=CC(NC2=C([N+](=O)[O-])C(Cl)=NC(SC)=N2)=C(C)C=C1.CNC(=O)C1=CC=C(C)C(N)=C1.CSC1=NC(Cl)=C([N+](=O)[O-])C(Cl)=N1 JSGFOGNTYVGPBT-UHFFFAOYSA-N 0.000 description 1
- BBVGLSVJACFTGU-UHFFFAOYSA-N CNC(=O)C1=CC(NC2=C([N+](=O)[O-])C(N(C)CC(C)(C)C)=NC(NCCCN(C)C)=N2)=C(C)C=C1.CSC1=NC(O)=C([N+](=O)[O-])C(O)=N1.CSC1=NC(O)=CC(O)=N1.O=S(=O)(O)O.O=[N+]([O-])O Chemical compound CNC(=O)C1=CC(NC2=C([N+](=O)[O-])C(N(C)CC(C)(C)C)=NC(NCCCN(C)C)=N2)=C(C)C=C1.CSC1=NC(O)=C([N+](=O)[O-])C(O)=N1.CSC1=NC(O)=CC(O)=N1.O=S(=O)(O)O.O=[N+]([O-])O BBVGLSVJACFTGU-UHFFFAOYSA-N 0.000 description 1
- QUKKZRFTSLKTIP-UHFFFAOYSA-N CNC(=O)C1=CC(NC2=C([N+](=O)[O-])C(N(C)CC(C)(C)C)=NC(S(C)(=O)=O)=N2)=C(C)C=C1.CNC(=O)C1=CC(NC2=C([N+](=O)[O-])C(N(C)CC(C)(C)C)=NC(SC)=N2)=C(C)C=C1.O=[Mn](=O)(=O)(=O)[K] Chemical compound CNC(=O)C1=CC(NC2=C([N+](=O)[O-])C(N(C)CC(C)(C)C)=NC(S(C)(=O)=O)=N2)=C(C)C=C1.CNC(=O)C1=CC(NC2=C([N+](=O)[O-])C(N(C)CC(C)(C)C)=NC(SC)=N2)=C(C)C=C1.O=[Mn](=O)(=O)(=O)[K] QUKKZRFTSLKTIP-UHFFFAOYSA-N 0.000 description 1
- SZEUAJDHVDJADA-GPIYMFQDSA-N CNC(=O)C1=CC(NC2=NC(N(C)CC(C)(C)C)=CC(N[C@@H]3CCN(C(=O)OC(C)(C)C)C3)=N2)=C(C)C=C1.[C-]#[N+]C1=C(N(C)CC(C)(C)C)N=C(NC2=C(C)C=CC(C(=O)NC)=C2)N=C1N[C@@H]1CCNC1 Chemical compound CNC(=O)C1=CC(NC2=NC(N(C)CC(C)(C)C)=CC(N[C@@H]3CCN(C(=O)OC(C)(C)C)C3)=N2)=C(C)C=C1.[C-]#[N+]C1=C(N(C)CC(C)(C)C)N=C(NC2=C(C)C=CC(C(=O)NC)=C2)N=C1N[C@@H]1CCNC1 SZEUAJDHVDJADA-GPIYMFQDSA-N 0.000 description 1
- HDEKONWVOHQJMN-UHFFFAOYSA-L CNC(=O)C1=CC([N-]C2=C([N+](=O)[O-])C(Cl)=NC(SC)=N2)=C(C)C=C1.CNC(=O)C1=CC([N-]C2=C([N+](=O)[O-])C(N(C)CC(C)(C)C)=NC(SC)=N2)=C(C)C=C1.CNCC(C)(C)C.Cl Chemical compound CNC(=O)C1=CC([N-]C2=C([N+](=O)[O-])C(Cl)=NC(SC)=N2)=C(C)C=C1.CNC(=O)C1=CC([N-]C2=C([N+](=O)[O-])C(N(C)CC(C)(C)C)=NC(SC)=N2)=C(C)C=C1.CNCC(C)(C)C.Cl HDEKONWVOHQJMN-UHFFFAOYSA-L 0.000 description 1
- KFSGLBITFPARQH-UHFFFAOYSA-N CNC(=O)C1=CC=C(C)C(N)=C1.CNC(=O)C1=CC=C(C)C(NC2=C(Br)C=NC(Cl)=N2)=C1.ClC1=NC(Cl)=C(Br)C=N1 Chemical compound CNC(=O)C1=CC=C(C)C(N)=C1.CNC(=O)C1=CC=C(C)C(NC2=C(Br)C=NC(Cl)=N2)=C1.ClC1=NC(Cl)=C(Br)C=N1 KFSGLBITFPARQH-UHFFFAOYSA-N 0.000 description 1
- SOWTZJIGDOCIDK-UHFFFAOYSA-N CNC(=O)C1=CC=C(C)C(N)=C1.CNC(=O)C1=CC=C(C)C(NC2=C(C#N)C(Cl)=NC(SC)=N2)=C1.CSC1=NC(Cl)=C(C#N)C(Cl)=N1 Chemical compound CNC(=O)C1=CC=C(C)C(N)=C1.CNC(=O)C1=CC=C(C)C(NC2=C(C#N)C(Cl)=NC(SC)=N2)=C1.CSC1=NC(Cl)=C(C#N)C(Cl)=N1 SOWTZJIGDOCIDK-UHFFFAOYSA-N 0.000 description 1
- OJONIBKNUJNJSX-UHFFFAOYSA-N CNC(=O)C1=CC=C(C)C(N)=C1.CNC(=O)C1=CC=C(C)C(NC2=NC=C(C#N)C(Cl)=N2)=C1.CNC(=O)C1=CC=C(C)C(NC2=NC=C(C#N)C(N(C)CC(C)(C)C)=N2)=C1.CNCC(C)(C)C.O=C(Cl)C1=CN=C(Cl)N=C1Cl Chemical compound CNC(=O)C1=CC=C(C)C(N)=C1.CNC(=O)C1=CC=C(C)C(NC2=NC=C(C#N)C(Cl)=N2)=C1.CNC(=O)C1=CC=C(C)C(NC2=NC=C(C#N)C(N(C)CC(C)(C)C)=N2)=C1.CNCC(C)(C)C.O=C(Cl)C1=CN=C(Cl)N=C1Cl OJONIBKNUJNJSX-UHFFFAOYSA-N 0.000 description 1
- VCRADTMQOVBIMV-UHFFFAOYSA-N CNC(=O)C1=CC=C(C)C(N2CC(=O)NC3=C(N(C)CC(C)(C)C)N=CN=C32)=C1.CNC(=O)C1=CC=C(C)C(NC2=NC=NC(N(C)CC(C)(C)C)=C2N)=C1.ClCCl Chemical compound CNC(=O)C1=CC=C(C)C(N2CC(=O)NC3=C(N(C)CC(C)(C)C)N=CN=C32)=C1.CNC(=O)C1=CC=C(C)C(NC2=NC=NC(N(C)CC(C)(C)C)=C2N)=C1.ClCCl VCRADTMQOVBIMV-UHFFFAOYSA-N 0.000 description 1
- PYJRCHKALHIXCQ-UHFFFAOYSA-N CNC(=O)C1=CC=C(C)C(NC2=C(Br)C=NC(Cl)=N2)=C1.CNC(=O)C1=CC=C(C)C(NC2=C(Br)C=NC(N(C)CC(C)(C)C)=N2)=C1.CNCC(C)(C)C.Cl Chemical compound CNC(=O)C1=CC=C(C)C(NC2=C(Br)C=NC(Cl)=N2)=C1.CNC(=O)C1=CC=C(C)C(NC2=C(Br)C=NC(N(C)CC(C)(C)C)=N2)=C1.CNCC(C)(C)C.Cl PYJRCHKALHIXCQ-UHFFFAOYSA-N 0.000 description 1
- JDGSQZOQRSTNAH-UHFFFAOYSA-N CNC(=O)C1=CC=C(C)C(NC2=C(Br)C=NC(N(C)CC(C)(C)C)=N2)=C1 Chemical compound CNC(=O)C1=CC=C(C)C(NC2=C(Br)C=NC(N(C)CC(C)(C)C)=N2)=C1 JDGSQZOQRSTNAH-UHFFFAOYSA-N 0.000 description 1
- WMRKYFIVWHGWKL-UHFFFAOYSA-N CNC(=O)C1=CC=C(C)C(NC2=C(Br)C=NC(N(C)CC(C)(C)C)=N2)=C1.CNC(=O)C1=CC=C(C)C(NC2=C(C#N)C=NC(N(C)CC(C)(C)C)=N2)=C1 Chemical compound CNC(=O)C1=CC=C(C)C(NC2=C(Br)C=NC(N(C)CC(C)(C)C)=N2)=C1.CNC(=O)C1=CC=C(C)C(NC2=C(C#N)C=NC(N(C)CC(C)(C)C)=N2)=C1 WMRKYFIVWHGWKL-UHFFFAOYSA-N 0.000 description 1
- GJAXPOJVDDLBNF-UHFFFAOYSA-N CNC(=O)C1=CC=C(C)C(NC2=C(C#N)C(Cl)=NC(SC)=N2)=C1.CNC(=O)C1=CC=C(C)C(NC2=C(C#N)C(N(C)CC(C)(C)C)=NC(SC)=N2)=C1.CNCC(C)(C)C Chemical compound CNC(=O)C1=CC=C(C)C(NC2=C(C#N)C(Cl)=NC(SC)=N2)=C1.CNC(=O)C1=CC=C(C)C(NC2=C(C#N)C(N(C)CC(C)(C)C)=NC(SC)=N2)=C1.CNCC(C)(C)C GJAXPOJVDDLBNF-UHFFFAOYSA-N 0.000 description 1
- CURKDSSYRGRFLO-UHFFFAOYSA-N CNC(=O)C1=CC=C(C)C(NC2=C(C#N)C(NC3CCNC3)=NC(N(C)CC(C)(C)C)=N2)=C1.CNC(=O)C1=CC=C(C)C(NC2=CC(NC3CCN(C(=O)OC(C)(C)C)C3)=NC(N(C)CC(C)(C)C)=N2)=C1 Chemical compound CNC(=O)C1=CC=C(C)C(NC2=C(C#N)C(NC3CCNC3)=NC(N(C)CC(C)(C)C)=N2)=C1.CNC(=O)C1=CC=C(C)C(NC2=CC(NC3CCN(C(=O)OC(C)(C)C)C3)=NC(N(C)CC(C)(C)C)=N2)=C1 CURKDSSYRGRFLO-UHFFFAOYSA-N 0.000 description 1
- BHVGIKKOGZBROJ-UHFFFAOYSA-N CNC(=O)C1=CC=C(C)C(NC2=CC(Cl)=NC=N2)=C1.CNC(=O)C1=CC=C(C)C(NC2=CC(N(C)CC(C)(C)C)=NC=N2)=C1.CNCC(C)(C)C Chemical compound CNC(=O)C1=CC=C(C)C(NC2=CC(Cl)=NC=N2)=C1.CNC(=O)C1=CC=C(C)C(NC2=CC(N(C)CC(C)(C)C)=NC=N2)=C1.CNCC(C)(C)C BHVGIKKOGZBROJ-UHFFFAOYSA-N 0.000 description 1
- LJSSZYVYZRMROT-JQXSOXDTSA-N CNC(=O)C1=CC=C(C)C(NC2=N(C#N)C(N[C@@H]3CCNC3)=NC(N(C)CC(C)(C)C)=N2)=C1.CNC(=O)C1=CC=C(C)C(NC2=NC(N[C@@H]3CCNC3)=C(C#N)C(N(C)CC(C)(C)C)=N2)=C1.CONC(=O)C1=CC=C(C)C(NC2=C(F)C=C(C#N)C(NCC(C)(C)C)=N2)=C1.[C-]#[N+]C1=C(N(C)CC(C)(C)C)N=C(N[C@@H]2CCNC2)N=C1NC1=CC(C(=O)NC)=CC=C1C.[C-]#[N+]C1=C(NC2=CC(C(=O)NOCC)=CC=C2C)N=C(N(C)CC(C)(C)C)N=C1N1CCCN(C)CC1 Chemical compound CNC(=O)C1=CC=C(C)C(NC2=N(C#N)C(N[C@@H]3CCNC3)=NC(N(C)CC(C)(C)C)=N2)=C1.CNC(=O)C1=CC=C(C)C(NC2=NC(N[C@@H]3CCNC3)=C(C#N)C(N(C)CC(C)(C)C)=N2)=C1.CONC(=O)C1=CC=C(C)C(NC2=C(F)C=C(C#N)C(NCC(C)(C)C)=N2)=C1.[C-]#[N+]C1=C(N(C)CC(C)(C)C)N=C(N[C@@H]2CCNC2)N=C1NC1=CC(C(=O)NC)=CC=C1C.[C-]#[N+]C1=C(NC2=CC(C(=O)NOCC)=CC=C2C)N=C(N(C)CC(C)(C)C)N=C1N1CCCN(C)CC1 LJSSZYVYZRMROT-JQXSOXDTSA-N 0.000 description 1
- WBRQCJYMLLJJMX-UHFFFAOYSA-N CNC(=O)C1=CC=C(C)C(NC2=NC(N(C)CC(C)(C)C)=NC(N(C)CC(C)(C)C)=C2O)=C1 Chemical compound CNC(=O)C1=CC=C(C)C(NC2=NC(N(C)CC(C)(C)C)=NC(N(C)CC(C)(C)C)=C2O)=C1 WBRQCJYMLLJJMX-UHFFFAOYSA-N 0.000 description 1
- GRDPJEOEYRSNQV-YPATWDRESA-N CNC(=O)C1=CC=C(C)C(NC2=NC(N(C)CC(C)(C)C)=NC(N[C@@H]3CCNC3)=C2C#N)=C1.CNC(=O)C1=CC=C(C)C(NC2=NC(NC3CCCC3)=C(C#N)C(N(C)CC(C)(C)C)=N2)=C1.CNC(=O)C1=CC=C(C)C(NC2=NC(NC3CCCC3)=NC(N(C)CC(C)(C)C)=C2C#N)=C1.CNC(=O)C1=CC=C(C)C(NC2=NC=NC(NCC(C)(C)C)=C2[N+](=O)[O-])=C1 Chemical compound CNC(=O)C1=CC=C(C)C(NC2=NC(N(C)CC(C)(C)C)=NC(N[C@@H]3CCNC3)=C2C#N)=C1.CNC(=O)C1=CC=C(C)C(NC2=NC(NC3CCCC3)=C(C#N)C(N(C)CC(C)(C)C)=N2)=C1.CNC(=O)C1=CC=C(C)C(NC2=NC(NC3CCCC3)=NC(N(C)CC(C)(C)C)=C2C#N)=C1.CNC(=O)C1=CC=C(C)C(NC2=NC=NC(NCC(C)(C)C)=C2[N+](=O)[O-])=C1 GRDPJEOEYRSNQV-YPATWDRESA-N 0.000 description 1
- SOJDFSYXVUCBPO-UHFFFAOYSA-N CNC(=O)C1=CC=C(C)C(NC2=NC(N(C)CC(C)(C)C)=NC=C2C#N)=C1 Chemical compound CNC(=O)C1=CC=C(C)C(NC2=NC(N(C)CC(C)(C)C)=NC=C2C#N)=C1 SOJDFSYXVUCBPO-UHFFFAOYSA-N 0.000 description 1
- TYZWLSSLSOBHRZ-UHFFFAOYSA-N CNC(=O)C1=CC=C(C)C(NC2=NC(N(C)CC(C)(C)C)=NC=C2C#N)=C1.CNC(=O)C1=CC=C(C)C(NC2=NC=CC(N(C)CC(C)(C)C)=C2[N+](=O)[O-])=C1.CONC(=O)C1=CC=C(C)C(NC2=NC=CC(N(C)CC(C)(C)C)=C2[N+](=O)[O-])=C1.CONC(=O)C1=CC=C(C)C(NC2=NC=NC(NCC(C)(C)C)=C2[N+](=O)[O-])=C1 Chemical compound CNC(=O)C1=CC=C(C)C(NC2=NC(N(C)CC(C)(C)C)=NC=C2C#N)=C1.CNC(=O)C1=CC=C(C)C(NC2=NC=CC(N(C)CC(C)(C)C)=C2[N+](=O)[O-])=C1.CONC(=O)C1=CC=C(C)C(NC2=NC=CC(N(C)CC(C)(C)C)=C2[N+](=O)[O-])=C1.CONC(=O)C1=CC=C(C)C(NC2=NC=NC(NCC(C)(C)C)=C2[N+](=O)[O-])=C1 TYZWLSSLSOBHRZ-UHFFFAOYSA-N 0.000 description 1
- WJDCEKPHOKHTEV-UHFFFAOYSA-N CNC(=O)C1=CC=C(C)C(NC2=NC(NC)=NC(N(C)CC(C)(C)C)=C2)=C1.CNC(=O)C1=CC=C(C)C(NC2=NC(NC)=NC(N(C)CC(C)(C)C)=C2Br)=C1.ClCCl Chemical compound CNC(=O)C1=CC=C(C)C(NC2=NC(NC)=NC(N(C)CC(C)(C)C)=C2)=C1.CNC(=O)C1=CC=C(C)C(NC2=NC(NC)=NC(N(C)CC(C)(C)C)=C2Br)=C1.ClCCl WJDCEKPHOKHTEV-UHFFFAOYSA-N 0.000 description 1
- SIKSDTIFIMMSQX-UHFFFAOYSA-N CNC(=O)C1=CC=C(C)C(NC2=NC(NC)=NC(N(C)CC(C)(C)C)=C2O)=C1 Chemical compound CNC(=O)C1=CC=C(C)C(NC2=NC(NC)=NC(N(C)CC(C)(C)C)=C2O)=C1 SIKSDTIFIMMSQX-UHFFFAOYSA-N 0.000 description 1
- TVTNNWSPNWYAGS-UHFFFAOYSA-N CNC(=O)C1=CC=C(C)C(NC2=NC(NC)=NC(NC)=C2Br)=C1 Chemical compound CNC(=O)C1=CC=C(C)C(NC2=NC(NC)=NC(NC)=C2Br)=C1 TVTNNWSPNWYAGS-UHFFFAOYSA-N 0.000 description 1
- ICQWECLDHQTLQY-UHFFFAOYSA-N CNC(=O)C1=CC=C(C)C(NC2=NC(NC)=NC(NCC(C)(C)C)=C2)=C1.CNC(=O)C1=CC=C(C)C(NC2=NC(NC)=NC(NCC(C)(C)C)=C2Br)=C1.ClCCl Chemical compound CNC(=O)C1=CC=C(C)C(NC2=NC(NC)=NC(NCC(C)(C)C)=C2)=C1.CNC(=O)C1=CC=C(C)C(NC2=NC(NC)=NC(NCC(C)(C)C)=C2Br)=C1.ClCCl ICQWECLDHQTLQY-UHFFFAOYSA-N 0.000 description 1
- AFXNPYWVBHSJDK-UHFFFAOYSA-N CNC(=O)C1=CC=C(C)C(NC2=NC(NC3CCS(=O)(=O)C3)=NC(N(C)CC(C)(C)C)=C2C#N)=C1 Chemical compound CNC(=O)C1=CC=C(C)C(NC2=NC(NC3CCS(=O)(=O)C3)=NC(N(C)CC(C)(C)C)=C2C#N)=C1 AFXNPYWVBHSJDK-UHFFFAOYSA-N 0.000 description 1
- PVMYZPKFHYKEBO-UHFFFAOYSA-N CNC(=O)C1=CC=C(C)C(NC2=NC(NCC(C)(C)CN(C)C)=NC(N(C)CC(C)(C)C)=C2C#N)=C1 Chemical compound CNC(=O)C1=CC=C(C)C(NC2=NC(NCC(C)(C)CN(C)C)=NC(N(C)CC(C)(C)C)=C2C#N)=C1 PVMYZPKFHYKEBO-UHFFFAOYSA-N 0.000 description 1
- VCAUFUTYWUVMRH-UHFFFAOYSA-N CNC(=O)C1=CC=C(C)C(NC2=NC(NCCC3CCCN3C)=NC(N(C)CC(C)(C)C)=C2C#N)=C1 Chemical compound CNC(=O)C1=CC=C(C)C(NC2=NC(NCCC3CCCN3C)=NC(N(C)CC(C)(C)C)=C2C#N)=C1 VCAUFUTYWUVMRH-UHFFFAOYSA-N 0.000 description 1
- XQQZEFAPYYTQLG-UHFFFAOYSA-N CNC(=O)C1=CC=C(C)C(NC2=NC(NCCCN(C)C)=NC(N(C)CC(C)(C)C)=C2C#N)=C1 Chemical compound CNC(=O)C1=CC=C(C)C(NC2=NC(NCCCN(C)C)=NC(N(C)CC(C)(C)C)=C2C#N)=C1 XQQZEFAPYYTQLG-UHFFFAOYSA-N 0.000 description 1
- XWMDTAZYXHPAKT-GFCCVEGCSA-N CNC(=O)C1=CC=C(C)C(NC2=NC(N[C@@H]3CCNC3)=NC=C2Br)=C1 Chemical compound CNC(=O)C1=CC=C(C)C(NC2=NC(N[C@@H]3CCNC3)=NC=C2Br)=C1 XWMDTAZYXHPAKT-GFCCVEGCSA-N 0.000 description 1
- WGOIUENFMBCIHH-UHFFFAOYSA-N CNC(=O)C1=CC=C(C)C(NC2=NC(O)=NC(N(C)CC(C)(C)C)=C2C#N)=C1 Chemical compound CNC(=O)C1=CC=C(C)C(NC2=NC(O)=NC(N(C)CC(C)(C)C)=C2C#N)=C1 WGOIUENFMBCIHH-UHFFFAOYSA-N 0.000 description 1
- ZHCXLVBVSHEAFZ-UHFFFAOYSA-N CNC(=O)C1=CC=C(C)C(NC2=NC(OC3CCN(C)CC3)=NC(N(C)C(C)C)=C2C#N)=C1 Chemical compound CNC(=O)C1=CC=C(C)C(NC2=NC(OC3CCN(C)CC3)=NC(N(C)C(C)C)=C2C#N)=C1 ZHCXLVBVSHEAFZ-UHFFFAOYSA-N 0.000 description 1
- KJJYYXGGYOPHJW-UHFFFAOYSA-N CNC(=O)C1=CC=C(C)C(NC2=NC(OC3CCN(C)CC3)=NC(N(C)C)=C2C#N)=C1 Chemical compound CNC(=O)C1=CC=C(C)C(NC2=NC(OC3CCN(C)CC3)=NC(N(C)C)=C2C#N)=C1 KJJYYXGGYOPHJW-UHFFFAOYSA-N 0.000 description 1
- MBTPWXKACUQHGU-UHFFFAOYSA-N CNC(=O)C1=CC=C(C)C(NC2=NC(OC3CCN(C)CC3)=NC(N(C)CC(C)(C)C)=C2C#N)=C1 Chemical compound CNC(=O)C1=CC=C(C)C(NC2=NC(OC3CCN(C)CC3)=NC(N(C)CC(C)(C)C)=C2C#N)=C1 MBTPWXKACUQHGU-UHFFFAOYSA-N 0.000 description 1
- HJENDDIDEFKETJ-UHFFFAOYSA-N CNC(=O)C1=CC=C(C)C(NC2=NC(OC3CCN(C)CC3)=NC(N(C)CC(C)C)=C2C#N)=C1 Chemical compound CNC(=O)C1=CC=C(C)C(NC2=NC(OC3CCN(C)CC3)=NC(N(C)CC(C)C)=C2C#N)=C1 HJENDDIDEFKETJ-UHFFFAOYSA-N 0.000 description 1
- CUMBKTMWWXQYMV-UHFFFAOYSA-N CNC(=O)C1=CC=C(C)C(NC2=NC(OC3CCN(C)CC3)=NC=C2Br)=C1 Chemical compound CNC(=O)C1=CC=C(C)C(NC2=NC(OC3CCN(C)CC3)=NC=C2Br)=C1 CUMBKTMWWXQYMV-UHFFFAOYSA-N 0.000 description 1
- UJTSGHZCFBCBNV-UHFFFAOYSA-N CNC(=O)C1=CC=C(C)C(NC2=NC(OCC(C)(C)CN(C)C)=NC(N(C)CC(C)(C)C)=C2C#N)=C1 Chemical compound CNC(=O)C1=CC=C(C)C(NC2=NC(OCC(C)(C)CN(C)C)=NC(N(C)CC(C)(C)C)=C2C#N)=C1 UJTSGHZCFBCBNV-UHFFFAOYSA-N 0.000 description 1
- PELLMBVSLWSKPX-UHFFFAOYSA-N CNC(=O)C1=CC=C(C)C(NC2=NC(OCC3CCCN(C)C3)=NC(N(C)CC(C)(C)C)=C2C#N)=C1 Chemical compound CNC(=O)C1=CC=C(C)C(NC2=NC(OCC3CCCN(C)C3)=NC(N(C)CC(C)(C)C)=C2C#N)=C1 PELLMBVSLWSKPX-UHFFFAOYSA-N 0.000 description 1
- XWIJXKSMVPFZJL-UHFFFAOYSA-N CNC(=O)C1=CC=C(C)C(NC2=NC(OCCCN(C)C)=NC(N(C)CC(C)(C)C)=C2C#N)=C1 Chemical compound CNC(=O)C1=CC=C(C)C(NC2=NC(OCCCN(C)C)=NC(N(C)CC(C)(C)C)=C2C#N)=C1 XWIJXKSMVPFZJL-UHFFFAOYSA-N 0.000 description 1
- BDEXPDWIVKOODT-UHFFFAOYSA-N CNC(=O)C1=CC=C(C)C(NC2=NC(SC)=NC(N(C)CC(C)(C)C)=C2)=C1.CNC(=O)C1=CC=C(C)C(NC2=NC(SC)=NC(N(C)CC(C)(C)C)=C2Br)=C1.ClCCl Chemical compound CNC(=O)C1=CC=C(C)C(NC2=NC(SC)=NC(N(C)CC(C)(C)C)=C2)=C1.CNC(=O)C1=CC=C(C)C(NC2=NC(SC)=NC(N(C)CC(C)(C)C)=C2Br)=C1.ClCCl BDEXPDWIVKOODT-UHFFFAOYSA-N 0.000 description 1
- BWEFYFUQJFZPOB-UHFFFAOYSA-N CNC(=O)C1=CC=C(C)C(NC2=NC(SC)=NC(NCC(C)(C)C)=C2C#N)=C1 Chemical compound CNC(=O)C1=CC=C(C)C(NC2=NC(SC)=NC(NCC(C)(C)C)=C2C#N)=C1 BWEFYFUQJFZPOB-UHFFFAOYSA-N 0.000 description 1
- DVIBIAHNHSNSEA-UHFFFAOYSA-N CNC(=O)C1=CC=C(C)C(NC2=NC=CC(N(C)CC(C)(C)C)=C2C#N)=C1.CNC(=O)C1=CC=C(C)C(NC2=NC=NC(NCC(C)(C)C)=C2Br)=C1.CNC(=O)C1=CC=C(C)C(NC2=NC=NC(NCC(C)(C)C)=C2C#N)=C1 Chemical compound CNC(=O)C1=CC=C(C)C(NC2=NC=CC(N(C)CC(C)(C)C)=C2C#N)=C1.CNC(=O)C1=CC=C(C)C(NC2=NC=NC(NCC(C)(C)C)=C2Br)=C1.CNC(=O)C1=CC=C(C)C(NC2=NC=NC(NCC(C)(C)C)=C2C#N)=C1 DVIBIAHNHSNSEA-UHFFFAOYSA-N 0.000 description 1
- SIEPZFXZAXOACO-UHFFFAOYSA-N CNC(=O)C1=CC=C(C)C(NC2=NC=NC(N(C)CC(C)(C)C)=C2)=C1.CNC(=O)C1=CC=C(C)C(NC2=NC=NC(N(C)CC(C)(C)C)=C2Br)=C1.ClCCl Chemical compound CNC(=O)C1=CC=C(C)C(NC2=NC=NC(N(C)CC(C)(C)C)=C2)=C1.CNC(=O)C1=CC=C(C)C(NC2=NC=NC(N(C)CC(C)(C)C)=C2Br)=C1.ClCCl SIEPZFXZAXOACO-UHFFFAOYSA-N 0.000 description 1
- BXPXCILOFPPTAH-UHFFFAOYSA-N CNC(=O)C1=CC=C(C)C(NC2=NC=NC(N(C)CC(C)(C)C)=C2C#N)=C1 Chemical compound CNC(=O)C1=CC=C(C)C(NC2=NC=NC(N(C)CC(C)(C)C)=C2C#N)=C1 BXPXCILOFPPTAH-UHFFFAOYSA-N 0.000 description 1
- KNSSBPRAEPTHSW-UHFFFAOYSA-N CNC(=O)C1=CC=C(C)C(NC2=NC=NC(N(C)CC(C)(C)C)=C2N)=C1.CNC(=O)C1=CC=C(C)C(NC2=NC=NC(N(C)CC(C)(C)C)=C2[N+](=O)[O-])=C1 Chemical compound CNC(=O)C1=CC=C(C)C(NC2=NC=NC(N(C)CC(C)(C)C)=C2N)=C1.CNC(=O)C1=CC=C(C)C(NC2=NC=NC(N(C)CC(C)(C)C)=C2[N+](=O)[O-])=C1 KNSSBPRAEPTHSW-UHFFFAOYSA-N 0.000 description 1
- YSNLGIQTWKSBCY-UHFFFAOYSA-N CNC(=O)C1=CC=C(C)C(NC2=NC=NC(NCC(C)(C)C)=C2)=C1.CNC(=O)C1=CC=C(C)C(NC2=NC=NC(NCC(C)(C)C)=C2Br)=C1.ClCCl Chemical compound CNC(=O)C1=CC=C(C)C(NC2=NC=NC(NCC(C)(C)C)=C2)=C1.CNC(=O)C1=CC=C(C)C(NC2=NC=NC(NCC(C)(C)C)=C2Br)=C1.ClCCl YSNLGIQTWKSBCY-UHFFFAOYSA-N 0.000 description 1
- CYWSCZNOFKSGET-UHFFFAOYSA-N CNC(=O)C1=CC=C(C)C(NC2=NC=NC(NCC(C)(C)C)=C2C#N)=C1 Chemical compound CNC(=O)C1=CC=C(C)C(NC2=NC=NC(NCC(C)(C)C)=C2C#N)=C1 CYWSCZNOFKSGET-UHFFFAOYSA-N 0.000 description 1
- AOXDYZRBOFGXEM-UHFFFAOYSA-N CNC(=O)C1=CC=C(C)C(NC2=NC=NC(NCC(C)(C)C)=C2[N+](=O)[O-])=C1 Chemical compound CNC(=O)C1=CC=C(C)C(NC2=NC=NC(NCC(C)(C)C)=C2[N+](=O)[O-])=C1 AOXDYZRBOFGXEM-UHFFFAOYSA-N 0.000 description 1
- ZVJSFCGRZSGGDS-UHFFFAOYSA-N CNC1=NC(NC2=CC(C(=O)NOC)=CC=C2C)=C(C#N)C(N(C)CC(C)(C)C)=N1.CONC(=O)C1=CC=C(C)C(NC2=NC(NCCC3CCCN3C)=NC(N(C)CC(C)(C)C)=C2C#N)=C1.CONC(=O)C1=CC=C(C)C(NC2=NC(OCC3CCCN(C)C3)=NC(N(C)CC(C)(C)C)=C2C#N)=C1.CONC(=O)C1=CC=C(C)C(NC2=NC(OCCC3CCCN3C)=NC(N(C)CC(C)(C)C)=C2C#N)=C1 Chemical compound CNC1=NC(NC2=CC(C(=O)NOC)=CC=C2C)=C(C#N)C(N(C)CC(C)(C)C)=N1.CONC(=O)C1=CC=C(C)C(NC2=NC(NCCC3CCCN3C)=NC(N(C)CC(C)(C)C)=C2C#N)=C1.CONC(=O)C1=CC=C(C)C(NC2=NC(OCC3CCCN(C)C3)=NC(N(C)CC(C)(C)C)=C2C#N)=C1.CONC(=O)C1=CC=C(C)C(NC2=NC(OCCC3CCCN3C)=NC(N(C)CC(C)(C)C)=C2C#N)=C1 ZVJSFCGRZSGGDS-UHFFFAOYSA-N 0.000 description 1
- FKCZMIRXWUJXQB-UHFFFAOYSA-N CNC1=NC(NC2=CC(C(N)=O)=CC=C2C)=C(C#N)C(N(C)CC(C)(C)C)=N1 Chemical compound CNC1=NC(NC2=CC(C(N)=O)=CC=C2C)=C(C#N)C(N(C)CC(C)(C)C)=N1 FKCZMIRXWUJXQB-UHFFFAOYSA-N 0.000 description 1
- UZDPIJOCXCMWDT-UHFFFAOYSA-M CNC1=NC(NC2=CC(C(N)=O)=CC=C2C)=C(C#N)C(N(C)CC(C)(C)C)=N1.CNC1=NC(NC2=CC(C3=NN=CN3)=CC=C2C)=C(C#N)C(N(C)CC(C)(C)C)=N1.CONC(=O)C1=CC=C(C)C(NC2=NC(NC3CCCC3)=NC(N(C)CC(C)(C)C)=C2C#N)=C1.C[N-]C(=O)C1=CC=C(C)C(NC2=NC(OC3CCN(C)CC3)=NC(N(C)CC(C)(C)C)=C2C#N)=C1 Chemical compound CNC1=NC(NC2=CC(C(N)=O)=CC=C2C)=C(C#N)C(N(C)CC(C)(C)C)=N1.CNC1=NC(NC2=CC(C3=NN=CN3)=CC=C2C)=C(C#N)C(N(C)CC(C)(C)C)=N1.CONC(=O)C1=CC=C(C)C(NC2=NC(NC3CCCC3)=NC(N(C)CC(C)(C)C)=C2C#N)=C1.C[N-]C(=O)C1=CC=C(C)C(NC2=NC(OC3CCN(C)CC3)=NC(N(C)CC(C)(C)C)=C2C#N)=C1 UZDPIJOCXCMWDT-UHFFFAOYSA-M 0.000 description 1
- FOTAFYVRYLXFSP-UHFFFAOYSA-N CNC1=NC(NC2=CC(C3=NN=CN3)=CC=C2C)=C(C#N)C(N(C)CC(C)(C)C)=N1 Chemical compound CNC1=NC(NC2=CC(C3=NN=CN3)=CC=C2C)=C(C#N)C(N(C)CC(C)(C)C)=N1 FOTAFYVRYLXFSP-UHFFFAOYSA-N 0.000 description 1
- CWHXDJUIMQJEBQ-UHFFFAOYSA-N CNCC(C)(C)C.CONC(=O)C1=CC=C(C)C(NC2=C(C#N)C(Cl)=NC(SC)=N2)=C1.CONC(=O)C1=CC=C(C)C(NC2=C(C#N)C(N(C)CC(C)(C)C)=NC(SC)=N2)=C1 Chemical compound CNCC(C)(C)C.CONC(=O)C1=CC=C(C)C(NC2=C(C#N)C(Cl)=NC(SC)=N2)=C1.CONC(=O)C1=CC=C(C)C(NC2=C(C#N)C(N(C)CC(C)(C)C)=NC(SC)=N2)=C1 CWHXDJUIMQJEBQ-UHFFFAOYSA-N 0.000 description 1
- POBYZTNFDOWQDU-UHFFFAOYSA-N CNCC(C)(C)C.CSC1=NC(NC2=C(C)C=CC(C3=NN=CN3)=C2)=C(C#N)C(Cl)=N1.CSC1=NC(NC2=C(C)C=CC(C3=NN=CN3)=C2)=C(C#N)C(N(C)CC(C)(C)C)=N1.Cl Chemical compound CNCC(C)(C)C.CSC1=NC(NC2=C(C)C=CC(C3=NN=CN3)=C2)=C(C#N)C(Cl)=N1.CSC1=NC(NC2=C(C)C=CC(C3=NN=CN3)=C2)=C(C#N)C(N(C)CC(C)(C)C)=N1.Cl POBYZTNFDOWQDU-UHFFFAOYSA-N 0.000 description 1
- AHUQNWPEPWHNMX-UHFFFAOYSA-N CNCC(C)(C)C.Cl.[C-]#[N+]C1=C(Cl)N=C(NC2=C(C)C=CC(C(=O)NOC)=C2)C(F)=C1.[C-]#[N+]C1=C(N(C)CC(C)(C)C)N=C(NC2=C(C)C=CC(C(=O)NOC)=C2)C(F)=C1 Chemical compound CNCC(C)(C)C.Cl.[C-]#[N+]C1=C(Cl)N=C(NC2=C(C)C=CC(C(=O)NOC)=C2)C(F)=C1.[C-]#[N+]C1=C(N(C)CC(C)(C)C)N=C(NC2=C(C)C=CC(C(=O)NOC)=C2)C(F)=C1 AHUQNWPEPWHNMX-UHFFFAOYSA-N 0.000 description 1
- BZFFGHKLSHRXLP-UHFFFAOYSA-N CONC(=O)C1=CC(NC2=NC(N(C)CC(C)(C)C)=C(C#N)C(N3CCCN(C)CC3)=N2)=C(C)C=C1.CONC(=O)C1=CC(NC2=NC(N(C)CC(C)(C)C)=CC(N3CCCN(C)CC3)=N2)=C(C)C=C1 Chemical compound CONC(=O)C1=CC(NC2=NC(N(C)CC(C)(C)C)=C(C#N)C(N3CCCN(C)CC3)=N2)=C(C)C=C1.CONC(=O)C1=CC(NC2=NC(N(C)CC(C)(C)C)=CC(N3CCCN(C)CC3)=N2)=C(C)C=C1 BZFFGHKLSHRXLP-UHFFFAOYSA-N 0.000 description 1
- NDOUYGHZQPFYSW-UHFFFAOYSA-N CONC(=O)C1=CC=C(C)C(N)=C1.CONC(=O)C1=CC=C(C)C(NC2=C(C#N)C(Cl)=NC(SC)=N2)=C1.CSC1=NC(Cl)=C(C#N)C(Cl)=N1 Chemical compound CONC(=O)C1=CC=C(C)C(N)=C1.CONC(=O)C1=CC=C(C)C(NC2=C(C#N)C(Cl)=NC(SC)=N2)=C1.CSC1=NC(Cl)=C(C#N)C(Cl)=N1 NDOUYGHZQPFYSW-UHFFFAOYSA-N 0.000 description 1
- OWBOYOWXFZKTOI-UHFFFAOYSA-M CONC(=O)C1=CC=C(C)C(N)=C1.F[K].[C-]#[N+]C1=C(Cl)N=C(N(C)CC(C)(C)C)C(F)=C1.[C-]#[N+]C1=C(NC2=CC(C(=O)NOC)=CC=C2C)N=C(N(C)CC(C)(C)C)C(F)=C1 Chemical compound CONC(=O)C1=CC=C(C)C(N)=C1.F[K].[C-]#[N+]C1=C(Cl)N=C(N(C)CC(C)(C)C)C(F)=C1.[C-]#[N+]C1=C(NC2=CC(C(=O)NOC)=CC=C2C)N=C(N(C)CC(C)(C)C)C(F)=C1 OWBOYOWXFZKTOI-UHFFFAOYSA-M 0.000 description 1
- MYKMRUPACBUTMG-UHFFFAOYSA-M CONC(=O)C1=CC=C(C)C(N)=C1.F[K].[C-]#[N+]C1=C(Cl)N=C(NCC(C)(C)C)C(F)=C1.[C-]#[N+]C1=C(NC2=CC(C(=O)NOC)=CC=C2C)N=C(NCC(C)(C)C)C(F)=C1 Chemical compound CONC(=O)C1=CC=C(C)C(N)=C1.F[K].[C-]#[N+]C1=C(Cl)N=C(NCC(C)(C)C)C(F)=C1.[C-]#[N+]C1=C(NC2=CC(C(=O)NOC)=CC=C2C)N=C(NCC(C)(C)C)C(F)=C1 MYKMRUPACBUTMG-UHFFFAOYSA-M 0.000 description 1
- SZKCMVYOUUCENX-UHFFFAOYSA-N CONC(=O)C1=CC=C(C)C(NC2=C(C#N)C(N(C)CC(C)(C)C)=NC(S(C)=O)=N2)=C1.CONC(=O)C1=CC=C(C)C(NC2=C(C#N)C(N(C)CC(C)(C)C)=NC(SC)=N2)=C1.O=I(=O)(=O)O[Na] Chemical compound CONC(=O)C1=CC=C(C)C(NC2=C(C#N)C(N(C)CC(C)(C)C)=NC(S(C)=O)=N2)=C1.CONC(=O)C1=CC=C(C)C(NC2=C(C#N)C(N(C)CC(C)(C)C)=NC(SC)=N2)=C1.O=I(=O)(=O)O[Na] SZKCMVYOUUCENX-UHFFFAOYSA-N 0.000 description 1
- NBXNRIBSKWIXSF-UHFFFAOYSA-N CONC(=O)C1=CC=C(C)C(NC2=NC(OC3CCN(C)CC3)=NC(N(C)CC(C)(C)C)=C2C#N)=C1 Chemical compound CONC(=O)C1=CC=C(C)C(NC2=NC(OC3CCN(C)CC3)=NC(N(C)CC(C)(C)C)=C2C#N)=C1 NBXNRIBSKWIXSF-UHFFFAOYSA-N 0.000 description 1
- HMWPDXANMYEDJF-UHFFFAOYSA-N CONC(=O)C1=CC=C(C)C(NC2=NC(OCC3CCCN(C)C3)=NC(N(C)CC(C)(C)C)=C2C#N)=C1 Chemical compound CONC(=O)C1=CC=C(C)C(NC2=NC(OCC3CCCN(C)C3)=NC(N(C)CC(C)(C)C)=C2C#N)=C1 HMWPDXANMYEDJF-UHFFFAOYSA-N 0.000 description 1
- ZATWWDWDCNXSEX-UHFFFAOYSA-N CONC(=O)C1=CC=C(C)C(NC2=NC=NC(N(C)CC(C)(C)C)=C2[N+](=O)[O-])=C1 Chemical compound CONC(=O)C1=CC=C(C)C(NC2=NC=NC(N(C)CC(C)(C)C)=C2[N+](=O)[O-])=C1 ZATWWDWDCNXSEX-UHFFFAOYSA-N 0.000 description 1
- QUFIFJMTUCAOLA-UHFFFAOYSA-N CONC(=O)C1=CC=C(C)C(NC2=NC=NC(NCC(C)(C)C)=C2[N+](=O)[O-])=C1 Chemical compound CONC(=O)C1=CC=C(C)C(NC2=NC=NC(NCC(C)(C)C)=C2[N+](=O)[O-])=C1 QUFIFJMTUCAOLA-UHFFFAOYSA-N 0.000 description 1
- ADWSSSYDGOCHTR-VFXUXTATSA-N CSC1=NC(Cl)=C(/C=N/O)C(Cl)=N1.CSC1=NC(Cl)=C(C#N)C(Cl)=N1.O=S(Cl)Cl Chemical compound CSC1=NC(Cl)=C(/C=N/O)C(Cl)=N1.CSC1=NC(Cl)=C(C#N)C(Cl)=N1.O=S(Cl)Cl ADWSSSYDGOCHTR-VFXUXTATSA-N 0.000 description 1
- IJXDLGNRFWMQLL-UHFFFAOYSA-N CSC1=NC(Cl)=C(C=O)C(Cl)=N1.CSC1=NC(O)=CC(O)=N1.O=P(Cl)(Cl)Cl Chemical compound CSC1=NC(Cl)=C(C=O)C(Cl)=N1.CSC1=NC(O)=CC(O)=N1.O=P(Cl)(Cl)Cl IJXDLGNRFWMQLL-UHFFFAOYSA-N 0.000 description 1
- GYOMOEYBLDYTIT-UHFFFAOYSA-N CSC1=NC(Cl)=C([N+](=O)[O-])C(Cl)=N1.CSC1=NC(O)=C([N+](=O)[O-])C(O)=N1.O=P(Cl)(Cl)Cl Chemical compound CSC1=NC(Cl)=C([N+](=O)[O-])C(Cl)=N1.CSC1=NC(O)=C([N+](=O)[O-])C(O)=N1.O=P(Cl)(Cl)Cl GYOMOEYBLDYTIT-UHFFFAOYSA-N 0.000 description 1
- 101100026251 Caenorhabditis elegans atf-2 gene Proteins 0.000 description 1
- OYPRJOBELJOOCE-UHFFFAOYSA-N Calcium Chemical compound [Ca] OYPRJOBELJOOCE-UHFFFAOYSA-N 0.000 description 1
- 241000282465 Canis Species 0.000 description 1
- 206010007572 Cardiac hypertrophy Diseases 0.000 description 1
- 208000006029 Cardiomegaly Diseases 0.000 description 1
- 206010063094 Cerebral malaria Diseases 0.000 description 1
- 206010008909 Chronic Hepatitis Diseases 0.000 description 1
- 208000000668 Chronic Pancreatitis Diseases 0.000 description 1
- 208000010833 Chronic myeloid leukaemia Diseases 0.000 description 1
- 206010048843 Cytomegalovirus chorioretinitis Diseases 0.000 description 1
- 206010012438 Dermatitis atopic Diseases 0.000 description 1
- XBPCUCUWBYBCDP-UHFFFAOYSA-N Dicyclohexylamine Chemical class C1CCCCC1NC1CCCCC1 XBPCUCUWBYBCDP-UHFFFAOYSA-N 0.000 description 1
- 102100023275 Dual specificity mitogen-activated protein kinase kinase 3 Human genes 0.000 description 1
- 238000012286 ELISA Assay Methods 0.000 description 1
- 238000008157 ELISA kit Methods 0.000 description 1
- 208000037487 Endotoxemia Diseases 0.000 description 1
- 241000713730 Equine infectious anemia virus Species 0.000 description 1
- 241000588724 Escherichia coli Species 0.000 description 1
- 241000713800 Feline immunodeficiency virus Species 0.000 description 1
- 108010024636 Glutathione Proteins 0.000 description 1
- 102000005720 Glutathione transferase Human genes 0.000 description 1
- 108010070675 Glutathione transferase Proteins 0.000 description 1
- 206010018634 Gouty Arthritis Diseases 0.000 description 1
- 208000031886 HIV Infections Diseases 0.000 description 1
- 208000037357 HIV infectious disease Diseases 0.000 description 1
- 206010019233 Headaches Diseases 0.000 description 1
- 208000035186 Hemolytic Autoimmune Anemia Diseases 0.000 description 1
- 208000005176 Hepatitis C Diseases 0.000 description 1
- 206010019755 Hepatitis chronic active Diseases 0.000 description 1
- 101001009252 Homo sapiens 2-hydroxyacyl-CoA lyase 1 Proteins 0.000 description 1
- 101001115394 Homo sapiens Dual specificity mitogen-activated protein kinase kinase 3 Proteins 0.000 description 1
- 101000628954 Homo sapiens Mitogen-activated protein kinase 12 Proteins 0.000 description 1
- 206010021143 Hypoxia Diseases 0.000 description 1
- XQFRJNBWHJMXHO-RRKCRQDMSA-N IDUR Chemical compound C1[C@H](O)[C@@H](CO)O[C@H]1N1C(=O)NC(=O)C(I)=C1 XQFRJNBWHJMXHO-RRKCRQDMSA-N 0.000 description 1
- 101150081923 IL4 gene Proteins 0.000 description 1
- DGAQECJNVWCQMB-PUAWFVPOSA-M Ilexoside XXIX Chemical compound C[C@@H]1CC[C@@]2(CC[C@@]3(C(=CC[C@H]4[C@]3(CC[C@@H]5[C@@]4(CC[C@@H](C5(C)C)OS(=O)(=O)[O-])C)C)[C@@H]2[C@]1(C)O)C)C(=O)O[C@H]6[C@@H]([C@H]([C@@H]([C@H](O6)CO)O)O)O.[Na+] DGAQECJNVWCQMB-PUAWFVPOSA-M 0.000 description 1
- 206010061598 Immunodeficiency Diseases 0.000 description 1
- 208000029462 Immunodeficiency disease Diseases 0.000 description 1
- 102100026018 Interleukin-1 receptor antagonist protein Human genes 0.000 description 1
- 101710144554 Interleukin-1 receptor antagonist protein Proteins 0.000 description 1
- 208000007766 Kaposi sarcoma Diseases 0.000 description 1
- AYFVYJQAPQTCCC-GBXIJSLDSA-N L-threonine Chemical compound C[C@@H](O)[C@H](N)C(O)=O AYFVYJQAPQTCCC-GBXIJSLDSA-N 0.000 description 1
