JP6055083B2 - 血管内ロータリ血液ポンプ - Google Patents
血管内ロータリ血液ポンプ Download PDFInfo
- Publication number
- JP6055083B2 JP6055083B2 JP2015507537A JP2015507537A JP6055083B2 JP 6055083 B2 JP6055083 B2 JP 6055083B2 JP 2015507537 A JP2015507537 A JP 2015507537A JP 2015507537 A JP2015507537 A JP 2015507537A JP 6055083 B2 JP6055083 B2 JP 6055083B2
- Authority
- JP
- Japan
- Prior art keywords
- blood pump
- pressure
- pumping device
- pressure sensor
- blood
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Active
Links
- 239000008280 blood Substances 0.000 title claims description 65
- 210000004369 blood Anatomy 0.000 title claims description 65
- 238000005086 pumping Methods 0.000 claims description 63
- 239000013307 optical fiber Substances 0.000 claims description 33
- 230000017531 blood circulation Effects 0.000 claims description 15
- 230000003287 optical effect Effects 0.000 claims description 13
- 239000011521 glass Substances 0.000 claims description 9
- 239000012528 membrane Substances 0.000 claims description 9
- 239000000835 fiber Substances 0.000 claims description 6
- 239000000919 ceramic Substances 0.000 claims description 5
- 239000007767 bonding agent Substances 0.000 claims description 3
- 239000011324 bead Substances 0.000 claims description 2
- 239000010408 film Substances 0.000 description 18
- 238000009530 blood pressure measurement Methods 0.000 description 9
- 238000011156 evaluation Methods 0.000 description 7
- 210000000709 aorta Anatomy 0.000 description 6
- 210000001765 aortic valve Anatomy 0.000 description 5
- 210000002318 cardia Anatomy 0.000 description 5
- 238000005259 measurement Methods 0.000 description 5
- 238000001361 intraarterial administration Methods 0.000 description 4
- 238000011084 recovery Methods 0.000 description 4
- 210000005166 vasculature Anatomy 0.000 description 4
- 210000002376 aorta thoracic Anatomy 0.000 description 3
- 239000011248 coating agent Substances 0.000 description 3
- 238000000576 coating method Methods 0.000 description 3
- 230000001419 dependent effect Effects 0.000 description 3
- 230000000694 effects Effects 0.000 description 3
- 230000006870 function Effects 0.000 description 3
- 210000005240 left ventricle Anatomy 0.000 description 3
- 239000007788 liquid Substances 0.000 description 3
- 229910001000 nickel titanium Inorganic materials 0.000 description 3
- HLXZNVUGXRDIFK-UHFFFAOYSA-N nickel titanium Chemical compound [Ti].[Ti].[Ti].[Ti].[Ti].[Ti].[Ti].[Ti].[Ti].[Ti].[Ti].[Ni].[Ni].[Ni].[Ni].[Ni].[Ni].[Ni].[Ni].[Ni].[Ni].[Ni].[Ni].[Ni].[Ni] HLXZNVUGXRDIFK-UHFFFAOYSA-N 0.000 description 3
- 229920000642 polymer Polymers 0.000 description 3
- 238000005452 bending Methods 0.000 description 2
- 210000005242 cardiac chamber Anatomy 0.000 description 2
- 230000023597 hemostasis Effects 0.000 description 2
- 238000007914 intraventricular administration Methods 0.000 description 2
- 239000004642 Polyimide Substances 0.000 description 1
- 229910004298 SiO 2 Inorganic materials 0.000 description 1
- 230000001174 ascending effect Effects 0.000 description 1
- 230000009286 beneficial effect Effects 0.000 description 1
- 230000005540 biological transmission Effects 0.000 description 1
- 230000036772 blood pressure Effects 0.000 description 1
- 230000000747 cardiac effect Effects 0.000 description 1
- 230000006835 compression Effects 0.000 description 1
- 238000007906 compression Methods 0.000 description 1
- 230000003205 diastolic effect Effects 0.000 description 1
- 210000001105 femoral artery Anatomy 0.000 description 1
- 239000012530 fluid Substances 0.000 description 1
- 238000005194 fractionation Methods 0.000 description 1
- 239000003365 glass fiber Substances 0.000 description 1
- 230000002439 hemostatic effect Effects 0.000 description 1
- 238000003780 insertion Methods 0.000 description 1
- 230000037431 insertion Effects 0.000 description 1
- 238000002955 isolation Methods 0.000 description 1
- 239000000463 material Substances 0.000 description 1
- 230000010355 oscillation Effects 0.000 description 1
- 239000004033 plastic Substances 0.000 description 1
- 229920003023 plastic Polymers 0.000 description 1
- 229920003223 poly(pyromellitimide-1,4-diphenyl ether) Polymers 0.000 description 1
- 229920001721 polyimide Polymers 0.000 description 1
