JP6526623B2 - 固定及び動的安定化のためにバルーンと共に使用される角張ったリング及び接合箔 - Google Patents
固定及び動的安定化のためにバルーンと共に使用される角張ったリング及び接合箔 Download PDFInfo
- Publication number
- JP6526623B2 JP6526623B2 JP2016500744A JP2016500744A JP6526623B2 JP 6526623 B2 JP6526623 B2 JP 6526623B2 JP 2016500744 A JP2016500744 A JP 2016500744A JP 2016500744 A JP2016500744 A JP 2016500744A JP 6526623 B2 JP6526623 B2 JP 6526623B2
- Authority
- JP
- Japan
- Prior art keywords
- balloon
- volume
- bone
- cavity
- disc
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Expired - Fee Related
Links
- 239000011888 foil Substances 0.000 title claims description 38
- 230000006641 stabilisation Effects 0.000 title description 9
- 238000011105 stabilization Methods 0.000 title description 9
- 239000000463 material Substances 0.000 claims description 71
- 210000000988 bone and bone Anatomy 0.000 claims description 46
- 206010061246 Intervertebral disc degeneration Diseases 0.000 claims description 21
- 208000018180 degenerative disc disease Diseases 0.000 claims description 16
- 208000021600 intervertebral disc degenerative disease Diseases 0.000 claims description 16
- -1 polyethylene Polymers 0.000 claims description 12
- 239000004696 Poly ether ether ketone Substances 0.000 claims description 11
- 229920001971 elastomer Polymers 0.000 claims description 11
- 229920002530 polyetherether ketone Polymers 0.000 claims description 11
- 239000004698 Polyethylene Substances 0.000 claims description 8
- 239000004433 Thermoplastic polyurethane Substances 0.000 claims description 8
- 229920001652 poly(etherketoneketone) Polymers 0.000 claims description 8
- 239000004814 polyurethane Substances 0.000 claims description 8
- 229920002803 thermoplastic polyurethane Polymers 0.000 claims description 8
- JUPQTSLXMOCDHR-UHFFFAOYSA-N benzene-1,4-diol;bis(4-fluorophenyl)methanone Chemical compound OC1=CC=C(O)C=C1.C1=CC(F)=CC=C1C(=O)C1=CC=C(F)C=C1 JUPQTSLXMOCDHR-UHFFFAOYSA-N 0.000 claims description 7
- 229920000139 polyethylene terephthalate Polymers 0.000 claims description 7
- 239000005020 polyethylene terephthalate Substances 0.000 claims description 7
- 230000007547 defect Effects 0.000 claims description 6
- 229920000573 polyethylene Polymers 0.000 claims description 4
- 229920002635 polyurethane Polymers 0.000 claims description 4
- 229920002379 silicone rubber Polymers 0.000 claims description 4
- 239000004945 silicone rubber Substances 0.000 claims description 4
- 229920001692 polycarbonate urethane Polymers 0.000 claims description 3
- 238000005553 drilling Methods 0.000 claims 2
- 238000000034 method Methods 0.000 description 63
- 238000005520 cutting process Methods 0.000 description 20
- 238000003780 insertion Methods 0.000 description 20
- 230000037431 insertion Effects 0.000 description 20
- 238000003466 welding Methods 0.000 description 15
- 238000001356 surgical procedure Methods 0.000 description 12
- 229920000642 polymer Polymers 0.000 description 11
- 238000005516 engineering process Methods 0.000 description 9
- 239000007943 implant Substances 0.000 description 9
- 238000013459 approach Methods 0.000 description 8
- 239000002639 bone cement Substances 0.000 description 8
- 230000007850 degeneration Effects 0.000 description 8
- 239000000806 elastomer Substances 0.000 description 7
- FAPWRFPIFSIZLT-UHFFFAOYSA-M Sodium chloride Chemical compound [Na+].[Cl-] FAPWRFPIFSIZLT-UHFFFAOYSA-M 0.000 description 6
- 238000013016 damping Methods 0.000 description 6
- 239000012530 fluid Substances 0.000 description 6
- 239000007788 liquid Substances 0.000 description 6
- 229910052751 metal Inorganic materials 0.000 description 6
- 239000002184 metal Substances 0.000 description 6
- 210000003484 anatomy Anatomy 0.000 description 5
- 230000008901 benefit Effects 0.000 description 5
- 238000000576 coating method Methods 0.000 description 5
- 230000000694 effects Effects 0.000 description 5
- 230000006870 function Effects 0.000 description 5
- 239000000203 mixture Substances 0.000 description 5
- 230000000087 stabilizing effect Effects 0.000 description 5
- 230000010339 dilation Effects 0.000 description 4
- 201000010099 disease Diseases 0.000 description 4
- 208000037265 diseases, disorders, signs and symptoms Diseases 0.000 description 4
- 238000009826 distribution Methods 0.000 description 4
- 238000005304 joining Methods 0.000 description 4
- 230000003902 lesion Effects 0.000 description 4
- 210000005036 nerve Anatomy 0.000 description 4
- 229920003229 poly(methyl methacrylate) Polymers 0.000 description 4
- 239000004926 polymethyl methacrylate Substances 0.000 description 4
- 229920002614 Polyether block amide Polymers 0.000 description 3
- 238000010521 absorption reaction Methods 0.000 description 3
- 239000000853 adhesive Substances 0.000 description 3
- 230000001070 adhesive effect Effects 0.000 description 3
- 210000001185 bone marrow Anatomy 0.000 description 3
- 239000001506 calcium phosphate Substances 0.000 description 3
- 229910000389 calcium phosphate Inorganic materials 0.000 description 3
- 235000011010 calcium phosphates Nutrition 0.000 description 3
- 230000008859 change Effects 0.000 description 3
- 238000006243 chemical reaction Methods 0.000 description 3
- 239000011248 coating agent Substances 0.000 description 3
- 239000002872 contrast media Substances 0.000 description 3
- 230000003412 degenerative effect Effects 0.000 description 3
- 239000000945 filler Substances 0.000 description 3
- 230000004927 fusion Effects 0.000 description 3
- 229910052588 hydroxylapatite Inorganic materials 0.000 description 3
- XYJRXVWERLGGKC-UHFFFAOYSA-D pentacalcium;hydroxide;triphosphate Chemical compound [OH-].[Ca+2].[Ca+2].[Ca+2].[Ca+2].[Ca+2].[O-]P([O-])([O-])=O.[O-]P([O-])([O-])=O.[O-]P([O-])([O-])=O XYJRXVWERLGGKC-UHFFFAOYSA-D 0.000 description 3
- 229920001296 polysiloxane Polymers 0.000 description 3
- 230000008569 process Effects 0.000 description 3
- 238000011084 recovery Methods 0.000 description 3
- 238000005096 rolling process Methods 0.000 description 3
- 239000011780 sodium chloride Substances 0.000 description 3
