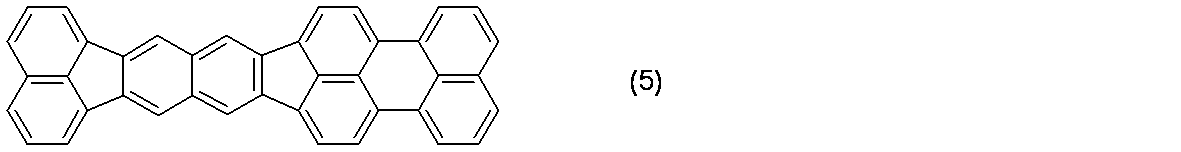JP2012144459A - 有機化合物、有機発光素子及び画像表示装置 - Google Patents
有機化合物、有機発光素子及び画像表示装置 Download PDFInfo
- Publication number
- JP2012144459A JP2012144459A JP2011002263A JP2011002263A JP2012144459A JP 2012144459 A JP2012144459 A JP 2012144459A JP 2011002263 A JP2011002263 A JP 2011002263A JP 2011002263 A JP2011002263 A JP 2011002263A JP 2012144459 A JP2012144459 A JP 2012144459A
- Authority
- JP
- Japan
- Prior art keywords
- group
- light emitting
- organic compound
- substituted
- organic
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Granted
Links
- KYLGOZJJUQHXKL-UHFFFAOYSA-N c(cc1ccc2ccc3-c4cc(cc(-c5ccc-6c7c5c-5ccc7-c7cccc8c7c-6ccc8)c-5c5)c5cc4-4)cc5c1c2c3c-4c5 Chemical compound c(cc1ccc2ccc3-c4cc(cc(-c5ccc-6c7c5c-5ccc7-c7cccc8c7c-6ccc8)c-5c5)c5cc4-4)cc5c1c2c3c-4c5 KYLGOZJJUQHXKL-UHFFFAOYSA-N 0.000 description 1
- JPRBNGOMCNZXSJ-UHFFFAOYSA-N c(ccc1c2)cc1cc(-c(cc1)c34)c2-c3ccc-2c4c1-c(cc1)c3c-2ccc-2c3c1-c1c-2cc(cccc2)c2c1 Chemical compound c(ccc1c2)cc1cc(-c(cc1)c34)c2-c3ccc-2c4c1-c(cc1)c3c-2ccc-2c3c1-c1c-2cc(cccc2)c2c1 JPRBNGOMCNZXSJ-UHFFFAOYSA-N 0.000 description 1
Images
Classifications
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07C—ACYCLIC OR CARBOCYCLIC COMPOUNDS
- C07C13/00—Cyclic hydrocarbons containing rings other than, or in addition to, six-membered aromatic rings
- C07C13/28—Polycyclic hydrocarbons or acyclic hydrocarbon derivatives thereof
- C07C13/32—Polycyclic hydrocarbons or acyclic hydrocarbon derivatives thereof with condensed rings
- C07C13/62—Polycyclic hydrocarbons or acyclic hydrocarbon derivatives thereof with condensed rings with more than three condensed rings
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07C—ACYCLIC OR CARBOCYCLIC COMPOUNDS
- C07C211/00—Compounds containing amino groups bound to a carbon skeleton
- C07C211/43—Compounds containing amino groups bound to a carbon skeleton having amino groups bound to carbon atoms of six-membered aromatic rings of the carbon skeleton
- C07C211/57—Compounds containing amino groups bound to a carbon skeleton having amino groups bound to carbon atoms of six-membered aromatic rings of the carbon skeleton having amino groups bound to carbon atoms of six-membered aromatic rings being part of condensed ring systems of the carbon skeleton
- C07C211/61—Compounds containing amino groups bound to a carbon skeleton having amino groups bound to carbon atoms of six-membered aromatic rings of the carbon skeleton having amino groups bound to carbon atoms of six-membered aromatic rings being part of condensed ring systems of the carbon skeleton with at least one of the condensed ring systems formed by three or more rings
-
- C—CHEMISTRY; METALLURGY
- C09—DYES; PAINTS; POLISHES; NATURAL RESINS; ADHESIVES; COMPOSITIONS NOT OTHERWISE PROVIDED FOR; APPLICATIONS OF MATERIALS NOT OTHERWISE PROVIDED FOR
- C09K—MATERIALS FOR MISCELLANEOUS APPLICATIONS, NOT PROVIDED FOR ELSEWHERE
