JP7017581B2 - Paufタンパク質に特異的に結合する抗体及びその用途 - Google Patents
Paufタンパク質に特異的に結合する抗体及びその用途 Download PDFInfo
- Publication number
- JP7017581B2 JP7017581B2 JP2019555140A JP2019555140A JP7017581B2 JP 7017581 B2 JP7017581 B2 JP 7017581B2 JP 2019555140 A JP2019555140 A JP 2019555140A JP 2019555140 A JP2019555140 A JP 2019555140A JP 7017581 B2 JP7017581 B2 JP 7017581B2
- Authority
- JP
- Japan
- Prior art keywords
- antibody
- seq
- cancer
- light chain
- heavy chain
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Active
Links
Images
Classifications
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07K—PEPTIDES
- C07K16/00—Immunoglobulins [IG], e.g. monoclonal or polyclonal antibodies
- C07K16/18—Immunoglobulins [IG], e.g. monoclonal or polyclonal antibodies against material from animals or humans
- C07K16/28—Immunoglobulins [IG], e.g. monoclonal or polyclonal antibodies against material from animals or humans against receptors, cell surface antigens or cell surface determinants
- C07K16/30—Immunoglobulins [IG], e.g. monoclonal or polyclonal antibodies against material from animals or humans against receptors, cell surface antigens or cell surface determinants from tumour cells
- C07K16/303—Liver or Pancreas
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K39/00—Medicinal preparations containing antigens or antibodies
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K39/00—Medicinal preparations containing antigens or antibodies
- A61K39/395—Antibodies; Immunoglobulins; Immune serum, e.g. antilymphocytic serum
- A61K39/39533—Antibodies; Immunoglobulins; Immune serum, e.g. antilymphocytic serum against materials from animals
- A61K39/39558—Antibodies; Immunoglobulins; Immune serum, e.g. antilymphocytic serum against materials from animals against tumor tissues, cells, antigens
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K47/00—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient
- A61K47/50—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient the non-active ingredient being chemically bound to the active ingredient, e.g. polymer-drug conjugates
- A61K47/51—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient the non-active ingredient being chemically bound to the active ingredient, e.g. polymer-drug conjugates the non-active ingredient being a modifying agent
- A61K47/68—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient the non-active ingredient being chemically bound to the active ingredient, e.g. polymer-drug conjugates the non-active ingredient being a modifying agent the modifying agent being an antibody, an immunoglobulin or a fragment thereof, e.g. an Fc-fragment
- A61K47/6835—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient the non-active ingredient being chemically bound to the active ingredient, e.g. polymer-drug conjugates the non-active ingredient being a modifying agent the modifying agent being an antibody, an immunoglobulin or a fragment thereof, e.g. an Fc-fragment the modifying agent being an antibody or an immunoglobulin bearing at least one antigen-binding site