- 229920001296 polysiloxane Polymers 0.000 description 1
- 229920002635 polyurethane Polymers 0.000 description 1
- 239000004814 polyurethane Substances 0.000 description 1
- 238000010926 purge Methods 0.000 description 1
- 230000004044 response Effects 0.000 description 1
- 239000004065 semiconductor Substances 0.000 description 1
- 229910001285 shape-memory alloy Inorganic materials 0.000 description 1
- 229910052710 silicon Inorganic materials 0.000 description 1
- 239000010703 silicon Substances 0.000 description 1
- 239000010409 thin film Substances 0.000 description 1
- 230000002792 vascular Effects 0.000 description 1
Images
Classifications
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61B—DIAGNOSIS; SURGERY; IDENTIFICATION
- A61B5/00—Measuring for diagnostic purposes; Identification of persons
- A61B5/02—Detecting, measuring or recording pulse, heart rate, blood pressure or blood flow; Combined pulse/heart-rate/blood pressure determination; Evaluating a cardiovascular condition not otherwise provided for, e.g. using combinations of techniques provided for in this group with electrocardiography or electroauscultation; Heart catheters for measuring blood pressure
- A61B5/021—Measuring pressure in heart or blood vessels
- A61B5/0215—Measuring pressure in heart or blood vessels by means inserted into the body
- A61B5/02154—Measuring pressure in heart or blood vessels by means inserted into the body by optical transmission
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61M—DEVICES FOR INTRODUCING MEDIA INTO, OR ONTO, THE BODY; DEVICES FOR TRANSDUCING BODY MEDIA OR FOR TAKING MEDIA FROM THE BODY; DEVICES FOR PRODUCING OR ENDING SLEEP OR STUPOR
- A61M60/00—Blood pumps; Devices for mechanical circulatory actuation; Balloon pumps for circulatory assistance
- A61M60/10—Location thereof with respect to the patient's body
- A61M60/122—Implantable pumps or pumping devices, i.e. the blood being pumped inside the patient's body
- A61M60/126—Implantable pumps or pumping devices, i.e. the blood being pumped inside the patient's body implantable via, into, inside, in line, branching on, or around a blood vessel
- A61M60/13—Implantable pumps or pumping devices, i.e. the blood being pumped inside the patient's body implantable via, into, inside, in line, branching on, or around a blood vessel by means of a catheter allowing explantation, e.g. catheter pumps temporarily introduced via the vascular system
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61M—DEVICES FOR INTRODUCING MEDIA INTO, OR ONTO, THE BODY; DEVICES FOR TRANSDUCING BODY MEDIA OR FOR TAKING MEDIA FROM THE BODY; DEVICES FOR PRODUCING OR ENDING SLEEP OR STUPOR
- A61M60/00—Blood pumps; Devices for mechanical circulatory actuation; Balloon pumps for circulatory assistance
- A61M60/10—Location thereof with respect to the patient's body
- A61M60/122—Implantable pumps or pumping devices, i.e. the blood being pumped inside the patient's body
- A61M60/165—Implantable pumps or pumping devices, i.e. the blood being pumped inside the patient's body implantable in, on, or around the heart
- A61M60/17—Implantable pumps or pumping devices, i.e. the blood being pumped inside the patient's body implantable in, on, or around the heart inside a ventricle, e.g. intraventricular balloon pumps
- A61M60/174—Implantable pumps or pumping devices, i.e. the blood being pumped inside the patient's body implantable in, on, or around the heart inside a ventricle, e.g. intraventricular balloon pumps discharging the blood to the ventricle or arterial system via a cannula internal to the ventricle or arterial system
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61M—DEVICES FOR INTRODUCING MEDIA INTO, OR ONTO, THE BODY; DEVICES FOR TRANSDUCING BODY MEDIA OR FOR TAKING MEDIA FROM THE BODY; DEVICES FOR PRODUCING OR ENDING SLEEP OR STUPOR
- A61M60/00—Blood pumps; Devices for mechanical circulatory actuation; Balloon pumps for circulatory assistance
- A61M60/20—Type thereof
- A61M60/205—Non-positive displacement blood pumps
- A61M60/216—Non-positive displacement blood pumps including a rotating member acting on the blood, e.g. impeller
