JP2012531534A - 新規なポリ尿素系繊維 - Google Patents
新規なポリ尿素系繊維 Download PDFInfo
- Publication number
- JP2012531534A JP2012531534A JP2012517720A JP2012517720A JP2012531534A JP 2012531534 A JP2012531534 A JP 2012531534A JP 2012517720 A JP2012517720 A JP 2012517720A JP 2012517720 A JP2012517720 A JP 2012517720A JP 2012531534 A JP2012531534 A JP 2012531534A
- Authority
- JP
- Japan
- Prior art keywords
- nmp
- solution
- polymer
- anhydrous
- polyurea
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Pending
Links
- 229920002396 Polyurea Polymers 0.000 title claims abstract description 101
- 239000000835 fiber Substances 0.000 title claims abstract description 65
- 125000003118 aryl group Chemical group 0.000 claims abstract description 59
- SECXISVLQFMRJM-UHFFFAOYSA-N N-Methylpyrrolidone Chemical compound CN1CCCC1=O SECXISVLQFMRJM-UHFFFAOYSA-N 0.000 claims description 159
- 229920000642 polymer Polymers 0.000 claims description 107
- CBCKQZAAMUWICA-UHFFFAOYSA-N 1,4-phenylenediamine Chemical compound NC1=CC=C(N)C=C1 CBCKQZAAMUWICA-UHFFFAOYSA-N 0.000 claims description 35
- ALQLPWJFHRMHIU-UHFFFAOYSA-N 1,4-diisocyanatobenzene Chemical group O=C=NC1=CC=C(N=C=O)C=C1 ALQLPWJFHRMHIU-UHFFFAOYSA-N 0.000 claims description 34
- 238000000034 method Methods 0.000 claims description 34
- LFQSCWFLJHTTHZ-UHFFFAOYSA-N Ethanol Chemical compound CCO LFQSCWFLJHTTHZ-UHFFFAOYSA-N 0.000 claims description 30
- 239000004202 carbamide Substances 0.000 claims description 28
- XSQUKJJJFZCRTK-UHFFFAOYSA-N Urea Chemical compound NC(N)=O XSQUKJJJFZCRTK-UHFFFAOYSA-N 0.000 claims description 22
- UXVMQQNJUSDDNG-UHFFFAOYSA-L Calcium chloride Chemical class [Cl-].[Cl-].[Ca+2] UXVMQQNJUSDDNG-UHFFFAOYSA-L 0.000 claims description 16
- 239000001110 calcium chloride Substances 0.000 claims description 14
- 229910001628 calcium chloride Inorganic materials 0.000 claims description 14
- 238000004519 manufacturing process Methods 0.000 claims description 13
- 230000008859 change Effects 0.000 claims description 7
- 230000008569 process Effects 0.000 claims description 7
- 238000001035 drying Methods 0.000 claims description 5
- 238000013019 agitation Methods 0.000 claims description 4
- 238000001914 filtration Methods 0.000 claims description 4
- 150000002576 ketones Chemical class 0.000 claims description 2
- 238000010189 synthetic method Methods 0.000 abstract description 4
- 230000007613 environmental effect Effects 0.000 abstract 1
- 239000000243 solution Substances 0.000 description 63
- 239000000523 sample Substances 0.000 description 58
- 239000000047 product Substances 0.000 description 47
- 229910052739 hydrogen Inorganic materials 0.000 description 44
- 238000006243 chemical reaction Methods 0.000 description 43
- 239000001257 hydrogen Substances 0.000 description 43
- 239000000463 material Substances 0.000 description 36
- 229920003235 aromatic polyamide Polymers 0.000 description 27
- 239000000376 reactant Substances 0.000 description 24
- XLYOFNOQVPJJNP-UHFFFAOYSA-N water Substances O XLYOFNOQVPJJNP-UHFFFAOYSA-N 0.000 description 24
- IJGRMHOSHXDMSA-UHFFFAOYSA-N Atomic nitrogen Chemical compound N#N IJGRMHOSHXDMSA-UHFFFAOYSA-N 0.000 description 21
- 229920000271 Kevlar® Polymers 0.000 description 21
- 238000002474 experimental method Methods 0.000 description 21
- 238000004458 analytical method Methods 0.000 description 18
- 230000015572 biosynthetic process Effects 0.000 description 18
- 238000001816 cooling Methods 0.000 description 18
- 239000002904 solvent Substances 0.000 description 18
- 239000000126 substance Substances 0.000 description 17
- 238000009826 distribution Methods 0.000 description 16
- 239000000203 mixture Substances 0.000 description 16
- 229910052757 nitrogen Inorganic materials 0.000 description 16
- 238000003786 synthesis reaction Methods 0.000 description 16
- -1 polyparaphenylene terephthalamide Polymers 0.000 description 15
- 239000004760 aramid Substances 0.000 description 14
- 238000005033 Fourier transform infrared spectroscopy Methods 0.000 description 12
- 239000011575 calcium Substances 0.000 description 12
- 125000005442 diisocyanate group Chemical group 0.000 description 12
- 238000005227 gel permeation chromatography Methods 0.000 description 12
- 125000002915 carbonyl group Chemical group [*:2]C([*:1])=O 0.000 description 11
- 150000004985 diamines Chemical class 0.000 description 11
- 238000002156 mixing Methods 0.000 description 11
- 238000005259 measurement Methods 0.000 description 10
- 230000004580 weight loss Effects 0.000 description 10
- 239000003153 chemical reaction reagent Substances 0.000 description 9
- 239000002657 fibrous material Substances 0.000 description 9
- 239000007787 solid Substances 0.000 description 9
- 229920002994 synthetic fiber Polymers 0.000 description 9
- AMQJEAYHLZJPGS-UHFFFAOYSA-N N-Pentanol Chemical compound CCCCCO AMQJEAYHLZJPGS-UHFFFAOYSA-N 0.000 description 8
- 229910001424 calcium ion Inorganic materials 0.000 description 8
- 230000000694 effects Effects 0.000 description 8
