JP2020075889A - Washing and sterilizing compositions - Google Patents
Washing and sterilizing compositions Download PDFInfo
- Publication number
- JP2020075889A JP2020075889A JP2018210531A JP2018210531A JP2020075889A JP 2020075889 A JP2020075889 A JP 2020075889A JP 2018210531 A JP2018210531 A JP 2018210531A JP 2018210531 A JP2018210531 A JP 2018210531A JP 2020075889 A JP2020075889 A JP 2020075889A
- Authority
- JP
- Japan
- Prior art keywords
- cleaning
- sterilizing composition
- surfactant
- acid
- sterilizing
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Pending
Links
- 230000001954 sterilising effect Effects 0.000 title claims abstract description 71
- 239000000203 mixture Substances 0.000 title claims abstract description 69
- 238000005406 washing Methods 0.000 title abstract description 7
- -1 alkane sulfonate Chemical class 0.000 claims abstract description 84
- 239000004094 surface-active agent Substances 0.000 claims abstract description 41
- CPKVUHPKYQGHMW-UHFFFAOYSA-N 1-ethenylpyrrolidin-2-one;molecular iodine Chemical compound II.C=CN1CCCC1=O CPKVUHPKYQGHMW-UHFFFAOYSA-N 0.000 claims abstract description 35
- 229920000153 Povidone-iodine Polymers 0.000 claims abstract description 35
- 229960001621 povidone-iodine Drugs 0.000 claims abstract description 35
- 229920003171 Poly (ethylene oxide) Polymers 0.000 claims abstract description 21
- KWIUHFFTVRNATP-UHFFFAOYSA-N glycine betaine Chemical compound C[N+](C)(C)CC([O-])=O KWIUHFFTVRNATP-UHFFFAOYSA-N 0.000 claims abstract description 18
- 235000014113 dietary fatty acids Nutrition 0.000 claims abstract description 16
- 239000000194 fatty acid Substances 0.000 claims abstract description 16
- 229930195729 fatty acid Natural products 0.000 claims abstract description 16
- 229960003237 betaine Drugs 0.000 claims abstract description 11
- BDHFUVZGWQCTTF-UHFFFAOYSA-M sulfonate Chemical compound [O-]S(=O)=O BDHFUVZGWQCTTF-UHFFFAOYSA-M 0.000 claims abstract description 10
- 150000008051 alkyl sulfates Chemical class 0.000 claims abstract description 9
- PEDCQBHIVMGVHV-UHFFFAOYSA-N Glycerol Natural products OCC(O)CO PEDCQBHIVMGVHV-UHFFFAOYSA-N 0.000 claims abstract description 7
- BACYUWVYYTXETD-UHFFFAOYSA-N N-Lauroylsarcosine Chemical class CCCCCCCCCCCC(=O)N(C)CC(O)=O BACYUWVYYTXETD-UHFFFAOYSA-N 0.000 claims abstract description 7
- 229920001214 Polysorbate 60 Polymers 0.000 claims abstract description 6
- 229930182478 glucoside Natural products 0.000 claims abstract description 6
- 235000011187 glycerol Nutrition 0.000 claims abstract description 6
- 239000004711 α-olefin Substances 0.000 claims abstract description 6
- 229930006000 Sucrose Natural products 0.000 claims abstract description 5
- 150000005215 alkyl ethers Chemical class 0.000 claims abstract description 5
- 239000005720 sucrose Substances 0.000 claims abstract description 5
- PSBDWGZCVUAZQS-UHFFFAOYSA-N (dimethylsulfonio)acetate Chemical compound C[S+](C)CC([O-])=O PSBDWGZCVUAZQS-UHFFFAOYSA-N 0.000 claims abstract description 4
- 229940117986 sulfobetaine Drugs 0.000 claims abstract description 4
- 238000004140 cleaning Methods 0.000 claims description 73
- QTBSBXVTEAMEQO-UHFFFAOYSA-N Acetic acid Chemical compound CC(O)=O QTBSBXVTEAMEQO-UHFFFAOYSA-N 0.000 claims description 12
- KRKNYBCHXYNGOX-UHFFFAOYSA-N citric acid Chemical compound OC(=O)CC(O)(C(O)=O)CC(O)=O KRKNYBCHXYNGOX-UHFFFAOYSA-N 0.000 claims description 12
- NBIIXXVUZAFLBC-UHFFFAOYSA-N Phosphoric acid Chemical compound OP(O)(O)=O NBIIXXVUZAFLBC-UHFFFAOYSA-N 0.000 claims description 8
- JVTAAEKCZFNVCJ-UHFFFAOYSA-N lactic acid Chemical compound CC(O)C(O)=O JVTAAEKCZFNVCJ-UHFFFAOYSA-N 0.000 claims description 8
- VEXZGXHMUGYJMC-UHFFFAOYSA-N Hydrochloric acid Chemical compound Cl VEXZGXHMUGYJMC-UHFFFAOYSA-N 0.000 claims description 6
- QAOWNCQODCNURD-UHFFFAOYSA-N Sulfuric acid Chemical compound OS(O)(=O)=O QAOWNCQODCNURD-UHFFFAOYSA-N 0.000 claims description 6
- 150000003839 salts Chemical class 0.000 claims description 5
- BJEPYKJPYRNKOW-REOHCLBHSA-N (S)-malic acid Chemical compound OC(=O)[C@@H](O)CC(O)=O BJEPYKJPYRNKOW-REOHCLBHSA-N 0.000 claims description 4
- KDYFGRWQOYBRFD-UHFFFAOYSA-N Succinic acid Natural products OC(=O)CCC(O)=O KDYFGRWQOYBRFD-UHFFFAOYSA-N 0.000 claims description 4
- 150000008044 alkali metal hydroxides Chemical class 0.000 claims description 4
- BJEPYKJPYRNKOW-UHFFFAOYSA-N alpha-hydroxysuccinic acid Natural products OC(=O)C(O)CC(O)=O BJEPYKJPYRNKOW-UHFFFAOYSA-N 0.000 claims description 4
- 229910000147 aluminium phosphate Inorganic materials 0.000 claims description 4
- KDYFGRWQOYBRFD-NUQCWPJISA-N butanedioic acid Chemical compound O[14C](=O)CC[14C](O)=O KDYFGRWQOYBRFD-NUQCWPJISA-N 0.000 claims description 4
- 239000004310 lactic acid Substances 0.000 claims description 4
- 235000014655 lactic acid Nutrition 0.000 claims description 4
- 239000001630 malic acid Substances 0.000 claims description 4
- 235000011090 malic acid Nutrition 0.000 claims description 4
- 230000000249 desinfective effect Effects 0.000 claims description 3
- 125000005526 alkyl sulfate group Chemical group 0.000 claims 1
- 150000002148 esters Chemical class 0.000 claims 1
- 238000004659 sterilization and disinfection Methods 0.000 abstract description 3
- 238000002360 preparation method Methods 0.000 description 13
- 238000011156 evaluation Methods 0.000 description 9
- 239000006260 foam Substances 0.000 description 9
- 239000002244 precipitate Substances 0.000 description 8
- 230000000844 anti-bacterial effect Effects 0.000 description 7
- 230000000052 comparative effect Effects 0.000 description 7
- DGAQECJNVWCQMB-PUAWFVPOSA-M Ilexoside XXIX Chemical compound C[C@@H]1CC[C@@]2(CC[C@@]3(C(=CC[C@H]4[C@]3(CC[C@@H]5[C@@]4(CC[C@@H](C5(C)C)OS(=O)(=O)[O-])C)C)[C@@H]2[C@]1(C)O)C)C(=O)O[C@H]6[C@@H]([C@H]([C@@H]([C@H](O6)CO)O)O)O.[Na+] DGAQECJNVWCQMB-PUAWFVPOSA-M 0.000 description 6
- 238000005187 foaming Methods 0.000 description 6
- 229910052708 sodium Inorganic materials 0.000 description 6
- 239000011734 sodium Substances 0.000 description 6
- 239000007788 liquid Substances 0.000 description 5
- 238000013459 approach Methods 0.000 description 4
- 235000019864 coconut oil Nutrition 0.000 description 4
- 239000003240 coconut oil Substances 0.000 description 4
- LFQSCWFLJHTTHZ-UHFFFAOYSA-N Ethanol Chemical compound CCO LFQSCWFLJHTTHZ-UHFFFAOYSA-N 0.000 description 3
- KWYUFKZDYYNOTN-UHFFFAOYSA-M Potassium hydroxide Chemical compound [OH-].[K+] KWYUFKZDYYNOTN-UHFFFAOYSA-M 0.000 description 3
- DNIAPMSPPWPWGF-UHFFFAOYSA-N Propylene glycol Chemical compound CC(O)CO DNIAPMSPPWPWGF-UHFFFAOYSA-N 0.000 description 3