- C09K11/00—Luminescent materials, e.g. electroluminescent or chemiluminescent
- C09K11/06—Luminescent materials, e.g. electroluminescent or chemiluminescent containing organic luminescent materials
-
- H—ELECTRICITY
- H05—ELECTRIC TECHNIQUES NOT OTHERWISE PROVIDED FOR
- H05B—ELECTRIC HEATING; ELECTRIC LIGHT SOURCES NOT OTHERWISE PROVIDED FOR; CIRCUIT ARRANGEMENTS FOR ELECTRIC LIGHT SOURCES, IN GENERAL
- H05B33/00—Electroluminescent light sources
- H05B33/12—Light sources with substantially two-dimensional [2D] radiating surfaces
- H05B33/14—Light sources with substantially two-dimensional [2D] radiating surfaces characterised by the chemical or physical composition or the arrangement of the electroluminescent material, or by the simultaneous addition of the electroluminescent material in or onto the light source
-
- H—ELECTRICITY
- H10—SEMICONDUCTOR DEVICES; ELECTRIC SOLID-STATE DEVICES NOT OTHERWISE PROVIDED FOR
- H10K—ORGANIC ELECTRIC SOLID-STATE DEVICES
- H10K50/00—Organic light-emitting devices
- H10K50/10—OLEDs or polymer light-emitting diodes [PLED]
- H10K50/11—OLEDs or polymer light-emitting diodes [PLED] characterised by the electroluminescent [EL] layers
-
- H—ELECTRICITY
- H10—SEMICONDUCTOR DEVICES; ELECTRIC SOLID-STATE DEVICES NOT OTHERWISE PROVIDED FOR
- H10K—ORGANIC ELECTRIC SOLID-STATE DEVICES
- H10K59/00—Integrated devices, or assemblies of multiple devices, comprising at least one organic light-emitting element covered by group H10K50/00
- H10K59/10—OLED displays
- H10K59/12—Active-matrix OLED [AMOLED] displays
- H10K59/121—Active-matrix OLED [AMOLED] displays characterised by the geometry or disposition of pixel elements
- H10K59/1213—Active-matrix OLED [AMOLED] displays characterised by the geometry or disposition of pixel elements the pixel elements being TFTs
-
- H—ELECTRICITY
- H10—SEMICONDUCTOR DEVICES; ELECTRIC SOLID-STATE DEVICES NOT OTHERWISE PROVIDED FOR
- H10K—ORGANIC ELECTRIC SOLID-STATE DEVICES
- H10K85/00—Organic materials used in the body or electrodes of devices covered by this subclass
- H10K85/60—Organic compounds having low molecular weight
- H10K85/615—Polycyclic condensed aromatic hydrocarbons, e.g. anthracene
- H10K85/624—Polycyclic condensed aromatic hydrocarbons, e.g. anthracene containing six or more rings
-
- H—ELECTRICITY
- H10—SEMICONDUCTOR DEVICES; ELECTRIC SOLID-STATE DEVICES NOT OTHERWISE PROVIDED FOR
- H10K—ORGANIC ELECTRIC SOLID-STATE DEVICES
- H10K85/00—Organic materials used in the body or electrodes of devices covered by this subclass
- H10K85/60—Organic compounds having low molecular weight
- H10K85/615—Polycyclic condensed aromatic hydrocarbons, e.g. anthracene
- H10K85/626—Polycyclic condensed aromatic hydrocarbons, e.g. anthracene containing more than one polycyclic condensed aromatic rings, e.g. bis-anthracene
-
- H—ELECTRICITY
- H10—SEMICONDUCTOR DEVICES; ELECTRIC SOLID-STATE DEVICES NOT OTHERWISE PROVIDED FOR