- A61K47/6851—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient the non-active ingredient being chemically bound to the active ingredient, e.g. polymer-drug conjugates the non-active ingredient being a modifying agent the modifying agent being an antibody, an immunoglobulin or a fragment thereof, e.g. an Fc-fragment the modifying agent being an antibody or an immunoglobulin bearing at least one antigen-binding site the antibody targeting a determinant of a tumour cell
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P35/00—Antineoplastic agents
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07K—PEPTIDES
- C07K16/00—Immunoglobulins [IG], e.g. monoclonal or polyclonal antibodies
- C07K16/18—Immunoglobulins [IG], e.g. monoclonal or polyclonal antibodies against material from animals or humans
- C07K16/28—Immunoglobulins [IG], e.g. monoclonal or polyclonal antibodies against material from animals or humans against receptors, cell surface antigens or cell surface determinants
- C07K16/30—Immunoglobulins [IG], e.g. monoclonal or polyclonal antibodies against material from animals or humans against receptors, cell surface antigens or cell surface determinants from tumour cells
-
- G—PHYSICS
- G01—MEASURING; TESTING
- G01N—INVESTIGATING OR ANALYSING MATERIALS BY DETERMINING THEIR CHEMICAL OR PHYSICAL PROPERTIES
- G01N33/00—Investigating or analysing materials by specific methods not covered by groups G01N1/00 - G01N31/00
- G01N33/48—Biological material, e.g. blood, urine; Haemocytometers
- G01N33/50—Chemical analysis of biological material, e.g. blood, urine; Testing involving biospecific ligand binding methods; Immunological testing
- G01N33/5005—Chemical analysis of biological material, e.g. blood, urine; Testing involving biospecific ligand binding methods; Immunological testing involving human or animal cells
- G01N33/5008—Chemical analysis of biological material, e.g. blood, urine; Testing involving biospecific ligand binding methods; Immunological testing involving human or animal cells for testing or evaluating the effect of chemical or biological compounds, e.g. drugs, cosmetics
- G01N33/502—Chemical analysis of biological material, e.g. blood, urine; Testing involving biospecific ligand binding methods; Immunological testing involving human or animal cells for testing or evaluating the effect of chemical or biological compounds, e.g. drugs, cosmetics for testing non-proliferative effects
- G01N33/5023—Chemical analysis of biological material, e.g. blood, urine; Testing involving biospecific ligand binding methods; Immunological testing involving human or animal cells for testing or evaluating the effect of chemical or biological compounds, e.g. drugs, cosmetics for testing non-proliferative effects on expression patterns
-
- G—PHYSICS
- G01—MEASURING; TESTING
- G01N—INVESTIGATING OR ANALYSING MATERIALS BY DETERMINING THEIR CHEMICAL OR PHYSICAL PROPERTIES
- G01N33/00—Investigating or analysing materials by specific methods not covered by groups G01N1/00 - G01N31/00