- HEMHJVSKTPXQMS-UHFFFAOYSA-M Sodium hydroxide Chemical compound [OH-].[Na+] HEMHJVSKTPXQMS-UHFFFAOYSA-M 0.000 description 3
- 150000001335 aliphatic alkanes Chemical group 0.000 description 3
- 150000001875 compounds Chemical class 0.000 description 3
- POULHZVOKOAJMA-UHFFFAOYSA-N dodecanoic acid Chemical compound CCCCCCCCCCCC(O)=O POULHZVOKOAJMA-UHFFFAOYSA-N 0.000 description 3
- NLKNQRATVPKPDG-UHFFFAOYSA-M potassium iodide Chemical compound [K+].[I-] NLKNQRATVPKPDG-UHFFFAOYSA-M 0.000 description 3
- ZCYVEMRRCGMTRW-UHFFFAOYSA-N 7553-56-2 Chemical compound [I] ZCYVEMRRCGMTRW-UHFFFAOYSA-N 0.000 description 2
- 239000005639 Lauric acid Substances 0.000 description 2
- 229920001213 Polysorbate 20 Polymers 0.000 description 2
- 239000000796 flavoring agent Substances 0.000 description 2
- 235000013355 food flavoring agent Nutrition 0.000 description 2
- 229910052740 iodine Inorganic materials 0.000 description 2
- 239000011630 iodine Substances 0.000 description 2
- 230000007794 irritation Effects 0.000 description 2
- 238000000034 method Methods 0.000 description 2
- 239000000256 polyoxyethylene sorbitan monolaurate Substances 0.000 description 2
- 235000010486 polyoxyethylene sorbitan monolaurate Nutrition 0.000 description 2
- 235000010483 polyoxyethylene sorbitan monopalmitate Nutrition 0.000 description 2
- 239000000249 polyoxyethylene sorbitan monopalmitate Substances 0.000 description 2
- 239000001818 polyoxyethylene sorbitan monostearate Substances 0.000 description 2
- 235000010989 polyoxyethylene sorbitan monostearate Nutrition 0.000 description 2
- 108700004121 sarkosyl Proteins 0.000 description 2
- 238000000926 separation method Methods 0.000 description 2
- XLYOFNOQVPJJNP-UHFFFAOYSA-N water Substances O XLYOFNOQVPJJNP-UHFFFAOYSA-N 0.000 description 2
- NOOLISFMXDJSKH-KXUCPTDWSA-N (-)-Menthol Chemical compound CC(C)[C@@H]1CC[C@@H](C)C[C@H]1O NOOLISFMXDJSKH-KXUCPTDWSA-N 0.000 description 1
- PZNPLUBHRSSFHT-RRHRGVEJSA-N 1-hexadecanoyl-2-octadecanoyl-sn-glycero-3-phosphocholine Chemical compound CCCCCCCCCCCCCCCCCC(=O)O[C@@H](COP([O-])(=O)OCC[N+](C)(C)C)COC(=O)CCCCCCCCCCCCCCC PZNPLUBHRSSFHT-RRHRGVEJSA-N 0.000 description 1
- RZRNAYUHWVFMIP-KTKRTIGZSA-N 1-oleoylglycerol Chemical compound CCCCCCCC\C=C/CCCCCCCC(=O)OCC(O)CO RZRNAYUHWVFMIP-KTKRTIGZSA-N 0.000 description 1
- HVYJSOSGTDINLW-UHFFFAOYSA-N 2-[dimethyl(octadecyl)azaniumyl]acetate Chemical compound CCCCCCCCCCCCCCCCCC[N+](C)(C)CC([O-])=O HVYJSOSGTDINLW-UHFFFAOYSA-N 0.000 description 1
- HJDITXMCJQRQLU-UHFFFAOYSA-N 2-[dodecanoyl(methyl)amino]acetate;tris(2-hydroxyethyl)azanium Chemical compound OCCN(CCO)CCO.CCCCCCCCCCCC(=O)N(C)CC(O)=O HJDITXMCJQRQLU-UHFFFAOYSA-N 0.000 description 1
- DDGPBVIAYDDWDH-UHFFFAOYSA-N 3-[dodecyl(dimethyl)azaniumyl]-2-hydroxypropane-1-sulfonate Chemical compound CCCCCCCCCCCC[N+](C)(C)CC(O)CS([O-])(=O)=O DDGPBVIAYDDWDH-UHFFFAOYSA-N 0.000 description 1
- CDOUZKKFHVEKRI-UHFFFAOYSA-N 3-bromo-n-[(prop-2-enoylamino)methyl]propanamide Chemical compound BrCCC(=O)NCNC(=O)C=C CDOUZKKFHVEKRI-UHFFFAOYSA-N 0.000 description 1
- LIFHMKCDDVTICL-UHFFFAOYSA-N 6-(chloromethyl)phenanthridine Chemical compound C1=CC=C2C(CCl)=NC3=CC=CC=C3C2=C1 LIFHMKCDDVTICL-UHFFFAOYSA-N 0.000 description 1
- QGZKDVFQNNGYKY-UHFFFAOYSA-O Ammonium Chemical compound [NH4+] QGZKDVFQNNGYKY-UHFFFAOYSA-O 0.000 description 1
- 241000233788 Arecaceae Species 0.000 description 1
- WWZKQHOCKIZLMA-UHFFFAOYSA-N Caprylic acid Natural products CCCCCCCC(O)=O WWZKQHOCKIZLMA-UHFFFAOYSA-N 0.000 description 1
- 239000005635 Caprylic acid (CAS 124-07-2) Substances 0.000 description 1
- JDRSMPFHFNXQRB-CMTNHCDUSA-N Decyl beta-D-threo-hexopyranoside Chemical compound CCCCCCCCCCO[C@@H]1O[C@H](CO)C(O)[C@H](O)C1O JDRSMPFHFNXQRB-CMTNHCDUSA-N 0.000 description 1
- 239000005977 Ethylene Substances 0.000 description 1
- 239000004909 Moisturizer Substances 0.000 description 1
- 239000002202 Polyethylene glycol Substances 0.000 description 1
- 239000004743 Polypropylene Substances 0.000 description 1
- 229920001219 Polysorbate 40 Polymers 0.000 description 1
- ZLMJMSJWJFRBEC-UHFFFAOYSA-N Potassium Chemical compound [K] ZLMJMSJWJFRBEC-UHFFFAOYSA-N 0.000 description 1
- DBMJMQXJHONAFJ-UHFFFAOYSA-M Sodium laurylsulphate Chemical compound [Na+].CCCCCCCCCCCCOS([O-])(=O)=O DBMJMQXJHONAFJ-UHFFFAOYSA-M 0.000 description 1
- GSEJCLTVZPLZKY-UHFFFAOYSA-N Triethanolamine Chemical compound OCCN(CCO)CCO GSEJCLTVZPLZKY-UHFFFAOYSA-N 0.000 description 1
- GCSPRLPXTPMSTL-IBDNADADSA-N [(2s,3r,4s,5s,6r)-2-[(2s,3s,4s,5r)-3,4-dihydroxy-2,5-bis(hydroxymethyl)oxolan-2-yl]-3,4,5-trihydroxy-6-(hydroxymethyl)oxan-2-yl] dodecanoate Chemical compound CCCCCCCCCCCC(=O)O[C@@]1([C@]2(CO)[C@H]([C@H](O)[C@@H](CO)O2)O)O[C@H](CO)[C@@H](O)[C@H](O)[C@H]1O GCSPRLPXTPMSTL-IBDNADADSA-N 0.000 description 1
- UEYVMVXJVDAGBB-ZHBLIPIOSA-N [(2s,3s,4s,5r)-3,4-dihydroxy-5-(hydroxymethyl)-2-[(2r,3r,4s,5s,6r)-3,4,5-trihydroxy-6-(hydroxymethyl)oxan-2-yl]oxyoxolan-2-yl]methyl tetradecanoate Chemical compound O([C@@H]1[C@@H]([C@@H](O)[C@H](O)[C@@H](CO)O1)O)[C@]1(COC(=O)CCCCCCCCCCCCC)O[C@H](CO)[C@@H](O)[C@@H]1O UEYVMVXJVDAGBB-ZHBLIPIOSA-N 0.000 description 1
- 239000002253 acid Substances 0.000 description 1
- 229910052783 alkali metal Inorganic materials 0.000 description 1
- 229910052784 alkaline earth metal Inorganic materials 0.000 description 1
- 125000000217 alkyl group Chemical group 0.000 description 1
- BTBJBAZGXNKLQC-UHFFFAOYSA-N ammonium lauryl sulfate Chemical compound [NH4+].CCCCCCCCCCCCOS([O-])(=O)=O BTBJBAZGXNKLQC-UHFFFAOYSA-N 0.000 description 1
- 229940063953 ammonium lauryl sulfate Drugs 0.000 description 1
- 239000007864 aqueous solution Substances 0.000 description 1
- 235000015278 beef Nutrition 0.000 description 1
- 230000005587 bubbling Effects 0.000 description 1
- 239000006229 carbon black Substances 0.000 description 1
- 239000001913 cellulose Substances 0.000 description 1
- 229920002678 cellulose Polymers 0.000 description 1
- 238000013329 compounding Methods 0.000 description 1
- 238000010411 cooking Methods 0.000 description 1
- 239000013078 crystal Substances 0.000 description 1
- 238000005520 cutting process Methods 0.000 description 1
- 125000003450 decanoic acid ester group Chemical group 0.000 description 1
- 229940073499 decyl glucoside Drugs 0.000 description 1
- 239000003599 detergent Substances 0.000 description 1
- 235000019329 dioctyl sodium sulphosuccinate Nutrition 0.000 description 1
- 238000007599 discharging Methods 0.000 description 1
- GVGUFUZHNYFZLC-UHFFFAOYSA-N dodecyl benzenesulfonate;sodium Chemical compound [Na].CCCCCCCCCCCCOS(=O)(=O)C1=CC=CC=C1 GVGUFUZHNYFZLC-UHFFFAOYSA-N 0.000 description 1