- H10K—ORGANIC ELECTRIC SOLID-STATE DEVICES
- H10K85/00—Organic materials used in the body or electrodes of devices covered by this subclass
- H10K85/60—Organic compounds having low molecular weight
- H10K85/631—Amine compounds having at least two aryl rest on at least one amine-nitrogen atom, e.g. triphenylamine
- H10K85/633—Amine compounds having at least two aryl rest on at least one amine-nitrogen atom, e.g. triphenylamine comprising polycyclic condensed aromatic hydrocarbons as substituents on the nitrogen atom
-
- H—ELECTRICITY
- H10—SEMICONDUCTOR DEVICES; ELECTRIC SOLID-STATE DEVICES NOT OTHERWISE PROVIDED FOR
- H10K—ORGANIC ELECTRIC SOLID-STATE DEVICES
- H10K85/00—Organic materials used in the body or electrodes of devices covered by this subclass
- H10K85/60—Organic compounds having low molecular weight
- H10K85/649—Aromatic compounds comprising a hetero atom
- H10K85/654—Aromatic compounds comprising a hetero atom comprising only nitrogen as heteroatom
-
- H—ELECTRICITY
- H10—SEMICONDUCTOR DEVICES; ELECTRIC SOLID-STATE DEVICES NOT OTHERWISE PROVIDED FOR
- H10K—ORGANIC ELECTRIC SOLID-STATE DEVICES
- H10K85/00—Organic materials used in the body or electrodes of devices covered by this subclass
- H10K85/60—Organic compounds having low molecular weight
- H10K85/649—Aromatic compounds comprising a hetero atom
- H10K85/657—Polycyclic condensed heteroaromatic hydrocarbons
- H10K85/6572—Polycyclic condensed heteroaromatic hydrocarbons comprising only nitrogen in the heteroaromatic polycondensed ring system, e.g. phenanthroline or carbazole
-
- H—ELECTRICITY
- H10—SEMICONDUCTOR DEVICES; ELECTRIC SOLID-STATE DEVICES NOT OTHERWISE PROVIDED FOR
- H10K—ORGANIC ELECTRIC SOLID-STATE DEVICES
- H10K85/00—Organic materials used in the body or electrodes of devices covered by this subclass
- H10K85/60—Organic compounds having low molecular weight
- H10K85/649—Aromatic compounds comprising a hetero atom
- H10K85/657—Polycyclic condensed heteroaromatic hydrocarbons
- H10K85/6576—Polycyclic condensed heteroaromatic hydrocarbons comprising only sulfur in the heteroaromatic polycondensed ring system, e.g. benzothiophene
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07C—ACYCLIC OR CARBOCYCLIC COMPOUNDS
- C07C2603/00—Systems containing at least three condensed rings
- C07C2603/02—Ortho- or ortho- and peri-condensed systems
- C07C2603/54—Ortho- or ortho- and peri-condensed systems containing more than five condensed rings
-
- C—CHEMISTRY; METALLURGY
- C09—DYES; PAINTS; POLISHES; NATURAL RESINS; ADHESIVES; COMPOSITIONS NOT OTHERWISE PROVIDED FOR; APPLICATIONS OF MATERIALS NOT OTHERWISE PROVIDED FOR
- C09K—MATERIALS FOR MISCELLANEOUS APPLICATIONS, NOT PROVIDED FOR ELSEWHERE
- C09K2211/00—Chemical nature of organic luminescent or tenebrescent compounds
- C09K2211/10—Non-macromolecular compounds
- C09K2211/1003—Carbocyclic compounds
- C09K2211/1011—Condensed systems
-
- H—ELECTRICITY
