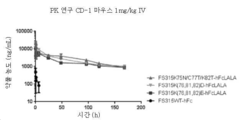
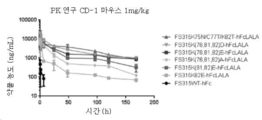
KR20180137487A - 재조합 폴리스타틴-fc 융합 단백질 및 듀시엔형 근이영양증 치료에서의 용도 - Google Patents
재조합 폴리스타틴-fc 융합 단백질 및 듀시엔형 근이영양증 치료에서의 용도 Download PDFInfo
- Publication number
- KR20180137487A KR20180137487A KR1020187028662A KR20187028662A KR20180137487A KR 20180137487 A KR20180137487 A KR 20180137487A KR 1020187028662 A KR1020187028662 A KR 1020187028662A KR 20187028662 A KR20187028662 A KR 20187028662A KR 20180137487 A KR20180137487 A KR 20180137487A
- Authority
- KR
- South Korea
- Prior art keywords
- cys
- glu
- lys
- ser
- gly
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Withdrawn
Links
- 108020001507 fusion proteins Proteins 0.000 title claims abstract description 161
- 201000006938 muscular dystrophy Diseases 0.000 title abstract description 24
- 229940121710 HMGCoA reductase inhibitor Drugs 0.000 claims abstract description 240
- 102000037865 fusion proteins Human genes 0.000 claims abstract description 157
- 238000000034 method Methods 0.000 claims abstract description 53
- 206010013801 Duchenne Muscular Dystrophy Diseases 0.000 claims abstract description 52
- 239000000203 mixture Substances 0.000 claims abstract description 35
- 208000024891 symptom Diseases 0.000 claims abstract description 23
- 230000003247 decreasing effect Effects 0.000 claims abstract description 9
- 108090000623 proteins and genes Proteins 0.000 claims description 179
- 210000003205 muscle Anatomy 0.000 claims description 173
- 102000004169 proteins and genes Human genes 0.000 claims description 169
- 108090000765 processed proteins & peptides Proteins 0.000 claims description 139
- 229920001184 polypeptide Polymers 0.000 claims description 127
- 102000004196 processed proteins & peptides Human genes 0.000 claims description 127
- 150000001413 amino acids Chemical class 0.000 claims description 122
- 208000037265 diseases, disorders, signs and symptoms Diseases 0.000 claims description 98
- 125000003275 alpha amino acid group Chemical group 0.000 claims description 87
- 230000027455 binding Effects 0.000 claims description 70
- 241000282414 Homo sapiens Species 0.000 claims description 55
- FWMNVWWHGCHHJJ-SKKKGAJSSA-N 4-amino-1-[(2r)-6-amino-2-[[(2r)-2-[[(2r)-2-[[(2r)-2-amino-3-phenylpropanoyl]amino]-3-phenylpropanoyl]amino]-4-methylpentanoyl]amino]hexanoyl]piperidine-4-carboxylic acid Chemical compound C([C@H](C(=O)N[C@H](CC(C)C)C(=O)N[C@H](CCCCN)C(=O)N1CCC(N)(CC1)C(O)=O)NC(=O)[C@H](N)CC=1C=CC=CC=1)C1=CC=CC=C1 FWMNVWWHGCHHJJ-SKKKGAJSSA-N 0.000 claims description 45
- 108010004165 polystatin Proteins 0.000 claims description 45
- 230000035772 mutation Effects 0.000 claims description 41
- HTTJABKRGRZYRN-UHFFFAOYSA-N Heparin Chemical compound OC1C(NC(=O)C)C(O)OC(COS(O)(=O)=O)C1OC1C(OS(O)(=O)=O)C(O)C(OC2C(C(OS(O)(=O)=O)C(OC3C(C(O)C(O)C(O3)C(O)=O)OS(O)(=O)=O)C(CO)O2)NS(O)(=O)=O)C(C(O)=O)O1 HTTJABKRGRZYRN-UHFFFAOYSA-N 0.000 claims description 38
- 229920000669 heparin Polymers 0.000 claims description 38
- 229960002897 heparin Drugs 0.000 claims description 38
- 102100039939 Growth/differentiation factor 8 Human genes 0.000 claims description 36
- 108010056852 Myostatin Proteins 0.000 claims description 36
- 238000006467 substitution reaction Methods 0.000 claims description 34
- 210000004027 cell Anatomy 0.000 claims description 32
- 208000035475 disorder Diseases 0.000 claims description 31
- 239000008194 pharmaceutical composition Substances 0.000 claims description 30
- 239000003814 drug Substances 0.000 claims description 29
- 230000001965 increasing effect Effects 0.000 claims description 22
- 229940124597 therapeutic agent Drugs 0.000 claims description 20
- 230000002829 reductive effect Effects 0.000 claims description 19
- 101710196692 Actin A Proteins 0.000 claims description 18
- 244000309466 calf Species 0.000 claims description 18
- 102220562703 Protein Tob2_L234A_mutation Human genes 0.000 claims description 13
- 238000010494 dissociation reaction Methods 0.000 claims description 13
- 230000005593 dissociations Effects 0.000 claims description 13
- 102000005962 receptors Human genes 0.000 claims description 13
- 230000007935 neutral effect Effects 0.000 claims description 12
- 108091033319 polynucleotide Proteins 0.000 claims description 10
- 102000040430 polynucleotide Human genes 0.000 claims description 10
- 239000002157 polynucleotide Substances 0.000 claims description 10
- 210000002027 skeletal muscle Anatomy 0.000 claims description 10
- 102100026120 IgG receptor FcRn large subunit p51 Human genes 0.000 claims description 9
- 101710177940 IgG receptor FcRn large subunit p51 Proteins 0.000 claims description 9
- 239000003937 drug carrier Substances 0.000 claims description 8
- 238000001990 intravenous administration Methods 0.000 claims description 8
- 208000028867 ischemia Diseases 0.000 claims description 8
- -1 pamrevlumab Chemical compound 0.000 claims description 8
- 239000003795 chemical substances by application Substances 0.000 claims description 7
- 230000004064 dysfunction Effects 0.000 claims description 6
- 238000007918 intramuscular administration Methods 0.000 claims description 6
- 230000033001 locomotion Effects 0.000 claims description 6
- 230000009756 muscle regeneration Effects 0.000 claims description 6
- 238000007911 parenteral administration Methods 0.000 claims description 6
- 230000009467 reduction Effects 0.000 claims description 6
- 238000007920 subcutaneous administration Methods 0.000 claims description 6
- 230000003442 weekly effect Effects 0.000 claims description 6
- 208000031229 Cardiomyopathies Diseases 0.000 claims description 5
- 102220639700 Leptin_R78L_mutation Human genes 0.000 claims description 5
- 206010028289 Muscle atrophy Diseases 0.000 claims description 5
- 230000006872 improvement Effects 0.000 claims description 5
- 206010028320 muscle necrosis Diseases 0.000 claims description 5
- 201000000585 muscular atrophy Diseases 0.000 claims description 5
- 102220226509 rs63750437 Human genes 0.000 claims description 5
- 208000010428 Muscle Weakness Diseases 0.000 claims description 4
- 206010028372 Muscular weakness Diseases 0.000 claims description 4
- 102220587527 Putative uncharacterized protein FLJ13197_K81D_mutation Human genes 0.000 claims description 4
- 108010023082 activin A Proteins 0.000 claims description 4
- 102220371048 c.225A>C Human genes 0.000 claims description 4
- 239000013604 expression vector Substances 0.000 claims description 4
- 230000001771 impaired effect Effects 0.000 claims description 4
- 239000002773 nucleotide Substances 0.000 claims description 4
- 125000003729 nucleotide group Chemical group 0.000 claims description 4
- 230000000241 respiratory effect Effects 0.000 claims description 4
- 102220017540 rs118203356 Human genes 0.000 claims description 4
- 102220050493 rs193921030 Human genes 0.000 claims description 4
- 210000000453 second toe Anatomy 0.000 claims description 4
- 102200087803 c.241A>G Human genes 0.000 claims description 3
- 208000010877 cognitive disease Diseases 0.000 claims description 3
- 239000012634 fragment Substances 0.000 claims description 3
- 238000001415 gene therapy Methods 0.000 claims description 3
- 230000002188 osteogenic effect Effects 0.000 claims description 3
- 229960004618 prednisone Drugs 0.000 claims description 3
- XOFYZVNMUHMLCC-ZPOLXVRWSA-N prednisone Chemical compound O=C1C=C[C@]2(C)[C@H]3C(=O)C[C@](C)([C@@](CC4)(O)C(=O)CO)[C@@H]4[C@@H]3CCC2=C1 XOFYZVNMUHMLCC-ZPOLXVRWSA-N 0.000 claims description 3
- 102200105315 rs748277951 Human genes 0.000 claims description 3
- 102200127051 rs9511510 Human genes 0.000 claims description 3
- 230000009747 swallowing Effects 0.000 claims description 3
- 230000000699 topical effect Effects 0.000 claims description 3
- 101001010152 Aplysia californica Probable glutathione transferase Proteins 0.000 claims description 2
- 206010061218 Inflammation Diseases 0.000 claims description 2
- 102220477772 Interferon-inducible GTPase 5_K82A_mutation Human genes 0.000 claims description 2
- 206010023201 Joint contracture Diseases 0.000 claims description 2
- 102220591431 Relaxin receptor 1_K81A_mutation Human genes 0.000 claims description 2
- 102220481747 Zinc finger SWIM domain-containing protein 3_C66S_mutation Human genes 0.000 claims description 2
- 230000006741 behavioral dysfunction Effects 0.000 claims description 2
- 210000000988 bone and bone Anatomy 0.000 claims description 2
- 238000012258 culturing Methods 0.000 claims description 2
- 229960001145 deflazacort Drugs 0.000 claims description 2
- FBHSPRKOSMHSIF-GRMWVWQJSA-N deflazacort Chemical compound C1CC2=CC(=O)C=C[C@]2(C)[C@@H]2[C@@H]1[C@@H]1C[C@H]3OC(C)=N[C@@]3(C(=O)COC(=O)C)[C@@]1(C)C[C@@H]2O FBHSPRKOSMHSIF-GRMWVWQJSA-N 0.000 claims description 2
- 229950002185 edasalonexent Drugs 0.000 claims description 2
- 210000004408 hybridoma Anatomy 0.000 claims description 2
- 230000004054 inflammatory process Effects 0.000 claims description 2
- 230000002401 inhibitory effect Effects 0.000 claims description 2
- 238000007913 intrathecal administration Methods 0.000 claims description 2
- 230000000921 morphogenic effect Effects 0.000 claims description 2
- JQLBBYLGWHUHRW-KUBAVDMBSA-N n-[2-[[(4z,7z,10z,13z,16z,19z)-docosa-4,7,10,13,16,19-hexaenoyl]amino]ethyl]-2-hydroxybenzamide Chemical compound CC\C=C/C\C=C/C\C=C/C\C=C/C\C=C/C\C=C/CCC(=O)NCCNC(=O)C1=CC=CC=C1O JQLBBYLGWHUHRW-KUBAVDMBSA-N 0.000 claims description 2
- 229950003481 pamrevlumab Drugs 0.000 claims description 2
- 102220311820 rs1376867262 Human genes 0.000 claims description 2
- 102220269398 rs1555403432 Human genes 0.000 claims description 2
- 102220077293 rs193922450 Human genes 0.000 claims description 2
- 230000007103 stamina Effects 0.000 claims description 2
- 208000008817 Myofibroma Diseases 0.000 claims 2
- 102220605043 Histone H4-like protein type G_K76A_mutation Human genes 0.000 claims 1
- 206010039710 Scleroderma Diseases 0.000 claims 1
- 102200058951 rs17560 Human genes 0.000 claims 1
- 102200065571 rs762020512 Human genes 0.000 claims 1
- 235000018102 proteins Nutrition 0.000 description 159
- 235000001014 amino acid Nutrition 0.000 description 116
- 229940024606 amino acid Drugs 0.000 description 109
- 108010040856 glutamyl-cysteinyl-alanine Proteins 0.000 description 67
- 201000010099 disease Diseases 0.000 description 66
- KOSRFJWDECSPRO-UHFFFAOYSA-N alpha-L-glutamyl-L-glutamic acid Natural products OC(=O)CCC(N)C(=O)NC(CCC(O)=O)C(O)=O KOSRFJWDECSPRO-UHFFFAOYSA-N 0.000 description 64
- 108010055341 glutamyl-glutamic acid Proteins 0.000 description 64
- WHUUTDBJXJRKMK-UHFFFAOYSA-N Glutamic acid Natural products OC(=O)C(N)CCC(O)=O WHUUTDBJXJRKMK-UHFFFAOYSA-N 0.000 description 41
- SITLTJHOQZFJGG-UHFFFAOYSA-N N-L-alpha-glutamyl-L-valine Natural products CC(C)C(C(O)=O)NC(=O)C(N)CCC(O)=O SITLTJHOQZFJGG-UHFFFAOYSA-N 0.000 description 40
- BEZTUFWTPVOROW-KJEVXHAQSA-N Thr-Tyr-Arg Chemical compound C[C@H]([C@@H](C(=O)N[C@@H](CC1=CC=C(C=C1)O)C(=O)N[C@@H](CCCN=C(N)N)C(=O)O)N)O BEZTUFWTPVOROW-KJEVXHAQSA-N 0.000 description 35
- BZDGLJPROOOUOZ-XGEHTFHBSA-N Val-Thr-Cys Chemical compound C[C@H]([C@@H](C(=O)N[C@@H](CS)C(=O)O)NC(=O)[C@H](C(C)C)N)O BZDGLJPROOOUOZ-XGEHTFHBSA-N 0.000 description 35
- 108010050848 glycylleucine Proteins 0.000 description 35
- VUTHNLMCXKLLFI-LAEOZQHASA-N Val-Asp-Gln Chemical compound CC(C)[C@@H](C(=O)N[C@@H](CC(=O)O)C(=O)N[C@@H](CCC(=O)N)C(=O)O)N VUTHNLMCXKLLFI-LAEOZQHASA-N 0.000 description 34
- WRDANSJTFOHBPI-FXQIFTODSA-N Ala-Arg-Cys Chemical compound C[C@@H](C(=O)N[C@@H](CCCN=C(N)N)C(=O)N[C@@H](CS)C(=O)O)N WRDANSJTFOHBPI-FXQIFTODSA-N 0.000 description 33
- BGGAIXWIZCIFSG-XDTLVQLUSA-N Ala-Tyr-Glu Chemical compound [H]N[C@@H](C)C(=O)N[C@@H](CC1=CC=C(O)C=C1)C(=O)N[C@@H](CCC(O)=O)C(O)=O BGGAIXWIZCIFSG-XDTLVQLUSA-N 0.000 description 33
- PIWWUBYJNONVTJ-ZLUOBGJFSA-N Asn-Asp-Asn Chemical compound C([C@@H](C(=O)N[C@@H](CC(=O)O)C(=O)N[C@@H](CC(=O)N)C(=O)O)N)C(=O)N PIWWUBYJNONVTJ-ZLUOBGJFSA-N 0.000 description 33
- ZMWDUIIACVLIHK-GHCJXIJMSA-N Asn-Cys-Ile Chemical compound CC[C@H](C)[C@@H](C(=O)O)NC(=O)[C@H](CS)NC(=O)[C@H](CC(=O)N)N ZMWDUIIACVLIHK-GHCJXIJMSA-N 0.000 description 33
- DDPXDCKYWDGZAL-BQBZGAKWSA-N Asn-Gly-Arg Chemical compound NC(=O)C[C@H](N)C(=O)NCC(=O)N[C@H](C(O)=O)CCCN=C(N)N DDPXDCKYWDGZAL-BQBZGAKWSA-N 0.000 description 33
- JZLFYAAGGYMRIK-BYULHYEWSA-N Asn-Val-Asp Chemical compound [H]N[C@@H](CC(N)=O)C(=O)N[C@@H](C(C)C)C(=O)N[C@@H](CC(O)=O)C(O)=O JZLFYAAGGYMRIK-BYULHYEWSA-N 0.000 description 33
- ATPDEYTYWVMINF-ZLUOBGJFSA-N Cys-Cys-Ser Chemical compound [H]N[C@@H](CS)C(=O)N[C@@H](CS)C(=O)N[C@@H](CO)C(O)=O ATPDEYTYWVMINF-ZLUOBGJFSA-N 0.000 description 33
- SFRQEQGPRTVDPO-NRPADANISA-N Cys-Gln-Val Chemical compound [H]N[C@@H](CS)C(=O)N[C@@H](CCC(N)=O)C(=O)N[C@@H](C(C)C)C(O)=O SFRQEQGPRTVDPO-NRPADANISA-N 0.000 description 33
- SKSJPIBFNFPTJB-NKWVEPMBSA-N Cys-Gly-Pro Chemical compound C1C[C@@H](N(C1)C(=O)CNC(=O)[C@H](CS)N)C(=O)O SKSJPIBFNFPTJB-NKWVEPMBSA-N 0.000 description 33
- RRJOQIBQVZDVCW-SRVKXCTJSA-N Cys-His-Leu Chemical compound CC(C)C[C@@H](C(=O)O)NC(=O)[C@H](CC1=CN=CN1)NC(=O)[C@H](CS)N RRJOQIBQVZDVCW-SRVKXCTJSA-N 0.000 description 33
- KVCJEMHFLGVINV-ZLUOBGJFSA-N Cys-Ser-Asn Chemical compound SC[C@H](N)C(=O)N[C@@H](CO)C(=O)N[C@H](C(O)=O)CC(N)=O KVCJEMHFLGVINV-ZLUOBGJFSA-N 0.000 description 33
- ALNKNYKSZPSLBD-ZDLURKLDSA-N Cys-Thr-Gly Chemical compound [H]N[C@@H](CS)C(=O)N[C@@H]([C@@H](C)O)C(=O)NCC(O)=O ALNKNYKSZPSLBD-ZDLURKLDSA-N 0.000 description 33
- MLZRSFQRBDNJON-GUBZILKMSA-N Gln-Ala-Lys Chemical compound C[C@@H](C(=O)N[C@@H](CCCCN)C(=O)O)NC(=O)[C@H](CCC(=O)N)N MLZRSFQRBDNJON-GUBZILKMSA-N 0.000 description 33
- WATXSTJXNBOHKD-LAEOZQHASA-N Glu-Asp-Val Chemical compound [H]N[C@@H](CCC(O)=O)C(=O)N[C@@H](CC(O)=O)C(=O)N[C@@H](C(C)C)C(O)=O WATXSTJXNBOHKD-LAEOZQHASA-N 0.000 description 33
- FBEJIDRSQCGFJI-GUBZILKMSA-N Glu-Leu-Ser Chemical compound [H]N[C@@H](CCC(O)=O)C(=O)N[C@@H](CC(C)C)C(=O)N[C@@H](CO)C(O)=O FBEJIDRSQCGFJI-GUBZILKMSA-N 0.000 description 33
- QXPRJQPCFXMCIY-NKWVEPMBSA-N Gly-Ala-Pro Chemical compound C[C@@H](C(=O)N1CCC[C@@H]1C(=O)O)NC(=O)CN QXPRJQPCFXMCIY-NKWVEPMBSA-N 0.000 description 33
- PMGDADKJMCOXHX-UHFFFAOYSA-N L-Arginyl-L-glutamin-acetat Natural products NC(=N)NCCCC(N)C(=O)NC(CCC(N)=O)C(O)=O PMGDADKJMCOXHX-UHFFFAOYSA-N 0.000 description 33
- FADYJNXDPBKVCA-UHFFFAOYSA-N L-Phenylalanyl-L-lysin Natural products NCCCCC(C(O)=O)NC(=O)C(N)CC1=CC=CC=C1 FADYJNXDPBKVCA-UHFFFAOYSA-N 0.000 description 33
- LXKNSJLSGPNHSK-KKUMJFAQSA-N Leu-Leu-Lys Chemical compound CC(C)C[C@@H](C(=O)N[C@@H](CC(C)C)C(=O)N[C@@H](CCCCN)C(=O)O)N LXKNSJLSGPNHSK-KKUMJFAQSA-N 0.000 description 33
- GJJQCBVRWDGLMQ-GUBZILKMSA-N Lys-Glu-Ala Chemical compound [H]N[C@@H](CCCCN)C(=O)N[C@@H](CCC(O)=O)C(=O)N[C@@H](C)C(O)=O GJJQCBVRWDGLMQ-GUBZILKMSA-N 0.000 description 33
- GRADYHMSAUIKPS-DCAQKATOSA-N Lys-Glu-Gln Chemical compound [H]N[C@@H](CCCCN)C(=O)N[C@@H](CCC(O)=O)C(=O)N[C@@H](CCC(N)=O)C(O)=O GRADYHMSAUIKPS-DCAQKATOSA-N 0.000 description 33
- PBIPLDMFHAICIP-DCAQKATOSA-N Lys-Glu-Glu Chemical compound [H]N[C@@H](CCCCN)C(=O)N[C@@H](CCC(O)=O)C(=O)N[C@@H](CCC(O)=O)C(O)=O PBIPLDMFHAICIP-DCAQKATOSA-N 0.000 description 33
- YBAFDPFAUTYYRW-UHFFFAOYSA-N N-L-alpha-glutamyl-L-leucine Natural products CC(C)CC(C(O)=O)NC(=O)C(N)CCC(O)=O YBAFDPFAUTYYRW-UHFFFAOYSA-N 0.000 description 33
- 108010047562 NGR peptide Proteins 0.000 description 33
- 101100342977 Neurospora crassa (strain ATCC 24698 / 74-OR23-1A / CBS 708.71 / DSM 1257 / FGSC 987) leu-1 gene Proteins 0.000 description 33
- HHOOEUSPFGPZFP-QWRGUYRKSA-N Phe-Asn-Gly Chemical compound [H]N[C@@H](CC1=CC=CC=C1)C(=O)N[C@@H](CC(N)=O)C(=O)NCC(O)=O HHOOEUSPFGPZFP-QWRGUYRKSA-N 0.000 description 33
- OLTFZQIYCNOBLI-DCAQKATOSA-N Pro-Cys-Lys Chemical compound C1C[C@H](NC1)C(=O)N[C@@H](CS)C(=O)N[C@@H](CCCCN)C(=O)O OLTFZQIYCNOBLI-DCAQKATOSA-N 0.000 description 33
- NXEYSLRNNPWCRN-SRVKXCTJSA-N Pro-Glu-Leu Chemical compound [H]N1CCC[C@H]1C(=O)N[C@@H](CCC(O)=O)C(=O)N[C@@H](CC(C)C)C(O)=O NXEYSLRNNPWCRN-SRVKXCTJSA-N 0.000 description 33
- KCRQEJSKXAIULJ-FJXKBIBVSA-N Thr-Gly-Arg Chemical compound [H]N[C@@H]([C@@H](C)O)C(=O)NCC(=O)N[C@@H](CCCNC(N)=N)C(O)=O KCRQEJSKXAIULJ-FJXKBIBVSA-N 0.000 description 33
- PRNGXSILMXSWQQ-OEAJRASXSA-N Thr-Leu-Phe Chemical compound [H]N[C@@H]([C@@H](C)O)C(=O)N[C@@H](CC(C)C)C(=O)N[C@@H](CC1=CC=CC=C1)C(O)=O PRNGXSILMXSWQQ-OEAJRASXSA-N 0.000 description 33
- AIISTODACBDQLW-WDSOQIARSA-N Trp-Leu-Arg Chemical compound C1=CC=C2C(C[C@H](N)C(=O)N[C@@H](CC(C)C)C(=O)N[C@@H](CCCN=C(N)N)C(O)=O)=CNC2=C1 AIISTODACBDQLW-WDSOQIARSA-N 0.000 description 33
- DTPWXZXGFAHEKL-NWLDYVSISA-N Trp-Thr-Glu Chemical compound [H]N[C@@H](CC1=CNC2=C1C=CC=C2)C(=O)N[C@@H]([C@@H](C)O)C(=O)N[C@@H](CCC(O)=O)C(O)=O DTPWXZXGFAHEKL-NWLDYVSISA-N 0.000 description 33
- 108010008355 arginyl-glutamine Proteins 0.000 description 33
- 108010063718 gamma-glutamylaspartic acid Proteins 0.000 description 33
- 108010009298 lysylglutamic acid Proteins 0.000 description 33
- FQNILRVJOJBFFC-FXQIFTODSA-N Ala-Pro-Asp Chemical compound C[C@@H](C(=O)N1CCC[C@H]1C(=O)N[C@@H](CC(=O)O)C(=O)O)N FQNILRVJOJBFFC-FXQIFTODSA-N 0.000 description 32
- MSWSRLGNLKHDEI-ACZMJKKPSA-N Ala-Ser-Glu Chemical compound [H]N[C@@H](C)C(=O)N[C@@H](CO)C(=O)N[C@@H](CCC(O)=O)C(O)=O MSWSRLGNLKHDEI-ACZMJKKPSA-N 0.000 description 32
- CREYEAPXISDKSB-FQPOAREZSA-N Ala-Thr-Tyr Chemical compound [H]N[C@@H](C)C(=O)N[C@@H]([C@@H](C)O)C(=O)N[C@@H](CC1=CC=C(O)C=C1)C(O)=O CREYEAPXISDKSB-FQPOAREZSA-N 0.000 description 32
- KLKARCOHVHLAJP-UWJYBYFXSA-N Ala-Tyr-Cys Chemical compound C[C@H](N)C(=O)N[C@@H](Cc1ccc(O)cc1)C(=O)N[C@@H](CS)C(O)=O KLKARCOHVHLAJP-UWJYBYFXSA-N 0.000 description 32
