JP2021517454A - 癌治療のためのアルギナーゼ抑制 - Google Patents
癌治療のためのアルギナーゼ抑制 Download PDFInfo
- Publication number
- JP2021517454A JP2021517454A JP2020541437A JP2020541437A JP2021517454A JP 2021517454 A JP2021517454 A JP 2021517454A JP 2020541437 A JP2020541437 A JP 2020541437A JP 2020541437 A JP2020541437 A JP 2020541437A JP 2021517454 A JP2021517454 A JP 2021517454A
- Authority
- JP
- Japan
- Prior art keywords
- cells
- arginase
- immune
- expression
- cell
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Pending
Links
- 206010028980 Neoplasm Diseases 0.000 title claims abstract description 167
- 108700024123 Arginases Proteins 0.000 title claims abstract description 129
- 102000004452 Arginase Human genes 0.000 title claims abstract description 128
- 201000011510 cancer Diseases 0.000 title claims abstract description 78
- 238000011282 treatment Methods 0.000 title claims description 49
- 230000001629 suppression Effects 0.000 title 1
- 210000002865 immune cell Anatomy 0.000 claims abstract description 143
- 210000001744 T-lymphocyte Anatomy 0.000 claims abstract description 102
- 230000014509 gene expression Effects 0.000 claims abstract description 95
- 230000000694 effects Effects 0.000 claims abstract description 91
- 238000000034 method Methods 0.000 claims abstract description 59
- 230000001771 impaired effect Effects 0.000 claims abstract description 50
- 108010019670 Chimeric Antigen Receptors Proteins 0.000 claims abstract description 44
- 108090000623 proteins and genes Proteins 0.000 claims abstract description 34
- 102000037982 Immune checkpoint proteins Human genes 0.000 claims abstract description 21
- 108091008036 Immune checkpoint proteins Proteins 0.000 claims abstract description 21
- 210000004027 cell Anatomy 0.000 claims description 143
- 102100030356 Arginase-2, mitochondrial Human genes 0.000 claims description 28
- 101000792835 Homo sapiens Arginase-2, mitochondrial Proteins 0.000 claims description 27
- 238000012546 transfer Methods 0.000 claims description 19
- 239000000427 antigen Substances 0.000 claims description 17
- 108091007433 antigens Proteins 0.000 claims description 17
- 102000036639 antigens Human genes 0.000 claims description 17
- 239000013598 vector Substances 0.000 claims description 16
- 108091008874 T cell receptors Proteins 0.000 claims description 14
- 102000016266 T-Cell Antigen Receptors Human genes 0.000 claims description 14
- 108020004999 messenger RNA Proteins 0.000 claims description 14
- 230000011664 signaling Effects 0.000 claims description 13
- 102100038282 V-type immunoglobulin domain-containing suppressor of T-cell activation Human genes 0.000 claims description 12
- 108020004707 nucleic acids Proteins 0.000 claims description 12
- 102000039446 nucleic acids Human genes 0.000 claims description 12
- 150000007523 nucleic acids Chemical class 0.000 claims description 12
- 102100040678 Programmed cell death protein 1 Human genes 0.000 claims description 11
- 101000666896 Homo sapiens V-type immunoglobulin domain-containing suppressor of T-cell activation Proteins 0.000 claims description 10
- 102100038929 V-set domain-containing T-cell activation inhibitor 1 Human genes 0.000 claims description 10
- 239000000203 mixture Substances 0.000 claims description 9
- 101001021491 Homo sapiens HERV-H LTR-associating protein 2 Proteins 0.000 claims description 8
- 230000001093 anti-cancer Effects 0.000 claims description 8
- 239000002773 nucleotide Substances 0.000 claims description 7
- 125000003729 nucleotide group Chemical group 0.000 claims description 7
- 102100035943 HERV-H LTR-associating protein 2 Human genes 0.000 claims description 6
- 101000955999 Homo sapiens V-set domain-containing T-cell activation inhibitor 1 Proteins 0.000 claims description 6
- 230000009261 transgenic effect Effects 0.000 claims description 5
- 102100022464 5'-nucleotidase Human genes 0.000 claims description 4
- 102000000506 Adenosine A2 Receptors Human genes 0.000 claims description 4
- 108010041368 Adenosine A2 Receptors Proteins 0.000 claims description 4
- 108010074708 B7-H1 Antigen Proteins 0.000 claims description 4
- 102100038078 CD276 antigen Human genes 0.000 claims description 4
- 101710185679 CD276 antigen Proteins 0.000 claims description 4
- 102100039498 Cytotoxic T-lymphocyte protein 4 Human genes 0.000 claims description 4
- 102100029722 Ectonucleoside triphosphate diphosphohydrolase 1 Human genes 0.000 claims description 4
- -1 Gi24 Proteins 0.000 claims description 4
- 101000678236 Homo sapiens 5'-nucleotidase Proteins 0.000 claims description 4
- 101000889276 Homo sapiens Cytotoxic T-lymphocyte protein 4 Proteins 0.000 claims description 4
- 101001012447 Homo sapiens Ectonucleoside triphosphate diphosphohydrolase 1 Proteins 0.000 claims description 4
- 101000868279 Homo sapiens Leukocyte surface antigen CD47 Proteins 0.000 claims description 4
- 101000831007 Homo sapiens T-cell immunoreceptor with Ig and ITIM domains Proteins 0.000 claims description 4
- 101000596234 Homo sapiens T-cell surface protein tactile Proteins 0.000 claims description 4
- 102000017578 LAG3 Human genes 0.000 claims description 4
- 101150030213 Lag3 gene Proteins 0.000 claims description 4
- 102100032913 Leukocyte surface antigen CD47 Human genes 0.000 claims description 4
- 102100024834 T-cell immunoreceptor with Ig and ITIM domains Human genes 0.000 claims description 4
- 102100035268 T-cell surface protein tactile Human genes 0.000 claims description 4
- 108091023040 Transcription factor Proteins 0.000 claims description 4
- 102000040945 Transcription factor Human genes 0.000 claims description 4
- 108010079206 V-Set Domain-Containing T-Cell Activation Inhibitor 1 Proteins 0.000 claims description 4
- IJJVMEJXYNJXOJ-UHFFFAOYSA-N fluquinconazole Chemical compound C=1C=C(Cl)C=C(Cl)C=1N1C(=O)C2=CC(F)=CC=C2N=C1N1C=NC=N1 IJJVMEJXYNJXOJ-UHFFFAOYSA-N 0.000 claims description 4
- 210000000822 natural killer cell Anatomy 0.000 claims description 4
- 206010025323 Lymphomas Diseases 0.000 claims description 3
- 102100024216 Programmed cell death 1 ligand 1 Human genes 0.000 claims description 3
- 210000004964 innate lymphoid cell Anatomy 0.000 claims description 3
- 208000032839 leukemia Diseases 0.000 claims description 3
- 108091058539 C10orf54 Proteins 0.000 claims description 2
- 229940124650 anti-cancer therapies Drugs 0.000 claims description 2
- 238000011319 anticancer therapy Methods 0.000 claims description 2
- 230000005754 cellular signaling Effects 0.000 claims description 2
- 101000834898 Homo sapiens Alpha-synuclein Proteins 0.000 claims 2
