JP2010036109A - 有機光触媒フィルムの製造方法 - Google Patents
有機光触媒フィルムの製造方法 Download PDFInfo
- Publication number
- JP2010036109A JP2010036109A JP2008201950A JP2008201950A JP2010036109A JP 2010036109 A JP2010036109 A JP 2010036109A JP 2008201950 A JP2008201950 A JP 2008201950A JP 2008201950 A JP2008201950 A JP 2008201950A JP 2010036109 A JP2010036109 A JP 2010036109A
- Authority
- JP
- Japan
- Prior art keywords
- type organic
- organic semiconductor
- film
- substrate
- manufacturing
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Granted
Links
- 238000004519 manufacturing process Methods 0.000 title claims abstract description 39
- 230000001699 photocatalysis Effects 0.000 title claims abstract description 26
- 239000004065 semiconductor Substances 0.000 claims abstract description 138
- 238000010438 heat treatment Methods 0.000 claims abstract description 21
- 239000000758 substrate Substances 0.000 claims description 84
- 239000011941 photocatalyst Substances 0.000 claims description 38
- IEQIEDJGQAUEQZ-UHFFFAOYSA-N phthalocyanine Chemical class N1C(N=C2C3=CC=CC=C3C(N=C3C4=CC=CC=C4C(=N4)N3)=N2)=C(C=CC=C2)C2=C1N=C1C2=CC=CC=C2C4=N1 IEQIEDJGQAUEQZ-UHFFFAOYSA-N 0.000 claims description 23
- 238000000034 method Methods 0.000 claims description 20
- LKKPNUDVOYAOBB-UHFFFAOYSA-N naphthalocyanine Chemical class N1C(N=C2C3=CC4=CC=CC=C4C=C3C(N=C3C4=CC5=CC=CC=C5C=C4C(=N4)N3)=N2)=C(C=C2C(C=CC=C2)=C2)C2=C1N=C1C2=CC3=CC=CC=C3C=C2C4=N1 LKKPNUDVOYAOBB-UHFFFAOYSA-N 0.000 claims description 15
- 239000000463 material Substances 0.000 claims description 14
- XMWRBQBLMFGWIX-UHFFFAOYSA-N C60 fullerene Chemical class C12=C3C(C4=C56)=C7C8=C5C5=C9C%10=C6C6=C4C1=C1C4=C6C6=C%10C%10=C9C9=C%11C5=C8C5=C8C7=C3C3=C7C2=C1C1=C2C4=C6C4=C%10C6=C9C9=C%11C5=C5C8=C3C3=C7C1=C1C2=C4C6=C2C9=C5C3=C12 XMWRBQBLMFGWIX-UHFFFAOYSA-N 0.000 claims description 13
- 229920001940 conductive polymer Polymers 0.000 claims description 10
- 125000002080 perylenyl group Chemical group C1(=CC=C2C=CC=C3C4=CC=CC5=CC=CC(C1=C23)=C45)* 0.000 claims description 10
- 230000015572 biosynthetic process Effects 0.000 claims description 9
- 150000004033 porphyrin derivatives Chemical class 0.000 claims description 9
- 229910003472 fullerene Inorganic materials 0.000 claims description 8
- 150000002790 naphthalenes Chemical class 0.000 claims description 8
- 238000001771 vacuum deposition Methods 0.000 claims description 8
- OKTJSMMVPCPJKN-UHFFFAOYSA-N Carbon Chemical compound [C] OKTJSMMVPCPJKN-UHFFFAOYSA-N 0.000 claims description 6
- 229920000642 polymer Polymers 0.000 claims description 5
- 239000002041 carbon nanotube Substances 0.000 claims description 3
- 229910021393 carbon nanotube Inorganic materials 0.000 claims description 3
- 239000000203 mixture Substances 0.000 claims description 3
- 230000000694 effects Effects 0.000 abstract description 18
- 239000010410 layer Substances 0.000 description 30
- 229910052751 metal Inorganic materials 0.000 description 25
- 239000002184 metal Substances 0.000 description 25
- 230000000052 comparative effect Effects 0.000 description 24
- 150000001875 compounds Chemical class 0.000 description 18
- 239000011521 glass Substances 0.000 description 16
- 125000004429 atom Chemical group 0.000 description 15
- -1 polyparaphenylene Polymers 0.000 description 13
- BASFCYQUMIYNBI-UHFFFAOYSA-N platinum Substances [Pt] BASFCYQUMIYNBI-UHFFFAOYSA-N 0.000 description 12
- 239000011701 zinc Substances 0.000 description 12
- 125000003118 aryl group Chemical group 0.000 description 10
- 239000003446 ligand Substances 0.000 description 10
- DGVVWUTYPXICAM-UHFFFAOYSA-N β‐Mercaptoethanol Chemical compound OCCS DGVVWUTYPXICAM-UHFFFAOYSA-N 0.000 description 10
- YXFVVABEGXRONW-UHFFFAOYSA-N Toluene Chemical compound CC1=CC=CC=C1 YXFVVABEGXRONW-UHFFFAOYSA-N 0.000 description 9
- 125000000217 alkyl group Chemical group 0.000 description 9
- 229910052697 platinum Inorganic materials 0.000 description 9
- 229910052725 zinc Inorganic materials 0.000 description 9
- 239000010949 copper Substances 0.000 description 8