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61M—DEVICES FOR INTRODUCING MEDIA INTO, OR ONTO, THE BODY; DEVICES FOR TRANSDUCING BODY MEDIA OR FOR TAKING MEDIA FROM THE BODY; DEVICES FOR PRODUCING OR ENDING SLEEP OR STUPOR
- A61M60/00—Blood pumps; Devices for mechanical circulatory actuation; Balloon pumps for circulatory assistance
- A61M60/20—Type thereof
- A61M60/205—Non-positive displacement blood pumps
- A61M60/216—Non-positive displacement blood pumps including a rotating member acting on the blood, e.g. impeller
- A61M60/237—Non-positive displacement blood pumps including a rotating member acting on the blood, e.g. impeller the blood flow through the rotating member having mainly axial components, e.g. axial flow pumps
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61M—DEVICES FOR INTRODUCING MEDIA INTO, OR ONTO, THE BODY; DEVICES FOR TRANSDUCING BODY MEDIA OR FOR TAKING MEDIA FROM THE BODY; DEVICES FOR PRODUCING OR ENDING SLEEP OR STUPOR
- A61M60/00—Blood pumps; Devices for mechanical circulatory actuation; Balloon pumps for circulatory assistance
- A61M60/40—Details relating to driving
- A61M60/403—Details relating to driving for non-positive displacement blood pumps
- A61M60/422—Details relating to driving for non-positive displacement blood pumps the force acting on the blood contacting member being electromagnetic, e.g. using canned motor pumps
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61M—DEVICES FOR INTRODUCING MEDIA INTO, OR ONTO, THE BODY; DEVICES FOR TRANSDUCING BODY MEDIA OR FOR TAKING MEDIA FROM THE BODY; DEVICES FOR PRODUCING OR ENDING SLEEP OR STUPOR
- A61M60/00—Blood pumps; Devices for mechanical circulatory actuation; Balloon pumps for circulatory assistance
- A61M60/50—Details relating to control
- A61M60/508—Electronic control means, e.g. for feedback regulation
- A61M60/515—Regulation using real-time patient data
- A61M60/531—Regulation using real-time patient data using blood pressure data, e.g. from blood pressure sensors
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61M—DEVICES FOR INTRODUCING MEDIA INTO, OR ONTO, THE BODY; DEVICES FOR TRANSDUCING BODY MEDIA OR FOR TAKING MEDIA FROM THE BODY; DEVICES FOR PRODUCING OR ENDING SLEEP OR STUPOR
- A61M60/00—Blood pumps; Devices for mechanical circulatory actuation; Balloon pumps for circulatory assistance
- A61M60/80—Constructional details other than related to driving
- A61M60/802—Constructional details other than related to driving of non-positive displacement blood pumps
- A61M60/81—Pump housings
- A61M60/816—Sensors arranged on or in the housing, e.g. ultrasound flow sensors
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61M—DEVICES FOR INTRODUCING MEDIA INTO, OR ONTO, THE BODY; DEVICES FOR TRANSDUCING BODY MEDIA OR FOR TAKING MEDIA FROM THE BODY; DEVICES FOR PRODUCING OR ENDING SLEEP OR STUPOR
- A61M60/00—Blood pumps; Devices for mechanical circulatory actuation; Balloon pumps for circulatory assistance
- A61M60/80—Constructional details other than related to driving
- A61M60/855—Constructional details other than related to driving of implantable pumps or pumping devices
- A61M60/857—Implantable blood tubes
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61B—DIAGNOSIS; SURGERY; IDENTIFICATION
- A61B2562/00—Details of sensors; Constructional details of sensor housings or probes; Accessories for sensors
- A61B2562/02—Details of sensors specially adapted for in-vivo measurements
- A61B2562/0247—Pressure sensors
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61M—DEVICES FOR INTRODUCING MEDIA INTO, OR ONTO, THE BODY; DEVICES FOR TRANSDUCING BODY MEDIA OR FOR TAKING MEDIA FROM THE BODY; DEVICES FOR PRODUCING OR ENDING SLEEP OR STUPOR
- A61M2205/00—General characteristics of the apparatus
- A61M2205/10—General characteristics of the apparatus with powered movement mechanisms
- A61M2205/103—General characteristics of the apparatus with powered movement mechanisms rotating
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61M—DEVICES FOR INTRODUCING MEDIA INTO, OR ONTO, THE BODY; DEVICES FOR TRANSDUCING BODY MEDIA OR FOR TAKING MEDIA FROM THE BODY; DEVICES FOR PRODUCING OR ENDING SLEEP OR STUPOR
- A61M2205/00—General characteristics of the apparatus
- A61M2205/33—Controlling, regulating or measuring
- A61M2205/3306—Optical measuring means