- QORWJWZARLRLPR-UHFFFAOYSA-H tricalcium bis(phosphate) Chemical compound [Ca+2].[Ca+2].[Ca+2].[O-]P([O-])([O-])=O.[O-]P([O-])([O-])=O QORWJWZARLRLPR-UHFFFAOYSA-H 0.000 description 3
- 238000002604 ultrasonography Methods 0.000 description 3
- XLYOFNOQVPJJNP-UHFFFAOYSA-N water Substances O XLYOFNOQVPJJNP-UHFFFAOYSA-N 0.000 description 3
- 208000003618 Intervertebral Disc Displacement Diseases 0.000 description 2
- 208000002193 Pain Diseases 0.000 description 2
- 206010058907 Spinal deformity Diseases 0.000 description 2
- 206010052428 Wound Diseases 0.000 description 2
- 238000002399 angioplasty Methods 0.000 description 2
- 230000036760 body temperature Effects 0.000 description 2
- 230000001112 coagulating effect Effects 0.000 description 2
- 238000005345 coagulation Methods 0.000 description 2
- 230000015271 coagulation Effects 0.000 description 2
- 239000002131 composite material Substances 0.000 description 2
- 238000012937 correction Methods 0.000 description 2
- 239000013013 elastic material Substances 0.000 description 2
- 239000012634 fragment Substances 0.000 description 2
- 239000011521 glass Substances 0.000 description 2
- 239000000017 hydrogel Substances 0.000 description 2
- 208000014674 injury Diseases 0.000 description 2
- 238000004519 manufacturing process Methods 0.000 description 2
- 239000003550 marker Substances 0.000 description 2
- 238000002324 minimally invasive surgery Methods 0.000 description 2
- 239000011414 polymer cement Substances 0.000 description 2
- 230000009467 reduction Effects 0.000 description 2
- 238000000926 separation method Methods 0.000 description 2
- 230000035939 shock Effects 0.000 description 2
- 239000007787 solid Substances 0.000 description 2
- 125000006850 spacer group Chemical group 0.000 description 2
- 210000000278 spinal cord Anatomy 0.000 description 2
- 239000000126 substance Substances 0.000 description 2
- 210000001519 tissue Anatomy 0.000 description 2
- 230000008733 trauma Effects 0.000 description 2
- 206010061728 Bone lesion Diseases 0.000 description 1
- 208000000094 Chronic Pain Diseases 0.000 description 1
- JOYRKODLDBILNP-UHFFFAOYSA-N Ethyl urethane Chemical compound CCOC(N)=O JOYRKODLDBILNP-UHFFFAOYSA-N 0.000 description 1
- 229910000861 Mg alloy Inorganic materials 0.000 description 1
- 240000005561 Musa balbisiana Species 0.000 description 1
- 235000018290 Musa x paradisiaca Nutrition 0.000 description 1
- 239000004952 Polyamide Substances 0.000 description 1
- 208000020339 Spinal injury Diseases 0.000 description 1
- RTAQQCXQSZGOHL-UHFFFAOYSA-N Titanium Chemical compound [Ti] RTAQQCXQSZGOHL-UHFFFAOYSA-N 0.000 description 1
- 208000027418 Wounds and injury Diseases 0.000 description 1
- WAIPAZQMEIHHTJ-UHFFFAOYSA-N [Cr].[Co] Chemical class [Cr].[Co] WAIPAZQMEIHHTJ-UHFFFAOYSA-N 0.000 description 1
- 238000004026 adhesive bonding Methods 0.000 description 1
- 230000032683 aging Effects 0.000 description 1
- 238000004873 anchoring Methods 0.000 description 1
- 210000001367 artery Anatomy 0.000 description 1
- 238000011882 arthroplasty Methods 0.000 description 1
- 239000012620 biological material Substances 0.000 description 1
- 230000015572 biosynthetic process Effects 0.000 description 1
- 210000004204 blood vessel Anatomy 0.000 description 1
- 230000008468 bone growth Effects 0.000 description 1
- 210000000459 calcaneus Anatomy 0.000 description 1
- 210000000845 cartilage Anatomy 0.000 description 1
- 230000015556 catabolic process Effects 0.000 description 1
- 239000004568 cement Substances 0.000 description 1
- 230000001684 chronic effect Effects 0.000 description 1
- 238000004140 cleaning Methods 0.000 description 1
- 239000004035 construction material Substances 0.000 description 1
- 229920001577 copolymer Polymers 0.000 description 1
- 238000002425 crystallisation Methods 0.000 description 1
- 230000008025 crystallization Effects 0.000 description 1
- 230000006378 damage Effects 0.000 description 1
- 230000006837 decompression Effects 0.000 description 1
- 238000006731 degradation reaction Methods 0.000 description 1
- 239000004744 fabric Substances 0.000 description 1
- 239000000499 gel Substances 0.000 description 1
- 238000010438 heat treatment Methods 0.000 description 1
- 238000002513 implantation Methods 0.000 description 1
- 238000011065 in-situ storage Methods 0.000 description 1
- 210000003734 kidney Anatomy 0.000 description 1
- 238000002595 magnetic resonance imaging Methods 0.000 description 1
- 238000012423 maintenance Methods 0.000 description 1
- 239000012528 membrane Substances 0.000 description 1
- 150000002739 metals Chemical class 0.000 description 1
- 230000004048 modification Effects 0.000 description 1
- 238000012986 modification Methods 0.000 description 1
- 210000000944 nerve tissue Anatomy 0.000 description 1
- 229910001000 nickel titanium Inorganic materials 0.000 description 1
- HLXZNVUGXRDIFK-UHFFFAOYSA-N nickel titanium Chemical compound [Ti].[Ti].[Ti].[Ti].[Ti].[Ti].[Ti].[Ti].[Ti].[Ti].[Ti].[Ni].[Ni].[Ni].[Ni].[Ni].[Ni].[Ni].[Ni].[Ni].[Ni].[Ni].[Ni].[Ni].[Ni] HLXZNVUGXRDIFK-UHFFFAOYSA-N 0.000 description 1
- 238000005457 optimization Methods 0.000 description 1
- 238000012856 packing Methods 0.000 description 1
- 239000002245 particle Substances 0.000 description 1
- 239000011236 particulate material Substances 0.000 description 1
- 230000035515 penetration Effects 0.000 description 1
- 230000002093 peripheral effect Effects 0.000 description 1
- 230000035699 permeability Effects 0.000 description 1
- VPRUMANMDWQMNF-UHFFFAOYSA-N phenylethane boronic acid Chemical compound OB(O)CCC1=CC=CC=C1 VPRUMANMDWQMNF-UHFFFAOYSA-N 0.000 description 1
- 229920000747 poly(lactic acid) Polymers 0.000 description 1
- 229920002647 polyamide Polymers 0.000 description 1
- 229920000515 polycarbonate Polymers 0.000 description 1
- 239000004417 polycarbonate Substances 0.000 description 1
- 239000004848 polyfunctional curative Substances 0.000 description 1
- 239000004626 polylactic acid Substances 0.000 description 1
- 238000006116 polymerization reaction Methods 0.000 description 1
- 230000002035 prolonged effect Effects 0.000 description 1
