JP4026041B2 - Neutral titania sol - Google Patents
Neutral titania sol Download PDFInfo
- Publication number
- JP4026041B2 JP4026041B2 JP10191599A JP10191599A JP4026041B2 JP 4026041 B2 JP4026041 B2 JP 4026041B2 JP 10191599 A JP10191599 A JP 10191599A JP 10191599 A JP10191599 A JP 10191599A JP 4026041 B2 JP4026041 B2 JP 4026041B2
- Authority
- JP
- Japan
- Prior art keywords
- sol
- tio
- titanium
- titania sol
- compound
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Expired - Lifetime
Links
- GWEVSGVZZGPLCZ-UHFFFAOYSA-N Titan oxide Chemical compound O=[Ti]=O GWEVSGVZZGPLCZ-UHFFFAOYSA-N 0.000 title claims description 95
- 230000007935 neutral effect Effects 0.000 title claims description 25
- NBIIXXVUZAFLBC-UHFFFAOYSA-N phosphoric acid Substances OP(O)(O)=O NBIIXXVUZAFLBC-UHFFFAOYSA-N 0.000 claims description 38
- 239000002245 particle Substances 0.000 claims description 34
- 229910010413 TiO 2 Inorganic materials 0.000 claims description 28
- 239000007864 aqueous solution Substances 0.000 claims description 24
- OGIDPMRJRNCKJF-UHFFFAOYSA-N titanium oxide Inorganic materials [Ti]=O OGIDPMRJRNCKJF-UHFFFAOYSA-N 0.000 claims description 22
- -1 titanium phosphate compound Chemical class 0.000 claims description 20
- 239000002253 acid Substances 0.000 claims description 12
- 229910000147 aluminium phosphate Inorganic materials 0.000 claims description 10
- 238000000034 method Methods 0.000 claims description 10
- XLYOFNOQVPJJNP-UHFFFAOYSA-N water Substances O XLYOFNOQVPJJNP-UHFFFAOYSA-N 0.000 claims description 10
- 239000006185 dispersion Substances 0.000 claims description 9
- 150000003609 titanium compounds Chemical class 0.000 claims description 9
- 238000006243 chemical reaction Methods 0.000 claims description 8
- 238000004519 manufacturing process Methods 0.000 claims description 8
- 239000011164 primary particle Substances 0.000 claims description 7
- 239000000243 solution Substances 0.000 claims description 7
- 239000011248 coating agent Substances 0.000 claims description 6
- 238000000576 coating method Methods 0.000 claims description 6
- 238000001914 filtration Methods 0.000 claims description 5
- 230000003472 neutralizing effect Effects 0.000 claims description 5
- 238000002156 mixing Methods 0.000 claims description 2
- 238000005406 washing Methods 0.000 claims description 2
- 235000011007 phosphoric acid Nutrition 0.000 description 20
- XJDNKRIXUMDJCW-UHFFFAOYSA-J titanium tetrachloride Chemical compound Cl[Ti](Cl)(Cl)Cl XJDNKRIXUMDJCW-UHFFFAOYSA-J 0.000 description 15
- VEXZGXHMUGYJMC-UHFFFAOYSA-N Hydrochloric acid Chemical compound Cl VEXZGXHMUGYJMC-UHFFFAOYSA-N 0.000 description 12
- 239000002002 slurry Substances 0.000 description 12
- 230000000052 comparative effect Effects 0.000 description 8
- 239000007787 solid Substances 0.000 description 8
- VHUUQVKOLVNVRT-UHFFFAOYSA-N Ammonium hydroxide Chemical compound [NH4+].[OH-] VHUUQVKOLVNVRT-UHFFFAOYSA-N 0.000 description 6
- 235000011114 ammonium hydroxide Nutrition 0.000 description 6
- 239000010936 titanium Substances 0.000 description 6
- 229910000349 titanium oxysulfate Inorganic materials 0.000 description 6
- 238000002834 transmittance Methods 0.000 description 6
- 230000002378 acidificating effect Effects 0.000 description 5
- 230000001699 photocatalysis Effects 0.000 description 5
- MWUXSHHQAYIFBG-UHFFFAOYSA-N Nitric oxide Chemical compound O=[N] MWUXSHHQAYIFBG-UHFFFAOYSA-N 0.000 description 4
- VYPSYNLAJGMNEJ-UHFFFAOYSA-N Silicium dioxide Chemical compound O=[Si]=O VYPSYNLAJGMNEJ-UHFFFAOYSA-N 0.000 description 4
- QGZKDVFQNNGYKY-UHFFFAOYSA-N ammonia Natural products N QGZKDVFQNNGYKY-UHFFFAOYSA-N 0.000 description 4
- 238000005259 measurement Methods 0.000 description 4
- 239000000203 mixture Substances 0.000 description 4
- 239000003973 paint Substances 0.000 description 4