- SYELZBGXAIXKHU-UHFFFAOYSA-N dodecyldimethylamine N-oxide Chemical compound CCCCCCCCCCCC[N+](C)(C)[O-] SYELZBGXAIXKHU-UHFFFAOYSA-N 0.000 description 1
- 229940079593 drug Drugs 0.000 description 1
- 239000003814 drug Substances 0.000 description 1
- 150000004665 fatty acids Chemical class 0.000 description 1
- 235000003599 food sweetener Nutrition 0.000 description 1
- 238000009472 formulation Methods 0.000 description 1
- RZRNAYUHWVFMIP-HXUWFJFHSA-N glycerol monolinoleate Natural products CCCCCCCCC=CCCCCCCCC(=O)OC[C@H](O)CO RZRNAYUHWVFMIP-HXUWFJFHSA-N 0.000 description 1
- 229940094506 lauryl betaine Drugs 0.000 description 1
- PYIDGJJWBIBVIA-UYTYNIKBSA-N lauryl glucoside Chemical compound CCCCCCCCCCCCO[C@@H]1O[C@H](CO)[C@@H](O)[C@H](O)[C@H]1O PYIDGJJWBIBVIA-UYTYNIKBSA-N 0.000 description 1
- 229940048848 lauryl glucoside Drugs 0.000 description 1
- 230000001333 moisturizer Effects 0.000 description 1
- 229940074096 monoolein Drugs 0.000 description 1
- DVEKCXOJTLDBFE-UHFFFAOYSA-N n-dodecyl-n,n-dimethylglycinate Chemical compound CCCCCCCCCCCC[N+](C)(C)CC([O-])=O DVEKCXOJTLDBFE-UHFFFAOYSA-N 0.000 description 1
- 229960002446 octanoic acid Drugs 0.000 description 1
- 229940049964 oleate Drugs 0.000 description 1
- ZQPPMHVWECSIRJ-KTKRTIGZSA-N oleic acid Chemical compound CCCCCCCC\C=C/CCCCCCCC(O)=O ZQPPMHVWECSIRJ-KTKRTIGZSA-N 0.000 description 1
- 235000019271 petrolatum Nutrition 0.000 description 1
- 229920001223 polyethylene glycol Polymers 0.000 description 1
- 229920000259 polyoxyethylene lauryl ether Polymers 0.000 description 1
- 239000000244 polyoxyethylene sorbitan monooleate Substances 0.000 description 1
- 235000010482 polyoxyethylene sorbitan monooleate Nutrition 0.000 description 1
- 229920001155 polypropylene Polymers 0.000 description 1
- 229940068977 polysorbate 20 Drugs 0.000 description 1
- 229940101027 polysorbate 40 Drugs 0.000 description 1
- 229940113124 polysorbate 60 Drugs 0.000 description 1
- 229920000053 polysorbate 80 Polymers 0.000 description 1
- 229940068968 polysorbate 80 Drugs 0.000 description 1
- 229910052700 potassium Inorganic materials 0.000 description 1
- 239000011591 potassium Substances 0.000 description 1
- 159000000001 potassium salts Chemical class 0.000 description 1
- CVHZOJJKTDOEJC-UHFFFAOYSA-N saccharin Chemical compound C1=CC=C2C(=O)NS(=O)(=O)C2=C1 CVHZOJJKTDOEJC-UHFFFAOYSA-N 0.000 description 1
- 229940081974 saccharin Drugs 0.000 description 1
- 235000019204 saccharin Nutrition 0.000 description 1
- 239000000901 saccharin and its Na,K and Ca salt Substances 0.000 description 1
- 239000000344 soap Substances 0.000 description 1
- 229940080264 sodium dodecylbenzenesulfonate Drugs 0.000 description 1
- 235000019333 sodium laurylsulphate Nutrition 0.000 description 1
- 159000000000 sodium salts Chemical class 0.000 description 1
- RLJSXMVTLMHXJS-UHFFFAOYSA-M sodium;4-decylbenzenesulfonate Chemical compound [Na+].CCCCCCCCCCC1=CC=C(S([O-])(=O)=O)C=C1 RLJSXMVTLMHXJS-UHFFFAOYSA-M 0.000 description 1
- 239000000243 solution Substances 0.000 description 1
- 239000002904 solvent Substances 0.000 description 1
- 239000008347 soybean phospholipid Substances 0.000 description 1
- 239000003381 stabilizer Substances 0.000 description 1
- 238000003860 storage Methods 0.000 description 1
- 239000000126 substance Substances 0.000 description 1
- 239000003765 sweetening agent Substances 0.000 description 1
- 239000003760 tallow Substances 0.000 description 1
- 239000003871 white petrolatum Substances 0.000 description 1
Abstract
Description
本発明は、洗浄殺菌組成物に関する。 The present invention relates to a cleaning and sterilizing composition.
ポビドンヨード製剤が優れた殺菌効果を有することは広く一般に知られている。また、優れた殺菌効果を有するポビドンヨード製剤を用いて手指を消毒することは従来から行われてきた。 It is widely known that povidone-iodine preparations have an excellent bactericidal effect. Further, it has been conventionally performed to disinfect a finger with a povidone iodine preparation having an excellent bactericidal effect.
ポビドンヨード製剤は、含ヨード水溶液であり、これを使用する際に、薬液が飛散して衣服等に付着すると褐色の染みになり、その洗浄に手数を要するという問題があった。また手指の消毒の際、液だれが生じると薬液が無駄になるという問題があった。 The povidone-iodine preparation is an iodine-containing aqueous solution, and when this is used, there is a problem in that if the drug solution scatters and adheres to clothes or the like, it becomes a brown stain, and cleaning it requires labor. In addition, there is a problem that the chemical liquid is wasted when dripping occurs during disinfection of fingers.
このような問題を解決するため、特許文献1には、ポビドンヨード製剤に所定の界面活性剤を加え、ポビドンヨード製剤を泡状にして使用することが記載されている。 In order to solve such a problem, Patent Literature 1 describes that a povidone-iodine preparation is added with a predetermined surfactant to make the povidone-iodine preparation into a foam and used.
特許文献1に記載されたようなポビドンヨード製剤を用いて手指を消毒する際、手指に汚れが付着していると、ポビドンヨード製剤の殺菌効果が充分に発揮されない。
そのため、ポビドンヨード製剤を用いて手指を消毒する際には、まず、石鹸等の洗浄剤で手指を洗浄することにより汚れを落とし、手指を乾燥させてからポビドンヨード製剤を手指に塗り付け、その後、ポビドンヨード製剤をすすいで洗い流すことが行われている。
すなわち、手指を消毒するためには、手指の洗浄と、ポビドンヨード製剤による殺菌という2工程が必要であった。そのため、迅速に手指を消毒できないという問題があった。
When disinfecting a finger using the povidone-iodine preparation as described in Patent Document 1, if the finger is soiled, the povidone-iodine preparation will not exhibit its bactericidal effect sufficiently.
Therefore, when disinfecting fingers with a povidone iodine preparation, first wash the fingers with a detergent such as soap to remove stains, dry the fingers, and then apply the povidone iodine preparation to the fingers, and then povidone iodine. It is common practice to rinse the formulation off.
That is, in order to disinfect a finger, two steps of washing the finger and sterilizing with a povidone iodine preparation were required. Therefore, there is a problem that the fingers cannot be disinfected quickly.
また、特許文献1に記載のポビドンヨード製剤には、界面活性剤としてジオクチルソジウムスルホサクシネートが含まれており、低温で保管すると、針状結晶が生じるという問題があった。 Further, the povidone-iodine preparation described in Patent Document 1 contains dioctylsodium sulfosuccinate as a surfactant, and there is a problem that needle-like crystals are generated when stored at low temperature.
本発明は、上記問題を解決するためにされたものであり、本発明の目的は、迅速に手指を殺菌することができる洗浄殺菌組成物を提供することである。 The present invention has been made to solve the above problems, and an object of the present invention is to provide a cleaning and sterilizing composition capable of rapidly sterilizing fingers.
本発明者らは、鋭意研究を重ねた結果、ポビドンヨード製剤に所定の界面活性剤を加えると、手指の洗浄及び殺菌を同時に行えることを見出し、本発明を完成させた。 As a result of intensive studies, the present inventors have found that by adding a predetermined surfactant to a povidone iodine preparation, it is possible to simultaneously clean and sterilize fingers, and have completed the present invention.
