KR20170000055A - Method for producing aromatics from light hydrocarbons using oxygen using carbon dioxide - Google Patents
Method for producing aromatics from light hydrocarbons using oxygen using carbon dioxide Download PDFInfo
- Publication number
- KR20170000055A KR20170000055A KR1020150088668A KR20150088668A KR20170000055A KR 20170000055 A KR20170000055 A KR 20170000055A KR 1020150088668 A KR1020150088668 A KR 1020150088668A KR 20150088668 A KR20150088668 A KR 20150088668A KR 20170000055 A KR20170000055 A KR 20170000055A
- Authority
- KR
- South Korea
- Prior art keywords
- low
- carbon
- carbon dioxide
- compound
- catalyst
- Prior art date
Links
Images
Classifications
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07C—ACYCLIC OR CARBOCYCLIC COMPOUNDS
- C07C1/00—Preparation of hydrocarbons from one or more compounds, none of them being a hydrocarbon
- C07C1/02—Preparation of hydrocarbons from one or more compounds, none of them being a hydrocarbon from oxides of a carbon
- C07C1/12—Preparation of hydrocarbons from one or more compounds, none of them being a hydrocarbon from oxides of a carbon from carbon dioxide with hydrogen
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07C—ACYCLIC OR CARBOCYCLIC COMPOUNDS
- C07C15/00—Cyclic hydrocarbons containing only six-membered aromatic rings as cyclic parts
- C07C15/02—Monocyclic hydrocarbons
- C07C15/04—Benzene
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07C—ACYCLIC OR CARBOCYCLIC COMPOUNDS
- C07C15/00—Cyclic hydrocarbons containing only six-membered aromatic rings as cyclic parts
- C07C15/02—Monocyclic hydrocarbons
- C07C15/06—Toluene
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07C—ACYCLIC OR CARBOCYCLIC COMPOUNDS
- C07C15/00—Cyclic hydrocarbons containing only six-membered aromatic rings as cyclic parts
- C07C15/02—Monocyclic hydrocarbons
- C07C15/067—C8H10 hydrocarbons
- C07C15/08—Xylenes
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07C—ACYCLIC OR CARBOCYCLIC COMPOUNDS
- C07C15/00—Cyclic hydrocarbons containing only six-membered aromatic rings as cyclic parts
- C07C15/40—Cyclic hydrocarbons containing only six-membered aromatic rings as cyclic parts substituted by unsaturated carbon radicals
- C07C15/42—Cyclic hydrocarbons containing only six-membered aromatic rings as cyclic parts substituted by unsaturated carbon radicals monocyclic
- C07C15/44—Cyclic hydrocarbons containing only six-membered aromatic rings as cyclic parts substituted by unsaturated carbon radicals monocyclic the hydrocarbon substituent containing a carbon-to-carbon double bond
- C07C15/46—Styrene; Ring-alkylated styrenes
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07C—ACYCLIC OR CARBOCYCLIC COMPOUNDS
- C07C2/00—Preparation of hydrocarbons from hydrocarbons containing a smaller number of carbon atoms
- C07C2/02—Preparation of hydrocarbons from hydrocarbons containing a smaller number of carbon atoms by addition between unsaturated hydrocarbons
- C07C2/04—Preparation of hydrocarbons from hydrocarbons containing a smaller number of carbon atoms by addition between unsaturated hydrocarbons by oligomerisation of well-defined unsaturated hydrocarbons without ring formation
- C07C2/06—Preparation of hydrocarbons from hydrocarbons containing a smaller number of carbon atoms by addition between unsaturated hydrocarbons by oligomerisation of well-defined unsaturated hydrocarbons without ring formation of alkenes, i.e. acyclic hydrocarbons having only one carbon-to-carbon double bond
- C07C2/08—Catalytic processes
-
- Y—GENERAL TAGGING OF NEW TECHNOLOGICAL DEVELOPMENTS; GENERAL TAGGING OF CROSS-SECTIONAL TECHNOLOGIES SPANNING OVER SEVERAL SECTIONS OF THE IPC; TECHNICAL SUBJECTS COVERED BY FORMER USPC CROSS-REFERENCE ART COLLECTIONS [XRACs] AND DIGESTS
- Y02—TECHNOLOGIES OR APPLICATIONS FOR MITIGATION OR ADAPTATION AGAINST CLIMATE CHANGE
- Y02P—CLIMATE CHANGE MITIGATION TECHNOLOGIES IN THE PRODUCTION OR PROCESSING OF GOODS
- Y02P20/00—Technologies relating to chemical industry
- Y02P20/50—Improvements relating to the production of bulk chemicals
- Y02P20/52—Improvements relating to the production of bulk chemicals using catalysts, e.g. selective catalysts
Abstract