- 239000002244 precipitate Substances 0.000 description 8
- 238000001228 spectrum Methods 0.000 description 8
- 238000003756 stirring Methods 0.000 description 8
- 239000012209 synthetic fiber Substances 0.000 description 8
- BHPQYMZQTOCNFJ-UHFFFAOYSA-N Calcium cation Chemical compound [Ca+2] BHPQYMZQTOCNFJ-UHFFFAOYSA-N 0.000 description 7
- JOYRKODLDBILNP-UHFFFAOYSA-N Ethyl urethane Chemical compound CCOC(N)=O JOYRKODLDBILNP-UHFFFAOYSA-N 0.000 description 7
- 229920003369 Kevlar® 49 Polymers 0.000 description 7
- 238000001556 precipitation Methods 0.000 description 7
- BDERNNFJNOPAEC-UHFFFAOYSA-N propan-1-ol Chemical compound CCCO BDERNNFJNOPAEC-UHFFFAOYSA-N 0.000 description 7
- OYPRJOBELJOOCE-UHFFFAOYSA-N Calcium Chemical compound [Ca] OYPRJOBELJOOCE-UHFFFAOYSA-N 0.000 description 6
- CURLTUGMZLYLDI-UHFFFAOYSA-N Carbon dioxide Chemical group O=C=O CURLTUGMZLYLDI-UHFFFAOYSA-N 0.000 description 6
- YMWUJEATGCHHMB-UHFFFAOYSA-N Dichloromethane Chemical compound ClCCl YMWUJEATGCHHMB-UHFFFAOYSA-N 0.000 description 6
- ZMXDDKWLCZADIW-UHFFFAOYSA-N N,N-Dimethylformamide Chemical compound CN(C)C=O ZMXDDKWLCZADIW-UHFFFAOYSA-N 0.000 description 6
- 239000004952 Polyamide Substances 0.000 description 6
- QAOWNCQODCNURD-UHFFFAOYSA-N Sulfuric acid Chemical compound OS(O)(=O)=O QAOWNCQODCNURD-UHFFFAOYSA-N 0.000 description 6
- YXFVVABEGXRONW-UHFFFAOYSA-N Toluene Chemical compound CC1=CC=CC=C1 YXFVVABEGXRONW-UHFFFAOYSA-N 0.000 description 6
- 150000001408 amides Chemical class 0.000 description 6
- 229910052791 calcium Inorganic materials 0.000 description 6
- 238000010790 dilution Methods 0.000 description 6
- 239000012895 dilution Substances 0.000 description 6
- 239000011521 glass Substances 0.000 description 6
- 239000012948 isocyanate Substances 0.000 description 6
- 150000002513 isocyanates Chemical class 0.000 description 6
- 239000007788 liquid Substances 0.000 description 6
- 230000000704 physical effect Effects 0.000 description 6
- 229920002647 polyamide Polymers 0.000 description 6
- 238000001308 synthesis method Methods 0.000 description 6
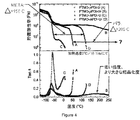
- 238000002411 thermogravimetry Methods 0.000 description 6
- RYHBNJHYFVUHQT-UHFFFAOYSA-N 1,4-Dioxane Chemical compound C1COCCO1 RYHBNJHYFVUHQT-UHFFFAOYSA-N 0.000 description 5
- 238000005481 NMR spectroscopy Methods 0.000 description 5
- 238000000921 elemental analysis Methods 0.000 description 5
- KWGKDLIKAYFUFQ-UHFFFAOYSA-M lithium chloride Chemical compound [Li+].[Cl-] KWGKDLIKAYFUFQ-UHFFFAOYSA-M 0.000 description 5
- 238000002844 melting Methods 0.000 description 5
- 230000008018 melting Effects 0.000 description 5
- 238000002360 preparation method Methods 0.000 description 5
- 238000000425 proton nuclear magnetic resonance spectrum Methods 0.000 description 5
- 238000003860 storage Methods 0.000 description 5
- 239000000725 suspension Substances 0.000 description 5
- NPDACUSDTOMAMK-UHFFFAOYSA-N 4-Chlorotoluene Chemical compound CC1=CC=C(Cl)C=C1 NPDACUSDTOMAMK-UHFFFAOYSA-N 0.000 description 4
- CSCPPACGZOOCGX-UHFFFAOYSA-N Acetone Chemical compound CC(C)=O CSCPPACGZOOCGX-UHFFFAOYSA-N 0.000 description 4
- VEXZGXHMUGYJMC-UHFFFAOYSA-N Hydrochloric acid Chemical compound Cl VEXZGXHMUGYJMC-UHFFFAOYSA-N 0.000 description 4
- 239000004793 Polystyrene Substances 0.000 description 4
- 150000001298 alcohols Chemical class 0.000 description 4
- 150000004984 aromatic diamines Chemical class 0.000 description 4
- 229910052799 carbon Inorganic materials 0.000 description 4
- 239000007795 chemical reaction product Substances 0.000 description 4
- 150000001875 compounds Chemical class 0.000 description 4
- 238000000113 differential scanning calorimetry Methods 0.000 description 4
- 239000000499 gel Substances 0.000 description 4
- 229920002223 polystyrene Polymers 0.000 description 4
- 238000000197 pyrolysis Methods 0.000 description 4
- 238000011160 research Methods 0.000 description 4
- HVLLSGMXQDNUAL-UHFFFAOYSA-N triphenyl phosphite Chemical compound C=1C=CC=CC=1OP(OC=1C=CC=CC=1)OC1=CC=CC=C1 HVLLSGMXQDNUAL-UHFFFAOYSA-N 0.000 description 4
- 239000003643 water by type Substances 0.000 description 4
- ZWEHNKRNPOVVGH-UHFFFAOYSA-N 2-Butanone Chemical compound CCC(C)=O ZWEHNKRNPOVVGH-UHFFFAOYSA-N 0.000 description 3
- IBOFVQJTBBUKMU-UHFFFAOYSA-N 4,4'-methylene-bis-(2-chloroaniline) Chemical compound C1=C(Cl)C(N)=CC=C1CC1=CC=C(N)C(Cl)=C1 IBOFVQJTBBUKMU-UHFFFAOYSA-N 0.000 description 3
- OKTJSMMVPCPJKN-UHFFFAOYSA-N Carbon Chemical compound [C] OKTJSMMVPCPJKN-UHFFFAOYSA-N 0.000 description 3
- 239000004970 Chain extender Substances 0.000 description 3
- UFHFLCQGNIYNRP-UHFFFAOYSA-N Hydrogen Chemical compound [H][H] UFHFLCQGNIYNRP-UHFFFAOYSA-N 0.000 description 3
- 150000001412 amines Chemical class 0.000 description 3
- 125000004429 atom Chemical group 0.000 description 3
- 239000002981 blocking agent Substances 0.000 description 3
- 239000006227 byproduct Substances 0.000 description 3