- G01N33/48—Biological material, e.g. blood, urine; Haemocytometers
- G01N33/50—Chemical analysis of biological material, e.g. blood, urine; Testing involving biospecific ligand binding methods; Immunological testing
- G01N33/53—Immunoassay; Biospecific binding assay; Materials therefor
- G01N33/575—Immunoassay; Biospecific binding assay; Materials therefor for cancer
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K39/00—Medicinal preparations containing antigens or antibodies
- A61K2039/505—Medicinal preparations containing antigens or antibodies comprising antibodies
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07K—PEPTIDES
- C07K2317/00—Immunoglobulins specific features
- C07K2317/20—Immunoglobulins specific features characterized by taxonomic origin
- C07K2317/24—Immunoglobulins specific features characterized by taxonomic origin containing regions, domains or residues from different species, e.g. chimeric, humanized or veneered
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07K—PEPTIDES
- C07K2317/00—Immunoglobulins specific features
- C07K2317/50—Immunoglobulins specific features characterized by immunoglobulin fragments
- C07K2317/56—Immunoglobulins specific features characterized by immunoglobulin fragments variable (Fv) region, i.e. VH and/or VL
- C07K2317/565—Complementarity determining region [CDR]
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07K—PEPTIDES
- C07K2317/00—Immunoglobulins specific features
- C07K2317/50—Immunoglobulins specific features characterized by immunoglobulin fragments
- C07K2317/56—Immunoglobulins specific features characterized by immunoglobulin fragments variable (Fv) region, i.e. VH and/or VL
- C07K2317/567—Framework region [FR]
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07K—PEPTIDES
- C07K2317/00—Immunoglobulins specific features
- C07K2317/70—Immunoglobulins specific features characterized by effect upon binding to a cell or to an antigen
- C07K2317/73—Inducing cell death, e.g. apoptosis, necrosis or inhibition of cell proliferation
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07K—PEPTIDES
- C07K2317/00—Immunoglobulins specific features
- C07K2317/90—Immunoglobulins specific features characterized by (pharmaco)kinetic aspects or by stability of the immunoglobulin
- C07K2317/92—Affinity (KD), association rate (Ka), dissociation rate (Kd) or EC50 value
-
- G—PHYSICS
- G01—MEASURING; TESTING
- G01N—INVESTIGATING OR ANALYSING MATERIALS BY DETERMINING THEIR CHEMICAL OR PHYSICAL PROPERTIES
- G01N2333/00—Assays involving biological materials from specific organisms or of a specific nature
- G01N2333/435—Assays involving biological materials from specific organisms or of a specific nature from animals; from humans
-
- G—PHYSICS
- G01—MEASURING; TESTING
- G01N—INVESTIGATING OR ANALYSING MATERIALS BY DETERMINING THEIR CHEMICAL OR PHYSICAL PROPERTIES
- G01N2500/00—Screening for compounds of potential therapeutic value
-
- G—PHYSICS
- G01—MEASURING; TESTING