Disclosed is a method for producing an aromatic compound from a low-carbon compound using carbon dioxide. The process for producing an aromatic compound can be characterized in that an aromatic compound is produced through a reaction between a low carbon compound of one of the low carbon compounds of C 3 to C 6 or a mixture of one or more low carbon compounds and carbon dioxide and a catalyst. By using carbon dioxide, the conversion of the low-carbon compound is increased and the selectivity of the aromatic compound is increased to improve the production yield of the aromatic compound. The addition of carbon dioxide to the hydrocarbon reactant of the low hydrocarbon reduces the production of coke, The stability can be greatly improved. The ratio of carbon dioxide to low carbon compound can be applied in the range of 0.1 to 100. Various types of low carbon compounds ranging from C 1 to C 8 (particularly, C 3 to C 6) may be applied, and a mixture of shale gas and petroleum by-products may be applied.
Description
The present invention relates to a technique for converting carbon dioxide and low-carbon compounds into aromatic compounds.
Aromatic compounds are the main intermediates for various products such as household necessities and industrial products, and are high-value-added compounds with high availability. Particularly, the benzene, xylene and C 9 aromatic compounds have various commercial applications such as polystyrene, phenol resin, polycarbonate, nylon, polyurethane, gasoline, polyester fiber, phthalic anhydride and the like.
The pyrolysis process, naphtha cracking process, and cycler process have been developed variously as a method of producing an aromatic compound, but the limit of the conventional process and the low utilization New efforts are being sought for maximizing aromatics and research is being concentrated.
In the pyrolysis process of coal tar, the raw coal is transferred to the coke oven in the coke oven during the metallurgical coke manufacturing process to obtain the solid coke, and the coke is recovered in the regeneration process to obtain the aromatic compound through the energy intensive process at the high temperature of 1200K to be. Coke is a solid material which differs depending on the process, but mainly consists of two or more benzene rings, and contains impurities such as sulfur and nitrogen compounds. Korean Patent Laid-Open No. 10-2014-0082097 discloses that application of an aromatic compound using a pyrolysis process of coke can be used as a single process but shows a disadvantage in selectivity due to an increase in byproducts, There is a problem that the unit price of the aromatic compound is increased due to consumption.
A method for obtaining a conventional aromatic compound is a method using naphtha cracking. However, the cost for synthesizing an aromatic compound is increasing due to an increase in raw material supply cost due to a decrease in productivity of naphtha.
Recently, the development of technologies for the conversion of low carbon compounds into aromatic compounds is required, and efforts are being made to overcome the limitations of the naphtha reforming process. The production of low-carbon compounds (light hydrocarbons) has increased the supply of shale gas in the US and the by-products in the petrochemical process, and has become interested in the possibility of utilization as a raw material. As a result, there is a growing need to convert inexpensive low-carbon compounds to high value-added products.
Oxyaromatization using existing oxygen has a disadvantage in that by-products of CO and CO2 are generated in a low-carbon compound due to strong oxidizing power by oxygen, and thus the selectivity of an aromatic compound is lowered. The aromatic compound Has a disadvantage in that the selectivity is lowered due to the nitrogen compound.
Also, a method of obtaining an aromatic compound by the Sikra process developed by UOP (Universal Oil Products Co.) in the United States has been put to practical use. The ZSM-5 (zeolite) added with gallium (Ga) is used as a catalyst to obtain an aromatic compound, but its application is limited due to the generation of coke and the limitation of the process life. The Sikra process is a technique for obtaining liquid aromatic compounds mainly using LPG gas as a main raw material. The aim of this study was to obtain high value-added aromatic compounds from low-priced LPG gas by focusing on the fact that aromatic compounds are high value-added products. The LPG gas mainly consists of propane (C 3 ) butane (C 4 ). In the course of the Sikra process, dehydrocyclodimerization, activation of the C 3 or C 4 CH bond, and dehydrogenation of the olefin intermediates are the rate determining steps of the reaction. . Thereafter, an aromatic compound is produced by dehydrogenation, oligomerization and cyclization.