- OUYCCCASQSFEME-QMMMGPOBSA-N L-tyrosine Chemical compound OC(=O)[C@@H](N)CC1=CC=C(O)C=C1 OUYCCCASQSFEME-QMMMGPOBSA-N 0.000 description 1
- KDXKERNSBIXSRK-UHFFFAOYSA-N Lysine Natural products NCCCCC(N)C(O)=O KDXKERNSBIXSRK-UHFFFAOYSA-N 0.000 description 1
- 239000004472 Lysine Substances 0.000 description 1
- 102100028397 MAP kinase-activated protein kinase 3 Human genes 0.000 description 1
- 101710141393 MAP kinase-activated protein kinase 3 Proteins 0.000 description 1
- 102100026299 MAP kinase-interacting serine/threonine-protein kinase 1 Human genes 0.000 description 1
- 101710139011 MAP kinase-interacting serine/threonine-protein kinase 1 Proteins 0.000 description 1
- 206010027480 Metastatic malignant melanoma Diseases 0.000 description 1
- 229920000168 Microcrystalline cellulose Polymers 0.000 description 1
- 108090000744 Mitogen-Activated Protein Kinase Kinases Proteins 0.000 description 1
- 102000004232 Mitogen-Activated Protein Kinase Kinases Human genes 0.000 description 1
- 102100026932 Mitogen-activated protein kinase 12 Human genes 0.000 description 1
- 108700015928 Mitogen-activated protein kinase 13 Proteins 0.000 description 1
- 102000056248 Mitogen-activated protein kinase 13 Human genes 0.000 description 1
- 241000699670 Mus sp. Species 0.000 description 1
- 206010028289 Muscle atrophy Diseases 0.000 description 1
- 208000000112 Myalgia Diseases 0.000 description 1
- 102000047918 Myelin Basic Human genes 0.000 description 1
- 101710107068 Myelin basic protein Proteins 0.000 description 1
- 208000033761 Myelogenous Chronic BCR-ABL Positive Leukemia Diseases 0.000 description 1
- 208000033776 Myeloid Acute Leukemia Diseases 0.000 description 1
- MBBZMMPHUWSWHV-BDVNFPICSA-N N-methylglucamine Chemical compound CNC[C@H](O)[C@@H](O)[C@H](O)[C@H](O)CO MBBZMMPHUWSWHV-BDVNFPICSA-N 0.000 description 1
- GRYLNZFGIOXLOG-UHFFFAOYSA-N Nitric acid Chemical compound O[N+]([O-])=O GRYLNZFGIOXLOG-UHFFFAOYSA-N 0.000 description 1
- 206010030113 Oedema Diseases 0.000 description 1
- 229910019213 POCl3 Inorganic materials 0.000 description 1
- 206010033645 Pancreatitis Diseases 0.000 description 1
- 206010033647 Pancreatitis acute Diseases 0.000 description 1
- 206010033649 Pancreatitis chronic Diseases 0.000 description 1
- 208000018737 Parkinson disease Diseases 0.000 description 1
- PCNDJXKNXGMECE-UHFFFAOYSA-N Phenazine Natural products C1=CC=CC2=NC3=CC=CC=C3N=C21 PCNDJXKNXGMECE-UHFFFAOYSA-N 0.000 description 1
- ZLMJMSJWJFRBEC-UHFFFAOYSA-N Potassium Chemical compound [K] ZLMJMSJWJFRBEC-UHFFFAOYSA-N 0.000 description 1
- 201000001263 Psoriatic Arthritis Diseases 0.000 description 1
- 208000036824 Psoriatic arthropathy Diseases 0.000 description 1
- WTKZEGDFNFYCGP-UHFFFAOYSA-N Pyrazole Chemical compound C=1C=NNC=1 WTKZEGDFNFYCGP-UHFFFAOYSA-N 0.000 description 1
- 239000012979 RPMI medium Substances 0.000 description 1
- 239000012980 RPMI-1640 medium Substances 0.000 description 1
- NPXOKRUENSOPAO-UHFFFAOYSA-N Raney nickel Chemical compound [Al].[Ni] NPXOKRUENSOPAO-UHFFFAOYSA-N 0.000 description 1
- 208000033464 Reiter syndrome Diseases 0.000 description 1
- 208000005074 Retroviridae Infections Diseases 0.000 description 1
- 241000219061 Rheum Species 0.000 description 1
- 239000006146 Roswell Park Memorial Institute medium Substances 0.000 description 1
- 206010039710 Scleroderma Diseases 0.000 description 1
- 206010040550 Shigella infections Diseases 0.000 description 1
- 201000010001 Silicosis Diseases 0.000 description 1
- 229920002472 Starch Polymers 0.000 description 1
- 108010090804 Streptavidin Proteins 0.000 description 1
- QAOWNCQODCNURD-UHFFFAOYSA-N Sulfuric acid Chemical compound OS(O)(=O)=O QAOWNCQODCNURD-UHFFFAOYSA-N 0.000 description 1
- JQAWYCUUFIMTHE-WLTAIBSBSA-N Thr-Gly-Tyr Chemical group [H]N[C@@H]([C@@H](C)O)C(=O)NCC(=O)N[C@@H](CC1=CC=C(O)C=C1)C(O)=O JQAWYCUUFIMTHE-WLTAIBSBSA-N 0.000 description 1
- AYFVYJQAPQTCCC-UHFFFAOYSA-N Threonine Natural products CC(O)C(N)C(O)=O AYFVYJQAPQTCCC-UHFFFAOYSA-N 0.000 description 1
- 239000004473 Threonine Substances 0.000 description 1
- 108090000190 Thrombin Proteins 0.000 description 1
- 206010043781 Thyroiditis chronic Diseases 0.000 description 1
- 206010044248 Toxic shock syndrome Diseases 0.000 description 1
- 231100000650 Toxic shock syndrome Toxicity 0.000 description 1
- 108091023040 Transcription factor Proteins 0.000 description 1
- 102000040945 Transcription factor Human genes 0.000 description 1
- 206010048873 Traumatic arthritis Diseases 0.000 description 1
- 239000007983 Tris buffer Substances 0.000 description 1
- 241000700605 Viruses Species 0.000 description 1
- IQKBGDZGAIKORK-UHFFFAOYSA-N [C-]#[N+]C1=C(N(C)CC(C)(C)C)N=C(NC2=C(C)C=CC(C(=O)NOC)=C2)C(F)=C1 Chemical compound [C-]#[N+]C1=C(N(C)CC(C)(C)C)N=C(NC2=C(C)C=CC(C(=O)NOC)=C2)C(F)=C1 IQKBGDZGAIKORK-UHFFFAOYSA-N 0.000 description 1
- QGOJMBYPTWMNCN-QGZVFWFLSA-N [C-]#[N+]C1=C(N(C)CC(C)(C)C)N=C(N[C@@H]2CCNC2)N=C1NC1=CC(C(=O)NC)=CC=C1C Chemical compound [C-]#[N+]C1=C(N(C)CC(C)(C)C)N=C(N[C@@H]2CCNC2)N=C1NC1=CC(C(=O)NC)=CC=C1C QGOJMBYPTWMNCN-QGZVFWFLSA-N 0.000 description 1
- ULPYREBFPLGQNG-UHFFFAOYSA-N [C-]#[N+]C1=C(NC2=CC(C(=O)NOC)=CC=C2C)N=C(NCC(C)(C)C)C(F)=C1 Chemical compound [C-]#[N+]C1=C(NC2=CC(C(=O)NOC)=CC=C2C)N=C(NCC(C)(C)C)C(F)=C1 ULPYREBFPLGQNG-UHFFFAOYSA-N 0.000 description 1
- FYISENICNBGCQA-UHFFFAOYSA-N [C-]#[N+]C1=CC(F)=C(NC2=CC(C(=O)NOC)=CC=C2C)N=C1NCC(C)(C)C Chemical compound [C-]#[N+]C1=CC(F)=C(NC2=CC(C(=O)NOC)=CC=C2C)N=C1NCC(C)(C)C FYISENICNBGCQA-UHFFFAOYSA-N 0.000 description 1
- WETWJCDKMRHUPV-UHFFFAOYSA-N acetyl chloride Chemical compound CC(Cl)=O WETWJCDKMRHUPV-UHFFFAOYSA-N 0.000 description 1
- 239000012346 acetyl chloride Substances 0.000 description 1
- 239000013543 active substance Substances 0.000 description 1
- 231100000354 acute hepatitis Toxicity 0.000 description 1
- 201000003229 acute pancreatitis Diseases 0.000 description 1
- 125000005073 adamantyl group Chemical group C12(CC3CC(CC(C1)C3)C2)* 0.000 description 1
- 229910052783 alkali metal Inorganic materials 0.000 description 1
- 229910052784 alkaline earth metal Inorganic materials 0.000 description 1
- 125000000304 alkynyl group Chemical group 0.000 description 1
- HSFWRNGVRCDJHI-UHFFFAOYSA-N alpha-acetylene Natural products C#C HSFWRNGVRCDJHI-UHFFFAOYSA-N 0.000 description 1
- 238000010640 amide synthesis reaction Methods 0.000 description 1
- 125000003277 amino group Chemical group 0.000 description 1
- 150000003863 ammonium salts Chemical class 0.000 description 1
- 230000036592 analgesia Effects 0.000 description 1
- 238000004458 analytical method Methods 0.000 description 1
- 238000010171 animal model Methods 0.000 description 1
- ODKSFYDXXFIFQN-UHFFFAOYSA-N arginine Natural products OC(=O)C(N)CCCNC(N)=N ODKSFYDXXFIFQN-UHFFFAOYSA-N 0.000 description 1
- 229910052786 argon Inorganic materials 0.000 description 1
- 230000002917 arthritic effect Effects 0.000 description 1
- 239000012131 assay buffer Substances 0.000 description 1
- 239000012298 atmosphere Substances 0.000 description 1
- 201000008937 atopic dermatitis Diseases 0.000 description 1
- 201000005000 autoimmune gastritis Diseases 0.000 description 1
- 201000000448 autoimmune hemolytic anemia Diseases 0.000 description 1
- 230000001580 bacterial effect Effects 0.000 description 1
- 239000002585 base Substances 0.000 description 1
- 125000003785 benzimidazolyl group Chemical group N1=C(NC2=C1C=CC=C2)* 0.000 description 1
- 125000002619 bicyclic group Chemical group 0.000 description 1
- 230000027455 binding Effects 0.000 description 1
- 239000011230 binding agent Substances 0.000 description 1
- 238000004166 bioassay Methods 0.000 description 1
- 230000004071 biological effect Effects 0.000 description 1
- 230000033228 biological regulation Effects 0.000 description 1
- 230000000903 blocking effect Effects 0.000 description 1
- 208000019664 bone resorption disease Diseases 0.000 description 1
- 238000010804 cDNA synthesis Methods 0.000 description 1
- 239000011575 calcium Substances 0.000 description 1
- 229910052791 calcium Inorganic materials 0.000 description 1
- 125000003178 carboxy group Chemical group [H]OC(*)=O 0.000 description 1
- 239000003054 catalyst Substances 0.000 description 1
- 230000003197 catalytic effect Effects 0.000 description 1
- 230000001413 cellular effect Effects 0.000 description 1
- 239000003795 chemical substances by application Substances 0.000 description 1
- 239000012069 chiral reagent Substances 0.000 description 1
- 238000013375 chromatographic separation Methods 0.000 description 1
- 208000037976 chronic inflammation Diseases 0.000 description 1
- 208000037893 chronic inflammatory disorder Diseases 0.000 description 1
- 239000003086 colorant Substances 0.000 description 1
- 239000012230 colorless oil Substances 0.000 description 1
- 238000002648 combination therapy Methods 0.000 description 1
- 238000013270 controlled release Methods 0.000 description 1
- 238000007796 conventional method Methods 0.000 description 1
- DOBRDRYODQBAMW-UHFFFAOYSA-N copper(i) cyanide Chemical compound [Cu+].N#[C-] DOBRDRYODQBAMW-UHFFFAOYSA-N 0.000 description 1
- ZYGHJZDHTFUPRJ-UHFFFAOYSA-N coumarin Chemical compound C1=CC=C2OC(=O)C=CC2=C1 ZYGHJZDHTFUPRJ-UHFFFAOYSA-N 0.000 description 1
- 230000008878 coupling Effects 0.000 description 1
- 238000010168 coupling process Methods 0.000 description 1
- 238000005859 coupling reaction Methods 0.000 description 1
- 125000001995 cyclobutyl group Chemical group [H]C1([H])C([H])([H])C([H])(*)C1([H])[H] 0.000 description 1
- 125000000113 cyclohexyl group Chemical group [H]C1([H])C([H])([H])C([H])([H])C([H])(*)C([H])([H])C1([H])[H] 0.000 description 1
- 125000001511 cyclopentyl group Chemical group [H]C1([H])C([H])([H])C([H])([H])C([H])(*)C1([H])[H] 0.000 description 1
- 125000001559 cyclopropyl group Chemical group [H]C1([H])C([H])([H])C1([H])* 0.000 description 1
- 208000001763 cytomegalovirus retinitis Diseases 0.000 description 1
- 230000003247 decreasing effect Effects 0.000 description 1
- 238000000432 density-gradient centrifugation Methods 0.000 description 1
- BGRWYRAHAFMIBJ-UHFFFAOYSA-N diisopropylcarbodiimide Natural products CC(C)NC(=O)NC(C)C BGRWYRAHAFMIBJ-UHFFFAOYSA-N 0.000 description 1
- 239000003085 diluting agent Substances 0.000 description 1
- IUNMPGNGSSIWFP-UHFFFAOYSA-N dimethylaminopropylamine Chemical compound CN(C)CCCN IUNMPGNGSSIWFP-UHFFFAOYSA-N 0.000 description 1
- 230000003292 diminished effect Effects 0.000 description 1
- 230000006806 disease prevention Effects 0.000 description 1
- 239000006185 dispersion Substances 0.000 description 1
- VHJLVAABSRFDPM-QWWZWVQMSA-N dithiothreitol Chemical compound SC[C@@H](O)[C@H](O)CS VHJLVAABSRFDPM-QWWZWVQMSA-N 0.000 description 1
- 229940079593 drug Drugs 0.000 description 1
- 238000012377 drug delivery Methods 0.000 description 1
- 238000001035 drying Methods 0.000 description 1
- 230000009977 dual effect Effects 0.000 description 1
- 229940073621 enbrel Drugs 0.000 description 1
- 229960000403 etanercept Drugs 0.000 description 1
- 125000002534 ethynyl group Chemical group [H]C#C* 0.000 description 1
- 239000013604 expression vector Substances 0.000 description 1
- 239000000796 flavoring agent Substances 0.000 description 1
- 235000013355 food flavoring agent Nutrition 0.000 description 1
- XZBIXDPGRMLSTC-UHFFFAOYSA-N formohydrazide Chemical compound NNC=O XZBIXDPGRMLSTC-UHFFFAOYSA-N 0.000 description 1
- 238000001640 fractional crystallisation Methods 0.000 description 1
- 125000002541 furyl group Chemical group 0.000 description 1
- 239000007903 gelatin capsule Substances 0.000 description 1
- 238000007429 general method Methods 0.000 description 1
- 229960003180 glutathione Drugs 0.000 description 1
- 208000024908 graft versus host disease Diseases 0.000 description 1
- 239000003979 granulating agent Substances 0.000 description 1
- 239000001963 growth medium Substances 0.000 description 1
- 231100000869 headache Toxicity 0.000 description 1
- 201000011066 hemangioma Diseases 0.000 description 1
- 208000006454 hepatitis Diseases 0.000 description 1
- 208000005252 hepatitis A Diseases 0.000 description 1
- 208000002672 hepatitis B Diseases 0.000 description 1
- 125000005842 heteroatom Chemical group 0.000 description 1
- 125000004051 hexyl group Chemical group [H]C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])* 0.000 description 1
- 210000005260 human cell Anatomy 0.000 description 1
- 208000033519 human immunodeficiency virus infectious disease Diseases 0.000 description 1
- 150000004677 hydrates Chemical class 0.000 description 1
- NPZTUJOABDZTLV-UHFFFAOYSA-N hydroxybenzotriazole Substances O=C1C=CC=C2NNN=C12 NPZTUJOABDZTLV-UHFFFAOYSA-N 0.000 description 1
- 206010020718 hyperplasia Diseases 0.000 description 1
- 230000007954 hypoxia Effects 0.000 description 1
- 125000002632 imidazolidinyl group Chemical group 0.000 description 1
- 125000002636 imidazolinyl group Chemical group 0.000 description 1
- 125000002883 imidazolyl group Chemical group 0.000 description 1
- 230000008105 immune reaction Effects 0.000 description 1
- 238000003018 immunoassay Methods 0.000 description 1
- 230000007813 immunodeficiency Effects 0.000 description 1
- 238000011534 incubation Methods 0.000 description 1
- PZOUSPYUWWUPPK-UHFFFAOYSA-N indole Natural products CC1=CC=CC2=C1C=CN2 PZOUSPYUWWUPPK-UHFFFAOYSA-N 0.000 description 1
- RKJUIXBNRJVNHR-UHFFFAOYSA-N indolenine Natural products C1=CC=C2CC=NC2=C1 RKJUIXBNRJVNHR-UHFFFAOYSA-N 0.000 description 1
- 125000001041 indolyl group Chemical group 0.000 description 1
- 230000001939 inductive effect Effects 0.000 description 1
- 230000002458 infectious effect Effects 0.000 description 1
- 230000008595 infiltration Effects 0.000 description 1
- 238000001764 infiltration Methods 0.000 description 1
- 210000004969 inflammatory cell Anatomy 0.000 description 1
- 230000002757 inflammatory effect Effects 0.000 description 1
- 206010022000 influenza Diseases 0.000 description 1
- 239000004615 ingredient Substances 0.000 description 1
- 230000000977 initiatory effect Effects 0.000 description 1
- 150000007529 inorganic bases Chemical class 0.000 description 1
- 230000003834 intracellular effect Effects 0.000 description 1
- 230000004068 intracellular signaling Effects 0.000 description 1
- 238000007917 intracranial administration Methods 0.000 description 1
- 238000007918 intramuscular administration Methods 0.000 description 1
- 238000007919 intrasynovial administration Methods 0.000 description 1
- 238000007913 intrathecal administration Methods 0.000 description 1
- 238000001990 intravenous administration Methods 0.000 description 1
- 229910052740 iodine Inorganic materials 0.000 description 1
- 208000028867 ischemia Diseases 0.000 description 1
- 125000000959 isobutyl group Chemical group [H]C([H])([H])C([H])(C([H])([H])[H])C([H])([H])* 0.000 description 1
- 125000001972 isopentyl group Chemical group [H]C([H])([H])C([H])(C([H])([H])[H])C([H])([H])C([H])([H])* 0.000 description 1
- 125000001786 isothiazolyl group Chemical group 0.000 description 1
- 125000000842 isoxazolyl group Chemical group 0.000 description 1
- 208000021601 lentivirus infection Diseases 0.000 description 1
- 238000012417 linear regression Methods 0.000 description 1
- 239000000314 lubricant Substances 0.000 description 1
- 229910001629 magnesium chloride Inorganic materials 0.000 description 1
- 159000000003 magnesium salts Chemical class 0.000 description 1
- 230000036210 malignancy Effects 0.000 description 1
- 239000000463 material Substances 0.000 description 1
- 239000002609 medium Substances 0.000 description 1
- 208000021039 metastatic melanoma Diseases 0.000 description 1
- 235000019813 microcrystalline cellulose Nutrition 0.000 description 1
- 239000008108 microcrystalline cellulose Substances 0.000 description 1
- 229940016286 microcrystalline cellulose Drugs 0.000 description 1
- 238000010369 molecular cloning Methods 0.000 description 1
- 125000002950 monocyclic group Chemical group 0.000 description 1
- 206010028417 myasthenia gravis Diseases 0.000 description 1
- 208000010125 myocardial infarction Diseases 0.000 description 1
- 208000031225 myocardial ischemia Diseases 0.000 description 1
- 125000004108 n-butyl group Chemical group [H]C([H])([H])C([H])([H])C([H])([H])C([H])([H])* 0.000 description 1
- 125000004123 n-propyl group Chemical group [H]C([H])([H])C([H])([H])C([H])([H])* 0.000 description 1
- 230000002232 neuromuscular Effects 0.000 description 1
- 210000000440 neutrophil Anatomy 0.000 description 1
- 229910017604 nitric acid Inorganic materials 0.000 description 1
- 150000002825 nitriles Chemical group 0.000 description 1
- 125000002868 norbornyl group Chemical group C12(CCC(CC1)C2)* 0.000 description 1
- 125000002347 octyl group Chemical group [H]C([*])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])[H] 0.000 description 1
- 230000003287 optical effect Effects 0.000 description 1
- 239000006186 oral dosage form Substances 0.000 description 1
- 210000000056 organ Anatomy 0.000 description 1
- 230000003204 osmotic effect Effects 0.000 description 1
- 125000002971 oxazolyl group Chemical group 0.000 description 1
- 125000004043 oxo group Chemical group O=* 0.000 description 1
- 229910052760 oxygen Inorganic materials 0.000 description 1
- 125000004430 oxygen atom Chemical group O* 0.000 description 1
- 230000007170 pathology Effects 0.000 description 1
- 230000037361 pathway Effects 0.000 description 1
- 239000008188 pellet Substances 0.000 description 1
- 125000001147 pentyl group Chemical group C(CCCC)* 0.000 description 1
- 150000002978 peroxides Chemical class 0.000 description 1
- 125000004193 piperazinyl group Chemical group 0.000 description 1
- 125000003386 piperidinyl group Chemical group 0.000 description 1
- 238000003752 polymerase chain reaction Methods 0.000 description 1
- 230000001124 posttranscriptional effect Effects 0.000 description 1
- 239000011591 potassium Substances 0.000 description 1
- 229910052700 potassium Inorganic materials 0.000 description 1
- 159000000001 potassium salts Chemical class 0.000 description 1
- 239000000843 powder Substances 0.000 description 1
- 125000004368 propenyl group Chemical group C(=CC)* 0.000 description 1
- 230000000069 prophylactic effect Effects 0.000 description 1
- 125000002568 propynyl group Chemical group [*]C#CC([H])([H])[H] 0.000 description 1
- 150000003180 prostaglandins Chemical class 0.000 description 1
- 229940076372 protein antagonist Drugs 0.000 description 1
- 229940121649 protein inhibitor Drugs 0.000 description 1
- 239000012268 protein inhibitor Substances 0.000 description 1
- 125000003373 pyrazinyl group Chemical group 0.000 description 1
- 125000003072 pyrazolidinyl group Chemical group 0.000 description 1
- 125000002755 pyrazolinyl group Chemical group 0.000 description 1
- 125000003226 pyrazolyl group Chemical group 0.000 description 1
- 125000002098 pyridazinyl group Chemical group 0.000 description 1
- 150000003222 pyridines Chemical class 0.000 description 1
- 125000004076 pyridyl group Chemical group 0.000 description 1
- 150000003230 pyrimidines Chemical class 0.000 description 1
- 125000000168 pyrrolyl group Chemical group 0.000 description 1
- 125000002943 quinolinyl group Chemical group N1=C(C=CC2=CC=CC=C12)* 0.000 description 1
- 208000002574 reactive arthritis Diseases 0.000 description 1
- 238000006268 reductive amination reaction Methods 0.000 description 1
- 230000010410 reperfusion Effects 0.000 description 1
- 201000005404 rubella Diseases 0.000 description 1
- 229930195734 saturated hydrocarbon Natural products 0.000 description 1
- 231100000241 scar Toxicity 0.000 description 1
- 125000002914 sec-butyl group Chemical group [H]C([H])([H])C([H])([H])C([H])(*)C([H])([H])[H] 0.000 description 1
- 238000011894 semi-preparative HPLC Methods 0.000 description 1
- 201000005113 shigellosis Diseases 0.000 description 1
- 230000019491 signal transduction Effects 0.000 description 1
- 230000011664 signaling Effects 0.000 description 1
- 239000000741 silica gel Substances 0.000 description 1
- 229910002027 silica gel Inorganic materials 0.000 description 1
- 238000009097 single-agent therapy Methods 0.000 description 1
- 229910052708 sodium Inorganic materials 0.000 description 1
- 239000011780 sodium chloride Substances 0.000 description 1
- QDRKDTQENPPHOJ-UHFFFAOYSA-N sodium ethoxide Chemical compound [Na+].CC[O-] QDRKDTQENPPHOJ-UHFFFAOYSA-N 0.000 description 1
- 239000007790 solid phase Substances 0.000 description 1
- 230000009870 specific binding Effects 0.000 description 1
- 208000010110 spontaneous platelet aggregation Diseases 0.000 description 1
- 238000010561 standard procedure Methods 0.000 description 1
- 235000019698 starch Nutrition 0.000 description 1
- 230000035882 stress Effects 0.000 description 1
- 238000007920 subcutaneous administration Methods 0.000 description 1
- 239000000126 substance Substances 0.000 description 1
- 238000006467 substitution reaction Methods 0.000 description 1
- 235000000346 sugar Nutrition 0.000 description 1
- 150000008163 sugars Chemical class 0.000 description 1
- 150000003462 sulfoxides Chemical class 0.000 description 1
- 125000004434 sulfur atom Chemical group 0.000 description 1
- 239000006228 supernatant Substances 0.000 description 1
- 230000001629 suppression Effects 0.000 description 1
- 239000000725 suspension Substances 0.000 description 1
- 238000013268 sustained release Methods 0.000 description 1
- 239000012730 sustained-release form Substances 0.000 description 1
- 208000024891 symptom Diseases 0.000 description 1
- 208000011580 syndromic disease Diseases 0.000 description 1
- 201000004595 synovitis Diseases 0.000 description 1
- 201000000596 systemic lupus erythematosus Diseases 0.000 description 1
- QTEBQBMTMVSRGP-OAHLLOKOSA-N tert-butyl (3r)-3-[[4-fluoro-6-[2-methyl-5-(methylcarbamoyl)anilino]pyrimidin-2-yl]amino]pyrrolidine-1-carboxylate Chemical compound CNC(=O)C1=CC=C(C)C(NC=2N=C(N[C@H]3CN(CC3)C(=O)OC(C)(C)C)N=C(F)C=2)=C1 QTEBQBMTMVSRGP-OAHLLOKOSA-N 0.000 description 1
- KPNVXTDWARRMFL-HXUWFJFHSA-N tert-butyl (3r)-3-[[6-[2,2-dimethylpropyl(methyl)amino]-2-[2-methyl-5-(methylcarbamoyl)anilino]pyrimidin-4-yl]amino]pyrrolidine-1-carboxylate Chemical compound CNC(=O)C1=CC=C(C)C(NC=2N=C(C=C(N[C@H]3CN(CC3)C(=O)OC(C)(C)C)N=2)N(C)CC(C)(C)C)=C1 KPNVXTDWARRMFL-HXUWFJFHSA-N 0.000 description 1
- 125000000999 tert-butyl group Chemical group [H]C([H])([H])C(*)(C([H])([H])[H])C([H])([H])[H] 0.000 description 1
- ILMRJRBKQSSXGY-UHFFFAOYSA-N tert-butyl(dimethyl)silicon Chemical group C[Si](C)C(C)(C)C ILMRJRBKQSSXGY-UHFFFAOYSA-N 0.000 description 1
- 150000003536 tetrazoles Chemical class 0.000 description 1
- 125000003831 tetrazolyl group Chemical group 0.000 description 1
- 125000000335 thiazolyl group Chemical group 0.000 description 1
- 125000001544 thienyl group Chemical group 0.000 description 1
- 125000004568 thiomorpholinyl group Chemical group 0.000 description 1
- 229930192474 thiophene Natural products 0.000 description 1
- 229960004072 thrombin Drugs 0.000 description 1
- 206010043554 thrombocytopenia Diseases 0.000 description 1
- 230000009772 tissue formation Effects 0.000 description 1
- 230000008733 trauma Effects 0.000 description 1
- 230000008736 traumatic injury Effects 0.000 description 1
- LENZDBCJOHFCAS-UHFFFAOYSA-N tris Chemical compound OCC(N)(CO)CO LENZDBCJOHFCAS-UHFFFAOYSA-N 0.000 description 1
- 201000008827 tuberculosis Diseases 0.000 description 1