- XSGBIBGAMKTHMY-WHFBIAKZSA-N Asn-Asp-Gly Chemical compound [H]N[C@@H](CC(N)=O)C(=O)N[C@@H](CC(O)=O)C(=O)NCC(O)=O XSGBIBGAMKTHMY-WHFBIAKZSA-N 0.000 description 32
- PZXPWHFYZXTFBI-YUMQZZPRSA-N Asp-Gly-Lys Chemical compound NCCCC[C@@H](C(O)=O)NC(=O)CNC(=O)[C@@H](N)CC(O)=O PZXPWHFYZXTFBI-YUMQZZPRSA-N 0.000 description 32
- QNFRBNZGVVKBNJ-PEFMBERDSA-N Asp-Ile-Gln Chemical compound CC[C@H](C)[C@@H](C(=O)N[C@@H](CCC(=O)N)C(=O)O)NC(=O)[C@H](CC(=O)O)N QNFRBNZGVVKBNJ-PEFMBERDSA-N 0.000 description 32
- SFJUYBCDQBAYAJ-YDHLFZDLSA-N Asp-Val-Phe Chemical compound OC(=O)C[C@H](N)C(=O)N[C@@H](C(C)C)C(=O)N[C@H](C(O)=O)CC1=CC=CC=C1 SFJUYBCDQBAYAJ-YDHLFZDLSA-N 0.000 description 32
- BGIRVSMUAJMGOK-FXQIFTODSA-N Cys-Ala-Met Chemical compound C[C@@H](C(=O)N[C@@H](CCSC)C(=O)O)NC(=O)[C@H](CS)N BGIRVSMUAJMGOK-FXQIFTODSA-N 0.000 description 32
- LBOLGUYQEPZSKM-YUMQZZPRSA-N Cys-Gly-Leu Chemical compound CC(C)C[C@@H](C(=O)O)NC(=O)CNC(=O)[C@H](CS)N LBOLGUYQEPZSKM-YUMQZZPRSA-N 0.000 description 32
- VAIWPXWHWAPYDF-FXQIFTODSA-N Glu-Asp-Gln Chemical compound [H]N[C@@H](CCC(O)=O)C(=O)N[C@@H](CC(O)=O)C(=O)N[C@@H](CCC(N)=O)C(O)=O VAIWPXWHWAPYDF-FXQIFTODSA-N 0.000 description 32
- BUZMZDDKFCSKOT-CIUDSAMLSA-N Glu-Glu-Glu Chemical compound OC(=O)CC[C@H](N)C(=O)N[C@@H](CCC(O)=O)C(=O)N[C@@H](CCC(O)=O)C(O)=O BUZMZDDKFCSKOT-CIUDSAMLSA-N 0.000 description 32
- BMWFDYIYBAFROD-WPRPVWTQSA-N Gly-Pro-Val Chemical compound CC(C)[C@@H](C(O)=O)NC(=O)[C@@H]1CCCN1C(=O)CN BMWFDYIYBAFROD-WPRPVWTQSA-N 0.000 description 32
- WSXTWLJHTLRFLW-SRVKXCTJSA-N Lys-Ala-Lys Chemical compound NCCCC[C@H](N)C(=O)N[C@@H](C)C(=O)N[C@@H](CCCCN)C(O)=O WSXTWLJHTLRFLW-SRVKXCTJSA-N 0.000 description 32
- AHFOKDZWPPGJAZ-SRVKXCTJSA-N Lys-Lys-Cys Chemical compound C(CCN)C[C@@H](C(=O)N[C@@H](CCCCN)C(=O)N[C@@H](CS)C(=O)O)N AHFOKDZWPPGJAZ-SRVKXCTJSA-N 0.000 description 32
- STGVYUTZKGPRCI-GUBZILKMSA-N Pro-Val-Cys Chemical compound SC[C@@H](C(O)=O)NC(=O)[C@H](C(C)C)NC(=O)[C@@H]1CCCN1 STGVYUTZKGPRCI-GUBZILKMSA-N 0.000 description 32
- 108010079005 RDV peptide Proteins 0.000 description 32
- WTPKKLMBNBCCNL-ACZMJKKPSA-N Ser-Cys-Glu Chemical compound C(CC(=O)O)[C@@H](C(=O)O)NC(=O)[C@H](CS)NC(=O)[C@H](CO)N WTPKKLMBNBCCNL-ACZMJKKPSA-N 0.000 description 32
- HHJFMHQYEAAOBM-ZLUOBGJFSA-N Ser-Ser-Ala Chemical compound [H]N[C@@H](CO)C(=O)N[C@@H](CO)C(=O)N[C@@H](C)C(O)=O HHJFMHQYEAAOBM-ZLUOBGJFSA-N 0.000 description 32
- SOACHCFYJMCMHC-BWBBJGPYSA-N Ser-Thr-Cys Chemical compound C[C@H]([C@@H](C(=O)N[C@@H](CS)C(=O)O)NC(=O)[C@H](CO)N)O SOACHCFYJMCMHC-BWBBJGPYSA-N 0.000 description 32
- VIBXMCZWVUOZLA-OLHMAJIHSA-N Thr-Asn-Asn Chemical compound C[C@H]([C@@H](C(=O)N[C@@H](CC(=O)N)C(=O)N[C@@H](CC(=O)N)C(=O)O)N)O VIBXMCZWVUOZLA-OLHMAJIHSA-N 0.000 description 32
- SINRIKQYQJRGDQ-MEYUZBJRSA-N Tyr-Lys-Thr Chemical compound C[C@@H](O)[C@@H](C(O)=O)NC(=O)[C@H](CCCCN)NC(=O)[C@@H](N)CC1=CC=C(O)C=C1 SINRIKQYQJRGDQ-MEYUZBJRSA-N 0.000 description 32
- YLBNZCJFSVJDRJ-KJEVXHAQSA-N Val-Thr-Tyr Chemical compound CC(C)[C@H](N)C(=O)N[C@@H]([C@@H](C)O)C(=O)N[C@@H](Cc1ccc(O)cc1)C(O)=O YLBNZCJFSVJDRJ-KJEVXHAQSA-N 0.000 description 32
- JVMKBJNSRZWDBO-FXQIFTODSA-N Arg-Cys-Ser Chemical compound [H]N[C@@H](CCCNC(N)=N)C(=O)N[C@@H](CS)C(=O)N[C@@H](CO)C(O)=O JVMKBJNSRZWDBO-FXQIFTODSA-N 0.000 description 31
- MEFGKQUUYZOLHM-GMOBBJLQSA-N Asn-Arg-Ile Chemical compound [H]N[C@@H](CC(N)=O)C(=O)N[C@@H](CCCNC(N)=N)C(=O)N[C@@H]([C@@H](C)CC)C(O)=O MEFGKQUUYZOLHM-GMOBBJLQSA-N 0.000 description 31
- VNXQRBXEQXLERQ-CIUDSAMLSA-N Asp-Ser-Lys Chemical compound C(CCN)C[C@@H](C(=O)O)NC(=O)[C@H](CO)NC(=O)[C@H](CC(=O)O)N VNXQRBXEQXLERQ-CIUDSAMLSA-N 0.000 description 31
- WVLZTXGTNGHPBO-SRVKXCTJSA-N Cys-Leu-Leu Chemical compound [H]N[C@@H](CS)C(=O)N[C@@H](CC(C)C)C(=O)N[C@@H](CC(C)C)C(O)=O WVLZTXGTNGHPBO-SRVKXCTJSA-N 0.000 description 31
- KGIHMGPYGXBYJJ-SRVKXCTJSA-N Cys-Lys-Lys Chemical compound NCCCC[C@@H](C(O)=O)NC(=O)[C@H](CCCCN)NC(=O)[C@@H](N)CS KGIHMGPYGXBYJJ-SRVKXCTJSA-N 0.000 description 31
- MBRWOKXNHTUJMB-CIUDSAMLSA-N Cys-Pro-Glu Chemical compound [H]N[C@@H](CS)C(=O)N1CCC[C@H]1C(=O)N[C@@H](CCC(O)=O)C(O)=O MBRWOKXNHTUJMB-CIUDSAMLSA-N 0.000 description 31
- IKFZXRLDMYWNBU-YUMQZZPRSA-N Gln-Gly-Arg Chemical compound NC(=O)CC[C@H](N)C(=O)NCC(=O)N[C@H](C(O)=O)CCCN=C(N)N IKFZXRLDMYWNBU-YUMQZZPRSA-N 0.000 description 31
- SBCYJMOOHUDWDA-NUMRIWBASA-N Glu-Asp-Thr Chemical compound [H]N[C@@H](CCC(O)=O)C(=O)N[C@@H](CC(O)=O)C(=O)N[C@@H]([C@@H](C)O)C(O)=O SBCYJMOOHUDWDA-NUMRIWBASA-N 0.000 description 31
- SAEBUDRWKUXLOM-ACZMJKKPSA-N Glu-Cys-Ala Chemical compound OC(=O)[C@H](C)NC(=O)[C@H](CS)NC(=O)[C@@H](N)CCC(O)=O SAEBUDRWKUXLOM-ACZMJKKPSA-N 0.000 description 31
- NKLRYVLERDYDBI-FXQIFTODSA-N Glu-Glu-Asp Chemical compound OC(=O)CC[C@H](N)C(=O)N[C@@H](CCC(O)=O)C(=O)N[C@@H](CC(O)=O)C(O)=O NKLRYVLERDYDBI-FXQIFTODSA-N 0.000 description 31
- RJIVPOXLQFJRTG-LURJTMIESA-N Gly-Arg-Gly Chemical compound OC(=O)CNC(=O)[C@@H](NC(=O)CN)CCCN=C(N)N RJIVPOXLQFJRTG-LURJTMIESA-N 0.000 description 31
- GWCRIHNSVMOBEQ-BQBZGAKWSA-N Gly-Arg-Ser Chemical compound [H]NCC(=O)N[C@@H](CCCNC(N)=N)C(=O)N[C@@H](CO)C(O)=O GWCRIHNSVMOBEQ-BQBZGAKWSA-N 0.000 description 31
- DJTXYXZNNDDEOU-WHFBIAKZSA-N Gly-Asn-Cys Chemical compound C([C@@H](C(=O)N[C@@H](CS)C(=O)O)NC(=O)CN)C(=O)N DJTXYXZNNDDEOU-WHFBIAKZSA-N 0.000 description 31
- NYEYYMLUABXDMC-NHCYSSNCSA-N Ile-Gly-Leu Chemical compound CC[C@H](C)[C@@H](C(=O)NCC(=O)N[C@@H](CC(C)C)C(=O)O)N NYEYYMLUABXDMC-NHCYSSNCSA-N 0.000 description 31
- HUEBCHPSXSQUGN-GARJFASQSA-N Leu-Cys-Pro Chemical compound CC(C)C[C@@H](C(=O)N[C@@H](CS)C(=O)N1CCC[C@@H]1C(=O)O)N HUEBCHPSXSQUGN-GARJFASQSA-N 0.000 description 31
- LSLUTXRANSUGFY-XIRDDKMYSA-N Leu-Trp-Asp Chemical compound [H]N[C@@H](CC(C)C)C(=O)N[C@@H](CC1=CNC2=C1C=CC=C2)C(=O)N[C@@H](CC(O)=O)C(O)=O LSLUTXRANSUGFY-XIRDDKMYSA-N 0.000 description 31
- GPSMLZQVIIYLDK-ULQDDVLXSA-N Phe-Lys-Val Chemical compound [H]N[C@@H](CC1=CC=CC=C1)C(=O)N[C@@H](CCCCN)C(=O)N[C@@H](C(C)C)C(O)=O GPSMLZQVIIYLDK-ULQDDVLXSA-N 0.000 description 31
- FTVRVZNYIYWJGB-ACZMJKKPSA-N Ser-Asp-Glu Chemical compound [H]N[C@@H](CO)C(=O)N[C@@H](CC(O)=O)C(=O)N[C@@H](CCC(O)=O)C(O)=O FTVRVZNYIYWJGB-ACZMJKKPSA-N 0.000 description 31
- YQQKYAZABFEYAF-FXQIFTODSA-N Ser-Glu-Gln Chemical compound [H]N[C@@H](CO)C(=O)N[C@@H](CCC(O)=O)C(=O)N[C@@H](CCC(N)=O)C(O)=O YQQKYAZABFEYAF-FXQIFTODSA-N 0.000 description 31
- SNXUIBACCONSOH-BWBBJGPYSA-N Ser-Thr-Ser Chemical compound OC[C@H](N)C(=O)N[C@@H]([C@H](O)C)C(=O)N[C@@H](CO)C(O)=O SNXUIBACCONSOH-BWBBJGPYSA-N 0.000 description 31
- NRUPKQSXTJNQGD-XGEHTFHBSA-N Thr-Cys-Arg Chemical compound [H]N[C@@H]([C@@H](C)O)C(=O)N[C@@H](CS)C(=O)N[C@@H](CCCNC(N)=N)C(O)=O NRUPKQSXTJNQGD-XGEHTFHBSA-N 0.000 description 31
- RIKLKPANMFNREP-FDARSICLSA-N Trp-Met-Ile Chemical compound C1=CC=C2C(C[C@H](N)C(=O)N[C@@H](CCSC)C(=O)N[C@@H]([C@@H](C)CC)C(O)=O)=CNC2=C1 RIKLKPANMFNREP-FDARSICLSA-N 0.000 description 31
- 108010069205 aspartyl-phenylalanine Proteins 0.000 description 31
- 108010027338 isoleucylcysteine Proteins 0.000 description 31
- NXQCSPVUPLUTJH-WHFBIAKZSA-N Cys-Ser-Gly Chemical compound SC[C@H](N)C(=O)N[C@@H](CO)C(=O)NCC(O)=O NXQCSPVUPLUTJH-WHFBIAKZSA-N 0.000 description 30
- MQQLYEHXSBJTRK-FXQIFTODSA-N Cys-Val-Cys Chemical compound CC(C)[C@@H](C(=O)N[C@@H](CS)C(=O)O)NC(=O)[C@H](CS)N MQQLYEHXSBJTRK-FXQIFTODSA-N 0.000 description 30
- IASQBRJGRVXNJI-YUMQZZPRSA-N Leu-Cys-Gly Chemical compound [H]N[C@@H](CC(C)C)C(=O)N[C@@H](CS)C(=O)NCC(O)=O IASQBRJGRVXNJI-YUMQZZPRSA-N 0.000 description 30
- QNBVTHNJGCOVFA-AVGNSLFASA-N Leu-Leu-Glu Chemical compound CC(C)C[C@H](N)C(=O)N[C@@H](CC(C)C)C(=O)N[C@H](C(O)=O)CCC(O)=O QNBVTHNJGCOVFA-AVGNSLFASA-N 0.000 description 30
- KNKHAVVBVXKOGX-JXUBOQSCSA-N Lys-Ala-Thr Chemical compound [H]N[C@@H](CCCCN)C(=O)N[C@@H](C)C(=O)N[C@@H]([C@@H](C)O)C(O)=O KNKHAVVBVXKOGX-JXUBOQSCSA-N 0.000 description 30
- SFQPJNQDUUYCLA-BJDJZHNGSA-N Lys-Cys-Ile Chemical compound CC[C@H](C)[C@@H](C(=O)O)NC(=O)[C@H](CS)NC(=O)[C@H](CCCCN)N SFQPJNQDUUYCLA-BJDJZHNGSA-N 0.000 description 30
- XQLBWXHVZVBNJM-FXQIFTODSA-N Pro-Ala-Ser Chemical compound OC[C@@H](C(O)=O)NC(=O)[C@H](C)NC(=O)[C@@H]1CCCN1 XQLBWXHVZVBNJM-FXQIFTODSA-N 0.000 description 30
- DXTOOBDIIAJZBJ-BQBZGAKWSA-N Pro-Gly-Ser Chemical compound [H]N1CCC[C@H]1C(=O)NCC(=O)N[C@@H](CO)C(O)=O DXTOOBDIIAJZBJ-BQBZGAKWSA-N 0.000 description 30
- OHKLFYXEOGGGCK-ZLUOBGJFSA-N Ser-Asp-Asn Chemical compound [H]N[C@@H](CO)C(=O)N[C@@H](CC(O)=O)C(=O)N[C@@H](CC(N)=O)C(O)=O OHKLFYXEOGGGCK-ZLUOBGJFSA-N 0.000 description 30
- UIGMAMGZOJVTDN-WHFBIAKZSA-N Ser-Gly-Ser Chemical compound OC[C@H](N)C(=O)NCC(=O)N[C@@H](CO)C(O)=O UIGMAMGZOJVTDN-WHFBIAKZSA-N 0.000 description 30
- MYNYCUXMIIWUNW-IEGACIPQSA-N Thr-Trp-Lys Chemical compound C[C@H]([C@@H](C(=O)N[C@@H](CC1=CNC2=CC=CC=C21)C(=O)N[C@@H](CCCCN)C(=O)O)N)O MYNYCUXMIIWUNW-IEGACIPQSA-N 0.000 description 30
- MLADEWAIYAPAAU-IHRRRGAJSA-N Val-Lys-His Chemical compound CC(C)[C@@H](C(=O)N[C@@H](CCCCN)C(=O)N[C@@H](CC1=CN=CN1)C(=O)O)N MLADEWAIYAPAAU-IHRRRGAJSA-N 0.000 description 30
- 108010037850 glycylvaline Proteins 0.000 description 30
- 108010018006 histidylserine Proteins 0.000 description 30
- 108010064235 lysylglycine Proteins 0.000 description 30
- 108010029020 prolylglycine Proteins 0.000 description 30
- GSNRZJNHMVMOFV-ACZMJKKPSA-N Cys-Asp-Glu Chemical compound C(CC(=O)O)[C@@H](C(=O)O)NC(=O)[C@H](CC(=O)O)NC(=O)[C@H](CS)N GSNRZJNHMVMOFV-ACZMJKKPSA-N 0.000 description 29
- UIPXCLNLUUAMJU-JBDRJPRFSA-N Ser-Ile-Ser Chemical compound [H]N[C@@H](CO)C(=O)N[C@@H]([C@@H](C)CC)C(=O)N[C@@H](CO)C(O)=O UIPXCLNLUUAMJU-JBDRJPRFSA-N 0.000 description 29
- DHPPWTOLRWYIDS-XKBZYTNZSA-N Thr-Cys-Glu Chemical compound [H]N[C@@H]([C@@H](C)O)C(=O)N[C@@H](CS)C(=O)N[C@@H](CCC(O)=O)C(O)=O DHPPWTOLRWYIDS-XKBZYTNZSA-N 0.000 description 29
- AAOPYWQQBXHINJ-DZKIICNBSA-N Val-Gln-Tyr Chemical compound CC(C)[C@@H](C(=O)N[C@@H](CCC(=O)N)C(=O)N[C@@H](CC1=CC=C(C=C1)O)C(=O)O)N AAOPYWQQBXHINJ-DZKIICNBSA-N 0.000 description 29
- HRHYJNLMIJWGLF-BZSNNMDCSA-N Tyr-Ser-Phe Chemical compound C([C@H](N)C(=O)N[C@@H](CO)C(=O)N[C@@H](CC=1C=CC=CC=1)C(O)=O)C1=CC=C(O)C=C1 HRHYJNLMIJWGLF-BZSNNMDCSA-N 0.000 description 28
- 108010050025 alpha-glutamyltryptophan Proteins 0.000 description 28
- UREQLMJCKFLLHM-NAKRPEOUSA-N Pro-Ile-Ser Chemical compound [H]N1CCC[C@H]1C(=O)N[C@@H]([C@@H](C)CC)C(=O)N[C@@H](CO)C(O)=O UREQLMJCKFLLHM-NAKRPEOUSA-N 0.000 description 27
- RIAKPZVSNBBNRE-BJDJZHNGSA-N Ser-Ile-Leu Chemical compound OC[C@H](N)C(=O)N[C@@H]([C@@H](C)CC)C(=O)N[C@@H](CC(C)C)C(O)=O RIAKPZVSNBBNRE-BJDJZHNGSA-N 0.000 description 27
- SURTWIXUHQNUGN-GUBZILKMSA-N Cys-Arg-Met Chemical compound CSCC[C@@H](C(=O)O)NC(=O)[C@H](CCCN=C(N)N)NC(=O)[C@H](CS)N SURTWIXUHQNUGN-GUBZILKMSA-N 0.000 description 24
- AFLBTVGQCQLOFJ-AVGNSLFASA-N Lys-Pro-Arg Chemical compound NCCCC[C@H](N)C(=O)N1CCC[C@H]1C(=O)N[C@@H](CCCN=C(N)N)C(O)=O AFLBTVGQCQLOFJ-AVGNSLFASA-N 0.000 description 22
- VEPBEGNDJYANCF-QWRGUYRKSA-N Gly-Lys-Lys Chemical compound NCCCC[C@@H](C(O)=O)NC(=O)[C@@H](NC(=O)CN)CCCCN VEPBEGNDJYANCF-QWRGUYRKSA-N 0.000 description 20
- 230000000694 effects Effects 0.000 description 18
- 241001465754 Metazoa Species 0.000 description 17
- 238000001727 in vivo Methods 0.000 description 17
- 210000001519 tissue Anatomy 0.000 description 16
- RIJCHEVHFWMDKD-SRVKXCTJSA-N Lys-Lys-Asn Chemical compound NCCCC[C@H](N)C(=O)N[C@@H](CCCCN)C(=O)N[C@@H](CC(N)=O)C(O)=O RIJCHEVHFWMDKD-SRVKXCTJSA-N 0.000 description 15
- 238000004458 analytical method Methods 0.000 description 15
- 239000000546 pharmaceutical excipient Substances 0.000 description 14
- 102000037280 Growth Differentiation Factor 2 Human genes 0.000 description 12
- 108010090290 Growth Differentiation Factor 2 Proteins 0.000 description 12
- 241000699670 Mus sp. Species 0.000 description 12
- FAPWRFPIFSIZLT-UHFFFAOYSA-M Sodium chloride Chemical compound [Na+].[Cl-] FAPWRFPIFSIZLT-UHFFFAOYSA-M 0.000 description 12
- 108010016616 cysteinylglycine Proteins 0.000 description 12
- 108020003175 receptors Proteins 0.000 description 12
- 108010069091 Dystrophin Proteins 0.000 description 11
- 241000880493 Leptailurus serval Species 0.000 description 11
- 230000037430 deletion Effects 0.000 description 11
- 238000012217 deletion Methods 0.000 description 11
- 150000007523 nucleic acids Chemical group 0.000 description 11
- 125000006850 spacer group Chemical group 0.000 description 11
- KDXKERNSBIXSRK-UHFFFAOYSA-N Lysine Natural products NCCCCC(N)C(O)=O KDXKERNSBIXSRK-UHFFFAOYSA-N 0.000 description 10
- 210000002966 serum Anatomy 0.000 description 10
- RWGDABDXVXRLLH-ACZMJKKPSA-N Cys-Glu-Asn Chemical compound C(CC(=O)O)[C@@H](C(=O)N[C@@H](CC(=O)N)C(=O)O)NC(=O)[C@H](CS)N RWGDABDXVXRLLH-ACZMJKKPSA-N 0.000 description 9
- 108010065920 Insulin Lispro Proteins 0.000 description 9
- IHCXPSYCHXFXKT-DCAQKATOSA-N Pro-Arg-Glu Chemical compound [H]N1CCC[C@H]1C(=O)N[C@@H](CCCNC(N)=N)C(=O)N[C@@H](CCC(O)=O)C(O)=O IHCXPSYCHXFXKT-DCAQKATOSA-N 0.000 description 9
- KXUKIBHIVRYOIP-ZKWXMUAHSA-N Val-Asp-Cys Chemical compound CC(C)[C@@H](C(=O)N[C@@H](CC(=O)O)C(=O)N[C@@H](CS)C(=O)O)N KXUKIBHIVRYOIP-ZKWXMUAHSA-N 0.000 description 9
- 108010060199 cysteinylproline Proteins 0.000 description 9
- 238000009472 formulation Methods 0.000 description 9
- 239000003446 ligand Substances 0.000 description 9
- 230000000670 limiting effect Effects 0.000 description 9
- 210000000689 upper leg Anatomy 0.000 description 9
- NSVOVKWEKGEOQB-LURJTMIESA-N Gly-Pro-Gly Chemical compound NCC(=O)N1CCC[C@H]1C(=O)NCC(O)=O NSVOVKWEKGEOQB-LURJTMIESA-N 0.000 description 8
- CGBYDGAJHSOGFQ-LPEHRKFASA-N Pro-Ala-Pro Chemical compound C[C@@H](C(=O)N1CCC[C@@H]1C(=O)O)NC(=O)[C@@H]2CCCN2 CGBYDGAJHSOGFQ-LPEHRKFASA-N 0.000 description 8
- OLHDPZMYUSBGDE-GUBZILKMSA-N Pro-Arg-Cys Chemical compound C1C[C@H](NC1)C(=O)N[C@@H](CCCN=C(N)N)C(=O)N[C@@H](CS)C(=O)O OLHDPZMYUSBGDE-GUBZILKMSA-N 0.000 description 8
- 108010008281 Recombinant Fusion Proteins Proteins 0.000 description 8
- 102000007056 Recombinant Fusion Proteins Human genes 0.000 description 8
- 230000001976 improved effect Effects 0.000 description 8
- 230000037431 insertion Effects 0.000 description 8
- 238000003780 insertion Methods 0.000 description 8
- 210000003141 lower extremity Anatomy 0.000 description 8
- 210000000056 organ Anatomy 0.000 description 8
- 239000000126 substance Substances 0.000 description 8
- PYZPXCZNQSEHDT-GUBZILKMSA-N Arg-Met-Asn Chemical compound CSCC[C@@H](C(=O)N[C@@H](CC(=O)N)C(=O)O)NC(=O)[C@H](CCCN=C(N)N)N PYZPXCZNQSEHDT-GUBZILKMSA-N 0.000 description 7
- 241000699666 Mus <mouse, genus> Species 0.000 description 7
- 210000004899 c-terminal region Anatomy 0.000 description 7
- 230000006870 function Effects 0.000 description 7
- 210000001087 myotubule Anatomy 0.000 description 7
- 239000012146 running buffer Substances 0.000 description 7
- 239000011780 sodium chloride Substances 0.000 description 7
- 210000003813 thumb Anatomy 0.000 description 7
- SNDBKTFJWVEVPO-WHFBIAKZSA-N Asp-Gly-Ser Chemical compound [H]N[C@@H](CC(O)=O)C(=O)NCC(=O)N[C@@H](CO)C(O)=O SNDBKTFJWVEVPO-WHFBIAKZSA-N 0.000 description 6
- 102000001039 Dystrophin Human genes 0.000 description 6
- TWYFJOHWGCCRIR-DCAQKATOSA-N Glu-Pro-Arg Chemical compound [H]N[C@@H](CCC(O)=O)C(=O)N1CCC[C@H]1C(=O)N[C@@H](CCCNC(N)=N)C(O)=O TWYFJOHWGCCRIR-DCAQKATOSA-N 0.000 description 6
- GMTXWRIDLGTVFC-IUCAKERBSA-N Gly-Lys-Glu Chemical compound [H]NCC(=O)N[C@@H](CCCCN)C(=O)N[C@@H](CCC(O)=O)C(O)=O GMTXWRIDLGTVFC-IUCAKERBSA-N 0.000 description 6
- 108060003951 Immunoglobulin Proteins 0.000 description 6
- AYFVYJQAPQTCCC-GBXIJSLDSA-N L-threonine Chemical compound C[C@@H](O)[C@H](N)C(O)=O AYFVYJQAPQTCCC-GBXIJSLDSA-N 0.000 description 6
- PVMPDMIKUVNOBD-CIUDSAMLSA-N Leu-Asp-Ser Chemical compound CC(C)C[C@H](N)C(=O)N[C@@H](CC(O)=O)C(=O)N[C@@H](CO)C(O)=O PVMPDMIKUVNOBD-CIUDSAMLSA-N 0.000 description 6
- ZXEUFAVXODIPHC-GUBZILKMSA-N Lys-Glu-Asn Chemical compound NCCCC[C@H](N)C(=O)N[C@@H](CCC(O)=O)C(=O)N[C@@H](CC(N)=O)C(O)=O ZXEUFAVXODIPHC-GUBZILKMSA-N 0.000 description 6
- 239000004472 Lysine Substances 0.000 description 6
- JDMKQHSHKJHAHR-UHFFFAOYSA-N Phe-Phe-Leu-Tyr Natural products C=1C=C(O)C=CC=1CC(C(O)=O)NC(=O)C(CC(C)C)NC(=O)C(NC(=O)C(N)CC=1C=CC=CC=1)CC1=CC=CC=C1 JDMKQHSHKJHAHR-UHFFFAOYSA-N 0.000 description 6
- XTDDIVQWDXMRJL-IHRRRGAJSA-N Val-Leu-His Chemical compound CC(C)C[C@@H](C(=O)N[C@@H](CC1=CN=CN1)C(=O)O)NC(=O)[C@H](C(C)C)N XTDDIVQWDXMRJL-IHRRRGAJSA-N 0.000 description 6
- SYSWVVCYSXBVJG-RHYQMDGZSA-N Val-Leu-Thr Chemical compound C[C@H]([C@@H](C(=O)O)NC(=O)[C@H](CC(C)C)NC(=O)[C@H](C(C)C)N)O SYSWVVCYSXBVJG-RHYQMDGZSA-N 0.000 description 6
- LLJLBRRXKZTTRD-GUBZILKMSA-N Val-Val-Ser Chemical compound CC(C)[C@@H](C(=O)N[C@@H](C(C)C)C(=O)N[C@@H](CO)C(=O)O)N LLJLBRRXKZTTRD-GUBZILKMSA-N 0.000 description 6
- 239000004480 active ingredient Substances 0.000 description 6
- 108010052670 arginyl-glutamyl-glutamic acid Proteins 0.000 description 6
- 210000003414 extremity Anatomy 0.000 description 6
- 230000004927 fusion Effects 0.000 description 6
- 102000018358 immunoglobulin Human genes 0.000 description 6
- 230000005764 inhibitory process Effects 0.000 description 6
- 108010073472 leucyl-prolyl-proline Proteins 0.000 description 6
- 108010017391 lysylvaline Proteins 0.000 description 6
- 102000039446 nucleic acids Human genes 0.000 description 6
- 108020004707 nucleic acids Proteins 0.000 description 6
- 108010048397 seryl-lysyl-leucine Proteins 0.000 description 6
- 230000019491 signal transduction Effects 0.000 description 6
- 230000000638 stimulation Effects 0.000 description 6
- 230000001225 therapeutic effect Effects 0.000 description 6
- 108010052774 valyl-lysyl-glycyl-phenylalanyl-tyrosine Proteins 0.000 description 6