The Sikra process is largely divided into three parts: a reactor, a Continuous Catalyst Regeneration (CCR), and a product recovery. The most important condition for obtaining the product at a high efficiency in the Sikra process is to regenerate the catalyst at a high temperature by hydrogen in the CCR regenerator under the control of the coke to regenerate the catalyst, but the hydrogen must be continuously flowed to the CCR regenerator, There is a disadvantage in that the cost for regenerating the catalyst is increased due to the supply of high-temperature energy, thereby increasing the production cost of the product. The Cyclar process has become a homework to obtain aromatic compounds, with control over coke not reducing activity on catalysts circulating in reactors and regenerators. The method of eliminating the coke generated in the catalyst is solved by raising the temperature of the CCR regenerator in the present Cyclar process, and the consumption of energy for catalyst regeneration can not be avoided. In addition, there is a risk of explosion of the regenerator when it reacts rapidly due to the explosive heat generated by using hydrogen as an oxidizing agent (see Non-Patent Document 1: Refining and Petrochemicals, 591-614).
According to U.S. Pat. No. 4,590,323, n-Octane was mixed with H-ZSM-5, which had not been treated as an aromaticization catalyst, at a ratio of WHSV = 1.0 and BTX (benzene (B)) of benzene, toluene, (T), xylenes (X)) of 18.5 wt% were obtained, whereas when Sc / H-ZSM-5 carrying 11.5 wt% Scandium was used, the ratio of BTX was 26.5 wt% Of the respondents increased by 47%. In this patent, the use of metal oxides of Groups IIIA, IVA and VA groups can increase the ratio to aromatic compounds. However, the above-mentioned patent does not show the effect of carbon dioxide and shows only the effect obtained by supporting metal oxide on simple ZSM-5.
US Published Patent Application 2005/0143610 discloses a technique for converting an alkane to an aromatic compound using platinum supported Pt / ZSM-5. In the case of ZSM-5 with 0.33% Pt supported on ZSM-5 mass, conversion of 46.9% and selectivity to BTX was 44.0% when 50% propane / nitrogen reactant was reacted at 470 ℃. However, in the above patent, no carbon dioxide is used, and since the rare metal Pt is used, there is a problem that the unit price of the catalyst is increased and the unit cost of generating BTX is increased.
US 5,149,679 discloses that a catalyst carrying 2 wt% or less of gallium on a zeolite having medium size micropores of ZSM-5, ZSM-11 or ZSM-12 is used for the conversion of low carbon compounds to aromatics ≪ / RTI > Rhenium, Rhodium, Nickel, Palladium, Platinum, and Iridium may be used in the above patent, but it is not necessarily a metal necessary for aromaticization of a low-carbon compound Respectively. In the above patent, 50% by weight of BTX was obtained when 100% propane gas was flowed at 500 ° C using Pt / Ga / ZSM-5 carrying platinum and gallium.
In the non-patent reference 2 (ACS Catalysis, 2 (2012) 1203-1210), the yield of aromatic compounds was measured using Ga, Zn-ZSM-5 in the reaction for producing an aromatic compound using C4 alcohol as a reactant . However, the method of obtaining aromatic compounds by modifying only the kind of ZSM-5 carrying metal or metal oxide, showing only the activity in an inert gas, and the introduction of an oxidative method using carbon dioxide has not yet been attempted It is not.
U.S. Patent No. 6,958,427 discloses that carbon dioxide is used to increase the selectivity of styrene monomer through oxidative dehydrogenation from ethylbenzene. In other words, carbon dioxide is converted into carbon dioxide by the concept of soft oxidant, thereby reducing by-products with low oxidizing power, thereby increasing the yield of styrene.