- G01N—INVESTIGATING OR ANALYSING MATERIALS BY DETERMINING THEIR CHEMICAL OR PHYSICAL PROPERTIES
- G01N2500/00—Screening for compounds of potential therapeutic value
- G01N2500/10—Screening for compounds of potential therapeutic value involving cells
Landscapes
- Health & Medical Sciences (AREA)
- Life Sciences & Earth Sciences (AREA)
- Chemical & Material Sciences (AREA)
- Immunology (AREA)
- Engineering & Computer Science (AREA)
- Medicinal Chemistry (AREA)
- General Health & Medical Sciences (AREA)
- Molecular Biology (AREA)
- Biomedical Technology (AREA)
- Organic Chemistry (AREA)
- Cell Biology (AREA)
- Urology & Nephrology (AREA)
- Hematology (AREA)
- Biochemistry (AREA)
- Microbiology (AREA)
- Public Health (AREA)
- Pharmacology & Pharmacy (AREA)
- Animal Behavior & Ethology (AREA)
- Veterinary Medicine (AREA)
- Bioinformatics & Cheminformatics (AREA)
- Food Science & Technology (AREA)
- Pathology (AREA)
- General Physics & Mathematics (AREA)
- Analytical Chemistry (AREA)
- Physics & Mathematics (AREA)
- Biotechnology (AREA)
- Genetics & Genomics (AREA)
- Biophysics (AREA)
- Proteomics, Peptides & Aminoacids (AREA)
- Epidemiology (AREA)
- Gastroenterology & Hepatology (AREA)
- General Chemical & Material Sciences (AREA)
- Nuclear Medicine, Radiotherapy & Molecular Imaging (AREA)
- Chemical Kinetics & Catalysis (AREA)
- Mycology (AREA)
- Tropical Medicine & Parasitology (AREA)
- Toxicology (AREA)
- Oncology (AREA)
- Peptides Or Proteins (AREA)
- Medicines Containing Antibodies Or Antigens For Use As Internal Diagnostic Agents (AREA)
Description
具体的には、配列番号1の重鎖CDR1;配列番号2または39の重鎖CDR2;及び配列番号3の重鎖CDR3を含む重鎖可変領域及び配列番号5の軽鎖CDR1;配列番号6の軽鎖CDR2;及び配列番号7の軽鎖CDR3を含む軽鎖可変領域を含むか、
具体的には、配列番号1の重鎖CDR1;配列番号2または39の重鎖CDR2;及び配列番号30の重鎖CDR3を含む重鎖可変領域及び配列番号5の軽鎖CDR1;配列番号6の軽鎖CDR2;及び配列番号31の軽鎖CDR3を含む軽鎖可変領域を含むか、または
実施例1-1.2H2及び20C5の発掘
PAUFタンパク質を標的とする抗体を開発するために、まずPUAFタンパク質に結合するマウスモノクローナル抗体を用いた。
前記実施例1-1で発掘した2H2、4E6、11G6及び20C5の4種の抗体中、PUAFタンパク質に親和度が最も高い抗体を発掘するために、次のような方法を行った。
前記実施例1-1で発掘した2H2または20C5のエピトープを確認するために、まず、韓国登録特許公報第10-1098186号(特許文献1)に公知となったPAUF特異的なヒトモノクローナル抗体である8F3、即ち、PMAb83と同一なエピトープを有するかを確認した。
実施例2-1.cPMAb22またはcPMAb205の製造
前記実施例1で発掘した2H2または20C5マウス抗体の拒否反応を最小限に抑えるために、前記抗体の可変領域は維持し、不変領域のみヒト由来のタンパク質に変えたキメラ抗体を製造した。
前記実施例2-1で製造したcPMAb22またはcPMAb205のPUAFタンパク質に対する特異度を確認するために、次のような方法を行った。
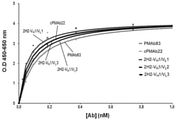
前記実施例2-1で製造したcPMAb22またはcPMAb205のPUAFタンパク質に対する親和度を確認するために、次のような方法を行った。
前記実施例2-1で製造したキメラ抗体cPMAb22の癌に対する治療効果を確認するために、膵臓癌細胞株BxPC-3に対する増殖抑制効果を確認した。
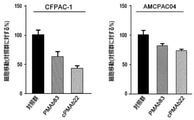
前記実施例2-1で製造したキメラ抗体cPMAb22の癌に対する治療効果を確認するために、膵臓癌細胞株CFPAC-1及び膵臓癌の患者に由来する膵臓癌細胞AMCPAC04の移動抑制効果を確認した。
前記実施例2-1で製造したキメラ抗体cPMAb22の癌に対する治療効果を確認するために、膵臓癌細胞株CFPAC-1及び膵臓癌患者に由来する膵臓癌細胞であるAMCPAC04に対する浸潤抑制効果を確認した。
実施例3-1.hPMAb22の製造
前記実施例1で発掘した2H2マウス抗体及び前記実施例2で製造したcPMAb22キメラ抗体の拒否反応を最小限に抑えるために、2H2及びcPMAb22のCDR(マウス由来)は維持し、CDRを除いた可変領域及び不変領域はヒト由来のタンパク質に変えたヒト化抗体を製造した。
前記実施例3-1で製造したヒト化抗体hPMAb22のPUAFタンパク質に対する特異度を確認するために、前記実施例2-2による方法を行った。
前記実施例3-1で製造したヒト化抗体hPMAb22のPUAFタンパク質に対する親和度を確認するために、次のような方法を行った。
前記実施例3-1で製造したhPMAb22のエピトープを確認するために、まず既存の抗PAUFヒト抗体であるPMAb83とエピトープが同一かを前記実施例1~3に基づいて確認した。