- 238000002484 cyclic voltammetry Methods 0.000 description 8
- 229910052741 iridium Inorganic materials 0.000 description 8
- 238000010030 laminating Methods 0.000 description 8
- 239000000243 solution Substances 0.000 description 8
- BQCADISMDOOEFD-UHFFFAOYSA-N Silver Chemical compound [Ag] BQCADISMDOOEFD-UHFFFAOYSA-N 0.000 description 7
- QVGXLLKOCUKJST-UHFFFAOYSA-N atomic oxygen Chemical compound [O] QVGXLLKOCUKJST-UHFFFAOYSA-N 0.000 description 7
- 210000004027 cell Anatomy 0.000 description 7
- 238000006243 chemical reaction Methods 0.000 description 7
- 229910052759 nickel Inorganic materials 0.000 description 7
- PXHVJJICTQNCMI-UHFFFAOYSA-N nickel Substances [Ni] PXHVJJICTQNCMI-UHFFFAOYSA-N 0.000 description 7
- 239000001301 oxygen Substances 0.000 description 7
- 229910052760 oxygen Inorganic materials 0.000 description 7
- 229910052763 palladium Inorganic materials 0.000 description 7
- KDLHZDBZIXYQEI-UHFFFAOYSA-N palladium Substances [Pd] KDLHZDBZIXYQEI-UHFFFAOYSA-N 0.000 description 7
- 230000000737 periodic effect Effects 0.000 description 7
- 229910052709 silver Inorganic materials 0.000 description 7
- 239000004332 silver Substances 0.000 description 7
- HCHKCACWOHOZIP-UHFFFAOYSA-N Zinc Chemical compound [Zn] HCHKCACWOHOZIP-UHFFFAOYSA-N 0.000 description 6
- 238000001720 action spectrum Methods 0.000 description 6
- 239000003054 catalyst Substances 0.000 description 6
- 238000000576 coating method Methods 0.000 description 6
- 150000004696 coordination complex Chemical class 0.000 description 6
- 239000003792 electrolyte Substances 0.000 description 6
- 229920000123 polythiophene Polymers 0.000 description 6
- XLYOFNOQVPJJNP-UHFFFAOYSA-N water Substances O XLYOFNOQVPJJNP-UHFFFAOYSA-N 0.000 description 6
- 229910021607 Silver chloride Inorganic materials 0.000 description 5
- 239000007864 aqueous solution Substances 0.000 description 5
- 239000011248 coating agent Substances 0.000 description 5
- 229910052802 copper Inorganic materials 0.000 description 5
- 238000005259 measurement Methods 0.000 description 5
- 238000007254 oxidation reaction Methods 0.000 description 5
- HKZLPVFGJNLROG-UHFFFAOYSA-M silver monochloride Chemical compound [Cl-].[Ag+] HKZLPVFGJNLROG-UHFFFAOYSA-M 0.000 description 5
- CXWXQJXEFPUFDZ-UHFFFAOYSA-N tetralin Chemical compound C1=CC=C2CCCCC2=C1 CXWXQJXEFPUFDZ-UHFFFAOYSA-N 0.000 description 5
- CSCPPACGZOOCGX-UHFFFAOYSA-N Acetone Chemical compound CC(C)=O CSCPPACGZOOCGX-UHFFFAOYSA-N 0.000 description 4
- WCUXLLCKKVVCTQ-UHFFFAOYSA-M Potassium chloride Chemical compound [Cl-].[K+] WCUXLLCKKVVCTQ-UHFFFAOYSA-M 0.000 description 4
- MCEWYIDBDVPMES-UHFFFAOYSA-N [60]pcbm Chemical compound C123C(C4=C5C6=C7C8=C9C%10=C%11C%12=C%13C%14=C%15C%16=C%17C%18=C(C=%19C=%20C%18=C%18C%16=C%13C%13=C%11C9=C9C7=C(C=%20C9=C%13%18)C(C7=%19)=C96)C6=C%11C%17=C%15C%13=C%15C%14=C%12C%12=C%10C%10=C85)=C9C7=C6C2=C%11C%13=C2C%15=C%12C%10=C4C23C1(CCCC(=O)OC)C1=CC=CC=C1 MCEWYIDBDVPMES-UHFFFAOYSA-N 0.000 description 4
- 229920006332 epoxy adhesive Polymers 0.000 description 4
- 150000002739 metals Chemical class 0.000 description 4
- 125000002496 methyl group Chemical group [H]C([H])([H])* 0.000 description 4
- 125000001997 phenyl group Chemical group [H]C1=C([H])C([H])=C(*)C([H])=C1[H] 0.000 description 4
- 238000013032 photocatalytic reaction Methods 0.000 description 4
- 229920000301 poly(3-hexylthiophene-2,5-diyl) polymer Polymers 0.000 description 4
- 229920006395 saturated elastomer Polymers 0.000 description 4
- 238000007740 vapor deposition Methods 0.000 description 4
- GWEVSGVZZGPLCZ-UHFFFAOYSA-N Titan oxide Chemical compound O=[Ti]=O GWEVSGVZZGPLCZ-UHFFFAOYSA-N 0.000 description 3
- 229910003077 Ti−O Inorganic materials 0.000 description 3
- 230000008859 change Effects 0.000 description 3
- 125000004093 cyano group Chemical group *C#N 0.000 description 3
- 238000000354 decomposition reaction Methods 0.000 description 3
- 238000000151 deposition Methods 0.000 description 3
- 230000008021 deposition Effects 0.000 description 3