- 239000003981 vehicle Substances 0.000 description 6
- XLYOFNOQVPJJNP-UHFFFAOYSA-N water Chemical compound O XLYOFNOQVPJJNP-UHFFFAOYSA-N 0.000 description 6
- 102000007469 Actins Human genes 0.000 description 5
- 108010085238 Actins Proteins 0.000 description 5
- 102000007350 Bone Morphogenetic Proteins Human genes 0.000 description 5
- 108010007726 Bone Morphogenetic Proteins Proteins 0.000 description 5
- 108091006020 Fc-tagged proteins Proteins 0.000 description 5
- DHNWZLGBTPUTQQ-QEJZJMRPSA-N Gln-Asp-Trp Chemical compound C1=CC=C2C(=C1)C(=CN2)C[C@@H](C(=O)O)NC(=O)[C@H](CC(=O)O)NC(=O)[C@H](CCC(=O)N)N DHNWZLGBTPUTQQ-QEJZJMRPSA-N 0.000 description 5
- HUFCEIHAFNVSNR-IHRRRGAJSA-N Glu-Gln-Tyr Chemical compound OC(=O)CC[C@H](N)C(=O)N[C@@H](CCC(N)=O)C(=O)N[C@H](C(O)=O)CC1=CC=C(O)C=C1 HUFCEIHAFNVSNR-IHRRRGAJSA-N 0.000 description 5
- DHMQDGOQFOQNFH-UHFFFAOYSA-N Glycine Chemical compound NCC(O)=O DHMQDGOQFOQNFH-UHFFFAOYSA-N 0.000 description 5
- CKLJMWTZIZZHCS-REOHCLBHSA-N L-aspartic acid Chemical compound OC(=O)[C@@H](N)CC(O)=O CKLJMWTZIZZHCS-REOHCLBHSA-N 0.000 description 5
- OIARJGNVARWKFP-YUMQZZPRSA-N Leu-Asn-Gly Chemical compound [H]N[C@@H](CC(C)C)C(=O)N[C@@H](CC(N)=O)C(=O)NCC(O)=O OIARJGNVARWKFP-YUMQZZPRSA-N 0.000 description 5
- AIQWYVFNBNNOLU-RHYQMDGZSA-N Leu-Thr-Val Chemical compound [H]N[C@@H](CC(C)C)C(=O)N[C@@H]([C@@H](C)O)C(=O)N[C@@H](C(C)C)C(O)=O AIQWYVFNBNNOLU-RHYQMDGZSA-N 0.000 description 5
- 108091028043 Nucleic acid sequence Proteins 0.000 description 5
- UAYHMOIGIQZLFR-NHCYSSNCSA-N Pro-Gln-Val Chemical compound [H]N1CCC[C@H]1C(=O)N[C@@H](CCC(N)=O)C(=O)N[C@@H](C(C)C)C(O)=O UAYHMOIGIQZLFR-NHCYSSNCSA-N 0.000 description 5
- NHOVZGFNTGMYMI-KKUMJFAQSA-N Tyr-Ser-Lys Chemical compound NCCCC[C@@H](C(O)=O)NC(=O)[C@H](CO)NC(=O)[C@@H](N)CC1=CC=C(O)C=C1 NHOVZGFNTGMYMI-KKUMJFAQSA-N 0.000 description 5
- KRAHMIJVUPUOTQ-DCAQKATOSA-N Val-Ser-His Chemical compound CC(C)[C@@H](C(=O)N[C@@H](CO)C(=O)N[C@@H](CC1=CN=CN1)C(=O)O)N KRAHMIJVUPUOTQ-DCAQKATOSA-N 0.000 description 5
- ZLNYBMWGPOKSLW-LSJOCFKGSA-N Val-Val-Asp Chemical compound CC(C)[C@H](N)C(=O)N[C@@H](C(C)C)C(=O)N[C@@H](CC(O)=O)C(O)=O ZLNYBMWGPOKSLW-LSJOCFKGSA-N 0.000 description 5
- 235000003704 aspartic acid Nutrition 0.000 description 5
- 108010047857 aspartylglycine Proteins 0.000 description 5
- 238000003556 assay Methods 0.000 description 5
- OQFSQFPPLPISGP-UHFFFAOYSA-N beta-carboxyaspartic acid Natural products OC(=O)C(N)C(C(O)=O)C(O)=O OQFSQFPPLPISGP-UHFFFAOYSA-N 0.000 description 5
- 230000004071 biological effect Effects 0.000 description 5
- 229940112869 bone morphogenetic protein Drugs 0.000 description 5
- 239000000969 carrier Substances 0.000 description 5
- 239000003085 diluting agent Substances 0.000 description 5
- 210000003194 forelimb Anatomy 0.000 description 5
- 235000013922 glutamic acid Nutrition 0.000 description 5
- 239000004220 glutamic acid Substances 0.000 description 5
- 108010013768 glutamyl-aspartyl-proline Proteins 0.000 description 5
- 210000002216 heart Anatomy 0.000 description 5
- 210000003292 kidney cell Anatomy 0.000 description 5
- 108010054155 lysyllysine Proteins 0.000 description 5
- 210000004962 mammalian cell Anatomy 0.000 description 5
- 210000004165 myocardium Anatomy 0.000 description 5
- 230000037361 pathway Effects 0.000 description 5
- 230000002265 prevention Effects 0.000 description 5
- 108010031719 prolyl-serine Proteins 0.000 description 5
- 150000003839 salts Chemical class 0.000 description 5
- 238000007910 systemic administration Methods 0.000 description 5
- 238000012360 testing method Methods 0.000 description 5
- 108010080629 tryptophan-leucine Proteins 0.000 description 5
- 210000001364 upper extremity Anatomy 0.000 description 5
- SUHLZMHFRALVSY-YUMQZZPRSA-N Ala-Lys-Gly Chemical compound NCCCC[C@H](NC(=O)[C@@H](N)C)C(=O)NCC(O)=O SUHLZMHFRALVSY-YUMQZZPRSA-N 0.000 description 4
- YJHKTAMKPGFJCT-NRPADANISA-N Ala-Val-Glu Chemical compound [H]N[C@@H](C)C(=O)N[C@@H](C(C)C)C(=O)N[C@@H](CCC(O)=O)C(O)=O YJHKTAMKPGFJCT-NRPADANISA-N 0.000 description 4
- OZNSCVPYWZRQPY-CIUDSAMLSA-N Arg-Asp-Glu Chemical compound [H]N[C@@H](CCCNC(N)=N)C(=O)N[C@@H](CC(O)=O)C(=O)N[C@@H](CCC(O)=O)C(O)=O OZNSCVPYWZRQPY-CIUDSAMLSA-N 0.000 description 4
- ZUVMUOOHJYNJPP-XIRDDKMYSA-N Arg-Trp-Gln Chemical compound [H]N[C@@H](CCCNC(N)=N)C(=O)N[C@@H](CC1=CNC2=C1C=CC=C2)C(=O)N[C@@H](CCC(N)=O)C(O)=O ZUVMUOOHJYNJPP-XIRDDKMYSA-N 0.000 description 4
- SRUUBQBAVNQZGJ-LAEOZQHASA-N Asn-Gln-Val Chemical compound CC(C)[C@@H](C(=O)O)NC(=O)[C@H](CCC(=O)N)NC(=O)[C@H](CC(=O)N)N SRUUBQBAVNQZGJ-LAEOZQHASA-N 0.000 description 4
- WONGRTVAMHFGBE-WDSKDSINSA-N Asn-Gly-Gln Chemical compound C(CC(=O)N)[C@@H](C(=O)O)NC(=O)CNC(=O)[C@H](CC(=O)N)N WONGRTVAMHFGBE-WDSKDSINSA-N 0.000 description 4
- HNXWVVHIGTZTBO-LKXGYXEUSA-N Asn-Ser-Thr Chemical compound C[C@@H](O)[C@@H](C(O)=O)NC(=O)[C@H](CO)NC(=O)[C@@H](N)CC(N)=O HNXWVVHIGTZTBO-LKXGYXEUSA-N 0.000 description 4
- QNNBHTFDFFFHGC-KKUMJFAQSA-N Asn-Tyr-Lys Chemical compound C1=CC(=CC=C1C[C@@H](C(=O)N[C@@H](CCCCN)C(=O)O)NC(=O)[C@H](CC(=O)N)N)O QNNBHTFDFFFHGC-KKUMJFAQSA-N 0.000 description 4
- DPNWSMBUYCLEDG-CIUDSAMLSA-N Asp-Lys-Ser Chemical compound [H]N[C@@H](CC(O)=O)C(=O)N[C@@H](CCCCN)C(=O)N[C@@H](CO)C(O)=O DPNWSMBUYCLEDG-CIUDSAMLSA-N 0.000 description 4
- DONWIPDSZZJHHK-HJGDQZAQSA-N Asp-Lys-Thr Chemical compound C[C@H]([C@@H](C(=O)O)NC(=O)[C@H](CCCCN)NC(=O)[C@H](CC(=O)O)N)O DONWIPDSZZJHHK-HJGDQZAQSA-N 0.000 description 4
- JSNWZMFSLIWAHS-HJGDQZAQSA-N Asp-Thr-Leu Chemical compound C[C@H]([C@@H](C(=O)N[C@@H](CC(C)C)C(=O)O)NC(=O)[C@H](CC(=O)O)N)O JSNWZMFSLIWAHS-HJGDQZAQSA-N 0.000 description 4
- NDNZRWUDUMTITL-FXQIFTODSA-N Cys-Ser-Val Chemical compound [H]N[C@@H](CS)C(=O)N[C@@H](CO)C(=O)N[C@@H](C(C)C)C(O)=O NDNZRWUDUMTITL-FXQIFTODSA-N 0.000 description 4
- LFQSCWFLJHTTHZ-UHFFFAOYSA-N Ethanol Chemical compound CCO LFQSCWFLJHTTHZ-UHFFFAOYSA-N 0.000 description 4
- 206010016654 Fibrosis Diseases 0.000 description 4
- XKBASPWPBXNVLQ-WDSKDSINSA-N Gln-Gly-Asn Chemical compound [H]N[C@@H](CCC(N)=O)C(=O)NCC(=O)N[C@@H](CC(N)=O)C(O)=O XKBASPWPBXNVLQ-WDSKDSINSA-N 0.000 description 4
- ZEEPYMXTJWIMSN-GUBZILKMSA-N Gln-Lys-Ser Chemical compound NCCCC[C@@H](C(=O)N[C@@H](CO)C(O)=O)NC(=O)[C@@H](N)CCC(N)=O ZEEPYMXTJWIMSN-GUBZILKMSA-N 0.000 description 4
- RJONUNZIMUXUOI-GUBZILKMSA-N Glu-Asn-Lys Chemical compound C(CCN)C[C@@H](C(=O)O)NC(=O)[C@H](CC(=O)N)NC(=O)[C@H](CCC(=O)O)N RJONUNZIMUXUOI-GUBZILKMSA-N 0.000 description 4
- CKOFNWCLWRYUHK-XHNCKOQMSA-N Glu-Asp-Pro Chemical compound C1C[C@@H](N(C1)C(=O)[C@H](CC(=O)O)NC(=O)[C@H](CCC(=O)O)N)C(=O)O CKOFNWCLWRYUHK-XHNCKOQMSA-N 0.000 description 4
- QDMVXRNLOPTPIE-WDCWCFNPSA-N Glu-Lys-Thr Chemical compound [H]N[C@@H](CCC(O)=O)C(=O)N[C@@H](CCCCN)C(=O)N[C@@H]([C@@H](C)O)C(O)=O QDMVXRNLOPTPIE-WDCWCFNPSA-N 0.000 description 4
- ZYRXTRTUCAVNBQ-GVXVVHGQSA-N Glu-Val-Lys Chemical compound CC(C)[C@@H](C(=O)N[C@@H](CCCCN)C(=O)O)NC(=O)[C@H](CCC(=O)O)N ZYRXTRTUCAVNBQ-GVXVVHGQSA-N 0.000 description 4
- HAOUOFNNJJLVNS-BQBZGAKWSA-N Gly-Pro-Ser Chemical compound NCC(=O)N1CCC[C@H]1C(=O)N[C@@H](CO)C(O)=O HAOUOFNNJJLVNS-BQBZGAKWSA-N 0.000 description 4
- SYOJVRNQCXYEOV-XVKPBYJWSA-N Gly-Val-Glu Chemical compound [H]NCC(=O)N[C@@H](C(C)C)C(=O)N[C@@H](CCC(O)=O)C(O)=O SYOJVRNQCXYEOV-XVKPBYJWSA-N 0.000 description 4
- MAABHGXCIBEYQR-XVYDVKMFSA-N His-Asn-Ala Chemical compound C[C@@H](C(=O)O)NC(=O)[C@H](CC(=O)N)NC(=O)[C@H](CC1=CN=CN1)N MAABHGXCIBEYQR-XVYDVKMFSA-N 0.000 description 4
- HIAHVKLTHNOENC-HGNGGELXSA-N His-Glu-Ala Chemical compound [H]N[C@@H](CC1=CNC=N1)C(=O)N[C@@H](CCC(O)=O)C(=O)N[C@@H](C)C(O)=O HIAHVKLTHNOENC-HGNGGELXSA-N 0.000 description 4
- HZWWOGWOBQBETJ-CUJWVEQBSA-N His-Thr-Cys Chemical compound C[C@H]([C@@H](C(=O)N[C@@H](CS)C(=O)O)NC(=O)[C@H](CC1=CN=CN1)N)O HZWWOGWOBQBETJ-CUJWVEQBSA-N 0.000 description 4
- CSTDQOOBZBAJKE-BWAGICSOSA-N His-Tyr-Thr Chemical compound C[C@H]([C@@H](C(=O)O)NC(=O)[C@H](CC1=CC=C(C=C1)O)NC(=O)[C@H](CC2=CN=CN2)N)O CSTDQOOBZBAJKE-BWAGICSOSA-N 0.000 description 4
- VEXZGXHMUGYJMC-UHFFFAOYSA-N Hydrochloric acid Chemical compound Cl VEXZGXHMUGYJMC-UHFFFAOYSA-N 0.000 description 4
- JHNJNTMTZHEDLJ-NAKRPEOUSA-N Ile-Ser-Arg Chemical compound CC[C@H](C)[C@H](N)C(=O)N[C@@H](CO)C(=O)N[C@@H](CCCN=C(N)N)C(O)=O JHNJNTMTZHEDLJ-NAKRPEOUSA-N 0.000 description 4
- VGSPNSSCMOHRRR-BJDJZHNGSA-N Ile-Ser-Lys Chemical compound CC[C@H](C)[C@@H](C(=O)N[C@@H](CO)C(=O)N[C@@H](CCCCN)C(=O)O)N VGSPNSSCMOHRRR-BJDJZHNGSA-N 0.000 description 4
- DCXYFEDJOCDNAF-REOHCLBHSA-N L-asparagine Chemical compound OC(=O)[C@@H](N)CC(N)=O DCXYFEDJOCDNAF-REOHCLBHSA-N 0.000 description 4
- RCFDOSNHHZGBOY-UHFFFAOYSA-N L-isoleucyl-L-alanine Natural products CCC(C)C(N)C(=O)NC(C)C(O)=O RCFDOSNHHZGBOY-UHFFFAOYSA-N 0.000 description 4
- TYYLDKGBCJGJGW-UHFFFAOYSA-N L-tryptophan-L-tyrosine Natural products C=1NC2=CC=CC=C2C=1CC(N)C(=O)NC(C(O)=O)CC1=CC=C(O)C=C1 TYYLDKGBCJGJGW-UHFFFAOYSA-N 0.000 description 4
- XOWMDXHFSBCAKQ-SRVKXCTJSA-N Leu-Ser-Leu Chemical compound CC(C)C[C@H](N)C(=O)N[C@@H](CO)C(=O)N[C@H](C(O)=O)CC(C)C XOWMDXHFSBCAKQ-SRVKXCTJSA-N 0.000 description 4
- LINKCQUOMUDLKN-KATARQTJSA-N Leu-Thr-Cys Chemical compound C[C@H]([C@@H](C(=O)N[C@@H](CS)C(=O)O)NC(=O)[C@H](CC(C)C)N)O LINKCQUOMUDLKN-KATARQTJSA-N 0.000 description 4
- DAYQSYGBCUKVKT-VOAKCMCISA-N Leu-Thr-Lys Chemical compound CC(C)C[C@H](N)C(=O)N[C@@H]([C@@H](C)O)C(=O)N[C@@H](CCCCN)C(O)=O DAYQSYGBCUKVKT-VOAKCMCISA-N 0.000 description 4
- YQFZRHYZLARWDY-IHRRRGAJSA-N Leu-Val-Lys Chemical compound CC(C)C[C@H](N)C(=O)N[C@@H](C(C)C)C(=O)N[C@H](C(O)=O)CCCCN YQFZRHYZLARWDY-IHRRRGAJSA-N 0.000 description 4
- KCXUCYYZNZFGLL-SRVKXCTJSA-N Lys-Ala-Leu Chemical compound [H]N[C@@H](CCCCN)C(=O)N[C@@H](C)C(=O)N[C@@H](CC(C)C)C(O)=O KCXUCYYZNZFGLL-SRVKXCTJSA-N 0.000 description 4
- MWVUEPNEPWMFBD-SRVKXCTJSA-N Lys-Cys-Lys Chemical compound NCCCC[C@H](N)C(=O)N[C@@H](CS)C(=O)N[C@H](C(O)=O)CCCCN MWVUEPNEPWMFBD-SRVKXCTJSA-N 0.000 description 4
- ODUQLUADRKMHOZ-JYJNAYRXSA-N Lys-Glu-Tyr Chemical compound C1=CC(=CC=C1C[C@@H](C(=O)O)NC(=O)[C@H](CCC(=O)O)NC(=O)[C@H](CCCCN)N)O ODUQLUADRKMHOZ-JYJNAYRXSA-N 0.000 description 4
- LUTDBHBIHHREDC-IHRRRGAJSA-N Lys-Pro-Lys Chemical compound NCCCC[C@H](N)C(=O)N1CCC[C@H]1C(=O)N[C@@H](CCCCN)C(O)=O LUTDBHBIHHREDC-IHRRRGAJSA-N 0.000 description 4
- YKBSXQFZWFXFIB-VOAKCMCISA-N Lys-Thr-Lys Chemical compound NCCCC[C@H](N)C(=O)N[C@@H]([C@H](O)C)C(=O)N[C@@H](CCCCN)C(O)=O YKBSXQFZWFXFIB-VOAKCMCISA-N 0.000 description 4
- 102100025751 Mothers against decapentaplegic homolog 2 Human genes 0.000 description 4
- 101710143123 Mothers against decapentaplegic homolog 2 Proteins 0.000 description 4
- 241000283973 Oryctolagus cuniculus Species 0.000 description 4
- JOXIIFVCSATTDH-IHPCNDPISA-N Phe-Asn-Trp Chemical compound C1=CC=C(C=C1)C[C@@H](C(=O)N[C@@H](CC(=O)N)C(=O)N[C@@H](CC2=CNC3=CC=CC=C32)C(=O)O)N JOXIIFVCSATTDH-IHPCNDPISA-N 0.000 description 4
- ZJPGOXWRFNKIQL-JYJNAYRXSA-N Phe-Pro-Pro Chemical compound C([C@H](N)C(=O)N1[C@@H](CCC1)C(=O)N1[C@@H](CCC1)C(O)=O)C1=CC=CC=C1 ZJPGOXWRFNKIQL-JYJNAYRXSA-N 0.000 description 4
- SJRQWEDYTKYHHL-SLFFLAALSA-N Phe-Tyr-Pro Chemical compound C1C[C@@H](N(C1)C(=O)[C@H](CC2=CC=C(C=C2)O)NC(=O)[C@H](CC3=CC=CC=C3)N)C(=O)O SJRQWEDYTKYHHL-SLFFLAALSA-N 0.000 description 4
- KIPIKSXPPLABPN-CIUDSAMLSA-N Pro-Glu-Asn Chemical compound NC(=O)C[C@@H](C(O)=O)NC(=O)[C@H](CCC(O)=O)NC(=O)[C@@H]1CCCN1 KIPIKSXPPLABPN-CIUDSAMLSA-N 0.000 description 4
- UIMCLYYSUCIUJM-UWVGGRQHSA-N Pro-Gly-Lys Chemical compound NCCCC[C@@H](C(O)=O)NC(=O)CNC(=O)[C@@H]1CCCN1 UIMCLYYSUCIUJM-UWVGGRQHSA-N 0.000 description 4
- HWLKHNDRXWTFTN-GUBZILKMSA-N Pro-Pro-Cys Chemical compound C1C[C@H](NC1)C(=O)N2CCC[C@H]2C(=O)N[C@@H](CS)C(=O)O HWLKHNDRXWTFTN-GUBZILKMSA-N 0.000 description 4
- FDMKYQQYJKYCLV-GUBZILKMSA-N Pro-Pro-Ser Chemical compound OC[C@@H](C(O)=O)NC(=O)[C@@H]1CCCN1C(=O)[C@H]1NCCC1 FDMKYQQYJKYCLV-GUBZILKMSA-N 0.000 description 4
- QPFJSHSJFIYDJZ-GHCJXIJMSA-N Ser-Asp-Ile Chemical compound CC[C@H](C)[C@@H](C(O)=O)NC(=O)[C@H](CC(O)=O)NC(=O)[C@@H](N)CO QPFJSHSJFIYDJZ-GHCJXIJMSA-N 0.000 description 4
- XKWABWFMQXMUMT-HJGDQZAQSA-N Thr-Pro-Glu Chemical compound [H]N[C@@H]([C@@H](C)O)C(=O)N1CCC[C@H]1C(=O)N[C@@H](CCC(O)=O)C(O)=O XKWABWFMQXMUMT-HJGDQZAQSA-N 0.000 description 4
- ZESGVALRVJIVLZ-VFCFLDTKSA-N Thr-Thr-Pro Chemical compound C[C@H]([C@@H](C(=O)N[C@@H]([C@@H](C)O)C(=O)N1CCC[C@@H]1C(=O)O)N)O ZESGVALRVJIVLZ-VFCFLDTKSA-N 0.000 description 4
- UDCHKDYNMRJYMI-QEJZJMRPSA-N Trp-Glu-Ser Chemical compound [H]N[C@@H](CC1=CNC2=C1C=CC=C2)C(=O)N[C@@H](CCC(O)=O)C(=O)N[C@@H](CO)C(O)=O UDCHKDYNMRJYMI-QEJZJMRPSA-N 0.000 description 4
- PWKMJDQXKCENMF-MEYUZBJRSA-N Tyr-Thr-Leu Chemical compound [H]N[C@@H](CC1=CC=C(O)C=C1)C(=O)N[C@@H]([C@@H](C)O)C(=O)N[C@@H](CC(C)C)C(O)=O PWKMJDQXKCENMF-MEYUZBJRSA-N 0.000 description 4
- SQUMHUZLJDUROQ-YDHLFZDLSA-N Tyr-Val-Asp Chemical compound [H]N[C@@H](CC1=CC=C(O)C=C1)C(=O)N[C@@H](C(C)C)C(=O)N[C@@H](CC(O)=O)C(O)=O SQUMHUZLJDUROQ-YDHLFZDLSA-N 0.000 description 4
- HJSLDXZAZGFPDK-ULQDDVLXSA-N Val-Phe-Leu Chemical compound CC(C)C[C@@H](C(=O)O)NC(=O)[C@H](CC1=CC=CC=C1)NC(=O)[C@H](C(C)C)N HJSLDXZAZGFPDK-ULQDDVLXSA-N 0.000 description 4
- KISFXYYRKKNLOP-IHRRRGAJSA-N Val-Phe-Ser Chemical compound CC(C)[C@@H](C(=O)N[C@@H](CC1=CC=CC=C1)C(=O)N[C@@H](CO)C(=O)O)N KISFXYYRKKNLOP-IHRRRGAJSA-N 0.000 description 4
- KSFXWENSJABBFI-ZKWXMUAHSA-N Val-Ser-Asn Chemical compound [H]N[C@@H](C(C)C)C(=O)N[C@@H](CO)C(=O)N[C@@H](CC(N)=O)C(O)=O KSFXWENSJABBFI-ZKWXMUAHSA-N 0.000 description 4
- 230000004913 activation Effects 0.000 description 4
- 125000000539 amino acid group Chemical group 0.000 description 4
- 108010038633 aspartylglutamate Proteins 0.000 description 4
- 210000004978 chinese hamster ovary cell Anatomy 0.000 description 4
- 239000003246 corticosteroid Substances 0.000 description 4
- 230000003111 delayed effect Effects 0.000 description 4
- 239000002552 dosage form Substances 0.000 description 4
- 239000003995 emulsifying agent Substances 0.000 description 4
- 230000004761 fibrosis Effects 0.000 description 4
- 239000004615 ingredient Substances 0.000 description 4
- 230000003993 interaction Effects 0.000 description 4
- 108010057821 leucylproline Proteins 0.000 description 4
- 108010003700 lysyl aspartic acid Proteins 0.000 description 4
- HQKMJHAJHXVSDF-UHFFFAOYSA-L magnesium stearate Chemical compound [Mg+2].CCCCCCCCCCCCCCCCCC([O-])=O.CCCCCCCCCCCCCCCCCC([O-])=O HQKMJHAJHXVSDF-UHFFFAOYSA-L 0.000 description 4
- 238000004519 manufacturing process Methods 0.000 description 4
- 238000005259 measurement Methods 0.000 description 4
- 239000002953 phosphate buffered saline Substances 0.000 description 4
- 238000002360 preparation method Methods 0.000 description 4
- 108010077112 prolyl-proline Proteins 0.000 description 4
- 238000010561 standard procedure Methods 0.000 description 4
- 238000002198 surface plasmon resonance spectroscopy Methods 0.000 description 4
- 108010071097 threonyl-lysyl-proline Proteins 0.000 description 4
- 210000003371 toe Anatomy 0.000 description 4
- 108010044292 tryptophyltyrosine Proteins 0.000 description 4
- 108010051110 tyrosyl-lysine Proteins 0.000 description 4
- QTBSBXVTEAMEQO-UHFFFAOYSA-N Acetic acid Chemical compound CC(O)=O QTBSBXVTEAMEQO-UHFFFAOYSA-N 0.000 description 3
- RMAWDDRDTRSZIR-ZLUOBGJFSA-N Ala-Ser-Asp Chemical compound [H]N[C@@H](C)C(=O)N[C@@H](CO)C(=O)N[C@@H](CC(O)=O)C(O)=O RMAWDDRDTRSZIR-ZLUOBGJFSA-N 0.000 description 3
- YVTHEZNOKSAWRW-DCAQKATOSA-N Arg-Lys-Ala Chemical compound [H]N[C@@H](CCCNC(N)=N)C(=O)N[C@@H](CCCCN)C(=O)N[C@@H](C)C(O)=O YVTHEZNOKSAWRW-DCAQKATOSA-N 0.000 description 3
- MECFLTFREHAZLH-ACZMJKKPSA-N Asn-Glu-Cys Chemical compound C(CC(=O)O)[C@@H](C(=O)N[C@@H](CS)C(=O)O)NC(=O)[C@H](CC(=O)N)N MECFLTFREHAZLH-ACZMJKKPSA-N 0.000 description 3
- PDECQIHABNQRHN-GUBZILKMSA-N Asp-Glu-Leu Chemical compound CC(C)C[C@@H](C(O)=O)NC(=O)[C@H](CCC(O)=O)NC(=O)[C@@H](N)CC(O)=O PDECQIHABNQRHN-GUBZILKMSA-N 0.000 description 3
- 101100512078 Caenorhabditis elegans lys-1 gene Proteins 0.000 description 3
- 241000282693 Cercopithecidae Species 0.000 description 3
- 108091026890 Coding region Proteins 0.000 description 3
- 108020004705 Codon Proteins 0.000 description 3
- SDXQKJAWASHMIZ-CIUDSAMLSA-N Cys-Glu-Met Chemical compound [H]N[C@@H](CS)C(=O)N[C@@H](CCC(O)=O)C(=O)N[C@@H](CCSC)C(O)=O SDXQKJAWASHMIZ-CIUDSAMLSA-N 0.000 description 3
- NITLUESFANGEIW-BQBZGAKWSA-N Cys-Pro-Gly Chemical compound [H]N[C@@H](CS)C(=O)N1CCC[C@H]1C(=O)NCC(O)=O NITLUESFANGEIW-BQBZGAKWSA-N 0.000 description 3
- 102000009109 Fc receptors Human genes 0.000 description 3
- 108010087819 Fc receptors Proteins 0.000 description 3
- 102000016970 Follistatin Human genes 0.000 description 3
- 108010014612 Follistatin Proteins 0.000 description 3
- QQLBPVKLJBAXBS-FXQIFTODSA-N Glu-Glu-Asn Chemical compound [H]N[C@@H](CCC(O)=O)C(=O)N[C@@H](CCC(O)=O)C(=O)N[C@@H](CC(N)=O)C(O)=O QQLBPVKLJBAXBS-FXQIFTODSA-N 0.000 description 3
- YQPFCZVKMUVZIN-AUTRQRHGSA-N Glu-Val-Gln Chemical compound [H]N[C@@H](CCC(O)=O)C(=O)N[C@@H](C(C)C)C(=O)N[C@@H](CCC(N)=O)C(O)=O YQPFCZVKMUVZIN-AUTRQRHGSA-N 0.000 description 3
- OCDLPQDYTJPWNG-YUMQZZPRSA-N Gly-Asn-Lys Chemical compound C(CCN)C[C@@H](C(=O)O)NC(=O)[C@H](CC(=O)N)NC(=O)CN OCDLPQDYTJPWNG-YUMQZZPRSA-N 0.000 description 3
- VLIJYPMATZSOLL-YUMQZZPRSA-N Gly-Lys-Cys Chemical compound C(CCN)C[C@@H](C(=O)N[C@@H](CS)C(=O)O)NC(=O)CN VLIJYPMATZSOLL-YUMQZZPRSA-N 0.000 description 3
- LBDXVCBAJJNJNN-WHFBIAKZSA-N Gly-Ser-Cys Chemical compound NCC(=O)N[C@@H](CO)C(=O)N[C@@H](CS)C(O)=O LBDXVCBAJJNJNN-WHFBIAKZSA-N 0.000 description 3
- BAYQNCWLXIDLHX-ONGXEEELSA-N Gly-Val-Leu Chemical compound CC(C)C[C@@H](C(O)=O)NC(=O)[C@H](C(C)C)NC(=O)CN BAYQNCWLXIDLHX-ONGXEEELSA-N 0.000 description 3