- 101000611936 Homo sapiens Programmed cell death protein 1 Proteins 0.000 claims 2
- 101000652359 Homo sapiens Spermatogenesis-associated protein 2 Proteins 0.000 claims 2
- 210000004544 dc2 Anatomy 0.000 claims 1
- 238000002560 therapeutic procedure Methods 0.000 abstract description 11
- 230000005764 inhibitory process Effects 0.000 abstract description 10
- 230000035772 mutation Effects 0.000 abstract description 10
- 238000012228 RNA interference-mediated gene silencing Methods 0.000 abstract description 6
- 230000009368 gene silencing by RNA Effects 0.000 abstract description 6
- 230000037361 pathway Effects 0.000 abstract description 6
- 238000012217 deletion Methods 0.000 abstract description 5
- 230000037430 deletion Effects 0.000 abstract description 5
- 230000001404 mediated effect Effects 0.000 abstract description 5
- 230000000903 blocking effect Effects 0.000 abstract description 3
- 229940080328 Arginase inhibitor Drugs 0.000 abstract description 2
- 238000003776 cleavage reaction Methods 0.000 abstract description 2
- 201000005787 hematologic cancer Diseases 0.000 abstract description 2
- 230000002489 hematologic effect Effects 0.000 abstract description 2
- 208000024200 hematopoietic and lymphoid system neoplasm Diseases 0.000 abstract description 2
- 230000007017 scission Effects 0.000 abstract description 2
- 101150026173 ARG2 gene Proteins 0.000 description 84
- 241000699670 Mus sp. Species 0.000 description 67
- 230000004614 tumor growth Effects 0.000 description 35
- 210000001185 bone marrow Anatomy 0.000 description 24
- 210000003171 tumor-infiltrating lymphocyte Anatomy 0.000 description 22
- 108091032973 (ribonucleotides)n+m Proteins 0.000 description 18
- 230000004913 activation Effects 0.000 description 18
- 230000007812 deficiency Effects 0.000 description 18
- 238000000338 in vitro Methods 0.000 description 18
- 230000002401 inhibitory effect Effects 0.000 description 17
- 239000004055 small Interfering RNA Substances 0.000 description 16
- 230000035899 viability Effects 0.000 description 16
- 241001465754 Metazoa Species 0.000 description 15
- 239000003112 inhibitor Substances 0.000 description 15
- 230000001965 increasing effect Effects 0.000 description 13
- 230000004083 survival effect Effects 0.000 description 13
- 108091033773 MiR-155 Proteins 0.000 description 12
- 238000002474 experimental method Methods 0.000 description 12
- 230000002452 interceptive effect Effects 0.000 description 12
- 102100021723 Arginase-1 Human genes 0.000 description 11
- 101710129000 Arginase-1 Proteins 0.000 description 11
- 102000004169 proteins and genes Human genes 0.000 description 11
- 108020004459 Small interfering RNA Proteins 0.000 description 10
- 230000035755 proliferation Effects 0.000 description 10
- 235000018102 proteins Nutrition 0.000 description 10
- 101710089372 Programmed cell death protein 1 Proteins 0.000 description 9
- 230000001506 immunosuppresive effect Effects 0.000 description 9
- 210000004881 tumor cell Anatomy 0.000 description 9
- ODKSFYDXXFIFQN-BYPYZUCNSA-N L-arginine Chemical compound OC(=O)[C@@H](N)CCCN=C(N)N ODKSFYDXXFIFQN-BYPYZUCNSA-N 0.000 description 8
- 229930064664 L-arginine Natural products 0.000 description 8
- 235000014852 L-arginine Nutrition 0.000 description 8
- 108010058846 Ovalbumin Proteins 0.000 description 8
- 230000002950 deficient Effects 0.000 description 8
- 238000004519 manufacturing process Methods 0.000 description 8
- 230000007246 mechanism Effects 0.000 description 8
- 229940092253 ovalbumin Drugs 0.000 description 8
- 102100025137 Early activation antigen CD69 Human genes 0.000 description 7
- 101000934374 Homo sapiens Early activation antigen CD69 Proteins 0.000 description 7
- 108091027967 Small hairpin RNA Proteins 0.000 description 7
- 239000003795 chemical substances by application Substances 0.000 description 7
- 230000006870 function Effects 0.000 description 7
- 230000003834 intracellular effect Effects 0.000 description 7
- 230000002829 reductive effect Effects 0.000 description 7
- 108091007741 Chimeric antigen receptor T cells Proteins 0.000 description 6
- 241000699666 Mus <mouse, genus> Species 0.000 description 6
- 102000008299 Nitric Oxide Synthase Human genes 0.000 description 6
- 108010021487 Nitric Oxide Synthase Proteins 0.000 description 6
- 210000004443 dendritic cell Anatomy 0.000 description 6
- 238000001727 in vivo Methods 0.000 description 6
- 230000001939 inductive effect Effects 0.000 description 6
- 150000003384 small molecules Chemical class 0.000 description 6
- 108700011259 MicroRNAs Proteins 0.000 description 5
- 230000006044 T cell activation Effects 0.000 description 5
- 230000005809 anti-tumor immunity Effects 0.000 description 5
- 230000033228 biological regulation Effects 0.000 description 5
- 238000002619 cancer immunotherapy Methods 0.000 description 5
- 238000002659 cell therapy Methods 0.000 description 5
- 230000030279 gene silencing Effects 0.000 description 5
- 230000028993 immune response Effects 0.000 description 5
- 238000009169 immunotherapy Methods 0.000 description 5
- 239000007924 injection Substances 0.000 description 5
- 238000002347 injection Methods 0.000 description 5
- 239000007928 intraperitoneal injection Substances 0.000 description 5
- 239000002679 microRNA Substances 0.000 description 5
- 230000001225 therapeutic effect Effects 0.000 description 5
- 108091033409 CRISPR Proteins 0.000 description 4
- 208000002250 Hematologic Neoplasms Diseases 0.000 description 4
- AHLPHDHHMVZTML-BYPYZUCNSA-N L-Ornithine Chemical compound NCCC[C@H](N)C(O)=O AHLPHDHHMVZTML-BYPYZUCNSA-N 0.000 description 4
- 239000006146 Roswell Park Memorial Institute medium Substances 0.000 description 4
- 230000005867 T cell response Effects 0.000 description 4
- 230000005975 antitumor immune response Effects 0.000 description 4
- 150000001875 compounds Chemical class 0.000 description 4
- 230000004048 modification Effects 0.000 description 4
- 238000012986 modification Methods 0.000 description 4
- 230000001737 promoting effect Effects 0.000 description 4
- 238000010186 staining Methods 0.000 description 4
- 230000002195 synergetic effect Effects 0.000 description 4
- 210000001519 tissue Anatomy 0.000 description 4
- 102100024222 B-lymphocyte antigen CD19 Human genes 0.000 description 3
- 101000980825 Homo sapiens B-lymphocyte antigen CD19 Proteins 0.000 description 3
- 101710120463 Prostate stem cell antigen Proteins 0.000 description 3
- 102100036735 Prostate stem cell antigen Human genes 0.000 description 3
- 230000006052 T cell proliferation Effects 0.000 description 3
- 230000000259 anti-tumor effect Effects 0.000 description 3