- 238000012512 characterization method Methods 0.000 description 3
- 230000007423 decrease Effects 0.000 description 3
- 238000011161 development Methods 0.000 description 3
- 230000018109 developmental process Effects 0.000 description 3
- 238000010586 diagram Methods 0.000 description 3
- 238000004090 dissolution Methods 0.000 description 3
- 239000003480 eluent Substances 0.000 description 3
- 238000002347 injection Methods 0.000 description 3
- 239000007924 injection Substances 0.000 description 3
- 230000007246 mechanism Effects 0.000 description 3
- 238000000302 molecular modelling Methods 0.000 description 3
- QJGQUHMNIGDVPM-UHFFFAOYSA-N nitrogen group Chemical group [N] QJGQUHMNIGDVPM-UHFFFAOYSA-N 0.000 description 3
- 125000001997 phenyl group Chemical group [H]C1=C([H])C([H])=C(*)C([H])=C1[H] 0.000 description 3
- 239000002861 polymer material Substances 0.000 description 3
- 238000006116 polymerization reaction Methods 0.000 description 3
- 238000012545 processing Methods 0.000 description 3
- 230000002194 synthesizing effect Effects 0.000 description 3
- 238000012360 testing method Methods 0.000 description 3
- 230000000007 visual effect Effects 0.000 description 3
- 238000005160 1H NMR spectroscopy Methods 0.000 description 2
- 241000408659 Darpa Species 0.000 description 2
- IAZDPXIOMUYVGZ-UHFFFAOYSA-N Dimethylsulphoxide Chemical compound CS(C)=O IAZDPXIOMUYVGZ-UHFFFAOYSA-N 0.000 description 2
- 238000001157 Fourier transform infrared spectrum Methods 0.000 description 2
- 241001112258 Moca Species 0.000 description 2
- LRHPLDYGYMQRHN-UHFFFAOYSA-N N-Butanol Chemical compound CCCCO LRHPLDYGYMQRHN-UHFFFAOYSA-N 0.000 description 2
- 241000209094 Oryza Species 0.000 description 2
- 235000007164 Oryza sativa Nutrition 0.000 description 2
- 239000004721 Polyphenylene oxide Substances 0.000 description 2
- WYURNTSHIVDZCO-UHFFFAOYSA-N Tetrahydrofuran Chemical compound C1CCOC1 WYURNTSHIVDZCO-UHFFFAOYSA-N 0.000 description 2
- 238000010521 absorption reaction Methods 0.000 description 2
- 125000001931 aliphatic group Chemical group 0.000 description 2
- 238000013459 approach Methods 0.000 description 2
- 230000009286 beneficial effect Effects 0.000 description 2
- 238000011088 calibration curve Methods 0.000 description 2
- 239000003054 catalyst Substances 0.000 description 2
- 239000003610 charcoal Substances 0.000 description 2
- 230000009920 chelation Effects 0.000 description 2
- 239000002826 coolant Substances 0.000 description 2
- 230000008878 coupling Effects 0.000 description 2
- 238000010168 coupling process Methods 0.000 description 2
- 238000005859 coupling reaction Methods 0.000 description 2
- 230000001186 cumulative effect Effects 0.000 description 2
- 238000000354 decomposition reaction Methods 0.000 description 2
- 239000008367 deionised water Substances 0.000 description 2
- 229910021641 deionized water Inorganic materials 0.000 description 2
- 229920006240 drawn fiber Polymers 0.000 description 2
- 238000011156 evaluation Methods 0.000 description 2
- 238000001704 evaporation Methods 0.000 description 2
- 230000008020 evaporation Effects 0.000 description 2
- 239000012467 final product Substances 0.000 description 2
- 239000012530 fluid Substances 0.000 description 2
- 230000010354 integration Effects 0.000 description 2
- 238000001000 micrograph Methods 0.000 description 2
- 230000003647 oxidation Effects 0.000 description 2
- 238000007254 oxidation reaction Methods 0.000 description 2
- 239000002245 particle Substances 0.000 description 2
- 238000006068 polycondensation reaction Methods 0.000 description 2
- 229920000570 polyether Polymers 0.000 description 2
- 230000009257 reactivity Effects 0.000 description 2
- 230000004044 response Effects 0.000 description 2
- 235000009566 rice Nutrition 0.000 description 2
- 150000003512 tertiary amines Chemical class 0.000 description 2
- 238000005979 thermal decomposition reaction Methods 0.000 description 2
- 150000003673 urethanes Chemical class 0.000 description 2
- 0 *c(cc1)ccc1N=C=O Chemical compound *c(cc1)ccc1N=C=O 0.000 description 1
- PISLZQACAJMAIO-UHFFFAOYSA-N 2,4-diethyl-6-methylbenzene-1,3-diamine Chemical compound CCC1=CC(C)=C(N)C(CC)=C1N PISLZQACAJMAIO-UHFFFAOYSA-N 0.000 description 1
- HGXVKAPCSIXGAK-UHFFFAOYSA-N 2,4-diethyl-6-methylbenzene-1,3-diamine;4,6-diethyl-2-methylbenzene-1,3-diamine Chemical compound CCC1=CC(CC)=C(N)C(C)=C1N.CCC1=CC(C)=C(N)C(CC)=C1N HGXVKAPCSIXGAK-UHFFFAOYSA-N 0.000 description 1
- XXXRVVGCCGOZOR-UHFFFAOYSA-N C(=O)=[N].[N] Chemical compound C(=O)=[N].[N] XXXRVVGCCGOZOR-UHFFFAOYSA-N 0.000 description 1
- 208000031968 Cadaver Diseases 0.000 description 1
- 229920000049 Carbon (fiber) Polymers 0.000 description 1
- VEXZGXHMUGYJMC-UHFFFAOYSA-M Chloride anion Chemical compound [Cl-] VEXZGXHMUGYJMC-UHFFFAOYSA-M 0.000 description 1
- YZCKVEUIGOORGS-OUBTZVSYSA-N Deuterium Chemical compound [2H] YZCKVEUIGOORGS-OUBTZVSYSA-N 0.000 description 1
- 238000005684 Liebig rearrangement reaction Methods 0.000 description 1