- RBTKNAXYKSUFRK-UHFFFAOYSA-N heliogen blue Chemical compound [Cu].[N-]1C2=C(C=CC=C3)C3=C1N=C([N-]1)C3=CC=CC=C3C1=NC([N-]1)=C(C=CC=C3)C3=C1N=C([N-]1)C3=CC=CC=C3C1=N2 RBTKNAXYKSUFRK-UHFFFAOYSA-N 0.000 description 3
- 125000001072 heteroaryl group Chemical group 0.000 description 3
- 229910052742 iron Inorganic materials 0.000 description 3
- XEEYBQQBJWHFJM-UHFFFAOYSA-N iron Substances [Fe] XEEYBQQBJWHFJM-UHFFFAOYSA-N 0.000 description 3
- 125000000959 isobutyl group Chemical group [H]C([H])([H])C([H])(C([H])([H])[H])C([H])([H])* 0.000 description 3
- 229910052762 osmium Inorganic materials 0.000 description 3
- 230000003647 oxidation Effects 0.000 description 3
- 238000006864 oxidative decomposition reaction Methods 0.000 description 3
- 229920000553 poly(phenylenevinylene) Polymers 0.000 description 3
- 150000004032 porphyrins Chemical class 0.000 description 3
- 229910052703 rhodium Inorganic materials 0.000 description 3
- 229910052707 ruthenium Inorganic materials 0.000 description 3
- 238000004528 spin coating Methods 0.000 description 3
- 239000000126 substance Substances 0.000 description 3
- 230000036962 time dependent Effects 0.000 description 3
- 239000010936 titanium Substances 0.000 description 3
- 125000000008 (C1-C10) alkyl group Chemical group 0.000 description 2
- RYGMFSIKBFXOCR-UHFFFAOYSA-N Copper Chemical compound [Cu] RYGMFSIKBFXOCR-UHFFFAOYSA-N 0.000 description 2
- 229920000265 Polyparaphenylene Polymers 0.000 description 2
- XLOMVQKBTHCTTD-UHFFFAOYSA-N Zinc monoxide Chemical compound [Zn]=O XLOMVQKBTHCTTD-UHFFFAOYSA-N 0.000 description 2
- 239000000956 alloy Substances 0.000 description 2
- 229910045601 alloy Inorganic materials 0.000 description 2
- 239000008346 aqueous phase Substances 0.000 description 2
- 239000012300 argon atmosphere Substances 0.000 description 2
- 239000012298 atmosphere Substances 0.000 description 2
- 125000002619 bicyclic group Chemical group 0.000 description 2
- 229910052799 carbon Inorganic materials 0.000 description 2
- 230000003197 catalytic effect Effects 0.000 description 2
- 210000005056 cell body Anatomy 0.000 description 2
- 239000000919 ceramic Substances 0.000 description 2
- MPMSMUBQXQALQI-UHFFFAOYSA-N cobalt phthalocyanine Chemical compound [Co+2].C12=CC=CC=C2C(N=C2[N-]C(C3=CC=CC=C32)=N2)=NC1=NC([C]1C=CC=CC1=1)=NC=1N=C1[C]3C=CC=CC3=C2[N-]1 MPMSMUBQXQALQI-UHFFFAOYSA-N 0.000 description 2
- 125000001495 ethyl group Chemical group [H]C([H])([H])C([H])([H])* 0.000 description 2
- 239000000835 fiber Substances 0.000 description 2
- 229910052736 halogen Inorganic materials 0.000 description 2
- 150000002367 halogens Chemical class 0.000 description 2
- 125000004435 hydrogen atom Chemical group [H]* 0.000 description 2
- AMGQUBHHOARCQH-UHFFFAOYSA-N indium;oxotin Chemical compound [In].[Sn]=O AMGQUBHHOARCQH-UHFFFAOYSA-N 0.000 description 2
- 125000001449 isopropyl group Chemical group [H]C([H])([H])C([H])(*)C([H])([H])[H] 0.000 description 2
- 125000002950 monocyclic group Chemical group 0.000 description 2
- 125000004108 n-butyl group Chemical group [H]C([H])([H])C([H])([H])C([H])([H])C([H])([H])* 0.000 description 2
- 125000003136 n-heptyl group Chemical group [H]C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])* 0.000 description 2
- 125000001280 n-hexyl group Chemical group C(CCCCC)* 0.000 description 2
- 125000000740 n-pentyl group Chemical group [H]C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])* 0.000 description 2
- 125000004123 n-propyl group Chemical group [H]C([H])([H])C([H])([H])C([H])([H])* 0.000 description 2
- 125000001624 naphthyl group Chemical group 0.000 description 2
- 229910052757 nitrogen Inorganic materials 0.000 description 2
- 125000004433 nitrogen atom Chemical group N* 0.000 description 2
- 230000003287 optical effect Effects 0.000 description 2
- 239000000123 paper Substances 0.000 description 2
- 229920002098 polyfluorene Polymers 0.000 description 2
- 239000001103 potassium chloride Substances 0.000 description 2