- 239000000758 substrate Substances 0.000 description 4
- LFQSCWFLJHTTHZ-UHFFFAOYSA-N Ethanol Chemical compound CCO LFQSCWFLJHTTHZ-UHFFFAOYSA-N 0.000 description 3
- UEZVMMHDMIWARA-UHFFFAOYSA-N Metaphosphoric acid Chemical compound OP(=O)=O UEZVMMHDMIWARA-UHFFFAOYSA-N 0.000 description 3
- OKKJLVBELUTLKV-UHFFFAOYSA-N Methanol Chemical compound OC OKKJLVBELUTLKV-UHFFFAOYSA-N 0.000 description 3
- GRYLNZFGIOXLOG-UHFFFAOYSA-N Nitric acid Chemical compound O[N+]([O-])=O GRYLNZFGIOXLOG-UHFFFAOYSA-N 0.000 description 3
- OAICVXFJPJFONN-UHFFFAOYSA-N Phosphorus Chemical compound [P] OAICVXFJPJFONN-UHFFFAOYSA-N 0.000 description 3
- HEMHJVSKTPXQMS-UHFFFAOYSA-M Sodium hydroxide Chemical compound [OH-].[Na+] HEMHJVSKTPXQMS-UHFFFAOYSA-M 0.000 description 3
- 239000011230 binding agent Substances 0.000 description 3
- 229910017604 nitric acid Inorganic materials 0.000 description 3
- 229910052698 phosphorus Inorganic materials 0.000 description 3
- 239000011574 phosphorus Substances 0.000 description 3
- 239000004065 semiconductor Substances 0.000 description 3
- 239000003381 stabilizer Substances 0.000 description 3
- 238000003860 storage Methods 0.000 description 3
- LYCAIKOWRPUZTN-UHFFFAOYSA-N Ethylene glycol Chemical compound OCCO LYCAIKOWRPUZTN-UHFFFAOYSA-N 0.000 description 2
- KFZMGEQAYNKOFK-UHFFFAOYSA-N Isopropanol Chemical compound CC(C)O KFZMGEQAYNKOFK-UHFFFAOYSA-N 0.000 description 2
- 229910019142 PO4 Inorganic materials 0.000 description 2
- RTAQQCXQSZGOHL-UHFFFAOYSA-N Titanium Chemical compound [Ti] RTAQQCXQSZGOHL-UHFFFAOYSA-N 0.000 description 2
- 239000003513 alkali Substances 0.000 description 2
- 230000000694 effects Effects 0.000 description 2
- 239000011521 glass Substances 0.000 description 2
- 230000003301 hydrolyzing effect Effects 0.000 description 2
- 230000001771 impaired effect Effects 0.000 description 2
- 238000005342 ion exchange Methods 0.000 description 2
- 235000010746 mayonnaise Nutrition 0.000 description 2
- 239000008268 mayonnaise Substances 0.000 description 2
- 239000002609 medium Substances 0.000 description 2
- 230000003287 optical effect Effects 0.000 description 2
- 239000010452 phosphate Substances 0.000 description 2
- 235000021317 phosphate Nutrition 0.000 description 2
- 239000011941 photocatalyst Substances 0.000 description 2
- 239000002994 raw material Substances 0.000 description 2
- 229910052719 titanium Inorganic materials 0.000 description 2
- JVKRKMWZYMKVTQ-UHFFFAOYSA-N 2-[4-[2-(2,3-dihydro-1H-inden-2-ylamino)pyrimidin-5-yl]pyrazol-1-yl]-N-(2-oxo-3H-1,3-benzoxazol-6-yl)acetamide Chemical compound C1C(CC2=CC=CC=C12)NC1=NC=C(C=N1)C=1C=NN(C=1)CC(=O)NC1=CC2=C(NC(O2)=O)C=C1 JVKRKMWZYMKVTQ-UHFFFAOYSA-N 0.000 description 1
- AFCIMSXHQSIHQW-UHFFFAOYSA-N [O].[P] Chemical class [O].[P] AFCIMSXHQSIHQW-UHFFFAOYSA-N 0.000 description 1
- 239000004063 acid-resistant material Substances 0.000 description 1
- 239000000654 additive Substances 0.000 description 1
- 230000002411 adverse Effects 0.000 description 1
- 239000012736 aqueous medium Substances 0.000 description 1
- 239000011324 bead Substances 0.000 description 1
- 230000003197 catalytic effect Effects 0.000 description 1
- 239000003795 chemical substances by application Substances 0.000 description 1
- 239000008119 colloidal silica Substances 0.000 description 1
- 150000001875 compounds Chemical class 0.000 description 1
- 238000007796 conventional method Methods 0.000 description 1
- 239000013078 crystal Substances 0.000 description 1
- 238000000502 dialysis Methods 0.000 description 1
- 239000002612 dispersion medium Substances 0.000 description 1
- 238000009826 distribution Methods 0.000 description 1
- 238000000909 electrodialysis Methods 0.000 description 1
- 238000007429 general method Methods 0.000 description 1
- 238000010438 heat treatment Methods 0.000 description 1