- H10—SEMICONDUCTOR DEVICES; ELECTRIC SOLID-STATE DEVICES NOT OTHERWISE PROVIDED FOR
- H10K—ORGANIC ELECTRIC SOLID-STATE DEVICES
- H10K2101/00—Properties of the organic materials covered by group H10K85/00
- H10K2101/90—Multiple hosts in the emissive layer
Landscapes
- Chemical & Material Sciences (AREA)
- Organic Chemistry (AREA)
- Engineering & Computer Science (AREA)
- Materials Engineering (AREA)
- Physics & Mathematics (AREA)
- Spectroscopy & Molecular Physics (AREA)
- Geometry (AREA)
- Microelectronics & Electronic Packaging (AREA)
- Optics & Photonics (AREA)
- Electroluminescent Light Sources (AREA)
- Organic Low-Molecular-Weight Compounds And Preparation Thereof (AREA)
Abstract
Description
以下に示す試薬、溶媒を反応容器内に投入した。
化合物X1:4.0g(13mmol)
化合物X2:2.8g(13mmol)
エタノール:65ml
以下に示す試薬、溶媒を反応容器内に投入した。
化合物X3:5.0g(10mmol)
化合物X4:6.1g(21mmol)
キシレン:100ml
以下に示す試薬、溶媒を反応容器内に投入した。
化合物X5:1.0g(1.5mmol)
化合物X6:0.55g(2.2mmol)
DMF:20ml
ビス(ジベンジリデンアセトン)パラジウム(0):0.84g(1.5mmol)
1,8―ジアザビシクロ[5.4.0]ウンデセ−7−エン:0.44g(2.9mmol)
トリシクロヘキシルフォスフィン:0.81g(2.9mmol)
以下に示す試薬、溶媒を反応容器内に投入した。
化合物X7:10g(55mmol)
化合物X2:12g(55mmol)
エタノール:200ml
以下に示す試薬、溶媒を反応容器内に投入した。
化合物X8:10g(28mmol)
化合物X4:17g(56mmol)
キシレン:100ml
以下に示す試薬、溶媒を反応容器内に投入した。
化合物X9:2.0g(3.6mmol)
化合物X10:2.0g(5.3mmol)
DMF:40ml
ビス(ジベンジリデンアセトン)パラジウム(0):2.0g(3.6mmol)
1,8―ジアザビシクロ[5.4.0]ウンデセ−7−エン:1.1g(7.1mmol)
トリシクロヘキシルフォスフィン:2.0g(7.1mmol)
DART−TOF−MASS:M+H=563.2
実施例2(1)において、化合物X2に代えて下記に示される化合物X11を使用する以外は、実施例2と同様の方法で合成を行うことにより、例示化合物XX−5を得た。
DART−TOF−MASS:M+H=805.3
実施例2(1)において、化合物X7に代えて下記に示される化合物X12を使用する以外は、実施例2と同様の方法で合成を行うことにより、例示化合物XX−10を得た。
DART−TOF−MASS:M+H=805.3
実施例1(3)において、化合物X6の代わりに下記に示されるX13を使用する以外は、実施例1と同様の方法で合成を行うことにより、例示化合物XY−1を得た。
DART−TOF−MASS:M+H=765.3
実施例2(1)において、化合物X2の代わりに下記に示されるX14を使用する以外は、実施例2と同様の方法で合成を行うことにより、例示化合物XY−3を得た。
DART−TOF−MASS:M+H=877.5
本実施例では、基板上に、陽極、ホール注入層、ホール輸送層、発光層、ホール・エキシトンブロッキング層、電子輸送層、陰極が順次形成された有機発光素子を作製した。以下に、本実施例で使用した材料の一部を示す。
実施例7において、G−2、G−3及びゲストを、表5に示される化合物に適宜変更する以外は、実施例7と同様の方法により有機発光素子を作製した。得られた素子について実施例7と同様に素子の特性を測定・評価した。測定の結果を表5に示す。尚、表5において、G−2として使用したH2、H7、H10、H15、H17、H19、H21及びH23、並びにG−3として使用したH2、H7、H10、H17、H19、H21、H23及びH24は、それぞれ表3に示されるホストである。
本実施例では、基板上に、陽極、ホール注入層、ホール輸送層、発光層、電子輸送層、電子注入層、陰極が順次形成された有機発光素子を作製した。尚、本実施例で作製される有機発光素子は共振構造を有している。以下に、本実施例で使用した材料の一部を示す。
以上により、有機発光素子を作製した。
実施例18において、G−13、G−14及びゲストを、表7に示される化合物に適宜変更する以外は、実施例18と同様の方法により有機発光素子を作製した。得られた素子について実施例18と同様に素子の特性を測定・評価した。測定の結果を表7に示す。尚、表7において、G−13として使用したH6及びH16、並びにG−14として使用したH21及びH24は、それぞれ表3に示されるホストである。
本実施例では、基板上に、陽極、ホール輸送層、第1発光層、第2発光層、ホール・エキシトンブロッキング層、電子輸送層、陰極が順次形成された有機発光素子を作製した。尚、本実施例の有機発光素子は発光層が複数あるので、各発光層に含まれるゲストが個別あるいは同時に発光する態様である。以下に、本実施例で使用した材料の一部を示す。
実施例21において、G−22、G−23、G−24及びゲストを、表9に示される化合物に適宜変更する以外は、実施例22と同様の方法により有機発光素子を作製した。得られた素子について実施例22と同様に素子の特性を測定・評価した。測定の結果を表9に示す。尚、表9において、G−22として使用したH18及びH23、G−23として使用したH18及びH23、並びにG−24として使用したH4及びH10は、それぞれ表3に示されるホストである。
Claims (9)
- 前記R1乃至R20が、それぞれ水素原子及び置換あるいは無置換のアリール基から選ばれる置換基であることを特徴とする、請求項1に記載の有機化合物。