- 235000011164 potassium chloride Nutrition 0.000 description 2
- 125000002914 sec-butyl group Chemical group [H]C([H])([H])C([H])([H])C([H])(*)C([H])([H])[H] 0.000 description 2
- 239000002904 solvent Substances 0.000 description 2
- 238000000859 sublimation Methods 0.000 description 2
- 230000008022 sublimation Effects 0.000 description 2
- 125000001424 substituent group Chemical group 0.000 description 2
- OGIDPMRJRNCKJF-UHFFFAOYSA-N titanium oxide Inorganic materials [Ti]=O OGIDPMRJRNCKJF-UHFFFAOYSA-N 0.000 description 2
- 229910052723 transition metal Inorganic materials 0.000 description 2
- 150000003624 transition metals Chemical class 0.000 description 2
- 239000002023 wood Substances 0.000 description 2
- GEYOCULIXLDCMW-UHFFFAOYSA-N 1,2-phenylenediamine Chemical compound NC1=CC=CC=C1N GEYOCULIXLDCMW-UHFFFAOYSA-N 0.000 description 1
- KMHSUNDEGHRBNV-UHFFFAOYSA-N 2,4-dichloropyrimidine-5-carbonitrile Chemical compound ClC1=NC=C(C#N)C(Cl)=N1 KMHSUNDEGHRBNV-UHFFFAOYSA-N 0.000 description 1
- BZSVVCFHMVMYCR-UHFFFAOYSA-N 2-pyridin-2-ylpyridine;ruthenium Chemical compound [Ru].N1=CC=CC=C1C1=CC=CC=N1.N1=CC=CC=C1C1=CC=CC=N1.N1=CC=CC=C1C1=CC=CC=N1 BZSVVCFHMVMYCR-UHFFFAOYSA-N 0.000 description 1
- XQNMSKCVXVXEJT-UHFFFAOYSA-N 7,14,25,32-tetrazaundecacyclo[21.13.2.22,5.03,19.04,16.06,14.08,13.020,37.024,32.026,31.034,38]tetraconta-1(36),2,4,6,8,10,12,16,18,20(37),21,23(38),24,26,28,30,34,39-octadecaene-15,33-dione 7,14,25,32-tetrazaundecacyclo[21.13.2.22,5.03,19.04,16.06,14.08,13.020,37.025,33.026,31.034,38]tetraconta-1(37),2,4,6,8,10,12,16,18,20,22,26,28,30,32,34(38),35,39-octadecaene-15,24-dione Chemical compound O=c1c2ccc3c4ccc5c6nc7ccccc7n6c(=O)c6ccc(c7ccc(c8nc9ccccc9n18)c2c37)c4c56.O=c1c2ccc3c4ccc5c6c(ccc(c7ccc(c8nc9ccccc9n18)c2c37)c46)c1nc2ccccc2n1c5=O XQNMSKCVXVXEJT-UHFFFAOYSA-N 0.000 description 1
- 239000004642 Polyimide Substances 0.000 description 1
- JUJWROOIHBZHMG-UHFFFAOYSA-N Pyridine Chemical group C1=CC=NC=C1 JUJWROOIHBZHMG-UHFFFAOYSA-N 0.000 description 1
- NVJHHSJKESILSZ-UHFFFAOYSA-N [Co].N1C(C=C2N=C(C=C3NC(=C4)C=C3)C=C2)=CC=C1C=C1C=CC4=N1 Chemical compound [Co].N1C(C=C2N=C(C=C3NC(=C4)C=C3)C=C2)=CC=C1C=C1C=CC4=N1 NVJHHSJKESILSZ-UHFFFAOYSA-N 0.000 description 1
- NUSORQHHEXCNQC-UHFFFAOYSA-N [Cu].N1C(C=C2N=C(C=C3NC(=C4)C=C3)C=C2)=CC=C1C=C1C=CC4=N1 Chemical compound [Cu].N1C(C=C2N=C(C=C3NC(=C4)C=C3)C=C2)=CC=C1C=C1C=CC4=N1 NUSORQHHEXCNQC-UHFFFAOYSA-N 0.000 description 1
- YIYFFLYGSHJWFF-UHFFFAOYSA-N [Zn].N1C(C=C2N=C(C=C3NC(=C4)C=C3)C=C2)=CC=C1C=C1C=CC4=N1 Chemical compound [Zn].N1C(C=C2N=C(C=C3NC(=C4)C=C3)C=C2)=CC=C1C=C1C=CC4=N1 YIYFFLYGSHJWFF-UHFFFAOYSA-N 0.000 description 1
- 238000010521 absorption reaction Methods 0.000 description 1
- 239000012790 adhesive layer Substances 0.000 description 1
- 230000002411 adverse Effects 0.000 description 1
- 238000004458 analytical method Methods 0.000 description 1
- 238000005513 bias potential Methods 0.000 description 1
- 125000002915 carbonyl group Chemical group [*:2]C([*:1])=O 0.000 description 1
- 231100000481 chemical toxicant Toxicity 0.000 description 1
- 229910017052 cobalt Inorganic materials 0.000 description 1
- 239000010941 cobalt Substances 0.000 description 1
- GUTLYIVDDKVIGB-UHFFFAOYSA-N cobalt atom Chemical compound [Co] GUTLYIVDDKVIGB-UHFFFAOYSA-N 0.000 description 1
- 239000004567 concrete Substances 0.000 description 1
- 150000001923 cyclic compounds Chemical class 0.000 description 1
- 230000007423 decrease Effects 0.000 description 1
- 229910003460 diamond Inorganic materials 0.000 description 1
- 239000010432 diamond Substances 0.000 description 1
- 238000004070 electrodeposition Methods 0.000 description 1
- 238000005868 electrolysis reaction Methods 0.000 description 1
- 125000006575 electron-withdrawing group Chemical group 0.000 description 1
- 239000010419 fine particle Substances 0.000 description 1
- 239000002803 fossil fuel Substances 0.000 description 1
- 239000007789 gas Substances 0.000 description 1
- 229910021397 glassy carbon Inorganic materials 0.000 description 1