- 108091034117 Oligonucleotide Proteins 0.000 description 1
- JLCPHMBAVCMARE-UHFFFAOYSA-N [3-[[3-[[3-[[3-[[3-[[3-[[3-[[3-[[3-[[3-[[3-[[5-(2-amino-6-oxo-1H-purin-9-yl)-3-[[3-[[3-[[3-[[3-[[3-[[5-(2-amino-6-oxo-1H-purin-9-yl)-3-[[5-(2-amino-6-oxo-1H-purin-9-yl)-3-hydroxyoxolan-2-yl]methoxy-hydroxyphosphoryl]oxyoxolan-2-yl]methoxy-hydroxyphosphoryl]oxy-5-(5-methyl-2,4-dioxopyrimidin-1-yl)oxolan-2-yl]methoxy-hydroxyphosphoryl]oxy-5-(6-aminopurin-9-yl)oxolan-2-yl]methoxy-hydroxyphosphoryl]oxy-5-(6-aminopurin-9-yl)oxolan-2-yl]methoxy-hydroxyphosphoryl]oxy-5-(6-aminopurin-9-yl)oxolan-2-yl]methoxy-hydroxyphosphoryl]oxy-5-(6-aminopurin-9-yl)oxolan-2-yl]methoxy-hydroxyphosphoryl]oxyoxolan-2-yl]methoxy-hydroxyphosphoryl]oxy-5-(5-methyl-2,4-dioxopyrimidin-1-yl)oxolan-2-yl]methoxy-hydroxyphosphoryl]oxy-5-(4-amino-2-oxopyrimidin-1-yl)oxolan-2-yl]methoxy-hydroxyphosphoryl]oxy-5-(5-methyl-2,4-dioxopyrimidin-1-yl)oxolan-2-yl]methoxy-hydroxyphosphoryl]oxy-5-(5-methyl-2,4-dioxopyrimidin-1-yl)oxolan-2-yl]methoxy-hydroxyphosphoryl]oxy-5-(6-aminopurin-9-yl)oxolan-2-yl]methoxy-hydroxyphosphoryl]oxy-5-(6-aminopurin-9-yl)oxolan-2-yl]methoxy-hydroxyphosphoryl]oxy-5-(4-amino-2-oxopyrimidin-1-yl)oxolan-2-yl]methoxy-hydroxyphosphoryl]oxy-5-(4-amino-2-oxopyrimidin-1-yl)oxolan-2-yl]methoxy-hydroxyphosphoryl]oxy-5-(4-amino-2-oxopyrimidin-1-yl)oxolan-2-yl]methoxy-hydroxyphosphoryl]oxy-5-(6-aminopurin-9-yl)oxolan-2-yl]methoxy-hydroxyphosphoryl]oxy-5-(4-amino-2-oxopyrimidin-1-yl)oxolan-2-yl]methyl [5-(6-aminopurin-9-yl)-2-(hydroxymethyl)oxolan-3-yl] hydrogen phosphate Polymers Cc1cn(C2CC(OP(O)(=O)OCC3OC(CC3OP(O)(=O)OCC3OC(CC3O)n3cnc4c3nc(N)[nH]c4=O)n3cnc4c3nc(N)[nH]c4=O)C(COP(O)(=O)OC3CC(OC3COP(O)(=O)OC3CC(OC3COP(O)(=O)OC3CC(OC3COP(O)(=O)OC3CC(OC3COP(O)(=O)OC3CC(OC3COP(O)(=O)OC3CC(OC3COP(O)(=O)OC3CC(OC3COP(O)(=O)OC3CC(OC3COP(O)(=O)OC3CC(OC3COP(O)(=O)OC3CC(OC3COP(O)(=O)OC3CC(OC3COP(O)(=O)OC3CC(OC3COP(O)(=O)OC3CC(OC3COP(O)(=O)OC3CC(OC3COP(O)(=O)OC3CC(OC3COP(O)(=O)OC3CC(OC3COP(O)(=O)OC3CC(OC3CO)n3cnc4c(N)ncnc34)n3ccc(N)nc3=O)n3cnc4c(N)ncnc34)n3ccc(N)nc3=O)n3ccc(N)nc3=O)n3ccc(N)nc3=O)n3cnc4c(N)ncnc34)n3cnc4c(N)ncnc34)n3cc(C)c(=O)[nH]c3=O)n3cc(C)c(=O)[nH]c3=O)n3ccc(N)nc3=O)n3cc(C)c(=O)[nH]c3=O)n3cnc4c3nc(N)[nH]c4=O)n3cnc4c(N)ncnc34)n3cnc4c(N)ncnc34)n3cnc4c(N)ncnc34)n3cnc4c(N)ncnc34)O2)c(=O)[nH]c1=O JLCPHMBAVCMARE-UHFFFAOYSA-N 0.000 description 1
- 238000005299 abrasion Methods 0.000 description 1
- 239000002253 acid Substances 0.000 description 1
- 229920006397 acrylic thermoplastic Polymers 0.000 description 1
- 239000011157 advanced composite material Substances 0.000 description 1
- 239000000010 aprotic solvent Substances 0.000 description 1
- 229920006231 aramid fiber Polymers 0.000 description 1
- 150000004982 aromatic amines Chemical group 0.000 description 1
- QVGXLLKOCUKJST-UHFFFAOYSA-N atomic oxygen Chemical compound [O] QVGXLLKOCUKJST-UHFFFAOYSA-N 0.000 description 1
- 238000005102 attenuated total reflection Methods 0.000 description 1
- 230000004888 barrier function Effects 0.000 description 1
- 230000008901 benefit Effects 0.000 description 1
- 230000033228 biological regulation Effects 0.000 description 1
- 230000000903 blocking effect Effects 0.000 description 1
- 238000004364 calculation method Methods 0.000 description 1
- 230000000711 cancerogenic effect Effects 0.000 description 1
- 239000004917 carbon fiber Substances 0.000 description 1
- 239000011203 carbon fibre reinforced carbon Substances 0.000 description 1
- 238000000160 carbon, hydrogen and nitrogen elemental analysis Methods 0.000 description 1
- 231100000357 carcinogen Toxicity 0.000 description 1
- 239000003183 carcinogenic agent Substances 0.000 description 1
- 230000015556 catabolic process Effects 0.000 description 1
- 239000002738 chelating agent Substances 0.000 description 1
- 150000001805 chlorine compounds Chemical class 0.000 description 1
- 238000000576 coating method Methods 0.000 description 1
- 238000009841 combustion method Methods 0.000 description 1
- 230000000295 complement effect Effects 0.000 description 1
- 239000002131 composite material Substances 0.000 description 1
- 238000005094 computer simulation Methods 0.000 description 1
- 239000012084 conversion product Substances 0.000 description 1
- 239000000498 cooling water Substances 0.000 description 1
- 229920001577 copolymer Polymers 0.000 description 1
- 239000007799 cork Substances 0.000 description 1
- 238000012937 correction Methods 0.000 description 1
- 239000013078 crystal Substances 0.000 description 1
- 230000003247 decreasing effect Effects 0.000 description 1
- 230000007123 defense Effects 0.000 description 1
- 238000006731 degradation reaction Methods 0.000 description 1
- 230000018044 dehydration Effects 0.000 description 1
- 238000006297 dehydration reaction Methods 0.000 description 1
- 229910052805 deuterium Inorganic materials 0.000 description 1
- 125000004427 diamine group Chemical group 0.000 description 1
- 238000009792 diffusion process Methods 0.000 description 1