- OUYCCCASQSFEME-UHFFFAOYSA-N tyrosine Natural products OC(=O)C(N)CC1=CC=C(O)C=C1 OUYCCCASQSFEME-UHFFFAOYSA-N 0.000 description 1
- 238000011144 upstream manufacturing Methods 0.000 description 1
- 230000002792 vascular Effects 0.000 description 1
- 239000003981 vehicle Substances 0.000 description 1
Classifications
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D239/00—Heterocyclic compounds containing 1,3-diazine or hydrogenated 1,3-diazine rings
- C07D239/02—Heterocyclic compounds containing 1,3-diazine or hydrogenated 1,3-diazine rings not condensed with other rings
- C07D239/24—Heterocyclic compounds containing 1,3-diazine or hydrogenated 1,3-diazine rings not condensed with other rings having three or more double bonds between ring members or between ring members and non-ring members
- C07D239/28—Heterocyclic compounds containing 1,3-diazine or hydrogenated 1,3-diazine rings not condensed with other rings having three or more double bonds between ring members or between ring members and non-ring members with hetero atoms or with carbon atoms having three bonds to hetero atoms with at the most one bond to halogen, directly attached to ring carbon atoms
- C07D239/46—Two or more oxygen, sulphur or nitrogen atoms
- C07D239/48—Two nitrogen atoms
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P1/00—Drugs for disorders of the alimentary tract or the digestive system
- A61P1/02—Stomatological preparations, e.g. drugs for caries, aphtae, periodontitis
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P1/00—Drugs for disorders of the alimentary tract or the digestive system
- A61P1/04—Drugs for disorders of the alimentary tract or the digestive system for ulcers, gastritis or reflux esophagitis, e.g. antacids, inhibitors of acid secretion, mucosal protectants
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P1/00—Drugs for disorders of the alimentary tract or the digestive system
- A61P1/16—Drugs for disorders of the alimentary tract or the digestive system for liver or gallbladder disorders, e.g. hepatoprotective agents, cholagogues, litholytics
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P1/00—Drugs for disorders of the alimentary tract or the digestive system
- A61P1/18—Drugs for disorders of the alimentary tract or the digestive system for pancreatic disorders, e.g. pancreatic enzymes
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P13/00—Drugs for disorders of the urinary system
- A61P13/12—Drugs for disorders of the urinary system of the kidneys
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P17/00—Drugs for dermatological disorders
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P17/00—Drugs for dermatological disorders
- A61P17/02—Drugs for dermatological disorders for treating wounds, ulcers, burns, scars, keloids, or the like
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P17/00—Drugs for dermatological disorders
- A61P17/06—Antipsoriatics
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P19/00—Drugs for skeletal disorders
- A61P19/02—Drugs for skeletal disorders for joint disorders, e.g. arthritis, arthrosis
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P19/00—Drugs for skeletal disorders
- A61P19/06—Antigout agents, e.g. antihyperuricemic or uricosuric agents
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P19/00—Drugs for skeletal disorders
- A61P19/08—Drugs for skeletal disorders for bone diseases, e.g. rachitism, Paget's disease
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P19/00—Drugs for skeletal disorders
- A61P19/08—Drugs for skeletal disorders for bone diseases, e.g. rachitism, Paget's disease
- A61P19/10—Drugs for skeletal disorders for bone diseases, e.g. rachitism, Paget's disease for osteoporosis
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P21/00—Drugs for disorders of the muscular or neuromuscular system
- A61P21/04—Drugs for disorders of the muscular or neuromuscular system for myasthenia gravis
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P25/00—Drugs for disorders of the nervous system
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P25/00—Drugs for disorders of the nervous system
- A61P25/02—Drugs for disorders of the nervous system for peripheral neuropathies
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P25/00—Drugs for disorders of the nervous system
- A61P25/14—Drugs for disorders of the nervous system for treating abnormal movements, e.g. chorea, dyskinesia
- A61P25/16—Anti-Parkinson drugs
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P25/00—Drugs for disorders of the nervous system
- A61P25/28—Drugs for disorders of the nervous system for treating neurodegenerative disorders of the central nervous system, e.g. nootropic agents, cognition enhancers, drugs for treating Alzheimer's disease or other forms of dementia
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P27/00—Drugs for disorders of the senses
- A61P27/02—Ophthalmic agents
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P29/00—Non-central analgesic, antipyretic or antiinflammatory agents, e.g. antirheumatic agents; Non-steroidal antiinflammatory drugs [NSAID]
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P29/00—Non-central analgesic, antipyretic or antiinflammatory agents, e.g. antirheumatic agents; Non-steroidal antiinflammatory drugs [NSAID]
- A61P29/02—Non-central analgesic, antipyretic or antiinflammatory agents, e.g. antirheumatic agents; Non-steroidal antiinflammatory drugs [NSAID] without antiinflammatory effect
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P3/00—Drugs for disorders of the metabolism
- A61P3/08—Drugs for disorders of the metabolism for glucose homeostasis
- A61P3/10—Drugs for disorders of the metabolism for glucose homeostasis for hyperglycaemia, e.g. antidiabetics
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P31/00—Antiinfectives, i.e. antibiotics, antiseptics, chemotherapeutics
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P31/00—Antiinfectives, i.e. antibiotics, antiseptics, chemotherapeutics
- A61P31/12—Antivirals
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P31/00—Antiinfectives, i.e. antibiotics, antiseptics, chemotherapeutics
- A61P31/12—Antivirals
- A61P31/14—Antivirals for RNA viruses
- A61P31/18—Antivirals for RNA viruses for HIV
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P35/00—Antineoplastic agents
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P35/00—Antineoplastic agents
- A61P35/04—Antineoplastic agents specific for metastasis
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P37/00—Drugs for immunological or allergic disorders
- A61P37/02—Immunomodulators
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P37/00—Drugs for immunological or allergic disorders
- A61P37/02—Immunomodulators
- A61P37/04—Immunostimulants
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P37/00—Drugs for immunological or allergic disorders
- A61P37/02—Immunomodulators
- A61P37/06—Immunosuppressants, e.g. drugs for graft rejection
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P37/00—Drugs for immunological or allergic disorders
- A61P37/08—Antiallergic agents
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P43/00—Drugs for specific purposes, not provided for in groups A61P1/00-A61P41/00
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P7/00—Drugs for disorders of the blood or the extracellular fluid
- A61P7/02—Antithrombotic agents; Anticoagulants; Platelet aggregation inhibitors
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P7/00—Drugs for disorders of the blood or the extracellular fluid
- A61P7/04—Antihaemorrhagics; Procoagulants; Haemostatic agents; Antifibrinolytic agents
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P7/00—Drugs for disorders of the blood or the extracellular fluid
- A61P7/06—Antianaemics
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P9/00—Drugs for disorders of the cardiovascular system
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P9/00—Drugs for disorders of the cardiovascular system
- A61P9/04—Inotropic agents, i.e. stimulants of cardiac contraction; Drugs for heart failure
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P9/00—Drugs for disorders of the cardiovascular system
- A61P9/10—Drugs for disorders of the cardiovascular system for treating ischaemic or atherosclerotic diseases, e.g. antianginal drugs, coronary vasodilators, drugs for myocardial infarction, retinopathy, cerebrovascula insufficiency, renal arteriosclerosis
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D213/00—Heterocyclic compounds containing six-membered rings, not condensed with other rings, with one nitrogen atom as the only ring hetero atom and three or more double bonds between ring members or between ring members and non-ring members
- C07D213/02—Heterocyclic compounds containing six-membered rings, not condensed with other rings, with one nitrogen atom as the only ring hetero atom and three or more double bonds between ring members or between ring members and non-ring members having three double bonds between ring members or between ring members and non-ring members
- C07D213/04—Heterocyclic compounds containing six-membered rings, not condensed with other rings, with one nitrogen atom as the only ring hetero atom and three or more double bonds between ring members or between ring members and non-ring members having three double bonds between ring members or between ring members and non-ring members having no bond between the ring nitrogen atom and a non-ring member or having only hydrogen or carbon atoms directly attached to the ring nitrogen atom
- C07D213/60—Heterocyclic compounds containing six-membered rings, not condensed with other rings, with one nitrogen atom as the only ring hetero atom and three or more double bonds between ring members or between ring members and non-ring members having three double bonds between ring members or between ring members and non-ring members having no bond between the ring nitrogen atom and a non-ring member or having only hydrogen or carbon atoms directly attached to the ring nitrogen atom with hetero atoms or with carbon atoms having three bonds to hetero atoms with at the most one bond to halogen, e.g. ester or nitrile radicals, directly attached to ring carbon atoms
- C07D213/78—Carbon atoms having three bonds to hetero atoms, with at the most one bond to halogen, e.g. ester or nitrile radicals
- C07D213/84—Nitriles
- C07D213/85—Nitriles in position 3
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D239/00—Heterocyclic compounds containing 1,3-diazine or hydrogenated 1,3-diazine rings
- C07D239/02—Heterocyclic compounds containing 1,3-diazine or hydrogenated 1,3-diazine rings not condensed with other rings
- C07D239/24—Heterocyclic compounds containing 1,3-diazine or hydrogenated 1,3-diazine rings not condensed with other rings having three or more double bonds between ring members or between ring members and non-ring members
- C07D239/28—Heterocyclic compounds containing 1,3-diazine or hydrogenated 1,3-diazine rings not condensed with other rings having three or more double bonds between ring members or between ring members and non-ring members with hetero atoms or with carbon atoms having three bonds to hetero atoms with at the most one bond to halogen, directly attached to ring carbon atoms
- C07D239/46—Two or more oxygen, sulphur or nitrogen atoms
- C07D239/47—One nitrogen atom and one oxygen or sulfur atom, e.g. cytosine
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D239/00—Heterocyclic compounds containing 1,3-diazine or hydrogenated 1,3-diazine rings
- C07D239/02—Heterocyclic compounds containing 1,3-diazine or hydrogenated 1,3-diazine rings not condensed with other rings
- C07D239/24—Heterocyclic compounds containing 1,3-diazine or hydrogenated 1,3-diazine rings not condensed with other rings having three or more double bonds between ring members or between ring members and non-ring members
- C07D239/28—Heterocyclic compounds containing 1,3-diazine or hydrogenated 1,3-diazine rings not condensed with other rings having three or more double bonds between ring members or between ring members and non-ring members with hetero atoms or with carbon atoms having three bonds to hetero atoms with at the most one bond to halogen, directly attached to ring carbon atoms
- C07D239/46—Two or more oxygen, sulphur or nitrogen atoms
- C07D239/50—Three nitrogen atoms
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D401/00—Heterocyclic compounds containing two or more hetero rings, having nitrogen atoms as the only ring hetero atoms, at least one ring being a six-membered ring with only one nitrogen atom
- C07D401/02—Heterocyclic compounds containing two or more hetero rings, having nitrogen atoms as the only ring hetero atoms, at least one ring being a six-membered ring with only one nitrogen atom containing two hetero rings
- C07D401/12—Heterocyclic compounds containing two or more hetero rings, having nitrogen atoms as the only ring hetero atoms, at least one ring being a six-membered ring with only one nitrogen atom containing two hetero rings linked by a chain containing hetero atoms as chain links
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D403/00—Heterocyclic compounds containing two or more hetero rings, having nitrogen atoms as the only ring hetero atoms, not provided for by group C07D401/00
- C07D403/02—Heterocyclic compounds containing two or more hetero rings, having nitrogen atoms as the only ring hetero atoms, not provided for by group C07D401/00 containing two hetero rings
- C07D403/12—Heterocyclic compounds containing two or more hetero rings, having nitrogen atoms as the only ring hetero atoms, not provided for by group C07D401/00 containing two hetero rings linked by a chain containing hetero atoms as chain links
Definitions
- This invention relates to N-heterocyclic compounds that are effective in blocking cytokine production, and in particular the expression of TNF-alpha (TNF- ⁇ ), via inhibition of p38 kinase.
- Compounds of the present invention are useful in the treatment of inflammatory diseases such as, for example, rheumatoid arthritis.
- cytokines such as IL-1 and TNF- ⁇ is implicated in a wide variety of inflammatory diseases, including rheumatoid arthritis (RA), psoriasis, multiple sclerosis, inflammatory bowel disease, endotoxin shock, osteoporosis, Alzheimer's disease and congestive heart failure, among others [Henry et al., Drugs Fut., 24:1345-1354 (1999); Salituro et al., Curr. Med. Chem., 6:807-823 (1999)].
- protein antagonists of cytokines such as, for example, monoclonal antibody to TNF- ⁇ (Enbrel) [Rankin et al., Br. J.
- TNF- ⁇ is a protein whose synthesis occurs in many cell types in response to an external stimulus, such as, for example, a mitogen, an infectious organism, or trauma. Signaling from the cell surface to the nucleus proceeds via several intracellular mediators including kinases that catalyze phosphorylation of proteins downstream in the signaling cascade. Important mediators for the production of TNF- ⁇ cytokine are the mitogen-activated protein (MAP) kinases, and in particular, p38 kinase.
- MAP mitogen-activated protein
- p38 Kinases are activated in response to various stress stimuli, including, but not limited to, proinflammatory cytokines, endotoxin, ultraviolet light, and osmotic shock. Activation of p38 requires dual phosphorylation by upstream MAP kinase kinases (MKK3 and MKK6) on threonine and tyrosine within a Thr-Gly-Tyr motif, characteristic of p38 isozymes.
- MKK3 and MKK6 upstream MAP kinase kinases
- p38 kinases phosphorylate and activate the transcription factors, ATF-2, MAX, CHOP, and C/ERPb, suggesting a role of p38 kinases in gene regulation.
- p38 kinases phosphorylate other protein kinases, such as MAPK activated protein kinase-2/3 (MAPKAP-K2/3, or MK2/3), and MAP-kinase-interacting kinase 1/2 (MNK1/2).
- MAPK activated protein kinase-2/3 MAPK activated protein kinase-2/3
- MNK1/2 MAP-kinase-interacting kinase 1/2
- Small molecule inhibitors of p38 are expected to have several advantages over protein inhibitors of TNF- ⁇ or IL-1. p38 inhibitors not only block the production of TNF- ⁇ and IL-1, but also directly interfere with many of their secondary biological effects. In addition, small molecule inhibitors are unlikely to induce immune reaction in patients, and are believed active following oral administration.
- the present invention provides novel compounds that are potent and selective inhibitors of p38 ⁇ and ⁇ , and as such, are also potent inhibitors of TNF- ⁇ expression in human cells.
- Compounds of the present invention are useful in the treatment of p38- and TNF- ⁇ expression-mediated inflammatory and other disorders, including, but not limited to, bone resorption, graft vs.
- Atherosclerosis atherosclerosis, arthritis, osteoarthritis, rheumatoid arthritis, gout, psoriasis, topical inflammatory disease states, adult respiratory distress syndrome, asthma, chronic pulmonary inflammatory disease, cardiac reperfusion injury, renal reperfusion injury, thrombus, glomerulonephritis, Chron's disease, ulcerative colitis, inflammatory bowel disease, multiple sclerosis, endotoxin shock, osteoporosis, Alzheimer's disease, congestive heart failure and cachexia.
- the compounds of the present invention are effective as inhibitors of inappropriate p38 activity, especially iso forms ⁇ and ⁇ , and in turn, of cytokine production, and in particular, of cellular TNF-alpha (TNF- ⁇ ) expression. Accordingly, compounds of the invention are useful for the inhibition, prevention and suppression of various pathologies associated with such activity, such as, for example, inflammation, asthma, arthritis, atherosclerosis, multiple sclerosis, psoriasis, autoimmune diseases, Alzeheimers disease and congestive heart failure, among others.
- the principles of the present invention provide a compound, including isomers, enantiomers, diastereomers, tautomers, pharmaceutically acceptable salts, prodrugs and solvates thereof, represented by Formula (I):
- one or two of W, Y and X are ⁇ N—;
- W, Y and X is selected from ⁇ C—CN, ⁇ C—F, ⁇ C—NO 2 , ⁇ C—Br, ⁇ C—NH 2 , ⁇ C—NHC(O)CH 3 and ⁇ C—Cl;
- V is —NR 5 —;
- Z is halogen or —N(R 1 )(R 2 );
- R 1 and R 2 are the same or different and are selected from hydrogen, alkyl, substituted alkyl, aryl, substituted aryl, cycloalkyl, substituted cycloalkyl, heterocyclyl or substituted heterocyclyl;
- R 5 is hydrogen or alkyl
- R 7 is hydrogen, alkyl, substituted alkyl, alkoxy, or halogen
- R 8 is hydrogen, alkyl, alkyloxy or cyano
- R 9 is —C(O)R 10 or unsubstituted or substituted heterocyclyl
- R 10 is —N(R 31 )(R 32 );
- R 31 and R 32 are the same or different and are selected from hydrogen, alkyl, substituted alkyl, alkoxy, aryl, substituted aryl, cycloalkyl, substituted cycloalkyl, heterocyclyl or substituted heterocyclyl;
- R 11 is hydrogen, halogen, O—R 35 or —N(R 12 )(R 13 );
- R 12 is hydrogen, alkyl, or substituted alkyl
- R 13 is —(CH 2 ) m R 14 ;
- —N(R 12 )(R 13 ) taken together may form a heterocyclyl or substituted heterocyclyl;
- n 0, 1, 2 or 3;
- R 14 is hydrogen, alkyl, substituted alkyl, —C(O)N(R 31 )(R 32 ), —N(R 33 )C(O)R 34 , aryl, substituted aryl, cycloalkyl, substituted cycloalkyl, heterocyclyl, substituted heterocyclyl, heteroaryl, substituted heteroaryl or
- R 15 is hydrogen, alkyl or substituted alkyl
- R 16 is hydrogen or alkyl; or R 33 is hydrogen, alkyl, or substituted alkyl;
- R 34 is alkyl, substituted alkyl, aryl or substituted aryl
- R 35 is hydrogen or -(lower alkyl)-R 36 ;
- R 36 is N(R 37 )(R 38 );
- R 37 is hydrogen, alkyl, or substituted alkyl
- R 38 is -(substituted alkyl)-R 14 ;
- N(R 37 )(R 38 ) taken together may form a heterocyclyl or substituted heterocyclyl.
- Preferred compounds of this invention are those of Formula (I) including a pharmaceutically acceptable salt thereof
- one or two of W, Y and X are ⁇ N—;
- W, Y and X is selected from ⁇ C—CN, ⁇ C—F, ⁇ C—NO 2 , ⁇ C—Br, ⁇ C—NH 2 , ⁇ C—NHC(O)CH 3 and ⁇ C—Cl;
- V is —NH—
- Z is —N(R 1 )(R 2 );
- R 1 and R 2 are the same or different and are selected from hydrogen, alkyl or substituted alkyl wherein alkyl is of 1 to 8 carbons;
- R 7 is hydrogen, alkyl of 1 to 4 carbons, alkoxy of 1 to 4 carbons, or halogen;
- R 8 is hydrogen
- R 9 is —C(O)R 10 or unsubstituted or substituted heterocyclyl
- R 10 is —NH 2 or unsubstituted or substituted —NH-alkyl, —NH-alkoxy, —NH-heterocyclyl, —NH-phenyl, or —NH—CH 2 -phenyl wherein alkyl and alkoxy are of 1 to 6 carbons;
- R 11 is hydrogen, halogen, O—R 35 or —N(R 12 )(R 13 ), wherein N(R 12 )(R 13 ) taken together may form a monocyclic heterocyclyl or substituted heterocyclyl of 5 to 7 atoms containing 1, 2, or 3 additional nitrogen atoms or wherein
- R 12 is hydrogen
- R 13 is alkyl of 1 to 4 carbons or
- R 15 and R 16 are independently selected from hydrogen and methyl
- R 35 is hydrogen or -(lower alkyl)-R 36 ;
- R 36 is N(R 37 )(R 38 );
- R 37 is hydrogen, alkyl, or substituted alkyl
- R 38 is -(substituted alkyl)-R 14 ;
- N(R 37 )(R 38 ) taken together may form a heterocyclyl or substituted heterocyclyl.
- the principles of the present invention also provide methods of inhibiting TNF- ⁇ expression in a mammal, wherein the methods comprise administering to the mammal an effective amount of a compound represented by Formula (I), or a prodrug or salt thereof.
- inhibiting TNF- ⁇ expression is intended to include inhibiting, suppressing and preventing conditions associated with inappropriate TNF- ⁇ expression, including, but not limited to, inflammation, asthma, arthritis, atherosclerosis, multiple sclerosis, psoriasis, autoimmune diseases, Alzheimer's disease and congestive heart failure.
- the principles of the present invention further provide methods of treating p38 kinase and TNF- ⁇ mediated disorders in a mammal, the methods comprising
- a p38 kinase mediated disorder means a disorder associated with inappropriate p38 kinase activity
- a TNF- ⁇ mediated disorder means a disorder associated with inappropriate TNF- ⁇ expression.
- disorders include, but are not limited to, inflammation, asthma, arthritis, atherosclerosis, multiple sclerosis, psoriasis, autoimmune diseases, Alzheimer ⁇ s disease and congestive heart failure.
- the compounds of the invention may be used in the manufacture of a pharmaceutical composition or medicament for the prophylactic or therapeutic treatment of disease states in mammals.
- the compounds of the present invention may be administered as pharmaceutical compositions as a monotherapy, or in combination with, for example, other anti-inflammatory, e.g. a steroid or NSAID (non-steroidal anti-inflammatory drug) and/or immunosuppressive agents.
- a steroid or NSAID non-steroidal anti-inflammatory drug
- immunosuppressive agents can involve the administration of the various pharmaceuticals as a single dosage form or as multiple dosage forms administered simultaneously or sequentially.
- Suitable routes of administration may be employed for providing a patient with an effective amount of a compound of the present invention.
- Suitable routes of administration may include, for example, oral, rectal, nasal, buccal, parenteral (such as, intravenous, intrathecal, subcutaneous, intramuscular, intrasternal, intrahepatic, intralesional, intracranial, intra-articular, and intra-synovial), transdermal (such as, for example, patches), and the like.