- R2及びR9が、置換あるいは無置換のアリール基であり、R1、R3乃至R8及びR10乃至R20が水素原子であることを特徴とする、請求項1又は2に記載の有機化合物。
- 分子構造中にターシャリーブチル基を2つ以上有することを特徴とする、請求項1乃至3のいずれか一項に記載の有機化合物。
- 陽極と陰極と、
前記陽極と前記陰極との間に配置される有機化合物層と、を有する有機発光素子であって、
前記有機化合物層の少なくとも一層に、請求項1乃至4のいずれか一項に記載の有機化合物が含まれることを特徴とする、有機発光素子。 - 前記有機化合物が、発光層に含まれることを特徴とする、請求項5に記載の有機発光素子。
- 黄色発光することを特徴とする、請求項6に記載の有機発光素子。
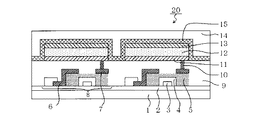
- 複数の画素を有する表示装置であって、
前記複数の画素が、請求項5乃至7のいずれか一項に記載の有機発光素子と、前記有機発光素子に電気接続されるTFT素子と、をそれぞれ有することを特徴とする、表示装置。 - 画像情報を入力するための入力部と、画像を出力するための表示部と、を有する画像入力装置であって、
前記表示部が複数の画素を有し、
前記複数の画素が、請求項5乃至7のいずれか一項に記載の有機発光素子と、前記有機発光素子に電気接続されるTFT素子と、をそれぞれ有することを特徴とする、画像入力装置。
Priority Applications (6)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| JP2011002263A JP5713686B2 (ja) | 2011-01-07 | 2011-01-07 | 有機化合物、有機発光素子及び画像表示装置 |
| KR1020137020177A KR101555110B1 (ko) | 2011-01-07 | 2011-12-09 | 유기 화합물, 유기 발광 디바이스, 및 화상 표시 디바이스 |
| EP11855218.1A EP2661418A4 (en) | 2011-01-07 | 2011-12-09 | ORGANIC CONNECTION, ORGANIC LIGHT-EMITTING DEVICE AND IMAGE DISPLAY DEVICE |
| CN201180063852XA CN103313957A (zh) | 2011-01-07 | 2011-12-09 | 有机化合物、有机发光器件和图像显示装置 |
| US13/978,058 US9397298B2 (en) | 2011-01-07 | 2011-12-09 | Organic compound, organic light-emitting device, and image display device |
| PCT/JP2011/079261 WO2012093578A1 (en) | 2011-01-07 | 2011-12-09 | Organic compound, organic light-emitting device, and image display device |
Applications Claiming Priority (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| JP2011002263A JP5713686B2 (ja) | 2011-01-07 | 2011-01-07 | 有機化合物、有機発光素子及び画像表示装置 |
Publications (3)
| Publication Number | Publication Date |
|---|---|
| JP2012144459A true JP2012144459A (ja) | 2012-08-02 |
| JP2012144459A5 JP2012144459A5 (ja) | 2014-02-20 |
| JP5713686B2 JP5713686B2 (ja) | 2015-05-07 |
Family
ID=46457438
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| JP2011002263A Active JP5713686B2 (ja) | 2011-01-07 | 2011-01-07 | 有機化合物、有機発光素子及び画像表示装置 |
Country Status (6)
| Country | Link |
|---|---|
| US (1) | US9397298B2 (ja) |
| EP (1) | EP2661418A4 (ja) |
| JP (1) | JP5713686B2 (ja) |
| KR (1) | KR101555110B1 (ja) |
| CN (1) | CN103313957A (ja) |
| WO (1) | WO2012093578A1 (ja) |
Cited By (3)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2013084833A1 (en) * | 2011-12-06 | 2013-06-13 | Canon Kabushiki Kaisha | Novel organic compound, organic light emitting device, and image display device |
| JP2015530735A (ja) * | 2012-07-23 | 2015-10-15 | メルク パテント ゲーエムベーハー | 2−ジアリールアミノフルオレン誘導体およびそれらを含む有機電子素子 |
| WO2016204150A1 (ja) * | 2015-06-16 | 2016-12-22 | 出光興産株式会社 | 化合物、有機エレクトロルミネッセンス素子用材料、有機エレクトロルミネッセンス素子及び電子機器 |
Families Citing this family (3)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JP5674707B2 (ja) * | 2012-05-22 | 2015-02-25 | 株式会社東芝 | 表示装置 |
| JPWO2013183280A1 (ja) | 2012-06-06 | 2016-01-28 | パナソニック株式会社 | インクジェット装置および有機elデバイスの製造方法 |