- 238000006471 dimerization reaction Methods 0.000 description 1
- 238000002845 discoloration Methods 0.000 description 1
- 229920001971 elastomer Polymers 0.000 description 1
- 239000000806 elastomer Substances 0.000 description 1
- 238000005516 engineering process Methods 0.000 description 1
- 150000002148 esters Chemical class 0.000 description 1
- 238000013213 extrapolation Methods 0.000 description 1
- 230000002349 favourable effect Effects 0.000 description 1
- 238000012681 fiber drawing Methods 0.000 description 1
- 238000007380 fibre production Methods 0.000 description 1
- 239000011888 foil Substances 0.000 description 1
- 239000000446 fuel Substances 0.000 description 1
- 125000000524 functional group Chemical group 0.000 description 1
- 230000004927 fusion Effects 0.000 description 1
- 230000014509 gene expression Effects 0.000 description 1
- 230000009477 glass transition Effects 0.000 description 1
- 238000010438 heat treatment Methods 0.000 description 1
- GNOIPBMMFNIUFM-UHFFFAOYSA-N hexamethylphosphoric triamide Chemical compound CN(C)P(=O)(N(C)C)N(C)C GNOIPBMMFNIUFM-UHFFFAOYSA-N 0.000 description 1
- 229920006158 high molecular weight polymer Polymers 0.000 description 1
- 125000004435 hydrogen atom Chemical group [H]* 0.000 description 1
- 125000002887 hydroxy group Chemical group [H]O* 0.000 description 1
- 239000005457 ice water Substances 0.000 description 1
- 230000006872 improvement Effects 0.000 description 1
- 239000012535 impurity Substances 0.000 description 1
- 238000002329 infrared spectrum Methods 0.000 description 1
- 238000013101 initial test Methods 0.000 description 1
- 238000007689 inspection Methods 0.000 description 1
- 230000003993 interaction Effects 0.000 description 1
- 238000010406 interfacial reaction Methods 0.000 description 1
- 239000000543 intermediate Substances 0.000 description 1
- IQPQWNKOIGAROB-UHFFFAOYSA-N isocyanate group Chemical group [N-]=C=O IQPQWNKOIGAROB-UHFFFAOYSA-N 0.000 description 1
- 238000002955 isolation Methods 0.000 description 1
- 239000004761 kevlar Substances 0.000 description 1
- 230000033001 locomotion Effects 0.000 description 1
- 230000014759 maintenance of location Effects 0.000 description 1
- 238000000691 measurement method Methods 0.000 description 1
- 230000004048 modification Effects 0.000 description 1
- 238000012986 modification Methods 0.000 description 1
- 239000000178 monomer Substances 0.000 description 1
- 239000004570 mortar (masonry) Substances 0.000 description 1
- 238000000465 moulding Methods 0.000 description 1
- 239000012299 nitrogen atmosphere Substances 0.000 description 1
- 150000002894 organic compounds Chemical class 0.000 description 1
- 239000011368 organic material Substances 0.000 description 1
- 239000001301 oxygen Substances 0.000 description 1
- 229910052760 oxygen Inorganic materials 0.000 description 1
- 239000013618 particulate matter Substances 0.000 description 1
- 230000037361 pathway Effects 0.000 description 1
- 239000012071 phase Substances 0.000 description 1
- 238000006366 phosphorylation reaction Methods 0.000 description 1
- 229920000075 poly(4-vinylpyridine) Polymers 0.000 description 1
- 229920003229 poly(methyl methacrylate) Polymers 0.000 description 1
- 229920000768 polyamine Polymers 0.000 description 1
- 238000012667 polymer degradation Methods 0.000 description 1
- 229920005594 polymer fiber Polymers 0.000 description 1
- 229920006254 polymer film Polymers 0.000 description 1
- 229920005862 polyol Polymers 0.000 description 1
- 150000003077 polyols Chemical class 0.000 description 1
- 239000004814 polyurethane Substances 0.000 description 1
- 229920003226 polyurethane urea Polymers 0.000 description 1
- 239000000843 powder Substances 0.000 description 1
- 230000001681 protective effect Effects 0.000 description 1
- 238000000746 purification Methods 0.000 description 1
- 238000010526 radical polymerization reaction Methods 0.000 description 1
- 150000003254 radicals Chemical class 0.000 description 1
- 238000010107 reaction injection moulding Methods 0.000 description 1
- 239000012429 reaction media Substances 0.000 description 1
- 230000009467 reduction Effects 0.000 description 1
- 230000001105 regulatory effect Effects 0.000 description 1
- 239000013557 residual solvent Substances 0.000 description 1
- 238000005070 sampling Methods 0.000 description 1
- 238000000926 separation method Methods 0.000 description 1
- 238000011172 small scale experimental method Methods 0.000 description 1
- 239000007790 solid phase Substances 0.000 description 1
- 239000011877 solvent mixture Substances 0.000 description 1
- 125000006850 spacer group Chemical group 0.000 description 1
- 241000894007 species Species 0.000 description 1
- 238000004611 spectroscopical analysis Methods 0.000 description 1
- 239000007921 spray Substances 0.000 description 1
- 239000003381 stabilizer Substances 0.000 description 1
- 230000000087 stabilizing effect Effects 0.000 description 1