- parenteral such as, intravenous, intrathecal, subcutaneous, intramuscular, intrasternal, intrahepatic, intralesional, intracranial, intra-articular, and intra-synovial
- transdermal such as, for example, patches
- oral dosage forms such as, for example, tablets, troches, dispersions, suspensions, solutions, capsules, soft gelatin capsules, and the like, may be preferred.
- Administration may also be by controlled or sustained release means and delivery devices. Methods for the preparation of such dosage forms are well known in
- compositions incorporating compounds of the present invention may include excipients, a pharmaceutically acceptable carrier, in addition to other therapeutic ingredients.
- Excipients such as starches, sugars, microcrystalline cellulose, diluents, lubricants, binders, coloring agents, flavoring agents, granulating agents, disintegrating agents, and the like may be appropriate depending upon the route of administration. Because of their ease of administration, tablets and capsules represent the most advantageous oral dosage unit forms. If desired, tablets may be coated by standard aqueous or nonaqueous techniques.
- the compounds of the present invention may be used in the form of pharmaceutically acceptable salts derived from inorganic or organic bases, and hydrates thereof. Included among such base salts are ammonium salts, alkali metal salts, such as sodium and potassium salts, alkaline earth metal salts, such as calcium and magnesium salts, salts with organic bases, such as dicyclohexylamine salts, N-methyl-D-glucamine, and salts with amino acids such as arginine, lysine, and so forth.
- base salts include ammonium salts, alkali metal salts, such as sodium and potassium salts, alkaline earth metal salts, such as calcium and magnesium salts, salts with organic bases, such as dicyclohexylamine salts, N-methyl-D-glucamine, and salts with amino acids such as arginine, lysine, and so forth.
- the present invention provides a novel compound of Formula (I) including isomers, enantiomers, diastereomers, tautomers, pharmaceutically acceptable salts, prodrugs and solvates thereof, comprising:
- one or two of W, Y and X are ⁇ N—;
- W, Y and X is selected from ⁇ C—CN, ⁇ C—F, ⁇ C—NO 2 , ⁇ C—Br, ⁇ C—NH 2 , ⁇ C—NHC(O)CH 3 and ⁇ C—Cl;
- V is —NR 5 —;
- Z is halogen or —N(R 1 )(R 2 );
- R 1 and R 2 are the same or different and are selected from hydrogen, alkyl, substituted alkyl, aryl, substituted aryl, cycloalkyl, substituted cycloalkyl, heterocyclyl or substituted heterocyclyl;
- R 5 is hydrogen or alkyl
- R 7 is hydrogen, alkyl, substituted alkyl, alkoxy, or halogen
- R 8 is hydrogen, alkyl, alkyloxy or cyano
- R 9 is —C(O)R 10 or unsubstituted or substituted heterocyclyl
- R 10 is —N(R 31 )(R 32 );
- R 31 and R 32 are the same or different and are selected from hydrogen, alkyl, substituted alkyl, alkoxy, aryl, substituted aryl, cycloalkyl, substituted cycloalkyl, heterocyclyl or substituted heterocyclyl;
- R 11 is hydrogen, halogen, O—R 35 or —N(R 12 )(R 13 );
- R 12 is hydrogen, alkyl, or substituted alkyl
- R 13 is —(CH 2 ) m R 14 ;
- —N(R 12 )(R 13 ) taken together may form a heterocyclyl or substituted heterocyclyl;
- n 0, 1, 2 or 3;
- R 14 is hydrogen, alkyl, substituted alkyl, —C(O)N(R 31 )(R 32 ), —N(R 33 )C(O)R 34 , aryl, substituted aryl, cycloalkyl, substituted cycloalkyl, heterocyclyl, substituted heterocyclyl, heteroaryl, substituted heteroaryl or
- R 15 is hydrogen, alkyl or substituted alkyl
- R 16 is hydrogen or alkyl
- R 33 is hydrogen, alkyl, or substituted alkyl
- R 34 is alkyl, substituted alkyl, aryl or substituted aryl
- R 35 is hydrogen or -(lower alkyl)-R 36 ;
- R 36 is N(R 37 )(R 38 );
- R 37 is hydrogen, alkyl, or substituted alkyl
- R 38 is -(substituted alkyl)-R 14 ;
- N(R 37 )(R 38 ) taken together may form a heterocyclyl or substituted heterocyclyl.
- the present invention provides a compound of Formula (I) including isomers, enantiomers, diastereomers, tautomers, pharmaceutically acceptable salts, prodrugs and solvates thereof,
- one or two of W, Y and X are ⁇ N—;
- W, Y and X is selected from ⁇ C—CN, ⁇ C—F, ⁇ C—NO 2 , ⁇ C—Br, ⁇ C—NH 2 , ⁇ C—NHC(O)CH 3 and ⁇ C—Cl;
- V is —NH—
- Z is —N(R 1 )(R 2 );
- R 1 and R 2 are the same or different and are selected from hydrogen, alkyl or substituted alkyl wherein alkyl is of 1 to 8 carbons;
- R 7 is hydrogen, alkyl of 1 to 4 carbons, alkoxy of 1 to 4 carbons, or halogen;
- R 8 is hydrogen
- R 9 is —C(O)R 10 or unsubstituted or substituted heterocyclyl
- R 10 is —NH 2 or unsubstituted or substituted —NH-alkyl, —NH-alkoxy, —NH-heterocyclyl, —NH-phenyl, or —NH—CH 2 -phenyl wherein alkyl and alkoxy are of 1 to 6 carbons;
- R 11 is hydrogen, halogen, O—R 35 or —N(R 12 )(R 13 ), wherein N(R 12 )(R 13 ) taken together may form a monocyclic heterocyclyl or substituted heterocyclyl of 5 to 7 atoms containing 1, 2, or 3 additional nitrogen atoms or wherein
- R 12 is hydrogen
- R 13 is alkyl of 1 to 4 carbons or
- R 15 and R 16 are independently selected from hydrogen and methyl
- R 35 is hydrogen or -(lower alkyl)-R 36 ;
- R 36 is N(R 37 )(R 38 );
- R 37 is hydrogen, alkyl, or substituted alkyl
- R 38 is -(substituted alkyl)-R 14 ;
- N(R 37 )(R 38 ) taken together may form a heterocyclyl or substituted heterocyclyl.
- the present invention provides a compound of Formula (I) including isomers, enantiomers, diastereomers, tautomers, pharmaceutically acceptable salts, prodrugs and solvates thereof,
- one or two of W, Y and X are ⁇ N—;
- W, Y and X is selected from ⁇ C—CN, ⁇ C—F, ⁇ C—NO 2 , ⁇ C—Br, ⁇ C—NH 2 , ⁇ C—NHC(O)CH 3 and ⁇ C—Cl;
- V is —NH—
- Z is —N(R 1 )(R 2 );
- R 1 and R 2 are the same or different and are selected from hydrogen or alkyl of 1 to 8 carbons;
- R 7 is hydrogen, methyl, methoxy, Cl, Br, or F
- R 8 is hydrogen
- R 9 is —C(O)R 10 or unsubstituted or substituted heterocyclyl
- R 10 is —NH 2 , or unsubstituted or substituted —NH-alkyl, —NH-alkoxy, —NH-phenyl, or —NH—CH 2 -phenyl wherein alkyl and alkoxy are of 1 to 6 carbons;
- R 11 is hydrogen, halogen, O—R 35 or —N(R 12 )(R 13 ) wherein N(R 12 )(R 13 ) taken together form a monocyclic heterocyclyl or substituted heterocyclyl of 5 to 7 atoms containing 1, 2, or 3 additional nitrogen atoms.
- the present invention provides a compound of Formula (I) including isomers, enantiomers, diastereomers, tautomers, pharmaceutically acceptable salts, prodrugs and solvates thereof,
- one of W, Y and X is ⁇ N—;
- W, Y and X is selected from ⁇ C—CN, ⁇ C—F, ⁇ C—NO 2 , ⁇ C—Br, ⁇ C—NH 2 , ⁇ C—NHC(O)CH 3 and ⁇ C—Cl;
- V is —NH—
- Z is —N(R 1 )(R 2 );
- R 1 and R 2 are the same or different and are selected from hydrogen or alkyl of 1 to 8 carbons;
- R 7 is hydrogen, methyl, methoxy, Cl, Br, or F
- R 8 is hydrogen
- R 9 is —C(O)R 10 or unsubstituted or substituted heterocyclyl
- R 10 is —NH 2 , or unsubstituted or substituted —NH-alkyl, —NH-alkoxy, —NH-phenyl, or —NH—CH 2 -phenyl wherein alkyl and alkoxy are of 1 to 6 carbons;
- R 11 is hydrogen, halogen, —O—R 35 or —N(R 12 )(R 13 ) wherein N(R 12 )(R 13 ) taken together form a monocyclic heterocyclyl or substituted heterocyclyl of 5 to 7 atoms containing 1, 2, or 3 additional nitrogen atoms; and
- R 15 and R 16 are independently selected from hydrogen and methyl.
- the present invention provides a compound of Formula (I) including isomers, enantiomers, diastereomers, tautomers, pharmaceutically acceptable salts, prodrugs and solvates thereof,
- R 10 is —NH 2 , unsubstituted or substituted —NH—CH 3 , —NH—C 2 H 5 , —NH—OCH 3 , or —NH—OC 2 H 5 .
- the present invention provides a compound of Formula (I) including isomers, enantiomers, diastereomers, tautomers, pharmaceutically acceptable salts, prodrugs and solvates thereof,
- R 9 is unsubstituted or substituted triazole, thiazole, oxadiazole or imidazole.
- the present invention provides a compound of Formula (I) including isomers, enantiomers, diastereomers, tautomers, pharmaceutically acceptable salts, prodrugs and solvates thereof,
- R 11 is hydrogen, halogen, —O-(substituted alkyl), —NH— (substituted alkyl) or
- the present invention provides a compound of Formula (I) including isomers, enantiomers, diastereomers, tautomers, pharmaceutically acceptable salts, prodrugs and solvates thereof,
- R 11 is hydrogen, halogen, —O-(substituted alkyl), —NH-(substituted alkyl) or
- the present invention provides a compound of Formula (I) including isomers, enantiomers, diastereomers, tautomers, pharmaceutically acceptable salts, prodrugs and solvates thereof,
- the remaining W, Y or X is selected from ⁇ C—CN, ⁇ C—F, ⁇ C—NO 2 , ⁇ C—Br, ⁇ C—NH 2 , ⁇ C—NHC(O)CH 3 and ⁇ C—Cl;
- V is —NH—
- Z is —N(R 1 )(R 2 );
- R 1 and R 2 are the same or different and are selected from hydrogen or alkyl of 1 to 8 carbons;
- R 7 is hydrogen, methyl, methoxy, Cl, Br, or F
- R 8 is hydrogen
- R 9 is —C(O)R 10 or unsubstituted or substituted heterocyclyl
- R 10 is —NH 2 , or unsubstituted or substituted —NH-alkyl, —NH-alkoxy, —NH-phenyl, or —NH—CH 2 -phenyl wherein alkyl and alkoxy are of 1 to 6 carbons;
- R 11 is hydrogen, halogen, —O—R 35 or —N(R 12 )(R 13 ), wherein N(R 12 )(R 13 ) taken together may form a monocyclic heterocyclyl or substituted heterocyclyl of 5 to 7 atoms containing 1, 2, or 3 additional nitrogen atoms; and
- R 15 and R 16 are independently selected from hydrogen and methyl.
- the present invention provides a compound of Formula (I) including isomers, enantiomers, diastereomers, tautomers, pharmaceutically acceptable salts, prodrugs and solvates thereof,
- R 10 is —NH 2 , unsubstituted or substituted —NH—CH 3 , —NH—C 2 H 5 , —NH—OCH 3 , or —NH—OC 2 H 5 .
- the present invention provides a compound of Formula (I) including isomers, enantiomers, diastereomers, tautomers, pharmaceutically acceptable salts, prodrugs and solvates thereof,
- R 9 is unsubstituted or substituted triazole, thiazole, oxadiazole or imidazole.
- the present invention provides a compound of Formula (I) including isomers, enantiomers, diastereomers, tautomers, pharmaceutically acceptable salts, prodrugs and solvates thereof,
- R 11 is hydrogen, halogen, —O-(substituted alkyl), —NH-(substituted alkyl) or
- the present invention provides a compound of Formula (I) including isomers, enantiomers, diastereomers, tautomers, pharmaceutically acceptable salts, prodrugs and solvates thereof,
- R 11 is hydrogen, halogen, —O-(substituted alkyl), —NH-(substituted alkyl) or
- the present invention provides a pharmaceutical composition
- a pharmaceutical composition comprising as an active ingredient, a compound of the invention or a prodrug or salt thereof, and a pharmaceutically acceptable carrier.
- the present invention provides a pharmaceutical composition further comprising one or more additional active ingredients.
- the present invention provides a pharmaceutical composition wherein the additional active ingredient is an anti-inflammatory compound or an immunosuppressive agent.
- the present invention provides a pharmaceutical composition wherein the additional active ingredient is chosen from a steroid and an NSAID.
- the present invention provides a method of inhibiting TNF- ⁇ expression in a mammal comprising administering to the mammal an effective amount of the pharmaceutical composition of the invention.
- the present invention provides a method of treating TNF- ⁇ mediated disorder comprising administering to a mammal in need of such treatment, an effective amount of a pharmaceutical composition of the invention.
- the present invention provides a method of treating TNF- ⁇ mediated disorder, wherein the TNF- ⁇ mediated disorder is an inflammatory disorder.
- the present invention provides a method of treating TNF- ⁇ mediated disorder, wherein the TNF- ⁇ mediated disorder is chosen from bone resorption, graft vs. host reaction, atherosclerosis, arthritis, osteoarthritis, rheumatoid arthritis, gout, psoriasis, topical inflammatory disease states, adult respiratory distress syndrome, asthma, chronic pulmonary inflammatory disease, cardiac reperfusion injury, renal reperfusion injury, thrombus, glomerulonephritis, Chron's disease, ulcerative colitis, inflammatory bowel disease, multiple sclerosis, endotoxin shock, osteoporosis, Alzheimer's disease, congestive heart failure and cachexia.
- the TNF- ⁇ mediated disorder is chosen from bone resorption, graft vs. host reaction, atherosclerosis, arthritis, osteoarthritis, rheumatoid arthritis, gout, psoriasis, topical inflammatory disease states, adult respiratory distress syndrome, asthma, chronic pulmonary
- the present invention provides a method of treating TNF- ⁇ mediated disorder wherein the pharmaceutical composition of the invention is administered with one or more additional anti-inflammatory or immunosuppressive agents as a single dose form or as separate dosage forms.
- the present invention provides a method of treating a condition associated with TNF- ⁇ expression in a mammal comprising administering to a mammal in need of such treatment, an effective amount of a pharmaceutical composition of the invention.
- the present invention provides a method of treating a condition associated with TNF- ⁇ expression in a mammal wherein the condition associated with TNF- ⁇ expression is an inflammatory disorder.
- the present invention provides a method of treating a condition associated with TNF- ⁇ expression, wherein the condition associated with TNF- ⁇ expression is chosen from bone resorption, graft vs. host reaction, atherosclerosis, arthritis, osteoarthritis, rheumatoid arthritis, gout, psoriasis, topical inflammatory disease states, adult respiratory distress syndrome, asthma, chronic pulmonary inflammatory disease, cardiac reperfusion injury, renal reperfusion injury, thrombus, glomerulonephritis, Crohn's disease, ulcerative colitis, inflammatory bowel disease, multiple sclerosis, endotoxin shock, osteoporosis, Alzheimer's disease, congestive heart failure and cachexia.
- the condition associated with TNF- ⁇ expression is chosen from bone resorption, graft vs. host reaction, atherosclerosis, arthritis, osteoarthritis, rheumatoid arthritis, gout, psoriasis, topical inflammatory disease states, adult respiratory distress syndrome,
- the present invention provides a method of treating a condition associated with TNF- ⁇ expression wherein the pharmaceutical composition of the invention is administered with one or more additional anti-inflammatory or immunosuppressive agents as a single dose form or as separate dosage forms.
- the present invention provides a method of treating a condition associated with p38 kinase activity in a mammal comprising administering to a mammal in need of such treatment, an effective amount of a pharmaceutical composition of the invention.
- the present invention provides a method of treating a condition associated with p38 kinase activity in a mammal wherein the condition associated with p38 activity is an inflammatory disorder.
- the present invention provides a method of treating a condition associated with p38 kinase activity is chosen from bone resorption, graft vs. host reaction, atherosclerosis, arthritis, osteoarthritis, rheumatoid arthritis, gout, psoriasis, topical inflammatory disease states, adult respiratory distress syndrome, asthma, chronic pulmonary inflammatory disease, cardiac reperfusion injury, renal reperfusion injury, thrombus, glomerulonephritis, Chron's disease, ulcerative colitis, inflammatory bowel disease, multiple sclerosis, endotoxin shock, osteoporosis, Alzheimer's disease, congestive heart failure and cachexia.
- a condition associated with p38 kinase activity is chosen from bone resorption, graft vs. host reaction, atherosclerosis, arthritis, osteoarthritis, rheumatoid arthritis, gout, psoriasis, topical inflammatory disease states, adult respiratory distress syndrome, asthma, chronic
- the present invention provides a method of treating a condition associated with p38 activity wherein the pharmaceutical composition of the invention is administered with one or more additional anti-inflammatory or immunosuppressive agents as a single dose form or as separate dosage forms.
- the present invention provides a compound including isomers, enantiomers, diastereomers, tautomers, pharmaceutically acceptable salts, prodrugs and solvates selected from:
- Alkyl is intended to include linear or branched hydrocarbon structures and combinations thereof of 1 to 20 carbons.
- “Lower alkyl” means alkyl groups of from 1 to about 10, preferably from 1 to about 8, and more preferably, from 1 to about 6 carbon atoms. Examples of such radicals include methyl, ethyl, n-propyl, isopropyl, n-butyl, isobutyl, s-butyl, t-butyl, pentyl, iso-amyl, hexyl, octyl and the like.
- Aryl means an aromatic hydrocarbon radical of 6 to about 16 carbon atoms, preferably of 6 to about 12 carbon atoms, and more preferably of 6 to about 10 carbon atoms.
- Examples of aryl groups are phenyl, which is preferred, 1-naphthyl and 2-naphthyl.
- Cycloalkyl refers to saturated hydrocarbon ring structures of from 3 to 12 carbon atoms, and preferably from 3 to 6 carbon atoms. Examples include cyclopropyl, cyclobutyl, cyclopentyl, cyclohexyl, norbornyl, adamantyl, and the like. “Lower cycloalkyl” refers to cycloalkyl of 3 to 6 carbons.
- Heterocyclyl refers to saturated, partially saturated or unsaturated monocyclic structures of from 3 to 8 atoms, preferably 5 or 6 atoms, and bicyclic structures of 9 or 10 atoms containing one or more carbon atoms and from 1 to 4 heteroatoms chosen from O, N, and S. The point of attachment of the heterocyclyl structure is at an available carbon or nitrogen atom.
- Examples include: imidazole, pyridine, indole, thiophene, benzopyranone, thiazole, furan, benzimidazole, quinoline, isoquinoline, quinoxaline, pyrimidine, pyrazine, tetrazole, pyrazole, pyrrolyl, pyridinyl, pyrazolyl, triazolyl, pyrimidinyl, pyridazinyl, oxazolyl, thiazolyl, imidazolyl, indolyl, thiophenyl, furanyl, tetrazolyl, 2-pyrrolinyl, 3-pyrrolinyl, pyrrolindinyl, 1,3-dioxolanyl, imidazolinyl, imidazolidinyl, pyrazolinyl, pyrazolidinyl, isoxazolyl, isothiazolyl, 1,2,3-oxadiazoly
- Alkoxy means a straight, branched or cyclic hydrocarbon configuration and combinations thereof, including from 1 to 20 carbon atoms, preferably from 1 to 8 carbon atoms, more preferably from 1 to about 4 carbon atoms, and an oxygen atom at the point of attachment.
- Suitable alkoxy groups include methoxy, ethoxy, n-propoxy, isopropoxy, n-butoxy, iso-butoxy, s-butoxy, t-butoxy, cyclopropyloxy, cyclohexyloxy, and the like.
- “Lower alkoxy” refers to alkoxy groups having from 1 to 4 carbon atoms.
- alkylthio refers to such groups having a sulfur atom at the point of attachment.
- Alkenyl refers to an unsaturated acyclic hydrocarbon radical in so much as it contains at least one double bond. “Lower alkenyl” refers to such radicals containing from about 2 to about 10 carbon atoms, preferably from about 2 to about 8 carbon atoms and more preferably 2 to about 6 carbon atoms. Examples of suitable alkenyl radicals include propenyl, buten-1-yl, isobutenyl, penten-1-yl, 2-methylbuten-1-yl, 3-methylbuten-1-yl, hexen-1-yl, hepten-1-yl, and octen-1-yl, and the like.
- Alkynyl refers to an unsaturated acyclic hydrocarbon radical containing at least one triple bond. Examples include ethynyl, propynyl, and the like.
- “Substituted alkyl” means an alkyl wherein one or more hydrogens, preferably one, two, or three hydrogens, attached to an aliphotic carbon are replaced with a substituent such as —N(R 31 )(R 32 ), alkoxy, alkylthio, halogen, cyano, carboxyl, hydroxyl, —SO 2 -alkyl, —CO 2 -alkyl, —C(O)-alkyl, nitro, cycloalkyl, substituted cycloalkyl, aryl, substituted aryl, heterocyclyl, substituted heterocyclyl, —C(O)—N(R 31 )(R 32 ), or —NH—C(O)-alkyl.
- substituent groups include methoxy, ethoxy, propoxy, amino, methylamino, dimethylamino, phenyl naphthyl, chlorine, fluorine, and the like.
- “Substituted cycloalkyl” means a cycloalkyl wherein one or more hydrogens, preferably one, two or three hydrogens, attached to a ring carbon are replaced with a substituent such as alkyl, substituted alkyl, —N(R 31 )(R 32 ), alkoxy, alkylthio, aryl, substituted aryl, halogen, cyano, carboxyl, hydroxyl, nitro, —SO 2 -alkyl, —CO 2 -alkyl, —C(O)-alkyl, —C(O)—N(R 31 )R 32 ), or —NH—C(O)-alkyl.
- cycloalkyl rings having a fused aryl, preferably phenyl, or cycloalkyl such as
- “Substituted aryl” means an aryl wherein one or more hydrogens, preferably one, two or three hydrogens, attached to an aromatic carbon are replaced with a substituent such as alkyl, substituted alkyl, —N(R 31 )(R 32 ), alkoxy, alkylthio, aryl, substituted aryl, halogen, cyano, nitro, carboxyl, hydroxyl, —SO 2 -alkyl, —CO 2 -alkyl, —C(O)-alkyl, —C(O)—N(R 31 )(R 32 ), or —NH—C(O)-alkyl.
- a substituent such as alkyl, substituted alkyl, —N(R 31 )(R 32 ), alkoxy, alkylthio, aryl, substituted aryl, halogen, cyano, nitro, carboxyl, hydroxyl, —SO 2 -alkyl, —
- substituents include methyl, isopropyl, methoxy, ethoxy, propoxy, amino, methylamino, dimethylamino, phenyl, chlorine, fluorine, —CO 2 CH 3 , —C(O)—NH 2 , and the like.
- “Substituted heterocyclyl” means a heterocyclyl substituted at one or more available carbon or nitrogen atoms, preferably at one or two carbon and/or nitrogen atoms, with a substituent such as alkyl, substituted alkyl, —N(R 31 )(R 32 ), alkoxy, alkylthio, aryl, substituted aryl, halogen, cyano, nitro, oxo, carboxyl, hydroxyl, —SO 2 -alkyl, —CO 2 -alkyl, —C(O)-alkyl, —C(O)—N(R 31 )(R 32 ), or —NH—C(O)-alkyl.
- substituent such as alkyl, substituted alkyl, —N(R 31 )(R 32 ), alkoxy, alkylthio, aryl, substituted aryl, halogen, cyano, nitro, oxo, carboxyl,
- Halogen is intended to include for example, F, Cl, Br and I.
- prodrug refers to a chemical compound that is converted to an active agent by metabolic processes in vivo.
- a prodrug of a compound of Formula I is intended to mean any compound that is converted to a compound of Formula I by metabolic processes in vivo.
- the use of prodrugs of compounds of Formula I in any of the methods described herein is contemplated and is intended to be within the scope of the invention.
- a protecting group refers to a group which is used to mask a functionality during a process step in which it would otherwise react, but in which reaction is undesirable.
- the protecting group prevents reaction at that step, but may be subsequently removed to expose the original functionality. The removal or “deprotection” occurs after the completion of the reaction or reactions in which the functionality would interfere.
- the typical functionalities that must be protected are amines.
- Suitable groups for that purpose are discussed in standard textbooks in the field of chemistry, such as Protective Groups in Organic Synthesis by T. W. Greene [John Wiley & Sons, New York, 1991], which is incorporated herein by reference. Particular attention is drawn to the chapter entitled “Protection for the Amino Group” (pages 309-405).
- Preferred protecting groups include BOC and Fmoc. Exemplary methods for protecting and deprotecting with these groups are found in Greene and Wuts on pages 318 and 327.
- Some of the compounds described herein contain one or more asymmetric centers and may thus give rise to enantiomers, diastereomers, and other stereoisometric forms which may be defined in terms of absolute stereochemistry as (R)— or (S)—, or as (D)- or (L)- for amino acids.
- the present invention is meant to include all such possible diastereomers as well as their racemic and optically pure forms.
- Optically active (R)- and (S)- or (D)- and (L)-isomers may be prepared using chiral synthons or chiral reagents, or optically resolved using conventional techniques.
- Compounds of the invention which incorporate chiral diamines may be resolved into pairs of enantiomers by known techniques. Where pure enantiomers of starting materials are not commercially available, they may be obtained by classic resolution, which may employ, for example, fractional crystallization of diastereomeric salts. Compounds of the invention may have more than one chiral center, for example wherein reductive amination of a homochiral intermediate leads to a mixture of diastereomers. Racemic intermediates and compounds of the invention may also be resolved by chromatographic separation, such as for example, HPLC using a column loaded with a homochiral support, to yield pure isomeric compounds.
- any carbon-carbon double bond appearing herein is selected for convenience only and is not intended to designate a particular configuration; thus a carbon-carbon double bond depicted arbitrarily herein as trans may be cis, trans, or a mixture of the two in any proportion.
- the compounds of the present invention have demonstrated utility as selective inhibitors of inappropriate p38 kinase activity, and in particular, isoforms p38 ⁇ and p38 ⁇ .
- compounds of the present invention have utility in the treatment of conditions associated with inappropriate p38 kinase activity.
- Such conditions include diseases in which cytokine levels are modulated as a consequence of intracellular signaling via p38, and in particular, diseases that are associated with an overproduction of such cytokines as Il-1, Il-4, IL-8, and in particular, TNF- ⁇ .
- compounds of the present invention are useful in the treatment and prevention of p-38 mediated conditions including, but not limited to, inflammatory diseases, autoimmune diseases, destructive bone disorders, proliferative disorders, angiogenic disorders, infectious diseases, neurodegenerative diseases, viral diseases, allergies, myocardial ischemia, reperfusion/ischemia in stroke, heart attacks, organ hypoxia, vascular hyperplasia, cardiac hypertrophy, thrombin-induced platelet aggregation, and conditions associated with prostaglandin endoperoxidase synthase-2.
- Inflammatory diseases which may be treated or prevented include, but are not limited to, acute pancreatitis, chronic pancreatitis, asthma, allergies and adult respiratory distress syndrome.
- Autoimmune diseases which may be treated or prevented include, but are not limited to, glomerulonephritis, rheumatoid arthritis, systemic lupus erythematosis, scleroderma, chronic thyroiditis, Grave ⁇ s disease, autoimmune gastritis, diabetes, autoimmune hemolytic anemia, autoimmune neutropenia, thrombocytopenia, atopic dermatitis, chronic active hepatitis, myasthenia gravis, multiple sclerosis, inflammatory bowel disease, ulcerative colitis, Crohn's disease, psoriasis, or graft vs. host disease.
- Destructive bone disorders which may be treated or prevented include, but are not limited to, osteoporosis, osteoarthritis and multiple myeloma-related bone disorder.
- Proliferative diseases which may be treated or prevented include, but are not limited to, acute myelogenous leukemia, chronic myelogenous leukemia, metastatic melanoma, Kaposi's sarcoma, and multiple myeloma.
- Infectious diseases which may be treated or prevented include, but are not limited to, sepsis, septic shock, and Shigellosis.
- Neurodegenerative diseases which may be treated or prevented by the compounds of this invention include, but are not limited to, Alzheimer's disease, Parkinson's disease, cerebral ischemias or neurodegenerative disease caused by traumatic injury.
- Angiogenic disorders which may be treated or prevented include solid tumors, ocular neovasculization, infantile haemangiomas.
- Viral diseases which may be treated or prevented include, but are not limited to, acute hepatitis infection (including hepatitis A, hepatitis B and hepatitis C), HIV infection and CMV retinitis.
- p38 inhibitors of this invention also exhibit inhibition of the expression of inducible pro-inflammatory proteins such as prostaglandin endoperoxide synthase-2 (PGHS-2), also referred to as cyclooxygenase-2 (COX-2).
- PGHS-2 prostaglandin endoperoxide synthase-2
- COX-2 cyclooxygenase-2
- additional p38 mediated conditions include edema, analgesia, fever and pain, such as neuromuscular pain, headache, pain caused by cancer, dental pain and arthritis pain.
- compounds of the present invention have utility in the treatment and prevention of diseases associated with cytokine production.
- compounds of the present invention are useful in the treatment and prevention of:
- Il-1 mediated diseases such as, for example, rheumatoid arthritis, osteoarthritis, stroke, endotoxemia and/or toxic shock syndrome, inflammatory reaction induced by endotoxin, inflammatory bowel disease, tuberculosis, atherosclerosis, muscle degeneration, cachexia, psoriatic arthritis, Reiter's syndrome, gout, traumatic arthritis, rubella arthritis, acute synovitis, diabetes, pancreatic ⁇ -cell disease and Alzheimer's disease;
- rheumatoid arthritis such as, for example, rheumatoid arthritis, osteoarthritis, stroke, endotoxemia and/or toxic shock syndrome, inflammatory reaction induced by endotoxin, inflammatory bowel disease, tuberculosis, atherosclerosis, muscle degeneration, cachexia, psoriatic arthritis, Reiter's syndrome, gout, traumatic arthritis, rubella arthritis, acute synovitis, diabetes, pancreatic ⁇ -cell disease and Alzheimer's
- IL-8 mediated diseases or conditions such as, for example, those characterized by massive neutrophil infiltration, such as psoriasis, inflammatory bowel disease, asthma, cardiac and renal reperfusion injury, adult respiratory distress syndrome, thrombosis and glomerulonephritis; and
- TNF-mediated diseases or conditions such as rheumatoid arthritis, rheumatoid spondylitis, osteoarthritis, gouty arthritis and other arthritic conditions, sepsis, septic shock syndrome, adult respiratory distress syndrome, cerebral malaria, chronic pulmonary inflammatory disease, silicosis, pulmonary sarcoisosis, bone resorption disease, reperfusion injury, graft vs.
- viral infections such as HIV, CMV, influenza and herpes
- veterinary viral infections such as lentivirus infections, including, but not limited to equine infectious anemia virus; or retro virus infections, including feline immunodeficiency virus, bovine immunodeficiency virus, or canine immunodeficiency virus.
- the compounds of formula I including a pharmaceutically acceptable salt or hydrate thereof may be administered by any suitable route as described previously to treat the above mentioned diseases and conditions.
- the method of administration will, of course, vary depending upon the type of disease being treated.
- the amount of active compound administered will also vary according to the method of administration and the disease being treated.
- An effective amount will be within the dosage range of about 0.1 to about 100 mg/kg, preferably about 0.2 to about 50 mg/kg, in a single or multiple doses administered at appropriate intervals throughout the day.
- IC 50 values concentration required to inhibit 50% of specific binding
- Preferred compounds have an IC 50 below 1 ⁇ M.
- cDNAs of human p38 ⁇ , ⁇ and ⁇ isozymes were cloned by PCR. These cDNAs were subcloned in the pGEX expression vector (Pharmacia). GST-p38 fusion protein was expressed in E. Coli and purified from bacterial pellets by affinity chromatography using glutathione agarose. p38 fusion protein was activated by incubating with constitutively active MKK6. Active p38 was separated from MKK6 by affinity chromatography. Constitutively active MKK6 was generated according to Raingeaud et al. [ Mol. Cell. Biol., 1247-1255 (1996)].
- PBMCs Peripheral blood mononuclear cells
- assay medium RPMI medium containing 10% fetal bovine serum
- 50 ⁇ l of cell suspension was incubated with 50 ⁇ l of test compound (4 ⁇ concentration in assay medium containing 0.2% DMSO) in 96 well-tissue culture plates for 5 minutes at room temperature.