- 239000003223 protective agent Substances 0.000 description 1
- 230000002787 reinforcement Effects 0.000 description 1
- 150000003839 salts Chemical class 0.000 description 1
- 239000000523 sample Substances 0.000 description 1
- 238000007789 sealing Methods 0.000 description 1
- 229910001285 shape-memory alloy Inorganic materials 0.000 description 1
- 210000004872 soft tissue Anatomy 0.000 description 1
- 238000003892 spreading Methods 0.000 description 1
- 230000007480 spreading Effects 0.000 description 1
- 239000010935 stainless steel Substances 0.000 description 1
- 229910001220 stainless steel Inorganic materials 0.000 description 1
- 230000002966 stenotic effect Effects 0.000 description 1
- 230000003319 supportive effect Effects 0.000 description 1
- 208000024891 symptom Diseases 0.000 description 1
- KKEYFWRCBNTPAC-UHFFFAOYSA-L terephthalate(2-) Chemical compound [O-]C(=O)C1=CC=C(C([O-])=O)C=C1 KKEYFWRCBNTPAC-UHFFFAOYSA-L 0.000 description 1
- 229920001169 thermoplastic Polymers 0.000 description 1
- 239000004416 thermosoftening plastic Substances 0.000 description 1
- 239000010936 titanium Substances 0.000 description 1
- 229910052719 titanium Inorganic materials 0.000 description 1
- 238000012876 topography Methods 0.000 description 1
Images
Classifications
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61F—FILTERS IMPLANTABLE INTO BLOOD VESSELS; PROSTHESES; DEVICES PROVIDING PATENCY TO, OR PREVENTING COLLAPSING OF, TUBULAR STRUCTURES OF THE BODY, e.g. STENTS; ORTHOPAEDIC, NURSING OR CONTRACEPTIVE DEVICES; FOMENTATION; TREATMENT OR PROTECTION OF EYES OR EARS; BANDAGES, DRESSINGS OR ABSORBENT PADS; FIRST-AID KITS
- A61F2/00—Filters implantable into blood vessels; Prostheses, i.e. artificial substitutes or replacements for parts of the body; Appliances for connecting them with the body; Devices providing patency to, or preventing collapsing of, tubular structures of the body, e.g. stents
- A61F2/02—Prostheses implantable into the body
- A61F2/30—Joints
- A61F2/44—Joints for the spine, e.g. vertebrae, spinal discs
- A61F2/441—Joints for the spine, e.g. vertebrae, spinal discs made of inflatable pockets or chambers filled with fluid, e.g. with hydrogel
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61B—DIAGNOSIS; SURGERY; IDENTIFICATION
- A61B17/00—Surgical instruments, devices or methods, e.g. tourniquets
- A61B17/56—Surgical instruments or methods for treatment of bones or joints; Devices specially adapted therefor
- A61B17/58—Surgical instruments or methods for treatment of bones or joints; Devices specially adapted therefor for osteosynthesis, e.g. bone plates, screws, setting implements or the like
- A61B17/68—Internal fixation devices, including fasteners and spinal fixators, even if a part thereof projects from the skin
- A61B17/70—Spinal positioners or stabilisers ; Bone stabilisers comprising fluid filler in an implant
- A61B17/7097—Stabilisers comprising fluid filler in an implant, e.g. balloon; devices for inserting or filling such implants
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61B—DIAGNOSIS; SURGERY; IDENTIFICATION
- A61B17/00—Surgical instruments, devices or methods, e.g. tourniquets
- A61B17/56—Surgical instruments or methods for treatment of bones or joints; Devices specially adapted therefor
- A61B17/58—Surgical instruments or methods for treatment of bones or joints; Devices specially adapted therefor for osteosynthesis, e.g. bone plates, screws, setting implements or the like
- A61B17/88—Osteosynthesis instruments; Methods or means for implanting or extracting internal or external fixation devices
- A61B17/885—Tools for expanding or compacting bones or discs or cavities therein
- A61B17/8852—Tools for expanding or compacting bones or discs or cavities therein capable of being assembled or enlarged, or changing shape, inside the bone or disc
- A61B17/8855—Tools for expanding or compacting bones or discs or cavities therein capable of being assembled or enlarged, or changing shape, inside the bone or disc inflatable, e.g. kyphoplasty balloons
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61F—FILTERS IMPLANTABLE INTO BLOOD VESSELS; PROSTHESES; DEVICES PROVIDING PATENCY TO, OR PREVENTING COLLAPSING OF, TUBULAR STRUCTURES OF THE BODY, e.g. STENTS; ORTHOPAEDIC, NURSING OR CONTRACEPTIVE DEVICES; FOMENTATION; TREATMENT OR PROTECTION OF EYES OR EARS; BANDAGES, DRESSINGS OR ABSORBENT PADS; FIRST-AID KITS
- A61F2/00—Filters implantable into blood vessels; Prostheses, i.e. artificial substitutes or replacements for parts of the body; Appliances for connecting them with the body; Devices providing patency to, or preventing collapsing of, tubular structures of the body, e.g. stents
- A61F2/02—Prostheses implantable into the body
- A61F2/30—Joints
- A61F2/46—Special tools or methods for implanting or extracting artificial joints, accessories, bone grafts or substitutes, or particular adaptations therefor
- A61F2/4603—Special tools or methods for implanting or extracting artificial joints, accessories, bone grafts or substitutes, or particular adaptations therefor for insertion or extraction of endoprosthetic joints or of accessories thereof
- A61F2/4611—Special tools or methods for implanting or extracting artificial joints, accessories, bone grafts or substitutes, or particular adaptations therefor for insertion or extraction of endoprosthetic joints or of accessories thereof of spinal prostheses
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61F—FILTERS IMPLANTABLE INTO BLOOD VESSELS; PROSTHESES; DEVICES PROVIDING PATENCY TO, OR PREVENTING COLLAPSING OF, TUBULAR STRUCTURES OF THE BODY, e.g. STENTS; ORTHOPAEDIC, NURSING OR CONTRACEPTIVE DEVICES; FOMENTATION; TREATMENT OR PROTECTION OF EYES OR EARS; BANDAGES, DRESSINGS OR ABSORBENT PADS; FIRST-AID KITS
- A61F2/00—Filters implantable into blood vessels; Prostheses, i.e. artificial substitutes or replacements for parts of the body; Appliances for connecting them with the body; Devices providing patency to, or preventing collapsing of, tubular structures of the body, e.g. stents
- A61F2/02—Prostheses implantable into the body
- A61F2/30—Joints