- PEDCQBHIVMGVHV-UHFFFAOYSA-N Glycerine Chemical compound OCC(O)CO PEDCQBHIVMGVHV-UHFFFAOYSA-N 0.000 description 3
- 241000238631 Hexapoda Species 0.000 description 3
- 102100026122 High affinity immunoglobulin gamma Fc receptor I Human genes 0.000 description 3
- 241000282412 Homo Species 0.000 description 3
- 101000913074 Homo sapiens High affinity immunoglobulin gamma Fc receptor I Proteins 0.000 description 3
- 206010020880 Hypertrophy Diseases 0.000 description 3
- KWHFUMYCSPJCFQ-NGTWOADLSA-N Ile-Thr-Trp Chemical compound CC[C@H](C)[C@@H](C(=O)N[C@@H]([C@@H](C)O)C(=O)N[C@@H](CC1=CNC2=CC=CC=C21)C(=O)O)N KWHFUMYCSPJCFQ-NGTWOADLSA-N 0.000 description 3
- RRSLQOLASISYTB-CIUDSAMLSA-N Leu-Cys-Asp Chemical compound [H]N[C@@H](CC(C)C)C(=O)N[C@@H](CS)C(=O)N[C@@H](CC(O)=O)C(O)=O RRSLQOLASISYTB-CIUDSAMLSA-N 0.000 description 3
- FEHQLKKBVJHSEC-SZMVWBNQSA-N Leu-Glu-Trp Chemical compound C1=CC=C2C(C[C@H](NC(=O)[C@H](CCC(O)=O)NC(=O)[C@@H](N)CC(C)C)C(O)=O)=CNC2=C1 FEHQLKKBVJHSEC-SZMVWBNQSA-N 0.000 description 3
- HVJVUYQWFYMGJS-GVXVVHGQSA-N Leu-Glu-Val Chemical compound [H]N[C@@H](CC(C)C)C(=O)N[C@@H](CCC(O)=O)C(=O)N[C@@H](C(C)C)C(O)=O HVJVUYQWFYMGJS-GVXVVHGQSA-N 0.000 description 3
- PGLGNCVOWIORQE-SRVKXCTJSA-N Lys-His-Ser Chemical compound [H]N[C@@H](CCCCN)C(=O)N[C@@H](CC1=CNC=N1)C(=O)N[C@@H](CO)C(O)=O PGLGNCVOWIORQE-SRVKXCTJSA-N 0.000 description 3
- MUBZPKHOEPUJKR-UHFFFAOYSA-N Oxalic acid Chemical compound OC(=O)C(O)=O MUBZPKHOEPUJKR-UHFFFAOYSA-N 0.000 description 3
- ONIBWKKTOPOVIA-UHFFFAOYSA-N Proline Natural products OC(=O)C1CCCN1 ONIBWKKTOPOVIA-UHFFFAOYSA-N 0.000 description 3
- JCLAFVNDBJMLBC-JBDRJPRFSA-N Ser-Ser-Ile Chemical compound [H]N[C@@H](CO)C(=O)N[C@@H](CO)C(=O)N[C@@H]([C@@H](C)CC)C(O)=O JCLAFVNDBJMLBC-JBDRJPRFSA-N 0.000 description 3
- XVNZSJIKGJLQLH-RCWTZXSCSA-N Thr-Arg-Met Chemical compound C[C@H]([C@@H](C(=O)N[C@@H](CCCN=C(N)N)C(=O)N[C@@H](CCSC)C(=O)O)N)O XVNZSJIKGJLQLH-RCWTZXSCSA-N 0.000 description 3
- ASJDFGOPDCVXTG-KATARQTJSA-N Thr-Cys-Leu Chemical compound [H]N[C@@H]([C@@H](C)O)C(=O)N[C@@H](CS)C(=O)N[C@@H](CC(C)C)C(O)=O ASJDFGOPDCVXTG-KATARQTJSA-N 0.000 description 3
- 108700019146 Transgenes Proteins 0.000 description 3
- ZMANZCXQSJIPKH-UHFFFAOYSA-N Triethylamine Chemical compound CCN(CC)CC ZMANZCXQSJIPKH-UHFFFAOYSA-N 0.000 description 3
- AVIQBBOOTZENLH-KKUMJFAQSA-N Tyr-Leu-Cys Chemical compound CC(C)C[C@@H](C(=O)N[C@@H](CS)C(=O)O)NC(=O)[C@H](CC1=CC=C(C=C1)O)N AVIQBBOOTZENLH-KKUMJFAQSA-N 0.000 description 3
- 230000002159 abnormal effect Effects 0.000 description 3
- 239000013543 active substance Substances 0.000 description 3
- 238000007792 addition Methods 0.000 description 3
- 210000003423 ankle Anatomy 0.000 description 3
- 238000002869 basic local alignment search tool Methods 0.000 description 3
- 239000011230 binding agent Substances 0.000 description 3
- 238000003766 bioinformatics method Methods 0.000 description 3
- 230000008859 change Effects 0.000 description 3
- 150000001875 compounds Chemical class 0.000 description 3
- 238000004590 computer program Methods 0.000 description 3
- 210000002808 connective tissue Anatomy 0.000 description 3
- 230000006378 damage Effects 0.000 description 3
- 239000000539 dimer Substances 0.000 description 3
- 239000000796 flavoring agent Substances 0.000 description 3
- 235000013355 food flavoring agent Nutrition 0.000 description 3
- 210000002683 foot Anatomy 0.000 description 3
- 230000013595 glycosylation Effects 0.000 description 3
- 238000006206 glycosylation reaction Methods 0.000 description 3
- 108010015792 glycyllysine Proteins 0.000 description 3
- 210000001624 hip Anatomy 0.000 description 3
- 210000005260 human cell Anatomy 0.000 description 3
- 230000002209 hydrophobic effect Effects 0.000 description 3
- 230000005847 immunogenicity Effects 0.000 description 3
- 238000000338 in vitro Methods 0.000 description 3
- 210000002414 leg Anatomy 0.000 description 3
- 239000000178 monomer Substances 0.000 description 3
- 238000010172 mouse model Methods 0.000 description 3
- 210000000663 muscle cell Anatomy 0.000 description 3
- 239000003755 preservative agent Substances 0.000 description 3
- 230000000750 progressive effect Effects 0.000 description 3
- 108010070643 prolylglutamic acid Proteins 0.000 description 3
- 230000008929 regeneration Effects 0.000 description 3
- 238000011069 regeneration method Methods 0.000 description 3
- 230000001105 regulatory effect Effects 0.000 description 3
- 102200136845 rs186964570 Human genes 0.000 description 3
- 239000000243 solution Substances 0.000 description 3
- 210000002435 tendon Anatomy 0.000 description 3
- 238000002560 therapeutic procedure Methods 0.000 description 3
- 108010073969 valyllysine Proteins 0.000 description 3
- 239000000080 wetting agent Substances 0.000 description 3
- 210000000707 wrist Anatomy 0.000 description 3
- IESDGNYHXIOKRW-YXMSTPNBSA-N (2s)-2-[[(2s)-1-[(2s)-6-amino-2-[[(2s,3r)-2-amino-3-hydroxybutanoyl]amino]hexanoyl]pyrrolidine-2-carbonyl]amino]-5-(diaminomethylideneamino)pentanoic acid Chemical compound C[C@@H](O)[C@H](N)C(=O)N[C@@H](CCCCN)C(=O)N1CCC[C@H]1C(=O)N[C@@H](CCCNC(N)=N)C(O)=O IESDGNYHXIOKRW-YXMSTPNBSA-N 0.000 description 2
- DIBLBAURNYJYBF-XLXZRNDBSA-N (2s)-2-[[(2s)-2-[[2-[[(2s)-6-amino-2-[[(2s)-2-amino-3-methylbutanoyl]amino]hexanoyl]amino]acetyl]amino]-3-phenylpropanoyl]amino]-3-(4-hydroxyphenyl)propanoic acid Chemical compound C([C@H](NC(=O)CNC(=O)[C@H](CCCCN)NC(=O)[C@@H](N)C(C)C)C(=O)N[C@@H](CC=1C=CC(O)=CC=1)C(O)=O)C1=CC=CC=C1 DIBLBAURNYJYBF-XLXZRNDBSA-N 0.000 description 2
- 108010059616 Activins Proteins 0.000 description 2
- 102000005606 Activins Human genes 0.000 description 2
- AAQGRPOPTAUUBM-ZLUOBGJFSA-N Ala-Ala-Asn Chemical compound [H]N[C@@H](C)C(=O)N[C@@H](C)C(=O)N[C@@H](CC(N)=O)C(O)=O AAQGRPOPTAUUBM-ZLUOBGJFSA-N 0.000 description 2
- KVWLTGNCJYDJET-LSJOCFKGSA-N Ala-Arg-His Chemical compound C[C@@H](C(=O)N[C@@H](CCCN=C(N)N)C(=O)N[C@@H](CC1=CN=CN1)C(=O)O)N KVWLTGNCJYDJET-LSJOCFKGSA-N 0.000 description 2
- XQGIRPGAVLFKBJ-CIUDSAMLSA-N Ala-Asn-Lys Chemical compound N[C@@H](C)C(=O)N[C@@H](CC(N)=O)C(=O)N[C@@H](CCCCN)C(=O)O XQGIRPGAVLFKBJ-CIUDSAMLSA-N 0.000 description 2
- OILNWMNBLIHXQK-ZLUOBGJFSA-N Ala-Cys-Ser Chemical compound [H]N[C@@H](C)C(=O)N[C@@H](CS)C(=O)N[C@@H](CO)C(O)=O OILNWMNBLIHXQK-ZLUOBGJFSA-N 0.000 description 2
- NHLAEBFGWPXFGI-WHFBIAKZSA-N Ala-Gly-Asn Chemical compound C[C@@H](C(=O)NCC(=O)N[C@@H](CC(=O)N)C(=O)O)N NHLAEBFGWPXFGI-WHFBIAKZSA-N 0.000 description 2
- SVHRPCMZTWZROG-DCAQKATOSA-N Arg-Cys-Lys Chemical compound C(CCN)C[C@@H](C(=O)O)NC(=O)[C@H](CS)NC(=O)[C@H](CCCN=C(N)N)N SVHRPCMZTWZROG-DCAQKATOSA-N 0.000 description 2
- SLKLLQWZQHXYSV-CIUDSAMLSA-N Asn-Ala-Lys Chemical compound NC(=O)C[C@H](N)C(=O)N[C@@H](C)C(=O)N[C@@H](CCCCN)C(O)=O SLKLLQWZQHXYSV-CIUDSAMLSA-N 0.000 description 2
- AYOAHKWVQLNPDM-HJGDQZAQSA-N Asn-Lys-Thr Chemical compound [H]N[C@@H](CC(N)=O)C(=O)N[C@@H](CCCCN)C(=O)N[C@@H]([C@@H](C)O)C(O)=O AYOAHKWVQLNPDM-HJGDQZAQSA-N 0.000 description 2
- SNYCNNPOFYBCEK-ZLUOBGJFSA-N Asn-Ser-Ser Chemical compound [H]N[C@@H](CC(N)=O)C(=O)N[C@@H](CO)C(=O)N[C@@H](CO)C(O)=O SNYCNNPOFYBCEK-ZLUOBGJFSA-N 0.000 description 2
- LGCVSPFCFXWUEY-IHPCNDPISA-N Asn-Trp-Tyr Chemical compound C1=CC=C2C(=C1)C(=CN2)C[C@@H](C(=O)N[C@@H](CC3=CC=C(C=C3)O)C(=O)O)NC(=O)[C@H](CC(=O)N)N LGCVSPFCFXWUEY-IHPCNDPISA-N 0.000 description 2
- JUWISGAGWSDGDH-KKUMJFAQSA-N Asp-Phe-Lys Chemical compound NCCCC[C@@H](C(O)=O)NC(=O)[C@@H](NC(=O)[C@@H](N)CC(O)=O)CC1=CC=CC=C1 JUWISGAGWSDGDH-KKUMJFAQSA-N 0.000 description 2
- AHWRSSLYSGLBGD-CIUDSAMLSA-N Asp-Pro-Glu Chemical compound OC(=O)C[C@H](N)C(=O)N1CCC[C@H]1C(=O)N[C@@H](CCC(O)=O)C(O)=O AHWRSSLYSGLBGD-CIUDSAMLSA-N 0.000 description 2
- CZIVKMOEXPILDK-SRVKXCTJSA-N Asp-Tyr-Ser Chemical compound [H]N[C@@H](CC(O)=O)C(=O)N[C@@H](CC1=CC=C(O)C=C1)C(=O)N[C@@H](CO)C(O)=O CZIVKMOEXPILDK-SRVKXCTJSA-N 0.000 description 2
- 201000006935 Becker muscular dystrophy Diseases 0.000 description 2
- 241000283690 Bos taurus Species 0.000 description 2
- 108091016585 CD44 antigen Proteins 0.000 description 2
- 241001164374 Calyx Species 0.000 description 2
- 241000283707 Capra Species 0.000 description 2
- 102000004420 Creatine Kinase Human genes 0.000 description 2
- 108010042126 Creatine kinase Proteins 0.000 description 2
- 102000036364 Cullin Ring E3 Ligases Human genes 0.000 description 2
- XXDATQFUGMAJRV-XIRDDKMYSA-N Cys-Leu-Trp Chemical compound [H]N[C@@H](CS)C(=O)N[C@@H](CC(C)C)C(=O)N[C@@H](CC1=CNC2=C1C=CC=C2)C(O)=O XXDATQFUGMAJRV-XIRDDKMYSA-N 0.000 description 2
- ZXCAQANTQWBICD-DCAQKATOSA-N Cys-Lys-Val Chemical compound CC(C)[C@@H](C(=O)O)NC(=O)[C@H](CCCCN)NC(=O)[C@H](CS)N ZXCAQANTQWBICD-DCAQKATOSA-N 0.000 description 2
- ALTQTAKGRFLRLR-GUBZILKMSA-N Cys-Val-Val Chemical compound CC(C)[C@@H](C(=O)N[C@@H](C(C)C)C(=O)O)NC(=O)[C@H](CS)N ALTQTAKGRFLRLR-GUBZILKMSA-N 0.000 description 2
- FBPFZTCFMRRESA-KVTDHHQDSA-N D-Mannitol Chemical compound OC[C@@H](O)[C@@H](O)[C@H](O)[C@H](O)CO FBPFZTCFMRRESA-KVTDHHQDSA-N 0.000 description 2
- 102000010834 Extracellular Matrix Proteins Human genes 0.000 description 2
- 108010037362 Extracellular Matrix Proteins Proteins 0.000 description 2
- 241000282326 Felis catus Species 0.000 description 2
- 102100038546 Fibronectin type III and SPRY domain-containing protein 1 Human genes 0.000 description 2
- 229930091371 Fructose Natural products 0.000 description 2
- 239000005715 Fructose Substances 0.000 description 2
- RFSUNEUAIZKAJO-ARQDHWQXSA-N Fructose Chemical compound OC[C@H]1O[C@](O)(CO)[C@@H](O)[C@@H]1O RFSUNEUAIZKAJO-ARQDHWQXSA-N 0.000 description 2
- 206010017577 Gait disturbance Diseases 0.000 description 2
- DBNLXHGDGBUCDV-KKUMJFAQSA-N Gln-Phe-Met Chemical compound [H]N[C@@H](CCC(N)=O)C(=O)N[C@@H](CC1=CC=CC=C1)C(=O)N[C@@H](CCSC)C(O)=O DBNLXHGDGBUCDV-KKUMJFAQSA-N 0.000 description 2
- FQCILXROGNOZON-YUMQZZPRSA-N Gln-Pro-Gly Chemical compound NC(=O)CC[C@H](N)C(=O)N1CCC[C@H]1C(=O)NCC(O)=O FQCILXROGNOZON-YUMQZZPRSA-N 0.000 description 2
- MFHVAWMMKZBSRQ-ACZMJKKPSA-N Gln-Ser-Cys Chemical compound C(CC(=O)N)[C@@H](C(=O)N[C@@H](CO)C(=O)N[C@@H](CS)C(=O)O)N MFHVAWMMKZBSRQ-ACZMJKKPSA-N 0.000 description 2
- RUFHOVYUYSNDNY-ACZMJKKPSA-N Glu-Ala-Ala Chemical compound OC(=O)[C@H](C)NC(=O)[C@H](C)NC(=O)[C@@H](N)CCC(O)=O RUFHOVYUYSNDNY-ACZMJKKPSA-N 0.000 description 2
- ITYRYNUZHPNCIK-GUBZILKMSA-N Glu-Ala-Leu Chemical compound [H]N[C@@H](CCC(O)=O)C(=O)N[C@@H](C)C(=O)N[C@@H](CC(C)C)C(O)=O ITYRYNUZHPNCIK-GUBZILKMSA-N 0.000 description 2
- GLWXKFRTOHKGIT-ACZMJKKPSA-N Glu-Asn-Asn Chemical compound [H]N[C@@H](CCC(O)=O)C(=O)N[C@@H](CC(N)=O)C(=O)N[C@@H](CC(N)=O)C(O)=O GLWXKFRTOHKGIT-ACZMJKKPSA-N 0.000 description 2
- QPRZKNOOOBWXSU-CIUDSAMLSA-N Glu-Asp-Arg Chemical compound OC(=O)CC[C@H](N)C(=O)N[C@@H](CC(O)=O)C(=O)N[C@H](C(O)=O)CCCN=C(N)N QPRZKNOOOBWXSU-CIUDSAMLSA-N 0.000 description 2
- HNVFSTLPVJWIDV-CIUDSAMLSA-N Glu-Glu-Gln Chemical compound OC(=O)CC[C@H](N)C(=O)N[C@@H](CCC(O)=O)C(=O)N[C@@H](CCC(N)=O)C(O)=O HNVFSTLPVJWIDV-CIUDSAMLSA-N 0.000 description 2
- MWMJCGBSIORNCD-AVGNSLFASA-N Glu-Leu-Leu Chemical compound [H]N[C@@H](CCC(O)=O)C(=O)N[C@@H](CC(C)C)C(=O)N[C@@H](CC(C)C)C(O)=O MWMJCGBSIORNCD-AVGNSLFASA-N 0.000 description 2
- ALMBZBOCGSVSAI-ACZMJKKPSA-N Glu-Ser-Asn Chemical compound C(CC(=O)O)[C@@H](C(=O)N[C@@H](CO)C(=O)N[C@@H](CC(=O)N)C(=O)O)N ALMBZBOCGSVSAI-ACZMJKKPSA-N 0.000 description 2
- LWYUQLZOIORFFJ-XKBZYTNZSA-N Glu-Thr-Cys Chemical compound C[C@H]([C@@H](C(=O)N[C@@H](CS)C(=O)O)NC(=O)[C@H](CCC(=O)O)N)O LWYUQLZOIORFFJ-XKBZYTNZSA-N 0.000 description 2
- MFYLRRCYBBJYPI-JYJNAYRXSA-N Glu-Tyr-Lys Chemical compound C1=CC(=CC=C1C[C@@H](C(=O)N[C@@H](CCCCN)C(=O)O)NC(=O)[C@H](CCC(=O)O)N)O MFYLRRCYBBJYPI-JYJNAYRXSA-N 0.000 description 2
- WQZGKKKJIJFFOK-GASJEMHNSA-N Glucose Chemical compound OC[C@H]1OC(O)[C@H](O)[C@@H](O)[C@@H]1O WQZGKKKJIJFFOK-GASJEMHNSA-N 0.000 description 2
- GRIRDMVMJJDZKV-RCOVLWMOSA-N Gly-Asn-Val Chemical compound [H]NCC(=O)N[C@@H](CC(N)=O)C(=O)N[C@@H](C(C)C)C(O)=O GRIRDMVMJJDZKV-RCOVLWMOSA-N 0.000 description 2
- PABFFPWEJMEVEC-JGVFFNPUSA-N Gly-Gln-Pro Chemical compound C1C[C@@H](N(C1)C(=O)[C@H](CCC(=O)N)NC(=O)CN)C(=O)O PABFFPWEJMEVEC-JGVFFNPUSA-N 0.000 description 2
- YIFUFYZELCMPJP-YUMQZZPRSA-N Gly-Leu-Cys Chemical compound NCC(=O)N[C@@H](CC(C)C)C(=O)N[C@@H](CS)C(O)=O YIFUFYZELCMPJP-YUMQZZPRSA-N 0.000 description 2
- 239000004471 Glycine Substances 0.000 description 2
- 102000008055 Heparan Sulfate Proteoglycans Human genes 0.000 description 2
- 229920002971 Heparan sulfate Polymers 0.000 description 2
- WMKXFMUJRCEGRP-SRVKXCTJSA-N His-Asn-His Chemical compound C1=C(NC=N1)C[C@@H](C(=O)N[C@@H](CC(=O)N)C(=O)N[C@@H](CC2=CN=CN2)C(=O)O)N WMKXFMUJRCEGRP-SRVKXCTJSA-N 0.000 description 2
- 101001030521 Homo sapiens Fibronectin type III and SPRY domain-containing protein 1 Proteins 0.000 description 2
- 102000009490 IgG Receptors Human genes 0.000 description 2
- 108010073807 IgG Receptors Proteins 0.000 description 2
- GUBGYTABKSRVRQ-QKKXKWKRSA-N Lactose Natural products OC[C@H]1O[C@@H](O[C@H]2[C@H](O)[C@@H](O)C(O)O[C@@H]2CO)[C@H](O)[C@@H](O)[C@H]1O GUBGYTABKSRVRQ-QKKXKWKRSA-N 0.000 description 2
- BABSVXFGKFLIGW-UWVGGRQHSA-N Leu-Gly-Arg Chemical compound CC(C)C[C@H](N)C(=O)NCC(=O)N[C@H](C(O)=O)CCCNC(N)=N BABSVXFGKFLIGW-UWVGGRQHSA-N 0.000 description 2
- BTNXKBVLWJBTNR-SRVKXCTJSA-N Leu-His-Asn Chemical compound [H]N[C@@H](CC(C)C)C(=O)N[C@@H](CC1=CNC=N1)C(=O)N[C@@H](CC(N)=O)C(O)=O BTNXKBVLWJBTNR-SRVKXCTJSA-N 0.000 description 2
- IFMPDNRWZZEZSL-SRVKXCTJSA-N Leu-Leu-Cys Chemical compound CC(C)C[C@H](N)C(=O)N[C@@H](CC(C)C)C(=O)N[C@@H](CS)C(O)=O IFMPDNRWZZEZSL-SRVKXCTJSA-N 0.000 description 2
- QNTJIDXQHWUBKC-BZSNNMDCSA-N Leu-Lys-Phe Chemical compound [H]N[C@@H](CC(C)C)C(=O)N[C@@H](CCCCN)C(=O)N[C@@H](CC1=CC=CC=C1)C(O)=O QNTJIDXQHWUBKC-BZSNNMDCSA-N 0.000 description 2
- SBANPBVRHYIMRR-UHFFFAOYSA-N Leu-Ser-Pro Natural products CC(C)CC(N)C(=O)NC(CO)C(=O)N1CCCC1C(O)=O SBANPBVRHYIMRR-UHFFFAOYSA-N 0.000 description 2
- PPGBXYKMUMHFBF-KATARQTJSA-N Leu-Ser-Thr Chemical compound [H]N[C@@H](CC(C)C)C(=O)N[C@@H](CO)C(=O)N[C@@H]([C@@H](C)O)C(O)=O PPGBXYKMUMHFBF-KATARQTJSA-N 0.000 description 2
- BTEMNFBEAAOGBR-BZSNNMDCSA-N Leu-Tyr-Lys Chemical compound CC(C)C[C@@H](C(=O)N[C@@H](CC1=CC=C(C=C1)O)C(=O)N[C@@H](CCCCN)C(=O)O)N BTEMNFBEAAOGBR-BZSNNMDCSA-N 0.000 description 2
- 108060001084 Luciferase Proteins 0.000 description 2
- CRNNMTHBMRFQNG-GUBZILKMSA-N Lys-Glu-Cys Chemical compound C(CCN)C[C@@H](C(=O)N[C@@H](CCC(=O)O)C(=O)N[C@@H](CS)C(=O)O)N CRNNMTHBMRFQNG-GUBZILKMSA-N 0.000 description 2
- XNKDCYABMBBEKN-IUCAKERBSA-N Lys-Gly-Gln Chemical compound NCCCC[C@H](N)C(=O)NCC(=O)N[C@H](C(O)=O)CCC(N)=O XNKDCYABMBBEKN-IUCAKERBSA-N 0.000 description 2
- WQDKIVRHTQYJSN-DCAQKATOSA-N Lys-Ser-Arg Chemical compound C(CCN)C[C@@H](C(=O)N[C@@H](CO)C(=O)N[C@@H](CCCN=C(N)N)C(=O)O)N WQDKIVRHTQYJSN-DCAQKATOSA-N 0.000 description 2
- TVHCDSBMFQYPNA-RHYQMDGZSA-N Lys-Thr-Arg Chemical compound [H]N[C@@H](CCCCN)C(=O)N[C@@H]([C@@H](C)O)C(=O)N[C@@H](CCCNC(N)=N)C(O)=O TVHCDSBMFQYPNA-RHYQMDGZSA-N 0.000 description 2
- CUHGAUZONORRIC-HJGDQZAQSA-N Lys-Thr-Asn Chemical compound C[C@H]([C@@H](C(=O)N[C@@H](CC(=O)N)C(=O)O)NC(=O)[C@H](CCCCN)N)O CUHGAUZONORRIC-HJGDQZAQSA-N 0.000 description 2
- TVOOGUNBIWAURO-KATARQTJSA-N Lys-Thr-Cys Chemical compound C[C@H]([C@@H](C(=O)N[C@@H](CS)C(=O)O)NC(=O)[C@H](CCCCN)N)O TVOOGUNBIWAURO-KATARQTJSA-N 0.000 description 2
- IEVXCWPVBYCJRZ-IXOXFDKPSA-N Lys-Thr-His Chemical compound NCCCC[C@H](N)C(=O)N[C@@H]([C@H](O)C)C(=O)N[C@H](C(O)=O)CC1=CN=CN1 IEVXCWPVBYCJRZ-IXOXFDKPSA-N 0.000 description 2
- DLCAXBGXGOVUCD-PPCPHDFISA-N Lys-Thr-Ile Chemical compound [H]N[C@@H](CCCCN)C(=O)N[C@@H]([C@@H](C)O)C(=O)N[C@@H]([C@@H](C)CC)C(O)=O DLCAXBGXGOVUCD-PPCPHDFISA-N 0.000 description 2
- KQAREVUPVXMNNP-WDSOQIARSA-N Lys-Trp-Met Chemical compound [H]N[C@@H](CCCCN)C(=O)N[C@@H](CC1=CNC2=C1C=CC=C2)C(=O)N[C@@H](CCSC)C(O)=O KQAREVUPVXMNNP-WDSOQIARSA-N 0.000 description 2
- 241000124008 Mammalia Species 0.000 description 2
- 229930195725 Mannitol Natural products 0.000 description 2
- DRXODWRPPUFIAY-DCAQKATOSA-N Met-Asn-Lys Chemical compound CSCC[C@H](N)C(=O)N[C@@H](CC(N)=O)C(=O)N[C@H](C(O)=O)CCCCN DRXODWRPPUFIAY-DCAQKATOSA-N 0.000 description 2
- ALTHVGNGGZZSAC-SRVKXCTJSA-N Met-Val-Arg Chemical compound CSCC[C@H](N)C(=O)N[C@@H](C(C)C)C(=O)N[C@H](C(O)=O)CCCNC(N)=N ALTHVGNGGZZSAC-SRVKXCTJSA-N 0.000 description 2
- 102100025748 Mothers against decapentaplegic homolog 3 Human genes 0.000 description 2
- 101710143111 Mothers against decapentaplegic homolog 3 Proteins 0.000 description 2
- 102000008934 Muscle Proteins Human genes 0.000 description 2
- 108010074084 Muscle Proteins Proteins 0.000 description 2
- 206010068871 Myotonic dystrophy Diseases 0.000 description 2
- 241001494479 Pecora Species 0.000 description 2
- MSHZERMPZKCODG-ACRUOGEOSA-N Phe-Leu-Phe Chemical compound C([C@H](N)C(=O)N[C@@H](CC(C)C)C(=O)N[C@@H](CC=1C=CC=CC=1)C(O)=O)C1=CC=CC=C1 MSHZERMPZKCODG-ACRUOGEOSA-N 0.000 description 2
- NBIIXXVUZAFLBC-UHFFFAOYSA-N Phosphoric acid Chemical compound OP(O)(O)=O NBIIXXVUZAFLBC-UHFFFAOYSA-N 0.000 description 2
- 241000288906 Primates Species 0.000 description 2
- TUYWCHPXKQTISF-LPEHRKFASA-N Pro-Cys-Pro Chemical compound C1C[C@H](NC1)C(=O)N[C@@H](CS)C(=O)N2CCC[C@@H]2C(=O)O TUYWCHPXKQTISF-LPEHRKFASA-N 0.000 description 2
- VPEVBAUSTBWQHN-NHCYSSNCSA-N Pro-Glu-Val Chemical compound [H]N1CCC[C@H]1C(=O)N[C@@H](CCC(O)=O)C(=O)N[C@@H](C(C)C)C(O)=O VPEVBAUSTBWQHN-NHCYSSNCSA-N 0.000 description 2
- PCWLNNZTBJTZRN-AVGNSLFASA-N Pro-Pro-Lys Chemical compound NCCCC[C@@H](C(O)=O)NC(=O)[C@@H]1CCCN1C(=O)[C@H]1NCCC1 PCWLNNZTBJTZRN-AVGNSLFASA-N 0.000 description 2