- 229910002804 graphite Inorganic materials 0.000 description 1
- 239000010439 graphite Substances 0.000 description 1
- 125000004051 hexyl group Chemical group [H]C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])* 0.000 description 1
- 125000002883 imidazolyl group Chemical group 0.000 description 1
- 230000006872 improvement Effects 0.000 description 1
- 229910052738 indium Inorganic materials 0.000 description 1
- APFVFJFRJDLVQX-UHFFFAOYSA-N indium atom Chemical compound [In] APFVFJFRJDLVQX-UHFFFAOYSA-N 0.000 description 1
- 150000002484 inorganic compounds Chemical class 0.000 description 1
- 229910010272 inorganic material Inorganic materials 0.000 description 1
- 238000003475 lamination Methods 0.000 description 1
- 239000007791 liquid phase Substances 0.000 description 1
- 239000011259 mixed solution Substances 0.000 description 1
- UFWIBTONFRDIAS-UHFFFAOYSA-N naphthalene-acid Natural products C1=CC=CC2=CC=CC=C21 UFWIBTONFRDIAS-UHFFFAOYSA-N 0.000 description 1
- QGLKJKCYBOYXKC-UHFFFAOYSA-N nonaoxidotritungsten Chemical compound O=[W]1(=O)O[W](=O)(=O)O[W](=O)(=O)O1 QGLKJKCYBOYXKC-UHFFFAOYSA-N 0.000 description 1
- 125000002347 octyl group Chemical group [H]C([*])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])[H] 0.000 description 1
- 150000002894 organic compounds Chemical class 0.000 description 1
- 239000005416 organic matter Substances 0.000 description 1
- 239000003960 organic solvent Substances 0.000 description 1
- 230000001590 oxidative effect Effects 0.000 description 1
- 125000004430 oxygen atom Chemical group O* 0.000 description 1
- 239000002245 particle Substances 0.000 description 1
- 150000002979 perylenes Chemical class 0.000 description 1
- CSHWQDPOILHKBI-UHFFFAOYSA-N peryrene Natural products C1=CC(C2=CC=CC=3C2=C2C=CC=3)=C3C2=CC=CC3=C1 CSHWQDPOILHKBI-UHFFFAOYSA-N 0.000 description 1
- 239000012071 phase Substances 0.000 description 1
- 238000005191 phase separation Methods 0.000 description 1
- 238000007539 photo-oxidation reaction Methods 0.000 description 1
- 230000001443 photoexcitation Effects 0.000 description 1
- 229920001721 polyimide Polymers 0.000 description 1
- 229920000128 polypyrrole Polymers 0.000 description 1
- 125000003373 pyrazinyl group Chemical group 0.000 description 1
- 125000004076 pyridyl group Chemical group 0.000 description 1
- 125000000168 pyrrolyl group Chemical group 0.000 description 1
- 125000001567 quinoxalinyl group Chemical group N1=C(C=NC2=CC=CC=C12)* 0.000 description 1
- 238000001228 spectrum Methods 0.000 description 1
- 238000004544 sputter deposition Methods 0.000 description 1
- 125000001174 sulfone group Chemical group 0.000 description 1
- 125000003375 sulfoxide group Chemical group 0.000 description 1
- XXWSZMQKQLNDLC-UHFFFAOYSA-N tetraaminoethylene Chemical group NC(N)=C(N)N XXWSZMQKQLNDLC-UHFFFAOYSA-N 0.000 description 1
- XOLBLPGZBRYERU-UHFFFAOYSA-N tin dioxide Chemical compound O=[Sn]=O XOLBLPGZBRYERU-UHFFFAOYSA-N 0.000 description 1
- 229910001887 tin oxide Inorganic materials 0.000 description 1
- 239000003440 toxic substance Substances 0.000 description 1
- 230000007704 transition Effects 0.000 description 1
- 238000002834 transmittance Methods 0.000 description 1
- 229910001930 tungsten oxide Inorganic materials 0.000 description 1
- 239000011787 zinc oxide Substances 0.000 description 1
Images
Classifications
-
- Y—GENERAL TAGGING OF NEW TECHNOLOGICAL DEVELOPMENTS; GENERAL TAGGING OF CROSS-SECTIONAL TECHNOLOGIES SPANNING OVER SEVERAL SECTIONS OF THE IPC; TECHNICAL SUBJECTS COVERED BY FORMER USPC CROSS-REFERENCE ART COLLECTIONS [XRACs] AND DIGESTS
- Y02—TECHNOLOGIES OR APPLICATIONS FOR MITIGATION OR ADAPTATION AGAINST CLIMATE CHANGE
- Y02E—REDUCTION OF GREENHOUSE GAS [GHG] EMISSIONS, RELATED TO ENERGY GENERATION, TRANSMISSION OR DISTRIBUTION
- Y02E10/00—Energy generation through renewable energy sources
- Y02E10/50—Photovoltaic [PV] energy
- Y02E10/549—Organic PV cells
-