- A61F2002/30001—Additional features of subject-matter classified in A61F2/28, A61F2/30 and subgroups thereof
- A61F2002/30003—Material related properties of the prosthesis or of a coating on the prosthesis
- A61F2002/3006—Properties of materials and coating materials
- A61F2002/30065—Properties of materials and coating materials thermoplastic, i.e. softening or fusing when heated, and hardening and becoming rigid again when cooled
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61F—FILTERS IMPLANTABLE INTO BLOOD VESSELS; PROSTHESES; DEVICES PROVIDING PATENCY TO, OR PREVENTING COLLAPSING OF, TUBULAR STRUCTURES OF THE BODY, e.g. STENTS; ORTHOPAEDIC, NURSING OR CONTRACEPTIVE DEVICES; FOMENTATION; TREATMENT OR PROTECTION OF EYES OR EARS; BANDAGES, DRESSINGS OR ABSORBENT PADS; FIRST-AID KITS
- A61F2/00—Filters implantable into blood vessels; Prostheses, i.e. artificial substitutes or replacements for parts of the body; Appliances for connecting them with the body; Devices providing patency to, or preventing collapsing of, tubular structures of the body, e.g. stents
- A61F2/02—Prostheses implantable into the body
- A61F2/30—Joints
- A61F2002/30001—Additional features of subject-matter classified in A61F2/28, A61F2/30 and subgroups thereof
- A61F2002/30003—Material related properties of the prosthesis or of a coating on the prosthesis
- A61F2002/3006—Properties of materials and coating materials
- A61F2002/3008—Properties of materials and coating materials radio-opaque, e.g. radio-opaque markers
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61F—FILTERS IMPLANTABLE INTO BLOOD VESSELS; PROSTHESES; DEVICES PROVIDING PATENCY TO, OR PREVENTING COLLAPSING OF, TUBULAR STRUCTURES OF THE BODY, e.g. STENTS; ORTHOPAEDIC, NURSING OR CONTRACEPTIVE DEVICES; FOMENTATION; TREATMENT OR PROTECTION OF EYES OR EARS; BANDAGES, DRESSINGS OR ABSORBENT PADS; FIRST-AID KITS
- A61F2/00—Filters implantable into blood vessels; Prostheses, i.e. artificial substitutes or replacements for parts of the body; Appliances for connecting them with the body; Devices providing patency to, or preventing collapsing of, tubular structures of the body, e.g. stents
- A61F2/02—Prostheses implantable into the body
- A61F2/30—Joints
- A61F2002/30001—Additional features of subject-matter classified in A61F2/28, A61F2/30 and subgroups thereof
- A61F2002/30316—The prosthesis having different structural features at different locations within the same prosthesis; Connections between prosthetic parts; Special structural features of bone or joint prostheses not otherwise provided for
- A61F2002/30329—Connections or couplings between prosthetic parts, e.g. between modular parts; Connecting elements
- A61F2002/30451—Connections or couplings between prosthetic parts, e.g. between modular parts; Connecting elements soldered or brazed or welded
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61F—FILTERS IMPLANTABLE INTO BLOOD VESSELS; PROSTHESES; DEVICES PROVIDING PATENCY TO, OR PREVENTING COLLAPSING OF, TUBULAR STRUCTURES OF THE BODY, e.g. STENTS; ORTHOPAEDIC, NURSING OR CONTRACEPTIVE DEVICES; FOMENTATION; TREATMENT OR PROTECTION OF EYES OR EARS; BANDAGES, DRESSINGS OR ABSORBENT PADS; FIRST-AID KITS
- A61F2/00—Filters implantable into blood vessels; Prostheses, i.e. artificial substitutes or replacements for parts of the body; Appliances for connecting them with the body; Devices providing patency to, or preventing collapsing of, tubular structures of the body, e.g. stents
- A61F2/02—Prostheses implantable into the body
- A61F2/30—Joints
- A61F2002/30001—Additional features of subject-matter classified in A61F2/28, A61F2/30 and subgroups thereof
- A61F2002/30316—The prosthesis having different structural features at different locations within the same prosthesis; Connections between prosthetic parts; Special structural features of bone or joint prostheses not otherwise provided for
- A61F2002/30535—Special structural features of bone or joint prostheses not otherwise provided for
- A61F2002/30537—Special structural features of bone or joint prostheses not otherwise provided for adjustable
- A61F2002/30538—Special structural features of bone or joint prostheses not otherwise provided for adjustable for adjusting angular orientation
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61F—FILTERS IMPLANTABLE INTO BLOOD VESSELS; PROSTHESES; DEVICES PROVIDING PATENCY TO, OR PREVENTING COLLAPSING OF, TUBULAR STRUCTURES OF THE BODY, e.g. STENTS; ORTHOPAEDIC, NURSING OR CONTRACEPTIVE DEVICES; FOMENTATION; TREATMENT OR PROTECTION OF EYES OR EARS; BANDAGES, DRESSINGS OR ABSORBENT PADS; FIRST-AID KITS
- A61F2/00—Filters implantable into blood vessels; Prostheses, i.e. artificial substitutes or replacements for parts of the body; Appliances for connecting them with the body; Devices providing patency to, or preventing collapsing of, tubular structures of the body, e.g. stents
- A61F2/02—Prostheses implantable into the body
- A61F2/30—Joints
- A61F2002/30001—Additional features of subject-matter classified in A61F2/28, A61F2/30 and subgroups thereof
- A61F2002/30316—The prosthesis having different structural features at different locations within the same prosthesis; Connections between prosthetic parts; Special structural features of bone or joint prostheses not otherwise provided for
- A61F2002/30535—Special structural features of bone or joint prostheses not otherwise provided for
- A61F2002/30537—Special structural features of bone or joint prostheses not otherwise provided for adjustable
- A61F2002/30545—Special structural features of bone or joint prostheses not otherwise provided for adjustable for adjusting a diameter
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61F—FILTERS IMPLANTABLE INTO BLOOD VESSELS; PROSTHESES; DEVICES PROVIDING PATENCY TO, OR PREVENTING COLLAPSING OF, TUBULAR STRUCTURES OF THE BODY, e.g. STENTS; ORTHOPAEDIC, NURSING OR CONTRACEPTIVE DEVICES; FOMENTATION; TREATMENT OR PROTECTION OF EYES OR EARS; BANDAGES, DRESSINGS OR ABSORBENT PADS; FIRST-AID KITS