- WGCNASOHLSPBMP-UHFFFAOYSA-N hydroxyacetaldehyde Natural products OCC=O WGCNASOHLSPBMP-UHFFFAOYSA-N 0.000 description 1
- 150000002484 inorganic compounds Chemical class 0.000 description 1
- 229910010272 inorganic material Inorganic materials 0.000 description 1
- 230000010354 integration Effects 0.000 description 1
- 239000000463 material Substances 0.000 description 1
- 239000012528 membrane Substances 0.000 description 1
- QSHDDOUJBYECFT-UHFFFAOYSA-N mercury Chemical compound [Hg] QSHDDOUJBYECFT-UHFFFAOYSA-N 0.000 description 1
- 229910052753 mercury Inorganic materials 0.000 description 1
- 229910021645 metal ion Inorganic materials 0.000 description 1
- 239000000693 micelle Substances 0.000 description 1
- 238000006386 neutralization reaction Methods 0.000 description 1
- 150000002894 organic compounds Chemical class 0.000 description 1
- 239000003960 organic solvent Substances 0.000 description 1
- 150000003016 phosphoric acids Chemical class 0.000 description 1
- 108090000765 processed proteins & peptides Proteins 0.000 description 1
- 239000010453 quartz Substances 0.000 description 1
- 150000003839 salts Chemical class 0.000 description 1
- 239000004576 sand Substances 0.000 description 1
- 239000007962 solid dispersion Substances 0.000 description 1
- 239000007858 starting material Substances 0.000 description 1
- JUWGUJSXVOBPHP-UHFFFAOYSA-B titanium(4+);tetraphosphate Chemical class [Ti+4].[Ti+4].[Ti+4].[O-]P([O-])([O-])=O.[O-]P([O-])([O-])=O.[O-]P([O-])([O-])=O.[O-]P([O-])([O-])=O JUWGUJSXVOBPHP-UHFFFAOYSA-B 0.000 description 1
- 238000000108 ultra-filtration Methods 0.000 description 1
Landscapes
- Inorganic Compounds Of Heavy Metals (AREA)
- Colloid Chemistry (AREA)
- Catalysts (AREA)
Description
【0001】
【産業上の利用分野】
本発明は、中性領域において透明かつ安定なチタニアゾルおよびその製造方法に関する。このゾルはバインダーと混合して基材に塗布し、例えば光触媒膜、半導体膜、紫外線カット皮膜の形成に用いることができる。
【0002】
【従来の技術とその問題点】
チタニアゾルは基材の表面に光触媒膜、半導体膜、紫外線カット膜などを形成するために有用である。チタニアゾルを得るための一般的な方法は、含水酸化チタンを塩酸または硝酸のような強酸で解膠する方法である。しかしながらこの方法で得られたゾルは解膠剤の強酸を含むためそのままでは取扱い上および使用上多くの制約がある。例えば輸送、貯蔵および他の取扱い用器具は耐酸性であることを必要とし、膜をその上に形成する基材も耐酸性材料に制限される。
【0003】
強酸で解膠したチタニアゾルは強酸性領域においてのみ安定であるため、ゾルから酸成分を除去すると分散粒子が凝集し、ゾルを形成しなくなる。このため強酸性のチタニアゾルへ安定剤として働く有機または無機化合物を添加した後、アルカリ中和またはイオン交換等によって中性化する方法が提案されている(特開昭63−123815,特開昭63−215520,特公平7−33255)。この場合ゾル中に添加した安定剤が比較的多く含まれるため、ゾルから形成された膜の光触媒活性や透明性を損なう。
【0004】
そこで本発明の課題は、このような欠点を持たない、すなわちチタニアゾルが本来持っている高い触媒活性および透明性に悪影響することなく中性域において安定なチタニアゾルおよびその製造方法を提供することである。
【0005】
【課題の解決方法】
本発明により、中性領域において透明性および分散安定性を有する水和リン酸チタン化合物被覆解膠酸化チタン粒子よりなる中性チタニアゾルが提供される。
【0006】
本発明はまた、上に記載した中性チタニアゾルの製造方法を提供する。この方法は、含水酸化チタンを塩酸または硝酸のような一塩基性強酸で解膠して得たチタニアゾルを出発原料とし、これを水溶性チタン化合物およびリン酸化合物の水溶液と混合し、反応液から酸を除去する。これによって中性領域において透明性および分散安定性を有するチタニアゾルを得ることができる。
【0007】
【具体的な実施方法】
硫酸チタニルの水溶液を加熱等により加水分解し、析出する含水酸化チタンを中和、濾過、水洗して得られるケーキを、塩酸、硝酸のような強酸で解膠することによりチタニアゾルを製造する方法は良く知られている。また、この際の反応条件の選択によって結晶形がルチル形、アナタース形または無定形の酸化チタン粒子が得られることも良く知られている。本発明では分散酸化チタン粒子の結晶形を問わないが、光触媒用途に対してアナタース形が好ましい。
【0008】
このようにして得られたチタニアゾルは強酸性(pH3以下)であり、透明で、一般に平均一次粒子径は50nm以下で、平均ミセル径は10〜150nmの範囲にある。先に述べたように、このゾルは中性領域において不安定である。そこで本発明は水溶性チタン化合物の水溶液と、リン酸化合物の水溶液をゾルと混合し、酸化チタン分散粒子を水溶液チタン化合物とリン酸化合物の反応によって生成した水和リン酸チタン化合物によって被覆する。この被覆によってゾルの等電点が酸性側にシフトし、中性領域において安定なゾルになるものと考えられる。
【0009】
使用し得る水溶性チタン化合物としては四塩化チタンおよび硫酸チタニルなどがある。リン酸化合物としては各種のリンの酸素酸、例えば正リン酸およびメタリン酸、ならびにそれらの水溶性塩がある。正リン酸またはメタリン酸が好ましい。これら化合物は直接またはゾルを中性にすることにより反応してゾル中の分散酸化チタンを被覆する水和リン酸チタン化合物を生成する。混合の順序は、分散酸化チタン粒子の不存在下にチタン化合物とリン酸化合物との反応が生起しない限り任意である。しかしながらチタニアゾルへ、水溶性チタン化合物の水溶液とリン酸化合物の水溶液を順次添加するのが好ましい。
【0010】
この時点ではゾルは依然として強酸性である。ゾルのpHを中性域へシフトさせる方法にはいくつかの方法がある。例えばイオン交換法、限外濾過、電気透析を含む透析等である。最も簡便でコストのかからない方法は、ゾルを中和、濾過して得られたケーキを水洗後、中性の水性媒体に再分散することである。中和剤としては水酸化ナトリウムのようなアルカリを使用することもできるが、夾雑金属イオンを持ち込まないアンモニア水を使用するのが好ましい。再分散は、ボールミル、サンドミル、ホモミキサー、ペイントシェーカーなどの装置を用いて行うことができる。
【0011】