- Y—GENERAL TAGGING OF NEW TECHNOLOGICAL DEVELOPMENTS; GENERAL TAGGING OF CROSS-SECTIONAL TECHNOLOGIES SPANNING OVER SEVERAL SECTIONS OF THE IPC; TECHNICAL SUBJECTS COVERED BY FORMER USPC CROSS-REFERENCE ART COLLECTIONS [XRACs] AND DIGESTS
- Y02—TECHNOLOGIES OR APPLICATIONS FOR MITIGATION OR ADAPTATION AGAINST CLIMATE CHANGE
- Y02P—CLIMATE CHANGE MITIGATION TECHNOLOGIES IN THE PRODUCTION OR PROCESSING OF GOODS
- Y02P70/00—Climate change mitigation technologies in the production process for final industrial or consumer products
- Y02P70/50—Manufacturing or production processes characterised by the final manufactured product
Landscapes
- Photovoltaic Devices (AREA)
- Hybrid Cells (AREA)
- Catalysts (AREA)
Abstract
【解決手段】p型有機半導体とn型有機半導体とを含む有機光触媒フィルムの製造方法であって、成膜時又は成膜後に加熱することを特徴とする製造方法。
【選択図】図4
Description
で表されるフタロシアニン誘導体が挙げられる。
で表されるナフタロシアニン誘導体が挙げられる。
で表されるポルフィリン誘導体が挙げられる。
で表されるペリレン誘導体が挙げられる。
で表されるナフタレン誘導体が挙げられる。
で表されるポリチオフェン誘導体が挙げられる。
で表されるフラーレン誘導体が挙げられる。
有機光触媒材料として、n型有機半導体である3,4,9,10−ペリレンテトラカルボキシル−ビスベンズイミダゾール(以下「PTCBI」と表記する)及びp型有機半導体であるフタロシアニン(以下「H2Pc」と表記する)を用いた。本発明においては、それぞれ昇華精製したものを用いた。
実施例1,2又は比較例1の有機光触媒被覆電極を用いた一室型三極式セルについて、それぞれ、電解質として、1mM 2−メルカプトエタノール水溶液(pH=10)を用い、常温、常圧下、電位走査速度を20mV/秒として、−0.2V〜+0.6Vの範囲でCV測定を行った。実施例1,2の結果を図2に、比較例1の結果を図3に示す。
実施例1,2又は比較例1の有機光触媒被覆電極を用いた一室型三極式セルについて、それぞれ、電解質として、5mM 2−メルカプトエタノール水溶液(pH=10)を用い、アルゴン雰囲気下、常温、常圧で、印加電位は0V(vs.Ag/AgCl)とし、作用スペクトルとして、400〜750nmのIPCE(各波長における入射光子数あたりの反応分子数)をモノクロメーターにより測定した。その結果を図4に示す。
有機光触媒材料として、n型有機半導体である[6,6]−フェニル−C61酪酸メチルエステル(以下「PCBM」と表記する)及びp型有機半導体であるポリ(3−ヘキシルチオフェン−2,5−ジイル)(以下「P3HT」と表記する)を用いた。実施例4においては、p型有機半導体である亜鉛フタロシアニン(以下「ZnPc」と表記する)を用いた。本発明においては、PCBM及びP3HTは市販のものをそのまま使用し、ZnPcは昇華精製したものを用いた。
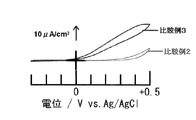
実施例3、4、比較例2又は3の有機光触媒被覆電極を用いた一室型三極式セルについて、それぞれ、常温、常圧下、電位走査速度を20mV/秒として、−0.3V〜+0.5Vの範囲でCV測定を行った。実施例3及び4の結果を図5に、比較例2及び3の結果を図6に示す。
実施例3,4、比較例2又は3の有機光触媒被覆電極を用いた一室型三極式セルについて、それぞれ、アルゴン雰囲気下、常温、常圧で、印加電位は0.3V(vs.Ag/AgCl)とし、作用スペクトルとして、400〜750nmのIPCEをモノクロメーターにより測定した。その結果を図7に示す。
混合フィルムの加熱効果に関する情報を得ることを目的として、2−メルカプトエタノールの光触媒的酸化反応に伴うアノード光電流値の経時変化を実施例3及び比較例2の電極について測定した。測定条件は、印加電位0.3V(vs.Ag/AgCl)で、ITO被覆ガラス基板側から白色光を100mW/cm2の光量で照射した。実施例3の結果を図8(a)に、比較例2の結果を図8(b)に示す。
Claims (14)
- p型有機半導体とn型有機半導体とを含む有機光触媒フィルムの製造方法であって、成膜時又は成膜後に加熱することを特徴とする製造方法。
- 前記有機光触媒フィルムがn型有機半導体の上にp型有機半導体が積層された二層膜であって、n型有機半導体が加熱された状態でp型有機半導体を積層する、請求項1に記載の製造方法。
- 真空蒸着法を用い、真空度が4.0×10−3Pa未満の条件で、室温下にある基体の上に前記n型有機半導体を積層した後、基体の温度を40〜150℃に制御することにより加熱されたn型有機半導体層の上にp型有機半導体を積層する、請求項2に記載の製造方法。
- 前記基体の温度を90〜120℃に制御する、請求項3に記載の製造方法。
- 前記真空度が5.0×10−4Paの条件で、前記基体の温度を120℃に制御する、請求項4に記載の製造方法。
- 前記p型有機半導体が、フタロシアニン誘導体、ナフタロシアニン誘導体、ポルフィリン誘導体及び導電性高分子からなる群から選ばれる少なくとも1種である、請求項1〜5のいずれか1項に記載の製造方法。
- 前記n型有機半導体が、フラーレン類、カーボンナノチューブ類、電子供与体をドープした導電性高分子、ペリレン誘導体、及びナフタレン誘導体からなる群から選ばれる少なくとも1種である、請求項1〜6のいずれか1項に記載の製造方法。
- 前記n型有機半導体とp型有機半導体との混合物を成膜した後、真空度が0.1Pa未満の条件で、50〜160℃に加熱する、請求項1に記載の製造方法。
- 前記n型有機半導体及びp型有機半導体を含む溶液を基体上に滴下し、該基体をスピンさせて成膜し、その後、真空度が0.1Pa未満の条件で、50〜160℃に加熱する、請求項8に記載の製造方法。
- 前記真空度が7.0×10−2Paの条件で、前記温度が95〜105℃である、請求項9に記載の製造方法。
- 前記n型有機半導体が低分子半導体であって、前記p型有機半導体が高分子半導体である、請求項8〜10のいずれか1項に記載の製造方法。
- 前記n型有機半導体がフラーレン誘導体であって、前記p型有機半導体が導電性高分子である、請求項11に記載の製造方法。
- 請求項1〜12のいずれか1項に記載の製造方法により得られた有機光触媒フィルム。
- 電極基材の表面に、請求項13に記載の有機光触媒フィルムを被覆してなる電極。
Priority Applications (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| JP2008201950A JP5544691B2 (ja) | 2008-08-05 | 2008-08-05 | 有機光触媒フィルムの製造方法 |
Applications Claiming Priority (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| JP2008201950A JP5544691B2 (ja) | 2008-08-05 | 2008-08-05 | 有機光触媒フィルムの製造方法 |
Publications (2)
| Publication Number | Publication Date |
|---|---|
| JP2010036109A true JP2010036109A (ja) | 2010-02-18 |
| JP5544691B2 JP5544691B2 (ja) | 2014-07-09 |
Family
ID=42009203
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| JP2008201950A Expired - Fee Related JP5544691B2 (ja) | 2008-08-05 | 2008-08-05 | 有機光触媒フィルムの製造方法 |
Country Status (1)
| Country | Link |
|---|---|
| JP (1) | JP5544691B2 (ja) |
Cited By (4)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JP2011147849A (ja) * | 2010-01-19 | 2011-08-04 | Tokyo Institute Of Technology | p−n接合型粒子、及びその製造方法、並びに有機光触媒 |