- 239000007858 starting material Substances 0.000 description 1
- 238000012916 structural analysis Methods 0.000 description 1
- 238000000859 sublimation Methods 0.000 description 1
- 230000008022 sublimation Effects 0.000 description 1
- 229920001059 synthetic polymer Polymers 0.000 description 1
- 230000009897 systematic effect Effects 0.000 description 1
- 229920001897 terpolymer Polymers 0.000 description 1
- ISXSCDLOGDJUNJ-UHFFFAOYSA-N tert-butyl prop-2-enoate Chemical compound CC(C)(C)OC(=O)C=C ISXSCDLOGDJUNJ-UHFFFAOYSA-N 0.000 description 1
- YLQBMQCUIZJEEH-UHFFFAOYSA-N tetrahydrofuran Natural products C=1C=COC=1 YLQBMQCUIZJEEH-UHFFFAOYSA-N 0.000 description 1
- 238000002076 thermal analysis method Methods 0.000 description 1
- 230000000930 thermomechanical effect Effects 0.000 description 1
- 238000012546 transfer Methods 0.000 description 1
- 239000011364 vaporized material Substances 0.000 description 1
- 238000011179 visual inspection Methods 0.000 description 1
- 238000005406 washing Methods 0.000 description 1
Images
Classifications
-
- D—TEXTILES; PAPER
- D01—NATURAL OR MAN-MADE THREADS OR FIBRES; SPINNING
- D01F—CHEMICAL FEATURES IN THE MANUFACTURE OF ARTIFICIAL FILAMENTS, THREADS, FIBRES, BRISTLES OR RIBBONS; APPARATUS SPECIALLY ADAPTED FOR THE MANUFACTURE OF CARBON FILAMENTS
- D01F6/00—Monocomponent artificial filaments or the like of synthetic polymers; Manufacture thereof
- D01F6/58—Monocomponent artificial filaments or the like of synthetic polymers; Manufacture thereof from homopolycondensation products
- D01F6/72—Monocomponent artificial filaments or the like of synthetic polymers; Manufacture thereof from homopolycondensation products from polyureas
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08G—MACROMOLECULAR COMPOUNDS OBTAINED OTHERWISE THAN BY REACTIONS ONLY INVOLVING UNSATURATED CARBON-TO-CARBON BONDS
- C08G18/00—Polymeric products of isocyanates or isothiocyanates
- C08G18/06—Polymeric products of isocyanates or isothiocyanates with compounds having active hydrogen
- C08G18/28—Polymeric products of isocyanates or isothiocyanates with compounds having active hydrogen characterised by the compounds used containing active hydrogen
- C08G18/30—Low-molecular-weight compounds
- C08G18/32—Polyhydroxy compounds; Polyamines; Hydroxyamines
- C08G18/3225—Polyamines
- C08G18/3237—Polyamines aromatic
- C08G18/324—Polyamines aromatic containing only one aromatic ring
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08G—MACROMOLECULAR COMPOUNDS OBTAINED OTHERWISE THAN BY REACTIONS ONLY INVOLVING UNSATURATED CARBON-TO-CARBON BONDS
- C08G18/00—Polymeric products of isocyanates or isothiocyanates
- C08G18/06—Polymeric products of isocyanates or isothiocyanates with compounds having active hydrogen
- C08G18/70—Polymeric products of isocyanates or isothiocyanates with compounds having active hydrogen characterised by the isocyanates or isothiocyanates used
- C08G18/72—Polyisocyanates or polyisothiocyanates
- C08G18/74—Polyisocyanates or polyisothiocyanates cyclic
- C08G18/76—Polyisocyanates or polyisothiocyanates cyclic aromatic
- C08G18/7614—Polyisocyanates or polyisothiocyanates cyclic aromatic containing only one aromatic ring
-
- D—TEXTILES; PAPER
- D01—NATURAL OR MAN-MADE THREADS OR FIBRES; SPINNING
- D01D—MECHANICAL METHODS OR APPARATUS IN THE MANUFACTURE OF ARTIFICIAL FILAMENTS, THREADS, FIBRES, BRISTLES OR RIBBONS
- D01D5/00—Formation of filaments, threads, or the like
- D01D5/40—Formation of filaments, threads, or the like by applying a shearing force to a dispersion or solution of filament formable polymers, e.g. by stirring
Landscapes
- Chemical & Material Sciences (AREA)
- Engineering & Computer Science (AREA)
- Chemical Kinetics & Catalysis (AREA)
- Polymers & Plastics (AREA)
- Textile Engineering (AREA)
- Health & Medical Sciences (AREA)
- Medicinal Chemistry (AREA)
- Organic Chemistry (AREA)
- Dispersion Chemistry (AREA)
- Mechanical Engineering (AREA)
- General Chemical & Material Sciences (AREA)
- Polyurethanes Or Polyureas (AREA)
- Artificial Filaments (AREA)
Applications Claiming Priority (5)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| US22035409P | 2009-06-25 | 2009-06-25 | |
| US61/220,354 | 2009-06-25 | ||
| US22229209P | 2009-07-01 | 2009-07-01 | |
| US61/222,292 | 2009-07-01 | ||
| PCT/US2010/039790 WO2010151645A1 (en) | 2009-06-25 | 2010-06-24 | Novel polyurea fiber |
Publications (2)
| Publication Number | Publication Date |
|---|---|
| JP2012531534A true JP2012531534A (ja) | 2012-12-10 |
| JP2012531534A5 JP2012531534A5 (enExample) | 2013-07-25 |
Family
ID=42543122
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| JP2012517720A Pending JP2012531534A (ja) | 2009-06-25 | 2010-06-24 | 新規なポリ尿素系繊維 |
Country Status (4)
| Country | Link |
|---|---|
| US (1) | US20110082274A1 (enExample) |
| EP (1) | EP2446074A1 (enExample) |
| JP (1) | JP2012531534A (enExample) |
| WO (1) | WO2010151645A1 (enExample) |