本発明において原料チタニアゾルの安定性および透明性を最大限保持する中性チタニアゾルを得るためには、被覆した分散酸化チタンの平均一次粒子径を50nm以下、平均分散粒子径を10〜200nmの範囲にとどめるのが好ましい。このためには被覆した酸化チタンケーキの再分散を徹底して実施することも勿論重要であるが、水和リン酸チタン化合物の被覆量および組成も重要である。好ましい被覆量は、原料のゾル中のTiO2 に換算した酸化チタンに対し、水和リン酸チタン化合物のチタンおよびリンをそれぞれTiO2 およびP2 O5 に換算し、この合計が7〜50重量%の範囲にある。当然のことながらこの被覆量が過少であれば効果が不十分であり、過多であれば粒子が巨大化して安定性および透明性を損なう。
【0012】
被覆水和リン酸チタン化合物の組成は、そのチタンとリンをそれぞれTiO2 とP2 O5 とに換算したTiO2 /P2 O5 の重量比が1.7〜7.0の範囲であることが好ましい。この比はTi/Pの原子比約1.3〜6.2に相当する。一方、公知のリン酸チタン、例えばTi(HPO4 )2 ,TiP2 O7 ,TiPO4 ,Ti(H2 PO4 )2 等はチタンとリンの原子比Ti/Pが1.0以下である。このような組成の相違も、チタン化合物およびリン酸化合物単独、および公知のリン酸チタン化合物には見られない独特の立体障害効果によって安定性に寄与しているものと考えられる。
【0013】
ここで透明性とは波長範囲400〜700nmにわたる透過率積分値(%T・nm)を意味し、具体的には厚さ1mm,光路長10mmの石英セルに固形分0.5重量%のゾルを入れて測定した時の透過率積分値(%T・nm)=透過率(%T)×(700−400nm)をもって評価される。本発明の中性チタニアゾルは、この透過率積分値の初期値および25℃2ケ月貯蔵後の値がいずれも4000%T・nm以上であり、高い透明性を保持している。
【0014】
また、ここでいう中性または中性域とは厳密な中性(pH7.0)のみならず、取扱い上および使用において接触する材料が特に耐酸性であることを要しない範囲のpH域、例えばpH7.0±1.5の領域を含むものと理解すべきである。
【0015】
本発明の中性チタニアゾルは、適当なバインダーへ添加して基材の表面に光触媒膜、半導体膜、紫外線カット膜などを形成するために使用することができる。その際ゾルはそのまま、適当な固形分濃度へ調節した後、または分散媒をメタノール、エタノール、イソプロパノール等の水混和性有機溶媒へ置換した後バインダーと混合することができ、さらに界面活性剤やエチレングリコール等の分散安定剤や他の慣用の添加剤を添加することも任意である。
【0016】
【実施例】
以下に限定を意図しない実施例によって本発明を具体的に説明する。
【0017】
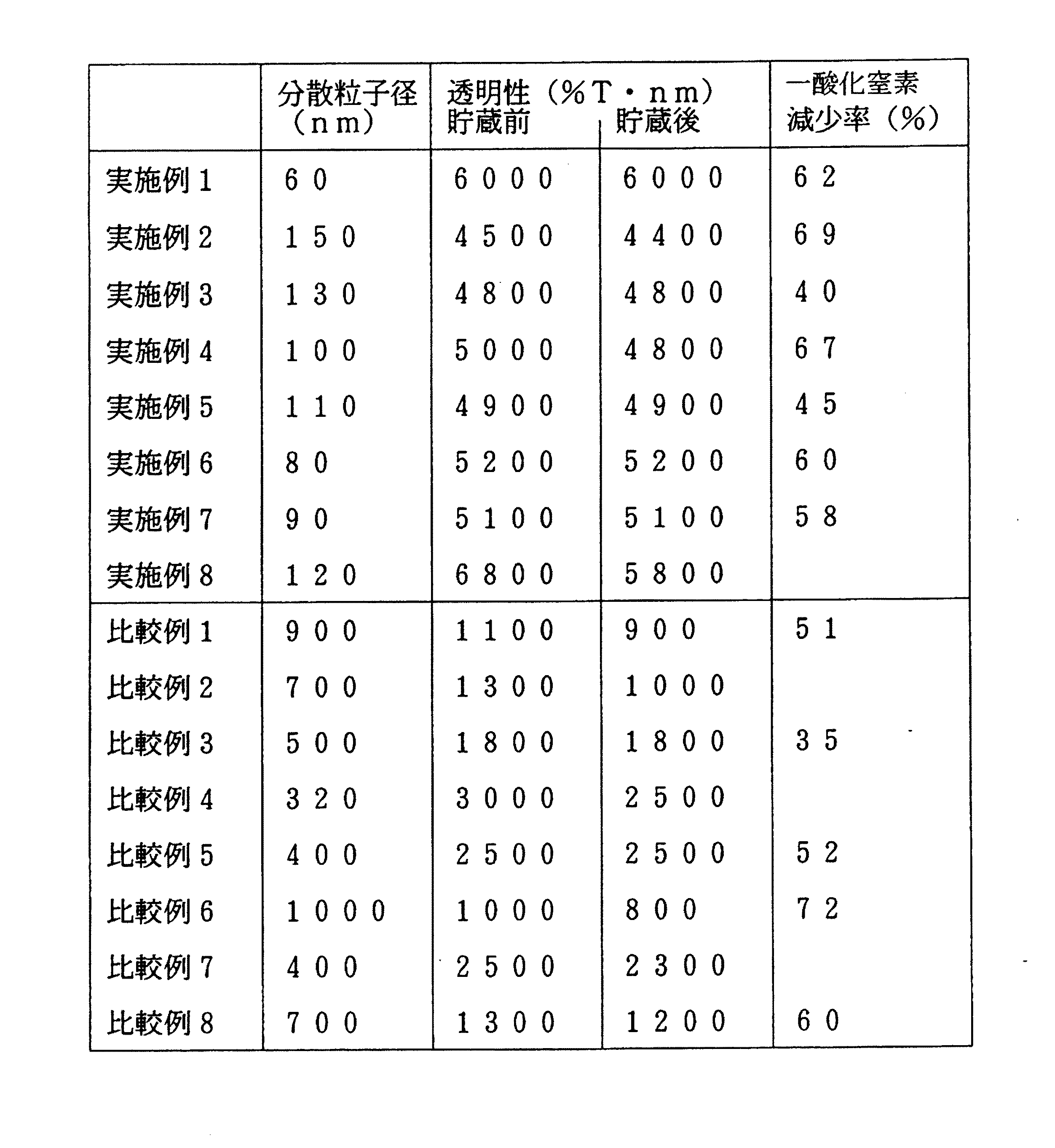
実施例1
解膠アナタース形チタニアゾルの製造
常法により硫酸チタニル水溶液を熱加水分解して得た含水酸化チタンスラリーをアンモニア水によりpH7に中和し、濾過、水洗して得たケーキを、ケーキの酸化チタン(TiO2 換算)に対して5重量%(HClに換算)の塩酸で解膠し、TiO2 に換算して32重量%濃度のアナタース形チタニアゾルを得た。このゾルの平均一次粒子径は7nm,平均分散粒子径は50nmであった。pH1.1
【0018】
中性被覆チタニアゾルの製造
上で得たアナタース形チタニアゾルをTiO2 換算で50g/Lの濃度に水で希釈し、この希釈ゾル2LへTiO2 換算15gの四塩化チタン水溶液を添加し、次いでP2 O5 に換算して5gの正リン酸を添加し、2時間反応させた。この反応液をアンモニア水でpH5.5に中和し、濾過して得たケーキを水洗後、固形分が25重量%になるように水を添加し、ペイントシェーカー(容器:400mLマヨネーズびん、媒体:直径1.5mmガラスビーズ30g,サンプル:100g,回転数:720rpm,分散時間:10分)を用いて再分散し、平均一次粒子径7nm,平均分散粒子径60nm,pH6.8の中性チタニアゾルを得た。
【0019】
実施例2
四塩化チタン水溶液の添加量をTiO2 換算で6.5g,正リン酸をP2 O5 換算で4gに変更した以外は実施例1に同じ。pH6.6、平均分散粒子径150nm
【0020】
実施例3
四塩化チタン水溶液の添加量をTiO2 換算で35g,正リン酸をP2 O5 換算で10gに変更した以外は実施例1に同じ。pH6.9、平均分散粒子径130nm
【0021】
実施例4
四塩化チタン水溶液の添加量をTiO2 換算で14g,正リン酸をP2 O5 換算で9gに変更した以外は実施例1に同じ。pH6.9、平均分散粒子径100nm
【0022】
実施例5
四塩化チタン水溶液の添加量をTiO2 換算で33g,正リン酸をP2 O5 換算で5gに変更した以外は実施例1に同じ。pH6.7、平均分散粒子径110nm
【0023】
実施例6
正リン酸の代わりにP2 O5 換算で5gのメタリン酸に変更した以外は実施例1に同じ。pH6.7、平均分散粒子径80nm
【0024】
実施例7
四塩化チタン水溶液の代わりにTiO2 換算で15gの硫酸チタニル水溶液に変更した以外は実施例1に同じ。pH6.5、平均分散粒子径90nm
【0025】
実施例8
解膠無定形チタニアゾルの製造
硫酸チタニル水溶液をアンモニア水でpH7に中和して加水分解し、得られた含水酸化チタンスラリーを濾過、水洗した。得られたケーキへ、酸化チタン(TiO2 換算)に対して25重量%(HCl換算)の塩酸を添加し、85℃で30分間加熱して解膠し、TiO2 に換算して50g/Lの無定形チタニアゾルを得た。このゾルの平均一次粒子径は5nm、平均分散粒子径は80nmであった。pH0.8
【0026】
中性被覆チタニアゾルの製造
上記で得た無定形チタニアゾル2Lへ、TiO2 換算15gの四塩化チタン水溶液を添加し、次いてP2 O5 換算5gの正リン酸を添加し、2時間反応させた。この反応液をアンモニア水でpH5.5に中和し、濾過して得たケーキを水洗後、固形分が10重量%になるように水に添加し、ペイントシェーカー(容器:400mLマヨネーズびん,媒体直径1.5mmガラスビーズ300g,サンプル:100g,回転数:720rpm,分散時間:10分)を用いて再分散し、pH6.5の中性チタニアゾルを得た。平均分散粒子径120nm
【0027】
比較例1
正リン酸を添加しなかったことを除き実施例1に同じ。生成物はゾルにならず、スラリーであった。pH6.7、平均分散粒子径900nm
【0028】
比較例2
四塩化チタン水溶液の添加量をTiO2 換算で4g、正リン酸をP2 O5 換算で2gに変更した以外は実施例1に同じ。生成物はゾルにならず、スラリーであった。pH6.9、平均粒子分散径700nm
【0029】
比較例3
四塩化チタン水溶液の添加量をTiO2 換算で10g、正リン酸をP2 O5 換算で20gに変更した以外は実施例1に同じ。生成物はゾルにならずスラリーであった。pH6.7、平均粒子分散径500nm
【0030】
比較例4
四塩化チタン水溶液の添加量をTiO2 換算で7g、正リン酸をP2 O5 換算で8gに変更した以外は実施例1に同じ。生成物はゾルにならずスラリーであった。pH6.8、平均分散粒子径320nm
【0031】
比較例5
四塩化チタン水溶液の添加量をTiO2 換算で25g、正リン酸をP2 O5 換算で3gに変更した以外は実施例1に同じ。生成物はゾルにならずスラリーであった。pH6.7、平均分散粒子径400nm
【0032】
比較例6
四塩化チタン水溶液および正リン酸を添加しなかったことを除き実施例1に同じ。生成物はゾルにならずスラリーであった。pH6.7、平均分散粒子径1000nm
【0033】
比較例7
四塩化チタン水溶液を添加しなかったことを除き実施例6に同じ。生成物はゾルにならずスラリー状であった。pH6.7、平均分散粒子径400nm
【0034】
比較例8
TiO2 換算で50g/Lの濃度に希釈した硫酸チタニル水溶液2Lを100℃で2時間熱加水分解してアナタース形の含水酸化チタンスラリーを得た。平均一次粒子径7nm,平均分散粒子径500nm
【0035】
このスラリーへTiO2 に換算して15gの四塩化チタン水溶液と、P2 O5 に換算して5gの正リン酸を添加し、2時間反応させ、アンモニア水でpH5.5に中和した後濾過し、得られたケーキを水洗した。このケーキを固形分が25重量%になるように水を添加し、実施例1と同じ条件でペイントシェーカーを使って再分散した。生成物はゾルにならず、スラリー状のままであった。pH6.7、平均粒子分散径700nm
【0036】
平均分散粒子径の測定
試料を固形分散濃度0.5重量%へ希釈し、粒度分布測定器(Microtrac UPA 日機装(株))で分散粒子径(体積平均径)を求めた。