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61M—DEVICES FOR INTRODUCING MEDIA INTO, OR ONTO, THE BODY; DEVICES FOR TRANSDUCING BODY MEDIA OR FOR TAKING MEDIA FROM THE BODY; DEVICES FOR PRODUCING OR ENDING SLEEP OR STUPOR
- A61M2205/00—General characteristics of the apparatus
- A61M2205/33—Controlling, regulating or measuring
- A61M2205/3331—Pressure; Flow
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61M—DEVICES FOR INTRODUCING MEDIA INTO, OR ONTO, THE BODY; DEVICES FOR TRANSDUCING BODY MEDIA OR FOR TAKING MEDIA FROM THE BODY; DEVICES FOR PRODUCING OR ENDING SLEEP OR STUPOR
- A61M2205/00—General characteristics of the apparatus
- A61M2205/33—Controlling, regulating or measuring
- A61M2205/3331—Pressure; Flow
- A61M2205/3334—Measuring or controlling the flow rate
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61M—DEVICES FOR INTRODUCING MEDIA INTO, OR ONTO, THE BODY; DEVICES FOR TRANSDUCING BODY MEDIA OR FOR TAKING MEDIA FROM THE BODY; DEVICES FOR PRODUCING OR ENDING SLEEP OR STUPOR
- A61M2205/00—General characteristics of the apparatus
- A61M2205/33—Controlling, regulating or measuring
- A61M2205/3331—Pressure; Flow
- A61M2205/3344—Measuring or controlling pressure at the body treatment site
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61M—DEVICES FOR INTRODUCING MEDIA INTO, OR ONTO, THE BODY; DEVICES FOR TRANSDUCING BODY MEDIA OR FOR TAKING MEDIA FROM THE BODY; DEVICES FOR PRODUCING OR ENDING SLEEP OR STUPOR
- A61M2205/00—General characteristics of the apparatus
- A61M2205/33—Controlling, regulating or measuring
- A61M2205/3331—Pressure; Flow
- A61M2205/3351—Controlling upstream pump pressure
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61M—DEVICES FOR INTRODUCING MEDIA INTO, OR ONTO, THE BODY; DEVICES FOR TRANSDUCING BODY MEDIA OR FOR TAKING MEDIA FROM THE BODY; DEVICES FOR PRODUCING OR ENDING SLEEP OR STUPOR
- A61M2205/00—General characteristics of the apparatus
- A61M2205/33—Controlling, regulating or measuring
- A61M2205/3331—Pressure; Flow
- A61M2205/3355—Controlling downstream pump pressure
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61M—DEVICES FOR INTRODUCING MEDIA INTO, OR ONTO, THE BODY; DEVICES FOR TRANSDUCING BODY MEDIA OR FOR TAKING MEDIA FROM THE BODY; DEVICES FOR PRODUCING OR ENDING SLEEP OR STUPOR
- A61M2210/00—Anatomical parts of the body
- A61M2210/12—Blood circulatory system
- A61M2210/125—Heart
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61M—DEVICES FOR INTRODUCING MEDIA INTO, OR ONTO, THE BODY; DEVICES FOR TRANSDUCING BODY MEDIA OR FOR TAKING MEDIA FROM THE BODY; DEVICES FOR PRODUCING OR ENDING SLEEP OR STUPOR
- A61M2210/00—Anatomical parts of the body
- A61M2210/12—Blood circulatory system
- A61M2210/127—Aorta
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61M—DEVICES FOR INTRODUCING MEDIA INTO, OR ONTO, THE BODY; DEVICES FOR TRANSDUCING BODY MEDIA OR FOR TAKING MEDIA FROM THE BODY; DEVICES FOR PRODUCING OR ENDING SLEEP OR STUPOR
- A61M2230/00—Measuring parameters of the user
- A61M2230/005—Parameter used as control input for the apparatus
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61M—DEVICES FOR INTRODUCING MEDIA INTO, OR ONTO, THE BODY; DEVICES FOR TRANSDUCING BODY MEDIA OR FOR TAKING MEDIA FROM THE BODY; DEVICES FOR PRODUCING OR ENDING SLEEP OR STUPOR
- A61M2230/00—Measuring parameters of the user
- A61M2230/30—Blood pressure
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61M—DEVICES FOR INTRODUCING MEDIA INTO, OR ONTO, THE BODY; DEVICES FOR TRANSDUCING BODY MEDIA OR FOR TAKING MEDIA FROM THE BODY; DEVICES FOR PRODUCING OR ENDING SLEEP OR STUPOR
- A61M60/00—Blood pumps; Devices for mechanical circulatory actuation; Balloon pumps for circulatory assistance
- A61M60/10—Location thereof with respect to the patient's body
- A61M60/122—Implantable pumps or pumping devices, i.e. the blood being pumped inside the patient's body
- A61M60/126—Implantable pumps or pumping devices, i.e. the blood being pumped inside the patient's body implantable via, into, inside, in line, branching on, or around a blood vessel
- A61M60/148—Implantable pumps or pumping devices, i.e. the blood being pumped inside the patient's body implantable via, into, inside, in line, branching on, or around a blood vessel in line with a blood vessel using resection or like techniques, e.g. permanent endovascular heart assist devices