| WO2016132750A1 (ja) * | 2015-02-18 | 2016-08-25 | 株式会社村田製作所 | 光二次電池、及び光二次電池用電極の製造方法 |
| WO2016132749A1 (ja) * | 2015-02-18 | 2016-08-25 | 株式会社村田製作所 | 光二次電池 |
| CN106362794A (zh) * | 2016-08-08 | 2017-02-01 | 上海师范大学 | N,n‑二(2,6‑异丙基苯基)‑3,4,9,10‑苝四羧酸酐的用途 |
Citations (9)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JPH01119662A (ja) * | 1987-10-30 | 1989-05-11 | Sumitomo Electric Ind Ltd | 有機物薄膜の製造法 |
| JPH01255660A (ja) * | 1988-04-06 | 1989-10-12 | Agency Of Ind Science & Technol | シリコンフタロシアニンポリマー薄膜の製法 |
| JPH0774377A (ja) * | 1993-08-31 | 1995-03-17 | Kawamura Inst Of Chem Res | 光電変換素子 |
| JPH10226883A (ja) * | 1997-02-18 | 1998-08-25 | Ricoh Co Ltd | フタロシアニン結晶質薄膜の作製法 |
| JP2000033265A (ja) * | 1998-07-17 | 2000-02-02 | Chisso Corp | オレフィン及び芳香族炭素化合物の選択酸化光触媒、並びにそれを用いた含酸素化合物の製造方法 |
| WO2005063393A1 (ja) * | 2003-12-26 | 2005-07-14 | Kansai Technology Licensing Organization Co., Ltd. | 有機光触媒を用いた水の電気分解方法 |
| JP2005203659A (ja) * | 2004-01-19 | 2005-07-28 | Sony Corp | 光電変換材料およびその製造方法ならびに光電変換素子およびその製造方法ならびに電子装置およびその製造方法 |
| WO2006115271A1 (ja) * | 2005-04-26 | 2006-11-02 | Osaka University | 有機光触媒 |
| JP2007273939A (ja) * | 2005-09-06 | 2007-10-18 | Kyoto Univ | 有機薄膜光電変換素子及びその製造方法 |
-
2008
- 2008-08-05 JP JP2008201950A patent/JP5544691B2/ja not_active Expired - Fee Related
Patent Citations (9)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JPH01119662A (ja) * | 1987-10-30 | 1989-05-11 | Sumitomo Electric Ind Ltd | 有機物薄膜の製造法 |
| JPH01255660A (ja) * | 1988-04-06 | 1989-10-12 | Agency Of Ind Science & Technol | シリコンフタロシアニンポリマー薄膜の製法 |
| JPH0774377A (ja) * | 1993-08-31 | 1995-03-17 | Kawamura Inst Of Chem Res | 光電変換素子 |
| JPH10226883A (ja) * | 1997-02-18 | 1998-08-25 | Ricoh Co Ltd | フタロシアニン結晶質薄膜の作製法 |
| JP2000033265A (ja) * | 1998-07-17 | 2000-02-02 | Chisso Corp | オレフィン及び芳香族炭素化合物の選択酸化光触媒、並びにそれを用いた含酸素化合物の製造方法 |
| WO2005063393A1 (ja) * | 2003-12-26 | 2005-07-14 | Kansai Technology Licensing Organization Co., Ltd. | 有機光触媒を用いた水の電気分解方法 |
| JP2005203659A (ja) * | 2004-01-19 | 2005-07-28 | Sony Corp | 光電変換材料およびその製造方法ならびに光電変換素子およびその製造方法ならびに電子装置およびその製造方法 |
| WO2006115271A1 (ja) * | 2005-04-26 | 2006-11-02 | Osaka University | 有機光触媒 |
| JP2007273939A (ja) * | 2005-09-06 | 2007-10-18 | Kyoto Univ | 有機薄膜光電変換素子及びその製造方法 |
Non-Patent Citations (1)
| Title |
|---|
| JPN6012055551; 芝田岳史 他: '導電性高分子/C60相互浸透型光電変換素子の基板加熱による効率改善' 2007年(平成19年)秋季第68回応用物理学会学術講演会講演予稿集第3分冊 , 20070904, Page1260, (社)応用物理学会 * |
Cited By (4)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JP2011147849A (ja) * | 2010-01-19 | 2011-08-04 | Tokyo Institute Of Technology | p−n接合型粒子、及びその製造方法、並びに有機光触媒 |
| WO2016132750A1 (ja) * | 2015-02-18 | 2016-08-25 | 株式会社村田製作所 | 光二次電池、及び光二次電池用電極の製造方法 |
| WO2016132749A1 (ja) * | 2015-02-18 | 2016-08-25 | 株式会社村田製作所 | 光二次電池 |
| CN106362794A (zh) * | 2016-08-08 | 2017-02-01 | 上海师范大学 | N,n‑二(2,6‑异丙基苯基)‑3,4,9,10‑苝四羧酸酐的用途 |
Also Published As
| Publication number | Publication date |
|---|---|
| JP5544691B2 (ja) | 2014-07-09 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| Han et al. | Highly ordered N‐doped carbon dots photosensitizer on metal–organic framework‐decorated ZnO nanotubes for improved photoelectrochemical water splitting | |
| Safaei et al. | Graphitic carbon nitride (gC 3 N 4) electrodes for energy conversion and storage: a review on photoelectrochemical water splitting, solar cells and supercapacitors | |
| Son et al. | Quinone and its derivatives for energy harvesting and storage materials | |
| Sherman et al. | Light-driven water splitting with a molecular electroassembly-based core/shell photoanode | |
| Han et al. | Reduced graphene oxide/mesoporous TiO2 nanocomposite based perovskite solar cells | |
| Ou et al. | Surface-supported metal–organic framework thin-film-derived transparent CoS1. 097@ N-doped carbon film as an efficient counter electrode for bifacial dye-sensitized solar cells | |
| Suzuki et al. | Application of carbon nanotubes to counter electrodes of dye-sensitized solar cells | |
| Brisse et al. | Inkjet printing NiO-based p-type dye-sensitized solar cells | |