In US Pat. No. 6,037,511, carbon dioxide can be used as a diluent. In the conversion of ethylbenzene to styrene monomers, an oxidative dehydrogenation reaction using carbon dioxide yielded a yield of styrene monomer of 48%. According to the patent, there is evidence that carbon dioxide can be used as an oxidizing agent, and hydrogen is generated by an oxidative dehydrogenation reaction. In addition, there is a growing interest in carbon dioxide by re-illuminating the fact that carbon dioxide can act as a soft oxidant and promoter (Non-Patent Document 2: Energy Env. Sci., 5 (2012) 9419 -9437). According to the non-patent document 2, carbon dioxide is converted from the surface of the catalyst to carbon monoxide, thereby forming active oxygen species on the surface of the catalyst, and the conversion rate of the dehydrogenation reaction is increased.
An industrial application using carbon dioxide is the synthesis of styrene from ethylbenzene through dehydrogenation. Catalyst deactivation by coke is promoted in propane and linear aliphatic hydrocarbons ranging from C 8 to C 12. The use of hydrogen or oxygen as a diluent initially contributed to an improvement in catalyst life. However, The risk has led to difficulties in the industrial application of the process. Korean Patent No. 10-0501842 relates to a process for applying carbon dioxide to the styrene synthesis process in ethylbenzene using carbon dioxide as an oxidizing agent. When carbon dioxide was used, the conversion of ethylbenzene was increased by 29.2% and the selectivity of styrene was increased by 6.6% at 550 ° C as compared with the step of synthesizing ethylbenzene with styrene by using water vapor as a diluent.
However, as described above, introduction of a process for obtaining an aromatic product using carbon dioxide as an oxidizing agent has not yet been attempted.
Therefore, the use of carbon dioxide in the present invention can be used as an oxidizing agent for conversion from a low-carbon compound to an aromatic compound, thereby improving the conversion of a low-carbon compound and the selectivity of an aromatic compound. Catalysts and processes.
In other words, by using carbon dioxide, the conversion of the low-carbon compound is increased and the selectivity of the aromatic compound is increased to improve the production yield of the aromatic compound. The addition of carbon dioxide to the hydrocarbon reactant of the low hydrocarbon reduces the production of coke, And a method for producing an aromatic compound capable of greatly improving the reaction stability.
Characterized in that an aromatic compound is prepared by reacting a low-carbon compound of one of C 3 to C 6 low-carbon compounds or a mixture of at least one low-carbon compound with carbon dioxide and a catalyst.
By using carbon dioxide, the conversion of low-carbon compounds and the selectivity of aromatic compounds are increased to improve the production yield of aromatic compounds and the addition of carbon dioxide to hydrocarbon reactants of low hydrocarbons reduces the production of coke, Can be greatly improved.
As a result of the oxidation reaction of petrochemicals, carbon dioxide as a by-product is generated, and the generated carbon dioxide can be used again as an oxidizing agent to lower the production cost of the aromatic product.
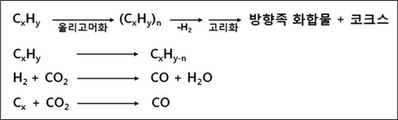
BRIEF DESCRIPTION OF THE DRAWINGS FIG. 1 is a diagram showing a process of obtaining an aromatic compound by contacting carbon dioxide and a low-carbon compound with a catalyst in a reactor in an embodiment of the present invention. FIG.
FIG. 2 is a graph comparing the yields when carbon dioxide is used as the flow gas and when the inert gas is used as the flow gas.
Hereinafter, embodiments of the present invention will be described in detail.
The present invention relates to a process for converting a low-carbon compound into an aromatic compound using carbon dioxide. It is an object of the present invention to overcome the incompleteness of a process due to a decrease in catalytic activity due to the formation of coke during the production of an aromatic compound from a low- By using carbon dioxide as an oxidizing agent for partial elimination of generated hydrogen, suppression of formation on the catalyst surface by reverse boudard reaction of coke, and oxidative dehydrogenation of dehydrogenation step of saturated hydrocarbon, the yield of aromatic compound (Yield) can be improved.
An aromatic compound preparation method according to an embodiment of the present invention may be characterized in that an aromatic compound is produced through a reaction between a low carbon compound of one of C 3 to C 6 low carbon compounds or a mixture of at least one low carbon compound and carbon dioxide and a catalyst have.