Description
Claims (13)
- 血管内ロータリ血液ポンプであって、カテーテル(10)と、そのカテーテル(10)の先端側に固定され大凡の長軸を有するポンピング装置(50)と、ポンピング装置(50)に堅固連結されており且つ感圧面(32)を有する少なくとも1個の圧力センサ(27,28A,30;28B,60)と、を備える血液ポンプにおいて、その感圧面(32)が、周囲に対し曝露されており且つ上記大凡の長軸に対し直交整列されていることを特徴とする血液ポンプ。
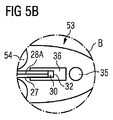
- 請求項1記載の血液ポンプであって、上記少なくとも1個の圧力センサがポンピング装置(50)のポンプハウジングに固定されており、そのハウジング内で少なくとも1個のインペラ(58)が回動する血液ポンプ。
- 請求項2記載の血液ポンプであって、インペラ(58)・カテーテル(10)間に少なくとも1個の第1血液通流開口(56)を備え、その第1血液通流開口(56)の付近且つ先端側に感圧面(32)が位置することとなるようポンプハウジングに圧力センサが固定されている血液ポンプ。
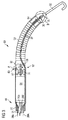
- 請求項1乃至3のうちいずれか一項記載の血液ポンプであって、ポンピング装置(50)が、少なくとも1個の第2血液通流開口(54)を有するフローカニューレ(53)をその先端に有し、血液ポンプ作動中には、ポンピング装置(50)がその第2血液通流開口を通じ血液を吸引又は吐出し、上記少なくとも1個の圧力センサが、その第2血液通流開口(54)の付近に配置されている血液ポンプ。
- 請求項4記載の血液ポンプであって、フローカニューレ(53)が、ソフトフレキシブルチップ(55)を、第2血液通流開口(54)の先端側に有し、圧力センサの先端(30)が、少なくとも部分的に、そのソフトフレキシブルチップ(55)内に配置されている血液ポンプ。
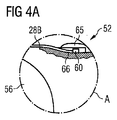
- 請求項1乃至5のうちいずれか一項記載の血液ポンプであって、圧力センサが、手元端側から先端側へと外側を通りポンピング装置(50)沿いに案内されており、且つ、ポンピング装置(50)が、圧力センサの先端(30;60)がその中に少なくとも部分的に配置される窪み(36;66)を有する外面を有する血液ポンプ。
- 請求項1乃至6のうちいずれか一項記載の血液ポンプであって、圧力センサの先端(30;60)が、ポンピング装置(50)の周を越えて径方向に突出しており、且つ、ポンピング装置(50)上、圧力センサの先端(30;60)の先端側前方に、ポンピング装置(50)の周を越えて同様に突出するバルジ(35;65)が設けられている血液ポンプ。
- 請求項7記載の血液ポンプであって、バルジ(35;65)がU字状又はO字状である血液ポンプ。
- 請求項7又は8記載の血液ポンプであって、バルジ(35;65)が接合剤のビーズである血液ポンプ。
- 請求項7又は8記載の血液ポンプであって、バルジ(35;65)が、ポンピング装置(50)の表面上に熔接又は半田付けされている血液ポンプ。
- 請求項7又は8記載の血液ポンプであって、バルジ(35;65)が、ポンピング装置(50)の一体部分を形成する血液ポンプ。
- 請求項1乃至11のうちいずれか一項記載の血液ポンプであって、圧力センサが、光ファイバ(28A;28B)を有する光学圧力センサであり、その感圧面(32)が膜であり、光ファイバ(28A,28B)がその膜から離隔した場所で終端する血液ポンプ。
- 請求項1乃至12のうちいずれか一項記載の血液ポンプであって、感圧面(32)が、そのセラミクス面又はガラス面が周囲に対し直に曝露するセラミクス又はガラス膜である血液ポンプ。
Applications Claiming Priority (3)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| DE102012207049.4A DE102012207049A1 (de) | 2012-04-27 | 2012-04-27 | Intravasale rotationsblutpumpe |
| DE102012207049.4 | 2012-04-27 | ||
| PCT/EP2013/058642 WO2013160407A1 (de) | 2012-04-27 | 2013-04-25 | Intravasale rotationsblutpumpe |
Publications (3)
| Publication Number | Publication Date |
|---|---|
| JP2015514529A JP2015514529A (ja) | 2015-05-21 |
| JP2015514529A5 JP2015514529A5 (ja) | 2016-06-16 |
| JP6055083B2 true JP6055083B2 (ja) | 2016-12-27 |
Family
ID=48182918
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| JP2015507537A Active JP6055083B2 (ja) | 2012-04-27 | 2013-04-25 | 血管内ロータリ血液ポンプ |
Country Status (7)
| Country | Link |
|---|---|
| US (1) | US9669142B2 (ja) |
| EP (1) | EP2841122B1 (ja) |
| JP (1) | JP6055083B2 (ja) |
| KR (1) | KR101915801B1 (ja) |
| AU (1) | AU2013254646B2 (ja) |
| DE (1) | DE102012207049A1 (ja) |
| WO (1) | WO2013160407A1 (ja) |
Families Citing this family (80)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US7393181B2 (en) | 2004-09-17 | 2008-07-01 | The Penn State Research Foundation | Expandable impeller pump |
| EP3115070B8 (en) | 2006-03-23 | 2019-05-08 | The Penn State Research Foundation | Heart assist device with expandable impeller pump |
| WO2012094641A2 (en) | 2011-01-06 | 2012-07-12 | Thoratec Corporation | Percutaneous heart pump |
| DE102012207053A1 (de) | 2012-04-27 | 2013-10-31 | Abiomed Europe Gmbh | Intravasale rotationsblutpumpe |
| DE102013008168A1 (de) | 2012-05-14 | 2013-11-14 | Thoratec Corporation | Laufrad für Katheterpumpe |
| US9872947B2 (en) | 2012-05-14 | 2018-01-23 | Tc1 Llc | Sheath system for catheter pump |
| US9446179B2 (en) | 2012-05-14 | 2016-09-20 | Thoratec Corporation | Distal bearing support |
| US8721517B2 (en) | 2012-05-14 | 2014-05-13 | Thoratec Corporation | Impeller for catheter pump |
| US9327067B2 (en) | 2012-05-14 | 2016-05-03 | Thoratec Corporation | Impeller for catheter pump |
| EP2858711B1 (en) | 2012-06-06 | 2018-03-07 | Magenta Medical Ltd. | Prosthetic renal valve |
| US9358329B2 (en) | 2012-07-03 | 2016-06-07 | Thoratec Corporation | Catheter pump |
| US9421311B2 (en) | 2012-07-03 | 2016-08-23 | Thoratec Corporation | Motor assembly for catheter pump |
| EP4186557A1 (en) | 2012-07-03 | 2023-05-31 | Tc1 Llc | Motor assembly for catheter pump |
| EP3834876B1 (en) | 2013-03-13 | 2022-09-14 | Tc1 Llc | Fluid handling system |
| CN113616920A (zh) | 2013-03-13 | 2021-11-09 | 马真塔医药有限公司 | 血液泵浦装置及制造血液泵浦的方法 |