- 100 ⁇ l of LPS (200 ng/ml stock) was then added to the cell suspension and the plate was incubated for 6 hours at 37° C.
- TNF ⁇ concentration in the medium was quantified using a standard ELISA kit (Pharmingen-San Diego, Calif.). Concentrations of TNF ⁇ and IC50 values for test compounds (concentration of compound that inhibited LPS-stimulated TNF ⁇ production by 50%) were calculated by linear regression analysis.
- Human monocytic THP-1 cells were maintained in RPMI 1640 medium supplemented with 10% fetal bovine serum. Cells (40,000 cells in 80 ⁇ l) were added to wells of 96-well flat-bottomed plates. Tested compounds (10 ⁇ l) or vehicle (3% DMSO) were added to wells. Subsequently, LPS (Sigma, #L7261; 10 ⁇ l/well) was added to the cells for a final concentration of 1 ⁇ g/mL. Plates were incubated overnight at 37° C. and 5% CO 2 . Supernatant (50 ⁇ l/well) was harvested for an ELISA assay.
- TNF was captured by an anti-human TNF antibody (R&D, #MAB610) which was pre-absorbed in high binding EIA plates (Costar, #3590). Captured TNF was recognized by a biotinlated anti-human TNF polyclonal antibody (R&D, #BAF210). Streptavidin conjugated with peroxidase was added to each well, and the activity of peroxidase was quantitated by a peroxide substrate kit (Pierce, #34062 and #34006).
- the assays were performed in V-bottomed 96-well plates.
- the final assay volume was 60 ⁇ l prepared from three 20 ⁇ l additions of enzyme, substrates (MBP and ATP) and test compounds in assay buffer (50 mM Tris pH 7.5, 10 mM MgCl 2 , 50 mM NaCl and 1 mM DTT).
- Bacterially expressed, activated p38 was pre-incubated with test compounds for 10 min. prior to initiation of reaction with substrates. The reaction was incubated at 25° C. for 45 min. and terminated by adding 5 ⁇ l of 0.5 M EDTA to each sample.
- the reaction mixture was aspirated onto a pre-wet filtermat using a Skatron Micro96 Cell Harvester (Skatron, Inc.), then wash with PBS.
- the filtermat was then dried in a microwave oven for 1 min., treated with MeltilLex A scintillation wax (Wallac), and counted on a Microbeta scintillation counter Model 1450 (Wallac). Inhibition data were analyzed by nonlinear least-squares regression using Prizm (GraphPad Software).
- the final concentration of reagents in the assays are ATP, 1 ⁇ M; [ ⁇ - 33 P]ATP, 3 nM; MBP (Sigma, # M1891), 2 ⁇ g/well; p38, 10 nM; and DMSO, 0.3%.
- compounds of Formula I wherein V is —NR 5 —; one or two of W, X and Y are N; and each of Z and R 11 are attached to the core pyrimidine or pyridine by —N— or —O—, may be prepared from trihalopyrimidine by sequential reactions with three different amines (1, 2, 3), or two different amines (1, 2) and an alcohol, and subsequent introduction of an additional substituent on the pyrimidine core.
- An alternative method of preparation may start from dihalocyano-methylsulfanyl-pyrimidine (Scheme 2).
- one of the amines will be an aniline and another will be a diamine suitably protected on its distal N.
- the amines themselves, the sequence of the three substitutions, as well as the position of the nitrile may be varied, and are not limited by the particular example shown in Scheme 1 or Scheme 2.
- Amine 1 corresponds to —N(R 5 )(R 6 ); Amine 2 corresponds to -Z; and Amine 3 corresponds to —R 11 and such designations are used interchangeably in the description below.
Landscapes
- Health & Medical Sciences (AREA)
- Chemical & Material Sciences (AREA)
- Organic Chemistry (AREA)
- Life Sciences & Earth Sciences (AREA)
- Pharmacology & Pharmacy (AREA)
- General Health & Medical Sciences (AREA)
- Veterinary Medicine (AREA)
- Chemical Kinetics & Catalysis (AREA)
- General Chemical & Material Sciences (AREA)
- Medicinal Chemistry (AREA)
- Nuclear Medicine, Radiotherapy & Molecular Imaging (AREA)
- Public Health (AREA)
- Animal Behavior & Ethology (AREA)
- Bioinformatics & Cheminformatics (AREA)
- Engineering & Computer Science (AREA)
- Immunology (AREA)
- Physical Education & Sports Medicine (AREA)
- Rheumatology (AREA)
- Neurology (AREA)
- Biomedical Technology (AREA)
- Diabetes (AREA)
- Neurosurgery (AREA)
- Orthopedic Medicine & Surgery (AREA)
- Cardiology (AREA)
- Oncology (AREA)
- Hematology (AREA)
- Virology (AREA)
- Heart & Thoracic Surgery (AREA)
- Dermatology (AREA)
- Communicable Diseases (AREA)
- Pain & Pain Management (AREA)
- Urology & Nephrology (AREA)
- Hospice & Palliative Care (AREA)
- Emergency Medicine (AREA)
- Tropical Medicine & Parasitology (AREA)
- Endocrinology (AREA)
- Transplantation (AREA)
- Pulmonology (AREA)
- Ophthalmology & Optometry (AREA)
- AIDS & HIV (AREA)
Abstract
N-heterocyclic compounds that block cytokine production via inhibition of p38 kinase are disclosed. In one embodiment, compounds of the present invention are represented by Formula (I):
Methods of production, pharmaceutical compositions and methods of treating conditions associated with inappropriate p38 kinase activity or TNF-α expression utilizing compounds of the present invention are also disclosed.
Description
This application is a continuation of Ser. No. 10/183,179, filed Jun. 26, 2002, now abandoned, which claims priority to provisional Application Ser. No. 60/301,020, filed Jun. 26, 2001, both of which are incorporated herein by reference in their entirety.
This invention relates to N-heterocyclic compounds that are effective in blocking cytokine production, and in particular the expression of TNF-alpha (TNF-α), via inhibition of p38 kinase. Compounds of the present invention are useful in the treatment of inflammatory diseases such as, for example, rheumatoid arthritis.
Overproduction of cytokines such as IL-1 and TNF-α is implicated in a wide variety of inflammatory diseases, including rheumatoid arthritis (RA), psoriasis, multiple sclerosis, inflammatory bowel disease, endotoxin shock, osteoporosis, Alzheimer's disease and congestive heart failure, among others [Henry et al., Drugs Fut., 24:1345-1354 (1999); Salituro et al., Curr. Med. Chem., 6:807-823 (1999)]. There is convincing evidence in human patients that protein antagonists of cytokines, such as, for example, monoclonal antibody to TNF-α (Enbrel) [Rankin et al., Br. J. Rheumatol., 34:334-342 (1995)], soluble TNF-α receptor-Fc fusion protein (Etanercept) [Moreland et al., Ann. Intern. Med., 130:478-486 (1999)] and or IL-1 receptor antagonist [Bresnihan et al., Arthritis Rheum., 41:2196-2204 (1998)], can provide effective treatment for chronic inflammatory diseases. As none of the current treatments for inflammatory diseases provide complete relief of symptoms, and as most current treatments are associated with various drawbacks such as side effects, improved methods for treating inflammatory diseases are desirable.
TNF-α is a protein whose synthesis occurs in many cell types in response to an external stimulus, such as, for example, a mitogen, an infectious organism, or trauma. Signaling from the cell surface to the nucleus proceeds via several intracellular mediators including kinases that catalyze phosphorylation of proteins downstream in the signaling cascade. Important mediators for the production of TNF-α cytokine are the mitogen-activated protein (MAP) kinases, and in particular, p38 kinase.
p38 Kinases are activated in response to various stress stimuli, including, but not limited to, proinflammatory cytokines, endotoxin, ultraviolet light, and osmotic shock. Activation of p38 requires dual phosphorylation by upstream MAP kinase kinases (MKK3 and MKK6) on threonine and tyrosine within a Thr-Gly-Tyr motif, characteristic of p38 isozymes.
Four iso-forms of p38 have been described. The α and β forms are expressed in inflammatory cells and are thought to be key mediators of TNF-α production. Inhibition of the enzymes p38α and β in cells results in reduced levels of expression of TNF-α, and such inhibitors are effective in animal models of inflammatory disease.
Molecular cloning of human p38α identified two isozymes, which are the splice variant product of a single gene. Three additional gene products have subsequently been identified, p38β, p38γ, and p38δ. p38 kinases phosphorylate and activate the transcription factors, ATF-2, MAX, CHOP, and C/ERPb, suggesting a role of p38 kinases in gene regulation. In addition, p38 kinases phosphorylate other protein kinases, such as MAPK activated protein kinase-2/3 (MAPKAP-K2/3, or MK2/3), and MAP-kinase-interacting kinase 1/2 (MNK1/2). Recently, activation of MK2 has been shown to be essential for LPS-induced TNF-α expression [Kotlyarov et al., Nature Cell Biol., 1:94-97 (1999)]. Mice lacking MK2 exhibit a 90% reduction in the production of TNF-α and are resistant to shock induced by LPS. The reduction in TNF-α amounts is due not to decreased production of the TNF-α mRNA, but rather to diminished production of the TNF-α protein, suggesting that MK2 regulates biosynthesis of TNF-α at a post-transcriptional level.
Ample evidence indicates that the p38 pathway serves an important role in inflammatory process mediated by IL-1 and TNF-α.
Small molecule inhibitors of p38 are expected to have several advantages over protein inhibitors of TNF-α or IL-1. p38 inhibitors not only block the production of TNF-α and IL-1, but also directly interfere with many of their secondary biological effects. In addition, small molecule inhibitors are unlikely to induce immune reaction in patients, and are believed active following oral administration.
The present invention provides novel compounds that are potent and selective inhibitors of p38α and β, and as such, are also potent inhibitors of TNF-α expression in human cells. Compounds of the present invention are useful in the treatment of p38- and TNF-α expression-mediated inflammatory and other disorders, including, but not limited to, bone resorption, graft vs. host reaction, atherosclerosis, arthritis, osteoarthritis, rheumatoid arthritis, gout, psoriasis, topical inflammatory disease states, adult respiratory distress syndrome, asthma, chronic pulmonary inflammatory disease, cardiac reperfusion injury, renal reperfusion injury, thrombus, glomerulonephritis, Chron's disease, ulcerative colitis, inflammatory bowel disease, multiple sclerosis, endotoxin shock, osteoporosis, Alzheimer's disease, congestive heart failure and cachexia.
The compounds of the present invention are effective as inhibitors of inappropriate p38 activity, especially iso forms α and β, and in turn, of cytokine production, and in particular, of cellular TNF-alpha (TNF-α) expression. Accordingly, compounds of the invention are useful for the inhibition, prevention and suppression of various pathologies associated with such activity, such as, for example, inflammation, asthma, arthritis, atherosclerosis, multiple sclerosis, psoriasis, autoimmune diseases, Alzeheimers disease and congestive heart failure, among others.
In one embodiment, the principles of the present invention provide a compound, including isomers, enantiomers, diastereomers, tautomers, pharmaceutically acceptable salts, prodrugs and solvates thereof, represented by Formula (I):
wherein:
one or two of W, Y and X are ═N—;
one of W, Y and X is selected from ═C—CN, ═C—F, ═C—NO2, ═C—Br, ═C—NH2, ═C—NHC(O)CH3 and ═C—Cl;
the remaining W, Y or X is ═CH—;
V is —NR5—;
Z is halogen or —N(R1)(R2);
R1 and R2 are the same or different and are selected from hydrogen, alkyl, substituted alkyl, aryl, substituted aryl, cycloalkyl, substituted cycloalkyl, heterocyclyl or substituted heterocyclyl;
R5 is hydrogen or alkyl;
R6 is
R7 is hydrogen, alkyl, substituted alkyl, alkoxy, or halogen;
R8 is hydrogen, alkyl, alkyloxy or cyano;
R9 is —C(O)R10 or unsubstituted or substituted heterocyclyl;
R10 is —N(R31)(R32);
R31 and R32 are the same or different and are selected from hydrogen, alkyl, substituted alkyl, alkoxy, aryl, substituted aryl, cycloalkyl, substituted cycloalkyl, heterocyclyl or substituted heterocyclyl;
R11 is hydrogen, halogen, O—R35 or —N(R12)(R13);
R12 is hydrogen, alkyl, or substituted alkyl;
R13 is —(CH2)mR14;
—N(R12)(R13) taken together may form a heterocyclyl or substituted heterocyclyl;
m is 0, 1, 2 or 3;
R14 is hydrogen, alkyl, substituted alkyl, —C(O)N(R31)(R32), —N(R33)C(O)R34, aryl, substituted aryl, cycloalkyl, substituted cycloalkyl, heterocyclyl, substituted heterocyclyl, heteroaryl, substituted heteroaryl or
R15 is hydrogen, alkyl or substituted alkyl;
R16 is hydrogen or alkyl; or R33 is hydrogen, alkyl, or substituted alkyl;
R34 is alkyl, substituted alkyl, aryl or substituted aryl;
R35 is hydrogen or -(lower alkyl)-R36;
R36 is N(R37)(R38);
R37 is hydrogen, alkyl, or substituted alkyl;
R38 is -(substituted alkyl)-R14; and
N(R37)(R38) taken together may form a heterocyclyl or substituted heterocyclyl.
Preferred compounds of this invention are those of Formula (I) including a pharmaceutically acceptable salt thereof
wherein:
one or two of W, Y and X are ═N—;
one of W, Y and X is selected from ═C—CN, ═C—F, ═C—NO2, ═C—Br, ═C—NH2, ═C—NHC(O)CH3 and ═C—Cl;
the remaining W, Y or X is ═CH—;
V is —NH—;
Z is —N(R1)(R2);
R1 and R2 are the same or different and are selected from hydrogen, alkyl or substituted alkyl wherein alkyl is of 1 to 8 carbons;
R6 is
R7 is hydrogen, alkyl of 1 to 4 carbons, alkoxy of 1 to 4 carbons, or halogen;
R8 is hydrogen;
R9 is —C(O)R10 or unsubstituted or substituted heterocyclyl;
R10 is —NH2 or unsubstituted or substituted —NH-alkyl, —NH-alkoxy, —NH-heterocyclyl, —NH-phenyl, or —NH—CH2-phenyl wherein alkyl and alkoxy are of 1 to 6 carbons;
R11 is hydrogen, halogen, O—R35 or —N(R12)(R13), wherein N(R12)(R13) taken together may form a monocyclic heterocyclyl or substituted heterocyclyl of 5 to 7 atoms containing 1, 2, or 3 additional nitrogen atoms or wherein
R12 is hydrogen;
R13 is alkyl of 1 to 4 carbons or
R15 and R16 are independently selected from hydrogen and methyl;
R35 is hydrogen or -(lower alkyl)-R36;
R36 is N(R37)(R38);
R37 is hydrogen, alkyl, or substituted alkyl;
R38 is -(substituted alkyl)-R14; and
N(R37)(R38) taken together may form a heterocyclyl or substituted heterocyclyl.
The principles of the present invention also provide methods of inhibiting TNF-α expression in a mammal, wherein the methods comprise administering to the mammal an effective amount of a compound represented by Formula (I), or a prodrug or salt thereof. As used herein, inhibiting TNF-α expression is intended to include inhibiting, suppressing and preventing conditions associated with inappropriate TNF-α expression, including, but not limited to, inflammation, asthma, arthritis, atherosclerosis, multiple sclerosis, psoriasis, autoimmune diseases, Alzheimer's disease and congestive heart failure.
The principles of the present invention further provide methods of treating p38 kinase and TNF-α mediated disorders in a mammal, the methods comprising
administering to a mammal in need of such treatment, an effective amount of a compound represented by Formula (I), or a prodrug or salt thereof. As used herein, a p38 kinase mediated disorder means a disorder associated with inappropriate p38 kinase activity; a TNF-α mediated disorder means a disorder associated with inappropriate TNF-α expression. Such disorders include, but are not limited to, inflammation, asthma, arthritis, atherosclerosis, multiple sclerosis, psoriasis, autoimmune diseases, Alzheimer□s disease and congestive heart failure.
Accordingly, the compounds of the invention, as well as prodrugs or salts thereof, may be used in the manufacture of a pharmaceutical composition or medicament for the prophylactic or therapeutic treatment of disease states in mammals. The compounds of the present invention may be administered as pharmaceutical compositions as a monotherapy, or in combination with, for example, other anti-inflammatory, e.g. a steroid or NSAID (non-steroidal anti-inflammatory drug) and/or immunosuppressive agents. Such combination therapies can involve the administration of the various pharmaceuticals as a single dosage form or as multiple dosage forms administered simultaneously or sequentially.
Any suitable route of administration may be employed for providing a patient with an effective amount of a compound of the present invention. Suitable routes of administration may include, for example, oral, rectal, nasal, buccal, parenteral (such as, intravenous, intrathecal, subcutaneous, intramuscular, intrasternal, intrahepatic, intralesional, intracranial, intra-articular, and intra-synovial), transdermal (such as, for example, patches), and the like. Due to their ease of administration, oral dosage forms, such as, for example, tablets, troches, dispersions, suspensions, solutions, capsules, soft gelatin capsules, and the like, may be preferred. Administration may also be by controlled or sustained release means and delivery devices. Methods for the preparation of such dosage forms are well known in the art.
Pharmaceutical compositions incorporating compounds of the present invention may include excipients, a pharmaceutically acceptable carrier, in addition to other therapeutic ingredients. Excipients such as starches, sugars, microcrystalline cellulose, diluents, lubricants, binders, coloring agents, flavoring agents, granulating agents, disintegrating agents, and the like may be appropriate depending upon the route of administration. Because of their ease of administration, tablets and capsules represent the most advantageous oral dosage unit forms. If desired, tablets may be coated by standard aqueous or nonaqueous techniques.
The compounds of the present invention may be used in the form of pharmaceutically acceptable salts derived from inorganic or organic bases, and hydrates thereof. Included among such base salts are ammonium salts, alkali metal salts, such as sodium and potassium salts, alkaline earth metal salts, such as calcium and magnesium salts, salts with organic bases, such as dicyclohexylamine salts, N-methyl-D-glucamine, and salts with amino acids such as arginine, lysine, and so forth.
[1] Thus, in a first embodiment, the present invention provides a novel compound of Formula (I) including isomers, enantiomers, diastereomers, tautomers, pharmaceutically acceptable salts, prodrugs and solvates thereof, comprising:
wherein:
one or two of W, Y and X are ═N—;
one of W, Y and X is selected from ═C—CN, ═C—F, ═C—NO2, ═C—Br, ═C—NH2, ═C—NHC(O)CH3 and ═C—Cl;
the remaining W, Y or X is ═CH—;
V is —NR5—;
Z is halogen or —N(R1)(R2);
R1 and R2 are the same or different and are selected from hydrogen, alkyl, substituted alkyl, aryl, substituted aryl, cycloalkyl, substituted cycloalkyl, heterocyclyl or substituted heterocyclyl;
R5 is hydrogen or alkyl;
R6 is
R7 is hydrogen, alkyl, substituted alkyl, alkoxy, or halogen;
R8 is hydrogen, alkyl, alkyloxy or cyano;
R9 is —C(O)R10 or unsubstituted or substituted heterocyclyl;
R10 is —N(R31)(R32);
R31 and R32 are the same or different and are selected from hydrogen, alkyl, substituted alkyl, alkoxy, aryl, substituted aryl, cycloalkyl, substituted cycloalkyl, heterocyclyl or substituted heterocyclyl;
R11 is hydrogen, halogen, O—R35 or —N(R12)(R13);
R12 is hydrogen, alkyl, or substituted alkyl;
R13 is —(CH2)mR14;
—N(R12)(R13) taken together may form a heterocyclyl or substituted heterocyclyl;
m is 0, 1, 2 or 3;
R14 is hydrogen, alkyl, substituted alkyl, —C(O)N(R31)(R32), —N(R33)C(O)R34, aryl, substituted aryl, cycloalkyl, substituted cycloalkyl, heterocyclyl, substituted heterocyclyl, heteroaryl, substituted heteroaryl or
R15 is hydrogen, alkyl or substituted alkyl;
R16 is hydrogen or alkyl; or
R33 is hydrogen, alkyl, or substituted alkyl;
R34 is alkyl, substituted alkyl, aryl or substituted aryl;
R35 is hydrogen or -(lower alkyl)-R36;
R36 is N(R37)(R38);
R37 is hydrogen, alkyl, or substituted alkyl;
R38 is -(substituted alkyl)-R14; and
N(R37)(R38) taken together may form a heterocyclyl or substituted heterocyclyl.
[2] In a preferred embodiment, the present invention provides a compound of Formula (I) including isomers, enantiomers, diastereomers, tautomers, pharmaceutically acceptable salts, prodrugs and solvates thereof,
wherein:
one or two of W, Y and X are ═N—;
one of W, Y and X is selected from ═C—CN, ═C—F, ═C—NO2, ═C—Br, ═C—NH2, ═C—NHC(O)CH3 and ═C—Cl;
the remaining W, Y or X is ═CH—;
V is —NH—;
Z is —N(R1)(R2);
R1 and R2 are the same or different and are selected from hydrogen, alkyl or substituted alkyl wherein alkyl is of 1 to 8 carbons;
R6 is
R7 is hydrogen, alkyl of 1 to 4 carbons, alkoxy of 1 to 4 carbons, or halogen;
R8 is hydrogen;
R9 is —C(O)R10 or unsubstituted or substituted heterocyclyl;
R10 is —NH2 or unsubstituted or substituted —NH-alkyl, —NH-alkoxy, —NH-heterocyclyl, —NH-phenyl, or —NH—CH2-phenyl wherein alkyl and alkoxy are of 1 to 6 carbons;
R11 is hydrogen, halogen, O—R35 or —N(R12)(R13), wherein N(R12)(R13) taken together may form a monocyclic heterocyclyl or substituted heterocyclyl of 5 to 7 atoms containing 1, 2, or 3 additional nitrogen atoms or wherein
R12 is hydrogen;
R13 is alkyl of 1 to 4 carbons or
R15 and R16 are independently selected from hydrogen and methyl;
R35 is hydrogen or -(lower alkyl)-R36;
R36 is N(R37)(R38);
R37 is hydrogen, alkyl, or substituted alkyl;
R38 is -(substituted alkyl)-R14; and
N(R37)(R38) taken together may form a heterocyclyl or substituted heterocyclyl.
[3] In a more preferred embodiment, the present invention provides a compound of Formula (I) including isomers, enantiomers, diastereomers, tautomers, pharmaceutically acceptable salts, prodrugs and solvates thereof,
wherein:
one or two of W, Y and X are ═N—;
one of W, Y and X is selected from ═C—CN, ═C—F, ═C—NO2, ═C—Br, ═C—NH2, ═C—NHC(O)CH3 and ═C—Cl;
the remaining W, Y or X is ═CH—;
V is —NH—;
Z is —N(R1)(R2);
R1 and R2 are the same or different and are selected from hydrogen or alkyl of 1 to 8 carbons;
R6 is
R7 is hydrogen, methyl, methoxy, Cl, Br, or F;
R8 is hydrogen;
R9 is —C(O)R10 or unsubstituted or substituted heterocyclyl;
R10 is —NH2, or unsubstituted or substituted —NH-alkyl, —NH-alkoxy, —NH-phenyl, or —NH—CH2-phenyl wherein alkyl and alkoxy are of 1 to 6 carbons; and
R11 is hydrogen, halogen, O—R35 or —N(R12)(R13) wherein N(R12)(R13) taken together form a monocyclic heterocyclyl or substituted heterocyclyl of 5 to 7 atoms containing 1, 2, or 3 additional nitrogen atoms.
[4] In another preferred embodiment, the present invention provides a compound of Formula (I) including isomers, enantiomers, diastereomers, tautomers, pharmaceutically acceptable salts, prodrugs and solvates thereof,
wherein:
one of W, Y and X is ═N—;
one of W, Y and X is selected from ═C—CN, ═C—F, ═C—NO2, ═C—Br, ═C—NH2, ═C—NHC(O)CH3 and ═C—Cl;
the remaining W, Y or X is ═CH—;
V is —NH—;
Z is —N(R1)(R2);
R1 and R2 are the same or different and are selected from hydrogen or alkyl of 1 to 8 carbons;
R6 is
R7 is hydrogen, methyl, methoxy, Cl, Br, or F;
R8 is hydrogen;
R9 is —C(O)R10 or unsubstituted or substituted heterocyclyl;
R10 is —NH2, or unsubstituted or substituted —NH-alkyl, —NH-alkoxy, —NH-phenyl, or —NH—CH2-phenyl wherein alkyl and alkoxy are of 1 to 6 carbons;
R11 is hydrogen, halogen, —O—R35 or —N(R12)(R13) wherein N(R12)(R13) taken together form a monocyclic heterocyclyl or substituted heterocyclyl of 5 to 7 atoms containing 1, 2, or 3 additional nitrogen atoms; and
R15 and R16 are independently selected from hydrogen and methyl.
[5] In another more preferred embodiment, the present invention provides a compound of Formula (I) including isomers, enantiomers, diastereomers, tautomers, pharmaceutically acceptable salts, prodrugs and solvates thereof,
wherein:
R10 is —NH2, unsubstituted or substituted —NH—CH3, —NH—C2H5, —NH—OCH3, or —NH—OC2H5.
[6] In another more preferred embodiment, the present invention provides a compound of Formula (I) including isomers, enantiomers, diastereomers, tautomers, pharmaceutically acceptable salts, prodrugs and solvates thereof,
wherein:
R9 is unsubstituted or substituted triazole, thiazole, oxadiazole or imidazole.
[7] In another more preferred embodiment, the present invention provides a compound of Formula (I) including isomers, enantiomers, diastereomers, tautomers, pharmaceutically acceptable salts, prodrugs and solvates thereof,
wherein:
R11 is hydrogen, halogen, —O-(substituted alkyl), —NH— (substituted alkyl) or
[8] In yet another preferred embodiment, the present invention provides a compound of Formula (I) including isomers, enantiomers, diastereomers, tautomers, pharmaceutically acceptable salts, prodrugs and solvates thereof,
wherein:
R11 is hydrogen, halogen, —O-(substituted alkyl), —NH-(substituted alkyl) or
In yet another preferred embodiment, the present invention provides a compound of Formula (I) including isomers, enantiomers, diastereomers, tautomers, pharmaceutically acceptable salts, prodrugs and solvates thereof,
wherein:
two of W, Y and X are ═N—;
the remaining W, Y or X is selected from ═C—CN, ═C—F, ═C—NO2, ═C—Br, ═C—NH2, ═C—NHC(O)CH3 and ═C—Cl;
V is —NH—;
Z is —N(R1)(R2);
R1 and R2 are the same or different and are selected from hydrogen or alkyl of 1 to 8 carbons;
R6 is
R7 is hydrogen, methyl, methoxy, Cl, Br, or F;
R8 is hydrogen;
R9 is —C(O)R10 or unsubstituted or substituted heterocyclyl;
R10 is —NH2, or unsubstituted or substituted —NH-alkyl, —NH-alkoxy, —NH-phenyl, or —NH—CH2-phenyl wherein alkyl and alkoxy are of 1 to 6 carbons;
R11 is hydrogen, halogen, —O—R35 or —N(R12)(R13), wherein N(R12)(R13) taken together may form a monocyclic heterocyclyl or substituted heterocyclyl of 5 to 7 atoms containing 1, 2, or 3 additional nitrogen atoms; and
R15 and R16 are independently selected from hydrogen and methyl.
[10] In yet another more preferred embodiment, the present invention provides a compound of Formula (I) including isomers, enantiomers, diastereomers, tautomers, pharmaceutically acceptable salts, prodrugs and solvates thereof,
wherein:
R10 is —NH2, unsubstituted or substituted —NH—CH3, —NH—C2H5, —NH—OCH3, or —NH—OC2H5.
[11] In yet another more preferred embodiment, the present invention provides a compound of Formula (I) including isomers, enantiomers, diastereomers, tautomers, pharmaceutically acceptable salts, prodrugs and solvates thereof,
wherein:
R9 is unsubstituted or substituted triazole, thiazole, oxadiazole or imidazole.
[12] In yet another more preferred embodiment, the present invention provides a compound of Formula (I) including isomers, enantiomers, diastereomers, tautomers, pharmaceutically acceptable salts, prodrugs and solvates thereof,
wherein:
R11 is hydrogen, halogen, —O-(substituted alkyl), —NH-(substituted alkyl) or
[13] In yet another more preferred embodiment, the present invention provides a compound of Formula (I) including isomers, enantiomers, diastereomers, tautomers, pharmaceutically acceptable salts, prodrugs and solvates thereof,
wherein:
R11 is hydrogen, halogen, —O-(substituted alkyl), —NH-(substituted alkyl) or
[14] In a second preferred embodiment, the present invention provides a pharmaceutical composition comprising as an active ingredient, a compound of the invention or a prodrug or salt thereof, and a pharmaceutically acceptable carrier.
[15] In a preferred embodiment, the present invention provides a pharmaceutical composition further comprising one or more additional active ingredients.
[16] In a more preferred embodiment, the present invention provides a pharmaceutical composition wherein the additional active ingredient is an anti-inflammatory compound or an immunosuppressive agent.
[17] In a more preferred embodiment, the present invention provides a pharmaceutical composition wherein the additional active ingredient is chosen from a steroid and an NSAID.
[18] In a third embodiment, the present invention provides a method of inhibiting TNF-α expression in a mammal comprising administering to the mammal an effective amount of the pharmaceutical composition of the invention.
[19] In a preferred embodiment, the present invention provides a method of treating TNF-α mediated disorder comprising administering to a mammal in need of such treatment, an effective amount of a pharmaceutical composition of the invention.
[20] In a more preferred embodiment, the present invention provides a method of treating TNF-α mediated disorder, wherein the TNF-α mediated disorder is an inflammatory disorder.
[21] In a more preferred embodiment, the present invention provides a method of treating TNF-α mediated disorder, wherein the TNF-α mediated disorder is chosen from bone resorption, graft vs. host reaction, atherosclerosis, arthritis, osteoarthritis, rheumatoid arthritis, gout, psoriasis, topical inflammatory disease states, adult respiratory distress syndrome, asthma, chronic pulmonary inflammatory disease, cardiac reperfusion injury, renal reperfusion injury, thrombus, glomerulonephritis, Chron's disease, ulcerative colitis, inflammatory bowel disease, multiple sclerosis, endotoxin shock, osteoporosis, Alzheimer's disease, congestive heart failure and cachexia.
[22] In a more preferred embodiment, the present invention provides a method of treating TNF-α mediated disorder wherein the pharmaceutical composition of the invention is administered with one or more additional anti-inflammatory or immunosuppressive agents as a single dose form or as separate dosage forms.
[23] In an even more preferred embodiment, the present invention provides a method of treating a condition associated with TNF-α expression in a mammal comprising administering to a mammal in need of such treatment, an effective amount of a pharmaceutical composition of the invention.
[24] In an even more preferred embodiment, the present invention provides a method of treating a condition associated with TNF-α expression in a mammal wherein the condition associated with TNF-α expression is an inflammatory disorder.
[25] In a more preferred embodiment, the present invention provides a method of treating a condition associated with TNF-α expression, wherein the condition associated with TNF-α expression is chosen from bone resorption, graft vs. host reaction, atherosclerosis, arthritis, osteoarthritis, rheumatoid arthritis, gout, psoriasis, topical inflammatory disease states, adult respiratory distress syndrome, asthma, chronic pulmonary inflammatory disease, cardiac reperfusion injury, renal reperfusion injury, thrombus, glomerulonephritis, Crohn's disease, ulcerative colitis, inflammatory bowel disease, multiple sclerosis, endotoxin shock, osteoporosis, Alzheimer's disease, congestive heart failure and cachexia.
[26] In a more preferred embodiment, the present invention provides a method of treating a condition associated with TNF-α expression wherein the pharmaceutical composition of the invention is administered with one or more additional anti-inflammatory or immunosuppressive agents as a single dose form or as separate dosage forms.
[27] In another more preferred embodiment, the present invention provides a method of treating a condition associated with p38 kinase activity in a mammal comprising administering to a mammal in need of such treatment, an effective amount of a pharmaceutical composition of the invention.
[28] In another more preferred embodiment, the present invention provides a method of treating a condition associated with p38 kinase activity in a mammal wherein the condition associated with p38 activity is an inflammatory disorder.
[29] In a more preferred embodiment, the present invention provides a method of treating a condition associated with p38 kinase activity is chosen from bone resorption, graft vs. host reaction, atherosclerosis, arthritis, osteoarthritis, rheumatoid arthritis, gout, psoriasis, topical inflammatory disease states, adult respiratory distress syndrome, asthma, chronic pulmonary inflammatory disease, cardiac reperfusion injury, renal reperfusion injury, thrombus, glomerulonephritis, Chron's disease, ulcerative colitis, inflammatory bowel disease, multiple sclerosis, endotoxin shock, osteoporosis, Alzheimer's disease, congestive heart failure and cachexia.