- GOMUXSCOIWIJFP-GUBZILKMSA-N Pro-Ser-Arg Chemical compound [H]N1CCC[C@H]1C(=O)N[C@@H](CO)C(=O)N[C@@H](CCCNC(N)=N)C(O)=O GOMUXSCOIWIJFP-GUBZILKMSA-N 0.000 description 2
- PRKWBYCXBBSLSK-GUBZILKMSA-N Pro-Ser-Val Chemical compound [H]N1CCC[C@H]1C(=O)N[C@@H](CO)C(=O)N[C@@H](C(C)C)C(O)=O PRKWBYCXBBSLSK-GUBZILKMSA-N 0.000 description 2
- LOUPRKONTZGTKE-WZBLMQSHSA-N Quinine Chemical compound C([C@H]([C@H](C1)C=C)C2)C[N@@]1[C@@H]2[C@H](O)C1=CC=NC2=CC=C(OC)C=C21 LOUPRKONTZGTKE-WZBLMQSHSA-N 0.000 description 2
- 241000700159 Rattus Species 0.000 description 2
- MMGJPDWSIOAGTH-ACZMJKKPSA-N Ser-Ala-Gln Chemical compound [H]N[C@@H](CO)C(=O)N[C@@H](C)C(=O)N[C@@H](CCC(N)=O)C(O)=O MMGJPDWSIOAGTH-ACZMJKKPSA-N 0.000 description 2
- OYEDZGNMSBZCIM-XGEHTFHBSA-N Ser-Arg-Thr Chemical compound [H]N[C@@H](CO)C(=O)N[C@@H](CCCNC(N)=N)C(=O)N[C@@H]([C@@H](C)O)C(O)=O OYEDZGNMSBZCIM-XGEHTFHBSA-N 0.000 description 2
- VGNYHOBZJKWRGI-CIUDSAMLSA-N Ser-Asn-Lys Chemical compound NCCCC[C@@H](C(O)=O)NC(=O)[C@H](CC(N)=O)NC(=O)[C@@H](N)CO VGNYHOBZJKWRGI-CIUDSAMLSA-N 0.000 description 2
- IFPBAGJBHSNYPR-ZKWXMUAHSA-N Ser-Ile-Gly Chemical compound [H]N[C@@H](CO)C(=O)N[C@@H]([C@@H](C)CC)C(=O)NCC(O)=O IFPBAGJBHSNYPR-ZKWXMUAHSA-N 0.000 description 2
- YUJLIIRMIAGMCQ-CIUDSAMLSA-N Ser-Leu-Ser Chemical compound [H]N[C@@H](CO)C(=O)N[C@@H](CC(C)C)C(=O)N[C@@H](CO)C(O)=O YUJLIIRMIAGMCQ-CIUDSAMLSA-N 0.000 description 2
- GZSZPKSBVAOGIE-CIUDSAMLSA-N Ser-Lys-Ala Chemical compound [H]N[C@@H](CO)C(=O)N[C@@H](CCCCN)C(=O)N[C@@H](C)C(O)=O GZSZPKSBVAOGIE-CIUDSAMLSA-N 0.000 description 2
- 229920002472 Starch Polymers 0.000 description 2
- 108090000054 Syndecan-2 Proteins 0.000 description 2
- KWQBJOUOSNJDRR-XAVMHZPKSA-N Thr-Cys-Pro Chemical compound C[C@H]([C@@H](C(=O)N[C@@H](CS)C(=O)N1CCC[C@@H]1C(=O)O)N)O KWQBJOUOSNJDRR-XAVMHZPKSA-N 0.000 description 2
- DSLHSTIUAPKERR-XGEHTFHBSA-N Thr-Cys-Val Chemical compound [H]N[C@@H]([C@@H](C)O)C(=O)N[C@@H](CS)C(=O)N[C@@H](C(C)C)C(O)=O DSLHSTIUAPKERR-XGEHTFHBSA-N 0.000 description 2
- DJDSEDOKJTZBAR-ZDLURKLDSA-N Thr-Gly-Ser Chemical compound C[C@@H](O)[C@H](N)C(=O)NCC(=O)N[C@@H](CO)C(O)=O DJDSEDOKJTZBAR-ZDLURKLDSA-N 0.000 description 2
- NCXVJIQMWSGRHY-KXNHARMFSA-N Thr-Leu-Pro Chemical compound C[C@H]([C@@H](C(=O)N[C@@H](CC(C)C)C(=O)N1CCC[C@@H]1C(=O)O)N)O NCXVJIQMWSGRHY-KXNHARMFSA-N 0.000 description 2
- AYFVYJQAPQTCCC-UHFFFAOYSA-N Threonine Natural products CC(O)C(N)C(O)=O AYFVYJQAPQTCCC-UHFFFAOYSA-N 0.000 description 2
- 239000004473 Threonine Substances 0.000 description 2
- DQDXHYIEITXNJY-BPUTZDHNSA-N Trp-Gln-Gln Chemical compound C1=CC=C2C(=C1)C(=CN2)C[C@@H](C(=O)N[C@@H](CCC(=O)N)C(=O)N[C@@H](CCC(=O)N)C(=O)O)N DQDXHYIEITXNJY-BPUTZDHNSA-N 0.000 description 2
- SCCKSNREWHMKOJ-SRVKXCTJSA-N Tyr-Asn-Ser Chemical compound N[C@@H](Cc1ccc(O)cc1)C(=O)N[C@@H](CC(N)=O)C(=O)N[C@@H](CO)C(O)=O SCCKSNREWHMKOJ-SRVKXCTJSA-N 0.000 description 2
- RYSNTWVRSLCAJZ-RYUDHWBXSA-N Tyr-Gln-Gly Chemical compound OC(=O)CNC(=O)[C@H](CCC(N)=O)NC(=O)[C@@H](N)CC1=CC=C(O)C=C1 RYSNTWVRSLCAJZ-RYUDHWBXSA-N 0.000 description 2
- RIVVDNTUSRVTQT-IRIUXVKKSA-N Tyr-Thr-Gln Chemical compound C[C@H]([C@@H](C(=O)N[C@@H](CCC(=O)N)C(=O)O)NC(=O)[C@H](CC1=CC=C(C=C1)O)N)O RIVVDNTUSRVTQT-IRIUXVKKSA-N 0.000 description 2
- XSQUKJJJFZCRTK-UHFFFAOYSA-N Urea Chemical compound NC(N)=O XSQUKJJJFZCRTK-UHFFFAOYSA-N 0.000 description 2
- JTWIMNMUYLQNPI-WPRPVWTQSA-N Val-Gly-Arg Chemical compound CC(C)[C@H](N)C(=O)NCC(=O)N[C@H](C(O)=O)CCCNC(N)=N JTWIMNMUYLQNPI-WPRPVWTQSA-N 0.000 description 2
- HPANGHISDXDUQY-ULQDDVLXSA-N Val-Lys-Phe Chemical compound CC(C)[C@@H](C(=O)N[C@@H](CCCCN)C(=O)N[C@@H](CC1=CC=CC=C1)C(=O)O)N HPANGHISDXDUQY-ULQDDVLXSA-N 0.000 description 2
- KZSNJWFQEVHDMF-UHFFFAOYSA-N Valine Natural products CC(C)C(N)C(O)=O KZSNJWFQEVHDMF-UHFFFAOYSA-N 0.000 description 2
- 241000251539 Vertebrata <Metazoa> Species 0.000 description 2
- 210000001766 X chromosome Anatomy 0.000 description 2
- 230000002378 acidificating effect Effects 0.000 description 2
- 239000000488 activin Substances 0.000 description 2
- 108010047495 alanylglycine Proteins 0.000 description 2
- 239000003708 ampul Substances 0.000 description 2
- 238000010171 animal model Methods 0.000 description 2
- 239000005557 antagonist Substances 0.000 description 2
- 230000010056 antibody-dependent cellular cytotoxicity Effects 0.000 description 2
- 108010092854 aspartyllysine Proteins 0.000 description 2
- 239000008228 bacteriostatic water for injection Substances 0.000 description 2
- 230000008901 benefit Effects 0.000 description 2
- 230000015572 biosynthetic process Effects 0.000 description 2
- 230000017531 blood circulation Effects 0.000 description 2
- 210000004204 blood vessel Anatomy 0.000 description 2
- 239000000872 buffer Substances 0.000 description 2
- 150000001720 carbohydrates Chemical class 0.000 description 2
- 210000000038 chest Anatomy 0.000 description 2
- 230000000052 comparative effect Effects 0.000 description 2
- 208000028831 congenital heart disease Diseases 0.000 description 2
- 229960001334 corticosteroids Drugs 0.000 description 2
- 230000034994 death Effects 0.000 description 2
- 238000011161 development Methods 0.000 description 2
- 230000018109 developmental process Effects 0.000 description 2
- 238000003745 diagnosis Methods 0.000 description 2
- 210000001198 duodenum Anatomy 0.000 description 2
- 235000019441 ethanol Nutrition 0.000 description 2
- 210000002744 extracellular matrix Anatomy 0.000 description 2
- 235000019197 fats Nutrition 0.000 description 2
- 230000002068 genetic effect Effects 0.000 description 2
- 210000004195 gingiva Anatomy 0.000 description 2
- 108010078144 glutaminyl-glycine Proteins 0.000 description 2
- 230000035876 healing Effects 0.000 description 2
- 210000003494 hepatocyte Anatomy 0.000 description 2
- HNDVDQJCIGZPNO-UHFFFAOYSA-N histidine Natural products OC(=O)C(N)CC1=CN=CN1 HNDVDQJCIGZPNO-UHFFFAOYSA-N 0.000 description 2
- 108010092114 histidylphenylalanine Proteins 0.000 description 2
- 230000002779 inactivation Effects 0.000 description 2
- 238000001802 infusion Methods 0.000 description 2
- 238000002347 injection Methods 0.000 description 2
- 239000007924 injection Substances 0.000 description 2
- 210000003734 kidney Anatomy 0.000 description 2
- 239000008101 lactose Substances 0.000 description 2
- 108010044311 leucyl-glycyl-glycine Proteins 0.000 description 2
- 239000000314 lubricant Substances 0.000 description 2
- 235000019359 magnesium stearate Nutrition 0.000 description 2
- 239000000594 mannitol Substances 0.000 description 2
- 235000010355 mannitol Nutrition 0.000 description 2
- 108010056582 methionylglutamic acid Proteins 0.000 description 2
- 108010085203 methionylmethionine Proteins 0.000 description 2
- 210000003098 myoblast Anatomy 0.000 description 2
- 230000017074 necrotic cell death Effects 0.000 description 2
- 230000001575 pathological effect Effects 0.000 description 2
- 230000007170 pathology Effects 0.000 description 2
- 239000001267 polyvinylpyrrolidone Substances 0.000 description 2
- 229920000036 polyvinylpyrrolidone Polymers 0.000 description 2
- 235000013855 polyvinylpyrrolidone Nutrition 0.000 description 2
- 239000013641 positive control Substances 0.000 description 2
- 230000002035 prolonged effect Effects 0.000 description 2
- 108010020755 prolyl-glycyl-glycine Proteins 0.000 description 2
- 108010015796 prolylisoleucine Proteins 0.000 description 2
- 108010053725 prolylvaline Proteins 0.000 description 2
- 239000013074 reference sample Substances 0.000 description 2
- 206010039722 scoliosis Diseases 0.000 description 2
- 238000004904 shortening Methods 0.000 description 2
- 230000011664 signaling Effects 0.000 description 2
- 239000002904 solvent Substances 0.000 description 2
- 210000000278 spinal cord Anatomy 0.000 description 2
- 239000008107 starch Substances 0.000 description 2
- 235000019698 starch Nutrition 0.000 description 2
- 239000008223 sterile water Substances 0.000 description 2
- 239000000758 substrate Substances 0.000 description 2
- 239000000829 suppository Substances 0.000 description 2
- 239000004094 surface-active agent Substances 0.000 description 2
- 239000000725 suspension Substances 0.000 description 2
- 239000002562 thickening agent Substances 0.000 description 2
- 210000000115 thoracic cavity Anatomy 0.000 description 2
- UBWXUGDQUBIEIZ-UHFFFAOYSA-N (13-methyl-3-oxo-2,6,7,8,9,10,11,12,14,15,16,17-dodecahydro-1h-cyclopenta[a]phenanthren-17-yl) 3-phenylpropanoate Chemical compound CC12CCC(C3CCC(=O)C=C3CC3)C3C1CCC2OC(=O)CCC1=CC=CC=C1 UBWXUGDQUBIEIZ-UHFFFAOYSA-N 0.000 description 1
- OZFAFGSSMRRTDW-UHFFFAOYSA-N (2,4-dichlorophenyl) benzenesulfonate Chemical compound ClC1=CC(Cl)=CC=C1OS(=O)(=O)C1=CC=CC=C1 OZFAFGSSMRRTDW-UHFFFAOYSA-N 0.000 description 1
- NHBKXEKEPDILRR-UHFFFAOYSA-N 2,3-bis(butanoylsulfanyl)propyl butanoate Chemical compound CCCC(=O)OCC(SC(=O)CCC)CSC(=O)CCC NHBKXEKEPDILRR-UHFFFAOYSA-N 0.000 description 1
- MIJDSYMOBYNHOT-UHFFFAOYSA-N 2-(ethylamino)ethanol Chemical compound CCNCCO MIJDSYMOBYNHOT-UHFFFAOYSA-N 0.000 description 1
- 244000215068 Acacia senegal Species 0.000 description 1
- 102000010825 Actinin Human genes 0.000 description 1
- 108010063503 Actinin Proteins 0.000 description 1
- 241000251468 Actinopterygii Species 0.000 description 1
- OYJCVIGKMXUVKB-GARJFASQSA-N Ala-Leu-Pro Chemical compound C[C@@H](C(=O)N[C@@H](CC(C)C)C(=O)N1CCC[C@@H]1C(=O)O)N OYJCVIGKMXUVKB-GARJFASQSA-N 0.000 description 1
- IORKCNUBHNIMKY-CIUDSAMLSA-N Ala-Pro-Glu Chemical compound C[C@H](N)C(=O)N1CCC[C@H]1C(=O)N[C@@H](CCC(O)=O)C(O)=O IORKCNUBHNIMKY-CIUDSAMLSA-N 0.000 description 1
- WQLDNOCHHRISMS-NAKRPEOUSA-N Ala-Pro-Ile Chemical compound [H]N[C@@H](C)C(=O)N1CCC[C@H]1C(=O)N[C@@H]([C@@H](C)CC)C(O)=O WQLDNOCHHRISMS-NAKRPEOUSA-N 0.000 description 1
- NCQMBSJGJMYKCK-ZLUOBGJFSA-N Ala-Ser-Ser Chemical compound [H]N[C@@H](C)C(=O)N[C@@H](CO)C(=O)N[C@@H](CO)C(O)=O NCQMBSJGJMYKCK-ZLUOBGJFSA-N 0.000 description 1
- GUBGYTABKSRVRQ-XLOQQCSPSA-N Alpha-Lactose Chemical compound O[C@@H]1[C@@H](O)[C@@H](O)[C@@H](CO)O[C@H]1O[C@@H]1[C@@H](CO)O[C@H](O)[C@H](O)[C@H]1O GUBGYTABKSRVRQ-XLOQQCSPSA-N 0.000 description 1
- QGZKDVFQNNGYKY-UHFFFAOYSA-O Ammonium Chemical compound [NH4+] QGZKDVFQNNGYKY-UHFFFAOYSA-O 0.000 description 1
- 229920000856 Amylose Polymers 0.000 description 1
- 101100533825 Arabidopsis thaliana FSD3 gene Proteins 0.000 description 1
- VXXHDZKEQNGXNU-QXEWZRGKSA-N Arg-Asp-Val Chemical compound CC(C)[C@@H](C(O)=O)NC(=O)[C@H](CC(O)=O)NC(=O)[C@@H](N)CCCN=C(N)N VXXHDZKEQNGXNU-QXEWZRGKSA-N 0.000 description 1
- HPSVTWMFWCHKFN-GARJFASQSA-N Arg-Glu-Pro Chemical compound C1C[C@@H](N(C1)C(=O)[C@H](CCC(=O)O)NC(=O)[C@H](CCCN=C(N)N)N)C(=O)O HPSVTWMFWCHKFN-GARJFASQSA-N 0.000 description 1
- HCIUUZGFTDTEGM-NAKRPEOUSA-N Arg-Ile-Cys Chemical compound CC[C@H](C)[C@@H](C(=O)N[C@@H](CS)C(=O)O)NC(=O)[C@H](CCCN=C(N)N)N HCIUUZGFTDTEGM-NAKRPEOUSA-N 0.000 description 1
- 239000004475 Arginine Substances 0.000 description 1
- IARGXWMWRFOQPG-GCJQMDKQSA-N Asn-Ala-Thr Chemical compound [H]N[C@@H](CC(N)=O)C(=O)N[C@@H](C)C(=O)N[C@@H]([C@@H](C)O)C(O)=O IARGXWMWRFOQPG-GCJQMDKQSA-N 0.000 description 1
- AKEBUSZTMQLNIX-UWJYBYFXSA-N Asn-Ala-Tyr Chemical compound C[C@@H](C(=O)N[C@@H](CC1=CC=C(C=C1)O)C(=O)O)NC(=O)[C@H](CC(=O)N)N AKEBUSZTMQLNIX-UWJYBYFXSA-N 0.000 description 1
- OLVIPTLKNSAYRJ-YUMQZZPRSA-N Asn-Gly-Lys Chemical compound C(CCN)C[C@@H](C(=O)O)NC(=O)CNC(=O)[C@H](CC(=O)N)N OLVIPTLKNSAYRJ-YUMQZZPRSA-N 0.000 description 1
- COWITDLVHMZSIW-CIUDSAMLSA-N Asn-Lys-Ser Chemical compound [H]N[C@@H](CC(N)=O)C(=O)N[C@@H](CCCCN)C(=O)N[C@@H](CO)C(O)=O COWITDLVHMZSIW-CIUDSAMLSA-N 0.000 description 1
- NPZJLGMWMDNQDD-GHCJXIJMSA-N Asn-Ser-Ile Chemical compound [H]N[C@@H](CC(N)=O)C(=O)N[C@@H](CO)C(=O)N[C@@H]([C@@H](C)CC)C(O)=O NPZJLGMWMDNQDD-GHCJXIJMSA-N 0.000 description 1
- KIJLEFNHWSXHRU-NUMRIWBASA-N Asp-Gln-Thr Chemical compound [H]N[C@@H](CC(O)=O)C(=O)N[C@@H](CCC(N)=O)C(=O)N[C@@H]([C@@H](C)O)C(O)=O KIJLEFNHWSXHRU-NUMRIWBASA-N 0.000 description 1
- ZEDBMCPXPIYJLW-XHNCKOQMSA-N Asp-Glu-Pro Chemical compound C1C[C@@H](N(C1)C(=O)[C@H](CCC(=O)O)NC(=O)[C@H](CC(=O)O)N)C(=O)O ZEDBMCPXPIYJLW-XHNCKOQMSA-N 0.000 description 1
- WSGVTKZFVJSJOG-RCOVLWMOSA-N Asp-Gly-Val Chemical compound [H]N[C@@H](CC(O)=O)C(=O)NCC(=O)N[C@@H](C(C)C)C(O)=O WSGVTKZFVJSJOG-RCOVLWMOSA-N 0.000 description 1
- KTTCQQNRRLCIBC-GHCJXIJMSA-N Asp-Ile-Ala Chemical compound [H]N[C@@H](CC(O)=O)C(=O)N[C@@H]([C@@H](C)CC)C(=O)N[C@@H](C)C(O)=O KTTCQQNRRLCIBC-GHCJXIJMSA-N 0.000 description 1
- YUELDQUPTAYEGM-XIRDDKMYSA-N Asp-Trp-Leu Chemical compound CC(C)C[C@@H](C(=O)O)NC(=O)[C@H](CC1=CNC2=CC=CC=C21)NC(=O)[C@H](CC(=O)O)N YUELDQUPTAYEGM-XIRDDKMYSA-N 0.000 description 1
- DCXYFEDJOCDNAF-UHFFFAOYSA-N Asparagine Natural products OC(=O)C(N)CC(N)=O DCXYFEDJOCDNAF-UHFFFAOYSA-N 0.000 description 1
- 208000006096 Attention Deficit Disorder with Hyperactivity Diseases 0.000 description 1
- 208000036864 Attention deficit/hyperactivity disease Diseases 0.000 description 1
- 241000271566 Aves Species 0.000 description 1
- 101100297347 Caenorhabditis elegans pgl-3 gene Proteins 0.000 description 1
- OYPRJOBELJOOCE-UHFFFAOYSA-N Calcium Chemical compound [Ca] OYPRJOBELJOOCE-UHFFFAOYSA-N 0.000 description 1
- 241000282552 Chlorocebus aethiops Species 0.000 description 1
- 235000001258 Cinchona calisaya Nutrition 0.000 description 1
- 108700010070 Codon Usage Proteins 0.000 description 1
- 208000032170 Congenital Abnormalities Diseases 0.000 description 1
- 206010010356 Congenital anomaly Diseases 0.000 description 1
- 108091035707 Consensus sequence Proteins 0.000 description 1
- 208000034656 Contusions Diseases 0.000 description 1
- 241000699800 Cricetinae Species 0.000 description 1
- 241000938605 Crocodylia Species 0.000 description 1
- 235000014375 Curcuma Nutrition 0.000 description 1
- 244000164480 Curcuma aromatica Species 0.000 description 1
- PKNIZMPLMSKROD-BIIVOSGPSA-N Cys-Ala-Pro Chemical compound C[C@@H](C(=O)N1CCC[C@@H]1C(=O)O)NC(=O)[C@H](CS)N PKNIZMPLMSKROD-BIIVOSGPSA-N 0.000 description 1
- QDFBJJABJKOLTD-FXQIFTODSA-N Cys-Asn-Arg Chemical compound [H]N[C@@H](CS)C(=O)N[C@@H](CC(N)=O)C(=O)N[C@@H](CCCNC(N)=N)C(O)=O QDFBJJABJKOLTD-FXQIFTODSA-N 0.000 description 1
- SQJSYLDKQBZQTG-FXQIFTODSA-N Cys-Asn-Met Chemical compound CSCC[C@@H](C(=O)O)NC(=O)[C@H](CC(=O)N)NC(=O)[C@H](CS)N SQJSYLDKQBZQTG-FXQIFTODSA-N 0.000 description 1
- BCSYBBMFGLHCOA-ACZMJKKPSA-N Cys-Glu-Cys Chemical compound SC[C@H](N)C(=O)N[C@@H](CCC(O)=O)C(=O)N[C@@H](CS)C(O)=O BCSYBBMFGLHCOA-ACZMJKKPSA-N 0.000 description 1
- KJJASVYBTKRYSN-FXQIFTODSA-N Cys-Pro-Asp Chemical compound C1C[C@H](N(C1)C(=O)[C@H](CS)N)C(=O)N[C@@H](CC(=O)O)C(=O)O KJJASVYBTKRYSN-FXQIFTODSA-N 0.000 description 1
- GGRDJANMZPGMNS-CIUDSAMLSA-N Cys-Ser-Leu Chemical compound [H]N[C@@H](CS)C(=O)N[C@@H](CO)C(=O)N[C@@H](CC(C)C)C(O)=O GGRDJANMZPGMNS-CIUDSAMLSA-N 0.000 description 1
- YNJBLTDKTMKEET-ZLUOBGJFSA-N Cys-Ser-Ser Chemical compound SC[C@H](N)C(=O)N[C@@H](CO)C(=O)N[C@@H](CO)C(O)=O YNJBLTDKTMKEET-ZLUOBGJFSA-N 0.000 description 1
- FTTZLFIEUQHLHH-BWBBJGPYSA-N Cys-Thr-Cys Chemical compound C[C@H]([C@@H](C(=O)N[C@@H](CS)C(=O)O)NC(=O)[C@H](CS)N)O FTTZLFIEUQHLHH-BWBBJGPYSA-N 0.000 description 1
- FNXOZWPPOJRBRE-XGEHTFHBSA-N Cys-Val-Thr Chemical compound C[C@H]([C@@H](C(=O)O)NC(=O)[C@H](C(C)C)NC(=O)[C@H](CS)N)O FNXOZWPPOJRBRE-XGEHTFHBSA-N 0.000 description 1
- 229920001353 Dextrin Polymers 0.000 description 1
- 239000004375 Dextrin Substances 0.000 description 1
- FEWJPZIEWOKRBE-JCYAYHJZSA-N Dextrotartaric acid Chemical compound OC(=O)[C@H](O)[C@@H](O)C(O)=O FEWJPZIEWOKRBE-JCYAYHJZSA-N 0.000 description 1
- 102100024746 Dihydrofolate reductase Human genes 0.000 description 1
- 102220521744 Dihydrofolate reductase_K84E_mutation Human genes 0.000 description 1
- 239000012591 Dulbecco’s Phosphate Buffered Saline Substances 0.000 description 1
- 208000014094 Dystonic disease Diseases 0.000 description 1
- 102100025682 Dystroglycan 1 Human genes 0.000 description 1
- 108010071885 Dystroglycans Proteins 0.000 description 1
- 241000196324 Embryophyta Species 0.000 description 1
- 102100034239 Emerin Human genes 0.000 description 1
- 201000009344 Emery-Dreifuss muscular dystrophy Diseases 0.000 description 1
- 241000588724 Escherichia coli Species 0.000 description 1
- 206010015995 Eyelid ptosis Diseases 0.000 description 1
- 208000037149 Facioscapulohumeral dystrophy Diseases 0.000 description 1
- 208000015220 Febrile disease Diseases 0.000 description 1
- 102100038545 Fibronectin type III and SPRY domain-containing protein 2 Human genes 0.000 description 1
- 201000008808 Fibrosarcoma Diseases 0.000 description 1
- 108090000331 Firefly luciferases Proteins 0.000 description 1
- 108010010803 Gelatin Proteins 0.000 description 1
- 206010071602 Genetic polymorphism Diseases 0.000 description 1
- ININBLZFFVOQIO-JHEQGTHGSA-N Gln-Thr-Gly Chemical compound C[C@H]([C@@H](C(=O)NCC(=O)O)NC(=O)[C@H](CCC(=O)N)N)O ININBLZFFVOQIO-JHEQGTHGSA-N 0.000 description 1
- FITIQFSXXBKFFM-NRPADANISA-N Gln-Val-Ser Chemical compound [H]N[C@@H](CCC(N)=O)C(=O)N[C@@H](C(C)C)C(=O)N[C@@H](CO)C(O)=O FITIQFSXXBKFFM-NRPADANISA-N 0.000 description 1
- CSMHMEATMDCQNY-DZKIICNBSA-N Gln-Val-Tyr Chemical compound [H]N[C@@H](CCC(N)=O)C(=O)N[C@@H](C(C)C)C(=O)N[C@@H](CC1=CC=C(O)C=C1)C(O)=O CSMHMEATMDCQNY-DZKIICNBSA-N 0.000 description 1
- IESFZVCAVACGPH-PEFMBERDSA-N Glu-Asp-Ile Chemical compound CC[C@H](C)[C@@H](C(O)=O)NC(=O)[C@H](CC(O)=O)NC(=O)[C@@H](N)CCC(O)=O IESFZVCAVACGPH-PEFMBERDSA-N 0.000 description 1
- NUSWUSKZRCGFEX-FXQIFTODSA-N Glu-Glu-Cys Chemical compound OC(=O)CC[C@H](N)C(=O)N[C@@H](CCC(O)=O)C(=O)N[C@@H](CS)C(O)=O NUSWUSKZRCGFEX-FXQIFTODSA-N 0.000 description 1