| US11033728B2 (en) | 2013-03-13 | 2021-06-15 | Tc1 Llc | Fluid handling system |
| US10583231B2 (en) | 2013-03-13 | 2020-03-10 | Magenta Medical Ltd. | Blood pump |
| US11077294B2 (en) | 2013-03-13 | 2021-08-03 | Tc1 Llc | Sheath assembly for catheter pump |
| EP3808390B1 (en) | 2013-03-15 | 2023-11-08 | Tc1 Llc | Catheter pump assembly including a stator |
| US9308302B2 (en) | 2013-03-15 | 2016-04-12 | Thoratec Corporation | Catheter pump assembly including a stator |
| WO2015023789A1 (en) * | 2013-08-14 | 2015-02-19 | Boston Scientific Scimed, Inc. | Medical device systems including an optical fiber with a tapered core |
| EP2851100A1 (en) * | 2013-09-20 | 2015-03-25 | Berlin Heart GmbH | Blood pump control system and method for controlling a blood pump |
| WO2015130768A2 (en) | 2014-02-25 | 2015-09-03 | KUSHWAHA, Sudhir | Ventricular assist device and method |
| US9827356B2 (en) | 2014-04-15 | 2017-11-28 | Tc1 Llc | Catheter pump with access ports |
| WO2015160943A1 (en) | 2014-04-15 | 2015-10-22 | Thoratec Corporation | Sensors for catheter pumps |
| US10583232B2 (en) | 2014-04-15 | 2020-03-10 | Tc1 Llc | Catheter pump with off-set motor position |
| EP3791920A1 (en) | 2014-04-15 | 2021-03-17 | Tc1 Llc | Catheter pump introducer systems and methods |
| US9901722B2 (en) | 2014-06-01 | 2018-02-27 | White Swell Medical Ltd | System and method for treatment of pulmonary edema |
| US10449279B2 (en) | 2014-08-18 | 2019-10-22 | Tc1 Llc | Guide features for percutaneous catheter pump |
| WO2016118784A1 (en) | 2015-01-22 | 2016-07-28 | Thoratec Corporation | Attachment mechanisms for motor of catheter pump |
| US9770543B2 (en) | 2015-01-22 | 2017-09-26 | Tc1 Llc | Reduced rotational mass motor assembly for catheter pump |
| EP3247421B1 (en) | 2015-01-22 | 2019-10-02 | Tc1 Llc | Motor assembly with heat exchanger for catheter pump |
| US9907890B2 (en) | 2015-04-16 | 2018-03-06 | Tc1 Llc | Catheter pump with positioning brace |
| US10149684B2 (en) | 2015-05-11 | 2018-12-11 | White Swell Medical Ltd | Systems and methods for reducing pressure at an outflow of a duct |
| WO2016185473A1 (en) | 2015-05-18 | 2016-11-24 | Magenta Medical Ltd. | Blood pump |
| EP3205359B1 (en) * | 2016-02-11 | 2018-08-29 | Abiomed Europe GmbH | Blood pump system |
| WO2017214118A1 (en) * | 2016-06-06 | 2017-12-14 | Abiomed, Inc. | Blood pump assembly having a sensor and a sensor shield |
| EP3804804A1 (en) | 2016-07-21 | 2021-04-14 | Tc1 Llc | Fluid seals for catheter pump motor assembly |
| WO2018017683A1 (en) | 2016-07-21 | 2018-01-25 | Thoratec Corporation | Gas-filled chamber for catheter pump motor assembly |
| EP3287154B1 (en) * | 2016-08-23 | 2019-10-09 | Abiomed Europe GmbH | Ventricular assist device |
| WO2018045299A1 (en) * | 2016-09-01 | 2018-03-08 | Abiomed, Inc. | Anti-suction blood pump inlet |
| JP7383476B2 (ja) | 2016-10-25 | 2023-11-20 | マジェンタ・メディカル・リミテッド | 心室補助デバイス |
| US10960189B2 (en) | 2016-11-01 | 2021-03-30 | White Swell Medical Ltd | Systems and methods for treatment of fluid overload |
| EP3544649B1 (en) | 2016-11-23 | 2023-06-07 | Magenta Medical Ltd. | Blood pumps |
| CN116549830A (zh) | 2017-03-02 | 2023-08-08 | 怀特斯维尔医疗有限公司 | 导管系统 |
| US11406393B2 (en) | 2017-03-19 | 2022-08-09 | White Swell Medical Ltd | Methods and devices for reducing pressure |
| US10478542B2 (en) | 2017-03-20 | 2019-11-19 | Abiomed, Inc. | Cannula having nitinol reinforced inflow region |
| EP3398624A1 (en) * | 2017-05-04 | 2018-11-07 | Abiomed Europe GmbH | Blood pump with reinforced catheter |
| CA3066361A1 (en) | 2017-06-07 | 2018-12-13 | Shifamed Holdings, Llc | Intravascular fluid movement devices, systems, and methods of use |
| EP3644846B1 (en) | 2017-08-03 | 2022-10-19 | Boston Scientific Scimed, Inc. | Systems for assessing fractional flow reserve |
| EP3710076B1 (en) | 2017-11-13 | 2023-12-27 | Shifamed Holdings, LLC | Intravascular fluid movement devices, systems, and methods of use |
| US10905808B2 (en) | 2018-01-10 | 2021-02-02 | Magenta Medical Ltd. | Drive cable for use with a blood pump |
| CN115089870A (zh) | 2018-01-10 | 2022-09-23 | 马真塔医药有限公司 | 心室辅助装置 |
| DE102018201030A1 (de) | 2018-01-24 | 2019-07-25 | Kardion Gmbh | Magnetkuppelelement mit magnetischer Lagerungsfunktion |