- 230000006907 apoptotic process Effects 0.000 description 3
- 210000003719 b-lymphocyte Anatomy 0.000 description 3
- 238000009172 cell transfer therapy Methods 0.000 description 3
- 230000002301 combined effect Effects 0.000 description 3
- 230000003247 decreasing effect Effects 0.000 description 3
- 238000011161 development Methods 0.000 description 3
- 208000037265 diseases, disorders, signs and symptoms Diseases 0.000 description 3
- 208000035475 disorder Diseases 0.000 description 3
- 238000000684 flow cytometry Methods 0.000 description 3
- 238000010362 genome editing Methods 0.000 description 3
- 230000012010 growth Effects 0.000 description 3
- 210000000987 immune system Anatomy 0.000 description 3
- 230000001976 improved effect Effects 0.000 description 3
- 210000001165 lymph node Anatomy 0.000 description 3
- 210000002540 macrophage Anatomy 0.000 description 3
- 210000003819 peripheral blood mononuclear cell Anatomy 0.000 description 3
- 239000013612 plasmid Substances 0.000 description 3
- 108010004131 poly(beta-D-mannuronate) lyase Proteins 0.000 description 3
- 230000008569 process Effects 0.000 description 3
- 102000005962 receptors Human genes 0.000 description 3
- 108020003175 receptors Proteins 0.000 description 3
- 230000009467 reduction Effects 0.000 description 3
- 230000001105 regulatory effect Effects 0.000 description 3
- 210000000130 stem cell Anatomy 0.000 description 3
- VDABVNMGKGUPEY-UHFFFAOYSA-N 6-carboxyfluorescein succinimidyl ester Chemical compound C=1C(O)=CC=C2C=1OC1=CC(O)=CC=C1C2(C1=C2)OC(=O)C1=CC=C2C(=O)ON1C(=O)CCC1=O VDABVNMGKGUPEY-UHFFFAOYSA-N 0.000 description 2
- QGZKDVFQNNGYKY-UHFFFAOYSA-N Ammonia Chemical compound N QGZKDVFQNNGYKY-UHFFFAOYSA-N 0.000 description 2
- IJGRMHOSHXDMSA-UHFFFAOYSA-N Atomic nitrogen Chemical compound N#N IJGRMHOSHXDMSA-UHFFFAOYSA-N 0.000 description 2
- 238000010354 CRISPR gene editing Methods 0.000 description 2
- 108091026890 Coding region Proteins 0.000 description 2
- 102000004190 Enzymes Human genes 0.000 description 2
- 108090000790 Enzymes Proteins 0.000 description 2
- 102000004388 Interleukin-4 Human genes 0.000 description 2
- 108090000978 Interleukin-4 Proteins 0.000 description 2
- 102000010787 Interleukin-4 Receptors Human genes 0.000 description 2
- 108010038486 Interleukin-4 Receptors Proteins 0.000 description 2
- ONIBWKKTOPOVIA-BYPYZUCNSA-N L-Proline Chemical compound OC(=O)[C@@H]1CCCN1 ONIBWKKTOPOVIA-BYPYZUCNSA-N 0.000 description 2
- 102000018697 Membrane Proteins Human genes 0.000 description 2
- 108010052285 Membrane Proteins Proteins 0.000 description 2
- 108091028043 Nucleic acid sequence Proteins 0.000 description 2
- AHLPHDHHMVZTML-UHFFFAOYSA-N Orn-delta-NH2 Natural products NCCCC(N)C(O)=O AHLPHDHHMVZTML-UHFFFAOYSA-N 0.000 description 2
- 241000209094 Oryza Species 0.000 description 2
- 235000007164 Oryza sativa Nutrition 0.000 description 2
- 206010061535 Ovarian neoplasm Diseases 0.000 description 2
- ONIBWKKTOPOVIA-UHFFFAOYSA-N Proline Natural products OC(=O)C1CCCN1 ONIBWKKTOPOVIA-UHFFFAOYSA-N 0.000 description 2
- 108010034634 Repressor Proteins Proteins 0.000 description 2
- 102000009661 Repressor Proteins Human genes 0.000 description 2
- 230000000735 allogeneic effect Effects 0.000 description 2
- 238000013459 approach Methods 0.000 description 2
- 230000008901 benefit Effects 0.000 description 2
- 239000012830 cancer therapeutic Substances 0.000 description 2
- 230000030570 cellular localization Effects 0.000 description 2
- 230000000295 complement effect Effects 0.000 description 2
- 230000004069 differentiation Effects 0.000 description 2
- 230000008030 elimination Effects 0.000 description 2
- 238000003379 elimination reaction Methods 0.000 description 2
- 102000034356 gene-regulatory proteins Human genes 0.000 description 2
- 108091006104 gene-regulatory proteins Proteins 0.000 description 2
- 230000003394 haemopoietic effect Effects 0.000 description 2
- 238000003306 harvesting Methods 0.000 description 2
- 230000003053 immunization Effects 0.000 description 2
- 238000002649 immunization Methods 0.000 description 2
- 239000002955 immunomodulating agent Substances 0.000 description 2
- 230000004941 influx Effects 0.000 description 2
- 230000007774 longterm Effects 0.000 description 2
- 230000037353 metabolic pathway Effects 0.000 description 2
- 230000004060 metabolic process Effects 0.000 description 2
- 210000003470 mitochondria Anatomy 0.000 description 2
- 229960003104 ornithine Drugs 0.000 description 2
- 239000008194 pharmaceutical composition Substances 0.000 description 2
- 210000001778 pluripotent stem cell Anatomy 0.000 description 2
- 229920000768 polyamine Polymers 0.000 description 2
- 230000003389 potentiating effect Effects 0.000 description 2
- 108090000765 processed proteins & peptides Proteins 0.000 description 2
- 239000000700 radioactive tracer Substances 0.000 description 2
- 210000003289 regulatory T cell Anatomy 0.000 description 2
- 230000002441 reversible effect Effects 0.000 description 2
- 235000009566 rice Nutrition 0.000 description 2
- 238000002741 site-directed mutagenesis Methods 0.000 description 2
- 210000004988 splenocyte Anatomy 0.000 description 2
- 208000024891 symptom Diseases 0.000 description 2
- 230000002992 thymic effect Effects 0.000 description 2
- 238000013518 transcription Methods 0.000 description 2
- 230000035897 transcription Effects 0.000 description 2
- 230000001052 transient effect Effects 0.000 description 2
- 238000013519 translation Methods 0.000 description 2
- 101100423701 Arabidopsis thaliana OVA1 gene Proteins 0.000 description 1
- 101710186578 Arginase-2, mitochondrial Proteins 0.000 description 1
- 239000004475 Arginine Substances 0.000 description 1
- 201000001320 Atherosclerosis Diseases 0.000 description 1
- 208000023275 Autoimmune disease Diseases 0.000 description 1
- 238000011357 CAR T-cell therapy Methods 0.000 description 1
- 238000011523 CAR-T cell immunotherapy Methods 0.000 description 1
- 208000024172 Cardiovascular disease Diseases 0.000 description 1
- 206010009944 Colon cancer Diseases 0.000 description 1
- 108090000695 Cytokines Proteins 0.000 description 1
- 102000004127 Cytokines Human genes 0.000 description 1
- 208000001382 Experimental Melanoma Diseases 0.000 description 1
- 208000018522 Gastrointestinal disease Diseases 0.000 description 1
- 208000032612 Glial tumor Diseases 0.000 description 1
- 206010018338 Glioma Diseases 0.000 description 1
- 102100041003 Glutamate carboxypeptidase 2 Human genes 0.000 description 1
- 108010009202 Growth Factor Receptors Proteins 0.000 description 1