すなわち、本発明の洗浄殺菌組成物は、界面活性剤と、ポビドンヨードとを含み、上記界面活性剤は、アルカンスルホン酸塩、アルキルアミドプロピルベタイン、アルキルグルコシド、アルキルジメチルアミンオキシド、アルキルベタイン、アルキルベンゼンスルホン酸塩、アルキル硫酸塩、α−オレフィンスルホン酸塩、グリセリン脂肪酸エステル、ショ糖脂肪酸エステル、スルホベタイン、ポリオキシエチレンアルキルエーテル、ポリオキシエチレンアルキルエーテル硫酸塩、ポリオキシエチレンアルキルエーテル酢酸塩、ポリオキシエチレンソルビタン脂肪酸エステル、ラウロイルサルコシン塩からなる群から選択される少なくとも1種であることを特徴とする。 That is, the cleaning and sterilizing composition of the present invention contains a surfactant and povidone iodine, and the surfactant is an alkane sulfonate, an alkylamidopropyl betaine, an alkyl glucoside, an alkyl dimethyl amine oxide, an alkyl betaine, an alkyl benzene sulfone. Acid salt, alkyl sulfate, α-olefin sulfonate, glycerin fatty acid ester, sucrose fatty acid ester, sulfobetaine, polyoxyethylene alkyl ether, polyoxyethylene alkyl ether sulfate, polyoxyethylene alkyl ether acetate, polyoxy It is characterized by being at least one selected from the group consisting of ethylene sorbitan fatty acid ester and lauroyl sarcosine salt.
本発明の洗浄殺菌組成物は、所定の界面活性剤を含む。
これらの界面活性剤は、洗浄能力に優れている。そのため、手指が汚れていたとしても、その汚れを落とすことができる。
また、本発明の洗浄殺菌組成物はポビドンヨードも含むので、手指の洗浄と同時に、手指の殺菌も行うことができる。
すなわち、1工程で迅速に手指の洗浄及び殺菌を行うことができる。
The cleaning and sterilizing composition of the present invention contains a predetermined surfactant.
These surfactants have excellent cleaning ability. Therefore, even if the fingers are dirty, the dirt can be removed.
Moreover, since the cleaning and sterilizing composition of the present invention also contains povidone iodine, it is possible to sterilize the fingers at the same time as cleaning the fingers.
That is, the fingers can be quickly washed and sterilized in one step.
また、これらの界面活性剤は、起泡性にも優れるので、本発明の洗浄殺菌組成物は泡ポンプ等によりわざわざ泡状にしなくても、手でこすり合わせるだけで泡立てることができる。このような泡は、洗浄力にも寄与する。 Further, since these surfactants are also excellent in foaming property, the cleaning and sterilizing composition of the present invention can be foamed by rubbing by hand without having to purposely foam the composition with a foam pump or the like. Such bubbles also contribute to detergency.
さらに、これらの界面活性剤は、低温安定性にも優れるので、低温で保管した場合であったとしても析出しにくい。 Furthermore, since these surfactants are also excellent in low temperature stability, they are unlikely to precipitate even when stored at low temperatures.
本発明の洗浄殺菌組成物では、上記洗浄殺菌組成物中の上記界面活性剤の濃度は、0.01〜10.0重量%であることが望ましい。
界面活性剤の濃度が、0.01重量%未満であると、充分な洗浄力が得られない。
界面活性剤の濃度が、10.0重量%を超えると、界面活性剤が析出しやすくなる。また、性能が頭打ちになり、費用対効果が低下してしまう。
In the cleaning / sterilizing composition of the present invention, the concentration of the surfactant in the cleaning / sterilizing composition is preferably 0.01 to 10.0% by weight.
If the concentration of the surfactant is less than 0.01% by weight, sufficient detergency cannot be obtained.
When the concentration of the surfactant exceeds 10.0% by weight, the surfactant tends to precipitate. In addition, the performance will reach a ceiling and cost effectiveness will decrease.
本発明の洗浄殺菌組成物では、上記界面活性剤は、アルキル硫酸塩及び/又はポリオキシエチレンアルキルエーテル硫酸塩であり、上記洗浄殺菌組成物中の上記界面活性剤の濃度は1.0重量%を超えることが望ましい。
界面活性剤が、アルキル硫酸塩及び/又はポリオキシエチレンアルキルエーテル硫酸塩であり、その濃度が1.0重量%を超えると、洗浄殺菌組成物の洗浄力及び泡立ちが良好になる。
In the cleaning and sterilizing composition of the present invention, the surfactant is an alkyl sulfate and / or a polyoxyethylene alkyl ether sulfate, and the concentration of the surfactant in the cleaning and sterilizing composition is 1.0% by weight. It is desirable to exceed.
When the surfactant is an alkyl sulfate and / or a polyoxyethylene alkyl ether sulfate, and the concentration thereof exceeds 1.0% by weight, the detergency and foaming of the cleaning / sterilizing composition will be good.
本発明の洗浄殺菌組成物では、上記洗浄殺菌組成物中の上記ポビドンヨードの濃度は、0.03〜20重量%であることが望ましい。
ポビドンヨードの濃度が、0.03重量%未満であると、ポビドンヨードの濃度が低いので、充分な殺菌効果を示さなくなる。
ポビドンヨードの濃度が、20重量%を超えると、ポビドンヨードの溶解度が上限に近づき、溶解できない、又は、析出しやすくなる。また、ポビドンヨードによる殺菌効果が上限に近づく。
In the cleaning and sterilizing composition of the present invention, the concentration of the povidone iodine in the cleaning and sterilizing composition is preferably 0.03 to 20% by weight.
When the concentration of povidone iodine is less than 0.03% by weight, the concentration of povidone iodine is low, so that a sufficient bactericidal effect cannot be obtained.
If the concentration of povidone-iodine exceeds 20% by weight, the solubility of povidone-iodine approaches the upper limit, and the povidone-iodine cannot be dissolved or is likely to precipitate. Also, the bactericidal effect of povidone iodine approaches the upper limit.
本発明の洗浄殺菌組成物は、さらに、塩酸、硫酸、クエン酸、コハク酸、酢酸、乳酸、リン酸、リンゴ酸、及び、これらの塩、並びに、アルカリ金属水酸化物からなる群から選択される少なくとも1種を含むことが望ましい。
これらの化合物を用いて本発明の洗浄殺菌組成物のpHを調整することができる。
The cleaning and sterilizing composition of the present invention is further selected from the group consisting of hydrochloric acid, sulfuric acid, citric acid, succinic acid, acetic acid, lactic acid, phosphoric acid, malic acid, and salts thereof, and alkali metal hydroxides. It is desirable to include at least one of
These compounds can be used to adjust the pH of the cleaning and sterilizing composition of the present invention.
本発明の洗浄殺菌組成物のpHは、1.5〜6.0であることが望ましい。
洗浄殺菌組成物のpHが上記範囲であると、洗浄殺菌組成物を皮膚と接触させたとしても、皮膚に対する刺激が少なくなる。
The cleaning and sterilizing composition of the present invention preferably has a pH of 1.5 to 6.0.
When the pH of the cleansing and sterilizing composition is in the above range, even if the cleansing and sterilizing composition is brought into contact with the skin, irritation to the skin is reduced.
本発明の洗浄殺菌組成物は、所定の界面活性剤を含む。
これらの界面活性剤は、洗浄能力に優れている。そのため、手指が汚れていたとしても、その汚れを落とすことができる。
また、本発明の洗浄殺菌組成物はポビドンヨードも含むので、手指の洗浄と同時に、手指の殺菌も行うことができる。
すなわち、1工程で迅速に手指の洗浄及び殺菌を行うことができる。
The cleaning and sterilizing composition of the present invention contains a predetermined surfactant.
These surfactants have excellent cleaning ability. Therefore, even if the fingers are dirty, the dirt can be removed.
Moreover, since the cleaning and sterilizing composition of the present invention also contains povidone iodine, it is possible to sterilize the fingers at the same time as cleaning the fingers.
That is, the fingers can be quickly washed and sterilized in one step.
以下、本発明の洗浄殺菌組成物について具体的な実施形態を示しながら説明する。しかしながら、本発明は、以下の実施形態に限定されるものではなく、本発明の要旨を変更しない範囲において適宜変更して適用することができる。 Hereinafter, the cleaning and sterilizing composition of the present invention will be described with reference to specific embodiments. However, the present invention is not limited to the following embodiments, and may be appropriately modified and applied without departing from the scope of the present invention.