| EP3746149A4 (en) | 2018-02-01 | 2021-10-27 | Shifamed Holdings, LLC | INTRAVASCULAR BLOOD PUMPS AND METHODS OF USE AND METHODS OF MANUFACTURING |
| EP3755215B1 (en) | 2018-02-23 | 2022-07-20 | Boston Scientific Scimed Inc. | System for assessing a vessel with sequential physiological measurements |
| CN112088022A (zh) | 2018-03-16 | 2020-12-15 | 阿比奥梅德公司 | 用于估计心脏泵的位置的系统和方法 |
| US11850073B2 (en) | 2018-03-23 | 2023-12-26 | Boston Scientific Scimed, Inc. | Medical device with pressure sensor |
| JP7138189B2 (ja) | 2018-04-06 | 2022-09-15 | ボストン サイエンティフィック サイムド,インコーポレイテッド | 圧力センサ付き医療装置 |
| EP4311482A3 (en) | 2018-04-18 | 2024-03-06 | Boston Scientific Scimed, Inc. | System for assessing a vessel with sequential physiological measurements |
| US11167121B2 (en) * | 2018-05-15 | 2021-11-09 | Cardiovascular Systems, Inc. | Intravascular pump with integrated isolated conductor(s) and methods thereof |
| US11141580B2 (en) * | 2018-05-15 | 2021-10-12 | Cardiovascular Systems, Inc. | Intravascular blood pump system with integrated conductor(s) in housing and methods thereof |
| DE102018208911A1 (de) * | 2018-06-06 | 2019-12-12 | Kardion Gmbh | Leitungsvorrichtung für ein Herzunterstützungssystem und Verfahren zum Herstellen einer Leitungsvorrichtung |
| US11013904B2 (en) | 2018-07-30 | 2021-05-25 | Cardiovascular Systems, Inc. | Intravascular pump with proximal and distal pressure or flow sensors and distal sensor tracking |
| CA3112595A1 (en) * | 2018-09-21 | 2020-03-26 | Abiomed, Inc. | Use of optical fiber sensor as a diagnostic tool in catheter-based medical devices |
| EP3873554A2 (en) * | 2018-12-21 | 2021-09-08 | Tc1 Llc | Implantable blood pump assembly including pressure sensor and methods of assembling same |
| EP3749383B1 (en) | 2019-01-24 | 2021-04-28 | Magenta Medical Ltd. | Ventricular assist device |
| US11724095B2 (en) | 2019-02-26 | 2023-08-15 | White Swell Medical Ltd | Devices and methods for treating edema |
| US11717652B2 (en) | 2019-02-26 | 2023-08-08 | White Swell Medical Ltd | Devices and methods for treating edema |
| US11931560B2 (en) | 2019-02-26 | 2024-03-19 | White Swell Medical Ltd | Devices and methods for treating edema |
| US11793996B2 (en) | 2019-02-26 | 2023-10-24 | White Swell Medical Ltd | Devices and methods for treating edema |
| US11660426B2 (en) | 2019-02-26 | 2023-05-30 | White Swell Medical Ltd | Devices and methods for treating edema |
| JP2022540616A (ja) | 2019-07-12 | 2022-09-16 | シファメド・ホールディングス・エルエルシー | 血管内血液ポンプならびに製造および使用の方法 |
| US11654275B2 (en) | 2019-07-22 | 2023-05-23 | Shifamed Holdings, Llc | Intravascular blood pumps with struts and methods of use and manufacture |
| EP4034221A4 (en) * | 2019-09-25 | 2023-10-11 | Shifamed Holdings, LLC | CATHETER BLOOD PUMPS AND FOLDABLE PUMP HOUSINGS |
| WO2021062265A1 (en) | 2019-09-25 | 2021-04-01 | Shifamed Holdings, Llc | Intravascular blood pump systems and methods of use and control thereof |
| DE102020102474A1 (de) | 2020-01-31 | 2021-08-05 | Kardion Gmbh | Pumpe zum Fördern eines Fluids und Verfahren zum Herstellen einer Pumpe |
| US20230086096A1 (en) * | 2021-09-17 | 2023-03-23 | Abiomed, Inc. | Intravascular blood pump |
| WO2023092122A1 (en) * | 2021-11-19 | 2023-05-25 | Shifamed Holdings, Llc | Intravascular blood pumps, motors, and fluid control |
| EP4245356A1 (en) * | 2022-03-15 | 2023-09-20 | Abiomed Europe GmbH | Sensor carrier, blood pump comprising a sensor carrier and guidewire comprising a sensor carrier |
Family Cites Families (15)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US5645531A (en) | 1993-05-26 | 1997-07-08 | Quest Medical, Inc. | Constant pressure blood mixture delivery system and method |
| US5911685A (en) | 1996-04-03 | 1999-06-15 | Guidant Corporation | Method and apparatus for cardiac blood flow assistance |
| US5964694A (en) * | 1997-04-02 | 1999-10-12 | Guidant Corporation | Method and apparatus for cardiac blood flow assistance |
| US6176822B1 (en) | 1998-03-31 | 2001-01-23 | Impella Cardiotechnik Gmbh | Intracardiac blood pump |
| DE19821307C1 (de) * | 1998-05-13 | 1999-10-21 | Impella Cardiotech Gmbh | Intrakardiale Blutpumpe |
| WO2000037139A1 (en) | 1998-12-23 | 2000-06-29 | A-Med Systems, Inc. | Left and right side heart support |
| DE19909159C1 (de) | 1999-03-02 | 2000-11-30 | Siemens Ag | Lichtwellenleiteranordnung |