- A61F2/00—Filters implantable into blood vessels; Prostheses, i.e. artificial substitutes or replacements for parts of the body; Appliances for connecting them with the body; Devices providing patency to, or preventing collapsing of, tubular structures of the body, e.g. stents
- A61F2/02—Prostheses implantable into the body
- A61F2/30—Joints
- A61F2002/30001—Additional features of subject-matter classified in A61F2/28, A61F2/30 and subgroups thereof
- A61F2002/30316—The prosthesis having different structural features at different locations within the same prosthesis; Connections between prosthetic parts; Special structural features of bone or joint prostheses not otherwise provided for
- A61F2002/30535—Special structural features of bone or joint prostheses not otherwise provided for
- A61F2002/30581—Special structural features of bone or joint prostheses not otherwise provided for having a pocket filled with fluid, e.g. liquid
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61F—FILTERS IMPLANTABLE INTO BLOOD VESSELS; PROSTHESES; DEVICES PROVIDING PATENCY TO, OR PREVENTING COLLAPSING OF, TUBULAR STRUCTURES OF THE BODY, e.g. STENTS; ORTHOPAEDIC, NURSING OR CONTRACEPTIVE DEVICES; FOMENTATION; TREATMENT OR PROTECTION OF EYES OR EARS; BANDAGES, DRESSINGS OR ABSORBENT PADS; FIRST-AID KITS
- A61F2/00—Filters implantable into blood vessels; Prostheses, i.e. artificial substitutes or replacements for parts of the body; Appliances for connecting them with the body; Devices providing patency to, or preventing collapsing of, tubular structures of the body, e.g. stents
- A61F2/02—Prostheses implantable into the body
- A61F2/30—Joints
- A61F2/46—Special tools or methods for implanting or extracting artificial joints, accessories, bone grafts or substitutes, or particular adaptations therefor
- A61F2002/4677—Special tools or methods for implanting or extracting artificial joints, accessories, bone grafts or substitutes, or particular adaptations therefor using a guide wire
Description
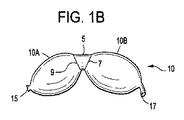
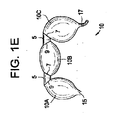
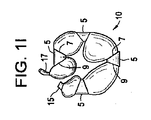
(1) 変性椎間板疾患、椎体骨欠損、及び脊髄運動セグメント不安定性の臨床結果を治療する複数容積部バルーンであって、直接的又は間接的にそれぞれが互いに接続された複数の単容積部を備え、穿孔部位が、前記単容積部の少なくとも2つの間に位置し、それぞれの単容積部は、(a)それぞれの単容積部が、可変容積の内容物を含むことができ、かつ(b)前記複数の単容積部のそれぞれが、別の単容積部に対して同じ又は異なる量及び形式の内容物を含むことができるように、個々に調節可能及び拡張可能である、複数容積部バルーン。
(2) 前記穿孔部位が、ガイドワイヤを含む、実施態様1に記載の複数容積部バルーン。
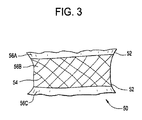
(3) 前記単容積部が、前記穿孔部位にて共に接合された少なくとも2つの箔片の隔室であり、前記接合が、超音波溶接、光源を使用する熱溶接、熱源を使用する熱溶接、圧接、スエージング、連結、接着及びスティッチングからなる群から選択される、実施態様1に記載の複数容積部バルーン。
(4) 前記材料が、箔、メッシュ、管、シリコーンゴム、エラストマゴム、ポリエーテルエーテルケトン(つまりPEEK)、ポリエーテルケトンケトン(つまりPEKK)、ポリエチレン(つまりPE)、ポリエチレンテレフタラート(つまりPET)、ポリウレタン(つまりPU)、熱可塑性ポリウレタン(つまりTPU)、及びポリカーボネートウレタン(つまりPCU)からなる群から選択される、実施態様1に記載の複数容積部バルーン。
(5) 変性椎間板疾患、椎体骨欠損、及び脊髄運動セグメント不安定性の臨床結果を治療するバルーンであって、バルーンを形成するために共に接合される、材料の少なくとも2つの部片を備え、前記材料が、マークを含み、前記マークが、少なくとも2つの隔室を前記バルーンにおいて形成するために共に接合されることができる、バルーン。
(7) 前記材料が、箔、メッシュ、布、管、シリコーンゴム、エラストマゴム、ポリエーテルエーテルケトン(つまりPEEK)、ポリエーテルケトンケトン(つまりPEKK)、ポリエチレン(つまりPE)、テレフタラート(つまりPET)、ポリウレタン(つまりPU)、熱可塑性ポリウレタン(つまりTPU)、及びポリカーボネートウレタン(つまりPCU)からなる群から選択される、実施態様5に記載のバルーン。
(8) 前記少なくとも2つの隔室は、(a)それぞれの隔室が、可変容積の内容物を含むことができ、かつ、(b)前記隔室のそれぞれが、別の隔室に対して同じ又は異なる容積の内容物を含むことができるように、個々に調節可能及び拡張可能である、実施態様5に記載のバルーン。
(9) 前記材料が、印刷された黒のマークを含む回路基板を含み、前記マークは、発熱光に晒されると共に接合される、実施態様5に記載の方法。
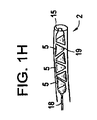
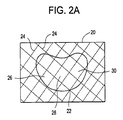
(10) 複数容積部バルーンを作製するために単容積部バルーンに角度をつける方法であって、
単容積部バルーンを提供することと、
少なくとも1つのリングを提供することであって、前記リングが、2つの端部を有し、前記リングの前記端部が、所定の角度に設定される、提供することと、
前記単容積部バルーンを前記リングに挿入することと、
前記単容積部バルーンを含む前記リングを脊柱の空洞に挿入することであって、前記空洞が、椎間板腔及び椎体における空間から成る群から選択される、挿入することと、
前記単容積部バルーンを拡張することと、を含み、前記リング内の前記単容積部バルーンの前記拡張が、複数容積部バルーンを形成し、前記リングの前記所定の角度が、前記複数容積部バルーンのそれぞれの容積部の前記角形成を決定する、方法。
(12) 前記リングを前記複数容積部バルーンに接合することを更に含み、前記接合が、超音波溶接、光源を使用する熱溶接、熱源を使用する熱溶接、圧接、スエージング、連結、接着及びスティッチングからなる群から選択される、実施態様10に記載の方法。
(13) 脊柱を整合及び安定させる方法であって、
バルーンカテーテルの遠位端に位置する複数容積部バルーンを含むバルーンカテーテルを脊柱の空洞に挿入することであって、前記空洞が、椎間板腔及び椎体における空間から成る群から選択される、挿入することと、
流体を挿入することによって前記複数容積部バルーンの第1の容積部を拡張することであって、前記第1の容積部の前記拡張が、前記脊柱までの高さを回復する、拡張することと、
凝固材料を挿入することによって前記複数容積部バルーンの第2の容積部を拡張することと、を含み、前記凝固材料が、前記第1の容積部の前記拡張によって創出された前記高さの回復を維持するために挿入の後に硬化する、方法。
(14) 前記流体が、水、食塩溶液、造影剤溶液、及びジェルからなる群から選択される、実施態様13に記載の方法。
(15) 前記凝固材料が、ポリマー、結晶性固体、エラストマ、及びガラスからなる群から選択される、実施態様13に記載の方法。
(17) 前記複数容積部バルーンの前記第2の容積部を膨張させた後、前記第1の容積部を除去するために前記穿孔部位に沿って引き裂くことと、
前記第1の容積部を前記脊柱の前記空洞から後退させて除去することと、
前記第1の容積部の前記拡張によって創出された前記高さの回復を維持するために前記硬化したポリマーを含む前記複数容積部バルーンの前記第2の容積部を保持することと、を更に含む、実施態様16に記載の方法。
(18) 前記第1の容積部及び第2の容積部が、異なる高さに拡張される、実施態様13に記載の方法。
(19) 前記第1の容積部の前記拡張が、また、クッション効果を提供するためであり、前記第1の容積部が、前記第2の容積部を上回る高さに拡張される、実施態様18に記載の方法。
(20) バルーンを患者の空洞に挿入する方法であって、
前記バルーンを管状形状へ丸めることと、
前記バルーンを前記バルーンの挿入前にきれいにされた前記患者の前記空洞に挿入することと、
一旦前記空洞の内側に置かれると前記バルーンを広げることと、を含む、方法。
(22) 前記バルーンが、ガイドワイヤ周りに管状形状に丸められる、実施態様20に記載の方法。
(23) 前記バルーンが、カバースリーブを介して前記空洞に挿入される、実施態様20に記載の方法。
(24) 前記空洞に挿入された後に前記バルーンを膨張させることを更に含み、前記バルーンを膨張させることが、前記空洞の適切な解剖学的構造を回復する、実施態様20に記載の方法。
(25) 骨を整合及び安定させるキットであって、
少なくとも1つの単容積部バルーンと、
少なくとも1つのリングと、を備え、それぞれのリングが、2つの端部を有し、前記リングの前記端部が、所定の角度に設定され、前記単容積部バルーンが、前記リングに挿入され、前記リングを含む前記単容積部バルーンを膨張させることが、複数容積部バルーンを作成し、前記リングの前記所定の角度が、前記複数容積部バルーンのそれぞれの容積部の前記角形成を決定する、キット。
Claims (4)
- 変性椎間板疾患、椎体骨欠損、及び脊髄運動セグメント不安定性を治療するための複数容積部バルーンであって、直接的又は間接的にそれぞれが互いに接続された複数の単容積部とガイドワイヤとを備え、穿孔部位が、前記単容積部の少なくとも2つの間に位置し、それぞれの前記単容積部は、(a)それぞれの前記単容積部が、可変容積の内容物を含むことができ、かつ(b)前記複数の単容積部のそれぞれが、別の前記単容積部に対して同じ又は異なる量及び形式の内容物を含むことができるように、個々に調節可能及び拡張可能であり、前記穿孔部位が、前記複数容積部バルーンを患者の体内に案内するための前記ガイドワイヤを含み、前記ガイドワイヤは前記穿孔部位を引き裂くことにより除去され得る、複数容積部バルーン。
- 前記単容積部が、前記穿孔部位にて共に接合された少なくとも2つの箔片の隔室である、請求項1に記載の複数容積部バルーン。
- 前記複数容積部バルーンが、箔材料またはメッシュ材料で形成されている、請求項1に記載の複数容積部バルーン。
- 前記複数容積部バルーンが、シリコーンゴム、エラストマゴム、ポリエーテルエーテルケトン(つまりPEEK)、ポリエーテルケトンケトン(つまりPEKK)、ポリエチレン(つまりPE)、ポリエチレンテレフタラート(つまりPET)、ポリウレタン(つまりPU)、熱可塑性ポリウレタン(つまりTPU)、及びポリカーボネートウレタン(つまりPCU)からなる群から選択される材料で作製されている、請求項1に記載の複数容積部バルーン。
Applications Claiming Priority (3)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| US13/804,992 US9585761B2 (en) | 2013-03-14 | 2013-03-14 | Angulated rings and bonded foils for use with balloons for fusion and dynamic stabilization |
| US13/804,992 | 2013-03-14 | ||