本発明の洗浄殺菌組成物は、界面活性剤と、ポビドンヨードとを含み、上記界面活性剤は、アルカンスルホン酸塩、アルキルアミドプロピルベタイン、アルキルグルコシド、アルキルジメチルアミンオキシド、アルキルベタイン、アルキルベンゼンスルホン酸塩、アルキル硫酸塩、α−オレフィンスルホン酸塩、グリセリン脂肪酸エステル、ショ糖脂肪酸エステル、スルホベタイン、ポリオキシエチレンアルキルエーテル、ポリオキシエチレンアルキルエーテル硫酸塩、ポリオキシエチレンアルキルエーテル酢酸塩、ポリオキシエチレンソルビタン脂肪酸エステル、ラウロイルサルコシン塩からなる群から選択される少なくとも1種であることを特徴とする。 The cleaning and sterilizing composition of the present invention contains a surfactant and povidone iodine, and the surfactant is an alkane sulfonate, an alkylamidopropyl betaine, an alkyl glucoside, an alkyldimethylamine oxide, an alkyl betaine, an alkylbenzene sulfonate. , Alkyl sulfate, α-olefin sulfonate, glycerin fatty acid ester, sucrose fatty acid ester, sulfobetaine, polyoxyethylene alkyl ether, polyoxyethylene alkyl ether sulfate, polyoxyethylene alkyl ether acetate, polyoxyethylene sorbitan It is characterized by being at least one selected from the group consisting of fatty acid ester and lauroyl sarcosine salt.
本発明の洗浄殺菌組成物は、所定の界面活性剤を含む。
これらの界面活性剤は、洗浄能力に優れている。そのため、手指が汚れていたとしても、その汚れを落とすことができる。
また、本発明の洗浄殺菌組成物はポビドンヨードも含むので、手指の洗浄と同時に、手指の殺菌も行うことができる。
すなわち、1工程で迅速に手指の洗浄及び殺菌を行うことができる。
The cleaning and sterilizing composition of the present invention contains a predetermined surfactant.
These surfactants have excellent cleaning ability. Therefore, even if the fingers are dirty, the dirt can be removed.
Moreover, since the cleaning and sterilizing composition of the present invention also contains povidone iodine, it is possible to sterilize the fingers at the same time as cleaning the fingers.
That is, the fingers can be quickly washed and sterilized in one step.
本発明の洗浄殺菌組成物では、洗浄殺菌組成物中のポビドンヨードの濃度は、0.03〜20重量%であることが望ましく、0.15〜10重量%であることがより望ましい。
ポビドンヨードの濃度が0.03重量%未満であると、ポビドンヨードの濃度が低いので、充分な殺菌効果を示さなくなる。
ポビドンヨードの濃度が20重量%を超えると、ポビドンヨードの溶解度が上限に近づき、溶解できない、又は、析出しやすくなる。また、ポビドンヨードによる殺菌効果が上限に近づく。
In the cleaning and sterilizing composition of the present invention, the concentration of povidone iodine in the cleaning and sterilizing composition is preferably 0.03 to 20% by weight, more preferably 0.15 to 10% by weight.
If the concentration of povidone iodine is less than 0.03% by weight, the concentration of povidone iodine is too low to show a sufficient bactericidal effect.
When the concentration of povidone iodine exceeds 20% by weight, the solubility of povidone iodine approaches the upper limit, and the povidone iodine cannot be dissolved or is easily precipitated. Also, the bactericidal effect of povidone iodine approaches the upper limit.
本発明の洗浄殺菌組成物中の有効ヨウ素の濃度は、30〜20000ppmであることが望ましく、150〜10000ppmであることがより望ましい。 The concentration of available iodine in the cleaning and sterilizing composition of the present invention is preferably 30 to 20,000 ppm, more preferably 150 to 10,000 ppm.
本発明の洗浄殺菌組成物に含まれる望ましい界面活性剤を以下に例示する。
アルカンスルホン酸塩としては、アルカンスルホン酸ナトリウムが挙げられる。
アルキルアミドプロピルベタインとしては、ラウリン酸アミドプロピルベタイン、ヤシ油脂肪酸アミドプロピルベタインが挙げられる。
アルキルグルコシドとしては、デシルグルコシド、ラウリルグルコシド、ヤシ油アルキルグルコシドが挙げられる。
アルキルジメチルアミンオキシドとしては、ラウリルジメチルアミンオキシド、ヤシ油アルキルジメチルアミンオキシドが挙げられる。
アルキルベタインとしては、ラウリルベタイン、ステアリルベタイン、ヤシ油アルキルベタインが挙げられる。
アルキルベンゼンスルホン酸塩としては、デシルベンゼンスルホン酸ナトリウム、ドデシルベンゼンスルホン酸ナトリウム、テトラシルベンゼンスルホン酸ナトリウムが挙げられる。
アルキル硫酸塩としては、ラウリル硫酸ナトリウム、ラウリル硫酸アンモニウム、ラウリル硫酸トリエタノールアミンが挙げられる。
α−オレフィンスルホン酸塩としては、α−オレフィンスルホン酸ナトリウムが挙げられる。
グリセリン脂肪酸エステルとしては、ポリグリセリンカプリル酸エステル、ポリグリセリンカプリン酸エステル、ポリグリセリンラウリン酸エステルが挙げられる。
ショ糖脂肪酸エステルとしては、ショ糖ラウリン酸エステル、ショ糖ミリスチン酸エステルが挙げられる。
スルホベタインとしては、ラウリルヒドロキシスルホベタインが挙げられる。
ポリオキシエチレンアルキルエーテルとしては、ポリオキシエチレンラウリルエーテルが挙げられる。
ポリオキシエチレンアルキルエーテル硫酸塩としては、ポリオキシエチレンラウリルエーテル硫酸ナトリウム、ポリオキシエチレンラウリルエーテル硫酸アンモニウム、ポリオキシエチレンラウリルエーテル硫酸トリエタノールアミンが挙げられる。
ポリオキシエチレンアルキルエーテル酢酸塩としては、ポリオキシエチレンアルキルエーテル酢酸ナトリウムが挙げられる。
ポリオキシエチレンソルビタン脂肪酸エステルとしては、ポリオキシエチレンソルビタンモノラウレート(ポリソルベート20)、ポリオキシエチレンソルビタンモノパルミテート(ポリソルベート40)、ポリオキシエチレンソルビタンモノステアレート(ポリソルベート60)、ポリオキシエチレンソルビタンモノオレート(ポリソルベート80)が挙げられる。
ラウロイルサルコシン塩としては、ラウロイルサルコシンナトリウム、ラウロイルサルコシンカリウム、ラウロイルサルコシントリエタノールアミンが挙げられる。
Examples of desirable surfactants contained in the cleaning and sterilizing composition of the present invention are shown below.
Examples of the alkane sulfonate include sodium alkane sulfonate.
Examples of the alkylamidopropyl betaine include lauric acid amidopropyl betaine and coconut oil fatty acid amidopropyl betaine.
Examples of the alkyl glucoside include decyl glucoside, lauryl glucoside, and coconut oil alkyl glucoside.
Examples of the alkyl dimethyl amine oxide include lauryl dimethyl amine oxide and coconut oil alkyl dimethyl amine oxide.
Examples of alkyl betaines include lauryl betaine, stearyl betaine, and coconut oil alkyl betaine.
Examples of the alkylbenzene sulfonate include sodium decylbenzene sulfonate, sodium dodecylbenzene sulfonate, and sodium tetrasylbenzene sulfonate.
Examples of the alkyl sulfates include sodium lauryl sulfate, ammonium lauryl sulfate, and triethanolamine lauryl sulfate.
Examples of the α-olefin sulfonate include sodium α-olefin sulfonate.
Examples of the glycerin fatty acid ester include polyglycerin caprylic acid ester, polyglycerin capric acid ester, and polyglycerin lauric acid ester.
Examples of the sucrose fatty acid ester include sucrose laurate and sucrose myristate.
Examples of sulfobetaines include lauryl hydroxysulfobetaine.
Examples of the polyoxyethylene alkyl ether include polyoxyethylene lauryl ether.
Examples of the polyoxyethylene alkyl ether sulfate include sodium polyoxyethylene lauryl ether sulfate, ammonium polyoxyethylene lauryl ether sulfate, and triethanolamine polyoxyethylene lauryl ether sulfate.
Examples of the polyoxyethylene alkyl ether acetate include sodium polyoxyethylene alkyl ether acetate.
Examples of the polyoxyethylene sorbitan fatty acid ester include polyoxyethylene sorbitan monolaurate (polysorbate 20), polyoxyethylene sorbitan monopalmitate (polysorbate 40), polyoxyethylene sorbitan monostearate (polysorbate 60), polyoxyethylene sorbitan mono Examples include oleate (polysorbate 80).
Examples of the lauroyl sarcosine salt include lauroyl sarcosine sodium, lauroyl sarcosine potassium, and lauroyl sarcosine triethanolamine.
これらの界面活性剤は、起泡性にも優れるので、泡ポンプ等によりわざわざ泡状にしなくても、手でこすり合わせるだけで泡立てることができる。このような泡は、洗浄力にも寄与する。 Since these surfactants also have excellent foaming properties, they can be foamed by rubbing them by hand without making them foamy with a foam pump or the like. Such bubbles also contribute to detergency.
さらに、これらの界面活性剤は、低温安定性にも優れるので、低温で保管した場合であったとしても析出しにくい。 Furthermore, since these surfactants are also excellent in low temperature stability, they are unlikely to precipitate even when stored at low temperatures.