【0037】
透明性の測定
試料を固形濃度0.5重量%へ希釈し、厚さ1mm,光路長10mmのセルへ入れ、分光光度計(U−3300(株)日立製作所)により透過率積分値(%T・nm)=透過率(%)×(700nm−400nm)を求めた。測定は初期値と、25℃×2ケ月貯蔵後の2回行った。
【0038】
光触媒活性の測定
試料をコロイダルシリカ(日産化学(株)製スノーテックス30)と固形分比1:1に混合し、全固形分5%になるように水で希釈し、7.6×2.6cmのガラス板にバーコーター#5を用いて塗布し、110℃で30分乾燥し、光触媒膜を形成した。この膜を400mlのベッセルに入れ、一酸化窒素を5ppm注入した後、水銀灯(3.7mW/cm2 )により5分間照射し、一酸化窒素の減少率を測定した。結果を表1に示す。
【0039】
【表1】
[0001]
[Industrial application fields]
The present invention relates to a titania sol that is transparent and stable in a neutral region and a method for producing the same. This sol is mixed with a binder and applied to a substrate, and can be used, for example, to form a photocatalytic film, a semiconductor film, or an ultraviolet cut film.
[0002]
[Prior art and its problems]
The titania sol is useful for forming a photocatalyst film, a semiconductor film, an ultraviolet cut film and the like on the surface of a substrate. A general method for obtaining a titania sol is a method of peptizing hydrous titanium oxide with a strong acid such as hydrochloric acid or nitric acid. However, since the sol obtained by this method contains a strong acid of a peptizer, there are many restrictions on handling and use as it is. For example, transportation, storage and other handling equipment needs to be acid resistant, and the substrate on which the membrane is formed is also limited to acid resistant materials.
[0003]
Since the titania sol peptized with a strong acid is stable only in a strongly acidic region, when the acid component is removed from the sol, the dispersed particles aggregate and do not form a sol. For this reason, a method has been proposed in which an organic or inorganic compound that acts as a stabilizer is added to a strongly acidic titania sol and then neutralized by alkali neutralization or ion exchange (Japanese Patent Laid-Open No. 63-123815, Japanese Patent Laid-Open No. 63). -215520, Japanese Patent Publication No. 7-33255). In this case, since a relatively large amount of stabilizer is contained in the sol, the photocatalytic activity and transparency of the film formed from the sol are impaired.
[0004]
Accordingly, an object of the present invention is to provide a titania sol that does not have such disadvantages, that is, is stable in a neutral region without adversely affecting the high catalytic activity and transparency inherent in the titania sol, and a method for producing the same. .
[0005]
[Solutions to the problem]
The present invention provides a neutral titania sol composed of hydrated titanium phosphate compound-coated peptized titanium oxide particles having transparency and dispersion stability in the neutral region.
[0006]
The present invention also provides a process for producing the neutral titania sol described above. In this method, a titania sol obtained by peptizing hydrous titanium oxide with a monobasic strong acid such as hydrochloric acid or nitric acid is used as a starting material, and this is mixed with an aqueous solution of a water-soluble titanium compound and a phosphoric acid compound. Remove the acid. As a result, a titania sol having transparency and dispersion stability in the neutral region can be obtained.
[0007]
[Specific implementation method]
A method for producing a titania sol by hydrolyzing an aqueous solution of titanyl sulfate by heating, etc., neutralizing, filtering and washing the precipitated hydrous titanium oxide with a strong acid such as hydrochloric acid or nitric acid. Well known. It is also well known that titanium oxide particles having a crystalline form of rutile, anatase or amorphous can be obtained by selecting reaction conditions. In the present invention, the crystal form of the dispersed titanium oxide particles is not limited, but the anatase form is preferable for the photocatalyst application.