- LZMQSTPFYJLVJB-GUBZILKMSA-N Glu-Leu-Cys Chemical compound CC(C)C[C@@H](C(=O)N[C@@H](CS)C(=O)O)NC(=O)[C@H](CCC(=O)O)N LZMQSTPFYJLVJB-GUBZILKMSA-N 0.000 description 1
- IRXNJYPKBVERCW-DCAQKATOSA-N Glu-Leu-Glu Chemical compound [H]N[C@@H](CCC(O)=O)C(=O)N[C@@H](CC(C)C)C(=O)N[C@@H](CCC(O)=O)C(O)=O IRXNJYPKBVERCW-DCAQKATOSA-N 0.000 description 1
- SWRVAQHFBRZVNX-GUBZILKMSA-N Glu-Lys-Asn Chemical compound [H]N[C@@H](CCC(O)=O)C(=O)N[C@@H](CCCCN)C(=O)N[C@@H](CC(N)=O)C(O)=O SWRVAQHFBRZVNX-GUBZILKMSA-N 0.000 description 1
- QJVZSVUYZFYLFQ-CIUDSAMLSA-N Glu-Pro-Ala Chemical compound [H]N[C@@H](CCC(O)=O)C(=O)N1CCC[C@H]1C(=O)N[C@@H](C)C(O)=O QJVZSVUYZFYLFQ-CIUDSAMLSA-N 0.000 description 1
- BFEZQZKEPRKKHV-SRVKXCTJSA-N Glu-Pro-Lys Chemical compound C1C[C@H](N(C1)C(=O)[C@H](CCC(=O)O)N)C(=O)N[C@@H](CCCCN)C(=O)O BFEZQZKEPRKKHV-SRVKXCTJSA-N 0.000 description 1
- ZALGPUWUVHOGAE-GVXVVHGQSA-N Glu-Val-His Chemical compound CC(C)[C@@H](C(=O)N[C@@H](CC1=CN=CN1)C(=O)O)NC(=O)[C@H](CCC(=O)O)N ZALGPUWUVHOGAE-GVXVVHGQSA-N 0.000 description 1
- PYUCNHJQQVSPGN-BQBZGAKWSA-N Gly-Arg-Cys Chemical compound C(C[C@@H](C(=O)N[C@@H](CS)C(=O)O)NC(=O)CN)CN=C(N)N PYUCNHJQQVSPGN-BQBZGAKWSA-N 0.000 description 1
- AIJAPFVDBFYNKN-WHFBIAKZSA-N Gly-Asn-Asp Chemical compound C([C@@H](C(=O)N[C@@H](CC(=O)O)C(=O)O)NC(=O)CN)C(=O)N AIJAPFVDBFYNKN-WHFBIAKZSA-N 0.000 description 1
- PEZZSFLFXXFUQD-XPUUQOCRSA-N Gly-Cys-Val Chemical compound [H]NCC(=O)N[C@@H](CS)C(=O)N[C@@H](C(C)C)C(O)=O PEZZSFLFXXFUQD-XPUUQOCRSA-N 0.000 description 1
- GNPVTZJUUBPZKW-WDSKDSINSA-N Gly-Gln-Ser Chemical compound [H]NCC(=O)N[C@@H](CCC(N)=O)C(=O)N[C@@H](CO)C(O)=O GNPVTZJUUBPZKW-WDSKDSINSA-N 0.000 description 1
- SOEATRRYCIPEHA-BQBZGAKWSA-N Gly-Glu-Glu Chemical compound [H]NCC(=O)N[C@@H](CCC(O)=O)C(=O)N[C@@H](CCC(O)=O)C(O)=O SOEATRRYCIPEHA-BQBZGAKWSA-N 0.000 description 1
- IKAIKUBBJHFNBZ-LURJTMIESA-N Gly-Lys Chemical compound NCCCC[C@@H](C(O)=O)NC(=O)CN IKAIKUBBJHFNBZ-LURJTMIESA-N 0.000 description 1
- VBOBNHSVQKKTOT-YUMQZZPRSA-N Gly-Lys-Ala Chemical compound [H]NCC(=O)N[C@@H](CCCCN)C(=O)N[C@@H](C)C(O)=O VBOBNHSVQKKTOT-YUMQZZPRSA-N 0.000 description 1
- AFMOTCMSEBITOE-YEPSODPASA-N Gly-Val-Thr Chemical compound NCC(=O)N[C@@H](C(C)C)C(=O)N[C@@H]([C@@H](C)O)C(O)=O AFMOTCMSEBITOE-YEPSODPASA-N 0.000 description 1
- 229920000084 Gum arabic Polymers 0.000 description 1
- 101001030525 Homo sapiens Fibronectin type III and SPRY domain-containing protein 2 Proteins 0.000 description 1
- 101000869690 Homo sapiens Protein S100-A8 Proteins 0.000 description 1
- 102000008100 Human Serum Albumin Human genes 0.000 description 1
- 108091006905 Human Serum Albumin Proteins 0.000 description 1
- 206010020524 Hydronephrosis Diseases 0.000 description 1
- 208000029422 Hypernatremia Diseases 0.000 description 1
- 206010062767 Hypophysitis Diseases 0.000 description 1
- MKWSZEHGHSLNPF-NAKRPEOUSA-N Ile-Ala-Val Chemical compound CC[C@H](C)[C@@H](C(=O)N[C@@H](C)C(=O)N[C@@H](C(C)C)C(=O)O)N MKWSZEHGHSLNPF-NAKRPEOUSA-N 0.000 description 1
- WEWCEPOYKANMGZ-MMWGEVLESA-N Ile-Cys-Pro Chemical compound CC[C@H](C)[C@@H](C(=O)N[C@@H](CS)C(=O)N1CCC[C@@H]1C(=O)O)N WEWCEPOYKANMGZ-MMWGEVLESA-N 0.000 description 1
- UIEZQYNXCYHMQS-BJDJZHNGSA-N Ile-Lys-Ala Chemical compound CC[C@H](C)[C@@H](C(=O)N[C@@H](CCCCN)C(=O)N[C@@H](C)C(=O)O)N UIEZQYNXCYHMQS-BJDJZHNGSA-N 0.000 description 1
- DGAQECJNVWCQMB-PUAWFVPOSA-M Ilexoside XXIX Chemical compound C[C@@H]1CC[C@@]2(CC[C@@]3(C(=CC[C@H]4[C@]3(CC[C@@H]5[C@@]4(CC[C@@H](C5(C)C)OS(=O)(=O)[O-])C)C)[C@@H]2[C@]1(C)O)C)C(=O)O[C@H]6[C@@H]([C@H]([C@@H]([C@H](O6)CO)O)O)O.[Na+] DGAQECJNVWCQMB-PUAWFVPOSA-M 0.000 description 1
- 208000026350 Inborn Genetic disease Diseases 0.000 description 1
- ONIBWKKTOPOVIA-BYPYZUCNSA-N L-Proline Chemical compound OC(=O)[C@@H]1CCCN1 ONIBWKKTOPOVIA-BYPYZUCNSA-N 0.000 description 1
- QNAYBMKLOCPYGJ-REOHCLBHSA-N L-alanine Chemical compound C[C@H](N)C(O)=O QNAYBMKLOCPYGJ-REOHCLBHSA-N 0.000 description 1
- HNDVDQJCIGZPNO-YFKPBYRVSA-N L-histidine Chemical compound OC(=O)[C@@H](N)CC1=CN=CN1 HNDVDQJCIGZPNO-YFKPBYRVSA-N 0.000 description 1
- AGPKZVBTJJNPAG-WHFBIAKZSA-N L-isoleucine Chemical compound CC[C@H](C)[C@H](N)C(O)=O AGPKZVBTJJNPAG-WHFBIAKZSA-N 0.000 description 1
- ROHFNLRQFUQHCH-YFKPBYRVSA-N L-leucine Chemical compound CC(C)C[C@H](N)C(O)=O ROHFNLRQFUQHCH-YFKPBYRVSA-N 0.000 description 1
- FFEARJCKVFRZRR-BYPYZUCNSA-N L-methionine Chemical compound CSCC[C@H](N)C(O)=O FFEARJCKVFRZRR-BYPYZUCNSA-N 0.000 description 1
- COLNVLDHVKWLRT-QMMMGPOBSA-N L-phenylalanine Chemical compound OC(=O)[C@@H](N)CC1=CC=CC=C1 COLNVLDHVKWLRT-QMMMGPOBSA-N 0.000 description 1
- QIVBCDIJIAJPQS-VIFPVBQESA-N L-tryptophane Chemical compound C1=CC=C2C(C[C@H](N)C(O)=O)=CNC2=C1 QIVBCDIJIAJPQS-VIFPVBQESA-N 0.000 description 1
- OUYCCCASQSFEME-QMMMGPOBSA-N L-tyrosine Chemical compound OC(=O)[C@@H](N)CC1=CC=C(O)C=C1 OUYCCCASQSFEME-QMMMGPOBSA-N 0.000 description 1
- KZSNJWFQEVHDMF-BYPYZUCNSA-N L-valine Chemical compound CC(C)[C@H](N)C(O)=O KZSNJWFQEVHDMF-BYPYZUCNSA-N 0.000 description 1
- ULXYQAJWJGLCNR-YUMQZZPRSA-N Leu-Asp-Gly Chemical compound CC(C)C[C@H](N)C(=O)N[C@@H](CC(O)=O)C(=O)NCC(O)=O ULXYQAJWJGLCNR-YUMQZZPRSA-N 0.000 description 1
- VWHGTYCRDRBSFI-ZETCQYMHSA-N Leu-Gly-Gly Chemical compound CC(C)C[C@H](N)C(=O)NCC(=O)NCC(O)=O VWHGTYCRDRBSFI-ZETCQYMHSA-N 0.000 description 1
- YOKVEHGYYQEQOP-QWRGUYRKSA-N Leu-Leu-Gly Chemical compound CC(C)C[C@H](N)C(=O)N[C@@H](CC(C)C)C(=O)NCC(O)=O YOKVEHGYYQEQOP-QWRGUYRKSA-N 0.000 description 1
- AUNMOHYWTAPQLA-XUXIUFHCSA-N Leu-Met-Ile Chemical compound [H]N[C@@H](CC(C)C)C(=O)N[C@@H](CCSC)C(=O)N[C@@H]([C@@H](C)CC)C(O)=O AUNMOHYWTAPQLA-XUXIUFHCSA-N 0.000 description 1
- WMIOEVKKYIMVKI-DCAQKATOSA-N Leu-Pro-Ala Chemical compound [H]N[C@@H](CC(C)C)C(=O)N1CCC[C@H]1C(=O)N[C@@H](C)C(O)=O WMIOEVKKYIMVKI-DCAQKATOSA-N 0.000 description 1
- SBANPBVRHYIMRR-GARJFASQSA-N Leu-Ser-Pro Chemical compound CC(C)C[C@@H](C(=O)N[C@@H](CO)C(=O)N1CCC[C@@H]1C(=O)O)N SBANPBVRHYIMRR-GARJFASQSA-N 0.000 description 1
- ROHFNLRQFUQHCH-UHFFFAOYSA-N Leucine Natural products CC(C)CC(N)C(O)=O ROHFNLRQFUQHCH-UHFFFAOYSA-N 0.000 description 1
- 201000009342 Limb-girdle muscular dystrophy Diseases 0.000 description 1
- 208000007623 Lordosis Diseases 0.000 description 1
- 239000005089 Luciferase Substances 0.000 description 1
- VHFFQUSNFFIZBT-CIUDSAMLSA-N Lys-Ala-Cys Chemical compound C[C@@H](C(=O)N[C@@H](CS)C(=O)O)NC(=O)[C@H](CCCCN)N VHFFQUSNFFIZBT-CIUDSAMLSA-N 0.000 description 1
- YKIRNDPUWONXQN-GUBZILKMSA-N Lys-Asn-Gln Chemical compound C(CCN)C[C@@H](C(=O)N[C@@H](CC(=O)N)C(=O)N[C@@H](CCC(=O)N)C(=O)O)N YKIRNDPUWONXQN-GUBZILKMSA-N 0.000 description 1
- ZQCVMVCVPFYXHZ-SRVKXCTJSA-N Lys-Asn-Lys Chemical compound NCCCC[C@H](N)C(=O)N[C@@H](CC(N)=O)C(=O)N[C@H](C(O)=O)CCCCN ZQCVMVCVPFYXHZ-SRVKXCTJSA-N 0.000 description 1
- KWUKZRFFKPLUPE-HJGDQZAQSA-N Lys-Asp-Thr Chemical compound [H]N[C@@H](CCCCN)C(=O)N[C@@H](CC(O)=O)C(=O)N[C@@H]([C@@H](C)O)C(O)=O KWUKZRFFKPLUPE-HJGDQZAQSA-N 0.000 description 1
- RFQATBGBLDAKGI-VHSXEESVSA-N Lys-Gly-Pro Chemical compound C1C[C@@H](N(C1)C(=O)CNC(=O)[C@H](CCCCN)N)C(=O)O RFQATBGBLDAKGI-VHSXEESVSA-N 0.000 description 1
- NVGBPTNZLWRQSY-UWVGGRQHSA-N Lys-Lys Chemical compound NCCCC[C@H](N)C(=O)N[C@H](C(O)=O)CCCCN NVGBPTNZLWRQSY-UWVGGRQHSA-N 0.000 description 1
- OBZHNHBAAVEWKI-DCAQKATOSA-N Lys-Pro-Asn Chemical compound NCCCC[C@H](N)C(=O)N1CCC[C@H]1C(=O)N[C@@H](CC(N)=O)C(O)=O OBZHNHBAAVEWKI-DCAQKATOSA-N 0.000 description 1
- MGKFCQFVPKOWOL-CIUDSAMLSA-N Lys-Ser-Asp Chemical compound C(CCN)C[C@@H](C(=O)N[C@@H](CO)C(=O)N[C@@H](CC(=O)O)C(=O)O)N MGKFCQFVPKOWOL-CIUDSAMLSA-N 0.000 description 1
- CTJUSALVKAWFFU-CIUDSAMLSA-N Lys-Ser-Cys Chemical compound C(CCN)C[C@@H](C(=O)N[C@@H](CO)C(=O)N[C@@H](CS)C(=O)O)N CTJUSALVKAWFFU-CIUDSAMLSA-N 0.000 description 1
- IOQWIOPSKJOEKI-SRVKXCTJSA-N Lys-Ser-Leu Chemical compound [H]N[C@@H](CCCCN)C(=O)N[C@@H](CO)C(=O)N[C@@H](CC(C)C)C(O)=O IOQWIOPSKJOEKI-SRVKXCTJSA-N 0.000 description 1
- ZVXSESPJMKNIQA-YXMSTPNBSA-N Lys-Thr-Pro-Pro Chemical compound NCCCC[C@H](N)C(=O)N[C@@H]([C@H](O)C)C(=O)N1CCC[C@H]1C(=O)N1[C@H](C(O)=O)CCC1 ZVXSESPJMKNIQA-YXMSTPNBSA-N 0.000 description 1
- 208000036626 Mental retardation Diseases 0.000 description 1
- 208000034819 Mobility Limitation Diseases 0.000 description 1
- 101000822667 Mus musculus Something about silencing protein 10 Proteins 0.000 description 1
- 206010028293 Muscle contractions involuntary Diseases 0.000 description 1
- 206010062575 Muscle contracture Diseases 0.000 description 1
- 206010028311 Muscle hypertrophy Diseases 0.000 description 1
- 208000021642 Muscular disease Diseases 0.000 description 1
- 201000009623 Myopathy Diseases 0.000 description 1
- 102000003505 Myosin Human genes 0.000 description 1
- 108060008487 Myosin Proteins 0.000 description 1
- KZNQNBZMBZJQJO-UHFFFAOYSA-N N-glycyl-L-proline Natural products NCC(=O)N1CCCC1C(O)=O KZNQNBZMBZJQJO-UHFFFAOYSA-N 0.000 description 1
- 206010028980 Neoplasm Diseases 0.000 description 1
- 101100068676 Neurospora crassa (strain ATCC 24698 / 74-OR23-1A / CBS 708.71 / DSM 1257 / FGSC 987) gln-1 gene Proteins 0.000 description 1
- 101000690429 Panax ginseng Floral homeotic protein AGAMOUS Proteins 0.000 description 1
- 241000009328 Perro Species 0.000 description 1
- FZBGMXYQPACKNC-HJWJTTGWSA-N Phe-Pro-Ile Chemical compound [H]N[C@@H](CC1=CC=CC=C1)C(=O)N1CCC[C@H]1C(=O)N[C@@H]([C@@H](C)CC)C(O)=O FZBGMXYQPACKNC-HJWJTTGWSA-N 0.000 description 1
- IIEOLPMQYRBZCN-SRVKXCTJSA-N Phe-Ser-Cys Chemical compound N[C@@H](CC1=CC=CC=C1)C(=O)N[C@@H](CO)C(=O)N[C@@H](CS)C(=O)O IIEOLPMQYRBZCN-SRVKXCTJSA-N 0.000 description 1
- CXOFVDLJLONNDW-UHFFFAOYSA-N Phenytoin Chemical compound N1C(=O)NC(=O)C1(C=1C=CC=CC=1)C1=CC=CC=C1 CXOFVDLJLONNDW-UHFFFAOYSA-N 0.000 description 1
- 206010035226 Plasma cell myeloma Diseases 0.000 description 1
- 239000002202 Polyethylene glycol Substances 0.000 description 1
- ZLMJMSJWJFRBEC-UHFFFAOYSA-N Potassium Chemical compound [K] ZLMJMSJWJFRBEC-UHFFFAOYSA-N 0.000 description 1
- ZCXQTRXYZOSGJR-FXQIFTODSA-N Pro-Asp-Ser Chemical compound [H]N1CCC[C@H]1C(=O)N[C@@H](CC(O)=O)C(=O)N[C@@H](CO)C(O)=O ZCXQTRXYZOSGJR-FXQIFTODSA-N 0.000 description 1
- LGSANCBHSMDFDY-GARJFASQSA-N Pro-Glu-Pro Chemical compound C1C[C@H](NC1)C(=O)N[C@@H](CCC(=O)O)C(=O)N2CCC[C@@H]2C(=O)O LGSANCBHSMDFDY-GARJFASQSA-N 0.000 description 1
- KWMUAKQOVYCQJQ-ZPFDUUQYSA-N Pro-Ile-Glu Chemical compound CC[C@H](C)[C@@H](C(=O)N[C@@H](CCC(=O)O)C(=O)O)NC(=O)[C@@H]1CCCN1 KWMUAKQOVYCQJQ-ZPFDUUQYSA-N 0.000 description 1
- ZLXKLMHAMDENIO-DCAQKATOSA-N Pro-Lys-Asp Chemical compound [H]N1CCC[C@H]1C(=O)N[C@@H](CCCCN)C(=O)N[C@@H](CC(O)=O)C(O)=O ZLXKLMHAMDENIO-DCAQKATOSA-N 0.000 description 1
- GMJDSFYVTAMIBF-FXQIFTODSA-N Pro-Ser-Asp Chemical compound [H]N1CCC[C@H]1C(=O)N[C@@H](CO)C(=O)N[C@@H](CC(O)=O)C(O)=O GMJDSFYVTAMIBF-FXQIFTODSA-N 0.000 description 1
- 102100032442 Protein S100-A8 Human genes 0.000 description 1
- 208000035955 Proximal myotonic myopathy Diseases 0.000 description 1
- 241000700157 Rattus norvegicus Species 0.000 description 1
- 208000004756 Respiratory Insufficiency Diseases 0.000 description 1
- 108091027981 Response element Proteins 0.000 description 1
- 241000283984 Rodentia Species 0.000 description 1
- 240000004808 Saccharomyces cerevisiae Species 0.000 description 1
- 101000828148 Saccharomyces cerevisiae (strain ATCC 204508 / S288c) Transposon Ty3-G Gag polyprotein Proteins 0.000 description 1
- 101000790437 Saccharomyces cerevisiae (strain ATCC 204508 / S288c) Transposon Ty3-I Gag polyprotein Proteins 0.000 description 1
- 206010039424 Salivary hypersecretion Diseases 0.000 description 1
- KNCJWSPMTFFJII-ZLUOBGJFSA-N Ser-Cys-Asp Chemical compound [H]N[C@@H](CO)C(=O)N[C@@H](CS)C(=O)N[C@@H](CC(O)=O)C(O)=O KNCJWSPMTFFJII-ZLUOBGJFSA-N 0.000 description 1
- MOVJSUIKUNCVMG-ZLUOBGJFSA-N Ser-Cys-Ser Chemical compound C([C@@H](C(=O)N[C@@H](CS)C(=O)N[C@@H](CO)C(=O)O)N)O MOVJSUIKUNCVMG-ZLUOBGJFSA-N 0.000 description 1
- HJEBZBMOTCQYDN-ACZMJKKPSA-N Ser-Glu-Asp Chemical compound [H]N[C@@H](CO)C(=O)N[C@@H](CCC(O)=O)C(=O)N[C@@H](CC(O)=O)C(O)=O HJEBZBMOTCQYDN-ACZMJKKPSA-N 0.000 description 1
- HMRAQFJFTOLDKW-GUBZILKMSA-N Ser-His-Glu Chemical compound [H]N[C@@H](CO)C(=O)N[C@@H](CC1=CNC=N1)C(=O)N[C@@H](CCC(O)=O)C(O)=O HMRAQFJFTOLDKW-GUBZILKMSA-N 0.000 description 1
- XUDRHBPSPAPDJP-SRVKXCTJSA-N Ser-Lys-Leu Chemical compound CC(C)C[C@@H](C(O)=O)NC(=O)[C@H](CCCCN)NC(=O)[C@@H](N)CO XUDRHBPSPAPDJP-SRVKXCTJSA-N 0.000 description 1
- FPCGZYMRFFIYIH-CIUDSAMLSA-N Ser-Lys-Ser Chemical compound [H]N[C@@H](CO)C(=O)N[C@@H](CCCCN)C(=O)N[C@@H](CO)C(O)=O FPCGZYMRFFIYIH-CIUDSAMLSA-N 0.000 description 1
- NUEHQDHDLDXCRU-GUBZILKMSA-N Ser-Pro-Arg Chemical compound OC[C@H](N)C(=O)N1CCC[C@H]1C(=O)N[C@@H](CCCN=C(N)N)C(O)=O NUEHQDHDLDXCRU-GUBZILKMSA-N 0.000 description 1
- GYDFRTRSSXOZCR-ACZMJKKPSA-N Ser-Ser-Glu Chemical compound OC[C@H](N)C(=O)N[C@@H](CO)C(=O)N[C@H](C(O)=O)CCC(O)=O GYDFRTRSSXOZCR-ACZMJKKPSA-N 0.000 description 1
- PYTKULIABVRXSC-BWBBJGPYSA-N Ser-Ser-Thr Chemical compound [H]N[C@@H](CO)C(=O)N[C@@H](CO)C(=O)N[C@@H]([C@@H](C)O)C(O)=O PYTKULIABVRXSC-BWBBJGPYSA-N 0.000 description 1
- RCOUFINCYASMDN-GUBZILKMSA-N Ser-Val-Met Chemical compound [H]N[C@@H](CO)C(=O)N[C@@H](C(C)C)C(=O)N[C@@H](CCSC)C(O)=O RCOUFINCYASMDN-GUBZILKMSA-N 0.000 description 1
- MTCFGRXMJLQNBG-UHFFFAOYSA-N Serine Natural products OCC(N)C(O)=O MTCFGRXMJLQNBG-UHFFFAOYSA-N 0.000 description 1
- VMHLLURERBWHNL-UHFFFAOYSA-M Sodium acetate Chemical compound [Na+].CC([O-])=O VMHLLURERBWHNL-UHFFFAOYSA-M 0.000 description 1
- 229910000831 Steel Inorganic materials 0.000 description 1
- 208000037140 Steinert myotonic dystrophy Diseases 0.000 description 1
- 108010090804 Streptavidin Proteins 0.000 description 1
- 229930006000 Sucrose Natural products 0.000 description 1
- CZMRCDWAGMRECN-UGDNZRGBSA-N Sucrose Chemical compound O[C@H]1[C@H](O)[C@@H](CO)O[C@@]1(CO)O[C@@H]1[C@H](O)[C@@H](O)[C@H](O)[C@@H](CO)O1 CZMRCDWAGMRECN-UGDNZRGBSA-N 0.000 description 1
- 206010042434 Sudden death Diseases 0.000 description 1
- FEWJPZIEWOKRBE-UHFFFAOYSA-N Tartaric acid Natural products [H+].[H+].[O-]C(=O)C(O)C(O)C([O-])=O FEWJPZIEWOKRBE-UHFFFAOYSA-N 0.000 description 1
- 208000000491 Tendinopathy Diseases 0.000 description 1
- FQPQPTHMHZKGFM-XQXXSGGOSA-N Thr-Ala-Glu Chemical compound [H]N[C@@H]([C@@H](C)O)C(=O)N[C@@H](C)C(=O)N[C@@H](CCC(O)=O)C(O)=O FQPQPTHMHZKGFM-XQXXSGGOSA-N 0.000 description 1
- ODSAPYVQSLDRSR-LKXGYXEUSA-N Thr-Cys-Asn Chemical compound [H]N[C@@H]([C@@H](C)O)C(=O)N[C@@H](CS)C(=O)N[C@@H](CC(N)=O)C(O)=O ODSAPYVQSLDRSR-LKXGYXEUSA-N 0.000 description 1
- UDQBCBUXAQIZAK-GLLZPBPUSA-N Thr-Glu-Glu Chemical compound [H]N[C@@H]([C@@H](C)O)C(=O)N[C@@H](CCC(O)=O)C(=O)N[C@@H](CCC(O)=O)C(O)=O UDQBCBUXAQIZAK-GLLZPBPUSA-N 0.000 description 1
- FIFDDJFLNVAVMS-RHYQMDGZSA-N Thr-Leu-Met Chemical compound [H]N[C@@H]([C@@H](C)O)C(=O)N[C@@H](CC(C)C)C(=O)N[C@@H](CCSC)C(O)=O FIFDDJFLNVAVMS-RHYQMDGZSA-N 0.000 description 1
- BDGBHYCAZJPLHX-HJGDQZAQSA-N Thr-Lys-Asn Chemical compound [H]N[C@@H]([C@@H](C)O)C(=O)N[C@@H](CCCCN)C(=O)N[C@@H](CC(N)=O)C(O)=O BDGBHYCAZJPLHX-HJGDQZAQSA-N 0.000 description 1
- MROIJTGJGIDEEJ-RCWTZXSCSA-N Thr-Pro-Pro Chemical compound C[C@@H](O)[C@H](N)C(=O)N1CCC[C@H]1C(=O)N1[C@H](C(O)=O)CCC1 MROIJTGJGIDEEJ-RCWTZXSCSA-N 0.000 description 1
- XHWCDRUPDNSDAZ-XKBZYTNZSA-N Thr-Ser-Glu Chemical compound C[C@H]([C@@H](C(=O)N[C@@H](CO)C(=O)N[C@@H](CCC(=O)O)C(=O)O)N)O XHWCDRUPDNSDAZ-XKBZYTNZSA-N 0.000 description 1
- FYBFTPLPAXZBOY-KKHAAJSZSA-N Thr-Val-Asp Chemical compound [H]N[C@@H]([C@@H](C)O)C(=O)N[C@@H](C(C)C)C(=O)N[C@@H](CC(O)=O)C(O)=O FYBFTPLPAXZBOY-KKHAAJSZSA-N 0.000 description 1
- QIVBCDIJIAJPQS-UHFFFAOYSA-N Tryptophan Natural products C1=CC=C2C(CC(N)C(O)=O)=CNC2=C1 QIVBCDIJIAJPQS-UHFFFAOYSA-N 0.000 description 1
- LGEYOIQBBIPHQN-UWJYBYFXSA-N Tyr-Ala-Ser Chemical compound OC[C@@H](C(O)=O)NC(=O)[C@H](C)NC(=O)[C@@H](N)CC1=CC=C(O)C=C1 LGEYOIQBBIPHQN-UWJYBYFXSA-N 0.000 description 1
- UMSZZGTXGKHTFJ-SRVKXCTJSA-N Tyr-Ser-Ser Chemical compound OC[C@@H](C(O)=O)NC(=O)[C@H](CO)NC(=O)[C@@H](N)CC1=CC=C(O)C=C1 UMSZZGTXGKHTFJ-SRVKXCTJSA-N 0.000 description 1
- 102220613750 Uncharacterized protein C19orf84_K76T_mutation Human genes 0.000 description 1
- 244000000188 Vaccinium ovalifolium Species 0.000 description 1
- QHDXUYOYTPWCSK-RCOVLWMOSA-N Val-Asp-Gly Chemical compound CC(C)[C@@H](C(=O)N[C@@H](CC(=O)O)C(=O)NCC(=O)O)N QHDXUYOYTPWCSK-RCOVLWMOSA-N 0.000 description 1
- SCBITHMBEJNRHC-LSJOCFKGSA-N Val-Asp-Val Chemical compound CC(C)[C@@H](C(=O)N[C@@H](CC(=O)O)C(=O)N[C@@H](C(C)C)C(=O)O)N SCBITHMBEJNRHC-LSJOCFKGSA-N 0.000 description 1
- CWSIBTLMMQLPPZ-FXQIFTODSA-N Val-Cys-Ala Chemical compound C[C@@H](C(=O)O)NC(=O)[C@H](CS)NC(=O)[C@H](C(C)C)N CWSIBTLMMQLPPZ-FXQIFTODSA-N 0.000 description 1
- FPCIBLUVDNXPJO-XPUUQOCRSA-N Val-Cys-Gly Chemical compound CC(C)[C@H](N)C(=O)N[C@@H](CS)C(=O)NCC(O)=O FPCIBLUVDNXPJO-XPUUQOCRSA-N 0.000 description 1
- XXDVDTMEVBYRPK-XPUUQOCRSA-N Val-Gln Chemical compound CC(C)[C@H](N)C(=O)N[C@H](C(O)=O)CCC(N)=O XXDVDTMEVBYRPK-XPUUQOCRSA-N 0.000 description 1
- RKIGNDAHUOOIMJ-BQFCYCMXSA-N Val-Glu-Trp Chemical compound C1=CC=C2C(C[C@H](NC(=O)[C@H](CCC(O)=O)NC(=O)[C@@H](N)C(C)C)C(O)=O)=CNC2=C1 RKIGNDAHUOOIMJ-BQFCYCMXSA-N 0.000 description 1
- UEHRGZCNLSWGHK-DLOVCJGASA-N Val-Glu-Val Chemical compound CC(C)[C@H](N)C(=O)N[C@@H](CCC(O)=O)C(=O)N[C@@H](C(C)C)C(O)=O UEHRGZCNLSWGHK-DLOVCJGASA-N 0.000 description 1
- SBJCTAZFSZXWSR-AVGNSLFASA-N Val-Met-His Chemical compound CC(C)[C@@H](C(=O)N[C@@H](CCSC)C(=O)N[C@@H](CC1=CN=CN1)C(=O)O)N SBJCTAZFSZXWSR-AVGNSLFASA-N 0.000 description 1