- 102000009465 Growth Factor Receptors Human genes 0.000 description 1
- 241000282412 Homo Species 0.000 description 1
- 101100005713 Homo sapiens CD4 gene Proteins 0.000 description 1
- 101000892862 Homo sapiens Glutamate carboxypeptidase 2 Proteins 0.000 description 1
- 101000623901 Homo sapiens Mucin-16 Proteins 0.000 description 1
- 101000914514 Homo sapiens T-cell-specific surface glycoprotein CD28 Proteins 0.000 description 1
- 102000004867 Hydro-Lyases Human genes 0.000 description 1
- 108090001042 Hydro-Lyases Proteins 0.000 description 1
- 206010021143 Hypoxia Diseases 0.000 description 1
- 206010062016 Immunosuppression Diseases 0.000 description 1
- 102000014150 Interferons Human genes 0.000 description 1
- 108010050904 Interferons Proteins 0.000 description 1
- 102000010782 Interleukin-7 Receptors Human genes 0.000 description 1
- 108010038498 Interleukin-7 Receptors Proteins 0.000 description 1
- 208000008839 Kidney Neoplasms Diseases 0.000 description 1
- 238000012218 Kunkel's method Methods 0.000 description 1
- 208000019693 Lung disease Diseases 0.000 description 1
- 241000124008 Mammalia Species 0.000 description 1
- 206010027476 Metastases Diseases 0.000 description 1
- 102100023123 Mucin-16 Human genes 0.000 description 1
- 101100260702 Mus musculus Tinagl1 gene Proteins 0.000 description 1
- 206010061309 Neoplasm progression Diseases 0.000 description 1
- 206010033128 Ovarian cancer Diseases 0.000 description 1
- 238000012220 PCR site-directed mutagenesis Methods 0.000 description 1
- 208000026149 Primary peritoneal carcinoma Diseases 0.000 description 1
- 206010060862 Prostate cancer Diseases 0.000 description 1
- 102000007066 Prostate-Specific Antigen Human genes 0.000 description 1
- 108010072866 Prostate-Specific Antigen Proteins 0.000 description 1
- 208000000236 Prostatic Neoplasms Diseases 0.000 description 1
- 206010038389 Renal cancer Diseases 0.000 description 1
- 206010039085 Rhinitis allergic Diseases 0.000 description 1
- FAPWRFPIFSIZLT-UHFFFAOYSA-M Sodium chloride Chemical compound [Na+].[Cl-] FAPWRFPIFSIZLT-UHFFFAOYSA-M 0.000 description 1
- 230000024932 T cell mediated immunity Effects 0.000 description 1
- 102100027213 T-cell-specific surface glycoprotein CD28 Human genes 0.000 description 1
- QJJXYPPXXYFBGM-LFZNUXCKSA-N Tacrolimus Chemical class C1C[C@@H](O)[C@H](OC)C[C@@H]1\C=C(/C)[C@@H]1[C@H](C)[C@@H](O)CC(=O)[C@H](CC=C)/C=C(C)/C[C@H](C)C[C@H](OC)[C@H]([C@H](C[C@H]2C)OC)O[C@@]2(O)C(=O)C(=O)N2CCCC[C@H]2C(=O)O1 QJJXYPPXXYFBGM-LFZNUXCKSA-N 0.000 description 1
- 108700019146 Transgenes Proteins 0.000 description 1
- XSQUKJJJFZCRTK-UHFFFAOYSA-N Urea Chemical compound NC(N)=O XSQUKJJJFZCRTK-UHFFFAOYSA-N 0.000 description 1
- GQLCLPLEEOUJQC-ZTQDTCGGSA-N [(1r)-3-(3,4-dimethoxyphenyl)-1-[3-[2-[2-[[2-[3-[(1r)-3-(3,4-dimethoxyphenyl)-1-[(2s)-1-[(2s)-2-(3,4,5-trimethoxyphenyl)butanoyl]piperidine-2-carbonyl]oxypropyl]phenoxy]acetyl]amino]ethylamino]-2-oxoethoxy]phenyl]propyl] (2s)-1-[(2s)-2-(3,4,5-trimethoxyph Chemical compound C([C@@H](OC(=O)[C@@H]1CCCCN1C(=O)[C@@H](CC)C=1C=C(OC)C(OC)=C(OC)C=1)C=1C=C(OCC(=O)NCCNC(=O)COC=2C=C(C=CC=2)[C@@H](CCC=2C=C(OC)C(OC)=CC=2)OC(=O)[C@H]2N(CCCC2)C(=O)[C@@H](CC)C=2C=C(OC)C(OC)=C(OC)C=2)C=CC=1)CC1=CC=C(OC)C(OC)=C1 GQLCLPLEEOUJQC-ZTQDTCGGSA-N 0.000 description 1
- 238000011467 adoptive cell therapy Methods 0.000 description 1
- 201000009961 allergic asthma Diseases 0.000 description 1
- 230000000172 allergic effect Effects 0.000 description 1
- 201000010105 allergic rhinitis Diseases 0.000 description 1
- 229910021529 ammonia Inorganic materials 0.000 description 1
- 230000003110 anti-inflammatory effect Effects 0.000 description 1
- 230000006023 anti-tumor response Effects 0.000 description 1
- 238000011394 anticancer treatment Methods 0.000 description 1
- 239000002246 antineoplastic agent Substances 0.000 description 1
- 101150088826 arg1 gene Proteins 0.000 description 1
- ODKSFYDXXFIFQN-UHFFFAOYSA-N arginine Natural products OC(=O)C(N)CCCNC(N)=N ODKSFYDXXFIFQN-UHFFFAOYSA-N 0.000 description 1
- 235000009697 arginine Nutrition 0.000 description 1
- 238000003556 assay Methods 0.000 description 1
- 208000006673 asthma Diseases 0.000 description 1
- 238000005842 biochemical reaction Methods 0.000 description 1
- 230000015572 biosynthetic process Effects 0.000 description 1
- 210000004369 blood Anatomy 0.000 description 1
- 239000008280 blood Substances 0.000 description 1
- 239000004202 carbamide Substances 0.000 description 1
- 238000012219 cassette mutagenesis Methods 0.000 description 1
- 230000015556 catabolic process Effects 0.000 description 1
- 230000003197 catalytic effect Effects 0.000 description 1
- 230000022534 cell killing Effects 0.000 description 1
- 230000004663 cell proliferation Effects 0.000 description 1
- 239000006285 cell suspension Substances 0.000 description 1
- 238000006243 chemical reaction Methods 0.000 description 1
- 238000005352 clarification Methods 0.000 description 1
- 208000029742 colonic neoplasm Diseases 0.000 description 1
- 230000001143 conditioned effect Effects 0.000 description 1
- 230000016396 cytokine production Effects 0.000 description 1
- 230000001086 cytosolic effect Effects 0.000 description 1
- 230000001472 cytotoxic effect Effects 0.000 description 1
- 230000003013 cytotoxicity Effects 0.000 description 1
- 231100000135 cytotoxicity Toxicity 0.000 description 1
- 230000006735 deficit Effects 0.000 description 1
- 210000001787 dendrite Anatomy 0.000 description 1
- 230000000779 depleting effect Effects 0.000 description 1
- 238000013461 design Methods 0.000 description 1
- 238000001784 detoxification Methods 0.000 description 1
- 238000010790 dilution Methods 0.000 description 1
- 239000012895 dilution Substances 0.000 description 1
- 239000012636 effector Substances 0.000 description 1
- 210000001671 embryonic stem cell Anatomy 0.000 description 1
- 230000002708 enhancing effect Effects 0.000 description 1
- 230000009088 enzymatic function Effects 0.000 description 1
- 108010087914 epidermal growth factor receptor VIII Proteins 0.000 description 1
- 230000008472 epithelial growth Effects 0.000 description 1
- 239000003797 essential amino acid Substances 0.000 description 1
- 230000001747 exhibiting effect Effects 0.000 description 1
- 230000003176 fibrotic effect Effects 0.000 description 1
- 238000012226 gene silencing method Methods 0.000 description 1
- 230000004077 genetic alteration Effects 0.000 description 1
- 231100000118 genetic alteration Toxicity 0.000 description 1
- 238000010353 genetic engineering Methods 0.000 description 1
- 210000003958 hematopoietic stem cell Anatomy 0.000 description 1