[0008]
The titania sol thus obtained is strongly acidic (pH 3 or less), transparent, and generally has an average primary particle diameter of 50 nm or less and an average micelle diameter in the range of 10 to 150 nm. As mentioned earlier, this sol is unstable in the neutral region. Therefore, in the present invention, an aqueous solution of a water-soluble titanium compound and an aqueous solution of a phosphoric acid compound are mixed with a sol, and the titanium oxide dispersed particles are coated with a hydrated titanium phosphate compound generated by the reaction of the aqueous titanium compound and the phosphoric acid compound. It is considered that this coating shifts the isoelectric point of the sol to the acidic side, resulting in a stable sol in the neutral region.
[0009]
Examples of water-soluble titanium compounds that can be used include titanium tetrachloride and titanyl sulfate. Examples of phosphoric acid compounds include various phosphorus oxygen acids such as orthophosphoric acid and metaphosphoric acid, and water-soluble salts thereof. Orthophosphoric acid or metaphosphoric acid is preferred. These compounds react either directly or by neutralizing the sol to produce a hydrated titanium phosphate compound that coats the dispersed titanium oxide in the sol. The order of mixing is arbitrary as long as the reaction between the titanium compound and the phosphate compound does not occur in the absence of the dispersed titanium oxide particles. However, it is preferable to sequentially add an aqueous solution of a water-soluble titanium compound and an aqueous solution of a phosphoric acid compound to the titania sol.
[0010]
At this point, the sol is still highly acidic. There are several methods for shifting the sol pH to the neutral range. For example, ion exchange method, ultrafiltration, dialysis including electrodialysis, and the like. The simplest and least expensive method is to wash the cake obtained by neutralizing and filtering the sol and then redispersing it in a neutral aqueous medium. An alkali such as sodium hydroxide can be used as the neutralizing agent, but it is preferable to use ammonia water that does not bring in contaminating metal ions. The redispersion can be performed using an apparatus such as a ball mill, a sand mill, a homomixer, or a paint shaker.
[0011]
In order to obtain a neutral titania sol that maintains the stability and transparency of the raw material titania sol in the present invention, the average primary particle diameter of the coated dispersed titanium oxide is 50 nm or less and the average dispersed particle diameter is in the range of 10 to 200 nm. It is preferable to stay. For this purpose, it is of course important to thoroughly redisperse the coated titanium oxide cake, but the coating amount and composition of the hydrated titanium phosphate compound are also important. The preferred coating amount is that titanium oxide and phosphorus of the hydrated titanium phosphate compound are converted to TiO 2 and P 2 O 5 , respectively, with respect to titanium oxide converted to TiO 2 in the sol of the raw material, and this total is 7 to 50 wt. % Range. As a matter of course, if the coating amount is too small, the effect is insufficient, and if it is excessive, the particles become enormous and the stability and transparency are impaired.
[0012]
The composition of the coated hydrated titanium phosphate compound is such that the weight ratio of TiO 2 / P 2 O 5 in which the titanium and phosphorus are converted to TiO 2 and P 2 O 5 is 1.7 to 7.0. It is preferable. This ratio corresponds to an atomic ratio of Ti / P of about 1.3 to 6.2. On the other hand, known titanium phosphates such as Ti (HPO 4 ) 2 , TiP 2 O 7 , TiPO 4 , Ti (H 2 PO 4 ) 2 have an atomic ratio Ti / P of titanium and phosphorus of 1.0 or less. . Such a difference in composition is also considered to contribute to stability by a unique steric hindrance effect that is not found in the titanium compound and the phosphate compound alone and in the known titanium phosphate compound.
[0013]
Here, the transparency means an integral value of transmittance (% T · nm) over a wavelength range of 400 to 700 nm. Specifically, a sol having a solid content of 0.5% by weight in a quartz cell having a thickness of 1 mm and an optical path length of 10 mm. The transmittance integral value (% T · nm) when measured by putting in is evaluated as transmittance (% T) × (700−400 nm). In the neutral titania sol of the present invention, the initial value of the transmittance integral value and the value after storage at 25 ° C. for 2 months are both 4000% T · nm or more, and high transparency is maintained.
[0014]
Further, the neutral or neutral region here is not only strictly neutral (pH 7.0), but also a pH region in a range that does not require that the material to be contacted in handling and use is particularly acid-resistant, for example, It should be understood to include the region of pH 7.0 ± 1.5.
[0015]
The neutral titania sol of the present invention can be added to an appropriate binder and used to form a photocatalytic film, a semiconductor film, an ultraviolet cut film or the like on the surface of the substrate. In this case, the sol can be mixed with a binder after adjusting to a suitable solid content concentration or after replacing the dispersion medium with a water-miscible organic solvent such as methanol, ethanol, isopropanol, etc. It is also optional to add a dispersion stabilizer such as glycol and other conventional additives.
[0016]
【Example】
Hereinafter, the present invention will be described specifically by way of examples not intended to be limited.
[0017]
Example 1
Production of peptized anatase titania sol A hydrous titanium oxide slurry obtained by hydrolyzing a titanyl sulfate aqueous solution by a conventional method is neutralized to pH 7 with aqueous ammonia, filtered and washed with a cake. Peptide with 5% by weight (converted to HCl) hydrochloric acid with respect to titanium oxide (converted to TiO 2 ) to obtain anatase titania sol having a concentration of 32% by weight converted to TiO 2 . This sol had an average primary particle size of 7 nm and an average dispersed particle size of 50 nm. pH 1.1
[0018]
The anatase titania sol obtained by the manufacturing <br/> neutral coated titania sol diluted with water to a concentration of 50 g / L in terms of TiO 2, was added titanium tetrachloride aqueous solution of this in terms of TiO 2 15g to dilute sol 2L Subsequently, 5 g of normal phosphoric acid was added in terms of P 2 O 5 and reacted for 2 hours. The reaction solution was neutralized with ammonia water to pH 5.5, and the cake obtained by filtration was washed with water, water was added so that the solid content was 25% by weight, and a paint shaker (container: 400 mL mayonnaise bottle, medium) : Neutral titania sol having an average primary particle size of 7 nm, an average dispersed particle size of 60 nm, and a pH of 6.8. Got.