- VHIZXDZMTDVFGX-DCAQKATOSA-N Val-Ser-Leu Chemical compound CC(C)C[C@@H](C(=O)O)NC(=O)[C@H](CO)NC(=O)[C@H](C(C)C)N VHIZXDZMTDVFGX-DCAQKATOSA-N 0.000 description 1
- 208000027418 Wounds and injury Diseases 0.000 description 1
- 210000001015 abdomen Anatomy 0.000 description 1
- 230000003187 abdominal effect Effects 0.000 description 1
- 239000000205 acacia gum Substances 0.000 description 1
- 235000010489 acacia gum Nutrition 0.000 description 1
- 235000011054 acetic acid Nutrition 0.000 description 1
- 230000003213 activating effect Effects 0.000 description 1
- 238000011360 adjunctive therapy Methods 0.000 description 1
- 239000002671 adjuvant Substances 0.000 description 1
- 235000004279 alanine Nutrition 0.000 description 1
- 125000003295 alanine group Chemical group N[C@@H](C)C(=O)* 0.000 description 1
- 108010087924 alanylproline Proteins 0.000 description 1
- 208000026935 allergic disease Diseases 0.000 description 1
- 229910000147 aluminium phosphate Inorganic materials 0.000 description 1
- 125000003277 amino group Chemical group 0.000 description 1
- 230000033115 angiogenesis Effects 0.000 description 1
- 239000000427 antigen Substances 0.000 description 1
- 108091007433 antigens Proteins 0.000 description 1
- 102000036639 antigens Human genes 0.000 description 1
- 210000000436 anus Anatomy 0.000 description 1
- 238000013459 approach Methods 0.000 description 1
- 239000012062 aqueous buffer Substances 0.000 description 1
- ODKSFYDXXFIFQN-UHFFFAOYSA-N arginine Natural products OC(=O)C(N)CCCNC(N)=N ODKSFYDXXFIFQN-UHFFFAOYSA-N 0.000 description 1
- 206010003119 arrhythmia Diseases 0.000 description 1
- 230000006793 arrhythmia Effects 0.000 description 1
- 125000003118 aryl group Chemical group 0.000 description 1
- 235000009582 asparagine Nutrition 0.000 description 1
- 229960001230 asparagine Drugs 0.000 description 1
- 230000000386 athletic effect Effects 0.000 description 1
- QVGXLLKOCUKJST-UHFFFAOYSA-N atomic oxygen Chemical compound [O] QVGXLLKOCUKJST-UHFFFAOYSA-N 0.000 description 1
- 210000004227 basal ganglia Anatomy 0.000 description 1
- 230000003542 behavioural effect Effects 0.000 description 1
- 238000005452 bending Methods 0.000 description 1
- WQZGKKKJIJFFOK-VFUOTHLCSA-N beta-D-glucose Chemical compound OC[C@H]1O[C@@H](O)[C@H](O)[C@@H](O)[C@@H]1O WQZGKKKJIJFFOK-VFUOTHLCSA-N 0.000 description 1
- 230000008827 biological function Effects 0.000 description 1
- 230000033228 biological regulation Effects 0.000 description 1
- 230000036770 blood supply Effects 0.000 description 1
- 210000004556 brain Anatomy 0.000 description 1
- 210000000481 breast Anatomy 0.000 description 1
- 239000007853 buffer solution Substances 0.000 description 1
- 239000007975 buffered saline Substances 0.000 description 1
- 230000003139 buffering effect Effects 0.000 description 1
- 210000001217 buttock Anatomy 0.000 description 1
- 102220397411 c.231G>C Human genes 0.000 description 1
- 239000011575 calcium Substances 0.000 description 1
- 229910052791 calcium Inorganic materials 0.000 description 1
- 239000002775 capsule Substances 0.000 description 1
- 239000004202 carbamide Substances 0.000 description 1
- 235000014633 carbohydrates Nutrition 0.000 description 1
- 230000004706 cardiovascular dysfunction Effects 0.000 description 1
- BGGXRVPCJUKHTQ-AHCAJXDVSA-L carumonam sodium Chemical compound [Na+].[Na+].O=C1N(S([O-])(=O)=O)[C@H](COC(=O)N)[C@@H]1NC(=O)C(=N/OCC([O-])=O)\C1=CSC(N)=N1 BGGXRVPCJUKHTQ-AHCAJXDVSA-L 0.000 description 1
- 238000000423 cell based assay Methods 0.000 description 1
- 238000004113 cell culture Methods 0.000 description 1
- 210000000170 cell membrane Anatomy 0.000 description 1
- 230000004663 cell proliferation Effects 0.000 description 1
- 230000015861 cell surface binding Effects 0.000 description 1
- 230000019522 cellular metabolic process Effects 0.000 description 1
- 239000001913 cellulose Substances 0.000 description 1
- 229920002678 cellulose Polymers 0.000 description 1
- 210000003169 central nervous system Anatomy 0.000 description 1
- 238000006243 chemical reaction Methods 0.000 description 1
- 239000003153 chemical reaction reagent Substances 0.000 description 1
- 230000001055 chewing effect Effects 0.000 description 1
- LOUPRKONTZGTKE-UHFFFAOYSA-N cinchonine Natural products C1C(C(C2)C=C)CCN2C1C(O)C1=CC=NC2=CC=C(OC)C=C21 LOUPRKONTZGTKE-UHFFFAOYSA-N 0.000 description 1
- 238000000576 coating method Methods 0.000 description 1
- 210000003477 cochlea Anatomy 0.000 description 1
- 229940110456 cocoa butter Drugs 0.000 description 1
- 235000019868 cocoa butter Nutrition 0.000 description 1
- 230000019771 cognition Effects 0.000 description 1
- 230000003930 cognitive ability Effects 0.000 description 1
- 230000001149 cognitive effect Effects 0.000 description 1
- 239000003086 colorant Substances 0.000 description 1
- 238000002648 combination therapy Methods 0.000 description 1
- 239000012141 concentrate Substances 0.000 description 1
- 239000000470 constituent Substances 0.000 description 1
- 230000008602 contraction Effects 0.000 description 1
- 208000006111 contracture Diseases 0.000 description 1
- VFLDPWHFBUODDF-FCXRPNKRSA-N curcumin Chemical compound C1=C(O)C(OC)=CC(\C=C\C(=O)CC(=O)\C=C\C=2C=C(OC)C(O)=CC=2)=C1 VFLDPWHFBUODDF-FCXRPNKRSA-N 0.000 description 1
- 235000018417 cysteine Nutrition 0.000 description 1
- XUJNEKJLAYXESH-UHFFFAOYSA-N cysteine Natural products SCC(N)C(O)=O XUJNEKJLAYXESH-UHFFFAOYSA-N 0.000 description 1
- 230000001086 cytosolic effect Effects 0.000 description 1
- 230000007850 degeneration Effects 0.000 description 1
- 230000001934 delay Effects 0.000 description 1
- 230000001419 dependent effect Effects 0.000 description 1
- 238000013461 design Methods 0.000 description 1
- 230000001627 detrimental effect Effects 0.000 description 1
- 235000019425 dextrin Nutrition 0.000 description 1
- 239000008121 dextrose Substances 0.000 description 1
- 235000014113 dietary fatty acids Nutrition 0.000 description 1
- 108020001096 dihydrofolate reductase Proteins 0.000 description 1
- 238000010790 dilution Methods 0.000 description 1
- 239000012895 dilution Substances 0.000 description 1
- 239000013024 dilution buffer Substances 0.000 description 1
- LOKCTEFSRHRXRJ-UHFFFAOYSA-I dipotassium trisodium dihydrogen phosphate hydrogen phosphate dichloride Chemical compound P(=O)(O)(O)[O-].[K+].P(=O)(O)([O-])[O-].[Na+].[Na+].[Cl-].[K+].[Cl-].[Na+] LOKCTEFSRHRXRJ-UHFFFAOYSA-I 0.000 description 1
- 239000007884 disintegrant Substances 0.000 description 1
- 239000002270 dispersing agent Substances 0.000 description 1
- 239000006185 dispersion Substances 0.000 description 1
- 239000002612 dispersion medium Substances 0.000 description 1
- 231100000673 dose–response relationship Toxicity 0.000 description 1
- 229940079593 drug Drugs 0.000 description 1
- 229940088679 drug related substance Drugs 0.000 description 1
- 230000002183 duodenal effect Effects 0.000 description 1
- 206010013932 dyslexia Diseases 0.000 description 1
- 208000024732 dysthymic disease Diseases 0.000 description 1
- 208000010118 dystonia Diseases 0.000 description 1
- 235000013601 eggs Nutrition 0.000 description 1
- 230000008030 elimination Effects 0.000 description 1
- 238000003379 elimination reaction Methods 0.000 description 1
- 239000000839 emulsion Substances 0.000 description 1
- 230000002708 enhancing effect Effects 0.000 description 1
- 210000003238 esophagus Anatomy 0.000 description 1
- 230000001747 exhibiting effect Effects 0.000 description 1
- 210000004709 eyebrow Anatomy 0.000 description 1
- 210000001097 facial muscle Anatomy 0.000 description 1
- 208000008570 facioscapulohumeral muscular dystrophy Diseases 0.000 description 1
- 229930195729 fatty acid Natural products 0.000 description 1
- 239000000194 fatty acid Substances 0.000 description 1
- 230000001605 fetal effect Effects 0.000 description 1
- 235000003599 food sweetener Nutrition 0.000 description 1
- 210000004744 fore-foot Anatomy 0.000 description 1
- 210000000245 forearm Anatomy 0.000 description 1
- 230000006097 fructosylation Effects 0.000 description 1
- 230000005021 gait Effects 0.000 description 1
- 210000001035 gastrointestinal tract Anatomy 0.000 description 1
- 229920000159 gelatin Polymers 0.000 description 1
- 239000008273 gelatin Substances 0.000 description 1
- 235000019322 gelatine Nutrition 0.000 description 1
- 235000011852 gelatine desserts Nutrition 0.000 description 1
- 208000016361 genetic disease Diseases 0.000 description 1
- 239000003862 glucocorticoid Substances 0.000 description 1
- ZDXPYRJPNDTMRX-UHFFFAOYSA-N glutamine Natural products OC(=O)C(N)CCC(N)=O ZDXPYRJPNDTMRX-UHFFFAOYSA-N 0.000 description 1
- 150000004676 glycans Chemical class 0.000 description 1
- 230000036252 glycation Effects 0.000 description 1
- 235000011187 glycerol Nutrition 0.000 description 1
- 230000012010 growth Effects 0.000 description 1
- 230000036541 health Effects 0.000 description 1
- 244000144980 herd Species 0.000 description 1
- 239000013628 high molecular weight specie Substances 0.000 description 1
- 235000011167 hydrochloric acid Nutrition 0.000 description 1
- 239000001257 hydrogen Substances 0.000 description 1
- 229910052739 hydrogen Inorganic materials 0.000 description 1
- 230000003301 hydrolyzing effect Effects 0.000 description 1
- 125000001165 hydrophobic group Chemical group 0.000 description 1
- 150000004679 hydroxides Chemical class 0.000 description 1
- 229920003063 hydroxymethyl cellulose Polymers 0.000 description 1
- 229940031574 hydroxymethyl cellulose Drugs 0.000 description 1
- 230000037417 hyperactivation Effects 0.000 description 1
- 206010020718 hyperplasia Diseases 0.000 description 1
- 238000000099 in vitro assay Methods 0.000 description 1
- 238000010874 in vitro model Methods 0.000 description 1
- 238000005462 in vivo assay Methods 0.000 description 1
- 230000001939 inductive effect Effects 0.000 description 1
- 239000003701 inert diluent Substances 0.000 description 1
- 230000000977 initiatory effect Effects 0.000 description 1
- 208000014674 injury Diseases 0.000 description 1
- 230000003434 inspiratory effect Effects 0.000 description 1
- 230000003834 intracellular effect Effects 0.000 description 1
- 230000001788 irregular Effects 0.000 description 1
- 230000007794 irritation Effects 0.000 description 1
- 230000000302 ischemic effect Effects 0.000 description 1
- AGPKZVBTJJNPAG-UHFFFAOYSA-N isoleucine Natural products CCC(C)C(N)C(O)=O AGPKZVBTJJNPAG-UHFFFAOYSA-N 0.000 description 1
- 229960000310 isoleucine Drugs 0.000 description 1
- JJWLVOIRVHMVIS-UHFFFAOYSA-N isopropylamine Chemical compound CC(C)N JJWLVOIRVHMVIS-UHFFFAOYSA-N 0.000 description 1
- 239000007951 isotonicity adjuster Substances 0.000 description 1
- 230000009191 jumping Effects 0.000 description 1
- 210000003127 knee Anatomy 0.000 description 1
- 201000003723 learning disability Diseases 0.000 description 1
- 239000007788 liquid Substances 0.000 description 1
- 239000006193 liquid solution Substances 0.000 description 1
- 210000004185 liver Anatomy 0.000 description 1
- 201000007270 liver cancer Diseases 0.000 description 1
- 208000014018 liver neoplasm Diseases 0.000 description 1
- 239000003589 local anesthetic agent Substances 0.000 description 1
- 210000005230 lumbar spinal cord Anatomy 0.000 description 1
- HWYHZTIRURJOHG-UHFFFAOYSA-N luminol Chemical compound O=C1NNC(=O)C2=C1C(N)=CC=C2 HWYHZTIRURJOHG-UHFFFAOYSA-N 0.000 description 1
- 210000004072 lung Anatomy 0.000 description 1
- 210000005265 lung cell Anatomy 0.000 description 1
- 230000004199 lung function Effects 0.000 description 1
- 239000008176 lyophilized powder Substances 0.000 description 1
- 125000003588 lysine group Chemical group [H]N([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])(N([H])[H])C(*)=O 0.000 description 1
- ZLNQQNXFFQJAID-UHFFFAOYSA-L magnesium carbonate Chemical compound [Mg+2].[O-]C([O-])=O ZLNQQNXFFQJAID-UHFFFAOYSA-L 0.000 description 1
- 239000001095 magnesium carbonate Substances 0.000 description 1
- 229910000021 magnesium carbonate Inorganic materials 0.000 description 1
- 230000005291 magnetic effect Effects 0.000 description 1
- 230000036210 malignancy Effects 0.000 description 1
- 210000003784 masticatory muscle Anatomy 0.000 description 1
- 239000012528 membrane Substances 0.000 description 1
- 229930182817 methionine Natural products 0.000 description 1
- VLPIATFUUWWMKC-UHFFFAOYSA-N mexiletine Chemical compound CC(N)COC1=C(C)C=CC=C1C VLPIATFUUWWMKC-UHFFFAOYSA-N 0.000 description 1
- 210000003632 microfilament Anatomy 0.000 description 1
- 230000005012 migration Effects 0.000 description 1
- 238000013508 migration Methods 0.000 description 1
- 230000003278 mimic effect Effects 0.000 description 1
- 230000004048 modification Effects 0.000 description 1
- 238000012986 modification Methods 0.000 description 1
- 230000037257 muscle growth Effects 0.000 description 1
- 230000012042 muscle hypertrophy Effects 0.000 description 1
- 230000003387 muscular Effects 0.000 description 1
- 201000000050 myeloid neoplasm Diseases 0.000 description 1
- 210000003365 myofibril Anatomy 0.000 description 1
- 201000009340 myotonic dystrophy type 1 Diseases 0.000 description 1
- 201000008709 myotonic dystrophy type 2 Diseases 0.000 description 1
- 210000004897 n-terminal region Anatomy 0.000 description 1
- 210000000492 nasalseptum Anatomy 0.000 description 1
- 239000013642 negative control Substances 0.000 description 1
- 230000003188 neurobehavioral effect Effects 0.000 description 1
- 238000006386 neutralization reaction Methods 0.000 description 1
- 210000001331 nose Anatomy 0.000 description 1
- 235000016709 nutrition Nutrition 0.000 description 1
- 230000035764 nutrition Effects 0.000 description 1
- 239000003921 oil Substances 0.000 description 1
- 235000019198 oils Nutrition 0.000 description 1
- 239000002674 ointment Substances 0.000 description 1
- 230000003287 optical effect Effects 0.000 description 1
- 230000003204 osmotic effect Effects 0.000 description 1
- 235000006408 oxalic acid Nutrition 0.000 description 1
- 239000001301 oxygen Substances 0.000 description 1
- 229910052760 oxygen Inorganic materials 0.000 description 1
- 238000004806 packaging method and process Methods 0.000 description 1
- 206010033675 panniculitis Diseases 0.000 description 1
- 239000012188 paraffin wax Substances 0.000 description 1
- 210000004197 pelvis Anatomy 0.000 description 1
- 230000035515 penetration Effects 0.000 description 1
- 238000011056 performance test Methods 0.000 description 1
- 150000002978 peroxides Chemical class 0.000 description 1
- 239000000825 pharmaceutical preparation Substances 0.000 description 1
- WVDDGKGOMKODPV-ZQBYOMGUSA-N phenyl(114C)methanol Chemical compound O[14CH2]C1=CC=CC=C1 WVDDGKGOMKODPV-ZQBYOMGUSA-N 0.000 description 1
- COLNVLDHVKWLRT-UHFFFAOYSA-N phenylalanine Natural products OC(=O)C(N)CC1=CC=CC=C1 COLNVLDHVKWLRT-UHFFFAOYSA-N 0.000 description 1
- 229960002036 phenytoin Drugs 0.000 description 1
- 235000011007 phosphoric acid Nutrition 0.000 description 1
- 238000000554 physical therapy Methods 0.000 description 1
- 230000004962 physiological condition Effects 0.000 description 1
- 239000006187 pill Substances 0.000 description 1
- 210000003635 pituitary gland Anatomy 0.000 description 1
- 239000013612 plasmid Substances 0.000 description 1
- 229920001223 polyethylene glycol Polymers 0.000 description 1
- 239000011591 potassium Substances 0.000 description 1
- 229910052700 potassium Inorganic materials 0.000 description 1
- 239000000843 powder Substances 0.000 description 1
- REQCZEXYDRLIBE-UHFFFAOYSA-N procainamide Chemical compound CCN(CC)CCNC(=O)C1=CC=C(N)C=C1 REQCZEXYDRLIBE-UHFFFAOYSA-N 0.000 description 1
- 229960000244 procainamide Drugs 0.000 description 1
- MFDFERRIHVXMIY-UHFFFAOYSA-N procaine Chemical compound CCN(CC)CCOC(=O)C1=CC=C(N)C=C1 MFDFERRIHVXMIY-UHFFFAOYSA-N 0.000 description 1
- 229960004919 procaine Drugs 0.000 description 1
- 230000008569 process Effects 0.000 description 1
- 208000026526 progressive weakness Diseases 0.000 description 1
- 230000000770 proinflammatory effect Effects 0.000 description 1
- 230000004952 protein activity Effects 0.000 description 1
- 230000006337 proteolytic cleavage Effects 0.000 description 1
- 201000003004 ptosis Diseases 0.000 description 1
- 230000009325 pulmonary function Effects 0.000 description 1
- 238000009613 pulmonary function test Methods 0.000 description 1
- 210000003314 quadriceps muscle Anatomy 0.000 description 1
- 238000012207 quantitative assay Methods 0.000 description 1
- 229960000948 quinine Drugs 0.000 description 1
- 239000002994 raw material Substances 0.000 description 1
- 238000010188 recombinant method Methods 0.000 description 1
- 238000011084 recovery Methods 0.000 description 1
- 210000000664 rectum Anatomy 0.000 description 1
- 201000004193 respiratory failure Diseases 0.000 description 1
- 201000009410 rhabdomyosarcoma Diseases 0.000 description 1
- CVHZOJJKTDOEJC-UHFFFAOYSA-N saccharin Chemical compound C1=CC=C2C(=O)NS(=O)(=O)C2=C1 CVHZOJJKTDOEJC-UHFFFAOYSA-N 0.000 description 1
- 239000004248 saffron Substances 0.000 description 1
- 208000026451 salivation Diseases 0.000 description 1
- 210000000518 sarcolemma Anatomy 0.000 description 1
- 210000002235 sarcomere Anatomy 0.000 description 1
- 230000028327 secretion Effects 0.000 description 1
- 210000002480 semicircular canal Anatomy 0.000 description 1
- 125000003607 serino group Chemical group [H]N([H])[C@]([H])(C(=O)[*])C(O[H])([H])[H] 0.000 description 1
- 210000000717 sertoli cell Anatomy 0.000 description 1