For example, an aromatic compound preparation method comprises introducing a low carbon compound of one of the C 3 to C 6 low carbon compounds or a mixture of one or more low carbon compounds into the flow gas, introducing carbon dioxide and mixing with the flow gas, Reacting the flow gas with the carbon dioxide under a catalyst, and recovering the aromatic compound produced as a result of the reaction.
At this time, the low-carbon compound (or the mixture) per catalyst unit may be introduced at a weight hourly space velocity (WHSV) of 0.1 to 10 h -1 and the molar ratio of the low-carbon compound to the carbon dioxide (or mixture) may range from 1 to 100.
For example, low-carbon compound is C 3 H 6, C 3 H 8, C 4 H 8, C 4
The reaction between the carbon dioxide and the low carbon compound and the catalyst can be carried out at a temperature of 300 to 800 ° C and a pressure of 0.5 to 200 atm.
The catalyst used may include at least one zeolite catalyst selected from the group consisting of MFI type zeolite catalyst, BEA type zeolite catalyst and MOR type zeolite catalyst. The zeolite catalyst may include at least one of Ga, Zn, Ni, Mg, Sn, Mo, Pt, Metal ions of La, metal or metal oxides. At this time, the metal that can be contained in the zeolite catalyst may be contained in the two dimer metal systems, or in the form of two or more metals in the zeolite catalyst in an amount of 0.1 to 25 wt%.
The aromatic compound produced as a result of the reaction may be characterized in that the yield of the C 9 aromatic compound containing at least one of benzene, toluene, xylene, ethylbenzene, styrene and durene is 40% or more.
BRIEF DESCRIPTION OF THE DRAWINGS FIG. 1 is a diagram showing a process of obtaining an aromatic compound by contacting carbon dioxide and a low-carbon compound with a catalyst in a reactor in an embodiment of the present invention. FIG. It has already been described in the background art that the reaction of oligomerization and cyclization for the production of aromatics can be used.
[Example 1]
The catalyst containing ZSM-5 (zeolite) produced was vaporized with butanol as a low-carbon compound to maintain the weight hourly space velocity WHSV = 1.74 h- 1 . The reaction was pre-treated at 500 ° C for 30 minutes in an inert gas atmosphere before the reaction, and the reaction temperature was 450 ° C. For the fresh catalyst, carbon dioxide and inert gas were flowed as a flow gas during the reaction and each case was compared. The gas after the reaction was not liquefied, and the product was analyzed using chromatography.
[Example 2]
Zn ion precursor is supported on the prepared ZSM-5 (zeolite) and calcined at 550 ° C to prepare a ZSM-5 catalyst carrying gallium oxide. Hereinafter, the reaction and the reaction analysis were carried out under the same conditions as in Example 1.
[Example 3]
Ga ion precursor is supported on the prepared ZSM-5, and then calcined at 550 DEG C to prepare a ZSM-5 carrying a zinc oxide. Hereinafter, the reaction and the reaction analysis were carried out under the same conditions as in Example 1.
FIG. 2 is a graph comparing the yields when carbon dioxide is used as the flow gas and when the inert gas is used as the flow gas.
The formula for calculating the yield was calculated on a% C basis. The% C criterion is to focus on the leveling of the carbon number of aromatic compounds converted by carbon dioxide. That is, the following formula (1) is a formula designed considering the fact that the number of carbon atoms of an aromatic compound is larger than that of a low-carbon compound (light hydrocarbon). This is applied to the above-described non-patent document 2 and various industrial yield calculations.
Table 1 is a table showing the yields of the aromatic compounds and the distribution of the aromatic compounds according to Examples 1, 2 and 3.
Here, 'C 1' is methane (Methane), 'C 2 = ' is ethylene (Ethylene), 'C 2 0 ' is ethane (Ethane), 'C 3 = ' propylene (propylene), 'C 3 0 ' Propane, and 'C 4 to C 9 ' may refer to all compounds that can be derived from C 4 to C 9 , respectively. In Table 1, each value represents% C-based yield, and 'Carrier gas' represents the reaction atmosphere.