[30] In a more preferred embodiment, the present invention provides a method of treating a condition associated with p38 activity wherein the pharmaceutical composition of the invention is administered with one or more additional anti-inflammatory or immunosuppressive agents as a single dose form or as separate dosage forms.
[31] In a fourth embodiment, the present invention provides a compound including isomers, enantiomers, diastereomers, tautomers, pharmaceutically acceptable salts, prodrugs and solvates selected from:
The following terms and abbreviations retain the indicated meaning throughout this disclosure.
- ATP=adenosine triphosphate
- cDNA=complementary DNA
- DCE=dichloroethylene
- DCM=dichloromethane=methylene chloride=CH2Cl2
- DIC=diisopropylcarbodiimide
- DIEA=N,N-diisopropylethylamine
- DMF=N,N-dimethylformamide
- DMSO=dimethyl sulfoxide
- DTT=dithiothreitol
- EDTA=ethylenediaminetetraacetic acid
- EIA=enzyme immunoassay
- ELISA=enzyme-linked immunosorbent assay
- Fmoc=9-fluorenylmethoxycarbonyl
- GST=glutathione S-transferase
- HOBt=1-hydroxybenzotriazole
- LPS=lipopolysaccharide
- MBP=myelin basic protein
- MES=2-(N-morpholino)ethanesulfonic acid
- mRNA=messenger RNA
- PCR=polymerase chain reaction
- Pr2NEt=dipropylethylamine
- i-Pr2NEt=diisopropylethylamine
- RPMI=Roswell Park Memorial Institute
- TBS=t-butyldimethylsilyl
- TFA=trifluoroacetic acid
- THF=tetrahydrofuran
“Alkyl” is intended to include linear or branched hydrocarbon structures and combinations thereof of 1 to 20 carbons. “Lower alkyl” means alkyl groups of from 1 to about 10, preferably from 1 to about 8, and more preferably, from 1 to about 6 carbon atoms. Examples of such radicals include methyl, ethyl, n-propyl, isopropyl, n-butyl, isobutyl, s-butyl, t-butyl, pentyl, iso-amyl, hexyl, octyl and the like.
“Aryl” means an aromatic hydrocarbon radical of 6 to about 16 carbon atoms, preferably of 6 to about 12 carbon atoms, and more preferably of 6 to about 10 carbon atoms. Examples of aryl groups are phenyl, which is preferred, 1-naphthyl and 2-naphthyl.
“Cycloalkyl” refers to saturated hydrocarbon ring structures of from 3 to 12 carbon atoms, and preferably from 3 to 6 carbon atoms. Examples include cyclopropyl, cyclobutyl, cyclopentyl, cyclohexyl, norbornyl, adamantyl, and the like. “Lower cycloalkyl” refers to cycloalkyl of 3 to 6 carbons.
“Heterocyclyl” refers to saturated, partially saturated or unsaturated monocyclic structures of from 3 to 8 atoms, preferably 5 or 6 atoms, and bicyclic structures of 9 or 10 atoms containing one or more carbon atoms and from 1 to 4 heteroatoms chosen from O, N, and S. The point of attachment of the heterocyclyl structure is at an available carbon or nitrogen atom. Examples include: imidazole, pyridine, indole, thiophene, benzopyranone, thiazole, furan, benzimidazole, quinoline, isoquinoline, quinoxaline, pyrimidine, pyrazine, tetrazole, pyrazole, pyrrolyl, pyridinyl, pyrazolyl, triazolyl, pyrimidinyl, pyridazinyl, oxazolyl, thiazolyl, imidazolyl, indolyl, thiophenyl, furanyl, tetrazolyl, 2-pyrrolinyl, 3-pyrrolinyl, pyrrolindinyl, 1,3-dioxolanyl, imidazolinyl, imidazolidinyl, pyrazolinyl, pyrazolidinyl, isoxazolyl, isothiazolyl, 1,2,3-oxadiazolyl, 1,2,3-triazolyl, 1,2,4-triazolyl, 1,3,4-thiadiazolyl, 2H-pyranyl, 4H-pyranyl, piperidinyl, 1,4-dithianyl, thiomorpholinyl, pyrazinyl, piperazinyl, 1,3,5-triazinyl, 1,2,5-trithianyl, benzo(b)thiophenyl, benzimidazolyl, quinolinyl, and the like.
“Alkoxy” means a straight, branched or cyclic hydrocarbon configuration and combinations thereof, including from 1 to 20 carbon atoms, preferably from 1 to 8 carbon atoms, more preferably from 1 to about 4 carbon atoms, and an oxygen atom at the point of attachment. Suitable alkoxy groups include methoxy, ethoxy, n-propoxy, isopropoxy, n-butoxy, iso-butoxy, s-butoxy, t-butoxy, cyclopropyloxy, cyclohexyloxy, and the like. “Lower alkoxy” refers to alkoxy groups having from 1 to 4 carbon atoms. Similarly, “alkylthio” refers to such groups having a sulfur atom at the point of attachment.
“Alkenyl” refers to an unsaturated acyclic hydrocarbon radical in so much as it contains at least one double bond. “Lower alkenyl” refers to such radicals containing from about 2 to about 10 carbon atoms, preferably from about 2 to about 8 carbon atoms and more preferably 2 to about 6 carbon atoms. Examples of suitable alkenyl radicals include propenyl, buten-1-yl, isobutenyl, penten-1-yl, 2-methylbuten-1-yl, 3-methylbuten-1-yl, hexen-1-yl, hepten-1-yl, and octen-1-yl, and the like.
“Alkynyl” refers to an unsaturated acyclic hydrocarbon radical containing at least one triple bond. Examples include ethynyl, propynyl, and the like.
“Substituted alkyl” means an alkyl wherein one or more hydrogens, preferably one, two, or three hydrogens, attached to an aliphotic carbon are replaced with a substituent such as —N(R31)(R32), alkoxy, alkylthio, halogen, cyano, carboxyl, hydroxyl, —SO2-alkyl, —CO2-alkyl, —C(O)-alkyl, nitro, cycloalkyl, substituted cycloalkyl, aryl, substituted aryl, heterocyclyl, substituted heterocyclyl, —C(O)—N(R31)(R32), or —NH—C(O)-alkyl. Examples of such substituent groups include methoxy, ethoxy, propoxy, amino, methylamino, dimethylamino, phenyl naphthyl, chlorine, fluorine, and the like.
“Substituted cycloalkyl” means a cycloalkyl wherein one or more hydrogens, preferably one, two or three hydrogens, attached to a ring carbon are replaced with a substituent such as alkyl, substituted alkyl, —N(R31)(R32), alkoxy, alkylthio, aryl, substituted aryl, halogen, cyano, carboxyl, hydroxyl, nitro, —SO2-alkyl, —CO2-alkyl, —C(O)-alkyl, —C(O)—N(R31)R32), or —NH—C(O)-alkyl. Examples of such groups include methyl, isopropyl, methoxy, ethoxy, propoxy, amino, methylamino, dimethylamino, phenyl, chlorine, fluorine and the like. Also included within this definition are cycloalkyl rings having a fused aryl, preferably phenyl, or cycloalkyl such as
“Substituted aryl” means an aryl wherein one or more hydrogens, preferably one, two or three hydrogens, attached to an aromatic carbon are replaced with a substituent such as alkyl, substituted alkyl, —N(R31)(R32), alkoxy, alkylthio, aryl, substituted aryl, halogen, cyano, nitro, carboxyl, hydroxyl, —SO2-alkyl, —CO2-alkyl, —C(O)-alkyl, —C(O)—N(R31)(R32), or —NH—C(O)-alkyl. Examples of such substituents include methyl, isopropyl, methoxy, ethoxy, propoxy, amino, methylamino, dimethylamino, phenyl, chlorine, fluorine, —CO2CH3, —C(O)—NH2, and the like.
“Substituted heterocyclyl” means a heterocyclyl substituted at one or more available carbon or nitrogen atoms, preferably at one or two carbon and/or nitrogen atoms, with a substituent such as alkyl, substituted alkyl, —N(R31)(R32), alkoxy, alkylthio, aryl, substituted aryl, halogen, cyano, nitro, oxo, carboxyl, hydroxyl, —SO2-alkyl, —CO2-alkyl, —C(O)-alkyl, —C(O)—N(R31)(R32), or —NH—C(O)-alkyl. Examples of such groups include methyl isopropyl, methoxy, ethoxy, propoxy, amino, methylamino, dimethylamino, phenyl, chlorine, fluorine and the like.
“Halogen” is intended to include for example, F, Cl, Br and I.
The term “prodrug” refers to a chemical compound that is converted to an active agent by metabolic processes in vivo. [See, e.g., N. Boder and J. J. Kaminski, Ann. Rep. Med. Chem. 22:303 (1987) and H. Bundgarrd, Adv. Drug Delivery Rev., 3:39 (1989)]. With regard to the present invention, a prodrug of a compound of Formula I is intended to mean any compound that is converted to a compound of Formula I by metabolic processes in vivo. The use of prodrugs of compounds of Formula I in any of the methods described herein is contemplated and is intended to be within the scope of the invention.
Terminology related to “protected,” “protecting” and/or “deprotecting” functionalities is used throughout this application. Such terminology is well understood by persons of skill in the art and is used in the context of processes which involve sequential treatment with a series of reagents. In this context, a protecting group refers to a group which is used to mask a functionality during a process step in which it would otherwise react, but in which reaction is undesirable. The protecting group prevents reaction at that step, but may be subsequently removed to expose the original functionality. The removal or “deprotection” occurs after the completion of the reaction or reactions in which the functionality would interfere. Thus, when a sequence of reagents is specified, as it is in the processes of the invention, the person of ordinary skill can readily envision those groups that would be suitable as “protecting groups” for the functionalities involved.
In the case of the present invention, the typical functionalities that must be protected are amines. Suitable groups for that purpose are discussed in standard textbooks in the field of chemistry, such as Protective Groups in Organic Synthesis by T. W. Greene [John Wiley & Sons, New York, 1991], which is incorporated herein by reference. Particular attention is drawn to the chapter entitled “Protection for the Amino Group” (pages 309-405). Preferred protecting groups include BOC and Fmoc. Exemplary methods for protecting and deprotecting with these groups are found in Greene and Wuts on pages 318 and 327.
Some of the compounds described herein contain one or more asymmetric centers and may thus give rise to enantiomers, diastereomers, and other stereoisometric forms which may be defined in terms of absolute stereochemistry as (R)— or (S)—, or as (D)- or (L)- for amino acids. The present invention is meant to include all such possible diastereomers as well as their racemic and optically pure forms. Optically active (R)- and (S)- or (D)- and (L)-isomers may be prepared using chiral synthons or chiral reagents, or optically resolved using conventional techniques. When the compounds described herein contain olefinic double bonds or other centers of geometric asymmetry, and unless specified otherwise, it is intended to include both (E)- and (Z)-geometric isomers. Likewise, all tautomeric forms are intended to be included.
Compounds of the invention which incorporate chiral diamines may be resolved into pairs of enantiomers by known techniques. Where pure enantiomers of starting materials are not commercially available, they may be obtained by classic resolution, which may employ, for example, fractional crystallization of diastereomeric salts. Compounds of the invention may have more than one chiral center, for example wherein reductive amination of a homochiral intermediate leads to a mixture of diastereomers. Racemic intermediates and compounds of the invention may also be resolved by chromatographic separation, such as for example, HPLC using a column loaded with a homochiral support, to yield pure isomeric compounds.
The configuration of any carbon-carbon double bond appearing herein is selected for convenience only and is not intended to designate a particular configuration; thus a carbon-carbon double bond depicted arbitrarily herein as trans may be cis, trans, or a mixture of the two in any proportion.
In view of the above definitions, other chemical terms used throughout this application can be easily understood by those of skill in the art. Terms may be used alone or in any combination thereof. The preferred and more preferred chain lengths of the radicals apply to all such combinations.
The compounds of the present invention have demonstrated utility as selective inhibitors of inappropriate p38 kinase activity, and in particular, isoforms p38α and p38β. As such, compounds of the present invention have utility in the treatment of conditions associated with inappropriate p38 kinase activity. Such conditions include diseases in which cytokine levels are modulated as a consequence of intracellular signaling via p38, and in particular, diseases that are associated with an overproduction of such cytokines as Il-1, Il-4, IL-8, and in particular, TNF-α.
As inhibitors of p-38 kinase activity, compounds of the present invention are useful in the treatment and prevention of p-38 mediated conditions including, but not limited to, inflammatory diseases, autoimmune diseases, destructive bone disorders, proliferative disorders, angiogenic disorders, infectious diseases, neurodegenerative diseases, viral diseases, allergies, myocardial ischemia, reperfusion/ischemia in stroke, heart attacks, organ hypoxia, vascular hyperplasia, cardiac hypertrophy, thrombin-induced platelet aggregation, and conditions associated with prostaglandin endoperoxidase synthase-2.
Inflammatory diseases which may be treated or prevented include, but are not limited to, acute pancreatitis, chronic pancreatitis, asthma, allergies and adult respiratory distress syndrome.
Autoimmune diseases which may be treated or prevented include, but are not limited to, glomerulonephritis, rheumatoid arthritis, systemic lupus erythematosis, scleroderma, chronic thyroiditis, Grave□s disease, autoimmune gastritis, diabetes, autoimmune hemolytic anemia, autoimmune neutropenia, thrombocytopenia, atopic dermatitis, chronic active hepatitis, myasthenia gravis, multiple sclerosis, inflammatory bowel disease, ulcerative colitis, Crohn's disease, psoriasis, or graft vs. host disease.
Destructive bone disorders which may be treated or prevented include, but are not limited to, osteoporosis, osteoarthritis and multiple myeloma-related bone disorder.
Proliferative diseases which may be treated or prevented include, but are not limited to, acute myelogenous leukemia, chronic myelogenous leukemia, metastatic melanoma, Kaposi's sarcoma, and multiple myeloma.
Infectious diseases which may be treated or prevented include, but are not limited to, sepsis, septic shock, and Shigellosis.
Neurodegenerative diseases which may be treated or prevented by the compounds of this invention include, but are not limited to, Alzheimer's disease, Parkinson's disease, cerebral ischemias or neurodegenerative disease caused by traumatic injury.
Angiogenic disorders which may be treated or prevented include solid tumors, ocular neovasculization, infantile haemangiomas.
Viral diseases which may be treated or prevented include, but are not limited to, acute hepatitis infection (including hepatitis A, hepatitis B and hepatitis C), HIV infection and CMV retinitis.
In addition, p38 inhibitors of this invention also exhibit inhibition of the expression of inducible pro-inflammatory proteins such as prostaglandin endoperoxide synthase-2 (PGHS-2), also referred to as cyclooxygenase-2 (COX-2). Accordingly, additional p38 mediated conditions include edema, analgesia, fever and pain, such as neuromuscular pain, headache, pain caused by cancer, dental pain and arthritis pain.
As a result of their p38 inhibitory activity, compounds of the present invention have utility in the treatment and prevention of diseases associated with cytokine production. For example, compounds of the present invention are useful in the treatment and prevention of:
Il-1 mediated diseases such as, for example, rheumatoid arthritis, osteoarthritis, stroke, endotoxemia and/or toxic shock syndrome, inflammatory reaction induced by endotoxin, inflammatory bowel disease, tuberculosis, atherosclerosis, muscle degeneration, cachexia, psoriatic arthritis, Reiter's syndrome, gout, traumatic arthritis, rubella arthritis, acute synovitis, diabetes, pancreatic β-cell disease and Alzheimer's disease;
IL-8 mediated diseases or conditions such as, for example, those characterized by massive neutrophil infiltration, such as psoriasis, inflammatory bowel disease, asthma, cardiac and renal reperfusion injury, adult respiratory distress syndrome, thrombosis and glomerulonephritis; and
TNF-mediated diseases or conditions such as rheumatoid arthritis, rheumatoid spondylitis, osteoarthritis, gouty arthritis and other arthritic conditions, sepsis, septic shock syndrome, adult respiratory distress syndrome, cerebral malaria, chronic pulmonary inflammatory disease, silicosis, pulmonary sarcoisosis, bone resorption disease, reperfusion injury, graft vs. host reaction, allograft rejections, fever and myalgias due to infection, cachexia secondary to infection, AIDS, ARC or malignancy, meloid formation, scar tissue formation, Crohn's disease, ulcerative colitis, pyresis, viral infections, such as HIV, CMV, influenza and herpes; and veterinary viral infections, such as lentivirus infections, including, but not limited to equine infectious anemia virus; or retro virus infections, including feline immunodeficiency virus, bovine immunodeficiency virus, or canine immunodeficiency virus.
The compounds of formula I including a pharmaceutically acceptable salt or hydrate thereof may be administered by any suitable route as described previously to treat the above mentioned diseases and conditions. The method of administration will, of course, vary depending upon the type of disease being treated. The amount of active compound administered will also vary according to the method of administration and the disease being treated. An effective amount will be within the dosage range of about 0.1 to about 100 mg/kg, preferably about 0.2 to about 50 mg/kg, in a single or multiple doses administered at appropriate intervals throughout the day.
The IC50 values (concentration required to inhibit 50% of specific binding) of compounds of the present invention for inhibition of p38 activity are below 5 μM. Preferred compounds have an IC50 below 1 μM.
cDNAs of human p38α, β and γ isozymes were cloned by PCR. These cDNAs were subcloned in the pGEX expression vector (Pharmacia). GST-p38 fusion protein was expressed in E. Coli and purified from bacterial pellets by affinity chromatography using glutathione agarose. p38 fusion protein was activated by incubating with constitutively active MKK6. Active p38 was separated from MKK6 by affinity chromatography. Constitutively active MKK6 was generated according to Raingeaud et al. [Mol. Cell. Biol., 1247-1255 (1996)].
TNF-α Production by LPS-Stimulated PBMCS
Heparinized human whole blood was obtained from healthy volunteers. Peripheral blood mononuclear cells (PBMCs) were purified from human whole blood by Ficoll-Hypaque density gradient centrifugation and resuspended at a concentration of 5×106/ml in assay medium (RPMI medium containing 10% fetal bovine serum). 50 μl of cell suspension was incubated with 50 μl of test compound (4× concentration in assay medium containing 0.2% DMSO) in 96 well-tissue culture plates for 5 minutes at room temperature. 100 μl of LPS (200 ng/ml stock) was then added to the cell suspension and the plate was incubated for 6 hours at 37° C. Following incubation, the culture medium was collected and stored at −20° C. TNFα concentration in the medium was quantified using a standard ELISA kit (Pharmingen-San Diego, Calif.). Concentrations of TNFα and IC50 values for test compounds (concentration of compound that inhibited LPS-stimulated TNFα production by 50%) were calculated by linear regression analysis.
Human monocytic THP-1 cells were maintained in RPMI 1640 medium supplemented with 10% fetal bovine serum. Cells (40,000 cells in 80 μl) were added to wells of 96-well flat-bottomed plates. Tested compounds (10 μl) or vehicle (3% DMSO) were added to wells. Subsequently, LPS (Sigma, #L7261; 10 μl/well) was added to the cells for a final concentration of 1 μg/mL. Plates were incubated overnight at 37° C. and 5% CO2. Supernatant (50 μl/well) was harvested for an ELISA assay. TNF was captured by an anti-human TNF antibody (R&D, #MAB610) which was pre-absorbed in high binding EIA plates (Costar, #3590). Captured TNF was recognized by a biotinlated anti-human TNF polyclonal antibody (R&D, #BAF210). Streptavidin conjugated with peroxidase was added to each well, and the activity of peroxidase was quantitated by a peroxide substrate kit (Pierce, #34062 and #34006).
The assays were performed in V-bottomed 96-well plates. The final assay volume was 60 μl prepared from three 20 μl additions of enzyme, substrates (MBP and ATP) and test compounds in assay buffer (50 mM Tris pH 7.5, 10 mM MgCl2, 50 mM NaCl and 1 mM DTT). Bacterially expressed, activated p38 was pre-incubated with test compounds for 10 min. prior to initiation of reaction with substrates. The reaction was incubated at 25° C. for 45 min. and terminated by adding 5 μl of 0.5 M EDTA to each sample. The reaction mixture was aspirated onto a pre-wet filtermat using a Skatron Micro96 Cell Harvester (Skatron, Inc.), then wash with PBS. The filtermat was then dried in a microwave oven for 1 min., treated with MeltilLex A scintillation wax (Wallac), and counted on a Microbeta scintillation counter Model 1450 (Wallac). Inhibition data were analyzed by nonlinear least-squares regression using Prizm (GraphPad Software). The final concentration of reagents in the assays are ATP, 1 μM; [γ-33P]ATP, 3 nM; MBP (Sigma, # M1891), 2 μg/well; p38, 10 nM; and DMSO, 0.3%.
General methods of synthesis for compounds of the present invention are illustrated by the following examples. Compounds of the invention may be prepared by standard techniques known in the art, involving both solution and solid phase chemistry. Starting materials are commercially available or may by readily prepared by one of skill in the art with known methods, or by methods disclosed herein. Specific embodiments described are presented by way of illustration only, and the invention is not limited thereto. Modifications and variations in any give material or process step will be readily apparent to one of skill in the art and all are to be included within the scope of the invention.
As illustrated in Scheme 1 and Scheme 2, compounds of Formula I wherein V is —NR5—; one or two of W, X and Y are N; and each of Z and R11 are attached to the core pyrimidine or pyridine by —N— or —O—, may be prepared from trihalopyrimidine by sequential reactions with three different amines (1, 2, 3), or two different amines (1, 2) and an alcohol, and subsequent introduction of an additional substituent on the pyrimidine core. An alternative method of preparation may start from dihalocyano-methylsulfanyl-pyrimidine (Scheme 2). Preferably, one of the amines will be an aniline and another will be a diamine suitably protected on its distal N. The person of skill will recognize that the amines themselves, the sequence of the three substitutions, as well as the position of the nitrile may be varied, and are not limited by the particular example shown in Scheme 1 or Scheme 2.
With respect to Formula I of the invention, Amine 1 corresponds to —N(R5)(R6); Amine 2 corresponds to -Z; and Amine 3 corresponds to —R11 and such designations are used interchangeably in the description below.
Preparation of Amines
Hydrogen chloride was bubbled through a solution of 3-nitro-p-tolunitrile (0.49 g, 3 mmol) in 40 mL of ethanol at room temp for 10 min. The solution was continued stirring at room temp for 60 min and the solvent was then evaporated under vacuum to dryness to give a white solid.
The intermediate so obtained was dissolved in 20 mL of ethanol, neutralized with sodium ethoxide solution and the resulting precipitate was removed by filtration. To the filtrate was added at room temp formic hydrazide (0.2 g, 3 mmol) and the solution was continued stirring at room temp for 2 h. After removal of volatiles in vacuo, the residue was dissolved in 30 mL of m-xylene and refluxed at 150° C. for 16 h. Removal of volatiles in vacuo and purification using flash chromatography afforded 0.26 g of the final product. (Yield: 43%). MS (m/z) calcd for C9H8N4O2 (MH+) 205.2, found, 205.1.
Coupling of Substituted Pyridines with Amines
A solution of 2,6-dichloro-3-cyano-5-fluoropyridine (1.0 g, 5.23 mmol), N-methyl-neopentylamine hydrochloride (830 mg, 6.0 mmol) and triethylamine (1.6 mL) in acetonitrile (20 mL) was stirred at room temp for 4 hours. Then volatiles were removed in vacuo and the residue was partitioned between ethyl acetate and water. The organic layer was dried (sodium sulfate) and concentrated in vacuo to afford the product (1.11 g, 83%). C12H15ClFN3 MS m/e=256 (M+H).
Preparation of Substituted Pyrimidines
To phosphoryl chloride (108 mL) chilled in ice bath was added dimethylformamide (35 mL). The mixture was allowed to stand at 20 degree for one hour, then 25 g of 2-methylsulfanyl-pyrimidine-4,6-diol was added slowly. After 30 minutes, the reaction mixture was heated to 100° C. for 6 hours. The reaction mixture was poured onto crushed ice and the precipitate was collected by filtration. The crude product was purified with flash chromatography to afford 11.13 g of 4,6-Dichloro-2-methylsulfanyl-pyrimidine-5-carbaldehyde (Yield=32%). 1H-NMR (300 MHz, CDCl3): δ 10.50 (s, 1H), 2.76 (s, 3H)
4,6-Dichloro-2-methylsulfanyl-pyrimidine-5-carbaldehyde (7.34 g, 33.09 mmol), hydroxylamine hydrochloride (2.31 g, 33.33 mmol), acetic acid (49.6 mL), and water (3.3 mL) were mixed, and heated to 60° C. for 2 hours. The reaction mixture was diluted with water and cooled under ice bath. The precipitate was collected and dried (Yield=6.41 g, 82%). MS (m/z): 238 (M+H).
4,6-Dichloro-2-methylsulfanyl-pyrimidine-5-carbaldehyde oxime (7.20 g, 30.38 mmol) was added to neat thionyl chloride (29.63 g, 245 mmol), then the mixture was heated to reflux for 4 hours. The reaction mixture was poured onto ice-water. The precipitate of 4,6-dichloro-2-methylsulfanyl-pyrimidine-5-carbonitrile was collected and dried (Yield=6.15 g, 92%). 1H-NMR (300 MHz, CDCl3) δ 2.75(s, 3H).
4,6-Dichloro-2-methylsulfanyl-pyrimidine-5-carbonitrile (2.19 g, 10 mmol), 3-amino-4,N-dimethyl-benzamide(1.64 g, 10 mmol), and DIEA(1.40 g, 18.8 mmol) were mixed in THF (20 mL). The resulting mixture was stirred at room temperature for overnight. The solvent was evaporated and the residue was partitioned between ethyl acetate and water. The organic layer was concentrated and the crude product was purified by flash chromatography to obtain 3-(6-chloro-5-cyano-2-methylsulfanyl-pyrimidin-4-ylamino)-4,N-dimethyl-benzamide (2.78 g, 80%). MS (m/z): 348 (M+H).
The following examples illustrate preferred embodiments of the present invention and do not limit the scope of the present invention, which is defined in the claims.
Compounds shown in Tables 1 and 2 have been synthesized according to the methods described herein and have been tested in accordance with the protocols described below. These compounds are provided by way of illustration only, and the invention is not intended to be limited thereto. Exemplary syntheses of some compounds are also provided.
| TABLE 1 | |||
| Ex | |||
| # | m/z | Rt | |
| 1 |
|
367 | 4.67 |
| 2 |
|
420,422 | |
| 3 |
|
433 | 3.09 |
| 4 |
|
405 | 2.58 |
| 5 |
|
499 | |
| 6 |
|
466 | 4.47 |
| 7 |
|
452 | 4.22 |
| 8 |
|
452 | 4.16 |
| 9 |
|
424 | 3.56 |
| 10 |
|
438 | 3.94 |
| 11 |
|
435 | |
| 12 |
|
486 | |
| 13 |
|
432 | |
| 14 |
|
466 | |
| 15 |
|
449 | |
| 16 |
|
466 | |
| 17 |
|
376 | |
| 18 |
|
475 | 7.3 |
| 19 |
|
405 | 6.2 |
| 20 |
|
335 | 4.6 |
| 21 |
|
445 | |
| 22 |
|
496 | 4.64 |
| 23 |
|
383 | 10.3 |
| 24 |
|
357 | |
| 25 |
|
399 | |
| 26 |
|
396 | |
| 27 |
|
500 | 11.0 |
| 28 |
|
366 | |
| 29 |
|
510 | 4.81 |
| 30 |
|
509 | 4.84 |
| 31 |
|
509 | 4.28 |
| 32 |
|
412 | 6.0 |
| 33 |
|
467 | |
| 34 |
|
||
| 35 |
|
405 | 5.96 |
| 36 |
|
480 | 4.67 |
| 37 |
|
496 | 5.06 |
| 38 |
|
494 | 4.83 |
| 39 |
|
468 | 4.69 |
| 40 |
|
396 | |
| 41 |
|
479 | |
| 42 |
|
495 | |
| 43 |
|
460 | 5.51 |
| 44 |
|
494 | 4.84 |
| 45 |
|
495 | 4.46 |
| 46 |
|
493 | 4.20 |
| 47 |
|
422 | |
| 48 |
|
399 | 7.5 |
| 49 |
|
406,409 | 5.7 |
| 50 |
|
353 | 6.0 |
| 51 |
|
367 | 6.4 |
| 52 |
|
413 | 7.7 |
| 53 |
|
367 | 4.53 |
| 54 |
|
389 | |
| 55 |
|
403 | |
| 56 |
|
387 | 6.9 |
| 57 |
|
373 | 7.0 |
| 58 |
|
451 | |
| 59 |
|
451 | |
| 60 |
|
451 | |
| 61 |
|
509 | 12.5 |
| 62 |
|
495 | 12.4 |
| TABLE 2 | |
| Example#70 |
|
| Example#71 |
|
| Example#72 |
|
| Example#73 |
|
| Example#74 |
|
| Example#75 |
|
| Example#76 |
|
| Example#77 |
|
| Example#78 |
|
A mixture of 2-chloro-6-[(2,2-dimethyl-propyl)-methyl-amino]-5-fluoro-nicotinonitrile (120 mg, 0.47 mmol), 3-amino-N-methoxy-4-methyl-benzamide (120 mg, 0.66 mmol) and potassium fluoride (30 mg, 0.51 mmol) in DMSO (1 mL) was heated to 150° C. overnight. The reaction mixture was allowed to cool down to room temp and then partitioned between water and ethyl acetate. The organic layer was dried (sodium sulfate) and concentrated under reduced pressure. The product (7.5 mg, 4%) was obtained after purification by silica gel chromatography with 30% EtOAc in hexane as eluent. C21H26FN5O2 MS m/e=400 (M+H).
A mixture of 2,6-dichloro-3-cyano-5-fluoropyridine (1.0 g, 5.23 mmol), neopentylamine (530 mg, 6.0 mmol) and triethylamine (1 mL) in acetonitrile (20 mL) was stirred at room temp for 4 h. After the solvent was removed under reduced pressure, the residue was partitioned between ethyl acetate and water. The organic layer was dried (sodium sulfate) and concentrated under reduced pressure to afford the product (1.11 g, 87%). C11H13ClFN3 MS m/e=242 (M+H).
A mixture of 2-chloro-6-[(2,2-dimethyl-propyl)-amino]-5-fluoro-nicotinonitrile (150 mg, 0.62 mmol), 3-amino-N-methoxy-4-methyl-benzamide (150 mg, 0.83 mmol) and potassium fluoride (30 mg, 0.51 mmol) in DMSO (1 mL) was heated to 150° C. overnight. The reaction mixture was allowed to cool to room temp and then partitioned between water and ethyl acetate. The organic layer was dried (sodium sulfate) and concentrated under reduced pressure. The product (2.5 mg, 1%) was obtained after purification by silica gel chromatography with 30% EtOAc in hexane as eluent. C20H24FN5O2 MS m/e=386 (M+H).
A mixture of 2,6-dichloro-5-fluoro-nicotinonitrile (830 mg, 4.34 mmol), 3-amino-N-methoxy-4-methyl-benzamide (576 mg, 3.2 mmol) and triethylamine (0.5 mL) in acetonitrile (10 mL) was heated to 70° C. overnight. Then the solvent was removed under reduced pressure and the reaction mixture was partitioned between water and ethyl acetate. The organic layer was dried (sodium sulfate) and concentrated under reduced pressure. The product (220 mg, 21%) was isolated after purification by silica gel chromatography. C15H12ClFN4O2 MS m/e=335 (M+H).
A mixture of 3-(6-chloro-5-cyano-3-fluoro-pyridin-2-ylamino)-N-methoxy-4-methyl-benzamide (52 mg, 0.15 mmol), neopentylamine (0.12 mL) and potassium fluoride (12 mg) in DMSO (1 mL) was heated to 150° C. overnight. The product (1.1 mg, 1.8%) was isolated after purification by HPLC. C20H24FN5O2 MS m/e=386 (M+H).
A mixture of 2,6-dichloro-5-fluoro-nicotinonitrile (830 mg, 4.34 mmol), 3-amino-N-methoxy-4-methyl-benzamide (576 mg, 3.2 mmol) and triethylamine (0.5 mL) in acetonitrile (10 mL) was heated to 70° C. overnight. Then the solvent was removed under reduced pressure and the reaction mixture was partitioned between water and ethyl acetate. The organic layer was dried (sodium sulfate) and concentrated under reduced pressure. The product (220 mg, 21%) was isolated after purification by silica gel chromatography. C15H12ClFN4O2 MS m/e=335 (M+H).
A mixture of 3-(6-chloro-5-cyano-3-fluoro-pyridin-2-ylamino)-N-methoxy-4-methyl-benzamide (55.6 mg, 0.166 mmol), N-methyl-neopentylamine (70 mg, 0.511 mmol), diisopropylethylamine (0.1 mL) and potassium fluoride (12 mg) in DMSO (1 mL) was heated to 150° C. overnight. The product (3.9 mg, 5.9%) was isolated after purification by HPLC. C21H26FN5O2 MS m/e=400 (M+H).