| DE10016422B4 (de) * | 2000-04-01 | 2013-10-31 | Impella Cardiosystems Ag | Parakardiale Blutpumpe |
| DE10040403A1 (de) | 2000-08-18 | 2002-02-28 | Impella Cardiotech Ag | Intrakardiale Blutpumpe |
| US6398738B1 (en) | 2000-09-25 | 2002-06-04 | Millar Instruments, Inc. | Method and apparatus for reconstructing a high fidelity pressure waveform with a balloon catheter |
| DE10060275A1 (de) * | 2000-12-05 | 2002-06-13 | Impella Cardiotech Ag | Verfahren zum Kalibrieren eines Drucksensors oder eines Flussensors an einer Rotationspumpe |
| AU2002220271A1 (en) | 2000-12-12 | 2002-06-24 | Datascope Investment Corp. | Intra-aortic balloon catheter having a fiberoptic sensor |
| US7822306B2 (en) | 2007-01-08 | 2010-10-26 | Commscope, Inc. Of North Carolina | Buoyancy neutral fiber optic cable |
| CA2721282C (en) * | 2009-03-17 | 2011-10-11 | Opsens Inc. | Eccentric pressure catheter with guidewire compatibility |
| DE102009047845A1 (de) | 2009-09-30 | 2011-03-31 | Abiomed Europe Gmbh | Herzunterstützungssystem |
-
2012
- 2012-04-27 DE DE102012207049.4A patent/DE102012207049A1/de not_active Withdrawn
-
2013
- 2013-04-25 JP JP2015507537A patent/JP6055083B2/ja active Active
- 2013-04-25 KR KR1020147032835A patent/KR101915801B1/ko active IP Right Grant
- 2013-04-25 WO PCT/EP2013/058642 patent/WO2013160407A1/de active Application Filing
- 2013-04-25 EP EP13718340.6A patent/EP2841122B1/de active Active
- 2013-04-25 US US14/396,708 patent/US9669142B2/en active Active
- 2013-04-25 AU AU2013254646A patent/AU2013254646B2/en active Active
Also Published As
| Publication number | Publication date |
|---|---|
| DE102012207049A1 (de) | 2015-08-13 |
| US20150141842A1 (en) | 2015-05-21 |
| US9669142B2 (en) | 2017-06-06 |
| WO2013160407A1 (de) | 2013-10-31 |
| AU2013254646B2 (en) | 2017-06-15 |
| JP2015514529A (ja) | 2015-05-21 |
| EP2841122A1 (de) | 2015-03-04 |
| KR20150008146A (ko) | 2015-01-21 |
| EP2841122B1 (de) | 2015-10-07 |
| AU2013254646A1 (en) | 2014-11-13 |
| KR101915801B1 (ko) | 2018-11-06 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| JP6055083B2 (ja) | 血管内ロータリ血液ポンプ | |
| JP7219794B2 (ja) | 血液ポンプシステム | |
| US11844940B2 (en) | Blood pump assembly having a sensor and a sensor shield | |
| KR102033944B1 (ko) | 혈관내 로터리 혈액 펌프 | |
| JP5347656B2 (ja) | カテーテル | |
| KR102644874B1 (ko) | 카테터 시스템 및 그 카테터 시스템을 포함하는 혈관내 혈액 펌프 | |
| CN109641092B (zh) | 用于心室内vad的集成传感器 | |
| JPH0239464Y2 (ja) |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| A521 | Request for written amendment filed |
Free format text: JAPANESE INTERMEDIATE CODE: A523 Effective date: 20160422 |
|
| A621 | Written request for application examination |
Free format text: JAPANESE INTERMEDIATE CODE: A621 Effective date: 20160422 |
|
| A871 | Explanation of circumstances concerning accelerated examination |
Free format text: JAPANESE INTERMEDIATE CODE: A871 Effective date: 20160422 |
|
| A975 | Report on accelerated examination |
Free format text: JAPANESE INTERMEDIATE CODE: A971005 Effective date: 20160527 |
|
| A131 | Notification of reasons for refusal |
Free format text: JAPANESE INTERMEDIATE CODE: A131 Effective date: 20160607 |
|
| A521 | Request for written amendment filed |
Free format text: JAPANESE INTERMEDIATE CODE: A523 Effective date: 20160906 |
|
| TRDD | Decision of grant or rejection written | ||
| A01 | Written decision to grant a patent or to grant a registration (utility model) |
Free format text: JAPANESE INTERMEDIATE CODE: A01 Effective date: 20161108 |
|
| A61 | First payment of annual fees (during grant procedure) |
Free format text: JAPANESE INTERMEDIATE CODE: A61 Effective date: 20161201 |
|
| R150 | Certificate of patent or registration of utility model |
Ref document number: 6055083 Country of ref document: JP Free format text: JAPANESE INTERMEDIATE CODE: R150 |
|
| R250 | Receipt of annual fees |
Free format text: JAPANESE INTERMEDIATE CODE: R250 |
|
| R250 | Receipt of annual fees |
Free format text: JAPANESE INTERMEDIATE CODE: R250 |
|
| R250 | Receipt of annual fees |
Free format text: JAPANESE INTERMEDIATE CODE: R250 |
|
| R250 | Receipt of annual fees |
Free format text: JAPANESE INTERMEDIATE CODE: R250 |
|
| R250 | Receipt of annual fees |
Free format text: JAPANESE INTERMEDIATE CODE: R250 |