| Luo et al. | Highly efficient metal-free sulfur-doped and nitrogen and sulfur dual-doped reduced graphene oxide counter electrodes for dye-sensitized solar cells | |
| Shan et al. | Modulating hole transport in multilayered photocathodes with derivatized p-type nickel oxide and molecular assemblies for solar-driven water splitting | |
| Chen et al. | Porphyrin-based conjugated polymers as intrinsic semiconducting photocatalysts for robust H2 generation under visible light | |
| Shahiduzzaman et al. | Compact TiO2/Anatase TiO2 single-crystalline nanoparticle electron-transport bilayer for efficient planar perovskite solar cells | |
| JP3995051B2 (ja) | 有機光触媒を用いた水の電気分解方法 | |
| TW200915641A (en) | Process for producing electroconductive polymer electrode and dye-sensitized solar cell comprising the electroconductive polymer electrode | |
| Kavan | Exploiting nanocarbons in dye-sensitized solar cells | |
| Gullace et al. | A platinum-free nanostructured gold counter electrode for DSSCs prepared by pulsed laser ablation | |
| Hilmi et al. | Universal low-temperature MWCNT-COOH-based counter electrode and a new thiolate/disulfide electrolyte system for dye-sensitized solar cells | |
| Ou et al. | Characterization of ZnO interlayers for organic solar cells: correlation of electrochemical properties with thin-film morphology and device performance | |
| Park et al. | Boosting Charge Transfer Efficiency by Nanofragment MXene for Efficient Photoelectrochemical Water Splitting of NiFe (OH) x Co-Catalyzed Hematite | |
| Park et al. | Stable organic-inorganic hybrid multilayered photoelectrochemical cells | |
| JP5544691B2 (ja) | 有機光触媒フィルムの製造方法 | |
| Zhao et al. | Molecular engineering of photocathodes based on polythiophene organic semiconductors for photoelectrochemical hydrogen generation | |
| Jiang et al. | Super-hydrophilic electrode encapsulated lead halide-perovskite photoanode toward stable and efficient photoelectrochemical water splitting | |
| Ji et al. | Ligand doping engineering induced robust internal electric field in MOFs/BiVO4 photoanode for water splitting | |
| JP5424594B2 (ja) | 有機光応答素子用反応セル |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| A621 | Written request for application examination |
Free format text: JAPANESE INTERMEDIATE CODE: A621 Effective date: 20110803 |
|
| A977 | Report on retrieval |
Free format text: JAPANESE INTERMEDIATE CODE: A971007 Effective date: 20120906 |
|
| A131 | Notification of reasons for refusal |
Free format text: JAPANESE INTERMEDIATE CODE: A131 Effective date: 20121030 |
|
| A521 | Request for written amendment filed |
Free format text: JAPANESE INTERMEDIATE CODE: A523 Effective date: 20121221 |
|
| A131 | Notification of reasons for refusal |
Free format text: JAPANESE INTERMEDIATE CODE: A131 Effective date: 20130723 |
|
| A521 | Request for written amendment filed |
Free format text: JAPANESE INTERMEDIATE CODE: A523 Effective date: 20130912 |
|
| TRDD | Decision of grant or rejection written | ||
| A01 | Written decision to grant a patent or to grant a registration (utility model) |
Free format text: JAPANESE INTERMEDIATE CODE: A01 Effective date: 20140408 |
|
| A61 | First payment of annual fees (during grant procedure) |
Free format text: JAPANESE INTERMEDIATE CODE: A61 Effective date: 20140428 |
|
| R150 | Certificate of patent or registration of utility model |
Ref document number: 5544691 Country of ref document: JP Free format text: JAPANESE INTERMEDIATE CODE: R150 |
|
| R250 | Receipt of annual fees |
Free format text: JAPANESE INTERMEDIATE CODE: R250 |
|
| R250 | Receipt of annual fees |
Free format text: JAPANESE INTERMEDIATE CODE: R250 |
|
| R250 | Receipt of annual fees |
Free format text: JAPANESE INTERMEDIATE CODE: R250 |
|
| R250 | Receipt of annual fees |
Free format text: JAPANESE INTERMEDIATE CODE: R250 |
|
| R250 | Receipt of annual fees |
Free format text: JAPANESE INTERMEDIATE CODE: R250 |
|
| LAPS | Cancellation because of no payment of annual fees |