This compound was prepared according to procedure for the synthesis of 3-(6-Chloro-5-cyano-2-methylsulfanyl-pyrimidin-4-ylamino)-4,N-dimethyl-benzamide. MS (m/z): 413 (M+H).
To a solution of 3-{5-cyano-6-[(2,2-dimethyl-propyl)-methyl-amino]-2-methylsulfanyl-pyrimidin-4-ylamino}-4,N-dimethyl-benzamide (0.20 g, 0.48 mmol) in acetic acid (8 mL) was added a solution of potassium permagnate (87 mg, 0.55 mmol) in water (10 mL). The resulting mixture was heated to 50° C. for 10 minutes. The reaction mixture was then diluted with water (20 mL), and the product was extracted with ethyl acetate. The product was obtained (199 mg) after drying and removing the solvent. MS (m/z): 445 (M+H).
3-{5-Cyano-6-[(2,2-dimethyl-propyl)-methyl-amino]-2-methanesulfonyl-pyrimidin-4-ylamino}-4,N-dimethyl-benzamide (44 mg, 0.1 mmol) and methylamine (1 mL, 1M in THF) were mixed in p-dioxane (1 mL) in a sealed tube. The mixture was heated to 80° C. for overnight. The solvent was removed in vacuo, and the product (23 mg) was purified by the silica gel column chromatography. MS (m/z): 396 (M+H).
To a portion of 576 mg of 1-methylpiperidine-4-ol (576 mg; 5 mmol) is added 616 mg of potassium tert.-butoxide (5.5 mmol) followed by 4.0 mL of DMSO. After stirring this mixture at r.t. for 1 h a portion of 1.0 mL of this mixture is added at r.t. to 19 mg of 3-{5-cyano-6-[(2,2-dimethyl-propyl)-methyl-amino]-2-methanesulfonyl-pyrimidin-4-ylamino}-4,N-dimethyl-benzamide (0.043 mmol) on 0.2 mL of DMSO. The mixture was heated at 60° C. for 3 h. At r.t. 5 mL of ethyl acetate is added and the organic layer is washed with brine (1×4 mL). The organic layer is dried (MgSO4), volatiles are removed in vacuo and the product is purified via reversed phase prep. HPLC. (Yield: 18.6 mg; 0.026 mmol; 56%). MS (m/z): 480 (M+H).
3-[5-Cyano-6-[(2,2-dimethyl-propyl)-methyl-amino]-2-(4-methyl-[1,4]diazepan-1-yl)-pyrimidin-4-ylamino]-4,N-dimethyl-benzamide was synthesized similar to the synthesis of 3-{5-cyano-6-[(2,2-dimethyl-propyl)-methyl-amino]-2-methylamino-pyrimidin-4-ylamino}-4,N-dimethyl-benzamide. MS (m/z): 479 (M+H).
4,6-Dichloro-2-methylsulfanyl-pyrimidine-5-carbonitrile (2.19 g, 10 mmol), 3-amino-N-methoxy-4-methyl-benzamide (1.80 g, 10 mmol) and DIEA (1.9 mL) in THF (80 mL) were stirred at room temperature for overnight. The solvent was removed in vacuo and the product (3.33 g, 92%) was obtained after purification by silica gel column chromatography.
3-(6-Chloro-5-cyano-2-methylsulfanyl-pyrimidin-4-ylamino)-N-methoxy-4-methyl-benzamide (3.33 g, 9.2 mmol), N-methyl-neopentylamine hydrochloride (2.05 g, 15 mmol) and DIEA (3.87 g, 30 mmol) in THF (10 mL) were heated to 60° C. for overnight. The solvent was removed in vacuo and the product (1.75 g) was obtained after purification by silica gel column chromatography.
To a solution of 3-{5-Cyano-6-[(2,2-dimethyl-propyl)-methyl-amino]-2-methylsulfanyl-pyrimidin-4-ylamino}-N-methoxy-4-methyl-benzamide (0.10 g) in ethanol (5 mL) was added the solution of sodium periodate (0.2 g) in water (1 mL). The resulting solution was heated to 70° C. for overnight. The solvent was evaporated and the residue was partitioned between water and ethyl acetate. The organic layer was separated, and concentrated, and the residue was purified by silica gel column chromatography to afford the sulfoxide product (90 mg). MS (m/z): 445 (M+H).
3-{5-Cyano-6-[(2,2-dimethyl-propyl)-methyl-amino]-2-methanesulfinyl-pyrimidin-4-ylamino}-N-methoxy-4-methyl-benzamide (40 mg) and 1-methyl-homopiperazine (0.05 mL) in THF (0.5 mL) were heated in sealed tube at 75° C. for overnight. After the solvent was removed in vacuo, the residue was purified by silica gel column chromatography to afford the product (6.8 mg). MS (m/z): 495 (M+H).
3-{5-Cyano-6-[(2,2-dimethyl-propyl)-methyl-amino]-2-methanesulfinyl-pyrimidin-4-ylamino}-N-methoxy-4-methyl-benzamide (33 mg) and methylamine (2.5 mL, 2 M in THF) were heated in a sealed tube at 75° C. for overnight. After the solvent was removed in vacuo, the residue was purified by silica gel column chromatography to afford the product (7.3 mg). MS (m/z): 412 (M+H).
3-{5-Cyano-6-[(2,2-dimethyl-propyl)-methyl-amino]-2-methanesulfinyl-pyrimidin-4-ylamino}-N-methoxy-4-methyl-benzamide (33 mg) and 1-N-Boc-3-(R)-aminopyrrolidine (30 mg), DIEA (0.2 mL) and p-dioxane (2 mL) were heated in a sealed tube at 75° C. for overnight. After the removal of the solvent in vacuo, the product was purified by silica gel column chromatography and treated with TFA/DCM (1:1) in order to remove the Boc-group. The product was then converted to hydrochloride salt by treating it with hydrochloric acid (1 M in ether) (Yield: 20 mg). MS (m/z): 567 (M+H).
3-{6-[(2,2-Dimethyl-propyl)-methyl-amino]-5-nitro-pyrimidin-4-ylamino}-4,N-dimethyl-benzamide (0.31 g, 0.8 mmol)) was hydrogenated under 40 psi of hydrogen pressure in the presence of 10% Pd/C for 4 hours in Parr instrument. The catalyst was filtered off through celite and the filtrate was concentrated to afford the product (0.24 g, yield 84%). MS (m/z): 357 (M+H).
3-{5-Amino-6-[(2,2-dimethyl-propyl)-methyl-amino]-pyrimidin-4-ylamino}-4,N-dimethyl-benzamide (16 mg), acetyl chloride (3.5 mg), triethylamine (0.02 ml) were stirred in methylene chloride (0.2 mL) at room temperature for overnight. The product (2.8 mg) was purified by preparative thin layer chromatography. MS (m/z): 399 (M+H).
To a solution of 3-{6-[(2,2-dimethyl-propyl)-methyl-amino]-2-methylsulfanyl-pyrimidin-4-ylamino}-4,N-dimethyl-benzamide (0.10 g) in methylene chloride (2 mL), was added sat. aq. sodium bicarbonate (0.05 mL) and bromine (0.013 mL). The resulting mixture was stirred at room temperature for 30 minutes, and ethyl acetate (30 mL) and magnesium sulfate (1 g) was added. After filtration and concentration, the residue was purified by silica gel column chromatography to afford the product (61.9 mg). MS (m/z): 466 (M+H).
3-{6-[(2,2-Dimethyl-propyl)-methyl-amino]-2-methylamino-pyrimidin-4-ylamino}-4,N-dimethyl-benzamide (33 mg), aq. sat. sodium bicarbonate (0.05 mL) and bromine (14 mg) were stirred in methylene chloride (1 mL) at room temperature for 4 hours. The product (14 mg) was purified with preparative thin layer chromatography. MS (m/z): 449 (M+1).
3-[6-(2,2-Dimethyl-propylamino)-2-methylamino-pyrimidin-4-ylamino]-4,N-dimethyl-benzamide (35 mg), aq. sat. sodium bicarbonate (0.05 mL) and bromine (21 mg) were stirred in methylene chloride (1 mL) at room temperature for overnight. The product (4 mg) was purified with preparative thin layer chromatography. MS (m/z): 435 (M+H).
To a solution of 4,6-dichloropyrimidine (2.0 g, 13.4 mmol) in p-dioxane (50 mL), 3-amino-4,N-dimethyl-benzamide (3.0 g, 18.3 mmol) and DIEA (2.3 mL) were added. The resulting mixture was heated to reflux for 4 days. The solvent was removed in vacuo, the residue was taken in water and ethyl acetate, and the ethyl acetate layer was separated and concentrated. The product was purified by silica gel column chromatography using ethyl acetate:hexane (1:1) as eluent to afford the pale white solid product (1.6 g, yield 43%). MS (m/z): 277 (M+H).
To a solution of 3-(6-chloro-pyrimidin-4-ylamino)-4,N-dimethyl-benzamide (0.4 g, 1.44 mmol) in DMSO (3 mL), neopentylamine (0.4 mL, 3.39 mmol) was added. The resulting solution was heated at 110° C. for 4 days. The product was purified by silica gel column chromatography using ethyl acetate as eluent to afford the product (0.45 g, yield 99%). MS (m/z): 328 (M+H).
To a solution of 3-{6-[(2,2-dimethyl-propyl)-amino]-pyrimidin-4-ylamino}-4,N-dimethyl-benzamide (0.45 g, 1.37 mmol) in methylene chloride (10 mL), was added sat. aq. sodium bicarbonate (2 mL) and bromine (0.07 mL, 1.37 mmol). The resulting mixture was stirred at room temperature for 18 hours, and the reaction was then diluted with water (20 mL). The organic layer was separated and the aqueous layer was extracted with DCM (10 mL). The combined organic layer was dried (sodium sulfate), filtered and concentrated. The residue was purified by silica gel column chromatography using 2% methanol in DCM as eluent to afford the product (314 mg, yield 56%). MS (m/z): 406 (M+H).
To a solution of 4,6-dichloropyrimidine (2.0 g, 13.4 mmol) in p-dioxane (50 mL), 3-amino-4,N-dimethyl-benzamide (3.0 g, 18.3 mmol) and DIEA (2.3 mL) were added. The resulting mixture was heated to reflux for 4 days. The solvent was removed in vacuo, the residue was taken in water and ethyl acetate, and the ethyl acetate layer was separated and concentrated. The product was purified by silica gel column chromatography using ethyl acetate:hexane (1:1) as eluent to afford the pale white solid product (1.6 g, yield 43%). MS (m/z): 277 (M+H).
To a solution of 3-(6-chloro-pyrimidin-4-ylamino)-4,N-dimethyl-benzamide (0.4 g, 1.44 mmol) in DMSO (3 mL), N-methylneopentylamine hydrochloride (0.4 g, 2.9 mmol) and DIEA (0.5 mL, 2.9 mmol) were added. The resulting solution was heated at 110° C. for 4 days. The product was purified by silica gel column chromatography using ethyl acetate as eluent to afford the product (0.46 g, yield 99%). MS (m/z): 342 (M+H).
To a solution of 3-{6-[(2,2-dimethyl-propyl)-methyl-amino]-pyrimidin-4-ylamino}-4,N-dimethyl-benzamide (0.46 g, 1.34 mmol) in methylene chloride (10 mL), was added sat. aq. sodium bicarbonate (2 mL) and bromine (0.07 mL, 1.37 mmol). The resulting mixture was stirred at room temperature for 18 hours, and the reaction was then diluted with water (20 mL). The organic layer was separated and the aqueous layer was extracted with DCM (10 mL). The combined organic layer was dried (sodium sulfate), filtered and concentrated. The residue was purified by silica gel column chromatography using 2% methanol in DCM as eluent to afford the product (250 mg, yield 44%). MS (m/z): 420 (M+H).
A mixture of 3-(R)-[4-fluoro-6-(2-methyl-5-methylcarbamoyl-phenylamino)-pyrimidin-2-ylamino]-pyrrolidine-1-carboxylic acid tert-butyl ester (0.36 g, 0.81 mmol), N-methyl-neopentylamine hydrochloride (411 mg, 3 mmol) and DIEA(0.4 mL) in 1,4-dioxane (0.5 mL) was stirred at 90° C. overnight. After removing the solvent under reduced pressure the desired product (81 mg) was purified by silica gel chromatography. C28H43N7O3 MS m/e=526 (M+H).
To a mixture of 3-(R)-[4-[(2,2-dimethyl-propyl)-methyl-amino]-6-(2-methyl-5-methylcarbamoyl-phenylamino)-pyrimidin-2-ylamino]-pyrrolidine-1-carboxylic acid tert-butyl ester (70 mg, 0.0001 mmol) and sodium cyanide (0.044 g, 0.89 mmol) in sat. aq. sodium bicarbonate (1 mL) and methylene chloride (3 mL) at room temp was added bromine (0.045 mL, 0.87 mmol). The resulting mixture was stirred for 16 h at room temp, then diluted with water and extracted with methylene chloride (2×15 mL). The combined organic layer was dried (sodium sulfate), filtered and concentrated under reduced pressure. This product was then treated with a mixture of trifluoroacetic acid and methylene chloride (1:1 v/v, 1 mL). The resulting solution was stirred at room temp for 2 h, then the solvent was removed under reduced pressure and the product was purified by HPLC. C24H34N8O MS m/e=451 (M+H).
To a mixture of 3-(R)-[6-[(2,2-dimethyl-propyl)-methyl-amino]-2-(2-methyl-5-methylcarbamoyl-phenylamino)-pyrimidin-4-ylamino]-pyrrolidine-1-carboxylic acid tert-butyl ester (128 mg, 0.0002 mmol) and sodium cyanide (0.044 g, 0.89 mmol) in sat. aq. sodium bicarbonate (1 mL) and methylene chloride (3 mL) was added bromine (0.045 mL, 0.87 mmol). The resulting mixture was continued stirring at room temp for 16 h, then diluted with water and extracted with methylene chloride (2×15 mL). The combined organic layer was dried (sodium sulfate), filtered and concentrated under reduced pressure. This product was then treated with a mixture of trifluoroacetic acid and methylene chloride (1:1 v/v, 1 mL). The resulting solution was stirred at room temp for 2 h. The solvent was removed under reduced pressure, and the product was purified by HPLC. C24H34N8O MS m/e=451 (M+H).
To a stirred mixture of 3-[4-[(2,2-dimethyl-propyl)-methyl-amino]-6-(4-methyl-[1,4]diazepan-1-yl)-pyrimidin-2-ylamino]-N-methoxy-4-methyl-benzamide (140 mg, 0.29 mmol) and sodium cyanide (0.044 g, 0.89 mmol) in sat. aq. sodium-bicarbonate (1 mL) and methylene chloride (3 mL) was added bromine (0.045 mL, 0.87 mmol). The resulting mixture was stirred for 16 h at room temp, then diluted with water and extracted with methylene chloride (2×15 mL). The combined organic layer was dried (sodium sulfate), filtered and concentrated under reduced pressure, and the product was purified by HPLC. C26H38N8O2 MS m/e=495 (M+H). Deprotection was performed as described in Example 75.
To a stirred mixture of 3-[2-[(2,2-dimethyl-propyl)-methyl-amino]-6-(4-methyl-[1,4]diazepan-1-yl)-pyrimidin-4-ylamino]-N-ethoxy-4-methyl-benzamide (50 mg, 0.1 mmol) and sodium cyanide (0.044 g, 0.89 mmol) in sat. aq. sodium bicarbonate (1 mL) and methylene chloride (3 mL) was added at room temp bromine (0.045 mL, 0.87 mmol). The resulting mixture was continued stirring for 16 h at room temp. The reaction mixture was diluted with water and extracted with methylene chloride (2×15 mL). The combined organic layer was dried (sodium sulfate), filtered and concentrated under reduced pressure, and the product was purified by HPLC. C27H40N8O2 MS m/e=509 (M+H).
To a solution of 2-methyl-5-(4H-[1,2,4]triazol-3-yl)-phenylamine (174 mg, 1 mmol) in 4 mL of DMF was added 0.17 mL of diisopropylethylamine (1 mmol) and 4,6-dichloro-2-methylsulfanyl-pyrimidine-5-carbonitrile (219 mg, 1 mmol). The resulting solution was stirred at room temperature overnight, then partitioned between ethyl acetate and water. The organic layer was washed with water, brine and dried over MgSO4. Removal of volatiles in vacuo and purification by flash chromatography gave 78 mg of the product (Yield: 22%). MS (m/z) calcd for C15H12ClN7S (MH+), 358.1, found, 358.3.
To a solution of 4-chloro-2-methylsulfanyl-6-[2-methyl-5-(4H-[1,2,4]triazol-3-yl)-phenylamino]-pyrimidine-5-carbonitrile (71 mg, 0.2 mmol) in 2 mL of n-BuOH was added 137 mg of N-(2,2-dimethylpropyl)methyl amine HCl salt (1 mmol) and 0.17 mL of diisopropylethylamine (1 mmol) and the resulting solution was stirred at 60° C. for 24 h. Removal of volatiles in vacuo and purification by flash chromatography gave 68 mg of the product (Yield: 81%) MS (m/z) calcd for C21H26N8S (MH+), 423.2, found, 423.3.
To a solution of 4-[(2,2-dimethyl-propyl)-methyl-amino]-2-methylsulfanyl-6-[2-methyl-5-(4H-[1,2,4]triazol-3-yl)-phenylamino]-pyrimidine-5-carbonitrile (76 mg, 0.18 mmol) in 3 mL of acetic acid was added 0.5 mL water followed by 50 mg of potassium permanganate (0.36 mmol). This solution was stirred at 60° C. for 10 minutes, then diluted with water and extracted with ethyl acetate. The organic layer was washed with water, brine and dried over MgSO4. Removal of volatiles in vacuo gave 58 mg of the crude product which was used in the next step without further purification (Yield: 71%). MS (m/z) calcd for C21H26N8O2S (MH+), 455.2, found, 455.3.
A solution of 4-[(2,2-dimethyl-propyl)-methyl-amino]-2-methanesulfonyl-6-[2-methyl-5-(4H-[1,2,4]triazol-3-yl)-phenylamino]-pyrimidine-5-carbonitrile (10 mg, 0.022 mmol) and 1-tert-butoxycarbony-3(R)-amino-pyrrolidine (0.08 g, 0.4 mmol) in 2 mL of acetonitrile was heated with stirring at 80° C. for 18 h. Volatiles were the removed in vacuo and the product was purified by flash chromatography.
This purified product was then dissolved in 3 mL of a solution of 50% TFA in CH2Cl2 (v/v) and stirred at room temp for 30 min. Removal of volatiles in vacuo and purification via prep. HPLC gave the product as TFA salt. The purified product was then dissolved in 1N HCl (g) in diethyl ether and evaporated to give 0.8 mg of the final product. (Yield: 8%). MS (m/z) calcd for C24H32N10 (MH+), 461.3, found, 461.4.
To a solution of 4-[(2,2-dimethyl-propyl)-methyl-amino]-2-methylsulfanyl-6-[2-methyl-5-(4H-[1,2,4]triazol-3-yl)-phenylamino]-pyrimidine-5-carbonitrile (60 mg, 0.142 mmol) in 3 mL of 50% ethanol/water (v/v) was added 0.4 mL of 50% Raney-Ni in water solution. The solution was refluxed under argon for 18 hours. The solution was evaporated under vacuum. The resulting residue was purified by flash chromatography to afford 5.6 mg of the final product (Yield: 10%). MS (m/z) calcd for C20H24N8 (MH+), 377.2, found, 377.4.
To a solution of 0.38 mL of 5-Bromo-2,4-dichloror-pyrimidine (70 mg; 3.1 mmol) in 5 mL of THF at 0° C. is added dropwise a solution of 0.644 mL of N,N-diisopropylethylamine (478 mg; 3.7 mmol) and of 506 mg of 3-Amino-4,N-dimethyl-benzamide ((3.1 mmol) in 2 mL of THF. This solution is continued stirring at 0° C. for 1 h, then at 25° C. for 30 min. After removal of volatiles in vacuo the product was purified via silica gel chromatography (20% ethyl acetate in hexanes) to yield 304 mg of a white powder (0.85 mmol; 25% yield). MS (m/z): 355 (M+H).
A mixture of 51 mg of 3-(5-Bromo-2-chloro-pyrimidin-4-ylamino)-4,N-dimethyl-benzamide (0.143 mmol), 59 mg of (2,2-dimethylpropyl)-methylamine, hydrochloride (0.430 mmol) and 0.112 mL of N,N-diisopropylethylamine (83 mg; 0.645 mmol) is heated to 120° C. for 18 h, then allowed to cool to r.t. 5 ml of ethyl acetate are added and the organic layer is washed with brine (1×5 ml). The aqueous layer is extracted with ethyl acetate (3×5 ml) and the combined organic layers are dried (MgSO4). After removal of volatiles in vacuo the product was purified via silica gel chromatography (20% ethyl acetate in hexanes) to yield 41 mg of an colorless oil (0.097 mmol; yield: 68%). MS (m/z): 420 (M+H).
A solution of 22 mg of 3-(5-bromo-2-chloro-pyrimidin-4-ylamino)-4,N-dimethyl-benzamide (0.06 mmol) and 46 mg of 3-amino-pyrrolidine-1-carboxylic acid tert.-butyl ester (0.247 mmol) is heated at 120° C. in 0.3 mL DMSO for 3 d. After addition of 5 mL of ethyl acetate at r.t. the organic layer is washed with a sat. solution of NaHCO3 in water (3×4 mL). The organic layer is dried (MgSO4), volatiles are removed in vacuo and the product is purified via prep. HPLC. (Yield: 5.7 mg; 0.013 mmol; 22 MS (m/z): 444 (M+); 455 (M+Na).
The purified product is dissolved in 1 mL of MeOH and 3 mL of a 1N solution of HCl in diethyl ether is added. The resulting solution is stirred at r.t for 30 min, then volatiles are removed in vacuo and the product is purified via prep. HPLC. (Yield: 4.3 mg; 0.0002 mmol; 0.4%). MS (m/z): 405 (M+H).
To a portion of 576 mg of 1-methylpiperidine-4-ol (576 mg; 5 mmol) is added 616 mg of potassium tert.-butoxide (5.5 mmol) followed by 4.0 mL of DMSO. After stirring this mixture at r.t. for 1 h a portion of 1.0 mL of this mixture is added at r.t. to 21 mg of 3-(5-Bromo-2-chloro-pyrimidin-4-ylamino)-4,N-dimethyl-benzamide (0.06 mmol). The resulting mixture is heated at 120° C. for 18 h and then allowed to cool to r.t. A portion of 5 mL of ethyl acetate is added and the resulting solution is washed with a sat. solution of NaHCO3 in water (3×4 mL). The organic layer is dried (MgSO4), volatiles are removed in vacuo and the product is purified via prep. HPLC. (Yield: 5.7 mg; 0.013 mmol; 22%). MS (m/z): 444 (M+); 455 (M+Na).
A mixture of 15 mg of 3-{5-Bromo-2-[2,2-dimethyl-propyl)-methyl-amino]-pyrimidin-4-ylamino}-4,N-dimethyl-benzamide (0.036 mmol) and 100 mg of CuCN (1.12 mmol) in 1.0 mL of 1-methyl-2-pyrrolidinone is heated to 140° C. for 18 h. The mixture was allowed to cool to r.t. and 2.0 mL of MeOH were added. After removing the precipitate by filtration volatiles were evaporated and the product was purified by prep. HPCL. Yield: 1.7 mg (0.005 mmol; 13%). MS (m/z): 367 (M+H).
To a solution of 1.0 g of 2,4-dichloropyrimidine-5-carbonyl chloride (4.73 mmol) in 5 ml of THF at 0° C. is added dropwise a 0.5 M solution of NH3 in 1,4-dioxane. The progress of the amide formation is followed via HPLC and the solution of NH3 in 1,4-dioxane is added until all of the acid chloride is consumed. Then volatiles are removed in vacuo to yield a white solid.
To the crude product from above is added at r.t. 25 mL of POCl3 and the resulting mixture is heated to 100° C. for 4 h. Volatiles are removed in vacuo, the crude product is absorbed on silica gel and washed off with 20% ethyl acetate in hexane to give a white solid.
To 20 mg of the product so obtained in 0.5 mL of THF is added at r.t. 0.022 mL of N,N-diisopropylethylamine 16.3 mg; 0.126 mmol) followed by 21 mg of 3-amino-4,N-dimethylbenzamide (0.126 mmol). The mixture is stirred at r.t. for 2 h, then 0.5 mL of THF are added followed by 32 mg of (2,2-dimethyl-propyl)-methyl-amine, hydrochloride (0.232 mmol) and 0.044 mL of N,N-diisopropylethylamine (32.6 mg; 0.252 mmol). The resulting mixture is heated at 60° C. for 18 h. Volatiles are removed in vacuo and the crude mixture containing two separable regioisomers is purified via reversed phase prep. HPLC. (Yield: 3.9 mg: 0.011 mmol; 8%). MS (m/z): 367 (M+H).
4,6-Dichloro-5-nitro-pyrimidine (0.20 g, 1.0 mmol), N-methyl-neopentylamine hydrochloride (0.14 g) and DIEA (0.2 mL) were stirred in acetone (5 mL) at 0° C. for 4 hours. The solvent was removed in vacuo and the crude product was used for the next reaction without purification.
(6-Chloro-5-nitro-pyrimidin-4-yl)-(2,2-dimethyl-propyl)-methyl-amine (1.0 mmol) was dissolved in p-dioxane (2 mL), and 3-amino-4,N-dimethyl-benzamide (0.2 g, 1.2 mmol) and DIEA (0.3 mL) were added. The resulting mixture was heated to 80° C. for overnight. The product (0.31 g, yield 80%) was purified by column silica gel chromatography. MS (m/z): 387 (M+H).
4,6-Dichloro-5-nitro-pyrimidine (193 mg, 1 mmol) was dissolved in 4 mL acetone at 0° C. To the solution was added N-(2,2-dimethylpropyl)methyl amine HCl salt (137 mg, 1 mmol) and diisopropylethylamine (0.17 mL, 1 mmol). The solution was stirred at 0° C. for 10 minutes, then room temperature for 3 hours, and evaporated in vacuo. The crude product was purified by flash chromatography to afford 180 mg of the product (Yield: 69%). MS (m/z) calcd for C10H15ClN4O2 (MH+), 259.1, found, 259.3.
A solution of (6-Chloro-5-nitro-pyrimidin-4-yl)-(2,2-dimethyl-propyl)-methyl-amine (185 mg, 0.72 mmol), diisopropylethylamine (0.13 mL, 0.72 mmol) and 2-Methyl-5-(4H-[1,2,4]triazol-3-yl)-phenylamine (126 mg, 0.72 mmol) in 3 mL of n-BuOH was heated with stirring at 80° C. for 18 h. The solvent was then evaporated in vacuo and the crude product was purified by flash chromatography to afford 87 mg of the product (30%). MS (m/z) calcd for C19H24N8O2 (MH+), 397.2, found, 397.3.
To a solution of N-(2,2-dimethyl-propyl)-N-methyl-N′-[2-methyl-5-(4H-[1,2,4]triazol-3-yl)phenyl]-5-nitro-pyrimidine-4,6-diamine (20 mg, 0.05 mmol) in 5 mL of methanol was added a catalytic amount of 10% Pd/C. The vessel was placed under a hydrogen atmosphere of 20 psi for 1 h at room temperature. The solution was filtered, and the filtrate was evaporated under vacuum to afford 4.6 mg of the product (Yield: 25%). MS (m/z) calcd for C19H26N8 (MH+), 367.2, found, 367.4.
2-Methylsulfanyl-pyrimidine-4,6-diol (1.58 g, 10 mmol) was dissolved in 10 mL of con. H2SO4 at 0° C. To the solution was added 0.84 mL of nitric acid drop-wise. The solution was stirred at 0° C. for 30 minutes, then room temperature for 2 h. The solution was poured into ice water. The yellowish solid precipitated out of the solution was collected, washed with cold water and dried to afford 400 mg of the product (Yield: 20%).
2-Methylsulfanyl-5-nitro-pyrimidine-4,6-diol (203 mg, 1 mmol) was dissolved in 4 mL of phosphorus oxychloride. The solution was refluxed at 120° C. for two hours. The solution was evaporated under vacuum. The oily residue was purified by flash chromatography to afford 80 mg of the product (Yield: 35%).
4,6-Dichloro-2-methylsulfanyl-5-nitro-pyrimidine (30 mg, 0.13 mmol) and 3-amino-4,N-dimethyl-benzamide (22 mg, 0.13 mmol) were dissolved in 2 mL of THF. The solution was stirred at 0° C. for 30 minutes, and evaporated under vacuum. The product thus obtained was directly used for the next reaction without purification.
The intermediate so obtained was dissolved in 2 mL of THF. To the solution was added N-(2,2-dimethylpropyl)methyl amine HCl salt (36 mg, 0.26 mmol) and diisopropylethylamine (0.05 mL, 0.26 mmol). The solution was stirred at room temperature for 1 h, and evaporated under vacuum. The residue was purified by flash chromatography to afford 5.5 mg of the product (Yield: 10%, two steps). MS (m/z) calcd for C20H28N6O3S (MH+), 433.2, found, 433.2.
3-{6-[(2,2-dimethyl-propyl)-methyl-amino]-2-methylsulfanyl-5-nitro-pyrimidin-4-ylamino}-4,N-dimethyl-benzamide (24 mg, 0.06 mmol) was dissolved in 1 mL acetic acid. To the solution was added two drops of water and potassium permanganate (18 mg, 0.12 mmol). The solution was stirred at 60° C. for 10 minutes. The solution was diluted with water and extracted with ethyl acetate. The organic layer was washed with water, brine, dried over MgSO4, and evaporated under vacuum. The residue was further purified by flash chromatography to afford 4.2 mg of the product (Yield: 15%). MS (m/z) calcd for C20H28N6O5S (MH+), 465.2, found, 465.2.
A solution of 3-{6-[(2,2-Dimethyl-propyl)-methyl-amino]-2-methanesulfonyl-5-nitro-pyrimidin-4-ylamino}-4,N-dimethyl-benzamide (4.2 mg, 0.01 mmol) and 3-(Dimethylamino)propylamine (0.2 mL) in 2 mL of acetonitrile was heated with stirring at 80° C. for 16 h. The solvent was then evaporated under vacuum and the crude product was purified by flash chromatography to afford 2.8 mg of the product (Yield: 64%). MS (m/z) calcd for C24H38N8O3 (MH+), 487.3, found, 487.3.
1-Methyl-piperidin-4-ol (56 mg, 0.5 mmol) was dissolved in 2 mL of DMSO. To the solution was added potassium tert-butoxide (56 mg, 0.5 mmol). The solution was stirred at room temperature for 1 h. The solution was then added to a solution of 3-{6-[(2,2-Dimethyl-propyl)-methyl-amino]-2-methanesulfonyl-5-nitro-pyrimidin-4-ylamino}-4,N-dimethyl-benzamide (4 mg, 0.01 mmol) in 0.5 mL of DMSO. The solution was stirred at 80° C. for 3 h, and extracted with ethyl acetate and water. The organic layer was washed with water, brine, dried over MgSO4, and evaporated under vacuum. The residue was purified by semi-preparative hplc column to afford 0.7 mg of the product (Yield: 16%). MS (m/z) calcd for C25H37N7O4 (MH+), 500.2, found, 500.1.
Although the present invention has been described in some detail by way of illustration and example, for purposes of clarity and understanding, it will be apparent that certain changes and modifications may be practiced within the scope of the appended claims.
Claims (8)
1. A compound of Formula (I),
or enantiomers, diastereomers, tautomers, and pharmaceutically acceptable salts thereof,
wherein:
Y and X are ═N—;
is selected from ═C—CN, ═C—F, ═C—Br, ═C—NH2, ═C—NHC(O)CH3 and ═C—Cl;
V is —NR5—;
Z is halogen or —N(R1)(R2);
R1 and R2 are the same or different and are selected from hydrogen, alkyl, substituted alkyl, cycloalkyl, substituted cycloalkyl, saturated heterocyclyl or substituted saturated heterocyclyl;
R5 is hydrogen or alkyl;
R6 is
R7 is alkyl, substituted alkyl, alkoxy, or halogen;
R8 is hydrogen, alkyl, alkyloxy or cyano;
R9 is —C(O)R10 or unsubstituted or substituted heterocyclyl;
R10 is —N(R31)(R32);
R31 and R32 are the same or different and are selected from hydrogen, alkyl, substituted alkyl, alkoxy, aryl, substituted aryl, cycloalkyl, substituted cycloalkyl, heterocyclyl or substituted heterocyclyl; and
R11 is hydrogen or halogen.