| PCT/US2014/021266 WO2014158982A1 (en) | 2013-03-14 | 2014-03-06 | Angulated rings and bonded foils for use with balloons for fusion and dynamic stabilization |
Publications (3)
| Publication Number | Publication Date |
|---|---|
| JP2016516470A JP2016516470A (ja) | 2016-06-09 |
| JP2016516470A5 JP2016516470A5 (ja) | 2019-01-10 |
| JP6526623B2 true JP6526623B2 (ja) | 2019-06-05 |
Family
ID=50489379
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| JP2016500744A Expired - Fee Related JP6526623B2 (ja) | 2013-03-14 | 2014-03-06 | 固定及び動的安定化のためにバルーンと共に使用される角張ったリング及び接合箔 |
Country Status (7)
| Country | Link |
|---|---|
| US (4) | US9585761B2 (ja) |
| EP (1) | EP2967898B1 (ja) |
| JP (1) | JP6526623B2 (ja) |
| CN (1) | CN105163689A (ja) |
| AU (1) | AU2014241511B2 (ja) |
| ES (1) | ES2646215T3 (ja) |
| WO (1) | WO2014158982A1 (ja) |
Families Citing this family (18)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| DE10154163A1 (de) * | 2001-11-03 | 2003-05-22 | Advanced Med Tech | Vorrichtung zum Aufrichten und Stabilisieren der Wirbelsäule |
| AU2008224435B2 (en) | 2007-03-15 | 2014-01-09 | Ortho-Space Ltd. | Prosthetic devices and methods for using same |
| US9839461B2 (en) * | 2011-08-18 | 2017-12-12 | Matthias Militz | Expansion device for bone expansion and medical device for bone expansion |
| WO2013057566A2 (en) | 2011-10-18 | 2013-04-25 | Ortho-Space Ltd. | Prosthetic devices and methods for using same |
| US20130282121A1 (en) * | 2012-03-22 | 2013-10-24 | Ann Prewett | Spinal facet augmentation implant and method |
| US9192420B2 (en) * | 2013-01-24 | 2015-11-24 | Kyphon Sarl | Surgical system and methods of use |
| US9358120B2 (en) | 2013-03-14 | 2016-06-07 | DePuy Synthes Products, Inc. | Expandable coil spinal implant |
| US9572676B2 (en) | 2013-03-14 | 2017-02-21 | DePuy Synthes Products, Inc. | Adjustable multi-volume balloon for spinal interventions |
| US9585761B2 (en) | 2013-03-14 | 2017-03-07 | DePuy Synthes Products, Inc. | Angulated rings and bonded foils for use with balloons for fusion and dynamic stabilization |
| WO2017046647A1 (en) | 2015-09-18 | 2017-03-23 | Ortho-Space Ltd. | Intramedullary fixated subacromial spacers |
| ITUA20162676A1 (it) | 2016-04-18 | 2017-10-18 | Alessandro Crippa | Dispositivo di ausilio alla respirazione e procedimento di fabbricazione del dispositivo |
| EP3573806A4 (en) | 2017-01-30 | 2019-12-11 | Ortho-Space Ltd. | MACHINING MACHINE AND METHOD FOR PROCESSING DIVING MADE ARTICLES |
| BR112020013107A2 (pt) * | 2017-12-28 | 2020-11-24 | Cardio Voyage Innovations, Llc | dispositivo de cifoplastia direcional e método de usar esse dispositivo |
| CN110974489B (zh) * | 2019-10-11 | 2022-01-25 | 杭州锐健马斯汀医疗器材有限公司 | 减轻人体组织与其他组织损伤的异形球囊及其制备方法 |
| CN110613534B (zh) * | 2019-10-11 | 2021-11-12 | 杭州锐健马斯汀医疗器材有限公司 | 减轻人体软组织损伤的可撕拉异形球囊及其制备方法 |
| US10821002B1 (en) * | 2019-12-10 | 2020-11-03 | Spica Medical Technologies, Llc | Inflatable spinal implants and related systems and methods |
| US11376131B2 (en) * | 2020-04-07 | 2022-07-05 | Ethicon, Inc. | Cortical rim-supporting interbody device and method |
| CN114652490B (zh) * | 2022-05-20 | 2022-09-02 | 茂名市人民医院 | 一种可折叠人工半月板 |
Family Cites Families (91)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US3012197A (en) | 1958-10-27 | 1961-12-05 | Gen Radio Co | Calibration apparatus |
| US3007535A (en) | 1960-10-31 | 1961-11-07 | Deere & Co | Implement attaching means |
| US3211152A (en) | 1961-11-15 | 1965-10-12 | Ernest A Stern | Safety endotracheal tube cuff |
| US4077394A (en) | 1976-08-25 | 1978-03-07 | Mccurdy Martin D | Integral pressure sensor probe for a cardiac assistance device |
| US4512766A (en) | 1982-12-08 | 1985-04-23 | Whitman Medical Corporation | Catheter valve |
| US4856510A (en) | 1988-04-06 | 1989-08-15 | Kowalewski Ryszard J | Tracheal tube inflator |
| US5772661A (en) | 1988-06-13 | 1998-06-30 | Michelson; Gary Karlin | Methods and instrumentation for the surgical correction of human thoracic and lumbar spinal disease from the antero-lateral aspect of the spine |
| JP2514087Y2 (ja) | 1990-05-25 | 1996-10-16 | 幸三 牧田 | 離脱式両端逆止弁付きバル―ン |
| DE4128332A1 (de) | 1991-08-27 | 1993-03-04 | Man Ceramics Gmbh | Wirbelknochenersatz |
| US5423850A (en) | 1993-10-01 | 1995-06-13 | Berger; J. Lee | Balloon compressor for internal fixation of bone fractures |
| US20030032963A1 (en) | 2001-10-24 | 2003-02-13 | Kyphon Inc. | Devices and methods using an expandable body with internal restraint for compressing cancellous bone |
| JP3333211B2 (ja) | 1994-01-26 | 2002-10-15 | レイリー,マーク・エイ | 骨の治療に関する外科的方法で使用される、改良に係る拡張可能な装置 |
| WO2003059213A2 (en) | 1994-01-26 | 2003-07-24 | Kyphon Inc. | Inflatable device for use in surgical protocol relating to fixation of bone |
| DE69534156T2 (de) | 1994-01-26 | 2005-09-29 | Kyphon Inc., Sunnyvale | Verbesserte aufblasbare Einrichtung für eine Verwendung in chirurgischen Protokollen in Bezug auf die Fixierung von Knochen |
| DE59511075D1 (de) | 1995-11-08 | 2007-02-08 | Zimmer Gmbh | Vorrichtung zum Einbringen eines Implantats, insbesondere einer Zwischenwirbelprothese |
| US7618451B2 (en) * | 2001-05-25 | 2009-11-17 | Conformis, Inc. | Patient selectable joint arthroplasty devices and surgical tools facilitating increased accuracy, speed and simplicity in performing total and partial joint arthroplasty |
| JP2001527437A (ja) * | 1997-03-07 | 2001-12-25 | ベイヤー、モルデキイ | 経皮的骨および脊椎安定化、固定および修復のためのシステム |
| WO1999062416A1 (en) | 1998-06-01 | 1999-12-09 | Kyphon Inc. | Expandable preformed structures for deployment in interior body regions |
| US6187000B1 (en) | 1998-08-20 | 2001-02-13 | Endius Incorporated | Cannula for receiving surgical instruments |
| US6228022B1 (en) | 1998-10-28 | 2001-05-08 | Sdgi Holdings, Inc. | Methods and instruments for spinal surgery |
| US6582451B1 (en) | 1999-03-16 | 2003-06-24 | The University Of Sydney | Device for use in surgery |
| US20020169473A1 (en) | 1999-06-02 | 2002-11-14 | Concentric Medical, Inc. | Devices and methods for treating vascular malformations |
| AU2001233098A1 (en) | 2000-01-27 | 2001-08-07 | Sterilis, Inc. | Cavity enlarger method and apparatus |