前記実施例3-1で製造したヒト化抗体hPMAb22の癌に対する治療効果を確認するために、膵臓癌細胞株CFPAC-1に対する増殖抑制効果を、前記実施例2-4に係る方法で確認した。
前記実施例3-1で製造したヒト化抗体hPMAb22の癌に対する治療効果を確認するために、膵臓癌患者に由来する膵臓癌細胞AMCPAC06及び卵巣癌細胞株OVCAR-5に対する移動抑制効果を、前記実施例2-5による方法で確認した。
前記実施例3-1で製造したヒト化抗体hPMAb22の癌に対する治療効果を確認するために、膵臓癌患者に由来する膵臓癌細胞AMCPAC04またはAMCPAC06及び卵巣癌細胞株OVCAR-5に対する浸潤抑制効果を、実施例2-6による方法で確認した。
前記実施例3-5~3-7を通じてPAUFタンパク質に特異的なヒト化抗体hPMAb22は試験管内(in vitro)で癌細胞の増殖、移動及び浸潤を抑制することを確認することにより、生体内(in vivo)でも同一な抗癌効果を示すかを確認した。
Claims (14)
- 配列番号1の重鎖CDR1;配列番号2または39の重鎖CDR2;及び配列番号3、4または30の重鎖CDR3を含む重鎖可変領域及び
配列番号5の軽鎖CDR1;配列番号6の軽鎖CDR2;及び配列番号7または31の軽鎖CDR3を含む軽鎖可変領域を含む、PAUF(Pancreatic adenocarcinoma upregulated factor)タンパク質に結合する抗体。 - 前記抗体の重鎖可変領域は、配列番号8、19、または32の重鎖FR1;配列番号9または20の重鎖FR2;配列番号10、21、または33の重鎖FR3;及び配列番号11、12、22、23、または34の重鎖FR4を含み、
前記抗体の軽鎖可変領域は、配列番号13、24、または35の軽鎖FR1;配列番号14または25の軽鎖FR2;配列番号15または26の軽鎖FR3;及び配列番号16、27または36の軽鎖FR4を含む、請求項1に記載の抗体。 - 前記重鎖可変領域は、配列番号17、28、または37のアミノ酸配列からなり;
前記軽鎖可変領域は、配列番号18、29、または38のアミノ酸配列からなるものである、請求項1に記載の抗体。 - 請求項1~3のいずれか一項に記載の抗体をコードするポリヌクレオチド。
- 請求項4に記載のポリヌクレオチドを含む発現ベクター。
- 請求項5に記載の発現ベクターが導入された形質転換体。
- 請求項1~3のいずれか一項に記載の抗体を含む、癌の予防または治療用医薬組成物。
- 請求項1~3のいずれか一項に記載の抗体をヒト以外の個体に投与する段階を含む、癌の予防または治療方法。
- 請求項1~3のいずれか一項に記載の抗体をヒト以外の個体に投与する段階を含む、癌細胞の増殖、移動、または浸潤を抑制する方法。
- 請求項1~3のいずれか一項に記載の抗体に薬物が結合された、抗体-薬物結合体。
- 請求項1~3のいずれか一項に記載の抗体を含む、癌診断用組成物。
- 請求項11に記載の組成物を含む、癌診断用キット。
- 請求項1~3のいずれか一項に記載の抗体を用いて、癌が疑われる個体の分離された生物学的試料からPAUF(Pancreatic adenocarcinoma upregulated factor)タンパク質を抗原抗体反応を通じて検出する段階を含む、癌の診断のための情報を提供する方法。
- (a)個体から分離された癌細胞に癌の予防または治療候補物質を処理する段階;
(b)前記候補物質が処理された癌細胞において、請求項1~3のいずれか一項に記載の抗体を用いて、PAUF(Pancreatic adenocarcinoma upregulated factor)タンパク質のレベルを測定する段階;及び
(c)前記段階(b)のPAUFタンパク質のレベルが候補物質を処理していない癌細胞のものより減少する場合、前記(a)段階で処理した候補物質を癌の予防または治療物質と判断する段階を含む、癌の予防または治療物質のスクリーニング方法。
Applications Claiming Priority (3)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| KR1020170096370A KR101856904B1 (ko) | 2017-07-28 | 2017-07-28 | Pauf 단백질에 특이적으로 결합하는 항체 및 이의 용도 |
| KR10-2017-0096370 | 2017-07-28 | ||
| PCT/KR2017/008893 WO2019022281A1 (ko) | 2017-07-28 | 2017-08-16 | Pauf 단백질에 특이적으로 결합하는 항체 및 이의 용도 |
Publications (2)
| Publication Number | Publication Date |
|---|---|
| JP2020520230A JP2020520230A (ja) | 2020-07-09 |
| JP7017581B2 true JP7017581B2 (ja) | 2022-03-03 |
Family
ID=62185493
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| JP2019555140A Active JP7017581B2 (ja) | 2017-07-28 | 2017-08-16 | Paufタンパク質に特異的に結合する抗体及びその用途 |
Country Status (20)
| Country | Link |
|---|---|
| US (1) | US11046779B2 (ja) |
| EP (1) | EP3660052B1 (ja) |
| JP (1) | JP7017581B2 (ja) |
| KR (1) | KR101856904B1 (ja) |
| CN (1) | CN110291110B (ja) |
| AR (1) | AR112579A1 (ja) |
| AU (1) | AU2017425111B2 (ja) |
| BR (1) | BR112019010687A2 (ja) |
| CA (1) | CA3044216C (ja) |
| ES (1) | ES3053649T3 (ja) |
| IL (1) | IL266576B2 (ja) |
| MX (1) | MX2020001027A (ja) |
| MY (1) | MY192517A (ja) |
| PH (1) | PH12020500172A1 (ja) |
| PL (1) | PL3660052T3 (ja) |
| RU (1) | RU2735102C1 (ja) |
| SG (1) | SG11201903573RA (ja) |
| TW (1) | TWI703155B (ja) |
| WO (1) | WO2019022281A1 (ja) |