本発明の洗浄殺菌組成物では、上記洗浄殺菌組成物中の上記界面活性剤の濃度は、0.01〜10.0重量%であることが望ましく、0.5〜5.0重量%であることがより望ましく、1.0〜4.0重量%であることがさらに望ましい。
界面活性剤の濃度が、0.01重量%未満であると、充分な洗浄力が得られない。
界面活性剤の濃度が、10.0重量%を超えると、界面活性剤が析出しやすくなる。また、性能が頭打ちになり、費用対効果が低下してしまう。
In the cleaning and sterilizing composition of the present invention, the concentration of the surfactant in the cleaning and sterilizing composition is preferably 0.01 to 10.0% by weight, and 0.5 to 5.0% by weight. It is more desirable that it is 1.0 to 4.0% by weight, and it is even more desirable.
If the concentration of the surfactant is less than 0.01% by weight, sufficient detergency cannot be obtained.
When the concentration of the surfactant exceeds 10.0% by weight, the surfactant tends to precipitate. In addition, the performance will reach a ceiling and cost effectiveness will decrease.
また、本発明の洗浄殺菌組成物において、界面活性剤が、アルキル硫酸塩及び/又はポリオキシエチレンアルキルエーテル硫酸塩である場合には、洗浄殺菌組成物中の界面活性剤の濃度は1.0重量%を超えることが望ましく、1.5重量%以上であることがより望ましく、2.0〜4.0重量%であることがさらに望ましい。
界面活性剤が、アルキル硫酸塩及び/又はポリオキシエチレンアルキルエーテル硫酸塩であり、その濃度が1.0重量%を超えると、洗浄殺菌組成物の洗浄力及び泡立ちが良好になる。
Further, in the cleaning and sterilizing composition of the present invention, when the surfactant is an alkyl sulfate and / or a polyoxyethylene alkyl ether sulfate, the concentration of the surfactant in the cleaning and sterilizing composition is 1.0. It is preferably more than 1.5% by weight, more preferably 1.5% by weight or more, still more preferably 2.0 to 4.0% by weight.
When the surfactant is an alkyl sulfate and / or a polyoxyethylene alkyl ether sulfate, and the concentration thereof exceeds 1.0% by weight, the detergency and foaming of the cleaning / sterilizing composition will be good.
本発明の洗浄殺菌組成物は、さらに、塩酸、硫酸、クエン酸、コハク酸、酢酸、乳酸、リン酸、リンゴ酸、及び、これらの塩、並びに、アルカリ金属水酸化物からなる群から選択される少なくとも1種を含むことが望ましい。
クエン酸、コハク酸、酢酸、乳酸、リン酸及びリンゴ酸の塩としては、アルカリ金属塩やアルカリ土類金属塩が挙げられ、ナトリウム塩、カリウム塩であることが望ましい。
アルカリ金属水酸化物としては、水酸化ナトリウム、水酸化カリウムが挙げられる。
これらの化合物を用いて本発明の洗浄殺菌組成物のpHを調整することができる。
The cleaning and sterilizing composition of the present invention is further selected from the group consisting of hydrochloric acid, sulfuric acid, citric acid, succinic acid, acetic acid, lactic acid, phosphoric acid, malic acid, and salts thereof, and alkali metal hydroxides. It is desirable to include at least one of
Examples of the salts of citric acid, succinic acid, acetic acid, lactic acid, phosphoric acid and malic acid include alkali metal salts and alkaline earth metal salts, and sodium salts and potassium salts are preferable.
Examples of the alkali metal hydroxide include sodium hydroxide and potassium hydroxide.
These compounds can be used to adjust the pH of the cleaning and sterilizing composition of the present invention.
本発明の洗浄殺菌組成物のpHは、1.5〜6.0であることが望ましく、3.0〜5.0であることがより望ましい。
洗浄殺菌組成物のpHが上記範囲であると、洗浄殺菌組成物を皮膚と接触させたとしても、皮膚に対する刺激が少なくなる。
The pH of the cleaning / sterilizing composition of the present invention is preferably 1.5 to 6.0, and more preferably 3.0 to 5.0.
When the pH of the cleansing and sterilizing composition is in the above range, even if the cleansing and sterilizing composition is brought into contact with the skin, irritation to the skin is reduced.
本発明の洗浄殺菌組成物は、その他の組成物として、ヨウ化カリウム、セルロース、グリセリン、プロピレングリコール、ポリエチレングリコール、サッカリン、L−メントール及びエタノールからなる群から選択される少なくとも1種を含んでいてもよい。
これらの化合物は、安定化剤、保湿剤、甘味剤、矯味剤、溶剤、着香剤等として機能する。
The cleaning and sterilizing composition of the present invention contains, as another composition, at least one selected from the group consisting of potassium iodide, cellulose, glycerin, propylene glycol, polyethylene glycol, saccharin, L-menthol and ethanol. Good.
These compounds function as a stabilizer, a moisturizer, a sweetener, a flavoring agent, a solvent, a flavoring agent, and the like.
本発明の洗浄殺菌組成物は、液体のまま用いてもよく、泡状にして用いてもよい。
なお、洗浄殺菌組成物は、界面活性剤を含むので、泡ポンプ等を用いることにより容易に泡状にすることができる。
The cleaning and sterilizing composition of the present invention may be used as a liquid as it is or in the form of foam.
Since the cleaning / sterilizing composition contains a surfactant, it can be easily foamed by using a foam pump or the like.
また、本発明の洗浄殺菌組成物は、手指を洗浄・殺菌する目的で使用する以外にも、創傷部等の生体部位を洗浄・殺菌する目的で使用してもよい。
また、鉗子等の医療器具、まな板等の調理器具、皿等の食器等の器具を洗浄・殺菌する目的で使用してもよい。
Further, the cleaning / sterilizing composition of the present invention may be used for the purpose of cleaning / sterilizing a living body site such as a wound, in addition to the purpose of cleaning / sterilizing fingers.
Further, it may be used for the purpose of cleaning and sterilizing medical instruments such as forceps, cooking instruments such as cutting boards, and dishes such as dishes.
以下に本発明をより具体的に説明する実施例を示すが、本発明はこれらの実施例に限定されるものではない。 Examples that more specifically describe the present invention will be shown below, but the present invention is not limited to these examples.
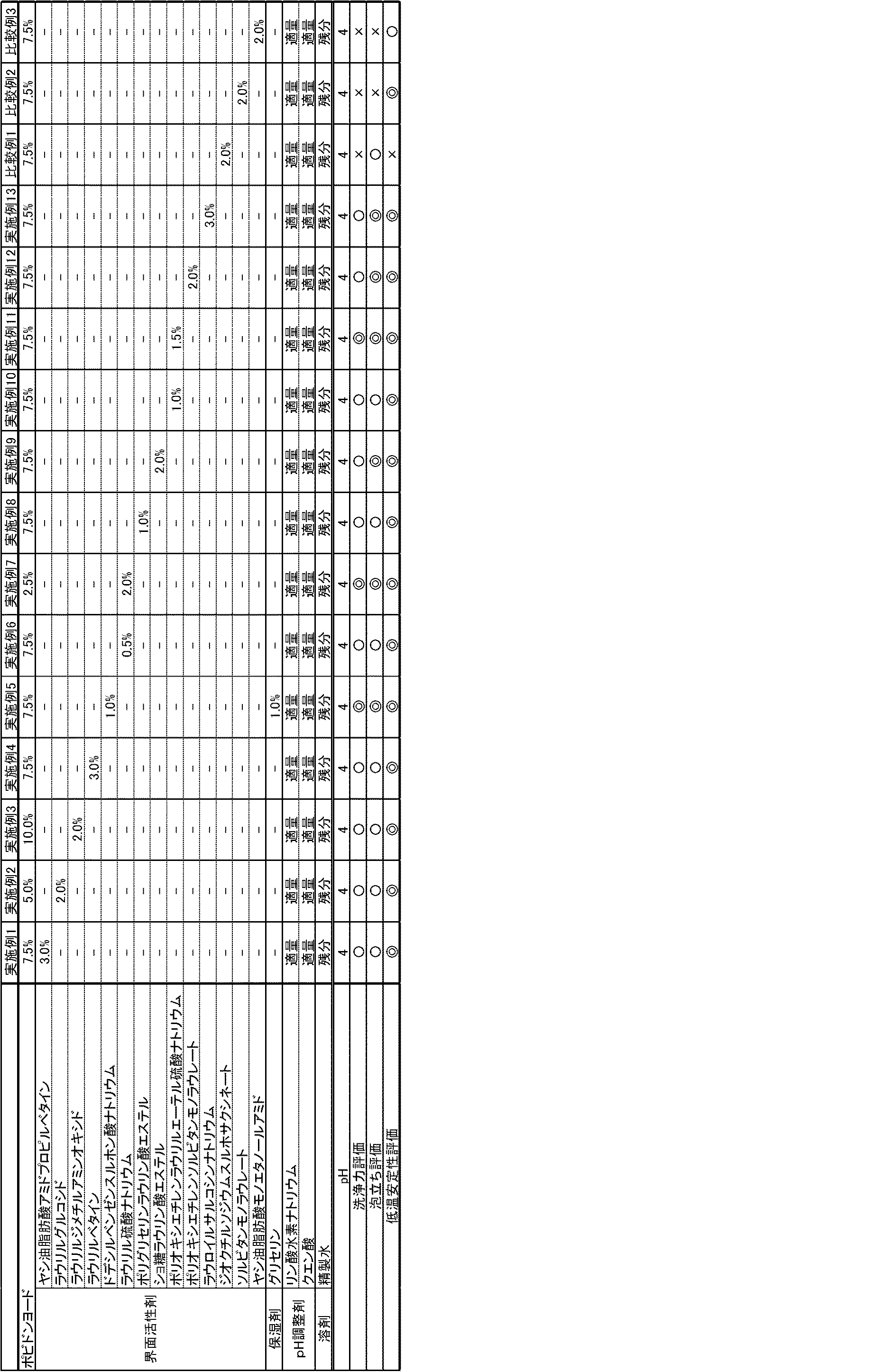
(実施例1〜13)並びに(比較例1〜3)
表1に示す配合量で実施例1〜13に係る洗浄殺菌組成物、並びに、比較例1〜3に係る洗浄殺菌組成物を作製した。
なお、表1中の「%」は、「重量%」を意味する。
(Examples 1 to 13) and (Comparative examples 1 to 3)
The cleaning and sterilizing compositions according to Examples 1 to 13 and the cleaning and sterilizing compositions according to Comparative Examples 1 to 3 were produced with the compounding amounts shown in Table 1.