| US6468200B1 (en) | 2000-03-06 | 2002-10-22 | Michael C. Fischi | Segmented peristaltic intra-aortic balloon pump |
| US6679886B2 (en) | 2000-09-01 | 2004-01-20 | Synthes (Usa) | Tools and methods for creating cavities in bone |
| WO2002056802A1 (en) | 2000-12-15 | 2002-07-25 | Spineology, Inc. | Annulus-reinforcing band |
| US6632235B2 (en) | 2001-04-19 | 2003-10-14 | Synthes (U.S.A.) | Inflatable device and method for reducing fractures in bone and in treating the spine |
| US20090234457A1 (en) | 2001-06-29 | 2009-09-17 | The Regents Of The University Of California | Systems, devices and methods for treatment of intervertebral disorders |
| US20030045923A1 (en) | 2001-08-31 | 2003-03-06 | Mehran Bashiri | Hybrid balloon expandable/self expanding stent |
| DE60232893D1 (de) | 2001-10-02 | 2009-08-20 | Rex Medical Lp | Wirbelimplantat |
| US7074226B2 (en) | 2002-09-19 | 2006-07-11 | Sdgi Holdings, Inc. | Oval dilator and retractor set and method |
| MXPA05008653A (es) | 2003-02-14 | 2006-04-27 | Depuy Spine Inc | Dispositivo de fusion intervertebral formado in situ. |
| US7655010B2 (en) | 2003-09-30 | 2010-02-02 | Depuy Spine, Inc. | Vertebral fusion device and method for using same |
| US20050278027A1 (en) | 2004-06-11 | 2005-12-15 | Hyde Edward R Jr | Annulus fibrosus stent |
| US20050288766A1 (en) | 2004-06-28 | 2005-12-29 | Xtent, Inc. | Devices and methods for controlling expandable prostheses during deployment |
| US7503920B2 (en) | 2004-08-11 | 2009-03-17 | Tzony Siegal | Spinal surgery system and method |
| US8236029B2 (en) | 2004-08-11 | 2012-08-07 | Nlt Spine Ltd. | Devices for introduction into a body via a substantially straight conduit to for a predefined curved configuration, and methods employing such devices |
| EP1833379B1 (en) | 2005-01-07 | 2016-08-24 | Stryker European Holdings I, LLC | Three-prong retractor with elastomeric sheath |
| US7643884B2 (en) | 2005-01-31 | 2010-01-05 | Warsaw Orthopedic, Inc. | Electrically insulated surgical needle assembly |
| WO2006092015A1 (en) | 2005-03-01 | 2006-09-08 | Columna Pty Ltd | Intervertebral disc restoration |
| US7141727B1 (en) | 2005-05-16 | 2006-11-28 | International Business Machines Corporation | Method and apparatus for fabricating a carbon nanotube transistor having unipolar characteristics |
| WO2006130796A2 (en) | 2005-06-02 | 2006-12-07 | Zimmer Spine, Inc. | Interbody fusion ring and method of using the same |
| CA2607914C (en) * | 2005-06-15 | 2010-08-03 | Pong-Jeu Lu | Orthopaedic paste delivering tool and method for continually delivering the paste, and devices and methods for facilitating the delivery of the orthopaedic paste |
| DE102005034529A1 (de) | 2005-07-23 | 2007-01-25 | Qualimed Innovative Medizinprodukte Gmbh | Ballondilatationskatheter |
| CN103272325B (zh) | 2005-08-11 | 2016-04-06 | 泰克尼恩研究和发展基金有限公司 | 用于在通道内运动的顶端推进装置 |
| US8591583B2 (en) | 2005-08-16 | 2013-11-26 | Benvenue Medical, Inc. | Devices for treating the spine |
| US8366773B2 (en) | 2005-08-16 | 2013-02-05 | Benvenue Medical, Inc. | Apparatus and method for treating bone |
| EP1924227B1 (en) | 2005-08-16 | 2014-12-17 | Benvenue Medical, Inc. | Spinal tissue distraction devices |
| US7699894B2 (en) | 2005-12-22 | 2010-04-20 | Depuy Spine, Inc. | Nucleus pulposus trial device and technique |
| US20070162132A1 (en) | 2005-12-23 | 2007-07-12 | Dominique Messerli | Flexible elongated chain implant and method of supporting body tissue with same |
| US20070191956A1 (en) | 2006-02-10 | 2007-08-16 | Replication Medical, Inc. | Radially extended support member for spinal nucleus implants and methods of use |
| US20070255406A1 (en) | 2006-04-27 | 2007-11-01 | Sdgi Holdings, Inc. | Devices, apparatus, and methods for bilateral approach to disc augmentation |
| US20090299476A1 (en) * | 2006-05-19 | 2009-12-03 | Ashish Diwan | Tissue prosthesis |
| US8236057B2 (en) | 2006-06-12 | 2012-08-07 | Globus Medical, Inc. | Inflatable multi-chambered devices and methods of treatment using the same |
| US7740659B2 (en) | 2006-06-29 | 2010-06-22 | Depuy Spine, Inc. | Insert for nucleus implant |
| EP2083760B1 (en) | 2006-10-16 | 2014-01-15 | Pioneer Surgical Technology, Inc. | FUSION DEVICE and SYSTEMS |
| EP2086472B1 (en) | 2006-11-16 | 2019-06-12 | Rex Medical, L.P. | Spinal implant |
| US7947078B2 (en) | 2007-01-09 | 2011-05-24 | Nonlinear Technologies Ltd. | Devices for forming curved or closed-loop structures |
| EP2124778B1 (en) | 2007-02-21 | 2019-09-25 | Benvenue Medical, Inc. | Devices for treating the spine |
| US20110092859A1 (en) | 2007-06-25 | 2011-04-21 | Neubardt Seth L | System for determining and placing spinal implants or prostheses |
| ATE527950T1 (de) | 2007-06-29 | 2011-10-15 | Synthes Gmbh | Flexible kettenimplantate und instrumente |
| US7922767B2 (en) | 2007-07-07 | 2011-04-12 | Jmea Corporation | Disk fusion implant |
| US20110054408A1 (en) * | 2007-07-10 | 2011-03-03 | Guobao Wei | Delivery systems, devices, tools, and methods of use |
| ES2374760T3 (es) | 2007-08-09 | 2012-02-21 | Nonlinear Technologies Ltd. | Dispositivo para la distracción de apofisis espinosas. |
| US7959684B2 (en) * | 2007-09-11 | 2011-06-14 | Joy Medical Devices Corporation | Method for forming a bioresorbable composite implant in a bone |
| US20090088788A1 (en) | 2007-09-28 | 2009-04-02 | Steven Mouw | Methods and apparatus having multiple separately actuatable expandable members |
| US20090118833A1 (en) | 2007-11-05 | 2009-05-07 | Zimmer Spine, Inc. | In-situ curable interspinous process spacer |
| US20090222096A1 (en) | 2008-02-28 | 2009-09-03 | Warsaw Orthopedic, Inc. | Multi-compartment expandable devices and methods for intervertebral disc expansion and augmentation |
| US20090234454A1 (en) | 2008-03-12 | 2009-09-17 | Nonlinear Technologies Ltd. | Spinal Implant With Structural Support And Bone Interface Surfaces Formed from UHMWPE |
| US8147555B2 (en) | 2008-03-31 | 2012-04-03 | Aflatoon Kamran | Artificial disc prosthesis for replacing a damaged nucleus |
| US8328812B2 (en) | 2008-07-27 | 2012-12-11 | NLT-Spine Ltd. | Tool and corresponding method for removal of material from within a body |