- 108010026333 seryl-proline Proteins 0.000 description 1
- RMAQACBXLXPBSY-UHFFFAOYSA-N silicic acid Chemical compound O[Si](O)(O)O RMAQACBXLXPBSY-UHFFFAOYSA-N 0.000 description 1
- 235000012239 silicon dioxide Nutrition 0.000 description 1
- 210000001057 smooth muscle myoblast Anatomy 0.000 description 1
- 239000011734 sodium Substances 0.000 description 1
- 229910052708 sodium Inorganic materials 0.000 description 1
- 239000001632 sodium acetate Substances 0.000 description 1
- 235000017281 sodium acetate Nutrition 0.000 description 1
- 239000001488 sodium phosphate Substances 0.000 description 1
- 229910000162 sodium phosphate Inorganic materials 0.000 description 1
- 239000007787 solid Substances 0.000 description 1
- 230000000392 somatic effect Effects 0.000 description 1
- 230000003238 somatosensory effect Effects 0.000 description 1
- 210000005070 sphincter Anatomy 0.000 description 1
- 230000002269 spontaneous effect Effects 0.000 description 1
- 239000003381 stabilizer Substances 0.000 description 1
- 239000010959 steel Substances 0.000 description 1
- 238000010254 subcutaneous injection Methods 0.000 description 1
- 239000007929 subcutaneous injection Substances 0.000 description 1
- 210000004304 subcutaneous tissue Anatomy 0.000 description 1
- 239000005720 sucrose Substances 0.000 description 1
- 230000004083 survival effect Effects 0.000 description 1
- 238000004114 suspension culture Methods 0.000 description 1
- 238000013268 sustained release Methods 0.000 description 1
- 239000012730 sustained-release form Substances 0.000 description 1
- 239000003765 sweetening agent Substances 0.000 description 1
- 208000011580 syndromic disease Diseases 0.000 description 1
- 238000003786 synthesis reaction Methods 0.000 description 1
- 230000009885 systemic effect Effects 0.000 description 1
- 239000003826 tablet Substances 0.000 description 1
- 239000000454 talc Substances 0.000 description 1
- 229910052623 talc Inorganic materials 0.000 description 1
- 235000012222 talc Nutrition 0.000 description 1
- 239000011975 tartaric acid Substances 0.000 description 1
- 235000002906 tartaric acid Nutrition 0.000 description 1
- 230000001988 toxicity Effects 0.000 description 1
- 231100000419 toxicity Toxicity 0.000 description 1
- 238000012546 transfer Methods 0.000 description 1
- 230000009261 transgenic effect Effects 0.000 description 1
- 230000001052 transient effect Effects 0.000 description 1
- 150000003626 triacylglycerols Chemical class 0.000 description 1
- RYFMWSXOAZQYPI-UHFFFAOYSA-K trisodium phosphate Chemical compound [Na+].[Na+].[Na+].[O-]P([O-])([O-])=O RYFMWSXOAZQYPI-UHFFFAOYSA-K 0.000 description 1
- 210000003454 tympanic membrane Anatomy 0.000 description 1
- OUYCCCASQSFEME-UHFFFAOYSA-N tyrosine Natural products OC(=O)C(N)CC1=CC=C(O)C=C1 OUYCCCASQSFEME-UHFFFAOYSA-N 0.000 description 1
- 241000701447 unidentified baculovirus Species 0.000 description 1
- 210000003932 urinary bladder Anatomy 0.000 description 1
- 206010046766 uterine cancer Diseases 0.000 description 1
- 208000012991 uterine carcinoma Diseases 0.000 description 1
- 239000004474 valine Substances 0.000 description 1
- 235000015112 vegetable and seed oil Nutrition 0.000 description 1
- 239000008158 vegetable oil Substances 0.000 description 1
- 230000007497 verbal memory Effects 0.000 description 1
- 230000002747 voluntary effect Effects 0.000 description 1
- 239000001993 wax Substances 0.000 description 1
- 230000003313 weakening effect Effects 0.000 description 1
- 238000009736 wetting Methods 0.000 description 1
- 108010027345 wheylin-1 peptide Proteins 0.000 description 1
- 210000005253 yeast cell Anatomy 0.000 description 1
Images
Classifications
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07K—PEPTIDES
- C07K14/00—Peptides having more than 20 amino acids; Gastrins; Somatostatins; Melanotropins; Derivatives thereof
- C07K14/435—Peptides having more than 20 amino acids; Gastrins; Somatostatins; Melanotropins; Derivatives thereof from animals; from humans
- C07K14/46—Peptides having more than 20 amino acids; Gastrins; Somatostatins; Melanotropins; Derivatives thereof from animals; from humans from vertebrates
- C07K14/47—Peptides having more than 20 amino acids; Gastrins; Somatostatins; Melanotropins; Derivatives thereof from animals; from humans from vertebrates from mammals
- C07K14/4701—Peptides having more than 20 amino acids; Gastrins; Somatostatins; Melanotropins; Derivatives thereof from animals; from humans from vertebrates from mammals not used
- C07K14/473—Peptides having more than 20 amino acids; Gastrins; Somatostatins; Melanotropins; Derivatives thereof from animals; from humans from vertebrates from mammals not used alpha-Glycoproteins
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07K—PEPTIDES
- C07K14/00—Peptides having more than 20 amino acids; Gastrins; Somatostatins; Melanotropins; Derivatives thereof
- C07K14/435—Peptides having more than 20 amino acids; Gastrins; Somatostatins; Melanotropins; Derivatives thereof from animals; from humans
- C07K14/46—Peptides having more than 20 amino acids; Gastrins; Somatostatins; Melanotropins; Derivatives thereof from animals; from humans from vertebrates
- C07K14/47—Peptides having more than 20 amino acids; Gastrins; Somatostatins; Melanotropins; Derivatives thereof from animals; from humans from vertebrates from mammals
- C07K14/4701—Peptides having more than 20 amino acids; Gastrins; Somatostatins; Melanotropins; Derivatives thereof from animals; from humans from vertebrates from mammals not used
- C07K14/4707—Muscular dystrophy
- C07K14/4708—Duchenne dystrophy
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K39/00—Medicinal preparations containing antigens or antibodies
- A61K39/395—Antibodies; Immunoglobulins; Immune serum, e.g. antilymphocytic serum
- A61K39/39533—Antibodies; Immunoglobulins; Immune serum, e.g. antilymphocytic serum against materials from animals
- A61K39/3955—Antibodies; Immunoglobulins; Immune serum, e.g. antilymphocytic serum against materials from animals against proteinaceous materials, e.g. enzymes, hormones, lymphokines
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K45/00—Medicinal preparations containing active ingredients not provided for in groups A61K31/00 - A61K41/00
- A61K45/06—Mixtures of active ingredients without chemical characterisation, e.g. antiphlogistics and cardiaca
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K9/00—Medicinal preparations characterised by special physical form
- A61K9/0012—Galenical forms characterised by the site of application
- A61K9/0019—Injectable compositions; Intramuscular, intravenous, arterial, subcutaneous administration; Compositions to be administered through the skin in an invasive manner
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P21/00—Drugs for disorders of the muscular or neuromuscular system
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P21/00—Drugs for disorders of the muscular or neuromuscular system
- A61P21/04—Drugs for disorders of the muscular or neuromuscular system for myasthenia gravis
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P21/00—Drugs for disorders of the muscular or neuromuscular system
- A61P21/06—Anabolic agents
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07K—PEPTIDES
- C07K14/00—Peptides having more than 20 amino acids; Gastrins; Somatostatins; Melanotropins; Derivatives thereof
- C07K14/435—Peptides having more than 20 amino acids; Gastrins; Somatostatins; Melanotropins; Derivatives thereof from animals; from humans
- C07K14/46—Peptides having more than 20 amino acids; Gastrins; Somatostatins; Melanotropins; Derivatives thereof from animals; from humans from vertebrates
- C07K14/47—Peptides having more than 20 amino acids; Gastrins; Somatostatins; Melanotropins; Derivatives thereof from animals; from humans from vertebrates from mammals
- C07K14/4701—Peptides having more than 20 amino acids; Gastrins; Somatostatins; Melanotropins; Derivatives thereof from animals; from humans from vertebrates from mammals not used
- C07K14/4702—Regulators; Modulating activity
- C07K14/4703—Inhibitors; Suppressors
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07K—PEPTIDES
- C07K16/00—Immunoglobulins [IGs], e.g. monoclonal or polyclonal antibodies
- C07K16/18—Immunoglobulins [IGs], e.g. monoclonal or polyclonal antibodies against material from animals or humans
- C07K16/22—Immunoglobulins [IGs], e.g. monoclonal or polyclonal antibodies against material from animals or humans against growth factors ; against growth regulators
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K39/00—Medicinal preparations containing antigens or antibodies
- A61K2039/505—Medicinal preparations containing antigens or antibodies comprising antibodies
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K39/00—Medicinal preparations containing antigens or antibodies
- A61K2039/545—Medicinal preparations containing antigens or antibodies characterised by the dose, timing or administration schedule
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K38/00—Medicinal preparations containing peptides
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K39/00—Medicinal preparations containing antigens or antibodies
- A61K39/395—Antibodies; Immunoglobulins; Immune serum, e.g. antilymphocytic serum
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07K—PEPTIDES
- C07K2317/00—Immunoglobulins specific features
- C07K2317/90—Immunoglobulins specific features characterized by (pharmaco)kinetic aspects or by stability of the immunoglobulin
- C07K2317/92—Affinity (KD), association rate (Ka), dissociation rate (Kd) or EC50 value
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07K—PEPTIDES
- C07K2319/00—Fusion polypeptide
- C07K2319/30—Non-immunoglobulin-derived peptide or protein having an immunoglobulin constant or Fc region, or a fragment thereof, attached thereto
Landscapes
- Health & Medical Sciences (AREA)
- Chemical & Material Sciences (AREA)
- Life Sciences & Earth Sciences (AREA)
- Medicinal Chemistry (AREA)
- Organic Chemistry (AREA)
- General Health & Medical Sciences (AREA)
- Veterinary Medicine (AREA)
- Public Health (AREA)
- Pharmacology & Pharmacy (AREA)
- Animal Behavior & Ethology (AREA)
- Proteomics, Peptides & Aminoacids (AREA)
- Biophysics (AREA)
- Biochemistry (AREA)
- Genetics & Genomics (AREA)
- Molecular Biology (AREA)
- Toxicology (AREA)
- Zoology (AREA)
- Gastroenterology & Hepatology (AREA)
- Bioinformatics & Cheminformatics (AREA)
- Epidemiology (AREA)
- Engineering & Computer Science (AREA)
- Physical Education & Sports Medicine (AREA)
- Orthopedic Medicine & Surgery (AREA)
- Neurology (AREA)
- Nuclear Medicine, Radiotherapy & Molecular Imaging (AREA)
- Chemical Kinetics & Catalysis (AREA)
- General Chemical & Material Sciences (AREA)
- Immunology (AREA)
- Endocrinology (AREA)
- Mycology (AREA)
- Microbiology (AREA)
- Dermatology (AREA)
- Medicines That Contain Protein Lipid Enzymes And Other Medicines (AREA)
- Peptides Or Proteins (AREA)
- Micro-Organisms Or Cultivation Processes Thereof (AREA)
- Prostheses (AREA)
- Medicines Containing Antibodies Or Antigens For Use As Internal Diagnostic Agents (AREA)
- Pharmaceuticals Containing Other Organic And Inorganic Compounds (AREA)
Applications Claiming Priority (3)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| US201662303954P | 2016-03-04 | 2016-03-04 | |
| US62/303,954 | 2016-03-04 | ||
| PCT/US2017/020723 WO2017152090A2 (en) | 2016-03-04 | 2017-03-03 | Recombinant follistatin-fc fusion proteins and use in treating duchenne muscular dystrophy |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| KR20180137487A true KR20180137487A (ko) | 2018-12-27 |
Family
ID=58358939
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| KR1020187028662A Withdrawn KR20180137487A (ko) | 2016-03-04 | 2017-03-03 | 재조합 폴리스타틴-fc 융합 단백질 및 듀시엔형 근이영양증 치료에서의 용도 |
Country Status (14)
| Country | Link |
|---|---|
| US (1) | US20200247856A1 (enExample) |
| EP (1) | EP3423479A2 (enExample) |
| JP (1) | JP2019509735A (enExample) |
| KR (1) | KR20180137487A (enExample) |
| CN (1) | CN109153708A (enExample) |
| AU (1) | AU2017228475A1 (enExample) |
| BR (1) | BR112018067747A2 (enExample) |
| CA (1) | CA3016576A1 (enExample) |
| EA (1) | EA201891990A1 (enExample) |
| IL (1) | IL261397A (enExample) |
| MA (1) | MA43588A (enExample) |
| MX (1) | MX2018010470A (enExample) |
| SG (1) | SG11201807400TA (enExample) |
| WO (1) | WO2017152090A2 (enExample) |
Families Citing this family (7)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US20180362604A1 (en) * | 2017-05-12 | 2018-12-20 | Shire Human Genetic Therapies, Inc. | Recombinant Follistatin-FC Fusion Proteins and Use in Treating Duchenne Muscular Dystrophy |
| TW201936201A (zh) | 2017-12-14 | 2019-09-16 | 美商堅固生物科技公司 | 基因之非病毒生產及遞送 |
| CN108303860B (zh) * | 2018-03-26 | 2019-11-08 | 西安电子科技大学 | 一种用于无掩模光刻扫描的分布式曝光方法 |
| WO2019191204A1 (en) * | 2018-03-28 | 2019-10-03 | Acceleron Pharma Inc. | Follistatin polypeptides for the treatment of muscle contracture |
| AU2020216361A1 (en) | 2019-01-29 | 2021-08-12 | Takeda Pharmaceutical Company Limited | Parathyroid hormone variants |
| EP4540273A1 (en) | 2022-06-15 | 2025-04-23 | UCB Biopharma SRL | Fusion protein for the prevention, treatment or amelioration of kidney diseases |
| JP2025520393A (ja) | 2022-06-15 | 2025-07-03 | ユーシービー バイオファルマ エスアールエル | フォリスタチン-fc融合タンパク質 |
Family Cites Families (12)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US6277375B1 (en) | 1997-03-03 | 2001-08-21 | Board Of Regents, The University Of Texas System | Immunoglobulin-like domains with increased half-lives |
| PT1355919E (pt) | 2000-12-12 | 2011-03-02 | Medimmune Llc | Moléculas com semivida longa, composições que as contêm e suas utilizações |
| MXPA04008150A (es) * | 2002-02-21 | 2005-06-17 | Wyeth Corp | Gasp1: una proteina que contiene dominio de folistatina. |
| EP1896503B1 (en) | 2005-05-31 | 2014-10-29 | Board of Regents, The University of Texas System | IgG1 ANTIBODIES WITH MUTATED Fc PORTION FOR INCREASED BINDING TO FcRn RECEPTOR AND USES TEHEREOF |
| WO2008030367A2 (en) * | 2006-09-01 | 2008-03-13 | The General Hospital Corporation | Selective myostatin inhibitors |
| US8580922B2 (en) | 2011-03-04 | 2013-11-12 | Shire Human Genetic Therapies, Inc. | Peptide linkers for polypeptide compositions and methods for using same |
| KR20160044598A (ko) * | 2011-03-29 | 2016-04-25 | 로슈 글리카트 아게 | 항체 Fc 변이체 |
| WO2013192131A1 (en) * | 2012-06-21 | 2013-12-27 | Indiana University Research And Technology Corporation | Incretin receptor ligand polypeptide fc-region fusion polypeptides and conjugates with altered fc-effector function |
| EP3626735A1 (en) * | 2012-12-18 | 2020-03-25 | Novartis AG | Stabilized insulin-like growth factor polypeptides |
| JP6475639B2 (ja) * | 2013-01-25 | 2019-02-27 | シャイアー ヒューマン ジェネティック セラピーズ インコーポレイテッド | デュシェンヌ筋ジストロフィーの治療におけるフォリスタチン |
| WO2014187807A1 (en) * | 2013-05-21 | 2014-11-27 | Arcarios B.V. | Follistatin derivatives |
| WO2015187977A1 (en) * | 2014-06-04 | 2015-12-10 | Acceleron Pharma, Inc. | Methods and compositions for treatment of disorders with follistatin polypeptides |
-
2017
- 2017-03-03 KR KR1020187028662A patent/KR20180137487A/ko not_active Withdrawn
- 2017-03-03 EP EP17711939.3A patent/EP3423479A2/en not_active Ceased
- 2017-03-03 CN CN201780025363.2A patent/CN109153708A/zh active Pending
- 2017-03-03 EA EA201891990A patent/EA201891990A1/ru unknown
- 2017-03-03 CA CA3016576A patent/CA3016576A1/en not_active Abandoned
- 2017-03-03 MX MX2018010470A patent/MX2018010470A/es unknown
- 2017-03-03 SG SG11201807400TA patent/SG11201807400TA/en unknown
- 2017-03-03 US US16/082,070 patent/US20200247856A1/en not_active Abandoned
- 2017-03-03 WO PCT/US2017/020723 patent/WO2017152090A2/en not_active Ceased
- 2017-03-03 MA MA043588A patent/MA43588A/fr unknown
- 2017-03-03 AU AU2017228475A patent/AU2017228475A1/en not_active Abandoned
- 2017-03-03 JP JP2018546471A patent/JP2019509735A/ja active Pending
- 2017-03-03 BR BR112018067747A patent/BR112018067747A2/pt not_active Application Discontinuation
-
2018
- 2018-08-27 IL IL261397A patent/IL261397A/en unknown
Also Published As
| Publication number | Publication date |
|---|---|
| US20200247856A1 (en) | 2020-08-06 |
| CN109153708A (zh) | 2019-01-04 |
| BR112018067747A2 (pt) | 2019-01-08 |
| IL261397A (en) | 2018-10-31 |
| MA43588A (fr) | 2021-06-02 |
| WO2017152090A3 (en) | 2017-10-19 |
| EP3423479A2 (en) | 2019-01-09 |
| AU2017228475A1 (en) | 2018-09-13 |
| SG11201807400TA (en) | 2018-09-27 |
| WO2017152090A2 (en) | 2017-09-08 |
| EA201891990A1 (ru) | 2019-04-30 |
| CA3016576A1 (en) | 2017-09-08 |
| MX2018010470A (es) | 2018-12-06 |
| JP2019509735A (ja) | 2019-04-11 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| JP6475639B2 (ja) | デュシェンヌ筋ジストロフィーの治療におけるフォリスタチン | |
| KR20180137487A (ko) | 재조합 폴리스타틴-fc 융합 단백질 및 듀시엔형 근이영양증 치료에서의 용도 | |
| CN110914294A (zh) | 重组卵泡抑素-fc融合蛋白及其在治疗杜氏肌营养不良中的用途 | |
| JP6469022B2 (ja) | デュシェンヌ型筋ジストロフィーの治療における抗−flt−1抗体 | |
| US10729746B2 (en) | Recombinant placenta growth factor for treating Duchenne muscular dystrophy | |
| HK1213804B (zh) | 治疗杜兴氏肌营养不良的卵泡抑素 | |
| HK1217637B (en) | Follistatin in treating duchenne muscular dystrophy |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| PA0105 | International application |
Patent event date: 20181004 Patent event code: PA01051R01D Comment text: International Patent Application |
|
| PG1501 | Laying open of application | ||
| PC1203 | Withdrawal of no request for examination |