- 230000002949 hemolytic effect Effects 0.000 description 1
- 230000002440 hepatic effect Effects 0.000 description 1
- 230000001146 hypoxic effect Effects 0.000 description 1
- 230000005746 immune checkpoint blockade Effects 0.000 description 1
- 230000037451 immune surveillance Effects 0.000 description 1
- 208000026278 immune system disease Diseases 0.000 description 1
- 230000036039 immunity Effects 0.000 description 1
- 230000007365 immunoregulation Effects 0.000 description 1
- 230000001024 immunotherapeutic effect Effects 0.000 description 1
- 238000010874 in vitro model Methods 0.000 description 1
- 230000000415 inactivating effect Effects 0.000 description 1
- 210000004263 induced pluripotent stem cell Anatomy 0.000 description 1
- 208000015181 infectious disease Diseases 0.000 description 1
- 238000003780 insertion Methods 0.000 description 1
- 230000037431 insertion Effects 0.000 description 1
- 230000003993 interaction Effects 0.000 description 1
- 229940079322 interferon Drugs 0.000 description 1
- 239000000543 intermediate Substances 0.000 description 1
- 201000010659 intrinsic asthma Diseases 0.000 description 1
- 230000001678 irradiating effect Effects 0.000 description 1
- 239000000644 isotonic solution Substances 0.000 description 1
- 201000010982 kidney cancer Diseases 0.000 description 1
- 230000002147 killing effect Effects 0.000 description 1
- 210000004185 liver Anatomy 0.000 description 1
- 210000005229 liver cell Anatomy 0.000 description 1
- 210000005228 liver tissue Anatomy 0.000 description 1
- 230000005923 long-lasting effect Effects 0.000 description 1
- 239000003550 marker Substances 0.000 description 1
- 230000013011 mating Effects 0.000 description 1
- 230000035800 maturation Effects 0.000 description 1
- 230000009401 metastasis Effects 0.000 description 1
- 238000006386 neutralization reaction Methods 0.000 description 1
- 230000003472 neutralizing effect Effects 0.000 description 1
- 229910052757 nitrogen Inorganic materials 0.000 description 1
- 229960003301 nivolumab Drugs 0.000 description 1
- 210000000056 organ Anatomy 0.000 description 1
- 210000002220 organoid Anatomy 0.000 description 1
- 201000008968 osteosarcoma Diseases 0.000 description 1
- 230000002018 overexpression Effects 0.000 description 1
- 230000036961 partial effect Effects 0.000 description 1
- 239000002245 particle Substances 0.000 description 1
- 244000052769 pathogen Species 0.000 description 1
- 229960002621 pembrolizumab Drugs 0.000 description 1
- 210000005259 peripheral blood Anatomy 0.000 description 1
- 239000011886 peripheral blood Substances 0.000 description 1
- 229940021222 peritoneal dialysis isotonic solution Drugs 0.000 description 1
- 230000002085 persistent effect Effects 0.000 description 1
- 239000000546 pharmaceutical excipient Substances 0.000 description 1
- 229920001481 poly(stearyl methacrylate) Polymers 0.000 description 1
- 238000011176 pooling Methods 0.000 description 1
- 230000004481 post-translational protein modification Effects 0.000 description 1
- 239000002243 precursor Substances 0.000 description 1
- 230000002265 prevention Effects 0.000 description 1
- 238000012545 processing Methods 0.000 description 1
- 230000005855 radiation Effects 0.000 description 1
- 230000007115 recruitment Effects 0.000 description 1
- 238000011160 research Methods 0.000 description 1
- 230000004044 response Effects 0.000 description 1
- 229950004008 rimiducid Drugs 0.000 description 1
- 210000002966 serum Anatomy 0.000 description 1
- 208000012201 sexual and gender identity disease Diseases 0.000 description 1
- 208000015891 sexual disease Diseases 0.000 description 1
- 230000019491 signal transduction Effects 0.000 description 1
- 239000002924 silencing RNA Substances 0.000 description 1
- 230000037432 silent mutation Effects 0.000 description 1
- 239000011780 sodium chloride Substances 0.000 description 1
- 239000000243 solution Substances 0.000 description 1
- 241000894007 species Species 0.000 description 1
- 210000000952 spleen Anatomy 0.000 description 1
- 230000004936 stimulating effect Effects 0.000 description 1
- 239000000758 substrate Substances 0.000 description 1
- 230000007761 synergistic anti-cancer Effects 0.000 description 1
- 238000003786 synthesis reaction Methods 0.000 description 1
- 231100000331 toxic Toxicity 0.000 description 1
- 230000002588 toxic effect Effects 0.000 description 1
- 231100000419 toxicity Toxicity 0.000 description 1
- 230000001988 toxicity Effects 0.000 description 1
- 238000001890 transfection Methods 0.000 description 1
- 230000001131 transforming effect Effects 0.000 description 1
- 230000001960 triggered effect Effects 0.000 description 1
- 230000005747 tumor angiogenesis Effects 0.000 description 1
- 230000005751 tumor progression Effects 0.000 description 1
- 230000004143 urea cycle Effects 0.000 description 1
- 230000003612 virological effect Effects 0.000 description 1
- 230000029663 wound healing Effects 0.000 description 1
Images
Classifications
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K35/00—Medicinal preparations containing materials or reaction products thereof with undetermined constitution
- A61K35/12—Materials from mammals; Compositions comprising non-specified tissues or cells; Compositions comprising non-embryonic stem cells; Genetically modified cells
- A61K35/14—Blood; Artificial blood
- A61K35/17—Lymphocytes; B-cells; T-cells; Natural killer cells; Interferon-activated or cytokine-activated lymphocytes
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K38/00—Medicinal preparations containing peptides
- A61K38/16—Peptides having more than 20 amino acids; Gastrins; Somatostatins; Melanotropins; Derivatives thereof
- A61K38/17—Peptides having more than 20 amino acids; Gastrins; Somatostatins; Melanotropins; Derivatives thereof from animals; from humans
- A61K38/177—Receptors; Cell surface antigens; Cell surface determinants
- A61K38/1774—Immunoglobulin superfamily (e.g. CD2, CD4, CD8, ICAM molecules, B7 molecules, Fc-receptors, MHC-molecules)
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K39/00—Medicinal preparations containing antigens or antibodies
- A61K39/395—Antibodies; Immunoglobulins; Immune serum, e.g. antilymphocytic serum
- A61K39/39533—Antibodies; Immunoglobulins; Immune serum, e.g. antilymphocytic serum against materials from animals
- A61K39/3955—Antibodies; Immunoglobulins; Immune serum, e.g. antilymphocytic serum against materials from animals against proteinaceous materials, e.g. enzymes, hormones, lymphokines
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K39/00—Medicinal preparations containing antigens or antibodies