| CN102046102B (zh) | 2008-07-27 | 2013-03-13 | 非线性技术有限公司 | 用于从椎间盘体内移除材料的工具与相应方法 |
| US8540667B2 (en) | 2008-11-12 | 2013-09-24 | Sanovas, Inc. | Multi-balloon catheter for extravasated drug delivery |
| US8128699B2 (en) | 2009-03-13 | 2012-03-06 | Warsaw Orthopedic, Inc. | Spinal implant and methods of implantation and treatment |
| US8535327B2 (en) | 2009-03-17 | 2013-09-17 | Benvenue Medical, Inc. | Delivery apparatus for use with implantable medical devices |
| WO2010118021A1 (en) | 2009-04-09 | 2010-10-14 | Synthes Usa, Llc | Minimally invasive spine augmentation and stabilization system and method |
| JP5636051B2 (ja) | 2009-08-19 | 2014-12-03 | ジンテス ゲゼルシャフト ミット ベシュレンクテル ハフツング | 骨を増強する方法及び装置 |
| US9615933B2 (en) | 2009-09-15 | 2017-04-11 | DePuy Synthes Products, Inc. | Expandable ring intervertebral fusion device |
| KR20120100930A (ko) | 2009-11-06 | 2012-09-12 | 신세스 게엠바하 | 최소 침습 극돌기간 스페이서 임플란트 및 방법 |
| US9039769B2 (en) | 2010-03-17 | 2015-05-26 | Globus Medical, Inc. | Intervertebral nucleus and annulus implants and method of use thereof |
| WO2011123439A2 (en) | 2010-03-31 | 2011-10-06 | Kitchen Michael S | Intervertebral spacer and methods of use |
| WO2012054240A1 (en) | 2010-10-18 | 2012-04-26 | Synthes Usa, Llc | Implant |
| US9498365B2 (en) | 2010-10-19 | 2016-11-22 | Apollo Endosurgery, Inc. | Intragastric implants with multiple fluid chambers |
| US8597239B2 (en) | 2011-03-01 | 2013-12-03 | Sanovas, Inc. | Abrading balloon catheter for extravasated drug delivery |
| US9180281B2 (en) | 2011-04-08 | 2015-11-10 | Sanovas, Inc. | Adjustable balloon catheter for extravasated drug delivery |
| US20120259216A1 (en) | 2011-04-08 | 2012-10-11 | Gerrans Lawrence J | Balloon catheter with drug delivery probe |
| US9839461B2 (en) | 2011-08-18 | 2017-12-12 | Matthias Militz | Expansion device for bone expansion and medical device for bone expansion |
| US9585761B2 (en) | 2013-03-14 | 2017-03-07 | DePuy Synthes Products, Inc. | Angulated rings and bonded foils for use with balloons for fusion and dynamic stabilization |
| US9572676B2 (en) | 2013-03-14 | 2017-02-21 | DePuy Synthes Products, Inc. | Adjustable multi-volume balloon for spinal interventions |
| US9480574B2 (en) | 2013-03-14 | 2016-11-01 | Benvenue Medical, Inc. | Spinal fusion implants and devices and methods for deploying such implants |
| US9358120B2 (en) | 2013-03-14 | 2016-06-07 | DePuy Synthes Products, Inc. | Expandable coil spinal implant |
-
2013
- 2013-03-14 US US13/804,992 patent/US9585761B2/en active Active
-
2014
- 2014-03-06 EP EP14717897.4A patent/EP2967898B1/en not_active Not-in-force
- 2014-03-06 WO PCT/US2014/021266 patent/WO2014158982A1/en active Application Filing
- 2014-03-06 CN CN201480015307.7A patent/CN105163689A/zh active Pending
- 2014-03-06 ES ES14717897.4T patent/ES2646215T3/es active Active
- 2014-03-06 JP JP2016500744A patent/JP6526623B2/ja not_active Expired - Fee Related
- 2014-03-06 AU AU2014241511A patent/AU2014241511B2/en not_active Ceased
-
2017
- 2017-01-17 US US15/408,148 patent/US10143561B2/en active Active
-
2018
- 2018-10-19 US US16/165,692 patent/US10828170B2/en active Active
-
2020
- 2020-09-30 US US17/060,072 patent/US11589999B2/en active Active
Also Published As
| Publication number | Publication date |
|---|---|
| AU2014241511A1 (en) | 2015-10-29 |
| EP2967898B1 (en) | 2017-09-06 |
| US20210015623A1 (en) | 2021-01-21 |
| US9585761B2 (en) | 2017-03-07 |
| ES2646215T3 (es) | 2017-12-12 |
| US20140277465A1 (en) | 2014-09-18 |
| CN105163689A (zh) | 2015-12-16 |
| EP2967898A1 (en) | 2016-01-20 |
| US10143561B2 (en) | 2018-12-04 |
| JP2016516470A (ja) | 2016-06-09 |
| AU2014241511B2 (en) | 2018-10-04 |
| US20190151107A1 (en) | 2019-05-23 |
| WO2014158982A1 (en) | 2014-10-02 |
| US11589999B2 (en) | 2023-02-28 |
| US20170119535A1 (en) | 2017-05-04 |
| US10828170B2 (en) | 2020-11-10 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| JP6526623B2 (ja) | 固定及び動的安定化のためにバルーンと共に使用される角張ったリング及び接合箔 | |
| EP2967695B1 (en) | Adjustable multi-volume balloon for spinal interventions | |
| JP4436045B2 (ja) | 骨のならびに脊椎治療における破損を整復するための可膨張性装置および方法 | |
| US8372115B2 (en) | Bone support device, system and method | |
| AU2010236479B2 (en) | Minimally invasive expandable contained vertebral implant and method | |
| EP2419052B1 (en) | Minimally invasive expandable vertebral implant | |
| JP2012523898A (ja) | 椎体終板を連結する植込み片およびその方法 | |
| AU2002258804A1 (en) | Inflatable device and method for reducing fractures in bone and in treating the spine | |
| US8226722B2 (en) | Sac for use in spinal surgery | |
| KR20080085089A (ko) | 해면 뼈 압밀 장치 |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| A621 | Written request for application examination |
Free format text: JAPANESE INTERMEDIATE CODE: A621 Effective date: 20170302 |
|
| A131 | Notification of reasons for refusal |
Free format text: JAPANESE INTERMEDIATE CODE: A131 Effective date: 20171205 |
|
| A521 | Request for written amendment filed |
Free format text: JAPANESE INTERMEDIATE CODE: A523 Effective date: 20180301 |
|
| A131 | Notification of reasons for refusal |
Free format text: JAPANESE INTERMEDIATE CODE: A131 Effective date: 20180828 |
|
| A521 | Request for written amendment filed |
Free format text: JAPANESE INTERMEDIATE CODE: A523 Effective date: 20181122 |
|
| A524 | Written submission of copy of amendment under article 19 pct |
Free format text: JAPANESE INTERMEDIATE CODE: A524 Effective date: 20181122 |
|
| TRDD | Decision of grant or rejection written | ||
| A01 | Written decision to grant a patent or to grant a registration (utility model) |
Free format text: JAPANESE INTERMEDIATE CODE: A01 Effective date: 20190423 |
|
| A61 | First payment of annual fees (during grant procedure) |
Free format text: JAPANESE INTERMEDIATE CODE: A61 Effective date: 20190508 |
|
| R150 | Certificate of patent or registration of utility model |
Ref document number: 6526623 Country of ref document: JP Free format text: JAPANESE INTERMEDIATE CODE: R150 |
|
| R250 | Receipt of annual fees |
Free format text: JAPANESE INTERMEDIATE CODE: R250 |
|
| LAPS | Cancellation because of no payment of annual fees |