In addition, "%" in Table 1 means "weight%."
(洗浄力評価)
各実施例及び各比較例に係る洗浄殺菌組成物の洗浄力を以下の方法により評価した。
(1)モデル汚れの作製
牛脂大豆レシチン、モノオレイン、白色ワセリン及びカーボンブラックを、重量比でそれぞれ、6:6:6:1となるように混合しモデル汚れとした。
(2)モデル汚れの付着
モデル汚れ0.1gをあらかじめ手洗いをして乾燥させた両手のひらに広げ、モデル汚れを付着させた。
(3)モデル汚れの洗浄
次に、モデル汚れを付着させた手のひらを水で少し濡らし、各洗浄殺菌組成物を1mL手のひらに落とし、15秒間手のひらを擦り合わせた後、15秒間流水で手のひらをすすいだ。
(4)モデル汚れの洗浄の観察
次に、手のひらを観察し、モデル汚れが完全に落ちているか否かを確認した。
モデル汚れが完全に落ちていない場合には、モデル汚れが完全に落ちるまで上記「(3)モデル汚れの洗浄」を繰り返した。
(Evaluation of detergency)
The cleaning power of the cleaning and sterilizing composition according to each example and each comparative example was evaluated by the following method.
(1) Preparation of model stains Beef tallow soy lecithin, monoolein, white petrolatum and carbon black were mixed at a weight ratio of 6: 6: 6: 1 to obtain model stains.
(2) Adhesion of model stains Model stains of 0.1 g were spread on the palms that had been washed by hand and dried to adhere the model stains.
(3) Washing of model dirt Next, the palm to which the model dirt has been attached is slightly wetted with water, 1 mL of each washing and sterilizing composition is dropped on the palm, the palm is rubbed for 15 seconds, and then the palm is rinsed with running water for 15 seconds. It is.
(4) Observation of washing model stains Next, the palm was observed to confirm whether the model stains were completely removed.
When the model stain is not completely removed, the above “(3) Model stain cleaning” is repeated until the model stain is completely removed.
洗浄力評価の結果を表1に示す。なお、評価基準は以下の通りである。
◎:「(3)モデル汚れの洗浄」を1〜2回行うことにより、モデル汚れが完全に落ちた。
〇:「(3)モデル汚れの洗浄」を3回行うことにより、モデル汚れが完全に落ちた。
×:「(3)モデル汚れの洗浄」を4回以上行うことにより、モデル汚れが完全に落ちた。
The results of the detergency evaluation are shown in Table 1. The evaluation criteria are as follows.
⊚: The model stain was completely removed by performing “(3) model stain cleaning” once or twice.
◯: The model dirt was completely removed by performing “(3) Model dirt cleaning” three times.
X: The model stain was completely removed by performing "(3) Cleaning the model stain" four times or more.
(泡立ち評価)
各実施例及び各比較例に係る洗浄殺菌組成物1mLを手のひらに落とし、15秒間擦り合わせた後、泡の状態を目視で観察することにより、各実施例及び各比較例に係る洗浄殺菌組成物の泡立ち評価を行った。
(Bubbling evaluation)
1 mL of the cleaning and sterilizing composition according to each example and each comparative example was dropped on the palm, rubbed for 15 seconds, and then visually observed for the state of bubbles, whereby the cleaning and sterilizing composition according to each example and each comparative example was observed. Was evaluated.
泡立ち評価の結果を表1に示す。なお、評価基準は以下の通りである。
◎:きめ細かい泡が形成される。
〇:大きな泡が形成される。
×:泡が形成されない。
The results of foaming evaluation are shown in Table 1. The evaluation criteria are as follows.
⊚: Fine bubbles are formed.
◯: Large bubbles are formed.
X: No bubbles are formed.
(低温安定性評価)
各実施例及び各比較例に係る洗浄殺菌組成物の低温安定性を以下の方法により評価した。
各実施例及び各比較例に係る洗浄殺菌組成物100mLを透明ポリプロピレン製容器に入れ、−7℃に設定した保管庫に保管し、凍結させ、1日後に取り出して室温に静置して自然融解させたときに、容器内の洗浄殺菌成物中に析出物や沈殿、分離等が発生しているかを確認した。さらに、洗浄殺菌組成物を取り出して泡ポンプ容器に充填して、液が吐出されるか確認した。
(Low temperature stability evaluation)
The low temperature stability of the cleaning and sterilizing composition according to each example and each comparative example was evaluated by the following method.
100 mL of the cleaning and sterilizing composition according to each example and each comparative example was placed in a transparent polypropylene container, stored in a storage set at -7 ° C, frozen, taken out one day later, and allowed to stand at room temperature to spontaneously melt. Then, it was confirmed whether or not precipitates, precipitates, separations, etc., were generated in the cleaning and sterilizing composition in the container. Further, the cleaning / sterilizing composition was taken out and filled in a foam pump container, and it was confirmed whether the liquid was discharged.
低温安定性評価の結果を表1に示す。なお、評価基準は以下の通りである。
◎:析出物や沈殿、分離等が発生しない。
〇:析出物や沈殿が発生するが、泡ポンプ容器から吐出するときに詰りが生じず、液が吐出する。
×:析出物や沈殿が発生し、泡ポンプ容器から吐出するときに詰りが生じて、液が吐出しない。
The results of low temperature stability evaluation are shown in Table 1. The evaluation criteria are as follows.
⊚: No precipitates, precipitates, separations, etc.
◯: Precipitates and precipitates are generated, but when discharged from the foam pump container, clogging does not occur and the liquid is discharged.
X: Precipitate or precipitate is generated, clogging occurs when discharging from the foam pump container, and the liquid does not discharge.
表1に示すように、各実施例に係る洗浄殺菌組成物は、洗浄力、泡立ち、低温安定性に優れていることが判明した。 As shown in Table 1, it was found that the cleaning and sterilizing composition according to each example was excellent in detergency, foaming and low temperature stability.
Claims (6)
ポビドンヨードとを含み、
前記界面活性剤は、アルカンスルホン酸塩、アルキルアミドプロピルベタイン、アルキルグルコシド、アルキルジメチルアミンオキシド、アルキルベタイン、アルキルベンゼンスルホン酸塩、アルキル硫酸塩、α−オレフィンスルホン酸塩、グリセリン脂肪酸エステル、ショ糖脂肪酸エステル、スルホベタイン、ポリオキシエチレンアルキルエーテル、ポリオキシエチレンアルキルエーテル硫酸塩、ポリオキシエチレンアルキルエーテル酢酸塩、ポリオキシエチレンソルビタン脂肪酸エステル、ラウロイルサルコシン塩からなる群から選択される少なくとも1種であることを特徴とする洗浄殺菌組成物。 A surfactant,
Including povidone-iodine,
Examples of the surfactant include alkane sulfonate, alkylamidopropyl betaine, alkyl glucoside, alkyl dimethylamine oxide, alkyl betaine, alkylbenzene sulfonate, alkyl sulfate, α-olefin sulfonate, glycerin fatty acid ester, sucrose fatty acid. At least one selected from the group consisting of ester, sulfobetaine, polyoxyethylene alkyl ether, polyoxyethylene alkyl ether sulfate, polyoxyethylene alkyl ether acetate, polyoxyethylene sorbitan fatty acid ester, and lauroyl sarcosine salt. A cleaning and sterilizing composition comprising:
前記洗浄殺菌組成物中の前記界面活性剤の濃度は1.0重量%を超える請求項1に記載の洗浄殺菌組成物。 The surfactant is an alkyl sulfate and / or a polyoxyethylene alkyl ether sulfate,
The cleaning and sterilizing composition according to claim 1, wherein the concentration of the surfactant in the cleaning and sterilizing composition exceeds 1.0% by weight.