Referring to FIG. 2, the yield of the aromatic compound in ZSM-5 using an inert gas as a flow gas in Example 1 was 44% (the yield of aromatic compound in the He reaction atmosphere in Example 1 of Table 1 The yield of aromatic compounds in ZSM-5 using carbon dioxide as a flow gas is 47% (the sum of the yields of aromatic compounds in a carbon dioxide (CO 2 ) reaction atmosphere in Example 2 of Table 1). The increase in the yield of aromatic compounds of carbon dioxide relative to the inert gas is 3%.
On the other hand, in Example 2 (an example using ZSM-5 impregnated with zinc oxide (ZnO) as a catalyst), the yield of an aromatic compound using an inert gas as a flow gas is 46%. However, the yield of aromatic compounds using carbon dioxide as a flow gas is 53%, and the yield difference is 7% as compared with the case where an inert gas is used as a flow gas. In other words, when ZSM-5 impregnated with ZnO is used as a catalyst rather than the yield difference in the embodiment using ZSM-5 as a catalyst, it can be seen that the yield difference according to the flow gas is larger.
Particularly, in Example 3 using ZSM-5 impregnated with gallium oxide (GaO) as a catalyst, the yield of aromatic compound in the case of using carbon dioxide as a flow gas was 59%, and the yield of aromatic compound when an inert gas was used as a flow gas Is 51%, showing a yield increase of 8%.
Through the comparative experiments of Examples 1 to 3, the yield of the aromatic compound was increased by the use of carbon dioxide, which is a result of the yield of the aromatic compound in the inert gas in the background art, Suggesting an improved yield of aromatic compounds.
While the present invention has been particularly shown and described with reference to exemplary embodiments thereof, it is to be understood that such detail is solved by the person skilled in the art without departing from the scope of the invention. will be. Accordingly, the actual scope of the present invention will be defined by the appended claims and their equivalents.
Claims (11)
Flowing said one low-carbon compound or said mixture into a flow gas;
Introducing the carbon dioxide and mixing with the flow gas;
Reacting the flow gas with the carbon dioxide under the catalyst; And
Recovering the aromatic compound produced as a result of the reaction
≪ / RTI >
Wherein the one low-carbon compound or the mixture has a weight hourly space velocity (WHSV) of 0.1 to 10 h -1 per catalyst unit.
Wherein the molar ratio of the one low-carbon compound or the mixture to the carbon dioxide is in the range of 1 to 100. The method of claim 1,
Wherein said one low carbon compound is selected from the group consisting of C 3 H 6 , C 3 H 8 , C 4 H 8 , C 4 H 10 , C 5 H 10 , C 5 H 12 , C 6 H 12 and C 6 H 14 Wherein the aromatic compound is selected from the group consisting of alumina, zirconium, zirconium, and zirconium.
Wherein the low-carbon compound is replaceable with another isomer.
Wherein the reaction is carried out at a temperature of 300 to 800 DEG C and a pressure of 0.5 to 200 atm.
Wherein the catalyst comprises at least one zeolite catalyst selected from the group consisting of an MFI type zeolite catalyst, a BEA type zeolite catalyst and a MOR type zeolite catalyst.
Wherein the zeolite catalyst contains a metal ion, metal or metal oxide of Ga, Zn, Ni, Mg, Sn, Mo, Pt, Pd and La.
Wherein the metal is contained in the two dimer metal systems or in the form of a mixture of two or more metals in the zeolite catalyst in an amount of 0.1 to 25% by weight.
Wherein the aromatic compound has a yield of 40% or more of a C 9 aromatic compound containing at least one of benzene, toluene, xylene, ethylbenzene, styrene, and durene.