| US9266324B2 (en) | 2012-07-05 | 2016-02-23 | Panasonic Intellectual Property Management Co., Ltd. | Inkjet apparatus and method for manufacturing organic EL device |
Citations (6)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JP2003347057A (ja) * | 2002-04-24 | 2003-12-05 | Eastman Kodak Co | 有機発光ダイオードデバイス |
| JP2005068087A (ja) * | 2003-08-26 | 2005-03-17 | Mitsui Chemicals Inc | ベンゾフルオランテン化合物、および該ベンゾフルオランテン化合物を含有する有機電界発光素子 |
| JP2008187185A (ja) * | 2002-12-31 | 2008-08-14 | Eastman Kodak Co | 高効率電場発光デバイス |
| JP2009001499A (ja) * | 2007-06-19 | 2009-01-08 | Mitsui Chemicals Inc | 芳香族炭化水素化合物、および該芳香族炭化水素化合物を含有する有機電界発光素子 |
| JP2009302470A (ja) * | 2008-06-17 | 2009-12-24 | Mitsui Chemicals Inc | 有機トランジスタ |
| JP2011501462A (ja) * | 2007-10-26 | 2011-01-06 | グローバル・オーエルイーディー・テクノロジー・リミテッド・ライアビリティ・カンパニー | 特定のフルオランテンホストを有するリン光oledデバイス |
Family Cites Families (8)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JP3824417B2 (ja) | 1997-04-04 | 2006-09-20 | 三井化学株式会社 | 炭化水素化合物および有機電界発光素子 |
| JP3781517B2 (ja) | 1997-07-22 | 2006-05-31 | 三井化学株式会社 | 有機電界発光素子 |
| JP4255610B2 (ja) * | 1999-12-28 | 2009-04-15 | 出光興産株式会社 | 白色系有機エレクトロルミネッセンス素子 |
| US6866947B1 (en) * | 1999-12-28 | 2005-03-15 | Idemitsu Kosan Co., Ltd. | Organic electroluminescence device emitting white light |
| JP4467170B2 (ja) * | 2000-11-22 | 2010-05-26 | 三井化学株式会社 | 炭化水素化合物および有機電界発光素子 |
| TWI314947B (en) * | 2002-04-24 | 2009-09-21 | Eastman Kodak Compan | Organic light emitting diode devices with improved operational stability |
| JP2009267135A (ja) | 2008-04-25 | 2009-11-12 | Mitsui Chemicals Inc | 有機トランジスタ |
| JP5911377B2 (ja) * | 2011-08-04 | 2016-04-27 | キヤノン株式会社 | 有機化合物およびこれを有する有機発光素子 |
-
2011
- 2011-01-07 JP JP2011002263A patent/JP5713686B2/ja active Active
- 2011-12-09 US US13/978,058 patent/US9397298B2/en active Active
- 2011-12-09 CN CN201180063852XA patent/CN103313957A/zh active Pending
- 2011-12-09 WO PCT/JP2011/079261 patent/WO2012093578A1/en not_active Ceased
- 2011-12-09 EP EP11855218.1A patent/EP2661418A4/en not_active Withdrawn
- 2011-12-09 KR KR1020137020177A patent/KR101555110B1/ko not_active Expired - Fee Related
Patent Citations (6)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JP2003347057A (ja) * | 2002-04-24 | 2003-12-05 | Eastman Kodak Co | 有機発光ダイオードデバイス |
| JP2008187185A (ja) * | 2002-12-31 | 2008-08-14 | Eastman Kodak Co | 高効率電場発光デバイス |
| JP2005068087A (ja) * | 2003-08-26 | 2005-03-17 | Mitsui Chemicals Inc | ベンゾフルオランテン化合物、および該ベンゾフルオランテン化合物を含有する有機電界発光素子 |
| JP2009001499A (ja) * | 2007-06-19 | 2009-01-08 | Mitsui Chemicals Inc | 芳香族炭化水素化合物、および該芳香族炭化水素化合物を含有する有機電界発光素子 |
| JP2011501462A (ja) * | 2007-10-26 | 2011-01-06 | グローバル・オーエルイーディー・テクノロジー・リミテッド・ライアビリティ・カンパニー | 特定のフルオランテンホストを有するリン光oledデバイス |
| JP2009302470A (ja) * | 2008-06-17 | 2009-12-24 | Mitsui Chemicals Inc | 有機トランジスタ |
Cited By (5)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2013084833A1 (en) * | 2011-12-06 | 2013-06-13 | Canon Kabushiki Kaisha | Novel organic compound, organic light emitting device, and image display device |