| ZA (1) | ZA202001042B (ja) |
Citations (2)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2007145466A1 (en) | 2006-06-14 | 2007-12-21 | Lg Life Sciences Ltd. | Gene family (lbfl313) associated with pancreatic cancer |
| KR101098186B1 (ko) | 2008-09-23 | 2011-12-23 | 한국생명공학연구원 | 피에이유에프(pauf) 특이적인 인간 단일클론항체, 이를 포함하는 암 치료용 조성물, 이를 이용하는 암의 진단방법 |
Family Cites Families (3)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| EP2703001A1 (en) * | 2012-08-26 | 2014-03-05 | XAX Kft. | Tumor vaccination |
| CN104936984B (zh) | 2012-11-16 | 2018-09-14 | 江原大学校产学协力团 | 特异性结合人和小鼠l1cam的抗体及其用途 |
| WO2019022274A1 (ko) | 2017-07-28 | 2019-01-31 | 동아대학교 산학협력단 | Pauf 단백질에 특이적으로 결합하는 항체 및 이의 용도 |
-
2017
- 2017-07-28 KR KR1020170096370A patent/KR101856904B1/ko active Active
- 2017-08-16 AU AU2017425111A patent/AU2017425111B2/en active Active
- 2017-08-16 SG SG11201903573RA patent/SG11201903573RA/en unknown
- 2017-08-16 CN CN201780072925.9A patent/CN110291110B/zh active Active
- 2017-08-16 PL PL17919096.2T patent/PL3660052T3/pl unknown
- 2017-08-16 US US16/346,928 patent/US11046779B2/en active Active
- 2017-08-16 ES ES17919096T patent/ES3053649T3/es active Active
- 2017-08-16 EP EP17919096.2A patent/EP3660052B1/en active Active
- 2017-08-16 RU RU2019113308A patent/RU2735102C1/ru active
- 2017-08-16 WO PCT/KR2017/008893 patent/WO2019022281A1/ko not_active Ceased
- 2017-08-16 IL IL266576A patent/IL266576B2/en unknown
- 2017-08-16 JP JP2019555140A patent/JP7017581B2/ja active Active
- 2017-08-16 MY MYPI2020000291A patent/MY192517A/en unknown
- 2017-08-16 BR BR112019010687-1A patent/BR112019010687A2/pt active Search and Examination
- 2017-08-16 CA CA3044216A patent/CA3044216C/en active Active
- 2017-08-16 MX MX2020001027A patent/MX2020001027A/es unknown
-
2018
- 2018-07-26 TW TW107125876A patent/TWI703155B/zh active
- 2018-07-26 AR ARP180102098 patent/AR112579A1/es unknown
-
2020
- 2020-01-23 PH PH12020500172A patent/PH12020500172A1/en unknown
- 2020-02-19 ZA ZA2020/01042A patent/ZA202001042B/en unknown
Patent Citations (2)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2007145466A1 (en) | 2006-06-14 | 2007-12-21 | Lg Life Sciences Ltd. | Gene family (lbfl313) associated with pancreatic cancer |
| KR101098186B1 (ko) | 2008-09-23 | 2011-12-23 | 한국생명공학연구원 | 피에이유에프(pauf) 특이적인 인간 단일클론항체, 이를 포함하는 암 치료용 조성물, 이를 이용하는 암의 진단방법 |
Non-Patent Citations (1)
| Title |
|---|
| Cancer Sci.,2009年,Vol.100,p.828-836 |
Also Published As
| Publication number | Publication date |
|---|---|
| CA3044216C (en) | 2023-05-23 |
| NZ760760A (en) | 2024-04-26 |
| CN110291110A (zh) | 2019-09-27 |
| CA3044216A1 (en) | 2019-01-31 |
| TW201910350A (zh) | 2019-03-16 |
| EP3660052B1 (en) | 2025-09-03 |
| MY192517A (en) | 2022-08-25 |
| MX2020001027A (es) | 2020-08-06 |
| IL266576B2 (en) | 2024-06-01 |
| SG11201903573RA (en) | 2019-05-30 |
| EP3660052A4 (en) | 2021-04-14 |
| AR112579A1 (es) | 2019-11-13 |
| PH12020500172A1 (en) | 2020-10-12 |
| PL3660052T3 (pl) | 2026-01-26 |
| ES3053649T3 (en) | 2026-01-23 |
| AU2017425111B2 (en) | 2020-06-18 |