Cited By (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JP2019182705A (ja) * | 2018-04-10 | 2019-10-24 | 国立大学法人茨城大学 | 高純度イソシアン酸の生成方法、生成装置、内蔵キット、ガス発生装置及び分析方法 |
Families Citing this family (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN107428896A (zh) * | 2015-02-03 | 2017-12-01 | 伊利诺伊大学董事会 | 用于聚合尿素生产的环状动态聚脲 |
Citations (5)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US2292443A (en) * | 1940-07-17 | 1942-08-11 | Du Pont | Process for preparing polymeric polyamides |
| US2813775A (en) * | 1955-02-21 | 1957-11-19 | Du Pont | Process for forming films and filaments directly from polymer intermediates |
| US2888438A (en) * | 1957-03-08 | 1959-05-26 | Du Pont | Polyureas of aromatic diamines and aromatic diisocyanates |
| JPS4985193A (enExample) * | 1972-08-09 | 1974-08-15 | Rhone Poulenc Textile | |
| JPH08319420A (ja) * | 1995-01-17 | 1996-12-03 | Teijin Ltd | ポリアミド溶液組成物及びそれを用いるフィブリッド、紙状シートの製造法 |
Family Cites Families (27)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US2511544A (en) * | 1937-11-12 | 1950-06-13 | Rinke Heinrich | Diol-dilsocyanate high molecular polymerization products |
| US2284896A (en) * | 1939-05-24 | 1942-06-02 | Du Pont | Process for making polymeric products and for modifying polymeric products |
| US2284869A (en) * | 1940-02-27 | 1942-06-02 | Frank J Hinderliter | Blowout preventer |
| DE906213C (de) | 1951-12-30 | 1954-03-11 | Hoechst Ag | Verfahren zur Herstellung ueberfaerbeechter Faerbungen auf Acetylcellulose sowie linearen Polyamiden oder Polyurethanen |
| GB876491A (en) * | 1957-07-25 | 1961-09-06 | Du Pont | Improvements in or relating to filamentary material of high tenacity and initial modulus |
| MX3465E (es) | 1960-10-14 | 1980-09-12 | Wander Ltd Wander Ag Wander S | Procedimiento para la obtencion de ureas n,n'-difenil |
| CH525898A (de) | 1961-09-11 | 1972-07-31 | Wander Ag Dr A | Verfahren zur Herstellung mehrbasischer Verbindungen |
| CH479557A (de) | 1961-09-11 | 1969-10-15 | Wander Ag Dr A | Verfahren zur Herstellung neuer mehrbasischer Verbindungen |
| NL290764A (enExample) * | 1962-03-29 | |||
| DE1213553B (de) | 1962-09-20 | 1966-03-31 | Bayer Ag | Verfahren zur Herstellung von wasserunloeslichen Azofarbstoffen |
| US3418275A (en) * | 1962-11-08 | 1968-12-24 | Du Pont | Aromatic polyamides from n,n'-diphenyl diamines |
| DE1544864A1 (de) * | 1963-07-24 | 1970-05-21 | Bayer Ag | Verfahren zur Herstellung von Polyurethan-Elastomeren |
| US3386942A (en) * | 1964-06-29 | 1968-06-04 | Eastman Kodak Co | Stabilization of non-yellowing segmented polyurethane copolymers of polymeric diols, functionally aliphatic diisocyanates and functionally aliphatic diamines |
| US3671542A (en) * | 1966-06-13 | 1972-06-20 | Du Pont | Optically anisotropic aromatic polyamide dopes |
| JPS572810B2 (enExample) | 1974-02-06 | 1982-01-19 | ||
| US4035344A (en) | 1974-11-29 | 1977-07-12 | The Firestone Tire & Rubber Company | Process for preparing ordered aromatic copolyamide urea polymer |
| JPS53149881A (en) | 1977-06-03 | 1978-12-27 | Asahi Glass Co Ltd | Strengthened cation exchange resin membrane and production thereof |
| JPS5742717A (en) | 1980-08-28 | 1982-03-10 | Mitsui Toatsu Chem Inc | Polycarbonate-urea and its production |
| US4433067A (en) | 1982-04-23 | 1984-02-21 | Texaco Inc. | Reaction injection molded elastomers prepared from amine terminated polyethers, amine terminated chain extender and aromatic polyisocyanate |
| US5414118A (en) | 1991-04-12 | 1995-05-09 | Asahi Kasei Kogyo Kabushiki Kaisha | Diaminourea compound and process for production thereof and high heat resistant polyurethaneurea and process for production thereof |
| US5635585A (en) | 1992-10-21 | 1997-06-03 | E. I. Du Pont De Nemours And Company | Polymerization of, and depolymerization to, cyclic ethers using selected metal compound catalysts |
| US5401825A (en) | 1993-05-05 | 1995-03-28 | Gould Inc. | Styrene type monomers containing substituents thereon, e.g. urea, and polymers and copolymers thereof |
| US5723568A (en) * | 1995-01-17 | 1998-03-03 | Teijin Limited | Polyamide solution composition and methods for producing fibrids and paper-like sheets using the same |
| JPH0913068A (ja) | 1995-06-30 | 1997-01-14 | Tonen Corp | グリース組成物 |
| JPH1044618A (ja) | 1996-08-06 | 1998-02-17 | Kemipuro Kasei Kk | ビス(p−トルエンスルホニルアミノカルボニルアミノ)−ジフェニル誘導体よりなる新規顕色剤 |
| US7015299B2 (en) * | 2001-04-30 | 2006-03-21 | Wilkinson W Kenneth | Melt spun thermoplastic polyurethanes useful as textile fibers |
| JP2004149626A (ja) | 2002-10-29 | 2004-05-27 | Ajinomoto Co Inc | 木質熱可塑性樹脂組成物 |
-
2010
- 2010-06-24 US US12/822,567 patent/US20110082274A1/en not_active Abandoned
- 2010-06-24 EP EP10727635A patent/EP2446074A1/en not_active Withdrawn
- 2010-06-24 JP JP2012517720A patent/JP2012531534A/ja active Pending
- 2010-06-24 WO PCT/US2010/039790 patent/WO2010151645A1/en not_active Ceased
Patent Citations (5)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US2292443A (en) * | 1940-07-17 | 1942-08-11 | Du Pont | Process for preparing polymeric polyamides |