- A61K39/46—Cellular immunotherapy
- A61K39/461—Cellular immunotherapy characterised by the cell type used
- A61K39/4611—T-cells, e.g. tumor infiltrating lymphocytes [TIL], lymphokine-activated killer cells [LAK] or regulatory T cells [Treg]
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K39/00—Medicinal preparations containing antigens or antibodies
- A61K39/46—Cellular immunotherapy
- A61K39/464—Cellular immunotherapy characterised by the antigen targeted or presented
- A61K39/4643—Vertebrate antigens
- A61K39/4644—Cancer antigens
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P35/00—Antineoplastic agents
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07K—PEPTIDES
- C07K16/00—Immunoglobulins [IGs], e.g. monoclonal or polyclonal antibodies
- C07K16/18—Immunoglobulins [IGs], e.g. monoclonal or polyclonal antibodies against material from animals or humans
- C07K16/28—Immunoglobulins [IGs], e.g. monoclonal or polyclonal antibodies against material from animals or humans against receptors, cell surface antigens or cell surface determinants
- C07K16/2803—Immunoglobulins [IGs], e.g. monoclonal or polyclonal antibodies against material from animals or humans against receptors, cell surface antigens or cell surface determinants against the immunoglobulin superfamily
- C07K16/2818—Immunoglobulins [IGs], e.g. monoclonal or polyclonal antibodies against material from animals or humans against receptors, cell surface antigens or cell surface determinants against the immunoglobulin superfamily against CD28 or CD152
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12N—MICROORGANISMS OR ENZYMES; COMPOSITIONS THEREOF; PROPAGATING, PRESERVING, OR MAINTAINING MICROORGANISMS; MUTATION OR GENETIC ENGINEERING; CULTURE MEDIA
- C12N15/00—Mutation or genetic engineering; DNA or RNA concerning genetic engineering, vectors, e.g. plasmids, or their isolation, preparation or purification; Use of hosts therefor
- C12N15/09—Recombinant DNA-technology
- C12N15/11—DNA or RNA fragments; Modified forms thereof; Non-coding nucleic acids having a biological activity
- C12N15/113—Non-coding nucleic acids modulating the expression of genes, e.g. antisense oligonucleotides; Antisense DNA or RNA; Triplex- forming oligonucleotides; Catalytic nucleic acids, e.g. ribozymes; Nucleic acids used in co-suppression or gene silencing
- C12N15/1137—Non-coding nucleic acids modulating the expression of genes, e.g. antisense oligonucleotides; Antisense DNA or RNA; Triplex- forming oligonucleotides; Catalytic nucleic acids, e.g. ribozymes; Nucleic acids used in co-suppression or gene silencing against enzymes
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12N—MICROORGANISMS OR ENZYMES; COMPOSITIONS THEREOF; PROPAGATING, PRESERVING, OR MAINTAINING MICROORGANISMS; MUTATION OR GENETIC ENGINEERING; CULTURE MEDIA
- C12N9/00—Enzymes; Proenzymes; Compositions thereof; Processes for preparing, activating, inhibiting, separating or purifying enzymes
- C12N9/14—Hydrolases (3)
- C12N9/78—Hydrolases (3) acting on carbon to nitrogen bonds other than peptide bonds (3.5)
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12Y—ENZYMES
- C12Y305/00—Hydrolases acting on carbon-nitrogen bonds, other than peptide bonds (3.5)
- C12Y305/03—Hydrolases acting on carbon-nitrogen bonds, other than peptide bonds (3.5) in linear amidines (3.5.3)
- C12Y305/03001—Arginase (3.5.3.1)
-
- A—HUMAN NECESSITIES
- A01—AGRICULTURE; FORESTRY; ANIMAL HUSBANDRY; HUNTING; TRAPPING; FISHING
- A01K—ANIMAL HUSBANDRY; AVICULTURE; APICULTURE; PISCICULTURE; FISHING; REARING OR BREEDING ANIMALS, NOT OTHERWISE PROVIDED FOR; NEW BREEDS OF ANIMALS
- A01K2217/00—Genetically modified animals
- A01K2217/07—Animals genetically altered by homologous recombination
- A01K2217/075—Animals genetically altered by homologous recombination inducing loss of function, i.e. knock out
-
- A—HUMAN NECESSITIES
- A01—AGRICULTURE; FORESTRY; ANIMAL HUSBANDRY; HUNTING; TRAPPING; FISHING
- A01K—ANIMAL HUSBANDRY; AVICULTURE; APICULTURE; PISCICULTURE; FISHING; REARING OR BREEDING ANIMALS, NOT OTHERWISE PROVIDED FOR; NEW BREEDS OF ANIMALS
- A01K2227/00—Animals characterised by species
- A01K2227/10—Mammal
- A01K2227/105—Murine
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K39/00—Medicinal preparations containing antigens or antibodies
- A61K2039/505—Medicinal preparations containing antigens or antibodies comprising antibodies
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K39/00—Medicinal preparations containing antigens or antibodies
- A61K2039/51—Medicinal preparations containing antigens or antibodies comprising whole cells, viruses or DNA/RNA
- A61K2039/515—Animal cells
- A61K2039/5156—Animal cells expressing foreign proteins
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K2239/00—Indexing codes associated with cellular immunotherapy of group A61K39/46
- A61K2239/38—Indexing codes associated with cellular immunotherapy of group A61K39/46 characterised by the dose, timing or administration schedule
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K2239/00—Indexing codes associated with cellular immunotherapy of group A61K39/46
- A61K2239/46—Indexing codes associated with cellular immunotherapy of group A61K39/46 characterised by the cancer treated
- A61K2239/50—Colon
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K2239/00—Indexing codes associated with cellular immunotherapy of group A61K39/46
- A61K2239/46—Indexing codes associated with cellular immunotherapy of group A61K39/46 characterised by the cancer treated
- A61K2239/57—Skin; melanoma
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K2300/00—Mixtures or combinations of active ingredients, wherein at least one active ingredient is fully defined in groups A61K31/00 - A61K41/00
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12N—MICROORGANISMS OR ENZYMES; COMPOSITIONS THEREOF; PROPAGATING, PRESERVING, OR MAINTAINING MICROORGANISMS; MUTATION OR GENETIC ENGINEERING; CULTURE MEDIA
- C12N2310/00—Structure or type of the nucleic acid
- C12N2310/10—Type of nucleic acid
- C12N2310/14—Type of nucleic acid interfering N.A.
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12N—MICROORGANISMS OR ENZYMES; COMPOSITIONS THEREOF; PROPAGATING, PRESERVING, OR MAINTAINING MICROORGANISMS; MUTATION OR GENETIC ENGINEERING; CULTURE MEDIA
- C12N2310/00—Structure or type of the nucleic acid
- C12N2310/10—Type of nucleic acid
- C12N2310/14—Type of nucleic acid interfering N.A.
- C12N2310/141—MicroRNAs, miRNAs