2. A compound of claim 1 , or enantiomers, diastereomers, tautomers, and pharmaceutically acceptable salts thereof, wherein:
V is —NH—;
Z is —N(R1)(R2);
R1 and R2 are the same or different and are selected from hydrogen, alkyl or substituted alkyl, wherein alkyl is of 1 to 8 carbons;
R7 is alkyl of 1 to 4 carbons, alkoxy of 1 to 4 carbons, or halogen;
R8 is hydrogen; and
R10 is —NH2 or unsubstituted or substituted —NH-alkyl, —NH-alkoxy, —NH-heterocyclyl, —NH-phenyl, or —NH—CH2-phenyl, wherein alkyl and alkoxy are of 1 to 6 carbons.
3. A compound of claim 2 , or enantiomers, diastereomers, tautomers, and pharmaceutically acceptable salts thereof, wherein:
R1 and R2 are the same or different and are selected from hydrogen or alkyl of 1 to 8 carbons;
R7 is methyl, methoxy, Cl, Br, or F; and
R10 is —NH2, or unsubstituted or substituted —NH-alkyl, —NH-alkoxy, —NH-phenyl, or —NH—CH2-phenyl, wherein alkyl and alkoxy are of 1 to 6 carbons; and
R11 is hydrogen or halogen.
4. A compound of claim 3 , or enantiomers, diastereomers, tautomers, and pharmaceutically acceptable salts thereof,
wherein:
R10 is —NH2, —NH—CH3, —NH—C2H5, —NH—OCH3, or —NH—OC2H5.
5. A compound of claim 3 , or enantiomers, diastereomers, tautomers, and pharmaceutically acceptable salts thereof,
wherein:
R9 is unsubstituted or substituted triazole, thiazole, oxadiazole or imidazole.
7. A pharmaceutical composition comprising, as an active ingredient, a compound, or a salt thereof, according to claim 1 , and a pharmaceutically acceptable carrier.
8. A method of treating a TNF-α mediated disorder, comprising administering to a mammal in need of such treatment an effective amount of a composition according to claim 7 , wherein the TNF-α mediated disorder is rheumatoid arthritis, psoriasis, Crohn's Disease, osteoarthritis or osteoporosis.
Priority Applications (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| US11/485,075 US7253174B2 (en) | 2001-06-26 | 2006-07-12 | N-heterocyclic inhibitors of TNF-α expression |
| US11/778,219 US8044040B2 (en) | 2001-06-26 | 2007-07-16 | N-heterocyclic inhibitors of TNF-alpha expression |
Applications Claiming Priority (3)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| US30102001P | 2001-06-26 | 2001-06-26 | |
| US10/183,179 US20030139435A1 (en) | 2001-06-26 | 2002-06-26 | N-heterocyclic inhibitors of TNF-alpha expression |
| US11/485,075 US7253174B2 (en) | 2001-06-26 | 2006-07-12 | N-heterocyclic inhibitors of TNF-α expression |
Related Parent Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| US10/183,179 Continuation US20030139435A1 (en) | 2001-06-26 | 2002-06-26 | N-heterocyclic inhibitors of TNF-alpha expression |
Related Child Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| US11/778,219 Division US8044040B2 (en) | 2001-06-26 | 2007-07-16 | N-heterocyclic inhibitors of TNF-alpha expression |
Publications (2)
| Publication Number | Publication Date |
|---|---|
| US20060276488A1 US20060276488A1 (en) | 2006-12-07 |
| US7253174B2 true US7253174B2 (en) | 2007-08-07 |
Family
ID=23161582
Family Applications (3)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| US10/183,179 Abandoned US20030139435A1 (en) | 2001-06-26 | 2002-06-26 | N-heterocyclic inhibitors of TNF-alpha expression |
| US11/485,075 Expired - Lifetime US7253174B2 (en) | 2001-06-26 | 2006-07-12 | N-heterocyclic inhibitors of TNF-α expression |
| US11/778,219 Expired - Fee Related US8044040B2 (en) | 2001-06-26 | 2007-07-16 | N-heterocyclic inhibitors of TNF-alpha expression |
Family Applications Before (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| US10/183,179 Abandoned US20030139435A1 (en) | 2001-06-26 | 2002-06-26 | N-heterocyclic inhibitors of TNF-alpha expression |
Family Applications After (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| US11/778,219 Expired - Fee Related US8044040B2 (en) | 2001-06-26 | 2007-07-16 | N-heterocyclic inhibitors of TNF-alpha expression |
Country Status (7)
| Country | Link |
|---|---|
| US (3) | US20030139435A1 (en) |
| EP (1) | EP1406875B1 (en) |
| JP (1) | JP4510442B2 (en) |
| AU (1) | AU2002316421B2 (en) |
| CA (1) | CA2451128A1 (en) |
| HU (1) | HUP0401711A3 (en) |
| WO (1) | WO2003002544A1 (en) |
Cited By (8)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US20070254896A1 (en) * | 2006-03-23 | 2007-11-01 | Jeannene Butler | Substituted pyrimidine kinase inhibitors |
| US20070270425A1 (en) * | 2005-12-21 | 2007-11-22 | Guozhang Xu | Substituted pyrimidinyl oxime kinase inhibitors |
| US20080015185A1 (en) * | 2001-06-26 | 2008-01-17 | Bristol-Myers Squibb Company | N-heterocyclic inhibitors of tnf-alpha expression |
| US8354420B2 (en) | 2010-06-04 | 2013-01-15 | Genentech, Inc. | Aminopyrimidine derivatives as LRRK2 inhibitors |
| US8815882B2 (en) | 2010-11-10 | 2014-08-26 | Genentech, Inc. | Pyrazole aminopyrimidine derivatives as LRRK2 modulators |
| US11518758B2 (en) | 2019-05-10 | 2022-12-06 | Deciphera Pharmaceuticals, Llc | Heteroarylaminopyrimidine amide autophagy inhibitors and methods of use thereof |
| US11530206B2 (en) | 2019-05-10 | 2022-12-20 | Deciphera Pharmaceuticals, Llc | Phenylaminopyrimidine amide autophagy inhibitors and methods of use thereof |
| US11590134B2 (en) | 2019-06-17 | 2023-02-28 | Deciphera Pharmaceuticals, Llc | Aminopyrimidine amide autophagy inhibitors and methods of use thereof |
Families Citing this family (68)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US7037916B2 (en) * | 1999-07-15 | 2006-05-02 | Pharmacopeia Drug Discovery, Inc. | Pyrimidine derivatives as IL-8 receptor antagonists |
| GB0016877D0 (en) * | 2000-07-11 | 2000-08-30 | Astrazeneca Ab | Chemical compounds |
| US6670357B2 (en) * | 2000-11-17 | 2003-12-30 | Bristol-Myers Squibb Company | Methods of treating p38 kinase-associated conditions and pyrrolotriazine compounds useful as kinase inhibitors |
| US6867300B2 (en) | 2000-11-17 | 2005-03-15 | Bristol-Myers Squibb Company | Methods for the preparation of pyrrolotriazine compounds useful as kinase inhibitors |
| KR20040062557A (en) * | 2001-11-01 | 2004-07-07 | 얀센 파마슈티카 엔.브이. | Aminobenzamide derivatives as glycogen synthase kinase 3β inhibitors |
| TWI329105B (en) | 2002-02-01 | 2010-08-21 | Rigel Pharmaceuticals Inc | 2,4-pyrimidinediamine compounds and their uses |
| KR101025675B1 (en) * | 2002-04-23 | 2011-03-30 | 브리스톨-마이어스 스큅 컴퍼니 | Pyrrolo-triazine Aniline Compounds Useful as Kinase Inhibitors |
| JP2005529890A (en) | 2002-04-23 | 2005-10-06 | ブリストル−マイヤーズ スクイブ カンパニー | Aryl ketone pyrrolotriazine compounds useful as kinase inhibitors |
| ES2445208T3 (en) * | 2002-07-29 | 2014-02-28 | Rigel Pharmaceuticals, Inc. | 2,4-Pyrimidinediamine compounds for use in methods to treat or prevent autoimmune diseases |
| BR0316680A (en) * | 2002-11-28 | 2005-10-18 | Schering Ag | Chk, pdk and akt inhibitor pyrimidines, their production and use as pharmaceutical agents |
| US6909001B2 (en) | 2002-12-12 | 2005-06-21 | Pharmacia Corporation | Method of making tricyclic aminocyanopyridine compounds |
| WO2004054505A2 (en) * | 2002-12-12 | 2004-07-01 | Pharmacia Corporation | Method of using aminocyanopyridine compounds as mitogen activated protein kinase-activated protein kinase-2 inhibitors |
| WO2004069829A1 (en) * | 2003-01-10 | 2004-08-19 | Pharmacopeia Drug Discovery, Inc. | (2s)-2-((pyrimidin-4-yl)amino)-4-methylpentanoic acid aminoethylamid derivatives as il-8 receptor modulators for the treatment of atherosclerosis and rheumatoid arthritis |
| SI1599468T1 (en) * | 2003-01-14 | 2008-02-29 | Arena Pharm Inc | 1,2,3-trisubstituted aryl and heteroaryl derivatives as modulators of metabolism and the prophylaxis and treatment of disorders related thereto such as diabetes and hyperglycemia |
| UA92450C2 (en) * | 2003-01-14 | 2010-11-10 | Арена Фармасьютикалз, Инк. | 1,2,3-trisubstituted aryl and heteroaryl derivatives as modulators of metabolism and the prophylaxis and treatment of disorders related thereto such as diabetes and hyperglycemia |
| MXPA05007485A (en) * | 2003-01-14 | 2006-01-30 | Arena Pharm Inc | 1,2,3-trisubstituted aryl and heteroaryl derivatives as modulators of metabolism and the prpphylaxis and treatment of disorders related thereto such as diabetes and hyperglycemia. |
| BRPI0407282A (en) | 2003-02-05 | 2006-01-31 | Bristol Myers Squibb Co | Process for preparing pyrrolotriazine kinase inhibitors |
| US20050014753A1 (en) * | 2003-04-04 | 2005-01-20 | Irm Llc | Novel compounds and compositions as protein kinase inhibitors |
| DK1618092T3 (en) * | 2003-05-01 | 2011-01-31 | Bristol Myers Squibb Co | Aryl-substituted pyrazole-amide compounds useful as kinase inhibitors |
| US7652044B2 (en) | 2003-06-03 | 2010-01-26 | Novartis A.G. | P-38 inhibitors |
| WO2004113305A2 (en) | 2003-06-16 | 2004-12-29 | Vertex Pharmaceuticals Incorporated | Diamino substituted quinazoline derivatives as promoters of smn2 |
| US7504396B2 (en) * | 2003-06-24 | 2009-03-17 | Amgen Inc. | Substituted heterocyclic compounds and methods of use |
| AR045047A1 (en) * | 2003-07-11 | 2005-10-12 | Arena Pharm Inc | ARILO AND HETEROARILO DERIVATIVES TRISUSTITUIDOS AS MODULATORS OF METABOLISM AND PROFILAXIS AND TREATMENT OF DISORDERS RELATED TO THEMSELVES |
| BRPI0413018B8 (en) | 2003-07-30 | 2021-05-25 | Rigel Pharmaceuticals Inc | compost, and use of a compost |
| NZ585188A (en) * | 2003-08-15 | 2011-09-30 | Novartis Ag | 2,4-Pyrimidinediamines useful in the treatment of neoplastic diseases, inflammatory and immune system disorders |
| PE20050483A1 (en) | 2003-10-31 | 2005-08-25 | Arena Pharm Inc | TETRAZOLE DERIVATIVES OF FORMULA (I), ITS PHARMACEUTICAL COMPOSITIONS AND PROCESSES TO PRODUCE PHARMACEUTICAL COMPOSITIONS |
| CA2545048A1 (en) * | 2003-11-20 | 2005-06-02 | Warner-Lambert Company Llc | Androgen receptor modulators |
| US7459562B2 (en) * | 2004-04-23 | 2008-12-02 | Bristol-Myers Squibb Company | Monocyclic heterocycles as kinase inhibitors |
| EP1763514A2 (en) | 2004-05-18 | 2007-03-21 | Rigel Pharmaceuticals, Inc. | Cycloalkyl substituted pyrimidinediamine compounds and their uses |
| EP1598343A1 (en) * | 2004-05-19 | 2005-11-23 | Boehringer Ingelheim International GmbH | 2-Arylaminopyrimidine derivatives as PLK inhibitors |
| EA011671B1 (en) * | 2004-06-04 | 2009-04-28 | Арена Фармасьютикалз, Инк. | Substituted aryl and heteroaryl derivatives as modulators of metabolism and the prophylaxis and treatment of disorders related thereto |
| US7253167B2 (en) * | 2004-06-30 | 2007-08-07 | Bristol-Myers Squibb Company | Tricyclic-heteroaryl compounds useful as kinase inhibitors |
| TW200600513A (en) * | 2004-06-30 | 2006-01-01 | Bristol Myers Squibb Co | A method for preparing pyrrolotriazine compounds |
| US7504521B2 (en) | 2004-08-05 | 2009-03-17 | Bristol-Myers Squibb Co. | Methods for the preparation of pyrrolotriazine compounds |
| US7148348B2 (en) | 2004-08-12 | 2006-12-12 | Bristol-Myers Squibb Company | Process for preparing pyrrolotriazine aniline compounds useful as kinase inhibitors |
| US7820654B2 (en) * | 2004-09-23 | 2010-10-26 | Dr. Reddy's Laboratories Ltd. | Pyrimidine compounds, process for their preparation and compositions containing them |
| WO2006037117A1 (en) * | 2004-09-27 | 2006-04-06 | Amgen Inc. | Substituted heterocyclic compounds and methods of use |
| GB2420559B (en) | 2004-11-15 | 2008-08-06 | Rigel Pharmaceuticals Inc | Stereoisomerically enriched 3-aminocarbonyl bicycloheptene pyrimidinediamine compounds and their uses |
| EP1814878B1 (en) | 2004-11-24 | 2012-01-04 | Rigel Pharmaceuticals, Inc. | Spiro-2, 4-pyrimidinediamine compounds and their uses |
| MY148521A (en) * | 2005-01-10 | 2013-04-30 | Arena Pharm Inc | Substituted pyridinyl and pyrimidinyl derivatives as modulators of metabolism and the treatment of disorders related thereto |
| DE602006010979D1 (en) | 2005-01-19 | 2010-01-21 | Rigel Pharmaceuticals Inc | PRODRUGS FROM 2,4-PYRIMIDIN DIAMIN COMPOUNDS AND THEIR USES |
| US20060247263A1 (en) * | 2005-04-19 | 2006-11-02 | Amgen Inc. | Substituted heterocyclic compounds and methods of use |
| US8278446B2 (en) * | 2005-12-21 | 2012-10-02 | Janssen Pharmaceutica N.V. | Process for preparing substituted diaminopyrimidine oximes |
| WO2007081630A2 (en) * | 2005-12-21 | 2007-07-19 | Janssen Pharmaceutica, N.V. | Substituted pyrimidinyl kinase inhibitors |
| EP2046763A2 (en) | 2006-07-31 | 2009-04-15 | Praecis Pharmaceuticals Incorporated | Aurora kinase inhibitors from an encoded small molecule library |
| JP2009545598A (en) * | 2006-08-01 | 2009-12-24 | プリーシス・ファーマシューティカルズ・インコーポレイテッド | P38 kinase inhibitor |
| CN101535276B (en) | 2006-10-23 | 2013-08-28 | 赛福伦公司 | 2,4-Diaminopyrimidine-fused bicyclic derivatives as ALK and c-MET inhibitors |
| US20100137398A1 (en) * | 2007-05-04 | 2010-06-03 | Novartis Ag | Use of hdac inhibitors for the treatment of gastrointestinal cancers |
| CA2693594A1 (en) * | 2007-07-17 | 2009-01-22 | Rigel Pharmaceuticals, Inc. | Cyclic amine substituted pyrimidinediamines as pkc inhibitors |
| AU2008317566B2 (en) | 2007-10-26 | 2014-05-01 | Academisch Ziekenhuis Leiden | Means and methods for counteracting muscle disorders |
| PL2252300T3 (en) | 2008-02-22 | 2017-04-28 | Rigel Pharmaceuticals, Inc. | Use of 2,4-pyrimidinediamines for the treatment of atherosclerosis |
| EP2119783A1 (en) | 2008-05-14 | 2009-11-18 | Prosensa Technologies B.V. | Method for efficient exon (44) skipping in Duchenne Muscular Dystrophy and associated means |
| US20110245156A1 (en) * | 2008-12-09 | 2011-10-06 | Cytokine Pharmasciences, Inc. | Novel antiviral compounds, compositions, and methods of use |
| AT509266B1 (en) * | 2009-12-28 | 2014-07-15 | Univ Wien Tech | SUBSTITUTED PYRIDINES AND PYRIMIDINES |
| JP2013517273A (en) * | 2010-01-13 | 2013-05-16 | グラクソスミスクライン・リミテッド・ライアビリティ・カンパニー | Compounds and methods |
| PH12013500547A1 (en) | 2010-09-22 | 2013-06-10 | Arena Pharm Inc | Modulators of the gpr119 receptor and the treatment of disorders related thereto |
| CN112251436A (en) | 2012-01-27 | 2021-01-22 | 比奥马林技术公司 | RNA-regulated oligonucleotides with improved properties for the treatment of duchenne muscular dystrophy and becker muscular dystrophy |
| CN102952084A (en) * | 2012-11-15 | 2013-03-06 | 大连九信生物化工科技有限公司 | A kind of synthetic method of 4,6-dichloro-2-methylthio-5-nitropyrimidine |
| EP3082819B1 (en) | 2013-12-20 | 2020-06-17 | Signal Pharmaceuticals, LLC | Substituted diaminopyrimidyl compounds, compositions thereof, and methods of treatment therewith |
| MX386419B (en) | 2015-01-06 | 2025-03-18 | Arena Pharm Inc | METHODS OF TREATMENT CONDITIONS RELATED TO THE S1P1 RECEPTOR. |
| EP3939965A1 (en) | 2015-06-22 | 2022-01-19 | Arena Pharmaceuticals, Inc. | Crystalline l-arginine salt of (r)-2-(7-(4-cyclopentyl-3-(trifluoromethyl)benzyloxy)-1,2,3,4-tetrahydrocyclo-penta[b]indol-3-yl)acetic acid for use in sipi receptor-associated disorders |
| WO2017151409A1 (en) | 2016-02-29 | 2017-09-08 | University Of Florida Research Foundation, Incorporated | Chemotherapeutic methods |
| HUE064145T2 (en) * | 2016-04-15 | 2024-03-28 | Epizyme Inc | Amine-substituted aryl or heteroaryl compounds as EHMT1 and EHMT2 inhibitors |
| MA47504A (en) | 2017-02-16 | 2019-12-25 | Arena Pharm Inc | COMPOUNDS AND TREATMENT METHODS FOR PRIMITIVE BILIARY ANGIOCHOLITIS |
| EP3619198B1 (en) * | 2017-07-18 | 2020-06-17 | Lonza Ltd | Method for preparation of chlorinated s-propylthiobarbituric acid |
| WO2019071147A1 (en) | 2017-10-05 | 2019-04-11 | Fulcrum Therapeutics, Inc. | P38 kinase inhibitors reduce dux4 and downstream gene expression for the treatment of fshd |
| US10342786B2 (en) | 2017-10-05 | 2019-07-09 | Fulcrum Therapeutics, Inc. | P38 kinase inhibitors reduce DUX4 and downstream gene expression for the treatment of FSHD |
| ES2987794T3 (en) | 2018-06-06 | 2024-11-18 | Arena Pharm Inc | Treatment procedures for S1P1 receptor-related conditions |
Citations (11)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US5428044A (en) | 1989-05-20 | 1995-06-27 | Fisons Plc | Anti-inflammatory 4-aminophenyl derivatives |
| US6107301A (en) | 1993-10-12 | 2000-08-22 | Dupont Pharmaceuticals Company | 1N-alkyl-N-arylpyrimidinamines and derivatives thereof |
| WO2001019825A1 (en) | 1999-09-15 | 2001-03-22 | Warner-Lambert Company | Pteridinones as kinase inhibitors |
| WO2001027089A1 (en) | 1999-10-13 | 2001-04-19 | Astrazeneca Ab | Pyrimidine derivatives |
| WO2001028561A1 (en) | 1999-10-21 | 2001-04-26 | Merck & Co., Inc. | Gram-positive selective antibacterial compounds, compositions containing such compounds and methods of treatment |
| WO2001047897A1 (en) | 1999-12-28 | 2001-07-05 | Pharmacopeia, Inc. | Cytokine, especially tnf-alpha, inhibitors |
| WO2001072717A1 (en) * | 2000-03-28 | 2001-10-04 | Astrazeneca Ab | 4-amino-5-cyano-2-anilino-pyrimidine derivatives and their use as inhibitors of cell-cycle kinases |
| WO2003002542A1 (en) | 2000-12-22 | 2003-01-09 | Bristol-Myers Squibb Company | N-heterocyclic inhibitors of tnf-alpha expression |
| WO2003032994A2 (en) | 2001-10-17 | 2003-04-24 | Boehringer Ingelheim Pharma Gmbh & Co. Kg | Novel tri-substituted pyrimidines, method for production and use thereof as medicament |
| WO2003032997A1 (en) | 2001-10-17 | 2003-04-24 | Boehringer Ingelheim Pharma Gmbh & Co. Kg | Pyrimidine derivatives, pharmaceutical agent containing said compounds, use and method for making same |
| WO2003063794A2 (en) | 2002-02-01 | 2003-08-07 | Rigel Pharmaceuticals, Inc. | 2,4-pyrimidinediamine compounds and their uses |
Family Cites Families (10)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JPS6110563A (en) * | 1984-06-25 | 1986-01-18 | Hokuriku Seiyaku Co Ltd | Pyrimidylaminophenylpropionic acid derivative |
| DE19651213A1 (en) * | 1996-12-10 | 1998-08-06 | Dystar Textilfarben Gmbh & Co | Water-soluble fiber-reactive dyes, processes for their preparation and their use |
| DE19715603A1 (en) * | 1997-04-15 | 1998-10-22 | Dystar Textilfarben Gmbh & Co | Azo dyes, process for their preparation and their use |
| MY132496A (en) * | 1998-05-11 | 2007-10-31 | Vertex Pharma | Inhibitors of p38 |
| GB9828511D0 (en) * | 1998-12-24 | 1999-02-17 | Zeneca Ltd | Chemical compounds |
| GB0004887D0 (en) * | 2000-03-01 | 2000-04-19 | Astrazeneca Uk Ltd | Chemical compounds |
| GB0004890D0 (en) * | 2000-03-01 | 2000-04-19 | Astrazeneca Uk Ltd | Chemical compounds |
| EP1343782B1 (en) | 2000-12-21 | 2009-05-06 | SmithKline Beecham Corporation | Pyrimidineamines as angiogenesis modulators |
| KR100874791B1 (en) | 2001-05-29 | 2008-12-18 | 바이엘 쉐링 파마 악티엔게젤샤프트 | CDV-inhibited pyrimidine, preparation method thereof and use as medicament |
| US20030139435A1 (en) * | 2001-06-26 | 2003-07-24 | Gulzar Ahmed | N-heterocyclic inhibitors of TNF-alpha expression |
-
2002
- 2002-06-26 US US10/183,179 patent/US20030139435A1/en not_active Abandoned
- 2002-06-26 HU HU0401711A patent/HUP0401711A3/en unknown
- 2002-06-26 JP JP2003508925A patent/JP4510442B2/en not_active Expired - Fee Related
- 2002-06-26 WO PCT/US2002/020341 patent/WO2003002544A1/en not_active Ceased
- 2002-06-26 EP EP02746724.0A patent/EP1406875B1/en not_active Expired - Lifetime
- 2002-06-26 AU AU2002316421A patent/AU2002316421B2/en not_active Ceased
- 2002-06-26 CA CA002451128A patent/CA2451128A1/en not_active Abandoned
-
2006
- 2006-07-12 US US11/485,075 patent/US7253174B2/en not_active Expired - Lifetime
-
2007
- 2007-07-16 US US11/778,219 patent/US8044040B2/en not_active Expired - Fee Related
Patent Citations (12)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US5428044A (en) | 1989-05-20 | 1995-06-27 | Fisons Plc | Anti-inflammatory 4-aminophenyl derivatives |
| US6107301A (en) | 1993-10-12 | 2000-08-22 | Dupont Pharmaceuticals Company | 1N-alkyl-N-arylpyrimidinamines and derivatives thereof |
| US6342503B1 (en) | 1993-10-12 | 2002-01-29 | Dupont Pharmaceuticals Company | 1N-alkyl-n-arylpyrimidinamines and derivatives thereof |
| WO2001019825A1 (en) | 1999-09-15 | 2001-03-22 | Warner-Lambert Company | Pteridinones as kinase inhibitors |
| WO2001027089A1 (en) | 1999-10-13 | 2001-04-19 | Astrazeneca Ab | Pyrimidine derivatives |
| WO2001028561A1 (en) | 1999-10-21 | 2001-04-26 | Merck & Co., Inc. | Gram-positive selective antibacterial compounds, compositions containing such compounds and methods of treatment |
| WO2001047897A1 (en) | 1999-12-28 | 2001-07-05 | Pharmacopeia, Inc. | Cytokine, especially tnf-alpha, inhibitors |
| WO2001072717A1 (en) * | 2000-03-28 | 2001-10-04 | Astrazeneca Ab | 4-amino-5-cyano-2-anilino-pyrimidine derivatives and their use as inhibitors of cell-cycle kinases |
| WO2003002542A1 (en) | 2000-12-22 | 2003-01-09 | Bristol-Myers Squibb Company | N-heterocyclic inhibitors of tnf-alpha expression |
| WO2003032994A2 (en) | 2001-10-17 | 2003-04-24 | Boehringer Ingelheim Pharma Gmbh & Co. Kg | Novel tri-substituted pyrimidines, method for production and use thereof as medicament |
| WO2003032997A1 (en) | 2001-10-17 | 2003-04-24 | Boehringer Ingelheim Pharma Gmbh & Co. Kg | Pyrimidine derivatives, pharmaceutical agent containing said compounds, use and method for making same |
| WO2003063794A2 (en) | 2002-02-01 | 2003-08-07 | Rigel Pharmaceuticals, Inc. | 2,4-pyrimidinediamine compounds and their uses |
Non-Patent Citations (6)
| Title |
|---|
| Chen et al., Biochemistry 1998, 37, p. 17735-17744. |
| Database Caplus, Accession No. 104:168491, JP61-10563 B (Ito et al.) Jan. 18, 1986 see CAS abstract. |
| Ulrich, Chapter 4: Crystallization, Kirk-Othmer Encyclopedia of Chemical Technology, Aug. 2002. * |
| Vippagunta et al., Cyrstalline Solids, Advanced Drug Delivery Reviews, 48, pp. 3-26, 2001. * |
| West, Solid Solutions, Solid State Chemistry and its applications, pp. 358 & 365, 1988. * |
| Wissner et al., J. Med. Chem. 2000, 43, pp. 3244-3256. |
Cited By (15)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US20080015185A1 (en) * | 2001-06-26 | 2008-01-17 | Bristol-Myers Squibb Company | N-heterocyclic inhibitors of tnf-alpha expression |
| US8044040B2 (en) * | 2001-06-26 | 2011-10-25 | Bristol-Myers Squibb Company | N-heterocyclic inhibitors of TNF-alpha expression |
| US8367825B2 (en) | 2005-12-21 | 2013-02-05 | Janssen Pharmaceutica N.V. | Substituted pyrimidinyl oxime kinase inhibitors |
| US20070270425A1 (en) * | 2005-12-21 | 2007-11-22 | Guozhang Xu | Substituted pyrimidinyl oxime kinase inhibitors |
| US8153791B2 (en) * | 2005-12-21 | 2012-04-10 | Janssen Pharmaceutica N.V. | Substituted pyrimidinyl oxime kinase inhibitors |
| US8013153B2 (en) | 2006-03-23 | 2011-09-06 | Janssen Pharmaceutica, N.V. | Substituted pyrimidine kinase inhibitors |
| US20070254896A1 (en) * | 2006-03-23 | 2007-11-01 | Jeannene Butler | Substituted pyrimidine kinase inhibitors |
| US8354420B2 (en) | 2010-06-04 | 2013-01-15 | Genentech, Inc. | Aminopyrimidine derivatives as LRRK2 inhibitors |
| US8815882B2 (en) | 2010-11-10 | 2014-08-26 | Genentech, Inc. | Pyrazole aminopyrimidine derivatives as LRRK2 modulators |
| US11518758B2 (en) | 2019-05-10 | 2022-12-06 | Deciphera Pharmaceuticals, Llc | Heteroarylaminopyrimidine amide autophagy inhibitors and methods of use thereof |
| US11530206B2 (en) | 2019-05-10 | 2022-12-20 | Deciphera Pharmaceuticals, Llc | Phenylaminopyrimidine amide autophagy inhibitors and methods of use thereof |
| US12071432B2 (en) | 2019-05-10 | 2024-08-27 | Deciphera Pharmaceuticals, Llc | Phenylaminopyrimidine amide autophagy inhibitors and methods of use thereof |
| US12479833B2 (en) | 2019-05-10 | 2025-11-25 | Deciphera Pharmaceuticals, Llc | Heteroarylaminopyrimidine amide autophagy inhibitors and methods of use thereof |
| US11590134B2 (en) | 2019-06-17 | 2023-02-28 | Deciphera Pharmaceuticals, Llc | Aminopyrimidine amide autophagy inhibitors and methods of use thereof |
| US12377097B2 (en) | 2019-06-17 | 2025-08-05 | Deciphera Pharmaceuticals, Llc | Aminopyrimidine amide autophagy inhibitors and methods of use thereof |
Also Published As
| Publication number | Publication date |
|---|---|
| HUP0401711A2 (en) | 2004-12-28 |
| EP1406875B1 (en) | 2013-07-31 |
| US20030139435A1 (en) | 2003-07-24 |
| JP4510442B2 (en) | 2010-07-21 |
| US8044040B2 (en) | 2011-10-25 |
| US20080015185A1 (en) | 2008-01-17 |
| US20060276488A1 (en) | 2006-12-07 |
| JP2004535447A (en) | 2004-11-25 |
| WO2003002544A1 (en) | 2003-01-09 |
| CA2451128A1 (en) | 2003-01-09 |
| EP1406875A4 (en) | 2005-05-04 |
| EP1406875A1 (en) | 2004-04-14 |
| HUP0401711A3 (en) | 2009-07-28 |
| AU2002316421B2 (en) | 2008-05-15 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| US7253174B2 (en) | N-heterocyclic inhibitors of TNF-α expression | |
| AU2002316421A1 (en) | N-heterocyclic inhibitors of TNF-ALPHA expression | |
| US6906067B2 (en) | N-heterocyclic inhibitors of TNF-α expression | |
| EP1242385B1 (en) | Cytokine, especially tnf-alpha, inhibitors | |
| US20020065270A1 (en) | N-heterocyclic inhibitors of TNF-alpha expression | |
| US6962915B2 (en) | Pyrazolo-pyrimidine aniline compounds useful as kinase inhibitors | |
| US20060069110A1 (en) | Substituted heterocyclic compounds and methods of use | |
| KR101693179B1 (en) | Nitrogen-containing heterocyclic compound or salt thereof | |
| EP1339687B1 (en) | 4-6-diphenyl pyridine derivatives as antiinflammatory agents | |
| US20050250770A1 (en) | Fused heterocyclic compounds | |
| CN1406230A (en) | 2,4-di (hetero-) arylamino (-oxy)-5-substituted pyrimidines as antineoplastic agents | |
| JP2007500129A (en) | p-38 kinase inhibitor | |
| JPWO1999031073A1 (en) | Novel pyrimidine-5-carboxamide derivatives | |
| JP2002514195A (en) | Substituted pyrimidine compounds and uses thereof | |
| US20040102464A1 (en) | Arylalkane-sulfonamides having endothelin-antagonist activity | |
| US7037916B2 (en) | Pyrimidine derivatives as IL-8 receptor antagonists | |
| EP1409463B1 (en) | N-heterocyclic inhibitors of tnf-alpha expression | |
| BG65128B1 (en) | Substituted pyramidine compounds and their application | |
| KR100647932B1 (en) | Quinazoline derivatives | |
| HUP0204168A2 (en) | Butyne diol derivatives, pharmaceutical composition containing the same and its preparation | |
| AU2002312590A1 (en) | N-heterocyclic inhibitors of TNF-alpha expression | |
| KR20000069328A (en) | Substituted pyrimidine compounds and their use |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| STCF | Information on status: patent grant |
Free format text: PATENTED CASE |
|
| CC | Certificate of correction | ||
| FPAY | Fee payment |
Year of fee payment: 4 |
|
| FPAY | Fee payment |
Year of fee payment: 8 |
|
| MAFP | Maintenance fee payment |
Free format text: PAYMENT OF MAINTENANCE FEE, 12TH YEAR, LARGE ENTITY (ORIGINAL EVENT CODE: M1553); ENTITY STATUS OF PATENT OWNER: LARGE ENTITY Year of fee payment: 12 |