[0019]
Example 2
The same as Example 1, except that the amount of titanium tetrachloride aqueous solution added was changed to 6.5 g in terms of TiO 2 and orthophosphoric acid to 4 g in terms of P 2 O 5 . pH 6.6, average dispersed particle size 150 nm
[0020]
Example 3
The same as Example 1 except that the addition amount of the titanium tetrachloride aqueous solution was changed to 35 g in terms of TiO 2 and orthophosphoric acid was changed to 10 g in terms of P 2 O 5 . pH 6.9, average dispersed particle size 130 nm
[0021]
Example 4
The same as Example 1, except that the amount of titanium tetrachloride aqueous solution added was changed to 14 g in terms of TiO 2 and orthophosphoric acid was changed to 9 g in terms of P 2 O 5 . pH 6.9, average dispersed particle size 100 nm
[0022]
Example 5
The same as Example 1, except that the amount of titanium tetrachloride aqueous solution added was changed to 33 g in terms of TiO 2 , and orthophosphoric acid to 5 g in terms of P 2 O 5 . pH 6.7, average dispersed particle size 110 nm
[0023]
Example 6
The same as Example 1 except that it was changed to 5 g of metaphosphoric acid in terms of P 2 O 5 instead of regular phosphoric acid. pH 6.7, average dispersed particle size 80 nm
[0024]
Example 7
The same as Example 1 except that the titanium tetrachloride aqueous solution was replaced with 15 g titanyl sulfate aqueous solution in terms of TiO 2 . pH 6.5, average dispersed particle size 90 nm
[0025]
Example 8
Production of peptized amorphous titania sol An aqueous solution of titanyl sulfate was neutralized with aqueous ammonia to pH 7 and hydrolyzed, and the resulting hydrous titanium oxide slurry was filtered and washed with water. To the obtained cake, 25% by weight (in terms of HCl) of hydrochloric acid was added to titanium oxide (in terms of TiO 2 ), heated at 85 ° C. for 30 minutes to pept, and converted to TiO 2 of 50 g / L. Amorphous titania sol was obtained. This sol had an average primary particle size of 5 nm and an average dispersed particle size of 80 nm. pH 0.8
[0026]
Production of neutral coated titania sol 15 g of titanium tetrachloride aqueous solution in terms of TiO 2 was added to 2 L of the amorphous titania sol obtained above, and then 5 g of orthophosphoric acid in terms of P 2 O 5 was added. Reacted for hours. The reaction solution was neutralized with aqueous ammonia to pH 5.5, the cake obtained by filtration was washed with water, added to water so that the solid content was 10% by weight, and a paint shaker (container: 400 mL mayonnaise bottle, medium) Using a glass bead of 1.5 mm in diameter, 300 g, sample: 100 g, rotation speed: 720 rpm, dispersion time: 10 minutes, re-dispersion was performed to obtain a neutral titania sol having a pH of 6.5. Average dispersed particle size 120nm
[0027]
Comparative Example 1
Same as Example 1 except that no orthophosphoric acid was added. The product was not a sol but a slurry. pH 6.7, average dispersed particle size 900 nm
[0028]
Comparative Example 2
The same as Example 1, except that the amount of titanium tetrachloride aqueous solution added was changed to 4 g in terms of TiO 2 , and orthophosphoric acid was changed to 2 g in terms of P 2 O 5 . The product was not a sol but a slurry. pH 6.9, average particle dispersion diameter 700 nm
[0029]
Comparative Example 3
The same as Example 1 except that the addition amount of the titanium tetrachloride aqueous solution was changed to 10 g in terms of TiO 2 , and orthophosphoric acid was changed to 20 g in terms of P 2 O 5 . The product was not a sol but a slurry. pH 6.7, average particle dispersion diameter 500 nm
[0030]
Comparative Example 4
The same as Example 1 except that the addition amount of the aqueous titanium tetrachloride solution was changed to 7 g in terms of TiO 2 and the normal phosphoric acid was changed to 8 g in terms of P 2 O 5 . The product was not a sol but a slurry. pH 6.8, average dispersed particle size 320 nm
[0031]
Comparative Example 5
The same as Example 1 except that the addition amount of the titanium tetrachloride aqueous solution was changed to 25 g in terms of TiO 2 , and orthophosphoric acid was changed to 3 g in terms of P 2 O 5 . The product was not a sol but a slurry. pH 6.7, average dispersed particle size 400 nm
[0032]
Comparative Example 6
Same as Example 1 except that no titanium tetrachloride aqueous solution and orthophosphoric acid were added. The product was not a sol but a slurry. pH 6.7, average dispersed particle size 1000 nm
[0033]
Comparative Example 7
Same as Example 6 except that no titanium tetrachloride aqueous solution was added. The product was not a sol but a slurry. pH 6.7, average dispersed particle size 400 nm
[0034]
Comparative Example 8
2 L of titanyl sulfate aqueous solution diluted to a concentration of 50 g / L in terms of TiO 2 was thermally hydrolyzed at 100 ° C. for 2 hours to obtain anatase type hydrous titanium oxide slurry. Average primary particle size 7nm, average dispersed particle size 500nm
[0035]
After adding 15 g of titanium tetrachloride aqueous solution in terms of TiO 2 and 5 g of normal phosphoric acid in terms of P 2 O 5 to this slurry, the mixture was reacted for 2 hours and neutralized to pH 5.5 with aqueous ammonia. Filter and wash the resulting cake with water. Water was added to the cake so that the solid content was 25% by weight, and the cake was redispersed using a paint shaker under the same conditions as in Example 1. The product did not become a sol and remained in slurry form. pH 6.7, average particle dispersion diameter 700 nm
[0036]
Measurement of average dispersed particle diameter The sample was diluted to a solid dispersion concentration of 0.5 wt%, and the dispersed particle diameter (volume average diameter) was determined with a particle size distribution analyzer (Microtrac UPA Nikkiso Co., Ltd.).
[0037]
Measurement of transparency The sample was diluted to a solid concentration of 0.5% by weight, put into a cell having a thickness of 1 mm and an optical path length of 10 mm, and transmittance integration with a spectrophotometer (U-3300, Hitachi, Ltd.). Value (% T · nm) = transmittance (%) × (700 nm−400 nm) was determined. The measurement was performed twice after the initial value and storage at 25 ° C. × 2 months.
[0038]
Measurement of photocatalytic activity A sample was mixed with colloidal silica (Snowtex 30 manufactured by Nissan Chemical Co., Ltd.) at a solid content ratio of 1: 1, diluted with water to a total solid content of 5%, and 7. A 6 × 2.6 cm glass plate was coated using a bar coater # 5 and dried at 110 ° C. for 30 minutes to form a photocatalytic film. This film was placed in a 400 ml vessel, 5 ppm of nitric oxide was injected, and then irradiated with a mercury lamp (3.7 mW / cm 2 ) for 5 minutes, and the reduction rate of nitric oxide was measured. The results are shown in Table 1.