| ZA202001042B (en) | 2021-05-26 |
| US11046779B2 (en) | 2021-06-29 |
| IL266576B1 (en) | 2024-02-01 |
| US20200055951A1 (en) | 2020-02-20 |
| CN110291110B (zh) | 2023-11-24 |
| AU2017425111A1 (en) | 2019-05-16 |
| WO2019022281A1 (ko) | 2019-01-31 |
| IL266576A (en) | 2019-07-31 |
| EP3660052A1 (en) | 2020-06-03 |
| BR112019010687A2 (pt) | 2020-02-18 |
| JP2020520230A (ja) | 2020-07-09 |
| RU2735102C1 (ru) | 2020-10-28 |
| TWI703155B (zh) | 2020-09-01 |
| KR101856904B1 (ko) | 2018-05-11 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| CA2855699C (en) | Anti-human trop-2 antibody having an antitumor activity in vivo | |
| CA2669731C (en) | Anti-human dlk-1 antibody showing anti-tumor activity in vivo | |
| US8017118B2 (en) | Anti-hDlk-1 antibody having an antitumor activity in vivo | |
| US11220543B2 (en) | Antibody binding specifically to CD66c and use thereof | |
| CN114437227A (zh) | 双特异抗体及其应用 | |
| EP3543259A2 (en) | Antibody binding to carbonic anhydrase and use thereof | |
| KR20090130335A (ko) | 암 세포 세포독성을 매개하는 인간화 및 키메라 항-cd59 항체 | |
| JP7017581B2 (ja) | Paufタンパク質に特異的に結合する抗体及びその用途 | |
| HK40025609A (en) | Antibody specifically binding to pauf protein and use thereof | |
| HK40025609B (en) | Antibody specifically binding to pauf protein and use thereof | |
| HK40073393A (en) | Bispecific antibody and use thereof | |
| WO2023190465A1 (ja) | 抗ヒトsema7a抗体 |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| A521 | Request for written amendment filed |
Free format text: JAPANESE INTERMEDIATE CODE: A523 Effective date: 20191118 |
|
| A621 | Written request for application examination |
Free format text: JAPANESE INTERMEDIATE CODE: A621 Effective date: 20191017 |
|
| A521 | Request for written amendment filed |
Free format text: JAPANESE INTERMEDIATE CODE: A523 Effective date: 20200515 |
|
| A131 | Notification of reasons for refusal |
Free format text: JAPANESE INTERMEDIATE CODE: A131 Effective date: 20201116 |
|
| A521 | Request for written amendment filed |
Free format text: JAPANESE INTERMEDIATE CODE: A523 Effective date: 20210201 |
|
| A131 | Notification of reasons for refusal |
Free format text: JAPANESE INTERMEDIATE CODE: A131 Effective date: 20210712 |
|
| A521 | Request for written amendment filed |
Free format text: JAPANESE INTERMEDIATE CODE: A523 Effective date: 20210921 |
|
| TRDD | Decision of grant or rejection written | ||
| A01 | Written decision to grant a patent or to grant a registration (utility model) |
Free format text: JAPANESE INTERMEDIATE CODE: A01 Effective date: 20220124 |
|
| A61 | First payment of annual fees (during grant procedure) |
Free format text: JAPANESE INTERMEDIATE CODE: A61 Effective date: 20220127 |
|
| R150 | Certificate of patent or registration of utility model |
Ref document number: 7017581 Country of ref document: JP Free format text: JAPANESE INTERMEDIATE CODE: R150 |
|
| R250 | Receipt of annual fees |
Free format text: JAPANESE INTERMEDIATE CODE: R250 |
|
| R250 | Receipt of annual fees |
Free format text: JAPANESE INTERMEDIATE CODE: R250 |