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12N—MICROORGANISMS OR ENZYMES; COMPOSITIONS THEREOF; PROPAGATING, PRESERVING, OR MAINTAINING MICROORGANISMS; MUTATION OR GENETIC ENGINEERING; CULTURE MEDIA
- C12N2501/00—Active agents used in cell culture processes, e.g. differentation
- C12N2501/70—Enzymes
- C12N2501/73—Hydrolases (EC 3.)
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12N—MICROORGANISMS OR ENZYMES; COMPOSITIONS THEREOF; PROPAGATING, PRESERVING, OR MAINTAINING MICROORGANISMS; MUTATION OR GENETIC ENGINEERING; CULTURE MEDIA
- C12N2510/00—Genetically modified cells
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12N—MICROORGANISMS OR ENZYMES; COMPOSITIONS THEREOF; PROPAGATING, PRESERVING, OR MAINTAINING MICROORGANISMS; MUTATION OR GENETIC ENGINEERING; CULTURE MEDIA
- C12N5/00—Undifferentiated human, animal or plant cells, e.g. cell lines; Tissues; Cultivation or maintenance thereof; Culture media therefor
- C12N5/06—Animal cells or tissues; Human cells or tissues
- C12N5/0602—Vertebrate cells
- C12N5/0634—Cells from the blood or the immune system
- C12N5/0636—T lymphocytes
Landscapes
- Health & Medical Sciences (AREA)
- Life Sciences & Earth Sciences (AREA)
- Chemical & Material Sciences (AREA)
- Engineering & Computer Science (AREA)
- General Health & Medical Sciences (AREA)
- Medicinal Chemistry (AREA)
- Immunology (AREA)
- Organic Chemistry (AREA)
- Bioinformatics & Cheminformatics (AREA)
- Genetics & Genomics (AREA)
- Zoology (AREA)
- Biomedical Technology (AREA)
- Pharmacology & Pharmacy (AREA)
- Animal Behavior & Ethology (AREA)
- Public Health (AREA)
- Veterinary Medicine (AREA)
- Wood Science & Technology (AREA)
- Microbiology (AREA)
- Biotechnology (AREA)
- Epidemiology (AREA)
- Cell Biology (AREA)
- General Engineering & Computer Science (AREA)
- Biochemistry (AREA)
- Molecular Biology (AREA)
- Mycology (AREA)
- Virology (AREA)
- Nuclear Medicine, Radiotherapy & Molecular Imaging (AREA)
- General Chemical & Material Sciences (AREA)
- Chemical Kinetics & Catalysis (AREA)
- Endocrinology (AREA)
- Hematology (AREA)
- Biophysics (AREA)
- Proteomics, Peptides & Aminoacids (AREA)
- Gastroenterology & Hepatology (AREA)
- Physics & Mathematics (AREA)
- Plant Pathology (AREA)
- Developmental Biology & Embryology (AREA)
- Oncology (AREA)
- Medicines Containing Material From Animals Or Micro-Organisms (AREA)
- Medicines Containing Antibodies Or Antigens For Use As Internal Diagnostic Agents (AREA)
Abstract
Description
・該アルギナーゼをコードする遺伝子を変異させるか、切断するか、欠失させること、
・該アルギナーゼをコードする該遺伝子の転写因子をコードする遺伝子を投与するか、変異させるか、切断するか、欠失させること、
・該アルギナーゼをコードするmRNAに結合することができるヌクレオチド配列をコードするか含むヌクレオチド配列を投与すること
によって、該アルギナーゼの発現を障害することによって障害される。例えば、この工程は、該アルギナーゼをコードするmRNAをコードするか該mRNAに結合するヌクレオチド配列を含む核酸分子を投与することを含み得る。
例1:Arg2−/−マウスにおける腫瘍増殖の障害および抗腫瘍免疫の増強
本例では、Charles River Laboratories,Inc.から入手したArg2欠乏(ダブルノックアウト)マウスを使用した。第1のアプローチとして、WTマウスおよびArg2−/−マウスの腫瘍増殖を比較した。2つの移植可能な腫瘍モデル、B16メラノーマモデルおよびMC38結腸癌モデルを使用した。両モデルでは、代理腫瘍抗原としてオボアルブミン(OVA)を発現する腫瘍変異体を使用した。
Arg2−/−マウスの障害されたMC38−OVA増殖がCD8+T細胞による増強された制御によるものであるかどうかを決定するために、CD8+T細胞枯渇実験を行った。
MC38腫瘍は、T細胞阻害性PDL1−PD1チェックポイント軸を遮断する抗体による免疫療法に感受性である。PDL1−PD1遮断によって誘導されるMC38腫瘍の増強された制御が、Arg2欠乏から生じるMC38腫瘍の増強された制御に関与する機構と協働するかどうかを決定するために、MC38−OVA腫瘍を担持するWTマウスおよびArg2−/−マウスを抗PD1抗体により処置した。
障害されたMC38‐OVA増殖が、BM由来細胞または非造血起源の細胞のArg2欠乏の結果であるかどうかを決定するために、相互骨髄(BM)キメラマウスを作製した。WTマウスおよびArg2−/−マウスに致死量未満の放射線を照射して、宿主BMを破壊した。次いで、WTマウスまたはArg2−/−マウス由来のBM細胞を全4種の対の組合せで移植することによって、造血作用を再構成した。
CD8+T細胞のArg2欠乏が腫瘍増殖の制御の改善に関与するかどうかを決定するために、交配によりOTIマウスにArg2変異を付与して、二重ホモ接合Arg2−/−OTIマウスを得た。
WTバックグラウンドでのArg2+/+OTI細胞およびArg2−/−OTI細胞によるMC38−OVA腫瘍増殖および動物生存率の制御を比較するために、インビボ系を開発した。
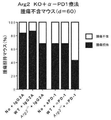
Arg2欠乏がCD8+T細胞のエフェクター機能に及ぼす影響をインビボでさらに調査するために、MC38−OVA腫瘍担持WTマウスに、同数のナイーブOTI CD8+T細胞およびArg2−/−OTI CD8+T細胞を移入した。OTI移入の1日後に、CpG−B+OVA−1ペプチドを用いて腫瘍担持レシピエントを免疫し、免疫化の7日後に、フローサイトメトリーにより、流入LN(dLN)および腫瘍内のOTI細胞を分析した。dLNおよび腫瘍内のArg2−/−OTI細胞ではともに、IFNγ+細胞の頻度が高かった(図8A)。腫瘍内のArg2−/−OTI細胞では、PD−1発現のレベルが有意に低かった(図8B)。
WT MC38−OVA腫瘍担癌マウスを対象に、養子Arg2−/−OTI移入とPD1遮断とを組み合わせる利点を調査した。WTマウスにMC38−OVA腫瘍を負荷し、5日後、腫瘍が触知可能になった際に、ドナーがキメラマウスでないことを除いて、図7Aに示すように養子移入細胞をマウスに投与し、翌日免疫した。T細胞移入後8日目、11日目および14日目に、腹腔内注射によりマウスに200μgの関連抗体を投与した。この設定では、T細胞固有のArg2欠乏とPD1遮断との相乗効果を再び観察した。Arg2−/−OT−I細胞または抗PD1抗体のみを投与されたマウスと比較して、併用処置を受けたマウスの方が、腫瘍増殖の大幅な減少(図9A)、長期の生存期間(図9B)および増加した腫瘍クリアランスを示した(図9C)。
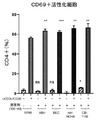
次に、ヒトCD4+T細胞およびCD8+T細胞でのARG阻害が、それらのインビトロでの活性化に影響を与えるかどうかを決定した。PBMCから精製したヒトT細胞を未処理のままにするか、抗CD3 Abおよび抗CD28 Abを用いてインビトロで活性化し、ARG阻害剤の存在下または非存在下で96ウェルプレート内のRPMI中で培養し、活性化24時間後の活性化(CD69染色)を評価した。ARG酵素機能の阻害により、CD4+T細胞(図10A)またはCD8+T細胞(図10B)内のCD69+細胞の頻度が増加し、ARG阻害によりインビトロでのヒトT細胞活性化が増加することが実証された。
Claims (20)
- 養子細胞移入により癌を治療するために、アルギナーゼ活性および/または発現が構成的または誘導的に障害された免疫細胞。
- 前記障害されたアルギナーゼ活性および/または発現が、障害されたアルギナーゼ2活性および/または発現である、請求項1に記載の免疫細胞。
- 白血病、リンパ腫および/または固形腫瘍からなる群から選択される1つ以上の癌を治療するための、請求項1または請求項2に記載の免疫細胞。
- 前記アルギナーゼ活性が、例えば、
・前記アルギナーゼをコードする遺伝子を変異させるか、切断するか、欠失させること、
・前記アルギナーゼをコードする前記遺伝子の転写因子をコードする遺伝子を投与するか、変異させるか、切断するか、欠失させること、
・前記アルギナーゼをコードするmRNAに結合することができるヌクレオチド配列をコードするか含むヌクレオチド配列を投与すること
によって、前記アルギナーゼの発現を障害することによって障害される、請求項1から3のいずれか一項に記載の免疫細胞。 - 前記アルギナーゼ活性が、前記アルギナーゼ活性および/または発現を障害するためのエクスビボ処理に前記細胞を曝すことによって障害される、請求項1から4のいずれか一項に記載の免疫細胞。
- 前記障害されたアルギナーゼ活性が、前記アルギナーゼをコードするmRNAに結合することができる核酸分子を前記免疫細胞にエクスビボで投与すること、またはそのような核酸分子をコードするベクターを投与することに起因する、請求項1から5のいずれか一項に記載の免疫細胞。
- T細胞、ナチュラルキラー細胞(NK細胞)、自然リンパ球系細胞および樹状細胞から選択される、請求項1から6のいずれか一項に記載の免疫細胞。
- CD3+T細胞および/またはCD4+T細胞および/またはCD8+T細胞から選択される、請求項7に記載の免疫細胞。
- キメラ抗原受容体(CAR)および/またはトランスジェニックT細胞受容体をさらに含み、前記CARが、リンカーを介してT細胞シグナル伝達ドメイン、好ましくはCD3ζシグナル伝達ドメインに融合された抗原結合ドメインを好ましくは含む、請求項1から8のいずれか一項に記載の免疫細胞。
- ネガティブ免疫チェックポイント調節因子を標的とするおよび/またはネガティブ免疫チェックポイント調節因子に特異的に結合する癌治療と組み合わせた、請求項1から9のいずれか一項に記載の免疫細胞。
- 前記癌治療が、免疫チェックポイント調節因子PD−L1/PD1、CTLA4、B7−H3(CD276)、B7−H4(B7x/B7S1/VTCN1)、HHLA2(B7H7/B7−H5)、VISTA(PD1H、DD1α、c10orf54、Gi24、Dies1、SISP1)、VSIG、LAG−3、TIGIT、CD96、CD39、CD73、アデノシンA2受容体、CD47、ブチロフィリン(BTN)および/またはTIM−3(T細胞−免疫グロブリン−ムチンドメイン3)を標的とする、請求項10に記載の免疫細胞。
- 前記癌治療が、PD1、PD−L1、CTLA4、B7−H3、B7−H4、HHLA2、VISTA、VSIG、LAG−3、TIGIT、CD96、CD39、CD73、アデノシンA2受容体、CD47、ブチロフィリン(BTN)および/またはTIM−3の群から選択される1つ以上に特異的に結合する抗体を含む、請求項10および/または11に記載の免疫細胞。
- 単離および/または精製されている、請求項1から12のいずれか一項に記載の免疫細胞。
- 特に養子細胞移入のための抗癌治療を調製する方法であって、
・免疫細胞を提供すること、および
・前記免疫細胞のアルギナーゼ活性および/または発現をエクスビボで障害することを含む方法。 - 養子細胞移入のための免疫細胞の抗癌活性を改善する方法であって、前記免疫細胞内のアルギナーゼ活性および/または発現をエクスビボで障害することを含む方法。
- 個体から、例えば治療される対象から以前に採取されている、請求項1から13のいずれか一項に記載の免疫細胞。
- 免疫細胞の抗癌活性を改善する方法であって、前記免疫細胞のアルギナーゼ活性および/または発現をエクスビボで障害することを含む方法。
- 養子細胞移入により癌を治療する方法であって、それを必要とする対象に、アルギナーゼ活性および/または発現が障害された免疫細胞を投与することを含む方法。
- 前記免疫細胞を投与する前に、前記免疫細胞内のアルギナーゼ活性および/または発現をエクスビボで障害することを含む、請求項18に記載の方法。
- 請求項1から13および16のいずれか一項に記載の免疫細胞を含む組成物。
Applications Claiming Priority (3)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| EP18153782.0 | 2018-01-28 | ||