[0039]
[Table 1]
Claims (5)
Priority Applications (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| JP10191599A JP4026041B2 (en) | 1999-04-09 | 1999-04-09 | Neutral titania sol |
Applications Claiming Priority (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| JP10191599A JP4026041B2 (en) | 1999-04-09 | 1999-04-09 | Neutral titania sol |
Publications (2)
| Publication Number | Publication Date |
|---|---|
| JP2000290015A JP2000290015A (en) | 2000-10-17 |
| JP4026041B2 true JP4026041B2 (en) | 2007-12-26 |
Family
ID=14313215
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| JP10191599A Expired - Lifetime JP4026041B2 (en) | 1999-04-09 | 1999-04-09 | Neutral titania sol |
Country Status (1)
| Country | Link |
|---|---|
| JP (1) | JP4026041B2 (en) |
Families Citing this family (11)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US7378371B2 (en) | 2001-12-21 | 2008-05-27 | Show A Denko K.K. | Highly active photocatalyst particles, method of production therefor, and use thereof |
| US7414009B2 (en) | 2001-12-21 | 2008-08-19 | Showa Denko K.K. | Highly active photocatalyst particles, method of production therefor, and use thereof |
| EP1462169A4 (en) * | 2001-12-21 | 2006-06-28 | Showa Denko Kk | HIGHLY ACTIVE PHOTOCATALYTIC PARTICLE, PROCESS FOR PRODUCTION AND USE OF SUCH PARTICLE |
| JP4982208B2 (en) * | 2007-02-27 | 2012-07-25 | テイカ株式会社 | Photosensitizer composition for use in photodynamic therapy |
| JP4977051B2 (en) * | 2008-01-30 | 2012-07-18 | テイカ株式会社 | Stable titanium oxide dispersion in the neutral range |
| JP5754884B2 (en) * | 2009-12-28 | 2015-07-29 | 日揮触媒化成株式会社 | Phosphoric acid (excluding phosphoric acid salt) -treated metal oxide fine particles and production method thereof, coating solution for forming a transparent film containing the phosphoric acid (excluding phosphoric acid salt) -treated metal oxide fine particles, and transparent Substrate with coating |
| JP5458129B2 (en) * | 2012-03-09 | 2014-04-02 | テイカ株式会社 | Stable titanium oxide dispersion in the neutral range |
| EP3257816A1 (en) * | 2016-06-14 | 2017-12-20 | Kronos International, Inc. | Production of nano-particulate titanium dioxide |
| JP2022078384A (en) * | 2019-03-29 | 2022-05-25 | 日本電産株式会社 | Optical member and its manufacturing method |
| CN113583481B (en) * | 2021-08-27 | 2022-03-25 | 河南佰利联新材料有限公司 | Titanium dioxide composite material and preparation method thereof |
| CN118515309B (en) * | 2024-07-23 | 2024-09-13 | 铜陵纳源材料科技有限公司 | Recycling method of titanium phosphate product |
-
1999
- 1999-04-09 JP JP10191599A patent/JP4026041B2/en not_active Expired - Lifetime
Also Published As
| Publication number | Publication date |
|---|---|
| JP2000290015A (en) | 2000-10-17 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| JP2783417B2 (en) | Manufacturing method of rutile type titanium oxide sol | |
| CN102498067B (en) | Stable nano-titanium oxide sol and its manufacturing method | |
| JP3080162B2 (en) | Titanium oxide sol and method for producing the same | |
| US5403513A (en) | Titanium oxide sol and process for preparation thereof | |
| JP4026041B2 (en) | Neutral titania sol | |
| JPH0212503B2 (en) | ||
| JP5657197B2 (en) | Titanium oxide particles and method for producing the same | |
| JP2000053421A (en) | Titanium oxide sol and its preparation | |
| JPH10158015A (en) | Production of surface-treated titanium dioxide sol | |
| JP2001206720A (en) | Titanium oxide sol and manufacturing method thereof | |
| CN101448742B (en) | Solution of particles containing titanium dioxide and titanium peroxide complex and preparation thereof | |
| JP3238349B2 (en) | Titanium oxide ceramic paint excellent in hydrophilicity, photocatalytic property and translucency and method for producing the same | |
| JP2008031023A (en) | Zirconia sol and method for producing the same | |
| JPS63185820A (en) | Production of modified titania sol | |
| JPS6149250B2 (en) | ||
| JPS6351974B2 (en) | ||
| JP2005170687A (en) | Neutral titanium oxide sol and method for producing the same | |
| JP3978636B2 (en) | Coating composition for photocatalyst film formation | |
| JP4891021B2 (en) | Method for producing niobium oxide fine particles | |
| JP3950691B2 (en) | Colloidal dispersion of cerium compound containing cerium III, process for its production and use thereof | |
| JP2833820B2 (en) | Surface treatment method of titanium oxide | |
| JP2820251B2 (en) | Titanium oxide sol | |
| JP3490012B2 (en) | Method for producing crystalline titanium oxide particle dispersion liquid | |
| JPH0980203A (en) | Modified metal oxide sol and its production | |
| JPH0339017B2 (en) |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| A977 | Report on retrieval |
Free format text: JAPANESE INTERMEDIATE CODE: A971007 Effective date: 20060823 |
|
| A131 | Notification of reasons for refusal |
Free format text: JAPANESE INTERMEDIATE CODE: A131 Effective date: 20061114 |
|
| A521 | Request for written amendment filed |
Free format text: JAPANESE INTERMEDIATE CODE: A523 Effective date: 20061207 |
|
| A131 | Notification of reasons for refusal |
Free format text: JAPANESE INTERMEDIATE CODE: A131 Effective date: 20070206 |
|
| A521 | Request for written amendment filed |
Free format text: JAPANESE INTERMEDIATE CODE: A523 Effective date: 20070313 |
|
| A131 | Notification of reasons for refusal |
Free format text: JAPANESE INTERMEDIATE CODE: A131 Effective date: 20070515 |
|
| A521 | Request for written amendment filed |
Free format text: JAPANESE INTERMEDIATE CODE: A523 Effective date: 20070521 |
|
| TRDD | Decision of grant or rejection written | ||
| A01 | Written decision to grant a patent or to grant a registration (utility model) |
Free format text: JAPANESE INTERMEDIATE CODE: A01 Effective date: 20070925 |
|
| A61 | First payment of annual fees (during grant procedure) |
Free format text: JAPANESE INTERMEDIATE CODE: A61 Effective date: 20070926 |
|
| FPAY | Renewal fee payment (event date is renewal date of database) |
Free format text: PAYMENT UNTIL: 20101019 Year of fee payment: 3 |
|
| R150 | Certificate of patent or registration of utility model |
Free format text: JAPANESE INTERMEDIATE CODE: R150 |
|
| FPAY | Renewal fee payment (event date is renewal date of database) |
Free format text: PAYMENT UNTIL: 20131019 Year of fee payment: 6 |
|
| R250 | Receipt of annual fees |
Free format text: JAPANESE INTERMEDIATE CODE: R250 |
|
| R250 | Receipt of annual fees |
Free format text: JAPANESE INTERMEDIATE CODE: R250 |
|
| EXPY | Cancellation because of completion of term |