Priority Applications (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| JP2018210531A JP2020075889A (en) | 2018-11-08 | 2018-11-08 | Washing and sterilizing compositions |
Applications Claiming Priority (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| JP2018210531A JP2020075889A (en) | 2018-11-08 | 2018-11-08 | Washing and sterilizing compositions |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| JP2020075889A true JP2020075889A (en) | 2020-05-21 |
Family
ID=70724877
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| JP2018210531A Pending JP2020075889A (en) | 2018-11-08 | 2018-11-08 | Washing and sterilizing compositions |
Country Status (1)
| Country | Link |
|---|---|
| JP (1) | JP2020075889A (en) |
Cited By (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US11937602B2 (en) | 2017-09-26 | 2024-03-26 | Ecolab Usa Inc. | Solid acid/anionic antimicrobial and virucidal compositions and uses thereof |
Citations (12)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO1986005359A1 (en) * | 1985-03-13 | 1986-09-25 | Gluck Bruno A | Antiseptic compositions |
| JPH01153644A (en) * | 1987-12-10 | 1989-06-15 | Meiji Seika Kaisha Ltd | Germicidal composition |
| JPH01290608A (en) * | 1988-05-18 | 1989-11-22 | Sunstar Inc | Germicidal composition |
| JP2002505894A (en) * | 1998-03-12 | 2002-02-26 | ザ、プロクター、エンド、ギャンブル、カンパニー | Tissue paper containing antibacterial skin lotion |
| JP2002241287A (en) * | 2001-02-20 | 2002-08-28 | Iwaki Seiyaku Co Ltd | Cutaneous ulcer-curing composition |
| JP2005054091A (en) * | 2003-08-06 | 2005-03-03 | Sunstar Inc | Detergent composition |
| JP2005306843A (en) * | 2004-03-25 | 2005-11-04 | Kose Corp | Skin detergent composition |
| JP2006117539A (en) * | 2004-10-19 | 2006-05-11 | Shiseido Co Ltd | Oily ointment |
| JP2009132694A (en) * | 2007-10-31 | 2009-06-18 | Meiji Seika Kaisha Ltd | Foam stabilizer composition |
| JP2011168553A (en) * | 2010-02-19 | 2011-09-01 | Cosmecure Co Ltd | Skin detergent |
| WO2011108245A1 (en) * | 2010-03-01 | 2011-09-09 | 株式会社オフテクス | Aqueous solution for cleaning contact lenses and kit for cleaning contact lenses comprising same |
| JP2020075888A (en) * | 2018-11-08 | 2020-05-21 | 株式会社ニイタカ | Washing and sterilizing compositions |
-
2018
- 2018-11-08 JP JP2018210531A patent/JP2020075889A/en active Pending
Patent Citations (12)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO1986005359A1 (en) * | 1985-03-13 | 1986-09-25 | Gluck Bruno A | Antiseptic compositions |
| JPH01153644A (en) * | 1987-12-10 | 1989-06-15 | Meiji Seika Kaisha Ltd | Germicidal composition |
| JPH01290608A (en) * | 1988-05-18 | 1989-11-22 | Sunstar Inc | Germicidal composition |
| JP2002505894A (en) * | 1998-03-12 | 2002-02-26 | ザ、プロクター、エンド、ギャンブル、カンパニー | Tissue paper containing antibacterial skin lotion |
| JP2002241287A (en) * | 2001-02-20 | 2002-08-28 | Iwaki Seiyaku Co Ltd | Cutaneous ulcer-curing composition |
| JP2005054091A (en) * | 2003-08-06 | 2005-03-03 | Sunstar Inc | Detergent composition |
| JP2005306843A (en) * | 2004-03-25 | 2005-11-04 | Kose Corp | Skin detergent composition |
| JP2006117539A (en) * | 2004-10-19 | 2006-05-11 | Shiseido Co Ltd | Oily ointment |
| JP2009132694A (en) * | 2007-10-31 | 2009-06-18 | Meiji Seika Kaisha Ltd | Foam stabilizer composition |
| JP2011168553A (en) * | 2010-02-19 | 2011-09-01 | Cosmecure Co Ltd | Skin detergent |
| WO2011108245A1 (en) * | 2010-03-01 | 2011-09-09 | 株式会社オフテクス | Aqueous solution for cleaning contact lenses and kit for cleaning contact lenses comprising same |
| JP2020075888A (en) * | 2018-11-08 | 2020-05-21 | 株式会社ニイタカ | Washing and sterilizing compositions |
Cited By (2)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US11937602B2 (en) | 2017-09-26 | 2024-03-26 | Ecolab Usa Inc. | Solid acid/anionic antimicrobial and virucidal compositions and uses thereof |
| US11950595B2 (en) | 2017-09-26 | 2024-04-09 | Ecolab Usa Inc. | Acid/anionic antimicrobial and virucidal compositions and uses thereof |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| ES2749962T3 (en) | Ready-to-use, reduced corrosion, storage-stable peroxycarboxylic acid antimicrobial compositions | |
| ES2383039T3 (en) | Disinfectant composition that includes glycerol monoalkyl ethers and bispyridinioalkanes and their use as a skin antiseptic | |
| US9730870B2 (en) | Sodium hypochlorite-based body wash compositions | |
| JP2023059954A (en) | Washing and sterilizing compositions | |
| US8193244B1 (en) | Antimicrobial agents | |
| BRPI0612409B1 (en) | COMPOSITIONS OF PEROXICARBOXYLIC ACID-BASED TENSOTIVES AND METHOD OF REDUCING THE POPULATION OF MICROORGANISMS ON AN OBJECT | |
| JP2009173768A (en) | Sterilizing detergent composition for toilet seat, sterilizing cleaning material containing the same, and sterilizing cleaning method using them | |
| JP2008297270A (en) | Quick-drying disinfectant and method for producing the same | |
| CN104797234B (en) | Mild antibiotic cleaning compositions | |
| KR100303721B1 (en) | Sterilization Cleaning Composition | |
| JP2023075279A (en) | Non-soap liquid cleanser composition comprising caprylic acid | |
| JP2020075889A (en) | Washing and sterilizing compositions | |
| US3118842A (en) | Multi-purpose cleaner-germicide-deodorizer composition | |
| JP2006347914A (en) | Skin cleaner composition | |
| JP2656301B2 (en) | Hand sanitizer cleaning agent | |
| CA3012079C (en) | Sanitising composition | |
| JP4638348B2 (en) | Food grade disinfecting composition | |
| JP4130383B2 (en) | Cleaning composition for medical equipment | |
| BR112021006553A2 (en) | antimicrobial compositions containing c3-c5 n-alkyl-gamma-butyrolactam and uses thereof | |
| JP6997560B2 (en) | Hair cleaning composition and hair cleaning method | |
| JPS60120800A (en) | Detergent composition | |
| KR20130058531A (en) | Dishwashing liquid detergent composition | |
| KR960002395B1 (en) | Antiseptic compositions | |
| CN106214513A (en) | Medical worker is with faintly acid handwashing liquid and preparation method thereof | |
| BR112015012443B1 (en) | CLEANING COMPOSITION, CLEANING METHOD, USE OF COMPOSITION AND METHOD TO PROVIDE CLEANING |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| A621 | Written request for application examination |
Free format text: JAPANESE INTERMEDIATE CODE: A621 Effective date: 20211012 |
|
| A977 | Report on retrieval |
Free format text: JAPANESE INTERMEDIATE CODE: A971007 Effective date: 20220627 |
|
| A131 | Notification of reasons for refusal |
Free format text: JAPANESE INTERMEDIATE CODE: A131 Effective date: 20220628 |
|
| A601 | Written request for extension of time |
Free format text: JAPANESE INTERMEDIATE CODE: A601 Effective date: 20220803 |
|
| A521 | Request for written amendment filed |
Free format text: JAPANESE INTERMEDIATE CODE: A523 Effective date: 20221027 |
|
| A02 | Decision of refusal |
Free format text: JAPANESE INTERMEDIATE CODE: A02 Effective date: 20230104 |
|
| A521 | Request for written amendment filed |
Free format text: JAPANESE INTERMEDIATE CODE: A523 Effective date: 20230404 |
|
| C60 | Trial request (containing other claim documents, opposition documents) |
Free format text: JAPANESE INTERMEDIATE CODE: C60 Effective date: 20230404 |
|
| A911 | Transfer to examiner for re-examination before appeal (zenchi) |
Free format text: JAPANESE INTERMEDIATE CODE: A911 Effective date: 20230424 |
|
| C21 | Notice of transfer of a case for reconsideration by examiners before appeal proceedings |
Free format text: JAPANESE INTERMEDIATE CODE: C21 Effective date: 20230425 |
|
| A912 | Re-examination (zenchi) completed and case transferred to appeal board |
Free format text: JAPANESE INTERMEDIATE CODE: A912 Effective date: 20230616 |