| US2813775A (en) * | 1955-02-21 | 1957-11-19 | Du Pont | Process for forming films and filaments directly from polymer intermediates |
| US2888438A (en) * | 1957-03-08 | 1959-05-26 | Du Pont | Polyureas of aromatic diamines and aromatic diisocyanates |
| JPS4985193A (enExample) * | 1972-08-09 | 1974-08-15 | Rhone Poulenc Textile | |
| JPH08319420A (ja) * | 1995-01-17 | 1996-12-03 | Teijin Ltd | ポリアミド溶液組成物及びそれを用いるフィブリッド、紙状シートの製造法 |
Cited By (2)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JP2019182705A (ja) * | 2018-04-10 | 2019-10-24 | 国立大学法人茨城大学 | 高純度イソシアン酸の生成方法、生成装置、内蔵キット、ガス発生装置及び分析方法 |
| JP7150257B2 (ja) | 2018-04-10 | 2022-10-11 | 国立大学法人茨城大学 | イソシアン酸の生成方法、生成装置、内蔵キット及びガス発生装置 |
Also Published As
| Publication number | Publication date |
|---|---|
| WO2010151645A1 (en) | 2010-12-29 |
| US20110082274A1 (en) | 2011-04-07 |
| EP2446074A1 (en) | 2012-05-02 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| Takayanagi et al. | N‐substituted poly (p‐phenylene terephthalamide) | |
| Du et al. | Synthesis of linear polyurethane bearing pendant furan and cross-linked healable polyurethane containing Diels–Alder bonds | |
| Li et al. | Feasible self-healing CL-20 based PBX: employing a novel polyurethane-urea containing disulfide bonds as polymer binder | |
| Riaz et al. | Microwave-assisted green synthesis of some nanoconjugated copolymers: characterisation and fluorescence quenching studies with bovine serum albumin | |
| Bazzar et al. | Novel fluorescent light-emitting polymer composites bearing 1, 2, 4-triazole and quinoxaline moieties: Reinforcement and thermal stabilization with silicon carbide nanoparticles by epoxide functionalization | |
| Bose et al. | Amphiphilic polypeptide-polyoxazoline graft copolymer conjugate with tunable thermoresponsiveness: Synthesis and self-assembly into various micellar structures in aqueous and nonaqueous media | |
| EP3789438A1 (en) | Polyamide resin film and resin laminate using same | |
| US3573260A (en) | Process of preparing solid molding powder from polytrimellitate amideimides | |
| KR20200096107A (ko) | 폴리아미드 수지 필름 및 이를 이용한 수지 적층체 | |
| Ghaemy et al. | Nanostructured composites of poly (triazole-amide-imide) s and reactive titanium oxide by epoxide functionalization: thermal, mechanical, photophysical and metal ions adsorption properties | |
| Balamurugan et al. | Click chemistry-assisted, bis-cholesteryl-appended, isosorbide-based, dual-responsive organogelators and their self-assemblies | |
| Yan et al. | Synthesis and characterization of semiaromatic polyamides with dicyclohexane units | |
| Chen et al. | The poor solubility of ureidopyrimidone can be used to form gels of low molecular weight N-alkyl urea oligomers in organic solvents | |
| Kricheldorf et al. | New polymer syntheses. 68. Kevlar-type polyaramides derived from 2-phenoxy-1, 4-diaminobenzene | |
| JP2012531534A (ja) | 新規なポリ尿素系繊維 | |
| US8129498B2 (en) | Polymeric medium | |
| Gómez-Valdemoro et al. | Novel aromatic polyamides with main chain and pendant 1, 2, 4-triazole moieties and their application to the extraction/elimination of mercury cations from aqueous media | |
| Tang et al. | Novel azide-rich fluorinated energetic polyurethane with excellent mechanical properties by a one-pot facile strategy | |
| Anas et al. | Side-chain functionality driven thermoresponsive and semicrystalline poly (L-glutamate) s and self-assembly | |
| Locatelli et al. | Synthesis of polyurea–polyether nanoparticles via spontaneous nanoprecipitation | |
| Jeyakumar et al. | Polyamide‐6, 6‐based blocky copolyamides obtained by solid‐state modification | |
| Tundidor-Camba et al. | Synthesis, characterization and film preparation of new co-polyimide based on new 3, 5-diamino-N-(pyridin-4-ylmethyl) benzamide, ODA and 6FDA | |
| Yu et al. | Synthesis and characterization of poly (2-cyano-1, 4-phenylene terephthalamide) and its copolymers by phosphorylation-assisted polycondensation reaction | |
| Mehdipour‐Ataei et al. | Preparation and characterization of thermally stable poly (amide‐urea) s functionalized with anthraquinone chromophore | |
| Nakano et al. | A new process for producing polyamide from polyester |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| A521 | Request for written amendment filed |
Free format text: JAPANESE INTERMEDIATE CODE: A523 Effective date: 20130610 |
|
| A621 | Written request for application examination |
Free format text: JAPANESE INTERMEDIATE CODE: A621 Effective date: 20130610 |
|
| RD04 | Notification of resignation of power of attorney |
Free format text: JAPANESE INTERMEDIATE CODE: A7424 Effective date: 20131219 |
|
| A977 | Report on retrieval |
Free format text: JAPANESE INTERMEDIATE CODE: A971007 Effective date: 20140825 |
|
| A131 | Notification of reasons for refusal |
Free format text: JAPANESE INTERMEDIATE CODE: A131 Effective date: 20140902 |
|
| A02 | Decision of refusal |
Free format text: JAPANESE INTERMEDIATE CODE: A02 Effective date: 20150217 |