| US9590199B2 (en) | 2011-12-06 | 2017-03-07 | Canon Kabushiki Kaisha | Organic compound, organic light emitting device, and image display device |
| JP2015530735A (ja) * | 2012-07-23 | 2015-10-15 | メルク パテント ゲーエムベーハー | 2−ジアリールアミノフルオレン誘導体およびそれらを含む有機電子素子 |
| WO2016204150A1 (ja) * | 2015-06-16 | 2016-12-22 | 出光興産株式会社 | 化合物、有機エレクトロルミネッセンス素子用材料、有機エレクトロルミネッセンス素子及び電子機器 |
| US10790449B2 (en) | 2015-06-16 | 2020-09-29 | Idemitsu Kosan Co., Ltd. | Compound, material for organic electroluminescence element, organic electroluminescence element, and electronic device |
Also Published As
| Publication number | Publication date |
|---|---|
| JP5713686B2 (ja) | 2015-05-07 |
| US20130292662A1 (en) | 2013-11-07 |
| CN103313957A (zh) | 2013-09-18 |
| WO2012093578A1 (en) | 2012-07-12 |
| EP2661418A4 (en) | 2016-02-24 |
| KR101555110B1 (ko) | 2015-09-22 |
| EP2661418A1 (en) | 2013-11-13 |
| US9397298B2 (en) | 2016-07-19 |
| KR20130112933A (ko) | 2013-10-14 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| JP5791445B2 (ja) | 新規有機化合物、それを有する有機発光素子及び表示装置 | |
| EP2243761B1 (en) | Phenanthrene derivatives, light-emitting device and image display apparatus using the same | |
| KR101555111B1 (ko) | 유기 화합물, 유기 전계발광 소자, 및 화상 표시 디바이스 | |
| JP5618647B2 (ja) | 新規有機化合物およびそれを有する有機発光素子 | |
| JP5713686B2 (ja) | 有機化合物、有機発光素子及び画像表示装置 | |
| EP2379472A1 (en) | Novel organic compound | |
| KR101490575B1 (ko) | 신규한 유기 화합물 및 이를 포함하는 유기 발광 소자 | |
| JP5627300B2 (ja) | 新規有機化合物およびそれを有する有機発光素子 | |
| JP4750893B1 (ja) | 新規有機化合物および有機発光素子 | |
| JP5578934B2 (ja) | 新規有機化合物 | |
| JP5704940B2 (ja) | 有機化合物、有機発光素子及び表示装置 | |
| JP5586981B2 (ja) | 新規有機化合物および有機発光素子 | |
| JP5645465B2 (ja) | 新規有機化合物 | |
| JP2012188355A (ja) | 新規有機化合物およびそれを有する有機発光素子 | |
| JP2012148987A (ja) | 有機化合物、有機発光素子及び表示装置 |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| A521 | Request for written amendment filed |
Free format text: JAPANESE INTERMEDIATE CODE: A523 Effective date: 20131226 |
|
| A621 | Written request for application examination |
Free format text: JAPANESE INTERMEDIATE CODE: A621 Effective date: 20131226 |
|
| A131 | Notification of reasons for refusal |
Free format text: JAPANESE INTERMEDIATE CODE: A131 Effective date: 20141111 |
|
| A521 | Request for written amendment filed |
Free format text: JAPANESE INTERMEDIATE CODE: A523 Effective date: 20150109 |
|
| TRDD | Decision of grant or rejection written | ||
| A01 | Written decision to grant a patent or to grant a registration (utility model) |
Free format text: JAPANESE INTERMEDIATE CODE: A01 Effective date: 20150210 |
|
| A61 | First payment of annual fees (during grant procedure) |
Free format text: JAPANESE INTERMEDIATE CODE: A61 Effective date: 20150310 |
|
| R151 | Written notification of patent or utility model registration |
Ref document number: 5713686 Country of ref document: JP Free format text: JAPANESE INTERMEDIATE CODE: R151 |