Priority Applications (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| KR1020150088668A KR101795851B1 (en) | 2015-06-22 | 2015-06-22 | Method for producing aromatics from light hydrocarbons using oxygen using carbon dioxide |
Applications Claiming Priority (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| KR1020150088668A KR101795851B1 (en) | 2015-06-22 | 2015-06-22 | Method for producing aromatics from light hydrocarbons using oxygen using carbon dioxide |
Publications (2)
| Publication Number | Publication Date |
|---|---|
| KR20170000055A true KR20170000055A (en) | 2017-01-02 |
| KR101795851B1 KR101795851B1 (en) | 2017-11-13 |
Family
ID=57810455
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| KR1020150088668A KR101795851B1 (en) | 2015-06-22 | 2015-06-22 | Method for producing aromatics from light hydrocarbons using oxygen using carbon dioxide |
Country Status (1)
| Country | Link |
|---|---|
| KR (1) | KR101795851B1 (en) |
Cited By (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US11578018B1 (en) | 2021-11-03 | 2023-02-14 | Sogang University Research & Business Development Foundation | Integrated process for producing BTX and hydrogen from shale gas with feeding of carbon dioxide |
Family Cites Families (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US7932425B2 (en) | 2006-07-28 | 2011-04-26 | Chevron Phillips Chemical Company Lp | Method of enhancing an aromatization catalyst |
-
2015
- 2015-06-22 KR KR1020150088668A patent/KR101795851B1/en active IP Right Grant
Cited By (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US11578018B1 (en) | 2021-11-03 | 2023-02-14 | Sogang University Research & Business Development Foundation | Integrated process for producing BTX and hydrogen from shale gas with feeding of carbon dioxide |
Also Published As
| Publication number | Publication date |
|---|---|
| KR101795851B1 (en) | 2017-11-13 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| AU2010313369B2 (en) | Process for the conversion of mixed lower alkanes to aromatic hydrocarbons | |
| JP5110316B2 (en) | Process for producing aromatic hydrocarbons and liquefied petroleum gas from hydrocarbon mixtures | |
| EP2229349B1 (en) | Continuous process for oxygen-free conversion of methane | |
| TWI648245B (en) | Process for converting paraffin to olefin and catalyst for use therein | |
| US20110257452A1 (en) | Regenerable Composite Catalysts for Hydrocarbon Aromatization | |
| AU2010313368B2 (en) | Process for the conversion of lower alkanes to aromatic hydrocarbons | |
| US11603500B2 (en) | Natural gas liquids upgrading process: two-step catalytic process for alkane dehydrogenation and oligomerization | |
| AU2010313367B2 (en) | Process for the conversion of propane and butane to aromatic hydrocarbons | |
| EP2215038B1 (en) | Process for producing light olefins from synthesis gas using dual sequential bed reactor | |
| CA2819367A1 (en) | Process for the conversion of mixed lower alkanes to aromatic hydrocarbons | |
| KR101895497B1 (en) | Hydrocracking catalysts for the production of light alkyl-aromatic hydrocarbons, preparation method thereof and method for producing light alkyl-aromatic hydrocarbons using the same | |
| CN108602732A (en) | Cyclic annular C5The production of compound | |
| KR101795851B1 (en) | Method for producing aromatics from light hydrocarbons using oxygen using carbon dioxide | |
| CN113493699B (en) | Method for producing aromatic hydrocarbon and/or liquid fuel from light hydrocarbon | |
| CN112830858B (en) | Method for producing aromatic hydrocarbon by dehydrogenating and aromatizing light hydrocarbon | |
| Kanitkar et al. | Light alkane aromatization: efficient use of natural gas | |
| EP3303269B1 (en) | Process for the selective hydrogenation of acetylene to ethylene | |
| KR102544676B1 (en) | Catalyst for Producing Light Olefin and Producing Method of Light Olefin Using the Same | |
| JP2020196685A (en) | Catalyst system for benzene production and method for producing benzene using the same | |
| JP6977453B2 (en) | Method for producing aromatic compounds | |
| KR20220020677A (en) | Catalyst for cycle run in fixed-bed reaction system of ethylene to propylene conversion reaction and hydrogen regeneration | |
| Kamaluddin et al. | Catalytic Oxidative Cracking for Light Olefin Production | |
| US20200078775A1 (en) | Regeneration of dehydroaromatization of catalyst with constant conversion | |
| Tan et al. | BTX from Light Hydrocarbons | |
| KR20200058088A (en) | Dissimilar metal supported catalyst for preparation of aromatic compounds by dehydroaromatization of ethane, and Method for preparing aromatic compounds using the same |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| A201 | Request for examination | ||
| E902 | Notification of reason for refusal | ||
| E90F | Notification of reason for final refusal | ||
| E701 | Decision to grant or registration of patent right | ||
| GRNT | Written decision to grant |