| EP18153782 | 2018-01-28 | ||
| PCT/EP2019/051806 WO2019145453A1 (en) | 2018-01-28 | 2019-01-24 | Arginase suppression for cancer treatment |
Publications (2)
| Publication Number | Publication Date |
|---|---|
| JP2021517454A true JP2021517454A (ja) | 2021-07-26 |
| JPWO2019145453A5 JPWO2019145453A5 (ja) | 2022-02-24 |
Family
ID=61074351
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| JP2020541437A Pending JP2021517454A (ja) | 2018-01-28 | 2019-01-24 | 癌治療のためのアルギナーゼ抑制 |
Country Status (7)
| Country | Link |
|---|---|
| US (1) | US20210177897A1 (ja) |
| EP (1) | EP3743510A1 (ja) |
| JP (1) | JP2021517454A (ja) |
| CN (1) | CN111902532A (ja) |
| AU (1) | AU2019211067A1 (ja) |
| CA (1) | CA3125797A1 (ja) |
| WO (1) | WO2019145453A1 (ja) |
Families Citing this family (3)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| PL417066A1 (pl) | 2016-05-04 | 2017-11-06 | Oncoarendi Therapeutics Spółka Z Ograniczoną Odpowiedzialnością | Inhibitory arginazy oraz ich zastosowania terapeutyczne |
| WO2019186497A1 (en) | 2018-03-29 | 2019-10-03 | Oncoarendi Therapeutics S.A. | Dipeptide piperidine derivatives |
| GB201912030D0 (en) * | 2019-08-21 | 2019-10-02 | Cancer Research Tech Ltd | Binding molecules |
Citations (2)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US20140120136A1 (en) * | 2012-10-12 | 2014-05-01 | The Babraham Institute | Mir-155 enhancement of cd8+ t cell immunity |
| US20150275209A1 (en) * | 2012-10-22 | 2015-10-01 | The United States Of America, As Represented By The Secretary, Dept. Of Health And Human Service | Compositions and methods for enhancing cancer immunotherapy |
Family Cites Families (8)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CA2431080A1 (en) * | 2003-06-02 | 2004-12-02 | Catherine Adele O'brien | Enhancement of anticancer immunity through inhibition of arginase |
| WO2007111626A2 (en) | 2006-03-23 | 2007-10-04 | The Johns Hopkins University | Arginase ii: a target for the prevention and treatment of atherosclerosis |
| MX2009005370A (es) | 2006-11-21 | 2009-10-16 | Univ Groningen | Uso de inhibidores de arginasa en el tratamiento de asma y rinitis alergica. |
| CA2852685C (en) | 2011-10-19 | 2019-02-26 | Mars, Incorporated | Inhibitors of arginase and their therapeutic application |
| AU2015252569C1 (en) | 2014-04-29 | 2020-12-03 | Bio-Cancer Treatment International Limited | Methods and compositions for modulating the immune system with Arginase l |
| EP3298131B1 (en) | 2015-05-20 | 2023-04-26 | The Regents of The University of California | Method for generating human dendritic cells for immunotherapy |
| AU2016343682A1 (en) | 2015-10-30 | 2018-06-14 | The Regents Of The University Of California | Methods of generating T-cells from stem cells and immunotherapeutic methods using the T-cells |
| CN110352063A (zh) * | 2016-11-08 | 2019-10-18 | 卡里塞拉生物科学股份公司 | 精氨酸酶抑制剂组合疗法 |
-
2019
- 2019-01-24 JP JP2020541437A patent/JP2021517454A/ja active Pending
- 2019-01-24 CA CA3125797A patent/CA3125797A1/en active Pending
- 2019-01-24 WO PCT/EP2019/051806 patent/WO2019145453A1/en active Application Filing
- 2019-01-24 US US16/963,380 patent/US20210177897A1/en active Pending
- 2019-01-24 EP EP19705288.9A patent/EP3743510A1/en active Pending
- 2019-01-24 CN CN201980021913.2A patent/CN111902532A/zh active Pending
- 2019-01-24 AU AU2019211067A patent/AU2019211067A1/en not_active Abandoned
Patent Citations (2)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US20140120136A1 (en) * | 2012-10-12 | 2014-05-01 | The Babraham Institute | Mir-155 enhancement of cd8+ t cell immunity |
| US20150275209A1 (en) * | 2012-10-22 | 2015-10-01 | The United States Of America, As Represented By The Secretary, Dept. Of Health And Human Service | Compositions and methods for enhancing cancer immunotherapy |
Non-Patent Citations (2)
| Title |
|---|
| CELL, vol. 167, JPN6022055028, 2016, pages 829 - 842, ISSN: 0004953686 * |
| THE JOURNAL OF IMMUNOLOGY, vol. 193, no. 4, JPN6022055029, 2014, pages 1690 - 1700, ISSN: 0004953685 * |
Also Published As
| Publication number | Publication date |
|---|---|
| CA3125797A1 (en) | 2019-08-01 |
| AU2019211067A1 (en) | 2020-08-13 |
| CN111902532A (zh) | 2020-11-06 |
| EP3743510A1 (en) | 2020-12-02 |
| WO2019145453A1 (en) | 2019-08-01 |
| US20210177897A1 (en) | 2021-06-17 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| US11931380B2 (en) | Inhibition of diacylglycerol kinase to augment adoptive T cell transfer | |
| JP6818720B6 (ja) | カスパーゼポリペプチドを使用して部分的なアポトーシスを誘導するための方法 | |
| JP7008350B2 (ja) | Car発現ベクター及びcar発現t細胞 | |
| CN107206024B (zh) | 改变cart细胞中的基因表达及其用途 | |
| JP6866385B2 (ja) | 造血幹細胞における遺伝子編集のための方法および組成物 | |
| JP7083527B2 (ja) | ユニバーサルドナー細胞及び関連方法 | |
| Tomuleasa et al. | Chimeric antigen receptor T-cells for the treatment of B-cell acute lymphoblastic leukemia | |
| CN116064401A (zh) | 具有增强的细胞毒性的修饰的天然杀伤细胞和天然杀伤细胞系 | |
| US20230348855A1 (en) | Reducing fratricide of immune cells expressing nkg2d-based receptors | |
| JP7235391B2 (ja) | 人工的に操作された免疫細胞 | |
| JP2021517454A (ja) | 癌治療のためのアルギナーゼ抑制 | |
| CA3076095A1 (en) | A grna targeting hpk1 and a method for editing hpk1 gene | |
| Huang et al. | CRISPR/Cas systems to overcome challenges in developing the next generation of T cells for cancer therapy | |
| JP2022531577A (ja) | 改変されたcd247遺伝子座からキメラ受容体を発現する細胞、関連ポリヌクレオチド、および方法 | |
| US20230040477A1 (en) | T-cell death associated gene 8 (tdag8) modulation to enhance cellular cancer therapies | |
| US20180066253A1 (en) | Methods and compositions for modifying endothelial cells | |
| Tang et al. | Dominant negative TGFβ receptor II and truncated TIM3 enhance the antitumor efficacy of CAR-T-cell therapy in prostate cancer | |
| WO2023222928A2 (en) | Compositions and methods for treating a refractory or relapsed cancer or a chronic infectious disease |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| A521 | Request for written amendment filed |
Free format text: JAPANESE INTERMEDIATE CODE: A523 Effective date: 20210525 |
|
| A521 | Request for written amendment filed |
Free format text: JAPANESE INTERMEDIATE CODE: A523 Effective date: 20220114 |
|
| A621 | Written request for application examination |
Free format text: JAPANESE INTERMEDIATE CODE: A621 Effective date: 20220114 |
|
| A977 | Report on retrieval |
Free format text: JAPANESE INTERMEDIATE CODE: A971007 Effective date: 20221223 |
|
| A131 | Notification of reasons for refusal |
Free format text: JAPANESE INTERMEDIATE CODE: A131 Effective date: 20221227 |
|
| A02 | Decision of refusal |
Free format text: JAPANESE INTERMEDIATE CODE: A02 Effective date: 20230714 |